chapter 41 Management of Upper Urinary Tract Obstruction
Technologic advances have significantly improved both the diagnostic and therapeutic alternatives available in the contemporary management of upper urinary tract obstruction. The obstructive processes may be intrinsic, extrinsic, congenital, or iatrogenic, and in many cases the cause of obstruction may not be immediately evident. Furthermore, making an accurate diagnosis of obstruction can also be challenging.
The treatments for upper tract obstruction range from ureteral stent placement to complex procedures involving ileal interposition or autotransplantation. Myriad skills are required for total surgical management of upper urinary tract obstruction. Not surprisingly, endourology, laparoscopy, and robotics are more prominent in the surgical management of upper urinary tract obstruction. As a result of the wide array of available treatments, the practicing urologist must have an understanding of the indications and risks of all the options.
This chapter provides a contemporary, state-of-the-art presentation of the major surgical management strategies for patients with upper urinary tract obstruction. The chapter is organized by the anatomic location of obstruction. The etiology, diagnosis, indications for intervention, risks, and therapeutic options (including endoscopic, laparoscopic, robotic, and open approaches) are thoroughly reviewed.
Ureteropelvic Junction Obstruction
The diagnosis of “ureteropelvic junction obstruction” results in a functionally significant impairment of urinary transport from the renal pelvis to the ureter. Although most cases are congenital, the problem may not become clinically apparent until much later in life (Jacobs et al, 1979). Acquired conditions such as stone disease, postoperative or inflammatory stricture, or urothelial neoplasm may also present clinically with symptoms and signs of obstruction at the level of the ureteropelvic junction (UPJ). Similarly, extrinsic obstruction can occur at this level as well. This section focuses primarily on the diagnosis and treatment of “congenital” UPJ obstruction, although these techniques may be applied to the management of certain acquired conditions, in particular urinary stones.
Pathogenesis
Congenital UPJ obstruction typically results from intrinsic disease. A frequently found defect is the presence of an aperistaltic segment of the ureter, perhaps similar to that found in primary obstructive megaureter. In these cases, histopathologic studies reveal that the spiral musculature normally present has been replaced by abnormal longitudinal muscle bundles or fibrous tissue (Allen, 1970; Foote et al, 1970; Hanna et al, 1976; Gosling and Dixon, 1978) (Fig. 41–1). This results in failure to develop a normal peristaltic wave for propagation of urine from the renal pelvis to the ureter. Recognition that this type of segmental defect is often responsible for UPJ obstruction is of utmost importance clinically because such ureters may appear grossly normal at the time of surgery, and, in fact, may often be calibrated to 14 French or greater. Further investigations in the etiology of UPJ obstruction have shown decreased interstitial cells of Cajal at the UPJ in children (Solari et al, 2003). In addition, the cytokine produced in the urothelium has also been proposed to exacerbate UPJ obstruction (Chiou et al, 2005). Other experimental studies have implicated transforming growth factor-β, epidermal growth factor expression, nitric oxide, and neuropeptide Y in UPJ stenosis (Knerr et al, 2001; Yang et al, 2003). A less frequent intrinsic cause of congenital UPJ obstruction is true ureteral stricture. Such congenital ureteral strictures are most frequently found at the UPJ, although they may be located at sites anywhere along the lumbar ureter. Abnormalities of ureteral musculature have been implicated as electron microscopy has demonstrated excessive collagen deposition at the site of the stricture (Hanna et al, 1976).
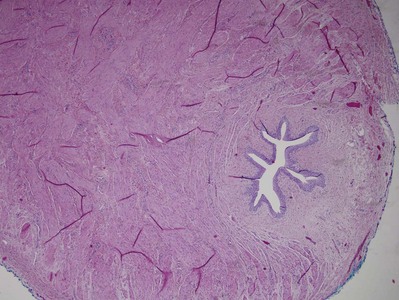
Figure 41–1 The microphotograph is taken through the ureteropelvic junction. There is marked attenuation of smooth muscles and smooth muscle in disarray and hypertrophy surrounding the urothelial lining.
Intrinsic obstruction at the UPJ may also result from kinks or valves produced by infoldings of the ureteral mucosa and musculature (Maizels and Stephens, 1980). In these cases, the obstruction may actually be at the level of the proximal ureter. This phenomenon appears to result from retention or exaggeration of congenital folds normally found in the ureter of developing fetuses. In some of these cases, the defects are bridged by ureteral adventitia. Grossly, this can manifest as external bands or adhesions that appear to be causing the obstruction. In fact, Johnston and colleagues, in 1977, reported that lysis of external adhesions can at times reestablish flow without pyeloplasty. In the majority of cases, however, these bands or adhesions are likely to be a secondary phenomenon associated with intrinsic obstruction, so operative pyeloplasty would generally be most effective. The presence of these kinks, valves, bands, or adhesions may also produce angulation of the ureter at the lower margin of the renal pelvis in such a manner that, as the pelvis dilates anteriorly and inferiorly, the ureteral insertion is carried further proximally. In these cases, the most dependent portion of the pelvis is inadequately drained and the apparent “high insertion” of the ureteral ostium is actually a secondary phenomenon (Kelalis, 1976). In at least some cases, however, the high insertion itself is likely the primary obstructing lesion because this phenomenon is found more frequently in the presence of renal ectopia or fusion anomalies (Zincke et al, 1974; Das and Amar, 1984). As such, a high insertion can have implications in the subsequent surgical management, particularly endourologic approaches.
Controversy persists regarding the potential role of “aberrant” vessels in the etiology of UPJ obstruction. Significant crossing vessels have been noted in up to 63% of cases of UPJ obstruction but as little as 20% of cases of normal kidneys (Quillin et al, 1996; Zeltser et al, 2004; Richstone et al, 2009). Although these lower pole vessels have often been referred to as aberrant, these segmental vessels, which may be branches from the main renal artery or arise directly from the aorta, are usually normal variants (Stephens, 1982). In some patients, these lower pole vessels cross the ureter posteriorly and truly have an aberrant course. Historically, it has been believed that the associated vessel alone does not cause the primary obstruction (Hanna, 1978). In fact, the true etiology is an intrinsic lesion at the UPJ or proximal ureter that causes dilatation and ballooning of the renal pelvis over the polar or aberrant vessel. Recent studies using three-dimensional multidetector row computed tomography (CT) demonstrated that the precise location of crossing vessels did not correspond to the obstructive transition point in patients with UPJ obstruction (Lawler et al, 2005). In contrast, one group found improvement in patients undergoing only ligation of crossing vessels (Keeley, Jr. et al, 1996). Richstone and colleagues reviewed histopathology from 95 patients with UPJ obstruction and found that 43% of 65 patients with a crossing vessel had no intrinsic abnormality (Richstone, 2009). Regardless, the presence of crossing vessels most certainly has a detrimental effect on the success rates of endopyelotomy (Van Cangh et al, 1994; Nakada et al, 1998). UPJ obstruction with concominant anatomic anomalies such as horseshoe kidney and pelvic kidney also present further surgical challenges. Preoperative spiral CT angiographic studies are useful in these patients to identify crossing vessels and to aid in surgical planning (Pozniak et al, 1997). Notably, the current emphasis on laparoscopic and robotic pyeloplasty has quelled the interest in the relevance of preoperative assessment of crossing vessels because this can be addressed at the time of reconstruction.
UPJ obstruction may also result from acquired lesions. In children, vesicoureteral reflux can lead to upper tract dilatation with subsequent elongation, tortuosity, and kinking of the ureter. In some cases, these changes may only mimic the radiographic findings of true UPJ obstruction. However, true UPJ obstruction may definitely coexist with vesicoureteral reflux, although it may be difficult to determine whether the anomalies are merely coincident or whether the upper tract ureteral obstruction has resulted from the reflux (Lebowitz and Johan, 1982). Diuretic renography is first line for differentiating between UPJ obstruction and reflux.
Other acquired causes of obstruction at the UPJ include benign lesions such as fibroepithelial polyps (Berger et al, 1982; Macksood et al, 1985), urothelial malignancy, stone disease, and postinflammatory or postoperative scarring or ischemia. For these acquired diseases, the techniques discussed in this section may be useful adjuncts for management of the obstruction as long as the primary problem is also addressed where appropriate. For instance, fibroepithelial polyps can be managed using retrograde ureteroscopy and holmium laser excision (Lam et al, 2003a).
Patient Presentation and Diagnostic Studies
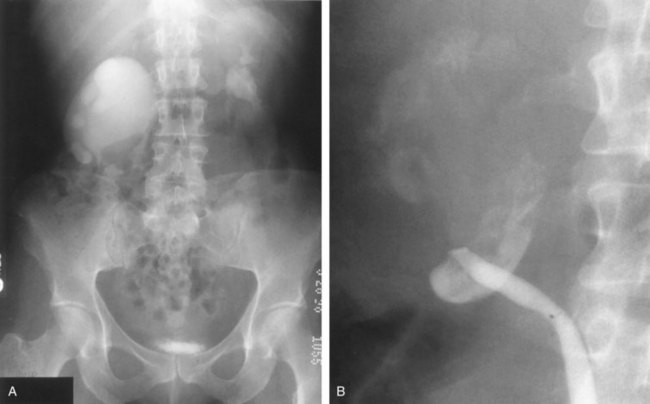
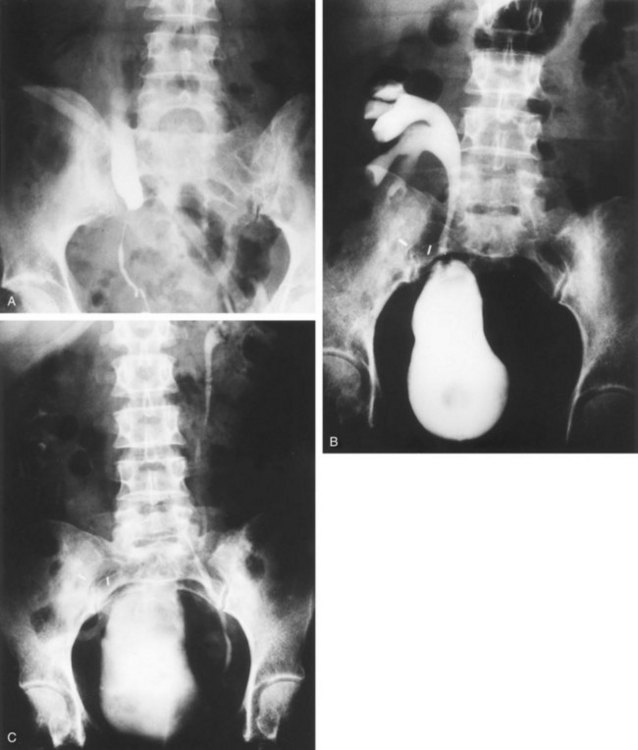
UPJ obstruction, although most often a congenital problem, can present clinically at any time of life. Historically, the most common presentation in neonates and infants was the finding of a palpable flank mass. However, the current widespread use of maternal, prenatal ultrasonography has led to a dramatic increase in the number of asymptomatic newborns being diagnosed with hydronephrosis, many of whom are subsequently found to have UPJ obstruction (Bernstein et al, 1988; Wolpert et al, 1989). A fraction of cases may also be found during evaluation of azotemia, which may result from bilateral obstruction in a functionally or anatomically solitary kidney. UPJ obstruction may also be incidentally found during studies performed to evaluate unrelated anomalies such as congenital heart disease (Roth and Gonzales, 1983). In older children or adults, intermittent abdominal or flank pain, at times associated with nausea or vomiting, is a frequent presenting symptom. Hematuria, either spontaneous or associated with otherwise relatively minor trauma, may also be an initial symptom. Laboratory findings of microhematuria, pyuria, or frank urinary tract infection might also bring an otherwise asymptomatic patient to the urologist. Rarely, hypertension may be a presenting finding (Riehle and Vaughan, 1981). Radiographic studies should be performed with a goal of determining both the anatomic site and the functional significance of an apparent obstruction. Although excretory urography remains a reasonable option for radiographic diagnosis, this study is less commonly used today. Classically, excretory urographic findings include delay in function associated with a dilated pelvicalyceal system. If the ureter is visualized, it should be of normal caliber. In some patients, symptoms may be intermittent and urography between painful episodes may be normal. In such cases the study should be repeated during an acute episode when the patient is symptomatic (Nesbit, 1956). Provocative testing with diuretic urography may allow accurate diagnosis in select cases. The patient should be well hydrated and the study then performed after injecting furosemide, 0.3 to 0.5 mg/kg (Malek, 1983) (Fig. 41–2).
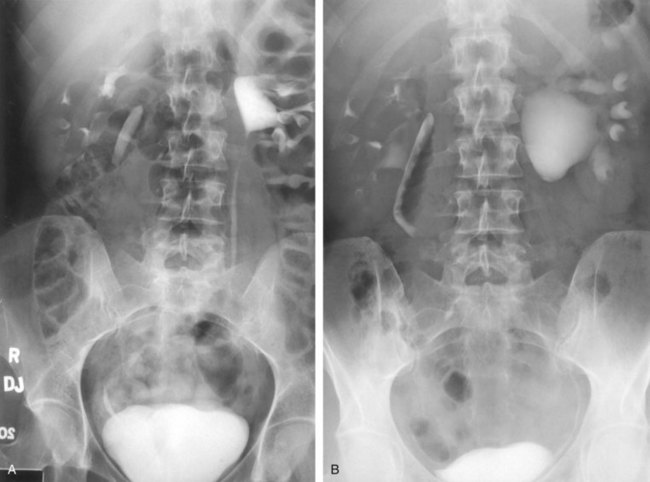
Figure 41–2 A, This patient with intermittent left flank pain underwent intravenous urography. The calyces are sharp bilaterally without evidence of obstruction. However, there is a “box-shaped” pelvis on the left side, which may be associated with intermittent obstruction. B, This intravenous urogram in the same patient was performed along with injection of intravenous furosemide, which brought out the obvious left-sided ureteropelvic junction obstruction. The patient’s symptoms were subsequently relieved with a left pyeloplasty.
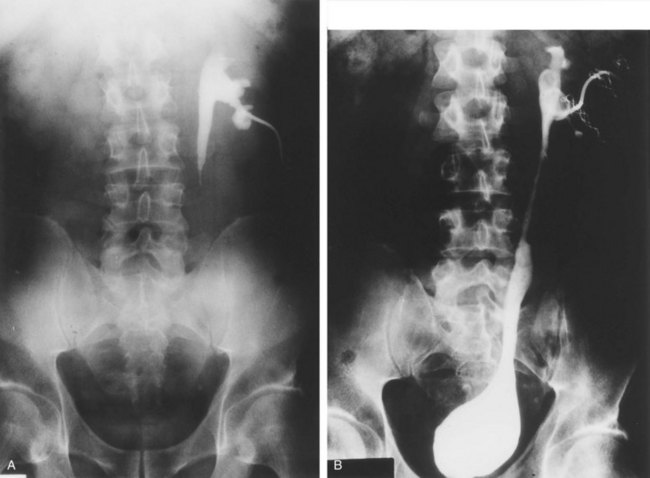
CT scan is frequently obtained for any patient presenting with acute flank pain (Fielding et al, 1997; Dalrymple et al, 1998; Vieweg et al, 1998) (Fig. 41–3). Moreover, CT scans provide detailed anatomic and functional information to aid in diagnosis of UPJ obstruction (Fig. 41–4A-C). Both ultrasonography and CT scanning also have a role in differentiating acquired causes of obstruction such as radiolucent calculi or urothelial tumors. In neonates and infants, the diagnosis of UPJ obstruction has generally been suggested either by routine performance of maternal ultrasonography or by the finding of a flank mass. In either setting, renal ultrasonography is usually the first radiographic study performed. Ideally, ultrasonography should be able to visualize dilatation of the collecting system to help differentiate UPJ obstruction from multicystic kidney and determine the level of obstruction. UPJ obstruction and multicystic kidneys are distinguishable in the majority of cases by ultrasound alone. With UPJ obstruction, the pelvis is visualized as a large, medial sonolucent area surrounded by smaller, rounded sonolucent structures representing dilated calyces. At times, dilated calyces will be seen connecting to the pelvis via dilated infundibula (Fig. 41–5).
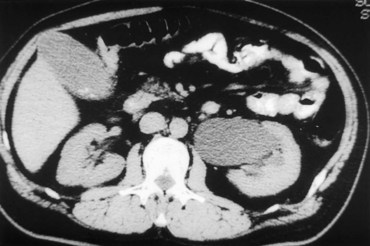
Figure 41–3 Noncontrast CT scan performed as the initial radiographic study in a patient presenting with left flank pain revealed hydronephrosis to the level of the ureteropelvic junction (UPJ). No calculus was visualized, and a presumed diagnosis of UPJ obstruction was considered. This proved correct on subsequent radiographic studies.
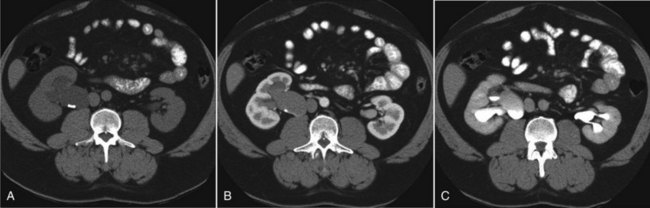
Figure 41–4 A, Contrast-enhanced CT scan identifies a classic ureteropelvic junction (UPJ) appearance in early-phase imaging. B, Early images reveal normal nephrogram and delayed filling of the obstructed, dilated UPJ. C, Delayed images demonstrate holdup of contrast drainage on the right compared with the normal left side.
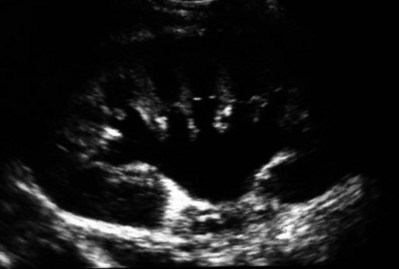
Figure 41–5 Classic ultrasound image of ureteropelvic junction obstruction, with dilated renal pelvis and infundibula and calyces. Note the ureter is not visualized in this image.
Occasionally, a solid-appearing renal cortex can be seen surrounding the sonolucent areas or separating the dilated calyces. In contrast, the cysts of multicystic kidneys are visualized as various-sized sonolucent areas in random distribution. Although the cysts may be connected, this is rarely visualized sonographically. Furthermore, little solid tissue is seen and that which is present has a random distribution among the cysts. Rarely, a large, centrally located cyst may cause confusion in the diagnosis (King et al, 1984a). In this setting, nuclear renography should be performed. Specifically, a technetium Tc99m-diethylenetriamenepentacetic acid (99mTc-DTPA) scan allows differentiation of these two entities. Multicystic kidneys rarely reveal concentration of this isotope. When uptake is seen, the areas of functioning tissue are initially discrete and are usually medial to the bulk of the mass, which itself remains a “cold” area. In contrast, neonatal kidneys with UPJ obstruction generally exhibit good concentration of the isotope. Furthermore, even with severe obstruction in which only a cortical rim remains, uptake of the isotope will be seen peripherally in the cortex, again helping to differentiate this from multicystic kidney (King et al, 1984a).
Diuretic renography is effective in predicting recoverability of function in cases in which intravenous urography has revealed nonvisualization. Diuretic renography allows quantification of the degree of obstruction and can help differentiate the level of obstruction. Today, 99mTc-MAG3 is the preferred isotope because of favorable imaging and dosimetry considerations over 99mTc-DTPA or radioiodinated hippuran (Roarke, 1998). Diuretic renography remains a commonly used study for diagnosing both UPJ and ureteral obstruction because it provides quantitative data regarding differential renal function and obstruction, even in hydronephrotic renal units. Diuretic renography is noninvasive and readily available in most medical centers. Ideally diuretic renography can be used to follow patients for functional loss, most effectively when a standard protocol is used. The diuretic is given 20 minutes into the study to allow time for filling of the collecting system. One study found diuretic renography to be useful in children to rule out concomitant UPJ obstruction with associated high-grade reflux (Stauss et al, 2003). There is evidence that the diuretic renography using MAG-3 is a most accurate study for patients with UPJ obstruction following therapeutic intervention (Niemczyk et al, 1999) (Fig. 41–6).
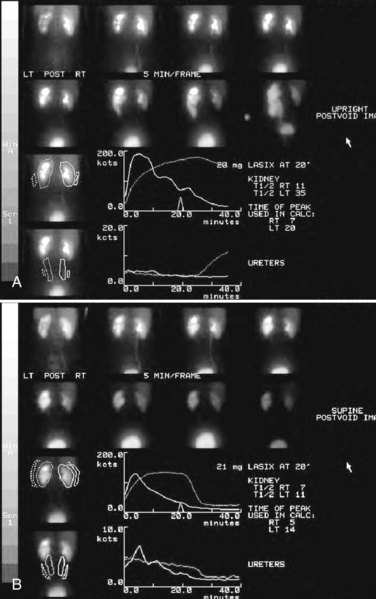
Figure 41–6 A, MAG-3 diuretic renography revealing functional ureteropelvic junction obstruction of the left kidney, with a T1/2 greater than 35 minutes. B, Follow-up MAG-3 diuretic renography reveals normal drainage of the left kidney following successful laparoscopic pyeloplasty.
