Tenon's Cyst Formation, Wound Healing, and Bleb Evaluation
Tenon's Cyst Formation and Management
Anthony Wells
![]()
Introduction and Definition
The development of a Tenon's cyst, also known as an encapsulated bleb, is a complication of filtering surgery that requires careful management to ensure surgical success.
Use of the word ‘cyst’ in bleb terminology may be confusing. A bleb that is ‘encysted’ by a thickening of Tenon's tissue is thick-walled, and often, but not always, associated with raised intraocular pressure (IOP). In contrast, the entity that most glaucoma surgeons refer to when they describe a bleb as ‘cystic’ is a thin-walled, avascular bleb. Thin-walled blebs are more often associated with low IOPs than high, carry their own risks and complications as described in Chapters 81, 82, and 83, and appear frequently in the literature since the advent of widespread antimetabolite use.1–3 Some reports from the 1960s may refer to Tenon's cysts as ‘cystic’ blebs,4–6 in contrast to current usage which was initiated by Van Buskirk in 1982.7 Tenon's cysts have also been referred to as ‘exteriorization of the anterior chamber,’ and ‘encysted bleb.’
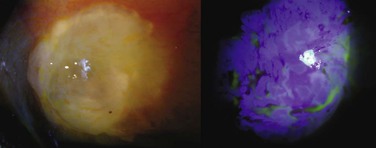
A practical definition of a Tenon's cyst, or encapsulated bleb, is a dome-shaped, elevated, tense, thick-walled, demarcated area centered over the trabeculectomy site. These tend to occur between 2 and 8 weeks after surgery.
In the current literature, definitions vary. All authors agree that a bleb with Tenon's encystment is dome-shaped in cross-section, thick-walled, and elevated; these are the critical signs. There are, however, limitations to the current morphologic definition: some Tenon's cysts have thick, opaque, highly vascularized walls, whereas others have thick but hypovascular walls. Both may be described within the spectrum of the encysted bleb; however, their fates may be quite different.
Whereas for most authors the diagnosis is made on purely morphologic grounds as described above, some authors8 include a functional component: raised IOP. While this approach arguably identifies those most at risk for developing progression of glaucoma and/or bleb failure, it might also fail to identify those whose IOP might be fluctuating, or where raised IOP is imminent. While an encysted bleb is unlikely to have a low IOP, there is probably a significant under-recognition of those with normal intraocular pressures, as noted in the Advanced Glaucoma Intervention Study (AGIS) where at least 50% of eyes with encysted blebs did not have raised IOP;9 the importance of such under-detection is unknown.
Some authors also choose to include a time component to the diagnosis, for example limiting the diagnosis to a 2–6-week post-trabeculectomy window. While this is certainly the time at which Tenon's cysts are most likely to occur, placing a morphologic diagnosis in a rigid time window is unnecessarily arbitrary. Tenon's cyst morphology may be identified many weeks following trabeculectomy, or after other interventions, for example cataract extraction in those with a previously functioning bleb.
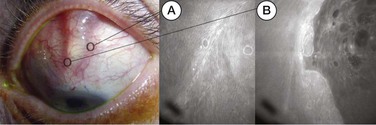
Most descriptions in the literature of Tenon's-cyst-type blebs do not attempt to describe peripheral drainage (mixed morphology blebs with central demarcated and peripheral diffuse components, Fig. 78-1-1) or ‘microcysts’ in adjacent or overlying conjunctival tissue, and it is unknown whether such presumptive evidence for trans-cyst-wall flow bears any relationship to eventual outcomes. Newer methods for assessment, such as new bleb grading schemes that can cope with blebs that have regional differences in morphology, may eventually provide insight into these issues.
Tenon's encystment of aqueous drainage reservoirs also occurs around glaucoma drainage devices (see Chapter 103), where it becomes an important guard against overdrainage and hypotony. It may also occur after nonpenetrating surgery (see Chapter 88) where Tenon's is generally left intact and antimetabolite use is less routine.
Incidence and Risk Factors
The incidence of Tenon's cyst after trabeculectomy has been reported between 6% and 29%.10,11 In male patients, where the risk is higher, it has been reported in as many as 48% of trabeculectomy cases by Campagna et al.11 The incidences reported in various studies need to be interpreted in view of the variable definitions and likely under-identification described above. Even when, as reported by the AGIS group, a raised IOP is not required for diagnosis, under-identification is still possible as attention may not be drawn to these cases9 and there is a wide range of possible bleb appearances consistent with the core definition of a Tenon's cyst.
Well-established risk factors include gender, with approximately twice the risk of Tenon's cysts in male patients as in females, and generally high statistical significance levels in most studies.9 Argon laser trabeculoplasty (ALT) has been implicated in several studies, but not all.11
Other implicated factors include the previous use of β-blockers,12 but these have not been supported in larger, more recent studies.9,11 Feldman et al.8 found higher rates of Tenon's cyst formation after sympathomimetic use; however, this has also not been identified since. Feldman et al.8 also identified an increased risk in patients with a history of Tenon's cyst formation in the fellow eye, which has not been further studied; logically, one would expect that this would be a significant risk factor for the second eye. The AGIS collaborators also found a relationship between the risk of developing a Tenon's cyst and educational status,9 which remains unexplained and may be a statistical anomaly.
Several factors could logically be linked to the risk of developing Tenon's cyst, but currently have no supporting evidence to refute or confirm associations. Surgical technique ought to play a role, in particular the excision of Tenon's and the use of antimetabolites. Counter to what might have been expected, the only report on Tenon's cyst formation that involves a large cohort of mitomycin C (MMC)-treated trabeculectomies had a higher rate of encapsulation than other reports.11 Limbus-based conjunctival flaps may also predispose to encapsulation according to some glaucoma surgeons, but this is unsubstantiated.13
Rates of glaucoma filtering surgery failure have been correlated with race, and similar mechanisms are presumably involved in Tenon's cyst formation. However, no studies to date have found a correlation between Tenon's cyst formation and race.
Etiology and Pathophysiology
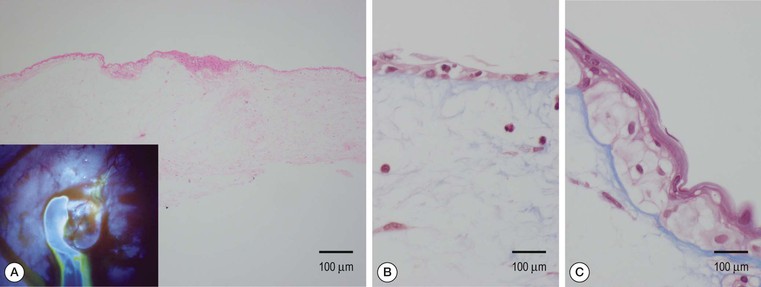
Histologic examination of Tenon's cysts7 reveals an inner wall of the cyst lined by low-cellularity fibrous connective tissue (Fig. 78-1-2), without an epithelial lining, and the bulk of the wall consists of collagenous tissue with multiple fibroblasts and occasional areas of lymphocytic infiltration. The outer wall is composed of loose connective tissue and the overlying conjunctiva.
