Bone
Bone is a living tissue, which makes up the body skeleton and is one of the hardest structures of the animal body. Bone possesses a certain degree of toughness and elasticity. It provides shape and support for the body. It also provides site of attachment for tendons and muscles, which are essential for locomotion. It protects the vital organs of the body. Bone serves as a storage site for minerals and provides the medium, the marrow for the development and storage of blood cells.
Classification of bones
Bones may be classified according to their shape, mode of development or by their histologic appearance.
Classification based on shape
Bones are classified according to their shape as long, short, flat and irregular bones.
Long bones
These bones are long and slender. They are longer than they are wider. The long bones include the bones of the arm—humerus, radius, ulna; leg—femur, tibia, fibula; fingers and toes—each phalanx (individual bone of finger or toe, all together the phalanges); palms of hands and soles of feet—metacarpals and metatarsals.
The long bones have a tubular diaphysis or the shaft, which is made of compact bone at the periphery, surrounding a central medullary cavity, which contains yellow marrow. The two bulky ends of the long bone are called the epiphysis, which are made of compact bone at the periphery and spongy bone at the center. The articular surface of the epiphysis is covered with hyaline cartilage, which provides a cushioning effect to the opposing bone ends during joint movements and absorbs stress. Epiphyseal line is present between the diaphysis and each epiphysis of an adult long bone. This line is a remnant of the epiphyseal plate.
Short bones
These bones are usually cube shaped of nearly equal length and width. They consist of spongy bone which is covered by a thin layer of compact bone. They contain bone marrow, but no marrow cavity. These bones include bones of wrist—carpals and ankle—tarsals.
Flat bones
These bones are thin, flat, curved, with no marrow cavity. Spongy bone is present between upper and lower layer of compact bone. Flat bones include bones of sternum, ribs, scapula, clavicle, and bones that form roof of the skull, parietal, frontal, temporal and occipital.
Irregular bones
These bones have complex shapes, notched or with ridges and are not included in any of the above categories. These bones are primarily made of spongy bone, which is covered with a thin layer of compact bone, with bone marrow, but no marrow cavity. The irregular bones include bones of vertebrae, facial bones (ethmoid, sphenoid) pelvic bones (ischium and pubis), calcareous (heel bone) and mandible.
Classification based on development
Developmentally, bones are classified as endochondral bones and intramembranous bones.
Classification based on microscopic structure
Histologically, bones are classified as mature bone and immature bone.
Mature bone is further classified as compact bone and cancellous bone.
Compact bone (cortical bone) consists of tightly, packed osteons or haversian systems, forming a solid mass. Since the bone mass is arranged in layers, it is also called lamellar bone (Fig. 9.1).
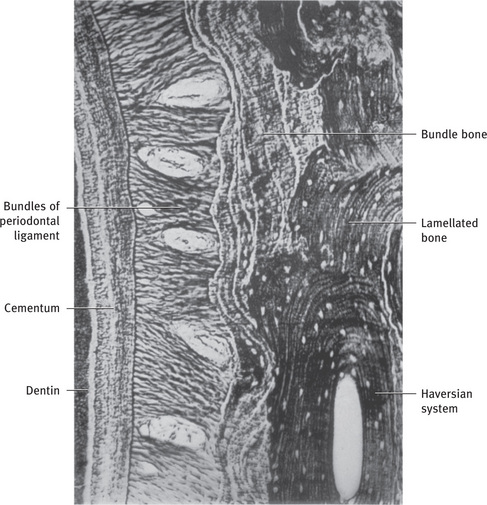
Fig. 9.1 Alveolar bone proper consisting of bundle bone and haversian bone on distal alveolar wall. A reversal line separates the two (silver impregnation).
Cancellous bone (spongy bone) has a honeycomb appearance, with large marrow cavities and sheets of trabeculae of bone in the form of bars and plates (Fig. 9.2).
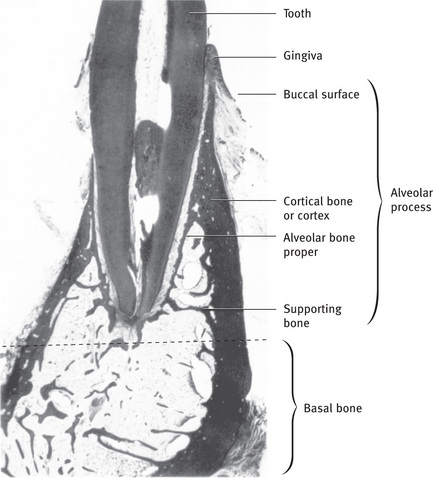
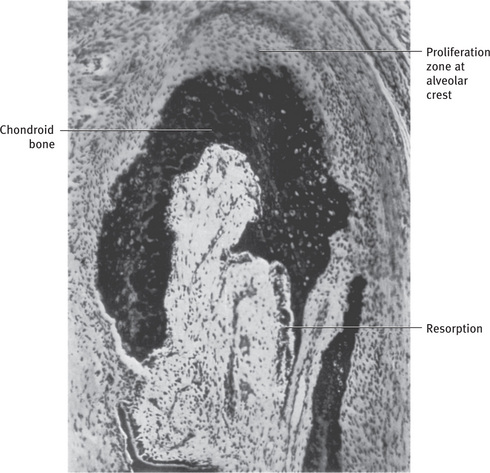
Fig. 9.2 Section through mandible showing relationship of tooth to alveolar process and basal bone. Note the supporting bone is made up of spongy (cancellous) bone From Bhaskar SN: Synopsis of oral histology, St Louis, 1962, The CV Mosby Co.
Woven or immature bone is the first formed bone with irregularly oriented collagen fibers of varying diameters. This type of bone is not usually seen after birth. However it is seen in the alveolar bone and during healing of fractures. Since this bone forms rapidly, it incorporates many osteocytes (Fig. 9.3).
Composition of bone
Bone is a connective tissue composed of cells, fibers and ground substance. The intercellular substance of bone consists of organic and inorganic substances. The inorganic part of bone is made of bone minerals.
The mineral component is composed of hydroxyapatite crystals, with carbonate content and low Ca/P ratio than the pure hydroxyapatite. Small amounts of calcium phosphate are also present. Bone crystals are in the form of thin plates or leaflike structures. They are packed closely with long axis nearly parallel to collagen fibrils axis. The narrow gaps between the crystals contain associated water and organic macromolecules.
The ions present are calcium phosphate, hydroxyl and carbonate. Citrate, magnesium, sodium, potassium, fluoride, iron, zinc, copper, aluminum, lead, strontium, silicon and boron are present in small quantities.
The organic matrix is known as osteoid and is made up of collagen and the noncollagenous proteins.
Collagen is the major organic component in mineralized bone tissues. Type I collagen (> 95%) is the principal collagen in mineralized bone and, together with type V collagen (< 5%), forms heterotypic fiber bundles that provide the basic structural integrity of connective tissues. The elasticity of collagen imparts resiliency to the tissue and helps to resist fractures. Alveolar bone contains type I, type V, type III and type XII collagen. Sharpey's fibers contain type III collagen with type I collagen. Type XII collagen is also present in alveolar bone and has been found to be expressed under conditions of mechanical strain. Types III and XII collagen fibers are produced by fibroblasts during the formation of the periodontal ligament. Types I, V and XII collagens are expressed by osteoblasts. The pink to red color of bone matrix seen in hematoxylin and eosin sections is due to the substantial collagen content.
In woven bone, fibers are interwoven with a great amount of interfibrillar space that is occupied by mineral crystals and associated acidic proteins. In mature bone, collagen fibers form organized sheets. The fibers are oriented perpendicular to each other with little interfibrillar space.
Noncollagenous proteins
Noncollagenous proteins comprise the remaining 10% of the total organic content of bone matrix. Most are endogenous proteins produced by bone cells, while some like albumin are derived from other sources such as blood and become incorporated into bone matrix during osteosynthesis.
Osteocalcin is the first noncollagenous protein to be recognized and represents less than 15% of the noncollagenous bone protein. Osteocalcin is also known as bone Gla protein as it contains the amino acid γ-carboxy glutamic acid. This protein has also been demonstrated in alveolar bone immunohistochemically.
Osteocalcin is a glycoprotein secreted by osteoblasts and is regulated by vitamin D3 and parathyroid hormone. The carboxy terminal segment of osteocalcin acts as a chemoattractant to osteoclast precursors, suggesting a role in bone resorption. It is also believed to be involved in bone calcification as it is a calcium binding protein. It is used as a marker of new bone formation.
Osteopontin and bone sialoprotein were previously, termed as bone sialoproteins I and II respectively. These have been demonstrated in alveolar bone, using immunohistochemistry. Both proteins are heavily glycosylated and phosphorylated, with high levels of acidic amino acids. Glutamic acid is predominant in bone sialoprotein and aspartate is predominant in osteopontin. Despite the structural similarities, these proteins have clearly different functional roles. Bone sialoprotein is restricted to mineralizing tissues, whereas osteopontin has a more generalized distribution. Bone sialoprotein is thought to function in the initiation of mineral crystal formation in vivo. In contrast, osteopontin is a potent inhibitor of hydroxyapatite crystal growth, and is enriched at cell matrix interface, where it can mediate the attachment of bone cells, including osteoclasts. Osteopontin transcription is strongly upregulated by vitamin D3, whereas bone sialoprotein transcription is suppressed by vitamin D3.
The expression of both proteins is stimulated by factors that stimulate bone formation, such as TGF-β family members and glucocorticoids. Osteopontin, vitronectin and fibronectin are termed RGD-containing proteins, as they have a specific amino acid sequence (Arg-Gly-Asp), which has important implications in the attachment of cells to the bone matrix.
Osteonectin comprises about 25% of noncollagenous proteins. It is bound to collagen and hydroxyapatite crystals. It is a secreted calcium binding glycoprotein, that interacts with extracellular matrix molecules. It has been proposed that, it may play a role in the regulation of cell adhesion, proliferation and modulation of cytokine activity, and in initiating hydroxyapatite crystal formation.
Proteoglycans are also present in the bone matrix. A large chondroitin sulfate proteoglycan, has been extracted from the nonmineralized bone matrix, while two small proteoglycans, biglycan and decorin (chondroitin sulfate proteoglycan I and II respectively) have been found in EDTA extracts of bone. Decorin and biglycan comprise < 10% of the noncollagenous proteins in bone, but this decreases with maturation of bone. A third small proteoglycan (chondroitin sulfate proteoglycan) has been found entirely associated with mineral crystals.
Biglycan is more prominent in developing bone and has been mineralized to pericellular areas. The precise function of biglycan is unknown, but similar to decorin, it can bind TGF-β and extracellular matrix macromolecules, including collagen, and thereby regulate fibrillogenesis. Decorin, as the name suggests, binds mainly within the gap region of collagen fibrils and decorates the fibril surface. The primary calcification in bones is reported to follow removal of decorin and fusion of collagen fibrils.
Lysyl oxidase and tyrosine rich acidic matrix proteins (TRAMP) are components of demineralized bone and dentin matrix. Lysyl oxidase is a critical enzyme for collagen crosslinking. TRAMP, also known as dermatopontin, binds decorin and TGF-β, and together these proteins regulate the cellular response to TGF-β.
Procollagen peptides, thrombospondin, fibronectin, vitronectin and alkaline phosphatase are the other proteins found in bone.
Matrix Gla protein and α2Hs glycoproteins are not secreted by osteoblasts but are of particular interest, with respect to regulation of mineralization. Matrix Gla protein is a mineral binding extracellular matrix protein secreted by vascular smooth muscle cells and chondrocytes that prevent mineralization in vascular tissues and cartilage. The absence of α2HS glycoprotein, which is produced by the liver, compromises the inhibition of apatite formation by serum.
Bone matrix also contains proteases, protease inhibitors and a variety of cytokines secreted by osteoblasts, that reg - ulate cell metabolism. These cells secrete several members of bone morphogenetic proteins (BMP) superfamily, including BMP-2, BMP-7, TGF-β, insulin like growth factors (IGF-I and IGF-II), platelet derived growth factor (PDGF) and fibroblast growth factor (FGF). IGF-I, PDGF and TGF-β increase the rapidity of bone formation and bone repair.
Bone histology
Osteoid is an unmineralized bone matrix on the surface, where active bone formation is taking place. It is approximately 5–10 μm before the commencement of mineralization. The mineralizing front is linear. Osteoid contains type I collagen fibers, parallel to the bone surface, embedded in ground substance of proteoglycans, glycoproteins and other proteins.
All mature bones have a dense outer sheet of compact bone and a central medullary cavity. The cavity is filled with red or yellow bone marrow in living bone. This cavity shows a network of bone trabeculae. Trabecular, spongy or cancellous bone are the terms used to describe this network.
The outer aspect of compact bone is surrounded by a condensed fibrocollagen layer, the periosteum which has two layers: an outer layer which is a dense, irregular connective tissue termed fibrous layer; and an inner osteogenic layer, next to the bone surface consisting of bone cells, their precursors and a rich vascular supply. The periosteum is active during fetal development. It is also important in the repair of fractures.
The inner surface of compact and cancellous bone are covered by a thin cellular layer called endosteum. In resting adult bone, quiescent osteoblasts and osteoprogenitor cells are present on the endosteal surfaces. These cells act as reservoir of new bone forming cells for remodeling or repair.
At the periosteal and endosteal surfaces, the lamellae are arranged in parallel layers surrounding the bony surface and are called circumferential lamellae.
