chapter 39 Renovascular Hypertension and Ischemic Nephropathy
Historical Background
Richard Bright, Physician Extraordinary to the Queen of England, was the first to associate proteinuria, fullness and hardness of the pulse, and dropsy with “hardening of the kidneys” (Bright, 1827). In 1856, Traube, from an analysis of pulse tracings, suggested that the abnormality might be high blood pressure, and Mohomed (1874) demonstrated “high tension in the arterial system” in association with renal disease.
The critical experimental work was the discovery of renin by Tigerstedt and Bergemann (1898), who noted an increase in arterial blood pressure in rabbits injected with a saline renal extract. They reasoned that the renal extract contained a pressor substance and coined the term renin. However, the significance of their work was not recognized until the critical experiments by Goldblatt and colleagues (1934), who produced diastolic hypertension in dogs by clamping the main renal arteries and corrected the hypertension by clamp removal.
Soon thereafter, Butler (1937) reported the first reversal of hypertension after nephrectomy in a patient with a small “pyelonephritic kidney”; 1 year later, Leadbetter and Burkland (1938) reported another cure of hypertension in a child with pathologic signs of a renal arterial lesion.
These clinical observations were paralleled by laboratory investigation, and in 1940, Page and Helmer, and Braun-Menendez and associates, independently reported that renin itself was not a pressor substance but acted as an enzyme to release a pressor peptide, now called angiotensin, from a circulating plasma globulin. Goormaghtigh and Grimson (1939), who had previously described the juxtaglomerular cells, described increased granularity of these cells in both animals and humans with renal hypertension and postulated that these cells were secreting excessive amounts of renin.
There followed an aggressive but disappointing clinical experience with nephrectomy for cure of hypertension in patients with unilateral renal disease. This experience led to the search for a way of proving that a renal lesion was actually causing the hypertension. Smith (1948), reviewing the literature, reported relief of hypertension in only 19% of 200 patients whose elevated blood pressure was thought to result from unilateral renal disease. Thus it became apparent that even if pressor mechanisms did underlie some forms of renal hypertension, there were no ways to measure them.
This challenge led to studies of the effect of renal artery constriction on renal function. In dogs, renal artery constriction resulted in a marked decrease in sodium and water excretion from the affected kidney (Blake et al, 1950; Pitts and Duggan, 1950). In 1964, Howard and Connor used these observations to develop a differential renal function test based on bilateral ureteral catheterization to identify the “ischemic kidney.” Another major advance was the development of translumbar aortography and the demonstration of its value in visualizing renal arterial lesions (Smith et al, 1952). By 1957, the first large series of studies of patients with renal arterial lesions was reported (Poutasse and Dustan, 1957).
In addition, interest in what would become known as the renin-angiotensin-aldosterone system (RAAS) was also emerging as new discoveries were made. Accordingly, it was determined that there were two forms of angiotensin (Skeggs et al, 1954), and angiotensin was sequenced and synthesized (Bumpus et al, 1957). These critical advancements led to an accurate radioimmunoassay for angiotensin, the development of angiotensin analogues, and angiotensin-converting enzyme (ACE) inhibitors, all major tools now used to identify the patient with renovascular hypertension (RVH). More recently, the presence of a family of angiotensin receptors has been clarified (Kang et al, 1994; Goodfriend et al, 1996), and specific blockers of the angiotensin receptor have been used clinically for treatment of hypertension.
It is now recognized that the RAAS is a critical integrated system regulating not only blood pressure, sodium balance, and potassium balance but also regional blood flow and, in particular, the glomerular filtration rate (GFR) (Gunning et al, 1996; Laragh and Blumenfeld, 1996). Moreover, there is an expanding body of literature implicating angiotensin II in cell proliferation and interstitial fibrosis (Mai et al, 1993; Eng et al, 1994; Stoll et al, 1995; Egido, 1996; Gunning et al, 1996).
Definitions
Hypertension
As strange as it may seem, it has been difficult to establish a precise definition of hypertension. The problem was best stated by Sir George Pickering, who wrote that “there is no dividing line. The relationship between arterial blood pressure and mortality is quantitative; the higher the pressure the worse the prognosis” (Pickering and Pickering, 1995; Pickering et al, 1996). Indeed, cumulative data obtained from insurance companies have validated this point! Untreated blood pressure in excess of 140/90 mm Hg is associated with excess mortality, and diastolic pressures below 70 mm Hg are optimal (Lew, 1973). For operational purposes, the World Health Organization has defined hypertension in adults as a systolic pressure greater than 160 mm Hg or a diastolic pressure greater than 95 mm Hg or both. In addition, consistent elevation of blood pressure should be established with repeated readings before evaluation is instituted. In children, there is a rise in blood pressure with age; an upper normal limit of 130/80 mm Hg is reached by 12 to 15 years of age.
Renal Arterial Disease versus Renovascular Hypertension
The development of arteriography provided an accurate means of identifying renal arterial disease and heralded the advent of renal arterial vascular repair (Freeman et al, 1954), which renewed enthusiasm for surgical management of the disease. However, it soon became apparent that normotensive patients undergoing arteriography for other reasons often had renal arterial disease (Eyler et al, 1962), especially those with arteriosclerotic disease (Wilms et al, 1990), and autopsy figures supported the radiologic findings (Holley et al, 1964). Accordingly, the finding of renal arterial disease alone is not sufficient justification to warrant correction in a hypertensive patient. The lesion must be functionally significant (i.e., it must reduce blood flow by an amount sufficient to activate renin release, initiating RVH). Hence, a practical definition of RVH is hypertension resulting from a renal arterial lesion that is relieved by correction of the offending lesion or removal of the kidney.
Pathology and Natural History
The two major pathologic entities that cause renal arterial disease are atherosclerosis obliterans (ASO) and fibrous dysplasia (FD). The Cleveland Clinic group has emphasized the importance of the various distinct histologic patterns, identifiable by angiographic techniques, that have predictable natural histories (Schreiber et al, 1984, 1989; Novick et al, 1994). Their classification is shown in Table 39–1.
Table 39–1 Classification and Natural History of Renovascular Disease
| Atherosclerosis: Proximal intimal plaques. Seen predominantly in males and usually in older age groups. Progressive in about 40% of patients; may dissect or thrombose. May involve renal arteries only or may involve carotid and coronary arteries, aorta, and other vessels. |
| Intimal fibroplasia: Collagenous disease involving intima; seen in children and young adults. Progressive; may dissect. May involve other vessels. |
| True fibromuscular hyperplasia: Diffusely involves media. Seen in children and young adults. Progressive. Radiographically indistinguishable from intimal fibroplasia. Very rare. |
| Medial fibroplasia: Series of collagenous rings involving media of main renal artery, often extending into branches. Usually seen in women in their 30s and 40s. Produces typical “string of beads” pattern in angiography. Does not dissect, thrombose, or rupture, and seldom progresses after 40 years of age. May involve other vessels. |
| Perimedial (subadventitial) fibroplasia: Dense collagenous collar involving media, just beneath adventitia of vessel. Tightly stenotic, with extensive collateral circulation on angiography. Seen mostly in women (“girlie disease”). Progressive. Involves renal arteries only. |
| Miscellaneous: Renal artery aneurysms, middle aortic syndrome, periarterial fibrosis, and post-traumatic intimal or medial disease. Variable in location and obstruction; occurs in diverse clinical settings. |
From Stewart BH, Dustan HP, Kiser WS, et al. Correlation of angiography and natural history in evaluation of patients with renovascular hypertension. J Urol 1970;104:231.
Atherosclerosis
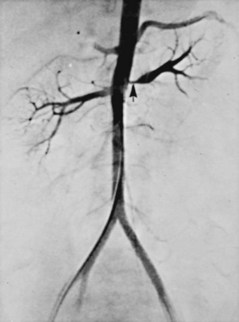
Approximately 70% of all renovascular lesions are caused by atherosclerosis (Novick et al, 1996). This disease may be limited to the renal artery but more commonly is a manifestation of generalized atherosclerosis, involving the abdominal aorta and coronary, cerebral, and lower extremity vessels. Atherosclerotic stenosis usually occurs in the proximal 2 cm of the renal artery, and distal arterial or branch involvement is distinctly uncommon. Owing to the proximal location of these lesions, oblique aortic views are often needed to adequately visualize the area of stenosis. The lesion involves the intima of the artery and, in two thirds of the cases, presents as an eccentric plaque (Fig. 39–1); in the remainder, the vessel is circumferentially involved, with narrowing of the lumen and destruction of the intima. Dissecting hematomas frequently complicate this disease, sometimes resulting in thrombosis of the entire vessel.
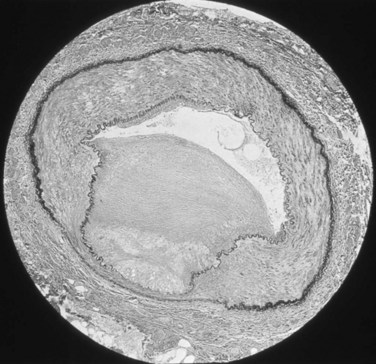
Figure 39–1 Histopathologic appearance of eccentric atherosclerotic plaque causing renal artery stenosis.
The natural history of atherosclerotic renal artery disease (RAD) has been studied by obtaining sequential abdominal aortography or duplex ultrasound scanning in patients with documented renal artery lesions who have been treated medically (Table 39–2). The largest of these studies have shown that progressive arterial obstruction occurs in 42% to 53% of patients with atherosclerotic renal artery disease, often within the first 2 years of radiographic follow-up. The incidence of progression to complete renal artery occlusion in these studies has ranged from 9% to 16%, and this has occurred more often in arteries that initially showed high degrees of stenosis.
Schreiber and colleagues (1984) reviewed the natural history of atherosclerotic renal artery stenosis (ARAS) in 85 patients who were followed with sequential renal angiograms obtained 3 to 172 months after an initial diagnostic angiogram. Progressive obstruction of the renal artery due to atherosclerosis occurred in 37 patients (44%), including 14 (16%) in whom such progression eventuated in complete occlusion of the involved renal artery. In patients in whom progressive disease developed, it occurred primarily within the first 2 years of angiographic follow-up. The rate of progression of ARAS correlated directly with the degree of stenosis on the initial angiogram. The majority of renal arteries with mild (50%) or moderate (50% to 75%) stenosis on the initial angiogram were unchanged on follow-up angiograms. In contrast, 39% of renal arteries with more than 75% stenosis on the initial angiogram progressed to complete occlusion. Other studies have since validated this observation that progression to 100% occlusion occurs more often and more rapidly in renal arteries that are initially involved with a high degree (>75%) of stenosis (Tollefson and Ernst, 1991; Zierler et al, 1994).
Clinical follow-up of patients in the same study (Schreiber et al, 1984) also revealed that significantly more patients with progressive disease developed deterioration of overall renal function compared with patients with stable disease. Interestingly, serial blood pressure control was equivalent in these two groups, indicating that blood pressure is not a useful clinical marker for progressive ARAS.
These natural history data clearly show that atherosclerotic renal artery disease progresses in many patients, and that loss of functioning renal parenchyma is a common sequela of such progression. Such loss of renal function due to progressive atherosclerotic renal artery obstruction can result in end-stage renal disease (ESRD). In this setting, ESRD occurs in older patients with generalized atherosclerosis who are not suitable candidates for transplantation and whose prognosis on chronic dialysis is poor in terms of both the quality of life and longevity. An early study identified 25 patients in whom ESRD was clearly a consequence of advanced atherosclerotic renal artery disease (Novick, 1994b). Seventeen of these patients were maintained on chronic dialysis, and, of these, 13 died within 1 year (mean survival 8.7 months). The causes of death on dialysis were myocardial infarction (6), infection (2), gastrointestinal bleeding (1), ruptured aortic aneurysm (1), mesenteric infarct (1), cardiogenic shock (1), and cerebrovascular accident (1). In a subsequent study, Mailloux and colleagues (1988) analyzed the survival of patients started on dialysis from 1970 to 1985 according to the primary renal diagnosis. Patients with renovascular disease as the cause of ESRD had the poorest survival, with a 27-month median survival time and a 12% 5-year survival rate. In another study, in which 51 patients with bilateral ARAS were followed for 52 months, 12% of the patients progressed to ESRD, and an average rate of decline of GFR of 4 mL/min/yr was noted (Baboolal et al, 1998). A crude mortality rate of 45% was reported. These data further highlight that ESRD from atherosclerotic renal artery disease does not respond well to renal replacement therapy.
The exact incidence of ESRD caused by atherosclerotic renal artery disease in the United States is not known. Fatica and colleagues (2001) reported an increase in incidence of renal vascular disease (RVD) as a cause for ESRD in patients starting dialysis treatment. This increase was from 1.4% to 2.1%, with an annual increase of 12%. This information was derived from the recorded diagnosis of these patients in the U.S. Renal Data System database, and the disease was not specifically searched for. No increase in mortality on dialysis was found in these patients when compared with other etiologies of ESRD.
When investigating the use of CT angiography, van Ampting and coworkers (2003) reported a 27% incidence of significant renal artery stenosis (RAS) in 49 patients over 45 years of age starting dialysis.
Uzu and coworkers (2002) reported a higher (50%) incidence in 44 patients with ESRD who were studied by magnetic resonance angiography when additional vascular disease (cerebral, coronary, or peripheral) was also diagnosed.
In a report from England, Scoble and colleagues (1989) prospectively performed renal arteriography in all new patients with ESRD during an 18-month period. Atherosclerotic renal artery disease was the cause of ESRD in 6% of all patients, and in 14% of patients older than 50 years. Approximately 300,000 patients in the United States are currently being maintained on chronic dialysis. Their median age is older than 60 years, and a majority show evidence of generalized atherosclerosis obliterans. Although the exact number of patients with ESRD caused by atherosclerotic renal artery disease is not known, based on the data previously described, there appear to be several thousand patients in this category.
Fibrous (Fibromuscular) Dysplasia
Fibrous dysplasia (FD) is a nonatherosclerotic noninflammatory vascular disorder with multiple subtypes depending upon the portion of the vessel wall that is primarily involved. The vast majority of cases affect the media (medial fibroplasia); less common is perimedial fibroplasia, intimal fibroplasia, and fibromuscular hyperplasia.
Intimal Fibroplasia
Primary intimal fibroplasia occurs in children and in young adults and constitutes approximately 10% of the total number of fibrous lesions. This lesion is characterized by a circumferential accumulation of collagen inside the internal elastic lamina (Fig. 39–2). Disruption and duplication of the elastica interna occur more often in younger patients, with dissecting hematomas as a complication in many patients. The possibility of atherosclerosis as a cause of renal artery disease in this group can be excluded histologically by the absence of lipid demonstrable with special staining techniques. Intimal fibroplasia with complicating medial dissection is characterized pathologically by large dissecting channels in the outer half of the media. These lesions are thought to develop because of defects in the internal elastica with resultant medial dissection and aneurysmal dilatation.
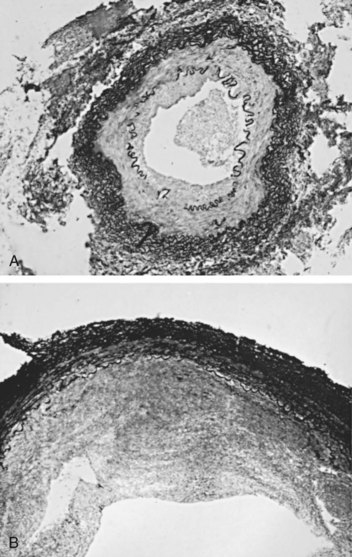
Figure 39–2 A, Photomicrograph of a cross section demonstrates intimal fibroplasia with focal fragmentation and partial absence of the elastica interna. B, Photomicrograph of cross section demonstrates severe renal arterial intimal fibroplasia with a dense cuff of intimal collagen apposed to the luminal surface of a partially disrupted elastica interna. A small recannulized channel is noted in the lower left.
(From Novick AC. Renal vascular hypertension in children. In: Kelalis PP, King LR, Belman AB, editors. Clinical pediatric urology. Philadelphia: WB Saunders; 1984.)
Angiography in primary intimal fibroplasia reveals a smooth, fairly focal stenosis, usually involving the proximal or midportion of the vessel or its branches (Fig. 39–3). Dissecting hematomas may distort the area of the stenosis. With nonoperative management, progressive renal artery obstruction and ischemic atrophy of the involved kidney invariably occur. Severe intimal fibroplasia may subsequently develop de novo in the contralateral renal artery. Although primary intimal fibroplasia most commonly affects the renal arteries, it may also occur as a generalized disorder with concomitant involvement of carotid, upper and lower extremity, and mesenteric vessels.
Medial Fibroplasia
Medial fibroplasia is the most common of the fibrous lesions, constituting 75% to 80% of the total number. It tends to occur in women between the ages of 25 and 50 years and often involves both renal arteries. It may involve other vessels in the body, most notably the carotid, mesenteric, and iliac arteries. Microscopically, the internal elastic membrane is focally and variably thinned and lost. Within the alternating thickened areas, much of the muscle is replaced by collagen, hence the term medial fibroplasia. In other areas, thinning of the media occurs to the point of complete loss, and microaneurysms can be seen as saccules lined by only the external elastica. In extreme cases, giant aneurysms may be found in association with medial fibroplasia.
Angiographically, medial fibroplasia demonstrates a typical “string of beads” appearance involving the distal two thirds of the main renal artery and branches (Fig. 39–4). The areas of stenosis are often overshadowed by contrast medium in the microaneurysms, making the degree of actual stenosis difficult to assess. The aneurysms themselves are greater in diameter than the normal renal artery proximal to the disease, and extreme collateral circulation is absent. These are important features in differentiating the lesion from perimedial fibroplasia. Schreiber and colleagues (1984) studied the natural history of renal artery disease due to medial fibroplasia in 66 patients who were followed with serial angiography. Progressive renal artery stenosis (RAS) occurred in 22 patients (33%), and, contrary to an earlier report, this occurrence was no different whether patients were older or younger than 40 years. Significantly, there were no cases of progression to total arterial occlusion in this group. Also, clinical follow-up revealed that serial decreases in either overall renal function or the size of the involved kidney seldom occurred in patients with progressive medial fibroplasia, suggesting that the risk of losing renal function is relatively small in patients with this disease who are managed medically.
