Epistaxis
Daniel B. Simmen, Nicholas S. Jones
Key Points
▪ Epistaxis is the most common otolaryngologic emergency.
▪ The most common etiology is idiopathic, followed by traumatic, iatrogenic, coagulopathic, and neoplastic.
▪ Management ranges from emergency replacement of blood loss, direct visualization and cautery, nasal packing, and surgical-endoscopic or external embolization.
▪ Localizing the site of bleeding is of the utmost importance.
▪ Avoid nasal packing if possible; it causes trauma to the nasal mucosa and makes it difficult to find the bleeding site.
▪ A primary aim is to occlude the vessel near its bleeding point.
▪ Terminal branches of the external and internal carotid arteries supply the mucosa of the nasal cavity with many anastomoses between these systems.
▪ Various anastomoses exist on the ipsilateral side and cross over to the contralateral side.
▪ The basic approach to treatment is to locate the site of bleeding, stop the bleeding, and treat the cause.
▪ The majority of posterior idiopathic bleeds are from the septum, usually from the septal branch of the sphenopalatine artery.
▪ Endoscopic bipolar suction diathermy treats most epistaxes.
▪ If a bleeding point cannot be found, ideally the nose is packed with an absorbable hemostatic agent that produces minimal mucosal trauma.
▪ Endoscopic sphenopalatine artery ligation has replaced the need for posterior nasal packs, other than in an emergency situation to control profuse bleeding.
▪ The branching pattern of the sphenopalatine artery is complex, but the most common are two or three branches medial to the crista ethmoidalis, but sometimes there are even more.
▪ Persistent posterior epistaxis can be controlled by percutaneous embolization.
Epistaxis is the most common otolaryngologic emergency and affects up to 60% of the population in their lifetime, and 6% require medical attention.1 It has been estimated that nosebleeds affect 108 per 100,000 population per year.2 In England and Wales, an average of 10.2 per 100,000 patients are admitted for an average stay of 2.9 days in a 3-month period,3 and in the United States, 17 per 100,000 (6%) are admitted.4 Peaks in incidence are seen in those under 10 years of age and in people aged over 40 years.5,6 Women of menstrual age have fewer hospital admissions with epistaxis, which may be due to estrogens that provide protection of the nasal vasculature.7 The etiology of epistaxis in the majority of patients is idiopathic,8 followed by primary neoplasms and traumatic or iatrogenic causes.
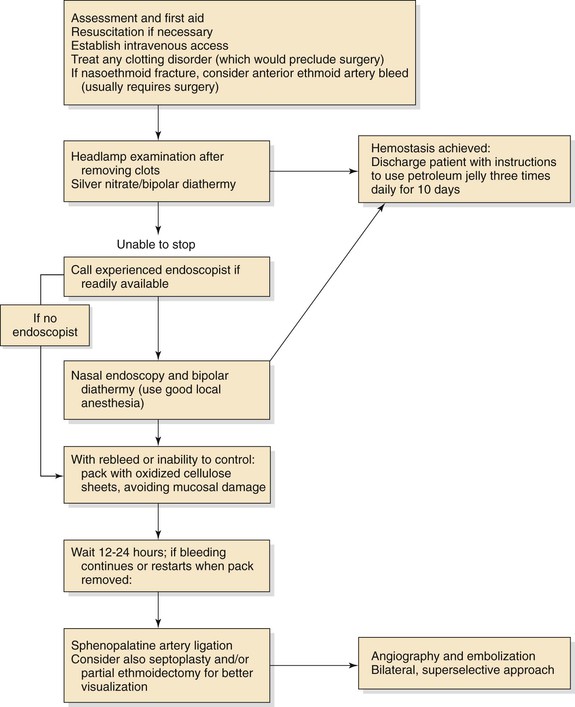
Decisions about the optimum therapeutic intervention and timing are often made on an ad hoc basis, and most units do not have a protocol (systematic algorithm) for the management of epistaxis9 despite some recently published guidelines and reviews.10-12 However, this protocol does not contain the timing of surgical intervention. The management of a patient with epistaxis ranges from the replacement of blood loss, direct visualization and cautery, nasal packing, and surgical (endoscopic or external) embolization. Recently, McGarry13 separated anterior epistaxis, bleeding from a source anterior to the plane of the piriform aperture, from posterior epistaxis, bleeding posterior to this plane. Further subdivision includes epistaxis from the lateral wall, septum, or nasal floor.13
Vascular Anatomy
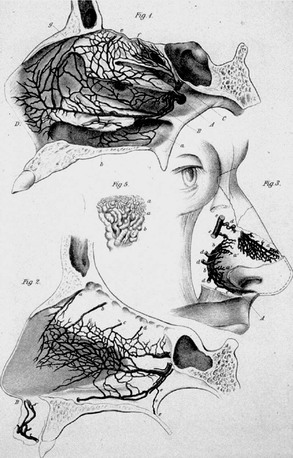
The nasal cavity is extremely vascular. Terminal branches of the external and internal carotid arteries supply the mucosa of the nasal cavity with frequent anastomoses between these systems (Fig. 42-1). The anterior nasal septum is the site of a plexus of vessels called the Kiesselbach area, or Little area, which is supplied by both systems.
