Aqueous Misdirection
Pradeep Y Ramulu, Steven J Gedde
Introduction
The combination of flat anterior chamber (AC) and elevated intraocular pressure (IOP) after surgery for angle closure was first described by von Graefe in 1869.1 Since that time, the syndrome defined by these findings has gone by many names, none of which is fully adequate. Since the constellation of IOP elevation with chamber shallowing was refractory to available treatments throughout most of the twentieth century, it earned the name malignant glaucoma. However, this term falsely implies the presence of tumor which can lead to unnecessary concern when used in the presence of patients. The term also fails to reflect the improved treatment outcomes now obtained with modern surgical techniques.
Later in the twentieth century, the term aqueous misdirection was introduced based on the belief that flow of aqueous humor is redirected into the vitreous cavity in eyes demonstrating IOP elevation and AC shallowing without other apparent cause (i.e. pupillary block or choroidal effusion or hemorrhage), though direct evidence for such misdirection of aqueous is lacking. This condition has also been referred to as ciliary block glaucoma, on the suggestion that contact of the ciliary body with the lens or anterior hyaloid face initiates the misdirection of aqueous, or direct lens block angle closure, on the belief this misdirection is a result of movement of the lens into the ciliary sulcus.
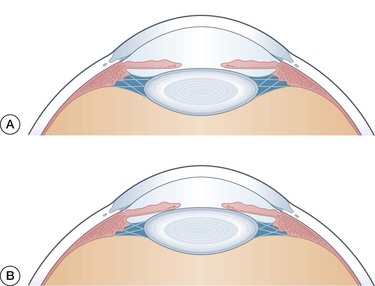
Patients with misdirection have shallowing of both the central and peripheral AC, often referred to as axial shallowing (Fig. 84-1). Vision is usually decreased, but may be nearly normal. In two series of patients with likely aqueous misdirection, approximately 40% of patients had acuities of 20/200 or worse, with equal numbers having acuities of 20/60 or better.2,3 Corneal edema is a frequent finding, and can account for some of the decrease in visual acuity (Fig. 84-2). Of the 14 phakic patients described by Harbour, nine (64%) had clinically evident corneal edema.2
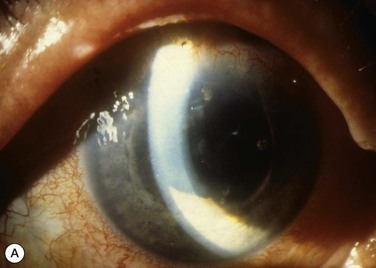

Misdirection can produce substantially elevated IOPs, with case series showing 20–54% of patients having an IOP ≥40.2–4 However, normal or even low-normal IOPs may be seen, with 21–50% of patients having an IOP of ≤22, and several patients described with an IOP of ≤10.2–5
Given the possibility of an IOP anywhere from the high single digits or higher, misdirection should be included in the differential of all postoperative shallow chambers except those with very low IOPs (Table 84-1). However, aqueous misdirection is a diagnosis of exclusion and definitive diagnosis of aqueous misdirection requires: (1) exclusion of pupillary block on the basis of lens status and/or the presence of a patent iridotomy or iridectomy; (2) exclusion of annular choroidal detachment and suprachoroidal hemorrhage with peripheral fundus exam and/or B-scan ultrasonography; (3) careful slit-lamp examination to rule out wound leak or overfiltration; and (4) careful ophthalmic and medication history to rule out causes of ciliary body edema.
Table 84-1
Differential Diagnosis for Aqueous Misdirection
| Clinical Entity | IOP | Distinguishing Features | Excluded by |
| Suprachoroidal hemorrhage | High | Sudden onset Severe pain Found in elderly, HTN, myopia, high pre-op IOP | Fundus exam B-scan US |
| CB edema/rotation | Normal to High | Spontaneous resolution Occurs with VKH, LPIs, PRP, SBP, medications | Ophthalmic Hx Medication Hx UBM |
| Pupillary block | High | Iris bombe Posterior synechiae | Patent iridectomy/iridotomy |
| Choroidal detachment | Low to Normal | Slow resolution May not be visible by fundoscopy in some cases | B-scan US |
| Overfiltration | Low to Low-nl | Formed bleb Absence of leak | SLE |
| Wound leak | Low to Low-nl | Seidel positivity | Seidel testing |
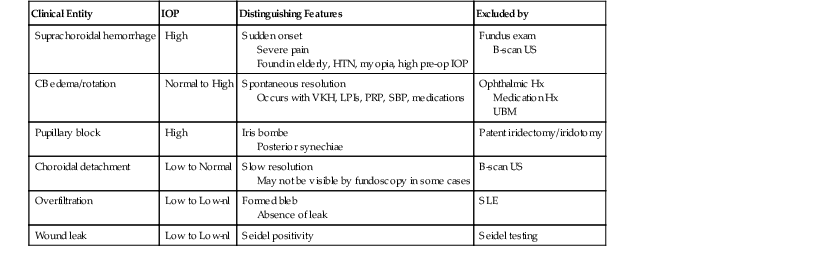
IOP = intraocular pressure, HTN = hypertension, US = ultrasound, CB = ciliary body, VKH = Vokt-Koyanagi-Harada, LPI = laser peripheral iridotomy, PRP = pan-retinal photocoagulation, SBP = scleral buckling procedure, Hx = history, nl = normal, SLE – slit lamp exam, UBM = ultrasound biomicroscopy.
