Breasts and Regional Lymphatics
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
Structure and Function
The breasts, or mammary glands, are present in both females and males, although in men they are rudimentary throughout life. The female breasts are accessory reproductive organs whose function is to produce milk for nourishing the newborn.
Surface Anatomy
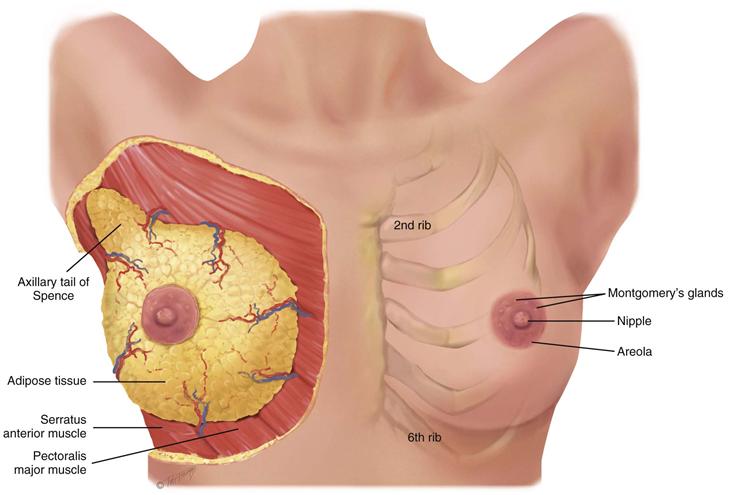
The breasts lie anterior to the pectoralis major and serratus anterior muscles (Fig. 17-1). The breasts are located between the second and sixth ribs, extending from the side of the sternum to the midaxillary line. The superior lateral corner of breast tissue, called the axillary tail of Spence, projects up and laterally into the axilla.
17-1 Copyright © (2006) © Pat Thomas, 2010.
The nipple is just below the center of the breast. It is rough, round, and usually protuberant; its surface looks wrinkled and indented with tiny milk duct openings. The areola surrounds the nipple for a 1- to 2-cm radius. In the areola are small elevated sebaceous glands, called Montgomery’s glands. These secrete a protective lipid material during lactation. The areola also has smooth muscle fibers that cause nipple erection when stimulated. Both the nipple and areola are more darkly pigmented than the rest of the breast surface; the color varies from pink to brown depending on the person’s skin color and parity (condition of giving birth).
Internal Anatomy
The breast is composed of (1) glandular tissue, (2) fibrous tissue including the suspensory ligaments, and (3) adipose tissue (Fig. 17-2). The glandular tissue contains 15 to 20 lobes radiating from the nipple, and these are composed of lobules. Within each lobule are clusters of alveoli that produce milk. Each lobe empties into a lactiferous duct. The 15 to 20 lactiferous ducts form a collecting duct system converging toward the nipple. There, the ducts form ampullae, or lactiferous sinuses, behind the nipple, which are reservoirs for storing milk.
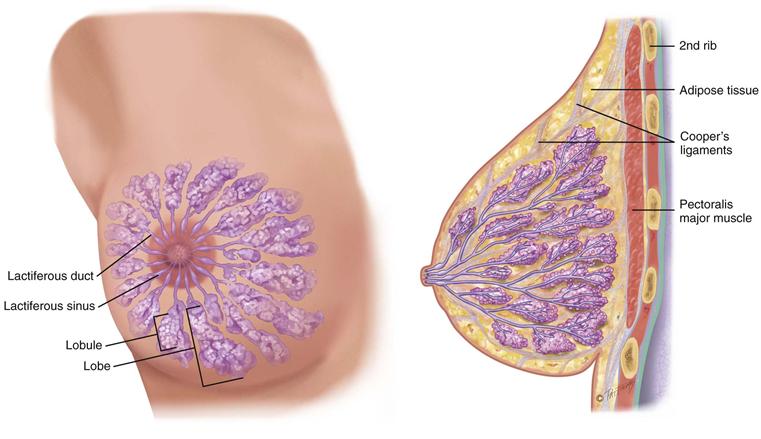
17-2 Copyright © (2006) © Pat Thomas, 2010.
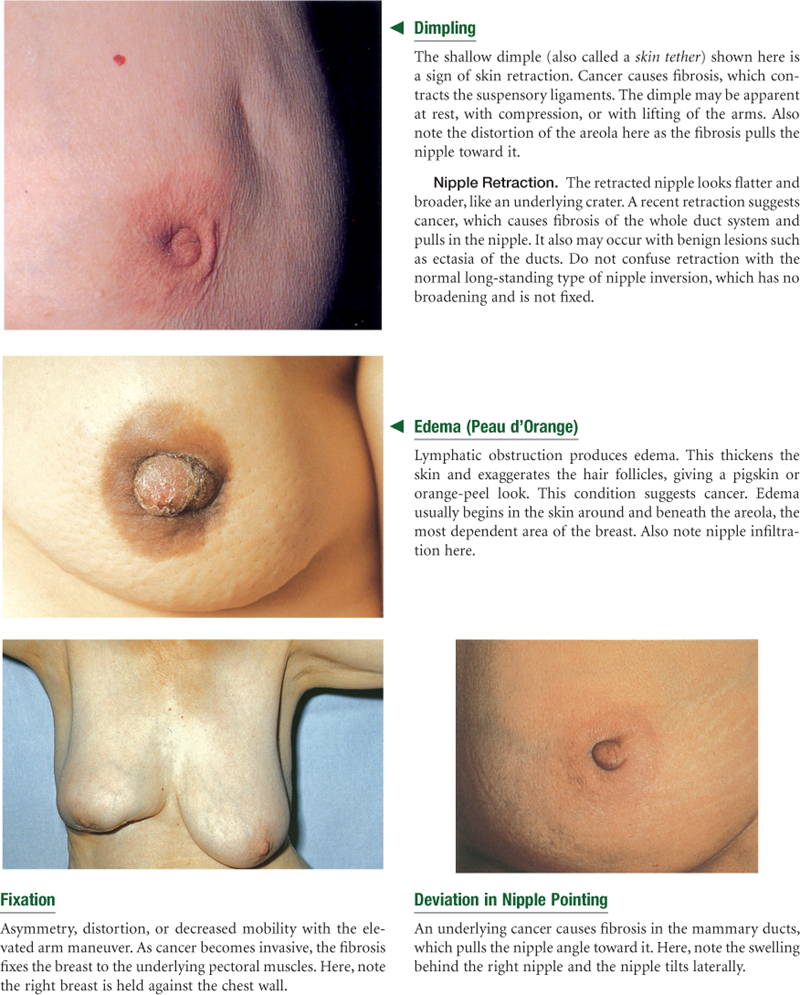
The suspensory ligaments, or Cooper’s ligaments, are fibrous bands extending vertically from the surface to attach on chest wall muscles. These support the breast tissue. They become contracted in cancer of the breast, producing pits or dimples in the overlying skin.
The lobes are embedded in adipose tissue. These layers of subcutaneous and retromammary fat actually provide most of the bulk of the breast. The relative proportion of glandular, fibrous, and fatty tissue varies depending on age, cycle, pregnancy, lactation, and general nutritional state.
The breast may be divided into four quadrants by imaginary horizontal and vertical lines intersecting at the nipple (Fig. 17-3). This makes a convenient map to describe clinical findings. In the upper outer quadrant, note the axillary tail of Spence, the cone-shaped breast tissue that projects up into the axilla, close to the pectoral group of axillary lymph nodes. The upper outer quadrant is the site of most breast tumors.
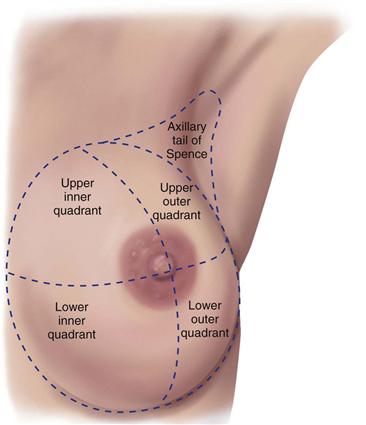
17-3
Lymphatics
The breast has extensive lymphatic drainage. Most of the lymph, more than 75%, drains into the ipsilateral (same side) axillary nodes. Four groups of axillary nodes are present (Fig. 17-4):
1. Central axillary nodes—high up in the middle of the axilla, over the ribs and serratus anterior muscle. These receive lymph from the other three groups of nodes.