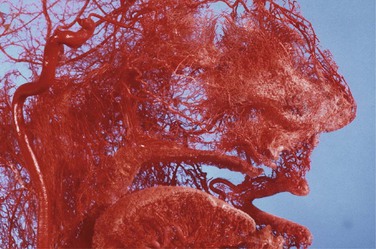
The terminal branches of the external carotid artery that supply the nasal cavity are the facial artery and the internal maxillary artery. The facial artery supplies the superior labial artery, which enters the nose and supplies the anterior nasal septum. The internal maxillary artery courses within the pterygopalatine fossa, and it terminates in the sphenopalatine, descending palatine, pharyngeal, infraorbital, and posterior-superior alveolar arteries. The branching of the sphenopalatine artery when entering the nasal cavity is a key point in the understanding of the management of posterior nosebleeds. The sphenopalatine artery enters the nasal cavity through the sphenopalatine foramen, where it then divides into conchal (posterolateral) and septal (posteromedial) branches.14,15 The descending palatine artery courses through the greater palatine canal and becomes the greater palatine artery, entering the nose through the incisive foramen to supply the anterior inferior septum, where it anastomoses with medial branches of the sphenopalatine artery. Importantly, the vidian artery shares an important anastomosis between the internal carotid artery and a branch of the sphenopalatine artery and therefore to the external carotid system (Fig. 42-2).
The internal carotid artery supplies the nasal mucosa via the ethmoid branches of the ophthalmic artery, and the ophthalmic artery is the first branch of the internal carotid artery. The posterior ethmoid artery passes through the posterior ethmoid canal into the anterior cranial fossa and divides into lateral and medial branches that supply the superior part of the posterior septum and lateral nasal wall. The anterior ethmoid artery enters the nasal cavity through the anterior ethmoid canal and passes anteromedially to the area of the anterior skull base (Fig. 42-3). There it crosses the anterior ethmoid roof to reach the fovea ethmoidalis and cribriform plate, and a nasal branch supplies the anterior superior part of the septum and its other branch, the anterior meningeal artery, which enters intracranially.
A fundamental aspect in the understanding of the vascular anatomy and its importance to epistaxis is the fact that various anastomoses on the ipsilateral side between the internal and external carotid systems exist, as does a crossover to the contralateral side (Fig. 42-4). The rich anastomoses that exist underpin the importance of a strategy to address the most distal site of any bleeding.
The maxillary sinus ostium serves as the dividing line between anterior and posterior epistaxis. Anterior bleeding is usually easier to access and is therefore less dangerous. Posterior epistaxis is more problematic to treat, because visualization is more difficult, and blood is often swallowed, which makes it more difficult to assess the amount of blood lost.
The term posterior bleeding is all too often used incorrectly to label bleeding that cannot be visualized with a headlamp. This is frequently incorrect because it often transpires after endoscopic examination and in many such patients, the bleeding is located high on the septum.
Management
Initial Assessment
The amount of blood loss should be estimated. One way is to ask patients whether they have lost enough blood to soak a handkerchief, facecloth, or towel—the latter would indicate a significant loss—and over what period (a minor but regular bleed can cause anemia). A clinical assessment of cardiac status and circulating blood volume should include looking to see if the patient is pale, sweating, has marked vasoconstriction or is cool, or has tachycardia; all of these would indicate significant hypovolemia. A reduction in blood pressure is often a late sign, particularly in young people who can maintain blood pressure until circulatory volume becomes critical. A transfusion threshold of 9 g/dL has been advocated, because this has been shown to improve outcome.16
Obtaining intravenous access, checking and correcting any clotting abnormalities, and taking blood to find their blood group and save blood for future crossmatching that may be needed for transfusion may be required. In our unit, patients admitted via the emergency department can be fast-tracked to the ENT emergency if stable (Box 42-1). This helps avoid unnecessary and counterproductive nasal packing in the emergency department and avoids patients being transferred before they are fit enough to travel.
First-aid measures include asking the patient to apply constant, firm pressure over the lower (nonbony) part of the nose for 20 minutes and to lean forward with the mouth open over a bowl so that further blood loss can be estimated. Otherwise, blood dripping postnasally will be swallowed, and the next warning sign could be several hundred milliliters of blood being vomited.
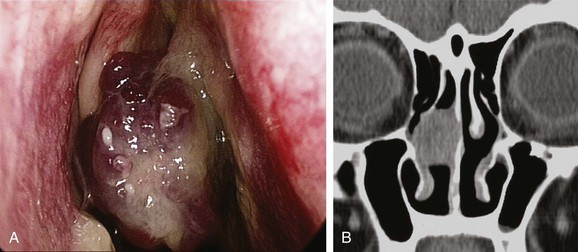
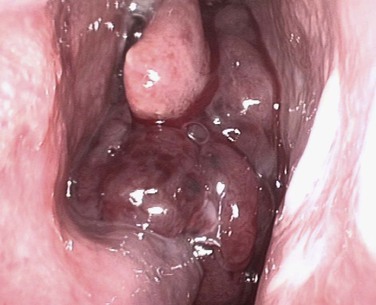
It is important to establish both the site and the cause of bleeding (Box 42-2), so the bleeding can be stopped and the cause treated. It must also be remembered that epistaxis is frequently idiopathic; however, it can be a manifestation of a possible underlying pathology (Figs. 42-5 through 42-12). The patient should undergo further investigation according to the history.
