Chapter 139 Atopic Dermatitis (Atopic Eczema)
Atopic dermatitis (AD), or eczema, is the most common chronic relapsing skin disease seen in infancy and childhood. It affects 10-30% of children worldwide and frequently occurs in families with other atopic diseases, such as asthma, allergic rhinitis, and food allergy. Infants with AD are predisposed to development of allergic rhinitis and/or asthma later in childhood, a process called “the atopic march.”
Etiology
AD is a complex genetic disorder that results in a defective skin barrier, reduced skin innate immune responses, and exaggerated T-cell responses to environmental allergens and microbes that lead to chronic skin inflammation.
Pathology
Acute AD skin lesions are characterized by spongiosis, or marked intercellular edema, of the epidermis. In AD, dendritic antigen-presenting cells (APCs) in the epidermis, such as Langerhans cells (LCs), exhibit surface-bound immunoglobulin (Ig) E molecules. These APCs play an important role in cutaneous allergen presentation to T helper type 2 (Th2) cells (Chapter 134). There is a marked perivenular T-cell infiltrate with occasional monocyte-macrophages in acute AD lesions. Mast cells are found in normal numbers but in different stages of degranulation. Chronic, lichenified AD is characterized by a hyperplastic epidermis with hyperkeratosis, and minimal spongiosis. There are predominantly IgE-bearing LCs in the epidermis, and macrophages in the dermal mononuclear cell infiltrate. Mast cell and eosinophil numbers are increased. Eosinophils contribute to allergic inflammation by secreting cytokines and mediators that augment inflammatory responses and induce tissue injury in AD through the production of reactive oxygen intermediates and release of toxic granule proteins.
Pathogenesis
Two forms of AD have been identified. Atopic eczema is associated with IgE-mediated sensitization (at onset or during the course of eczema) and occurs in 70-80% of patients with AD. Nonatopic eczema is not associated with IgE-mediated sensitization and is seen in 20-30% of patients with AD. Both forms of AD are associated with eosinophilia. In atopic eczema, circulating T cells expressing the skin homing receptor cutaneous lymphocyte-associated antigen (CLA) produce increased levels of Th2 cytokines, including interleukin-4 (IL-4) and IL-13, which induce isotype switching to IgE synthesis. Another cytokine, IL-5, plays an important role in eosinophil development and survival. These CLA+ T cells also produce abnormally low levels of interferon-γ (IFN-γ), a Th1 cytokine known to inhibit Th2 cell function. Nonatopic eczema is associated with lower IL-4 and IL-13 production than is atopic eczema. Another Th2 cytokine, IL-31, induces marked pruritus in experimental animals.
Compared with the skin of healthy subjects, both unaffected skin and acute skin lesions of patients with AD have an increased number of cells expressing IL-4 and IL-13; however, acute AD does not involve significant numbers of cells that express IFN-γ or IL-12. Chronic AD skin lesions, by contrast, have significantly fewer cells that express IL-4 and IL-13 but increased numbers of cells that express IL-5, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-12, and IFN-γ than acute AD lesions. Thus, chronic AD, unlike acute AD, is characterized by a shift from a Th2-dominant to a Th1-dominant profile. The increased expression of IL-12 in eosinophils, inflammatory dendritic epidermal cells, and macrophages in chronic AD skin lesions may play a role in initiating this switch to Th1 cell development. Persistent skin inflammation may be associated with a relative lack of T-regulatory cells in the skin of AD subjects and increased expression of IL-17.
