Expanding the Differential Diagnosis
Going Beyond IDC-NOS
So you found the architectural distortion on her screening mammogram. She had a diagnostic workup and you found a small abnormal area, no more than 1 or 2 cm. Ultrasound-guided core biopsy showed invasive lobular carcinoma (ILC). She opted for mastectomy even though she looked like a good breast conservation candidate. The pathologist found 7 cm of tumor! How could I have underestimated the size by so much? How could we not have seen such a big invasive cancer last year?
This scenario is actually quite common with ILC, which invades the breast in thin strands of cells much like a spider web. In this chapter we will review the less common breast cancers, including some pathology correlation that may help you expand your differential diagnosis beyond invasive ductal carcinoma (IDC).
Invasive ductal carcinoma–not otherwise specified (IDC-NOS) and ductal carcinoma in situ (DCIS) are by far the most commonly diagnosed breast cancers, making up about 85% of new cases. The remaining 15% of breast cancers often have features that suggest that the diagnosis is something less common.
IDC-NOS simply means that the cancer is differentiated enough to form ducts but does not display other differentiating features, such as making mucin (as in mucinous carcinoma) or papillary formations (as in papillary carcinoma). IDC-NOS typically presents as a spiculated mass or developing asymmetry. DCIS, the nonobligatory predecessor to IDC, typically presents as either coarse heterogeneous or fine pleomorphic calcifications.
The “other” breast cancers can be broadly divided into ILC, the well-differentiated subtypes of IDC, tumors of stromal origin, and metastatic carcinomas. Some rare types of breast malignancies also display characteristic features that suggest the diagnosis. An understanding of these less common breast malignancies will improve skills in detection and diagnosis.
Invasive Lobular Carcinoma
ILC is very different from IDC. On histologic examination, it is characterized by a lack of e-cadherin. Why are we boring you with pathology? Although the study of Latin among premedical students is less common these days, e-cadherin sounds rather like “adhere,” which explains ILC in a nutshell. The cells in ILC lose their ability to adhere to one another, resulting in cancers that are very infiltrative in the breast, like a spider web. ILC is characterized by lines and sheets of cells invading into the breast tissue (Fig. 11-1). This insidious growth pattern and failure to elicit a desmosplastic reaction can make ILC difficult to detect on clinical examination and mammography.
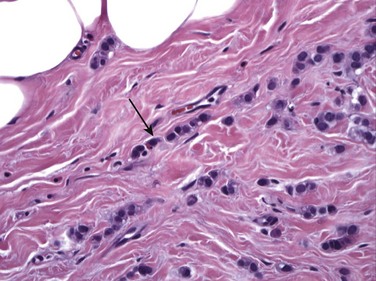
FIGURE 11-1 Histology of ILC.
ILC is characterized by single file sheets of cells (arrow). Note that there is little lymphocytic response or edema. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
Let’s think about how a spider web of tumor cells in the breast (ILC) is going to be different from IDC, where a mass of adherent tumor cells is more characteristic. How would a web of tumor cells feel on clinical examination? Although IDC typically presents as a firm palpable mass on clinical examination, ILC more often presents with an ill-defined mass or thickening of the tissue, or with nipple retraction if the web becomes tethered to the subareolar tissues. If the web of tumor cells is dense enough, ILC can also present as a firm palpable lump.
How would a web of tumor cells look on a mammogram or ultrasonography (US)? The mammogram may show only very subtle findings or be completely normal, even in the setting of extensive ILC (Fig. 11-2). Only the densest part of the tumor will be visible on mammography. ILC may appear as a mass with ill-defined or spiculated margins or as architectural distortion with or without a central mass. When a central mass is present, then it is considered a spiculated mass. When there is architectural distortion without a central mass, these lesions are often called the “dark star.” Calcifications are uncommon, though ILC can have associated DCIS. ILC is often seen in only one view, most commonly the craniocaudal (CC) view. This makes sense because the CC view has better compression of the breast tissue than does the mediolateral oblique (MLO) view. Greater compression can bring out the subtle distortion to better advantage. The extent of disease of ILC is not easily assessed on mammography because the edges of that web are not well defined.
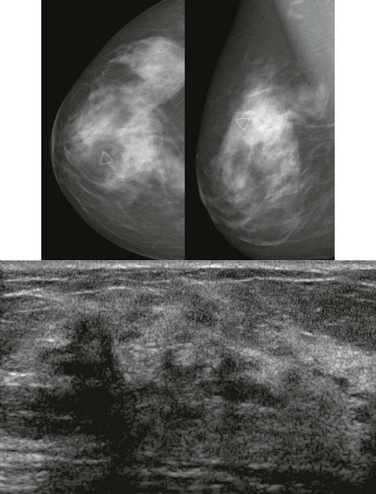
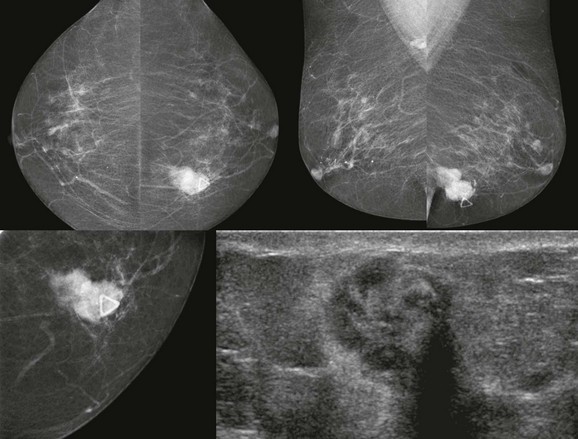
FIGURE 11-2 ILC.
A 49-year-old woman with a firm palpable lump in the right breast. Mammogram is normal in the palpable area that is marked by a triangle. US shows multiple hypoechoic areas with shadowing but no discrete mass, which is characteristic of ILC. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
When the tumor is very large, the breast affected with ILC can appear to be getting smaller on mammography—the “shrinking breast” (Fig. 11-3). This is not due to the breast becoming physically smaller, but to the decreased compressibility of the breast tissue that is full of webs of cancer cells. If the contralateral breast compresses to a thickness of 5 cm, a breast with extensive ILC may only compress to 8 cm. Although this results in the appearance of a smaller breast on mammography, breast size is typically symmetric on clinical examination.
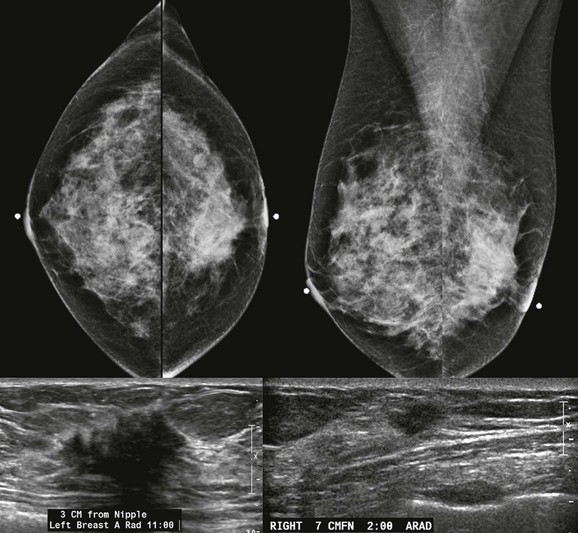
FIGURE 11-3 Shrinking Breast.
