Female Genitourinary System
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
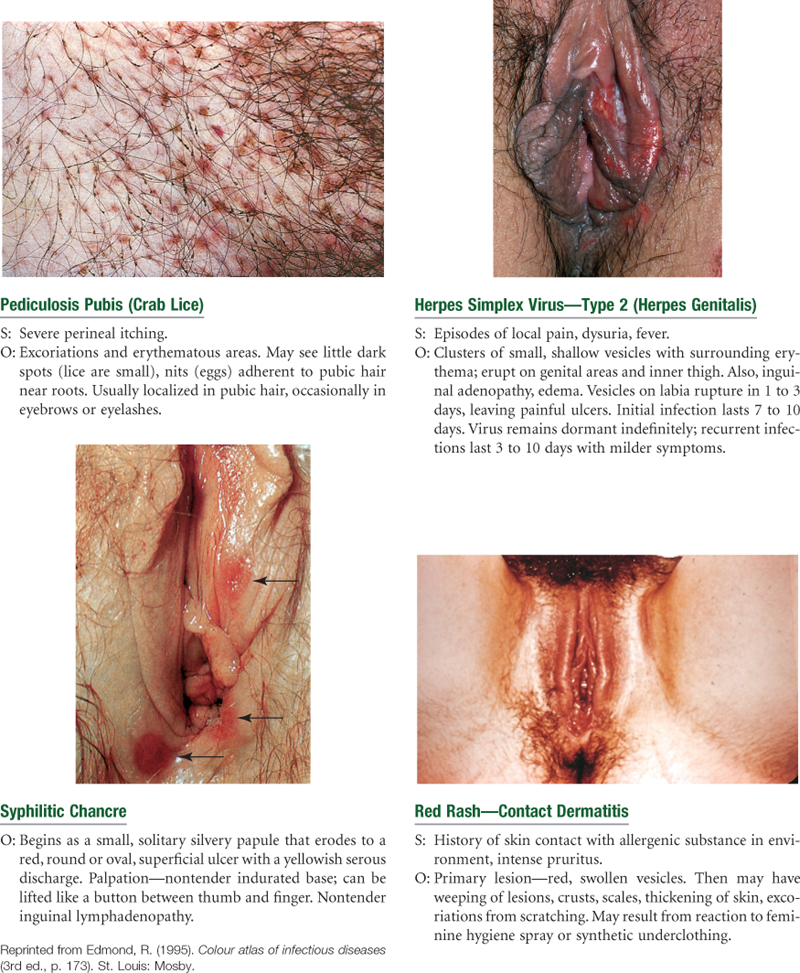
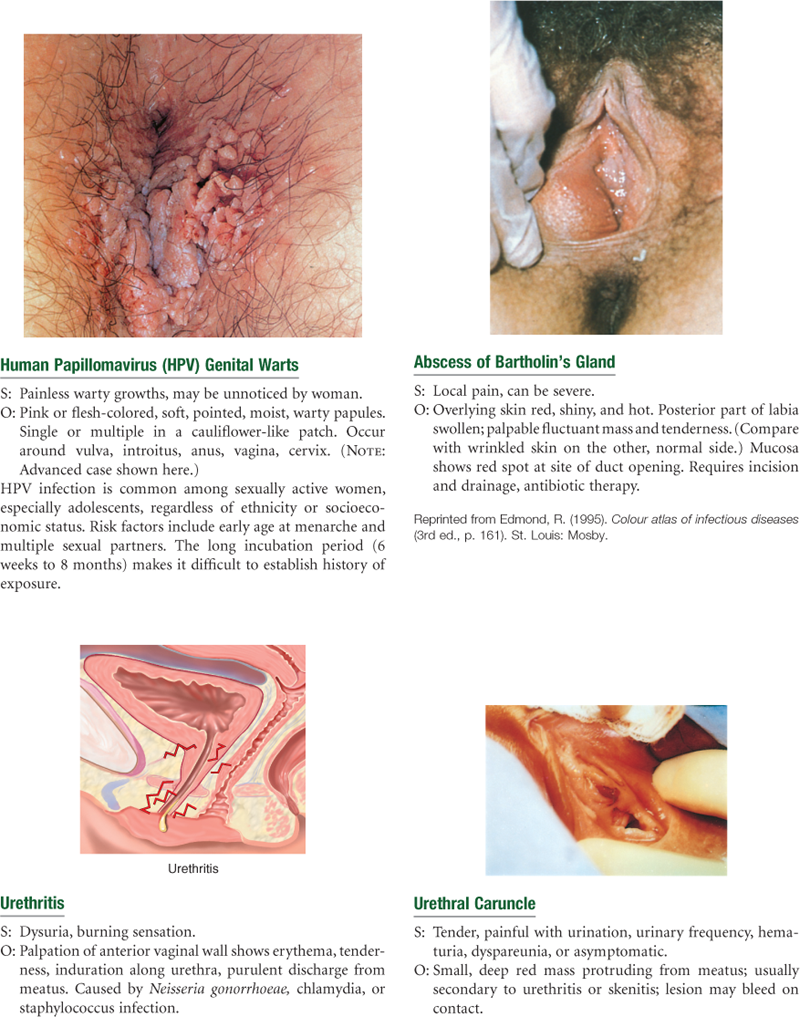
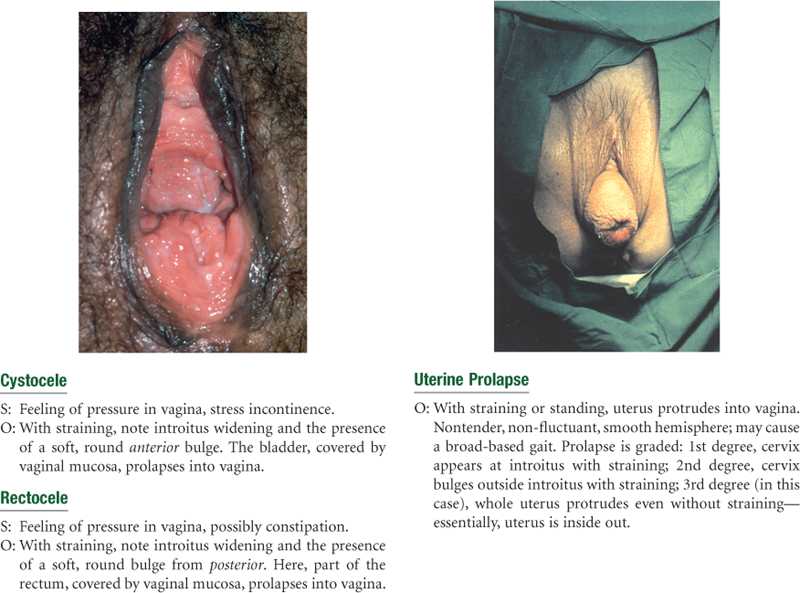
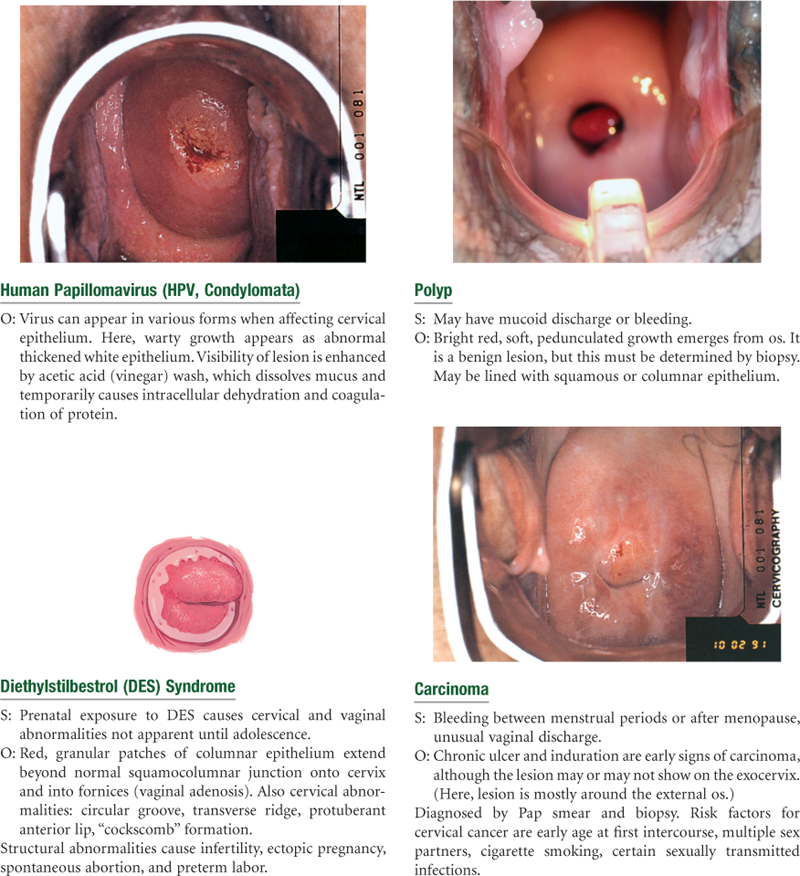
Sexually Transmitted Infections (STIs)
• Physical Examination Summary Checklists
Structure and Function
External Genitalia
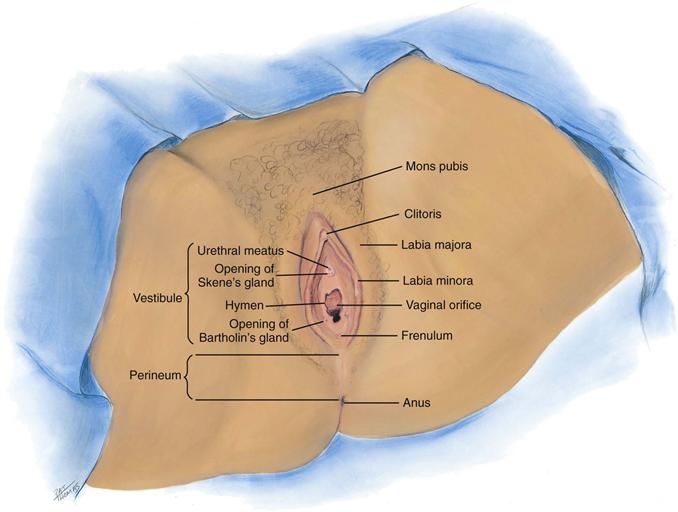
The external genitalia are called the vulva, or pudendum (Fig. 26-1). The mons pubis is a round, firm pad of adipose tissue covering the symphysis pubis. After puberty, it is covered with hair in the pattern of an inverted triangle. The labia majora are two rounded folds of adipose tissue extending from the mons pubis down and around to the perineum. After puberty, hair covers the outer surfaces of the labia, whereas the inner folds are smooth and moist and contain sebaceous follicles.
26-1
Inside the labia majora are two smaller, darker folds of skin, the labia minora. These are joined anteriorly at the clitoris where they form a hood, or prepuce. The labia minora are joined posteriorly by a transverse fold, the frenulum, or fourchette. The clitoris is a small, pea-shaped erectile body, homologous with the male penis and highly sensitive to tactile stimulation.
