chapter 55 Laparoscopic Surgery of the Kidney
Surgical treatment is central to addressing many urologic conditions affecting the kidneys. Decades of experience have demonstrated the efficacy and durability of operative excision for malignancies, as well as reconstruction for obstruction. Unfortunately, traditional open surgical approaches, although extremely effective, are associated with significant postoperative discomfort and recuperation. Studies also demonstrate permanent body surface alterations with flank incisions resulting in significantly larger postoperative surface area and volume changes on the operated flank compared to the noninvolved flank. Indeed, patients report dissatisfaction with the body changes occurring in up to 60% of flank incisions (Chatterjee et al, 2004; Kobayashi et al, 2004).
Minimally invasive surgical approaches were born out of the desire to address secondary issues related to surgery, including incisional pain, convalescence, and cosmesis. Initially applied to treat stone disease, advances in video technology and surgical tools have now been used to treat the most complex of renal pathologies, with less morbidity compared to standard operative approaches. Clayman and associates initiated this revolution in renal surgery in 1990 when they introduced the laparoscopic nephrectomy (Clayman et al, 1991). This was performed in an octogenarian with a kidney mass, and although the procedure took over 7 hours, its impact on postoperative recovery was immediately apparent. Subsequently, this approach has been applied to treat every aspect of operative renal disease.
Multiple studies have demonstrated that laparoscopic renal surgery provides recuperative as well as cosmetic advantages in contrast to open surgery (Kerbl et al, 1994a; Dunn et al, 2000; Gill et al, 2003). With experience, this is now routinely accomplished without compromise to surgical outcomes. As such, the laparoscopic technique has evolved into the preferred alternative in treating surgical diseases of the kidney. This chapter will discuss indications, present techniques, review results, and outline potential complications of laparoscopy applied to the kidney.
Patient Evaluation and Preparation
Basic laparoscopic principles are thoroughly discussed in Chapter 9. Patient selection and preparation for renal surgery is similar to that for comparable open surgery. A pertinent history and physical are necessary to identify potential issues that could arise during surgery. Prior abdominal, retroperitoneal, or renal surgery is not a contraindication to laparoscopic surgery; however, type and extent of prior abdominal surgery will drive trocar location, as well as choice of transperitoneal or extraperitoneal access (Chen et al, 1998; Cadeddu et al, 1999). In addition, degree of obesity may influence the type of access and location of trocars (Fugita et al, 2004; Hedican et al, 2004; Kapoor et al, 2004). Surgeon experience and available equipment will facilitate or deter use of the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, CA) in treating renal pathology. Informed consent is obtained with a detailed discussion of potential complications including the potential for open conversion.
Coagulopathies should be corrected to minimize chances of intraoperative or postoperative bleeding. With the increased use of cardiac stenting, more patients are on chronic antiplatelet therapy. A conversation should occur with the patient’s cardiologist to plan cessation of therapy to minimize risk of perioperative coronary artery thrombosis. If necessary, complex laparoscopic renal procedures have been performed while patients continue aspirin therapy. Uremic patients with prolonged bleeding time may benefit from desmopresssin acetate (desamino-D-arginine vasopressin [DDAVP] 0.3 to 0.4 µg/kg) given intravenously an hour before surgery to improve platelet function (Mannucci et al, 1983). A side effect of this therapy, iatrogenic hyponatremia, has been reported (Pruthi et al, 2002).
Laboratory and imaging studies are obtained as indicated by each patient’s medical history. Patients should have blood typed and screened. Cross matching of blood is at the discretion of each surgeon and based on operative experience and complexity of the procedure. Bowel preparation is helpful to improve visualization and working space.
Imaging studies that define the pathology should be available in the operative suite to define anatomy and, along with operative site marking, minimize the risk of wrong side surgery. Angiography, embolization, and stent placement are not routine and related to each individual procedure.
Anesthesic Considerations in Laparoscopy
The majority of laparoscopic renal procedures require a general anesthetic, and a patient’s pulmonary and cardiac function must tolerate this anesthetic approach (Monk and Weldon, 1992). The pneumoperitoneum can impact upon patients with severe cardiopulmonary disease by compromising ventilation and venous return (Arthure, 1970; Hodgson et al, 1970; Nunn 1987; Lew et al, 1992). Patients with chronic pulmonary disease may not be able to compensate for the pneumoperitoneum-induced hypercarbia and may require working at lower pressures, use of helium as an insufflant, or open conversion (Monk and Weldon, 1992; Wolf et al, 1996; Makarov et al, 2007). The use of valveless trocar systems has also been associated with less carbon dioxide absorption (Herati et al, 2009).
Considerations in Obese Patients
Obesity is not a contraindication for laparoscopic surgery, but can make retraction and identification of anatomic structures challenging. It is for these reasons that laparoscopy in obese patients is associated with an increased risk of open conversion when compared with nonobese patients (Fazeli-Matin et al, 1999). In addition, although complication rates for laparoscopy in obese patients are higher when compared with laparoscopy in the general population (Mendoza et al, 1996), pulmonary and wound complications are lower with laparoscopy when compared with an open approach (Kapoor et al, 2004; Montgomery et al, 2005). Other factors to account for in the obese population include the increased distance to the operative field, which calls for modifying trocar location and number, as well as the use of longer instrumentation (Doublet and Belair, 2000; Jacobs et al, 2000). Consideration must also be given to the weight of the pannus, which may raise the intra-abdominal pressure and limit working space. The potential for rhabdomyolysis, a rare but devastating complication in the obese as well as very muscular individuals, must also be considered (Troppmann and Perez, 2003).
Surgical Approaches and Obtaining Access
There are now five laparoscopic approaches to renal surgery: transperitoneal, retroperitoneal, hand-assisted, robotic, and laparoendoscopic single-site surgery (LESS)/natural orifice transluminal endoscopic surgery (NOTES). Each approach can have discrete advantages and limitations depending upon the clinical situation and surgeon familiarity. To date there have been no studies that have shown a definitive recuperative advantage of any one of these approaches. Potential differences in cosmesis may exist, but have also not been well studied to date.
Transperitoneal Laparoscopy
The transperitoneal approach is the traditional and most widely used method of addressing renal pathology. It provides the largest working space, facilitates orientation by providing readily identifiable anatomic landmarks, affords greater versatility in angles and direction of laparoscopic trocars and instruments, and can result in the smallest size and number of ports. The equipment is mature and techniques are well defined and, like any approach, it requires significant expertise in instrument manipulation and suturing.
Patient Positioning and Trocar Placement
For most transperitoneal renal surgery, the patient is initially positioned supine for intravenous (IV) access, the induction of general anesthesia, and endotracheal intubation. A bladder catheter and orogastric tube are placed for decompression of the bladder and stomach during insufflation, trocar placement, and dissection. Sequential compression stockings are placed for deep venous thrombosis prophylaxis. When performing transperitoneal procedures, including robotic and LESS surgeries, patients are positioned in a 30- to 45-degree flank-up position. Care is taken to pad all pressure points. The patient is secured to the operating table to allow lateral tilting of the table (Fig. 55–1). Tilting the table away from the affected kidney will help move bowel out of the operative field. There is no need to flex the table or use a kidney rest as there is with open surgery. The equipment in the operating room is situated to maximize the use of space and allow all members of the surgical team to view the procedure (Fig. 55–2). During the skin preparation and draping, the entire flank and abdomen are included in case conversion to an open procedure is required.
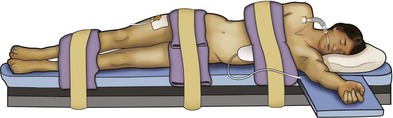
Figure 55–1 The patient is placed in a modified flank position, with the operative side tilted up 30 to 45 degrees using a gel roll or a rolled blanket supporting the back. The lower arm is placed on a padded armrest, and the other arm is flexed at the elbow and rested over the chest. Wide cloth or silk tape is used to secure the patient to the operating table to allow for table rotation during the surgery.
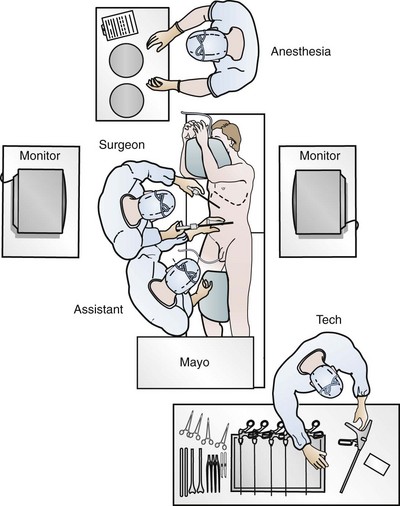
Figure 55–2 The operating room configured for left nephrectomy. Two monitors allow the assistant to follow the procedure. The scrub tech is positioned to easily assist with instrument passage and exchange.
Once a pneumoperitoneum is established, three to five trocars are initially placed to complete the dissection (Fig. 55–3). There are a variety of trocar configurations that are effective for each procedure.
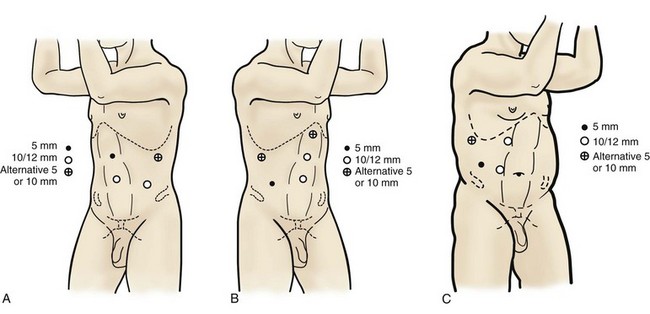
Figure 55–3 Trocar sites for left-sided (A) and right-sided (B) procedures. A 12-mm trocar is placed lateral to the rectus at the level of the umbilicus, a second 10-mm trocar is placed at the umbilicus, and a 5-mm trocar is inserted in the midline between the umbilicus and the xiphoid process. C, In obese patients, all trocars are shifted laterally. Optional accessory subcostal, subxiphoid, and low midline trocar positions, which may be helpful for retraction, are also shown.
A 12-mm trocar is placed lateral to the rectus at the level of the umbilicus; this trocar is used for instrumentation and the passage of sutures, bulldog clamps, or staplers to secure and divide hilar vessels. In short patients, this may be placed in the midline, halfway between the umbilicus and pubis. A 10-mm trocar is placed at the umbilicus for the camera, and a 5- or 10-mm port is inserted in the midline 2 cm below the xiphoid process. In obese patients, all trocar sites are moved laterally (see Fig. 55–3C). Additional trocars for retraction may be needed for visualization or assist with organ entrapment (Fig. 55–4).
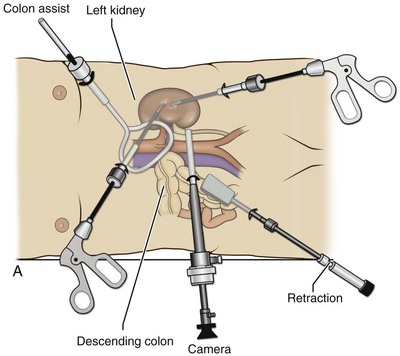
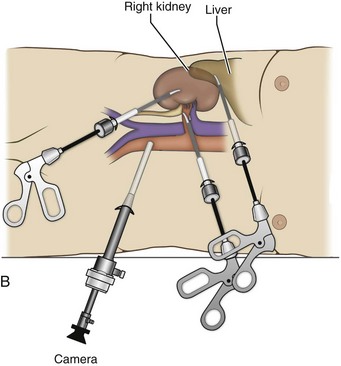
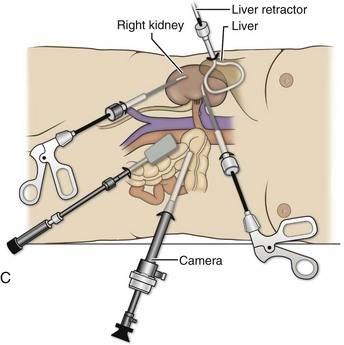
Figure 55–4 Options for additional trocar placement and instrumentation. A, Additional retraction during left-sided procedures can be accomplished with the use of a blunt instrument passed through a 5- or 10-mm trocar placed above the symphysis pubis, or a 5-mm instrument and retractor passed through a subcostal incision. B, In right-sided procedures, the liver and bowel can be retracted through a 3- or 5-mm trocar placed in the midline. C, In right-sided procedures, the liver and bowel can be retracted through a 5-mm trocar with a 5-mm instrument. An optional 10-mm lower midline trocar may also be placed for retraction, freeing the two other working hands for dissection.
Retroperitoneal Laparoscopy
The retroperitoneal approach mimics open surgery because the peritoneal cavity is avoided. A potential space is created to visualize the surgical field. It may be preferred for selected cases of laparoscopic partial nephrectomy, cyst marsupialization, pyeloplasty, renal biopsy, or in patients who have had peritonitis or who have undergone multiple prior abdominal surgeries.
