Oncogenes and Signal Transduction
Signaling: An Overview
Oncogenes
Signal Transduction by Protein Tyrosine Kinase Receptors
RTKs and Cancer
Signaling Pathways of Tyrosine Kinase Receptors
RAS
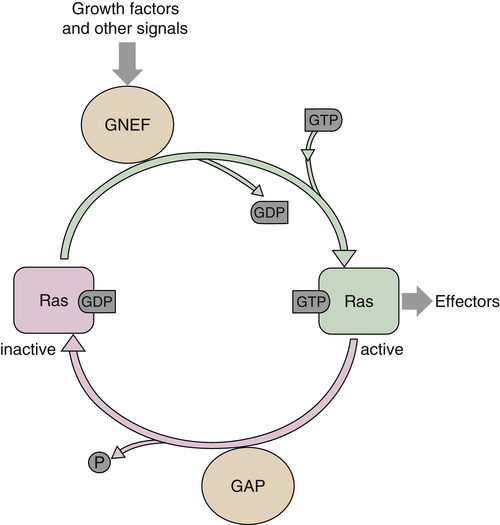
Ras Functions
Ras and Cancer
Signaling Downstream of RAS
Ras>Raf>Map Kinase Cascade
Functions of the MAP Kinase Pathway
Raf/Mek/MapK and Cancer
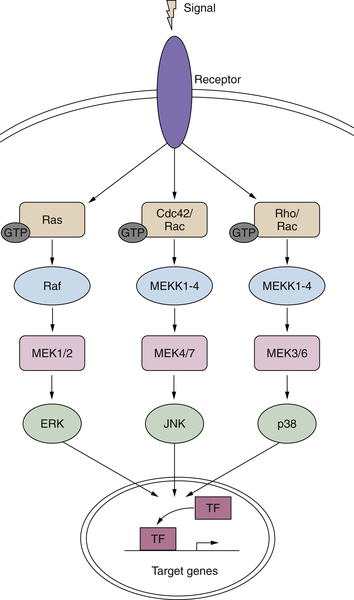
Other MAP Kinases
C-Myc
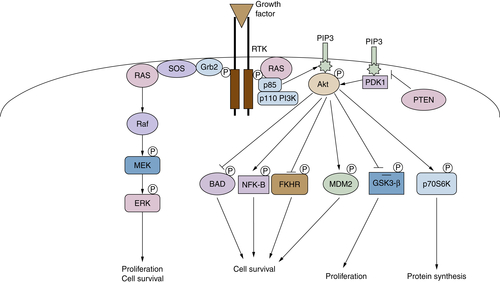
Oncogenes and Survival Signaling
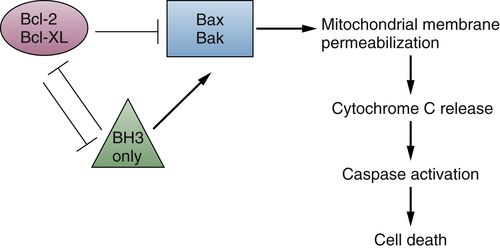
The Bcl-2 Family
PI3K-Dependent Pathways
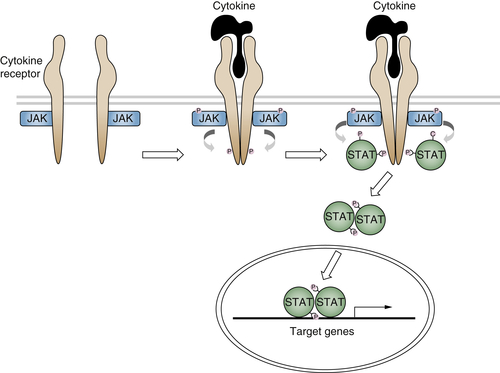
Cytokine Receptor Signaling
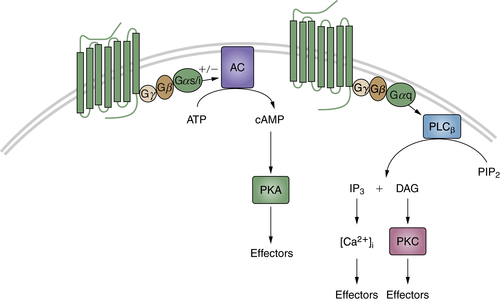
Neurotransmitters
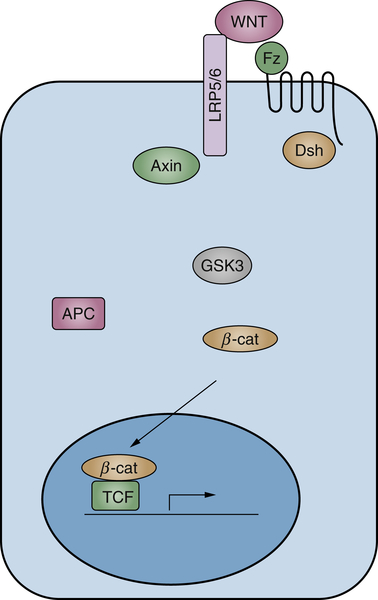
Wnt Signaling
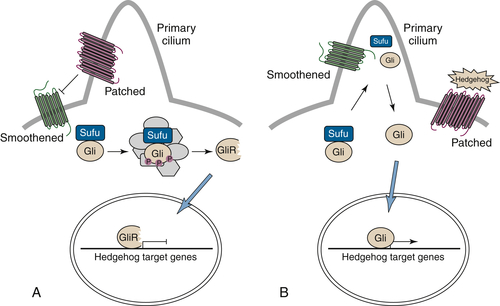
