chapter 47 Percutaneous Approaches to the Upper Urinary Tract Collecting System
History and Introduction
Commonly ascribed to Goodwin and colleagues (1955), the first therapeutic percutaneous nephrostomy actually was performed by Thomas Hillier in 1865 (Bloom et al, 1989). Hillier, at the Hospital for Sick Children at Great Ormond Street, repeatedly aspirated the hydronephrotic kidney of a young boy for symptom relief over a 4-year period until his death at 8 years of age. Subsequently there were a few reports of diagnostic percutaneous renal aspirations, but it was not until Goodwin and colleagues published their landmark report in 1955 that therapeutic percutaneous nephrostomy was rediscovered. Even then, the utility of percutaneous access to the upper urinary tract collecting system was limited to drainage of obstructed kidneys until Fernström and Johansson (1976) reported the percutaneous removal of renal calculi, termed “percutaneous pyelolithotomy.”
In the 30-plus years since that report, a number of procedures have been performed using the convenient and safe percutaneous route to accessing the upper urinary tract collecting system including drainage of an obstructed kidney, nephrolithotomy, endopyelotomy, and resection of urothelial tumors. More recently, percutaneous access to portions of the kidney other than the collecting system has expanded the diagnostic and therapeutic choices for patients with renal diseases. Other chapters in this book address these indications, including Chapters 49 and 56. This chapter addresses only percutaneous access to the upper urinary tract collecting system, focusing on the creation, maintenance, and postprocedure management of the percutaneous tract. The final section of the chapter reviews the general complications of percutaneous access to the upper urinary tract collecting system. Specific aspects of the procedures performed through the percutaneous access are covered in Chapters 7, 41, 48, and 53.
Indications for Percutaneous Access
Simple Drainage or Access
Percutaneous drainage of the upper urinary tract collecting system can be for diagnostic or therapeutic indications. The only remaining popular indication for diagnostic percutaneous nephrostomy is to perform a Whitaker test, which requires placement of a small-caliber nephrostomy tube through which contrast material is instilled at specific flow rates while pressures are measured to assess for ureteral obstruction (see more detailed description in Chapter 41). In other cases, diagnostic nephrostography is performed as an adjunct to therapeutic percutaneous nephrostomy.
Therapeutic percutaneous nephrostomy tubes can be placed to drain the kidney (see Chapter 7) and access the upper urinary tract for direct instillation of therapeutic agents (see Chapter 53) or to perform a surgical procedure. Percutaneous nephrostomy is indicated to drain the upper urinary tract collecting system in cases of obstruction at an intrarenal location, at the ureteropelvic junction, or anywhere in the ureter. Obstruction of the lower urinary tract is best treated by drainage of the bladder rather than the kidney, unless secondary obstruction of the upper tract has developed that is refractory to vesical drainage. An alternative to percutaneous drainage is drainage through a ureteral catheter or stent placed in a retrograde fashion (cephalad from the bladder to the kidney, as opposed to antegrade, which is placement from the kidney toward the bladder). The choice of antegrade or retrograde drainage of the upper urinary tract collecting system depends on the indication for the procedure, patient’s medical condition, particular anatomy of the patient, and preferences of both the patient and the physician.
All things being equal, a retrograde route to drainage is preferred over the antegrade route. This includes most cases of acute and chronic ureteral obstruction without infection (Rosevear et al, 2007; Wenzler et al, 2008). In the setting of upper urinary tract collecting system obstruction complicated by infection, however, drainage is an emergency and in many such cases percutaneous rather than retrograde drainage may be best (Ng et al, 2002), unless retrograde drainage can be obtained expeditiously and assuredly. Percutaneous nephrostomy tubes and retrograde ureteral stents are generally equivalent in their capacity to resolve fever in patients with upper urinary tract obstruction and fever (Pearle et al, 1998), but in a given patient circumstances may dictate preference for one access over the other. Retrograde placement of a ureteral stent generally requires regional or general anesthesia, whereas a percutaneous nephrostomy tube can be inserted under local anesthesia; this is an important consideration in an ill patient. Because the percutaneous route has a greater initial success rate than the retrograde one in cases in which the collecting system is dilated, it might be preferred in a patient who needs rapid intervention. This is especially true when the ureteral obstruction is long, severe, or involving the ureteral orifice—all of which can make retrograde stent placement more difficult. Conversely, untreated coagulopathy is a contraindication to percutaneous access, but internal ureteral stents can be placed safely in an anticoagulated patient. A final consideration is patient preference. Although the health-related quality of life for patients with percutaneous nephrostomy tubes and those with internal ureteral stents is similar, a given patient may prefer one route over the other (Joshi et al, 2001). For additional discussion of percutaneous nephrostomy versus internal ureteral stent for drainage of the upper urinary tract, see Chapter 7.
Percutaneous Surgery
Access into the upper urinary tract collecting system may be indicated to directly instill therapeutic agents. This includes chemolysis of urinary calculi and intracavitary topical therapy for urothelial carcinoma. In most cases, the access elected for the instillation will already be present after the preceding surgical procedure.
An important indication for percutaneous access into the upper urinary tract collecting system is for intrarenal or intraureteral surgery. This includes percutaneous endopyelotomy and endoureterotomy (see Chapter 41), nephrolithotomy, treatment of calyceal diverticula and hydrocalyces, and antegrade ureteroscopic treatment of large ureteral stones (see Chapter 48); percutaneous resection of urothelial tumors (see Chapter 53); and less common procedures such as management of fungal bezoars. For all of these procedures skillfully attaining access, properly managing postoperative drainage, and preventing and treating complications related to the percutaneous access are major components of the procedure. The remainder of this chapter centers on these topics.
Anatomic Considerations
Given that the visualization of the kidney and surrounding structures during standard percutaneous entry guided by fluoroscopy or ultrasonography is limited, understanding of the renal and perirenal anatomy is critical for obtaining access that is both effective and safe. Even armed with this knowledge, variations in anatomy can make access challenging for the experienced surgeon and prohibitive for the inexperienced one.
Perirenal Anatomy
The kidneys are well-protected organs, situated retroperitoneally and surrounded by adipose tissue. The mobility of the kidneys is limited by the short renal hilar vessels, although nephroptosis (“falling kidney”) can occur, especially in thin women with a paucity of perirenal fat. In such cases the kidney not only descends but also rotates anteriorly. This can be troublesome during percutaneous punctures with the patient in a prone position.
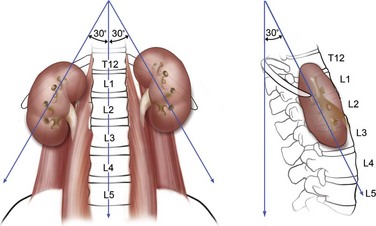
The kidneys lie adjacent to the vertebral bodies, usually extending from the 11th or 12th thoracic to the 2nd or 3rd lumbar vertebrae (Fig. 47–1). The right kidney is displaced a few centimeters inferior to the left kidney. The longitudinal axis of the kidneys parallels the lateral edges of the psoas muscles, about 30 degrees from vertical, with the lower poles lateral to the upper poles. The kidneys are also tilted 30 degrees off the frontal plane, with the lower poles anterior to the upper poles. Finally, the kidneys are rotated out of the frontal plane as well, with the lateral aspect of the kidney posterior to the medial aspect, such that each kidney is rotated 30 degrees posteriorly from the renal hilum.
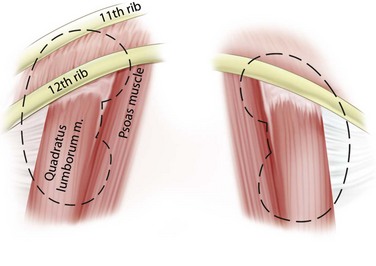
Immediately posterior to the kidneys are the quadratus lumborum and psoas muscles, except at the upper poles where the diaphragm is posterior (Fig. 47–2). The pleura can be violated during percutaneous entry into the upper pole of the kidney. This risk is greater as the access to the kidney is moved cephalad. The lung itself lies above the 11th rib, so direct lung injury is unlikely unless the 10th intercostal space (superior to the 11th rib) is used as the entry site. The ribs curve inferiorly from medial to lateral, such that more portions of the kidney can be approached subcostally with a medial as opposed to lateral access site.
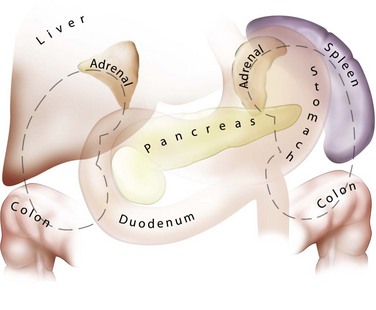
The lateral, anterior, and medial perirenal relationships are more varied than the posterior ones (Fig. 47–3). On the right side, the liver is anterior to the upper pole of the kidney and can extend in some individuals to cover the entire anterior surface. On the left, the spleen covers less of the kidney anteriorly. Both the liver and spleen can extend lateral to the kidneys and are therefore at risk of injury with a lateral puncture into the kidney. The ascending and descending colon can be lateral or even posterior to the right and left kidneys, respectively. The apposition of the colon to the kidney varies with location; it is greatest on the left side and at the lower pole. In one study of computed tomograms the left colon was posterior in 16.1% of cases, and the right colon was posterior in 9% of cases at the level of the lower pole. At the midaspect of the kidney the colon was posterior in 5.2% and 2.8%, respectively, and at the upper pole 1.1% and 0.4%, respectively (Boon et al, 2001). Additional visceral relations to the kidney include the adrenal glands (medial to the upper pole of both kidneys), the duodenum and gallbladder (anterior and medial to the right kidney), and the tail of the pancreas (anterior and medial to the left kidney). These structures can be injured with a misdirected or excessively deep puncture.
Renal Parenchyma and Collecting System
The renal parenchyma is composed of cortical and medullary tissue. The renal cortex is the outermost layer. It contains the glomeruli and proximal and distal convoluted tubules. The more interior medulla contains the renal pyramids. These are inverted cones (the base of which is superficial and the apex is deep) that comprise the loops of Henle and the collecting ducts, which coalesce at the apex of the pyramid into papillary ducts that open on the surface of the renal papillae. There are approximately 20 papillary ducts draining into each papilla. The columns of Bertin are invaginations of cortical tissue that surround the renal pyramids except at their apices.
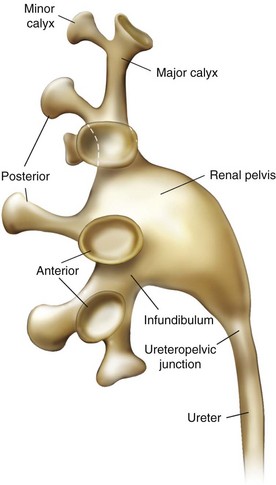
The renal papillae drain into the minor calyces, which are the most peripheral portions of the intrarenal collecting system. If only one papilla drains into a minor calyx, it is described as a simple calyx. When there are two or more papillae entering the calyx, it is termed a compound calyx. The outermost wall of the calyx, into which the papilla is set, is the calyceal fornix. There are 5 to 14 minor calyces in each kidney (mean of 8, with 70% of kidneys having 7 to 9 minor calyces) (Sampaio and Mandarim-de-Lacerda, 1988). There are three drainage zones: the upper pole, the middle region, and the lower pole. Compound calyces are the rule in the upper pole, are common in the lower pole, and are rare among the middle calyces. The minor calyces, either directly or after coalescing into major calyces, drain by infundibula into the renal pelvis (Fig. 47–4). Occasionally a minor calyx will open directly into the renal pelvis without an intervening infundibulum. Some infundibula are unusually narrow, even if they drain adequately, and can present an obstacle to endoscopy, especially with the relatively large rigid nephroscope.
The compound calyces of the poles of the kidney are oriented facing their respective poles. The simple calyces usually come in pairs, one facing anteriorly and one facing posteriorly (Fig. 47–5). The upper pole calyceal system almost always contains at least one compound calyx, and in some cases this is the only calyx in the system. Drainage of the upper pole into the renal pelvis is by a single midline infundibulum in the majority of kidneys. The lower pole system often contains a compound calyx as well. The calyceal drainage from the lower pole is via a single infundibulum in about half of kidneys and through a series of paired anterior and posterior calyces in approximately half of kidneys. With compound calyces rare in the middle calyceal system, the middle calyces are typically arranged in a series of paired anterior and posterior calyces. In about two thirds of kidneys, there are two major calyceal systems—an upper and lower one—and the middle calyces drain into either or both systems. In the other third of kidneys, the middle calyceal system is distinct from the upper and lower systems, either coalescing into a middle major calyx before emptying into the renal pelvis or with drainage of the middle minor calyces directly into the renal pelvis through short infundibula.
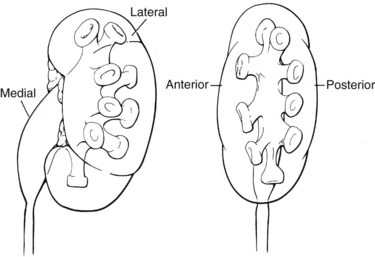
Figure 47–5 Calyceal orientation of polar and middle calyces.
(From Smith AD, editor. Controversies in endourology. Philadelphia: WB Saunders; 1995.)
An important consideration for percutaneous renal surgery is determining the anterior-posterior orientation of the calyces because access (from the typical posterior or posterolateral approach) into a posterior calyx allows relatively straight entry into the rest of the kidney, whereas percutaneous puncture of an anterior calyx requires an acute angulation to enter the renal pelvis, which may not be possible with rigid instrumentation (Fig. 47–6). Efforts have been made to determine which calyces are likely to be anterior and which are likely to be posterior, solely on the basis of their medial-lateral position on anterior-posterior radiography. The distinction pertains to the middle and lower calyceal system, which contains (in almost all middle systems and approximately half of the lower system) paired anterior and posterior minor calyces. The upper pole system, with its almost uniformly compound calyceal system, is less problematic in this regard. Paired anterior and posterior calyces usually enter about 90 degrees from each other. As such, the relative medial-lateral orientation (on anterior-posterior radiography) is determined by the relationship of this 90-degree unit to the frontal plane of the kidney. In a Brödel-type kidney this unit is rotated anteriorly, such that the posterior calyces are about 20 degrees behind the frontal plane and the anterior calyces are 70 degrees in front of the frontal plane. The posterior calyces are lateral, and the anterior calyces are medial in this case. The Hodson-type kidney is the opposite; the calyceal pairs are rotated posteriorly, with the posterior calyces 70 degrees behind the frontal plane and appearing medial and the anterior calyces 20 degrees in front of the frontal plane and appearing lateral (see Fig. 47–6). Most right kidneys have a Brödel-type orientation (posterior calyces are lateral), and most left kidneys have a Hodson-type orientation (posterior calyces are medial). Because variation is considerable, the lateral-medial orientation of the calyces on antero-posterior radiography cannot be used to reliably determine the optimal calyx for entry and additional maneuvers are required to determine the exact calyceal anatomy.
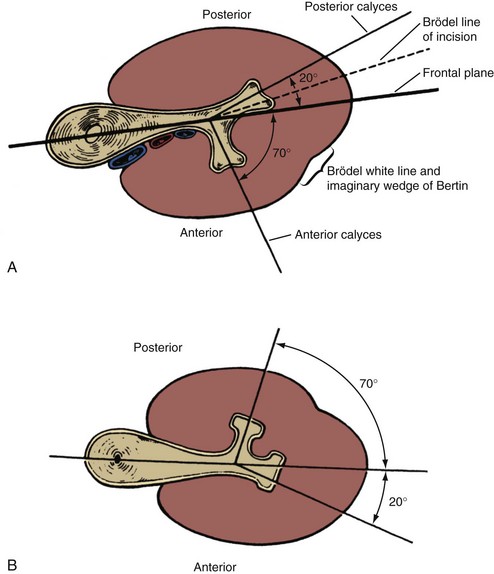
Figure 47–6 Relation of anterior and posterior calyces to renal parenchyma in Brödel-type kidney (A) and Hodson-type kidney (B). The optimal site of percutaneous entry from the posterior aspect of the kidney is into a posterior calyx because the path into the renal pelvis is fairly straight. If entry is into an anterior calyx (from the posterior aspect of the kidney) then an acute angulation must be made to enter the renal pelvis, which may not be possible with rigid instrumentation.
(From Smith AD, editor. Controversies in endourology. Philadelphia: WB Saunders; 1995.)
Intrarenal Vasculature
Although the renal arterial anatomy is variable, in general the main renal artery divides into an anterior and a posterior branch. The former then divides within or before the renal sinus into four anterior segmental arteries: the apical and lower segmental arteries (which supply the tip of the upper pole and the entire lower pole, respectively), and the upper and middle segmental arteries (which supply the remainder of the anterior half of the kidney). The posterior branch of the renal artery supplies the remainder of the posterior half of the kidney (Fig. 47–7). Once the anterior segmental arteries and the posterior branch of the renal artery enter the renal parenchyma, they divide into interlobar arteries, which are also called infundibular arteries owing to their course adjacent to the calyceal infundibula of the renal collecting system. At the cortico-medullary junction, near the base of the renal pyramids, each interlobar artery usually divides into two arcuate arteries that run along the renal pyramid. The next division is into the interlobular arteries, which run along the outer surface of the renal pyramids and are derived at right angles from the arcuate arteries. The final divisions, the afferent arterioles of the glomeruli, come off the interlobular arteries in the peripheral renal cortex. Each renal arteriole is an “end-artery,” meaning that each cell in the kidney derives blood supply from one arteriole. For this reason renal arterial vascular injury must be avoided in order to prevent loss of renal function. The potential for arterial injury is least in Brödel’s line, an avascular plane approximately at the lateral margin of the kidney, extending from the superior apex of the kidney (limited by the circulation of the apical anterior segmental artery) to the lower pole of the kidney (limited by the circulation of the lower anterior segmental artery). Additionally, the safest place to percutaneously access the collecting system is directly into the calyceal fornix because this will avoid the interlobar (infundibular) arteries adjacent to the calyceal infundibula and the arcuate arteries that skirt the renal pyramid (Sampaio et al, 1992).
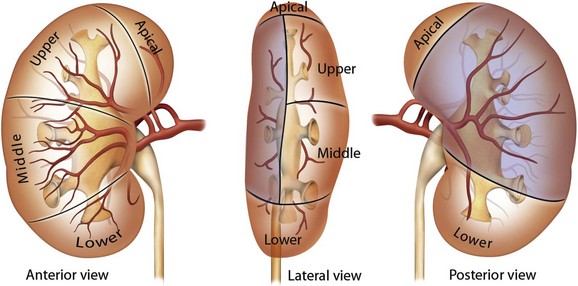
Figure 47–7 Arterial supply to the kidney. The kidney is supplied by the anterior and posterior branches of the main renal artery. The anterior branch supplies both the anterior half of the kidney and the polar regions via four segmental branches. The posterior branch of the renal artery supplies the posterior aspect of the kidney (represented by the shaded region). An avascular plane, known as Brödel line, separates the anterior and posterior circulations.
The venous anatomy of the kidney does not have the same defined structure as the arteries. Moreover, there is free cross-circulation among the intrarenal veins via arcades. This enhances vascular outflow from the kidneys, reduces the risk of venous congestion, and makes renal venous injury less damaging to renal function than arterial injury.
Key Points: Anatomic Considerations
Obtaining Percutaneous Access
Adequate percutaneous access to the upper urinary tract collecting system is a critical part of the percutaneous procedure. Over time more urologists are being trained to attain percutaneous access, but only a minority of urologists maintains this skill in practice (Bird et al, 2003; Lee et al, 2004a). Although there is some controversy regarding the success and complications associated with percutaneous access attained by urologists versus radiologists (Watterson et al, 2006; El-Assmy et al, 2007), the critical distinction in ensuring effective access for a subsequent percutaneous procedure is not who obtains access but rather that the access for urologic surgery is directed by a urologist. The optimal situation is for the urologist to be present at the time access is attained, either performing the access procedure or actively directing the radiologist as to which calyx to enter and at what angle. If that is not possible, then the urologist should discuss beforehand with the radiologist attaining access the pertinent considerations in that particular patient.
Periprocedural Antimicrobials
Prevention of infectious complications is a major consideration when considering percutaneous access into the upper urinary tract collecting system. In an elective setting, confirmation that urine is sterile is optimal. In cases of anatomic abnormality, recent hospitalization, distant coexistent infection, recent catheterization, or other situations that suggest increased likelihood of bacteriuria, a urine culture is recommended. In the setting of an externalized catheter or staghorn renal calculus, bacteriuria is likely and a preoperative urine culture should be considered standard practice. In other cases a screening urinalysis may be adequate, with subsequent culture if the urinalysis is suspicious for infection. When there is bacteriuria, a therapeutic course of culture-directed antimicrobials should be administered to sterilize the urine. Optimally, a repeat urine culture should be obtained to confirm urine sterility, although this might not be practical. In cases with colonized exogenous material such as an externalized urinary catheter or an infected calculus, it may not be possible to eradicate the infection before the percutaneous procedure. The goal then is to suppress the bacterial count before intervention.
The American Urological Association recommends periprocedural antimicrobial prophylaxis for all cases of percutaneous renal surgery (Wolf et al, 2008). Although there are no randomized, controlled trials to support this recommendation, nonrandomized data suggest a postprocedure urinary infection rate of 35% to 40% if antimicrobial prophylaxis is not used compared with 0% to 17% if prophylaxis is used (Charton et al, 1986; Darenkov et al, 1994). The need for antimicrobial coverage for simple percutaneous drainage of the upper urinary tract collecting system is not certain.
Antimicrobial coverage should include organisms common to the urinary tract (Escherichia coli, Proteus sp., Klebsiella sp., Enterococcus sp.) and the skin (Staphylococcus aureus, coagulase-negative Staphylococcus sp., group A Streptococcus sp.). Recommended agents include first- and second-generation cephalosporins; aminoglycosides (or aztreonam in patients with renal insufficiency) plus either metronidazole or clindamycin; ampicillin/sulbactam; or a fluoroquinolone. When the antimicrobial is being administered only for prophylaxis (i.e., not treatment of known or presumed infection), the duration of treatment should be for 24 hours or less. A short (≤24 hours) course of antimicrobials at the time of nephrostomy tube removal also can be considered (Wolf et al, 2008).
Patient Positioning
The prone position was described by Goodwin and colleagues (1955) for percutaneous access to the upper urinary tract collecting system, and over time this position became standard. It has the advantage of presenting a large surface area (the patient’s back) that provides many choices of access sites and a stable horizontal working surface. The posterior or posterolateral approach is the most direct one to the desirable posterior calyces and comes closest to approaching the kidney through Brödel’s avascular line. Prone positioning does have some disadvantages, however. It is associated with a decrease in cardiac index (Hatada et al, 1991), and in cases where inadequate padding is provided it is associated with decreased pulmonary capacity, although if enough padded support ensures free abdominal and chest wall movement, then pulmonary capacity is greater in the prone compared with the supine position (Edgcombe et al, 2008). The anesthesiologist has poor access to the airway with the patient in the prone position. Prone positioning might not be possible in patients with morbid obesity and/or spinal concavity, and the prone position can be associated with neuromusculoskeletal complications such as nerve compression or stretch injury, ocular or facial injury, and rhabdomyolysis. Finally, the prone position requires that the surgeon stand, often holding instruments at a distance using outstretched arms, which leads to surgeon fatigue. To address these deficiencies, urologists have introduced supine and lateral decubitus positioning for percutaneous renal surgery.
Valdivia Uria and colleagues (1987) first reported the supine approach to percutaneous nephrostolithotomy, culminating in their 1998 review of 557 patients with percutaneous nephrostolithotomy performed in this position (Valdivia Uria et al, 1998). They reported few complications including no hydrothoraces or pneumothoraces, no colon injuries, and only a 0.5% rate of major hemorrhage (see later for further discussion of complications). Variations of this position include completely supine, supine with the ipsilateral side elevated, and supine combined with ipsilateral flank elevation and asymmetric lithotomy position (Falahatkar et al, 2008; Papatsoris et al, 2008; Scoffone et al, 2008; Zhou et al, 2008). In the prone position, with a posterior or posterolateral skin entry, the posterior calyces are the best to enter because they allow access to the renal pelvis and the rest of the kidney. In the supine position, the skin entry site is lateral or anterolateral so the best calyces to enter are often the anterior ones. The angle of the access sheath when doing a procedure such as percutaneous nephrostolithotomy in the supine position is angled toward horizontal (compared with vertical during percutaneous nephrostolithotomy in the prone position), which reduces the pressure in the collecting system and facilitates stone fragments washing out through the sheath. Supine positioning does not require repositioning after induction of anesthesia, and the urethra is more easily accessed than in the prone position. The supine position is a safer position with regard to neuromusculoskeletal complications. Finally, because the percutaneous entry is more lateral than during a prone procedure, the instruments are closer to the surgeon, resulting in less physical strain on the surgeon and the opportunity to sit during the procedure.