Patient Positioning and Trocar Placement
Using this approach, patients are placed in a full-flank position. Modest table flexion can help increase the distance between the ribs and iliac crest to facilitate trocar placement. An axillary roll is required, as well as care in securing the patient to the bed. Arms may be secured on pillows or a purpose-built armrest. A 15-mm transverse incision is made in the posterior axillary line, midway between the tip of the 12th rib and the iliac crest (Fig. 55–5A). After deepening the dissection downward through the lumbodorsal fascia, the retroperitoneum is entered, and a working space may be developed using blunt dissection with the tip of a finger in the space between the psoas muscle and the kidney (Fig. 55–5B). A simple balloon created from two fingers of a size-8 or -9 glove may then be inserted and filed with CO2 or saline to further develop the retroperitoneal working space (Fig. 55–5C). A blunt tip trocar is then passed through the incision, and the trocar cuff is expanded and cinched to the skin to prevent leakage of CO2 (Fig. 55–5D). An alternative entry approach involves entry with the 0-degree lens and visual obturator through the initial incision (Fig. 55–6A). Entry into the retroperitoneum may be confirmed by the appearance of the characteristic yellow retroperitoneal fat, insufflation is initiated, and blunt dissection using only the laparoscope is performed to develop a working space (Fig. 55–6B). Caution must be used not to enter too anteriorly because inadvertent peritoneal entry or colon injury may occur; entering too posteriorly may result in bleeding from the quadratus lumborum or psoas muscles. Once the working space is established through either approach, pertinent structures may be identified for orientation and additional trocars placed. Typically, a 5-mm trocar is placed just off the tip of the 12th rib, and a 12-mm trocar is placed posteriorly and superiorly relative to the camera port (see Fig. 55–5A).
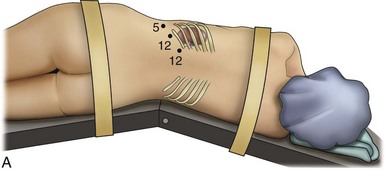
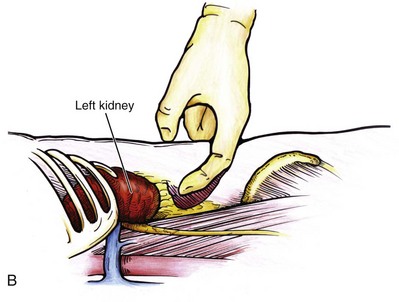
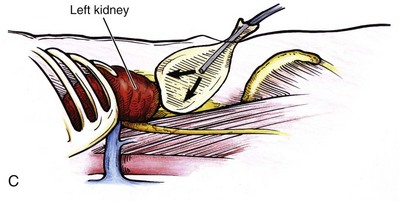
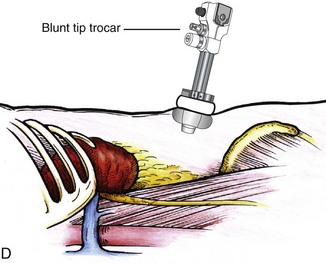
Figure 55–5 Trocar placement for retroperitoneal kidney surgery. A, With the patient in the full lateral position, the hips flexed, and the kidney rest elevated, a 15-mm incision is made 2 cm below the tip of the 12th rib, between the rib and the anterior superior iliac spine. B, The index finger is inserted through the incision and used for blunt dissection to create a hole from the skin through the muscle into the retroperitoneal space. If the finger is in the correct position, the surgeon should feel the smooth surface of psoas muscle and the lower pole of the kidney covered by Gerota fascia. C, To quickly create the working space, insert a balloon created from the finger of a size 8 or 9 glove, secured with silk suture over a simple red rubber catheter. The balloon is then filled with 600 to 800 mL of saline. D, A Blunt Tip Trocar (U.S. Surgical, Norwalk, CT) is used to seal the trocar site. Because of its low profile, it will not obstruct the view or take up useful space in the retroperitoneum. The balloon/collar configuration eliminates the need for sutures and allows 360-degree rotation.
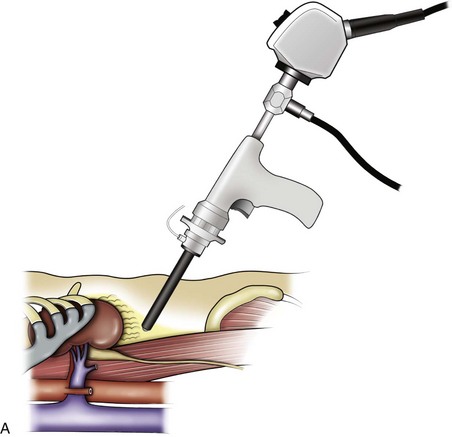
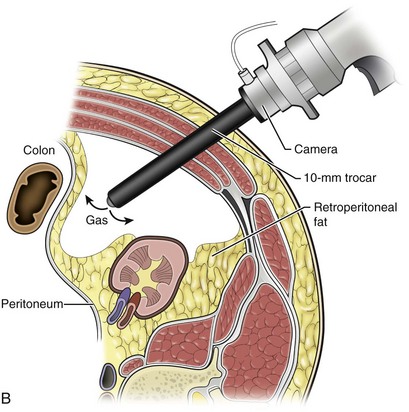
Figure 55–6 A, Standing behind the patient, the surgeon initially develops a space bluntly between the psoas muscle and the kidney using the visual obturator with the 0-degree laparoscope through it. B, Together, they are used to bluntly push the peritoneum medially, creating a working space large enough to allow placement of additional trocars.
The greatest limitations of the retroperitoneal approach are the limited working space and more subtle anatomic landmarks. The smaller working space limits the distance between trocars, potentially leading to awkward hand positioning. Also, the pathology is much closer to the lens, and frequent smudging of the tip may occur. If additional space is needed during the procedure, initial retroperitoneal access can be expanded to a transperitoneal approach through opening the peritoneum under direct vision. Despite these limitations, the retroperitoneal approach may be preferred in some cases, and with adequate experience, a wide variety of laparoscopic renal surgery may be performed.
Hand-Assisted Laparoscopy
Hand assistance offers a bridge between open surgery and laparoscopy (Nakada et al, 1997). It offers more intuitive assistance from the human hand with dissection and retraction, simultaneously providing tactile feedback to the surgeon. Several manufacturers make devices for this purpose. An incision large enough for the hand must be created and can also be used as an extraction site. This incision site is usually larger than one used for intact extraction during pure laparoscopic surgery. This technique may be advantageous for the novice laparoscopic surgeon and in patients with significant scaring about the kidney, or if a difficult dissection is anticipated. Hand assistance may also be employed in the event of an emergency, such as bleeding, by extending a trocar site and placing a hand port to assist in repair of an injury or vascular control. Indications are identical to those for laparoscopic nephrectomy or laparoscopic partial nephrectomy.
Patient Positioning and Trocar Placement
The patient is positioned just as for pure laparoscopic kidney surgery. The initial incision is for the hand port that is made through the skin and fascia and into the peritoneal cavity. Location will depend on handedness of the surgeon, operative side, and body habitus of the patient (Figs. 55-7 and 55-8). Care must be taken to avoid making the incision too large, because gas may escape, making the procedure more difficult due to decreased working space. Once the hand-assisted device is placed, the peritoneum is insufflated, and the additional trocars may be placed under direct vision by placing the camera through the hand port.
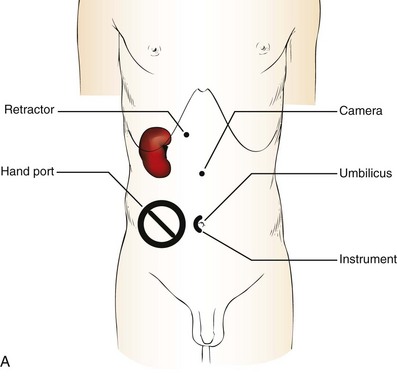
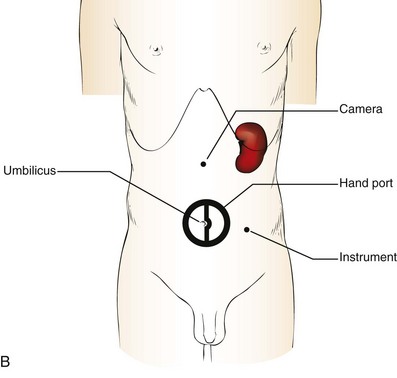
Figure 55–7 Port placement for a right-handed surgeon for hand assistance. A, For a right-sided kidney, the hand-assisted device is placed in the right lower quadrant for insertion of the left hand, and dissection is performed with instruments in the right hand placed through an umbilical trocar. The camera is placed several centimeters above the umbilicus in the midline. On the right side, retraction of the liver is usually necessary to allow visualization and dissection of the renal hilum. A liver or bowel retractor can be placed through a subcostal trocar to assist with visualization or irrigation/aspiration. B, For the left kidney, the hand-assisted device and left hand are placed though a periumbilical incision and dissection is performed with the right hand using an instrument placed in the subcostal margin just medial to the nipple. The camera is placed several centimeters lateral to the edge of the actual hand-assisted device (not the edge of the incision). Additional assistance can be delivered through the most lateral trocar site.
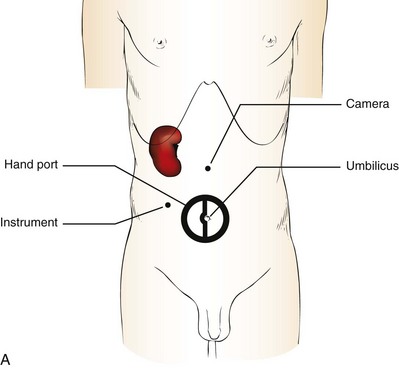
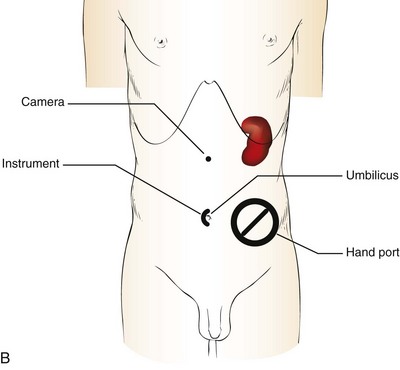
Figure 55–8 Port placement for a left-handed surgeon for hand assistance A, When operating on the right kidney, a left-handed surgeon places the hand-assisted port in the periumbilical location for insertion of the right hand. The working port for the left hand is placed lateral to the rectus muscle, in line with or just inferior to the level of the umbilicus. The camera is placed through a lateral trocar in the anterior axillary line. Additional assistance with retraction of the liver can be accomplished through a subcostal trocar. B, For a left-handed surgeon operating on the left kidney the hand-assisted port is placed in the left lower quadrant for insertion of the right hand. The left hand works with the instrument passed through an umbilical trocar, and the camera is placed midway between the umbilicus and the xiphoid process. Additional assistance with retraction or aspiration can be accomplished through a fourth trocar placed at the subcostal margin.
There are some limitations in location of port placement, and the hand may potentially get in the way of visualization or dissection instrumentation. Some leakage of gas may occur about the device, resulting in difficulty in maintaining the pneumoperitoneum. These devices exert 30 to 100 mm Hg of pressure on the arm, which may account for surgeons developing tingling, numbness, or pain in the forearm or hand (Monga et al, 2004).
Robotic-Assisted Laparoscopy
The da Vinci computer–aided surgical system has been used to perform what has commonly become known as robotic surgery. This device uses a computer system and a series of mechanical arms to translate surgeon movements familiar to open surgery to a laparoscopic platform. As such, instrument movement of right and left is preserved, as opposed to pure laparoscopic surgery where it is reversed. Also, hand-eye association is preserved and, a dual lens system provides 3-dimensional (3D) depth perception. The addition of instrument articulation facilitates dissection and suturing for those lacking laparoscopic abilities. These additions have obviated the need for high-level laparoscopic skills, in some instances to allow more surgeons to offer a minimally invasive approach to their patients.
Patient Positioning and Trocar Placement
Patient positioning will depend, in part, upon tumor location if partial nephrectomy is to be undertaken. In addition, the timing of docking the robot must be considered because with the robot docked, table rotation will not be possible. The entire procedure may be performed with the aid of the robot, or the initial part of the operation may be performed using standard laparoscopy, with use of the robot only for tumor excision and renorrhaphy.
Most authors prefer a flank position with the table modestly flexed (Aron et al, 2008; Deane et al, 2008; Benway et al, 2009). As the robotic procedure requires more trocars than standard laparoscopy, this allows more adequate spacing of instruments and less clashing of the robotic arms. Inclining the table may provide additional space at the back of the patient for the robot and other equipment (Fig. 55–9).
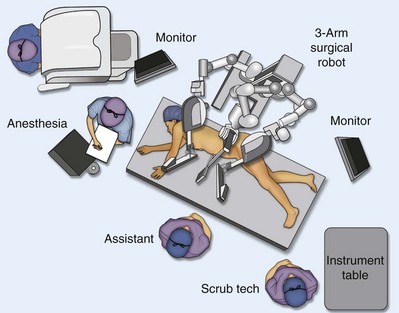
Figure 55–9 Operating room configured for left-sided robotic-assisted laparoscopic partial nephrectomy.
Robotic trocars plus the additional assistant and camera ports are typically used for the procedure. A port-in-port technique of robotic-assisted partial nephrectomy (RaPN) has also been described, wherein the righthand and lefthand working ports are actually standard 12-mm laparoscopic ports (Aron et al, 2008). The robotic 8-mm ports are inserted through these 12-mm ports to perform RaPN. This arrangement can be advantageous in event of an intraoperative complication or robotic malfunction, wherein emergent conversion to pure laparoscopic surgery is necessary. The robot can be undocked expeditiously and standard LPN performed in the usual fashion without requiring additional time to insert new ports or working through 8-mm robotic ports that preclude passage of the necessary CT-1 or CT-X needles.