The labial structures encircle a boat-shaped space, or cleft, termed the vestibule. Within it are numerous openings. The urethral meatus appears as a dimple 2.5 cm posterior to the clitoris. Surrounding the urethral meatus are the tiny, multiple paraurethral (Skene’s) glands. Their ducts are not visible but open posterior to the urethra at the 5 and 7 o’clock positions.
The vaginal orifice is posterior to the urethral meatus. It appears either as a thin median slit or as a large opening with irregular edges, depending on the presentation of the membranous hymen. The hymen is a thin, circular or crescent-shaped fold that may cover part of the vaginal orifice or may be absent completely. On either side and posterior to the vaginal orifice are two vestibular (Bartholin’s) glands, which secrete a clear lubricating mucus during intercourse. Their ducts are not visible but open in the groove between the labia minora and the hymen.
Internal Genitalia
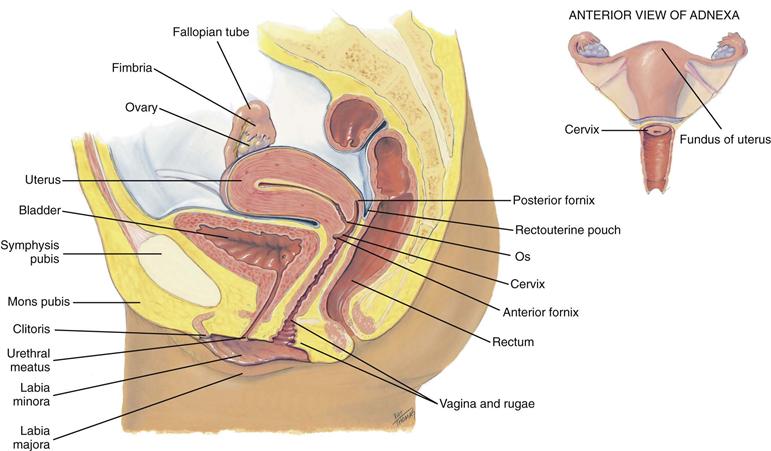
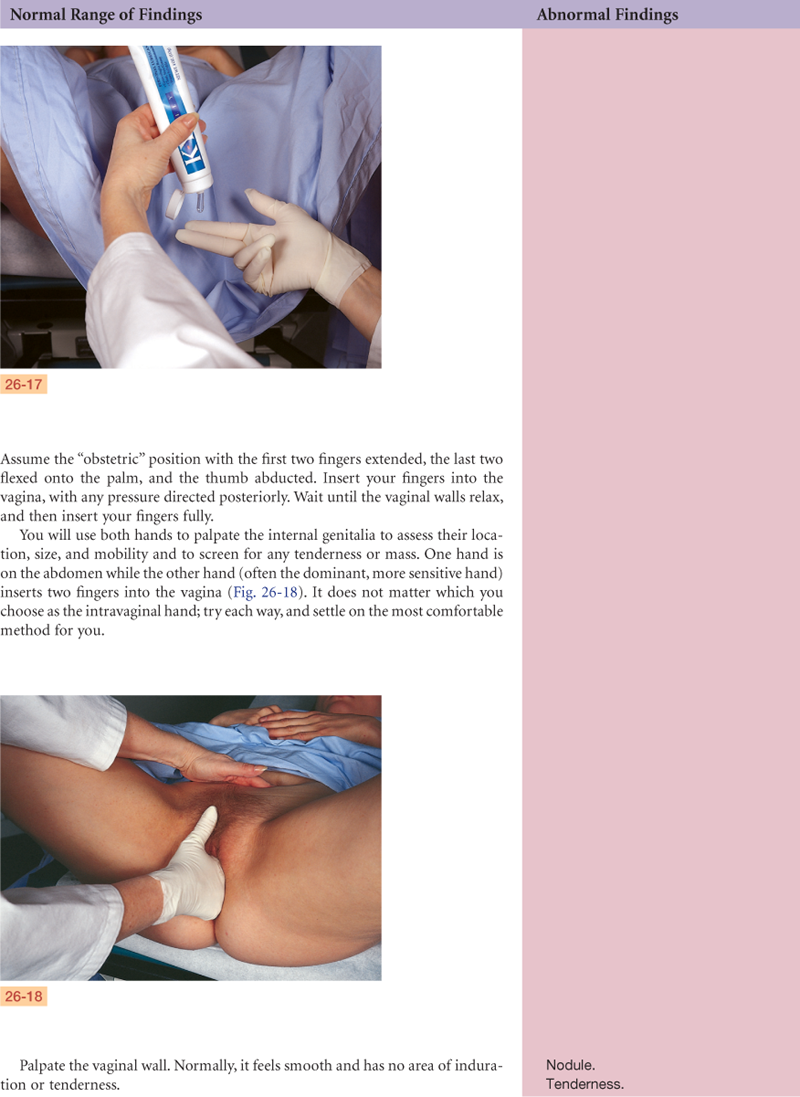
The internal genitalia include the vagina, a flattened, tubular canal extending from the orifice up and backward into the pelvis (Fig. 26-2). It is 9 cm long and sits between the rectum posteriorly and the bladder and urethra anteriorly. Its walls are in thick transverse folds, or rugae, enabling the vagina to dilate widely during childbirth.
