Male Hypogonadism
The two major functions of the testis are to produce sufficient amounts of testosterone and of sperm to support the development and maintenance of male sexual function, body function, and fertility. Male hypogonadism is a clinical syndrome that results from a failure of the testes to produce adequate amounts of testosterone; this is almost always associated with impaired sperm production (androgen deficiency and impairment of sperm production), or an isolated impairment of sperm production or function with normal testosterone production. Hypogonadism is the most common disorder of testis function encountered in clinical practice.
Because testis function is controlled by the hypothalamus and the pituitary, male hypogonadism may be caused by a primary disorder of the testis (primary hypogonadism); it may be secondary to a disorder of the pituitary or hypothalamus (secondary hypogonadism); or in some instances, there may be defects at both levels (combined primary and secondary hypogonadism).
Identifying men with secondary hypogonadism has important clinical implications that may affect management.113 For example, secondary hypogonadism can be caused by a pituitary adenoma that may be associated with clinical manifestations related to tumor mass (e.g., headaches, visual field defects); to deficiency or excessive secretion of other anterior pituitary hormones; or to diabetes insipidus (polyuria) resulting from hypothalamic antidiuretic hormone deficiency. Such patients require management of the underlying hypothalamic or pituitary disorder in addition to testosterone replacement therapy. Secondary hypogonadism may be reversible with treatment of the underlying condition (e.g., nutritional deficiency) or discontinuation of an offending medication (e.g., glucocorticoids, opioids), or it may be associated with a chronic systemic illness that is not curable, such as chronic kidney disease (CKD). Impaired spermatogenesis and infertility caused by gonadotropin deficiency in men with secondary hypogonadism may be treated with gonadotropin or GnRH therapy, and sperm production and fertility may be restored. In contrast, infertility caused by primary testicular disease is usually not treatable with hormone therapy and requires other fertility options, such as the use of donor sperm, ART (e.g., ICSI), or adoption.
Clinical Manifestations
Androgen Deficiency and Impairment in Sperm Production
Because testosterone has different roles during fetal, prepubertal, and adult life, the manifestations of androgen deficiency differ depending on the stage of sexual development.9,113
Fetal Androgen Deficiency.
During fetal development, testosterone and its conversion to DHT have vital roles in directing male internal and external genital differentiation and development. Fetal androgen deficiency (e.g., from congenital defects in testosterone biosynthesis enzymes) or androgen resistance/insensitivity (e.g., from AR mutations or 5α-reductase deficiency) manifests at birth with varying degrees of ambiguous genitalia and 46,XY DSD (i.e., male pseudohermaphroditism) (Table 19-1).36,62,140 Depending on the severity of androgen deficiency or resistance/insensitivity, the phenotype of individuals with these disorders may range from that of a normal female to that of an otherwise normal male with microphallus, pseudovaginal perineoscrotal hypospadias, bifid scrotum, and/or cryptorchidism of varying severity. These disorders are described in greater detail in Chapter 23.
TABLE 19-1
Clinical Manifestations of Androgen Deficiency
| Fetal Androgen Deficiency | |
| Symptoms | Signs |
| Ambiguous genitalia | Ambiguous genitalia (46,XY DSD) |
| Normal female genitalia | |
| Microphallus (resembling clitoromegaly) | |
| Pseudovaginal perineoscrotal hypospadias | |
| Bifid scrotum | |
| Cryptorchidism | |
| Prepubertal Androgen Deficiency | |
| Symptoms | Signs |
| Delayed puberty | Eunuchoidism |
| Lack of sexual interest or desire (libido) | Infantile genitalia |
| Reduced nighttime or morning spontaneous erections | Small testes |
| Breast enlargement and tenderness | Lack of male hair pattern growth, no acne |
| Reduced motivation and initiative | Disproportionately long arms and legs relative to height |
| Diminished strength and physical performance | Pubertal fat distribution |
| No ejaculate or ejaculation (spermarche) | Poorly developed muscle mass |
| Inability to father children (infertility) | High-pitched voice |
| Reduced peak bone mass, osteopenia or osteoporosis | |
| Gynecomastia | |
| Small prostate gland | |
| Aspermia, severe oligozoospermia or azoospermia | |
| Adult Androgen Deficiency | |
| Symptoms | Signs |
| Incomplete sexual development | Eunuchoidism |
| Lack of sexual interest or desire (libido) | Small or shrinking testes |
| Reduced nighttime or morning spontaneous erections | Loss of male hair (axillary and pubic hair) |
| Breast enlargement and tenderness | Gynecomastia |
| Inability to father children (infertility) | Aspermia or azoospermia or severe oligozoospermia |
| Height loss, history of minimal-trauma fracture | Low bone mineral density (osteopenia or osteoporosis) |
| Hot flushes, sweats | Height loss, minimal-trauma or vertebral compression fracture |
| Reduced shaving frequency | Unexplained reduction in prostate size or PSA |
| Less Specific Symptoms | Less Specific Signs |
| Decreased energy, vitality | Mild normocytic, normochromic anemia (normal female range) |
| Decreased motivation, self-confidence | Depressed mood, mild depression or dysthymia |
| Feeling sad or blue, irritability | Reduced muscle bulk and strength |
| Weakness, decreased physical or work performance | Increased body fat or body mass index |
| Poor concentration and memory | Fine facial skin wrinkling (lateral to orbits and mouth) |
| Increased sleepiness | |
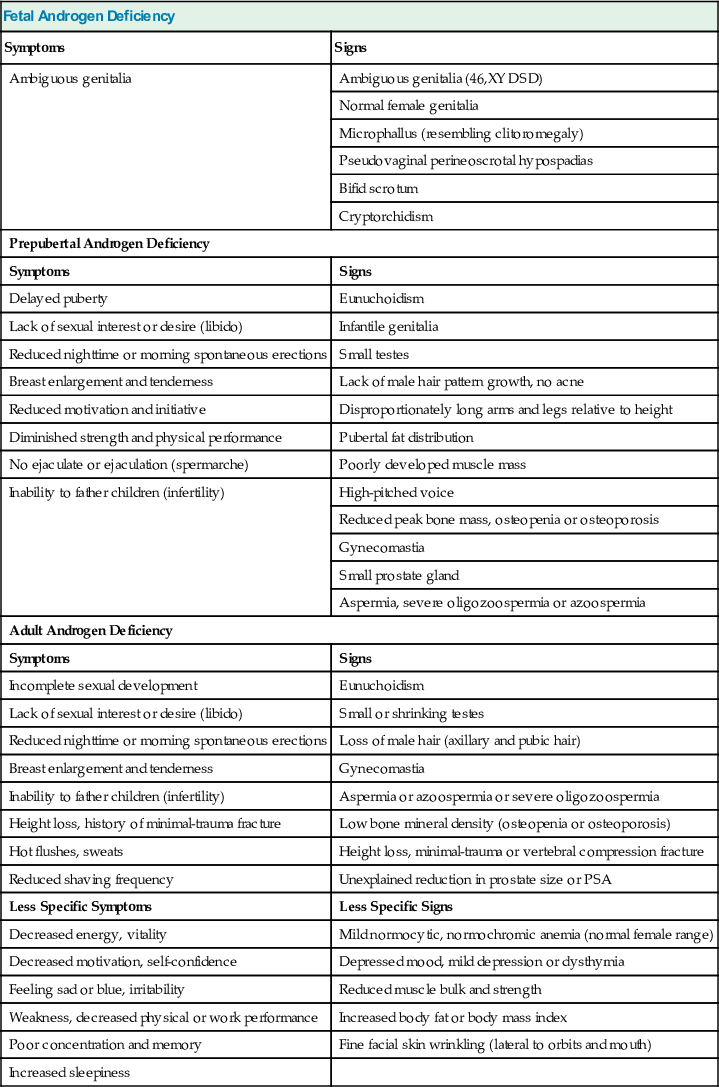
Prepubertal Androgen Deficiency.
The increase in testosterone levels that occurs at the time of puberty is responsible for development of secondary sexual characteristics; an increase in muscle mass and reduction and redistribution of body fat; long bone growth and eventually closure of epiphyses resulting in cessation of growth; stimulation of sexual interest (libido), spontaneous erections, and sexual activity; and initiation of spermatogenesis and seminal fluid production.141 Prepubertal androgen deficiency causes eunuchoidism (Fig. 19-14; see Table 19-1),141 which is characterized most notably by infantile genitalia with a small penis and a poorly developed scrotum that lacks rugal folds and pigmentation. The testes are small, usually less than 2 cm in length and from 2 mL to less than 4 mL in volume. Hair is thin and fine, and there is a lack of androgen-dependent hair growth (i.e., absence of a male hair pattern in all body areas) and no temporal hair recession. The pubic hair pattern is more typical of females, with the shape of an inverted triangle in the pubic area (female escutcheon) rather in a diamond shape with hair extending from the pubic area to the umbilicus (male escutcheon), and there is little hair extending to the thighs. Acne does not develop because sebum production is not stimulated by androgens.
Eunuchoidism is typified by a distinctive body habitus, characterized by poor muscle mass development (especially in the shoulders and chest), prepubertal fat distribution (predominantly in the face, chest, and hips), and excessively long arms and legs relative to height. Arm span exceeds height by greater than 5 cm, and the distance from the crown of the head to the symphysis pubis is less than 5 cm than the distance from the symphysis to the floor. The voice is high-pitched in the absence of androgen-dependent laryngeal enlargement and vocal cord thickening. Relatively long arms and legs result from a failure of long bone epiphyses to close; epiphyseal closure is mediated normally by increased estradiol derived from aromatization of the increased testosterone produced at the time of puberty.
Prepubertal androgen deficiency may not be recognized or diagnosed until adulthood. Compromise in peak bone mass accrual due to androgen deficiency may manifest as low BMD for age, and prolonged severe androgen deficiency increases the risk of osteoporosis and fractures as these men become older. Despite the absence of pubertal development, these individuals may develop gynecomastia (benign breast enlargement) that is caused by androgen deficiency rather than by the relatively high ratio of estradiol to testosterone levels associated with pubertal gynecomastia. Motivation and initiative are reduced and, together with poor muscle mass and strength, may contribute to poor physical performance (e.g., in athletics or the military). These men have reduced sexual interest or desire (libido) and lack spontaneous erections at night or on awakening in the morning. Hematocrit remains in the female range due to inadequate androgen stimulation of erythropoiesis. The prostate and seminal vesicles remain small without androgen stimulation, and seminal fluid production is absent, resulting in aspermia (lack of ejaculate) and failure to undergo spermarche (first ejaculation). Seminal fluid may be present in men with mild or partial prepubertal androgen deficiency or in those treated with androgens. However, these men usually have severe oligozoospermia or azoospermia, and most are infertile.
Adult Androgen Deficiency.
Some individuals with prepubertal androgen deficiency who are not diagnosed or are inadequately treated as boys present as adults with features of eunuchoidism and other manifestations of prepubertal androgen deficiency (see Table 19-1). Their condition is usually clinically obvious because of inadequate sexual development for their chronologic age.
In adults, testosterone is needed to maintain sexual function, some secondary sexual characteristics, muscle and bone mass, and sperm production. Clinical manifestations of androgen deficiency are nonspecific and may be modified by the severity and duration of androgen deficiency, the presence of comorbid illnesses, previous testosterone treatment, or variations in target-organ sensitivity to androgens. Therefore, the clinical diagnosis of androgen deficiency acquired as an adult can be challenging, particularly in older men.113
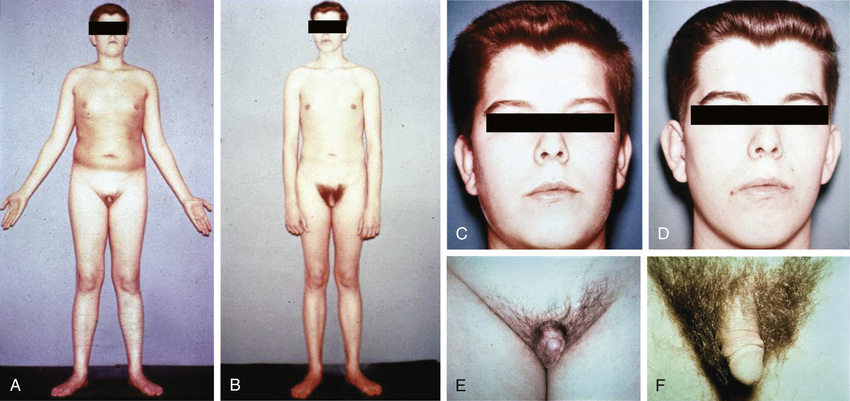
Some clinical symptoms and signs are suggestive of androgen deficiency. Adults most commonly present with sexual dysfunction (diminished libido as manifested by reduced sexual interest or desire, reduced spontaneous and sexually evoked erections, and erectile dysfunction); gynecomastia (benign breast enlargement that may be accompanied by tenderness); and infertility (inability to father children despite unprotected intercourse) associated with oligozoospermia or azoospermia and small or shrinking testes with severe impairment in spermatogenesis. Secondary sexual characteristics do not regress to a prepubertal state; however, with long-standing, severe androgen deficiency, there may be loss of androgen-dependent hair, such as reduced facial hair associated with reduced shaving frequency and loss of axillary and pubic hair (Fig. 19-15). Men with rapid and profound decreases in testosterone levels (e.g., from GnRH agonist treatment of prostate cancer) may have hot flushes and sweats due to vasomotor instability, similar to those experienced by menopausal women. Because testosterone and its active metabolite, estradiol, have an important role in the maintenance of bone mass, men with chronic androgen deficiency may present with osteopenia or osteoporosis on BMD measurement (e.g., by dual energy x-ray absorptiometry [DXA] scan) or with a minimal-trauma bone or vertebral compression fracture that may be associated with height loss. An unexplained reduction in prostate size or in the level of prostate-specific antigen (PSA) is uncommon but may occur as a result of long-term, severe acquired androgen deficiency.
Other symptoms and signs are much less specific for androgen deficiency but may occur, commonly in conjunction with clinical manifestations described previously that are more suggestive of androgen deficiency. Men with low testosterone concentrations often complain of diminished energy and joie de vivre (vitality), poor motivation and social aggressiveness, depressed mood and irritability that may be diagnosed as subsyndromal depression (mild depression or dysthymia), increased sleepiness, or poor concentration and memory. Men with severe androgen deficiency may have a mild hypoproliferative normocytic, normochromic anemia within the female range in the absence of androgen stimulation of erythropoiesis. With long-standing deficiency, reduced muscle bulk and strength associated with weakness and reduced physical and work performance may occur. The latter symptoms may occur in conjunction with an increase in body fat, but androgen deficiency is not a cause of clinically obvious obesity per se. Skin changes and reduced sebum production with severe, long-standing androgen deficiency may be associated with fine facial wrinkling that is particularly noticeable on the lateral corners of the orbits (lateral canthus) and mouth. Testis size may be small, especially with severe impairment of spermatogenesis, but in most men with acquired adult androgen deficiency, testis size is normal to slightly reduced.
