24 Proprioception and Neuromuscular Control
Human beings are unique in their capacity to propel themselves through their environment in an upright posture. This ability is achieved through a complex interaction of lower limb muscle activity coordinated by the central nervous system (CNS). To maintain balance and postural control we rely on sensory information from the periphery from our visual, vestibular, and somatosensory systems. The nervous system integrates this peripheral afferent information to maintain postural control during stance.
Control of locomotion, including walking or running, occurs through complex neural pathways in the spinal cord called central pattern generators or limb controllers. These motor programs for locomotion are automatic but are modulated by the CNS through feedback and feedforward mechanisms. The feedforward mechanism operates on the premise of initiating a motor response in anticipation of a load or activity that will disrupt the integrity of a joint and gauges the response from previous experiences. In contrast, the feedback system operates directly in response to a potentially destabilizing event by using a normal reference point to monitor the muscle activity necessary to restore homeostasis.1
Both feedback and feedforward systems rely on processing of afferent information from the periphery at different levels of the CNS (spinal cord, brainstem and cerebellum, and cerebral cortex), with the end result being coordinated muscle activity during movement to maintain joint stability.1 The motor response varies depending on joint position, type of force, direction of force, and which higher center predominates in processing the information.
Segmental spinal reflexes involve the processing of afferent input between peripheral receptors in the muscle spindle and Golgi tendon organs at the musculotendinous junction with the efferent output of motor neurons in the ventral horn of the spinal cord. On the most basic level, monosynaptic reflexes produce an excitatory or inhibitory efferent motor response to the stimulus received from the periphery. Along with the physiologic properties of the muscle itself (length-tension curve), these peripheral receptors potentially assist in modulating muscle stiffness, with muscle tension varying according to the amount of afferent input.1
The afferent information received in the cortical area of the brain from peripheral mechanoreceptors produces a voluntary motor response to potential disturbances in functional joint stability. The latency of the response is usually greater than 120 msec and sometimes longer, depending on the amount of information in the environment being processed. In addition to the response to an environmental stimulus, the potential exists for a theorized motor program operating under the assumption that the individual components of performing skilled movements, such as swinging a bat, that require sequential steps would be difficult to enact successfully without having a preprogrammed set of instructions to optimize efficiency, speed, and coordinated muscle activity.2
The function of the cerebellum and brainstem is to integrate peripheral feedback from the environment with the motor commands from the cerebral cortex to enable humans to perform skilled and coordinated movement. The action of these neural centers allows the adjustments needed to carry out an intended motor skill with precision and efficiency.2
A PubMed search of the terms proprioception and neuromuscular control was performed in October 2010. The results identified 305 references, the majority (260) of which have been published during the last decade. However, when the search is limited to higher levels of evidence, including randomized controlled trials (RCTs), systematic reviews, and metaanalysis studies, the actual number is 37 high-quality studies. In one such study, Riemann et al3 performed a literature review to identify sensorimotor assessment techniques, many of which are described throughout this chapter. Their conclusions indicate that the complex interactions and relationships among the individual components of the sensorimotor system make measuring and analyzing specific characteristics and functions difficult. Additionally, the specific assessment techniques used to measure a variable can influence the results obtained. Optimizing the application of sensorimotor research to clinical settings can best be accomplished through the use of common nomenclature to describe the underlying physiologic mechanisms and specific measurement techniques.
Definitions
Review of the orthopedic and musculoskeletal rehabilitation literature identifies many different versions of definitions for the terms associated with joint proprioception and neuromuscular control. In Goetz’s Textbook of Clinical Neurology, proprioception is defined as any postural, positional, or kinetic information provided to the CNS by sensory receptors in muscles, tendons, joints, or skin.4 Other texts define proprioception as “awareness of the position and movements of our limbs, fingers, and toes derived from receptors in the muscles, tendons and joints.”5 Sherrington’s classic definition of proprioception is “afferent information arising from the proprioceptive field,” and mechanoreceptors or proprioceptors were identified as being the source of the origination of this afferent information.6
These original definitions of the term proprioception continue to be used today; however, a more advanced definition of the sensory functions that encompass human proprioceptive function is clearly needed. In a classic monograph titled Physiologie des Muskelsinnes, Goldsheider7 proposed that muscle sense be divided into four distinct and separate sensory functions. These functions were described as sensation of passive movements, sensation of active movements, sensation of position, and appreciation or sensation of heaviness and resistance. These original classifications or definitions have been expanded to decrease confusion. The sensation of passive movements is considered to be a product of sensations induced by external forces that result in a change in limb position with noncontracting muscles. The sensation of active movement (or kinesthesia as it is now better known) encompasses the appreciation of change in position of a limb with contracting muscles. Appreciation of the position of a limb in space has been termed stagnosia, and finally, in the presence of tension, appreciation of force applied during a voluntary contraction has been termed dynamaesthesia.8 Although these expanded definitions found in the classic literature provide additional information about human proprioception, adaptations of these classic definitions have been suggested and are used for the purposes of this chapter (Box 24-1).
Afferent neurobiology of the joint
Early work on afferent proprioceptive function of the human joint included investigations into the role of joint- and muscle-based afferent receptors in human active and passive movement and detection of joint position.8 In 1898 Goldsheider proposed that the sensation of passive movements was solely the product of joint-based receptors. This view is still widely accepted today for passive movements.7,8
The view up until the 1970s about the sensory feedback of active human movements was that when voluntary movement was initiated by the cerebral cortex, only low-level control was presented by the receptors in muscles and tendons. This sensory information from the muscles and tendons yielded information to the spinal cord and some subcortical extrapyramidal parts of the brain such as the cerebellum but played no contributing role in conscious sensation, which remained in the province of the joint receptors.8 In the early 1970s, however, important research by Goodwin et al9 and Eklund10 independently demonstrated the important role that muscular receptors play in contributing to sensations of active movement qualitatively. This section of the chapter focuses on both joint- and muscle-based afferent receptors to allow the clinician a more complete understanding of the sources of afferent information in the human body. This will later lead to a greater understanding of how specific treatment strategies can be used clinically to improve proprioceptive and neuromuscular function in both upper and lower extremity rehabilitation (Box 24-2).
Classification of Afferent Mechanoreceptor
Mechanoreceptors are sensory neurons or peripheral afferents located within joint capsular tissues, ligaments, tendons, muscle, and skin.11,12 Deformation or stimulation of the tissues in which the mechanoreceptors lie produces gated release of sodium, which elicits an action potential.13 Four primary types of afferent mechanoreceptors have been classified and are commonly present in noncontractile capsular and ligamentous structures in human joints (Table 24-1).
Type I articular receptors are traditionally globular or ovoid corpuscles with a very thin capsule. They are numerous in the capsular tissues of all the limb joints, as well as the apophyseal joints of the vertebral column. Wyke12 reported that the population of type I receptors appears to be more dense in proximal joints than in distal joints. Type I receptors are typically located in the superficial layers of the joint capsule.
Physiologically, type I receptors are low-threshold, slowly adapting mechanoreceptors. A proportion of type I receptors are always active in every joint position.12 The resting discharge of type I receptors allows the body to know where the limb is placed and receive constant input on limb position in virtually any joint position. The type I receptor is categorized as both a static and dynamic mechanoreceptor12 whose discharge pattern signals static joint position, changes in intraarticular pressure, and the direction, amplitude, and velocity of joint movements.
Type II mechanoreceptors are elongated, conical corpuscles with thick multilaminated connective tissue capsules. These type II corpuscles are present in the fibrous capsules of all joints but are reported to be present in greater number in distal joints than in proximal joints.1 Type II corpuscles are located in the deeper layers of the fibrous joint capsule, particularly at the border between the fibrous capsule and the subsynovial fibroadipose tissue and often alongside articular blood vessels. Type II mechanoreceptors are low-threshold, rapidly adapting receptors and are reported to be entirely inactive in immobile joints.12 These receptors become activated for very brief moments (1 second or less) at the onset of joint movement. The type II receptor is considered to be a dynamic mechanoreceptor whose brief, high-velocity discharges signal joint acceleration and deceleration during both active and passive joint movements.
The type I and type II mechanoreceptors described in the preceding paragraphs are the primary receptors located in the joint capsule. Type III receptors are primarily confined to the joint ligamentous structures. These type III receptors are found in both intrinsic and extrinsic ligamentous structures12 and are similar in nature to the Golgi tendon organs found in tendons, as discussed in later sections of this chapter. Type III receptors are found predominantly in the superficial surfaces of the joint ligaments, near their bony attachments. Research delineating the type III mechanoreceptor classifies this receptor as a high-threshold, slowly adapting structure, again similar in nature to the Golgi tendon organ. These type III receptors are completely inactive in immobile joints and become active or stimulated only toward the extreme ranges of joint motion where the ligamentous structures become taut. When considerable stress is generated in the joint ligaments, the type III receptor will become actively stimulated. Wyke14 also reported that type III receptors become activated with longitudinal traction on the limbs; the receptors remain activated centripetally at a high velocity only if extreme joint displacement or joint traction is maintained.
The final joint receptor to be discussed in this section is the type IV receptor. These receptors are noncorpuscular, unlike type I, II, and III receptors, and are represented by plexuses of small unmyelinated nerve fibers or free nerve endings. Type IV receptors are typically distributed throughout the fibrous joint capsule, adjacent periosteum, and articular fat pads. The type IV receptor represents the pain receptor system of articular tissues and is entirely inactive in normal circumstances. Marked mechanical deformation or chemical irritation such as exposure of the nerve endings to agents such as histamine, bradykinin, and other inflammatory exudates produced by damaged or necrosing tissues can stimulate activation of the type IV receptor.12,14,15
Afferent mechanoreceptors in the lower extremity
The distribution of afferent articular nerves in synovial joints consists of medium and large myelinated fibers innervating the small end-organs or mechanoreceptors throughout joint tissue. These nerves represent approximately 55% of the total quantity of articular nerves, with the remaining 45% consisting of small unmyelinated fibers that transmit nociception or pain sensation.12
Type I or Ruffini receptors located in the superficial layers of the joint capsule are low-threshold, slowly adapting mechanoreceptors. These receptors respond to changing mechanical stress and are always active because of the gradient pressure difference in the joint capsule. They undergo deformation with natural movement because of their location in the superficial portion of the joint capsule. In the limbs, type I receptors are found to be more densely distributed in the proximal joints of the hip and are not as prevalent in the distal joints of the ankle.12 Ruffini receptors have also been found in the meniscofemoral, cruciate, and collateral ligaments of the knee.2
Type II or pacinian receptors are located in the deep layers of the joint capsule, the meniscofemoral, cruciate, and collateral ligaments of the knee. In addition, type II receptors are located in the intraarticular and extraarticular fat pads of all synovial joints. These pacinian receptors are more prevalent in distal joints such as the ankle and are less densely distributed in proximal joints such as the hip. They function as rapidly adapting, low-threshold receptors and respond to acceleration, deceleration, and passive joint movement but are silent during inactivity and joint movement at constant velocity.2
Type III or Golgi tendon organ–like endings are found predominantly in intraarticular and extraarticular joint ligaments, including the collateral ligaments and cruciate ligaments in the knee.12 These receptors have also been identified in the menisci of the knee.2 Type III Golgi tendon organ–like endings are structurally identical to the Golgi tendon organ receptors and function as slowly adapting, high-threshold receptors with a function similar to that of the Golgi tendon organs found in tendons.
Type IV free nerve endings function as the pain receptor or nociception system in synovial joints. These type IV receptor nerve endings are found throughout the joints of the extremities in the fibrous capsule and adjacent periosteum and in the articular fat pads and are the most prevalent receptor type in the knee menisci. They are completely inactive in normal situations and are activated by marked mechanical deformation or chemical stimuli resulting from an inflammatory response.2
Afferent joint receptors in the upper extremity
The classification system mentioned earlier for the four primary types of mechanoreceptors found in human noncontractile capsular and ligamentous tissues described by Wyke12,14 provides generalized information about the location of these receptors in the human body. Vangsness et al16 studied the neural histology of the human shoulder joint, including the glenohumeral ligaments, labrum, and subacromial bursa. They found two types of mechanoreceptors and free nerve endings in the glenohumeral joint capsular ligaments. Two types of slowly adapting Ruffini end-organs and rapidly adapting pacinian corpuscles were identified in the superior, middle, and inferior portions of the glenohumeral ligaments. The most common mechanoreceptor was the classic Ruffini end-organ in the capsular ligaments of the glenohumeral joint. Pacinian corpuscles were less abundant overall; however, Kikuchi17 and Shimoda18 reported that type II pacinian corpuscles were more commonly found in the capsular ligaments of the human glenohumeral joint than in the human knee. Analysis of the coracoclavicular and acromioclavicular ligaments showed equal distribution of type I and II mechanoreceptors. Morisawa et al13 identified type I, II, III, and IV mechanoreceptors in human coracoacromial ligaments. These reviews show how the capsular ligaments of the glenohumeral joint aid in the provision of afferent proprioceptive input by their inherent distribution of both type I Ruffini mechanoreceptors and the more rapidly adapting pacinian receptors. A rapidly adapting receptor such as the pacinian receptor can identify changes in tension in the joint capsular ligaments but quickly decreases its input once the tension becomes constant.16 In this way the type II receptor has the ability to monitor acceleration and deceleration of the tension on a ligament.
