Pituitary Hormones and Their Control by the Hypothalamus
Pituitary Gland and Its Relation to the Hypothalamus
The Anterior and Posterior Lobes of the Pituitary Gland
The pituitary gland (Figure 76-1), also called the hypophysis, is a small gland—about 1 centimeter in diameter and 0.5 to 1 gram in weight—that lies in the sella turcica, a bony cavity at the base of the brain, and is connected to the hypothalamus by the pituitary (or hypophysial) stalk. Physiologically, the pituitary gland is divisible into two distinct portions: the anterior pituitary, also known as the adenohypophysis, and the posterior pituitary, also known as the neurohypophysis. Between these portions is a small, relatively avascular zone called the pars intermedia, which is much less developed in the human being but is larger and much more functional in some animals.
Embryologically, the two portions of the pituitary originate from different sources—the anterior pituitary from Rathke's pouch, which is an embryonic invagination of the pharyngeal epithelium, and the posterior pituitary from a neural tissue outgrowth from the hypothalamus. The origin of the anterior pituitary from the pharyngeal epithelium explains the epithelioid nature of its cells, and the origin of the posterior pituitary from neural tissue explains the presence of large numbers of glial-type cells in this gland.
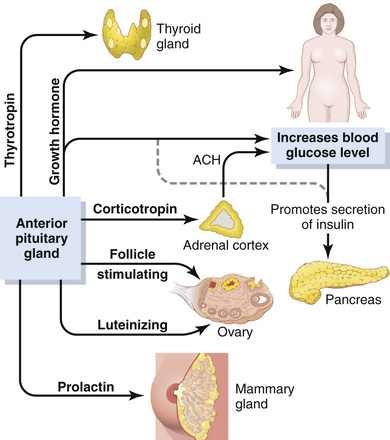
Six major peptide hormones plus several other hormones of lesser importance are secreted by the anterior pituitary, and two important peptide hormones are secreted by the posterior pituitary. The hormones of the anterior pituitary play major roles in the control of metabolic functions throughout the body, as shown in Figure 76-2.
• Growth hormone promotes growth of the entire body by affecting protein formation, cell multiplication, and cell differentiation.
• Adrenocorticotropin (corticotropin) controls the secretion of some of the adrenocortical hormones, which affect metabolism of glucose, proteins, and fats.
• Thyroid-stimulating hormone (thyrotropin) controls the secretion rate of thyroxine and triiodothyronine by the thyroid gland, and these hormones control the rates of most intracellular chemical reactions in the body.
• Prolactin promotes mammary gland development and milk production.
• Two separate gonadotropic hormones, follicle-stimulating hormone and luteinizing hormone, control growth of the ovaries and testes, as well as their hormonal and reproductive activities. The two hormones secreted by the posterior pituitary play other roles.
• Antidiuretic hormone (also called vasopressin) controls the rate of water excretion into the urine, thus helping to control the concentration of water in the body fluids.
• Oxytocin helps express milk from the glands of the breast to the nipples during suckling and helps in the delivery of the baby at the end of gestation.
The Anterior Pituitary Gland Contains Several Different Cell Types That Synthesize and Secrete Hormones.
Usually, there is one cell type for each major hormone formed in the anterior pituitary gland. With special stains attached to high-affinity antibodies that bind with the distinctive hormones, at least five cell types can be differentiated (Figure 76-3). Table 76-1 provides a summary of these cell types, the hormones they produce, and their physiological actions. These five cell types are:
1. Somatotropes—human growth hormone (hGH)
2. Corticotropes—adrenocorticotropin (ACTH)
3. Thyrotropes—thyroid-stimulating hormone (TSH)
4. Gonadotropes—gonadotropic hormones, which include both luteinizing hormone (LH) and follicle-stimulating hormone (FSH)
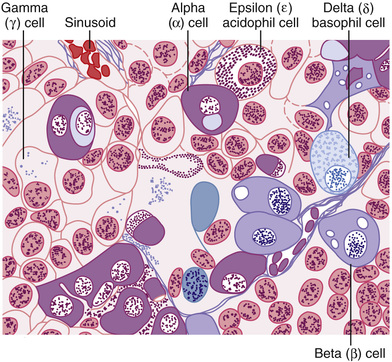
Table 76-1
Cells and Hormones of the Anterior Pituitary Gland and Their Physiological Functions
| Cell | Hormone | Chemistry | Physiological Action |
| Somatotropes | Growth hormone (GH) (somatotropin) | Single chain of 191 amino acids | Stimulates body growth; stimulates secretion of insulin-like growth factor-1; stimulates lipolysis; inhibits actions of insulin on carbohydrate and lipid metabolism |
| Corticotropes | Adrenocorticotropic hormone (ACTH) (corticotropin) | Single chain of 39 amino acids | Stimulates production of glucocorticoids and androgens by the adrenal cortex; maintains size of zona fasciculata and zona reticularis of cortex |
| Thyrotropes | Thyroid-stimulating hormone (TSH) (thyrotropin) | Glycoprotein of two subunits, α (89 amino acids) and β (112 amino acids) | Stimulates production of thyroid hormones by thyroid follicular cells; maintains size of follicular cells |
| Gonadotropes | Follicle-stimulating hormone (FSH) | Glycoprotein of two subunits, α (89 amino acids) and β (112 amino acids) | Stimulates development of ovarian follicles; regulates spermatogenesis in the testis |
| Luteinizing (LH) hormone | Glycoprotein of two subunits, α (89 amino acids) and β (115 amino acids) | Causes ovulation and formation of the corpus luteum in the ovary; stimulates production of estrogen and progesterone by the ovary; stimulates testosterone production by the testis | |
| Lactotropes-Mammotropes | Prolactin (PRL) | Single chain of 198 amino acids | Stimulates milk secretion and production |

About 30 to 40 percent of the anterior pituitary cells are somatotropes that secrete growth hormone, and about 20 percent are corticotropes that secrete ACTH. Each of the other cell types accounts for only 3 to 5 percent of the total; nevertheless, they secrete powerful hormones for controlling thyroid function, sexual functions, and milk secretion by the breasts.
