Resistance of the Body to Infection
II. Immunity and Allergy
The human body has the ability to resist almost all types of organisms or toxins that tend to damage the tissues and organs. This capability is called immunity. Much of immunity is acquired immunity that does not develop until after the body is first attacked by a bacterium, virus, or toxin; often weeks or months are required for the immunity to develop. An additional element of immunity that results from general processes, rather than from processes directed at specific disease organisms, is called innate immunity. It includes the following aspects:
1. Phagocytosis of bacteria and other invaders by white blood cells and cells of the tissue macrophage system, as described in Chapter 34.
2. Destruction of swallowed organisms by the acid secretions of the stomach and the digestive enzymes.
3. Resistance of the skin to invasion by organisms.
4. Presence in the blood of certain chemicals and cells that attach to foreign organisms or toxins and destroy them. Some of these are (1) lysozyme, a mucolytic polysaccharide that attacks bacteria and causes them to dissolute; (2) basic polypeptides, which react with and inactivate certain types of gram-positive bacteria; (3) the complement complex that is described later, a system of about 20 proteins that can be activated in various ways to destroy bacteria; and (4) natural killer lymphocytes that can recognize and destroy foreign cells, tumor cells, and even some infected cells.
This innate immunity makes the human body resistant to such diseases as some paralytic viral infections of animals, hog cholera, cattle plague, and distemper—a viral disease that kills a large percentage of dogs that become afflicted with it. Conversely, many animals are resistant or even immune to many human diseases, such as poliomyelitis, mumps, human cholera, measles, and syphilis, which are very damaging or even lethal to human beings.
Acquired (Adaptive) Immunity
In addition to its generalized innate immunity, the human body has the ability to develop extremely powerful specific immunity against individual invading agents such as lethal bacteria, viruses, toxins, and even foreign tissues from other animals. This ability is called acquired or adaptive immunity. Acquired immunity is caused by a special immune system that forms antibodies and/or activated lymphocytes that attack and destroy the specific invading organism or toxin. It is with this acquired immunity mechanism and some of its associated reactions, especially the allergies, that this chapter is concerned.
Acquired immunity can often bestow an extreme degree of protection. For instance, certain toxins, such as the paralytic botulinum toxin or the tetanizing toxin of tetanus, can be protected against in doses as high as 100,000 times the amount that would be lethal without immunity. It is for this reason that the treatment process known as immunization is so important in protecting human beings against disease and against toxins, as explained later in this chapter.
Basic Types of Acquired Immunity—Humoral and Cell Mediated
Two basic but closely allied types of acquired immunity occur in the body. In one of these, the body develops circulating antibodies, which are globulin molecules in the blood plasma capable of attacking the invading agent. This type of immunity is called humoral immunity or B-cell immunity (because B lymphocytes produce the antibodies). The second type of acquired immunity is achieved through formation of large numbers of activated T lymphocytes that are specifically crafted in the lymph nodes to destroy the foreign agent. This type of immunity is called cell-mediated immunity or T-cell immunity (because the activated lymphocytes are T lymphocytes). We shall see shortly that both the antibodies and the activated lymphocytes are formed in the lymphoid tissues of the body. Let us discuss the initiation of the immune process by antigens.
Both Types of Acquired Immunity are Initiated by Antigens
Because acquired immunity does not develop until after invasion by a foreign organism or toxin, it is clear that the body must have some mechanism for recognizing this invasion. Each toxin or each type of organism almost always contains one or more specific chemical compounds in its makeup that are different from all other compounds. In general, these are proteins or large polysaccharides, and it is they that initiate the acquired immunity. These substances are called antigens (antibody generators).
For a substance to be antigenic, it usually must have a high molecular weight of 8000 or greater. Furthermore, the process of antigenicity usually depends on regularly recurring molecular groups, called epitopes, on the surface of the large molecule. This factor also explains why proteins and large polysaccharides are almost always antigenic, because both of these substances have this stereochemical characteristic.
Lymphocytes are Responsible for Acquired Immunity
Acquired immunity is the product of the body's lymphocytes. In people who have a genetic lack of lymphocytes or whose lymphocytes have been destroyed by radiation or chemicals, no acquired immunity can develop. Within days after birth, such a person dies of fulminating bacterial infection unless he or she is treated by heroic measures. Therefore, it is clear that the lymphocytes are essential to the survival of the human being.
The lymphocytes are located most extensively in the lymph nodes, but they are also found in special lymphoid tissues such as the spleen, submucosal areas of the gastrointestinal tract, thymus, and bone marrow. The lymphoid tissue is distributed advantageously in the body to intercept invading organisms or toxins before they can spread too widely.
In most instances, the invading agent first enters the tissue fluids and then is carried by lymph vessels to the lymph node or other lymphoid tissue. For instance, the lymphoid tissue of the gastrointestinal walls is exposed immediately to antigens invading from the gut. The lymphoid tissue of the throat and pharynx (the tonsils and adenoids) is well located to intercept antigens that enter by way of the upper respiratory tract. The lymphoid tissue in the lymph nodes is exposed to antigens that invade the peripheral tissues of the body, and the lymphoid tissue of the spleen, thymus, and bone marrow plays the specific role of intercepting antigenic agents that have succeeded in reaching the circulating blood.
