Gastrointestinal Hormones and Neurotransmitters
Diego V. Bohórquez, Rodger A. Liddle
The GI tract relies on hormones and neurotransmitters to integrate signals arising in the lumen with whole-body homeostasis. As an example, satiety in the brain is to a great extent induced by the presence of food in the gut. This process begins with ingestion of nutrients that stimulate sensory cells in the intestinal epithelium that modulate food intake via the release of specific chemical messengers. GI hormones and neurotransmitters are intimately involved with every aspect of the digestive process, including ingestion and absorption of nutrients, so it is not surprising that these transmitters are essential for life.1,2 In this chapter, the critical role of the regulatory transmitters in GI function is analyzed by covering the following aspects: their synthesis and secretion from sensory epithelial cells, how food or other GI luminal factors trigger their release, the most representative members, and their importance in the context of disease.
Hormones and Neurotransmitters
The sensory cells of the GI epithelium, enteroendocrine cells, as well as neurons of the enteric nervous system are the main producers of chemical messengers, which are released in the form of hormones or neurotransmitters. Enteroendocrine cells reside in the intestinal mucosa as single cells that are scattered among more numerous enterocytes—the absorptive cells of the gut. Most enteroendocrine cells are oriented with their apical surface open to the lumen where they are exposed to food and other contents within the gut lumen. Upon stimulation, enteroendocrine cells release hormones from their basolateral surface, and these enter the paracellular space where they are taken up into the blood. In contrast to enteroendocrine cells, enteric neurons are found below the mucosal epithelium, and even though villi and crypts are richly innervated, enteric neurons are not believed to be directly exposed to food in the gut.
Unlike other endocrine organs where endocrine cells are concentrated in a single organ, the function of scattered hormone-containing cells of the GI tract has been questioned, and it becomes important to distinguish hormonal from neuronal actions.
Definitions
Established criteria exist for determining whether a candidate transmitter is a true hormone or a neurotransmitter. The first hormone to be discovered was secretin, when it was shown that injection of intestinal extracts into the blood stimulated pancreatic secretion.3 Since then the following criteria have been established to prove that a substance functions as a hormone. First, the stimulation of one organ must cause a distant response by acting through the blood. Second, the response must be independent of neural stimulation. Third, no response should occur in the absence of the secretory organ. And fourth, the response should be reproducible by applying pure amounts of the candidate hormone onto the target tissue. There are over 30 GI hormones that meet these criteria, and their singularities are discussed in “The Transmitters” section of this chapter.
Demonstrating that a chemical is a neurotransmitter is perhaps more challenging, but the following criteria are agreed to define a neurotransmitter. First, the candidate molecule must be present within a presynaptic neuron. Second, the transmitter must be released in response to presynaptic depolarization. And third, specific candidate receptors must be present on the postsynaptic cell.
Although hormones are commonly thought to reside exclusively in the endocrine system and neurotransmitters in the nervous system, both systems work closely together. Indeed, some cells exert both endocrine and neural actions. For example, peripheral sensory cells such as taste cells of the tongue and solitary chemosensory olfactory cells of the nose are known as paraneurons and can release both hormones in the bloodstream and neurotransmitters at synaptic connections.4 There is growing evidence that enteroendocrine cells have similar dual function.5,6 These observations extend the continuum between the endocrine and nervous systems.
Moreover, one transmitter can act both as a hormone and a neurotransmitter, depending on its location. Upon the ingestion of food, for example, CCK is typically released from enteroendocrine cells into the bloodstream to act as a hormone. However, CCK is also abundant in nerves of the GI tract and brain, where it is released at synaptic terminals to act as a neurotransmitter. This conservation of transmitters allows the same messenger to have different physiologic actions at different locations, and is made possible by the manner in which the transmitter is delivered to its target tissues.
Modes of Transmitter Release
Enteroendocrine transmitters can be released onto their targets through endocrine, paracrine, and autocrine signaling or synaptic neurotransmission (Fig. 4-1).
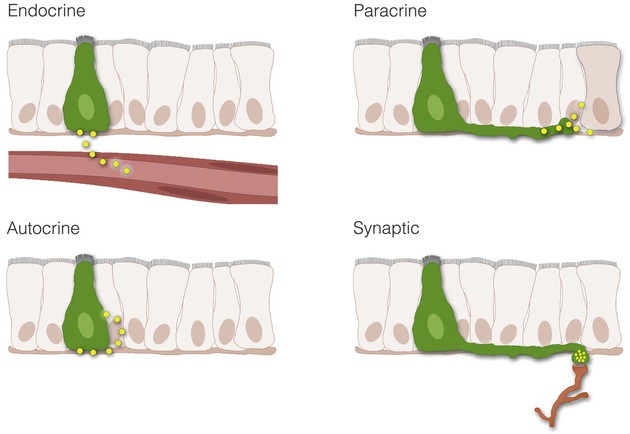
Endocrine
Endocrine communication occurs when transmitters are secreted into the bloodstream. The most common endocrine transmitters are peptides, lipids, and monoamines, collectively known as hormones. In the GI tract the most predominant type of hormone is the peptide form (e.g., peptide YY, gastrin, secretin). Hormones bind to specific receptors on the surface of target cells at remote sites and regulate metabolic processes.7
Paracrine
In contrast to endocrine mechanisms used to reach distant targets through the blood, signaling cells of the GI tract can also produce transmitters that act on neighboring cells. This process is known as paracrine signaling and is typical of enteroendocrine cells that produce somatostatin.8 Paracrine transmitters are secreted locally and cannot diffuse far. They bind to receptors on nearby cells to exert their biological actions. Once released, the transmitter is rapidly taken up by the target cell, is catabolized by extracellular enzymes, or becomes adherent to extracellular matrix, thus limiting the transmitter's ability to act at distant sites. Because paracrine signals act locally, their onset of action is generally rapid and can be terminated abruptly. By comparison, endocrine signaling takes much longer, and termination of signaling requires clearance of hormone from the circulation. Paracrine transmitters can be peptides (e.g., somatostatin) or monoamines (e.g., histamine).
Autocrine
Some cells possess cell surface receptors for their own messengers. In this way, when a messenger is released it can act on the same secreting cell. This mode of transmission is known as autocrine and has been demonstrated for several growth factors. Autocrine signaling has been implicated in the growth of certain cancers, including colorectal cancer (see Chapter 1).9
Neurotransmission
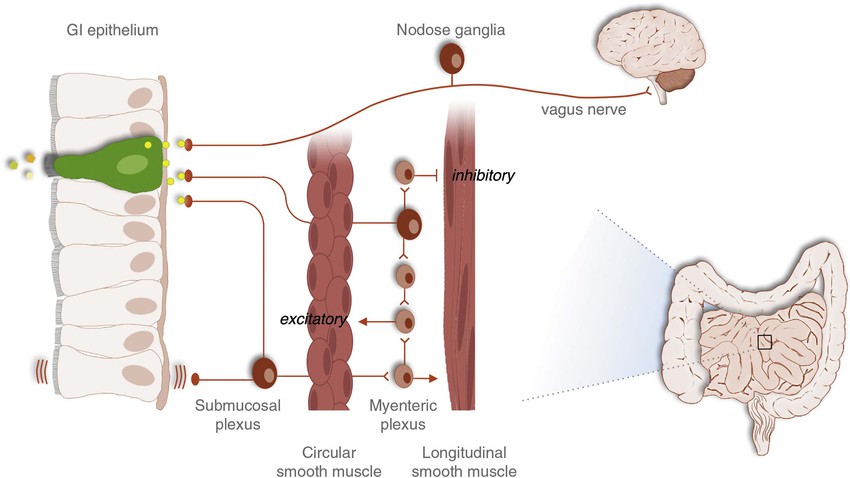
A fourth form of signaling in the GI tract is synaptic neurotransmission. This form of signaling is primarily used by the enteric nervous system, which is a complex network of nerve cells that must communicate efficiently to regulate numerous GI functions (Fig. 4-2). When neurons of the GI tract are activated, signals in the form of neurotransmitters are released at nerve-to-nerve junctions known as synapses. These structures help neurons deliver neurotransmitters at specific locations on the target cell and influence the function of other neurons, muscle cells, epithelial and secretory cells, and other specialized cells of the GI tract (e.g., enteric glia). Neurotransmitters are critical for the processes of digestion, including the coordination of gut motility and secretion. Although the GI tract secretes a variety of neurotransmitters, the most common are peptides like vasoactive intestinal polypeptide (VIP), or small molecules like acetylcholine and norepinephrine. Other molecules, such as nitric oxide (NO), can simply diffuse across the synaptic cleft to exert an effect on the postsynaptic cell. Some nerves actually release peptides or neurotransmitters directly into the blood. This process is called neurocrine signaling and may be used to cause systemic effects, depending on the transmitter released.
Box 4-1 lists the major hormones and neurotransmitters of the GI tract. Their actions depend on specific receptors located on target tissues; the specificity of neurotransmitter action is dependent on the precise location at which the nerve synapses with the target cell. Adjusting their synthesis, catabolism, or secretion regulates the transmitter concentration within the releasing cell. Once secreted, the concentration of a transmitter can be quickly modulated by catabolism or, in the case of neurotransmitters, reuptake into the secretory neuron. Many peptide transmitters have very short half-lives that are generally within the 2- to 5-minute range. This allows for rapid initiation and termination of signaling.
Transducing Signals From the Gastrointestinal Lumen
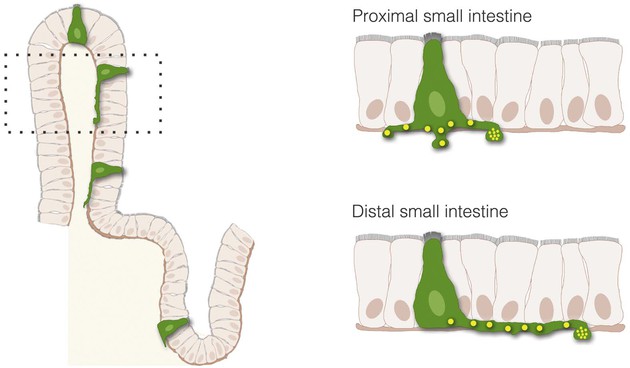
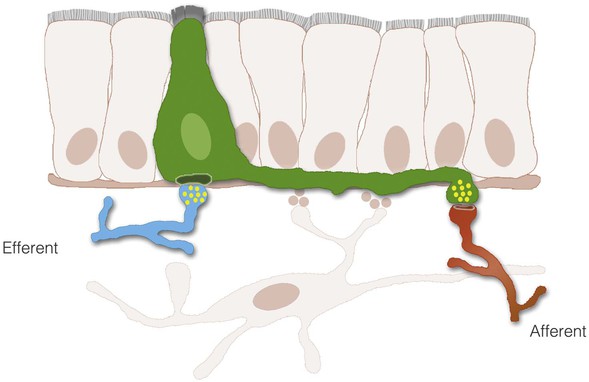
The process of nutrient sensing involves the activation of cell surface receptors that trigger the release of transmitters. The transmitters then either enter the bloodstream or activate sensory afferent nerves. Although the cells releasing the transmitters, enteroendocrine cells, are thought to interact with nerves indirectly through paracrine or endocrine signals, a new concept is emerging in which enteroendocrine cells and nerves actually communicate through synaptic connections.5,6 With the use of transgenic and advanced optical tools, enteroendocrine cells have been described to have several anatomic features observed in neurons, including dendritic-like spines and axon-like processes. The axon-like cytoplasmic processes vary in length from crypt to villus and from proximal to distal small intestine (Fig. 4-3). Moreover, enteroendocrine cells have the molecular components, genes, and proteins of synapses and connect to sensory neurons through synaptic-like connections (Fig. 4-4). These connections may have broad applications in the biology of GI function, including the transmission of sensory signals from nutrients and the GI microbiota. Some key components involved in the transduction of signals from the lumen of the gut to the rest of the body are discussed next.