The diagnosis of UPJ obstruction can generally be made with a high degree of certainty on the basis of the clinical presentation and the results of any one or more of the imaging studies already cited. It is preferable to have a combination of anatomic and functional studies, such as retrograde pyelogram and diuretic renography, in order to best plan therapy. Retrograde pyelography thus retains a role for confirmation of the diagnosis and for demonstration of the exact site and nature of obstruction before repair. In most cases, this study is performed at the time of the planned operative intervention in order to avoid the risk of introducing infection in the face of obstruction. However, retrograde pyelography is indicated emergently whenever the UPJ obstruction requires acute decompression, such as in the setting of infection or compromised renal function. In cases in which cystoscopic retrograde manipulation has been unsuccessful or may be hazardous, particularly in neonates or infants, placement of a percutaneous nephrostomy is preferred. This allows the performance of antegrade studies that will help define the nature and exact anatomic site of obstruction. It also allows decompression of the system in cases of associated infection or compromised renal function and allows assessment of recoverability of renal function after decompression. When there remains some doubt as to the clinical significance of a dilated collecting system, placement of a percutaneous nephrostomy tube allows access for dynamic pressure perfusion studies. First described by Whitaker in 1973, the renal pelvis is continuously perfused at 10 mL/min with normal saline solution or dilute radiographic contrast solution under fluoroscopic control. Renal pelvic pressure is monitored during the infusion, and the pressure gradient across the UPJ is determined. During the infusion, the bladder is continuously drained with an indwelling catheter to prevent transmission of intravesical pressures. Renal pelvic pressure ranging up to 12 to 15 cm H2O during this infusion suggests a nonobstructed system. In contrast, pressures in excess of 15 to 22 cm H2O are highly suggestive of a functional obstruction. Pressures between these extremes may be nondiagnostic (O’Reilly, 1986).
Although pressure perfusion studies can often provide valuable information regarding the functional significance of an apparent obstruction, these studies can at times be inaccurate. This inaccuracy may be a result of variations in renal pelvic anatomy and compliance (Koff et al, 1986) or positional variations (Ellis et al, 1995). As such, the urologist must be a diagnostician and collate the clinical presentation and results of all diagnostic studies performed in order to identify the best clinical intervention.
Indications and Options for Intervention
Contemporary indications for intervention for UPJ obstruction include the presence of symptoms associated with the obstruction, impairment of overall renal function or progressive impairment of ipsilateral function, development of stones or infection, or, rarely, causal hypertension. The primary goal of intervention is relief of symptoms and preservation or improvement of renal function. Traditionally, such intervention should be a reconstructive procedure aimed at restoring nonobstructed urinary flow. This is especially true for neonates, infants, or children in whom early repair is desirable because these patients will have the best chance for improvement in renal function after relief of obstruction (Bejjani and Belman, 1982; Roth and Gonzales, 1983; Wolpert et al, 1989). However, timing of the repair in neonates remains controversial (DiSandro and Kogan, 1998; Koff, 1998, 2000; Hanna, 2000; Shokeir and Nijman, 2000), mostly because of difficulty in defining those kidneys truly at risk for functional obstruction. In a prospective study of 104 neonates with primary unilateral hydronephrosis suspected of being caused by UPJ obstruction, after a mean follow-up of 21 months, only 7 (7%) required pyeloplasty for functional obstruction, defined as a progression of hydronephrosis or a 10% reduction in differential glomerular filtration rate on serial ultrasonography and diuretic renography (Koff and Campbell, 1994). All treated patients had a return of renal function to predetermination levels, supporting selective nonoperative management of neonatal hydronephrosis.
UPJ obstruction may not become apparent until middle age or later (Jacobs et al, 1979). Occasionally, if the patient is asymptomatic and the physiologic significance of the obstruction seems indeterminate, careful observation with serial follow-up studies may be appropriate, typically using diuretic renography. However, the majority of affected patients may ultimately benefit from reconstructive intervention (Jacobs et al, 1979; Clark and Malek, 1987; O’Reilly, 1989). When intervention is indicated, the procedure of choice has historically been dismembered pyeloplasty. However, less invasive endourologic approaches have a role as an alternative in many centers (Brannen et al, 1988; Motola et al, 1993a; Kletscher et al, 1995; Cohen et al, 1996; Nadler et al, 1996; Thomas et al, 1996; Tawfiek et al, 1998; Lechevallier et al, 1999; Gerber and Kim, 2000; Nakada, 2000; Conlin, 2002). Most recently, laparoscopic and robotic pyeloplasty has gained acceptance as primary therapy at centers with appropriate skills and technology (DiMarco et al, 2006; Rassweiler et al, 2007).
Although success rates with most endourologic techniques have not proven to be comparable with those of open, laparoscopic, or robotic pyeloplasty, it has been suggested that the success rates may be significantly improved with careful patient selection. In an important prospective study, Van Cangh and colleagues (1994) achieved an overall success rate for endopyelotomy of 73%. However, these investigators found the presence of crossing vessels to be a major determinant of outcome (42% success rate in the setting of a crossing vessel vs. 86% success without a crossing vessel). Furthermore, when endopyelotomy was applied to patients with “a high degree of obstruction,” the success rate was only 60% compared with an 81% success rate for those patients with “low-grade” obstruction. When patients with both a crossing vessel and a high degree of obstruction were excluded from analysis, the success rate improved to 95%, which is comparable with that of open pyeloplasty. However, other studies have suggested a less important role for these factors in regard to their impact on a successful outcome (Gupta et al, 1997; Danuser et al, 1998; Nakada et al, 1998).
Although the indications for intervention for UPJ obstruction are similar regardless of technique, it is critical to discuss the risks and benefits of all available options with patients. As such, each patient should be advised individually on the basis of all the anatomic and functional information available preoperatively. In this setting, many patients will opt for a minimally invasive approach, even with the understanding that success rates may be lower, or secondary intervention may become necessary. As a result of studies linking crossing vessels to hindered endourologic successes, there is increased interest in intraoperative management of the UPJ and crossing vessel by either an open or laparoscopic approach (Conlin, 2002). Therefore for “secondary” UPJ obstruction, it remains reasonable to recommend an open or laparoscopic approach to any patient who has failed primary endourologic management and an endourologic approach to those who have failed open or laparoscopic repair. The results of endourologic management in this setting remain generally excellent (Jabbour et al, 1998; Canes et al, 2008).
Rarely, nephrectomy may be the procedure of choice. Indications for nephrectomy as primary therapy include diminished function or nonfunction of the involved renal moiety and a normal contralateral kidney on the basis of radiographic and nuclear studies. These patients may be symptomatic with urinary tract infections or pain. In such cases, ultrasonography or CT scanning is generally performed and will reveal only a thin shell of parenchyma remaining. Renography can provide quantitative measures of renal function, and generally kidneys with less than 15% differential function are nonsalvageable in adults. If the potential for salvageability of function is still unclear, an internal stent or percutaneous nephrostomy may be placed for temporary relief of obstruction and renal function studies subsequently repeated. Nephrectomy may also be considered for patients in whom the obstruction has led to extensive stone disease with chronic infection and significant loss of function in the face of a normal contralateral kidney. Removal of the kidney may also be chosen over reconstruction for patients in whom repeated attempts at repair have already failed and in whom further intervention would therefore be extremely complicated. This option should be considered only when the contralateral kidney is essentially normal.
Options for Intervention
Endourologic Management
Operative intervention for UPJ obstruction has historically provided a widely patent, dependently positioned, well-funneled UPJ. In addition, the option to reduce the size of the renal pelvis is readily available with this approach. Although pyeloplasty has stood the test of time with a published success rate of 95%, several less invasive alternatives to standard operative reconstruction exist (Clark et al, 1987). The advantages of endourologic approaches include reduced hospital stays and postoperative recovery. However, the success rate does not approach that of open, laparoscopic, or robotic pyeloplasty. Furthermore, whereas open, laparoscopic, or robotic pyeloplasty can be applied to almost any anatomic variation of UPJ obstruction, consideration of any of the less invasive alternatives require that the surgeon take into account the degree of hydronephrosis, ipsilateral renal function, concomitant calculi, and possibly the presence of crossing vessels. Of note, Albani and colleagues (2004) reported contemporary long-term results with various endopyelotomy approaches to have a success rate of 67%, with the majority of failures in the first 32 months. More recently, DiMarco and colleagues (2006) reported long-term follow-up of more than 400 patients undergoing either percutaneous antegrade endopyelotomy or pyeloplasty. The 3-, 5-, and 10-year success rates were superior for pyeloplasty, 85% versus 63%, 80% versus 55%, and 75% versus 41%. Moreover, Rassweiler and colleagues (2007) compared retrograde laser endopyelotomy with laparoscopic retroperitoneal pyeloplasty in 256 patients in a 10-year single surgeon experience and found success rates were 73% for laser endopyelotomy compared with 94% for pyeloplasty.
Endourologic management of UPJ obstruction was introduced by Ramsay and colleagues in 1984 as a “percutaneous pyelolysis” and then popularized in the United States by Badlani and colleagues (1986), who coined the term “endopyelotomy.” Although various nuances in the technique have been described (Korth et al, 1988; Van Cangh et al, 1989; Ono et al, 1992), the basic concept of the endopyelotomy is a full-thickness lateral incision through the obstructing proximal ureter, from the ureteral lumen out to the peripelvic and periureteral fat. A stent is placed across the incision and left to heal, in keeping with the original work of Davis in 1943, who performed an “intubated ureterotomy” to repair UPJ obstruction. Subsequently, alternative techniques using a retrograde approach to the UPJ were developed. The retrograde approach most used today is the ureteroscopic approach, typically using the holmium laser to incise the UPJ under direct visual control. Alternatively, a cautery wire balloon endopyelotomy, which incises the UPJ under fluoroscopic control, or percutaneous endopyeloplasty may be used (Gill et al, 2002). Recently, Vaarala and colleagues reported a small series of 64 patients who underwent either antegrade or retrograde cold knife or cautery wire balloon endopyelotomy. In this study, success rates ranged from 79% to 83%, without statistically significant differences among the three treatments (Vaarala et al, 2008). Of note, transplantation complications are particularly suited to endoscopic management, either antegrade or retrograde (Schumacher et al, 2006; Gdor et al, 2008a). As far as efficacy is concerned, there is little evidence for significant differences between endopyelotomy techniques. The differences lie in technical considerations and complications.
Percutaneous Antegrade Endopyelotomy
Indications and Contraindications
The indications to intervene for any patient with UPJ obstruction include the presence of symptoms, progressive or overall impairment of renal function, development of upper tract stones or infection, or, rarely, causal hypertension. Historically, a percutaneous approach for definitive management of UPJ obstruction was offered only to those patients undergoing percutaneous removal of associated stones or to those who had previously failed open pyeloplasty. However, encouraging results ultimately led many centers to offer percutaneous endopyelotomy as primary therapy for almost any patient with UPJ obstruction. Even with the acceptance of laparoscopic pyeloplasty, percutaneous endopyelotomy is also appropriate for those patients with UPJ obstruction and concomitant pyelocalyceal stones, which can then be managed simultaneously. Contraindications to a percutaneous endopyelotomy are similar to the contraindications to any endourologic approach and include a long segment (>2 cm) of obstruction, active infection, or untreated coagulopathy. Whereas the impact of crossing vessels is controversial, the mere presence of crossing vessels is not a contraindication to an endopyelotomy (Motola et al, 1993b; Nakada et al, 1998; Lam et al, 2003b). However, significant entanglement of the UPJ by crossing vessels can occasionally be identified and this may render any endourologic approach unsuccessful. When such entanglement is suggested by intravenous or retrograde pyelography (Fig. 41–7), it can be reliably verified using three-dimensional helical CT (Kumon et al, 1997).
Patient Preparation
Patients undergoing a percutaneous endopyelotomy undergo preoperative evaluation and preparation as if they were undergoing any percutaneous, laparoscopic, or open renal intervention. The evaluation includes an assessment for any comorbidity that may increase the risk of anesthesia. Sterile urine should be ensured at the time of definitive intervention. If upper tract infection cannot be cleared because of obstruction, temporization should be accomplished using internal stenting or percutaneous nephrostomy drainage alone. The patient should be counseled as to the risks and benefits of the procedure, and in particular the fact that the success rate of any endourologic approach, including percutaneous endopyelotomy, may be less than that of operative intervention. Patients should also be counseled of the risk of bleeding requiring transfusion, urinary leak, drainage-related complications, and hydropneumothorax, particularly if upper pole access is used.
Technique
An endopyelotomy cannot be performed safely by any route until access across the UPJ is established. This can be accomplished in a retrograde fashion cystoscopically or in an antegrade manner percutaneously. For retrograde access, the UPJ can almost always be traversed using a hydrophilic wire passed through an open-end catheter. Once the hydrophilic wire is successfully positioned in the pyelocalyceal system, the open-end catheter is advanced over it into the renal pelvis. The wire can then be withdrawn so that contrast material can be injected through the open-end catheter to guide subsequent percutaneous access.
With the patient in the prone position, the site for percutaneous access is chosen to allow straightforward access to the UPJ. Generally, a midposterior or superolateral calyx is chosen, although, occasionally, an inferolateral calyx may be used. Typically, the UPJ can be intubated in an antegrade fashion when the tract is initially established with fluoroscopic control. Alternatively, once the tract is dilated and nephroscopy is performed, a wire can again be passed in a retrograde fashion through the open-end catheter and grasped from above so that through-and-through access is reestablished. In either case, as soon as access is obtained with one wire, an introducing catheter is used to pass a second wire as a “safety wire,” so a working and a safety wire are now both in place. At this point, percutaneous access is complete and the endopyelotomy may be performed.
In the original descriptions of the technique both from the Institute of Urology in London (Ramsay et al, 1984) and from Long Island Jewish Hospital in New York (Badlani et al, 1986), the endopyelotomy was performed using a cold knife technique under direct vision. With one or two wires in place across the UPJ, a direct vision “endopyelotome” is used. This hook-shaped cold knife may be used to completely incise the UPJ in a full-thickness manner, from the ureteral lumen to periureteral and peripelvic fat (Fig. 41–8). Rigorous anatomic studies have shown the incision should generally be made laterally because this is the location devoid of crossing vessels (Sampaio, 1998). However, in cases of high insertion, the incision should instead “marsupialize” the proximal ureter into the renal pelvis, such that an anterior or posterior incision may be required (Fig. 41–9). When such incisions are done under direct vision, any crossing vessel can be directly visualized and avoided. In addition to the endopyelotome, the holmium laser or the cutting balloon catheter may also be used to perform an antegrade endopyelotomy.
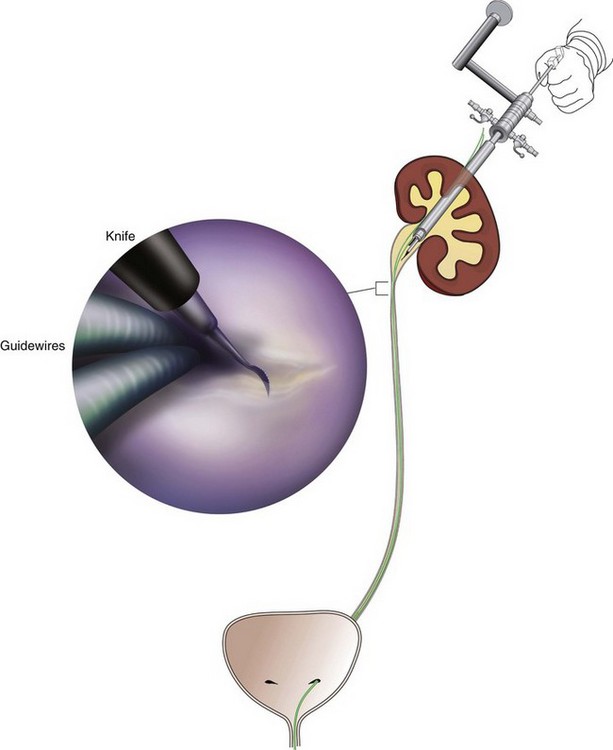
Figure 41–8 Percutaneous nephroscopic view of “cold knife” endopyelotomy (inset). The line of incision is delineated by two guidewires, which have been passed across the ureteropelvic junction in an antegrade fashion through a superior calyx using a rigid nephroscope through a 30-Fr sheath. The lateral incision is performed under direct visual control.
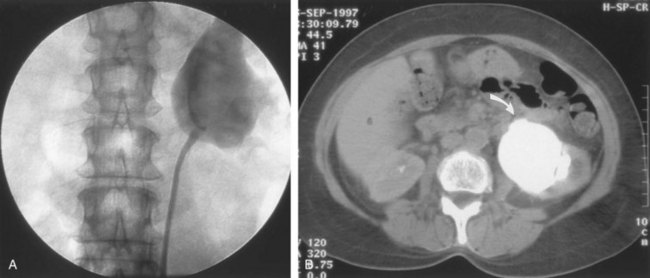
Figure 41–9 A, Retrograde study in this patient with left ureteropelvic junction obstruction reveals a “high insertion” of the left ureter. B, CT scan in this same patient reveals the ureter inserting on the anatomically anterior aspect of the renal pelvis. A marsupializing incision must be made in a true posterior direction from the ureter into the renal pelvis.
Once the incision is complete, stenting is accomplished. There remains no consensus as to the optimal stent size or duration for endopyelotomy. A No. 14/7-Fr endopyelotomy stent may be used, passed in an antegrade fashion with the larger diameter end of the stent positioned across the UPJ. In some cases, especially when the patient has not been prestented, passage of this large-caliber stent may be difficult. In those instances, a No. 10/7-Fr endopyelotomy stent or even a standard No. 8-Fr internal stent may be used without compromising the ultimate outcome. Once proper positioning of the stent is determined fluoroscopically, any remaining safety wires are withdrawn. One group showed no difference between larger and standard stents in a porcine study of endopyelotomies (Moon et al, 1995). Alternatively, Danuser and colleagues (2001) demonstrated improved success rates using a modified 27-Fr stent following percutaneous endopyelotomy at nearly 2 years follow-up.
In the setting of a high insertion, the incision can often be extended to the dependent portion of the renal pelvis under direct vision, bridging the gap between the lateral wall of the ureter and the medial wall of the pelvis, across the periureteral and peripelvic fat. Once the incision is complete, the stent is already in place and nephrostomy drainage is instituted for 24 to 48 hours.
Postoperative Care
Avoidance of strenuous activity for 8 to 10 days after the procedure is recommended. The ideal stent size, duration of stent placement, and radiographic follow-up after endopyelotomy remains unclear (Canes et al, 2008). One study did report a benefit to larger stents in patients undergoing antegrade endopyelotomy (71% vs. 93%); however, a large-bore (27-Fr) catheter was used for the initial 3 weeks postoperatively (Danuser et al, 2001). On the other hand, Kletscher and colleagues (1995) reported no benefit to larger stents as did Hwang and colleagues (1996). Wolf and colleagues (1997) reported improved success using larger stents (12 Fr) in endoureterotomy patients in a retrospective review. Regarding stent duration, less is known. The original report and recommendation of 6 weeks by Davis (1943) is still often used, although Mandhani and colleagues (2003) identified no difference in results when comparing 57 patients stented for 2 weeks compared with 4 weeks. Although the need for prophylactic antibiotics while the stent is indwelling is not literature based, many use a daily suppressive dose.
Once the stent is removed, the patient returns 1 month later for clinical follow-up and radiographic evaluation. This generally includes a history, physical, urinalysis, and diuretic renography. If the patient remains asymptomatic and the diuretic renography reveals normal drainage (normal 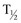 ), reevaluation is performed at 6 months and then at 12-month intervals. Most literature indicates that the majority of endopyelotomy failures occur within the first year of the procedure; however, longer-term studies demonstrate failures well beyond that timeframe (Nadler et al, 1996; Albani et al, 2004; DiMarco et al, 2006; Doo et al, 2007). For most adults, 2- and 3-year follow-up is justified because studies indicate even at 36 months some late failures are identified, but relatively few are identified at 60 months (Doo et al, 2007).
), reevaluation is performed at 6 months and then at 12-month intervals. Most literature indicates that the majority of endopyelotomy failures occur within the first year of the procedure; however, longer-term studies demonstrate failures well beyond that timeframe (Nadler et al, 1996; Albani et al, 2004; DiMarco et al, 2006; Doo et al, 2007). For most adults, 2- and 3-year follow-up is justified because studies indicate even at 36 months some late failures are identified, but relatively few are identified at 60 months (Doo et al, 2007).
Results
The immediate and long-term results of percutaneous endopyelotomy are well established. Although percutaneous endopyelotomy compares favorably with open operative pyeloplasty in terms of postoperative pain, length of hospital stay, and return to prehospitalization activities (Brooks et al, 1995; Karlin et al, 1988), retrograde endopyelotomy and laparoscopy also offer favorable convalescence.
Gerber and Lyon, in 1994, reviewed the outcome of percutaneous endopyelotomy in 672 patients reported from 12 centers and found a success rate ranging from 57% to 100% (mean, 73.5%) at follow-up ranging from 2 to 96 months. Currently, success rates approaching 85% to 90% are being reported at experienced centers, with little difference in outcome noted in those patients undergoing the procedure for primary versus secondary UPJ obstruction (Motola et al, 1993a; Kletscher et al, 1995; Shalhav et al, 1998). Of note, Knudsen and colleagues (2004) reported long-term results in 80 patients using the cold knife and holmium laser for antegrade endopyelotomy, with 55-month follow-up. This series had a success rate of 67%, slightly lower than otherwise reported. Interestingly, DiMarco and colleagues (2006) reported on 182 antegrade endopyelotomies with a recurrence-free survival at a single center over 10 years as low as 41%. Of note, Schumacher and colleagues (2006) reported on three successful antegrade endopyelotomies in transplanted kidneys in 2006.