T and B Lymphocytes Promote “Cell-Mediated” Immunity or “Humoral” Immunity.
Although most lymphocytes in normal lymphoid tissue look alike when studied under a microscope, these cells are distinctly divided into two major populations. One of the populations, the T lymphocytes, is responsible for forming the activated lymphocytes that provide “cell-mediated” immunity, and the other population, the B lymphocytes, is responsible for forming antibodies that provide “humoral” immunity.
Both types of lymphocytes are derived originally in the embryo from pluripotent hematopoietic stem cells that form common lymphoid progenitor cells as one of their most important offspring as they differentiate. Almost all of the lymphocytes that are formed eventually end up in the lymphoid tissue, but before doing so, they are further differentiated or “preprocessed” in the following ways.
The lymphoid progenitor cells that are destined to eventually form activated T lymphocytes first migrate to and are preprocessed in the thymus gland, and thus they are called “T” lymphocytes to designate the role of the thymus. They are responsible for cell-mediated immunity.
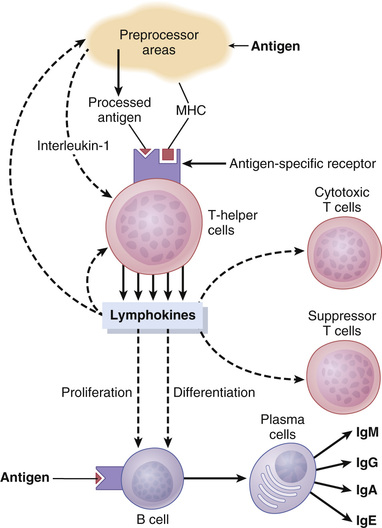
The other population of lymphocytes—the B lymphocytes that are destined to form antibodies—are preprocessed in the liver during mid fetal life and in the bone marrow in late fetal life and after birth. This population of cells was first discovered in birds, which have a special preprocessing organ called the bursa of Fabricius. For this reason, these lymphocytes are called “B” lymphocytes to designate the role of the bursa, and they are responsible for humoral immunity. Figure 35-1 shows the two lymphocyte systems for the formation, respectively, of (1) the activated T lymphocytes and (2) the antibodies.
Preprocessing of the T and B Lymphocytes
Although all lymphocytes in the body originate from lymphocyte-committed stem cells of the embryo, these stem cells are incapable of forming directly either activated T lymphocytes or antibodies. Before they can do so, they must be further differentiated in appropriate processing areas as follows.
The Thymus Gland Preprocesses the T Lymphocytes.
The T lymphocytes, after origination in the bone marrow, first migrate to the thymus gland. Here they divide rapidly and at the same time develop extreme diversity for reacting against different specific antigens. That is, one thymic lymphocyte develops specific reactivity against one antigen, and then the next lymphocyte develops specificity against another antigen. This process continues until there are thousands of different types of thymic lymphocytes with specific reactivities against many thousands of different antigens. These different types of preprocessed T lymphocytes now leave the thymus and spread by way of the blood throughout the body to lodge in lymphoid tissue everywhere.
The thymus also makes certain that any T lymphocytes leaving the thymus will not react against proteins or other antigens that are present in the body's own tissues; otherwise, the T lymphocytes would be lethal to the person's own body in only a few days. The thymus selects which T lymphocytes will be released by first mixing them with virtually all the specific “self-antigens” from the body's own tissues. If a T lymphocyte reacts, it is destroyed and phagocytized instead of being released, which happens to up to 90 percent of the cells. Thus, the only cells that are finally released are those that are nonreactive against the body's own antigens—they react only against antigens from an outside source, such as from a bacterium, a toxin, or even transplanted tissue from another person.
Most of the preprocessing of T lymphocytes in the thymus occurs shortly before birth of a baby and for a few months after birth. Beyond this period, removal of the thymus gland diminishes (but does not eliminate) the T-lymphocytic immune system. However, removal of the thymus several months before birth can prevent development of all cell-mediated immunity. Because this cellular type of immunity is mainly responsible for rejection of transplanted organs, such as hearts and kidneys, one can transplant organs with much less likelihood of rejection if the thymus is removed from an animal a reasonable time before its birth.
Liver and Bone Marrow Preprocess the B Lymphocytes.
In humans, B lymphocytes are preprocessed in the liver during mid fetal life and in the bone marrow during late fetal life and after birth. B lymphocytes are different from T lymphocytes in two ways: First, instead of the whole cell developing reactivity against the antigen, as occurs for the T lymphocytes, the B lymphocytes actively secrete antibodies that are the reactive agents. These agents are large proteins that are capable of combining with and destroying the antigenic substance, which is explained elsewhere in this chapter and in Chapter 34. Second, the B lymphocytes have even greater diversity than the T lymphocytes, thus forming many millions of types of B-lymphocyte antibodies with different specific reactivities. After preprocessing, the B lymphocytes, like the T lymphocytes, migrate to lymphoid tissue throughout the body, where they lodge near but slightly removed from the T-lymphocyte areas.
T Lymphocytes and B-Lymphocyte Antibodies React Highly Specifically Against Specific Antigens—Role of Lymphocyte Clones
When specific antigens come in contact with T and B lymphocytes in the lymphoid tissue, certain of the T lymphocytes become activated to form activated T cells, and certain of the B lymphocytes become activated to form antibodies. The activated T cells and antibodies, in turn, react highly specifically against the particular types of antigens that initiated their development. The mechanism of this specificity is the following.