The relationship of the wound healing response and scar tissue formation to failure of trabeculectomy is well established.14–16 It is less well understood why some patients have a healing response that gives the morphologic appearance of encapsulation, but probably involved are variations in tissue responses to trauma, aqueous cytokine profiles, and possibly multiple other factors, including surgical technique.
Preventative Measures
Antimetabolite Use and Surgical Technique
Since the presumed etiology of Tenon's cyst formation hinges on wound healing responses after trabeculectomy, factors such as antimetabolites that inhibit healing responses might be expected to result in altered rates of Tenon's cyst formation.
Surgical technique, particularly the conjunctival incision type, has been implicated in bleb morphology outcomes in some studies.13,17 Although there are currently no studies that address Tenon's cyst formation outcomes resulting from different incision types, there is limited anecdotal support for decreased numbers of Tenon's cysts after fornix-based conjunctival flaps. It is noteworthy that none of the eyes in AGIS that received antifibrotics developed encapsulated blebs, although only 21 eyes out of a total 789 eyes (2.6%) received antimetabolites.
Management Options
Investigations (Imaging)
There are no investigations specific to Tenon's cyst formation, and the diagnosis is made clinically. Simple bleb photographs by a standardized technique (e.g. as seen at www.blebs.net)18 may be useful for clinical studies, particularly for application of bleb grading systems by an independent observer.
Newer imaging modalities, in particular infrared optical coherence tomography (OCT) instruments, are able to reveal details of bleb morphology that may not be visible to the naked eye,19–24 but the role of these in clinical management is currently undefined. Confocal imaging, both reflectance25 and fluorescence26 (see Fig. 78-1-2) modes, is able to show ultrastructural details at cellular level, but the large degree of regional variation may make interpretation difficult, and the role of these instruments is also currently undefined.
When to Intervene
Raised IOP following trabeculectomy surgery may be due to a range of causes, and before considering management of a Tenon's cyst it is important to (1) confirm the diagnosis and (2) decide whether any treatment is required.
The diagnosis is confirmed by eliminating other potential causes for raised IOP such as: obstruction of the inner ostium by tissue, blood, or vitreous; and underfiltration because the scleral flap is too tight, primary failure of filtration as might occur if the sclerostomy is incomplete, and simple bleb fibrosis in the absence of a Tenon's cyst. Examination of the sclerostomy with a goniolens is an essential part of the work-up of an eye with raised IOP post trabeculectomy, and the internal sclerostomy is visibly open in eyes with encapsulated blebs. A mass of fibrotic tissue overlying the trabeculectomy flap may closely resemble Tenon's encapsulation, but may be distinguished by transillumination using a narrow slit-lamp beam or, with a blunt forceps tip or similar instrument after topical anesthetic, the characteristic texture of the fluid-filled structure is usually easy to distinguish from a solid mass of scar tissue. Anterior segment OCT or ultrasound biomicroscopy will also differentiate a solid from a cystic mass, but these are usually not necessary. Primary bleb failure and simple bleb fibrosis without Tenon's cyst formation do not have the characteristic dome shape of encapsulation.
No IOP-lowering treatment may be required if the IOP is in an acceptable range. Such a decision will be based on the glaucoma status of the eye, the desired target IOP, and hence the estimated vulnerability of the optic nerve to further damage. If IOP is considered to be too high or there are other Tenon's cyst-related complications, such as dellen, the surgeon should consider additional medical and/or surgical management. A Cochrane database review found only one small, randomized trial of 25 eyes comparing bleb needling to medical management of bleb encapsulation and concluded that the success rate for the medically managed group was significantly higher than the needled bleb group.27
Medical and Other Conservative Treatment
If required, short-term medical control of IOP is usually achieved with aqueous suppression, using carbonic anhydrase inhibitors such as brinzolamide (Azopt®) or dorzolamide (Trusopt®), α-adrenergic agonists such as brimonidine (Alphagan®) or β-receptor blocking agents such as timolol (Timoptol®) or levobunolol (Betagan®). Since it is aqueous flow that maintains a bleb, there is a theoretical risk that decreasing aqueous flow early after filtering surgery may decrease bleb size and increase the chance of bleb collapse or fibrosis.
Since the underlying process of Tenon's cyst formation is one of fibrosis, inhibition of fibrosis by suppression of inflammation, fibroblast activity, and hypervascularization is normally attempted. There are multiple approaches described, but these generally hinge on the use of topical steroids such as prednisolone acetate. Others have recommended stopping all steroids in the presence of a Tenon's cyst.28
Options primarily directed against fibroblast function, such as 5-fluorouracil (5-FU) and mitomycin C (MMC), probably also have a role, although this is poorly defined at present. Subconjunctival injections of 5-FU are commonly used after trabeculectomy as an antiscarring agent29 and may decrease fibrotic wound healing responses in filtering blebs (Fig. 78-1-3). MMC, which has more potent effects on fibroblast function and survival, has been described in a similar role either given as a subconjunctival injection30 or applied transconjunctivally.31 Some surgeons are reluctant to give subconjunctival injections of MMC because of concerns about adverse long-term outcomes, such as scleral melting, scleral ischemia, or damage to underlying ocular tissues. More information about the use and actions of 5-FU and MMC can be found in Section 16.
Alternative approaches include the systemic use of steroids, colchicine, and nonsteroidal anti-inflammatories as described by Molteno et al.,32 although this is either not commonly used, or saved as an option when all else has failed, given the potential systemic risks. The use of antivascular endothelial growth factor (VEGF) treatment has been reported33 for Tenon's cyst treatment.
Most textbooks and many authors of scientific reports describe digital massage, either by the surgeon at the slit lamp or by the patient at home, in detail.34–36 Transiently increasing the IOP to high levels by globe massage may ‘hydrodissect’ channels for aqueous flow through the Tenon's encapsulation and free scarred areas of conjunctiva. The resulting transient IOP elevation is unlikely to be dangerous, so there is probably little harm in trying this approach, although at present there are only anecdotal reports of improvement in outcomes resulting from globe massage. The most commonly described technique is to have the patient massage the inferior part of their closed and supraducted eye via the inferior eyelid firmly with two fingers for up to a minute.
