Chapter 20 Otoscopy
Video Otoscopy
Ear diseases are one of the most commonly encountered conditions for which clients seek veterinary care from the veterinary practitioner and veterinary dermatologist. These diseases frequently pose diagnostic and therapeutic challenges to the veterinarian and can be very frustrating conditions for the clients to treat. Video otoscopy has revolutionized the practice of veterinary otology. It allows for better visualization of the ear, which aids in the diagnosis and management of the ear disease, makes possible digital documentation of the ear disease, and is instrumental in educating clients about ear disease.
Indications
In the past decade, video otoscopy has become more widely utilized for the evaluation and treatment of diseases of the external ear canal and middle ear. Indications for the use of the handheld otoscope still exist; however, the video otoscope has the advantages of superior magnification and lighting, complete visualization of the ear canal and tympanic membrane when instruments (e.g., biopsy forceps, grasping forceps, curettes, and catheters) are used through the instrument channel, and digital capture capability to document the ear disease.
Most video otoscopes are much more durable than the traditional rigid endoscopes; therefore, they can be used in the awake animal in the examination room, as well as in the sedated or anesthetized animal, so they are very versatile. Examination of the external ear canal and tympanic membrane should be part of the general examination, regardless of whether clients complain of any ear disease in their pets. It is important for veterinarians to become familiar with the normal structures in the ear so that they are then able to determine whether an abnormality exists.
The video otoscope is one of the few pieces of equipment that can be used in the examination room and that can allow the client to visualize the ear canal and tympanic membrane along with the veterinarian. Direct visualization of the ear and any abnormalities present will improve the client’s compliance with prescribed therapies and on reexamination offers positive reinforcement for the treatments that were performed. It is an invaluable piece of equipment to use when the ear canal and tympanic membrane are evaluated in a patient with otitis. The most common complaints that clients may have when their pets have ear disease are head shaking, ear scratching, otic discharge, foul odor from the ear, and pain when the ear is manipulated. In some instances, neurologic signs, such as facial nerve paralysis, Horner’s syndrome, vestibular disturbances, or deafness, may be present and may indicate the need to evaluate the patient for middle or inner ear disease.
The initial diagnostic plan for a patient with acute otitis externa should include cytologic analysis of otic exudates in addition to the otic examination so that it can be determined whether an infective agent is present in the ear canal and, if so, what initial treatments should be recommended. In patients with chronic, recurrent otitis externa, in addition to cytologic analysis of otic exudates and an otic examination, diagnostics are needed to determine the cause of the recurrent otitis and to evaluate the patient for concurrent otitis media. These may include, but are not limited to, bacterial culture and sensitivity testing (C/S), a food trial, allergy testing, blood work, biopsies, radiographic imaging of the bulla, deep ear flushing, and myringotomy.
Additionally, in patients with chronic otitis externa, the predisposing factors, primary causes, and perpetuating factors must be identified to control the ear disease. Primary causes of otitis externa are those that result in ear disease in the absence of other factors. Primary causes include parasitic diseases, hypersensitivity disorders, foreign bodies, disorders of keratinization, autoimmune disease, and neoplasia.
Predisposing factors are conditions affecting the patient that increase the risk of developing ear disease but that do not trigger the ear disease. Predisposing factors include conformational abnormalities (e.g., pendulous pinnae, stenotic ear canals, and excessive hair) and ear canal epithelial compromise (e.g., swimming, inappropriate treatments such as hair plucking, trauma from cotton swabs, and irritating topical otic products).
If these primary causes and predisposing factors are not diagnosed and controlled, perpetuating factors such as bacterial and yeast infections, otitis media, and progressive pathologic changes (e.g., stenosis, hyperplasia, fibrosis, and calcification) develop. These perpetuating factors prevent the resolution of the otitis externa even if the primary cause is identified and controlled. The most commonly isolated infectious organisms from the horizontal ear canal in dogs with otitis externa are Malassezia pachydermatis, Staphylococcus intermedius, and Pseudomonas aeruginosa.
Indications for the Use of Video Otoscopy in the Examination Room
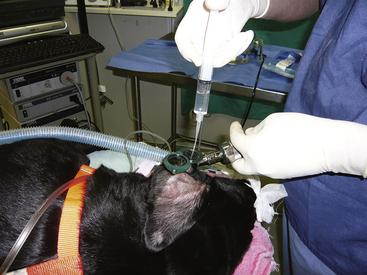
For the beginner, the use of the video otoscope in the awake animal can be frustrating because it requires hand–eye coordination. However, the best way to become familiar with using the equipment is to perform video otoscopic examinations on every patient. In the awake patient, proper technical restraint is a must. A technician experienced in handling awake animals for otic examinations will help to expedite the procedure, which results in less discomfort for the patient. Examinations are best performed on an examination table with the animal in sternal recumbency or standing. In the dog, it is best to have the technician stand on the opposite side of the ear to be examined and gently grasp the muzzle with one hand while keeping the other hand on the dorsum of the neck. In the cat, less restraint is usually best: the technician should place a hand on the dorsum of the neck or, if necessary, scruff the cat while the other hand controls the back end.
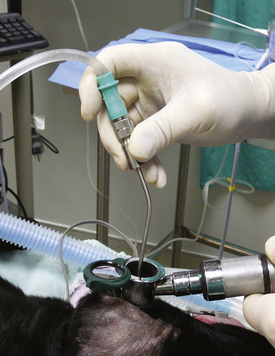
For evaluation of the ear, the tip of the video otoscope should be placed at the intertragic incisure (incisura intertragica). Then, the external ear canal needs to be gently extended into a straight line by grasping the ear pinna and pulling it up and outward while inserting the otoendoscope. Advance the otoendoscope into the horizontal ear canal as the canal is straightened while looking at the monitor. The focal distance of the video otoscope is such that it is not necessary to penetrate deeply into the horizontal ear canal to visualize the entire canal and tympanic membrane. In the awake cat, it is difficult to visualize the entire rostral and dorsal portion of the tympanic membrane.
Both ears should be examined, and if there is a complaint of unilateral otitis, I will begin with the least affected or unaffected ear first. The least affected or unaffected ear should be used to show the client a normal ear as compared with the clinically affected ear. If both ears are abnormal, then it is helpful to have pictures of a normal ear to show the client. After examination of one ear, the otoendoscope should be disinfected before it is inserted into the second ear. At minimum, I will wipe the otoendoscope with isopropyl alcohol before examining the second ear. If there is a considerable amount of exudate on the otoendoscope, I will soak the otoendoscope in glutaraldehyde for 20 minutes and rinse it thoroughly with tap water, wipe it dry, before placing it in the second ear. Depending on the severity of the ear disease, a complete otoscopic examination may not be possible. However, even if only the vertical ear canal can be viewed with the otoendoscope, the client can be shown the pathology of the ear, which will help them to understand the necessity of additional treatments and diagnostics. A plan for future management of the ear can then be formulated.
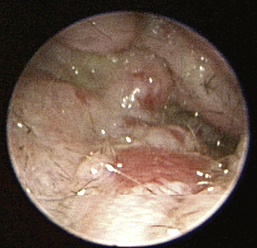
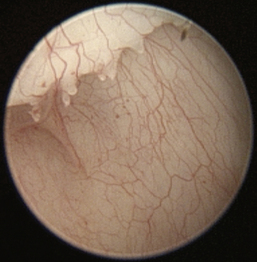
The normal vertical and horizontal ear canal should be pink, and in most animals, the cartilage is flexible; thus the otoendoscope should advance easily down the canal. There can be small amounts of wax in the ear canal. In animals with otitis externa, the changes detected in the ear canal are related to the severity of the ear disease. The first changes noted in the ear canals in cases of acute otitis externa include erythema and glandular hyperplasia (Figure 20-1). The amount of exudate in the ear canal is variable and should be documented, as should its color and consistency. A swab sample from each ear should be obtained for cytologic evaluation. If numerous rod bacteria are noted cytologically or the infection has worsened since the last visit, then a bacterial C/S is indicated. In more severe cases of otitis externa, glandular hyperplasia progresses, resulting in stenosis of the canal, sometimes to the point where the otoendoscope can no longer be advanced into the vertical ear canal. The degree of stenosis and hyperplasia should also be documented. Ulcerations may also be noted, especially in ears secondarily infected with P. aeruginosa. Parasites such as Otodectes cynotis may be visualized rapidly moving about in the ear canal with the video otoscope and are usually found in conjunction with dark brown ceruminous (“coffee ground”) exudate. On occasion, masses or foreign bodies may be seen in the ear canal during the video otoscopic examination.