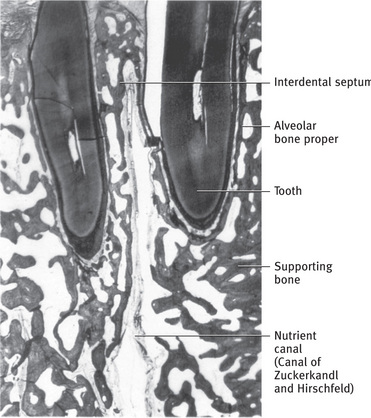
Deep to the circumferential lamellae, the lamellae are arranged as small concentric layers around a central vascular canal. Haversian (vascular) canal (about 50 μ in diameter) and the concentric lamellae together is known as the osteon or haversian system (Fig. 9.4). There may be up to 20 concentric lamellae within each osteon. Osteon is the basic metabolic unit of bone. A cement line of mineralized matrix delineates the haversian system. This cement line contains little or no collagen, and is strongly basophilic, because it has a high content of glycoproteins and proteoglycans. It marks the limit of bone erosion prior to the formation of osteon, and is therefore also known as reversal line (Fig. 9.5). This line appears to be highly irregular as it is formed by the scalloped outline of the Howship's lacunae. This line has to be distinguished from the more regular appearance of the resting line, which denotes the period of rest during the formation of bone (Fig. 9.4). The collagen fibers within each lamella spiral along the length of lamella, but have different orientations to those in adjacent lamella. This pattern is to withstand torsion stresses.
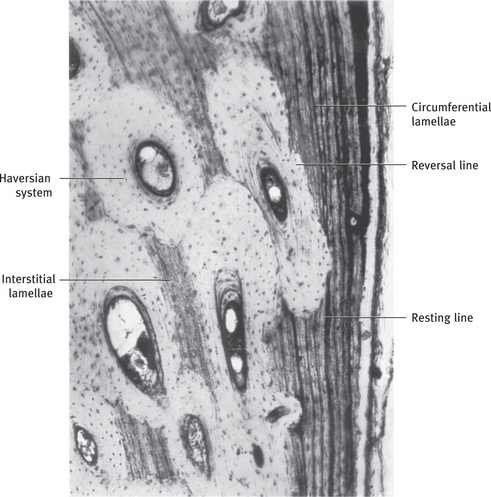
Fig. 9.4 Appositional growth of mandible by formation of circumferential lamellae. These are replaced by haversian bone; remnants of circumferential lamellae in the depth persisting as interstitial lamellae.
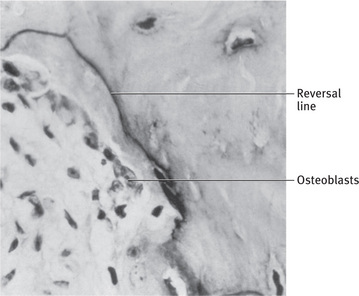
Fig. 9.5 Reversal line in bone From Bhaskar SN: Synopsis of oral histology, St Louis, 1962, The CV Mosby Co.
Adjacent haversian canals are interconnected by Volkmann's canals, channels that contain blood vessels, creating a rich vascular network, throughout the compact bone. Osteocytes are present in lacunae, at the junctions of the lamellae. Small canaliculi radiate from lacunae to haversian canal to provide a passage way through the hard matrix. The canaliculi connect all the osteocytes in an osteon together. This connecting system permits nutrients and wastes to be relayed from one osteocyte to the other. The adult bones, between the osteons, contain interstitial lamellae, which are remnants of osteons, left behind during remodeling.
Spongy bone
Spongy bone and compact bone have the same cells and intercellular matrix, but differ in the arrangement of components. Spongy bone looks like a poorly organized tissue in contrast to compact bone.
The bony substance consists of large slender spicules called trabeculae. The trabeculae are up to 50 μm thick. The trabeculae are oriented along lines of stress to withstand the forces applied to bone (Fig. 9.2). The marrow spaces are large. The trabeculae surround the marrow spaces from where they derive their nutrition through diffusion.
Hemopoietic tissue in bones
Red marrow is present in young bone and yellow marrow in old bone. Red marrow is found within cavities of spongy bone of long bones and diploe of flat bones. In newborn infants, the medullary cavity and all areas of spongy bone contain red bone marrow. Red marrow contains stem cells of both the fibroblast/mesenchymal type and blood cell lineage. Yellow marrow is seen in epiphysis of long bones. In old bone, the marrow is yellow, with loss of hemopoietic potential and increased accumulation of fat cells.
Yellow marrow of the medullary cavity can revert to red marrow, if a person is anemic and needs increased red blood cell production.
Bone cells
Osteoblasts
Osteoblasts are mononucleated cells responsible for the synthesis and secretion of the macromolecular organic constituents of bone matrix. These cells are derived from osteoprogenitor cells of mesenchymal origin, which are present in the bone marrow and other connective tissues. Periosteum also serves as an important reservoir of osteoblasts, particularly during childhood growth, after skeletal fractures or with bone forming tumors.
Morphology
Osteoblasts are basophilic, plump cuboidal or slightly flattened cells. These cells are found on the forming surfaces of growing or remodeling bone. These cells produce the organic matrix of bone (Osteoid) which primarily consists of type I collagen and the balance being noncollagenous proteins (Fig. 9.6).
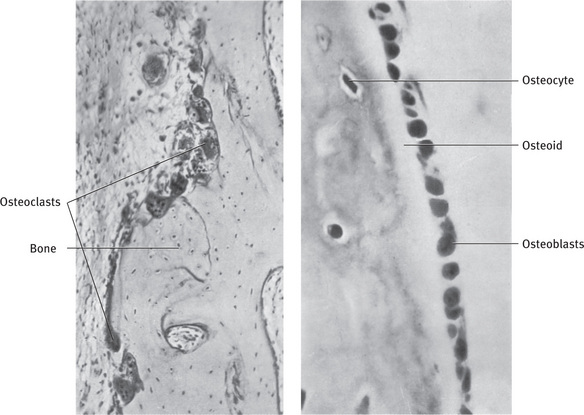
Fig. 9.6 Resorption and apposition of bone. Left, Osteoclasts in Howship's lacunae. Right, Osteoblasts along bone trabecula. Layer of osteoid tissue is a sign of bone formation.
Osteoblasts exhibit abundant and well developed protein synthetic organelles. The intense cytoplasmic basophilia is due to an abundance of rough endoplasmic reticulum. The procollagen and other organic constituents of bone matrix synthesized by this organelle, enter its lumen and are carried by transfer vesicles to the Golgi complex and assembled within Golgi complex in secretory granules. A pale juxtanuclear area indicates the site of the Golgi complex. These granules release their contents along the surface of cell opposed to forming bone which, assemble extracellularly as fibrils to form osteoid. The presence of the noncollagenous proteins within the secretory collagen granule or in a distinct population of granules is debatable. But, the noncollagenous proteins are also released along the surface of osteoblasts apposed to osteoid and diffuse from osteoblast surface, towards the mineralization front, where they participate in regulating mineral deposition. Nucleus is situated eccentrically in the part of the cell that is farthest away from the adjacent bone surface.
Organic matrix is deposited around the cell bodies and their cytoplasmic processes resulting in the formation of canaliculi. The cytoplasmic processes are not seen in H and E sections, but in other preparations, it is known that, they are in contact with one another and also with processes of osteocytes in lacunae beneath them. The cells contact one another by adherens and gap junctions. These are functionally connected to microfilaments and enzymes associated with intracellular secondary messenger systems. This arrangement provides for intercellular adhesion and cell to cell communication and ensures that osteoblast layer completely covers the osteoid surface and functions in a coordinated manner.
Osteoblasts also contain prominent bundles of actin, myosin and cytoskeletal proteins which are associated with maintenance of cell shape, attachment and motility.
Formation
Osteoblasts are derived from undifferentiated pluripotent mesenchymal stem cells. Osteoprogenitor cells are divided into two types, determined (DOPCs) and inducible osteogenic precursor cells (IOPCs). These DOPCs are present in the bone marrow, endosteum and periosteum and differentiate into osteoblasts under the influence of systemic and bone derived growth factors. The IOPCs represent mesenchymal cells present in other organs and tissues that may differentiate into bone forming cells when stimulated.
The osteoprogenitor cells express transcription factors cbfa1/Runx-2 and osterix which are essential for osteoblast differentiation. cbfa1 is a member of runt related family of transcription factors. It triggers the expression of BSP, osteopontin, osteocalcin and type I collagen. Osterix, a zinc fingercontaining transcription factor, is similar to Runx-2.
Functions
The main function of osteoblast is the formation of new bone via synthesis of various proteins and polysaccharides. Other functions include the regulation of bone remodeling and mineral metabolism. Osteoblasts also play a significant role in the mineralization of osteoid. Osteoblasts secrete type I collagen which is widely distributed and not unique to osteoblasts whereas, osteocalcin and cbfa-1 (osteoblast specific transcription factor) are specific to cells of osteoblast lineage. These provide useful markers of osteoblast phenotype. Osteoblasts also secrete small amounts of type V collagen, osteonectin, osteopontin, RANKL, osteoprotegerin, proteoglycans, latent proteases and growth factors including bone morphogenetic proteins. Osteoblasts exhibit high levels of alkaline phosphatase on outer surface of plasma membrane which is used as a cytochemical marker to distinguish preosteoblasts from fibroblasts. Total alkaline phosphatase activity has been recognized as a reliable indicator of osteoblast function. Osteoblasts express receptors for various hormones including PTH, vitamin D3, estrogen and glucocorticoids, which are involved in the regulation of osteoblast differentiation. The osteoblasts recognize the resorptive signal and transmit it to the osteoclast.
RANKL is a membrane bound TNF related factor, that is expressed by osteoblasts/stromal cells. The presence of RANKL is vital in osteoclast differentiation.
Cbfa1 expressed by osteoprogenitor cells, regulates the expression of OPG (osteoprotegerin) which is a potent inhibitor of osteoclast formation and function.
Regulation of osteoblast activity
The overall integrity of bone is controlled by hormones, proteins secreted by hematopoietic bone marrow cells and bone cells.
Role of parathormone (PTH)
In response to hypocalcemia, the hormone activates a mechanism for the release of calcium from bone, the principal body store house. PTH does so, by an indirect effect mediated by PTH receptors on bone stromal cells including osteoblasts, as osteoclasts are devoid of PTH receptors. PTH regulates serum calcium levels by stimulation of bone resorption and can also have anabolic effects in vivo that appear to be mediated through TGF-β and IGF-I. These opposing effects of PTH are consistent with the apparent coupling of bone formation and remodeling.
Cells of mesenchymal lineage that differentiate into osteoblasts were shown to produce PTHrP very early during differentiation, and PTHrP levels decreased with further cell maturation. PTHR1 is expressed at a later stage in differentiation, by committed preosteoblasts. PTHrP (parathyroid hormone related protein) resembles PTH in its amino terminal sequence and the two have similar structural requirements for binding and activation of their common receptor, type 1 PTH receptor (PTHR1). Persistently increased levels of local PTHrP favor increased osteoclast formation through stimulation of RANKL production. Hence, PTHrP needs to be regulated in terms of concentration, location and time, so that it is presented briefly to target cells. PTH is secreted as a hormone in response to a hypocalcemic signal in order to regulate calcium homeostasis by promoting bone resorption, whereas PTHrP functions as a bone cytokine to control bone mass.
While vitamin D3 stimulates bone resorption, it is also essential for normal bone growth and mineralization. It also promotes calcium absorption from the intestine. It stimulates synthesis of osteocalcin and osteopontin by osteoblasts and suppresses collagen production. The action of parathormone and vitamin D3 is that, they enhance bone resorption at high concentrations (pharmacological) and support bone formation at low (physiologic) concentrations.
Growth hormone is required for attaining normal bone mass which is mediated by the local production of IGF-I. It binds to membrane bound growth hormone receptors on activated osteoblasts.
Insulin targets osteoblasts directly and stimulates bone matrix formation and mineralization and indirectly affects bone formation through stimulation of IGF-I (insulin like growth factor) produced in the liver.
Bone morphogenetic proteins
These are the only factors that can initiate osteoblastogenesis from uncommitted progenitor cells. These proteins are expressed during embryonic development as well as in adulthood. BMPs 2, 4 and 6 direct the pluripotent cells to commit to an osteoblastic pathway. BMPs can also increase the differentiation of committed cells to the osteoblast lineage. BMPs can also upregulate cbfa1 under certain conditions during osteoblast differentiation. cbfa1, in turn, activates osteoblast-specific genes such as osteopontin, bone sialoprotein, type I collagen and osteocalcin.
TGF-β During the early stages of bone formation, the action of transforming growth factor-β is to recruit and stimulate osteoprogenitor cells to proliferate, providing a pool of early osteoblasts. In contrast, during later stages of osteoblast differentiation, TGF-β blocks differentiation and mineralization. These effects appear to be highly dependant on bone cell source, dose applied and local environment, which may be a result of inhibition of DNA synthesis at high TGF-β concentration. Additionally, it inhibits the expression of Runx-2 and osteocalcin genes, whose expression is controlled by cbfa1/ Runx-2 in osteoblast like cell lines.
IGF I and II (insulin like growth factors) increase proliferation and play a major role in stimulating mature osteoblast function. IGF-1 upregulates osterix but not cbfa1. IGF-1 along with BMP-2 acts synergistically on osterix expression.
FGF (fibroblast growth factors) are a family of structurally related polypeptides that play a critical role in angiogenesis and mesenchymal cell mitogenesis. In normal adult tissues, the most abundant proteins are FGF-1 and FGF-2. FGF-2 is expressed by osteoblasts and is more potent than FGF-1. These factors exert their effect on bone formation, primarily through increased proliferation of osteoprogenitor cells and promotion of osteogenic differentiation.
Glucocorticoids promote differentiation of osteoblasts and stimulate bone matrix formation in vitro. But, prolonged treatment with glucocorticoids in vivo, results in bone loss, which can be attributed to increased PTH production in response to the inhibitory effects of glucocorticoids on calcium absorption and depletion of osteogenic precursor cells.
PDGF (platelet derived growth factor) The isoforms of this factor have a strong chemotactic effect on osteoblasts and other connective tissue cells and may act to recruit mesenchymal cells, during bone development and remodeling. It has similar effects to fibroblast growth factor in promoting osteogenesis. It acts as a potent mitogen for all cells of mesenchymal origin. PDGF may also have direct and indirect effects on bone resorption by the upregulation of collagenase transcription and an increase in IL-6 expression in osteoblasts.
