Ascites and Spontaneous Bacterial Peritonitis
Bruce A. Runyon
Ascites is of Greek derivation (“askos”) and refers to a bag or sack. The word is a noun and describes pathologic fluid accumulation within the peritoneal cavity. The adjective ascitic is used in conjunction with the word fluid to describe the liquid per se. Therefore, “ascitic fluid” is preferred to “ascites fluid.”
Pathogenesis of Ascites
Cirrhotic Ascites
Ascites occurs in the setting of cirrhosis as a result of the sequence of events detailed in Figure 93-1. The most recent theory of ascitic fluid formation, the “peripheral arterial vasodilation hypothesis,” proposes that both older hypotheses, the underfill and overflow theories, are correct, but that each is operative at a different stage.1 The first abnormality that develops appears to be portal hypertension. Portal pressure increases above a critical threshold, and circulating nitric oxide levels increase. Nitric oxide leads to vasodilatation. As the state of vasodilatation worsens, plasma levels of vasoconstrictor sodium-retentive hormones increase, renal function deteriorates, and ascitic fluid forms—that is, hepatic decompensation occurs.
In the setting of volume overload in a patient with cirrhosis and ascites, the explanation for the neurohumoral excitation, which is characteristic of volume depletion, may relate to volume sensors. Animals have sophisticated systems for detecting and preserving vascular perfusion pressures and intravascular osmolality. An organism's ability to detect changes in intravascular volume (especially volume overload) is limited, however, and is linked to pressure receptors. This observation may explain, in part, the paradox of dramatic volume overload in the face of sympathetic nervous traffic and hormone levels that are indicative of intravascular volume depletion.
Noncirrhotic Ascites
The mechanism of fluid retention in patients with malignancy-related ascites depends on the location of the tumor. Peritoneal carcinomatosis appears to cause ascites through the production of proteinaceous fluid by tumor cells lining the peritoneum. Extracellular fluid enters the peritoneal cavity to reestablish oncotic balance. Fluid accumulates in patients with massive liver metastases because of portal hypertension caused by stenosis or occlusion of portal veins by tumor nodules or tumor emboli.2 In patients with hepatocellular carcinoma, ascites arises because of the underlying cirrhosis-related portal hypertension, tumor-induced portal vein thrombosis, or both. Chylous ascites in patients with malignant lymphoma appears to be caused by lymph node obstruction by tumor and rupture of chyle-containing lymphatics.
Ascites can complicate high-output or low-output heart failure or nephrotic syndrome. As in cirrhosis, effective arterial blood volume appears to be decreased, and the vasopressin, renin-aldosterone, and sympathetic nervous systems are activated.3 These changes lead to renal vasoconstriction and sodium and water retention. Fluid then “weeps” from the congested hepatic sinusoids as lymph, as in cirrhosis. TB, Chlamydia infection, and coccidioidomycosis probably cause ascites through the production of proteinaceous fluid, as in peritoneal carcinomatosis. SBP does not appear to cause fluid to accumulate; infection develops only in preexisting ascites.
In patients with pancreatic or biliary ascites, fluid accumulates by leakage of pancreatic juice or bile into the peritoneal cavity or forms secondary to a “chemical burn” of the peritoneum. After abdominal surgery, especially extensive retroperitoneal dissection, lymphatics may be transected and may leak lymph for varying amounts of time. The mechanism of development of ascites in this condition is similar to that for malignant chylous ascites, namely, lymphatic leak.
Clinical Features
History
Most patients (≈85%) with ascites in the United States have cirrhosis. The 3 most common causes of cirrhosis are excess alcohol use, chronic hepatitis C, and nonalcoholic steatohepatitis (NASH) related in many cases to obesity. As the obesity epidemic evolves, NASH could become the most common cause of cirrhosis. Many patients have 2 of these conditions, and some have all 3.4 In approximately 15% of patients with ascites, a nonhepatic cause of fluid retention is identified (Table 93-1).
TABLE 93-1
Causes of Ascites
| Cause | % |
| Cirrhosis (with or without peritoneal infection) | 85 |
| Miscellaneous portal hypertension-related disorder (including 5% with two causes) | 8 |
| Cardiac disease | 3 |
| Peritoneal carcinomatosis | 2 |
| Miscellaneous nonportal hypertension–related disorders | 2 |
Ascites frequently develops during a patient's first episode of decompensation of alcoholic liver disease. Ascites can develop early in alcoholic liver disease in the precirrhotic alcoholic hepatitis stage. At this stage, portal hypertension and the resulting predisposition to sodium retention are reversible with abstinence from alcohol. Patients with precirrhotic alcoholic liver disease may lose their predisposition to fluid retention when they reduce or cease consumption of alcohol.
Evidence is accumulating that cirrhosis unrelated to alcohol use can also be reversible with effective therapy.5 Whether a decompensated cirrhotic liver can revert to a normal liver, however, remains to be seen. Many patients with cirrhosis and ascites will ultimately require liver transplantation.
Patients with ascites should be questioned about risk factors for liver disease other than alcohol, such as injection drug use, blood transfusions prior to 1992, sex with a same-gender partner, acupuncture, tattoos, ear piercing, and country of origin. Commonly, the cause of ascites in a middle-aged or elderly woman is viral hepatitis–induced cirrhosis resulting from a remote, often forgotten blood transfusion. Another cause of “cryptogenic” cirrhosis and ascites is NASH from long-standing obesity.6 Many patients who have been obese will spontaneously lose 50 or even 100 pounds after their liver disease decompensates. Unless the patient is questioned about lifetime maximum body weight and usual adult body weight, the possibility of NASH-related cirrhosis may not be considered. With careful history-taking and appropriate laboratory testing, the percentage of patients with cirrhosis who are now labeled cryptogenic is approaching zero (see Chapter 74).6
Patients with a long history of stable cirrhosis and the sudden development of ascites should be suspected of harboring a hepatocellular carcinoma that has precipitated the decompensation. Patients with ascites who have a history of cancer should be suspected of having malignancy-related ascites. Cancer in the past, however, does not guarantee a malignant cause of ascites. For example, patients with tobacco-related lung cancer and a history of alcohol abuse may have ascites due to cirrhosis. Breast, lung, colon, and pancreatic cancers are regularly complicated by ascites.2 Abdominal pain is a helpful distinguishing feature. Malignancy-related ascites frequently is painful, whereas cirrhotic ascites usually is not, unless bacterial peritonitis or alcoholic hepatitis is superimposed.
A history of heart failure may raise the possibility of cardiac ascites. Alcoholic patients in whom ascites develops may have alcoholic cardiomyopathy or alcoholic liver disease, but usually not both.
Tuberculous peritonitis usually manifests as fever and abdominal pain. Many affected patients are immigrants from an endemic area. In the United States, more than half of the patients with tuberculous peritonitis have underlying alcoholic cirrhosis that may contribute to the formation of ascitic fluid.
Ascites may occur in patients with acute pancreatitis with necrosis or a ruptured pancreatic duct from chronic pancreatitis or trauma. Often, troublesome ascites also may develop in a small percentage of patients undergoing hemodialysis. Fitz-Hugh–Curtis syndrome caused by chlamydia or gonorrhea may cause inflammatory ascites in a sexually active woman. Patients in whom ascites and anasarca develop in the setting of diabetes mellitus should be suspected of having nephrotic ascites. Ascites in a patient with symptoms and signs of myxedema should prompt assessment of thyroid function. Serositis in a patient with a connective tissue disease may be complicated by ascites.7
Physical Examination
On the basis of the history and the appearance of the abdomen, the diagnosis of ascites is readily suspected and usually confirmed easily on physical examination. The presence of a full, bulging abdomen should lead to percussion of the flanks. If the degree of flank dullness is greater than usual (i.e., if the percussed air-fluid level is higher than that normally found on the lateral aspect of the abdomen with the patient supine), the examiner should check for “shifting.” If flank dullness is absent, checking for shifting is unnecessary. Approximately 1500 mL of fluid must be present before dullness is detected.8 If flank dullness is not present, the chance that the patient has ascites is less than 10%.8 A fluid wave is not worth testing for.8
Gaseous distention of the bowel, a thick panniculus, and an ovarian mass can mimic ascites. Gaseous distention should be readily apparent on percussion. Ovarian masses usually cause tympanitic flanks with central dullness. The speed of increase in abdominal girth also can be helpful: ascites develops in days to weeks, whereas thickening of the omentum and panniculus takes months to years. An obese abdomen may be diffusely dull to percussion, and abdominal US may be required to determine if fluid is present. US can detect as little as 100 mL of fluid in the abdomen.
The presence of palmar erythema, large pulsatile spider telangiectasias, large abdominal wall collateral veins, or fetor hepaticus is suggestive of parenchymal liver disease and portal hypertension (see Chapter 74). The presence of large veins on the patient's back suggests inferior vena cava blockage. An immobile mass in the umbilicus, the Sister Mary Joseph nodule, is suggestive of peritoneal carcinomatosis.
The neck veins of patients with ascites should always be examined. Alcoholic cardiomyopathy with cardiac ascites can mimic cirrhosis with ascites; an elevated jugular venous pressure helps with this aspect of the differential diagnosis. Constrictive pericarditis is one of the few curable causes of ascites. Most patients with cardiac ascites have impressive jugular venous distention. Some have no visible jugular venous distention but such high central venous pressures that their bulging forehead veins rise to the top of their skulls. When present, peripheral edema in patients with liver disease is usually found in the lower extremities and occasionally may involve the abdominal wall. Patients with nephrotic syndrome or cardiac failure may have total body edema (anasarca).
Diagnosis
Although the diagnosis of ascites may be suspected on the basis of the history and physical examination, final confirmation is based on successful abdominal paracentesis or detection of ascites on imaging. Determination of the cause of ascites is based on the results of the history, physical examination, and ascitic fluid analysis. In general, few other tests are required.
Abdominal Paracentesis
Indications
Abdominal paracentesis9 with appropriate ascitic fluid analysis is probably the most rapid and cost-effective method of diagnosing the cause of ascites. Also, because of the possibility of ascitic fluid infection in a patient with cirrhosis admitted to the hospital, a surveillance paracentesis performed on admission may detect unexpected infection.10 Not all patients with ascitic fluid infection are symptomatic; many have subtle symptoms, such as mild confusion noticed only by the family. An emergency department study has demonstrated that a physician's impression regarding the presence or absence of ascitic fluid infection is inaccurate.11
Detection of infection at an early asymptomatic stage may reduce mortality. Therefore, ascitic fluid should be sampled in all inpatients and outpatients with new-onset ascites and in all patients with ascites who are admitted to the hospital. Paracentesis should be repeated in patients (whether hospitalized or not) in whom symptoms, signs, or laboratory abnormalities suggestive of infection develop (e.g., abdominal pain or tenderness, fever, encephalopathy, hypotension, renal failure, acidosis, peripheral leukocytosis).
Contraindications
Few contraindications to paracentesis have been recognized. Coagulopathy is a potential contraindication; however, most patients with cirrhosis and ascites have coagulopathy, and if mild to moderate coagulopathy were viewed as a contraindication to paracentesis, few patients with cirrhosis would undergo this procedure.12 Coagulopathy should preclude paracentesis only when clinically evident fibrinolysis or DIC is present.12 These conditions occur in fewer than 1 per 1000 paracenteses. No data are available to support cutoff values for coagulation parameters beyond which paracentesis should be avoided. Overall coagulation is usually normal in the setting of cirrhosis, despite abnormal tests of coagulation, because there is a balanced deficiency of procoagulants and anticoagulants.13 Even after multiple paracenteses, bloody ascites usually does not develop in patients with marked prolongation of the prothrombin time. Patients with cirrhosis and without clinically obvious coagulopathy simply do not bleed excessively from needlesticks unless a blood vessel is entered.12
Studies regarding complications of paracentesis in patients with ascites have documented no deaths or infections caused by paracentesis.10,12 No episodes of hemoperitoneum or entry of the paracentesis needle into the bowel have been reported in these studies. Complications have included only abdominal wall hematomas in approximately 2% of paracenteses, even though 71% of the patients had an abnormal prothrombin time and 21% had a prothrombin time prolonged by more than 5 seconds.12 Complication rates may be higher when paracentesis is performed by an inexperienced operator.
Transfusion of blood products (fresh frozen plasma or platelets) routinely before paracentesis in patients with cirrhosis and coagulopathy, presumably to prevent hemorrhagic complications, is not supported by data. Because a hematoma that necessitates blood transfusion develops in only approximately 1% of patients who undergo paracentesis without prophylactic transfusion of plasma or platelets, approximately 100 to 200 units of fresh frozen plasma or platelets would have to be given to prevent the transfusion of approximately 2 units of red blood cells (RBCs). In a prospective study of 1100 therapeutic paracenteses, no blood products were given prior to the procedure, nor were they needed after the procedure despite a platelet count as low as 19,000/mm3 [0.19 × 109/L]) and INR as high as 8.7.14
Patient Position and Choice of Needle and Entry Site
The volume of fluid in the abdomen and the thickness of the abdominal wall determine, in part, how the patient should be positioned in preparation for paracentesis. Patients with a large volume of ascites and thin abdominal wall can be “tapped” successfully in the supine position, with the head of the bed or examining table elevated slightly. Patients with less fluid can be placed in the lateral decubitus position and tapped in the midline or in the right or left lower quadrant while supine (see later). Patients with small amounts of fluid may be tapped successfully only in the face-down position or with US guidance.15
The choice of the site for inserting the needle has changed over the years because of the increasing prevalence of obesity and frequency of therapeutic paracentesis. Paracentesis in obese patients poses special challenges. In obese patients, the abdominal wall usually is substantially thicker in the midline than in the lower quadrants on US examination.15 The abdominal wall may be even thicker than the length of a 3.5-inch paracentesis needle. Also, on physical examination, determining whether ascites is present or absent in the obese patient is frequently difficult. US examination is helpful in confirming the presence of fluid and in guiding the paracentesis needle. Preferably, the needle is inserted into the left lower quadrant, rather than the right lower quadrant, because the cecum may be distended with gas from lactulose therapy (see Chapter 94). Also, the right lower quadrant is more likely than the left to have a surgical scar (e.g., from an appendectomy). When therapeutic paracentesis is performed, more fluid can be obtained using a lower quadrant needle insertion site than a midline site.
The needle must be placed several centimeters from a surgical scar. The bowel may be adherent to the peritoneal surface of the abdomen near a scar, and a needle inserted there may enter the bowel.10 A long midline scar precludes midline paracentesis. An appendectomy scar precludes a right lower quadrant site, in general.
I usually choose a site in the left lower quadrant 2 fingerbreadths (3 cm) cephalad and 2 fingerbreadths medial to the anterior superior iliac spine.15 In a patient with multiple abdominal scars, US guidance may be required.
In a patient who is not overweight, I prefer to use a standard metal 1.5-inch, 22-gauge needle. Paracentesis in obese patients requires the use of a longer needle—for example, one that is 3.5 inches and 22 gauge. Steel needles are preferable to plastic-sheathed cannulas because plastic sheaths may shear off into the peritoneal cavity, with the potential to kink and obstruct the flow of fluid after the cannula is removed. Metal needles do not puncture the bowel unless the bowel is adherent to a scar or severe gaseous distention is present.
