Male Genitourinary System
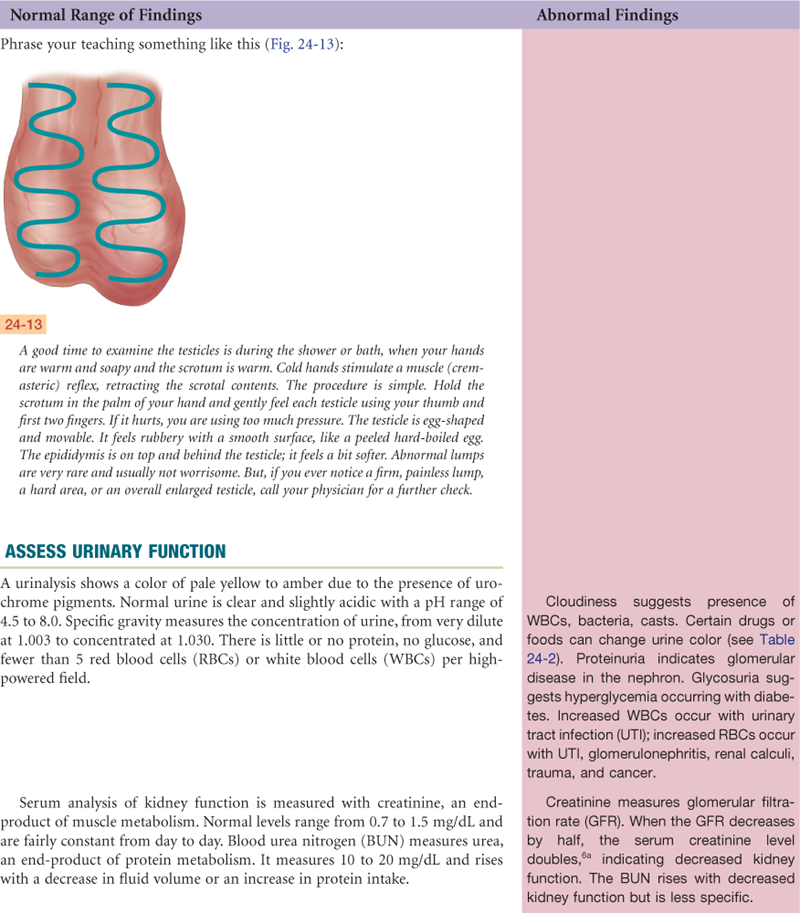
Self-Care—Testicular Self-Examination (TSE)
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
Sexually Transmitted Infections (STIs)
• Physical Examination Summary Checklist
Structure and Function
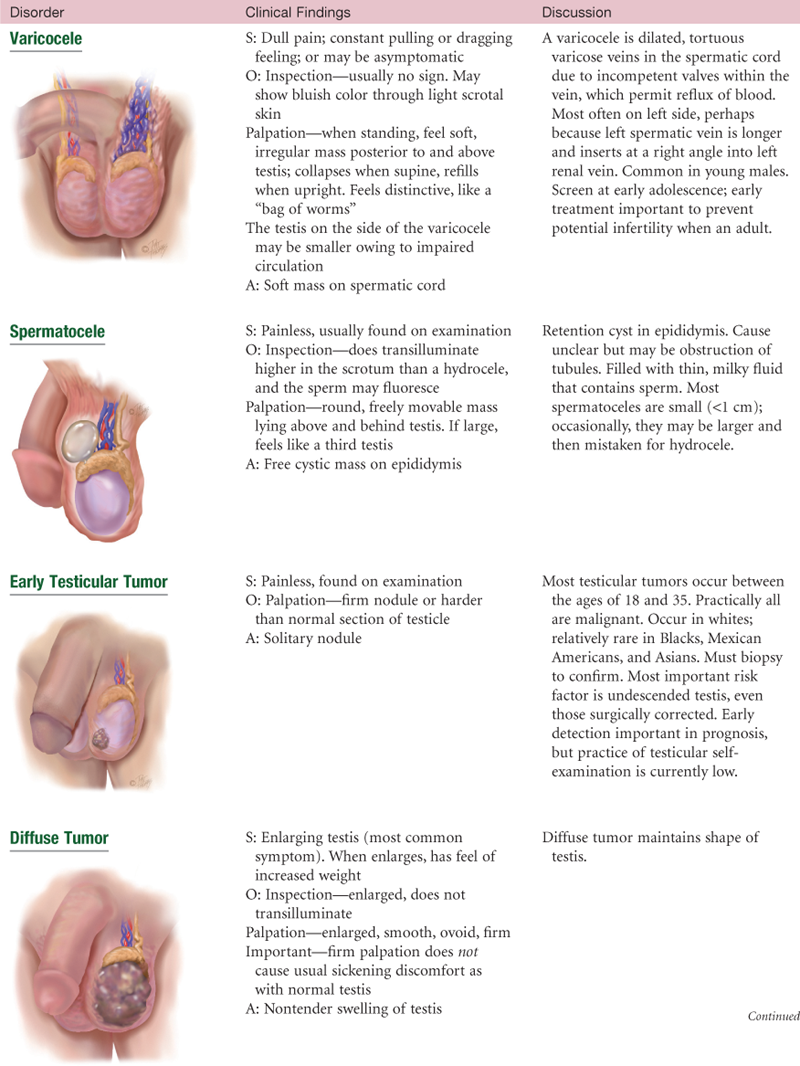
The Male Genitalia
The male genital structures include the penis and scrotum externally and the testis, epididymis, and vas deferens internally. Glandular structures accessory to the genital organs (the prostate, seminal vesicles, and bulbourethral glands) are discussed in Chapter 25.