When percutaneous endopyelotomy does fail, several options exist including a retrograde endopyelotomy, repeat percutaneous endopyelotomy, or laparoscopic, robotic, or open operative intervention. There remains a role for spiral CT angiography in failed endopyelotomy, to rule out a crossing vessel. If a significant vessel is found, repeat endopyelotomy is generally not recommended (Nakada, 2000). Alternatively, operative intervention is generally offered to any patient who has failed an endourologic approach. On the basis of available data, the results of laparoscopic pyeloplasty will not be compromised (Motola et al, 1993b; Gupta et al, 1997; Conlin et al 2002).
Complications
The complications associated with percutaneous endopyelotomy are analogous to those associated with percutaneous nephrolithotomy (Badlani et al, 1988; Weiss et al, 1988; Cassis et al, 1991; Malden et al, 1992; Bellman, 1996), and hemorrhage is a risk of any percutaneous upper tract procedure including endopyelotomy. However, because in patients with UPJ obstruction the renal parenchyma is generally thinner than that associated with a normal kidney, and because the collecting system is dilated, this risk may be different than that in the general population of stone patients undergoing percutaneous manipulation. Acute management in this setting is generally conservative to start: bed rest, hydration, and transfusion if necessary. The nephrostomy tube should not be irrigated acutely. Rather, it is preferable to allow the pyelocalyceal system to tamponade the bleeding. When continued bleeding does not respond to these conservative measures, the next step is selective angiographic embolization. Generally, the urologist should have a low threshold to proceeding to angiography in order to minimize the need for transfusion and potential exploration. Successful angiographic embolization often obviates the need for operative “exploration” that can lead to nephrectomy.
Infection is a risk of any urinary tract manipulation including percutaneous endopyelotomy, and all attempts should be made to sterilize the urinary tract before the procedure. Whereas the role of prophylactic antibiotics at the outset of the procedure in the setting of a sterile urine is unproven, most urologists give a second-generation cephalosporin “on call” to the procedure. Consideration should be given to the use of prophylactic antibiotics while the endopyelotomy stent is indwelling for the month after the procedure, especially in women who are more prone to bacteruria.
Persistent obstruction is rare in the early postoperative period because of the internal stent. Occasionally, the stent can be obstructed from blood clots, and continued nephrostomy drainage for a few days typically allows the problem to resolve spontaneously.
Percutaneous Endopyeloplasty
Percutaneous endopyeloplasty is a hybrid technique described as an endoscopic Heineke Mikuliz repair performed through a percutaneous tract. In other words, endopyeloplasty combines percutaneous endopyelotomy and an endoscopic Fenger plasty. Initial and short-term reports by Gill and colleagues have been published demonstrating both feasibility and safety, and success rates have been similar to percutaneous endopyelotomy in limited comparative series (Gill et al, 2002; Canes et al, 2008). Latest reports include 55 patients with short-term follow-up with more than 90% success by these same authors (Stein et al, 2007). Early reports identified endopyeloplasty to be more effective in primary rather than secondary UPJ obstruction, most likely because tissue scarring may inhibit the endoscopic reconstruction.
Simultaneous Percutaneous Endopyelotomy and Nephrolithotomy
Percutaneous endopyelotomy is particularly favorable when the UPJ obstruction is associated with upper tract stone disease because the stones can be managed concomitantly. In such cases, percutaneous access is again established with a wire across the UPJ. The stone should be removed before the endopyelotomy so that stone fragments do not migrate into the peripyeloureteral tissue, as can happen if the endopyelotomy is performed first. Otherwise, localized obstruction may result from fibrosis or granuloma formation (Giddens et al, 2000; Streem, 2000). The urologist must take care to assure that the UPJ obstruction is not a result of edema from the concominant stone disease, in particular with stone disease in the renal pelvis. In this circumstance, initial management of the stone percutaneously and subsequent radiographic assessment of the UPJ once the stone has been removed are most prudent. In addition, if a nephrostomy tube is retained, a Whitaker test is straightforward and definitive to assess for persistent obstruction. Conversely, UPJ obstruction and solitary lower pole calculi do not represent a dilemma regarding UPJ edema, and combined percutaneous management remains most efficient. Alternatively, laparoscopic pyeloplasty and concominant stone removal is also effective for these patients.
Retrograde Ureteroscopic Endopyelotomy
A ureteroscopic approach to endopyelotomy was first suggested in 1985 when Bagley and colleagues reported a combined percutaneous and flexible ureteroscopic procedure approach for management of an “obliterated” UPJ. Subsequently, Inglis and Tolley (1986) reported a ureteroscopic “pyelolysis” for UPJ obstruction. Shortly thereafter, Clayman and colleagues (1990) reported an initial experience in a small number of patients performing ureteroscopic endopyelotomy with a No. 3- or 5-Fr cutting electrode passed under direct vision using large, rigid or flexible ureteroscopes. In that series, however, a No. 8-Fr nephrostomy tube was placed at the outset of the procedure and left indwelling for at least 48 hours. As such, that series still represented a “combined” endourologic approach to endopyelotomy. Stents were routinely left in place for 6 to 8 weeks, after which diagnostic studies were performed. With a mean follow-up approaching 1 year, a success rate of 81% was achieved in 16 patients. However, two patients developed distal ureteral strictures, probably resulting from the larger-diameter rigid instrumentation.
Thomas and colleagues (1996) subsequently reported their experience with ureteroscopic endopyelotomy. Again, only relatively larger-diameter ureteroscopic instrumentation was available such that preoperative stent placement was routine, and some male patients required perineal urethrostomy. The endopyelotomy incision itself was performed with either cold knife or electrocautery attachments to the ureteroscope. The authors achieved a success rate of approximately 90%, although nephrectomy was ultimately performed in two patients, one of which was done urgently for bleeding. Cold knife ureteroscopic endopyelotomies are still being reported (e.g., Butani and Eschghi reported on a single surgeon experience with 135 cases from 1998-2004). Although three rigid ureteroscope and preprocedure stents were necessary, this group identified 96% success rates in primary procedures with an average 5-year follow-up (Butani and Eschghi, 2008). Notably, the complication rate was only 2.7%.
Advances in instrumentation and technique now allow a ureteroscopic approach to be performed reliably at a single setting (Conlin and Bagley, 1998), and this is now considered the standard. The main advantage of a ureteroscopic approach is that it allows direct visualization of the UPJ and assurance of a properly situated, full-thickness endopyelotomy incision without the need for percutaneous access. Another advantage of the ureteroscopic approach is a decrease in cost compared with the use of the cautery wire balloon, assuming ureteroscopic equipment and electroincision or holmium laser is already available. Moreover, the risks and morbidity of percutaneous access are avoided with the ureteroscopic procedure. Gettman and colleagues found that the retrograde ureteroscopic endopyelotomy was more cost effective than hot wire cutting balloon endopyelotomy, antegrade endopyelotomy, and pyeloplasty for treating UPJ obstruction when taking into account treatment failures (Gettman et al, 2003).
Indications and Contraindications
The indications and contraindications to a ureteroscopic endopyelotomy include functionally significant obstruction, as defined earlier. Contraindications include relatively long areas of obstruction and upper tract stones, which are best managed simultaneously with alternative approaches, usually percutaneously or laparoscopically. Another consideration is that in patients with significant hydronephrosis, the evidence indicates an antegrade endopyelotomy may be more efficacious (Lam et al, 2003b).
Technique
The instrument that allows the most straightforward retrograde access to the UPJ, as well as providing an effective working channel, is a small caliber (≤7-Fr) semirigid ureteroscope. In women, the UPJ can often be reached with a 6.9-Fr semirigid ureteroscope. In men, small-caliber (≤7.5-Fr) actively deflecting flexible ureteroscopes are typically used, and today with availability of improved ureteral access sheaths and improved flexible ureteroscopes, many retrograde endopyelotomies are done using the flexible ureteroscope.
General anesthesia is used in order to minimize patient movement during ureteroscopy and the subsequent incision of the UPJ. In preparation for the endopyelotomy, a retrograde pyelogram is performed under fluoroscopic control at the outset of the procedure. A hydrophilic guidewire is passed cystoscopically under fluoroscopic control and coiled in the pyelocalyceal system. The cystoscope is then withdrawn and exchanged for the semirigid ureteroscope. The ureteroscope is passed alongside the guidewire to the level of the UPJ. If the distal ureter is too narrow to allow easy passage of the ureteroscope, the intramural ureter can be dilated using a 5-mm balloon or a No. 9- or 10-Fr “introducing” catheter. If the ureter is still too narrow at any point to easily accommodate the ureteroscope, then an internal stent is placed and the procedure postponed for 5 to 10 days to allow “passive” ureteral dilatation. Alternatively, an actively deflecting flexible ureteroscope may be used, and in most cases a ureteral access sheath is quite useful. The sheath allows for rapid transfer of the ureteroscope for assessment of the UPJ. Once the flexible ureteroscope is passed to the UPJ, a 200-µm holmium fiber is placed through the working channel and the UPJ incised in the appropriate location, as suggested by the radiographic studies (Figs. 41-10 and 41-11).
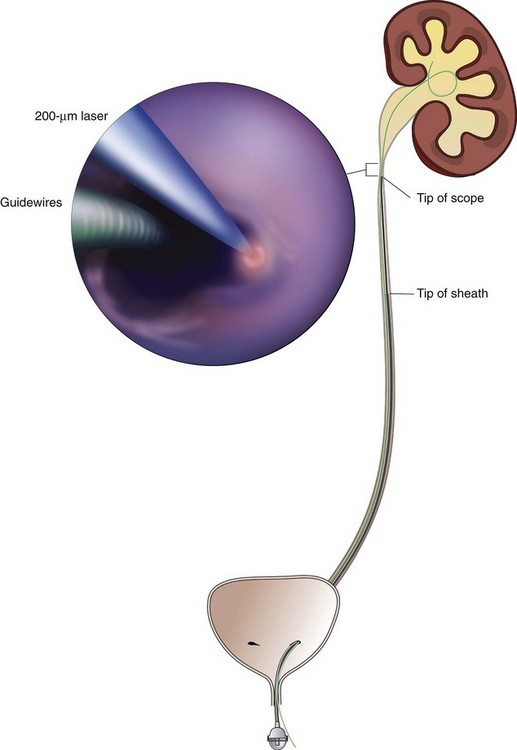
Figure 41–10 Flexible ureteroscopic endopyelotomy using holmium laser, demonstrating endoscopic view of the ureteropelvic junction (inset). A safety wire is in place, and the ureteroscope is passed through a ureteral access sheath as a lateral incision is being made under endoscopic view, using holmium laser fiber. A properly sited, complete incision is straightforward with this direct visualization technique.
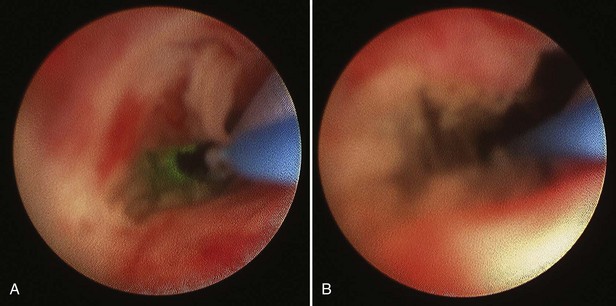
Figure 41–11 A, Endoscopic view of ureteropelvic junction (UPJ) stenosis with safety wire and laser fiber during incision. B, Following incision, note full-thickness incision and minimal bleeding with capacious UPJ.
Once the UPJ is reached with the ureteroscope, the renal pelvis is drained to assist movement across the UPJ during the incision. When using a semirigid ureteroscope, the 200- or 365-µm holmium laser fiber is inserted through the working channel as the ureteroscope is positioned at the proximal extent of the UPJ or in the renal pelvis itself. At a setting of 0.8 to 1.2 Joules and a frequency of 10 to 15 Hz, the UPJ is incised, usually in a posterolateral direction, while the ureteroscope is withdrawn back down across the UPJ. This procedure is repeated, and the incision gradually deepened to extend into the peripelvic and periureteral retroperitoneal space. Because this is done gradually and under direct vision, any visualized vessels, and thus potentially significant bleeding, are usually avoided.
The incision is carried caudally into normal ureteral tissue, until the UPJ is widely patent. Injection of contrast material through the ureteroscope can demonstrate extravasation and confirm an adequate depth of incision, although this is generally not necessary because the entire procedure has been performed under direct vision. Balloon dilation up to 24-Fr can also be performed to complete the incision. If any small bleeding points are visualized ureteroscopically, they can be treated by defocusing the holmium laser. Similarly, the balloon can be reinflated to allow tamponade for 10 minutes to see if the bleeding will subside. The ureteroscope is then withdrawn from the ureter while the safety wire is left in place in the renal pelvis for subsequent passage of a stent. Experimental studies have shown that 36-Fr balloon dilation alone can create linear incisions in the UPJ (Pearle et al, 1994). Although retrograde balloon dilation alone has been reported for treatment of UPJ obstruction, long-term follow-up studies have shown a diminishing success rate over time, as low as 42% (McClinton et al, 1993; Webber et al, 1997).
Once the ureteroscope has been removed, a stent is advanced over the remaining wire using fluoroscopic guidance. A Foley catheter is left indwelling, again to obviate the risk of reflux and extravasation at the site of the endopyelotomy incision and to rapidly identify any significant bleeding. Diuretic renography is performed 4 weeks after stent removal to assess results. Clinical and radiographic follow-up is then continued at 6- to 12-month intervals for 24 to 32 months.
Results
Biyani and colleagues (1997) described their initial experience with a ureteroscopic approach using holmium laser energy. With a mean follow-up of slightly more than 12 months, they achieved a success rate of 87.5% in a small group of patients. One patient developed a urinoma, which was managed conservatively. In 1998, Renner and colleagues reported a larger series of patients undergoing ureteroscopic laser endopyelotomy. Using a semirigid ureteroscope, the UPJ was incised at a posterolateral location unless vessels were visualized in that area, in which case a contralateral incision was made. Tawfiek and colleagues (1998) reported the Jefferson Medical College experience with ureteroscopic endopyelotomy. These investigators combined endoluminal ultrasound with their ureteroscopic approach in order to definitively identify crossing vessels or a ureteropelvic septum, which is present in patients with high-inserting ureters. The authors believed this helped them definitively site their endopyelotomy incision. Different modalities were used for the endopyelotomy itself including electrocautery and holmium laser. An 87.5% success rate was achieved in 32 patients. There were no significant bleeding complications, and all patients were discharged within 24 hours of the procedure.
Gerber and colleagues and Matin and colleagues reported experiences with ureteroscopic holmium laser endopyelotomy, demonstrating success rates of 70% to 80% with follow-up out to 5 years (Gerber et al, 2000; Matin et al, 2003). More recently, Yanke reported on 128 retrograde ureteroscopic endopyelotomies with a 60% success rate at 20 months while Rassweiler and colleagues reported 73% success in 113 patients at 63 months (Rassweiler et al, 2007; Yanke et al, 2008). Improved results were reported by Conlin and associates (91% success rates) with retrograde endopyelotomy in patients when culling patients with crossing vessels greater than 4 mm using preoperative ultrasonography (Conlin, 2002). Giddens and colleagues also published excellent results when culling patients with anterior and posterior crossing vessels from retrograde endopyelotomy using endoluminal ultrasound (Giddens et al, 2000). To date, the use of endoluminal ultrasound to identify crossing vessels has been controversial, and although it may play a role in preoperative decision making, similar data can be obtained using the less invasive spiral CT angiography. Regardless, the best endopyelotomy success rates still lag behind those of open or laparoscopic pyeloplasty.
Complications
Complications of this approach have diminished in frequency and severity with the refinement of ureteroscopic instrumentation and the introduction of small-caliber holmium laser fibers. Postprocedural ureteral strictures are rare in contemporary series, and angiographic embolization and nephrectomy are rare using the retrograde approach. Most complications are minor and relate primarily to urinary leak, stent migration, and infection (Gerber and Kim, 2000; Tawfiek et al, 1998). Recently Castle and colleagues reported on ureteroarterial fistula 2 weeks following retrograde laser endopyelotomy, which could be fulgurated ureteroscopically (Castle et al, 2009).
Retrograde Cautery Wire Balloon Endopyelotomy
Use of a cautery wire balloon for management of UPJ obstruction was first reported in a clinical series by Chandhoke and colleagues in 1993. Use of this device gained initial acceptance by many clinicians because standard cystoscopic techniques and real-time fluoroscopy are all that is necessary for its use. Because the procedure is guided fluoroscopically, such vessels may increase the risk of hemorrhage after activation of the cautery wire balloon. Some authors recommended preoperative imaging for such vessels with relatively noninvasive techniques such as CT or three-dimensional CT angiography (Fig. 41–12) (Streem and Geisinger, 1995; Quillin et al, 1996; Nakada et al, 1998; Herts et al, 1999; Nakada, 2000). Nadler and colleagues (1996) reported on 28 patients 2 or more years after cautery wire balloon endopyelotomy. With a mean follow-up of 32.5 months, subjective improvement was noted in 61% of patients, and 81% had a patent UPJ on the basis of diuretic renography or Whitaker testing. More recent studies have demonstrated lower success rates than these initial series (32% to 63%) and perhaps that high-grade hydronephrosis has a negative impact on success (Albani et al, 2004; Sofras et al, 2004). El-Nahas and colleagues reported a small prospective randomized trial comparing retrograde ureteroscopic endopyelotomy to the hot-wire balloon endopyelotomy in 40 patients. Although not statistically significant, they found superior success rates (85% compared with 65%) and lower complication rates with the ureteroscopic endopyelotomy (El-Nahas et al, 2006). Ponsky and Streem reported on 64 patients undergoing either ureteroscopic endopyelotomy or hot wire balloon endopyelotomy and found equivalent success rates with both procedures yet higher major complication rates in the cautery wire balloon endopyelotomy, specifically transfusion and selective embolization (Ponsky and Streem, 2006). The major complication associated with cautery wire balloon incision is hemorrhage. Although injury to crossing vessels has been reported using the cutting balloon catheter, strict adherence to lateral incision principles minimizes this risk (Sampaio et al, 1993; Streem and Geisinger, 1995; Wagner et al, 1996). In addition, although some believe crossing vessels hinder success rates primarily, others believe hemorrhage is a real concern (Aslan et al, 1998). Currently, improved ureteroscopic instrumentation and the benefits of direct endoscopic visualization make ureteroscopic endopyelotomy the more pervasive retrograde approach.
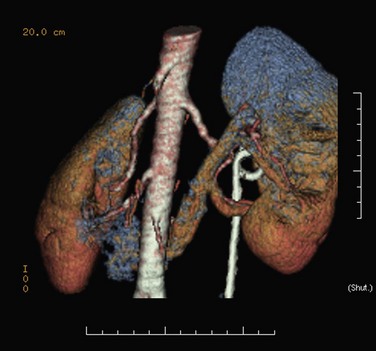
Figure 41–12 Spiral CT angiography with three-dimensional reconstruction clearly identifies an accessory lower pole crossing vessels at the level of the ureteropelvic junction (UPJ) in a patient with UPJ obstruction. This case was managed successfully with laparoscopic pyeloplasty.
Key Points: Endourologic Management of UPJ Obstruction
Laparoscopic and Robotic Intervention
Laparoscopic Pyeloplasty
Laparoscopic approach to pyeloplasty was first introduced in 1993 by Schuessler and colleagues (1993) and has been developed worldwide as a viable minimally invasive alternative to open pyeloplasty and endopyelotomy. Relative to both open pyeloplasty and endopyelotomy, laparoscopic pyeloplasty is associated with greater technical complexity and a steeper learning curve. In the hands of the experienced laparoscopic surgeons, it has been shown to provide lower patient morbidity, shorter hospitalization, and faster convalescence, with the reported success rates matching those of open pyeloplasty (≥90%). Following the similar surgical principles of anatomic dissection and repair used in open pyeloplasty, laparoscopic pyeloplasty has been shown to provide the success rates surpassing those of endopyelotomy by approximately 10% to 30%.
Indications and Contraindications
The indications and contraindications for a laparoscopic repair are similar to those for either an endourologic or an open operative procedure. Indications to intervene include the presence of clinical symptoms of ureteropelvic junction obstruction, the progressive impairment of renal function, and the development of ipsilateral upper tract calculi or infection. Cases requiring the transposition of crossing vessels obstructing ureteropelvic junction or the size reduction for massively dilated renal pelvis are suitable for the laparoscopic approach. Absolute contraindications to intervene include the presence of uncorrected coagulopathy, the absence of adequate treatment of active urinary tract infection, and the presence of cardiopulmonary compromise unsuitable for surgery. The objective of the laparoscopic surgery is to provide a tension-free, water-tight repair with a funnel-shaped drainage product to relieve clinical symptoms and to preserve renal function.
Techniques
Four laparoscopic techniques for pyeloplasty have been described in the literature including the standard transperitoneal approach, retroperitoneal approach, anterior extraperitoneal approach, and robotic-assisted approach. For each approach, a dismembered Andersen-Hynes pyeloplasty, which is preferred by most surgeons, or one of the nondismembered methods such as Y-V plasty and flap pyeloplasty (Culp) analogous to those described for the open pyeloplasty can be used.