If a steeply angled limbal bleb surface disrupts tear film coverage of the adjacent peripheral cornea, dellen may result. Management of this may be conservative, as one would treat dellen from any cause, using lubricants and topical antibiotics as indicated. Resolution of the abrupt change in limbal contour, via successful management of the Tenon's cyst, will result in resolution of dellen. In some cases, especially if IOP is difficult to control, persistent dellen may be sufficient reason to embark on surgical management.
Surgical Treatment
Needling.
Using a needle and adjunctive compounds including antifibrotic medications and steroids to treat Tenon's cyst is well established.37,38 This is performed in carefully selected patients by some surgeons at the slit lamp in the clinic, or alternatively under general or regional anesthetic. General anesthetic (GA) is most appropriate if the patient is a ‘squeezer’ or has difficulty with being still; all of the disadvantages of a GA are present, but excellent control of the eye and procedure is afforded, and the lack of iatrogenic subconjunctival fluid (as might occur following sub-Tenon's or peribulbar anesthetic) results in a great view of bleb extension in response to breaking the barrier to aqueous flow.
Pretreatment with weak adrenergic agonists, such as adrenaline 0.1% or alternatively a 50% solution of 2.5% phenylephrine in balanced salt solution or tetracaine, will constrict conjunctival blood vessels and decrease the risk of subconjunctival hemorrhage. Blood in the bleb area is not only distressing for the patient, but will also contain cytokines that are likely to promote bleb fibrosis and increase risk of surgical failure.
The general approach to needling a Tenon's cyst is to enter the conjunctiva some distance from the trabeculectomy site while avoiding blood vessels, advance the needle tip in the loose subconjunctival tissue towards the sclerostomy, and to then incise or perforate the posterior wall of the cyst with the needle tip (Fig. 78-1-4 and Video 78-1![]() ). There are multiple subtle variations according to individual preference: some surgeons prefer to make an individual puncture in the cyst wall with the idea that bleeding may be less likely, some like to make multiple punctures, and others prefer to make a large linear cut by moving the bevel of the needle in a sweeping motion. For needling of bleb encapsulation where the blockage to aqueous flow is peripheral to the scleral flap, it is not usually necessary to enter the anterior chamber with the needle tip, which improves the safety of needling in this situation. A conjunctival suture is generally not required, since the small entry site is well away from the sclerostomy and closes quickly.
). There are multiple subtle variations according to individual preference: some surgeons prefer to make an individual puncture in the cyst wall with the idea that bleeding may be less likely, some like to make multiple punctures, and others prefer to make a large linear cut by moving the bevel of the needle in a sweeping motion. For needling of bleb encapsulation where the blockage to aqueous flow is peripheral to the scleral flap, it is not usually necessary to enter the anterior chamber with the needle tip, which improves the safety of needling in this situation. A conjunctival suture is generally not required, since the small entry site is well away from the sclerostomy and closes quickly.
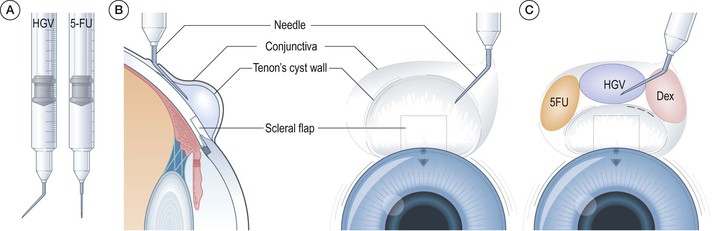
Many surgeons feel that a smaller needle, such as 29-gauge or 29.5-gauge, gives sufficient flow while minimizing the risk of overdrainage and hypotony. Others may feel that a larger aperture resulting from a 23-gauge to 25-gauge needle is more likely to stay open, especially in the cases of a very thick encapsulation. Although literature on these decisions is sparse, there is evidence to suggest that if the IOP is below 12 mmHg the day after the needling, the needling is more likely to succeed.38
It is usual to give adjunctive treatment with needling procedures. Most surgeons will give subconjunctival dexametasone and/or 5-FU; some use MMC, typically transconjunctivally. Another suggested alternative is use of 1 mg of bevacizumad (avastin) with needle revision.39 Given the high pH of 5-FU, great care must be taken to avoid this entering the anterior chamber; 5-FU should be injected slowly with the needle tip well away from the sclerostomy. The anterior chamber should be observed during 5-FU injection and if 5-FU is seen to enter the anterior chamber in bulk, it should be immediately irrigated with balanced salt solution via a paracentesis.
The author prefers to use Healon GV (HGV) loaded into an insulin syringe, with an approximately 135° bend 6–10 mm from the bevel of the 29-gauge needle. This syringe and needle combination works well from a mechanical perspective, giving good access and angulation while allowing a surgeon with average hand size to perform the needling and depress the plunger with one hand. The HGV viscodissects adherent Tenon's and temporarily maintains the bleb space, prevents leakage of 5-FU from the needle entry to the ocular surface, avoiding epithelial toxicity, and the HGV–5-FU combination forms a depot of slow-release 5-FU.
Complications from needling procedures are relatively less than those of full incisional surgery to manage IOP, and include subconjunctival and anterior chamber hemorrhage, inadvertent buttonhole via needle tip perforation of the central bleb, accidental damage to the lens or corneal endothelium, hypotony, corneal epithelial toxicity from repeated 5-FU injections, and a low risk of infection. By far the most common complication is failure to control IOP, although needling procedures can potentially be repeated several times and the chances of success may be cumulative.
An alternative approach of using Nd: YAG laser instead of a needle to perforate the Tenon's encapsulation was described by Ofner and Smith in 1988,40 but is not widely used.
Surgical Excision.
Opening the conjunctiva and surgically removing the dome-shaped encapsulation tissue should be considered a procedure of last resort in somebody who has vision-threatening visual field loss, an uncontrolled IOP, and has not responded to other management options. The usual technique is to elevate the conjunctiva and dissect it free from the Tenon's cyst surface (the author prefers a limbal incision and fornix-based conjunctival flap approach, although posterior approaches are also described and effective41), open and remove the top of the dome, and then close the conjunctiva in a watertight fashion. This approach may also remove resistance to aqueous flow and result in loss of IOP control, hence post-procedure hypotony and the complications that may result are common. Tenon's cyst may also recur following excision.
