18 Dermatopathology
Of all organ systems, the skin has the greatest number of lesions described, perhaps because the skin is subject to a wide variety of environmental exposures and no special procedures are necessary to visualize its surface.
RELEVANT CLINICAL HISTORY (IN ADDITION TO AGE AND GENDER)
TABLE 18–1 RELEVANT CLINICAL HISTORY
| HISTORY RELEVANT TO ALL SPECIMENS | HISTORY RELEVANT FOR DERMATOPATHOLOGY SPECIMENS |
|---|---|
| Organ/tissue resected or biopsied | Site, duration, and appearance of the lesion (especially for incisional biopsies) |
| Purpose of the procedure | |
| Gross appearance of the organ/tissue/lesion sampled | Systemic diseases that affect the skin |
| Any unusual features of the clinical presentation | Clinical differential diagnosis |
| Any unusual features of the gross appearance | Family history (see Table 7-50) |
| Prior surgery/biopsies – results | Previous similar lesions |
| Prior malignancy | |
| Prior treatment (radiation therapy, chemotherapy, drug use that can change the histologic appearance of tissues) | |
| Compromised immune system |
GENERAL CONSIDERATIONS
The ability to clearly visualize the entire epidermis in a perpendicular section is important for diagnosis, and at times for prognosis (e.g., malignant melanoma). Therefore, try to maintain vertical orientation at all times in sections. Any specimen that is labeled “excision,” regardless of the type of specimen, must have the margins evaluated by inking and submission of appropriate sections. Diagrams are used for any difficult or complicated specimens.
Never cut through small vesicular lesions in any type of specimen. The overlying tissue layer is important for diagnosis, but is fragile and easily detached and lost. Cut the specimen so as to leave the vesicle intact or submit small specimens whole and request that the histotechnologists bisect the specimen after processing.
SKIN PUNCH BIOPSIES
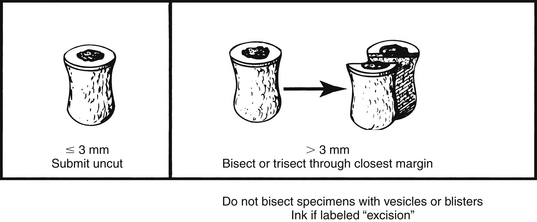
Punch biopsies are performed to completely excise small lesions, to sample large lesions, or to evaluate an inflammatory process or a systemic disease (e.g., pustular psoriasis). Punches can be 2, 3, 4, 5, 6, 8, or 10 mm in diameter.
PROCESSING THE SPECIMEN
Punch biopsies greater than 4 mm are bisected or trisected, depending on size. If there is a discrete lesion, cut in a plane to demonstrate the closest margin. If the lesion is very small (i.e., leveling the block might remove the lesional tissue), cut the punch biopsy on either side of the lesion. Do not section through vesicles or blisters – submit whole and request sectioning by the laboratory.
SAMPLE DICTATION
Received in formalin labeled with the patient’s name and unit number and “5 mm punch, left leg” is a 5 mm in diameter by 5 mm (depth) punch biopsy with tan/white skin. There is a flat homogeneously brown lesion with slightly irregular margins, 0.3 × 0.3 cm, on the skin surface. The lesion is less than 0.1 cm from the nearest margin, but does not grossly involve the margin.
SKIN SHAVE BIOPSIES
Shave biopsies are usually performed to remove nonmalignant lesions (e.g., seborrheic keratoses, actinic keratoses, or fibroepithelial polyps) or for diagnosis of basal cell carcinomas. Shave biopsies of pigmented lesions should be strongly discouraged and interpreted with caution. The diagnosis of melanoma may be difficult in such a specimen due to limited sampling and the depth of invasion may be impossible to assess. Specimens are inked if designated “excisions.”
PROCESSING THE SPECIMEN
SKIN CURETTINGS
Skin scrapings (curettings) of seborrheic or actinic keratosis or basal cell carcinoma may be performed.
PROCESSING THE SPECIMEN
SKIN ELLIPSES
These specimens are excisions of malignant tumors (squamous cell carcinoma or basal cell carcinoma), typical or atypical melanocytic nevi (and to rule out melanoma), or large benign lesions (e.g., epithelial inclusion cysts). Occasionally, ellipses are submitted for the evaluation of panniculitis or large vessel vasculitis.