A three-arm configuration includes four to five total trocars, a 12-mm periumbilical camera port, an 8-mm subcostal robotic trocar in the anterior axillary line, an 8-mm robotic trocar in the posterior axillary line placed above the iliac crest, and one 12-mm assistant trocar in the low midline to allow passage of sutures, bulldog clamps, suction, or retraction. An additional 12-mm subxiphoid trocar may be used, if necessary, for additional retraction or passage of bulldog clamps if the angle is more optimal (Fig. 55–10A). A four-arm configuration involves five to six total trocars, using the same general configuration as the three-arm technique, but shifting the robotic trocars slightly to avoid clashing of the arms (Fig. 55–10B).
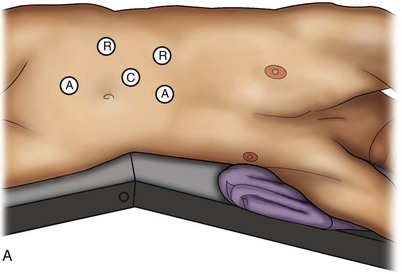
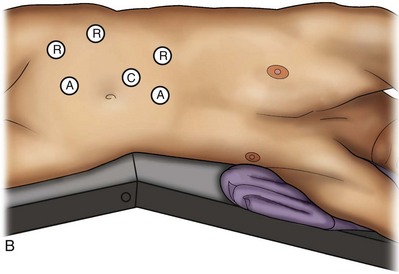
Figure 55–10 Trocar placement for robotic-assisted laparoscopic renal surgery. A, Three-arm system configuration. B, Four-arm system configuration. A, assistant trocars, R, robotic trocars, C, camera port.
The shortcomings of the device include the initial capital investment and ongoing disposable expenses that make robotic procedures more expensive than other laparoscopic approaches. In case of instrument malfunction, the surgeon must either have laparoscopic skills or abort and perform the surgery in an open manner. Robotic cases also require a skilled bedside assistant and use more, and potentially larger, port sites, whereas most laparoscopic renal procedures can be completed in a solo fashion using a mechanical endoscope holder and two working trocars.
Laparoendoscopic Single-Site Surgery (LESS) and Natural Orifice Transluminal Endoscopic Surgery (NOTES)
Laparoendoscopic single-site (LESS) surgery describes laparoscopic techniques that consolidate all ports within a single skin incision, often concealed within the umbilicus (Box et al, 2008). It has evolved to improve upon cosmesis associated with standard laparoscopic surgery and uses commercially available multichannel working ports placed in the umbilicus or below the lower abdominal hair line to minimize postoperative visible scars. Initial clinical cases of virtually every type of extirpative and reconstructive urologic procedures have now been performed by LESS surgery.
Patient Positioning and Trocar Placement
Because LESS surgery remains a procedure in evolution, there is no widely accepted method of positioning or gaining access. Modified and full-flank positioning have been described and are identical to standard transperitoneal or retroperitoneal kidney surgery, respectively. Once pneumoperitoneum has been established, one may cluster multiple traditional low-profile trocars close together within a single, small extraction incision (Fig. 55–11). Alternatively, newer purpose-specific access devices (Fig. 55–12) may be used in combination with conventional laparoscopic or flexible instrumentation. The devices are secured using preplaced fascial sutures or with an inner and outer ring drawn together with a cylindrical sleeve. Characteristics of single-site access options are described in Table 55–1.
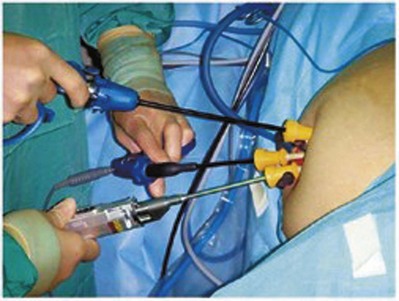
Figure 55–11 Laparoendoscopic single-site surgery performed using three low-profile trocars inserted through a single small extraction incision. A flexible laparoscope and flexible instrumentation may be used.
(From Tracy CR, Raman JD, Cadeddu JA, Rane A. Laparoendoscopic single-site surgery in urology: where have we been and where are we heading? Nat Clin Pract Urol 2008;5:561–8.)
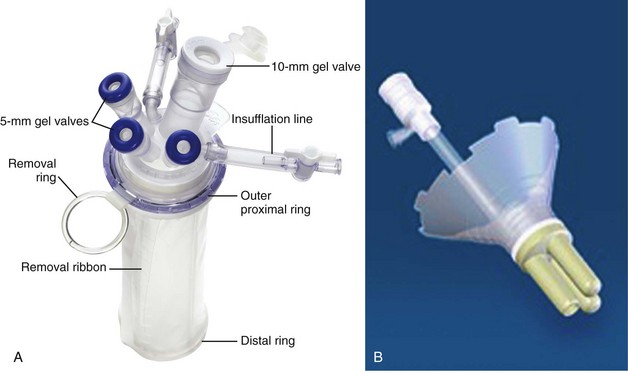
Figure 55–12 Purpose-specific devices for laparoendoscopic single-site surgery. A, The TriPort system (Advanced Surgical Concepts, Bray, Ireland). B, The Uni-X system (Pnavel Systems, Cleveland, OH). Both allow for passage of multiple instruments through a single incision.
Table 55–1 Access Options for Laparoendoscopic Single Site (LESS) Surgery
| ACCESS TYPE | DESCRIPTION |
|---|---|
| Keyhole | Use of three closely approximated periumbilical trocars placed side by side in a single skin incision or three separate incisions |
| No additional device required | |
| Typically used with articulating camera and specialized instrumentation | |
| Insufflation through trocar | |
| TriPort (Advanced Surgical Concepts, Bray, Ireland) | Open or closed access, may be used with multiple incision sizes (5-mm to hand-assisted incision) |
| Anchored by inner (intra-abdominal) and outer rings drawn together with cylindrical sleeve | |
| 3-Port (one 12-mm and two 5-mm) configuration or 4-port (two 12-mm and two 5-mm) configuration available (Quadport) | |
| Insufflation through valve housing | |
| Uni-X (Pnavel Systems, Cleveland, OH) | Open access technique, requires 2-cm fascial incision |
| Anchored with preplaced fascial sutures | |
| Single port encompassing three 5-mm access ports | |
| Typically used with articulating camera and specialized instrumentation | |
| Insufflation through valve housing | |
| Gelport (Applied Medical, Rancho Santa Margarita, CA) | Open access technique, requires 2.5- to 5-cm fascial incision |
| Anchored by inner and outer rings drawn together with cylindrical sleeve | |
| Can accommodate all trocar sizes | |
| May allow for wider spacing of trocars | |
| Insufflation through trocar placed through device |
These approaches are technically challenging and the most difficult of the minimally invasive techniques. To date, no differences in postoperative morbidity or recovery have been reported (Raman et al, 2009). Advances in instrumentation may increase applicability and dissemination into mainstream urologic practice.
NOTES involves using a natural orifice to perform the entire operation. In the gastrointestinal (GI) and surgical literature, the mouth, vagina, and rectum have been used to remove organs such as the appendix and gall bladder (Rao et al, 2006; Zorron et al, 2007; Palanivelu et al, 2008). In urology, to date experience with pure NOTES surgery in humans is limited (Kaouk et al, 2009). Several authors have reported hybrid approaches with NOTES and standard laparoscopic techniques, using the vagina as an access and extraction site for nephrectomy (Branco et al, 2008; Alcaraz et al, 2010; Sotelo et al, 2010).
Simple Nephrectomy
Benign Indications
Laparoscopic simple nephrectomy is indicated in the treatment of most benign renal diseases. Renovascular hypertension that is not correctable with medication or angiographic repair may be managed with a simple nephrectomy. Patients with chronic pain syndromes may benefit from nephrectomy, including symptomatic acquired renal cystic disease, autosomal dominant polycystic kidney disease (ADPKD), chronic hydronephosis not amenable to surgical repair, loin pain/hematuria syndrome. Chronic infectious processes that are recalcitrant to antibiotic therapy may also be approached laparoscopically, including chronic pyelonephritis, xanthogranulomatous pyelonephritis (XGP), and renal tuberculosis. These conditions are associated with a higher rate of conversion to open surgery due to perinephric inflammatory changes and loss of tissue planes (Gupta et al, 1997; Bercowsky et al, 1999). In some cases, subcapsular nephrectomy may be necessary to complete the procedure. Hand assistance may also be of benefit and avoid open conversion in cases with dense scar tissue formation or inflammatory reaction (Rosoff et al, 2006). Successful retroperitoneal laparoscopic simple nephrectomy has also been reported in 30 of 31 patients with nonfunctioning kidneys secondary to tuberculosis (Lee et al, 2002). Other benign conditions that may call for nephrectomy include patients with a multicystic dysplastic kidney or a symptomatic failed renal transplant. Again, in this later case, a subcapsular dissection may be required.
Procedure
Reflection of the Colon
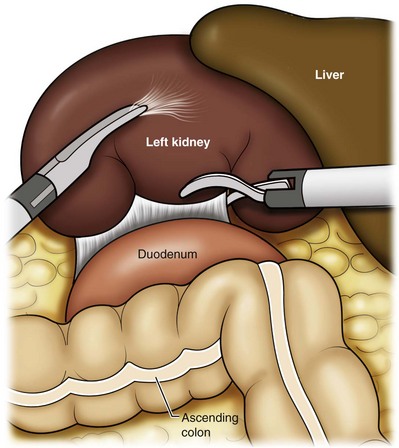
For a left nephrectomy and all renal surgery, the line of Toldt is incised from below the lower pole of the kidney inferiorly to above the spleen superiorly (Fig. 55–13). The inferior limit of this incision may be extended inferiorly, if the bowel does not move sufficiently. The lienocolic ligament should be incised to allow the spleen to fall medially along with the pancreas and the colon (see Fig. 55–13). Care must be taken to avoid injuring the diaphragm with this maneuver. The thin colorenal attachments are incised and the colon is swept medially (Fig. 55–14). Care should be taken to avoid making a hole in the mesentery. Mesenteric fat has a brighter hue of yellow compared with the retroperitoneal or Gerotas fat, which allows for identification of the correct plane of dissection. If the operative field is not adequately visualized, a paddle retractor may be placed in an additional lower midline trocar to aid in retracting the colon, pancreas, and spleen medially (see Fig. 55–4A). Blunt and sharp dissection is necessary to move these structures off the anterior surface of the kidney and hilum.
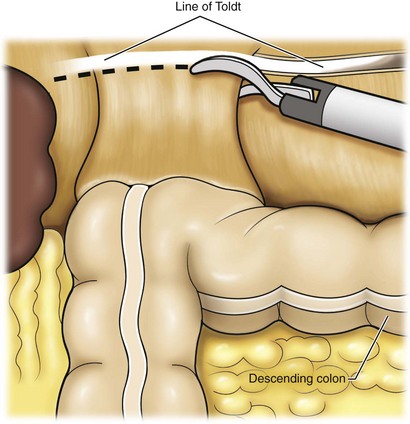
Figure 55–13 Incision of the white line of Toldt with endoshears, bipolar cautery, or ultrasonic energy allows reflection of the colon. Continuing superiorly allows incision of the lienocolic ligament, facilitating reflection of the spleen, pancreas, and colon.
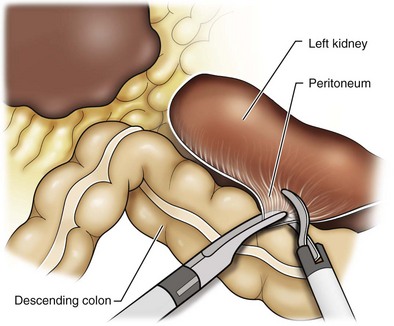
Figure 55–14 Medial traction on the colon helps identify additional colorenal attachments and assists in differentiating the undersurface of the large bowel mesentery. Care must be taken at this step to avoid creating a mesenteric window.
During a right-sided nephrectomy, the peritoneal incision is carried medially and parallel to the lateral border of the venacava and duodenum. A lateral (anterior axillary line) or high midline port may be needed to retract the liver anteriorly (see Fig 55–4B and C). Care must be taken to avoid thermal injury to the duodenum and gall bladder during incision of the peritoneum. Medial traction on the colon reveals colorenal attachments that must be divided to complete the colon dissection. Again, a low midline retractor may be helpful for visualization. A Kocher maneuver may be required to fully expose the medial portion of the kidney and the connective tissue overlying the renal hilum (Fig. 55–15).
Dissection of the Ureter
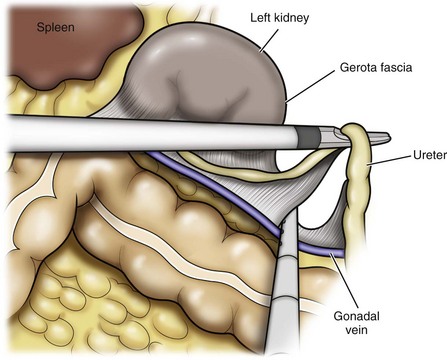
Once the colon has been adequately mobilized, the psoas muscle and tendon should be identified. Following this structure medially, the gonadal vessels are usually first encountered. These should be swept medially and the ureter is usually located just deep to these vessels. Peristalsis of the ureter can help differentiate between these two structures. Once identified, the ureter is elevated and followed proximally to the lower pole of the kidney. The ureter is not divided at this time, because it can be used to help elevate the kidney (Fig. 55–16). The tissue posterior to the ureter and lower pole of the kidney is swept anteriorly to further expose the psoas muscle. Care should be taken to try and stay above the psoas fascia to minimize postoperative thigh numbness. The instrument in the subxyphoid trocar is used to slide under the kidney all the way to the sidewall. This allows the surgeon to lift the kidney and place medial lymphatic and vascular attachments on stretch.