There are some disadvantages to the supine position for percutaneous renal surgery, however. First, it is not familiar to most urologists because the prone position is used in most training programs. Second, the reduced pressure in the collecting system results in a lower volume and thus less room for visualization and manipulation. Third, upper pole calyceal access is more difficult in the supine compared with prone position. Finally, with optimal placement of pads and bolsters, the prone position may provide better ventilation than the supine position (Edgcombe et al, 2008).
A systematic review including literature through 2008, which included 9 studies of supine and 25 for prone percutaneous nephrolithotomy, found that for cases involving obese patients or staghorn calculi, prone percutaneous nephrolithotomy was associated with decreased operative time, similar rate of hemorrhage, and improved stone-free rate (de la Rosette et al, 2008b). In the only randomized study, however, operative time favored the supine position without differences in other parameters (Falahatkar et al, 2008).
Less commonly used for percutaneous renal surgery is the flank (lateral decubitus) position, which was first described by Kerbl and colleagues (1994). This position allows simultaneous access to the anterior and posterior aspects of the kidney and appears to be particularly useful for morbidly obese patients or those with spinal deformities in whom both supine and prone positioning are difficult (Gofrit et al, 2002; Basiri et al, 2008b; Karami et al, 2009).
Although the supine and flank positions offer some potential benefits, overall experience is limited and the only North American report is a single case (Kerbl et al, 1994). As such, the remainder of this chapter concerns access into a posterior calyx from a posterior or posterolateral direction with the patient in the prone position, which remains the standard.
The initial step in a percutaneous procedure is often cystoscopic retrograde placement of a ureteral catheter. This can be performed with the patient in prone position (using a flexible cystoscope) or in lithotomy with subsequent prone repositioning. In the “upside-down” bladder the bubble of air from initial introduction of the cystoscope often approximates the location of the ureteral orifices. Prone cystoscopy can be performed on a standard operating or fluoroscopy table but is simplified by the use of “spreader bars” on the foot of the table (also called a “split-leg table”). The legs are spread by abducting the legs with the knees straight. This provides better access to the external urethral meatus.
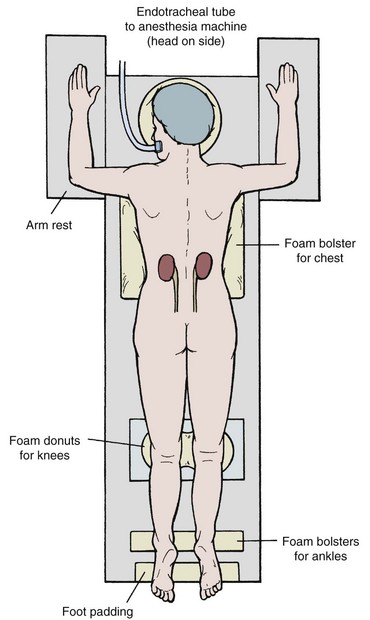
Careful placement of padding is important in the prone position (Fig. 47–8). Support the head with padding in a neutral position that allows access to the mouth. Make sure there is not undue pressure on the facial bones, nose, and ears. Place the ipsilateral arm above the head to get it out of the operative field, with the shoulder and elbow at right angles with the use of generous padding. Position the contralateral arm the same way, or leave it straight and tucked at the side. Rest the lateral aspects of the chest on rolled blankets or other bulky foam or gel bolsters to allow for chest and abdominal wall expansion. Alternatively, purpose-made pads and supports provide more assured patient positioning (Papatsoris et al, 2009). Provide support under the ankles to take pressure off the feet, and pad the knees and feet. Prepare the perineum and ipsilateral flank sterilely, and cover unsterile areas with drapes. Cover the flank with an adherent drape that incorporates a fluid collection pouch.
Choice of Access Site to the Collecting System
The access site into the upper urinary tract collecting system is a critical determinant of the success of the subsequent procedure. In the prone position, the preferred calyces are the posterior ones (or the posterior aspect of compound calyces), which allow better access to the remainder of the collecting system. Usually, the anterior calyces can be approached through a posterior calyx. In cases that involve a calyceal diverticulum, narrow infundibulum, or pathology in an eccentric anterior calyx, direct puncture into an anterior location might be required. The ability to access the rest of the collecting system is limited with such access. Percutaneous access should never be directly into an infundibulum or the renal pelvis, which greatly increases the risk of vascular injury (Sampaio et al, 1992). The state of the renal parenchyma overlying the intended calyx of entry also must be considered because if it is thin, the tract into the collecting system may not close well after nephrostomy tube removal.
An upper pole calyx is generally the most versatile site through which to enter the upper urinary tract collecting system. The renal pelvis, lower pole calyces, and ureter usually can be entered with a rigid nephroscope from a well-placed upper pole access. Because of the posterior tilt of the upper pole of the kidney, the lower pole does not offer assured access to the upper pole. Access to the middle calyces will usually require a separate access or use of flexible instrumentation. Often, a middle calyx will offer adequate access to the ureteropelvic junction, as needed in cases such as endopyelotomy. In other cases, the calyx of entry should be selected on the basis of the distribution of the pathology to be treated. Efforts should be made to select a calyx that will allow treatment through a single site with rigid instrumentation. If this is impossible, then the site that will allow the largest portion of the pathology to be treated should be selected. The remaining pathology can be addressed with a second (and rarely a third or more) access or with flexible instrumentation through the initial access site.
Subcostal access is the safest route to the kidney because pleural injuries are rare with entry below the 12th rib. Nonetheless, if entry directly above the 12th rib (11th intercostal space) provides the best access to the optimal calyx, then the benefit generally exceeds the risk (Fig. 47–9). Entry above the 11th rib, however, has a greater potential for pleural and even lung injury, so when the best access calls for a direct puncture above the 11th rib, additional maneuvers should be considered to make access safer. A subcostal access sheath placed into a lower calyx can be tilted cephalad, displacing the kidney caudally to allow access to an upper pole calyx at a lower intercostal level (Karlin and Smith, 1989). Another alternative is to angle the access tract cephalad from a subcostal entry site (Liatsikos et al, 2005; Rehman et al, 2008). This approach provides limited access to the rest of the kidney. These alternatives can result in damage if the kidney is moved caudally with excessive force. Access above the 10th rib is associated with a high incidence of pleural violation and lung injury and should be avoided unless absolutely necessary. Thorascopically guided access superior to the 10th rib can be performed to reduce the risk of lung injury (Finelli and Honey, 2001).
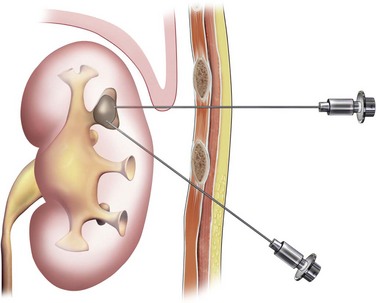
Figure 47–9 Subcostal and supracostal percutaneous access to an upper pole calyx. The supracostal approach provides more direct access and provides a better angle for endoscopy of the rest of the kidney.
Although standard preoperative ultrasonography or intravenous urography is sufficient in most cases for treatment planning, in complex cases cross-sectional imaging with computed tomography (CT) or magnetic resonance imaging (MRI) might be helpful. CT is a standard at many centers. For more accurate representation of the renal collecting system, three-dimensional imaging reconstructions are useful (Hubert et al, 1997; Buchholz, 2000; Ng et al, 2005; Thiruchelvam et al, 2005; Patel et al, 2009).
Retrograde Assistance for Access into the Collecting System
One of the advantages of urologist participation in obtaining the initial percutaneous access is the opportunity to provide retrograde transurethral assistance. This assistance can take many forms, from placing a ureteral catheter, to inserting a flexible ureteroscope, to obtaining the percutaneous access using a retrograde inserted device.
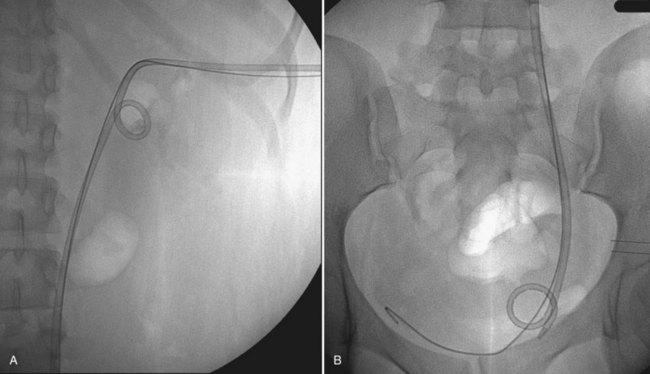
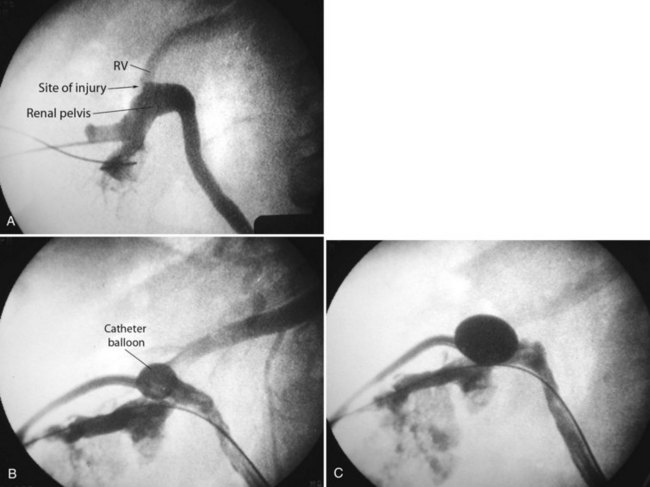
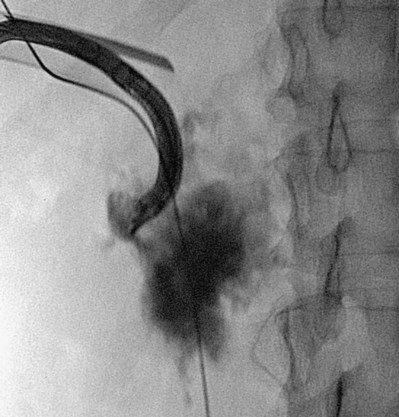
The simplest form of retrograde transurethral assistance is to place a 5- or 6-Fr straight ureteral catheter up into the renal pelvis (Fig. 47–10 on the Expert Consult website![]() ). Air and/or contrast material can be injected to delineate and dilate the intrarenal collecting system anatomy (Fig. 47–11 on the Expert Consult website
). Air and/or contrast material can be injected to delineate and dilate the intrarenal collecting system anatomy (Fig. 47–11 on the Expert Consult website![]() ), and a guidewire can be passed from below and grasped by the nephroscope to establish through-and-through access from the external urethral meatus to the percutaneous entry site. A dual-lumen catheter can be placed as well. The small caliber of either catheter, however, does not provide much outflow from the kidney and may not prevent stone or tumor fragments from passing into the ureter along the catheter. A ureteral occlusion balloon catheter, which incorporates an approximately 15-Fr spherical balloon on the distal tip, more consistently prevents material from migrating down the ureter. The balloon should be carefully inflated in the renal pelvis, making sure the balloon is not in the ureter—which could lead to ureteral rupture—and then gently pull down to occlude the ureteropelvic junction (Fig. 47–12). Another alternative is to place a ureteral access sheath (usually 11 to 15 Fr) over a retrograde-inserted guidewire (Landman et al, 2003). The large outer diameter of the sheath effectively prevents particles from passing around the sheath into the ureter, and the large inner diameter affords excellent outflow of small stone particles. The disadvantages of employing a ureteral access sheath include the potential ureteral trauma from passing such a large device into the ureter and clogging of the catheter lumen by oversized stone fragments.
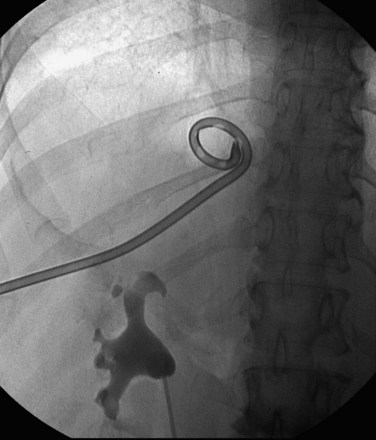
), and a guidewire can be passed from below and grasped by the nephroscope to establish through-and-through access from the external urethral meatus to the percutaneous entry site. A dual-lumen catheter can be placed as well. The small caliber of either catheter, however, does not provide much outflow from the kidney and may not prevent stone or tumor fragments from passing into the ureter along the catheter. A ureteral occlusion balloon catheter, which incorporates an approximately 15-Fr spherical balloon on the distal tip, more consistently prevents material from migrating down the ureter. The balloon should be carefully inflated in the renal pelvis, making sure the balloon is not in the ureter—which could lead to ureteral rupture—and then gently pull down to occlude the ureteropelvic junction (Fig. 47–12). Another alternative is to place a ureteral access sheath (usually 11 to 15 Fr) over a retrograde-inserted guidewire (Landman et al, 2003). The large outer diameter of the sheath effectively prevents particles from passing around the sheath into the ureter, and the large inner diameter affords excellent outflow of small stone particles. The disadvantages of employing a ureteral access sheath include the potential ureteral trauma from passing such a large device into the ureter and clogging of the catheter lumen by oversized stone fragments.
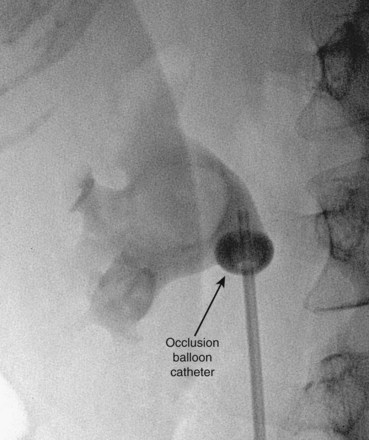
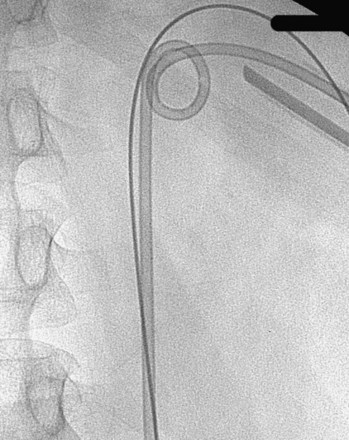
Figure 47–12 Occlusion balloon inflated and snugged down at ureteropelvic junction of contrast-filled upper tract collecting system.
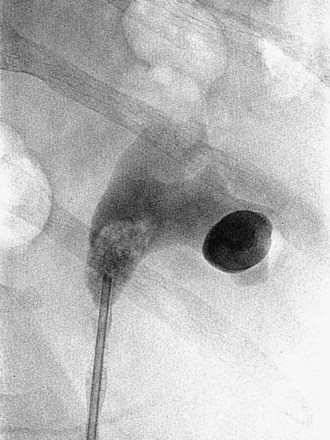
Figure 47–11 Retrogram pyelogram with air and contract reveals air in upper pole compound calyx and posterior lower pole calyx.
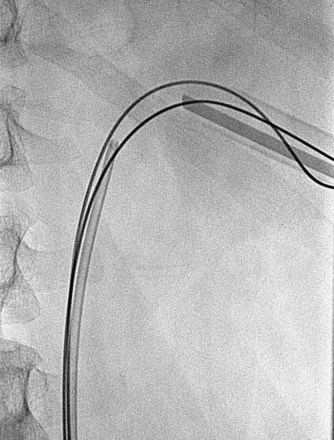
A ureteroscope passed retrograde can greatly facilitate percutaneous entry into the intrarenal collecting system (Grasso et al, 1995; Kidd and Conlin, 2003; Patel et al, 2008) by allowing the surgeon to observe and correct the percutaneous placement of a needle. A basket can then be passed through the ureteroscope to grasp the end of the percutaneous guidewire; pulling this out the urethra provides through-and-through access (Fig. 47–13). Even if the pathology being addressed is so large that direct visualization of the percutaneous needle is obscured (e.g., complete staghorn calculus), the ureteroscope can still be used to rapidly attain through-and-through access. Moreover, the ureteroscope may have better access to some sites in the kidney than the nephroscope and can be used to assist in the procedure (e.g., fragment or relocate stones, fulgurate small tumors).
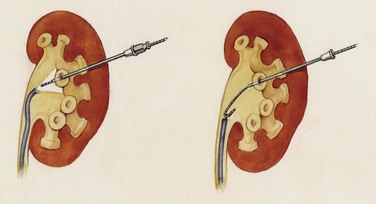
Figure 47–13 Ureteroscope retrograde assistance. The needle can be directly visualized entering the calyx, and a wire can be grasped and pulled down the ureter and out the urethra.
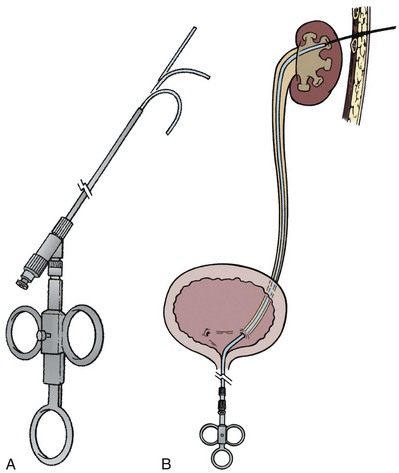
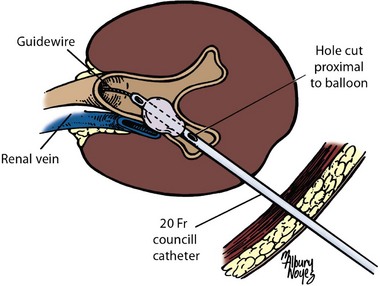
The “ultimate” retrograde assistance to percutaneous access into the upper urinary tract collecting system is the retrograde approach to percutaneous access. Although the antegrade approach is much more commonly performed, a retrograde approach may be selected when the surgeon has limited experience with antegrade percutaneous renal puncture or in situations in which there might be a technical advantage to the retrograde approach such as morbid obesity or a hypermobile or abnormally situated kidney (Mokulis and Peretsman, 1997). The Lawson Retrograde Nephrostomy Wire Puncture Set (Cook Urological; Spencer, IN) is the device commercially available for this use. After retrograde placement of a guidewire into the renal pelvis, pass the 7-Fr Torcon catheter—which is actively deflectable from 0 to 140 degrees (Fig. 47–14A)—over the guidewire and into the desired calyx. Insert the 3-Fr polytetrafluoroethylene (PTFE) sheath containing the 0.017-inch stainless steel puncture wire through the Torcon catheter. Advance the puncture wire through the kidney and body wall under fluoroscopic control, withdrawing and repositioning it if any obstacles such as a rib are encountered (Fig. 47–14B). Make a small skin incision and grasp the wire externally. Use the fascial dilators in an antegrade fashion until the Torcon catheter can be advanced through the tract. Once the end of the catheter exits the skin, exchange the puncture wire for a standard 0.035 inch guidewire, thus attaining through-and-through access.
Antegrade Approach to Access into the Collecting System: Needles and Guidewires
The antegrade approach to percutaneous access into the upper urinary tract collecting system is the standard. It affords the most control of the skin entry site and can be guided by ureteroscopy or a variety of imaging modalities.
The general scheme of antegrade access is to place a needle through the skin into the upper urinary tract collecting system. Through the needle a guidewire is placed and then over the guidewire are placed catheters and other devices, eventually enlarging the tract until the desired lumen is reached for the purpose of the procedure. This is the Seldinger technique, described (for vascular access) by Sven-Ivar Seldinger (1953). The standard choices for the needle are a 21-gauge needle through which is passed a 0.018-inch guidewire or an 18-gauge needle through which is passed a standard 0.035-inch guidewire. Both needles have a blunt sheath and a sharp obturator (Fig. 47–15). The 21-gauge needle has the advantage of causing relatively minor injury as it is passed through tissue. Multiple passes can generally be made with little risk of hemorrhage from the needle itself; the option to place and replace the needle multiple times is advantageous because getting the tip of the needle into the right spot in the kidney is the most difficult aspect of percutaneous access into the upper urinary tract collecting system. The 18-gauge needle is more traumatic, and multiple passes should be avoided. The advantage of the 18-gauge needle is that it is stiffer. In a number of circumstances the 21-gauge needle does not maintain trajectory adequately (e.g., scarred kidney, obese patient) and the 18-gauge needle is more effective. In addition, the 0.018-inch guidewire that passes through the 21-gauge needle (Fig. 47–16) must be exchanged for a standard 0.035-inch guidewire for subsequent tract dilation or catheter placement. This requires an extra step, which adds to the complexity of the procedure and increases the risk of loss of access. Balancing the reduced efficacy of the 21-gauge needle and its increased potential for loss of access, versus the increased risk of trauma with the 18-gauge needle, it is recommend that the 21-gauge needle be used when the operator is less experienced or if minimizing trauma is paramount. The 18-gauge needle should be used when an experienced operator is confident that the tip of the needle can be placed within the desired calyx with just a few attempts.
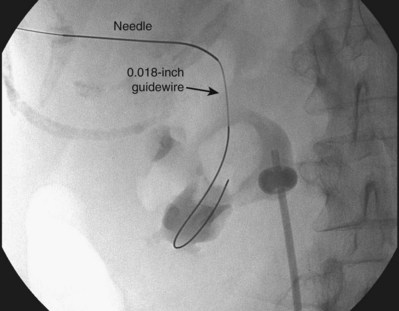
Figure 47–16 0.018-inch guidewire passed through percutaneous needle. This wire is exchanged for a 0.035-inch guidewire for subsequent manipulation.
Two methods can be used to exchange the 0.018-inch for a 0.035-inch guidewire. A coaxial introducer incorporates a small catheter within a slightly larger catheter. After inserting the 0.018-inch guidewire and removing the needle, the coaxial introducer is advanced over the guidewire until the end is within the renal collecting system. The surgeon should remove the inner catheter and 0.018-inch guidewire and then advance a 0.035-inch guidewire through the outer catheter into the collecting system. The second method is the use of a graduated introducer, which fits over the 0.018-inch guidewire at its tip but then enlarges to a lumen that will accept the 0.035-inch guidewire. A few centimeters back from the tip there is a hole through which the 0.035-inch guidewire can pass. After inserting the 0.018-inch guidewire and removing the needle, pass the graduated introducer over the guidewire until it is a few centimeters inside the kidney, such that the side hole is within the renal collecting system. Remove the 0.018-inch guidewire and then insert an angled or J-tipped 0.035-inch guidewire through the introducer until the end of the wire exits the catheter within the collecting system. With either the coaxial or graduated introducer, a stiffener can be used to assist in passing the device over the relatively insecure 0.018-inch guidewire.
The safest initial 0.035-inch guidewire to use for upper urinary tract percutaneous access is a PTFE-coated J-wire. The “J” tip makes the guidewire unlikely to perforate out of the collecting system. This guidewire will not easily pass down the ureter, however, which is more easily done with a floppy tip PTFE-coated guidewire or a hydrophilic guidewire with a straight or angled tip.
Antegrade Approach to Access into the Collecting System: Technique of Initial Access
The initial percutaneous access to the upper urinary tract collecting system described by Goodwin and colleagues (1955) was “blind.” In 1974 Pedersen first reported ultrasonographic guidance (Pedersen, 1974). Although “blind” access is still occasionally reported and in expert hands can be successful, in most cases the initial antegrade percutaneous access into the upper urinary tract collecting system is obtained with real-time imaging guidance. Ultrasonography and fluoroscopy are most commonly employed, with the choice based on patient characteristics and physician preference (Basiri et al, 2008a).