Several authors have also studied the labrum and subacromial bursa. Vangsness et al16 reported that no evidence of mechanoreceptors was found in the glenoid labrum; however, free nerve endings were noted in the fibrocartilage tissue in the peripheral half. The subacromial bursa was found to have diffuse, yet copious free nerve endings, with no evidence of larger, more complex mechanoreceptors. Ide et al19 also studied the subacromial bursa, taken from three cadavers, and found a copious supply of free nerve endings, most of which were present on the roof side of the subacromial arch, which is exposed to impingement-type stress. Unlike the study by Vangsness et al,16 Ide et al19 did find evidence of both Ruffini and pacinian mechanoreceptors in the subacromial bursa. Their findings suggest that the subacromial bursa receives both nociceptive and proprioceptive stimuli and may play a role in regulation of shoulder movement. Further research into the exact distribution of these important structures in the human shoulder is indicated to give clinicians further information and enhance the understanding of proprioceptive function of the shoulder.
Afferent receptors of contractile structures in the upper extremity
In addition to the afferent structures found in noncontractile tissues of the human shoulder (joint capsule, subacromial bursa, and intrinsic and extrinsic ligaments), significant contributions to the regulation of human movement and proprioceptive feedback are obtained from receptors located in contractile structures.
Two of the primary mechanisms for afferent feedback from the muscle-tendon unit are the muscle spindle and the Golgi tendon organ.15,20 Research classifying muscle spindles has traditionally grouped intrafusal muscle fibers into two groups based on the type of afferent projections.20,21 These two groups consist of nuclear bag and nuclear chain fibers. Nuclear chain fibers project from large afferent axons.20,21 Nuclear bag fibers are innervated by γ1 (dynamic) motor neurons and are more sensitive to the rate of change in muscle length, such as that occurring during rapid stretch of a muscle during an eccentric contraction or passive stretch.20 Intrafusal nuclear chain fibers are innervated by γ2 (static) motor neurons and are more sensitive to static muscle length. The combination of nuclear chain and nuclear bag fibers allows afferent communication from the muscle-tendon unit to remain sensitive over a wide range of joint motion during both reflex and voluntary activation (Table 24-2).
Muscle spindles provide much of the primary information for motor learning, including muscle length and joint position. Upper levels of the CNS can bias the sensitivity of muscle spindle input and sampling.20 Muscle spindles do not occur in similar density in all muscles in the human body. Spindle density is probably related to muscle function, with greater densities of muscle spindles being reported in muscles that initiate and control fine movements or maintain posture. Muscles that cross the front of the shoulder, such as the pectoralis major and biceps, have a very high number of muscle spindles per unit of muscle weight.22 Muscles with attachment to the coracoid, such as the biceps, pectoralis minor, and coracobrachialis, also have high spindle densities. Lower spindle densities have been reported for the rotator cuff muscle-tendon units, with the subscapularis and infraspinatus having greater densities than the supraspinatus and teres minor.22 This lower rotator cuff spindle density probably suggests synergistic mechanoreceptor activation with the scapulothoracic musculature during movement of the glenohumeral joint.20,23 This coupled or shared mechanoreceptor activation is an example of the kinetic link or proximal-to-distal sequencing that occurs with predictable or programmed movement patterns in the human body.24 This kinetic link activation concept is further demonstrated by the deltoid/rotator cuff force couple23 and other important biomechanical features of the human glenohumeral joint and is discussed later in this chapter.
The second major aspect of musculotendinous afferent activity is the Golgi tendon organ. These tendinous mechanoreceptors are present in the human shoulder and respond to the tension generated by muscular contraction.15,20 Activation of the Golgi tendon organs relays afferent feedback about muscle tension and joint position. Additionally, as a protective mechanism, activation of the tension-sensitive Golgi tendon organ produces a protective mechanism that causes relaxation of the agonist muscle that is undergoing tension, with simultaneous stimulation of antagonistic musculature.
Clincal assessment of proprioception in the lower extremity
The two primary tests measuring proprioception and kinesthetic awareness in the knee joint are the threshold to detection of passive motion (TTDPM) for movement sense and reproduction of angular position for joint position sense. The TTDPM test has been more standardized in the literature.2,25,26 The method described by Barrack et al27 and Skinner et al28 involves placing the subject in a seated position with the leg hanging freely over the seat and suspended by a motorized pulley system in 90° of flexion (Fig. 24-1). Tactile, visual, and auditory cues are eliminated with the use of custom-fitted Jobst air splints and wearing of a blindfold. Initiation of movement into either flexion or extension proceeds at a rate of angular deflection of 0.5°/sec. When subjects initially detect movement to occur, they engage a control switch to indicate that the test leg has been moved.28
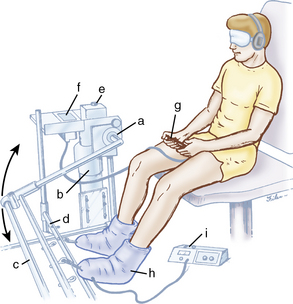
Figure 24-1 Proprioceptive testing device. a, Rotational transducer; b, motor; c, moving arm; d, stationary arm; e, control panel; f, digital microprocessor; g, handheld disengage switch; h, pneumatic compression boot; and i, pneumatic compression device. The threshold for detecting passive movement is assessed by measuring angular displacement until the subject senses motion in the knee.
(From Lephart, S.M., Kocher, M.S., Fu, F.H., et al. [1992]: Proprioception following anterior cruciate ligament reconstruction. J. Sport Rehabil., 1:188–196.)
Testing for joint position sense involves passive movement of the extremity to a specified angle by the clinician, holding of the position for several seconds, and passive return of the extremity to the starting reference position. The patient is then asked to actively move the extremity to the specified angle without visual input. The difference between the actual and replicated angle can be calculated as either an absolute or a real angular error. With absolute error, only the magnitude of the error is determined, and whether the subject overestimates or underestimates knee position is not considered. Real error calculations, however, consider both the magnitude and direction of the error and can be used to determine whether a subject overestimates or underestimates the reference angle.29 Barrack et al30 demonstrated through studies on proprioception that extremities with no evidence of pathologic conditions have a high degree of symmetry in joint position sense.
Because essentially no standard protocols have been established for measuring joint position sense or for performing joint replication tests, many variations exist, including apparatuses used for angular measurement, starting reference angle, active or passive reproduction, and open chain (seated) versus closed chain (standing).31 Lattanzio et al25 and Marks and Quinney32 used closed chain weight-bearing joint replications and reported a high degree of accuracy. Their results may be due to the fact that proprioceptive input is greater in the standing weight-bearing position, in which multiple joints are being loaded.
Single-limb postural stability tests have also been used for measuring the amount of sway in individuals with complaints of ankle instability. Tropp et al33 developed such a test for measuring ankle instability that has been used with variations throughout the years. Individuals stand for 60 seconds on a force platform, and the instantaneous center of pressure is recorded along a graph; the magnitude of sway is compared with that on the uninvolved side.
Single-leg hop tests are often used for assessing stability in patients with pathologic knee or ankle conditions. Variations of the test include single-leg or triple-leg hop tests for distance, the crossover hop test, and the timed hop test. The relationship of hop tests to functional parameters such as instability, proprioception, and leg strength has been inconclusive in studies to date.
Assessment of proprioception and neuromuscular control in the upper extremity with specific reference to the human shoulder
Determination of which patients require particular emphasis in rehabilitation on restoring proprioception and neuromuscular control requires the use of clinical assessment techniques. In this section, techniques used in research investigations, as well as in clinical applications, to allow the clinician to perform a detailed evaluation are reviewed.
Primary Measures of Proprioception and Neuromuscular Control for the Shoulder
Evaluation of proprioception and neuromuscular control in the human shoulder encompasses both afferent and efferent neural function, as well as the resulting muscular activation patterns.15 Proprioception for the purposes of this and many other articles, texts, and chapters2,15,34 consists of three major submodalities: kinesthesia, joint position sense, and sensation of resistance. Separate techniques can be used to assess each of these aspects of proprioception.
Measurement of Kinesthesia
Assessment of glenohumeral joint kinesthesia has been performed with a test called the TTDPM. This test assesses the subject’s or patient’s ability to detect a passive movement occurring typically at very slow angular velocities.2,15,35 Elaborate testing devices have been used in several studies that have reported on the TTDPM, such as an instrumented (motorized) shoulder wheel35 and other devices such as the one used by the University of Pittsburgh, whose characteristics are described next (Fig. 24-2).2 Extensive research2,15,36 using the TTDPM test has resulted in the selection and recommendation of slow angular velocities (0.5° to 2°/sec) to enhance the reliability of data acquisition. In addition to the device used, blindfolds, earphones, and a pneumatic cuff are recommended to eliminate cues from the visual, auditory, and tactile realm. This ensures that only joint kinesthesia is being assessed and not simply visual or auditory responses to perceived movement.
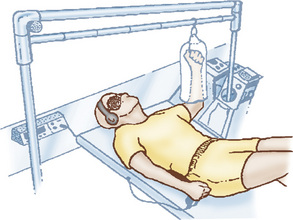
Figure 24-2 Upper extremity proprioceptive testing device.
(From Pollack, R. [2000]: Role of shoulder stabilization relative to restoration of neuromuscular control and joint kinematics. In Lephart, S.M., and Fu, F.H. [eds.]: Proprioception and Neuromuscular Control. Champaign, IL: Human Kinetics.)
Physiologically, the TTDPM test is designed to selectively stimulate the Ruffini or Golgi-type mechanoreceptors in the articular structures being tested. Testing is typically performed for internal and external rotation of the glenohumeral joint in varying positions of elevation in the scapular and coronal planes. Testing in the literature has been done at the midrange and end-range positions of glenohumeral rotation.2,15,36 As stated earlier, TTDPM in the human shoulder was measured by Blaiser et al,34 and passive motion was found to be enhanced (smaller amount of movement before detection) at or near the end range of external rotation versus the midrange of external rotation or internal rotation.
Normative data on 40 healthy college-aged individuals undergoing the TTDPM test were reported by Warner et al37 from both neutral rotational starting positions and 30° of humeral rotation with 90° of glenohumeral joint abduction. They found an average of 1.5° to 2.2° for all testing conditions, with no significant difference measured between the dominant or preferred hand relative to the nondominant extremity.38 Allegrucci et al39 measured shoulder kinesthesia in healthy athletes who performed unilateral upper extremity sports, such as baseball, tennis, or volleyball. The TTDPM test was performed with the shoulder in 90° of abduction and both 0° and 75° of external rotation and compared bilaterally. The results showed that the athletes had greater difficulty detecting passive motion in the dominant extremity than in the nondominant extremity. Consistent with earlier research,34 Allegrucci et al39 measured greater sensitivity to passive movement with the shoulder in 75° of external rotation bilaterally than with the shoulder in a more neutral condition. The findings in this study suggest that athletes in unilaterally dominant upper extremity sports may have a proprioceptive deficit in the dominant arm that may interfere with optimal afferent feedback regarding joint position.39 This finding provides a rationale for proprioceptive upper extremity training in athletes from this population.
Measurement of Joint Position Sense
Joint position sense is the ability of the subject to appreciate where the extremity is oriented in space. Testing procedures to assess joint position sense are called joint angular replication tests. These tests typically place the extremity in a particular position to allow the subject to appreciate the spatial orientation of the extremity. After this period of joint positioning, the subject’s extremity is returned to a starting position. The subject then reapproximates the position initially selected as closely as possible, without any visual, auditory, or tactile cues. Researchers have used both active2,15,36,40,41 and passive41 angular replication tests for assessment of the glenohumeral joint, and various apparatuses have been used to facilitate the accuracy of joint angular replication testing. Voight et al41 used an isokinetic dynamometer with 90° of abduction and elbow flexion and standard isokinetic stabilization to perform active angular joint replication testing via a fatigue paradigm. They also used the passive mode of the isokinetic dynamometer set at 2°/sec to perform passive joint angular replication testing. Various authors2,42,43 have used complex three-dimensional spatial tracking devices and multiple positions of active joint angular replication testing to quantify arm position.
In the most clinically applicable research study on active joint angular reproduction, Davies and Hoffman40 tested subjects in a seated position with an electronic digital inclinometer (EDI).* Reference angles were chosen in several ranges and verified with the EDI; the patient then attempted to replicate the angular position, with the EDI being used to verify the position of the extremity. Angles chosen were greater than 90° and less than 90° of flexion and abduction, external rotation greater than 45° and less than 45°, and internal rotation greater than 45° and less than 45°. Normative data developed by Davies and Hoffman for 100 male subjects without pathologic shoulder conditions showed the average of the seven measurements to be 2.7°.40 This represents the average difference between the seven reference angles and the actual matched angles by the subjects over the seven measurements.