Vascular endothelial growth factor (VEGF) acts directly on osteoblasts to promote osteoblast migration, proliferation and differentiation in an autocrine manner. It is also a mediator of osteoinductive factors like TGF-β, IGF-1, FGF-2, which upregulate VEGF expression in osteoblasts. VEGF also influences osteoblasts indirectly via its effects on endothelial cells. It stimulates the production of bone forming factors for osteoblasts by endothelial cells.
The Wnt/β-catenin signaling pathway has been shown to have important roles in the maintenance of self-renewal of stem cells in epidermal and hematopoietic cells, and regulation of bone formation. This pathway is proposed to directly promote osteogenesis through actions on Runx-2 gene.
Bone lining cells
Once osteoblasts have completed their function, they are either entrapped in the bone matrix and become osteocytes or remain on the surface as lining cells. Osteoblasts flatten, when bone is not forming and extend along the bone surface and hence the name. These cells contain very few organelles but, retain gap junctions with osteocytes, while retaining its vitality.
But 50–70% of osteoblasts present at the remodeling site cannot be accounted for after enumeration of lining cells and osteocytes. It has been proposed that missing osteoblasts die by apoptosis. Growth factors and cytokines produced in bone microenvironment influence this process. Tumor necrosis factor (TNF) promotes apoptosis. TGF-β and IL-6 have antiapoptotic effects. Glucocorticoids and estrogen withdrawal promote apoptosis in osteoblasts and osteocytes.
Osteocytes
Osteoblasts produce the extracellular matrix, osteoid. As the osteoblasts form the bone matrix, they get entrapped within the matrix they secrete, and are called osteocytes. The number of osteoblasts that become osteocytes, depends on the rapidity of bone formation. Embryonic (woven) bone and repair bone, show more osteocytes, than lamellar bone, as they are formed rapidly. There are approximately ten times more osteocytes than osteoblasts in an individual bone. The average half life of human osteocytes is approximately 25 years. The life span of osteocytes exceeds that of active osteoblasts, which is estimated to be only three months in human bones.
During the preparation of ground sections, the osteocytes are lost, but the spaces are filled with debris and appear black, when viewed under the microscope, using transmitted light.
Within the bone matrix, the osteocyte reduces in size, creating a space around it, called the osteocytic lacuna. Under the electron microscope, it has been observed that, a thin layer of uncalcified tissue, lines the lacuna. The lacunae can appear ovoid or flattened. Narrow extensions of these lacunae form channels called canaliculi. Osteocytic processes are present within these canaliculi. Canaliculi do not usually extend through and beyond the reversal line surrounding an osteon, and so do not communicate with neighboring systems. These processes contain bundles of microfilaments and some smooth endoplasmic reticulum. At the distal end, these processes contact the processes of adjacent cells, i.e. other osteocytes through gap junctions. They also maintain contact with osteoblasts and bone lining cells on the surface.
The canaliculi penetrate the bone matrix and permit diffusion of nutrients, gases and waste products between osteocytes and blood vessels. Osteocytes also sense the changes in environment and send signals that affect response of other cells involved in bone remodeling. This interconnecting system maintains the bone integrity and bone vitality. Failure of the interconnecting system between osteocytes and osteoblasts leads to sclerosis and death of bone.
Mature inactive osteocytes possess an ellipsoid cell body with long axis parallel to the surrounding bony lamellae. The nucleus is oval with a narrow rim of faintly basophilic cytoplasm. The cell has very few organelles, but contains sufficient rough endoplasmic reticulum and large Golgi region, to suggest that these cells are capable of keeping the bone matrix in a state of good repair. Osteocytes secrete a few matrix proteins. Lysosomes have also been described in the older osteocytes.
Old osteocytes retract their processes from the canaliculi, and when dead, their lacunae and canaliculi may get plugged with debris. The death of the osteocytes leads to resorption of the matrix by osteoclasts.
Transformation of osteoblasts into osteocytes
At the end of bone forming phase, osteoblasts can have one of four different fates
(a) Become embedded in the bone as osteocytes
(b) Transform into inactive osteoblasts and become bone lining cells
(d) Transdifferentiate into cells that deposit chondroid or chondroid bone.
The transformation process is proposed to involve three cells, preosteoblasts which differentiate into osteoblasts, and these osteoblasts which become trapped as osteocytes.
Preosteoblasts are less cuboidal in shape and are located at a distance from the bone surface, do not deposit bone matrix, but can still divide. These cells produce type I collagen precursor molecules, which later assemble into collagen fibrils after post transitional modification.
Preosteoblasts differentiate into active bone matrix secreting osteoblasts, which are cuboidal in shape, and ultimately deposit the bone matrix.
As the bone matrix deposition continues, osteoblasts become embedded in the secretory product, the osteoid. Cells at this early stage of osteoblast to osteocyte differentiation are called large osteocytes. These cells are large with a well developed Golgi apparatus for collagen storage.
On mineralization of the osteoid, there is a reduction in the endoplasmic reticulum and Golgi apparatus in the osteocyte, suggesting a decrease in protein synthesis.
Four schemes have been proposed to explain how an osteoblast could get trapped within bone matrix.
- Osteoblasts are unpolarized and lay down bone in all directions, i.e. the cells become trapped in their own secretions.
- Individual osteoblasts are polarized, but those within same generation are polarized differently to those in adjacent layers. As a result, bone is deposited in all directions and osteoblasts become trapped.
- Osteoblasts of each generation are polarized in the same direction. One generation buries the preceding one in bone matrix.
- Within one generation, some osteoblasts slow down rate of bone deposition or stop laying down bone, so that they become trapped by the secretion of their neighboring cells.
It has also been proposed that osteoblasts are highly polarized and function as a unit to lay down bone synchronously. All cells move away from osteogenic front as bone matrix is deposited, ultimately resulting in acellular bone.
Decision to transform into osteocyte
During bone formation, processes on the vascular surface of the osteocytes continue to grow to enable osteocytes to remain in contact with active osteoblast layer and to modulate their activity. When these vascular facing processes stop growing, they produce a signal that induces the recruitment of these osteoblasts with which they are losing contact. The committed osteoblasts are then transformed into osteoblastic osteocytes. The signal to stop growing a vascular process, may be issued by the osteoblasts, with which they have contact or it may be due to the gradual reduction in the vascular supply to the osteocyte, as new layers of bone are laid down on the osteogenic front.
Several factors have been reported to modulate osteoblast function and are involved in controlling the decision to transform into an osteocyte.
- The transcription factors Runx-2 and osterix are important for osteoblast differentiation.
- Leptin is a gene product synthesized by adipocytes but may serve an important signal to modulate osteoblast function. It has also been shown that leptin protects cells from apoptosis and facilitates transformation from osteoblast to pre-osteocyte.
- TGF-β related signaling mechanism can increase the propensity of osteoblast to mature into an osteocyte and decrease the duration of its productive functioning by shortening its life span.
Two different mechanisms of transformation have been proposed:
- Stationary osteoblasts transform into osteocytes by selfburial
- Dynamic osteoblasts are selected to transform into osteocytes by secretory activity of neighboring osteoblasts.
But, there is not only one mechanism for transforming osteoblasts into osteocytes, since different mechanisms exist in different bones, different types of bone formation, different positions within a bone, and different vertebrate species.
Osteoclasts
The word “osteoclast” is derived from the Greek words for “bone and broken”. Osteoclast is a type of bone cell that removes bone tissue by removing the mineralized matrix of bone.
Morphology
Osteoclasts lie in resorption bays called Howship's lacunae. Osteoclast is a large cell approximately 40–100 μm in diameter with 15 to 20 closely packed nuclei. Osteoclasts with many nuclei resorb more bone than osteoclasts with few nuclei (Fig. 9.6). The different nuclei are proposed to be of different ages and there is evidence of apoptosis. These cells are variable in shape due to their motility. The cytoplasm of the osteoclast shows acid phosphatase containing vesicles and vacuoles. The presence of acid phosphatase distinguishes the osteoclast from other multinucleated giant cells. Mitochondria are extensive and distributed throughout the cytoplasm, except below the ruffled border. Rough endoplasmic reticulum is relatively sparse for the size of the cell. Golgi complex is extensive and arranged in stacks. The cytoplasm also contains microtubules, which transport vesicles between Golgi stacks and ruffled membrane. Cathepsin containing vesicles and vacuoles are present close to the ruffled border indicating resorptive activity of these cells.
Formation of osteoclast
Multinucleated giant cells are derived from hemopoietic cells of monocyte macrophage lineage. The earliest identifiable hematopoietic precursor that can form osteoclast is the granulocytemacrophage colony forming unit (CFU-GM). The early precursor cells proliferate and differentiate to form post mitotic committed precursor cells. These committed precursors then differentiate and fuse to form immature multinucleated giant cells. These are activated to form bone resorbing osteoclasts. The differentiation into osteoclasts is through a mechanism involving cell–cell interaction with osteoblast stromal cells.
The formation of osteoclast requires the presence of RANK ligand (receptor activator of nuclear factor κB) and M-CSF (macrophage colony stimulating factor). These two membrane bound proteins are produced by neighboring stromal cells and osteoblasts, thus requiring direct contact between these cells and osteoclast precursors.
M-CSF acts through its receptor on osteoclast precursors c-Fms (colony stimulating factor 1 receptor) and thereby provides signals required for proliferation. M-CSF also enhances osteoclast activity by preventing osteoclast apoptosis.
RANKL belongs to the TNF (tumor necrosis factor) family of ligands. RANKL is also called osteoclast differentiation factor (ODF), TNF related induced cytokine (TRANCE), or osteoprotegerin ligand (OPGL).
RANK is homotrimeric TNF receptor family member. As RANKL binds to RANK on surface of M-CSF triggered osteoclast precursors, a number of signaling pathways are activated committing the cell to osteoclast lineage. These are initially mediated by TRAF 6 TNF (receptor-associated factor), leading to activation of NF-κβ, the (AP)-1 transcription factor complex and ultimately NFATc1 nuclear factor of activated T cells.
RANKL has been implicated in the fusion of osteoclast precursors into multinucleated giant cells, their differentiation into mature osteoclasts, their attachment to bone surface and their activation to resorb bone.
Osteoprotegerin (OPG) is a member of the TNF receptor family and is expressed by osteoblasts. It recognizes RANKL, and blocks the interaction between RANK and RANKL, leading to an inhibition of osteoclast differentiation and activation. Cbfa1 contributes to the expression of OPG. The balance between RANKL-RANK signaling and the levels of biologically active OPG, regulates development and activation of osteoclasts and bone metabolism.
Regulation of osteoclast activity
Estrogen suppresses the production of bone resorbing cytokines including IL-1 and IL-6. Estrogen deficiency results in marked bone resorption by increasing osteoclast activity. It is suggested that estrogen prevents excessive bone loss before and after menopause by decreasing the life span of osteoclast, by promoting apoptosis. Estrogen increases production of TGF-β by osteoblasts which stimulate apoptosis of osteoclasts.
Vitamin D3 and parathyroid hormone (PTH) Vitamin D3 promotes the differentiation of osteoclasts from monocyte macrophage stem cell precursors in vitro. In vivo, enhanced osteoclastic bone resorption has been observed when vitamin D is applied at high dose. This effect is caused by the stimulation of RANKL production by osteoclasts. PTH is secreted in response to changes in blood calcium and affects bone formation and resorption. PTH binds to osteoblasts and induces the production of M-CSF and RANKL that stimulate the maturation and action of osteoclasts.
Calcitonin is a particularly potent inhibitor of osteoclast activity, but its effects are transient likely, due to downregulation of calcitonin receptor on osteoclast in the sustained presence of hormone. Calcitonin inhibits proliferation and differentiation of osteoclast precursors. It reduces the dimension of ruffled border and their dissociation into monocytic cells. Calcitonin also promotes osteoclast apoptosis.
Several factors expressed by osteoblasts/lymphocytes have an impact on osteoclastogenesis to enhance (IL-1, IL-6, IL-8 and IL-11) or limit (IL-4, IL-10, IL-12, IL-13, IL-18) osteoclast formation.
IL-6 is produced by stromal or osteoblast lineage cells in response to PTH and vitamin D3 and on stimulation by IL-1, TNFα, TGFβ, PDGF and IGF-2. IL-6 alone or in concert with other agents stimulates osteoclastogenesis. But it is a much less potent stimulator of osteoclast generation than IL-1 and TNF-α.
Osteoclast precursors interact with bone matrix to trigger their own differentiation by producing IL-1. IL-1 can also induce osteoclast formation by increasing expression of RANKL on surface of osteoblasts and marrow stromal cells.
TNFα stimulates differentiation of osteoclast progenitors into osteoclasts in the presence of M-CSF independent of RANKL-RANK interaction.
OCIL (osteoclast inhibitory lectin) has been recognized as an inhibitor of osteoclast formation. OCIL and its related proteins OCILrP1 and OCILrP2 are type II membrane bound C-lectins expressed by osteoblasts.
TGF-β and interferon-γ inhibit proliferation and differentiation of committed precursors into mature osteoclasts. TGF-β also promotes apoptosis of osteoclasts. But, TGF-β is also believed to enhance osteoclast differentiation in hematopoietic cells stimulated with RANKL and M-CSF.
IFN-γ interferes with the osteoclast differentiation induced by RANKL and this mechanism is critical for the suppression of pathological bone resorption associated with inflammation.
Bisphosphonates suppress bone resorption via injury to osteoclasts when they solubilize bisphosphonate contaminated bone. These do not suppress osteoclastogenesis. They are capable of inducing osteoclast apoptosis. Bisphosphonates suppress bone resorption without consistent reduction in osteoclast numbers which suggests that these compounds might act through a mechanism that is distinct from OPG. These are used as antibone resorbing agents in various diseases associated with stimulated bone resorption.