The development of AD skin lesions is orchestrated by local tissue expression of proinflammatory cytokines and chemokines. Cytokines, such as tumor necrosis factor-α (TNF-α) and IL-1 from keratinocytes, mast cells, and dendritic cells, bind to receptors on vascular endothelium. The ligand-receptor pair activates cellular signaling, including the NF-κB pathway, and induces expression of vascular endothelial cell adhesion molecules (VCAM). These events proceed from tethering, activation, and adhesion to the endothelium, followed by extravasation of inflammatory cells. Once the inflammatory cells infiltrate the tissue, they respond to chemotactic gradients established by chemokines, which are released at sites of injury or infection. Chemokines play a central role in defining the nature of the inflammatory infiltrate in AD. The chemotactic protein, CCL27, is highly upregulated in AD and preferentially attracts CLA+ T cells to the skin. Other C-C chemokines, RANTES (“regulated on activation, normal T expressed and secreted”), monocyte chemotactic protein-4 (MCP-4), and eotaxin are increased in AD skin lesions, resulting in chemotaxis of eosinophils, macrophages, and Th2 lymphocytes expressing their receptor (CCR3). Selective recruitment of CCR4-expressing Th2 cells into skin affected by AD may also be mediated by macrophage-derived chemokine (MDC) and TARC (expand), which are increased in AD. Elevated IL-5 and GM-CSF in chronic AD may lead to enhanced survival of eosinophils and monocyte-macrophages as well as LCs.
Research has identified the mechanisms leading to barrier dysfunction in AD. In healthy people, the skin acts as a protective barrier against external irritants, moisture loss, and infection. Proper function of the skin depends on adequate moisture and lipid content, functional immune responses, and structural integrity. Severely dry skin is a hallmark of AD. This is a result of compromise of the physical and chemical structures of the epidermal barrier, which leads to excess transepidermal water loss. Filaggrin, a component of the cytoskeleton, and its breakdown products are critical to skin barrier function. Genetic mutations in the filaggrin gene family have been identified in up to 50% of severe patients with AD. Such patients have increased risk of bacterial, viral, and fungal infection related to impairment of innate immunity, including a loss of barrier function and impaired generation of antimicrobial peptides.
Clinical Manifestations
AD typically begins in infancy. Approximately 50% of patients experience symptoms in the 1st year of life, and an additional 30% are diagnosed between 1 and 5 yr of age. Intense pruritus, especially at night, and cutaneous reactivity are the cardinal features of AD. Scratching and excoriation cause increased skin inflammation that contributes to the development of more pronounced eczematous skin lesions. Foods (cow milk, egg, peanut, tree nuts, soy, wheat, fish, shellfish), inhalant allergens, bacterial infection, reduced humidity, excessive sweating, and irritants (wool, acrylic, soaps, toiletries, fragrances, detergents) can exacerbate (trigger) pruritus and scratching.
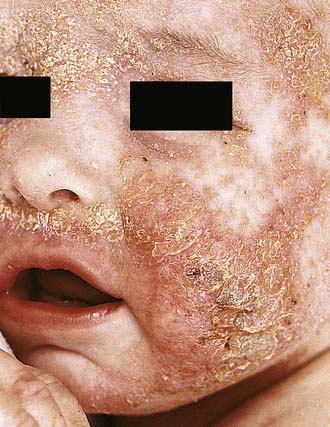
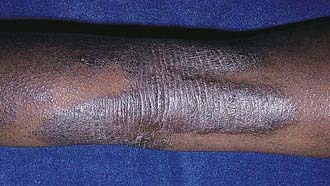
Acute AD skin lesions are intensely pruritic with erythematous papules (Figs. 139-1 and 139-2). Subacute dermatitis manifests as erythematous, excoriated, scaling papules. In contrast, chronic AD is characterized by lichenification (Fig. 139-3), or thickening of the skin with accentuated surface markings, and fibrotic papules (prurigo nodularis). In chronic AD, all three types of skin reactions may coexist in the same individual. Most patients with AD have dry, lackluster skin irrespective of their stage of illness. Skin reaction pattern and distribution vary with the patient’s age and disease activity. AD is generally more acute in infancy and involves the face, scalp, and extensor surfaces of the extremities. The diaper area is usually spared. Older children and children with chronic AD have lichenification and localization of the rash to the flexural folds of the extremities. AD often goes into remission as the patient grows older, leaving an adolescent or adult with skin prone to itching and inflammation when exposed to exogenous irritants.
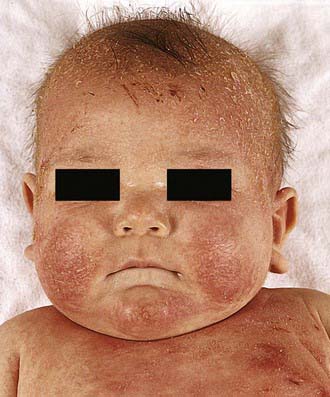
Figure 139-1 Atopic dermatitis, typical cheek involvement.
