Prostaglandins and Related Compounds
Prostaglandins and their related compounds—prostacyclins (PGI), thromboxanes (TXA), leukotrienes (LT) and lipoxins are collectively known as eicosaniods, since they all contain 20 carbons (Greek : eikosi-twenty). Eicosanoids are considered as locally acting hormones with a wide range of biochemical functions.
History
Prostaglandins (PGs) were first discovered in human semen by Ulf von Euler (of Sweden) in 1930. These compounds were found to stimulate uterine contraction and reduce blood pressure. von Euler presumed that they were synthesized by prostate gland and hence named them as prostaglandins. It was later realized that PGs and other eicosanoids are synthesized in almost all the tissues (exception–erythrocytes). By then, however, the name prostaglandins was accepted worldwide, and hence continued.
The prostaglandins E and F were first isolated from the biological fluids. They were so named due to their solubility in ether (PGE) and phosphate buffer (PGF, F for fosfat, in Swedish). All other prostaglandins discovered later were denoted by a letter—PGA, PGH etc.
Structure of prostaglandins
Prostaglandins are derivatives of a hypothetical 20-carbon fatty acid namely prostanoic acid, hence known as prostanoids. This has a cyclopentane ring (formed by carbon atoms 8 to 12) and two side chains, with carboxyl group on one side. Prostaglandins differ in their structure due to substituent group and double bond on cyclopentane ring. The different prostaglandins are given in Fig.32.1.
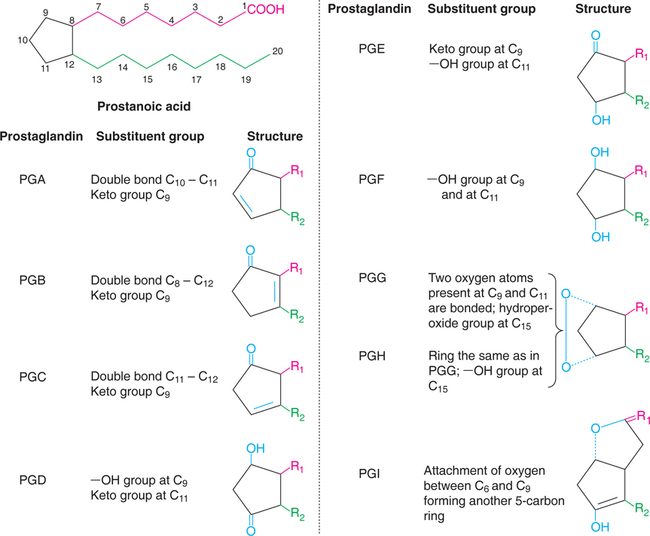
The structures of the most important prostaglandins (PGF2 and PGF2α), prostacyclins (PGI2), thromboxanes (TXA2) and leukotrienes (LTA4) along with arachidonic acid are depicted in Fig.32.2. A subscript numeral indicates the number of double bonds in the two side chains. A subscript α-denotes that the hydroxyl group at C9 of the ring and the carboxyl group are on the same side of the ring.
Synthesis of prostaglandins
Arachidonic acid (5,8,11,14-eicosatetraenoic acid) is the precursor for most of the prostaglandins in humans. The biosynthesis of PGs was described by scene Bergstrom and Bengt Samuelsson (1960). It occurs in the endoplasmic reticulum in the following stages, as depicted in Fig.32.3.
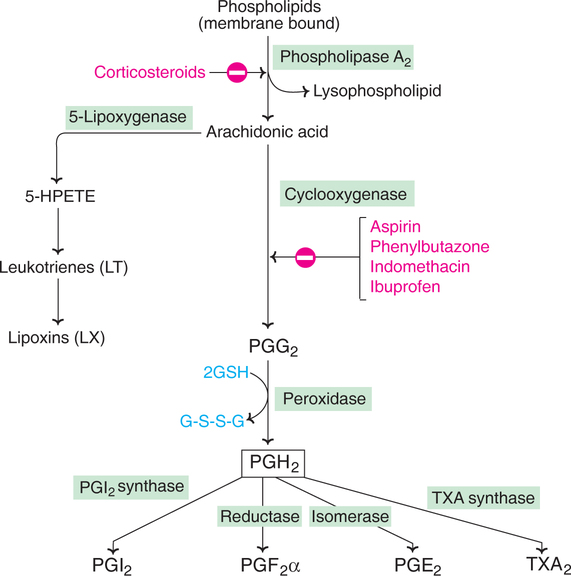
1. Release of arachidonic acid from membrane bound phospholipids by phospholipase A2—this reaction occurs due to a specific stimuli by hormones such as epinephrine or bradykinin.
