Chapter 685 General Considerations
The genetically and clinically heterogeneous group of disorders of skeletal development and growth are referred to as skeletal dysplasias, bone dysplasias, and osteochondrodysplasias. Their prevalence is estimated to be about 1/4,000 births. They can be divided into the osteodysplasias typified by osteogenesis imperfecta (Chapter 692) and the chondrodysplasias. The latter result from mutations of genes that are essential for skeletal development and growth. The clinical picture is dominated by skeletal abnormalities. The manifestations may be restricted to the skeleton, but in most cases nonskeletal tissues are also involved. The disorders may be lethal in utero or mild with features that go undetected.
The chondrodysplasias are distinguished from other forms of short stature by a disproportionality of skeletal manifestations. The importance of cartilage in bone formation is noted in Figure 685-1. There are two basic categories: predominantly with short limbs and predominantly with short trunks. Efforts to define the extent of clinical heterogeneity resulted in the delineation of >100 distinct entities. Many of these disorders result from mutations of a relatively small group of genes, the “chondrodysplasia genes.” An International Working Group on Bone Dysplasias has named and classified these disorders into groups based on genetic cause if known or on similarities of clinical and radiographic manifestations, which often imply a common pathogenesis and a common genetic basis, if the cause is unknown (Table 685-1). The better-defined chondrodysplasia groups, such as the achondroplasia and type II collagenopathy groups, contain graded series of disorders that range from very severe to very mild. This may be true for other groups as more mutations are found and the full spectrum of clinical phenotypes associated with mutations of a given gene is defined. These disorders are clinical phenotypes distributed along spectra of phenotypic abnormality associated with mutations of particular genes. For mutations of some genes such as COL2A1, the distribution is fairly continuous, with clinical phenotypes merging into one another across a broad range. There is much less clinical overlap for mutations of some other genes, such as FGFR3, in which the distribution is discontinuous. Because most clinicians and most reference materials refer to the disorders as distinct entities, this vernacular continues to be used.
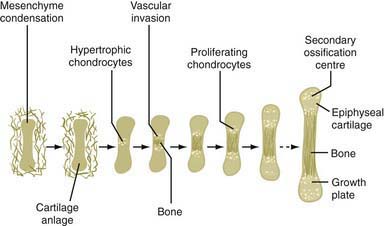
Figure 685-1 The importance of cartilage in bone formation.
(From Horton WA: Skeletal development: Insights from targeting the mouse genome, Lancet 362:560, 2005.)
Most chondrodysplasias require the analysis of information from the history, physical examination, skeletal radiographs, family history, and laboratory testing to make a diagnosis. The process involves recognizing complex patterns that are characteristic of the different disorders (Tables 685-2, 685-3, 685-4, and 685-5). Comprehensive descriptions of disorders and references are at the Online Mendelian Inheritance in Man (OMIM) Internet site (see the references).
Table 685-2 MAJOR PROBLEMS ASSOCIATED WITH SKELETAL DYSPLASIAS
| PROBLEM | EXAMPLE |
|---|---|
| Lethality* | Thanatophoric dysplasia |
| Associated anomalies† | Ellis-van Creveld syndrome |
| Short stature | Common to almost all |
| Cervical spine dislocations | Larsen syndrome |
| Severe limb bowing | Metaphyseal dysplasia, Schmid type |
| Spine curvatures | Metatropic dysplasia |
| Clubfeet | Diastrophic dysplasia |
| Fractures | Osteogenesis imperfecta |
| Pneumonias, aspirations | Campomelic dysplasia |
| Spinal cord compression | Achondroplasia |
| Joint problems (hips, knees) | Most skeletal dysplasias |
| Hearing loss | Common (greatest with cleft palate) |
| Myopia/cataracts | Stickler syndrome |
| Immunodeficiency‡ | Cartilage-hair hypoplasia, Schimke immuno-osseous dysplasia |
| Poor body image | Variable, but common to all |
| Sex reversal | Campomelic dysplasia |
* Mostly due to severely reduced size of thorax.
† See Table 685-3.
‡ At least four additional disorders, all involving the metaphyses, can have immunodeficiency.