Prevalence and Risk Factors
Procedures Predisposing to Aqueous Misdirection
Aqueous misdirection is usually a surgical complication, and generally follows surgery performed on eyes with angle-closure glaucoma. The incidence of aqueous misdirection is difficult to determine as the complication is rare, occurs variably with the type of operation performed, and can be confused with other clinical entities which produce a similar clinical picture. Several different procedures have been associated with aqueous misdirection, and are discussed separately below.
Trabeculectomy
Trabeculectomy remains the most common glaucoma surgery performed in the United States,6 and series of patients with aqueous misdirection have shown trabeculectomy, alone or in combination with another procedure, to be the inciting event in over 75% of cases (Table 84-2). Trabeculectomy less frequently represents the precipitating event in pseudophakic misdirection, accounting for only 40% of cases in two small series.2,7
Table 84-2
Surgical Events Most Commonly Preceding Aqueous Misdirection
| Author | Year | Inclusion Criteria | # of Cases | Inciting Event/Surgery |
| Harbour2 | 1996 | Shallow AC Patent PI Normal B-scan/indirect exam Refractory to medical rx | 24 | 10 Trab (42%) 6 ECCE (25%) 3 ECCE/Trab (13%) 4 YAG LPI (17%) 1 CRVO (4%) |
| Byrnes22 | 1995 | Shallow AC High IOP Patent PI (19/21 patients) Refractory to medical rx | 21 | 11 Trab (52%) 4 Trab/CE (19%) 4 Trab/Molteno (19%) 2 CE/PKP (10%) |
| Little7 | 1993 | Pseudophakia ‘Diagnosis of malignant glaucoma’ | 7 | 4 ECCE (57%) 2 ECCE/Trab (29%) 1 Trab (14%) |
| Trope27 | 1994 | Shallow AC Elevated IOP Patent PI No visible choroidals | 14 | 12 Trab (86%) 2 ECCE (14%) |
| Tsai3 | 1997 | Shallow AC Elevated IOP Patent PI ‘Posterior misdirection of fluid’ | 19 | 12 Trab (63%) 2 ECCE/Trab (11%) 2 ECCE (11%) 1 ECCE/Iridectomy (5%) 1 Molteno GDI (5%) 1 None (5%) |
| Sharma71 | 2006 | ‘Prolonged’ AC shallowing Elevated IOP (6–32 mm Hg) ‘Absence of papillary block’ | 4 | 2 Trab (50%) 1 YAG LPI (25%) 1 None (25%) |
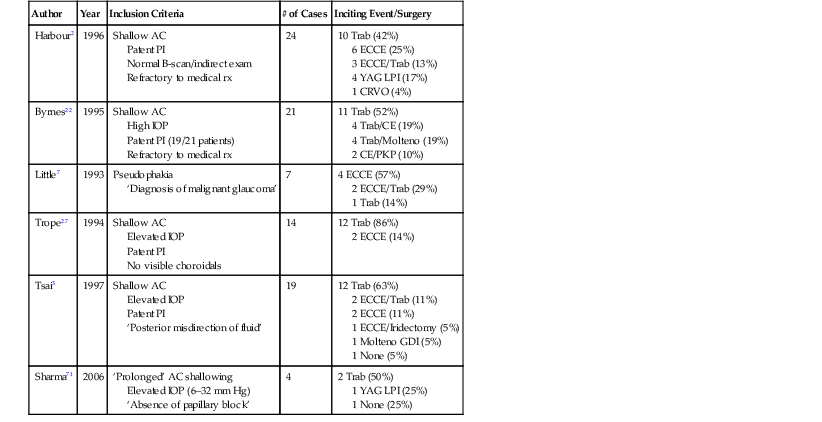
AC = anterior chamber, PI = peripheral iridectomy/iridotomy, Trab = trabeculectomy, ECCE = extracapsular cataract extraction, LPI = laser peripheral iridotomy, CRVO = central retinal vein occlusion, PKP = penetrating keratoplasty, GDI = glaucoma drainage implant.