2. Pectoral (anterior)—along the lateral edge of the pectoralis major muscle, just inside the anterior axillary fold.
3. Subscapular (posterior)—along the lateral edge of the scapula, deep in the posterior axillary fold.
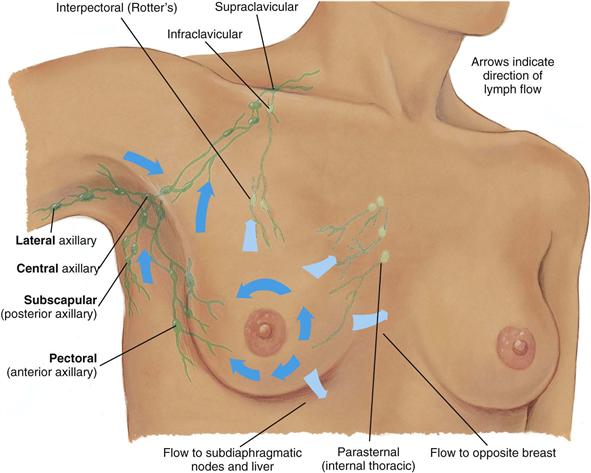
17-4
From the central axillary nodes, drainage flows up to the infraclavicular and supraclavicular nodes.
A smaller amount of lymphatic drainage does not take these channels but, instead, flows directly up to the infraclavicular group, deep into the chest, or into the abdomen, or directly across to the opposite breast.
 Developmental competence
Developmental competence
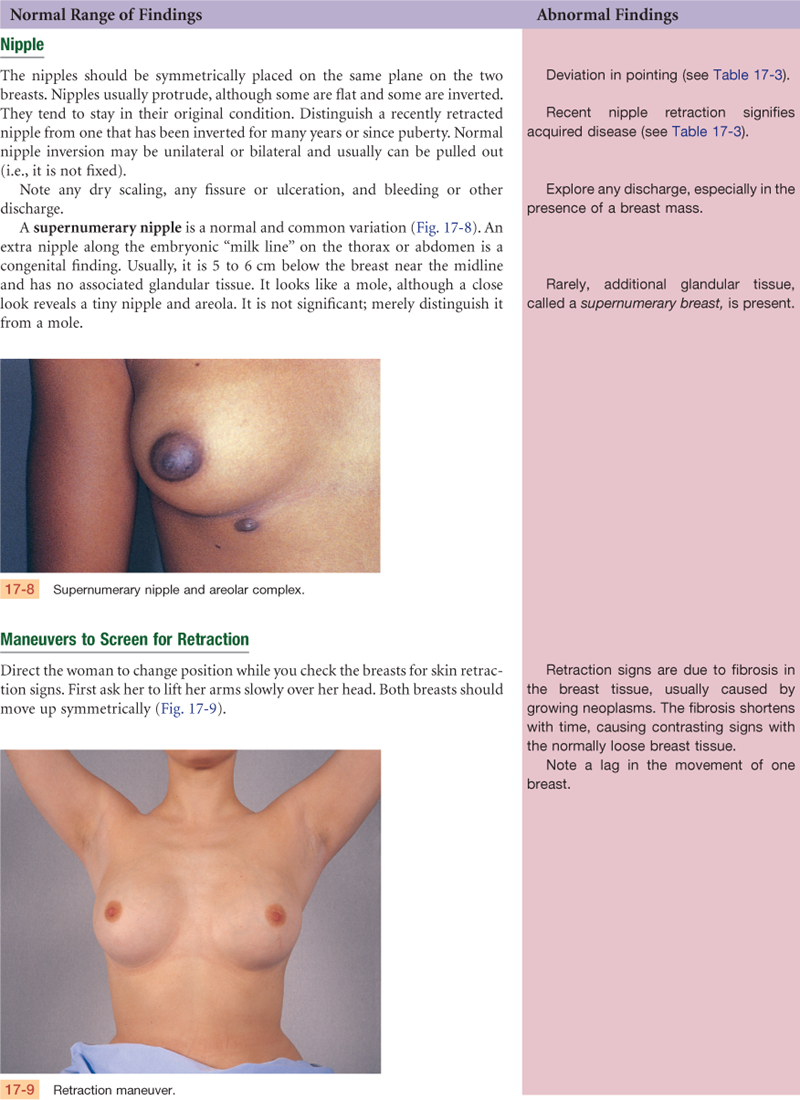
During embryonic life, ventral epidermal ridges, or “milk lines,” are present and curve down from the axilla to the groin bilaterally (Fig. 17-5). The breast develops along the ridge over the thorax, and the rest of the ridge usually atrophies. Occasionally a supernumerary nipple (i.e., an extra nipple) persists and is visible somewhere along the track of the mammary ridge (see Fig. 17-8).
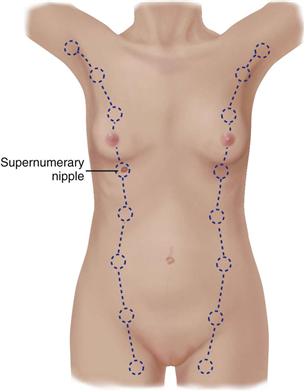
17-5
At birth, the only breast structures present are the lactiferous ducts within the nipple. No alveoli have developed. Little change occurs until puberty.
The Adolescent
At puberty, the estrogen hormones stimulate breast changes. The breasts enlarge, mostly as a result of extensive fat deposition. The duct system also grows and branches, and masses of small, solid cells develop at the duct endings. These are potential alveoli.
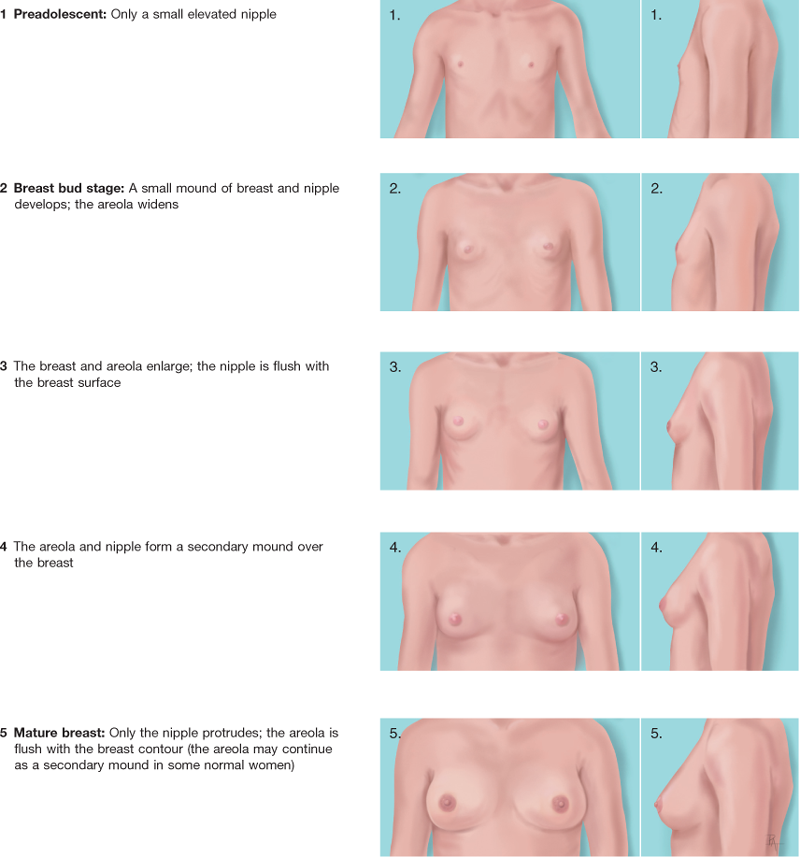
A 1997 study of 17,077 girls in the United States ages 3 through 12 years indicates that puberty is occurring earlier than classically used norms.14 The onset of breast development occurred at an average (mean) age between 8 and 9 years for African-American girls and by 10 years for white girls. Occasionally, one breast may grow faster than the other, producing a temporary asymmetry. This may cause some distress; reassurance is necessary. Tenderness is common also. Although the age of onset varies widely, the five stages of breast development follow this classic description of sexual maturity rating, or Tanner staging (Table 17-1).