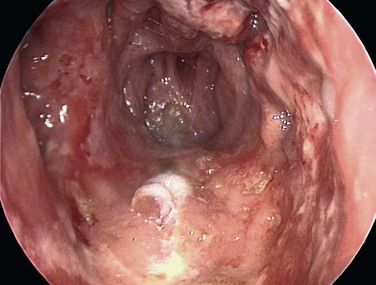
Headlamp Examination with Local Anesthesia
The key to controlling most epistaxes is to find the site of the bleeding, and whereas chemical cautery with silver nitrate can be used, bipolar diathermy is more effective. Protection from blood contamination is important. A plastic apron for both parties is helpful to avoid clothes being stained, and eye protection is advisable if there is active bleeding, because some patients reflexively blow away any fluid dripping down their upper lip, and this can create a bloody aerosol. Once the clots have been evacuated, the nasal airway should be inspected, initially with a headlamp; if the bleeding point cannot be located, inspection with an endoscope is warranted.
Epistaxis in Children
Young children usually bleed from a vessel just inside the nose at the mucocutaneous junction on the septum, and this invariably stops spontaneously. In children with epistaxis, when no prominent vessel can be seen, the regular local application of a cream can help,20 whereas petroleum jelly alone does not help.21 A Cochrane Collaboration systematic analysis of the efficacy of topical treatments for idiopathic epistaxis in children concluded that the best treatment still needs to be defined.22 As many as 5% to 10% of children with recurrent nosebleeds may have undiagnosed von Willebrand disease.23,24 One study advocated the use of β-blockers in children with recurrent primary epistaxis resistant to conventional management, and this was given 1.5 to 2 mg/kg/day, divided into three doses, as a second-line therapy for terminating epistaxis.25 This therapy, however, cannot be recommended until a prospective and randomized study can support this therapy. Children with leukemia and those receiving chemotherapy often have epistaxis associated with thrombocytopenia. Older children, adolescents, and adults often bleed from the Kiesselbach area or from a maxillary spur.
Epistaxis in Adults
The caudal end of the septum is where several branches of the external and internal carotid anastomose, in the Kiesselbach area, and it is the most common site of bleeding.26 Less commonly, bleeding comes from further back on the septum, and a septal deviation may make it difficult to visualize (Fig 42-13). Some patients with seasonal allergic rhinitis complain of more nosebleeds in the hay fever season, and topical nasal steroids aggravate the bleeding in approximately 4% of users. Many people believe that a nosebleed signifies a release of pressure and may herald a stroke, and it is important to address these anxieties. Although many patients are hypertensive when they have their nosebleeds, few remain so on follow-up, and the association between hypertension and epistaxis is disputed.27 Many clinicians report that epistaxis is not related to hypertension.28-30 However, patients with hypertension tend to have nosebleeds that are more likely to lead to admission and to be associated with comorbidity.31
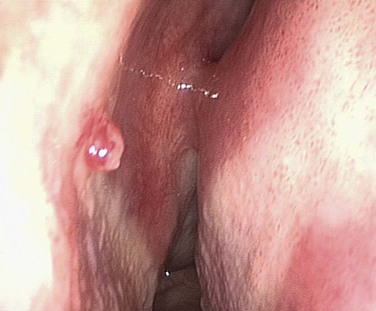
Bleeding disorders can come to medical attention de novo with nosebleeds, although this is rare. A range of drugs have been linked with epistaxis, and warfarin is one of the most common causes.3,32 Nearly a third of patients admitted with epistaxis who were taking warfarin had an international normalized ratio above the upper limit of the therapeutic range.33 The need to reverse anticoagulation is uncertain, as long as the patient falls within the therapeutic range.34 In overanticoagulated patients, fresh frozen plasma, clotting factor extracts, and vitamin K help. Vitamin K takes more than 6 hours to work, and it can delay anticoagulation for 7 days after warfarin is started. If the international normalized ratio is greater than 4, the warfarin should be stopped, and fresh frozen plasma should be given.35 Clotting factor extracts should be given with extreme caution because of the risk of thromboembolic complications. Tranexamic acid, an antifibrinolytic agent, has not been shown to help.36 Other drugs associated with bleeding include aspirin, which interferes with platelet function for up to 7 days, clopidogrel, and nonsteroidal antiinflammatory drugs.37 In patients without a history of a bleeding disorder or anticoagulant therapy, routine clotting studies do not add to the management.38 A higher incidence of epistaxis is seen in patients with a high alcohol intake, even in the absence of any evidence of abnormality when testing coagulation.39 Indirect markers of alcohol overuse include the mean corpuscular volume, γ-glutamyltransferase, aspartate aminotransferase, and alanine aminotransferase.40 Inquiry about drinking habits should be made, and help should be offered to affected patients.