Transperitoneal Laparoscopic Approach
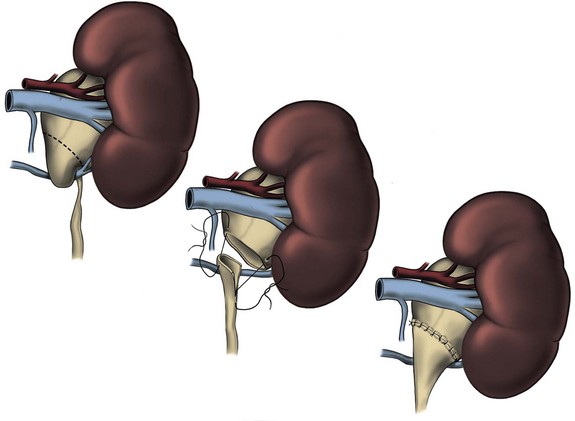
The initial transperitoneal approach to laparoscopic pyeloplasty was first described by Schuessler and colleagues (1993) and Kavoussi and colleagues (1993), and this approach has been the most widely used laparoscopic method due to its associated large working space and familiar anatomy. Before the laparoscopic portion of the procedure, cystoscopy with retrograde pyelography is first performed to define the anatomy and confirm the diagnosis, followed by placement of a ureteral stent and a urethral Foley catheter. The patient is placed in a 45-degree lateral decubitus position, and access to the peritoneal cavity is obtained via either the Veress needle or the Hassan access technique. Three to five laparoscopic ports are placed after the creation of CO2 pneumoperitoneum. Typically the umbilical port is for the laparoscope use. Colonic mobilization to expose the retropetioneal structures is the initial step of the laparoscopic procedure, although transmesenteric approach without bowel mobilization has been reported if renal pelvis or ureter can be readily recognized through the descending colonic mesentery (Romero et al, 2006). After medial mobilization of the colon, the ureter is identified and dissected in the cephalad direction to achieve mobilization of the ipsilateral proximal ureter, ureteropelvic junction, and renal pelvis (Fig. 41–13A). Extensive dissection of the ureter and excessive electrocautery use in close proximity to the ureter should be avoided to minimize injury to its vascular supply. At this time, the anatomy of the proximal ureter, renal pelvis, and nearby vasculature are carefully examined to determine the etiology of the ureteropelvic junction obstruction and the appropriate type of surgical repair. The general methods and principles of various types of surgical repair for laparoscopic pyeloplasty are identical to those described for open pyelplasty. If dismembered pyeloplasty is to be performed, which is suitable for the presence of crossing vessels, the renal pelvis is first transected circumferentially above the ureteropelvic junction and the lateral aspect of the proximal ureter is spatulated (Fig. 41–13B). The renal pelvis and proximal ureter are then transposed to the opposite side of the crossing vessel, if such vessel is present, and the ureteropelvic anastomosis is then completed with intracorporeal suturing techniques (Fig. 41–13C-D). In the presence of redundant renal pelvis, reduction pelvioplasty may be performed by excising redundant renal pelvic tissue and closing the pyelotomy. The actual laparoscopic suturing maneuver can be accomplished either freehand or with a semiautomated device (EndoStitch, US Surgical, Newark, CT). Either continuous running or simple interrupted suturing method may be used in the dismembered laparoscopic pyeloplasty, typically with the 4-0 absorbable suture. A surgical drain is placed after the completion of the anastomosis, and one of the trocar sites is typically used as the drain exit site.
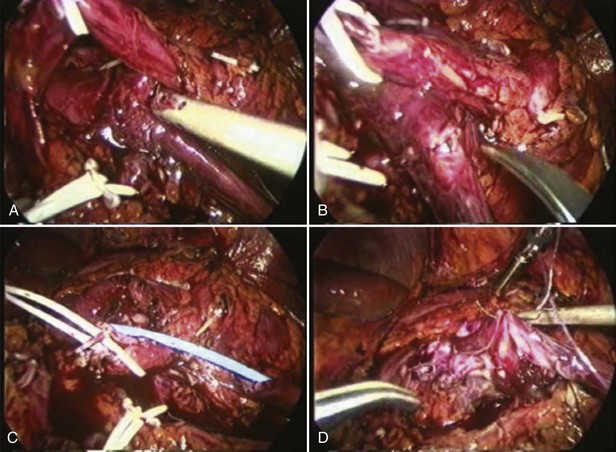
Figure 41–13 A, Transperitoneoscopic view of a patient with left pyelplasty. The patient’s head is to the left. Note the proximal ureter is sandwiched between an anterior crossing lower pole renal artery (wrapped with white vessel loop and retracted upward) and a posterior crossing lower pole vessel (wrapped with white vessel loop). The suction-irrigator tip points at the left ureteropelvic junction. B, The left proximal ureter is being transected with laparoscopic scissors, revealing the preplaced ureteral stent within the ureteral lumen. C, The ureteral stent is transposed anterior to the anteriorly crossing vessel following circumferential transection of the proximal left ureter. D, Ureteropelvic anastomosis is completed with intracorporeal suturing techniques.
Retroperitoneal Laparoscopic Approach
The initial retroperitoneoscopic approach to pyeloplasty was first reported by Janetschek and colleagues (1996). Cystoscopy with retrograde pyelography and ureteral stent placement are first performed as described earlier. For the retroperitoneal approach, the patient is usually positioned in the flank position with the use of flexion and elevation of the kidney rest. Following Hassan access technique to enter the retroperitoneum, a retroperitoneal working space can be created with balloon dilation. Following CO2 pneumoretroperitoneum, three to four laparoscopic ports are used to perform the laparoscopic pyeloplasty. The ureter is usually identified early in the procedure, and the dissection, mobilization, and ureteropelvic junction repair steps are identical to those described for the transperitoneal approach (Fig. 41–14).
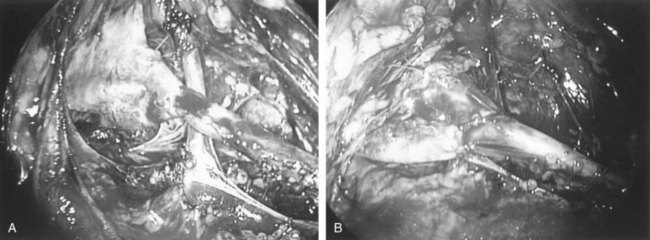
Figure 41–14 A, Retroperitoneal laparoscopic photograph of a case with right ureteropelvic junction (UPJ) obstruction. Note the presence of a lower pole crossing renal vessel anterior to the UPJ. B, Intraoperative view of completed ureteropelvic anastomosis after retroperitoneal laparoscopic dismembered pyeloplasty.
Anterior Extraperitoneal Laparoscopic Approach
The anterior extraperitoneal laparoscopic approach to pyeloplasty was first described by Hsu and colleagues (2003). Cystoscopy with retrograde pyelography and ureteral stent placement are first performed as described earlier. For the anterior extraperitoneal approach, the patient is placed in a 45-degree lateral decubitus position. Access to the preperitoneal space is obtained using open surgical techniques via a 10-mm incision, after which a large preperitoneal space is created with balloon dilation. Following CO2 insufflation and placement of four ports, the boundary between the retroperitoneal fat and peritoneal sac is identified and developed, allowing medial mobilization of the peritoneal sac containing the bowel contents en bloc. Subsequently, full exposure of the anterior aspects of the retroperitoneal structures including the ipsilateral ureter and kidney comes into view. The proximal ureter, ureteropelvic junction, and renal pelvis are identified, dissected, mobilized, and repaired as in the transperitoneal laparoscopic pyeloplasty. The entire procedure is completed in an extraperitoneal manner. A surgical drain is similarly placed at the end of the procedure.
Robotic-Assisted Laparoscopic Approach
The robotic-assisted laparoscopic pyeloplasty in the experimental setting was first reported by Sung and colleagues (1999). Its feasibility was subsequently confirmed with worldwide clinical application in recent years (Palese et al, 2005; Mufarrij et al, 2007; Schwentner et al, 2007; Yanke et al, 2008). The most widely used robotic system in the clinical setting today is the da Vinci Robot (Intuitive Surgical, Sunnyvale, CA), and the reported benefits of the robot include enhanced three-dimensional vision, motion scaling, tremor reduction, improved dexterity, and increased range of motion. Typically the procedure is performed in a transperitoneal manner providing a larger working space for the robotic arms, although the feasibility of retroperitoneal approach has been recently demonstrated in a small number of patients (Kaouk et al, 2008). Ureteral stent may be placed via a cystoscopic retrograde or laparoscopic antegrade manner. In both transperitoneal and retroperitoneal approaches, at least four different trocars are used in a robotic-assisted procedure including three for the robotic arms and one for the surgical assistant to perform suction, irrigation, retraction, and suture introduction. Following the initial laparoscopic access and trocar placement, the robotic system is placed in close proximity to the operating table and the robotic arms are attached to the laparoscope and specifically designed laparoscopic instruments. The surgeon at the console operates via the control of the robotic arms, while the assistant remains at the bedside and performs suction, retraction, exchange of laparoscopic instruments, suture needle introduction, and removal. The general surgical steps are identical to those described for non-robotic-assisted laparoscopic pyeloplasty.
Postoperative Care and Complications
Typically, a clear liquid diet is initiated on postoperative day 1 and advanced rapidly. Perioperative prophylactic antibiotic coverage is maintained. The Foley catheter is usually removed 24 to 36 hours postoperatively, and the surgical drain is removed before hospital discharge if the drain output remains negligible. If the drain output increases after the Foley catheter removal, the Foley catheter should be replaced for 7 days to eliminate urinary reflux along the stent in the treated ureter and decrease urinary extravasation at the ureteropelvic anastomosis. The ureteral stent is typically removed 4 to 6 weeks later in an outpatient setting, and follow-up including the use of imaging studies such as diuretic renal scan is performed as for any open pyeloplasty. Most of the complications of laparoscopic pyeloplasty are similar to those of general laparoscopic procedures including colonic injury, hemorrhage, ileus, pneumonia, congestive heart failure, thrombophlebitis, and urinoma formation. In the first 100 cases of laparoscopic pyeloplasty performed at Johns Hopkins (Jarrett et al, 2002), such complications occurred in 12% of the patients. Another large-scale review involving 189 cases of laparoscopic pyeloplasty identified approximately 2% to 2.3% intraoperative complication rate and 12.9% to 15.8% postoperative complication rate (Rassweiler et al, 2008). A typical postoperative appearance of a laparoscopic pyeloplasty patient is illustrated in Figure 41–15.
Results
Most of the published laparoscopic pyeloplasty reports have used the classic Andersen-Hynes dismembered technique because most laparoscopic surgeons attempt to duplicate the well-established principles of open surgery (Janetschek et al, 2000; Eden et al, 2001; Soulie et al, 2001; Jarrett et al, 2002; Turk et al, 2002; Inagaki et al, 2005; Bachmann et al, 2006; Rassweiler et al, 2008). The overwhelming majority of patients in these recent series had primary laparoscopic pyeloplasties, and the mean operative times are in the range of 119 to 252 minutes. In the experienced hands, the entire procedure can be consistently performed in less than 3.5 hours (Jarrett et al, 2002), reflecting greater confidence in intracorporeal suturing and knot tying. Perioperative complication rates are low, ranging from 2% to 15.8%, demonstrating the safety of the laparoscopic procedure. Open conversion rates are also low, in the range of 0% to 5.5%. Furthermore, blood transfusion risks are low, being limited to anecdotal reports. Postoperative analgesic use is generally minimal. Mean length of hospital stay ranges from 2.6 to 4.5 days, and such average has decreased to 3.8 days in the series reported since 2000. With mean follow-up times of 14 to 26 months, the rates of surgical success (defined as durable clinical and/or radiographic success) reach the range of 87% to 99%, with the majority of contemporary series reporting success rates of greater than 95%. The safety and efficacy of laparoscopic pyeloplasty have also been demonstrated in the pediatric population including patients younger than 1 year (Metzelder et al, 2006).
Recently the transmesenteric approach to laparoscopic pyeloplasty has been analyzed (Romero et al, 2006). In contrast to the traditional retrocolic approach involving transperitoneal bowel mobilization described previously, a longitudinal incision is made in the colonic mesentery overlying the ureteropelvic junction region, followed by dissection of the ureter and renal pelvis. The laparoscopic pyeloplasty is then completed in the standard transperitoneal fashion. Recognition of the renal pelvis and/or ureter through the colonic mesentery is necessary in a successful transmesenteric procedure, which is performed more commonly in cases involving renal malrotation, horseshoe kidneys, left ureteropelvic junction obstruction, and younger patients (who presumably tend to have less visceral fat allowing better transmesenteric visualization of renal pelvis/ureter). Compared with the conventional retrocolic approach, the transmesenteric approach has been found to provide shorter operative time (by a mean of 22.5%) without significant increase in complication or patency outcomes.
Most failures from laparoscopic pyeloplasty occur in the first 2 years, although up to 30% of failed cases may occur after 2 years postoperatively (Madi et al, 2008). For the patients who fail laparoscopic pyeloplasty, open surgery has been used as a salvage procedure, with success rates of approximately 86% (Thomas et al, 2005). However, most cases can be well managed with endoscopic intervention such as endopyelotomy, with success rates of approximately 70% (Varkarakis et al, 2004).
More data on robotic-assisted laparoscopic pyeloplasty have emerged recently (Palese et al, 2005; Mufarrij et al, 2007; Schwentner et al, 2007; Yanke et al, 2008). Like the conventional laparoscopic studies, the overwhelming majority of the patients in these recent series had primary robotic-assisted laparoscopic pyeloplasties. The mean operative times are in the range of 108 to 244.8 minutes. Perioperative complication rates are low (<10.3%). Open conversion rates are also relatively low (0% to 6.8%). Postoperative analgesic use is generally minimal. Mean length of hospital stay is in the range of 2.2 to 2.8 days. With mean follow-up times of 11 to 39.1 months, the rates of surgical success (defined as durable clinical and/or radiographic success) are in the range of 94.7% to 100%. These results were similar to those from the historic laparoscopic series in the literature. The feasibility of the robotic approach has also been demonstrated in the pediatric patients (Atug et al, 2005; Lee et al, 2006). The additional reported benefits provided by the robot include better three-dimensional magnification, increased range of motion, ease of dissection, and suturing. However, the value of the robot in the setting of clinical pyeloplasty remains controversial and has been addressed by one recent study (Link et al, 2006). In this study comparing robotic and laparoscopic pyeloplasty in a prospective manner, the mean operative time and total room time for robotic cases were found to be significantly longer than laparoscopic cases by 19.5 and 39 minutes, respectively. Robotic cases were also found to be more costly than laparoscopic cases (2.7 times) due to longer operative time, increased cosumables costs, and depreciation of the robot system. In the hands of experienced laparoscopic surgeons, the use of the robot does not seem to provide significant clinical or cost advantage compared with conventional laparoscopic approach. In additional to cost, additional concerns for the robotic-assisted laparoscopic pyeloplasty include limited instrumentation and need for experienced bedside laparoscopic assistance (Peschel et al, 2004).
No prospective randomized trial has been successfully completed to compare laparoscopic with open pyeloplasty to date. The unwillingness of the patients to undergo randomization due to the different levels of perceived invasiveness appears to be the most significant barrier to completing such studies. In a retrospective study, Bauer and colleagues (1999) compared 42 laparoscopic pyeloplasties and 35 open pyeloplasties. With a minimum follow-up of 12 months for each of the patients, the two groups were found to be equivalent in pain relief (90% vs. 91%, respectively) and relief of obstruction (98% vs. 94%, respectively). In another retrospective study, Soulie and colleagues examined 26 laparoscopic pyeloplasties and 28 open pyeloplasties (Soulie et al, 2001). The two groups were found to be equivalent in mean operating time (165 vs. 145 minutes, respectively); mean blood loss (92 mL vs. 84 mL, respectively); perioperative complication rate (11.5% vs. 14.3%, respectively); mean hospital stay (4.5 days vs. 5.5 days, respectively); and radiologic success (89% vs. 89%, respectively). However, more laparoscopic patients were found to have returned to normal activity by postoperative day 15 (90% vs. 70%, respectively). In a third retrospective study, Klingler and colleagues (2003) compared 40 laparoscopic pyeloplasties with 15 open pyeloplasties. In this series the laparoscopic group was found to have lower mean postoperative visual analogue scale score (day 1, 3.5 vs. 5.4; day 5, 0.9 vs. 3.1) and shorter mean hospital stay (5.9 vs. 13.4 days). In a fourth retrospective study, Simforoosh and colleagues (2004) compared 37 laparoscopic pyeloplasties and 32 open pyeloplasties and found equivalent clinical and radiologic success rates between the two approaches (89% and 83.8% for laparoscopic group and 96.5% and 87% for open group, respectively) with a mean follow-up of 16.5 months (laparoscopic group) and 11.4 months (open group). Finally, in a fifth retrospective study, Calvert and colleagues (2008) examined the differences between 49 laparoscopic and 51 open pyeloplasty patients. Compared with open cases, laparoscopic cases were found to have significantly longer mean operating time (159 vs. 91 minutes) and significantly shorter mean time to normal diet (38 vs. 72 hours).
In the presence of the crossing vessel, dismembered pyeloplasty with transposition of the ureteropelvic junction anastomosis anterior to the crossing vessel has been the conventional teaching. Recently, an alternative approach has been reported to successfully manage ureteropelvic junction obstruction associated with the crossing vessel without the need for ureteral transection and reanastomosis. The technique involves laparoscopic mobilization of the entire ureteropelvic junction, proximal ureter, and renal pelvis from surrounding tissues without ureteral transection (Meng et al, 2003; Boylu et al, 2009). The crossing vessel, in addition, may be mobilized and transposed superiorly and fixed to the peripelvic tissues with metallic clips or sutures (Simforoosh et al, 2005; Gundeti et al, 2008; Masood et al, 2009). Such technique has been described to be most appropriate for the ureteropelvic junction obstruction cases involving normal-appearing ureter with peristalsis and unfavorable pelvic anatomy, in which ureteropelvic anasomosis cannot be achieved in a tension-free manner after ureteral transposition.
Primary ureteropelvic junction obstruction associated with renal anomalies such as horseshoe kidneys and pelvic kidneys have also been managed with laparoscopic pyeloplasty safely and successfully (Janetschek et al 1996; Hsu et al 2003; Bovie et al 2004). Furthermore, secondary ureteropelvic junction obstruction has similarly been managed with success. In a retrospective review, Sundaram and colleagues (2003) identified 36 cases of laparoscopic transperitoneal pyeloplasty for secondary ureteropelvic junction obstruction, mostly following failed retrograde or antegrade endopyelotomies. Mean operative time was 6.2 hours, longer than the reported times associated with primary ureteropelvic junction obstruction. Open conversion was necessary in one patient, and postoperative complication occurred in eight patients. With a mean follow-up of 21.8 months, the overall success rate of a greater than 50% decrease in pain, a patent ureteropelvic junction, and stable or improved function of the affected renal unit was 83% (30 of 36 patients). Shapiro and colleagues (2009) identified nine cases of laparoscopic transperitoneal pyeloplasty for secondary ureteropelvic junction obstruction after a failed open procedure. Mean operative time was 204 minutes. At a median follow-up of 66 months, 89% (eight of nine) patients had clinical and radiologic resolution of ureteropelvic junction obstruction, with stable renal function, pain-free status, and patent ureteropelvic junction.
Special Situations of Laparoscopic and Robotic-Assisted Laparoscopic Management of Ureteropelvic Junction Obstruction
Laparoscopic and Robotic-Assisted Laparoscopic Ureterocalicostomy
Ureterocalicostomy has been completed successfully via both laparoscopic and robotic-assisted laparoscopic approaches. Gill and colleagues (2004) performed laparoscopic ureterocalicostomy in two patients with ureteropelvic junction obstruction associated with small renal pelvis and dilated lower pole calix. In both patients a double-J ureteral stent was first placed into the ipsilateral ureter cystoscopically. With the patient in a 45- to 60-degree flank position, a transperitoneal approach using three or four ports was used to gain access to the ipsilateral renal unit laparoscopically. A circular rim of the tip of the thin lower pole renal parenchyma was identified and excised. The ureteropelvic junction was transected, followed by ligation of the renal pelvic opening. The ureter was spatulated laterally, and end-to-end ureterocaliceal anastomosis with mucosa-to-mucosa apposition over the preplaced double-J stent was performed with free-hand intracorporeal suturing and knot-tying techniques. The general reconstructive principles are identical to those of open ureterocalicostomy described previously including the need to achieve tension-free, water-tight, dependent drainage. Casale and colleagues (2008) reported successful robotic-assisted laparoscopic ureterocalicostomy in nine pediatric patients, following the identical reconstructive principles described earlier. Mean operative time was 168 minutes, and feasibility of the use of robot was well demonstrated. All patients were found to have no evidence of obstruction on diuretic radionuclide imaging at 12 months postoperatively.
Laparoscopic and Robotic-Assisted Pyeloplasty with Concomitant Pyelolithotomy
Presence of calculi in the setting of ureteropelvic junction obstruction can be managed laparoscopically with success. In a retrospective review, Ramakumar and colleagues (2002) reported 20 cases of laparoscopic pyeloplasty with concomitant extraction of renal stones through the pyelotomy site under laparoscopic guidance. In the series, extraction of the caliceal stones was assisted by the use of a flexible cystoscope introduced through a 10- to 12-mm port site. At a mean follow-up of 3 months, 90% of patients were stone free, and 90% patients had patent ureteropelvic junction radiographically. In another retrospective review, Stein and colleagues (2008) reported 15 cases of laparoscopic pyeloplasty with concomitant pyelolithotomy, involving the use of laparoscopic graspers, flexible cystoscopes, and/or laparoscopic irrigation. The overall stone-free rate was 80%. Robotic-assisted laparoscopic pyeloplasty with concomitant pyelolithotomy has also been demonstrated in eight patients recently, using the similar instruments including laparoscopic graspers (Atug et al, 2005). To complete the pyelolithotomy, one of the robotic arms was temporarily undocked to allow passage of a flexible nephroscope into the renal pelvis to gain visualization of the stones in the collecting system. In this small series, all patients were rendered stone free.
