Thyroid Metabolic Hormones
The thyroid gland, located immediately below the larynx on each side of and anterior to the trachea, is one of the largest of the endocrine glands, normally weighing 15 to 20 grams in adults. The thyroid secretes two major hormones, thyroxine and triiodothyronine, commonly called T4 and T3, respectively. Both of these hormones profoundly increase the metabolic rate of the body. Complete lack of thyroid secretion usually causes the basal metabolic rate to fall 40 to 50 percent below normal, and extreme excesses of thyroid secretion can increase the basal metabolic rate to 60 to 100 percent above normal. Thyroid secretion is controlled primarily by thyroid-stimulating hormone (TSH) secreted by the anterior pituitary gland.
The thyroid gland also secretes calcitonin, a hormone involved in calcium metabolism that is discussed in Chapter 80.
The purpose of this chapter is to discuss the formation and secretion of the thyroid hormones, their metabolic functions, and regulation of their secretion.
Synthesis and Secretion of the Thyroid Metabolic Hormones
About 93 percent of the metabolically active hormones secreted by the thyroid gland is thyroxine, and 7 percent is triiodothyronine. However, almost all the thyroxine is eventually converted to triiodothyronine in the tissues, so both are functionally important. The functions of these two hormones are qualitatively the same, but they differ in rapidity and intensity of action. Triiodothyronine is about four times as potent as thyroxine, but it is present in the blood in much smaller quantities and persists for a much shorter time compared with thyroxine.
Physiological Anatomy of the Thyroid Gland
As shown in Figure 77-1, the thyroid gland is composed of large numbers of closed follicles (100 to 300 micrometers in diameter) that are filled with a secretory substance called colloid and lined with cuboidal epithelial cells that secrete into the interior of the follicles. The major constituent of colloid is the large glycoprotein thyroglobulin, which contains the thyroid hormones. Once the secretion has entered the follicles, it must be absorbed back through the follicular epithelium into the blood before it can function in the body. The thyroid gland has a blood flow about five times the weight of the gland each minute, which is a blood supply as great as that of any other area of the body, with the possible exception of the adrenal cortex.
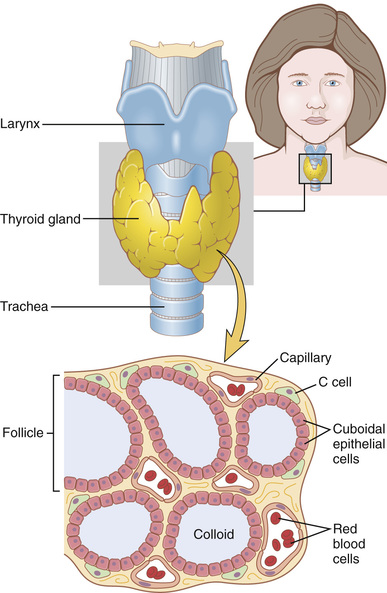
The thyroid gland also contains C cells that secrete calcitonin, a hormone that contributes to regulation of plasma calcium ion concentration, as discussed in Chapter 80.
Iodine is Required for Formation of Thyroxine
To form normal quantities of thyroxine, about 50 milligrams of ingested iodine in the form of iodides are required each year, or about 1 mg/week. To prevent iodine deficiency, common table salt is iodized with about 1 part sodium iodide to every 100,000 parts sodium chloride.
Fate of Ingested Iodides.
Iodides ingested orally are absorbed from the gastrointestinal tract into the blood in about the same manner as chlorides. Normally, most of the iodides are rapidly excreted by the kidneys, but only after about one fifth are selectively removed from the circulating blood by the cells of the thyroid gland and used for synthesis of the thyroid hormones.
Iodide Pump—the Sodium-Iodide Symporter (Iodide Trapping)
The first stage in the formation of thyroid hormones, shown in Figure 77-2, is transport of iodides from the blood into the thyroid glandular cells and follicles. The basal membrane of the thyroid cell has the specific ability to pump the iodide actively to the interior of the cell. This pumping is achieved by the action of a sodium-iodide symporter, which co-transports one iodide ion along with two sodium ions across the basolateral (plasma) membrane into the cell. The energy for transporting iodide against a concentration gradient comes from the sodium-potassium adenosine triphosphatase (ATPase) pump, which pumps sodium out of the cell, thereby establishing a low intracellular sodium concentration and a gradient for facilitated diffusion of sodium into the cell.
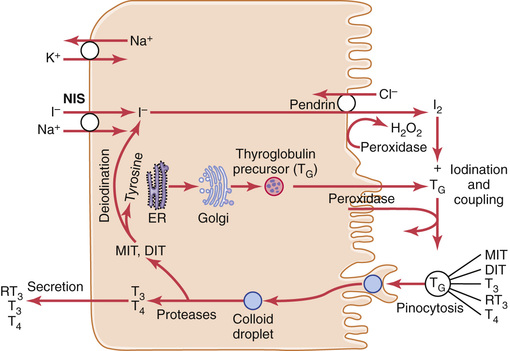
This process of concentrating the iodide in the cell is called iodide trapping. In a normal gland, the iodide pump concentrates the iodide to about 30 times its concentration in the blood. When the thyroid gland becomes maximally active, this concentration ratio can rise to as high as 250 times. The rate of iodide trapping by the thyroid is influenced by several factors, the most important being the concentration of TSH; TSH stimulates and hypophysectomy greatly diminishes the activity of the iodide pump in thyroid cells.
Iodide is transported out of the thyroid cells across the apical membrane into the follicle by a chloride-iodide ion counter-transporter molecule called pendrin. The thyroid epithelial cells also secrete into the follicle thyroglobulin that contains tyrosine amino acids to which the iodine will bind, as discussed in the next section.
Thyroglobulin and Chemistry of Thyroxine and Triiodothyronine Formation
Formation and Secretion of Thyroglobulin by the Thyroid Cells.
The thyroid cells are typical protein-secreting glandular cells, as shown in Figure 77-2. The endoplasmic reticulum and Golgi apparatus synthesize and secrete into the follicles a large glycoprotein molecule called thyroglobulin, with a molecular weight of about 335,000.
Each molecule of thyroglobulin contains about 70 tyrosine amino acids, and they are the major substrates that combine with iodine to form the thyroid hormones. Thus, the thyroid hormones form within the thyroglobulin molecule. That is, the thyroxine and triiodothyronine hormones formed from the tyrosine amino acids remain part of the thyroglobulin molecule during synthesis of the thyroid hormones and even afterward as stored hormones in the follicular colloid.