26-2
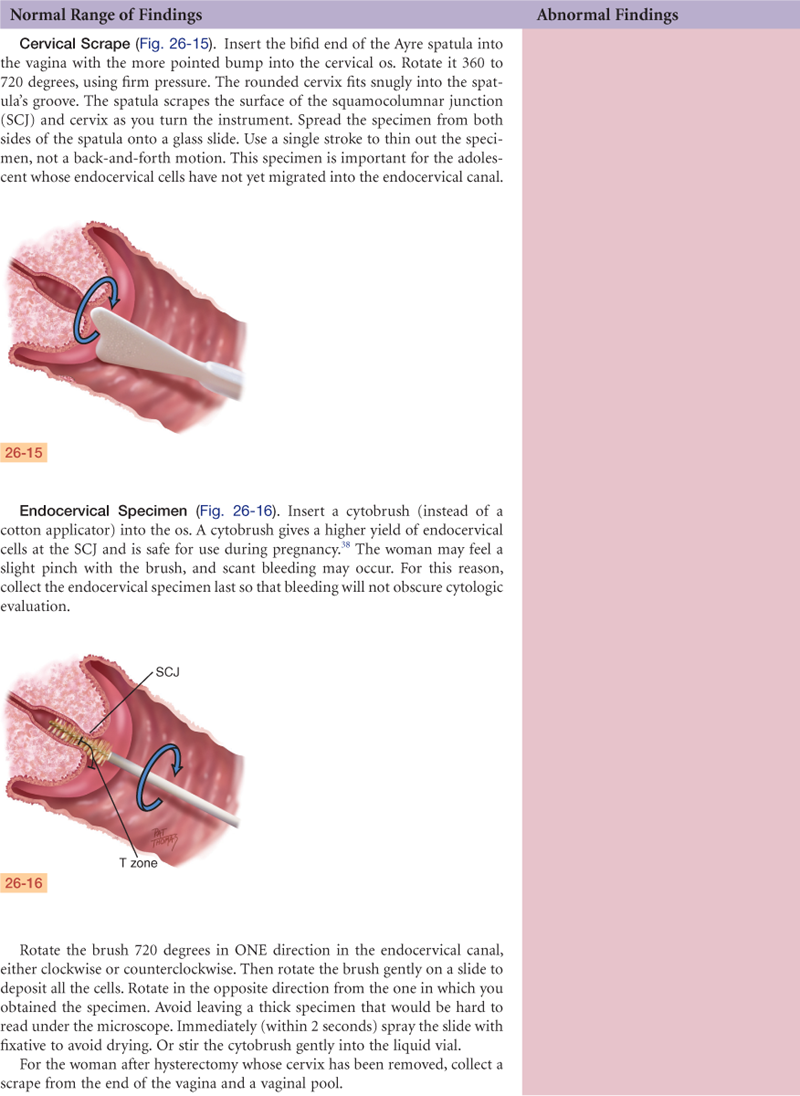
At the end of the canal, the uterine cervix projects into the vagina. In the nulliparous female, the cervix appears as a smooth doughnut-shaped area with a small circular hole, or os. After childbirth, the os is slightly enlarged and irregular. The cervical epithelium is of two distinct types. The vagina and cervix are covered with smooth, pink, stratified squamous epithelium. Inside the os, the endocervical canal is lined with columnar epithelium that looks red and rough. The point where these two tissues meet is the squamocolumnar junction and is not visible.
A continuous recess is present around the cervix, termed the anterior fornix in front and the posterior fornix in back. Behind the posterior fornix, another deep recess is formed by the peritoneum. It dips down between the rectum and cervix to form the rectouterine pouch, or cul-de-sac of Douglas.
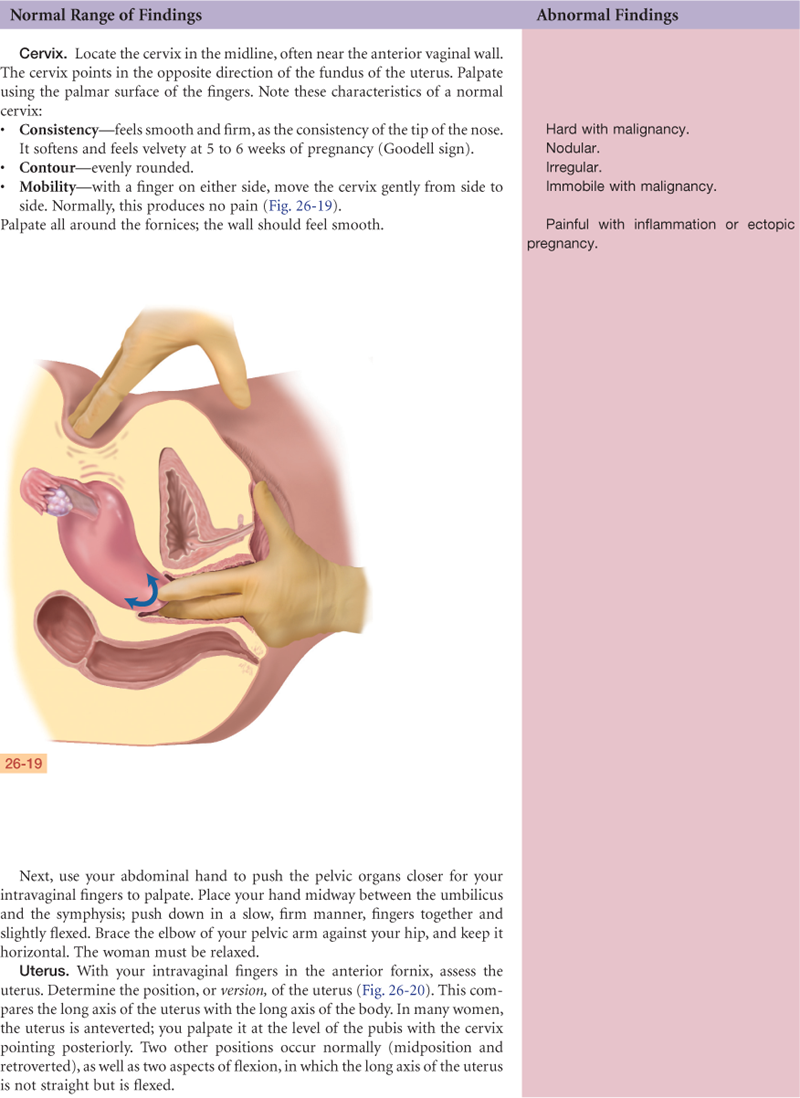
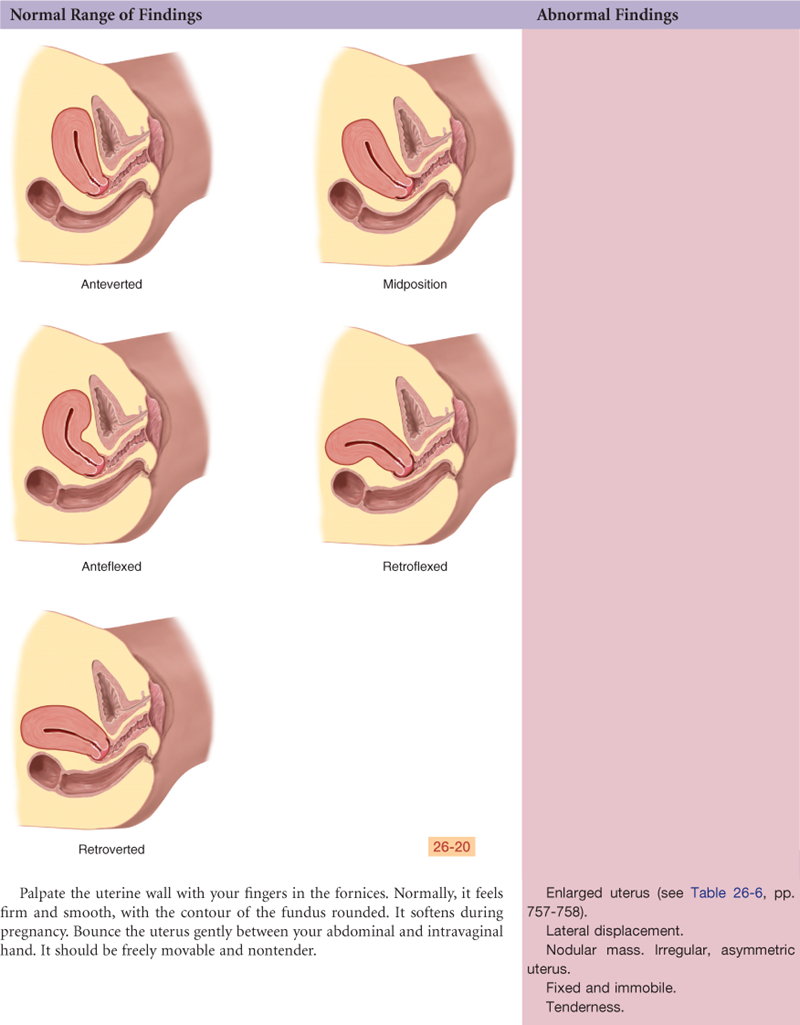
