Extracorporeal Shockwave Lithotripsy
Methods and Physical Principles
In extracorporeal SWL a source external to the patient’s body generates a shockwave. Specifically, the energy source rapidly deposits pulses of energy into a fluid environment, which results in the generation of a shockwave. Shockwaves are surfaces that divide material ahead, not yet affected by the disturbance, from that behind, which has been compressed as a consequence of energy input at the source (Sturtevant, 1996). These waves move faster than the speed of sound, and the stronger the initial shock, the faster the shockwave moves. Their behavior is characteristic of the propagation of nonlinear waves. Although the shockwaves in lithotripters generate large pressures, they are relatively weak in that they induce only slight compression and deformation of a material. The uniqueness of the shockwave lithotripter is in its exploitation of shockwave focusing. Relatively weak, nonintrusive waves are generated externally and transmitted through the body. The shockwaves build to sufficient strength only at the target, where they generate enough force to fragment a stone.
Generator Type
There are three primary types of shockwave generators: electrohydraulic (spark gap), electromagnetic, and piezoelectric.
Electrohydraulic (Spark Gap) Generator
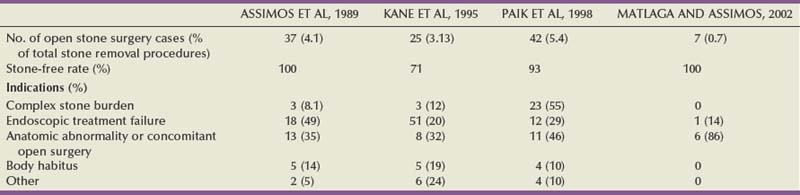
In the electrohydraulic shockwave lithotripter, a spherically expanding shockwave is generated by an underwater spark discharge (Cleveland et al, 2000). High voltage is applied to two opposing electrodes positioned about 1 mm apart. The high-voltage spark discharge causes the explosive vaporization of water at the electrode tip. For the spherically expanding shockwave to be focused onto a calculus the electrode is placed at one focus (termed F1) of an ellipsoid, and the target (the kidney stone) is placed at the other focus (termed F2). Figure 48–14 (on the Expert Consult website![]() ) shows a hemiellipsoid reflector and a spark gap typical of those used in the older electrohydraulic machines. This arrangement allows the projection of the majority of the original shockwave energy from the electrode tip to the stone, provided the electrode tip is precisely at F1. The body of the electrode varies in orientation among machines in that it is positioned within the ellipsoid to provide an easy means of replacement as it deteriorates.
) shows a hemiellipsoid reflector and a spark gap typical of those used in the older electrohydraulic machines. This arrangement allows the projection of the majority of the original shockwave energy from the electrode tip to the stone, provided the electrode tip is precisely at F1. The body of the electrode varies in orientation among machines in that it is positioned within the ellipsoid to provide an easy means of replacement as it deteriorates.
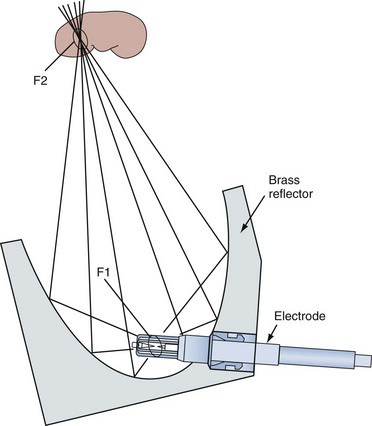
Figure 48–14 Schematic view of an electrohydraulic shockwave generator. An electrode is used to generate a shockwave.
The clear advantage of this generator is its effectiveness in breaking kidney stones (Lingeman, 1997). Disadvantages are the substantial pressure fluctuations from shock to shock and a relatively short electrode life. New longer life electrodes (like the NewTrode by HMT) have been developed to overcome these drawbacks. Another issue to consider is that as the electrode deteriorates, it wears down, and a 1-mm displacement of the electrode tip off of F1 can shift F2 up to 1 cm off of the initial target.
Electromagnetic Generator
Whereas the electrohydraulic lithotripter produces focused shockwaves by bouncing spherically expanding shocks off of an ellipsoid reflector, the electromagnetic generators produce either plane or cylindrical shockwaves. The plane waves are focused by an acoustic lens (Fig. 48–15); the cylindrical waves are reflected by a parabolic reflector (Fig. 48–16) and transformed into a spherical wave. The basic design of an electromagnetic generator is simple. Figure 48–15 shows a system that uses a water-filled shock tube containing two conducting cylindrical plates separated by a thin insulating sheet. When an electrical current is sent through one or both of the conductors, a strong magnetic field is produced between the conductors, moving the plate against the water and thereby generating a pressure wave. The electromagnetic force that is generated, termed magnetic pressure, causes a corresponding pressure (shockwave) in the water. The shock front produced is a plane wave that is of the same diameter as the current-carrying plates. The energy in the shockwave is concentrated onto the target by focusing it with an acoustic lens. The electromagnetic system that uses a cylindrical source (see Fig. 48–16) also has a cylindrical coil surrounded by a cylindrical membrane that is pushed away from the coil by the induction of a magnetic field between the two components. In both systems the pressure pulse has only one focal point (F2) that is positioned on the target.
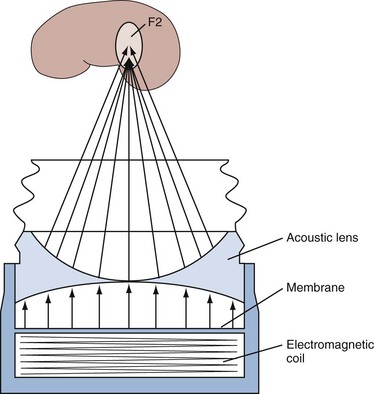
Figure 48–15 Schematic view of an electromagnetic shockwave generator that uses an acoustic lens to focus the shockwave. An electromagnetic coil is used to generate the shockwave.
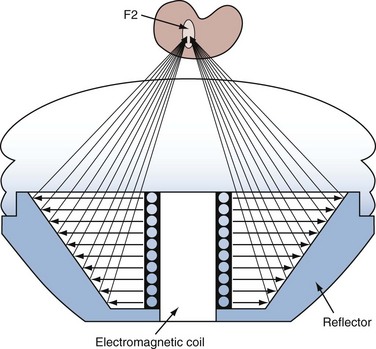
Figure 48–16 Schematic view of an electromagnetic shockwave generator that uses a parabolic reflector to focus the shockwave. An electromagnetic coil is used to generate the shockwave.
Electromagnetic generators are more controllable and reproducible than electrohydraulic generators because they do not incorporate a variable in their design such as the underwater spark discharge. Other advantages include the introduction of energy into the patient’s body over a large skin area, which may cause less pain. In addition, a small focal point can be achieved with high-energy densities, which may increase its effectiveness in breaking stones. This generator will deliver several hundred thousand shockwaves before servicing, thereby eliminating the need for frequent electrode replacement, which is required with most electrohydraulic machines. A disadvantage of this design may be that the small focal region of high energy results in an increased rate of subcapsular hematoma formation. The rate of subcapsular hematoma formation for the Storz Modulith has been suggested to be 3.1% to 3.7% (Dhar et al, 2004). Piper and associates (2001) suggested that perinephric hematomas may occur in up to 12% of patients treated with a DoLi S lithotripter. In contrast, perinephric hematomas were reported to occur in approximately 0.6% of patients undergoing SWL with the unmodified Dornier HM3 machine (Chaussy and Schmiedt, 1984; Knapp et al, 1987).
Piezoelectric Generator
The piezoelectric lithotripter also produces plane shockwaves with directly converging shockfronts. These generators are made of a mosaic of small, polarized, polycrystalline, ceramic elements (barium titanate), each of which can be induced to rapidly expand by the application of a high-voltage pulse (Fig. 48–17). Owing to the limited power of a single piezoelectric element, 300 to 3000 crystals are necessary for the generation of a sufficiently large shock pressure. The piezoelectric elements are usually placed on the inside of a spherical dish to permit convergence of the shockfront. The focus of the system is at the geometric center of the spherical dish.
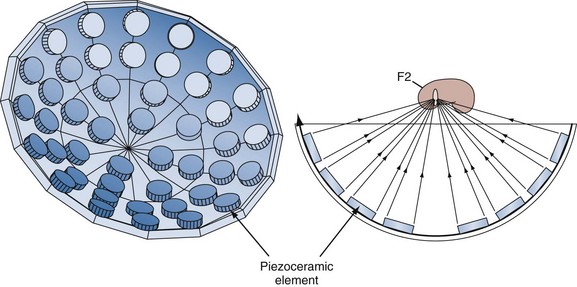
Figure 48–17 Schematic view of a piezoelectric shockwave generator. Numerous polarized polycrystalline ceramic elements are positioned on the inside of a spherical dish.
The advantages of this generator include the focusing accuracy, a long service life, and the possibility of an anesthetic-free treatment because of the relatively low-energy density at the skin entry point of the shockwave. For this reason, piezoelectric lithotripters in general tend to produce less discomfort than do lithotripters with other energy sources. A major disadvantage of this system is the insufficient power it delivers, which hampers its ability to effectively break renal stones. The piezoelectric energy sources produce some of the highest peak pressures of any lithotripter, but the actual energy delivered to the stone per shockwave pulse is several orders of magnitude lower than that delivered by an electrohydraulic machine because of the extremely tiny volume of F2.
Other Generators
Microexplosive generators have also been produced but have not gained widespread acceptance. The explosion of tiny lead azide pellets within a parabolic reflector generates the device’s shockwave (Kuwahara et al, 1987). Despite the effectiveness of this type of generator in producing shockwaves, this technology has not met with commercial success because of concerns about the storage and handling of the volatile lead azide pellets. Still other methods of shockwave generation use a laser beam or a multistage light gas gun, but these too have not been well received commercially.
Imaging Systems
There are three basic designs used by lithotripter manufacturers for stone localization. They are fluoroscopy alone, ultrasonography alone, and the combination of ultrasonography and fluoroscopy.
Fluoroscopy Alone
The original Dornier HM3 lithotripter used two x-ray converters arranged at oblique angles to the patient and 90 degrees from each other to localize the stone effectively at F2. To reduce the cost of lithotripters, an adjustable C-arm has been subsequently introduced on many devices. There is presently a remarkable similarity in the fluoroscopic systems used among manufacturers. This appears to be primarily the result of a common theme in the industry to develop multifunctional tables around these machines. The fluoroscopic system typically consists of a high-quality digitized x-ray imaging system mounted on a rotatable C-arm with an isocentrically integrated shockwave source. Because the shockwave head can be rotated out of the field of the fluoroscopic system, the table can be used for routine urologic fluoroscopic applications.
The primary advantages of fluoroscopy still include its familiarity to most urologists, the ability to visualize radiopaque calculi throughout the urinary tract, the ability to use iodinated contrast agents to aid in stone localization, and the ability to display anatomic detail. The disadvantages include the exposure of the staff and patient to ionizing radiation, the high maintenance demands of the equipment, and the inability to visualize radiolucent calculi without the use of radiographic contrast agents.
Ultrasonography Alone
Ultrasonic localization was initially designed to aid multifunctional lithotripters for treatment of both urinary and biliary stones. It is presently used in several low-cost machines because it is inexpensive to manufacture and to maintain compared with fluoroscopic systems. Another major advantage of this technology is in the treatment of children and infants when one is concerned about the dose of ionizing radiation. In addition, ultrasonography can localize slightly opaque or nonopaque calculi.
Despite its advantages there are a number of significant disadvantages of ultrasound imaging. Sonographic localization of a kidney stone requires a highly trained operator. To complicate the issue of stone detection is the fact that it is almost impossible to view a kidney stone in areas such as the middle third of the ureter or when there is an indwelling ureteral catheter. Once a stone is fragmented, it is difficult to identify each individual stone piece. Unfortunately, these disadvantages tend to overshadow the advantages of ultrasound imaging.
Combination of Ultrasonography and Fluoroscopy
As the demand for interdisciplinary lithotripters has increased, the lithotripsy industry has responded, in some cases combining ultrasonography and fluoroscopy for stone localization. There are clearly advantages to these setups, but each system has a drawback that limits one of the functions of the system.
Anesthesia
The approach to anesthesia for lithotripsy has changed considerably since clinical SWL began in 1980. At that time, regional or general anesthesia was used in all instances because the unmodified HM3 device (15.6-cm ellipsoid; 80-nF generator) produced a powerful shockwave and treatment at recommended energy levels caused intolerable pain. Subsequently, urologists and lithotripter manufacturers recognized that the HM3 is considerably more powerful at the recommended energy setting than is necessary for the fragmentation of most renal calculi, an observation that spawned interest in less powerful lithotripters with lessened anesthesia requirements (Marberger et al, 1988; Wilbert et al, 1987). Several researchers have noted that the original HM3 lithotripter without modification produces excellent clinical results when it is used at lower energy settings (Pettersson et al, 1989; Tiselius, 1991; Tolley et al, 1991). In addition, such settings create a smaller lesion at F2 in experimental animals (Connors et al, 2000).
The discomfort experienced during SWL is related directly to the energy density of the shockwave as it passes through the skin as well as the size of the focal point. In the past decade several new and useful anesthetic techniques adaptable to SWL have been produced that were not available at the time SWL was introduced and include short-acting parenteral sedative-narcotics and topical agents.
Short-acting agents, such as the narcotic alfentanil and the sedative-hypnotics midazolam and propofol, have been used in various combinations to allow most SWL treatments with any lithotripter (including the unmodified Dornier HM3) to be accomplished comfortably for the patient without the need for general or regional anesthesia, if the patient so desires. Monk and associates (1991) compared two sedative-analgesic techniques (midazolam-alfentanil vs. fentanyl-propofol) and found that both techniques provided adequate anesthesia for SWL with use of an unmodified Dornier HM3 lithotripter. Anesthesia and recovery times were significantly shorter than those recorded for epidural anesthesia techniques. These findings have been confirmed by others (Nelson et al, 2001; Burmeister et al, 2002; Ozcan et al, 2002).
Another approach to minimize anesthesia requirements during SWL has been the use of topical agents. EMLA cream, a eutectic mixture of lidocaine and prilocaine, has been shown significantly to reduce anesthesia requirements during SWL (Basar et al, 2003). A topical agent, EMLA cream should be applied at least 45 minutes before SWL. The combination of topical agents and short-acting intravenous agents is likely to minimize the amount of these agents required and to shorten recovery times.
Not all patients are well served by treatment with a low-energy SWL technique, and therefore a variety of factors need to be considered in choosing the preferred approach for SWL. Calculi composed of cystine, calcium oxalate monohydrate, or brushite are known to be resistant to fragmentation; if their presence is anticipated, delivery of higher levels of shockwave energy with attendant increased anesthesia requirements should be expected (Dretler, 1988; Klee et al, 1991). Thin patients have more pain during SWL because the converging shockwave is more concentrated at the point of skin penetration. Children and extremely anxious individuals may be served best by general anesthesia. If a lengthy treatment session is anticipated (i.e., bilateral SWL or treatment of ureteral and renal stones), the larger amount of topical and intravenous agents required lessens their appeal.
One important observation regarding the issue of general anesthesia versus intravenous sedation was reported by Sorensen and colleagues (2002) and Eichel and colleagues (2001). In a comparison of patients treated with the DoLi 50 lithotripter, those patients who received general anesthesia experienced a significantly greater stone-free rate than did those patients who underwent intravenous sedation. One possible explanation for this finding is the more controlled respiratory excursion that is conferred by the general anesthetic.
Lithotripter Comparisons
Shockwave lithotripters are considered by the U.S. Food and Drug Administration to be Class II devices. As such, for a lithotripter to be brought to market simply requires documentation that the device has the same intended use and the same technologic characteristics as a predicate device that has already been approved and brought to market. Specific testing that evaluates, in a proscribed manner, the lithotripter’s treatment efficacy and safety is not required. Largely as a consequence of this practice, few, if any, appropriately designed comparative trials of lithotripters exist in the published literature. In addition, there are no validated standards within the lithotripsy industry regarding a method of quantification of the power and efficiency of lithotripters, a problem further compounded by a lack of knowledge of the number of shockwaves that can be safely administered to a kidney during any single SWL session with any lithotripter. Although there is general consensus that re-treatment rates are an appropriate indicator of lithotripter effectiveness, the lack of clinical agreement about the appropriate outcome of lithotripsy (i.e., stone free vs. residual fragments of various size) further hampers comparisons of lithotripters.
Only a small part of the literature published to date on the outcomes of SWL presents data that are stratified sufficiently to permit a meaningful comparative analysis. Surprisingly, despite the proliferation of lithotripters and the variety of solutions devised for stone targeting and shockwave delivery, no other lithotripter system has convincingly equaled or surpassed the results produced by the unmodified Dornier HM3 device. That the most effective lithotripter was invented first is a remarkable achievement for Dornier. In general, the less powerful lithotripters with smaller focal points result in lower stone-free rates or higher re-treatment rates. Additionally, it is now recognized that SWL inflicts a trauma similar to a renal contusion, which occasionally can result in adverse clinical sequelae. Potential concerns about the long-term effects of lithotripsy with the unmodified Dornier HM3 device may have been one motivating factor in the trend within the lithotripsy industry toward, at first, lower power but eventually higher power lithotripters with smaller focal points, with the goal that the efficacy of lithotripsy could be maintained while producing fewer deleterious effects on renal tissue (Figs. 48–18 [on the Expert Consult website![]() ] and 48–19). Unfortunately, the newer lithotripters are less efficacious than the original Dornier device, and no published information is available to suggest that newer lithotripters produce fewer adverse effects for equivalent degrees of efficacy.
] and 48–19). Unfortunately, the newer lithotripters are less efficacious than the original Dornier device, and no published information is available to suggest that newer lithotripters produce fewer adverse effects for equivalent degrees of efficacy.
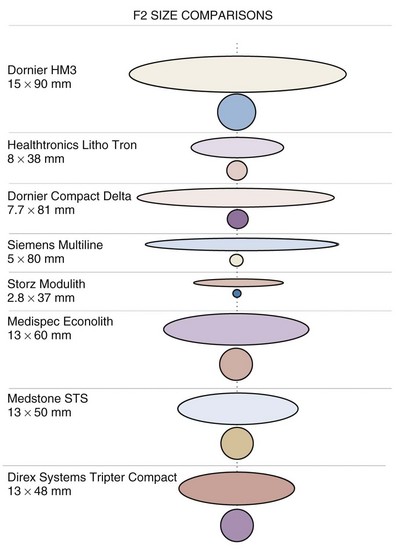
Figure 48–19 Comparison of the focal zones of selected clinical lithotripters showing their dimensions along the axis of the lithotripter (ellipses) and in the focal plane at the focus (circles).
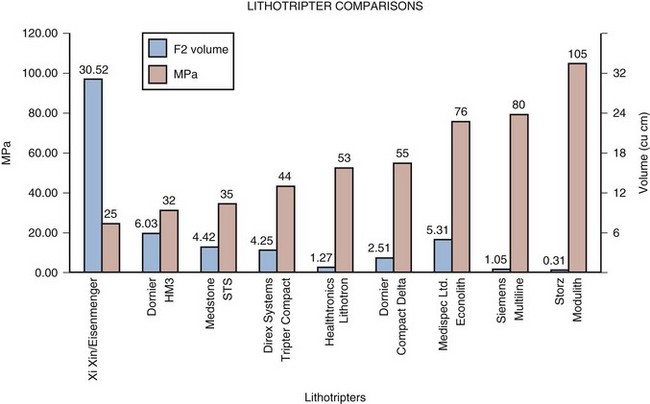
Figure 48–18 Comparison of the peak amplitude and size of the focal volume of nine different lithotripters. The general trend (from left to right) is a decrease in the device focal volume and an increase in the peak positive pressure. At the far left is the Xi Xin/Eisenmenger lithotripter, a new design that goes against the trend with a diminished peak amplitude and enlarged focal area.
Mechanisms of Stone Comminution
Present knowledge in the field of SWL suggests that comminution of a renal stone in a lithotripter field is the consequence of failure of the stone material due to the mechanical stresses produced either directly by the incident shockwave or indirectly by the collapse of cavitation bubbles. These events could be occurring simultaneously or separately at the surface of the stone or within the interior of the stone (Fig. 48–20). Several potential mechanisms for SWL stone breakage have been described: spall fracture, squeezing, shear stress, superfocusing, acoustic cavitation, and dynamic fatigue.
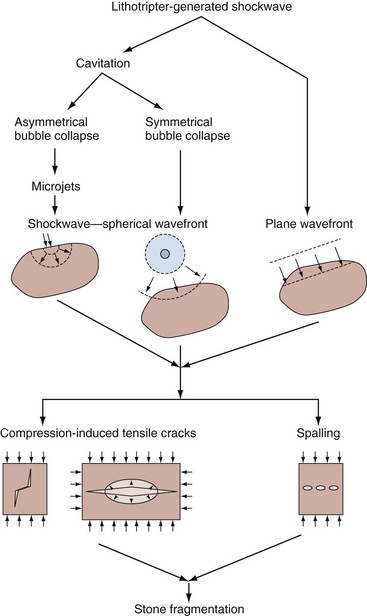
Figure 48–20 Summary of how the various mechanical forces generated by a lithotripsy shockwave might cause a kidney stone to fracture.
(Reproduced with permission of Dr. Bradley Sturtevant.)
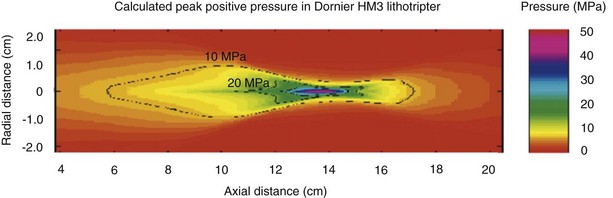
Before each of these mechanisms is discussed, consideration of the typical shockwave profile is required. A typical pressure pulse generated by an electrohydraulic shockwave lithotripter is shown in Figure 48–21. It involves an initial short and steep compressive front with pressures of about 40 MPa that is followed by a longer, lower-amplitude negative (tensile) pressure of 10 MPa, with the entire pulse lasting for a duration of 4 µsec. Note that the ratio of the positive to negative peak pressures is approximately 5 : 1. Pressure measurements near the focal region of a Dornier unmodified HM3 indicate a 6-dB beam, of a width of approximately 15 mm. Because kidney stones are also generally of this dimension, the wavefront incident on the stone can be considered a plane wave (Muller, 1990; Cleveland et al, 2000).
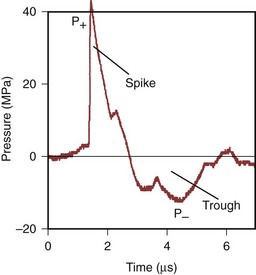
Figure 48–21 A typical pressure pulse at the lithotripter focus (F2) as measured by a polyvinylidene difluoride membrane hydrophone. First, there is a steep positive pressure front of about 40 MPa, which is followed by a negative pressure of 10 MPa, with the entire pulse lasting for a duration of 4 µsec.
(From Coleman AJ, Saunders JE, Preston RC, et al. Pressure waveforms generated by a Dornier extracorporeal shockwave lithotriptor. Ultrasound Med Biol 1987;13:651–7.)
The first mechanism by which a stone might break is through spall fracture. Once the shockwave enters the stone it will be reflected at sites of impedance mismatch. One such location is at the distal surface of the stone at the stone-fluid (urine) interface (although there could be other internal sites, such as cavities in the stone and interfaces of crystalline and matrix materials). As the shockwave is reflected, it is inverted in phase to a tensile (negative) wave. If the tensile wave exceeds the tensile strength of the stone, there is an induction of nucleation and growth of microcracks that eventually coalesce, resulting in stone fragmentation, which is termed spallation. The failure plane is located perpendicular to the applied tensile stress. This mechanism is thought to be of considerable importance in that kidney stones, like most brittle materials, will be much more likely to fail under tension rather than compression (Johrde and Cocks, 1985). Lokhandwalla and Sturtevant (2000) have suggested that the trailing negative pressure of the lithotripter pulse also exerts tensile stresses of an order of magnitude similar to that of the spall mechanism. Contributing factors to the effectiveness of spallation in generating stone breakage appear to be the size and the shape of the stone as well as its physical properties (i.e., fracture toughness, acoustic speed, density, and void dimensions). More spherically shaped stones may focus the tensile wave after reflection and thus further increase the tensile stress. Stones with larger diameters may allow sufficient tensile stress to be generated so that the tensile strength of the stone can more easily be exceeded. If these factors are important, then smaller, irregularly shaped stones may not fracture by spallation.
Eisenmenger (1998) first suggested that the second mechanism for stone breakage, termed squeezing-splitting or circumferential compression, occurs because of the difference in sound speed between the stone and the surrounding fluid. The shockwave inside the stone advances faster through the stone than the shockwave propagating in the fluid outside the stone. The shockwave that propagates in the fluid outside the stone thus produces a circumferential force on the stone, resulting in a tensile stress in the stone that is at its maximum at the proximal and distal ends of the stone. The resulting squeezing force could split the stone either in a plane parallel to the shockwave propagation direction or, depending on the elastic properties of the stone, possibly in a plane parallel to the shockwave front. It has been theorized that squeezing should be enhanced when the entire stone falls within the diameter of the focal zone. Thus, current third-generation lithotripters that have very small focal zones will not make use of this mechanism, as the stone size is typically greater than the focal zone, whereas the original Dornier HM3 machine would.
The third mechanism is shear stress. Shear stress will be generated by shear waves (also termed transverse waves) that develop as the shockwave passes into the stone. The shear waves propagate through the stone and will result in regions of high shear stress inside the stone. In contrast to compression waves, which move the molecules in the direction of propagation, a shear wave results in translation of molecules transverse to the direction of propagation, and therefore the molecules are not compressed but are shifted sideways by the wave. Many materials are weak in shear, particularly if they consist of layers, because the bonding strength of the matrix between layers often has a low ultimate shear stress. Calcium oxalate stones commonly possess alternating layers of mineral and matrix, and the shear stress induced by the transverse wave could cause such stones to fail. Theoretical work by Sapozhnikov and colleagues (2003) suggests that the shear wave mechanism will lead to a tensile strain in cylindrical stones that is 5 to 10 times larger than that induced by spall. They also suggest that cracks will be initiated in the center of the stone and grow in a direction perpendicular to the axis of the stone.
The fourth mechanism for stone breakage, superfocusing, is the amplification of stresses inside the stone due to the geometry of that stone. The shockwave that is reflected at the distal surface of the stone can be focused either by refraction or by diffraction from the corners of the stone. Several groups have demonstrated that these reflected waves can be focused to regions of high stress in the interior of the stone and that this can lead to failure (Gracewski et al, 1993; Xi and Zhong, 2001). The regions of high stress (both tensile and shear) are dependent on the geometry of the stone as well as its elastic properties.
The fifth potential mechanism for SWL stone breakage is cavitation (Coleman et al, 1987; Crum, 1988; Vakil and Everbach, 1993; Zhong and Chuong, 1993; Zhong et al, 1993). Cavitation is defined as the formation and subsequent dynamic behavior of bubbles. The lithotripter-generated pressure field has been found to induce cavitation in both in vitro and in vivo studies. The negative pressure in the trailing part of the pulse causes bubbles to grow at nucleation sites. A nucleation site is an inhomogeneity in the fluid, which leads to preferential formation of free gas under stress. During the negative pressure wave, the pressure inside the bubble falls below the vapor pressure of the fluid, and the bubble fills with vapor and grows rapidly in size (almost three orders of magnitude). As these bubbles grow, they oscillate in size for about 200 µsec and then collapse violently, giving rise to high pressures and temperatures. In the absence of any boundaries, a cavitation bubble remains spherical during collapse, releasing energy primarily by sound radiation, the majority of which is in the form of a shockwave (see Fig. 48–21). This shockwave generates a positive and negative wave and therefore can induce all of the fragmentation mechanisms described in the preceding. However, in the presence of a boundary, a liquid jet, also termed a cavitation microjet, forms inside the bubble during the collapse (Crum, 1979, 1988). This jet can accelerate to extremely large speeds because it converts most of its kinetic energy from the collapse of the cavity interface to the jet itself. The typical bubble radii found in SWL vary from 1 µm to 1 mm, and bubble jet velocities range from 22 m/sec to 800 m/sec. In actual jet-impact cases the duration of the pressure pulse is only a few microseconds, and in most instances, the peak pressure lasts for only about 1 µsec. If the liquid jet is near the surface of a stone, it creates a locally compressive stress field in the stone, which propagates spherically into the stone interior.