Millions of Specific Types of Lymphocytes Are Stored in the Lymphoid Tissue.
Millions of different types of preformed B lymphocytes and preformed T lymphocytes capable of forming highly specific types of antibodies or T cells have been stored in the lymph tissue, as explained earlier. Each of these preformed lymphocytes is capable of forming only one type of antibody or one type of T cell with a single type of specificity, and only the specific type of antigen can activate it. Once the specific lymphocyte is activated by its antigen, it reproduces wildly, forming tremendous numbers of duplicate lymphocytes (Figure 35-2). If it is a B lymphocyte, its progeny will eventually secrete the specific type of antibody that then circulates throughout the body. If it is a T lymphocyte, its progeny are specific sensitized T cells that are released into the lymph and then carried to the blood and circulated through all the tissue fluids and back into the lymph, sometimes circulating around and around in this circuit for months or years.
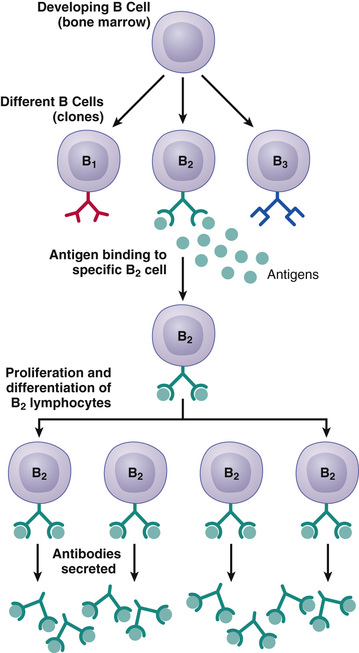
All the different lymphocytes that are capable of forming one specific antibody or T cell are called a clone of lymphocytes. That is, the lymphocytes in each clone are alike and are derived originally from one or a few early lymphocytes of its specific type.
Origin of the Many Clones of Lymphocytes
Only several hundred to a few thousand genes code for the millions of different types of antibodies and T lymphocytes. At first, it was a mystery how it was possible for so few genes to code for the millions of different specificities of antibodies or T cells that can be produced by the lymphoid tissue. This mystery has now been solved.
The whole gene for forming each type of T cell or B cell is never present in the original stem cells from which the functional immune cells are formed. Instead, there are only “gene segments”—actually, hundreds of such segments—but not whole genes. During preprocessing of the respective T- and B-cell lymphocytes, these gene segments become mixed with one another in random combinations, in this way finally forming whole genes.
Because there are several hundred types of gene segments, as well as millions of different combinations in which the segments can be arranged in single cells, one can understand the millions of different cell gene types that can occur. For each functional T or B lymphocyte that is finally formed, the gene structure codes for only a single antigen specificity. These mature cells then become the highly specific T and B cells that spread to and populate the lymphoid tissue.
Mechanism for Activating a Clone of Lymphocytes
Each clone of lymphocytes is responsive to only a single type of antigen (or to several similar antigens that have almost exactly the same stereochemical characteristics). The reason for this is the following: In the case of the B lymphocytes, each of these has on its cell surface membrane about 100,000 antibody molecules that will react highly specifically with only one type of antigen. Therefore, when the appropriate antigen comes along, it immediately attaches to the antibody in the cell membrane; this leads to the activation process, which is described in more detail subsequently. In the case of the T lymphocytes, molecules similar to antibodies, called surface receptor proteins (or T-cell markers), are on the surface of the T-cell membrane, and these are also highly specific for one specified activating antigen. An antigen therefore stimulates only those cells that have complementary receptors for the antigen and are already committed to respond to it.
Role of Macrophages in the Activation Process.
Aside from the lymphocytes in lymphoid tissue, literally millions of macrophages are also present in the same tissue. These macrophages line the sinusoids of the lymph nodes, spleen, and other lymphoid tissue, and they lie in apposition to many of the lymph node lymphocytes. Most invading organisms are first phagocytized and partially digested by the macrophages, and the antigenic products are liberated into the macrophage cytosol. The macrophages then pass these antigens by cell-to-cell contact directly to the lymphocytes, thus leading to activation of the specified lymphocytic clones. The macrophages, in addition, secrete a special activating substance, interleukin-1, that promotes still further growth and reproduction of the specific lymphocytes.
Role of the T Cells in Activation of the B Lymphocytes.
Most antigens activate both T lymphocytes and B lymphocytes at the same time. Some of the T cells that are formed, called T-helper cells, secrete specific substances (collectively called lymphokines) that activate the specific B lymphocytes. Indeed, without the aid of these T-helper cells, the quantity of antibodies formed by the B lymphocytes is usually slight. We discuss this cooperative relationship between helper T cells and B cells after describing the mechanisms of the T-cell system of immunity.
Specific Attributes of the B-Lymphocyte System—Humoral Immunity and the Antibodies
Formation of Antibodies by Plasma Cells.