PGE2 (Prostaglandins of E series) can act as powerful mediators of bone resorption and can also influence bone formation. The prostaglandins, induce osteoclast formation through increased expression of RANKL on the surface of immature osteoblasts and stromal cells. PGE2 can also stimulate bone formation when administered systemically.
Bone formation
Intramembranous ossification
Intramembranous ossification is the direct formation of bone within highly vascular sheets of condensed primitive mesenchyme. This process occurs in the flat bones of the skull and clavicles. It begins approximately towards the end of second month of gestation. The sequence of events is as follows.
Formation of bone matrix within the fibrous membrane
At the site where a bone will develop, there is initially loose mesenchyme, which appears as widely separated, pale staining, stellate cells with interconnecting cytoplasmic processes.
Then a center of osteogenesis develops in association with capillaries that grow into the mesenchyme. These mesenchymal cells in the center become round and basophilic with thick interconnecting cytoplasmic processes. These cells differentiate into osteoblasts and secrete the organic bone matrix. Once surrounded by bone matrix, these cells are called osteocytes. The matrix soon begins to calcify. The earliest crystals appear in association with extracellular matrix vesicles produced by osteoblasts. Crystal formation subsequently extends into collagen fibrils in the surrounding matrix. Even after calcification begins, the osteocytes obtain nutrients and oxygen by diffusion along bone canaliculi.
Formation of woven bone
The first small mass of newly formed bone matrix is an irregular shaped spicule. The bony spicules gradually lengthen into longer anastomosing structures called trabeculae. The trabeculae extend in a radial pattern leading to an anastomosing network of trabeculae characteristic of spongy bone. The spicules and trabeculae are easily recognized in Hematoxylin and Eosin sections, because the matrix stains a bright pink color, and they are also covered with large rounded osteoblasts that have intensely basophilic cytoplasm. These trabeculae enclose local blood vessels. This early membrane bone is termed woven bone.
At this stage, few mesenchymal cells remain undifferentiated. But, before these cells disappear, they leave a layer of flat cells called osteogenic cells on trabeculae which do not have osteoblasts. In richly vascular areas, these osteogenic cells give rise to osteoblasts that form bone matrix. In areas, with no capillary blood supply, they form chondroblasts which lay down cartilage.
Appositional growth mechanism and formation of compact bone plates
The new layers of bone matrix are deposited on preexisting bone surfaces. The osteogenic cells on the surface of spicules and trabeculae are always in a superficial position repeating the process again and again. This is appositional growth, which results in build up of bone tissue one layer at a time. Every generation of osteoblasts produce their own canaliculi. Hence, all the new osteocytes remain linked through canaliculi to bone surface above and to osteocytes below. As the trabeculae increase in width due to appositional growth, neighboring capillaries are incorporated to provide nutrition to osteocytes in deeper layers. New bone is deposited on some surfaces and resorbed at other sites leading to remodeling of trabeculae. This remodeling maintains shape and size of bone throughout life.
Continued appositional growth and remodeling of trabeculae converts cancellous bone to compact bone. Cancellous bone is in the central part of bones as the trabeculae do not increase in size. The vascular tissue in cancellous bone differentiates into red marrow.
Formation of osteon
As layers of bone tissue build up by apposition, the trabeculae thicken and the soft tissue spaces get narrowed. This process converts cancellous bone to compact bone. As cancellous bone gets converted to compact bone, a number of narrow canals are formed lined by osteogenic cells. These canals enclose vessels that were present in soft tissue spaces of cancellous network. The consecutive lamellae of bone become added to the bony walls of spaces in cancellous bone, which is called osteon or haversian system. These osteons are called primitive osteons as they are short, compared to those in long bones.
While these changes occur, external to the woven bone, there is condensation of vascular mesenchyme called the periosteum.
The mechanism of intramembranous ossification involves bone morphogenetic proteins (BMPs) and activation of transcription factor called cbfa1. BMPs activate cbfa1 gene in mesenchymal cells. The cbfa1 transcription factor transforms mesenchymal cells into osteoblasts. It is believed that the proteins activate genes for osteocalcin, osteopontin and other bone specific extracellular matrix proteins.
Differences between immature bone and mature bone (woven bone and lamellar bone)
Woven bone is an immature bone characterized by intertwined collagen fibers oriented in many directions, hence the name. In lamellar bone, a distinctive, orderly arrangement is seen, which is the result of repeated addition of uniform lamellae to bony surfaces during appositional growth. The direction of collagen fibrils in any given lamella lies at right angles to that of the fibrils in the adjacent lamella.
In woven bone, there is a great amount of interfibrillar space that is occupied by mineral crystals and acidic proteins. In mature bone, the interfibrillar space is less.
The matrix of woven bone in hematoxylin and eosin section is tinged with blue indicating that it has higher proteoglycan content. Lamellar bone shows comparatively uniform acidophilic staining of the matrix.
Rates of deposition and mineralization are faster for woven bone than lamellar bone. Hence, woven bone shows higher proportion of osteocytes than lamellar bone.
Woven bone is enriched in BAG-75 (bone acidic glycoprotein- 75) and BSP (bone sialoprotein). Lamellar bone is enriched in osteocalcin. Osteopontin and type 1 collagen are seen in both woven and lamellar bone matrices.
Mineral density is lower and water content higher in woven bone matrix.
Woven bone can be entirely removed by osteoclasts, whereas only a portion of lamellar matrix of a given bone is resorbed at one time.
Matrix vesicles participate in initiation of mineralization of woven bone, whereas collagen mediated mechanism is operative in calcification of lamellar bone.
In woven bone, the osteocytes are isodiametric whereas in lamellar bone, osteocytes are flattened and oblate with their short axis parallel to the thickness of the lamellae.
Intracartilaginous (endochondral) bone formation
This type of ossification involves the replacement of a cartilaginous model by bone, and occurs at the extremities of all long bones, vertebrae, ribs, articular extremity of the mandible and base of the skull. The process involves the following steps.
Formation of a cartilaginous model
At the site where a limb will later emerge, the embryo shows outgrowth of mesoderm covered by ectoderm. The mesenchymal cells in this site condense, differentiate into chondroblasts and form the cartilage matrix, resulting in the development of a hyaline cartilage model. This process begins late in the second month of development. The model is surrounded by a perichondrium, made up of an inner chondrogenic layer and outer fibrous layer. No osteoblasts are produced by the cells in the chondrogenic layer, because differentiation is taking place in an avascular environment. Fibroblasts in fibrous layer produce collagen and a dense fibrous covering is formed.
The growth of the cartilage model is by interstitial and appositional growth. Increase in the length is by interstitial growth, due to repeated division of chondrocytes, along with production of additional matrix by the daughter cells. Widening of the model is due to further addition of matrix to its periphery by new chondroblasts, derived from the chondrogenic layer of the perichondrium. This is called appositional growth. In case of long bones as the differentiation of cartilage cells moves towards the metaphysis, the cells organize into longitudinal columns which are subdivided into three zones.
Zone of proliferation. The cells are small and flat, and constitute a source of new cells.
Zone of hypertrophy and maturation. This is the broadest zone. The chondrocytes hypertrophy, and in the early stages secrete type II collagen. As hypertrophy proceeds, proteoglycans are secreted. The increased cell size and increased cell secretion, lead to an increase in the size of the cartilaginous model. As the chondrocytes reach maximum size, they secrete type X collagen and noncollagenous proteins. Subsequently, there is partial breakdown of proteoglycans, creating a matrix environment receptive for mineral deposition.
Zone of provisional mineralization. Matrix mineralization begins in the zone of mineralization by formation of matrix vesicles. These membrane bound vesicles bud off from the cell and form independent units in the longitudinal septa of the cartilage.
Formation of bone collar
The capillaries grow into the perichondrium that surrounds midsection of the model. The cells in the inner layer of the perichondrium differentiate into osteoblasts in a vascular environment and form a thin collar of bone matrix around the mid region of the model. At this stage, periochondrium is referred to as periosteum as the differentiation of cells from the inner layer of the perichondrium is giving rise to bone. Vascularization of the middle of the cartilage occurs, and chondroclasts resorb most of the mineralized cartilage matrix. The bone collar holds together the shaft, which has been weakened by disintegration of the cartilage. Hence, more space is created for vascular ingrowth.
Formation of periosteal bud
Periosteal capillaries accompanied by osteogenic cells invade the calcified cartilage in the middle of the model and supply its interior. The osteogenic cells and the vessels comprise a structure called the periosteal bud. The periosteal capillaries grow into the cartilage model and initiate development of a primary ossification center. Osteogenic cells in the periosteal bud give rise to osteoblasts that deposit bone matrix on the residual calcified cartilage. This results in the formation of cancellous bone that has remnants of calcified cartilage. This is the mixed spicule. The network of mixed spicules is called primary spongiosa. The calcified cartilage in the trabeculae in hematoxylin and eosin sections stains pale blue to mauve, whereas bone matrix appears bright pink to red.
Formation of medullary cavity
As the primary ossification center enlarges, spreading proximally and distally, osteoclasts break down the newly formed spongy bone and open up a medullary cavity in the center of the shaft. Hematopoietic stem cells enter the medullary cavity giving rise to myeloid tissue.
The two ends of the developing bone are at this stage still composed entirely of cartilage. The midsection of the bone becomes the diaphysis and the cartilaginous ends of bone become the epiphysis. Hence, the primary center of ossification is the diaphyseal center of ossification.
Formation of secondary ossification center
At birth, most of the long bones have a bony diaphysis surrounding remnants of spongy bone, a widening medullary cavity, and two cartilaginous epiphysis. Shortly before or after birth, secondary ossification centers appear in one or both epiphysis. Initially chondrocytes in the middle of the epiphysis hypertrophy and mature, and the matrix partitions between their lacunae calcify.
Periosteal buds carry mesenchymal cells and blood vessels and process is same as that occurring in a primary ossification center, except that the spongy bone in the interior is retained and no medullary cavity forms in the epiphysis. The ossification spreads from secondary center in all directions. Eventually, the cartilage in the middle of epiphysis gradually gets replaced by cancellous bone. When secondary ossification is complete, hyaline cartilage remains at two places—on the articular surface as articular cartilage and at the junction of the diaphysis and epiphysis, where it forms the epiphyseal plates.
This plate continues to form new cartilage, which is replaced by bone, a process that increases the length of the bone. Long bones have one or two secondary ossification centers. Short bones have one ossification center. The union of primary and secondary ossification center is called epiphyseal line.
Long bones development depends on endochondral bone formation, which requires balance between hypertrophic cartilage (HC) formation and its ossification. Dysregulation of this process may result in skeletal dysplasia and heterotopic ossification. Endochondral ossification requires the precise orchestration of HC, vascularization, extracellular matrix remodeling, and the recruitment of osteoclasts and osteoblasts. Matrix metalloproteinase-9 (MMP-9), vascular endothelial growth factor (VEGF) and osteoclasts have all been shown to regulate endochondral ossification.
Calcification is the process of deposition of insoluble calcium salts in a tissue.
Mechanism of calcification. It is not entirely clear if the first formed solid phase is amorphous or crystalline. It is widely conceded that the first formed solid phase is amorphous. This initial phase is subsequently transformed to hydroxyapatite.
Amorphous calcium phosphate appears as microscopic spheres 30–100 nm in diameter, comprising randomly packed apatite crystals each of about 0.95 nm diameter.
Under normal conditions, there is insufficient concentration of available calcium and phosphate ions in blood and tissue fluid, for calcium phosphate to crystallize or precipitate spontaneously. The critical factor is the local (Ca2+) x. (Pi) ion product. Pi is the total free inorganic orthophosphate. When factors operate locally, to raise this ion product, calcium phosphate separates out in the solid phase and undergoes solid phase transition to a number of crystalline arrangements. Once the microcrystals have begun to form, they continue to grow and also catalyse further crystallization of calcium phosphate even at sites where the Ca2+. Pi product does not exceed the plasma level.
Theories of calcification
Traditionally, calcification has been treated from the point of view of precipitation dynamics, with initially the only ions needed being calcium and phosphate. The extracellular fluid is supersaturated with respect to the basic calcium phosphate, yet the mineralization is not a widespread phenomenon. Thus for precipitation to occur, the conditions in the osteoid matrix must be in some way especially favorable to this process. Interplay of various factors may contribute to conditions that may favor calcification.
A) Nucleation theory
Neumann and Neumann (1953) put forward the theory of epitactic nucleation based on the concept of seeding or epitaxy. A nucleus is formed, probably in relation to collagen, effective in aggregating calcium and phosphate ions. The hydroxyapatite crystals then grow spontaneously by addition of these from the saturated surrounding fluids. Acceptance of this theory led to many attempts to determine the nature and distribution of nucleation sites.
a) Ground substance. The components of ground substance, mainly sulfated glycosaminoglycans and proteoglycans, have been suggested for this function. Qualitative and quantitative changes of proteoglycans in the cartilaginous matrix prior to mineralization have been reported, thus supporting this phenomenon.
b) Collagen. The initial mineral deposits in the organic matrix of bone appear to occur at discrete sites in, or on the collagen fibrils.
The dimensions of water filled pore space in the matrix, particularly within the collagen macromolecular structure are crucial in allowing both the ingress of ions, the formation of ion clusters and the aggregation of such clusters to form nuclei from which crystal growth can continue. As mineralization progresses, axial periodicity of the collagen is first accentuated; this suggests that growth follows a specific fibril controlled pattern. Apart from collagen, other tissues closely associated with collagen also serve this function, e.g. phosphoproteins, including osteonectin. Phosphoproteins induce apatite formation, but are more involved in controlling the shape, size and orientation of bone crystals.
c) Mitochondria in osteoblasts play an important indirect role in the calcification process. Mitochondria are the earliest storage sites of calcium and phosphate in the form of amorphous calcium phosphate. This stored mineral is made available extracellularly, liberated directly due to cell destruction, or released indirectly in the form of constituent ions. This then supports the growth of crystals forming extracellularly, in association with matrix vesicles.