(From Eichenfield LF, Friedan IJ, Esterly NB: Textbook of neonatal dermatology, Philadelphia, 2001, WB Saunders, p 242.)
Laboratory Findings
There are no specific laboratory tests to diagnose AD. Many patients have peripheral blood eosinophilia and increased serum IgE levels. Serum IgE measurement or prick skin testing can identify the allergens to which patients are sensitized. The diagnosis of clinical allergy to these allergens requires confirmation by history and environmental challenges.
Diagnosis and Differential Diagnosis
AD is diagnosed on the basis of 3 major features: pruritus, an eczematous dermatitis that fits into a typical presentation, and a chronic or chronically relapsing course (Table 139-1). Associated features, such as a family history of asthma, hay fever, elevated IgE, and immediate skin test reactivity, are variably present.
Table 139-1 CLINICAL FEATURES OF ATOPIC DERMATITIS
MAJOR FEATURES
ASSOCIATED FEATURES
Many inflammatory skin diseases, immunodeficiencies, skin malignancies, genetic disorders, infectious diseases, and infestations share symptoms with AD and should be considered and excluded before a diagnosis of AD is established (Table 139-2). Severe combined immunodeficiency syndrome (Chapter 120.1) should be considered for infants presenting in the 1st year of life with diarrhea, failure to thrive, generalized scaling rash, and recurrent cutaneous and/or systemic infection. Histiocytosis (Chapter 501) should be excluded in any infant with AD and failure to thrive. Wiskott-Aldrich syndrome (Chapter 120.2), an X-linked recessive disorder associated with thrombocytopenia, immune defects, and recurrent severe bacterial infections, is characterized by a rash almost indistinguishable from that in AD. Hyper-IgE syndrome (Chapter 120.2) is characterized by markedly elevated serum IgE values, recurrent deep-seated bacterial infections, chronic dermatitis, and refractory dermatophytosis.
Table 139-2 DIFFERENTIAL DIAGNOSIS OF ATOPIC DERMATITIS
CONGENITAL DISORDERS
CHRONIC DERMATOSES
INFECTIONS AND INFESTATIONS
MALIGNANCIES
AUTOIMMUNE DISORDERS
IMMUNODEFICIENCIES
METABOLIC DISORDERS
From Leung DYM, Sampson HA, Geha RS, et al: Pediatric allergy principles and practice, St Louis, 2003, Mosby, p 562.
Adolescents who present with an eczematous dermatitis but no history of childhood eczema, respiratory allergy, or atopic family history may have allergic contact dermatitis (Chapter 647). A contact allergen may be the problem in any patient whose AD does not respond to appropriate therapy. Sensitizing chemicals, such as parabens and lanolin, can be irritants for patients with AD and are commonly found as vehicles in therapeutic topical agents. Topical glucocorticoid contact allergy has been reported in patients with chronic dermatitis who are undergoing topical corticosteroid therapy. Eczematous dermatitis has also been reported with HIV infection as well as with a variety of infestations such as scabies. Other conditions that can be confused with AD include psoriasis, ichthyoses, and seborrheic dermatitis.
Treatment
The treatment of AD requires a systematic, multifaceted approach that incorporates skin hydration, topical anti-inflammatory therapy, identification and elimination of flare factors, and, if necessary, systemic therapy. Assessment of the severity also helps direct therapy (Table 139-3).
Table 139-3 CATEGORIZATION OF PHYSICAL SEVERITY OF ATOPIC ECZEMA
From Lewis-Jones S, Mugglestone MA; Guideline Development Group: Management of atopic eczema in children aged up to 12 years: summary of NICE guidance, BMJ 335:1263–1264, 2007.