Recognizing Signals Through Cell Surface Receptors
GI epithelial cells recognize molecules in the lumen using membrane-bound receptors. When activated, receptors transduce signals from the outside of the cell into the cytoplasm. Although the process is rather complex, there are key checkpoints at which the signaling cascade can be regulated. Some of these checkpoints occur at the moment of receptor activation, desensitization, internalization, and/or resensitization. Because of their regulatory potential, these are attractive targets for therapeutic intervention.
Receptors are grouped into major families depending on their structures and signaling mechanisms. The major families of cell surface receptors include G protein–coupled receptors, enzyme-coupled receptors, and ion channels.
G Protein–Coupled Receptors
G protein–coupled receptors (GPCRs) are typified by their 7-transmembrane domains. They are the most common family of protein receptors and have broad physiologic applications, ranging from sensing light in the retina, to allowing vision to sensing nutrients in the GI tract, to regulating food intake. When stimulated by a specific ligand, GPCRs undergo conformational changes, leading to their association with a G protein, hence their name. These G proteins are bound to the intracellular surface of the cell membrane10,11 and are composed of 3 distinct subunits: α, β, and γ. It is the Gα subunit that confers the name of the G protein (Table 4-1). The G proteins that stimulate an effector (e.g., adenylate cyclase) are classified as Gαs (s for “stimulatory”), whereas those that inhibit an effector are called Gαi (i for “inhibitory”).12-14 When the G protein acts on the effector, this causes a rapid increase in the intracellular concentrations of a second messenger (e.g., cyclic adenosine monophosphate [cAMP] or calcium). The second messenger then changes the activity of one or more protein kinases to catalyze the phosphorylation of an existing protein and ultimately modify the physiologic activity.
TABLE 4-1
Classification of G Protein α Subunits and Their Signaling Pathways
| Class | Signaling Pathways |
| Gαs | Adenylate cyclase, calcium channels |
| Gαi and Gαo | Adenylate cyclase, cyclic guanosine monophosphate, phosphodiesterase, c-Src, STAT 3 |
| Gαq | Phospholipase C-β |
| Gα12 and Gα13 | Sodium-hydrogen exchange |
In general the GPCR signaling mechanism involves the following events. In the resting state, the G protein complex does not interact with the receptor. When the ligand or first messenger binds to the receptor, the receptor changes its conformation and binds to the G protein complex. Once bound, there is a molecular substitution in the Gα subunit—a guanosine diphosphate (GDP) is replaced by a guanosine triphosphate (GTP). This replacement causes activation of the Gα subunit. The active Gα subunit then separates from the β and γ subunits and moves laterally in the membrane to activate an effector. Working through different Gα subunits, the activity of an effector can be up- or down-regulated. When the interaction is completed, the GTP bound to the Gα subunit is hydrolyzed back to GDP and dissociated from Gα. In this way, Gα moves back to reunite with the other 2 subunits. The effector then induces an increase in the intracellular concentration of a second messenger. The 2 most common second messengers are cAMP and calcium, and the mechanisms involving each are discussed next.
Signaling Through Cyclic Adenosine Monophosphate
This second messenger cAMP is a classic downstream effector of β-adrenergic receptors, a family of GPCRs that have been well characterized. These receptors are coupled to Gαs and activate adenyl cyclase, which catalyzes the conversion of ATP to cAMP. High concentrations of cAMP then modify the activity of protein kinase A (PKA), which ultimately modulates rate-limiting enzymes involved in important physiologic functions. For example, modulation of glycogen phosphorylase increases the conversion of glycogen to glucose-1 phosphate, leading to a rise in blood glucose levels.
Signaling Through Calcium
GPCRs associated with Gαq subunits use Ca2+ as a second messenger (see Table 4-1). An increase in intracellular concentrations of Ca2+ can result from the activation of voltage-gated Ca2+ channels, ligand-gated Ca2+ channels, or the release of cytosolic Ca2+ activated by membrane phospholipids. The latter is triggered by activation of GPCRs associated with Gαq. When active, Gαq moves along the cell membrane to activate the enzyme phospholipase Cβ. Phospholipase Cβ then cleaves the membrane phospholipid phosphatidyl inositol bisphosphate into diacylglycerol and inositol 1,4,5 trisphosphate (IP3), generating 2 potential signaling molecules. Diacylglycerol in the presence of Ca2+ activates protein kinase C. In addition, a rise in Ca2+ levels from internal stores can also activate Ca2+–calmodulin kinase. In this way, 2 different kinases are activated, Ca-calmodulin kinase by increasing cytosolic Ca2+ and protein kinase C by the action of diacylglycerol and Ca2+. These kinases then catalyze the phosphorylation of target proteins within the cell. Following receptor activation, IP3 moves from the plasma membrane into the cytoplasm to bind IP3 receptors located on the endoplasmic reticulum and mitochondria. IP3 receptor binding causes release of Ca2+ from intracellular organelles to further increase cytoplasmic Ca2+ concentrations. Ultimately, Ca2+ cytoplasmic concentrations are restored to normal by active transport out of the cell or by reuptake into intracellular Ca+2 stores.
If the cell is over-stimulated, a process of adaptation occurs to prevent the cell from over-responding. Attenuation of signaling occurs through either ligand-induced receptor desensitization or receptor internalization. The receptor is desensitized by means of phosphorylation. Phosphorylation can also further label the receptor for internalization, which is accomplished by activation of specific receptor kinases and recruitment of arrestin-like molecules that uncouple the receptor from the G protein.15 Uncoupling and subsequent receptor internalization ends signaling and eventually restores cell responsiveness.
Enzyme–Coupled Receptors
The most representative of the enzyme-coupled receptors are the tyrosine kinase receptor family. These receptors are primarily targets of growth factors like EGF and are unique in that they are both a receptor and a tyrosine kinase. When activated, the receptors catalyze the transfer of phosphate from ATP to the target proteins. Enzyme-coupled receptors are composed of 3 domains: a ligand-binding extracellular domain, a transmembrane domain, and a cytoplasmic domain. The cytoplasmic domain contains a protein tyrosine kinase region and substrate region for agonist-activated receptor phosphorylation. In this way, phosphorylation from other kinases or autophosphorylation can occur to modulate the activity of the tyrosine kinase receptor.16 In general, receptor tyrosine kinases exist in the cell membrane as monomers. However, with ligand binding, these receptors dimerize, autophosphorylate, and initiate other intracellular signal transduction pathways that ultimately modulate physiologic function.17 Receptor tyrosine kinases are further discussed in Chapter 1 in relation to cellular growth and neoplasia.18
There are several other types of enzyme-coupled receptors, including receptor guanylate cyclases, non-receptor tyrosine kinases, receptor tyrosine phosphatases, and receptor serine/threonine kinases. Although these receptors act through different enzymes, the signaling principles remain similar to those of tyrosine kinase receptors.
Ion Channel–Coupled Receptors
Ion channel–coupled receptors are involved in rapid signaling between cells. This type of receptor is important in tissues where electrical impulses drive signaling, like nerve cells and muscle. In nerve cells, ion channels open or close in response to a relatively small number of neurotransmitters and allow the flow of particular ions across the plasma membrane. The kinetics of the ion flow depends on the concentration inside and outside the cell. This flow of ions regulates the excitability of the target cell to ultimately trigger processes such as neurotransmission, muscle contraction, electrolyte and fluid secretion, or hormone release.
An example of this type of receptor is the transient receptor potential cation channel M5, better known as TRPM5. This ion channel receptor is activated by elevated intracellular Ca2+ concentrations and is a key component in the transduction of the taste signals bitter, sweet, and umami.19 It has been recently shown to mediate the release of opioids and hormones like CCK from enteroendocrine cells.20 Thus, ion channel–coupled receptors can be attractive targets to modulate the function of sensory cells in the epithelium of the GI tract.
Nutrient Chemosensing
Lipids
Lipids in the intestinal lumen are potent inducers of satiety and modulators of whole body metabolism. Although the mechanisms are not completely understood, it has been recently demonstrated that specific lipids are recognized by cell surface receptors that activate the release of several hormones, including CCK, peptide YY, and glucagon-like peptide 1.
The lipids can be in the form of triglycerides or free fatty acids of various chain lengths. Different lipids are recognized by different receptors. The Gq-coupled GPCRs 40 (i.e., FFAR1) and 120 respond to medium- and long-chain fatty acids, whereas the Gαi-coupled GPR41 (i.e., FFAR3) and GPR43 (i.e., FFAR2) bind to short-chain fatty acids of 2 to 5 carbons.21 It is possible some GPCRs respond to lipids in the lumen of the gut. Other non-GPCRs are also involved in lipid sensing, such as the immunoglobulin-like domain-containing receptor (ILDR).21a ILDR is expressed in CCK cells and is activated by the combination of fatty acids and lipoproteins, suggesting that fatty acids must be absorbed to stimulate CCK secretion. Although the specific location of most nutrient receptors has yet to be determined, it may be that at least some lipids have to be digested and absorbed prior to activating hormone release. This hypothesis is supported by studies in which the infusion of lipid in the intestine triggers hormone secretion but only if chylomicrons, lipoprotein particles formed from absorbed lipids, are allowed to form.22
Some lipid-generated sensory signals appear to travel through afferent fibers of the vagus nerve. Infusion of lipids into the duodenum increases brown fat temperature, and this effect is abolished if lipids are infused along with tetracaine, a potent local anesthetic used to block vagal afferents activation.23 Signals traveling through afferent nerves or the bloodstream ultimately induce homeostatic changes (e.g., satiety, body temperature, GI motility) in response to the presence of nutrients in the GI lumen.
