Goniosynechialysis
Chaiwat Teekhasaenee
Introduction
Primary angle closure (PAC) is an anatomical disorder of the eye characterized by iris apposition to the angle wall. Although laser iridotomy is considered to be the definitive treatment, up to 58% of patients with PAC continue to have elevated intraocular pressure (IOP) requiring postoperative glaucoma medication.1–5 The incidence of residual angle closure is increased in patients who have had prolonged angle closure prior to the iridotomy.6,7 Postoperative IOP control depends on the amount of trabecular meshwork damage and extent of peripheral anterior synechiae (PAS). Progressive irreversible endothelial damage and subsequent occlusion of the Schlemm's canal and trabecular meshwork will eventually occur if the PAS remain untreated.8
There is a direct but nonlinear relationship between IOP and degrees of synechial closure. The IOP is usually elevated when >180° of the angle is closed by the PAS. When >270° of the angle is closed, medical therapy is usually ineffective and filtering surgery becomes necessary.9 Nevertheless, filtering surgery has several potentially serious complications which are more prevalent in eyes with PAC.10 Flat anterior chamber and malignant glaucoma tend to occur more frequently. The routine use of anti-metabolites increases postoperative endophthalmitis and bleb leak. In addition, filtration is subjected to closure by the healing process and the success rate decreases over time. A more logical approach is to eliminate the PAS and restore the trabecular function prior to the ultrastructural changes. Filtration through the natural trabecular pathway should be more physiologic and reliable than the artificial channel of filtering surgery.
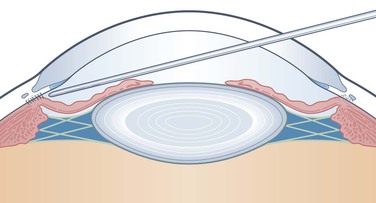
Argon laser peripheral iridoplasty can open an appositionally closed angle, but does not eliminate PAS. Goniosynechialysis (GSL) is a surgical procedure designed to strip the synechiae from the angle wall and restore the trabecular outflow. Shaffer first described the procedure by using a cyclodialysis spatula and intraoperative gonioscopy.11 However, anterior chamber collapse and uncontrollable hemorrhage made the procedure unpopular. Chandler and Simmons proposed anterior chamber deepening as a diagnostic and therapeutic procedure for the unresolved acute PAC.12 Nevertheless, the procedure does not eliminate extensive or long-standing PAS. Most PAS are firmly established and resistant to such gentle efforts. Campbell and Vela described a modified technique employing a viscoelastic to maintain the anterior chamber and control hemorrhaging, while using an irrigating spatula to separate the PAS (Fig. 108-1).9 The procedure was successful in 80% of eyes with minimal complications if the PAS had been present for less than one year. Later studies have confirmed the effectiveness of GSL in reducing PAS and improving IOP control.13–17 A study has shown an increase in the tonographic outflow facility following removal of the synechiae.14 The procedure is effective in separating the synechiae not only in PAC but also in secondary angle closure following vitreoretinal or filtering surgery.15,18
The success of GSL depends not only on the preoperative duration but also the recurrence of the PAS. Although pupillary block has been eliminated by iridectomy, progressive angle closure can continue if non-pupillary block mechanisms are also present. It has been shown that nearly 60% of patients with a successful surgical iridectomy19 or 38% with a patent laser iridotomy still have a positive dark room prone provocative test.5,20 Recurrent attacks and further angle closure have been reported in both the attacked and the fellow eyes.3,6,21,22 Factors contributing to the reformation of PAS are early postoperative inflammation, plateau iris configuration, and the presence of an enlarged or anteriorly situated lens. It has been shown that cataract formation progresses rapidly following an acute attack.1 A thickening cataractous lens would crowd the anterior chamber and force the iris back against the trabecular meshwork.