Ultrasonographic Guidance
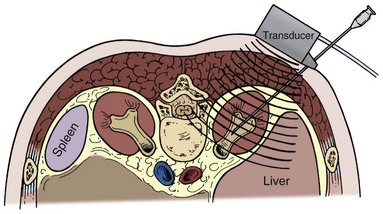
Ultrasonography has the advantages of portability (the mobile ultrasound machine is easier to maneuver than a C-arm fluoroscopy unit and does not require a radiolucent operating table), the ability to rapidly evaluate different aspects of the kidney, and no delivery of ionizing radiation. In addition, retrograde injection of contrast material or air is not necessary because the collecting system can readily be distinguished within the kidney on the basis of ultrasonographic appearance alone. The disadvantages of ultrasonography include less clear visualization of the percutaneous needle (although etched needles made for enhanced sonographic appearance are available), a limited field of view compared with fluoroscopy, and difficulty monitoring the subsequent steps of the procedure. Ultrasonography is the first choice in imaging when retrograde access cannot be attained or is difficult to attain, such as in kidneys above urinary diversions, transplanted kidneys, kidneys above a completely obstructed ureter, or when radiation exposure is a concern. Additionally, when percutaneous access is necessary for simple drainage of the kidney, such that the exact site of access is not critical as long as puncture into the renal pelvis or an infundibulum is avoided, ultrasonography is more expedient and convenient than fluoroscopy.
Using a hand-held 3.5- or 5-MHz ultrasound transducer, inspect the kidney and select a calyx for percutaneous entry. Needle guides can be placed on the transducer to direct the needle in the plane of visualization of the probe. Some prefer to place the needle freehand instead, moving the transducer around to gain different views of the kidney and needle. Observe the needle as it is advanced until it appears that the tip is within the collecting system (Fig. 47–17). Removing the obturator and aspirating urine confirms entry.
Fluoroscopic Guidance
Fluoroscopic guidance is more commonly employed for gaining antegrade access to the upper urinary tract collecting system for percutaneous renal surgery. Although retrograde instillation of air and/or contrast material is not absolutely essential (Tabibi et al, 2007), most urologists find that it enhances fluoroscopically guided percutaneous access (Fig. 47–18). Fluoroscopy provides excellent delineation of the intrarenal collecting system anatomy and pathology, a wide field of view (that can be collimated down to reduce radiation exposure), and the ability to monitor all steps of the procedure. In some cases combining the techniques is an excellent approach, using ultrasonography to guide the initial needle placement and then using fluoroscopy (after injection of air and contrast through the sonographically guided needle) to confirm that the desired calyx has been accessed and to monitor the subsequent steps of the procedure (Osman et al, 2005). If the entry site is incorrect, then fluoroscopy of the air-and-contrast filled collecting system can be used to guide another needle into the desired calyx. With this combination, retrograde assistance is not necessary. This technique is especially useful in accessing nondilated systems without retrograde assistance (Patel and Hussain, 2004).
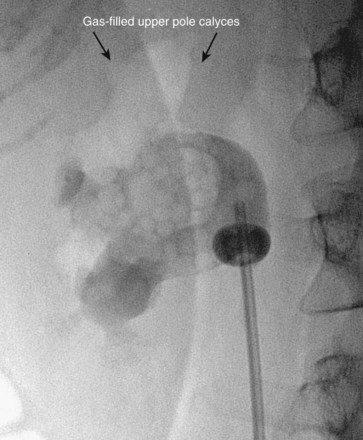
Figure 47–18 Injection of air into contrast-filled upper tract collecting system reveals posterior calyces, in this case most clearly the upper pole calyces. Compare with Figure 47–12, before injection of air.
There are two well-described methods of fluoroscopic guidance for antegrade percutaneous access into the upper urinary tract collecting system: the “eye-of-the-needle” technique and the “triangulation” technique (Miller et al, 2007). Both have their proponents, and there is no clear advantage of one over the other. Through the retrograde device, inject contrast material to delineate the collecting system after first taking note of any radiopaque pathology for later reference. Comparing a spot film of the unopacified collecting system with the opacified view is useful in this regard. Once the options for the calyces of entry are identified, inject air to define which calyces are posterior. In the prone position, air rises up the posterior calyces. The “double-contrast” pyelogram (both contrast material and air) provides the best determination of the pertinent intrarenal anatomy.
To perform the “eye-of-the-needle” technique, first inspect the kidney with the fluoroscopy unit directly above the patient (directed vertically) and select the desired calyx. Next, rotate the top of the fluoroscopic unit 30 degrees toward the operator, which brings the fluoroscopic view more or less end-on with the posterior calyces. The unit can be additionally rotated slightly cephalad or caudad to line it up more exactly with the axis of the calyx. Place the tip of a hemostat on the skin and move it until it is directly over the desired calyx. Mark this site and make an incision large enough to accept the needle and initial dilators. Place the tip of the access needle into this incision, and then move the shaft of the needle while keeping the tip in place until the needle is directly in line with the axis of the fluoroscopy unit; doing so gives the appearance of a “bull’s eye” with the hub of the needle (appearing as a circle) around the shaft (which appears as a dot). The fluoroscopic view is as if the operator is looking down the shaft of the needle at the targeted calyx—thus the term “eye of the needle” (Fig. 47–19A). Advance the needle straight in, while checking with fluoroscopy and adjusting the angle of the needle as needed to maintain the “bull’s-eye” appearance. If the needle is more than a few centimeters deep and readjustment is necessary, the needle may have to be withdrawn before a new trajectory can be followed. The 21-gauge needle can be difficult to control during this step. If this occurs, substitute the more easily passed 18-gauge needle. Once the needle’s axis is fixed and it is thought that the needle is approaching or is in the kidney (typically a “pop” can be felt when the renal capsule is punctured), then rotate the fluoroscopy unit away from the operator, back to vertical or even 10 to 15 degrees beyond vertical. Now the needle appears “in profile” as a straight line. With the cephalo-caudad and medial-lateral axes of the needle now fixed, advance or withdraw the needle to change its anterior-posterior position (depth) to get the tip of the needle into the desired calyx (Fig. 47–19B). Aspiration of urine or air after removing the obturator confirms entry. Instillation of contrast material can be used to confirm entry as well, but if the needle is misplaced, the extravasated contrast material can obscure subsequent fluoroscopic visualization. If a gently passed guidewire stays within the contours of the collecting system, then this confirms proper entry with risking troublesome extravasation of contrast material.
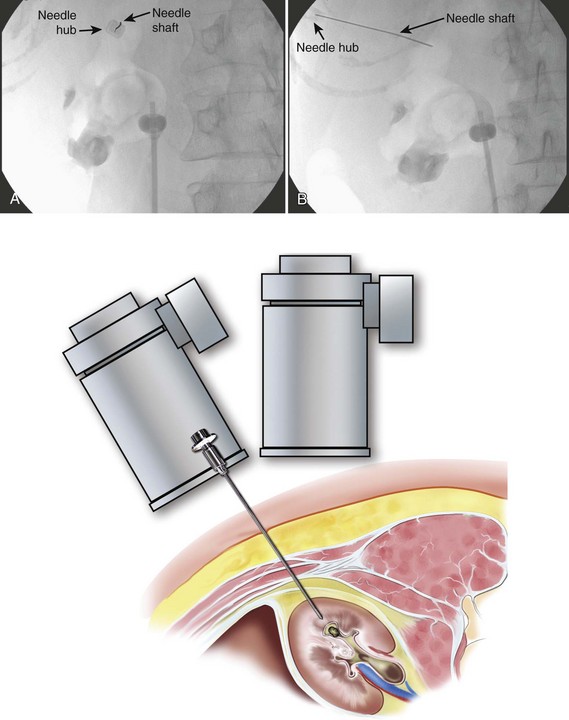
Figure 47–19 “Eye-of-the-needle” fluoroscopic guidance. A, With the top of the fluoroscopic unit rotated 30 degrees toward the operator, the needle is lined up directly over the intended calyx of entry. B, With the fluoroscopy unit rotated away from the operator, the needle is now seen in profile, and in the figure has already been advanced under fluoroscopic control to achieve collecting system entry.
It should be remembered that the ionizing radiation presents a small but real risk. When the operator must grasp the access needle within the fluoroscopy field, it is best to hold the needle with a hemostat, sponge forceps, or purpose-built needle holder to reduce radiation exposure to the operator’s hand. Collimating the field down as much as possible while still maintaining an adequate field of view reduces radiation exposure to the patient and all personnel in the room. Moving a collimated field around to maintain the object of interest in the field is preferable to maintaining a wide field that includes the operator’s hands and unnecessary body parts of the patient. For additional information about radiation safety, see Chapter 4.
To use the “triangulation” technique, inspect the kidney with the fluoroscopy unit directly above the patient to select the desired calyx and hold the needle in the approximate position of the desired angle of entry. Rotate the top of the fluoroscopy unit cephalad and lateral, and widen the field of view with the collimator such that medial-lateral (left-right) movements of the needle are apparent. Move the shaft of the needle while keeping its tip in place until the needle is aimed toward the desired calyx (Fig. 47–20A). Then rotate the top of the fluoroscopy unit medially 45 degrees. While keeping the medial-lateral orientation of the needle constant, move the needle in the cephalo-caudad (up-down) plane until the needle is again aimed toward the desired calyx (Fig. 47–20B). Resting the forearm on the patient’s back will help stabilize the needle in one plane while moving in the other. Move the fluoroscopy unit back and forth between these two positions until the needle remains aimed at the desired calyx on both views. Advance the needle under fluoroscopic guidance while monitoring the anterior-posterior direction (depth) of the needle tip. If the needle position in the medial-lateral and cephalo-caudad planes is maintained, the needle should enter the targeted calyx.
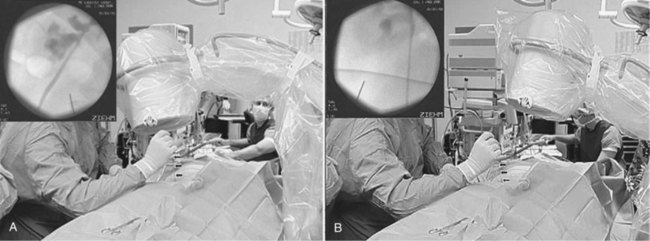
Figure 47–20 Triangulation fluoroscopic guidance. A, With the top of the fluoroscopy unit rotated laterally and cephalad, adjust the access needle (arrows) medial-lateral orientation of the needle. B, After rotating top of the fluoroscopy unit rotated medially, and while keeping medial-lateral orientation of the needle constant, move the needle in the cephalo-caudad plane until the needle is again aimed toward the desired calyx.
(From Miller NL, Matlaga BR, Lingeman JE. Techniques for fluoroscopic percutaneous renal access. J Urol 2007;178:15–23.)
With the “eye-of-the-needle” technique, the proper cephalo-caudad and medial-lateral axes of the needle are verified and maintained on a single fluoroscopic view and the confirmatory view is necessary only to confirm the depth of the needle tip. For the “triangulation” technique, one fluoroscopic view is used to assess the medial-lateral axis and another is used to assess the cephalo-caudad axis, with the depth of the needle tip being assessed on both views (Miller et al, 2007). The advantage of the triangulation technique over the “eye-of-the-needle” technique is that the needle cannot be passed too deeply because the depth of advancement is monitored continuously. The disadvantage of the “triangulation” technique is that maintaining both the medial-lateral and cephalo-caudad planes is difficult because both are not being monitored at the same time as in the “eye-of-the-needle” technique. Use of the 18-gauge rather than a 21-gauge needle is recommended with the “triangulation” technique to help maintain angle of entry.
Advanced Guidance
In some complex cases, percutaneous access into the kidney guided by CT or MRI might be considered. The initial access is obtained into the desired calyx with the patient on the CT or MRI table, similar to the techniques used for needle biopsy (Barbaric et al, 1997; Hagspiel et al, 1998; Thanos et al, 2006). Bowel and other viscera can be imaged to protect against their injury, and this approach is especially useful in cases of anatomic abnormalities (LeMaitre et al, 2000; Matlaga et al, 2003) and for nondilated collecting systems (Merkle et al, 1999; Egilmez et al, 2007).
Three-dimensional fluoroscopy has the potential to provide images with a level of quality equivalent to CT. This modality has proven useful in other disciplines such as orthopedic surgery (Kendoff et al, 2009). In a study of this technology applied to percutaneous access of porcine kidneys, it appeared to be effective (Soria et al, 2009), although simultaneous retrograde access and injection of contrast material was still helpful. Clinical application of three-dimensional fluoroscopy to percutaneous renal collecting system access has not yet been reported.
Three-dimensional ultrasonography accurately represents the renal collecting system (Ghani et al, 2008) and appears useful for renal imaging (Kim et al, 2008) and for teaching percutaneous access into an in vitro renal model (John et al, 2009). Given that three-dimensional ultrasonography has been applied to other therapeutic urologic applications (such as percutaneous drainage of prostatic abscesses) (Varkarakis et al, 2004), investigation of its use for clinical percutaneous access of the collecting system is anticipated.
There has been one report of a novel image localization system that projects the ultrasonographic puncture tract onto the fluoroscopy screen, which incorporates some of the advantages of both modalities (Mozer et al, 2007).
In addition to advanced image guidance of percutaneous access to the intrarenal collecting system, technologic enhancements have been applied to the initial needle puncture. The Urorobotics Laboratory at Johns Hopkins University has developed the robotic percutaneous access to the kidney with remote center of motion device (PAKY-RCM) (Cadeddu et al, 1997b). This is a robotic arm with 7 degrees of freedom that places a needle into the intrarenal collecting system as directed by the control device that pivots the tip of the needle about a fixed point on the skin. In a nonrandomized clinical trial, the PAKY-RCM system was equivalent to an expert physician in gaining access to the collecting system in terms of time and accuracy (Su et al, 2002). In a randomized trial in an in vitro kidney model, the PAKY-RCM took slightly more time but was more accurate than manual needle insertion (Challacombe et al, 2005b). This device can also be controlled at distance with telepresence technology (Bove et al, 2003; Netto et al, 2003). A percutaneous access needle with an impedance-based sensing system to detect entry into the collecting system has also been developed, but clinical use has not been reported (Hernandez et al, 2001; Roberts et al, 2002).
“Blind” Access
The upper urinary tract collecting system can also be accessed “blindly,” without any imaging guidance (Chien and Bellman, 2002). The only situation in which this should be considered is if sonography is not available and there is complete ureteral obstruction (precluding retrograde instillation of contrast material or opacification of the collecting system with intravenous contrast). The lumbar notch, also known as the superior lumbar triangle or Grynfeltt lumbar triangle, has been reported to be a reliable landmark for blind percutaneous renal access (Fig. 47–21). The lumbar notch is an area of muscular insufficiency through which hernias can occur. It is located posteriorly below the 12th rib. The superior border is the 12th rib and latissimus dorsi muscle, the lateral border is the transversus abdominis and external oblique muscles, the medial border is the quadratus lumborum and sacrospinalis muscles, and the inferior border is the internal oblique muscle. Insert a needle 3 to 4 cm deep into the notch at a 30-degree cephalad angle to enter the collecting system. Another blind approach to the collecting system is to insert a needle directly perpendicular to the body surface 1 to 1.5 cm lateral to the L1 vertebral body, which will lead directly to the renal pelvis if anatomy is normal. If fluoroscopy is available, then air and contrast material can be injected through a blindly placed needle to fluoroscopically assess its position and guide another needle if needed. In the only randomized clinical trial comparing “blind” access to image-guided access, entry into the collecting system was successful in 50% and 90% of cases, respectively (Basiri et al, 2007). Use of the technique is not recommended in most settings.
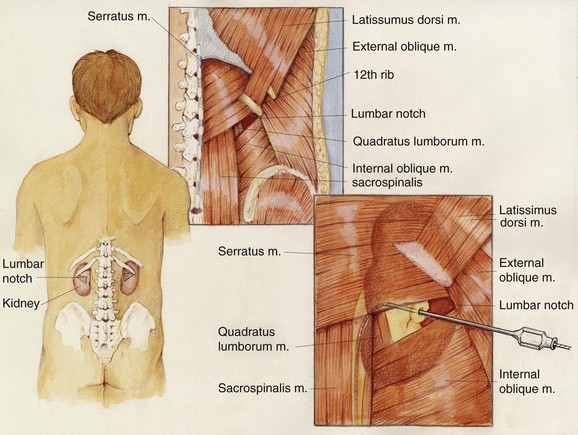
Figure 47–21 The lumbar notch is a useful anatomic landmark for blind percutaneous access to the renal collecting system. It is bounded superiorly by the latissimus dorsi muscle and the 12th rib, medially by the sacrospinalis and quadratus lumborum muscles, laterally by the transversus abdominis and external oblique muscles, and inferiorly by the internal oblique muscle.
Working Access
Once good access to the collecting system is attained, exchange the initial guidewire through a catheter for a different one as needed. Dilate the tract over the guidewire with stiff plastic dilators or a metal fascial cutter to enlarge the tract to 8 to 12 Fr. If the goal of the procedure is simple drainage, then a small-caliber nephrostomy tube can be placed over the single guidewire to complete the procedure.
Safety Wire
If ureteroscopic assistance is used, the first guidewire inserted into the kidney can be grasped with the ureteroscope and pulled down the ureter and out the external urethral meatus. With this through-and-through access, the guidewire cannot be lost. In all other situations, the goal for a therapeutic percutaneous procedure is to get two guidewires down the ureter into the bladder, generally a super-stiff (working) guidewire and a floppy tip or J-tipped PTFE-coated (safety) guidewire. One important exception is in cases in which a dependent lower pole has been accessed percutaneously. If there is extreme angulation to get from the lower pole calyx to the ureteropelvic junction and down the ureter, placing a super-stiff guidewire down the ureter may put undue force on the kidney and risk tearing parenchyma. In such cases, the flexible safety guidewire should still be directed down the ureter if possible, but the stiff working wire over which the dilation is performed can simply be directed toward the upper pole. Additionally, in some cases the pathology (e.g., calyceal obstruction, impacted ureteral stone, large staghorn stone) prevents the surgeon from getting a guidewire down the ureter or even into the remainder of the intrarenal collecting system. In such cases attempts should be made to get as much guidewire as possible into the upper urinary tract collecting system. A guidewire with a moveable core is useful in this setting because it can be coiled more tightly.
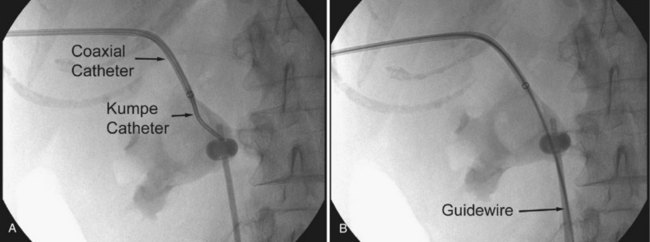
There are several techniques for getting a guidewire down the ureter. The safest maneuver is to place a stiff angled-tip hydrophilic guidewire adjacent to the initial stiff angled-tip hydrophilic guidewire using a coaxial or dual-lumen catheter. Remove the coaxial or dual-lumen catheter and place an angled tip catheter (Kumpe, Cobra, or coudé tip) over the angled-tip hydrophilic guidewire to help direct it down the ureter. Once the stiff angled-tip hydrophilic guidewire is down the ureter, optimally all the way into the bladder, use the coaxial or dual-lumen catheter to place a second guidewire down the ureter. In cases of secure access, the initial wire can be exchanged for a stiff angled-tip hydrophilic guidewire through an angled tip catheter to get down the ureter right away (Fig. 47–22); alternatively, the surgeon can use a stiff angled-tip hydrophilic guidewire as the initial wire.
Dilation of Tract
Once there is adequate wire access into the upper urinary tract collecting system, dilate the tract to allow insertion of working instruments. In most cases the goal of dilation for percutaneous renal surgery is to place a 30-Fr inner-diameter/34-Fr outer-diameter plastic access sheath. In some cases a smaller sheath, with an inner diameter measuring 12- to 24-Fr, is adequate. Renal access sheaths generally have a beveled tip, such that one side of the sheath extends farther than the other part. This bevel is used to maintain access into part of the collecting system on one side of the sheath while allowing extra mobility on the other side. Sheath repositioning is facilitated by the bevel. In cases of pathology at the edge of the collecting system, however, the bevel is a disadvantage because the entire tip of the sheath cannot be placed into the collecting system. Both opaque and clear sheaths are available; some prefer the clear sheaths because structures next to the sheath can be visualized.
In the early years of therapeutic percutaneous renal procedures, the tract was dilated gradually over the course of many days by placing sequentially larger tubes. Acute dilation of the tract was first reported by Castañeda-Zúñiga and associates in 1982 (Castañeda-Zúñiga et al, 1982). There are several dilator systems available. Regardless of the method used, is it imperative that the dilator does not pass too far into the collecting system. If this occurs, an infundibulum, the renal pelvis, or the ureteropelvic junction can be torn or perforated, either directly by the dilator or indirectly by a stone pushed aside by the dilator. The dilator should be passed just into the calyx. It is better to dilate a bit “short” of the targeted calyx than to dilate too far and create trauma. The initial inspection with the nephroscope confirms entry of the sheath into the calyx. If the sheath is not deep enough, then simply replace the dilating device and redilate after advancing the device. The dilators should not be used to dilate the skin; the skin should be incised to allow entry of the final sheath without pressure on the skin edges.
Rigid metal dilators, introduced by Alken (1985), are a series of progressively enlarging coaxial stainless steel rods that pass over an 8-Fr guide rod. The first step in tract dilation is to pass the 8-Fr guide rod over a 0.035-inch guidewire. The end of the guide rod has a ball that prevents advancement of the first dilating rod beyond the tip. The ball is positioned at the intended depth of the dilation (Fig. 47–23). After passing the first rod, each successive metal rod is passed sequentially over the former until the desired tract is achieved, up to a 30-Fr rod over which is passed a 30/34-Fr plastic sheath. The advantages of the rigid metal dilator system are that it is the most effective dilator, able to dilate even when there is dense perirenal scarring from prior procedures, and that is it inexpensive on a per-case basis because it is reusable. The disadvantage is that, for the same reason they are so effective, the rigid metal dilators can do considerable damage. The depth of the dilation can be hard to maintain accurately, especially when pushing hard against tough scar tissue. One group has modified the rigid metal rods, tapering the ends and adding centimeter markings (Shen et al, 2007).
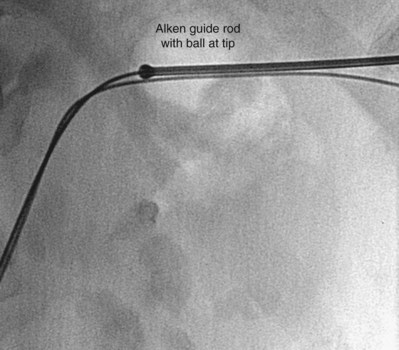
Figure 47–23 Alken guide rod is positioned with the ball at the tip of the rod positioned at the intended depth of the dilation.
Semirigid plastic dilation sets (often referred to as “Amplatz” dilators, after Kurt Amplatz, the senior author in the initial publication [Rusnak et al, 1982]) consist of progressively larger firm plastic (polyurethane) dilators that are passed over an 8-Fr PTFE guiding catheter that fits over a 0.035-inch guidewire (Fig. 47–24). The dilators are passed one after the other, not coaxially like the rigid metal dilators but progressively, advancing one dilator, removing it, advancing the next largest dilator, and so on until the final tract diameter is achieved. The working sheath is passed over the final dilator and then the dilator and 8-Fr catheter are removed, leaving the working wire and sheath in place. The dilators are made in increments of 2 Fr, but if the tissue being dilated is soft, then not every dilator needs to be used. The advantages of the semirigid plastic dilation system are that trauma to the collecting system is theoretically less likely than with the rigid metal dilators (although experienced urologists have found no difference between the two systems in terms of safety), but the disadvantage is that hemorrhage can occur each time a dilator is withdrawn. Current semirigid plastic dilators are sold as disposable devices, so they are more expensive on a per-case basis than rigid metal dilators.