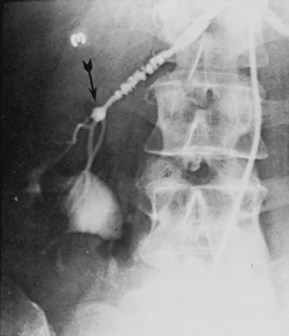
Figure 39–4 Selective right renal arteriogram reveals medial fibroplasia involving the main renal artery with typical “string of beads” appearance.
(From Novick AC. Renal vascular hypertension in children. In: Kelalis PP, King LR, Belman AB, editors. Clinical pediatric urology. Philadelphia: WB Saunders; 1984.)
Perimedial Fibroplasia
Perimedial fibroplasia occurs predominantly in young women between the ages of 15 and 30 years and has therefore been referred to, rather crudely, as girlie disease. It constitutes about 10% to 15% of the total number of fibrous lesions and occurs only in the renal artery. This is a tightly stenotic lesion that, pathologically, consists of a collar of dense collagen enveloping the renal artery for variable lengths and thicknesses. The collagen is deposited in the outer border of the media, usually replaces a considerable portion of the media, and may replace it completely in some areas (Fig. 39–5). Islands of smooth muscle are occasionally seen trapped within the collagenous ring. Special stains show that the lesion is confined within the external elastic lamina and contained in all cases by intact adventitial connective tissue. The arterial lumen may be further compromised by a process of secondary intimal fibroplasia. It has been suggested that this secondary thickening of the intima is related to slowing of blood flow through a narrowed arterial segment, with resultant platelet and fibrin deposition and subsequent fibrous organization.
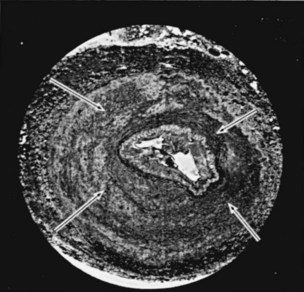
Figure 39–5 Cross section of the main renal artery in a girl with perimedial fibroplasia demonstrates a dense collagenous collar (arrows) involving the outer media of the vessel, which causes a severe progressive stenosis.
(From Novick AC. Renal vascular hypertension in children. In: Kelalis PP, King LR, Belman AB, editors. Clinical pediatric urology. Philadelphia: WB Saunders; 1984.)
The arteriogram in perimedial fibroplasia may give the appearance of arterial beading, but careful observation shows that the caliber of the normal segment of the vessel is not exceeded by the “bead” (Fig. 39–6). This fact, along with the frequent occurrence of extensive collateral circulation, differentiates this lesion angiographically from that of medial fibroplasia. Perimedial fibroplasia produces severe stenosis, and, although complicating thrombosis or dissection is relatively uncommon, progressive obstruction with ischemic renal atrophy occurs in almost all patients managed nonoperatively.
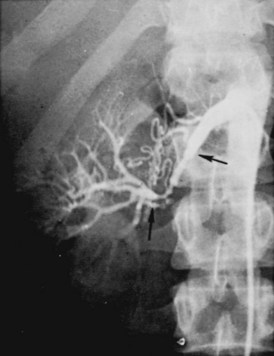
Figure 39–6 Renal arteriogram in a patient with perimedial fibroplasia shows slightly irregular, yet severe, stenosis of the midrenal artery (arrows) associated with extensive collateral circulation to the kidney. The small size of the arterial irregularities and the presence of collateral circulation distinguishes this lesion radiographically from medial fibroplasia.
(From Novick AC. Renal vascular hypertension in children. In: Kelalis PP, King LR, Belman AB, editors. Clinical pediatric urology. Philadelphia: WB Saunders; 1984.)
Fibromuscular Hyperplasia
Fibromuscular hyperplasia is an extremely rare disease, constituting only 2% to 3% of fibrous lesions, and tends to occur in children and young adults. This is the only renal arterial disease in which true hyperplasia of the smooth muscle cells is present. The renal artery shows a concentric thickening of its wall with a mixture of proliferating smooth muscle and fibrous tissue in variable quantity. Angiographically, fibromuscular hyperplasia presents as a smooth stenosis of the renal artery or its branches and, from a radiographic standpoint, may be indistinguishable from intimal fibroplasia. Most patients with this disease have developed progressive vascular obstruction when followed with serial angiographic studies.
Key Points: Pathology of Renal Artery Stenosis
Physiology of the Renin-Angiotensin-Aldosterone System
The RAAS plays a fundamental role in maintaining arterial blood pressure as well as extracellular volume. The system is composed of a series of proteins and peptides that react in a cascade, ultimately resulting in a widespread series of actions. Local renin-angiotensin systems are widely present in several organ systems and exert numerous local actions. The main components of the system are angiotensinogen, renin, ACE, and various angiotensins, the most important being angiotensin II (AII). AII is a powerful vasoconstrictor that increases peripheral vascular resistance to raise blood pressure. In addition, AII stimulates sodium reabsorption directly and through stimulation of aldosterone synthesis. The primary role of the RAAS is to maintain tissue perfusion, especially in cases of hypovolemia.
The basic cascade involves conversion of angiotensinogen to angiotensin I (AI) through the action of renin. This is the rate-limiting step for the entire system, and, accordingly, control of renin release regulates the activity of the whole system. ACE then acts on AI to produce AII, which exerts a wide variety of immediate and delayed actions on the vascular system and kidneys, as well as stimulates production of aldosterone from the adrenal cortex. In healthy subjects with normal dietary sodium intake, the RAAS probably plays a minor role in day-to-day blood pressure control. Several disease states, however, activate the RAAS. True hypovolemia or hypotension is the physiologic stimulus for AII secretion. Inappropriate activation of the RAAS occurs in cases of perceived hypovolemia, such as renal artery stenosis, congestive heart failure, or advanced hepatic disease, and leads to hypertension, inappropriate salt and fluid retention, or both.
Angiotensinogen
Angiotensinogen is a 452–amino acid protein and is the source of all angiotensins (Kageyama et al, 1984). It is formed as preangiotensinogen and loses the signal peptide as it becomes secreted from the cell as angiotensinogen. It functions as a serine protease inhibitor (serpin) similar to α1-antitrypsin and antithrombin III, with which it shares some structural homology (Carrell et al, 1987). It is present in plasma in two forms, a smaller (52- to 60-kD) predominant molecule and a larger (450- to 500-kD) molecule that increases in pregnancy and after estrogen treatment (Tewksbury and Dart, 1982). The larger form is probably composed of the smaller molecule bound to other plasma proteins. Renin acts on the smaller form of angiotensinogen preferentially, cleaving AI off the larger molecule. Renin reacts with much less affinity with the larger form, also forming AI.
The liver is the primary site of synthesis of angiotensinogen, which is not stored but secreted directly after production. Angiotensinogen mRNA is widely present in several tissues that are regulated by local renin-angiotensin systems, including the central nervous system (CNS), kidney, adrenal, heart, and leukocytes (Dzau et al, 1987). Several hormones stimulate angiotensinogen synthesis by the liver, including estrogens and glucocorticoids. Stressful stimuli, such as infection or tissue injury, also increase plasma angiotensinogen levels (Hoj Nielsen and Knudsen, 1987). Feedback control through the RAAS is also present, with AII increasing and renin decreasing plasma levels of angiotensinogen.
Renin
Renin is a single–polypeptide chain aspartyl protease that is secreted from the juxtaglomerular cells of the afferent arteriole. The kidney is the major site of renin production, although renin mRNA is found in several other tissues where a local renin-angiotensin system functions. It is produced as pre-prorenin, and both active renin and prorenin are secreted (Atlas et al, 1980). The function of circulating prorenin is not clear, and it does not appear that prorenin is transformed to active renin in the circulation (Sealey et al, 1977). The action of renin is very specific, restricted to cleavage of a single bond, separating AI from angiotensinogen. Because renin controls the rate-limiting step of the RAAS, control of renin secretion regulates the activity of the RAAS. Several mechanisms affect the secretion of renin, as described in the following sections.
As the first and rate-limiting step for production of angiotensin II, targeting renin is an attractive option for inhibiting the RAAS. Recently a new class of orally effective medications targeting renin has been developed and approved for treatment of hypertension, direct renin inhibitors (DRI). The first of these medications is aliskiren, is a competitive analog and specific inhibitor of human renin, with therapeutic potential similar to other available antagonists of the RAAS (ACE inhibitors, angiotensin receptor blockers) (Nussberger et al, 2002).
Macula Densa Mechanism
The macula densa region of the thick ascending loop of Henle comes in close proximity to the juxtaglomerular cells and influences renin release. Reduction of distal tubule salt delivery stimulates renin secretion, and vice versa. Although sodium was initially thought to be responsible for this action, it now appears that the signal for macula densa–controlled renin release is the alteration of tubular chloride concentration (Lorenz et al, 1990).
Baroreceptor Mechanism
The juxtaglomerular cells of the afferent arteriole act as their own baroreceptors by responding directly to stretch of the afferent arteriole (Tobian et al, 1959). Diminished cell stretch, as a result of renal hypoperfusion, hyperpolarizes the juxtaglomerular cells, resulting in decreased intracellular calcium and increased renin release.
Neural Mechanism
The juxtaglomerular cells are richly innervated by β-adrenergic sympathetic nerve fibers. Stimulation of these β-adrenergic nerves leads to increased renin secretion (Keeton and Campbell, 1980). Dopamine is also stimulatory to renin release, although the limited number of dopaminergic nerve endings results in a much smaller role (Mizoguchi et al, 1983). Renal nerve stimulation is the mechanism through which renin release is increased as a result of exercise and tilting.
Endocrine and Paracrine Mechanisms
Several local and systemic hormones affect the rate of renin secretion. Foremost among these are prostaglandins. Prostaglandin E2 and I2 (prostacyclin), as well as exogenously administrated arachidonic acid, stimulate renin secretion (Franco-Saenz et al, 1980; Whorton et al, 1980). This prostaglandin effect is independent of the other mechanisms controlling renin release. AII inhibits renin release as a feedback mechanism. Other inhibitors of renin release include endothelin, vasopressin, and atrial natriuretic peptide.
Intracellular Mechanisms
Agents that increase adenylate cyclase activity increase the secretion of renin, including β-adrenergic agonists, prostaglandin E2, prostaglandin I2, dopamine, histamine, and parathyroid hormone. This is because cyclic adenosine monophosphate (cAMP) is an important second messenger in renin release. Intracellular calcium concentrations are also important in controlling renin release. AII, vasopressin, and adenosine increase intracellular calcium levels and inhibit renin secretion through their effect on intracellular calcium levels.
Angiotensin-Converting Enzyme
ACE is a zinc-containing single-chain glycoprotein enzyme. It is also known as kininase II and is a dipeptidyl carboxypeptidase (Ehlers and Riordan, 1989). It splits two amino acids off the carboxy terminus of AI to form AII and, at the same time, functions in the kallikrein-kinin system by inactivating bradykinin. ACE is found in a wide variety of organs, where it is primarily expressed on endothelial, epithelial, and neuroepithelial cells. A high concentration of ACE is found in the kidney, ileum, duodenum, and uterus (Lieberman and Sastre, 1983). Although pulmonary endothelial ACE was presumed to be the major site of ACE activity for the systemic RAAS, it is now believed that peripheral sites might play an equal role. The majority of circulating ACE originates from endothelial cells and macrophages.
ACE is expressed in several tissues where local renin-angiotensin systems function. Renal ACE is localized to the glomerular endothelial cells and the proximal tubule brush border, where it might play a role in cleaving filtered protein for reabsorption (Danilov et al, 1987). Within the CNS, ACE is found in several locations, where it functions in the local renin-angiotensin system. This local CNS renin-angiotensin system is thought to have dipsogenic and hypertensive effects as well as to stimulate vasopressin secretion (Strittmatter and Snyder, 1987). Adrenal ACE is found predominantly in the medulla, where it is thought to stimulate catecholamine secretion (Peach et al, 1971). ACE is found abundantly in the testes and prostate, in the Leydig cells, and also in cytoplasmic droplets in sperm (Pandey et al, 1984; Yotsumoto et al, 1984). In the female reproductive tract, ACE is found in follicular and fallopian tube oocytes (Brentjens et al, 1986). The precise role of ACE in the reproductive system has not been elucidated. Several hormones and disease states affect the level and activity of ACE. Corticosteroids, as well as thyroid hormones, stimulate ACE activity (Friedland et al, 1978; Smallridge et al, 1983). The serum ACE level is increased in silicosis, primary biliary cirrhosis, and sarcoidosis (Studdy et al, 1983). As mentioned previously, ACE is not the rate-limiting step in the RAAS cascade; so, changes in serum ACE levels do not directly affect the activity of the systemic RAAS (circulating AII levels).
In addition to ACE, several angiotensinases act in the RAAS to lesser degrees. The physiologic contribution of these enzymes to the function of the RAAS is not clear. Most of these enzymes are present in body tissues such as the kidney. Among these are aminopeptidase A and angiotensinase A, B, and C. Nonspecific angiotensinases hydrolyze AII and angiotensin III (AIII), inactivating them rapidly.
Angiotensin II
The role of the RAAS in the control of blood pressure and extracellular volume is carried out through the integration of a variety of actions performed by AII. Vasoconstriction and the release of aldosterone occur immediately and are of short duration, supporting the role of AII in maintaining tissue perfusion in hypovolemia. Other actions such as vascular growth and ventricular hypertrophy are slower in onset and longer in duration, lasting for several days or weeks.
Effect of Angiotensin II on Glomerular Circulation
One of the most important actions of AII is the autoregulation of the GFR in response to changes in renal perfusion. These are affected through changes in vascular resistance as well as mesangial cell tone. AII causes a marked increase in efferent arteriolar resistance in cases of renal hypoperfusion but does not affect afferent arteriolar resistance unless there is an increase in renal perfusion pressure. The result of this disproportionate increase in efferent over afferent resistance is an increase in capillary hydraulic pressure, and subsequently in filtration pressure, maintaining the GFR in the face of decreased renal perfusion (Hall et al, 1977). It is through inhibition of this action that ACE inhibitors result in a decrease in GFR in cases of renal artery stenosis. This effect of AII on the glomerular circulation is thought to be mediated through differential induction of vasodilatory prostaglandins from the afferent and efferent vessels (Hura and Kunau, 1988).
In addition to its effects on the glomerular vessels, AII directly results in mesangial cell contraction, leading to a decrease in the filtration coefficient of the glomerulus (Blantz et al, 1976).
Tubular Effects of Angiotensin II
AII-induced increases in the filtration fraction lead to an increase in the oncotic pressure in the postglomerular vessels. This leads to an increase in fluid reabsorption in the proximal tubules. AII receptors are also present on the proximal tubule brush border and basolateral sides, and AII is produced in large amounts locally within the proximal tubules. AII is present within the proximal renal tubule in much higher concentration than in the plasma (Seikaly et al, 1990). The effect of AII on sodium reabsorption is bimodal; physiologic concentrations of AII stimulate sodium reabsorption in the proximal tubule, whereas higher concentrations inhibit sodium transport (Harris and Young, 1977).
Medullary Effects
AII decreases medullary blood flow, leading to increased medullary hypertonicity and concentration of urine (Arendshorst and Finn, 1977).
Vascular Effects
AII raises blood pressure by increasing peripheral vascular resistance through a direct effect on vascular smooth muscle cells, causing them to contract. Medium-sized and small arteries are more responsive to AII than large vessels. Contraction occurs mainly in the vessels of the kidney, skin, mesentery, coronary arteries, and brain. Vessels of the lung and skeletal muscle are less responsive to AII. In addition to vasoconstriction, AII stimulates vascular smooth muscle cell growth, leading to a hypertrophic response (Geisterfer et al, 1988). Such smooth muscle proliferation results in left ventricular hypertrophy in cases of chronic stimulation of the RAS. AII is also involved in inflammatory processes including atherosclerosis. This cascade of cardiovascular events including hypertension, ventricular hypertophy, and atherosclerosis is felt to be central to the development of heart failure. With this mechanism in mind, interruption of the RAAS through multiple pharmacologic channels has become a major objective in lowering cardiac mortality associated with hypertension and heart failure.
Adrenal Effects
AII acts directly on the adrenal glomerulosa cells to stimulate aldosterone secretion. This is accomplished through increased desmolase activity and increased conversion of corticosterone to aldosterone (Aguilera, 1993). This augments the salt reabsorptive actions of AII to conserve sodium.
CNS Renin-Angiotensin-Aldosterone System
The CNS is affected mainly by the local renin-angiotensin system, but high circulating levels of AII may also affect CNS function. Central AII results in an increase in blood pressure as well as increased drinking and salt appetite (Sweet et al, 1971; Fitzsimons, 1980). Central AII also leads to increased secretion of corticotropin, prolactin, luteinizing hormone, oxytocin, and vasopressin (Unger et al, 1988).
Gonadal Renin-Angiotensin-Aldosterone System
Gonadal RAAS is present in both the testis and the ovary. The function of testicular RAAS is not clear; in the ovary, RAAS may play a role in oocyte maturation.
Angiotensin II Receptor Subtypes
Nonpeptide receptor antagonists have provided definite proof of at least two major angiotensin receptor subtypes, named AT1 and AT2. Both receptors are polypeptides containing 360 amino acids spanning the cell membrane several times. They are functionally distinct with a sequence homology of 30%. The gene for the AT1 receptor is located on chromosome 3, and the gene for the AT2 receptor is located on the X chromosome (Goodfriend et al, 1996). AT1 receptors are blocked by DuP 753 (losartan), and AT2 receptors are blocked by tetrahydroimidazopyridines such as PD 123177. AT1 receptors have a higher affinity for AII than AIII, but AT2 receptors bind both AII and AIII equally. AT1 receptors have been further subtyped into two isoforms, AT1A and AT1B, although the function of the subtypes is not clear.
The AT1A receptor is expressed in the liver, kidney, aorta, uterus, adrenals, ovary, spleen, and lung as well as in the hypothalamus. The AT1B receptor is expressed in the pituitary, adrenals, kidney, uterus, and liver and is absent in the heart, brain, and spleen. In fetal life, the AT2 receptor is widely present in the adrenals, kidney, liver, skin, tongue, and brain. In the adult, this distribution becomes restricted to the adrenals, uterus, ovary, heart, and some nuclei in the brain.