Before exposure to a specific antigen, the clones of B lymphocytes remain dormant in the lymphoid tissue. Upon entry of a foreign antigen, macrophages in lymphoid tissue phagocytize the antigen and then present it to adjacent B lymphocytes. In addition, the antigen is presented to T cells at the same time, and activated T-helper cells are formed. These helper cells also contribute to extreme activation of the B lymphocytes, as discussed later.
The B lymphocytes specific for the antigen immediately enlarge and take on the appearance of lymphoblasts. Some of the lymphoblasts further differentiate to form plasmablasts, which are precursors of plasma cells. In the plasmablasts, the cytoplasm expands and the rough endoplasmic reticulum vastly proliferates. The plasmablasts then begin to divide at a rate of about once every 10 hours for about nine divisions, giving in 4 days a total population of about 500 cells for each original plasmablast. The mature plasma cell then produces gamma globulin antibodies at an extremely rapid rate—about 2000 molecules per second for each plasma cell. In turn, the antibodies are secreted into the lymph and carried to the circulating blood. This process continues for several days or weeks until finally exhaustion and death of the plasma cells occur.
Formation of “Memory” Cells Enhances the Antibody Response to Subsequent Antigen Exposure.
A few of the lymphoblasts formed by activation of a clone of B lymphocytes do not go on to form plasma cells but instead form moderate numbers of new B lymphocytes similar to those of the original clone. In other words, the B-cell population of the specifically activated clone becomes greatly enhanced, and the new B lymphocytes are added to the original lymphocytes of the same clone. They also circulate throughout the body to populate all the lymphoid tissue; immunologically, however, they remain dormant until activated once again by a new quantity of the same antigen. These lymphocytes are called memory cells. Subsequent exposure to the same antigen will cause a much more rapid and much more potent antibody response this second time around, because there are many more memory cells than there were original B lymphocytes of the specific clone.
Figure 35-3 shows the differences between the primary response for forming antibodies that occurs on first exposure to a specific antigen and the secondary response that occurs after second exposure to the same antigen. Note the 1-week delay in the appearance of the primary response, its weak potency, and its short life. The secondary response, by contrast, begins rapidly after exposure to the antigen (often within hours), is far more potent, and forms antibodies for many months rather than for only a few weeks. The increased potency and duration of the secondary response explain why immunization is usually accomplished by injecting antigen in multiple doses with periods of several weeks or several months between injections.
Nature of the Antibodies
Antibodies are gamma globulins called immunoglobulins (Ig) that have molecular weights between 160,000 and 970,000 and constitute about 20 percent of all the plasma proteins.
All the immunoglobulins are composed of combinations of light and heavy polypeptide chains. Most are a combination of two light and two heavy chains, as shown in Figure 35-4. However, some of the immunoglobulins have combinations of as many as 10 heavy and 10 light chains, which give rise to high-molecular-weight immunoglobulins. Yet, in all immunoglobulins, each heavy chain is paralleled by a light chain at one of its ends, thus forming a heavy-light pair, and there are always at least 2 and as many as 10 such pairs in each immunoglobulin molecule.
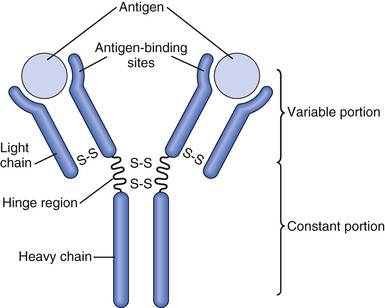
Figure 35-4 shows a designated end of each light and heavy chain, called the variable portion; the remainder of each chain is called the constant portion. The variable portion is different for each specific antibody, and it is this portion that attaches specifically to a particular type of antigen. The constant portion of the antibody determines other properties of the antibody, establishing such factors as antibody diffusivity in the tissues, adherence to specific structures within the tissues, attachment to the complement complex, ease with which the antibodies pass through membranes, and other biological properties of the antibody. A combination of noncovalent and covalent bonds (disulfide) holds the light and heavy chains together.
Specificity of Antibodies.
Each antibody is specific for a particular antigen; this characteristic is caused by its unique structural organization of amino acids in the variable portions of both the light and heavy chains. The amino acid organization has a different steric shape for each antigen specificity, so when an antigen comes in contact with it, multiple prosthetic groups of the antigen fit as a mirror image with those of the antibody, thus allowing rapid and tight bonding between the antibody and the antigen. When the antibody is highly specific, there are so many bonding sites that the antibody-antigen coupling is exceedingly strong, held together by (1) hydrophobic bonding, (2) hydrogen bonding, (3) ionic attractions, and (4) van der Waals forces. It also obeys the thermodynamic mass action law.

Ka is called the affinity constant and is a measure of how tightly the antibody binds with the antigen.
Note especially in Figure 35-4 that there are two variable sites on the illustrated antibody for attachment of antigens, making this type of antibody bivalent. A small proportion of the antibodies, which consist of combinations of up to 10 light and 10 heavy chains, have as many as 10 binding sites.
Five General Classes of Antibodies.
There are five general classes of antibodies, respectively named IgM, IgG, IgA, IgD, and IgE. “Ig” stands for immunoglobulin, and the other five respective letters designate the respective classes.