Topical Treatment
A randomized controlled trial of silver nitrate cautery and Naseptin, a proprietary cream that contains neomycin, versus Naseptin alone showed both to be effective.41 A study of patients who applied weekly triamcinolone 0.025% and daily petroleum jelly resulted in 89% of patients having no further bleeding.42 Various hemostatic compounds have been used but without consistent evidence of their efficacy. Collagen-derived particles with bovine-derived thrombin have been found to be better than nasal packs.43 Use of topical bovine-derived gelatin granules and human thrombin in anterior epistaxis has been reported, but cost limits its use, and more studies of its efficacy are needed.
Cautery
Most anterior epistaxis can be controlled with identification of the bleeding point using a headlamp and cautery. The vast majority of posterior bleeding sites can be identified by endoscopy without general anesthesia.44 The majority of posterior idiopathic bleeds are from the septum, usually from the septal branch of the sphenopalatine artery as it runs submucosally (Fig. 42-14), although some report bleeding from the lateral aspect of the middle or inferior meatus or from the posterior end of the turbinates (Fig 42-15).45 When the site of bleeding cannot clearly be identified with a headlamp, the use of a rigid nasal endoscope by an experienced endoscopist is best. The key is to identify the site of the bleeding and gain control using silver nitrate cautery or bipolar suction diathermy. Recent studies have shown that the use of endoscopic bipolar diathermy treats most epistaxis.46,47 Preparing the nose with phenylephrine or cocaine for its decongestant and anesthetic effects often helps. It is worth noting that phenylephrine has a significant decongestant effect at 6 minutes and maximum anesthetic effect after 9 minutes,48 and time must be allowed for it to take effect. An injection of local anesthetic and epinephrine gives better analgesia, if there is a sizable vessel that needs bipolar diathermy; otherwise, the patient may feel a smarting sensation.
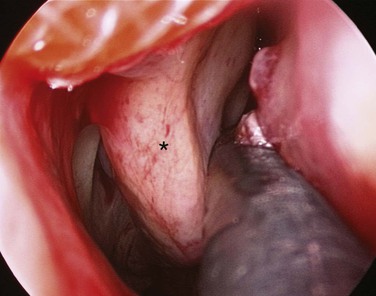
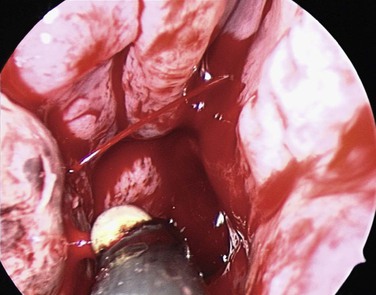
To stop the bleeding, either bipolar forceps or unipolar suction diathermy can be used. Some of the bipolar suction forceps now available help to remove the blood and smoke at the same time. The bulbous head of the unipolar suction device allows cautery of a sizable vessel anywhere in the nose, and its large suction channel is less likely to become blocked by congealed blood. Simultaneous bilateral cautery should be avoided to minimize the risk of causing a septal perforation. After cautery, patients should be advised against nose blowing for about 10 days to allow the area to heal. A greasy antiseptic barrier cream should be applied several times daily for 2 weeks to prevent the eschar from drying and coming off, with a resulting rebleed. The ointment should not be placed directly on the area treated but is best placed inside the rim of the nostril on the tip of the finger and then “milked” upward by massaging the nostril rims, and then it can be sniffed up. This advice can also be given to patients with a crusted septal area from picking or excessive drying. Whereas most nosebleeds come from the Little area—or in young children, from the mucocutaneous junction—a small proportion of bleeds come from higher up on the septum or more posteriorly. Endoscopy is helpful in defining the bleeding point. If the nose is packed before endoscopy is done, this can complicate detection of the exact site of bleeding, because this often causes mucosal trauma and misleads the clinician.
Rarely, nasal tumors can come to medical attention with epistaxis or a serosanguinous discharge, and it is important to see whether a nasal mass is present, especially beyond a septal deviation. If crusting is evident on the septum, it is worth asking whether this has been a problem. Crusting can follow an abrasion, picking, or vasculitis, such as Wegener granulomatosis.49 If it looks likely that picking or blowing any crust off is perpetuating the problem, it is usually not helpful to confront the patient about this. Instead, if the clinician casually mentions that nose picking is very common (“you see people doing it in cars or waiting for a train, and many people do it when nobody is watching”), and says that dried crusts are irritating, and then asks if they have to remove the crusts, the patient may confess to doing so, and some progress can be made.
If the bleeding point is not identified, gently packing the nose with fine sheets of oxidized cellulose (Surgicel Fibrillar; Ethicon, Blue Ash, OH) avoids mucosal damage and promotes coagulation to control bleeding.
The majority of posterior idiopathic bleeds are from the septum, usually from the septal branch of the sphenopalatine artery as it runs submucosally. The key is to identify the site of the bleeding and to gain control using bipolar suction diathermy. A severe septal deviation can make it difficult to define the bleeding point.49 Control of the bleeding avoids the discomfort associated with nasal packing and avoids admission.50 A cost-benefit analysis of 38 adult patients with epistaxis concluded that £6804 (about $10,260 U.S. dollars) could be saved by avoiding admission in 28 patients.46 Endoscopic cauterization achieves hemostasis in over 80% of patients with posterior epistaxis at the first attempt and over 90% after a second attempt.38 The bleeding point can be cauterized with the help of a nasal endoscope, which has a reported failure rate of 17% to 33%.51,52 Complications associated with this procedure are uncommon, but isolated reports describe palatal numbness from thermal damage to the greater palatine nerve, damage to the lacrimal duct, and possible damage to the optic nerve when cautery is used in a patient who has already had an ethmoidectomy.53 If by some misfortune bleeding is torrential because of trauma to the internal carotid artery in the sphenoid, prompt packing of the sphenoid sinus is required with a firm gauze pack.54 The patient is then resuscitated, and the help of an interventional radiologist is sought.