Somatotropes stain strongly with acid dyes and are therefore called acidophils. Thus, pituitary tumors that secrete large quantities of human growth hormone are called acidophilic tumors.
Posterior Pituitary Hormones Are Synthesized by Cell Bodies in the Hypothalamus.
The bodies of the cells that secrete the posterior pituitary hormones are not located in the pituitary gland but are large neurons, called magnocellular neurons, located in the supraoptic and paraventricular nuclei of the hypothalamus. The hormones are then transported in the axoplasm of the neurons' nerve fibers passing from the hypothalamus to the posterior pituitary gland. This mechanism is discussed later in the chapter.
Hypothalamus Controls Pituitary Secretion
Almost all secretion by the pituitary is controlled by either hormonal or nervous signals from the hypothalamus. Indeed, when the pituitary gland is removed from its normal position beneath the hypothalamus and transplanted to some other part of the body, its secretion rates of the different hormones (except for prolactin) fall to very low levels.
Secretion from the posterior pituitary is controlled by nerve signals that originate in the hypothalamus and terminate in the posterior pituitary. In contrast, secretion by the anterior pituitary is controlled by hormones called hypothalamic releasing and hypothalamic inhibitory hormones (or factors) secreted within the hypothalamus and then conducted, as shown in Figure 76-4, to the anterior pituitary through minute blood vessels called hypothalamic-hypophysial portal vessels. In the anterior pituitary, these releasing and inhibitory hormones act on the glandular cells to control their secretion. This system of control is discussed in the next section of this chapter.
The hypothalamus receives signals from many sources in the nervous system. Thus, when a person is exposed to pain, a portion of the pain signal is transmitted into the hypothalamus. Likewise, when a person experiences some powerful depressing or exciting thought, a portion of the signal is transmitted into the hypothalamus. Olfactory stimuli denoting pleasant or unpleasant smells transmit strong signals directly and through the amygdaloid nuclei into the hypothalamus. Even the concentrations of nutrients, electrolytes, water, and various hormones in the blood excite or inhibit various portions of the hypothalamus. Thus, the hypothalamus is a collecting center for information concerning the internal well-being of the body, and much of this information is used to control secretions of the many globally important pituitary hormones.
Hypothalamic-Hypophysial Portal Blood Vessels of the Anterior Pituitary Gland
The anterior pituitary is a highly vascular gland with extensive capillary sinuses among the glandular cells. Almost all the blood that enters these sinuses passes first through another capillary bed in the lower hypothalamus. The blood then flows through small hypothalamic-hypophysial portal blood vessels into the anterior pituitary sinuses. Figure 76-4 shows the lowermost portion of the hypothalamus, called the median eminence, which connects inferiorly with the pituitary stalk. Small arteries penetrate into the median eminence and then additional small vessels return to its surface, coalescing to form the hypothalamic-hypophysial portal blood vessels. These vessels pass downward along the pituitary stalk to supply blood to the anterior pituitary sinuses.
Hypothalamic Releasing and Inhibitory Hormones Are Secreted Into the Median Eminence.
Special neurons in the hypothalamus synthesize and secrete the hypothalamic releasing and inhibitory hormones that control secretion of the anterior pituitary hormones. These neurons originate in various parts of the hypothalamus and send their nerve fibers to the median eminence and tuber cinereum, an extension of hypothalamic tissue into the pituitary stalk.
The endings of these fibers are different from most endings in the central nervous system, in that their function is not to transmit signals from one neuron to another but rather to secrete the hypothalamic releasing and inhibitory hormones into the tissue fluids. These hormones are immediately absorbed into the hypothalamic-hypophysial portal system and carried directly to the sinuses of the anterior pituitary gland.
Hypothalamic Releasing and Inhibitory Hormones Control Anterior Pituitary Secretion.
The function of the releasing and inhibitory hormones is to control secretion of the anterior pituitary hormones. For most of the anterior pituitary hormones, it is the releasing hormones that are important, but for prolactin, a hypothalamic inhibitory hormone probably exerts more control. The major hypothalamic releasing and inhibitory hormones, which are summarized in Table 76-2, are the following:
1. Thyrotropin-releasing hormone (TRH), which causes release of thyroid-stimulating hormone
2. Corticotropin-releasing hormone (CRH), which causes release of adrenocorticotropin
3. Growth hormone–releasing hormone (GHRH), which causes release of growth hormone, and growth hormone inhibitory hormone (GHIH), also called somatostatin, which inhibits release of growth hormone
4. Gonadotropin-releasing hormone (GnRH), which causes release of the two gonadotropic hormones, luteinizing hormone (LH) and follicle-stimulating hormone (FSH)
5. Prolactin inhibitory hormone (PIH), which causes inhibition of prolactin secretion
Table 76-2
Hypothalamic Releasing and Inhibitory Hormones That Control Secretion of the Anterior Pituitary Gland
| Hormone | Structure | Primary Action on Anterior Pituitary |
| Thyrotropin-releasing hormone (TRH) | Peptide of 3 amino acids | Stimulates secretion of TSH by thyrotropes |
| Gonadotropin-releasing hormone (GnRH) | Single chain of 10 amino acids | Stimulates secretion of FSH and LH by gonadotropes |
| Corticotropin-releasing hormone (CRH) | Single chain of 41 amino acids | Stimulates secretion of ACTH by corticotropes |
| Growth hormone–releasing hormone (GHRH) | Single chain of 44 amino acids | Stimulates secretion of growth hormone by somatotropes |
| Growth hormone inhibitory hormone (somatostatin) | Single chain of 14 amino acids | Inhibits secretion of growth hormone by somatotropes |
| Prolactin-inhibiting hormone (PIH) | Dopamine (a catecholamine) | Inhibits synthesis and secretion of prolactin by lactotropes |
Additional hypothalamic hormones include one that stimulates prolactin secretion and perhaps others that inhibit release of the anterior pituitary hormones. Each of the more important hypothalamic hormones is discussed in detail as the specific hormonal systems controlled by them are presented in this and subsequent chapters.