Few prospective studies of trabeculectomy have evaluated the incidence of aqueous misdirection. Gedde, in the Tube vs. Trabeculectomy (TVT) Study, described one case of aqueous misdirection amongst 105 trabeculectomies (1.0%),8 while Jampel reported two cases of aqueous misdirection occurring in 465 trabeculectomies (0.4%) performed as part of the Collaborative Initial Glaucoma Treatment Study (CIGTS).9 Both of these studies took place in Western populations in which nearly all patients had open-angle glaucoma, and may not apply to other populations in which angle-closure glaucoma is more common.
Aqueous misdirection after trabeculectomy is typically seen in the early postoperative period. Among Harbour's 13 cases of misdirection after trabeculectomy, seven occurred in the first postoperative week (53%) and 10 (77%) occurred within the first month. Adjunctive antimetabolite therapy was administered in only four of the 13 cases, but accounted for two of the three cases presenting greater than one month after surgery, including one case presenting 6 months postoperatively.2 Misdirection may also occur after postoperative manipulation of the operative site, such as suture lysis or bleb needling.10
Glaucoma Drainage Implant Surgery
Three case series have described the rate of aqueous misdirection after placement of a glaucoma drainage implant (GDI). Nguyen and colleagues looked retrospectively at 107 eyes receiving Baerveldt glaucoma implants between 1992 and 1996, reporting aqueous misdirection in four eyes (3.7%).11 The TVT study prospectively evaluated for complications resulting from Baerveldt placement, noting aqueous misdirection in three of 107 (2.8%) cases.8 A similar frequency of aqueous misdirection (4.3%) was also noted in a series of Ahmed glaucoma valves.12 These demonstrate slightly more aqueous misdirection occurring after implantation of non-valved GDIs as compared to trabeculectomy, though these differences are not statistically significant.
The timing of misdirection after GDI placement is variable, and does not cluster around the immediate postoperative period or the opening of the vicryl ligature. Greenfield described a series of 10 cases of aqueous misdirection occurring after placement of a Baerveldt (non-valved) glaucoma implant secured with a vicryl ligature.4 Nearly all eyes were pseudophakic and had had previous trabeculectomy. Six of 10 cases (60%) occurred within 2 months of surgery, with two cases presenting over 4 months after placement of the implant.
Cataract Surgery
While aqueous misdirection can occur after cataract extraction (CE) combined with glaucoma procedures such as trabeculectomy, studies focusing on pseudophakic eyes have noted most cases occurring after cataract extraction alone.2,7 However, cataract extraction appears to be a less-frequent cause of misdirection than trabeculectomy, with case series showing CE (when not combined with glaucoma surgery) to be the inciting factor in 10–38% of cases (Table 84-2). As cataract extraction is performed substantially more than trabeculectomy, but it is observed to cause misdirection less often, the rate of misdirection from cataract removal is likely significantly lower. Little and colleagues found four cases of misdirection from CE alone occurring among 12,000 cataract extractions performed over the study period, estimating an incidence of 0.03%,7 15 times lower than the incidence reported for trabeculectomy in CIGTS.9
While, no data exist on the relative rates of misdirection in extracapsular cataract extraction (ECCE) and phacoemulsification, theoretical reasoning suggests that phacoemulsification may initiate less misdirection. Both the avoidance of lens expression and the use of smaller, self-sealing wounds should permit less chamber shallowing, less vitreous movement, and less opportunity for choroidal expansion, all of which may play an inciting role in the initiation of misdirection.13 Additionally, routine in-the-bag IOL placement with phacoemulsification should eliminate the possibility of sulcus IOL – ciliary body touch, which has been suggested to predispose towards aqueous misdirection, particularly with larger IOLs used in smaller eyes.14–16
Iridotomy and Iridectomy
Surgical iridectomy was the first operation associated with malignant glaucoma, with Von Graefe reporting a 2% incidence of the complication in his original study.1 Subsequent reports from the early 20th century confirmed a 2–4% incidence of malignant glaucoma after surgical iridectomy, with the incidence of aqueous misdirection likely being somewhat lower.17
The advent of the Argon and YAG lasers has nearly eliminated the use of surgical iridectomy,6 but has not eliminated aqueous misdirection after the treatment of pupillary block. Reports of malignant glaucoma following laser peripheral iridotomy (LPI) include cases which may have occurred by mechanisms other than aqueous misdirection, i.e. ciliary body swelling occurring from usage of sulfa medications or central retinal vein occlusion.18,19 However, a likely case of aqueous misdirection resulting from LPI was described by Robinson,20 and case series with good criteria for diagnosing aqueous misdirection have attributed LPI as the causative event in 0–29% of cases (Table 84-2).
Corneal Transplantation
Penetrating keratoplasty (PK) is a very rare setting for aqueous misdirection. Jacoby described a single case of likely misdirection after combined CE and PK,21 and Byrnes' series included two patients (10%) with misdirection after combined PK and CE (Table 84-2).22 As all cases occurred after PK combined with CE, it remains unclear whether corneal transplantation alone presents a risk of aqueous misdirection.