Laparoscopic Dismembered Tubularized Flap Pyeloplasty
Presence of a significant upper ureteral defect following the excision of ureteropelvic junction stricture may also be managed laparoscopically with success. Kaouk and colleagues (2002) described a case of laparoscopic pyeloplasty for secondary ureteropelvic junction obstruction, in which a 3-cm upper ureteral defect was found following excision of the long stricture. Using a four-port transperitoneal approach, a wide-base renal pelvic flap was created and tubularized to bridge the defect, using intracorporeal freehand suturing techniques. At a 2-month follow-up, excretory urography and diuretic renal scan confirmed a widely patent upper ureter.
Laparoscopic Calicovesicostomy
Presence of a large-capacity bladder in the setting of ureteropelvic junction obstruction associated with a low-lying obstructed renal unit can be managed successfully using an unconventional laparoscopic reconstructive strategy. Hsu and colleagues (2006) described a case of laparoscopic management of ureteropelvic junction obstruction involving a horseshoe kidney with a unilateral hydronephrotic yet functioning lower pole moiety, ipsilateral ureteral duplication with high bifurcation, and complex anomalous renal vasculature. Rather than performing tedious anatomic dissection and complex ureteral reconstruction in such scenario as required in conventional laparoscopic pyeloplasty, a nephrotomy was created at the most dependent portion of the hydronephrotic lower pole moiety and then laparoscopically anastomosed to the bladder dome vesicostomy using intracorporeal freehand suturing and knot-tying techniques. At the 4-month follow-up, patent calicovesicostomy was confirmed endoscopically and clinically.
Key Points: Laparoscopic and Robotic Intervention
Open Operative Intervention
General Surgical Principles
Several types of surgical incisions have been used for a pyeloplasty in the management of ureteropelvic junction (UPJ) obstruction. An anterior extraperitoneal approach is chosen by some because it allows surgical repair with minimal mobilization of the pelvis and proximal ureter. Alternatively, a posterior lumbotomy provides direct exposure to the UPJ and again allows repair with minimal mobilization of the surrounding structures. Like the anterior extraperitoneal approach, posterior lumbotomy is best suited to relatively thin patients without previous ipsilateral surgery. The authors’ personal preference for most patients undergoing primary surgical repair of UPJ obstruction is an extraperitoneal flank approach. This incision may be subcostal but is usually performed through the bed of the 12th rib or carried anteriorly off its tip. The extraperitoneal flank approach is advantageous in that it is familiar to all urologists and provides excellent exposure without regard to body habitus. In the presence of other renal anomalies associated with the UPJ, such as horseshoe or pelvic kidney, anterior extraperitoneal approaches are often preferable, although laparoscopic management may be considered in this setting.
Before the definitive surgical management, drainage of a kidney with UPJ obstruction is recommended only in select circumstances including infection associated with the obstruction or azotemia resulting from obstruction in a solitary kidney or bilateral disease. Procedural drainage may be of value in the uncommon scenario of severe, unrelenting pain requiring emergent relief of obstruction. For any of these situations, such drainage can be achieved by placement of an internal ureteral stent or a percutaneous nephrostomy tube.
The clinical indications for placement of stents or nephrostomy tubes intraoperatively remain controversial and vary among the urologists. For adults, the authors’ preference is for routine placement of a soft, inert, self-retaining internal ureteral stent, which is removed 4 to 6 weeks postoperatively. Such stents in adults can be easily removed in an outpatient office setting under local anesthesia. Routine use of internal ureteral stents offers several advantages, especially in the early postoperative period. Such practice appears to decrease the amount and length of time of urinary extravasation at the surgical repair site, thereby decreasing the risk of secondary fibrosis. Decreased urinary extravasation also allows earlier removal of external drains. For the uncomplicated pyeloplasty in adult patients, there appears to be no advantage to using both a nephrostomy tube and a stent because this may result in a prolonged hospital stay and an increased incidence of infection (Wollin et al, 1989). Instead, nephrostomy tubes may be reserved for complicated procedures such as those required for secondary UPJ obstruction or those associated with active inflammation. However, if a percutaneous nephrostomy tube had been placed preoperatively, it is generally left in place to allow proximal diversion and access for antegrade radiographic studies during the postoperative period.
Although the use of internal stents and nephrostomy tubes remains somewhat controversial, provision of external drainage from the site of surgical repair is absolutely necessary. Such external drainage may be achieved with a Penrose or closed suction drain placed near, but not on, the suture line and brought out through a separate stab incision. This practice helps to minimize the risk of urinoma formation leading to possible disruption of the suture line, scarring, or sepsis.
Historical Notes
The historical aspects of UPJ repair were previously examined by Kay in 1989 and by Schaeffer and Grayhack in 1986. The first reconstructive procedure was performed by Trendelenburg in 1886; however, the patient died of postoperative complications. In 1891 Kuster divided the ureter and reanastomosed it to the renal pelvis, thus apparently performing the first successful dismembered pyeloplasty. Kuster’s technique, however, was prone to recurrent stricture. In 1892 Fenzer applied the Heineke-Mikulicz principle to UPJ repair. This surgical technique involves transverse closure of a longitudinal incision. However, this technique can cause shortening of the suture line on one side, thus resulting in buckling or kinking of the UPJ with recurrent obstruction. In 1916 Schwyzer introduced the Y-V-pyeloplasty, which was subsequently modified by Foley in 1937. However, this technique was best applied to high ureteral insertions and was essentially unsuitable when the UPJ itself was already in a dependent position. Later on, flap techniques were developed that were more universally applicable including the spiral flap of Culp and DeWeerd (1951) and the vertical flap of Scardino and Prince (1953). Thompson and colleagues (1969) reported the use of a renal capsular flap for complex cases in which an adequate amount of renal pelvis is not available for repair.
In 1949 Nesbit followed the principle of Kuster’s dismembered procedure and further modified it by creating an elliptical anastomosis to decrease the likelihood of stricture formation at the site of repair. Also in 1949, Anderson and Hynes described their modifications of this dismembered technique that involved anastomosis of the spatulated ureter to a projection of the lower aspect of the pelvis after a redundant portion was excised. Use of healing by secondary intention was also investigated in the similar time period. The techniques of intubated ureterotomy were popularized by Davis in 1943, but they had been previously described by Fiori in 1905, Albarran in 1909, and Keyes in 1915.
Although a variety of techniques have been described for the management of UPJ obstruction, several basic principles must always be applied to maximize the success of surgical repair. For any procedure, the resultant anastomosis should be widely patent and completed in a watertight fashion without tension. In addition, the reconstructed UPJ should allow a funnel-shaped transition between the pelvis and the ureter that is in a position of dependent drainage.
Dismembered Pyeloplasty
Indications
At present, a dismembered pyeloplasty is preferred by most urologists in the surgical repair of UPJ obstruction because this procedure is almost universally applicable to the different clinical scenarios. This approach can be used regardless of whether the ureteral insertion is high on the pelvis or already dependent. It also permits reduction of a redundant pelvis or straightening of a tortuous proximal ureter. Furthermore, anterior or posterior transposition of the UPJ can be achieved when the obstruction is due to accessory or aberrant lower pole vessels. In addition, unlike the flap techniques, only a dismembered pyeloplasty allows complete excision of the anatomically or functionally abnormal UPJ itself. It is important to note that a dismembered pyeloplasty is not well suited to UPJ obstruction associated with lengthy or multiple proximal ureteral strictures or to patients in whom the UPJ obstruction is associated with a small, relatively inaccessible intrarenal pelvis.
Technique
Surgical exposure to the UPJ is achieved by first identifying the proximal ureter in the retroperitoneum. The proximal ureter is then dissected cephalad to the renal pelvis, leaving a large amount of periureteral tissue to preserve the ureteral blood supply. A marking stitch of fine suture is then placed on the lateral aspect of the proximal ureter, below the level of the obstruction, to assist proper orientation for the subsequent repair. In a similar fashion, the medial and lateral aspects of the dependent portion of the renal pelvis are marked with traction sutures (Fig. 41–16A). The UPJ tissue is excised, and the proximal ureter is then spatulated on its lateral aspect. The apex of this lateral, spatulated aspect of the proximal ureter is brought to the inferior border of the renal pelvis, while the medial side of the ureter is brought to the superior aspect (Fig. 41–16B). The anastomosis is then performed with fine interrupted or running absorbable sutures, placed full thickness through the ureteral and renal pelvic walls, in a watertight manner (Fig. 41–16C). As discussed earlier, the authors’ preference for adult patients is to routinely perform the anastomosis over an internal ureteral stent, which is left indwelling.
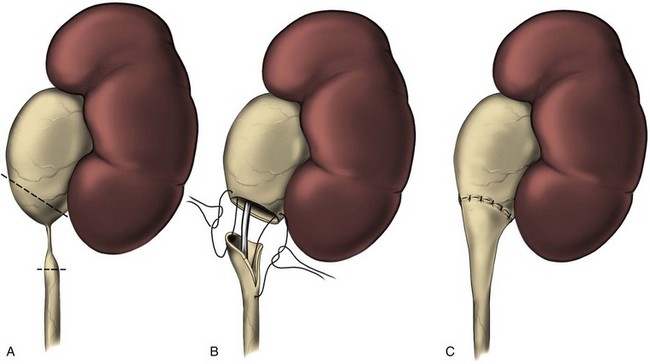
Figure 41–16 A, Traction sutures are placed on the medial and lateral aspects of the dependent portion of the renal pelvis in preparation for dismembered pyeloplasty. A traction suture is also placed on the lateral aspect of the proximal ureter, below the level of obstruction. This will help maintain proper orientation for the subsequent repair. B, Ureteropelvic junction is excised. The proximal ureter is spatulated on its lateral aspect. The apex of this lateral, spatulated aspect of the ureter is then brought to the inferior border of the pelvis while the medial side of the ureter is brought to the superior edge of the pelvis. C, Anastomosis is then performed with fine interrupted or running absorbable sutures placed full thickness through the ureteral and renal pelvis walls in a watertight fashion. In general, we prefer to leave an indwelling internal stent for adult patients. The stent is removed 4 to 6 weeks later.
If the renal pelvis is exceptionally redundant, a “reduction” pyeloplasty can be performed by excising the redundant portion of the pelvis (Fig. 41–17A-C). The cephalad aspect of the pelvis is then closed with running absorbable sutures down to the dependent portion, which will subsequently be anastomosed to the ureter. In the event that aberrant or accessory lower pole vessels are found in association with the UPJ obstruction, a dismembered pyeloplasty allows transposition of the UPJ in relation to these vessels (Fig. 41–18).
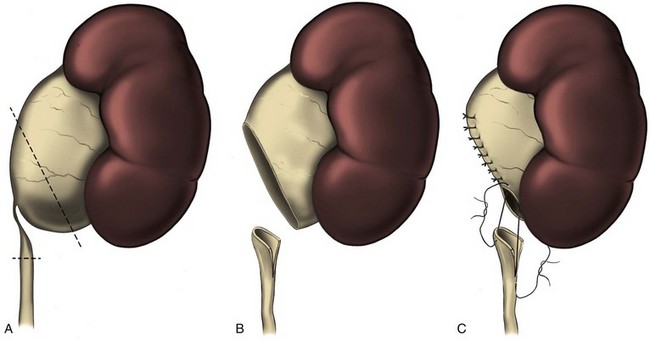
Figure 41–17 A, For large or redundant renal pelves, a reduction pyeloplasty is performed by excising the redundant portion between traction sutures. B, The cephalad aspect of the pelvis is then closed with running absorbable suture down to the dependent portion. C, The dependent aspect of the pelvis is then anastomosed to the proximal ureter.
Results
The overall success of open dismembered pyeloplasty has been favorable in the literature. In a retrospective review, Persky and colleagues (1977) noted that none of their 109 dismembered pyeloplasties for UPJ obstruction required subsequent nephrectomy. In another retrospective review involving 111 patients with UPJ obstruction undergoing open surgical repair over a 15-year-period, Clark and Malek (1987) found 95% success in resolution of clinical symptoms and 91% success in decompression of pelviocalyceal system on urography following one surgical repair. Of the 111 patients with open pyeloplasty, 95 (86%) patients underwent dismembered pyeloplasty. Examining the functional outcomes on the basis of split-function analysis from preoperative and postoperative renal scans, O’Reilly (1989) found that open Anderson-Hynes dismembered pyeloplasty arrests functional deterioration in almost every case and improves function significantly in the majority in 26 consecutive patients with UPJ obstruction.
Flap Procedures
Foley Y-V-Plasty
Indications
The Foley Y-V-plasty was originally designed for repair of a UPJ obstruction secondary to a high ureteral insertion. Like other flap techniques, however, its use has generally been replaced by the more versatile dismembered pyeloplasty. Like other flap techniques, the Foley Y-V-plasty is specifically contraindicated when transposition of lower pole vessels is necessary. In situations requiring concomitant reduction of redundant renal pelvis, this technique is also of little value.
Technique
In Foley Y-V-plasty, the renal pelvis and proximal ureter are first exposed, and a widely based triangular or V-shaped flap is outlined with methylene blue or fine stay sutures. The base of the V is positioned on the dependent, medial aspect of the ipsilateral renal pelvis and the apex at the UPJ. The incision from the apex of the flap (the stem of the Y) will then be performed along the lateral aspect of the proximal ureter. The surgical incision in the ureter should be long enough to completely traverse the area of stenosis and extend for several millimeters into the normal-caliber ureter (Fig. 41–19A). The renal pelvic flap and ureterotomy are then created. A fine scalpel blade is used for the initial pelvic incision, after which a Potts or a fine Metzenbaum scissors is used to complete the flap and ureterotomy (Fig. 41–19B). An internal ureteral stent is now placed and the repair performed over it. First, the apex of the pelvic flap is approximated to the apex (inferior aspect) of the ureterotomy incision using fine absorbable suture. The posterior walls are then approximated using fine interrupted or running suture (Fig. 41–19C). Interrupted technique is likely to minimize pursing or buckling of the suture line, as well as local tissue ischemia. Anastomosis of the anterior walls is then performed, thereby completing the surgical repair (Fig. 41–19D).
Figure 41–19 A, Foley Y-V-plasty is best applied to a ureteropelvic junction (UPJ) obstruction associated with a high insertion of the ureter. The flap is outlined with tissue marker or stay sutures. The base of the V is positioned on the dependent, medial aspect of the renal pelvis and the apex at the UPJ. The incision from the apex of the flap, which represents the stem of the Y, is then carried along the lateral aspect of the proximal ureter well into an area of normal caliber. B, The flap is developed with fine scissors. The apex of the pelvic flap is then brought to the most inferior aspect of the ureterotomy incision. C, The posterior walls are then approximated using interrupted or running fine absorbable suture. D, The anastomosis is completed with approximation of the anterior walls of the pelvic flap and ureterotomy.
Culp-DeWeerd Spiral Flap
Indications
The Culp-DeWeerd spiral flap is generally best suited for large, readily accessible extrarenal pelves in which the ureteral insertion is already in a dependent, oblique position. Although most of these patients are also good candidates for a standard or reduction dismembered pyeloplasty, the spiral flap may be of significant value when both UPJ obstruction and a relatively long segment of proximal ureteral narrowing or stricture occur in the same setting.
Technique
The spiral flap is first outlined with a broad base positioned obliquely on the dependent aspect of the renal pelvis. To maximize preservation of the flap blood supply, the base is placed in a position anatomically lateral to the UPJ, that is, between the ureteral insertion and the renal parenchyma. The pelvic flap itself may be spiraled posteriorly to anteriorly or vice versa. In either case, the anatomically medial line of incision (farthest from the parenchyma) is carried down the proximal ureter, completely traversing through the obstructed segment (Fig. 41–20A). Appropriate placement of the apex of the flap is determined by the length of flap needed. This, in turn, depends on the length of proximal ureter to be bridged. The longer the flap required, the farther away the apex will be from the base. However, to preserve vascular integrity of the flap, the ratio of flap length to width should not be greater than 3 : 1. In general, the outline of the flap should be made longer than what may initially be perceived as necessary because the flap will shrink once the pelvis is incised. If the flap is found to be too long, excess length can be reduced by trimming back the apex, thereby preserving its blood supply. Once the flap is created, the apex is rotated down to the most inferior aspect of the ureterotomy (Fig. 41–20B). The anastomosis with fine absorbable sutures is subsequently performed over an internal stent (Fig. 41–20C).
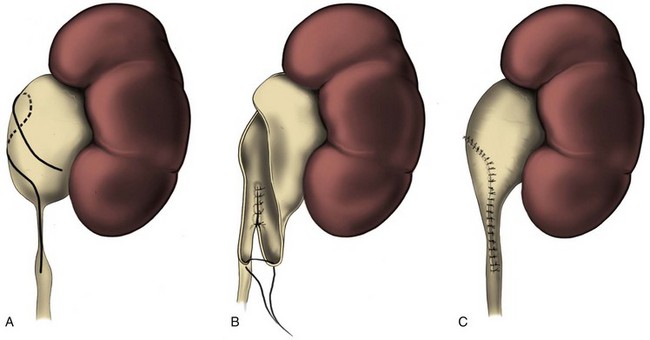
Figure 41–20 A, Spiral flap may be indicated for relatively long areas of proximal ureteral obstruction when the ureteropelvic junction (UPJ) is already in a dependent position. The spiral flap is outlined with the base situated obliquely on the dependent aspect of the renal pelvis. The base of the flap is positioned anatomically lateral to the UPJ, between the ureteral insertion and the renal parenchyma. The flap is spiraled posteriorly to anteriorly or vice versa. The anatomically medial line of incision is carried down completely through the obstructed proximal ureteral segment into normal-caliber ureter. The site of the apex for the flap is determined by the length of flap required to bridge the obstruction. The longer the segment of proximal ureteral obstruction, the farther away is the apex because this will make the flap longer. However, to preserve vascular integrity to the flap, the ratio of flap length-to-width should not exceed 3:1. B, Once the flap is developed, the apex is rotated down to the most inferior aspect of the ureterotomy. C, The anastomosis is then completed, usually over an internal stent, again using fine absorbable sutures.
Scardino-Prince Vertical Flap
Indications
The Scardino-Prince vertical flap technique generally has limited clinical application. It may be appropriately used only when a dependent UPJ is situated at the medial margin of a large, square (“box-shaped”) extrarenal pelvis (Fig. 41–21A). Its use in most instances has been replaced by a standard dismembered pyeloplasty, although the vertical flap may be preferable for relatively long areas of proximal ureteral narrowing. It is important to note that the vertical flap technique generally cannot produce as long a flap as the spiral flap.
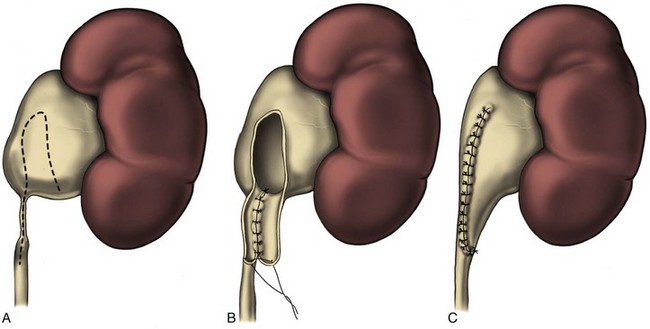
Figure 41–21 A, Vertical flap technique may be used when a dependent ureteropelvic junction (UPJ) is situated at the medial margin of a large, box-shaped extrarenal pelvis. In contrast to the spiral flap, the base of the vertical flap is situated more horizontally on the dependent aspect of the renal pelvis, between the UPJ and the renal parenchyma. The flap itself is formed by two straight incisions converging from the base vertically up to the apex on either the anterior or the posterior aspect of the renal pelvis. As for the spiral flap, the position of the apex determines the length of the flap, which should be a function of the length of proximal ureter to be bridged. The medial incision of the flap is carried down the proximal ureter completely through the strictured area into normal-caliber ureter. B, The apex of the flap is rotated down to the most inferior aspect of the ureterotomy. C, The flap is then closed by approximating the edges with interrupted or running fine absorbable sutures.
Technique
The Scardino-Prince vertical flap is similar to the spiral flap technique except that the base of the flap is positioned more horizontally on the dependent aspect of the renal pelvis, between the UPJ and the renal parenchyma. The flap itself is created by straight incisions converging from the base vertically to the apex on either the anterior or the posterior aspects of the renal pelvis. The site of the apex and the length of the flap are determined by the length of proximal ureter to be bridged. The medial incision is carried down the proximal ureter, completely traversing through the stenotic area and into normal-caliber ureter, using fine scissors (Fig. 41–21B). The apex of the flap is then rotated down and approximated to the most inferior aspect of the ureterotomy. Finally, the flap is closed with interrupted or running fine absorbable sutures (Fig. 41–21C).
Intubated Ureterotomy
Indications
The Davis intubated ureterotomy, which is rarely used today, was developed for surgical repair of lengthy or multiple ureteral strictures. If these strictures are found in association with UPJ obstruction, the intubated ureterotomy may be combined with any of the standard pyeloplasty procedures. However, in such situation, the intubated ureterotomy would be best combined with a spiral flap procedure. Compared with the vertical flap, the spiral flap can be made longer, which allows more of the strictured area to be bridged by a pelvic flap, thereby leaving a shorter area to rely on healing by “secondary intention.” In fact, in this specific clinical setting, any flap technique would be preferable to a dismembered repair, at least in regard to blood supply preservation of and subsequent healing.