Balloon dilators (Fig. 47–25) were developed to obviate the repetitive dilations of the rigid metal and semirigid plastic dilation systems, which are both time consuming and potentially dangerous. This is the most common dilation method for percutaneous renal surgery today (Benway and Nakada, 2008). The appropriate working sheath is back-loaded onto the balloon dilation catheter, which is passed over the working wire until the radiopaque marker is at the intended depth of dilation (Fig. 47–26). The dilating balloon is inflated with a pressure syringe. A “waist” appears at the site(s) of greatest resistance, usually the abdominal wall fascia and the renal capsule (Fig. 47–26B). Once the balloon is fully expanded (Fig. 47–26C), the working sheath is passed over the balloon (Fig. 47–26D) (Fig. 47–27 on the Expert Consult website![]() ). The balloon catheter has a “shoulder,” which is the portion between the end of the balloon and the point at which the maximal diameter is achieved. The sheath should not be passed beyond the maximal diameter of the balloon because this can cause significant injury. Balloon dilators, which are expensive one-time use devices, are less effective than rigid metal and semirigid plastic dilation systems in densely scarred tissue but are more effective when the kidney is hypermobile (Kumar and Keeley, 2008). Most (Heggagi et al, 1991; Davidoff and Bellman, 1997; Safak et al, 2003; Kukreja et al, 2004), but not all (Gonen et al, 2008a), studies have suggested that hemorrhage and transfusion rates are less with the balloon dilators compared with rigid metal and semirigid plastic dilators.
). The balloon catheter has a “shoulder,” which is the portion between the end of the balloon and the point at which the maximal diameter is achieved. The sheath should not be passed beyond the maximal diameter of the balloon because this can cause significant injury. Balloon dilators, which are expensive one-time use devices, are less effective than rigid metal and semirigid plastic dilation systems in densely scarred tissue but are more effective when the kidney is hypermobile (Kumar and Keeley, 2008). Most (Heggagi et al, 1991; Davidoff and Bellman, 1997; Safak et al, 2003; Kukreja et al, 2004), but not all (Gonen et al, 2008a), studies have suggested that hemorrhage and transfusion rates are less with the balloon dilators compared with rigid metal and semirigid plastic dilators.
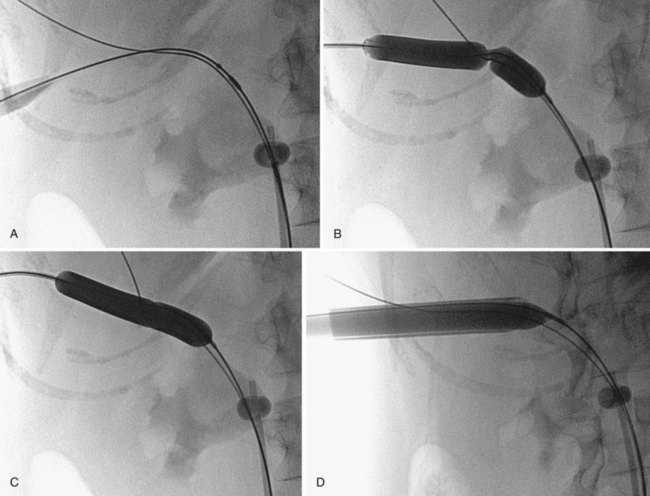
Figure 47–26 Balloon dilation of tract and placement of working sheath. A, Balloon catheter is inserted over wire, with distal radiopaque marker is at the intended depth of dilation. B, “Waist” appears as balloon is inflated. C, Balloon is fully expanded. D, Sheath is passed over balloon, taking care not to advance it beyond the point of maximal diameter of the balloon.
Figure 47–27 Working sheath being passed over dilating balloon in lower pole access site. The working and safety guidewires are both going down the ureter.
In an effort to further simplify dilation of the renal access tract, a number of single-step techniques have been described. The simplest is passage of the final semirigid plastic dilator without prior dilation by the smaller dilators (Frattini et al, 2001). This technique is reported to work well in comparison with other methods, even in patients with prior renal surgery, but the forward force that needs to be applied is appreciable (Ziaee et al, 2007; Amjadi et al, 2008; Falahatkar et al, 2009). Devices designed specifically for single-step dilation include a balloon dilator with an expandable sheath (Pathak and Bellman, 2005; Baldwin et al, 2006; Maynes et al, 2008) and a rigid dilator with an expandable sheath (Goharderakhshan et al, 2001). Preliminary results with these devices appear favorable.
The most common cause of difficult tract dilation is prior renal surgery (Joel et al, 2005). Densely scarred kidneys present a challenge. If even semirigid plastic or rigid metal dilators fail, novel uses of devices such as Collings knives and atherotomy cutting balloons can be employed (Davis et al, 1991; Williams et al, 2008).
Modifications in Special Situations
In cases of anomalous kidneys (malrotated, ptotic, ectopic, horseshoe, and other fused kidneys), alteration of the percutaneous approach to the upper urinary tract collecting system may be necessary. As noted earlier, in some situations real-time guidance of the needle puncture with CT or MRI might be considered, but in most cases preoperative CT or magnetic resonance suffices. Because the orientation of some organs might change with patient position, imaging in the intended position of surgery might be useful. Preoperative cross-sectional imaging of anomalous kidneys helps plan patient position, choice of calyx, and orientation of the tract, taking into account distance of the kidney from the skin, calyceal orientation, vasculature, and the relative orientation of adjacent organs.
Horseshoe kidneys frequently require percutaneous intervention. Although antegrade access into horseshoe kidneys is somewhat different than into normal kidneys, standard techniques can generally still be used. CT or MRI should be considered for preoperative assessment of horseshoe kidneys, both to assess for the possibility of retro-renal colon (Skoog et al, 1985) and to assess the vasculature and relationship of the calyces to the anticipated puncture site. In a series of 12 patients undergoing percutaneous nephrolithotomy in horseshoe kidneys, 5 had bowel posterior to the kidney on CT (Al-Otaibi and Hosking, 1999). Horseshoe kidneys often have extra and eccentric calyces that can be hard to access. In other ways, however, percutaneous access to a horseshoe kidney is more favorable than in normal kidneys. The antero-postero tilt of the kidney is prominent, which makes the upper pole the most superficial and posterior aspect of the horseshoe kidney. In addition, the upper pole is usually inferior to the ribs. Upper pole access is useful in horseshoe kidneys because this is the easiest calyx to enter, the puncture rarely needs to be supra-costal, and it provides excellent access to most of the kidney and the ureter owing to the alignment of long axis of the moiety (Fig. 47–28). The initial entry into a horseshoe kidney is more medial than in normal kidneys and can pass through the paraspinous musculature. The distance to the lower pole and ureter can be great in an obese or muscular patient, such that extra-long rigid nephroscopes or flexible nephroscopy may be necessary. In some cases, middle calyceal access is preferred because the upper pole is so far away from the pathology, but lower pole calyces are usually not safely accessible with direct percutaneous puncture. The vasculature of horseshoe kidneys is aberrant, but vessels enter and exit the kidney in an antero-medial location (except for some at the isthmus), so direct vessel injury is rare with well-planned access (Janetschek and Kunzel, 1988). Overall, among a total of 256 percutaneous procedures (primarily nephrolithotomy) recently reported in horseshoe kidneys, there were 11 (4.3%) major hemorrhagic complications and only 1 (<0.4%) colon injury (Al-Otaibi and Hosking, 1999; Shokeir et al, 2004; Lojanapiwat, 2005; Darabi Mahboub et al, 2007; Mosavi-Bahar et al, 2007; Viola et al, 2007; Majidpour and Yousefinejad, 2008; Miller et al, 2008; Symons et al, 2008; Gupta et al, 2009).
Figure 47–28 Percutaneous access sheath (radiolucent except for a radiopaque stripe) in upper pole of horseshoe kidney. Note medially directed lower pole calyx. Although not apparent from this radiograph, the access site is subcostal.
Another way to access anomalous kidneys is with an anterior approach using laparoscopic assistance. This is most applicable to pelvic ectopic kidneys because a posterior approach is often blocked by the bony pelvis, although laparoscopic assistance has been used for percutaneous nephrolithotomy of a large calculus in an anteriorly directed calyx in the isthmus of a horseshoe kidney (Maheshwari et al, 2004b) and a large calculus in an anterior diverticulum of a horseshoe kidney (Wong and Zimmerman, 2005). The bowels can be mobilized off of the surface of the kidney laparoscopically, and the access needle is passed through the anterior abdominal wall into the kidney under vision from the laparoscope. Retrograde assistance is still desirable because opacifying the collecting system assists in simultaneous fluoroscopic confirmation of calyceal entry and wire passage. Dilation and sheath insertion can be assessed both laparoscopically and fluoroscopically. In 10 series of laparoscopically directed antegrade percutaneous nephrolithotomy into pelvic kidneys published since 2001 that described a total of 33 patients, all procedures were reported to be successful and without complications (Troxel et al, 2002; Maheshwari et al, 2004a; Santos et al, 2004; Aron et al, 2005b; Aquil et al, 2006; Goel et al, 2006; Matlaga et al, 2006; El-Kappany et al, 2007; Mousavi-Bahar et al, 2008; Gupta et al, 2009). As a simpler alternative to laparoscopic assistance, anterior percutaneous access into pelvic kidneys using a supine oblique position with a pad under the ipsilateral hemipelvis, as well as using ultrasonography to assess for intervening bowel, has been reported to be a safe technique in selected patients (Desai and Jasani, 2000; Mosavi-Bahar et al, 2007). If bone does not intervene between the appropriate calyx and the skin, then a posterior approach can be used just above the iliac bone (Atmaca et al, 2007) or through the greater sciatic foramen (Watterson et al, 2001).
Transplanted kidneys also mandate changes in the approach to percutaneous access of the upper urinary tract. The transplanted kidney, typically positioned extraperitoneally in the iliac fossa, has variable angles of the calyces depending on the placement of the kidney by the transplant surgeon. The usual approach is from the antero-lateral direction, with the patient in the supine position. Percutaneous nephrostomy is the preferred approach to obstruction and for endoscopy of the collecting system in the transplant kidney (Mostafa et al, 2008). Ultrasonography is most convenient for initial access to the collecting system because retrograde assistance for fluoroscopy is difficult owing to the site and angle of the reimplanted ureter. Fluoroscopy can then be used as needed to facilitate subsequent steps. The dense fibrosis that often surrounds a transplant kidney can make dilation of the tract difficult, and semirigid plastic or rigid metal dilators may be necessary if balloon dilation fails. Of 42 percutaneous procedures in transplant kidneys reported in six recent series, using ultrasonographically directed percutaneous access with or without adjunctive fluoroscopy, all but one were technically successful and there were no complications (Francesca et al, 2002; Klingler et al, 2002; Challacombe et al, 2005a; He et al, 2007; Krambeck et al, 2008a; Rifaioglu et al, 2008).
Key Points: Obtaining Percutaneous Access
Postprocedural Nephrostomy Drainage
The final surgical consideration after percutaneous renal surgery is which drain(s), if any, to insert into the upper urinary tract collecting system. Options include an externalized nephrostomy tube or nephroureteral stent, an internal or externalized ureteral stent, or no drainage tube at all. Additional discussion of these options can be found in Chapter 7.
Nephrostomy Tube
For years the standard drainage after percutaneous surgery of the upper urinary tract collecting system has been an externalized nephrostomy tube. There are a variety of choices for postoperative nephrostomy tubes.
Balloon Catheters
The Foley and Councill catheters used for transurethral drainage can be used as nephrostomy tubes as well (Fig. 47–29). These tubes come in a range of diameters; typically 16- to 24-Fr catheters are used for postoperative nephrostomy drainage. The 5-mL retention balloon might be too large for some collecting systems and does not need to be completely inflated. The balloon can cause calyceal obstruction if it is pulled into an infundibulum. Saline or water should be used to inflate the balloon because viscous contrast material might hinder emptying of the balloon when removal is attempted. An advantage of the Councill catheter is the ability to pass a small-caliber catheter through the end hole and down the ureter, providing more secure access to the upper urinary tract collecting system and maintaining ureteral patency. All nephrostomy tubes, even ones with robust internal retention devices, should be fixed to the skin externally with a suture or other mechanism. External fixation of a tube, however, does not necessarily prevent internal dislodgement of the tube. Especially in a large patient, the distance from the skin to the upper urinary tract collecting system can change with patient movement, and the tube (fixed at the skin) can pull out of the kidney. The potential for tube dislodgement is one of the best arguments in favor of nephrostomy tubes that have some extension down the ureter to maintain a conduit to the upper urinary tract collecting system even if the renal pelvic portion of the tube is pulled out of the kidney.
Malecot Catheter
The wings of the Malecot catheter expand when the catheter is at rest, providing a modest but atraumatic and nonobstructive retention mechanism (Fig. 47–30A). When the catheter is being placed or removed, a stiffener is inserted through it to push on the distal end of the Malecot tip and straighten the wings. During removal this stiffener can misalign with the Malecot tip if the tube is not straight; pulling the catheter back until the tube is straight helps the stiffener line up properly. The Malecot catheter is also available with an extension that is directed down the ureter. This modification is called a “re-entry” catheter because it simplifies placing a guidewire through the Malecot catheter and down the ureter into the bladder (see Fig. 47–30A). The extension is long enough (18 cm) so that in most patients the Malecot tube can be withdrawn until the wings are externalized and a guidewire can be placed into the ureter. Malecot catheters for renal use are large-bore catheters, ranging from 16 to 30 Fr, although Malecot catheters as small as 8 Fr are available.
Cope Catheter
A more secure retention mechanism is provided by Cope nephrostomy tubes. A string exits the catheter a few centimeters from the distal tip and then re-enters the catheter near the tip (Fig. 47–31). Pulling on the string forms a secure coil that is not easily dislodged from the renal pelvis. The string is fixed at the external end of the tube with a locking mechanism or by wrapping it around the tube and fixing it in place with a rubber cuff. Cope catheters employ the same coil shape used in pigtail ureteral stents. The active reinforcement of the coil strength by the string provides more secure retention than the passive coil of a pigtail, and as such Cope catheters have replaced pigtail catheters for most percutaneous uses. Cope nephrostomy tubes, ranging from 6 to 14 Fr in diameter, can be used for simple upper urinary tract drainage and instillation procedures, as well as after percutaneous surgery.
Nephroureteral Stent
The Cope retention mechanism is also used in nephroureteral stents. A nephroureteral stent has a renal coil like that of a Cope nephrostomy tube, but the tube continues on to a ureteral extension that travels down the ureter to end in a passive pigtail that rests in the bladder (Fig. 47–32). The ureteral portion can be the same diameter as the nephrostomy portion, or it can be narrower. A nephroureteral stent is passed percutaneously over a wire that ends in the bladder. Once the end is coiled generously in the bladder, careful inspection of the fluoroscopy image reveals the location of the side holes in the renal coil. By moving the catheter in and out while pulling on the string and rotating the external portion of the tube clockwise, the Cope retention coil is formed in the renal pelvis (Figs. 47-33 and 47-34 on the Expert Consult website![]() ) A nephroureteral stent offers excellent control of the entire upper urinary tract, from renal pelvis to bladder, and is unlikely to become dislodged. Nephroureteral stents are available in diameters of 8.5 or 10.2 Fr, and the standard lengths (from renal to bladder coil) are 20 to 28 cm.
) A nephroureteral stent offers excellent control of the entire upper urinary tract, from renal pelvis to bladder, and is unlikely to become dislodged. Nephroureteral stents are available in diameters of 8.5 or 10.2 Fr, and the standard lengths (from renal to bladder coil) are 20 to 28 cm.
Circle Catheter
A final type of nephrostomy tube is the circle nephrostomy tube (Fig. 47–35), which is secure, easily exchanged, causes little trauma, rarely occludes, and provides excellent drainage and avenue for irrigation of the renal pelvis. The circle nephrostomy tube requires two percutaneous access sites to the kidney, and this tube is most useful when maintenance of two tracts is desired, such as for irrigation of the renal pelvis or if more than one access is necessary for second-look nephroscopy (Kim et al, 2005). After obtaining access at two distant calyces, a flexible nephroscope or flexible ureteroscope passed over one wire is used to grasp the wire coming from the other site. When the endoscope is withdrawn, the wire is now in position to guide placement of the circle nephrostomy tube. Radiopaque markers on the tube delineate the location of the drainage holes, which should be maintained within the intrarenal collecting system. External drainage of the circle nephrostomy tube requires a Y-connector.
General Considerations
The advantages of a postoperative nephrostomy tube include good drainage and control of the upper urinary tract, and maintenance of percutaneous access for additional procedures. It was initially thought that a postoperative nephrostomy served to tamponade the nephrostomy tract and reduce hemorrhage, but subsequent studies have suggested that is not the case. When hemorrhage does occur, however, the larger caliber of a nephrostomy tube provides better drainage of the upper urinary tract collecting system than an internal ureteral stent. In addition, if a large perforation has occurred during the procedure, the additional diversion of urine away from the site might be advantageous. When a nephrostomy tube is left in place following percutaneous renal surgery, it is usually in the dilated access site. At least one group has attempted to reduce the discomfort associated with supracostal percutaneous renal surgery by placing a small-caliber postoperative nephrostomy tube in a new subcostal site and leaving the dilated supracostal access site without a nephrostomy tube (although there was no control cohort for comparison) (Kim et al, 2006).
Including along with the nephrostomy a tube that goes down the ureter provides the greatest control and assurance of drainage. Because entry of a tube into the bladder is associated with additional symptoms, though, such a tube should only be used when needed. Considerations include the size of the patient (which determines to a large extent the risk of tube dislodgement), the importance of maintaining drainage, and the desire for ureteral intubation (e.g., ureteral obstruction that might resolve if intubated, ureteral injury that should be bypassed). Aside from the choice of retention mechanism, the main remaining consideration is the diameter of the nephrostomy tube.
A number of studies have compared the impact of nephrostomy tube diameter after percutaneous renal surgery including one prospective trial with alternating assignment to 28-Fr versus 9-Fr nephrostomy tubes (Maheshwari et al, 2000) and four randomized controlled trials comparing large-caliber tubes (20 to 24 Fr) to small-caliber tubes (8 to 18 Fr) (Liatsikos et al, 2002; Pietrow et al, 2003; Desai et al, 2004; Marcovich et al, 2004). Among the five studies, comprising a total of 170 patients with nephrostomy tubes, four revealed less pain and two reported less urinary leakage in the patients with smaller tubes. Bleeding was not increased in the groups with smaller tubes in any of the studies. Only one study found no benefit to the smaller tube (Marcovich et al, 2004). Although tube diameter is not related to bleeding overall, the removal of larger tubes occasionally can be followed by immediate hemorrhage; this is rare with smaller tubes. As such, large-caliber nephrostomy tubes should be removed in a radiology suite where there is the opportunity for immediate replacement of the tube. Small-caliber tubes can be removed safely at the bedside after a period of clamping to assess clinically for distal ureteral obstruction.
“Tubeless” with Ureteral Stent
A “tubeless” percutaneous procedure—one that omits the postoperative nephrostomy tube—was initially proposed by Wickham and colleagues (1984). This practice never met with widespread acceptance, especially after Winfield and colleagues (1986) reported disastrous outcomes with this technique. The concept was revived in 1997 by Bellman and colleagues (1997), with the addition of an internal ureteral stent left in place for a week or two. Since then, many studies have evaluated the practice of omitting the nephrostomy tube after percutaneous renal surgery. Although this technique is called “tubeless,” most series employ a ureteral stent for at least a short period postoperatively.
Options for ureteral stenting without a nephrostomy tube after percutaneous renal surgery include an internal ureteral stent that is removed cystoscopically, an internal ureteral stent with an attached string that exits out the flank to allow removal of the ureteral stent without cystoscopy, and an externalized (out the urethra) ureteral stent that is removed along with the urethral catheter to which it is attached. The potential advantages of omitting the nephrostomy tube after percutaneous renal surgery include decreased pain and analgesic use, avoidance of an external drainage device, abbreviated hospital stay, and decreased health care costs (secondary to shortening the duration of hospitalization). The seminal report of Bellman and colleagues (1997) described placing an internal ureteral stent antegrade, looking at the tract through the nephroscope as the sheath is withdrawn, and leaving a percutaneous guidewire in place for a few hours in the recovery room before removing it if there is no hemorrhage. Among 50 patients undergoing tubeless percutaneous renal procedures, compared with a cohort of patients with a nephrostomy tube placed after surgery, hospitalization was briefer, narcotic use was less, return to normal activities was sooner, and costs were less. Since then, many studies including several randomized controlled trials have evaluated omission of nephrostomy tube postoperatively with placement of an internal ureteral stent (Table 47–1). It is important to note that none of these studies included patients with significant bleeding or perforation, or in whom secondary nephroscopy was intended.
Table 47–1 Randomized Controlled Trials of PCN versus Internal Ureteral Stent after Percutaneous Renal Surgery
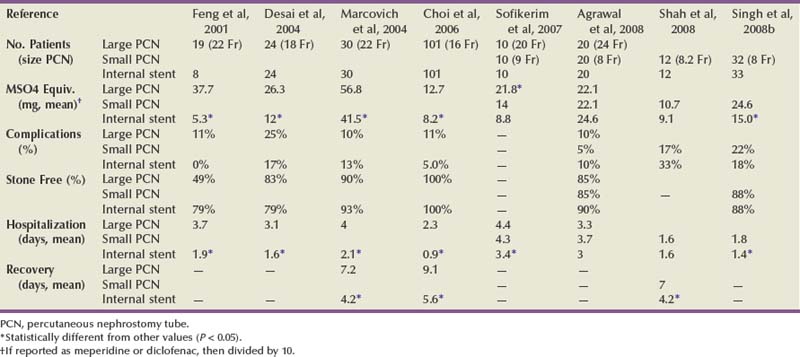
Of the six trials in Table 47–1 (Feng et al, 2001; Desai et al, 2004; Marcovich et al, 2004; Sofikerim et al, 2007; Agrawal et al, 2008; Singh et al, 2008b) that compared postoperative drainage with a large-caliber percutaneous nephrostomy tube (16 to 24 Fr) to drainage with an internal ureteral stent and no nephrostomy tube, five revealed a benefit to omission of the nephrostomy tube in terms of reduced narcotic use and decreased length of hospitalization. Both studies that evaluated return to normal activity found a benefit to omitting the nephrostomy tube, and in one study (Feng et al, 2001) omission of the nephrostomy tube was associated with decreased cost. Among the four studies (Desai et al, 2004; Marcovich et al, 2004; Choi et al, 2006; Shah et al, 2008) that compared postoperative drainage with a small-caliber percutaneous nephrostomy tube (8 to 9 Fr) with drainage with an internal ureteral stent and no nephrostomy tube, the benefit of omitting the nephrostomy tube was less apparent, with only one study suggesting a decrease in narcotic use, two indicating a reduced hospital stay, and one finding a more rapid return to normal activity. In none of these comparisons in Table 47–1, however, did there seem to be any danger to nephrostomy tube omission; technical success rates and complications appeared similar between the groups.
There are some disadvantages to using an internal ureteral stent as an alternative to a nephrostomy tube, however, including loss of the percutaneous tract for a secondary procedure and the cost, inconvenience, and discomfort associated with an internal ureteral stent that requires cystoscopic removal at a later date. In order to obviate the problems associated with the ureteral stent, several groups have offered alternatives including insertion of an externalized ureteral stent or insertion of an internal stent with an attached string that exits out the flank. In both cases the stent can then be removed before hospital discharge without an additional procedure. Goh and Wolf (1999) first reported in 1999 the use of an externalized ureteral stent (single pigtail) as an alternative to a postoperative nephrostomy tube. Since then there have been a number of reports of this option (Lojanapiwat et al, 2001; Abou-Elela et al, 2007; Karami et al, 2007; Rana and Mithani, 2007; Al-Ba’adani et al, 2008) including two randomized controlled trials, one comparing an externalized ureteral stent to a large-caliber nephrostomy tube, which suggested a benefit in terms of reduced narcotic use and length of hospital stay (Tefekli et al, 2007), and one comparing internal and external ureteral stents, which revealed no difference between the two techniques except for a lack of outpatient stent-related symptoms in the external stent group (Gonen et al, 2009). Shpall and colleagues (2007) described the modification of leaving the attached string on the stent and placing the stent “upside down” so that the string can exit the flank. They described removing the stent as an outpatient 3 and 12 days postoperatively, but since then others have reported removing the stent at the bedside on the first postoperative day (Berkman et al, 2008). Use of a Polaris Loop stent (Boston Scientific, Natick, MA) placed “upside down” offers less resistance at the time of removal (Fig. 47–36).