Proteins and Amino Acids
Proteins can also be potent stimulants of GI hormone secretion. Most proteins stimulate hormone secretion only when digested to peptones and amino acids. Recently, enteroendocrine cells have been found to express several classes of amino acid receptors that mediate hormone secretion. The calcium-sensing receptor (CaSR), which was originally identified for its ability to detect and respond to extracellular Ca2+ and regulate calcium homeostasis in the kidney and parathyroid gland,24 also recognizes L-amino acids and di- and tripeptides.25 A clear role for CaSR has been established in the regulation of L-amino acid–stimulated gastrin release by gastrin (G) cells and gastric acid secretion by parietal cells.26,27 The aromatic amino acids phenylalanine and tryptophan are the most potent amino acids for stimulating CaSR and are also the most potent for stimulating CCK secretion. The discovery of CaSR in CCK cells and its link to secretion support its physiologic importance as a nutrient sensor in the GI tract. CaSR also appears to mediate the secretion of glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide (GLP)-1, and peptide tyrosine tyrosine (PYY).28-30
Another amino acid–sensing receptor closely related to CaSR is the G protein–coupled receptor GPRC6A. GPRC6A responds to basic amino acids and is expressed in taste cells and enteroendocrine cells of the distal small intestine where the secretion of GLP-1 is mediated.31 Genetic deletion of GPRC6A leads to diet-induced obesity, implying that this receptor is important for metabolic regulation.32 Finally, the taste receptors T1R1/T1R3 also recognize acidic amino acids and do not appear to be restricted to taste cells of the tongue, but instead are distributed in chemosensory cells throughout the body. Together, CaSR, GPRC6A, and T1R1/T1R3 respond to all of the 20 L-amino acids and represent a comprehensive mechanism to sense amino acid nutrient stimuli.
Partially digested protein in the form of peptones can also stimulate hormone secretion. The G protein–coupled receptor GPR93 is not only a lysophosphatidic acid receptor but is also activated by peptone.33 GPR93 is expressed in enterocytes and enteroendocrine cells, where its activation has been coupled to CCK secretion.34 Thus, GPR93 may be the mechanism by which peptone stimulates CCK release following a meal.
Some intact proteins stimulate hormone secretion indirectly through a class of endogenous luminally active hormone releasing factors including luminal CCK releasing factor (LCRF)35 and diazepam binding inhibitor (DBI).36 The most potent proteins are those that compete for trypsin binding and allow the endogenous releasing factor to escape proteolytic digestion within the gut lumen.
Tastants
Sensing tastants in various foods is important to regulate pleasure, reward, food intake, and other important metabolic functions. The GI tract detects chemicals and toxins through specific receptors expressed by specialized chemosensory cells. These cells are best characterized in the tongue, where they are concentrated in taste buds. Taste receptor cells can detect chemicals that give rise to the 5 different flavors: sweet, salty, sour, bitter, and umami—the savory taste of soy sauce. Although this is an active area of research, only the sensing mechanisms for sweet, bitter, and umami flavors are well understood. These 3 flavors are mediated by the activation of 2 families of GPCRs: taste-1 receptors (T1Rs) and taste-2 receptors (T2Rs). In humans, there are 30 T2R proteins and 3 T1Rs, T1R1, T1R2, and T1R3.37-39
Sweet and umami flavors are recognized by T1Rs. In the tongue, T1R1 and T1R2 are expressed in separate taste receptor cells, but always along with T1R3. In this way, the receptors form heterodimers that allow detection of sweet ligands in the case of T1R2 + T1R3, and umami in the case of T1R1 + T1R3.40 T1Rs are also expressed in enteroendocrine cells.41 Here, the binding of glucose to T1R2 + T1R3 receptors in enteroendocrine cells in the gut lumen leads to secretion of incretin hormones like glucagon-like peptide (GLP)-1. GLP-1 ultimately modulates a wide variety of functions, including insulin secretion, nutrient absorption, and gut motility.42 Consequently, gut-expressed taste signaling has become an active area of research to develop therapies for diet-related disorders like type 2 diabetes.43
Bitter perception functions as a warning signal against the ingestion of toxic substances through direct taste aversion, induction of the pharyngeal gag reflex, and nausea. The wide array of T2Rs present in the tongue and gut are set to recognize bitter compounds such as toxic alkaloids in plants.44 It is believed that bitter compounds that bypass T2Rs in the tongue are recognized by T2Rs in the gut, serving as a backup mechanism for inducing a protective response such as vomiting.45 Activation of T2Rs and their associated Gα-gustducin protein result in a rapid increase in cytosolic Ca2+, which stimulates membrane depolarization and hormone release. In the gut, bitter chemicals can stimulate the release of CCK from enteroendocrine cells to slow gastric emptying and decrease appetite, reducing the likelihood of toxin absorption.42,45,46
Sensing the Microbiome
Enteroendocrine cells typically have a small, narrow opening to the luminal surface. Although it has been long been assumed that nutrients stimulate enteroendocrine cells at their apical portion, there are some reports that absorbed and not luminal nutrients stimulate gut hormone release.22 It is possible that the apical portion of enteroendocrine cells open to the gut lumen may serve to sense bacterial inputs. Evidence supporting this hypothesis comes from the fact that some bacterial Toll-like receptors (e.g., TLRs 4, 5, and 9) are exclusively expressed in enteroendocrine cells.47 When these specific TLRs are stimulated with bacterial ligands (e.g., LPS or flagellin), CCK and several chemokines are secreted. Remarkably, cytokines and defensins are secreted from an enteroendocrine cell line (i.e., STC-1) only in response to bacterial ligands and not to fatty acids. Moreover, silencing MyD88, a central mediator of TLR signaling, reduces CCK secretion stimulated by bacterial ligands but not by fatty acids.48 This evidence suggests that there may be 2 different sensing pathways in enteroendocrine cells, 1 for bacteria and another for nutrients.
It has long been assumed that chemosensory receptors on enteroendocrine cells reside on the apical surface, which is open to the gut lumen. However, this has not yet been demonstrated, and recent evidence suggests that some nutrients stimulate enteroendocrine cells when exposed to the basal lateral surface. Because gut microbiota reside in the lumen of the GI tract, it is likely that Toll-like receptors are located on microvilli. In the future, elucidating the location of receptors on enteroendocrine cells may facilitate the design of drugs to target specific receptors and modulate the secretion of hormones involved in appetite regulation and insulin secretion.
Other Factors That Stimulate Transmitter Release
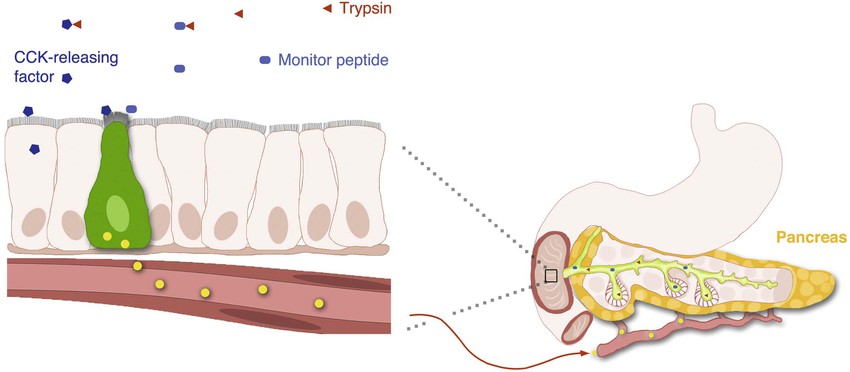
There is evidence that GI hormones can be released by certain non-nutrient factors present in the lumen of the gut (Fig. 4-5). CCK was the first hormone shown to be regulated by an intraluminal releasing factor.49,50 Luminal CCK-releasing factor (LCRF) was purified from intestinal washings and shown to stimulate CCK release when instilled into the lumen of animals. Other luminal factors causing the release of CCK are the diazepam-binding inhibitor (DBI) and the pancreatic monitor peptide.51,52 It has been proposed that a secretin releasing factor regulates secretin secretion in an acid-sensitive way.53 The pancreatic secretory trypsin inhibitor, better known as monitor peptide, is an endogenous trypsin inhibitor produced by pancreatic acinar cells.54 When secreted into the duodenum, monitor peptide directly stimulates CCK secretion from I cells. These proteins act directly on enteroendocrine cells, most likely through cell surface receptors. The existence of these releasing factors highlights the existence of underappreciated bioactive molecules within the lumen of the gut.
The Transmitters
The same factors that stimulate transmitter release simultaneously modulate the expression of specific transmitter genes. The genes for most of the known GI peptides have now been identified and are regulated within cell types by specific gene regulatory elements. Gut hormone gene expression is generally linked to peptide production and regulated according to the physiologic needs of the organism. Once a biological response is elicited, signals may then be sent back to the endocrine cell to “turn off” hormone secretion. This negative feedback mechanism is common to many physiologic systems and avoids excess production and secretion of hormone.
All GI peptides are synthesized via transcription of DNA into messenger RNA, which is subsequently translated into precursor proteins known as preprohormones. The newly translated protein contains a signal sequence that directs it to the endoplasmic reticulum to prepare the peptide precursor for structural modifications.55 These precursors are transported to the Golgi apparatus where further structure modifications occur before the peptide is packaged in secretory granules. Secretory granules may be targeted for immediate release or stored in close proximity to the plasma membrane, ready to be released. Although many hormones are produced from a single gene, there can be multiple molecular forms in tissues and blood. The different molecular forms result from differences in pre-translational or post-translational processing. A common pre-translational processing mechanism is the alternative splicing of mRNA, which generates unique peptides from the same gene.
Post-translational modifications can occur by cleavage of precursor molecules, where enzymatic cleavage of the signal peptide produces a prohormone. Other post-translational features that result in mature GI peptides include peptide cleavage to smaller forms (e.g., somatostatin), amidation of the carboxyl terminus (e.g., gastrin), and sulfation of tyrosine residues (e.g., CCK). These processing steps are critical for biological activity of the hormone. For example, sulfated CCK is 100-fold more potent than its unsulfated form. The vast biochemical complexity of gastroenteropancreatic hormones is evident in the different tissues that secrete these peptides. GI peptides are secreted from endocrine as well as nervous tissue, so the distinct tissue involved often determines the processing steps for production of the peptide. Many hormone genes are capable of manufacturing alternatively spliced mRNAs or proteins that undergo different post-translational processing and ultimately produce hormones of different sizes. These modifications are important for receptor binding, signal transduction, and consequent cellular responses.56 The discussion that follows outlines the major characteristics of GI transmitters, including neuropeptides, neurotransmitters, and other transmitters.
Gut Neuropeptides
Gastrin
As discussed in more detail in Chapter 50, gastrin is the major hormone that stimulates gastric acid secretion. Gastrin was found to have growth-promoting effects on the gastric mucosa and possibly some cancers.57 Human gastrin is the product of a single gene located on chromosome 17. The active hormone is generated from a precursor peptide called preprogastrin. Human preprogastrin contains 101 amino acids (AAs), including a signal peptide (21 AAs), spacer sequence (37 AAs), gastrin component (34 AAs), and a 9-AA extension at the carboxyl terminus. The enzymatic processing of preprogastrin produces all the known physiologically active forms of gastrin.