Technique
A flap is outlined as described previously, with the ureterotomy to be made completely through the long, strictured area (Fig. 41–22A). The flap is then created, with minimal dissection of the ureter in order to preserve its blood supply. Unlike the uncomplicated pyeloplasties, these cases require routine nephrostomy tube drainage to prevent postoperative urinoma formation. Nephrostomy drainage in these cases also allows access for subsequent antegrade radiographic studies during the postoperative period.
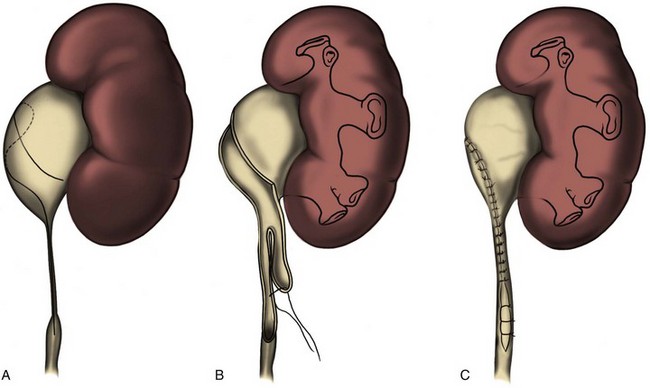
Figure 41–22 A, Intubated ureterotomy may be of value when a ureteropelvic junction obstruction is associated with extremely long or multiple ureteral strictures. A spiral flap is outlined and developed as described in Figure 41-20. The ureterotomy incision will be carried completely through the long strictured areas or through each of the multiple areas of stricture. B, The flap is developed, taking care to use minimal dissection of the ureter in order to preserve its blood supply. In contrast to uncomplicated repairs, nephrostomy tube drainage is used routinely. A self-retaining, soft, inert internal ureteral stent is then placed and positioned proximally in the renal pelvis or lower infundibulum and distally in the bladder. The apex of the flap is then brought as far down as possible over the stent on the ureterotomy, and the flap is closed with interrupted or running absorbable suture. C, The distal aspect of the ureterotomy is left open to heal secondarily by ureteral regeneration. A few fine absorbable sutures may be loosely placed to keep the sides of the ureter in apposition to the stent.
On the basis of the original description, the ureteral intubation is achieved with a stenting catheter that is placed across the stenotic area to the distal ureter or bladder. Proximally, it is brought out through the renal cortex alongside a nephrostomy tube. Currently, most urologists use a self-retaining, soft, inert, internal ureteral stent instead. The apex of the flap is brought over the stent as far down as possible on the ureterotomy, and the flap is closed with either interrupted or running absorbable suture (Fig. 41–22B). The distal aspect of the ureterotomy is then left open for secondary healing via ureteral regeneration (Fig. 41–22C).
An antegrade nephrostogram is usually obtained 6 weeks after the surgery. If there is no extravasation, the ureteral stent is removed cystoscopically and an antegrade radiographic study is repeated. When ureteral patency without extravasation is ensured with such study, the nephrostomy tube is clamped and subsequently removed.
Ureterocalycostomy
Indications
Ureterocalycostomy may be used as a primary reconstructive procedure whenever a UPJ obstruction or proximal ureteral stricture is associated with a relatively small intrarenal pelvis (Fig. 41–23A). When the UPJ is associated with rotational anomalies such as horseshoe kidney (Levitt et al, 1981), ureterocalycostomy may be useful to provide completely dependent drainage. Furthermore, ureterocalycostomy is a well-accepted salvage technique for the failed pyeloplasty (Ross et al, 1990).
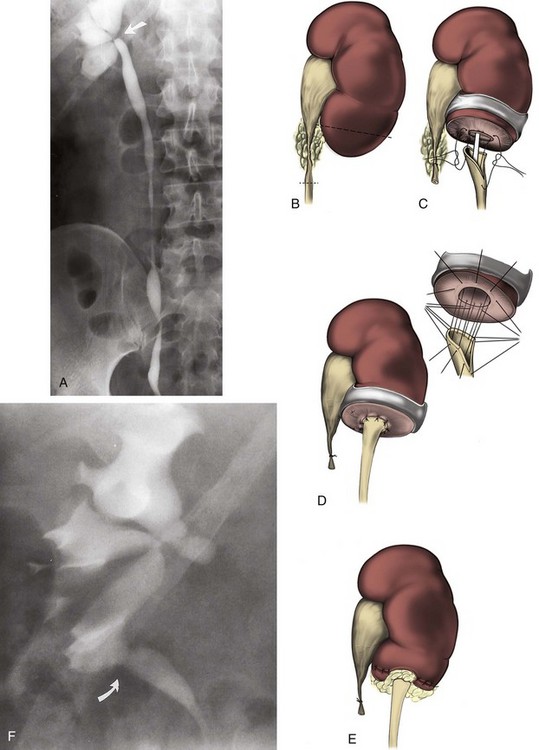
Figure 41–23 A, This patient complained of progressive right flank pain, and was found on this retrograde study to have a ureteropelvic junction obstruction (arrow) associated with a small intrarenal pelvis. This situation may be best managed with a ureterocalycostomy. B, The ureter is identified in the retroperitoneum and dissected proximally as far as possible. The kidney is mobilized as much as necessary to gain access to the lower pole and to subsequently perform the anastomosis without tension. A lower pole nephrectomy is performed, removing as much parenchyma as necessary to widely expose a dilated lower pole calyx. C, The proximal ureter is spatulated laterally. The anastomosis should subsequently be performed over an internal stent, and consideration should also be given to leaving a nephrostomy tube. The initial sutures are placed at the apex of the ureteral spatulation, and the lateral wall of the calyx with a second suture is placed 180 degrees from that. D, Anastomosis is then completed in an “open” fashion, placing each suture circumferentially (inset) but not securing them down until the anastomosis has been completed. E, Renal capsule is closed over the cut surface of the parenchyma whenever possible. However, the capsule should not be closed near the anastomosis itself because that may compromise the lumen by extrinsic compression. Instead, the anastomosis should be protected with a graft of perinephric fat or a peritoneal or omental flap. F, Intravenous urogram 2 months after right ureterocalycostomy reveals a widely patent ureterocalyceal anastomosis at the lower pole (arrow).
Technique
The ureter is first identified in the retroperitoneum and dissected proximally with a generous amount of periureteral tissue. For secondary procedures, however, extensive scarring may preclude adequate identification and dissection of the renal pelvis itself (Fig. 41–23B). The kidney is then mobilized to gain access to the lower pole. An important technical point in ureterocalycostomy is that the parenchyma overlying the lower pole calyx must be resected rather than simply incised because a simple nephrotomy may lead to a secondary stricture (Couvelaire et al, 1964).
The proximal ureter is first spatulated laterally, and the ureterocalyceal anastomosis is completed over an internal stent. Leaving an indwelling nephrostomy tube should also be considered in these cases. The first suture is placed at the apex of the ureteral spatulation and lateral wall of the calyx, and the second suture is placed 180 degrees apart. The remainder of the anastomosis is then performed using an interrupted “open” suture technique. That is, each suture placed is left untied until the final one is in place (Fig. 41–23C). This method seems to provide a more accurate anastomosis under direct vision. When the full set of circumferential sutures has been placed, the sutures are secured down together (Fig. 41–23D). The renal capsule is closed over the cut surface of the parenchyma if possible. However, such closure should not be close enough to the anastomosis itself to cause extrinsic compression on the anastomosis. Instead, the anastomosis should be covered with perinephric fat or a peritoneal or omental flap (Fig. 41–23E). A follow-up urogram is generally obtained at 1 month following the ureteral stent extraction (Fig. 41–23F).
“Salvage” Procedures
Failed open pyeloplasty is a challenging problem that is usually best managed initially using an endourologic approach. In some cases, such approach may not be applicable. In these cases, successful reconstruction can at times be achieved using one of the flap or dismembered techniques already described. The secondary open operative reconstruction may be significantly aided by the placement of a ureteral catheter to aid intraoperative identification and dissection of the ureter and renal pelvis. In these situations, there is often a relatively long length of proximal ureteral stenosis to repair, and wide mobilization of the kidney and ureter is generally a necessity. This helps to bridge the area of stenosis and allows a tension-free secondary pyeloplasty.
Several other options are available for these secondary and often complex repairs. These surgical alternatives include those generally available for any extensive ureteral problem such as ileoureteral replacement and autotransplantation with a Boari flap pyelovesicostomy. For cases in which function of the involved kidney is already significantly compromised and the contralateral kidney is normal, nephrectomy can be considered.
Postoperative Care and Management of Complications
In general, external drains are removed 24 to 48 hours after cessation of urinary drainage, and internal ureteral stents, if placed, are removed on an outpatient basis approximately 4 to 6 weeks after the surgery. If a nephrostomy tube is used, a nephrostogram is obtained no sooner than 7 to 10 days postoperatively, or even later for particularly complicated repairs. If nephrostogram demonstrates a patent anastomosis without obstruction or extravasation, the tube is clamped for 12 to 24 hours and removed if there is no flank pain, fever, or leakage around the tube.
If urinary drainage persists after 7 to 10 days in cases without the use of internal ureteral stent or recurs after the removal of external drain, retrograde studies with an attempt to place an internal stent should be considered. This generally resolves the problem immediately, and the internal stent is removed 1 month later. If an attempt to place an internal stent is unsuccessful, a percutaneous nephrostomy should be placed. If drainage persists following nephrostomy tube placement, an internal or internal/external stent should be placed in an antegrade fashion. At times, despite the appropriate use of stents, drains, and nephrostomy tubes, urinary extravasation may result in urinoma formation, which is best managed with direct percutaneous drainage of the fluid collection, using ultrasound or CT guidance. Follow-up of the functional result is typically achieved with a urogram or renogram obtained approximately 4 weeks postoperatively or 4 weeks after removal of any stent or nephrostomy tube. Earlier studies are indicated if the patient develops symptoms.
Key Points: Open Operative Intervention
Retrocaval Ureter
Etiology and Diagnosis
Retrocaval ureter is a rare congenital urologic anomaly. It occurs as a consequence of the persistence of the posterior cardinal veins during embryologic development (Considine, 1966). Its presence should be suspected with the finding of a characteristic S-shaped deformity on intravenous or retrograde pyelography (Fig. 41–24A). Today, a definitive diagnosis can be made noninvasively using three-dimensional CT imaging (Fig. 41–24B) (Pienkny et al, 1999). Procedural intervention is indicated in the presence of functionally significant obstruction leading to pain or renal function deterioration.
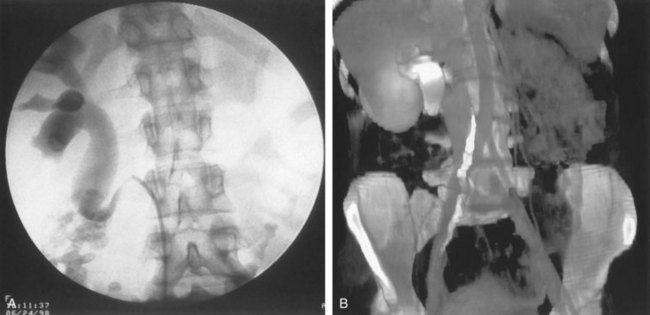
Figure 41–24 A, Retrograde pyelography in a patient with right-sided hydronephrosis. This study reveals a typical S-shaped deformity secondary to the ureter coursing laterally to medially posterior to the inferior vena cava. B, Three-dimensional spiral CT demonstrates the presence of a retrocaval ureter.
Operative Intervention
Open Surgical Management
The standard repair of retrocaval ureter is open surgical pyelopyelostomy. In this procedure, the ureter, dilated renal pelvis, and inferior vena cava are identified and dissected using the standard open surgical techniques. The dilated renal pelvis is then transected, following which the ureter is transposed to its normal anatomic position anterior to the vena cava (Fig. 41–25). Pyelopyelostomy is then performed circumferentially with absorbable sutures in a tension-free, water-tight manner. Surgical drain and internal ureteral stent are typically used.
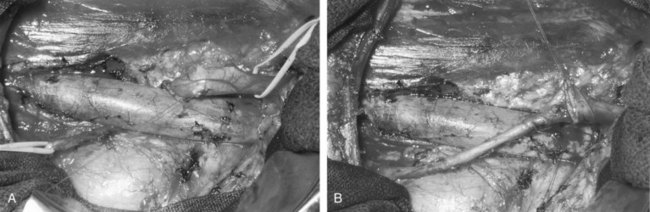
Figure 41–25 A, Intraoperative photograph of a patient with retrocaval ureter undergoing surgical repair via a retroperitoneal flank approach. Right side of the photo represents the cephalad direction. Note the dilated proximal right ureter passing behind the inferior vena cava. B, Right ureteropelvic anastomosis has been completed, following transection of the right renal pelvis and transposition of the ureter anterior to the inferior vena cava.
Laparoscopic Surgical Management
Retrocaval ureter has been managed successfully with the laparoscopic approach in the clinical setting as shown by a series of sporadic case reports in recent years (Baba et al, 1994; Matsuda et al, 1996; Polascik et al, 1998; Salomon et al, 1999; Gupta et al, 2001; Ramalingam et al, 2003). Either a transperitoneal or a retroperitoneal approach may be used laparoscopically. A double-J ureteral stent is first placed into the ipsilateral ureter cystoscopically. Following transperitoneal or retroperitoneal laparoscopic access, the ipsilateral ureter is identified and mobilized off the inferior vena cava. The ureter is then divided at the most distal segment of the dilated ureter. Redundant segment of dilated proximal ureter and stenotic segment of ureter are excised if present. The ureteral ends are positioned anterolateral to the vena cava, spatulated for 1.5 to 2 cm on opposite ends, and then anastomosed with absorbable sutures using intracorporeal suturing techniques over the stent. Tension-free, water-tight anastomosis is the objective. A surgical drain is then left in place before formal laparoscopic exit. The surgical drain is typically removed within a few days postoperatively, and the ureteral stent is typically removed 4 to 6 weeks postoperatively.
More recently, retrocaval ureter has been managed successfully with the robotic-assisted laparoscopic approach (Muffarrij et al, 2007; Hemal et al, 2008; Smith et al, 2009). A transperitoneal approach providing a large working space is typically used. The general principles of laparscopic ureteral dissection, division, transposition, and anastomosis are identical to those described in conventional laparoscopic approach. At least four different ports are involved, including three for the robot and one for the surgical assistant providing suction, irrigation, suture introduction, and retraction.
The overall clinical results of the laparoscopic repair with or without the use of robots in the literature have been favorable, providing minimal postoperative patient morbidity, short convalescence, and anastomotic patency on short-term radiographic follow-up.
Key Points: Retrocaval Ureter
Ureteral Stricture Disease
Etiology
Common etiologies of ureteral stricture formation include ischemia, surgical and nonsurgical trauma, periureteral fibrosis, malignancy, or congenital (Table 41–1). Proper evaluation and treatment of a ureteral stricture is essential to preserve renal function and rule out the presence of malignancy. Although the classic radiographic presentation of a transitional cell carcinoma of the ureter is a radiolucent filling defect within the lumen with the characteristic goblet sign, it may appear as a benign stricture. In addition, metastatic tumors such as cervical, prostate, ovarian, breast, and colon cancer may appear as a ureteral stricture (Lau et al, 1998). Although the incidence of ureteral strictures in the general population is unknown, it is clear that the presence of ureteral calculi and associated treatment of stones are risk factors. Roberts and colleagues (1998) evaluated 21 patients with impacted ureteral stones and found that impaction for more than 2 months’ duration was associated with a 24% incidence of stricture formation. Any ureteral instrumentation can lead to the development of a ureteral stricture. As advances in ureteroscopic technology have provided smaller, more flexible instruments with better optics, these endoscopic procedures have become less traumatic and are now associated with a long-term complication rate of less than 1% (Harmon et al, 1997). Other causes of benign ureteral strictures include radiation; abdominal aortic aneurysm; infections such as tuberculosis and schistosomiasis; endometriosis; and trauma including iatrogenic injury from previous abdominal or pelvic surgery or post–renal ablation injury (El Abd et al, 1996; Lacquet et al, 1997; Ramanathan et al, 1998; Oh et al, 2000; Johnson et al, 2004). Patients with presumed idiopathic ureteral strictures should be evaluated using CT scan to rule out the presence of an intrinsic ureteral malignancy or a lesion causing extrinsic compression.
Table 41–1 Etiology of Ureteral Stricture
Diagnostic Studies and Indications for Intervention
An intravenous pyelogram, retrograde pyelogram, or diagnostic ureteroscopy can define the location and length of the ureteral stricture reliably. CT can also delineate ureteral stricture disease, and CT urography can be used to help delineate the anatomy. Subsequent ureteroscopy with biopsy or barbotage should be performed in any patient for whom the etiology of the stricture is not certain. Diuretic renography will provide differential renal function and evaluate the renal unit for functional obstruction. It is critical to assess the renal unit for function before starting treatment because endourologic therapies generally require 25% function of the ipsilateral moiety to have reasonable success rates (Wolf et al, 1997). Once a ureteral stricture is diagnosed, indications for intervention include the need to rule out malignancy, compromised renal function, recurrent pyelonephritis, and pain associated with functional obstruction.
Endourologic Options for Intervention
Ureteral Stent Placement
Ureteral stent placement is effective acutely in treating most ureteral strictures, in particular intrinsic ureteral strictures. Wenzler and colleagues reported good success rates in treating intrinsic ureteral obstruction, with 88% success rates at 26 months (Wenzler et al, 2008). Although intrinsic ureteral strictures can be managed with endoscopic options, cases of extrinsic ureteral compression are better suited to percutaneous drainage or surgical management. In cases in which the patient is not a candidate for definitive repair, or has a poor prognosis, chronic stent placement with periodic stent changes may be considered. The use of chronic stent placement must be guarded, particularly when treating ureteral obstruction from extrinsic compression because in many cases, adequate drainage is short-lived (Chung et al, 2004; Docimo et al, 1989). Some urologists have placed two stents to maintain ureteral patency in cases in which a single stent did not provide adequate drainage (Yohannes et al, 2001). Recently Rosevear and colleagues (2007) reported 84% success rates in their series at 16 months using ureteral stents with 68% of the cases being for malignancy. The remainder included patients with retroperitoneal fibrosis and other benign extrinsic diseases. In 2006 Borin and colleagues reported the initial experience with a full-length metal stent for malignant, extrinsic ureteral obstruction (Borin, 2006). Liatsikos reported on 50 patients treated with the full metallic stent, and although concerns arose regarding stent exchange and encrustation, overall the study supported use of the stent at 12-month intervals (Liatsikos et al, 2010).
Retrograde Balloon Dilation
Retrograde dilatation of ureteral strictures has historically been part of the urologic armamentarium. The technique was rarely definitive and usually required repeated dilatations on a regular basis. In the early 1980s angiographic and vascular balloons were introduced into urologic practice, and the technique of balloon dilatation with temporary internal stenting became an accepted mode of treatment (Banner et al, 1983; Finnerty et al, 1984).
As for any patient with a ureteral stricture, the indications to intervene include functionally significant obstruction. Contraindications to this approach include active infection or a stricture longer than 2 cm because dilatation alone will rarely be successful in this setting. Moreover, any endoscopic technique is likely to fail with strictures greater than 2 cm (Fig. 41–26A-D).
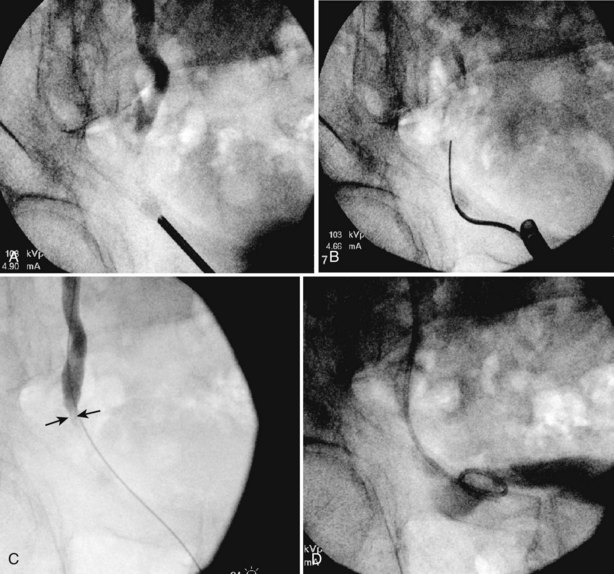
Figure 41–26 A, Antegrade and retrograde contrast demonstrating a distal ureteral stricture following prior traumatic ureteroscopic stone procedure. B, Image demonstrating complex guidewire access across this long and narrow stricture with retained proximal stones. C, The stricture is greater than 2 cm, and minimal contrast would pass through antegrade. D, Image following ureteral stent placement. This patient required ureteral reimplantation and stone removal.
A retrograde approach is indicated whenever access across the strictured area is easily accomplished using transurethral techniques. The procedure generally begins with a retrograde pyelogram performed under fluoroscopic control to precisely delineate the site and length of stricture. A floppy-tipped guidewire is passed in a retrograde fashion across the strictured area and coiled proximally in the pyelocalyceal system. This is most easily accomplished by passing an open-end catheter up to the level of the stricture to use as a guide for the hydrophilic or floppy-tipped wire. Passage of the open-end catheter through the strictured area over the wire will then aid subsequent passage of a balloon catheter. Techniques for bypassing difficult areas of obstruction have been described in detail (Mata et al, 1994).