Hedgehog/Patched Signaling
Implications for Cancer Therapy
Table 2-1
Targeted Therapeutics Directed against Oncogene Products
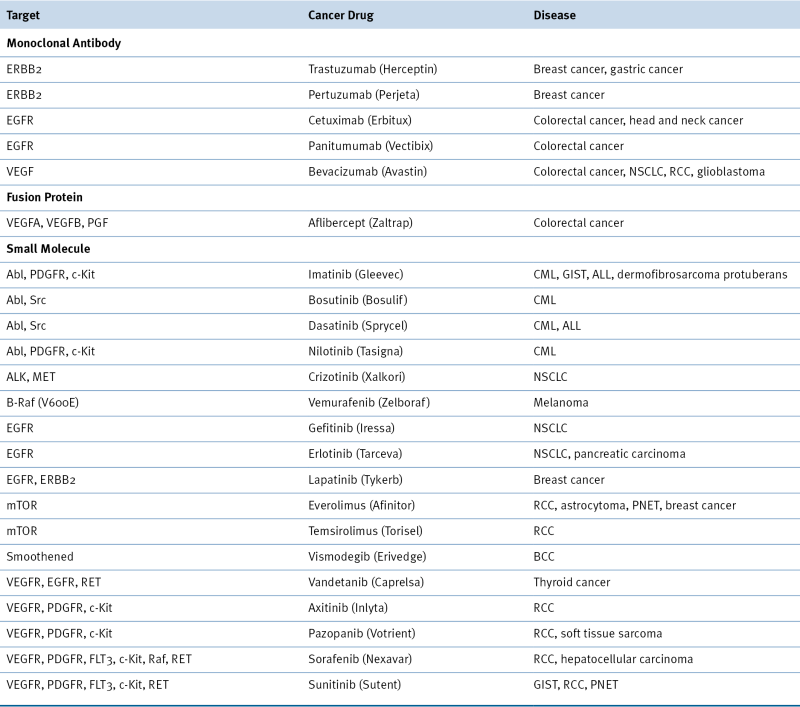
Drugs included in this table have been approved by the Food and Drug Administration (FDA).
ALL, acute lymphoblastic leukemia; BCC, basal cell carcinoma; CML, chronic myeloid leukemia; GIST, gastrointestinal stromal tumor; NSCLC, non-small-cell lung carcinoma; PGF, placental growth factor; PNET, pancreatic neuroendocrine tumor; RCC, renal cell carcinoma.
References
1. Pawson T. Dynamic control of signaling by modular adaptor proteins. Curr Opin Cell Biol. 2007;19:112–116.
2. Hunter T., Sefton B.M. Transforming gene product of Rous sarcoma virus phosphorylates tyrosine. Proc Natl Acad Sci U S A. 1980;77:1311–1315.