Technique
The optimal technique of diagnostic and therapeutic paracentesis has been reviewed in detail.9
Diagnostic Paracentesis
Drapes, gown, hat, and mask are optional, but sterile gloves should be used when paracentesis is performed. The skin is disinfected with an iodine solution. The skin and subcutaneous tissue should be infiltrated with a local anesthetic. The sterile package insert enclosing the gloves can be used as a sterile field on which to place syringes, needles, gauze, and other supplies. When sterile gloves are not used, ascitic fluid cultures frequently grow skin contaminants; a single viable organism will grow to detectable levels in blood culture bottles.
To prevent leakage of fluid after the needle is withdrawn, a special technique is required. The previously used term “Z tract” led to confusion about the precise technique. It does not involve manipulating the needle up and down, which could lead to tissue injury. The technique of needle insertion is accomplished by displacing (with one gloved hand) the skin approximately 2 cm downward and then slowly inserting the paracentesis needle mounted on the syringe held in the other hand. The hand holding the syringe stabilizes the syringe and retracts its plunger simultaneously. A steady hand and experience are needed. The skin is released only after the needle has penetrated the peritoneum and fluid flows. When the needle is ultimately removed, the skin resumes its original position and seals the needle pathway. (If the needle were inserted straight into the peritoneum from the skin surface, the fluid would leak out easily because the pathway would be straight.)
The needle should be advanced slowly through the abdominal wall in approximately 5-mm increments. Slow insertion allows the operator to see blood if a vessel is entered, so that the needle can be withdrawn immediately before further damage is done. Slow insertion also allows the bowel to move away from the needle, thereby avoiding bowel puncture. The syringe that is attached to the needle should be aspirated intermittently during insertion. If continuous suction is applied, bowel or omentum may be drawn to the end of the needle as soon as the needle enters the peritoneal cavity, thereby occluding flow and resulting in an apparently unsuccessful tap. Slow insertion also allows time for the elastic peritoneum to “tent” over the end of the needle and be pierced by it. The most common causes of an unsuccessful paracentesis are continuous aspiration during insertion of the needle and rapid insertion and withdrawal of the needle before the peritoneum is pierced. If the operator is certain that the needle tip is inserted far enough but no fluid is apparent, the syringe and needle can be twisted 90 degrees to pierce the peritoneum, thereby permitting flow of fluid.
Approximately 30 mL of fluid is obtained using one or more syringes. I prefer to use a 5- or 10-mL syringe for the initial portion of a diagnostic tap and then twist this syringe off the needle and replace it with a 20- or 30-mL syringe to obtain the remainder of the sample. The initial use of a small syringe allows the operator to have better control and to see fluid more easily as it enters the hub of the syringe. The syringe and attached needle are then pulled out of the abdomen, and the needle is removed and discarded. A sterile needle is then placed on the larger syringe, and an appropriate amount of fluid is inoculated into each of a pair of prepared blood culture bottles (see later). Usually, 5 to 10 mL is inoculated into 50-mL bottles, and 10 to 20 mL into 100-mL bottles. The next aliquot is placed into a “purple-top” ethylenediaminetetraacetic acid (EDTA) tube for a cell count, and the final aliquot is placed into a “red-top” tube for chemistries. Inoculating the culture bottles first with a sterile needle minimizes contamination. The fluid must be placed promptly into the anticoagulant-containing tube to avoid clotting; clotted fluid cannot be analyzed for cell count.
Therapeutic Paracentesis
Therapeutic paracentesis is similar to diagnostic paracentesis except that a larger-bore needle is used and additional equipment is required. In the patient who is not overweight, I prefer to use a standard metal 1.5-inch, 16- to 18-gauge needle. Obese patients may require a longer needle (e.g., 3.5 inches and 18 gauge). A set of 15-gauge 5-hole needles has been produced specifically for therapeutic abdominal paracentesis; these needles may replace the spinal needles generally used for paracentesis in obese patients. The 15-gauge needles have a removable sharp inner component and a blunt outer cannula; they range in length from 3.25 to 5.9 inches. A tiny scalpel nick is required to permit the large needle to enter the skin.
An old method of using a 60-mL syringe, stopcock, and collection bag is tedious; use of vacuum bottles (1 or 2 L) connected to the needle with noncollapsible tubing is much faster. Use of a pump is even faster than vacuum bottles. Unless the needle is allowed to drift subcutaneously, the needle (or blunt steel cannula) can be left in the abdomen during a therapeutic paracentesis without injury. Larger-bore needles or cannulas permit more rapid removal of fluid but leave larger defects if they enter vessels or the bowel inadvertently.
Once fluid is flowing, the needle should be stabilized to ensure steady flow. Not unusually, flow ceases intermittently. With respiratory movement, the needle may gradually work its way out of the peritoneal cavity and into the soft tissue, and some serosanguineous fluid may appear in the needle hub or tubing. When this happens, the pump should be turned off or a clamp placed on the tubing connected to the vacuum bottle. The tubing should be removed from the needle and the needle twisted a few degrees. If flow does not resume, the needle should be twisted a bit more. If flow still does not resume, the needle should be inserted in 1- to 2-mm increments until brisk dripping of fluid from the needle hub is seen. The tubing can then be reattached and more fluid removed. Occasionally, fluid cannot be aspirated but drips from the needle hub. In this situation, fluid can be allowed to drip into a sterile container for collection, as in a lumbar puncture.
As the fluid is removed, the bowel and omentum draw closer to the needle and eventually block the flow of ascitic fluid. The patient must then be repositioned so that gravity causes the fluid to pool near the needle. It is useful to reposition the patient a few times during a total paracentesis to maximize the amount of fluid removed. Excessive manipulation of the needle should be avoided to minimize the risk of trauma to the bowel or blood vessels.
After samples of fluid are obtained for testing, a total of 2 to 4 L of fluid is removed to relieve the pressure of tense ascites in patients with new or diuretic-sensitive ascites. A sodium-restricted diet and diuretics are prescribed to reduce the fluid further. If a patient is known to be diuretic-resistant, a “total tap” is performed—that is, all of the fluid that is accessible is removed. If less is removed, the tap will need to be repeated soon (see later).
Ascitic Fluid Analysis
Gross Appearance
Non-neutrocytic (i.e., ascitic fluid polymorphonuclear neutrophil [PMN] count less than 250/mm3 [0.25 × 109/L]) ascitic fluid is transparent and usually slightly yellow (Fig. 93-2). Ascitic fluid with a very low protein concentration may have no pigment and look like water. The opacity of many cloudy ascitic fluid specimens is caused by neutrophils. The presence of neutrophils leads to a shimmering effect when a glass tube containing the fluid is rocked back and forth in front of a light. Fluid with an absolute neutrophil count less than 1000/mm3 (1.0 × 109/L) may be nearly clear. Fluid with a count greater than 5000/mm3 (5.0 × 109/L) is quite cloudy, and fluid with a count greater than 50,000/mm3 (50.0 × 109/L) resembles mayonnaise.
Ascitic fluid specimens frequently are blood-tinged or frankly bloody. An RBC count of 10,000/mm3 (10.0 × 109/L) is the threshold for a pink appearance; lower concentrations result in clear or turbid fluid. Ascitic fluid with an RBC count greater than 20,000/mm3 (20.0 × 109/L) is distinctly red. Many ascitic fluid specimens are bloody because of a traumatic tap; these specimens are blood-streaked and frequently clot unless the fluid is transferred immediately to the anticoagulant-containing tube for the cell count. By contrast, nontraumatic or remotely traumatic blood-tinged ascitic fluid is homogeneous and does not clot because it has already clotted and the clot has lysed. Some patients with portal hypertension have bloody hepatic lymph, resulting in bloody ascitic fluid—perhaps because of rupture of lymphatics that are under high pressure. Samples from patients with hepatocellular carcinoma are regularly bloody, but only about 10% of samples from patients with peritoneal carcinomatosis are red.2 Although many physicians have the impression that TB results in bloody ascitic fluid, less than 5% of tuberculous samples are hemorrhagic in my experience.
Ascitic fluid frequently is lipid-laden. Lipid opacifies the fluid. The degree of opalescence of ascitic fluid ranges from slightly cloudy to completely opaque and chylous. Most opaque, milky fluid samples have a TG concentration greater than 200 mg/dL (2.26 mmol/L) and usually greater than 1000 mg/dL (11.30 mmol/L). Fluid that has the appearance of dilute skim milk has a TG concentration between 100 mg/dL (1.13 mmol/L) and 200 mg/dL (2.26 mmol/L). A substantial minority of cirrhotic ascitic fluid samples are neither transparent nor frankly milky. These opalescent samples have slightly elevated TG concentrations ranging from 50 mg/dL (0.56 mmol/L) to 200 mg/dL (2.26 mmol/L).16 The opacity of these fluids does not have the shimmering characteristics of ascitic fluid with an elevated WBC count. The lipid usually layers out when a tube of ascitic fluid is placed in a refrigerator for 48 to 72 hours. In contrast to findings in older published reports, most patients with chylous or opalescent ascites have cirrhosis.16,17
Dark-brown fluid with a bilirubin concentration greater than that of serum usually indicates biliary perforation.18 Deeply jaundiced patients have bile-stained ascitic fluid, but the bilirubin level and the degree of pigmentation are visually less than those of the corresponding serum. Pancreatic ascites may be pigmented because of the effect of pancreatic enzymes on RBCs. The RBCs may have to be centrifuged before the discolored supernatant is revealed. The degree of pigmentation ranges from tea-colored to jet black, as in pancreatic necrosis. Black ascitic fluid also may be found in patients with malignant melanoma.
Tests
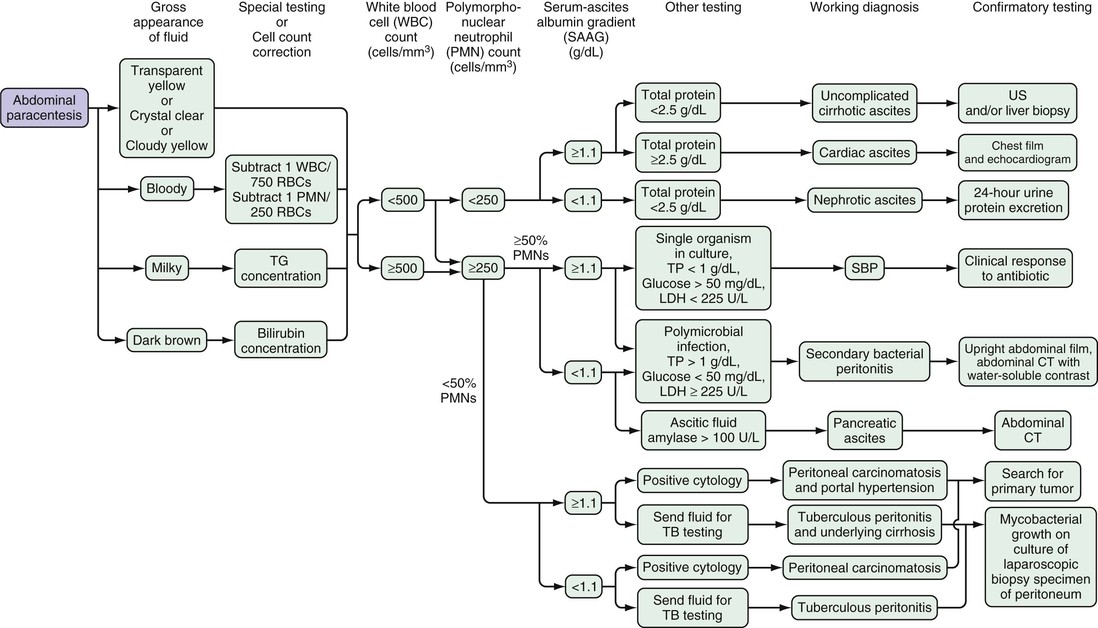
The practice of ordering every available body fluid test on every ascitic fluid specimen is expensive and can be more confusing than helpful, especially when unexpectedly abnormal results are encountered. An algorithm for the analysis of ascitic fluid is shown in Figure 93-2. The basic concept is that screening tests are performed on the initial specimen; additional testing is performed only when necessary as indicated by the results of the screening tests. Further testing may require another paracentesis, but because most specimens consist of ascitic fluid resulting from uncomplicated cirrhosis, no further testing is needed in a majority of cases. Also, because laboratories frequently store the fluid for a few days, additional testing can often be ordered on the stored fluid.
On the basis of cost analysis, tests can be classified as routine, optional, unusual, and unhelpful (Table 93-2).10 The cell count is the single most helpful ascitic fluid test. Only approximately 10 µL of fluid is required for a standard manual hemocytometer count. Therefore, if only 1 drop of fluid can be obtained, it should be sent for cell count. More fluid is almost always obtainable, however. The fluid should be submitted in an anticoagulant-containing tube (i.e., EDTA) to prevent clotting. Because the decision to begin empirical antibiotic treatment of suspected ascitic fluid infection is based largely on the absolute neutrophil count (which should have a turnaround time of a few minutes) rather than the culture (which takes 12 to 48 hours to demonstrate growth), the cell count is more important than the culture in the early detection and treatment of ascitic fluid infection.
TABLE 93-2
Ascitic Fluid Laboratory Tests
| Routine | Optional | Unusual | Unhelpful |
Cell count Albumin Total protein | Amylase Culture in blood culture bottles Glucose Gram stain LDH | Bilirubin Cytology TB smear, culture, and PCR test TG | Cholesterol Fibronectin Lactate pH |
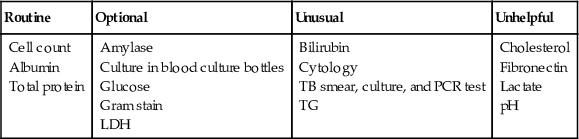
Even samples from asymptomatic outpatients undergoing therapeutic paracentesis should be sent for a cell and differential count; the information obtained can lead to early, life-saving treatment of bacterial infection.
Cell Count
Surprisingly, ascitic fluid cell counts have not been standardized. Some laboratories count mesothelial cells in addition to WBCs and label the sum as “nucleated cells.” The usefulness of mesothelial cell counts is not clear. The WBC count in uncomplicated ascites in the setting of cirrhosis is usually less than 500/mm3 (0.5 × 109/L) (see Fig. 93-2).10,19 During diuresis in patients with cirrhosis and ascites, the WBC count can concentrate to more than 1000/mm3 (1.0 × 109/L).19 A diagnosis of diuresis-related elevation of the ascitic fluid WBC count, however, requires that a prediuresis count be available, that normal lymphocytes predominate in the fluid, and that unexplained clinical symptoms or signs (e.g., fever or abdominal pain) be absent.
The upper limit of normal for the absolute PMN count in uncomplicated ascitic fluid in cirrhosis is usually stated to be lower than 250/mm3 (0.25 × 109/L).10,19 The short survival of PMNs results in relative stability of the absolute PMN count during diuresis.19 Therefore, the 250/mm3 (0.25 × 109/L) cutoff value remains reliable even after diuresis.