Prognosis
In general terms, the prognosis for IOP control following Tenon's encapsulation is excellent,42 although a frequent finding is that complete success rates without medications are significantly decreased after conservative treatment.11,42 In other words, long-term IOP-lowering medications are more likely to be required. Reports of success rates for conservative management alone range from over 70% to 100% (Fig. 78-1-5).35,43,44 but one retrospective study showed a significantly higher failure rate at a mean of 2.5–3 years for patients receiving either needling or medical treatment for bleb encapsulation compared to control, non-encapsulated consecutive eyes at the same institution.45
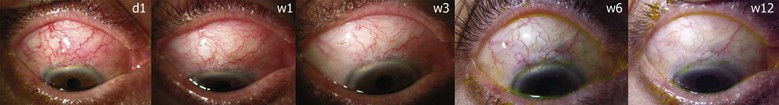
A lack of consistent definition, as alluded to above, does preclude accurate prognostic information for subtypes of Tenon's cyst, for example thick opaque domes may be destined for eventual fibrosis and failure, while more translucent dome-shaped areas of central bleb demarcation may eventually become thin and avascular. Further research using better bleb assessment tools18 should improve our understanding of these variants.
Long-term bleb morphology outcomes have not been described for either conservatively managed or more aggressively managed Tenon's encapsulation. A proportion may proceed to thin and avascular central blebs, which carry their own risks, including bleb-related endophthalmitis and chronic leaks with hypotony. In many cases the fibrous encapsulation may resorb, leaving diffuse bleb morphology and a controlled IOP. Others may simply scar down and fail, with loss of IOP control. The predictors of these varied outcomes are not well understood at present.
If the Tenon's cyst does eventually progress to trabeculectomy failure, IOP control is lost and there is risk of glaucoma progression. Late needling procedures may be successful, but frequently repeat surgery is necessary.
Wound Healing and Bleb Evaluation after Trabeculectomy
Anthony Wells, Tina T Wong, Jonathan G Crowston
The Wound Healing Response
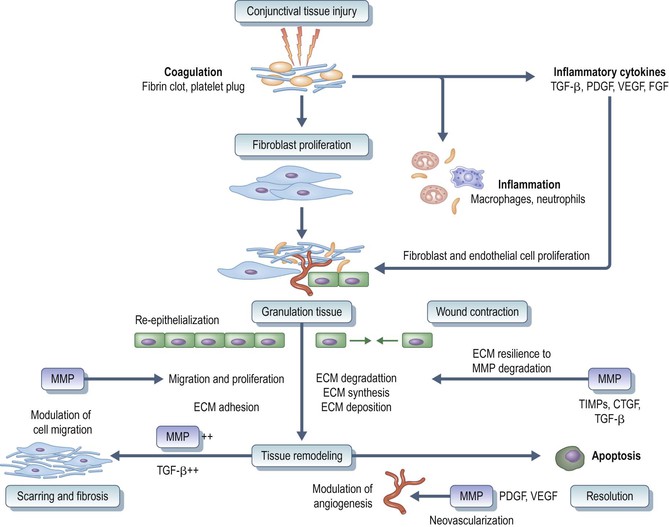
The wound healing process is a key determinant of the final intraocular pressure after trabeculectomy and glaucoma drainage device surgery (see also Chapter 113). Modulation of the postoperative wound healing response is often required in order to achieve adequate intraocular pressure lowering and to maximize surgical success.
Wound repair is a physiological event, whereby tissue injury induces a repair process that finally leads to the restoration of tissue structure and function. The wound healing response involves several interconnecting cellular pathways (Fig. 78-2-1). Surgical trauma to conjunctival, episcleral and iris tissue leads to the leakage of plasma proteins into the filtration site, precipitation of the clotting cascade, and complement activation. The fibroblast is the effector cell responsible for subconjunctival scar formation.1 Platelets aggregate and are incorporated into an insoluble fibrin mesh, forming a blood clot. This ensures hemostasis and provides a provisional matrix for the migration of inflammatory cells and fibroblasts.1,2 Wound healing can be divided into three overlapping phases: (1) the inflammatory phase; (2) the proliferative phase; and (3) the remodeling phase.
The Inflammatory Phase
Inflammation is a localized protective response to damage permitting preservation of structural integrity and cellular function. Neutrophils and monocytes are recruited to the wound during early inflammation. Neutrophils are evident at the wound site within minutes of wound formation3 and peak around day 2 in subconjunctival tissues.1 Proteolytic enzymes, collagenase, and elastase are released by activated neutrophils, aiding their penetration of the endothelium basement membrane. Neutrophils phagocytose contaminating bacteria, and clean the wound site by protease-mediated digestion of extracellular matrix.
Macrophages are derived locally or from circulating monocytes, which differentiate into tissue macrophages. Monocyte accumulation is driven by the presence of monocytic chemoattractants, including collagen fragments and transforming growth factor-β (TGF-β).4 Blood-borne monocytes enter the wound and bind to the extracellular matrix via integrins. Macrophages contribute to tissue debridement and phagocytosis and secrete a variety of soluble factors required for tissue formation and the transition from an inflammatory response to tissue repair (Fig. 78-2-2). Tissue macrophages are a major source of proinflammatory growth factors. These include platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), epidermal growth factor (EGF), and TGF-β.
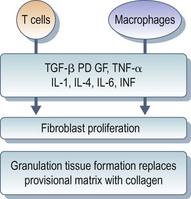
Unlike neutrophils, macrophages are essential for normal wound healing. They play important roles in orchestrating the wound healing response both by altering the cytokine environment and by interacting with lymphocytes and fibroblasts. Macrophages are necessary for the normal initiation and maintenance of fibroblast activity.5,6 Macrophage-depleted wounds show a delay in the formation and reduction in the quantity of granulation tissue.7
Immunohistochemical studies of a rat model of conjunctival healing after trabeculectomy revealed extensive macrophage infiltrates throughout the healing process, suggesting that these cells also participate in conjunctival healing.8
T lymphocytes are essential for normal wound healing events.6,9 A dual role has been proposed where T cells stimulate fibroblasts, macrophages, and endothelial cells in the early stages, but downregulate healing in the later stages.10
The Proliferative Phase
The proliferative phase results in reepithelialization and the formation of granulation tissue from fibroplasia and neovascularization.
Reepithelialization.
As conjunctiva is resutured to cover the sclerostomy and subconjunctival tissues at the end of surgery, healing occurs by primary intention at the conjunctival–corneal interface in trabeculectomies with fornix-based conjunctival incisions. Although these defects are small, they can be of important clinical significance, as insufficient healing at this interface is a common cause of postoperative bleb leak.
Reepithelialization of the wound begins within hours of tissue injury and involves epithelial cell migration over the wound edge. First, the conjunctival epithelial cells differentiate into a more motile phenotype. Modifications include the loss of hemidesmosomal links between the epidermis and basement membrane,11 alterations in integrin expression,12 and the formation and assembly of intracellular α smooth-muscle actin filaments.13 After an initial lag of 1–2 days, epithelial cell proliferation will occur at the wound edge and provide more cells for migration.
Granulation Tissue Formation.