In the kidneys, AT1 receptors are located predominantly in the glomeruli and tubulointerstitium, whereas AT2 receptors are located in the large cortical blood vessels (Goldfarb et al, 1994).
Almost all the vascular effects of AII, including vasoconstriction, aldosterone release, and β-adrenergic stimulation, are mediated by the AT1 receptor (Timmermans et al, 1992). The development of AT1 receptor antagonists (e.g., losartan) has produced a new class of drugs, as well as an effective tool for blocking the RAAS in a variety of disease states, including hypertension. Further, these new drugs modulate cardiac and renal injury responses to disease.
The function of the AT2 receptor has not been fully defined; however, it may act in a manner antagonistic to the AT1 receptor, especially in the cardiovascular system, where it exerts antiproliferative, antihypertrophic, and proapoptotic functions (Horiuchi et al, 1999). AT2 receptors are thus thought to mediate protective actions that counterbalance the potentially harmful actions mediated through the AT1 receptors. AT2 receptors are also believed to play a crucial role during gestational growth and development, mainly due to the widespread distribution of these receptors in most body tissues during fetal life. Reexpression of these receptors in adult life occurs as a response to vascular injury or inflammation (Horiuchi et al, 1999).
The signal transduction mechanism initiated by binding of AII to the AT1 receptor has been well described; however, the signal transduction mechanism for the AT2 receptor is not yet known. Binding of AII to the AT1 receptor leads to the dissociation of subunits of a guanine nucleotide–binding protein, which activates phospholipase C to generate diacylglycerol and inositol triphosphate. Inositol triphosphate releases calcium from the endoplasmic reticulum, and AII also increases calcium entry through the cell membrane. The intracellular calcium, as well as diacylglycerol, activates protein kinase C and other enzymes that phosphorylate protein and ultimately regulates the specific cellular function induced by AII (Goodfriend et al, 1996).
Other Angiotensins
The parent peptide of the angiotensin family is the decapeptide AI. Several other peptides are formed within the RAAS, some of which have weak activity compared with AII, and some of which have undetermined activity. As previously mentioned, AII (also called angiotensin 1-8) is the major active peptide in the system; it is an octapeptide formed by the removal of terminal histidine and leucine from the carboxy terminus of AI. AIII (or angiotensin 2-8) is similar to AII but lacks the aspartyl amino acid at the amino terminus of the polypeptide chain. It can be formed from AII or directly from AI. Angiotensin 1-7 lacks the three amino acids at the carboxy terminus of AI and has undetermined receptor activity. AIV is a hexapeptide lacking the two terminal amino acids at both ends of the AI polypeptide chain (Goodfriend et al, 1996).
The actions of angiotensin 1-7 have been defined. It appears to be formed from AI directly by a different enzyme than ACE, called neprilysin. ACE inhibitors thus increase the levels of circulating angiotensin 1-7. It acts in an opposing fashion to AII, producing vasodilatation and natriuresis, and also has antiproliferative effects on vascular smooth muscle (Chappell and Ferrario, 1999).
Pathophysiology of Renovascular Hypertension
The classic experiments on RVH were performed by Goldblatt and colleagues (1934), who demonstrated that hypertension could be produced by constricting the renal artery in the dog. Two models of experimental Goldblatt hypertension are described: the two-kidney, one-clip (2K,1C) model, in which one renal artery is clipped and the contralateral kidney is in place and normal; and the one-kidney, one-clip (1K,1C) model, in which one renal artery is clipped and the contralateral kidney is removed. RVH results in both models, but the evolution and the pathophysiologic mechanisms are different. These models provide the basis for understanding the mechanism and evolution of RVH in humans.
Two-Kidney, One-Clip Model
In the two-kidney, one-clip model, the renal artery to one kidney is clipped, resulting in ischemia of the clipped kidney. The RAAS is activated as a result of renal hypoperfusion, resulting in generalized vasoconstriction and systemic hypertension. The adrenal cortex is also stimulated, resulting in secondary hyperaldosteronism and promoting sodium retention by the stenotic kidney. This is the early phase of RVH and is totally mediated by high circulating levels of AII. The normal contralateral kidney is subjected to higher-than-normal perfusion pressure and reacts by suppression of renin secretion as well as “pressure” natriuresis, excreting higher-than-normal levels of sodium and water. Renal vein renin (RVR) from the normal kidney is equal to the arterial value, indicating no secretion by the kidney. In this manner, both kidneys work against each other, with the normal kidney preventing the systemic blood pressure and sodium content from reaching levels high enough to suppress renin release from the stenotic kidney.
Briefly, the two-kidney, one-clip model is characterized by the unilateral release of renin from the ischemic kidney accompanied by contralateral suppression of renin release from the normal kidney, sodium retention by the stenotic kidney, and excretion by the contralateral kidney; euvolemia; and hypertension dependent on AII-induced vasoconstriction. Accordingly, unclipping of the ischemic kidney, ACE inhibitors, or AII antagonists result in a marked decrease in blood pressure.
One-Kidney, One-Clip Model
In the one-kidney, one-clip model, one renal artery is clipped and the contralateral kidney is removed. The solitary ischemic kidney secretes renin, activating the RAAS and resulting in systemic hypertension. Owing to the absence of the normal contralateral kidney, pressure natriuresis does not occur, and the stenotic kidney avidly conserves sodium and fluid, producing volume expansion. The elevation of blood pressure, sodium retention, and volume expansion gradually suppress renin release from the ischemic kidney. Accordingly, although the generating mechanism of hypertension is similar in both models, hypertension in the one-kidney, one-clip model is largely maintained by volume and sodium excess, in the face of normal circulating AII levels. ACE inhibitors or AII antagonists do not result in marked decrease of blood pressure. Under conditions of sodium depletion, hypertension once again becomes dependent on AII, with a marked response to ACE inhibition.
In addition, both models do not remain static but, rather, pass through an acute phase, a transitional phase, and then a final chronic phase (Table 39–3). In cases of two-kidney, one-clip hypertension, after several days or weeks a chronic phase is eventually reached in which unclipping of the stenotic kidney fails to normalize blood pressure. In this chronic phase, the elevated perfusion pressure, as well as high levels of AII, results in widespread arteriolar damage to the contralateral kidney. The excretory function (natriuresis) of the contralateral kidney declines, resulting in extracellular volume expansion, a decrease in circulating AII levels, and the gradual development of a “volume-dependent” type of hypertension. ACE inhibition or removal of the stenotic kidney fails to cure the hypertension in this phase of the disease unless sodium depletion is instituted. Systemic vasoconstriction continues to play a role in maintaining hypertension in the chronic phase, with increased sensitivity to AII, increased vasopressin secretion, and increased sympathetic nervous system activity.
Table 39–3 Phases of Experimental Renovascular Hypertension
| Acute Phase |
| Renin dependency |
| Transitional Phase |
| Chronic Phase |
Human Correlates of Experimental Renovascular Hypertension
The situation in cases of human RVH is not as simple as the experimental models, but in most cases human unilateral RAS resembles the two-kidney, one-clip model. A similar sequence of events ensues, with activation of the RAAS resulting in hypertension and secondary hyperaldosteronism, sometimes resulting in hypokalemia. Relief of the stenotic lesion by revascularization or nephrectomy carries a higher chance of amelioration of hypertension if carried out before the development of a chronic phase in which parenchymal damage in the contralateral kidney maintains the blood pressure elevation.
Bilateral renal artery stenosis in humans does not clearly follow either experimental model but rather is a mixed picture with some characteristics of each. In this respect, a beneficial blood pressure response is seen with ACE inhibition, indicating overactivity of the RAAS, and there is also evidence of volume expansion with frequent pulmonary edema, as well as diuresis after revascularization. Different reasons for this mixed picture are possible, mainly owing to the asymmetrical development of renal artery stenosis, starting with unilateral disease and progressing to bilateral disease. Undetermined renal parenchymal damage may occur to the contralateral normal kidney before the onset of bilateral disease as well. Volume overload would then be exacerbated by the development of bilateral disease.
The one-kidney, one-clip model clinically resembles cases of stenosis to a solitary functioning kidney, unilateral renal artery stenosis with parenchymal damage to the contralateral kidney (nephrosclerosis or atheroembolism), and transplant renal artery stenosis.
Pathophysiology of Ischemic Nephropathy
In addition to RVH, a second, equally important phenomenon resulting from renal artery stenosis is deterioration of renal function, termed ischemic nephropathy (IN). This is a clinical syndrome that occurs through different pathophysiologic mechanisms, is distinct from RVH, and can occur in the absence of elevated blood pressure.
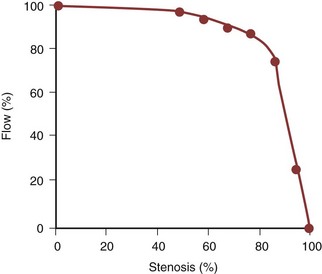
IN is the result of chronic hypoperfusion of the total functioning renal mass. This occurs in the setting of bilateral severe stenosis or stenosis to a functionally or anatomically solitary kidney. The pathophysiology of renal injury as a result of chronic ischemia is poorly understood. This injury is not simply cell death due to a lack of oxygen and nutrients, because the oxygen demand of the kidney never exceeds the supply. Experiments studying the effects of acute renal ischemia do not lend themselves to the explanation of chronic ischemic injury. For ischemic injury to occur, the reduction in renal blood flow needs to exceed the compensatory ability of the kidneys. Renal autoregulation fails to maintain the GFR when renal perfusion decreases below 70 to 80 mm Hg. This occurs when the luminal diameter of the renal artery is stenosed by more than 70% of the original size. At this point, the stenosis becomes hemodynamically significant, resulting in a gradual deterioration of the GFR with an accompanying rise in the serum creatinine level (Fig. 39–7).
Critical reduction of renal blood flow results in IN without affecting renal viability, because renal blood flow is severalfold higher than blood flow to other organs such as the liver or heart. This flow rate far exceeds the needs of the kidney for oxygenation but is necessary to drive glomerular filtration. It is estimated that the kidney needs only about 10% of its blood flow to maintain its oxygen requirement. Under conditions of chronic ischemia, collateral circulation to the kidney develops from the adrenal, lumbar, and ureteric vascular beds and can sustain renal viability even in cases of complete occlusion of the renal artery.
Reduction of renal blood flow activates the RAAS to produce angiotensin II, which maintains glomerular capillary hydrostatic pressure (and GFR) through efferent arteriolar vasoconstriction. Reduction of renal blood flow also leads to redistribution of blood within the kidney and diminished cortical blood flow to prevent medullary anoxia.
The cellular mechanisms by which a decrease in renal size and IN develop are not well understood. Several mechanisms play a role, including vascular mediators (endothelin, thromboxane, prostacyclin, and nitric oxide); calcium accumulation in or adenosine triphosphate (ATP) depletion of the ischemic cells; production of oxygen free radicals; or disruption of cellular membrane polarity (Textor, 1994). The exact role played by each of these mechanisms is not well defined. The action of repetitive cycles of hypoperfusion may allow renal injury to continue before repair is complete, whereas a disturbance in cellular repair processes may hinder recovery between these episodes (Textor, 1996). The point at (or mechanism through) which these changes become irreversible after relief of renal ischemia is even more poorly understood.
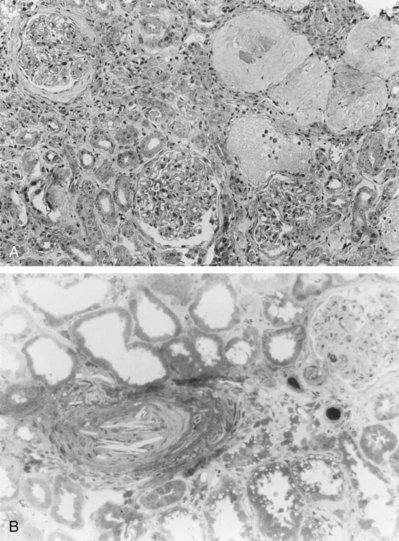
Although the exact mechanisms through which chronic ischemic injury of the kidneys occur are not clear, the resulting structural changes within the chronically ischemic kidney have been well identified. Tubular changes are usually most prominent, in the form of patchy tubular necrosis and atrophy. Glomeruli decrease in size, with wrinkling of the glomerular tuft and thickening of the Bowman capsule. Localized or global glomerular sclerosis is also seen. Hypercellularity of the juxtaglomerular apparatus is commonly seen. Blood vessels show the effects of comorbid conditions, such as essential hypertension, diabetes, and hyperlipidemia, with arteriolar thickening and hyalinosis.
Atheroembolism (Cholesterol Embolism)
Renal cholesterol embolism occurs most commonly in older hypertensive patients, with severe abdominal aortic ASO as the most common associated underlying cause, and contributes significantly to renal dysfunction in cases of IN (Vidt et al, 1989). Atheroembolism can occur spontaneously but more commonly follows manipulation of the atherosclerotic aorta by surgery, angiography, or the use of thrombolytic agents. In a review of 221 cases of cholesterol embolism, 69% were spontaneous, and an inciting event precipitated embolism in 31%. Renal manifestations were present in 50% of cases, and the mortality rate was 81% (Fine et al, 1987).
Atheromatous fragments lodged in blood vessels are highly thrombogenic, leading to occlusion of the vessels in which they lodge, and, at the same time, an inflammatory reaction and fibrosis are incited. Cholesterol crystals can remain histologically detectable in blood vessels for up to 9 months after the event. Diagnosis of atheroembolism is made by examination of biopsy samples from the affected tissue, most commonly skin, muscle, or kidney. Cholesterol microemboli are seen within the renal vasculature (needle-shaped cholesterol crystals, which appear birefringent on frozen section and are dissolved by the solvents used in permanent sections, leaving empty cholesterol clefts) and generally portend a poorer prognosis. In a study of 44 patients from the Cleveland Clinic, findings reflective of atheroembolism were identified on intraoperative biopsy of 16 patients (36%) at the time of open surgical renal revascularization. Patients with atheroembolism had a significantly decreased 5-year survival (54% versus 85%), compared with patients who did not exhibit histologic evidence of atheroembolism. Patients with atheroembolism had a significantly higher incidence of postoperative atherosclerotic complications as well as renal and renovascular complications (Krishnamurthi et al, 1999).
The organs most commonly affected by atheroembolism are the kidney, spleen, pancreas, and gastrointestinal tract. Virtually all organs can be affected, however, leading to multisystem organ disease. Cutaneous manifestations are the most common extrarenal manifestation, in the form of livedo reticularis (lacy bluish discoloration affecting the lower extremities), digital cyanosis or gangrene, ulceration, or subcutaneous nodules. Retinal emboli (Hollenhorst plaques) can occur, leading to visual symptoms, or can be silent.
Renal affection takes the form of deteriorating renal function, usually after a precipitating event. Decline of renal function can vary in severity from mild to rapid acute renal failure. Gradual improvement in renal function occurs after the event, but recurrent episodes lead to progressive loss of renal function with time (Siemons et al, 1987). Symptoms of renal involvement are usually absent, but new-onset hypertension or worsening of preexisting hypertension may occur.
A preventive approach to atheroembolism entails avoiding unnecessary or rough manipulation of atherosclerotic vessels, as well as avoiding prolonged anticoagulation in patients at risk for developing atheroemboli. Management of cholesterol embolism is supportive, with removal of the inciting trauma if present, cessation of anticoagulation, control of hypertension, and institution of renal replacement therapy as needed. Increased awareness of this condition has led to early diagnosis and institution of aggressive supportive care, resulting in a decline in mortality from the disease. A 1999 study of intensive multiorgan support for 67 patients reported a mortality rate of only 16%, with 32% of the survivors requiring long-term dialysis therapy (Belenfant et al, 1999).
Key Points: Ischemic Nephropathy
Clinical Features of Renovascular Hypertension
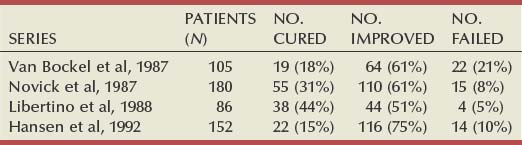
Although RVH is the most common correctable cause of secondary hypertension (with the exception of hypertension due to oral contraceptive use), the prevalence of RVH is probably less than 1% for patients with mild or moderate hypertension (Lewin et al, 1985). For this reason, before subjecting patients to numerous diagnostic procedures that are potentially invasive and costly, enough clinical suspicion needs to be generated to prevent unnecessary investigations of the patient with essential hypertension. Patients with RAS may also present with renal impairment (IN) in the presence or absence of RVH.
Clinical Clues
Symptoms suggestive of RVH are rare, with the exception of flank pain due to segmental infarction or arterial dissection, and generalized nonspecific symptoms in cases of Takayasu arteritis. The clinical course of hypertension, however, may be suggestive of a renovascular cause.
The age at presentation is a clue. The onset of hypertension before the age of 30 years or after the age of 55 years is more commonly associated with renovascular disease, typically FD in young patients and ASO in those older than 55 years. In the Cooperative Study for Renovascular Hypertension, the average age at the onset of essential hypertension was 35 years; for hypertension secondary to FD, the average age of onset was 33 years; and for atherosclerotic renal artery stenosis (ASO-RAS), the average age of onset was 46 years (Maxwell et al, 1972).
A family history of hypertension suggests essential hypertension, although there are reports of familial fibromuscular disease of the renal arteries, especially in females (Major et al, 1977; Pannier-Moreau et al, 1997).
Sudden onset and shorter duration of hypertension is usually associated with RVH; it may also be associated with a better chance of cure after treatment.
Hypertension that is difficult to control on two or three medications is more likely to be associated with renovascular disease. A sudden increase in severity or difficulty of control of previous mild or well-controlled hypertension is also suggestive of the development of RVH on top of preexisting essential hypertension.
Accelerated, malignant hypertension, or hypertensive crises are more frequently associated with RVH than essential hypertension (Simon et al, 1972).
Hypertension associated with episodes of pulmonary edema, evidence of generalized atherosclerotic disease, or gradual impairment of renal function is also suggestive of RVH.
Smoking is a risk factor for developing atherosclerotic disease. In a retrospective study comparing patients having documented RVH with patients having essential hypertension, 88% of patients with ASO-RAS were smokers, compared with 42% of patients with essential hypertension. Patients with FD also showed a higher incidence of smoking (71%) (Nicholson et al, 1983).