Numerous investigators have exposed either aluminum foil or brass plates to the focused shockwave generated by a Dornier HM3 machine and observed significant microjet damage (pitting) on the surfaces of these metals. If this event occurs at the surface of a kidney stone, erosion of this surface would be expected; Averkiou and Crum (1996) reported this event for SWL-treated plaster of Paris target stones. To determine if cavitation is the primary mechanism of stone fragmentation, investigators have developed in-vitro systems that would eliminate or dampen cavitational events. Such systems have included a viscous medium that possesses a much lower number of nucleation sites and a chamber that allows one to increase the ambient pressure that surrounds the growing cavitation bubbles (Vakil et al, 1991; Delius, 1997; Stonehill et al, 1998). These in-vitro systems have shown reduced stone damage along with a reduction in cavitation activity. Work by Bailey and associates (1998, 1999), in which the positive and negative waves were inverted with a pressure release reflector, also showed a reduction in stone comminution. All of these studies suggest that cavitation plays a significant role in damaging brittle objects.
The final mechanism of stone fragmentation to be considered defines stone breakage in terms of a dynamic fracture process, in which the damage induced by SWL accumulates during the course of the treatment, leading to the eventual destruction of the stone. Essential to this process is nucleation, growth, and coalescence of flaws within the stone caused by a tensile or shear stress (Fig. 48–22). Because renal calculi are not homogeneous but rather have either a lamellar crystalline structure bonded by an organic matrix material or an agglomeration of crystalline and noncrystalline material there are numerous sites of preexisting flaws (microcracks). All of the fracture mechanisms described have the potential to generate progressive damage to the interior of the stone. By use of the cohesive-zone model, a mathematical approach of predicting the qualitative features of transient microcrack damage accumulation, Lokhandwalla and Sturtevant (2000) were able to calculate the number of shockwaves required for a spall-like failure to occur in a typical calcium oxalate monohydrate calculus. The values they determined had a range of two orders of magnitude (30 to 3000 shocks), which is well within the clinical dose presently used to treat patients. These investigators further suggested that mechanisms other than spall are also likely to inflict damage to stones, and spall may be a factor only in a small portion of the stone.
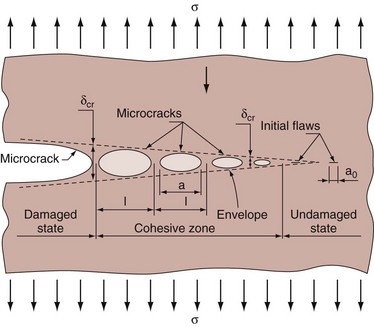
Figure 48–22 Coalescence of microcracks with a main crack. The undamaged stone has some initial flaws or microcracks of known length. These flaws occur in renal calculi either at a lamellar crystalline structure bonded by an organic matrix material or at an agglomerate of crystalline and noncrystalline material. When stressed, these microcracks grow until they coalesce with the main crack; and because this event is repeated throughout the stone, it eventually fragments.
(Reproduced with permission of Dr. Bradley Sturtevant.)
Bioeffects: Clinical Studies
Acute Extrarenal Damage
SWL induces acute injury in a variety of extrarenal tissues (Evan et al, 1991, 1998). SWL has been associated with trauma to organs such as the liver and skeletal muscle, as evidenced by elevated levels of bilirubin, lactate dehydrogenase, serum aspartate transaminase, and creatine phosphokinase within 24 hours of treatment (Lingeman et al, 1986a; Ruiz Marcellan and Ibarz Servio, 1986; Parr et al, 1988). These parameters begin to fall within 3 to 7 days of SWL treatment and are normal at 3 months. Other findings of damage outside the kidney have included reports of visceral injuries, such as perforation of the colon, hepatic hematoma, splenic rupture, pancreatitis, and abdominal wall abscess. Extrarenal vascular complications have been reported to occur as well, such as rupture of the hepatic artery, rupture of the abdominal aorta, and iliac vein thrombosis. Thoracic events, such as pneumothorax and urinothorax, have even been described. Fortunately, these events are all exceedingly rare, and have generally been presented as isolated incidents.
In addition, early clinical studies noted that shockwaves could induce cardiac arrhythmia, an observation that led to electrocardiographic synchronization with R-wave triggering on the Dornier HM3 device (Chaussy and Schmiedt, 1984). However, later clinical studies with non–water bath lithotripters have concluded that treating ungated to cardiac rhythm is safe.
Although the lithotripter is characterized by the spatial distribution of its acoustic output (the focal zone, or F2), it is known that high acoustic pressure does extend beyond this zone (Fig. 48–23). Therefore, it is reasonable to expect that organs other than the kidney are exposed to stresses sufficient to cause injury. One such organ is the pancreas; a retrospective follow-up study from the Mayo Clinic suggested that patients who underwent SWL for the treatment of kidney stones in 1985 were at increased risk for developing diabetes mellitus compared with controls (Krambeck et al, 2005). The development of diabetes was related to the total number of shockwaves and the power level of the lithotripter. Although these data are provocative, there were a number of limitations of the study, including that the stone disease of the SWL cohort was more severe than the control cohort, a family history of diabetes was not ascertained for either group, and the data for the SWL group were collected by self-report questionnaire whereas the control group was examined by chart review. Several other groups have subsequently investigated this subject, however; and these findings have not been confirmed by any other study (Sato et al, 2008; Makhlouf et al, 2009). At present, then, there have yet to be any corroborative analyses confirming a relationship between diabetes and SWL.
Acute Renal Injury: Structural and Functional Changes
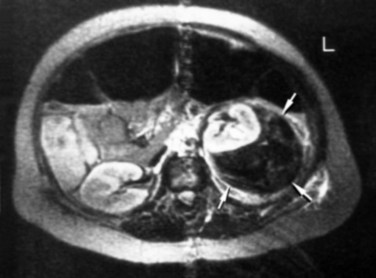
Virtually all patients who undergo SWL for renal stones demonstrate hematuria after approximately 200 shockwaves. Hematuria is so common that it may be considered an incidental finding, and its severity is rarely of concern. Although hematuria was initially considered to be a consequence of irritation of the urothelium as stones were fragmented by shockwaves, it is now known that such is not the case. Detailed morphologic studies have demonstrated that shockwaves rupture blood vessels and can damage surrounding renal tubules (Fig. 48–24). SWL is now known to induce such structural changes in the treated kidney in the majority, if not all, SWL patients, regardless of the type of lithotripter employed (Table 48–9).
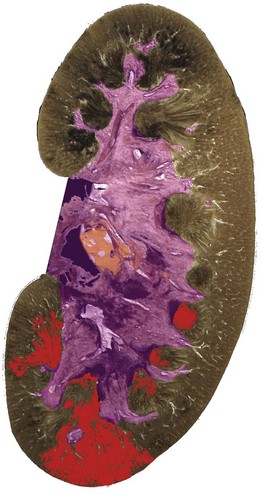
Figure 48–24 Macroscopic photomicrograph of a coronal section through the kidney of a juvenile pig (about 6 weeks old) treated with 2000 shocks at 24 kV by an unmodified Dornier HM3 lithotripter and examined 4 hours after treatment. The region of intraparenchymal hemorrhage has been colored red by an automated computer color recognition program. Note that the lesion involves multiple papillae and in some regions extends through the cortex to the renal capsule, where a subcapsular hematoma may develop.
Table 48–9 Renal Side Effects of Shockwave Lithotripsy in Experimental Animal Models (Canine and Porcine)
| Acute Histologic Changes |
| Chronic Histologic Changes |
In porcine subjects, the preferred animal model for studying acute renal injury, SWL traumatizes vessels ranging in size from the glomerular and cortical capillaries and vasa recta, to the larger arcuate and intralobular vessels. The resulting hemorrhagic lesion generally extends from cortex to medulla and comprises torn blood vessels with platelet aggregation and red blood cells in the interstitial space (Figs. 48-25 and 48-26 on the Expert Consult website![]() ). Affected renal corpuscles typically show breaks in the Bowman capsule, blood in the urinary space, and damage to the podocytes and mesangial cells (Fig. 48–27 on the Expert Consult website
). Affected renal corpuscles typically show breaks in the Bowman capsule, blood in the urinary space, and damage to the podocytes and mesangial cells (Fig. 48–27 on the Expert Consult website![]() ). Renal tubules often contain blood cell casts, and the tubular cells may show ischemic changes. In the setting of a more severe injury, complete necrosis of the endothelium and vascular smooth muscle may result. A typical clinical dose of 2000 shockwaves with the Dornier HM3 lithotripter, operated at 24 kV with shockwaves delivered at 2 Hz produces a lesion measuring 5% to 6% of the functional renal volume (Fig. 48–28).
). Renal tubules often contain blood cell casts, and the tubular cells may show ischemic changes. In the setting of a more severe injury, complete necrosis of the endothelium and vascular smooth muscle may result. A typical clinical dose of 2000 shockwaves with the Dornier HM3 lithotripter, operated at 24 kV with shockwaves delivered at 2 Hz produces a lesion measuring 5% to 6% of the functional renal volume (Fig. 48–28).
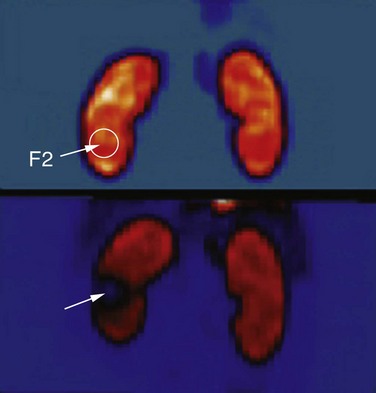
Figure 48–28 Shockwave lithotripsy–treated and control kidneys imaged by positron emission tomographic scanning before and immediately after treatment with 3500 shockwaves to the lower pole, at level six, with a DoLi 50 device. The site of F2 (lower pole) on the shocked kidney shows a 50% reduction of renal blood flow (arrow).
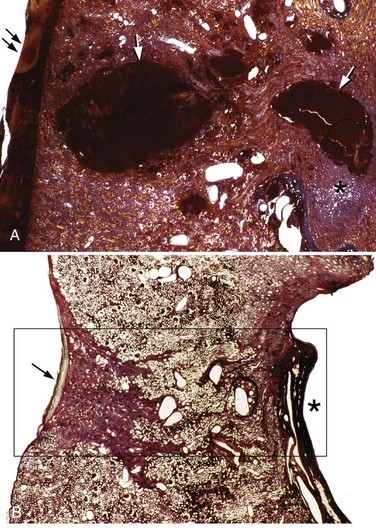
Figure 48–25 Light micrographs of an acute shockwave lithotripsy (SWL)-induced lesion at F2 (A) and subsequent chronic changes at a similar site 3 months after SWL treatment (B). Each pig kidney was treated with 2000 shockwaves at 24 kV by an unmodified Dornier HM3 lithotripter. The acute lesion is characterized by numerous sites of hemorrhage (arrows) that extend from an individual renal papilla (asterisk) to the outer cortex of the kidney. Note a subcapsular hematoma (double arrows, A). The tissue section in B is similar in location to that seen in A but is shown at 3 months after SWL. A rectangle outlines the site of F2. Within that region there is complete loss of the renal papilla (the asterisk indicates where it should be), and only scar tissue is found in the adjacent cortical tissue (arrow).
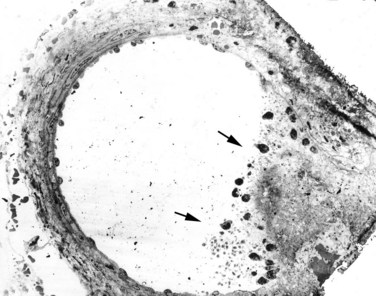
Figure 48–26 Low-magnification transmission electron micrograph demonstrating injury to a medium-sized artery located within F2 of a pig treated with 2000 shockwaves at 24 kV. The shockwave-induced injury to the right side of this vessel resulted in a rupture site that permitted extravasation of blood into the nearby interstitium. The site of injury in the vessel wall is plugged with a clot (arrows).
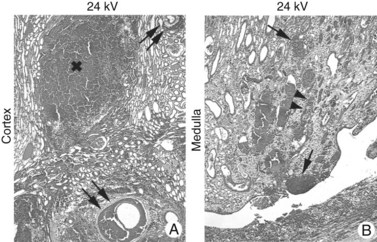
Figure 48–27 This series of light microscope panels depicts the injury seen in the cortex and medulla from an animal treated with 2000 shockwaves at 24 kV by an unmodified Dornier HM3 lithotripter. A and B illustrate extensive injury in both cortex and medulla. Within the cortex, disruption of arterial walls with hemorrhage (double arrows) is noted near sites of intraparenchymal bleeding (x). The first site of injury appears to occur in the renal medulla, where damage is noted to small vessels, which causes intraparenchymal hemorrhage (arrows) adjacent to damaged collecting ducts (arrowheads).
There have been reports of moderate to severe renal injury occurring after SWL, generally manifesting as a hemorrhagic event. Hematoma rates range from less than 1% up to as high as 20%, depending on the type of lithotripter used and the treatment parameters employed, as well as the radiographic modality and timing of imaging follow-up. In addition, the later generation lithotripters that have small focal areas and extremely high peak positive pressures are reported to produce higher clinically significant hematoma rates (3% to 12%), a trend that is worrisome (Thuroff et al, 1988; Ueda et al, 1993; Kohrmann et al, 1995; Piper et al, 2001). Several risk factors for the development of a post-SWL hematoma have been identified (Table 48–10). Dhar and associates (2004) reported that the probability of a subcapsular hematoma increased 2.2 times for every 10-year increase in the patient’s age. Knapp and associates (1988) found patients with existing hypertension to be at increased risk for the development of perinephric hematomas as a consequence of SWL. In particular, those patients having unsatisfactory control of their hypertension at the time of SWL had the highest incidence of hematoma formation. Additional risk factors for hemorrhage were diabetes mellitus, coronary artery disease, and obesity, all of which suggest a link to a vascular disorder. The appearance of renal hematomas can range in severity from a mild contusion localized within the renal parenchyma to a large hematoma (Fig. 48–29) associated with severe bleeding, possibly necessitating blood transfusion or rarely even angiographic embolization. Although some hematomas may persist for many months to years, it has been reported that most resolve within weeks and without long-term sequelae.
Table 48–10 Acute Renal Side Effects: Risk Factors for Shockwave Lithotripsy
Chronic Renal Injury: Structural and Functional Changes
At present there is a paucity of information on the chronic injury induced by SWL, due in great part to the lack of experimental animal studies. Nonetheless, it is well accepted that shockwaves damage blood vessels, and the resulting hemorrhage initiates an inflammatory response that ultimately leads to scar formation. Parenchymal fibrosis, a precursor to renal scarring, is seen as early as 1 month after SWL, and scar formation has also been reported to be a dose-dependent phenomenon. Clinically there are four potential chronic renal changes that may be associated with SWL treatment. They are an accelerated rise in systemic blood pressure, a decrease in renal function, an increase in the rate of stone recurrence, and the induction of brushite stone disease. All four effects appear to be linked to the observation that the acute renal injury at F2 progresses to scar formation.
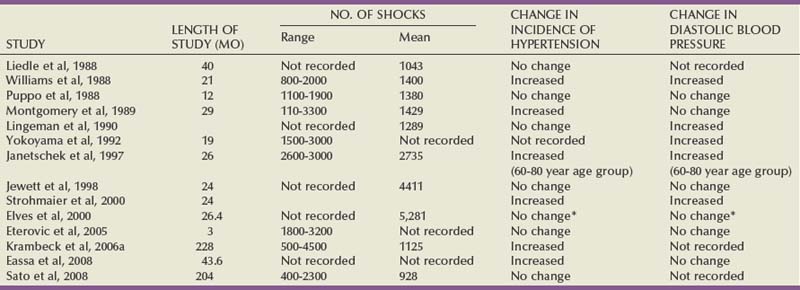
The possibility that SWL might be associated with significant changes in systemic blood pressure was first suggested by Peterson and Finlayson (1986) and has been investigated by others (Table 48–11). Lingeman and coworkers (1987b) reported that 8.2% of 243 patients who were normotensive at the time of SWL developed blood pressure changes requiring antihypertensive medication. Mean follow-up in this group of patients was 1.5 years, giving an annualized incidence of hypertension of 5.5%. Similar data have been reported by Williams and Thomas (1989). After these reports suggesting that hypertension could be a long-term complication of SWL, a large study involving almost 1000 patients was undertaken at the Methodist Hospital of Indiana (Lingeman et al, 1990). This study found a small but statistically significant change in diastolic blood pressure associated with SWL therapy. The observed effect of SWL on diastolic pressure change persisted even after controlling statistically for other variables that might be associated with variation in blood pressure, such as age, sex, pretreatment baseline blood pressure, and number of treatment sessions. Janetschek and colleagues (1997) performed a prospective study that demonstrated age was a significant risk factor for post-SWL hypertension, with an increase in intrarenal resistive index observed in patients 60 years of age and older. The mechanism of hypertension after SWL is not well elucidated. Although subcapsular hematomas can induce hypertension, such changes are generally transient. There has been a report, however, of mesangial proliferation in porcine models after SWL that could induce hypertensive changes.
The possibility that SWL treatment might be associated with a long-term reduction in renal function has been suggested by several investigators. Williams and associates (1988) found a significant decrease in the percentage of effective renal plasma flow 17 to 21 months after SWL for patients with two kidneys. Orestano and colleagues (1989) noted that patients receiving more than 2500 shocks had a reduction in creatinine clearance and a prolongation of 131I-Hippuran transit time 30 days after SWL in the treated kidney; in some cases, similar findings were noted in the contralateral kidney. Lingeman and associates have reported that patients with a solitary kidney demonstrated elevated serum creatinine levels 5 years after SWL (Brito et al, 1990). These observations stand in contrast to the early reports by Chaussy and Fuchs (1986), which suggested a significant increase in renal function 3 months to 1 year after SWL. In addition, a longer follow-up study of patients treated in Munich has failed to confirm this increase in renal function (Liedle et al, 1988).
An additional concern is that stone recurrence rates may be higher after SWL because of residual stone debris (Pearle et al, 1999). A study by Carr and associates (1996) documented new stone formation in 298 consecutive patients who initially were determined to be stone free after SWL and compared those findings with those of 62 patients treated by PNL. Their data showed a significant increase in the rate of new stone formation within 1 year of SWL treatment compared with PNL. The authors suggested that fine sand debris generated from SWL treatment remained in the kidney and gravity acted to position it as a nidus in the calyceal system.
There has been reported to be a significant increase in the number of calcium phosphate stone formers during the past 3 decades (Mandel et al, 2003; Parks et al, 2004). An intriguing finding in Parks and colleagues’ work was that when all kidney stone formers were analyzed for the number of SWL procedures, the calcium phosphate stone formers had received a significantly higher number of procedures than did the idiopathic calcium oxalate stone formers when rates were adjusted for number of stones and duration of stone disease. Furthermore, the brushite stone formers had received a significantly greater number of SWL treatments than had the apatite stone formers. The histopathologic examination of the brushite stone formers revealed advanced levels of tissue changes in the renal cortex and papilla that included interstitial fibrosis, tubular atrophy, glomerular obsolescence, and deposition of large amounts of biologic hydroxyapatite in the lumens of inner medullary collection ducts (Evan et al, 2005). Although these data do not establish a cause-and-effect relationship, clearly there is an association between brushite stone disease and high levels of SWL treatment sessions. Because apatite stone disease is likely to be related to higher urine pH levels in these patients, animal studies that showed the initial site of SWL injury to be localized to the microvessels and collecting duct of the renal papilla may explain the loss of control over normal urinary fluid pH.
Mechanism for Tissue Injury
The mechanism for the traumatic effects of SWL is not known, although Delius and colleagues (1988) have speculated that the violent collapse of cavitation bubbles generated by the shockwaves is primarily responsible for the cellular changes (Table 48–12). This concept is based on data showing that cavitation bubbles are present during shockwave application and that lithotripter shockwaves can cavitate water and blood in vitro (Coleman et al, 1987). Crum (1988) documented that SWL does produce acoustic cavitation, possibly as the result of the high intensity of the shockwave amplitude, and noted that the cavitation microjets are sufficiently forceful to pit or deform metal test foils. Zhong and coworkers (2001) suggested that it is expansion of the bubbles in a vessel that will lead to rupture of the wall of that blood vessel, testing this in an in-vitro setting.
Table 48–12 Reversible and Irreversible Injury
| Reversible Changes |
| Irreversible Changes Resulting in Loss of Renal Tissue |
No group had been able to positively detect and validate acoustic cavitation within the kidney during SWL treatment until Bailey and associates (2005) created a passive cavitation detection system using two confocal spherical bowl PZT transducers. This device was used for coincidence detection of cavitation bubble emissions within a 2 × 2 × 2-mm sampling volume centered on F2 of a Dornier HM3 lithotripter. An ultrasound scan head targeted at this spot was used to image echogenicity in and around the sample volume. Signal (passive cavitation detection, hyperechoic spots) was intense in the urinary space during SWL treatment, and a signal was also seen in the renal cortex after only 1000 shocks. At that time a small fluid space was noted at the site of the parenchymal signal. These data suggest that once blood vessels have been ruptured and blood has collected in pools there is a greater potential for cavitation to occur. The pooling of blood provides a large fluid-filled space for cavitation bubbles to grow and collapse. In this model the accuracy of tissue targeting was confirmed by inducing a lesion with high-intensity focused ultrasound. Further evidence that cavitation plays a role in tissue injury comes from a study by Evan and associates (2002) in which the degree of tissue injury was compared between a standard rigid reflector and a pressure release reflector. The pressure release reflector generates a shockwave in which the negative tail precedes the positive peak, resulting in a suppression of cavitation activity. No injury was detected in the kidneys treated with the pressure release reflector; the standard rigid reflector induced the expected lesion.
Techniques to Optimize SWL Outcome
For all lithotripters the urologist has the ability to control a number of device parameters that may affect the ultimate treatment outcome (Table 48–13). These parameters include the acoustic output and focal volume that are employed, optimal coupling, the number of shockwaves administered, the rate at which they are dispensed, and the power or voltage that is used. In addition, other intraoperative factors that may affect stone breakage can be controlled, such as anesthetic technique.
Table 48–13 Factors That Induce the Degree of Renal Trauma Associated with Shockwave Lithotripsy
| Aggravating Factors |
| Mitigating Factors |
Although all lithotripters generate waveforms that are fundamentally similar, lithotripters may be distinguished from one another by the peak pressure and spatial extent of their acoustic field. The physics of acoustics dictate that the pressure field of a lithotripter is focused not at a particular point in space but rather is distributed over a volume of space. Most commonly, that focal zone is a cigar-shaped region, although the volume of that zone may differ greatly among devices. Recent in-vitro studies suggest that the focal width generated by a lithotripter affects stone breakage; a wider focal width has been reported to increase the likelihood of stone breakage (Sapozhnikov et al, 2007). Because the kidney tends to move, as a consequence of respiratory motion, the stone may move in and out of a narrow focal zone. Another potential drawback of a narrow focal zone is that less energy may be deposited into the stone. When the focal zone is narrower than the stone being treated, the tensile stress inside of a stone is reduced; for the stone to be subjected to the full force of shear stress, the outer surface of the stone must be subjected to high-pressure shockwave energy.
The first generation lithotripter was a water bath design that was a large, stationary machine. The present generation of lithotripters have dry treatment heads, which make them smaller and more easily transportable. However, they require a coupling medium, such as gel or oil, to join the patient to the device. Optimal coupling permits the efficient transfer of energy from the lithotripter to the patient; poor coupling will reduce stone breakage. Most commonly, energy transfer through a coupling medium is attenuated by air pockets in the coupling interface itself. Decoupling and recoupling, which may occur during repositioning of a patient during SWL, can generate large volume air pockets in the coupling medium. Such air pockets can have a dramatic effect on treatment efficacy: air pockets of just 2% of the coupling interface reduce breakage by 20% to 40% (Pishchalnikov et al, 2006; Neucks et al, 2008). Although there is no way to monitor coupling during treatment, simple steps can minimize the likelihood of air pockets developing. Dispensing gel from a squirt bottle and rubbing the gel by had to cover the treatment head and skin degrades the coupling interface. Improved coupling can be achieved by delivering a large volume of gel as a mound dispensed from the stock jug and allowing the gel to spread upon contact between the treatment head and the skin.
During an SWL treatment session the urologist can directly control the rate at which shockwaves are delivered and the number of shockwaves dispensed. In a recent literature review and meta-analysis of randomized control trials evaluating different shockwave delivery rates, a rate of 60 shocks per minute was found to break stones more effectively than 120 shocks per minute (Semins et al, 2008). Cavitation is thought to play a role in this effect, because the dynamic bubbles are given a longer time interval to dissipate with a slower rate and therefore have less of a shielding effect and energy draw from subsequent shocks. The disadvantage of a slow rate is, of course, a longer treatment time, particularly if the number of shockwaves being delivered is predetermined. However, slowing the rate has also been shown to be protective of kidney vasculature (Evan et al, 2007). The lowest number of shockwaves possible should be used to reduce renal injury, but this number is generally predetermined and many patients are likely to be overtreated. Risks and benefits need to be weighed regarding possible overtreatment versus need for further procedures when deciding on shockwave number.
Another parameter urologists adjust is the energy setting on the machine. Increasing the power setting on most electromagnetic lithotripters actually narrows the focal zone, which, as discussed earlier, decreases stone breakage and may also increase the risk of renal injury and renal hematoma (Connors et al, 2000). In the past several years, studies have shown that “ramping up” the energy of the lithotripter can be protective of renal injury (Fig. 48–30). Lesion size is decreased after pretreatment with low-energy shockwaves (100 to 2000 at 12 kV followed by 24 kV) (Willis et al, 2006). Interestingly, Connors and coworkers (2009) showed in a pig model that the voltage that is initiated is less important than the actual ramping up, as pretreatment groups with 100 shockwaves at 18 kV or 24 kV both had significantly smaller lesions than just treating with 2000 shockwaves at 24 kV without pretreatment. The reduction in renal injury is thought to be secondary to vasoconstriction because the same beneficial effect was blocked when dopamine was administered (Willis et al, 2006). In addition, ramping up the voltage has been shown to result in better stone breakage when using the same total shockwave energy (Zhou et al, 2004). With regard to voltage parameters, the technique of ramping up appears to both improve stone breakage and reduce tissue injury.
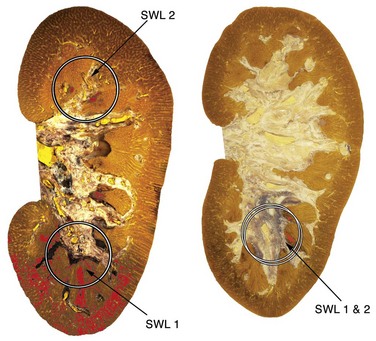
Figure 48–30 On the left is a coronal section of a kidney from an animal treated with 2000 shocks at 24 kV first to the lower pole (shockwave lithotripsy [SWL] 1) and then an additional 2000 shocks at 24 kV to the upper pole (SWL 2) of the same kidney. The typical lesion (in red) is seen at the lower pole; however, a greatly reduced lesion is seen on the upper pole. These data suggested that a pretreatment protocol might reduce the lesion induced by a clinical dose of shockwaves. At right, lesion size is shown in an animal first treated at the lower pole with 500 shocks at 12 kV (SWL 1) and then treated again at the lower pole with 2000 shocks at 24 kV (SWL 2). A greatly reduced lesion is also noted for this protocol.
The original lithotripter, the Dornier HM3, required a general anesthetic for treatment. However, later generations of lithotripters were developed for treatment to be performed without anesthesia. To minimize treatment discomfort the lithotripters were designed with wider aperture, which will spread the acoustic field across a broader area of the patient’s skin, reducing skin surface pain. However, this wider aperture resulted in a narrow focal zone, which had a deleterious effect on stone breakage. Interestingly, the higher pressures used with these newer machines result in higher adverse event rates as well. The effect of respiratory motion, as described previously, further hampers stone targeting and acts to reduce stone breakage rates.
To reduce stone motion, urologists can perform SWL with general anesthesia, which will control the patient’s respiratory rate and volume. Two clinical studies compared the outcome of SWL performed with intravenous sedation and SWL performed with general endotracheal anesthesia. General anesthesia yielded significantly better outcomes: 78% to 87% stone-free rates versus 51% to 55% with intravenous sedation (Eichel et al, 2001; Sorensen et al, 2002). Because general anesthesia is associated with superior outcomes this may be the anesthesia of choice for SWL, unless contraindicated for medical reasons.