A 52-year-old woman noted thickening in her left breast. On clinical examination, the breasts are symmetric in size but the texture of the left breast is diffusely thickened. On mammography, the left breast appears to be smaller than the right. Biopsy showed ILC. Did you also notice the one-view asymmetry in the right medial breast? US of this area shows a small hypoechoic mass. Biopsy of the right breast showed IDC.
On US, ILC may present as a defined mass (see Fig. 11-3) but more often presents as ill-defined areas of shadowing without a distinct mass (see Fig. 11-2). The lines and arcs of tumor can produce bands of shadowing, like the edges of a spider web. Although tumor size is often underestimated by US, this modality is more accurate than clinical examination or mammography in assessing extent of disease.
Magnetic resonance imaging (MRI) is helpful in evaluating the extent of disease with ILC, showing more extensive disease than does mammography in 39% of cases. A cautionary note—ILC may show only faint enhancement with progressive kinetics on MRI. The washout pattern is much less common for ILC than IDC. Remember—when interpreting breast MRI and deciding on your management recommendation, the morphologic appearance always trumps the curve.
ILC is often larger at diagnosis than IDC and is often multifocal. Likewise, ILC is more commonly associated with positive margins and more often treated with mastectomy than IDC. Axillary metastasis is less common, however, so overall ILC has a similar prognosis to IDC. The most evil variant of ILC is pleomorphic ILC. These tumors show more variability of the nuclear size and less uniform cell size. A few recent studies suggest that pleomorphic ILC may have a higher predisposition to metastasis and a worse prognosis compared with usual ILC.
When ILC metastasizes, it may spread to strange places like peritoneal surfaces, the stomach, uterus, ovaries, and bladder. ILC may therefore present with ascites, hydronephrosis, or pelvic masses.
As might be expected, fine-needle aspiration (FNA) of ILC is less sensitive than for IDC. This is logical because FNA of ILC attempts to obtain material from single-file sheets of tumor cells with intervening normal tissue, rather than from a more easily targeted mass of adherent tumor cells that is seen with IDC.
Lobular carcinoma in situ (LCIS) is characterized by a monomorphic population of cells expanding lobules. It is typically mammographically occult and, as such, is an incidental finding on biopsy. However, proliferative lesions such as LCIS are common on biopsy of MRI-detected lesions.
Pleomorphic LCIS is characterized by increased cellular atypia compared with usual LCIS. Some small series suggest a higher incidence of associated invasive carcinoma when core biopsy shows pleomorphic LCIS (Boxes 11-1 and 11-2).
Radial scar can mimic ILC on mammography. Both frequently present as distortion without a central mass (the “dark star”). Radial scar is not actually a scar; it is not due to trauma or surgery, but does look somewhat like a scar at histologic examination. Benign lobules and ducts are entrapped by dense central fibrosis and elastosis, which results in the appearance of architectural distortion on the mammogram (Fig. 11-4). The cause is unclear, but these are proliferative lesions. Hyperplasia, atypical ductal hyperplasia, and papillomas are common in the surrounding tissue. From 10% to 30% of radial scars are associated with DCIS or IDC. Multiple small foci of carcinoma are not uncommon. A history of biopsy showing radial scar is also associated with a higher risk of breast cancer (relative risk of two times that of women without this history).

FIGURE 11-4 Radial Scar with Associated DCIS.
Architectural distortion is present in the right breast (circle). On US, there is a corresponding hypoechoic, irregular mass. Ultrasound-guided core biopsy showed radial scar. Excisional biopsy showed radial scar with associated DCIS.
Radial scar may also mimic an invasive cancer, particularly tubular carcinoma, at both gross and histopathologic examination. A myosin stain is sometimes used to identify myoepithelial cells in the basement membrane, which will be present in radial scar but not in invasive carcinoma.
Radial scar is a general term that may refer to any of these pathologic entities: radial sclerosing lesion, sclerosing papillary proliferation, and complex sclerosing lesion. Radial scar/complex sclerosing lesion is characterized by a dense central fibrotic nidus surrounded by circumferentially radiating ducts and lobules. Complex sclerosing lesions are similar, but larger (usually > 10 mm) and less organized. Sclerosing papillary proliferation is a sclerosing lesion with associated papillomas.
Management of architectural distortion having the dark star appearance on mammography is somewhat controversial. Up to 29% of these women will have invasive carcinoma. At initial mammographic interpretation, either core biopsy or diagnostic surgical biopsy can be performed. Core biopsy showing a benign result will often result in a recommendation for excision of the area due the underlying distortion. On the other hand, surgeons may prefer to know before surgery if the pathologic finding is consistent with radial scar, requiring only a small volume excision, or invasive carcinoma, which requires a larger volume excision and nodal sampling. Hence, core biopsy can aid in biopsy planning (Box 11-3).
If a core biopsy is performed for a different type of BI-RADS 4 lesion, such as calcifications or a focal asymmetry, and shows an incidental minute radial scar that was completely contained within the core sample, excisional biopsy is probably not necessary.
Subtypes of IDC
IDC-NOS is the most common type of breast cancer. Essentially, the carcinoma is differentiated enough to form ducts, but that is all. There are several subtypes of IDC that are better differentiated—they make mucin, form tubules, etc. Because these cancers are more differentiated, they tend to grow slowly (except medullary carcinoma) and be circumscribed (except tubular). They are typically grade I and have a better prognosis than IDC-NOS overall (Boxes 11-4 and 11-5).
Tubular carcinoma is characterized by the formation of tubules or small ductules. It typically presents as a small mass, often with long spicules (Fig. 11-5) like cat whiskers, and is often multifocal. Tubular carcinoma has an excellent outcome with a cause-specific survival rate of 98% at 10 years. Axillary metastasis is very uncommon, and distant disease is even less common. If you could pick your type of invasive cancer to be diagnosed with, this is the one.
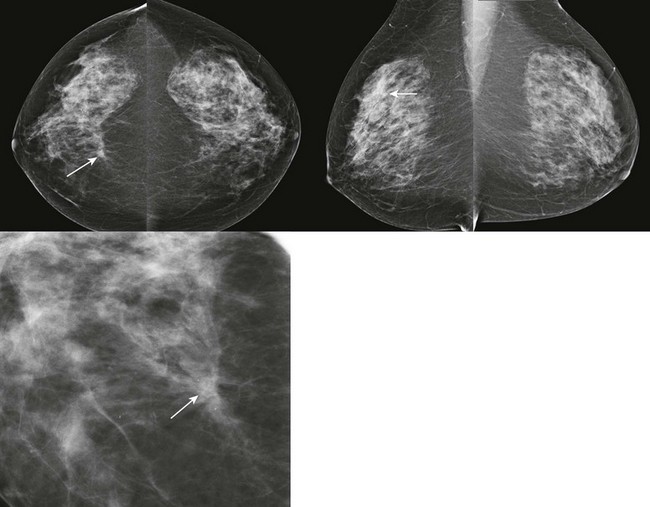
FIGURE 11-5 Tubular Carcinoma.
There is a small spiculated mass in the right breast at 1 o’clock, middle third (arrows). Notice the long spicules along the lateral margin of the mass.
One of our prior fellows (a “Valley Girl” from California), pointed out that “tubular” was synonymous with “cool” during the 1980s. So think, “Like, tubular, man!” and you’ll remember that this is the cool, easy-going invasive cancer despite the appearance of a spiculated mass. A word of caution—if you have teenage kids, we don’t recommend using this language (or related terms like “nifty”) in their presence unless you really want to see their eyes roll!