3. McBride O.W., Swan D.C., Santos E., Barbacid M., Tronick S.R., Aaronson S.A. Localization of the normal allele of T24 human bladder carcinoma oncogene to chromosome 11. Nature. 1982;300:773–774.
4. Doolittle R.F., Hunkapiller M.W., Hood L.E. et al. Simian sarcoma virus onc gene, v-sis, is derived from the gene (or genes) encoding a platelet-derived growth factor. Science. 1983;221:275–277.
5. Waterfield M.D., Scrace G.T., Whittle N. et al. Platelet-derived growth factor is structurally related to the putative transforming protein p28sis of simian sarcoma virus. Nature. 1983;304:35–39.
6. Downward J., Yarden Y., Mayes E. et al. Close similarity of epidermal growth factor receptor and v-erb-B oncogene protein sequences. Nature. 1984;307:521–527.
7. Carpenter G., Cohen S. Epidermal growth factor. J Biol Chem. 1990;265:7709–7712.
8. Blume-Jensen P., Hunter T. Oncogenic kinase signalling. Nature. 2001;411:355–365.
9. Lemmon M.A., Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2010;141:1117–1134.
10. Ullrich A., Schlessinger J. Signal transduction by receptors with tyrosine kinase activity. Cell. 1990;61:203–212.
11. Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2000;103:211–225.
12. Pietras K., Sjoblom T., Rubin K., Heldin C.H., Ostman A. PDGF receptors as cancer drug targets. Cancer Cell. 2003;3:439–443.
13. Nister M., Libermann T.A., Betsholtz C. et al. Expression of messenger RNAs for platelet-derived growth factor and transforming growth factor-alpha and their receptors in human malignant glioma cell lines. Cancer Res. 1988;48:3910–3918.
14. Maxwell M., Naber S.P., Wolfe H.J. et al. Coexpression of platelet-derived growth factor (PDGF) and PDGF-receptor genes by primary human astrocytomas may contribute to their development and maintenance. J Clin Invest. 1990;86:131–140.
15. Marra F. Chemokines in liver inflammation and fibrosis. Front Biosci. 2002;7:d1899–d1914.
16. Saharinen P., Eklund L., Pulkki K., Bono P., Alitalo K. VEGF and angiopoietin signaling in tumor angiogenesis and metastasis. Trends Mol Med. 2011;17:347–362.
17. Sherr C.J., Rettenmier C.W., Sacca R., Roussel M.F., Look A.T., Stanley E.R. The c-fms proto-oncogene product is related to the receptor for the mononuclear phagocyte growth factor, CSF-1. Cell. 1985;41:665–676.
18. Gherardi E., Birchmeier W., Birchmeier C., Vande Woude G. Targeting MET in cancer: rationale and progress. Nat Rev Cancer. 2012;12:89–103.
19. Yarden Y., Pines G. The ERBB network: at last, cancer therapy meets systems biology. Nat Rev Cancer. 2012;12:553–563.
20. Runeberg-Roos P., Saarma M. Neurotrophic factor receptor RET: structure, cell biology, and inherited diseases. Ann Med. 2007;39:572–580.
21. Birchmeier C., Birchmeier W., Gherardi E., Vande Woude G.F. Met, metastasis, motility and more. Nat Rev Mol Cell Biol. 2003;4:915–925.
22. Carroll M., Tomasson M.H., Barker G.F., Golub T.R., Gilliland D.G. The TEL/platelet-derived growth factor beta receptor (PDGF beta R) fusion in chronic myelomonocytic leukemia is a transforming protein that self-associates and activates PDGF beta R kinase-dependent signaling pathways. Proc Natl Acad Sci U S A. 1996;93:14845–14850.
23. Greco A., Mariani C., Miranda C. et al. The DNA rearrangement that generates the TRK-T3 oncogene involves a novel gene on chromosome 3 whose product has a potential coiled-coil domain. Mol Cell Biol. 1995;15:6118–6127.
24. Greco A., Pierotti M.A., Bongarzone I., Pagliardini S., Lanzi C., Della Porta G. TRK-T1 is a novel oncogene formed by the fusion of TPR and TRK genes in human papillary thyroid carcinomas. Oncogene. 1992;7:237–242.