Specific Areas in the Hypothalamus Control Secretion of Specific Hypothalamic Releasing and Inhibitory Hormones.
All or most of the hypothalamic hormones are secreted at nerve endings in the median eminence before being transported to the anterior pituitary gland. Electrical stimulation of this region excites these nerve endings and, therefore, causes release of essentially all the hypothalamic hormones. However, the neuronal cell bodies that give rise to these median eminence nerve endings are located in other discrete areas of the hypothalamus or in closely related areas of the basal brain. The specific loci of the neuronal cell bodies that form the different hypothalamic releasing or inhibitory hormones are still poorly known, so it would be misleading to attempt delineation here.
Physiological Functions of Growth Hormone
All the major anterior pituitary hormones, except for growth hormone, exert their principal effects mainly by stimulating target glands, including thyroid gland, adrenal cortex, ovaries, testicles, and mammary glands. The functions of each of these pituitary hormones are so intimately concerned with the functions of the respective target glands that, except for growth hormone, their functions are discussed in subsequent chapters along with the target glands. Growth hormone, however, does not function through a specific target gland but exerts its effects directly on all or almost all tissues of the body.
Growth Hormone Promotes Growth of Many Body Tissues
Growth hormone, also called somatotropic hormone or somatotropin, is a small protein molecule that contains 191 amino acids in a single chain and has a molecular weight of 22,005. It causes growth of almost all tissues of the body that are capable of growing. It promotes increased sizes of the cells and increased mitosis, with development of greater numbers of cells and specific differentiation of certain types of cells such as bone growth cells and early muscle cells.
Figure 76-5 shows typical weight charts of two growing littermate rats; one received daily injections of growth hormone, and the other did not receive growth hormone. This figure shows marked enhancement of growth in the rat given growth hormone, in the early days of life and even after the two rats reached adulthood. In the early stages of development, all organs of the treated rat increased proportionately in size; after adulthood was reached, most of the bones stopped lengthening, but many of the soft tissues continued to grow. This outcome results from the fact that once the epiphyses of the long bones have united with the shafts, further lengthening of bone cannot occur, even though many other tissues of the body can continue to grow throughout life.
Growth Hormone Has Several Metabolic Effects
Aside from its general effect in causing growth, growth hormone has multiple specific metabolic effects, including (1) increased rate of protein synthesis in most cells of the body; (2) increased mobilization of fatty acids from adipose tissue, increased free fatty acids in the blood, and increased use of fatty acids for energy; and (3) decreased rate of glucose utilization throughout the body. Thus, in effect, growth hormone enhances body protein, decreases fat stores, and conserves carbohydrates.
Growth Hormone Promotes Protein Deposition in Tissues
Although the precise mechanisms by which growth hormone increases protein deposition are not fully understood, a series of different effects are known, all of which could lead to enhanced protein deposition.
Enhancement of Amino Acid Transport Through the Cell Membranes.
Growth hormone directly enhances transport of most amino acids through the cell membranes to the interior of the cells. This increases the amino acid concentrations in the cells and is presumed to be at least partly responsible for the increased protein synthesis. This control of amino acid transport is similar to the effect of insulin in controlling glucose transport through the membrane, as discussed in Chapters 68 and 79.
Enhancement of RNA Translation to Cause Protein Synthesis by the Ribosomes.
Even when the amino acid concentrations are not increased in the cells, growth hormone still increases RNA translation, causing protein to be synthesized in greater amounts by the ribosomes in the cytoplasm.
Increased Nuclear Transcription of DNA to Form RNA.
Over more prolonged periods (24 to 48 hours), growth hormone also stimulates transcription of DNA in the nucleus, causing formation of increased quantities of RNA. This promotes more protein synthesis and growth if sufficient energy, amino acids, vitamins, and other requisites for growth are available. In the long run, this may be the most important function of growth hormone.
Decreased Catabolism of Protein and Amino Acids.
In addition to the increase in protein synthesis, there is a decrease in the breakdown of cell protein. A probable reason for this decrease is that growth hormone also mobilizes large quantities of free fatty acids from the adipose tissue, and these are used to supply most of the energy for the body's cells, thus acting as a potent “protein sparer.”
Summary.
Growth hormone enhances almost all facets of amino acid uptake and protein synthesis by cells, while at the same time reducing the breakdown of proteins.
Growth Hormone Enhances Fat Utilization for Energy
Growth hormone has a specific effect in causing release of fatty acids from adipose tissue and, therefore, increasing the concentration of fatty acids in body fluids. Also, in tissues throughout the body, growth hormone enhances conversion of fatty acids to acetyl coenzyme A (acetyl-CoA) and its subsequent utilization for energy. Therefore, under the influence of growth hormone, fat is used for energy in preference to use of carbohydrates and proteins.