New methods have been developed to estimate the number of ascitic fluid cells.20 Dipsticks can detect an ascitic fluid PMN count greater than 250/mm3 (0.25 × 109/L) in 90 to 120 seconds. Urine-specific dipsticks have been used to date but are not very sensitive.21 An ascitic fluid–specific dipstick has been developed and calibrated to a PMN count of 250/mm3 (0.25 × 109/L).22
Any inflammatory process can result in an elevated ascitic fluid WBC count. SBP is the most common cause of inflammation of ascitic fluid and the most common cause of an elevated ascitic WBC count (see later). The total WBC count, as well as the absolute PMN count, is elevated in SBP, and PMNs usually account for more than 70% of the total WBC count. Also, in tuberculous peritonitis and peritoneal carcinomatosis, the total ascitic WBC count is frequently elevated, but usually with a predominance of lymphocytes.2
In most instances, bloody ascitic fluid is the result of a slightly traumatic tap. Leakage of blood into the peritoneal cavity leads to an elevated ascitic fluid WBC count. Because neutrophils predominate in blood, the ascitic fluid differential count may be altered by contamination of ascitic fluid with blood. To correct for this, 1 PMN is subtracted from the absolute ascitic fluid PMN count for every 250 RBCs19 (see Fig. 93-2). If the leakage of blood occurred at a remote time, the PMNs will have lysed, and the corrected PMN count will be a negative number. If the corrected PMN count in a bloody specimen is 250/mm3 (0.25 × 109/L) or greater, the patient must be assumed to be infected.
Serum-Ascites Albumin Gradient
Before the 1980s, the ascitic fluid total protein concentration was used to classify ascites as either exudative (at least 2.5 g/dL [25 g/L]) or transudative (<2.5 g/dL [25 g/L]). Unfortunately, this classification does not work well in ascitic fluid, and these terms as applied to ascitic fluid were never carefully defined or validated. Attempts at using combinations of LDH and serum-to-ascitic fluid ratios of LDH and protein also have not been shown to classify ascitic fluid accurately into exudates and transudates.23
The serum-ascites albumin gradient (SAAG) has been proved to categorize ascites better than the total protein concentration or other parameters24 (Box 93-1). The SAAG is based on oncotic-hydrostatic balance. Portal hypertension results in an abnormally high hydrostatic pressure gradient between the portal bed and ascitic fluid. A similarly large difference must exist between ascitic fluid and intravascular oncotic forces. Albumin exerts greater oncotic force per gram than that exerted by other proteins. Therefore, the difference between the serum and ascitic fluid albumin concentrations correlates directly with portal pressure.
Calculating the SAAG involves measuring the albumin concentration of serum and ascitic fluid specimens and simply subtracting the ascitic fluid value from the serum value. Unless a laboratory error has been made, the serum albumin concentration is always the larger value. The gradient is calculated by subtraction and is not a ratio. If the SAAG is 1.1 g/dL (11 g/L) or greater, the patient can be considered to have portal hypertension with an accuracy of approximately 97%.24 Also, if the serum albumin minus ascitic fluid total protein gradient is 1.1 g/dL (11 g/L) or greater, the patient has portal hypertension because the ascitic fluid albumin concentration cannot be greater than the ascitic fluid total protein concentration. Conversely, if the SAAG is less than 1.1 g/dL (11 g/L), the patient is unlikely to have portal hypertension. The SAAG does not explain the pathogenesis of ascites formation, nor does it explain where the albumin came from (i.e., liver or bowel). It simply gives the physician an indirect but accurate index of portal pressure. The accuracy of the test is excellent, even with ascitic fluid infection, diuresis, therapeutic paracentesis, IV infusions of albumin, and various causes of liver disease.24
Measurement of the ascitic fluid albumin concentration has been routine in some laboratories since the 1980s. The accuracy of the albumin assay at low albumin concentrations (e.g., <1 g/dL [10 g/L]) should be confirmed because many patients with ascites have a serum albumin concentration in the range of 2.0 g/dL (20 g/L) and an ascitic fluid albumin concentration in the range of 0 to 1.0 g/dL (0 to 10 g/L). If a patient with cirrhosis has a serum albumin level of less than 1.1 g/dL (11 g/L), as occurs in less than 1% of patients with cirrhosis and ascites, the SAAG will be falsely low.
The accuracy of the SAAG is also reduced when specimens of serum and ascites are not obtained nearly simultaneously. The specimens should be obtained on the same day, preferably within the same hour. Both serum and ascitic fluid albumin concentrations change over time; however, these values change in parallel, so the difference is stable. Arterial hypotension may result in a decrease in the portal pressure and a narrowing of the SAAG. Lipid interferes with the assay for albumin, and chylous ascites may result in a falsely high SAAG.
Serum hyperglobulinemia (serum globulin level > 5 g/dL [50 g/L]) leads to a high ascitic fluid globulin concentration and can narrow the albumin gradient by contributing to the oncotic forces. A narrowed gradient caused by high serum globulin levels occurs in only approximately 1% of ascitic fluid specimens. To correct the SAAG in the setting of a high serum globulin level, the following formula is used25:
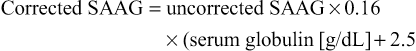
Approximately 5% of patients with ascites have “mixed” ascites (i.e., 2 causes of ascites) (see Table 93-1). Most of these patients have portal hypertension from cirrhosis as well as another cause of ascites, such as tuberculous peritonitis or peritoneal carcinomatosis.24 The albumin gradient is high (≥1.1 g/dL [11 g/L]) in mixed ascites, as a reflection of the underlying portal hypertension.24
The presence of a high SAAG does not confirm a diagnosis of cirrhosis; it simply indicates the presence of portal hypertension. Many causes of portal hypertension other than cirrhosis are recognized (Table 93-3; also see Table 93-1 and Chapter 92). A low SAAG does not confirm a diagnosis of peritoneal carcinomatosis. Although peritoneal carcinomatosis is the most common cause of a low SAAG, other causes exist (see Box 93-1). The SAAG needs to be determined only on the first paracentesis specimen in a given patient; it does not need to be repeated on subsequent specimens, if the first value is definitive. If the first result is borderline (e.g., 1.0 or 1.1 g/dL [10 or 11 g/L]), repeating the paracentesis and analysis usually provides a definitive result. High-albumin-gradient and low-albumin-gradient have replaced the modifiers “transudative” and “exudative” in the classification of ascites.24
TABLE 93-3
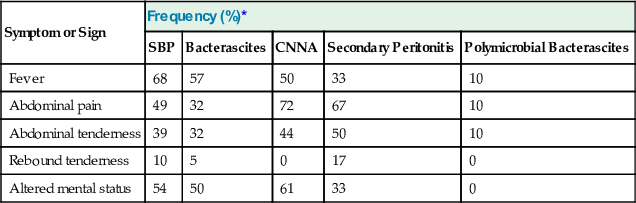
Symptoms and Signs of Ascitic Fluid Infection
| Symptom or Sign | Frequency (%)* | ||||
| SBP | Bacterascites | CNNA | Secondary Peritonitis | Polymicrobial Bacterascites | |
| Fever | 68 | 57 | 50 | 33 | 10 |
| Abdominal pain | 49 | 32 | 72 | 67 | 10 |
| Abdominal tenderness | 39 | 32 | 44 | 50 | 10 |
| Rebound tenderness | 10 | 5 | 0 | 17 | 0 |
| Altered mental status | 54 | 50 | 61 | 33 | 0 |
Culture
The most common bacterial infection of ascitic fluid, SBP, is monomicrobial, with a low bacterial concentration (median colony count of only 1 organism/mL).26 In the past, culture methodology for ascitic fluid was based on the notion that most episodes of ascitic fluid infection were polymicrobial with high colony counts, as in surgical peritonitis. The older method of culture consisted of inoculation (in the microbiology laboratory) of each of 3 agar plates and some broth with a few drops of fluid. This method of culturing ascitic fluid, as is used for urine or stool, is predictably insensitive for detecting monomicrobial infections with a low colony count. SBP is more like bacteremia in terms of the number of bacteria present; culturing ascitic fluid as if it were blood has a high yield.26 In fact, the sensitivity of culture in detecting bacterial growth in neutrocytic ascites (i.e., ascitic fluid with a PMN count of ≥ 250/mm3 [0.25 × 109/L]) depends on the method of culture used. The older method of culture was found to detect bacterial growth in approximately 50% of neutrocytic samples, whereas bedside inoculation of blood culture bottles with ascitic fluid detects growth in approximately 80%.10 Multiple prospective studies have demonstrated the superiority of the blood culture bottle method.10 Also, bedside inoculation is superior to delayed laboratory inoculation of blood culture bottles in the laboratory.27 Gene probes have become commercially available for the detection of bacteremia; hopefully, they will also lead to rapid (30-minute) and accurate detection of organisms in ascitic fluid. Culture will continue to be required, however, for assessment of the susceptibility of the organism to antibiotics.
Total Protein
The protein concentration in ascitic fluid in the setting of cirrhosis is determined almost entirely by the serum protein concentration and portal pressure. A patient with cirrhosis and a relatively high serum protein concentration will have a relatively high ascitic fluid protein concentration. Because of this relationship, almost 20% of ascitic fluid samples in patients with cirrhosis will have a protein concentration greater than 2.5 g/dL (25 g/L). The ascitic fluid total protein concentration does not increase during SBP; it remains stable before, during, and after infection.28 In fact, patients with the lowest ascitic protein concentrations are the most susceptible to spontaneous peritonitis.29 During a 10-kg diuresis, however, the ascitic fluid total protein concentration doubles, and 67% of such patients with cirrhosis and ascites have a protein concentration greater than 2.5 g/dL (25 g/L) by the end of diuresis.19 In almost one third of patients with malignancy-related ascites, the ascites is caused by massive liver metastases or hepatocellular carcinoma, and the ascitic fluid in these patients has a low protein concentration.2 In cardiac ascites, the ascitic fluid protein concentration is usually greater than 2.5 g/dL (25 g/L).30
The SAAG classifies fluid by the presence or absence of portal hypertension and is much more physiologic and intuitive than the old exudate/transudate classification. The exudate/transudate method of classification of ascites places many patients with cirrhosis and ascites and all patients with cardiac ascites in the exudate category, and many patients with malignant ascites and essentially all patients with spontaneously infected ascites in the transudate category. Clearly, this method of classification is not useful.24 The albumin gradient classifies cardiac ascites in the high-SAAG category, similar to ascites in cirrhosis. The high SAAG of cardiac ascites is presumably the result of high right-sided cardiac pressures. In patients with cardiac ascites, the SAAG may narrow with diuresis; such narrowing does not happen in patients with cirrhosis.
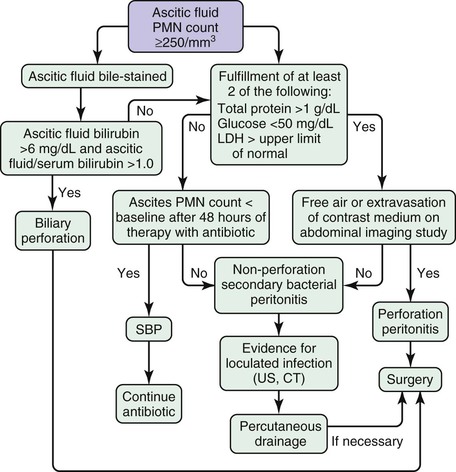
The combination of ascitic fluid total protein, glucose, and LDH is of value in distinguishing SBP from intestinal perforation with leakage of intestinal contents into ascites31 (Fig. 93-3). Patients who have neutrocytic ascitic fluid, in whom the clinical picture suggests bacterial peritonitis (rather than peritoneal carcinomatosis or tuberculous peritonitis) and who meet 2 of the following 3 criteria, are likely to have surgical peritonitis and merit immediate radiologic evaluation to determine if intestinal perforation with leakage of intestinal contents into ascites has occurred: total protein greater than 1 g/dL (10 g/L), glucose less than 50 mg/dL (2.8 mmol/L), and LDH greater than the upper limit of normal for serum.31
Glucose
The glucose molecule is small enough to diffuse readily into body fluid cavities. Therefore, the concentration of glucose in ascitic fluid is similar to that in serum, unless glucose is being consumed by ascitic fluid WBCs or bacteria.31 In early SBP, the ascitic fluid glucose concentration is similar to that of sterile fluid.28 By contrast, in SBP detected late in its course (as well as in the setting of intestinal perforation into ascitic fluid), the ascitic fluid glucose concentration usually drops to 0 mg/dL (0 mmol/L) because of large numbers of stimulated neutrophils and bacteria.31
Lactate Dehydrogenase
The LDH molecule is too large to enter ascitic fluid readily from blood,31 and the ascitic fluid concentration of LDH usually is less than half of the serum level in uncomplicated ascites in the setting of cirrhosis. In SBP, the ascitic fluid LDH level rises because of the release of LDH from neutrophils, and the ascitic fluid concentration is greater than that of serum. In secondary peritonitis, the LDH level is even higher than that seen in SBP and may be several-fold higher than the serum LDH level.31
Amylase
In uncomplicated ascites in the setting of cirrhosis, the ascitic fluid amylase concentration usually is half that of the serum value, approximately 50 U/L.32 In patients with acute pancreatitis or intestinal perforation (with release of luminal amylase into the ascitic fluid), the fluid amylase concentration is elevated markedly, usually greater than 2000 U/L, and approximately 5-fold greater than simultaneous serum values.31-33
Gram Stain
Gram stains of body fluids demonstrate bacteria only when more than 10,000 bacteria/mL are present. The median concentration of bacteria in ascitic fluid in SBP is only 1 organism/mL, similar to the colony count in bacteremia.26 Requesting an ascitic fluid Gram stain to detect bacteria in SBP is analogous to requesting a Gram stain of blood to detect bacteremia. Bacteria are detected on Gram stain only with overwhelming infection, as in advanced SBP or asplenic pneumococcal sepsis. Gram stain of ascitic fluid is most helpful in the diagnosis of free perforation of the intestine into ascitic fluid. In this setting, sheets of multiple different bacteria are found. Gram stain of the centrifuged sediment of 50 mL of ascites has a sensitivity of only 10% for visualizing bacteria in SBP.26
Smear and Culture for TB
A direct smear of ascitic fluid to detect mycobacteria is almost never positive because of the rarity of tuberculous peritonitis and the low concentration of mycobacteria in ascitic fluid in tuberculous peritonitis.34 The older literature suggests that 1 L of fluid should be cultured. The largest centrifuge tube found in most laboratories, however, has a capacity of 50 mL. In general, only 1 50-mL aliquot of fluid is centrifuged, and the pellet is cultured. In contrast to a sensitivity of approximately 50% for ascitic fluid mycobacterial culture with optimal processing, laparoscopy with histology and culture of peritoneal biopsies has a sensitivity approaching 100% for detecting tuberculous peritonitis.34 Tuberculous peritonitis can easily be confused with SBP, because both conditions are associated with abdominal pain and fever and half of the patients with tuberculous peritonitis have cirrhosis. A negative bacterial culture and predominance of mononuclear cells in the differential count, however, provide clues to the diagnosis of tuberculous peritonitis. DNA probes have become available to detect mycobacteria and probably will replace older methods of detection.35 Nevertheless, cultures still will be required to determine susceptibility to antimicrobial agents.