Granulation tissue formation is initiated by the release of growth factors synthesized by platelets, injured cells, and macrophages. The new matrix consists of loose connective tissue, fibroblasts, new blood vessels, and macrophages. Macrophages now secrete cytokines to stimulate fibroplasia and angiogenesis, whereas fibroblasts remodel the extracellular matrix to aid cell migration and proliferation. Angiogenesis leads to new blood vessel formation and provides necessary oxygen and nutrients for cell metabolism.
Angiogenesis.
Angiogenesis is initiated within days of wound formation in response to low oxygen tension and lactic acid build-up that follows from tissue trauma and surgical manipulation. Vascular endothelial cells proliferate to produce capillary buds, which network to form a capillary bed. Proangiogenic factors including vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) are secreted by inflammatory cells, particularly macrophages and platelets. Blocking antibodies to these two growth factors almost completely inhibits wound angiogenesis.14
Fibroplasia.
Fibroplasia is the deposition of extracellular matrix by large numbers of fibroblasts that have entered the wound site. Fibroplasia is initiated by growth factors, such as TGF-β and PDGF, that stimulate extracellular matrix production, fibroblast proliferation, and the differentiation of fibroblasts into myofibroblasts. Not all fibroblasts undergo differentiation, as in vitro studies have shown that successful fibroplasia depends on the presence of the fibroblast, rather than the myofibroblast.15 Myofibroblasts show altered responses to cytokines and are characterized by the presence of intracellular microfilament bundles containing α smooth-muscle actin (ASMA), the actin isoform expressed by smooth muscle cells.16 Myofibroblasts assist in wound closure by wound contraction and extracellular matrix production.
In addition to chemotaxis to soluble factors, fibroblasts are attracted to the wound by contact guidance and haplotaxis. Haplotaxis is the process of fibroblast migration up a surface-bound adhesion gradient, while contact guidance is migration along discontinuities in the matrix to which they are attached. This highlights the interdependence between fibroblasts and the extracellular matrix. While fibroblasts synthesize and remodel the extracellular matrix, the matrix in turn regulates fibroblast motility.
Fibroblast movement into and through the fibrin clot requires proteolytic cleavage of the extracellular matrix. As fibroblasts migrate over the fibronectin interface, traction forces are established with the underlying substrate inducing wound contraction. This process is facilitated by the release of matrix metalloproteinases (MMP).17 This family of enzymes degrade extracellular matrix, providing a ‘path’ for fibroblast movement. MMPs are secreted as inactive proenzymes that require activation to initiate extracellular matrix degradation. In addition, tissue inhibitors of matrix metalloproteinases (TIMPs) inhibit MMPs. The ratio of MMP to TIMP determines the balance between tissue degradation and extracellular matrix synthesis.
Fibroblasts bind to various components in the matrix, which include fibrin, fibronectin, and vitronectin through the use of cell-binding domains of the matrix proteins via integrins.18 Syndecans, transmembrane heparin sulfate proteoglycans, can also mediate cell–matrix interactions via the heparin-binding domain of fibronectin.19 This binding domain is structurally distinct from the integrin domain. These and other cell-binding domains collectively regulate the strength of cell binding and hence cell motility.20
Collagen production is stimulated by the autocrine action of TGF-β and paracrine interleukin-4 (IL-4) from mast cells.21 Collagen is the major component of the wound matrix in animal models of glaucoma filtration surgery.2,22
Remodeling Phase
Although classified as the final stage of wound repair, matrix reformation and remodeling overlaps with granulation tissue formation. However, unlike granulation tissue synthesis, this stage continues for many months after wounding. This final phase is characterized by matrix remodeling, cell differentiation, maturation, and apoptosis. As the wound is remodeled, fibroblasts differentiate into myofibroblasts. Once the wound matures and restoration of tissue structure occurs, fibroblast and myofibroblast numbers are reduced through the induction of apoptotic cell death.23,24
Wound Remodeling.
Extracellular matrix (ECM) remodeling defines the process where coincident ECM synthesis and breakdown refine the wound matrix. Plasminogen activators and matrix metalloproteinases are the chief mediators of ECM degradation. Remodeling initially consists of hyaluronan and fibronectin removal. Maturation of the remodeling phase is associated with deposition of proteoglycans and the replacement of collagen type III with collagen type I, which occurs synchronously with a reduction in fibroblast numbers. This reflects the transition of a matrix favoring cell proliferation and migration to that of high tensile strength and resistance to deformation.
Apoptosis.
A key event in the conversion of highly cellular granulation tissue to a less cellular scar is fibroblast death by apoptosis. Fibroblast apoptosis has been demonstrated in skin, kidney, and lung wounds.23,25,26 The molecular pathways that induce fibroblast apoptosis are not fully understood. A reduction in mechanical tension of connective tissue has been proposed. Grinnell and colleagues have demonstrated that the release of mechanical tension is a trigger for fibroblast apoptosis in cell culture experiments. Fibroblasts in a mechanically loaded collagen matrix demonstrated little apoptosis whereas apoptosis was rapidly induced when the tension within the gel was released.27 The antifibrosis agent mitomycin C has also been shown to induce Tenon's fibroblast apoptosis in cell culture, suggesting that the induction of fibroblast apoptosis may be a mechanism whereby this cytotoxic agent limits scar formation post trabeculectomy.28,29
Precise regulation of fibroblast death is a critical factor in the terminal stages of the healing response.29 Fibroblast apoptosis must not occur too prematurely to ensure that the defect is repaired, but also should not be delayed in order to limit functional impairment that would result from excess scar formation and wound contracture.
Apoptotic failure results in prolonged fibroblast survival and excess scar production.24 Analysis of biopsies taken from keloid and hypercellular scars in human skin revealed persistent fibroblastic activity and inflammation for up to 10 years following the original surgery. This differed from normal scars, where biopsies taken 1 year after wound formation displayed minimal evidence of fibroblast activity.30 In contrast, excessive or premature fibroblast apoptosis can result in insufficient wound healing. The impaired healing observed in full-thickness skin wounds of non-obese diabetic (NOD) mice was associated with higher levels of fibroblast apoptosis compared to identical wounds in control mice.31 Fibroblast apoptosis has not been demonstrated directly in subconjunctival wound healing in vivo. The dramatic reduction in fibroblast numbers observed in a rabbit model of glaucoma filtration surgery, 31 days after surgery, may be a consequence of apoptosis.22
Clinical Assessment of the Healing Bleb
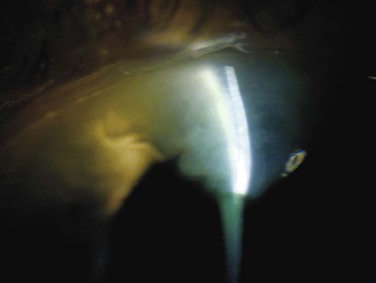
Bleb morphology impacts on IOP lowering and the risk of postoperative complications and therefore is an important determinant of the long-term success of trabeculectomy. After weeks to months, remodeling of the sclerostomy and scleral flap area results in establishment of a fistula (Fig. 78-2-3) between the anterior chamber and the sub-Tenon's space through which aqueous flows. In the vast majority of cases, the amount of resistance to aqueous flow, the facility of outflow, and IOP are determined not by the fistula but by characteristics of the sub-Tenon's–episcleral fascia interface, here referred to as the bleb tissues. Although good published evidence is awaited, it is very likely that early recognition of undesirable patterns of healing in bleb tissues could guide interventions to optimize surgical outcomes.