On physical examination, clues suggestive of RVH include severe hypertension, the presence of an upper abdominal or epigastric bruit (with both systolic and diastolic components), severe hypertensive retinopathy (grade III or IV), and evidence of generalized atherosclerosis.
Laboratory Investigations
The presence of mild proteinuria is not uncommon in RVH; however, nephrotic-range proteinuria has also been described with RVH (Kumar and Shapiro, 1980; Chen et al, 1995) and has been reversed by renal revascularization (Zimbler et al, 1987). Apart from renal artery disease, proteinuria may be the result of coexisting disease such as diabetes or glomerulosclerosis.
Azotemia in the presence of generalized atherosclerosis, with or without the presence of hypertension, is strongly suggestive of a renal arterial cause.
Hypokalemia (serum potassium level 3.4 mEq/L) especially in the absence of diuretic use is strongly suggestive of RVH resulting in secondary hyperaldosteronism. In the Cooperative Study for Renovascular Hypertension, 16% of patients with RVH were found to have hypokalemia (Maxwell et al, 1972).
Clinical Features of Ischemic Nephropathy
Epidemiologic Considerations
Epidemiologic studies indicate that atherosclerotic renal artery disease is quite common in patients with generalized atherosclerosis obliterans, regardless of whether or not RVH is present. These studies have involved renal angiography in patients with a documented abdominal aortic aneurysm, aorto-occlusive disease, or lower extremity occlusive disease. In such patients, the overall incidence of ARAS has ranged from 31% to 61%, with significant (>50%) ARAS present in 14% to 42% of patients (Table 39–4). In a detailed study by Olin and colleagues (1990), significant ARAS was present in 41 of 108 patients (38%) with an abdominal aortic aneurysm, 7 of 21 patients (33%) with aorto-occlusive disease, and 74 of 189 patients (39%) with lower-extremity occlusive disease.
Table 39–4 Prevalence of Atherosclerotic Renal Artery Stenosis (ARAS) in Patients with Peripheral Vascular Disease
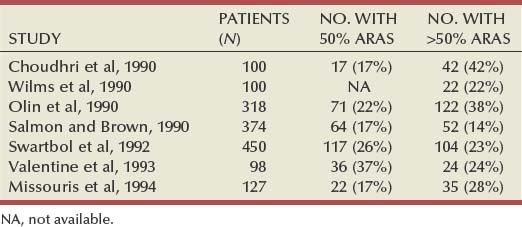
Other studies have evaluated the prevalence of atherosclerotic renal artery disease in patients with coronary artery disease by obtaining abdominal aortography at the time of coronary angiography. Vetrovec and colleagues (1989) noted significant (>50%) ARAS in 22 of 76 patients (29%) with coronary artery disease. In a much larger study, Harding and colleagues (1992) found significant (>50%) ARAS in 164 of 817 patients (20%) with coronary artery disease. In the latter study, the prevalence of significant ARAS was greater in patients with more severe coronary artery disease; significant ARAS was present in 29% of patients with three-vessel disease and in 39% of patients with left-main disease. More recently, the Mayo Clinic group (Rihal et al, 2002) reported a 19% incidence of greater than 50% stenosis, 7% with greater than 70% stenosis, and 3.7% with bilateral stenosis in 297 hypertensive patients undergoing coronary angiography. Similar results are reported by Aqel and colleagues (2003) in a smaller group of 90 veterans with hypertension. A 16% incidence of severe stenosis and 6% incidence of bilateral disease were reported in this study. None of these studies reported an increase in complications related to the addition of abdominal angiography to coronary angiography. This would present a logical rationale for screening the renal arteries in patients with risk factors such as peripheral vascular disease, renal insufficiency, and hypertension.
In a large population-based study of nonselected subjects, Hansen and colleagues (2002) screened 870 patients for renal artery stenosis using duplex ultrasonography. An overall prevalence of 6.8% was noted, with 12% having bilateral disease, and stenosis was associated with hypertension, advancing age, and elevated LDL.
Another study has shown an increased prevalence of ARAS in patients with diabetes mellitus. Sawicki and colleagues (1991) evaluated 5194 consecutive autopsy protocols from patients who died between 1980 and 1988. Significant (>50%) ARAS was present in 4.3% of all patients but in 8.3% of all diabetic patients; all but one of the latter had type II diabetes mellitus. Bilateral ARAS was found in 30% of the nondiabetic patients with ARAS but in 43% of diabetic patients with ARAS. These data suggest that the presence of type II diabetes increases the risk of ARAS and that the latter is more likely to involve both kidneys.
These studies show that ARAS is commonly present in patients with generalized atherosclerosis, particularly those with peripheral vascular disease or coronary artery disease. Although hypertension is also commonly present in such older patients with ARAS, it is far more likely to be idiopathic (essential) than renovascular in origin. Therefore, the prevalence of anatomic ARAS is much greater than that of atherosclerotic RVH.
Screening and Diagnosis
The screening of patients for atherosclerotic renal artery disease is based, in part, on an early study by Gifford and associates (1965). These investigators found that in 53 of 75 older patients (71%) with unilateral renal atrophy, the renal atrophy was caused by stenosing atherosclerotic renal artery disease. Of equal importance was the finding that 22 of these 53 patients (42%) also had unsuspected atherosclerotic renal artery disease involving the opposite, normal-sized kidney. Subsequently, Lawrie and associates (1980) reviewed 40 patients with renal atrophy caused by total arterial occlusion and noted contralateral ARAS in 31 patients (78%). These observations underscore the high incidence of renal artery disease, often bilateral, in patients with generalized atherosclerosis and diminished renal size. The additional finding of even mild azotemia in this setting further enhances the likelihood that underlying large vessel occlusive disease is present.
Harding and associates (1992) evaluated clinical variables associated with ARAS in a study of 1235 patients undergoing simultaneous cardiac catheterization and abdominal aortography. A multivariate logistic-regression analysis identified the following five risk factors as strongly predictive of significant ARAS: higher age, coronary artery disease, a history of congestive heart failure, female gender, and peripheral vascular disease. An elevated serum creatinine level was also predictive of ARAS by univariate logistic-regression analysis. Hypertension was not helpful in identifying patients with ARAS.
Another important clinical clue to the presence of significant ARAS is the development of progressive azotemia after medical control of the blood pressure in patients with significant hypertension. This problem strongly suggests the presence of perfusion-dependent renal function due to significant underlying renal artery obstruction (Textor et al, 1983). In addition to reducing flow across a stenotic renal artery by lowering the systemic blood pressure, antihypertensive medications can impair renal function in such patients through other mechanisms as well. β-Adrenergic blockers produce a fall in cardiac output, which may occasionally diminish effective renal plasma flow and the GFR. ACE inhibitors can lead to deterioration of renal function through loss of efferent arteriolar vasoconstrictor tone in the kidney (Hricik et al, 1983).
Finally, atherosclerotic renal artery disease should also be suspected in older patients with renal insufficiency and no obvious cause for the latter. Corradi and associates (1993) obtained renal angiography in 51 consecutive patients with the following criteria: age older than 60 years, creatinine clearance less than 50 mL/min, no analgesic abuse, proteinuria less than 1 g/day, clinical signs of generalized atherosclerosis, and no biochemical or radiographic findings indicative of glomerulopathy, diabetic nephropathy, polycystic disease, obstructive nephropathy, or pyelonephritis. Angiographic studies revealed significant renal artery stenosis in 29 patients (56.8%); renal artery stenosis was present bilaterally in 10 patients (19.6%) and unilaterally in 19 patients (37.2%).
The studies described indicate that clinical screening for atherosclerotic renal artery disease is appropriate in older patients with most or all of the following features: (1) evidence of generalized atherosclerosis; (2) a decrease in the size of one or both kidneys; (3) renal insufficiency, even of a mild extent, particularly in patients with no obvious underlying cause; (4) the development of progressive azotemia after restoration of normotension with medical antihypertensive therapy; (5) coronary artery disease; (6) a history of congestive heart failure; and (7) peripheral vascular disease. It should be emphasized that patients with atherosclerotic renal artery disease may or may not have significant hypertension, and this should not influence the decision to investigate the patient for this disease.
Diagnostic Evaluation
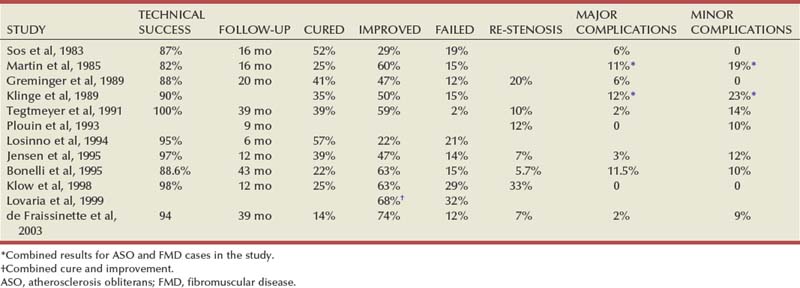
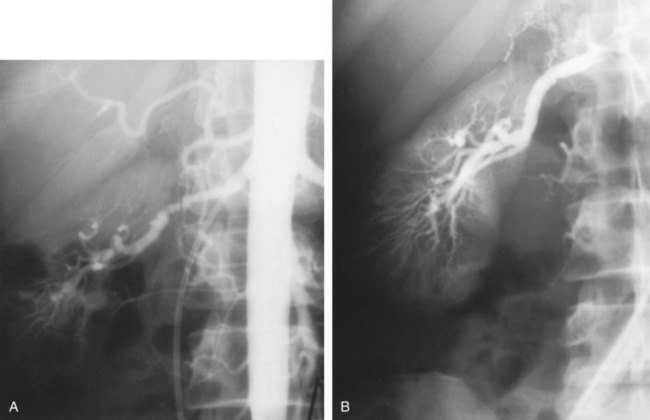
Several noninvasive studies are available to diagnose renal artery stenosis. Most of these studies are intended for hypertensive patients, aiming to identify those patients with a renovascular cause of hypertension. Among these studies are rapid-sequence intravenous urography, ultrasonography, peripheral renin activity assays, captopril testing, and radionuclide renal scanning (with and without ACE inhibition). A variety of modern noninvasive tests have become available and have largely superseded the previously mentioned tests; these are duplex ultrasonography (DUS), magnetic resonance angiography (MRA), and CT angiography (CTA). These tests offer anatomic information only, with no functional information. The definitive diagnosis of RAS, however, is documented by angiographic study of the aorta and renal vessels, which remains the “gold standard” against which all other diagnostic modalities are compared.
Diagnostic evaluation for patients presenting with suspected RVH differs from that of patients presenting with suspected IN. For patients with suspected RVH, a number of tests are available for functional diagnosis of RVH. These tests (plasma renin activity [PRA], captopril test, captopril renography, and RVR assays) diagnose hyperactivity of the RAAS but provide no anatomic information regarding the offending arterial lesion. Captopril renography and RVR assays can localize the ischemic kidney as well. Anatomic delineation of the arterial lesion guides the treatment decisions and is obtained by intra-arterial angiography. A variety of noninvasive anatomic studies (DUS, MRA, and spiral CTA) can be used before angiography in cases in which clinical suspicion of RVH is not confirmed by functional tests.
The pathophysiology of RVH is different and distinct from that of IN. The diagnostic evaluation of patients with suspected IN is hampered by the lack of functional tests and the inability to determine conclusively that an anatomic lesion in the renal artery is responsible for impairment of renal function. Noninvasive anatomic testing is used to confirm a clinical suspicion of renal artery stenosis, which is definitively diagnosed by intra-arterial angiography. Stabilization or improvement of renal function after revascularization remains the final proof of the cause, provided that ischemic damage to the kidney has not become irreversible.
Intravenous Urogram
Until better methods were developed, a modification of the standard intravenous urogram, called the hypertensive, rapid-sequence, or minute-sequence urogram was used as a screening test for RVH. Several findings are suggestive of RVH, including a delayed appearance of contrast material in the calyces of the affected kidney (most important), disparity of renal size of more than 1.5 cm (most common finding), delayed hyperconcentration of contrast material within the affected collecting system, retention of contrast material in a nonobstructed collecting system, and notching of the pelvicalyceal system by collateral vessels.
The poor sensitivity and specificity of the rapid-sequence urogram, as well as the development of multiple other more sensitive diagnostic tests, have led to the discontinuation of its use for diagnosing renovascular disease.
Peripheral Plasma Renin Activity
Measurement of peripheral PRA is a functional test designed to diagnose overactivity of the RAAS. Originally intended as a screening test for RVH, it provides no anatomic information and has no value for diagnosing IN. In order to obtain meaningful results from this test, all antihypertensive medications should be discontinued for 2 weeks, and the PRA should be indexed to the sodium intake. Blood should be collected at noon after 4 hours of patient ambulation. When the test is standardized as mentioned, a sensitivity and specificity of 80% and 84%, respectively, can be expected (Pickering et al, 1984).
Important limitations to this test have restricted its general use. Sixteen percent of patients with essential hypertension have elevated PRA, whereas up to 20% of patients with RVH have normal PRA (Brunner et al, 1972). In addition, discontinuation of all antihypertensive medication in a population of patients with severe, sometimes life-threatening hypertension is not generally feasible.
Captopril Test
Measurement of peripheral PRA before and after an oral dose of captopril is called the captopril test. This is a functional test of RVH that does not provide anatomic information. The test is based on the observation that after the administration of ACE inhibitors, patients with RVH show a higher reactionary rise of PRA than patients with essential hypertension (Case and Laragh, 1979). Patients may continue to take β blockers, but all diuretics and ACE inhibitors need to be discontinued for at least 1 week before the test. A normal- or high-salt diet is needed. Blood should be drawn with the patient in the same position before and after captopril administration, after measurements of blood pressure are stable. An oral dose of 25 mg of captopril is used, and blood is drawn again 1 hour after the dose.
Criteria for a positive test are the presence of all the following: postcaptopril PRA greater than 12 ng/ml/hr, an absolute increase in PRA greater than 10 ng/ml/hr, and a 400% increase in baseline PRA (150% increase if the baseline PRA was more than 3 ng/ml/hr) (Muller et al, 1986). The test is generally safe, with the main risk being an excessive fall of blood pressure in hyper-reninemic patients who are also volume depleted. Overall sensitivity is about 74% and specificity is around 89% (Pickering et al, 1996). The test is not reliable in patients who are azotemic, nor is it sufficiently accurate for use in children (Gauthier et al, 1991).
The low sensitivity of the captopril test makes it unsuitable for use as a general screening test for RVH. The major strength of the captopril test is its accuracy in excluding RVH, especially in patients with low clinical suspicion. The high negative predictive value (approximately 95%) of captopril testing has been confirmed in several studies (Gosse et al, 1989; Svetkey et al, 1989; Frederickson et al, 1990).
Renal Vein Renins
The primary criterion for the functional diagnosis of RVH is hypersecretion of renin from the ischemic kidney combined with contralateral suppression of renin secretion. Calculation of net renin secretion from any kidney is performed by subtracting the renin value in the arterial blood to the kidney (inflow) from the renin value in the venous blood from the kidney (outflow). Because renin values in both the aorta and the inferior vena cava (IVC) are the same, IVC renin is used instead of aortic renin (Sealey et al, 1973). RVR assays are useful for localizing the ischemic kidney in unilateral renal artery stenosis, as well as the more ischemic kidney in bilateral cases. A moderate sodium intake should be maintained at the time of the sampling, which is usually performed with the patient in the supine position. Blood samples from both renal veins as well as from the IVC are obtained.
Hypersecretion of renin from the ischemic kidney (>50% of PRA) confirms the diagnosis of RVH. Contralateral suppression of renin secretion (renal vein − IVC renin = 0) indicates an appropriate response of the normal contralateral kidney to the elevated blood pressure and predicts a cure of hypertension after revascularization. Increasing severity of stenosis reduces blood flow to the ischemic kidney, resulting in an increased RVR increment (after subtracting IVC renin) higher than 50% of the total peripheral PRA (Vaughan et al, 1973). In patients with high PRA and RVR failing to show an increment above 50% from both kidneys, sampling from segmental renal veins may be performed to localize the segment of kidney responsible for hypersecretion of renin (Schambelan et al, 1974). Administration of captopril results in accentuation of renin secretion from the ischemic kidney (similar to the captopril test), which increases accuracy in diagnosing RVH (Simon and Coleman, 1994). This is particularly useful when RVR values are equivocal, in cases of branch stenosis, and in cases of coexisting hypertension or renal disease.
Captopril Renography
Radionuclide renography without ACE inhibition has limited use for the functional or anatomic diagnosis of renovascular disease. The physiologic principle of captopril renography is the loss of preferential vasoconstriction of the efferent arteriole that is mediated by AII and maintains the glomerular pressure gradient in cases of RAS. This loss of postglomerular pressure results in a decreased GFR of the kidney distal to the stenosis, which is measured noninvasively by radionuclide renography.
The study is performed in well-hydrated patients on liberal salt intake. ACE inhibitors are discontinued for 3 to 5 days before the study, but other antihypertensives may be continued (Setaro et al, 1991). Oral hydration is continued on the day of the procedure. Oral captopril (25 to 50 mg) is usually used, although IV enalapril (0.04 mg/kg) can be used as well (Sfakianakis and Sfakianakis, 1988). The captopril renogram is obtained 1 hour after the captopril dose. The use of furosemide has also been suggested to improve the accuracy of ACE renography (Erbsloh-Moller et al, 1991).
Considerable debate continues around the optimal radionuclide agent to use for captopril renography. The most commonly used agents are technetium 99m (99mTc)–diethylenetriaminepentaacetic acid (DTPA) and iodine 131 (131I)–orthoiodohippurate (OIH), with the recent addition of 99mTc-mercaptoacetyltriglycine (MAG3). The imaging characteristics of 99mTc compounds are better than that of 131I compounds. On the other hand, OIH and MAG3 are excreted by both glomerular filtration and tubular secretion, whereas DTPA is excreted by glomerular filtration only, making it less optimal for patients with renal dysfunction. To date, 99mTc-MAG3 has shown the best results for captopril renography, especially in patients with impaired renal function (Dondi, 1991). Studies have shown that 99mTc-ethylenedicysteine (EC) can be used as an imaging agent for captopril renography to diagnose RVH and predict the outcome of revascularization (Ugur et al, 1999).