Cytologic Examination
Cytologic studies can be expected to detect malignancy only when tumor cells line the peritoneal cavity and exfoliate into the ascitic fluid (i.e., in peritoneal carcinomatosis). In the past, massive liver metastases and hepatocellular carcinoma superimposed on cirrhosis were not recognized as causes of “malignant” ascites.36 Cytologic studies should not be expected to detect tumor when the peritoneum is uninvolved, as in ascites resulting from portal hypertension in patients with hepatocellular carcinoma or massive liver metastases or from lymph node obstruction in patients with malignant lymphoma.2 In one study in which the location and type of tumor that caused ascites were confirmed by a standard test, only approximately two thirds of patients with malignancy-related ascites were found to have peritoneal carcinomatosis, but nearly 100% of patients with peritoneal carcinomatosis were reported to have positive findings on cytologic examination of ascitic fluid; the remaining one third of patients with massive liver metastases, chylous ascites caused by lymphoma, or hepatocellular carcinoma had negative cytologic findings.2 Therefore, the sensitivity of cytology is approximately 100% for detecting peritoneal carcinomatosis but much lower for detecting malignancy-related ascites caused by conditions other than peritoneal carcinomatosis. Cytologic studies should not be falsely positive if performed carefully; I have never encountered a false-positive result.
Because hepatocellular carcinoma rarely metastasizes to the peritoneum, a positive ascitic fluid cytology in a patient with hepatocellular carcinoma is unusual enough to be the subject of a case report.37 Measurement of the serum AFP concentration (which is always higher in serum than in ascitic fluid) may be of value in detecting hepatocellular carcinoma; serum AFP is much more sensitive than ascitic fluid cytology for this purpose (see Chapter 96).2 In malignancy-related ascites, the fluid may have an elevated PMN count, presumably because dying tumor cells attract neutrophils.2 The elevated PMN count may cause confusion with SBP; however, a predominance of lymphocytes in malignancy-related ascites is usual. Flow cytometry and magnetic enrichment of ascitic fluid as an adjunct to cytology may further increase diagnostic accuracy.38
TG
A TG level should be measured in opalescent or frankly milky ascitic fluid (see Fig. 93-2). By definition, chylous ascites has a TG concentration greater than 200 mg/dL (2.26 mmol/L) and greater than the serum level; usually, the level is greater than 1000 mg/dL (11.30 mmol/L).39 In sterile but slightly cloudy ascitic fluid specimens in the setting of cirrhosis, in the absence of an elevated cell count, the TG concentration is elevated: 64 ± 40 mg/dL (0.72 ± 0.45 mmol/L), compared with 18 ± 9 mg/dL (0.20 ± 0.10 mmol/L) in clear ascites in the setting of cirrhosis.16
Tests That Are Seldom Helpful
Tests that have been proposed to be helpful in the analysis of ascitic fluid but shown subsequently to be of no benefit include determination of pH, lactate, fibronectin, and cholesterol. The studies that attempted to validate the value of pH and lactate included small numbers of patients and used suboptimal culture techniques. In the 2 largest studies, which did not have some of the deficiencies of smaller, earlier studies, the ascitic fluid pH and lactate were found not to be helpful.40,41 The pH was found to have no impact on decision making regarding the use of empirical antibiotic therapy.40
Fibronectin and cholesterol were proposed to be useful in detecting malignant ascites. The basic premise in studies of these markers was that ascitic fluid cytologic examination is insensitive. Unfortunately, the design of the studies was problematic, several subgroups of malignancy-related ascites (e.g., massive liver metastases, hepatocellular carcinoma with cirrhosis) were not considered, and appropriate control groups (e.g., patients with ascites caused by conditions other than cirrhosis or peritoneal carcinomatosis) were not included. Other studies have demonstrated that in patients with massive liver metastases, ascitic fluid fibronectin and cholesterol concentrations are not abnormally elevated.42,43 Therefore, in patients with malignancy-related ascites and negative cytologic findings, these “humoral tests of malignancy” are usually negative. Additionally, patients with high-protein ascites in the absence of cirrhosis nearly always have ascitic fibronectin and cholesterol elevations despite the absence of malignancy.2,42,43
CEA in ascitic fluid was proposed as a helpful marker for detecting malignant ascites.44 The study that attempted to validate this proposal, however, was flawed, and more studies, with various subgroups of patients, are required before testing for ascitic fluid CEA can be considered validated.
Measurement of adenosine deaminase has been proposed as a useful test for detecting peritoneal TB. In the United States, however, where greater than 50% of patients with tuberculous peritonitis have underlying cirrhosis, the adenosine deaminase level has been found to be too insensitive to be helpful.34
Differential Diagnosis of Ascites
Although cirrhosis is the cause of ascites in most patients with ascites evaluated by a primary care provider, a cause other than liver disease is found in approximately 15% of patients (see Table 93-1). Approximately 5% of patients have 2 causes of ascites (i.e., mixed ascites).24 Usually, these patients have cirrhosis plus peritoneal carcinomatosis or tuberculous peritonitis (see Table 93-1). Because TB is potentially fatal but curable and frequently occurs in patients with cirrhosis and preexisting ascites, the physician must not assume that liver disease is the only cause of ascites in a febrile alcoholic patient if the ascitic fluid analysis is atypical. If the ascitic fluid lymphocyte count is unusually high, for example, tuberculous peritonitis may be present. Liver diseases other than cirrhosis (e.g., alcoholic hepatitis or fulminant hepatic failure) may cause ascites (see Table 93-1).
An algorithm for the differential diagnosis of ascites is shown in Figure 93-2. This proposed strategy is applicable to a majority of patients with ascites, including many with the causes listed in Table 93-1. Not every patient (including patients with rare causes of ascites) can be categorized readily with such an algorithm, however. Many patients with enigmatic ascites eventually are found to have 2 or even 3 causes of ascites (e.g., heart failure, cirrhosis caused by NASH, diabetic nephropathy). In these cases, the sum of predisposing factors leads to sodium and water retention, even though each factor alone may not be severe enough to cause fluid overload.
In most patients with ascites, cirrhosis is the cause. Cirrhotic ascites, especially when low in protein, is complicated frequently by SBP (see later).29 Other forms of ascites are rarely complicated by spontaneous peritonitis.
The intestine can perforate, with spillage of contents in patients with ascites of any cause, cirrhosis or otherwise. The ascitic fluid analysis in intestinal perforation is dramatically different from that in SBP (see Fig. 93-3).31,45 Distinguishing SBP from surgical peritonitis in a patient with cirrhosis is critical to the patient's survival; SBP is treated with antibiotics alone, whereas surgical peritonitis is treated with antibiotics and emergency surgical intervention (see Chapter 38).
Cancer accounts for fewer than 10% of cases of ascites (see Table 93-1). Not all cases of malignancy-related ascites are caused by peritoneal carcinomatosis (see earlier); the characteristics of the ascitic fluid and the treatments differ depending on the pathophysiology of the ascites2 (Box 93-2).
Heart failure accounts for less than 5% of cases of ascites. Cardiac ascites is characterized by a high albumin gradient, high ascitic fluid protein concentration, and normal blood hematocrit value.30 The gradient may narrow with diuresis, in contrast to cirrhosis. Patients with cardiac ascites often have alcoholic cardiomyopathy, with cardiomegaly on a chest radiograph and 4-chamber enlargement of the heart on an echocardiogram. Clinically, heart failure may mimic cirrhosis, including the presence of small nonbleeding esophageal varices and hepatic encephalopathy.46 Ascites in the setting of cirrhosis is characterized by a high albumin gradient, as in cardiac ascites, but a low protein concentration, and patients with cirrhosis and ascites have a lower mean blood hematocrit value of 32%.30 Serum pro-brain-type natriuretic peptide also can be useful in distinguishing cardiac ascites from ascites due to cirrhosis. The median value is 6100 pg/mL in the former and only 166 pg/mL in the latter.47
In the United States, tuberculous peritonitis generally is a disease of Asian and Latin American immigrants to the West Coast, poor African Americans, and the elderly. Tuberculous peritonitis was a rare disease between 1955 and 1985, but it subsequently increased in prevalence because of the advent of AIDS.48 Fifty percent of patients with tuberculous peritonitis have underlying cirrhosis (and thus mixed ascites). Although most patients with liver disease are not unusually predisposed to the hepatotoxicity of anti-TB drugs, they tolerate drug toxicity less well than do patients with a normal liver.49 Underdiagnosis can lead to unnecessary deaths from untreated TB, whereas overdiagnosis and overtreatment of suspected but unproved tuberculous peritonitis may lead to unnecessary deaths from the hepatotoxicity of isoniazid and other anti-TB drugs. If the clinical circumstances (e.g., fever in an immigrant from an area endemic for TB) and results of the initial ascitic fluid analysis (high lymphocyte count) suggest TB, strong consideration should be given to an urgent laparoscopy with histologic examination and culture of peritoneal biopsy specimens. If at laparoscopy the peritoneum demonstrates the typical “millet-seed” and “violin-string” appearance, anti-TB therapy can be started immediately. Blind peritoneal biopsy may be performed in patients without cirrhosis; however, in patients with cirrhosis, the predictable presence of peritoneal collateral veins makes blind biopsy potentially hazardous, and laparoscopically guided biopsy is preferable. Suspected tuberculous peritonitis is one of the few remaining indications for diagnostic laparoscopy. Peritoneal coccidioidomycosis can mimic tuberculous peritonitis, including its appearance at laparoscopy, and can occur in patients without AIDS.50 The high sensitivities of cytology for peritoneal carcinomatosis and US-guided biopsy for focal liver lesions have obviated the need for laparoscopy in detecting tumor, for all practical purposes.2
Pancreatic ascites, an uncommon condition, occurs in patients with clinically obvious severe acute pancreatitis or a history of chronic pancreatitis or pancreatic trauma (see Chapters 58 and 59).32 Ordering an ascitic fluid amylase level on all ascitic fluid samples is unnecessary; the test is indicated only in patients in whom pancreatitis is suspected or the initial ascitic fluid is nondiagnostic (see Table 93-2). Patients with alcohol-related pancreatic ascites may also have underlying alcoholic cirrhosis. Pancreatic ascites frequently is neutrocytic and may also be complicated by bacterial infection. Patients with an ascitic fluid neutrophil count of 250/mm3 (0.25 × 109/L) or greater merit empirical antibiotic coverage, at least until the cause of the elevated neutrophil count is explained.
Nephrogenous ascites is a poorly understood form of ascites that develops in patients undergoing hemodialysis.51 On careful evaluation, most patients with ascites in the setting of hemodialysis are found to have another cause of ascites, usually cirrhosis from alcohol abuse or from hepatitis C. The presence of a second cause of fluid overload explains why these patients have ascites, whereas a majority of patients on dialysis do not. “Urine ascites,” due to leakage of urine into the abdominal cavity, is most likely to result from a surgical complication and is characterized by a low protein concentration, low SAAG, and ascitic fluid-to-serum creatinine level ratio greater than 1.
Although the nephrotic syndrome used to be a common cause of ascites in children, it is rare in adults.52 When it occurs in adults, a second cause of ascites usually is present, just as in nephrogenous ascites. The ascitic fluid is usually characterized by a low protein concentration and low SAAG and can be complicated by SBP.
Chlamydial (or rarely gonococcal) peritonitis should be suspected in sexually active young women with fever and neutrocytic, high-protein, low-gradient ascites and no evidence of liver disease. This infection responds rapidly to oral doxycycline and is one of the few curable causes of ascites.
In some patients, pathologic accumulation of fluid develops in the peritoneal cavity as a result of leakage from a ruptured viscus (e.g., “bile ascites” from a ruptured gallbladder).18,31 The ascitic fluid analysis is critical to the preoperative diagnosis of this condition (see earlier and Fig. 93-3).
Chylous ascites develops when intra-abdominal lymphatics containing chyle rupture. Cirrhosis is the cause of chylous ascites in more than 90% of the patients whom I have encountered (see Table 93-1).17,24 The high lymphatic flow and pressure are presumed to be the cause of lymphatic rupture in patients with cirrhosis. In addition, retroperitoneal surgery and radical pelvic surgery in patients with cancer can transect lymphatics and thereby lead to chylous ascites. In the past, this form of ascites was caused by a malignancy in nearly 90% of cases.39
Additional causes of ascites include ambulatory peritoneal dialysis, Budd-Chiari syndrome, myxedema, connective tissue disease, postoperative ascites, and rare entities. With the iatrogenic form of ascites associated with peritoneal dialysis, the patient is usually not under the care of a gastroenterologist. Although Budd-Chiari syndrome is regularly complicated by ascites, hepatic vein thrombosis is rare and accounts for less than 0.1% of cases of ascites (see Chapter 85). Ascites in patients with myxedema appears to be related to heart failure53; treatment of the hypothyroidism cures the fluid retention. Serositis with development of ascites may complicate systemic lupus erythematosus (see Chapter 36).7
Ascites after abdominal surgery (often after cholecystectomy in the setting of asymptomatic gallstones and abnormal liver biochemical test results) is a common mode of presentation of previously undiagnosed cirrhosis.54 Resection of hepatocellular carcinoma in the setting of cirrhosis regularly leads to hepatic decompensation, which all too often starts a downward spiral ending in death.55
Aggressive hormone administration to induce ovulation can lead to ascites from ovarian hyperstimulation syndrome.56 Other rare causes of ascites include the POEMS syndrome (polyneuropathy, organomegaly, endocrinopathy, M component, and skin changes) and hemophagocytic syndrome.57,58 The latter is a rare syndrome that usually occurs in patients with leukemia or lymphoma and can masquerade as decompensated cirrhosis.58 Ascites that recurs or does not resolve after liver transplantation appears to be due to relative hepatic venous outflow obstruction or hepatitis C but frequently is enigmatic.59,60
Complications
Ascitic Fluid Infection, Including SBP
Ascitic fluid infection can be classified into 5 categories based on ascitic culture results, PMN count, and presence or absence of a surgical source of infection (Box 93-3). An abdominal paracentesis must be performed and ascitic fluid analyzed before a confident diagnosis of ascitic fluid infection can be made. A “clinical” diagnosis of infected ascitic fluid without a paracentesis is inadequate.
Classification
Of the 3 subtypes of spontaneous ascitic fluid infection, the prototype is SBP. The diagnosis of SBP is made when there is a positive ascitic fluid culture and an elevated ascitic fluid absolute PMN count (i.e., at least 250/mm3 [0.25 × 109/L]) without evidence of an intra-abdominal surgically treatable source of infection.10 When Correia and Conn coined the term spontaneous bacterial peritonitis in 1975, their goal was to distinguish this form of infection from surgical peritonitis.61 Therefore, although many patients with SBP have a focus of infection (e.g., urinary tract infection or pneumonia), the diagnosis of SBP is still appropriate unless the focus requires surgical intervention (e.g., a ruptured viscus). I have not encountered a convincing case of polymicrobial SBP; all of the patients presumed to have SBP in whom ascitic fluid cultures initially grew more than 1 organism eventually were found to have surgical peritonitis or an erroneous culture result (e.g., a pathogen plus a contaminant or 2 colony morphologies of 1 species of bacteria).
The criteria for a diagnosis of monomicrobial non-neutrocytic bacterascites (MNB) include (1) a positive ascitic fluid culture for a single organism, (2) an ascitic fluid PMN count lower than 250/mm3 (0.25 × 109/L), and (3) no evidence of an intra-abdominal surgically treatable source of infection.62 In the older literature, MNB was either grouped with SBP or labeled “asymptomatic bacterascites.” Because many patients with bacterascites have symptoms, the modifier asymptomatic seems inappropriate.