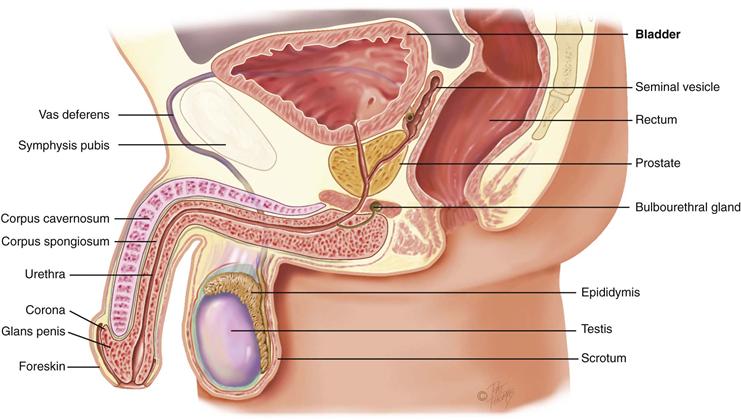
Penis
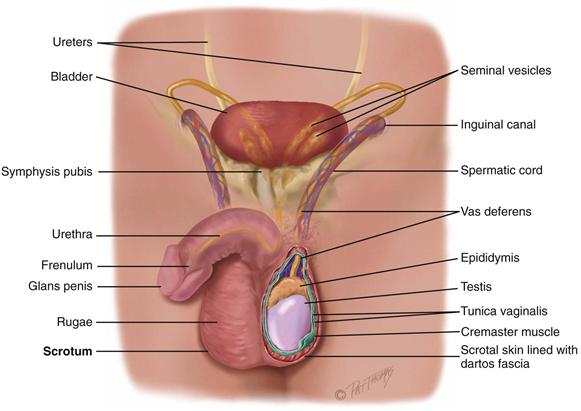
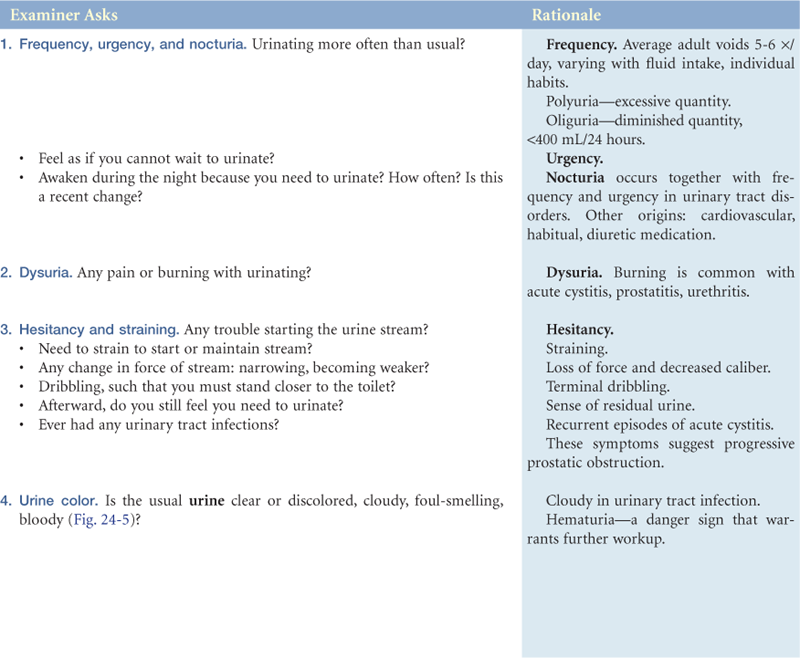
The penis is composed of three cylindric columns of erectile tissue: the two corpora cavernosa on the dorsal side and the corpus spongiosum ventrally (Fig. 24-1). At the distal end of the shaft, the corpus spongiosum expands into a cone of erectile tissue, the glans. The shoulder where the glans joins the shaft is the corona. The urethra is a conduit for both the genital and the urinary systems. It transverses the corpus spongiosum, and its meatus forms a slit at the glans tip. Over the glans, the skin folds in and back on itself forming a hood or flap. This is the foreskin or prepuce. Often, it is surgically removed shortly after birth by circumcision. The frenulum is a fold of the foreskin extending from the urethral meatus ventrally.
24-1 Copyright © (2010) © Pat Thomas, 2010.
Scrotum
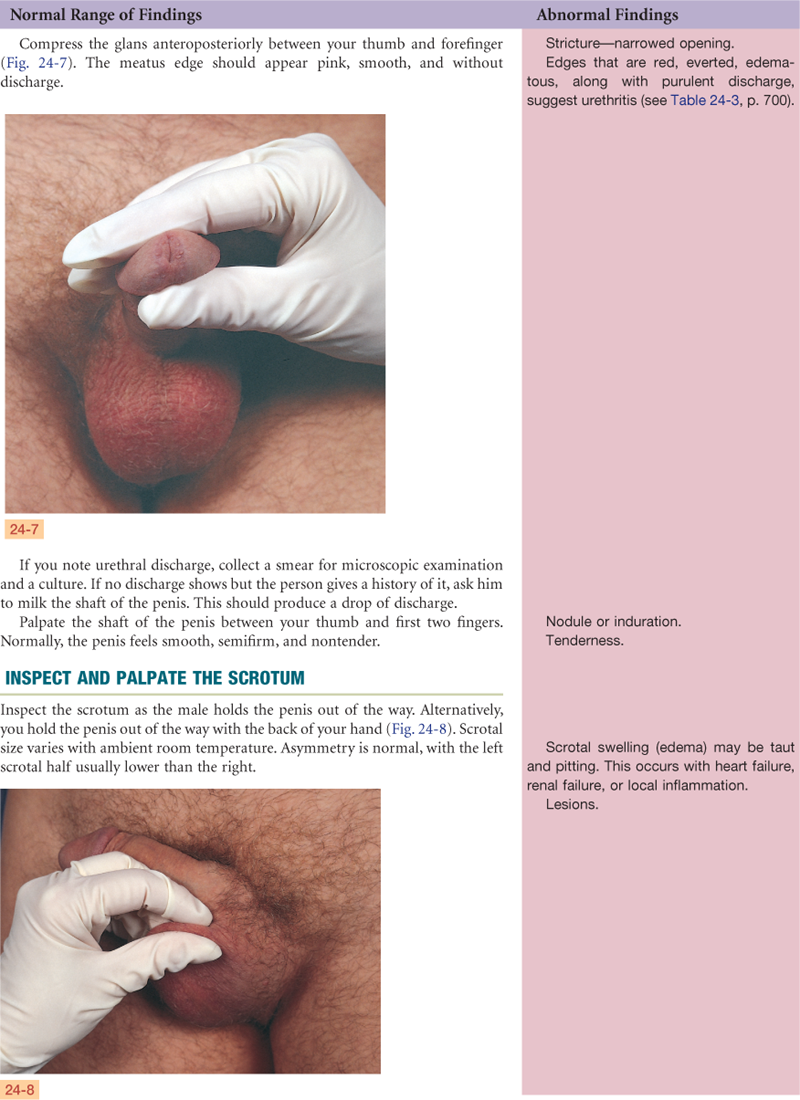
The scrotum is a loose protective sac, which is a continuation of the abdominal wall. After adolescence, the scrotal skin is deeply pigmented and has large sebaceous follicles. The scrotal wall consists of thin skin lying in folds, or rugae, and the underlying cremaster muscle. The cremaster muscle controls the size of the scrotum by responding to ambient temperature. This is to keep the testes at 3° C below abdominal temperature, the best temperature for producing sperm. When it is cold, the muscle contracts, raising the sac and bringing the testes closer to the body to absorb heat necessary for sperm viability. As a result, the scrotal skin looks corrugated. When it is warmer, the muscle relaxes, the scrotum lowers, and the skin looks smoother.
