28
Surgical Incisions, Implants, and Wound Closure
After studying this chapter the learner will be able to:
• Describe the anatomy of the skin and underlying tissues.
• Discuss the rationale for placement of the surgical incision.
• Identify several surgical incisions and their associated surgical procedure.
• Identify several absorbable sutures and their application.
• Identify several nonabsorbable sutures and their application.
• List several different types of needles and their application.
Key Terms and Definitions
Allogeneic graft Tissue taken from the same species for implantation into a different individual of the same species.
Approximation Bringing the edges of an incision together.
Autologous graft Tissue taken from one area of a person’s body for implantation into another area of the same person’s body.
Evert edges Turning the edges of the skin outward with pick-ups for approximation.
Figure of 8 Suturing method used to provide hemostasis of a large vessel.
Free tie Strand of suture without a needle.
Fusiform (Elliptical) Oval-shaped incision that follows the natural lines of tension in the integument.
Implantation Process of placing material or tissue into a surgical site.
Ligature Strand of suture material used to tie or bind. Suture ligature is a free tie. Tie on a passer is a tie held in the tip of a clamp.
Prosthesis Artificial part worn on the outside or implanted inside the body.
Pursestring Circular suturing method for closure of a round opening.
Stick tie Suture on a needle in a needle holder.
Suture Strand of material used for sewing tissue together or ligating a structure.
Undermine Method of releasing the skin edges by sharp dissection to approximate skin. Care is taken not to interrupt circulation to the tissue.
Xenograft Tissue taken from one species for implantation into another species.
 Evolve Website
Evolve Website
• Historical Perspective
• Student Interactive Questions
• Glossary
The Surgical Incision
Anatomy and Physiology of the Integument
Tissue structure and function vary according to location in the body. Basic tissue types are described in Table 28-1.
The skin contributes to the health and well being of the patient. Intact skin is an effective barrier to most harmful elements. Wounded, nonintact skin is an open avenue for microbial entry. Wounds occur intentionally or unintentionally. When treated properly, most wounds heal without incident. Unfavorable outcomes occur when wound healing is disrupted by poor circulation, infection, or immune dysfunction.
Skin is a multifunction body cover, and skin assessment is an important measurement of generalized wellness. Skin color, texture, and condition can be the best predictors of how well a surgical site will heal. The most intricate procedure can be done in deep tissue layers, but the superficial layers are what the patient sees and measures the outcome against. Durability and viability of the skin of the perioperative patient are influenced by many factors. Within reasonable limits, in the absence of hemorrhage and sepsis, wound healing is predictable. Disregard for the principles of tissue handling and wound management can lead to complications. The intent of this chapter is to provide an overview of body tissues, surgical incisions, and surgical site closure.
The skin is the largest and heaviest organ of the body. The two main layers that compose the integument are the epidermis and the dermis. The thickness of the skin and its layers is determined by its location. The combined thickness of the epidermis and dermis ranges from 4 mm on the back to 1.5 mm on the scalp. Figure 28-1 shows a cross-section of the integument (skin) and its layers. Areas involving bone will incorporate vascularized periosteum over the bone.
TABLE 28-1
Four Basic Histologic Tissue Types


From Fortunato NM, McCullough SM: Plastic and reconstructive surgery, St. Louis, 1998, Mosby.
There are two basic skin types: glabrous and hairy. Glabrous, smooth skin is very thick and is found on the palms and soles. The surface is marked by ridges and sulci arranged in unique configurations referred to as dermatographics, or fingerprints. These ridges first appear in the fingertips during the thirteenth week of fetal life. A marked absence of hair follicles and oil glands is characteristic of this tissue. Hairy, thin skin has hair follicles, sweat glands, and oil glands.
Langer’s Lines
Natural lines of tension are formed by the relationship of the skin to the underlying musculature (Fig. 28-2). Austrian anatomist Karl Langer (1819-1887) described how incisions could be more cosmetic if natural cleavage lines were followed when planning the surgical incision. The collagen fibers in the epidermis and dermis form an elliptical shape when the skin is incised in fusiform fashion along the natural lines. The angle of the incision should be no more than 30 degrees at each margin. The surgeon may undermine the tissue to minimize the distance between the skin edges. Closure is better when the edges meet during approximation. As the incision heals, tension of the skin is relaxed, causing minimal pulling and widening of the bridge of scar tissue.
Epidermis
The epidermis is the outermost layer of skin. It is organized into five levels of stratified squamous epithelium and contains no organs, glands, nerve endings, or blood vessels. This layer renews itself every 15 to 30 days, depending on the body surface area, the age of the individual, and the individual’s generalized condition. The basic anatomy and physiology of epidermal layers are as follows:
• Stratum corneum: Keratinized cells make up 75% of the epidermal thickness. Cells are shed from this level, which is referred to as the horny layer. It is thinner in hairy, thin-skinned areas.

• FIG. 28-2 Langer’s lines. A, Anterior view. B, Posterior view. C, Forearm. D, Relaxed skin tension lines of face. (From Fortunato NM, McCullough SM: Plastic and reconstructive surgery, St Louis, 1998, Mosby.)
• Stratum granulosum: This level is arranged in three to five layers. Mitotic activity creates cells for renewal of epidermal layers.
• Stratum spinosum: This layer creates cells for renewal of epidermal layers.
• Stratum basale: A single cell layer that lies between the junction of the epidermis and the dermis. Intense mitosis in this layer in combination with the basal layer and the spinosum causes epidermal regeneration. As cells are generated they migrate upward, toward the surface. Melanocytes, located between basal cells and in hair follicles, create melanin, which causes skin pigmentation. Melanin enters and accumulates in keratocytes, causing superficial skin tone and providing ultraviolet protection. Exposure to sunlight causes darkening of existing melanin and accelerated generation of new melanin.
Each epithelial layer consists of keratin-producing cells (keratinocytes). Keratin is modified into functional components such as hair and fingernails on select body surfaces. Overactivity of the spinosum and basal levels can increase epidermal thickness in normally thin areas, causing psoriasis.
Dermis
The dermis is composed of papillary and reticular layers of flexible connective tissue. Superficially, the dermis has an irregular surface of papilla-like fingers that project into the strata basale of the epidermal layers. The dermis, regardless of location, is a loose, areolar connective tissue that contains pain and touch receptors, glands, blood vessels, and lymphatics. It is the key layer in wound repair and tissue healing.
Glandular Structures and Ducts in the Integument
Oil Glands (Sebaceous Glands)
Sebaceous and sudoriferous glands are found within the dermal layers of the skin. Sebaceous glands are referred to as holocrine glands because the oily secretion sebum also carries cellular debris. The sebaceous duct empties into a hair follicle or, on nonhairy areas, directly onto the surface of the skin. Sebum is a lubricant with minor antibacterial and antifungal properties. The palms and soles lack sebaceous glands, but these glands are numerous on the scalp and face and around natural body orifices. Oily skin types sometimes have increased scar formation because the oil forms a mechanical barrier to healing.
Sweat Glands (Sudoriferous Glands)
Sudoriferous glands are found on every area of the body except the lips, nipples, and glans penis. Eccrine and apocrine glands are two types of sudoriferous glands. Eccrine glands are widely distributed over the entire surface of the body and are responsible for producing 700 to 900 g of sweat per 24 hours. These simple structures are embedded in the dermis and have a funnel-shaped exit path (pore) leading directly to the skin’s surface. Secretions are produced in response to physical activity and cool the body by evaporation. These glands are most numerous on the palms, soles, and forehead.

• FIG. 28-3 Abdominal tissue layer at the arcuate line. A, Above the arcuate line. B, Below the arcuate line.
Apocrine glands are located only in the axillary, perineal, and areolar areas in combination with eccrine glands. Apocrine glands are larger than eccrine glands and are embedded in subcutaneous tissue. These glands secrete a viscous fluid and have ducts that open into hair follicles. Initially the secretion is odorless, but it quickly develops an odor caused by bacterial decomposition. These glands secrete in response to stress or excitement. In animals, apocrine and sebaceous glands are thought to release pheromones, which are hormones thought to cause sexual attraction. Modified apocrine glands are found in the ear canals and secrete cerumen (earwax).
Blood Supply and Innervation
The dermis contains a rich supply of blood and lymph. In some areas, arterial and venous communication is by direct shunting, without using a capillary mechanism. Arteriovenous shunts allow for thermoregulation and blood pressure control. Capillary networks are located in the papillary layer to nourish the epidermis. Effector innervation of the dermis is derived from postganglionic fibers of the sympathetic ganglia. Affector innervation is a superficial dermal network of free nerve endings, hair follicles, and encapsulated sensory organs.
Tissue Layers Under the Integument
Subcutaneous Adipose Layer
Underneath the dermis is a loose, fatty layer that is referred to as the subcutaneous layer. Beneath the subcutaneous tissue is a layer of striated muscle. The looseness of this structure allows for movement of the skin over supporting musculature. In males the distribution is through the nape of the neck, deltoids, triceps, abdomen, lumbosacral region, and buttocks. In females the fatty layer extends through the breasts, abdomen, buttocks, epitrochanteric area, and anterior thighs.
Fascia
The fascia is a fibrous areolar tissue that supports the superficial skin layers and encases the muscle. Fascia throughout the body covers the muscles anteriorly and posteriorly. The superficial fascia is located directly underneath the integument and is the point to which injection of a local anesthetic agent should extend for the best effect. Sensory nerve fibers run through this area, and an anesthetic agent is easily absorbed. Adipose cells occupy areolar spaces, rendering the fascia soft and pliable, and permitting vessels, nerves, and lymphatics to pass through the layers.
The deep fascia is tough and less pliable. It runs the length of the muscle bundle and terminates in fibrous tendons that attach to bones beneath the periosteum. The anterior fascia of the abdomen is arranged in three layers that merge around the rectus abdominis muscles. The internal and external oblique muscles cover and surround the rectus muscles to the level of the linea circularis, or the landmark known as the arcuate line, sometimes called the semicircular line of Douglas (Fig. 28-3). The arcuate line is formed by three fascial merges one third the distance between the umbilicus and the pubis. Below the level of the arcuate line the fascial layers are fully anterior with no posterior fascial component. The rectus abdominis is behind the layered fascia.
Peritoneum
Lying beneath the posterior fascia of the abdomen is the peritoneum—the thin, two-layer serous membrane that lines the interior of the abdominal cavity (parietal peritoneum) and surrounds the organs (visceral peritoneum). The inferior aspect of the peritoneum overlies or is superior to the dome of the urinary bladder. In other words, the bladder is infraperitoneal, or below the peritoneal cavity. The stomach is superior to the peritoneal sac with the fundus situated supraperitoneally. The kidneys and the aorta are behind the peritoneum (retroperitoneal).
Accessory Appendages to the Integument
Modifications in the epidermal layer cause varied degrees of keratin deposition. Thickness and durability are functionally related to the location of keratinization. Hair and fingernails are modified keratin.
Other skin appendages include glands, blood vessels, and sensory organs. Glands arise in the dermis, and some exit the body through ducts that penetrate the epidermis. Other glands empty into the superior segment of hair follicles.
Hair Follicles
Hair follicles are keratinized epidermal epithelium that terminates in the dermal layers. The follicle is nourished by a capillary bed. Loss of this blood supply results in death of the follicle. Small bundles of smooth muscle cells, referred to as arrector pili, form attachments to the surrounding connective tissue in a diagonal fashion. As arrector pili contract, the shaft of the hair is straightened to an upright position. This contraction causes the superficial skin to dimple and pucker, creating “goose bumps.”
Nails (Ungues)
The dorsal tip of each phalanx is tipped with a plate of specialized keratinized cells. Proximally, the nail root is covered by stratum corneum, which is referred to as the eponychium, or cuticle. The nail plate rests on a bed of epidermis (nailbed). Nails are chemically similar to the surface epidermis. Peripheral blood supply may be assessed through the translucent nail plate but should not be the sole determinant of oxygenation and well-being of the patient.
Surgical Landmarks
The primary reference point for abdominal incisions is the umbilicus. Secondary surface landmarks include the xyphoid, the pubis, and the iliac crests. Incisions may be vertical, horizontal, or oblique and may occur in various areas of the torso (Fig. 28-4). Specialty incisions, such as those involving the head, limbs, breast, reconstructive procedures, and other organ systems, are described in their respective chapters.
The direction of the incisional line is determined by the anatomic plane in the body (Fig. 28-5). Some specialty instruments are constructed to dissect, debulk, or separate layers in a specific pattern according to the angle of the natural tissue arrangement. One example is a sagittal saw that is designed to cut bone along the sagittal plane.
Placement of the Surgical Incision
Before the procedure begins the surgeon chooses the most suitable incision for the procedure being performed. All incisions incorporate, with varying degrees of success, certain characteristics that include:

• FIG. 28-6 Dissecting tissue layers of the abdomen, A, Subcutaneous fat (yellow). B, Anterior fascia (white). C, Muscle (red). D, Skin through muscle dissected. This white peritoneum is shown for dissection. E, Open peritoneum with viscera beneath.
• Condition of the patient
• Knowledge of previous surgery in the same region and the presence of adhesions
• Ease and speed of entry into tissues
• Size of the body habitus and the natural lines of tissue tension (Langer’s lines)
• Maximum exposure of intended surgical site and adjacent structures (i.e., muscles, nerves, vessels, lymphatics)
• Ability to extend the incision if necessary
• Minimum trauma and scar formation
• Least postoperative discomfort
• Maximum postoperative wound strength
Abdominal Surgery
A laparotomy involves surgically opening the abdominal wall and entering the peritoneal cavity (Fig. 28-6). The skin and subcutaneous tissue are incised and the blood vessels are ligated or electrocoagulated. Both the posterior fascia and the peritoneum may be cut at the same time, thus exposing the contents of the abdominal cavity. Various types of incisions are used in a laparotomy, but each follows essentially the same technique.
Types of Abdominal Incisions
The incisions discussed in the following sections are applicable to open abdominal or pelvic procedures for specific organs or organ systems. The usual anterior surface incisions are depicted in Figure 28-7.
Laparoscopy is performed through either one puncture or multiple (usually one to five) incisions that are smaller (usually 5-10 mm), separate, and distinct. Endoscopy and laparoscopy are discussed in detail in Chapter 32.
Midline Incision
A midline incision can be upper abdominal, lower abdominal, or a combination of both going around the umbilicus. The patient may have thicker deposits of adipose under the planned incision line. Depending on the intended procedure, a full midline incision can begin in the epigastrium at the level of the xyphoid process and may extend inferiorly to the suprapubic region. The surgeon is careful to avoid cutting bowel that may be close to the surface in the peritoneal cavity. Most surgeons curve the incision around the periphery of the umbilicus to avoid cutting through the structure.