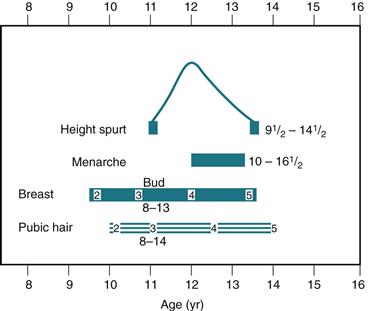
Full development from stage 2 to stage 5 takes an average of 3 years, although the range is 1.5 to 6 years. During this time, pubic hair develops, and axillary hair appears 2 years after the onset of pubic hair. The beginning of breast development precedes menarche (beginning of menstruation) by about 2 years. Menarche occurs in breast development stage 3 or 4, usually just after the peak of the adolescent growth spurt around age 12 years. Note the relationship of these events (Fig. 17-6). This aids in assessing the development of adolescent girls and increases their knowledge about their own development.
Breasts of the nonpregnant woman change with the ebb and flow of hormones during the monthly menstrual cycle. Nodularity increases from midcycle up to menstruation. During the 3 to 4 days before menstruation, the breasts feel full, tight, heavy, and occasionally sore. The breast volume is smallest on days 4 to 7 of the menstrual cycle.
The Pregnant Woman
During pregnancy, breast changes start during the second month and are an early sign of pregnancy for most women. Pregnancy stimulates the expansion of the ductal system and supporting fatty tissue as well as development of the true secretory alveoli. Thus the breasts enlarge and feel more nodular. The nipples are larger, darker, and more erectile. The areolae become larger and grow a darker brown as pregnancy progresses, and the tubercles become more prominent. (The brown color fades after lactation, but the areolae never return to the original color.) A venous pattern is prominent over the skin surface (see Fig. 29-4 on p. 808).
After the fourth month, colostrum may be expressed. This thick, yellow fluid is the precursor for milk, containing the same amount of protein and lactose but practically no fat. The breasts produce colostrum for the first few days after delivery. It is rich with antibodies that protect the newborn against infection, so breastfeeding is important. Milk production (lactation) begins 1 to 3 days postpartum. The whitish color is from emulsified fat and calcium caseinate.
The Aging Woman
After menopause, ovarian secretion of estrogen and progesterone decreases, which causes the breast glandular tissue to atrophy. This is replaced with fibrous connective tissue. The fat envelope atrophies also, beginning in the middle years and becoming marked in the eighth and ninth decades. These changes decrease breast size and elasticity so the breasts droop and sag, looking flattened and flabby. Drooping is accentuated by the kyphosis in some older women.
The decreased breast size makes inner structures more prominent. A breast lump may have been present for years but is suddenly palpable. Around the nipple, the lactiferous ducts are more palpable and feel firm and stringy because of fibrosis and calcification. The axillary hair decreases.
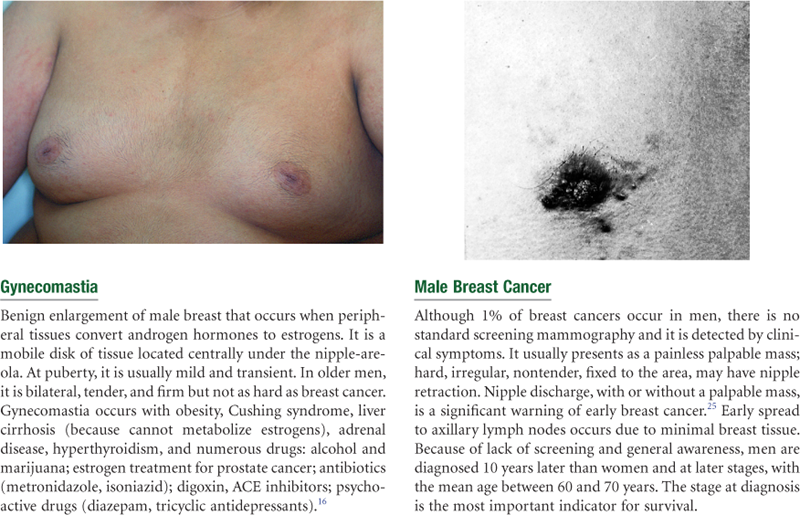
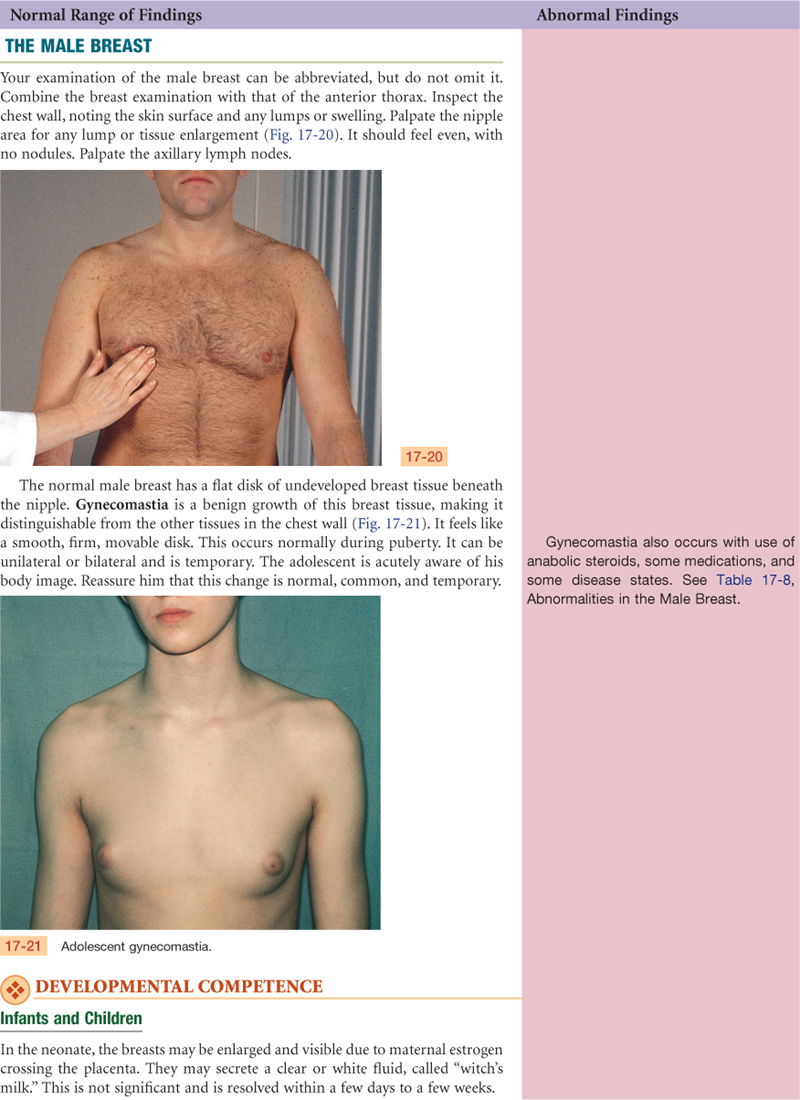
The Male Breast
The male breast is a rudimentary structure consisting of a thin disk of undeveloped tissue underlying the nipple. The areola is well developed, although the nipple is relatively very small. During adolescence, it is common for the breast tissue to temporarily enlarge, producing gynecomastia (see Fig. 17-21). This condition is usually temporary, but reassurance is necessary for the adolescent male, whose attention is riveted on his body image. Gynecomastia may reappear in the aging male and may be due to testosterone deficiency.