At this point, the open-end catheter is withdrawn and replaced with a high-pressure, 4-cm-long, 5- to 8-mm balloon. Under fluoroscopic control, the balloon catheter is positioned across the strictured area with proper position ensured by visualization of radiopaque markers at the tips of the balloon. Balloon inflation is then begun, and a waist will be visualized at the strictured area, which will disappear with progressive balloon inflation (Fig. 41–27). After 10 minutes of tamponade, the balloon is deflated and withdrawn. A guidewire is still in place, and this is used to pass an internal stent, which is left indwelling for 2 to 4 weeks. Follow-up diuretic renography is generally performed approximately 1 month after stent extraction and at 6- to 12-month intervals thereafter.
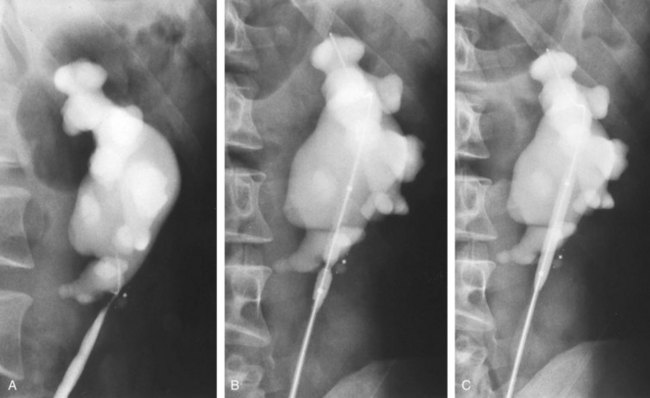
Figure 41–27 A, Retrograde study confirms a short stricture at the level of the ureteropelvic junction in this patient with a horseshoe kidney referred after failed ureteroscopic management of a ureteral calculus impacted at that level. B, The stricture has been traversed with a guidewire, over which a high-pressure balloon has been passed. A waist is evident at the level of the stricture during initial balloon inflation. C, Balloon inflation and stricture dilatation are complete with disappearance of the waist.
Occasionally, access across the involved area cannot be obtained using fluoroscopic control alone. In such cases direct ureteroscopic visualization can aid initial passage of the guidewire, and the procedure can be continued as described. Alternatively, a low-profile balloon can be passed through the ureteroscope and the stricture dilated under direct vision.
Antegrade Balloon Dilation
At times, retrograde access across a strictured area is impossible. In such cases, access can be obtained using an antegrade approach and fluoroscopic control (Mitty et al, 1983; Banner and Pollack, 1984), with or without direct antegrade ureteroscopic visualization (De Jonge et al, 1986). Percutaneous nephrostomy drainage is established, and, in cases associated with infection or compromised renal function, percutaneous drainage alone is instituted to allow resolution of infection and return to baseline renal function. Once that is accomplished, the percutaneous tract is used for access for a fluoroscopically or ureteroscopically guided approach. The procedure is then analogous to a retrograde approach. Under fluoroscopic guidance, an antegrade contrast agent study is used to definitively define the site and length of the stricture. A floppy-tipped guidewire or glidewire is passed antegrade across the level of obstruction, following which a balloon catheter is passed, and the balloon progressively inflated until the waist disappears. The balloon catheter is withdrawn over a wire and replaced with an internal stent, and a nephrostomy tube is also left indwelling. A follow-up nephrostogram is obtained within 24 to 48 hours to ensure proper positioning of a functional internal stent, and, at that time, the nephrostomy tube can be removed. Alternatively, access can be maintained by the use of an internal/external stent, which can be capped to allow internal drainage.
Results
Initial reports of retrograde and antegrade balloon dilatation of ureteral strictures suggested that results were better when the stricture was anastomotic and of relatively short duration and length (King et al, 1984b; Chang et al, 1987; Netto et al, 1990). Goldfischer and Gerber (1997) reviewed the literature in regard to results of balloon dilatation of ureteral strictures and found reported success rates ranging from 50% to 76%. In that review, the best results were obtained in patients with iatrogenic, nonanastomotic strictures such as those following ureteroscopic instrumentation. In that setting, a success rate of 85% was achieved compared with a rate of 50% for anastomotic strictures. Alternatively, Ravery and colleagues found a 40% success rate using retrograde balloon dilation in treating inflammatory ureteral strictures at 16 months’ follow-up (Ravery et al, 1998). Richter and colleagues (2000) reviewed their results with balloon dilatation in 114 patients with a minimum 2-year follow-up. As in other series, balloon dilatation was more successful for patients with relatively short strictures. In addition, these authors noted the significance of an intact vascular supply on the success of this procedure. One series of transplant ureteral strictures demonstrated 79% success at 29 months using percutaneous balloon dilation in 14 transplant patients. Notably, these are short, anastomotic strictures in patients on immunosuppression (Voegeli et al, 1988). For longer ureteral strictures and those associated with compromised vascular supply, an incisional approach (endoureterotomy) was recommended as a more successful endourologic alternative to balloon dilatation. Of note, in experimental models, balloon dilation created longitudinal incisions similar to endoureterotomy, possibly explaining some of the success seen using balloon dilation in ureteral strictures (Nakada et al, 1996).
Endoureterotomy
Endoluminal ureteral incision is a logical extension of balloon dilatation for “minimally invasive” management of ureteral strictures. As for balloon dilatation, access to and across the strictured area can be obtained in a retrograde or antegrade fashion, although a retrograde approach is preferable whenever possible because it is less invasive. The procedure is performed under direct vision using ureteroscopic control, or it can be guided fluoroscopically using the hot wire cutting balloon catheter. Generally, radiographic follow-up using diuretic renography is recommended for up to 3 years to detect most late failures (Wolf et al, 1997).
Retrograde Ureteroscopic Approach
A retrograde study is performed under fluoroscopic control at the outset of the procedure. Whenever possible, a floppy-tipped guidewire or hydrophilic glidewire is passed across the level of obstruction, as outlined earlier. If a wire cannot be passed across the strictured area using fluoroscopic control alone, the flexible ureteroscope is passed to the level of obstruction and the guidewire advanced through the ureteroscope across the involved area under direct vision. The ureteroscope is then withdrawn, but a safety wire is always left in place across the stricture. The ureteroscope is then reintroduced and passed alongside the guidewire to the level of obstruction.
The position for the endoureterotomy incision is chosen as a function of the level of the ureter involved. In general, lower ureteral strictures are incised in an anteromedial direction, taking care to stay away from the iliac vessels. In contrast, upper ureteral strictures are incised laterally or posterolaterally, again away from the great vessels (Meretyk et al, 1992) (Fig. 41–28).
The ureterotomy incision itself can be performed using a cold knife (Schneider et al, 1991; Yamada et al, 1995), a cutting electrode (Conlin et al, 1996), or a holmium laser (Fig. 41–29). Today, the availability of small-caliber laser fibers and actively deflecting flexible ureteroscopes make the holmium laser a common approach to endoscopic incision. In all cases, the incision is made from the ureteral lumen out to periureteral fat in a full-thickness fashion. Proximally and distally, the endoureterotomy should encompass 2 to 3 mm of normal ureteral tissue. In certain instances, the stricture must be balloon dilated to gain access across the stricture. Similarly, the strictures may be balloon dilated after endoincision, to enlarge the incision. Once the endoureterotomy incision is complete, the remaining guidewire is used to pass an internal stent. In general, the larger-diameter stents should be considered because larger stents (12-Fr) have been associated with improved results in some series (Hwang et al, 1996; Wolf et al, 1997). Similarly, Wolf and colleagues (1997) found benefit in the injection of triamcinolone ureteroscopically following endoureterotomy. Steroids and other biologic response modifiers may have a role in the future in managing select strictures.
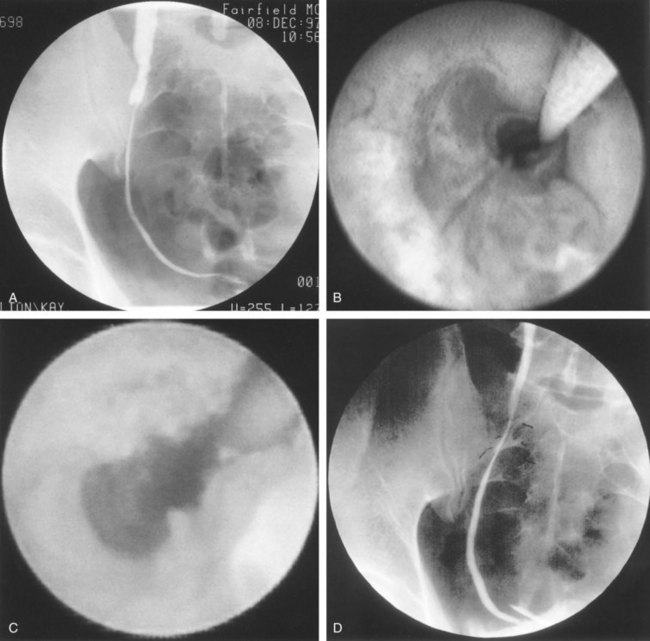
Figure 41–29 A, Antegrade study confirms hydronephrosis at the level of the iliac ureter in this patient referred for management after a complicated open ureterolithotomy. B, Ureteroscopic view confirms a tight ureteral stricture. C, Ureteral lumen is widely patent after holmium laser endoureterotomy. D, Follow-up ureterogram confirms resolution of the stricture.
Results
The success of holmium laser endoureterotomy ranges from 66% to 83% in series of greater than 10 patients with greater than 12 months’ follow-up (Lane et al, 2006; Hibi et al, 2007; Gdor et al, 2008a). There is early evidence that strictures related to stone impaction and prior stone treatment may have lower success rates (56% in one series) than typical benign strictures (Gdor et al, 2008b). Of note, as the use of ureteroscopy and laser lithotripsy continues to grow, more strictures involving impacted stones may be encountered, and this may become a growing clinical problem. Gdor and colleagues also reported 67% success in treating transplant ureteral strictures using the holmium laser at 58 months’ follow-up (Gdor et al, 2008a). At this time the current available reports and the familiarity of ureteroscopy, coupled with relative availability of the holmium laser, make retrograde laser endoureterotomy an attractive initial management strategy for short ureteral strictures.
Cautery Wire Balloon Incision
Cautery wire balloon incision has been used for ureteral strictures in the same manner as it has for UPJ obstruction. The procedure requires safety wire access across the strictured area and is performed under fluoroscopic control. The cautery wire balloon is passed in a retrograde or antegrade fashion and positioned across the stricture using radiopaque markers. In the proximal ureter, the incision should be posterolateral, whereas in the distal ureter, an anteromedial incision is used. Fluoroscopically guided cautery wire balloon incision should not be used in close proximity to the great vessels such as at the iliac level of the ureter. As for any form of endourologic management, success using this technique is dependent on the length and vascularity of the involved segment (Chandhoke et al, 1993b; Cohen et al, 1996; Wolf et al, 1997).
Antegrade Approach
When direct visual ureteroscopic access to the strictured area cannot be accomplished in a retrograde fashion, an antegrade approach may be used. Nephrostomy tube drainage is instituted, and any associated infection or compromised renal function is allowed to resolve before definitive incision. The percutaneous tract is dilated to a size large enough to allow a working sheath through which a flexible ureteroscope is passed. The procedure is then performed in a fashion analogous to a retrograde approach. A safety wire should be in place at all times alongside the ureteroscope, across the obstructed area and coiled distally in the bladder.
Combined Retrograde/Antegrade Approach
Rarely, a ureteral stricture is associated with an area of complete ureteral obliteration across which a wire cannot be passed to allow subsequent balloon dilatation or ureteroscopic endoureterotomy. In such cases a combined retrograde/antegrade approach has been described (Cardella et al, 1985; Conlin et al, 1996; Beaghler et al, 1997; Knowles et al, 2001). The obstructed area is defined radiographically with a simultaneous antegrade and retrograde pyelogram. Ureteroscopes are passed simultaneously in both a retrograde and an antegrade manner, and the two opposing ureteral ends are localized under fluoroscopic guidance. A working guidewire is then passed from one end of the ureter, through and through to the other lumen, using a combination of fluoroscopic and direct visual control. For completely obliterated ureteral segments, this is most easily accomplished using the stiff end of a guidewire passed through a semirigid ureteroscope via the retrograde approach, although when a semirigid ureteroscope cannot be placed, a flexible ureteroscope or even an open-end ureteral catheter can be used to stabilize the wire from above or below. A “cut to the light” technique can be helpful in this setting. The ureteral segments are aligned as closely as possible under endoscopic and fluoroscopic guidance, and the light source to one of the ureteroscopes is turned off. The light from the opposite ureteroscope is then used to aid incisional restoration of urinary continuity. The strictured area is then recannulated using the stiff end of a guidewire, a small electrocautery electrode, or holmium laser. Once through-and-through control is obtained with a guidewire, a stent is passed and left in place for 8 to 10 weeks. As with other endourologic approaches to ureteral strictures, success rates are inversely related to the length of the strictured area. Although success rates may be uncertain, internalization of urinary flow, even when dependent on long-term stent placement, can be a quality-of-life advantage for certain high-risk patients. Knowles and colleagues reported a 90% patency rate in 10 patients treating obliterated distal ureteral segments using cautery wire balloon incision at 36 months’ follow-up, 3 of whom required the combined approach (Knowles et al, 2001).
Key Points: Endourologic Management of Ureteral Strictures
Surgical Repair
Before any surgical repair, it is essential to conduct careful evaluation of the nature, location, and length of the ureteral stricture. Preoperative assessment typically includes an intravenous pyelogram (or antegrade nephrostogram) and a retrograde pyelogram if indicated. Other studies such as a nuclear medicine renogram to assess renal function and ureteroscopy, ureteral barbotage, or brushing to rule out carcinoma should be individualized. On the basis of such information, the appropriate surgical procedure can then be planned for the patient (Table 41–2).
Table 41–2 Bridging Various Ureteral Defect Lengths with Different Reconstructive Surgical Techniques
| TECHNIQUE | URETERAL DEFECT LENGTH (cm) |
|---|---|
| Ureteroureterostomy | 2-3 |
| Ureteroneocystostomy | 4-5 |
| Psoas hitch | 6-10 |
| Boari flap | 12-15 |
| Renal descensus | 5-8 |
Open Ureteroureterostomy
A short defect involving the upper ureter or midureter, either in the form of stricture or as a consequence of recent injury, is most appropriate for ureteroureterostomy. On the other hand, a lower ureteral stricture is usually best managed by ureteroneocystostomy with or without a psoas hitch or Boari flap. In the transplant setting, a donor ureteral stricture may be managed by a ureteroureterostomy to a healthy, native ureter. Because tension on the anastomosis almost always leads to stricture formation, only short defects should be managed by end-to-end ureteroureterostomy. Determination of whether or not enough ureteral mobility can be achieved to allow tension-free ureteroureterostomy usually cannot be made until the time of surgery.
The choice of surgical incision depends on the level of the ureteral stricture. A flank incision is appropriate for the upper ureter. A Gibson or a lower midline incision is suitable for the middle and lower ureter. If the patient has sustained an iatrogenic ureteral injury from a previous surgery performed through a Pfannenstiel incision, the same incision may be used for the ureteral reconstruction. In such situation, proximal ureteral dissection may be difficult through the Pfannenstiel incision, requiring cephalad extension of the lateral portion of the incision in a “hockey stick” fashion. Extraperitoneal dissection is usually performed except in cases of transperitoneal surgical ureteral injury.
Following surgical incision, the retroperitoneal space is developed as the peritoneum is mobilized and retracted medially. Frequently, the ureter can be easily identified as it crosses the iliac vessels. A Penrose drain or vessel loop may be placed around the ureter to assist its atraumatic handling. Direct handling of the ureter with forceps should be minimized. Care should be taken to preserve its adventitia, which loosely attaches the blood supply to the ureter.
During ureteral dissection and mobilization, enough mobility must be achieved to avoid tension after the excision of the diseased ureter. With a gunshot injury, devitalized tissue and an adjacent segment of normal-appearing ureter should be excised to eliminate late ischemia and stricture formation from the blast effect. Once both ends of the ureter have been adequately trimmed to healthy areas, mobilized, and correctly oriented, they are spatulated for approximately 5 to 6 mm. Spatulation is performed for both ureteral segments at 180 degrees apart. If a grossly dilated ureter is involved, it may be transected obliquely and not spatulated in order to match the circumference of the nondilated segment. A fine, absorbable suture is placed in the corner of one ureteral segment and the apex of the other, and the two ends of the suture are tied outside the ureteral lumen. The opposite corner and apex are similarly sutured and approximated. The anastomosis may then be completed by running these two sutures continuously and tying them to each other or in an interrupted fashion (Fig. 41–30). A double-J ureteral stent should be placed before completion of the anastomotic closure. Observation of reflux of methylene blue irrigant from the bladder to the ureterotomy can be used to verify the appropriate placement of the distal stent in the bladder. Retroperitoneal fat or omentum may be used to cover the anastomosis. A surgical drain is placed, and a Foley catheter is generally left indwelling for 1 to 2 days. The surgical drain may be removed if there is minimal output for 24 to 48 hours. If the surgical procedure is not performed entirely in a retroperitoneal manner, it is important to determine the nature of the fluid from the surgical drain, which can be achieved by checking the creatinine level of the fluid. If there is no urinary extravasation, the drain can then be removed. The double-J ureteral stent is removed endoscopically, usually 4 to 6 weeks postoperatively.
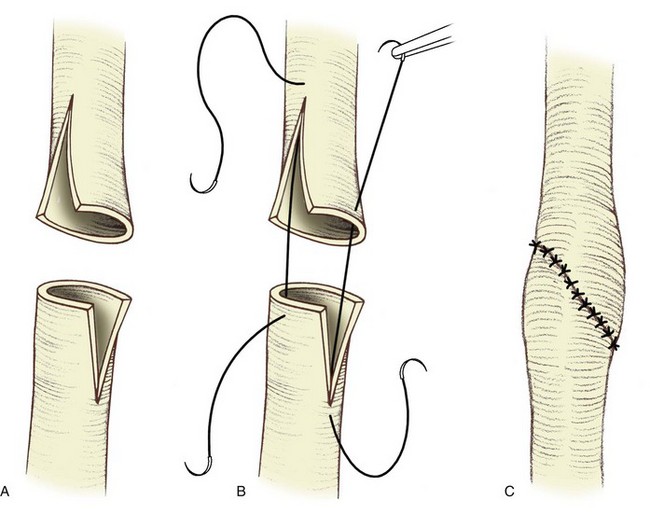
Figure 41–30 A, Spatulated ureteral ends. B, Placement of sutures. C, End-to-end ureteroureterostomy.
The success rate for a tension-free, watertight ureteroureterostomy is high—more than 90% (Carlton et al, 1969; Guiter et al, 1985). If a urinary fistula is suspected, a plain abdominal radiograph should first be obtained to verify the position of the double-J stent. The proximity of a drain to the anastomosis should also be checked because it may exacerbate a leak. Suction should be stopped if a suction drain device is used because straight drainage may assist closure of the ureteral leakage site. Reflux from voiding or bladder spasms may also contribute to prolonged urinary extravasation, a problem that can be managed by Foley catheter drainage and anticholinergics. Prolonged urinary leakage from the anastomosis may require the placement of a nephrostomy tube for proximal urinary diversion.
Laparoscopic Ureteroureterostomy
A laparoscopic approach may be offered to patients with ureteral stricture disease. Nezhat and colleagues (1992) first reported laparoscopic management of an obstructed ureter due to endometriosis. In this case, ureteroureterostomy was performed laparoscopically over a ureteral stent following resection of the obstructed ureteral site. In their more recent retrospective review involving eight laparoscopic ureteroureterostomy patients, seven were found to have patent anastomosis with relatively short follow-up, ranging from 2 to 6 months (Nezhat et al, 1998). More recently, the robotic-assisted approach has been applied to laparoscopic ureteroureterostomy in a small number of patients (Mufarrij et al, 2007; Passerotti et al, 2008). The overall clinical experience in laparoscopic ureteroureterostomy is limited worldwide. However, in the hands of the experienced laparoscopic surgeons, it appears to be a viable minimally invasive approach applicable to almost any patient with a relatively short area of obstruction.
Open Ureteroneocystostomy
Ureteroneocystostomy to manage vesicoureteral reflux is covered elsewhere in the text. Ureteroneocystostomy without a psoas hitch or Boari flap in an adult is appropriate for injury or obstruction affecting the distal 3 to 4 cm of the ureter. A lower midline, Pfannenstiel, or Gibson incision may be used, and the extraperitoneal approach is generally more preferable. Following surgical incision, the ureter is usually identified as it crosses the iliac vessels, dissected distally, and transected at the level of the obstruction. Following adequate proximal ureteral mobilization, direct ureteroneocystostomy is performed only if a tension-free anastomosis is possible. Otherwise, a psoas hitch or Boari flap should be used as an adjunct. A direct, nontunneled anastomosis may be performed if postoperative reflux is acceptable. Otherwise, a submucosal tunnel is created for antireflux anastomosis. A double-J stent and surgical drain are used as described for ureteroureterostomy described earlier.
The issue of reluxing versus antirefluxing anastomosis in ureteroneocystostomy in adults has been examined previously. In a retrospective review of adult patients with ureteroneocystostomy, no significant difference in the preservation of renal function or risk of stenosis was identified in the refluxing versus antirefluxing procedures (Stefanovic et al, 1991). However, it is unclear if a nonrefluxing anastomosis decreases the risk of pyelonephritis in an adult patient.