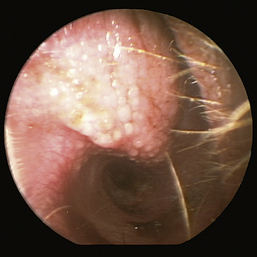
Figure 20-1 Mild erythema and glandular hyperplasia associated with otitis externa are some of the first changes noted in the ear canal.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
In cases of chronic otitis externa, the tympanic membrane may not be visualized due to excessive exudate, stenosis, hyperplasia, or a combination of these. If the tympanic membrane is visible, the color and gross appearance of the pars flaccida and pars tensa should be noted. Documentation of the findings on the video otoscopic examination of the vertical and horizontal ear canal as well the tympanic membrane is important for comparison during future reevaluations; this documentation should consist of digital images as well as other findings recorded in the patient record.
I find that most patients that allow an otoscopic examination with a handheld otoscope will allow one performed with a video otoscope. However, it is important to note that some dogs and cats will not allow a video otoscopic examination to be performed, despite proper restraint or the experience of the video otoscope user. In addition, some animals’ ears may be too painful to allow any otic examination.
Fogging of the lens during the video otoscopic examination in the awake patient can be a source of frustration. Fogging occurs when the cool lens comes into contact with the warm ear canal. There are a few procedures that can be used to control lens fogging. I use a combination of isopropyl alcohol wipes and a commercially available lens defogger (UltraStop). Before inserting the otoendoscope into the ear, I wipe the tip with an alcohol-soaked gauze sponge and then spray the defogger on the tip. This procedure will need to be repeated if the tip comes into contact with the ear canal epithelium (and subsequently accumulates some wax on the tip) or comes into contact with exudate in the ear canal. Another technique that has been used to defog the lens is to warm the tip of the otoendoscope in a bowl of warm water.
Indications for the Use of Video Otoscopy for Deep Ear Flushing, Myringotomy, Biopsy, and Foreign Body Retrieval
If the animal has chronic otitis externa, it may have concurrent otitis media. The most common clinical sign of otitis media is recurrent otitis externa. Rarely do my patients with otitis media have concurrent neurologic signs, but some will. Neurologic signs that are suggestive of middle and inner ear disease include facial nerve paralysis, Horner’s syndrome, vestibular disturbances, and hearing loss. Not every animal with chronic recurrent otitis externa requires anesthesia and a deep ear flush, so I have some guidelines I use to help make that decision. The following are cases in which I will consider performing anesthesia and a deep ear flush and examination:
Instrumentation
The standard otoendoscope is a straightforward 0-degree telescope with a length of 7 to 8.5 cm, a tip diameter of 4.75 to 5 mm, and a 2-mm working channel, which is fully immersible and able to be sterilized in ethylene oxide (gas) and autoclaved. The camera is a digital single-chip or three-chip camera of high resolution, with automatic white balance. The standard light source is halogen; however, I prefer the xenon light source, which gives off a “whiter” and brighter light. In addition, if the camera is to be used for other types of endoscopy, the xenon light is the preferred light source. The working channel allows the passage of 5F or smaller catheters, endoscopic forceps, and video otoscopic–compatible curettes. Some may choose to use a rigid cystoscope or arthoscope to examine the ear canal and tympanic membrane. Because of the delicate nature of these scopes, they should only be used in the anesthetized animal.
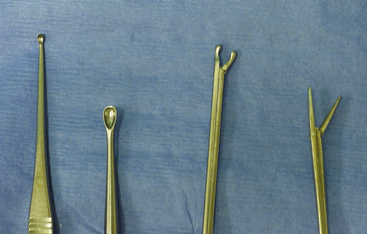
A dual-port adapter is available with some video otoscopes and can be attached to an external suction device and a fluid bag to provide simultaneous passive suctioning and flushing. External suction and flushing devices are also available and can be used by attachment of a catheter to the unit for directed suction and flushing. A medical-grade monitor is preferred for viewing because the image projected is superior to nonmedical grade monitors. The digital capture system functions like a computer and is invaluable for image documentation. Most have the capability of capturing still images and video clips. Biopsy and grasping forceps and curettes are available for obtaining samples, removing foreign bodies, and dislodging cerumen, respectively. Brushes are available for removing loose hairs and debris.
Instrument Disinfection
After the video otoscope and any associated equipment (e.g., curette, forceps, or dual-port adapter) is used, it is important to clean and disinfect the equipment. The clinician can remove exudate in the port of the otoendoscope by passing the manufacturer’s cleaning brush in and out of the port. The otoendoscope along with any equipment used during the procedure is first cleaned with an enzymatic cleaner. The soaking time will be indicated by the manufacturer of the cleaner, but soaking times should never exceed those recommended by the manufacturer of the otoendoscope to prevent damage. The otoendoscope and equipment are scrubbed with brushes and towels and then transferred to a distilled water bath to rinse off the enzymatic cleaner. After the otoendoscope and equipment are cleaned, they are disinfected by soaking in glutaraldehyde for the length of time indicated by the manufacturer of the otoendoscope. Alternatively, the curette and forceps may be gas sterilized. Some otoendoscopes may also be gas sterilized or autoclaved.
Patient Preparation
Once the decision has been made to perform a deep ear flush, the patient’s ear needs to be prepared for the procedure. To do an appropriate flush, one needs the vertical and horizontal ear canals to be as open as possible so that the tympanic membrane can be visualized. To do this, the animal should be started on topical and systemic glucocorticoids 2 to 3 weeks before the deep ear flushing. The only exceptions to this would be in an animal with neurologic disease where glucocorticoids may be contraindicated or in an animal that has a medical reason for not being able to be treated with glucocorticoids. In older patients I will usually run a complete blood count and biochemical profile before the administration of glucocorticoids to ensure that no metabolic problems are present that would preclude their use. If, when the animal returns for the deep ear flush, the ear canal has not opened up and the vertical and horizontal ear canals are still stenotic and hyperplastic, then the prognosis for medical management of this case is poor and treatment of the otitis will require surgical management. If the ear canals have opened up with the treatment, then the animal should be anesthetized for the deep ear flush.
Otic Anatomy
It is important to understand the anatomy of the external and middle ear when evaluating a patient with ear disease. First, in order to diagnose otic disease, one must be familiar with the appearance of the normal structures of the ear. Secondly, when one is performing a deep ear flush, it is imperative that one is aware of the delicate structures of the ear to avoid damaging them and causing neurologic complications.
The opening of the external ear canal is bounded by the helix (the free, slightly folded margin of cartilage at the base of the pinna) rostrally, the tragus laterally, and the antitragus caudally. The antitragus is a thin, elongated piece of cartilage caudal to the tragus and separated from it by the intertragic incisure (incisura intertragica). This anatomic region is the area in which I will insert the otoscopic cone or otoendoscope for the otoscopic examination. The vertical ear canal runs for about 1 inch, extending ventrally and slightly rostrally before taking a medial turn and forming the horizontal ear canal. There is a prominent cartilaginous ridge that separates the vertical and horizontal ear canals and, when the ear is in its normal position, makes otic examination of the horizontal ear canal difficult without the clinician elevating this ridge by grasping the ear pinna and lifting the ear.
The horizontal ear canal is composed of auricular and annular cartilage. The auricular cartilage rolls as it forms a tube. A separate cartilaginous band, the annular cartilage, fits within the base of this tube. The annular cartilage overlaps with the osseous external acoustic meatus and articulates via ligamentous tissue, giving the external ear canal flexibility.
In most breeds, hairs are present in the external ear canal, decreasing in number from distal to proximal. A very few fine hairs are found at the entrance of the cartilaginous external acoustic meatus. I find that these hairs are a useful landmark for locating the tympanic membrane when flushing an ear.