Phakic IOL
A single case of misdirection has been reported after placement of a phakic IOL in a 23-year-old high myope.23 Pupillary block was ruled out with the placement of two preoperative LPIs, and the eye normalized after explant of the IOL.
Vitrectomy
Massicotte reported two cases of aqueous misdirection after pars plana vitrectomy and suggested that both resulted from fibrin forming on the ciliary body, resulting in ciliary body – anterior hyaloid touch and posterior redirection of aqueous.24 Indeed, one case resolved after tPA injection, though vitreous gas was also removed at the same time, possibly removing a gas-mediated fluid block as suggested by Francis.25
Ocular Features Predisposing to Aqueous Misdirection
Angle Closure
Malignant glaucoma, since its first description by Von Graefe, has been noted to occur primarily in eyes with angle closure.1 Chandler argued that shallow anterior chamber depth was a particular risk factor for subsequent malignant glaucoma, and noted very shallow anterior chambers in all six of his patients.17
Regardless of the inciting procedure, most cases of aqueous misdirection occur in eyes with angle-closure glaucoma (ACG). Case series consisting mostly of patients with misdirection after trabeculectomy reveal angle-closure glaucoma in 50–79% of patients (Table 84-3). Additionally, in Harbour's six cases of misdirection occurring from CE alone, two occurred in CACG while one occurred in a patient with a previous LPI.2 Likewise, in Greenfield's 10 patients with misdirection after placement of a Baerveldt implant, four had CACG while one had nanophthalmos.4
Table 84-3
Types of Glaucoma Seen in Series of Patients with Misdirection
| Author | Year | # Cases (Total) | CACG Cases (%) | AAC Cases (%) | POAG Cases (%) | PXFG Cases (%) | No Glauc (%) |
| Harbour2 | 1996 | 24 | 13 (54) | 3 (13) | 2 (8) | 0 | 6 (25) |
| Byrnes22 | 1995 | 21 | 14 (67) | 4 (19) | 0 | 0 | 3 (14) |
| Tsai3,# | 1997 | 19 | 15 (79) | ND | 2 (11) | 2 (11) | 0 |
| Trope27,* | 1994 | 14 | 10 (71) | 0 | 2 (14) | 4 (29) | 0 |
| Sharma71 | 2006 | 5 | 2 (40) | 2 (40) | 1 (20) | 0 | 0 |
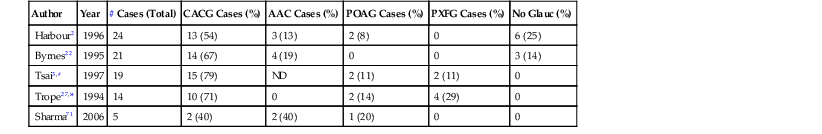
# No distinction was made between AAC and CACG.
* Two patients had both PXFG and CACG.
While one would expect surgery in eyes with angle-closure glaucoma to have a higher rate of aqueous misdirection, most studies of surgery in angle-closure glaucoma have not specifically looked for aqueous misdirection as a complication, often incorporating it under the umbrella of postoperative shallow chambers. Additionally, most such studies are limited by a retrospective design and small sample size. A report by Lowe in 1978 looked at the rate of malignant glaucoma in 800 glaucoma surgeries performed in patients with narrow angles.26 Six cases (0.8%) of malignant glaucoma resulted, two from surgical iridectomy, one from sclerectomy, and three from iridencleisis. This represents a rate five times higher than that reported in CIGTS,9 though the two studies evaluated different procedures, and the true rate of aqueous misdirection may have been lower in Lowe's study as some cases may have represented shallow chambers from mechanisms other than aqueous misdirection.
Acute-Angle Closure
Von Graefe noted all of his malignant glaucoma cases in patients with chronic angle closure, concluding that acute-angle closure (AAC) did not result in aqueous misdirection.1 However, among the 14 phakic eyes with misdirection described by Harbour, seven resulted from procedures performed in the context of CACG and three occurred from procedures performed for AAC, demonstrating that misdirection can indeed occur in the context of acute-angle closure.2 Given the rarity of this complication, the relative rates of misdirection in CACG and AAC remain unknown.
Pseudoexfoliation
While the majority of patients in all of the series described above had misdirection occurring in the context of angle-closure glaucoma, four of Trope's 14 patients (29%) and two of Tsai's 19 patients (11%) had pseudoexfoliation glaucoma.3,27 While these do not clearly represent a grossly abnormal fraction of patients with exfoliative glaucoma, there are theoretical reasons to believe that pseudoexfoliation may predispose to aqueous misdirection. Rieser described a case of a patient with pseudoexfoliation and phacodonesis who experienced postoperative aqueous misdirection resolving with a Chandler procedure.28 The authors suggested that loose zonules might have facilitated anterior movement of the lens, allowing for concomitant vitreous movement and posterior redirection of aqueous humor flow.