Histologically, tubular carcinoma can mimic a radial scar and vice versa. The pathologist may use actin stain to check for myoepithelial cells that are present in the basement membrane of tubules associated with radial scar, but absent in tubular carcinoma.
Medullary carcinoma is a very rapidly growing cancer that typically presents as a palpable mass in young or middle-aged women (Fig. 11-6). The median age of diagnosis is 51. “Medulla” means marrow. Like basal cell (triple negative) IDC, medullary carcinoma is characterized by a brisk lymphocytic response, absence of fibroglandular differentiation, and pleomorphic nuclei. There is usually little or no associated DCIS. So at first glance under the microscope medullary carcinoma resembles an aggressive IDC. There is considerable interobserver variability in the pathologic diagnosis of medullary carcinoma that may explain the variable patient outcomes reported across different series. The prognosis for women with this cancer is better than for typical IDC in most series.
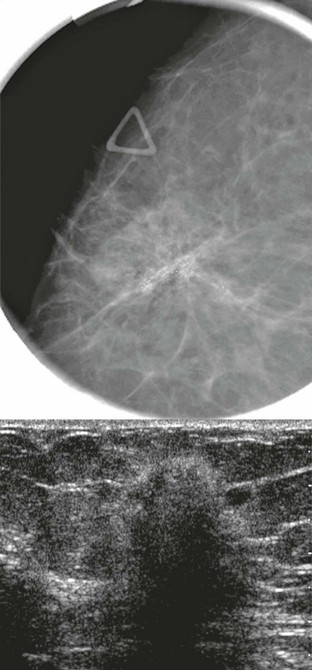
FIGURE 11-6 Medullary Carcinoma.
This 44-year-old woman presented with a palpable mass (triangle) that corresponds to an oval, relatively circumscribed, high-density mass. US demonstrates a hypoechoic, solid, oval mass with posterior acoustic shadowing.
On mammography, medullary carcinoma typically presents as a round or oval, circumscribed mass without calcifications. It is often palpable.
Mucinous (colloid) carcinoma is characterized by tumor cells floating in a pool of mucin (Fig. 11-7). This lesion presents most commonly in older women with a median age of 71. Your last experience with an upper respiratory infection should remind you that mucin is a soft semiliquid. As a result, the mammographic appearance is usually a low-density mass rather than a dense clump of cells like most cancers (Fig. 11-8). The margins are typically fairly well-circumscribed. The US appearance can be relatively isoechoic, and often there is posterior acoustic enhancement. Like proliferative fibroadenomas, these masses are very T2 hyperintense on MRI, but tend to be more heterogeneous on T1 postcontrast sequences.
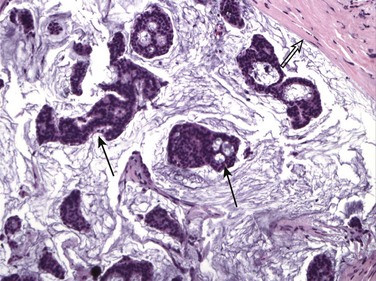
FIGURE 11-7 Histologic Appearance of Mucinous Carcinoma.
Islands of cancer cells (arrows) are floating in mucin. Note the abrupt margin with normal breast tissue (open arrow). (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
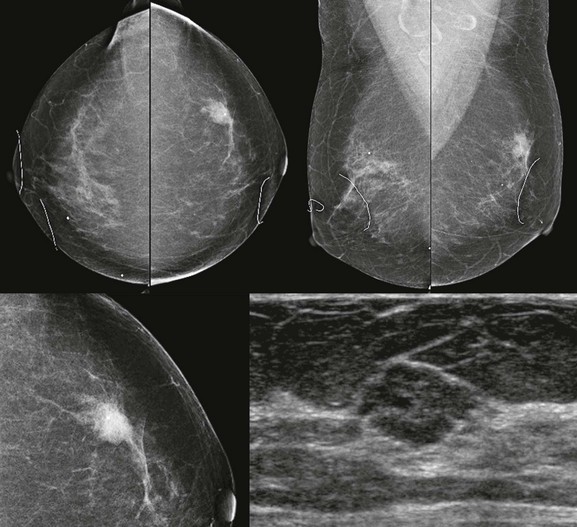
FIGURE 11-8 Mucinous Carcinoma.
A 67-year-old woman with a round mass with ill-defined margins in the left breast at 1 o’clock. On US, there is an isoechoic lobular mass with abrupt defined margins.
There may be a paucity of neoplastic cells in mucinous carcinoma. For this reason, a core biopsy containing mucin may represent either a benign mucocoele or mucinous carcinoma and should be excised even in the absence of neoplastic cells.
Papillary carcinoma represents 1% to 2% of breast cancers. Papillary carcinoma shares some similarities with benign papillomas. Both most commonly present as intraductal or intracystic masses (Fig. 11-9), may be associated with nipple discharge, and are frequently located in the subareolar region. Papillary carcinoma often presents as a palpable mass. On mammography, papillary carcinoma is usually a round or oval, circumscribed, equal- to high-density mass. On US, the most common finding is an intraductal or intracystic mass. These cancers are termed encapsulated (or intracystic) papillary carcinoma. Because the cancer is largely confined to the contents of the cyst, these lesions behave and are treated in a manner similar to DCIS. If the cancer invades through the wall of the cyst, the size and degree of invasion determine management and outcome. So, a woman with a 3-cm palpable intracystic papillary carcinoma may only have a 5-mm invasive component, and is treated and staged based on the 5-mm measurement. The prognosis for these tumors is excellent.
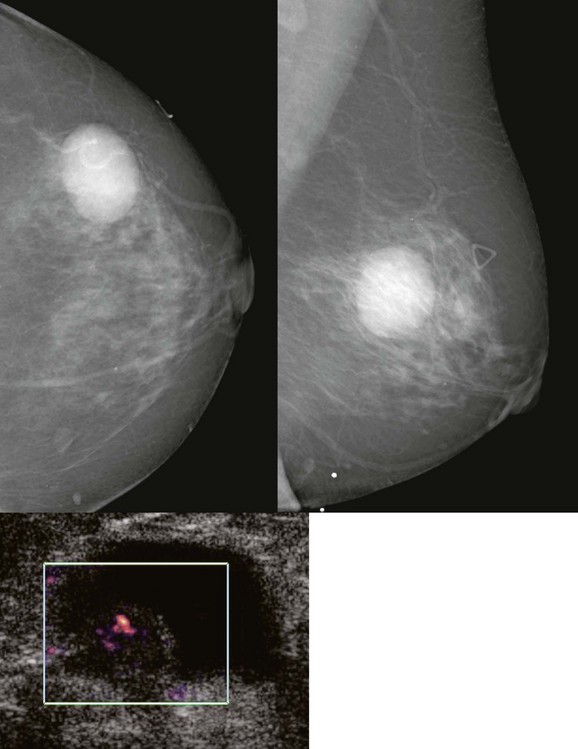
FIGURE 11-9 Intracystic Papillary Carcinoma.
This patient presented with a palpable lump that corresponds to an oval, circumscribed, high-density mass on mammography and a complex mass on US. At histologic examination, only a few millimeters of invasive tumor were present. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
Solid papillary carcinoma is a rare variant of DCIS that occurs in older women. Invasive papillary carcinoma that is not intracystic is extremely rare.