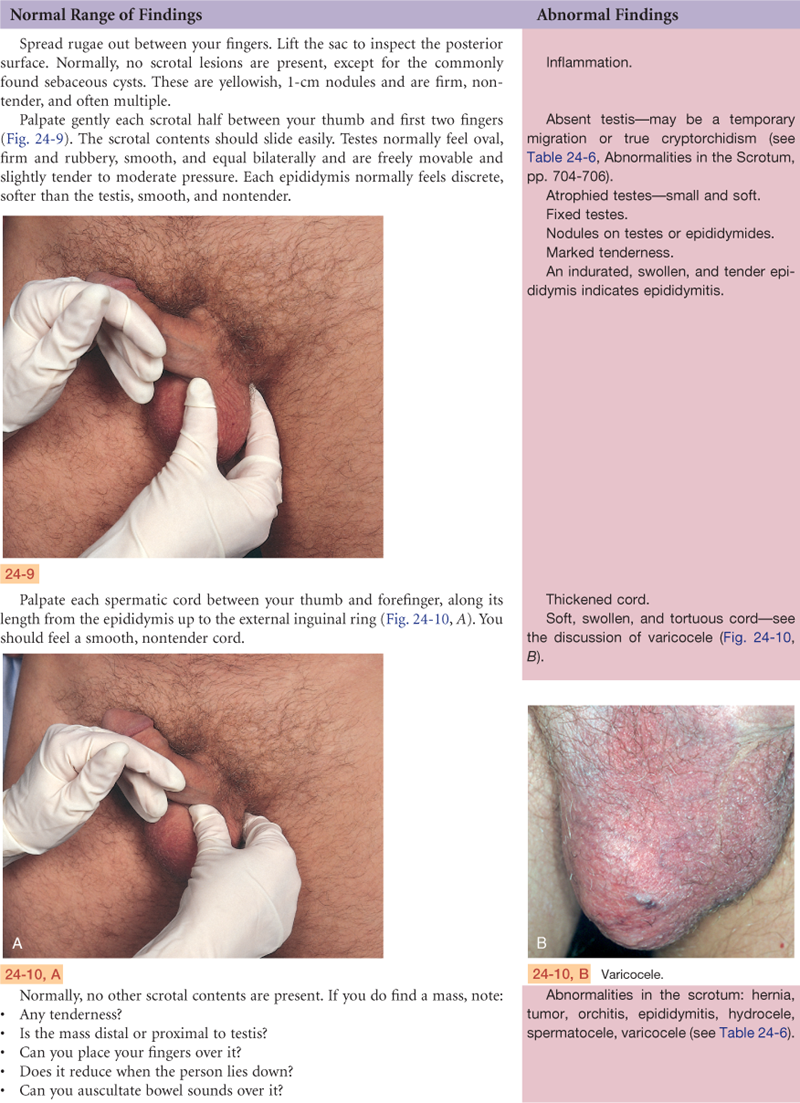
Inside, a septum separates the sac into two halves. In each scrotal half is a testis, which produces sperm. The testis has a solid oval shape, which is compressed laterally and measures 4 to 5 cm long by 3 cm wide in the adult. The testis is suspended vertically by the spermatic cord (Fig. 24-2). The left testis is lower than the right because the left spermatic cord is longer. Each testis is covered by a double-layered membrane, the tunica vaginalis, which separates it from the scrotal wall. The two layers are lubricated by fluid so that the testis can slide a little within the scrotum; this helps prevent injury.
Sperm are transported along a series of ducts. First, the testis is capped by the epididymis, which is a markedly coiled duct system and the main storage site of sperm. It is a comma-shaped structure, curved over the top and the posterior surface of the testis. Occasionally (in 6% to 7% of males), the epididymis is anterior to the testis.
The lower part of the epididymis is continuous with a muscular duct, the vas deferens. This duct approximates with other vessels (arteries and veins, lymphatics, nerves) to form the spermatic cord. The spermatic cord ascends along the posterior border of the testis and runs through the tunnel of the inguinal canal into the abdomen. Here, the vas deferens continues back and down behind the bladder, where it joins the duct of the seminal vesicle to form the ejaculatory duct. This duct empties into the urethra.
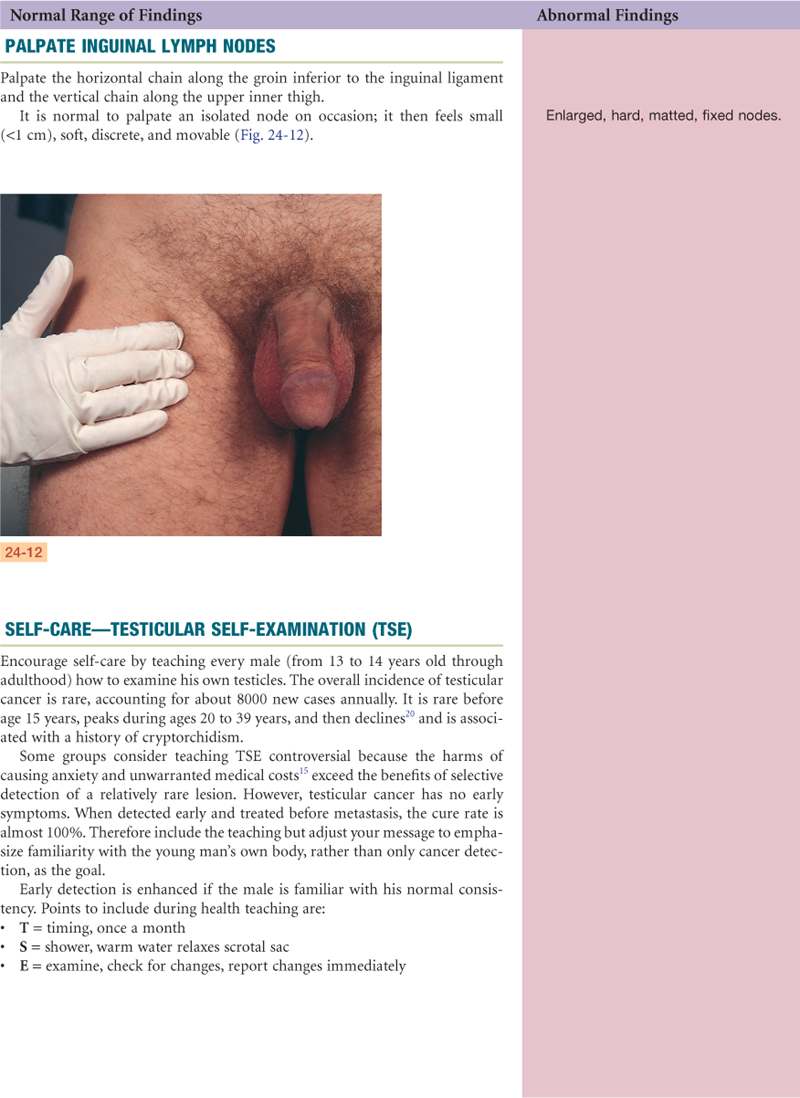
The lymphatics of the penis and scrotal surface drain into the inguinal lymph nodes, whereas those of the testes drain into the abdomen. Abdominal lymph nodes are not accessible to clinical examination.