Aphakia
While aphakia is a rare cause of aqueous misdirection, such eyes may be predisposed to this complication as vitreous can easily come forward along the ciliary body resulting in posterior redirection of aqueous.29,30 Numerous authors have described cases of misdirection in eyes left aphakic after intracapsular cataract extraction (ICCE), including Sugar, who described a case of probable bilateral misdirection,30 and Epstein, who described three eyes with aphakic aqueous misdirection amongst his series of five patients treated with YAG anterior hyaloidotomy.31 The anterior hyaloid face can also cause pupillary block in aphakic eyes, so as with all cases of misdirection, a patent iridotomy or iridectomy is required to make the diagnosis.
Preoperative IOP
Chandler suggested that malignant glaucoma was more common in eyes with elevated preoperative pressures. In his case series of six patients, all had elevated preoperative pressures ranging from 32–70 mmHg.17 However, malignant glaucoma from these earlier reports may have resulted from undetected choroidal detachments, which would be expected to occur frequently in eyes with a large, sudden drop in IOP. Also, given the rarity of this complication and the fact that many eyes with ACG have severely elevated IOPs at the time of surgery, the importance of this risk factor remains unknown.
Miotic Use
Miotic therapy has been noted to precipitate aqueous misdirection in previously operated eyes,32,33 as well as to cause presumed misdirection in eyes with no previous surgery.28,34 Levene described several cases of likely bilateral misdirection, with misdirection in the first eye initiated by surgery, and misdirection in the second eye initiated by miotic therapy.32
Vitreous Inflammation
Lass reported a case of aqueous misdirection occurring in a patient with nocardia endophthalmitis following a penetrating corneal injury.35 B-scan showed extensive inflammatory debris over the ciliary body, and a mechanism for aqueous misdirection much like misdirection after vitrectomy was postulated, with a fibrin/enlarged ciliary body complex touching the anterior hyaloid and leading to posterior misdirection of aqueous.
ROP
Kushner reported three cases of chamber shallowing and elevated IOP occurring in patients aged 2–25 with a history of retinopathy of prematurity.36 All cases resolved with cycloplegia, and the authors suggested that these findings resulted from aqueous misdirection resulting from ciliolenticular block in these eyes with a small pars plana. However, the absence of iridotomy and ultrasonography in these cases makes it difficult to rule out other reasons for chamber shallowing and elevated IOP.
Spontaneous Misdirection
Cases of possible spontaneous misdirection in the absence of surgery or miotic use have been reported. Fanous described a 45-year-old hyperopic female presenting with unilateral high pressure and shallow AC,37 with the authors speculating a ciliary block mechanism primarily based on the response to cycloplegia. A more compelling case was presented by Schwartz and Anderson, who described an 85-year-old male who presented with unilateral pressure elevation, shallow chamber, and myopic shift.38 At surgery, posterior pressure was observed to be pushing vitreous through the surgically created iridotomy, and fluid less viscous than vitreous was drained behind an unusually solid anterior vitreous face, strongly suggesting aqueous misdirection as the mechanism.
Prevention
Misdirection in one eye quite clearly confers an increased risk of misdirection in the contralateral eye. In Chandler's series of six patients with likely misdirection, three of four who underwent a similar operation in the second eye experienced misdirection again.17 Several other case series and individual case reports have demonstrated likely bilateral aqueous misdirection,1–3,30 and the contralateral eye should receive early prophylaxis against angle closure and should be assumed to be at high risk of misdirection should surgery be required.
Etiology/Pathophysiology of Aqueous Misdirection
A full understanding of aqueous misdirection is lacking, resulting in much speculation regarding its pathophysiology. An adequate description of the mechanisms involved needs to explain all aspects of the complication, including predisposing factors, initiation, perpetuation of symptoms, and resolution with appropriate treatment.
Predisposition.
A striking feature of aqueous misdirection is its strong predilection for eyes with preoperative shallow chambers and angle closure. Chandler also felt that these eyes had an IOP which could not be normalized preoperatively,17 leading others to suggest that misdirection may have already taken place in some of these eyes.39,40 Indeed, convincing cases of spontaneous misdirection in the absence of surgery, laser or miotic use have been presented, suggesting that ‘inciting’ events may actually only be exacerbating events.
Anterior lens position may represent the likely link between angle closure and subsequent misdirection (Fig. 84-3). Such a lens position not only allows for iridolenticular contact and subsequent angle closure, but would also facilitate vitreous movement up against the ciliary body and subsequent redirection of fluid posteriorly.17 Anterior lens movement may also be facilitated by miotics or pseudoexfoliation,32 both likely risk factors for misdirection.
Quigley suggested choroidal expansion as an alternate explanation tying angle closure to malignant glaucoma.13 In this model, acute choroidal expansion occurring during or after surgery would lead to a posterior–anterior pressure gradient leading to vitreous compression. Hyperopic eyes with angle closure would be particularly prone to angle closure due to a thicker scleral wall preventing trans-scleral egress of fluid from the choroidal extravascular space.