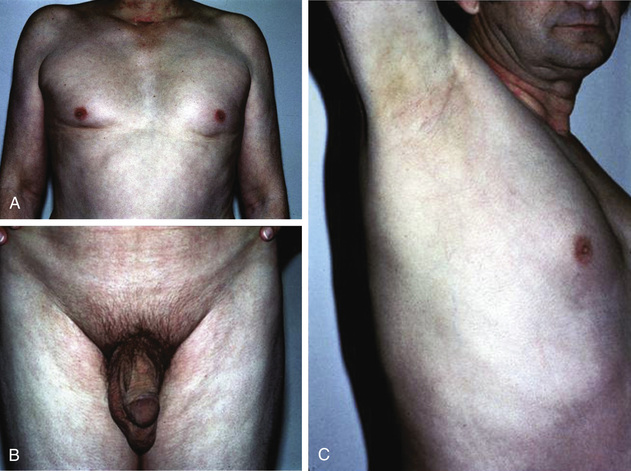
Because clinical manifestations are nonspecific, older men may have a number of medical or comorbid conditions and medications that contribute to symptoms and signs that are consistent with androgen deficiency, presenting a particular diagnostic challenge (Fig. 19-16). Symptoms and signs of comorbid illnesses may mask, mimic, or contribute to clinical manifestations of androgen deficiency in older men. Elderly men may present with muscle loss and mobility impairment, fragility fracture or osteoporosis, and reduced vitality and depressed mood. On close examination, however, older men with severe, long-standing androgen deficiency usually manifest objective evidence of androgen deficiency.
Isolated Impairment of Sperm Production or Function
Most men with male infertility have hypogonadism manifested by an isolated impairment of sperm production with normal androgen production. These men present as adults with infertility and demonstrate oligozoospermia or azoospermia, sperm with abnormal morphologic appearance (teratospermia) or reduced or absent motility (asthenospermia), or a combination of abnormalities on seminal fluid analysis. They do not have manifestations of androgen deficiency, and serum testosterone concentrations are normal. Testes may be small (if spermatogenesis is severely impaired) or normal sized. Testes may not be palpable if cryptorchidism or anorchia is present.
History and Physical Examination
Clinical evaluation of male hypogonadism involves a careful history and physical examination directed at determining whether there are symptoms and signs of androgen deficiency or isolated impairment of sperm production and at identifying potential common causes of hypogonadism.9 Because adults with androgen deficiency present commonly with sexual dysfunction, gynecomastia, and infertility, the differential diagnosis of these conditions and causes other than hypogonadism of these presenting complaints should be considered. Laboratory evaluation of serum testosterone, gonadotropins, and seminal fluid (in men who are concerned with infertility) are performed to confirm the diagnosis of hypogonadism and to determine whether there is predominantly primary or secondary hypogonadism.
The history should include inquiry regarding symptoms of androgen deficiency. These may be grouped as relating to several areas:
1. Development: genital abnormalities and the potential need for surgical correction (e.g., hypospadias, microphallus, cryptorchidism); delayed sexual development or growth and need for hormone therapy; family history of delayed puberty or reproductive disorders; psychological impact of delayed puberty or growth; difficulty in school or learning disability; inability or reduced ability to smell
2. Sexual function: poor erections; reduced spontaneous, nighttime, or morning erections; inability to perform sexually; decreased sexual activity; inability to father children despite unprotected sexual intercourse (>1 year); small or shrinking testes
3. Brain function: poor general well-being; reduced sexual desire, interest, and motivation (libido); poor energy and vitality and excessive fatigue; poor motivation and initiative, passivity, low self-confidence, and low self-esteem; depressed mood and irritability; difficulty sleeping; hot flushes and sweats; poor concentration and memory
4. Body function: decreased muscle bulk and strength; reduced physical activity or performance; breast enlargement or tenderness, especially if recent in onset; height loss, history of low-trauma or vertebral compression fractures, osteopenia, or osteoporosis; body hair loss (chest, axillary, or pubic); reduced beard growth and shaving frequency
The initial history may also include inquiry concerning the potential cause of hypogonadism. With primary hypogonadism, there may be a history of mumps involving the testes; testicular trauma, irradiation, or surgery; medication use (spironolactone, ketoconazole, cytotoxic agents); or chronic liver or kidney failure. With secondary hypogonadism, headaches, visual complaints or reduced peripheral vision, history of pituitary disease, chronic lung disease or congestive heart failure (CHF), wasting conditions (e.g., acquired immunodeficiency syndrome [AIDS], cancer), nutritional deficiencies, recent acute illness, morbid obesity, or use of certain medications (e.g., opioids, CNS-active drugs, glucocorticoids, anabolic steroids, megestrol acetate, medroxyprogesterone acetate, nutritional supplements) may be noted.
The patient should be questioned regarding conditions that are relative or absolute contraindications to testosterone treatment, including a history of severe BPH and lower urinary tract symptoms as assessed by the American Urological Association (AUA) symptom score or International Prostate Symptom Score (IPSS), history of prostate or breast cancer, history or symptoms of untreated obstructive sleep apnea syndrome (daytime sleepiness, snoring with sleep disruption, witnessed apnea episodes), history of severe CHF, and polycythemia or hyperviscosity.
In patients with suspected prepubertal androgen deficiency, physical examination should include measurements of total arm span, height, and the distances from the crown of the head to the symphysis pubis and from the symphysis pubis to the floor to determine whether the patient has excessively long arms and legs (see Fig. 19-14). Eunuchoidal body proportions are characterized by an arm span that is at least 5 cm greater than height and a crown-to-symphysis distance that is at least 5 cm less than the symphysis-to-floor distance; such proportions are indicative of prepubertal androgen deficiency. Men with Klinefelter syndrome may have disproportionately long legs relative to arms and a greater ratio of lower- to upper-body segment measurements but a relatively normal ratio of arm span to height. Eunuchoidism is also characterized by infantile genitalia (micropenis or small penis, unrugated and nonpigmented scrotum); small testes or, rarely, absence of the testes; cryptorchidism; sparse or absent facial, axillary, chest, extremity, and pubic hair; poorly developed upper body musculature; fat predominance in the face, chest, and hips; and gynecomastia. Patients with Kallmann syndrome may have anosmia or hyposmia that may be tested with an odor identification and threshold test using readily identifiable, common household odorants (e.g., alcohol swab, peppermint, cinnamon, cocoa, coffee, cigarette, orange, soap) or more formally, such as with the University of Pennsylvania scratch and sniff test.
The physical findings of androgen deficiency acquired in adulthood are usually subtler than those of prepubertal androgen deficiency (see Fig. 19-15). In patients with severe, long-standing adult androgen deficiency, there may be loss of androgen-dependent facial, axillary, chest, extremity, and pubic hair; however, there are ethnic variations in body hair in androgen-dependent areas (e.g., less in Asians and Hispanics). The skin may be dry, and there may be fine wrinkling lateral to orbits or mouth in patients with severe, long-standing androgen deficiency. Patients should be carefully examined for the presence of palpable breast tissue or gynecomastia; presence, size, and consistency of the testes; and palpable abnormalities in the scrotum, such as varicocele, epididymal enlargement, or tenderness or absence of the vas deferens. A digital rectal examination (DRE) should be performed primarily to determine whether there are palpable abnormalities, such as a prostate nodule or induration, and also to assess the size of the prostate. Careful examination for kyphosis and measurement of height are useful for detecting significant height loss (>5 cm) associated with osteoporotic vertebral compression fractures that may be asymptomatic.
Proper technique is needed to examine the male breast. The thumb and index finger are used to grasp and gently pinch the periareolar area of the breast and to palpate glandular breast tissue, which is rubbery in consistency and firmer than the surrounding adipose tissue (Fig. 19-17). With this technique, gynecomastia can usually be distinguished from excessive breast adipose tissue, called pseudogynecomastia, which is often associated with generalized obesity. Gynecomastia is usually bilateral and relatively symmetric, but occasionally it is asymmetric and more prominent on one side. If present, asymmetric gynecomastia may suggest breast carcinoma, which is usually rock-hard and irregular and may be associated with skin dimpling (peau d'orange), nipple retraction or discharge, and axillary lymphadenopathy. The diameter of palpable breast tissue is used as an objective measure of gynecomastia. Gynecomastia of recent onset is usually tender on palpation, and men usually complain of nipple irritation associated with rubbing against clothing.
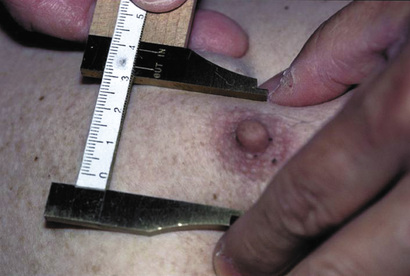
Examination of the testes and scrotum may be performed with the patient either lying on his back or standing, but the latter position is preferred because it relaxes the scrotum, making some abnormalities (e.g., varicocele) more easily detected. In patients with retractile testes positioned high in the scrotum, it may be possible to palpate the testes only after placing the scrotum in warm water, after a warm bath, or by having the patient assume a squatting position. The testes may be very difficult to examine and palpate in morbidly obese men who have excessive folds of fat overlying the scrotum, in the presence of a large hydrocele, if the testis is tender (e.g., with epididymo-orchitis or testicular torsion), or occasionally in some men who are sensitive to palpation for unclear reasons. In these instances, testicular ultrasound may be required to confirm the presence of the testis, estimate its size, and detect abnormalities.
Although ultrasonographic size estimates are more accurate, testis size can be estimated by measuring length and width with a ruler or calipers or by comparing testis volume with that of ellipsoid models of known volume (Prader orchidometer) (Fig. 19-18). Normal testis size varies with age and ethnicity. Normal prepubertal testis size is approximately 1.6 to 2.9 cm in length and 1.0 to 1.8 cm in width, or 1 to 4 mL in volume. Testis size greater than 4 mL suggests the onset of puberty. In adults, normal testes usually measure 3.5 to 5.5 cm in length and 2.0 to 3.0 cm in width or 15 to 30 mL in volume.8,9 In addition to size, testes should be palpated for consistency or firmness and for presence of a mass representing a benign or malignant testicular tumor. The testicular examination in men with Klinefelter syndrome is notable for very small (usually <3 mL), firm testes.
Differential Diagnosis
Because sexual dysfunction, gynecomastia, and infertility are often presenting complaints in adults with androgen deficiency, it is important to consider the differential diagnosis of these conditions and to be familiar with other common causes of these manifestations when evaluating men who present with these complaints.
Sexual Dysfunction
Normal sexual function requires successive, coordinated physiologic events—libido, erection, ejaculation, orgasm, and detumescence—that occur in a defined sequence and require normal psychological, CNS, peripheral nerve, vascular, and genital function.142
Sexual dysfunction may involve specific disorders of libido or sexual desire, erectile dysfunction, ejaculatory disorders, orgasmic dysfunction, or failure of detumescence. These may occur in isolation, but specific disorders of sexual function commonly occur together because these processes are interrelated and because a specific cause (e.g., androgen deficiency) can affect more than one of the physiologic processes that mediate normal sexual function. Male sexual dysfunction is detailed in Chapter 20. Men with androgen deficiency often present with sexual dysfunction, and it is important to consider the differential diagnosis of this complaint in the evaluation.
Androgen deficiency often results in reduced libido or sexual desire (hypoactive sexual desire disorder), loss or reduction of spontaneous evening and morning or sexually stimulated erections (erectile dysfunction), and, if severe, reduced or absent ejaculation. In many men with androgen deficiency, erectile response to intense erotic stimuli (and, occasionally, spontaneous erections) may be preserved, suggesting that the androgen requirement for sexual function is variable.143 However, persistent erectile dysfunction may cause performance anxiety, and, together with hypoactive sexual desire and depressed mood associated with androgen deficiency, this may contribute to the eventual loss of erotically stimulated erections and, secondarily, to orgasmic dysfunction. Androgen deficiency may also affect nitric oxide (NO) production and maximal smooth muscle relaxation and vasodilatation within the penis, reducing the ability to produce an erection that is sufficient to satisfactorily complete sexual intercourse and further contributing to the severity of erectile dysfunction.144,145
Clinically, men with androgen deficiency most commonly present with hypoactive sexual desire disorder and erectile dysfunction. Severely androgen-deficient men may present with reduced ejaculation, but these individuals usually also complain of hypoactive sexual desire disorder and erectile dysfunction.
Hypoactive Sexual Desire Disorder and Erectile Dysfunction.
Libido, the desire or drive for sexual activity, is generated by external visual, auditory, and tactile stimuli as well as internal psychic stimuli acting on cortical and subcortical brain regions such as the limbic system (amygdala, hippocampus, anterior thalamic nuclei, and prefrontal cortex) and the temporal lobe. Stimuli from these areas are relayed to the medial preoptic area, which serves to integrate central inputs and sends impulses to the paraventricular nuclei; these, in turn, send projections to the thoracolumbar and sacral spinal cord centers that regulate penile erection. This neural pathway explains why brain disorders that cause hypoactive sexual desire disorder are usually accompanied by varying degrees of erectile dysfunction (see later discussion).142 In particular, there is a loss of the spontaneous evening and morning erections that are associated with brain activation of sexual neural pathways during rapid eye movement (REM) sleep and dreaming. Clinically, libido may be influenced by previous or recent sexual activity and by experiences, psychosocial background, overall state of general health, androgen sufficiency, and brain function.
The neurotransmitter systems that regulate the physiology of normal libido are not known precisely. However, there is evidence that central dopamine neurotransmission may be important in mediating CNS regulation of sexual desire and erections. In humans, treatment with dopamine receptor agonists (e.g., bromocriptine, pergolide) may stimulate spontaneous erections, and in 20% to 30% of men with Parkinson disease, levodopa therapy is associated with stimulation of libido and spontaneous erections. The use of pharmacologic agents with dopamine receptor antagonist activity is frequently associated with reduced libido and erectile dysfunction. However, these agents also affect a number of other neurotransmitter systems. Dopamine antagonism (e.g., by neuroleptic or antipsychotic agents) results in elevated prolactin levels that suppress endogenous gonadotropin and testosterone secretion and may contribute to reduced libido and erectile dysfunction.
Hypoactive sexual desire disorder is defined as persistent or recurrent deficiency or absence of desire for sexual activity resulting in marked personal distress or interpersonal difficulty or both.142,146,147 It is estimated to affect more than 15% of men. The causes of hypoactive sexual desire disorder are primarily disorders that affect normal brain function and are usually associated with erectile dysfunction, in particular loss of spontaneous evening or morning erections (Table 19-2). Erectile dysfunction is defined as the inability to achieve or maintain penile erection that is adequate for completion of satisfactory sexual intercourse or activity.148 Erectile dysfunction is a common condition that increases with aging. It is estimated to affect fewer than 10% of men younger than 40 years of age but approximately 50% of men between 40 and 70 years of age, with 35% of men in the latter age group having moderate or complete erectile dysfunction.