 Culture and genetics
Culture and genetics
The timing of puberty is influenced by genetic and environmental factors, with genetics determining about 50% to 80% of the variation.13 The 1997 study of 17,077 young girls showed that African-American girls begin puberty about 1 to 1.5 years earlier than white girls and start menstruating about 8.5 months earlier.14 The onset of breast development occurs at an average age of 8.87 years for African-American girls and 10 years for white girls (Hispanic ethnicity occurs in both groups). Menses begins at an average age of 12.16 years for African-American girls and at almost 13 years for white girls. For over 50 years, precocious puberty had been defined as breast budding occurring younger than 8 years; however, the 1997 study showed 6.5% of white girls and 27.2% of African-American girls had breast or pubic hair development before age 8 years.14
As to environmental factors, obesity may contribute to early onset of puberty, as girls with early onset of breast budding have higher body mass index (BMI) scores than age-matched girls without budding.14 It is not clear whether this correlation applies among different racial and ethnic groups.33 Among girls with normal BMIs, signs of puberty occurred before 8 years in fewer than 5% of white girls, 12% of Black girls, and 19% of Mexican-American girls.28 But girls with overweight or obese BMI levels had a significantly higher occurrence of early breast budding and early menarche. Thus the ongoing epidemic of childhood obesity in the United States is a major determinant of early-age pubertal milestones.28
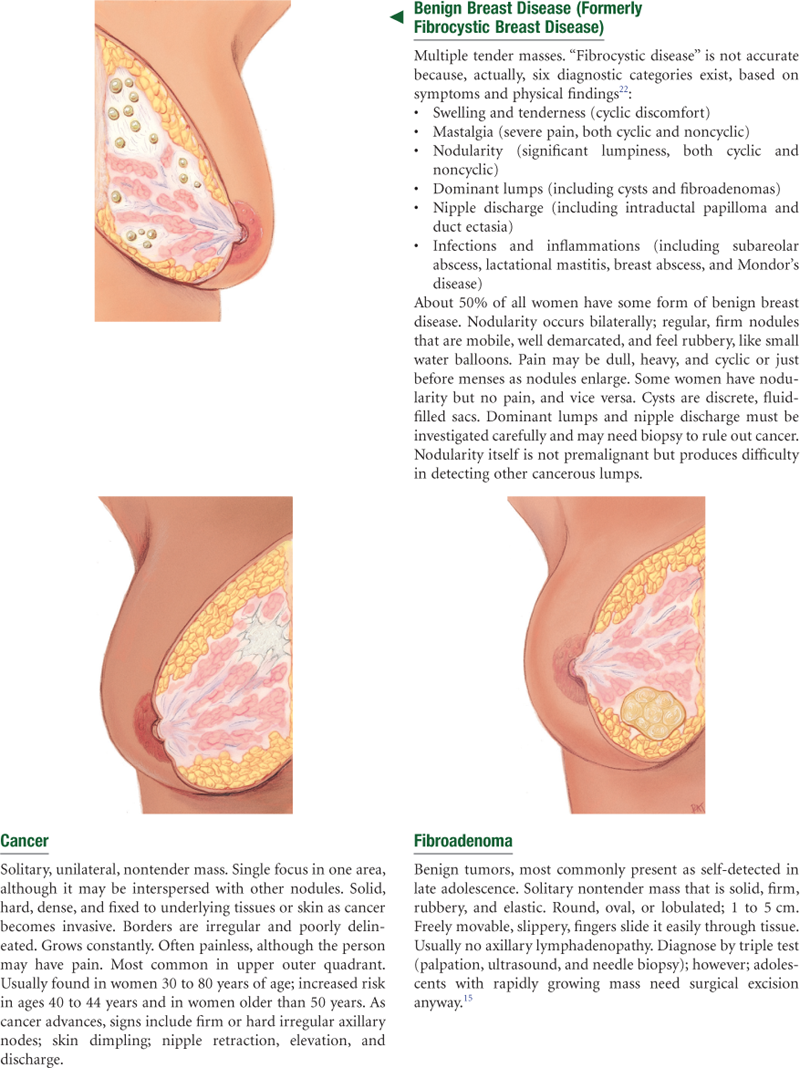
Breast Cancer
The genetic contribution to breast cancer involves specific gene mutations at the BRCA1 and BRCA2 locations. Women with these mutations are at increased risk for breast and ovarian cancer.
White women have a higher incidence of breast cancer than African-American women starting at age 45 years. In contrast, African-American women have a higher incidence before age 45 years and they are more likely to die of their disease at every age.4 Women from Asian-American, Hispanic, and American-Indian groups have a lower incidence and death rates from breast cancer than whites and African Americans have.
The disparity in death rates may be due, in part, to insufficient use of screening measures and lack of access to health care. The good news is that, in the United States, the percentage of women ages 40 years and older who report having had a mammogram in the past 2 years increased from 29% in 1987 to 70% in 2000. However, women least likely to have had a recent mammogram include those with less than a high school education, with no health insurance, or who are recent immigrants.4 Data are inconsistent regarding African-American and white women’s utilization of mammograms; some studies report higher rates among African Americans, and some report lower rates.10 Somewhat fewer Hispanic women than white women (42.3% vs. 44.6%) report adhering to screening mammography guidelines, and Hispanic women were more likely than white women to have never had a mammogram in their lifetime. Low-income women have multiple barriers to screening mammography, including lack of insurance coverage, lack of access to care, not having a regular health care provider, and lack of comprehensive breast cancer knowledge, not merely screening awareness.2 Culturally sensitive interventions aimed at increasing screening measures must occur if we are to serve all women.
Regarding spread of disease, African-American women were significantly more likely to be diagnosed with regional or distant breast cancer (compared with local) than were white women.5 African-American women had lower 5-year survival rates, which is due, in part, to underuse of mammography, taking longer time to medical consultation after a diagnosis of breast cancer, less likely to receive surgical removal of their tumors, noncompliance with planned therapy, and incomplete adherence to treatment regimens.5
Diet is another environmental factor in breast cancer risk, noted because breast cancer incidence varies among countries. One study of a large French postmenopausal cohort examined two dietary patterns: (1) “alcohol/Western” (meat products, French fries, appetizers, rice/pasta, potatoes, pizza, pies, canned fish, eggs, alcohol, cakes, mayonnaise, butter/cream); and (2) “healthy/Mediterranean” (vegetables, fruits, seafood, olive oil, sunflower oil). The first pattern had a positive association with breast cancer risk, especially with estrogen or progesterone receptor positive tumors. Other diet studies have found that only alcohol intake, being overweight, and weight gain have shown consistent and positive associations with breast cancer risk.21 Premenopausal African-American women had lower cancer risk when following a “prudent” diet (whole grains, vegetables, fruit, fish), especially those with a BMI below 25. Although the evidence is not consistent to generate preventive diet guidelines for all women, some subgroups of women may benefit from a prudent diet.