The uterus is a pear-shaped, thick-walled, muscular organ. It is flattened anteroposteriorly, measuring 5.5 to 8 cm long by 3.5 to 4 cm wide and 2 to 2.5 cm thick. It is freely movable, not fixed, and usually tilts forward and superior to the bladder (a position labeled as anteverted and anteflexed, see p. 745).
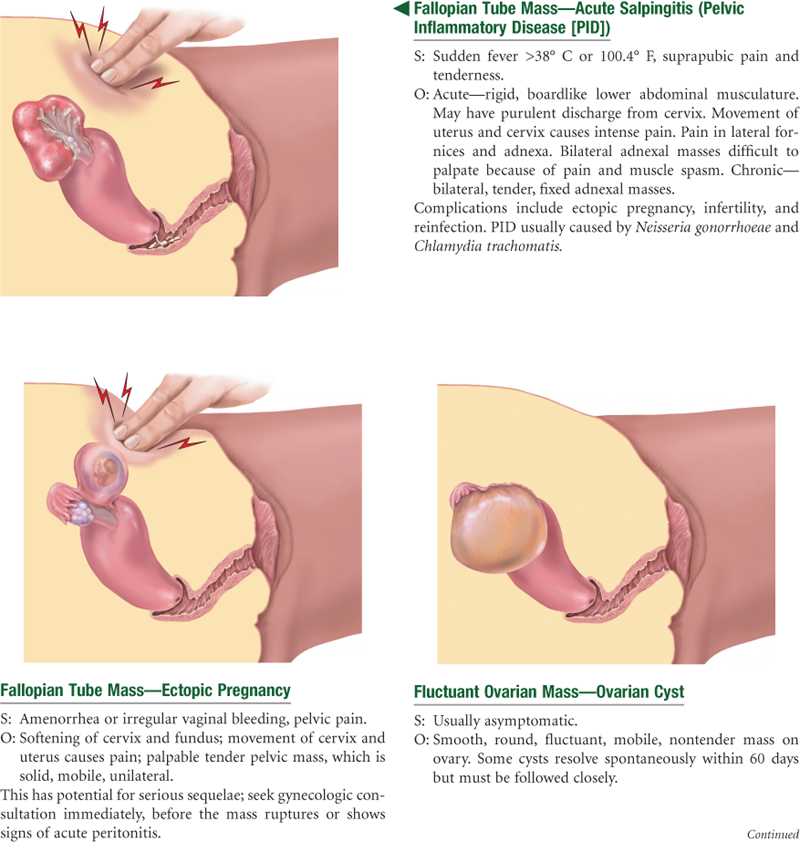
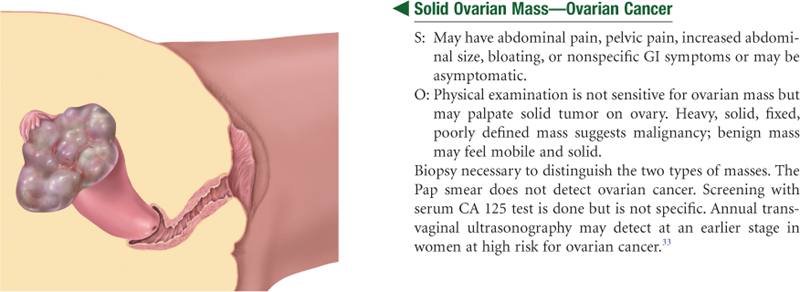
The fallopian tubes are two pliable, trumpet-shaped tubes, 10 cm in length, extending from the uterine fundus laterally to the brim of the pelvis. There they curve posteriorly, their fimbriated ends located near the ovaries. The two ovaries are located one on each side of the uterus at the level of the anterior superior iliac spine. Each is oval-shaped, 3 cm long by 2 cm wide by 1 cm thick, and serves to develop ova (eggs) and the female hormones.
 Developmental Competence
Developmental Competence
Infants and Adolescents
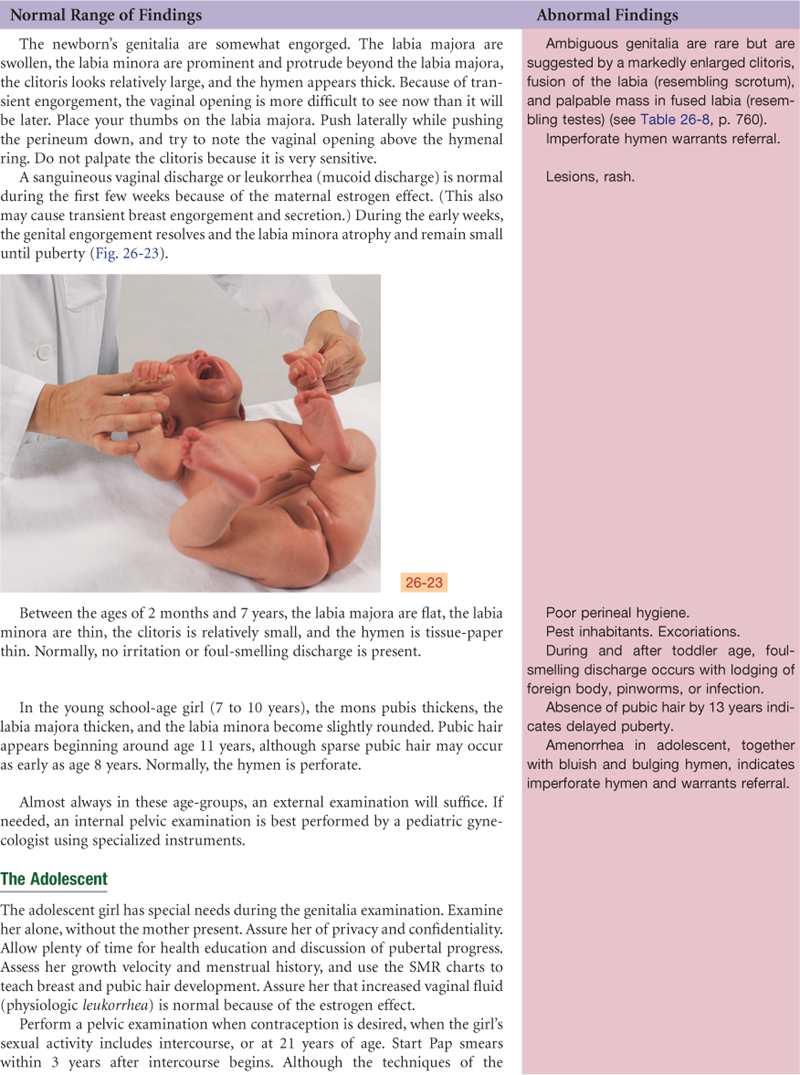
At birth, the external genitalia are engorged because of the presence of maternal estrogen. The structures recede in a few weeks, remaining small until puberty. The ovaries are located in the abdomen during childhood. The uterus is small with a straight axis and no anteflexion.