For the purpose of our present limited discussion, two of these classes of antibodies are of particular importance: IgG, which is a bivalent antibody and constitutes about 75 percent of the antibodies of the normal person, and IgE, which constitutes only a small percentage of the antibodies but is especially involved in allergy. The IgM class is also interesting because a large share of the antibodies formed during the primary response are of this type. These antibodies have 10 binding sites that make them exceedingly effective in protecting the body against invaders, even though there are not many IgM antibodies.
Mechanisms of Action of Antibodies
Antibodies act mainly in two ways to protect the body against invading agents: (1) by direct attack on the invader and (2) by activation of the “complement system” that then has multiple means of its own for destroying the invader.
Direct Action of Antibodies on Invading Agents.
Figure 35-5 shows antibodies (designated by the red Y-shaped bars) reacting with antigens (designated by the shaded objects). Because of the bivalent nature of the antibodies and the multiple antigen sites on most invading agents, the antibodies can inactivate the invading agent in one of several ways, as follows:
1. Agglutination, in which multiple large particles with antigens on their surfaces, such as bacteria or red cells, are bound together into a clump
2. Precipitation, in which the molecular complex of soluble antigen (such as tetanus toxin) and antibody becomes so large that it is rendered insoluble and precipitates
3. Neutralization, in which the antibodies cover the toxic sites of the antigenic agent
4. Lysis, in which some potent antibodies are occasionally capable of directly attacking membranes of cellular agents and thereby cause rupture of the agent
These direct actions of antibodies often are not strong enough to play a major role in protecting the body against the invader. Most of the protection comes through the amplifying effects of the complement system described next.
Complement System for Antibody Action
“Complement” is a collective term that describes a system of about 20 proteins, many of which are enzyme precursors. The principal actors in this system are 11 proteins designated C1 through C9, B, and D, shown in Figure 35-6. All these are present normally among the plasma proteins in the blood, as well as among the proteins that leak out of the capillaries into the tissue spaces. The enzyme precursors are normally inactive, but they can be activated by the so-called classical pathway.
Classical Pathway.
The classical pathway is initiated by an antigen-antibody reaction. That is, when an antibody binds with an antigen, a specific reactive site on the “constant” portion of the antibody becomes uncovered, or “activated,” and this in turn binds directly with the C1 molecule of the complement system, setting into motion a “cascade” of sequential reactions, shown in Figure 35-6, beginning with activation of the proenzyme C1. The C1 enzymes that are formed then activate successively increasing quantities of enzymes in the later stages of the system so that from a small beginning, an extremely large “amplified” reaction occurs. Multiple end products are formed, as shown to the right in the figure, and several of these cause important effects that help to prevent damage to the body's tissues caused by the invading organism or toxin. Among the more important effects are the following:
1. Opsonization and phagocytosis. One of the products of the complement cascade, C3b, strongly activates phagocytosis by both neutrophils and macrophages, causing these cells to engulf the bacteria to which the antigen-antibody complexes are attached. This process is called opsonization. It often enhances the number of bacteria that can be destroyed by many hundredfold.
2. Lysis. One of the most important of all the products of the complement cascade is the lytic complex, which is a combination of multiple complement factors and is designated C5b6789. This has a direct effect of rupturing the cell membranes of bacteria or other invading organisms.
3. Agglutination. The complement products also change the surfaces of the invading organisms, causing them to adhere to one another, thus promoting agglutination.
4. Neutralization of viruses. The complement enzymes and other complement products can attack the structures of some viruses and thereby render them nonvirulent.
5. Chemotaxis. Fragment C5a initiates chemotaxis of neutrophils and macrophages, thus causing large numbers of these phagocytes to migrate into the tissue area adjacent to the antigenic agent.
6. Activation of mast cells and basophils. Fragments C3a, C4a, and C5a activate mast cells and basophils, causing them to release histamine, heparin, and several other substances into the local fluids. These substances in turn cause increased local blood flow, increased leakage of fluid and plasma protein into the tissue, and other local tissue reactions that help inactivate or immobilize the antigenic agent. The same factors play a major role in inflammation (which was discussed in Chapter 34) and in allergy, as we discuss later.
7. Inflammatory effects. In addition to inflammatory effects caused by activation of the mast cells and basophils, several other complement products contribute to local inflammation. These products cause (1) the already increased blood flow to increase still further, (2) the capillary leakage of proteins to be increased, and (3) the interstitial fluid proteins to coagulate in the tissue spaces, thus preventing movement of the invading organism through the tissues.
Special Attributes of the T-Lymphocyte System—Activated T Cells and Cell-Mediated Immunity
Release of Activated T Cells from Lymphoid Tissue and Formation of Memory Cells.
Upon exposure to the proper antigen, as presented by adjacent macrophages, the T lymphocytes of a specific lymphocyte clone proliferate and release large numbers of activated, specifically reacting T cells in ways that parallel antibody release by activated B cells. The principal difference is that instead of releasing antibodies, whole activated T cells are formed and released into the lymph. These T cells then pass into the circulation and are distributed throughout the body, passing through the capillary walls into the tissue spaces, back into the lymph and blood once again, and circulating again and again throughout the body, sometimes lasting for months or even years.
Also, T-lymphocyte memory cells are formed in the same way that B memory cells are formed in the antibody system. That is, when a clone of T lymphocytes is activated by an antigen, many of the newly formed lymphocytes are preserved in the lymphoid tissue to become additional T lymphocytes of that specific clone; in fact, these memory cells even spread throughout the lymphoid tissue of the entire body. Therefore, on subsequent exposure to the same antigen anywhere in the body, release of activated T cells occurs far more rapidly and much more powerfully than during first exposure.