An upper midline incision above the umbilicus offers excellent exposure of and rapid entry into the epigastric region. The upper midline incision is made carefully to accommodate the position of the falciform ligament of the liver. A lower midline incision can begin inferior to the umbilicus and extend to the pubis for lower bowel, gynecologic, or obstetric pelvic procedures. Care is taken at the lower margin to avoid perforating the urinary bladder. An indwelling urinary catheter can help keep the bladder decompressed.
The midline incision enters the body through fascial planes that are relatively avascular. Many vessels and nerve endings are spared. The muscles are easily separated and retracted for visualization and when the procedure is complete it is easy to close. If the incision must be lengthened, the extension is easier to make. When closing, the surgeon may request a visceral retainer to protect the underlying organs and prevent an inadvertent suture from passing through the bowel.
Paramedian Incision
The paramedian incision is a vertical incision made approximately 4 cm (approximately 2 inches) lateral to the midline on either side in the upper or lower abdomen. After the skin and subcutaneous tissue are incised, the rectus sheath is split vertically and the muscle is retracted laterally. This incision allows quick entry into and excellent exposure of the abdominal cavity. It limits trauma, avoids nerve injury, is easily extended, and gives a firm closure. Examples of use include access to the biliary tract or pancreas in the right upper quadrant and access to the left lower quadrant for resection of the sigmoid colon.
Subcostal Upper Quadrant Oblique Incision
A right or left oblique incision begins in the epigastrium and extends laterally and obliquely just below the lower costal margin. One example is the Kocher incision in the right subcostal region, which was developed by Swiss surgeon and Nobel laureate Emil Theodor Kocher (1841-1917). It continues through the rectus muscle, which is either retracted or transversely divided. Although this type of incision affords limited exposure except for upper abdominal viscera, it provides good cosmetic results because it follows skin lines and produces limited nerve damage. Despite being painful, it is a strong incision postoperatively. Examples of use include biliary procedures.
Bilateral subcostal incisions that join in the midline may be preferred for procedures that involve the stomach and/or pancreas. A modified bilateral subcostal incision (chevron incision or rooftop incision) is made for increased visibility during a liver transplantation or resection. The chevron incision can be extended superiorly toward the xyphoid to create the Mercedes incision for greater access to the inferior aspect of the diaphragm.
McBurney’s Incision
Charles McBurney (1845-1913) described a method of diagnosing appendicitis in 1889 by pressing on the right lower quadrant, just below the umbilicus and 4 cm (2 inches) medial from the anterior superior iliac spine. This area of the abdomen is referred to as McBurney’s point.
McBurney’s incision involves a muscle-splitting incision that extends through the fibers of the external oblique muscle. The incision is deepened, the internal oblique and transversalis muscles are split and retracted, and the peritoneum is entered. This is a fast and easy incision, but exposure is limited. Its primary use is for appendectomy. Some surgeons modify this incision in a transverse plane referred to as the Rockey-Davis or Lanz incision to conceal the scar in a natural skinfold.
Thoracoabdominal Incision
For a thoracoabdominal incision the patient is placed in a lateral position. Either a right or a left incision begins at a point midway between the xyphoid process and umbilicus and extends across the abdomen to the seventh or eighth costal interspace and along the interspace into the thorax. The rectus, oblique, serratus, and intercostal muscles are divided in the line of incision down to the peritoneum and pleura. This converts the pleural and peritoneal cavities into one main cavity, thus allowing excellent exposure for the upper end of the stomach and lower end of the esophagus. Examples of use include esophageal varices and the repair of a hiatal hernia.
Midabdominal Transverse Incision
The midabdominal transverse incision starts on either the right or left side and slightly above or below the umbilicus. It may be carried laterally to the lumbar region between the ribs and crest of the ilium. The intercostal nerves are protected by cutting the posterior rectus sheath and peritoneum in the direction of the divided muscle fibers.
Transverse abdominal incisions are sometimes used for infants because the abdomen is wider than it is long. Better exposure to the intraabdominal cavity is attained. In some cases the same is true for extremely short, stout adults with large abdominal girth.
The advantages are rapid incision, easy extension, a provision for retroperitoneal approach, and a secure postoperative wound. Examples of use include choledochojejunostomy and transverse colostomy.
Pfannenstiel Incision
German gynecologist Herman Johannes Pfannenstiel (1862-1909) developed the Pfannenstiel incision, a curvilinear transverse incision across the lower abdomen and within or superior to the hairline of the pubis. The incision follows the Langer’s lines of the natural skinfolds. The rectus fascia is incised transversely below the arcuate line, and the muscles are separated. The peritoneum is incised vertically in the midline. This lower curved incision provides good exposure and strong closure for pelvic procedures. Its primary use is for urologic and gynecologic procedures and cesarean section. One disadvantage is that exposure may be limited. The scar is obscured by the patient’s pubic hair.
One modification is the Maylard transverse incision. This incision is not curved but straight. It is made above the level of the curvilinear Pfannenstiel incision for greater access to the pelvic and urologic organs.
Inguinal Incision (Lower Oblique)
An oblique incision of the right or left inguinal region extends from the pubic tubercle to the anterior iliac crest, slightly above and parallel to the inguinal crease. Incision of the external oblique fascia provides access to the cremaster muscle, inguinal canal, and spermatic cord structures. Great care is taken not to entrap ilioinguinal nerves or the spermatic cord. Its primary use is for inguinal herniorrhaphy.
Wound Closure
Closure of a surgical site or other wound is performed after necessary hemostasis has been achieved. Wounds include deep and superficial structures. Methods of wound closure include sutures, staples, clips, tapes, and glues.
Suture Basics
The noun form of suture is used for any strand of material used for ligating or approximating tissue; it is also synonymous with stitch. The verb to suture denotes the act of sewing by bringing tissues together and holding them in approximation until healing has taken place.
If the material is tied around a blood vessel to occlude the lumen, it is called a ligature or tie. A suture attached to a needle for a single stitch for hemostasis is referred to as a stick tie or suture ligature. A free tie is a single strand of material handed to the surgeon or first assistant to ligate a vessel. A tie handed to the surgeon in the tip of a forceps or clamp is referred to as a tie on a passer.
Suturing Techniques
Halsted Suture Technique
The education a physician receives during postgraduate surgical training exerts a lasting influence on his or her surgical techniques. The classic example of the influence of a professor on his students is that of Dr. William Stewart Halsted (1852-1922).
Halsted, a professor of surgery at Johns Hopkins Hospital in Baltimore from 1893 to 1922, perfected and brought into use the fine-pointed hemostat for occluding vessels, the Penrose drain, and rubber gloves. He is best known for his principles of gentle tissue handling. The silk suture technique he initiated in 1883, or a modification of it, is in use today. Its features are as follows:
1. Interrupted individual sutures are used for greater strength along the wound. Each stitch is taken and tied separately in a figure of 8 pattern for deeper tissues. If one knot slips, all the others hold. Halsted also believed that interrupted sutures were a barrier to infection, for he thought that if one area of a wound became infected, the microorganisms traveled along a continuous suture to infect the entire wound.
2. Sutures are as fine as is consistent with security. A suture stronger than the tissue it holds is not necessary.
3. Sutures are cut close to the knots. Long ends cause irritation and increase inflammation. Only external stitches have tails for ease of removal after healing.
4. A separate needle is used for each skin stitch.
5. Dead space in the wound is eliminated. Dead space is that space caused by separation of wound edges that have not been closely approximated by sutures. Serum or blood clots may collect in a dead space and prevent healing by keeping the cut edges of tissue separated.
6. Two fine sutures are used in situations usually requiring one large one.
7. Silk is not used in the presence of infection. The interstices (braid pattern) can harbor microorganisms.
8. Tension is not placed on tissue. Approximation versus strangulation preserves the blood supply.
Halsted’s principles were based on use of the only suture materials available to him: silk and surgical gut. With the advent of less reactive synthetic materials, wound closure may be safely and more quickly performed with different techniques without complications.
Principles of Suturing
The strength of the wound is related to the condition of the tissue and the number of stitches in the edges. Care is taken not to place more sutures than necessary to approximate the edges. The amount of tissue incorporated into each stitch directly influences the rate of healing.1 The adequacy of the blood supply to the tissue needs to be preserved for healing to take place.
Methods of Suturing
The edges of the wound are intentionally directed by the placement of sutures during closure. Suturing techniques are depicted in Figure 28-8. Examples of suturing techniques that direct the wound edges for specific healing mechanisms include but are not limited to:
1. Everting sutures: These interrupted (individual stitches) or continuous (running stitch) sutures are used to evert edges.
a. Simple continuous (running): This suture can be used to close multiple layers with one suture. The suture is not cut until the full length is incorporated into the tissue (see Fig. 28-8, A).
b. Continuous running/locking (blanket stitch): A single suture is passed in and out of the tissue layers and looped through the free end before the needle is passed through the tissue for another stitch. Each new stitch locks the previous stitch in place (see Fig. 28-8, B).
c. Simple interrupted: Each individual stitch is placed, tied, and cut in succession from one suture (see Fig. 28-8, C).
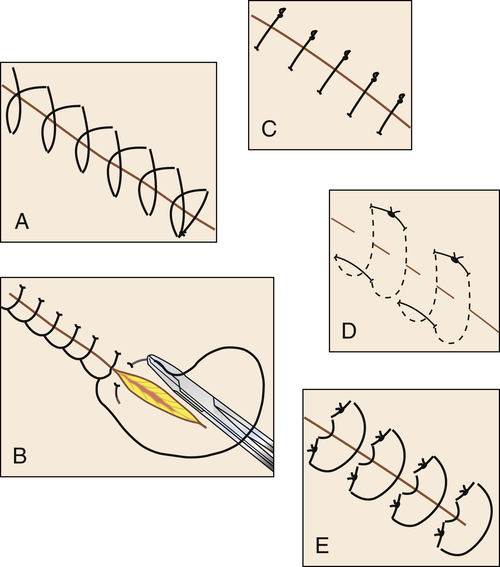
• FIG. 28-8 Examples of suturing techniques. A, Simple continuous. B, Continuous locking. C, Simple interrupted. D, Horizontal mattress. E, Vertical mattress.
d. Horizontal mattress: Stitches are placed parallel to wound edges. Each single bite takes the place of two interrupted stitches (see Fig. 28-8, D).
e. Vertical mattress: This suture uses deep and superficial bites, with each stitch crossing the wound at right angles. It works well for deep wounds. Edges approximate well (see Fig. 28-8, E).
2. Inverting sutures: These sutures are commonly used for two-layer anastomosis of hollow internal organs, such as the bowel and stomach. Placing two layers prevents passing suture through the lumen of the organ and creating a path for infection. A single layer is placed for other structures, such as the trachea, bronchus, and ureter. The edges are turned in toward the lumen to prevent serosal and mucosal adhesions. The number of layers is proportional to the quality of the blood supply. Stitches can be interrupted or continuous.
a. Halsted suture: A two-layer modification of the horizontal mattress suture used for friable tissue.
b. Connell suture: A continuous single-layer suture of gut used for hemostasis in the inner layer of bowel with a separate outer inverting layer of alternating horizontal and vertical mattress sutures.
c. Cushing suture: A continuous vertical mattress suture that unites half of the lumen, followed by a second continuous vertical mattress suture that completes the second half of the circumference.
d. Grey-Turner sutures: A series of inverted interrupted horizontal or vertical mattress stitches.
e. Purse string suture: A continuous stitch that encircles and closes a lumen while inverting the edges.
Knot Placement
Each suture placed in tissue usually requires the placement of a knot to secure the ends. Interrupted stitches require individual knots, and therefore placement of each knot can influence how well the wound heals and the cosmetic result. The following list of principles concerns knots and knot tying:
1. The knot should be tied away from:
a. Vital structures, such as the eye
b. Sources of contamination, such as the mouth
c. Potential irritants, such as the nares
d. Potential sources of increased inflammation, such as the incision line
2. The knot should be tied toward:
a. The better blood supply
b. The area that provides the best security of the knot
c. Where the mark would be less noticeable, if possible
Cutting Sutures
Care is taken to prevent excess suture from remaining in the wound. Suture tails are trimmed close to the knot inside the body, but a short tail may be left for external stitches to facilitate removal. Considerations for cutting suture include:
1. Scissors should be stabilized by the index finger on the screw (tripod stance), the blades are angled slightly and slide down to the area just above the knot, and the suture is cut with the tips of the scissors.
2. The tips of the scissors must be visible to ensure that other structures are not injured by the cutting motion.
3. A hemostat and/or a second suture should be available in the event that the knot is inadvertently cut, releasing the sutured tissue.
4. A hemostat may be placed on one of the suture ends to stabilize the suture to be cut.
5. If removing a suture, a forceps is used to grasp the suture at the knot. Cut the suture between the knot and the skin. Extract the cut suture with the forceps.
Retention Sutures
Interrupted nonabsorbable sutures are placed through tissue on each side of the primary suture line, a short distance from it, to relieve tension on it. Heavy strands are used in sizes ranging from 0 through 5. The tissue through which retention sutures are passed includes skin, subcutaneous tissue, and fascia and may include rectus muscle and peritoneum of an abdominal incision.
After abdominal surgical procedures, retention sutures are used frequently in patients in whom slow healing is expected because of malnutrition, obesity, carcinoma, or infection; in geriatric patients; in patients receiving cortisone; and in patients with respiratory problems.2
Retention sutures may be used as a precautionary measure to prevent wound disruption when postoperative stress on the primary suture line from distention, vomiting, or coughing is anticipated. Retention sutures should be removed as soon as the danger of sudden increases in intraabdominal pressure is over, usually on the fourth or fifth postoperative day. Retention sutures are also used to support wounds for healing by second intention and for secondary closure after wound disruption for healing by third intention.
Retention Bridges, Bolsters, and Bumpers
To prevent heavy retention suture from cutting into skin, several different types of bridges, bolsters, or bumpers are used:
• Bridges are plastic devices placed on the skin to span the incision. The retention suture is brought through the skin on both sides of the incision and through holes on each side of the bridge and is fastened over the bridge. One type allows adjustment of tension on the edges of the incision during the postoperative healing period.
• Bumpers are segments of plastic or rubber tubing. One end of the suture is threaded through the tubing before the suture is tied. It covers the entire retention suture strand that is on the skin surface to prevent irritation (Fig. 28-9). Compression bolsters are made from polyethylene foam held in place with malleable aluminum buttons to secure and distribute tension of retention sutures.