TABLE 19-2
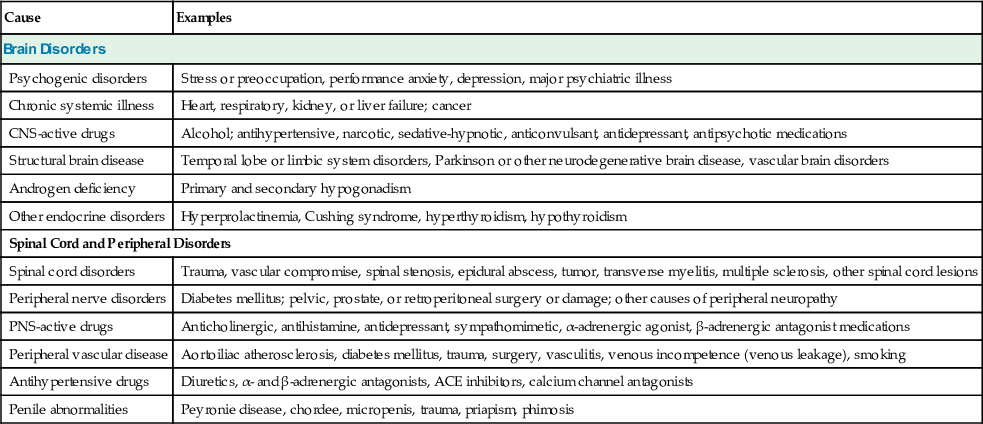
Causes of Hypoactive Sexual Desire Disorder and Erectile Dysfunction
| Cause | Examples |
| Brain Disorders | |
| Psychogenic disorders | Stress or preoccupation, performance anxiety, depression, major psychiatric illness |
| Chronic systemic illness | Heart, respiratory, kidney, or liver failure; cancer |
| CNS-active drugs | Alcohol; antihypertensive, narcotic, sedative-hypnotic, anticonvulsant, antidepressant, antipsychotic medications |
| Structural brain disease | Temporal lobe or limbic system disorders, Parkinson or other neurodegenerative brain disease, vascular brain disorders |
| Androgen deficiency | Primary and secondary hypogonadism |
| Other endocrine disorders | Hyperprolactinemia, Cushing syndrome, hyperthyroidism, hypothyroidism |
| Spinal Cord and Peripheral Disorders | |
| Spinal cord disorders | Trauma, vascular compromise, spinal stenosis, epidural abscess, tumor, transverse myelitis, multiple sclerosis, other spinal cord lesions |
| Peripheral nerve disorders | Diabetes mellitus; pelvic, prostate, or retroperitoneal surgery or damage; other causes of peripheral neuropathy |
| PNS-active drugs | Anticholinergic, antihistamine, antidepressant, sympathomimetic, α-adrenergic agonist, β-adrenergic antagonist medications |
| Peripheral vascular disease | Aortoiliac atherosclerosis, diabetes mellitus, trauma, surgery, vasculitis, venous incompetence (venous leakage), smoking |
| Antihypertensive drugs | Diuretics, α- and β-adrenergic antagonists, ACE inhibitors, calcium channel antagonists |
| Penile abnormalities | Peyronie disease, chordee, micropenis, trauma, priapism, phimosis |
Hypoactive Sexual Desire Disorder and Erectile Dysfunction Due to Brain Disorders.
Psychogenic disorders commonly cause hypoactive sexual desire and erectile dysfunction; these disorders include stress or preoccupation associated with life circumstances or situations, illness, marital discord, or underlying maternal transference or gender identity issues; performance anxiety associated with fear of failure or preoccupation with the adequacy of erections during sexual intercourse; major depression or dysthymia (moderate or complete erectile dysfunction occurs in 60% to 90% of men with moderate to severe depression); and major psychiatric illness such as psychotic or personality disorders.142,146,147
Chronic systemic illness (chronic heart disease, respiratory illness, kidney or liver failure, or cancer) and poor general health are usually associated with reduced libido and spontaneous erections.142,146,147 A number of CNS-active medications may cause hypoactive sexual desire disorder and erectile dysfunction, including alcohol, centrally acting antihypertensive medications, narcotics, sedative-hypnotic drugs, anticonvulsants, antidepressants, and antipsychotic medications. In addition to their direct effects on brain neurotransmitter function, both chronic illness and CNS-active medications may also be associated with androgen deficiency. Structural brain disease, such as infiltrative or destructive lesions of the temporal lobe or limbic system, Parkinson or other neurodegenerative brain disease, or vascular brain disorders such as stroke or vasculitis, may reduce libido and spontaneous erections.
Androgen deficiency is commonly associated with reduction or loss of libido and spontaneous erections.149,150 Sexual dysfunction is usually a prominent presenting complaint in young, severely androgen-deficient men and in older men who are treated with medical therapies (e.g., GnRH agonist treatment) or surgical castration for advanced prostate cancer. In contrast, older men with less severe androgen deficiency may have sexual dysfunction that is also related to underlying depression, chronic systemic illness, or use of certain medications.151 Comorbid conditions contribute to the nonspecificity of presenting complaints of androgen deficiency (e.g., sexual dysfunction) as men age. Testosterone treatment of severe androgen deficiency in young men usually improves sexual desire, interest, and thoughts; attentiveness to erotic stimuli; and the frequency, duration, and rigidity of spontaneous evening and morning erections.150,152,153
Other endocrine disorders can cause hypoactive sexual desire disorder and erectile dysfunction; examples include hyperprolactinemia, Cushing syndrome (glucocorticoid excess), hyperthyroidism, and hypothyroidism. In addition to their direct effects on brain function, hyperprolactinemia and glucocorticoid excess also suppress GnRH and gonadotropin secretion and induce androgen deficiency that contributes to sexual dysfunction. Anecdotally, some men with androgen deficiency due to severe hyperprolactinemia who are treated with testosterone alone do not fully recover sexual function and may require additional therapy with dopamine receptor agonists, but this has not been demonstrated conclusively. Dopamine receptor agonists lower elevated prolactin levels and may also have direct affects in the brain to activate neuronal systems involved in stimulating libido and erections.
Erectile Dysfunction Due to Spinal Cord or Peripheral Disorders.
External and internal erotic stimuli from the brain are relayed via descending neural pathways in the lateral spinal columns to stimulate the parasympathetic sacral (S2-S4) spinal erection center, resulting in psychogenic erections. Efferent parasympathetic nervous system stimulation from the sacral center travels via the nervi erigentes (pelvic splanchnic nerve) and the pelvic plexus and enters the penis via the cavernosal nerve. This stimulation causes relaxation of the smooth muscles that form sponge-like interconnected trabecular spaces within the corpora cavernosa of the penis and vasodilation of the cavernosal arterioles and vascular sinusoids. As a result, blood flow and pressure into the trabecular spaces within the corpora increase several-fold and cause engorgement of the penis (tumescence). Expansion of the trabecular spaces against the thick fibrous sheath (tunica albuginea) surrounding the corpora compresses subtunical venules and impedes venous outflow, resulting in sustained penile tumescence (i.e., an erection).142,148
Afferent somatic (via the pudendal nerve) and parasympathetic impulses in response to sensory stimulation of the penis with sexual intercourse or masturbation also act to stimulate erections via a reflex arc through the sacral spinal erection center, resulting in reflexogenic erections. Pudendal nerve stimulation also triggers the reflex contraction of the ischiocavernosus and bulbocavernosus muscles, resulting in vascular compression at the base of the penis, further increasing cavernosal blood pressure and maximal penile rigidity, leading to the plateau phase of erection.
The primary neurotransmitter that mediates penile smooth muscle relaxation and erection is NO. In response to parasympathetic cholinergic (acetylcholine-mediated) stimulation, NO is synthesized from its precursor, L-arginine, by the enzyme nitric oxide synthase (NOS) and is released by corporal sinusoidal endothelial cells and postganglionic noncholinergic, nonadrenergic nerve terminals. NO then enters adjacent smooth muscle cells, where it activates guanylate cyclase and increases intracellular cyclic guanosine monophosphate (cGMP). cGMP activates cGMP-dependent protein kinase, which phosphorylates a number of proteins, including myosin light chains and ion channels that ultimately decrease intracellular calcium concentrations, causing smooth muscle relaxation, increase in penile blood flow, and erection. cGMP is hydrolyzed and inactivated by the enzyme, phosphodiesterase type 5 (PDE5). In addition to cGMP, other neurotransmitters induce cavernosal smooth muscle relaxation, including prostaglandin E1 (PGE1), which activates adenylate cyclase and increases cAMP and cAMP-dependent protein kinase.
Knowledge of the neurotransmitter systems that control erections has been used to design pharmacologic treatments for erectile dysfunction (detailed in Chapter 20).148,154,155 The most commonly used treatments are oral PDE5 inhibitors, such as sildenafil, vardenafil, and tadalafil, which act to inhibit the breakdown of cGMP, resulting in more sustained smooth muscle relaxation and improved penile erection after erotic stimulation. Injection of intracavernosal PGE1 or insertion of intraurethral PGE1 pellets acts to increase cavernosal cAMP concentrations and induce smooth muscle relaxation and penile erection even in the absence of sexual stimulation. Intracavernosal injections of papaverine, a nonspecific phosphodiesterase inhibitor (which inhibits the breakdown of both cGMP and cAMP), combined with phentolamine, an α1- and α2-adrenergic receptor antagonist vasodilator (bi-mix), or the two combined together with PGE1 (tri-mix), are also used to induce smooth muscle relaxation and erection.
Studies in experimental animals and in vitro have found that androgen deficiency impairs penile nerve, trabecular smooth muscle, vascular endothelial, and tunica albuginea structure and function; reduces both endothelial and neuronal NOS synthesis and activity; and causes accumulation of adipocytes in the subtunical region of the corpora cavernosa.145 These changes are reversed with androgen administration, suggesting a direct penile effect of androgens in addition to their central role in maintaining penile erections. In humans, ARs are expressed in the corpora cavernosa tissue. However, there is no conclusive evidence to support the notion that androgen treatment has a direct effect in the penis to enhance the response to PDE5 inhibitor therapy in androgen-deficient men with erectile dysfunction. In practice, symptomatic men with androgen deficiency and sexual dysfunction are usually treated with testosterone replacement, which can variably improve erectile dysfunction, particularly in younger hypogonadal men with severe androgen deficiency and no comorbid illness. If erectile dysfunction does not improve with testosterone treatment alone, they are given additional therapy for erectile dysfunction (e.g., addition of a PDE5 inhibitor). In some hypogonadal men, PDE5 inhibitor treatment alone may be sufficient to improve erectile dysfunction but is not adequate to treat reduced libido or other symptoms of androgen deficiency.153
In addition to the brain disorders that cause hypoactive sexual desire disorder and erectile dysfunction, spinal cord and peripheral disorders (e.g., peripheral nervous system disorders, peripheral vascular disease, medications that affect peripheral nerve and vascular function, penile abnormalities) may also cause erectile dysfunction that is usually not associated with hypoactive sexual desire (see Table 19-2). However, long-standing, severe erectile dysfunction may cause performance anxiety or depression, which may secondarily reduce libido. Furthermore, peripheral disorders that cause erectile dysfunction may also affect brain function and alter sexual interest and drive, contributing to erectile dysfunction. Tricyclic antidepressants may affect both peripheral and CNS function.
Spinal cord disorders, such as spinal cord injury due to trauma, vascular compromise, spinal stenosis, epidural abscess, tumor, transverse myelitis, multiple sclerosis, or other spinal cord lesions, usually cause erectile dysfunction. In general, the severity of erectile dysfunction associated with spinal cord injury and the response to treatment vary with the cord level involved, the severity of the lesion (i.e., complete versus incomplete), and the time since the injury. Peripheral nerve disorders, particularly those that affect the autonomic nervous system, may disrupt the normal regulation of penile erectile tissue and cause erectile dysfunction. For example, erectile dysfunction may be caused by diabetes mellitus or other diseases that cause peripheral neuropathy (e.g., amyloidosis, vasculitis, heavy metal toxicity, renal failure, multiple system atrophy, acute intermittent porphyria) or by pelvic, prostate, or retroperitoneal surgery or damage (e.g., abdominoperineal resection of the rectum, pelvic lymph node dissection, prostatectomy, aortoiliac bypass, lumbar sympathectomy). Peripheral nervous system medications, including anticholinergic agents, antihistamines, antidepressants, sympathomimetic medications, α-adrenergic agonists, and β-adrenergic antagonists, often impair erectile function by affecting peripheral nervous system regulation of erectile tissue of the penis, and many also cause erectile dysfunction by altering neurotransmitter function in the nervous system and penis.
The blood supply of the penis is derived from the internal iliac (hypogastric) artery, a branch of the common iliac artery that bifurcates from the aorta.142 The internal iliac artery gives rise to the internal pudendal artery, which branches into the dorsal penile, bulbourethral, and cavernosal arteries. The cavernosal arteries run through the middle of the corpora cavernosa and give off corkscrew-shaped branches, the helicine arteries, that open directly into the lacunar spaces. Smooth muscle relaxation of lacunar spaces increases blood flow into the corpora cavernosa, resulting in penile tumescence. Blood from the lacunar spaces or cavernosal sinusoids collects in the subtunical plexus and is delivered via emissary veins to the deep dorsal vein, which ultimately drains into the internal and common internal iliac veins and then into the inferior vena cava. With filling of the lunar spaces of the corpora cavernosa and penile tumescence against the fibrous tunica albuginea, venous outflow from the subtunical venous plexus is impeded, and sustained tumescence or erection ensues. Disorders of arterial inflow or venous output may cause erectile dysfunction.
Peripheral vascular disease due to aortoiliac atherosclerosis is probably the most common cause of erectile dysfunction in aging men.142,148 These men usually have absent or severely diminished femoral artery pulses, and some present clinically with Leriche syndrome (absent femoral pulses, buttock or leg claudication, and erectile dysfunction). Other men with iliac atherosclerosis may be able to achieve an erection, but with penetration and use of the hip muscles for thrusting during sexual intercourse, blood is diverted from the penis to the hips, resulting in premature detumescence and loss of erection; this is known as the pelvic steal syndrome. Atherosclerotic large- and small-vessel disease may contribute to erectile dysfunction in men with diabetes mellitus, hypertension, CKD, smoking, and other atherosclerotic risk factors. Erectile dysfunction occurs in about 50% of men with diabetes mellitus. Smoking, specifically nicotine, also causes direct vasoconstriction of the corpora cavernosum and erectile dysfunction. Other conditions that compromise aortoiliac circulation, such as pelvic trauma, irradiation, and vasculitis, are less common causes of erectile dysfunction. Chronic pressure on the pudendal artery from bicycle riding, especially with some bicycle seats, may cause penile ischemia and erectile dysfunction; in addition, pressure on the pudendal nerve may cause penile numbness and contribute to sexual dysfunction. Penile venous incompetence (venous leakage) may cause premature loss of erections and inability to maintain erections sufficient to complete intercourse.
Many antihypertensive medications, including diuretics, α- and β-adrenergic antagonists, angiotensin-converting enzyme inhibitors, and calcium channel antagonists, have been implicated as causes of erectile dysfunction. Penile abnormalities, such as Peyronie disease or chordee (fibrosis or scarring of the tunica albuginea resulting in bending of the penis), micropenis or microphallus, penile trauma, phimosis (inability to retract the foreskin over the penis), and priapism (painful extended erections) may also cause erectile dysfunction.
Evaluation of Erectile Dysfunction.
The cause of erectile dysfunction is usually strongly suspected on the basis of a careful medical, psychiatric, and medication history and physical examination.142,148 Erectile dysfunction of psychogenic origin usually occurs abruptly, is transient, is intermittent or associated with a stressful situation, occurs with only some partners but not with others or does not occur with masturbation. Spontaneous evening and morning erections are usually maintained in psychogenic erectile dysfunction but lost with organic causes. Spontaneous erections may be detected by formal measurements of nocturnal penile tumescence (NPT) in a sleep laboratory or by breakage of wires of different tensile strength in a snap gauge (RigiScan), but these assessments are not routinely performed in practice and usually are not necessary.
Patients with nonpsychogenic brain disorders, spinal cord or peripheral nervous system disorders, vascular disorders, or penile abnormalities that cause organic erectile dysfunction usually exhibit clinical manifestations of the underlying disorder, and offending drugs that impair erectile function are revealed with a careful review of medications. Androgen deficiency is a cause of reduced libido and erectile dysfunction and occurs in 15% to 20% of men who complain of sexual dysfunction in a general medical clinic.156 Therefore, evaluation of men who present with sexual dysfunction should include inquiry regarding other symptoms of androgen deficiency, examination for signs such as small testis size and gynecomastia, and confirmation of androgen deficiency by measurement of serum testosterone levels (see later discussion).