Preprogastrin is processed into progastrin and gastrin peptide fragments of various sizes by sequential enzymatic cleavage. The 2 major forms of gastrin are G34 and G17, although smaller forms exist. The common feature of all gastrins is an amidated tetrapeptide (Try-Met-Asp-Phe-NH2) carboxyl terminus, which imparts full biological activity. Modification by sulfation at tyrosine residues produces alternative gastrin forms of equal biological potency. A nonamidated form of gastrin known as glycine-extended gastrin is produced by colonic mucosa. Glycine-extended gastrin has been shown in animal models to stimulate proliferation of normal colonic mucosa and enhance the development of colorectal cancer. It is not known whether local production of this form of gastrin contributes to human colon carcinogenesis, and the receptor for glycine-extended gastrin has not been identified.58
Most gastrin is produced in endocrine cells of the gastric antrum.59 Much smaller amounts of gastrin are produced in other regions of the GI tract, including the proximal stomach, duodenum, jejunum, ileum, and pancreas. Gastrin has also been found outside the GI tract, including in the brain, adrenal gland, respiratory tract, and reproductive organs, although its biological role in these sites is unknown.
The receptors for gastrin and CCK are related and constitute the so-called gastrin-CCK receptor family. The CCK-1 and CCK-2 (previously known as CCK-A and -B) receptor complementary DNAs were cloned from the pancreas and brain, respectively, after which it was recognized that the CCK-2 receptor is identical to the gastrin receptor of the stomach.60
The CCK-1 receptor is present in the gallbladder and, in most species, the pancreas. The CCK-1 receptor has a 1000-fold higher affinity for CCK than for gastrin. The CCK-1 and CCK-2 gastrin receptors have over 50% sequence homology and respond differently to various receptor antagonists and to gastrin.
Gastrin is released from specialized endocrine cells (G cells) into the circulation in response to a meal. The specific components of a meal that stimulate gastrin release include protein, peptides, and amino acids. Gastrin release is profoundly influenced by the pH of the stomach. Fasting and increased gastric acidity inhibit gastrin release, whereas a high gastric pH is a strong stimulus for its secretion.
Hypergastrinemia occurs in pathologic states associated with decreased acid production, such as chronic atrophic gastritis (see Chapter 52). Serum gastrin levels can also become elevated in patients on prolonged acid-suppressive medications, such as histamine receptor antagonists and proton pump inhibitors. Hypergastrinemia in these conditions is caused by stimulation of gastrin production by the alkaline pH environment. Another important but far less common cause of hypergastrinemia is a gastrin-producing tumor, also known as Zollinger-Ellison syndrome (see Chapter 33).
The gastrin analog pentagastrin has been used clinically to stimulate histamine and gastric acid secretion in diagnostic tests of acid secretory capacity.
Cholecystokinin
CCK is a peptide transmitter produced primarily by enteroendocrine cells of the proximal small intestine and is secreted into the blood following ingestion of a meal. Circulating CCK binds to specific CCK-1 receptors on the gallbladder, pancreas, smooth muscle of the stomach, and peripheral nerves to stimulate gallbladder contraction and pancreatic secretion, regulate gastric emptying and bowel motility, and induce satiety.61 These effects serve to coordinate the ingestion, digestion, and absorption of dietary nutrients. Ingested fat and protein are the major food components that stimulate CCK release.
CCK was originally identified as a 33–amino acid peptide. However, since its discovery larger and smaller forms of CCK have been isolated from blood, intestine, and brain. All forms of CCK are produced from a single gene by post-translational processing of a preprohormone. Forms of CCK ranging in size from CCK-58 to CCK-8 have similar biological activities.62
CCK is the major hormonal regulator of gallbladder contraction. It also plays an important role in regulating meal-stimulated pancreatic secretion (see Chapter 56). In many species, this latter effect is mediated directly through receptors on pancreatic acinar cells, but in humans, in whom pancreatic CCK-1 receptors are less abundant, CCK appears to stimulate pancreatic secretion indirectly through enteropancreatic neurons that possess CCK-1 receptors. In some species, CCK has trophic effects on the pancreas, although its potential role in human pancreatic neoplasia is speculative. CCK also has been shown to delay gastric emptying.63 This action may be important in coordinating the delivery of food from the stomach to the intestine. CCK has been proposed as a major mediator of satiety and food intake, an effect that is particularly noticeable when food is in the stomach or intestine. CCK inhibits gastric acid secretion by binding to CCK-1 receptors on somatostatin (D) cells in the antrum and oxyntic mucosa. Somatostatin acts locally to inhibit gastrin release from adjacent G cells and directly inhibits acid secretion from parietal cells.64
Clinically, CCK has been used together with secretin to stimulate pancreatic secretion for pancreatic function testing. It is also used radiographically or scintigraphically to evaluate gallbladder contractility. There are no known diseases of CCK excess. Low CCK levels have been reported in individuals with celiac disease who have reduced intestinal mucosal surface area and in those with bulimia nervosa.65,66 Elevated levels of CCK have been reported in some patients with chronic pancreatitis (see Chapter 59), presumably because of reduced pancreatic enzyme secretion and interruption of negative feedback regulation of CCK release.67
Secretin
The first hormone, secretin, was discovered when it was observed that intestinal extracts, when injected intravenously into dogs, caused pancreatic secretion.68 Secretin is released by acid in the duodenum and stimulates pancreatic fluid and bicarbonate secretion, leading to neutralization of acidic chyme in the intestine (see Chapter 56). Secretin also inhibits gastric acid secretion (see Chapter 50) and intestinal motility.
Human secretin is a 27–amino acid peptide and, similar to many other GI peptides, is amidated at the carboxyl terminus. It is the founding member of the secretin-glucagon-VIP family of structurally related GI hormones. Secretin is selectively expressed in specialized enteroendocrine cells of the small intestine called S cells.69
The secretin receptor is a member of a large family of GPCRs that is structurally similar to receptors for glucagon, calcitonin, parathyroid hormone, pituitary adenylate cyclase–activating peptide (PACAP), and vasoactive intestinal polypeptide (VIP).
One of the major physiologic actions of secretin is stimulation of pancreatic fluid and bicarbonate secretion (see Chapter 56). Pancreatic bicarbonate, on reaching the duodenum, neutralizes gastric acid and raises the duodenal pH, thereby “turning off” secretin release (negative feedback). It has been suggested that acid-stimulated secretin release is regulated by an endogenous intestinal secretin-releasing factor.70 This peptide stimulates secretin release from S cells until the flow of pancreatic proteases is sufficient to degrade the releasing factor and terminate secretin release.
Although the primary action of secretin is to produce pancreatic fluid and bicarbonate secretion, it is also an enterogastrone, a substance that is released when fat is present in the GI lumen and that inhibits gastric acid secretion. In physiologic concentrations, secretin inhibits gastrin release, gastric acid secretion, and gastric motility.71 The most common clinical application of secretin is in the diagnosis of gastrin-secreting tumors,72 as discussed in Chapter 33.
Vasoactive Intestinal Polypeptide
VIP is a neuromodulator that has broad significance in intestinal physiology. VIP is a potent vasodilator that increases blood flow in the GI tract and causes smooth muscle relaxation and epithelial cell secretion.73,74 As a chemical messenger, VIP is released from nerve terminals and acts locally on cells bearing VIP receptors. VIP belongs to a family of GI peptides, including secretin and glucagon, that are structurally related. The VIP receptor is a GPCR that stimulates intracellular cAMP generation.
Like other GI peptides, VIP is synthesized as a precursor molecule that is cleaved to an active peptide of 28 amino acids. VIP is expressed primarily in neurons of the peripheral-enteric and central nervous systems (CNS) and is released along with other peptides, including primarily PHI and/or PHM (see Box 4-1).75
VIP is an important neurotransmitter throughout the central and peripheral nervous systems.76 Because of its wide distribution, VIP has effects on many organ systems; most notably, in the GI tract, VIP stimulates fluid and electrolyte secretion from intestinal epithelium and bile duct cholangiocytes.77,78
VIP, along with NO, is a primary component of nonadrenergic, noncholinergic nerve transmission in the gut.79 GI smooth muscle exhibits a basal tone, or sustained tension, caused by rhythmic depolarizations of the smooth muscle membrane potential. VIP serves as an inhibitory transmitter of this rhythmic activity, causing membrane hyperpolarization and subsequent relaxation of GI smooth muscle. Accordingly, VIP is an important neuromodulator of sphincters of the GI tract, including the lower esophageal sphincter and sphincter of Oddi. In certain pathologic conditions, such as achalasia and Hirschsprung's disease, the lack of VIP innervation is believed to play a major role in defective esophageal relaxation and bowel dysmotility, respectively.80,81
Unlike GI endocrine cells that line the mucosa of the gut, VIP is produced and released from neurons, and it is likely that most measurable VIP in serum is of neuronal origin. Normally, serum VIP levels are low and do not appreciably change with a meal. However, in pancreatic cholera, also known as Verner-Morrison syndrome and manifested by watery diarrhea, hypokalemia, and achlorhydria,82 VIP levels can be extraordinarily high.77 VIP-secreting tumors usually produce a voluminous diarrhea (see Chapter 33).83
Glucagon
Glucagon is synthesized and released from pancreatic alpha cells and from intestinal L cells of the ileum and colon. Pancreatic glucagon is a 29–amino acid peptide that regulates glucose homeostasis via gluconeogenesis, glycogenolysis, and lipolysis and is counter-regulatory to insulin. The gene for glucagon encodes not only preproglucagon but also glucagon-like peptides (GLPs). This precursor peptide consists of a signal peptide, a glucagon-related polypeptide, glucagon, and GLP-1 and GLP-2. Tissue-specific peptide processing occurs through prohormone convertases that produce glucagon in the pancreas and GLP-1 and GLP-2 in the intestine (Fig. 4-6).84,85
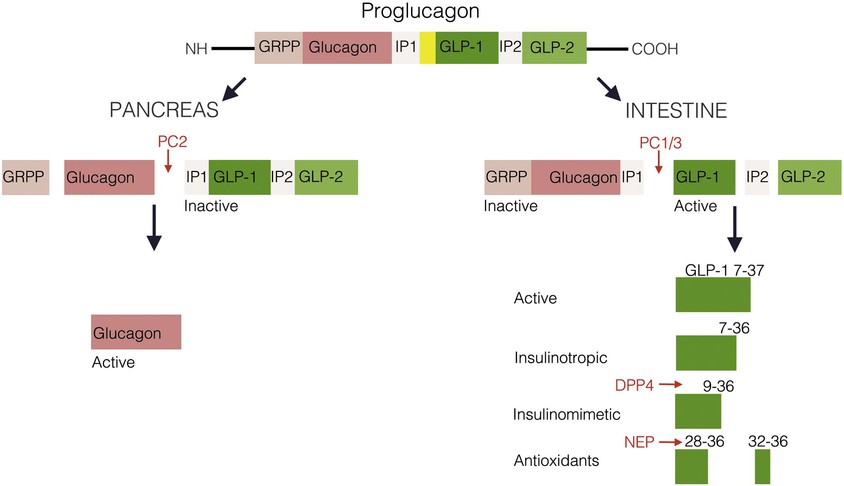
Glucagon and GLP-1 regulate glucose homeostasis.86 Glucagon is released into the blood from alpha cells in the endocrine pancreas in response to a meal and binds to GPCRs on skeletal muscle and the liver to exert its glucoregulatory effects. GLP-1 stimulates insulin secretion and augments the insulin-releasing effects of glucose on the pancreatic beta cell (see enteroinsular axis under “Diabetes and the Gastrointestinal Tract” later in the chapter). GLP-1 analogs have been developed for the treatment of type 2 diabetes mellitus. A long-acting human GLP-1 analog improves beta cell function and can lower body weight in patients with type 2 diabetes.87,88 GLP-2 is an intestinal growth factor that increases villus height, stimulates intestinal crypt proliferation and prevents enterocyte apoptosis. GLP-2 agonists have therapeutic implications in the maintenance of the GI mucosal mass and the reversal of villus atrophy and are being developed for the treatment of short bowel syndrome.