The ability of growth hormone to promote fat utilization, together with its protein anabolic effect, causes an increase in lean body mass. However, mobilization of fat by growth hormone requires several hours to occur, whereas enhancement of protein synthesis can begin in minutes under the influence of growth hormone.
“Ketogenic” Effect of Excessive Growth Hormone.
Under the influence of excessive amounts of growth hormone, fat mobilization from adipose tissue sometimes becomes so great that large quantities of acetoacetic acid are formed by the liver and released into the body fluids, thus causing ketosis. This excessive mobilization of fat from the adipose tissue also frequently causes a fatty liver.
Growth Hormone Decreases Carbohydrate Utilization
Growth hormone causes multiple effects that influence carbohydrate metabolism, including (1) decreased glucose uptake in tissues such as skeletal muscle and fat, (2) increased glucose production by the liver, and (3) increased insulin secretion.
Each of these changes results from growth hormone–induced “insulin resistance,” which attenuates insulin's actions to stimulate uptake and utilization of glucose in skeletal muscle and adipose tissue and to inhibit gluconeogenesis (glucose production) by the liver; this leads to increased blood glucose concentration and a compensatory increase in insulin secretion. For these reasons, growth hormone's effects are called diabetogenic, and excess secretion of growth hormone can produce metabolic disturbances similar to those found in patients with type II (non–insulin-dependent) diabetes, who are also resistant to the metabolic effects of insulin.
We do not know the precise mechanism by which growth hormone causes insulin resistance and decreased glucose utilization by the cells. However, growth hormone–induced increases in blood concentrations of fatty acids likely contribute to impairment of insulin's actions on tissue glucose utilization. Experimental studies indicate that raising blood levels of fatty acids above normal rapidly decreases the sensitivity of the liver and skeletal muscle to insulin's effects on carbohydrate metabolism.
Necessity of Insulin and Carbohydrate for the Growth-Promoting Action of Growth Hormone
Growth hormone fails to cause growth in animals that lack a pancreas; it also fails to cause growth if carbohydrates are excluded from the diet. These phenomena show that adequate insulin activity and adequate availability of carbohydrates are necessary for growth hormone to be effective. Part of this requirement for carbohydrates and insulin is to provide the energy needed for the metabolism of growth, but there seem to be other effects as well. Especially important is the ability of insulin to enhance transport of some amino acids into cells, in the same way that it stimulates glucose transport.
Growth Hormone Stimulates Cartilage and Bone Growth
Although growth hormone stimulates increased deposition of protein and increased growth in almost all tissues of the body, its most obvious effect is to increase growth of the skeletal frame. This results from multiple effects of growth hormone on bone, including (1) increased deposition of protein by the chondrocytic and osteogenic cells that cause bone growth, (2) increased rate of reproduction of these cells, and (3) a specific effect of converting chondrocytes into osteogenic cells, thus causing deposition of new bone.
There are two principal mechanisms of bone growth. First, in response to growth hormone stimulation, the long bones grow in length at the epiphyseal cartilages, where the epiphyses at the ends of the bone are separated from the shaft. This growth first causes deposition of new cartilage, followed by its conversion into new bone, thus elongating the shaft and pushing the epiphyses farther and farther apart. At the same time, the epiphyseal cartilage is progressively used up, so by late adolescence, no additional epiphyseal cartilage remains to provide for further long bone growth. At this time, bony fusion occurs between the shaft and the epiphysis at each end, so no further lengthening of the long bone can occur.
Second, osteoblasts in the bone periosteum and in some bone cavities deposit new bone on the surfaces of older bone. Simultaneously, osteoclasts in the bone (discussed in detail in Chapter 80) remove old bone. When the rate of deposition is greater than that of resorption, the thickness of the bone increases. Growth hormone strongly stimulates osteoblasts. Therefore, the bones can continue to become thicker throughout life under the influence of growth hormone; this is especially true for the membranous bones. For instance, the jaw bones can be stimulated to grow even after adolescence, causing forward protrusion of the chin and lower teeth. Likewise, the bones of the skull can grow in thickness and give rise to bony protrusions over the eyes.
Growth Hormone Exerts Much of Its Effect Through Intermediate Substances Called Somatomedins
When growth hormone is supplied directly to cartilage chondrocytes cultured outside the body, proliferation or enlargement of the chondrocytes usually fails to occur. Yet, growth hormone injected into the intact animal does cause proliferation and growth of the same cells.
In brief, growth hormone causes the liver (and, to a much less extent, other tissues) to form several small proteins called somatomedins that have the potent effect of increasing all aspects of bone growth. Many of the somatomedin effects on growth are similar to the effects of insulin on growth. Therefore, the somatomedins are also called insulin-like growth factors (IGFs).
At least four somatomedins have been isolated, but by far the most important of these is somatomedin C (also called insulin-like growth factor-1, or IGF-I). The molecular weight of somatomedin C is about 7500, and its concentration in the plasma closely follows the rate of growth hormone secretion.
The pygmies of Africa have a congenital inability to synthesize significant amounts of somatomedin C. Therefore, even though their plasma concentration of growth hormone is either normal or high, they have diminished amounts of somatomedin C in the plasma, which apparently accounts for the small stature of these people. Some other dwarfs (e.g., Laron syndrome) have a similar problem.