Nasal Packing
If a bleeding point cannot be found using a headlamp, ideally, an experienced endoscopic surgeon should try and locate the bleeding point. However, if nobody is available who has these skills, the nose is packed with an absorbable hemostatic agent that produces minimal mucosal trauma. Various nonabsorbable packs have been used, but their insertion is uncomfortable, as is their presence once in position; insertion can cause local mucosal trauma, which can complicate localization of the bleeding point.
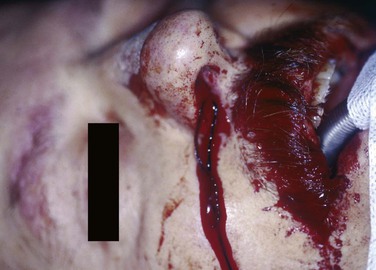
If an anterior pack fails, and no experienced surgeon is available who can examine, localize, and cauterize the bleeding point, more packing to tamponade the bleeding point may be required. This requires good local anesthesia and analgesia to enable adequate pressure to be applied with the packs in position. Various proprietary packs are available. The insertion of a nasal pack has conventionally meant that the patient has to be admitted, although one study discharged 46 of 62 patients whose nasal airway was packed, and follow-up was arranged on an outpatient basis after 48 hours. However, 28% of those discharged had unplanned returns.55 If anterior packing fails, a posterior balloon may need to be placed and inflated in the postnasal space. An anterior pack is then placed, and then gentle traction is used to pull the balloon forward against the anterior pack; this is held in place by a clip over the catheter anteriorly, as it emerges through the anterior pack.56 It is vital that any clip used to secure the catheter does not rest on the skin of the nostril, because this can produce necrosis of the area in as little as 4 hours (Fig. 42-16). Posterior packs were required in 9% of patients in one series.57
The morbidity and physical discomfort associated with nasal packing includes marked pain, hypoxia, alar necrosis, and toxemia and is well described in the literature.53,58-60 Unfortunately, nasal packing is used routinely in some departments, where expertise in nasal endoscopy is not available, and only 7% of units in the United Kingdom have suction diathermy available.9 Packing not only traumatizes the nasal lining, it can also cause cardiorespiratory complications and local infection.58
The role of prophylactic systemic antibiotics in patients who have nasal packs is not well established, and wide variations are apparent in current practice in England.54 The main concern is to avoid toxic shock syndrome. We would prescribe antibiotics if the patient has any cardiac anomalies that would require antibiotic prophylaxis for surgery. One study concluded that systemic prophylactic antibiotics are unnecessary in the majority of epistaxis patients who have nasal packs.61 The use of topical antibiotics may be more appropriate, cheaper, and just as effective.
If the patient does not rebleed within 12 to 24 hours, the packs should be removed. The nose should be inspected with a rigid endoscope, if this has not already been done prior to packing, to exclude any pathology that may have been responsible for the bleeding. Posterior epistaxis controlled by posterior nasal packing has a failure rate between 26% and 52% and a complication rate between 2% and 68.8%.61,62 The complications noticed were synechiae, angina, periorbital cellulitis, sinusitis, toxic shock syndrome, hypoxia, and otitis media.62,63
It has been stated, “Gone are the days when a patient had an uncomfortable posterior nasal pack inserted then spent several days on the ward only to bleed again on its removal.”64 Endoscopic sphenopalatine artery ligation (ESPAL) has replaced the need for posterior nasal packs, other than in emergency situations, to control profuse bleeding.
The goal is to achieve a high success rate and low morbidity. Elderly patients with multiple medical problems—arteriosclerosis, hypertension, diabetes, hepatic and renal disease—tolerate packing especially poorly, and complications occur frequently; therefore the physician should consider an early surgical intervention, rather than packing the nose, in this population.60
Maxillary Artery Ligation
Another technique to control posterior epistaxis is by ligation of the internal maxillary artery in the pterygopalatine fossa.65 This has a reported success rate of approximately 90%,66 and treatment failures are due to difficulty finding the artery and its branches. This technique is also associated with a complication rate of 28%.66 Because this procedure is done through a Caldwell-Luc approach, complications include rhinosinusitis, facial pain, oroantral fistula, and facial and dental paresthesias, and dissection in the pterygopalatine fossa can result in blindness, ophthalmoplegia, and decreased lacrimation.67
Ligation of the External Carotid Artery
Ligation of the external carotid artery has been advocated, but the rich anastomoses of vessels in the nose makes it somewhat ineffective.67 Ligation of the external carotid artery prevents embolization, which may be desirable because of bleeding in the distribution of that vessel, but this area is also supplied by cross-anastomoses from contralateral vessels; for this reason, ligation of the external carotid artery is best avoided. Cerebrovascular ischemia and infarction has been reported following external carotid ligation in elderly atherosclerotic patients, whose cerebral circulation has partially relied on anastamotic connections from the external to the internal carotid system.