Glucose-Dependent Insulinotropic Polypeptide
GIP was discovered based on its ability to inhibit gastric acid secretion (enterogastrone effect) and was originally termed gastric inhibitory polypeptide. It was subsequently shown that the effects on gastric acid secretion occur only at very high concentrations that are above the physiologic range. However, GIP has potent effects on insulin release that (like GLP-1) potentiates glucose-stimulated insulin secretion.89 Based on this action, GIP was redefined as glucose-dependent insulinotropic polypeptide.
GIP is a 42–amino acid peptide produced by K cells in the mucosa of the small intestine. GIP is released into the blood in response to ingestion of glucose or fat. In the presence of elevated blood glucose levels, GIP binds to its receptor on pancreatic beta cells, activating adenylate cyclase and other pathways that increase intracellular calcium concentrations, leading to insulin secretion. Importantly, however, the effects on insulin secretion occur only if hyperglycemia exists; GIP does not stimulate insulin release under normoglycemic conditions.
GIP receptors are also expressed on adipocytes through which GIP augments triglyceride storage, which may contribute to fat accumulation. Based on the insulinotropic properties of GIP, coupled with its effects on adipocytes, it has been proposed that GIP may play a role in obesity and development of insulin resistance associated with type 2 diabetes mellitus.90 Consistent with this proposal was the experimental finding that mice lacking the GIP receptor do not gain weight when placed on a high-fat diet.91 It remains to be seen whether GIP antagonists can be used to treat obesity. In rare circumstances, receptors for GIP may be aberrantly expressed in the adrenal cortex, resulting in food-dependent Cushing's syndrome.92,93
Pancreatic Polypeptide Family
Originally isolated during the preparation of insulin, pancreatic polypeptide (PP) is the founding member of the PP family.94 The PP family of peptides includes neuropeptide Y (NPY) and peptide tyrosine tyrosine (PYY), which were discovered because of the presence of a C-terminal tyrosine amide.95,96 PP is stored and secreted from specialized pancreatic endocrine cells (PP cells),97 whereas NPY is a principal neurotransmitter found in the central and peripheral nervous systems.98 PYY has been localized to enteroendocrine cells throughout the GI tract but is found in greatest concentrations in the ileum and colon, where it is produced in L cells along with glucagon-like peptides.99
The PP-PYY-NPY family of peptides functions as endocrine, paracrine, and neurocrine transmitters in the regulation of a number of actions that result from binding to 1 of 5 receptor subtypes.100 PP inhibits pancreatic exocrine secretion, gallbladder contraction, and gut motility.101 PYY inhibits vagally stimulated gastric acid secretion and other motor and secretory functions.102 An abbreviated form of PYY lacking the first 2 amino acids of the normally produced 36 amino acid peptide, PYY3-36, has been shown to reduce food intake when administered to humans, indicating that intestinally released peptide may play a role in regulating meal size.103 NPY is one of the most abundant peptides in the CNS and, in contrast to PYY3-36, is a potent stimulant of food intake.104 Peripherally, NPY affects vascular and GI smooth muscle function.105
Substance P and the Tachykinins
Substance P belongs to the tachykinin family of peptides, which includes neurokinin A and neurokinin B. The tachykinins are found throughout the peripheral and central nervous systems and are important mediators of neuropathic inflammation.106 Tachykinins, as a group, are encoded by 2 genes that produce preprotachykinin A and pre-protachykinin B. Common to both is a well-conserved C-terminal pentapeptide. Transcriptional and translational processing produce substance P, neurokinin A, and/or neurokinin B, which are regulated in large part by alternative splicing. These peptides function primarily as neuropeptides. Substance P is a neurotransmitter of primary sensory afferent neurons and binds to specific receptors in lamina I of the spinal cord.107 Three receptors for this family of peptides have been identified: NK-1, NK-2, and NK-3.108 Substance P is the primary ligand for the NK-1 receptor, neurokinin A for the NK-2 receptor, and neurokinin B for the NK-3 receptor. However, all these peptides can bind and signal through all 3 receptor subtypes.
Substance P has been implicated as a primary mediator of neurogenic inflammation. In the intestine, Clostridium difficile–initiated experimental colitis results from toxin-induced release of substance P and consequent activation of the NK-1 receptor.109 These inflammatory sequelae can be blocked by substance P receptor antagonists. Substance P receptors are more abundant in the intestine of patients with ulcerative colitis and Crohn's disease.110 NK-1 receptor antagonists such as aprepitant are potent antiemetic drugs (see Chapter 15).
Somatostatin
Somatostatin is a 14–amino acid cyclic peptide that was initially identified as an inhibitor of growth hormone secretion. Since its discovery, it has been found in almost every organ in the body and throughout the GI tract. In the gut, somatostatin is produced by D cells in the gastric and intestinal mucosa and islets of the pancreas, as well as enteric neurons.111 Somatostatin has a number of pharmacologic effects that are mostly inhibitory.
In the stomach, somatostatin plays an important role in regulating gastric acid secretion.112 In the antrum, D cells are open to the lumen, where they are directly exposed to acid. A low gastric pH stimulates D cells that lie in close proximity to gastrin-producing cells to secrete somatostatin and inhibit gastrin release by a paracrine effect (see Chapter 50). Reduced gastrin secretion decreases the stimulus for acid production and the pH of the stomach contents rises. Thus, some of the inhibitory effects of gastric acid on gastrin release (see earlier, “Gastrin”) are mediated by somatostatin.
Somatostatin release is also influenced by mechanical stimulation, dietary components of a meal, including protein, fat, and glucose, and other hormones and neurotransmitters.113 Muscarinic stimulation appears to be the most important neural stimulus to somatostatin secretion.
At least 5 somatostatin receptors have been identified that account for divergent pharmacologic properties.114 Receptor subtypes 2 and 3 couple to inhibitory G proteins, but receptor subtype 1 does not. Only somatostatin receptor subtype 3 inhibits adenylate cyclase. The inhibitory effects of somatostatin are mediated by a decrease in cAMP, Ca2+ channel inhibition, or K+ channel opening.
In the gut, somatostatin has broad inhibitory actions. In addition to effects on gastric acid, somatostatin reduces pepsinogen secretion. Somatostatin profoundly inhibits pancreatic enzyme, fluid, and bicarbonate secretion and reduces bile flow.115 The effects of somatostatin on gut motility are largely inhibitory, with the exception that it stimulates the migrating motor complex, possibly through effects on motilin. Somatostatin also reduces intestinal transport of nutrients and fluid, reduces splanchnic blood flow, and has inhibitory effects on tissue growth and proliferation.116,117
Because of its varied physiologic effects, somatostatin has several clinically important pharmacologic uses. Many endocrine cells possess somatostatin receptors and are sensitive to inhibitory regulation. Therefore, somatostatin and more recently developed somatostatin analogs are used to treat conditions of hormone excess produced by endocrine tumors (e.g., acromegaly, carcinoid tumors, islet cell tumors [including gastrinomas]).118 Its ability to reduce splanchnic blood flow and portal venous pressure has led to somatostatin analogs being used to treat esophageal variceal bleeding (see Chapter 92).119 The inhibitory effects on secretion have been exploited by using somatostatin analogs to treat some forms of diarrhea and reduce fluid output from pancreatic fistulas. Many endocrine tumors express abundant somatostatin receptors, making it possible to use radiolabeled somatostatin analogs (e.g., octreotide) to image even small tumors throughout the body.
Motilin
Motilin is a 22–amino acid peptide produced by endocrine cells of the duodenal epithelium.120 It is not released by the stimulation of food but instead is secreted into the blood under fasting conditions in a periodic and recurrent pattern that is synchronized with the migrating motor complex (MMC). Elevations in blood motilin levels regulate the phase III contractions that initiate in the antroduodenal region and progress toward the distal gut.
Motilin binds to specific receptors on smooth muscle cells of the esophagus, stomach, and small and large intestines through which it exerts propulsive activity.121 Agonists to the motilin receptor (e.g., erythromycin) have pronounced effects on GI motility, which occasionally produces undesired side effects of abdominal cramping and diarrhea.122 However, motilin agonists may be useful to treat conditions of impaired gastric and intestinal motility and are being investigated for the treatment of constipation-predominant irritable bowel syndrome.123
Leptin
Leptin is a 167–amino acid protein that is secreted primarily from adipocytes. Blood leptin levels reflect total body fat stores.124 Small amounts of leptin are produced by the chief cells of the stomach and by the placenta and are present in breast milk. Its primary action appears to be to reduce food intake. Leptin is a member of the cytokine family of signaling molecules. Five different forms of leptin receptors have been reported.125 A short form of the receptor appears to transport leptin from the blood across the blood-brain barrier, where it has access to the hypothalamus. A long form of the leptin receptor is located in hypothalamic nuclei, where leptin binds and activates the Janis kinase signal transduction and translation system (JAK STAT).126
Peripheral administration of leptin reduces food intake. However, this effect is reduced as animals become obese. Interestingly, when injected into the CNS, obese animals respond normally to leptin and reduce food intake, suggesting that leptin “resistance” in obesity occurs at the level of the leptin receptor that transports leptin across the blood-brain barrier.127 Leptin's ability to reduce food intake occurs within the brain by decreasing NPY (a potent stimulant of food intake) and by increasing α-melanocyte-stimulating hormone (α-MSH), an inhibitor of food intake.128 Peripherally, leptin acts synergistically with CCK to reduce meal size.129 In obese rats lacking the leptin receptor, the synergistic effects of leptin plus CCK to reduce meal size are lost but could be restored with genetic reconstitution of the leptin receptor in the brain.130 One might expect loss of leptin-CCK synergy on meal size in those rare cases of human obesity caused by leptin receptor defects or even with leptin resistance.