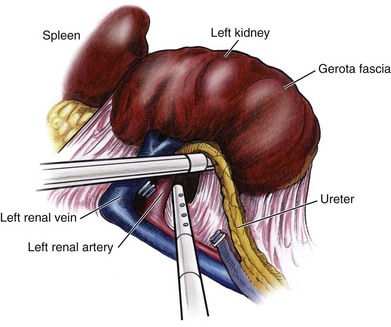
Identification of the Renal Hilum
Safe dissection of the renal hilum requires medial retraction of the colon and bowel by gravity or an additional retractor, as well as anterior retraction of the kidney, lifting it out of the renal fossa. With the ureter and lower pole of the kidney elevated, vessels entering the renal hilum can be identified and bluntly dissected using the tip of the irrigator-aspirator. Firm elevation of the lower pole assists in identification and dissection of the renal hilar vessels (Fig. 55–17). This is accomplished by gently placing the lateral grasper under the ureter and kidney until it abuts the abdominal sidewall. It is important to be sure that the grasper is against the muscle and not into the renal parenchyma. A gentle, layer-by-layer dissection is performed with the irrigator-aspirator until the renal vein is uncovered. There is usually an anterior bundle of connective tissue that needs to be incised in order to fully expose and visualize the vein. Gonadal, lumbar, and accessory venous branches can be clipped and divided as necessary.
Securing the Renal Blood Vessels
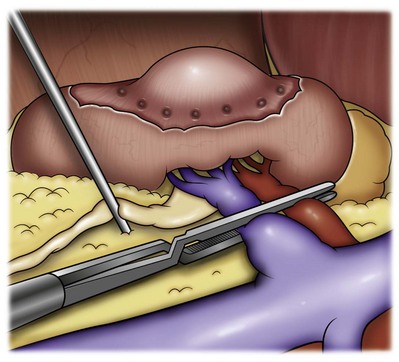
By clearing off inferior attachments and lymphatics, one can identify the renal artery. If the irrigator-aspirator tip is not precise enough for meticulous dissection, a hook electrode can be used to dissect the lymphatic vessels free of the vein and artery. With an endovascular gastrointestinal anastomosis (GIA) stapler, the artery is divided first, followed by the vein (Fig. 55–18). We prefer to use staples in this area because three rows of staples will be left on the vessel stump and are unlikely to be dislodged during subsequent dissection. In some instances clips may be needed. We prefer to use at least five clips on the patient side, when possible.
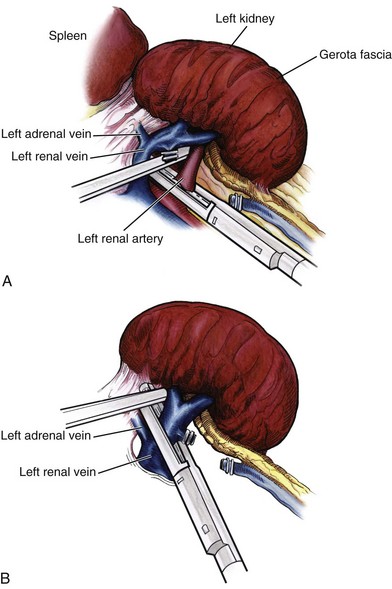
Figure 55–18 A, First, the renal artery is stapled using an endovascular gastrointestinal anastomosis (GIA) stapler. B, The renal vein is secured lateral to the adrenal vein with the GIA stapler. If clips are used on the gonadal or adrenal vessels, the surgeon must be careful to exclude them from the jaws of the stapler.
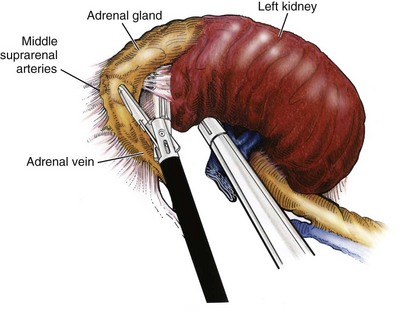
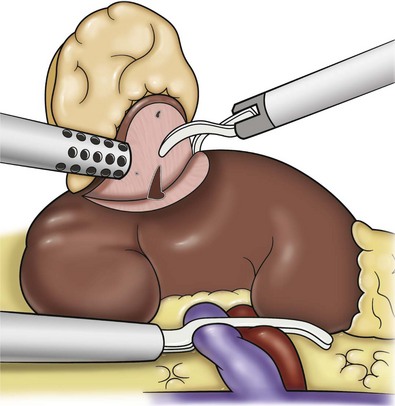
Isolation of the Upper Pole
Once the hilar vessels have been divided, the dissection continues posteriorly and superiorly to the upper pole. The adrenal gland is preserved in cases of simple nephrectomy by staying close to the upper pole (Fig. 55–19). This is accomplished by incising the Gerota fascia anteriorly, just above the hilum. Gerota fat is then gently peeled off circumferentially above the upper pole of the kidney. At this point during the dissection, it may be necessary to clip and transect the ureter. This allows the kidney to be rotated anteriorly above the liver (right) or spleen (left) to facilitate incision of the uppermost attachments under direct vision. In cases of extreme fibrosis, a subcapsular nephrectomy can be performed once the artery and vein have been controlled (Moore et al, 1998). Long, blunt instruments, such as the closed stapler or the 10-mm Ligasure Atlas (Valleylab, Boulder, CO) are particularly well suited for reaching and freeing the upper pole attachments.
Organ Entrapment and Extraction
The kidney can be removed intact or through morcellation. When morcellation is performed, the specimen should be placed into a sturdy entrapment sac (Urban et al, 1993). This minimizes the risk of rupture during mechanical morcellation of the tissue (Landman et al, 2000; Pautler et al, 2002). Using ring forceps and a Kocher clamp, the kidney and collecting system can be morcellated and removed in small pieces (Fig. 55–20). Alternatively, the kidney can be removed intact through an incision after placement into a sac (Fig. 55–21). The kidney can be worked out of an extended trocar site or Pfannenstiel incision. For trocar sites greater than 5 mm, it is helpful to place the closure sutures before removing the kidney. Once the sutures are placed, trocars can be reinserted for kidney entrapment and removal.
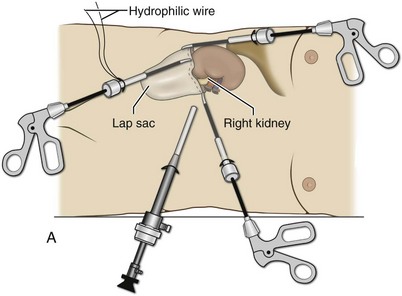
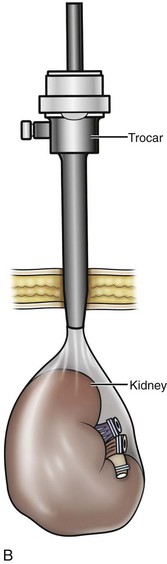
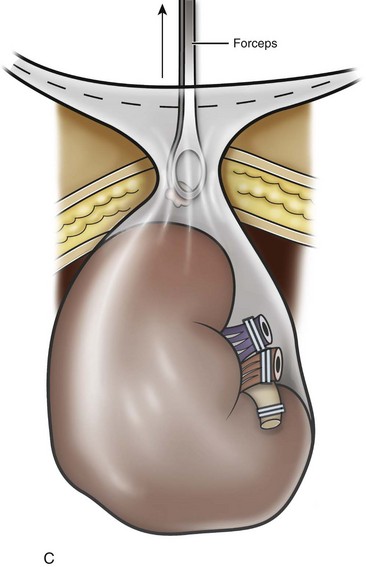
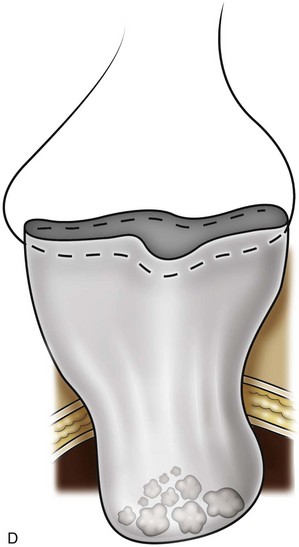
Figure 55–20 Removal of the morcellated specimen. A, The LapSac (Cook Urological, Spencer, IN) entrapment sac is introduced through the lateral 10-mm trocar site after passing a hydrophilic wire through the opening of the LapSac. The entrapment sac is then released within the abdomen. The wire facilitates opening the bag and placement of the specimen. A lateral 5- or 3-mm port may be necessary to assist with holding placement of the specimen inside the LapSac. B, Once the specimen is within the LapSac, the wire is removed, the bag cinched closed, and the opening withdrawn through the 10-mm trocar site. C, The entrapment sac is pulled tightly up against the abdominal wall, with two hands pushing part of the specimen to appear through the opening of the LapSac. After the site is carefully draped, manual morcellation with ring forceps or a Kelly clamp can be used. D, The entrapment sac is removed once the remaining specimen fragments are small enough to be extracted through the trocar site. Only the tissue visible from the opening is grasped. Blind passes into the bag may injure surrounding bowel segments.
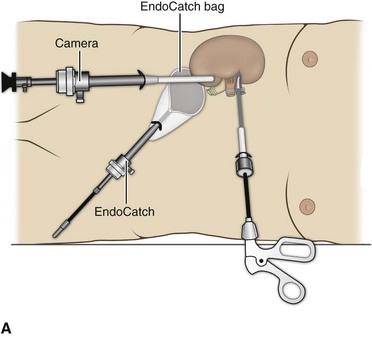
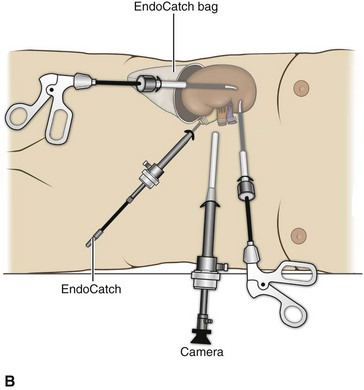
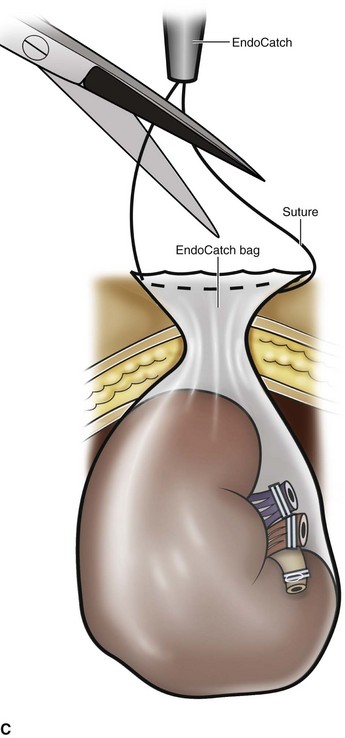
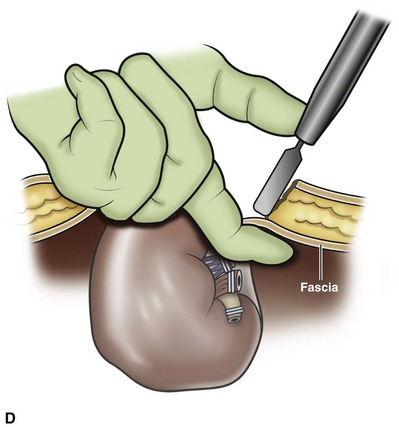
Figure 55–21 Removal of the intact specimen. A, The camera is moved to the lateral port site and an EndoCatch device is placed through the umbilical trocar site to entrap the specimen. B, Alternatively, the specimen may be extracted through a Pfannenstiel incision. To accomplish this, the EndoCatch device is brought in through a separate 10-mm suprapubic incision. C, The trocar is then removed, bringing the EndoCatch device with it through the trocar site, and the suture is cut and clamped. D, A 4- to 6-cm incision is made including one of the trocar sites. The surgeon’s finger protects the specimen and underlying structures from injury.
Postoperative Management
The orogastric tube is removed at the conclusion of the procedure. The patient can begin a diet as tolerated. The Foley catheter should be removed once the patient is comfortably ambulating. The patient is discharged when tolerating a regular diet. Unrestricted activity can usually be resumed according to the patient’s comfort.
Results
The postoperative results of laparoscopic nephrectomy are comparable to that of open surgery, with much less pain and shorter convalescence. Postoperative pain control requirements are approximately four times less than with traditional open incisions. Hospital stays have been decreased by 50%, and the time to full convalescence has been reported to be markedly less than with open removal. In early series, the mean operative times were greater than 300 minutes. However, with advances in techniques, experience, and equipment, current operative times have decreased dramatically (Kerbl et al, 1994a, 1994b; Nicol et al, 1994; Parra et al, 1995; Baba et al, 1996; Rassweiler et al, 1998).