The Role of Endoscopic Sphenopalatine Artery Ligation
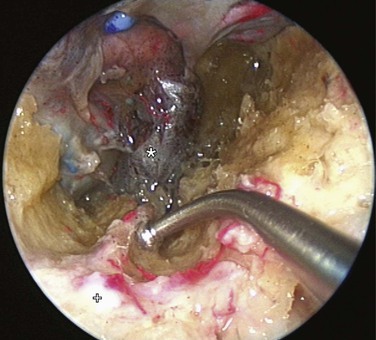
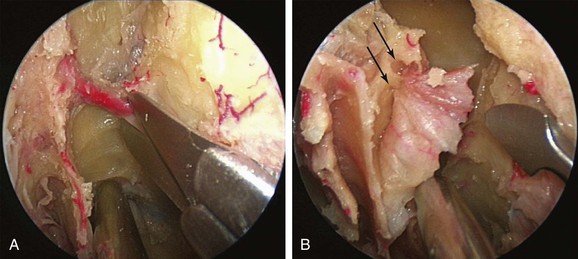
If bleeding cannot be controlled after endoscopic examination and cautery and/or nasal packing, sedation and examination under a local or general anesthetic are indicated. Bipolar diathermy of any bleeding points or ESPAL is then the treatment of choice.10 Clipping or diathermy of the sphenopalatine artery is currently the accepted treatment for management of persistent posterior epistaxis,50,62,68-72 because it produces less morbidity compared with embolization or ligation of the maxillary or external carotid artery.73,74 The main complication in 25% of patients was nasal crusting. Pooled case series data have shown that 98% of patients had epistaxis controlled by clipping or diathermy of the sphenopalatine artery.75 The endoscopic sinus surgeon should locate the sphenopalatine artery at the level of the ethmoid crest (Fig. 42-17).75 An incision is made over the posterior fontanelle, a submucosal flap is lifted, and the anterior branch is identified with its origin just posterior to the crista ethmoidalis. The anterior branch is then identified and is either clipped or diathermied (Fig. 42-18). In the majority of cases, the sphenopalatine foramen opens into the middle and superior meatus.
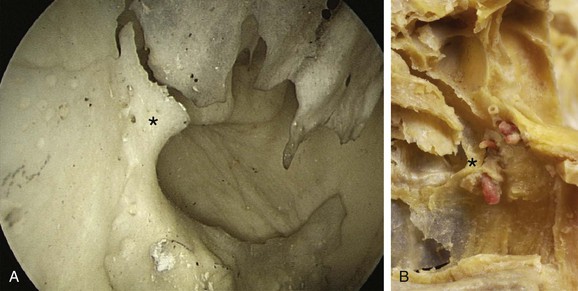
The sphenopalatine artery normally starts to branch lateral to the ethmoid crest, and these branches vary widely. It is important that the surgeon who undertakes ligation or cautery of the artery is aware that over 97% of individuals have two or more branches medial to the ethmoid crest, 67% have three or more branches, and 35% have four or more branches. The endoscopic sinus surgeon views the sphenopalatine artery at the level of the sphenopalatine foramen or a few millimeters medial to it. Making a mucosal incision in the posterior fontanelle area, anterior to the horizontal part of the base of the middle turbinate as it joins the lateral nasal wall, a submucosal flap is lifted, and the artery is identified posterior to the crista ethmoidalis and is clipped. Some authors describe identifying the artery and clipping it, but they did not dissect more to verify whether any other branches were present.68,71,72 It may be because of the mistaken belief that the artery enters as a single trunk into the nose, which has led to failure of these procedures. One study specifically looked at the branching pattern of this artery from the endoscopic surgeon's point of view.51,76 They found two branches of the sphenopalatine artery near the sphenopalatine foramen. In their study, 16% branched within the foramen and reached the nose together; in 42% the branches had an anteroposterior relationship, with the septal branch being posterior to the posterior nasal branch, and in the remaining 42%, the septal branch exited through a separate foramen posterior to the sphenopalatine foramen.51
In a study by Simmen and colleagues,76 they found that over 97% of specimens had two or more branches medial to the crista ethmoidalis, 30% had three branches, and in one specimen, 10 branches were identified. The variation in 77 specimens is illustrated in Figure 42-19.