B) Role of matrix vesicles
The crystals have been found to be formed in association with matrix vesicles. Matrix vesicles are small membrane bound structures, 25 to 250 nm in diameter, lying free in the matrix, where calcification is known to be underway. These are rounded outgrowths of cell membrane that bud from osteoblasts, chondrocytes and odontoblasts. The vesicles are rich in phospholipids, especially phosphatidyl serine, a lipid with high affinity for calcium ions. Vesicles also contain annexins. Annexins in the vesicles form a calcium channel, thus incorporating the ion within the matrix vesicles. Matrix vesicles accumulate Ca2+ and their membranes furnish binding sites for the nucleation of hydroxyapatite crystals.
The mineralization of bone requires the presence of extracellular matrix vesicles, since the first step of mineralization is initiated inside these organelles. They serve as a site for Ca2+ and Pi accumulation by creating a specific environment where deposition of initial amorphous mineral complexes (nucleation) occurs and where hydroxyapatite (HA) is produced. The needle like crystals form on the inner surface of vesicle membrane.
Calcification
The extracellular matrix contains sufficiently high levels of Ca2+ and Pi concentrations to sustain the nucleation process and to propagate mineralization. Ion channels and transporters present in vesicle membrane are responsible for Ca2+ and Pi uptake into these organelles. The hydroxyapatite crystals are released into the extracellular matrix after reaching a certain thickness. The release of the crystals is proposed to be triggered by phospholipases leading to tissue calcification which affects the membrane fluidity. The released crystals serve as a template for the formation of crystalline arrays.
Their essential role in the initiation of calcification remains disputed and it is difficult to exclude the problem of artifactual precipitation of crystals in vesicles during specimen preparation for electron microscopy. They have not always been found in mineralizing fronts in osteoid of more mature bone and some have failed to find calcium tightly bound to matrix vesicles when using rapid freezing and freeze substitution of tissue to preserve ultrastructure.
C) Alkaline phosphatase is the enzyme which participates in the process of calcification. This enzyme hydrolyses a broad range of organic phosphate containing substrates and increases the local inorganic phosphate concentration. This enzyme resides in matrix vesicles budding from cell membranes of chondrocytes, osteoblasts and odontoblasts. Alkaline phosphatase hydrolyses PPi, which is an inhibitor of hydroxyapatite formation and also provides inorganic phosphates for the formation of hydroxyapatite crystals. It is used as a marker of active tissue mineralization.
Inhibitors of calcification
Collagen and other potential nucleating agents occur in tissues that do not calcify. The probable reason is that, the collagen molecules are packed closely together in soft tissues than in bone, which impede phosphate ion access to intrafibrillary nucleation sites. Pyrophosphate, diphosphonates or adenosine triphosphate can delay or prevent the transformation of amorphous calcium phosphate to hydroxyapatite. Other potential inhibitors include citrate, magnesium, and proteins like albumin. Components of the bone matrix may act locally and inhibit mineralization e.g. proteoglycans.
Bone resorption
Bone resorption is the removal of mineral and organic components of extracellular matrix of bone under the action of osteolytic cells, of which the most important is the osteoclast.
Sequence of events of bone resorption
The first phase involves the formation of osteoclast progenitors in the hematopoietic tissues, followed by their vascular dissemination and the generation of resting preosteoclasts and osteoclasts in the bone itself. The second phase consists of activation of osteoclasts at the surface of mineralized bone. Osteoblasts play a major role by retracting, to expose the mineral to the osteoclast and releasing a soluble factor that activates these cells. The third phase involves the activated osteoclasts resorbing the bone.
Alterations in the osteoclast
Immediately before the resorption event, the osteoclasts undergo changes by assuming a polarity of structure and function. These changes facilitate bone resorption. The two distinct alterations are the development of a ruffled border and a sealing zone at the plasma membrane. These changes occur only in the region of the cell that is next to the bone surface. The ruffled border consists of many infoldings of the cell membrane, resulting in fingerlike projections of the cytoplasm. Thus, an extensive surface is created well suited for an intensive exchange between the cell and bone.
At the periphery of the ruffled border, the plasma membrane is smooth and apposed closely to the bone surface. The adjacent cytoplasm, devoid of cell organelles contains contractile actin microfilaments, surrounded by two vinculin rings. This region is called the clear (sealing) zone. This zone serves to attach the cell very closely to the surface of bone and creates an isolated microenvironment, in which resorption can take place without diffusion of the hydrolytic enzymes produced by the cell into adjacent tissue. When osteoclasts arrive at the resorption site, they use the sealing zone to attach themselves to the bone surface. The attachment of the osteoclast cell membrane to the bone matrix at the sealing zone is due to the presence of cell membrane proteins known as integrins, especially αVβ3. Integrins are a large family of heteromeric cell surface receptors composed of noncovalently bound α and β subunits which interact with extracellular matrix molecules, serum constituents and adhesion molecules of immunoglobulin family. αVβ3, a vitronectin receptor, is expressed by resorbing osteoclasts. These integrins bind to specific amino acid sequences present in proteins of the bone matrix, namely, RGD (Arg-Gly-Asp).
Removal of hydroxyapatite
The initial phase involves the dissolution of the mineral phase by the action of hydrochloric acid (HCl). The protons for the acid arise from the activity of cytoplasmic carbonic anhydrase II, which is synthesized in the osteoclast. The protons are then released across the ruffled border into the resorption zone by an ATP consuming proton pump. This leads to a fall in pH to 2.5–3.0 in the osteoclast resorption space. The proton pump is an absolute requirement for normal bone resorption to take place.
Degradation of organic matrix
Organic constituents of bone tissue remain after the dissolution of mineralized component. Next step involves the digestion of organic components of matrix. Proteolytic enzymes are synthesized by osteoclasts, namely, cathepsin-K and MMP-9 (matrix metalloproteinase). The enzymes are synthesized in rough endoplasmic reticulum, transported to Golgi complexes and moved to the ruffled border in transport vesicles, and the contents of the same are released into sealed compartment, creating extracellular lysosomes. As a result, a visible depression or Howship's lacunae is excavated into the bone.
Cathepsin-K is the most important enzyme in bone resorption. It is a collagenolytic papain like cysteine protease expressed in osteoclasts. In vivo studies, have shown that, activation of cathepsin-K occurs intracellularly before secretion into lacunae and onset of bone resorption. The processing of procathepsin-K to mature cathepsin-K occurs as osteoclast approaches bone. Cathepsin-K degrades major amount of type I collagen and other noncollagenous proteins, which have been demineralized by the acidic environment of the resorptive zone.
MMP-9 (collagenase B) is believed to be required for osteoclast migration. MMP-13 is proposed to be involved in bone resorption and osteoclast differentiation.
Removal of degradation products from lacunae
Once liberated from bone, the free organic and nonorganic particles of bone matrix are taken in or endocytosed from the resorption lacunae, across the ruffled border, into the osteoclast. These are then packed in membrane bound vesicles within cytoplasm of osteoclast. These vesicles and their contents pass across the cell and fuse with FSD (functional secretory domain) a specialized region of the basal membrane. Then the vesicles are released by exocytosis. The changes in the cytoplasm framework in the cell and presence of clusters of matrix fragments in the region directly outside the cell next to FSD, indicate that matrix fragments have been expelled from the cell by exocytosis into extracellular space away from bone.
Following resorption, osteoclasts undergo apoptosis, which provides a mechanism for limiting resorptive activity. Factors like TGF-β and estrogen promote apoptosis. PTH and IL-1, act as suppressors prolonging osteoclast activity.
Differences between the resorbed and unresorbed surfaces
The resorbing surface is scalloped and exhibits scattered osteoclasts unlike osteoblasts, which line the bone surface.
Osteoclasts lie in the Howship's lacunae or resorption bays, whereas osteoblasts are present on the bone surface (Fig. 9.6).
The side of the osteoclast cell adjacent to bone contains few nuclei than the opposite side. Osteoblast is a mononucleated cell on the bone surface.
Role of TRAP in bone resorption
Tartrate resistant acid phosphatase (TRAP) is synthesized as a latent inactive proenzyme. The molecule is cleaved by an enzyme—cysteine proteinases converting it into an active form. This active enzyme plays a role in bone resorption inside and outside the osteoclast cell.
Extracellular role of TRAP
TRAP accumulates extracellularly in bone matrix, immediately adjacent to ruffled border of resorbing osteoclasts. Osteopontin, bone sialoprotein and osteonectin act as substrates for TRAP. Osteopontin is highly expressed at the bone surface, opposite sealing zone of resorbing osteoclasts, and is essential for resorption to take place. Osteopontin enables osteoclasts to adhere to bone surface by binding with integrin αVβ3, which are abundantly present at the sealing zone. TRAP can remove phosphate groups from osteopontin, an event that consequently disrupts adhesion of osteoclasts to the bone. This suggests that the enzyme might regulate osteoclast adhesion to the bone and also enable migration of osteoclasts to adjacent sites of resorption. The ability of TRAP to degrade phosphoproteins in bone by dephosphorylation may illustrate a preliminary stage in the degradation of the bone matrix. TRAP can hydrolyse and liberate pyrophosphate from bone matrix which is an inhibitor of resorption. This hydrolysis event would enable osteoclasts to begin bone resorption activity.
Intracellular role of TRAP
Intracellularly TRAP has been found to be co-localized with organic products of bone degradation released from bone matrix during resorption and endocytosed into osteoclasts. It has been put forward that TRAP containing vesicles fuse with transcytotic vesicles transporting the matrix degradation products from ruffled border to FSD (functional secretory domain) of osteoclasts. In this location, TRAP is secreted out of cells together with matrix degradation products. After this stage, both entities leak into the circulation at a rate that corresponds to the amount of resorption activity being undertaken by the osteoclast.
The enzyme is synthesized to help dispose of the products of bone breakdown within transcytotic vesicles. Along with fragments of bone matrix, it is released into the extracellular environment as an active enzyme by exocytosis at the FSD. TRAP then subsequently leaks into the circulation through the interstitial fluid.
Extracellular fate of TRAP
Once secreted TRAP is exposed to physiological influences present in body fluids. It binds to α2 macroglobulin, a high molecular weight molecule in serum. It has been proposed that, α2 macroglobulin may be a carrier molecule for TRAP, that mediates clearance of enzyme from areas of bone resorption and then the circulation. Ultimately, TRAP has the fate of all circulating enzymes. Its structure becomes compromised, leading to its inactivation as a catalyst. It loses its binuclear iron center, which is then recycled, and the iron free enzyme protein is broken down by proteases in the plasma and the liver. The fragments that result from these events are eventually metabolized by the liver and/or removed in the urine.
The concentration of osteoclast derived TRAP in serum can be assessed by immunoassay, and has a quantitative and dynamic relation to amount of resorption taking place on a day by day basis.
The immunoassay would provide additional information in the hospital's clinical laboratory for diagnosis and monitoring of bone resorption conditions.
Bone remodeling
Bone remodeling is performed by clusters of bone resorbing osteoclasts and bone forming osteoblasts arranged within temporary anatomical structures known as Basic Multicellular Units (BMUs). Traversing and encasing the BMU is a canopy of cells that creates a bone-remodeling compartment (BRC).
An active BMU consists of a leading front of bone resorbing osteoclasts. Reversal cells of unclear phenotype follow the osteoclasts, covering the newly exposed bone surface and prepare it for deposition of replacement bone. Osteoblasts occupy the tail portion of the BMU and secrete and deposit unmineralized bone matrix known as osteoid and direct its formation and mineralization into mature lamellar bone. At any given time, the process of bone synthesis and bone breakdown go on simultaneously (Fig. 9.7) and the status of the bone represents the net result of a balance between the two processes. This phenomenon is called ‘coupling’ of bone resorption and formation.
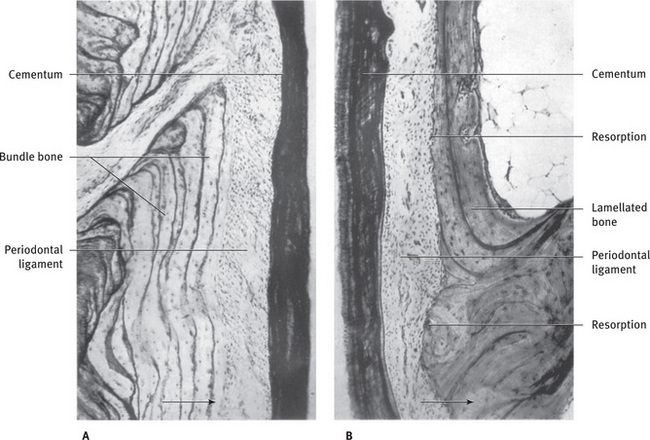
Fig. 9.7 Alveolar bone showing remodeling with (A) Apposition of bundle bone on distal alveolar wall and (B) Resorption of bone on mesial alveolar wall. Mesial drift indicated by arrow From Weinmann JP: Angle Orthod 11:83, 1941.
The main functions of remodeling are to prevent the accumulation of damaged and fatigued bone by regenerating new bone, allow the bone to respond to changes in mechanical forces and to facilitate mineral homeostasis.
Sequence of events in bone remodeling
Bone turnover rates of 30% to 100% per year are common in rapidly growing children. In adulthood, the rate is slow. The turnover rate of trabecular bone and endosteal surface of cortical bone is more than cortical bone turnover. The sequence of events is as follows:
Activation stage. The cells of the osteoblast lineage interact with hematopoietic cells to initiate osteoclast formation. This stage of bone remodeling involves detection of an initiating remodeling signal. This signal can take several forms, like direct mechanical strain on the bone that results in structural damage or hormonal action on bone cells in response to more systemic changes in homeostasis. Daily activity also places mechanical strain on the skeleton and osteocytes probably sense changes in these physical forces and translate them into biological signals that initiate bone remodeling.