The middle ear consists of an air-filled tympanic cavity, three auditory ossicles, and the tympanic membrane. The tympanic membrane is located at a 45-degree angle in relation to the central axis of the horizontal part of the external ear canal. The tympanic membrane is a semitransparent membrane that separates the external ear canal from the middle ear, is thin in the center and thicker at the periphery, and is divided into two sections, the small upper pars flaccida and the larger lower pars tensa (Figure 20-2). The pars flaccida is the pink, small, loosely attached region forming the upper quadrant of the tympanic membrane that contains small blood vessels. The pars flaccida is usually flat; however, even in the healthy ear one may identify a bulging pars flaccida (Figure 20-3). The exception is the Cavalier King Charles Spaniel, in which a bulging pars flaccida may be indicative of a disease known as primary secretory otitis media (PSOM) (Figure 20-4).
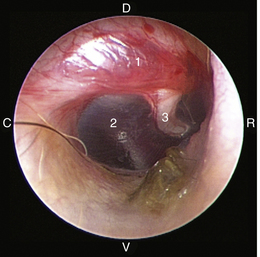
Figure 20-2 Normal anatomy of the canine tympanic membrane of the right ear. 1, Pars flaccida; 2, pars tensa; 3, stria mallearis; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
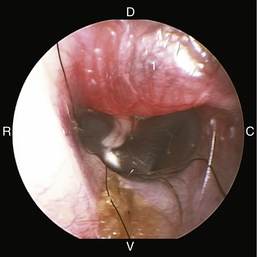
Figure 20-3 Bulging pars flaccida in a dog without any external or middle ear disease. 1, Pars flaccida; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
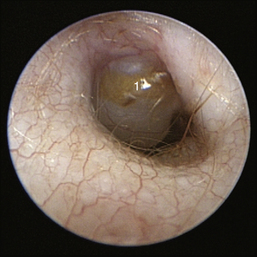
Figure 20-4 Bulging pars flaccida in a Cavalier King Charles Spaniel with primary secretory otitis media (PSOM). 1, Pars flaccida.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
The pars tensa, the thin, tough, gray structure with radiating strands, occupies the remainder of the membrane. The pars tensa is attached to the osseous ring of the external acoustic meatus. The manubrium of the malleus attaches to the medial surface of the tympanic membrane. The outline of the manubrium of the malleus, the stria mallearis, may be visualized when the tympanic membrane is viewed externally. The pars tensa has a concave shape when viewed externally because of the tension applied to the internal surface of the membrane, where the manubrium of the malleus is attached. The point of greatest depression is called the umbo.
The tympanic cavity consists of a small epitympanic recess, a large ventral bulla, and the tympanic bulla proper. In the dog, there is an incomplete bony septum, the bulla septum, which allows communication between the tympanic bulla proper and the ventral tympanic bulla. On the medial wall of the tympanic cavity, there is a bony eminence, the promontory, which houses the cochlea, and lies opposite the tympanic membrane medial to the epitympanic recess. At the caudolateral portion of the promontory, a foramen called the cochlear (round) window is located. The cochlear window is covered by a thin membrane that oscillates to dissipate the vibratory energy of the perilymph in the scala tympani. The vestibular (oval) window lies on the dorsolateral surface of the promontory immediately adjacent to the pars flaccida (Figure 20-5). It is covered by a thin diaphragm. The footplate of the stapes is attached to the diaphragm over the vestibular window. When flushing the middle ear, one must be very careful to avoid damaging the promontory or the cochlear window and to avoid damaging the inner ear, causing vestibular disturbances or deafness.
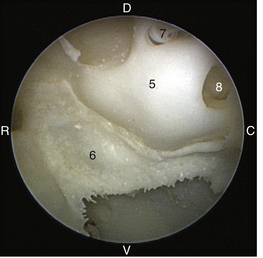
Figure 20-5 Left ear of a canine skeleton (tympanic membrane removed) illustrating middle ear structures: 5, promontory; 6, bulla septum; 7, vestibular (oval) window; 8, cochlear (round) window; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
The middle ear cavity of the cat is different from the dog’s and is divided by a septum into two distinct regions, the tympanic cavity and the bullar cavity. These two cavities are connected through a narrow foramen between the septum and the petrous bone. In the small dorsolateral compartment lie the auditory ossicles, the osteum of the auditory tube, and the tympanic membrane. The larger ventromedial compartment is an air-filled tympanic bulla. The bony septum should be perforated surgically when middle ear disease is present in both cavities for proper drainage of the middle ear cavity. Rough handling of the bony septum may result in damage to the postganglionic sympathetic nerves. The nerves, which are visible submucosally as fine strands over the cochlear promontory, should be avoided during surgical removal of the septum in the cat.
The three auditory ossicles, the malleus, incus, and stapes, are the bones that transmit and amplify air vibrations from the tympanic membrane to the inner ear. The malleus is attached to the tympanic membrane, the petrous temporal bone, and the incus. The incus is suspended between the malleus and the stapes. The footplate (base) of the stapes is attached to the vestibular window, which is in direct contact with the perilymph fluid. The vestibular window is approximately 18 to 20 times smaller in area than the tympanic membrane.
Procedure for Deep Ear Flush with the Video Otoscope
The video otoscope is an invaluable tool for evaluating and flushing the middle ear of an anesthetized patient with otitis media. I would encourage standardizing the orientation of the video otoscope relative to the animal when utilizing the video otoscope in the anesthetized patient. That way, the surgeon avoids becoming disoriented when the anatomy of the ear is abnormal. Next, make sure that the monitor is easily visible while examining the ear, which means that it is directly in front or to the left or right. It is very awkward for the surgeon to try to view the monitor over his or her shoulder.
The animal should be anesthetized and have an endotracheal tube in place for the deep ear flush. The endotracheal tube will prevent aspiration of any fluid that passes from the middle ear into the auditory tube to the nasopharynx and then to the oropharynx and respiratory tract if a myringotomy and middle ear flush are performed. Before the deep ear flush, the first procedure I perform is radiographic imaging to evaluate the middle ear. I prefer to perform computed tomography (CT) on my patients, but in general practice, this is often not available, and bulla radiographs suffice. Radiographic imaging not only allows evaluation of the middle ear for changes consistent with otitis media (i.e., soft tissue density in the bulla) but it also allows identification of significant changes in the bulla, such as sclerosis or lysis, that may indicate osteomyelitis or a tumor, respectively, which will aid in determining the prognosis for medical treatment of the ear disease.
Just before cleaning the ear, one should obtain swab samples from the external ear canal for bacterial C/S and cytologic examination. Video otoscopic images are taken of the external ear canal for documentation. With the patient in lateral recumbency, preferably on a tub table and a heating pad (those specifically designed for veterinary surgical procedures), the external ear canal is soaked with an ear cleaner or ceruminolytic agent for 10 minutes. I prefer to use a ceruminolytic agent because these products are the best for removing exudate from the ear canal. However, the majority of these products are ototoxic if they enter into the middle ear cavity. Once the ear has been cleaned, if the tympanic membrane is ruptured or torn, I will repeatedly flush sterile saline into the external ear canal to remove the ceruminolytic agent. There is, however, one commercially available ceruminolytic agent, Cerumene, that has been shown to be nonototoxic.