Cutaneous Hydration
Because patients with AD have impaired skin barrier function from reduced lipid levels, they present with diffuse, abnormally dry skin, or xerosis. Lukewarm soaking baths for 15-20 min followed by the application of an occlusive emollient to retain moisture provide symptomatic relief. Hydrophilic ointments of varying degrees of viscosity can be used according to the patient’s preference. Occlusive ointments are sometimes not well tolerated because of interference with the function of the eccrine sweat ducts and may induce the development of folliculitis. In these cases, less occlusive agents should be used.
Hydration by baths or wet dressings promotes transepidermal penetration of topical glucocorticoids. Dressings may also serve as effective barriers against persistent scratching, in turn promoting healing of excoriated lesions. Wet dressings are recommended for use on severely affected or chronically involved areas of dermatitis refractory to skin care. It is critical that wet dressing therapy be followed by topical emollient application to avoid potential drying and fissuring from the therapy. Wet dressing therapy can be complicated by maceration and secondary infection and should be closely monitored by a physician.
Topical Corticosteroids
Topical corticosteroids are the cornerstone of anti-inflammatory treatment for acute exacerbations of AD. Patients should be carefully instructed on their use of topical glucocorticoids in order to avoid potential adverse effects. There are 7 classes of topical glucocorticoids, ranked according to their potency as determined by vasoconstrictor assays (Table 139-4). Because of their potential adverse effects, the ultra-high-potency glucocorticoids should not be used on the face or intertriginous areas and should be used only for very short periods on the trunk and extremities. Mid-potency glucocorticoids can be used for longer periods to treat chronic AD involving the trunk and extremities. Long-term control can be maintained with twice-weekly applications of topical fluticasone or mometasone to areas that have healed but are prone to relapse, once control of AD is achieved after a daily regimen of topical corticosteroids. Compared with creams, ointments have a greater potential to occlude the epidermis, resulting in enhanced systemic absorption. Adverse effects of topical glucocorticoids can be divided into local adverse effects and systemic adverse effects, the latter of which result from suppression of the hypothalamic-pituitary-adrenal axis. Local adverse effects include the development of striae and skin atrophy. Systemic adverse effects are related to the potency of the topical corticosteroid, site of application, occlusiveness of the preparation, percentage of the body surface area covered, and length of use. The potential for adrenal suppression from potent topical corticosteroids is greatest in infants and young children with severe AD requiring intensive therapy.
Table 139-4 SELECTED TOPICAL CORTICOSTEROID PREPARATIONS*
GROUP 1
GROUP 2
GROUP 3
GROUP 4
GROUP 5
GROUP 6
GROUP 7
Hydrocortisone (Hytone) 2.5% & 1% ointment/cream
* Representative corticosteroids are listed by group from 1 (superpotent) through 7 (least potent).
Adapted from Stoughton RB: Vasoconstrictor assay-specific applications. In Malbach HI, Surber C, editors: Topical corticosteroids, Basel, Switzerland, 1992, Karger, pp 42–53.
Topical Calcineurin Inhibitors
The nonsteroidal topical calcineurin inhibitors are effective in reducing AD skin inflammation. Pimecrolimus cream 1% (Elidel) is indicated for mild to moderate AD. Tacrolimus ointment 0.1% and 0.03% (Protopic) is indicated for moderate to severe AD. Both are approved for short-term or intermittent long-term treatment of AD in patients ≥2 yr whose disease is unresponsive to or who are intolerant of other conventional therapies or for whom these therapies are inadvisable owing to potential risks. Topical calcineurin inhibitors may be better than topical corticosteroids in the treatment of patients whose AD is poorly responsive to topical steroids, of patients with steroid phobia, and of patients with face and neck dermatitis, in which ineffective, low-potency topical corticosteroids are usually used because of fears of steroid-induced skin atrophy.
Tar Preparations
Coal tar preparations have antipruritic and anti-inflammatory effects on the skin; however, the anti-inflammatory effects are usually not as pronounced as those of topical glucocorticoids or calcineurin inhibitors. Tar preparations are useful in reducing the potency of topical glucocorticoids required in long-term maintenance therapy of AD. Tar shampoos can be particularly beneficial for scalp dermatitis. Adverse effects associated with tar preparations include skin irritation, folliculitis, and photosensitivity.