Key Points: Shockwave Lithotripsy
Percutaneous Nephrolithotomy
Fernstrom and Johansson first reported the technique of creating a percutaneous track specifically to remove a stone in 1976. Subsequent reports have established PNL as a routinely used technique to treat patients with large or otherwise complex calculi (Alken et al, 1981; Wickham and Kellett, 1981; Segura et al, 1982; Clayman et al, 1984). Advances in surgical technique and technology have enabled the continuous evolution of PNL, allowing the urologist to remove calculi percutaneously with increasing efficiency. Because the percutaneous approach to stone removal is superior to the open approach in terms of morbidity, convalescence, and cost, PNL has replaced open surgical removal of large or complex calculi at most institutions (Matlaga and Assimos, 2002). Here the specific aspects of percutaneous techniques as they relate to stone removal are delineated.
Preparation of the Patient
The initial evaluation of the patient who is being considered for PNL should be a complete history and physical examination. A complete medical history will identify those patients with an absolute contraindication to PNL, such as uncorrected coagulopathy, as well as those with an active, untreated urinary tract infection. The placement of a percutaneous nephrostomy drain, without manipulation of the calculus, may be an appropriate therapy if the stone is associated with obstruction of the renal unit and sepsis. If it is medically feasible, aspirin and other antiplatelet medications should be discontinued 7 days before the date of surgery (Mak and Amoroso, 2003).
Preoperative laboratory evaluation of patients scheduled for PNL should include a complete blood cell count as well as serum electrolyte determinations and renal function tests. Martin and colleagues (2000) have reported that it is unnecessary to obtain screening coagulation studies before PNL for an otherwise healthy patient. Urine culture is mandatory for all patients who undergo PNL; perioperative antibiotics can be appropriately tailored to culture-specific organisms. Typing and screening of the patient’s blood should be performed, although preoperative crossmatching is usually not necessary.
Historically, it has been viewed as mandatory to evaluate the patient’s collecting system by either intravenous urography or retrograde pyelography. Recently, as more patients are initially evaluated with helical CT, it is no longer mandatory to have these studies at the preoperative evaluation (Park and Pearle, 2006). In most cases, the decision to perform PNL may be based on the stone burden displayed on the CT images. The main advantage of CT is the ability to assess the spatial relationship of the kidney relative to the stone and that of the kidney in relation to adjacent peritoneal and retroperitoneal structures. Retrorenal colon has been reported to be present in less than 1% of all patients, but its incidence may be higher in those who have undergone jejunoileal bypass, those in a nursing home, or those with spinal cord injury (Sherman et al, 1985). These patients may benefit from initial CT. Patients with ectopic kidneys, both congenital and iatrogenic (e.g., due to renal allograft, autotransplantation), as well as patients with dysmorphic body habitus due to congenital malformations such as spinal dysraphism may also benefit from cross-sectional imaging before PNL; intra-abdominal structures, such as the bowel, may be located between the skin and the renal access point. A KUB radiograph may be obtained immediately before the procedure to verify stone location. Retrograde pyelography can be performed at the time of the surgical procedure, acquiring information about calyceal anatomy that may aid in selecting the targeted puncture site. However, for certain patients, such as those with calyceal diverticula, for whom the surgical approach is affected by the diverticulum’s relationship to the collecting system, intravenous or retrograde pyelography may be required at the time of initial evaluation. Radionuclide scanning may be necessary in select patients, particularly those harboring staghorn calculi, to evaluate differential renal function.
Antibiotics
Although there are no prospective, randomized controlled trials confirming the need for antibiotic prophylaxis during PNL, it is generally accepted that antibiotic prophylaxis will reduce infectious complications. The antimicrobial of choice is a first- or second-generation cephalosporin or an aminoglycoside with metronidazole or clindamycin; ampicillin/sulbactam or a fluoroquinolone is recommended as an alternative. Mariappan and associates (2005) have reported a prospective controlled study that also found that oral ciprofloxacin 1 week before PNL significantly reduced the risk of postoperative urosepsis. Importantly, urinary calculi may harbor bacteria even though bacteriuria is only intermittently present. This is particularly true in the patient who has been taking antibiotics in the past. For those patients who do have preoperative bacteriuria, stone cultures produced bacteria in 77% of cases in a series reported by Larsen and associates (1986). The most frequently identified organisms were Proteus mirabilis, Escherichia coli, Klebsiella species, Pseudomonas species, Enterococcus species, and Enterobacter species. However, sterile urine does not preclude postoperative bacteriuria, because Charton and colleagues (1986) reported a 35% incidence of bacteriuria after PNL among patients with sterile preoperative urine culture specimens in whom prophylactic antibiotic therapy was not used. Mariappan and associates (2005) have also reported that the best correlate with post-PNL sepsis is stone culture or renal pelvic urine culture, not bladder urine culture. The fragmentation of stones, despite sterile urine, may release preformed bacterial endotoxins and viable bacteria that place the patient at risk for septic complications (Scherz and Parsons, 1987; McAleer et al, 2002, 2003; Paterson et al, 2003). Therefore, patients who have radiographic or clinical features suggestive of struvite or in whom infection is suspected should receive broad-spectrum antibiotics before surgery to reduce the risk of sepsis. Antibiotic treatment may also reduce bleeding secondary to inflammation and friability of renal parenchyma. Approximately one third of patients with an indwelling ureteral stent will, despite sterile urine on a preoperative analysis, be colonized with bacteria; Enterococcus and Staphylococcus epidermidis are the most frequent offending organisms (Reid et al, 1992; Lifshitz et al, 1999b). For patients with indwelling stents, then, a course of antibiotic prophylaxis, particularly for gram-positive organisms, may be beneficial before instrumentation.
Anesthesia
PNL can be performed after the administration of general, epidural, or local anesthesia. Local anesthesia, usually in combination with intravenous sedatives and analgesics, has been reported in a number of centers (Clayman et al, 1983; Hulbert et al, 1986; Preminger et al, 1986; Ohlsen and Kinn, 1993). Local anesthesia may be an option when general anesthesia is contraindicated. A local anesthetic, such as lidocaine, can be delivered into the access tract by use of an 8.3-Fr anesthetic injection catheter with multiple side holes or with a dual-lumen ureteral access catheter (Dalela et al, 2004). Regional anesthesia (e.g., epidural, spinal) can be used for percutaneous procedures, but several problems may be associated with these regional anesthetic techniques. First, a relatively high block is necessary to eliminate all renal pain. Second, distention of the renal pelvis during PNL may cause a vasovagal reaction that is not always prevented by regional anesthesia (Grasso and Taylor, 1997). General anesthesia is usually preferred when a more lengthy procedure is planned because it is the best means of protecting the airway when patients are in a prone position. In cases in which upper pole puncture is contemplated, general anesthesia should be used because it permits control of respiratory movements, which is essential to minimize the risk of pulmonary complications. A close relationship between the surgeon and the anesthesia team is essential to optimize the outcome of a PNL procedure. The anesthesiologist should be aware that pulmonary injuries, including hydrothorax and pneumothorax, can occur during PNL; to that end, the anesthesiologist should monitor airway pressures, end-tidal carbon dioxide levels, and oxygen saturation and should auscultate the lungs frequently. Acute anemia due to blood loss or dilution may also occur, emphasizing the need for frequent hemodynamic assessments. Because of the large amounts of fluids administered to the patient during nephroscopy there is a potential risk for hypothermia, a disorder associated with an increased risk of morbid cardiac events. Warming of irrigation fluids as well as patient warming devices may attenuate this risk.
The fundamental techniques of gaining and maintaining are reviewed in Chapter 47.
Stone Removal
After the nephrostomy access has been appropriately dilated and the Amplatz sheath positioned the urologist can proceed with stone removal by endoscopic techniques. In the early days of PNL several authors reported the successful extraction of renal calculi with Randall forceps (modified to allow passage over a guide wire) or stone baskets under only fluoroscopic, not visual, guidance (Castaneda-Zuniga et al, 1982; Pollack and Banner, 1984). However, fluoroscopically guided stone removal is no longer recommended because it is not as safe or as efficient as the removal of calculi under direct vision.
Physiologic solutions should be used for irrigation during PNL to minimize the risk of dilutional hyponatremia in the event of large-volume extravasation (Carson, 1986). The height of the irrigant should be maintained at 80 cm or less above the patient to minimize intrapelvic pressure and to prevent fluid absorption through pyelovenous backflow (Miller and Whitfield, 1985). The use of an Amplatz working sheath also prevents elevated intrapelvic pressures. Rigid nephroscopy is performed initially, and stones up to 1 cm in diameter can be grasped with rigid graspers or stone baskets and extracted intact through the 30-Fr Amplatz sheath. Stones larger than 1 cm require fragmentation before extraction. Several intracorporeal lithotripsy techniques are available.
Rigid nephroscopy is the preferred method for stone removal; however, only the simplest intrarenal collecting systems can be completely inspected with a rigid nephroscope through a single access. Therefore, flexible nephroscopy should be used during every PNL to survey the entire intrarenal collecting system for residual stone fragments. Pressurization of irrigation fluid (to 300 mm Hg) during flexible nephroscopy is necessary to adequately distend the collecting system and to improve visualization (the use of an Amplatz sheath is mandatory when pressurized irrigation is used). The entire collecting system should be examined systematically, including the proximal ureter. Injection of contrast material through the flexible nephroscope is helpful in maintaining orientation and verifying that each calyx has been inspected. Small stone fragments can be removed with a stone basket passed through the flexible instrument, and larger stones can be fragmented with laser or EHL. Alternatively, fragments may be flushed or manipulated into the renal pelvis with a combination of high-pressure irrigant and a floppy-tipped J wire, where they may be retrieved more easily with rigid instruments. The goal of PNL is complete or nearly complete clearance of stone material at the time of the primary procedure, which greatly simplifies secondary procedures, if necessary. Furosemide is administered intravenously when the nephrostomy tube is placed at the conclusion of PNL to promote and to maintain diuresis.
Special Situations
Calyceal Diverticula
Calyceal diverticula are nonsecretory, transitional cell epithelium-lined cystic cavities within the renal parenchyma. A narrow communication with the pelvicalyceal system almost always exists. The incidence of calyceal diverticula diagnosed on routine intravenous pyelography ranges from 0.21% to 0.45% (Hulbert et al, 1986). Calculi have been reported in 9.5% to 50% of calyceal diverticula (Jones et al, 1991b). Percutaneous access into stone-bearing diverticula poses several unique problems. Direct puncture is often difficult because of the small size of the cavity and the frequent occurrence of calyceal diverticula in the upper pole of the kidney. After successful puncture is achieved, negotiation of a guide wire into the renal pelvis is often not possible. A similar situation can occur when a stone fills a calyx so completely that a guide wire cannot be passed through the infundibulum into the renal pelvis or in the rare case of infundibular stenosis. To overcome these difficulties, a special access technique is required.
Contrast media can be instilled through a ureteral catheter to localize the diverticulum. However, if the stones are visible on fluoroscopy it is often preferred to puncture directly onto the stone (Ohlsen and Kinn, 1993). Direct puncture into the diverticulum allows use of rigid instruments that provide superior visualization compared with flexible instruments that are used in an indirect approach. Optimal visualization is essential in trying to identify the communication between the diverticulum and the renal collecting system. Direct access is also beneficial because fulguration of the urothelium can easily be achieved by a resectoscope equipped with a roller-ball electrode. Jones and colleagues (1991b) reported that direct percutaneous access into the diverticulum could be established in all but 2 of 24 patients. Likewise, Shalhav and associates (1998) reported that in a group of 30 patients with calyceal diverticula, direct access was performed in 28 patients. When direct puncture fails, a neighboring calyx can be punctured and the diverticulum entered indirectly by perforating the wall of the diverticulum or by entering in a retrograde fashion through the diverticular neck (Hedelin et al, 1988). However, the results with an indirect approach are inferior (Jarrett and Smith, 1986).
Kim and associates (2005a) have described a single-stage technique for the treatment of patients with calculi residing in a calyceal diverticulum (Fig. 48–31). Once the diverticulum is punctured, a 0.035-inch movable core J wire is coiled within the diverticulum. It is important to ensure that not only the floppy tip of the wire but also the solid core is coiled within the diverticulum, so that sufficient stabilization is provided for proper placement of coaxial dilators. A second 0.035-inch movable core J wire serving as a safety wire is then passed through the 10-Fr sheath of the coaxial dilator. With two guide wires coiled within the diverticular lumen, balloon dilation of the track can be performed safely. Care should be taken to avoid perforation of the back wall of the diverticulum. Once the balloon dilator is inflated, the working sheath is passed over the balloon as close as possible to the diverticulum without advancing the balloon. In small diverticula, this results in the placement of the sheath outside the diverticulum. An 11-Fr alligator forceps is passed through the rigid nephroscope and used to follow the wire and gently spread renal parenchyma to allow entry into the calyceal diverticulum under direct vision. Stone material is extracted with grasping forceps or ultrasonic lithotripsy. Careful inspection of the urothelium with the rigid nephroscope is performed in an effort to identify a flattened renal papilla, which suggests an obstructed calyx rather than a diverticulum. The neck of the diverticulum is often difficult to identify because it can be diminutive. Methylene blue injected through the ureteral catheter can facilitate visualization of the ostium. Once a guidewire is passed into the renal pelvis, the neck of the diverticulum can be balloon dilated or incised.
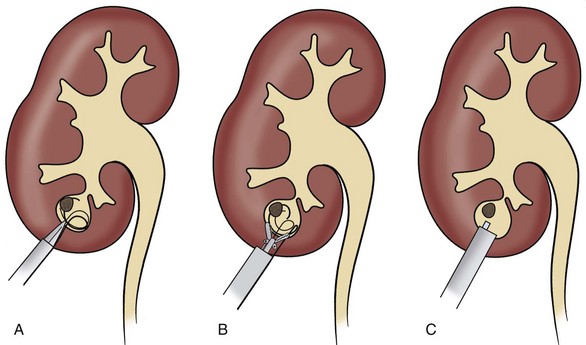
Figure 48–31 Access into small diverticula. A, Balloon dilator is advanced as far as possible without perforating the back wall of the diverticulum. The working sheath is placed just outside the diverticulum. B, Alligator forceps spread the parenchyma and allow advancement of the nephroscope under vision into the diverticulum. The working sheath is then advanced over the nephroscope and into the diverticulum (C).
Because calyceal diverticula are lined by a nonsecretory endothelium, most authors advocate fulguration at the time of PNL because this will ablate 76% to 100% of diverticula (Monga et al, 2000; Kim et al, 2005b). Auge and associates (2002b) and Turna and associates (2007) have also reported the results of long-term studies in which they found that PNL accompanied by ablation of the diverticulum is associated with superior stone-free rates. Alternatively, Hulbert and coworkers (1986) reported treating 10 patients with calyceal diverticula and suggested that trauma to the wall of the diverticulum caused by the dilation process is sufficient to ablate the diverticular lumen. In this series, a nephrostomy tube was left in place for 2 weeks. However, Donnellan and colleagues (1999) reported that treatment of 20 patients with calyceal diverticula by dilation or incision of the diverticular neck without fulguration resulted in complete ablation of the diverticulum in only 30% of patients, leading the authors to conclude that fulguration should be performed routinely to ensure diverticular ablation. Typically, after ablation of the diverticulum, a nephrostomy tube is placed for 48 to 72 hours.
Horseshoe Kidney
Horseshoe kidney is the most common congenital renal anomaly, with an estimated incidence of 1 in 400 and a male-to-female ratio of 2 : 1 (Jones et al, 1991a). The unique location and orientation of the horseshoe kidney are due to the incomplete cephalad migration and malrotation of the kidney, a consequence of the entrapment of the isthmus under the inferior mesenteric artery (Hohenfellner et al, 1992). The UPJ is commonly deformed owing to the high insertion of the ureter into a typically elongated renal pelvis. The course of the proximal ureter is similarly aberrant; it drapes ventrally over the renal symphysis, where it may be compressed by vessels supplying the lower pole and isthmus. Ureteral obstruction that may result from these anomalies can give rise to hydronephrosis, urinary stasis, sepsis, and calculi formation in up to 70% of patients (Jones et al, 1991a; Lampel et al, 1996).
In considering PNL in a horseshoe kidney, the characteristic lower and centrally oriented position of the kidney, the orientation of the collecting system, and the abnormal blood supply should be taken into account. Janetschek and Kunzel (1988) performed postmortem examinations of six horseshoe kidneys, in situ, and found normal renal arteries in all specimens. However, accessory arteries entering the renal hilum, aberrant polar and isthmus arteries originating from the aorta, and hypogastric and common iliac arteries were noted as well; all blood vessels, except a select few supplying the isthmus, entered the kidney from its ventromedial aspect. Therefore, a puncture of the dorsal or dorsolateral aspect of the kidney will be well away from major renal vessels.
Skoog and associates (1985) have reported an association between horseshoe kidney and retrorenal colon. A preoperative CT can be of assistance in determining the presence of a retrorenal colon as well as in defining the stone-bearing calyces. The lower pole calyces lie within a coronal plane, angled medially, and are seldom suitable for direct puncture (Al-Otaibi and Hosking, 1999). However, the upper pole calyces are more posterior and lateral and are often subcostal, providing a convenient and relatively safe route for PNL access. The standard site for PNL (inside the posterior axillary line just caudad to the 12th rib) is punctured, but the angle of the puncture is caudad rather than cephalad. Because most of the calyces of horseshoe kidneys point either dorsomedially or dorsolaterally, they are more favorably positioned for puncture than are normal renal units (Janetschek and Kunzel, 1988). Because of the malrotation of the kidney, the renal pelvis may be more anteriorly located and the length of the nephrostomy track may exceed the length of the rigid nephroscope, necessitating the use of flexible nephroscopy or multiple accesses. Flexible nephroscopy may also be required to gain access to the lower medial calyces, where stones are often found.
Raj and associates (2003) reported a multicenter analysis of 24 patients with calculi in a horseshoe kidney who underwent PNL. The overall stone-free rate was greater than 90%, most accesses were upper pole, and flexible nephroscopy was performed in almost all patients. The authors noted that the rigid nephroscope was rarely sufficient to remove the whole stone burden. Miller and associates (2008) have reported a similar series of patients undergoing PNL of horseshoe kidneys; upper pole access was used in all cases, as was flexible nephroscopy.
Transplantation and Pelvic Kidneys
Urolithiasis is uncommon in patients who have undergone renal transplantation, with a reported incidence of 0.5% to 3% (Harper et al, 1994; Shoskes et al, 1995; Del Pizzo and Sklar, 1999). Factors that may predispose transplant recipients to form calculi include metabolic abnormalities, foreign bodies (nonabsorbable suture material, forgotten stents), recurrent infection, and papillary necrosis. On occasion, calculi may have been present in the donor kidney (Pardalidis et al, 1994). Typical renal colic does not occur because the transplantation kidney and ureter are denervated; the presentation may instead resemble acute rejection or acute tubular necrosis (Harper et al, 1994; Rhee et al, 1999). Benoit and colleagues (1996) reported that in a series of 1500 transplantation patients, 12 (0.8%) were diagnosed with urinary calculi. Three patients presented with obstructive anuria, one patient presented with abdominal pain, and eight patients were asymptomatic and diagnosed by ultrasonography.
Renal allografts present a unique anatomic situation for PNL. The most common surgical anatomy is for the donor left kidney to be placed extraperitoneally into the recipient’s right iliac fossa; alternatively, the right kidney is transplanted in the left iliac fossa. In either case, the renal pelvis is located medially, requiring that the kidney be rotated 180 degrees on its axis. Thus, the posterior calyces point anteriorly and, consequently, an anterior approach to the kidney is similar to a posterior approach to native kidneys. In the usual percutaneous approach to a transplanted kidney the patient is placed in the lithotomy position, which allows simultaneous cystoscopic access to the bladder. A ureteral catheter is inserted for instillation of contrast material. Access is most safely established (i.e., avoiding intraperitoneal contents) into the lower pole with the skin puncture as caudal as possible. Percutaneous access to transplanted kidneys is actually facilitated by their superficial location. However, scar formation around the graft may make the initial needle puncture and tract dilation more difficult (Rhee et al, 1999). Once access is established, PNL can be performed by standard methods (Pardalidis et al, 1994). Del Pizzo and Sklar (1999) have reported the use of a mini-perc technique in 14 transplantation patients in whom access was established with the aid of intraoperative ultrasonography with a 16-Fr suprapubic peel-away introducer and sheath. No data are available to show an advantage of the mini-perc technique compared with the larger diameter working sheaths. Although delayed closure of the access site after catheter removal in immunosuppressed transplantation patients is a concern, most authors report that even nephrostomy tracts dilated to 30 French closed normally (Caldwell and Burns, 1988; Gedroyc et al, 1989; Del Pizzo and Sklar, 1999). Rifaioglu and associates (2008) have described that not all patients will require tract dilation to 30 French, and in some cases acceptable outcomes may be achieved with percutaneous flexible nephroscopy alone.
Patients with ectopic pelvic kidneys necessitate a different and more complicated approach for PNL as a result of their unique anatomy. The incidence of pelvic kidney has been estimated to range from 1 in 2200 to 1 in 3000 in autopsy series (Zafar and Lingeman, 1996). The pelvic kidney is retroperitoneal, posterior to the peritoneum and anterior to the sacrum. Interposing bowel loops between the kidney and the anterior abdominal wall prevent a direct puncture through the anterior abdominal wall. Eshghi and colleagues (1985) first described a laparoscopically assisted PNL technique for pelvic kidneys, a technique that has been further characterized by others (Zafar and Lingeman, 1996; Holman and Toth, 1998; Matlaga et al, 2006). Desai and associates (2007) have reported on PNL in pelvic ectopic kidneys using only ultrasound guidance. Although the stone-free rate was 100%, a bowel injury did occur in one patient. Consequently, if laparoscopy will not be used to assist guidance and reflect intervening organs away from the anticipated course of percutaneous access, percutaneous access could be obtained utilizing CT guidance (Fig. 48–32), as described by Matlaga and associates (2003).
Morbid Obesity
Patients suffering from morbid obesity present technical as well as anesthetic challenges during any surgical intervention (Freedman et al, 2002). Often, one or more comorbid factors complicate management of these patients. General anesthesia may be a special concern for patients in the prone position because of restricted respiratory capacity that may require higher ventilation pressures intraoperatively. Some studies suggest an increased postoperative morbidity in morbidly obese patients, mostly because of the higher incidence of wound infection and thromboembolic events. Pearle and coworkers (1998) first examined the issue of PNL among the obese when they evaluated a group of obese patients undergoing PNL. They found that performing PNL, with standard, prone techniques, was not associated with an increased risk of morbidity; in fact, complication and transfusion rates as well as the length of hospitalization was no different. Koo and colleagues (2004) have subsequently reported that the outcome of PNL is independent of the patient’s BMI. El-Assmy and colleagues (2006b) also reported that the stone-free outcome of PNL is also unrelated to BMI.
Positioning the morbidly obese patient for PNL can be difficult; and in particular for large patients, two surgical tables may need to be secured together (Hofmann and Stoller, 1992). Patient positioning should be closely inspected, and pressure points should be identified and padded. Hofmann and Stoller (1992) described a morbidly obese patient who developed acute rhabdomyolysis secondary to crush injuries from placement in the lateral approach, so if this technique is used care must be taken to minimize muscle crush injury. The major difficulty in performing PNL in the morbidly obese patient is the long distance from the skin to the collecting system, which may exceed the length of the working sheath or the length of the rigid nephroscope. Extralong Amplatz working sheaths (≥20 cm) and extralong rigid nephroscopes are now available that can overcome this challenge. Alternatively, the Amplatz sheath can be secured by a suture, allowing easy retrieval even when it migrates under the skin. On occasion, when the long Amplatz sheath is not sufficient to reach the kidney, an incision can be made through the subcutaneous tissue to the muscles of the flank, and the PNL track is created from the level of the muscle sheath (Curtis et al, 1997). Alternative instrumentation can also be employed: Giblin and colleagues (1995) described the successful use of a 30-Fr gynecologic laparoscope (with a working length of 27 cm) in patients in whom the SSD precluded the use of standard access sheaths and nephroscopes. Another possibility is to dilate the tract and place a nephrostomy tube for 1 week to let the tract mature. In some cases, maturation of the track allows the kidney to fall back posteriorly closer to the skin, allowing the use of standard nephroscopic instrumentation. Flexible nephroscopy can also be performed through the mature tract, reducing the necessity of rigid nephroscopy (Hofmann and Stoller, 1992). Liberal use of flexible nephroscopy in obese patients improves the stone-free rate and decreases the need for additional access (Pearle et al, 1998).
After stone removal, consideration should be given to the type of nephrostomy tube used. Tube displacement tends to occur more often in morbidly obese patients, so balloon-type catheters or reentry Malecot catheters may be preferable (Carson et al, 1988). Alternatively, if a Cope loop catheter is used, placement of a ureteral catheter should be considered to ensure that access to the kidney is not lost should the nephrostomy tube become displaced.
Bilateral Simultaneous Percutaneous Nephrolithotomy
Patients with large, bilateral stone burdens present a formidable challenge to the urologist; rendering these patients stone free can require separate staged procedures and multiple anesthetics. In 1987, Colon-Perez and associates (1987) first reported simultaneous bilateral PNL in a series of three patients. Since that time the procedure has evolved and several dictums have been established. It is recommended that either the side that is more symptomatic or the side that is more difficult be treated first. Patients may be positioned prone or with the treated side elevated.
Later series have expanded on this initial report. Most groups have reported that there is a marked advantage in terms of hospitalization and convalescence, suggesting that a simultaneous bilateral PNL provides the patient a less morbid, more rapid method of stone resolution than does staged PNL or a sandwich technique (Dushinski and Lingeman, 1997; Nadler et al, 1998; Holman et al, 2002). Silverstein and associates (2004) also reported that stone-free rates, blood loss per operation, and transfusion rates for simultaneous and staged bilateral PNL were similar. Furthermore, the reduced total operative time, hospital stay, and total blood loss along with the requirement for only one anesthesia session makes simultaneous bilateral PNL an attractive option for select patients. Desai and associates (2007) have issued a caveat that patients with exceptionally large stone burdens or complex intrarenal collecting system anatomy should not be selected for a simultaneous bilateral PNL. Bagrodia and associates (2009) performed an outcome cost analysis of simultaneous bilateral PNL, confirming the superior morbidity and convalescence of this approach.
Taking all of the evidence together, should a simultaneous bilateral approach be selected the second side should be pursued only if no significant bleeding is encountered on the first side, the first side did not take an undue amount of time, the patient is clinically stable, and the anesthesia team is agreeable.
Complications
Even for the most experienced urologist, major complications can still occur in up to 7% of patients undergoing PNL, and minor complications may be encountered in up to 25% of patients (Preminger et al, 2005). Hemorrhage is the most significant complication of PNL, with transfusion rates reported to be from less than 1% to 10%. Bleeding from an arteriovenous fistula or pseudoaneurysm that requires angiographic embolization occurs in less than 0.5% of patients. Other potential complications include sepsis (postoperative temperature above 38.5° C (101.3° F) is found in almost one fourth of patients undergoing PNL), adjacent organ injury (bowel, spleen), failed access, and perforation of renal pelvis and ureter. The need for open surgery is rare and mostly reported as part of early experience in various studies. The mortality rate of PNL is between 0.046% and 0.8% (Lang, 1987; Lee et al, 1987; Henriksson et al, 1989; Jones et al, 1991a; Lam et al, 1992b; Segura et al, 1994; Dyer et al, 1997; Matlaga et al, 2004). When supracostal puncture is performed, the risk of pneumothorax or pleural effusion requiring drainage may be higher (see Table 48–14). Finally, failure of equipment is an often ignored but significant potential complication. During initial experiences with PNL, concern was expressed about the extent of renal damage caused by the creation and dilation of the transparenchymal nephrostomy tract. However, although some scarring does occur along the percutaneous tract, there is little or no significant effect on renal function (Eshghi et al, 1989).
Because the kidney is an extremely vascular organ, some degree of bleeding occurs during every PNL. Significant bleeding usually requires cessation of the procedure because of impaired visualization. In most cases the source of hemorrhage is venous and placement of a nephrostomy tube is usually sufficient to control the bleeding. If bleeding persists despite the placement of a nephrostomy tube, clamping the tube for a time may facilitate the tamponade of any bleeding points. If these measures do not control the hemorrhage, a Kaye nephrostomy tamponade balloon catheter should be placed. The Kaye nephrostomy tube incorporates a low-pressure 12-mm balloon that may be left inflated for prolonged periods to tamponade bleeding from the nephrostomy tract (Kaye and Clayman, 1986). If bleeding persists despite placement of a Kaye catheter, immediate angiography should be performed to identify a possible arteriovenous fistula or false aneurysm. Angiography is both diagnostic and therapeutic, because arteriovenous fistulas and false aneurysms are best managed by embolization. In the rare event that bleeding cannot be controlled with angiography, partial nephrectomy may be required.