Table 685-3 ASSOCIATED ANOMALIES IN SKELETAL DYSPLASIAS
| ANOMALY | EXAMPLE |
|---|---|
| Heart defects | Ellis-van Creveld syndrome, Jeune syndrome |
| Polydactyly | Short rib polydactyly, Majewski type |
| Cleft palate | Diastrophic dysplasia |
| Ear cysts | Diastrophic dysplasia |
| Spinal cord compression | Achondroplasia |
| Encephalocele | Dyssegmental dysplasia |
| Hemivertebrae | Dyssegmental dysplasia |
| Micrognathia | Campomelic dysplasia |
| Nail dysplasia | Ellis-van Creveld syndrome |
| Conical teeth, oligodontia | Ellis-van Creveld syndrome |
| Multiple oral frenulae | Ellis-van Creveld syndrome |
| Dentinogenesis imperfecta | Osteogenesis imperfecta |
| Pretibial skin dimples | Campomelic dysplasia |
| Cataracts, retinal detachment | Stickler syndrome |
| Intestinal atresia | Saldino-Noonan |
| Renal cysts | Saldino-Noonan |
| Campodactyly | Diastrophic dysplasia |
| Craniosynostosis | Thanatophoric dysplasia |
| Ichthyosis | Chondrodystrophica punctata |
| Hitchhiker thumb | Diastrophic dysplasia |
| Sparse scalp hair | Cartilage-hair hypoplasia |
| Hypertelorism | Robinow syndrome |
| Hypoplastic nasal bridge | Acrodysostosis |
| Clavicular agenesis | Cleidocranial dysplasia |
| Genital hypoplasia | Robinow syndrome |
| Tail | Metatropic dysplasia |
| Omphalocele | Beemer-Langer syndrome |
| Blue sclera | Osteogenesis imperfecta |
Table 685-4 LETHAL NEONATAL DWARFISM
USUALLY FATAL*
OFTEN FATAL
OCCASIONALLY FATAL
* A few prolonged survivors have been reported in most of these disorders.
Table 685-5 USUALLY NONLETHAL DWARFING CONDITIONS RECOGNIZABLE AT BIRTH OR WITHIN FIRST FEW MONTHS OF LIFE
MOST COMMON
LESS COMMON
Clinical Manifestations
Growth Related
The hallmark of the chondrodysplasias is disproportionate short stature. Although this refers to a disproportion between the limbs and the trunk, most disorders exhibit some shortening of both, and subtle degrees of disproportion may be difficult to appreciate, especially in premature, obese, or edematous infants. Disproportionate shortening of the limbs should be suspected if the upper limbs do not reach the mid pelvis in infancy or the upper thigh after infancy. Disproportionate shortening of the trunk is indicated by a short neck, small chest, and protuberant abdomen. Skeletal disproportion is usually accompanied by short stature (length and height below the 3rd percentile); these measurements are occasionally within the low-normal range early in the course of certain conditions.
There may also be disproportionate shortening of different segments of the limbs; the particular pattern can provide clues for specific diagnoses. Shortening is greatest in the proximal segments (upper arms and legs) in achondroplasia; this is termed rhizomelic shortening. Disproportionate shortening of the middle segments (forearms and lower legs) is called mesomelic shortening; acromelic shortening involves the hands and feet.
With some exceptions, there is a strong correlation between the age at onset and the clinical severity. Many of the lethal neonatal chondrodysplasias are evident during routine fetal ultrasound examinations performed at the end of the 1st trimester of gestation (see Table 685-4). Gestational standards exist for long-bone lengths; discrepancies are often detected between biparietal diameter of the skull and long-bone lengths. Many disorders become apparent around the time of birth; others manifest during the 1st yr of life. A number of disorders manifest in early childhood and a few in late childhood or later.
Non–Growth Related
Most patients also have problems unrelated to growth. Skeletal deformities, such as abnormal joint mobility, protuberances at and around joints, and angular deformities, are common and usually symmetric. Skeletal abnormalities can adversely affect nonskeletal tissues. Impaired growth at the base of the skull and of vertebral pedicles reduces the size of the spinal canal in achondroplasia and can contribute to spinal cord compression. Short ribs reduce thoracic volume, which can compromise breathing in patients with short trunk chondrodysplasias. Cleft palate is common to many disorders, presumably reflecting defective palatal growth.
Manifestations may be unrelated to the skeleton; they reflect expression of mutant genes in nonskeletal tissues. Examples include retinal detachment in spondyloepiphyseal dysplasia congenita, sex reversal in campomelic dysplasia, congenital heart malformations in Ellis-van Creveld syndrome, immunodeficiency in cartilage-hair hypoplasia, and renal dysfunction in asphyxiating thoracic dystrophy. These nonskeletal problems provide valuable clues to specific diagnoses and must be managed clinically (see Table 685-3).