Renal Cystic Disease
Renal cysts are extremely common and are present in more than one third of patients more than 50 years old (Laucks and McLachlan, 1981; Carrim et al, 2003). They rarely require surgical intervention, but indications include cyst-associated pain, infection, or obstruction (Hoenig et al, 1997; Wolf, 1998; Roberts et al, 2001; Doumas et al, 2004; Camargo et al, 2005). The increased use of cross-sectional imaging has also increased the detection of indeterminate renal cystic lesions and complex renal cysts, bringing an increased number to the attention of urologists. Classification schema have been developed to help clinicians make determinations regarding management, the most popular being the Bosniak system (Table 55–2) (Israel and Bosniak, 2005). Although these schema can be extremely useful, they are not diagnostic, and surgery may be required in some cases to exclude malignancy.
Table 55–2 Renal Cyst Classification Based on Updated Bosniak Criteria
| TYPE | DESCRIPTION | RECOMMENDED MANAGEMENT |
|---|---|---|
| I | A benign simple cyst with a hairline thin wall that does not contain septa, calcifications, or solid components. It measures water density and does not enhance. | No follow-up necessary |
| II | A benign cyst that may contain a few hairline thin septa in which “perceived”* enhancement may be present. Fine calcification or a short segment of slightly thickened calcification may be present in the wall or septa. Uniformly high-attenuation lesions (3 cm) (so-called high-density cysts) that are well marginated and do not enhance are included in this group. | No follow-up necessary |
| IIF | Cysts that may contain multiple hairline thin septa or minimal smooth thickening of their wall or septa. Perceived enhancement of their septa or wall may be present. Their wall or septa may contain calcification that may be thick and nodular, but no measurable contrast enhancement is present. These lesions are generally well marginated. Totally intrarenal nonenhancing high-attenuation renal lesions >3 cm are also included in this category. | Follow-up imaging required |
| III | “Indeterminate” cystic masses that have thickened irregular or smooth walls or septa in which measurable enhancement is present. These are surgical lesions, although some will prove to be benign (e.g., hemorrhagic cysts, chronic infected cysts, and multiloculated cystic nephroma), some will be malignant, such as cystic renal cell carcinoma and multiloculated cystic renal cell carcinoma. | Surgical treatment |
| IV | These are clearly malignant cystic masses that can have all the criteria of category III, but also contain enhancing soft tissue components adjacent to, but independent of, the wall or septum. These lesions include cystic carcinomas and require surgical removal. | Surgical treatment |
From Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology 2005;66:484–8.
Indications
First-line therapy and diagnosis of symptomatic renal cysts often involves percutaneous image-guided needle aspiration, with or without the use of a sclerosing agent, to prevent recurrence. If symptoms temporarily resolve and recur when fluid reaccumulates, this increases the likelihood that surgical treatment will be successful in resolving the pain (Rané et al, 2004). Caution should be exercised in use of cyst aspiration and sclerosing agents in peripelvic cysts, because fibrosis may occur (Wehle and Grabstald, 1986; Hulbert et al, 1988; Santiago et al, 1998; McDougall, 2000).
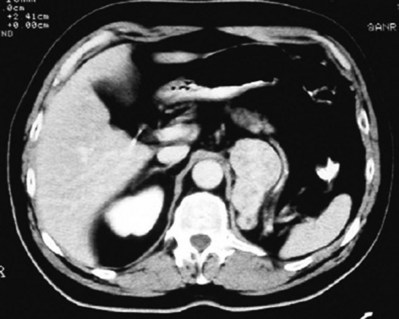
In addition to causing pain, cysts may compress the renal parenchyma or other adjacent organs, cause ureteral obstruction and obstructive uropathy, spontaneously bleed, cause hypertension, or become infected. Laparoscopic decortication or unroofing may be used to treat these cysts, which are typically simple in character (Fig. 55–22). Cysts with complex appearance, such as thickened septa, calcification, or enhancement (Bosniak class III-IV), may be explored and sampled laparoscopically to rule out renal cell carcinoma due to their increased risk of harboring malignancy (Cloix et al, 1996; Santiago et al, 1998). Options include cryoablation, enucleation, partial nephrectomy, or radical nephrectomy. If there is any question of cyst proximity to the collecting system, cystoscopy and placement of an open-ended ureteral catheter may be performed to ensure the integrity of the collecting system after cyst excision.
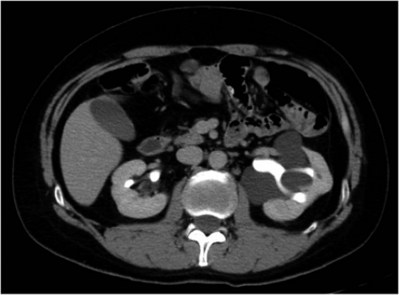
Figure 55–22 Axial CT scan in delayed phase after IV contrast administration, demonstrating peripelvic cysts in a patient who presented with left flank pain.
A subset of patients with autosomal dominant polycystic kidney disease (ADPKD) may develop cyst-associated pain. Laparoscopic cyst decortication, marsupialization, or unroofing can be of benefit to these patients by offering a minimally invasive treatment that is successful in relieving pain in up to 83% of cases (Lifson et al, 1998; Dunn et al, 2001; Lee et al, 2003). In patients with end-stage renal disease, bilateral synchronous laparoscopic nephrectomy may be performed in patients with enlarged, symptomatic, or infected kidneys (Gill et al, 2001; Rehman et al, 2001; Bendavid et al, 2004; Desai et al, 2008). Because an incision will be required to remove kidneys that are often quite enlarged, a hand port may be placed in the midline and used bilaterally to facilitate the dissection (Rehman et al, 2001; Jenkins et al, 2002).
Procedure
Depending on cyst location, a transperitoneal or retroperitoneal approach may be used as previously described. Intraoperative ultrasonography may be used to identify the cyst or cysts in question. It is usually easier to dissect out the cyst wall before evacuating fluid. The wall of the cyst can then be grasped and excised, cutting along the junction between the cyst wall and the renal parenchyma (Fig. 55–23). If suspicious lesions are noted in the base, biopsies may be taken using the 5-mm biopsy forceps. If no evidence of malignancy is seen, the remaining cyst wall may be fulgarated with either electrocautery or the argon beam coagulator. Care should be taken when ablating the surface, because inadvertent entry into the collecting system can easily occur (Cherullo et al, 1999). Moreover, these surfaces can be friable and prone to significant bleeding. Packing with hemostatic agents or suturing may be necessary. One should have a low level of suspicion for entry into the collecting system and, as such, a low threshold for leaving a drain (Fig. 55–24). If malignancy is noted, extirpative surgery or cryoablation may be used to treat the remainder of the lesion.
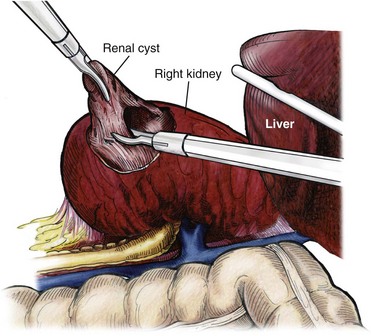
Figure 55–23 The cyst fluid is aspirated with a laparoscopic aspiration needle. After decompression of the cyst, the wall can easily be grasped and manipulated. The cyst is elevated with a grasper and scissors or ultrasonic shears to circumferentially excise the cyst wall. The edge of the cyst is carefully inspected, and biopsies are performed using the 5-mm laparoscopic biopsy forceps as needed.
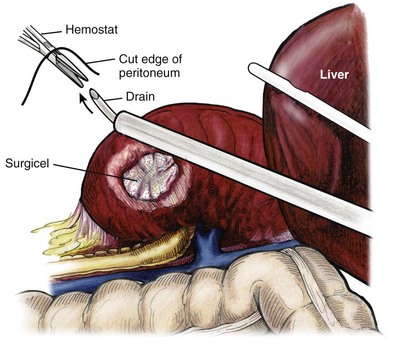
Figure 55–24 Drain placement after renal cyst excision. If the collecting system has been entered, it is closed and a drain placed. To insert the drain, a hemostat is passed through a small stab incision in the side and advanced into the abdominal cavity under direct vision. A drain is placed through a trocar site and advanced toward the open hemostat using the trocar to direct the drain. The colon is brought back over the kidney and attached to the sidewall to “reperitonealize” the kidney and drain.
When treating central or perihilar cysts, it may not be feasible to remove a large portion of the cyst wall. In these cases, it is helpful to place a pedicle of autologous fat into the defect to act as a wick (Nieh and Bihrle, 1993).
Results
Laparoscopic treatment of symptomatic renal cysts has been found to be effective in both decompression and pain control. A study evaluating treatment durability at a mean follow-up of 26 months demonstrated a 100% pain-free rate in patients treated for a solitary symptomatic renal cyst (Lifson et al, 1998). In a separate study with a mean follow-up of 60 months, 80% to 90% of patients have complete resolution of pain after laparoscopic cyst decortication. In patients with ADPKD, additional benefits of cyst decortication have been noted, including decreased blood pressure (Dunn et al, 2001; Lee et al, 2003). Recurrence of pain in this group is higher than with simple cysts, and durability is moderate (Brown et al, 1996). No significant changes in renal function were noted postoperatively.
Patients undergoing surgery for cystic renal disease are inherently a heterogeneous group, made up of patients with simple renal cysts, complex or indeterminate cysts, and ADPKD. Interpretation of incidence reports of renal cell carcinoma in these series must take this fact into account. That said, the reported incidence of renal cell carcinoma in cystic lesions is between 3% and 20% (Rubenstein et al, 1993; Lifson et al, 1998; Roberts et al, 2001; Limb et al, 2002).
Renal Biopsy for Medical Renal Disease
Histologic information is a key component in making treatment decisions and for prognosis in patients with proteinuria or unexplained renal insufficiency (Morel-Maroger, 1982; Gault and Muehrcke, 1983; Manaligod and Pirani, 1985). Although the modality of choice is typically ultrasound-guided percutaneous renal biopsy, laparoscopic biopsy may be preferred in certain situations, such as failed percutaneous biopsy, renal anatomic anomalies, risk of bleeding complications, morbid obesity, multiple renal cysts, or solitary kidneys.
Procedure
The patient is placed in the full-flank position with the table flexed to increase working space between the costal margin and the iliac crest. Retroperitoneal access is preferred and obtained using techniques described later in this chapter. Renal biopsy is accomplished with the use of two trocars. With blunt dissection, Gerota fascia is opened, and the lower pole of the kidney is exposed (Fig. 55–25A). In obese patients, intraoperative ultrasonography may be required to localize the kidney when copious retroperitoneal or perinephric fat is present. A 5-mm biopsy forceps is used to take samples of cortical tissue; hemostasis is achieved with the argon beam coagulator; and adjunctive hemostatic measures are used as necessary (Fig. 55–25B).
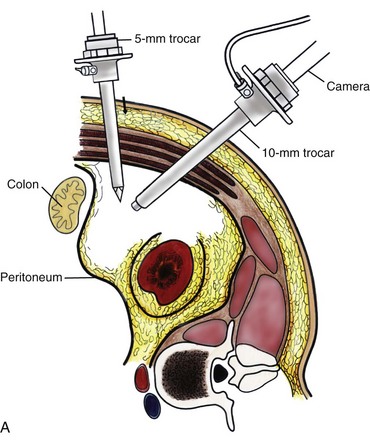
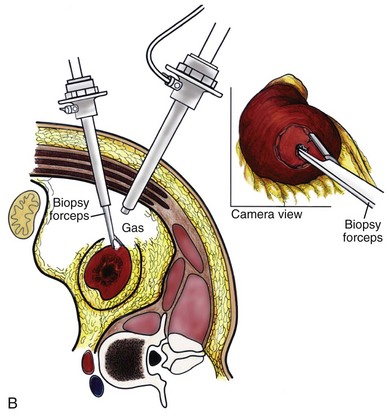
Figure 55–25 A, After establishing a working space, a 5-mm trocar is placed under direct vision. The working instruments are passed through this port. The camera can be used to assist with dissection and is frequently cleaned to maintain adequate visualization. B, Gerota fascia is opened with the use of the scissors. A 5-mm two-tooth laparoscopic biopsy forceps is used to take two to three samples from the lower pole of the kidney.
Results
A multi-institutional series reporting on outcomes of laparoscopic renal biopsy in 74 patients over 9 years showed a mean operative time of 123 minutes, mean estimated blood loss of 67 cc, and discharge within 24 to 48 hours unless other preexisting medical conditions required a longer stay (Shetye et al, 2003). Tissue obtained from 96% of patients was adequate for diagnosis, with a complication rate of 13.5%. The authors concluded that laparoscopic renal biopsy may be performed safely with a high success rate, and, that as experience grows, the complication rate and operative duration would likely decrease. In another series of 17 patients, using balloon dilatation to create the working space demonstrated 100% success in obtaining renal tissue adequate for diagnosis. Mean operative time was 35 minutes (excluding anesthesia time), complication rate was 11%, and 15 of 17 patients were discharged within 24 hours. In published series of laparoscopic renal biopsy, hemorrhage is the most common complication. Caution should be used in resumption of anticoagulation in patients who require it postoperatively. If signs or symptoms of postoperative anemia or hypovolemia occur, a low threshold should be used to evaluate the patient with computed tomography (CT).