Resorption stage. In this stage, osteoblasts respond to signals generated by osteocytes and recruit osteoclast precursors to the remodeling site. In addition to recruitment of osteoclast precursors, osteoblast expression of the master osteoclastogenesis cytokines, M-CSF, RANKL and Osteoprotegerin (OPG) is also modulated in response to PTH. OPG expression is reduced and M-CSF and RANKL production increased to promote osteoclast formation and subsequent activity. M-CSF promotes proliferation and survival of osteoclast precursors and directs spreading, motility and cytoskeletal organization in mature cells. RANKL also promotes proliferation of osteoclast precursors and additionally coordinates the differentiation of osteoclast precursors to multinucleated osteoclasts, promotes resorption activity and prolongs the life of the mature cells. Matrix metalloproteinases (MMPs), including MMP-13, are also secreted from osteoblasts in response to mechanical and endocrine remodeling signals. MMPs degrade the unmineralized osteoid that lines the bone surface and expose RGD adhesion sites within mineralized bone, that are necessary for osteoclast attachment.
The osteoclasts tunnel into surface of bone, which lasts for three weeks. In haversian canals, closest to the surface, osteoclasts travel along a vessel, resorb the haversian lamellae, and a part of circumferential lamellae, and form a resorption tunnel or cutting cone. Resorption tunnels can be distinguished from Haversian canals or Volkmann's canals as they have irregular etched outlines and presence of osteoclasts along the border.
Reversal stage. Resorption phase is followed by the reversal phase, comprising the differentiation of osteoblast precursors and discontinuation of bone resorption with osteoclast apoptosis. Following osteoclast-mediated resorption, the Howship's lacunae remain covered with undigested demineralized collagen matrix. A mononuclear cell of undetermined lineage removes these collagen remnants and prepares the bone surface for subsequent osteoblast-mediated bone formation. This reversal cell is from the osteoblast lineage, based on cell morphology and the positive expression for alkaline phosphatase.
However, the mesenchymal bone lining cells are more ideally equipped to deposit the collagenous matrix that forms along osteopontin-rich cement lines within Howship's lacunae. The final role of the reversal cells may be to receive or produce coupling signals that allow transition from bone resorption to bone formation within the BMU.
In the next stage, activated osteoblasts lay down new bone material, until the resorbed bone is entirely replaced by new one. Once mesenchymal stem cells or early osteoblast progenitors have returned to the resorption lacunae, they differentiate and secrete molecules that ultimately form replacement bone.
Collagen type I is the primary organic component of bone. Noncollagenous proteins, including proteoglycans, Gla-containing proteins (matrix Gla protein and osteocalcin) and lipids comprise the remaining organic material.
These osteoblasts lay down a new set of haversian lamellae, encircling a vessel upon a reversal line. This cement line is a thin layer of glycoproteins comprising bone sialoprotein and osteopontin, that acts as a cohesive mineralized layer between the old bone and new bone to be secreted. The entire area of osteon, where active formation occurs is termed the filling cone. Fragments of lamellae from old bone haversian systems are left behind as interstitial lamellae (Fig. 9.4).
For bone to assume its final form, hydroxyapatite is incorporated into this newly deposited osteoid.
Osteoblasts that become encased in the new bone are transformed into osteocytes. The osteoblasts become quiescent at the end of bone remodeling and form flattened lining cells on the bone surface until a new remodeling cycle is triggered.
Mediators of bone remodeling
Hormones
Parathyroid hormone (PTH) is produced in the parathyroid glands in response to hypocalcemia, stimulating bone resorption. A stimulating role in bone formation has also been established through the synthesis of IGF 1 and TGF β. This dual effect of resorption and formation is explained by the fact that the continuous supply of PTH stimulates bone resorption through the synthesis of RANKL on the part of the osteoblastic cells, while at intermittent doses, it would stimulate the formation of bone, associated with an increase of the growth factors and with a decrease in the apoptosis of osteoblasts.
Calcitonin is secreted when blood calcium levels rise. It inhibits bone resorption and promotes calcium salt deposition in bone matrix, effectively reducing blood calcium levels. As blood calcium levels fall, calcitonin release wanes as well. Calcitonin also reduces the number and activity of osteoclasts.
Vitamin D metabolites
The major active metabolite of vitamin D is 1,25-dihydroxycholecalciferol. This has been shown to affect bone formation and also to cause bone resorption. Its effect on bone resorption appears to be by the differentiation of committed progenitor cells into mature cells. It favors the intestinal absorption of calcium and phosphate and therefore bone mineralization. It is necessary for normal growth of the skeleton.
Estrogen receptors are present on osteoblasts, osteocytes and osteoclasts. They favor bone formation, increasing the number and function of osteoblasts. It has also been proposed that these hormones are believed to increase the levels of OPG, which inhibits resorption.
Growth hormones act directly on the osteoblasts stimulating their activity and increasing the synthesis of collagen, osteocalcin and alkaline phosphatase. Indirect action is produced through an increase in the synthesis of IGF-I and II by osteoblasts, which stimulate the proliferation and differentiation of osteoblasts.
Glucocorticoids at high doses inhibit the synthesis of IGF-1 by osteoblasts and suppress BMP-2 and Cbfa-1 which are essential for the formation of osteoblasts. But, at physiological doses they are believed to have osteogenic capacity favoring osteoblastic differentiation.
Local factors
IL-1 does not have direct action on the osteoclast, but like PTH acts via the osteoblast. It has a direct promotional effect on osteoclast formation. It inhibits the apoptosis of osteoclasts.
TNF-α and TNF-β stimulate bone resorption in vitro. The bone resorbing effect is mediated through osteoblasts. TNF is also believed to inhibit bone collagen and noncollagenous protein synthesis.
Prostaglandins are local pathological mediators of bone destruction, in particular, where there is inflammation.
IGF-I and II increase the number and function of osteoblasts stimulating collagen synthesis. They mediate osteoblast– osteoclast interaction and participate in bone remodeling.
BMPs are highly abundant in bone tissue and participate in the formation of bone and cartilage. They are the most important factors for osteoblast differentiation.
Other growth factors like PDGF, FGF and EGF may also play an important role in physiologic remodeling and an even more important role in remodeling associated with skeletal repair. PDGF stimulates protein synthesis by osteoblasts and also favors bone resorption. FGF is a mitogen for osteoblasts. In bone, EGF has both, a formative and destructive function.
Bacterial products such as lipopolysaccharide, capsular material, lipoteichoic acids and peptidoglycans may act as foreign antigens and induce monocytes, macrophages and then bone cells to produce prostaglandins and cytokines such as IL-1, leading to bone resorption.
Mechanical factors Under muscular action, tension is transmitted to the bone, which is detected by osteocyte network. These osteocytes produce prostaglandins and IGF-1 which stimulate osteoblast activity leading to increased bone formation. Absence of muscular activity accelerates resorption.
Markers of bone turnover
The markers of bone formation are (Serum markers)
The markers of bone resorption are (Urinary markers)
- collagen crosslink fragments (first to be hydrolysed)
- urine N-telopeptide (N terminus of collagen fibrils)
Serum markers
- Serum β2 macroglobulin is a marker for high bone remodeling. It plays a role as bone derived growth factor regulating osteoblasts and osteoclasts. It has been proposed as a bone remodeling biological marker in high bone turnover conditions, especially vitamin D deficiency and secondary hyperparathyroidism.
Pathologies caused by improper control of remodeling are; osteoporosis, osteopetrosis, malignant bone tumors inflammatory joint diseases, hyperparathyroidism, Paget's disease and hyperthyroidism.
Alveolar bone
The alveolar process is defined as that part of the maxilla and the mandible that forms and supports the sockets of the teeth.
Functions of alveolar bone are
- Anchors the roots of teeth to the alveoli, which is achieved by the insertion of Sharpey's fibers into the alveolar bone proper.
- Helps to move the teeth for better occlusion.
- Helps to absorb and distribute occlusal forces generated during tooth contact.
- Supplies vessels to periodontal ligament.
- Houses and protects developing permanent teeth, while supporting primary teeth.
Development of alveolar process
Near the end of the second month of fetal life, the maxilla as well as the mandible form a groove that is open towards the surface of the oral cavity (Figs. 9.8, 9.9).
Fig. 9.8 Development of mandible as intramembranous bone lateral to Meckel's cartilage (human embryo 45 mm in length).
Fig. 9.9 Development of mandibular symphysis and formation of bony septa between developing teeth. (A) Newborn infant. Symphysis wide open. Mental ossicle (radiograph). (B) Child 9 months of age. Symphysis partly closed. Mental ossicles fused to mandible and bony septa between developing teeth seen (radiograph). (C) Frontal section through mandibular symphysis of newborn infant. Connective tissue in midline connects plates of cartilage on either side. Cartilage is later replaced by bone.
Tooth germs develop within the bony structures at late bell stage. Bony septa and bony bridge begin to form and separate the individual tooth germs from one another, keeping individual tooth germs in clearly outlined bony compartments. Along with tooth germs, alveolar nerves and vessels are also present. At this stage, dental follicle surrounds each tooth germ, which is located between tooth germs and its bony compartment. Even prior to root formation, tooth germs within bony compartments show bodily movement in various directions to adjust to growing jaws. This movement causes bony remodeling of bony compartment through bone resorption and bone deposition. The major changes in the alveolar process begin to occur with development of roots and tooth eruption. As roots develop, the alveolar process increases in height. Also, the cells in the dental follicle start to differentiate into periodontal ligament and cementum. At the same time, some cells in the dental follicle differentiate into osteoblasts and form alveolar bone proper.
Hence, an alveolar process in the strict sense of the word develops only during the eruption of the teeth. It is important to realize that, during growth, part of the alveolar process is gradually incorporated into the maxillary or mandibular body while it grows at a fairly rapid rate at its free borders. During the period of rapid growth, a tissue may develop at the alveolar crest that combines characteristics of cartilage and bone. It is called chondroid bone (Fig. 9.10). The alveolar process forms with the development and the eruption of teeth, and conversely, it gradually diminishes in height after the loss of teeth.
Structure of the alveolar bone
Anatomically, no distinct boundary exists between the body of the maxilla or the mandible and their respective alveolar processes. In some places, the alveolar process is fused with and partly masked by, bone that is not functionally related to the teeth. In the anterior part of the maxilla, the palatine process fuses with the oral plate of the alveolar process. In the posterior part of the mandible, the oblique line is superimposed laterally on the bone of the alveolar process. As a result of its adaptation to function, two parts of the alveolar process can be distinguished, the alveolar bone proper and the supporting alveolar bone.
Alveolar bone proper
The alveolar bone proper consists partly of lamellated and partly of bundle bone and is about 0.1–0.4 mm thick. It surrounds the root of the tooth and gives attachment to principal fibers of the periodontal ligament.
Lamellated bone
The lamellar bone contains osteons each of which has a blood vessel in a haversian canal. Blood vessel is surrounded by concentric lamellae to form osteon. Some lamellae of the lamellated bone are arranged roughly parallel to the surface of the adjacent marrow spaces, whereas others form haversian systems.
Bundle bone
Bundle bone is that bone in which the principal fibers of the periodontal ligament are anchored. The term ‘bundle’ was chosen, because, the bundles of the principal fibers continue into the bone as Sharpey's fibers (Fig. 9.11). The bundle bone is characterized by the scarcity of the fibrils in the intercellular substance. These fibrils, more over, are all arranged at right angles to Sharpey's fibers. The bundle bone contains fewer fibrils than does lamellated bone, and therefore it appears dark in routine hematoxylin and eosin stained sections and much lighter in preparations stained with silver than does lamellated bone. These fibers are mineralized at the periphery and have a larger diameter. These fibers are less numerous than the corresponding fiber bundles in the cementum on the opposite side of the periodontal ligament. The collagen adjacent to bone is always less mature than that adjacent to cementum. In some areas, the alveolar bone proper consists mainly of bundle bone. Bundle bone is formed in areas of recent bone apposition. Lines of rest are seen in bundle bone.
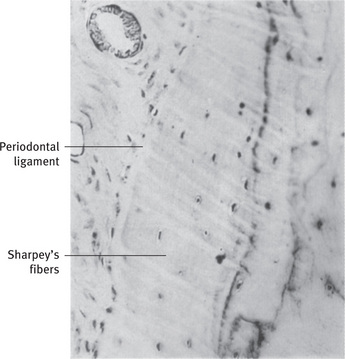
Fig. 9.11 Histologic section showing Sharpey's fibers in alveolar bone proper From Bhaskar SN: Synopsis of oral histology, St Louis, 1962, The CV Mosby Co.
Radiographically, it is also referred to as the lamina dura, because, of increased radiopacity, which is due to the presence of thick bone without trabeculations, that X-rays must penetrate and not to any increased mineral content.
The alveolar bone proper, which forms the inner wall of the socket is perforated by many openings that carry branches of the interalveolar nerves and blood vessels into the periodontal ligament, and it is therefore called the cribriform plate (Fig. 9.12). Bone between the teeth is called interdental septum and is composed entirely of cribriform plate. The interdental and interradicular septa contain the perforating canals of Zuckerkandl and Hirschfeld (nutrient canals) which house the interdental and interradicular arteries, veins, lymph vessels and nerves (Fig. 9.13).
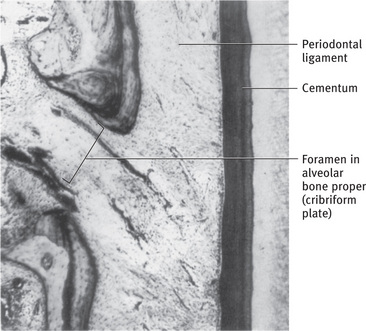
Fig. 9.12 Histologic section showing foramen in alveolar bone proper (cribriform plate) From Bhaskar SN: Synopsis of oral histology, St Louis, 1962, The CV Mosby Co.