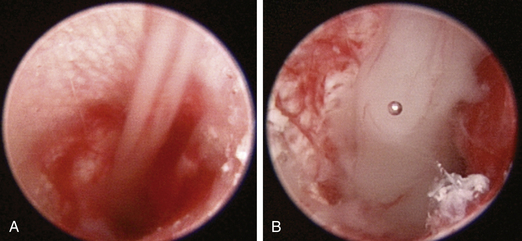
After the external ear canal has been soaked with a ceruminolytic agent or ear cleaner, the external ear canal is flushed with warm sterile isotonic saline with the use of a bulb syringe to remove large debris and exudate. This is followed by flushing with warm sterile isotonic saline with the use of a handheld otoscope with an 8F polypropylene urinary catheter attached to a 12-mL syringe passed through an otoscopic cone. An 8F red rubber feeding tube may be cut to size and used to flush the external ear canal. External flushing devices, such as the Vet Pump 2, available from the manufacturer of the video otoscope, may be used in place of the manual flushing described above. Once the ear is clean, the tympanic membrane is evaluated with the video otoscope and images are taken for documentation. If the tympanic membrane is ruptured or torn, as previously stated, it is important to flush copious amounts of sterile saline into the ear canal to remove the ceruminolytic agent. In addition, once a ruptured or torn tympanic membrane has been identified, do not infuse the ceruminolytic agent into the bulla. Once the exudate has been removed from the external ear canal, I attach the dual-port adapter to the port on the otoendoscope. The dual-port adapter is used to perform passive suctioning and flushing of the external ear canal, which expands the ear canals, further magnifies the ear, prevents lens fogging, and continues to clean the ear. A warmed bag of sterile saline is attached to a Venoset on one port of the dual-port adapter, and a suction hose is attached to the other port. A tear or rupture can be identified by bubbles coming from the tympanic membrane. Once the patency of the tympanic membrane has been determined, I will remove the saline from the ear canal and evaluate the tympanic membrane with the otoendoscope because saline in the ear canal can sometimes cause the tympanic membrane to look opaque (which would be considered abnormal) when it is not. If the tympanic membrane is ruptured, then the patient has otitis media. A myringotomy does not need to be performed, and samples from the middle ear for cytologic and culture analysis can be obtained through the tear in the tympanic membrane. I find it helpful to have technical assistance in obtaining these samples. I will turn the otoendoscope to where the port is ventral, without moving the camera. This allows easy access to the port and keeps the instruments aimed ventrally so that the auditory ossicles and promontory are avoided. I will have a technician stand behind the patient’s head and hold the ear in one hand and the video otoscope in the other. This allows me to have my hands free to flush the middle ear (Figure 20-6). An open-end 3½F Tomcat catheter attached to a 12-mL syringe is placed through the port of the otoendoscope. Saline is flushed into the middle ear cavity and aspirated back for culture, and a second sample is flushed into the middle ear and aspirated back for cytologic analysis. After sample retrieval, the middle ear is flushed with warm sterile saline through the open-ended Tomcat catheter. I will flush and aspirate back 1 mL of sterile saline at a time and will finish when the saline flushed from the middle ear cavity is clear.
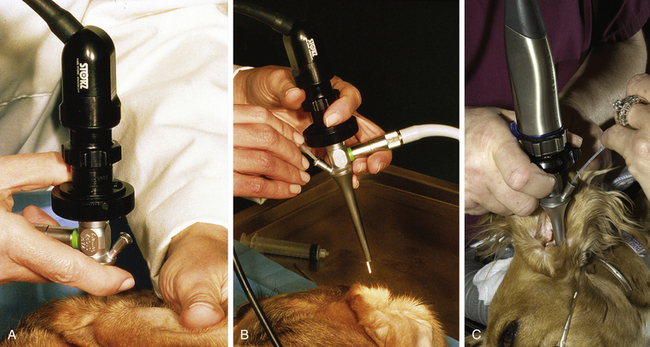
Figure 20-6 A, The normal position of the otoendoscope (port is dorsal) and camera in the ear canal of a dog. B, Rotation of the otoendoscope allowing the port to be ventral. A Tomcat catheter is placed through the port. C, Placement of the otoendoscope in the ear canal with the port ventral. The clinician is flushing the middle ear with the catheter and the technician is holding the otoendoscope and camera.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
If the tympanic membrane is intact, then one must evaluate the tympanic membrane to determine whether it is abnormal or normal and couple that with the results from the radiographic imaging. A tympanic membrane, specifically the pars tensa, that is opaque, gray, peeling, bulging, or hemorrhagic would be considered abnormal. At times, it may be difficult to determine whether the “structure” that one is visualizing is indeed the tympanic membrane, exudate in front of the membrane, or, in the case of a ruptured tympanic membrane, middle ear structures. In those situations, I will pass an open-ended 3½F Tomcat catheter through the port on the otoendoscope and gently touch the structure in question.
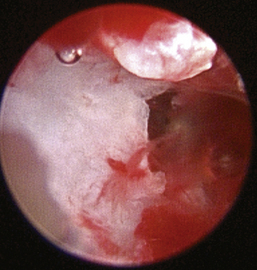
Once it has been determined that the tympanic membrane is intact and is abnormal or if there is radiographic evidence of otitis media, then a myringotomy incision would need to be made to be able to obtain samples from the middle ear and flush the middle ear cavity. This can be accomplished by passing a myringotomy needle (specially designed for use with the otoendoscope), open-ended 3½F Tomcat catheter, or 5F polypropylene urinary catheter (cut down in length) through the port on the otoendoscope into the caudoventral quadrant of the pars tensa. The myringotomy needle, Tomcat catheter, or 5F polypropylene urinary catheter is used to make the myringotomy incision (Figure 20-7). To obtain samples from the middle ear cavity for bacterial culture and cytologic analysis, an open-ended 3½F Tomcat catheter or 5F polypropylene urinary catheter is attached a 12-mL syringe placed through the port of the otoendoscope. One milliliter of saline is flushed into the middle ear cavity and aspirated back for culture, and a second sample is flushed into the middle ear and aspirated back for cytologic analysis. Then the middle ear cavity is flushed in the same fashion as discussed previously when the tympanic membrane was ruptured. The normal tympanum has been shown experimentally to heal in 21 to 35 days. Therefore, if the ear is kept free from infection after the myringotomy procedure, the tympanic membrane should heal in 3 to 4 weeks.
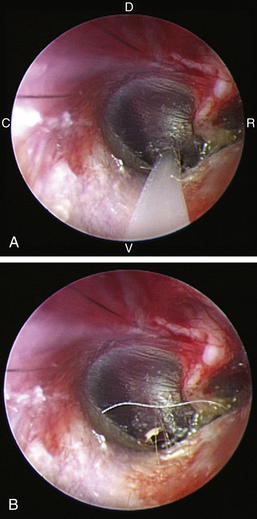
Figure 20-7 A, Use of a Tomcat catheter to make a myringotomy incision into the caudoventral quadrant of the pars tensa of a cadaver dog’s right ear. C, Caudal; D, dorsal; R, rostral; V, ventral. B, Myringotomy incision.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
Rarely, while the external ear canal is being flushed, the ear canal epithelium will begin to peel. This is from soaking the ear with saline and ear cleaners or ceruminolytic agents, which results in maceration of the stratum corneum. Unfortunately, if this occurs, it is best to abort the ear flush and reschedule; in my experience, once the ear canal epithelium begins to peel, it will continue to peel and make it very difficult, if not impossible, to visualize the tympanic membrane. Fortunately, this does not occur very frequently.
Once the ear canal has been cleaned, masses and foreign bodies may now be identified that were unable to be seen (if present) before the flush because of the exudate inhibiting visualization. The video otoscope, along with specific forceps, can be used to remove or sample masses in the ear canal or remove foreign bodies. For biopsies, the biopsy forceps are inserted into the port on the otoendoscope. It is best to keep the tip of the otoendoscope in the vertical ear canal, pass the biopsy forceps into the port, and extend it out into the lumen of the ear canal. Then have the technician slowly move the video otoscope closer to the mass to be removed or sampled. If the tip of the otoendoscope is too close to the mass, once the biopsy forceps are advanced through the port, they may extend past the structure in question and potentially rupture the tympanic membrane. The mass or biopsy sample from the mass should be submitted for histopathologic evaluation. It is advisable to perform the biopsy procedure as the final procedure because there will be some bleeding from the biopsy site, which makes visualization of deeper structures difficult. Foreign bodies may be removed with grasping forceps under direct visualization of the video otoscope. The procedure is the same as previously described for the use of the biopsy forceps.
Complications of Deep Ear Flushing and Myringotomy
I rarely see any complications after deep ear flushing in the dog; however, I go over the neurologic side effects (i.e., facial nerve paralysis, Horner’s syndrome, vestibular disturbances, and hearing loss) that may occur subsequent to a deep ear flush and myringotomy with the client and have the client sign a consent form before the procedure. In cats, complications after ear flushing are common. Part may be due to the location of the postganglionic nerves, which run through the middle ear cavity submucosally below the septum of the tympanic bulla over the cochlear promontory. Damage to these nerves results in Horner’s syndrome. Fortunately, most complications will resolve within 3 to 6 weeks. It is important to perform a neurologic examination on any animal that has otitis as well as question the client on his or her perception of the animal’s hearing ability before the ear flushing.
In some cases of chronic, recurrent otitis externa, surgical intervention, consisting of a total ear canal ablation and bulla osteotomy, may be warranted. Surgery is indicated when there is inadequate response of the external ear infection to medical management due to poor client compliance or the presence of a resistant organism; otitis media fails to respond to myringotomy, ear canal flushing, and medical management; or progressive pathologic changes of the ear (i.e., calcification, proliferation, and stenosis) have resulted in permanent occlusion of the ear canal. Fortunately, this is rarely necessary if aggressive medical therapy has been used.