Oxidation of the Iodide Ion.
The first essential step in the formation of thyroid hormones is conversion of iodide ions to an oxidized form of iodine, either nascent iodine (I0) or I3−, which is then capable of combining directly with the amino acid tyrosine. This oxidation of iodine is promoted by the enzyme peroxidase and its accompanying hydrogen peroxide, which provide a potent system capable of oxidizing iodides. The peroxidase is either located in the apical membrane of the cell or attached to it, thus providing the oxidized iodine at exactly the point in the cell where the thyroglobulin molecule issues forth from the Golgi apparatus and through the cell membrane into the stored thyroid gland colloid. When the peroxidase system is blocked or when it is hereditarily absent from the cells, the rate of formation of thyroid hormones falls to zero.
Iodination of Tyrosine and Formation of the Thyroid Hormones—“Organification” of Thyroglobulin.
The binding of iodine with the thyroglobulin molecule is called organification of the thyroglobulin. Oxidized iodine even in the molecular form will bind directly but slowly with the amino acid tyrosine. In thyroid cells, however, the oxidized iodine is associated with thyroid peroxidase enzyme (Figure 77-2) that causes the process to occur within seconds or minutes. Therefore, almost as rapidly as thyroglobulin is released from the Golgi apparatus or as it is secreted through the apical cell membrane into the follicle, iodine binds with about one sixth of the tyrosine amino acids within the thyroglobulin molecule.
Figure 77-3 shows the successive stages of iodination of tyrosine and final formation of the two important thyroid hormones, thyroxine and triiodothyronine. Tyrosine is first iodized to monoiodotyrosine and then to diiodotyrosine. Then, during the next few minutes, hours, and even days, more and more of the iodotyrosine residues become coupled with one another.
The major hormonal product of the coupling reaction is the molecule thyroxine (T4), which is formed when two molecules of diiodotyrosine are joined together; the thyroxine then remains part of the thyroglobulin molecule. Or one molecule of monoiodotyrosine couples with one molecule of diiodotyrosine to form triiodothyronine (T3), which represents about one fifteenth of the final hormones. Small amounts of reverse T3 (RT3) are formed by coupling of diiodotyrosine with monoiodotyrosine, but RT3 does not appear to be of functional significance in humans.
Storage of Thyroglobulin.
The thyroid gland is unusual among the endocrine glands in its ability to store large amounts of hormone. After synthesis of the thyroid hormones has run its course, each thyroglobulin molecule contains up to 30 thyroxine molecules and a few triiodothyronine molecules. In this form, the thyroid hormones are stored in the follicles in an amount sufficient to supply the body with its normal requirements of thyroid hormones for 2 to 3 months. Therefore, when synthesis of thyroid hormone ceases, the physiological effects of deficiency are not observed for several months.
Release of Thyroxine and Triiodothyronine From the Thyroid Gland
Most of the thyroglobulin is not released into the circulating blood; instead, thyroxine and triiodothyronine are cleaved from the thyroglobulin molecule, and then these free hormones are released. This process occurs as follows: The apical surface of thyroid cells sends out pseudopod extensions that close around small portions of the colloid to form pinocytic vesicles that enter the apex of the thyroid cell. Then lysosomes in the cell cytoplasm immediately fuse with these vesicles to form digestive vesicles containing digestive enzymes from the lysosomes mixed with the colloid. Multiple proteases among the enzymes digest the thyroglobulin molecules and release thyroxine and triiodothyronine in free form, which then diffuse through the base of the thyroid cell into the surrounding capillaries. Thus, the thyroid hormones are released into the blood.
Some of the thyroglobulin in the colloid enters the thyroid cell by endocytosis after binding to megalin, a protein located on the lumen membrane of the cells. The megalin-thyroglobulin complex is then carried across the cell by transcytosis to the basolateral membrane, where a portion of the megalin remains bound to thyroglobulin and is released into the capillary blood.
About three quarters of the iodinated tyrosine in the thyroglobulin never become thyroid hormones but remain monoiodotyrosine and diiodotyrosine. During the digestion of the thyroglobulin molecule to cause release of thyroxine and triiodothyronine, these iodinated tyrosines also are freed from the thyroglobulin molecules. However, they are not secreted into the blood. Instead, their iodine is cleaved from them by a deiodinase enzyme that makes virtually all this iodine available again for recycling within the gland for forming additional thyroid hormones. In the congenital absence of this deiodinase enzyme, many persons become iodine deficient because of failure of this recycling process.
Daily Rate of Secretion of Thyroxine and Triiodothyronine.
About 93 percent of the thyroid hormone released from the thyroid gland is normally thyroxine and only 7 percent is triiodothyronine. However, during the ensuing few days, about one half of the thyroxine is slowly deiodinated to form additional triiodothyronine. Therefore, the hormone finally delivered to and used by the tissues is mainly triiodothyronine—a total of about 35 micrograms of triiodothyronine per day.
Transport of Thyroxine and Triiodothyronine to Tissues
Thyroxine and Triiodothyronine Are Bound to Plasma Proteins.
Upon entering the blood, more than 99 percent of the thyroxine and triiodothyronine combines immediately with several of the plasma proteins, all of which are synthesized by the liver. They combine mainly with thyroxine-binding globulin and much less so with thyroxine-binding prealbumin and albumin.
Thyroxine and Triiodothyronine Are Released Slowly to Tissue Cells.
Because of high affinity of the plasma-binding proteins for the thyroid hormones, these substances—in particular, thyroxine—are released to the tissue cells slowly. Half the thyroxine in the blood is released to the tissue cells about every 6 days, whereas half the triiodothyronine—because of its lower affinity—is released to the cells in about 1 day.
Upon entering the tissue cells, both thyroxine and triiodothyronine again bind with intracellular proteins, with the thyroxine binding more strongly than the triiodothyronine. Therefore, they are again stored, but this time in the target cells themselves, and they are used slowly over a period of days or weeks.
Thyroid Hormones Have Slow Onset and Long Duration of Action.