2. Oxidation and cyclization of arachidonic acid to PGG2 which is then converted to PGH2 by a reduced glutathione dependent peroxidase.
3. PGH2 serves as the immediate precursor for the synthesis of a number of prostaglandins, including prostacyclins and thromboxanes.
The above pathway is known as cyclic pathway of arachidonic acid. In the linear pathway of arachidonic acid, leukotrienes and lipoxins are synthesized (details given later).
Inhibition of PG synthesis
A number of structurally unrelated compounds can inhibit prostaglandin synthesis. Corticosteroids (e.g. cortisol) prevent the formation of arachidonic acid by inhibiting the enzyme phospholipase A2.
Many non-steroidal anti-inflammatory drugs inhibit the synthesis of prostaglandins, prostacyclins and thromboxanes. They do so by blocking the enzyme cyclooxygenase.
Aspirin inhibits PG synthesis
Aspirin (acetyl salicylic acid) has been used since nineteenth century as an antipyretic (fever-reducing) and analgesic (pain relieving). The mechanism of action of aspirin however, was not known for a long period. It was only in 1971, John Vane discovered that aspirin inhibits the synthesis of PG from arachidonic acid. Aspirin acetylates serine at the active site of cyclooxygenase and irreversibly inhibits. Other antiinflammatory drugs, such as indomethacin and phenylbutazone act as reversible inhibitors of the enzyme cyclooxygenase. Paracetamol is also a reversible inhibitor.
Biochemical actions of prostaglandins
Prostaglandins act as local hormones in their function. They, however, differ from the true hormones in many ways. Prostaglandins are produced in almost all the tissues in contrast to hormonal synthesis which occurs in specialized glands. PGs are not stored and they are degraded to inactive products at the site of their production. Further, PGs are produced in very small amounts and have low half-lives.
Prostaglandins are involved in a variety of biological functions. The actions of PGs differ in different tissues. Sometimes, PGs bring about opposing actions in the same tissue.
Overproduction of PGs results in many symptoms which include pain, fever, nausea, vomiting and inflammation.
Prostaglandins mediate the regulation of blood pressure, inflammatory response, blood clotting, reproductive functions, response to pain, fever etc.
1. Regulation of blood pressure : The prostaglandins (PGE, PGA and PGI2) are vasodilator in function. This results in increased blood flow and decreased peripheral resistance to lower the blood pressure. PGs serve as agents in the treatment of hypertension.
2. Inflammation : The prostaglandins PGE1 and PGE2 induce the symptoms of inflammation (redness, swelling, edema etc.) due to arteriolar vasodilation. This led to the belief that PGs are natural mediators of inflammatory reactions of rheumatoid arthritis (involving joints), psoriasis (skin), conjunctivitis (eyes) etc. Corticosteroids are frequently used to treat these inflammatory reactions, since they inhibit prostaglandin synthesis.
3. Reproduction : Prostaglandins have widespread applications in the field of reproduction. PGE2 and PGF2 are used for the medical termination of pregnancy and induction of labor. Prostaglandins are administered to cattle to induce estrus and achieve better rate of fertilization.
4. Pain and fever : It is believed that pyrogens (fever producing agents) promote prostaglandin biosynthesis leading to the formation of PGE2 in the hypothalamus, the site of regulation of body temperature. PGE2 along with histamine and bradykinin cause pain. Migraine is also due to PGE2. Aspirin and other non-steroidal drugs inhibit PG synthesis and thus control fever and relieve pain.
5. Regulation of gastric secretion : In general, prostaglandins (PGE) inhibit gastric secretion. PGs are used for the treatment of gastric ulcers. However, PGs stimulate pancreatic secretion and increase the motility of intestine which often causes diarrhea.
6. Influence on immune system : Macrophages secrete PGE which decreases the immunological functions of B-and T-lymphocytes.
7. Effects on respiratory function : PGE is a bronchodilator whereas PGF acts as a constrictor of bronchial smooth muscles. Thus, PGE and PGF oppose the actions of each other in the lungs. PGE1 and PGE2 are used in the treatment of asthma.
8. Influence on renal functions : PGE increases glomerular filtration rate (GFR) and promotes urine output. Excretion of Na+ and K+ is also increased by PGE.
9. Effects on metabolism : Prostaglandins influence certain metabolic reactions, probably through the mediation of cAMP. PGE decreases lipolysis, increases glycogen formation and promotes calcium mobilization from the bone.