Antihistamines
Systemic antihistamines act primarily by blocking the histamine H1 receptors in the dermis, thereby reducing histamine-induced pruritus. Histamine is only one of many mediators that induce pruritus of the skin, so patients may derive minimal benefit from antihistaminic therapy. Because pruritus is usually worse at night, sedating antihistamines (hydroxyzine, diphenhydramine) may offer an advantage with their soporific side effects when used at bedtime. Doxepin hydrochloride has both tricyclic antidepressant and H1- and H2-receptor blocking effects. Short-term use of a sedative to allow adequate rest may be appropriate in cases of severe nocturnal pruritus. Studies of newer nonsedating antihistamines have shown variable effectiveness in controlling pruritus in AD, although they may be useful in the small subset of patients with AD and concomitant urticaria.
Systemic Corticosteroids
Systemic corticosteroids are rarely indicated in the treatment of chronic AD. The dramatic clinical improvement that may occur with systemic corticosteroids is frequently associated with a severe rebound flare of AD after therapy discontinuation. Short courses of oral corticosteroids may be appropriate for an acute exacerbation of AD while other treatment measures are being instituted in parallel. If a short course of oral corticosteroids is given, it is important to taper the dosage and begin intensified skin care, particularly with topical corticosteroids and frequent bathing followed by application of emollients, to prevent rebound flaring of AD.
Cyclosporine
Cyclosporine is a potent immunosuppressive drug that acts primarily on T cells by suppressing cytokine gene transcription. Cyclosporine forms a complex with an intracellular protein, cyclophilin, and this complex in turn inhibits calcineurin, a phosphatase required for activation of NFAT (nuclear factor of activated T cells), a transcription factor necessary for cytokine gene transcription. Cyclosporine (5 mg/kg/day) for short-term and long-term (1 yr) use has been beneficial for children with severe, refractory AD. Possible adverse effects include renal impairment and hypertension.
Phototherapy
Natural sunlight is often beneficial to patients with AD as long as sunburn and excessive sweating are avoided. Many phototherapy modalities are effective for AD, including ultraviolet A-1, ultraviolet B (UVB), narrow-band UVB, and psoralen plus ultraviolet A (PUVA). Phototherapy is generally reserved for patients in whom standard treatments fail. Maintenance treatments are usually required for phototherapy to be effective. Short-term adverse effects with phototherapy include erythema, skin pain, pruritus, and pigmentation. Long-term adverse effects include predisposition to cutaneous malignancies.
Unproven Therapies
Other therapies that may be considered in patients with refractory AD are as follows.
Interferon-γ
IFN-γ is known to suppress Th2-cell function. Several studies, including a multicenter, double-blind, placebo-controlled trial and several open trials, have demonstrated that treatment with recombinant human IFN-γ results in clinical improvement of AD. Reduction in clinical severity of AD correlated with the ability of IFN-γ to decrease total circulating eosinophil counts. Influenza-like symptoms are commonly observed side effects during the treatment course.
Omalizumab
Treatment of patients who have severe AD and elevated serum IgE values with monoclonal anti-IgE may be considered in those with allergen-induced flares of AD. However, there have been no published double-blind, placebo-controlled trials of its use. Most reports have been case studies and show inconsistent responses to anti-IgE.
Allergen Immunotherapy
In contrast to its acceptance for treatment of allergic rhinitis and extrinsic asthma, immunotherapy with aeroallergens in the treatment of AD is controversial. There are reports of both disease exacerbation and improvement. Studies suggest specific immunotherapy in patients with AD sensitized to dust mite allergen showed improvement in severity of skin disease as well as reduction in topical steroid use. However, well-controlled studies are still required to determine the role for immunotherapy with this disease, particularly in children.
Probiotics
Perinatal administration of the probiotic Lactobacillus rhamnosus strain GG has been shown to reduce the incidence of AD in at-risk children during the first 2 yr of life. The treatment response has been found to be more pronounced in patients with positive skin prick test results and elevated IgE values. Other studies have not demonstrated a benefit.