After injection of a large quantity of thyroxine into a human being, essentially no effect on the metabolic rate can be discerned for 2 to 3 days, thereby demonstrating that there is a long latent period before thyroxine activity begins. Once activity does begin, it increases progressively and reaches a maximum in 10 to 12 days, as shown in Figure 77-4. Thereafter, it decreases with a half-life of about 15 days. Some of the activity persists for as long as 6 weeks to 2 months.
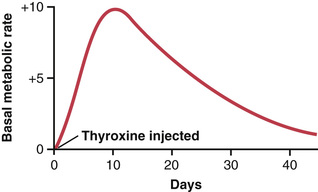
The actions of triiodothyronine occur about four times as rapidly as those of thyroxine, with a latent period as short as 6 to 12 hours and maximal cellular activity occurring within 2 to 3 days.
Most of the latency and the prolonged period of action of these hormones are probably caused by their binding with proteins both in the plasma and in the tissue cells, followed by their slow release. However, we shall see in subsequent discussions that part of the latent period also results from the manner in which these hormones perform their functions in the cells.
Physiological Functions of the Thyroid Hormones
Thyroid Hormones Increase Transcription of Large Numbers of Genes
The general effect of thyroid hormone is to activate nuclear transcription of large numbers of genes (Figure 77-5). Therefore, in virtually all cells of the body, great numbers of protein enzymes, structural proteins, transport proteins, and other substances are synthesized. The net result is a generalized increase in functional activity throughout the body.
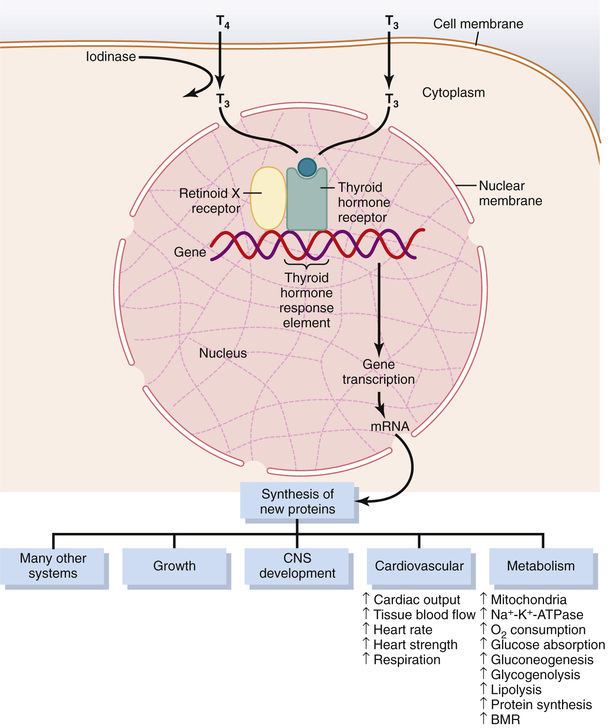
Most of the Thyroxine Secreted by the Thyroid Is Converted to Triiodothyronine.
Before acting on the genes to increase genetic transcription, one iodide is removed from almost all the thyroxine, thus forming triiodothyronine. Intracellular thyroid hormone receptors have a high affinity for triiodothyronine. Consequently, more than 90 percent of the thyroid hormone molecules that bind with the receptors is triiodothyronine.
Thyroid Hormones Activate Nuclear Receptors.
The thyroid hormone receptors are either attached to the DNA genetic strands or located in proximity to them. The thyroid hormone receptor usually forms a heterodimer with retinoid X receptor (RXR) at specific thyroid hormone response elements on the DNA. After binding with thyroid hormone, the receptors become activated and initiate the transcription process. Large numbers of different types of messenger RNA are then formed, followed within another few minutes or hours by RNA translation on the cytoplasmic ribosomes to form hundreds of new intracellular proteins. However, not all the proteins are increased by similar percentages—some are increased only slightly, and others at least as much as sixfold. It is believed that most of the actions of thyroid hormone result from the subsequent enzymatic and other functions of these new proteins.
Thyroid hormones also appear to have nongenomic cellular effects that are independent of their effects on gene transcription. For example, some effects of thyroid hormones occur within minutes, too rapidly to be explained by changes in protein synthesis, and are not affected by inhibitors of gene transcription and translation. Such actions have been described in several tissues, including the heart and pituitary, as well as adipose tissue. The site of nongenomic thyroid hormone action appears to be the plasma membrane, cytoplasm, and perhaps some cell organelles such as mitochondria. Nongenomic actions of thyroid hormone include the regulation of ion channels and oxidative phosphorylation and appear to involve the activation of intracellular secondary messengers such as cyclic adenosine monophosphate (cAMP) or protein kinase signaling cascades.
Thyroid Hormones Increase Cellular Metabolic Activity
The thyroid hormones increase the metabolic activities of almost all the tissues of the body. The basal metabolic rate can increase to 60 to 100 percent above normal when large quantities of the hormones are secreted. The rate of utilization of foods for energy is greatly accelerated. Although the rate of protein synthesis is increased, at the same time the rate of protein catabolism is also increased. The growth rate of young people is greatly accelerated. The mental processes are excited, and the activities of most of the other endocrine glands are increased.
Thyroid Hormones Increase the Number and Activity of Mitochondria.
When thyroxine or triiodothyronine is given to an animal, the mitochondria in most cells of the animal's body increase in size and number. Furthermore, the total membrane surface area of the mitochondria increases almost directly in proportion to the increased metabolic rate of the whole animal. Therefore, one of the principal functions of thyroxine might be simply to increase the number and activity of mitochondria, which in turn increases the rate of formation of adenosine triphosphate to energize cellular function. However, the increase in the number and activity of mitochondria could be the result of increased activity of the cells, as well as the cause of the increase.
Thyroid Hormones Increase Active Transport of Ions Through Cell Membranes.
One of the enzymes that increases its activity in response to thyroid hormone is Na-K-ATPase. This increased activity in turn increases the rate of transport of both sodium and potassium ions through the cell membranes of some tissues. Because this process uses energy and increases the amount of heat produced in the body, it has been suggested that this might be one of the mechanisms by which thyroid hormone increases the body's metabolic rate. In fact, thyroid hormone also causes the cell membranes of most cells to become leaky to sodium ions, which further activates the sodium pump and further increases heat production.
Effect of Thyroid Hormone on Growth
Thyroid hormone has both general and specific effects on growth. For instance, it has long been known that thyroid hormone is essential for the metamorphic change of the tadpole into the frog.