Captopril renographic diagnostic criteria have not been well standardized. The following are the criteria suggested by the Consensus Panel on Captopril Renography (Nally et al, 1991). Two categories of information are used: asymmetry of renal size and function as suggested by the scintigraphic images, and specific captopril-induced changes in the renogram. These changes on the postcaptopril renogram include a delayed time to maximal activity (>11 minutes), significant asymmetry of peak activity of each kidney, marked cortical retention of radionuclide, and a marked decrease in the GFR of the ipsilateral kidney. For radionuclides with tubular excretion (131I-OIH and 99mTc-MAG3), the ratio of 20-minute counts to peak counts can also be used. Normally less than 0.3, a 0.15 change is considered significant. A small poorly functioning (30%) kidney that shows no change after ACE inhibition, as well as bilateral symmetrical change after ACE inhibition, is considered to be moderately indicative of RVH (Taylor et al, 1998).
The use of AT1 receptor antagonists for hypertension does not preclude the use of captopril renography as a diagnostic test. In a study comparing captopril renography in patients using AT1 receptor antagonists to hypertensive controls, Picciotto and colleagues (2003) found equally accurate results in both groups.
Overall, the sensitivity of captopril renography appears to be approximately 90% to 93%, and the specificity approximately 93% to 98% (if the test is used in patients with a strong clinical suspicion). More importantly, captopril renography is predictive of a cure or improvement in blood pressure after revascularization in 80% to 90% of cases (Nally, 1996). The presence of bilateral renal artery stenosis, renal artery stenosis to a solitary kidney, or impaired renal function (serum creatinine level > 2.5 to 3 mg/dL) decreases the accuracy of captopril renography.
Duplex Ultrasonography
DUS of the renal arteries is a noninvasive anatomic study that has shown an excellent ability to diagnose renal artery stenosis. It combines the use of real-time B-mode renal ultrasonography with color-coded pulsed Doppler to obtain blood flow velocities within the major abdominal vessels. Flow velocity at the renal hilum and inside the renal parenchyma can be measured as well. The basis for diagnosing renal artery stenosis is the altered flow pattern distal to the stenosis, with a turbulent jet during systole and a decrease in diastolic flow. Measurements are obtained at the proximal main renal artery using a standardized angle of incidence, and several indices as well as waveform analysis are used to diagnose stenosis. More than 180 cm/sec peak systolic velocity (PSV) in the renal artery is indicative of RAS (normal renal PSV averages 100 ± 25 cm/sec). PSV has been found to be the most important single indicator of RAS (Miralles et al, 1996). The ratio of the renal PSV to the aortic PSV (renal PSV/aortic PSV) is called the renal aortic ratio (RAR). A ratio of 3.5 or more indicates severe (>60%) stenosis. The use of the RAR is not possible in cases of aortic occlusive or aneurysmal disease because of the abnormal aortic flow patterns in these situations. Measurement of the flow patterns within the distal hilar renal artery branches, as well as within the parenchyma, can be analyzed to provide indices indicative of arterial stenosis or increased renovascular resistance (Nazzal et al, 1997; Riehl et al, 1997). These indices (acceleration time, acceleration index, resistive index) have not proved as universally reliable as the aforementioned (PSV, RAR) measurements. Intraluminal ultrasonography has also been used to measure flow rate directly within the renal artery (van der Hulst et al, 1996; Chavan et al, 1998), and this has been useful to evaluate the flow rate before and after angioplasty (Savader et al, 1998).
Diagnostic categories obtained by DUS include normal, mild stenosis (60%), severe stenosis (>60%), and technically unsatisfactory study (unable to visualize the renal arteries). Renal artery occlusion is indirectly diagnosed by the findings of a small kidney and the inability to detect a renal artery flow pattern.
DUS offers significant advantages as a diagnostic tool for RAS. It is noninvasive, uses portable equipment that is relatively inexpensive and widely available, does not use iodinated contrast material, and has no effect on renal function. Azotemia does not affect the results of the study, and no discontinuation of antihypertensive medications is required. Repeated evaluations can be performed, which is particularly useful for postintervention and follow-up examinations.
The main disadvantage of DUS is its dependence on operator skill, with less reliable results obtained from less experienced vascular laboratories. In obese patients and patients with a lot of bowel gas, the renal arteries may be difficult to visualize. Accessory renal arteries are difficult to visualize, and occluded renal arteries cannot be diagnosed with certainty. Finally, DUS offers anatomic data with no indication of the functional significance of the lesion.
In a prospective study comparing DUS with angiography in 102 patients, DUS was found to have a sensitivity and specificity of 98%, correctly identifying the lesions in 182 of 187 arteries with different degrees of stenosis (Olin et al, 1995). In another prospective study in 41 patients, DUS (compared with angiography) revealed a sensitivity of 95% and a specificity of 90% (Strandness, 1994). In a more recent prospective study evaluating 53 patients with both DUS and angiography, a DUS sensitivity of only 75% was reported, with 103 (of 112) arteries and 12 (of 16) stenoses detected by DUS. Specificity for detecting RAS was 100% (Mollo et al, 1997).
In a study using slightly different diagnostic criteria (PSV > 210 cm/sec) and evaluating 46 patients, the sensitivity and specificity of DUS were 89.5% and 90.7%, respectively. Notably, in the same study, only 23 patients were hypertensive, and captopril renography was positive in only 5 cases (Miralles et al, 1993). The superior utility of DUS over captopril renography reported in this study was not validated in a more recent study prospectively comparing both DUS and captopril renography to angiography in 28 patients. After 11 (of 45) renal arteries were excluded from the study because no Doppler signal could be obtained, the accuracy of both diagnostic modalities was comparable, with a sensitivity and specificity for DUS and captopril renography of 78% and 81%, and 83% and 81%, respectively (Kaplan-Pavlovcic and Nadja, 1998).
Continuous developments in equipment and techniques of DUS are occurring and promise an increased accuracy and utility. Among these developments are the use of power Doppler imaging, three-dimensional imaging, harmonic imaging, and ultrasonic contrast agents. Power Doppler imaging is more sensitive, especially in the detection of low flow rates (Murphy et al, 1997). Three-dimensional imaging uses computer technology to produce a three-dimensional image of the region of interest. Harmonic imaging also improves the imaging of structures that might be moving, like the proximal renal arteries. The use of biodegradable microbubbles in the circulation increases the echogenicity and enhances the visualization of the renal vessels (Missouris et al, 1996).
Magnetic Resonance Angiography
MRA is a modern noninvasive anatomic diagnostic modality that has become available for diagnosing RAS. The techniques used for MRA are time of flight or phase contrast. Gadolinium DTPA enhances the blood signal, resulting in improved images of the aorta and the proximal renal arteries. MRA has the advantage of being noninvasive, not using radiation, having a low technical failure rate, and not using iodinated contrast material, the latter factor making the technique suitable for use in patients with renal insufficiency (Ghantous et al, 1999). Multiple projections can be obtained, and the morphology of the kidney can be evaluated as well. Functional information, including individual renal blood flow and GFR, can also be obtained (Grist, 1994).
The emergence of nephrogenic systemic fibrosis (NSF) with the use of gadolinium MRI contrast agents in patients with renal impairment has received much attention lately and has severely limited contrast MRI use in these patients. This is a rare but potentially life-threatening condition that results from dissociation of gadolinium from its parent compound and chelation with endogenous cations, thus liberating toxic free gadolinium. Affected patients develop multisystem fibrosis affecting primarily the skin, with devastating consequences. Most of the reported cases occurred in patients on dialysis; some studies report that the condition is more likely to occur in patients with acidosis (Broome et al, 2007).
MRA provides an image quality that is inferior compared with angiographic images. Only the proximal parts of the main renal arteries are visualized, without the ability to image the distal arterial tree. MRA is contraindicated in patients with magnetic implants and claustrophobia, and it uses sophisticated expensive equipment that might not be widely available.
MRA was compared with intra-arterial angiography in 103 patients, all of whom had both studies performed. When MRA was used, all main renal arteries and 31 of 33 accessory renal arteries were visualized, and 61 of 65 stenoses were diagnosed and graded correctly. The overall sensitivity was 93% and the specificity was 90% (Hany et al, 1998). Another study evaluated 62 patients with both MRA and angiography (Thornton et al, 1999). MRA had a sensitivity of 88% and a specificity of 98% and visualized 93% of all the arteries seen on angiography. The reproducibility of interpretation and interobserver variability of MRA were found to be comparable with those of conventional angiography in 54 patients who underwent both diagnostic procedures (Gilfeather et al, 1999).
A prospective study compared gadolinium-enhanced MRA to color DUS in 45 patients, and the results of both examinations were referenced to angiography in all cases (De Cobelli et al, 2000). Out of 13 accessory renal arteries detected by digital subtraction angiography (DSA), 12 were detected by MRA and only 3 were detected by DUS. The sensitivity and specificity of MRA were 94% and 91% for diagnosing all degrees of stenosis, whereas the sensitivity and specificity of DUS were 71% and 76%. For the detection of stenoses of more than 50%, the corresponding values were 93% and 95% for MRA, compared with 93% and 89% for DUS. Another group (Leung et al, 1999) compared the two modalities (and referenced results to DSA studies) and reported similar results, with slightly better sensitivity and specificity for MRA (90% and 86%) compared with DUS (81% and 87%). This study also confirmed the superior ability of MRA over DUS in detecting accessory renal arteries (96% vs. 5%). MRA was more sensitive in detecting RAS due to ASO than in detecting RAS due to FD, according to this study; this could be related to the more distal nature of most FD lesions. MRA can also diagnose renal artery aneurysms, and is useful for following the size of renal artery aneurysms over time, although aneurysms distal to the primary branches of the renal artery are not well visualized on MRA (Browne et al, 2004).
Improved techniques and increased experience with MRA continue to increase its accuracy and utility. The use of time-resolved imaging to decrease artifacts, decrease venous overlap, and improve spatial resolution and contrast enhancement has been reported (Van Hoe et al, 2000). Gadolinium has been administered intra-arterially in vitro in an attempt to improve visualization of blood vessels (Omary et al, 1999). Multiecho gradient echo technique for three-dimensional MRA has been reported to produce high vessel-to-background contrast with decreased bowel-related artifact (Papachristopoulos et al, 1999). Although the recognition of nephrogenic systemic fibrosis (NSF), a serious complication of using gadolinium-based contrast agents for MRI in patients with renal insufficiency, has hampered the use of MRI in many patients with renal artery stenosis, several precautions have been recommended that may allow MRI contrast agents to be used with minimal risk. Cyclic gadolinium–based contrast agents have less chance of causing NSF if using smaller doses of contrast, and if avoiding gadolinium agents altogether in patients with estimated GFR of less than 30 mL/min or in patients with metabolic acidosis (Boyd et al, 2007; Othersen et al, 2007).
MRI has the potential to provide other important information, in addition to anatomical depiction of the arterial stenosis. Gadolinium clearance can be used to measure single-kidney glomerular filtration rate, and renal volume can be measured and followed to evaluate recovery after revascularization. Loss of corticomedullary differentiation and increased cortical-medullary transit time can also indicate renal ischemia, and reversal of these findings may indicate successful revascularization (Chrysochou et al, 2008).
Computed Tomography Angiography
The availability of spiral CT technology with rapid acquisition time has made possible imaging of the renal arteries. Scanning through the area of the renal arteries using thin (2-mm) slices during the arterial phase of contrast material injection is performed while the patient holds a single breath. Three-dimensional reconstruction of the axial image is then carried out, showing the abdominal aorta with its main branches. Atherosclerotic disease affecting the aorta and the renal arteries can be visualized, as well as renal morphology and parenchymal lesions. Spiral CTA lacks the ability to define disease distal to the main stem renal artery, and a relatively large volume of intravenous (IV) iodinated contrast material is required to perform the study; however, CTA might offer the advantages over MRA of cost, convenience, and widespread availability (Prokop et al, 1998).
Comparison between CTA and DSA in 82 patients revealed a sensitivity and specificity of 96% and 99%, respectively, for hemodynamically significant stenosis, with only 1 stenosis (of 34) missed on CTA. Thirty-three accessory renal arteries were visualized in this study, as well as 5 adrenal masses (Wittenberg et al, 1999). In another study evaluating 50 patients with suspected RAS, CTA showed a sensitivity of 90% and a specificity of 97% and demonstrated 27 of 28 accessory renal arteries (Kim et al, 1998). Modern CT technology can also be used to perform virtual endoscopy, which can be helpful for evaluating renal artery stenoses and positioning arterial stents (Neri et al, 2000). Multiple detector–row CT angiography is a new method of CT angiography that also promises more accurate depiction of the renal and other visceral arteries (Fleischmann, 2003; Pannu et al, 2002).
In order to clarify the various limitations of noninvasive imaging modalities in respect to accessory renal arteries as well as proximal branches of the renal artery, a study compared DUS, CTA, and MRA to angiography. The points of comparison were the ability to evaluate accessory renal arteries and proximal branches. Fifty-six patients underwent angiography; of those patients, 45 underwent DUS, 52 underwent CTA, and 28 underwent MRA. When angiography was used, 28 accessory renal arteries as well as 21 proximal (>2 cm from the aorta) renal artery branches were visualized. DUS depicted 21% of the accessory arteries and 0% of the proximal branches. MRA depicted 73% of the accessory arteries (and four additional arteries not seen on angiography), and 0% of the proximal branches. CTA depicted 92% of the accessory arteries and 76% of the proximal branches. Although experience with modalities differs among institutions, this study offers an interesting comparison of the limitations of these techniques (Halpern et al, 1999).
In a more recent study comparing MRA against CTA with intra-arterial DSA as the reference standard, there was no significant difference in the accuracy of both MRA and CTA, with sensitivity around 92% and specificity around 99% for both modalities. Patient acceptance was also measured, and estimated to be best for CTA, although the time consumed for reconstruction of the images was most in CTA (Willmann et al, 2003).
Contrast Arteriography
Intra-arterial angiography remains the gold standard for diagnosing renal artery disease, and it is the test against which the results of other tests are compared. The availability of modern interventional techniques (angioplasty and arterial stents) has allowed angiography to become a combined diagnostic and therapeutic procedure. However, for several reasons, angiography is not suitable for use as a preliminary screening tool for all patients suspected of having renal artery stenosis. It is an expensive test that cannot be done on an outpatient basis. It is also an invasive test using ionizing radiation and requiring arterial puncture, manipulation of arterial catheters, and injection of iodinated contrast material.
Complications of arterial puncture and manipulation include bleeding, hematoma, dissection, thrombosis, and distal embolization of atherosclerotic plaque, as well as cholesterol embolization. The use of iodinated contrast material carries the risk of allergic reaction and volume overload. The contrast load also leads to a transient impairment of renal function, especially in patients with preexisting renal insufficiency and in diabetic patients. Several advancements in the field of angiography have been directed at decreasing side effects from iodinated contrast material. These advancements include decreasing the contrast load through digital postprocessing, as well as the use of contrast agents with decreased nephrotoxicity, such as carbon dioxide or gadodiamide (Spinosa et al, 1999). The advent of digital subtraction angiography (DSA) has allowed reduction of the contrast load with a concomitant reduction in catheter size. Although the spatial resolution of DSA is lower than that of conventional angiography, the contrast resolution of DSA is superior. The ability to subtract bone and soft tissue with DSA is an important advantage, making conventional cut film angiography obsolete, and DSA the most commonly used technique today.
Early in the development of digital techniques, it was suggested that IV studies (IV-DSA) would be accurate and supersede the arterial approach (Wilms et al, 1986). The attraction of visualizing the renal arteries angiographically with an outpatient procedure was not realized, however. Several disadvantages led to the gradual abandonment of IV-DSA in favor of intra-arterial studies. These disadvantages include the need for a large contrast material volume, the frequent need for central venous catheters to deliver this large volume, poor image quality owing to overlying visceral arteries, frequent uninterpretable studies, and poor opacification, especially in elderly patients or those with cardiac dysfunction (Buonocore et al, 1981; Smith et al, 1982).
Carbon dioxide has been introduced as a contrast agent for intra-arterial injection in an effort to reduce contrast nephrotoxicity from iodinated contrast material. An injected bolus of carbon dioxide displaces blood from the vessels to be imaged and provides sufficient contrast for adequate imaging using DSA technology and postprocessing enhancement. The injected carbon dioxide is cleared by the lungs without deleterious effects. Carbon dioxide has no effect on renal function, making it an ideal agent for use in patients with renal insufficiency. It is nonallergenic, cheap, and presents no problems with fluid overload. Smaller softer arterial catheters can be used for injection, minimizing trauma to the arterial walls. This technique can also provide information that is not visualized using standard iodinated contrast material, including small arteriovenous shunts, small tumor vessels, and minute amounts of arterial bleeding (Hawkins et al, 1994).
In a prospective study comparing carbon dioxide angiography with standard contrast arteriography in 100 patients, the overall accuracy of carbon dioxide DSA was 97% (Schreier et al, 1996). Carbon dioxide DSA was successfully used for guiding angioplasty and renal artery stenting in 17 patients in another study, with only one transient rise in the serum creatinine level (Caridi et al, 1999). Similarly, no cases of contrast nephropathy were reported in a study employing carbon dioxide DSA as the main imaging technique for patients with renal impairment (Fitridge et al, 1999). Carbon dioxide renal angiography has been used safely in children (Kriss et al, 1997). Although carbon dioxide is generally regarded as a benign contrast medium, a case of fatal complications (rhabdomyolysis and intestinal infarction) after carbon dioxide angiography has been reported (Rundback et al, 1997).
Ailawadi and colleagues (2003) evaluated the use of gadolinium as a contrast agent for intra-arterial angiography in patients with renal insufficiency and found it to be safe and adequate for imaging the renal arteries as well as first- and second-order branches. No comparison with conventional contrast angiography was performed, but this might be an option to decrease nephrotoxic side effects of angiography. Significant concern about NSF in patients with renal insufficiency will undoubtedly limit the use of gadolinium-based contrast agents in patients with renal impairment.