PNL can lead to some absorption of irrigation fluid; therefore, the use of physiologic irrigating solutions is mandatory. The amount of absorbed fluid depends mostly on the irrigant pressure and the length of the procedure. Larger amounts of fluid absorption may occur with extravasation of fluid due to collecting system perforation. Extravasation usually occurs into the retroperitoneal tissue and may be noted by medial displacement of the kidney during fluoroscopy. Minor perforations are common during PNL; premature termination of the procedure is usually not necessary if a low-pressure system (i.e., Amplatz sheath) is being used. However, with more significant perforations, termination of the procedure and nephrostomy drainage are advisable. Intraperitoneal extravasation is a less common but potentially more serious complication than retroperitoneal extravasation. Because the patient is prone, abdominal distention may be difficult to recognize, although the anesthesiologist will usually note a gradual rise in the patient’s diastolic blood pressure with consequent narrowing of the pulse pressure and increase in central venous pressure; in advanced cases, ventilation may become difficult because of increased abdominal pressure. Early recognition of major extravasation is crucial. Some authors have suggested that irrigant input and output should be monitored during PNL; and if a discrepancy of more than 500 mL is encountered, cessation of the procedure should be considered (Lee et al, 1986; Segura, 1993). However, a strict accounting of irrigating fluid balance is often not possible during PNL and should not be considered mandatory. Intraperitoneal extravasation may be treated by vigorous diuresis; alternatively, peritoneal drainage has been reported (Carson and Nesbitt, 1985).
When a supracostal puncture is performed, extravasation of irrigant into the pleural cavity may occur. The use of a working sheath tends to minimize extravasation into this space because intrarenal pressure remains low. The chest should be examined at the end of PNL procedures in which a supracostal puncture is used. Fluoroscopy with use of the C-arm is usually sufficient to examine for pneumothorax or hydrothorax (Ogan et al, 2003). If the surgeon has a high index of suspicion for a thoracic complication, a chest radiograph may be obtained postoperatively. If a greater than 10% pneumothorax or hydrothorax occurs, aspiration is generally sufficient because lung injury is extremely rare. Should the pneumothorax recur, a chest tube should be placed. Ogan and Pearle (2002) have described placement of a chest tube under fluoroscopic guidance at the time of the PNL.
Colonic injury is an unusual complication and is often diagnosed on postoperative nephrostogram, although passage of gas or feculent material through the nephrostomy track, intraoperative diarrhea, and hematochezia or peritonitis are all signs of a possible colonic perforation. Typically, the injury is retroperitoneal; thus, signs and symptoms of peritonitis are infrequent. If the perforation is extraperitoneal, management may be expectant, with placement of a ureteral catheter or double-J stent to decompress the collecting system and withdrawal of the nephrostomy tube from an intrarenal position to an intracolonic position to serve as a colostomy tube (Gerspach et al, 1997). The colostomy tube is left in place for a minimum of 7 days and is removed after a nephrostogram or a retrograde pyelogram shows no communication between the colon and the kidney (LeRoy et al, 1985; Wolf, 1998).
Ureteroscopic Management of Ureteral Stones
Basic ureteroscopic techniques and intracorporeal lithotripsy techniques have been reviewed elsewhere. Herein, issues of anesthesia and specific points of technique and complications of ureteral stone management are reviewed.
The increasing miniaturization of and improvements in the technology of ureteroscopy have greatly altered the anesthetic considerations associated with this procedure. Ureteroscopy was initially performed exclusively under general or regional anesthesia as a consequence of the large-caliber ureteroscopes that were available. However, as ureteroscopes have become more diminutive, intravenous sedation, or sedoanalgesia, has provided another practical anesthetic option for patients harboring ureteral stones. The short duration of modern sedoanalgesic agents allows rapid relief of pain, efficient titration of analgesia, and rapid recovery after the procedure. Ureteroscopy can be safely and efficiently performed under local or intravenous sedation, with success rates equivalent to those of patients undergoing general or regional anesthesia (Cybulski et al, 2004). Because most ureteral orifices will accommodate a 6- to 7-Fr device, immediate access to ureteral calculi with a 7-Fr flexible ureteroscope is possible. Furthermore, the small-diameter fibers of the holmium:YAG laser allow the immediate treatment of ureteral calculi.
The ability to fragment and remove calculi with a ureteroscope has dramatically advanced the urologist’s ability to render patients stone free with a single procedure. Early stone-free results with flexible ureteroscopy did not exceed those of in-situ SWL (Drach et al, 1986; Bagley, 1990; Frang et al, 1992; Mogensen and Andersen, 1994). However, as smaller ureteroscopes were introduced and the holmium:YAG laser became available, success rates for ureteroscopy have increased (Preminger et al, 2007). Although it is a more invasive technique than SWL, ureteroscopy with small, rigid or flexible endoscopes is the most efficient technique for treatment and removal of ureteral stones. Patients desiring a single procedure with maximal efficacy should be counseled on the advantages of a ureteroscopic approach.
When the ureteral orifice is too narrow to accommodate a ureteroscope, dilation may be accomplished with serial dilators, balloons, or even the ureteroscope itself. The anatomy of male patients may not allow a rigid ureteroscope to be easily passed above the iliac vessels, but a flexible ureteroscope can usually be advanced over a guidewire. The entire ureter can be more easily accessed with a rigid ureteroscope in female patients. Once the stone is visualized, fragmentation with the lithotrite of choice is performed. Complete fragmentation to a size less than that of the safety wire (0.035 inch) should allow passage of all fragments without sequelae. Alternatively, fragmentation to a size sufficient for extraction by a stone retrieval device achieves a stone-free state for the patient at the end of the procedure.
If significant ureteral edema or manipulation occurs, a post-ureteroscopy stent should be placed to prevent colic and obstruction. There have been two unique meta-analyses addressing this subject (Makarov et al, 2008; Nabi et al, 2007); both have found that for uncomplicated ureteroscopy, a ureteral stent may be safely omitted. If the clinical situation does not allow primary ureteroscopy (sepsis, inability to advance the ureteroscope, ureteral injury, equipment failure), a ureteral stent is placed and the problem corrected. Secondary or post-stent ureteroscopy has the advantage of working through a dilated ureter, often allowing the use of larger rigid ureteroscopes.
Steinstrasse
Steinstrasse, literally “street of stones,” describes the accumulation of stone fragments in the ureter obstructing the collecting system after SWL. This complication has been reported in 2% to 10% of SWL cases and directly correlates with increasing stone burden (Coptcoat et al, 1987; Weinerth et al, 1989; Sulaiman et al, 1999; Madbouly et al, 2002). Large stone burden, staghorn calculi, bilateral SWL, and preexisting ureteral obstruction are all known to be risk factors for steinstrasse (Weinerth et al, 1989). Steinstrasse may occur in as many as 41% of patients undergoing SWL monotherapy for staghorn calculi, although with the increasing use of PNL in the treatment of patients with large stones the incidence of steinstrasse has lessened (Coptcoat et al, 1987; Wirth et al, 1992).
The spectrum of presentation of patients suffering from steinstrasse is variable. Some patients may be entirely asymptomatic; others will present with the typical symptoms of renal colic, urinary tract infection, and renal insufficiency, with a time to presentation ranging from hours to months after SWL. More than 70% of steinstrasse occurs in the distal ureter (Coptcoat et al, 1987; Fedullo et al, 1988; Al-Awadi et al, 1999; Sulaiman et al, 1999).
The placement of a ureteral stent before SWL therapy may decrease the risk of steinstrasse. However, even when a ureteral stent is placed, steinstrasse may still occur. The management of patients suffering from steinstrasse depends on their clinical presentation. Patients with steinstrasse who are asymptomatic can often be observed initially; spontaneous stone clearance occurs in up to 60% to 80% of cases (Fedullo et al, 1988; Preminger et al, 1989; Kim et al, 1991). However, patients with bilateral obstruction, obstruction of a solitary kidney, severe refractory pain, or infected hydronephrosis should be managed with prompt urinary tract decompression by internal ureteral stenting, nephrostomy tube drainage, or ureteroscopic management. Large leading fragments unlikely to pass spontaneously and failure of expectant management are additional indications for intervention (Coptcoat et al, 1987; Weinerth et al, 1989).
When intervention is required, a minimally invasive approach is often successful. Coptcoat and associates (1988b) reported that percutaneous nephrostomy alone permitted passage of all stone fragments in 75% of cases. Dretler (1994) also recommended placement of a percutaneous nephrostomy tube for patients with pain or sepsis, reserving endoscopic intervention for afebrile patients with large leading fragments or after at least 4 weeks of observation. However, Weinerth and colleagues (1989) found that seven of nine patients initially treated with percutaneous nephrostomy eventually required ureteroscopic basket extraction or lithotripsy. The use of prophylactic antibiotics in expectantly managed cases is recommended, as are frequent radiologic and ultrasound examinations. Failure of steinstrasse to resolve within 3 to 4 weeks may necessitate intervention (Coptcoat et al, 1987).
Should patients require a ureteroscopic intervention, success rates have been reported to be good (Coptcoat et al, 1988a; Weinerth et al, 1989). However, caution is warranted in undertaking the retrograde endoscopic treatment of a patient with steinstrasse; the complication rate of ureteroscopy may be increased over that of percutaneous nephrostomy or SWL (Al-Awadi et al, 1999). Alternatively, SWL of the lead fragment of steinstrasse also has a high reported success rate and has been associated with a low rate of complications (Kim et al, 1991; Sulaiman et al, 1999). Finally, PNL, with or without concomitant ureteroscopy, can also be used to treat patients suffering from steinstrasse (Coptcoat et al, 1988b). PNL is particularly appealing if significant stone burden remains in the kidney, as is commonly the case. Open ureterolithotomy has been reported by multiple authors as a salvage procedure for steinstrasse but is rarely required in the modern endoscopic era (Kim et al, 1991; Al-Awadi et al, 1999; Sulaiman et al, 1999).
In summary, steinstrasse is a well-known complication of SWL, with most cases resolving spontaneously. When intervention is required, percutaneous nephrostomy alone has a success rate of more than 70%. Ureteroscopic intervention is definitive and predictable, with an immediate success rate approaching 100%. Additional SWL may be successful, but the results are less predictable than with ureteroscopy. PNL may be used as a salvage procedure, with open ureterolithotomy reserved for failure or for complications of less invasive modalities.
Complications
As modern ureteroscopes have become smaller and less traumatic, as safer intracorporeal lithotripters have become widely available, and as a better understanding of the technical principles of ureteroscopy has been developed, the number of complications arising from the management of ureteral stones has been steadily decreasing.
Fortunately, most of the complications caused by ureteral stones and their management respond favorably to simple drainage of urine with ureteral catheters or stents.
Perforation
As is true with most ureteroscopic complications, the incidence of ureteral perforation has decreased over time, as technology and technique continue to improve. In a series of ureteroscopic procedures reported in 1992, ureteral perforation was reported in approximately 15% of cases. More recent series report a perforation rate of 0% to 4%. A number of actions may result in a ureteral perforation; some of the more commonly encountered scenarios include the splitting of a ureter after balloon dilation or a traumatic injury due to forceful and misdirected manipulation of a stone. In some cases an intracorporeal device such as a lithotrite or electrocautery can cause full-thickness damage to the ureter. The incidence of ureteral perforation varies among the different intracorporeal lithotrites, with the highest incidence of ureteral perforation occurring with electrohydraulic lithotripsy. Baskets and grasping devices may also cause injury, because in the process of grasping a stone or lesion it is possible to inadvertently snare part of the ureter, which may result in a perforation. In addition, the pressurized irrigation can cause perforation or calyceal rupture.
Adherence to the tenets of safe ureteroscopy will minimize the likelihood of a ureteral perforation. Nonetheless, such a complication may still occur and it is important for the urologist to be familiar with the treatment of this event. When a ureteral perforation is recognized the ureteroscopic procedure should be terminated and a stent should be placed across the injury. The risk of perforation underscores the importance of using saline as an irrigant, to prevent electrolyte derangements due to fluid extravasation. In cases of a severe injury, with significant extravasation of fluid, a percutaneous nephrostomy drain may also be necessary. Urinoma can result from perforation as well and may need to be drained. Antibiotics should be given because of the risk of infected urine and abscess formation. In general, a stent should be left in place for approximately 4 weeks after injury. Subsequent imaging after ureteral stent removal is mandatory, to evaluate for a proper healing and adequate drainage.
Schuster and associates (2002) found a significant association of ureteral perforation with increased operative time. This suggests that if the procedure is difficult and not progressing it is wise for the urologist to stop, place a stent, and plan for a staged procedure. The best prevention strategies are similar to the principles discussed earlier, including controlled maneuvers without force, knowing the margin of safety of the equipment being used, and always using both a working and safety guidewire.
Stricture
The development of a postoperative ureteral stricture is one of the more serious complications that may occur after ureteroscopy. Approximately 2 decades ago, the reported incidence of ureteral stricture after ureteroscopy was as high as 10%. More recently, however, the incidence of a postoperative stricture is reported to be much lower at 0.5% to 4%. It is likely that the improvements in surgical technology and technique are responsible for this dramatic reduction.
Although the cause of a ureteral stricture is likely multifactorial, certain factors have been reported to increase the patient’s risk for stricture development. Roberts and coworkers (1998) reported a 24% stricture rate for stones that had been impacted an average of 11 months. Impacted stones are defined by the inability to pass a wire or catheter beyond the stone or stones that have been present and not moved for 2 months or more. Meng and coworkers (2003) have reported that a ureteral perforation may also increase the risk of stricture formation, finding that strictures occurred in 5.9% of patients who suffered a ureteral perforation during ureteroscopy. It may be that an inflammatory response that results in devascularization and ischemia promote this process, because such local changes can result in a cicatrization of the ureter. Patients with a history of ureteral surgery, pelvic radiation, and impacted stones are also at greater risk secondary to altered blood flow and poor healing. Devascularization injury can also result in ureteral necrosis, which necessitates open or laparoscopic repair. However, some patients develop a ureteral stricture in the presence of no intraoperative misadventures, suggesting that there is much about this process that remains to be elucidated.
To reduce the risk of stricture, care should be taken during all parts of the procedure, because the traumas that may increase the risk of this event are many and varied. Overly aggressive manipulation of a ureteroscope across a narrow segment of ureter, as well as trauma or perforation from injudicious manipulation of intracorporeal devices or lithotrites may all increase the risk of stricture. Because of the reported occurrence of a postoperative ureteral stricture even after an uncomplicated ureteroscopy, it is recommended that all patients undergo postoperative imaging after ureteroscopic instrumentation to ensure that such a complication is recognized. Although most patients with a stricture will be symptomatic, some will not be, so imaging is necessary in all patients to exclude such cases of silent obstruction because there is a risk of renal loss if it is not identified. In one study 23% of those with postoperative obstruction, 2.9% of the total cohort, had silent obstruction (Weizer et al, 2002).
The management of a postureteroscopic ureteral stricture will depend primarily on its length and location, although other factors such as the time elapsed since injury, nature of trauma, and patient-specific parameters merit consideration. For many short strictures, endoscopic management may be appropriate, and incision and dilation may yield a good result. For longer strictures, however, ureteral reconstruction can be more complex, requiring either open or laparoscopic repair. The types of reconstruction available are essentially the same as those for ureteral avulsion. If a stricture is short but has failed endoureterotomy, a ureteroureterostomy may be possible after the affected portion has been resected.
Submucosal Stone/Lost Stone
The submucosal stone and the lost stone represent two points on a continuum of iatrogenic displacement of a ureteral calculus into the wall of the ureter. Extrusion of a urinary calculus may occur in up to 2% of ureteroscopic procedures. When the stone only migrates to the submucosa, a problematic complication can develop, because removal of such stones is difficult. If submucosal stones are encountered, laser excision followed by ureteral stent placement is recommended. Submucosal stones are of concern, because they can increase the risk of ureteral stricture formation.
Complete extrusion of a calculus, also known as a “lost stone,” can occur in the setting of a ureteral perforation. In most cases, if the fragment is completely outside the collecting system it can be left in place. Attempts to retrieve the stone may exacerbate the injury as well as increase the risk of significant irrigant extravasation. When an extruded stone is recognized, the procedure should be terminated and a ureteral stent placed. Antibiotics should be administered to prevent the theoretical risk of abscess formation, although such a complication would be rare. One of the most serious sequelae of such an event is the later development of a ureteral stricture; for this reason patients who suffer this complication should undergo postoperative imaging. Additionally, patients with an extruded stone should also undergo imaging to confirm the location of the stone and they should be informed of this finding. It is possible that, in the future, the “lost” stone could be confused for a ureteral stone, and it is important for the patient to be aware that such a situation exists.
Avulsion
Perhaps the most catastrophic complication that can occur during a ureteroscopic procedure is avulsion of the ureter. Fortunately, such a complication is a rare occurrence, reported in less than 1% of all cases; in fact, a recent review of over 1000 patients undergoing ureteroscopy reported no episodes at all of ureteral avulsion (Krambeck et al, 2006b). Ureteral avulsion generally occurs as a consequence of overly forceful manipulation of a large or impacted calculus. It has been reported that the proximal one third of the ureter may be at greatest risk for avulsion, because it is the portion of the ureter that has the least muscular tissue support. A ureteral avulsion is usually diagnosed when a portion of the ureter is withdrawn from the patient, along with the stone and basket or grasper.
There are a number of maneuvers the urologist can undertake to avoid a ureteral avulsion. Blind basketing, the removal of a ureteral calculus without the aid of endoscopy, may increase the risk of ureteral avulsion and should never be considered an appropriate method of stone extraction. In fact, even endoscopically guided basketing should be reserved for small stones only. In general, a safe ureteroscopic procedure relies on the placement of both safety and working guidewires. Before engaging a stone in a basket or grasping device, it should be endoscopically evaluated, to determine if it is of a size that would be likely to be extracted out of the ureter. When the stone is engaged in the basket or grasper, the stone and device should both be kept under direct vision as they are extracted, so that the size of the stone can be continually compared with the size of the ureteral lumen. If the stone appears to be too large to be removed intact, it should be fragmented into smaller pieces that may either pass spontaneously or be extracted. If a stone too large to pass through the ureter is inadvertently engaged with a basket or grasping device, it should be released or replaced more proximally. Should it not be possible to release the stone the basket should be disassembled and the ureteroscope passed alongside of the basket and the entrapped stone fragmented in situ. The use of a grasping device, rather than a basket, may simplify the release of an entrapped stone. The great benefit of having a safety wire in place is that should a stone become entrapped in the ureter a ureteral stent may be placed that will passively dilate the upper urinary tract and perhaps permit a more straightforward procedure at a later date.
Should a ureteral avulsion occur, a reasoned and considered approach to the management of the affected patient is advisable. Although it may be tempting to perform an immediate primary repair at the time of injury, in general a delayed repair is recommended. The patient should undergo immediate diversion of the renal unit with the placement of a percutaneous nephrostomy drain. In some cases, a urinoma may develop as a consequence of urinary extravasation; these collections are generally amenable to percutaneous drainage. Subsequent ureteral reconstruction techniques depend on the location of the injury and the amount of viable ureter that remains. For extensive injuries the treatment options are generally limited to ileal interposition (ileal ureter) or renal autotransplantation. Although a transureteroureterostomy may be a replacement option for some patients who suffer a ureteral injury, this repair technique is contraindicated in stone formers. For a more distal ureteral injury, a ureteral reimplantation with either a psoas hitch or Boari flap may also be successful. Nephrectomy has been reported to be an option for these patients as well; however, given the recurrent nature of stone disease and the fact that stone formers may be at increased risk for hypertension and diabetes, this approach is controversial.
Ureteroscopic Management of Intrarenal Calculi
Huffman and associates (1983) first described the removal of large renal pelvic calculi with a rigid rod-lens ureteroscope and an ultrasonic lithotripter. However, rigid ureteroscopy is generally limited to stones in ureteral locations, because it is usually impossible to negotiate a rigid ureteroscope into the renal collecting system. In the decades since this initial report of ureteroscopic treatment of renal calculi, the therapeutic potential of ureteroscopy has been greatly advanced, primarily owing to the popularization of flexible, fiberoptic ureteroscopy. Over the previous 2 decades, advances in fiberoptic technology have facilitated the development of practical flexible ureteroscopes; concomitant improvements in flexible intracorporeal lithotripters as well as intracorporeal grasping devices have promoted the use of flexible ureteroscopes for the treatment of renal calculi with a high degree of success.
Fuchs and Fuchs (1990) reported the first large series of patients with renal calculi who were treated by flexible ureteroscopy. In all cases, flexible ureteroscopy was performed following a 1- to 2-week period of ureteral stent placement. The overall stone-free rate was 87%, and the only complications reported were two cases of sepsis. The subsequent introduction of more miniaturized, flexible, actively deflectable ureteroscopes (7.5 Fr) has permitted the majority of ureteroscopic procedures to be performed without routine ureteral dilation. In a large series of patients with renal stones who underwent treatment with holmium laser lithotripsy, ureteral dilation was necessary in only 185 (31%) (Sofer and Denstedt, 2000). Numerous baskets and graspers now allow full deflection of flexible ureteroscopes, facilitating treatment of often difficult-to-access lower pole stones (Lukasewycz et al, 2004; Pearle et al, 2005).
As ureteroscopic technology has advanced, and as surgeons have gained increasing comfort with this technique, this intervention has become an increasingly common treatment for patients with renal calculi. Single session success rates, generally defined as successful fragmentation with complete stone clearance or the presence of “clinically insignificant” residual fragments, are reported to be in the 70% to 80% range (Mariani, 2007; Ricchiuti et al, 2007; Breda et al, 2008). Even greater success rates may be achieved after a secondary procedure. Complication rates are low, with fever and urinary tract infection being the most commonly encountered adverse events. Even the treatment of staghorn calculi has been described, although this is not a widely used technique (Mariani, 2007)
Technique
Proximal ureteral and intrarenal calculi can be accessed with actively deflectable, flexible ureteroscopes. Although, historically, a ureteral stent was left indwelling before ureterorenoscopy, this maneuver is currently necessary only when difficulty is encountered introducing the flexible ureteroscope into the ureter. If necessary, the stent is left for 2 to 4 weeks before the procedure (Erhard et al, 1996). The use of small-diameter ureteroscopes minimizes the necessity of ureteral dilation for stone access and may decrease associated morbidity. In cases of multiple stones or large stone burden requiring multiple passages of a basket, a ureteral access sheath may facilitate stone removal. A comparative study of ureteroscopic lithotripsy with or without access sheath showed that operative time was significantly reduced by use of the access sheath, despite a greater mean stone burden in the access sheath group (Kourambas et al, 2001).
When a retrograde ureteroscopic approach is used to treat patients with intrarenal calculi, two wires are placed initially. The flexible ureteroscope is passed over one working wire in a monorail fashion. Saline is used for irrigation. When an implement is present within the working channel, simple gravity irrigation is inadequate and pressurized irrigation is required. The holmium:YAG laser lithotripter is used in almost all cases, and the laser energy required usually does not exceed 1 J and 15 Hz. Stones in lower pole calyces can be treated in situ or moved, with flexible graspers or a basket, into a position that allows better visualization. A head-down patient position with the ipsilateral flank elevated may help with stone and fragment visualization because fragments tend to migrate superiorly and are thus more easily localized during treatment. When access into the lower pole is difficult, prone positioning of the patient with the head down 20 degrees has been shown to provide the broadest angle of entry to the lower pole infundibulum (Bercowsky et al, 1999). The goal of holmium laser lithotripsy is to reduce the stone to fine dust and to small fragments 2 mm or less in diameter. If the stone is large, the collecting system may often become lined with fine dust and debris, which can obscure residual stones. Furthermore, poor visualization may lead to perforation. In such cases, either the irrigant in the intrarenal collecting system may be aspirated through the ureteroscope or a ureteral stent can be placed and the situation approached in a staged fashion.
Fragmentation of stones in situ in the lower pole can be challenging. If the lower pole infundibulum is accommodating, the most straightforward way to treat the stone is to engage it in a nitinol basket and displace it to the renal pelvis or an upper pole calyx. In this way it is generally a straight passage of the scope, with minimal deflection of the tip, which will simplify ureteroscopic laser lithotripsy. Residual fragments also should be more likely to evacuate spontaneously from the kidney.
Ureteral Access Sheath
The utilization of a ureteral access sheath as an adjunct to ureteroscopy was first reported by Takayasu and Aso in 1974 as a means to simplify access to the intrarenal collecting system. It was not until more than 2 decades later that the ureteral access sheath was rediscovered and refined, simplifying the deployment and safety of these devices. The present generation of ureteral access sheaths consists of a hydrophilic outer coating, as well as a tapered transition from obturator to sheath, which facilitates their retrograde placement. The walls of the sheaths are designed not only for a slim profile but also for strength and often are reinforced so as to resist kinking.
Kourambas and associates (2001) have reported that the ureteral access sheath can be successfully deployed in over 90% of attempted placements. In this randomized, controlled study these authors also found that the use of an access sheath decreased operating room time, simplified reentry of the ureter, and, likely as a consequence of these two points, was associated with decreased operating room costs. In a larger, retrospective study from the same institution, L’Esperance and associates (2005) reported on a series of 173 patients undergoing ureteroscopy with an access sheath and compared these outcomes to those of 83 patients undergoing ureteroscopy without an access sheath. They found that the stone-free rate was significantly higher when the access sheath was employed (79% vs. 67%; P = .042). Portis and associates (2006) also reported on a prospective series of patients undergoing ureteroscopy with a ureteral access sheath and commented that the sheath facilitated the active extraction of all stone fragments. Finally, ureteral access sheaths may have an added benefit when treating infection-related calculi, because the use of a sheath has been reported to maintain low intrapelvic pressures during ureteroscopy (Rehman et al, 2003). Additionally, as irrigating fluid drains through the access sheath external to the patient, the need for periodic emptying of the patient’s bladder during a prolonged procedure is eliminated.
Open Stone Surgery
Historically, most patients with symptomatic upper urinary tract calculi underwent open surgical lithotomy. Those patients with a small to moderate stone burden typically underwent pyelolithotomy, radial nephrolithotomy, or ureterolithotomy. For those patients harboring staghorn calculi, more extensive procedures were required, including anatrophic nephrolithotomy, extended pyelolithotomy combined with radial lithotomies, and bench surgery with autotransplantation. However, the introduction of SWL and the development of endourologic techniques for stone removal have dramatically diminished the role of open stone surgery, especially for stone removal procedures, and open surgery is now one of the least common treatments of patients harboring upper urinary tract calculi. Matlaga and Assimos (2002) reported that of 986 surgical procedures for stone removal performed at their institution between 1998 and 2001, 0.7% were open surgical procedures. Others have reported similar findings (Table 48–14), with an incidence of open stone surgery ranging from 0.3% to 5.4% (Assimos et al, 1989; Segura, 1990; Kane et al, 1995; Paik et al, 1998).
Renal Calculi
Minimally invasive techniques have clear advantages over open surgical techniques for patients harboring small to moderate burdens in otherwise normal kidneys. In 1985, Brannen and associates retrospectively compared PNL and open surgery for the treatment of patients with renal and proximal ureteral stones. Although the overall stone-free rate was similar, those patients treated by PNL experienced a shorter hospital stay, a lower narcotic requirement, and a shorter recovery period. Preminger and associates (1985) compared 88 patients undergoing PNL with 41 patients undergoing open stone surgery and found that PNL was associated with lower postoperative morbidity, more rapid convalescence, greater satisfaction of the patient, and reduced hospital costs for stones smaller than 2.5 cm. Brown and associates (1986) have also demonstrated that PNL is more cost effective than open stone surgery because of its reduced morbidity.
A more controversial issue has been the treatment of patients harboring staghorn calculi, a condition that carries a significant risk of mortality if it is untreated. Boyce and Elkins (1974) established anatrophic nephrolithotomy as a standard treatment of patients with staghorn stones in the United States. Overall, the reported stone-free rate after open surgery for struvite calculi is about 85%, with a 30% stone recurrence rate during 6 years (Griffith, 1978). In comparing results for a combination of PNL and SWL with reported results of anatrophic nephrolithotomy, Kahnoski and associates (1986) reported stone-free rates (85%) similar to those for the open surgical procedures, although convalescence and hospital stay for PNL were shorter and blood loss was less.
Snyder and Smith (1986) compared PNL with anatrophic nephrolithotomy for patients with staghorn calculi. They reported that although the retained stone fragment rate was higher for PNL than for anatrophic nephrolithotomy (13% vs. 0%), shorter procedure times, reduced need for blood transfusions and narcotics, and far more rapid return to work were achieved with PNL.