Antigen-Presenting Cells, MHC Proteins, and Antigen Receptors on the T Lymphocytes.
T-cell responses are extremely antigen specific, like the antibody responses of B cells, and are at least as important as antibodies in defending against infection. In fact, acquired immune responses usually require assistance from T cells to begin the process, and T cells play a major role in helping to eliminate invading pathogens.
Although B lymphocytes recognize intact antigens, T lymphocytes respond to antigens only when they are bound to specific molecules called MHC proteins on the surface of antigen-presenting cells in the lymphoid tissues (Figure 35-7). The three major types of antigen-presenting cells are macrophages, B lymphocytes, and dendritic cells. The dendritic cells, the most potent of the antigen-presenting cells, are located throughout the body, and their only known function is to present antigens to T cells. Interaction of cell adhesion proteins is critical in permitting the T cells to bind to antigen-presenting cells long enough to become activated.
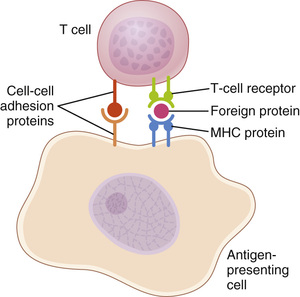
The MHC proteins are encoded by a large group of genes called the major histocompatibility complex (MHC). The MHC proteins bind peptide fragments of antigen proteins that are degraded inside antigen-presenting cells and then transport them to the cell surface. There are two types of MHC proteins: (1) MHC I proteins, which present antigens to cytotoxic T cells, and (2) MHC II proteins, which present antigens to T-helper cells. The specific functions of cytotoxic and T-helper cells are discussed later.
The antigens on the surface of antigen-presenting cells bind with receptor molecules on the surfaces of T cells in the same way that they bind with plasma protein antibodies. These receptor molecules are composed of a variable unit similar to the variable portion of the humoral antibody, but its stem section is firmly bound to the cell membrane of the T lymphocyte. There are as many as 100,000 receptor sites on a single T cell.
Several Types of T Cells and Their Different Functions
It has become clear that there are multiple types of T cells. They are classified into three major groups: (1) T-helper cells, (2) cytotoxic T cells, and (3) suppressor T cells. The functions of each of these T cells are distinct.
T-Helper Cells are the Most Numerous of the T Cells
The T-helper cells are by far the most numerous of the T cells, usually constituting more than three quarters of all of them. As their name implies, they help in the functions of the immune system, and they do so in many ways. In fact, they serve as the major regulator of virtually all immune functions, as shown in Figure 35-8. They do this by forming a series of protein mediators, called lymphokines, that act on other cells of the immune system, as well as on bone marrow cells. Among the most important lymphokines secreted by the T-helper cells are the following:
Specific Regulatory Functions of the Lymphokines.
In the absence of the lymphokines from the T-helper cells, the remainder of the immune system is almost paralyzed. In fact, it is the T-helper cells that are inactivated or destroyed by the human immunodeficiency virus (HIV), which leaves the body almost totally unprotected against infectious disease, therefore leading to the now well-known debilitating and lethal effects of acquired immunodeficiency syndrome (AIDS). Some of the specific regulatory functions are described in the following sections.
Stimulation of Growth and Proliferation of Cytotoxic T Cells and Suppressor T Cells.
In the absence of T-helper cells, the clones for producing cytotoxic T cells and suppressor T cells are activated only slightly by most antigens. The lymphokine interleukin-2 has an especially strong stimulatory effect in causing growth and proliferation of both cytotoxic and suppressor T cells. In addition, several of the other lymphokines have less potent effects.
Stimulation of B-Cell Growth and Differentiation to Form Plasma Cells and Antibodies.
The direct actions of antigens to cause B-cell growth, proliferation, formation of plasma cells, and secretion of antibodies are also slight without the “help” of the T-helper cells. Almost all the interleukins participate in the B-cell response, but especially interleukins 4, 5, and 6. In fact, these three interleukins have such potent effects on the B cells that they have been called B-cell stimulating factors or B-cell growth factors.
Activation of the Macrophage System.
The lymphokines also affect the macrophages. First, they slow or stop the migration of the macrophages after they have been chemotactically attracted into the inflamed tissue area, thus causing great accumulation of macrophages. Second, they activate the macrophages to cause far more efficient phagocytosis, allowing them to attack and destroy increasing numbers of invading bacteria or other tissue-destroying agents.
Feedback Stimulatory Effect on the T-Helper Cells.
Some of the lymphokines, especially interleukin-2, have a direct positive feedback effect in stimulating activation of the T-helper cells. This acts as an amplifier by further enhancing the helper cell response, as well as the entire immune response to an invading antigen.