Chinese Herbal Medications
Several placebo-controlled clinical trials have suggested that patients with severe AD may benefit from treatment with traditional Chinese herbal therapy. The subjects had significantly reduced skin disease and decreased pruritus. The beneficial response of Chinese herbal therapy is often temporary, and effectiveness may wear off despite continued treatment. The possibility of hepatic toxicity, cardiac side effects, or idiosyncratic reactions remains a concern. The specific ingredients of the herbs also remain to be elucidated, and some preparations have been found to be contaminated with corticosteroids. At present, Chinese herbal therapy for AD is considered investigational.
Antimetabolites
Mycophenolate mofetil is a purine biosynthesis inhibitor used as an immunosuppressant in organ transplantation that has been used for treatment of refractory AD. Aside from immunosuppression, herpes retinitis and dose-related bone marrow suppression have been reported with its use. Of note, not all patients benefit from treatment. Therefore, the medication should be discontinued if the disease does not respond within 4-8 wk. Methotrexate is an antimetabolite with potent inhibitory effects on inflammatory cytokine synthesis and cell chemotaxis. Methotrexate has been used for patients with recalcitrant AD. In AD, dosing is more frequent than the weekly dosing used for psoriasis. Azathioprine is a purine analogue with anti-inflammatory and antiproliferative effects that has been used for severe AD. Myelosuppression is a significant adverse effect, and thiopurine methyl transferase levels may identify individuals at risk for it. Before any of these drugs is used, patients should be referred to an AD specialist who is familiar with treatment of severe AD, because well-controlled clinical trials of the use of antimetabolites in AD are lacking.
Avoiding Triggers
It is essential to identify and eliminate triggering factors, both during the period of acute symptoms and on a long-term basis to prevent recurrences.
Irritants
Patients with AD have a low threshold response to irritants that trigger their itch-scratch cycle. Soaps or detergents, chemicals, smoke, abrasive clothing, and exposure to extremes of temperature and humidity are common triggers. Patients with AD should use soaps with minimal defatting properties and a neutral pH. New clothing should be laundered before wearing to decrease levels of formaldehyde and other chemicals. Residual laundry detergent in clothing may trigger the itch-scratch cycle; using a liquid rather than powder detergent and adding a 2nd rinse cycle facilitates removal of the detergent.
Every attempt should be made to allow children with AD to be as normally active as possible. A sport such as swimming may be better tolerated than others that involve intense perspiration, physical contact, or heavy clothing and equipment. Rinsing off chlorine immediately and lubricating the skin after swimming are important. Although ultraviolet light may be beneficial to some patients with AD, high–sun protection factor (SPF) sunscreens should be used to avoid sunburn.
Foods
Food allergy is co-morbid in approximately 40% of infants and young children with moderate to severe AD (Chapter 145). Undiagnosed food allergies in patients with AD may induce eczematous dermatitis in some patients and urticarial reactions, wheezing, or nasal congestion in others. Increased severity of AD symptoms and younger age correlate directly with the presence of food allergy. Removal of food allergens from the diet leads to significant clinical improvement but requires a great deal of education, because most common allergens (egg, milk, peanut, wheat, soy) contaminate many foods and are difficult to avoid.
Potential allergens can be identified by a careful history and performing selective skin prick tests or in vitro blood testing for allergen-specific IgE. Negative skin and blood test results for allergen-specific IgE have a high predictive value for excluding suspected allergens. Positive results of skin or blood tests using foods often do not correlate with clinical symptoms and should be confirmed with controlled food challenges and elimination diets. Extensive elimination diets, which can be nutritionally deficient, are rarely required. Even with multiple positive skin test results, the majority of patients react to fewer than 3 foods under controlled challenge conditions.
Aeroallergens
In older children, AD flares can occur after intranasal or epicutaneous exposure to aeroallergens such as fungi, animal dander, grass, and ragweed pollen. Avoiding aeroallergens, particularly dust mites, can result in clinical improvement of AD. Avoidance measures for dust mite–allergic patients include using dust mite–proof encasings on pillows, mattresses, and box springs; washing bedding in hot water weekly; removing bedroom carpeting; and decreasing indoor humidity levels with air conditioning.