25. Scheijen B., Griffin J.D. Tyrosine kinase oncogenes in normal hematopoiesis and hematological disease. Oncogene. 2002;21:3314–3333.
26. Meisenhelder J., Suh P.G., Rhee S.G., Hunter T. Phospholipase C-gamma is a substrate for the PDGF and EGF receptor protein-tyrosine kinases in vivo and in vitro. Cell. 1989;57:1109–1122.
27. Escobedo J.A., Navankasattusas S., Kavanaugh W.M., Milfay D., Fried V.A., Williams L.T. cDNA cloning of a novel 85 kd protein that has SH2 domains and regulates binding of PI3-kinase to the PDGF beta-receptor. Cell. 1991;65:75–82.
28. Nishimura R., Li W., Kashishian A. et al. Two signaling molecules share a phosphotyrosine-containing binding site in the platelet-derived growth factor receptor. Mol Cell Biol. 1993;13:6889–6896.
29. Kazlauskas A., Feng G.S., Pawson T., Valius M. The 64-kDa protein that associates with the platelet-derived growth factor receptor beta subunit via Tyr-1009 is the SH2-containing phosphotyrosine phosphatase Syp. Proc Natl Acad Sci U S A. 1993;90:6939–6943.
30. Arvidsson A.K., Rupp E., Nånberg E. et al. Tyr-716 in the platelet-derived growth factor beta-receptor kinase insert is involved in GRB2 binding and Ras activation. Mol Cell Biol. 1994;14:6715–6726.
31. Anderson D., Koch C.A., Grey L., Ellis C., Moran M.F., Pawson T. Binding of SH2 domains of phospholipase C gamma 1, GAP, and Src to activated growth factor receptors. Science. 1990;250:979–982.
32. Kaplan D.R., Morrison D.K., Wong G., McCormick F., Williams L.T.P.D.G.F. beta-receptor stimulates tyrosine phosphorylation of GAP and association of GAP with a signaling complex. Cell. 1990;61:125–133.
33. Molloy C.J., Bottaro D.P., Fleming T.P., Marshall M.S., Gibbs J.B., Aaronson S.A. PDGF induction of tyrosine phosphorylation of GTPase activating protein. Nature. 1989;342:711–714.
34. Ralston R., Bishop J.M. The product of the protooncogene c-src is modified during the cellular response to platelet-derived growth factor. Proc Natl Acad Sci U S A. 1985;82:7845–7849.
35. Berridge M.J., Irvine R.F. Inositol phosphates and cell signalling. Nature. 1989;341:197–205.
36. Griner E.M., Kazanietz M.G. Protein kinase C and other diacylglycerol effectors in cancer. Nat Rev Cancer. 2007;7:281–294.
37. Engelman J.A., Luo J., Cantley L.C. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet. 2006;7:606–619.
38. Vanhaesebroeck B., Stephens L., Hawkins P. PI3K signalling: the path to discovery and understanding. Nat Rev Mol Cell Biol. 2012;13:195–203.
39. Karnoub A.E., Weinberg R.A. Ras oncogenes: split personalities. Nat Rev Mol Cell Biol. 2008;9:517–531.
40. Buday L., Downward J. Epidermal growth factor regulates p21ras through the formation of a complex of receptor, Grb2 adapter protein, and Sos nucleotide exchange factor. Cell. 1993;73:611–620.
41. Margolis B., Skolnik E.Y. Activation of Ras by receptor tyrosine kinases. J Am Soc Nephrol. 1994;5:1288–1299.
42. Vigil D., Cherfils J., Rossman K.L., Der C.J. Ras superfamily GEFs and GAPs: validated and tractable targets for cancer therapy? Nat Rev Cancer. 2010;10:842–857.
43. Barbacid M. ras genes. Annu Rev Biochem. 1987;56:779–827.
44. Santos E., Nebreda A.R. Structural and functional properties of ras proteins. FASEB J. 1989;3:2151–2163.
45. Lassar A.B., Thayer M.J., Overell R.W., Weintraub H. Transformation by activated ras or fos prevents myogenesis by inhibiting expression of MyoD1. Cell. 1989;58:659–667.