Cytotoxic T Cells Are “Killer” Cells
The cytotoxic T cell is a direct-attack cell that is capable of killing microorganisms and, at times, even some of the body's own cells. For this reason, these cells are called killer cells. The receptor proteins on the surfaces of the cytotoxic cells cause them to bind tightly to the organisms or cells that contain the appropriate binding-specific antigen. Then they kill the attacked cell in the manner shown in Figure 35-9. After binding, the cytotoxic T cell secretes hole-forming proteins, called perforins, that literally punch round holes in the membrane of the attacked cell. Then fluid flows rapidly into the cell from the interstitial space. In addition, the cytotoxic T cell releases cytotoxic substances directly into the attacked cell. Almost immediately, the attacked cell becomes greatly swollen, and it usually dissolves shortly thereafter.
Of special importance is that these cytotoxic killer cells can pull away from the victim cells after they have punched holes and delivered cytotoxic substances and then move on to kill more cells. Indeed, some of these cells persist for months in the tissues.
Some of the cytotoxic T cells are especially lethal to tissue cells that have been invaded by viruses because many virus particles become entrapped in the membranes of the tissue cells and attract T cells in response to the viral antigenicity. The cytotoxic cells also play an important role in destroying cancer cells, heart transplant cells, or other types of cells that are foreign to the person's own body.
Suppressor T Cells
Much less is known about the suppressor T cells than about the others, but they are capable of suppressing the functions of both cytotoxic and T-helper cells. These suppressor functions are believed to prevent the cytotoxic cells from causing excessive immune reactions that might be damaging to the body's own tissues. For this reason, the suppressor cells are classified, along with the T-helper cells, as regulatory T cells. It is probable that the suppressor T-cell system plays an important role in limiting the ability of the immune system to attack a person's own body tissues, called immune tolerance, as we discuss in the next section.
Tolerance of the Acquired Immunity System to One's Own Tissues—Role of Preprocessing in the Thymus and Bone Marrow
The process of acquired immunity would destroy the individual's own body if a person became immune to his or her own tissues. The immune mechanism normally “recognizes” a person's own tissues as being distinctive from bacteria or viruses, and the person's immunity system forms few antibodies or activated T cells against his or her own antigens.
Most Tolerance Results from Clone Selection During Preprocessing.
Most tolerance is believed to develop during preprocessing of T lymphocytes in the thymus and of B lymphocytes in the bone marrow. The reason for this belief is that injecting a strong antigen into a fetus while the lymphocytes are being preprocessed in these two areas prevents development of clones of lymphocytes in the lymphoid tissue that are specific for the injected antigen. Experiments have shown that specific immature lymphocytes in the thymus, when exposed to a strong antigen, become lymphoblastic, proliferate considerably, and then combine with the stimulating antigen—an effect that is believed to cause the cells to be destroyed by the thymic epithelial cells before they can migrate to and colonize the total body lymphoid tissue.
It is believed that during the preprocessing of lymphocytes in the thymus and bone marrow, all or most of the clones of lymphocytes that are specific to damage the body's own tissues are self-destroyed because of their continual exposure to the body's antigens.
Failure of the Tolerance Mechanism Causes Autoimmune Diseases.
Sometimes people lose immune tolerance of their own tissues. This phenomenon occurs to a greater extent the older a person becomes. It usually occurs after destruction of some of the body's own tissues, which releases considerable quantities of “self-antigens” that circulate in the body and presumably cause acquired immunity in the form of either activated T cells or antibodies.
Several specific diseases that result from autoimmunity include (1) rheumatic fever, in which the body becomes immunized against tissues in the joints and heart, especially the heart valves, after exposure to a specific type of streptococcal toxin that has an epitope in its molecular structure similar to the structure of some of the body's own self-antigens; (2) one type of glomerulonephritis, in which the person becomes immunized against the basement membranes of glomeruli; (3) myasthenia gravis, in which immunity develops against the acetylcholine receptor proteins of the neuromuscular junction, causing paralysis; and (4) systemic lupus erythematosus (SLE), in which the person becomes immunized against many different body tissues at the same time, a disease that causes extensive damage and even death when SLE is severe.
Immunization by Injection of Antigens
Immunization has been used for many years to produce acquired immunity against specific diseases. A person can be immunized by injecting dead organisms that are no longer capable of causing disease but that still have some of their chemical antigens. This type of immunization is used to protect against typhoid fever, whooping cough, diphtheria, and many other types of bacterial diseases.
Immunity can be achieved against toxins that have been treated with chemicals so that their toxic nature has been destroyed even though their antigens for causing immunity are still intact. This procedure is used in immunizing against tetanus, botulism, and other similar toxic diseases.
And, finally, a person can be immunized by being infected with live organisms that have been “attenuated.” That is, these organisms either have been grown in special culture media or have been passed through a series of animals until they have mutated enough that they will not cause disease but do still carry specific antigens required for immunization. This procedure is used to protect against smallpox, yellow fever, poliomyelitis, measles, and many other viral diseases.
Passive Immunity
Thus far, all the acquired immunity we have discussed has been active immunity—that is, the person's own body develops either antibodies or activated T cells in response to invasion of the body by a foreign antigen. However, temporary immunity can be achieved in a person without injecting any antigen. This temporary immunity is achieved by infusing antibodies, activated T cells, or both obtained from the blood of someone else or from some other animal that has been actively immunized against the antigen.
Antibodies last in the body of the recipient for 2 to 3 weeks, and during that time, the person is protected against the invading disease. Activated T cells last for a few weeks if transfused from another person but only for a few hours to a few days if transfused from an animal. Such transfusion of antibodies or T lymphocytes to confer immunity is called passive immunity.