Infections
Patients with AD have increased susceptibility to bacterial, viral, and fungal skin infections. Antistaphylococcal antibiotics are very helpful for treating patients who are heavily colonized or infected with Staphylococcus aureus. Erythromycin and azithromycin are usually beneficial for patients who are not colonized with a resistant S. aureus strain; however, a first-generation cephalosporin (cephalexin) is recommended for macrolide-resistant S. aureus. Topical mupirocin is useful in the treatment of localized impetiginous lesions, with systemic antibiotic for widespread infections. Cytokine-mediated skin inflammation contributes to skin colonization with S. aureus; this fact indicates the importance of combining effective anti-inflammatory therapy with antibiotics for treating moderate to severe AD to avoid the need for repeated courses of antibiotics, which can lead to the emergence of antibiotic-resistant strains of S. aureus.
Herpes simplex virus (HSV) can provoke recurrent dermatitis and may be misdiagnosed as S. aureus infection. The presence of punched-out erosions, vesicles, and infected skin lesions that fail to respond to oral antibiotics suggests HSV infection, which can be diagnosed by a Giemsa-stained Tzanck smear of cells scraped from the vesicle base or by viral polymerase chain reaction or culture. Topical corticosteroids should be temporarily discontinued if HSV infection is suspected. Reports of life-threatening dissemination of HSV infections in patients with AD who have widespread disease mandate antiviral treatment. Dermatophyte infections also can contribute to exacerbation of AD disease activity. Patients with dermatophyte infection or IgE antibodies to Malassezia furfur (formerly known as Pityrosporum ovale) may benefit from a trial of topical or systemic antifungal therapy.
Complications
Staphylococcus aureus is found in >90% of AD skin lesions. Antibiotic therapy is required if honey-colored crusting, folliculitis, impetigo, and pyoderma are present. Regional lymphadenopathy is common in such cases. The importance of S. aureus in AD is supported by the observation that patients with severe AD, even those without overt infection, may show clinical response to combined treatment with antistaphylococcal antibiotics and topical corticosteroids.
AD is associated with recurrent viral skin infections. The most serious viral infection is Kaposi varicelliform eruption, or eczema herpeticum, which is caused by HSV and affects patients of all ages. The incubation period of 5-12 days is followed by disseminated eruption of multiple, itchy, vesiculopustular lesions. The vesicular lesions are umbilicated, tend to crop, and often become hemorrhagic and crusted. Persons with AD are susceptible to eczema vaccinatum, which is similar in appearance to eczema herpeticum and historically follows smallpox (vaccinia virus) vaccination. Cutaneous warts and molluscum contagiosum are additional viral infections affecting children with AD.
Patients with AD have been found to have a greater susceptibility to Trichophyton rubrum fungal infections than nonatopic control subjects. There has been particular interest in the role of M. furfur in AD because it is a lipophilic yeast commonly present in the seborrheic areas of the skin. IgE antibodies against M. furfur have been found in patients with head and neck dermatitis. A reduction of AD severity has been observed in those patients after treatment with antifungal agents.
Exfoliative dermatitis may develop in patients with extensive skin involvement. It is associated with generalized redness, scaling, weeping, crusting, systemic toxicity, lymphadenopathy, and fever and is usually caused by superinfection (e.g., with toxin-producing S. aureus or HSV infection) or inappropriate therapy. In some cases, the withdrawal of systemic glucocorticoids used to control severe AD precipitates exfoliative erythroderma.
Eyelid dermatitis and chronic blepharitis may result in visual impairment from corneal scarring. Atopic keratoconjunctivitis is usually bilateral and can have disabling symptoms that include itching, burning, tearing, and copious mucoid discharge. Vernal conjunctivitis is associated with papillary hypertrophy or cobblestoning of the upper eyelid conjunctiva. It typically occurs in younger patients and has a marked seasonal incidence with spring exacerbations. Keratoconus is a conical deformity of the cornea believed to result from chronic rubbing of the eyes in patients with AD. Cataracts may be a primary manifestation of AD or from extensive use of systemic and topical glucocorticoids, particularly around the eyes.