46. Klesse L.J., Parada L.F. p21 ras and phosphatidylinositol-3 kinase are required for survival of wild-type and NF1 mutant sensory neurons. J Neurosci. 1998;18:10420–10428.
47. Terada K., Kaziro Y., Satoh T. Ras is not required for the interleukin 3-induced proliferation of a mouse pro-B cell line, BaF3. J Biol Chem. 1995;270:27880–27886.
48. Johnson L. et al. K-ras is an essential gene in the mouse with partial functional overlap with N-ras [published erratum appears in Genes Dev 1997 Dec 1;11(23):3277]. Genes Dev. 1997;11:2468–2481.
49. Umanoff H., Edelmann W., Pellicer A., Kucherlapati R. The murine N-ras gene is not essential for growth and development. Proc Natl Acad Sci U S A. 1995;92:1709–1713.
50. Schubbert S., Shannon K., Bollag G. Hyperactive Ras in developmental disorders and cancer. Nat Rev Cancer. 2007;7:295–308.
51. Serrano M., Lin A.W., McCurrach M.E., Beach D., Lowe S.W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell. 1997;88:593–602.
52. Cichowski K., Jacks T. NF1 tumor suppressor gene function: narrowing the GAP. Cell. 2001;104:593–604.
53. Kolch W., Heidecker G., Lloyd P., Rapp U.R. Raf-1 protein kinase is required for growth of induced NIH/3T3 cells. Nature. 1991;349:426–428.
54. Wellbrock C., Karasarides M., Marais R. The RAF proteins take centre stage. Nat Rev Mol Cell Biol. 2004;5:875–885.
55. Maurer G., Tarkowski B., Baccarini M. Raf kinases in cancer roles and therapeutic opportunities. Oncogene. 2011;30:3477–3488.
56. Dhillon A.S., Hagan S., Rath O., Kolch W. MAP kinase signalling pathways in cancer. Oncogene. 2007;26:3279–3290.
57. Anjum R., Blenis J. The RSK family of kinases: emerging roles in cellular signalling. Nat Rev Mol Cell Biol. 2008;9:747–758.
58. Bermudez O., Pages G., Gimond C. The dual-specificity MAP kinase phosphatases: critical roles in development and cancer. Am J Physiol Cell Physiol. 2010;299:C189–C202.
59. Musgrove E.A., Caldon C.E., Barraclough J., Stone A., Sutherland R.L. Cyclin D as a therapeutic target in cancer. Nat Rev Cancer. 2011;11:558–572.
60. Beroukhim R. et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463:899–905.
61. Davies H., Bignell G.R., Cox C. et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954.
62. Palanisamy N., Ateeq B., Kalyana-Sundaram S. et al. Rearrangements of the RAF kinase pathway in prostate cancer, gastric cancer and melanoma. Nat Med. 2010;16:793–798.
63. Wagner E.F., Nebreda A.R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat Rev Cancer. 2009;9:537–549.
64. Van Beveren C., van Straaten F., Curran T., Muller R., Verma I.M. Analysis of FBJ-MuSV provirus and c-fos (mouse) gene reveals that viral and cellular fos gene products have different carboxy termini. Cell. 1983;32:1241–1255.
65. Maki Y., Bos T.J., Davis C., Starbuck M., Vogt P.K. Avian sarcoma virus 17 carries the jun oncogene. Proc Natl Acad Sci U S A. 1987;84:2848–2852.
66. Miller A.D., Curran T., Verma I.M. c-fos protein can induce cellular transformation: a novel mechanism of activation of a cellular oncogene. Cell. 1984;36:51–60.
67. Shaulian E., Karin M. AP-1 as a regulator of cell life and death. Nat Cell Biol. 2002;4:E131–E136.
68. Adhikary S., Eilers M. Transcriptional regulation and transformation by Myc proteins. Nat Rev Mol Cell Biol. 2005;6:635–645.
69. Dang C.V. MYC on the path to cancer. Cell. 2012;149:22–35.
70. Vita M., Henriksson M. The Myc oncoprotein as a therapeutic target for human cancer. Semin Cancer Biol. 2006;16:318–330.