Initiation.
Actual forward movement of the lens may occur postoperatively for several reasons. Intraoperative chamber shallowing may place the vitreous or lens directly in contact with adjacent structures, i.e. the ciliary body (Fig. 84-4). However, not all cases of misdirection occur immediately after surgery with vitreous detachment (as suggested by Shaffer)29 or eye rubbing possibly initiating later ciliovitreal touch. Alternately, the ciliary body may come to the lens or vitreous as a result of enlargement, i.e. after miotic use32,34 or fibrin deposition with anterior vitreous inflammation.24
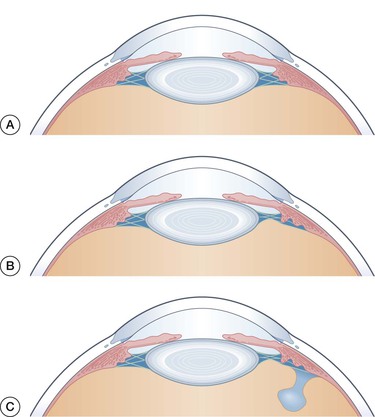
Ciliovitreal or ciliolenticular contact alone may not be sufficient to initiate misdirection. Grant and Epstein demonstrated that the normal vitreous provides very little resistance to outflow.41,42 Herniation of vitreous against the ciliary body may limit the anterior hyaloidal surface area through which posteriorly directed fluid must flow, thereby increasing vitreous resistance to flow.42 Alternately, a long-term moderate increase in flow through the vitreous may mimic the increased outflow resistance noted to occur by Grant and Epstein when the vitreous perfusion pressure was elevated.41,42 Vitreous resistance to flow may also increase as a result of acute choroidal expansion creating a greater posterior to anterior fluid flux within the vitreous.13
Perpetuation.
Once initiated, aqueous misdirection will rarely resolve without intervention, suggesting that the mechanism is somehow perpetuated (Fig. 84-5). Perpetuation of misdirection may occur because of changes that continue the posterior flow of aqueous and/or changes that limit the flow of posteriorly directed fluid through the vitreous. Shaffer argued that the vitreous may form a ball valve leading to continued posterior flow of aqueous.40 Others have argued that posterior flow is perpetuated by inflammatory welding of the ciliary body to the vitreous, with Shaffer noting ciliary body tissue attached to anterior hyaloid excised during surgery on a patient with possible misdirection.40
Changes in the vitreous may also provide increased resistance to the forward movement of posteriorly directed fluid. Epstein and others have shown that flow through the vitreous, particularly at elevated IOPs, severely impairs outflow resistance,41–43 possibly due to compaction of vitreous as suggested by Quigley.44 Indeed, many authors have found a very dense anterior hyaloid at the time of surgery.38,40 Robbins has shown that overall vitreous volume increases with elevated IOP, which may contribute to further angle closure and further increase IOP.45 Ultrasound biomicroscopy (UBM) studies have shown anteriorly rotated ciliary bodies in eyes with misdirection,27,46–48 suggesting an increase in posterior segment volume from diffuse expansion of large portions of vitreous, accumulations of aqueous-like material within the vitreous, or increases in choroidal volume.
Treatment.
We have learned much about the pathogenesis of aqueous misdirection from the treatments that have been effective in resolving the condition (Fig. 84-6). The importance of the ciliary body has been demonstrated by the effectiveness of cycloplegia, which thins the ciliary body and increases the zonular diameter,49 and laser shrinkage of the ciliary body processes either through direct laser application through an iridectomy50,51 or via trans-scleral cyclophotocoagulation.52 Such treatments may break ciliovitreal adhesions or apposition preventing continued posterior flow of fluid. The importance of the anterior hyaloid has been demonstrated by Epstein, who was able to resolve some cases of misdirection with Nd : YAG treatment to this structure,31 and Chandler who noted that ICCE with violation of the anterior vitreous face was more likely to result in resolution of misdirection.17 More posterior vitreous is doubtlessly important as well given the effectiveness of Chandler's procedure, and vitrectomy, which can work occasionally even when the anterior hyaloid face is preserved. Lens removal does not routinely normalize IOP, suggesting that ciliolenticular contact is not central to the mechanism.