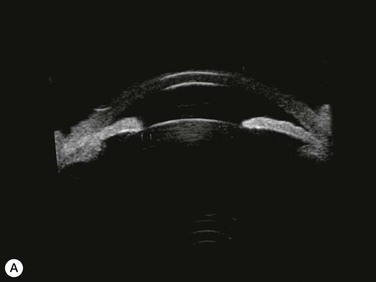
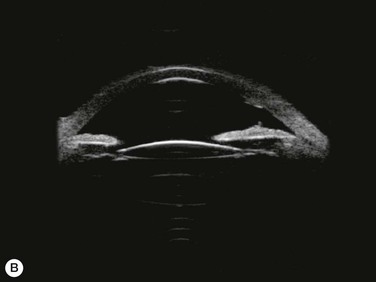
Substantial increases in anterior chamber depth and angle width following cataract extraction with intraocular lens (IOL) implantation have been demonstrated in eyes with PAC (Fig. 108-2A,B).23–25 The narrower the preoperative anterior chamber angle, the greater the alteration of angle configuration. Whereas iridolenticular contact was observed in a phakic eye, there was no iris and IOL contact in a pseudophakic one (Fig. 108-2A,B). The iris plane shifted backward, deepening the central anterior chamber by approximately 850 µm. Preoperative biometry performed in a series of 52 eyes of 48 consecutive Thai middle-aged patients who had developed acute PAC within 6 months showed that average natural lens thickness and central anterior chamber depth were 4.83 mm and 1.8 mm, respectively.26 Since the IOL (PMMA, silicone, acrylic) thickness in the 20–25 D power ranges from 0.75 mm to 1.42 mm. Replacement of the natural lens with the IOL provided up to 4 mm more axial distance within the anterior segment eliminating angle crowding and appositional closure. In addition, anterior chamber deepening with a viscoelastic during IOL implantation might break recent PAS.
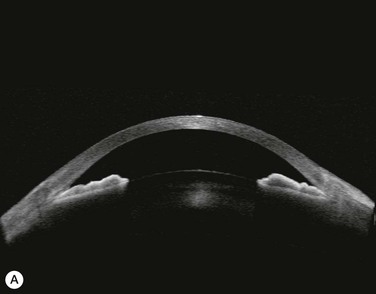
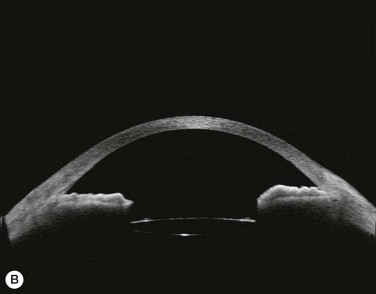
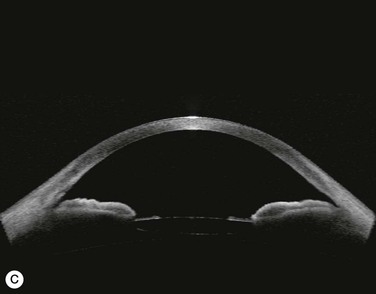
Several studies have shown that extracapsular cataract extraction (ECCE) with IOL implantation is effective in opening the angle and controlling IOP in refractory PAC.27–30 Currently, ECCE has been replaced by phacoemulsification which offers a higher surgical success rate, less postoperative inflammation and fewer complications. In addition, the clear corneal approach spares the superior conjunctiva for possible filtering surgery, if needed. Phacoemulsification and IOL implantation has been reported to be highly effective in patients with uncontrolled PAC.31,32 However, up to 32% of the patients still had persistent PAS and required long-term treatment with glaucoma medication following lens removal either by ECCE or phacoemulsification and IOL implantation alone (Fig. 108-2A).5,32 Goniosynechialysis is the next step to eliminate the remaining synechiae and control the IOP (Fig. 108-2C).
Goniosynechialysis becomes more effective when performed after lens removal. An increase in the anterior chamber space provides ample room to perform the procedure and reduces the chance of synechial reformation. Combined phacoemulsification and GSL (phaco-GSL) has been shown to be safe and highly effective in controlling IOP (<21 mmHg) and decreasing PAS in >90% of 52 eyes that developed acute PAC within 6 months and had persistent IOP elevation following laser treatment.26,33,34 Postoperative IOP was reduced to below mid-teens regardless of the preoperative IOP level. Recurrence of the PAS, although uncommon, might occur during the first three months. The success of phaco-GSL has been stable since the third postoperative month for up to 14 years, providing a long-lasting control or even a permanent cure.26 A recent multicenter study also demonstrates a success rate (< 21 mmHg) of 85.9% in 109 eyes with uncontrolled PAC/PACG at one and three years following phaco-GSL.35
Indications
Goniosynechialysis is best for patients who have had previously normal trabecular meshwork that has recently developed PAS on the basis of acute or chronic PAC, surgical procedures such as cataract extraction and penetrating keratoplasty, or trauma. Patients with acute PAC should have uncontrolled IOP and persistent PAS following successful laser iridotomy and laser peripheral iridoplasty. The extent of PAS should be >180° and correlates with the IOP. If not, an open-angle mechanism may coexist and makes the prognosis unfavorable. The shorter the duration of synechial closure, the better the prognosis. However, a postoperative fibrinous anterior chamber reaction commonly occurs if the operation is performed during the first four weeks after the acute attack. The optimal timing for phaco-GSL is suggested at week six after the attack when the eye becomes quiet.26
Contraindications
Goniosynechialysis is contraindicated in patients who are likely to have ultrastructural changes of the trabecular meshwork from long-standing PAS. The presence of severe glaucomatous optic nerve cupping or extensive visual field loss should suggest long-standing asymptomatic closure despite a history of a recent acute attack. Patients with congenital angle anomalies and secondary angle closure following uveitis or anterior membranous pulling from neovascularization, iridocorneal endothelial syndrome, or posterior polymorphous dystrophy, are poor candidates.