Subjective Data
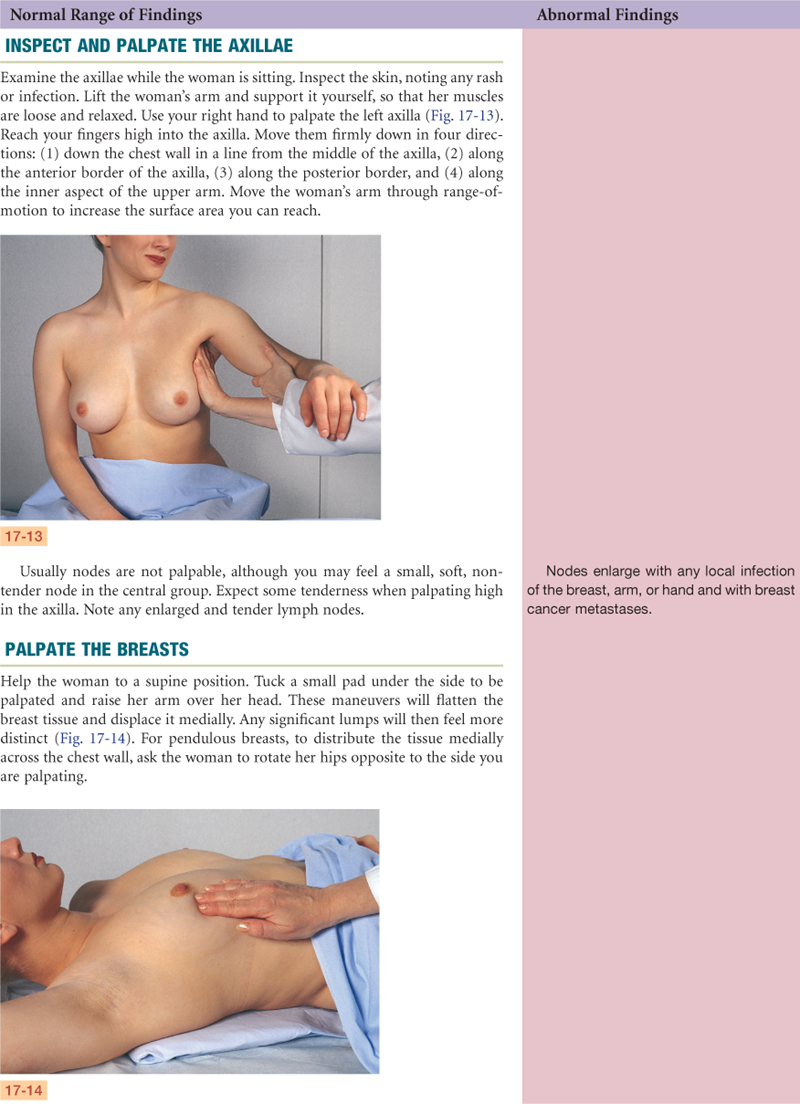
Axilla
In Western culture, the female breasts signify more than their primary purpose of lactation. Women are surrounded by messages that feminine norms of beauty and desirability are enhanced by and depend on the size of the breasts and their appearance. Women leaders have tried to refocus this attitude, stressing women’s self-worth as individual human beings, not as stereotyped sexual objects. The intense cultural emphasis is slow to change, and the breasts still are crucial to a woman’s self-concept and her perception of her femininity. Matters pertaining to the breast affect the body image and generate deep emotional responses.
This emotionality may take strong forms that you observe as you discuss the woman’s history. One woman may be acutely embarrassed talking about her breasts, as evidenced by lack of eye contact, minimal response, nervous gestures, or inappropriate humor. Another woman may talk wryly and disparagingly about the size or development of her breasts. A young adolescent is acutely aware of her own development in relation to her peers. Or, a woman who has found a breast lump may come to you with fear, high anxiety, and even panic. Although many breast lumps are benign, women initially assume the worst possible outcome—cancer, disfigurement, and death. While you are collecting the subjective data, tune in to cues for these behaviors that call for a straightforward and reasoned attitude.


TABLE 17-2
| Risk Factors That Cannot Be Changed | Lifestyle-Related Risk Factors |
| Female gender, age >50 years Personal history of breast cancer Mutation of BRCA1 and BRCA2 genes First-degree relative with breast cancer (mother, sister, daughter) High breast tissue density Biopsy-confirmed atypical hyperplasia High-dose radiation to chest Early menarche (<12 years) or late menopause (>55 years) | Nulliparity or first child after age 30 years Recent oral contraceptive use Never breastfed a child Recent and long-term use of estrogen and progestin Alcohol intake of ≥1 drink daily Obesity (especially after menopause) and high-fat diet Physical inactivity |
Data adapted from American Cancer Society, 2010.
Objective Data
| Preparation | Equipment needed |