Peripheral pulses, in particular the presence of femoral pulses, should be tested to assess for peripheral vascular disease. Diagnosis of penile vascular insufficiency may be suspected by Doppler ultrasound measurement of the ratio of penile to brachial systolic blood pressure (penile/brachial index). A penile/brachial index greater than 0.75 is normal, whereas an index of less than 0.60 is suggestive of vascular erectile dysfunction. If there is a clinical suspicion of spinal cord disease, perineal and penile sensation should be assessed. A cremasteric reflex (stroking of the inner thigh associated with contraction of the ipsilateral cremasteric muscle and pulling up of the scrotum and testis) and a bulbocavernosus reflex (squeezing of the glans penis associated with contraction of the anal sphincter) should be elicited to assess spinal cord levels L1-L2 and S2-S4, respectively. Finally, the penis should be examined for abnormalities, such as penile plaques, angulation, or tight, unretractable foreskin.
Ejaculatory Disorders and Orgasmic Dysfunction.
After the plateau phase of erection is achieved, sympathetic nervous system stimulation from the thoracolumbar (T10-L2) spinal erection center travels via the hypogastric nerve and pelvic plexus, enters the penis via the cavernosal nerve, and causes α-adrenergic receptor–mediated contraction of the cauda epididymis, vas deferens, accessory sex glands (the bulbourethral or Cowper glands and the urethral glands or glands of Littre), prostate, seminal vesicles, and ejaculatory ducts that moves sperm and semen into the posterior urethra (emission). It also stimulates closure of the internal urethral sphincter to prevent retrograde ejaculation of sperm into the bladder.142 After emission, continued sensory stimulation of the penis with sexual intercourse or masturbation stimulates reflex rhythmic contractions of the ischiocavernosus and bulbocavernosus muscles, resulting in expulsion of semen from the urethra (ejaculation).142
Like erection, ejaculation is under considerable control by higher brain centers, with both voluntary and involuntary regulation.142 Premature ejaculation is ejaculation that occurs before or shortly after vaginal penetration during sexual intercourse and is followed by a rapid loss of erection.157 The cause of premature ejaculation is usually a psychological disturbance such as performance anxiety; it is rarely the result of an organic cause. There is evidence that serotoninergic neurotransmission inhibits sexual function and ejaculation. Selective serotonin reuptake inhibitors retard ejaculation, an effect that is used therapeutically to treat premature ejaculation.157,158 Other men with psychological disorders such as excessive anxiety may have retarded ejaculation (inability to ejaculate), either in isolation or in combination with impaired libido and erections. The ejaculate is composed of spermatozoa (10%) and seminal fluid (90%), the latter derived mostly from the seminal vesicles (65%) and the prostate gland (30%). Because secretions from these accessory sex glands are androgen dependent, severe androgen deficiency may result in absent or reduced ejaculation. Absent or reduced ejaculation may also be caused by urethral abnormalities. Autonomic neuropathy, such as that caused by diabetes mellitus, sympatholytic drugs, thoracolumbar sympathectomy, extensive retroperitoneal or pelvic surgery, or bladder neck surgery, may be associated with absent or reduced ejaculation by causing retrograde ejaculation into the bladder.
Orgasm, the pleasurable sensation associated with ejaculation, usually occurs simultaneously with ejaculation and is mediated by CNS activation via ascending pathways from the spinal cord erection centers to regions of the temporal lobe and limbic system.142 Because of impaired libido and erectile dysfunction, men with androgen deficiency may also fail to achieve an orgasm. Isolated absence of orgasm in the presence of normal libido, erections, and ejaculation is relatively rare and is almost always caused by a psychological disorder.
Disorders of Detumescence.
After ejaculation, the thoracolumbar sympathetic outflow acts via α-adrenergic receptor stimulation to cause contraction of trabecular smooth muscle, which results in collapse of lacunar spaces, vasoconstriction of arterioles of the corpora cavernosa (reducing blood flow into the penis), and decompression of subtunical venules, leading to an increase in venous outflow and a flaccid penis (detumescence).142 Premature detumescence may contribute to erectile dysfunction, such as that caused by penile venous incompetence. Intracorporal injection of an α-adrenergic receptor antagonist, phentolamine, together with papaverine and PGE1, causes sustained lacunar smooth muscle relaxation, arteriole vasodilatation, and penile tumescence and is used to treat erectile dysfunction caused by premature detumescence.
Priapism is failure of detumescence with persistence of erection lasting for longer than 4 hours that is unrelated to sexual stimulation and is usually painful.142,159 An erection that persists for more than 4 hours is an emergency and may be complicated by ischemia, thrombosis, and vascular damage that contribute further to erectile dysfunction; if ischemia is severe, it can cause gangrene and eventual loss of the penis. Priapism may be idiopathic, or it may be caused by medications (e.g., intracavernosal injection therapy for erectile dysfunction, phenothiazines, trazodone, cocaine), by hematologic disorders such as sickle cell disease or chronic myelogenous leukemia, by neurologic disorders such as spinal cord injury, or by infiltrative diseases such as amyloidosis. The initial treatment is administration of the α-adrenergic receptor agonist, pseudoephedrine; if this is unsuccessful, aspiration of blood from the corpora cavernosa is performed with local anesthesia.
Gynecomastia
Gynecomastia is benign enlargement of the male breast caused by proliferation of glandular breast tissue.160-162 On inspection, it is difficult to distinguish gynecomastia from increased adipose tissue deposition within the breast in the absence of glandular proliferation (pseudogynecomastia), which is commonly present in obese men and boys. Detection of glandular breast tissue requires a careful and properly performed physical examination (see earlier discussion), feeling for a firm, rubbery, finely lobular, freely mobile disc of tissue that extends concentrically from under the nipple and areola. Initially, gynecomastia of relatively recent and rapid onset may be painful and associated with tenderness. With time, glandular tissue is replaced by fibrous tissue and tenderness resolves, although palpable tissue remains. In contrast, pseudogynecomastia is soft, nondiscrete, and irregularly lobular, similar to subcutaneous fat in the abdomen.
Gynecomastia is usually present bilaterally but may be asymmetric in size and variably symptomatic. If palpable breast tissue is present unilaterally, the major concern is male breast cancer. Breast cancer is usually rock-hard and indurated, eccentrically located from the nipple and areola, and fixed to underlying tissue; it may be associated with skin dimpling with retraction of hair follicles (peau d'orange), nipple retraction, nipple bleeding or discharge, or axillary lymphadenopathy.163 Other chest wall tumors may cause unilateral breast enlargement, including lipomas, sebaceous or dermoid cysts, hematomas, fat necrosis, lymphangiomas, neurofibromas, and lymphomas.
The primary hormones that regulate breast tissue development are estrogens, which stimulate the growth and differentiation of breast epithelium to form ducts (ductal hyperplasia), and progesterone, which controls acinar development and the formation of glandular buds (glandular formation).160,162 GH, IGF-1, insulin, thyroid hormone, and cortisol play permissive roles in breast development. Androgens inhibit the growth and differentiation of breast tissue. Prolactin stimulates differentiated breast acinar cells to produce milk, but high progesterone levels inhibit lactogenesis. Therefore, milk production requires a reduction in high progesterone levels in the presence of high prolactin levels, as occurs in the first few days after delivery. Milk production (galactorrhea) is rarely seen in men with hyperprolactinemia and gynecomastia, because progesterone levels are not usually high enough for breast acinar development to occur, and they do not decline in the presence of high prolactin levels to stimulate lactogenesis.
Gynecomastia develops in clinical situations in which the levels or activity of estrogens is relatively high in comparison with androgens (i.e., high estrogen-to-androgen ratio). This hormonal milieu may result from high estrogen or low androgen concentrations or activity. Androgen deficiency, because it decreases the inhibitory influence of androgens on breast development, is a major cause of gynecomastia. However, the differential diagnosis of other causes of gynecomastia should be considered in patients who present with breast enlargement with or without tenderness.
Causes of Gynecomastia.
Physiologic gynecomastia occurs normally in neonatal and pubertal boys. Transient gynecomastia (neonatal gynecomastia) occurs in 60% to 90% of neonatal boys as a result of exposure in utero to high concentrations of maternal estrogens; it resolves within several weeks after delivery (Table 19-3).160-162 At the time of puberty, breast enlargement greater than 0.5 cm in diameter, which is often tender, initially occurs in 60% to 70% of boys by 14 years of age and then regresses within 1 to 2 years. This pubertal gynecomastia is thought to be caused by a transient rise in serum concentrations of estrogen relative to testosterone during puberty.
TABLE 19-3
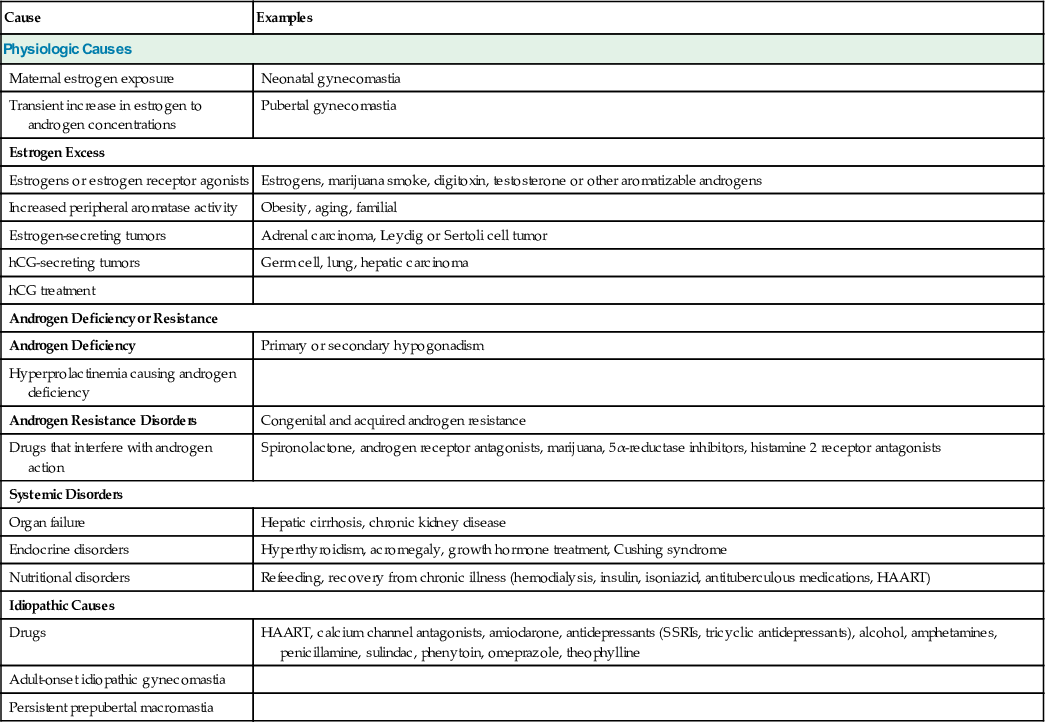
Causes of Gynecomastia
| Cause | Examples |
| Physiologic Causes | |
| Maternal estrogen exposure | Neonatal gynecomastia |
| Transient increase in estrogen to androgen concentrations | Pubertal gynecomastia |
| Estrogen Excess | |
| Estrogens or estrogen receptor agonists | Estrogens, marijuana smoke, digitoxin, testosterone or other aromatizable androgens |
| Increased peripheral aromatase activity | Obesity, aging, familial |
| Estrogen-secreting tumors | Adrenal carcinoma, Leydig or Sertoli cell tumor |
| hCG-secreting tumors | Germ cell, lung, hepatic carcinoma |
| hCG treatment | |
| Androgen Deficiency or Resistance | |
| Androgen Deficiency | Primary or secondary hypogonadism |
| Hyperprolactinemia causing androgen deficiency | |
| Androgen Resistance Disorders | Congenital and acquired androgen resistance |
| Drugs that interfere with androgen action | Spironolactone, androgen receptor antagonists, marijuana, 5α-reductase inhibitors, histamine 2 receptor antagonists |
| Systemic Disorders | |
| Organ failure | Hepatic cirrhosis, chronic kidney disease |
| Endocrine disorders | Hyperthyroidism, acromegaly, growth hormone treatment, Cushing syndrome |
| Nutritional disorders | Refeeding, recovery from chronic illness (hemodialysis, insulin, isoniazid, antituberculous medications, HAART) |
| Idiopathic Causes | |
| Drugs | HAART, calcium channel antagonists, amiodarone, antidepressants (SSRIs, tricyclic antidepressants), alcohol, amphetamines, penicillamine, sulindac, phenytoin, omeprazole, theophylline |
| Adult-onset idiopathic gynecomastia | |
| Persistent prepubertal macromastia | |
Pathologic gynecomastia may result from excessive estrogen levels or action or from androgen deficiency or resistance/insensitivity in isolation. In some conditions, both estrogen excess and androgen deficiency contribute to proliferation of glandular breast tissue.160-162 For example, in most conditions that cause gynecomastia as a result of excessive estrogen exposure, high circulating estrogen concentrations inhibit endogenous gonadotropin and testosterone secretion and cause secondary hypogonadism, which also contributes to the growth of breast tissue. Also, some disorders of the testes that cause androgen deficiency (i.e., primary hypogonadism), such as Klinefelter syndrome, result in high circulating LH levels that stimulate aromatase activity in Leydig cells, leading to higher levels of estradiol relative to testosterone and contributing to the pathogenesis of gynecomastia.
Estrogen excess disorders that cause gynecomastia include exposure to exogenous estrogens (e.g., diethylstilbestrol treatment of prostate cancer, contact with an estrogen-containing cream or cosmetic, accidental occupational exposure to estrogens, ingestion of estrogen-containing nutritional supplements or excessive amounts of phytoestrogens) and exposure to ER agonists such as marijuana smoke (unidentified phenolic components but not active cannabinoids164) or digitoxin. Ingestion of normal dietary amounts of phytoestrogens (e.g., soybean isoflavones) does not usually cause gynecomastia.165 Uncommonly, administration of testosterone or other aromatizable androgens, usually to prepubertal boys or men with long-standing, severe androgen deficiency, induces or worsens gynecomastia by initially causing relatively higher estradiol than testosterone levels.
Increased peripheral aromatase activity with increased conversion of androgens to estrogens in excessive amounts of adipose tissue is thought to cause mild to moderate gynecomastia in men with obesity.160-162 Also, increased aromatization of androgens to estrogens with increasing amounts of adipose tissue (including that within the breast) probably contributes substantially to the increased prevalence of gynecomastia with aging, which occurs in up to 65% of men 50 to 80 years of age.160-162 Familial gynecomastia, an autosomal dominant or X-linked genetic disorder caused by constitutive activation of the CYP19A1 (aromatase) gene that results in increased peripheral conversion of androgen to estrogen, is a very rare cause of gynecomastia that manifests as prepubertal gynecomastia persisting into adulthood.