Blood levels of leptin increase as obesity develops, and leptin appears to reflect total fat content.131 At the cellular level, large adipocytes produce more leptin than small adipocytes. Because of its effects on food intake, it was initially thought that exogenous leptin could be used therapeutically to treat obesity. However, only a very modest effect on weight loss has been demonstrated in clinical trials. Leptin deficiency has been reported as a cause of obesity in a few families, but this condition is extremely rare.132,133 Mutation of the leptin receptor has been described as a cause of obesity in at least one family.134
Ghrelin
Ghrelin is a 28–amino acid peptide produced by the stomach and is the natural ligand for the growth hormone secretagogue (GHS) receptor.135 When administered centrally or peripherally, ghrelin stimulates growth hormone secretion, increases food intake, and produces weight gain.136,137 Circulating ghrelin levels increase during periods of fasting or under conditions associated with negative energy balance, such as starvation or anorexia. In contrast, ghrelin levels are low after eating and in obesity. Ghrelin appears to play a central role in the neurohormonal regulation of food intake and energy homeostasis.
The gastric fundus is the most abundant source of ghrelin, although lower amounts of ghrelin are found in the intestine, pancreas, pituitary, kidney, and placenta. Ghrelin is produced by distinctive endocrine cells known as P/D1 cells138,139 that are of 2 types, open and closed. The open type is exposed to the lumen of the stomach, where it comes into contact with gastric contents, whereas the closed type lies in close proximity to the capillary network of the lamina propria.140 Both cell types secrete hormone into the bloodstream. Based on its structure, ghrelin is a member of the motilin family of peptides and, like motilin, ghrelin stimulates gastric contraction and enhances stomach emptying.
The observations that circulating ghrelin levels increase sharply before a meal and fall abruptly after a meal suggest that it serves as a signal for initiation of feeding. The inhibitory effects of food on plasma ghrelin levels can be reproduced by ingestion of glucose and appear to be unrelated to the physical effects of a meal on gastric distention. Circulating ghrelin levels are low in states of positive energy balance such as obesity and are inversely correlated with body mass index.141,142 Conversely, ghrelin levels are high in fasting, cachexia, and anorexia. Importantly, weight loss increases circulating ghrelin levels.143
Ghrelin released from the stomach acts on the vagus nerve to exert its effects on feeding. However, it is also active when delivered to the CNS and, in this location, ghrelin activates NPY- and agouti-related protein-producing neurons in the arcuate nucleus of the hypothalamus, which is involved in the regulation of feeding.137,144
Gastric bypass patients do not demonstrate the premeal increase in plasma ghrelin that is seen in normal individuals.145 This lack of ghrelin release may be one of the mechanisms contributing to the overall effectiveness of gastric bypass surgery for inducing weight loss.
Prader-Willi syndrome is a congenital obesity syndrome characterized by severe hyperphagia, growth hormone deficiency, and hypogonadism. Although common obesity is ordinarily associated with low ghrelin levels, patients with Prader-Willi syndrome have high circulating ghrelin levels that do not decline after a meal.146,147 The levels of ghrelin in this syndrome are similar to those that can stimulate appetite and increase food intake in individuals receiving infusions of exogenous ghrelin, suggesting that abnormal ghrelin secretion may be responsible for the hyperphagia in Prader-Willi syndrome.148
Neurotransmitters
Acetylcholine
Acetylcholine is synthesized in cholinergic neurons and is the principal regulator of GI motility and pancreatic secretion. Acetylcholine is stored in nerve terminals and released by nerve depolarization. Released acetylcholine binds to postsynaptic muscarinic and/or nicotinic receptors. Nicotinic acetylcholine receptors belong to a family of ligand-gated ion channels and are homopentamers or heteropentamers composed of α, β, γ, δ, and ε subunits.149 The α subunit is believed to be the mediator of postsynaptic membrane depolarization following acetylcholine receptor binding. Muscarinic receptors belong to the heptahelical GPCR family. There are 5 known muscarinic cholinergic receptors (M1 to M5). Muscarinic receptors can be further classified based on receptor signal transduction, with M1, M3, and M5 stimulating adenylate cyclase and M2 and M4 inhibiting this enzyme. Acetylcholine is degraded by the enzyme acetylcholinesterase, and the products may be recycled through high-affinity transporters on the nerve terminal.
Catecholamines
The primary catecholamine neurotransmitters of the enteric nervous system include norepinephrine and dopamine. Norepinephrine is synthesized from tyrosine and released from postganglionic sympathetic nerve terminals that innervate enteric ganglia and blood vessels. Tyrosine is converted to dopa by tyrosine hydroxylase. Dopa is initially converted into dopamine by dopa decarboxylase and packaged into secretory granules. Norepinephrine is formed from dopamine by the action of dopamine β-hydroxylase in the secretory granule. After an appropriate stimulus, norepinephrine-containing secretory granules are released from nerve terminals and bind to adrenergic receptors.
Adrenergic receptors are G protein–coupled, have 7 typical membrane-spanning domains, and are of 2 basic types, α and β. α-Adrenergic receptors are further classified into α1A, α1B, α2A, α2B, α2C, and α2D. Similarly, β receptors include β1, β2, and β3. Adrenergic receptors are known to signal through various G proteins, resulting in stimulation or inhibition of adenylate cyclase and other effector systems. Norepinephrine signaling is terminated by intracellular monoamine oxidase or by rapid reuptake by an amine transporter. The actions of adrenergic receptor stimulation regulate smooth muscle contraction, intestinal blood flow, and GI secretion.
Dopamine
Dopamine is an important mediator of GI secretion, absorption, and motility and is the predominant catecholamine neurotransmitter of the central and peripheral nervous systems. In the CNS, dopamine regulates food intake, emotions, and endocrine responses. Peripherally, it controls hormone secretion, vascular tone, and GI motility. Characterization of dopamine in the GI tract has been challenging for several reasons. First, dopamine can produce inhibitory and excitatory effects on GI motility.150 Generally the excitatory response, which is mediated by presynaptic receptors, occurs at a lower agonist concentration than the inhibitory effect, which is mediated by postsynaptic receptors. Second, localization of dopamine receptors has been hampered by identification of dopamine receptors in locations that appear to be species specific.151 Third, studies of dopamine in GI tract motility have often used pharmacologic amounts of this agonist. Therefore, the interpretation of results has been confounded by the ability of dopamine to activate adrenergic receptors at high doses.
Classically, dopamine was thought to act via 2 distinct receptor subtypes, type 1 and type 2. Molecular cloning has now demonstrated 5 dopamine receptor subtypes, each with a unique molecular structure and gene locus.151 Dopamine receptors are integral membrane GPCRs, and each receptor subtype has a specific pharmacologic profile when exposed to agonists and antagonists. After release from the nerve terminal, dopamine is cleared from the synaptic cleft by a specific dopamine transporter.
Serotonin
Serotonin (5-hydroxytryptamine [5-HT]) has long been known to play a role in GI neurotransmission.152 The GI tract contains more than 95% of the total body serotonin, and serotonin is important in various processes, including epithelial secretion, bowel motility, nausea, and emesis.153 Serotonin is synthesized from tryptophan, an essential amino acid, and is converted to its active form in nerve terminals. Secreted serotonin is inactivated in the synaptic cleft by reuptake via a serotonin-specific transporter. Most plasma serotonin is derived from the gut, where it is found in mucosal enterochromaffin cells and the enteric nervous system. Serotonin mediates its effects by binding to a specific receptor. There are 7 different serotonin receptor subtypes found on enteric neurons, enterochromaffin cells, and GI smooth muscle (5-HT1 to 5-HT7).
The actions of serotonin are complex (Fig. 4-7).154 It can cause smooth muscle contraction through stimulation of cholinergic nerves or relaxation by stimulating inhibitory NO-containing neurons.153 Serotonin released from mucosal cells stimulates sensory neurons, initiating a peristaltic reflex and secretion (via 5-HT4 receptors) and modulates sensation through activation of 5-HT3 receptors.152 The myenteric plexus contains serotoninergic interneurons that project to the submucosal plexus and ganglia extrinsic to the bowel wall. Extrinsic neurons activated by serotonin participate in bowel sensation and may be responsible for abdominal pain, nausea, and symptoms associated with irritable bowel syndrome. Intrinsic neurons activated by serotonin are primary components of the peristaltic and secretory reflexes responsible for normal GI function. Serotonin may also activate vagal afferent pathways and, in the CNS, modulates appetite, mood, and sexual function. Because of these diverse effects, it is not surprising that selective serotonin reuptake inhibitor drugs (SSRIs), commonly used to treat depression and anxiety, have prominent GI side effects when compared with placebo treatment.
Serotonin and its receptor have been implicated in the pathogenesis of motility disorders of the GI tract.155 Characterization of specific serotonin receptor subtypes has led to the development of selective agonists and antagonists for the treatment of irritable bowel syndrome and chronic constipation and diarrhea. For example, 5-HT3 receptor antagonists, which reduce intestinal secretion, are used to treat diarrhea-predominant irritable bowel syndrome. 5-HT4 receptor agonists elicit prokinetic effects and are used to treat constipation-predominant irritable bowel syndrome and other motility disorders.156,157
Serotonin can also be enzymatically converted to melatonin by serotonin N-acetyltransferase.158 Other than the pineal gland, the GI tract is the major source of the body's melatonin. Melatonin is produced in enterochromaffin cells and released into the blood after ingestion of a meal. A number of actions on the GI tract have been described for melatonin, including reducing gastric acid and pepsin secretion, inducing smooth muscle relaxation, and preventing epithelial injury through an antioxidant effect.159 It has been proposed that melatonin released after a meal may contribute to postprandial somnolence.160
Histamine
In the GI tract, histamine is best known for its central role in regulating gastric acid secretion (see Chapter 50) and intestinal motility. Histamine is produced by enterochromaffin-like cells of the stomach and intestine as well as enteric nerves. Histamine is synthesized from L-histidine by histidine decarboxylase and activates 3 GPCR subtypes. H1 receptors are found on smooth muscle and vascular endothelial cells and are linked to phospholipase C (PLC) activation. As such, the H1 receptor mediates many of the allergic responses induced by histamine. H2 receptors are present on gastric parietal cells, smooth muscle, and cardiac myocytes. H2 receptor binding stimulates Gs (G proteins that stimulate adenylate cyclase) and activates adenylate cyclase. H3 receptors are present in the CNS and GI tract enterochromaffin cells. These receptors signal through Gi and inhibit adenylate cyclase.161 Histamine can also interact with the N-methyl-D-aspartate (NMDA) receptor and enhance activity of NMDA-bearing neurons independently of the 3 known histamine receptor subtypes.