Family and Reproductive History
A family history might identify relatives with the condition; a mendelian inheritance pattern may be elicited. Because the presentation can vary in some disorders, features that might be related to the disorder should be identified. Special attention should be given to mild degrees of short stature, disproportion, deformities, and other manifestations such as precocious osteoarthritis because they may be overlooked. Physical examination of relatives may be useful, as may the review of their photographs, radiographs, and medical records.
A reproductive history might reveal previous stillbirths, fetal losses, and other abnormal pregnancy outcomes resulting from a skeletal dysplasia. Pregnancy complications, such as polyhydramnios or reduced fetal movement, are common in bone dysplasias, especially neonatal lethal variants.
Even though most of the skeletal dysplasias are genetic, it is common to have no family history of the disorder. New mutations are common for autosomal dominant disorders, especially lethal disorders in the perinatal period (thanatophoric dysplasia, osteogenesis imperfecta). Most cases of achondroplasia result from new mutations. Germ cell mosaicism, in which a parent has clones of mutant germ cells, has been observed in osteogenesis imperfecta and in other dominant disorders. A negative family history is usually seen in recessive disorders. Prenatal diagnosis is available for disorders that have a genetic locus identified. Appropriateness of the testing depends on many factors, and genetic counseling is warranted for these families.
Radiographic Features
Radiographic evaluation for a chondrodysplasia should include plain films of the entire skeleton. Efforts should be made to identify which bones and which parts of bones (epiphyses, metaphyses, diaphyses) are most affected. If possible, films taken at different ages should be examined because the radiographic changes evolve with time. Films taken before puberty are generally more informative because pubertal closure of the epiphyses obliterates many of the signs needed for a radiographic diagnosis. Prenatal diagnosis may also be possible with fetal ultrasound.
Diagnosis
If an infant or child is short with disproportionate features, a diagnosis is established by matching the observed clinical picture (defined primarily from clinical, family, and gestational histories; physical examination; and radiographic evaluation) with clinical phenotypes of well-documented disorders. Pediatricians should be able to gather most of this information and, in consultation with a radiologist, diagnose the common chondrodysplasias. A number of reference texts and online databases provide information about the disorders and comprehensive lists of current references. For less common disorders and for infants and children whose phenotypes do not closely match well-established clinical phenotypes, consultation with experts in the bone dysplasia field is warranted.
Laboratory testing has not been useful in diagnosing chondrodysplasias except in osteogenesis imperfecta, in which analysis of collagen synthesis by skin fibroblasts or genes whose products are involved in collagen biosynthesis, has helped establish a diagnosis. Osteogenesis imperfecta is not a chondrodysplasia, but it is often in the differential diagnosis, especially for newborns with severe skeletal deformities (Chapter 692). Reduced plasma levels of COMP (cartilage oligomeric matrix protein) have been detected in patients with pseudoachondroplasia and multiple epiphyseal dysplasias in which COMP mutations have been found.
Molecular genetic testing for chondrodysplasias is very useful, especially for disorders in which recurrent mutations occur (typical achondroplasia has the same FGFR3 mutation). Mutation testing for achondroplasia is available, although the diagnosis is usually made clinically. The greatest utility for testing may be for prenatal diagnosis for couples where both parents have typical (heterozygous) achondroplasia. Their children are at a 25% risk of the much more severe homozygous achondroplasia, which can be detected by mutation analysis. Another example is in disorders resulting from mutations of DTDST. These disorders are inherited in an autosomal recessive manner, and a limited number of mutant alleles have been found. If the mutations are identified in the patient, they should be detectable in the parents and potentially used for prenatal diagnosis. Mutational analysis is now commercially available for many of the skeletal dysplasias and is increasingly used to confirm clinical diagnosis and for future pregnancy planning.
Many of the chondrodysplasias have distinct histologic changes of the skeletal growth plate. Sometimes such tissues obtained at biopsy or discarded from a surgical procedure are helpful diagnostically. It is uncommon to make a diagnosis histologically if it was not already suspected on clinical grounds. An exception is for the lethal neonatal chondrodysplasias, in which an aborted fetus is macerated, thus making a clinical and radiographic assessment difficult.