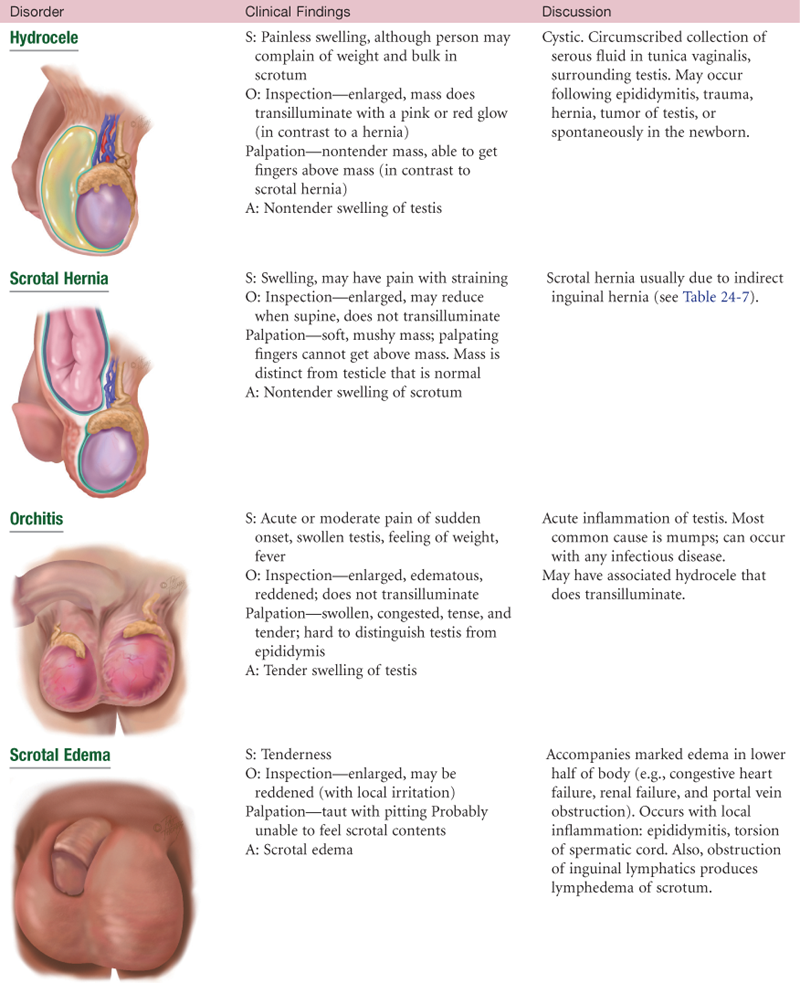
Inguinal Area
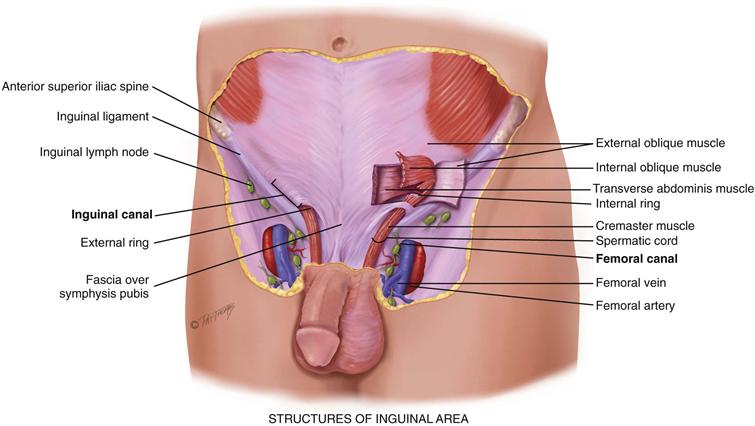
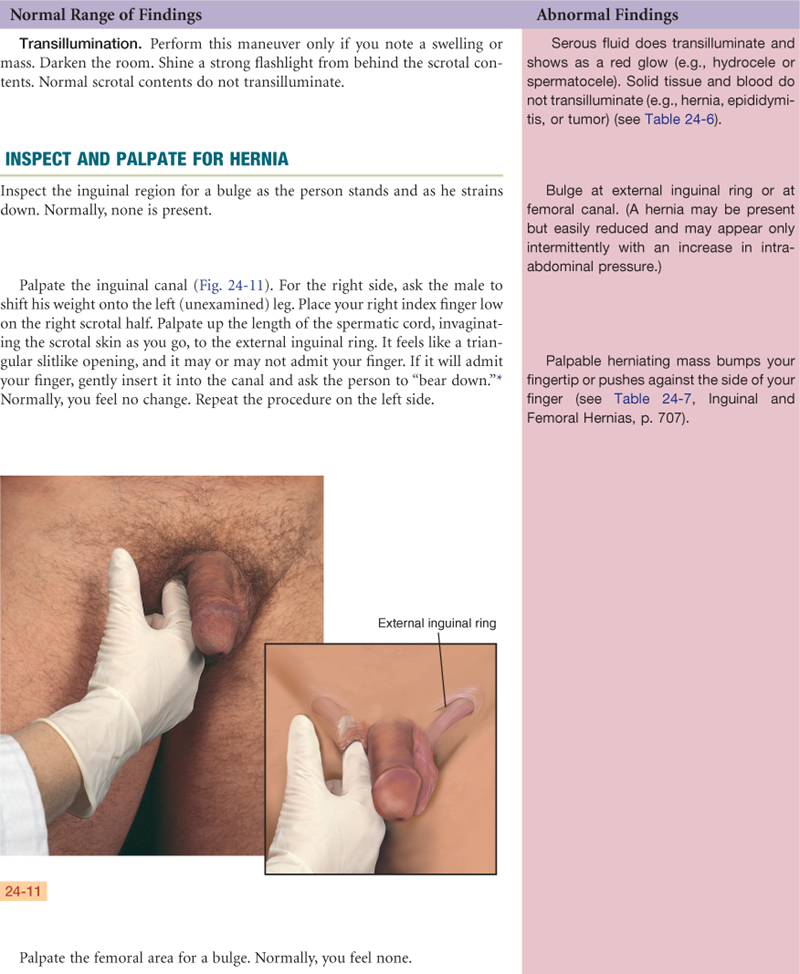
The inguinal area, or groin, is the juncture of the lower abdominal wall and the thigh (Fig. 24-3). Its diagonal borders are the anterior superior iliac spine and the symphysis pubis. Between these landmarks lies the inguinal ligament (Poupart ligament). Superior to the ligament lies the inguinal canal, a narrow tunnel passing obliquely between layers of abdominal muscle. It is 4 to 6 cm long in the adult. Its openings are an internal ring, located 1 to 2 cm above the midpoint of the inguinal ligament, and an external ring, located just above and lateral to the pubis.