A meta-analysis undertaken by the AUA documented stone-free rates of 81.6% for open stone surgery, 80.8% for combined PNL and SWL, 73.3% for PNL, and only 50% for SWL monotherapy (Segura et al, 1994; Preminger et al, 2005). The more invasive the procedure, the greater the stone-free rate; however, morbidity was higher as well. Although SWL carried the lowest morbidity, a greater number of unplanned post-treatment interventions were necessary. The AUA concluded that for most patients, neither SWL monotherapy nor open stone surgery should be the first-line treatment of staghorn stones. As a guideline, PNL, followed by SWL or repeated PNL procedures as warranted, should be used for most patients with struvite staghorn calculi.
There are no strict guidelines that define which patient should undergo an open surgical procedure for stone removal. Some indications, such as a stone burden too large for PNL, clearly rely on the surgeon’s judgment and experience and the availability of equipment. Also, those patients harboring calculi that may require multiple PNL or SWL treatments may be good candidates for an open procedure. Although a single open surgical procedure may seem to be the optimal procedure in the short term, the inevitable scar tissue that develops will compromise any future stone removal procedures. A small group of patients whose conditions are refractory to PNL, SWL, and ureteroscopy may require an open surgical procedure as a salvage technique.
Nephrectomy remains an option for patients with nonfunctioning kidneys or stone disease with a normal contralateral kidney. Partial nephrectomy is also an option for a stone in a localized area of irrevocably poor function. In addition, patients with an associated anatomic abnormality requiring open operative intervention, such as UPJ obstruction and infundibular stenosis, may be candidates for an open surgical approach. Some patients requiring open surgery unrelated to their urologic problem may also benefit from a simultaneously performed open procedure.
Ureteral Calculi
Although ureterolithotomy has been a time-honored technique for many decades it is seldom performed in the modern, endourologic era. A meta-analysis undertaken by Segura and associates (1997) for the AUA demonstrated median stone-free rates of 87% and 90% for stones in the distal ureter treated by open surgical removal and ureteroscopy, respectively. In the proximal ureter, the stone-free rate for ureterolithotomy was 97% compared with 83% and 72% for SWL and ureteroscopy, respectively. Although the results of open surgery in the proximal ureter were somewhat better than those of minimally invasive techniques, the greater morbidity and longer hospitalization associated with open surgery favored a primary endourologic solution for ureteral stones. Further miniaturization of ureteroscopes, combined with the now widespread availability of the holmium:YAG laser, has increased the success rate for a ureteroscopic approach to proximal ureteral calculi. Grasso and Bagley (1998) have reported a large series of patients undergoing ureteroscopy for proximal ureteral calculi, finding a 97% stone-free rate. Open surgery for patients with ureteral stones is now indicated only as a salvage procedure, when a planned abdominal operation coincides with a symptomatic ureteral stone episode, or when another ureteral abnormality requires open surgical repair.
Laparoscopic Renal Stone Removal
Laparoscopic ureterolithotomy has been previously discussed; the focus of the following section is on laparoscopic treatment of renal calculi. The advent of laparoscopic renal stone removal procedures has provided the urologist with another means to circumvent open stone surgery. Every type of “lithotomy” procedure has been reported by use of a laparoscopic approach (Raboy et al, 1992; Winfield et al, 1993; Ruckle and Segura, 1994; Van Cangh et al, 1995; Harmon et al, 1996; Deger et al, 2004). However, a laparoscopic approach to stone removal should be considered only if the results with SWL or endoscopic approaches are expected to be poor.
There are certain cases in which a laparoscopic lithotomy may be considered a reasonable therapy. Situations that may benefit from a laparoscopic approach include the following: pyeloplasty with pyelolithotomy; patients harboring stones in poorly functioning polar areas or with nonfunctioning kidneys; pelvic kidneys containing a large stone volume, in which laparoscopic techniques can be used to reflect overlying bowel, allowing pyelolithotomy or percutaneous stone removal; and ureterolithotomy for the extremely rare endoscopic failure. In present practice, however, the utilization of laparoscopy in the treatment of renal calculi has been reported to be limited; Desai and Assimos (2008) reported that only 1% of patients require such an approach. In this series, the most common indication for a laparoscopic approach was renal stones with a concomitant UPJ obstruction.
Key Points: Stone Removal: Surgical Techniques and Technology
Albala DM, Assimos DG, Clayman RV, et al. Lower pole I: a prospective randomized trial of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy for lower pole nephrolithiasis-initial results. J Urol. 2001;166(6):2072-2080.
Auge BK, Munver R, Kourambas J, et al. Endoscopic management of symptomatic caliceal diverticula: a retrospective comparison of percutaneous nephrolithotripsy and ureteroscopy. J Endourol. 2002;16:557-563.
de la Rosette JJ, Zuazu JR, Tsakiris P, et al. Prognostic factors and percutaneous nephrolithotomy morbidity: a multivariate analysis of a contemporary series using the Clavien classification. J Urol. 2008;180:2489-2493.
Hollingsworth JM, Rogers MA, Kaufman SR, et al. Medical therapy to facilitate urinary stone passage: a meta-analysis. Lancet. 2006;368:1171-1179.
Kim SC, Tinmouth WW, Kuo RL, et al. Using and choosing a nephrostomy tube after percutaneous nephrolithotomy for large or complex stone disease: a treatment strategy. J Endourol. 2005;19(3):348-352.
Krambeck AE, Gettman MT, Rohlinger AL, et al. Diabetes mellitus and hypertension associated with shock wave lithotripsy of renal and proximal ureteral stones at 19 years of followup. J Urol. 2006;175:1742-1747.
L’Esperance JO, Ekeruo WO, Scales CDJr, et al. Effect of ureteral access sheath on stone-free rates in patients undergoing ureteroscopic management of renal calculi. Urology. 2005;66:252-255.
Lingeman JE, Siegel YI, Steele B, et al. Management of lower pole nephrolithiasis: a critical analysis. J Urol. 1994;151(3):663-667.
Makarov DV, Trock BJ, Allaf ME, Matlaga BR. The effect of ureteral stent placement on post-ureteroscopy complications: a meta-analysis. Urology. 2008;71:796-800.
Monga M, Smith R, Ferral H, et al. Percutaneous ablation of caliceal diverticulum: long-term followup. J Urol. 2000;163(1):28-32.
Pearle MS, Lingeman JE, Leveillee R, et al. Prospective, randomized trial comparing shock wave lithotripsy and ureteroscopy for lower pole caliceal calculi 1 cm or less. J Urol. 2005;173:2005-2009.
Pishchalnikov YA, Neucks JS, VonDerHaar RJ, et al. Air pockets trapped during routine coupling in dry head lithotripsy can significantly decrease the delivery of shock wave energy. J Urol. 2006;176:2706-2710.
Preminger GM, Assimos DG, Lingeman JE, et al. Chapter 1: AUA guideline on management of staghorn calculi: diagnosis and treatment recommendations. J Urol. 2005;173(6):1991-2000.
Preminger GM, Tiselius HG, Assimos DG, et al. 2007 guideline for the management of ureteral calculi. J Urol. 2007;178:2418-2434.
Sapozhnikov OA, Maxwell AD, MacConaghy B, Bailey MR. A mechanistic analysis of stone fracture in lithotripsy. J Acoust Soc Am. 2007;121:1190-1202.
Semins MJ, Trock BJ, Matlaga BR. The effect of shock wave rate on the outcome of shock wave lithotripsy: a meta-analysis. J Urol. 2008;179:194-197. discussion 197
Semins MJ, Trock BJ, Matlaga BR. The safety of ureteroscopy during pregnancy: a systematic review and meta-analysis. J Urol. 2009;181:139-143.
Willis LR, Evan AP, Connors BA, et al. Prevention of lithotripsy-induced renal injury by pre-treating kidneys with low energy shockwaves. J Am Soc Nephrol. 2006;17:336-673.
Ackermann D, Griffith DP, Dunthorn M, et al. Calculation of stone volume and urinary stone staging with computer assistance. J Endourol. 1989;3:355.
Ackermann DK, Fuhrimann R, Pfluger D, et al. Prognosis after extracorporeal shock wave lithotripsy of radiopaque renal calculi: a multivariate analysis. Eur Urol. 1994;25:105-109.
Aghamir SK, Mohseni MG, Ardestani A. Treatment of ureteral calculi with ballistic lithotripsy. J Endourol. 2003;17:887-890.
Ahmed K, Khan MS, Thomas K, et al. Management of cystinuric patients: an observational, retrospective, single-centre analysis. Urol Int. 2008;80:141-144.
Al-Awadi KA, Abdul Halim H, Kehinde EO, Al-Tawheed A. Steinstrasse: a comparison of incidence with and without J stenting and the effect of J stenting on subsequent management. BJU Int. 1999;84:618-621.
Al-Kohlany KM, Shokeir AA, Mosbah A, et al. Treatment of complete staghorn stones: a prospective randomized comparison of open surgery versus percutaneous nephrolithotomy. J Urol. 2005;173:469-473.
Albala DM, Assimos DG, Clayman RV, et al. Lower pole I: a prospective randomized trial of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy for lower pole nephrolithiasis—initial results. J Urol. 2001;166:2072-2080.
Albani JM, Yost AJ, Streem SB. Ureteropelvic junction obstruction: determining durability of endourological intervention. J Urol. 2004;171(Pt. 1):579-582.
Al-Basam S, Bennett JD, Layton ZA, et al. Treatment of caliceal diverticular stones: transdiverticular percutaneous nephrolithotomy with creation of a neoinfundibulum. J Vasc Interv Radiol. 2000;11:885-889.
Alken P, Hutschenreiter G, Gunther R, et al. Percutaneous stone manipulation. J Urol. 1981;125:463-466.
Al-Otaibi K, Hosking DH. Percutaneous stone removal in horseshoe kidneys. J Urol. 1999;162(Pt. 1):674-677.
Andren-Sandberg A. Permanent impairment of renal function demonstrated by renographic follow-up in ureterolithiasis. Scand J Urol Nephrol. 1983;17:81-84.
Andreoni C, Portis AJ, Clayman RV. Retrograde renal pelvic access sheath to facilitate flexible ureteroscopic lithotripsy for the treatment of urolithiasis in a horseshoe kidney. J Urol. 2000;164(4):1290-1291.
Arrabal-Martin M, Fernandez-Rodriguez A, Arrabal-Polo MA, et al. Extracorporeal renal lithotripsy: evolution of residual lithiasis treated with thiazides. Urology. 2006;68:956-959.
Assimos DG, Boyce WH, Harrison LH, et al. The role of open stone surgery since extracorporeal shock wave lithotripsy. J Urol. 1989;142(Pt. 1):263-267.
Assimos DG, Leslie SW, Ng C, et al. The impact of cystinuria on renal function. J Urol. 2002;168:27-30.
Atug F, Castle EP, Burgess SV, Thomas R. Concomitant management of renal calculi and pelvi-ureteric junction obstruction with robotic laparoscopic surgery. BJU Int. 2005;96:1365-1368.
Auge BK, Lallas CD, Pietrow PK, et al. In vitro comparison of standard ultrasound and pneumatic lithotrites with a new combination intracorporeal lithotripsy device. Urology. 2002;60:28-32.
Auge BK, Munver R, Kourambas J, et al. Endoscopic management of symptomatic caliceal diverticula: a retrospective comparison of percutaneous nephrolithotripsy and ureteroscopy. J Endourol. 2002;16:557-563.
Averkiou MA, Crum L. Cavitation: its role in stone comminution and renal injury. In: Lingeman JE, Preminger GM, editors. Topics in clinical urology: new developments in the management of urolithiasis. New York: Igaku-Shoin; 1996:3-20.
Bagley DH. Removal of upper urinary tract calculi with flexible ureteropyeloscopy. Urology. 1990;35:412-416.
Bagrodia A, Gupta A, Raman JD, et al. Impact of body mass index on cost and clinical outcomes after percutaneous nephrolithotomy. J Urol. 2009;181(1):149-153.
Bailey MR, Blackstock DT, Cleveland RO, et al. Comparison of electrohydraulic lithotripters with rigid and pressure-release ellipsoidal reflectors. I. Acoustic fields. J Acoust Soc Am. 1998;104:2517-2524.
Bailey MR, Blackstock DT, Cleveland RO, et al. Comparison of electrohydraulic lithotripters with rigid and pressure-release ellipsoidal reflectors. II. Cavitation fields. J Acoust Soc Am. 1999;106:1149-1160.
Bailey MR, Pishchalnikov YA, Sapozhnikov OA, et al. Cavitation detection during shock-wave lithotripsy. Ultrasound Med Biol. 2005;31:1245-1256.
Ball AJ, Leveillee RJ, Patel VR, et al. Laparoscopic pyeloplasty and flexible nephroscopy: simultaneous treatment of ureteropelvic junction obstruction and nephrolithiasis. JSLS. 2004;8:223-228.
Ballenger EG, Frontz WA, Hamer HG, Lewis B. History of urology. Prepared under the auspices of the American Urological Association. Baltimore: Williams & Wilkins; 1933.
Bani-Hani AH, Segura JW, LeRoy AJ. Urinary matrix calculi: our experience at a single institution. J Urol. 2005;173:120-123.
Barbey F, Joly D, Rieu P, et al. Medical treatment of cystinuria: critical reappraisal of long-term results. J Urol. 2000;163:1419-1423.
Basar H, Ohta N, Kageyama S, et al. Treatment of ureteral and renal stones by electrohydraulic lithotripsy. Int Urol Nephrol. 1997;29:275-280.
Basar H, Yilmaz E, Ozcan S, et al. Four analgesic techniques for shockwave lithotripsy: eutectic mixture local anesthetic is a good alternative. J Endourol. 2003;17:3-6.
Batter SJ, Dretler SP. Ureterorenoscopic approach to the symptomatic caliceal diverticulum. J Urol. 1997;158(Pt. 1):709-713.
Beck EM, Riehle RAJr. The fate of residual fragments after extracorporeal shock wave lithotripsy monotherapy of infection stones. J Urol. 1991;145:6-9. discussion 10
Begun FP, Jacobs SC, Lawson RK. Use of a prototype 3F electrohydraulic electrode with ureteroscopy for treatment of ureteral calculous disease. J Urol. 1988;139:1188-1191.
Benoit G, Blanchet P, Eschwege P, et al. Occurrence and treatment of kidney graft lithiasis in a series of 1500 patients. Clin Transplant. 1996;10:176-180.
Bercowsky E, Shalhav AL, Elbahnasy AM, et al. The effect of patient position on intrarenal anatomy. J Endourol. 1999;13:257-260.
Bhatta KM, Prien ELJr, Dretler SP. Cystine calculi—rough and smooth: a new clinical distinction. J Urol. 1989;142:937-940.
Biyani CS, Joyce AD. Urolithiasis in pregnancy. I. Pathophysiology, fetal considerations and diagnosis. BJU Int. 2002;89:811-818.
Blandy JP, Singh M. The case for a more aggressive approach to staghorn stones. J Urol. 1976;115:505-506.
Boll DT, Patil NA, Paulson EK, et al. Renal stone assessment with dual-energy multidetector CT and advanced postprocessing techniques: improved characterization of renal stone composition—pilot study. Radiology. 2009;250:813-820.
Borghi L, Meschi T, Amato F, et al. Nifedipine and methylprednisolone in facilitating ureteral stone passage: a randomized, double-blind, placebo-controlled study. J Urol. 1994;152:1095-1098.
Boridy IC, Maklad N, Sandler CM. Suspected urolithiasis in pregnant women: imaging algorithm and literature review. AJR Am J Roentgenol. 1996;167:869-875.
Boyce WH, Elkins IB. Reconstructive renal surgery following anatrophic nephrolithotomy: followup of 100 consecutive cases. J Urol. 1974;111:307-312.
Boyle JA, Campbell S, Duncan AM, et al. Serum uric acid levels in normal pregnancy with observations on the renal excretion of urate in pregnancy. J Clin Pathol. 1966;19:501-503.
Brannen GE, Bush WH, Correa RJ, et al. Kidney stone removal: percutaneous versus surgical lithotomy. J Urol. 1985;133:6-12.
Breda A, Ogunyemi O, Leppert JT, et al. Flexible ureteroscopy and laser lithotripsy for single intrarenal stones 2 cm or greater—is this the new frontier? J Urol. 2008;179:981-984.
Brito CG, Lingeman JE, Newman DM. Long-term follow-up of renal function in ESWL treated patients with a solitary kidney. J Urol. 1990;143(Suppl.):299.
Brown J. Diagnostic and treatment patterns for renal colic in US emergency departments. Int Urol Nephrol. 2006;38:87-92.
Brown MW, Carson CC3rd, Dunnick NR, et al. Comparison of the costs and morbidity of percutaneous and open flank procedures. J Urol. 1986;135:1150-1152.
Brownlee N, Foster M, Griffith DP, et al. Controlled inversion therapy: an adjunct to the elimination of gravity-dependent fragments following extracorporeal shock wave lithotripsy. J Urol. 1990;143:1096-1098.
Burgher A, Beman M, Holtzman JL, et al. Progression of nephrolithiasis: long-term outcomes with observation of asymptomatic calculi. J Endourol. 2004;18:534-539.
Burke BJ, Washowich TL. Ureteral jets in normal second- and third-trimester pregnancy. J Clin Ultrasound. 1998;26:423-426.
Burmeister MA, Brauer P, Wintruff M, et al. A comparison of anaesthetic techniques for shock wave lithotripsy: the use of a remifentanil infusion alone compared to intermittent fentanyl boluses combined with a low dose propofol infusion. Anaesthesia. 2002;57:877-881.
Burns JR, Finlayson B, Gauthier J. Calcium oxalate retention in subjects with crystalluria. Urol Int. 1984;39:36-39.
Butler EL, Cox SM, Eberts EG, et al. Symptomatic nephrolithiasis complicating pregnancy. Obstet Gynecol. 2000;96(Pt. 1):753-756.
Caldwell TC, Burns JR. Current operative management of urinary calculi after renal transplantation. J Urol. 1988;140:1360-1363.
Calvano CJ, Moran ME, White MD, et al. Experimental utilization of the holmium laser in a model of ureteroscopic lithotripsy: energy analysis. J Endourol. 1999;13:113-115.
Candau C, Saussine C, Lang H, et al. Natural history of residual renal stone fragments after ESWL. Eur Urol. 2000;37:18-22.
Carr LK, D’A Honey J, Jewett MA, et al. New stone formation: a comparison of extracorporeal shock wave lithotripsy and percutaneous nephrolithotomy. J Urol. 1996;155:1565-1567.
Carson CC. Complications of percutaneous stone extraction: prevention and treatment. Semin Urol. 1986;4:161-169.
Carson CC, Nesbitt JA. Peritoneal extravasation during percutaneous lithotripsy. J Urol. 1985;134:725-727.
Carson CC3rd, Danneberger JE, Weinerth JL. Percutaneous lithotripsy in morbid obesity. J Urol. 1988;139:243-245.
Cass AS. Comparison of first generation (Dornier HM3) and second generation (Medstone STS) lithotriptors: treatment results with 13,864 renal and ureteral calculi. J Urol. 1995;153(Pt. 1):588-592.
Cass AS. Extracorporeal shockwave lithotripsy or percutaneous nephrolithotomy for lower pole nephrolithiasis? J Endourol. 1996;10:17-20.
Castaneda-Zuniga WR, Clayman R, Smith A, et al. Nephrostolithotomy: percutaneous techniques for urinary calculus removal. AJR Am J Roentgenol. 1982;139:721-726.
Charton M, Vallancien G, Veillon B, et al. Urinary tract infection in percutaneous surgery for renal calculi. J Urol. 1986;135:15-17.
Chaussy C, Fuchs G. [Extracorporeal lithotripsy in the treatment of renal lithiasis. 5 years’ experience.]. J Urol (Paris). 1986;92:339-343. [in French]
Chaussy C, Fuchs G. Current state and future developments of noninvasive treatment of human urinary stones with extracorporeal shock wave lithotripsy. J Urol. 1989;141(Pt. 2):782-789.
Chaussy C, Fuchs G, Kahn R, et al. Transurethral ultrasonic ureterolithotripsy using a solid-wire probe. Urology. 1987;29:531-532.
Chaussy C, Schmiedt E. Extracorporeal shock wave lithotripsy (ESWL) for kidney stones: an alternative to surgery? Urol Radiol. 1984;6:80-87.
Chen RN, Streem SB. Extracorporeal shock wave lithotripsy for lower pole calculi: long-term radiographic and clinical outcome. J Urol. 1996;156:1572-1575.
Chow GK, Streem SB. Contemporary urological intervention for cystinuric patients: immediate and long-term impact and implications. J Urol. 1998;160:341-344. discussion 344–5
Cicerello E, Merlo F, Gambaro G, et al. Effect of alkaline citrate therapy on clearance of residual renal stone fragments after extracorporeal shock wave lithotripsy in sterile calcium and infection nephrolithiasis patients. J Urol. 1994;151:5-9.
Clayman RV, McClennan BL, Garvin TJ, et al. Lithostar: an electromagnetic acoustic shock wave unit for extracorporeal lithotripsy. J Endourol. 1989;3:307-313.
Clayman RV, Surya V, Miller RP, et al. Percutaneous nephrolithotomy: an approach to branched and staghorn renal calculi. JAMA. 1983;250:73-75.
Clayman RV, Surya V, Miller RP, et al. Percutaneous nephrolithotomy: extraction of renal and ureteral calculi from 100 patients. J Urol. 1984;131:868-871.
Cleveland RO, Bailey MR, Hartennaum B, et al. Design and characterization of a research electrohydraulic lithotriptor patterned after the Dornier HM3. Rev Scientific Inst. 2000;71:2514-2525.
Coe FL, Parks JH, Lindheimer MD. Nephrolithiasis during pregnancy. N Engl J Med. 1978;298:324-326.
Cohen TD, Preminger GM. Management of calyceal calculi. Urol Clin North Am. 1997;24:81-96.
Coleman AJ, Saunders JE, Crum LA, et al. Acoustic cavitation generated by an extracorporeal shockwave lithotripter. Ultrasound Med Biol. 1987;13:69-76.
Coll DM, Varanelli MJ, Smith RC, et al. Relationship of spontaneous passage of ureteral calculi to stone size and location as revealed by unenhanced helical CT. AJR Am J Roentgenol. 2002;178:101-103.
Colon-Perez B, Canto RJ, Ramos ME. Simultaneous bilateral nephrostolithotomy: immediate results in 3 cases. J Endourol. 1987;1:209-211.
Connors BA, Evan AP, Blomgren PM, et al. Reducing shock number dramatically decreases lesion size in a juvenile kidney model. J Endourol. 2006;20:607-611.
Connors BA, Evan AP, Blomgren PM, et al. Effect of initial shock wave voltage on shock wave lithotripsy–induced lesion size during step-wise voltage ramping. BJU Int. 2009;103:104-107.
Connors BA, Evan AP, Willis LR, et al. The effect of discharge voltage on renal injury and impairment caused by lithotripsy in the pig. J Am Soc Nephrol. 2000;11:310-318.
Consensus conference. Prevention and treatment of kidney stones. JAMA. 1988;260:977-981.
Coptcoat MJ, Ison KT, Watson G, et al. Lasertripsy for ureteric stones in 120 cases: lessons learned. Br J Urol. 1988;61:487-489.
Coptcoat MJ, Webb DR, Kellett MJ, et al. The treatment of 100 consecutive patients with ureteral calculi in a British stone center. J Urol. 1987;137:1122-1123.
Coptcoat MJ, Webb DR, Kellet MJ, et al. The steinstrasse: a legacy of extracorporeal lithotripsy? Eur Urol. 1988;14:93-95.
Coughlin BF, Risius B, Streem SB, et al. Abdominal radiograph and renal ultrasound versus excretory urography in the evaluation of asymptomatic patients after extracorporeal shock wave lithotripsy. J Urol. 1989;142:1419-1423. discussion 1423–24
Coury TA, Sonda LP, Lingeman JE, et al. Treatment of painful caliceal stones. Urology. 1988;32:119-123.
Crum L. Surface oscillations and jet development in pulsating bubbles. J Physique. 40, 1979. C8–285–C8–288
Crum L. Cavitation microjets as a contributory mechanism for renal calculi disintegration in ESWL. J Urol. 1988;140:1587-1590.
Cummings JM, Boullier JA, Izenberg SD, et al. Prediction of spontaneous ureteral calculous passage by an artificial neural network. J Urol. 2000;164:326-328.
Curran MJ, Little AF, Bouyounes B, et al. Retroperitoneoscopic technique for treating symptomatic caliceal diverticula. J Endourol. 1999;13:723-725.
Curtis R, Thorpe AC, Marsh R. Modification of the technique of percutaneous nephrolithotomy in the morbidly obese patient. Br J Urol. 1997;79:138-140.
Cybulski PA, Joo H, Honey RJ. Ureteroscopy: anesthetic considerations. Urol Clin North Am. 2004;31:43-47. viii
D’a Honey RJ, Luymes J, Weir MJ, et al. Mechanical percussion inversion can result in relocation of lower pole stone fragments after shock wave lithotripsy. Urology. 2000;55:204-206.
Dafnis E, Sabatini S. The effect of pregnancy on renal function: physiology and pathophysiology. Am J Med Sci. 1992;303:184-205.
Dalela D, Goel A, Singh P, et al. Renal capsular block: a novel method for performing percutaneous nephrolithotomy under local anesthesia. J Endourol. 2004;18:544-546.
Dash A, Schuster TG, Hollenbeck BK, et al. Ureteroscopic treatment of renal calculi in morbidly obese patients: a stone-matched comparison. Urology. 2002;60:393-397.
Daudon M, Estepa L, Kebede M, et al. [Urinary calculi and crystalluria in HIV? patients treated with indinavir sulfate.]. Presse Med. 1997;26:1612-1615. [in French]
de la Rosette JJ, Zuazu JR, Tsakiris P, et al. Prognostic factors and percutaneous nephrolithotomy morbidity: a multivariate analysis of a contemporary series using the Clavien classification. J Urol. 2008;180:2489-2493.
Deger S, Tuellmann M, Schoenberger B, et al. Laparoscopic anatrophic nephrolithotomy. Scand J Urol Nephrol. 2004;38:263-265.
Delius M. Minimal static excess pressure minimises the effect of extracorporeal shock waves on cells and reduces it on gallstones. Ultrasound Med Biol. 1997;23:611-617.
Delius M, Jordan M, Eizenhoefer H, et al. Biological effects of shock waves: kidney haemorrhage by shock waves in dogs—administration rate dependence. Ultrasound Med Biol. 1988;14:689-694.
Del Pizzo JJ, Sklar GN. O’Brien peel-away sheath: an alternative for allograft percutaneous nephroscopy. J Endourol. 1999;13:31-33.
Delvecchio FC, Auge BK, Munver R, et al. Shock wave lithotripsy causes ipsilateral renal injury remote from the focal point: the role of regional vasoconstriction. J Urol. 2003;169:1526-1529.
Denstedt JD. Use of Swiss Lithoclast for percutaneous nephrolithotripsy. J Endourol. 1993;7:477-480.
Denstedt JD. Intracorporeal lithotriptors. In: Smith AD, editor. Smith’s textbook of endourology. St. Louis: Quality Medical; 1996:60-77.
Denstedt JD, Clayman RV. Electrohydraulic lithotripsy of renal and ureteral calculi. J Urol. 1990;143:13-17.
Denstedt JD, Clayman RV, Picus DD. Comparison of endoscopic and radiological residual fragment rate following percutaneous nephrolithotripsy. J Urol. 1991;145:703-705.
Denstedt JD, Eberwein PM, Singh RR. The Swiss Lithoclast: a new device for intracorporeal lithotripsy. J Urol. 1992;148(Pt. 2):1088-1090.
Denstedt JD, Razvi H. Management of urinary calculi during pregnancy. J Urol. 1992;148(Pt. 2):1072-1074. discussion 1074–5
Desai M. Ultrasonography-guided punctures with and without puncture guide. J Endourol. 2009;23(10):1641-1643.
Desai M, Grover R, Manohar T, Ganpule A. Simultaneous bilateral percutaneous nephrolithotomy: a single-center experience. J Endourol. 2007;21:508-514.
Desai RA, Assimos DG. Role of laparoscopic stone surgery. Urology. 2008;71:578-580.
Desnos E. The history of urology to the nineteenth century. In: Murphy LJT, editor. The history of urology. Springfield (IL): Charles C. Thomas, 1972.
Deyoe LA, Cronan JJ, Breslaw BH, et al. New techniques of ultrasound and color Doppler in the prospective evaluation of acute renal obstruction: do they replace the intravenous urogram? Abdom Imaging. 1995;20:58-63.
Dhar NB, Thornton J, Karafa MT, et al. A multivariate analysis of risk factors associated with subcapsular hematoma formation following electromagnetic shock wave lithotripsy. J Urol. 2004;172(Pt. 1):2271-2274.