In humans, the effect of thyroid hormone on growth is manifest mainly in growing children. In children with hypothyroidism, the rate of growth is greatly retarded. In children with hyperthyroidism, excessive skeletal growth often occurs, causing the child to become considerably taller at an earlier age. However, the bones also mature more rapidly and the epiphyses close at an early age, so the duration of growth and the eventual height of the adult actually may be shortened.
An important effect of thyroid hormone is to promote growth and development of the brain during fetal life and for the first few years of postnatal life. If the fetus does not secrete sufficient quantities of thyroid hormone, growth and maturation of the brain both before birth and afterward are greatly retarded and the brain remains smaller than normal. Without specific thyroid therapy within days or weeks after birth, the child without a thyroid gland will remain mentally deficient throughout life. This condition is discussed later in the chapter.
Effects of Thyroid Hormone on Specific Body Functions
Stimulation of Carbohydrate Metabolism.
Thyroid hormone stimulates almost all aspects of carbohydrate metabolism, including rapid glucose uptake by cells, enhanced glycolysis, enhanced gluconeogenesis, increased rate of absorption from the gastrointestinal tract, and even increased insulin secretion with its resultant secondary effects on carbohydrate metabolism. All these effects probably result from the overall increase in cellular metabolic enzymes caused by thyroid hormone.
Stimulation of Fat Metabolism.
Essentially all aspects of fat metabolism are also enhanced under the influence of thyroid hormone. In particular, lipids are mobilized rapidly from the fat tissue, which decreases the fat stores of the body to a greater extent than almost any other tissue element. Mobilization of lipids from fat tissue also increases the free fatty acid concentration in the plasma and greatly accelerates the oxidation of free fatty acids by the cells.
Effect on Plasma and Liver Fats.
Increased thyroid hormone decreases the concentrations of cholesterol, phospholipids, and triglycerides in the plasma, even though it increases the free fatty acids. Conversely, decreased thyroid secretion greatly increases the plasma concentrations of cholesterol, phospholipids, and triglycerides and almost always causes excessive deposition of fat in the liver as well. The large increase in circulating plasma cholesterol in prolonged hypothyroidism is often associated with severe atherosclerosis, as discussed in Chapter 69.
One of the mechanisms by which thyroid hormone decreases plasma cholesterol concentration is to increase significantly cholesterol secretion in the bile and consequent loss in the feces. A possible mechanism for the increased cholesterol secretion is that thyroid hormone induces increased numbers of low-density lipoprotein receptors on the liver cells, leading to rapid removal of low-density lipoproteins from the plasma by the liver and subsequent secretion of cholesterol in these lipoproteins by the liver cells.
Increased Requirement for Vitamins.
Because thyroid hormone increases the quantities of many bodily enzymes and because vitamins are essential parts of some of the enzymes or coenzymes, thyroid hormone increases the need for vitamins. Therefore, a relative vitamin deficiency can occur when excess thyroid hormone is secreted, unless at the same time increased quantities of vitamins are made available.
Increased Basal Metabolic Rate.
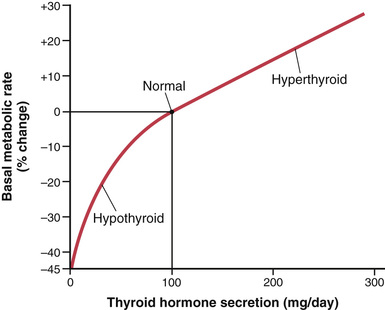
Because thyroid hormone increases metabolism in almost all cells of the body, excessive quantities of the hormone can occasionally increase the basal metabolic rate 60 to 100 percent above normal. Conversely, when no thyroid hormone is produced, the basal metabolic rate falls to almost one-half normal. Figure 77-6 shows the approximate relation between the daily supply of thyroid hormones and the basal metabolic rate. Extreme amounts of the hormones are required to cause high basal metabolic rates.
Decreased Body Weight.
A greatly increased amount of thyroid hormone almost always decreases body weight, and a greatly decreased amount of thyroid hormone almost always increases body weight; however, these effects do not always occur because thyroid hormone also increases the appetite, which may counterbalance the change in the metabolic rate.
Increased Blood Flow and Cardiac Output.
Increased metabolism in the tissues causes more rapid utilization of oxygen than normal and the release of greater than normal quantities of metabolic end products from the tissues. These effects cause vasodilation in most body tissues, thus increasing blood flow. The rate of blood flow in the skin especially increases because of the increased need for heat elimination from the body. As a consequence of the increased blood flow, cardiac output also increases, sometimes rising to 60 percent or more above normal when excessive thyroid hormone is present and falling to only 50 percent of normal in severe hypothyroidism.
Increased Heart Rate.
The heart rate increases considerably more under the influence of thyroid hormone than would be expected from the increase in cardiac output. Therefore, thyroid hormone seems to have a direct effect on the excitability of the heart, which in turn increases the heart rate. This effect is especially important because the heart rate is one of the sensitive physical signs that the clinician uses in determining whether a patient has excessive or diminished thyroid hormone production.
Increased Heart Strength.
The increased enzymatic activity caused by increased thyroid hormone production apparently increases the strength of the heart when only a slight excess of thyroid hormone is secreted. This effect is analogous to the increase in heart strength that occurs in mild fevers and during exercise. However, when thyroid hormone is increased markedly, heart muscle strength becomes depressed because of long-term excessive protein catabolism. Indeed, some severely thyrotoxic patients die of cardiac decompensation secondary to myocardial failure and increased cardiac load imposed by the increase in cardiac output.
Normal Arterial Pressure.
The mean arterial pressure usually remains about normal after administration of thyroid hormone. Because of increased blood flow through the tissues between heartbeats, the pulse pressure is often increased, with the systolic pressure elevated 10 to 15 mm Hg in hyperthyroidism and the diastolic pressure reduced a corresponding amount.
Increased Respiration.
The increased rate of metabolism increases the utilization of oxygen and the formation of carbon dioxide; these effects activate all the mechanisms that increase the rate and depth of respiration.
Increased Gastrointestinal Motility.
In addition to increased appetite and food intake, which has been discussed, thyroid hormone increases both the rates of secretion of the digestive juices and the motility of the gastrointestinal tract. Hyperthyroidism therefore often results in diarrhea, whereas lack of thyroid hormone can cause constipation.
Excitatory Effects on the Central Nervous System.