Culture-negative neutrocytic ascites (CNNA) is diagnosed when (1) the ascitic fluid culture grows no bacteria, (2) the ascitic fluid PMN count is 250/mm3 (0.25 × 109/L) or greater, (3) no antibiotics have been given (not even a single dose), and (4) no other explanation for an elevated ascitic PMN count (e.g., hemorrhage into ascites, peritoneal carcinomatosis, TB, or pancreatitis) can be identified.63 This variant of ascitic fluid infection is seldom diagnosed when sensitive culture methods are used.26
Secondary bacterial peritonitis is diagnosed when (1) the ascitic fluid culture is positive (usually for multiple organisms), (2) the PMN count is 250/mm3 (0.25 × 109/L) or greater, and (3) an intra-abdominal surgically treatable primary source of infection (e.g., perforated intestine, perinephric abscess) has been identified.31,45 The importance of distinguishing this variant from SBP is that secondary peritonitis usually requires emergency surgical intervention (see Chapter 38).
Polymicrobial bacterascites is diagnosed when (1) multiple organisms are seen on Gram stain or cultured from the ascitic fluid and (2) the PMN count is lower than 250/mm3 (0.25 × 109/L).64 This diagnosis should be suspected when the paracentesis is traumatic or unusually difficult because of ileus or when stool or air is aspirated into the paracentesis syringe. Polymicrobial bacterascites is essentially diagnostic of intestinal perforation by the paracentesis needle.
Clinical Setting
The spontaneous variants of ascitic fluid infection—SBP, CNNA, and MNB—occur almost exclusively in the setting of severe liver disease. The liver disease usually is chronic (cirrhosis), but may be acute (fulminant hepatic failure) or subacute (alcoholic hepatitis). Cirrhosis of all causes can be complicated by spontaneous ascitic fluid infection. Spontaneous infection of noncirrhotic ascites is rare.
Essentially all patients with SBP have an elevated serum bilirubin level and prolonged prothrombin time because of advanced cirrhosis.10 Ascites appears to be a prerequisite for the development of SBP. The peritonitis is unlikely to precede the development of ascites. Usually, the infection develops when the volume of ascites is at its maximum.
Secondary bacterial peritonitis and polymicrobial bacterascites can develop with ascites of any type. The only prerequisite, in addition to the presence of ascites, is an intra-abdominal surgical source of infection.31 Such an infection can result from penetration of a needle into the bowel during attempted paracentesis.64
Pathogenesis
Currently available evidence suggests that the spontaneous forms of ascitic fluid infection are the result of overgrowth of a specific organism in the intestine, translocation of that microbe from the intestine to mesenteric lymph nodes, and resulting spontaneous bacteremia and subsequent colonization of susceptible ascitic fluid65,66 (Fig. 93-4). When bacteria enter the fluid in the abdomen, by whatever route, a battle ensues between the virulence factors of the organism and the immune defenses of the host.67 The ascitic fluid protein concentration does not change with development of spontaneous infection.28 Low-protein ascitic fluid (e.g., protein content < 1 g/dL [10 g/L]) is particularly susceptible to SBP.29 The endogenous antimicrobial (opsonic) activity of human ascitic fluid correlates directly with the protein concentration of the fluid.66 Patients with deficient ascitic fluid opsonic activity are predisposed to SBP.68 Patients with detectable ascitic fluid opsonic activity appear to be protected from SBP unless they are exposed to a particularly virulent organism (e.g., Salmonella).67,68
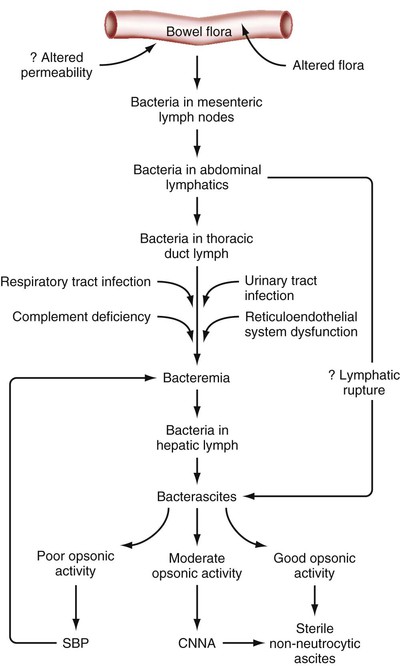
Studies in both patients and animals with cirrhosis demonstrate that MNB is common.62,69 Pieces of bacterial DNA are commonly present in serum and ascitic fluid of patients with cirrhosis.70 In both humans and rats, most episodes of bacterascites resolve without antibiotic treatment.62,69 The fluid frequently becomes sterile without an increase in ascitic PMNs. Apparently, the host's defense mechanisms are able to eradicate the invading bacteria on most occasions. Uncontrolled infection probably develops only when the defenses are weak or the organism is virulent (see Fig. 93-4). Bacterascites probably is more common than SBP. Conceivably, ascitic fluid in the setting of cirrhosis is colonized regularly by bacteria, and almost just as regularly, the colonization resolves. The entry of PMNs into the fluid probably signals failure of the peritoneal macrophages to control the infection.71 A majority of episodes of MNB appear to resolve in rats and humans with cirrhosis, whereas untreated SBP is frequently fatal. In summary, MNB probably represents an early stage of ascitic fluid infection, which can resolve or progress to CNNA or to SBP.
Most episodes of CNNA are diagnosed by insensitive culture methods for which the numbers of bacteria are insufficient to reach the threshold of detectability.26 Inoculation of ascitic fluid into blood culture bottles can lead to detection of a single organism in the cultured aliquot of fluid, whereas the older method of culture by inoculation of agar plates and broth probably requires at least 100 organisms/mL (see earlier).26 Even when optimal culture methods are used, however, a variable percentage of specimens of neutrocytic ascitic fluid grow no bacteria. A study of rapid sequential paracenteses (before the initiation of antibiotic treatment) in patients with CNNA demonstrated that, in most cases, the PMN count dropped spontaneously and the culture results remained negative in the second specimen.72 When sensitive culture techniques are used, CNNA probably results from (1) previous antibiotic treatment (even 1 dose), (2) an inadequate volume of fluid inoculated, or (3) spontaneously resolving SBP in which the paracentesis is performed after all bacteria have been killed by host defenses but before the PMN count has normalized.
The pathogenesis of secondary bacterial peritonitis is more straightforward than that of SBP. When the intestine perforates, billions of bacteria flood into the ascitic fluid. In the absence of a frank perforation, bacteria may cross inflamed tissue planes and enter the fluid. The pathogenesis of polymicrobial bacterascites is also obvious.64 A paracentesis needle enters the bowel, and the bowel contents are released into the ascites.
Symptoms and Signs
Although 87% of patients with SBP are symptomatic at the time the infection is diagnosed, the symptoms and signs of infection are often subtle, such as a slight change in mental status.62 Without prompt paracentesis, the diagnosis and treatment of infected ascites may be delayed, often resulting in the death of the patient. The symptoms and signs manifested in all 5 variants of ascitic fluid infection are listed in Table 93-3.
Frequency
Since the 1980s, routine paracenteses at the time of hospitalization in patients with ascites have provided data regarding the frequency of ascitic fluid infection. In the 1980s, approximately 10% of patients with ascites were infected at the time of hospital admission; of the subgroup of patients with cirrhosis, about 27% were infected.10 At present, because of measures to prevent SBP, the frequency has dropped significantly (see later). Of patients with culture-positive ascitic fluid, about two thirds have neutrocytic ascitic fluid (SBP), and one third have MNB.62 The frequency of CNNA depends largely on the culture technique (see earlier). Polymicrobial bacterascites occurs in only 1 in 1000 paracenteses. Secondary bacterial peritonitis is found in only 0% to 2% of patients with ascites at the time of hospital admission.10,31
Bacteriology
Escherichia coli, streptococci (mostly pneumococci), and Klebsiella caused most episodes of SBP and MNB in the past in patients who were not receiving selective intestinal decontamination. Now more Gram-positive and resistant Gram-negative organisms are being isolated (Table 93-4).73 Selective intestinal decontamination and recent exposure to antibiotics are causing a change in the bacteria isolated from patients in whom an ascitic fluid infection develops. We may encounter organisms for which we have no effective antibiotics.74 The antibiotic susceptibility of bacteria that cause infections in patients with cirrhosis should be tested periodically in each hospital, and the preferred empirical antibiotic regimens should be adapted accordingly.73
TABLE 93-4
Pathogens in Ascitic Fluid Infection
| Organism | Frequency (%)* | |||
| SBP | Monomicrobial Non-Neutrocytic Bacterascites | Secondary Bacterial Peritonitis | SBP with SID | |
| Monomicrobial | ||||
| Escherichia coli | 37 | 27 | 20 | 0 |
| Klebsiella pneumoniae | 17 | 11 | 7 | 7 |
| Streptococcus pneumoniae | 12 | 9 | 0 | 29 |
| Streptococcus viridans | 9 | 2 | 0 | 0 |
| Staphylococcus aureus | 0 | 7 | 13 | 0 |
| Miscellaneous Gram-positive | 14 | 30 | 0 | 50 |
| Miscellaneous Gram-negative | 10 | 14 | 7 | 7 |
| Polymicrobial | 1 | 0 | 53 | 7 |
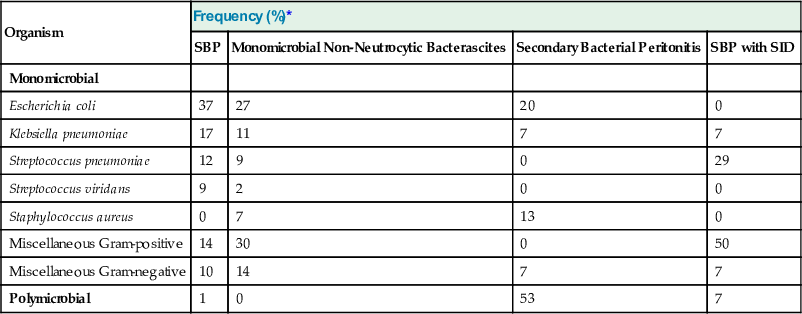
CNNA is, by definition, culture-negative, and polymicrobial bacterascites is, by definition, polymicrobial. The most apparent difference between the spontaneous forms of ascitic fluid infection and the secondary forms (secondary peritonitis and polymicrobial bacterascites) is that the former always are monomicrobial and the latter usually are polymicrobial. Although older papers reported that anaerobic bacteria were present in approximately 6% of cases of SBP, the detection of anaerobes probably reflected unrecognized cases of secondary bacterial peritonitis. In more recent series, anaerobes have been found in approximately 1% of cases of SBP and MNB.26,62
Risk Factors
Patients with cirrhosis are unusually predisposed to bacterial infection because of multiple defects in immune defense. The concept that cirrhosis is a form of acquired immunodeficiency (in the generic sense) is relatively new. In a prospective study, a bacterial infection occurred in 34% of 405 patients with cirrhosis at the time of admission to the hospital or during the hospitalization.75 Low ascitic fluid total protein concentrations, as well as the phagocytic (both motile and stationary) dysfunction associated with cirrhosis, are risk factors for bacterial infection.
Paracentesis itself has been proposed as a risk factor for ascitic fluid infection, but this theoretical risk has not been substantiated in prospective studies of paracentesis-related complications.12 SBP is statistically more likely to be diagnosed on the first paracentesis than on subsequent taps.12 Needle-induced ascitic fluid infections do not occur unless the bowel is penetrated by the paracentesis needle.12,64 Fortunately, bowel penetration occurs in only 1 in 1000 taps. One would expect bacteria of the skin flora to be isolated more frequently if poor paracentesis technique were the cause of many cases of SBP; yet skin flora microorganisms are seldom isolated from ascitic fluid when sterile technique is used.26 Iatrogenic peritonitis is most likely to occur when the paracentesis needle enters the bowel during a difficult paracentesis.
GI hemorrhage is an under-recognized risk factor for the development of spontaneous bacteremia and SBP (see Chapters 20 and 92). The cumulative probability of infection during a single hospitalization for bleeding is approximately 40%.76 The risk appears to peak 48 hours after the onset of hemorrhage. The high risk of infection probably is mediated by a shock-induced increase in the translocation of bacteria from the intestine to extraintestinal sites. Urinary tract infections also constitute an under-recognized risk factor for SBP.77
Diagnosis
Timely diagnosis of ascitic fluid infection requires a high index of suspicion and a low threshold for performing a paracentesis. Clinical deterioration, especially fever or abdominal pain, in a patient with ascites should raise the suspicion of infection and prompt a paracentesis. If the ascitic fluid PMN count is elevated, the working diagnosis is ascitic fluid infection until proved otherwise. Although peritoneal carcinomatosis, pancreatitis, hemorrhage into ascites, and TB can lead to an elevated ascitic fluid PMN count, most cases of neutrocytic ascites are caused by bacterial infection. A predominance of PMNs in the WBC differential count lends further support to the diagnosis of infection. An elevated absolute ascitic fluid PMN count with a predominance of neutrophils in a clinical setting compatible with infection should prompt empirical antibiotic therapy (see later).
Although SBP is approximately 20 times as common as surgical peritonitis in a patient with ascites, secondary peritonitis should be considered in any patient with neutrocytic ascites (see Chapter 38).45 Clinical symptoms and signs do not distinguish patients with secondary peritonitis from those with SBP (see Fig. 93-3).31 Even with free perforation of the colon into ascitic fluid, a classic surgical abdomen does not develop. Peritoneal signs require contact of inflamed visceral and parietal peritoneal surfaces, and such contact does not occur when there is a large volume of fluid separating these surfaces. Intestinal perforation can be suspected and pursued if a specimen of ascites is neutrocytic and meets 2 of the following 3 criteria (see Fig. 93-3): (1) total protein greater than 1 g/dL (10 g/L), (2) glucose less than 50 mg/dL (2.8 mmol/L), and (3) LDH greater than the upper limit of normal for serum.31 In the setting of a perforated viscus, cultures of ascitic fluid nearly always disclose multiple organisms, except in gallbladder rupture, which is usually monomicrobial.18 Brown ascitic fluid with a bilirubin concentration that is greater than 6 mg/dL (102 µmol/L) and greater than the serum level is indicative of biliary or proximal small intestinal perforation into ascites.18 An ascitic fluid amylase level that is greater than 5-fold that of the serum level also may be indicative of intestinal rupture (but not gallbladder rupture) with the release of luminal amylase.31,32
The initial ascitic fluid analysis is helpful in delineating which patients are likely to have a ruptured viscus (see Fig. 93-3). Within minutes of the detection of neutrocytic ascitic fluid, these patients should undergo abdominal CT to confirm and localize the site of rupture.45 If perforation is documented, emergency surgical intervention is the next step. Timing is crucial; after septic shock occurs, death is nearly certain. Antibiotic therapy without surgical intervention in the treatment of a ruptured viscus is predictably unsuccessful.