Regardless of the testing methodology, active joint angular position replication tests primarily involve stimulation of both joint and muscle receptors and provide a thorough assessment of the afferent pathways of the human shoulder.2,15
Assessment of Neuromuscular Control of the Shoulder
Several methods have been used by clinicians and researchers to assess neuromuscular control of the shoulder. Widespread use of electromyographic (EMG) studies to measure muscular activity during shoulder rehabilitative exercise,44-47 functional movement patterns such as the throwing motion48 and tennis serve and groundstrokes,49 and abnormal muscular activity patterns during planar motions50-52 and functional activities53 is reported in the scientific and clinical literature. Most of these studies comparing muscular activity expressed the contribution or activity of the muscle in terms of the amount of muscle activity relative to the maximal activity assessed via a maximal isolated manual muscle test (MMT). This is commonly referred to as %MMT or %MVC (maximum voluntary contraction) and allows comparison and expression of the relative activity of human muscle activity during activities of daily living (ADLs) and sport-specific movement patterns.48,49
Muscular Strength Testing
Another important aspect of assessing neuromuscular control is measurement of muscular strength. Methods such as the MMT and the use of handheld dynamometers and isokinetic apparatuses have been used extensively for the documentation of both upper and lower extremity strength. Further discussion is beyond the scope of this chapter; however, the reader is referred to Chapter 25.
Closed Kinetic Chain Upper Extremity Testing
Closed kinetic chain (CKC) upper extremity tests are also used to assess neuromuscular control of the shoulder. Although widespread use of CKC training techniques has been reported in the physical medicine and rehabilitation literature,54-58 currently existing evaluation methods to properly assess CKC function of the upper extremity are limited.
One of the “gold standards” in physical education for gross assessment of upper extremity strength has been the push-up. This test has been used to generate sport-specific normative data in normal populations,56,59 but it is not typically considered appropriate for use in patients with shoulder dysfunction. The positional demands placed on the anterior capsule and the increased joint loading limit the effectiveness of this test in musculoskeletal rehabilitation. Modification of the push-up has been reported, and the modified push-up has been used clinically as an acceptable alternative to assess CKC function in the upper extremities.
Davies developed the CKC upper extremity stability test in an attempt to provide a means of assessing the functional ability of the upper extremity more accurately.56,60,61 The test is initiated in the starting position of a standard push-up for males and modified (off the knees) push-up for females. Two strips of tape are placed parallel to each other, 3 feet apart on the floor. The subject or patient then moves both hands back and forth and touches each line alternatively as many times as possible in 15 seconds. Each touch of the line is counted and tallied to generate the CKC upper extremity stability test score. Normative results have been established, with males averaging 18.5 touches and females averaging 20.5 touches in 15 seconds. The CKC upper extremity stability test has been subjected to a test-retest reliability measure, with an intraclass correlation coefficient of 0.927 being generated, which is indicative of high clinical reliability between sessions with this examination method.61
Effects of aging, instability, and injury on lower extremity proprioception
The effects of age and injury have been correlated with diminished proprioceptive sense.62 Studies63,64 have shown decreased proprioceptive acuity in older adults with testing, and it has been suggested that this decreased capacity for movement sense results in a higher incidence of falling and joint degeneration in this population. However, it has also been found that with regular physical activity, the age-related decline in proprioception can be lessened through dampening of the effect of disuse atrophy on the neuromuscular system.2 In addition to age-related deficits, injuries to the lower extremity joints sustained as a result of repetitive microtrauma or a single traumatic event can create an environment in which degenerative changes occur in the joint along with disruption of the neuromuscular response. The presence of pain and inflammation in a joint produces an inhibitory effect on neuromuscular activation with decreased afferent mechanoreceptor signals.65 Hurley and Newham66 and Sharma and Pai67 demonstrated arthrogenous muscle inhibition in patients with degenerative arthritis. The inability to achieve full voluntary muscle contraction may lead to continued overload on the joints through the loss of dynamic control and attenuation of force.
Loss of capsuloligamentous stability has been shown to cause proprioceptive deficits as a result of inadequate activation of mechanoreceptors leading to delayed muscle reaction latencies. Barrack et al68 found decreased proprioception in a group of ballet dancers and attributed this clinical loss of proprioception to the hyperlaxity found in the ligamentous restraints in this population. It is theorized that without adequate tension in the capsuloligamentous restraints, insufficient stimulation of the mechanoreceptors used for proprioception occurs and results in decreased motor control. A study by Garn and Newton69 also showed that individuals suffering from chronic ankle instability have diminished proprioception with a low threshold for passive plantar flexion. A similar study by Lentell et al70 tested subjects with chronic lateral ankle instability who demonstrated decreased passive movement sense, with the uninvolved ankle being used as the control. Subjects in this study demonstrated no evidence of everter strength contributing to the functional instability. Therefore, the chronic instability was due to loss of mechanoreceptor function from ligamentous laxity and the resultant delayed muscular reflex. Lephart and Fu2 and Nawoczenski et al71 confirmed this decreased muscular stabilization in a study involving subjects with ankle instability. The results of their studies supported this loss of motor control, with a delay in onset latency in the peroneal muscles when subjected to sudden inversion stress.
Effects of Knee Injury on Proprioception
Degenerative arthritis in the knee causes pain, inflammation, and muscular inhibition, which results in decreased functional performance during gait and weight-bearing activities.2 When combined with pain and altered muscle activity, the inadequate ligamentous tension resulting from narrowing of the joint space contributes to the interruption in afferent signals for proprioception and neuromuscular control. The goal of joint replacement surgery is to restore function through resurfacing joints, retensioning soft tissue structures, and ultimately restoring dynamic stability. Research performed by Warren et al72 and Barrett et al73 suggested that joint replacement surgery may actually improve joint position sense, with subjects showing significant improvement in position sense 6 months postoperatively. Furthermore, correlations have been made between improved functional outcomes and gait parameters and proprioceptive scores, thus suggesting a relationship between restoration of proprioception and improved functional outcomes.
The results of studies to date on the selection of joint prostheses and the effects of retaining versus sacrificing the posterior cruciate ligament (PCL) on proprioception have been inconclusive. However, it has been theorized that by restoring joint integrity and retensioning soft tissue structures, retention of the PCL will enhance dynamic joint stability through preservation of the neural reflexive pathway.73-75
Studies in the literature have consistently demonstrated decreases in proprioceptive sense and altered muscle patterns after rupture of the anterior cruciate ligament (ACL).1,2,29 Loss of stability of the ACL causes alterations in muscle activity and reflex patterns, primarily the ACL-hamstring reflex. Measuring the ACL-hamstring reflex in patients with ACL rupture, Beard et al76 showed significant reflex latency delays that were directly correlated with functional instability. Using EMG studies, Limbird et al77 showed variations in muscle activation patterns with increased hamstring activation and concomitant decreased quadriceps activity with joint loading during gait. Andriacchi and Birac78 had similar findings in patients performing normal activities of ambulation, stair climbing, and jogging. With the loss of stability and neural sensory input, many individuals experience functional disability in performance of normal ADLs.
Effects of pathologic shoulder conditions on proprioception and neuromuscular control
In this section the normal afferent neurobiology of the joint and periarticular structures is reviewed, and examples of how proprioception and neuromuscular control are affected in pathologic conditions of the shoulder are provided. Examples of both glenohumeral joint instability and pathologic rotator cuff conditions are presented, as well as dysfunction of the scapulothoracic joint.
Effects of Glenohumeral Joint Instability on Proprioception
Several studies have addressed the influence of glenohumeral joint instability on proprioception. One of the most common clinical maladies seen by clinicians is anterior glenohumeral joint instability. Speer et al79 studied the effects of a simulated Bankart lesion in cadavers. Coupled anterior/posterior translations were assessed in the presence of sequentially applied loads of 50 N in the anterior, posterior, superior, and inferior directions. The effects of a simulated Bankart lesion were small increases (maximum of 3.4 mm) in anterior and inferior translation of the humeral head relative to the glenoid in all positions of elevation and in posterior translation at 90° of elevation only.79 The relevance of this article to the current discussion on proprioception is that Speer et al79 concluded that detachment of the anterior inferior labrum from the glenoid (Bankart lesion) alone does not create large enough increases in humeral head translation to allow anterior glenohumeral joint dislocation. They indicated that permanent stretching or elongation of the inferior glenohumeral ligament may also occur and is necessary to produce full dislocation of the glenohumeral joint. This elongation or permanent stretching of the ligamentous structures may lead to alterations in the intrinsic tensile relationships of the glenohumeral joint capsule and capsular ligaments. The authors concluded that capsular elongation may be responsible for the high incidence of anterior reconstructions that fail to address anterior glenohumeral joint instability and do not fully restore normal capsular tension in the anterior structures.
Blaiser et al34 examined the proprioceptive ability of subjects without known pathologic shoulder conditions and compared them with individuals with clinically determined generalized joint laxity. Individuals with greater glenohumeral joint laxity were found to have less sensitive proprioception than were those with less glenohumeral joint laxity. The authors found enhanced proprioception at or near the end range of external rotation, a position at which the anterior capsular structures have greater internal tension. They concluded that decreased joint angular reposition sense is one characteristic in individuals with increased glenohumeral joint laxity.
Smith and Brunolli35 examined kinesthesia after glenohumeral joint dislocation in 8 subjects and compared their inherent joint position sense with that in 10 normal subjects by using an instrumented modification of a shoulder wheel. Their results indicated a significant decrease in joint awareness in the involved shoulders after shoulder dislocation in comparison to all uninvolved shoulders tested in the study.
Barden et al80 tested subjects with multidirectional instability (MDI) for joint angular replication in multiple positions, including overhead reaching and abduction with external rotation. Subjects with MDI exhibited significantly greater hand position error than did control subjects without instability. This study showed significant proprioceptive deficits in patient with MDI.
Lephart et al36 studied glenohumeral joint proprioception in 90 subjects in three experimental groups. One group consisted of 40 normal college-aged subjects, another group consisted of 30 patients with anterior instability, and the third group included 20 subjects who underwent surgical reconstruction for shoulder instability. No significant difference was found between extremities (dominant versus nondominant) in the normal subjects’ proprioceptive ability; however, subjects with anterior instability had significant differences between the normal and unstable shoulders. Finally, Lephart et al36 found no significant difference in the operated extremity versus the uninjured extremity after reconstructive surgery. This study was performed at least 6 months after subjects underwent open or arthroscopic repair for chronic, recurrent shoulder anterior instability. The authors concluded that these results provide evidence, consistent with the studies mentioned earlier, for partial deafferentation leading to proprioceptive deficits when the capsuloligamentous structures are damaged. Reconstructive surgery in this experiment appeared to restore normal joint proprioception 6 months or more after the surgical procedure.
Safran et al81 used a testing device to study 21 collegiate baseball pitchers to determine whether bilateral differences in joint angular replication (JAR) and kinesthesia were present between extremities. They found that JAR was more accurate in the nondominant extremity when moving from a position of 75° of external rotation into internal rotation. Measurements were taken in 90° of abduction. No difference in proprioceptive ability was observed when moving from 75° of external rotation to end range of motion (ROM) between the extremities. Six collegiate pitchers with reports of shoulder pain were tested by Safran et al81 and found to have a kinesthetic deficit in the injured dominant shoulder versus the nondominant shoulder when moving from neutral rotation into internal rotation. These results show JAR to be bilaterally symmetric from 75° of external rotation to end ROM between extremities in healthy skilled baseball pitchers despite increases in laxity and training effects. Additionally, despite a small sample size, Safran et al81 did show very importantly that pitchers with a recent report of injury involving the shoulder do have kinesthetic deficits in the injured arm that may affect further performance.
The finding of reduced proprioception in unstable shoulders has prompted researchers to examine the effect of surgical stabilization procedures on restoring proprioception following surgery. Rokito et al82 studied the effects of two open surgical procedures for recurrent unidirectional anterior instability. Thirty subjects underwent an open inferior capsular shift procedure involving an approach that detached the subscapularis from the lesser tuberosity to gain exposure. Twenty-five underwent anterior capsulolabral reconstruction with a transverse splitting approach to the subscapularis for exposure. At 6 months postoperatively patients underwent proprioceptive testing, and the group with transverse splitting of the subscapularis had no deficits in proprioception and mean strength with respect to the contralateral uninvolved extremity. However, the group that underwent open capsular shift with subscapularis detachment had significant deficits in proprioception and mean strength that did not return to full functional values until 1 year postoperatively. This study shows that deficits in proprioception and strength following an open approach with detachment of the subscapularis require up to 1 year for return to the same functional level as the contralateral baseline extremity.