A Few Other Lesions
This section is for the detail-oriented people.
Invasive cribriform carcinoma, like tubular carcinoma, presents as a spiculated mass but has an excellent prognosis.
Invasive micropapillary carcinoma represents less than 2% of breast cancers and has no specific imaging or clinical features. About 70% have associated cribriform or micropapillary DCIS. About 75% of patients have metastatic adenopathy at diagnosis; this tumor has a poorer prognosis than the other ductal subtypes.
Invasive apocrine carcinoma is an uncommon lesion that has a similar mammographic appearance and prognosis as IDC-NOS.
Granular cell tumor is most commonly seen in the tongue, but may present as a subcutaneous spiculated mass in the breast (Fig. 11-10). It is more common in African-American women. These tumors are benign but may be locally aggressive, so they require wide local excision.
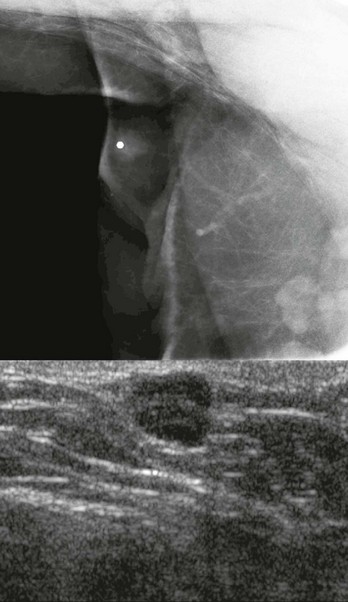
FIGURE 11-10 Granular Cell Tumor.
There is a small spiculated mass in a subcutaneous location. This would be an uncommon location for IDC-NOS. Wide excision is indicated.
Adenoid cystic carcinoma represents 0.1% of breast cancers. These tumors are typically seen in the salivary glands, so why in the breast? Remember way back to your embryology class? The breast and salivary glands are both modified sweat glands. Adenoid cystic carcinomas are most common in older women and present as a mammographic or palpable mass that is relatively well circumscribed (Fig. 11-11). These patients have an excellent prognosis; axillary metastasis is rare.
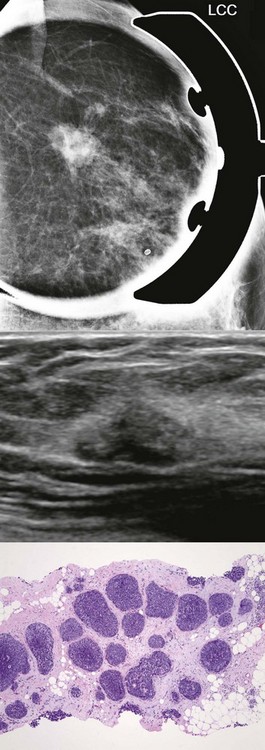
FIGURE 11-11 Adenoid Cystic Carcinoma.
This 46-year-old woman’s screening detected an oval mass that has mixed echogenicity by US. Core biopsy (hematoxylin-eosin) showed solid nests of tumor cells that lack a myoepithelial cell layer.
Metaplastic carcinoma (carcinosarcoma) contains both epithelial (carcinoma) and sarcomatous elements. The nonfibroglandular component may be epithelial (e.g., squamous) or mesenchymal (e.g., chondroid, spindle cell, osseous). These lesions typically grow rapidly and present as a palpable mass. On imaging, the appearance is nonspecific, though they often present as a mass with partly circumscribed and partly spiculated margins without calcifications (Fig. 11-12). Prognosis is similar to that for other high-grade invasive breast carcinomas. Spindle cell carcinoma is a subtype of metaplastic carcinoma.
The Fibroepithelial Lesions: Phyllodes to Fibroadenoma (Box 11-6)
Phyllodes tumor is a very rapidly growing tumor that typically occurs in middle-aged or older women. It usually presents as a very large but defined oval mass (Fig. 11-13). Phyllodes tumor is classified with a group of masses known as fibroepithelial lesions that originate from the lobule. The old name for phyllodes tumor is “cystosarcoma phyllodes,” which hints at the behavior of this tumor. Like all sarcomas, wide excision is the primary and most effective treatment. Even low-grade phyllodes tumors can be locally aggressive. High-grade phyllodes tumor can metastasize hematogenously, most frequently to the lungs. Sentinel lymph node biopsy is not indicated for phyllodes tumor because lymphatic metastasis is rare. These tumors can undergo sarcomatous degeneration, in which cartilage or bone can be seen within the tumor. The prognosis is then poor. Spindle cell sarcoma in the breast most commonly represents a subtype of phyllodes tumor.
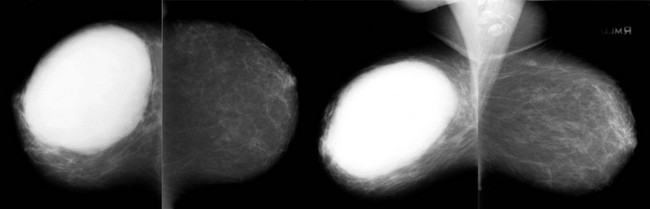
FIGURE 11-13 Phyllodes Tumor.
Large rapidly growing mass in a 56-year-old woman. The mass is oval and relatively circumscribed. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
Benign fibroadenomas are also fibroepithelial lesions. They are the most common breast lesion, occurring in 25% of asymptomatic women. You may run across certain descriptive pathologic terms for fibroadenomas, including pericanalicular, intracanalicular, and complex. These descriptions refer to growth patterns of fibroadenomas that contain cysts, adenosis, or other hyperplastic changes. The bottom line is that all of these histologic subtypes are managed similarly.
When small, phyllodes tumor can mimic a fibroadenoma on imaging (Table 11-1). Phyllodes tumors tend to arise in older women and to be faster growing than fibroadenomas. Ill-defined margins and internal cystic spaces on imaging are also suggestive of phyllodes tumor.
TABLE 11-1
Differentiation between Fibroadenoma and Phyllodes Tumor
| FEATURE | FIBROADENOMA | PHYLLODES TUMOR |
| Patient age at diagnosis | Young | Middle age or older |
| Size | 1-3 cm | Very large |
| Growth | Mild or none | Very rapid |
| Margins | Circumscribed | Ill-defined |
| Ultrasound findings | Internal septations | Cleft-like spaces |
On histologic examination, phyllodes tumor is very similar to fibroadenoma, but with considerably more proliferation. They are two ends of the same spectrum of disease. Phyllodes means “leaf,” and the histologic appearance of the tumor is characterized by leaf-like growth of cells. It may be difficult to differentiate a proliferative fibroadenoma from a low-grade phyllodes tumor, and there are no special stains to tell the two apart. Occasionally, a core biopsy result may read “proliferative fibroadenoma versus phyllodes,” and excision is then needed to make the diagnosis.
Juvenile fibroadenoma is a variant of fibroadenoma that typically presents in adolescents and young women as a rapidly growing and often very large mass. When very large (over 5-10 cm), they are referred to as giant fibroadenomas. Histologically, these lesions show hypercellular stromal proliferation. A very large mass in a young woman typically represents a giant fibroadenoma. Because phyllodes tumor and giant fibroadenoma both grow rapidly, patient age can usually suggest the correct diagnosis. Both are usually managed by excision.