Preoperative Evaluation and Treatment
The extent of PAS must be evaluated and correlated with the level of IOP. The duration of the PAS must be determined. A-scan ultrasonic biometry is not only important for IOL power calculation but also for accurate diagnosis. Eyes with an extremely narrow anterior chamber depth < 1.8 mm or asymmetrical anterior chamber depth > 0.2 mm may have subclinical lens dislocation. A capsular tension ring, an IOL with suture loops for scleral fixation, or anterior vitreous tapping may be necessary.
The patients should be treated with hypotensive medications to maximally reduce the IOP. Systemic mannitol may be necessary in patients with very high pressure. In addition, frequent steroids are needed to prevent postoperative reactions. Pilocarpine is discontinued in eyes undergoing phaco-GSL.
Surgical Technique
A clear view of the angle structures by gonioscopy is essential in performing GSL. A blind procedure can injure not only the trabecular meshwork but also the nearby structures resulting in further outflow damage and hemorrhage.
Goniosynechialysis can be performed with a surgical loupe with headlight illumination. This technique provides an unrestricted range of surgical access. However, an operating microscope provides a much clearer surgical view. An operating microscope equipped with an effortless tilting mechanism and straight eyepieces allows an efficient surgical approach (Fig. 108-3).
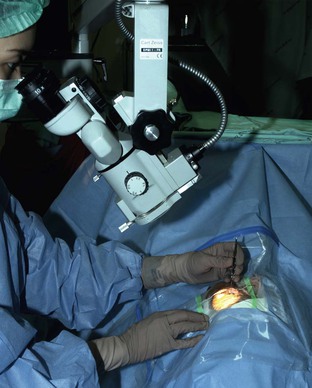
Goniosynechialysis can be performed under topical, intraocular, peribulbar, or retrobulbar anesthesia. Patients should be draped so that there is no restriction in turning their position. The patient's head along with the body will be rotated >45° to the left or right while the microscope is being tilted until a proper surgical access is accomplished (Fig. 108-3). If the rotation is limited, the microscope must be tilted to almost a horizontal axis, making the surgical procedure cumbersome. Performing GSL of the temporal angle in patients with a high nasal bridge or deep-set eye can be quite demanding. Ocular movement toward the treated angle can facilitate the surgical attempt.
Procedure
Three beveled corneal paracenteses are made with a 15° sharp blade evenly spaced 120° apart at 3:00, 7:00, and 11:00 o'clock positions in the right eye and at 2:00, 5:00, and 9:00 in the left. In the presence of a corneal pannus, the paracenteses should be performed more anteriorly to avoid hemorrhage that can form a thin layer between the cornea and the goniolens obscuring a gonioscopic view. The paracentesis track should point toward the opposite angle and be just wide enough for a snug fit of a Swan knife. Too large paracentesis results in a leakage of the viscoelastic and a flat anterior chamber.
Anterior chamber deepening procedure is performed by depressing the posterior lip of the paracenteses, allowing egress of the aqueous from the anterior chamber. A muscle hook is used to indent the limbus forcing aqueous flow from the posterior chamber through the pupil into the anterior chamber. The aqueous is released from the anterior chamber again. The procedure is repeated until nearly all the aqueous is released. A viscoelastic is then injected into the anterior chamber to sufficiently deepen the anterior chamber and raise IOP. Excessive viscoelastic can result in extreme IOP elevation and corneal edema. The peripheral iris will be stretched and bowed posteriorly toward the empty posterior chamber (Fig. 108-1). In eyes with extremely high IOP and shallow anterior chamber, anterior vitreous tapping may be necessary.