Diamond DA, Rickwood AM, Lee PH, et al. Infection stones in children: a twenty-seven-year review. Urology. 1994;43:525-527.
Dimarco DS, Gettman MT, McGee SM, et al. Long-term success of antegrade endopyelotomy compared with pyeloplasty at a single institution. J Endourol. 2006;20:707-712.
Di Silverio F, Gallucci M, Alpi G. Staghorn calculi of the kidney: classification and therapy. Br J Urol. 1990;65:449-452.
Donnellan SM, Harewood LM, Webb DR. Percutaneous management of caliceal diverticular calculi: technique and outcome. J Endourol. 1999;13:83-88.
Drach GW, Dretler S, Fair W, et al. Report of the United States cooperative study of extracorporeal shock wave lithotripsy. J Urol. 1986;135:1127-1133.
Dretler SP. Stone fragility—a new therapeutic distinction. J Urol. 1988;139:1124-1127.
Dretler SP. Ureteroscopic fragmentation followed by extracorporeal shock wave lithotripsy: a treatment alternative for selected large or staghorn calculi. J Urol. 1994;151:842-846.
Dretler SP. Management of the lower ureteral stone. AUA Update Series. 1995;18:62-67.
Dretler SP, Polykoff G. Calcium oxalate stone morphology: fine tuning our therapeutic distinctions. J Urol. 1996;155:828-833.
Dudley NE. Sir Henry Morris, Bart. (1844-1926). Invest Urol. 1973;11:170-171.
Dushinski JW, Lingeman JE. Simultaneous bilateral percutaneous nephrolithotomy. J Urol. 1997;158:2065-2068.
Dushinski JW, Lingeman JE. High-speed photographic evaluation of holmium laser. J Endourol. 1998;12:177-181.
Dyer RB, Assimos DG, Regan JD. Update on interventional uroradiology. Urol Clin North Am. 1997;24:623-652.
Eassa WA, Sheir KZ, Gad HM, et al. Prospective study of the long-term effects of shock wave lithotripsy on renal function and blood pressure. J Urol. 2008;179:964-968.
Eichel L, Batzold P, Erturk E. Operator experience and adequate anesthesia improve treatment outcome with third-generation lithotripters. J Endourol. 2001;15:671-673.
Eisenmenger W. Shock wave measuring techniques in liquids. Proceedings of the 16th International Congress on Acoustics and 135th Meeting of the Acoustical Society of America, 1998. 2793–4
El-Anany FG, Hammouda HM, Maghraby HA, et al. Retrograde ureteropyeloscopic holmium laser lithotripsy for large renal calculi. BJU Int. 2001;88:850-853.
Elashry OM, DiMeglio RB, Nakada SY, et al. Intracorporeal electrohydraulic lithotripsy of ureteral and renal calculi using small caliber (1.9F) electrohydraulic lithotripsy probes. J Urol. 1996;156:1581-1585.
El-Assmy A, El-Nahas AR, Madbouly K, et al. Extracorporeal shock-wave lithotripsy monotherapy of partial staghorn calculi: prognostic factors and long-term results. Scand J Urol Nephrol. 2006;40:320-325.
El-Assmy AM, Shokeir AA, El-Nahas AR, et al. Outcome of percutaneous nephrolithotomy: effect of body mass index. Eur Urol. 2006;52(1):199-204.
Elbahnasy AM, Clayman RV, Shalhav AT, et al. Lower pole calyceal stone clearance after shockwave lithotripsy, percutaneous nephrolithotomy, and flexible ureteroscopy: Impact of radiographic spatial anatomy. J Endourol. 1998;12:113-119.
Elbahnasy AM, Shalhav AL, Hoenig DM, et al. Lower caliceal stone clearance after shock wave lithotripsy or ureteroscopy: the impact of lower pole radiographic anatomy. J Urol. 1998;159:676-682.
El-Feel A, Abouel-Fettouh H, Abdel-Hakim AM. Laparoscopic transperitoneal ureterolithotomy. J Endourol. 2007;21:50-54.
El-Moula MG, Abdallah A, El-Anany F, et al. Laparoscopic ureterolithotomy: our experience with 74 cases. Int J Urol. 2008;15:593-597.
El-Nahas AR, El-Assmy AM, Mansour O, Sheir KZ. A prospective multivariate analysis of factors predicting stone disintegration by extracorporeal shock wave lithotripsy: the value of high-resolution noncontrast computed tomography. Eur Urol. 2007;51:1688-1693. discussion 1693–4
Elves AW, Tilling K, Menezes P, et al. Early observations of the effect of extracorporeal shockwave lithotripsy on blood pressure: a prospective randomized control clinical trial. BJU Int. 2000;85:611-615.
Erhard M, Salwen J, Bagley DH. Ureteroscopic removal of mid and proximal ureteral calculi. J Urol. 1996;155:38-42.
Eshghi AM, Roth JS, Smith AD. Percutaneous transperitoneal approach to a pelvic kidney for endourological removal of staghorn calculus. J Urol. 1985;134:525-527.
Eshghi M, Schiff RG, Smith AD. Renal effects of percutaneous stone removal. Urology. 1989;33:120-124.
Esuvaranathan K, Tan EC, Tung KH, et al. Stones in horseshoe kidneys: results of treatment by extracorporeal shock wave lithotripsy and endourology. J Urol. 1991;146:1213-1215.
Eterovic D, Situm M, Juretic-Kuscic L, et al. A decrease in blood pressure after pyelolithotomy but not extracorporeal lithotripsy. Urol Res. 2005;33:93-98.
Evan AP, Coe FL, Lingeman JE, et al. Renal crystal deposits and histopathology in patients with cystine stones. Kidney Int. 2006;69:2227-2235.
Evan AP, Coe FL, Rittling SR, et al. Apatite plaque particles in inner medulla of kidneys of calcium oxalate stone formers: osteopontin localization. Kidney Int. 2005;68:145-154.
Evan AP, McAteer JA, Connors BA, et al. Renal injury during shock wave lithotripsy is significantly reduced by slowing the rate of shock wave delivery. BJU Int. 2007;100(3):624-627.
Evan AP, Willis LR, Connors B, et al. Shock wave lithotripsy–induced renal injury. Am J Kidney Dis. 1991;17:445-450.
Evan AP, Willis LR, Lingeman JE, et al. Renal trauma and the risk of long-term complications in shock wave lithotripsy. Nephron. 1998;78:1-8.
Evan AP, Willis LR, McAteer JA, et al. Kidney damage and renal functional changes are minimized by waveform control that suppresses cavitation in shock wave lithotripsy. J Urol. 2002;168(Pt. 1):1556-1562.
Evans WP, Resnick MI. Horseshoe kidney and urolithiasis. J Urol. 1981;125:620-621.
Fedullo LM, Pollack HM, Banner MP, et al. The development of steinstrassen after ESWL: frequency, natural history, and radiologic management. AJR Am J Roentgenol. 1988;151:1145-1147.
Fernstrom I, Johansson B. Percutaneous pyelolithotomy: a new extraction technique. Scand J Urol Nephrol. 1976;10:257-259.
Fine JK, Pak CY, Preminger GM. Effect of medical management and residual fragments on recurrent stone formation following shock wave lithotripsy. J Urol. 1995;153:27-32. discussion 33
Fischer CP, Sonda LP3rd, Diokno AC. Use of cryoprecipitate coagulum in extracting renal calculi. Urology. 1980;15:6-13.
Flohr TG, McCollough CH, Bruder H, et al. First performance evaluation of a dual-source CT (DSCT) system. Eur Radiol. 2006;16:256-268.
Floratos DL, de la Rosette JJ. Lasers in urology. BJU Int. 1999;84:204-211.
Fong YK, Ho SH, Peh OH, et al. Extracorporeal shockwave lithotripsy and intracorporeal lithotripsy for proximal ureteric calculi—a comparative assessment of efficacy and safety. Ann Acad Med Singapore. 2004;33:80-83.
Frang D, Hamvas A, Kalman J, et al. Experience with the Direx Tripter X-1 shock-wave lithotripter. Int Urol Nephrol. 1992;24:481-490.
Frankenschmidt A, Sommerkamp H. Shock wave lithotripsy during pregnancy: a successful clinical experiment. J Urol. 1998;159:501-502.
Freedman DS, Khan LK, Serdula MK, et al. Trends and correlates of class 3 obesity in the United States from 1990 through 2000. JAMA. 2002;288:1758-1761.
Freiha GS, Glickman RD, Teichman JM. Holmium:YAG laser-induced damage to guidewires: experimental study. J Endourol. 1997;11:331-336.
Fuchs AM, Wolfson BA, Fuchs GJ. Staghorn stone treatment with extracorporeal shock wave lithotripsy monotherapy: long term results. J Endourol. 1991;5:45.
Fuchs GJ. Ultrasonic lithotripsy in the ureter. Urol Clin North Am. 1988;15:347-359.
Fuchs GJ, David RD. Flexible ureterorenoscopy, dilatation of narrow calyceal neck, and ESWL: a new, minimally invasive approach to stones in caliceal diverticula. J Endourol. 1989;3:255-263.
Fuchs GJ, Fuchs AM. [Flexible endoscopy of the upper urinary tract: a new minimally invasive method for diagnosis and treatment.]. Urologe A. 1990;29:313-320. [in German]
Galvin DJ, Pearle MS. The contemporary management of renal and ureteric calculi. BJU Int. 2006;98:1283-1288.
Gaur DD, Agarwal DK, Purohit KC, et al. Retroperitoneal laparoscopic ureterolithotomy for multiple upper mid ureteral calculi. J Urol. 1994;151:1001-1002.
Gaur DD, Trivedi S, Prabhudesai MR, et al. Laparoscopic ureterolithotomy: technical considerations and long-term follow-up. BJU Int. 2002;89:339-343.
Gedroyc WM, MacIver D, Joyce MR, et al. Percutaneous stone and stent removal from renal transplants. Clin Radiol. 1989;40:174-177.
Gerber GS. Management of lower-pole caliceal stones. J Endourol. 2003;17:501-503.
Gerspach JM, Bellman GC, Stoller ML, et al. Conservative management of colon injury following percutaneous renal surgery. Urology. 1997;49:831-836.
Gertner JM, Coustan DR, Kliger AS, et al. Pregnancy as state of physiologic absorptive hypercalciuria. Am J Med. 1986;81:451-456.
Gettman MT, Segura JW. Struvite stones: diagnosis and current treatment concepts. J Endourol. 1999;13:653-658.
Giblin JG, Lossef S, Pahira JJ. A modification of standard percutaneous nephrolithotripsy technique for the morbidly obese patient. Urology. 1995;46:491-493.
Gil-Vernet J. New surgical concepts in removing renal calculi. Urol Int. 1965;20:255-288.
Gil-Vernet JM, Culla A. Advances in intraoperative renal radiography: 3-dimensional radiography of the kidney. J Urol. 1981;125:614-619.
Gluckman GR, Stoller M, Irby P. Laparoscopic pyelocaliceal diverticula ablation. J Endourol. 1993;7:315-317.
Goel A, Hemal AK. Upper and mid-ureteric stones: a prospective unrandomized comparison of retroperitoneoscopic and open ureterolithotomy. BJU Int. 2001;88:679-682.
Goldwasser B, Cohan RH, Dunnick NR, et al. Role of linear tomography in evaluation of patients with nephrolithiasis. Urology. 1989;33:253-256.
Goodman TM. Ureteroscopy with pediatric cystoscope in adults. Urology. 1977;9:394.
Goodwin WE, Casey WC, Woolf W. Percutaneous trocar (needle) nephrostomy in hydronephrosis. JAMA. 1955;157:891-894.
Gorton E, Whitfield HN. Renal calculi in pregnancy. Br J Urol. 1997;80(Suppl. 1):4-9.
Gracewski SM, Dahake G, Ding Z, et al. Internal stress wave measurements in solids subjected to lithotripter pulses. J Acoust Soc Am. 1993;94(Pt. 1):652-661.
Graff J, Diederichs W, Schulze H. Long-term followup in 1,003 extracorporeal shock wave lithotripsy patients. J Urol. 1988;140:479-483.
Graham JB, Nelson JB. Percutaneous caliceal irrigation during extracorporeal shock wave lithotripsy for lower pole renal calculi. J Urol. 1994;152(Pt. 2):2227.
Grasso M, Bagley D. Small diameter, actively deflectable, flexible ureteropyeloscopy. J Urol. 1998;160:1648-1653. discussion 1653–4
Grasso M, Beaghler M, Loisides P. The case for primary endoscopic management of upper urinary tract calculi. II. Cost and outcome assessment of 112 primary ureteral calculi. Urology. 1995;45:372-376.
Grasso M, Chalik Y. Principles and applications of laser lithotripsy: experience with the holmium laser lithotrite. J Clin Laser Med Surg. 1998;16:3-7.
Grasso M, Conlin M, Bagley D. Retrograde ureteropyeloscopic treatment of 2 cm or greater upper urinary tract and minor staghorn calculi. J Urol. 1998;160:346-351.
Grasso M, Ficazzola M. Retrograde ureteropyeloscopy for lower pole caliceal calculi. J Urol. 1999;162:1904-1908.
Grasso M, Lang G, Loisides P, et al. Endoscopic management of the symptomatic caliceal diverticular calculus. J Urol. 1995;153:1878-1881.
Grasso M, Taylor F. Techniques for percutaneous renal access. In: Sosa RE, Jenkins AD, Albala DM, Perlmutter AP, editors. Textbook of endourology. Philadelphia: WB Saunders; 1997:99-113.
Green DF, Lytton B. Early experience with direct vision electrohydraulic lithotripsy of ureteral calculi. J Urol. 1985;133:767-770.
Griffith DP. Struvite stones. Kidney Int. 1978;13:372-382.
Griffith DP, Gibson JR, Clinton CW, et al. Acetohydroxamic acid: clinical studies of a urease inhibitor in patients with staghorn renal calculi. J Urol. 1978;119:9-15.
Griffith DP, Valiquette L. PICA/burden: a staging system for upper tract urinary stones. J Urol. 1987;138:253-257.
Grocela JA, Dretler SP. Intracorporeal lithotripsy: instrumentation and development. Urol Clin North Am. 1997;24:13-23.
Gupta NP, Ansari MS, Kesarvani P, et al. Role of computed tomography with no contrast medium enhancement in predicting the outcome of extracorporeal shock wave lithotripsy for urinary calculi. BJU Int. 2005;95:1285-1288.
Gupta NP, Singh DV, Hemal AK, et al. Infundibulopelvic anatomy and clearance of inferior caliceal calculi with shock wave lithotripsy. J Urol. 2000;163:24-27.
Gur U, Lifshitz DA, Lask D, et al. Ureteral ultrasonic lithotripsy revisited: a neglected tool. J Endourol. 2004;18:137-140.
Harewood LM, Agarwal D, Lindsay S, et al. Extraperitoneal laparoscopic caliceal diverticulectomy. J Endourol. 1996;10:425-430.
Harmon WJ, Kleer E, Segura JW. Laparoscopic pyelolithotomy for calculus removal in a pelvic kidney. J Urol. 1996;155:2019-2020.
Harper JM, Samuell CT, Hallson PC, et al. Risk factors for calculus formation in patients with renal transplants. Br J Urol. 1994;74:147-150.
Harvey EB, Boice JDJr, Honeyman M, et al. Prenatal x-ray exposure and childhood cancer in twins. N Engl J Med. 1985;312:541-545.
Hattery RR, King BF. Technique and application of MR urography. Radiology. 1995;194:25-27.
Haupt G, Haupt A. In vitro comparison of 4 ultrasound lithotripsy devices. J Urol. 2003;170(5):1731-1733.
Hedelin H, Geterud K, Grenabo L, et al. Percutaneous surgery for stones in pyelocaliceal diverticula. Br J Urol. 1988;62:206-208.
Heidenreich A, Desgrandchamps F, Terrier F. Modern approach of diagnosis and management of acute flank pain: review of all imaging modalities. Eur Urol. 2002;41:351-362.
Hendricks SK, Ross SO, Krieger JN. An algorithm for diagnosis and therapy of management and complications of urolithiasis during pregnancy. Surg Gynecol Obstet. 1991;172:49-54.
Henriksson C, Geterud K, Grenabo L, et al. Percutaneous renal and ureteric stone extraction: report on the first 500 operations. Scand J Urol Nephrol. 1989;23:291-297.
Hepp W. Survey of the development of shock wave lithotripsy. Wessling (Germany): Dornier Medizintechnik GmbH; 1984.
Herman JR. Urology, a view through the retrospectoscope. Hagerstown (MD): Harper & Row; 1973.
Hirschowitz BI, Peters CW, Curtiss LE. Preliminary report on a long fiberscope for examination of stomach and duodenum. Med Bull (Ann Arbor). 1957;23:178-180.
Hjollund Madsen E. The value of tomography for the demonstration of small intrarenal calcifications. Br J Radiol. 1972;45:203-206.
Hockley NM, Lingeman JE, Hutchinson CL. Relative efficacy of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy in the management of cystine calculi. J Endourol. 1989;3:273-285.
Hofbauer J, Hobarth K, Marberger M. Electrohydraulic versus pneumatic disintegration in the treatment of ureteral stones: A randomized, prospective trial. J Urol. 1995;153(Pt. 1):623-625.
Hofmann R, Stoller ML. Endoscopic and open stone surgery in morbidly obese patients. J Urol. 1992;148(Pt. 2):1108-1111.
Hohenfellner M, Schultz-Lampel D, Lampel A, et al. Tumor in the horseshoe kidney: clinical implications and review of embryogenesis. J Urol. 1992;147:1098-1102.
Hollenbeck BK, Schuster TG, Faerber GJ, et al. Flexible ureteroscopy in conjunction with in situ laser lithotripsy for lower pole calculi. J Urol. 2001;165(Suppl.):375.
Hollingsworth JM, Rogers MA, Kaufman SR, et al. Medical therapy to facilitate urinary stone passage: a meta-analysis. Lancet. 2006;368:1171-1179.
Holm-Nielsen A, Jorgensen T, Mogensen P, et al. The prognostic value of probe renography in ureteric stone obstruction. Br J Urol. 1981;53:504-507.
Holman E, Salah MA, Toth C. Comparison of 150 simultaneous bilateral and 300 unilateral percutaneous nephrolithotomies. J Endourol. 2002;16:33-36.
Holman E, Toth C. Laparoscopically assisted percutaneous transperitoneal nephrolithotomy in pelvic dystopic kidneys: experience in 15 successful cases. J Laparoendosc Adv Surg Tech A. 1998;8:431-435.
Horowitz E, Schmidt JD. Renal calculi in pregnancy. Clin Obstet Gynecol. 1985;28:324-338.
Horrigan TJ, Reese CS, Parres JA, et al. A study of resistive indices in the arcuate arteries of the kidney over the course of gestation. J Perinatol. 1996;16:467-469.
Houshiar AM, Ercole CJ. Urinary calculi during pregnancy: when are they cause for concern? Postgrad Med. 1996;100:131-138.
Howarth AT, Morgan DB, Payne RB. Urinary excretion of calcium in late pregnancy and its relation to creatinine clearance. Am J Obstet Gynecol. 1977;129:499-502.
Hoznek A, Herard A, Ogiez N, et al. Symptomatic caliceal diverticula treated with extraperitoneal laparoscopic marsupialization fulguration and gelatin resorcinol formaldehyde glue obliteration. J Urol. 1998;160:352-355.
Hsu TH, Streem SB. Metabolic abnormalities in patients with caliceal diverticular calculi. J Urol. 1998;160:1640-1642.
Huang S, Patel H, Bellman GC. Cost effectiveness of electrohydraulic lithotripsy v Candela pulsed-dye laser in management of the distal ureteral stone. J Endourol. 1998;12:237-240.
Hubner W, Porpaczy P. Treatment of caliceal calculi. Br J Urol. 1990;66:9-11.
Hubner WA, Irby P, Stoller ML. Natural history and current concepts for the treatment of small ureteral calculi. Eur Urol. 1993;24:172-176.
Huffman JL, Bagley DH, Lyon ES. Extending cystoscopic techniques into the ureter and renal pelvis: experience with ureteroscopy and pyeloscopy. JAMA. 1983;250:2002-2005.
Hulbert JC, Reddy PK, Hunter DW, et al. Percutaneous techniques for the management of caliceal diverticula containing calculi. J Urol. 1986;135:225-227.
Humphreys MR, Miller NL, Williams JCJr, et al. A new world revealed: early experience with digital ureteroscopy. J Urol. 2008;179:970-975.
Husmann DA, Milliner DS, Segura JW. Ureteropelvic junction obstruction with a simultaneous renal calculus: long-term followup. J Urol. 1995;153:1399-1402.
Hussain Z, Inman RD, Elves AW, et al. Use of glyceryl trinitrate patches in patients with ureteral stones: a randomized, double-blind, placebo-controlled study. Urology. 2001;58(4):521-525.
Ibrahim AI, Shetty SD, Awad RM, et al. Prognostic factors in the conservative treatment of ureteric stones. Br J Urol. 1991;67:358-361.
Inci K, Sahin A, Islamoglu E, et al. Prospective long-term followup of patients with asymptomatic lower pole caliceal stones. J Urol. 2007;177:2189-2192.
Irving SO, Calleja R, Lee F, et al. Is the conservative management of ureteric calculi of > 4 mm safe? BJU Int. 2000;85:637-640.
Janetschek G, Frauscher F, Knapp R, et al. New onset hypertension after extracorporeal shock wave lithotripsy: age related incidence and prediction by intrarenal resistive index. J Urol. 1997;158:346-351.
Janetschek G, Kunzel KH. Percutaneous nephrolithotomy in horseshoe kidneys. Applied anatomy and clinical experience. Br J Urol. 1988;62:117-122.
Jarrett TW, Smith AD. Percutaneous treatment of calyceal diverticula. In: Smith AD, editor. Controversies in endourology. Philadelphia: WB Saunders, 1986.
Jenkins AD, Gillenwater JY. Extracorporeal shock wave lithotripsy in the prone position: treatment of stones in the distal ureter or anomalous kidney. J Urol. 1988;139:911-915.
Jewett MA, Bombardier C, Logan AG, et al. A randomized controlled trial to assess the incidence of new onset hypertension in patients after shock wave lithotripsy for asymptomatic renal calculi. J Urol. 1998;160:1241-1243.
Johrde LG, Cocks FH. Fracture strength studies of renal calculi. J Mater Sci Lett. 1985;4:1264-1265.
Jones DJ, Wickham JE, Kellett MJ. Percutaneous nephrolithotomy for calculi in horseshoe kidneys. J Urol. 1991;145:481-483.
Jones JA, Lingeman JE, Steidle CP. The roles of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy in the management of pyelocaliceal diverticula. J Urol. 1991;146:724-727.
Joseph P, Mandal AK, Singh SK, et al. Computerized tomography attenuation value of renal calculus: can it predict successful fragmentation of the calculus by extracorporeal shock wave lithotripsy? A preliminary study. J Urol. 2002;167:1968-1971.
Kachel TA, Vijan SR, Dretler SP. Endourological experience with cystine calculi and a treatment algorithm. J Urol. 1991;145:25-28.
Kahnoski RJ, Lingeman JE, Coury TA, et al. Combined percutaneous and extracorporeal shock wave lithotripsy for staghorn calculi: an alternative to anatrophic nephrolithotomy. J Urol. 1986;135:679-681.
Kajikawa H, Kameoka H, Nishimoto N, et al. [Ectopic ureteral opening in four males: including a case of inverted Y ureteral duplication.]. Hinyokika Kiyo. 1985;31:2039-2048. [in Japanese]
Kane CJ, Bolton DM, Stoller ML. Current indications for open stone surgery in an endourology center. Urology. 1995;45:218-221.
Kang DE, Maloney MM, Haleblian GE, et al. Effect of medical management on recurrent stone formation following percutaneous nephrolithotomy. J Urol. 2007;177:1785-1788. discussion 1788–9
Kang HW, Lee H, Teichman JM, et al. Dependence of calculus retropulsion on pulse duration during Ho:YAG laser lithotripsy. Lasers Surg Med. 2006;38:762-772.
Kapoor DA, Leech JE, Yap WT, et al. Cost and efficacy of extracorporeal shock wave lithotripsy versus ureteroscopy in the treatment of lower ureteral calculi. J Urol. 1992;148(Pt. 2):1095-1096.
Kavoussi LR, Albala DM, Basler JW, et al. Percutaneous management of urolithiasis during pregnancy. J Urol. 1992;148(Pt. 2):1069-1071.
Kaye KW, Clayman RV. Tamponade nephrostomy catheter for percutaneous nephrostolithotomy. Urology. 1986;27:441-445.
Keeley FX, Gialas I, Pillai M, et al. Laparoscopic ureterolithotomy: the Edinburgh experience. BJU Int. 1999;84:765-769.
Keeley FXJr, Pillai M, Smith G, et al. Electrokinetic lithotripsy: safety, efficacy and limitations of a new form of ballistic lithotripsy. BJU Int. 1999;84:261-263.
Keeley FXJr, Tilling K, Elves A, et al. Preliminary results of a randomized controlled trial of prophylactic shock wave lithotripsy for small asymptomatic renal calyceal stones. BJU Int. 2001;87:1-8.
Kelleher JP, Wakefield AJ, Gordon I, et al. Renal injury in complete ureteric obstruction: a functional and morphological study. Urol Res. 1991;19:245-248.
Kim SC, Kuo RL, Tinmouth WW, et al. Percutaneous nephrolithotomy for caliceal diverticular calculi: a novel single stage approach. J Urol. 2005;173:1194-1198.
Kim SC, Matlaga BR, Tinmouth WW, et al. In vitro assessment of a novel dual probe ultrasonic intracorporeal lithotriptor. J Urol. 2007;177:1363-1365.
Kim SC, Oh CH, Moon YT, et al. Treatment of steinstrasse with repeat extracorporeal shock wave lithotripsy: experience with piezoelectric lithotriptor. J Urol. 1991;145:489-491.
Kim SC, Tinmouth WW, Kuo RL, et al. Using and choosing a nephrostomy tube after percutaneous nephrolithotomy for large or complex stone disease: a treatment strategy. J Endourol. 2005;19:348-352.
Kirkali Z, Esen AA, Mungan MU. Effectiveness of extracorporeal shockwave lithotripsy in the management of stone-bearing horseshoe kidneys. J Endourol. 1996;10:13-15.
Klee LW, Brito CG, Lingeman JE. The clinical implications of brushite calculi. J Urol. 1991;145:715-718.
Kletscher BA, Segura JW, LeRoy AJ, et al. Percutaneous antegrade endopyelotomy: review of 50 consecutive cases. J Urol. 1995;153(Pt. 1):701-703.
Knapp PK, Scott J, Lingeman JE. Magnetic resonance imaging following extracorporeal shock wave lithotripsy with the Dornier HM3 lithotripter. J Urol. 1987;137:287.
Knapp PM, Kulb TB, Lingeman JE, et al. Extracorporeal shock wave lithotripsy–induced perirenal hematomas. J Urol. 1988;139:700-703.
Knispel HH, Klan R, Heicappell R, et al. Pneumatic lithotripsy applied through deflected working channel of miniureteroscope: results in 143 patients. J Endourol. 1998;12:513-515.
Knoll LD, Segura JW, Patterson DE, et al. Long-term followup in patients with cystine urinary calculi treated by percutaneous ultrasonic lithotripsy. J Urol. 1988;140:246-248.
Knudsen BE, Cook AJ, Watterson JD, et al. Percutaneous antegrade endopyelotomy: long-term results from one institution. Urology. 2004;63:230-234.
Koga S, Arakaki Y, Matsuoka M, et al. Staghorn calculi—long-term results of management. Br J Urol. 1991;68:122-124.
Kohan AD, Armenakas NA, Fracchia JA. Indinavir urolithiasis: an emerging cause of renal colic in patients with human immunodeficiency virus. J Urol. 1999;161:1765-1768.
Kohrmann KU, Rassweiler J, Alken P. The recurrence rate of stones following ESWL. World J Urol. 1993;11:26-30.
Kohrmann KU, Rassweiler JJ, Manning M, et al. The clinical introduction of a third generation lithotriptor: Modulith SL 20. J Urol. 1995;153:1379-1383.
Koo BC, Burtt G, Burgess NA. Percutaneous stone surgery in the obese: outcome stratified according to body mass index. BJU Int. 2004;93:1296-1299.
Kourambas J, Byrne RR, Preminger GM. Does a ureteral access sheath facilitate ureteroscopy? J Urol. 2001;165:789-793.
Kourambas J, Delvecchio FC, Munver R, et al. Nitinol stone retrieval-assisted ureteroscopic management of lower pole renal calculi. Urology. 2000;56:935-939.