Pyelolithotomy, Nephrolithotomy, and Ureterolithotomy
Advances in stone therapy have made pyelolithotomy, nephrolithotomy, and ureterolithotomy almost obsolete. However, a select subgroup of stone patients will benefit from these approaches. Depending on local demographics and incidence of stone disease, the indications for laparoscopic stone extraction may vary but primarily include patients with renal stones who are undergoing simultaneous laparoscopic pyeloplasty (Ball et al, 2004; Stein et al, 2008). In these patients, a flexible cystoscope may be passed through one of the trocars to remove stones. A laparoscopic grasper may also be used to remove stones, but patients should always undergo a complete nephroscopy to ensure stone clearance. Other patients who may benefit include those with unusual anatomy (pelvic kidney) (Chang and Dretler, 1996; Harmon et al, 1996; Hoenig et al, 1997), stones resistant to fragmentation (Jordan et al, 1997), and failed extracorporeal shock wave lithotripsy (ESWL), ureteroscopy, or percutaneous procedures.
Treating stone disease in ectopic kidneys can be particularly challenging. Ectopic kidneys frequently have anomalous vasculature, and therefore access into the collecting system carries a higher risk of bleeding. Laparoscopy may be used to assist in the placement of percutaneous access to minimize the risk of bleeding complications (Eshghi et al, 1985; Tóth et al, 1993; Troxel et al, 2002).
A retrospective comparative study of 12 patients who underwent percutaneous nephrolithotomy (PCNL), and 16 patients who underwent laparoscopic retroperitoneal pyelolithotomy, revealed no benefit to the laparoscopic approach. Laparoscopy was also associated with longer operative time, longer recovery, inferior cosmesis, and required more advanced skills (Goel and Hemal, 2003). The authors concluded that PCNL should remain the gold standard for treating large renal stones, unless the patient is undergoing surgery for concomitant pathology.
Robotic approaches to stone disease have also been described for the treatment of staghorn calculi, with or without simultaneous pyeloplasty. In a study of 12 patients undergoing robotic extended pyelolithotomy, 11 were stone free after surgery as assessed by postoperative CT scan. Mean operative time was 158 minutes, with mean estimated blood loss of 100 cc (Badani et al, 2006).
Laparoscopic anatrophic nephrolithotomy has been reported in animal models and small case series (Kaouk et al, 2003; Simforoosh et al, 2008). Initial reports are encouraging, but, to date, renal functional outcomes in humans have not been evaluated with nuclear renal scans. Further studies are required to assess the feasibility of this procedure.
Large or impacted ureteral stones may be managed laparoscopically with ureterolithotomy (Fig. 55–26). In a series of 93 patients undergoing this procedure, successful stone removal was achieved in 85 patients, with eight failures due to inability to locate the stone (Gaur et al, 2002). Mean operative time was reported as 80 minutes, and in many of the patients, the ureter was left open, leading to shorter operative time but more complications with prolonged urinary leakage. Other authors have also reported this approach for impacted or large ureteral stones (Hemal et al, 2003). In this series of 31 patients, success was reported in all cases, no stent was used, and closure of the ureterolithotomy in all cases resulted in a prolonged urinary leakage rate of 6.5% at 48 hours.
Nephropexy
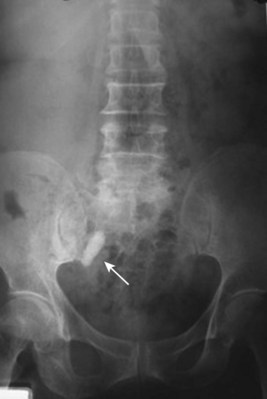
Renal ptosis, although rare, is a real cause of chronic flank or upper abdominal pain. The precise origin of symptoms is unknown, but is likely secondary to either transient ischemia or urinary obstruction (Moss, 1997). It is usually defined as the descent of the kidney by more than two vertebral bodies, but before a definitive diagnosis is made, objective proof that is associated with pain should be obtained. The typical patient with a ptotic kidney is a young, thin female who complains of pain while in an upright position. Supine and erect intravenous pyelography (IVP) can be used for diagnosis, with the finding of interest being descent of the symptomatic kidney by two vertebral bodies (Fig. 55–27). Nuclear imaging may also quantify obstruction of blood flow or drainage in the upright position. Color Doppler sonography in both the supine and upright positions can also be used to evaluate differential blood flow. If a ptotic kidney is present, the expected finding would be diminished blood flow while in the erect position. Before surgical repair, obstruction, decreased blood flow, or significant descent correlating with pain should be documented.
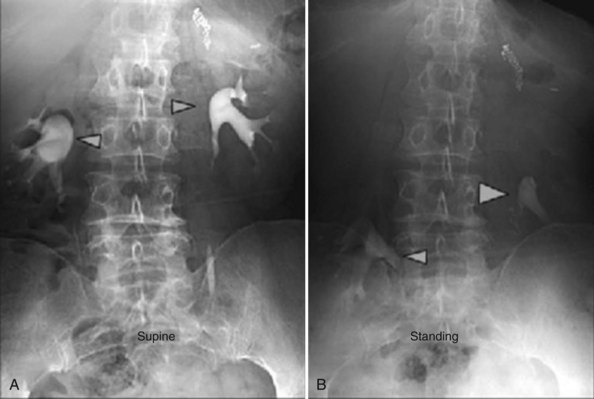
Figure 55–27 Intravenous pyelogram demonstrating bilateral ptotic kidneys in the supine (A) and standing (B) positions.
(From El-Moula MG, Izaki H, Kishimoto T, et al. Laparoscopic nephropexy. J Laparoendosc Adv Surg Tech A 2008;18:230–6.)
Procedure
Surgical repair is performed using either a standard transperitoneal or retroperitoneal approach to fully mobilize the affected kidney and expose the fascia overlying the psoas and quadratus lumborum muscles (Chueh et al, 2002; Matsui et al, 2004) (Fig. 55–28). Beginning at the upper pole, interrupted sutures are placed to secure the lateral edge of the renal capsule to the fascia overlying the muscle (Fig. 55–29). Sutures may also be placed between the anterior renal capsule and the parietal peritoneum for additional support. Using sutures with preplaced Lapra-Ty clips (Ethicon Endosurgery, Cincinnati, OH), the initial pass is made through the fascia or peritoneum, and a second clip is placed on the suture after the pass through the kidney. An alternative technique describes the use of tension-free vaginal tape to secure the kidney (Hübner et al, 2004). By placing the needles such that the tape is passed around the lower pole of the kidney and out through the abdominal wall, the kidney is secured. Sometimes this is found in conjunction with a ureteropelvic junction obstruction that should be adressed simultaneously (Boylu et al, 2009).
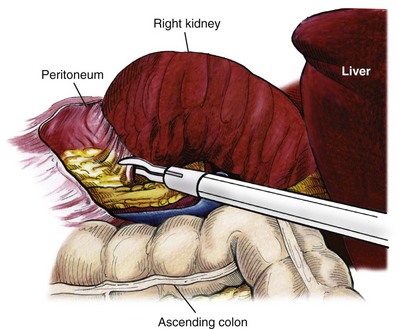
Figure 55–28 The kidney is stripped of overlying Gerota fascia down to the surface of the renal capsule. All remaining attachments are divided, allowing full mobility for repositioning.
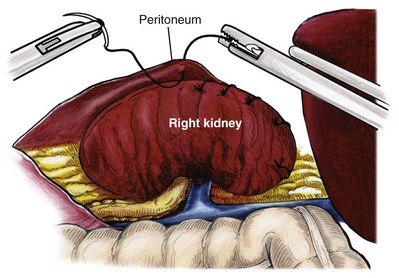
Figure 55–29 Nephropexy. Once the kidney is free of lateral and posterior attachments, multiple 2-0 sutures are placed into the capsule and the lateral edge of the fascia overlying the abdominal wall. Sutures may also be placed between the anterior renal capsule and the parietal peritoneum for additional support.
Results
A retrospective study of 30 patients undergoing laparoscopic nephropexy with a median follow-up of 5.9 years demonstrated improvement in all patients, 11 with complete relief of symptoms (Plas et al, 2001). Significant improvement in renal function was measured by renal scan in 9 of 10 patients undergoing the study postoperatively. Two patients developed a recurrent ptotic kidney with greater than 5 cm of descent documented by IVP. An additional study of 48 patients, with a median follow-up of just more than 8 years, revealed that 94% of patients had no signs of significant ptosis on postoperative imaging, and 91% of patients had improvement in their pain symptoms (Gözen et al, 2008). Mean operative time was 95 minutes, and mean estimated blood loss was 50 cc. Patient satisfaction was high, and the authors concluded that laparoscopic nephropexy provided a minimally invasive approach to these patients with good long-term clinical outcomes.
Calyceal Diverticulectomy
Patients with symptomatic calyceal diverticula containing stones can be managed laparoscopy. ESWL and ureteroscopy may be employed, but due to infundibular stenosis, stone clearance rates are low (Jones et al, 1991; Pang et al, 1992; Stream et al, 1992). The principles of treatment include the removal of stones and widening of the infundibular stenosis to prevent urine stasis or ablation of the diverticula cavity. In the past, symptomatic calyceal diverticula have been managed with partial nephrectomy, with marsupialization and fulgaration of the diverticulum, and occasionally simple nephrectomy. More recently, percutaneous treatment has been used, but stones or symptoms may recur if the cavity is not fully ablated (Donnellan et al, 1999). A laparoscopic approach may be indicated with large, peripheral diverticula, or for centrally located diverticula with proximity to the renal hilum.
Procedure
The location of the diverticula will be the primary driver for choosing either a transperitoneal or retroperitoneal approach. Once the renal dissection is completed, locating the diverticulum may prove difficult. The presence of dense adhesions on the surface of the kidney overlying the divericulum may be present along with a “dimpling” effect on the renal capsule. Intraoperative ultrasonography may also be used to aid in the location of the divertiulum or to confirm a suspected location. Once localized, the overlying parenchyma is incised and opened to expose the diverticulum, which is subsequently opened with cautery. Stones may then be removed, and the argon beam coagulator or monopolar cautery device can be used to fulgarate the diverticular lining. The collecting system is closed with suture, and perirenal fat may be placed into the defect to further decrease the likelihood of recurrence. A drain is usually left in place.
Results
Published reports on laparoscopic calyceal diverticulectomy are limited to small series and case reports. Excellent results have been reported by numerous authors, demonstrating the definitive nature of this treatment modality (Miller et al, 2002; Gluckman et al, 1993; Ruckle and Segura et al, 1994; Harewood et al, 1996; Wolf, 2000; Canales and Monga, 2003; Wyler et al, 2005). Probably due to the rarity of this surgery, complications are also rare and have not been reported yet in the literature.
Nephrolysis
Chyluria is caused by lymphatic rupture or fistulous connection into the pyelocalyceal system. It is a rare problem worldwide but is commonly seen in tropical countries when filariasis (Wuchereria bancrofti or Brugia malayi), the most common cause, is endemic (Tandon et al, 2004). Rarely, schistosomiasis may also be a cause of chyluria, and nonparasitic chyluria is rare. Other reported etiologies include tuberculosis (Wilson and White, 1976), idiopathic lymphorenal fistula (Eisner et al, 2009), prior surgery (Kim and Joudi, 2009), pregnancy (Onyeije et al, 1997), thoracic duct obstruction (Garrido et al, 1995), mesenteric adenitis (Cohen et al, 1984), renal vasculitis (El-Reshaid et al, 1998), and metanephric adenoma (McNeil et al, 2008).
Diagnosis
Patients typically present with milky white urine and may have nephrotic range proteinuria. Initial evaluation includes urinalysis and culture, urine for chyle, and complete blood count to check for eosinophila. Evaluation to localize the fistula may include cystoscopy with retrograde pyelogram, CT, magnetic resonance imaging (MRI), or lymphangiography (Fig. 55–30).
Treatment
Chyluria is often self-limited, and many patients may be managed conservatively. In filariasis-associated cases, this involves treatment with a course of diethylcarbamazine (DEC) in combination with a low-fat diet (Tandon et al, 2004). Retrograde instillation of silver nitrate or povidone iodine into the collecting system as a sclerosing agent has also been described, and these agents are commonly used as first-line treatment with comparable success rates of approximately 80% (Dalela et al, 2004a; Goel et al, 2004). When conservative management fails or if cases are particularly severe, surgical intervention is undertaken.
Procedure and Results
Nephrolysis involves the complete mobilization of the kidney and skeletonization of the renal hilar vessels and upper ureter with ligation of the lymphatic channels. The procedure may be performed laparoscopically by either a transperitoneal or retroperitoneal approach and ensures complete lymphatic dissociation of the affected kidney (Chiu et al, 1995, Gomella et al, 1998). The use of an omental wrap around the hilum has also been described to provide an additional barrier against recurrence (Dalela et al, 2004b). Authors commonly performing this procedure report excellent success rates—approaching 100%.
Radical Nephrectomy
Indications for Malignancy
Laparoscopic approaches to malignancy have been performed for over 20 years. The indications for radical nephrectomy are similar to those for open surgery. Kidneys with tumors as large as 25 cm have been successfully removed laparoscopically, and cytoreductive nephrectomy has been performed in patients with metastatic disease (Walther et al, 1999). Moreover, tumors with low- level caval thrombi have also been removed (Martin et al, 2008).