• FIG. 28-9 Suturing incised abdominal tissue layers. A, Peritoneum (continuous stitch, taper-point needle). B, Muscle (interrupted stitch). C, Anterior fascia (interrupted stitch, cutting needle). D, Subcutaneous (not always sutured, taper-point needle). E, Skin (interrupted stitch, cutting needle). F, Retention sutures. Note bumpers to protect skin.
• Buttons and beads are used as bolsters and bumpers to prevent the suture from retracting or cutting into skin or friable tissue. The suture is pulled through holes and tied over a sterile button (e.g., with pull-out tendon sutures). Beads may be placed on the ends of pull-out subcuticular skin sutures. The devices are used most frequently in plastic and orthopedic surgery.
Traction Suture
A traction suture may be used to retract tissue to the side or out of the way, such as the tongue in a surgical procedure in the mouth. Usually a nonabsorbable suture is placed through the part. Other materials that may be used to retract or ligate vessels include:
• Umbilical tape: Aside from its original use for tying the umbilical cord on a newborn, cotton umbilical tape may be used as a heavy tie or as a traction suture. It may be placed around a portion of bowel or a great vessel to retract it. These should be counted and accounted for at the end of the procedure. Cotton should be moistened with sterile saline before use. Umbilical tapes should not be cut into pieces.
• Vessel loop: A length of thin flat silicone can be placed around a vessel, nerve, or other tubular structure for retraction. It can be tightened around a blood vessel for temporary vascular occlusion. These should be counted and accounted for at the end of the procedure. Vessel loops should not be cut into pieces.
• Aneurysm needle: An aneurysm needle is an instrument with a blunt needle on the end for passing suture. The eye is on the distal end of the needle. The needle forms a right or oblique angle to the handle, which is one continuous unit with the needle. The needles are made in symmetric pairs, right and left. The surgeon uses them to place a ligature around a deep, large vessel, such as in a thyroidectomy or in thoracic surgery. They can be used to pass a suture tape around an incompetent cervix to perform cerclage. These should be counted and accounted for at the end of the procedure. (Refer to Fig. 34-16 in Chapter 34.)
Endoscopic Suturing
Endoscopic sutures are available as ligatures and preknotted loops or with curved or straight, permanently swaged needles for use through an endoscope. The ligatures are fashioned into loosely knotted loops before being passed through the endoscope to tie off vessels and tissue pedicles.3 After the loop is placed around the target site, the knot is slid into position and tightened. The ends are cut with endoscopic scissors and removed through the endoscope. Suture with a permanently swaged needle is placed through either a 3-mm suture introducer for a straight needle or an 8-mm suture introducer for a curved needle. Used to suture vessels, reconstruct organs, approximate opposing tissue surfaces, and anastomose tubular structures, the technique varies according to the method used for knot tying.3 The methods of endoscopic knot tying are as follows:
• Extracorporeal method: The swaged needle and both ends of the suture are brought outside the body through the trocar. The needle is cut off, and the knot is loosely fashioned. The knot is reintroduced into the body through the trocar by means of a knot-sliding cannula. It is snugged into position and tightened against the tissue. The ends of the suture are cut close to the knot with endoscopic scissors. Excess suture ends are removed through the endoscope.
• Intracorporeal method: The needle and suture are passed through the tissue with an endoscopic needle holder. Endoscopic instruments are used to tie the knot and cut the suture inside the body.
Specifications for Suture Material
• It must be sterile when placed in tissue. Sterile techniques must be rigidly followed in handling suture material. For example, if the end of a strand drops over the side of any sterile surface, discard the strand. Almost all postoperative wound infections are initiated along or adjacent to suture lines. Affinity for bacterial contamination varies with the physical characteristics of the material.
• It must be predictably uniform in tensile strength by size and material. Tensile strength is the measured pounds of tension or pull that a strand will withstand before it breaks when knotted. Minimum knot-pull strengths are specified for each basic raw material and for each size of that material by the U.S. Pharmacopeia (USP). Tensile strength decreases as the diameter of the strand decreases.
• It must be as small in diameter as is safe to use on each type of tissue. The strength of the suture usually needs to be no greater than the strength of the tissue on which it is used. Smaller sizes are less traumatic during placement in tissue and leave less suture mass to cause tissue reaction. The surgeon ties small-diameter sutures more gently and thus is less apt to strangulate tissue. A small-diameter suture is flexible, easy to manipulate, and leaves minimal scar on skin.
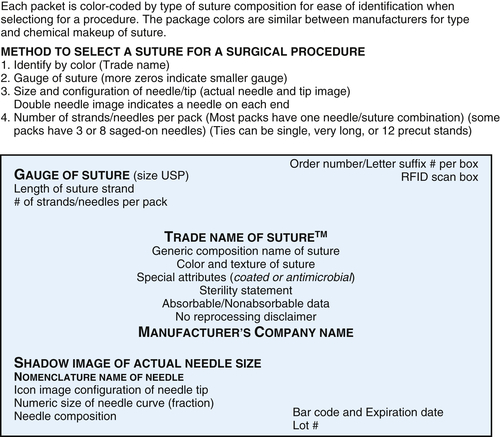
• USP-determined sizes range from heavy 10 (largest) to very fine 12-0 (smallest); ranges vary with materials. Taking size 1 as a starting point, sizes increase with each number above 1 and decrease with each 0 (zero) added. The more zeroes in the number, the smaller the size of the strand. As the number of zeroes increases, the size of the strand decreases. In addition to this system of size designation, the manufacturer’s labels on boxes and packets may include metric measures for suture diameters. These metric equivalents vary slightly by types of materials. Box 28-1 shows how suture gauge is measured on a scale in numeric descriptions from smallest to largest.
• It must have knot security, remain tied, and give support to tissue during the healing process. However, sutures in the skin are always removed 3 to 10 days postoperatively, depending on the site of incision and cosmetic result desired. Because they are exposed to the external environment, skin sutures can be a source of microbial contamination of the wound that inhibits healing by first intention.
• It must cause as little foreign body tissue reaction as possible. All suture materials are foreign bodies, but some are more inert (less reactive) than others.
Choice of Suture Material
The surgeon selects the type of suture material best suited to maintain tensile strength and promote healing. Figure 28-10 shows the labeling of a generic suture packet. When selecting a suture, specific notations about the characteristics of each suture are listed on the box and the packet. Surgical sutures are classified as either absorbable or nonabsorbable:
1. Absorbable sutures: Sterile strands prepared from collagen derived from healthy mammals or from a synthetic polymer. They are capable of being absorbed by living mammalian tissue but may be treated or coated to modify resistance to absorption. Some synthetic sutures are coated with a Triclosan antimicrobial chemical.4 They may be colored by a dye approved by the U.S. Food and Drug Administration (FDA).
2. Nonabsorbable sutures: Strands of natural or synthetic material that effectively resist enzymatic digestion or absorption in living tissue. During the healing process, suture mass becomes encapsulated and may remain for years in tissues without producing any ill effects.
Sutures may be dyed with a color additive approved by the FDA. They may be modified with respect to body, texture, or capillarity. Capillarity refers to a characteristic of nonabsorbable sutures that allows the passage of tissue fluids along the strand, permitting infection, if present, to be drawn along the suture line. Suture materials may be untreated or may be treated to reduce capillarity. Noncapillarity is the characteristic of some nonabsorbable sutures in which the nature of the raw material or specific processing meets USP tests that establish them as resistant to wicking transfer of body fluids.
The two classifications of suture materials are subdivided into monofilament and multifilament strands:
1. Monofilament suture: A strand consisting of a single threadlike structure that is noncapillary. Monofilament suture is generally less traumatic when pulled through tissue. The smooth surface glides easily without catching on tissue. Some monofilament suture retains its curved shape when removed from the packet. This is referred to as memory. Manufacturers recommend that the suture not be tugged on to straighten the strand. It can weaken the integrity.
2. Multifilament suture: A strand made of more than one threadlike structure held together by braiding or twisting. This suture strand is capillary unless it is treated to resist capillarity or is absorbable. Multifilament suture has a rougher surface and can be somewhat traumatic as it is pulled through tissue layers. Gut suture has memory and will weaken if stretched for straightening.
The following factors influence suture choice:
• Biologic characteristics of the material in tissue (e.g., absorbable vs. nonabsorbable, capillary vs. noncapillary, or inertness).
• Healing characteristics of tissue. Tissues that normally heal slowly, such as skin, fascia, and tendons, usually are closed with nonabsorbable sutures. Absorbable suture placed through the skin may cause a stitch abscess to develop because it is inclined to act as a culture medium for microorganisms in the pores of the skin. Tissues that heal rapidly, such as stomach, colon, and bladder, may be closed with absorbable sutures. Braided suture can be the nidus for stone formation if placed in moisture reservoirs, such as the urinary bladder.
• Presence or absence of infection, contamination, and/or drainage. If infection is present, sutures may be the origin of granuloma formation with subsequent discharge of suture and sinus formation. Foreign bodies in potentially contaminated tissues may convert contamination to infection. Foreign bodies in the presence of some body fluids may cause stone formation, as in the urinary or biliary tract. Braided suture is not used in the presence of infection.
• Patient problems such as obesity, debility, advanced age, and diseases, which influence the rate of healing and time desired for wound support.
• Physical characteristics of the material such as ease of passage through tissue, knot tying, and other personal preferences of the surgeon.
Absorbable Sutures (Table 28-2)
Surgical Gut
Surgical gut is collagen derived from the submucosa of sheep intestine or serosa of beef intestine. Many elaborate mechanical and chemical cleaning processes occur before intestinal ribbons of collagen are spun into strands of various sizes, ranging from the heaviest (size 3) to the finest (size 6-0). Gut is packed in an alcohol solution. The manufacturer recommends that gut suture not be rinsed before use. Although the larger sizes are made from two or more ribbons (multifilament), the behavior of surgical gut is that of a monofilament suture. Surgical gut is digested by body enzymes and absorbed by tissue so that no permanent foreign body remains.
Gut is available in plain (untreated) or chromic (treated with chromium salts). Plain surgical gut loses tensile strength relatively quickly. It is used to ligate small vessels and to suture subcutaneous fat. It is not used to suture any layers of tissue likely to be subjected to tension during healing. Fast-absorbing plain surgical gut is specially treated to speed absorption and tensile strength loss. It may be used for epidermal suturing where sutures are needed for no more than a week.
Chromic surgical gut is treated in a chromium salt solution to resist absorption by tissues for varying lengths of time, depending on the strength of the solution and duration and method of the process. Chromic surgical gut is used for ligation of larger vessels and for suture of tissues in which nonabsorbable materials are not usually recommended because they may act as a nidus for stone formation, as in the urinary or biliary tracts.
Collagen Sutures
Collagen sutures are extruded from a homogeneous dispersion of pure collagen fibrils from the flexor tendons of beef. Both plain and chromic types are similar in appearance to surgical gut and may be dyed blue. Sizes range from 4-0 through 8-0. These sutures are used primarily in ophthalmic surgery.
Handling Characteristics of Surgical Gut and Collagen
1. Most surgical gut and collagen sutures are sealed in packets that contain an alcohol solution to keep the material pliable. This solution is mostly alcohol and water but may be irritating to ophthalmic tissues. Hold the packet over a basin and open carefully to avoid spilling fluid on the sterile field or splashing it into your own eyes. Rinsing is necessary only for surgical gut or collagen sutures to be implanted into the eye. Do not soak or the suture will weaken.
2. Surgical gut and collagen sutures should be used immediately after removal from their packets. When the material is removed and not used at once, the alcohol evaporates and the strand loses pliability. Excessive exposure to water or saline will reduce the tensile strength. Before unwinding, the strand can be dipped momentarily in water or saline solution at room temperature; heat will coagulate the protein.
TABLE 28-2
| Suture Name | Strand Formation | Sizes Available | Colors | Tensile Strength | Notes |
| Plain gut | Twisted multifilament | 6-0 to 3 | Yellow Blue Black | 5-10 days | Natural. Duration 70 days. Packed in alcohol solution. Not used in presence of infection. |
| Chromic gut | Twisted multifilament | 7-0 to 3 | Brown Blue Black | 14-21 days | Natural. Duration 90 days. Packed in alcohol solution. Not used in presence of infection. |
| Collagen | Twisted multifilament | 4-0 to 8-0 | Blue | Natural. Available as plain or chromic. Used in eye surgery. Not used in presence of infection. | |
| Maxon∗ | Monofilament | 7-0 to 2 | Green Clear | 60 days | Synthetic. Duration 6 months. Nonantigenic, nonpyogenic. Prolonged approximation without stress. Hydrolytic absorption by tissues. Can be used in presence of infection. |
| PDS, PDSII† | Monofilament | 9-0 to 2 10-0 to 7-0 | Violet Blue Clear | 90 days | Synthetic. Duration 6 months. Nonantigenic, nonpyogenic. Prolonged approximation without stress. Hydrolytic absorption by tissues. Can be used in presence of infection. Antibacterial IRGACARE (a form of Triclosan). |
| Monocryl† | Monofilament | 6-0 to 2 | Golden Clear Undyed Violet | 21 days | Synthetic. Duration 91-119 days. Antibacterial IRGACARE (a form of Triclosan) |
| Vicryl† | Braided | Coated 9-0 to 2 Undyed 8-0 to 1 | Violet Undyed | 40 days | Synthetic. Duration 90 days. Coating is a lubricant. Available as 42-day fast absorbing “Vicryl Rapide” Antibacterial IRGACARE (a form of Triclosan). Not used in presence of infection. |
| Velosorb∗ | Braided | 6-0 to 1 | Violet Undyed | 5-10 days | Synthetic. Duration 40-50 days. Not used in presence of infection. |
| Polysorb∗ | Braided | 8-0 to 2 | Violet Undyed | 21 days | Synthetic. Duration 56-70 days. Not used in presence of infection. |
Biosyn∗ Caprosyn∗ | Monofilament | 6-0 to 1 | Violet Undyed | 10 days to 3 weeks | Synthetic. Duration 90-110 days (Biosyn) Duration 56 days (Caprosyn) |

3. Unwind the strand carefully. The suture is stiff and has folded memory. Handle it as little as possible. Never pull or stretch surgical gut; this weakens it. Do not straighten suture by running fingers down the length of the strand; excessive handling with gloves can cause fraying.
Synthetic Absorbable Polymers
Polymers, either dyed (colored) or undyed (white or clear), are extruded into absorbable suture strands. These synthetic sutures are absorbed by a slow hydrolysis process in the presence of tissue fluids. They are used for ligating or suturing except when extended approximation of tissues under stress is required. They are inert, nonantigenic, and nonpyrogenic and produce only a mild tissue reaction during absorption. They may be monofilament or multifilament, coated or uncoated.