Krambeck AE, Gettman MT, BaniHani AH, et al. Management of nephrolithiasis after Cohen cross-trigonal and Glenn-Anderson advancement ureteroneocystostomy. J Urol. 2007;177:174-178.
Krambeck AE, Gettman MT, Rohlinger AL, et al. Diabetes mellitus and hypertension associated with shock wave lithotripsy of renal and proximal ureteral stones at 19 years of followup. J Urol. 2006;175:1742-1747.
Krambeck AE, Murat FJ, Gettman MT, et al. The evolution of ureteroscopy: a mondern single-institution series. Mayo Clin Proc. 2006;81(4):468-473.
Krambeck AE, Rohlinger AL, Lohse CM, et al. Long term effects of shock wave lithotripsy for nephrolithiasis: a nineteen year study. J Endourol. 2005;19(Suppl.):A33.
Kuo RL, Lingeman JE, Leveillee RJ, et al. A randomized clinical trial of ureteroscopy and percutaneous nephrolithotomy for lower pole stones between 11 and 25 mm. J Endourol. 2003;17:A31.
Kuo RL, Paterson RF, Siqueira TMJr, et al. In vitro assessment of ultrasonic lithotriptors. J Urol. 2003;170:1101-1104.
Kuo RL, Paterson RF, Siqueira TMJr, et al. In vitro assessment of lithoclast ultra intracorporeal lithotripter. J Endourol. 2004;18:153-156.
Kupeli B, Irkilata L, Gurocak S, et al. Does tamsulosin enhance lower ureteral stone clearance with or without shock wave lithotripsy? Urology. 2004;64(6):1111-1115.
Kupeli B, Isen K, Biri H, et al. Extracorporeal shockwave lithotripsy in anomalous kidneys. J Endourol. 1999;13:349-352.
Kuwahara M, Kambe K, Kurosu S, et al. Clinical application of extracorporeal shock wave lithotripsy using microexplosions. J Urol. 1987;137:837-840.
Laing FC, Benson CB, DiSalvo DN, et al. Distal ureteral calculi: detection with vaginal US. Radiology. 1994;192:545-548.
Lam HS, Lingeman JE, Barron M, et al. Staghorn calculi: analysis of treatment results between initial percutaneous nephrostolithotomy and extracorporeal shock wave lithotripsy monotherapy with reference to surface area. J Urol. 1992;147:1219-1225.
Lam HS, Lingeman JE, Mosbaugh PG, et al. Evolution of the technique of combination therapy for staghorn calculi: a decreasing role for extracorporeal shock wave lithotripsy. J Urol. 1992;148(Pt. 2):1058-1062.
Lam HS, Lingeman JE, Russo R, et al. Stone surface area determination techniques: a unifying concept of staghorn stone burden assessment. J Urol. 1992;148(Pt. 2):1026-1029.
Lam JS, Greene TD, Gupta M. Treatment of proximal ureteral calculi: holmium:YAG laser ureterolithotripsy versus extracorporeal shock wave lithotripsy. J Urol. 2002;167:1972-1976.
Lampel A, Hohenfellner M, Schultz-Lampel D, et al. Urolithiasis in horseshoe kidneys: therapeutic management. Urology. 1996;47:182-186.
Lane BR, Singh D, Meraney A, Streem SB. Novel endourologic applications for holmium laser. Urology. 2005;65:991-993.
Lang EK. Percutaneous nephrostolithotomy and lithotripsy: a multi-institutional survey of complications. Radiology. 1987;162(Pt. 1):25-30.
Larsen EH, Gasser TC, Madsen PO. Antimicrobial prophylaxis in urologic surgery. Urol Clin North Am. 1986;13:591-604.
Lee WJ, Smith AD, Cubelli V, et al. Percutaneous nephrolithotomy: analysis of 500 consecutive cases. Urol Radiol. 1986;8:61-66.
Lee WJ, Smith AD, Cubelli V, et al. Complications of percutaneous nephrolithotomy. AJR Am J Roentgenol. 1987;148:177-180.
Lee YH, Huang WC, Tsai JY, et al. The efficacy of potassium citrate–based medical prophylaxis for preventing upper urinary tract calculi: a midterm followup study. J Urol. 1999;161:1453-1457.
Lee YH, Tsai JY, Jiaan BP, et al. Prospective randomized trial comparing shock wave lithotripsy and ureteroscopic lithotripsy for management of large upper third ureteral stones. Urology. 2006;67:480-484.
LeRoy AJ, Williams HJJr, Bender CE, et al. Colon perforation following percutaneous nephrostomy and renal calculus removal. Radiology. 1985;155:83-85.
L’Esperance JO, Ekeruo WO, Scales CDJr, et al. Effect of ureteral access sheath on stone-free rates in patients undergoing ureteroscopic management of renal calculi. Urology. 2005;66:252-255.
Lewis DF, Robichaux AG3rd, Jaekle RK, et al. Urolithiasis in pregnancy: diagnosis, management and pregnancy outcome. J Reprod Med. 2003;48:28-32.
Liatsikos EN, Bernardo NO, Dinlenc CZ, et al. Caliceal diverticular calculi: is there a role for metabolic evaluation? J Urol. 2000;164:18-20.
Liatsikos EN, Dinlenc CZ, Fogarty JD, et al. Efficiency and efficacy of different intracorporeal ultrasonic lithotripsy units on a synthetic stone model. J Endourol. 2001;15:925-928.
Liedle B, Jocham D, Lunz C, et al. Five year follow-up of urinary stone treatment with extracorporeal shock wave lithotripsy. J Endourol. 1988;2:157-162.
Lifshitz DA, Lingeman JE, Zafar FS, et al. Alterations in predicted growth rates of pediatric kidneys treated with extracorporeal shockwave lithotripsy. J Endourol. 1998;12:469-475.
Lifshitz DA, Shalhav AL, Lingeman JE, et al. Metabolic evaluation of stone disease patients: a practical approach. J Endourol. 1999;13:669-678.
Lifshitz DA, Winkler HZ, Gross M, et al. Predictive value of urinary cultures in assessment of microbial colonization of ureteral stents. J Endourol. 1999;13:735-738.
Lingeman JE. Stones. J Urol. 1995;153:1408.
Lingeman JE. Extracorporeal shock wave lithotripsy: development, instrumentation, and current status. Urol Clin North Am. 1997;24:185-211.
Lingeman JE, Coury TA, Newman DM, et al. Comparison of results and morbidity of percutaneous nephrostolithotomy and extracorporeal shock wave lithotripsy. J Urol. 1987;138:485-490.
Lingeman JE, Kulb TB, Newman DM, et al. Hypertension following ESWL. J Urol. 1987;137(Suppl.):142.
Lingeman JE, Newman D, Mertz JH, et al. Extracorporeal shock wave lithotripsy: the Methodist Hospital of Indiana experience. J Urol. 1986;135:1134-1137.
Lingeman JE, Saywell RMJr, Woods JR, et al. Cost analysis of extracorporeal shock wave lithotripsy relative to other surgical and nonsurgical treatment alternatives for urolithiasis. Med Care. 1986;24:1151-1160.
Lingeman JE, Siegel YI, Steele B. Metabolic evaluation of infected renal lithiasis: clinical relevance. J Endourol. 1995;9:51-54.
Lingeman JE, Siegel YI, Steele B, et al. Management of lower pole nephrolithiasis: a critical analysis. J Urol. 1994;151:663-667.
Lingeman JE, Woods JR, Toth PD. Blood pressure changes following extracorporeal shock wave lithotripsy and other forms of treatment for nephrolithiasis. JAMA. 1990;263:1789-1794.
Locke DR, Newman RC, Steinbock GS, et al. Extracorporeal shock-wave lithotripsy in horseshoe kidneys. Urology. 1990;35:407-411.
Logarakis NF, Jewett MA, Luymes J, et al. Variation in clinical outcome following shock wave lithotripsy. J Urol. 2000;163:721-725.
Lokhandwalla M, Sturtevant B. Fracture mechanics model of stone comminution in ESWL and implications for tissue damage. Phys Med Biol. 2000;45:1923-1940.
Lotan Y, Gettman MT, Roehrborn CG, et al. Management of ureteral calculi: a cost comparison and decision making analysis. J Urol. 2002;167:1621-1629.
Loughlin KR. Management of urologic problems during pregnancy. Urology. 1994;44:159-169.
Lukasewycz S, Skenazy J, Hoffman N, et al. Comparison of nitinol tipless stone baskets in an in vitro caliceal model. J Urol. 2004;172:562-564.
MacNeily AE, Goldenberg SL, Allen GJ, et al. Sonographic visualization of the ureter in pregnancy. J Urol. 1991;146:298-301.
Madbouly K, Sheir KZ, Elsobky E. Impact of lower pole renal anatomy on stone clearance after shock wave lithotripsy: fact or fiction? J Urol. 2001;165:1415-1418.
Madbouly K, Sheir KZ, Elsobky E, et al. Risk factors for the formation of a steinstrasse after extracorporeal shock wave lithotripsy: a statistical model. J Urol. 2002;167:1239-1242.
Maheshwari PN, Oswal AT, Andankar M, et al. Is antegrade ureteroscopy better than retrograde ureteroscopy for impacted large upper ureteral calculi? J Endourol. 1999;13:441-444.
Maikranz P, Coe FL, Parks J, et al. Nephrolithiasis in pregnancy. Am J Kidney Dis. 1987;9:354-358.
Mak S, Amoroso P. Stop those antiplatelet drugs before surgery!. BJU Int. 2003;91:593-594.
Makarov DV, Trock BJ, Allaf ME, Matlaga BR. The effect of ureteral stent placement on post-ureteroscopy complications: a meta-analysis. Urology. 2008;71:796-800.
Makhlouf AA, Thorner D, Ugarte R, Monga M. Shock wave lithotripsy not associated with development of diabetes mellitus at 6 years of follow-up. Urology. 2009;73:4-8. discussion 8
Mandel N, Mandel I, Fryjoff K, et al. Conversion of calcium oxalate to calcium phosphate with recurrent stone episodes. J Urol. 2003;169:2026-2029.
Marberger M. Disintegration of renal and ureteral calculi with ultrasound. Urol Clin North Am. 1983;10:729-742.
Marberger M, Turk C, Steinkogler I. Painless piezoelectric extracorporeal lithotripsy. J Urol. 1988;139:695-699.
Mardis HK, Parks JH, Muller G, et al. Outcome of metabolic evaluation and medical treatment for calcium nephrolithiasis in a private urological practice. J Urol. 2004;171:85-88.
Mariani AJ. Combined electrohydraulic and holmium:YAG laser ureteroscopic nephrolithotripsy of large (greater than 4 cm) renal calculi. J Urol. 2007;177:168-173. discussion 173
Mariappan P, Smith G, Bariol SV, et al. Stone and pelvic urine culture and sensitivity are better than bladder urine as predictors of urosepsis following percutaneous nephrolithotomy: a prospective clinical study. J Urol. 2005;173:1610-1614.
Marshall S. Coagulum pyelolithotomy. Urol Clin North Am. 1983;10:659-664.
Marshall VF. Fiber optics in urology. J Urol. 1964;91:110-114.
Martin JH, Rosser CJ, Linebach RF, et al. Are coagulation studies necessary before percutaneous nephrostomy? Tech Urol. 2000;6:205-207.
Matin SF, Streem SB. Metabolic risk factors in patients with ureteropelvic junction obstruction and renal calculi. J Urol. 2000;163(6):1676-1678.
Matlaga BR, Assimos DG. Changing indications of open stone surgery. Urology. 2002;59:490-493. discussion 493–4
Matlaga BR, Hodges SJ, Shah OD, et al. Percutaneous nephrostolithotomy: predictors of length of stay. J Urol. 2004;172(Pt. 1):1351-1354.
Matlaga BR, Kawamoto S, Fishman E. Dual source computed tomography: a novel technique to determine stone composition. Urology. 2008;72:1164-1168.
Matlaga BR, Kim SC, Watkins SL, et al. Percutaneous nephrolithotomy for ectopic kidneys: over, around, or through. Urology. 2006;67:513-517.
Matlaga BR, Miller NL, Terry C, et al. The pathogenesis of calyceal diverticular calculi. Urol Res. 2007;35:35-40.
Matlaga BR, Shah OD, Zagoria RJ, et al. Computerized tomography guided access for percutaneous nephrostolithotomy. J Urol. 2003;170:45-47.
McAleer IM, Kaplan GW, Bradley JS, et al. Staghorn calculus endotoxin expression in sepsis. Urology. 2002;59:601.
McAleer IM, Kaplan GW, Bradley JS, et al. Endotoxin content in renal calculi. J Urol. 2003;169:1813-1814.
McCullough DL. Extracorporeal shock wave lithotripsy and residual stone fragments in lower calices. J Urol. 1989;141:140.
Menezes P, Kumar PV, Timoney AG. A randomized trial comparing lithoclast with an electrokinetic lithotripter in the management of ureteric stones. BJU Int. 2000;85:22-25.
Meretyk S, Bigg S, Clayman RV, et al. Caveat emptor: caliceal stones and the missing calix. J Urol. 1992;147:1091-1095.
Meretyk S, Gofrit ON, Gafni O, et al. Complete staghorn calculi: random prospective comparison between extracorporeal shock wave lithotripsy monotherapy and combined with percutaneous nephrostolithotomy. J Urol. 1997;157:780-786.
Michel W, Funke PJ, Tunn UW, et al. Pyelocalyceal diverticula. Int Urol Nephrol. 1985;17:225-230.
Middleton AWJr, Pfister RC. Stone-containing pyelocaliceal diverticulum: embryogenic, anatomic, radiologic and clinical characteristics. J Urol. 1974;111:2-6.
Miller NL, Matlaga BR, Handa SE, et al. The presence of horseshoe kidney does not affect the outcome of percutaneous nephrolithotomy. J Endourol. 2008;22:1219-1225.
Miller OF, Kane CJ. Time to stone passage for observed ureteral calculi: a guide for patient education. J Urol. 1999;162(Pt. 1):688-690. discussion 690–1
Miller RA, Whitfield HN. Lithotripsy for renal stone disease. BMJ. 1985;291:967.
Miller SD, Ng CS, Streem SB, et al. Laparoscopic management of caliceal diverticular calculi. J Urol. 2002;167:1248-1252.
Mogensen P, Andersen JT. Primary in situ extracorporeal shock wave lithotripsy for ureteral calculi. Scand J Urol Nephrol Suppl. 1994;157:159-163.
Monga M, Smith R, Ferral H, et al. Percutaneous ablation of caliceal diverticulum: long-term followup. J Urol. 2000;163:28-32.
Monk TG, Boure B, White PF, et al. Comparison of intravenous sedative-analgesic techniques for outpatient immersion lithotripsy. Anesth Analg. 1991;72:616-621.
Montgomery G, Cole R, Palfrey E. Does extracorporeal shock wave lithotripsy cause hypertension? Br J Urol. 1989;64:567-571.
Morse RM, Resnick MI. Ureteral calculi: natural history and treatment in an era of advanced technology. J Urol. 1991;145:263-265.
Mostafavi MR, Ernst RD, Saltzman B. Accurate determination of chemical composition of urinary calculi by spiral computerized tomography. J Urol. 1998;159:673-675.
Motola JA, Badlani GH, Smith AD. Results of 212 consecutive endopyelotomies: an 8-year followup. J Urol. 1993;149:453-456.
Muller M. Comparison of Dornier lithotripters: measurement of shock wave fields and fragmentation effectiveness. Biomed Tech. 1990;35:250-262.
Mulvaney WP. Attempted disintegration of calculi by ultrasonic vibrations. J Urol. 1953;70:704-707.
Mulvaney WP, Beck CW. The laser beam in urology. J Urol. 1968;99:112-115.
Murphy LJT. The history of urology. Springfield (IL): Charles C. Thomas; 1972.
Murray MJ, Chandhoke PS, Berman CJ, et al. Outcome of extracorporeal shockwave lithotripsy monotherapy for large renal calculi: effect of stone and collecting system surface areas and cost-effectiveness of treatment. J Endourol. 1995;9:9-13.
Murthy PV, Rao HS, Meherwade S, et al. Ureteroscopic lithotripsy using mini-endoscope and Swiss lithoclast: experience in 147 cases. J Endourol. 1997;11:327-330.
Nabi G, Cook J, N’Dow J, McClinton S. Outcomes of stenting after uncomplicated ureteroscopy: systematic review and meta-analysis. BMJ. 2007;334:572.
Nadler RB, Monk TG, Elashry O, et al. Simultaneous bilateral percutaneous nephrolithotomy with subarachnoid spinal anesthesia. J Endourol. 1998;12:27-31.
Nadler RB, Rao GS, Pearle MS, et al. Acucise endopyelotomy: assessment of long-term durability. J Urol. 1996;156:1094-1097. discussion 1097–8
Nadler RB, Rubenstein JN, Eggener SE, et al. The etiology of urolithiasis in HIV infected patients. J Urol. 2003;169:475-477.
Nadler RB, Stern JA, Kimm S, et al. Coronal imaging to assess urinary tract stone size. J Urol. 2004;172:962-964.
Nakamoto T, Sagami K, Yamasaki A, et al. Long-term results of endourologic treatment of urinary calculi: investigation of risk factors for recurrence or regrowth. J Endourol. 1993;7:297-301.
Natalin R, Xavier K, Okeke Z, Gupta M. Impact of obesity on ureteroscopic laser lithotripsy of urinary tract calculi. Int Braz J Urol. 2009;35:36-41. discussion 41–2
Nelson CP, Francis TA, Wolf JSJr. Comparison of shockwave lithotripsy outcomes in patients receiving sufentanil or lidocaine spinal anesthesia. J Endourol. 2001;15:473-477.
Netto NRJr, Claro JF, Lemos GC, et al. Renal calculi in lower pole calices: what is the best method of treatment? J Urol. 1991;146:721-723.
Neucks JS, Pishchalnikov YA, Zancanaro AJ, et al. Improved acoustic coupling for shock wave lithotripsy. Urol Res. 2008;36:61-66.
Newman DM, Lingeman JE, Mosbaugh PM, et al. Two year follow up of patients treated with extracorporeal shock wave lithotripsy. J Endourol. 1988;2:163-171.
Nicely ER, Maggio MI, Kuhn EJ. The use of a cystoscopically placed cobra catheter for directed irrigation of lower pole caliceal stones during extracorporeal shock wave lithotripsy. J Urol. 1992;148(Pt. 2):1036-1039.
Nijman RJ, Ackaert K, Scholtmeijer RJ, et al. Long-term results of extracorporeal shock wave lithotripsy in children. J Urol. 1989;142(Pt. 2):609-611. discussion 619
Obek C, Onal B, Kantay K, et al. The efficacy of extracorporeal shock wave lithotripsy for isolated lower pole calculi compared with isolated middle and upper caliceal calculi. J Urol. 2001;166:2081-2084. discussion 2085
O’Connor KP, Choyke PL, Pahira JJ. Matrix urinary calculi. J Endourol. 1990;29:265-267.
Ogan K, Corwin TS, Smith T, et al. Sensitivity of chest fluoroscopy compared with chest CT and chest radiography for diagnosing hydropneumothorax in association with percutaneous nephrostolithotomy. Urology. 2003;62:988-992.
Ogan K, Pearle MS. Oops we got in the chest: fluoroscopic chest tube insertion for hydrothorax after percutaneous nephrostolithotomy. Urology. 2002;60:1098-1099.
Ohlsen H, Kinn AC. Percutaneous extraction of upper urinary calculi under fluoroscopic control—still a valuable complement to ESWL. Scand J Urol Nephrol. 1993;27:311-321.
Older RA, Jenkins AD. Stone disease. Urol Clin North Am. 2000;27:215-229. vii
Orestano F, Caronia N, Gallo G, et al. Functional aspects of the kidney after shock wave lithotripsy. In: Lingeman JE, Newman DM, editors. Shock wave lithotripsy 2: urinary and biliary lithotripsy. New York: Plenum Press; 1989:15-17.
Ozcan S, Yilmaz E, Buyukkocak U, et al. Comparison of three analgesics for extracorporeal shock wave lithotripsy. Scand J Urol Nephrol. 2002;36:281-285.
Pace KT, Tariq N, Dyer SJ, et al. Mechanical percussion, inversion and diuresis for residual lower pole fragments after shock wave lithotripsy: a prospective, single blind, randomized controlled trial. J Urol. 2001;166:2065-2071.
Pace KT, Weir MJ, Tariq N, et al. Individual patient variation and inter-rater variability of lower calyceal infundibular width on routine intravenous pyelography. J Urol. 2000;163(Suppl.):341.
Paik ML, Wainstein MA, Spirnak JP, et al. Current indications for open stone surgery in the treatment of renal and ureteral calculi. J Urol. 1998;159:374-378. discussion 378–9
Paller MS, Ferris TF. The kidney and hypertension in pregnancy. In: Brenner BM, editor. Brenner and Rector’s the kidney. 5th ed. Philadelphia: WB Saunders; 1996:1731-1736.
Pardalidis NP, Waltzer WC, Tellis VA, et al. Endourologic management of complications in renal allografts. J Endourol. 1994;8:321-327.
Pareek G, Hedican SP, Lee FTJr, Nakada SY. Shock wave lithotripsy success determined by skin-to-stone distance on computed tomography. Urology. 2005;66:941-944.
Park S, Pearle MS. Imaging for percutaneous renal access and management of renal calculi. Urol Clin North Am. 2006;33:353-364.
Parker BD, Frederick RW, Reilly TP, et al. Efficiency and cost of treating proximal ureteral stones: shock wave lithotripsy versus ureteroscopy plus holmium:yttrium-aluminum-garnet laser. Urology. 2004;64(6):1102-1106.
Parks JH, Worcester EM, Coe FL, et al. Clinical implications of abundant calcium phosphate in routinely analyzed kidney stones. Kidney Int. 2004;66:777-785.
Parr KL, Lingeman JE, Jordan M, et al. Creatinine kinase concentrations and electrocardiographic changes in extracorporeal shock-wave lithotripsy. Urology. 1988;32:21-23.
Parulkar BG, Hopkins TB, Wollin MR, et al. Renal colic during pregnancy: a case for conservative treatment. J Urol. 1998;159:365-368.
Paterson RF, Kuo RL, Lingeman JE. Staghorn calculus endotoxin expression in sepsis. Urology. 2003;62:197. author reply 198
Patterson DE, Segura JW, LeRoy AJ. Long term follow-up of patients treated by percutaneous ultrasonic lithotripsy for struvite staghorn calculi. J Endourol. 1987;1:177-180.
Pearle MS, Lingeman JE, Leveillee R, et al. Prospective, randomized trial comparing shock wave lithotripsy and ureteroscopy for lower pole caliceal calculi 1 cm or less. J Urol. 2005;173:2005-2009.
Pearle MS, Nadler R, Bercowsky E, et al. Prospective randomized trial comparing shock wave lithotripsy and ureteroscopy for management of distal ureteral calculi. J Urol. 2001;166:1255-1260.
Pearle MS, Nakada SY, Womack JS, et al. Outcomes of contemporary percutaneous nephrostolithotomy in morbidly obese patients. J Urol. 1998;160(Pt. 1):669-763.
Pearle MS, Watamull LM, Mullican MA. Sensitivity of noncontrast helical computerized tomography and plain film radiography compared to flexible nephroscopy for detecting residual fragments after percutaneous nephrostolithotomy. J Urol. 1999;162:23-26.
Perks AE, Schuler TD, Lee J, et al. Stone attenuation and skin-to-stone distance on computed tomography predicts for stone fragmentation by shock wave lithotripsy. Urology. 2008;72:765-769.
Perlmutter A, Miller L, Trimble LA, et al. Toradol, an NSAID used for renal colic, decreases renal perfusion and ureteral pressure in a canine model of unilateral ureteral obstruction. J Urol. 1993;149:926-930.
Peterson JC, Finlayson B. Effects of ESWL on blood pressure. In: Gravenstein JS, Peter K, editors. Extracorporeal shock wave lithotripsy for renal stone disease: technical and clinical aspects. Boston: Butterworths, 1986.
Pettersson B, Tiselius HG, Andersson A, et al. Evaluation of extracorporeal shock wave lithotripsy without anesthesia using a Dornier HM3 lithotriptor without technical modifications. J Urol. 1989;142:1189-1192.
Piergiovanni M, Desgrandchamps F, Cochand-Priollet B, et al. Ureteral and bladder lesions after ballistic, ultrasonic, electrohydraulic, or laser lithotripsy. J Endourol. 1994;8:293-299.
Pietrow PK, Auge BK, Weizer AZ, et al. Durability of the medical management of cystinuria. J Urol. 2003;169:68-70.
Pietrow PK, Auge BK, Zhong P, Preminger GM. Clinical efficacy of a combination pneumatic and ultrasonic lithotrite. J Urol. 2003;169:1247-1249.
Piper NY, Dalrymple N, Bishoff JT. Incidence of renal hematoma formation after ESWL using the new Dornier Doli-S lithotriptor. J Urol. 2001;165:377.
Pishchalnikov YA, Neucks JS, VonDerHaar RJ, et al. Air pockets trapped during routine coupling in dry head lithotripsy can significantly decrease the delivery of shock wave energy. J Urol. 2006;176:2706-2710.
Pittomvils G, Vandeursen H, Wevers M, et al. The influence of internal stone structure upon the fracture behaviour of urinary calculi. Ultrasound Med Biol. 1994;20:803-810.
Pollack HM, Banner MP. Percutaneous extraction of renal and ureteral calculi: technical considerations. AJR Am J Roentgenol. 1984;143:778-784.
Poon M, Beaghler M, Baldwin D. Flexible endoscope deflectability: changes using a variety of working instruments and laser fibers. J Endourol. 1997;11:247-249.
Portis AJ, Rygwall R, Holtz C, et al. Ureteroscopic laser lithotripsy for upper urinary tract calculi with active fragment extraction and computerized tomography followup. J Urol. 2006;175:2129-2133. discussion 2133–4
Portis AJ, Yan Y, Pattaras JG, et al. Matched pair analysis of shock wave lithotripsy effectiveness for comparison of lithotriptors. J Urol. 2003;169:58-62.
Preminger GM, Assimos DG, Lingeman JE, et al. Chapter 1: AUA guideline on management of staghorn calculi: diagnosis and treatment recommendations. J Urol. 2005;173:1991-2000.
Preminger GM, Clayman RV, Curry T, et al. Outpatient percutaneous nephrostolithotomy. J Urol. 1986;136:355-357.
Preminger GM, Clayman RV, Hardeman SW, et al. Percutaneous nephrostolithotomy vs open surgery for renal calculi: a comparative study. JAMA. 1985;254:1054-1058.
Preminger GM, Kettelhut MC, Elkins SL, et al. Ureteral stenting during extracorporeal shock wave lithotripsy: help or hindrance? J Urol. 1989;142:32-36.
Preminger GM, Tiselius HG, Assimos DG, et al. 2007 guideline for the management of ureteral calculi. J Urol. 2007;178:2418-2434.
Psihramis KE, Jewett MA, Bombardier C, et al. Lithostar extracorporeal shock wave lithotripsy: the first 1,000 patients. Toronto Lithotripsy Associates. J Urol. 1992;147:1006-1009.
Puppo P, Germinale F, Ricciotti G. Hypertension after extracorporeal shock wave lithotripsy: a false alarm. J Endourol. 1988;3:401-404.
Raboy A, Ferzli GS, Ioffreda R, et al. Laparoscopic ureterolithotomy. Urology. 1992;39:223-225.
Raj GV, Auge BK, Weizer AZ, et al. Percutaneous management of calculi within horseshoe kidneys. J Urol. 2003;170:48-51.
Ramakumar S, Lancini V, Chan DY, et al. Laparoscopic pyeloplasty with concomitant pyelolithotomy. J Urol. 2002;167:1378-1380.
Raman JD, Bagrodia A, Gupta A, et al. Natural history of residual fragments following percutaneous nephrostolithotomy. J Urol. 2009;181:1163-1168.
Ramsay JW, Miller RA, Kellett MJ, et al. Percutaneous pyelolysis: indications, complications and results. Br J Urol. 1984;56:586-588.
Rane A, Rao P, Rao P. Single-port-access nephrectomy and other laparoscopic urologic procedures using a novel laparoscopic port (R-port). Urology. 2008;72:260-263. discussion 263–4
Raney AM. Electrohydraulic ureterolithotripsy: preliminary report. Urology. 1978;12:284-285.
Raney AM, Handler J. Electrohydraulic nephrolithotripsy. Urology. 1975;6:439-442.
Rasmussen PE, Nielsen FR. Hydronephrosis during pregnancy: a literature survey. Eur J Obstet Gynecol Reprod Biol. 1988;27:249-259.