Prognosis
AD generally tends to be more severe and persistent in young children, particularly if they have null mutations in their filaggrin genes. Periods of remission occur more frequently as patients grow older. Spontaneous resolution of AD has been reported to occur after age 5 yr in 40-60% of patients affected during infancy, particularly for mild disease. Earlier studies suggested that approximately 84% of children outgrow their AD by adolescence; however, later studies reported that AD resolves in approximately 20% of children monitored from infancy until adolescence and becomes less severe in 65%. Of those adolescents treated for mild dermatitis, >50% may experience a relapse of disease as adults, which frequently manifests as hand dermatitis, especially if daily activities require repeated hand wetting. Predictive factors of a poor prognosis for AD include widespread AD in childhood, filaggrin gene null mutations, concomitant allergic rhinitis and asthma, family history of AD in parents or siblings, early age at onset of AD, being an only child, and very high serum IgE levels.
Prevention
Breast-feeding or a feeding with a hypoallergenic hydrolyzed formula may be beneficial. Probiotics may also reduce the incidence or severity of AD, but this possibility is unproven. If an infant with AD is diagnosed with food allergy, the breast = feeding mother will need to eliminate the implicated food allergen from her diet. Identification and elimination of triggering factors is the mainstay for prevention of flares as well as for the long-term treatment of AD.
Alm B, Aberg N, Erdes L, et al. Early introduction of fish decreases the risk of eczema in infants. Arch Dis Child. 2009;94:11-15.
Baumer JH. Atopic eczema in children, NICE. Arch Dis Child Educ Pract Ed. 2008;93:93-97.
Bieber T. Atopic dermatitis. N Engl J Med. 2008;358:1483-1494.
Boguniewicz M, Nicol N, Kelsay K, et al. A multidisciplinary approach to evaluation and treatment of atopic dermatitis. Semin Cutan Med Surg. 2008;27:115-127.
Boyle RJ, Bath-Hextall FJ, Leonardi-Bee J, et al: Probiotics for treating eczema (review), Cochrane Database System Rev (4): CD006135, 2008.
De Benedetto A, Agnihothri R, McGirt LY, et al. Atopic dermatitis: a disease caused by innate immune defects? J Invest Dermatol. 2009;129:14-30.
Esparza-Gordillo J, Weidinger S, Fölster-Holst R, et al. A common variant on chromosome 11q13 is associated with atopic dermatitis. Nat Genet. 2009;41:596-601.
Jung T, Stingl G. Atopic dermatitis: therapeutic concepts evolving from new pathophysiologic insights. J Allergy Clin Immunol. 2008;122:1074-1081.
Lee J, Seto D, Bielory L. Meta-analysis of clinical trials of probiotics for prevention and treatment of pediatric atopic dermatitis. J Allergy Clin Immunol. 2008;121:116-121.
Krakowski AC, Eichenfield LF, Dohil MA. Management of atopic dermatitis in the pediatric population. Pediatrics. 2008;122:812-824.
Lewis-Jones S, Mugglestone MA. Guideline Development Group: Management of atopic eczema in children aged up to 12 years: summary of NICE guidance. BMJ. 2007;335:1263-1264.
O’Regan GM, Sandilands A, McLean WH, et al. Filaggrin in atopic dermatitis. J Allergy Clin Immunol. 2008;122:689-693.
Ownby DR, Johnson CC. Does exposure to cats or dogs in early life alter a child’s risk of atopic dermatitis? J Pediatr. 2010;158(2):184-186.
Paller AS, Eichenfield LF, Kirsner RS, et al. Three times weekly tacrolimus ointment reduces relapse in stabilized atopic dermatitis: a new paradigm for use. Pediatrics. 2008;122:e1210-e1218.
Rosenlund H, Bergström A, Aim JA, et al. Allergic disease and atopic sensitization in children in relation to measles vaccination and measles infection. Pediatrics. 2009;123:771-778.
Taylor AL, Dunstan JA, Prescott SL. Probiotic supplementation for the first 6 months of life fails to reduce the risk of atopic dermatitis and increases the risk of allergen sensitization in high-risk children: a randomized controlled trial. J Allergy Clin Immunol. 2007;119:184-191.
Treadwell PA. Eczema and infection. Pediatr Infect Dis J. 2008;27:551-552.