Cost-Effective Approach for Diagnosis
The diagnostic approach for patients with renal artery stenosis should be tailored according to the predominant clinical picture, that is, patients with IN should be worked up differently from patients with hypertension as the presenting clinical problem. As mentioned previously, patients with IN constitute the majority of patients presenting for treatment today, may not have hypertension as part of the clinical picture, and cannot be diagnosed by functional testing. In these patients, an anatomic diagnosis is pursued directly. Patients with a strong suspicion of renal arterial disease should probably undergo intra-arterial angiography (iodinated contrast material or carbon dioxide) directly. Patients with mild or moderate suspicion of having renal artery stenosis should undergo a noninvasive anatomic test (DUS, MRA, or CTA). The choice of diagnostic procedure depends on patient’s level of renal function (azotemic patients more suitable for DUS than CTA), as well as the experience with the various modalities at different centers. Positive findings on noninvasive testing should lead to definitive confirmation of the lesion and therapeutic planning through intra-arterial angiography. Negative findings on these tests should be interpreted with understanding of the limitations of each technique and should lead to further noninvasive testing if technical factors are suspected, or to no further testing if a technically perfect study is clearly negative in a patient with minimal clinical suspicion.
Patients with suspected RVH present a different diagnostic challenge. Functional evaluation of the RAAS is possible and is usually performed as a first step in the diagnosis before anatomic diagnosis. The steps used to screen and select patients for further study differ according to different centers and have changed over the last years, mainly owing to the introduction of reliable noninvasive anatomic imaging techniques.
Patients should be ranked according to the clinical findings suspicious for RVH into patients with low, moderate, and high suspicion. Like patients with IN, patients with a high suspicion for RVH are probably best served by performing angiography directly. Negative findings on other studies would not prevent these patients from progressing to angiography in any case. In bilateral cases, RVR assays can be used to localize the more ischemic side.
Patients with low or moderate suspicion of RVH are more problematic. In these cases, it is probably reasonable to perform captopril renography as a preliminary test. A positive test should be followed by further testing, preferably definitive diagnosis through angiography. Patients with low clinical suspicion and a technically satisfactory negative study should not be subjected to further testing. Technically unsatisfactory studies should probably be followed by a noninvasive test of a different type (e.g., DUS). These tests (DUS, MRA, or CTA) will provide no functional information or prediction of cure after intervention. To date, anatomic confirmation of the disease and treatment planning still require intra-arterial angiography.
The role of renin assays has decreased significantly since the 1980s. Few centers use the captopril test to exclude patients from having RVH, because of its consistently high negative predictive value. RVR assays are rarely used to diagnose RVH before a noninvasive test indicates the anatomic presence of stenosis; their role has focused on localizing the more ischemic kidney in bilateral cases.
Key Points: Diagnostic Evaluation
Renovascular Disease in Children
Renovascular disease is estimated to cause 5% to 10% of childhood hypertension (Wyszyńūska et al, 1992). The causes of renovascular hypertension in children are more variable than adults, and diagnosis is usually more difficult, particularly in young children and infants. Blood pressure measurement in children requires consideration of the child’s age, sex and height. Inaccurate cuff size and positioning will result in inaccurate measurements, and blood pressure should be taken in a relaxed environment and in all four limbs. Causes of renovascular hypertension in children include fibromuscular dysplasia, vasculitis (Takayasu disease, Kawasaki disease, polyarteritis nodosa), syndromes such as neurofibromatosis, and extrinsic compression from tumors such as Wilms tumor or neuroblastoma. As in adults, intra-arterial digital subtraction angiography remains the definitive diagnostic procedure in children. In smaller children, general anesthesia will be required, and small-caliber coronary angiography catheters can be used. In general, life-long medical treatment is avoided in children with renovascular hypertension, and most children will be referred for surgical or endovascular revascularization. Medical management is useful in emergency control of blood pressure, optimization of blood pressure control in preparation for revascularization, and rare cases of widespread distal arterial disease not amenable to surgical or endovascular intervention.
Management of Patients with Renal Artery Disease
Management options for patients with renal artery disease resulting in renovascular hypertension or ischemic nephropathy includes medical management, surgical revascularization (or nephrectomy), and endovascular treatment.
Medical Management
Medical treatment of renovascular hypertension is often the preferred treatment for patients with well-controlled mild hypertension in the absence of a threat to kidney function (from ischemic nephropathy or progressive arterial disease). Medical treatment is also reasonable in patients with advanced age and multiple comorbidities that place them at high risk for surgical or endovascular intervention. Some patients with ischemic nephropathy and hypertension who have poor potential for recovery of renal function (see later), are also managed medically. New classes of antihypertensive medications, including newer ACE inhibitors and angiotensin receptor antagonists (and potentially renin inhibitors), provide specific blockade of RAAS overactivity and adequate control of blood pressure.
Optimal medical therapy aims to reduce overall risk of cardiovascular disease in addition to lowering blood pressure, particularly in patients with atherosclerotic renal artery disease. This approach is important because patients with renal artery ASO usually have a large burden of systemic arterial disease and multiple risk factors for cardiovascular mortality. Strict blood pressure control is essential to a target blood pressure below 140/90, or below 130/80 in diabetics and patients with renal disease and proteinuria (Chobanian et al, 2003). Use of angiotensin receptor antagonists or ACE inhibitors as first-line therapy is attractive and usually effective in controlling blood pressure, although renal function has to be carefully monitored during use. Combination treatment with diuretics, calcium channel blockers, β or α blockers, and vasodilators is based on clinical results and patient comorbidities. In addition to blood pressure control, a complete treatment regimen includes treatment of dyslipidemia (to low-density lipoprotein cholesterol <100), strict management of diabetes (tight glucose control, medical nutrition, foot and eye care, physical activity), antiplatelet agents (aspirin, clopidogrel, or ticlopidine), smoking cessation, and management of complications of renal insufficiency (anemia, hyperparathyroidism, etc.), which are frequently present in patients with renal impairment.
Selection of Patients for Surgical or Endovascular Therapy
Renovascular Hypertension
In patients with RVH secondary to FD, the decision for intervention (surgery or angioplasty) is guided by the specific type of disease as determined by angiographic findings and the associated natural history (Novick et al, 1996). Medical management of hypertension is the preferred initial treatment for patients with medial fibroplasia, because loss of renal function from progressive obstruction is uncommon with this disease. Interventive treatment in the latter category is reserved for patients whose blood pressure is difficult to control with multiple drugs. Conversely, renal artery stenosis secondary to intimal or perimedial fibroplasia generally progresses and often eventuates in ischemic renal atrophy. Furthermore, these lesions tend to occur in younger patients and to cause hypertension that is extremely difficult to control. Early interventional therapy in these patients is therefore indicated both to preserve renal function and to minimize the need for long-term antihypertensive medication.
In selecting patients with FD for surgical renal revascularization, the efficacy of percutaneous transluminal angioplasty (PTA) must also be considered. The results of angioplasty for FD of the main renal artery have been excellent and equal to those obtained with surgical revascularization; therefore, angioplasty is the initial treatment of choice in such cases. However, as many as 30% of patients with FD have branch renal arterial involvement, which increases the technical difficulty of, and often precludes, angioplasty. Therefore surgical renal revascularization is the primary interventional treatment in this category.
In patients with atherosclerotic RVH, more vigorous attempts at medical management are warranted, because these patients are older and often have extrarenal vascular disease. Therefore multiple-drug regimens that control the blood pressure are often the preferred approach. Indeed, the advent of new β-blocking agents and converting enzyme inhibitors has enhanced the efficacy of medical antihypertensive therapy. Intervention with surgery or endovascular therapy is best reserved for patients whose hypertension cannot be adequately controlled or when renal function is threatened by advanced vascular disease (Novick et al, 1996).
Ischemic Nephropathy
Anatomic Severity and Extent of Renal Artery Disease
After angiographic diagnosis of ARAS, and with knowledge of the natural history of this disease, one can identify those patients in whom such disease poses a significant threat to overall renal function. This designation applies to patients with high-grade (>75%) arterial stenosis affecting their entire renal mass, namely, where such stenosis is present bilaterally or involves a solitary kidney (Fig. 39–8). In these patients, the risk of complete renal arterial occlusion is significant, and, if this occurs, the clinical outcome is a critical decrease in functioning renal mass with resulting renal failure. Intervention to restore normal renal arterial blood flow is indicated in such patients for the purpose of preserving renal function. In a study from the Cleveland Clinic (Novick et al, 1987), surgical revascularization for preservation of renal function was performed in 161 patients with ARAS bilaterally or in a solitary kidney. Postoperatively, renal function was improved in 93 patients (58%), stable in 50 patients (31%), and deteriorated in only 18 patients (11%).
Figure 39–8 Atherosclerotic renal artery disease poses the greatest threat to overall renal function in patients with high-grade arterial stenosis to a solitary (right) kidney (A), and bilateral high-grade renal artery stenosis (B), as demonstrated on these abdominal aortograms.
The benefit of undertaking revascularization for the preservation of renal function in patients with unilateral ARAS and an unobstructed contralateral renal artery is not established. If the contralateral kidney is anatomically and functionally normal, revascularization for this purpose is clearly not warranted. If the opposite kidney is functioning but involved with some type of parenchymal disorder, revascularization of the ischemic kidney may benefit some patients, but specific indications for this approach are not well defined. Dean and associates (1991) reviewed the renal functional outcome after surgical revascularization in 53 patients with IN. The postoperative estimated GFR was improved significantly in 41 patients treated for bilateral ARAS but was unaltered in 12 patients with unilateral ARAS.
Complete occlusion of the renal artery most often eventuates in irreversible ischemic damage of the involved kidney. However, in some patients with gradual arterial occlusion, the viability of the kidney can be maintained through the development of collateral arterial supply (Morris, 1956; Zinman and Libertino, 1977; Schefft et al, 1980). Helpful clinical clues suggesting renal salvability in such cases include (1) angiographic demonstration of retrograde filling of the distal renal arterial tree by collateral vessels on the side of total arterial occlusion (Fig. 39–9), (2) a renal biopsy showing well-preserved glomeruli, (3) kidney size greater than 9 cm, and (4) function of the involved kidney on isotope renography or intravenous pyelography. When such criteria are present, restoration of normal renal arterial flow can lead to recovery of renal function.
Level of Renal Function
In general, revascularization to preserve renal function in patients with ARAS is most likely to be beneficial in those who have not yet sustained severe, permanent impairment of overall renal function. In a study from the Cleveland Clinic (Bedoya et al, 1989), we evaluated the effect of baseline renal function on the outcome of surgical revascularization in elderly patients with ARAS. The majority of these patients had preoperative serum creatinine levels of less than 3 mg/dL; postoperative renal function was stable or improved in 89% of patients in this category.
Revascularization to preserve renal function is generally not worthwhile in patients with severe azotemia (serum creatinine level >4 mg/dL), because advanced underlying renal parenchymal disease is inevitably present and obviates improvement in renal function with restored perfusion. This observation has been recorded by several groups including our own (Mercier et al, 1990; Chaikof et al, 1994). Severe nephrosclerosis is the most common form of renal parenchymal disease in such patients; however, renal cholesterol embolization may be an additional complicating feature (Fig. 39–10) (Vidt et al, 1989). In patients with atherosclerotic ischemic renal disease and a serum creatinine level greater than 4 mg/dL, our policy is to perform a renal biopsy to evaluate the severity of renal parenchymal involvement from one or both of these disorders.
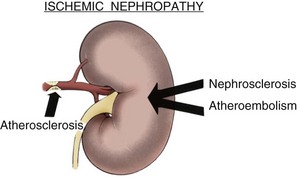
Figure 39–10 Patients with atherosclerotic renal artery disease often have renal parenchymal involvement with varied degrees of nephrosclerosis or atheroembolic disease.
Some patients present with severe impairment of overall renal function that has developed acutely after the initiation of medical antihypertensive therapy (Hricik et al, 1983; Textor and Novick, 1983). Such rapidly progressive renal insufficiency may be a manifestation of perfusion-dependent renal function due to underlying ARAS. If this problem is detected promptly, renal function often improves after discontinuation of the offending antihypertensive medications. Intervention to relieve renal arterial obstruction can prevent permanent renal damage in patients with this type of severe acute renal insufficiency.
In addition to the absolute level of renal function, the rate of decline in overall renal function is an important determinant of the outcome after intervention in atherosclerotic ischemic renal disease. In a study by Dean and associates (1991), patients with a rapid rate of deterioration in estimated GFR during the 6 months preceding surgical revascularization achieved the greatest benefit in terms of postoperative improvement of estimated GFR. Unfortunately, their data precluded definition of a critical rate of decline in estimated GFR that would predict retrieval of renal function by revascularization. Nevertheless, rapid deterioration of overall renal function in association with ARAS suggests a strong possibility of retrieval of function by intervention to restore normal renal arterial flow.
Occasional patients with ESRD from IN have had salvable renal function on revascularization (Wasser et al, 1981; Kaylor et al, 1989). The basis for this has been the presence of chronic bilateral total renal arterial occlusion in which, fortuitously, the viability of one or both kidneys has been maintained through collateral vascular supply. In such cases, revascularization can yield dramatic recovery of renal function (Fig. 39–11). Unfortunately, this clinical presentation is rare, and a less favorable outcome of bilateral arterial occlusion on renal viability is far more common.
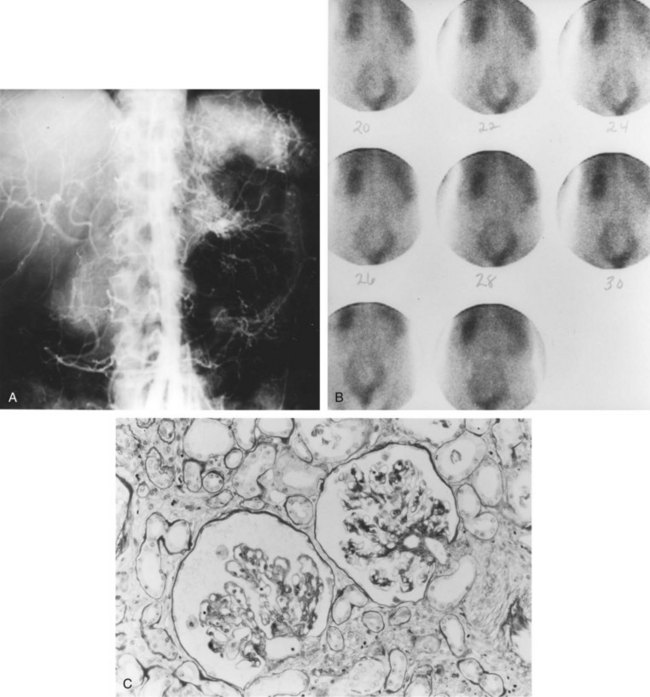
Figure 39–11 A, This abdominal aortogram was obtained in a 60-year-old woman who had been undergoing chronic dialysis for 9 months with no urine output. The aortogram shows complete bilateral renal artery occlusion with evidence of collateral vascular supply to the left kidney. B, Technetium renal scan confirms blood flow to the left kidney from collateral supply. C, Left renal biopsy shows hypoperfused but viable glomerular tissue, indicating a salvageable kidney. Left renal revascularization led to recovery of renal function and discontinuation of dialysis in this patient.
We reported nine patients with ESRD, caused by atherosclerotic renal artery occlusion, who underwent surgical revascularization with subsequent recovery of renal function (Kaylor et al, 1989). The duration of dialysis preoperatively ranged from 1 to 13 months. In all cases, renal viability was being maintained by collateral vascular supply. Postoperatively, renal function improved immediately, and no patient required subsequent dialysis. Excellent overall rehabilitation was achieved in all patients. Six patients were alive at a mean interval of 4 years postoperatively and with satisfactory renal function (mean serum creatinine level of 2.7 mg/dL). Three patients died at a mean interval of 6.8 years postoperatively, and they all maintained satisfactory renal function (mean serum creatinine level of 3 mg/dL) until death. Although some patients with ESRD caused by atherosclerotic arterial occlusion can be salvaged in this manner, in most such cases irreversible loss of renal function has occurred. Therefore the most effective approach is to relieve arterial obstruction before ESRD develops.
Finally, it is important to emphasize that patients with ESRD and ARAS without complete occlusion are not appropriate candidates for revascularization to restore function. In such cases, main renal arterial blood flow is preserved, albeit at a reduced level, yet the supplied renal parenchyma is without function. The basis for this is the presence of severe unalterable parenchymal damage that precludes recovery of renal function with improved perfusion.
Renal Histopathology
Evaluation of renal histopathology can help to determine the presence of salvable renal function in selected patients with atherosclerotic renal ischemic disease. In patients with ARAS and severe existing impairment of overall renal function (serum creatinine level >4 mg/dL), renal biopsy findings can help to predict whether revascularization is likely to forestall further progressive renal insufficiency. In patients with atherosclerotic renal artery occlusion, renal biopsy findings can indicate whether the involved kidney is viable and functionally salvable on the basis of collateral vascular supply. The two predominant morphologic lesions in such patients are arteriolar nephrosclerosis and atheroembolic renal disease, with the former more often encountered. Both these diagnoses can be established on a frozen-section histopathologic examination, and, therefore the renal biopsy can be done at the same time as surgical revascularization, if the biopsy findings are favorable. In patients with arteriolar nephrosclerosis, the most important favorable criterion is histologic evidence that a majority of examined glomeruli are intact and viable (Zinman et al, 1977; Schefft et al, 1980). The presence of tubular atrophy, interstitial fibrosis, and arteriolar sclerosis are of lesser importance and do not necessarily preclude recovery of renal function; these findings may merely reflect the histologic changes of chronic reversible renal ischemia. However, the finding of widespread glomerular hyalinization indicates irreversible ischemic renal injury that obviates any benefit from relief of renal arterial obstruction (Fig. 39–12A). The finding of extensive atheroembolic disease would also preclude renal revascularization (Fig. 39–12B). Regarding the technique for renal biopsy in such patients, a more representative specimen of renal tissue is obtained with an open surgical (or laparoscopic) wedge biopsy than with a closed needle biopsy.
Surgical Revascularization
Although repair of renal artery disease by revascularization is usually the main goal of surgical intervention, ablative surgery (nephrectomy) is occasionally more appropriate. The original surgery for renovascular hypertension, nephrectomy is a suitable alternative in hypertensive patients with a unilateral small kidney and markedly decreased ipsilateral function. Renal vein renin evaluation can confirm the kidney as the cause of hypertension, and laparoscopic nephrectomy can provide an attractive minimally invasive alternative to open nephrectomy in such cases (Fig. 39–13).