Redman JF. Partial nephrectomy. Urol Clin North Am. 1983;10:677-684.
Rehman J, Monga M, Landman J, et al. Characterization of intrapelvic pressure during ureteropyeloscopy with ureteral access sheaths. Urology. 2003;61:713-718.
Reid G, Denstedt JD, Kang YS, et al. Microbial adhesion and biofilm formation on ureteral stents in vitro and in vivo. J Urol. 1992;148:1592-1594.
Reiter WJ, Schon-Pernerstorfer H, Dorfinger K, et al. Frequency of urolithiasis in individuals seropositive for human immunodeficiency virus treated with indinavir is higher than previously assumed. J Urol. 1999;161:1082-1084.
Renner C, Rassweiler J. Treatment of renal stones by extracorporeal shock wave lithotripsy. Nephron. 1999;81(Suppl. 1):71-81.
Resim S, Ekerbicer HC, Kiran G, Kilinc M. Are changes in urinary parameters during pregnancy clinically significant? Urol Res. 2006;34:244-248.
Rhee BK, Bretan PNJr, Stoller ML. Urolithiasis in renal and combined pancreas/renal transplant recipients. J Urol. 1999;161:1458-1462.
Ricchiuti DJ, Smaldone MC, Jacobs BL, et al. Staged retrograde endoscopic lithotripsy as alternative to PCNL in select patients with large renal calculi. J Endourol. 2007;21:1421-1424.
Rifaioglu MM, Berger AD, Pengune W, Stoller ML. Percutaneous management of stones in transplanted kidneys. Urology. 2008;72:508-512.
Roberts WW, Cadeddu JA, Micali S, et al. Ureteral stricture formation after removal of impacted calculi. J Urol. 1998;159:723-726.
Rocco F, Mandressi A, Larcher P. Surgical classification of renal calculi. Eur Urol. 1984;10:121-123.
Rodriguez PN, Klein AS. Management of urolithiasis during pregnancy. Surg Gynecol Obstet. 1988;166:103-106.
Ross AE, Handa S, Lingeman JE, Matlaga BR. Kidney stones during pregnancy: an investigation into stone composition. Urol Res. 2008;36:99-102.
Rous SN, Turner WR. Retrospective study of 95 patients with staghorn calculus disease. J Urol. 1977;118:902-904.
Rouvalis P. Electronic lithotripsy for vesical calculus with “Urat-1”: an experience of 100 cases and an experimental application of the method to stones in the upper urinary tract. Br J Urol. 1970;42:486-491.
Roy C, Saussine C, Jahn C, et al. Fast imaging MR assessment of ureterohydronephrosis during pregnancy. Magn Reson Imaging. 1995;13:767-772.
Rubenstein JN, Gonzalez CM, Blunt LW, et al. Safety and efficacy of percutaneous nephrolithotomy in patients with neurogenic bladder dysfunction. Urology. 2004;63(4):636-640.
Ruckle HC, Segura JW. Laparoscopic treatment of a stone-filled, caliceal diverticulum: a definitive, minimally invasive therapeutic option. J Urol. 1994;151:122-124.
Rudnick DM, Bennett PM, Dretler SP. Retrograde renoscopic fragmentation of moderate-size (1.5-3.0-cm) renal cystine stones. J Endourol. 1999;13:483-485.
Ruiz Marcellan FJ, Ibarz Servio L. Evaluation of renal damage in extracorporeal lithotripsy by shock waves. Eur Urol. 1986;12:73-75.
Rupel E, Brown R. Nephroscopy with removal of stone following nephrostomy for obstructive calculus anuria. J Urol. 1941;46:177.
Rutchik SD, Resnick MI. Ureteropelvic junction obstruction and renal calculi: pathophysiology and implications for management. Urol Clin North Am. 1998;25:317-321.
Saltel E, Angel JB, Futter NG, et al. Increased prevalence and analysis of risk factors for indinavir nephrolithiasis. J Urol. 2000;164:1895-1897.
Sampaio FJ, Aragao AH. Inferior pole collecting system anatomy: its probable role in extracorporeal shock wave lithotripsy. J Urol. 1992;147:322-324.
Sampaio FJ, Aragao AH. Limitations of extracorporeal shockwave lithotripsy for lower caliceal stones: anatomic insight. J Endourol. 1994;8:241-247.
Santa-Cruz RW, Leveillee RJ, Krongrad A. Ex vivo comparison of four lithotripters commonly used in the ureter: what does it take to perforate? J Endourol. 1998;12:417-422.
Sapozhnikov OA, Cleveland RO, Bailey MR, et al. Modeling of stresses generated by lithotripter shock wave in cylindrical kidney stones. In: Chapelon JY, Lafon C, editors. Proceedings of the 3rd international symposium of therapeutic ultrasound. Lyon (France): INSERM; 2003:323-328.
Sapozhnikov OA, Maxwell AD, MacConaghy B, Bailey MR. A mechanistic analysis of stone fracture in lithotripsy. J Acoust Soc Am. 2007;121:1190-1202.
Sato Y, Tanda H, Kato S, et al. Shock wave lithotripsy for renal stones is not associated with hypertension and diabetes mellitus. Urology. 2008;71:586-591. discussion 591–2
Saw KC, Lingeman JE. Lesson 20: management of calyceal stones. AUA Update Series. 1999;20:154-159.
Saw KC, McAteer JA, Monga AG, et al. Helical CT of urinary calculi: effect of stone composition, stone size, and scan collimation. AJR Am J Roentgenol. 2000;175:329-332.
Scarpa RM, De Lisa A, Porru D, et al. Holmium:YAG laser ureterolithotripsy. Eur Urol. 1999;35:233-238.
Scherz HC, Parsons CL. Prophylactic antibiotics in urology. Urol Clin North Am. 1987;14:265-271.
Schulman A, Herlinger H. Urinary tract dilatation in pregnancy. Br J Radiol. 1975;48:638-645.
Schultheiss D, Engel RM, Crosby RW, et al. Max Brödel (1870-1941) and medical illustration in urology. J Urol. 2000;164:1137-1142.
Schuster TG, Hollenbeck BK, Faerber GJ, et al. Ureteroscopic treatment of lower pole calculi: comparison of lithotripsy in situ and after displacement. J Urol. 2002;168:43-45.
Schwartz G, Lipschitz S, Becker JA. Detection of renal calculi: the value of tomography. AJR Am J Roentgenol. 1984;143:143-145.
Segura JW. Surgical management of urinary calculi. Semin Nephrol. 1990;10:53-63.
Segura JW. Percutaneous nephrolithotomy: technique, indication, and complications. AUA Update Series. 12, 1993.
Segura JW. Staghorn calculi. Urol Clin North Am. 1997;24:71-80.
Segura JW. Intracorporeal lithotripsy. AUA Update Series. 1999;18:66-71.
Segura JW, Erickson SB, Wilson DM, et al. Infected renal lithiasis: results of long-term surgical and medical management. In: Smith LH, Robertson WG, Finlayson B, editors. Urolithiasis: clinical and basic research. New York: Plenum Press, 1981.
Segura JW, LeRoy AJ. Percutaneous ultrasonic lithotripsy. Urology. 1984;23(5 Spec. No.):7-10.
Segura JW, Patterson DE, LeRoy AJ, et al. Percutaneous removal of kidney stones: preliminary report. Mayo Clin Proc. 1982;57:615-619.
Segura JW, Preminger GM, Assimos DG, et al. Nephrolithiasis Clinical Guidelines Panel summary report on the management of staghorn calculi. The American Urological Association Nephrolithiasis Clinical Guidelines Panel. J Urol. 1994;151:1648-1651.
Segura JW, Preminger GM, Assimos DG, et al. Ureteral Stones Clinical Guidelines Panel summary report on the management of ureteral calculi. The American Urological Association. J Urol. 1997;158:1915-1921.
Selmy GI, Hassouna MM, Khalaf IM, et al. Effects of verapamil, prostaglandin F2 alpha, phenylephrine, and noradrenaline on upper urinary tract dynamics. Urology. 1994;43:31-35.
Semins MJ, Trock BJ, Matlaga BR. The effect of shock wave rate on the outcome of shock wave lithotripsy: a meta-analysis. J Urol. 2008;179:194-197. discussion 197
Semins MJ, Trock BJ, Matlaga BR. The safety of ureteroscopy during pregnancy: a systematic review and meta-analysis. J Urol. 2009;181:139-143.
Shah J, Whitfield HN. Urolithiasis through the ages. BJU Int. 2002;89:801-810.
Shalhav AL, Soble JJ, Nakada SY, et al. Long-term outcome of caliceal diverticula following percutaneous endosurgical management. J Urol. 1998;160:1635-1639.
Sheir KZ, Madbouly K, Elsobky E. Extracorporeal shock wave lithotripsy in anomalous kidneys: 11-year experience with two second-generation lithotripters. Urology. 2003;62:10-15.
Sherman JL, Hopper KD, Greene AJ, et al. The retrorenal colon on computed tomography: a normal variant. J Comput Assist Tomogr. 1985;9:339-341.
Shokeir AA, Abdulmaaboud M. Resistive index in renal colic: a prospective study. BJU Int. 1999;83:378-382.
Shokeir AA, El-Nahas AR, Shoma AM, et al. Percutaneous nephrolithotomy in treatment of large stones in horseshoe kidneys. Urology. 2004;64:426-429.
Shokeir AA, Mahran MR, Abdulmaaboud M. Renal colic in pregnant women: role of renal resistive index. Urology. 2000;55:344-347.
Shoskes DA, Hanbury D, Cranston D, et al. Urological complications in 1,000 consecutive renal transplant recipients. J Urol. 1995;153:18-21.
Silverman DE, Stamey TA. Management of infection stones: the Stanford experience. Medicine (Baltimore). 1983;62:44-51.
Silverstein AD, Terranova SA, Auge BK, et al. Bilateral renal calculi: assessment of staged v synchronous percutaneous nephrolithotomy. J Endourol. 2004;18:145-151.
Singal RK, Denstedt JD. Contemporary management of ureteral stones. Urol Clin North Am. 1997;24:59-70.
Singh M, Marshall V, Blandy J. The residual renal stone. Br J Urol. 1975;47:125-129.
Skoog SJ, Reed MD, Gaudier FAJr, et al. The posterolateral and the retrorenal colon: implication in percutaneous stone extraction. J Urol. 1985;134:110-112.
Smith AD, Lange PH, Fraley EE. Applications of percutaneous nephrostomy: new challenges and opportunities in endo-urology. J Urol. 1979;121:382.
Smith MJ, Boyce WH. Anatrophic nephrotomy and plastic calyrhaphy. J Urol. 1968;99:521-527.
Snyder JA, Smith AD. Staghorn calculi: percutaneous extraction versus anatrophic nephrolithotomy. J Urol. 1986;136:351-354.
Sofer M, Denstedt J. Flexible ureteroscopy and lithotripsy with the holmium:YAG laser. Can J Urol. 2000;7:952-956.
Sofer M, Watterson JD, Wollin TA, et al. Holmium:yag laser lithotripsy for upper urinary tract calculi in 598 patients. J Urol. 2002;167(1):31-34.
Sorensen C, Chandhoke P, Moore M, et al. Comparison of intravenous sedation versus general anesthesia on the efficacy of the Doli 50 lithotriptor. J Urol. 2002;168:35-37.
Soygur T, Akbay A, Kupeli S. Effect of potassium citrate therapy on stone recurrence and residual fragments after shockwave lithotripsy in lower caliceal calcium oxalate urolithiasis: a randomized controlled trial. J Endourol. 2002;16:149-152.
Sozen S, Kupeli B, Tunc L, et al. Management of ureteral stones with pneumatic lithotripsy: report of 500 patients. J Endourol. 2003;17(9):721-724.
Spencer JA, Chahal R, Kelly A, et al. Evaluation of painful hydronephrosis in pregnancy: Magnetic resonance urographic patterns in physiological dilatation versus calculous obstruction. J Urol. 2004;171:256-260.
Spirnak JP, Resnick MI. Anatrophic nephrolithotomy. Urol Clin North Am. 1983;10:665-675.
Spore SS, Teichman JM, Corbin NS, et al. Holmium:YAG lithotripsy: Optimal power settings. J Endourol. 1999;13:559-566.
Stein RJ, Turna B, Nguyen MM, et al. Laparoscopic pyeloplasty with concomitant pyelolithotomy: technique and outcomes. J Endourol. 2008;22:1251-1255.
Stonehill MA, Williams JC, Bailey MR, et al. An acoustically matched high pressure chamber for control of cavitation in shock wave lithotripsy: mechanisms of shock wave damage in vitro. Methods Cell Sci. 1998;19:303-310.
Stothers L, Lee LM. Renal colic in pregnancy. J Urol. 1992;148:1383-1387.
Streem SB, Yost A. Extracorporeal shock wave lithotripsy in patients with bleeding diatheses. J Urol. 1990;144:1347-1348.
Streem SB, Yost A. Treatment of caliceal diverticular calculi with extracorporeal shock wave lithotripsy: patient selection and extended followup. J Urol. 1992;148(Pt. 2):1043-1046.
Streem SB, Yost A, Dolmatch B. Combination “sandwich” therapy for extensive renal calculi in 100 consecutive patients: immediate, long-term and stratified results from a 10-year experience. J Urol. 1997;158:342-345.
Streem SB, Yost A, Mascha E. Clinical implications of clinically insignificant stone fragments after extracorporeal shock wave lithotripsy. J Urol. 1996;155:1186-1190.
Strohmaier WL, Schmidt J, Lahme S, et al. Arterial blood pressure following different types of urinary stone therapy. Presented at the 8th European Symposium on Urolithiasis, Parma, Italy, 1999. Eur Urol. 2000;38:753-757.
Sturtevant B. The shock wave physics of lithotripsy. In: Smith AD, editor. Smith’s textbook of endourology. St. Louis: Quality Medical; 1996:1-29.
Sulaiman MN, Buchholz NP, Clark PB. The role of ureteral stent placement in the prevention of steinstrasse. J Endourol. 1999;13:151-155.
Sumino Y, Mimata H, Tasaki Y, et al. Predictors of lower pole renal stones clearance after extracorporeal shock wave lithotripsy. J Urol. 2002;168:1344-1347.
Sundaram CP, Saltzman B. Urolithiasis associated with protease inhibitors. J Endourol. 1999;13:309-312.
Swanson SK, Heilman RL, Eversman WG. Urinary tract stones in pregnancy. Surg Clin North Am. 1995;75:123-142.
Swartz HM, Reichling BA. Hazards of radiation exposure for pregnant women. JAMA. 1978;239:1907-1908.
Swartz MA, Lydon-Rochelle MT, Simon D, et al. Admission for nephrolithiasis in pregnancy and risk of adverse birth outcomes. Obstet Gynecol. 2007;109:1099-1104.
Symons SJ, Ramachandran A, Kurien A, et al. Urolithiasis in the horseshoe kidney: a single-centre experience. BJU Int. 2008;102:1676-1680.
Tan MO, Kirac M, Onaran M, et al. Factors affecting the success rate of extracorporeal shock wave lithotripsy for renal calculi in children. Urol Res. 2006;34:215-221.
Tan PK, Tan SM, Consigliere D. Ureteroscopic lithoclast lithotripsy: a cost-effective option. J Endourol. 1998;12:341-344.
Taub DA, Suh RS, Faerber GJ, Wolf JSJr. Ureteroscopic laser papillotomy to treat papillary calcifications associated with chronic flank pain. Urology. 2006;67:683-687.
Teh CL, Zhong P, Preminger GM. Laboratory and clinical assessment of pneumatically driven intracorporeal lithotripsy. J Endourol. 1998;12:163-169.
Teichman JM, Champion PC, Wollin TA, et al. Holmium:YAG lithotripsy of uric acid calculi. J Urol. 1998;160(Pt. 1):2130-2132.
Teichman JM, Rao RD, Rogenes VJ, et al. Ureteroscopic management of ureteral calculi: electrohydraulic versus holmium:YAG lithotripsy. J Urol. 1997;158:1357-1361.
Teichman JM, Vassar GJ, Bishoff JT, et al. Holmium:YAG lithotripsy yields smaller fragments than lithoclast, pulsed dye laser or electrohydraulic lithotripsy. J Urol. 1998;159:17-23.
Teichman JM, Vassar GJ, Glickman RD. Holmium:yttrium-aluminum-garnet lithotripsy efficiency varies with stone composition. Urology. 1998;52:392-397.
Theiss M, Wirth MP, Frohmuller HG. Extracorporeal shock wave lithotripsy in patients with renal malformations. Br J Urol. 1993;72(Pt. 1):534-538.
Thiruchelvam N, Mostafid H, Ubhayakar G. Planning percutaneous nephrolithotomy using multidetector computed tomography urography, multiplanar reconstruction and three-dimensional reformatting. BJU Int. 2005;95:1280-1284.
Thornbury JR, Parker TW. Ureteral calculi. Semin Roentgenol. 1982;17:133-139.
Thuroff S, Thorsten B, Chaussy C. Anatomy related shockwave power using Siemens Lithostar multiline. J Urol. 1988;159(Suppl.):34.
Timmons JWJr, Malek RS, Hattery RR, et al. Caliceal diverticulum. J Urol. 1975;114:6-9.
Tiselius HG. Anesthesia-free in situ extracorporeal shock wave lithotripsy of ureteral stones. J Urol. 1991;146:8-12.
Tolley DA, Downey P. Current advances in shock wave lithotripsy. Curr Opin Urol. 1999;9:319-323.
Tolley DA, Wallace DM, Tiptaft RC. First UK consensus conference on lithotriptor terminology—1989. Br J Urol. 1991;67:9-12.
Trinchieri A, Montanari E, Zanetti G, Lizzano R. The impact of new technology in the treatment of cystine stones. Urol Res. 2007;35:129-132.
Tunc L, Tokgoz H, Tan MO, et al. Stones in anomalous kidneys: results of treatment by shock wave lithotripsy in 150 patients. Int J Urol. 2004;11:831-836.
Turk I, Deger S, Roigas J, et al. Laparoscopic ureterolithotomy. Tech Urol. 1998;4:29-34.
Turna B, Raza A, Moussa S, et al. Management of calyceal diverticular stones with extracorporeal shock wave lithotripsy and percutaneous nephrolithotomy: long-term outcome. BJU Int. 2007;100:151-156.
Turna B, Stein RJ, Smaldone MC, et al. Safety and efficacy of flexible ureterorenoscopy and holmium:YAG lithotripsy for intrarenal stones in anticoagulated cases. J Urol. 2008;179:1415-1419.
Ueda S, Matsuko K, Yamashita T, et al. Perirenal hematomas caused by SWL with EDAP LT-01 lithotriptor. J Endourol. 1993;7:11-15.
Ueno A, Kawamura T, Ogawa A, et al. Relation of spontaneous passage of ureteral calculi to size. Urology. 1977;10:544-546.
Ulrich JC, York JP, Koff SA. The renal vascular response to acutely elevated intrapelvic pressure: resistive index measurements in experimental urinary obstruction. J Urol. 1995;154:1202-1204.
Vakil N, Everbach EC. Transient acoustic cavitation in gallstone fragmentation: a study of gallstones fragmented in vivo. Ultrasound Med Biol. 1993;19:331-342.
Vakil N, Gracewski SM, Everbach EC. Relationship of model stone properties to fragmentation mechanisms during lithotripsy. J Lithotr Stone Dis. 1991;3:304-310.
Van Cangh PJ, Abi Aad AS, Lorge F, et al. Laparoscopic nephrolithotomy: the value of intracorporeal sonography and color Doppler. Urology. 1995;45:516-519.
Van Cangh PJ, Jorion JL, Wese FX, et al. Endoureteropyelotomy: percutaneous treatment of ureteropelvic junction obstruction. J Urol. 1989;141:1317-1321. discussion 1321–2
Van Cangh PJ, Wilmart JF, Opsomer RJ, et al. Long-term results and late recurrence after endoureteropyelotomy: a critical analysis of prognostic factors. J Urol. 1994;151:934-937.
Vandeursen H, Baert L. Electromagnetic extracorporeal shock wave lithotripsy for calculi in horseshoe kidneys. J Urol. 1992;148(Pt. 2):1120-1122.
Vassar GJ, Chan KF, Teichman JM, et al. Holmium:YAG lithotripsy: photothermal mechanism. J Endourol. 1999;13:181-190.
Vassar GJ, Teichman JM, Glickman RD. Holmium:YAG lithotripsy efficiency varies with energy density. J Urol. 1998;160:471-476.
Vaughan EDJr, Gillenwater JY. Recovery following complete chronic unilateral ureteral occlusion: functional, radiographic and pathologic alterations. J Urol. 1971;106:27-35.
Vorreuther R, Corleis R, Klotz T, et al. Impact of shock wave pattern and cavitation bubble size on tissue damage during ureteroscopic electrohydraulic lithotripsy. J Urol. 1995;153(Pt. 1):849-853.
Vorreuther R, Klotz T, Heidenreich A, et al. Pneumatic v electrokinetic lithotripsy in treatment of ureteral stones. J Endourol. 1998;12:233-236.
Waldmann TB, Lashley DB, Fuchs EF. Unenhanced computerized axial tomography to detect retained calculi after percutaneous ultrasonic lithotripsy. J Urol. 1999;162:312-314.
Wang LJ, Wong YC, Chuang CK, et al. Predictions of outcomes of renal stones after extracorporeal shock wave lithotripsy from stone characteristics determined by unenhanced helical computed tomography: a multivariate analysis. Eur Radiol. 2005;15:2238-2243.
Watterson JD, Girvan AR, Cook AJ, et al. Safety and efficacy of holmium:YAG laser lithotripsy in patients with bleeding diatheses. J Urol. 2002;168:442-445.
Weinerth JL, Flatt JA, Carson CC3rd. Lessons learned in patients with large steinstrasse. J Urol. 1989;142:1425-1427.
Weizer AZ, Auge BK, Silverstein AD, et al. Routine postoperative imaging is important ureteroscopic stone manipulation. J Urol. 2002;168:46-50.
Weizer AZ, Springhart WP, Ekeruo WO, et al. Ureteroscopic management of renal calculi in anomalous kidneys. Urology. 2005;65:265-269.
Wershub LP. Urology, from antiquity to the 20th century. St. Louis: WH Green; 1970.
Whelan JP, Finlayson B, Welch J, et al. The blast path: theoretical basis, experimental data and clinical application. J Urol. 1988;140:401-404.
White MD, Moran ME, Calvano CJ, et al. Evaluation of retropulsion caused by holmium:YAG laser with various power settings and fibers. J Endourol. 1998;12:183-186.
White WM, Zite NB, Gash J, et al. Low-dose computed tomography for the evaluation of flank pain in the pregnant population. J Endourol. 2007;21:1255-1260.
Wickham JE, Coe N, Ward JP. One hundred cases of nephrolithotomy under hypothermia. J Urol. 1974;112:702-705.
Wickham JE, Kellett MJ. Percutaneous nephrolithotomy. Br J Urol. 1981;53:297-299.
Wilbert DM, Reichenberger H, Noske E, et al. New generation shock wave lithotripsy. J Urol. 1987;138:563-565.
Williams CM, Kaude JV, Newman RC, et al. Extracorporeal shock-wave lithotripsy: long-term complications. AJR Am J Roentgenol. 1988;150:311-315.
Williams CM, Thomas WCJr. Permanently decreased renal blood flow and hypertension after lithotripsy. N Engl J Med. 1989;321:1269-1270.
Williams G, Blandy JP, Tresidder GC. Communicating cysts and diverticula of the renal pelvis. Br J Urol. 1969;41:163-170.
Williams JCJr, Paterson RF, Kopecky KK, et al. High resolution detection of internal structure of renal calculi by helical computerized tomography. J Urol. 2002;167:322-326.
Williams JCJr, Saw KC, Paterson RF, et al. Variability of renal stone fragility in shock wave lithotripsy. Urology. 2003;61:1092-1096. discussion 1097
Willis LR, Evan AP, Connors BA, et al. Prevention of lithotripsy-induced renal injury by pre-treating kidneys with low energy shockwaves. J Am Soc Nephrol. 2006;17:336-673.
Winfield HN, Donovan JF, Godet AS, et al. Laparoscopic partial nephrectomy: initial case report for benign disease. J Endourol. 1993;7:521-526.
Wirth MP, Theiss M, Frohmuller HG. Primary extracorporeal shock wave lithotripsy of staghorn renal calculi. Urol Int. 1992;48:71-75.
Wolf JSJr. Management of intraoperatively diagnosed colonic injury during percutaneous nephrostolithotomy. Tech Urol. 1998;4:160-164.
Wolf JSJr, Bennett CJ, Dmochowski RR, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol. 2008;179:1379-1390.
Wolf JSJr, Carroll PR, Stoller ML. Cost-effectiveness v patient preference in the choice of treatment for distal ureteral calculi: a literature-based decision analysis. J Endourol. 1995;9:243-248.
Wollin TA, Denstedt JD. The holmium laser in urology. J Clin Laser Med Surg. 1998;16:13-20.
Wu CF, Chen CS, Lin WY, et al. Therapeutic options for proximal ureter stone: extracorporeal shock wave lithotripsy versus semirigid ureterorenoscope with holmium:yttrium aluminum garnet laser lithotripsy. Urology. 2005;65:1075-1079.
Wuernschimmel E, Lipsky H. Laparoscopic treatment of an upper ureteral stone. J Laparoendosc Surg. 1993;3:301-307.
Wulfsohn MA. Pyelocaliceal diverticula. J Urol. 1980;123:1-8.
Wyler SF, Bachmann A, Jayet C, et al. Retroperitoneoscopic management of caliceal diverticular calculi. Urology. 2005;65:380-383.
Xi X, Zhong P. Dynamic photoelastic study of the transient stress field in solids during shock wave lithotripsy. J Acoust Soc Am. 2001;109:1226-1239.
Yang SS, Hong JS. Electrohydraulic lithotripsy of upper ureteral calculi with semirigid ureteroscope. J Endourol. 1996;10:27-30.
Yavascaoglu I, Yenihayat K, Oktay B, et al. Pneumatic lithotriptor—a useful tool for challenging renal stone surgery. Int Urol Nephrol. 1999;31:283-289.
Yilmaz S, Sindel T, Arslan G, et al. Renal colic: comparison of spiral CT, US and IVU in the detection of ureteral calculi. Eur Radiol. 1998;8:212-217.
Yokoyama M, Shoji F, Yanagizawa R. Blood pressure changes following extracorporeal shock wave lithotripsy for urolithiasis. J Urol. 1992;147:553-558.
Young HH, McKay RW. Congenital valvular obstruction of the prostatic urethra. Surg Gynecol Obstet. 1929;48:509.
Yow RM, Bunts RC. Calyceal diverticulum. J Urol. 1955;73:663-670.
Zafar FS, Lingeman JE. Value of laparoscopy in the management of calculi complicating renal malformations. J Endourol. 1996;10:379-383.
Zanetti G, Montanari E, Mandressi A, et al. Long-term results of extracorporeal shock wave lithotripsy in renal stone treatment. J Endourol. 1991;5:61.
Zanetti G, Seveso M, Montanari E, et al. Renal stone fragments following shock wave lithotripsy. J Urol. 1997;158:352-355.
Zeng GQ, Zhong WD, Cai YB, et al. Extracorporeal shock wave versus pneumatic ureteroscopic lithotripsy in treatment of lower ureteral calculi. Asian J Androl. 2002;4(4):303-305.
Zhong P, Chuong CJ. Propagation of shock waves in elastic solids caused by cavitation microjet impact. I. Theoretical formulation. J Acoust Soc Am. 1993;94:19-28.
Zhong P, Chuong CJ, Preminger GM. Propagation of shock waves in elastic solids caused by cavitation microjet impact. II. Application in extracorporeal shock wave lithotripsy. J Acoust Soc Am. 1993;94:29-36.
Zhong P, Tong HL, Cocks FH, et al. Transient oscillation of cavitation bubbles near stone surface during electrohydraulic lithotripsy. J Endourol. 1997;11:55-61.
Zhong P, Zhou Y, Zhu S. Dynamics of bubble oscillation in constrained media and mechanisms of vessel rupture in SWL. Ultrasound Med Biol. 2001;27:119-134.
Zhou Y, Cocks FH, Preminger GM, Zhong P. The effect of treatment strategy on stone comminution efficiency in shock wave lithotripsy. J Urol. 2004;172:349-354.
Zhu S, Kourambas J, Munver R, et al. Quantification of the tip movement of lithotripsy flexible pneumatic probes. J Urol. 2000;164:1735-1739.
Zink RA, Frohmueller HG, Eberhardt JE. Urosepsis following ESWL. J Urol. 1988;139(Suppl.):265.