Estrogen-secreting tumors of the adrenal gland or testis are uncommon causes of gynecomastia. Feminizing adrenal tumors are usually malignant and large, manifesting with a palpable abdominal mass. In contrast, estrogen-secreting Leydig or Sertoli tumors are usually small and benign. Feminizing Sertoli tumors (in particular, the large cell calcifying variety) may occur in isolation or in association with autosomal dominant disorders such as Peutz-Jeghers syndrome (multiple intestinal polyps and mucocutaneous pigmented macules) or the Carney complex (cardiac or cutaneous myxomas, pigmented skin lesions, and endocrinopathy, including functioning endocrine tumors of the adrenal and testis). hCG-secreting tumors (e.g., germ cell, lung, gastric, renal cell, or hepatic carcinomas in adults; hepatoblastomas in boys) or hCG treatment of gonadotropin deficiency increases aromatase activity in Leydig cells and stimulates excessive secretion of estradiol relative to testosterone, causing relative rapid onset of symptomatic gynecomastia.
Disorders and drugs that cause androgen deficiency, such as conditions that cause either primary or secondary hypogonadism (including medications such as cytotoxic agents) or androgen resistance, are major causes of gynecomastia.160-162 Although prolactin acts on the breast to facilitate milk production in developed glandular tissue, the major mechanism by which hyperprolactinemia causes gynecomastia is inhibition of endogenous gonadotropin and testosterone production (inducing androgen deficiency), which acts indirectly to stimulate breast development by reducing the inhibitory influence of androgens on the breast. Hyperprolactinemia is a main reason that a number of CNS-active medications, such as antipsychotics, antidepressants, and sedatives, are associated with gynecomastia. Drugs that interfere with androgen action, such as spironolactone (in contrast to eplerenone, a selective aldosterone receptor antagonist that does not cause gynecomastia), AR antagonists (e.g., flutamide, bicalutamide, nilutamide), marijuana, and histamine 2 (H2) receptor antagonists, may cause gynecomastia.
Androgen deficiency contributes to the pathogenesis of gynecomastia in systemic disorders such as major organ failure—and, in particular, in hepatic cirrhosis and CKD, which are commonly associated with combined primary and secondary hypogonadism—and in endocrine disorders such as acromegaly and Cushing syndrome, which may be associated with secondary hypogonadism.160-162 In hepatic cirrhosis, there is reduced catabolism of Δ4-androstenedione, resulting in increased peripheral conversion of Δ4-androstenedione to estrone and increased circulating estrogen levels. Also, in both hepatic cirrhosis and hyperthyroidism, increased serum concentrations of SHBG, which binds testosterone with greater affinity than estradiol, result in relatively higher free estradiol compared with free testosterone levels and thereby contribute to stimulation of breast tissue and development of gynecomastia. LH levels are often elevated in men with hyperthyroidism, which stimulates relatively more estradiol than testosterone secretion by Leydig cells of the testes. Excessive GH with acromegaly or GH treatment and excessive cortisol with Cushing syndrome directly stimulate breast tissue growth in addition to causing secondary hypogonadism, both of which contribute to the pathogenesis of gynecomastia.
Gynecomastia often accompanies nutritional disorders, in particular during nutritional repletion after a period of starvation and weight loss (refeeding gynecomastia) and analogously during recovery from chronic illness.160-162 In both starvation and severe chronic illness that is commonly associated with anorexia and weight loss, central GnRH production and concomitant gonadotropin and testosterone secretion are markedly suppressed. With refeeding or restitution of appetite and weight gain, there is activation of the hypothalamic-pituitary-testicular axis and restoration of gonadal function, similar to what occurs during puberty but occurring more rapidly (a “second puberty”), resulting in transiently higher levels of estrogen relative to androgen levels and inducing gynecomastia. Refeeding gynecomastia was described initially in World War II prisoners who developed painful gynecomastia after liberation and nutritional repletion. Analogously, refeeding-like gynecomastia may occur in stage 5 CKD with the initiation of hemodialysis, in type 1 diabetes mellitus (T1DM) with insulin therapy, in tuberculosis with antituberculosis medications, and in human immunodeficiency virus (HIV) infection or AIDS with highly active antiretroviral treatment (HAART). As mentioned, these chronic systemic disorders also cause androgen deficiency that may contribute to the pathogenesis of gynecomastia. HAART also may cause lipohypertrophy and fat accumulation in the breast (pseudogynecomastia), and efavirenz has estrogenic activity.
The mechanisms of gynecomastia associated with a number of drugs are not entirely clear, and these cases are usually classified as idiopathic. Such drugs include HAART, calcium channel blockers (e.g., nifedipine, verapamil), amiodarone, antidepressants (selective serotonin reuptake inhibitors, tricyclic antidepressants), alcohol, amphetamines, penicillamine, sulindac, phenytoin, omeprazole (much less commonly than H2-receptor antagonists), and theophylline.160-162,166,167
In a number of cases of adult-onset gynecomastia, the cause remains idiopathic. Most of these cases are probably caused by increased aromatization of androgens to estrogens associated with increased peripheral adiposity, enhanced breast production of estrogens, enhanced sensitivity to estrogens, or some combination of these factors. Rarely, boys may develop severe pubertal gynecomastia (female size breast development, Tanner stage III through V) that persists to adulthood (persistent pubertal macromastia). This disorder is not associated with specific hormonal or receptor abnormalities and remains idiopathic.
Evaluation.
Most gynecomastia is asymptomatic and of mild degree but can be appreciated on a properly performed, careful physical examination (as described earlier). Mild, asymptomatic gynecomastia found incidentally on examination and in isolation does not warrant evaluation. However, breast enlargement that is recent and rapid in onset, large (>5 cm in obese men, >2 cm in lean men), symptomatic (i.e., associated with breast pain, tenderness, or galactorrhea), asymmetric, or suspicious for malignancy (eccentrically located, rock-hard, fixed to overlying or underlying tissues, or associated with bloody nipple discharge or lymphadenopathy) should trigger further evaluation.
A careful history, including medication history, and physical examination usually identify potential predisposing conditions or medications causing gynecomastia that in older men may be mulifactorial.160-162 Clinical evaluation should focus on evidence of androgen deficiency; assessment of prescription and over-the-counter medications, substance abuse, herbal or nutritional supplement intake, cosmetic use, and usual dietary intake; symptoms and signs of systemic illness (e.g., hepatic or renal disease), malignancy, or endocrine disorders (e.g., thyroid, GH, cortisol excess); and history of recent recovery from malnutrition, severe weight loss, or chronic illness. At a minimum, the initial laboratory evaluation comprises serum testosterone, LH, FSH, TSH, and renal and liver function tests. Evaluation also usually includes measurements of estradiol, prolactin, and β-hCG, although elevations of these hormones usually affect testosterone and gonadotropin concentrations. Breast enlargement suspicious for malignancy should be evaluated by mammography and biopsy.
Treatment.
Pubertal gynecomastia usually regresses spontaneously without treatment in 1 to 2 years and by age 17 in about 90% of cases. In adults, spontaneous regression of symptoms (breast pain and tenderness, nipple sensitivity) associated with inflammatory glandular proliferation usually occurs within 6 months, after which progressive stromal fibrosis causes more or less permanent palpable breast tissue and only partial regression of gynecomastia by 1 year.
Initial treatment of gynecomastia is directed at correction of the underlying cause of breast enlargement or discontinuation or replacement of a potentially offending medication.168 Testosterone replacement therapy in androgen-deficient men may result in partial regression of gynecomastia, especially if breast enlargement is of recent onset. Prophylactic low-dose breast irradiation (10 to 15 Gy over 1 to 3 days) may be used before androgen deprivation therapy in men with prostate cancer to prevent the development of gynecomastia; this is more common in surgical orchidectomy and in AR antagonist monotherapy rather than combined therapy with a GnRH agonist or antagonist. ER antagonists (tamoxifen, 10 to 20 mg daily, or raloxifene, 60 mg daily) are effective in treating pubertal and adult gynecomastia and preventing gynecomastia induced by androgen deprivation therapy. For unclear reasons, aromatase inhibitors (e.g., anastrazole) are not effective. Although tamoxifen is not approved for treatment of gynecomastia, it has been shown to be effective in the treatment of idiopathic gynecomastia, resulting in partial regression in approximately 80% and complete regression in about 60% of cases. A gel formulation of DHT, a nonaromatizable androgen, is used to treat gynecomastia in some countries outside the United States.
Gynecomastia of recent onset, during the initial phase of ductal proliferation, periductal inflammation and edema, and subareolar fat accumulation, is usually responsive to medical therapy (e.g., androgen replacement in hypogonadal men, ER antagonist therapy). With long-standing gynecomastia (>1 year), there is progressive stromal fibrosis of the breast that is not responsive to medical treatment. In these cases, surgical reduction mammoplasty (i.e., removal of breast tissue [subcutaneous mastectomy] with or without periareolar adipose tissue [liposuction]) is necessary, especially if breast enlargement is severe, painful, socially embarrassing, or disfiguring.
Infertility
Infertility is defined as the inability of a sexually active couple to achieve conception despite 1 year of unprotected intercourse. The probability of conception in a sexually active couple is approximately 85% by 1 year. Approximately 15% of couples in the reproductive age group are infertile, and a male factor contributes to the cause (either in isolation or in combination with a female factor) in about half of the cases. Therefore, male infertility is a common condition, affecting approximately 7% of men.169
Causes of Male Infertility.
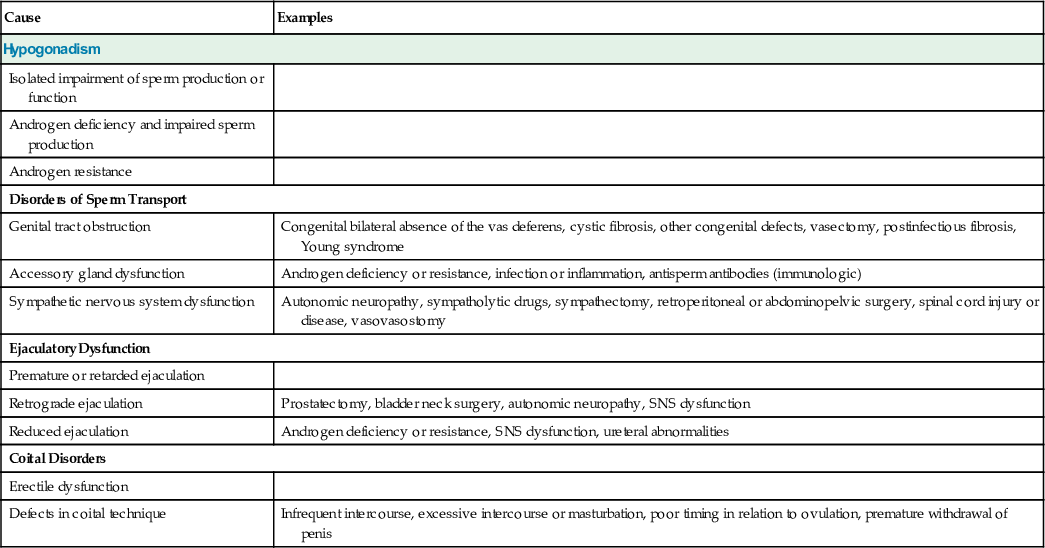
In about 80% to 90% of infertile men, infertility is caused by primary or secondary hypogonadism, manifested mostly by an isolated impairment of sperm production or function, much less commonly by androgen deficiency and impaired spermatogenesis, and rarely by androgen resistance (Table 19-4).170,171 The evaluation and specific causes of hypogonadism are discussed in detail in subsequent sections. Most men with isolated impairment in sperm production have a primary disorder of the testes that is idiopathic in 60% to 70% of cases (if one includes both idiopathic oligozoospermia or azoospermia and varicocele, given that relationship of varicocele to the pathogenesis of infertility is unclear). If isolated impairment of spermatogenesis is severe in men with primary hypogonadism, serum FSH levels may be selectively elevated as a result of reduced negative feedback by inhibin B from Sertoli cells of the testis. In men with less severely impaired spermatogenesis, serum gonadotropin levels are normal, but this is still classified with disorders of primary hypogonadism because gonadotropin treatment has not been demonstrated to improve fertility.
TABLE 19-4
Causes of Male Infertility
| Cause | Examples |
| Hypogonadism | |
| Isolated impairment of sperm production or function | |
| Androgen deficiency and impaired sperm production | |
| Androgen resistance | |
| Disorders of Sperm Transport | |
| Genital tract obstruction | Congenital bilateral absence of the vas deferens, cystic fibrosis, other congenital defects, vasectomy, postinfectious fibrosis, Young syndrome |
| Accessory gland dysfunction | Androgen deficiency or resistance, infection or inflammation, antisperm antibodies (immunologic) |
| Sympathetic nervous system dysfunction | Autonomic neuropathy, sympatholytic drugs, sympathectomy, retroperitoneal or abdominopelvic surgery, spinal cord injury or disease, vasovasostomy |
| Ejaculatory Dysfunction | |
| Premature or retarded ejaculation | |
| Retrograde ejaculation | Prostatectomy, bladder neck surgery, autonomic neuropathy, SNS dysfunction |
| Reduced ejaculation | Androgen deficiency or resistance, SNS dysfunction, ureteral abnormalities |
| Coital Disorders | |
| Erectile dysfunction | |
| Defects in coital technique | Infrequent intercourse, excessive intercourse or masturbation, poor timing in relation to ovulation, premature withdrawal of penis |
Disorders of spermatogenesis caused by primary hypogonadism may be associated with chromosomal or genetic disorders. There is an 8- to 10-fold increase in the prevalence of chromosomal abnormalities among infertile men with impaired spermatogenesis—specifically, sex chromosomal aneuploidy (e.g., Klinefelter syndrome) or Robertsonian translocations of two nonhomologous chromosomes, most commonly involving chromosomes 13 and 14 or chromosomes 14 and 21.172 The long arm of the Y chromosome (Yq), specifically the azoospermia factor (AZF) region (Yq11), contains a number of genes that encode for proteins that have important roles in spermatogenesis. This region contains highly homologous palindromic DNA repeat sequences that are susceptible to rearrangement and deletions. Small deletions in the AZF region (Y chromosome microdeletions) are the most common genetic cause of impaired sperm production and male infertility; they are found in 5% to 10% of men with azoospermia or severe oligozoospermia (sperm concentration <5 million/mL).172 Y chromosome microdeletions have been identified in three regions: in the AZFa region, microdeletions are uncommon but are usually associated with azoospermia and Sertoli cell–only histology; in the AZFb region, they are usually associated with severe oligozoospermia and germ cell arrest at the pachytene primary spermatocyte stage; and in the AZFc region, where the majority of Y chromosome microdeletions reside, they are usually associated with germ cell arrest at the spermatid stage or hypospermatogenesis with some mature spermatids present. Occasionally, microdeletions in the AZFb and AZFc regions are associated with azoospermia and Sertoli cell–only histology. Genes encoding a number of candidate proteins for male infertility include DDX3Y (DEAD box Y, an ATP-dependent RNA helicase), RBMY (RNA-binding motif Y-linked, an RNA-binding protein), and DAZ (deleted in azoospermia, another RNA-binding protein) in the AZFa, AZFb, and AZFc regions, respectively.172,173
Approximately 15% to 20% of male infertility is caused by disorders of sperm transport from the testes to the urethra, most commonly by genital tract obstruction. Congenital bilateral absence of the vas deferens (CBAVD) is present in 1% to 2% of men with infertility.172,174,175 Seventy-five percent of men with CBAVD are heterozygous for the cystic fibrosis transmembrane conductance regulator gene (CFTR), which encodes for an epithelial chloride channel, or carry compound heterozygous mutations of CTFR. They do not have obvious clinical manifestations of cystic fibrosis, although some manifest abnormalities on sweat chloride testing and sinopulmonary infections. Conversely, almost all men with cystic fibrosis have CBAVD. CBAVD is also commonly associated with absence of the seminal vesicles, ejaculatory ducts, and epididymides due to fetal atrophy of these wolffian duct derivatives; in 10% of cases, there is also renal agenesis or hypoplasia.