PROCESSING THE SPECIMEN
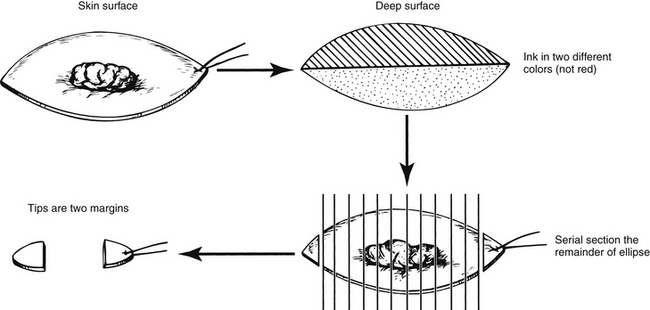
Describe any lesions including color, borders, ulceration, shape, and distance from margins. Describe any scars from prior biopsies (length, recent or well-healed).
Serially section the entire specimen along the short axis at 2 to 3 mm intervals.
Submit the most distal sections (“tips”) as two of the margins in two cassettes. If the ellipse is small and unoriented, both tips can be placed in one cassette. Each tip is taken as a 0.1 to 0.2 cm en face margin.
Submit the remainder of the specimen in one or more cassettes.
A simple diagram showing orientation and sections is very helpful in interpreting the sections histologically. This is especially true for large or irregularly shaped skin excisions.
For pigmented lesions, the initial cut is made through the thickest or darkest portion of the lesion (most likely area to have deep invasion). Describe the gross depth of invasion or involvement of subcutaneous tissues.
GROSS DIFFERENTIAL DIAGNOSIS
Seborrheic Keratosis
This is a rounded, raised, lesion sharply demarcated from any surrounding skin with a “stuck on” appearance. The color is usually dark brown, black, or gray and has a dirty appearance due to the presence of horn cysts containing keratinous debris.
Epidermal Inclusion Cyst
These cysts are often received in multiple fragments. The wall of the cyst is thin (1 to 2 mm) and has a smooth inner lining. The cyst is filled with white or yellow friable, often malodorous, material corresponding to keratinaceous debris. Some are due to traumatic or iatrogenic introduction of keratinizing epithelium into deep soft tissues. If located near the nipple, the lesion may be in a lactiferous sinus (squamous metaplasia of lactiferous ducts or recurrent subareolar abscess).
Fibroepithelial Polyp (Acrochordon)
A flesh colored papule often designated “skin tag” by the clinician.
Basal Cell Carcinoma
Translucent papule or plaque with a yellow or pearly hue. Central ulceration with a rolled border is common in larger lesions. The outer margin tends to be sharply demarcated from the surrounding skin.
MICROSCOPIC SECTIONS
SAMPLE DICTATION
Received in formalin labeled with the patient’s name, unit number, and “right shoulder” is a 2.5 × 1 × 0.8 cm (depth) skin ellipse with tan/brown skin and an orienting suture at one tip (designated by the surgeon as 12 o’clock). There is a variegated brown and black lesion (1 × 0.8 cm) with markedly irregular borders with notching located in the center of the ellipse. Within the lesion there is a raised black nodule (0.3 × 0.3 cm) that grossly appears to extend through the epidermis into the dermis and is 0.2 cm from the deep resection margin. The closest margin is the 3 o’clock margin, which is 0.1 cm from the lesion. The 3 o’clock margin is inked black and the 9 o’clock margin is inked blue.
PATHOLOGIC PROGNOSTIC/DIAGNOSTIC FEATURES SIGN-OUT CHECKLIST FOR SKIN CARCINOMAS
Table 18–2 HISTOLOGIC GRADE – SQUAMOUS CELL CARCINOMA
| Grade 1 | Well differentiated tumors are characterized by squamous epithelium that frequently shows easily recognizable and often abundant keratinization. Intercellular bridges are readily apparent. There is minimal pleomorphism, and mitotic figures are mainly basally located. |
| Grade 2 | Moderately differentiated tumors show more structural disorganization in which squamous epithelial derivation is less obvious. Nuclear and cytoplasmic pleomorphism are more pronounced, and mitotic figures may be numerous. Keratin formation is typically limited to keratin pearls, horn cysts, and scattered individually keratinized cells. |
| Grade 3 | In poorly differentiated tumors it may be difficult to establish squamous differentiation, usually by identification of rare intercellular bridges or small foci of keratinization. |
| Grade 4 | Used to denote anaplastic or undifferentiated tumors. |
From Protocol for the Examination of Specimens from Patients with Squamous Cell Carcinoma of the Skin (www.cap.org).