In contrast to patients with peritonitis resulting from perforation of a viscus, patients with secondary peritonitis unrelated to perforation tend not to have a diagnostic initial ascitic fluid analysis.31 The need to make the diagnosis of secondary peritonitis in patients without free perforation is less urgent, and there may be time to evaluate the response of the ascitic PMN count and fluid culture to treatment with antibiotics. It is best to repeat the paracentesis to assess the response to treatment after 48 hours of therapy; by 48 hours, the ascitic PMN count will be lower than the pretreatment value and the ascitic culture will be negative in essentially every patient with SBP who has been treated with an appropriate antibiotic.31 Before 48 hours of treatment, the ascitic PMN count may rise to a value higher than baseline in either SBP or secondary peritonitis.31 The culture remains positive in secondary peritonitis and becomes rapidly negative in SBP (see Fig. 93-3).31 Whereas antibiotics alone cannot control secondary peritonitis, medical therapy cures SBP rapidly.31
Treatment
Patients with an ascitic fluid PMN count of 250/mm3 (0.25 × 109/L) or greater and a clinical scenario compatible with ascitic fluid infection should receive empirical antibiotic treatment (Table 93-5).10,78 Patients with hemorrhage into the ascitic fluid, peritoneal carcinomatosis, pancreatic ascites, or tuberculous peritonitis may have an elevated ascitic PMN count that is unrelated to SBP and usually do not require empirical antibiotic treatment. If they do receive antibiotics, the ascitic PMN count usually fluctuates randomly, in contrast to the dramatic reduction in PMN count typical of SBP. If the clinical picture is unclear initially, the physician should err on the side of antibiotic treatment (with a non-nephrotoxic antibiotic). If ascitic fluid cultures are negative, the antibiotic can be stopped after 48 hours. In patients with uninfected neutrocytic ascitic fluid (except those with hemorrhage), lymphocytes usually predominate in the ascitic fluid differential count, in contrast to those with SBP, in whom PMNs predominate. In patients with bloody ascitic fluid, a “corrected” PMN count should be calculated (as discussed earlier). Antibiotic therapy is not necessary for patients with bloody fluid unless the corrected ascitic fluid PMN count is 250/mm3 (0.25 × 109/L) or greater.
TABLE 93-5
Treatment of Ascitic Fluid Infection
| Diagnosis | Treatment |
| SBP | 5 days of IV antibiotic to which the organism is highly susceptible (e.g., cefotaxime 2 g every 8 hours empirically followed by more specific therapy after susceptibility results are available) |
| Monomicrobial non-neutrocytic bacterascites | 5 days of IV antibiotic to which the organism is highly susceptible, if the patient is symptomatic or persistently culture-positive; not all patients with bacterascites require treatment |
| Culture-negative neutrocytic ascites | 5 days of IV third-generation cephalosporin (e.g., cefotaxime 2 g every 8 hours) |
| Secondary bacterial peritonitis | Surgical intervention plus ≈ 2 weeks of IV cephalosporin (e.g., cefotaxime 2 g every 8 hours) plus an antianaerobic drug such as metronidazole* |
| Polymicrobial bacterascites | IV third-generation cephalosporin (e.g., cefotaxime 2 g every 8 hours) plus an antianaerobic drug such as metronidazole* Duration is determined by clinical response and serial ascitic fluid PMN counts and cultures |
The decision to begin empirical antibiotic treatment in patients with bacterascites must be individualized. Many episodes resolve without treatment62; however, the hospital mortality rate of 32% in patients when MNB is attributable, at least, in part, to infection.62 Therefore, treatment appears to be warranted in many patients. By definition, the ascitic PMN count is lower than 250/mm3 (0.25 × 109/L) in this variant of ascitic fluid infection, and the PMN count cannot be the only parameter on which to base the decision about empirical therapy. Most patients with MNB in whom the colonization does not resolve progress to SBP and have symptoms or signs of infection at the time of the paracentesis that documents bacterascites.62 Therefore, patients with cirrhosis and ascites who have convincing symptoms or signs of infection should receive treatment regardless of the ascitic fluid PMN count. Empirical treatment can be discontinued after only 2 to 3 days if the culture demonstrates no growth. Asymptomatic patients may not need treatment.62 The paracentesis should be repeated for cell count and culture in patients without clinical evidence of infection, as soon as it is known that the initial culture result is positive. If the PMN count has risen to at least 250/mm3 (0.25 × 109/L) or if symptoms or signs of infection have developed, antibiotics should be started. Culture results usually are negative in patients without a rise in the ascitic fluid PMN count on repeat paracentesis and without clinical evidence of infection, and these persons do not require antibiotics62 because colonization has been eradicated by host immune defenses.
The physician will not know initially that the ascitic fluid culture is destined to be negative in a patient with CNNA; therefore, empirical antibiotic treatment should be started. When the preliminary culture demonstrates no growth, it is helpful to repeat the paracentesis after 48 hours of therapy to assess the response of the PMN count to antibiotics. A dramatic decline in PMN count (always below the baseline pretreatment value and frequently a reduction of more than 80%) confirms a response to treatment.31 In such cases, a few more days of therapy is probably warranted. A stable ascitic fluid PMN count, especially with a predominance of lymphocytes and monocytes, suggests a nonbacterial (or mycobacterial) cause of ascitic fluid neutrocytosis, and the fluid should be sent for cytologic examination and mycobacterial culture. Because a negative culture result may be due to insensitive culture techniques, the prevalence of CNNA in a hospital that still uses older methods of culture (sending a tube or syringe of fluid to the laboratory) can be reduced by convincing the microbiology laboratory to accept and process ascitic fluid submitted in blood culture bottles.26
Gram stain of the ascitic fluid is most helpful in detecting secondary peritonitis, in which multiple different bacterial forms are seen, but is of little value in guiding the choice of empirical antibiotic treatment for spontaneous ascitic infections (see earlier). I have found that use of the Gram stain did not help narrow the antibiotic coverage in even 1 patient of approximately 700 with SBP. Only approximately 10% of Gram stains demonstrate organisms in SBP.26 If a Gram stain indicates secondary peritonitis, coverage for anaerobic flora, in addition to coverage for aerobic and facultative anaerobic flora, is required, as is an emergency search for the source of the infection (see Fig. 93-3 and Table 93-5).31 Therefore, a positive Gram stain may lead to broader antibiotic coverage, rather than narrower coverage. Narrow coverage based on a misinterpretation of the significance of the results of the Gram stain may lead to uncontrolled infection and death before resistance of the isolated organism to the chosen antibiotic becomes apparent.
Until the results of susceptibility testing are available, relatively broad-spectrum antibiotic therapy is warranted in patients with suspected ascitic fluid infection. After sensitivities are known, the spectrum of coverage can usually be narrowed. The antibiotics that have been recommended for empirical treatment have changed over the years. In the late 1970s, the combination of ampicillin and gentamicin was promoted, but this recommendation was not based on susceptibility testing or efficacy data. One study has documented an adjusted odds ratio of 4.0 for aminoglycosides as a risk factor for renal dysfunction in patients with cirrhosis.79 Evidence that newer aminoglycosides are less nephrotoxic than gentamicin is lacking.
Several antibiotics are now available for the treatment of ascitic fluid infection. Cefotaxime, a third-generation cephalosporin, has been shown in a controlled trial to be superior to ampicillin plus tobramycin for the treatment of SBP.80 Fully 98% of causative organisms were susceptible to cefotaxime, which did not result in superinfection or nephrotoxicity.80 Cefotaxime or a similar third-generation cephalosporin appears to be the treatment of choice for suspected SBP.10 Anaerobic coverage is not needed.26 Cefotaxime, 2 g IV every 8 hours, has been shown to result in excellent ascitic fluid levels (20-fold killing power after 1 dose).81 In patients with a serum creatinine level greater than 3 mg/dL, the dosing interval may be extended to 12 hours.81 Neither a loading dose nor an intraperitoneal dose appears to be necessary or appropriate. The clinician should, however, order the first dose “STAT,” to avoid a delay in administration of the life-saving agent. More broad-spectrum antibiotics are warranted if local susceptibility testing demonstrates increasing resistance to third-generation cephalosporins.
Other Intravenous Antibiotics
Amoxicillin-clavulanic acid has been shown to be as effective as cefotaxime in a randomized trial but is not available in a parenteral formulation in the United States.82 Other antibiotics have been recommended as well but have been less well studied than has cefotaxime. Some newer drugs have been used to treat SBP (without any data on antibiotic penetration into the ascitic fluid) on the basis of their spectrum of coverage and formulary constraints.
Intravenous Albumin
Renal impairment occurs in 33% of episodes of SBP.83 SBP leads to increased intraperitoneal nitric oxide production, which in turn further increases systemic vasodilatation and promotes renal failure (see Chapter 94).84 IV albumin (1.5 g/kg of body weight at the time the infection is detected, and 1 g/kg on day 3) can increase intravascular volume and, in combination with cefotaxime, has been shown in a large randomized trial to reduce the risk of renal failure and improve survival compared with cefotaxime without albumin.85 Albumin appears to be effective by decreasing systemic vasodilatation.86 Because of the survival advantage, the use of IV albumin as an adjunct to antibiotic treatment has been recommended.87
Oral Antibiotics
Oral ofloxacin has been reported in a controlled trial to be as effective as parenteral cefotaxime in the treatment of SBP in patients who do not have vomiting, shock, bleeding, or renal failure.88 The dose studied was 400 mg twice daily.88 Another study has demonstrated the efficacy of IV ciprofloxacin, 200 mg every 12 hours for 2 days, followed by oral ciprofloxacin, 500 mg every 12 hours for 5 days.89 Because of the possibility of fluoroquinolone resistance in patients receiving fluoroquinolones to prevent SBP (see later), however, the empirical use of a fluoroquinolone to treat suspected SBP should be avoided.90 Fortunately, bacterial isolates from patients with SBP who were receiving fluoroquinolones for prophylaxis of this disorder remain susceptible to cefotaxime.78
Narrowing the Spectrum of Coverage
After the results of susceptibility testing are available, an antibiotic with a narrower spectrum of activity usually can be substituted for the broad-spectrum drug (e.g., pneumococci will usually be sensitive to penicillin, and many E. coli species will usually be sensitive to ampicillin).
Duration of Treatment
Infectious disease subspecialists generally recommend 10 to 14 days of antibiotic therapy for life-threatening infections; however, no data are available to support this duration of treatment in spontaneous ascitic fluid infections. The ascitic fluid culture becomes sterile after 1 dose of cefotaxime in 86% of patients.31 After 48 hours of therapy, the ascitic fluid PMN count is always less than the pretreatment value in patients with a spontaneous ascitic fluid infection treated with appropriate antibiotics; frequently, an 80% reduction is observed at 48 hours.31 A randomized controlled trial involving 100 patients has demonstrated that 5 days of treatment is as efficacious as 10 days in patients with SBP or CNNA.91 I have been treating SBP and CNNA for 5 days since the late 1980s, with excellent results. The average duration of oral ofloxacin treatment was 8 days in the only published trial.88
Follow-up Paracentesis
On the basis of a large database of repeat paracenteses during and after the treatment of SBP,31 a follow-up paracentesis does not appear to be needed if the setting (advanced cirrhosis with symptoms and signs of infection), bacterial isolate (monomicrobial with a characteristic organism), and response to treatment (dramatic reduction in symptoms and signs of infection) are typical.31 Paracentesis should be repeated after 48 hours of treatment if the course is atypical.31
Treatment of Ascitic Fluid Infection Other Than SBP
Because of the predictable presence of anaerobes, patients with suspected secondary peritonitis require empirical antibiotic coverage that is broader in spectrum than that used for SBP. They also require an emergency evaluation to assess the need for surgical intervention (see earlier, Box 93-4, and Fig. 93-3). Cefotaxime plus metronidazole appears to provide excellent initial empirical therapy of suspected secondary peritonitis.31
Polymicrobial bacterascites (from needle perforation of the bowel) is tolerated relatively well. Peritonitis developed in only 1 in 10 patients with a needle perforation of the intestine with spillage of intestinal contents into ascitic fluid in the one relevant study.64 The single episode of paracentesis-related peritonitis was not fatal. Patients with low-protein ascitic fluid appear to be at most risk for development of a PMN response and clinical peritonitis related to needle perforation of the intestine.64 Most of the patients with a higher ascitic protein concentration (e.g., >1 g/dL [10 g/L]) did not receive antibiotics, yet did well. Many physicians, however, probably would feel uncomfortable in withholding antibiotic treatment if needle perforation is suspected. If a decision to treat is made, anaerobic coverage should be included (e.g., cefotaxime plus metronidazole [see Table 93-5]). Whether or not treatment is begun, a follow-up paracentesis is helpful (if it can be performed safely) to monitor the ascitic fluid PMN count and culture results. If a decision is made to defer antibiotic treatment initially and the number of organisms in the ascitic fluid does not decrease or the PMN count rises in the second specimen, antibiotic treatment should then be initiated (see Table 93-5).
Prognosis
In the past, 48% to 95% of patients with a spontaneous ascitic fluid infection died during the hospitalization in which the diagnosis was made, despite antibiotic treatment.10,23 By 1991, mortality rates were low (<5% if antibiotics are administered in a timely fashion), probably because of earlier detection and treatment of infection, as well as the avoidance of nephrotoxic antibiotics.91 The trial in which cefotaxime plus albumin was studied reported an inpatient mortality rate of 10%.85 Even now, however, some patients are cured of their infection but die of liver failure or GI bleeding because of the severity of the underlying liver disease. In fact, spontaneous ascitic fluid infection is a marker of end-stage liver disease and has been proposed as an indication for liver transplantation in a patient who is otherwise a candidate.
To maximize survival, it is important that paracentesis is performed in all patients with ascites at the time of hospitalization, so that infection can be detected and treated promptly. The ascitic fluid cell count should be reviewed as soon as the results are available (≈60 minutes), and appropriate treatment should be instituted if indicated. The first dose of antibiotic should be given immediately. Because dipstick test results (see earlier) are available in 90 to 120 seconds, this tool may speed treatment of SBP and improve survival.20,22
Paracentesis should be repeated during the hospitalization if any manifestation of clinical deterioration develops, including abdominal pain, fever, change in mental status, renal failure, acidosis, peripheral leukocytosis, or GI bleeding. If the physician waits to perform a paracentesis until convincing symptoms and signs of peritoneal infection have developed, the infection is likely to be advanced by the time the diagnosis is made. No survivors of SBP have been reported when the diagnosis was made after the serum creatinine level had risen above 4 mg/dL (350 µmol/L).