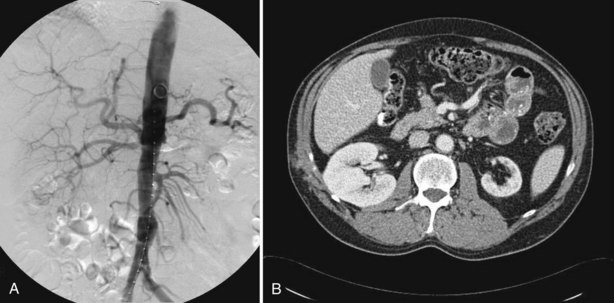
Figure 39–13 A 47-year-old male with poorly controlled hypertension (215/115) on four medications. CT scan shows a small left kidney (A), and angiography confirms an occluded left renal artery with minimal right renal artery stenosis (B). Renin evaluations from the infrarenal inferior vena cava, right and left renal veins were 15.4, 14.9, and 91.5 µg/L/hr, respectively. After laparoscopic left nephrectomy, blood pressure improved to 145/85 on one medication.
Preoperative Preparation
When surgical revascularization is indicated for renal artery disease, it is important to accurately define the general medical condition of the patient, because this will determine the risk of undertaking a major vascular operation. Most patients with renal arterial FD are young and otherwise healthy, and the operative risk is minimal in this group (Novick, 1994a). In patients with atherosclerotic renovascular disease, the preoperative evaluation should include a thorough search for coronary artery disease, because this has been the leading cause of operative death after surgical revascularization. In our program, in addition to a careful history, a physical examination, and an ECG, a thallium cardiac stress test is done on all operative candidates in this category. If any of these assessments suggest the presence of coronary artery disease, our policy is to perform coronary cineangiography and left-sided ventriculography. Myocardial revascularization is recommended for patients with significant correctable coronary artery disease before renal revascularization (Novick et al, 1981).
Cerebrovascular accident has also been a significant cause of death after renal revascularization in patients with atherosclerosis, albeit a less common complication than myocardial infarction. The approach to patients whose history or examination suggests extracranial cerebrovascular disease is analogous to that employed for patients with suspected coronary artery disease. In such patients, carotid arteriography is obtained preoperatively, and if significant occlusive disease is found, endarterectomy is recommended before renal revascularization (Novick et al, 1981).
In patients with generalized atherosclerosis and renal artery disease, cardiac function is often compromised to various degrees. In these patients, hypertension increases the workload on the left ventricle, which decreases cardiac reserve and renders the heart less efficient. In addition to having an impaired myocardium, these patients also often have a decreased intravascular volume because of prior treatment with diuretic agents. These patients can benefit from a careful hemodynamic assessment in an intensive care unit for 12 to 24 hours before surgical revascularization. In the intensive care unit, Swan-Ganz arterial and urethral catheters are placed for measurement of blood pressure, pulmonary capillary wedge pressure, pulmonary artery pressure, cardiac output, total peripheral resistance, and urinary output. While these parameters are being monitored, intravenous vasodilators can be administered to control the blood pressure and decrease the cardiac afterload, while the intravascular space is carefully expanded with isotonic fluid. Afterload reduction and fluid repletion, in this manner, optimize perioperative cardiac function by increasing cardiac output and decreasing cardiac work. This approach can enhance the safety of surgical renal revascularization in patients with generalized atherosclerosis.
Operative Techniques
Advances in both surgical renovascular reconstruction and medical antihypertensive therapy have limited the role of total or partial nephrectomy in the management of patients with renal artery disease. These operations are only occasionally indicated in patients with severe arteriolar nephrosclerosis, severe renal atrophy, uncorrectable renovascular lesions, and renal infarction.
A variety of surgical revascularization techniques are available for treating patients with significant renal artery disease. Aortorenal bypass with a free graft of autogenous hypogastric artery or saphenous vein remains a popular method in patients with a healthy abdominal aorta. Polytetrafluoroethylene aortorenal bypass grafts have been successfully employed by some authors, usually when an autogenous graft is not available. Renal endarterectomy also continues to be used occasionally to treat atherosclerotic renal artery disease. Patients with complex branch renal artery lesions are managed with extracorporeal microvascular reconstruction and autotransplantation.
In older patients, severe atherosclerosis of the abdominal aorta may render an aortorenal bypass or endarterectomy technically difficult and potentially hazardous to perform. In such cases, several authors prefer alternative surgical approaches that allow renal revascularization to be safely and effectively accomplished while avoiding operation on a badly diseased aorta. The most effective alternate bypass techniques have been a splenorenal bypass for left renal revascularization and a hepatorenal bypass for right renal revascularization. The absence of occlusive disease involving the origin of the celiac artery is an important prerequisite for these operations. A 1995 study indicated the presence of significant celiac artery stenosis in 50% or more of patients with ARAS (Fergany et al, 1995). This information underscores the importance of obtaining preoperative lateral aortography to evaluate the celiac artery origin in patients who are being considered for hepatorenal or splenorenal bypass.
Use of the supraceliac or lower thoracic aorta for renal revascularization is a more recent surgical alternative in patients with significant atherosclerosis of the abdominal aorta and its major visceral branches. The supraceliac aorta is often relatively disease-free in such patients and can be used to achieve renovascular reconstruction with an interposition saphenous vein graft. Simultaneous aortic replacement and renal revascularization have been associated with an increased risk of operative mortality, and this approach is best reserved for patients with a fixed indication for aortic replacement, such as a significant aortic aneurysm or symptomatic aortoiliac occlusive disease.
Results
Reports from several centers indicate that the techniques described previously for surgical renovascular reconstruction can be safely performed with a high technical success rate. Patients with FD are usually otherwise healthy, and operative morbidity and mortality after revascularization in this group have been minimal (Novick et al, 1987; Hansen et al, 1992). Operative mortality rates of 2.1% (Novick et al, 1987), 3.1% (Hansen et al, 1992), 3.4% (Bredenberg et al, 1992), and 6.1% (Libertino et al, 1992) have been reported after surgical revascularization in patients with atherosclerotic renal artery disease. An increased risk of operative mortality has been observed with bilateral simultaneous renal revascularization (Hallett et al, 1987), or when renal revascularization is performed in conjunction with another major vascular operation such as aortic replacement (Lawrie et al, 1989). Most studies have indicated a high technical success rate for surgical vascular reconstruction with postoperative thrombosis or stenosis rates of less than 10% (Novick et al, 1987; Van Bockel et al, 1987; Libertino et al, 1992).
In a retrospective study from the Cleveland Clinic, Tsoukas and colleagues (2001), evaluated the results of simultaneous aortic and renal revascularization and reported a 13% postoperative mortality rate for bilateral renal revascularization versus 7% for patients with unilateral renal revascularization, when both were performed simultaneously with aortic replacement. The 5-year survival rate for patients with preoperative serum creatinine more than 2 mg/dL was 53% compared with 85% for patients with lower creatinine. The authors recommend adjunctive use of endovascular techniques to decrease the magnitude of the surgical procedure for patients with concomitant aortic and bilateral renal artery atherosclerotic disease.
In evaluating the results of surgical revascularization for RVH, most studies have considered patients to be cured if the blood pressure is 140/90 mm Hg, or less, postoperatively. Patients have been considered to be improved if they have either shown a reduction in diastolic pressure of 10 to 15 mm Hg, or more, or become normotensive on medication. Failures have been those who have not qualified for either of the aforementioned categories. The results of surgical treatment for RVH vary according to the underlying pathologic diagnosis. In patients with FD, 50% to 60% are cured, 30% to 40% are improved, and the failure rate is less than 10% (Novick et al, 1987; Hansen et al, 1992). In patients undergoing revascularization for atherosclerotic RVH (Table 39–5), the failure rate is approximately the same; however, fewer patients are cured and more patients are improved postoperatively. The explanation for this is that RVH is often superimposed on existing essential hypertension in older patients. A study by Van Bockel and colleagues (1987) highlighted the excellent long-term results after reconstructive surgery for atherosclerotic RVH, with a mean follow-up of 8.9 years; postoperative hypertension was cured or improved in 83 of 105 patients (79%).
During the past 15 years, more centers have been performing surgical revascularization to preserve renal function in patients with high-grade atherosclerotic arterial occlusive disease affecting both kidneys or a solitary kidney. These are generally older patients with diffuse atherosclerosis, ostial renal artery lesions, and varied degrees of renal functional impairment. Studies from several centers (Table 39–6) have indicated improvement or stabilization of renal function postoperatively in 75% to 89% of patients. Considering the significant risks of progressive occlusive disease and renal failure that have been associated with medical management of such patients, these results demonstrate a favorable influence of revascularization on the natural history of untreated atherosclerotic renal artery disease.
We have reviewed the contemporary experience at the Cleveland Clinic, between 1980 and 1992, with surgical renal revascularization employing an extra-anatomic bypass operation (Fergany et al, 1995). A total of 175 operations were done, including hepatorenal bypass (n = 59), splenorenal bypass (n = 54), iliorenal bypass (n = 37), thoracic aortorenal bypass (n = 23), renal autotransplantation (n = 1), and superior mesenterorenal bypass (n = 1). There were five operative deaths (2.9%) and seven cases of postoperative graft thrombosis (4%). All patients with poorly controlled hypertension were cured or improved postoperatively. Among patients with IN, postoperative renal function improved in 35%, remained stable in 47%, and deteriorated in 18%. These extra-anatomic techniques have become an important component of the surgical armamentarium for ARAS.
In another study, we analyzed long-term clinical outcomes and survival after surgical revascularization for ARAS in 222 patients treated from 1974 to 1987 at the Cleveland Clinic (Steinbach et al, 1997). The indications for surgical revascularization were treatment of hypertension in 60 patients, preservation of renal function in 12 patients, and both control of hypertension and preservation of renal function in 148 patients. The mean postoperative follow-up interval was 7.4 years.
In this study, there were five operative deaths (2.2%), and postoperative thrombosis or stenosis of the repaired renal artery occurred in 16 patients (7.3%). Long-term improvement or stabilization of renal function was achieved in 71.3% of patients. Actuarial 5- and 10-year survival for the entire series from the time of revascularization was 81% and 53%, respectively. The expected 5- and 10-year survival for a comparable healthy population is 89% and 77%, respectively. Using a multivariate analysis, factors correlating with diminished long-term survival were age greater than 60 years (P = .002), coronary artery disease (P = .031), and previous vascular operations (P = .001). These data support the long-term therapeutic efficacy of surgical revascularization in patients with ARAS. The merits of newer forms of therapy, such as PTA and endovascular stenting, must ultimately be weighed against these results.
Cherr and colleagues (2002), reported on the results of surgical revascularization in 500 patients with atherosclerotic renal artery disease from 87 to 99. Unilateral surgery was performed in 40%, bilateral in 60%, and combined aortic and renal surgery in 41%. Mortality rate was 4.6% overall. Hypertension was cured, improved and unchanged in 12%, 73%, and 15%, respectively. Renal function was improved in 43%, unchanged in 47% and deteriorated in 10%.
The results of renal autotransplantation for renal artery stenosis due to various pathologies (fibromuscular disease [FMD], ASO, and Takayasu arteritis) were reported by Chiche and colleagues (2003). Results of surgery were better in the FMD patients with early or delayed nephrectomy performed in five of eight patients with ASO. Hypertension was cured or improved in 96% of patients with FMD, and 89% of patients with Takayasu disease.
Secondary Renal Revascularization
Relatively few reports have addressed the management of patients with recurrent renal artery stenosis after failed surgical revascularization or PTA. For patients in the latter category, repeat PTA may be attempted; however, ultimately, many of these patients will require surgical revascularization. There has been controversy concerning whether the prior performance of PTA increases the technical difficulty or compromises the outcome of surgical renal revascularization. A report by Dean and associates (1987) indicated that the task of arterial reconstruction may be rendered more complicated by a perivascular inflammatory response induced by PTA. Reports from McCann and colleagues (1988) and Martinez and coworkers (1990) found that surgical revascularization is not more technically difficult after PTA than when done primarily, and that the same satisfactory results can be achieved. The latter study comprised 53 patients treated surgically after failed PTA for renal artery stenosis. Three patients underwent nephrectomy because a nonviable kidney was found at operation. Successful surgical revascularization was achieved in all the remaining 50 patients. The authors noted no significant fibrosis or inflammation around the previously dilated renal artery. PTA necessitated performing a more complicated revascularization operation in only 1 patient. These data indicate that if the kidney is viable at operation, prior performance of PTA does not increase the technical difficulty of surgical renovascular reconstruction.
Recurrent renal artery stenosis after surgical revascularization is typically a late complication occurring weeks, months, or even years postoperatively. If the involved kidney is functionally salvable, then another attempt to restore normal renal arterial flow is indicated. There has been scant experience with PTA or stenting in this setting; however, this is an appropriate initial approach for focal stenotic lesions. Secondary surgical revascularization has constituted the predominant reported approach, and this may be technically complicated (Stanley et al, 1985; Erturk et al, 1989). Reoperation often entails dissection in a surgical field obliterated by fibrous scar tissue, and it is most efficacious to employ a secondary reconstructive technique that avoids the site of previous surgery. In patients with recurrent renal artery stenosis after an abdominal aortorenal bypass, alternate approaches that may be used for secondary revascularization include hepatorenal bypass, splenorenal bypass, thoracic aortorenal bypass, iliorenal bypass, and renal autotransplantation.
Percutaneous Transluminal Angioplasty
Percutaneous dilatation of arterial stenoses (angioplasty) was originally introduced by Dotter and Judkins in 1964; however, their technique of using coaxial catheters of increasing diameter was limited to the femoral and popliteal arteries and did not gain wide acceptance. It was the development of the balloon catheter by Gruntzig and colleagues in 1978 that permitted the widespread use of angioplasty for dilatation of the renal, coronary, and almost all other visceral arteries. Numerous modifications have been made to the original technique since its introduction, and there are currently several approaches to performing renal PTA.
Technique
All angioplasty techniques require a high-quality angiogram before dilatation to accurately delineate the lesion and allow assessment of proper equipment needs and approach. A proper-sized balloon should be selected to correspond to the original diameter of the renal artery as measured on the angiogram. Because there is 15% to 20% magnification on the angiogram, the result is the intentional overdilatation of the renal artery by 1 mm. Progress of angioplasty is monitored as the balloon inflates, and a postdilatation angiogram is obtained to assess the results and diagnose complications. PTA should be performed only when a skilled vascular surgeon is immediately available in case inadvertent occlusion or disruption of the renal artery creates a surgical emergency.
The original Gruntzig coaxial technique uses an 8- or 9-Fr renal guiding catheter through which a 4.3- or 4.5-Fr balloon catheter is passed over a guidewire traversing the stenotic segment through a femoral arterial puncture. In selected circumstances, an axillary approach may be used. Modification of the original technique and balloon catheters have allowed the use of a 5-Fr femoral artery puncture through which a 5-Fr diagnostic catheter is passed to the renal artery using the Seldinger technique. After the lesion is negotiated with an appropriate guidewire, the diagnostic catheter is exchanged with a 5-Fr balloon catheter and the angioplasty is performed (Tegtmeyer et al, 1980).
Mechanism
The principal mechanism through which the increase in arterial diameter after PTA occurs in cases of ASO is fracture of the atherosclerotic plaque. Stretching of the arterial wall with tearing of the media and adventitia also occurs but contributes to a smaller degree in cases of ASO than in FD. This stretching effect probably occurs after the atherosclerotic plaque is fractured and becomes more pronounced with increasing plaque circumference and a decrease in the uninvolved area of the vessel wall.
Complications
Renal angioplasty is a complex and technically demanding procedure that should be performed only by skilled interventional radiologists to prevent the occurrence of potentially serious complications. The complications of PTA include those of standard angiography (complications related to arterial puncture and to the use of iodinated contrast material), as well as specific complications related to manipulation of the renal arteries. Transient deterioration of renal function is the most frequently occurring complication and is related to the contrast load delivered during the procedure. Adequate hydration, minimizing the volume of contrast material, separating the diagnostic procedure from the PTA (by several days), and possibly the use of carbon dioxide or non-nephrotoxic contrast agents may decrease the incidence of this complication.
Technical mishaps during PTA may lead to an intimal dissection or even thrombosis of the renal artery. A small intimal flap is usually inconsequential, healing without sequelae. A larger flap compromising blood flow is usually managed by placing an arterial stent across the dissection. Thrombosis of the renal artery can be managed by injecting a thrombolytic agent through the renal artery or by emergency surgery. Rupture of the renal artery, a rare complication, is also managed by emergency surgery after reinflation of the balloon catheter to control retroperitoneal hemorrhage. Overall, the complication rate can be expected to be 5% to 10%.
Fibrous Dysplasia
Several studies have reported the results of PTA on blood pressure in cases of FD. Cure of hypertension is usually defined as a diastolic blood pressure of 90 mm Hg, or less, with no antihypertensive medication. Improvement of hypertension is usually defined as either a decrease in diastolic pressure of 15 mm Hg or a diastolic pressure of 90, or less, with a decrease in the antihypertensive medication needed to maintain normal blood pressure. Standardization of the results of these studies is difficult for several reasons. Several definitions are not uniform, including significant stenosis (varying from >50% to >80% of the original lumen) and technical success (defined as no stenosis or insignificant residual stenosis). Measures of success are also variable, with most studies relying on clinical parameters (blood pressure response) as the measure of success. Follow-up angiographic studies are rarely available. In addition, most studies combine ASO and FD patients, and sometimes results or complication rates are not referred specifically to one of the disease groups. Further complicating the interpretation of results is the reporting of results per patient in some studies and per lesion in others. The results of these studies are summarized in Table 39–7. Data per patient are used, and represent primary procedures only.
Overall, PTA is usually performed in cases of FD without stent placement and has become the primary modality of treatment for these lesions. With the use of modern equipment and increasing experience with the technique, technical success has been more than 90% in most modern studies. A beneficial blood pressure response, that is, cure of hypertension or improvement in blood pressure control, can be expected in more than 80%, and up to 100% of cases (Fig. 39–14). The incidence of major complications (when reported for FD patients alone) was 6% or less. At short to moderate follow-up intervals, restenosis of the treated artery was reported in up to one third of cases; however, most of these cases have been successfully redilated. Rare reports describe the use of renal artery stents for resistant fibrous lesions (Joffre et al, 1992; Imamura et al, 1998); however, these are usually not needed for this form of RAS.