Angus J.C., Campbell K.L. Uses and indications for video-otoscopy in small animal practice. Vet Clin North Am Small Anim Pract. 2001;31:809-828.
Cole L.K. Otoscopic evaluation of the ear canal. Vet Clin North Am Small Anim Pract. 2004;34:397-410.
Evans H.E. The ear. In Evans H.E., editor: Miller’s anatomy of the dog, ed 3, Philadelphia: Saunders, 1993.
Mansfield P.D., Steiss J.E., Boosinger T.R., et al. The effects of four, commercial ceruminolytic agents on the middle ear. J Am Anim Hosp Assoc. 1997;33:479-486.
Atlas Pages 595-597
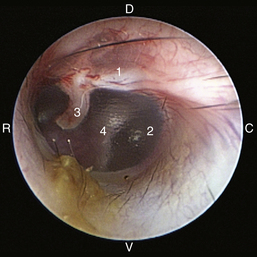
Figure 20-8 Normal canine tympanic membrane of the left ear. 1, Pars flaccida; 2, pars tensa; 3, stria mallearis; 4, bulla septum; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
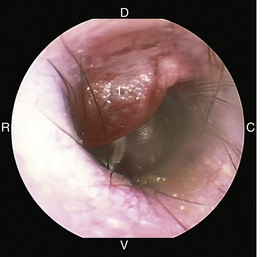
Figure 20-9 Bulging pars flaccida in a dog without otitis media. 1, Pars flaccida; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
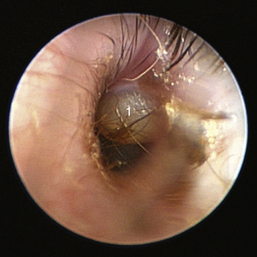
Figure 20-10 Bulging pars flaccida in a Cavalier King Charles spaniel dog with primary secretory otitis media. 1, Pars flaccida.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
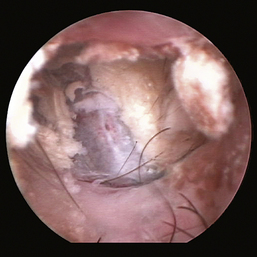
Figure 20-11 Bulging pars tensa in a dog with otitis media.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
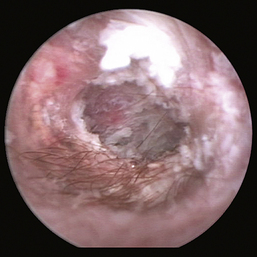
Figure 20-12 Scaly pars tensa in a dog with otitis media.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
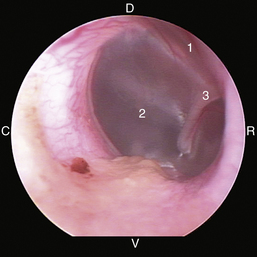
Figure 20-13 Normal feline tympanic membrane. 1, Pars flaccida; 2, pars tensa; 3, stria mallearis; C, caudal; D, dorsal; R, rostral; V, ventral.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
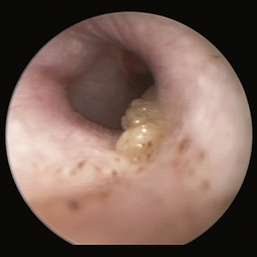
Figure 20-14 Bulging, opaque pars tensa in a cat with otitis media.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
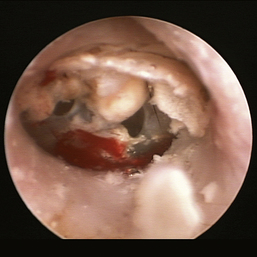
Figure 20-15 Feline tympanic membrane with two tears in the pars tensa.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
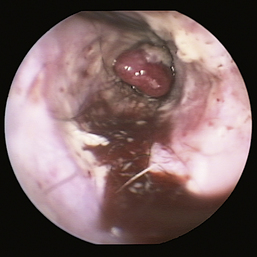
Figure 20-16 Ceruminal gland adenoma with blood noted in the external ear canal of a dog.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
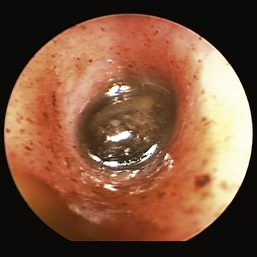
Figure 20-17 Ceruminolith in the horizontal ear canal of a cat.
(Courtesy of Dr. Lynette Cole, The Ohio State University, Columbus, Ohio.)
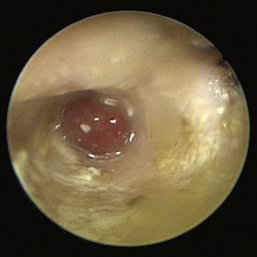
Figure 20-18 Nasopharyngeal polyp in the horizontal ear canal of a cat.
(Courtesy of Andrew Hillier, D.V.M.)
Conservative Management of Chronic Otitis Media in Dogs
David K. Saylor and Jarod E. Williams
Chronic canine otitis media (OM) is a frustrating problem in small animal practice. OM is most commonly the result of bacterial otitis externa extension through a ruptured tympanum. Less commonly, OM can result from bacterial nasopharyngitis with eustachian tube retrograde translocation of bacteria to the tympanic bulla. General symptoms include scratching, rubbing, head shaking, exudation, odor, periaural pain and pain on temporomandibular joint extension, head tilt, Horner’s syndrome, facial neuropathies, and ataxia.1 Examination of the canal is often unsuccessful without sedation or general anesthesia. Most patients have been treated with multiple antimicrobials over a period of months to years. The majority of cases are unilateral, but bilateral OM does occur often in conjunction with atopic dermatitis.2,3 A polymicrobial gram-positive or gram-negative aerobic recovery from middle and external ear segments has been reported in multiple studies.4-7 Given the spectrum of potential pathogens and often significant antimicrobial resistance, specific culture-guided therapy is required. Enteral and parenteral antibiotic delivery has often been shown to be inferior to topical delivery methods in humans.8 The same observations should be assumed in dogs in light of the relatively extensive canal and bulla volume comparatively. Definitive treatment is often unsuccessful without surgical intervention. Total ear canal ablation with lateral bulla ostectomy (TEA/LBO) and lateral ear canal resection techniques have been well characterized9,10 but are invasive and traumatic. A number of seemingly “end-stage ears” can be salvaged noninvasively.11 Careful transtympanic bulla curettage (TBC) and lavage of the bulla followed by medical management with otic topical medications has a high rate of success. The goal of TBC is to resolve infection and eliminate pain. Significant hearing loss is generally present before TBC. Restoration of hearing is uncommon and not considered a procedural goal. The main limiting factor with conservative management is the patency of the lumen of the canal. Successful TBC is unlikely when the canal lumen has been severely compromised by fibrosis, stenosis, or glandular hyperplasia. Progression of OM to otitis interna with neurologic complications is a potential outcome without TEA/LBO.12 The majority of cases of OM can be managed without TEA/LBO with successful TBC and culture-guided local antimicrobial–corticosteroid therapy.
Instrumentation
Otoscopic evaluation with a rigid endoscope is superior to that achieved with a handheld otoscope. Visualization of the tympanic cavity and its contents is an essential part of the examination. The rigid scope can be visually directed into the bulla. The magnification and superior lighting provided with the rigid endoscope are invaluable. The 1.9-mm, 30-degree visual angled integrated endoscope (Karl Storz model No. 27030BA) with ports for irrigation, suction, and biopsy can be used in any sized dog. Irrigation is necessary to keep the end of the scope clear. The operating ports and specialized endoscopic cup forceps of the 1.9-mm endoscope are often too small to be effective at meaningful TBC. Our personal preference is to use this scope for examination and a handheld otoscope for introduction of instruments into the canal and bulla. Instruments used in TBC include Noyes alligator forceps, various curettes, and McIlwraith peapod arthroscopic rongeurs (Figure 20-19). These rongeurs are the most effective tool. They can be purchased with cup sizes of 2, 4, 5, and 6 mm. Tomcat catheters are used for flushing, and various diameter Frazier suction tips are used for suction. A 1.9-mm rigid endoscope with an arthroscopy sheath for irrigation is a suitable substitute for the integrated scope.