Without surgical intervention, the mortality rate for secondary peritonitis in hospitalized patients with ascites approaches 100%. When secondary peritonitis is diagnosed early and treated with emergency laparotomy, the mortality rate is 50% to 67%.31,45
Prevention
The identification of risk factors for SBP (including an ascitic fluid protein concentration < 1.0 g/dL, variceal hemorrhage, and previous episode of SBP) has led to controlled trials of prophylactic antibiotics.29,92-94 Norfloxacin, 400 mg/day orally, has been reported to reduce the risk of SBP in inpatients with low-protein ascites and those with previous SBP.92,93 Norfloxacin, 400 mg orally twice daily for 7 days, helps prevent infection in patients with variceal hemorrhage94 and is cost-effective in preventing recurrent SBP.95 IV ceftriaxone 1 g daily for 7 days has been found to be even more effective than norfloxacin in the setting of GI bleeding; this regimen allows administration of antibiotics to patients who are vomiting blood.96 Oral antibiotics select for resistant organisms in the intestinal flora in patients, and in animals these organisms can then cause spontaneous ascitic fluid infection.90,97 Despite this concern, 2 randomized trials of primary prevention of ascitic fluid infection with prophylactic norfloxacin or ciprofloxacin have demonstrated a survival advantage for the antibiotic-treated patients (Table 93-6).98-100
TABLE 93-6
Prevention of SBP
| Indication | Drug | Results |
| Prior SBP | Norfloxacin 400 mg orally once daily until death or liver transplantation | 66% reduction in recurrence |
| Cirrhosis with GI hemorrhage | Norfloxacin 400 mg orally twice daily × 7 days, or | 73% reduction in infection |
| Ceftriaxone 1 g IV/day × 7 days | 67% reduction in infection compared with norfloxacin | |
Cirrhosis with ascitic fluid total protein < 1.5 g/dL and either Child-Turcotte-Pugh score ≥ 9 and total bilirubin ≥ 3 mg/dL, or Creatinine ≥ 1.2 mg/dL, or Blood urea nitrogen ≥ 25 mg/dL, or Serum sodium ≤ 130 mEq/L | Norfloxacin 400 mg/day orally × 1 year | 89% reduction in SBP 32% reduction in hepatorenal syndrome 52% increase in 3-month survival 25% increase in 1-year survival |
| Cirrhosis with ascitic fluid total protein < 1.5 g/dL | Ciprofloxacin 500 mg orally daily × 1 year | 31% reduction in infection 30% improvement in survival |

Trimethoprim/sulfamethoxazole has also been shown to prevent SBP in an animal model and in patients; in animals survival was increased.101,102 The recommended dose for patients is 1 double-strength tablet daily.102
Use of parenteral antibiotics to prevent endoscopic sclerotherapy–related or band ligation–related infections in nonbleeding patients does not appear to be warranted, as indicated by a controlled trial.103 Active bleeding, not endoscopic treatment, appears to be the risk factor for ascitic fluid infection. On the other hand, bacterial infection is associated with failure to control variceal hemorrhage.104 This observation provides additional incentive to try to prevent, detect, and treat infections aggressively in this setting to minimize mortality related not only to infection, but also to hemorrhage.
Cellulitis
Cellulitis of the lower extremities or abdominal wall is a common cause of soft tissue infection in obese patients with edema. One study has documented a 19% cumulative probability of cellulitis during hospitalization of patients with cirrhosis and ascites, compared with only a 4% likelihood of SBP.105 Risk factors for cellulitis included obesity (which is increasing in frequency in patients with cirrhosis), homelessness, and greater degree of edema.105 Another study has demonstrated a 22% risk of renal failure in patients with cellulitis and cirrhosis.106 A high index of suspicion and low threshold for treatment with a first-generation cephalosporin or other antibiotic may help decrease morbidity and mortality from uncontrolled cellulitis.
Tense Ascites
Some patients with ascites do not seek medical attention until they can no longer breathe or eat comfortably because of the pressure of the intra-abdominal fluid on the diaphragm. Tense ascites requires urgent therapeutic paracentesis (see later). Contrary to folklore, tense ascites can be drained without untoward hemodynamic effects.107 “Total paracentesis,” even more than 22 L, has been demonstrated to be safe.107 In the setting of tense ascites, therapeutic paracentesis improves venous return and hemodynamics; the myth of paracentesis-related hemodynamic catastrophes was based on anecdotal observations in small numbers of patients.
Pleural Effusions
Pleural effusions can occur in patients with cirrhosis and ascites. They usually are unilateral and right-sided but occasionally may be bilateral and larger on the right side than on the left. A unilateral left-sided effusion suggests TB. A large effusion in a patient with cirrhotic ascites is designated hepatic hydrothorax.108 Most carefully studied patients with hepatic hydrothorax have been shown to have a small defect in the right hemidiaphragm. With large diaphragmatic defects, ascites may be undetectable on clinical examination despite a large pleural effusion.
The most common symptom associated with hepatic hydrothorax is shortness of breath. Infection of the fluid can occur, usually as a result of SBP and transmission of bacteria across the diaphragm.109 The analysis of uncomplicated hepatic hydrothorax fluid is similar but not identical to that of ascitic fluid, because the pleural fluid is subject to hydrostatic pressures different from those that affect the portal bed. The total protein concentration is higher (by ≈ 1.0 g/dL [10 g/L]) in the pleural fluid than in ascitic fluid.108
The treatment of hepatic hydrothorax was difficult until the transjugular intrahepatic portosystemic shunt (TIPS) became available (see later).108 The effusions tend to occur in patients who are the least adherent to treatment regimens or in whom ascites is most refractory to therapy. Some authors have recommended chest tube insertion and sclerosing of the pleurae with tetracycline; however, chest tubes inserted to treat hepatic hydrothorax are usually difficult to remove,110 Moreover, shortness of breath may recur when the tube is clamped, and fluid may leak around the insertion site of the tube. A peritoneovenous shunt (see later) can be considered when the patient with hepatic hydrothorax has large-volume ascites, but the shunt usually clots after a short time. Direct surgical repair of the diaphragmatic defect can be considered, but the patients typically are poor operative candidates. Video thoracoscopic suture of the hole in the diaphragm followed by pleurodesis has been reported to be successful.111 Sodium restriction plus use of diuretics with intermittent thoracentesis is the safest and most effective first-line therapy of hepatic hydrothorax. TIPS placement has been reported to be successful and constitutes reasonable second-line treatment.108 If the patient is a candidate for liver transplantation, proceeding with a transplant evaluation may be the best approach.
Abdominal Wall Hernias
Abdominal wall hernias are common in patients with ascites. They usually are umbilical or incisional and occasionally inguinal. Up to 20% of patients with cirrhosis and ascites have umbilical hernias at the time of hospitalization.112 Some of these hernias incarcerate or perforate. Because of these potential complications, elective surgical treatment should be considered in a patient with a hernia and ascites. Insertion of mesh should be avoided because of the potential for the mesh to become infected. The ascitic fluid should be medically removed preoperatively because the hernia recurs in 73% of patients who have ascites at the time of hernia repair but in only 14% of those who have no ascitic fluid at the time of repair.113 Nevertheless, hernia repair is not without hazard. Successful laparoscopic repair of a recurrent strangulated umbilical hernia has been described.114 TIPS has also been reported to lead to good control of symptoms and may obviate the need for surgical repair.115 Many transplant surgeons prefer to avoid repair of the hernia or postpone it until the time of liver transplantation. An elastic abdominal binder can be used as a temporizing measure to reduce pain and hernia enlargement.
Surgical repair of a hernia or TIPS should be performed urgently in patients with skin ulceration, crusting, or black discoloration and emergently for refractory incarceration or rupture. Rupture is the most feared complication of an umbilical hernia. If TIPS is used, it must be performed prior to bacteremia. Infection of the TIPS may be difficult to eradicate.
Treatment of Ascites
Appropriate treatment of ascites depends on the cause of fluid retention. Accurate determination of the etiology of ascites is crucial. The SAAG is helpful diagnostically and for therapeutic decision making. Patients with a low SAAG usually do not have portal hypertension and do not respond to salt restriction and diuretics (except for those with nephrotic syndrome). Conversely, patients with a high SAAG have portal hypertension and are usually responsive to these measures.10
Low-Albumin-Gradient Ascites
Peritoneal carcinomatosis is the most common cause of low-albumin-gradient ascites.2 Peripheral edema in affected patients can be managed with diuretics. By contrast, patients without peripheral edema who receive diuretics lose only intravascular volume, without loss of ascitic fluid. The mainstay of treatment of nonovarian peritoneal carcinomatosis is outpatient therapeutic paracentesis. Patients with peritoneal carcinomatosis usually live only a few months. Patients with ovarian malignancy are an exception to this rule and may exhibit a good response to surgical debulking and chemotherapy.
Ascites caused by tuberculous peritonitis (without cirrhosis) is cured by anti-TB therapy. Diuretics do not speed weight loss unless the patient has underlying portal hypertension from cirrhosis. Pancreatic ascites may resolve spontaneously, require endoscopic placement of a stent in the pancreatic duct or operative intervention, or respond to treatment with somatostatin.116 A postoperative lymphatic leak from a distal splenorenal shunt or radical lymphadenectomy also may resolve spontaneously but on occasion may require surgical intervention or placement of a peritoneovenous shunt. Chlamydia peritonitis is cured by doxycycline. Ascites caused by lupus serositis may respond to glucocorticoids.7 Dialysis-related ascites may respond to aggressive dialysis.51
High-Albumin-Gradient Ascites
Cirrhosis is the most common cause of liver disease that leads to high-albumin-gradient ascites (see Table 93-1). Many patients with cirrhosis experience multiple insults to the liver, including excessive alcohol use, NASH, and chronic hepatitis C.4 One of the most important steps in treating high-albumin-gradient ascites in a patient with alcoholic liver disease, with or without other causes of liver injury, is to convince the patient to stop drinking alcohol. In a period of months, abstinence from alcohol can result in healing of the reversible component of alcoholic liver disease, and the ascites may resolve or become more responsive to medical therapy.
Baclofen has been shown in a randomized controlled trial to reduce alcohol craving and alcohol consumption, specifically in patients with alcoholic liver disease.117 It can be started at 5 mg orally 3 times daily and increased to 10 mg 3 times daily or even higher until craving is suppressed.
Similarly, patients with other forms of treatable liver disease (e.g., autoimmune hepatitis, chronic hepatitis B with reactivation, hemochromatosis, Wilson disease) should receive specific therapy for these diseases. Occasionally, cirrhosis due to causes other than alcohol or hepatitis B is reversible5; however, these diseases are usually less reversible than alcoholic liver disease, and by the time ascites is present, these patients may be better candidates for liver transplantation than for protracted medical therapy.
Hospitalization
Outpatient treatment of patients with small-volume ascites can be attempted initially. However, patients with large-volume ascites and those who are resistant to outpatient treatment frequently require hospitalization for definitive diagnosis and management.10 Many of these patients also have GI hemorrhage, encephalopathy, infection, or hepatocellular carcinoma. An intensive period of inpatient education and treatment may be required to convince the patient that the prescribed diet and diuretics are actually effective and worth the effort to prevent future hospitalizations.
Precipitating Cause
Determining the immediate precipitant of ascites (e.g., dietary indiscretion or nonadherence to therapy with diuretics) may be of value. Ascites may be precipitated by IV saline given perioperatively or to treat variceal hemorrhage or by sodium bicarbonate tablets; in such cases the ascites may resolve without the need for long-term treatment.
Diet Education
Fluid loss and weight change are related directly to sodium balance in patients with portal hypertension–related ascites. In the presence of avid renal retention of sodium, dietary sodium restriction is essential. The patient and the food preparer should be educated by a dietitian about a sodium-restricted diet. Severely sodium-restricted diets (e.g., 500 mg [22 mmol] of sodium per day) are feasible (but not palatable) in an inpatient setting but unrealistic for outpatients. The dietary sodium restriction that I recommend for both inpatients and outpatients is 2000 mg (88 mmol) per day. Protein is not restricted unless the patient has hepatic encephalopathy refractory to 2 drugs on a vegetable protein diet (see Chapter 94).
Fluid Restriction
Indiscriminate restriction of fluid in the treatment of cirrhotic ascites is inappropriate and serves only to alienate patients, nurses, and dietitians. Sodium restriction, not fluid restriction, results in weight loss; fluid follows sodium passively. The chronic hyponatremia usually seen in patients with cirrhotic ascites is seldom morbid. Attempts to correct hyponatremia rapidly in this setting can lead to more complications than those related to the hyponatremia. Severe hyponatremia (e.g., serum sodium concentration < 120 mmol/L) does warrant fluid restriction in the patient with cirrhosis and ascites but fortunately occurs in only 1.2% of patients.118 Unless the decline in sodium concentration is rapid, symptoms of hyponatremia usually do not develop in patients with cirrhosis until the serum sodium concentration is below 110 mmol/L.
Role of Bed Rest
Although bed rest has traditionally been prescribed, no controlled trials support this practice; bed rest was part of the treatment of heart failure in the past and was extrapolated to the treatment of cirrhotic ascites without data.119 An upright posture may aggravate the plasma renin elevation found in most patients with cirrhosis and ascites and, theoretically, increase renal sodium retention. In all likelihood, however, strict bed rest is unnecessary and may lead to decubitus ulcer formation in emaciated patients.
Urine Sodium Excretion
Urinary sodium excretion is a helpful parameter to follow in patients with portal hypertension–related ascites. Because urine is the most important route of excretion of sodium in the absence of diarrhea or hyperthermia, and because dietary intake is the only source of nonparenteral sodium, dietary intake and urinary excretion of sodium should be roughly equivalent, if the patient's weight is stable. Nonurinary sodium losses are less than 10 mmol/day in these patients.120 A suboptimal decline in body weight may be the result of inadequate natriuresis, failure to restrict sodium intake, or both. Monitoring 24-hour urinary sodium excretion and daily weight will clarify the issue. Patients who are adherent to an 88 mmol/day sodium diet and who excrete more than 78 mmol/day of sodium in the urine should lose weight. If the weight is increasing despite urinary losses in excess of 78 mmol/day, one can assume that the patient is consuming more sodium than is prescribed in the diet.
Urine Sodium-to-Potassium Ratio
Although 24-hour urine specimens constitute the diagnostic standard, studies have demonstrated that random urine specimens tested for sodium-to-potassium ratios correlate well with 24-hour specimens.121,122 When a random urine sodium-to-potassium ratio is greater than 1, the patient should lose fluid weight. Patients who do not lose weight despite a random urine sodium-to-potassium ratio greater than 1 probably are not adherent to the diet. Only the 10% to 15% of patients who have significant spontaneous natriuresis with urine sodium-to-potassium ratios greater than 1 can be considered for dietary sodium restriction as sole therapy of ascites (i.e., without diuretics).10 When given a choice, however, most patients would prefer to take some diuretics with more liberal intake of sodium than to take no pills with severe restriction of sodium intake.
Avoidance of Urinary Bladder Catheters
Many physicians promptly insert a bladder catheter in hospitalized patients with cirrhosis to monitor urine output accurately. Unfortunately, many of these immunocompromised patients have urinary tract infections on hospital admission,77 and urethral trauma from insertion of the catheter in the setting of cystitis can lead to bacteremia. Prolonged catheterization predictably leads to cystitis and possibly sepsis in these patients. A prospective study has confirmed the infectious consequences of these catheters.123 I insert urinary catheters only briefly and only in the ICU setting; these portals of entry for bacteria should be removed as soon as possible.
Diuretics
Spironolactone is the mainstay of treatment for patients with cirrhosis and ascites but increases natriuresis slowly. Single-agent diuretic therapy with spironolactone requires several days to induce weight loss. Although spironolactone alone has been shown to be superior to furosemide alone in the treatment of ascites,124 I prefer to start spironolactone and furosemide together on the first hospital day in initial doses of 100 mg and 40 mg, respectively, each taken once in the morning.10 Amiloride, 10 mg/day, can be substituted for spironolactone; amiloride is less widely available and more expensive than spironolactone but more rapidly effective, and it does not cause gynecomastia. A newer potassium-sparing diuretic, eplerenone, has been used in the treatment of heart failure and does not cause gynecomastia, but studies of its use in cirrhosis are lacking.