Effects of Glenohumeral Joint Instability on Neuromuscular Control
Lephart and Fu2 defined neuromuscular control as the unconscious efferent response to an afferent signal concerning dynamic joint stability. Several studies highlighting changes in neuromuscular control in subjects with glenohumeral joint instability have been published. Glousman et al,53 using an indwelling EMG electrode, studied the muscular activity patterns of normal healthy baseball pitchers and compared them with throwers with anterior glenohumeral joint instability. The results of the study showed marked increases in muscular activation of the supraspinatus and biceps muscle, as well as selective increases in the infraspinatus muscle during the early cocking and follow-through phases.53 Also of interest was the finding of decreased muscular activation of the pectoralis major, latissimus dorsi, subscapularis, and serratus anterior muscles in the throwing athletes with anterior glenohumeral joint instability. This study showed neuromuscular compensations in the group with glenohumeral joint instability, as evidenced by increased activation of the primary dynamic stabilizers. Inhibition of the serratus anterior in the group with anterior instability may decrease scapular stability and further jeopardize joint congruity through improper scapulothoracic muscle sequencing.
McMahon and et al83 tested normal shoulders and those with anterior instability and monitored them via indwelling EMG muscular activation patterns. Planar motions of flexion, abduction, and scapular-plane elevation (scaption) were studied in 30° increments. Significant decreases in serratus anterior muscle activity were measured in all three planar motions in the group of subjects with anterior glenohumeral joint instability. None of the other muscles—rotator cuff, deltoid, or scapular—showed a significant difference in testing during standard planar movement patterns. This study clearly shows the importance of the scapulothoracic musculature and dynamic stabilization during both aggressive overhead and common ADL-type movement patterns.
Finally, Kronberg et al50 used intramuscular electrodes to compare shoulder muscle activity in patients with generalized joint laxity and normal control subjects. Increased subscapularis muscular activity was measured during internal rotation in the subjects with increased glenohumeral joint laxity, as well as increased middle and anterior deltoid activity during abduction and flexion. These studies clearly show the increased demand required by the dynamic stabilizers in subjects with joint laxity and glenohumeral joint instability. Application of the resistive exercise progressions and use of the kinetic chain exercise series listed later in this chapter have these research-based rationales and can directly enhance neuromuscular control of the shoulder complex.
Effects of Rotator Cuff Dysfunction on Neuromuscular Control in the Shoulder
Research similar to that discussed in the preceding section in which muscular activation patterns in patients with rotator cuff impingement were measured has been published. Ludewig and Cook51 studied 52 male construction workers, 26 of whom had unilateral shoulder impingement and 26 had no symptoms of impingement or other pathologic shoulder condition. Similar to subjects in the previously discussed research studies on glenohumeral joint instability, those with unilateral impingement demonstrated a decrease in serratus anterior muscle activation during active elevation of the arm in comparison to normal, uninjured subjects.51 Additionally, increases in upper and lower trapezius muscle activity were found in the subjects with unilateral impingement. This altered neuromuscular control mechanism also resulted in abnormal scapular posturing consisting of decreased upward rotation with elevation, increased anterior tipping, and increased medial rotation. These scapular modifications are thought to be contributing factors to rotator cuff impingement and demonstrate the importance of optimal and coordinated muscular control of the scapulothoracic and glenohumeral joints.
Effects of fatigue on lower extremity proprioception
Muscle fatigue reduces the force-generating capacity of the neuromuscular system, which essentially leads to increased laxity in the knee joint.84 Skinner et al28 found an increase in laxity of the ACL measured with a KT1000* arthrometer after a fatigue protocol. Similarly, Weisman et al85 found increased laxity in the medial collateral ligament in athletes at a university after participation in various sporting activities. Furthermore, studies in the literature2,28,86,87 have shown a decrease in the sensitivity of muscle receptors under fatigue conditions. The consequences of decreased proprioceptive sense from fatigue can be deleterious because of the possibility of sustaining injuries under these conditions when higher-level activities are performed. Skinner et al28 studied the effects of fatigue on joint position sense and knee angle reproduction in a group of healthy, highly trained male recruits in the Special Forces division of the Navy. Subjects underwent an interval running program followed by isokinetic measurement of knee extension and flexion. Fatigue was determined by the percent decrement in work output measured from pretraining to posttraining conditions on an isokinetic device. The authors concluded that after fatigue ensues, values on angular replication tests are significantly decreased, but no significant changes were noted in the threshold of movement sense. They determined that loss of muscle receptor efficiency as a result of fatigue played a key role in angular replication errors. The authors concluded that the dual role of afferent input by the receptors in the contractile and noncontractile elements of the knee is important for proprioceptive sense.
Lattanzio et al25 conducted a study involving healthy male and female subjects performing three different cycling protocols (ramp, continuous, and interval training) at a percentage of their ![]() o2max. In this study, methods for determining the threshold for detection of movement were similar, but angular replications were performed with subjects in the standing weight-bearing position instead of the seated open kinetic chain (OKC) protocol used in the study of Skinner et al. The results of the study of Lattanzio et al25 were similar to those of Skinner et al, with statistically significant decrements in joint replication in male subjects after the three different fatigue protocols. Female subjects similarly showed significant differences in joint replication after the continuous and interval programs, but not with the ramp protocol for joint angular replication. The conclusions drawn by the authors of this study were that anatomic gender differences possibly account for the variation in proprioception in response to fatigue.
o2max. In this study, methods for determining the threshold for detection of movement were similar, but angular replications were performed with subjects in the standing weight-bearing position instead of the seated open kinetic chain (OKC) protocol used in the study of Skinner et al. The results of the study of Lattanzio et al25 were similar to those of Skinner et al, with statistically significant decrements in joint replication in male subjects after the three different fatigue protocols. Female subjects similarly showed significant differences in joint replication after the continuous and interval programs, but not with the ramp protocol for joint angular replication. The conclusions drawn by the authors of this study were that anatomic gender differences possibly account for the variation in proprioception in response to fatigue.
Finally, Barrack et al30 and Barrett et al73 studied the effects of total knee replacement on knee joint proprioception. This research paradigm is of particular interest because insertion of a total knee joint prosthesis results in removal of most joint receptors in the human knee. Both groups of investigators found no significant loss of proprioception in the extremity that underwent total knee replacement in comparison to the contralateral extremity 6 months postoperatively. These groups of authors both concluded that their research again points to the important role that muscle-based mechanoreceptors play in knee joint proprioception.
Effects of muscular fatigue on upper extremity proprioception and neuromuscular control
The role of specific afferent receptors in the human body has been examined with different methods to better understand the role of joint and muscular afferents. Provins62 reported a decrease in the ability to detect passive motion of the finger when digital nerves containing both joint and cutaneous afferents were blocked by local anesthesia. He concluded that both types of afferent feedback may be equally important when joint proprioception is analyzed.
Zuckerman et al88 injected lidocaine into the subacromial space and glenohumeral joint to assess proprioception in young and old male subjects. They found no adverse effects from the injection of lidocaine in either location and proposed that compensatory extracapsular feedback ensured intact proprioception after injection. No differences in joint position sense and TTDPM testing were noted between the dominant and nondominant extremity; however, a decline in proprioception with age was measured in the young (20 to 30 years of age) and older (50 to 70 years of age) subjects.
Several studies have been performed on the human shoulder to investigate the effect of muscular fatigue on various indices of joint proprioception and neuromuscular control. Carpenter et al89 tested subjects using a TTDPM test with the shoulder in 90° of abduction and 90° of external rotation. After an isokinetic fatigue protocol, subjects’ detection of passive motion was marred or decreased by 171% for internal rotation and 179% for external rotation. In preexercise testing, Carpenter et al found increased sensitivity when moving into external rotation versus internal rotation but no difference between the dominant and nondominant extremities.89 These authors concluded that the effect of muscular fatigue on joint proprioception may play a role in injury and decrease athletic performance.
Voight et al41 tested subjects with an active and passive joint angular replication protocol after isokinetically induced muscular fatigue of the glenohumeral joint internal and external rotators. No significant difference in shoulder joint angular replication was found between the dominant and nondominant extremities. Significant decreases in accuracy were noted after muscular fatigue in both the active and passive joint angular replication tests. Pederson et al90 tested the ability of healthy subjects to discriminate movement velocity of the glenohumeral joint in the transverse plane. The results of their study showed that subjects had a decrement in discrimination of movement velocity after a hard isokinetic horizontal flexion/extension exercise fatigue protocol versus a light exercise condition.
Myers et al91 used an active angular replication test and neuromuscular control test to examine the effects of muscle fatigue in normal shoulders. A concentric isokinetic internal and external rotation fatigue protocol was used. Fatigue of the internal and external rotators of the shoulder decreased subjects’ accuracy in detecting both midrange and end-range absolute angular error but did not have a negative effect on neuromuscular control in a bilaterally assessed unilateral CKC stability-type test measuring postural sway velocity.
Additional research by Myers et al92 has demonstrated that patients with anterior glenohumeral instability have alterations in muscle activation. Therefore, clinicians can implement therapeutic exercises that address the suppressed muscles as the scientific foundation of a rehabilitation program. Myers et al93,94 found that capsuloligamentous injury to the shoulder decreases proprioceptive input to the CNS and thereby results in decreased neuromuscular control. Consequently, clinicians need to address the mechanical instability but also implement functional rehabilitation interventions to return an athlete to competition. Tripp et al95 demonstrated that functional fatigue affects the acuity of the entire upper extremity.
Lin et al96 investigated the effects of scapular taping on shoulder proprioception and EMG activity in several muscles of the shoulder complex. The magnitude of proprioceptive feedback was significantly lower in the taping conditions. The results suggest that scapular tape affects the activity of the shoulder muscles and ultimately that these effects are related to the proprioceptive feedback provided by the tape. Further research will continue to be needed to better understand and demonstrate the efficacy of shoulder and scapular taping because it is a very popular technique with many anecdotal reports demonstrating effectiveness; however, high-level research support for this technique is limited at this time.
The consistent finding in these studies of a decrement in proprioception after muscular fatigue has led researchers to emphasize the importance of the muscle-based receptors. The use of active joint angular positioning tests has been reported to stimulate both joint and muscle mechanoreceptors and is considered to be a more functional assessment of the afferent pathways.2,15,91 The exact mechanism by which muscle-based proprioception is affected is not entirely clear or known. Muscle fatigue is thought to desensitize the muscle spindle threshold and thereby lead to decrements in both joint position sense and neuromuscular control. Djupsjobacka et al97-99 reported alterations in muscle spindle output in the presence of lactic acid, potassium chloride, arachidonic acid, and bradykinin. Intramuscular concentrations of these substances are altered during muscular exertion and fatigue. This consistent relationship has provided further rationale and support for improvement in muscular endurance of the dynamic stabilizers of the glenohumeral joint. This topic is covered in detail in the application section of this chapter.
Effects of training on proprioception in the lower extremity
Some studies in the literature have investigated the notion of injury prevention and improvement in neuromuscular stabilization through proprioceptive training. A review of research in this section will provide the reader with important references that support the use of proprioceptive training of the lower part of the body for both injury prevention and rehabilitation.
In a prospective study by Cerulli et al,100 600 semiprofessional and amateur soccer players were monitored for three seasons to determine the frequency of ACL injury in players who underwent a progressive proprioceptive training program and in a control group who performed only traditional strengthening exercises. The results showed significant differences between the experimental and control groups, with the proprioception training group sustaining fewer lesions of the ACL than the control group who performed traditional strengthening exercises.
Osborne et al101 studied the effects of ankle disk training in eight individuals who sustained an inversion ankle sprain within the preceding years and who had not received any formal rehabilitation. The subjects performed 15 minutes of daily training on a disk with the involved leg in an 8-week training program. After completion of the 8-week training program, the subjects were tested for onset latencies with surface EMG electrodes on the muscles of the ankle influencing stability to measure the motor response to a simulated inversion sprain on a platform. The results showed significant improvements in anterior tibialis latency times in both the trained and untrained control ankles.
A similar study by Eils and Dieter102 showed significant improvements in muscle reaction times and patterns of muscle coactivation in 30 subjects with chronic ankle sprains. The subjects performed a multistation proprioceptive exercise program that included 12 stations with various devices once weekly for 6 weeks. The frequency of once per week and the types of exercises were chosen for their ability to be implemented easily into a rehabilitation program. The exercises included a Biodex balance system,* inversion boards, minitrampoline, and ankle disk. The subjects performing the exercises showed significant improvement over the control group in position sense and reported subjective improvements in functional stability.