Tubular adenoma and lactating adenoma are also fibroepithelial lesions with histologic and imaging features similar to those of fibroadenoma, but with a paucity of stroma and a greater epithelial component at histologic examination. Lactating adenomas present in women who are pregnant or lactating. Some pathologists believe that these are literally fibroadenomas on steroids—that they may represent preexisting fibroadenomas that are stimulated by the hormones of pregnancy and lactation. Tubular adenomas and lactating adenomas are managed the same as run-of-the-mill fibroadenomas.
Ductal adenoma and pleomorphic adenoma are actually considered variants of intraductal papilloma rather than fibroadenoma. Nevertheless, they are benign and can be managed like fibroadenomas.
Breast Sarcomas
Yes, these neoplasms are rare, but there are some cases in which you really should think about them, so read on!
All of the breast sarcomas are more common after radiation therapy to the breast. Sarcomas in general are characterized by rapid growth, hematogenous metastasis most frequently to the lungs, and poor prognosis. Lymphatic metastasis is rare, so sentinel lymph node sampling is not typically performed.
The primary treatment for sarcoma is wide local excision. The surgeon will often try to have a 1 cm margin around the lesion. Radiation therapy and chemotherapy are less effective than for carcinomas.
Angiosarcoma is the most common breast sarcoma. In the irradiated breast, cutaneous presentation (Fig. 11-14) is more common than within the breast parenchyma (Fig. 11-15). A diagnosis of cutaneous angiosarcoma should be considered for women with prior lumpectomy treated with radiation therapy who present with apparent breast bruising but no recallable trauma.
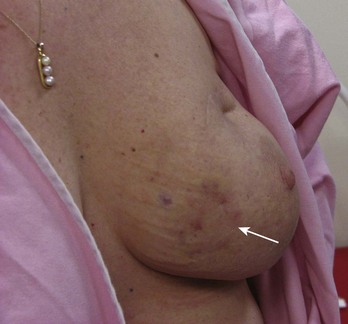
FIGURE 11-14 Cutaneous Angiosarcoma.
This 57-year-old woman had lumpectomy with radiation therapy 7 years ago and now notes bruising of her breast (arrow), though she does not recall trauma.
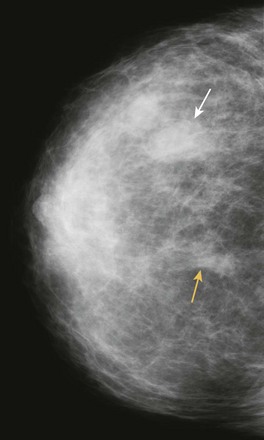
FIGURE 11-15 Parenchymal Angiosarcoma.
This 54-year-old woman developed a new mass (white arrow) in a different location than her prior lumpectomy scar (yellow arrow) from 10 years earlier. Clinically, she presented with a warm, red breast that was suspicious for inflammatory recurrence. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
Pseudoangiomatous stromal hyperplasia (PASH) is a benign myofibroblastic proliferation that can mimic angiosarcoma at histologic examination (hence, the pseudoangiomatous designation). PASH is uncommon and most frequently presents as an asymmetry rather than a mass or calcifications. A core biopsy showing PASH is benign, so excision is not indicated if the imaging findings are concordant.
Osteosarcoma presents as a really strange looking calcification almost anywhere in the body (Fig. 11-16). The primary and metastatic lesions may show increased activity on bone scintigraphy.
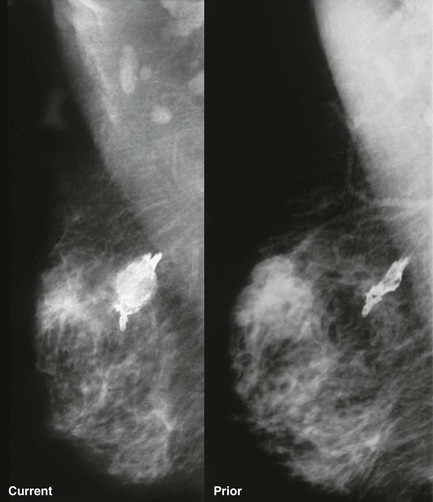
FIGURE 11-16 Osteosarcoma of the Breast.
The calcification appears lucent-centered, although not typical for dystrophic calcification. It increased in size and became palpable since the mammogram from 6 months earlier. (Reprinted from Harvey JA, Fondreist JT, Smith MM. Densely calcified breast mass. Invest Radiol 1994;29:516-517.)
Sarcomas other than angiosarcoma and osteosarcoma are rare and typically associated with phyllodes tumor.
Metastasis to the Breast
Metastatic lesions in the breast are usually a late finding of malignancy. They typically present as multiple round bilateral breast masses (Fig. 11-17), and may also be present in the subcutaneous tissues of the axilla. The mass margins are usually ill-defined compared with those of multiple cysts or fibroadenomas. Metastasis can also present as axillary or intramammary adenopathy (Fig. 11-18).
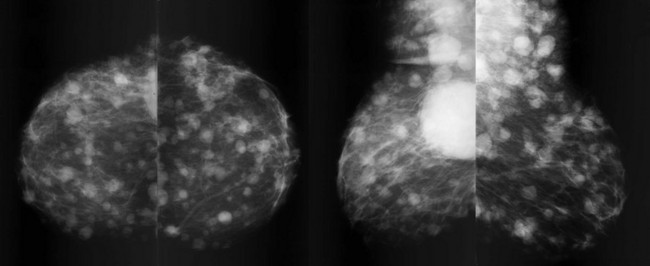
FIGURE 11-17 Metastatic Lung Carcinoma.
Multiple bilateral masses are present, including in the subcutaneous fat of the axilla. The margins of the masses are ill-defined. This mammographic appearance should not be confused with multiple benign masses, which are discussed in Chapter 8, Multiple Masses. (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
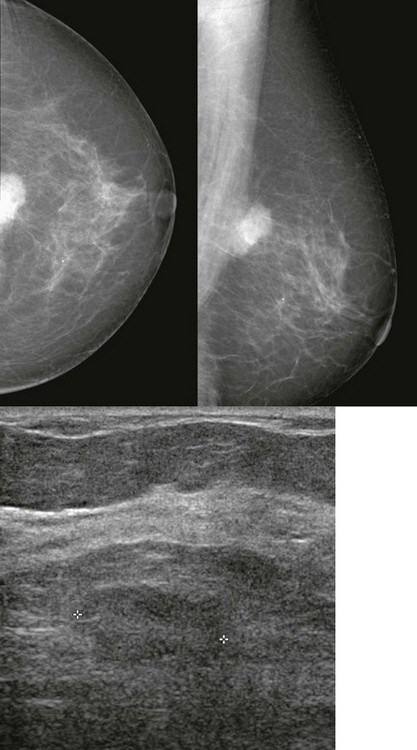
FIGURE 11-18 Metastatic Intramammary Lymph Node.
Although the mammographic appearance is nonspecific, the location and US appearance are consistent with an abnormal intramammary lymph node. Diagnosis: metastatic melanoma.
Melanoma is the most common nonbreast primary neoplasm to metastasize to the breast (Box 11-7).
Non-Hodgkin lymphoma (NHL) rarely involves the breast. Breast involvement usually occurs in women with known extramammary disease. NHL of the breast most commonly presents as a solitary mass, and multiple masses are less common. Primary breast lymphoma is very rare (Fig. 11-19).