Other causes of genital tract obstruction include other congenital defects of the epididymides and vas deferens (e.g., epididymal cysts associated with prenatal diethylstilbestrol exposure); vasectomy (surgical ligation of the vas deferens); postinfectious fibrosis (e.g., associated with gonorrhea, Chlamydia infection, other sexually transmitted diseases; tuberculosis; leprosy); and Young syndrome, a rare, congenital primary ciliary dyskinesia syndrome characterized by bronchiectasis, recurrent sinopulmonary infections, and obstructive azoospermia caused by thickened, inspissated mucous secretions obstructing the epididymides.
Although a causal link to infertility has not been clearly established, other genital tract abnormalities may contribute to impaired sperm transport and the pathogenesis of infertility in some men. Accessory gland dysfunction, such as reduced seminal vesicle and prostate secretions associated with disorders that cause severe androgen deficiency or resistance, may contribute to reduced fertility, although the main effects of these disorders are to impair spermatogenesis and cause sexual dysfunction. Infection or inflammation of the epididymides, seminal vesicles, or prostate gland may affect fertility directly by impairing sperm maturation or function or secondarily by causing scarring of the genital tract or induction of antisperm antibodies in semen (resulting in sperm agglutination and impaired sperm function).176,177 Sympathetic nervous system dysfunction (e.g., associated with autonomic neuropathy, sympatholytic drugs, sympathectomy, retroperitoneal or abdominopelvic surgery, spinal cord injury or disease, vasovasostomy) may contribute to impaired sperm transport and male infertility.
Ejaculatory dysfunction may cause or contribute to male infertility by preventing normal or efficient deposition of sperm into the vagina and female genital tract. Premature or retarded ejaculation may contribute to infertility if ejaculation occurs during arousal or foreplay before vaginal penetration or after withdrawal from the vagina. Retrograde ejaculation of semen into the bladder rather than the urethra occurs with neuromuscular failure of normal bladder sphincter contraction during ejaculation. Retrograde ejaculation may be associated with prostatectomy or bladder neck surgery (e.g., transurethral resection of the prostate [TURP]), autonomic neuropathy (e.g., complicating diabetes mellitus), or sympathetic nervous system dysfunction, and in particular with sympatholytic drugs (e.g., α-adrenergic receptor antagonists such as prazosin or terazosin), retroperitoneal or abdominopelvic surgery (e.g., retroperitoneal lymph node dissection), and spinal cord injury or disease. Reduced ejaculation caused by androgen deficiency or resistance, sympathetic nervous system dysfunction, or urethral abnormalities may contribute to reduced sperm delivery to the female genital tract.
Erectile dysfunction may contribute to male infertility by causing unsuccessful completion of intercourse. Coital disorders are uncommon causes of male infertility, but they are potentially correctable with proper education. Infrequent sexual intercourse, excessive intercourse with other partners or masturbation, intercourse during menses rather than just before or around the time of ovulation, and premature withdrawal of the penis during intercourse may contribute to reduced fertility.
Evaluation.
Because a coexisting female factor contributes to infertility in 30% of cases, it is important for the female partner to undergo evaluation for ovulation (menstrual periods, androgenization) and for cervical disorders (postcoital testing) and uterine and tubal disorders (hysterosalpingogram, pelvic ultrasound). In men, the history and physical examination are usually able to identify the potential cause of male infertility.170-172
In addition to an assessment of general health and medical comorbid conditions, the initial clinical evaluation should focus on the following:
• Symptoms and signs of androgen deficiency or resistance (as detailed elsewhere in this chapter)
• Scrotal examination for presence of a varicocele, presence and size of the testes, and presence or absence of firm, fibrous cords of the vas deferens
• Family history or evidence of cystic fibrosis
• Previous vasectomy or vasovasostomy
• History or manifestations of genitourinary infections
• Medications, particularly ones that cause androgen deficiency or resistance and sympatholytic agents
• Ejaculatory problems, in particular absent or reduced ejaculate
• Autonomic neuropathy (e.g., complicating diabetes mellitus)
• Retroperitoneal or abdominopelvic surgery
The initial laboratory evaluation of male infertility should begin with at least two or three seminal fluid analyses performed over a period of a few months (see later discussion) to assess semen volume, sperm count and concentration, and sperm motility and morphologic appearance, with the aim of identifying men who have impaired sperm production or function, the major cause of male infertility. The presence of leukocytes in semen (>106/mL, termed leukospermia or pyospermia) may suggest a genitourinary inflammation or infection, but routine cultures are not usually helpful in guiding treatment. Agglutination of spermatozoa in semen (i.e., sticking of motile sperm to each other) suggests the presence of antisperm antibodies, which can be measured in semen and and may indicate an immunologic cause of male infertility.
Seminal fluid fructose is derived mostly from the seminal vesicles (60%) and to a lesser extent from the prostate gland (30%). Absent or low seminal fluid fructose and low semen volume suggest either congenital absence of the vas deferens and seminal vesicles or obstruction of the ejaculatory ducts. Dilated seminal vesicles may be detected on transrectal ultrasonography to confirm the presence of ejaculatory duct obstruction. In men who have little or no ejaculate, a postejaculatory urine specimen should be collected and examined for the presence of sperm, indicating retrograde ejaculation.
If there are clinical manifestations of androgen deficiency, serum testosterone levels should be measured on at least two occasions to confirm androgen deficiency, and measurements of LH and FSH should be performed to determine whether the patient has primary or secondary hypogonadism (see later discussion). Identification of infertile men with secondary hypogonadism potentially has important therapeutic implications. In men with impaired sperm production due to gonadotropin deficiency, spermatogenesis may be stimulated and fertility restored with the use of gonadotropin or GnRH therapy. Secondary hypogonadism is one of few treatable causes of male infertility. Elevated levels of testosterone, LH, and FSH suggest androgen resistance.
Measurements of FSH levels specifically as a marker of Sertoli cell and seminiferous tubule function are useful in identifying men with severe defects in spermatogenesis and impairment of seminiferous tubule and Sertoli cell function; such patients often demonstrate selective elevation in serum FSH concentrations with normal or high-normal LH levels due to loss of negative feedback inhibition of pituitary FSH secretion by inhibin B.178 However, men with less severe seminiferous tubule dysfunction and impairment of spermatogenesis and those with azoospermia due to genital tract obstruction (obstructive azoospermia) have normal serum FSH levels.
Genetic disorders make up a small but significant proportion of the causes of male infertility. Because ART, and specifically ICSI, which involves direct injection of spermatozoa into the cytoplasm of an ovum (discussed later), is commonly used to treat male infertility, the potential exists for transmission of genetic defects to offspring. Therefore, genetic testing and counseling should be undertaken for men who are considering ICSI, particularly for those with severe oligozoospermia or azoospermia.170-172
Men in whom bilateral congenital absence of the vas or genital tract obstruction is suspected (i.e., those with low semen volume, low fructose level, and nonpalpable vas deferens in the scrotum) and those who have unexplained obstructive azoospermia should undergo genetic testing for CFTR mutations and genetic counseling before ICSI. In men with severe oligozoospermia (sperm concentration <5 million/mL) or azoospermia, testing for Y chromosome microdeletions in the AZF region should be performed routinely. There is a high prevalence of sex chromosome and autosomal chromosome defects, often in the absence of other phenotypic abnormalities, in men with moderately impaired spermatogenesis and infertility. Therefore, karyotyping is recommended before ICSI for infertile men with impaired sperm production, and in particular for those with a sperm concentration of less than 10 million/mL.
In azoospermic men with normal semen volume, a normal fructose level, and a normal FSH level in whom it is unclear whether azoospermia is caused by germ cell failure, genital tract obstruction, or both, surgical exploration of the scrotum and testis biopsy are needed. Biopsy is also used to harvest sperm for ICSI, even in men with known severe impairment in spermatogenesis, such as those with Klinefelter syndrome.170,171,179,180
Specialized tests of in vitro sperm function, such as detailed examination of sperm motility using computer-assisted semen analysis (CASA), cervical mucus penetration tests, acrosome reaction, and human zona pellucida binding tests, may be useful in some men who are considering intrauterine insemination or in vitro fertilization. However, these tests should be performed only in highly specialized laboratories that have demonstrated excellent quality control. Even in such laboratories, there is a high rate of clinical false-positive and false-negative results, limiting the clinical utility of these tests.
Treatment.
In men with infertility caused by primary hypogonadism (whether due to androgen deficiency and impairment of sperm production or to isolated impairment of sperm production or function), defects in sperm production are not treatable, although spontaneous recovery of spermatogenesis may occur at variable times after discontinuation of cytotoxic drugs or ionizing radiation. Because intratesticular testosterone concentrations are normally approximately 100-fold higher than serum levels, exogenous testosterone treatment of men with androgen deficiency cannot deliver sufficient amounts of testosterone to support sperm production in the testis.
In men with secondary hypogonadism, on the other hand, sperm production can be stimulated with gonadotropin or GnRH treatment, or spermatogenesis may recover sufficiently to restore fertility after discontinuation of drugs that suppress gonadotropins, such as androgenic anabolic steroids, progestins, glucocorticoids, and drugs causing hyperprolactinemia.
Most men with a varicocele and infertility have abnormal seminal fluid. However, varicocele repair has not been demonstrated to be effective in restoring fertility to these men. Therefore, unless a varicocele is very large or symptomatic, surgical repair is not recommended.12,181 Although the efficacy of treatment is unclear, infertile men with leukospermia or sperm agglutination are usually treated empirically with a 14-day course of antibiotics, such as doxycycline, trimethoprim-sulfamethoxazole, or a fluoroquinolone. Although high-dose prednisone (40 to 60 mg for several months) has been shown to be effective in treating infertile men with antisperm antibodies, the risks of high-dose glucocorticoid treatment are substantial, and this therapy cannot be recommended given the safer alternative of ICSI.
Although ICSI is costly, it is used increasingly to treat male infertility, and it dramatically improves the prognosis for men with impaired sperm production regardless of the cause.179,180 Spermatozoa that are ejaculated or obtained by testicular biopsy (testicular sperm extraction [TESE]) or from the epididymis (microsurgical epididymal sperm aspiration [MESA]) are used for ICSI and other ARTs. With ICSI, fertilization rates of about 60% and pregnancy rates of approximately 20% are achieved, irrespective of the cause of male infertility or source of spermatozoa. ICSI after TESE using microsurgical testis biopsy or fine-needle aspiration to retrieve sperm has been successful in restoring fertility to men with primary hypogonadism who had severe impairments in spermatogenesis and azoospermia that were previously thought to be untreatable (e.g., Klinefelter syndrome, prolonged azoospermia after chemotherapy).182 Because of the potential for chromosomal abnormalities and transmission of Y chromosome microdeletions and CFTR mutations that cause infertility in male offspring, genetic testing and counseling should be conducted if ICSI is being considered (see earlier discussion).172
Obstruction of the epididymides or the ejaculatory ducts can be corrected surgically, such as with end-to-end anastomosis of the epididymides or transurethral resection of the ejaculatory ducts. More commonly, MESA is used to obtain spermatozoa that can be incubated with ova in vitro (in vitro fertilization [IVF]) or directly injected into an ovum (ICSI), and this method is more successful in restoring fertility than surgical options. In contrast, microsurgical reanastomosis of the vas (vasovasostomy) to reverse vasectomy is less costly and more successful in restoring fertility than MESA followed by IVF or ICSI. Return of sperm in the ejaculate occurs in approximately 90% of men who undergo vasectomy reversal, but restoration of fertility occurs in only about 50%, probably because of stenosis or blockage of the previous vasovasostomy, epididymal blockage, or the development of antisperm antibodies in response to the vasectomy.183
In men with retrograde ejaculation, collection of spermatozoa in alkalinized postejaculation urine, followed by extensive washing and intrauterine insemination (IUI) or ICSI, has been used successfully to treat infertility. With proper education, coital disorders that contribute to infertility may be corrected. Also, the timing of intercourse may be optimized to occur a few days before and after ovulation (the period of highest probability for conception) based on basal body temperature measurements or, more accurately, on commercially available rapid urinary LH kits to estimate the timing of ovulation in the female partner.
If the treatment options described previously are not available or affordable to infertile couples who desire children, artificial insemination with donor sperm or adoption may be considered.
Diagnosis of Male Hypogonadism
Clinical Manifestations of Androgen Deficiency
The diagnosis of male hypogonadism requires clinical manifestations consistent with androgen deficiency and unequivocally low serum testosterone levels. In community-dwelling middle-aged to older men, the crude prevalence of symptomatic androgen deficiency is 2% to 9%, depending on the constellation of symptoms and signs and the biochemical definition of androgen deficiency used (i.e., the threshold used for a single low testosterone level).184-186 This prevalence increases with age and is much higher in a primary care setting.187 In community populations, the prevalence of low testosterone levels alone, without consideration of symptoms and signs of androgen deficiency, is much higher than that of clinical androgen deficiency. This underscores the importance of making a diagnosis of hypogonadism only in men who have clinical manifestations and consistently low testosterone levels. Both the clinical and the biochemical diagnosis of androgen deficiency can be challenging, especially in older adults.
Although the manifestations of fetal androgen deficiency or resistance (ambiguous genitalia and 46,XY DSD) and those of prepubertal androgen deficiency (eunuchoidism) are usually clinically obvious, the clinical diagnosis of androgen deficiency in adults is more difficult. As described previously, the symptoms and signs of androgen deficiency are nonspecific and have a broad differential diagnosis. Moreover, clinical manifestations may be modified by a number of factors, such as the severity and duration of androgen deficiency, age, comorbid illnesses, medications, previous testosterone treatment, and individual variations in androgen sensitivity, all of which contribute to variability in clinical presentation that may confound the diagnosis.113 Because the manifestations of androgen deficiency in adults are nonspecific, other potential causes (such as depression, comorbid illness, or medications) should be considered in the differential diagnosis to explain clinical features in any individual patient.188
The degree and duration of androgen deficiency have impressive effects on clinical manifestations. The severe and relatively rapid suppression of testosterone levels in men with prostate cancer treated with a GnRH agonist or orchidectomy results in prominent clinical manifestations with notable reductions in erectile function, libido, energy, and mood; hot flushes and sleep disturbances; infertility; decreases in muscle mass and strength, BMD (associated with an increase in fracture risk), and body hair; gynecomastia; increases in body fat; anemia; and possibly increases in the risks for diabetes mellitus and for cardiovascular events.189,190 In contrast, men with mild androgen deficiency may have few or no referable manifestations; such patients have “subclinical” androgen deficiency that may or may not be associated with clinically significant outcomes. The latter situation is analogous to that observed in other endocrine disorders such as subclinical hypothyroidism or asymptomatic primary hyperparathyroidism.
Aging is associated with alterations in body functions, such as declines in sexual function, muscle mass and strength, and BMD, that result in clinical manifestations similar to those of androgen deficiency.139 These alterations associated with aging may also be caused in part by age-related androgen deficiency. To add to the clinical complexity in older men, age-associated comorbid illnesses and medications used to treat these illnesses may modify the symptoms and signs of androgen deficiency, and in many instances, they may also contribute to the cause of androgen deficiency. Therefore, it is understandable why the diagnosis of clinical androgen deficiency is challenging in older men, and particularly in frail elderly men who have multiple comorbid illnesses and are taking numerous medications.