71. Chipuk J.E., Moldoveanu T., Llambi F., Parsons M.J., Green D.R. The BCL-2 family reunion. Mol Cell. 2010;37:299–310.
72. Strasser A., Cory S., Adams J.M. Deciphering the rules of programmed cell death to improve therapy of cancer and other diseases. EMBO J. 2011;30:3667–3683.
73. Cantley L.C. The phosphoinositide 3-kinase pathway. Science. 2002;296:1655–1657.
74. Engelman J.A. Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer. 2009;9:550–562.
75. Chang H.W. et al. Transformation of chicken cells by the gene encoding the catalytic subunit of PI 3-kinase. Science. 1997;276:1848–1850.
76. Song M.S., Salmena L., Pandolfi P.P. The functions and regulation of the PTEN tumour suppressor. Nat Rev Mol Cell Biol. 2012;13:283–296.
77. Taniguchi T. Cytokine signaling through nonreceptor protein tyrosine kinases. Science. 1995;268:251–255.
78. Wang X., Lupardus P., Laporte S.L., Garcia K.C. Structural biology of shared cytokine receptors. Annu Rev Immunol. 2009;27:29–60.
79. Stark G.R., Darnell Jr. J.E. The JAK-STAT pathway at twenty. Immunity. 2012;36:503–514.
80. Kisseleva T., Bhattacharya S., Braunstein J., Schindler C.W. Signaling through the JAK/STAT pathway, recent advances and future challenges. Gene. 2002;285:1–24.
81. Quintas-Cardama A., Kantarjian H., Cortes J., Verstovsek S. Janus kinase inhibitors for the treatment of myeloproliferative neoplasias and beyond. Nat Rev Drug Discov. 2011;10:127–140.
82. Cocault L., Bouscary D., Le Bousse Kerdiles C. et al. Ectopic expression of murine TPO receptor (c-mpl) in mice is pathogenic and induces erythroblastic proliferation. Blood. 1996;88:1656–1665.
83. Pikman Y., Lee B.H., Mercher T. et al. MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med. 2006;3 e270.
84. Yan R., Luo H., Darnell Jr. J.E., Dearolf C.R.A. JAK-STAT pathway regulates wing vein formation in Drosophila. Proc Natl Acad Sci U S A. 1996;93:5842–5847.
85. Lacronique V., Boureux A., Valle V.D. et al. A TEL-JAK2 fusion protein with constitutive kinase activity in human leukemia. Science. 1997;278:1309–1312.
86. Pilati C., Amessou M., Bihl M.P. et al. Somatic mutations activating STAT3 in human inflammatory hepatocellular adenomas. J Exp Med. 2011;208:1359–1366.
87. Yu H., Pardoll D., Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798–809.
88. Dorsam R.T., Gutkind J.S. G-protein-coupled receptors and cancer. Nat Rev Cancer. 2007;7:79–94.
89. Landis C.A., Masters S.B., Spada A., Pace A.M., Bourne H.R., Vallar L. GTPase inhibiting mutations activate the alpha chain of Gs and stimulate adenylyl cyclase in human pituitary tumours. Nature. 1989;340:692–696.
90. Lyons J., Landis C.A., Harsh G. et al. Two G protein oncogenes in human endocrine tumors. Science. 1990;249:655–659.
91. Markwalder R., Reubi J.C. Gastrin-releasing peptide receptors in the human prostate: relation to neoplastic transformation. Cancer Res. 1999;59:1152–1159.
92. Uemura H., Hasumi H., Ishiguro H., Teranishi J., Miyoshi Y., Kubota Y. Renin-angiotensin system is an important factor in hormone refractory prostate cancer. Prostate. 2006;66:822–830.
93. Clerc P., Leung-Theung-Long S., Wang T.C. et al. Expression of CCK2 receptors in the murine pancreas: proliferation, transdifferentiation of acinar cells, and neoplasia. Gastroenterology. 2002;122:428–437.
94. Lappano R., Maggiolini M. G protein-coupled receptors: novel targets for drug discovery in cancer. Nat Rev Drug Discov. 2011;10:47–60.
95. Reubi J.C. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev. 2003;24:389–427.
96. van Essen M., Krenning E.P., Kam B.L., de Jong M., Valkema R., Kwekkeboom D.J. Peptide-receptor radionuclide therapy for endocrine tumors. Nat Rev Endocrinol. 2009;5:382–393.
97. Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–480.
98. Grigoryan T., Wend P., Klaus A., Birchmeier W. Deciphering the function of canonical Wnt signals in development and disease: conditional loss- and gain-of-function mutations of beta-catenin in mice. Genes Dev. 2008;22:2308–2341.
99. Reya T., Clevers H. Wnt signalling in stem cells and cancer. Nature. 2005;434:843–850.
100. Clevers H., Nusse R. Wnt/beta-catenin signaling and disease. Cell. 2012;149:1192–1205.
101. MacDonald B.T., Tamai K., He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17:9–26.
102. Nusse R., Varmus H.E. Wnt genes. Cell. 1992;69:1073–1087.
103. Brown A.M., Wildin R.S., Prendergast T.J., Varmus H.E. A retrovirus vector expressing the putative mammary oncogene int-1 causes partial transformation of a mammary epithelial cell line. Cell. 1986;46:1001–1009.
104. Bafico A., Gazit A., Wu-Morgan S.S., Yaniv A., Aaronson S.A. Characterization of Wnt-1 and Wnt-2 induced growth alterations and signaling pathways in NIH3T3 fibroblasts. Oncogene. 1998;16:2819–2825.
105. Bafico A., Liu G., Goldin L., Harris V., Aaronson S.A. An autocrine mechanism for constitutive Wnt pathway activation in human cancer cells. Cancer Cell. 2004;6:497–506.
106. Akiri G., Cherian M.M., Vijayakumar S., Liu G., Bafico A., Aaronson S.A. Wnt pathway aberrations including autocrine Wnt activation occur at high frequency in human non-small-cell lung carcinoma. Oncogene. 2009;28:2163–2172.
107. Vijayakumar S., Liu G., Rus I.A. et al. High-frequency canonical Wnt activation in multiple sarcoma subtypes drives proliferation through a TCF/beta-catenin target gene, CDC25A. Cancer Cell. 2011;19:601–612.
108. Polakis P. Drugging Wnt signalling in cancer. EMBO J. 2012;31:2737–2746.
109. Seshagiri S., Stawiski E.W., Durinck S. et al. Recurrent R-spondin fusions in colon cancer. Nature. 2012;488:660–664.
110. Lum L., Beachy P.A. The Hedgehog response network: sensors, switches, and routers. Science. 2004;304:1755175–1755179.
111. Briscoe J., Therond P. Hedgehog signaling: from the Drosophila cuticle to anti-cancer drugs. Dev Cell. 2005;8:143–151.
112. Ingham P.W., Nakano Y., Seger C. Mechanisms and functions of Hedgehog signalling across the metazoa. Nat Rev Genet. 2011;12:393–406.
113. Ryan K.E., Chiang C. Hedgehog secretion and signal transduction in vertebrates. J Biol Chem. 2012;287:17905–17913.
114. Hahn H. et al. Mutations of the human homolog of Drosophila patched in the nevoid basal cell carcinoma syndrome. Cell. 1996;85:841–851.
115. Xie J., Murone M., Luoh S.M. et al. Activating Smoothened mutations in sporadic basal-cell carcinoma. Nature. 1998;391:90–92.
116. Yauch R.L., Gould S.E., Scales S.J. et al. A paracrine requirement for hedgehog signalling in cancer. Nature. 2008;455:406–410.
117. Ng J.M., Curran T. The Hedgehog’s tale: developing strategies for targeting cancer. Nat Rev Cancer. 2011;11:493–501.
118. Weinstein I.B. Cancer. Addiction to oncogenes—the Achilles heal of cancer. Science. 2002;297:63–64.
119. Bollag G., Hirth P., Tsai J. et al. Clinical efficacy of a RAF inhibitor needs broad target blockade in BRAF-mutant melanoma. Nature. 2010;467:596–599.
120. Flaherty K.T., Puzanov I., Kim K.B. et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809–819.
121. Poulikakos P.I., Rosen N. Mutant BRAF melanomas—dependence and resistance. Cancer Cell. 2011;19:11–15.
122. Prahallad A., Sun C., Huang S. et al. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature. 2012;483:100–103.