The half-life of spironolactone is approximately 24 hours in normal control subjects but is markedly prolonged in patients with cirrhosis; almost 1 month is required to reach a steady state.125 In view of its long half-life, dosing the drug multiple times per day is unnecessary. A loading dose may be appropriate but has not been studied. Single daily doses maximize adherence; 25-, 50-, and 100-mg spironolactone tablets are available generically. Furosemide also should be given once a day.126
If the combination of spironolactone, 100 mg/day (or amiloride, 10 mg/day) and furosemide, 40 mg/day orally, is ineffective in increasing urinary sodium or decreasing body weight, the doses of both drugs should be increased simultaneously as needed (e.g., spironolactone, 200 mg plus furosemide, 80 mg, then 300 mg plus 120 mg, and finally 400 mg plus 160 mg). In my experience, as well as in a randomized trial, starting both drugs at once speeds the onset of diuresis, whereas slowly increasing the daily dose of spironolactone to 400 mg or even higher before adding furosemide delays diuresis and results in hyperkalemia.127
The 100 : 40 ratio of the daily doses of spironolactone and furosemide usually maintains normokalemia. The ratio of doses can be adjusted to correct abnormal serum potassium levels. Occasionally, an alcoholic patient who has had no recent food intake will have hypokalemia at the time of admission (usually for alcoholic hepatitis) and for a variable interval thereafter. Such a patient should receive spironolactone alone until the serum potassium normalizes; furosemide can then be added. When combined with a sodium-restricted diet in a study of almost 4000 patients, the regimen of spironolactone and furosemide has been demonstrated to achieve successful diuresis in more than 90% of patients.128
IV diuretics cause acute decreases in the glomerular filtration rate in patients with cirrhosis and ascites and generally should be avoided.129 Many patients are given IV furosemide when they are hospitalized because of failure of outpatient treatment of ascites in the setting of cirrhosis. The approach of switching from oral to IV administration is effective for heart failure, but in patients with cirrhosis, repeated doses of IV furosemide regularly lead to azotemia and then to an erroneous diagnosis of hepatorenal syndrome. The correct diagnosis is diuretic-induced azotemia, which resolves when the diuretics are withheld and fluid is administered IV. Some physicians give IV albumin with IV furosemide, but a randomized crossover study has shown no benefit to albumin in this setting.130 Repeated IV dosing of furosemide appears to be too “harsh” for the patient with cirrhosis; oral diuretics are better tolerated.
If rapid weight loss is desired, therapeutic paracentesis should be performed (see later). No limit has been identified for acceptable daily weight loss in patients who have massive edema. As soon as the edema has resolved, a reasonable maximum weight loss is probably 0.5 kg/day.131 Encephalopathy, a serum sodium concentration less than 120 mmol/L despite fluid restriction, and a serum creatinine level greater than 2.0 mg/dL (180 mmol/L) are indications to discontinue diuretics and reassess the patient. Abnormalities in potassium levels almost never prohibit diuretic use, because the ratio of the 2 diuretics can be readjusted. Patients with parenchymal renal disease (e.g., diabetic nephropathy) usually require relatively higher doses of furosemide and lower doses of spironolactone; otherwise, they develop hyperkalemia. Patients in whom complications develop despite a careful attempt at diuretic treatment usually require second-line therapy. Prostaglandin inhibitors (e.g., NSAIDs) should be avoided in patients with cirrhosis and ascites because they inhibit diuresis, may promote renal failure, and may cause GI bleeding.132
Reducing the quantity of fluid in the abdomen can improve the patient's comfort and prevent hepatic hydrothorax and hernias. Also, by concentrating the ascitic fluid, diuresis increases the opsonic activity of ascitic fluid 10-fold and theoretically may be of value in preventing spontaneous ascitic fluid infection.133
Blood Pressure
An issue that nurses regularly raise is whether diuretics should be withheld when a patient's blood pressure is low. No data exist to support this practice in the setting of cirrhosis. Baseline blood pressure, mental status, and creatinine must be factored into the decision to continue, hold, or discontinue diuretics. The baseline blood pressure is usually low (e.g., 60 to 100 mm Hg systolic) in a patient with cirrhosis and ascites. Unless it has dropped rapidly or the patient has confusion or azotemia, diuretics should be given. Blood pressure correlates with survival in cirrhosis; if mean arterial pressure is 82 mm Hg or less, 2-year survival is only 20% compared with 70% for higher blood pressures.134 A mean arterial pressure of 82 mm Hg correlates with a systolic blood pressure of approximately 100 mm Hg.
Beta blockers reduce survival in patients with refractory ascites, probably by reducing blood pressure.135 Beta blockers should be stopped if blood pressure is less than 100 mg Hg systolic. Midodrine has been shown to increase blood pressure and increase survival in hypotensive patients with cirrhosis and ascites.136 Midodrine can be started at 5 mg orally 3 times daily and titrated up to achieve an increase in blood pressure of approximately 10 mm Hg, a systolic blood pressure above 100 mm Hg, or both.
Goal of Inpatient Treatment
Hospitalized patients who are stable, with ascites as their major problem, can be discharged after they are demonstrated to be responding to the medical regimen and are normokalemic, are not azotemic, and have a normal or slightly to moderately reduced serum sodium level. In the past, patients with ascites frequently occupied hospital beds for prolonged durations because of uncertainty regarding the diagnosis and optimal treatment and because of iatrogenic complications. Complete resolution of ascites should not be a prerequisite for discharge from the hospital. Following discharge from the hospital, a patient should be seen in the outpatient setting within 7 to 14 days.
Role of Sodium Bicarbonate
Mild renal tubular acidosis develops in some patients with cirrhosis and ascites. Although oral sodium bicarbonate administration has been recommended in this setting, such treatment increases sodium intake dramatically and cannot be advocated in the absence of evidence to support its use.
Vaptans
The vaptans are a relatively new class of drugs that have been used in patients with cirrhosis to increase urinary water excretion.137,138 Initially the focus of treatment with vaptans was an increase in serum sodium137; however, this is not a very important end point. Additionally, the largest randomized trial that included only patients with cirrhosis demonstrated that the vaptan was not clinically beneficial in long-term management of ascites in cirrhosis. Even more concerning was an increase in mortality in the vaptan-treated group.138
Outpatient Management
After discharge from the hospital, the patient's body weight, blood pressure at home (using a prescribed blood pressure cuff), orthostatic symptoms, and serum electrolyte, urea, and creatinine levels should be monitored. Random urine specimens for a sodium-to-potassium ratio can be collected to help guide treatment decisions. The subsequent frequency of follow-up evaluations is determined by the response to treatment and stability of the patient. I usually evaluate these patients every 1 to 4 weeks until they clearly are responding to treatment and are not experiencing problems. Intensive outpatient follow-up helps prevent subsequent hospitalizations.
Diuretic doses and dietary sodium intake are adjusted to achieve weight loss and negative sodium balance. Patients who are gaining fluid weight despite diuretic therapy should not be considered to have diuretic-resistant ascites (see later) until they are demonstrated to be adherent to the prescribed diet. Monitoring the urine sodium concentration provides insight into adherence. Patients who have a random urine sodium-to-potassium ratio greater than 1 should be losing weight if they are consuming less than 88 mmol of sodium per day. In my experience, most patients who initially are thought to be diuretic-resistant eventually are found to be nonadherent to the diet; they demonstrate weight gain and urinary sodium excretion as high as 500 mmol/day or more. Diet education is crucial to the successful management of such patients. Patients with truly diuretic-resistant ascites excrete nearly sodium-free urine despite maximal doses of diuretics. During long-term follow-up, abstinent alcoholic patients may become more sensitive to diuretics. In these cases, the dose of diuretics may be tapered and the drugs even discontinued.
Refractory Ascites
Refractory ascites is defined as ascites unresponsive to a sodium-restricted diet and high-dose diuretic treatment. Refractoriness may manifest as minimal or no weight loss despite diuretics or the development of complications of diuretics.139 Studies have shown that ascites in the setting of cirrhosis is refractory to standard medical therapy in fewer than 10% of patients.124,128
Viable options for patients refractory to routine medical therapy include careful attention to medications, liver transplantation, serial therapeutic paracenteses, TIPS, and peritoneovenous shunts (Fig. 93-5).10
Careful Attention to Medications
Ascites can be made refractory by drugs that reduce blood pressure. These include beta blockers, acetylcholinesterase inhibitors, and angiotensin receptor blockers (see Fig. 93-5).135 The risks versus benefits of each of these drugs must be considered carefully. If a patient's systolic blood pressure is below 100 mm Hg and azotemia is present, the beta blocker, acetylcholinesterase inhibitor, or angiotensin receptor blocker should be stopped. If ascites continues to be refractory to diuretics, midodrine can be added and carefully titrated upward to achieve the desired blood pressure and natriuresis. These simple steps can cause diuretic-resistant patients to become diuretic-sensitive.
Liver Transplantation
Liver transplantation should be considered among the treatment options for patients with cirrhosis and ascites—whether the fluid is diuretic-sensitive or diuretic-refractory (see Chapter 97). In many areas of the United States, patients with ascites are not offered transplantation until hepatorenal syndrome has developed (see Chapter 94). The 12-month survival rate for patients with ascites refractory to medical therapy is only 32%.140 The survival rate following liver transplantation is much higher.
In patients who are candidates for liver transplantation, procedures that could make a future transplant difficult should be avoided. Surgery in the right upper quadrant causes adhesions that become vascularized and difficult to remove during transplant surgery.
Serial Paracenteses
Therapeutic abdominal paracentesis is one of the oldest medical procedures. In the 1980s, after 2000 years of use, scientific data regarding large-volume paracentesis were reported, and patients were documented to tolerate large-volume paracentesis well, just as patients had in the 1940s and earlier.141 In one large randomized controlled trial, therapeutic paracentesis plus IV infusion of colloid led to fewer minor (asymptomatic) changes in serum electrolyte and creatinine levels than those reported with diuretic therapy; however, no differences in morbidity or mortality rates could be demonstrated.141 Therapeutic paracentesis now appears to be first-line therapy for patients in whom ascites is tense and second-line therapy for patients with cirrhosis whose ascites is refractory to diuretics (see Fig. 93-5).10 The world record for volume of fluid removed at one time appears to be 41 L.142
Colloid Replacement
A controversial issue regarding therapeutic paracentesis is the role of colloid replacement. In one study, patients with tense ascites were randomized to receive IV albumin (10 g/L of fluid removed) or no albumin after therapeutic paracentesis.143 More statistically significant (asymptomatic) changes in serum electrolyte, plasma renin, and serum creatinine levels developed in the patients who did not receive albumin than in those who received albumin, but no greater frequency of clinical morbidity or mortality was seen. Although another study has documented that the patients who have a postparacentesis rise in plasma renin levels have a decreased life expectancy compared with those who have stable renin levels, no study has demonstrated decreased survival rate in patients not given a plasma expander compared with patients given albumin after paracentesis.144 The phrase “paracentesis-induced circulatory dysfunction” has been coined to describe the rise in plasma renin levels after paracentesis.145
The confusion regarding albumin infusion relates, in part, to the design of the relevant studies. In the studies from Barcelona, patients with “tense” ascites could be entered into the trial of albumin versus no albumin, and 31% of these patients were not even receiving diuretics.142 It seems more appropriate to study the population in which repeated paracenteses are really needed, specifically the diuretic-resistant group, rather than all patients with tense ascites.146 Another group of investigators has shown that patients with cirrhosis and diuretic-resistant ascites tolerate a 5-L paracentesis without a change in plasma renin levels.147 My approach to patients with tense ascites is to take off enough fluid (4 to 5 L) to relieve intra-abdominal pressure and then to rely on diuretics to eliminate the remainder. To remove all of the fluid by paracentesis when most of it can be removed with diuretics seems inappropriate, partly because paracentesis removes opsonins, whereas diuresis concentrates opsonins.133 Repeated therapeutic paracenteses should be reserved for the 10% of patients in whom diuretic treatment fails to relieve the ascites.
A meta-analysis has reported a survival advantage with albumin infusion after large-volume paracenteses.148 Therefore, it seems reasonable to (1) avoid serial large-volume paracenteses in patients with diuretic-sensitive ascites, (2) withhold albumin after taps of 5 L or less, and (3) consider albumin infusion (6 to 8 g/L of fluid removed) after taps of larger volume in patients with diuretic-resistant ascites.10,148
A small randomized trial has shown that terlipressin may be equivalent to albumin after therapeutic paracentesis in preventing paracentesis-induced circulatory dysfunction; when this drug receives approval for use in the United States, and if further studies support its efficacy, terlipressin would be an alternative to albumin.149
Transjugular Intrahepatic Portosystemic Shunt (TIPS)
TIPS is a side-to-side portacaval shunt that is placed by an interventional radiologist (or hepatologist), usually with the use of local anesthesia. TIPS placement was first used for the treatment of refractory variceal bleeding, but it also has been advocated for diuretic-resistant ascites150 (see Chapter 92). TIPS was received with great enthusiasm in the 1990s, similar to the enthusiasm for the peritoneovenous shunt in the 1970s. As with peritoneovenous shunting, TIPS was overused until serious complications and suboptimal efficacy were reported. Four large-scale randomized trials in patients with diuretic-resistant ascites have demonstrated consistently superior efficacy of TIPS over repeated paracenteses, but no survival advantage.151-154 Several meta-analyses have confirmed the efficacy of TIPS but with a higher frequency of hepatic encephalopathy.155-159 One meta-analysis has demonstrated a trend toward improved survival in patients undergoing TIPS placement.156 Another meta-analysis, which analyzed individual patient data, did show improved transplant-free survival with TIPS.159 Although TIPS dysfunction was common when an uncoated (or uncovered) stent was used, polytetrafluoroethylene-coated stents have been reported to improve patency and survival when compared with uncoated stents in a nonrandomized study and to improve patency, with no survival advantage, when compared with uncoated stents in a randomized trial.160,161 Also, the 4 older TIPS trials preceded development and implementation of the MELD score, which predicts 90-day mortality after TIPS placement (see Chapter 92). Trials using the coated stent and selecting patients according to their MELD scores may demonstrate a survival advantage for TIPS compared with repeated paracenteses.
TIPS also is useful in the treatment of hepatic hydrothorax and umbilical hernia.116,126 A direct intrahepatic portosystemic shunt connects the portal vein directly to the inferior vena cava and has applicability in patients with Budd-Chiari syndrome (see Chapter 85).162
Peritoneovenous Shunt
In the mid-1970s, the peritoneovenous shunt was promoted as a new “physiologic” treatment for the management of ascites. Reports of shunt failure, fatal complications following shunt insertion, and randomized trials demonstrating no survival advantage have led to the relegation of this procedure to third-line therapy in patients with cirrhosis and ascites10,128 (see Fig. 93-5). Patients who are not candidates for liver transplantation and who have a scarred abdomen that is not amenable to repeated paracenteses, who are not candidates for a TIPS, or in whom an attempt at TIPS placement has failed make up the small subset of candidates for a peritoneovenous shunt. A randomized trial has shown that even an uncoated TIPS stent has better “assisted patency” than a peritoneovenous shunt.163
Novel Treatments
Novel treatment options for patients with refractory ascites include weekly infusions of IV albumin, ascites reinfusion, ultrafiltration, terlipressin infusion (not available in the United States), partial splenic artery embolization, peritoneal-urinary drainage of the fluid using a surgically implanted pump, and percutaneous placement of a peritoneovenous shunt by an interventional radiologist.164-171 More data are needed before these treatments can be recommended.
Prognosis
Cirrhosis complicated by ascites is associated with significant morbidity and mortality, related, in part, to the severe underlying liver disease and, in part, to the ascites per se. In half of the patients in whom cirrhosis is detected before decompensation (i.e., development of ascites, jaundice, encephalopathy, or GI hemorrhage), ascites occurs within 10 years.172 When ascites appears, the expected mortality rate is approximately 50% in just 2 years.173 With liver transplantation, survival is improved dramatically.