| The woman is sitting up facing the examiner. Use a short gown, open at the back, and lift it up to the woman’s shoulders during inspection. During palpation when the woman is supine, cover one breast with the gown while examining the other. Be aware that many women are embarrassed to have their breasts examined; use a sensitive but matter-of-fact approach. After your examination, be prepared to teach the woman breast self-examination. | Small pillow Ruler marked in centimeters Pamphlet or teaching aid for BSE |
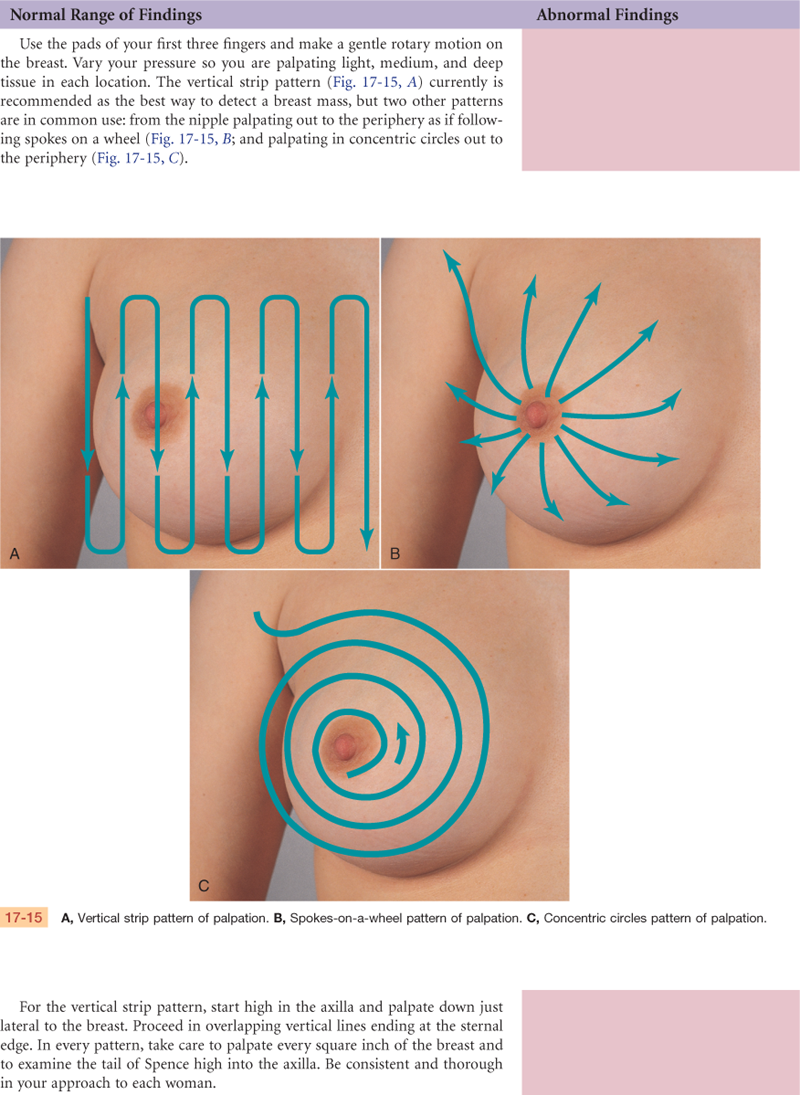
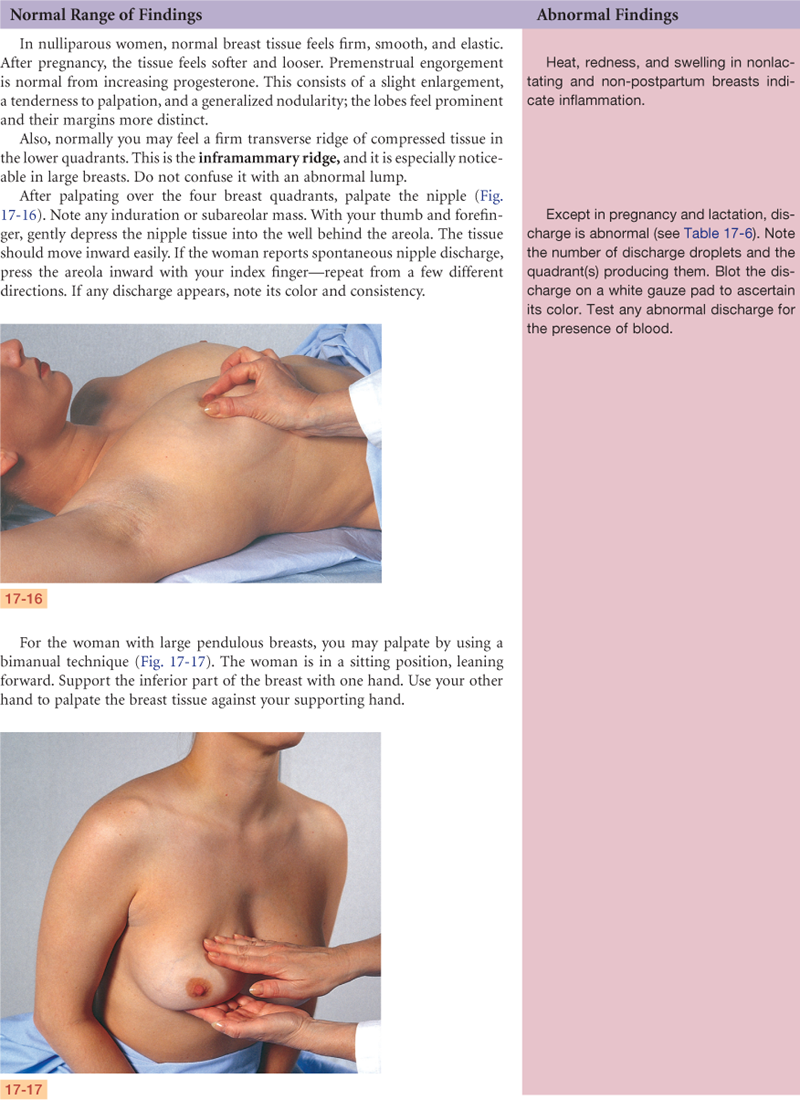
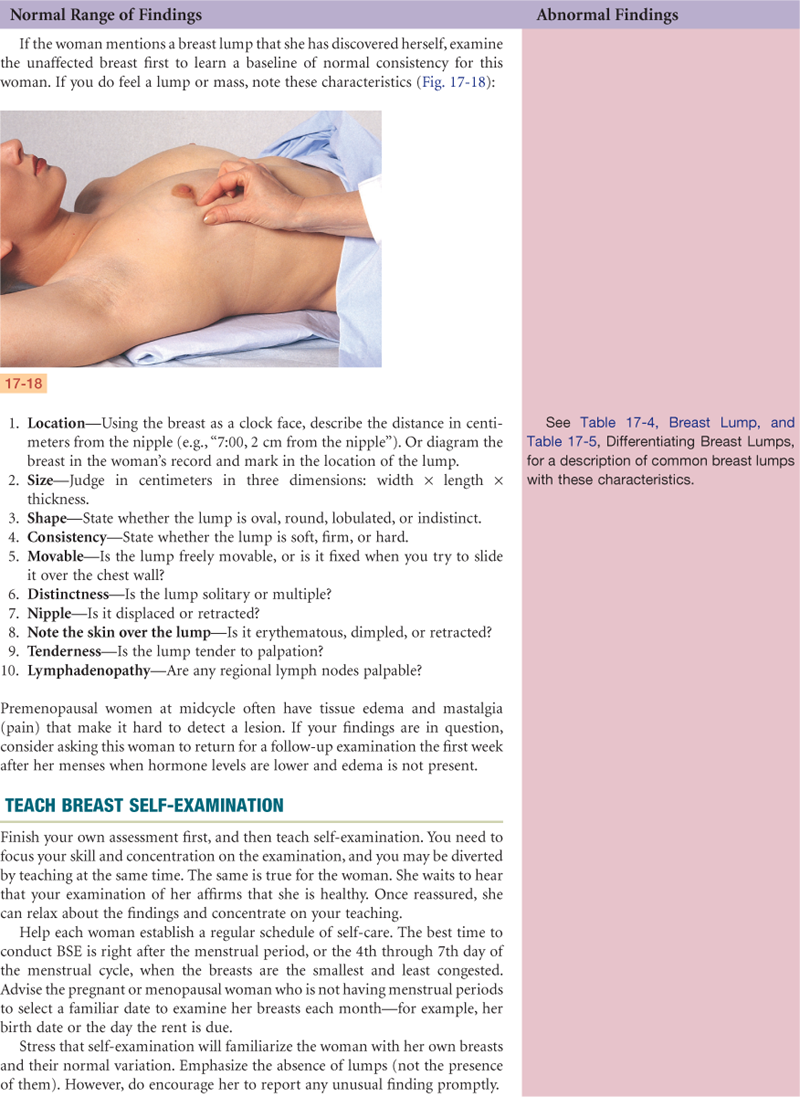


 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
FEMALE
Subjective
States no breast pain, lump, discharge, rash, swelling, or trauma. No history of breast disease herself; does have mother with fibrocystic disease. No history of breast surgery. Never been pregnant. Performs BSE monthly.
Objective
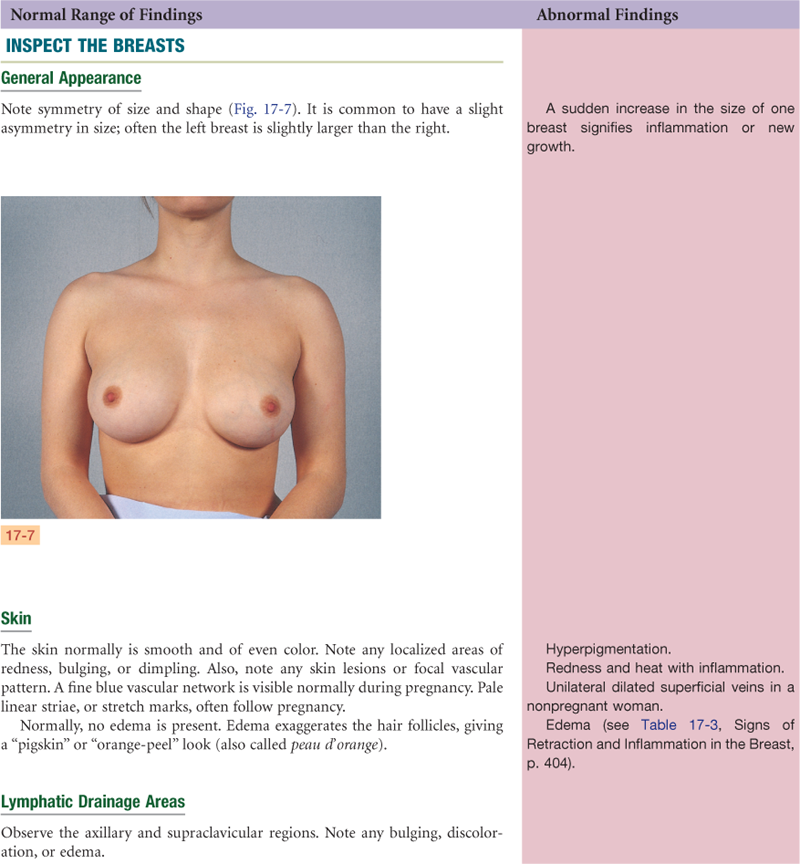
Inspection: Breasts symmetric. Skin smooth with even color and no rash or lesions. Arm movement shows no dimpling or retractions. No nipple discharge, no lesions.
Palpation: Breast contour and consistency firm and homogeneous. No masses or tenderness. No lymphadenopathy.
Assessment
Healthy breast structure
Has knowledge of breast self-exam
MALE
Subjective
No pain, lump, rash, or swelling.
Objective
No masses or tenderness. No lymphadenopathy.
Focused Assessment: Clinical Case Study 1
J.G. is a 32-year-old white female high school teacher, married, with no children. She reports good health until finding “lump in right breast 2 weeks ago.”
Subjective
2 weeks PTA—noticed lump in R breast on self-examination. Lump firm, nonmovable area “the size of a quarter,” in upper outer quadrant of breast, tender on touch only. No skin changes, no nipple discharge, on no medications. Last breast exam by MD 3 months before was reported normal. Did not notice lump on previous self-exam 1 month before. No history of breast disease in self or family.
2 days PTA—saw MD, who confirmed presence of lump and recommended biopsy as outpatient. Last menstrual period 1/25 (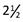 weeks PTA). States that for the past 2 days she has been so nervous that she has been unable to sleep well or to concentrate at work. “I just know it’s cancer.”
weeks PTA). States that for the past 2 days she has been so nervous that she has been unable to sleep well or to concentrate at work. “I just know it’s cancer.”
Objective
Voice trembling and breathless during history. Sitting posture stiff and rigid. BP, 148/78 mm Hg; TPR, 37°-92-16.