24-3 Copyright © (2010) © Pat Thomas, 2010.
Inferior to the inguinal ligament is the femoral canal. It is a potential space located 3 cm medial to and parallel with the femoral artery. You can use the artery as a landmark to find this space.
Knowledge of these anatomic areas in the groin is useful because they are potential sites for a hernia, which is a loop of bowel protruding through a weak spot in the musculature.
 Developmental Competence
Developmental Competence
Infants
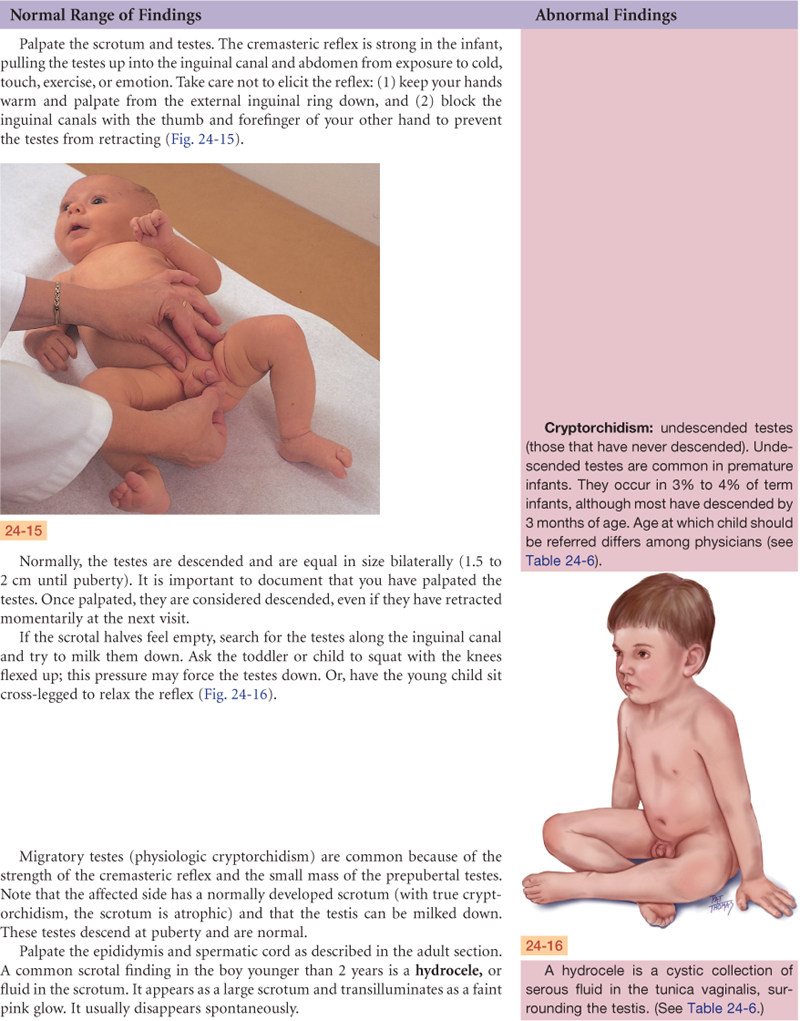
Prenatally, the testes develop in the abdominal cavity near the kidneys. During the later months of gestation, the testes migrate, pushing the abdominal wall in front of them and dragging the vas deferens, the blood vessels, and nerves behind. The testes descend along the inguinal canal into the scrotum before birth. At birth, each testis measures 1.5 to 2 cm long and 1 cm wide. Only a slight increase in size occurs during the prepubertal years.
Adolescents
Puberty begins sometime between the ages of 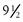 and
and 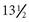 years. The first sign is enlargement of the testes. Next, pubic hair appears, and then penis size increases. The stages of development are documented in Tanner’s sexual maturity ratings (SMR) (Table 24-1).
years. The first sign is enlargement of the testes. Next, pubic hair appears, and then penis size increases. The stages of development are documented in Tanner’s sexual maturity ratings (SMR) (Table 24-1).
TABLE 24-1
Sexual Maturity Ratings (SMR) in Boys
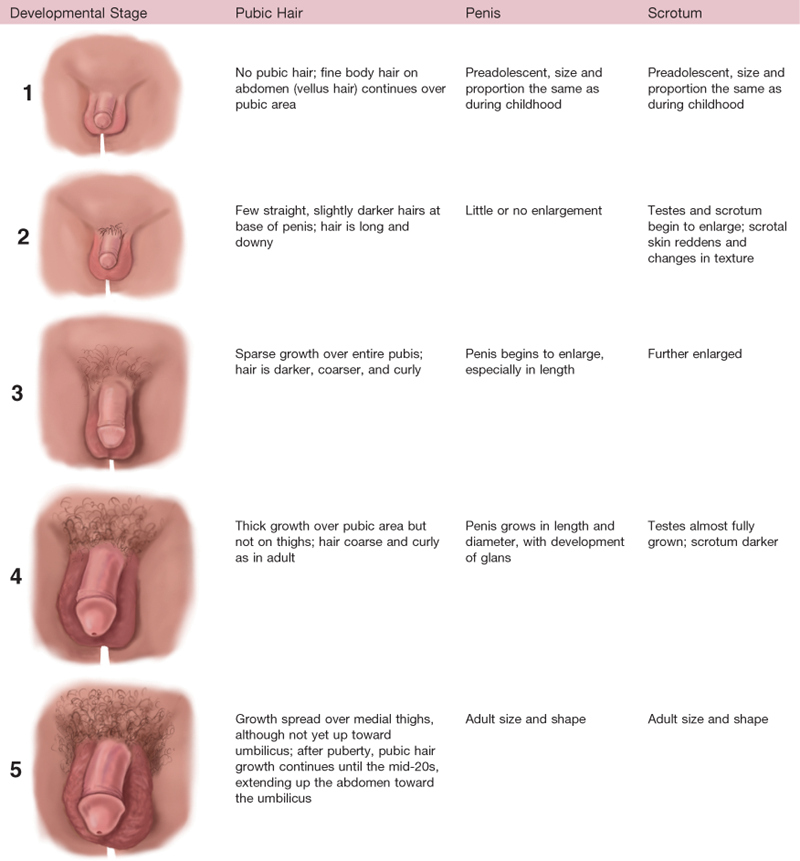
Adapted from Tanner, J.M. (1962). Growth at adolescence, Oxford, England: Blackwell Scientific Publications.