Management Options
Medical Management
Medical therapy should be instituted once a diagnosis of aqueous misdirection is made. Table 84-4 shows a regimen that is commonly used to treat this condition. Mydriatic-cycloplegic drops tighten the zonules and pull the lens–iris diaphragm posteriorly in phakic eyes.49 In aphakic eyes, mydriatic-cycloplegic therapy is expected to be less beneficial, but may move the ciliary body ring outward away from the hyaloid53 and increase the surface area for fluid transfer into the anterior chamber through a dilated pupil.54 Beta blockers, alpha agonists, and carbonic anhydrase inhibitors suppress aqueous production with the effect of lowering the intraocular pressure (IOP) and decreasing the amount of fluid that is misdirected into the vitreous cavity. Osmotic agents dehydrate the vitreous and reduce vitreous volume and pressure.55 Use of an anti-inflammatory agent can decrease the likelihood of peripheral anterior synechiae formation while the anterior chamber is shallow, and may prevent permanent adhesions of the hyaloid to the ciliary body. Any of the above medications should be withheld if there is a contraindication to their use, and they may be discontinued if side effects develop.
Table 84-4
Standard Medical Regimen for Aqueous Misdirection
| Mydriatic-cycloplegic | Atropine 1% qid |
| Phenylephrine 2.5–10% qid | |
| Aqueous suppressants | Timolol 0.5% bid |
| Brimonidine tid | |
| Acetazolamide 250 mg po qid | |
| Osmotics | Isosorbide 45% 1.5 mL/kg po qd, or |
| Mannitol 20% 2 g/kg IV over 45 min qd | |
| Anti-inflammatory | Prednisolone acetate 1% qid |

This medical regimen should be continued until the anterior chamber forms and the IOP normalizes. It has been reported that 50% of cases of aqueous misdirection are relieved within 5 days of using this approach.56,57 If the condition resolves, medications are gradually withdrawn over several days. However, there is a significant risk that aqueous misdirection may recur following discontinuation of a cycloplegic. or even in the context of continued cycloplegic use. Indeed, Debrouwere and colleagues noted that 100% of their medically treated patients (n = 10) relapsed, with time to relapse extending all the way out to 2 years.58 In cases in which laser therapy or surgical management have not been used or have proved ineffective, continued treatment with atropine 1% daily is recommended.
Surgical Management
Surgical management is directed toward correcting the block to aqueous flow and reestablishing a normal anterior circulation of aqueous. Figure 84-7 outlines an algorithm that may be used in the surgical management of aqueous misdirection.
Laser Therapy
Because laser therapy has relatively low risk, we recommend that it be performed early in the therapeutic course. Laser therapy requires a clear visualization of the structures to be treated. Topical application of glycerin can be used to clear corneal edema and allow laser treatment in some cases.
Direct Argon Laser Photocoagulation of Ciliary Processes.
Argon laser treatment of ciliary processes has been reported to relieve aqueous misdirection.50,51 Laser burns are applied to the ciliary processes that are visible through a peripheral iridectomy, with or without the aid of a gonioscopic lens. Typical laser settings include power of 100 to 300 milliwatts, duration of 0.1 to 0.2 second, and spot size of 50 to 100 microns. Laser shrinkage of the ciliary processes presumably works by relieving ciliohyaloidal block of the anterior flow of aqueous humor.50 Long-term outcomes with such treatment, however, are unknown.
Nd : YAG Laser Hyaloidotomy.
There have been several reports of the successful treatment of aqueous misdirection with Nd : YAG laser hyaloidotomy in aphakic and pseudophakic eyes.6,28,59–62 Disruption of the anterior hyaloid establishes a communication between the anterior and posterior segments. The procedure may be performed without a contact lens, or using an iridotomy or gonioscopic lens. Laser energy is typically set at 4 to 6 millijoules. A central hyaloidotomy may not be adequate to produce a communication with the anterior chamber because of the intervening lens capsule and intraocular lens. The hyaloidotomy should be performed peripheral to the lens optic, and may be done through a peripheral iridectomy. As with medical treatment, hyaloidotomy may only serve as a temporizing measure before more definitive treatment, as relapse was noted in three of four eyes undergoing hyaloidotomy with a time to recurrence ranging from 1 to 5 months.58
Cyclodestructive Procedures.
Cyclocryotherapy63 and cyclophotocoagulation52,64,65 have been reported as treatments for aqueous misdirection. Success can occasionally be long-term, with one series of five cases showing remission in only one patient (resolving with repeat cyclophotocoagulation) over a follow-up period ranging from 1 to 8 years.52 The presumed mechanism involves shrinkage of the ciliary body, thus eliminating ciliohyaloidal touch, decreasing posterior aqueous flow, or decreasing the potential for the ciliary body to expand.
Incisional Surgery
Incisional surgery is indicated when medical management and laser therapy are unsuccessful in resolving aqueous misdirection. Several surgical approaches have been described for treating aqueous misdirection, and clear superiority of one procedure has not been demonstrated.
Slit-Lamp Needle Revision.
Transcorneal needling to disrupt the anterior hyaloid face and reform the anterior chamber may be performed if a Nd : YAG laser is not accessible.66
Posterior Sclerotomy and Air Injection (Chandler Procedure).