In general, thyroid hormone increases the rapidity of cerebration, although thought processes may be dissociated; conversely, lack of thyroid hormone decreases rapidity of cerebration. A person with hyperthyroidism is likely to be extremely nervous and have many psychoneurotic tendencies, such as anxiety complexes, extreme worry, and paranoia.
Effect on the Function of the Muscles.
A slight increase in thyroid hormone usually makes the muscles react with vigor, but when the quantity of hormone becomes excessive, the muscles become weakened because of excess protein catabolism. Conversely, lack of thyroid hormone causes the muscles to become sluggish, and they relax slowly after a contraction.
Muscle Tremor.
One of the most characteristic signs of hyperthyroidism is a fine muscle tremor. This symptom is not the coarse tremor that occurs in Parkinson's disease or when a person shivers because it occurs at the rapid frequency of 10 to 15 times per second. The tremor can be observed easily by placing a sheet of paper on the extended fingers and noting the degree of vibration of the paper. This tremor is believed to be caused by increased reactivity of the neuronal synapses in the areas of the spinal cord that control muscle tone. The tremor is an important means for assessing the degree of thyroid hormone effect on the central nervous system.
Effect on Sleep.
Because of the exhausting effect of thyroid hormone on the musculature and on the central nervous system, persons with hyperthyroidism often have a feeling of constant tiredness, but because of the excitable effects of thyroid hormone on the synapses, it is difficult to sleep. Conversely, extreme somnolence is characteristic of hypothyroidism, with sleep sometimes lasting 12 to 14 hours a day.
Effect on Other Endocrine Glands.
Increased thyroid hormone increases the rates of secretion of several other endocrine glands, but it also increases the need of the tissues for the hormones. For instance, increased thyroxine secretion increases the rate of glucose metabolism almost everywhere in the body and therefore causes a corresponding need for increased insulin secretion by the pancreas. Also, thyroid hormone increases many metabolic activities related to bone formation and, as a consequence, increases the need for parathyroid hormone. Thyroid hormone also increases the rate at which adrenal glucocorticoids are inactivated by the liver. This increased rate of inactivation leads to feedback increase in adrenocorticotropic hormone production by the anterior pituitary and, therefore, an increased rate of glucocorticoid secretion by the adrenal glands.
Effect of Thyroid Hormone on Sexual Function.
For normal sexual function, thyroid secretion needs to be approximately normal. In men, lack of thyroid hormone is likely to cause loss of libido; a great excess of the hormone, however, sometimes causes impotence.
In women, lack of thyroid hormone often causes menorrhagia and polymenorrhea—that is, excessive and frequent menstrual bleeding, respectively. Yet, strangely enough, in other women a lack of thyroid hormone may cause irregular periods and occasionally even amenorrhea (absence of menstrual bleeding).
Hypothyroidism in women, as in men, is likely to result in a greatly decreased libido. To make the picture still more confusing, in women with hyperthyroidism, oligomenorrhea (greatly reduced bleeding) is common, and occasionally amenorrhea occurs.
The action of thyroid hormone on the gonads cannot be pinpointed to a specific function but probably results from a combination of direct metabolic effects on the gonads, as well as excitatory and inhibitory feedback effects operating through the anterior pituitary hormones that control sexual functions.
Regulation of Thyroid Hormone Secretion
To maintain normal levels of metabolic activity in the body, precisely the right amount of thyroid hormone must be secreted at all times; to achieve this ideal level of secretion, specific feedback mechanisms operate through the hypothalamus and anterior pituitary gland to control the rate of thyroid secretion. These mechanisms are described in the following sections.
TSH (From the Anterior Pituitary Gland) Increases Thyroid Secretion
TSH, also known as thyrotropin, is an anterior pituitary hormone; it is a glycoprotein with a molecular weight of about 28,000. This hormone, also discussed in Chapter 75, increases secretion of thyroxine and triiodothyronine by the thyroid gland. It has the following specific effects on the thyroid gland:
1. Increased proteolysis of the thyroglobulin that has already been stored in the follicles, releasing the thyroid hormones into the circulating blood and diminishing the follicular substance
2. Increased activity of the iodide pump, which increases the rate of “iodide trapping” in the glandular cells, sometimes increasing the ratio of intracellular to extracellular iodide concentration in the glandular substance to as much as eight times normal
3. Increased iodination of tyrosine to form the thyroid hormones
4. Increased size and increased secretory activity of the thyroid cells
5. Increased number of thyroid cells plus a change from cuboidal to columnar cells and much infolding of the thyroid epithelium into the follicles
In summary, TSH increases all the known secretory activities of the thyroid glandular cells.
The most important early effect after administration of TSH is to initiate proteolysis of thyroglobulin, which causes release of thyroxine and triiodothyronine into the blood within 30 minutes. The other effects require hours or even days and weeks to develop fully.
Cyclic Adenosine Monophosphate Mediates the Stimulatory Effect of TSH.
Most of the varied effects of TSH on the thyroid cell result from activation of the “second messenger” cAMP system of the cell.
The first event in this activation is binding of TSH with specific TSH receptors on the basal membrane surfaces of the thyroid cell. This binding then activates adenylyl cyclase in the membrane, which increases the formation of cAMP inside the cell. Finally, the cAMP acts as a second messenger to activate protein kinase, which causes multiple phosphorylations throughout the cell. The result is both an immediate increase in secretion of thyroid hormones and prolonged growth of the thyroid glandular tissue itself.
This method for control of thyroid cell activity is similar to the function of cAMP as a “second messenger” in many other target tissues of the body, as discussed in Chapter 75.
Anterior Pituitary Secretion of TSH is Regulated by Thyrotropin-Releasing Hormone From the Hypothalamus
Anterior pituitary secretion of TSH is controlled by a hypothalamic hormone, thyrotropin-releasing hormone (TRH), which is secreted by nerve endings in the median eminence of the hypothalamus. From the median eminence, TRH is then transported to the anterior pituitary by way of the hypothalamic-hypophysial portal blood, as explained in Chapter 75.
TRH is a tripeptide amide—pyroglutamyl-histidyl-proline-amide. TRH stimulates the anterior pituitary gland cells to increase their output of TSH. When the blood portal system from the hypothalamus to the anterior pituitary gland becomes blocked, the rate of secretion of TSH by the anterior pituitary decreases greatly but is not reduced to zero.