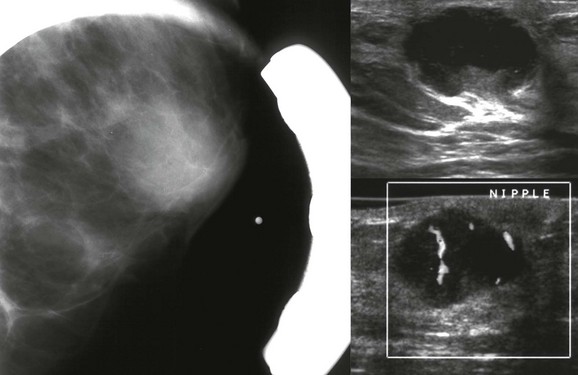
FIGURE 11-19 Primary Breast Lymphoma.
A middle-aged woman presents with a palpable retroareolar mass. Mammography shows an obscured mass that is markedly hypoechoic and vascular by US. Core biopsy revealed non-Hodgkin lymphoma (diffuse large B cell). There was no evidence of extramammary disease and the patient was treated with chemotherapy and radiation therapy.
Rhabdomysarcoma and leukemic infiltrates can present as palpable thickening with asymmetry on mammography and poorly defined areas of hyperechogenic breast tissue on US (Fig. 11-20). Fat necrosis differs in appearance; it is typically subcutaneous and better defined.
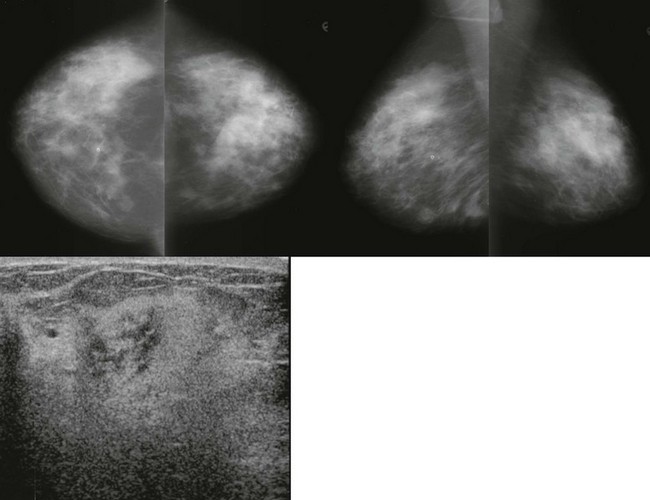
FIGURE 11-20 Leukemia of the Breast.
A 28-year-old woman with bilateral palpable breast masses. The mammogram shows bilateral irregular masses. On US, the palpable lumps correspond to areas where the echotexture is very heterogeneous. In some regions, the finding is very hyperechoic. The patient has a history of leukemia, thought to be in remission. Unfortunately, this represents recurrence.
Anderson, WF, Chu, KC, Chang, S, Sherman, ME. Comparison of age-specific incidence rate patterns for different histopathologic types of breast carcinoma. Cancer Epidemiol Biomarkers Prev. 2004;13:1128–1135.
Harvey, JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology. 2007;242:683–694.
Harvey, JA, Fechner, RE, Moore, MM. Apparent ipsilateral decrease in breast size on mammography: A sign of infiltrating lobular carcinoma. Radiology. 2000;213:883–889.
Harvey, JA, Fondreist, JT, Smith, MM. Densely calcified breast mass. Invest Radiol. 1994;29:516–517.
Kawashima, M, Tamaki, Y, Nonaka, T, et al. MR imaging of mucinous carcinoma of the breast. Am J Roentgenol. 2002;179(1):179–183.
Lagrange, JL, Ramaioli, A, Chateau, MC, et al. Sarcoma after radiation therapy: Retrospective multiinstitutional study of 80 histologically confirmed cases. Radiology. 2000;216(1):197–205.
Lam, WWM, Chu, WCW, Tse, GM, Ma, TK. Sonographic appearance of mucinous carcinoma of the breast. Am J Roentgenol. 2004;182(4):1069–1074.
Livi, L, Paiar, F, Meldolesi, E, et al. Tubular carcinoma of the breast: Outcome and loco-regional recurrence in 307 patients. Eur J Surg Oncol. 2005;31(1):9–12.
Lopez, JK, Bassett, LW. Invasive lobular carcinoma of the breast: Spectrum of mammographic, US, and MR imaging findings. Radiographics. 2009;29:165–176.
McGhan, L, Wasif, N, Gray, R, et al. Use of preoperative magnetic resonance imaging for invasive lobular cancer: Good, better, but maybe not the best? Ann Surg Oncol. 2010;17(0):255–262.
Mercado, CL, Hamele-Bena, D, Oken, SM, et al. Papillary lesions of the breast at percutaneous core-needle biopsy. [see comment]. Radiology. 2006;238(3):801–808.
Mylonas, I, Janni, W, Friese, K, Gerber, B. Unexpected metastatic lobular carcinoma of the breast with intraabdominal spread and subsequent port-site metastasis after diagnostic laparoscopy for exclusion of ovarian cancer. Gynecol Oncol. 2004;95(2):405–408.
Sabate, JM, Gomez, A, Torrubia, S, Flotats, A. Osteosarcoma of the breast. Am J Roentgenol. 2002;179(1):277–278.
Schnitt, SJ, Collins, LC. Biopsy Interpretation of the Breast. Philadelphia: Lippincott Williams & Wilkins; 2009.
Sheppard, DG, Whitman, GJ, Fornage, BD, et al. Tubular carcinoma of the breast: Mammographic and sonographic features. Am J Roentgenol. 2000;174(1):253–257.
Shin, HJ, Kim, HH, Kim, SM, et al. Papillary lesions of the breast diagnosed at percutaneous sonographically guided biopsy: Comparison of sonographic features and biopsy methods. Am J Roentgenol. 2008;190(3):630–636.
Sinclair, DS, Olsen, J, Spigos, DG, Freedy, L. Phyllodes (phylloides or cystosarcoma phyllodes) tumor: Wide local excision is the preferred method of treatment. Am J Roentgenol. 2000;175(3):859–861.
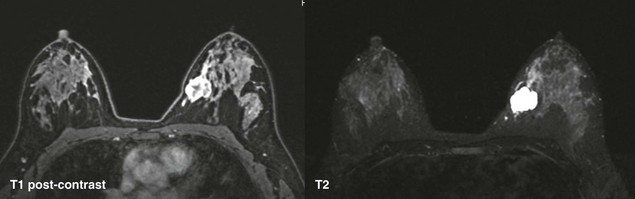
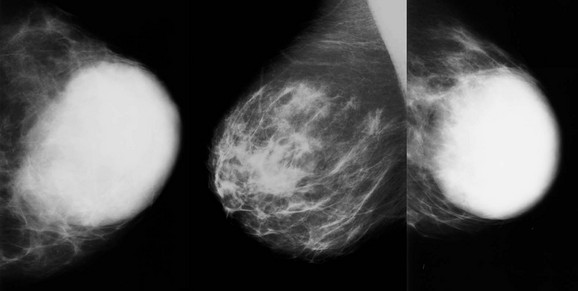
CASE 11-1. A 62-year-old woman with screening MRI. What is the most likely diagnosis of the left breast mass?