It has been postulated that most, if not all, of the growth effects of growth hormone result from somatomedin C and other somatomedins, rather than from direct effects of growth hormone on the bones and other peripheral tissues. Even so, experiments have demonstrated that injection of growth hormone directly into the epiphyseal cartilages of bones of living animals causes growth of these cartilage areas, and the amount of growth hormone required for this growth to occur is minute. Some aspects of the somatomedin hypothesis are still questionable. One possibility is that growth hormone can cause the formation of enough somatomedin C in the local tissue to cause local growth. It is also possible that growth hormone is directly responsible for increased growth in some tissues and that the somatomedin mechanism is an alternative means of increasing growth but not always a necessary one.
Short Duration of Action of Growth Hormone but Prolonged Action of Somatomedin C.
Growth hormone attaches only weakly to the plasma proteins in blood. Therefore, it is released from the blood into the tissues rapidly, having a half-time in blood of less than 20 minutes. By contrast, somatomedin C attaches strongly to a carrier protein in the blood that, like somatomedin C, is produced in response to growth hormone. As a result, somatomedin C is released only slowly from the blood to the tissues, with a half-time of about 20 hours. This slow release greatly prolongs the growth-promoting effects of the bursts of growth hormone secretion shown in Figure 76-6.
Regulation of Growth Hormone Secretion
After adolescence, secretion decreases slowly with aging, finally falling to about 25 percent of the adolescent level in very old age.
Growth hormone is secreted in a pulsatile pattern, increasing and decreasing. The precise mechanisms that control secretion of growth hormone are not fully understood, but several factors related to a person's state of nutrition or stress are known to stimulate secretion: (1) starvation, especially with severe protein deficiency; (2) hypoglycemia or low concentration of fatty acids in the blood; (3) exercise; (4) excitement; (5) trauma; and (6) ghrelin, a hormone secreted by the stomach before meals. Growth hormone also characteristically increases during the first 2 hours of deep sleep, as shown in Figure 76-6. Table 76-3 summarizes some of the factors that are known to influence growth hormone secretion.
Table 76-3
Factors That Stimulate or Inhibit Secretion of Growth Hormone
| Stimulate Growth Hormone Secretion | Inhibit Growth Hormone Secretion |
| Decreased blood glucose Decreased blood free fatty acids Increased blood amino acids (arginine) Starvation or fasting, protein deficiency Trauma, stress, excitement Exercise Testosterone, estrogen Deep sleep (stages II and IV) Growth hormone–releasing hormone Ghrelin | Increased blood glucose Increased blood free fatty acids Aging Obesity Growth hormone inhibitory hormone (somatostatin) Growth hormone (exogenous) Somatomedins (insulin-like growth factors) Increased blood glucose Increased blood free fatty acids Aging |
The normal concentration of growth hormone in the plasma of an adult is between 1.6 and 3 ng/ml; in a child or adolescent, it is about 6 ng/ml. These values may increase to as high as 50 ng/ml after depletion of the body stores of proteins or carbohydrates during prolonged starvation.
Under acute conditions, hypoglycemia is a far more potent stimulator of growth hormone secretion than is an acute decrease in protein intake. Conversely, in chronic conditions, growth hormone secretion seems to correlate more with the degree of cellular protein depletion than with the degree of glucose insufficiency. For instance, the extremely high levels of growth hormone that occur during starvation are closely related to the amount of protein depletion.
Figure 76-7 demonstrates the effect of protein deficiency on plasma growth hormone and then the effect of adding protein to the diet. The first column shows very high levels of growth hormone in children with extreme protein deficiency during the protein malnutrition condition called kwashiorkor; the second column shows the levels in the same children after 3 days of treatment with more than adequate quantities of carbohydrates in their diets, demonstrating that the carbohydrates did not lower the plasma growth hormone concentration. The third and fourth columns show the levels after treatment with protein supplements for 3 and 25 days, respectively, with a concomitant decrease in the hormone.
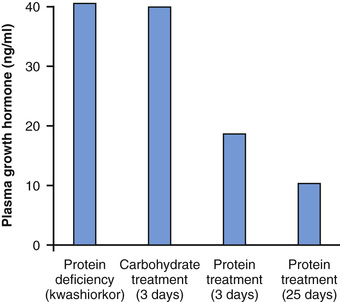
These results demonstrate that under severe conditions of protein malnutrition, adequate calories alone are not sufficient to correct the excess production of growth hormone. The protein deficiency must also be corrected before the growth hormone concentration will return to normal.
Role of the Hypothalamus, Growth Hormone–Releasing Hormone, and Somatostatin in Controlling Growth Hormone Secretion
From the preceding description of the many factors that can affect growth hormone secretion, one can readily understand the perplexity of physiologists as they attempted to unravel the mysteries of regulation of growth hormone secretion. It is known that growth hormone secretion is controlled by two factors secreted in the hypothalamus and then transported to the anterior pituitary gland through the hypothalamic-hypophysial portal vessels. They are growth hormone–releasing hormone (GHRH) and growth hormone inhibitory hormone (also called somatostatin). Both of these are polypeptides; GHRH is composed of 44 amino acids, and somatostatin is composed of 14 amino acids.
The part of the hypothalamus that causes secretion of GHRH is the ventromedial nucleus; this is the same area of the hypothalamus that is sensitive to blood glucose concentration, causing satiety in hyperglycemic states and hunger in hypoglycemic states. The secretion of somatostatin is controlled by other nearby areas of the hypothalamus. Therefore, it is reasonable to expect that some of the same signals that modify a person's behavioral feeding instincts also alter the rate of growth hormone secretion.
In a similar manner, hypothalamic signals depicting emotions, stress, and trauma can all affect hypothalamic control of growth hormone secretion. In fact, experiments have shown that catecholamines, dopamine, and serotonin, each of which is released by a different neuronal system in the hypothalamus, all increase the rate of growth hormone secretion.