The molecular mechanism by which TRH causes TSH-secreting cells of the anterior pituitary to produce TSH is first to bind with TRH receptors in the pituitary cell membrane. This binding in turn activates the phospholipase second messenger system inside the pituitary cells to produce large amounts of phospholipase C, followed by a cascade of other second messengers, including calcium ions and diacyl glycerol, which eventually leads to TSH release.
Effects of Cold and Other Neurogenic Stimuli on TRH and TSH Secretion.
One of the best-known stimuli for increasing TRH secretion by the hypothalamus, and therefore TSH secretion by the anterior pituitary gland, is exposure of an animal to cold. This effect almost certainly results from excitation of the hypothalamic centers for body temperature control. Exposure of rats for several weeks to severe cold increases the output of thyroid hormones sometimes to more than 100 percent of normal and can increase the basal metabolic rate as much as 50 percent. Indeed, persons moving to arctic regions have been known to develop basal metabolic rates that are 15 to 20 percent above normal.
Various emotional reactions can also affect the output of TRH and TSH and therefore indirectly affect the secretion of thyroid hormones. Excitement and anxiety—conditions that greatly stimulate the sympathetic nervous system—cause an acute decrease in secretion of TSH, perhaps because these states increase the metabolic rate and body heat and therefore exert an inverse effect on the heat control center.
Neither these emotional effects nor the effect of cold is observed after the hypophysial stalk has been cut, demonstrating that both of these effects are mediated by way of the hypothalamus.
Feedback Effect of Thyroid Hormone to Decrease Anterior Pituitary Secretion of TSH
Increased thyroid hormone in the body fluids decreases secretion of TSH by the anterior pituitary. When the rate of thyroid hormone secretion rises to about 1.75 times normal, the rate of TSH secretion falls essentially to zero. Almost all this feedback depressant effect occurs even when the anterior pituitary has been separated from the hypothalamus. Therefore, as shown in Figure 77-7, it is probable that increased thyroid hormone inhibits anterior pituitary secretion of TSH mainly by a direct effect on the anterior pituitary gland itself. Regardless of the mechanism of the feedback, its effect is to maintain an almost constant concentration of free thyroid hormones in the circulating body fluids.
 Antithyroid Substances Suppress Thyroid Secretion
Antithyroid Substances Suppress Thyroid Secretion
The best known antithyroid drugs are thiocyanate, propylthiouracil, and high concentrations of inorganic iodides. The mechanism by which each of these drugs blocks thyroid secretion is different from the others and can be explained as follows.
 Thiocyanate Ions Decrease Iodide Trapping.
Thiocyanate Ions Decrease Iodide Trapping.
The same active pump that transports iodide ions into the thyroid cells can also pump thiocyanate ions, perchlorate ions, and nitrate ions. Therefore, the administration of thiocyanate (or one of the other ions as well) in a high enough concentration can cause competitive inhibition of iodide transport into the cell—that is, inhibition of the iodide-trapping mechanism.
The decreased availability of iodide in the glandular cells does not stop the formation of thyroglobulin; it merely prevents the thyroglobulin that is formed from becoming iodinated and therefore from forming thyroid hormones. This deficiency of thyroid hormones in turn leads to increased secretion of TSH by the anterior pituitary gland, which causes overgrowth of the thyroid gland even though the gland still does not form adequate quantities of thyroid hormones. Therefore, the use of thiocyanates and some other ions to block thyroid secretion can lead to the development of a greatly enlarged thyroid gland, which is called a goiter.
 Propylthiouracil Decreases Thyroid Hormone Formation.
Propylthiouracil Decreases Thyroid Hormone Formation.
Propylthiouracil (along with other similar compounds, such as methimazole and carbimazole) prevents formation of thyroid hormone from iodides and tyrosine. The mechanism of this action is partly to block the peroxidase enzyme that is required for iodination of tyrosine and partly to block the coupling of two iodinated tyrosines to form thyroxine or triiodothyronine.
Propylthiouracil, like thiocyanate, does not prevent formation of thyroglobulin. The absence of thyroxine and triiodothyronine in the thyroglobulin can lead to tremendous feedback enhancement of TSH secretion by the anterior pituitary gland, thus promoting growth of the glandular tissue and forming a goiter.
 Iodides in High Concentrations Decrease Thyroid Activity and Thyroid Gland Size.
Iodides in High Concentrations Decrease Thyroid Activity and Thyroid Gland Size.
When iodides are present in the blood in a high concentration (100 times the normal plasma level), most activities of the thyroid gland are decreased, but often they remain decreased for only a few weeks. The effect is to reduce the rate of iodide trapping so that the rate of iodination of tyrosine to form thyroid hormones is also decreased. Even more important, the normal endocytosis of colloid from the follicles by the thyroid glandular cells is paralyzed by the high iodide concentrations. Because this is the first step in release of thyroid hormones from the storage colloid, there is almost immediate shutdown of thyroid hormone secretion into the blood.
Because iodides in high concentrations decrease all phases of thyroid activity, they slightly decrease the size of the thyroid gland and especially decrease its blood supply, in contradistinction to the opposite effects caused by most of the other antithyroid agents. For this reason, iodides are frequently administered to patients for 2 to 3 weeks before surgical removal of the thyroid gland to decrease the necessary amount of surgery, and especially to decrease the amount of bleeding.
 Diseases of the Thyroid
Diseases of the Thyroid
 Hyperthyroidism
Hyperthyroidism
Most effects of hyperthyroidism are obvious from the preceding discussion of the various physiological effects of thyroid hormone. However, some specific effects should be mentioned, especially in connection with the development, diagnosis, and treatment of hyperthyroidism.
 Causes of Hyperthyroidism (Toxic Goiter, Thyrotoxicosis, Graves' Disease).
Causes of Hyperthyroidism (Toxic Goiter, Thyrotoxicosis, Graves' Disease).
In most patients with hyperthyroidism, the thyroid gland is increased to two to three times its normal size, with tremendous hyperplasia and infolding of the follicular cell lining into the follicles, so the number of cells is increased greatly. Also, each cell increases its rate of secretion severalfold; radioactive iodine uptake studies indicate that some of these hyperplastic glands secrete thyroid hormone at rates 5 to 15 times normal.