CASE 11-2. A 47-year-old woman for screening. What are your BI-RADS assessment and differential diagnosis for the mass in the right breast at 10 o’clock? (Reprinted with permission from Harvey JA. Unusual breast cancers: Useful clues to expanding the differential diagnosis. Radiology 2007;242:683-694.)
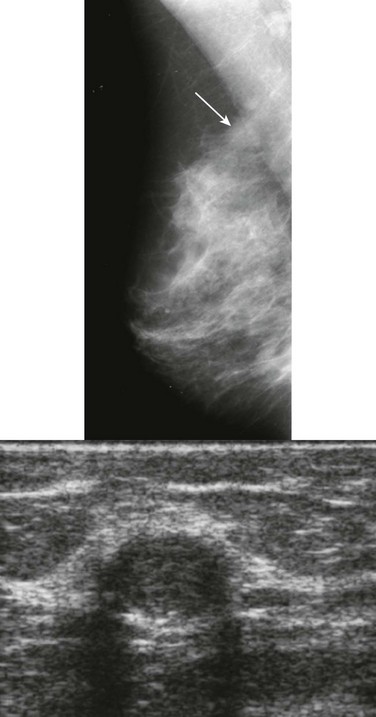
CASE 11-3. A 58-year-old woman with rapidly growing mass noted about 6 months earlier. What might you consider in the differential diagnosis?
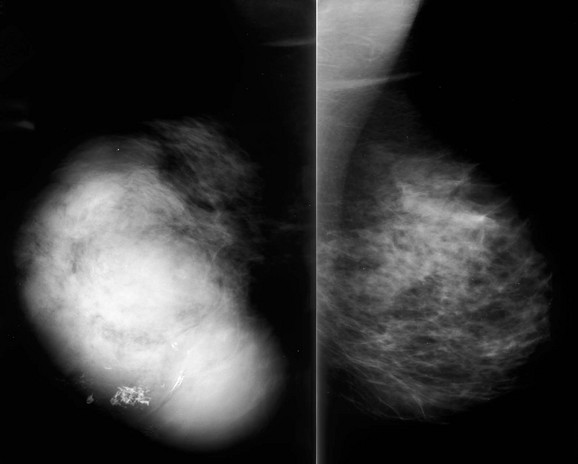
CASE 11-4. A 22-year-old woman presents with a rapidly growing mass in the left breast. What is the most likely diagnosis?
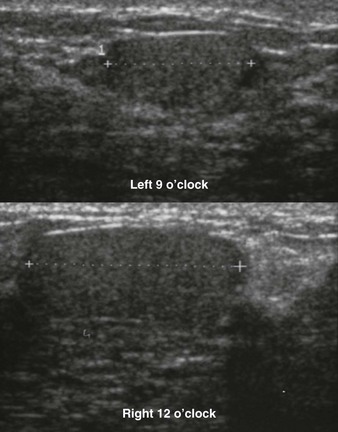
CASE 11-5. A 20-year-old woman who is 8 months pregnant presents for evaluation of several palpable lumps in both breasts. What is the most likely diagnosis given her US below? The other palpable lumps all looked similar. How would you manage her?
CASE 11-6. Ultrasound-guided core biopsy of the mass below (arrow) shows tubular adenoma. What is your recommendation given the imaging findings and result?
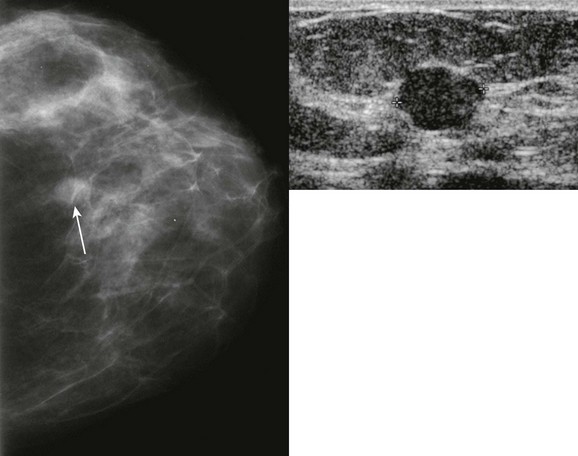
CASE 11-7. A 64-year-old woman with a history of left mastectomy is recalled from screening for diagnostic mammography and US. Which subtype of invasive ductal carcinoma is the most likely diagnosis?
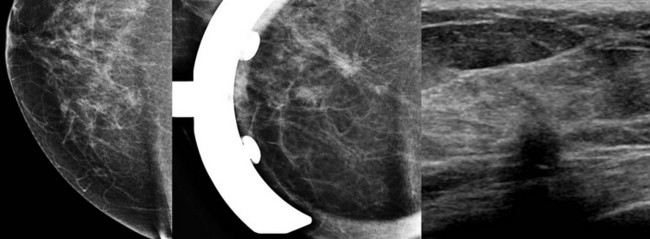
CASE 11-8. A 73-year-old woman presents with a palpable mass in her right breast. What is the most likely diagnosis based on the diagnostic mammogram and US?
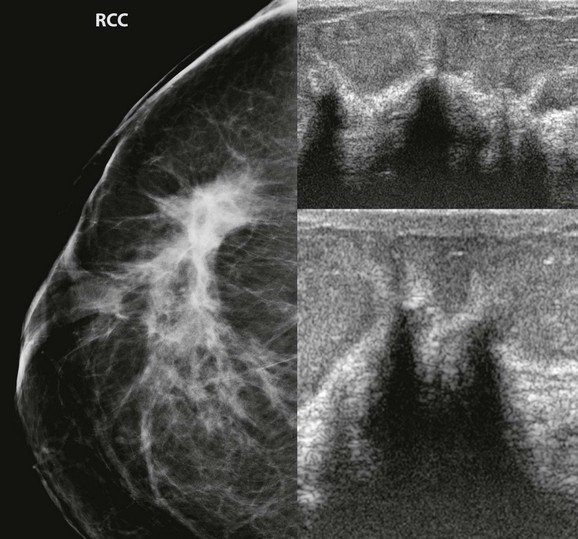
CASE 11-9. This 69-year-old woman was recalled from screening to evaluate a developing asymmetry (arrow). US was negative. Stereotactic biopsy shows pseudoangiomatous stromal hyperplasia. What do you recommend?
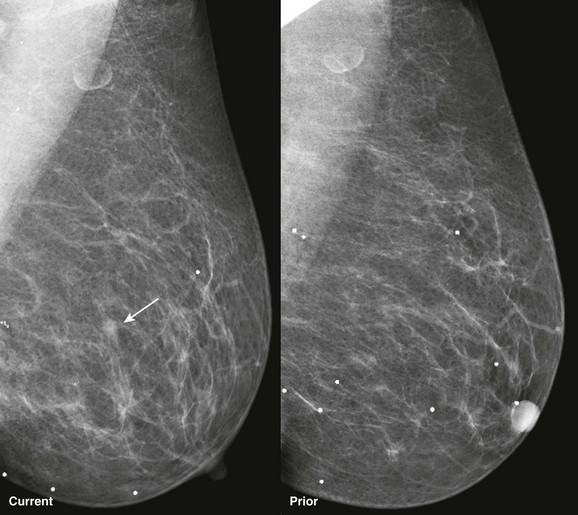
CASE 11-10. A 31-year-old woman at high risk due to family history of breast cancer has a suspicious enhancing mass on screening MRI. It is hyperintense on T2. The MRI-guided biopsy shows stromal fibrosis and focal mucin deposition. What is your recommendation for this biopsy result?