If IOP control is poor and the bleb seems to be failing, it is important to verify that the changes result from the bleb itself, not from obstruction proximal to the bleb, such as an obstructed internal ostium.
It is also important to avoid placing too much emphasis on IOP when assessing bleb morphology and function in the early postoperative period. There may be low ciliary body aqueous production from inflammation, choroidal detachments, effects of inadvertently used topical medications, or crossover effects of fellow eye or systemic medications. Over-reliance on IOP to guide bleb management will therefore be misleading soon after surgery. Similarly, high IOP and a low bleb early after surgery are probably from a scleral flap that is too tightly sutured, and the sutures may require adjustment,32 laser lysis, or removal, which will reestablish flow and expand the bleb.
Bleb Assessment Criteria
Vascularity.
Increased bleb vascularity, especially in the first weeks after surgery, has been associated with surgical failure.33,34 Increased vascularity results from inflammatory mediators, dilation, and proliferation of local vascular networks. In the current literature, bleb vascularity usually refers to a general assessment of vascularity over the central bleb area. It is, however, becoming clear that specific regional areas of increased vascularity are also important: for example, hypervascularity of the tissues surrounding the central bleb area, 6 weeks after trabeculectomy with limbus-based conjunctival flap, was associated with a sixfold increased risk of bleb failure.35
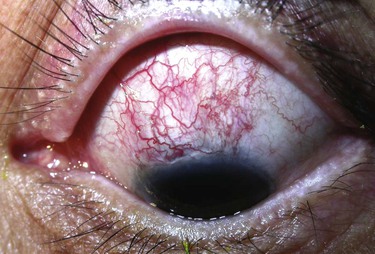
Corkscrew Vessels.
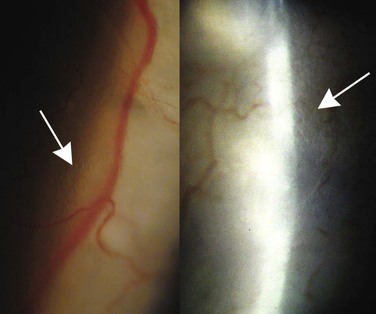
The presence of corkscrew vessels (Fig. 78-2-4) in the early weeks following glaucoma filtering surgery denotes an increased risk of surgical failure by scarring,33 and this may be independent of the degree of vascularity.36 Although the process is not well understood, it is likely to involve contraction of Tenon's tissue caused by migration of fibroblasts,37,38 which produces a concertina effect on an existing, often dilated, vessel. Corkscrew vessels therefore probably signify recruitment of fibroblasts to the bleb, ongoing wound healing, and contraction of the bleb space, all of which may lead to bleb failure.
Dragged Vessels and Conjunctival Suture Line Contraction.
Dragging or straightening of conjunctival vessels (Fig. 78-2-5) also represents fibroblast-mediated tissue contraction.37 The dragged vessels usually appear peripheral to the bleb space and the advancing line of fibrosis anterior to them typically decreases bleb size and function. In blebs formed from a limbus-based conjunctival flap, with a prominent and inflamed suture line, contraction of the suture line (Fig. 78-2-6) heralds shrinkage and possibly disappearance of the bleb. This process is slow, and unless efforts are made to recognize it by taking measurements or photographs, is typically not noted; if the bleb is thin-walled, IOP will remain low until very late in the process.
Elevation.
With the exception of flat or nonexistent blebs, lower blebs tend be associated with better long-term outcomes. A low bleb with large surface area without focal thinning or demarcation by fibrotic tissue tends to give excellent IOP control; hypovascular thin-walled ‘cystic’ blebs are undesirable as discussed below. In Tenon's cyst, or bleb encapsulation, with thickened bleb walls, bleb height is typically increased,34 occasionally to dramatic levels. The amount of elevation may or may not reflect IOP, but in a significant proportion of encapsulated blebs the IOP is raised because aqueous cannot easily diffuse through the thickened bleb wall, and this may be associated with poor bleb outcomes.33 The contour by which the maximum elevation is reached also has an influence on the success of surgery. A steep contour at the anterior edge of the bleb may produce areas of bleb-edge tear film bubbles, best seen immediately after a blink (Fig. 78-2-7) and these areas of nonwetting can predispose to dysesthesia and dellen formation.
Bleb Area and Extent.
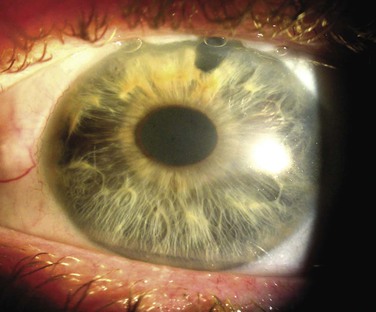
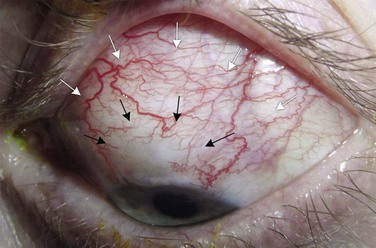
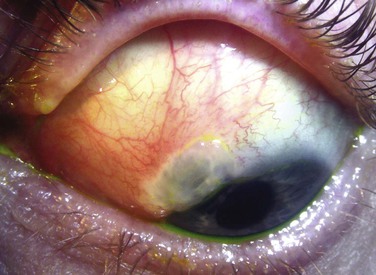
There is a complex interrelationship between bleb wall thickness, transmural permeability to aqueous, the area of the bleb, and IOP. In general, large bleb areas are desirable and associated with good IOP control and a lower risk of long-term complications. There are, of course, exceptions. A small-area, thin-walled bleb with high transmural flow, which at the extreme might become a ‘sweating’ or leaking bleb, will usually produce a low IOP. A very large encapsulated bleb can have a huge surface area, but high resistance to transmural flow and thus high IOP. It is helpful to distinguish between separate bleb areas; traditional descriptions of blebs do not allow for the possibility of mixed-morphology blebs,39,40 such as a central thin-walled demarcated area surrounded by a large area of diffuse conjunctival elevation (Fig. 78-2-8). Whereas the localized or focal parts of the bleb are easy to discern, the full extent of diffuse conjunctival elevation may be more difficult. Using ultrasound biomicroscopy to measure the total area of the bleb, Jinza et al.41 reported an inverse relationship between bleb area and IOP. Blebs that extend circumferentially to the inferior bulbar conjunctiva (Fig. 78-2-9) are relatively common in recent times when antimetabolite use has become more widespread. These rarely result in any significant complications but frequently produce a mild foreign body sensation, and patients may notice that their eyes appear ‘wet.’ These eyes are also prone to spontaneous subconjunctival bleeds.42 Blebs may also extend onto the cornea. Such blebs may be asymptomatic but can also lead to dysesthesia and foreign body sensation (Fig. 78-2-10). Corneal blebs tend to be thin-walled and well demarcated posteriorly, typically with a scalloped anterior edge.