Inspection: Breasts symmetric, nipples everted. No skin lesions, no dimpling, no retraction, no fixation.
Palpation: Left breast firm, no mass, no tenderness, no discharge. Right breast firm, with 2 cm × 2 cm × 1 cm mass at 10 o’clock position, 5 cm from the nipple. Lump is firm, oval, with smooth discrete borders, nonmovable, tender to palpation. No other mass. No discharge. No lymphadenopathy.
Assessment
Lump in R breast
Anxiety R/T threat to health status
Focused Assessment: Clinical Case Study 2
D.B. is a 62-year-old Black female bank comptroller, married, with no children. History of hypertension, managed by diuretic medication and diet. No other health problems until yearly company physical exam 3 days PTA, when MD “found a lump in my right breast.”
Subjective
3 days PTA—MD noted lump in R breast during yearly physical exam. MD did not describe lump but told D.B. it was “serious” and needed immediate biopsy. D.B. has not felt it herself. States has noted no skin changes, no nipple discharge. No previous history of breast disease. Mother died age 54 years of breast cancer, no other relative with breast disease. D.B. has had no term pregnancies; two spontaneous abortions, ages 28, 31 years. Menopause completed at age 52 years.
Aware of BSE but has never performed it. “I feel so bad. If only I had been doing it. I should have found this myself.” Married 43 years. States husband supportive, but “I just can’t talk to him about this. I can’t even go near him now.”
Objective
Inspection: Breasts symmetric when sitting, arms down. Nipples flat. No lesions, no discharge. As lifts arms, left breast elevates, right breast stays fixed. Dimple in right breast, 9 o’clock position, apparent at rest and with muscle contraction. Leaning forward reveals left breast falls free, right breast flattens.
Palpation: Left breast feels soft and granular throughout, no mass. Right breast soft and granular, with large, stony hard mass in outer quadrant. Lump is 5 cm × 4 cm × 2 cm, at 9 o’clock position, 3 cm from nipple. Borders irregular, mass fixed to tissues, no pain with palpation.
One firm, palpable lymph node in center of right axilla. No palpable nodes on the left.