Handling Characteristics of Synthetic Absorbable Polymers
• Synthetic absorbable sutures have an expiration date on the package. Therefore rotate stock. “First in, first out” is a good rule to follow.
• Synthetic absorbable sutures are packaged and used dry. Do not soak or dip in water or normal saline solution. The material hydrolyzes in water, so excessive exposure to moisture will reduce the tensile strength. It is smooth and soft and will retain its pliability.
Nonabsorbable Sutures (Table 28-3)
Surgical Silk
Surgical silk is a natural animal product made from the fiber spun by silkworm larvae in making their cocoons. From the raw state, each fiber is processed to remove natural waxes and gums. Fibers are braided or twisted together to form a multifilament suture strand. Braided silk is used more frequently because surgeons prefer its high tensile strength and better handling qualities. Surgical silk is treated to render it noncapillary. They lose tensile strength if wet. Therefore they should not be moistened before use.
TABLE 28-3
| Suture Name | Strand Formation | Sizes Available | Colors | Tissue Effects | Notes |
Silk Sofsilk∗ Perma-hand† | Braided | 9-0 to 5 | Black White | Acute tissue inflammation | Natural fiber coated with silicon or wax. Encapsulates. Not used in presence of infection. Not detectable after 2 years. |
| Cotton† | Twisted strands | Umbilical tape | White | Minimal tissue inflammation | Natural fiber. Moisten before use. Single-strand suture rarely used. |
| Ethilon† | Monofilament | 11-0 to 2 | Black Green Clear | Minimal tissue inflammation | Synthetic nylon |
| Nurolon† | Braided | 6-0 to 1 | Black | Minimal tissue inflammation | Synthetic nylon Not used in presence of infection. |
Dermalon∗ Monosof∗ | Monofilament | 11-0 to 2 | Blue Black Clear | Minimal tissue inflammation | Synthetic nylon. Uncoated. |
| Surgilon∗ | Braided | 6-0 to 1 | Black White | Minimal tissue inflammation | Synthetic nylon. Silicone coated. Not used in presence of infection. |
| Prolene† | Monofilament | 10-0 to 2 | Blue Clear | Minimal tissue inflammation | Synthetic. Can be used in a contaminated wound. Available with attached Teflon pledgets. |
| Pronova† | Monofilament | 8-0 to 5-0 | Blue | Minimal tissue inflammation | Synthetic. Can be used in a contaminated wound. |
| Surgipro II∗ | Monofilament | 8-0 to 2 | Blue | Minimal tissue inflammation | Synthetic. Can be used in a contaminated wound. |
| Vascufil∗ | Monofilament | 7-0 to 2-0 | Blue | Minimal tissue inflammation | Synthetic. Less memory. Can be used in a contaminated wound. |
| Novafil∗ | Monofilament | 7-0 to 2 | Blue Clear | Minimal tissue inflammation | Synthetic. Can be used in a contaminated wound. |
| Ti-Cron∗ | Braided | 7-0 to 5 | Blue White | Minimal tissue inflammation | Synthetic. Coated with silicone. Available uncoated. Available with precut pledgets. Not used in presence of infection. |
| Surgidac∗ | Braided | 6-0 to 4-0 | White | Minimal tissue inflammation | Synthetic Not used in presence of infection. |
| Mersilene† | Braided Monofilament | 10-0 to 5 | Green White | Minimal tissue inflammation | Synthetic. Braided form not used in presence of infection. |
| Ethibond† | Braided | 7-0 to 5 | Green White | Minimal tissue inflammation | Synthetic. Coated with polybutilate. Encapsulates. Available with attached Teflon pledgets. Not used in presence of infection. |
| Steel | Monofilament Multifilament | 6-0 to 7 | Silver | Minimal tissue inflammation | Low carbon alloy. Do not handle with serrated instrument. Only use wire scissors to cut. Do not use with metal implant of different alloy. |

Silk is not a truly nonabsorbable material. It disappears within 2 years. It causes less tissue reaction than does surgical gut, but it is not as inert as most of the other nonabsorbable materials. It is used frequently in the serosa of the gastrointestinal tract and to close fascia in the absence of infection.
Surgical Cotton
Cotton is a natural cellulose fiber. Suture is made from individual, long-staple cotton fibers. Single-strand cotton suture is rarely used because it causes an inflammatory reaction. Cotton umbilical tapes are used to tie umbilical cords or retract tissue structures during a surgical procedure. It should be moistened before it is handed to the surgeon. Moisture prevents clinging to the surgeon’s gloves.
Surgical Stainless Steel
Stainless steel sutures are drawn from 316L-SS (L for low carbon) iron alloy wire. This is the same metal formula used in the manufacture of surgical stainless steel implants and prostheses. Two different kinds of metal alloy should not be embedded in the tissues simultaneously. This combination creates an unfavorable electrolytic reaction. Some implants and prostheses are made of Vitallium, titanium, or tantalum. Suture material in the wound must be compatible with these metals.
Before the availability of surgical stainless steel from suture manufacturers, commercial steel was purchased by weight, using the Brown and Sharpe (B&S) scale for diameter variations. Many surgeons still refer to surgical stainless steel size by the B&S gauge, from 18 (the largest diameter) to 40 (the smallest). One manufacturer labels surgical stainless steel with both B&S gauge and equivalent USP diameter classifications. Both monofilament and twisted multifilament stainless steel strands are available.
Surgical stainless steel is inert in tissue and has high tensile strength. It gives the greatest strength of any suture material to a wound before healing begins and supports a wound indefinitely. Some surgeons use stainless steel for abdominal wall or sternal closure or for retention sutures to reduce the danger of wound disruption in the presence of contributing factors. It may be used in the presence of infection or in patients in whom slow healing is expected. It is used for secondary repair or resuturing after wound disruption.
Unlike most other suture materials, steel lacks elasticity. A suture secured too tightly may act as a knife and cut through tissue. Stainless steel sutures are harder to handle than any other suture material. A painstaking knot-tying and twisting technique with a nonserrated instrument is required. The serrations of the instrument will create weak areas along the suture. Wire suture is only cut with wire scissors.
Handling Characteristics of Surgical Stainless Steel Suture
1. Surgical stainless steel strands are malleable and kink rather easily. Kinks in the strand can make it practically useless. Therefore use care in handling to keep the strand straight.
2. Barbs on the end of a strand can tear gloves, puncture the skin, or traumatize tissue. Sternal wire suture has been known to extrude through the skin if the cut end is not buried deep into the tissue.
3. If surgical stainless steel must be threaded through a needle, some surgeons prefer one or two twists of the end around the strand just below the eye of the needle to prevent unthreading during suturing.
Synthetic Nonabsorbable Polymers
Synthetic nonabsorbable sutures have higher tensile strength and elicit less tissue reaction than does silk. They retain their strength in tissue. Knot tying with most of these materials is more difficult than with silk. Additional throws are required to secure the knot. The surgeon may sacrifice some handling characteristics and ease of knot tying for strength, durability, and nonreactivity of the synthetics. These advantages may outweigh the disadvantages. Synthetic nonabsorbable sutures are available as monofilament or braided strands.
Surgical Nylon
Nylon is a polyamide polymer derived by chemical synthesis from coal, air, and water. It produces minimal tissue reaction. Nylon has high tensile strength, but it degrades by hydrolysis in tissue at a rate of about 15% to 20% per year. It may be used in all tissues where a nonabsorbable suture is acceptable, except when long-term support is critical. It is available in three forms: monofilament, uncoated multifilament, and coated multifilament.
Polyester Fiber
A polymer of terephthalic acid and polyethylene, Dacron polyester fiber is braided into a multifilament suture strand that is available in two forms: uncoated fiber and coated fiber.
1. Uncoated polyester fiber suture is closely braided to provide a flexible, pliable strand that is relatively easy to handle. However, uncoated braided polyester fiber suture has a tendency to drag and exert a sawing or tearing effect when passed through tissue. It may be used in all tissues in which a multifilament nonabsorbable suture is indicated.
2. Coated polyester fiber suture has a lubricated surface for smooth passage through tissue. Sutures are available with different coating materials:
a. Polybutilate is the only coating developed specifically as a surgical lubricant. This polyester material adheres strongly to the braided polyester fiber. Polyester fiber coated with polybutilate provides a strand superior to any other braided material, coated or uncoated, in decreasing drag through tissue.
b. Polyfluoroethylene (PTFE), a commercial product known by the name Teflon and manufactured by DuPont, is used as a coating bonded to the surface or impregnated into spaces (interstices) in the braid of the polyester fiber strand. Minute particles of this coating can flake off the strand. Because these particles are insoluble and resistant to enzymes, foreign body granulomas may be produced.
c. Silicone, a commercial lubricant, provides a slippery coating but does not bond well to polyester fiber. It can become dislodged in tissues as the strand is tied.
Polypropylene Suture
A polymerized propylene is extruded into a monofilament strand. It is the most inert of the synthetic materials and almost as inert as stainless steel. Polypropylene is an acceptable substitute for stainless steel in situations in which strength and nonreactivity are required, and it is easier to handle. The suture may be left in place for prolonged healing. It can be used in the presence of infection.
Handling Characteristics of Synthetic Nonabsorbable Polymers
1. Physical damage to suture materials can occur from the time the suture is removed from a packet if the strand is mishandled. Handle all sutures and needles as little as possible. Avoid pulling or stretching. Sutures should be handled without using instruments except when grasping the free end during an instrument tie. Clamping instruments—especially needle holders and forceps with serrations—on strands can crush, cut, and weaken them.
2. All synthetic materials require a specific knot-tying technique. Knot security requires additional flat and square ties. Multifilament materials are generally easier to tie than are monofilament sutures.
Barbed Suture (Table 28-4)
Barbed Suture
This synthetic self-anchoring suture is used for low tension suturing without the need of knotted ends. Barbed synthetic suture is available in absorbable and nonabsorbable material. Handling barbed suture is done with care. The barbs are tiny but still can snag on sponges or drapes. Use of barbed suture has many applications in several specialties. Barbed suture has two different configurations: unidirectional barbs and bidirectional barbs.
Unidirectional barbed suture has a needle on one end and a loop on the other end. The loop can be used to anchor the suture in place of a knot. The barbs are designed to pull through the tissue in one direction and maintain placement without moving. At the completion of closure the needle can be cut off without losing stability of the wound.
Bidirectional barbed suture is double-armed and has raised barbs that are angled from the center to the ends in both directions. During closure the suture is placed with a slight reverse torque to anchor the barbs into the dermis from the center in one direction, and then the reverse is performed in the opposite direction. The result is a well-approximated incision line.
TABLE 28-4
| Barbed Suture | Quill Surgical Specialties Corp | V-Loc Covidien | Stratafix Ethicon |
Unidirectional Knotless Loop end | Polypropylene: Clear Nonabsorbable Size 0 | V-Loc PBT: Polybutester: Blue Nonabsorbable Size 3-0 to 1 Endo 3-0 to 0 | Polypropylene: Undyed Nonabsorbable, Spiral Size 5-0 to 1 |
Monoderm: Violet or Clear Absorbable Size 4-0 to 0 90-120 days | V-Loc 180: Green or clear Absorbable Size 4-0 to 0 Endo 3-0 to 0 180 days | PGA-PCL: Clear Absorbable, Spiral Size 4-0 to 0 90-120 days | |
PDO: Violet Absorbable Size 4-0 to 2 180 days | V-Loc 90: Violet or undyed Absorbable Size 4-0 to 2-0 90-110 days | PDO: Violet and Clear Absorbable, Spiral Size 4-0 to 1 120-180 days | |
Bidirectional Double-armed Knotless | Polypropylene: Clear Nonabsorbable Size 2-0 to 2 | Polypropylene: Clear Nonabsorbable, Spiral Size 5-0 to 1 | |
Nylon: Blue Nonabsorbable Size 1 to 2 | |||
Monoderm: Violet or Clear Absorbable Size 4-0 to 0 90-120 days | PGA-PCL: Violet or Clear Absorbable, Spiral Size 5-0 to 1 | ||
PDO: Violet Absorbable Size 4-0 to 2 180 days | PDO: Violet and Clear Absorbable, Spiral Size 5-0 to 1 120-180 days |

Surgical Needles
Except for simple ligating with free ties, surgical needles are needed to safely carry suture material through tissue with the least amount of trauma. The best surgical needles are made of high-quality tempered steel that is:
• Strong enough that it does not break or fracture easily
• Rigid enough to prevent excessive bending, yet flexible enough to prevent breaking if bent
• Sharp enough to penetrate tissue with minimal resistance (yet it need not be stronger than the tissue it penetrates)
• Approximately the same diameter as the suture material it carries to minimize trauma in passage through tissue
• Appropriate in shape and size for the type, condition, and accessibility of the tissue to be sutured
• Free from corrosion and burrs to prevent infection and tissue trauma
Because needles are made of steel, theoretically they are detectable by x-ray if inadvertently lost in tissue. The location in tissue may preclude the needle from appearing on a radiograph. For example, the angle of the needle or its position behind bone may obstruct detection. The smaller the size of the needle, the more likely the image is to be obstructed. All needles should be accounted for in their entirety so that they do not become foreign bodies in tissue. Lost needles can become embedded in linen or trash, causing harm to unsuspecting support personnel.
Many shapes and sizes of surgical needles are available. Names vary from one manufacturer to another; general classification only, not nomenclature, is standardized. They may be straight like a sewing needle or curved. All surgical needles have three basic components: the point, the body (or shaft), and the eye. They are classified according to these three components.
Point of the Needle
Points of surgical needles are honed to the configuration and sharpness desired for specific types of tissues. The basic shapes are cutting, tapered, and blunt (Fig. 28-11).
Cutting Point
A razor-sharp, honed cutting point may be preferred when tissue is difficult to penetrate, such as skin, tendon, and tough tissues in the eye. These make a slight cut in tissue as they penetrate. The location and degree of sharpness of cutting edges vary. Cutting needles are less traumatic as they pass through tissues.
Conventional Cutting Needles
Two opposing cutting edges form a triangular configuration with a third edge on the body of the needle. Cutting edges are on the inside curvature of a curved needle. Cutting edges may be honed to precision sharpness to ensure smooth passage through tissue and a minute needle path that heals quickly.
Reverse-Cutting Needles
A triangular configuration extends along the body of the needle. The edges near the point are sharpened or honed to precision points. The two opposing cutting edges are on the outer curvature of a curved needle.
Side-Cutting Needles
Trocar Point
Sharp cutting tips are at the points of tapered needles. All three edges of the tip are sharpened to provide cutting action with the smallest possible hole in tissue as it penetrates.