Patient Evaluation and Anesthesia
Preoperative diagnostic testing includes a complete blood count (CBC), chemistry panel, and urinalysis. Otoscopy and any procedure involving the middle ear are conducted under general anesthesia. An intravenous catheter is placed, and intravenous fluids are recommended. A balanced neuroleptanalgesic preanesthetic combination is recommended for most patients because the affected ear is often very painful. Anesthetic induction agents are chosen based on the patient assessment. Inhalant anesthesia is provided for maintenance during the procedure. Postoperative analgesia is highly recommended for 2 to 3 days postprocedure.
External Ear Canal Evaluation
Samples are collected from the ear canal for aerobic bacterial culture. External ear canal exudate sampling has been shown to be sufficient for prediction of aural tissue infection in patients with chronic otitis externa and media.5 The canal is lavaged and suctioned to remove gross obstructive debris (Figures 20-20 and 20-21). The 1.9-mm endoscope is introduced into the canal, and the lining is visualized. Ulcerations in the lining are common findings. They can be focal or involve large sections of the epithelium (Figure 20-22). The degree of hyperplasia of the lining varies. Occasionally, obstructive fibrosis and stenosis secondary to severe infection are found in some canals. These ears are more effectively treated with TEA/LBO.
Middle Ear Evaluation
The middle ear starts at the tympanic membrane. In most truly chronic cases it is rare to find an intact tympanum (Figure 20-23). However, intact tympani can be present with OM.11 In this case, nasopharyngitis or secretory OM should be considered as potential origins. If the eardrum is intact, follow recommendations for medical management described previously in this chapter. If the tympanum is not present and the bulla is empty (Figure 20-24), copious lavage with warm saline can be performed through the endoscope irrigation port. This is often sufficient for cases without marked bulla soft tissue proliferation. More often, mucus, debris, and proliferative tissues are present, filling the bulla (Figures 20-26 and 20-28). The endoscope is removed, and curettage is performed through a standard otoscope. One should be familiar with the position of the round window, the oval window, and the opening to the eustachian tube and consider the spatial relationship of a straight ear canal and the medial wall of the bulla. Manipulate the pinna to straighten the canal and introduce instruments in a rostral, ventral direction. It is difficult to damage any of these structures with proper placement of instruments. Overzealous flushing is more of a concern. In dogs a bony ridge separates the tympanic cavity from the tympanic bulla and, in a few individuals, may cause difficulty in placing the scope or instruments into the tympanic bulla (see Figures 20-24 and 20-25). When alligator forceps, curettes, or a peapod arthroscopic rongeur is used, proliferative tissues and attached debris can be removed. Progress is visualized with intermittent rigid endoscopic visualization. The TBC procedural goal is to debride proliferative tissue and flush out all debris, which results in visualization of bulla periosteum (see Figures 20-24 and 20-25). The medial wall of the bulla should be visible at the end of the procedure (Figure 20-27).
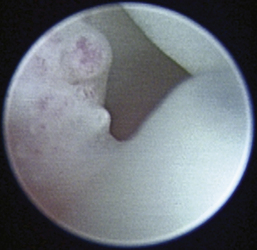
Figure 20-23 Proliferative tissue and mucopurulent discharge frame a large defect in the tympanic membrane of the dog in Figure 20-22.
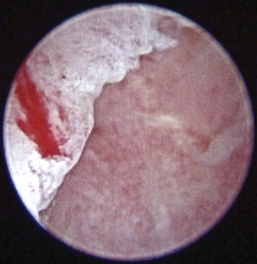
Figure 20-24 Appearance of the tympanic bulla of the dog in Figures 20-22 and 20-23 after debridement and irrigation. Note the inflamed appearance of the medial wall of the bulla and the sclerosis of the dorsal bony ridge when compared to the same structures in a different dog in Figure 20-25.
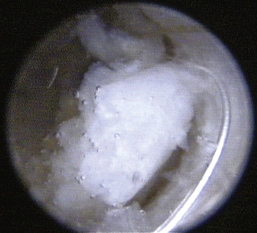
Figure 20-26 Mucopurulent discharge and debris filling the tympanic bulla of an 11-year-old Golden retriever with a chronic history of otitis and recent development of a head tilt.
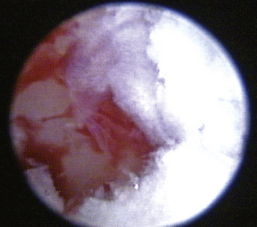
Figure 20-27 Appearance of the medial wall of the tympanic bulla of the dog in Figure 20-26 after curettage and copious irrigation.
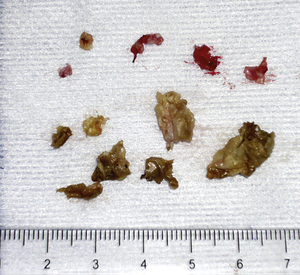
Figure 20-28 Debris and proliferative tissue removed from the tympanic bulla of the dog in Figures 20-26 and 20-27.
Potential Complications and Care after Treatment
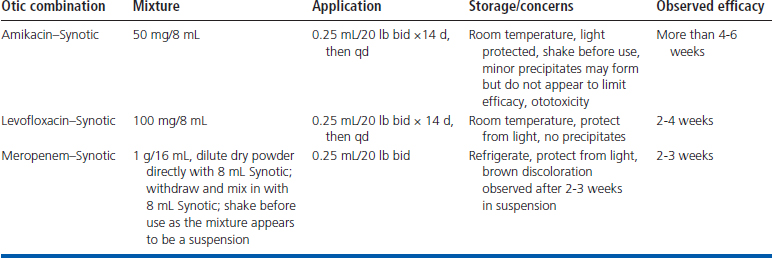
Most patients are discharged the day of the procedure on an opioid analgesic and eardrops on the basis of case medical history and in-house cytologic results. Various combinations of dimethylsulfoxide, corticosteroids, and parenteral antibiotics have been previously reported13 and have been effectively used. Otic application of parenteral formulas has been reported in the treatment of resistant P. aeruginosa otitis externa in dogs.14 Mixed solution shelf lives and solution precipitates bring into question the usefulness of such approaches. That said, we have been able to successfully clear numerous complex infections including multidrug–resistant gram-negative bacilli and methicillin-resistant Staphylococcus aureus with careful periodic reevaluation using local therapies alone (Table 20-1). Not all antibiotics are created equal. Some drugs within classes, because of their molecular structures or mechanism of action, are reported to be more or less susceptible to resistance development over time.15,16 We choose compounds reported to have lower resistance potential because most cases require prolonged treatment of weeks to months. With this approach, we do not find resistant bacterial selection an issue over the course of treatment, and successful management of even chronic, stubborn, and resistant OM can be achieved without the systemic administration of antimicrobials. While the OM infection is treated, it is imperative to determine the cause of the chronic otitis externa so that an appropriate maintenance program can be started when the OM has resolved. It is also extremely important that clients understand that a maintenance program is essential in preventing future ear infections.
Possible complications of TBC include Horner’s syndrome, facial nerve paralysis, deafness, vestibular abnormalities, and, rarely, meningitis or encephalitis. Always use caution when placing instruments in the tympanic cavity and flush with care. The use of amikacin in an ear with a ruptured eardrum is controversial because of the ototoxicity of aminoglycosides instilled in the middle ear. Candidates for this procedure are more often than not hearing impaired in the affected ear. The goal of this procedure is to resolve the infection, eliminate pain, and salvage the ear canal. The purpose is not to restore hearing. Antimicrobial agents are chosen more for their effectiveness than any concern about ototoxicity. If iatrogenic hearing loss is of significant concern, a nonototoxic agent should be employed.
1. Gotthelf L. Diagnosis and treatment of otitis media in dogs and cats. Vet Clin North Am Small Anim Pract. 2004;34(2):469-487.
2. Rosser E.Jr. Causes of otitis externa. Vet Clin North Am Small Anim Pract. 2004;34(2):459-468.
3. Saridomichelakis M.N., Farmaki R., Leontides L.S., et al. Aetiology of canine otitis externa: a retrospective study of 100 cases. Vet Dermatol. 2007;18(5):341-347.
4. Cole L.K., Kwochka K.W., Kowalski J.J., et al. Microbial flora and antimicrobial susceptibility patterns of isolated pathogens from the horizontal ear canal and middle ear in dogs with otitis media. J Am Vet Med Assoc. 1998;212:534-538.