10. Platelet aggregation and thrombosis : The prostaglandins, namely prostacyclins (PGI2), inhibit platelet aggregation. On the other hand, thromboxanes (TXA2) and prostaglandin E2 promote platelet aggregation and blood clotting that might lead to thrombosis. PGI2, produced by endothelial cells lining the blood vessels, prevents the adherence of platelets to the blood vessels. TXA2 is released by the platelets and is responsible for their spontaneous aggregation. Thus, prostacyclins and thromboxanes are antagonists in their action. In the overall effect PGI2 acts as a vasodilator, while TXA2 is a vasoconstrictor. The balance between PGI2 and TXA2 is important in the regulation of hemostasis and thrombosis.
The mechanism of action of prostaglandins is not known for certain. It is believed that PGs may act through the mediation of cyclic nucleotides. PGE increases cAMP levels whereas PGF elevates cGMP.
Biomedical applications of PGs
Prostaglandins perform diversified functions. And for this reason, PGs (or other derivatives) are the most exploited in therapeutic applications. They are used in the treatment of gastric ulcers, hypertension, thrombosis, asthma etc. Prostaglandins are also employed in the medical termination of pregnancy, prevention of conception, induction of labor etc.
Inhibitors of prostaglandin synthesis (e.g. aspirin, ibuprofen) are utilized in controlling fever, pain, migraine, inflammation etc.
Leukotrienes
Leukotrienes are synthesized by leucocytes, mast cells, lung, heart, spleen etc., by lipoxygenase pathway of arachidonic acid. The synthesis of different leukotrienes (A4, B4, C4, D4 and E4) through the intermediate, 5–hydroperoxyeicosatetraenoic acid (5–HPETE) is depicted in Fig.32.4.
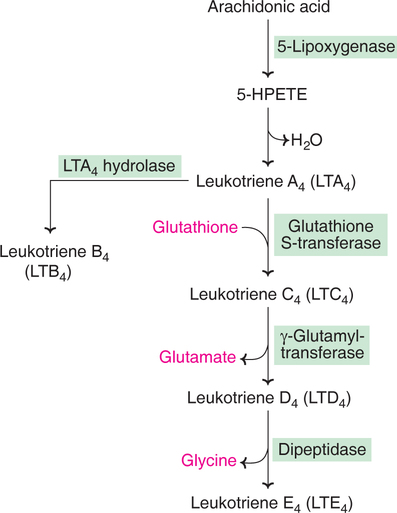
Anaphylaxis is a violent and fatal allergic reaction. It is now known that leukotrienes (C4, D4 and E4) are the components of slow-reacting substances of anaphylaxis (SRS-A), released after immunological challenge. SRS–A is 100–1,000 times more potent than histamine or prostaglandins in its action as a stimulant of allergic reactions. Leukotrienes are implicated in asthma, inflammatory reactions, hypersensitivity (allergy) and heart attacks.
Leukotrienes cause contraction of smooth muscles, bronchoconstriction, vasoconstriction, adhesion of white blood cells and release of lysosomal enzymes.
Lipoxins are involved in vasoactive, and immunoregulatory functions. There is a strong evidence to support that lipoxins act as counterregulatory compounds of immune response.
Dietary marine lipids in relation to PGs, LTs and heart diseases
Eskimos of Greenland have a low incidence of coronary heart diseases, despite the fact that they consume high quantities of fat and cholesterol. This is due to the high intake of marine lipids containing unsaturated fatty acids (UFA). The most predominant UFA in the fish foods consumed by Eskimos is 5, 8, 11, 14, 17-eicosapentaenoic acid (EPA). EPA is the precursor for leukotrienes-5 series which are much lower in their activity than the leukotriene-4 series, produced from arachidonic acid. Further, eicosapentaenoic acid inhibits the formation thromboxanes (TXA2). As already described, TXA2 promotes platelet aggregation and thrombosis.
The diet rich in marine lipids (with EPA) decreases plasma cholesterol and triacylglycerols. These factors, along with reduced synthesis of TXA2 are believed to be responsible for the low incidence of heart attacks in Eskimos.
Summary 
1. Prostaglandins (PGs) and related compounds prostacyclins (PGI), thromboxanes (TXA) and leukotrienes (LT) are collectively known as eicosanoids. They are the derivatives of a hypothetical 20 carbon fatty acid, namely prostanoic acid. Prostaglandins are synthesized from arachidonic acid, released from the membrane bound phospholipids. Corticosteroids and aspirin inhibit PG synthesis.
2. Prostaglandins act as local hormones and are involved in a wide range of biochemical functions. In general, PGs are involved in the lowering of blood pressure, induction of inflammation, medical termination of pregnancy, induction of labor, inhibition of gastric HCI secretion, decrease in immunological response and increase in glomerular filtration rate. Thromboxanes (TXA2) and prostaglandin E1 promote while prostacyclins (PGI2) inhibit platelet aggregation.
 The prostaglandins speak :
The prostaglandins speak :