Previous testosterone treatment that has been discontinued may affect the clinical manifestations of androgen deficiency, depending on the duration of therapy and the time since discontinuation. It is also likely that clinical manifestations of androgen deficiency are affected by individual variations in androgen action on specific target organs. Alterations in androgen sensitivity may result from individual or tissue-specific differences in the activity of the AR or the ER and associated coregulators or from differences in active metabolism to estradiol or DHT, or inactivation of testosterone.
In men with clinical manifestations suggestive of androgen deficiency, the diagnosis of hypogonadism is confirmed biochemically by measurement of consistently low serum testosterone concentrations (Fig. 19-19).113
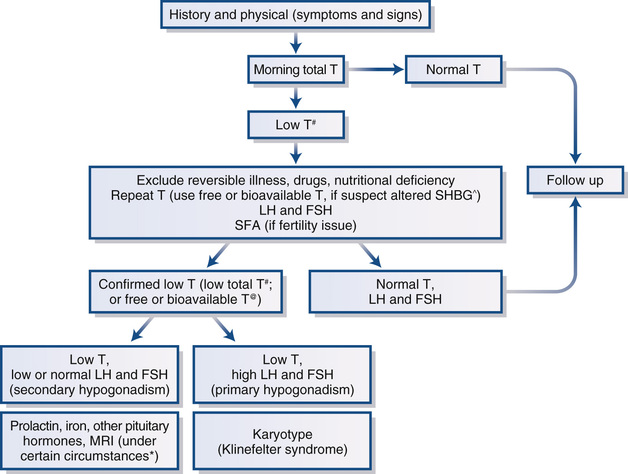
Testosterone Measurements
As with the clinical manifestations, the biochemical confirmation of androgen deficiency presents its own difficulties. Testosterone levels exhibit both biologic and assay variability. Total testosterone concentrations are affected by alterations in SHBG, and testosterone levels may be suppressed transiently with illness, certain medications, and some nutritional deficiencies.113 Therefore, the biochemical diagnosis of androgen deficiency requires demonstration of consistently and unequivocally low serum testosterone levels on at least two occasions and preferably measured in the morning. In men who have conditions that alter SHBG, accurate and reliable free or bioavailable testosterone measurements are needed to confirm the diagnosis of hypogonadism. Finally, the diagnosis of hypogonadism should not be made during acute or subacute illness.
The threshold level of circulating total or free testosterone below which symptoms and signs of androgen deficiency occur and for which testosterone treatment will improve clinical manifestations is not known. However, the concept of a single threshold testosterone level is probably not valid, nor is it clinically useful, because thresholds vary with the specific symptom and the androgen target organ or tissue. In general, symptoms and signs of androgen deficiency are more likely to occur with a total testosterone level that is below the lower limit of the normal range for young healthy men (approximately 280 to 300 ng/dL or 2.8 to 3.0 ng/mL [9.7 to 10.4 nmol/L], when using an accurate and reliable assay). The likelihood and severity of clinical manifestations of androgen deficiency increase with a greater decline in testosterone level below normal.
Variability in Testosterone Levels.
Because serum testosterone levels exhibit both biologic and assay variability, a single measurement is not a reliable indicator of an individual's average concentration. Serum testosterone levels exhibit both ultradian and circadian variation, providing physiologic sources of biologic variability. Ultradian fluctuations in serum testosterone levels, characterized by peaks of incremental amplitude that average approximately 240 ng/dL (40% fractional amplitude) with a 95-minute duration,45 have been reported in a small number of young men; more chaotic peaks with lower amplitude have been reported in older men.191 As described previously, the circadian variation in serum testosterone peaks at about 8 AM and has a maximum excursion averaging 140 ng/dL.64 The circadian variation in testosterone is blunted but still present in older men, with a maximum excursion averaging 60 ng/dL. In young men, serum testosterone levels are 20% to 25% lower at 4 PM than at 8 AM (i.e., over the course of usual clinic hours).192 This difference decreases with age: in 70-year-old men, testosterone levels are 10% lower at 4 PM than at 8 AM. Most importantly, many young and old men who have testosterone concentrations that are below normal in the afternoon have consistently normal levels in the morning. Testosterone levels are also suppressed with glucose infusion or food intake,193,194 and thus measurements should preferably be done in the fasting state. Given these findings and the fact that normal ranges of testosterone concentration are usually based on morning blood samples, testosterone measurements to confirm the diagnosis of hypogonadism should preferably be performed in the morning in the fasting state.
There is also substantial day-to-day variation in serum testosterone concentrations, underscoring the need to repeat the measurement to confirm low levels, particularly if the first result was only moderately below normal. Among men with serum testosterone levels of less than 300 ng/dL on an initial test, 30% to 35% were found to have a normal level on repeat testing.195 Among community-dwelling middle-aged to older men who had an initial serum testosterone concentration of less than 250 ng/dL, 20% were found to have an average testosterone level higher than 300 ng/dL (i.e., within the normal range) when six samples were drawn over the subsequent 6 months.196 However, none of the subjects who had an initial average serum testosterone concentration of less than 250 ng/dL on two separate samples obtained 1 to 3 days apart had an average level higher than 300 ng/dL in six samples drawn over the next 6 months. These findings support the need to measure testosterone on at least two occasions to confirm the diagnosis of androgen deficiency.
Total Testosterone Assays.
Total testosterone assays are performed in most local laboratories and are readily available to clinicians. Therefore, total testosterone is recommended as the initial measurement for the assessment of androgen deficiency. In local clinical laboratories, total testosterone is usually measured by automated platform-based direct immunoassays on unextracted serum or plasma. However, there is substantial variability in results from different assays, mostly because the accreditation of laboratories has been based on the reproducibility of results in comparison to other laboratories using the same method or kit, rather than on the accuracy of results. For example, when identical quality control samples were assayed by different methods or kits, the reported measured values ranged from 160 to 508 ng/dL. Moreover, the lower limit of the normal range in some assays was as low as 132 to 210 ng/dL (clearly in the hypogonadal range for most conventional assays).197 In contrast, the lower limit of the normal range based on conventional radioimmunoassays after extraction is approximately 280 to 300 ng/dL.
Most commercial reference laboratories now measure testosterone by liquid chromatography tandem mass spectrometry methods after solid phase extraction that have the potential to be more accurate than immunoassays. To address the problems in the quality control of testosterone assays, the U.S. Centers for Disease Control and Prevention (CDC) has initiated a program to standardize and harmonize testosterone assays using accuracy-based quality control standards to which most commercial reference laboratories participate.198 Recently, the College of American Pathologists also instituted an accuracy-based quality control program that unfortunately is not mandatory for certification.
The prevalence of low testosterone concentrations is high in a number of clinical conditions, namely, presence of a pituitary or sellar mass; irradiation; disease; use of certain medications, such as opiates or high-dose glucocorticoids; HIV disease with weight loss; late-stage CKD, especially with hemodialysis; moderate to severe chronic lung disease; infertility; BMD levels that reveal osteoporosis or low-trauma fracture; and type 2 diabetes mellitus (T2DM). If clinical manifestations consistent with androgen deficiency are present in men with these conditions, testosterone measurements should be performed.113
Total Testosterone Affected by Alterations in Sex Hormone–Binding Globulin.
Because a substantial proportion (30% to 40%) of circulating testosterone is bound tightly to SHBG, alterations in SHBG concentration may affect total testosterone levels without altering free or bioavailable testosterone. Conditions that suppress SHBG levels (even within the broad normal range) lower total testosterone (sometimes to below the normal range) without affecting circulating free or bioavailable testosterone levels (Table 19-5).113 Common conditions that lower SHBG concentrations include moderate obesity, often associated with T2DM; protein-losing states, such as nephrotic syndrome; administration of glucocorticoids, progestins, or androgens; hypothyroidism; acromegaly; and familial SHBG deficiency. SHBG concentrations are increased with increasing age; hepatic cirrhosis and inflammation (hepatitis of any cause); estrogens; hyperthyroidism; anticonvulsants; and HIV disease.
TABLE 19-5
Conditions Associated with Alterations in SHBG Concentrations
| Decreased SHBG Concentrations | Increased SHBG Concentrations |
| Moderate obesity, type 2 diabetes mellitus | Aging |
| Nephrotic syndrome | Hepatic cirrhosis and hepatitis |
| Glucocorticoids, progestins, and androgens | Estrogens |
| Hypothyroidism | Hyperthyroidism |
| Acromegaly | Anticonvulsants |
| Familial SHBG deficiency | HIV disease |
If conditions that affect SHBG concentration are present in a patient or if total testosterone concentrations are close to the lower limit of the normal range, serum free or bioavailable testosterone measurements should be obtained to confirm androgen deficiency. Unfortunately, accurate and reliable assays for free or bioavailable testosterone are not performed routinely in most local clinical laboratories. Although direct free testosterone assays using automated platform-based analogue immunoassay methods are available, these assays are inaccurate and are affected by alterations in SHBG, so they offer no advantage over total testosterone measurements and are not recommended.199-201
The gold standard method for measurement of free testosterone levels is equilibrium dialysis or centrifugal ultrafiltration. Free testosterone concentrations may be calculated accurately from measurements of total testosterone, SHBG, and albumin using affinity constants for binding of testosterone to its binding proteins and published formulas. Calculated free testosterone values are comparable to those measured by equilibrium dialysis.200 However, calculated values depend on the specific testosterone and SHBG assays employed and the formula used to estimate free testosterone.202 There are a number of formulae used for calculation of free testosterone that all demonstrate some bias relative to the equilibrium dialysis method, largely as a result of assuming a single or two identical, noninteracting binding sites on SHBG.110 However, they all provide reasonable approximations of free testosterone in the normal to low range of values that are less prone to misinterpretation and overdiagnosis of hypogonadism than total testosterone measurements when alterations in SHBG levels are present.
Bioavailable testosterone is measured by ammonium sulfate precipitation of SHBG-bound testosterone and measurement of free and albumin-bound testosterone in the supernatant. Bioavailable testosterone levels may also be calculated from measurements of total testosterone, SHBG, and albumin. These accurate and reliable measurements of free and bioavailable testosterone are available in commercial laboratories and should be used to confirm androgen deficiency in men who have conditions that affect SHBG and in those with total testosterone levels near the lower limit of the normal range. In a recent report, 60% of over 3700 men in a single health care system who had low total testosterone levels were found to have normal calculated free testosterone concentrations using the laboratory reference range that was utilized by practitioners to make clinical decisions. These findings highlight the potential clinical importance of using free testosterone to confirm a biochemical diagnosis of hypogonadism.203
Transient Suppression of Testosterone.
In evaluating men for a diagnosis of male hypogonadism, it is important to recognize that serum testosterone levels may be suppressed transiently during acute (particularly critical) and subacute illness and recovery; with the short-term use of certain medications, such as opioids, high-dose glucocorticoids, and CNS-active medications or recreational drugs that suppress gonadotropin and testosterone production; and during transient malnutrition, such as that associated with illness, eating disorders, or excessive or prolonged strenuous exercise (resulting in low energy intake relative to energy expenditure). In such situations, measurement of serum testosterone should be delayed until the patient is completely recovered from the illness, the offending drugs are discontinued, the malnutrition is corrected, or the excessive exercise is stopped.113
Case-Finding in Androgen Deficiency
In the absence of evidence for long-term clinically meaningful health benefits greater than risks for testosterone treatment of androgen deficiency, screening for androgen deficiency in the general population or in all elderly men is not indicated. Existing case-finding instruments lack sufficient specificity and sensitivity to be clinically useful. In certain clinical conditions, there is a high prevalence of low testosterone concentrations and androgen deficiency, and measurement of serum testosterone should be performed. These conditions include hypothalamic-pituitary mass, disease, surgery, or radiation therapy; medications that suppress testosterone production (e.g., opioids, glucocorticoids); HIV-associated weight loss and other wasting syndromes; and osteoporosis or minimal-trauma fragility fracture, especially in young men. In patients with a chronic disease in which low testosterone and hypogonadism are common (e.g., T2DM, CKD, chronic obstructive pulmonary disease [COPD]), serum testosterone should be measured if symptoms or signs indicative of androgen deficiency (e.g., sexual dysfunction, weakness) are present.113
Seminal Fluid Analysis
If infertility is a main complaint, whether or not androgen deficiency is also present, seminal fluid analysis should be performed to determine the presence and degree of impairment of sperm production. Seminal fluid analyses are performed on ejaculated semen samples obtained by masturbation after a standardized period (usually 48 hours) of abstinence from ejaculation. Semen collection after withdrawal of the penis from the partner just before ejaculation during sexual intercourse (coitus interruptus) is usually incomplete and is not recommended, but it may be an option if masturbation is not possible or is not permitted for personal or religious reasons. Seminal fluid analyses should be performed in a specialized laboratory that employs standardized procedures, such as those outlined by the World Health Organization (WHO), and is certified and qualified to carry out these procedures.29
According to recently revised WHO criteria based on approximately 1800 to 1900 men from 14 countries whose partners became pregnant in 12 months or less (Table 19-6), the lower limit of normal sperm concentration is 15 million/mL; semen volume is 1.5 mL; total sperm count is 39 million per ejaculate; total sperm motility (both progressive and nonprogressive) is 40% and progressive sperm motility is 32%; and the percentage of sperm with normal morphologic forms, using strict criteria to eliminate spermatozoa with even mild abnormalities, is at least 4%.29 Values below these lower limits may be classified as falling into the subfertile range, and such values are found in independent (unscreened) populations. In another study, the subfertile ranges were defined as follows: sperm concentration, less than 13.5 million/mL; motility, less than 32%; and strict morphologic structure, less than 9%. The respective fertile ranges were greater than 48 million/mL for sperm concentration, 63% for motility, and 12% for strict morphologic structure. Values between these ranges indicated indeterminate fertility.204
TABLE 19-6
Normal Seminal Fluid Analysis
| Parameter | Normal Value |
| Sperm concentration | ≥15 million/mL |
| Semen volume | ≥1.5 mL |
| Sperm count | ≥39 million per ejaculate |
| Sperm motility | ≥40% (progressive sperm motility >32%) |
| Sperm morphology | ≥4% normal forms (by strict criteria excluding sperm with mild abnormalities) |
Sperm counts and concentrations exhibit extreme variability (Fig. 19-20)9 for a number of reasons, including variations in sexual activity and abstinence, completeness of collection, recent illness (especially febrile illness) that may suppress spermatogenesis, and lifestyle factors such as frequent hot tub use. Therefore, at least two or three seminal fluid analyses, separated by at least 2 weeks, should be performed to assess sperm production adequately. Also, to assess motility, freshly collected semen (within 1 hour of ejaculation) should be used, necessitating collection at or near the laboratory in which the analysis is to be performed.
Gonadotropin Measurements
Androgen Deficiency and Impaired Sperm Production.