The complete change in development from a preadolescent to an adult takes around 3 years, although the normal range is 2 to 5 years (Fig. 24-4). The chart shown in Figure 24-4 is useful in teaching a boy the expected sequence of events and in reassuring him about the wide range of normal ages when these events are experienced.
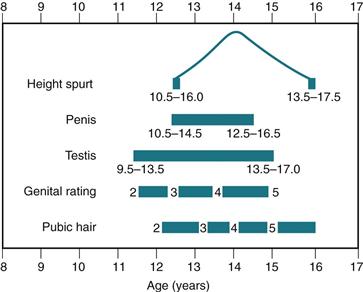
24-4
Data from the NHANES III showed that U.S. boys now have earlier genital maturation and pubic hair growth than traditional Tanner staging.13 The median age of attaining stage 2 for pubic hair development was 12 years. African-American boys showed pubic hair growth about 9 months earlier than white boys and over 1 year earlier than Mexican-American boys. Toward the end of puberty, or Tanner stage 5, African-American boys were 1 year younger than white or Mexican-American boys in completing genital development. Although these racial/ethnic differences could not be explained, the study suggested racial differences in the interactions between insulin, glucose, and androgens and racial/ethnic differences in diet, lifestyle, and exposure to environmental factors.
Adults and Aging Adults
The male does not experience a definite end to fertility as the female does. Around age 40 years, the production of sperm begins to decrease, although it continues into the 80s and 90s. After age 55 to 60 years, testosterone production declines very gradually so that resulting physical changes are not evident until later in life.
In the aging male, the amount of pubic hair decreases and the remaining hair turns gray. Penis size decreases. Due to decreased tone of the dartos muscle, the scrotal contents hang lower, the rugae decrease, and the scrotum looks pendulous. The testes decrease in size and are less firm to palpation. Increased connective tissue is present in the tubules, so these become thickened and produce less sperm.
In general, declining testosterone production leaves the older male with a slower and less intense sexual response, and an erection takes longer to develop and is less full or firm. Ejaculation is shorter and less forceful, and the volume of seminal fluid is less than when the man was younger. After ejaculation, rapid detumescence (return to the flaccid state) occurs, especially after 60 years of age. This occurs in a few seconds as compared with minutes or hours in the younger male. The refractory state (when the male is physiologically unable to ejaculate) lasts longer, from 12 to 24 hours as compared with 2 minutes in the younger male.
Sexual Expression in Later Life
Chronologic age by itself should not mean a halt in sexual activity. The just-mentioned physical changes need not interfere with the libido and pleasure from sexual intercourse. The older male is capable of sexual function as long as he is in reasonably good health and has an interested, willing partner. Even chronic illness does not mean a complete end to sexual desire or activity.
The danger is in the male misinterpreting normal age changes as a sexual failure. Once this idea occurs, it may demoralize the man and place undue emphasis on performance rather than on pleasure. In the absence of disease, a withdrawal from sexual activity may be due to loss of spouse; depression; preoccupation with work; marital or family conflict; side effects of medications such as antihypertensives, psychotropics, antidepressants, antispasmodics, sedatives, tranquilizers or narcotics, and estrogens; heavy use of alcohol; lack of privacy (living with adult children or in a nursing home); economic or emotional stress; poor nutrition; or fatigue.
 Culture and Genetics
Culture and Genetics
Circumcision
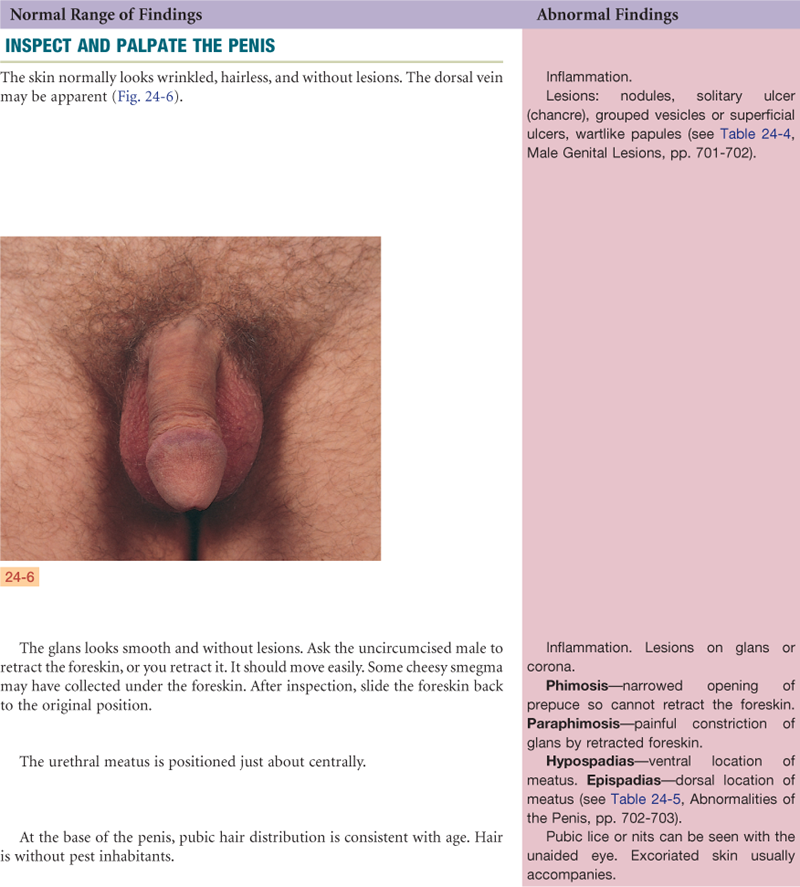
During pregnancy or the immediate neonatal period, parents will ask you about whether to circumcise the male infant. There are religious and cultural indications for circumcision; other indications include preventing phimosis and inflammation of the glans penis and foreskin, decreasing the incidence of cancer of the penis, and decreasing the incidence of urinary tract infections in infancy.