Taper Point
These needles are used in soft tissues, such as intestine and peritoneum, which offer a small amount of resistance to the needle as it passes through. They tend to push the tissue aside as they go through, rather than cut it. The body tapers to a sharp point at the tip.
Blunt Point
These tapered needles are designed with a rounded blunt point at the tip. They are used primarily for suturing friable tissue, such as liver and kidney. Because the blunt point will not cut through tissue, it is less apt to puncture a vessel in these organs than is a sharp-pointed needle. Blunt needles also may be used in some tissues to reduce the potential for needlesticks, especially in general and gynecologic surgery.
Body of the Needle
The body, or shaft, varies in wire gauge, length, shape, and finish. The nature and location of tissue to be sutured influence the selection of needles with these variable features. Most manufacturers have designated a specific alphanumeric code to describe each needle they produce. Examples of alphanumeric codes for taper-point needles can be found in Table 28-5.
Considerations relating to the body of the needle are as follows:
1. Tough or fibrosed tissue requires a heavier-gauge needle than the fine-gauge diameter needed in microsurgery.
2. The depth of the bite (placement) through tissue determines the appropriate needle length.
3. The body of the needle may be round, oval, flat, or triangular. The point determines the shape: round or oval bodies have trocar, taper, or blunt points; flat or triangular bodies have cutting edges. The shape may also be straight or curved (Fig. 28-12).
TABLE 28-5
Select Examples of Common Taper-Point Needles (Alphanumeric Codes) and Representative Manufacturers
| Purpose | Ethicon | Covidien | Configuration |
| General closure | CT | GS 24 | ½ circle taper 40 mm |
| General closure | CT 1 | GS 21 | 37 mm |
| Cardiovascular | RB | EV 23 | 17 mm |
| Gastrointestinal | SH | EV 20 | 25 mm |
| Multilayer closure | CTX | GS 25 | 48 mm |
| Cardiovascular | BB | CV 15 | ⅜ circle taper 17 mm |
| Urologic | UR 6 | GU 46 | ⅜ circle taper 27 mm |

a. Straight needles are used in readily accessible tissue. They have cutting points for use in skin, which is their most frequent use, or tapered points for use in intestinal tissue held by a pursestring device. They are not used with a needle holder.
b. Curved needles are used to approximate most tissues, because quick needle turnout is an advantage. The curvature may be ¼, ⅜, ½, or ⅝ circle; half-curved with only the tip curved; or compound curved. Curved needles always are armed in a needle holder before being handed to the surgeon.
c. J-shaped needles range from 15.5 cm to 17.5 cm in length. The width of the J bend at the tip is available in 7- to 9-mm curvature. They are used for 10- to 14-mm fascial incisions created for trocar use during laparoscopy. They are multiuse needles that can be threaded with the suture of the surgeon’s choice at the point of use. Their unique shape enables the surgeon to close the deep layers of the wound without perforation of underlying organs while visualizing closure of the accessory ports with the laparoscopic camera.
4. Curved needles that have longitudinal ribbed depressions or grooves along the body on the inside and outside curvature can be cross-locked in the needle holder. This feature virtually eliminates twisting or turning of the needle in any position in the needle holder.
5. In all needles the body must have a smooth finish. Many needles have a surface coating of microthin plastic or silicone to enhance smooth passage through tissue. Others have a black surface finish to enhance visibility at the surgical site.
Eye of the Needle
The eye is the segment of the needle where the suture strand is attached. Surgical needles are classified as eyed, French eye, or swaged or atraumatic. Swaged-on suture is inserted directly into the end of the needle by the manufacturer (Fig. 28-13, C).
Eyed Free Needle
The closed eye of an eyed surgical needle is like that of any household sewing needle. Needles that are threaded by the scrub person are referred to as free needles. The shape of the enclosed eye may be round, oblong, or square. The end of the suture strand is pulled 2 to 4 inches (5-10 cm) through the eye so that the short end is about one sixth the length of the long end. Unlike regular sewing thread, there is no knot placed in the end of the suture.
Disposable varieties are preferred. Some specialty needles will be packed in-house on a needle rack. Care is taken when handling these needles. Careful counting and accountability are required. In some cases, if a swaged suture breaks during a procedure the surgeon may request a free needle to complete the suturing process.
French Eye Needle
Sometimes referred to as spring eye or split eye, a French eye needle has a slit from the inside of the eye to the end of the needle through which the suture strand is drawn. To thread a French eye after arming the needle in a needle holder, 2 to 3 inches (5-7.5 cm) of the strand is secured between the fingers holding the needle holder. The strand is pulled taut across the center of the V-shaped area above the eye and drawn down through the slit into the eye (Fig. 28-13, B, and Fig. 28-14).
French eye needles, as a general rule, are used with pliable braided materials, primarily silk and cotton, of medium or fine size. These needles are not practical for surgical gut; the strand may fray, or the eye may break because the diameter is usually too large for the slit.
Handling of Free Needles
Eyed and French eye needles have the following disadvantages for the scrub person, surgeon, and patient:
• Each needle must be carefully inspected by the scrub person before and after use for dull or burred points and defects in the eye.
• Care must be taken to avoid puncturing gloves with the needle point when threading.
• If the scrub person must choose an appropriate needle to thread, the needle should be the same approximate diameter as the suture size requested by the surgeon.

• FIG. 28-14 To thread French eye needle, pull strand taut across center of V-shaped area and draw down through slit into eye.
• Needles can unthread prematurely. This is an annoyance to the surgeon and prolongs operating time for the patient. To avoid this, the surgeon may prefer the suture strand threaded double with both ends pulled the same length through the eye. The scrub person may lock the suture strand by threading the short end through the eye twice in the same direction.
• Two strands of suture material are pulled through tissue when threaded needles are used. The bulk of the double strand through the eye creates a larger hole than the size of the needle or suture material, causing additional trauma to tissue.
Swaged-on Needle
A swaged-on needle is a continuous unit with the suture strand. The needle is swaged onto the end of the strand in the manufacturing process. This eliminates threading at the operating bed and minimizes tissue trauma, because a single strand of material is drawn through tissue (Fig. 28-15). The diameter of the needle matches the size of the strand. The surgeon uses a new sharp needle with every suture strand. Usually referred to as swaged needles, three types of swaged-on needle-suture attachments are available.
1. Single-armed attachment: One needle is swaged to a suture strand.
2. Double-armed attachment: A needle is swaged to each end of the suture strand. The two needles are not necessarily the same size and shape. These are used when the surgeon wishes to place a suture and then continue to approximate surrounding tissue on both sides from a midpoint in the strand. Barbed suture is packed this way, for example.
3. Controlled-release needle attachment: This attachment is secure, so the suture strand will not separate from the needle inadvertently, but it will release rapidly when pulled off intentionally. Usually only single-armed. The surgeon grasps the suture strand just below the needle, pulling the strand taut, and releases the needle with a straight tug of the needle holder on the needle. This facilitates fast separation of the needle from the suture when desired. This type of needle is referred to as a pop-off needle.
Placement of the Needle in the Needle Holder
Needle holders have specially designed jaws to securely grasp surgical needles without damage if they are used correctly. Care is taken not to use needle holders for anything other than holding needles. The jaw can weaken or spring, causing the needle to twist and turn when suturing. The scrub person should observe the following principles in handling needles and needle holders:
1. Select a needle holder with appropriate-size jaws for the size of the needle to be used. An extremely small needle requires a needle holder with very-fine-tipped jaws. As the wire gauge of the needle increases, the jaws of the needle holder selected should be proportionately wider and heavier. Curved jaws or angulated handles may be needed for placement of the needle in tissues.
2. Select an appropriate-length needle holder for the area of tissue to be sutured. When the surgeon works deep inside the abdomen, chest, or pelvic cavity, a longer needle holder will be needed than is needed in superficial areas.
3. Clamp the body of the needle in an area one fourth to one half the distance from the eye to the point (Fig. 28-16). Never clamp the needle holder over the swaged area. This is the weakest area of a swaged-on needle because it is hollow before the suture strand is attached. Pressure on or near the needle-suture juncture may break the needle.

• FIG. 28-16 Correct position of curved needle in needle holder for right-handed surgeon, about one third down from swage or eye.
4. Place the needle securely in the tip of the needle holder jaws, and close the needle holder in the first or second ratchet. If the needle is held too tightly in the jaws or the needle holder is defective, the needle may be damaged or notched in such a manner that it will have a tendency to bend or break on successive passes through tissue.
If the needle breaks it must be accounted for in its entirety. All pieces and fragments must be retrieved from the patient’s body and the surrounding environment for personnel safety.
5. Pass the needle holder with the needle point up and directed toward the surgeon’s thumb when grasped so that it is ready for use without readjustment. If a hands-free technique is preferred, place the needle holder on a tray or magnetic mat with the needle point facing down.
6. Hand the needle holder to the surgeon so that the suture strand is free and not entangled with the needle holder. Hold the free end of the suture in one hand while passing the loaded needle holder with the other hand. Protect the end of the suture material from dragging across the sterile field. The assistant may take hold of the free end to keep the strand straight for the surgeon and to keep it from falling over the side of the sterile field. This is referred to as trailing the suture.
7. Hand the needle holder to an assistant to pull the needle out through tissue. A hemostat or other tissue forceps is not used for this purpose because the instrument may be damaged or may damage the needle. The needle should be grasped as far back as possible to avoid damage to the point or cutting edges.
Considerations in the Choice of Sutures and Needles
The surgeon chooses from available types and sizes of sutures and needles, the ones that best suit each purpose. In general, fine sizes are used for plastic, ophthalmic, pediatric, and vascular surgery; medium sizes are used for all other kinds of surgery; heavy sizes are used for retention and for anchoring bone. In general, cutting needles are used in tough tissue such as skin, fascia, tendon, and mucous membranes, including the cervix, palate, tongue, and nose. Medium tissue calls for round taper-point or cutting needles. Round taper-point needles generally are used for nerve, peritoneum, muscle, and other soft tissue, such as lung and intestine, subcutaneous tissue, and dura.
It is almost impossible to learn the needle-tissue-suture-surgeon combinations by memory alone because of the unlimited number of combinations. Learning the general classification of needles, sutures, and tissues is the first step; practical experience is necessary to remember the combinations. Try to learn the suture and needle combinations most commonly used.
The preference card usually lists the surgeon’s usual suture-and-needle routine by tissue layer. Some cards list swaged-on sutures by order number, but this can be confusing. Some of the order numbers include alphanumeric labels that refer to how many sutures are packed in a box. This has nothing to do with the suture itself. Others list sutures by size and materials and needles by size and shape, which is easier to follow. Suture and needle sizes are as variable as patient sizes. Therefore the surgeon may unexpectedly request a smaller or larger size out of routine for a particular patient’s situation.
Each manufacturer produces different needle styles. Most types have a comparable counterpart in another brand name. Table 28-5 describes cross-referencing of needle types by alphanumeric codes among different manufacturers.
Packaging and Preparation of Sutures and Needles
Suture material is individually packaged and supplied sterile by the manufacturer. It is sterilized by cobalt-60 irradiation or ethylene oxide gas. Reprocessing of suture material is not recommended. It can be damaged and is not guaranteed to be sterile.
Swaged needles come in sterile packets attached to the suture material. The suture needles are counted as the packet is dispensed to the field and confirmed as they are opened. When the swaged-on suture is opened in the field the contents are verified. Discard the outer sterile packet as soon as the suture is opened. Saving the empty packet on the field is not a reliable way to keep track of the number of suture needles used.
Disposable eyed and French eye needles are individually packaged and sterilized by the manufacturer. Needles are counted when dispensed to the sterile field one at a time and only as needed. Single-use needles ensure a sharper point with less tissue trauma.
Preparation of Reusable Needles
Standard sets of reusable eyed and French eye needles may be prepared for each surgical procedure where they are used. This necessitates preparing more needles in a package than will actually be used. The margin for error in counting increases when too many needles are present on the field. Choose needles for each procedure according to each surgeon’s preferences as listed on the preference card.
Packaging of Suture Materials
A strand of sterile suture material is supplied with as many as four coverings.
Box
Each box is sealed in cellophane and contains one, two, or three dozen packets of sterile suture material. The label on the box may be color-coded by suture material (e.g., light blue for silk, tan for chromic gut, yellow for plain surgical gut). Competing manufacturers frequently select similar colors for box labels and suture material identification. Most boxes fit into a suture cabinet rack. Many synthetic sutures have expiration dates because the material degrades with time. Each box is provided with a lot number that corresponds to the lot number on the individual suture packets.
Overwrap
Each individual suture packet has a sealed outer overwrap. The overwrap is peeled back to expose the inner primary packet for aseptic transfer to the sterile table. The circulating nurse must not contaminate the sterile inner primary packet as the overwrap is peeled apart and the packet is transferred onto the sterile table or presented to the scrub person.
Primary Sterile Packet
Suture material, with or without swaged needles, is sealed in a sterile primary inner packet that is to be opened by the scrub person. The primary packet may be made of foil, paper, or plastic, or combinations of these. Labels may be color-coded by material, the same as the box. If a swaged needle is enclosed, a silhouette of the needle is included on the label, along with the size and type of suture material.
A single strand of material or multiple strands may be in the primary packet. The packet may be designed for dispensing individual strands from a multiple suture packet. Packaging configurations and considerations include:
1. Suture packets should be opened only as needed, to minimize waste.
2. Custom kits with multiple suture packets within a single package facilitate dispensing sutures to and organizing them on the sterile table. The kit may have appropriate sutures, with or without swaged needles, to meet the requirements of a particular surgeon or procedure. The packets are organized in order of use. The contents are listed on the cover of the package to facilitate counts. Expiration date will be printed on the outer label if synthetic suture is inside.
3. Sterile suture packets are labeled “Do Not Resterilize.” Component layers of the packaging materials cannot withstand exposure to the heat of steam sterilization without potential physical damage to the contents and packets. The manufacturer will not guarantee product stability or sterility for packets resterilized in the hospital or in secondary processors, or for strands removed from packets and sterilized.
4. Some suture materials have an expiration date stamped on the box and primary packet to indicate the stability of the material. Oldest sutures should be used first. Sutures should not be used past this expiration date because the chemical composition may have started to degrade, and the safety and stability of the suture is in question.
Inner Dispenser Matrix
Suture material is contained within the primary sterile packet in a manner that facilitates removing or dispensing it. This may be a paper folder, reel, or plastic tray that may or may not be removed with the suture strands. The inner matrix keeps the suture from tangling as it is pulled out for use.