Wall Thickness.
Wall thickness is inferred from the degree of translucency or transparency. Wall thickness determines the amount of transconjunctival flow. Blebs that appear to be less translucent tend to have lower transmural outflow facility and higher IOPs, but this is not an absolute relationship36 since the arrangement of connective tissues should also alter permeability, as proposed by Maumenee in the 1960s.43 Few blebs have uniform wall thickness, so resistance to transmural flow will vary across the bleb. Aqueous flow will take the route of least resistance and most will pass through the most permeable part of the bleb area. It is not known whether thicker parts of the bleb with less transmural flow and therefore less exposure to potential pro- or antifibrotic factors in the aqueous are more likely to fibrose, and similarly whether thinner areas might be exposed to more wall tension and different cytokine compositions, which may therefore tend to further thin or reinforce the wall in this area. Whatever turns out to be the case, it is well known that blebs tend to remodel significantly over time.
Avascular Areas (‘Cystic Blebs’).
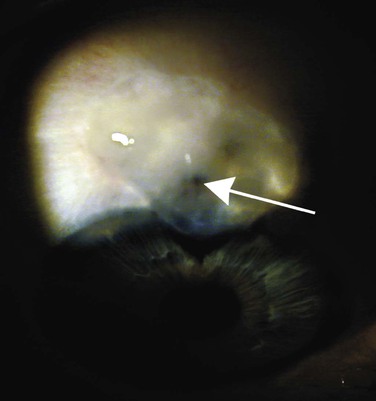
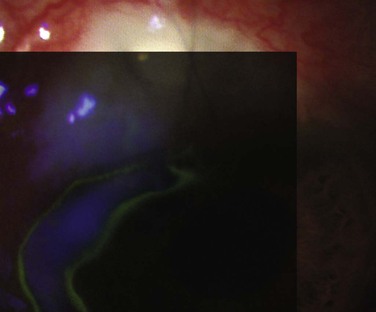
Although much of the literature in recent times has concentrated on the role of antimetabolites in producing thin-walled, leaking blebs, surgical technique also plays a significant role.44 Thin and avascular, or ‘cystic,’ bleb areas were common in full-thickness procedures (Fig. 78-2-11) prior to trabeculectomy, long before antimetabolite use entered glaucoma surgery.45 The importance of recognition of avascular and thin areas is that they predispose to many of the long-term complications of initially successful trabeculectomy including bleb leaks (Fig. 78-2-12), hypotony, dysesthesia, and bleb-related endophthalmitis (Fig. 78-2-13). Examination of these blebs over time, for example by serial photographs, shows that they are far from static and are continually remodeling themselves. The processes that drive remodeling are not well understood but mechanical stress,46 MMP activity,38 and fibroblast apoptosis probably all contribute.28,31
Subconjunctival Blood.
Blood contains multiple inflammatory mediators, and provokes inflammatory and healing responses all over the body. Blood is a nearly ubiquitous finding in the subconjunctival space immediately following glaucoma filtering surgery but minor blood presence is thought to have minimal impact on outcome. One might expect the impact of minor peri- and intrableb subconjunctival hemorrhage to be trivial, since cytokines and other mediators are not continuously released and there is dilution from aqueous flow. This is supported by a study by Sacu et al.36 Larger volumes of subconjunctival blood, enough to form a clot and persist, may provoke more local tissue response, provide a depot of inflammatory mediators, and also provide a scaffold for fibroblasts to bridge the sub-Tenon's space. Recurrent inferior subconjunctival hemorrhages in patients with blebs that extend to the inferior bulbar conjunctiva are potentially concerning but usually benign and self-limiting.42
Microcysts.
This is the sign most commonly associated with good bleb function (Fig. 78-2-14), and almost certainly reflects transconjunctival aqueous flow.47–49 Satisfactory IOP control and the presence of microcysts are consistently associated in the literature.33,36,50 In some very low, homogeneous, and diffuse blebs, the only signs of bleb function may be the presence of microcysts and the detection of a tiny optically empty space using a high-power, very narrow slit beam. Microcysts are usually not distributed evenly throughout the entire bleb area, which may reflect local variations in transmural flow. Although the relationship between bleb function and microcysts is strong, not all well-functioning blebs have visible microcysts, and microcysts can still be present when the bleb is underperforming and the IOP is very high. Microcysts are evidence of transconjunctival filtration and hence flow, but are not evidence of adequate bleb function.
Bleb Leak.
Bleb leaks are best detected by a variant of the Seidel test. The basis for this is that concentrated fluorescein, for example 2% solution or a moistened strip, does not fluoresce under cobalt-blue light, but dilute solutions will. After instilling topical anesthetic drops, the authors prefer to use a fluorescein strip, slightly dampened with more topical anesthetic and, holding the upper lid up in downgaze to expose the bleb, carefully apply the flat of the strip to suspect areas of the surface. In this way multiple leaks will not be missed, and mislocation of the leak because of high flow and intermittent ‘flooding’ can be avoided. Alternatively, a drop of 2% fluorescein can be applied, with the bleb surface well exposed, and the area observed for fluorescence as aqueous dilutes the high concentration of fluorescein in the tear film. If the IOP is very low, lack of flow may mask a leak; gentle digital pressure can produce some flow through the leak and make it visible, although low IOP is not just a result of a bleb leak, as discussed below.
Early Bleb Leak.