Circumcision reduces HIV acquisition in men by 53% to 60%, as shown in three randomized trials and numerous observational studies in sub-Saharan Africa,10,24 and it reduces HIV transmission to uninfected women sexual partners.27 Further, circumcision significantly reduced the incidence of herpes simplex virus type 2 (HSV-2) and the prevalence of human papillomavirus (HPV).22 Certain types of HPV cause cervical cancer. The mechanism may be that the presence of the foreskin increases susceptibility to small abrasions, allowing more contact time between pathogens and the mucosa of the partner.9 The risk for other STIs (Trichomonas vaginalis, bacterial vaginosis) is reduced in women with circumcised partners as well.21
In the United States, the public health implications of these findings are being debated. Routine neonatal circumcision rates have dropped from a high of 80% after World War II to about 65% of newborns in 1999, in part because the American Academy of Pediatrics does not endorse the procedure. Because of this, Medicaid does not cover routine neonatal circumcision costs in 16 states. This is disadvantageous especially to African-American and Hispanic male infants who are overrepresented in poorer groups whose only medical coverage is Medicaid.
Circumcision carries a very small but possible risk for complications. Most are minor and treatable: pain, bleeding, swelling, or inadequate skin removal. Serious complications are rare and include excess bleeding, wound infection, and urinary retention.28 Neonates certainly are capable of perceiving pain; therefore parents need to be apprised of pain-relief measures for the circumcision procedure. These include dorsal penile nerve block and a lidocaine-prilocaine cream (EMLA).29
Kidney Disease
Chronic kidney disease (CKD) is determined by blood tests, urinalysis, and imaging studies that show decreased kidney function or kidney damage lasting 3 months or longer. This can lead progressively and irreversibly to end-stage renal disease (ESRD), when the person survives only by kidney transplant or dialysis. CKD is a global health problem. It has two main causes, hypertension and diabetes, which comprise 70% or more of patients who progress to ESRD and are having dialysis.
In the United States, the majority of patients with CKD are white; however, racial and ethnic groups are disproportionately affected. When compared with whites, the per million prevalence rates are 47% higher for Hispanics, 230% higher for American Indian/Alaskan Natives, and 420% higher for African Americans.23 Reasons for these astounding differences are being actively researched. One factor may be long-term underutilization of antihypertensive medications, the renin-angiotensin-aldosterone system (RAAS) inhibitors, by African Americans.19 Socioeconomic factors also limit access to health care for lower income groups. In the United States, 22% of whites are uninsured or receiving public-funded health care, compared with 55% of Hispanics and 45% of African Americans.26
Subjective Data
1. Frequency, urgency, and nocturia
6. Penis—pain, lesion, discharge
7. Scrotum, self-care behaviors, lump




Objective Data
| Preparation | Equipment needed |
| Position the male standing with undershorts down and appropriate draping. The examiner should be sitting. Alternatively, the male may be supine for the first part of the examination and stand to check for a hernia. It is normal for a male to feel apprehensive about having his genitalia examined, especially by a female examiner. Younger adolescents usually have more anxiety than older adolescents. But any male may have difficulty dissociating a necessary, matter-of-fact step in the physical examination from the feeling this is an invasion of his privacy. His concerns are similar to those experienced by the female during the examination of the genitalia: modesty, fear of pain, cold hands, negative judgment, or memory of previously uncomfortable examinations. In addition, he may fear comparison with others or fear having an erection during the examination and that this would be misinterpreted by the examiner. This normal apprehension becomes manifested in different behaviors. Many act resigned and embarrassed and avoid eye contact. An occasional man will laugh and make jokes to cover embarrassment. Also, a man may refuse examination by a female and insist on a male examiner. | Gloves: wear gloves during every male genitalia examination Occasionally: glass slide for urethral specimen Materials for cytology Flashlight |
| Take time to consider these feelings, as well as to explore your own. It is normal for you to feel embarrassed and apprehensive too. You may worry about your age, lack of clinical experience, causing pain, or even that your movements might “cause” an erection. Some examiners feel guilty when this occurs. You need to accept these feelings and work through them so that you can examine the male in a professional way. Discuss these concerns with an experienced examiner. Your demeanor is important. Your unresolved discomfort magnifies any discomfort the man may have. | |
| Your demeanor should be confident and relaxed, unhurried yet businesslike. Do not discuss genitourinary history or sexual practices while you are performing the examination. This may be perceived as judgmental. Use a firm, deliberate touch, not a soft, stroking one. If an erection does occur, do not stop the examination or leave the room. This only focuses more attention on the erection and increases embarrassment. Reassure the male that this is only a normal physiologic response to touch, just as when the pupil constricts in response to bright light. Proceed with the rest of the examination. |


 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
Urinates four or five times/day, clear, straw-colored. No nocturia, dysuria, or hesitancy. No pain, lesions, or discharge from penis. Does not do testicular self-examination. No history of genitourinary disease. Sexually active in a monogamous relationship. Sexual life satisfactory to self and partner. Uses birth control via barrier method (partner uses diaphragm). No known STI contact.
Objective
No lesions, inflammation, or discharge from penis. Scrotum—testes descended, symmetric, no masses. No inguinal hernia.
Assessment
Genital structures normal
Focused Assessment: Clinical Case Study
R.C. is a 19-year-old student who 2 days PTA noted acute onset of painful urination, frequency, and urgency. Noted some thick penile discharge.
Subjective
States has no side pain, no abdominal pain, no fever, and no genital skin rash. R.C. is concerned he has an STI because of episode of unprotected intercourse with a new partner 6 days PTA. Has no known allergies.
Objective
Vital signs 37° C-72-16. No lesions or inflammation around penis or scrotum. Urethral meatus has mild edema with purulent urethral discharge. No pain on palpation of genitalia. Testes symmetric with no masses. No lymphadenopathy.