Most of the control of growth hormone secretion is probably mediated through GHRH rather than through the inhibitory hormone somatostatin. GHRH stimulates growth hormone secretion by attaching to specific cell membrane receptors on the outer surfaces of the growth hormone cells in the pituitary gland. The receptors activate the adenylyl cyclase system inside the cell membrane, increasing the intracellular level of cyclic adenosine monophosphate (cAMP). This increase has both short-term and long-term effects. The short-term effect is to increase calcium ion transport into the cell; within minutes, this increase causes fusion of the growth hormone secretory vesicles with the cell membrane and release of the hormone into the blood. The long-term effect is to increase transcription in the nucleus by the genes to stimulate synthesis of new growth hormone.
When growth hormone is administered directly into the blood of an animal over several hours, the rate of endogenous growth hormone secretion decreases. This decrease demonstrates that growth hormone secretion is subject to typical negative feedback control, as is true for essentially all hormones. The nature of this feedback mechanism and whether it is mediated mainly through inhibition of GHRH or enhancement of somatostatin, which inhibits growth hormone secretion, are uncertain.
In summary, our knowledge of the regulation of growth hormone secretion is not sufficient to describe a composite picture. Yet, because of the extreme secretion of growth hormone during starvation and its important long-term effect to promote protein synthesis and tissue growth, we can propose the following: the major long-term controller of growth hormone secretion is the long-term state of nutrition of the tissues, especially their level of protein nutrition. That is, nutritional deficiency or excess tissue need for cellular proteins—for instance, after a severe bout of exercise when the muscles' nutritional status has been taxed—in some way increases the rate of growth hormone secretion. Growth hormone, in turn, promotes synthesis of new proteins while at the same time conserving the proteins already present in the cells.
 Abnormalities of Growth Hormone Secretion
Abnormalities of Growth Hormone Secretion
 Panhypopituitarism.
Panhypopituitarism.
Panhypopituitarism means decreased secretion of all the anterior pituitary hormones. The decrease in secretion may be congenital (present from birth), or it may occur suddenly or slowly at any time during life, most often resulting from a pituitary tumor that destroys the pituitary gland.
 Panhypopituitarism in the Adult.
Panhypopituitarism in the Adult.
Panhypopituitarism first occurring in adulthood frequently results from one of three common abnormalities. Two tumorous conditions, craniopharyngiomas or chromophobe tumors, may compress the pituitary gland until the functioning anterior pituitary cells are totally or almost totally destroyed. The third cause is thrombosis of the pituitary blood vessels. This abnormality occasionally occurs when a new mother experiences circulatory shock after the birth of her baby.
The general effects of adult panhypopituitarism are (1) hypothyroidism, (2) depressed production of glucocorticoids by the adrenal glands, and (3) suppressed secretion of the gonadotropic hormones so that sexual functions are lost. Thus, the picture is that of a lethargic person (from lack of thyroid hormones) who is gaining weight (because of lack of fat mobilization by growth, adrenocorticotropic, adrenocortical, and thyroid hormones) and has lost all sexual functions. Except for the abnormal sexual functions, the patient can usually be treated satisfactorily by administering adrenocortical and thyroid hormones.
 Dwarfism.
Dwarfism.
Most instances of dwarfism result from generalized deficiency of anterior pituitary secretion (panhypopituitarism) during childhood. In general, all the physical parts of the body develop in appropriate proportion to one another, but the rate of development is greatly decreased. A child who has reached the age of 10 years may have the bodily development of a child aged 4 to 5 years, and the same person at age 20 years may have the bodily development of a child aged 7 to 10 years.
A person with panhypopituitary dwarfism does not pass through puberty and never secretes sufficient quantities of gonadotropic hormones to develop adult sexual functions. In one third of such dwarfs, however, only growth hormone is deficient; these persons do mature sexually and occasionally reproduce. In one type of dwarfism (the African pygmy and the Laron dwarf), the rate of growth hormone secretion is normal or high, but there is a hereditary inability to form somatomedin C, which is a key step for the promotion of growth by growth hormone.
 Treatment with Human Growth Hormone.
Treatment with Human Growth Hormone.
Growth hormones from different species of animals are sufficiently different from one another that they will cause growth only in the one species or, at most, closely related species. For this reason, growth hormone prepared from lower animals (except, to some extent, from primates) is not effective in human beings. Therefore, the growth hormone of the human being is called human growth hormone to distinguish it from the others.
In the past, because growth hormone had to be prepared from human pituitary glands, it was difficult to obtain sufficient quantities to treat patients with growth hormone deficiency, except on an experimental basis. However, human growth hormone can now be synthesized by Escherichia coli bacteria as a result of successful application of recombinant DNA technology. Therefore, this hormone is now available in sufficient quantities for treatment purposes. Dwarfs who have pure growth hormone deficiency can be completely cured if treated early in life. Human growth hormone may also prove to be beneficial in other metabolic disorders because of its widespread metabolic functions.
 Gigantism.
Gigantism.
Occasionally, the acidophilic, growth hormone–producing cells of the anterior pituitary gland become excessively active, and sometimes even acidophilic tumors occur in the gland. As a result, large quantities of growth hormone are produced. All body tissues grow rapidly, including the bones. If the condition occurs before adolescence, before the epiphyses of the long bones have become fused with the shafts, height increases so that the person becomes a giant—up to 8 feet tall.
The giant ordinarily has hyperglycemia, and the beta cells of the islets of Langerhans in the pancreas are prone to degenerate because they become overactive owing to the hyperglycemia. Consequently, in about 10 percent of giants, full-blown diabetes mellitus eventually develops.