At puberty, estrogens stimulate the growth of cells in the reproductive tract and the development of secondary sex characteristics. The first signs of puberty are breast and pubic hair development, beginning between the ages of 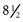 and 13 years. These signs are usually concurrent, but it is not abnormal if they do not develop together. They take about 3 years to complete.
and 13 years. These signs are usually concurrent, but it is not abnormal if they do not develop together. They take about 3 years to complete.
Menarche occurs during the latter half of this sequence, just after the peak of growth velocity. Irregularity of the menstrual cycle is common during adolescence because of the girl’s occasional failure to ovulate. With menarche, the uterine body flexes on the cervix. The ovaries now are in the pelvic cavity.
Tanner’s table on the five stages of pubic hair development (sexual maturity rating [SMR]) is helpful in teaching girls the expected sequence of sexual development (Table 26-1). These data may not generalize to all racial groups; mature Asian women normally have fine, sparse pubic hair.
TABLE 26-1
Sexual Maturity Ratings (SMR) in Girls
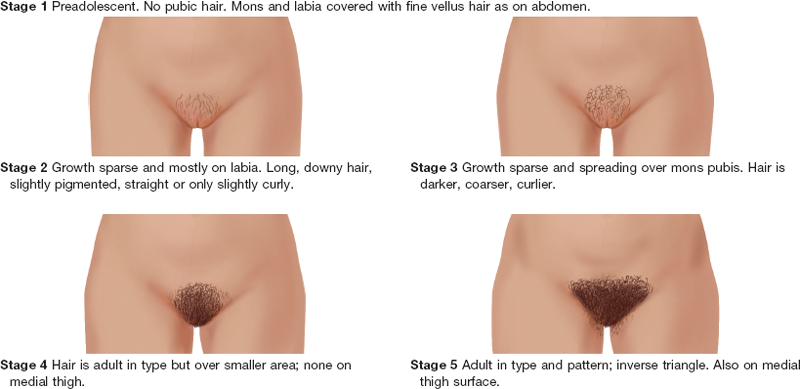
Adapted from Tanner, J.M. (1962). Growth at adolescence. Oxford, England: Blackwell Scientific.
Data from NHANES III found that contemporary U.S. girls’ development is consistent with Tanner’s findings.40 However, African-American and Mexican-American girls had pubic hair and achieved menarche at younger ages than white girls. The mean age at onset of pubic hair and menarche was 9.5 and 12.1 years for African-American girls; 10.3 and 12.2 years for Mexican-American girls; and 10.5 and 12.7 years for white girls. Thus African-American girls on average enter puberty first, followed by Mexican-American and then white girls.34,40
The ongoing U.S. epidemic of childhood obesity shows that menarche is significantly more likely to occur in preteen girls with an elevated body mass index (BMI).34 The median age of achieving menarche was 5.4 months earlier in obese preteen girls than in girls with normal BMI. The association of adiposity with menarche was independent of race or ethnicity. In conclusion, BMI should be a major consideration when a clinician evaluates pubertal development.34
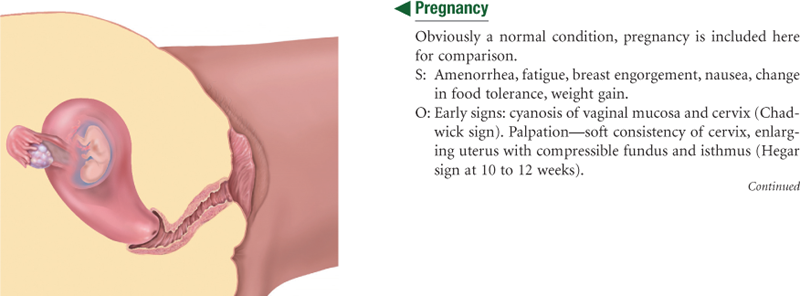
The Pregnant Woman
A complete discussion of the pregnant woman follows in Chapter 29. In summary, shortly after the first missed menstrual period, the genitalia show signs of the growing fetus. The cervix softens (Goodell sign) at 4 to 6 weeks, and the vaginal mucosa and cervix look cyanotic (Chadwick sign) at 8 to 12 weeks. These changes occur because of increased vascularity and edema of the cervix and hypertrophy and hyperplasia of the cervical glands. The isthmus of the uterus softens (Hegar sign) at 6 to 8 weeks.
The greatest change is in the uterus itself. It increases in capacity by 500 to 1000 times its nonpregnant state, at first because of hormone stimulation and then because of the increasing size of its contents.12 The nonpregnant uterus has a flattened pear shape. Its early growth encroaches on the space occupied by the bladder, producing the symptom of urinary frequency. By 10 to 12 weeks’ gestation, the uterus becomes globular in shape and is too large to stay in the pelvis. At 20 to 24 weeks, the uterus has an oval shape. It rises almost to the liver, displacing the intestines superiorly and laterally.
A clot of thick, tenacious mucus forms in the spaces of the cervical canal (the mucus plug), which protects the fetus from infection. The mucus plug dislodges when labor begins at the end of term, producing a sign of labor called “bloody show.” Cervical and vaginal secretions increase during pregnancy and are thick, white, and more acidic. The increased acidity occurs because of the action of Lactobacillus acidophilus, which changes glycogen into lactic acid. The acidic pH keeps pathogenic bacteria from multiplying in the vagina, but the increase in glycogen increases the risk for candidiasis (commonly called a yeast infection) during pregnancy.
The Aging Woman
In contrast to the slowly declining hormones in the aging male, the female’s hormonal milieu decreases rapidly. Menopause is cessation of the menses. Usually this occurs around 48 to 51 years, although a wide variation of ages from 35 to 60 years exists. The stage of menopause includes the preceding 1 to 2 years of decline in ovarian function, shown by irregular menses that gradually become farther apart and produce a lighter flow. Ovaries stop producing progesterone and estrogen. Because cells in the reproductive tract are estrogen dependent, decreased estrogen levels during menopause bring dramatic physical changes.
The uterus shrinks in size because of decreased myometrium. The ovaries atrophy to 1 to 2 cm and are not palpable after menopause. Ovulation still may occur sporadically after menopause. The sacral ligaments relax and the pelvic musculature weakens, so the uterus droops. Sometimes it may protrude, or prolapse, into the vagina. The cervix shrinks and looks paler with a thick, glistening epithelium.
The vagina becomes shorter, narrower, and less elastic because of increased connective tissue. Without sexual activity, the vagina atrophies to one-half its former length and width. The vaginal epithelium atrophies, becoming thinner, drier, and itchy. This results in a fragile mucosal surface that is at risk for bleeding and vaginitis. Decreased vaginal secretions leave the vagina dry and at risk for irritation and pain with intercourse (dyspareunia). The vaginal pH becomes more alkaline, and glycogen content decreases from the decreased estrogen. These factors also increase the risk for vaginitis because they create a suitable medium for pathogens.