Transperitoneal
Procedure
Access and trocar placement are similar to what has been described for simple nephrectomy. With larger masses, caval involvement or organ invasion, additional trocars or a hand port may be needed. The procedure for laparoscopic radical nephrectomy (LRN) is essentially identical to laparoscopic simple nephrectomy. The main distinguishing feature is that Gerota fascia is left intact during dissection. The adrenal may be removed en bloc with the kidney when indicated (Fig. 55–31A). Alternatively, this fascia is opened over the upper medial aspect of the kidney when adrenal-sparing nephrectomy is performed (Fig. 55–31B). Suspect lymph nodes may be removed, and a full hilar or retroperitoneal dissection can be carried out if deemed necessary. Excision of part of adjacent muscle or involved organs, such as the pancreas, liver, spleen, and bowel, has also been reported (Molina et al, 2004).
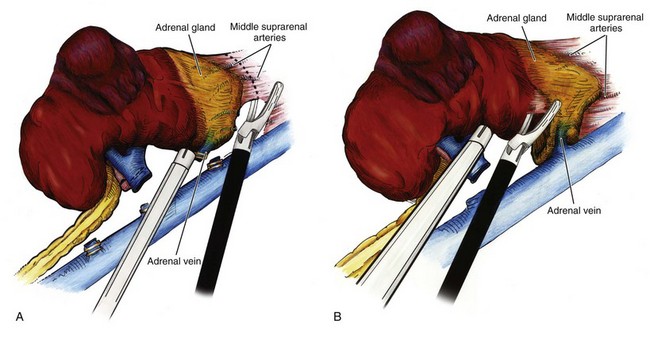
Figure 55–31 A, Inclusion of the adrenal gland during right laparoscopic radical nephrectomy can be readily accomplished using ultrasonic or bipolar shears to control the multiple arterial branches to the adrenal gland. Inferior retraction of the specimen facilitates exposure of this surgical plane. B, Adrenal-sparing, right radical nephrectomy. Use of a blunt instrument above the hilum to put anterior and inferior traction on the kidney helps to expose the correct plane and place the connective tissue on stretch. Ultrasonic or bipolar shears are again useful to avoid any bleeding that may be encountered in this plane.
Results
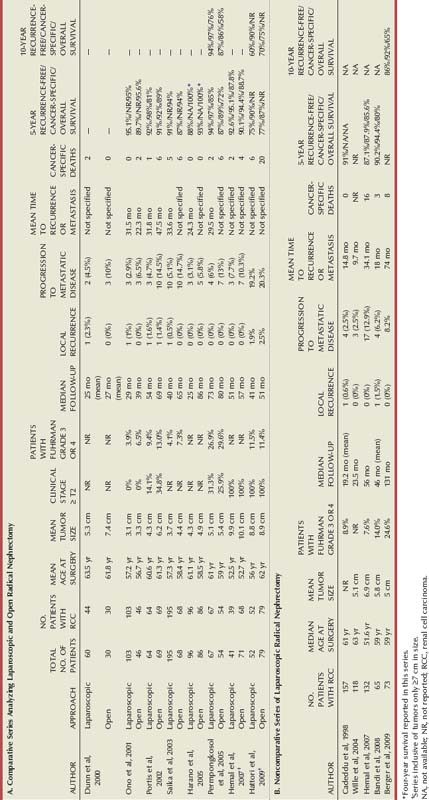
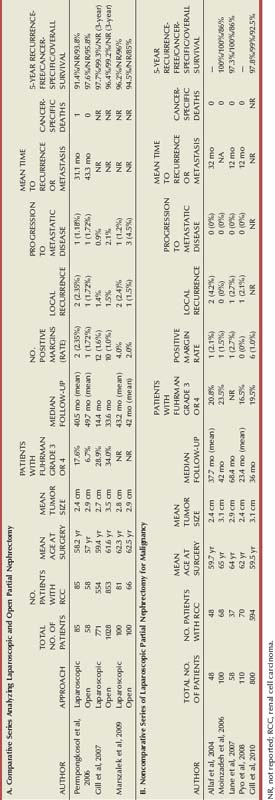
Long-term cancer-specific survival data is now widely available from multiple centers around the world that perform laparoscopic radical nephrectomy (Table 55–3). Five- and 10-year outcomes show oncologic equivalence to open radical nephrectomy in treatment of renal cancer. Indeed, LRN has become the standard of care for most renal malignancies.
A multi-institutional study from centers performing LRN compared the surgical and disease-specific outcomes between open and laparoscopic radical nephrectomy, with long-term follow-up (Portis et al, 2002). Median follow-up was 54 months, and recurrence-free survival was 91% and 92%, respectively, for the two groups at 5 years. The 5-year cancer-specific survival was 98% for the laparoscopic cohort and 92% for the open cohort.
A comparative analysis of 67 patients undergoing LRN, with 54 patients undergoing open radical nephrectomy, evaluated perioperative and oncologic outcomes (Permpongkosol et al, 2005). The LRN group showed a longer mean operative time (256 vs. 193 minutes). However, this finding likely reflects the learning curve for laparoscopy, because the first 34 patients and last 33 patients in the LRN group had a significant operative time difference. Complications occurred in 15% of patients in the LRN and open groups, and blood transfusions were required in 6% and 20% of the patients, respectively. Most importantly, the calculated disease-free survival rates for laparoscopic and open radical nephrectomy were 95% and 89%, respectively, at 10 years. The actuarial survival rates for laparoscopic and open radical nephrectomy were 87% and 75%, respectively, at 10 years. These differences were not found to be statistically significant.
Most recently, 10-year oncologic outcomes data following LRN were reported. Recurrence-free, cancer-specific, and overall survival rates were 86%, 92%, and 65%, respectively, at 10 years postnephrectomy (Berger et al, 2009). Of 73 patients undergoing LRN, no patient developed local recurrence, and 6 (8.2%) developed metastatic disease at a mean time to recurrence of 74 months. Although outcomes in this study were not compared with an open radical nephrectomy cohort, results are quite comparable to those for open surgery.
Perioperative outcomes in contemporary groups undergoing laparoscopic and open radical nephrectomy have also been extensively studied. In a series of 54 laparoscopic and 34 open radical nephrectomies, no differences in age, body mass index (BMI), American Society of Anesthesiologists (ASA) score, tumor size, specimen weight, or operative time were noted (Gill et al, 2000). The laparoscopic approach was shown to have less blood loss, shorter hospital course, lower analgesic requirement, and shorter return to convalescence. Complications were noted in 13% of patients in the laparoscopic group and 24% of patients in the open group. Similar findings have been previously reported by multiple authors (Kerbl et al, 1994a; McDougall et al, 1996; Hemal et al, 2007).
Retroperitoneal
Procedure
The patient is positioned, and trocars are placed for retroperitoneal access as described earlier in the chapter. After identifying the psoas muscle and tendon, medial dissection in this plane will reveal the ureter. Elevation of the ureter will allow visualization and subsequent elevation of the lower pole of the kidney. This will place the main renal vessels on stretch, facilitating their dissection. The arterial pulsation may be indirectly visualized through overlying connective tissue, and just as with the transperitoneal approach, gentle layer-by-layer dissection with the suction-irrigator will allow the renal vessels to come more directly into view. Use of the right-angle dissector will allow the artery to be circumferentially freed from the surrounding tissue, and the endovascular stapler or clips are used to divide the artery and vein sequentially. When operating on the left kidney, a lumber vein will typically require dissection, ligation, and division to allow unencumbered access to the main hilum. Care must be taken to continuously reorient to anatomic relationships to ensure that the inferior vena cava is not mistakenly identified as the renal vein. The kidney may be removed intact or after morcellation, as previously described.
Results
When compared with transperitoneal laparoscopic radical nephrectomy, outcomes of the retroperitoneal approach are quite similar with regard to complication rates, analgesic requirements, hospital course, and return to convalescence.
A randomized study comparing transperitoneal and retroperitoneal laparoscopic approaches was performed in 102 patients (52 transperitoneal, 50 retroperitoneal) with a mean tumor size of 5 cm (Desai et al, 2005). No difference was seen in blood loss, narcotic requirement, hospital stay, or complication rate. However, there was a significant difference noted in operative time, which favored the retroperitoneal approach (150 vs. 207 minutes). A second randomized study of 40 patients compared the number and size of trocars, pathologic stage, blood loss, operative time, complication rate, and hospital stay (Nambirajan et al, 2004). No statistical difference was noted in any of these outcomes, including operative time.
Hand-Assisted
Procedure
The patient is positioned, and the hand port and trocars are placed as previously described. The steps in hand-assisted laparoscopic nephrectomy (HALN) are similar to those for pure laparoscopic surgery, but the nondominant hand is used throughout for retraction and dissection. To incise the white line of Toldt, the nondominant hand retracts the colon medially while the dominant hand uses an endoscopic scissors to divide the attachments (Fig. 55–32). The irrigator-aspirator is then used to help identify and dissect the correct plane posterior to the large bowel mesentery and anterior to Gerota fascia. The surgeon’s hand and fingers may be used to simultaneously place lateral traction on the kidney and medial traction on the bowel, helping to demonstrate the correct plane. For a left nephrectomy, the hand may also be used to gently retract the spleen and pancreas medially while the lienorenal attachments are divided. Similarly, on the right, the hand is used to retract the liver anteriorly, exposing the upper pole and facilitating its dissection.
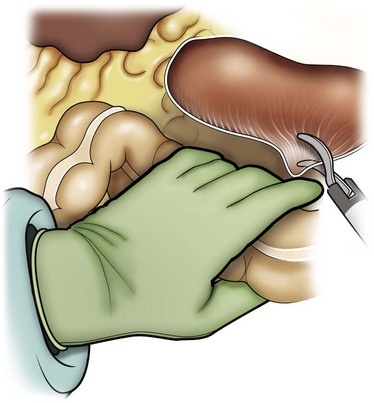
Figure 55–32 The nondominant hand is used to retract the colon medially and to dissect tissue planes, while the dominant hand uses endoscopic scissors to divide colon attachments.
After the colon is sufficiently mobilized, the psoas muscle is identified, which will allow the ureter to be elevated. On the left, the gonadal vein is typically elevated in the packet along with the ureter, but on the right, the gonadal vein is reflected medially. With the ureter elevated, the hand can bluntly dissect and elevate the entire kidney off the psoas muscle, and the ureter is then followed up to the renal hilum. The fingers are then used to place anterolateral traction on the kidney, while the thumb pushes the bowel and mesentery medially. The hilum should begin to come into view at this point, and the irrigator-aspirator can be used to gently dissect the overlying connective tissue while the hand is used to keep the vessels on stretch. Once the vessels are sufficiently skeletonized, the endovascular stapler or clips are used to ligate and divide the artery and vein sequentially (Fig. 55–33A and B). The lateral and superior attachments may then be divided, using a Ligasure device or harmonic scalpel while the hand keeps them on traction. The hand should not be used to dissect the adrenal gland free from the upper pole of the kidney, because this will typically result in bleeding. Ultrasonic energy, Ligasure, or bipolar cautery may be used to divide the fragile attachments between the kidney and the adrenal gland, just as with pure laparoscopy.
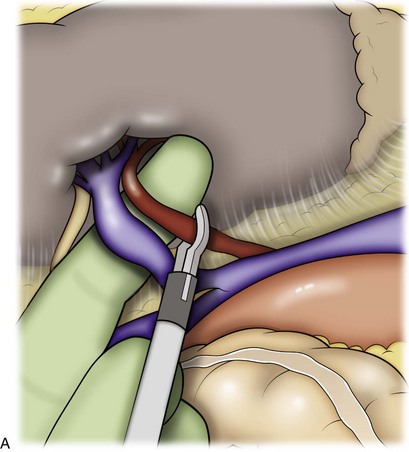
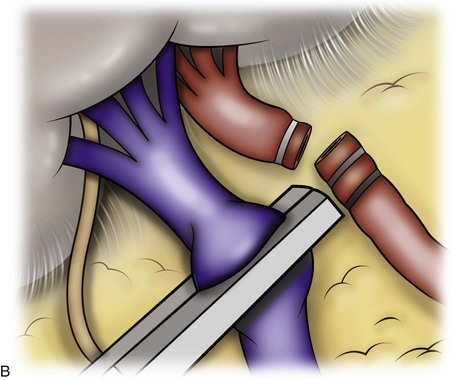
Figure 55–33 A, With the renal hilum on stretch and the bowel retracted medially to expose the vessels, the fingers can be used to palpate the renal artery and guide a stapler or clip applier to secure and divide the artery. B, Once the artery is divided, the renal vein is freed circumferentially and divided with an endovascular stapler.
A rolled laparotomy sponge is often placed through the hand port at the beginning of the procedure to assist with retraction, absorb blood, and to allow the surgeon to hold pressure if needed. In addition, the tip of the irrigator-aspirator can be placed into the compressed sponge to facilitate suction when needed. It is critical to remember removal of the sponge at the end of the case. The previously made hand-port incision allows rapid removal of sponge and the intact specimen. However, hand-port metastases have been reported, and it is recommended to place the specimen in a removal device before extraction to prevent any possibility of direct contact with wound edges.
The inserted hand can also facilitate closure of trocar sites greater than 10 mm with a suture-passing device. Wound complications such as hernias and infections have been reported at the hand-port site, with hernias typically presenting 3 months or more after surgery and with an overall 4% incidence (Okeke et al, 2002; Wolf, 2005). Care must be taken to copiously irrigate and close the hand-port incision, ideally closing the peritoneum separately from the fascia to minimize chance of hernia.