Unlike other neurotransmitters, there is no known transporter responsible for termination of histamine's action. However, histamine is metabolized to telemethylhistamine by histamine N-methyltransferase and is then degraded to telemethylimidazoleacetic acid by monoamine oxidase B and an aldehyde dehydrogenase.
Nitric Oxide
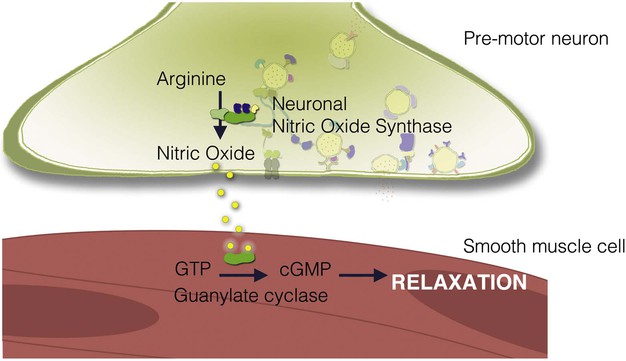
NO is a unique chemical messenger produced from L-arginine by the enzyme nitric oxide synthase (NOS).162 Three types of NOS are known. Types I and III are also known as endothelial NOS and neuronal NOS, respectively, and are constitutively active. Small changes in NOS activity can occur through elevations in intracellular calcium. The inducible form of NOS (type II) is apparent only when cells become activated by specific inflammatory cytokines. This form of NOS is capable of producing large amounts of NO and is calcium independent. NOS is often co-localized with VIP and PACAP in neurons of the enteric nervous system.163
NO, being an unstable gas, has a relatively short half-life. Unlike most neurotransmitters and hormones, NO does not act via a membrane-bound receptor. Instead, NO readily diffuses into adjacent cells to activate guanylate cyclase directly (Fig. 4-8). NO activity is terminated by its oxidation to nitrate and nitrite. Many enteric nerves use NO to signal neighboring cells and induce epithelial secretion, vasodilation, or muscle relaxation. NO is also produced by macrophages and neutrophils to help kill invading organisms.164
Cannabinoids and Other Chemical Transmitters
Cannabinoids
There are 3 categories of cannabinoids: synthetic, phytocannabinoids found in plants, and endocannabinoids. Endocannabinoids in particular have similar functions to neurotransmitters, in that they participate in synaptic transmission.165 In contrast to typical neurotransmitters, however, the flow of endocannabinoid signaling is retrograde to conventional neurotransmitters.166 Because of their liphophilic nature, endocannibinoids are membrane-bound molecules thought to be enriched in postsynaptic neurons. Thus, when released, endoccanabinoids move from postsynapses to act on presynaptic cannabinoid receptors and depress presynaptic function.167 In this manner, endocannabinoid signaling helps postsynaptic neurons regulate the secretion of transmitters from the sensory cell.
There are several types of endocannabinoid ligands, including arachidonoylethanolamine (anandamide), 2-arachidonoyl (2-AG), 2-arachidonyl glyceryl ether (Noladin ether), N-arachidonoyl-dopamine (NADA), virodhamine (OAE), and lysophosphatidylinositol. Endocanabinoids, as well as other cannabinoids, modulate metabolism and behavior by acting on the GPCR cannabinoid receptors CB1 and CB2. Both receptors are distributed throughout the body, although CB1 is primarily found in neurons and epithelial chemosensory cells, and CB2 is mainly present in cells of the immune system.
In the GI tract, CB1 receptors are also involved in counteracting proinflammatory responses and preventing the development of colitis.168 In addition to activating classical cannabinoid receptors, endocannabinoids can also stimulate GPCRs such as GPR119. Importantly, GPR119 is a receptor found in enteroendocrine cells of the small intestine, and its activation by endocannabinoids triggers the release of satiety-inducing hormones like CCK and peptide YY.168 These findings have made the field of GI endoccanabinoid research an active area for the development of therapeutic treatments.
Adenosine
Adenosine is an endogenous nucleoside that acts through any of 4 GPCR subtypes.169 Adenosine causes relaxation of intestinal smooth muscle and stimulates intestinal secretion. Adenosine can also cause peripheral vasodilation and activation of nociceptors that participate in neural pain pathways.
Cytokines
Cytokines are a group of polypeptides produced by various immunomodulatory cells and are involved in cell proliferation, immunity, and inflammation. Cytokines are induced by specific stimuli, such as toxins produced by pathogens, and often elicit a complex response involving other cellular mediators to eradicate the foreign substance. Cytokines may be categorized as interleukins (ILs), TNFs, lymphotoxins, interferons (IFNs), colony-stimulating factors (CSFs), and others.170 Interleukins can be further subtyped into at least 35 separate substances, IL-1 to IL-35. There are 2 TNFs, TNF-α and TNF-β (also known as lymphotoxin-α). IFNs are produced during viral or bacterial infection and come in 2 varieties, IFN-α (also known as leukocyte-derived interferon or interferon-β) and IFN-γ. IFN-α is produced by T lymphocytes and is used clinically for the treatment of viral hepatitis (see Chapters 79 and 80). The major CSFs are granulocyte mononuclear phagocyte CSF, mononuclear phagocyte CSF, and granulocyte CSF. These agents are used for chemotherapy-induced neutropenia and marrow support after bone marrow transplantation. Chemokines initiate and propagate inflammation and are of 2 groups, CXC (α chemokines) and CC (β chemokines). Other cytokines, such as transforming growth factor (TGF)-β and platelet-derived growth factor (PDGF), have proliferative effects.
The Importance of Hormones and Neurotransmitters
Growth and Abnormal Growth of the Gastrointestinal Mucosa
Growth of GI tissues is a balance between cellular proliferation and senescence. Many factors participate in maintenance of the GI mucosa. Nutrients and other luminal factors stimulate growth of the intestinal mucosa and are necessary to maintain normal digestive and absorptive functions. Hormones and transmitters serve as secondary messengers that are normally secreted in response to food ingestion and mediate many of the nutrient effects on the GI tract. They play a key role in cellular proliferation. Alterations in intestinal proliferation are manifested by atrophy, hyperplasia, dysplasia, or malignancy (see Chapter 1).
These responses are mediated by specific growth factors. There are several growth factors that have important effects on the GI tract, including peptides of the EGF, TGF-β, IGF, FGF, and PDGF families, hepatocyte growth factors, trefoil factors, and many cytokines (including ILs).171 Below are outlined important properties of some of these receptors.
Growth Factor Receptors
Growth factors regulate cellular proliferation by interacting with specific cell surface receptors. These receptors are membrane proteins that possess specific binding sites for the growth factor ligand. An unusual form of signaling occurs when the ligand interacts with its receptor within the same cell. For example, PDGF receptors present on the intracellular surface of fibroblast cell lines are activated by intracellular ligand. This process is known as intracrine signaling. Most peptide growth factors, however, interact with receptors on different cells to regulate proliferation.
Growth factor receptors can be single polypeptide chains containing 1 membrane-spanning region, such as the receptor for EGF, or they may be composed 2 subunit heterodimers, with 1 subunit containing a transmembrane domain and the other residing intracellularly but covalently bound to the transmembrane subunit. Heterodimers may also dimerize to form a receptor composed of 4 subunits (e.g., IGF receptor). Binding of the ligand to its receptor usually causes aggregation of 2 or more receptors and activation of intrinsic tyrosine kinase activity. Growth factor receptors also have the ability to autophosphorylate when bound to ligand. In addition, receptor tyrosine kinase activity may phosphorylate other intracellular proteins important in signal transduction. Autophosphorylation attenuates the receptor's kinase activity and often leads to down-regulation and internalization of the receptor. Mutation of the receptor at its autophosphorylation site may lead to constitutive receptor activity and cellular transformation. Growth factor receptors may couple to various intracellular signaling pathways, including adenylate cyclase, phospholipase C, calcium-calmodulin protein kinases, MAP kinase, and nuclear transcription factors. Thus, growth factors play important and varied roles in most cells of the GI tract. It is not surprising, therefore, that mutations in growth factor receptors or downstream signaling proteins can lead to unregulated cell growth and neoplasia (see Chapter 1).
An important action of growth factors is their ability to modulate the expression of transacting transcription factors that can regulate expression of many other genes.172 Early response genes such as jun and fos are activated rapidly after ligand binding and control the expression of many other genes involved in cellular proliferation. Other important transcriptional factors include c-myc and nuclear factor (NF)-κB. The latter is found in the cytoplasm in an inactive form and, following ligand binding, translocates to the nucleus, where it activates other transcription factors. NF-κB is a key target for strategies to regulate cellular proliferation and inflammation. In its phosphorylated form Rb-1, originally identified in retinoblastoma, is an inhibitor of cellular proliferation that complexes with the transcription factor p53. Dephosphorylation of Rb-1 releases p53, which activates other genes leading to cellular proliferation.
Almost all growth factors of the GI tract exert paracrine effects. However, many growth factors also possess autocrine and even intracrine actions. It has become apparent that growth factors and other signaling molecules secreted into the lumen of the gut can have important local biological actions. Distant effects of growth factors found in the circulation may be important for growth of certain types of cancers, particularly lung and colon cancer.
Epidermal Growth Factor
EGF was the first growth factor to be discovered. It is the prototype for a family of growth factors that are structurally related and have similarly related receptors. Other members of the family include TGF-α, amphiregulin, and heparin-binding EGF. EGF is identical to urogastrone (originally isolated from urine), which was shown to inhibit gastric acid secretion and promote healing of gastric ulcers. EGF is secreted from submaxillary glands and Brunner's glands of the duodenum. It is likely that EGF interacts with luminal cells of the GI tract to regulate proliferation. EGF has important trophic effects on gastric mucosa, and the wide distribution of EGF receptors suggests that EGF has mitogenic actions on various cells throughout the gut. The EGF receptor has been reported to be responsible for gastric hyperplasia in patients with Ménétrier's disease.173 Moreover, 2 patients were effectively treated with a monoclonal antibody that blocks ligand binding to the EGF receptor.174 EGF receptors are important targets for the treatment of human cancer, including metastatic colorectal cancer.175
Transforming Growth Factor-α
TGF-α is produced by most epithelial cells of the GI tract and acts through the EGF receptor. Therefore, it shares trophic properties with EGF. It is believed to play a key role in gastric reconstitution after mucosal injury. Moreover, this peptide appears to be important in GI neoplasia, because most gastric and colon cancers produce TGF-α (see Chapters 54 and 127).