Assessment
Lump in R breast
Ineffective coping R/T effects of breast lump
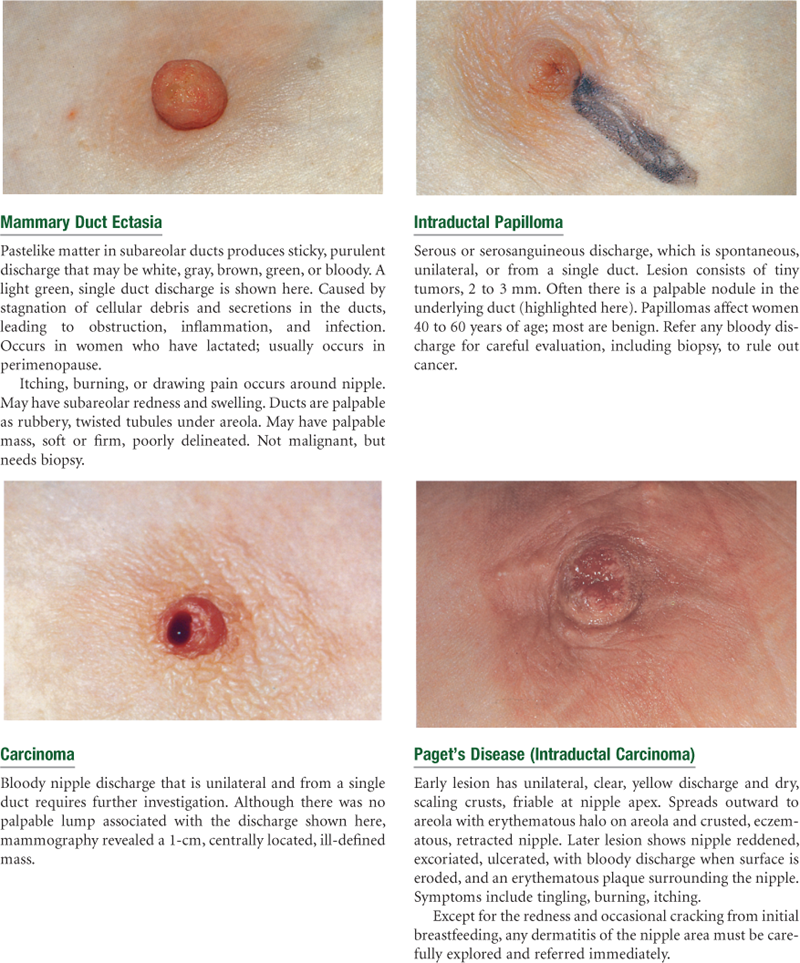
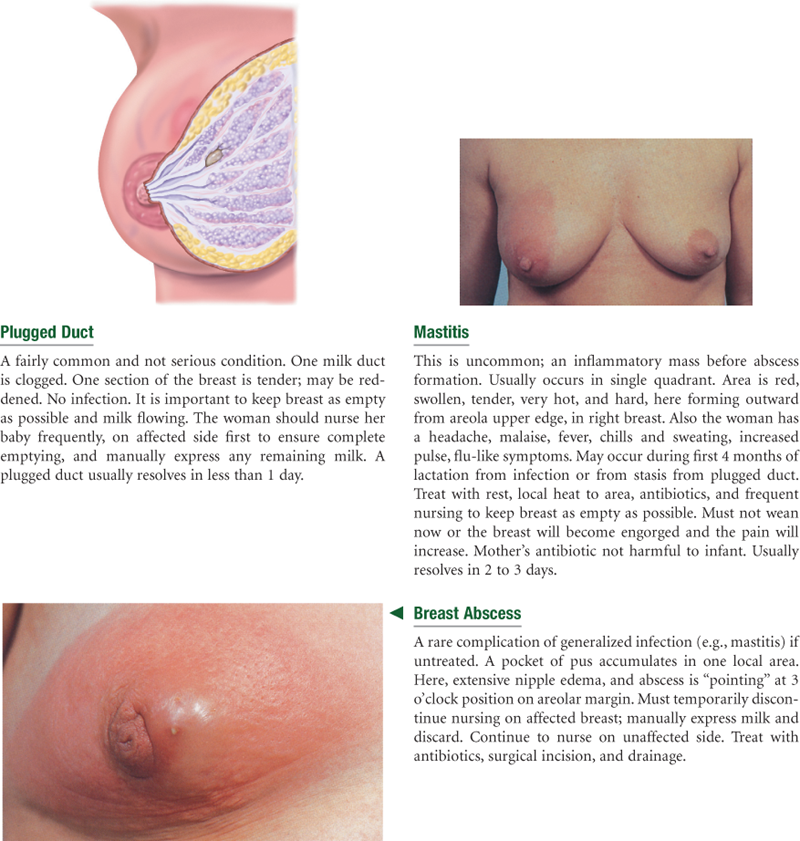
Abnormal Findings for Advanced Practice
Summary Checklist: Breasts and Regional Lymphatics Examination
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
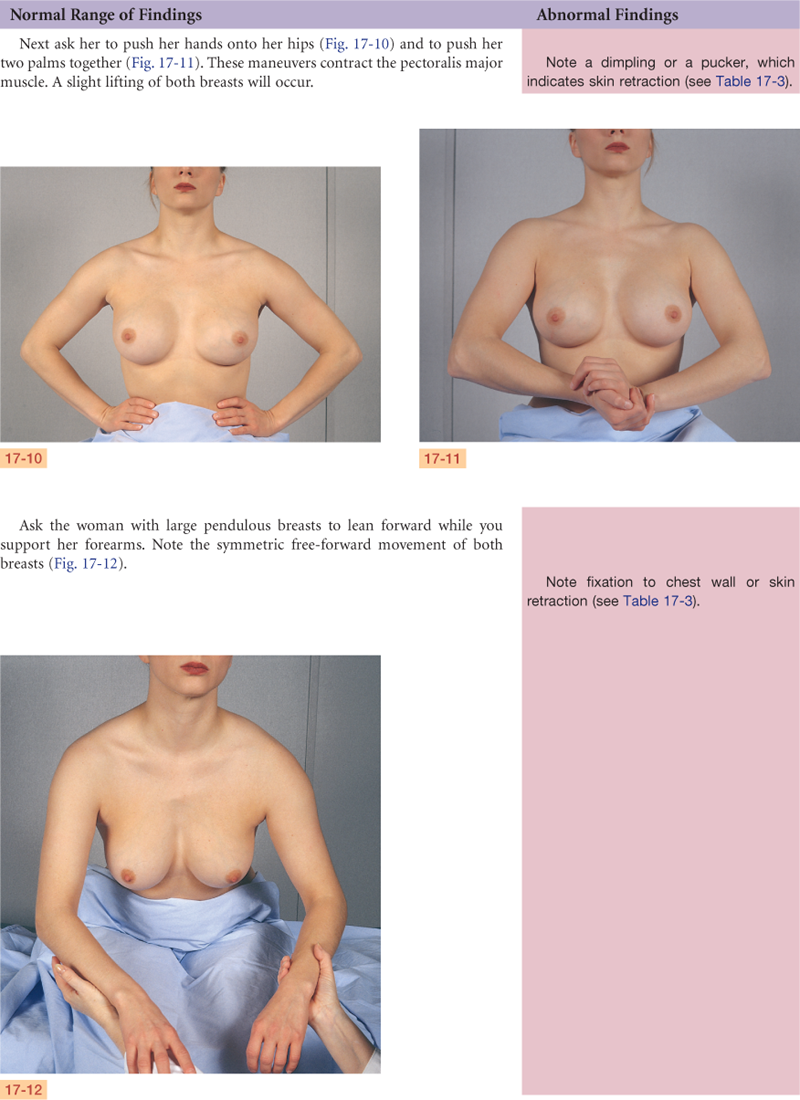
1. Inspect breasts as the woman sits, raises arms overhead, pushes hands on hips, leans forward.
2. Inspect the supraclavicular and infraclavicular areas.
3. Palpate the axillae and regional lymph nodes.
4. With woman supine, palpate the breast tissue, including tail of Spence, the nipples, and areolae.
Bibliography
1. Agurs-Collins T, Rosenberg L, Makambi K, et al. Dietary patterns and breast cancer risk in women participating in the Black Women’s Health Study. American Journal of Clinical Nutrition. 2009;90(3):621–628.
2. Ahmed NU, Ford JG, Fair AM, et al. Breast cancer knowledge and barriers to mammography in a low-income managed care population. Journal of Cancer Education. 2009;24(4):261–266.
3. Aksglaede L, Sørensen K, Peterson JH, et al. Recent decline in age at breast development: the Copenhagen Puberty Study. Pediatrics. 2009;123(5):e932–e939.
4. American Cancer Society. Breast cancer: cancer facts and figures 2009-2010. Atlanta: Author; 2010.
5. Baquet CR, Mishra SI, Commiskey P, et al. Breast cancer epidemiology in blacks and whites: disparities in incidence, mortality, survival rates and histology. Journal of the National Medical Association. 2008;100(5):480–488.
6. Barron MA, Fishel RS. Talk to your patients about breast disease. Nurse Practitioner. 2007;32(10):21–32.
7. Borrayo EA, Hines L, Byers T, et al. Characteristics associated with mammography screening among both Hispanic and non-Hispanic white women. Journal of Women’s Health. 2009;18(10):1585–1594.
8. Chiarelli AM, Majpruz V, Brown P, et al. The contribution of clinical breast examination to the accuracy of breast screening. Journal of the National Cancer Institute. 2009;101(18):1236–1243.
9. Colditz GA, Rosner B. Cumulative risk of breast cancer to age 70 years according to risk factor status: data from the Nurses’ Health Study. American Journal of Epidemiology. 2000;152:950–964.
10. Conway-Philips R, Millon-Underwood S. Breast cancer screening behaviors of African American women: a comprehensive review, analysis, and critique of nursing research. ABNF Journal. 2009;20(4):97–101.
11. Cottet V, Touvier M, Fornier A, et al. Postmenopausal breast cancer risk and dietary patterns in the E3N-EPIC Prospective Cohort Study. American Journal of Epidemiology. 2009;170(10):1257–1267.
12. DiVall SA, Radovick S. Endocrinology of female puberty. Current Opinion in Endocrinology, Diabetes, and Obesity. 2009;16(1):1–4.
13. Gajdos ZKZ, Hirschhorn JN, Palmert M. What controls the timing of puberty? An update on progress from genetic investigation. Current Opinion in Endocrinology, Diabetes, and Obesity. 2009;16(1):16–24.
14. Herman-Giddens ME, Slora EJ, Wasserman RC, et al. Secondary sexual characteristics and menses in young girls seen in office practice: a study from the Pediatric Research in Office Settings network. Pediatrics. 1997;99:505–512.
15. Jayasinghe Y, Simmons PS. Fibroadenomas in adolescence. Current Opinion in Obstetrics & Gynecology. 2009;21(5):402–406.
16. Johnson RE, Murad MH. Gynecomastia: pathophysiology, evaluation, and management. Mayo Clinic Proceedings. 2009;84(11):1010–1015.
17. Katapodi MC, Dodd MJ, Lee KA, et al. Underestimation of breast cancer risk: influence on screening behavior. Oncology Nursing Forum. 2009;36(3):306–314.
18. Kearney AJ, Murray M. Breast cancer screening recommendations: is mammography the only answer? Journal of Midwifery & Women’s Health. 2009;54(5):393–400.
19. Lee CH, Dershaw DD, Kopans D, et al. Breast cancer screening with imaging: recommendations from the Society of Breast Imaging and the ACR on the use of mammography, breast MRI, breast ultrasound, and other technologies for the detection of clinically occult breast cancer. Journal of the American College of Radiology. 2010;7(1):18–27.
20. Lindberg NM, Stevens VJ, Smith KS, et al. A brief intervention designed to increase breast cancer self-screening. American Journal of Health Promotion. 2009;23(5):320–323.
21. Lof M, Weiderpass E. Impact of diet on breast cancer risk. Current Opinion in Obstetrics & Gynecology. 2009;21(1):80–85.
22. Love S, Lindsey K. Dr Susan Love’s breast book. 4th ed. Cambridge, MA: Da Capo Lifelong Books; 2005.
23. Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Archives of Disease in Childhood. 1969;44:291–303.
24. Mellington TE, Fields MM. Targeting breast cancer with hormonal treatment options. Nurse Practitioner. 2008;33(5):17–22.
25. Morrogh M, King TA. The significance of nipple discharge of the male breast. Breast Journal. 2009;15(6):632–638.
26. Neal L, Tortorelli CL, Nassar A. Clinician’s guide to imaging and pathologic findings in benign breast disease. Mayo Clinic Proceedings. 2010;85(3):274–279.
27. Pierce JP. Diet and breast cancer prognosis: making sense of the Women’s Healthy Eating and Living and Women’s Intervention Nutrition Study Trials. Current Opinion in Obstetrics & Gynecology. 2009;21(1):86–91.
28. Rosenfield RL, Lipton RB, Drum ML. Thelarche, pubarche, and menarche attainment in children with normal and elevated body mass index. Pediatrics. 2009;123(1):84–88.
29. Schonberg M. Breast cancer screening: at what age to stop? Consultant. 2010;50(5):196–205.
30. Tanner JM. Growth at adolescence. 2nd ed. Oxford, UK: Blackwell Scientific; 1962.
31. Thind A, Diamant A, Hoq L, et al. Method of detection of breast cancer in low-income women. Journal of Women’s Health. 2009;18(11):1807–1811.
32. Thomas E. Men’s awareness and knowledge of male breast cancer. American Journal of Nursing. October 2010;110(10):32–42.
32. U.S. Department of Health and Human Services, Agency for Healthcare Research and Quality. Screening for breast cancer. Retrieved May 2010 from www.ahrq.gov/clinic/uspstf09/breastcancer/brcanrs.htm; 2009.
33. Wu T, Ronis D. Correlates of recent and regular mammography screening among Asian-American women. Journal of Advanced Nursing. 2009;65(11):2434–2446.