Externally, the mons pubis looks smaller because the fat pad atrophies. The labia and clitoris gradually decrease in size. Pubic hair becomes thin and sparse.
Declining estrogen levels produce some physiologic changes in the female sexual response cycle: reduced amount of vaginal secretion and lubrication during excitement; shorter duration of orgasm; and rapid resolution. However, these changes do not affect sexual pleasure and function. As with the male, the older female is capable of sexual expression and function given reasonably good health and an interested partner. Aging women greatly outnumber their male counterparts, and aging women are more likely to be single, whereas males their same age are more likely to be married.
 Culture and genetics
Culture and genetics
The increased use of the Papanicolaou (Pap) test in the United States has resulted in a 74% decline in the cervical cancer death rate between 1955 and 1992.2 Today, however, cervical cancer occurs most often in Hispanic women; their incidence is over twice that of white women. African-American women have a 50% higher incidence rate than white women. Relative to white women, Hispanic women are less likely to die of cervical cancer despite their lower socioeconomic status (SES), known as the “Hispanic paradox.”11 Reasons are unclear but include differences in comorbid conditions, social support, cultural influences, and religion/faith.
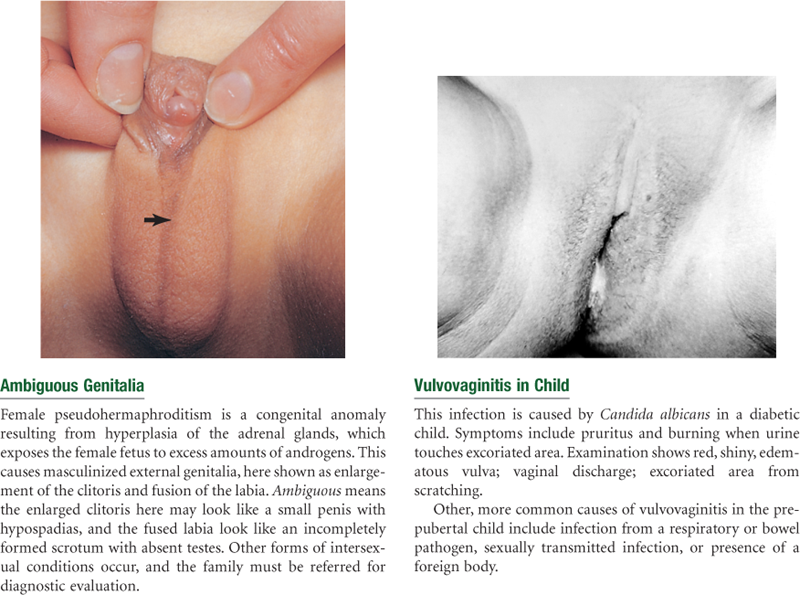
Female circumcision, known as infibulation or female genital mutilation, is an invasive surgical procedure that is performed on girls before puberty. It is practiced within Aboriginal, Christian, and Muslim families who have emigrated to the United States from western and southern Asia, the Middle East, and large areas of Africa. It is a social custom, not a religious practice. This procedure involves removal, partial or total, of the clitoris and is believed to inhibit sexual pleasure. There are about 130 to 140 million women alive today who have had this procedure.22 The procedure is outlawed in the United States.
Objective Data
| Preparation | Equipment needed |
| Assemble the equipment before helping the woman into position. Arrange within easy reach. Familiarize yourself with the vaginal speculum before the examination. Practice opening and closing the blades, locking them into position, and releasing them. Try both metal and plastic types. Note that the plastic speculum locks and unlocks with a resounding click that can be alarming to the uninformed woman. | Gloves Gooseneck lamp with a strong light Vaginal speculum of appropriate size (Fig. 26-3): Graves speculum—useful for most adult women, available in varying lengths and widths |
| Pederson speculum—narrow blades, useful for young or postmenopausal women with narrowed introitus Large cotton-tipped applicators (rectal swabs) Materials for cytologic study: Glass slide with frosted end Sterile cytobrush or cotton-tipped applicator Liquid-based cytology vial Ayre spatula Spray fixative Specimen container for gonorrhea culture (GC)/chlamydia Small bottle of normal saline solution, potassium hydroxide (KOH), and acetic acid (white vinegar) Lubricant |

26-3
Position
Initially, the woman should be sitting up. An equal-status position is important to establish trust and rapport before the vaginal examination.
For the examination, the woman should be placed in the lithotomy position, with the examiner sitting on a stool. Help the woman into lithotomy position, with the body supine, feet in stirrups and knees apart, and buttocks at edge of examining table (Fig. 26-4). Ask the woman to lift her hips as you guide them to the edge of the table. Some women prefer to leave their shoes or socks on. Or, you can place an examination glove over each of the stirrups to warm the stirrups and keep her feet from slipping.
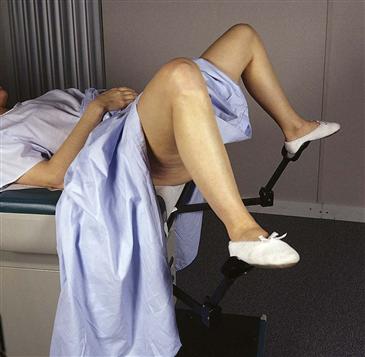
26-4
Place her arms at her sides or across the chest, not over the head, because this position only tightens the abdominal muscles. The traditional mode is to drape the woman fully, covering the stomach and legs, exposing only the vulva to your view. Be sure to push down the drape between the woman’s legs and elevate her head so that you can see her face.
The lithotomy position leaves many women feeling helpless and vulnerable. Indeed, many women tolerate the pelvic examination because they consider it basic for health care, yet they find it embarrassing and uncomfortable. Previous examinations may have been painful or the previous examiner’s attitude hurried and patronizing.
The examination need not be this way. You can help the woman relax, decrease her anxiety, and retain a sense of control by using these measures:
• Have her empty the bladder before the examination.
• Position the examination table so that her perineum is not exposed to an inadvertent open door.
• Ask if she would like a friend, family member, or chaperone present. Position this person by the woman’s head to maintain privacy.
• Elevate her head and shoulders to a semisitting position to maintain eye contact.
• Place the stirrups so that the legs are not abducted too far.
• Explain each step in the examination before you do it.
• Assure the woman she can stop the examination at any point should she feel any discomfort.
• Use a gentle, firm touch and gradual movements.
• Communicate throughout the examination. Maintain a dialogue to share information.
• Use the techniques of the educational or mirror pelvic examination (Fig. 26-5). This is a routine examination with some modifications in attitude, position, and communication. First, the woman is considered an active participant, one who is interested in learning and in sharing decisions about her own health care. The woman props herself up on one elbow, or the head of the table is raised. Her other hand holds a mirror between her legs, above the examiner’s hands. The woman can see all that the examiner is doing and has a full view of her genitalia.
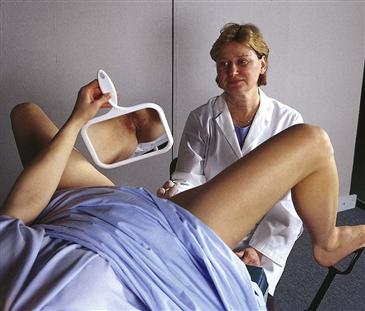
26-5
The mirror works well for teaching normal anatomy and its relationship to sexual behavior. Even women who are in a sexual relationship or who have had children may be surprisingly uninformed about their own anatomy. You will find the woman’s enthusiasm on seeing her own cervix is rewarding too.
The mirror pelvic examination also works well when abnormalities arise, because the woman can see the rationale for treatment and can monitor progress at the next appointment. She is more willing to comply with treatment when she shares in the decision.