5. Cole L.K., Kwochka K.W., Hillier A., et al. Comparison of bacterial organisms and their susceptibility patterns from otic exudate and ear tissue from the vertical ear canal of dogs undergoing total ear canal ablation. Vet Ther. 2005;6(3):252-260.
6. Hariharan H., Coles M., Poole D., et al. Update on antimicrobial susceptibilities of bacterial isolates from canine and feline otitis externa. Can Vet J. 2006;47:253-255.
7. Colombini S., Merchant S.R., Hosgood G. Microbial flora and antimicrobial susceptibility patterns from dogs with otitis media. Vet Dermatol. 2000;11:235-239.
8. Esposito S., D’Errico G., Montanaro C. Topical and oral treatment of chronic otitis media with ciprofloxacin. Arch Otolaryngol Head Neck Surg. 1990;116:557-559.
9. Mason K.L., Harvey C.E., Orsher R.J. Total ear canal ablation with lateral bulla ostectomy for end-stage otitis in dogs. Vet Surg. 1988;17(5):263-268.
10. Lanz O., Wood B. Surgery of the ear and pinna. Vet Clin North Am Small Anim Pract. 2004;34(2):567-599.
11. Palmeiro B.S., Morris D.O., Wiemelt S.P., et al. Evaluation of otitis media after lavage of the tympanic bulla and long-term antimicrobial drug treatment in dogs: 44 cases (1998-2002). J Am Vet Med Assoc. 2004;225:548-553.
12. Spangler E.A., Dewey C.W. Meningoencephalitis secondary to bacterial otitis media/interna in a dog. J Am Anim Hosp Assoc. 2000;36:239-243.
13. Boothe D.M. Small animal clinical pharmacology and therapeutics. Philadelphia: Saunders; 2000.
14. Nutall T.J. Use of ticarcillin in the management of canine otitis externa complicated by. Pseudomonas aeruginosa, J Small Anim Pract. 1998;39:165-168.
15. Cunha B.A. Antibiotic resistance. Med Clin North Am. 2000;84(6):1407-1429.
16. Kotra L.P., Haddad J., Mobashery S. Aminoglycosides: perspectives on mechanisms of action and resistance and strategies to counter resistance. Antimicrob Agents Chemother. 2000;44:3249-3256.
Conservative Management of Feline Inflammatory Polyps
David K. Saylor and Jarod E. Williams
Feline inflammatory polyps are benign growths that most commonly are found in young cats.1 Infectious causes have been theorized. However, polymerase chain reaction studies have failed to reliably identify etiologic viral organisms.2 Most arise in the tympanic bulla (TB) or eustachian tube (ET). The polyps can then extend either through the tympanic membrane into the horizontal ear canal or down the ET into the nasopharynx (NP). Polyps can be single or multiple, involve one or both ears, and can be found in the NP.3 Symptoms depend on the location of the polyp(s). Polyps in the NP typically cause chronic stertorous respiration and have been associated with chronic rhinitis. Polyps in the ear canal cause recurrent otitis externa and media and can lead to head tilt, nystagmus, and ataxia. The recommended treatment is traction avulsion of the polyp followed by ventral bulla osteotomy (VBO).1,3-5 Simple removal of the polyp with traction avulsion is often followed by regrowth and relapse of symptoms. Polyrecurrence has been reported to be greater in cats with aural components, and recurrence may be decreased by corticosteroid therapy.6
Traction avulsion of the polyp followed by transtympanic bulla curettage (TBC) of the dorsolateral compartment (DLC) of the bulla and ventromedial compartment (VMC) septotomy to allow drainage of the ventromedial compartment is less invasive compared with VBO. Most cats are treated on an outpatient basis, which reduces hospitalization time and costs. Recurrence of polyps with this technique is approximately 10% (unpublished data) with postoperative local installation of an otic steroid–antibiotic combination. If polyps recur, procedural repetition or VBO can be performed.
Instrumentation
Video otoscopy performed with a video otoscope or a rigid endoscope improves visualization of the ear canal, tympanic membrane, and middle ear because of the superior magnification and lighting. This advantage is important in evaluating the ear canal and TB after removal of the polyp. A handheld otoscope is used to allow passage of larger instruments for removal of smaller polyps and proliferative tissues within the canal and DLC (Figure 20-29). A peapod arthroscopic rongeur is preferred for removal of soft tissue. Curettes of varying sizes are used to remove granulation tissue from the surface of the TB septum. A Frazier suction tip is used to remove discharge and irrigation fluids. Open-ended Tomcat catheters are used for flushing the canal and middle ear. We prefer using the 1.9-mm integrated scope (Karl Storz model No. 27030BA) for visualization of the canal. The scope has a 30-degree viewing angle and three ports for biopsy, irrigation, and suction. Irrigation is essential for visualization of the canal and bulla anatomy.
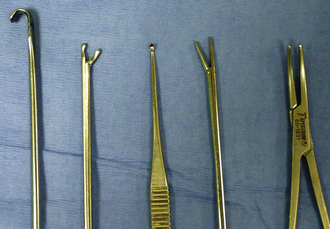
Figure 20-29 Instruments for polyp removal and débridement of tympanic bulla (left to right): spay hook for retraction of soft palate, peapod arthroscopic rongeur for grasping small polyps and proliferative tissue, small bone curette for curettage and puncture of the septum, alligator forceps for removal of tissue, and mosquito hemostat for traction and avulsion of polyps.
Feline Middle Ear Anatomy
The feline TB is divided into the VMC and DLC by a bony septum. The larger VMC is palpable caudal to the angle of the jaw and surrounds the ventral, medial, and caudal aspects of the DLC. This compartment is entered when VBO is performed. It is frequently filled with mucopurulent discharge but rarely contains polyps. The eardrum forms the major portion of the lateral wall of the smaller DLC, and the septum forms the medial wall. The auditory os of the ET occupies the dorsomedial and rostral extremity of this compartment. It is believed that most polyps originate in this area. As polyps grow they either extend down the ET to lie in the NP or more commonly extend laterally through the eardrum to sit in the ear canal.
The two compartments communicate through a narrow fissure where the septum is incomplete dorsally.7 The caudal aspect of this fissure enlarges to form a foramen that houses the round window of the cochlea. The promontory lies medial to the foramen along the dorsal wall of the bulla primarily in the VMC but does communicate with the DLC along the fissure above the septum. The tympanic plexus lies on the surface of the promontory. Damage to this structure causes Horner’s syndrome.8,9 A transient Horner’s syndrome is a common sequela of polyp removal even if a bulla osteotomy is not performed. The round (cochlear) window, oval (vestibular) window, and auditory ossicles are all found in the dorsal aspect of the bulla. Avoiding curettage in this area minimizes iatrogenic trauma that produces loss of hearing and vestibular deficits.
Procedure
The procedure is performed under general anesthesia. The preanesthetic evaluation consists of a CBC, chemistry profile, and feline leukemia virus and feline immunodeficiency serologic testing. A balanced neuroleptanalgesic preanesthetic combination is recommended to reduce the amount of induction and maintenance agents required. Although some clinicians prefer radiographs or CT scans to evaluate the TB, we find the added cost of imaging unnecessary, specifically in cats less than 3 years of age. In middle-aged to geriatric cats, imaging of the middle ear is highly recommended because of the higher incidence of malignant tumor presence.10 Palpate the soft palate to check for a mass in the NP. The soft palate is then retroflexed with a spay hook for direct visualization (Figure 20-30, A). Evaluation of the NP is much easier if surgical loops and a headlamp are used. Polyps in the NP are grasped with a hemostat or right-angle forceps and removed via traction avulsion (Figure 20-30, B). On removal of the polyp, suction is used to clear the NP, and the contralateral ET orifice is examined.
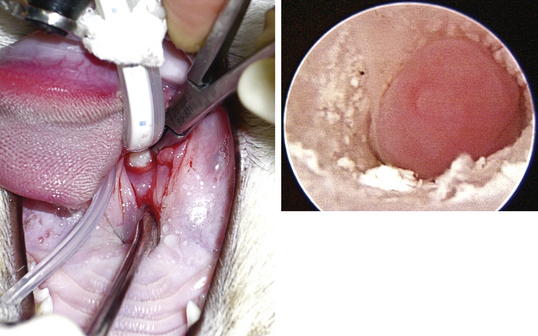
Figure 20-30 Inflammatory polyps in the nasopharynx and right horizontal ear canal of a 10-month-old, female, spayed domestic short hair.