Molecular Genetics
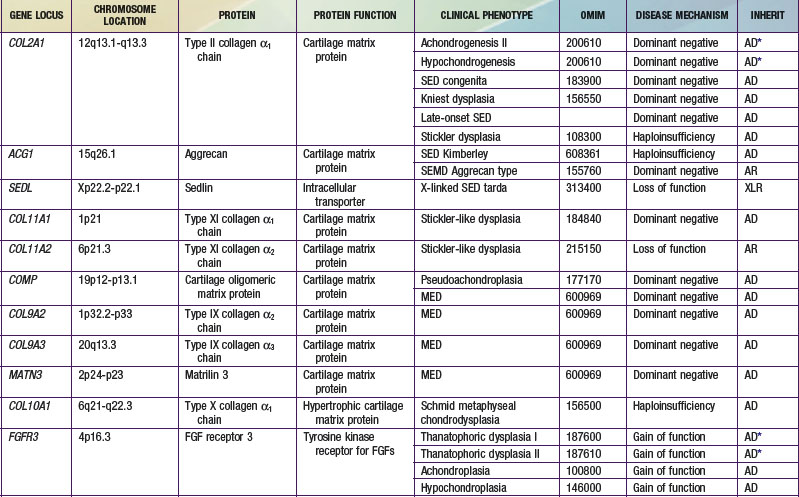
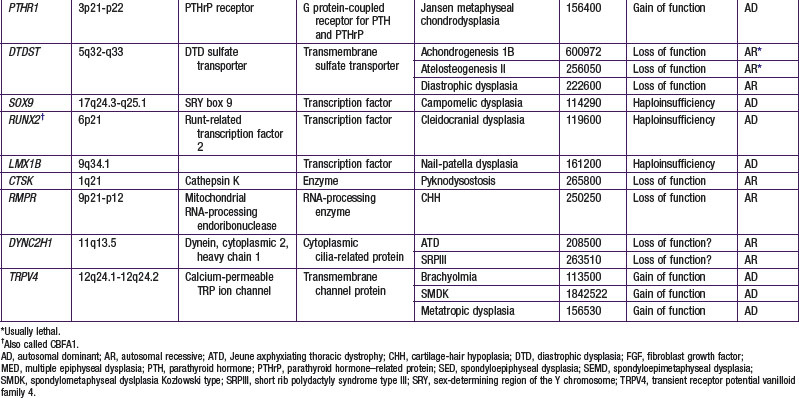
A number of chondrodysplasia genes have been identified (see Table 685-1). They encode several categories of proteins, including cartilage matrix proteins, transmembrane receptors, ion transporters, and transcription factors. The number of identified gene loci is smaller than anticipated from the number of recognized clinical phenotypes. The majority of patients have disorders that map to <10 loci; mutations at 2 loci (COL2A1 and FGFR3) account for more than half of all cases. There may be a limited number of genes whose function is critical to skeletal development, especially linear bone growth; mutations in these genes give rise to a wide range of chondrodysplasia clinical phenotypes. However, new genes harboring mutations that cause chondrodysplasias continue to be identified with advances in detection technology.
Mutations at the COL2A1 and FGFR3 loci illustrate different genetic characteristics. COL2A1 mutations are distributed throughout the gene, with few instances of recurrence in unrelated persons. In contrast, FGFR3 mutations are restricted to a few locations within the gene, and occurrence of new mutations at these sites in unrelated persons is the rule. There is a strong correlation between clinical phenotype and mutation site for FGFR3, but not COL2A1, mutations.
Pathophysiology
Chondrodysplasia mutations act through different mechanisms. Most mutations involving cartilage matrix proteins cause disease when only 1 of the 2 copies (alleles) of the relevant gene is mutated. These mutations usually act through a dominant negative mechanism in which the protein products of the mutant allele interfere with the assembly and function of multimeric molecules that contain the protein products of both the normal and mutant alleles. The type II collagen molecule is a triple helix composed of 3 collagen chains, which are the products of the type II collagen gene COL2A1. When chains from both normal and mutant alleles are combined to form triple helices, most molecules contain at least 1 mutant chain. It is not known how many mutant chains are required to produce a dysfunctional molecule but, depending on the mutation, it theoretically could be as few as 1.
Mutations involving type X collagen differ from the model just described. They map to the region of the chain that is responsible for chain recognition; the chains must recognize each other before they can assemble into collagen molecules. Mutations are thought to disrupt this process. As a result, none of the mutant chains are incorporated into molecules. This mechanism is haploinsufficiency because the products of the mutant allele are functionally absent and the normal allele is insufficient for normal function. Mutations involving ion transport genes also act through a loss of function of the transporters. Alternatively, mutations of transmembrane receptors studied to date appear to act through a gain of function; the mutant receptors initiate signals in a constitutive manner independent of their normal ligands.
Regardless of genetic mechanism, the mutations ultimately disrupt endochondral ossification, the biologic process responsible for the development and linear growth of the skeleton (see Fig. 685-1). Indeed, a wide range of morphologic abnormalities of the skeletal growth plate, the anatomic structure in which endochondral ossification occurs, have been described in the chondrodysplasias.