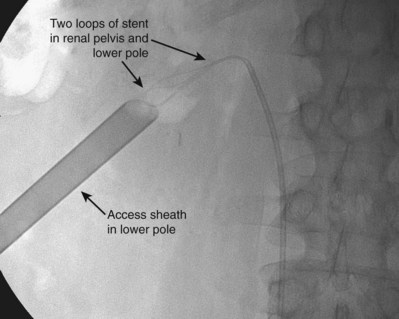
Figure 47–36 Use of a Polaris Loop stent (Boston Scientific, Natick, MA) placed “upside down.” An attached string is left exiting the flank, which allows removal of the stent without an additional procedure.
These modifications of nephrostomy tube omission avoid many of the disadvantages of an internal ureteral stent but still leave the problem of loss of access in case a secondary procedure is required. With improved endoscopes, better ancillary tools, and growing experience with percutaneous surgery, the need for secondary procedures is declining. In properly selected patients including those who do not for some other reason need external drainage (e.g., pyonephrosis, significant bleeding, significant collecting system injury) and those who are unlikely to need a secondary procedure, omission of the postoperative nephrostomy tube appears to be safe and effective.
No Drainage Tube
Recently, some authors have reintroduced the idea of a “totally tubeless” percutaneous renal surgery, omitting both the nephrostomy tube and ureteral catheter. The procedure is done in selected patients, with low-volume stones, atraumatic single access, and no hemorrhage, perforation, or obstruction (Aghamir et al, 2004; Karami and Gholamrezaie, 2004; Gupta et al, 2005; Mandhani et al, 2007; Crook et al, 2008b; Mouracade et al, 2008). In light of the findings of studies comparing internal stents with nephrostomy tubes, it is not surprising that the three randomized controlled trials of “totally tubeless” percutaneous renal surgery summarized in Table 47–2 (Aghamir et al, 2008; Crook et al, 2008a; Istanbulluoglu et al, 2009) also found reduced duration and intensity of convalescence in the groups without nephrostomy tubes. The more important comparison would be “totally tubeless” versus internal stent without nephrostomy tube, which to date has not been reported.
Adjuncts to Drainage without Nephrostomy Tubes
One of the concerns with any approach that omits the nephrostomy tube after percutaneous renal surgery is that of hemorrhage from the tract. Although the evidence from randomized controlled trials suggests that the size or presence of the nephrostomy tube does not affect the degree of postoperative bleeding, there is still a potential for dramatic bleeding after any percutaneous procedure—and such bleeding would be more problematic without a nephrostomy tube in place. Several groups have described adjuncts intended to enhance hemostasis of the percutaneous tract including direct monopolar cauterization of the tract (Mouracade et al, 2008), cryotreatment of the tract (Okeke et al, 2009), and insertion/instillation of hemostatic agents into the tract including oxidized cellulose (Aghamir et al, 2006), gelatin sponge (Singh et al, 2008a), gelatin granules plus thrombin (Lee et al, 2004b; Nagele et al, 2006), and fibrin glue (Noller et al, 2004). One nonrandomized comparison of percutaneous nephrolithotomy with and without electrode cauterization of the tract reported a lower blood transfusion rate in the first group (1.2% vs. 6.5%, respectively) (Jou et al, 2004). A nonrandomized comparison of tract cryotherapy without a nephrostomy tube in 30 patients versus a 24-Fr nephrostomy tube without tract cryotherapy in 30 patients suggested that there was shorter hospitalization and less hemorrhage and urine leakage in the patients treated with cryotherapy. The high rate of postoperative hemorrhage requiring angio-embolization in the control group, 13%, confounds the analysis (Okeke et al, 2009). There have been three randomized controlled trials of hemostatic adjuncts for percutaneous renal surgery without nephrostomy tubes, comparing patients managed without hemostatic adjuncts with those managed with gelatin sponge (Singh et al, 2008a), oxidized cellulose (Aghamir et al, 2006), and fibrin glue (Shah et al, 2006). None of the three studies found an impact of the adjunct on measures of hemostasis (postoperative hematocrit, hemorrhage, and/or blood transfusions). Narcotic use was reduced in both studies in which it was evaluated. Hospital stay and urinary leakage were each assessed in two studies, and each was improved in one study. Some nonrandomized studies also have suggested a shortened hospitalization and/or reduced narcotic use after tract hemostatic maneuvers, but because improvements in hemostasis and urinary leakage are not consistently found, the mechanism for improvement in these secondary outcomes is not clear (Mikhail et al, 2003; Aron et al, 2004; Borin et al, 2005). Overall, the utility of any of adjuncts for tract hemostasis is not certain and further study is necessary.
Key Points: Postprocedural Nephrostomy Drainage
Training in Percutaneous Access and Procedures
Efforts to enhance training for percutaneous surgery of the upper urinary tract have focused on obtaining percutaneous access. Summarizing their own experience and other reports (Allen et al, 2005; Tanriverdi et al, 2007), de la Rosette and colleagues (de la Rosette et al, 2008a) estimated that a trainee must perform approximately 24 percutaneous nephrolithotomies to attain proficiency, whereas competence is not achieved until the experience includes 60 cases and excellence is not obtained until more than 100 cases have been performed. The American College of Radiology’s “Practice Guideline for the Performance of Percutaneous Nephrostomy” recommends that a physician must have performed at least 15 percutaneous nephrostomies as primary operator, with acceptable outcomes, in order to be considered qualified as a supervising physician (ACR, 2007). Training during residency is the most effective way to develop the skills, but maintenance of skills requires ongoing experience. In a survey of urologists who completed a single residency program, those trained in percutaneous access were more likely to perform percutaneous surgical procedures than those not trained in percutaneous access (92% vs. 33%). Of those who performed percutaneous surgery, urologists with training did more procedures than those without training (14.0 vs. 3.3 procedures annually). However, only about a third of respondents in both groups continued to attain their own percutaneous access (27% vs. 11%) (Lee et al, 2004a).
Nonhuman models can also assist training (Laguna et al, 2002). An inanimate trainer is marketed by Limbs and Things, Ltd (Bristol, UK). Several biologic models using ex-vivo porcine kidneys incorporate a body wall simulator composed of a chicken carcass (Hammond et al, 2004; Hacker et al, 2007), full-thickness porcine skin including subcutaneous tissue and muscle (Zhang et al, 2008), or silicon (Strohmaier and Giese, 2005). The most advanced trainer is the PERC Mentor (Simbionix Ltd, Lod, Israel), a computer-assisted simulator for percutaneous access procedures guided by fluoroscopy (Fig. 47–37). Some of the biologic models can be used to teach steps in the subsequent procedure (e.g., nephrolithotomy, endopyelotomy), but the main focus is on attaining percutaneous access. The PERC Mentor has undergone preclinical validation, confirming that training on the PERC Mentor improves performance on the PERC Mentor (Knudsen et al, 2006), and that trained individuals more proficiently attain percutaneous renal access in a porcine in-vivo model (Margulis et al, 2005), but validation of the impact on performance in the human operating room is lacking (Stern et al, 2007). At the least, these trainers can serve as an introduction for the trainee by orienting them to the necessary anatomic considerations and familiarizing them with the physical movements of the procedure. It is unknown if these or other devices can be used to validate the skills of trainees and practitioners.
Taking simulation even farther, Bruyère and colleagues (2008) used computer-assisted design and rapid prototyping based on CT images to create a silicon model of a patient’s kidney, complete with artificial flank and respiratory motion. After practicing percutaneous nephrolithotomy on the model several times, the percutaneous nephrolithotomy on the patient went smoothly using the practiced technique. Creating a specific renal model for each patient is too expensive and laborious for routine application currently, but with anticipated technical improvements, this iteration of “personalized medicine” might play a role in training and treatment planning.
Complications
Given enough time and access sites to the upper urinary tract collecting system, in most cases the primary goal of the percutaneous procedure can be achieved. The challenge with percutaneous surgery, then, is to do so while also avoiding complications. If complications do occur, then prompt recognition and management will usually avoid major morbidity. Some complications of percutaneous renal surgery are specific to the procedure including migration of stone outside the kidney, retained foreign bodies such as tips of wires and probes, and tumor seeding of the percutaneous tract. In this section the general complications of percutaneous renal surgery are discussed.
Acute Hemorrhage
Acute hemorrhage is the most common significant complication of percutaneous access into the upper urinary tract collecting system. Percutaneous nephrostomy alone results in hemorrhage requiring transfusion in 0.5% to 4% of procedures (Radecka and Magnusson, 2004; Wah et al, 2004; ACR, 2007; Rana et al, 2007). With the addition of percutaneous nephrolithotomy, likely owing to the larger caliber of the percutaneous tract and increased intrarenal manipulation, the incidence of hemorrhage to the point of transfusion rises to 6% to 20% (Kukreja et al, 2004; Netto et al, 2005; Preminger et al, 2005; Muslumanoglu et al, 2006; Chew et al, 2009). This wide variation in transfusion rate, which likely is more variable than the rate of significant hemorrhage, reflects differences in the complexity of the procedure, patient factors, and physician triggers for use of blood products. Factors associated with hemorrhage during percutaneous surgery include patient characteristics, multiple access sites, supracostal access, increasing tract size, tract dilation with methods other than balloon dilation, prolonged operative time, and renal pelvic perforation (Stoller et al, 1994; Martin et al, 1999; Kukreja et al, 2004; Netto et al, 2005; Hegarty and Desai, 2006; Chew et al, 2009).
Technical errors predispose to hemorrhage as well. Infundibular entry risks injury to interlobar (infundibular) arteries. Access into an anterior calyx or any calyx that does not afford direct access to the pathology invites overly aggressive torquing of the sheath and rigid endoscope, which also can lead to hemorrhage. If direct access cannot be obtained, then flexible instrumentation should be considered. Additionally, misuse of any tool—lithotrites (ultrasonic, pneumatic, electro-hydraulic, or laser), resectoscopes, wires, sheaths, graspers, baskets, and so on—can cause hemorrhage.
Most hemorrhage occurs from the renal parenchyma, and in most cases this hemorrhage is not significant. Small arteries and veins are always injured to some degree by percutaneous entry into the kidney. Parenchymal bleeding is minimized by proper entry and dilation and by careful manipulation of the sheath, but it still can occur. The access sheath provides intraoperative tamponade of parenchymal bleeding. Postoperatively, hemostasis is achieved by collapse of the parenchyma onto itself. Unless the postoperative nephrostomy tube is as large as or larger than the sheath used during the procedure, it likely does not contribute to hemostasis. There is no difference in measures of postoperative bleeding between small (8- to 18-Fr) and large (20- to 28-Fr) tubes (Maheshwari et al, 2000; Liatsikos et al, 2002; Pietrow et al, 2003; Desai et al, 2004; Marcovich et al, 2004), and randomized controlled trials suggest that hemorrhage is no greater when the nephrostomy tube is omitted altogether (Feng et al, 2001; Desai et al, 2004; Marcovich et al, 2004; Choi et al, 2006; Sofikerim et al, 2007; Tefekli et al, 2007; Aghamir et al, 2008; Agrawal et al, 2008; Crook et al, 2008a; Shah et al, 2008; Singh et al, 2008b; Gonen et al, 2009; Istanbulluoglu et al, 2009). If there is noticeable bleeding from the tract after sheath removal following an otherwise unremarkable procedure, this suggests bleeding from intraparenchymal vessels. Hemostatic maneuvers such as cauterization or placement of hemostatic material can be considered, but in general the best management is to insert and occlude a nephrostomy tube, apply pressure to the incision, and let the collecting system clot off. A tubeless approach is not advised in such cases because maintenance of percutaneous access to the upper tract might facilitate management. Nephrostomy tubes should not be irrigated the day or evening of the procedure if they are not draining; it is best to let the collecting system remain occluded to tamponade bleeding. By the next morning, it is safe to gently irrigate the tube because hemostasis is more certain.
If the procedure was not complicated by bleeding, but severe hemorrhage occurs following sheath removal and is refractory to the hemostatic measures described earlier, then use of a Kaye Nephrostomy Tamponade Balloon (Cook Urological, Spencer, IN) should be considered. This is a nephrostomy tube surrounded by a balloon (Fig. 47–38) (Kaye and Clayman, 1986), which is inflated up to 36 Fr to tamponade the parenchymal bleeding just as the sheath did intraoperatively. This device should be removed under fluoroscopic guidance with guidewire access down the ureter in case tube reinsertion is required for recurrent bleeding.
Intraoperative hemorrhage from an injured vein or artery within the collecting system mandates cessation of the procedure if vision is lost. In most cases, especially if the injury appears to be venous, then placing a nephrostomy tube and letting the collecting system clot off is effective. If this is not effective, then Gupta and colleagues (1997) have described inserting a Councill catheter as a nephrostomy tube, with the balloon inflated slowly at the site where contrast material enters into the venous system until repeated nephrostography reveals no more extravasation of contrast material (Fig. 47–39). A hole should be cut into the tube proximal to the balloon to provide drainage of calyces obstructed by the balloon (Fig. 47–40). An additional alternative, described in one case of a large venous injury, is to place a large-bore catheter through the injury site into the main renal vein and then back it out several days later (Shaw et al, 2005). A small arterial injury can sometimes be addressed with fulguration under direct vision, but if this is not successful and bleeding does not cease with pressure, or in cases of significant arterial hemorrhage, then selective angio-embolization will likely be required (see later).
Delayed Hemorrhage
Postoperative hemorrhage can occur with the nephrostomy tube in place, at time of tube removal, or after discharge from the hospital. Approximately 1% of major percutaneous procedures are complicated by delayed hemorrhage requiring treatment (Kessaris et al, 1995; Martin et al, 2000; Richstone et al, 2008). In one large series, the incidence was greatest following percutaneous resection of upper tract urothelial carcinoma (3.2%) and least following percutaneous endopyelotomy (0.8%) (Richstone et al, 2008). Delayed hemorrhage is usually due to arteriovenous fistulas or arterial pseudoaneurysms, with the latter being more common. Arteriovenous fistulas occur when a paired set of artery and vein are injured, and arterial blood enters directly into vein (Fig. 47–41). The weak vein wall cannot sustain the high arterial pressure and ruptures. Bleeding is most commonly noted into the collecting system but can be outside the kidney as well. The latter should be suspected if the hematocrit falls but the urine remains relatively clear. It can be confirmed with CT or ultrasonography. An arterial pseudoaneurysm occurs when an artery is injured, clots off, and then intermittently ruptures, often clotting off again at variable intervals (Fig. 47–42A). Continuous bleeding suggests an arteriovenous fistula, and intermittent bleeding suggests arterial pseudoaneurysm, but the distinction is not critical because treatment is the same. In a large series of Kessaris and colleagues (1995), of the 0.8% of 2200 patients requiring treatment for delayed hemorrhage after percutaneous renal surgery, 24% of the hemorrhages occurred within 24 hours of surgery, 41% between 2 and 7 days after surgery, and 35% more than 7 days later. Any report of bright red blood in the urine after percutaneous renal surgery should prompt hospital admission and consideration of angiography, which is diagnostic in more than 90% of cases (Richstone et al, 2008). The standard treatment of renal arteriovenous fistulae and arterial pseudoaneurysms is selective angio-embolization, which is highly effective; among three series reporting 72 patients undergoing selective angio-embolization after percutaneous renal surgery, success was noted in 68 (94%) (Patterson et al, 1985; Martin et al, 2000; Richstone et al, 2008). Nephrectomy may be required if selective angio-embolization fails, and partial or total renal loss may occur if angio-embolization is not selective enough (Fig. 47–42B). Additionally, the embolization coils can migrate to unwanted locations, immediately or delayed.
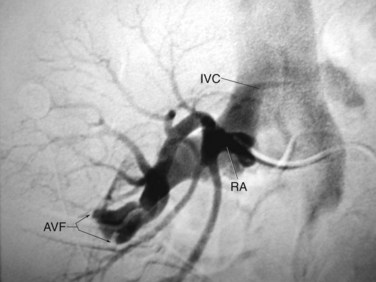
Figure 47–41 Angiography of the renal artery (RA) is followed promptly by contrast material appearing in the inferior vena cava (IVC), suggesting the presence of an arteriovenous fistula (AVF).
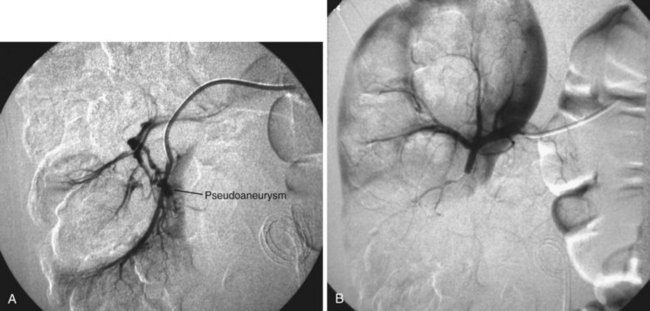
Figure 47–42 A, Angiography demonstrates a pseudoaneurysm in the lower pole of the right kidney. B, Angio-embolization was successful at occluding the pseudoaneurysm but also devascularized a large portion of the lower pole.
A recently introduced alternative to angio-embolization is endovascular placement of a covered stent to occlude the site of arterial injury (Sprouse and Hamilton, 2002; Areste et al, 2005). This treatment maintains patency of the feeding artery, thereby preserving renal parenchyma, and obviates the risk of migration of embolization coils (Fig. 47–43). Another alternative is ultrasonographically guided percutaneous puncture of an arterial pseudoaneurysm, with injection of thrombin or fibrin tissue adhesive (Benjaminov and Atri, 2002; Lagana et al, 2006).
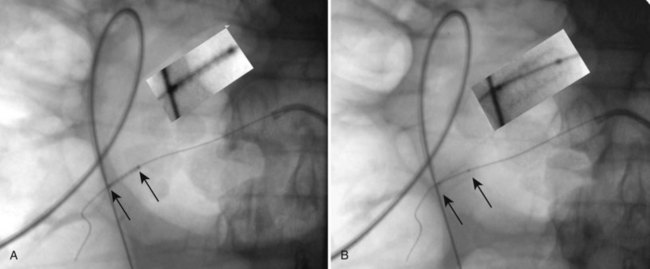
Figure 47–43 Covered arterial stent placed in branch of renal artery injured during endopyelotomy. A, Stent before balloon expansion. B, Stent after balloon expansion. Inset in each figure is a 2× enlargement of stent (indicated by arrows).
Untreated coagulopathy is an absolute contraindication to percutaneous renal surgery. Percutaneous access can be safely obtained if coagulopathy is reversed or anticoagulant medications are stopped. Anticoagulants can be restarted once the urine clears, preferably after nephrostomy tube removal, but the patient should be closely monitored.
Collecting System Injury
Tears in the infundibulum are not uncommon during percutaneous surgery of the upper urinary tract collecting system. They generally do not cause problems intraoperatively as long as there is no hemorrhage. Ureteral injuries are rare but can occur as a result of inflating the ureteral balloon occlusion catheter in the ureter or during other ureteral manipulation. These injuries generally will heal over a ureteral stent. Renal pelvic perforation can occur during initial access or during dilation. Pushing on a renal pelvic stone too hard during lithotripsy or misusing a lithotripter or resectoscope can also perforate the renal pelvis. Renal pelvic perforation is usually recognized intraoperatively (Fig. 47–44). Collapse of a previously distended renal pelvis is a usual sign if the perforation is not visualized directly at first. A perforation that has not been recognized intraoperatively might be heralded by postoperative abdominal distention, ileus, and/or fever. If noted intraoperatively, abort the procedure unless it is near completion, in which case the task can be completed at lower irrigation pressure if the patient is doing well clinically. At the completion of a case in which renal pelvic perforation has been noted, insert a nephroureteral stent or a nephrostomy tube plus an internal ureteral stent to optimize drainage and then wait 2 to 7 days before nephrostography and tube removal, depending on the severity of the injury. If renal pelvic perforation is detected postprocedure, despite adequate drainage of the collecting system, then placement of a percutaneous drain into the urinoma might be required. There have been reports of massive intra-abdominal collection of extravasated fluid after percutaneous renal surgery (Peterson et al, 1985; Pugach et al, 1999; Ghai et al, 2003).
Visceral Injury
Any abdominal organ close to the kidney can be injured during percutaneous renal surgery including the colon, duodenum, jejunum, spleen, liver, and biliary system.
Colon injury occurs during percutaneous renal surgery in the prone position at a rate of less than 1% (Segura et al, 1985; Lee et al, 1987; Gerspach et al, 1997; El-Nahas et al, 2006; Michel et al, 2007). As would be expected on the basis of the anatomy, with the apposition of the colon to the kidney being greatest on the left side and at the lower pole, the left colon is injured twice as often as the right colon and the majority of colon injuries involve access to the lower pole (El-Nahas et al, 2006). Additional risk factors include advanced patient age, dilated colon, prior colon surgery or disease, thin body habitus, and the presence of a horseshoe kidney (Goswami et al, 2001; El-Nahas et al, 2006; Michel et al, 2007; Korkes et al, 2009). Injury might be less likely with the patient in the supine position; one study reported a retrorenal colon on CT in 1.9% of patients in the supine position versus 10% in prone patients (Hopper et al, 1987). The clinical incidence of colon injury, however, is much lower than this figure would suggest.
Intraoperative detection of colonic injury confers easier management. If not determined intraoperatively, colon injury should be considered postoperatively if a patient develops unexplained fever, prolonged ileus, unexplained leukocytosis, rectal bleeding, evidence of peritoneal inflammation, or fecaluria or pneumouria. Clinically apparent nephrocolonic fistula may be the presenting sign, or the injury may not be noted until the time of postoperative nephrostogram. Most colon injuries are extraperitoneal and can be managed conservatively (Gerspach et al, 1997; El-Nahas et al, 2006; Korkes et al, 2009). The main principle of care is prompt and separate drainage of the colon and urinary collecting system. Back out the offending nephrostomy tube into the colon to serve as a colostomy tube, considering exchanging it for a larger tube to enhance colonic drainage, and obtain separate access to the upper urinary tract with either a new percutaneous access that does not traverse the colon or a retrograde-placed ureteral stent. Administer broad-spectrum antibiotics. Give nothing by mouth for a few days, and then start clear liquids. If there is no increase of colostomy output, then administer high-calorie protein supplementation and eventually a regular diet. Parenteral feeding is usually not required. Confirm lack of communication between the colon and collecting system with contrast injection of the tubes before removing them. If the injury is intraperitoneal, or if the patient develops peritonitis or sepsis, then open surgical repair may be required.
Duodenal and jejunal injuries are even less common than colonic injury, described in only a few cases reports (Culkin et al, 1985; Morris et al, 1991; Kumar et al, 1994; Ahmed and Reeve, 1995; Santiago et al, 1998; Lopes-Neto et al, 2000; Al-Assiri et al, 2005; Ricciardi et al, 2007). Detection is by clinical signs and symptoms of peritonitis or by noting a nephroenteric fistula during postoperative nephrostography. Although open surgery may be required, conservative management using percutaneous intraduodenal catheterization or simple nasogastric or nasoduodenal drainage, combined with drainage of the upper urinary tract, fasting, and parenteral feeding, has been successful.
Studies based on CT and MRI have suggested that splenic and hepatic injuries should be unlikely unless the kidney is accessed above the 10th rib, although access above the 11th or 12th rib might traverse these organs in rare cases (Hopper and Yakes, 1990; Robert et al, 1999). If splenomegaly or hepatomegaly is present, these relationships change and access guided by CT is recommended. Splenic or hepatic injuries to orthotopic and normal-sized organs occur almost exclusively with supracostal upper pole renal access. Splenic injury might require laparotomy and potentially splenectomy owing to hemorrhage (Kondas et al, 1994; Shah et al, 2007), but conservative management has been successful as well (Goldberg et al, 1989; Santiago et al, 1998; Carey et al, 2006; Schaeffer et al, 2008). Liver injury is less likely to be associated with significant hemorrhage—indeed the liver can be traversed percutaneously to obtain biliary access (Nadler et al, 2002), and purposeful transhepatic percutaneous renal surgery has been reported (Matlaga et al, 2006). As such, liver injuries during percutaneous renal surgery can be managed conservatively (El-Nahas et al, 2008).