Friden et al31 conducted a study in subjects with ACL deficiencies who performed traditional lower extremity strengthening exercises and in subjects who performed traditional rehabilitation along with perturbation training. The perturbation program consisted of progressive exercises using rocker boards and roller boards, with advancement to the next phase after successful completion of the task without evidence of instability or pain. After the training program, the perturbation group was found to have significantly greater success with subjective reports of stability during completion of higher-level activities than did the traditional training group. Beard et al76 conducted a similar study but used hamstring reflex latencies and the Lyshom rating scale for measuring functional outcomes. They concluded that the group that underwent perturbation training had improved functional outcomes in knee stability while performing ADLs. Additionally, numerous studies have evaluated the effectiveness of neuromuscular training programs, including perturbation training, in patients with ACL injuries.52,103-107 Numerous studies have also evaluated the effectiveness of neuromuscular training programs, including perturbation training, in patients with chronic/functional ankle instability.108-111
One very important aspect inherent in most lower extremity proprioceptive training programs is inclusion of the entire lower extremity kinetic chain in the exercise. Most proprioceptive exercises in the literature described herein use multiple joint-training positions and CKC positioning environments that allow the entire lower extremity kinetic chain to be included. Research112-114 has emphasized the importance of examining the entire kinematic chain from the trunk and hip musculature throughout the lower extremity for postural control mechanisms in rehabilitation and prevention of injury. Furthermore, using absolute angular error measurements, Miura et al115 found that local and general fatigue affects knee proprioception, decreases muscular power, and has effects on different mechanisms in the proprioceptive pathway. Consequently, to prevent injury caused by a fatigue-induced decline in proprioception, local muscle training by itself is not enough, but neuromuscular training, including central programming, is essential for the entire lower extremity kinetic chain.
Neuromuscular training for rehabilitation and prevention of sports injuries
Zech et al116 performed a systematic review of the use of neuromuscular training for rehabilitation of sports injuries. Fifteen studies met the inclusion criteria and demonstrated the effectiveness of neuromuscular training in increasing functionality and decreasing the incidence of recurrence after ankle and knee injuries. However, no studies demonstrated the effectiveness of neuromuscular training in rehabilitating sports injuries in the shoulder, only for lower body injuries. Though used inherently and recommended for shoulder rehabilitation, this systematic review showed much less scientific support for the use of neuromuscular control exercises in the upper extremity. Hübscher et al117 performed a systematic review of the use of neuromuscular training for rehabilitation of sports injuries. Thirty-two articles were identified, but only seven methodically well conducted studies were included in the review. Multiintervention training was effective in reducing the risk for lower limb injuries. Balance training alone decreased the incidence of ankle sprains in athletes. Interestingly, exercise interventions were more effective in athletes with a history of sports injuries than in those without. This probably relates to the concept of inadequate rehabilitation following the initial sports injury. Additional research on the use of balance training to improve neuromuscular control was performed by Zech et al.118 They completed a systematic review of balance training for neuromuscular control and enhancement of performance. Twenty RCTs met the inclusion criteria. Balance training was effective in improving postural sway and functional balance, and larger effect sizes were demonstrated for training programs of longer duration. It is controversial whether balance training was effective in improving jumping performance, agility, and neuromuscular control.
Clinical application: techniques to improve lower extremity proprioception and neuromuscular control
Lower extremity injuries occur often in competitive and recreational sports. These injuries are sometimes caused by physical contact with another individual, but they usually result from a noncontact injury in which the external forces in the environment exceed the internal forces of the body.100 Some of the more common injuries involve damage to the ligamentous and cartilaginous components in the knee and ankle. Injuries that do not involve another person occur when the player or individual attempts to suddenly change the rate of speed or course of direction or when an obstacle in the external environment causes overload on the static joint restraints. The questions that have received recent attention in the literature are the degree to which these injuries can be prevented and, once an individual is injured, the ways in which recurrent injuries to an existing compromised system can be prevented through dynamic neuromuscular stabilization.119
For a patient with ACL deficiency, neuromuscular training is achieved through coordinated muscle activation in response to controlled perturbation forces imparted on the joint. One strategy for dynamic stabilization is cocontraction of opposing muscle groups to essentially stabilize the knee in a rigid posture. This strategy may be successful for simple tasks, but with higher-level activities such as sports, stabilization is achieved through selective motor recruitment that is dependent on the task. A force feedback mechanism in which stability is achieved through varied patterns of muscle recruitment, depending on the situational needs of the task, has been discussed.1,2 This theory acknowledges that different patterns of movement require varied muscular stabilization, depending on the direction, speed, and amount of force occurring at the joint.
At the University of Delaware, Snyder-Mackler et al1,120 designed a rehabilitation program based on the premise of achieving dynamic muscular stabilization during normal and higher-level skills through neuromuscular perturbation training. They use the term copers for individuals who successfully perform varied high-level activities without experiencing functional instability. In copers, the muscular strategies used for joint stability allow normal joint movement, whereas deleterious compressive and shear forces at the joint are minimized. The term given to individuals who are unsuccessful in maintaining joint stability during lower extremity weight-bearing tasks is noncopers. In the group of noncopers, a cocontraction stiffening strategy is used with all tasks, which results in inefficient movement strategies and functional instability. Furthermore, with this inadequate coping mechanism, progressive deterioration of joint surfaces and capsuloligamentous restraints occurs as a result of excessive shear and compressive forces at the joint.1,2
The faculty at the University of Delaware designed a program using the guidelines of Fitzgerald et al to implement a neuromuscular training program for patients with ACL deficiency in an attempt to restore functional stability during higher-level sporting activities. In selection of individuals for the program, certain criteria had to be met to ensure a successful outcome of the training.
The program is designed to identify individuals who would be successful rehabilitation candidates through a screening process. The criteria include isolated injury to the ACL, infrequent episodes of instability (< 1), a passing score on functional hop tests, and a passing percentage on two subjective rating scales for functional knee impairment. Before a stabilization program is initiated, the early focus of rehabilitation is on decreasing joint effusion, restoring ROM, and increasing quadriceps and hamstring strength to allow stabilization through muscle recruitment.1 When these goals have been met, an advanced neuromuscular training program can be initiated. The program is progressive in nature and designed for specificity of sport or activity.
The program consists of 10 treatment sessions at a frequency of two to three times per week and is progressive in nature with three phases of implementation (early phase, sessions 1 to 4; middle phase, sessions 5 to 7; and late phase, sessions 8 to 10). Progression of the program is based on the symptomatic patient response (i.e., increased effusion or pain), which is used as a guideline, and the ability of the patient to perform successful motor strategies to counteract the perturbation force, which includes no episodes of falling or instability. All three phases include the use of rocker boards, roller boards, and platforms with an introduction to sport-specific agility drills in the middle to later stages.
In the early phase of perturbation training, patients are subjected to perturbation forces on all three devices in slow predictable directions with the use of verbal cues as necessary for the onset and direction of the forces. Initially, the direction of the applied forces is in the anterior/posterior and medial/lateral directions with progression to diagonal and rotational planes. The clinical implication of this phase thus involves the application of progressive variable perturbation forces in multiple directions (sagittal, transverse, and frontal planes) in a controlled manner to retrain the nervous system in a number of applications or situational needs while avoiding the use of rigid cocontraction strategies.2
The middle phase of training continues with perturbation training and requires successful adaptation of strategies in the early phase. Variations in the parameters of training, including predictability, speed, amplitude, intensity, and direction of force, are advanced, along with the implementation of light sport-specific drills while the individual is wearing a functional knee brace. In the last phase of treatment, sport-specific movements are emphasized with the use of agility drills. Initially, training begins in straight planes and then moves to variable-direction drills such as cutting and changing direction on command. The drills are initially performed at 50% and progress to 100% in the later stages. Examples of some of the agility drills are side shuffles, shuttle running, and cutting maneuvers at 45° and 90° angles. With no evidence of instability, patients then perform sport-specific activities while undergoing perturbations on roller boards and platforms to simulate the competitive demands of the sporting environment. Before returning to full athletic competition, athletes are required to pass a posttreatment ACL screening involving measures similar to those used during prescreening for acceptance into the program.2
Guidelines for implementing lower extremity proprioceptive training
Regardless of whether surgery or conservative care is chosen to restore stability and function to a degenerative or unstable joint, rehabilitation is crucial for reestablishing neuromuscular control or dynamic stability in a compromised joint. The loss of neuromuscular control results from damage to the mechanoreceptors within the capsuloligamentous structures of the joint and from interruption of the afferent sensory pathways that play a crucial role in producing smooth, coordinated movement.2
Several considerations are important when a rehabilitation program is designed to restore proprioception and dynamic stability. In selecting exercises for training, a focus on restoring function to the individual should remain at the forefront. Exercises chosen should then focus on an individual’s deficits in strength, ROM, and balance and, most importantly, on the individual’s ability to meet the demands of stability while performing daily or sporting activities.
Traditionally, a combination of OKC and CKC exercises have been used in rehabilitation. OKC exercises have been defined as movements in which the distal segment is free to move in space, and CKC movements occur when the distal segment is fixed or meets considerable resistance.121 Emphasis on the use of CKC exercises has predominated because they are thought to more closely resemble the functional demands placed on the lower extremity during a variety of activities. Another advantage of CKC exercises is the simultaneous movement of multiple joints, which requires cocontraction of opposing muscle groups to control joint movement. CKC exercises can also reduce shear forces across joint surfaces as a result of the stability of joint-through-joint compression forces and cocontraction of opposing muscle groups. The advantage of OKC exercises is their ability to isolate targeted muscle groups for strengthening.
In a study by Snyder-Mackler et al,122 isolated OKC quadriceps strengthening was found to be superior to CKC exercises in improving quadriceps function in patients after ACL surgery because the involvement of other muscle groups in performing the CKC exercises did not isolate the quadriceps as effectively. In fact, most functional activities, such as ambulation, use a combination of both OKC and CKC muscle activation patterns, and therefore both should be incorporated in the design of a successful program.
In the early stages of rehabilitation, the development of an exercise program should identify deficits in ROM, strength, and joint effusion, and progression should not exceed the rate of natural healing or limitations in the involved structures. Any number of exercises will elicit proprioception training based on the fact that deformation of the joint mechanoreceptors occurs with active, active assisted, and passive movements and provides sensory input to improve neural mechanisms.2 Performing ROM exercises on an immobilized joint and weight shifting early after an ankle sprain or surgery on the ACL are examples of early forms of proprioception training.
When sufficient healing has taken place in the subacute stages of recovery, initiation of resistance exercises for building muscular strength and endurance will enable sufficient muscle recruitment patterns for the dynamic stabilization needed for advanced forms of training. Proprioceptive training in this stage may involve two-legged stance exercises on an unstable surface such as the Biodex stability balance system (Fig. 24-3), which can be advanced to a functional squatting movement pattern. The exercise progression can include single-leg stance (Fig. 24-4) and single-leg stance with partial squatting on the machine with the benefit of a visual cursor to assess weight distribution so that compensatory patterns can be avoided.
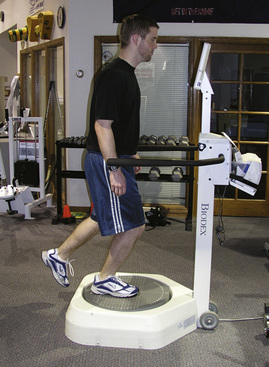
Figure 24-4 Single-leg balance on Biodex balance system. The level of difficulty is progressed by decreasing the stability of the platform or removing visual cues by having the patient close his eyes.
Other exercises that are beneficial for proprioception involve the use of rocker boards for directional perturbations to activate selective muscle recruitment patterns in a variety of planes of movement. These training techniques can be advanced to sport-specific activities such as tossing a ball against a trampoline while manual perturbations are applied to the board (Fig. 24-5). Movement patterns such as a straight plane or multidirectional lunge are also useful for selective motor recruitment in functional or sport-related activities (Figs. 24-6 and 24-7). These patterns can be performed on balance pads or exercise mats to enhance motor control through maintenance of balance while performing initially slow and then more rapid movements beyond the base of support (Fig. 24-8).

Figure 24-5 Chest pass with a weighted ball incorporates plyometric exercises with proprioceptive exercise, with difficulty being increased by manual perturbations of the rocker board while the patient throws and catches the ball.

Figure 24-6 Combination of proximal pelvic control with lower extremity proprioception while the patient performs lunges on Thera-Band balance pads.

Figure 24-7 A lunge being performed with rotation of the torso while a weighted ball is held outside the base of support for the integration of upper and lower extremity movement patterns.
Figure 24-8 Single-leg balance on Thera-Band balance pads while opposite extremity movements are resisted beyond the base of support in functional planes.
The goal of proprioceptive training is to reestablish stability or dynamic neuromuscular control and should emphasize return to functional or sport-specific activity. In the later stages of training, exercise should focus on restoring and ideally optimizing the adaptive neuromuscular response to situational needs. If stabilization training has yielded the appropriate muscular response toward the final stages of rehabilitation, more advanced exercises incorporating sport-specific drills should be performed. Exercises on a Fitter board* or slide board can be performed to challenge the patient with higher-velocity movements while performing sport- specific drills involving full body movement patterns on a yielding surface (Figs. 24-9 and 24-10). Agility drills, including progressively quicker changes in direction while pivoting on the involved extremity, should be incorporated in the final stages of rehabilitation for athletes who perform such maneuvers in the competitive arena.
Clinical application: techniques to improve proprioception and neuromuscular control of the upper extremity with specific reference to the shoulder
Application of the basic science information on proprioception and neuromuscular control of the shoulder to clinical practice allows clinicians to most appropriately provide stability to the glenohumeral joint and optimize shoulder girdle arthrokinematics. Several areas are covered in this section, including the use of CKC and joint approximation exercises, joint oscillation exercises, postoperative interventions, and techniques to improve muscular endurance of the rotator cuff and scapular musculature.