Assessment
Urethral discharge
Deficient knowledge about STI prevention R/T lack of information recall
Abnormal Findings for Advanced Practice
TABLE 24-7
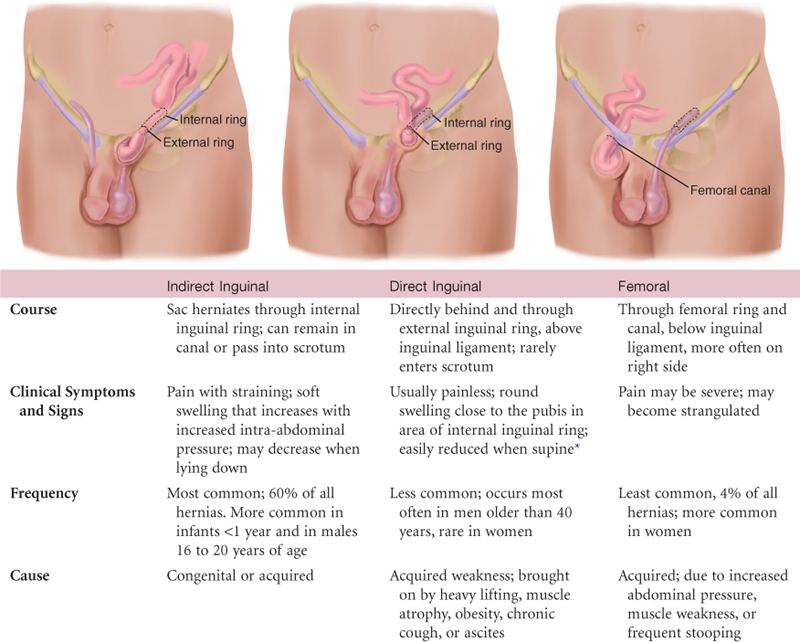
*Reducible—contents will return to abdominal cavity by lying down or gentle pressure. Incarcerated—herniated bowel cannot be returned to abdominal cavity. Strangulated—blood supply to hernia is shut off. Accompanied by nausea, vomiting, and tenderness.
Summary Checklist: Male Genitalia Examination
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
Bibliography
1. American Cancer Society. What are the risk factors for testicular cancer? Retrieved June 15, 2010, from www.cancer.org/docroot/cri/content/cri_2_4_2x_what_are_the_risk_factors_for_testicular_cancer_41.asp; 2009.
2. Baumann BM, Welsh BE, Rogers CJ, et al. Nurses using volumetric bladder ultrasound in the pediatric ED. American Journal of Nursing. 2008;108(4):73–76.
3. Bohnenkamp S, Yoder LH. The medical-surgical nurse’s guide to testicular cancer. Medsurg Nursing. 2009;18(2):116–123.
4. Bradway C, Rodgers J. Evaluation and management of genitourinary emergencies. Nurse Practitioner. 2009;34(5):37–44.
5. Brady MT. Newborn circumcision. Archives of Pediatrics & Adolescent Medicine. 2010;164(1):94–96.
6. Cochran SD, Mays VM. Physical health complaints among lesbians, gay men, and bisexual and homosexually experienced heterosexual individuals: results from the California quality of life survey. American Journal of Public Health. 2007;97(11):2048–2055.
7. Copstead LC, Banasik JL. Pathophysiology. 4th ed. St. Louis: Saunders; 2010.
7. Crestodina LR. Assessment and management of urinary incontinence in the elderly male. Nurse Practitioner. 2007;32(9):27–35.
8. Fitzgerald M, Walker SM. Infant pain management: a developmental neurobiological approach. Nature Clinical Practice Neurology. 2009;5(1):35–50.
9. Golden MR, Wasserheit JN. Prevention of viral sexually transmitted infections. New England Journal of Medicine. 2009;360(13):1349–1351.
10. Gray RH, Wawer MJ, Serwadda D, et al. The role of male circumcision in the prevention of human papillomavirus and HIV infection. Journal of Infectious Diseases. 2009;199(1):1–3.
11. Hayes-Lattin B, Nichols CR. Testicular cancer: a prototypic tumor of young adults. Seminars in Oncology. 2009;36(5):432–438.
12. Heidelbaugh JJ. Management of erectile dysfunction. American Family Physician. 2010;81(3):305–312.
13. Herman-Giddens ME, Wang L, Koch G. Secondary sexual characteristics in boys. Archives of Pediatrics & Adolescent Medicine. 2001;155(1):1022–1028.
14. Hornor G. Sexual behavior in children: normal or not? Journal of Pediatric Health Care. 2004;18(2):57–64.
15. Joffe A. Should we teach testicular self-exam? Contemporary Pediatrics. 2009;26(8):33–34.
16. Jones L, Felblinger D, Cooper L. Mycoplasma genitalium. Nurse Practitioner. 2009;34(8):50–52.
17. Leung-Chen P. Syphilis makes another comeback. American Journal of Nursing. 2008;108(2):28–30.
18. Marshall W, Tanner J. Variations in the pattern of pubertal changes in boys. Archives of Disease in Childhood. 1970;45(239):13.
19. Pearson MZ. Racial disparities in chronic kidney disease: current data and nursing roles. Nephrology Nursing Journal. 2008;35(5):485–489.
20. Ries LAG, Melbert D, Krapcho M, et al. SEER cancer statistics reviews, 1975-2005. Bethesda, MD: National Cancer Institute; 2008; Retrieved June 15, 2010, from http://seer.cancer.gov/csr/1975_2005/; 2008.
21. Sobngwi-Tambekou J, Talijaard D, Nieuwoudt M, et al. Male circumcision and Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis: observation after a randomized controlled trial for HIV prevention. Sexually Transmitted Infections. 2009;85(1):116–120.
22. Tobian AAR, Serwadda D, Quinn TC, et al. Male circumcision for the prevention of HSV-2 and HPV infections and syphilis. New England Journal of Medicine. 2009;360(13):1298–1309.
23. U.S. Renal Data System (USRDS). (2007). USRDS 2007 annual data report: atlas of end-stage renal disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Chronic Kidney Disease, Economic Costs, Clinical Indicators.
24. Viscidi RP, Shah KV. Adult male circumcision: will it reduce disease caused by human papillomavirus? Journal of Infectious Diseases. 2010;201(10):1447–1449.
25. Wallace MA. Assessment of sexual health in older adults. The American Journal of Nursing. 2008;108(7):52–61.
26. Ward MM. Socioeconomic status and the incidence of ESRD. American Journal of Kidney Diseases. 2008;51(4):563–572.
27. Wawer MJ, Makumbi F, Kigozi G, et al. Circumcision in HIV-infected men and its effect on HIV transmission to female partners in Rakai, Uganda: a randomized controlled trial. Lancet. 2009;374(1):229–237.
28. Weiss HA, Larke N, Halperin D, et al. Complications of circumcision in male neonates, infants and children: a systematic review. BMC Urology. 2010;10(1):2.
29. Yamada J, Stinson J, Lamba J, et al. A review of systematic reviews on pain interventions in hospitalized infants. Pain Research & Management. 2008;13(5):413–420.
*At times, phrase your questions so that all is right for the person to acknowledge a problem.
*Avoid the old direction, “turn your head and cough.” For one thing, a brief cough does not give the steady, increased intra-abdominal pressure you need. For another, the person is likely to cough right in your face.