There have been a few reported cases of biliary peritonitis due to injury of the gallbladder or biliary tree, which require laparotomy owing to the high mortality rate of bile peritonitis (Martin et al, 1996; Saxby, 1996; Kontothanassis and Bissas, 1997; Ricciardi et al, 2007). Postoperative pancreatitis, without evidence of direct pancreatic injury, has been described as well (Chitale et al, 2005).
Pleural Injury
Hydrothorax, and occasionally pneumothorax, is a risk of percutaneous access to the upper urinary tract collecting system. Access below the 12th rib rarely results in hydrothorax or pneumothorax (<0.5%) (Munver et al, 2001; Radecka et al, 2003; Lojanapiwat and Prasopsuk, 2006). The incidence of pleural complications with punctures above the 12th rib (the 11th intercostal space) is generally considered an acceptable risk if that approach provides optimal access to the upper urinary tract. Access above the 11th rib or higher carries a much greater risk of pleural injury. Among 16 reports that distinguished between access above the 12th rib and access above the 11th rib, reporting a total of 1384 supracostal percutaneous accesses, the incidence of hydro/pneumothorax that required intervention varied from 0% to 18% for access superior to the 12th rib (weighted mean of 4.6%) and from 0% to 100% for access superior to the 11th rib (weighted mean of 24.6%) (Young et al, 1985; Picus et al, 1986; Narasimham et al, 1991; Golijanin et al, 1998; Kekre et al, 2001; Munver et al, 2001; Gupta et al, 2002; Wong and Leveillee, 2002; Muzrakchi et al, 2003; Radecka et al, 2003; Aron et al, 2005c; Lojanapiwat and Prasopsuk, 2006; Yadav et al, 2006; Shaban et al, 2008; Sukumar et al, 2008; Yadav et al, 2008).
Nephropleural fistula (urinothorax) is a direct and persistent communication between the intrarenal collecting system and the intrathoracic cavity (Ray et al, 2003; Shleyfer et al, 2006; Handa et al, 2007). It can follow percutaneous renal access of the upper urinary tract in the setting of pleural transgression. Some degree of distal ureteral obstruction usually contributes to the problem. Most commonly, nephropleural fistula is diagnosed after the percutaneous tube is removed, but it can occur in the setting of a recognized hydrothorax when the persistent communication is documented on nephrostography at the time of intended tube removal. Lallas and colleagues (2004) reported that nephropleural fistula never occurred in association with subcostal access but did complicate 2.3% of punctures superior to the 12th rib and 6.3% of punctures superior to the 11th rib.
Pleural complications of supracostal percutaneous access can often be detected with chest fluoroscopy during or at the conclusion of the procedure. Fluid can be seen tracking along the lateral borders of the chest cavity and compressing the ipsilateral lung. Although postoperative chest radiography is more sensitive than intraoperative fluoroscopy, in the series by Ogan and colleagues (2003), thoracostomy was never required on the basis of postoperative chest radiography when intraoperative chest fluoroscopy was negative. Nonetheless, formal chest radiography is recommended following all cases of supracostal percutaneous renal access.
Thoracostomy is not necessary for all patients with hydrothorax. If hydrothorax is noted intraoperatively, then insert a small-caliber (8-Fr to 12-Fr) Cope nephrostomy tube as the thoracostomy (Fig. 47–45), using fluoroscopic guidance and the same general techniques as for antegrade percutaneous renal access (Ogan and Pearle, 2002). A Heimlich valve, rather than water seal drainage, is all that is necessary in the absence of lung injury. If a hydrothorax is noted on the postoperative chest radiograph, then place a small-caliber tube only if the effusion is large or there is evidence of respiratory compromise or hemodynamic instability. A large-bore thoracostomy tube as for lung injury is rarely required.
Metabolic and Physiologic Complications
Normal saline should be the irrigant for percutaneous renal surgery, with the exception of glycine or similar nonelectrolytic isotonic fluids when monopolar electrocautery is used. Irrigation with water during percutaneous renal surgery risks intravascular hemolysis, which can be fatal. At least one death associated with water irrigation during percutaneous nephrolithotomy has been described (Bennett et al, 1984), and the author is aware of an unreported case. Intravascular or extravascular extravasation of nonelectrolytic isotonic fluid from continued irrigation in the setting of a large venous injury or collecting system perforation, respectively, can result in hyponatremia and other electrolyte abnormalities, renal or hepatic dysfunction, and mental status changes. When normal saline is used in uncomplicated cases, the amount of fluid absorption is generally clinically insignificant (Kukreja et al, 2002; Koroglu et al, 2003), although in one study 28% of patients absorbed more than a liter (Malhotra et al, 2001). A large amount of saline extravasation can lead to clinically significant respiratory distress or cardiac failure due to volume overload.
Venous gas embolism is a rare but potentially fatal complication of percutaneous renal surgery. The gas (in this case, air) enters the venous system and passes through the right heart into the pulmonary circulation, blocking the output of the right heart, which results in hypoxemia, hypercapnia, and depressed cardiac output. Gas can also pass through a patent foramen ovale to enter the arterial system, which can result in neurologic deficits. Among six reported cases of venous gas embolism in association with percutaneous renal surgery, three were associated with air pyelography in combination with percutaneous access of the kidney (Miller et al, 1984; Cadeddu et al, 1997a; Droghetti et al, 2002), one involved percutaneous surgery without air pyelography (Turillazzi et al, 2009) and two occurred after retrograde injection of air into the renal pelvis but before percutaneous puncture (Varkarakis et al, 2003; Song et al, 2007). Venous gas embolism is indicated by hypoxemia, evidence of pulmonary edema, increased airway pressure, hypotension, jugular venous distention, facial plethora, dysrhythmias, and auscultation of a mill-wheel cardiac murmur and/or the appearance of a widened QRS complex with right heart strain patterns on electrocardiography. The most sensitive measure is a sudden decrease in capnometry reading of the P(end-tidal)CO2. Swift response is required and includes rapid ventilation with 100% oxygen, positioning the patient head down with the right side up, and general resuscitative maneuvers.
Postoperative Fever and Sepsis
After percutaneous nephrolithotomy, 15% to 30% of patients have a fever. Risk factors for fever after percutaneous nephrolithotomy include infectious stones, preoperative urinary tract infection, hydronephrosis, and indwelling ureteral stent or nephrostomy tube (Charton et al, 1986; Troxel and Low, 2002; Aghdas et al, 2006). Most patients with fever after percutaneous nephrolithotomy, assuming appropriate antimicrobial prophylaxis, do not have infection (Cadeddu et al, 1998). Sepsis occurs in 1% to 2% of patients after percutaneous nephrolithotomy (Dogan et al, 2007; Gonen et al, 2008b). Sepsis implies an infection, which is not always present; “systemic inflammatory response syndrome” is a more accurate description. Preoperative urine cultures should be treated. Even if bacteriologic cure is not possible (e.g., infectious stone, indwelling nephrostomy tube), bacterial counts should be suppressed as much as possible to reduce the risk of infectious complications. Nonetheless, a negative urine culture does not guarantee against sepsis because the voided urine culture may not reflect the intrarenal urine (Rao et al, 1991). Because the febrile patient who will progress to sepsis cannot be predicted, careful observation, appropriate diagnostic evaluation, and initiation of antimicrobial therapy and other supportive care is indicated if a postoperative fever does not resolve promptly.
If pus is aspirated upon initial percutaneous to the upper urinary tract, the safest measure is to abort the procedure and leave a nephrostomy tube for drainage. Aron and associates (2005a), on the basis of their experience in 19 patients with purulent fluid from the kidney at initial puncture for percutaneous nephrolithotomy, suggested that aborting the procedure might not always be necessary. They continued the procedure in 12 patients and delayed it in 7. Of the 12 patients in whom the procedure was continued, two (17%) had postprocedure sepsis. Among the seven patients in whom the procedure was delayed (for 3 to 7 days), two (29%) had sepsis after the second procedure (both of whom had additional portions of the kidney containing undrained pus that were discovered at the time of the delayed percutaneous nephrolithotomy). An important difference was in the quality of the aspirated material. One patient in whom percutaneous nephrolithotomy was continued and two in whom the procedure was delayed had “frank pus” as opposed to “purulent fluid” in the other cases. All three of these patients with frank pus were among the four who developed sepsis. The authors recommend that if frank pus is aspirated, then the procedure should be aborted. If the aspirate is a cloudy nonviscous liquid, it might be safe to proceed. This approach has not yet been validated in additional studies. This report also suggests that not all patients with “purulent fluid” are infected. Of the 19 patients, the culture of the aspirate revealed bacteria in only 6. This suggests that infection may have been sterilized by previous antibiotic usage. The turbid fluid may indicate a sterile inflammatory response to the stone, or the fluid may consist of debris related to the renal calculus. Another important point is that a single nephrostomy tube may not drain all areas of infection in the kidney, especially if there is intrarenal obstruction.
Neuromusculoskeletal Complications
Prone positioning for percutaneous renal surgery has the potential for a number of neuromusculoskeletal injuries (Shermak et al, 2006; Edgcombe et al, 2008). Excessive pressure on neural and vascular structures, whether directly from the operative table or indirectly through the positioning of limbs, may lead to short- or long-term disability. Most reported injuries associated with prone positioning are related to the head and neck region including ocular injury resulting in visual loss, facial nerve injury or necrosis over facial bones or the tip of the nose, and cerebrovascular accident due to carotid or vertebrobasilar artery dissection. Careful padding of the head, in a neutral and nonextended position, is important. Malpositioning of the extremities can lead to peripheral nerve injury (Winfree and Kline, 2005). The shoulder and elbow should not be abducted more than 90 degrees, so as to prevent brachial plexopathy, and generous padding at the elbow and forearm reduces the risk of nerve compression. Knees need to be padded. Ankles should be elevated to reduce pressure on the dorsum of the foot.
Venous Thromboembolism
The incidence of venous thromboembolism with percutaneous renal surgery is low (<3% in older series of percutaneous nephrolithotomy) (Segura et al, 1985; Lee et al, 1987), but there are no recent data. The American Urological Association’s Best Practice Statement for the prevention of deep vein thrombosis in patients undergoing urologic surgery does not include percutaneous renal surgery among procedures for which prophylaxis against venous thromboembolism is recommended (Forrest et al, 2009). Early ambulation is the best measure to reduce the already low risk of venous thromboembolism.
Tube Dislodgement
Whether the percutaneous nephrostomy tube is intended to be short or long term, inadvertent tube dislodgement risks poor patient outcomes. The tube does not need to fall out of the patient completely; especially in a patient with a large subcutaneous layer, the tube can remain attached at the skin but pull out from within the kidney when the distance between the skin and kidney increases with patient movement. Nonetheless, all tubes should be secured at the skin to reduce the risk of at least one mechanism of tube removal. Tubes vary in their inherent ability to resist removal. Malecot tubes are the easiest to pull out, and circle nephrostomy tubes are the hardest. The Cope retention mechanism is more secure than Malecot wings but does not retain as well as a balloon (Canales et al, 2005). For long-term indwelling, circle nephrostomy tubes and Cope nephrostomy tubes are most commonly used. Ironically, the Malecot tube is also the one most likely to become entrapped; tissue can grow over the wings, making removal difficult and traumatic (Sardina et al, 1995; Tasca and Cacciola, 2004).
If a nephrostomy tube has been in for only a short time, then complete dislodgement often leads to complete loss of percutaneous access. Using a nephrostomy tube with a ureteral extension, whether it is only partially down the ureter as in the Malecot re-entry tube or all the way into the bladder as in a nephroureteral stent, will increase the chance of having some access back into the kidney even if the renal retention device pulls out of the kidney. For tubes that have been in place more than a few weeks, the tract is usually mature enough that carefully probing with an angled hydrophilic wire and judicious use of contrast material to outline the tract can allow restoration of the percutaneous access. Tube malposition can usually be corrected under fluoroscopic control, but guidance by CT might be helpful as well (Jones and McGahan, 1999).
Collecting System Obstruction
Transient ureteral obstruction owing to ureteral edema or blood clot occurs commonly. To assess for this, nephrostomy tubes should be removed after nephrostography or after a period of clamping to assess clinically for distal ureteral obstruction. Much more uncommon is stricture formation, which can occur in the ureter, at the ureteropelvic junction, or in an infundibulum. If the stricture occurs early in the postprocedure course, then a nephrocutaneous fistula will develop. If it develops late, then hydronephrosis or hydrocalyx will occur. In one large series there was a 2% rate of infundibular stenosis after percutaneous nephrolithotomy (Parsons et al, 2002). The obstructions developed in the areas that had been accessed percutaneously. Predisposing factors in this and other smaller reports (Ballanger et al, 1987; Weir and Honey, 1999; Buchholz, 2001) include large stone burden requiring multiple or long procedures and prolonged nephrostomy tube drainage, previous open stone surgery, diabetes mellitus, and obesity. The ureter (Culkin et al, 1987; Lopes-Neto et al, 2008) and the ureteropelvic junction (Green et al, 1987; Ben Slama et al, 2005) are smaller in caliber than the intrarenal collecting system and are therefore also susceptible to trauma.
Obstruction after percutaneous renal surgery should respond to endoscopic treatment in most cases, but open surgical reconstruction or excision with partial nephrectomy or total nephrectomy may be required.
Loss of Renal Function
Despite the direct puncture of renal parenchyma and enlargement of sometimes multiple tracts to up to 34 Fr, the kidney suffers little permanent damage after uncomplicated percutaneous renal surgery. On the basis of several different techniques of study, a kidney loses negligible function after percutaneous renal surgery (Ekelund et al, 1986; Chen et al, 1992; Saxby, 1997; Kilic et al, 2006), and in one study there appeared to be less damage to the kidney after percutaneous nephrolithotomy than after shock wave lithotripsy (LeChevallier et al, 1993). In the setting of impaired function, especially due to obstructing calculi, percutaneous surgery will often improve renal function (Chandhoke et al, 1992; Chatham et al, 2002; Bilen et al, 2008). Percutaneous renal surgery also causes no significant change in function of solitary kidneys (Jones et al, 1991; Liou and Streem, 2001; Canes et al, 2009). In one study of patients surveyed a mean of 19 years after treatment, there was no difference between shock wave lithotripsy and percutaneous nephrolithotomy in the development of renal insufficiency or hypertension (Krambeck et al, 2008b).
When there is renal loss following percutaneous renal surgery, it usually owes to disastrous vascular injury or the angio-embolization used to treat hemorrhage. In the original American Urological Association guideline on staghorn calculi (Segura et al, 1994), renal loss after percutaneous nephrolithotomy was estimated at 1.6%; data were insufficient to calculate a new figure in the 2005 update of the guidelines (Preminger et al, 2005).
Death
Death after percutaneous renal surgery is extremely rare, and when it occurs it is usually due to underlying cardiovascular conditions. In the current American Urological Association guideline on management of staghorn calculi, the median death estimate for percutaneous nephrolithotomy was zero, which reflects the paucity of data on the subject (Preminger et al, 2005).
Key Points: Complications
Basiri A, Ziaee AM, Kianian HR, et al. Ultrasonographic versus fluoroscopic access for percutaneous nephrolithotomy: a randomized clinical trial. J Endourol. 2008;22:281-284.
Cadeddu JA, Chen R, Bishoff J, et al. Clinical significance of fever after percutaneous nephrolithotomy. Urology. 1998;52:48-50.
de la Rosette JJ, Tsakiris P, Ferrandino MN, et al. Beyond prone position in percutaneous nephrolithotomy: a comprehensive review. Eur Urol. 2008;54:1262-1269.
Desai MR, Kukreja R, Desai MM, et al. A prospective randomized comparison of type of nephrostomy drainage following percutaneous nephrostolithotomy: large bore versus small bore versus tubeless. J Urol. 2004;172:565-567.
El-Nahas AR, Shokeir AA, El-Assmy AM, et al. Colonic perforation during percutaneous nephrolithotomy: study of risk factors. Urology. 2006;67:937-941.
Gupta M, Bellman GC, Smith AD. Massive hemorrhage from renal vein injury during percutaneous renal surgery: endourological management. J Urology. 1997;157:795-797.
Miller NL, Matlaga BR, Lingeman JE. Techniques for fluoroscopic percutaneous renal access. J Urol. 2007;178:15-23.
Munver R, Delvecchio FC, Newman GE, et al. Critical analysis of supracostal access for percutaneous renal surgery. J Urol. 2001;166:1242-1246.
Netto NRJr, Ikonomidis J, Ikari O, et al. Comparative study of percutaneous access for staghorn calculi. Urology. 2005;65:659-662.
Ogan K, Corwin TS, Smith T, et al. Sensitivity of chest fluoroscopy compared with chest CT and chest radiography for diagnosing hydropneumothorax in association with percutaneous nephrostolithotomy. Urology. 2003;62:988-992.
Patel B, Mason BM, Hoenig DM. Retrograde endoscopic-assisted percutaneous renal access: a novel “lasso” technique to achieve rapid secure access to the collecting system. J Endourol. 2008;22:591-596.
Preminger GM, Assimos DG, Lingeman JE, et al. Chapter 1: AUA guideline on management of staghorn calculi: diagnosis and treatment recommendations. J Urol. 2005;173:1991-2000.
Richstone L, Reggio E, Ost MC, et al. Hemorrhage following percutaneous renal surgery: characterization of angiographic findings. J Endourol. 2008;22:1129-1135.
Sampaio FJ, Zanier JF, Aragao AH, et al. Intrarenal access: 3-dimensional anatomical study. J Urol. 1992;148:1769-1773.
Abou-Elela A, Emran A, Mohsen MA, et al. Safety and efficacy of tubeless percutaneous renal surgery. J Endourol. 2007;21:977-984.
ACR. Practice guideline for the performance of percutaneous nephrostomy. ACR practice guidelines and technical standards. American College of Radiology, Reston (VA), 2007, 705-715 http://www.acr.org/guidelines [accessed 08.05.09]
Aghamir SMK, Hosseini SR, Gooran S. Totally tubeless percutaneous nephrolithotomy. J Endourol. 2004;18:647-648.
Aghamir SMK, Khazaeli MH, Meisami A. Use of Surgicel for sealing nephrostomy tract after totally tubeless percutaneous nephrolithotomy. J Endourol. 2006;20:293-295.
Aghamir SMK, Mohammadi A, Mosavibahar SH, et al. Totally tubeless percutaneous nephrolitotomy in renal anomalies. J Endourol. 2008;22:2131-2134.
Aghdas FS, Akhavizadegan H, Aryanpoor A, et al. Fever after percutaneous nephrolithotomy: contributing factors. Surg Infect. 2006;7:367-371.
Agrawal MS, Agrawal M, Gupta A, et al. A randomized comparison of tubeless and standard percutaneous nephrolithotomy. J Endourol. 2008;22:439-442.
Ahmed M, Reeve R. Iatrogenic duodeno-cutaneous fistula at percutaneous nephrolithotomy managed conservatively. Br J Urol. 1995;75:416-418.
Al-Assiri M, Binsaleh S, Libman J, et al. Jejunal perforation during percutaneous nephrolithotrypsy. Sci World J. 2005;5:496-499.
Al-Ba’adani TH, Al-Kohlany KM, Al-Adimi A, et al. Tubeless percutaneous nephrolithotomy: the new gold standard. Int Urol Nephrol. 2008;40:603-608.
Al-Otaibi K, Hosking DH. Percutaneous stone removal in horseshoe kidneys. J Urol. 1999;162:674-677.
Alken P. The telescope dilators. World J Urol. 1985;3:7-10.
Allen D, O’Brien T, Tiptaft R, et al. Defining the learning curve for percutaneous nephrolithotomy. J Endourol. 2005;19:279-282.
Amjadi M, Zolfaghari A, Elahian A, et al. Percutaneous nephrolithotomy in patients with previous open nephrolithotomy: one-shot versus telescopic technique for tract dilatation. J Endourol. 2008;22:423-425.
Aquil S, Rana M, Zaidi Z. Laparoscopic assisted percutaneous nephrolithotomy (PCNL) in ectopic pelvic kidney. J Pakistan Med Assoc. 2006;56:381-383.
Areste N, Moyano MJ, Naranjo D, et al. [Endovascular treatment with “covered stent” of arteriovenous fistula secondary to percutaneous renal biopsy]. Nefrologia. 2005;25:449-450.
Aron M, Goel R, Kesarwani PK, et al. Hemostasis in tubeless PNL: point of technique. Urol Int. 2004;73:244-247.
Aron M, Goel R, Gupta NP, et al. Incidental detection of purulent fluid in kidney at percutaneous nephrolithotomy for branched renal calculi. J Endourol. 2005;19:136-139.
Aron M, Gupta NP, Goel R, et al. Laparoscopy-assisted percutaneous nephrolithotomy (PCNL) in a previously operated ectopic pelvic kidney. Surg Laparosc Endosc Percutan Tech. 2005;15:41-43.
Aron M, Yadav R, Goel R, et al. Multi-tract percutaneous nephrolithotomy for large complete staghorn calculi. Urol Int. 2005;75:327-332.
Atmaca AF, Akbulut Z, Altinova S, et al. Standard percutaneous nephrolithotomy on a pelvic kidney without any ancillary modalities. Int Urol Nephrol. 2007;39:723-725.
Baldwin DD, Maynes LJ, Desai PJ, et al. A novel single step percutaneous access sheath: the initial human experience. J Urol. 2006;175:156-161.
Ballanger P, Baron JC, Teillac P, et al. [Acquired stenosis of the upper urinary tract after percutaneous nephrolithotomy]. Ann Urol. 1987;21:296-299.
Barbaric ZL, Hall T, Cochran ST, et al. Percutaneous nephrostomy: placement under CT and fluoroscopy guidance. AJR. 1997;169:151-155.
Basiri A, Mehrabi S, Kianian H, et al. Blind puncture in comparison with fluoroscopic guidance in percutaneous nephrolithotomy: a randomized controlled trial. Urol J. 2007;4:79-83.
Basiri A, Ziaee AM, Kianian HR, et al. Ultrasonographic versus fluoroscopic access for percutaneous nephrolithotomy: a randomized clinical trial. J Endourol. 2008;22:281-284.
Basiri A, Ziaee SA, Nasseh H, et al. Totally ultrasonography-guided percutaneous nephrolithotomy in the flank position. J Endourol. 2008;22:1453-1457.
Bellman GC, Davidoff R, Candela J, et al. Tubeless percutaneous renal surgery. J Urol. 1997;157:1578-1582.
Ben Slama MR, Zaafrani R, Ben Mouelli S, et al. [Ureterocalicostomy: last resort in the treatment of certain forms of ureteropelvic junction stenosis. Report of 5 cases]. Progres Urol. 2005;15:646-649.
Benjaminov O, Atri M. Percutaneous thrombin injection for treatment of an intrarenal pseudoaneurysm. AJR. 2002;178:364-366.
Bennett MJ, Smith RW, Fuchs E. Sudden cardiac arrest during percutaneous ultrasonic nephrostolithotomy. Anesthesiology. 1984;60:245-246.
Benway BM, Nakada SY. Balloon dilation of nephrostomy tracts. J Endourol. 2008;22:1875-1876.
Berkman DS, Lee MW, Landman J, et al. Tubeless percutaneous nephrolithotomy (PCNL) with reversed Polaris Loop stent: reduced postoperative pain and narcotic use. J Endourol. 2008;22:2245-2249.
Bilen CY, Inci K, Kocak B, et al. Impact of percutaneous nephrolithotomy on estimated glomerular filtration rate in patients with chronic kidney disease. J Endourol. 2008;22:895-900.
Bird VG, Fallon B, Winfield HN. Practice patterns in the treatment of large renal stones. J Endourol. 2003;17:355-363.
Bloom DA, Morgan RJ, Scardino PL. Thomas Hillier and percutaneous nephrostomy. Urology. 1989;33:346-350.