When combined with phacoemulsification, GSL should be performed after lens removal and IOL implantation. The phacoemulsification can be performed either through a temporal clear corneal or scleral incision. If performed through the scleral incision, complete hemostasis is necessary to avoid a blood film on the cornea that can interfere with the gonioscopic view. Performing phacoemulsification in an eye with a shallow anterior chamber requires a competent skill. Inadvertent corneal damage or lens dislocation can occur.
Intraoperative gonioscopy is performed with a surgical direct goniolens. Deepening of the AC and raising the IOP with a cohesive viscoelastic not only allows a clear gonioscopic view by preventing the formation of corneal folds but also stops hemorrhage, if accidentally occurs. Goniosynechialysis will proceed only if a clear view of the angle structures is achieved and the presence of PAS is confirmed. Intracameral injection of acetylcholine chloride, 1 : 100 (Miochol-E, Novartis Ophthalmics, Inc., Duluth, GA) may be given to constrict the pupil unless the sphincter is paralyzed. A blunt-tipped Swan knife is introduced into the anterior chamber through the paracentesis and advanced toward the opposite angle under direct visualization. The Swan knife, while snugly engaged in the paracentesis track, can be used to either immobilize or shift and tilt the eye in the opposite direction to ensure the best possible surgical access. The knife tip is then precisely pressed against the most peripheral iris next to the point of angle adhesion and gently pressed posteriorly until the trabecular meshwork is fully exposed (Fig. 108-4). Only anterior to posterior movements of the knife tip are used to strip the synechiae from the angle wall. Horizontal sweeping of the knife or excessive force that can cause hemorrhage or cyclodialysis is avoided. The procedure is repeated in adjacent areas through the remaining paracenteses until the entire angle is opened. Limited hemorrhage might occur but usually localizes in the viscoelastic and does not disperse to obscure the surgical field. At the end of the procedure, the entire viscoelastic is removed and the temporal corneal incision is closed by stromal hydration or suturing. A large air bubble is injected to deepen the anterior chamber and push the iris posteriorly away from the angle wall. Intracameral injection of acetylcholine chloride is given to tighten the peripheral iris away from the angle if the iris sphincter is reactive. Alternatively, surgical pupilloplasty can be performed to constrict a paralytic, dilated pupil. Intracameral tissue plasminogen activator may be considered if the procedure has to be performed in an inflamed eye with a recent attack. Several modifications of the procedure including intraoperative visualization with a double-mirror goniolens,36 a dental mirror37 or intraocular endoscopy,38 scleral indentation39 and separation of the PAS by pulling with iris micro-forceps have been described.
Complications
Postoperative complications include a fibrinous aqueous reaction, transient IOP elevation, limited hyphema and photophobia from the paralytic mydriasis. A precise surgical technique, complete removal of the viscoelastic, and frequent steroids should prevent these mild complications. Surgical pupilloplasty or creating an artificial pupil may help alleviate the photophobia. The latter is achieved by performing a small, 4 mm diameter central circular capsulorrhexis and leaving the lens epithelium beneath the capsular remnant intact. Late postoperative opacification of the anterior capsule remnant results in a second smaller pupil. The third pupil can also be created by performing an Nd : YAG laser posterior capsulotomy (Fig. 108-5).
Postoperative Management
Patients are treated with antiglaucoma medications and frequent topical steroids. When the inflammation subsides, the medications are then slowly tapered off within a month. Occasionally, a release of aqueous through the paracenteses at the slit lamp may be performed if a high IOP rise occurs. Gonioscopy typically reveals opening of the angle structures with irregular pigmentation on the newly exposed trabecular meshwork and the angle wall (Fig. 108-6). Angle recession or a cyclodialysis cleft are undesirable findings. If successful, anterior segment optical coherence tomography or ultrasound biomicroscopy should demonstrate separation of the PAS and reopening more than half of the entire angle (Figs 108-2, 108-7).



Conclusion
Nearly 60% of patients with PAC continue to have persistent PAS and elevated IOP following laser treatment. Irreversible changes in the trabecular meshwork will eventually occur if the PAS remain untreated. Early stripping of the synechiae by GSL can restore the outflow. Filtration through the natural trabecular pathway should be more physiologic and reliable than the artificial channel of filtering surgery. Goniosynechialysis becomes more effective when performed after lens removal. Replacement of the natural lens with an IOL provides ample room within the anterior segment to perform the procedure and prevent PAS reformation. A clear view of the angle structures by gonioscopy is essential in performing the surgery. In addition to simplicity and safety, phaco-GSL offers a long-lasting control of IOP in >90% of PAC patients with a recent acute attack and uncontrolled IOP following laser treatment.