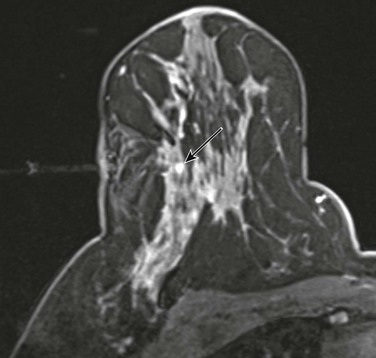
CASE 11-11. This 41-year-old woman is recalled due to architectural distortion (arrows) on her baseline screening mammogram. A corresponding mass is identified on US (arrows). What is your differential diagnosis? Ultrasound-guided core biopsy shows florid hyperplasia without atypia. What is your recommendation?
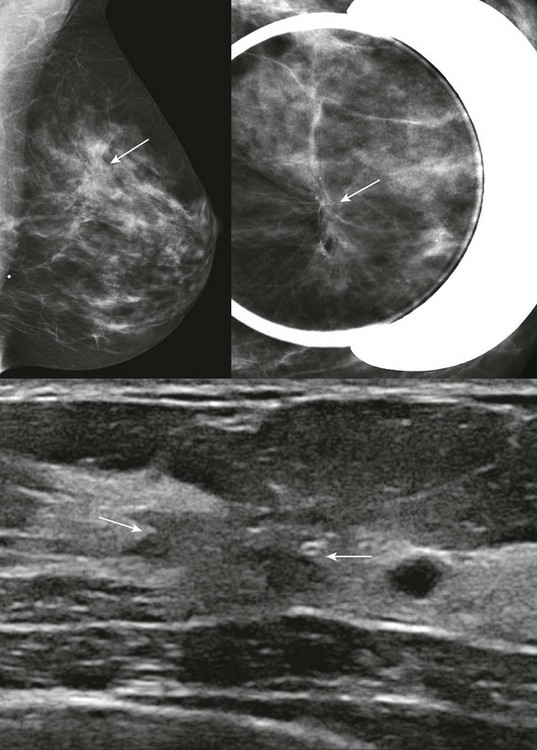
CASE 11-12. This 52-year-old woman has a very firm palpable lump measuring about 5 cm in the lower inner quadrant of her right breast (marked by the triangle). Below are her diagnostic mammogram and US. What are the findings? What is your differential diagnosis? BI-RADS assessment?
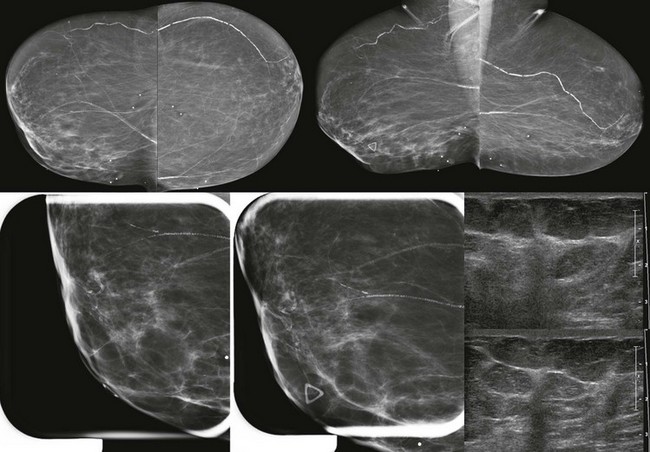
CASE 11-1. This is suspicious for a mucinous carcinoma. The mass is hyperintense on T2, but has heterogeneous enhancement and ill-defined margins. This should not be mistaken for a fibroadenoma.
CASE 11-2. This mass resembles a fibroadenoma, but the margins are ill-defined, and there are some internal cleft-like cystic spaces. This is concerning for phyllodes tumor, BI-RADS 4. USCNB confirmed the diagnosis. Although phyllodes tumor typically presents as a rapidly growing palpable mass in a middle-aged or older woman, these tumors may occasionally be detected at screening.
CASE 11-3. A rapidly growing mass should always be concerning for either a very poorly differentiated IDC, carcinosarcoma, or phyllodes tumor. Did you notice the coarse calcifications? The diagnosis in this case is phyllodes tumor with osteosarcomatous and chondrosarcomatous degeneration. The prognosis is poor when sarcomatous elements are present within a phyllodes tumor.
CASE 11-4. A large, rapidly growing, round or oval mass in a middle-aged or older woman would be most concerning for phyllodes tumor. However, this is a young woman, and giant fibroadenoma—which this represents—would be most likely.
CASE 11-5. The masses are all oval, circumscribed, and homogeneously hypoechoic with a parallel orientation. The findings suggest lactating adenomas.
Biopsy is tricky here. Doing an ultrasound-guided core biopsy may result in a milk fistula this late in pregnancy. A fine-needle aspiration may be reasonable with a smaller risk of fistula. The surgeons will not want to excise anything until after she delivers her baby.
If these were multiple circumscribed masses on mammography and all similar in appearance, you would most likely give her BI-RADS 2. Instead of biopsy, we elected to see her back in 4 months; she was 3 months post partum and did not choose to breastfeed her infant. The masses decreased considerably in size, consistent with lactating adenomas. No further evaluation was needed.
CASE 11-6. The biopsy result of tubular adenoma is concordant. Tubular adenomas can be managed similarly to fibroadenomas. A follow-up mammogram or US can be performed in 6 to 12 months. Excision is not indicated.
CASE 11-7. Among the major subtypes of IDC, tubular carcinoma is most likely to appear as a small spiculated mass and is the diagnosis in this case. There was DCIS associated with the invasive tumor.
CASE 11-8. Mammography shows a large area of architectural distortion with skin retraction. US reveals extensive shadowing at the edge of the fibroglandular tissue. Diagnosis: ILC.
CASE 11-9. Pseudoangiomatous stromal hyperplasia (PASH) is a concordant result for this developing asymmetry. A mammogram in 6 months is reasonable to ensure stability.
CASE 11-10. The finding of mucin on a core biopsy may be due to a benign mucoceole, but may also be due to mucinous carcinoma. Excision should be recommended. Diagnosis: mucinous carcinoma.
CASE 11-11. There is persistent architectural distortion without a central mass on mammography (“the dark star”). On US, there is an irregular hypoechoic mass with spiculated margins. The echogenicity of the mass on US is higher than that of most breast cancers, and there is no echogenic margin. The appearance on mammography and US suggests that this may represent a radial scar. ILC, IDC-NOS, or tubular carcinoma is also possible. The patient has no history of prior breast surgery, so this is not a surgical scar. Excisional biopsy showed radial scar/complex sclerosing lesion. Core biopsy often does not result in a diagnosis of radial scar because a larger sample is often needed to display the diagnostic morphologic appearance of this lesion. A benign result should be considered discordant, and likely to be a radial scar.
CASE 11-12. There is mild asymmetry in the area of the palpable region in the right breast. There is no distortion or discrete mass on mammography. On US, there are areas of shadowing with sonographic distortion. The presence of a clinically suspicious mass with asymmetry and sonographic shadowing suggests the diagnosis of ILC, which was the diagnosis. Congratulations if you were thinking ILC. Good job!