In most giants, panhypopituitarism eventually develops if they remain untreated because the gigantism is usually caused by a tumor of the pituitary gland that grows until the gland is destroyed. This eventual general deficiency of pituitary hormones usually causes death in early adulthood. However, once gigantism is diagnosed, further effects can often be blocked by microsurgical removal of the tumor or by irradiation of the pituitary gland.
 Acromegaly.
Acromegaly.
If an acidophilic tumor occurs after adolescence—that is, after the epiphyses of the long bones have fused with the shafts—the person cannot grow taller, but the bones can become thicker and the soft tissues can continue to grow. This condition, shown in Figure 76-8, is known as acromegaly. Enlargement is especially marked in the bones of the hands and feet and in the membranous bones, including the cranium, nose, bosses on the forehead, supraorbital ridges, lower jawbone, and portions of the vertebrae, because their growth does not cease at adolescence. Consequently, the lower jaw protrudes forward, sometimes as much as half an inch, the forehead slants forward because of excess development of the supraorbital ridges, the nose increases to as much as twice normal size, the feet require size 14 or larger shoes, and the fingers become extremely thickened so that the hands are almost twice normal size. In addition to these effects, changes in the vertebrae ordinarily cause a hunched back, which is known clinically as kyphosis. Finally, many soft tissue organs, such as the tongue, the liver, and especially the kidneys, become greatly enlarged.
 Possible Role of Decreased Growth Hormone Secretion in Causing Changes Associated with Aging
Possible Role of Decreased Growth Hormone Secretion in Causing Changes Associated with Aging
In people who have lost the ability to secrete growth hormone, some features of the aging process accelerate. For instance, a 50-year-old person who has been without growth hormone for many years may have the appearance of a person aged 65 years. The aged appearance seems to result mainly from decreased protein deposition in most tissues of the body and increased fat deposition in its place. The physical and physiological effects are increased wrinkling of the skin, diminished rates of function of some of the organs, and diminished muscle mass and strength.
As one ages, the average plasma concentration of growth hormone in an otherwise normal person changes approximately as follows:
| Age (years) | ng/ml |
| 5 to 20 | 6 |
| 20 to 40 | 3 |
| 40 to 70 | 1.6 |
Thus, it is possible that some of the normal aging effects result from diminished growth hormone secretion. In fact, some studies of growth hormone therapy in older people have demonstrated three important beneficial effects: (1) increased protein deposition in the body, especially in the muscles; (2) decreased fat deposits; and (3) a feeling of increased energy. Other studies, however, have shown that treatment of elderly patients with recombinant growth hormone may produce several undesirable adverse effects including insulin resistance and diabetes, edema, carpal tunnel syndrome, and arthralgias (joint pain). Therefore, recombinant growth hormone therapy is generally not recommended for use in healthy elderly patients with normal endocrine function. ![]()
Posterior Pituitary Gland and Its Relation to the Hypothalamus
The posterior pituitary gland, also called the neurohypophysis, is composed mainly of glial-like cells called pituicytes. The pituicytes do not secrete hormones; they act simply as a supporting structure for large numbers of terminal nerve fibers and terminal nerve endings from nerve tracts that originate in the supraoptic and paraventricular nuclei of the hypothalamus, as shown in Figure 76-9. These tracts pass to the neurohypophysis through the pituitary stalk (hypophysial stalk). The nerve endings are bulbous knobs that contain many secretory granules. These endings lie on the surfaces of capillaries, where they secrete two posterior pituitary hormones: (1) antidiuretic hormone (ADH), also called vasopressin, and (2) oxytocin.
If the pituitary stalk is cut above the pituitary gland but the entire hypothalamus is left intact, the posterior pituitary hormones continue to be secreted normally, after a transient decrease for a few days; they are then secreted by the cut ends of the fibers within the hypothalamus and not by the nerve endings in the posterior pituitary. The reason for this is that the hormones are initially synthesized in the cell bodies of the supraoptic and paraventricular nuclei and are then transported in combination with “carrier” proteins called neurophysins down to the nerve endings in the posterior pituitary gland, requiring several days to reach the gland.
ADH is formed primarily in the supraoptic nuclei, whereas oxytocin is formed primarily in the paraventricular nuclei. Each of these nuclei can synthesize about one sixth as much of the second hormone as of its primary hormone.
When nerve impulses are transmitted downward along the fibers from the supraoptic or paraventricular nuclei, the hormone is immediately released from the secretory granules in the nerve endings by the usual secretory mechanism of exocytosis and is absorbed into adjacent capillaries. Both the neurophysin and the hormone are secreted together, but because they are only loosely bound to each other, the hormone separates almost immediately. The neurophysin has no known function after leaving the nerve terminals.
 Chemical Structures of Antidiuretic Hormone and Oxytocin
Chemical Structures of Antidiuretic Hormone and Oxytocin
Both oxytocin and ADH (vasopressin) are polypeptides, each containing nine amino acids. Their amino acid sequences are the following:
Vasopressin: Cys-Tyr-Phe-Gln-Asn-Cys-Pro-Arg-GlyNH2
Oxytocin: Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-GlyNH2
Note that these two hormones are almost identical except that in vasopressin, phenylalanine and arginine replace isoleucine and leucine of the oxytocin molecule. The similarity of the molecules explains their partial functional similarities. ![]()
Physiological Functions of Antidiuretic Hormone
The injection of extremely minute quantities of ADH—as small as 2 nanograms—can cause decreased excretion of water by the kidneys (antidiuresis). This antidiuretic effect is discussed in Chapter 29. Briefly, in the absence of ADH, the collecting tubules and ducts become almost impermeable to water, which prevents significant reabsorption of water and therefore allows extreme loss of water into the urine, also causing extreme dilution of the urine. Conversely, in the presence of ADH, the permeability of the collecting ducts and tubules to water increases greatly and allows most of the water to be reabsorbed as the tubular fluid passes through these ducts, thereby conserving water in the body and producing very concentrated urine.