Table 18–3 AJCC (7TH EDITION) CLASSIFICATION OF CUTANEOUS SQUAMOUS CELL CARCINOMA AND OTHER CUTANEOUS CARCINOMAS
| TUMOR | ||
| TX | Primary tumor cannot be assessed. | |
| T0 | No evidence of primary tumor | |
| Tis | Carcinoma in situ | |
| T1 | Tumor 2 cm or less in greatest dimension with less than two high-risk features∗ | |
| T2 | Tumor greater than 2 cm in greatest dimension or Tumor any size with two or more high-risk features∗ | |
| T3 | Tumor with invasion of maxilla, mandible, orbit, or temporal bone | |
| T4 | Tumor with invasion of skeleton (axial or appendicular) or perneural invasion of skull base | |
| ∗HIGH-RISK FEATURES FOR THE PRIMARY TUMOR (T) STAGING | ||
| Depth/invasion | ||
| Anatomic location | ||
| Differentiation | Poorly differentiated or undifferentiated | |
| REGIONAL LYMPH NODES | ||
| NX | Regional lymph nodes cannot be assessed. | |
| N0 | No regional lymph node metastasis | |
| N1 | Metastasis in a single ipsilateral lymph node, 3 cm or less in greater dimension. | |
| N2 | Metastasis in a single ipsilateral lymph node, more than 3 cm but not more than 6 cm in greatest dimension; or in multiple ipsilateral lymph nodes, none more than 6 cm in greatest dimension; or in bilateral or contralateral lymph nodes, none more than 6 cm in greatest dimension. | |
| N2a | Metastasis in a single ipsilateral lymph node, more than 3 cm but not more than 6 cm in greatest dimension. | |
| N2b | Metastasis in multiple ipsilateral lymph nodes, none more than 6 cm in greatest dimension. | |
| N2c | Metastasis in bilateral or contralateral lymph nodes, none more than 6 cm in greatest dimension. | |
| N3 | Metastasis in a lymph node, more than 6 cm in greatest dimension | |
| DISTANT METASTASIS | ||
| M0 | No distant metastasis | |
| M1 | Distant metastasis | |
Note: Used for squamous cell and basal cell carcinomas of the skin and adenocarcinomas developing from sweat or sebaceous glands. The classification is not used for carcinomas of the eyelid, melanomas, or Merkel cell carcinomas.
From the AJCC Cancer Staging Manual, Seventh Edition. New York, Springer-Verlag, 2009. Used with the permission of the American Joint Committee on Cancer (AJCC), Chicago, Illinois.
This checklist incorporates information from the CAP Cancer Committee protocols for reporting on cancer specimens (see www.cap.org/) and the ADASP (see www.adasp.org). The underlined elements are considered to be scientifically validated or regularly used data elements that must be present in reports of cancer-directed surgical resection specimens from ACS CoC-approved cancer programs. The specific details of reporting the elements may vary among institutions.
PATHOLOGIC PROGNOSTIC/DIAGNOSTIC FEATURES SIGN-OUT CHECKLIST FOR MELANOMA OF THE SKIN
TABLE 18–4 BRESLOW DEPTH OF INVASION
Measurements should be performed with an ocular reticule.
Adapted from Breslow A: Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann Surg 172:902-908, 1970.
| Level I | Melanoma confined to the epidermis and epidermal appendages |
| Level II | Extension into the papillary dermis by single cells, and sometimes small clusters of cells, with, at most, only a few cells extending to the interface between the papillary and reticular dermis |
| Level III | Extension of tumor cells throughout the papillary dermis, filling it and impinging upon the reticular dermis but not invading it |
| Level IV | Invasion of the reticular dermis |
| Level V | Invasion of the subcutaneous fat |
Adapted from Clark WH Jr, From L, Bernardino EH, et al: Histogenesis and biologic behavior of primary human malignant melanoma of the skin. Cancer Res 29:705-727, 1969.
TABLE 18–6 IMMUNOHISTOCHEMICAL MELANOMA MARKERS
| TYPE OF CELL IN THE LYMPH NODE | S100 | MART-1 |
|---|---|---|
| Metastatic melanoma | POS | High∗ |
| Nevus cells | POS | POS |
| Dendritic cells | POS | neg |
| Nerves, ganglion cells | POS | neg |
∗ About 20% of metastatic melanomas are negative for HMB45 and MART-1.
Table 18–7 AJCC (7TH EDITION) CLASSIFICATION OF MELANOMA OF THE SKIN
Note: This classification is not used for melanomas of sites other than skin (e.g., ocular, mucosal, urethral, etc.).
From the AJCC Cancer Staging Manual, Seventh Edition. New York, Springer-Verlag, 2009. Used with the permission of the American Joint Committee on Cancer (AJCC), Chicago, Illinois.