Cortical plates
Cortical plates consist of compact bone and form the outer and inner plates of the alveolar processes. The cortical plates, continuous with the compact layers of the maxillary and mandibular body, are generally much thinner in the maxilla, than in the mandible. They are thickest in the premolar and molar region of the lower jaw, especially on the buccal side. In the maxilla, the outer cortical plate is perforated by many small openings through which blood and lymph vessels pass.
In the region of the anterior teeth of both jaws, the supporting bone usually is very thin. No spongy bone is found here, and the cortical plate is fused with the alveolar bone proper (Fig. 9.14). In such areas, notably in the premolar and molar regions of the maxilla, defects of the outer alveolar wall are fairly common. Such defects, where periodontal tissues and covering mucosa fuse, do not impair the firm attachment and function of the tooth.
Fig. 9.14 Gross relations of alveolar processes. (A) Horizontal section through upper alveolar process. (B) Labiolingual section through upper lateral incisor. (C) Labiolingual section through lower canine. (D) Labiolingual section through lower second molar. (E) Labiolingual section through lower third molar From Sicher H and Tandler J: Anatomie für Zahnärzte [Anatomy for dentists], Vienna, 1928, Julius Springer Verlag.
Bone underlying the gingiva is the cortical plate. Both cribriform plate and cortical plate are compact bone separated by spongy bone.
Histologically, the cortical plates consist of longitudinal lamellae and haversian systems (Fig. 9.4). In the lower jaw, circumferential or basic lamellae reach from the body of the mandible into the cortical plates.
Spongy bone
Spongy bone fills the area between the cortical plates and the alveolar bone proper. It contains trabeculae of lamellar bone. These are surrounded by marrow that is rich in adipocytes and pluripotent mesenchymal cells.
The trabeculae contain osteocytes in the interior and osteoblasts or osteoclasts on the surface. These trabeculae of the spongy bone buttress the functional forces to which alveolar bone proper is exposed. The cancellous component in maxilla is more than in the mandible. The study of radiographs permits the classification of the spongiosa of the alveolar process into two main types.
In type I the interdental and interradicular trabeculae are regular and horizontal in a ladder like arrangement (Fig. 9.15).
Fig. 9.15 Supporting trabeculae between alveoli. (A) Radiograph of mandible. (B) Mesiodistal section through mandibular molars showing alveolar bone proper and supporting bone. (C) Type I alveolar spongiosa. Note regular horizontal trabeculae. (D) Type II alveolar spongiosa. Note irregularly arranged trabeculae (Courtesy Dr N Brescia, Chicago).
Type II shows irregularly arranged, numerous, delicate interdental and interradicular trabeculae. Both types show a variation in thickness of trabeculae and size of marrow spaces. The architecture of type I is seen most often in the mandible and fits well into the general idea of a trajectory pattern of spongy bone. Type II, although evidently functionally satisfactory, lacks a distinct trajectory pattern, which seems to be compensated for by the greater number of trabeculae in any given area. This arrangement is more common in the maxilla.
From the apical part of the socket of lower molars, trabeculae are sometimes seen radiating in a slightly distal direction.
These trabeculae are less prominent in the upper jaw, because of the proximity of the nasal cavity and the maxillary sinus. In the condylar process, in the angle of the mandible, in the maxillary tuberosity, and in other isolated foci, hematopoietic cellular marrow is found.
Crest of the alveolar septa
The shape of the outlines of the crest of the alveolar septa in the roentgenogram is dependent on the position of the adjacent teeth. In a healthy mouth the distance between the cementoenamel junction and the free border of the alveolar bone proper is fairly constant. If the neighboring teeth are inclined, the alveolar crest is oblique. In the majority of individuals the inclination is most pronounced in the premolar and molar regions, with the teeth being tipped mesially. Then the cementoenamel junction of the mesial tooth is situated in a more occlusal plane than that of the distal tooth, and the alveolar crest therefore slopes distally (Fig. 9.16). Cortical bone and alveolar bone meet at the alveolar crest usually 1.5 to 2 mm below the level of the cementoenamel junction on the tooth it surrounds.
Internal reconstruction of alveolar bone
Mesial drift and continuous tooth eruption elicit remodeling of alveolar bone proper. During the mesial drift of a tooth, bone is apposed on the distal and resorbed on the mesial alveolar wall (Fig. 9.7). The distal wall is made up almost entirely of bundle bone. However, the osteoclasts in the adjacent marrow spaces remove part of the bundle bone, when it reaches a certain thickness. In its place, lamellated bone is deposited.
On the mesial alveolar wall of a drifting tooth, the sign of active resorption is the presence of Howship's lacunae containing osteoclasts. Bundle bone, however, on this side is always present in some areas but forms merely a thin layer (Fig. 9.17). This is because the mesial drift of a tooth does not occur simply as a bodily movement. Thus resorption does not involve the entire mesial surface of the alveolus at one and the same time. Moreover, periods of resorption alternate with periods of rest and repair. It is during these periods of repair that bundle bone is formed, and detached periodontal fibers are again secured. Islands of bundle bone are separated from the lamellated bone by reversal lines that turn their convexities towards the lamellated bone (Fig. 9.17).
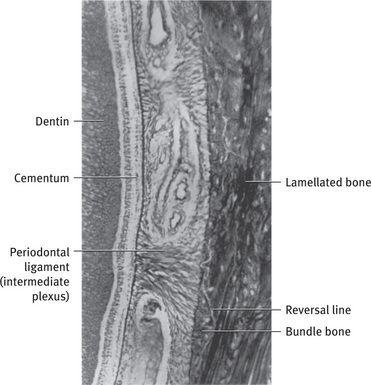
Fig. 9.17 Mesial alveolar wall where alveolar bone proper consists mostly of lamellated bone and islands of bundle bone, which anchor principal fibers of periodontal ligament.
During these changes, compact bone may be replaced by spongy bone or spongy bone may change into compact bone. This type of internal reconstruction of bone can be observed in physiologic mesial drift or in orthodontic mesial or distal movement of teeth. In these movements an interdental septum shows apposition on one surface and resorption on the other. If the alveolar bone proper is thickened by apposition of bundle bone, the interdental marrow spaces widen and advance in the direction of apposition. Conversely, if the plate of the alveolar bone proper is thinned by resorption, apposition of bone occurs on those surfaces that face the marrow spaces. The result is a reconstructive shift of the interdental septum.
Alterations in the structure of the alveolar bone are of great importance in connection with the physiologic eruptive movements of the teeth. These movements are directed mesioocclusally. At the alveolar fundus the continual apposition of bone can be recognized by resting lines separating parallel layers of bundle bone. When the bundle bone has reached a certain thickness, it is resorbed partly from the marrow spaces and then replaced by lamellated bone or spongy trabeculae. The presence of bundle bone indicates the level at which the alveolar fundus was situated previously.
Age changes
- Alveolar sockets appear jagged and uneven.
- The marrow spaces have fatty infiltration.
- The alveolar process in edentulous jaws decreases in size.
- Loss of maxillary bone is accompanied by increase in size of the maxillary sinus.
- Internal trabecular arrangement is more open, which indicates bone loss.
- The distance between the crest of the alveolar bone and CEJ increases with age—approximately by 2.81 mm.
Clinical considerations
Bone, although one of the hardest tissues of the human body, is biologically a highly plastic tissue. Where bone is covered by a vascularized connective tissue, it is exceedingly sensitive to pressure, whereas tension acts generally as a stimulus to the production of new bone. It is this biologic plasticity that enables the orthodontist to move teeth without disrupting their relations to the alveolar bone. Bone is resorbed on the side of pressure and apposed on the side of tension; thus the entire alveolus is allowed to shift with the tooth. It has been shown that on the pressure side there is an increase in the level of cyclic adenosine monophosphate (cAMP) in cells. This may play some role in bone resorption. At sites of alveolar bone compression, osteoclasts proliferate and initial resorption of the superficial bone takes place. It is believed that, the initial response may involve osteoblasts which can produce collagenolytic enzymes to remove a portion of unmineralized extracellular matrix, thereby, facilitating access of osteoclast precursors to the bone surface. Osteoblastic cells also produce cytokines and chemokines, which can attract monocyte precursors and promote osteoclast differentiation. In quiescent areas, retraction or apoptotic death of bone lining cells will expose the mineralized bone surface to osteoclasts.
At sites of tension, osteoblasts are activated to produce osteoid that subsequently mineralizes to form new bone.
The adaptation of bone to function is quantitative as well as qualitative. Whereas, increase in functional forces leads to formation of new bone, decreased function leads to a decrease in the volume of bone. This can be observed in the supporting bone of teeth that have lost their antagonists. Here the spongy bone around the alveolus shows pronounced rarefaction. The bone trabeculae are less numerous and very thin (Fig. 9.18). The alveolar bone proper, however, is generally well preserved because it continues to receive some stimuli from the tension of the periodontal tissues.
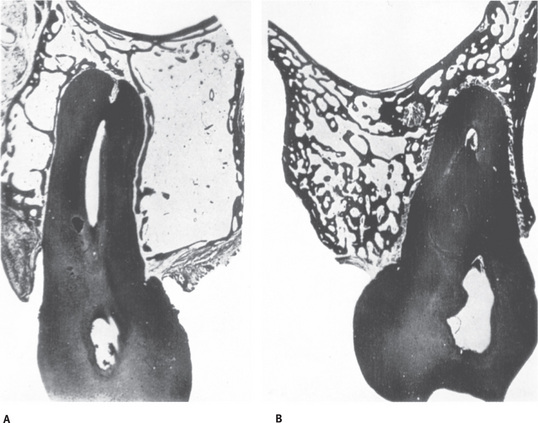
Fig. 9.18 Osteoporosis of alveolar process caused by inactivity of tooth that has no antagonist. Labiolingual sections through upper molars of same individual. (A) Disappearance of bony trabeculae after loss of function. Plane of mesiobuccal root. Alveolar bone proper remains intact. (B) Normal spongy bone in plane of mesiobuccal root of functioning tooth From Kellner E: Z Stomatol 18:59, 1920.
During healing of fractures or extraction wounds, an embryonic type of bone is formed, which only later is replaced by mature bone. The embryonic bone also called immature or coarse fibrillar bone, is characterized, among other aspects, by the greater number, size and irregular arrangement of the osteocytes than are found in mature bone (Fig. 9.3). The greater number of cells and the reduced volume of calcified intercellular substance render this immature bone more radiolucent than mature bone. This explains why bony callus cannot be seen in radiographs at a time when histologic examination of a fracture reveals a well-developed union between the fragments and why a socket after an extraction wound appears to be empty at a time, when it is almost filled with immature bone. The visibility in radiographs lags 2 or 3 weeks behind actual formation of new bone.
The most frequent and harmful change in the alveolar process is that which is associated with periodontal disease. The bone resorption is almost universal, occurs more frequently in posterior teeth, is usually symmetrical, occurs in episodic spurts, is both of the horizontal and vertical type (i.e., occurs from the gingival and tooth side, respectively), and is intimately related to bacterial plaque and pocket formation. It has been shown, for example, endotoxins produced by the gramnegative bacteria of the plaque lead to an increase in cAMP, which increases the osteoclastic activity.
Resorption after tooth loss has been shown to follow a predictable pattern. The labial aspect of the alveolar crest is the principal site of resorption, which reduces first in width and later in height.
The pattern of resorption is different in the maxilla and mandible. The residual alveolar ridge resorbs downward and outward in the mandible, whereas, in the maxilla the resorption is upwards and inwards. Nontraumatic loss of anterior maxillary teeth is followed by a progressive loss of bone mainly from the labial side. In the deciduous dentition, loss of a retained second deciduous molar, which has no succedaneous permanent tooth to replace it, is also associated with bone loss. The cause for resorption of alveolar bone after tooth loss has been assumed to be due to disuse atrophy, decreased blood supply, localized inflammation or unfavorable prosthesis pressure.
Alveolar ridge defects and deformities can also be the result of congenital defects, trauma, periodontal disease or surgical ablation, as in the case of tumor surgery.
Lamina dura is an important diagnostic landmark in determining health of the periapical tissues. Loss of density usually means infections, inflammation and resorption of bone socket.
Therapeutic considerations
Traditional treatment methods for promoting bone healing primarily utilize bone grafts or synthetic materials to fill the defects and provide structural support.
Bone grafting to stimulate bone deposition has been used in periodontal surgery since the 1970s. It involves a surgical procedure to place bone or bone substitute material into a bone defect with the objective of producing new bone and possibly the regeneration of periodontal ligament and cementum.
Autografts utilize the patient's bone, which can be obtained from intraoral or extraoral sites. They are the best materials for bone grafting, are very well accepted by the body and may produce the fastest rate of bone growth; however, there is the potential risk of additional discomfort and a secondary procedure. With autografts, the patient is assured of protection from disease transmission and/or immune reaction.
Allografts are obtained from another human source, typically highly processed bone powder from human cadavers. The age and health of the donor can affect the rate of bone regeneration. The risk of disease transmission and/or rejection is handled by processing and quality control. The allografts are freeze-dried at ultra-low temperatures and dried under high vacuum. They are available either demineralized or non-demineralized. Unlike synthetic bone, which only provides scaffolding for osteoconduction, allografts include growth factors which are also osteoinductive. Allografts induce bone growth and provide an environment that increases the body's regenerative process.
Xenografts are obtained from animal sources; usually cows and/or pigs. They include processed animal bone or growth proteins. Again, the risk of disease transmission and/or rejection is reduced by processing.
The drawbacks of using bone grafts include a lack of viable bone tissue available for autografts and allografts transplantation and potential disease transmission with allografts and xenografts.
In cases where bone grafts from human or animal sources are not feasible, synthetic graft materials (alloplasts) are used.
Synthetic bone grafting materials
Examples of synthetic materials include natural and synthetic hydroxyapatites, ceramics, calcium carbonate (natural coral), silicon-containing glasses, and synthetic polymers.