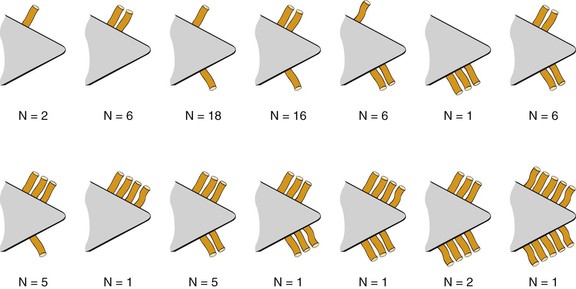
Various reports have claimed the success rate of ESPAL to be between 92% and 100% in controlling epistaxis with this technique.69,71,75 Failure to clip all the branches of the sphenopalatine artery may be the reason for continued epistaxis after the procedure. All divisions of the sphenopalatine artery occur in the pterygomaxillary fissure and enter the nasal fossa as separate blood vessels. The arrangement of these branches in relation to the sphenopalatine foramen varies widely. The largest and most anteriorly based branch is the posterior lateral nasal branch, which must be clipped and divided so that the flap can be lifted superiorly to locate the nasopalatine branch, which must be clipped and divided as well. Because such wide variations are found in the branching pattern of the sphenopalatine artery, careful dissection is required even after these two branches are clipped. A thorough search by careful dissection should be done all around the sphenopalatine foramen to find any other branches that can be clipped. Clipping or diathermy of the sphenopalatine artery has a 0% to 8% failure rate75 and is not associated with any serious complications. The main complication, as previously mentioned, is failure to control epistaxis, and it is usually due to failure to clip all the branches of the sphenopalatine artery. The arrangements of these branches are unpredictable, as seen in our study, and can vary between sides in the same individual. In addition, the branches of this artery can come out through separate foramina, and if the surgeon is not aware of this, it can result in missing a few branches. Other complications are uncommon and include nasal crusting, palatal numbness, acute sinusitis, decreased lacrimation, and septal perforation.71 A long-term study of ESPAL showed a 93% success rate.77 We do not advocate packing and repacking for refractory epistaxis if this can be avoided.
Embolization
Arterial embolization has been shown to be effective in the treatment of intractable epistaxis.78-80 However, the procedure carries a risk of complications, which include cerebrovascular accident, hemiplegia, ophthalmoplegia, facial nerve palsy, seizures, and soft tissue necrosis.81-84 However, an analysis of 31 cases of embolization of the internal maxillary artery by Siniluoto and colleagues85 did not come across any major, persistent complications. Because interventional neuroradiology is increasingly available, embolization has become an option when initial treatment fails. It is also important to state at this point that the arterial embolization technique is mainly effective for the external carotid artery supply; it is extremely dangerous for the internal carotid artery supply. Embolization for the terminal branches of the ophthalmic artery is therefore not advocated because of the high risk of causing blindness as a result of a reflux of embolic materials.86
We would advocate the use of this arterial embolization technique for patients with refractory epistaxis who are unfit for surgery or in whom surgery has failed to control the bleeding (Fig. 42-20). Persistent posterior epistaxis can be controlled by percutaneous embolization of bleeding arteries. The success rate for this procedure is between 71% and 95% with a complication rate of 27%.50,65,85
Bleeding from the Anterior Ethmoid Artery
An uncommon cause of severe epistaxis is associated with a nasoethmoid fracture, in which case the bleed originates from the anterior ethmoid artery. These patients form a distinct subgroup, whose management differs in that they require surgery to the anterior ethmoid artery, either via a traditional external approach or via an endoscopic approach; this is because the bleeding rarely subsides with conservative measures (Fig. 42-21).87
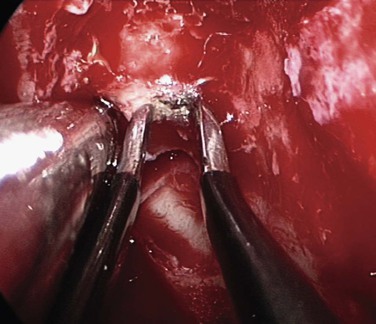
The anterior ethmoid artery can also be damaged during sinus surgery. It is at risk as it passes through the roof of the ethmoid sinuses on its way from the orbit to the anterior cranial fossa.88 Damage to the artery can lead to profuse epistaxis, intraorbital bleeding, and at times intracranial bleeding.89 The development of a retroorbital hemorrhage is an emergency, because it can lead to blindness. The bony canal in which the anterior ethmoid artery crosses the skull base can be dehiscent, making it vulnerable to damage.90 A dehiscence of the anterior ethmoid artery of 11% to 40% has been reported.91 Bony protrusions from the lateral end of the bony canal of the artery are apparent, both where it emerges from the orbit and medially, where it goes through the lateral lamella. Some authors have stated that the artery lies behind the frontal recess and can act as a landmark when approaching this area.92,93 Stammberger90 located the anterior ethmoid artery 1 to 2 mm behind the junction of the posterior wall of the frontal recess and anterior ethmoid air cells. Simmen and colleagues94 showed a mean distance from the posterior wall of the frontal recess to the artery of 11 mm (range 6 to 15 mm). In most cases the position of the artery is mirrored on both sides. However, it is variable and, consequently, it is not safe to use the artery as a landmark for any endoscopic intervention, especially in locating the frontal recess. The anterior ethmoid artery is always seen between the second and third lamella, and the most common site to find the artery is in the suprabullar recess (85%). If pneumatization of the ethmoid is marked, the artery is likely to be lying below the skull base, where it is more prone to surgical damage. Preoperative knowledge of suprabullar recess and supraorbital cells by careful interpretation of CT scans is helpful in avoiding damage to the anterior ethmoid artery (Fig. 42-22).