Preparation of Suture Material
The length of each strand of suture material within the primary packet varies; the shortest is 5 inches (approximately 13 cm), and the longest is 60 inches (150 cm). The most commonly used lengths range from 18 to 30 inches (45-75 cm). The length the surgeon prefers should be noted on the preference card. The scrub person may have to cut the strands to the desired length, depending on the lengths available.
Standard Length
The term standard length refers to a 60-inch (150-cm) strand of nonabsorbable material or a 54-inch (135-cm) strand of absorbable material without a swaged needle referred to as a free tie. It is not handed to the surgeon in this length. The scrub person may cut it into a half-length, or third-length for use as a free tie (suture ligature) or thread it for a stick tie or suture, as shown in Figures 28-17 and 28-18.
Ligating Reels
Twelve-foot (approximately 4-m) lengths of nonabsorbable or 54-inch (135-cm) lengths of absorbable suture are wound on disposable plastic reels. These reels are color-coded by material and have size identification. The surgeon keeps the reel in the palm of the hand for a series of free ties. The reel is radiopaque in case it is inadvertently dropped in a body cavity. These are counted items.
Precut Lengths
Most suture materials are supplied in precut lengths of 12 to 18 inches and are ready for use as free ties or for threading. These facilitate handling for the scrub person. They are dispensed individually from some primary packets or may be removed and placed in a fold of a towel near the sterile field. Packets contain from 3 to 17 strands, depending on the material.

• FIG. 28-17 Scrub person preparing one third–length sutures. One free end of full-length strand is passed from right to left hand. At same time, loop is caught around third finger of right hand. Other loop is caught around third finger of left hand while each end is held and suture is adjusted to equal lengths (thirds). Then each loop is cut with scissors.

• FIG. 28-18 Sequence of scrub person preparing half-lengths. A, Suture loops are separated by fingers of left hand while unwinding. B, Full length is gently unwound and straightened before cutting. Scrub person does not pull hard or test strand, but keeps firm grasp on both ends to prevent suture from snapping away and possibly becoming contaminated. C, Suture is bent in half, and loop is cut.
Swaged Needle-Suture
The manufacturer predetermines lengths of sutures; however, the surgeon has a wide variety of choices to meet all suturing needs. The scrub person must remember that a strand can be shortened but not extended. An appropriate length for location of tissue must be handed to the surgeon. A packet may contain one suture strand armed with a single or double needle(s) or multiple strands with swaged needles.
The sterile package will depict the needle image of a single-armed suture or two needles in a double-armed suture. The package will also indicate how many sutures are enclosed. The number of suture needles recorded on the pack is reported to the circulating nurse during the count.
Surgical Staples
Surgeons can join many tissues with staples. This involves inserting stainless steel or titanium staples through tissues with a stapler—a device specifically designed for this purpose. Some surgical procedures have become simplified or feasible since the advent of surgical stapling techniques. Titanium internal staples are preferred.
As Hümer Hültl recognized in 1908, for internal stapling to be successful, fine wire as the basic material must form a B shape when stapled into tissue. This shape allows blood to flow through tissues, preventing necrosis secondary to devascularization beyond the staple line. Sufficient pressure must be exerted, however, to provide hemostasis of cut tissues.
Skin staples form a D shape when placed with the stapler. The box shape of the staple allows for adequate blood flow through the dermal layers. The length and width of the staples must accommodate tissue being approximated or transected. The number of staples varies with the length of the staple line.
Individual titanium ligation clips used for hemostasis form a completely occlusive, flat closure. Unlike the protection of blood flow in the shape of internal staples and skin staples, complete closure of the clipped structure is critical to prevent blood loss.
Advantages of Using Staples
Staples can be used safely in many types of tissues and have a wide range of applications.
• Stapling is a rapid method of ligating, anastomosing, and approximating tissues. The time saved, compared with suturing techniques, reduces blood loss and total operating and anesthesia time for the patient.
• Wound healing may be accelerated because of minimal trauma and the nonreactive nature of metallic staples.
• Staples produce an even surface and an airtight, leakproof closure.
• Staples can be placed through an endoscopic trocar.
Stapling Instruments
Each stapler is designed for stapling specific tissues (i.e., skin, fascia, bronchus, gastrointestinal tract, vessels). The surgeon selects the correct instrument for the desired application. The consequences of an erroneous staple application, however, are much more difficult to correct than those of manually placed sutures. The surgeon must learn when and how to use each instrument. Whether the stapler is reusable or a single-use disposable, the basic technical mechanics of stapling are the same.
Staplers either fire a single staple or simultaneously fire straight or circular rows of staples. The rows can be double- or triple-staggered staple lines. A different instrument must be used for each type of firing.
Skin Stapler
To approximate skin edges, the stapler fires a single staple with each squeeze of the trigger. Edges of both cuticular and subcuticular layers are aligned, with the edges slightly raised in an everted direction with Adson pick-ups with teeth, as close to their original configuration on the horizontal plane as possible. Care is taken not to perforate the skin with the tips of the pick-ups. The stapler is positioned over the line of incision so that the staple will be placed evenly on each side. The staple forms a rectangular D shape over the incision. As many staples as needed are placed to close the incision.
Skin staplers are supplied preloaded with different quantities of staples in varying widths (i.e., crown span). The most appropriate stapler should be chosen for the selected use. For example, an average range of 28 to 35 staples is needed to close most abdominal incisions. More may be needed to close the chest; fewer may be needed for an inguinal herniorrhaphy.
Skin staples are removed 5 to 7 days postoperatively. Extractors are used for this purpose. As it heals, the skin flattens out to form an even surface with excellent cosmetic results if the staples have been properly placed lightly over the skin. Embedded staples are difficult to remove, and results may be less than desired.
Internal Staplers
Internal staplers are used to separate, anastomose, or occlude vessels and internal organs. They are manufactured as reusable stainless steel instruments with cartridges, single-use instruments with cartridges, or single-use single-fire disposable instruments.
Several internal staplers used for separation and anastomosis have self-contained knives built in. Some occlusion models do not have knives. They only create a reinforced staple line. The surgeon can trim away excess tissue along the staple line with a scalpel.
Staplers with cartridges and knives can be used on the same patient throughout the entire procedure. As the staples are fired and the knife used, the scrub person carefully changes the cartridge without disturbing the next load of staples. Figure 28-19 shows the three most common styles of internal staplers that can be reloaded or purchased as single-fire instruments.

• FIG. 28-19 Internal staplers. A, Terminal end stapler. B, Internal anastomosis stapler with linear knife. C, End-to-end stapler with circular knife.
To avoid unnecessary contamination of a sterile instrument, the package should not be opened until the surgeon determines the correct size for the intended use of the stapler. Staplers should never be flipped or dumped onto the field. The impact can cause misalignment of the staples or internal knife and result in misfire. This could cause problems during the procedure and possibly cause an anastomic leak postoperatively. The circulating nurse should open the package aseptically and the scrub person should lift the instrument out of the sterile pack. A misfired stapler and its packaging should be given to the supervisor in charge for inspection by the manufacturer.
The numbers and sizes of staples in cartridges vary; the length of the linear jaws or the diameter of the circular head varies for internal use. Internal staplers are available with either stainless steel or titanium staples. The surgeon may prefer titanium, because it creates less distortion when CT scans are used. Titanium staples are safer for use in MRI because they are nonmagnetic. Care is taken to discard any used staple device in the sharps container at the end of the surgical procedure for the safety of the surgical services personnel.
Linear Stapler
Two staggered or side-by-side straight double or triple rows of staples are placed simultaneously in tissue with a linear stapler. This stapler is used throughout the alimentary tract and in thoracic surgery for transection and resection of internal tissues. The tissue is positioned in the straight jaws of the stapler. The gap between the jaws must be adequate for the thickness of tissues. Terminal anastomosis (aka thoracoabdominal [TA]) staplers have no knife built in. Excess tissue is trimmed away after firing with a scalpel before the instrument is removed. TA is commonly used to close a side or end of a structure like a segment of bowel or the lobe of a lung.
Gastrointestinal anastomosis (GIA) staplers have a knife that is run between the two staple lines to separate tissue after firing. GIA is commonly used to join two tubular segments side-by-side to form a pouch. They can also be used to close a terminal end of an organ. GIA is commercially available without a knife, referred to as SGIA. These are not commonly used.
The number of staples that will be fired depends on the length of the stapler jaws. The TA and GIA instruments can be reloaded with a second staple cartridge for use on the same patient. The instrument is discarded at the end of the surgical procedure. Single-use disposable models are available.
Intraluminal Circular Stapler
With the circular stapler a double row of staggered staples is placed in a circle for intraluminal end-to-end anastomosis (EEA) of tubular hollow organs in the gastrointestinal tract. Because the diameter of the lumen of organs in the alimentary tract varies, the surgeon must choose a stapler with an appropriate head circumference size. The number of staples the instrument fires depends on its head size. The instrument can be reloaded with a new staple-cutting head for second use on the same patient. Single-fire models are available. The EEA stapler is available curved or straight.
A circular knife within the head of the EEA stapler trims tissue to produce a round lumen as the stapler is fired. The result is a circular anastomosis and two distinct tissue rings, intact circles of tissue from the anastomosed region referred to as doughnuts. These are sent as two separate specimens to be examined for intactness. Care is taken to keep the distal and proximal tissue circles separate, so if cancerous margins are present they will be correctly identified if a second resection is required.
Ligating and Dividing Stapler
A double row of two staples each ligates tissue and vessels that are then divided simultaneously between the staple lines with a cutting knife incorporated into the stapler. The staple configuration after firing is flat like a ligating clip. Hemostasis is attained. This disposable stapler is used primarily to ligate and divide omental vessels or other small, soft tubular structures. The working end is curved to encircle the vessel as it is ligated and cut. Some types have built-in CO2 cartridges referred to as powered LDS (ligate-divide-staple).
Endoscopic Stapler
Endoscopic staplers are available for ligating and dividing and for linear stapling. The stapling device is passed through an 11- or 12-gauge laparoscopic trocar. It may be reloaded several times with staple cartridges for multiple firings. The stapler is discarded in the sharps container after single-patient use.
Clips
Multi-load or single-load ligating clips are used to completely occlude vessels as needed. Titanium is the usual composition. Clips are applied to individual vessels with a clip application instrument and are designed to close flat. They do not cut tissue. The purpose is to permanently occlude the vessel. The instrument can be a reusable ring-handled stainless instrument or a disposable preloaded plastic applicator. Care is taken not to prematurely squeeze the handles as the instrument is passed to the surgeon. Squeezing will cause the clip to fall from the jaws of the instrument before it can be applied to the vessels.
Tissue Adhesives
Conventional suturing and stapling materials hold tissues in apposition during the healing process. Sutures and staples will not fuse or bond tissues. Ancient Egyptians used resins and gums to hold tissue surfaces together. Research is ongoing for an ideal tissue adhesive that will bond tissue, effect hemostasis, promote regeneration of cells, and serve as a barrier to microbes, fluid, and air. Biologic and synthetic tissue adhesives have extensive potential applications for wound closure and reconstruction.
Biologic Adhesives
Fibrin sealants, most commonly called fibrin glue, act as biologic adhesives and hemostatic agents. The components are fibrinogen, cryoprecipitate from human plasma; calcium chloride; and reconstituted thrombin of bovine origin. When applied directly to tissues, thrombin converts fibrinogen to fibrin to produce a clot. Fibrin sealants can be applied to deeper tissues as a liquid, gel, or aerosol spray to control bleeding and approximate tissues technically difficult to approximate by suturing, especially after resections or traumatic injuries of friable or highly vascular tissues, such as liver, spleen, and lung. Each component is applied simultaneously with separate syringes to prevent congealing before application to the tissue site. It is not mixed ahead of use or it will congeal and be useless. A sample formula and supplies for fibrin glue compound are found in Box 28-2.
Fibrin glue may also be used for microsurgical anastomoses of blood vessels, nerves, and other structures such as fallopian tubes; for reconstruction of the middle ear; to fix ocular implants; to close superficial lacerations and fistula tracts; and to secure some skin grafts. It may be used as a carrier for demineralized bone powder to promote osteoregeneration.
Tisseel is a topical fibrin sealant produced by Baxter Healthcare Corporation that can be applied by a duo syringe or a spray. Tisseel is derived from the human blood proteins fibrin and thrombin and is applied in the same manner as earlier forms of fibrin sealant. No bovine components are used. Aprotinin is an antifibrinolytic protein ingredient that may cause an allergic reaction in some patients. The risk for transmissible viral or prion disease is low, but it still must be considered. Tisseel is used alone and cannot be used with any other hemostatic material—an embolic event can occur. More information is available at http://tisseel.com/us/tisseel_advantage.html.
Autologous or Homologous Plasma
Plasma collected from the patient (autologous) or a single donor (homologous) is processed into a cryoprecipitate containing clotting factor XIII to produce fibrinogen. Autologous plasma is obtained preoperatively and prepared either in the blood bank or in the OR. A single donor must be tested and found to be negative for human immunodeficiency virus (HIV) and hepatitis before plasma is processed.
Autologous or homologous fibrinogen is warmed to 98.6° F (37° C) immediately before use. Thrombin is reconstituted to 1000 units/mL. Equal volumes of fibrinogen and thrombin are applied simultaneously.
Pooled-Donor Plasma
Fibrin glue commercially prepared from pooled-donor plasma (i.e., blended from multiple donors) has significantly greater bonding strength than does autologous or single-donor plasma. The fibrinogen must undergo purification and viral inactivation, however, to prevent transmission of bloodborne pathogens. Used extensively in Europe, this product has not been approved by the FDA for use in the United States.
Synthetic Adhesives
Synthetic glue-like adhesive substances that polymerize in contact with body tissues effect hemostasis and hold tissues together.
Cyanoacrylate
Butyl cyanoacrylate and octyl cyanoacrylate derivatives may be used for topical skin closure (skin glue). Both types of synthetic adhesives are applied to clean, dry, easily approximated wounds and require no additional dressing material. The glue should be introduced into the wound. The adhesive forms a closure over the incision and secures the closure over the skin. The incision remains visible for inspection and the adhesive forms a protective, occlusive antimicrobial barrier. Conversely, if the wound is contaminated, an infectious process can be sequestered.
Skin glue is available from several manufacturers and comes in blue, violet, and clear varieties. Before using skin glue, check the expiration date. The glue dries after 2½ minutes and remains on the wound for 5 to 10 days. The glue is applied with an applicator along approximated, clean wound edges that are not under tension. The product can be applied in a long strand or in a series of closely aligned dots. Thick layers can produce an exothermic effect and the patient can experience a burning sensation. This can be minimized by applying thin layers over top of each other. Adherence is compromised by a hairy or greasy surface. A protective dressing or splint can be applied after the glue is completely dry. The patient should be instructed to keep the area dry for 48 hours and not to apply any ointments, which could cause the glue to peel off prematurely. If the glue must be removed from tissue sooner, acetone can be used.