The viability of the bleb depends, at least in part, on its inflation by aqueous flowing from the anterior chamber, and so a watertight conjunctival closure at the end of surgery is imperative. Leaks from the wound in the first days after surgery provide a low-resistance alternative to aqueous egress via the sub-Tenon's space. If the leak allows bulk flow of aqueous, the rest of the bleb can collapse, allowing apposition of the inflamed inner bleb walls, which are likely to adhere. If such adherence is over most or all of the bleb area, bleb failure is probably inevitable. Most of the time, the leak is small enough that the bleb space is maintained, and these can be managed conservatively with good results, although early leak may be a risk factor for subsequent bleb failure (see also Chapter 75).51 Hypotony can occur in the presence of bleb leak, in particular where the scleral flap does not provide adequate resistance to flow.52 Early resistance to flow in the first weeks after surgery is largely due to resistance at the level of the scleral flap and related to scleral suture tension. However, if there is bulk flow from the anterior chamber because of poor scleral flap construction or closure, a weak conjunctival closure will often leak also; thus, hypotony is often seen in association with early postoperative bleb leaks. These are often confused. The decision to revisit the scleral flap should be made on IOP levels, and the decision to resuture the conjunctiva should be made on bleb elevation and extent.
Late Bleb Leak.
Bleb leaks after the first few weeks may rarely occur from unusual causes, such as conjunctival erosion over a retained suture end, but are almost universally from thin and avascular areas, usually referred to as cystic blebs (see also Chapter 81). The leak may be relatively slow, with the Siedel test appearance of a ‘sweating’ bleb, or fast enough for the patient to complain of intermittent or continuous watering. Once the fistula from the anterior chamber to the bleb is well established there is usually only a little resistance to flow into the bleb, so leakage from blebs late after surgery may result in hypotony. Bleb-related endophthalmitis, which may be rapid and devastating, is associated with late bleb leaks and thin bleb walls.
Bleb Grading Systems
Recognition that the success of glaucoma surgery depends on more than just the final IOP has generated increasing interest in quantifying and recording bleb characteristics33,53,54 for clinical studies. The ability to accurately and efficiently encode blebs for analysis ought to give better understanding of the natural history of adverse outcomes, and the ability to test interventions to avoid complications and optimize the results of trabeculectomy.
Since glaucoma surgery has been described, many authors have described blebs in recognizable patterns such as cystic, encysted, flat, and diffuse; sometimes with a global vascularity assessment. These descriptors are useful for describing common appearances but lack the depth of description required for following changes over time. Additionally, there is a vast range of possible variations within each of these descriptions. Other bleb classification or grading systems, such as that utilized by Kronfeld in 196955 and by Migdal and Hitchings in 1983,56 were useful, but had limitations, such as the assignment of a single vascularity grading for the entire bleb area, and coarse grading scales requiring a forced-choice assessment.54
Previous systems for bleb grading and classification have suffered from limitations in some combination of flexibility, reproducibility, or scale. The Indiana Bleb Appearance Grading Scale (IBAGS, 2003), described by Cantor et al.53 was a significant advance on previous systems because it isolated height, extent, and vascularity scoring. The IBAGS, however, does not describe vascularity of the bleb away from the central area, which may provide an important prognostic factor,35 and does not allow for the description of mixed-morphology blebs.40
Other bleb grading scales that have been developed include the system described by Picht and Grehn33 and the Moorfields Bleb Grading System (MBGS).57,58 The MBGS and the IBAGS are the most recent. Work comparing these systems for reproducibility and agreement has recently been published.40
In the MBGS (details and reference photographs available at www.blebs.net), four parameters are scored, and in addition area and vascularity are subdivided (Table 78-1).
Table 78-1
| Parameter | Symbol | Range | Represents | Notes | |
| IBAGS | Height | H | 0–4 | Flat (0) to high (4) | |
| Extent | E | 0–3 | <1 hour to >4 | ||
| Vascularity | V | 0–4 | Avascular to extensive | V1 = avascular and cystic | |
| Siedel | S | 0–2 | S0 = no leak, S1 = pinpoint leaks, S2 = streaming | ||
| MBGS | Central Area | 1a | 1–5 | 0% of photo area to 100% | Demarcated central area |
| Maximal Area | 1b | 1–5 | Total area elevated | ||
| Height | 2 | 1–4 | Low to high bleb | ||
| Central vascularity | 3a | 1–5 | |||
| Peripheral vascularity | 3b | 1–5 | Avascular to severe | 2 = ‘normal’ | |
| Non-bleb vascularity | 3c | 1–5 | |||
| Subconjunctival blood | scb | 0–1 | i.e. Yes or No; Yes if larger than scleral trap door | ||
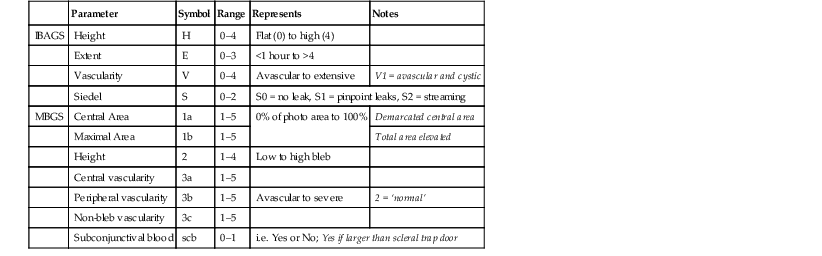
a Indiana Bleb Appearance Grading Scale (IBAGS, 2003).
b Moorfields Bleb Grading System (MBGS).
Many blebs have a central area that is delimited by a rim of subconjunctival fibrosis from a more diffuse peripheral area, referred to as a ‘mixed-morphology’ bleb (see Fig. 78-2-8). In a mixed-morphology cystic and diffuse bleb, a thin cystic central zone is separated from a peripheral diffuse area. A mixed encysted and diffuse bleb contains a thick-walled central area separated from a peripheral diffuse area by the encysted inner bleb wall. The MBGS is capable of describing these variant appearances. Vascularity grading in the MBGS is scored for three zones: central bleb, bleb edge, and non-bleb conjunctiva. If there is no demarcation – in other words, there is no central encysted or cystic area and the bleb is completely diffuse – the central and peripheral bleb vascularity scores are the same. Capture of information about vascularity and bleb size in this way allows for numerical description of a wide spectrum of bleb morphology.
Microcysts are not included in either system, which reflects the original intention of the IBAGS and MBGS to be photographic grading systems for clinical studies, and the difficulty of photographing microcysts. MBGS and IBAGS both omit assessment of corkscrew vessels, which may have prognostic value over and above vascularity grading.
Increasing knowledge of the cellular pathways that regulate wound healing will lead to the development of more sophisticated molecular approaches aimed at inhibiting postoperative scarring. Careful examination of the postoperative bleb provides important information regarding wound healing activity. Postoperative application of antifibrosis therapy should not rely on waiting for the IOP to rise, as scar tissue will already have formed. Rather, antiscarring activity should be titrated against the level of active scarring, which can be determined from the bleb appearance and anterior chamber activity.