Boon JM, Shinners B, Meiring JH. Variations of the position of the colon as applied to percutaneous nephrostomy. Surg Radiol Anat. 23, 2001. 421–405
Borin JF, Sala LG, Eichel L, et al. Tubeless percutaneous nephrolithotomy using hemostatic gelatin matrix. J Endourol. 2005;19:614-617.
Bove P, Stoianovici D, Micali S, et al. Is telesurgery a new reality? Our experience with laparoscopic and percutaneous procedures. J Endourol. 2003;17:137-142.
Bruyère F, Leroux C, Brunereau L, et al. Rapid prototyping model for percutaneous nephrolithotomy training. J Endourol. 2008;22:91-96.
Buchholz NP. Three-dimensional CT scan stone reconstruction for the planning of percutaneous surgery in a morbidly obese patient. Urol Int. 2000;65:46-48.
Buchholz NP. Double infundibular obliteration with abscess formation after percutaneous nephrolithotomy. Urol Int. 2001;66:46-48.
Cadeddu JA, Arrindell D, Moore RG. Near fatal air embolism during percutaneous nephrostomy placement. J Urol. 159, 1997.
Cadeddu JA, Bzostek A, Schreiner S, et al. A robotic system for percutaneous renal access. J Urol. 1997;158:1589-1593.
Cadeddu JA, Chen R, Bishoff J, et al. Clinical significance of fever after percutaneous nephrolithotomy. Urology. 1998;52:48-50.
Canales BK, Hendlin K, Braasch M, et al. Percutaneous nephrostomy catheters: drainage flow and retention strength. Urology. 2005;66:261-265.
Canes D, Hegarty NJ, Kamoi K, et al. Functional outcomes following percutaneous surgery in the solitary kidney. J Urology. 2009;181:154-160.
Carey RI, Siddiq FM, Guerra J, et al. Conservative management of a splenic injury related to percutaneous nephrostolithotomy. J Soc Laparoendoscopic Surg. 2006;10:504-506.
Castañeda-Zúñiga WR, Smith AD, et al. Single-step dilation of nephrostomy tract. J Urol. 1982;127:341.
Challacombe B, Dasgupta P, Tiptaft R, et al. Multimodal management of urolithiasis in renal transplantation. BJU Int. 2005;96:385-389.
Challacombe B, Patriciu A, Glass J, et al. A randomized controlled trial of human versus robotic and telerobotic access to the kidney as the first step in percutaneous nephrolithotomy. Comput Aided Surg. 2005;10:165-171.
Chandhoke PS, Albala DM, Clayman RV. Long-term comparison of renal function in patients with solitary kidneys and/or moderate renal insufficiency undergoing extracorporeal shock wave lithotripsy or percutaneous nephrolithotomy. J Urol. 1992;147:1226-1230.
Charton M, Vallancien G, Veillon B, et al. Urinary tract infection in percutaneous surgery for renal calculi. J Urol. 1986;135:15-17.
Chatham JR, Dykes TE, Kennon WG, et al. Effect of percutaneous nephrolithotomy on differential renal function as measured by mercaptoacetyl triglycine nuclear renography. Urology. 2002;59:522-525.
Chen KK, Chen MT, Yeh SH, et al. Radionuclide renal function study in various surgical treatments of upper urinary stones. Chung Hua i Hsueh Tsa Chih Chin Med J. 1992;49:319-327.
Chew BH, Chiu RYW, Hamidizadeh R, et al. Success and complication rates in 729 percutaneous nephrolithotomies at a single centre (abstract #1381). J Urol. 2009;181(Suppl):493-494.
Chien GW, Bellman GC. Blind percutaneous renal access. J Urol. 2002;16:93-96.
Chitale S, Mbakada R, Burgess N. Pancreatitis following percutaneous nephrolithotomy. Scand J Urol Nephrol. 2005;39:251-253.
Choi M, Brusky J, Weaver J, et al. Randomized trial comparing modified tubeless percutaneous nephrolithotomy with tailed stent with percutaneous nephrostomy with small-bore tube. J Endourol. 2006;20:766-770.
Crook TJ, Lockyer CR, Keoghane SR, et al. A randomized controlled trial of nephrostomy placement versus tubeless percutaneous nephrolithotomy. J Urol. 2008;180:612-614.
Crook TJ, Lockyer CR, Keoghane SR, et al. Totally tubeless percutaneous nephrolithotomy. J Endourol. 2008;22:267-271.
Culkin DJ, Wheeler JSJr, Canning JR. Nephro-duodenal fistula: a complication of percutaneous nephrolithotomy. J Urol. 1985;134:528-530.
Culkin DJ, Wheeler JSJ, Canning JR. Distal ureteral obstruction complicating percutaneous nephrolithotomy. Urology. 1987;30:364-368.
Darabi Mahboub MR, Zolfaghari M, Ahanian A. Percutaneous nephrolithotomy of kidney calculi in horseshoe kidney. Urol J. 2007;4:147-150.
Darenkov AF, Derevianko II, Martov AG, et al. [The prevention of infectious-inflammatory complications in the postoperative period in percutaneous surgical interventions in patients with urolithiasis]. Urologiia i Nefrologiia. 1994;2:24-26.
Davidoff R, Bellman GC. Influence of technique of percutaneous tract creation on incidence of renal hemorrhage. J Urol. 1997;157:1229-1231.
Davis BE, Noble MJ, Mebust WK. Use of the Collings knife electrode for percutaneous access in difficult endourology cases. J Urol. 1991;145:257-261.
de la Rosette JJ, Laguna MP, Rassweiler JJ, et al. Training in percutaneous nephrolithotomy-a critical review. Eur Urol. 2008;54:994-1001.
de la Rosette JJ, Tsakiris P, Ferrandino MN, et al. Beyond prone position in percutaneous nephrolithotomy: a comprehensive review. Eur Urol. 2008;54:1262-1269.
Desai MR, Jasani A. Percutaneous nephrolithotripsy in ectopic kidneys. J Endourol. 2000;14:289-292.
Desai MR, Kukreja R, Desai MM, et al. A prospective randomized comparison of type of nephrostomy drainage following percutaneous nephrostolithotomy: large bore versus small bore versus tubeless. J Urol. 2004;172:565-567.
Dogan HS, Guliyev F, Cetinkaya YS, et al. Importance of microbiological evaluation in management of infectious complications following percutaneous nephrolithotomy. Int Urol Nephrol. 2007;39:737-742.
Droghetti L, Giganti M, Memmo A, et al. Air embolism: diagnosis with single-photon emission tomography and successful hyperbaric oxygen therapy. Br J Anaesth. 2002;89:775-778.
Edgcombe H, Carter K, Yarrow S. Anaesthesia in the prone position. Br J Anaesth. 2008;100:165-183.
Egilmez H, Oztoprak I, Atalar M, et al. The place of computed tomography as a guidance modality in percutaneous nephrostomy: analysis of a 10-year single-center experience. Acta Radiol. 2007;48:806-813.
Ekelund L, Lindstedt E, Lundquist SB, et al. Studies on renal damage from percutaneous nephrolitholapaxy. J Urol. 1986;135:682-685.
El-Assmy AM, Shokeir AA, Mohsen T, et al. Renal access by urologist or radiologist for percutaneous nephrolithotomy-is it still an issue? J Urol. 2007;187:916-920.
El-Kappany HA, El-Nahas AR, Shoma AM, et al. Combination of laparoscopy and nephroscopy for treatment of stones in pelvic ectopic kidneys. J Endourol. 2007;21:1131-1136.
El-Nahas AR, Shokeir AA, El-Assmy AM, et al. Colonic perforation during percutaneous nephrolithotomy: study of risk factors. Urology. 2006;67:937-941.
El-Nahas AR, Mansour AM, Ellaithy R, et al. Case report: conservative treatment of liver injury during percutaneous nephrolithotomy. J Endourol. 2008;22:1649-1652.
Falahatkar S, Moghaddam AA, Salehi M, et al. Complete supine percutaneous nephrolithotripsy comparison with the prone standard technique. J Endourol. 2008;22:2513-2517.
Falahatkar S, Neiroomand H, Akbarpour M, et al. One-shot versus metal telescopic dilation technique for tract creation in percutaneous nephrolithotomy: comparison of safety and efficacy. J Endourol. 2009;23:615-618.
Feng MI, Tamaddon K, Mikhai lA, et al. Prospective randomized study of various techniques of percutaneous nephrolithotomy. Urology. 2001;58:345-350.
Fernström I, Johansson B. Percutaneous pyelolithotomy: a new extraction technique. Scand J Urol Nephrol. 1976;10:257-259.
Finelli A, Honey RJDA. Thoracoscopy-assisted high intercostal percutaneous renal access. J Endourol. 2001;15:581-585.
Forrest JB, Clemens JQ, Finamore P, et al. AUA Best Practice Statement for the prevention of deep vein thrombosis in patients undergoing urologic surgery. J Urol. 2009;181:1170-1177.
Francesca F, Felipetto R, Mosca F, et al. Percutaneous nephrolithotomy of transplanted kidney. J Endourol. 2002;16:225-227.
Frattini A, Barbieri A, Salsi P, et al. One shot: A novel method to dilate the nephrostomy access for percutaneous lithotripsy. J Endourol. 2001;15:919-923.
Gerspach JM, Bellman GC, Stoller ML, et al. Conservative management of colon injury following percutaneous renal surgery. Urology. 1997;49:831-836.
Ghai B, Dureja GP, Arvind P. Massive intraabdominal extravasation of fluid: a life threatening complication following percutaneous nephrolithotomy. Int Urol Nephrol. 2003;35:315-318.
Ghani KR, Pilcher J, Patel U, et al. Three-dimensional ultrasound reconstruction of the pelvicaliceal system: an in-vitro study. World J Urol. 2008;26:493-498.
Goel R, Yadav R, Gupta NP, et al. Laparoscopic assisted percutaneous nephrolithotomy (PCNL) in ectopic kidneys: two different techniques. Int Urol Nephrol. 2006;38:75-78.
Gofrit ON, Shapiro A, Donchin Y, et al. Lateral decubitus position for percutaneous nephrolithotripsy in the morbidly obese or kyphotic patient. J Endourol. 2002;16:383-386.
Goh M, Wolf JSJr. Almost totally tubeless percutaneous nephrostolithotomy: further evolution of the technique. J Endourol. 1999;13:177-180.
Goharderakhshan RZ, Schwartz BF, Rudnick DM, et al. Radially expanding single-step nephrostomy tract dilator. Urology. 2001;58:693-696.
Goldberg SD, Gray RR, St Louis EL, et al. Nonoperative management of complications of percutaneous renal nephrostomy. Can J Surg. 1989;32:192-195.
Golijanin D, Katz R, Verstandig A, et al. The supracostal percutaneous nephrostomy for treatment of staghorn and complex kidney stones. J Endourol. 1998;12:403-405.
Gonen M, Istanbulluoglu OM, Cicek T, et al. Balloon dilatation versus Amplatz dilatation for nephrostomy tract dilatation. J Endourol. 2008;22:901-904.
Gonen M, Turan H, Ozturk B, et al. Factors affecting fever following percutaneous nephrolithotomy: a prospective clinical study. J Endourol. 2008;22:2135-2138.
Gonen M, Ozturk B, Ozkardes H. Double-J stenting compared with one night externalized ureteral catheter placement in tubeless percutaneous nephrolithotomy. J Endourol. 2009;23:27-31.
Goodwin WE, Casey WC, Woolf W. Percutaneous trocar (needle) nephrostomy in hydronephrosis. JAMA. 1955;157:891-894.
Goswami AK, Shrivastava P, Mukherjee A, et al. Management of colonic perforation during percutaneous nephrolithotomy in horseshoe kidney. J Endourol. 2001;15:989-991.
Grasso M, Lang G, Taylor FC. Flexible ureteroscopically assisted percutaneous renal access. Tech Urol. 1995;1:39-43.
Green DF, Lytton B, Glickman M. Ureteropelvic junction obstruction after percutaneous nephrolithotripsy. J Urol. 1987;138:599-602.
Gupta M, Bellman GC, Smith AD. Massive hemorrhage from renal vein injury during percutaneous renal surgery: endourological management. J Urol. 1997;157:795-797.
Gupta NP, Mishra S, Seth A, et al. Percutaneous nephrolithotomy in abnormal kidneys: single-center experience. Urology. 2009;73:710-714.
Gupta R, Kumar A, Kapoor R, et al. Prospective evaluation of safety and efficacy of the supracostal approach for percutaneous nephrolithotomy. BJU Int. 2002;90:809-813.
Gupta V, Sadasukhi TC, Sharma KK, et al. Tubeless and stentless percutaneous nephrolithotomy. BJU Int. 2005;95:905-906.
Hacker A, Wendt-Nordahl G, Honeck P, et al. A biological model to teach percutaneous nephrolithotomy technique with ultrasound- and fluoroscopy-guided access. J Endourol. 2007;21:545-550.
Hagspiel KD, Kandarpa K, Silverman SG. Interactive MR-guided percutaneous nephrostomy. J Magn Reson Imaging. 1998;8:1319-1322.
Hammond L, Ketchum J, Schwartz BF. A new approach to urology training: a laboratory model for percutaneous nephrolithotomy. J Urol. 2004;172:1950-1952.
Handa A, Agarwal R, Aggarwal AN. Urinothorax: an unusual cause of pleural effusion. Singapore Med J. 2007;48:e289-e292.
Hatada T, Kusunoki M, Sakiyama T, et al. Hemodynamics in the prone jackknife position during surgery. Am J Surg. 1991;162:55-58.
He Z, Li X, Chen L, et al. Minimally invasive percutaneous nephrolithotomy for upper urinary tract calculi in transplanted kidneys. BJU Int. 2007;99:1467-1471.
Hegarty NJ, Desai MM. Percutaneous nephrolithotomy requiring multiple tracts: comparison of morbidity with single-tract procedures. J Endourol. 2006;20:753-760.
Heggagi MA, Karsza A, Szüle EJr. Use of different types of dilator systems in the prevention of complications of percutaneous (PC) renal surgery. Acta Chir Hung. 1991;32:365-369.
Hernandez DJ, Sinkov VA, Roberts WW, et al. Measurement of bio-impedance with a smart needle to confirm percutaneous kidney access. J Urol. 2001;166:1520-1523.
Hopper KD, Sherman JL, Luethke JM, et al. The retrorenal colon in the supine and prone patient. Radiology. 1987;162:443-446.
Hopper KD, Yakes WE. The posterior intercostal approach for percutaneous renal procedures: risk of puncturing the lung, spleen, and liver as determined by CT. AJR. 1990;154:115-117.
Hubert J, Blum A, Cormier L, et al. Three-dimensional CT-scan reconstruction of renal calculi: a new tool for mapping-out staghorn calculi and follow-up of radiolucent stones. Eur Urol. 1997;31:297-301.
Istanbulluoglu MO, Ozturk B, Gonen M, et al. Effectiveness of totally tubeless percutaneous nephrolithotomy in selected patients: a prospective randomized study. Int Urol Nephrol. 2009;41:541-545.
Janetschek G, Kunzel KH. Percutaneous nephrolithotomy in horseshoe kidneys: applied anatomy and clinical experience. Br J Urol. 1988;62:117-122.
Joel AB, Rubenstein JN, Hsieh MH, et al. Failed percutaneous balloon dilation for renal access: incidence and risk factors. Urology. 2005;66:29-32.
John BS, Rowland D, Patel U, et al. Evaluation of the accuracy of 3-dimensional ultrasonography of the kidney using an in vitro renal model. J Ultrasound Med. 2009;28:155-162.
Jones CD, McGahan JP. Computed tomographic evaluation and guided correction of malpositioned nephrostomy catheters. Abdom Imaging. 1999;24:422-425.
Jones DJ, Kellett MJ, Wickham JE. Percutaneous nephrolithotomy and the solitary kidney. J Urol. 1991;145:477-479.
Joshi HB, Adams S, Obadeyi OO, et al. Nephrostomy tube or “JJ” ureteric stent in ureteric obstruction: assessment of patient perspectives using quality-of-life survey and utility analysis. Eur Urol. 2001;39:695-701.
Jou YC, Cheng MC, Sheen JH, et al. Electrocauterization of bleeding points for percutaneous nephrolithotomy. Urology. 2004;64:443-446.
Karami H, Gholamrezaie HR. Totally tubeless percutaneous nephrolithotomy in selected patients. J Endourol. 2004;18:475-476.
Karami H, Jabbari M, Arbab AH. Tubeless percutaneous nephrolithotomy: 5 years of experience in 201 patients. J Endourol. 2007;21:1411-1413.
Karami H, Arbab AH, Rezaei A, et al. Percutaneous nephrolithotomy with ultrasonography-guided renal access in the lateral decubitus flank position. J Endourol. 2009;23:33-35.
Karlin GS, Smith AD. Approaches to the superior calix: renal displacement technique and review of options. J Urol. 1989;142:774-777.
Kaye KW, Clayman RV. Tamponade nephrostomy catheter for percutaneous nephrostolithotomy. Urology. 1986;27:441-445.
Kekre NS, Gopalakrishnan GG, Gupta GG, et al. Supracostal approach in percutaneous nephrolithotomy: experience with 102 cases. J Endourol. 2001;15:789-791.
Kendoff D, Citak M, Gardner MJ, et al. Intraoperative 3D imaging: value and consequences in 248 cases. J Trauma Inj Infect Crit Care. 2009;66:232-238.
Kerbl K, Clayman RV, Chandhoke PS, et al. Percutaneous stone removal with the patient in a flank position. J Urol. 1994;151:686-688.
Kessaris DN, Bellman GC, Pardalidis NP, et al. Management of hemorrhage after percutaneous renal surgery. J Urol. 1995;153:604-608.
Kidd CR, Conlin MJ. Ureteroscopically assisted percutaneous renal access. Urology. 2003;61:1244-1245.
Kilic S, Altinok T, Altunoluk B, et al. Long-term effects of percutaneous nephrolithotomy on renal morphology and arterial vascular resistance as evaluated by color Doppler ultrasonography: preliminary report. Urol Res. 2006;34:178-183.
Kim HC, Yang DM, Lee SH, et al. Usefulness of renal volume measurements obtained by a 3-dimensional sonographic transducer with matrix electronic arrays. J Ultrasound Med. 2008;27:1673-1681.
Kim SC, Tinmouth WW, Kuo RL, et al. Using and choosing a nephrostomy tube after percutaneous nephrolithotomy for large or complex stone disease: a treatment strategy. J Endourol. 2005;19:348-352.
Kim SC, Ng JC, Matlaga BR, et al. Use of lower pole nephrostomy drainage following endorenal surgery through an upper pole access. J Urol. 2006;175:580-584.
Klingler HC, Kramer G, Lodde M, et al. Urolithiasis in allograft kidneys. Urology. 2002;59:344-348.
Knudsen BE, Matsumoto ED, Chew BH, et al. A randomized, controlled, prospective study validating the acquisition of percutaneous renal collecting system access skills using a computer based hybrid virtual reality surgical simulator: phase I. J Urol. 2006;176:2173-2178.
Kondas J, Szentgyorgyi E, Vaczi L, et al. Splenic injury: a rare complication of percutaneous nephrolithotomy. Int Urol Nephrol. 1994;26:399-404.
Kontothanassis D, Bissas A. Biliary peritonitis complicating percutaneous nephrostomy. Int Urol Nephrol. 1997;29:529-531.
Korkes F, Neto ACL, Lucio JII, et al. Management of colon injury after percutaneous renal surgery. J Endourol. 2009;23:569-573.
Koroglu A, Togal T, Cicek M, et al. The effects of irrigation fluid volume and irrigation time on fluid electrolyte balance and hemodynamics in percutaneous nephrolithotripsy. Int Urol Nephrol. 2003;35:1-6.
Krambeck AE, LeRoy AJ, Patterson DE, et al. Percutaneous nephrolithotomy success in the transplant kidney. J Urol. 180, 2008. 2545–2459
Krambeck AE, LeRoy AJ, Patterson DE, et al. Long-term outcomes of percutaneous nephrolithotomy compared to shock wave lithotripsy and conservative management. J Urol. 2008;179:2233-2237.
Kukreja R, Desai M, Patel S, et al. Factors affecting blood loss during percutaneous nephrolithotomy: prospective study. J Endourol. 2004;18:715-722.
Kukreja RA, Desai MR, Sabnis RB, et al. Fluid absorption during percutaneous nephrolithotomy: does it matter? J Endourol. 2002;16:221-224.
Kumar A, Banerjee GK, Tewari A, et al. Isolated duodenal injury during relook percutaneous nephrolithotomy. Br J Urol. 1994;74:382-383.
Kumar V, Keeley FXJr. Percutaneous nephrolithotomy: why do we use rigid dilators? J Endourol. 2008;22:1877-1879.
Lagana D, Carrafiello G, Mangini M, et al. Multimodal approach to endovascular treatment of visceral artery aneurysms and pseudoaneurysms. Eur J Radiol. 2006;59:104-111.
Laguna MP, Hatzinger M, Rassweiler J. Simulators and endourological training. Curr Opin Urol. 2002;12:209-215.
Lallas CD, Delvecchio FC, Evans BR, et al. Management of nephropleural fistula after supracostal percutaneous nephrolithotomy. Urology. 2004;64:241-245.
Landman J, Venkatesh R, Lee DI, et al. Combined percutaneous and retrograde approach to staghorn calculi with application of the ureteral access sheath to facilitate percutaneous nephrolithotomy. J Urol. 2003;169:64-67.
LeChevallier E, Siles S, Ortega JC, et al. Comparison by SPECT of renal scars after extracoporeal shock wave lithotripsy and percutaneous nephrolithotomy. J Endourol. 1993;7:465-467.
Lee CL, Anderson JK, Monga M. Residency training in percutaneous renal access: does it affect urological practice? J Urol. 2004;171:592-595.
Lee DI, Uribe C, Eichel L, et al. Sealing percutaneous nephrolithotomy tracts with gelatin matrix hemostatic sealant: initial clinical use. J Urol. 2004;171:575-578.
Lee WJ, Smith AD, Cubelli V, et al. Complications of percutaneous nephrolithotomy. AJR. 1987;148:177-180.
LeMaitre L, Mestdagh P, Marecaux-Delomez J, et al. Percutaneous nephrostomy: placement under laser guidance and real-time CT fluoroscopy. Eur Radiol. 2000;10:892-895.
Liatsikos EN, Hom D, Dinlenc CZ, et al. Tail stent versus re-entry tube: a randomized comparison after percutaneous stone extraction. Urology. 2002;59:15-19.
Liatsikos EN, Kapoor R, Lee B, et al. “Angular percutaneous renal access”. Multiple tracts through a single incision for staghorn calculous treatment in a single session. Eur Urol. 2005;48:832-837.
Liou LS, Streem SB. Long-term renal functional effects of shock wave lithotripsy, percutaneous nephrolithotomy and combination therapy: a comparative study of patients with solitary kidney. J Urol. 2001;166:33-37.
Lojanapiwat B, Soonthornphan S, Wudhikarn S. Tubeless percutaneous nephrolithotomy in selected patients. J Endourol. 2001;15:711-713.
Lojanapiwat B. Percutaneous nephrolithotomy (PCNL) in kidneys with fusion and rotation anomalies. J Med Assoc Thailand. 2005;88:1426-1429.
Lojanapiwat B, Prasopsuk S. Upper-pole access for percutaneous nephrolithotomy: comparison of supracostal and infracostal approaches. J Endourol. 2006;20:491-494.
Lopes-Neto AC, de Mattos MH, da Silva MN, et al. Extensive ureteral stricture after percutaneous nephrolithotomy. Archivos Espanoles de Urologia. 2008;61:559-561.
Lopes-Neto AC, Tobias-Machado M, Juliano RV, et al. Duodenal damage complicating percutaneous access to kidney. Sao Paulo Med J. 2000;118:116-117.
Maheshwari PN, Andankar MG, Bansal M. Nephrostomy tube after percutaneous nephrolithotomy: large bore or pigtail catheter? J Endourol. 2000;14:735-737.