The diagnosis of hypogonadism is confirmed in men with symptoms and signs consistent with androgen deficiency and in whom low testosterone levels are found on at least two occasions. Testosterone levels should not be measured shortly after an acute illness, medication use, or nutritional deficiency that could transiently lower testosterone. Furthermore, an accurate assay of low free or bioavailable testosterone should be performed for men who have conditions that alter SHBG or a total testosterone level near the lower limit of normal. In men with confirmed hypogonadism, measurements of serum gonadotropins, LH, and FSH should be measured to distinguish primary from secondary hypogonadism (see Fig. 19-19).113
Men with primary hypogonadism caused by a disorder of the testis have low serum testosterone in association with elevated LH and FSH levels as a result of reduced negative feedback suppression of gonadotropin secretion by testosterone and inhibin B (Fig. 19-21).9 In contrast, men with secondary hypogonadism caused by a disorder of the pituitary or the hypothalamus (or both) have low testosterone in association with low gonadotropin levels or inappropriately normal LH and FSH given the presence of low testosterone levels (Fig. 19-22).9 In most local clinical laboratories, LH and FSH are measured by newer-generation nonradioactive immunoassays that have sufficient sensitivity to distinguish between normal and low concentrations.
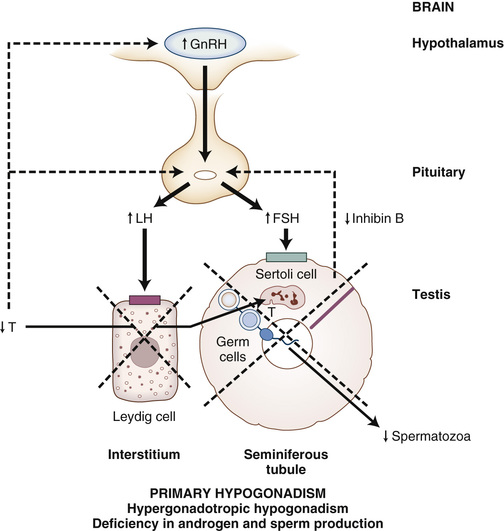
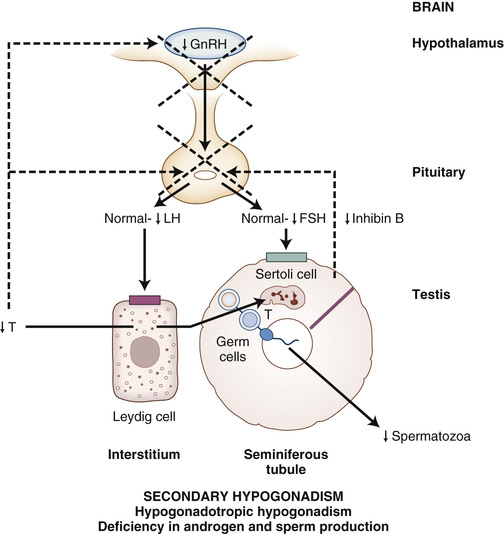
Aging, some systemic illnesses (e.g., hemochromatosis), and certain medications (e.g., glucocorticoids) may cause defects in both the testes and the hypothalamus or pituitary, resulting in combined primary and secondary hypogonadism. In most cases, a hormonal pattern consistent with either primary or secondary hypogonadism predominates. For example, men with hemochromatosis have defects in both the pituitary and the testes due to iron overload, but they usually have low testosterone and gonadotropin levels, consistent mostly with secondary hypogonadism. Men with late-stage CKD have both testis and hypothalamic-pituitary dysfunction but usually have low testosterone and elevated LH and FSH concentrations, the latter mostly due to reduced clearance of gonadotropins by the kidney. However, in the presence of comorbid illness, nutritional deficiency, or certain medications, men with CKD may have suppression of gonadotropins and testosterone into the normal range and hormonal pattern consistent with secondary hypogonadism. Some men have more than one disorder influencing the gonadal axis, one affecting the testis and another affecting the hypothalamus or pituitary. This may result in a hormonal pattern that is predominantly consistent with either primary or secondary hypogonadism or in a combined pattern (e.g., very low testosterone levels with only slightly elevated or high-normal gonadotropin levels that are lower than expected given the presence of very low testosterone concentrations).
Distinguishing primary from secondary hypogonadism helps to define the specific cause of hypogonadism and has important clinical and therapeutic implications.9 Secondary hypogonadism may be caused by a destructive process in the pituitary or hypothalamus, such as a pituitary adenoma. A large pituitary adenoma (macroadenoma) may cause space-occupying tumor mass effects such as headaches, visual field defects, hydrocephalus, or cerebrospinal fluid rhinorrhea, or it may result in impaired or excessive secretion of some anterior pituitary hormones, leading to clinical manifestations and therapeutic implications beyond the treatment of androgen deficiency alone. Secondary hypogonadism may be caused by disorders that are transient, such as an acute illness, certain medications (e.g., opioids, glucocorticoids), or malnutrition associated with illness. In such cases, androgen deficiency may resolve with treatment of and recovery from the illness or malnutrition or discontinuation of the offending medication. Finally, in men with secondary hypogonadism who have gonadotropin deficiency but otherwise normal testes, gonadotropin or GnRH treatment may be used to stimulate spermatogenesis and androgen production and to restore fertility in men who wish to father children. In contrast, infertility in men with primary hypogonadism is not treatable with hormone therapy.
Isolated Impairment of Sperm Production or Function.
Most men with isolated impairment of sperm production have low sperm counts or abnormalities in sperm motility or morphologic appearance (or both) but no clinical manifestations of androgen deficiency and normal levels of testosterone and gonadotropins. Most men with isolated impairment of sperm production or function are classified as having primary hypogonadism with an isolated defect in the seminiferous tubule compartment of the testes (Table 19-7); in such cases, there is no response to gonadotropin treatment, as there is in secondary hypogonadism. Men with severe seminiferous tubule failure and azoospermia or severe oligozoospermia may demonstrate a selective elevation in FSH levels as a result in reduced negative feedback from inhibin B with normal LH levels (Fig. 19-23).9 Occasionally, isolated impairment in sperm production is caused by gonadotropin deficiency (i.e., secondary hypogonadism) (Table 19-8); this may occur in men who are taking high doses of testosterone and in those who have androgen-secreting tumors, congenital adrenal hyperplasia, or, rarely, isolated FSH deficiency.
TABLE 19-7
Causes of Primary Hypogonadism
| Common Causes | Uncommon Causes |
| Androgen Deficiency and Impairment of Sperm Production | |
| Congenital or Developmental Disorders | |
| Klinefelter syndrome (XXY) and variants | Myotonic dystrophy |
| Uncorrected cryptorchidism | |
| Noonan syndrome | |
| Bilateral congenital anorchia | |
| Polyglandular autoimmune syndrome | |
| Testosterone biosynthetic enzyme defects | |
| CAH (testicular adrenal rest tumors) | |
| Complex genetic syndromes | |
| Down syndrome | |
| LH receptor mutation | |
| Acquired Disorders | |
| Bilateral surgical castration or trauma | Orchitis |
| Drugs (spironolactone, ketoconazole, abiraterone, enzalutamide, alcohol, chemotherapy agents) | |
| Ionizing radiation | |
| Systemic Disorders | |
| Chronic liver disease (hepatic cirrhosis)* | Malignancy (lymphoma, testicular cancer) |
| Chronic kidney disease* | Sickle cell disease* |
| Aging* | Spinal cord injury |
| Vasculitis (polyarteritis) | |
| Infiltrative disease (amyloidosis, leukemia) | |
| Isolated Impairment of Sperm Prduction or Function | |
| Congenital or Developmental Disorders | |
| Cryptorchidism | Myotonic dystrophy |
| Varicocele | Sertoli cell–only syndrome |
| Y chromosome microdeletions | Primary ciliary dyskinesia |
| Down syndrome | |
| FSH receptor mutation | |
| Acquired Disorders | |
| Orchitis | Environmental toxins |
| Ionizing radiation | |
| Chemotherapy agents | |
| Thermal trauma | |
| Systemic Disorders | |
| Acute febrile illness | Spinal cord injury |
| Malignancy (testicular cancer, Hodgkin disease)* | |
| Idiopathic azoospermia or oligozoospermia | |
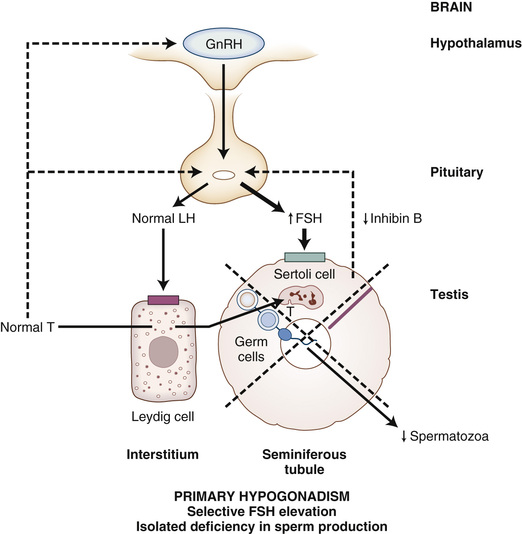
TABLE 19-8
Causes of Secondary Hypogonadism
| Common Causes | Uncommon Causes |
| Androgen Deficiency and Impairment of Sperm Production | |
| Congenital or Developmental Disorders | |
| Constitutional delayed puberty | IHH and variants |
| Hemochromatosis | IHH |
| Kallmann syndrome | |
| Congenital adrenal hypoplasia | |
| Isolated LH deficiency, LHβ mutations | |
| Complex genetic syndromes | |
| Acquired Disorders | |
| Hypopituitarism | |
| Pituitary or hypothalamic tumor | |
| Surgical hypophysectomy, pituitary or cranial irradiation | |
| Vascular compromise, traumatic brain injury | |
| Granulomatous or infiltrative disease | |
| Infection | |
| Pituitary stalk disease | |
| Lymphocytic or autoimmune hypophysitis | |
| Systemic Disorders | |
| Glucocorticoid excess (Cushing syndrome)* | Chronic systemic illness* |
| Chronic organ failure* | Spinal cord injury |
| Chronic liver disease (hepatic cirrhosis), chronic kidney disease, chronic lung disease, chronic heart failure | Transfusion-related iron overload (β-thalassemia) |
| Chronic systemic illness* | Sickle cell disease |
| Type 2 diabetes mellitus | Cystic fibrosis |
| Malignancy | |
| Rheumatic disease (rheumatoid arthritis) | |
| HIV disease | |
| Starvation,* malnutrition,* eating disorders, endurance exercise | |
| Morbid obesity, obstructive sleep apnea | |
| Acute and critical illness | |
| Aging* | |
| Isolated Impairment of Sperm Production or Function | |
| Congenital or Developmental Disorders | |
| Congenital adrenal hyperplasia (21-hydroxylase deficiency, 11β-hydroxylase deficiency) | |
| Isolated FSH deficiency, FSHβ mutations | |
| Acquired Disorders | |
| Testosterone, androgenic anabolic steroids | Androgen- or hCG-secreting tumors |
| Malignancy (Hodgkin disease, testicular cancer)* | Hyperprolactinemia |
Men with nonfunctioning or gonadotropin-secreting pituitary tumors often have secondary hypogonadism with clinical manifestations of androgen deficiency and low testosterone levels.53 Many of these men secrete excessive amounts of intact FSH and biologically inactive free α-, FSHβ-, and LHβ-subunits but rarely intact LH. Therefore, a gonadotropin-secreting pituitary tumor should be suspected in a man who has clinical manifestations of androgen deficiency, low testosterone, and elevated FSH but normal or low LH (or, rarely, elevated LH but normal or low FSH), which is an atypical gonadotropin pattern for men with androgen deficiency.
Rarely, disorders of androgen action or androgen resistance manifest in adults (Table 19-9). These men usually present with clinical manifestations similar to those of men with mild androgen deficiency, usually with an almost normal male phenotype and frequently with varying degrees of hypospadias, cryptorchidism, scrotal abnormalities, or impairment in sperm production. Usually, both serum testosterone and gonadotropin levels are elevated (Fig. 19-24).9
TABLE 19-9
Causes of Androgen Resistance
| Common Causes | Uncommon Causes |
| Congenital or Developmental Disorders | |
| Kennedy disease (spinal and bulbar muscular atrophy) | |
| Partial androgen insensitivity syndrome (AR mutations) | |
| 5α-Reductase type 2 deficiency | |
| Complete androgen insensitivity syndrome (female phenotype) | |
| Acquired Disorders | |
| AR antagonists (bicalutamide, nilutamide) | Celiac disease |
| Drugs (spironolactone, cyproterone acetate, marijuana, histamine 2 receptor antagonists) | |

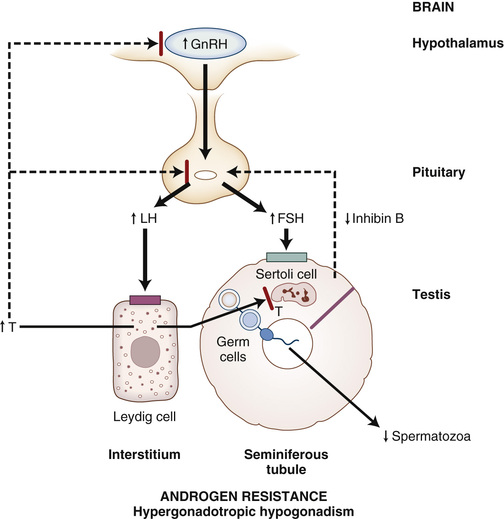
Further Evaluation
Testosterone measurements and assessment of sperm production, combined with measurement of gonadotropin levels, allow classification of the causes of male hypogonadism into primary or secondary hypogonadism and subclassification of the latter into disorders causing both androgen deficiency and impairment in sperm production and those causing isolated impairment of sperm production or function (see Tables 19-7 and 19-8).
Once hypogonadism has been classified as primary or secondary hypogonadism, further evaluation includes a history (including medication review), physical examination, and laboratory testing to identify a specific cause or causes of hypogonadism. For example, in men with primary hypogonadism and suggestive clinical manifestations such as very small testes and gynecomastia, low or low-normal testosterone, azoospermia, and markedly elevated gonadotropins, a karyotype may be obtained to confirm the diagnosis of Klinefelter syndrome.
In men with secondary hypogonadism, further evaluation may include measurements of serum prolactin (in almost all cases) to exclude hyperprolactinemia; iron saturation and ferritin to screen for hereditary hemochromatosis, especially in men with other manifestations of iron overload (e.g., liver failure, diabetes, and CHF) and in young men with unexplained selective gonadotropin deficiency; further testing to exclude excessive secretion or deficiency of anterior pituitary hormones; and magnetic resonance imaging (MRI) of the sella turcica to exclude a pituitary or hypothalamic tumor or infiltrative disease. Computed tomography of the sella turcica usually detects a pituitary macroadenoma but is less sensitive than sella MRI in detecting smaller tumors and infiltrative disease.113
It is not cost effective to perform sella MRI in all men with secondary hypogonadism. This modality should be reserved for men with severe androgen deficiency (e.g., serum testosterone <150 ng/dL) due to secondary hypogonadism, particularly with distinctly low gonadotropin levels205,206; discordant LH and FSH levels associated with androgen deficiency; hyperprolactinemia (especially with prolactin levels >100 to 200 ng/mL); clinical and biochemical evidence of excessive secretion of other pituitary hormones (e.g., free α-subunit secretion, Cushing syndrome, acromegaly), panhypopituitarism, or diabetes insipidus; tumor mass effects, such as severe headache, visual field defects, or visual impairment; and no obvious functional cause of secondary hypogonadism (e.g., morbid obesity, long-acting opioid, or chronic high-dose glucocorticoid therapy).
In men who have severe androgen deficiency caused by primary or secondary hypogonadism or who have sustained a low-trauma or fragility fracture, DXA scanning to assess BMD should be performed to exclude osteopenia or osteoporosis.113