Transforming Growth Factor-β
A family of TGF-β peptides exerts various biological actions, including stimulation of proliferation, differentiation, embryonic development, and formation of extracellular matrix.176 In contrast with the TGF-α receptor, there are 3 distinct TGF-β receptors.177 TGF-β modulates cell growth and proliferation in nearly all cell types and can enhance its own production from cells. It is likely that TGF-β plays a critical role in inflammation and tissue repair. TGF-β augments collagen production by recruitment of fibroblasts through its chemoattractant properties. This action can have beneficial or deleterious effects, depending on its site of deposition and abundance. For example, TGF-β may play a key role in the development of adhesions following surgery.178
Insulin-like Growth Factors
Alternative splicing of the insulin gene produces 2 structurally related peptides, insulin-like growth factor (IGF) I and IGF II.179 IGFs signal through at least 3 different IGF receptors. The IGF I receptor is a tyrosine kinase, and the IGF II receptor is identical to the mannose 6-phosphate receptor. Although the exact function of IGFs in the GI tract is not clearly understood, they have potent mitogenic activity in intestinal epithelium. IGF II appears to be critical for embryonic development.
Fibroblast Growth Factor and Platelet-Derived Growth Factor
At least 7 related fibroblast growth factors (FGFs) have been identified.180 These peptides have mitogenic effects on various cell types, including mesenchymal cells, and likely play an important role in organogenesis and neovascularization.181 Although not unique to the GI tract, PDGF is one of the most thoroughly studied growth factors. It is important for fibroblast growth, and its receptor is expressed in the liver and throughout the GI tract, where it appears to promote wound healing.
Trefoil Factors
Trefoil factors (pS2, spasmolysin, and intestinal trefoil factor, which are also known as TTF1, 2, and 3, respectively) are a family of proteins expressed throughout the GI tract.182 They share a common structure, having 6 cysteine residues and 3 disulfide bonds, creating a cloverleaf appearance that stabilizes the peptide within the gut lumen. The pS2 peptide is produced in the gastric mucosa, spasmolysin is found in the antrum and pancreas, and intestinal trefoil factor is produced throughout the small and large intestines. These peptides are produced by mucous neck cells in the stomach or goblet cells in the intestine and are secreted onto the mucosal surface of the gut. It is likely that trefoil factors act on the apical surface of the epithelial cells, where they have growth-promoting properties on the GI mucosa.
Other peptides signaling through GPCRs may also have growth-promoting effects. Three important examples include gastrin, CCK, and gastrin-releasing peptide (GRP). Gastrin stimulates the growth of enterochromaffin-like cells of the stomach and induces proliferation of the oxyntic mucosa containing parietal cells.183 Gastrin binds to CCK-2 receptors of the stomach and activates PLC and Ras pathways, which ultimately results in activation of protein kinase C and MAP kinase, respectively. MAP kinase, which can also be activated by tyrosine kinase receptors typical of growth factors, causes the phosphorylation of transcription factors that are involved in cellular proliferation. In some cells, cAMP and protein kinase A exert synergistic effects on cellular growth through activation of nuclear transcription factors such as cAMP-responsive element binding (protein) (CREB). However, in other cells, cAMP antagonizes proliferation. Therefore, depending on the cell type, the effects of growth factors such as EGF, IGF, and PDGF may be enhanced by hormones that stimulate cAMP production. Certain colon cancer cells possess CCK-2 receptors and respond to the proliferative effects of gastrin. Moreover, gastrin may be produced by some colon cancers, enabling it to exert an autocrine effect to promote cancer growth.184 Whether circulating gastrin initiates colon cancer development is unknown.
Diabetes Mellitus and the Gastrointestinal Tract
GI hormones play an important role in the regulation of insulin secretion and glucose homeostasis. These hormones control processes that facilitate the digestion and absorption of nutrients, as well as disposal of nutrients that have reached the bloodstream. In particular, gut peptides control postprandial glucose levels through 3 different mechanisms: (1) stimulation of insulin secretion from pancreatic beta cells, (2) inhibition of hepatic gluconeogenesis by suppression of glucagon secretion, and (3) delaying the delivery of carbohydrates to the small intestine by inhibiting gastric emptying.185 Each of these actions reduces the blood glucose excursions that normally occur after eating.
Approximately 50% of the insulin released after a meal is the result of GI hormones that potentiate insulin secretion.186 This interaction is known as the enteroinsular axis, and the gut peptides that stimulate insulin release are known as incretins. The major incretins are GLP-1 and GIP. GLP-1 not only stimulates insulin secretion but also increases beta cell mass, inhibits glucagon secretion, and delays gastric emptying. GIP stimulates insulin secretion when glucose levels are elevated and decreases glucagon-stimulated hepatic glucose production.187 On ingestion of a meal, glucose, as it is absorbed, stimulates GLP-1 and GIP secretion. Circulating glucose then stimulates beta cell production of insulin, and this effect is substantially augmented by incretins acting in conjunction with glucose to increase insulin levels.
Postprandial hyperglycemia may also be controlled by delaying the delivery of food from the stomach to the small intestine, allowing the rise in insulin to keep pace with the rate of glucose absorption. Several gut hormones that delay gastric emptying have been shown to reduce postprandial glucose excursions (Box 4-2).185 Amylin (islet amyloid polypeptide) is a 37–amino acid peptide synthesized primarily in the beta cells of the pancreatic islets together with insulin. Although it was originally recognized for its ability to form amyloid deposits in association with beta cell loss, it has more recently been found to suppress glucagon secretion, delay gastric emptying, and induce satiety.188 Insulin resistance in obese patients is associated with increased levels of both insulin and amylin.
Type 2 diabetes mellitus is characterized by high circulating insulin levels and insulin resistance. In addition, insulin levels do not increase appropriately after a meal, and significant hyperglycemia occurs, which is consistent with an impaired incretin effect. GIP secretion is preserved in type 2 diabetes, but the insulinotropic effect of GIP is reduced.189 Although the precise cause is unknown, the defect in GIP-stimulated insulin release is most pronounced in the late phase of insulin secretion. In contrast to GIP, GLP-1 secretion is reduced in insulin-resistant type 2 diabetics. The lower GLP-1 levels are caused by impaired secretion rather than increased degradation of the hormone.190 Unlike GIP, the insulin response to infusion of GLP-1 is preserved, indicating that the beta cell can respond normally to this incretin hormone. These observations suggest that GLP-1 administration could be a viable treatment for the hyperglycemia associated with diabetes.191 The growing evidence that beta cell failure may develop in type 2 diabetes supports the use of incretin hormones, such as GLP-1, or agents that delay GLP-1 degradation by the enzyme dipeptidyl peptidase-4 (DPP-4) to enhance beta cell function.192,193 Several incretin analogs are now used clinically for the treatment of diabetes.194
Gastrointestinal Regulation of Appetite
During a meal, ingested nutrients interact with cells of the mouth and GI tract. Endocrine cells of the stomach and small intestine possess receptors that are linked to the secretion of GI hormones. GI peptides are then released into the surrounding space, where they exert paracrine actions or are taken up into the circulation, where they function as hormones.195 Each of these transmitters facilitates the ingestion, digestion, absorption, or distribution of nutrients that are essential for the organism. Some GI hormones control the size of an ingested meal and are known as satiety signals. Satiety hormones share several qualities.196 First, they decrease meal size. Second, blocking their endogenous activity leads to increased meal size. Third, reduction of food intake is not the result of an aversion to food. Fourth, secretion of the hormone is caused by ingestion of food that normally causes cessation of eating (Table 4-2). Most satiety signals interact with specific receptors on nerves leading from the GI tract to the hindbrain.
TABLE 4-2
GI Peptides That Regulate Satiety and Food Intake
| Reduce Food Intake | Increase Food Intake |
| Cholecystokinin (CCK) | Ghrelin |
| Glucagon-like peptide-1 | |
| Peptide tyrosine tyrosine (PYY3-36) | |
| Gastrin-releasing peptide | |
| Amylin | |
| Apolipoprotein A-IV | |
| Somatostatin |
CCK is one of the most extensively studied satiety hormones. In a time- and dose-dependent manner, CCK reduces food intake in animals and humans,197 an effect that is mediated by CCK-1 receptors residing on vagus nerve endings.198 The effect of CCK on food intake is a proved physiologic action, because administration of a CCK receptor antagonist induces hunger and results in larger meal sizes. CCK also delays the rate at which food empties from the stomach, which may explain why the satiety actions of CCK are most apparent when the stomach is distended. Together, these findings indicate that CCK provides a signal for terminating a meal.
GLP-1 is produced by enteroendocrine cells in the ileum and colon and is released in response to food in the intestine. Although the primary action of GLP-1 is to stimulate insulin secretion, it also delays gastric emptying. Moreover, infusion of GLP-1 increases satiety and produces feelings of fullness, thereby reducing food intake without causing aversion.199 GLP-1 receptors are found in the periventricular nucleus, dorsal medial nucleus, and arcuate nucleus of the hypothalamus, which are important areas in the regulation of hunger. Like CCK, central administration of GLP-1 suppresses food intake.
PYY is also produced by enteroendocrine cells of the ileum and colon. Two forms of PYY are released into the circulation, PYY1-36 and PYY3-36. PYY1-36 binds to all subtypes of the neuropeptide Y family of receptors, whereas PYY3-36 has strong affinity for the Y2 receptor. When administered to animals, PYY3-36 causes a reduction in food intake, and mice lacking the Y2 receptor are resistant to the anorexigenic effects of PYY3-36, indicating that PYY3-36 signals satiety through this receptor.200 PYY3-36 has been shown in humans to decrease hunger scores and caloric intake.201 Interestingly, most of the GI peptide receptors involved in satiety are also found in the brain, where they mediate similar satiety effects. This may represent conservation of peptide signals that serve similar purposes.
Leptin is referred to as an adiposity signal because it is released into the blood in proportion to the amount of body fat and is considered a long-term regulator of energy balance. Together with CCK, leptin reduces food intake and produces a greater reduction in body weight than either agent alone.129 Therefore, it appears that long-term regulators of energy balance can affect short-term regulators through a decrease in meal size, which may promote weight reduction.
Hunger and initiation of a meal are intimately related. Ghrelin is intriguing because it is the only known circulating GI hormone that has orexigenic effects.145 Produced by the stomach, ghrelin levels increase abruptly before the onset of a meal and decrease rapidly after eating, suggesting that it signals initiation of a meal. Consistent with this role are studies demonstrating that administration of antighrelin antibodies or a ghrelin receptor antagonist suppresses food intake.202 It is not known if ghrelin is responsible for the hunger pains and audible bowel sounds that occur in people who are hungry.
Bariatric surgery, in particular Roux-en-Y gastric bypass, is the most effective procedure for long-term weight loss in morbid obesity. Although it had been assumed that weight loss accompanying this procedure was the result of reduced gastric capacity and calorie malabsorption, recent evidence of reduced ghrelin release and exaggerated PYY release after a meal has suggested that hormonal factors may contribute to reduced calorie intake.203