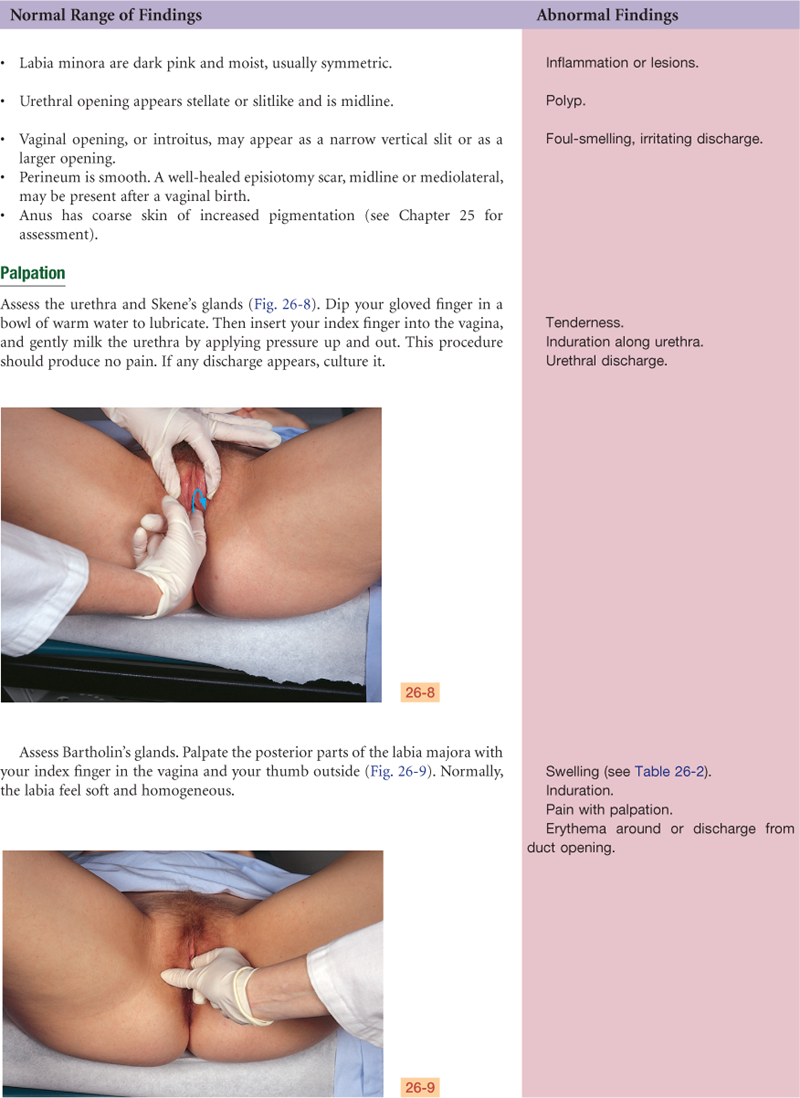

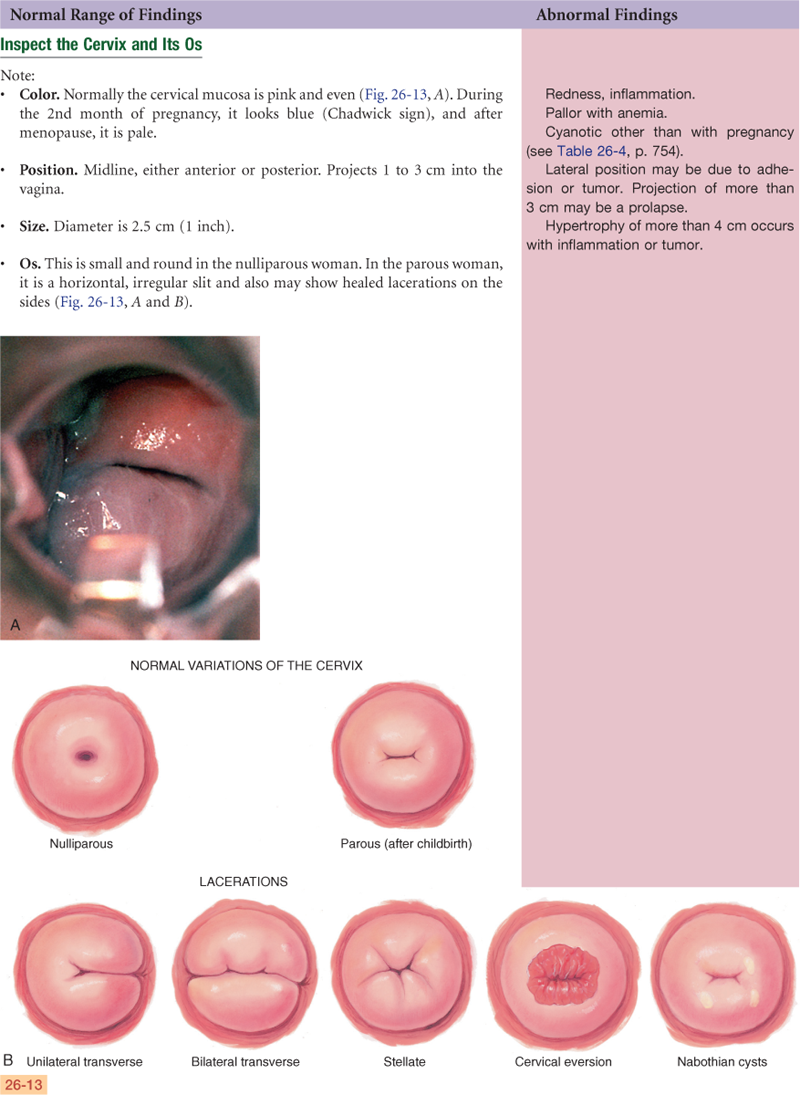




 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
Menarche age 12 years, cycle usually q 28 days, duration 5 days, flow moderate, no dysmenorrhea, LMP April 3. Grav 0/Para 0/Ab 0. Gyne checkups yearly. Last Pap test 1 year PTA, negative.
No urinary problems, no irritating or foul-smelling vaginal discharge, no sores or lesions, no history pelvic surgery. Satisfied with sexual relationship with husband, uses vaginal diaphragm for birth control, no plans for pregnancy at this time. Not aware of any STIcontact to herself or husband.
Objective
External genitalia: No swelling, lesions, or discharge. No urethral swelling or discharge.
Internal: Vaginal walls have no bulging or lesions, cervix pink with no lesions, scant clear mucoid discharge.
Bimanual: No pain on moving cervix, uterus anteflexed and anteverted, no enlargement or irregularity.
Adnexa: Ovaries not enlarged.
Rectal: No hemorrhoids, fissures, or lesions; no masses or tenderness; stool brown with guaiac test negative.
Assessment
Genital structures intact and appear healthy
Focused Assessment: Clinical Case Study 1
J.K., 27-year-old white married newspaper reporter, Grav 0/Para 0/Ab 0. Presents at clinic with “urinary burning, vaginal itching, and discharge × 4 days.”
Subjective
3 weeks PTA—treated at clinic for bronchitis with erythromycin. Improved within 5 days.
4 to 5 days PTA—noted burning on urination; intense vaginal itching; thick, white, “smelly” discharge. Warm water douche—no relief.
No previous history vaginal infection, urinary tract infection, or pelvic surgery. Monogamous sexual relationship, has used low-estrogen birth control pills for 3 years with no side effects.
Objective
Vulva and vagina erythematous and edematous. Thick, white, curdlike discharge clinging to vaginal walls. Cervix pink, no lesions.
Bimanual examination: No pain on palpating cervix, uterus not enlarged, ovaries not enlarged.
Specimens: Pap smear, GC/chlamydia to lab. KOH prep shows mycelia and spores of Candida albicans.