Closed Kinetic Chain (Joint Approximation) Exercises
Exercises that produce approximation of the glenohumeral joint and are characterized by a fixed distal aspect of the extremity are typically referred to as joint approximation or CKC upper extremity exercises. The approximation of the joint surfaces and the multiple joint loading inherent in CKC exercises are reported to increase mechanoreceptor stimulation2,123 and produce muscular cocontraction. The presence of muscular cocontraction around the human shoulder is particularly beneficial because of the important role that the musculature surrounding the scapulothoracic joint plays in stabilizing and controlling movement of the shoulder.124,125
Significantly less EMG research has been published on upper extremity CKC exercise than on upper extremity OKC exercise. Moesley et al46 published a comprehensive analysis of the scapular muscles during traditional rehabilitation exercises. Two CKC upper extremity exercises were included in their analysis. These exercises were the push-up with a plus and the press-up. The push-up with a plus includes maximal protraction of the scapula during the end of the ascent phase of a modified push-up and produces very high levels of serratus anterior muscle activity. The press-up exercise did not elicit high levels of muscular activity in the trapezius or serratus but instead produced high activation levels in the pectoralis minor. Decker et al126 confirmed the importance of the plus position for serratus anterior activation and concluded that exercises emphasizing scapular upward rotation and accentuated protraction produce the highest levels of muscular activity in the serratus anterior.
Kibler et al125 published EMG research on a series of very low level CKC exercises for the upper extremity, including weight-bearing upper extremity weight shifts and exercises on the rocker board or biomechanical ankle platform system with the upper extremity. Muscle activation levels during these exercises were very low in the rotator cuff, deltoid, and scapular muscles; however, low levels of activity were present in virtually all these muscles during these activities. This indicates that high degrees of coactivation and cocontraction are inherent in this type of exercise.
Research has demonstrated the important role that joint compression plays in glenohumeral joint CKC exercise. Warner et al37 studied the effects of applying a 5-, 25-, and 50-lb compressive force to cadaveric shoulder specimens. These amounts of compression resulted in decreases in anterior humeral head translation in neutral elevation from 11 to 2 mm with 5 and 25 lb of compressive force and from 21.5 to 1.4 mm at 45° of abduction, respectively. This study shows the potential benefit of a compressive load in the provision of glenohumeral joint stability and points out the important application that CKC exercises may have in enhancement of neuromuscular control in patients with glenohumeral joint instability.
Application of CKC exercises clinically is facilitated by a thorough review of glenohumeral joint anatomy. It is imperative that the clinician realize the osseous relationship of the glenohumeral joint. The human glenoid is oriented slightly inferiorly with the arm held at the side and tilted anteriorly 30° from the coronal plane of the body.127,128 This anterior version of the scapula is aligned with 30° of retrotorsion of the humeral head, and optimal bony congruity occurs with the arm placed in the scapular plane.128 Understanding these important relationships will guide the clinician in shoulder positioning and ROM selection during joint approximation exercises with the arm placed in the scapular plane.128
Research on CKC upper extremity training is limited. Lephart et al86 used five neuromuscular control exercises that emphasized joint positioning, joint approximation and compression, and muscular cocontraction in one experimental group, in addition to traditional OKC shoulder rehabilitation exercises in patients with glenohumeral joint instability. They found significant improvements in kinesthetic ability, scapular slide testing, and isokinetically documented protraction and retraction strength in the group that performed these neuromuscular control exercises during rehabilitation.
Application of these concepts in patients with rotator cuff dysfunction and glenohumeral joint instability is pictured in Figures 24-11 to 24-15. Guidelines for the time-based sets of exercise depend on the patient, with the ability of the patient to maintain the desired scapulothoracic stabilization being a governing factor. Careful monitoring of the medial and inferior borders of the scapula is important to ensure proper neuromuscular control and avoid the development of undesired motor patterning.124 Sets of exercises lasting up to 30 or 45 seconds are desired in the later stages of rehabilitation.

Figure 24-11 Closed chain wall scapular-plane rhythmic stabilization. The patient’s arm is placed on a medicine ball or small exercise ball in varying degrees of abduction in the scapular plane. The clinician performs rhythmic stabilization with the patient remaining as stable as possible over the ball; varying the position of the hand contacts by progressing further toward the patient’s hand increases the intensity of the exercise.
Figure 24-12 Quadruped rhythmic stabilization exercise progression with instruction to maintain scapular protraction or the “plus” position during repeated multidirectional challenges.
Figure 24-13 Tripod rhythmic stabilization technique with the involved extremity in the closed chain position and maintenance of the “plus” position to increase activation of the serratus anterior muscle. The clinician alternately provides multidirectional challenges to the non–weight-bearing limb as the patient attempts to isometrically hold the pictured position.
Joint Oscillation Exercises
Rehabilitative exercises involving joint oscillation have increased in popularity in recent years. Appliances such as the Bodyblade,* BOING (body oscillation integrates neuromuscular gain),† and resistance bar‡ have facilitated the use of joint oscillation exercises. Rapid oscillation of these devices coupled with external loads such as light weights, manual resistance, and TheraTubing‡ can provide additional emphasis on particular muscle groups during these exercises. Figures 24-16 to 24-19 show exercises using oscillatory devices or manual contact that require the rotator cuff and scapular musculature to respond to external cues induced via the oscillation and stretch imparted during the exercise. The ability of these time-based exercises to promote local muscular endurance is increased by manipulation of set duration and rest cycles.129 Progression to the external rotation oscillation exercise in Figure 24-17 by using the 90° abducted scapular-plane position is followed as rehabilitation progresses to more closely approximate the glenohumeral and scapulothoracic positions inherent in overhead sport-specific movement patterns.130Figure 24-20 shows the push-up with a plus exercise. Care is taken with this exercise to protect the shoulder complex by descending only approximately one half the distance of a standard push-up and then maximally protracting the scapula on the ascent phase to increase activity of the serratus anterior muscle.46
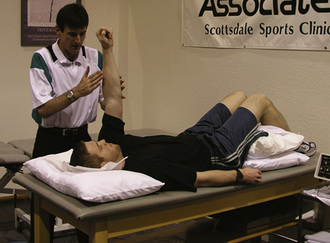
Figure 24-16 Supine modified rhythmic stabilization technique performed in 90° of shoulder flexion with scapular protraction.

Figure 24-18 Biped closed kinetic chain exercise using the Bodyblade to provide joint oscillation in the non–weight-bearing upper extremity and a medicine ball to decrease surface stability of the closed chain upper extremity. Scapular position can be altered in this exercise, depending on the intended goal of muscular activation.

Figure 24-19 “Statue of Liberty” external rotation oscillation exercise using Thera-band elastic resistance and a resistance bar for oscillation. A scapular-plane position in 90° of elevation is used while the contralateral extremity provides support to decrease the role of the deltoid in actively holding the exercising extremity in the 90° position.

Figure 24-20 Push-up with a plus and maximal protraction of the scapula to elicit higher levels of serratus anterior activity. A, Starting position. B, Ending position of movement.
Holt et al131 investigated four different positions of exercise with the Bodyblade and its effect on infraspinatus muscle activity. These four positions included (1) standing with the glenohumeral joint in a neutral abduction/adduction position for internal and external rotation, (2) side-lying position with the glenohumeral joint in neutral abduction/adduction for internal and external rotation (see Fig. 24-17), (3) 90° of glenohumeral joint scapular-plane elevation with stabilization, and (4) 90° of scapular-plane elevation with the unsupported arm. The results of a repeated-measures analysis of variance showed that the side-lying internal/external rotation oscillatory pattern elicited the highest levels of infraspinatus muscle activation.131 Further research such as this is needed to guide clinicians in the use of oscillatory-type exercise.
Vibration Training on Performance
One of the fastest growing areas focusing on proprioceptive and neuromuscular control deals with the effects of vibration training on performance. Marin and Rhea132 performed a metaanalysis of the effects of vibration training on muscle strength. Thirty-one studies met the inclusion criteria. Many factors influence the training responses and effect sizes demonstrated by the studies. These studies show that gender, training status, exercise protocol, and type of vibration platform all influence the outcomes. It appears that vertical platforms produce chronic adaptations whereas oscillating platforms have a more profound effect on acute responses to the exercises. The results of the vibration exercise can be used by exercise professionals to enhance muscular strength based on the aforementioned criteria. Marin and Rhea132 also completed a metaanalysis of the effects of vibration training on muscle power. Vertical platforms were more effective than oscillating platforms in producing a larger treatment effect for chronic adaptations. However, age is a moderator of the response to vibration exercise for power. The results of the vibration exercise can be used by exercise professionals to enhance muscular power in selected subjects and specific protocols.
Muscular Endurance Exercise
Exercises to increase endurance and fatigue resistance of the rotator cuff and scapular musculature would have a direct effect on improving performance and enhancing proprioception and neuromuscular control. All the exercises described in this chapter can be used to promote local muscular endurance by increasing the work duration and decreasing the rest periods in the exercise format. Exercises using sets of 15 to 20 repetitions and 15 to 20 repetition maximum loading schemes are geared toward improving local muscular endurance.129 Current practice in orthopedic and sports physical therapy usually includes exercises with this type of prescription or recommendation.54,57 The use and integration of joint oscillation, joint approximation or closed chain, plyometric, and isotonic and isokinetic training of targeted muscles or muscle groups have clear benefits and research-oriented rationales as outlined in this chapter and other sources.15,54,57,60,129
One additional study performed by Ellenbecker and Roetert121 specifically evaluated the relative muscular fatigue of the rotator cuff with isokinetic testing. Seventy-two elite junior tennis players underwent isokinetic fatigue testing consisting of 20 reciprocal concentric contractions of the internal and external rotators with 90° of glenohumeral joint abduction. The results showed significantly different fatigue responses between the internal and external rotators. Analysis of the relative fatigue ratio, which compares the work performed in the second half of the testing protocol with the work performed in the first half, showed that the internal rotators fatigued to a level of only 83%. The external rotators, however, fatigued to a level of 69% over the 20 testing repetitions.121 This study demonstrated a greater relative degree of muscular fatigue in the external rotators, even in healthy trained subjects, and provides an important rationale for the inclusion of copious amounts of endurance-oriented training of the external rotators in patients with rotator cuff dysfunction or glenohumeral joint instability.
Chen et al133 demonstrated the effects of muscular fatigue on glenohumeral joint kinematics. Subjects were studied radiographically as they elevated their shoulders before and after a series of rehabilitation exercises that produced substantial levels of muscular fatigue in the shoulder. They found significantly greater amounts of superiorly directed humeral head translation documented radiographically with arm elevation after fatigue. This study shows the important role that the rotator cuff plays in maintaining glenohumeral joint congruity and stabilizing the humeral head within the glenoid.34 Repeated attempts to enhance muscular endurance based on these studies, as well as on earlier literature citations linking muscular fatigue of the glenohumeral rotators to decrements in proprioception, are clinically indicated.
Postoperative Applications
The use of treatment techniques to enhance proprioception and neuromuscular control is indicated for the shoulder after surgery. Methods proposed for addressing glenohumeral joint instability include primarily capsular plication and the application of thermal energy to produce capsular shortening, which may acutely alter glenohumeral joint proprioception.20,134 Myers et al135 measured joint position sense, kinesthesia, and shoulder function in patients who underwent thermal capsular shrinkage for glenohumeral joint instability. No significant differences were found in active and passive angular reproduction of joint position sense 6 to 24 months postoperatively. The acute effects of thermal capsular shrinkage on glenohumeral joint proprioception are not completely understood; however, this study shows that full return of proprioceptive function is expected after rehabilitation.
Early postoperative proprioceptive training consists of passive angular joint repositioning in available ROM with the elimination of visual cues. Replication of either the contralateral extremity position or repeated movements can be done passively initially and then be progressed to active angular joint position replication as patient status and tissue healing allow. Early application of the joint approximation exercises and rhythmic stabilization exercises described earlier in this chapter is also beneficial in the early progression after rotator cuff repair and open and arthroscopic stabilization.136
Neuromuscular control is a result of the efferent response to the afferent signals generated through the sensory system. Proprioception plays a critical role in this feedback system. When the proprioceptive pathways are injured or disrupted, not only inefficient motor responses but also greater risk for injury are possible, especially in the area of athletics in which overcoming resistance from opposing players is coupled with environmental obstacles in an often fast-paced arena. In preparing athletes for competition, the clinician should consider the effects of fatigue and disuse on proprioception and also their potential impact when designing a comprehensive training or rehabilitation program.
Use of the clinically oriented exercise progressions highlighted in this chapter, including techniques such as joint approximation, joint oscillation, and local muscular endurance applications, provides the framework for objectively based rehabilitation programs using the scientific concepts reviewed in this chapter.