Allergy and Hypersensitivity
An important undesirable side effect of immunity is the development, under some conditions, of allergy or other types of immune hypersensitivity. There are several types of allergy and other hypersensitivities, some of which occur only in people who have a specific allergic tendency.
Allergy Caused by Activated T Cells: Delayed-Reaction Allergy
Delayed-reaction allergy is caused by activated T cells and not by antibodies. In the case of poison ivy, the toxin of poison ivy in itself does not cause much harm to the tissues. However, upon repeated exposure, it does cause the formation of activated helper and cytotoxic T cells. Then, after subsequent exposure to the poison ivy toxin, within a day or so, the activated T cells diffuse from the circulating blood in large numbers into the skin to respond to the poison ivy toxin. At the same time, these T cells elicit a cell-mediated type of immune reaction. Remembering that this type of immunity can cause release of many toxic substances from the activated T cells, as well as extensive invasion of the tissues by macrophages along with their subsequent effects, one can well understand that the eventual result of some delayed-reaction allergies can be serious tissue damage. The damage normally occurs in the tissue area where the instigating antigen is present, such as in the skin in the case of poison ivy, or in the lungs to cause lung edema or asthmatic attacks in the case of some airborne antigens.
“Atopic” Allergies Associated with Excess IgE Antibodies
Some people have an “allergic” tendency. Their allergies are called atopic allergies because they are caused by a nonordinary response of the immune system. The allergic tendency is genetically passed from parent to child and is characterized by the presence of large quantities of IgE antibodies in the blood. These antibodies are called reagins or sensitizing antibodies to distinguish them from the more common IgG antibodies. When an allergen (defined as an antigen that reacts specifically with a specific type of IgE reagin antibody) enters the body, an allergen-reagin reaction takes place and a subsequent allergic reaction occurs.
A special characteristic of the IgE antibodies (the reagins) is a strong propensity to attach to mast cells and basophils. Indeed, a single mast cell or basophil can bind as many as half a million molecules of IgE antibodies. Then, when an antigen (an allergen) that has multiple binding sites binds with several IgE antibodies that are already attached to a mast cell or basophil, this causes immediate change in the membrane of the mast cell or basophil, perhaps resulting from a physical effect of the antibody molecules to contort the cell membrane. At any rate, many of the mast cells and basophils rupture; others release special agents immediately or shortly thereafter, including histamine, protease, slow-reacting substance of anaphylaxis (which is a mixture of toxic leukotrienes), eosinophil chemotactic substance, neutrophil chemotactic substance, heparin, and platelet activating factors. These substances cause such effects as dilation of the local blood vessels; attraction of eosinophils and neutrophils to the reactive site; increased permeability of the capillaries with loss of fluid into the tissues; and contraction of local smooth muscle cells. Therefore, several different tissue responses can occur, depending on the type of tissue in which the allergen-reagin reaction occurs. Among the different types of allergic reactions caused in this manner are the following.
Anaphylaxis.
When a specific allergen is injected directly into the circulation, the allergen can react with basophils of the blood and mast cells in the tissues located immediately outside the small blood vessels if the basophils and mast cells have been sensitized by attachment of IgE reagins. Therefore, a widespread allergic reaction occurs throughout the vascular system and closely associated tissues. This reaction is called anaphylaxis. Histamine is released into the circulation and causes body-wide vasodilation, as well as increased permeability of the capillaries with resultant marked loss of plasma from the circulation. Occasionally, a person who experiences this reaction dies of circulatory shock within a few minutes unless treated with epinephrine to oppose the effects of the histamine.
Also released from the activated basophils and mast cells is a mixture of leukotrienes called slow-reacting substance of anaphylaxis. These leukotrienes can cause spasm of the smooth muscle of the bronchioles, eliciting an asthma-like attack, sometimes causing death by suffocation.
Urticaria.
Urticaria results from antigen entering specific skin areas and causing localized anaphylactoid reactions. Histamine released locally causes (1) vasodilation that induces an immediate red flare and (2) increased local permeability of the capillaries that leads to local circumscribed areas of swelling of the skin within another few minutes. The swellings are commonly called hives. Administration of antihistamine drugs to a person before exposure will prevent the hives.
Hay Fever.
In hay fever, the allergen-reagin reaction occurs in the nose. Histamine released in response to the reaction causes local intranasal vascular dilation, with resultant increased capillary pressure and increased capillary permeability. Both these effects cause rapid fluid leakage into the nasal cavities and into associated deeper tissues of the nose, and the nasal linings become swollen and secretory. Here again, use of antihistamine drugs can prevent this swelling reaction. However, other products of the allergen-reagin reaction can still cause irritation of the nose, eliciting the typical sneezing syndrome.
Asthma.
Asthma often occurs in the “allergic” type of person. In such a person, the allergen-reagin reaction occurs in the bronchioles of the lungs. Here, an important product released from the mast cells is believed to be the slow-reacting substance of anaphylaxis (a mixture of three leukotrienes), which causes spasm of the bronchiolar smooth muscle. Consequently, the person has difficulty breathing until the reactive products of the allergic reaction have been removed. Administration of antihistamine medication has less effect on the course of asthma because histamine does not appear to be the major factor eliciting the asthmatic reaction.