Assessment
Candida vaginitis
Pain R/T infectious process
Focused Assessment: Clinical Case Study 2
Brenda, 17-year-old white high school student, comes to clinic for pelvic examination.
Subjective
Menarche 12 years, cycle q 30 days, duration 6 days, mild cramps relieved by acetaminophen. LMP March 10. No dysuria, vaginal discharge, vaginal itching. Relationship involving intercourse with one boyfriend for 8 months PTA. For birth control, boyfriend uses condoms “sometimes.” Wants to start birth control pills. Never had pelvic examination. Never had teaching about breast self-examination or STIsexcept AIDS. Smokes cigarettes,  PPD, started age 11 years.
PPD, started age 11 years.
Objective
Breasts: Symmetric, no lesions or discharge, palpation reveals no mass or tenderness.
External genitalia: No redness, lesions, or discharge.
Internal genitalia: Vaginal walls and cervix pink with no lesions or discharge. Specimens obtained. Acetic acid wash shows no acetowhitening.
Bimanual: No tenderness to palpation, uterus anteverted with no enlargement, ovaries not enlarged.
Rectum: No masses, fissure, or tenderness. Stool brown and guaiac test negative.
Specimens: GC, chlamydia, Pap smear to lab.
Assessment
Breast and pelvic structures appear healthy
Deficient knowledge regarding: breast self-examination; birth control measures; STI prevention; cigarette smoking R/T lack of exposure
Abnormal Findings for Advanced Practice
Summary Checklist: Female Genitalia Examination
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
Bibliography
1. Ackerson K. Personal influences that affect motivation in Pap smear testing among African American women. Journal of Obstetric, Gynecologic, and Neonatal Nursing. 2010;39(2):136–146.
2. American Cancer Society. Cancer facts & figures 2010. Atlanta: Author; 2010.
3. Amies AM, Miller L, Lee SK. The effect of vaginal speculum lubrication on the rate of unsatisfactory cervical cytology diagnosis. Obstetrics and Gynecology. 2002;100(5):889–892.
4. Arbyn M, Bergeron C, Klinkhamer P, et al. Liquid compared with conventional cervical cytology. Obstetrics and Gynecology. 2008;111(1):167–177.
5. Banikarim C, Chacko M. Pelvic inflammatory disease in adolescents. Seminars in Pediatric Infectious Diseases. 2005;16:175–180.
6. Bartoszek MP. Recognizing polycystic ovary syndrome in the primary care setting. Nurse Practitioner. 2009;34(7):23–29.
7. Beerman H, van Dorst EBL, Kuenen-Boumeester V, et al. Superior performance of liquid-based versus conventional cytology in a population-based cervical cancer screening program. Gynecologic Oncology. 2009;112(3):572–576.
8. Berecki-Gisolf J, Begum N, Dobson AJ. Symptoms reported by women in midlife: menopausal transition or aging? Menopause. 2009;16(5):1021–1029.
9. Bruce ML, Baril C. Save the date: screening tips and new vaccines for female HPV. Nurse Practitioner. 2008;33(9):29–34.
10. Burns N, Briggs P, Gaudet CA. Chlamydia screening in teenage girls. Nurse Practitioner. 2007;32(6):41–43.
11. Coker AL, DeSimone CP, Eggleston KS, et al. Ethnic disparities in cervical cancer survival among Texas women. Journal of Women’s Health. 2009;18(10):1577–1582.
12. Cunningham FG, Leveno KJ, Bloom SL, et al. Williams’ obstetrics. 23rd ed New York: McGraw-Hill; 2010.
13. Daley A. Exercise and premenstrual symptomatology: a comprehensive review. Journal of Women’s Health. 2009;18(6):895–899.
14. Daley AM, Cromwell PF. How to perform a pelvic exam for the sexually active adolescent. Nurse Practitoner. 2002;27:28–45.
15. Dowling-Castronovo A, Specht JK. Assessment of transient urinary incontinence in older adults. American Journal of Nursing. 2009;109(2):62–72.
16. Espindola D, Kennedy KA, Fischer EG. Management of abnormal uterine bleeding and the pathology of endometrial hyperplasia. Obstetrics and Gynecology Clinics of North America. 2007;34(4):717–737.
17. Harer WB, Valenzuela G, Lebo D. Lubrication of the vaginal introitus and speculum does not affect Papanicolaou smears. Obstetrics and Gynecology. 2002;100(5):887–888.
18. Jones S. A step-by-step approach to HIV/AIDS. Nurse Practitioner. 2006;31:26–41.
19. Kelsey B. Contraceptive considerations for obese women. The Nurse Practitioner. 2010;35(3):25–32.
20. Kimberlin DW, Rouse DJ. Genital herpes. New England Journal of Medicine. 2004;350:1970–1977.
21. Kirkland LG. New developments in the management of STDs. Nurse Practitioner. 2006;31(12):12–23.
22. Kontoyannis M, Katsetos C. Female genital mutilation. Health Science Journal. 2010;4(1):31–36.
23. Kulp JL, Taylor HS. New theories on the causes and treatment of endometriosis. Contemporary Ob/Gyn. 2009;54(4):34–41.
24. Likes WM. Vulvar cancer in the wake of increasing incidence. Nurse Practitioner. 2009;34(2):45–50.
25. Lockwood-Rayermann S, Donovan HS, Rambo D, et al. Women’s awareness of ovarian cancer risks and symptoms. American Journal of Nursing. 2009;109(9):36–46.
26. Mao AJ, Anastasi JK. Diagnosis and management of endometriosis: the role of the advanced practice nurse in primary care. Journal of the American Academy of Nurse Practitioners. 2010;22(2):109–116.
27. Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Archives of Disease in Childhood. 1969;44:291–303.
28. McCloskey C. Updated office testing skills for vaginal infections. Nurse Practitioner. 2010;35(2):46–52.
29. Miller MM, Wilson JM, Waldrop J. Current acceptance of the HPV vaccine. Nurse Practitioner. 2008;33(4):18–23.
30. Nelson W, Moser RP, Gaffey A, et al. Adherence to cervical cancer screening guidelines for U.S women aged 25-64: data from the 2005 Health Information National Trends Survey (HINTS). Journal of Women’s Health. 2009;18(11):1759–1768.
31. Noone J. Strategies for contraceptive success. Nurse Practitioner. 2007;32(6):29–36.
32. Norton P, Brubaker L. Urinary incontinence in women. Lancet. 2006;367(9504):57–67.
33. Roett MA, Evans P. Ovarian cancer: an overview. American Family Physician. 2009;80(6):609–616.
34. Rosenfield RL, Lipton RB, Drum ML. Thelarche, pubarche, and menarche attainment in children with normal and elevated body mass index. Pediatrics. 2009;123(1):84–88.
35. Scarinci IC, Garcés-Palacio IC, Partridge EE. An examination of acceptability of HPV vaccination among African American women and Latina immigrants. Journal of Women’s Health. 2007;16(8):1224–1233.
36. Siebers AG, Klinkhamer PJJM, Grefte JMM, et al. Comparison of liquid-based cytology with conventional cytology for detection of cervical cancer precursors: a randomized controlled trial. Journal of the American Medical Association. 2009;302(16):1757–1764.
37. Smith RA, Cokkinides V, Brawley OW. Cancer screening in the United States, 2008: a review of current American Cancer Society guidelines. CA: A Cancer Journal for Clinicians. 2008;58:161–179.
38. Stillson T, Knight AL, Elswick RK. The effectiveness and safety of two cervical cytologic techniques during pregnancy. Journal of Family Practice. 1997;45(2):159–163.
39. Warman J. Cervical cancer screening in young women: saving lives with prevention and detection. Oncology Nursing Forum. 2010;37(1):33–38.
40. Wu T, Mendola P, Buck GM. Ethnic differences in the presence of secondary sex characteristics and menarche among U.S girls: the NHANES III, 1988-1994. Pediatrics. 2002;110:752–757.