Treatment
The first step is to establish the correct diagnosis. This allows one to predict a prognosis and to anticipate the medical and surgical problems associated with a particular disorder. Establishing a diagnosis helps to distinguish between lethal disorders and nonlethal disorders in a premature or newborn infant (see Tables 685-4 and 685-5). A poor prognosis for long-term survival might argue against initiating extreme lifesaving measures for thanatophoric dysplasia or achondrogenesis types Ib or II, whereas such measures may be indicated for infants with spondyloepiphyseal dysplasia congenita or diastrophic dysplasia, which have a good prognosis if the infant survives the newborn period.
Because there is no definitive therapy to normalize bone growth in any of the disorders, management is directed at preventing and correcting skeletal deformities, treating nonskeletal complications, providing genetic counseling, and helping patients and families learn to cope. Each disorder has its own unique set of problems, and consequently management must be tailored to each disorder. Medical information for a few disorders can be found at the Medical Information on Dwarfism website (see references).
There are a number of problems common to many chondrodysplasias for which general recommendations can be made. Children with most chondrodysplasias should avoid contact sports and other activities that cause injury or stress to joints. Good dietary habits should be established in childhood to prevent or minimize obesity in adulthood. Dental care should be started early to minimize crowding and malalignment of teeth. Children and relatives should be given the opportunity to participate in support groups, such as the Little People of America and Human Growth Foundation.
Two controversial approaches have been used to increase bone length. Surgical limb lengthening has been employed for a few disorders. Its greatest success has been in achondroplasia in which nonskeletal tissues tend to be redundant and easily stretched. The procedure is usually performed during adolescence. Pharmacologic doses of human growth hormone comparable to those used to treat Turner syndrome have also been tried in several disorders; the results have been equivocal.
Apajasalo M, Sintonen H, Rautonen J, Kaitila I. Health-related quality of life of patients with genetic skeletal dysplasias. Eur J Pediatr. 1998;157:114-121.
Gripp K, Slavotinek A, Hall JG, et al. Handbook of normal physical measurements, ed 2. Oxford, UK: Oxford University Press; 2006.
Horton WA. Skeletal development: insights from targeting the mouse genome. Lancet. 2003;362:560-569.
Horton WA, Hecht JT. Chondrodysplasias: general concepts and diagnostic and management considerations. In: Royce PM, Steinmann B, editors. Connective tissue and its heritable disorders: molecular, genetic, and medical aspects. ed 2. New York: Wiley-Liss; 2002:901-908.
Kozlowski K, Masel J, Sillence DO, et al. Gracile bone dysplasias. Pediatr Radiol. 2002;32:629-634.
Krakow D, Lachman RS, Rimoin DL. Guidelines for the prenatal diagnosis of fetal skeletal dysplasias. Genet Med. 2009;11:127-133.
Lachman RS. Neurologic abnormalities in the skeletal dysplasias: a clinical and radiological perspective. Am J Med Genet. 1997;69:33-43.
Lachman RS. Taybi and Lachman’s radiology of syndromes, metabolic disorders and skeletal dysplasias, ed 2. New York: Elsevier; 2007.
Schramm T, Gloning KP, Minderer S, et al. Prenatal sonographic diagnosis of skeletal dysplasias. Ultrasound Obstet Gynecol. 2009;34(2):160-170.
Smits P, Bolton AD, Funari V, et al. Lethal skeletal dysplasia in mice and humans lacking the golgin GMAP-210. N Engl J Med. 2010;362:206-216.
Spranger J, Brill PW, Poznanski A. Bone dysplasias. An atlas of genetic disorders of skeletal development, ed 2. New York: Oxford University Press; 2002.
Spranger J, Maroteaux P. The lethal osteochondrodysplasias. Adv Hum Genet. 1990;19:1-103. 331-332
Superti-Furga A, Unger S. Nosology and classification of genetic skeletal disorders: 2006 revision. Am J Med Genet A. 2007;143:1-18.
Unger S, Lachman RS, Rimoin DL. Chondrodysplasias. In: Rimoin DL, Conner JM, Pyeritz RE, et al, editors. Emery and Rimoin’s principles and practice of medical genetics. New York: Churchill Livingstone; 2007:3709.
Medical information on dwarfism. web.memberclicks.com/mc/page.do?orgId=lpamrs.
Online Mendelian Inheritance in Man (OMIM). www.ncbi.nlm.nih.gov/sites/entrez?db=omim.