Synthetic materials carry no risk of disease transmission or immune system rejection. They help create an environment that facilitates the body's regenerative process.
A guided tissue regeneration approach has evolved which is an epithelial exclusionary technique. This technique facilitates the cells of the periodontal ligament to differentiate into osteoblasts, cementoblasts and fibroblasts resulting in regeneration of the lost periodontium. It involves use of membranes, nonresorbable and/or resorbable, which prevent the downgrowth of the epithelium thereby promoting regeneration. Example of a resorbable material is collagen and nonresorbable material is ePTFE (expanded polytetrafluoroethylene) membrane.
Biologically mediated strategies
Biologically mediated strategies include materials, such as enamel matrix proteins, that can be premixed with vehicle solution. They are intended as an adjunct to periodontal surgery for topical application onto exposed root surfaces or bone to treat intrabony defects and furcations due to moderate or severe periodontitis. Enamel matrix protein leave only a resorbable protein matrix on the root surface, which makes bone more likely to regenerate. These bone morphogenetic proteins help to initiate a cascade of events leading to the differentiation of progenitor cells into phenotypes involved in periodontal regeneration.
Bone tissue engineering has emerged as a new therapeutic alternative to promote bone healing. This approach aims to regenerate or repair bone tissue with various combinations of polymeric scaffolds, cells and inductive factors into a system that actively stimulates tissue formation.
- Which simply guide and support bone regeneration— Osteoconductive
- Which actively stimulate bone regeneration via the delivery of inductive factors—Osteoinductive.
These scaffolds are a porous composite material composed mainly of hydroxyapatite in a collagen I matrix. These materials promote osteoblasts and osteoprogenitor cell attachment and differentiation to enhance bone tissue formation.
Other materials used for bone tissue engineering include synthetic, biocompatible and biodegradable polymers such as poly (lactide-co-glycolide)
– For filling small defects, injectable polymers such as alginate or poly (ethylene glycol) are preferred.
Cells. Osteoblasts and osteoprogenitor cells have been incorporated into various scaffolds to enhance bone repair. These cells must be obtained from patient or donor. Stem cells derived from bone marrow periosteum, adipose tissue, skeletal muscle or baby teeth can be induced to differentiate into diverse cell types such as muscle, nerve, cartilage, bone and fat.
Direct or gene therapy approaches to the delivery of osteoinductive factors are also promising approaches for bone regeneration.
Controlled release of proteins from a polymeric scaffold allows localized sustained protein delivery and may be a more effective means to enhance bone formation.
Delivery of BMP-2 from absorbable collagen sponge induces bone formation and heals bony defects. BMP-2 can also be delivered from synthetic materials such as poly (ethylene glycol) hydrogels to promote repair of bone defects. Other than BMP-2, sustained release of FGF-2 and IGF-1 can also accelerate bone formation.
References
Atkins, G.J., Panagiota Kostakis, Beiquing, P.A.N., et al. RANKL expression is related to the differentiation state of human osteoblasts. J Bone Mineral Res. 2003; 18:1088.
Avery, J., Steele, P.F., Nancy, A. Oral Development and Histology, 3rd. Stuttgart: Thieme; 2001.
Balcerzak, M., Hamade, E., Le Zhang, et al. The roles of annexins and alkaline phosphatase in mineralization process. Acta Biochima Polonica. 2003; 50:1019.
Barbara Pavan, Dan Zhou, Brandon Whitman, Marco Fornari and Mary Tecklenburg: Bone biomineralisation: in depth analysis of hydroxyapatite crystallization through experiments and stimulations: http://www.icors2010.org/abstractfiles/ICORS20100493.4500VER.1.pdf.
Bartold, P.M., Songtao Shi, Gronthos, S. Stem cells and periodontal regeneration. Periodontology 2000. 2006; 40:164.
Berkovitz, B.K., Holland, G.H., Moxham, B.J. Oral anatomy, histology and embryology, 3rd. St Louis: Mosby; 2002. [pp. 205-219].
Bezerra, M.C., Carvalho, J.F., Prokopowitsch, A.S., et al. RANK, RANKL and Osteoprotegerin in arthritic bone loss. Braz J Med Biol Res. 2005; 38(2):161–170.
Bhaskar, S.N. Radiographic interpretation for the dentist, 3. St Louis: The CV Mosby Co; 1979.
Bhaskar, S.N., Mohammed, C., Weinmann, J. A morphological and histochemical study of osteoclasts. J Bone Joint Surg (Am). 1956; 38:1335.
Brodie, A.G. Some recent observations on the growth of the mandible. Angle Orthod. 1940; 10:63.
Brodie, A.G. On the growth pattern of the human head from the third month to the eighth year of life. Am J Anat. 1941; 68:209.
Bull, H., Murray, P.G., Thomas, D., et al. Acid phosphatases. Molecular Pathology. 2002; 55:65.
Cormack, D.H. HAMS Histology, 9th. Philadelphia: Lippincott; 1979.
Cormack, D.H., Ham, A.W. Ham's Histology, 9th Sub. Philadelphia: Lippincott Williams and Wilkins; 1987.
Council on Dental Materials, Instruments and Equipment, American Dental Association: Hydroxyapatite, beta tricalcium phosphate and autogenous and allogeneic bone for filling periodontal defects, alveolar ridge augmentation, and pulp capping. J Am Dent Assoc. 1984; 108:822.
David, C.V. Bone replacement grafts—the bone substitutes. Dental Clin North Am Saunders. 1998; 42:491.
Dodds, R.A., James, I.E., Rieman, D., et al. Human osteoclast Cathepsin-K is processed intracellularly prior to attachment and bone resorption. J Bone Miner Res. 2001; 16(3):478.
Fernandez, et al. Physiological bases of bone regeneration II. The remodeling process. Med Oral Patol Oral Cir Bucal. 2006; 11:E151.
Francis, H.J. Effects of growth factors and cytokines on osteoblast differentiation. Periodontology 2000. 2006; 41:48.
Giannobile, W.V., Rios, H.F., Lang, N.P.. 5th. Bone as a tissue. Clin Periodontol and Implant Dentistry; 1. Wiley-Blackwell, 2008.
Glimcher, M.J. Composition, structure and organization of bone and other mineralized tissues and the mechanism of calcification. In: Greep, R.O., Aptwood, E.B., eds. The handbook of physiology, 7. Washington, DC: American Physiological Society; 1976. [Section 7, 25-116].
Gorski JP: Division of molecular biology and biochemistry, School of biological sciences Is all bone the same. Distinctive distribution and properties of non collagenous matrix proteins in lamellar vs woven bone imply the existence of different underlying osteogenic mechanisms.
Hall, D.A. Glycoproteins and proteoglycans.the aging of connective tissue. New York: Academic Press, Inc.; 1976.
Hideo Orimo:. The mechanism of mineralization and the role of alkaline phosphatase in health and disease. J Nippon Med Sch. 2010; 77:4.
Takayanagi, Hiroshi, Akinori Takooka, Kojiro Sato, et al. Immunological Reviews. 2005; 208:181.
Hock, J.M., Krishnan, V., Onyia, J.E., et al. Osteoblast apoptosis and bone turnover. Journal of bone and mineral research. 2001; 16:975.
Ilcho, M., Garant, R.P. Development and general structure of the periodontium. Periodontology 2000. 2000; 24:9.
Ishimi, Y., Miyaura, C., Jin, C.H., et al. IL-6 is produced by osteoblasts and induces bone resorption Journal of immunology. 1990; 145(10):3297.
Kaban, L.B., Glowacki, J. Augmentation of rat mandibular ridge with demineralized bone implants. J Dent Res. 1984; 63:998.
Karsenty, G. Mini review: Transcriptional control of osteoblast differentiation. Endocrinology. 2001; 142:7.
Katagiri, T., Takashi, N. Regulatory mechanisms of osteoblast and osteoclast differentiation (Review Article) Bone Biology. Oral Diseases. 2002; 8:147.
Lavelle, L.B. Applied oral physiology, 2nd. Wright; 1998.
Lawrence, R.G. Physiology and pathophysiology of bone remodeling. Clinical Chemistry. 1999; 45:1352.
Lehlinger, A.L. Mitochondria calcium ion transport. Biochem J. 1970; 119:129.
Lindhe, J., Lang, N.P., Karring, T.. 5th. Clin Periodontol and Implant Dentistry; 2. Willey-Blackwell, 2008.
Manappalli, J.J. Complete denture prosthodontics physical and biomechanical considerations, Ist. Arya Publishing House; 2004. [13-14].
Manolagas Stavros, C. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocrine Reviews. 2000; 21(2):115.
Marieb, E.N. Human Anatomy and physiology, 3rd. Benjamin/ Cummings Publishing Company; 1995.
Marks, S.C., Schneider, G. Evidence for a relationship between lymphoid cells and osteoclasts. Am J Anat. 1978; 152:331.
Martin, J.T. Osteoblast derived PTHrP is a physiological regulator of bone formation. J Clin Invest. 2005; 115:2322.
Millet, P.J. Synthetic function and regulation of osteoblasts: current knowledge and applications. MJM. 1995; 19951:138.
Nanci, A. Ten Cate's Oral Histology: Development, Structure and Function, 7th. Mosby; 2005.
Nanci, A., Bosshardt, D.B. Structure of periodontal tissues in health and disease. Periodontology 2000. 2006; 40:11.
Nancy, A., Whitson, S.W., Bianco, P.Nanci A., ed. Bone in Ten Cates Oral Histology Developement Structure Function, 6th, Elsevier, 2005. [pp. 111-144].
Newman, M.G., Takei, H., Klokkevold, P.R., Carranza, F.A. Carranza's Clinical Periodontology, 10th. Philadelphia: WB Saunders; 2006.
Orban, B. A contribution to the knowledge of the physiologic changes in the periodontal membrane. J Am Dent Assoc. 1929; 16:405.
Ortega, N., Ke Wang, Napoleone, F., et al. Complementary interplay between matrix metalloproteinase-9, vascular endothelial growth factor and osteoclast function drives endochondral bone formation. Dis Model Mech. 2010; 3:224.
Proff, P., Romer, P. The molecular mechanism behind bone remodeling. Clin Oral Invest. 2009; 13:355.
Raggatt, L.J., Partridge, N.C. Cellular and molecular mechanisms of bone remodeling. J Biol Chem. 2010; 285:25103.
Ritchey, B., Orban, B. The crests of the interdental alveolar septa. J Periodontol. 1953; 24:75.
Robert, L.J., Robert, S.W., Teresita, B., et al. Osteoblast programed cell death (Apoptosis): Modulation by growth factors and cytokine. Journal of bone and mineral research. 1998; 13:793.
Rodan, S.B., Rodan, G.A. Integrin function in osteoclasts Journal of Endocrinology. 1997; 154(suppl):S47–S56.
Roodman, D.C. Regulation of osteoclast differentiation. Ann N Y Acad Sci. 2006; 1068:100.
Roodman, G.D. Advances in bone biology:The Osteoclast. Endocrine reviews. 1996; 17:308.
Roux, S., Orcel, P. Bone loss-Factors that regulate osteoclast differentiation— An update. Arthritis res. 2000; 2(6):451.
Sajeda Meghji. Bone remodeling. Br Dent J. 1992; 172:235.
Seibel, M.J., Woitge, H.W. Basic principles and clinical applications of biochemical markers of bone metabolism: biochemical and technical aspects. Clin Densitom. 1999; 2(3):299.
Sicher, H., DuBrul, E.L. Oral anatomy, 6th. St Louis: The CV Mosby Co; 1975.
Sodek, J., McKee, D.M. Molecular and cellular biology of alveolar bone. Periodontology. 2000; vol 24:99.
Sonoyama, W., Seo, B.M., Human, S.S. Hertwig's epithelial root sheath cells play crucial roles in cementum formation. J Dent Res. 2007; 86(7):594.
Standring, S. Gray's Anatomy, 40th. Edinburgh: Churchill Livingstone; 2009.
Standring, S. GRAY'S anatomy, the anatomical basis of clinical practice, 39th. Elsevier Churchill Livingstone; 2005.
Strewle, G.J. Local and systemic control of the osteoblast. J Clin Invest 107. 2001; 3:271.
Susan, X., Hsiong, Mooney, D.J. Regeneration of vascularised bone. Periodontology 2000. 2006; 41:109.
Szulc, P., Seeman, E., Delmas, P.D. Biochemical measurements of bone turnover in children and adolescents. Osteoporos Int. 2000; 11(4):281.
Tamara, A., Franz-Odendaal, Hall, B.K., et al. Buried alive: how osteoblasts become osteocytes. Developmental Dynamics. 2006; 235:176.
Suda, Tatsuo, Nakamura, Ichiro, et al. Regulation of osteoclast function. Journal of bone and mineral research. 1997; 12:869.
Ueland, T. GH/IGF-1 and bone resorption in vivo and in vitro. European Journal of endocrinology. 2005; 52(3):327.
Urist, M.R., Biochemistry of calcification. 2. Bourne, G.H., eds. The biochemistry and physiology of bone; 4. Academic Press, Inc, New York, 1976.
Vaananen, H.K., Zhao, H., Mulari, M., Halleen, J.M. The cell biology of osteoclast function. Journal of Cell Science. 2000; 113(3):377.
Weinmann, J.P. Bone changes related to eruption of the teeth. Angle Orthod. 1941; 11:83.
Weinmann, J.P., Sicher, H. Bone and bones; fundamentals of bone biology, 2. St Louis: The CV Mosby Co; 1955.
Yunshan Hu, et al. Isolation of a human homolog of osteoclast inhibitory lectin that inhibits the formation and function of osteoclasts. Journal of bone and mineral research. 2004; 219:89.
Zimmermann, B., Wachtel, H.C., Noppe, C. Patterns of mineralization in vivo. Cell Tissue Res. 1991; 263:483.