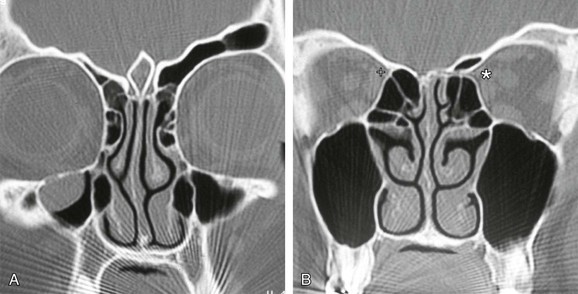
It should be emphasized that it is usually unnecessary, and indeed undesirable, to locate the anterior ethmoid artery at surgery during most procedures, because this will increase the risk of damaging it.
Hot Water Irrigation for Epistaxis
Hot water irrigation as a noninvasive treatment for posterior epistaxis has received some renewed attention.95,96,98 In the nineteenth century, obstetricians used hot water to irrigate a postpartum bleeding wound, and Guice97 introduced this procedure for stanching blood flow in cases of epistaxis in 1879. Sven-Eric Stangerup's98 studies involving experiments with rabbits showed that irrigating the nose at 40° to 46° C (104° to 114.8° F) does not lead to any histologic changes of the mucosa. Higher temperatures result in vasodilation and, in particular, in edema of the mucosa. Only temperatures above 52° C (125.6° F) trigger necrosis. It can be assumed that the temperature-induced mucosal edema leads to local compression of the bleeding vessel; at the same time, it may propagate the cascade for hemostasis.95,98
After an examination with nasal endoscopy, topical anesthesia of the relevant nasal cavity is administered using 4% tetracaine. Stangerup's modified bladder catheter is then inserted into the affected nasal cavity, the balloon is inflated in the epipharynx, and the catheter is gently pulled back, until the choana is blocked off. The affected nasal cavity is irrigated continuously with tap water at a temperature of 50° C (122° F), until the bleeding decreases. The treatment lasts approximately 3 minutes, with the patient sitting upright, so that the irrigation water can flow forward from the nose into a basin. After irrigation with 500 mL of hot water, the catheter can be removed. In a prospective study that included 84 patients, Schlegel and associates95 showed a success rate of 82% without any complications. This technique avoids painful packing, hospitalization, or immediate surgery and also allows the patient to breathe normally through an open nasal cavity after the treatment.
Management of Epistaxis in Patients with Hereditary Hemorrhagic Telangiectasia
Hereditary hemorrhagic telangiectasia (HHT) is an autosomal-dominant multisystemic disorder characterized by telangiectasia that affects cutaneous and mucosal surfaces and arteriovenous malformations in the pulmonary, cerebral, and hepatic circulations (Fig. 42-23). The most common symptom in this population is epistaxis, which affects more than 90% of individuals. The severity of attacks is variable, and episodes of epistaxis can be functionally and socially debilitating for the patient and may require frequent hospital admission; in addition, a set of patients exists for whom the bleeding is intractable.99 Many methods of treatment for epistaxis as a result of HHT have been described in the literature; these include medical strategies, such as the use of hormonal manipulation and antifibrinolytic agents, as well as surgical options, which include laser coagulation (Fig. 42-24),100 septodermoplasty,101 and nasal closure,102 but no one method has proved itself entirely successful or without significant side effects.
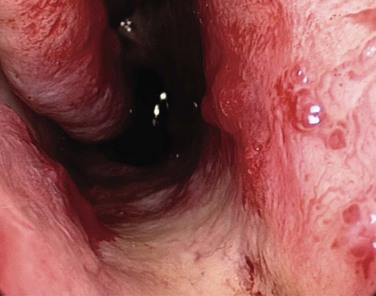
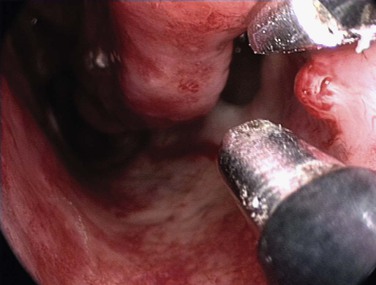
A recent prospective study by Hitchings and colleagues99 investigated the effect of various surgical options on quality of life scores in these patients. The authors have concluded that nasal closure should be offered to patients with moderate to severe epistaxis that has proved unresponsive to other treatment options (bipolar or laser coagulation treatment, septodermoplasty), and subjectively 88% (7/8) of patients treated with nasal closure reported a complete cessation of their nosebleeds. Closure of the nasal cavity, the Young procedure, is based on the principle that the absence of desiccating airflow through the nasal cavity prevents the initiation of mucosal breakdown overlying the fragile telangiectasias.102 An alternative to nasal closure is the use of nasal obturators.103 In recent years, the monoclonal antibody bevacizumab has been advocated in HHT for intractable epistaxis, with use both topically104 and by local injection.105-107 In addition, some evidence supports the use of tamoxifen to reduce the need for blood transfusions.108
Summary
The role of rigid nasal endoscopy as part of the initial assessment of patients who come to medical attention with epistaxis, with direct visualization and control of the bleeding point, is effective in the majority of patients and reduces the need for nasal packing. Endoscopic sphenopalatine artery ligation is well established as the treatment of choice, where cautery and nasal packing fails. An algorithm for the management of epistaxis is shown in Figure 42-25.