Conclusion
Introduction
Afferent Mechanoreceptor Classification
Clinical Assessment of Proprioception in the Lower Extremity
Assessment of Proprioception and Neuromuscular Control in the Upper Extremity
Effects of Aging, Instability, and Injury on Lower Extremity Proprioception
Effects of Fatigue on Lower Extremity Proprioception
Effects of Muscular Fatigue on Upper Extremity Proprioception and Neuromuscular Control
Techniques to Improve Lower Extremity Proprioception and Neuromuscular Control
Guidelines for Implementing Lower Extremity Proprioceptive Training
Techniques to Improve Proprioception and Neuromuscular Control of the Upper Extremity
1 Williams G.N., Chmielewski T., Rudolph K.S., et al. Dynamic knee stability: Current theory and implications for clinical scientists. J. Orthop. Sports Phys. Ther.. 2001;31:546-566.
2 Lephart S.M., Fu F.H. Proprioception and Neuromuscular Control in Joint Stability. Champaign, IL: Human Kinetics; 2000.
3 Riemann B.L., Myers J.B., Lephart S.M. Sensorimotor system measurement techniques. J. Athl. Train.. 2002;37:85-98.
4 Goetz C.G. Textbook of Clinical Neurology, 1st ed. Philadelphia: Saunders; 1999.
5 Adams R.D., Victor M., Ropper A.H. Principles of Neurology, 6th ed. New York: McGraw-Hill; 1997.
6 Sherrington C. The Integrative Action of the Nervous System. New York: Scribner’s Son; 1906.
7 Goldscheider A. Gesammelte Abhandlungen. Leipzig, Germany, Barth: II. Physiologie des Muskelsinnes; 1898.
8 Roland P.E., Ladegaard-Pedersen H. A quantitative analysis of sensations of tension and of kinesthesia in man: Evidence for a peripherally originating muscular sense and for a sense of effort. Brain. 1977;100:671-692.
9 Goodwin G.M., McCloskey D.I., Matthews P.B.C. The contribution of muscle afferents to kinesthesia shown by vibration induced illusions of movement and by the effects of paralyzing joint afferents. Brain. 1972;95:705-748.
10 Eklund G. Position sense and state of contraction: The effects of vibration. J. Neurol. Neurosurg. Psychiatry. 1972;35:606-611.
11 Grigg P. Peripheral mechanisms in proprioception. J. Sport Rehabil.. 1994;3:2-17.
12 Wyke B. Articular neurology—A review. Physiotherapy. 1972;58:94-99.
13 Morisawa Y., Kawakami T., Uermura H., et al. Mechanoreceptors in the coraco-acromial ligament. A study of the aging process. J. Shoulder Elbow Surg. 1994;3:S45.
14 Wyke B.D. The neurology of joints. Ann. R. Coll. Surg. Engl.. 1967;41:25.
15 Myers J.B., Lephart S.M. The role of the sensorimotor system in the athletic shoulder. J. Athl. Train.. 2000;35:351-363.
16 Vangsness C.T., Ennis M., Taylor J.G., et al. Neural anatomy of the glenohumeral ligaments, labrum, and subacromial bursa. Arthroscopy. 1995;11:180-184.
17 Kikuchi T. Histological studies on the sensory innervation of the shoulder joint. J. Iwate Med. Assoc.. 1968;20:554-567.
18 Shimoda F. Innervation, especially sensory innervation of the knee joint and motor organs around it in early stage of human embryo. Arch. Histol. (Jpn.). 1955;9:91-108.
19 Ide K., Shirai Y., Ito H., et al. Sensory nerve supply in the human subacromial bursa. J. Shoulder Elbow Surg.. 1996;5:371-382.
20 Nyland J.A., Caborn D.N.M., Johnson D.L. The human glenohumeral joint: A proprioceptive and stability alliance. Knee Surg. Sports Traumatol. Arthrosc.. 1998;6:50-61.
21 Barker D., Banks R.W., Harker D.W., et al. Studies of the histochemistry, ultrastructure, motor innervation, and regeneration of mammalian intrafusal muscle fibers. Exp. Brain Res.. 1976;44:67-88.
22 Voss H. Tabelle der absoluten und relativen Muskel-spindelzahlen der menschlichen Skelettmuskulatur. Anat. Anz.. 1971;129:562-572.
23 Inman V.T., Saunders J.B., Abbot L.C. Observations on the function of the shoulder joint. J. Bone Joint Surg.. 1944;26:1-30.
24 Marshall R.N., Elliot B.C. Long-axis rotation: The missing link in proximal to distal segmental sequencing. J. Sports Sci.. 2000;18:247-254.
25 Lattanzio P.J., Petrella R.J., Sproule J.R., et al. Effects of fatigue on knee proprioception. Clin. J. Sports Med.. 1997;7:22-27.
26 Pincivero D.M., Coelho A.J. Proprioceptive measures warrant scrutiny. Biomechanics. 2001;3:77-86.
27 Barrack R.L., Skinner H.B., Buckley S.L. Proprioception in the anterior cruciate–deficient knee. Am. J. Sports Med.. 1989;17:1-6.
28 Skinner H.B., Wyatt M.P., Hodgdon J.A., et al. Effect of fatigue on joint position sense of the knee. J. Orthop. Res.. 1986;4:112-118.
29 Beynnon B.D., Good L., Risberg M.A. The effect of bracing on proprioception of knees with anterior cruciate ligament injury. J. Orthop. Sports Phys. Ther.. 2002;32:11-23.
30 Barrack R.L., Skinner H.B., Cook S.D., et al. Effect of articular disease and total knee arthroplasty on knee joint-position sense. J. Neurophysiol.. 1983;50:684-687.
31 Friden T., Roberts M., Ageberg E., et al. Review of knee proprioception and the relation to extremity function after an anterior cruciate ligament rupture. J. Orthop. Sports Phys. Ther.. 2001;31:568-576.
32 Marks R., Quinney H.A. Effect of fatiguing maximal isokinetic quadriceps contractions on ability to estimate knee position. Percept. Mot. Skills. 1993;77:1195-2002.
33 Tropp H., Ekstrand J., Gillquist J. Factors affecting stabilometry recordings of single limb stance. Am. J. Sports Med.. 1984;12:185-188.
34 Blaiser R.B., Carpenter J.E., Huston L.J. Shoulder proprioception: Effect of joint laxity, joint position, and direction of motion. Orthop. Rev.. 1994;23:45-50.
35 Smith R.L., Brunolli J. Shoulder kinesthesia after anterior glenohumeral joint dislocation. Phys. Ther.. 1989;69:106-112.
36 Lephart S.M., Warner J.J.P., Borsa P.A., Fu F.H. Proprioception of the shoulder joint in healthy, unstable, and surgically repaired shoulders. J. Shoulder Elbow Surg.. 1994;3:371-380.
37 Warner J.J.P., Bowen M.K., Deng X., et al. Effect of joint compression on inferior stability of the glenohumeral joint. J. Shoulder Elbow Surg.. 1999;8:31-36.
38 Warner J.J.P., Lephart S., Fu F.H. Role of proprioception in pathoetiology of shoulder instability. Clin. Orthop. Relat. Res.. 1996;330:35-39.
39 Allegrucci M., Whitney S.L., Lephart S.M., et al. Shoulder kinesthesia in healthy unilateral athletes participating in upper extremity sports. J. Orthop. Sports. Phys. Ther.. 1995;21:220-226.
40 Davies G.J., Hoffman S.D. Neuromuscular testing and rehabilitation of the shoulder complex. J. Orthop. Sports Phys. Ther.. 1993;18:449-457.
41 Voight M.L., Hardin J.A., Blackburn T.A., et al. The effects of muscle fatigue on and the relationship of arm dominance to shoulder proprioception. J. Orthop. Sports Phys. Ther.. 1996;23:348-352.
42 Jerosch J.G. Effects of shoulder instability on joint proprioception. In: Lephart S.M., Fu F.H., editors. Proprioception and Neuromuscular Control in Joint Stability. Champaign, II: Human Kinetics, 2000.
43 Slobounov S.M., Poole S.T., Simon R.F., et al. The efficacy of modern technology to improve healthy and injured shoulder joint position sense. J. Sport Rehabil.. 1999;8:10-23.
44 Ballantyne B.T., O’Hare S.J., Paschall J.L., et al. Electromyographic activity of selected shoulder muscles in commonly used therapeutic exercises. Phys. Ther.. 1993;73:668-682.
45 Blackburn T.A., McLeod W.D., White B., et al. EMG analysis of posterior rotator cuff exercises. Athl. Train.. 1990;25:40-45.
46 Moesley J.B., Jobe F.W., Pink M. EMG analysis of the scapular muscles during a shoulder rehabilitation program. Am. J. Sports Med.. 1992;20:128-134.
47 Townsend H., Jobe F.W., Pink M., et al. Electromyographic analysis of the glenohumeral muscles during a baseball rehabilitation program. Am. J. Sports Med.. 1991;19:264-272.
48 DiGiovine N.M., Jobe F.W., Pink M., et al. An electromyographic analysis of the upper extremity in pitching. J. Shoulder Elbow Surg.. 1994;1:15-25.
49 Rhu K.N., McCormick J., Jobe F.W., et al. An electromyographic analysis of shoulder function in tennis players. Am. J. Sports Med.. 1988;16:481-485.
50 Kronberg M., Brostrom L.A., Nemeth G. Differences in shoulder muscle activity between patients with generalized joint laxity and normal controls. Clin. Orthop. Relat. Res.. 1991;209:181-192.
51 Ludewig P.M., Cook T.M. Alterations in shoulder kinematics and associated muscle activity in people with symptoms of shoulder impingement. Phys. Ther.. 2000;80:276-291.
52 McLeod T.C., Armstrong T., Miller M., Sauers J.L. Balance improvements in female high school basketball players after a 6-week neuromuscular training program. J. Sport Rehabil.. 2009;18:465-481.
53 Glousman R., Jobe F., Tibone J., et al. Dynamic electromyographic analysis of the throwing shoulder with glenohumeral instability. J. Bone Joint Surg. Am.. 1988;70:220-226.
54 Ellenbecker T.S. Rehabilitation of shoulder and elbow injuries in tennis players. Clin. Sports Med.. 1995;14:87-110.
55 Ellenbecker T.S., Cappel K. Clinical application of closed kinetic chain exercises in the upper extremities. Orthop. Phys. Ther. Clin. North Am.. 2000;9:231-245.
56 Ellenbecker T.S., Manske R., Davies G.J. Closed kinetic chain testing techniques of the upper extremities. Orthop. Phys. Ther. Clin. North Am.. 2000;9:219-245.
57 Wilk K.E., Arrigo C. Current concepts in the rehabilitation of the athletic shoulder. J. Orthop. Sports Phys. Ther.. 1993;18:365-378.
58 Wilk K.E., Arrigo C.A., Andrews J.R. Closed and open kinetic chain exercises for the upper extremity. J. Sport Rehabil.. 1996;5:88-102.
59 Roetert E.P., Ellenbecker T.S. Complete Conditioning for Tennis. Champaign, IL: Human Kinetics; 1998.
60 Ellenbecker T.S., Davies G.J. The application of isokinetics in testing and rehabilitation of the shoulder complex. J. Athl. Train.. 2000;35:338-350.
61 Goldbeck T.G., Davies G.J. Test-retest reliability of the closed kinetic chain upper extremity stability test: A clinical field test. J. Sport Rehabil.. 2000;9:35-45.
62 Provins K.A. The effect of peripheral nerve block on the appreciation and execution of finger movements. J. Physiol.. 1958;143:55-67.
63 Kaplan F.S., Nixon J.E., Reitz M., et al. Age-related changes in joint proprioception and sensation of joint position. Acta Orthop. Scand.. 1985;56:72-74.
64 Skinner H.B., Barrack R.L., Cook S.D. Age related decline in proprioception. Clin. Orthop. Relat. Res.. 1984;184:208-211.
65 Beard D.J., Kyberd P.J., Ferguson C.M., et al. Proprioception after rupture of the anterior cruciate ligament. J. Bone Joint Surg. Br.. 1993;73:311-315.
66 Hurley M.V., Newham D.J. The influence of arthrogenous muscle inhibition on quadriceps rehabilitation of patients with early, unilateral osteoarthritic knees. Br. J. Rheumatol.. 1993;32:127-131.
67 Sharma L., Pai Y.C. Impaired proprioception and osteoarthritis. Curr. Opin. Rheumatol.. 1997;9:253-258.
68 Barrack R.L., Skinner H.B., Brunet M.E., Cook S.D. Joint laxity and proprioception in the knee. Phys. Sports Med.. 1983;11:130-135.
69 Garn S.N., Newton R.A. Kinesthetic awareness in subjects with multiple ankle sprains. Phys. Ther.. 1988;68:1667-1671.
70 Lentell G.G., Baas B., Lopez D., et al. The contributions of proprioceptive deficit, muscle function, and anatomic laxity to functional instability of the ankle. J. Orthop. Sports Phys. Ther.. 1995;21:206-215.
71 Nawoczenski D.A., Ownen G., Ecker B., et al. Objective evaluation of peroneal response to sudden inversion stress. J. Orthop. Sports Phys. Ther.. 1985;25:107-109.