Maheshwari PN, Bhandarkar DS, Andankar MG, et al. Laparoscopically guided transperitoneal percutaneous nephrolithotomy for calculi in pelvic ectopic kidneys. Surg Endosc. 2004;18:1151.
Maheshwari PN, Bhandarkar DS, Shah RS, et al. Laparoscopy-assisted transperitoneal percutaneous nephrolithotomy for recurrent calculus in isthmic calix of horseshoe kidney. J Endourol. 2004;18:858-861.
Majidpour HS, Yousefinejad V. Percutaneous management of urinary calculi in horseshoe kidneys. Urology J. 2008;5:188-191.
Malhotra SK, Khaitan A, Goswami AK, et al. Monitoring of irrigation fluid absorption during percutaneous nephrolithotripsy: the use of 1% ethanol as a marker. Anaesthesia. 2001;56:1103-1106.
Mandhani A, Goyal R, Vijjan V, et al. Tubeless percutaneous nephrolithotomy-should a stent be an integral part? J Urol. 2007;178:921-924.
Marcovich R, Jacobson AI, Singh J, et al. No panacea for drainage after percutaneous nephrolithotomy. J Endourol. 2004;18:743-747.
Margulis V, Matsumoto ED, Knudsen BE, et al. Percutaneous renal collecting system access: Can virtual reality shorten the learning curve? J Urol. 2005;173(Suppl):315.
Martin E, Lujan M, Paez A, et al. Puncture of the gall bladder: an unusual cause of peritonitis complicating percutaneous nephrostomy. Br J Urol. 1996;77:464-465.
Martin X, Tajra LC, Gelet A, et al. Complete staghorn stones: percutaneous approach using one or multiple percutaneous accesses. J Endourol. 1999;13:367-368.
Martin X, Murat F-J, Feitosa LC, et al. Severe bleeding after nephrolithotomy: results of hyperselective embolization. Eur Urol. 2000;37:136-139.
Matlaga BR, Shah OD, Zagoria RJ, et al. Computerized tomography guided access for percutaneous nephrostolithotomy. J Urol. 2003;170:45-47.
Matlaga BR, Kim SC, Watkins SL, et al. Percutaneous nephrolithotomy for ectopic kidneys: over, around, or through. Urology. 2006;67:513-517.
Maynes LJ, Desai PJ, Zuppan CW, et al. Comparison of a novel one-step percutaneous nephrolithotomy sheath with a standard two-step device. Urology. 2008;71:223-237.
Merkle EM, Hashim M, Wendt M, et al. MR-guided percutaneous nephrostomy of the nondilated upper urinary tract in a porcine model. AJR. 1999;172:1221-1225.
Michel MS, Trojan L, Rassweiler JJ. Complications in percutaneous nephrolithotomy. Eur Urol. 2007;51:899-906.
Mikhail AA, Kaptein JS, Bellman GC. Use of fibrin glue in percutaneous nephrolithotomy. Urology. 2003;61:910-914.
Miller NL, Matlaga BR, Lingeman JE. Techniques for fluoroscopic percutaneous renal access. J Urol. 2007;178:15-23.
Miller NL, Matlaga BR, Handa SE, et al. The presence of horseshoe kidney does not affect the outcome of percutaneous nephrolithotomy. J Endourol. 2008;22:1219-1225.
Miller RA, Kellett MJ, Wickham JE. Air embolism, a new complication of percutaneous nephrolithotomy. What are the implications? J Urol. 1984;90:337-339.
Mokulis JA, Peretsman SJ. Retrograde percutaneous nephrolithotomy using the Lawson technique for management of complex nephrolithiasis. J Endourol. 1997;11:125-130.
Morris DB, Siegelbaum MH, Pollack HM, et al. Renoduodenal fistula in a patient with chronic nephrostomy drainage: a case report. J Urol. 1991;146:835-837.
Mosavi-Bahar SH, Amirzargar MA, Rahnavardi M, et al. Percutaneous nephrolithotomy in patients with kidney malformations. J Endourol. 2007;21:520-524.
Mostafa SA, Abbaszadeh S, Taheri S, et al. Percutaneous nephrostomy for treatment of posttransplant ureteral obstructions. Urol J. 2008;5:79-83.
Mouracade P, Spie R, Lang H, et al. Tubeless percutaneous nephrolithotomy: what about replacing the Double-J stent with a ureteral catheter? J Endourol. 2008;22:273-275.
Mousavi-Bahar SH, Amir-Zargar MA, Gholamrezaie HR. Laparoscopic assisted percutaneous nephrolithotomy in ectopic pelvic kidneys. Int J Urol. 2008;15:276-278.
Mozer P, Conort P, Leroy A, et al. Aid to percutaneous renal access by virtual projection of the ultrasound puncture tract onto fluoroscopic images. J Endourol. 2007;21:460-465.
Munver R, Delvecchio FC, Newman GE, et al. Critical analysis of supracostal access for percutaneous renal surgery. J Urol. 2001;166:1242-1246.
Muslumanoglu AY, Tefekli A, Karadag MA, et al. Impact of percutaneous access point number and location on complication and success rates in percutaneous nephrolithotomy. Urol Int. 2006;77:340-346.
Muzrakchi AA, Szmigielski W, Omar AJS, et al. Is the 10th and 11th intercostal space a safe approach for percutaneous nephrostomy and nephrolithotomy? Cardiovasc Intervent Radiol. 2003;27:503-506.
Nadler RB, Rubenstein JN, Kim SC, et al. Percutaneous hepatolithotomy: the Northwestern University experience. J Endourol. 2002;16:293-297.
Nagele U, Schilling D, Anastasiadis AG, et al. Closing the tract of mini-percutaneous nephrolithotomy with gelatine matrix hemostatic sealant can replace nephrostomy tube placement. Urology. 2006;68:489-493.
Narasimham DL, Jacobsson B, Vijayan P, et al. Percutaneous nephrolithotomy through an intercostal approach. Acta Radiol. 1991;32:162-165.
Netto NRJr, Ikonomidis J, Ikari O, et al. Comparative study of percutaneous access for staghorn calculi. Urology. 2005;65:659-662.
Netto NRJr, Mitre AI, Lima SV, et al. Telementoring between Brazil and the United States: initial experience. J Endourol. 2003;17:217-220.
Ng CK, Yip SKH, Sim LSJ, et al. Outcome of percutaneous nephrostomy for the management of pyonephrosis. Asian J Surg. 2002;25:215-219.
Ng CS, Herts BR, Streem SB. Percutaneous access to upper pole renal stones: role of prone 3-dimensional computerized tomography in inspiratory and expiratory phases. J Urol. 2005;173:124-126.
Noller MW, Baughman SM, Morey AF, et al. Fibrin sealant enables tubeless percutaneous stone surgery. J Urol. 2004;172:166-169.
Ogan K, Pearle MS. Oops we got in the chest: fluoroscopic chest tube insertion for hydrothorax after percutaneous nephrostolithotomy. Urology. 2002;60:1098-1099.
Ogan K, Corwin TS, Smith T, et al. Sensitivity of chest fluoroscopy compared with chest CT and chest radiography for diagnosing hydropneumothorax in association with percutaneous nephrostolithotomy. Urology. 2003;62:988-992.
Okeke Z, Andonian S, Srinivasan A, et al. Cryotherapy of the nephrostomy tract: a novel technique to decrease the risk of hemorrhage after tubeless percutaneous renal surgery. J Endourol. 2009;23:417-420.
Osman M, Wendt-Nordahl G, Heger K, et al. Percutaneous nephrolithotomy with ultrasonography-guided renal access: experience from over 300 cases. BJU Int. 2005;96:875-878.
Papatsoris A, Masood J, El-Husseiny T, et al. Improving patient positioning to reduce complications in prone percutaneous nephrolithotomy. J Endourol. 2009;23:831-832.
Papatsoris AG, Zaman F, Panah A, et al. Simultaneous anterograde and retrograde endourologic access: “the Barts technique. J Endourol. 2008;22:2665-2666.
Parsons JK, Jarrett TW, Lancini V, et al. Infundibular stenosis after percutaneous nephrolithotomy. J Urol. 2002;167:35-38.
Patel B, Mason BM, Hoenig DM. Retrograde endoscopic-assisted percutaneous renal access: a novel “lasso” technique to achieve rapid secure access to the collecting system. J Endourol. 2008;22:591-596.
Patel U, Hussain FF. Percutaneous nephrostomy of nondilated renal collecting systems with fluoroscopic guidance: technique and results. Radiology. 2004;233:226-233.
Patel U, Walkden RM, Ghani KR, et al. Three-dimensional CT pyelography for planning of percutaneous nephrostolithotomy: accuracy of stone measurement, stone depiction and pelvicalyceal reconstruction. Eur Radiol. 2009;19:1280-1288.
Pathak AS, Bellman GC. One-step percutaneous nephrolithotomy sheath versus standard two-step technique. Urology. 2005;66:953-957.
Patterson DE, Segura JW, LeRoy AJ, et al. The etiology and treatment of delayed bleeding following percutaneous lithotripsy. J Urol. 1985;133:447-451.
Pearle MS, Pierce HL, Miller GL, et al. Optimal method of urgent decompression of the collecting system for obstruction and infection due to ureteral calculi. J Urol. 1998;160:1260-1264.
Pedersen JF. Percutaneous nephrostomy guided by ultrasound. J Urol. 1974;112:157-159.
Peterson GN, Krieger JN, Glauber DT. Anaesthetic experience with percutaneous lithotripsy. A review of potential and actual complications. Anaesthesia. 1985;40:460-464.
Picus D, Weyman PJ, Clayman RV, et al. Intercostal-space nephrostomy for percutaneous stone removal. AJR. 1986;147:393-397.
Pietrow PK, Auge BK, Lallas CD, et al. Pain after percutaneous nephrolithotomy: impact of nephrostomy tube size. J Endourol. 2003;17:411-414.
Preminger GM, Assimos DG, Lingeman JE, et al. Chapter 1: AUA guideline on management of staghorn calculi: diagnosis and treatment recommendations. J Urol. 2005;173:1991-2000.
Pugach JL, Moore RG, Parra RO, et al. Massive hydrothorax and hydro-abdomen complicating percutaneous nephrolithotomy. J Urol. 1999;162:2110.
Radecka E, Brehmer M, Holmgren K, et al. Complications associated with percutaneous nephrolithotripsy: supra- versus subcostal access. A retrospective study. Acta Radiol. 2003;44:447-451.
Radecka E, Magnusson A. Complications associated with percutaneous nephrostomies. A retrospective study. Acta Radiol. 2004;45:184-188.
Rana AM, Mithani S. Tubeless percutaneous nephrolithotomy: call of the day. J Endourol. 2007;21:169-172.
Rana AM, Zaidi Z, El-Khalid S. Single-center review of fluoroscopy-guided percutaneous nephrostomy performed by urologic surgeons. J Endourol. 2007;21:688-691.
Rao PN, Dube DA, Weightman NC, et al. Prediction of septicemia following endourological manipulation for stones in the upper urinary tract. J Urol. 1991;146:955-960.
Ray K, Rattan S, Yohannes T. Urinothorax: unexpected cause of a pleural effusion. Mayo Clin Proc. 2003;78:1433-1434.
Rehman J, Chughtai B, Schulsinger D, et al. A percutaneous subcostal approach for intercostal stones. J Endourol. 2008;22:497-502.
Ricciardi S, Sallustio P, Troisi R. Life-threatening biliary complications after percutaneous nephro-lithotomy: a case report. Acta Chir Belg. 2007;107:336-337.
Richstone L, Reggio E, Ost MC, et al. Hemorrhage following percutaneous renal surgery: characterization of angiographic findings. J Endourol. 2008;22:1129-1135.
Rifaioglu MM, Berger AD, Pengune W, et al. Percutaneous management of stones in transplanted kidneys. Urology. 2008;72:508-512.
Robert M, Maubon A, Roux JO, et al. Direct percutaneous approach to the upper pole of the kidney: MRI anatomy with assessment of the visceral risk. J Endourol. 1999;13:17-20.
Roberts WW, Fugita OE, Kavoussi LR, et al. Measurement of needle-tip bioimpedance to facilitate percutaneous access of the urinary and biliary systems: first assessment of an experimental system. Invest Radiol. 2002;37:91-94.
Rosevear HM, Kim SP, Wenzler DL, et al. Retrograde ureteral stents for extrinsic ureteral obstruction: 9 years of experience at the University of Michigan. Urology. 2007;70:846-850.
Rusnak B, Castañeda-Zúñiga W, Kotula F, et al. An improved dilator system for percutaneous nephrostomies. Radiology. 1982;144:174.
Safak M, Gogus C, Soygur T. Nephrostomy tract dilation using a balloon dilator in percutaneous renal surgery: experience with 95 cases and comparison with the fascial dilator system. Urol Int. 2003;71:382-384.
Sampaio FJ, Zanier JF, Aragao AH, et al. Intrarenal access: 3-dimensional anatomical study. J Urol. 1992;148:1769-1773.
Sampaio FJB, Mandarim-de-Lacerda CA. 3-Dimensional and radiological pelviocaliceal anatomy for endourology. J Urol. 1988;140:1352-1355.
Santiago L, Bellman GC, Murphy J, et al. Small bowel and splenic injury during percutaneous renal surgery. J Urol. 1998;159:2071-2072.
Santos ARD, Rocha Filho DCB, Tajra LCF. Management of lithiasis in pelvic kidney through laparoscopy-guided percutaneous transperitoneal nephrolithotripsy. Int Braz J Urol. 2004;30:32-34.
Sardina JI, Bolton DM, Stoller ML. Entrapped Malecot nephrostomy tube: etiology and management. J Urol. 1995;153:1882-1883.
Saxby MF. Biliary peritonitis following percutaneous nephrolithotomy. Br J Urol. 1996;77:465-466.
Saxby MF. Effects of percutaneous nephrolithotomy and extracorporeal shock wave lithotripsy on renal function and prostaglandin excretion. Scand J Urol Nephrol. 1997;32:141-144.
Schaeffer AJ, Handa SE, Lingeman JE, et al. Transsplenic percutaneous nephrolithotomy. J Endourol. 2008;22:2481-2484.
Scoffone CM, Cracco CM, Cossu M, et al. Endoscopic combined intrarenal surgery in Galdakao-modified supine Valdivia position: a new standard for percutaneous nephrolithotomy? Eur Urol. 2008;54:1393-1403.
Segura JW, Patterson DE, LeRoy AJ, et al. Percutaneous removal of kidney stones: review of 1000 cases. J Urol. 1985;134:1077-1081.
Segura JW, Preminger GM, Assimos DG, et al. Nephrolithiasis clinical guidelines panel summary report on the management of staghorn calculi. J Urol. 1994;151:1648-1651.
Seldinger S-I. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 1953;39:368-376.
Shaban A, Kodera A, El Ghoneimy MN, et al. Safety and efficacy of supracostal access in percutaneous renal surgery. J Endourol. 2008;22:29-34.
Shah HN, Hegde S, Shah JN, et al. A prospective, randomized trial evaluating the safety and efficacy of fibrin sealant in tubeless percutaneous nephrolithotomy. J Urol. 2006;176:2488-2493.
Shah HN, Hegde SS, Mahajan AP, et al. Splenic injury: rare complication of percutaneous nephrolithotomy: report of two cases with review of literature. J Endourol. 2007;21:919-922.
Shah HN, Sodha HS, Khandkar AA, et al. A randomized trial evaluating type of nephrostomy drainage after percutaneous nephrolithotomy: small bore v tubeless. J Endourol. 2008;22:1433-1439.
Shaw G, Wah TM, Kellett MJ, et al. Management of renal-vein perforation during a challenging percutaneous nephrolithotomy. J Endourol. 2005;19:722-723.
Shen CH, Cheng MC, Lin CT, et al. Innovative metal dilators for percutaneous nephrostomy tract: report on 546 cases. Urology. 2007;70:418-421.
Shermak M, Shoo B, Deune EG. Prone positioning precautions in plastic surgery. Plast Reconstr Surg. 2006;117:1584-1588.
Shleyfer E, Nevzorov R, Jotkowitz AB, et al. Urinothorax: An unexpected cause of pleural effusion. Eur J Intern Med. 2006;17:300-302.
Shokeir AA, El-Nahas AR, Shoma AM, et al. Percutaneous nephrolithotomy in treatment of large stones within horseshoe kidneys. Urology. 2004;64:426-429.
Shpall AI, Parekh AR, Bellman GC. Tubeless percutaneous nephrolithotomy with antegrade stent tether: clinical experience. J Endourol. 2007;21:973-976.
Singh I, Saran RN, Jain M. Does sealing of the tract with absorbable gelatin (Spongostan) facilitate tubeless PCNL? A prospective study. J Endourol. 2008;22:2485-2493.
Singh I, Singh A, Mittal G. Tubeless percutaneous nephrolithotomy: is it really less morbid? J Endourol. 2008;22:427-434.
Skoog SJ, Reed MD, Gaudier FAJr, et al. The posterolateral and the retrorenal colon: Implication in percutaneous stone extraction. J Urol. 1985;134:110-112.
Sofikerim M, Demirci D, Huri E, et al. Tubeless percutaneous nephrolithotomy: safe even in supracostal access. J Endourol. 2007;21:967-972.
Song SH, Hong B, Park HK, et al. Paradoxical air embolism during percutaneous nephrolithotomy: a case report. J Korean Med Sci. 2007;22:1071-1073.
Soria F, Delgado MI, Sanchez FM, et al. Effectiveness of three-dimensional fluoroscopy in percutaneous nephrostomy: an animal model study. Urology. 2009;73:649-652.
Sprouse LR2nd, Hamilton INJr. The endovascular treatment of a renal arteriovenous fistula: placement of a covered stent. J Vasc Surg. 2002;36:1066-1068.
Stern J, Zeltser IS, Pearle MS. Percutaneous renal access simulators. J Endourol. 2007;21:270-273.
Stoller ML, Wolf JSJr, St. Lezin MA. Estimated blood loss and transfusion rates associated with percutaneous nephrolithotomy. J Urol. 1994;152:1977-1981.
Strohmaier WL, Giese A. Ex vivo training model for percutaneous renal surgery. Urol Res. 2005;33:191-193.
Su LM, Stoianovic ID, Jarrett TW, et al. Robotic percutaneous access to the kidney: comparison with standard manual access. J Endourol. 2002;16:471-475.
Sukumar S, Nair B, Kumar PG, et al. Supracostal access for percutaneous nephrolithotomy: less morbid, more effective. Int Urol Nephrol. 2008;40:263-267.
Symons SJ, Ramachandran A, Kurien A, et al. Urolithiasis in the horseshoe kidney: a single-centre experience. BJU Int. 2008;102:1676-1680.
Tabibi A, Akhavizadegan H, Nouri-Mahdavi K, et al. Percutaneous nephrolithotomy with and without retrograde pyelography: a randomized clinical trial. Int Braz J Urol. 2007;33:19-24.
Tanriverdi O, Boylu U, Kendirci M, et al. The learning curve in the training of percutaneous nephrolithotomy. Eur Urol. 2007;52:206-211.
Tasca A, Cacciola A. Mini-invasive management of a rare complication of percutaneous stone treatment: entrapped nephrostomy tube. Urol Int. 2004;72:165-167.
Tefekli A, Altunrende F, Tepeler K, et al. Tubeless percutaneous nephrolithotomy in selected patients: a prospective randomized comparison. Int Urol Nephrol. 2007;39:57-63.
Thanos L, Mylona S, Stroumpouli E, et al. Percutaneous CT-guided nephrostomy: a safe and quick alternative method in management of obstructive and nonobstructive uropathy. J Endourol. 2006;20:486-490.
Thiruchelvam N, Mostafid H, Ubhayakar G. Planning percutaneous nephrolithotomy using multidetector computed tomography urography, multiplanar reconstruction and three-dimensional reformatting. BJU Int. 2005;95:1280-1284.
Troxel SA, Low RK. Renal intrapelvic pressure during percutaneous nephrolithotomy and its correlation with the development of postoperative fever. J Urol. 2002;168:1348-1351.
Troxel SA, Low RK, Das S. Extraperitoneal laparoscopy-assisted percutaneous nephrolithotomy in a left pelvic kidney. J Endourol. 2002;16:655-657.
Turillazzi E, Pomara C, Bisceglia R, et al. Vascular air embolism complicating percutaneous nephrolithotomy: medical malpractice or fatal unforeseeable complication? Urology. 2009;73:e1-e4.
Valdivia Uria J, Lachares Santamaria E, Villarroya Rodriguez S, et al. [Percutaneous nephrolithectomy: simplified technic (preliminary report)]. Arch Esp Urol. 1987;40:177-180.
Valdivia Uria JG, Valle Gerhold J, Lopez Lopez JA, et al. Technique and complications of percutaneous nephroscopy: experience with 557 patients in the supine position. J Urol. 1998;160:1975-1988.
Varkarakis J, Su LM, Hsu TH. Air embolism from pneumopyelography. J Urol. 2003;169:267.
Varkarakis J, Sebe P, Pinggera GM, et al. Three-dimensional ultrasound guidance for percutaneous drainage of prostatic abscesses. Urology. 2004;63:1017-1020.
Viola D, Anagnostou T, Thompson TJ, et al. Sixteen years of experience with stone management in horseshoe kidneys. Urol Int. 2007;78:214-218.
Wah TM, Weston MJ, Irving HC. Percutaneous nephrostomy insertion: outcome data from a prospective multi-operator study at a UK training centre. Clin Radiol. 2004;59:255-261.
Watterson JD, Cook A, Sahajpal R, et al. Percutaneous nephrolithotomy of a pelvic kidney: a posterior approach through the greater sciatic foramen. J Urol. 2001;166:209-210.
Watterson JD, Soon S, Jana K. Access related complications during percutaneous nephrolithotomy: urology versus radiology at a single academic institution. J Urol. 2006;176:142-145.
Weir MJ, Honey JD. Complete infundibular obliteration following percutaneous nephrolithotomy. J Urol. 1999;161:1274-1275.
Wenzler DL, Kim SP, Rosevear HM, et al. Success of ureteral stents for intrinsic ureteral obstruction. J Endourol. 2008;22:295-299.
Wickham JE, Miller RA, Kellett MJ, et al. Percutaneous nephrolithotomy: one stage or two? Br J Urol. 1984;56:582-585.
Williams SK, Bird VG, Maurici G, et al. Borrowing from interventional radiology: novel technique to dilate scarred nephrostomy tract. Urology. 2008;72:1156-1158.
Winfield HN, Weyman P, Clayman RV. Percutaneous nephrostolithotomy: complications of premature nephrostomy tube removal. J Urol. 1986;136:77-79.
Winfree CJ, Kline DG. Intraoperative positioning nerve injuries. Surg Neurol. 2005;63:5-18.
Wolf JSJr, Bennett CJ, Dmochowski RR, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol. 2008;179:1379-1390.
Wong C, Leveillee RJ. Single upper-pole percutaneous access for treatment of ≥5-cm complex branched staghorn calculi: is shockwave lithotripsy necessary? J Endourol. 2002;16:477-481.
Wong C, Zimmerman RA. Laparoscopy-assisted transperitoneal percutaneous nephrolithotomy for renal caliceal diverticular calculi. J Endourol. 2005;19:608-613.
Yadav R, Aron M, Gupta NP, et al. Safety of supracostal punctures for percutaneous renal surgery. Int J Urol. 2006;13:1267-1270.
Yadav R, Gupta NP, Gamanagatti S, et al. Supra-twelfth supracostal access: when and where to puncture? J Endourol. 2008;22:1209-1212.
Young AT, Hunter DW, Castaneda-Zuniga WR, et al. Percutaneous extraction of urinary calculi: use of the intercostal approach. Radiology. 1985;154:633-638.
Zhang Y, Ou TW, Jia JG, et al. Novel biologic model for percutaneous renal surgery learning and training in the laboratory. Urology. 2008;72:513-516.
Zhou X, Gao X, Wen J, et al. Clinical value of minimally invasive percutaneous nephrolithotomy in the supine position under the guidance of real-time ultrasound: report of 92 cases. Urol Res. 2008;36:111-114.
Ziaee SA, Karami H, Aminsharifi A, et al. One-stage tract dilation for percutaneous nephrolithotomy: is it justified? J Endourol. 2007;21:1415-1420.