Once the NP has been cleared, otic examination is performed with the handheld otoscope. Affected canals are generally filled with mucopurulent discharge and waxy debris that can hide the polyp. The discharge is cleared with gentle flushing and suction. Once the canal is clean, a 1.9-mm rigid endoscope is used to evaluate the canal. Polyps appear as smooth pink masses that tend to fill the lumen of the horizontal canal (see Figure 20-30, B, and Figure 20-32). Larger polyps can extend into the vertical canal and in extreme cases may be visible in the opening of the vertical canal. The polyp is grasped with a hemostat and removed via traction avulsion. Always seat the hemostat as deep into the canal as possible to enable complete removal of the polyp. Typically there is a stalk associated with one end of the polyp indicative of a narrow base of attachment (Figure 20-31). After removal of the polyp reevaluate the canal, tympanum, and bulla using the rigid endoscope (Figure 20-33). Findings vary in the horizontal canal. It can be empty, or smaller polyps and inflammatory tissue can be found extending into the tympanic cavity. Intact tympani are rarely present.
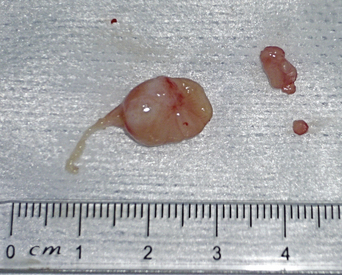
Figure 20-31 Large nasopharyngeal polyp and smaller polyps from the right ear canal of the cat (see Figure 20-30).
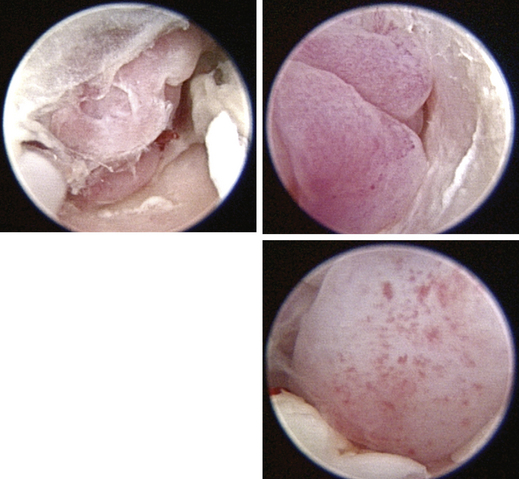
Figure 20-32 Multiple polyps in the right ear canal of a 2-year-old, male, neutered domestic short hair with a history of chronic otitis.
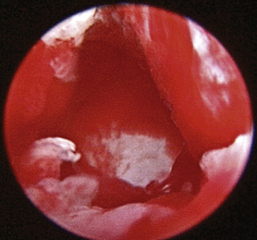
Figure 20-33 Appearance of the septum after debridement of the polyps of the cat (see Figure 20-32).
Proliferative tissue, satellite polyps, and fibrinous debris that extend into the TB are commonly present. The TB is often lined with inflammatory tissue. At this point the goal is to remove proliferative tissue until the bony septum is visible. With a handheld otoscope and a peapod arthroscopic rongeur, grasp and remove soft tissue. Alternately, a small curette can be used to carefully loosen tissue from the septum. Smaller instruments that can be passed through the operative channels of the rigid scope or video otoscope are generally too small to be effective. The curette is then used to puncture the septum by placing progressive force in a caudoventral direction. A pop is felt as the VMC septotomy is successfully performed. Remove the curette and insert the rigid endoscope. A stream of mucus flowing into the DLC is a typical finding (Figure 20-34). This mucus can be collected for bacterial culture. The hole in the septum can be enlarged, or a second puncture can be created to allow easier access into the VMC (Figure 20-35). A Tomcat catheter is used to gently lavage the VMC. Even tenacious or caseated mucus can be removed with this method (Figure 20-36). The VMC is lavaged but not curetted. The TB is liberally lavaged with a saline flush until the returning fluid is clear. Carefully suction all fluid and debris from the pharynx at the conclusion of the procedure.
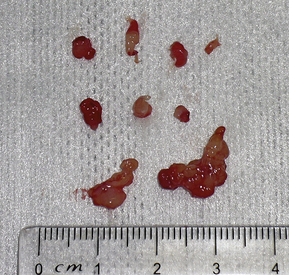
Figure 20-36 Polyps and proliferative tissue removed from the right ear canal and tympanic bulla of the cat (see Figures 20-32 through 20-35).
Potential Complications and Care after Treatment
Horner’s syndrome is the most common complication of this procedure, but it tends to resolve in most cases. If Horner’s syndrome persists, functional impairment is uncommon.5 Signs of otitis interna can result from curettage in the dorsal aspect of the bulla. Damage to the oval window can lead to head tilt, nystagmus, and ataxia. Always try to avoid this segment while removing proliferative tissue. Damage to the round window and/or auditory ossicles can cause deafness in the affected ear. Pharyngeal swelling from removal of a nasopharyngeal polyp or discharge from the ET can lead to obstruction and respiratory distress on extubation. We commonly administer a single dose of intra-operative corticosteroids to lessen postoperative nasopharyngeal swelling and edema. Otic medications are instilled immediately after final canal cleansing. Cats must be watched closely in the immediate postoperative period until awake and moving.
Cats are discharged with an otic steroid–antibiotic combination and an opioid analgesic (buprenorphine 0.015 mg/kg per os up to every 6 hours, as needed). Pending the results of bacterial cultures, we commonly use a mixture of fluocinolone acetonide with dimethyl-sulfoxide (Synotic) and levofloxacin 6.25 mg/mL, given at 0.2 mL twice a day in the affected ear(s) for 2 to 4 weeks on the basis of the subjective severity of the pathologic condition. Oral administration of postoperative antibiotics and corticosteroids is rarely needed with this approach. Reexamination is recommended near the end of medication duration. If mucopurulent discharge is observed in the bottom of the ear canal, exudate is cultured and the ear canal and TB are reevaluated under anesthesia. The canal is flushed and cleaned, and proliferative tissue is removed, if present. Consider VBO in refractory cases.
Polypectomy and TBC as described are minimally invasive and appear to be highly effective in the resolution of feline aural and nasopharyngeal inflammatory polyps. In our practice, recurrence rates are approximately 10 percent. Most cases of recurrence can be definitively managed by procedural repetition with appropriate culture-guided infection and inflammation control. In refractory cases, VBO is an effective treatment with excellent long-term results for patients that fail to respond to polypectomy and TBC.
1. Donnelly K.E., Tillson M.D. Feline inflammatory polyps and ventral bulla osteotomy. Compend Contin Educ Vet. 2004;26(6):446-453.
2. Veir J.K., Lappin M.R., Foley J.E., et al. Feline inflammatory polyps; historical, clinical and PCR findings for feline calicivirus and feline herpes virus-1 in 28 cases. J Feline Med Surg. 2002;4:195-199.
3. MacPhail C.M., Kudnig S.T., Lappin M.R. Atypical manifestations of feline inflammatory polyps in three cats. J Feline Med Surg. 2007;9:219-225.
4. Faulkner J.E., Budsberg S.C. Results of ventral bulla osteotomy for treatment of middle ear polyps in cats. J Am Anim Hosp Assoc. 1990;26:496-499.
5. Kapatkin A.S., Matthiesen D.T. Results of surgery and long term follow up in 31 cats with nasopharyngeal polyps. J Am Anim Hosp Assoc. 1990;26:387-392.
6. Anderson D.M., Robinson R.K., White R.A.S. Management of inflammatory polyps in 37 cats. Vet Rec. 2000;146:684-687.
7. Little C.J.L., Lane J.G. The surgical anatomy of the feline bulla tympanica. J Small Anim Pract. 1986;27:371-378.
8. Pope E.R., Constantinescu G.M. Feline respiratory tract polyps. In: Bonagura J., editor. Kirk’s current veterinary therapy XIII. Philadelphia: Saunders, 2000.
9. LeCouter R. Feline vestibular diseases—new developments. J Feline Med Surg. 2003;5(2):101-108.
10. Allen H.S., Broussard J.B., Noone K. Nasopharyngeal disease in cats: a retrospective study of 53 cases (1991-1998). J Am Anim Hosp Assoc. 1999;35:457-461.