Graves' disease, the most common form of hyperthyroidism, is an autoimmune disease in which antibodies called thyroid-stimulating immunoglobulins (TSIs) form against the TSH receptor in the thyroid gland. These antibodies bind with the same membrane receptors that bind TSH and induce continual activation of the cAMP system of the cells, with resultant development of hyperthyroidism. The TSI antibodies have a prolonged stimulating effect on the thyroid gland, lasting for as long as 12 hours, in contrast to a little over 1 hour for TSH. The high level of thyroid hormone secretion caused by TSI in turn suppresses anterior pituitary formation of TSH. Therefore, TSH concentrations are less than normal (often essentially zero) rather than enhanced in almost all patients with Graves' disease.
The antibodies that cause hyperthyroidism almost certainly occur as the result of autoimmunity that has developed against thyroid tissue. Presumably, at some time in the person's history, an excess of thyroid cell antigens was released from the thyroid cells, resulting in formation of antibodies against the thyroid gland.
 Thyroid Adenoma.
Thyroid Adenoma.
Hyperthyroidism occasionally results from a localized adenoma (a tumor) that develops in the thyroid tissue and secretes large quantities of thyroid hormone. This presentation is different from the more usual type of hyperthyroidism in that it is usually not associated with evidence of any autoimmune disease. As long as the adenoma continues to secrete large quantities of thyroid hormone, secretory function in the remainder of the thyroid gland is almost totally inhibited because the thyroid hormone from the adenoma depresses the production of TSH by the pituitary gland.
 Symptoms of Hyperthyroidism
Symptoms of Hyperthyroidism
The symptoms of hyperthyroidism are obvious from the preceding discussion of the physiology of the thyroid hormones: (1) a high state of excitability, (2) intolerance to heat, (3) increased sweating, (4) mild to extreme weight loss (sometimes as much as 100 pounds), (5) varying degrees of diarrhea, (6) muscle weakness, (7) nervousness or other psychic disorders, (8) extreme fatigue but inability to sleep, and (9) tremor of the hands.
 Exophthalmos.
Exophthalmos.
Most people with hyperthyroidism exhibit some degree of protrusion of the eyeballs, as shown in Figure 77-8. This condition is called exophthalmos. A major degree of exophthalmos occurs in about one third of patients with hyperthyroidism, with the condition sometimes becoming so severe that the eyeball protrusion stretches the optic nerve enough to damage vision. Much more often, the eyes are damaged because the eyelids do not close completely when the person blinks or is asleep. As a result, the epithelial surfaces of the eyes become dry and irritated and often infected, resulting in ulceration of the cornea.

The cause of the protruding eyes is edematous swelling of the retro-orbital tissues and degenerative changes in the extraocular muscles. In most patients, immunoglobulins that react with the eye muscles can be found in the blood. Furthermore, the concentration of these immunoglobulins is usually highest in patients who have high concentrations of TSIs. Therefore, there is much reason to believe that exophthalmos, like hyperthyroidism itself, is an autoimmune process. The exophthalmos is usually greatly ameliorated with treatment of the hyperthyroidism.
 Diagnostic Tests for Hyperthyroidism.
Diagnostic Tests for Hyperthyroidism.
For the usual case of hyperthyroidism, the most accurate diagnostic test is direct measurement of the concentration of “free” thyroxine (and sometimes triiodothyronine) in the plasma, using appropriate radioimmunoassay procedures.
The following tests also are sometimes used:
1. The basal metabolic rate is usually increased to +30 to +60 in severe hyperthyroidism.
2. The concentration of TSH in the plasma is measured by radioimmunoassay. In the usual type of thyrotoxicosis, anterior pituitary secretion of TSH is so completely suppressed by the large amounts of circulating thyroxine and triiodothyronine that there is almost no plasma TSH.
3. The concentration of TSI is measured by radioimmunoassay. This concentration is usually high in thyrotoxicosis but low in thyroid adenoma.
 Treatment in Hyperthyroidism.
Treatment in Hyperthyroidism.
The most direct treatment for hyperthyroidism is surgical removal of most of the thyroid gland. In general, it is desirable to prepare the patient for surgical removal of the gland before the operation by administering propylthiouracil, usually for several weeks, until the basal metabolic rate of the patient has returned to normal. Then, administration of high concentrations of iodides for 1 to 2 weeks immediately before operation causes the gland to recede in size and its blood supply to diminish. With use of these preoperative procedures, the operative mortality is less than 1 in 1000, whereas before the development of modern procedures, operative mortality was 1 in 25.
 Treatment of the Hyperplastic Thyroid Gland With Radioactive Iodine.
Treatment of the Hyperplastic Thyroid Gland With Radioactive Iodine.
Eighty to 90 percent of an injected dose of iodide is absorbed by the hyperplastic, toxic thyroid gland within 1 day after injection. If this injected iodine is radioactive, it can destroy most of the secretory cells of the thyroid gland. Usually 5 millicuries of radioactive iodine is given to the patient, whose condition is reassessed several weeks later. If the patient is still in a hyperthyroid state, additional doses are administered until normal thyroid status is reached.
 Hypothyroidism
Hypothyroidism
The effects of hypothyroidism, in general, are opposite to those of hyperthyroidism, but a few physiological mechanisms are peculiar to hypothyroidism. Hypothyroidism, like hyperthyroidism, is often initiated by autoimmunity against the thyroid gland (Hashimoto's disease), but in this case the autoimmunity destroys the gland rather than stimulates it. The thyroid glands of most of these patients first demonstrate autoimmune “thyroiditis,” which means thyroid inflammation. Thyroiditis causes progressive deterioration and finally fibrosis of the gland, with resultant diminished or absent secretion of thyroid hormone. Several other types of hypothyroidism also occur that are often associated with development of enlarged thyroid glands, called thyroid goiter, as described in the following sections.
 Endemic Colloid Goiter Caused by Dietary Iodide Deficiency.
Endemic Colloid Goiter Caused by Dietary Iodide Deficiency.
The term “goiter” means a greatly enlarged thyroid gland. As pointed out in the discussion of iodine metabolism, about 50 milligrams of iodine are required each year for the formation of adequate quantities of thyroid hormone. In certain areas of the world, notably in the Swiss Alps, the Andes, and the Great Lakes region of the United States, insufficient iodine is present in the soil for the foodstuffs to contain even this minute quantity. Therefore, in the days before iodized table salt, many people who lived in these areas developed extremely large thyroid glands, called endemic goiters.