Methyl Methacrylate
Methyl methacrylate is used to augment fixation of pathologic fractures and to stabilize prosthetic devices in bone. It is an acrylic, cementlike substance commonly referred to as bone cement. It is a drug supplied in two sterile components that must be mixed together immediately before use. One component is a colorless, highly volatile, flammable, liquid methyl methacrylate monomer in an ampule. The powder component may contain antibiotic gentamycin or tobramycin. Studies are being done to determine whether the inclusion of silver nanoparticles could help prevent infection.5 Other products such as dressings and urinary catheters have been using silver successfully for this purpose.
This powerful liquid solvent must be handled carefully. The other component is a white powder mixture of polymethyl methacrylate, methyl methacrylate–styrene copolymer, and barium sulfate in a packet. The barium sulfate provides radiopacity to the substance. When the powder and liquid are mixed, an exothermic polymeric reaction forms a soft, pliable, doughlike mass. This reaction liberates heat as high as 230° F (110° C). As the reaction progresses, the substance becomes hard in a few minutes. The mixing and kneading of the entire contents of the liquid ampule and powder packet must be thorough and should continue for at least 4 minutes. The substance must be adequately soft and pliable for application to bone. The completion of polymerization occurs in the patient. After it hardens, it holds a prosthesis firmly in a fixed position.
A hazard to OR personnel has been reported in regard to the use of methyl methacrylate in the OR. Some personnel have experienced dizzy spells, difficulty breathing, and/or nausea and vomiting after mixing of methyl methacrylate. The fumes can cause severe eye irritation in people wearing gas-permeable contact lenses during the use of this chemical compound. The monomer and several of its ingredients are potent allergenic sensitizers when vapor is inhaled. Disposable mixing bowls with covers and vapor suction ports should be used.
Patients can experience physiologic symptoms when the bone cement is used. Most common problems include hypoxia, hypotension, cardiac dysrhythmia, pulmonary vascular resistance, and cardiac arrest.6 The anesthesia provider must monitor the patient closely and should be alerted to the placement of the cement. The chemicals in the cement can cause many of these symptoms, but other causes including fat embolism have been cited.
Implantable Materials
Tissue deficiencies may require additional reinforcement or bridging material to obtain adequate wound healing. Sometimes edges of fascia, for example, cannot be brought together without excessive tension. In obese or older patients the fascia cannot withstand this tension because of weakness caused by the infiltration of fat. Other implantable materials are used to produce subsurface modifications, such as breast enlargement.
Biologic or synthetic mesh materials are used to fill congenital, traumatic, or acquired defects in fascia or a body wall and to reinforce fascia, as in hernia repair.
Implants must be sterile and compatible with the recipient’s tissues. The surgeon should note any allergies or sensitivities to metallic substances if any are to be used. Care is taken to assure the correct size and handling regardless of the type of implantable used. Powderless gloves are recommended to prevent particulate distribution in the wound.
Administrative controls concerning implantable materials include documentation of the source material, the lot number in case of a recall, and special preparatory handling. The circulating nurse dispenses the implant to the sterile field and the scrub person readies the material for use. Implants should not be handled excessively to prevent damage to the surface or contamination with particulate or lint from the field.
Examples of implant uses and characteristics include but are not limited to:
• Dimensional subsurface space-holding devices, as in tissue expanders
• Load bearing as in orthopedic implants
• Passage creation as in a stent
• Bone integration for increased density
• Flexion with low friction, as in hernia mesh
• Subsurface modification such as testicular implants to replace missing parts
• Mechanical-electrical regulation of a body system such as pacemakers
• Drug delivery devices
Implants can be permanent or temporary and composed of many materials. Any implant that is removed (explanted) is usually sent to the pathology laboratory for accession (gross identification). Refer to Figure 28-20 for a collective comparison of implants for tissue repair and replacement.
Biologic Materials
Cargile Membrane
A thin membrane is obtained from the submucosal layer of the cecum of the ox. Cargile membrane is rarely used, although it is still commercially available in a 4 × 6 inch (10 × 15 cm) sheet to cover peritoneum to prevent adhesions, for isolating ligations, as a covering for packing in submucous nasal resections, and as a dural substitute.
Fascia Lata
Strips of fascia lata are obtained from the fibrous connective tissue that covers thigh muscles of beef cattle. In lieu of commercial fascia lata (allogeneic graft), the surgeon may strip a piece of fascia from the patient’s thigh (an autologous graft). Fascia lata also is obtained from cadavers and freeze-dried (an allogeneic graft). Fascia lata contains collagen. It increases the amount of tissue already present and becomes a living part of the tissue it supports. It is used to strengthen weakened fascial layers or to fill in defects in fascia.
Synthetic Materials
Synthetic Meshes
Synthetic meshes offer several advantages for reinforcing or bridging fascial or other tissue deficiencies:
1. They are easily cut to desired size for the defect.
2. They are easily sutured underneath the edges of tissue to create a smooth surface.
3. They are pliable to preclude erosion into major structures.
5. They are porous to allow free drainage of exudate.
6. Fibrous tissue easily grows through openings to incorporate mesh into tissue to maximize tensile strength.
The manufacturer’s instructions must be followed for each type of mesh product. Unused mesh should be discarded and not reprocessed.
Polyester Fiber Mesh (Mersilene Mesh)
Mesh remains soft and pliable in tissue but has limited elasticity. It is the least inert of the synthetic meshes. It is not preferred in the presence of infection or in contaminated wounds because of its multifilament construction.2 Polyester fibers are knitted by a process that interlocks each fiber juncture to prevent unraveling when cut. However, a minimum of 1 × 4 inch (6.5 mm) of mesh should extend beyond the suture line.
Polyglactin 910 Mesh (Vicryl Mesh)
Mesh is knitted fibers of undyed and uncoated polyglactin 910. Because it is absorbed by hydrolysis, this mesh is intended for use as a buttress to provide temporary support during healing. The mesh acts as a scaffold for ingrowth of connective tissue.2 It may be used to support a traumatized spleen, kidney, or abdominal wall and to support facial fascia. Absorption is essentially complete in 60 to 90 days.
Polypropylene Mesh (Prolene Mesh, Marlex Mesh)
Knitted mesh of polypropylene has high tensile strength and good elasticity. However, Marlex mesh is stiffer and exhibits greater fiber fatigue than does Prolene mesh. Because polypropylene is inert, it may be used in the presence of infection or during healing by second intention. Mesh is used to span and reinforce traumatic abdominal wall defects, incisional ventral hernias, large inguinal hernias, and other fascial deficiencies. It stimulates rapid tissue ingrowth through interstices of mesh. Mesh remains soft and pliable in tissues. It will not unravel when cut.
Polytetrafluoroethylene (Gore-Tex Soft Tissue Patch)
A sheet of expanded PTFE may be used to repair hernias and tissue deficiencies that require prosthetic material. This material is flexible, soft, and porous to allow tissue ingrowth. It is not used in the presence of infection. It must be handled only with clean gloves or rubber-shod forceps. Only Gore-Tex suture should be used.
Tissue Replacement Materials
For centuries surgeons have sought materials to replace parts of anatomy. Tissue may be absent or distorted because of congenital deformity, traumatic injury, degenerative disease, or surgical resection. Replacement or substitution of tissue may be possible with biologic dressings or implanted materials, or with synthetic prosthetic materials implanted in the body. An overview of the types of biologic and synthetic tissue replacement materials is given here. Their uses are referenced in other chapters by surgical specialty or procedure.
Tissue replacement materials may require reconstitution with sterile saline before use. The circulating nurse should record the lot number of the replacement material and the lot number of the saline used for preparation. Avoid the use of saline with preservative when preparing tissue replacement material. Small vials of saline diluent may contain a bacteriostatic preservative and may alter the tissue. IV saline is suitable for this process because it has no preservative.
Biologic Wound Cover
A biologic dressing temporarily covers an open surface defect in skin and underlying soft tissues. Although defects are usually the result of trauma such as burns, vascular or pressure necrosis can cause skin ulcers. Open wounds quickly become contaminated. The dressing arrests loss of fluid, reduces or eliminates microbial growth, and minimizes scarring. It promotes production of granulation tissue and epithelialization before healing by second or third intention. A fibrin-elastin biologic bonding adheres the dressing to exposed surfaces. Biologic dressings are dermal replacements. The source determines the type of dressing.
Autograft
Skin is grafted from one part of the patient’s body to another part. Skin grafts are described in Chapter 40 of this text.
Allograft
Human tissue obtained from one genetically dissimilar person (i.e., unmatched donor) is grafted to another person. This is referred to as an allograft. Negative HIV and hepatitis B virus (HBV) status of donor and recipient should be determined and documented before use. Any natural body tissue transferred from one human to another must be free of infection.
Cryopreserved Skin
Allograft skin provides a protective covering that initially acquires, and then eventually loses, vascular connection with underlying tissue. A cadaver usually is the source of skin for a dermal allograft. Cryopreservation maintains viability of skin during prolonged storage. Skin is frozen by cooling at a rate of 1.8° F to 9° F (1° C-5° C) to –94° F (–70° C) until frozen and then stored in a liquid nitrogen freezer. Immediately before use, skin is warmed by immersion in sterile water at 107.6° F (42° C), the maximum compatible with cellular viability.
Skin should be warmed at a rate of 90° F to 126° F (50° C-70° C) per minute. (The patient’s own skin can be cryopreserved for prolonged storage for later use as an autograft.) Allografts may be obtained from a skin bank.
For storage of allografts or autografts up to 14 days, skin may be placed in isotonic saline solution or tissue nutrient medium and refrigerated at 33.8° F to 50° F (1° C-10° C).
Amniotic Membrane
Prepared from human placenta, amniotic membranes can be used as biologic dressings to promote the healing of burns, skin ulcers, and infected wounds and to cover defects such as spina bifida. The placenta has two loosely connected membranes: amnion is used for partial-thickness wounds, and chorion is used for full-thickness defects.7 Membranes are prepared by cleaning blood and clots from the placenta immediately after delivery, placing the placenta in an iodophor solution, and refrigerating it at 39° F (4° C). Membranes should be stripped from the placenta within 36 hours after delivery. Amnion can be used fresh, preserved, frozen, or dried.7 It may be obtained from a tissue bank that prepares and stores amniotic membranes.
Xenograft
With xenograft, skin obtained from a dissimilar species may be placed on human tissue as a temporary dressing.
Porcine Dermis
Porcine (pig) skin is used to cover body surfaces denuded of full-thickness skin until permanent skin grafting can be accomplished. Vascularization does not occur, but the xenograft adheres tightly while reepithelialization proceeds underneath it. It may remain in place for as long as 2 weeks before it dries up and peels off spontaneously. Porcine biologic dressings are available in rolls or strips. They may be prepared fresh for refrigeration, fresh frozen, irradiated and then frozen, or dried. Some dressings are soaked in an iodophor and should not be placed on a patient allergic to iodine. Dressings must be prepared and used according to the manufacturer’s instructions.
Artificial Skin
A skin substitute may be prepared from a layer of collagen obtained from the dermis of a calf or pig and coated with autologous epithelium obtained from the recipient. Another type is synthesized from a bilayered polymeric membrane. The top layer is silicone elastomer. The bottom layer is a porous, cross-linked network of collagen and glycosaminoglycan. This artificial skin is biodegradable, but it can be used as a temporary covering that is similar to porcine xenografts.
Biologic Materials
Autologous tissues may be grafted or transferred from one part of the patient’s body to another. Allograft tissues or organs may be transplanted from another human. Xenograft biomaterials may be used to supplement tissues.
Standards are set by the American Association of Tissue Banks for screening donors and retrieving, processing, and preserving allogeneic tissues, including skin, cartilage, bone, and blood vessels. Potential donors of allografts are tested for HIV and HBV. Excluded from donating are people who are HIV-positive, have a history of hepatitis, have an active infection, have an immune disorder, or have a suspected prion disease.
Bone Grafts
A bone graft affords structural support and a pattern for regrowth of bone within a skeletal defect. Cancellous bone is porous. Its porosity permits tissue fluid to reach deeper into it than into cortical bone, and thus most of the bone cells live. Cortical bone is used for bridging large skeletal defects, because it gives greater strength. It may be fixed in the recipient site by means of metallic sutures or screws. Bone obtained from the crest of the ilium or a rib is cancellous and cortical bone; cortical bone is obtained from the tibia. The main purpose of a bone graft is to stimulate new bone growth.
Autologous bone, which is obtained from the patient, usually is taken from the ilium, tibia, or ribcage at the time of the surgical procedure. Calvarial bone from the frontal, parietal, or occipital cranial bones may be harvested for maxillofacial bone grafts. A free bone graft with its vascular pedicle, such as a free fibular graft, may be obtained for revascularization by microvascular anastomosis after removal of dead (avascular) bone. A separate, small sterile table may be prepared for the instrumentation required for the donor site. If the recipient area is potentially contaminated, the donor site must not be cross-contaminated from the recipient site.
Allogeneic bone, which is obtained from a cadaver, is dead bone. This bone is weaker than autologous bone, thus requiring a longer time of immobilization. Union occurs from bone regeneration in the recipient with this type of bone graft. It may be desirable, however, to spare the patient the added operating time and trauma of removing an autologous graft.
Composite bone grafts are freeze-dried allografts combined with autologous particulate cancellous bone and marrow. A crib formed from a cadaver bone (e.g., rib) is packed with the patient’s bone particles and marrow. When implanted to reconstruct bony defects, the composite graft induces bone regeneration in the recipient site. The freeze-dried allograft is biodegradable by slow resorption. Eventually it is replaced by mature, functional bone.
Decalcified bone and demineralized bone chips or powder, prepared from homogeneous bone, also are used to stimulate bone regeneration or to fill defects in bone. This material is sterilized and stored at room temperature. For use, it is soaked in Ringer’s lactate solution. The powder then becomes a paste that can be used to fill a depressed area or to caulk an irregularity (e.g., in craniofacial reconstruction).
Bone Bank
Bone may be preserved and stored in a bone bank until needed. Autologous bone may be preserved after the surgical procedure by storage in a bone bank for subsequent grafting into the same patient. Bone such as a rib or femoral head may be salvaged from patients (i.e., living donors) who are free of malignancy or infection for an allogeneic graft into another person. Bone may also be obtained from cadavers (i.e., nonliving donors).