Without ADH, the luminal membranes of the tubular epithelial cells of the collecting ducts are almost impermeable to water. However, immediately inside the cell membrane are a large number of special vesicles that have highly water-permeable pores called aquaporins. When ADH acts on the cell, it first combines with membrane receptors that activate adenylyl cyclase and cause the formation of cAMP inside the tubular cell cytoplasm. This formation causes phosphorylation of elements in the special vesicles, which then causes the vesicles to insert into the apical cell membranes, thus providing many areas of high water permeability. All this occurs within 5 to 10 minutes. Then, in the absence of ADH, the entire process reverses in another 5 to 10 minutes. Thus, this process temporarily provides many new pores that allow free diffusion of water from the tubular fluid through the tubular epithelial cells and into the renal interstitial fluid. Water is then absorbed from the collecting tubules and ducts by osmosis, as explained in Chapter 29 in relation to the urine-concentrating mechanism of the kidneys.
Regulation of Antidiuretic Hormone Production
Increased Extracellular Fluid Osmolarity Stimulates ADH Secretion.
When a concentrated electrolyte solution is injected into the artery that supplies the hypothalamus, the ADH neurons in the supraoptic and paraventricular nuclei immediately transmit impulses into the posterior pituitary to release large quantities of ADH into the circulating blood, sometimes increasing the ADH secretion to as high as 20 times normal. Conversely, injection of a dilute solution into this artery causes cessation of the impulses and therefore almost total cessation of ADH secretion. Thus, the concentration of ADH in the body fluids can change from small amounts to large amounts, or vice versa, in only a few minutes.
Somewhere in or near the hypothalamus are modified neuron receptors called osmoreceptors. When the extracellular fluid becomes too concentrated, fluid is pulled by osmosis out of the osmoreceptor cell, decreasing its size and initiating appropriate nerve signals in the hypothalamus to cause additional ADH secretion. Conversely, when the extracellular fluid becomes too dilute, water moves by osmosis in the opposite direction, into the cell, which decreases the signal for ADH secretion. Although some researchers place these osmoreceptors in the hypothalamus (possibly even in the supraoptic nuclei), others believe that they are located in the organum vasculosum, a highly vascular structure in the anteroventral wall of the third ventricle.
Regardless of the mechanism, concentrated body fluids stimulate the supraoptic nuclei, whereas dilute body fluids inhibit them. A feedback control system is available to control the total osmotic pressure of the body fluids.
Further details on the control of ADH secretion and the role of ADH in controlling renal function and body fluid osmolality are presented in Chapter 29.
Low Blood Volume and Low Blood Pressure Stimulate ADH Secretion—Vasoconstrictor Effects of ADH.
Whereas minute concentrations of ADH cause increased water conservation by the kidneys, higher concentrations of ADH have a potent effect of constricting the arterioles throughout the body and therefore increasing the arterial pressure. For this reason, ADH has another name, vasopressin.
One of the stimuli for causing intense ADH secretion is decreased blood volume. This occurs strongly when the blood volume decreases 15 to 25 percent or more; the secretory rate then sometimes rises to as high as 50 times normal. The cause of this effect is the following.
The atria have stretch receptors that are excited by overfilling. When excited, they send signals to the brain to inhibit ADH secretion. Conversely, when the receptors are unexcited as a result of underfilling, the opposite occurs, with greatly increased ADH secretion. Decreased stretch of the baroreceptors of the carotid, aortic, and pulmonary regions also stimulates ADH secretion. For further details about this blood volume-pressure feedback mechanism, refer to Chapter 29.
Physiological Functions of Oxytocin
Oxytocin Causes Contraction of the Pregnant Uterus.
The hormone oxytocin, in accordance with its name, powerfully stimulates contraction of the pregnant uterus, especially toward the end of gestation. Therefore, many obstetricians believe that this hormone is at least partially responsible for causing birth of the baby. This belief is supported by the following facts: (1) In a hypophysectomized animal, the duration of labor is prolonged, indicating a possible effect of oxytocin during delivery; (2) the amount of oxytocin in the plasma increases during labor, especially during the last stage; and (3) stimulation of the cervix in a pregnant animal elicits nervous signals that pass to the hypothalamus and cause increased secretion of oxytocin. These effects and this possible mechanism for aiding in the birth process are discussed in more detail in Chapter 83.
Oxytocin Aids in Milk Ejection by the Breasts.
Oxytocin also plays an especially important role in lactation—a role that is far better understood than its role in delivery. In lactation, oxytocin causes milk to be expressed from the alveoli into the ducts of the breast so that the baby can obtain it by suckling.
This mechanism works as follows: The suckling stimulus on the nipple of the breast causes signals to be transmitted through sensory nerves to the oxytocin neurons in the paraventricular and supraoptic nuclei in the hypothalamus, which causes release of oxytocin by the posterior pituitary gland. The oxytocin is then carried by the blood to the breasts, where it causes contraction of myoepithelial cells that lie outside of and form a latticework surrounding the alveoli of the mammary glands. In less than a minute after the beginning of suckling, milk begins to flow. This mechanism is called milk letdown or milk ejection. It is discussed further in Chapter 83 in relation to the physiology of lactation.