Laparoscopic Ureteroneocystostomy
Successful laparoscopic application to ureteroneocystostomy has been reported by several investigators (Ehrlich et al, 1993; Reddy et al, 1994; Yohannes et al, 2001). Successful use of the robot in such clinical setting has also been reported (Mufarrij et al, 2007; Laungani et al, 2008; Williams et al, 2009). In the management of distal ureteral stricture, laparoscopic ureteroneocystostomy is usually performed transperitoneally incorporating intracorporeal suturing techniques. Large working space provided by the transperitoneal approach is especially advantageous in the setting of robotic involvement. Ureteral stenting is typically used postoperatively as in open surgery. The overall clinical experience for laparoscopic management of distal ureteral strictures is limited in the literature. However, the clinical outcomes have been reported to be favorable to and comparable with those of open surgical data while providing minimal postoperative morbidity, as in many other laparoscopic urologic procedures.
Open Psoas Hitch
The psoas hitch is an effective method to bridge a defect of the lower third of the ureter. However, a ureteral defect extending proximal to the pelvic brim usually requires more than a psoas hitch alone. Indications include distal ureteral stricture, injury, and failed ureteroneocystostomy (Prout and Koontz, 1970; Ehrlich et al, 1978; Rodo Salas et al, 1991). A psoas hitch may also be used in conjunction with other maneuvers such as a transureteroureterostomy (TUU) in more complicated urinary tract reconstruction. Generally, a small, contracted bladder with limited mobility is considered as a contraindication. In addition to the preoperative radiographic and endoscopic evaluation described previously, urodynamic studies may provide information regarding detrusor capacity and compliance before the surgery. Bladder outlet obstruction or neurogenic dysfunction, if present, needs to be treated preoperatively.
To gain access to the distal ureter, a Pfannenstiel or lower midline incision is usually used. Extraperitoneal approach is preferred, if possible. In such scenario, the space of Retzius is developed and the bladder mobilized by freeing its peritoneal attachments and dividing the vas deferens or round ligament. With traction, the ipsilateral dome of the bladder should be able to reach the level proximal to the iliac vessels. Additional mobility can be achieved by dividing the contralateral superior vesical artery. The ipsilateral ureter is identified as it crosses the iliac vessels, mobilized, and divided just above the diseased segment. An anterior cystotomy, generally created in a vertical or oblique fashion, is frequently made to assist manual displacement of the bladder toward the ipsilateral ureter. The ureter is delivered into the lumen of the bladder at the ipsilateral superolateral aspect of the dome, followed by the tension-free anastomosis with or without a submucosal tunnel. The ipsilateral bladder dome is secured to the psoas minor tendon or the psoas major muscle using several absorbable sutures. Care should be taken to avoid injury to the genitofemoral nerve and the femoral nerve in the vicinity when placing these sutures. Alternatively, psoas fixation may be performed before ureteroneocystostomy. A double-J stent is used usually, followed by closure of cystotomy with absorbable sutures (Fig. 41–31).
Figure 41–31 A, In psoas hitch, an anterior cystotomy is performed following bladder mobilization. B, The bladder dome is fixed to the ipsilateral psoas tendon, and the ureteral reimplantation is completed in a tension-free manner.
Relative to simple ureteroneocystostomy, psoas hitch can provide an additional 5 cm of length. Relative to Boari flap, the advantages of psoas hitch include increased technical simplicity and decreased risk for vascular compromise and voiding difficulties. The success rate of ureteroneocystostomy with a psoas hitch is greater than 85% in both adults and children on the basis of recent reports (Mathews and Marshall, 1997; Ahn and Loughlin, 2001). Complications occur uncommonly but have included urinary fistula, ureteral obstruction, bowel injury, iliac vein injury, and urosepsis (Fig. 41–32).
Laparoscopic Psoas Hitch
Ureteroneocystostomy with psoas hitch has been performed laparoscopically with success (Nezhat et al, 2004). Successful robotic-assisted application has also been reported (Mufarrij et al, 2007; Patil et al, 2008; Schimpf and Wagner, 2009). Preoperative ureteral stenting is generally performed, and the procedure is typically completed via the transperitoneal approach. Overall, the clinical experience with such procedure is quite limited in the literature. However, on the basis of short-term and intermediate-term follow-up data to date, the clinical outcomes appear to be satisfactory and equivalent in experienced hands.
Open Boari Flap
When the diseased ureteral segment is too long or when ureteral mobility is too limited to perform a tension-free ureteroureterostomy, a Boari flap may be a useful alternative. Boari first described the use of this technique in the canine model in 1894. A Boari flap can be constructed to bridge a 10- to 15-cm ureteral defect, and a spiraled bladder flap can reach the renal pelvis in some circumstances, especially on the right side. As in a psoas hitch, evaluation of bladder function should be performed preoperatively in addition to the ureteral evaluation. Bladder outlet obstruction and neurogenic dysfunction, if present, should be addressed preoperatively. A small bladder capacity is likely to be associated with difficult or inadequate Boari flap creation, warranting consideration of alternative methods in the preoperative surgical planning.
In Boari flap, a Pfannenstiel incision may be used at the time of surgery, although a midline incision is more preferable and allows easier access to the upper ureter. The bladder is mobilized from its peritoneal attachments, and the umbilical ligaments are divided. The contralateral bladder pedicle is divided and ligated, allowing greater mobility toward the ipsilateral ureter, and the ipsilateral bladder pedicle including the superior vesical artery is preserved. The affected ureter is carefully mobilized, with care being taken to preserve its blood supply. The diseased segment is then excised. Following the identification of the ipsilateral superior vesical artery or one of its branches, a posterolateral bladder flap is outlined on the basis of this vascular supply. The flap continues obliquely across the anterior bladder wall, with the base of the flap being at least 4 cm in width and the tip of the flap being at least 3 cm in width. The flap length should equal the estimated ureteral defect plus an addition of 3 to 4 cm if a nonrefluxing anastomosis is planned. Furthermore, the ratio of flap length to base width should not be greater than 3 : 1 to help minimize flap ischemia.
Following bladder flap creation, the distal end of the flap is pexed to the psoas minor tendon or psoas major muscle with several absorbable sutures. The ureter is delivered through a small opening created in the posterior flap, and a tension-free mucosa-to-mucosa refluxing anastomosis is performed after spatulation of the distal ureteral end. Alternatively, nonrefluxing tunneled anastomosis can be used. The flap is then tubularized anteriorly and closed using absorbable suture. Furthermore, the ureteral adventitia may be secured to the distal aspect of the flap, and the base of the flap may be secured to the psoas (Fig. 41–33).
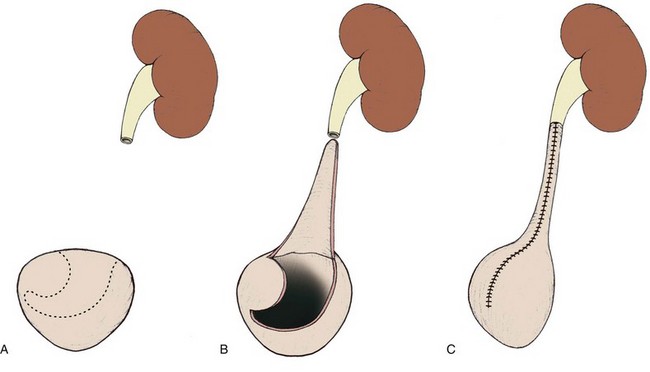
Figure 41–33 A, In Boari flap, the intended flap is first marked on the anterior and lateral aspects of the mobilized bladder. B, The flap is created, ensuring good vascular supply. C, Ureteroneocystostomy is completed, with the longitudinal bladder tube closure.
The number of reported patients treated with a Boari flap is small, yet the results are good if a well-vascularized flap is used (Ockerblad, 1947; Scott, 1972; Thompson, 1974; Middleton, 1980; Benson et al, 1990; Motiwala et al, 1990) (Fig. 41–34). The most common complication is clearly recurrent stricture formation, resulting from either ischemia or excessive tension on the anastomosis. Rare pseudodiverticulum has also been reported (Berzeg et al, 2003).
Laparoscopic Boari Flap
Laparoscopic Boari flap has been uncommonly yet successfully performed in the clinical setting. Kavoussi and colleagues reported three successful cases for distal ureteral obstruction, in which a transperitoneal approach was used (Fugita et al, 2001). Following the same principles in open surgery, the bladder flap was created and anastomosed to the ureteral end over a stent in a tension-free, water-tight manner. Operative time ranged from 120 to 330 minutes, and blood loss ranged from 400 to 600 mL. Two patients were discharged home within 3 days postoperatively, while 1 patient was hospitalized for 13 days for Clostridium difficile colitis. With a follow-up of more than 6 months, there was radiographically demonstrated patency of the anastomosis. In this report, the information of the length of distal ureteral stricture was not available. However, in the clinical experience of one of the authors (TH), ureteral loss of 8 to 12 cm can be comfortably bridged with Boari flap in the laparoscopic setting, as in open surgery. More recently, laparoscopic boari flap assisted by the robot has been successfully performed (Schimpf and Wagner, 2009). Transperitoneal approach was used in all cases reported thus far.
Renal Descensus
Renal mobilization, which was originally described by Popescu in 1964, can provide additional length to bridge a defect in the upper ureter or decrease tension on a ureteral repair (Harada et al, 1964; Passerini-Glazel et al, 1994). A transperitoneal, subcostal, midline, or paramedian incision may be used to gain access to the kidney and the appropriate level of the ureter. Following entry to the Gerota’s fascia, the kidney is completely mobilized and rotated inferiorly and medially on its vascular pedicle. The lower pole of the kidney is then secured to the retroperitoneal muscle using several absorbable sutures. Up to 8 cm of additional length may be gained using this technique. In such cases the renal vessels—especially the renal vein—limit the extent to which the kidney can be mobilized. As a solution, the technique for division of the renal vein with reanastomosis more inferiorly to the inferior vena cava may be performed but rarely applied clinically.
Intubated Ureterotomy
The Davis intubated ureterotomy has been described previously in this chapter. Because of the development of the more effective surgical treatment alternatives, this procedure is described primarily for historical interest. An intubated ureterotomy is generally used for a ureteral stricture too long for conventional ureteroureterostomy or ureteroneocystostomy and has been performed to treat stricture up to 10 to 12 cm in length. An innovative modification to this procedure has incorporated a buccal mucosal patch graft in a small number of patients with good results (Naude, 1999).
Open Transureteroureterostomy
The initial clinical application of TUU was described Higgins in 1934. In the management of ureteral stricture, a TUU may be used when ureteral length is insufficient for anastomosis to the bladder (Brannan, 1975). The only absolute contraindication is insufficient length of the donor ureter to reach the contralateral recipient ureter in a tension-free manner. However, any disease process that may affect both ureters represents a relative contraindication. Absolute contraindications include the presence of a diseased recipient ureter or a donor ureter of inadequate length. Relative contraindications include history of nephrolithiasis, retroperitoneal fibrosis, urothelial malignancy, chronic pyelonephritis, and abdominopelvic radiation. Reflux to the recipient ureter, if present, needs to be identified and corrected simultaneously. Therefore a voiding cystogram should be performed preoperatively, in addition to the other imaging and endoscopic studies previously described for thorough evaluation of both ureters.
In performing a TUU, a midline, transperitoneal approach is used to gain access to both ureters. Following medial colonic mobilization, the affected ureter is mobilized, preserving the adventitia with the ureteral blood supply, and divided just proximal to the level of obstruction. The contralateral colon is medially mobilized. Only the portion of recipient ureter needed for the anastomosis is exposed, which is generally 5 cm proximal to the level of division of the affected ureter. A tunnel under the sigmoid colon mesentery is created proximal to the inferior mesenteric artery to avoid ureteral tethering by this vessel, after which the donor ureter is then brought through the tunnel to the recipient side. Mobilization of the recipient ureter should be minimized to help preserve the integrity of its vascular supply. An anteromedial ureterotomy is made in the recipient ureter, which is then anastomosed to the spatulated donor ureteral end in a tension-free, watertight manner using either interrupted or running absorbable sutures. A double-J ureteral stent is usually passed from the donor renal pelvis, through the anastomosis and into the bladder. A second ureteral stent may also be placed throughout the length of the recipient ureter if the ureter is found to be adequately large in diameter.
The clinical success of TUU has been demonstrated by multiple investigators. Hendren and Hensle (1980) reported 75 cases of pediatric TUU without compromising a single recipient kidney. Hodges and colleagues (1980) reported a similar success in a large group of children and adults. However, two patients required revision due to ureteral kinking by the inferior mesenteric artery. The successful application of TUU was further confirmed more recently by Pesce and colleagues (Pesce et al, 2001). In two other recent studies, nephrectomy for ureteral stenosis was found to be rarely necessary (Mure et al, 2000; Sugarbaker et al, 2003).
Laparoscopic Transureteroureterostomy
Laparoscopic TUU is rarely performed and has been reported in three pediatric patients recently (Piaggio et al, 2007). The transperitoneal laparoscopic procedure was completed with the patient in the supine, Trendelenburg position. The general principles of donor and recipient ureteral dissection, donor ureteral ligation and transposition under the rectosigmoid mesentery, longitudinal ureterotomy at the medial aspect of the recipient ureter, and end-to-side anastomosis in a tension-free, water-tight manner are identical to those described for open surgery. Mean surgical time was 264 minutes (range: 200 to 313 minutes). Transient urine leak occurred in one of the three patients, with spontaneous resolution. There was normal renal function without complication at a mean follow-up of 6 months. Long-term clinical data to validate laparoscopic TUU are not yet available.
Open Ileal Ureteral Substitution
Surgical management of long length of ureteral defect or loss, especially the proximal ureter, is particularly challenging (Benson et al, 1990). Reconstruction of the ureter with tissue lined with urothelium is most preferable because urothelium is not absorptive and is resistant to the inflammatory and potentially carcinogenic effects of urine (Harzmann et al, 1986). Incorporation of other tissue in ureteral repair is, therefore, reserved for situations in which a defect cannot be bridged by other methods or the bladder is unsuitable for reconstruction. In this scenario, ileal interposition has been demonstrated to be a satisfactory option for complicated ureteral reconstruction. On the other hand, the appendix and fallopian tube have been found to be unreliable ureteral substitutes.
Shoemaker reported the first ileal ureter in a woman with tuberculous involvement of the urinary tract in 1909. Later, the metabolic and physiologic effects of the ileal ureter have been investigated in the canine model (Hinman and Oppenheimer, 1958; Martinez et al, 1965). When an isoperistaltic segment of ileum is directly anastomosed to the bladder, reflux and renal pelvic pressure increase are generally seen only during voiding. The retrograde transmission of intravesical pressure is dependent on the length of ileum segment used in interposition and the voiding pressure. In patients with ileal segments longer than 15 cm, Waldner and colleagues found no reflux into the renal pelvis in a report involving 19 cases of ileal ureter with refluxing ileo-vesical anastomosis (1999). Comparing dogs with tapered versus nontapered ileal segments, Waters and colleagues (1981) found no difference in renal perfusion pressure or metabolic derangements.
A large clinical experience in ileal ureter involving 89 patients was reported by Boxer and colleagues (1979). Only 12% of patients with normal preoperative renal function developed significant metabolic problems postoperatively, and preoperative renal function was identified to be an important prognostic factor. In a separate study, nearly half of those with a serum creatinine of greater than 2 mg/dL developed hyperchloremic metabolic acidosis, requiring conversion to a conduit (Koch and McDougal, 1985). In the same study, patients with bladder dysfunction also experienced more complications. No sufficient clinical data exist to establish the superiority of a tapered segment, a nonrefluxing anastomosis, or a shorter, segmental replacement over a standard ileal substitution (Waters et al, 1981). Therefore the general contraindications to an ileal ureteral substitution are baseline renal insufficiency with a serum creatinine of greater than 2 mg/dL, bladder dysfunction or outlet obstruction, inflammatory bowel disease, or radiation enteritis.
Before the surgical procedure, a full mechanical and antibiotic bowel preparation is often used. A long midline incision is made. The ipsilateral colon is mobilized medially, and the affected ureter is dissected proximally to the level of healthy tissue. The proximal anastomosis may be performed at the level of the renal pelvis if the entire upper ureter is unhealthy. The length of the ureteral defect is measured, and an appropriate segment of distal ileum is chosen. The segment should be at least 15 cm away from the ileocecal valve, and adequate blood supply should be confirmed before harvesting. The mesentery is usually divided more extensively than with a standard ileal conduit to provide greater mobility. Occasionally, a segment of colon may be more accessible than ileum and is harvested using the similar surgical principles. In the presence of a scarred or intrarenal pelvis, ileocalycostomy may be performed (McQuitty et al, 1995). In this circumstance, excision of a piece of lower pole renal parenchymal tissue is helpful in preventing stenosis at the anastomosis, as in a typical ureterocalicostomy. Following bowel division, the distal end of the ileal segment is marked for orientation and bowel-to-bowel continuity is reestablished. A small window is made in the colonic mesentery, through which the segment of ileum is delivered laterally. Alternatively, the cecum and ascending colon can be reflected superiorly to avoid mesenteric window creation in performing right ureteral reconstruction. The orientation of the ileal segment is checked to ensure isoperistalsis, and the anastomoses are performed at the level of the renal pelvis or lower pole calyx and at the bladder (Fig. 41–35). Bilateral ileal ureteral substitution may be achieved by using a longer segment that travels intraperitoneally from one kidney to the other and then to the bladder. An alternative to such is to use two separate bowel segments.
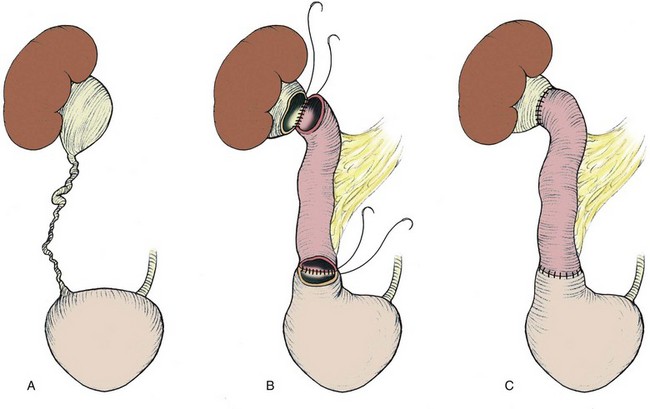
Figure 41–35 A, In ileal ureteral substitution, the affected ureter is first identified and dissected, which is followed by removal of the diseased portion. B, A piece of ileum is brought through the colonic mesentery to bridge the renal pelvis and the bladder. C, Both proximal and distal anastomoses are completed in full-thickness, water-tight, tension-free manner.
Perioperative complications associated with ileal ureter include early urinary extravasation or urinoma formation and obstruction from edema, a mucus plug, or a kink in the segment. Ischemic necrosis of the ileal segment may occur and should be considered if signs of an acute abdomen are present. Significant electrolyte abnormalities and renal insufficiency are unusual if preoperative renal function is normal. Patients with worsening metabolic abnormalities associated with a progressively dilating ileal ureter should be evaluated for vesicourethral dysfunction. Furthermore, malignancy arising from ileal ureter segment has been reported in four cases in the literature (Austen et al, 2004), and it is recommended that regular endoscopic examination should be performed starting at postoperative year 3 for early detection of such malignancy. However, Matiaga and colleagues (2003) recently confirmed the safety and reliability of ileal ureter creation for complex ureteral stricture/loss in 18 patients with a mean follow-up of 18.6 months. This conclusion was further supported by Bonfig and colleagues (2004) in another contemporary series involving 43 patients with a mean follow-up of 40.8 months.
Laparoscopic Ileal Ureteral Substitution
The clinical experience in laparoscopic ileal ureteral substitution is limited worldwide, yet this procedure appears to hold significant promise. Gill and colleagues (2000a) reported successful laparoscopic ileal ureter replacement using a transperitoneal, three-port approach. The entire procedure including freehand suturing and knot tying was performed using the intracorporeal laparoscopic techniques. Although the operative procedure was relatively long at 8 hours, there was minimal postoperative morbidity and hospital stays were short (4 days), as in most other laparoscopic procedures. More recently, robotic-assisted laparoscopic ileal ureter was performed via a transperitoneal, four-port approach successfully (Wagner et al, 2008). Total operative time was 9 hours, with an acceptable hospital stay of 5 days. No complication was reported in either study.
Autotransplantation
In 1963 Hardy performed the first autotransplantation for a patient with proximal ureteral injury. Since then, clinical autotransplantation has been performed for a variety of problems including extensive ureteral loss or stricture (Hardy, 1963; Novick and Stewart, 1981; Chuang et al, 1999; Wotkowicz et al, 2004). Generally, autotransplant is considered when the contralateral kidney is absent or poorly functioning or when other methods for ureteral substitution or repair are not feasible. The kidney is harvested with maximal vessel length as in a typical live donor nephrectomy for allotransplantation, and the renal vessels are anastomosed to the iliac vessels to reestablish renal perfusion. A healthy segment of the proximal ureter is anastomosed to the bladder (Bodie et al, 1986). Alternatively, the ipsilateral renal pelvis may be anastomosed directly to the bladder (Kennelly et al, 1993).
Recently, laparoscopy has been successfully incorporated in autotransplantation for severe ureteral loss. Nephrectomy can be performed laparoscopically as in any typical laparoscopic donor nephrectomy, followed by renal graft retrieval, bench preparation, and autotransplantation in the ipsilateral iliac fossa via a Gibson incision using the standard open surgical techniques (Fabrizio et al, 2000; Meng et al, 2003; Blueblond-Langner et al, 2004). The use of laparoscopy in autotransplantation has been shown to provide reduced postoperative analgesic need and faster recovery because a large open upper abdominal or flank incision for renal harvest is avoided. Laparoscopic nephrectomy in autotransplantation is most commonly performed transperitoneally. However, retroperitoneal approach for such purpose has been applied successfully by Gill and colleagues (2000b).
Key Points: Surgical Repair of Ureteral Stricture