Bone used for allografts must be clean and sterile. Immediately after removal, bone marrow, fat, and blood are rinsed out with sterile distilled water or normal saline solution. The bone may be put in sterile nested glass jars or double plastic or metal containers. If it is to be used for an allogeneic graft, a small piece of bone is put into a sterile Petri dish and sent to the laboratory for culture tests. Bone should never be used until negative results of culture and serology are received. Several methods are used to preserve bone:
• Freezing is the most common method of preserving bone. Bone is quick-frozen in a freezer at –94° F (–70° C) or in liquid nitrogen. If bone will be used within 6 months, it can be stored in a refrigerator freezer at –4° F to –5° F (–20° C to –20.6° C). For prolonged storage of more than 6 months, the freezer temperature must be maintained below –4° F (–20° C) to avoid damage from a buildup of ice crystals. Bone frozen by liquid nitrogen is stored in vapor at about –238° F (–150° C). The container initially is placed on a shelf labeled “Not Ready for Use.” It is labeled “Sterile” and moved to the freezer compartment labeled “Ready for Use” when negative results of culture and serology tests are recorded on the identification card. Bone is thawed rapidly immediately before grafting.
• Freeze-drying requires specialized equipment that removes moisture as the freezing process takes place in a condenser with a vacuum cycle.
• Ethylene oxide sterilization ensures safety of bone. It must be aerated for 72 hours before storage at room temperature or in a refrigerator. When protected from air and contamination, sterilized bone can be stored indefinitely, although a 1-year expiration date is recommended if bone is placed in a heat-sealed, peel-apart package. In lieu of ethylene oxide sterilization, packaged bone may be shipped in dry ice to a center equipped to sterilize it by irradiation.
• Formaldehyde solution, 0.25% to 1% concentration, may have a bacteriostatic effect around the graft site in infected or contaminated wounds, such as in osteomyelitis. During storage, temperature is maintained at 35.6° F to 39° F (2° C-4° C) in a refrigerator.
The container must be labeled with donor information and not used until laboratory reports are available. The donor must be seronegative for hepatitis B surface antigen and HIV. Bone from a living donor is quarantined for 90 days, awaiting results of a repeat test for HIV.
Xenograft Bone Implant
Coralline hydroxyapatite, which is composed of skeletons of sea coral, and collagen may be used to replace facial or cranial bone. This material has hardness, mineral content, and porosity similar to that of human bone. These implants stimulate bone growth into the porous architecture of the coral.
Organ Transplants
Some whole body organs can be transplanted from one human to another. This is done in an effort to sustain life by compensating for physiologic deficits or inadequate function of vital organs. Organ transplants are discussed in Chapter 45 of this text.
Tissue Transplants
Skin and blood vessels are frequently transplanted from one part of the body to another. These are referred to as autografts, because the patient is both donor and recipient. The transplanted tissue becomes a part of the living tissue in the recipient site.
Some tissues can be transplanted from one person to another to restore function, such as the cornea, or to provide support in structures, such as cartilage in nasal reconstruction. These are referred to as allografts. Some allografts are commercially prepared, such as lyophilized human dura mater and human umbilical cord vein graft.
Human Dura Mater
A trimmed and measured piece of cadaver dura mater is freeze-dried, sterilized by exposure to ethylene oxide, and stored in a vacuum container. It may be stored at room temperature indefinitely, provided that the vacuum is maintained. The graft is reconstituted by the addition of normal saline solution to the container for a minimum of 30 minutes. Most of these grafts are used for closure of dural defects, but they may be used also to repair abdominal and thoracic wall and diaphragmatic defects.
Neurologic tissue for transplant may harbor the prion responsible for the development of Creutzfeldt-Jakob disease (CJD). Routine sterilization does not render this material safe for use in the presence of CJD. Routine testing does not reveal CJD contamination.
Human Umbilical Cord Vein Graft
A glutaraldehyde tanning process converts an umbilical vein into an inert, antithrombogenic graft. A polyester mesh covering over the outer surface allows tissue ingrowth and provides added strength. Commercially supplied, an allograft-modified human umbilical vein graft is an acceptable graft material for arterial reconstruction when an autologous saphenous vein is not available.
The glutaraldehyde is thoroughly rinsed from the graft with sterile heparinized IV saline or Ringer’s lactate solution before implantation. A series of three basins filled with sterile solution of choice are used for the rinse process. The basins should be set up on a separate sterile surface, away from the main sterile field, and discarded after use.
After rinsing, the graft should remain in sterile heparinized IV saline solution to keep it moist until implanted. Only noncrushing clamps should be used to avoid damage to the graft during handling.
Xenograft Biomaterials
In addition to porcine skin used as biologic dressings, artificial skin derived from the collagen of a calf or pig, allogeneic bone, and other materials derived from animals are commercially prepared for tissue replacement. Some are supplied in glutaraldehyde solution and require rinsing before use, as previously described.
Arteriovenous Shunts
Enzymatically treated bovine carotid artery xenografts are used for blood access in patients on hemodialysis therapy who have poor blood vessels or in whom it is difficult to create either fistulas or shunts. Femoral arteriovenous bovine shunts can be punctured innumerable times with a low incidence of thrombus formation.
Collagen
Collagen is used in its natural form, such as a processed bovine graft and microfibrillar hemostatic powder, and restructured into membranes or films. It can be injected into middle to deep dermis to fill and smooth nasolabial furrows and facial creases. It is implanted to correct soft tissue defects and contours. The duration of effect is 4 to 6 months. Collagen can be altered by a variety of techniques to change its physical properties and duration of action in tissue.
Corium
Corium implants are prepared from porcine dermis to replace tissue loss or to support tissues. They can be used as a fascia lata substitute or dural replacement or to repair tympanic membrane, hernia, or bladder sling. Corium will form a collagen matrix to close a defect in soft tissues around teeth. Available in sterile sheets of several sizes, corium implant (ZenoDERM) is freeze-dried or air-dried before sterilization by gamma irradiation. It should not be resterilized.
Human Skin Equivalent
Bioengineered skin product, referred to as Apligraf, is derived from bovine collagen and human tissue taken from discarded foreskins of circumcised newborns. The mixture takes 5 days to generate and grow under sterile conditions in the laboratory. It is specially ordered for each patient and cannot be stored for additional uses. The cell culture mixture is applied to nonhealing wounds, such as venous stasis ulcers after debridement. More information can be found at www.dermnetnz.org/procedures/bioengineered-skin.html.
Synthetic Materials
A prosthesis is a permanent or temporary replacement for a missing or malfunctioning structure. Some synthetic implants replace vital structures, such as diseased heart valves and blood vessels. Devices such as pacemakers assist the function of vital organs. Other materials are used to repair or replace defects.
Care is taken to determine whether the patient has an allergy to any metals.8 Some metallic devices can have a nickel component, and patients who are sensitive to nickel can have an augmented inflammatory response to the metal. Prosthetic materials implanted into the body must:
• Be compatible with physiologic processes
• Produce no or minimal tissue reaction
• Be sterile so they will not cause infection or become a culture medium
• Be noncarcinogenic or other disease causative
• Have viable and adequate tissue coverage, unless used as a biologic dressing over denuded skin surfaces
• Have adequate blood supply through or around them
• Be stable so that they will not degenerate or change shape if used for permanent function
• Contour or conform to normal tissue configuration as desired
Permanently implanted devices can provide support, restore function, and augment or restore body contour. Inorganic substances cannot unite with tissue, however. Their physiologic responses may be predictable. All synthetic materials implanted in contact with blood will activate coagulation and promote the process of thrombosis. The surface of some materials is less thrombogenic (i.e., less likely to form clots) than others.
The magnitude of the inflammatory response they stimulate varies in patients. An immune response may cause chronic inflammation from bacterial adhesion, such as biofilm. Infection that develops around a prosthesis usually necessitates its removal. Most infections arise from microorganisms inoculated into the wound at the time of implantation. Therefore meticulous sterile technique is mandatory.
Prosthetic implants must not be flash sterilized. They must be sterilized in a standard cycle for the agent used (see Chapter 17). The cycle should be monitored with a biologic indicator, and a negative biologic test result should be confirmed before the implant is used. Implants sterilized by the manufacturer are preferred because the sterilization controls are closely monitored. Biologic testing is imperative for any implant.
Carbon Fiber
Pure carbon fibers braided into a strip are used for ligament replacement and articular resurfacing. Inert in tissue, carbon fiber stimulates regrowth of connective tissue and cartilage. The fibers may be braided with polypropylene or coated with a resorbable gelatin or lyophilized dura. The prosthesis should be soaked in normal saline solution before implantation to facilitate handling.
Metal
Stainless steel, a cobalt alloy (with the trade name Vitallium), and titanium are manufactured into prosthetic implants. Used primarily for stabilization of bone, metal implants must be strong enough to withstand the stress of weight bearing or muscular action and must not corrode in body tissues. They are never reused because of the weakening that can occur with use.
Special care must be taken in handling metal implants to protect the surfaces. A simple scratch on a metal implant can lead to its corrosion in the body. The implant will be bathed continuously by weakly chloride body fluids. If corrosion begins, the implant may fail and have to be removed. It is very important therefore that all metal implants be protected from scratches. This can be accomplished by:
1. Wrapping each implant individually, or wrapping sets with each size implant (i.e., screws and plates) in a separate compartment, for both storage and sterilization. Most prostheses come from the manufacturer in protective coverings or cases. Some of these are suitable for adequate sterilization, with subsequent placement in the sterile field to minimize handling before implantation.
2. Preventing implants from coming into contact with other hard surfaces of metal or glass, both during storage and sterilization and on the instrument table.
3. Not handling or transferring an unprotected implant with any type of forceps. Implants of one metal should not come into contact with those of another metal because an electrochemical reaction occurs between metals. Two different metals are not implanted in the same patient for this reason. Instruments used for insertion also should be of the same metal as the implant (e.g., a stainless steel screwdriver and screw).
Methyl Methacrylate
A highly refined methyl methacrylate mixture can be molded and shaped to fit a defect in bone. When it hardens, this material looks and feels very much like bone. It is used to repair a skull. In some patients the chemicals in the cement can cause neurotoxic effects.6
Polyester Fiber
Polyester fibers (Dacron) woven or knitted into seamless cylinders are used to replace major arteries.
Polyethylene
Polyethylene tubing may be inserted into structures such as fallopian tubes or ureters to give support during healing or to bridge a defect in tissue continuity. Polyethylene may be combined with silicon to produce a thromboresistant coating for vascular grafts and artificial hearts.
Implants of porous polyethylene are used for anatomic reconstruction, such as of the external ear. The porosity of the implant encourages both soft tissue and vascular ingrowth. Collagen deposited along the framework adds strength. Porous polyethylene is a strong, flexible material that can be molded or shaped to the desired configuration. When dipped into boiling normal saline solution, the material becomes pliable for molding by hand. An implant can be shaped by cutting with a scalpel blade. Glove powder, lint, and dust particles must not adhere to the implant, because they can cause a foreign body reaction around the implant.
Polytetrafluoroethylene (Teflon)
Some prostheses or parts of prosthetic devices are made of the polymer PTFE. It may be woven into a fabric for arterial grafts, extruded into tubing for struts, or molded into a solid configuration for valves or joints. Its flexibility makes it a useful replacement for tissues when motion is desirable.
Silicone
Silicone is one of the most inert of the synthetic polymers used for implantation. It has a durable and nonthrombogenic surface. It is used in many forms: gel, sponge, film, tubing, liquid, and preformed molded anatomic structures. It may be coated with polyurethane or polyester or used as an elastomer to coat polyester. A medical-grade silicone elastomer (Silastic) in one form or another is used in virtually every surgical specialty for tissue reconstruction or replacement. Silicone may migrate from a ruptured or leaking gel- or liquid-filled implant and cause systemic illness.
Complete instructions for cleaning and sterilizing silicone implants before use are supplied by the manufacturer with each type of prosthesis. These instructions must be followed meticulously. Implants are not handled with bare hands, and care must be taken to ensure that they do not pick up lint and dust. Gloves worn during handling must be entirely free of powder. Skin oil, lint, dust, powder, and other surface contaminants can evoke foreign body reactions around the implant in tissue.
Skin Closure
In addition to sutures, staples, and skin glue, other materials may be used to hold skin edges in approximation.
Wound Silo
A sterile, clear plastic, round or tubular cover can be placed over areas of evisceration or dehiscence until the abdomen can be primarily closed. The silo is sometimes referred to as a Bogota bag after the Colombian surgeons who described its use after performing open fasciotomies of the abdomen in compartment syndrome. This device is useful in covering congenital gastroschisis of the abdomen in newborns until a repair can be made.
The silo creates a see-through barrier over the abdomen and viscera as a temporary closure device. It can be used with a wound vacuum dressing. Some facilities use empty sterilized 3-L IV bags sutured or clipped to the skin. Commercial silos are available in adult and pediatric sizes.
Skin Closure Strips
Adhesive-backed strips of microporous nylon (Proxi-Strip) or polypropylene (Steri-Strip) or rayon acetate are placed at intervals across the line of incision. They may be used to approximate skin edges of superficial lacerations, as the primary closure of skin in conjunction with subcuticular suture, or in conjunction with interrupted skin sutures or staples. Often they are used after early suture or staple removal to support the wound during healing. A skin tackifier, such as tincture of benzoin, may be recommended by the manufacturer for ensuring adhesion to skin.
Sterile strips are available in various widths and lengths. They are ethylene oxide gas–sterilized in peel packets by the manufacturer. Skin closure strips have the following advantages:
• They may be used in the emergency department on superficial lacerations to eliminate the need for sutures that would require local anesthesia for placement and subsequent return of the patient for suture removal.
• They eliminate foreign body tissue reaction of suture material in skin.
• They have enough porosity to permit adequate ventilation of clean or contaminated wounds.
• They permit removal of sutures within 32 to 48 hours postoperatively. Crosshatch scarring (referred to as railroad tracks) and the possibility of infection are reduced when sutures are removed early. Skin closure strips provide long-term wound reinforcement and support.
• They permit visibility of the healing wound so that the surgeon can see how well the wound edges have coated. Some strips are translucent; others have a color tone or opacity that does not afford this advantage.
• They minimize skin irritation, because they are hyporeactive.
• They can be applied and removed rapidly.
• They can be easily cut to meet exact length requirements.
The team should keep in mind that the external part of the closure is what the patient sees and on which an opinion of the entire surgical experience is based. The shape of the repaired site and scar formation can lead the patient to believe that surgery was unsuccessful. Proper closure and wound care can help minimize scarring and preserve the patient’s self-esteem.