Atherosclerotic Renal Artery Stenosis
The pattern of arterial disease in cases of ASO is different from that in FD. Renal artery stenosis in cases of ASO is usually bilateral and ostial, or very proximal in the main renal artery. In most ostial cases, this represents encroachment of the atherosclerotic plaque in the abdominal aorta on the origin of the renal artery rather than primary renal artery disease. The patients affected by atherosclerotic RAS are also different from patients with FD of the renal arteries. Patients with ASO are generally older and have a number of comorbid medical conditions, as well as generalized atherosclerosis affecting the coronary or carotid arteries or the peripheral vascular tree. Associated essential hypertension and nephrosclerosis are usually present. All the previously mentioned factors, as well as the propensity for atheroembolism in patients with generalized ASO, make PTA for ASO-RAS less successful and associated with higher morbidity (and some mortality) than in cases of FD. The presence of renal insufficiency or borderline renal function in a significant percentage of patients leads to an increased incidence of contrast nephrotoxicity as well.
The results of PTA on blood pressure in cases of RVH secondary to ASO are summarized in Table 39–8. Because of the previous factors, the cure rate is lower than that for FD, commonly around 15%, and less in cases with bilateral PTA. Failure to improve the blood pressure occurs in 15% to 85% in the reported series. Technical success at the time of angioplasty varies from 57% to 92% (Fig. 39–15), and is lower in cases in which separate results for ostial stenosis are reported (62% to 72%). Restenosis rates of 8% to 22%, and as high as 68% in some earlier series, are reported. Restenosis of ostial lesions is higher, reaching 35%, although more recent studies of ostial angioplasty show improved results (Eldrup-Jorgensen et al, 1995; Hoffman et al, 1998). Major complications requiring surgical intervention are reported in 5% to 24% of cases, and a mortality rate of 1% to 2% is also generally reported. More recent reports tend to show higher rates of technical success and lower complication rates (Martin et al, 1986), reflecting improved equipment and increasing expertise (Table 39–9); however, major complications and mortality rates reflect the fact that PTA is a serious procedure with significant attendant risks in this patient population. Renal artery stents are an adjunct to renal PTA that has been developed to improve the results obtained in ASO patients (see later).
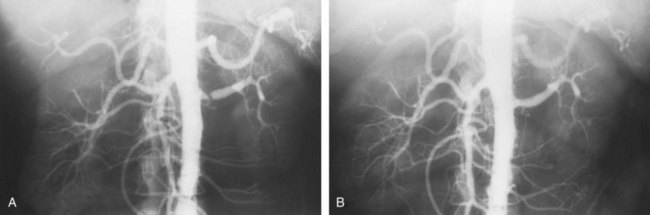
Figure 39–15 A, Aortography shows high-grade left renal artery stenosis from nonostial atherosclerotic plaque. B, After percutaneous transluminal angioplasty, aortography shows relief of left renal artery stenosis.
Table 39–9 Technical Success, Restenosis, and Complications of Angioplasty in Atherosclerosis Patients
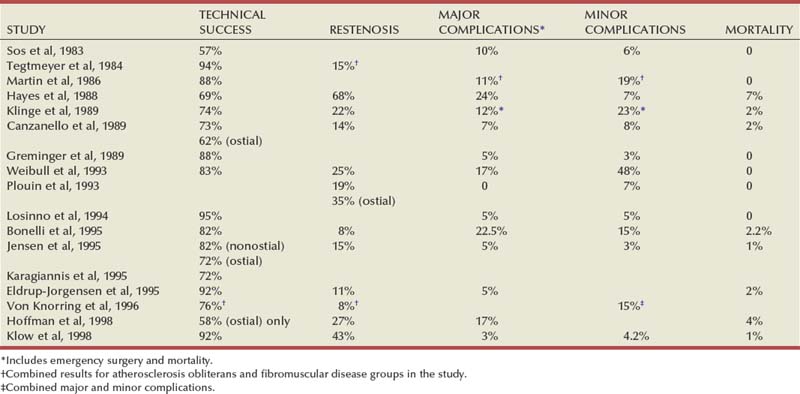
With the advent of newer, more potent antihypertensive medications, as well as the increasing awareness of IN as a cause of renal failure, renal revascularization in patients with ASO is increasingly being performed with the intent of preserving or improving renal function, rather than the intent of curing RVH. Several studies have reported the results of PTA on renal function in patients with ASO-RAS (Table 39–10). Clinical benefit is defined as an improvement in renal function or a stabilization of renal function, in view of the steadily progressive decline in renal function associated with IN. With technically successful angioplasty, improvement can be expected in 15% to 43% of cases, and stabilization of renal function in 26% to 50% of cases. The remaining cases continue to suffer a decline in renal function. Although a mean follow-up of more than 100 months was achieved in one of these studies (Paulsen et al, 1999), follow-up for the rest of the studies is generally short to intermediate, ranging from 1 to 3 years. Percutaneous revascularization has also been reported to be successful in improving blood pressure control and renal function in a diabetic subset of ASO patients (Zuccala et al, 1998).
Inflammatory Renal Artery Stenosis
Takayasu arteritis is one of the causes of RAS in children and accounts for a majority of cases of RAS in South and East Asian countries. In the absence of active disease or inflammation, angioplasty has been performed to relieve RVH as well as to prevent renal loss. In a series of 24 patients aged 5 to 16 years, all with inactive disease, technically successful dilatation was achieved in 92%, with a restenosis rate of 20% after a mean of 33 months of follow-up, and these were successfully redilated. Half the children in this study were cured of hypertension, and the other half improved. No complications were specifically reported in this study (Sharma et al, 1996). In a slightly larger group of 31 children (age range 5 to 14 years) in which less importance was given to the presence of active disease, technical success was possible in 87% of cases, with a restenosis rate of 26% at a mean follow-up of 23 months. Thirty-one percent of the patients were cured, 61% improved, and 8% failed clinically. One acute occlusion of the renal artery was reported; it was successfully redilated (Tyagi et al, 1997).
These excellent results were not duplicated in a smaller Canadian study of six patients in which only one patient obtained clinical benefit and the remaining five patients proceeded to surgical autotransplantation (D’Souza et al, 1998). All these studies were in agreement that long stenoses starting at the ostium of the renal artery were much less likely to have a successful result with PTA.
Management of RAS secondary to Takayasu disease in adults with PTA has also been successful (Dong et al, 1987; Deyu et al, 1998; Sharma et al, 1998). Technical success was obtained in more than 80% of patients in these studies, with overall clinical benefit in the range of 85%.
Children
RAS in children is caused by a variety of arterial diseases, including FD, middle aortic syndrome, and neurofibromatosis. In Asian countries, a significant proportion of RAS in children is also caused by aortoarteritis (Takayasu disease). The main limitation to performing PTA in children was the lack of suitably small-sized equipment. With modern angiographic techniques and digital imaging, PTA has been reported in several studies and is becoming a primary mode of management for children with RVH. The largest series reporting on PTA for treatment of FD in children comprises 16 children, 12 of whom were diagnosed with FD. Three patients required a secondary PTA. Overall, 9 patients were cured and 2 were improved; in 5 patients, the treatment failed, and 4 of these were treated surgically. The PTA failed in all 3 patients with multiple stenoses in this study (none had FD). There were no reports of complications in this study (Courtel et al, 1998).
Reports of smaller studies of PTA in children with FD also demonstrate the technical success and beneficial blood pressure response obtained with this treatment modality (Lund et al, 1984; Millan et al, 1985; Chevalier et al, 1987; Simunic et al, 1990). Angioplasty has been successfully used in very young children, with reports of 18-, 15-, and even 9-month-old children being treated using small coronary angioplasty equipment (Hofbeck et al, 1998; Lee et al, 1999; Liang et al, 1999).
Endovascular Stenting
With increasing experience with PTA, limitations of the technique, especially in regard to atherosclerotic lesions, have become well recognized. These limitations involve mainly ostial lesions that represent engulfment of the renal ostium in aortic plaque. These cases represent a significant percentage of ASO-RAS cases, and elastic recoil of the plaque leads to poor initial results and frequent restenosis. Renal artery stents are an adjunct to PTA that was introduced in an effort to oppose this recoil force and provide better results for angioplasty, especially in ostial lesions. Almost all renal artery stents in the literature have been placed to treat ASO of the renal arteries (estimated at 97%), with many fewer stents placed in cases of FD, transplant renal arteries, and other miscellaneous renal arterial abnormalities (Rees, 1999).
Arterial stents are radiopaque, expandable metallic wire mesh tubes that have been widely used in the peripheral vascular system. Experience in other parts of the vascular as well as the biliary tree has led to the use of stents in the renal artery, although none are specifically approved by the U.S. Food and Drug Administration for use in the renal artery. Different stents are currently available and include the Palmaz, Strecker, Wallstent, and Nitinol stents. Stent expansion either is spontaneous on extrusion from a delivery catheter (self-expandable) or occurs on inflation of a balloon on which the stent is preloaded (balloon expandable).
Technique
Several techniques for stent placement in the renal artery have been developed. It is essential in all cases to obtain a high-quality angiographic study (immediately before stent placement at the same sitting or separately) in order to precisely delineate the lesion and assess balloon and stent length and diameter. The stent used should be long enough to traverse the entire lesion, taking into consideration shortening of the stent with expansion. Excessive length beyond the lesion is undesirable because the presence of the stent may excite an intimal hyperplastic reaction, placing healthy segments of the vessel at risk. This might also obstruct a site suitable for later bypass surgery. In cases of ostial lesions, the stent should be placed to protrude 1 to 2 mm into the aortic lumen to prevent restenosis due to recoil of the aortic plaque.
Indications
Current indications for stent placement are poor immediate results during PTA as well as restenosis after PTA. Stents are also used to treat angioplasty complications (artery dissection and intimal flaps) and thus have markedly reduced the incidence of emergency surgery for these complications. “Primary” stent placement is becoming increasingly popular in cases of RAS in which PTA alone is unlikely to be successful (ostial lesions).
Results
The results of several studies reporting on endovascular stent placement in the renal arteries are summarized in Tables 39-11 and 39-12. Most of the published experience in the renal arteries has been described using the Palmaz stent. With the exception of two of the earliest studies using Strecker and Wallstents, the reported technical success for stent placement has consistently been above 95%, and in most studies has been 99% or 100% (Fig. 39–16). With short to moderate follow-up intervals, restenosis of the renal artery after stent placement has ranged from 6% to 38%. A single early study (Wilms et al, 1991) reported a 71% restenosis rate; however, most larger contemporary series average a restenosis rate of 15% to 20%. Restenosis commonly occurs within the stent owing to a hyperplastic intimal reaction. In other cases, an intimal lining about 1 mm thick covers the luminal surface of the stent. Renal arteries that are dilated and stented to a luminal diameter of less than 6 mm are more prone to develop restenosis.
Figure 39–16 A, Aortography shows high-grade left renal artery stenosis from ostial atherosclerotic plaque. B, After endovascular stenting, aortography shows relief of left renal artery stenosis.
The blood pressure response to renal revascularization by PTA and stenting is shown in Table 39–11. The low cure rates (31% at most) reflect the pathophysiologic nature of atherosclerotic renal artery disease in which anatomic stenosis is frequently associated with concomitant essential hypertension and renal parenchymal damage, precluding a simple pathologic explanation for hypertension. Revascularization for preservation of renal function has become the focus of intervention in cases of ASO-RAS. The result of renal artery stent placement (for renal function preservation or improvement) in various studies is shown in Table 39–12. Because of the progressive nature of renal dysfunction due to ASO, a clinical benefit is defined as improvement or stabilization (halt of decline) of renal function after revascularization. Improvement in renal function was achieved in 13% to 60% of cases, and stabilization was achieved in 24% to 75% of cases, with the remaining cases continuing to have progressive dysfunction.
Perhaps the clearest situation of IN is RAS to a solitary kidney, and it is on these patients that the effect of revascularization on renal function can be most clearly identified. In a study of 21 such patients, an improvement in the serum creatinine level was obtained after stent placement in 42% of cases, and stabilization in 29% of cases. Within the group of improved patients were 4 who were relieved of dialysis dependence. These results were associated with a major complication rate of 19% including a single early (and another late) procedure-related mortality (Shannon et al, 1998).
In the only prospective study comparing PTA alone versus PTA with stenting in ostial atherosclerosis, a total of 85 patients were randomized to either treatment group. Technical success was higher in the stented group (88% vs. 57%), and patency at 6 months was 75% for patients in the stent group versus 29% for PTA alone. In patients with successful primary procedures, restenosis occurred in 14% of the patients with stents and in 48% of patients with PTA alone. Stenting for immediate or late failure of PTA was required in 12 (of 42) patients in the PTA group. This study reflects the overall higher success of PTA with stenting in treating ostial ASO when compared with PTA alone, and probably also justifies the increasing trend to perform primary stenting in these cases to avoid exposing patients to a secondary procedure (Van de Ven et al, 1999).
Several studies addressed the factors affecting outcome after stenting and recommended selecting patients for interventional treatment based on their findings; Kennedy and colleagues (2003) and Zeller and colleagues (2003) both report that baseline renal insufficiency correlates with decreased survival and increased adverse events after renal artery stenting. In Zeller’s study, the 5-year survival rate was 30% for patients with creatinine more than 2.5 mg/dL versus 95% in patients with creatinine less than 1.2 mg/dL. Major causes of death were cardiac events, cerebral events, and malignancy.
Perkovic and colleagues (2002) studied several parameters for patients undergoing renal artery stenting and reported baseline renal impairment, older age (>70), and diabetes as predictors of poor outcome. ACE inhibitor therapy after stenting appeared to improve outcome in this series.
Complications
Complications of renal artery stent placement are similar to those of renal PTA, with the added complications of stent delivery. The incidence of access site complications is higher because of the larger arterial puncture needed. The incidence of intimal injury and dissection is lower than that of PTA, because stent placement itself is a treatment for these complications. A higher incidence of contrast nephrotoxicity owing to a larger contrast load is noted, but the increasing use of carbon dioxide as a contrast agent may decrease the incidence of this complication. The literature has rare reports of endovascular infection from renal artery stents, resolving after conservative therapy with IV antibiotics (Gordon et al, 1996), or progressing to form mycotic aneurysm of the aorta and renal artery necessitating major excision and vascular reconstructive surgery (Deitch et al, 1998).
Major complications (including death) are reported in 0% to 20% of cases (Table 39–13). Minor complications are reported in 0% to 40% of cases. Mortality directly related to the procedure is reported in almost all large series in the range of 3%. This attests to the fact that PTA with stent placement is not a benign procedure and is associated with definite risk. In a review of complications of stent placement in 50 patients, a complication rate of 20% was noted, 10% of patients suffered major complications, and a 10% rate of cholesterol embolism was reported (Beek et al, 1997).
In a meta-analysis of renal artery intervention studies earlier than 1998, including more than 1300 patients, initial technical success of stent placement was 98%, with major complications occurring in 11%. The overall cure rate for hypertension was 20%, whereas hypertension was improved in 49%. Renal function improved in 30% and stabilized in 38% of patients. The restenosis rate at follow-up of 6 to 29 months was 17%. Stent placement had a higher technical success rate and a lower restenosis rate than did renal PTA (98% versus 77% and 17% versus 26%, respectively; P < .001). The complication rate was not different between the two treatments (Leertouwer et al, 2000).
Proper patient selection for endovascular intervention in cases of ASO-RAD should be a careful process that takes into account the patient’s chances of a successful outcome. This process should be as rigorus as patient selection for open surgery to minimize morbidity and poor outcomes from endovascular intervention. Recent studies have suggested that renal artery intervention (with or without stenting) may not improve overall patient outcomes compared with optimal medical treatment alone (van Jaarsveld et al, 2000), and a large prospective randomized multicenter study (CORAL [Cardiovascular Outcomes in Renal Atherosclerotic Lesions]) is currently underway attempting to provide informed clinical decisions regarding patient outcomes.
Complications of PTA with stenting were not found to be significantly different from those of PTA alone in a prospective study comparing both procedures. Specifically, bleeding-related complications were 19% in both groups; cholesterol embolism was 10% in both groups; and access site pseudoaneurysm and renal artery injury were slightly higher with stent placement (7% vs. 5%). Transient deterioration of renal function from contrast nephrotoxicity was noted in 24% of patients undergoing PTA and 21% of patients undergoing PTA with stent placement (Van de Ven et al, 1999).
Recent advances in renal artery intervention include the use of distal protection devices (DPD), and the use of drug-eluting stents (DES) to minimize restenosis. Distal protection devices include filters, baskets, nets, and balloons, most primarily intended for use in carotid artery stenting, and are intended to prevent distal embolization during manipulation of the renal artery. Although clinically apparent distal embolization is uncommon, such atheroemboli may be associated with decreased survival (Krishnamurthi et al, 1999). Experience with DPD has been promising but is still preliminary, with studies reporting retrieval of debris in 60% to 100% of cases (Holden et al 2006, Henry et al, 2001). Difficulty in deploying the devices in the distal renal artery due to early bifurcation or tortuosity may be encountered, resulting in successful use of such a device in three of five cases (Hagspiel et al, 2005). Future device innovations tailored to the renal artery and increasing experience with DPD will further clarify their role in renal artery interventions.
Drug-eluting stents have been used increasingly in coronary interventions, with the intention of decreasing in-stent restenosis, particularly of smaller vessels. Drug-eluting stents have been studied prospectively in the renal artery in 105 patients (Zähringer et al, 2007). Although both Sirolimus and Paclitaxel are used for stent coating, this study used Sirolimus-coated stents only. There was no significant difference in results between the bare stent group and the DES group after 2 years of follow-up, with clinical patency at 88.5% for the bare metal stent group and 98% for the DES group. Blood pressure–lowering effects did not differ significantly between the two groups either. Based on the results of this study, it may seem that DES do not offer an additional advantage in renal artery intervention over bare metal stents, but this may be due to the larger size of the renal arteries (about 6 to 7 mm) compared with the coronary arteries (5 mm or less), where restenosis may be more common. In the future, DES may prove useful in smaller renal arteries or branch disease.