Table 18–8 AJCC (7TH EDITION) CLASSIFICATION – MERKEL CELL CARCINOMA
| TUMOR | |
| TX | Primary tumor cannot be assessed. |
| T0 | No evidence of primary tumor (e.g., nodal/metastatic presentation without associated primary) |
| Tis | In situ primary tumor |
| T1 | Less than or equal to 2 cm maximum tumor dimension |
| T2 | Greater than 2 cm but not more than 5 cm maximum tumor dimension |
| T3 | Over 5 cm maximum tumor dimension |
| T4 | Primary tumor invades bone, muscle, fascia, or cartilage |
| REGIONAL LYMPH NODES | |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| cN0 | Nodes negative by clinical exam∗ (no pathologic node exam performed) |
| pN0 | Nodes negative by pathologic exam |
| N1 | Metastasis in regional lymph node(s) |
| N1a | Micrometastasis |
| N1b | Macrometastasis |
| N2 | In transit metastasis |
| ∗ Clinical detection of nodal disease may be via inspection, palpation, and/or imaging. Micrometastases are diagnosed after sentinel or elective lymphadenectomy. A micrometastasis is a pathologically documented metastasis (of any size) in a node not detected by clinical or radiologic examination. Macrometastases are defined as clinically detectable nodal metastases confirmed by therapeutic lymphadenectomy or needle biopsy. In transit metastasis: a tumor distinct from the primary lesion and located either (1) between the primary lesion and the draining regional lymph nodes or (2) distal to the primary lesion. |
|
| DISTANT METASTASIS | |
| M0 | No distant metastasis |
| M1 | Metastasis beyond regional lymph nodes |
| M1a | Metastasis to skin, subcutaneous tissues, or distant lymph nodes |
| M1b | Metastasis to lung |
| M1c | Metastasis to all other visceral sites |
This system does not include Merkel Cell carcinoma of the eyelid.
From the AJCC Cancer Staging Manual, Seventh Edition. New York, Springer-Verlag, 2009. Used with the permission of the American Joint Committee on Cancer (AJCC), Chicago, Illinois.
This checklist incorporates information from the CAP Cancer Committee protocols for reporting on cancer specimens (see www.cap.org/) and the ADASP (see www.adasp.org). The underlined elements are considered to be scientifically validated or regularly used data elements that must be present in reports of cancer directed surgical resection specimens from ACS CoC-approved cancer programs. The specific details of reporting the elements may vary among institutions.
LARGE SKIN EXCISIONS
Large resections are usually carried out after the lesion has been biopsied and a diagnosis made.
PROCESSING THE SPECIMEN
Even the smallest ellipses must be cross-sectioned perpendicular to the scar (i.e., do not bisect longitudinally) in order to evaluate the closest (lateral) margins.
LIP EXCISIONS
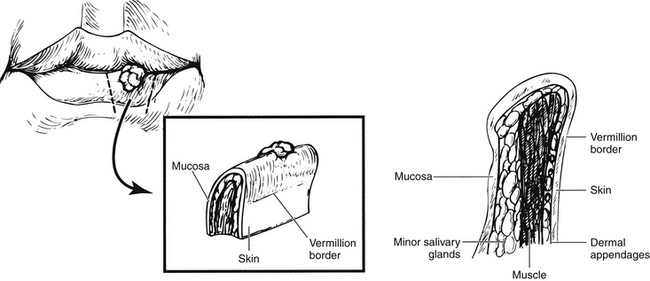
Squamous cell carcinoma is the most common neoplasm of the lip. The specimens are more complicated because there is often both a mucosal and skin surface (Fig. 18-3). There are three margins: the side margins (lateral and medial) margins and a deep margin.
PROCESSING THE SPECIMEN
FORESKIN
The foreskins of newborn infants are generally not submitted for histologic examination unless there are gross abnormalities. Circumcisions of older males are performed in two age groups:
PROCESSING THE SPECIMEN
FINGERNAILS aND TOENAILS
Clippings
Toenail clippings may be sent for the evaluation of fungal infection. Inform the histology laboratory that the specimen consists of a nail as these specimens are usually very difficult to section and may require special techniques for softening.1,2 Order a PAS stain (for fungi).
Nail bed biopsies are usually submitted for the evaluation of pigmented lesions. Do not order a PAS stain on these specimens.
Tumor
Subungual melanomas occur in all ethnic groups but are proportionately more common in persons of color. These lesions may present as linear pigmented streaks of the nail, if the melanoma cells involve the nail matrix. Specimens consisting of only the nail (and not the matrix) will not be diagnostic because melanocytes are not present. The appropriate specimen is a punch biopsy of the nail matrix. If a nail is received with a pigmented area, it is submitted for microscopic examination for evaluation of an atypical melanocytic lesion or melanoma.