72 Warren P.J., Olankun T.K., Cobb A.G., Bentley G. Proprioception after knee arthroplasty: The influence of prosthetic design. Clin. Orthop. Relat. Res.. 1993;297:182-187.
73 Barrett D.S., Cobb A.G., Bently G. Joint proprioception in normal osteoarthritic and replaced knees. J. Bone Joint Surg. Br.. 1991;73:53-56.
74 Andriacchi T.P., Galante J.O. Retention of the posterior cruciate ligament in total knee arthroplasty. J. Arthroplasty. 1988;3(Suppl.):S13-S19.
75 Dorr L.D., Ochsner J.L., Growley J., Perry J. Functional comparisons of posterior cruciate retained versus sacrificed in total knee arthroplasty. Clin. Orthop. Relat. Res.. 1988;236:36-43.
76 Beard D.J., Dodd C.F., Trundle H.R., Simpson A.W. Proprioception enhancement for anterior cruciate ligament deficiency. J. Bone Joint Surg. Br.. 1994;76:654-659.
77 Limbird T.J., Shiavir R., Frazer M., Borra H. EMG profiles of knee joint musculature during walking: Changes induced by anterior cruciate ligament deficiency. J. Orthop. Res.. 1988;6:630-638.
78 Andriacchi T.P., Birac D. Functional testing in the anterior cruciate ligament–deficient knee. Clin. Orthop. Relat. Res.. 1993;288:40-47.
79 Speer K.P., Deng X., Borrero S., et al. Biomechanical evaluation of a simulated Bankart lesion. J. Bone Joint Surg. Am.. 1994;76:1819-1826.
80 Barden J.M., Balyk R., Raso J., et al. Dynamic upper limb proprioception in multidirectional shoulder instability. Clin. Orthop. Relat. Res.. 2004;420:181-189.
81 Safran M.R., Borsa P.A., Lepahrt S.M, et al. Shoulder proprioception in baseball pitchers. J. Shoulder Elbow Surg.. 2001;10:438-444.
82 Rokito A.S., Birdzell M.G., Cuomoa F., et al. Recovery of shoulder strength and proprioception after open surgery for recurrent anterior instability: A comparison of two surgical techniques. J. Shoulder Elbow Surg.. 2010;19:564-569.
83 McMahon P.J., Jobe F.W., Pink M.M., et al. Comparative electromyographic analysis of shoulder muscles during planar motions: Anterior glenohumeral instability versus normal. J. Shoulder Elbow Surg.. 1996;5:118-123.
84 Skinner H.B., Wyatt M.P., Stone M.L., et al. Exercise related knee joint laxity. Am. J. Sports Med.. 1986;14:30-34.
85 Weisman G., Pope M.H., Hohnson R.J. Cyclic loading in knee ligament injuries. Am. J. Sports Med.. 1980;8:24-30.
86 Lephart S.M., Henry T.J., Riemann B.L., et al. The effects of neuromuscular control exercises on functional stability in the unstable shoulder. J. Athl. Train.. 1998;33:S15.
87 Lattanzio P.J., Petrella R.J. Knee proprioception: A review of mechanisms, measurements, and implications of muscular fatigue. Orthopedics. 1998;21:463-470.
88 Zuckerman J.D., Gallagher M.A., Lehman C., et al. Normal shoulder proprioception and the effect of lidocaine injection. J. Shoulder Elbow Surg.. 1999;8:11-16.
89 Carpenter J.E., Blaiser R.B., Pellizon G.G. The effects of muscle fatigue on shoulder joint position sense. Am. J. Sports Med.. 1998;26:262-265.
90 Pederson J., Jonn J., Hellstrom F., et al. Localized muscle fatigue decreases the acuity of the movement sense in the human shoulder. Med. Sci. Sports Exerc.. 1999;31:1047-1052.
91 Myers J.B., Guskiewicz K.M., Schneider R.A., et al. Proprioception and neuromuscular control of the shoulder after muscle fatigue. J. Athl. Train.. 1999;34:362-367.
92 Myers J.B., Ju Y.Y., Hwang J.H., et al. Reflexive muscle activation alterations in shoulders with anterior glenohumeral instability. Am. J. Sports Med.. 2004;32:1013-1021.
93 Myers J.B., Lephart S.M. The role of the sensorimotor system in the athletic shoulder. J. Athl. Train.. 2000;35:351-363.
94 Myers J.B., Wassinger C.A., Lephart S.M. Sensorimotor contribution to shoulder stability: Effect of injury and rehabilitation. Man. Ther.. 2006;11:197-201.
95 Trip B.L., Yochem E.M., Uhl T.L. Functional fatigue and upper extremity sensorimotor system acuity in baseball athletes. J. Athl. Train.. 2007;42:90-98.
96 Lin J.J., Hung C.J., Yang P.L. The effects of scapular taping on electromyographic muscle activity and proprioception feedback in healthy shoulders. J. Orthop. Res.. 2011;29:53-57.
97 Djupsjobacka M., Johansson H., Bergenheim M. Influences on the gamma muscle spindle system from muscle afferents stimulated by increased intramuscular concentrations of arachidonic acid. Brain Res.. 1994;663:293-302.
98 Djupsjobacka M., Johansson H., Bergenheim M., et al. Influences on the gamma muscle spindle system from muscle afferents stimulated by increased intramuscular concentrations of bradykinin and 5-HT. Neurosci. Res.. 1995;22:325-333.
99 Djupsjobacka M., Johansson H., Bergenheim, et al. Influences on the gamma muscle spindle system from contralateral muscle afferents stimulated by KCl and lactic acid. Neurosci. Res.. 1995;21:301-309.
100 Cerulli G., Benoit D.L., Caraffa A., et al. Proprioceptive training and prevention of anterior cruciate ligament injuries in soccer. J. Orthop. Sports Phys. Ther.. 2001;31:655-661.
101 Osborne M.D., Chou L.S., Laskowski E.R., et al. The effect of ankle disk training on muscle reaction time in subjects with a history of ankle sprain. Am. J. Sports Med.. 2001;29:627-632.
102 Eils E., Dieter R. A multi-station proprioceptive exercise program in patients with ankle instability. Med. Sci. Sports Exerc.. 2001;33:1991-1998.
103 Risberg M.A., Mork M., Jenssen H.K., Holm I. Design and implementation of a neuromuscular training program following anterior cruciate ligament reconstruction. J. Orthop. Sports Phys. Ther.. 2001;31:620-631.
104 Hewett T.E., Paterno M.V., Myer G.D. Strategies for enhancing proprioception and neuromuscular control of the knee. Clin. Orthop. Relat. Res.. 2002;402:76-94.
105 Mandelbaum B.R., Silvers H.J., Watanabe D.S., et al. Effectiveness of a neuromuscular and proprioceptive training program in preventing anterior cruciate ligament injuries in female athletes: 2 year follow-up. Am. J. Sports Med.. 2005;33:1003-1010.
106 Hurd W.J., Chmielewski T.L., Snyder-Mackler L. Perturbation-enhanced neuromuscular training alters muscle activity in female athletes. Knee Surg. Sports Traumatol. Arthrosc.. 2006;14:60-69.
107 Biel A., Dudzinski K. Rehabilitation outcome in patients recovering from reconstruction of the anterior cruciate ligament; a preliminary report. Ortop. Traumatol. Rehabil.. 2005;7:401-405.
108 Hale S.A., Hertel J., Olmsted-Kramer L.C. The effect of a 4-week comprehensive rehabilitation program on postural control and lower extremity function in individuals with chronic ankle instability. J. Orthop. Sports Phys. Ther.. 2007;37:303-311.
109 Wikstrom E.A., Bishop M.D., Inamdar A.D., Hass C.J. Gait termination control strategies are altered in chronic ankle instability subjects. Med. Sci. Sports Exerc.. 2010;42:197-205.
110 Gutierrez G.M., Kaminski T.W., Douex A.T. Neuromuscular control and ankle instability. P MR. 2009;1:359-365.
111 Kynsburg A., Panics G., Halasi T. Long-term neuromuscular training and ankle joint position sense. Acta Physiol. Hung.. 2010;97:183-191.
112 Blackburn J.T., Riemann B.L., Myers J.B., Lephart S.M. Kinematic analysis of the hip and trunk during bilateral stance on firm, foam and multiaxial support surfaces. Clin. Biomech. (Bristol, Avon). 2003;18:655-661.
113 Riemann B.L., Myers J.B., Lephart S.M. Comparison of the ankle, knee, hip and trunk corrective action shown during single-leg stance on firm, foam, and multiaxial surfaces. Arch. Phys. Med. Rehabil.. 2003;84:90-95.
114 Zazulak B.T., Hewett T.E., Reeves N.P., et al. Deficits in neuromuscular control of the trunk predict knee injury risk; a prospective biomechanical-epidemiologic study. Am. J. Sports Med.. 2007;35:1123-1130.
115 Miura K., Ishibashi Y., Tsuda E., et al. The effect of local and general fatigue on knee proprioception. Arthroscopy. 2004;20:414-418.
116 Zech A., Hübscher M., Vogt L., et al. Neuromuscular training for rehabilitation of sports injuries: A systematic review. Med. Sci. Sports Exerc.. 2009;41:1831-1841.
117 Hübscher M., Zech A., Pfeifer K., et al. Neuromuscular training for sports injury prevention; a systematic review. Med. Sci. Sports Exerc.. 2010;42:413-421.
118 Zech A., Hübscher M., Vogt L., et al. Balance training for neuromuscular control and performance enhancement: A systematic review. J. Athl. Train.. 2010;45:392-403.
119 Lephart S.M., Pincivero D.M., Giraldo J.L., Fu F.H. The role of proprioception in the management and rehabilitation of athletic injuries. Am. J. Sports Med.. 1997;25:130-137.
120 Fitzgerals G.K., Axe M.J., Snyder-Mackler L. The efficacy of perturbation training in nonoperative anterior cruciate ligament rehabilitation programs for physically active individuals. Phys. Ther.. 2000;80:128-140.
121 Ellenbecker T.S., Roetert E.P. Testing isokinetic muscular fatigue of shoulder internal and external rotation in elite junior tennis players. J. Orthop. Sports Phys. Ther.. 1999;29:275-281.
122 Snyder-Mackler L.A., Delitto S.L., Straka S.W. Strength of the quadriceps femoris muscle and functional recovery after reconstruction of the anterior cruciate ligament. J. Bone Joint Surg. Am.. 1995;77:1166-1173.
123 Palmitier R.A., An K.N., Scott S.G., et al. Kinetic chain exercise in knee rehabilitation. Sports Med.. 1991;11:402-413.
124 Kibler W.B. The role of the scapula in athletic shoulder function. Am. J. Sports Med.. 1998;26:325-337.
125 Kibler W.B., Livingstone B., Bruce R. Current concepts in shoulder rehabilitation. Adv. Oper. Orthop.. 1995;3:249-297.
126 Decker M.J., Hintermeister R.A., Faber K.J., Hawkins R.J. Serratus anterior muscle activity during selected rehabilitation exercises. Am. J. Sports Med.. 1999;27:784-791.
127 Poppen N.K., Walker P.S. Normal and abnormal motion of the shoulder. J. Bone Joint Surg. Am.. 1976;58:195-201.
128 Saha A.K. Mechanism of shoulder movements and a plea for the recognition of “zero-position” of glenohumeral joint. Clin. Orthop. Relat. Res.. 1983;173:3-10.
129 Kraemer W.J., Fleck S.J. Designing Resistance Training Programs, 3 rd ed. Champaign, IL: Human Kinetics; 2003.
130 Elliot B., Marsh T., Blanksby B. A three dimensional cinematographic analysis of the tennis serve. Int. J. Sport Biomech.. 1986;2:260-271.
131 Holt S., O’Brien M., Davies G.J., et al An investigation of shoulder muscle electrical activity during Bodyblade exercises Presented at Wisconsin State Physical Therapy Association Spring Meeting 2000
132 Marin P.J., Rhea M.R. Effects of vibration training on muscle strength; a meta-analysis. J. Strength Cond. Res.. 2010;24:548-556.
133 Chen S.K., Simonion P.T., Wickiewicz T.L., Warren R.F. Radiographic evaluation of glenohumeral kinematics: A muscle fatigue model. J. Shoulder Elbow Surg.. 1999;8:49-52.
134 Lu Y., Hayashi K., Edwards R.B., et al. The effect of monopolar radiofrequency treatment pattern on joint capsular healing. In vitro and in vivo studies using an ovine model. Am. J. Sports Med.. 2000;28:711-719.
135 Myers J.B., Lephart S.M., Riemann B.L., et al. Evaluation of shoulder proprioception following thermal capsulorraphy. Med. Sci. Sports Exer.. 2000;32:S123.
136 Ellenbecker T.S., Mattalino A.J. Glenohumeral joint range of motion and rotator cuff strength following arthroscopic anterior stabilization with thermal capsulorraphy. J. Orthop. Sports Phys. Ther.. 1999;29:160-167.