The following mechanism results in the development of large endemic goiters: Lack of iodine prevents production of both thyroxine and triiodothyronine. As a result, no hormone is available to inhibit production of TSH by the anterior pituitary, which causes the pituitary to secrete excessively large quantities of TSH. The TSH then stimulates the thyroid cells to secrete tremendous amounts of thyroglobulin colloid into the follicles, and the gland grows larger and larger. However, because of lack of iodine, thyroxine and triiodothyronine production does not occur in the thyroglobulin molecule and therefore does not cause the normal suppression of TSH production by the anterior pituitary. The follicles become tremendous in size, and the thyroid gland may increase to 10 to 20 times its normal size.
 Idiopathic Nontoxic Colloid Goiter.
Idiopathic Nontoxic Colloid Goiter.
Enlarged thyroid glands similar to those of endemic colloid goiter can also occur in people who do not have iodine deficiency. These goitrous glands may secrete normal quantities of thyroid hormones, but more frequently, the secretion of hormone is depressed, as in endemic colloid goiter.
The exact cause of the enlarged thyroid gland in patients with idiopathic colloid goiter is not known, but most of these patients show signs of mild thyroiditis; therefore, it has been suggested that the thyroiditis causes slight hypothyroidism, which then leads to increased TSH secretion and progressive growth of the noninflamed portions of the gland. This theory could explain why these glands are usually nodular, with some portions of the gland growing while other portions are being destroyed by thyroiditis.
In some persons with colloid goiter, the thyroid gland has an abnormality of the enzyme system required for formation of the thyroid hormones. The following abnormalities are often encountered:
1. A deficient iodide-trapping mechanism, in which iodine is not pumped adequately into the thyroid cells
2. A deficient peroxidase system, in which the iodides are not oxidized to the iodine state
3. Deficient coupling of iodinated tyrosines in the thyroglobulin molecule so that the final thyroid hormones cannot be formed
4. Deficiency of the deiodinase enzyme, which prevents recovery of iodine from the iodinated tyrosines that are not coupled to form the thyroid hormones (this is about two thirds of the iodine), thus leading to iodine deficiency
Finally, some foods contain goitrogenic substances that have a propylthiouracil-type of antithyroid activity, thus also leading to TSH-stimulated enlargement of the thyroid gland. Such goitrogenic substances are found especially in some varieties of turnips and cabbages.
 Physiological Characteristics of Hypothyroidism.
Physiological Characteristics of Hypothyroidism.
Whether hypothyroidism is due to thyroiditis, endemic colloid goiter, idiopathic colloid goiter, destruction of the thyroid gland by irradiation, or surgical removal of the thyroid gland, the physiological effects are the same. They include fatigue and extreme somnolence, with persons sleeping up to 12 to 14 hours a day, extreme muscular sluggishness, a slowed heart rate, decreased cardiac output, decreased blood volume, sometimes increased body weight, constipation, mental sluggishness, failure of many trophic functions in the body as evidenced by depressed growth of hair and scaliness of the skin, development of a froglike, husky voice, and, in severe cases, development of an edematous appearance throughout the body called myxedema.
 Myxedema.
Myxedema.
Myxedema develops in persons who have almost total lack of thyroid hormone function. Figure 77-9 shows such a patient, demonstrating bagginess under the eyes and swelling of the face. In this condition, for reasons that are not explained, greatly increased quantities of hyaluronic acid and chondroitin sulfate bound with protein form excessive tissue gel in the interstitial spaces, which causes the total quantity of interstitial fluid to increase. Because of the gel nature of the excess fluid, it is mainly immobile and the edema is the nonpitting type.
 Atherosclerosis in Hypothyroidism.
Atherosclerosis in Hypothyroidism.
As pointed out earlier, lack of thyroid hormone increases the quantity of blood cholesterol because of altered fat and cholesterol metabolism and diminished liver excretion of cholesterol in the bile. The increase in blood cholesterol is usually associated with increased atherosclerosis. Therefore, many hypothyroid patients, particularly those with myxedema, develop atherosclerosis, which in turn results in peripheral vascular disease, deafness, and coronary artery disease with subsequent early death.
 Diagnostic Tests for Hypothyroidism.
Diagnostic Tests for Hypothyroidism.
The tests already described for the diagnosis of hyperthyroidism give opposite results in hypothyroidism. The free thyroxine in the blood is low. The basal metabolic rate in myxedema ranges between −30 and −50. In addition, the secretion of TSH by the anterior pituitary when a test dose of TRH is administered is usually greatly increased (except in the rare instances of hypothyroidism caused by depressed response of the pituitary gland to TRH).
 Treatment of Hypothyroidism.
Treatment of Hypothyroidism.
Figure 77-4 shows the effect of thyroxine on basal metabolic rate, demonstrating that the hormone normally has a duration of action of more than 1 month. Consequently, a steady level of thyroid hormone activity is easily maintained in the body via daily oral ingestion of one or more tablets containing thyroxine. Furthermore, proper treatment of hypothyroidism results in such complete normality that formerly myxedematous patients have lived into their 90s after undergoing treatment for more than 50 years.
 Cretinism
Cretinism
Cretinism is caused by extreme hypothyroidism during fetal life, infancy, or childhood. This condition is characterized especially by failure of body growth and by mental retardation. It results from congenital lack of a thyroid gland (congenital cretinism), from failure of the thyroid gland to produce thyroid hormone because of a genetic defect of the gland, or from a lack of iodine in the diet (endemic cretinism).
A neonate without a thyroid gland may have a normal appearance and function because it was supplied with some (but usually not enough) thyroid hormone by the mother while in utero. A few weeks after birth, however, the neonate's movements become sluggish and both physical and mental growth begin to be greatly retarded. Treatment of the neonate with cretinism at any time with adequate iodine or thyroxine usually causes normal return of physical growth, but unless the cretinism is treated within a few weeks after birth, mental growth remains permanently retarded. This state results from retardation of the growth, branching, and myelination of the neuronal cells of the central nervous system at this critical time in the normal development of the mental powers.
Skeletal growth in a child with cretinism is characteristically more inhibited than is soft tissue growth. As a result of this disproportionate rate of growth, the soft tissues are likely to enlarge excessively, giving the child with cretinism an obese, stocky, and short appearance. Occasionally the tongue becomes so large in relation to the skeletal growth that it obstructs swallowing and breathing, inducing a characteristic guttural breathing that sometimes chokes the child. ![]()