Aspiration of fluid from the vitreous using an 18-gauge needle inserted through the pars plana and reformation of the anterior chamber with an air bubble was originally advocated by some surgeons as the surgical procedure of choice for aqueous misdirection.40,56,57 Patients are generally maintained on atropine postoperatively to prevent recurrence.
Pars Plana Vitrectomy.
With the advent of modern vitreoretinal surgical techniques, pars plana vitrectomy has replaced many other procedures for managing aqueous misdirection. Removal of the anterior hyaloid is the most important portion of the procedure, as core vitrectomy alone can lead to frequent relapse.58 In phakic eyes, difficulties excising the anterior hyaloid without damaging the crystalline lens contribute to a higher rate of surgical failure compared with pseudophakic and aphakic eyes.2,3,22 Vitrectomy may be combined with pars plana lensectomy in phakic eyes with marked corneal edema, dense cataract, or inability to deepen the anterior chamber during vitrectomy.2 Pars plana insertion of an aqueous shunt in conjunction with pars plana vitrectomy has been reported in patients with synechial angle-closure glaucoma.67 Tube insertion through the pars plana can reduce the risk of recurrent aqueous misdirection by preventing an increase in vitreous volume and pressure, and provides long-term IOP control. As fluid is being directly drained from the posterior segment, hyaloidectomy and iridectomy may not be necessary.
Lens Extraction.
Lens extraction has been shown to effectively treat aqueous misdirection.3,56,57,68,69 However, this procedure should be combined with a posterior capsulotomy and anterior vitrectomy using an anterior or posterior approach.3,69,70 Success rates of this procedure are variable, with several groups reporting consistent success, and one study demonstrating eventual relapse in three of four cases.58 Alternately, vitreous removal with a core vitrectomy before lens extraction has been described.71
Zonulo-Hyaloido-Vitrectomy.
Zonulo-hyaloido-vitrectomy performed with an anterior vitrector has been shown to be effective in managing pseudophakic aqueous misdirection in studies with mean follow-up times under a year.72,73 In this procedure, a vitreous cutter is introduced into the anterior chamber through a paracentesis incision and passed through a peripheral iridectomy to remove zonules and anterior hyaloids or through the pars plana and brought forwards through the anterior hyaloids and iris (Fig. 84-8).
Prognosis
While the term ‘malignant glaucoma’ was initially used to describe this condition because of its poor response to conventional therapy, more favorable outcomes have been achieved in recent years when the surgeon is diligent in making the correct diagnosis and instituting appropriate treatment.
With the staged treatment algorithm described in Figure 84-7, misdirection can be eliminated in nearly all cases. For instance, in Greenfield's series of 10 patients with misdirection after GDI placement, a final IOP of 18 or less was achieved after a mean follow-up of 9 months in 9 of 10 patients (90%).4 Six of 10 eyes (60%) required additional medications to achieve this IOP. Control was accomplished with cycloplegia in one eye, with Nd : YAG hyaloidotomy in four eyes, and with vitrectomy (with or without lensectomy or lens explantation) in five eyes.4 Patients with a successful initial treatment to medicines or laser are at lifetime risk of recurrence, with relapse into misdirection sometimes occurring months or even years later.58
Good IOP results were also seen in Harbour's 24 patients undergoing PPV for aqueous misdirection.2 Twenty-two of 24 (92%) achieved an IOP of 18 or less, with only six of 24 patients (25%) remaining on IOP-lowering medicines. Only one patient retained a shallow anterior chamber.
Despite generally good IOP control with therapy, patients with misdirection are at significant risk of losing acuity. In Harbour's series of patients undergoing PPV, several serious postoperative complications were described, including corneal decompensation in 21% of eyes, trabeculectomy failure in 23% of eyes, and choroidal effusion in 13% of eyes.2 Other reports have detailed other serious complications, including suprachoroidal hemorrhage and vitreous hemorrhage.3
Case series describe a final acuity of 20/200 or less in 21–50% of eyes.2–4 Though visual potential is often limited preoperatively, many eyes experience a significant drop in acuity. These outcomes generally reflect patients treated in the 1980s and early 1990s. With improved vitrectomy techniques and increasing awareness of the need for removing the anterior hyaloid with surgery, current outcomes may be better than described above.
Precautions must be taken to protect the fellow eye, since patients with aqueous misdirection in one eye are at high risk to develop it in the other.28,30,74,75 A peripheral iridectomy should be performed if the angle is occludable, because the presence of angle-closure glaucoma is strongly associated with aqueous misdirection. Miotic therapy should be avoided, as these agents have been reported to precipitate the condition. If the fellow eye requires surgery, cycloplegics and osmotic agents should be used preemptively, and the surgeon must monitor the eye closely for the postoperative development of aqueous misdirection. Prophylactic vitrectomy with zonulo-hyaloido-iridectomy or vitrectomy with posterior tube shunt placement may be considered.