Results
Most outcomes are comparable between pure laparoscopy and the hand-assisted technique including efficacy, operative times, complication rates, narcotic requirements, length of hospital stay, and return to convalescence. Some comparative studies between laparoscopic and hand-assisted techniques have demonstrated that the hand-assisted procedures can require shorter operative times, but patients had more abdominal pain and wound complications (Nelson and Wolf, 2002).
Comparative studies between HALN and standard laparoscopic approaches have shown that choice of approach does not impact on oncologic outcome. Gabr and colleagues (2009) studied and compared 147 patients who underwent standard laparoscopic nephrectomy and 108 who underwent hand-assisted laparoscopic nephrectomy. Patients were followed for a mean of 35.2 months, and multivariate analysis showed that the approach did not impact recurrence-free, cancer-specific, or overall survival. A multi-institutional study of 95 patients undergoing HALN evaluated the impact of tumor size on outcome (Stifelman et al, 2003). Patients were grouped into those with tumors larger than 7 cm and those with tumors smaller than 7 cm. Short-term outcomes at mean follow-up of 12 months were not different between the two groups, including positive margin rate, local recurrence, and metastasis. An additional short-term outcome study investigating a series of 60 patients undergoing HALN for T1 or T2 tumors demonstrated no recurrence or trocar site seeding at mean follow-up of 11 months (Patel and Leveillee, 2003).
Radical Nephrectomy: Special Considerations
Large Tumors
Experience with laparoscopy for large tumors (>7 cm) has grown substantially as confidence with renal laparoscopy has increased (Steinberg et al, 2004; Hemal et al, 2007; Berger et al, 2008; Rosoff et al, 2009). Large tumors present several surgical challenges. The bulk of the mass can decrease working space and alter normal anatomic landmarks. This can result in disorientation with potential injury to surrounding structures. Continuous intraoperative reference to preoperative imaging as well as use of ultrasonography is helpful. Flexible endoscopes may be used to see around structures. The weight of large tumors may cause the surgeon to apply additional force for manipulation, potentially resulting in tumor rupture. It may be beneficial to use a hand port in these instances or additional trocars. Consideration may also be given to lymphadenectomy with larger tumors as indicated.
En Bloc Stapling
En bloc stapling of the renal hilum has been reported from several centers. An evaluation of 80 patients with mean follow-up of 35.2 months after either open or laparoscopic radical nephrectomy, with routine use of en bloc hilar stapling, demonstrated no clinical evidence of arteriovenous fistula (White et al, 2007). Half of the patients also underwent CT arteriography at a minimum interval of 12 months postoperatively, and no patient had radiographic evidence of arteriovenous fistula. Another study evaluated patient outcomes in 433 patients undergoing laparoscopic radical nephrectomy or nephroureterectomy, of whom 26 (6%) underwent en bloc stapling only when hilar dissection was deemed “difficult” by the surgeon (Rapp et al, 2004). No cases of arteriovenous fistula were noted at a mean follow-up of 26 months. However, the authors noted that arteriovenous fistula may be a more long-term complication after en bloc hilar stapling, and thus longer follow-up is needed to properly assess these patients.
Port-Site Recurrence
Since its inception, a concern for port-site seeding and recurrence continues to be raised for performance of laparoscopic surgery for urologic malignancy. In an international survey of 20 centers performing 2604 laparoscopic radical nephrectomies, no port-site seeding was reported (Micali et al, 2004). A recent review of all reported cases of port-site seeding in laparoscopy for urologic malignancy revealed a total of 28 cases. The majority involved aggressive upper tract transitional cell carcinoma, and six involved renal cell carcinoma (Eng et al, 2008). The etiology of port-site recurrence is thought to be multifactorial and related to tumor aggressiveness, immune status of the patient, local wound factors, and surgical technique. The effect of pneumoperitoneum, aerosolization of tumor cells, insufflation gas type, and laparoscopic wound closure techniques have been studied by multiple authors and is noncontributory (Ikramuddin et al, 1998; Tsivian et al, 2000; Gupta et al, 2002; Burns et al, 2005; Halpin et al, 2005; Jingli et al, 2006). As with open surgery, the most common etiology is technical error and associated tumor spillage. Animal studies demonstate that direct contact between tumor and port-site enhances tumor growth (Bouvy et al, 1996), and the use of an impermeable specimen retrieval bag is recommended in all cases. Although the overall incidence of port-site metastasis is low—estimated at 0.09% to 0.18% (Micali et al, 2004; Rassweiler et al, 2003)—care should be taken in specimen handling and extraction to help minimize risk factors over which the surgeon has the most control.
Specimen Extraction
An area of controversy surrounding specimen extraction has been morcellation due to the concerns for inadequate pathology and staging, peritoneal or port-site seeding, bag rupture, and recurrence. Although there is an advantage in terms of shorter incision length, there has been no benefit demonstrated in postoperative analgesic requirement (Hernandez et al, 2003). A multi-institutional study of the safety and efficacy of specimen morcellation in 188 patients with clinical stage T1 or T2 renal cell carcinomas revealed 11 patients with recurrent disease (10 metastatic to lungs or viscera and 1 patient with port-site, renal fossa, and lymph node recurrence) (Wu et al, 2009). This demonstrates that mechanical morcellation may be safely performed in selected patients. However, the ability to properly stage the patient remains in question, with studies demonstrating conflicting results. The feasibility of pathologic evaluation has been studied in comparative fashion by evaluating both fresh and formalin-fixed specimens before and after morcellation (Landman et al, 2000). No alteration in determination of histology, grade, or local invasiveness of tumor was seen. Only specimen size could not be assessed following morcellation. A separate study on 23 morcellated specimens concluded that pathologic tumor stage in both renal cell and transitional cell carcinoma is severely limited by morcellation and must rely partially on diagnostic imaging for lesion size, capsule, and renal vein involvement (Rabban et al, 2001). Additionally, the incidence of pathologic stage T3a tumors was evaluated retrospectively in a series of 1781 patients and found to be 7.2% overall (Granberg et al, 2007). Imaging in the overwhelming majority of these patients did not predict stage pT3a disease. The authors concluded that without imaging that can more reliably predict fat invasion, accurate staging would be difficult if morcellation is performed.
If the specimen is to be morcellated, the surgeon should strictly adhere to proper technique including the use of a purpose-built sac, adequate draping, and change of gowns, gloves, and instruments after morcellation. The LapSac (Cook Urological, Spencer, IN) has been shown to be impermeable to bacteria and tumor cells, even after its use for morcellation (Urban et al, 1993). The sac is prepared by passing a moistened hydrophilic wire alternating through every third hole in the sac, which is then rolled from the bottom up and passed through a 12-mm trocar site. The trocar is replaced leaving the wire and drawstrings outside the trocar. Graspers are used to place the specimen in the sac, which is held open by the wire, and the wire is removed (Wakabayashi et al, 2003). The drawstrings are grasped and brought through the periumbilical incision along with the neck of the sac, which is held tightly against the abdomen. Enlarging the trocar site by 1 cm will allow small amounts of tissue to protrude through the mouth of the sac. The morcellation process is performed with a ring forceps, working with alternating bites on the protruding tissue. Deep passes with the forceps should be avoided to prevent unintentional incorporation of bowel into the forceps. Pneumoperitoneum should also be maintained during the process to allow monitoring of the sac intracorporeally to avoid injury to structures resting against the sac or sac perforation.
Lymphadenectomy
Lymphadenectomy at the time of nephrectomy, open or laparoscopic, remains controversial. It has been shown that presence of two or more adverse pathologic predictors (grade, sarcomatoid features, tumor size, stage, and necrosis) results in a higher likelihood of lymph node metastasis (Blute et al, 2004). In addition, patients with preoperatively or intraoperatively suspicious lymph nodes have been shown to have improved survival (median of 5-month benefit) when undergoing a lymph node dissection compared with those who did not (Pantuck et al, 2003). A retrospective study of 50 patients undergoing LRN alone versus 50 undergoing LRN with laparoscopic lymph node dissection (LND) determined that 10% of the patients undergoing LND had positive nodes (Chapman et al, 2008). All patients were preoperatively node negative by cross-sectional imaging evaluation, and those with positive nodes all had high-grade lesions, stages T3 or T4. However, a randomized study comparing groups undergoing radical nephrectomy, with or without lymphadenectomy at the time of surgery, has demonstrated no survival benefit of lymph node dissection in patients with clinically negative lymph nodes (Blom et al, 2009).
It is clear that not every patient with renal cell carcinoma requires lymphadenectomy. However, a subset of patients with suspicious lymph nodes may derive benefit from lymphadenectomy. Of note, there is no consensus on the extent of node dissection to be performed. The additional group that may benefit includes those with higher-stage tumors in the absence of suspicious lymph nodes, although survival data to support lymphadenectomy in this population is lacking.
Local Recurrence
The incidence of isolated local recurrence after nephrectomy with curative intent is approximately 1.8% (Itano et al, 2000; Margulis et al, 2009). Isolated local recurrence is defined as recurrence in the ipsilateral retroperitoneal lymph nodes, renal fossa, or adrenal gland without evidence of distant metastasis (Fig. 55–34). In a study of 54 patients with isolated local recurrence managed with open surgical resection, median recurrence-free and cancer-specific survival of 11 and 61 months, respectively, were observed (Margulis et al, 2009). Perioperative systemic therapy with various combinations of immunotherapy, chemotherapy, and targeted tyrosine kinase inhibitors was used in 69% of cases. Given their rarity, published laparoscopic experience in surgically addressing these recurrences is quite limited to date. A series of five patients (one open conversion for vena caval invasion) undergoing a hand-assisted approach to isolated local recurrence has demonstrated that the procedure may be safely performed in selected patients (Bandi et al, 2008). At a mean follow-up of 43 months, cancer-specific and disease-free survival rates were 60% and 20%, respectively. The small number of patients in this report makes the results difficult to interpret. An open surgical resection can offer durable local control and cancer-specific survival in carefully selected patients; larger comparative laparoscopic series with sufficient follow-up are clearly needed to determine the efficacy of laparoscopy in these scenarios.
Renal Vein and Caval Tumor Thrombus
Several centers have now published their experiences with laparoscopy for renal cancers with associated tumor thrombus into the renal vein or inferior vena cava (Desai et al, 2003; Hsu et al, 2003; Martin et al, 2008; Guzzo et al, 2009). After complete laparoscopic mobilization of the kidney and ligation of the renal artery, a laparoscopic DeBakey procedure, vessel loop, or hand assistance is typically employed to “milk” the tumor thrombus distally toward the kidney. This allows either the endovascular stapler to be deployed proximal to the thrombus, or a laparoscopic Satinsky clamp to be placed to isolate a cuff of the vena cava such that the cuff may be excised to allow intact specimen extraction. When using a hand port, the vena cava may then be oversewn using proline suture. The use of intraoperative ultrasonography has also been described to aid in assessing the location of the proximal extent of the tumor thrombus (Hsu et al, 2003). To date, the approach has been limited to low-level caval thrombi, but results have been comparable with the open surgical experience.
Cytoreductive Nephrectomy
Patients with advanced renal cell carcinoma may require cytoreductive nephrectomy before the initiation of systemic secondary therapies. A comparative study of open versus laparoscopic cytoreductive nephrectomy in a selected group of patients with metastatic disease—but without local invasion, venous involvement, or bulky adenopathy—demonstrated similar one-year survival between the two groups (61% vs. 65%) (Rabets et al, 2004). In addition, the laparoscopic group had less blood loss, shorter hospital stay, and shorter interval between surgery and the initiation of systemic therapy (36 vs. 61 days). Other studies have shown similar results (Eisenberg et al, 2006; Matin et al, 2006a), although the shorter interval to systemic therapy has not been consistently observed.
Surgical Salvage after Failed Ablative Therapies
Nephrectomy following ablation is technically challenging due to resulting loss of tissue planes surrounding the lesion. A multi-institutional review of treatment outcomes for primary radiofrequency ablation (RFA) or cryotherapy revealed residual or recurrent disease in a median of 8.7% of patients (Matin et al, 2006b). Although a subgroup of these patients will undergo successful salvage ablative therapy, some may not be candidates for repeat ablation due to disease progression, tumor size, or failed repeat ablation. A report of 10 patients undergoing salvage surgery in this patient population has shown that laparoscopic nephrectomy was only possible in 4 patients, and the remainder required either open partial or radical nephrectomy (Nguyen et al, 2008). Extensive perinephric fibrosis was cited as the main factor complicating surgery.
Partial Nephrectomy
Kidney tumors are increasingly being diagnosed incidentally, at a small size and early stage, in asymptomatic healthy patients (Jayson et al, 1998; Leslie et al, 2003). Although the majority of imaging-detected incidentally found renal lesions are benign simple cysts, one must be vigilant to not miss an early renal cancer. Recent data regarding the prevalence of unsuspected, early-stage chronic kidney disease underscore an important point: Renal functional preservation and nephron-sparing strategies are important considerations when making management decisions in patients with a small renal mass (SRM) (Huang et al, 2006). The desire to provide a minimally invasive alternative to treat patients with small renal masses led to the application of laparoscopic techniques to nephron-sparing surgery.
The first transperitoneal laparoscopic partial nephrectomy (LPN) was reported in 1993 by Winfield and colleagues, with the retroperitoneal approach introduced one year later (Gill et al, 1994). Initially, LPN was applied to treat small clinical T1a exophytic renal masses (Fig. 55–35). With increasing laparoscopic confidence and experience, the indications of LPN have been expanded to almost all patients with challenging tumor anatomy in complex clinical settings.
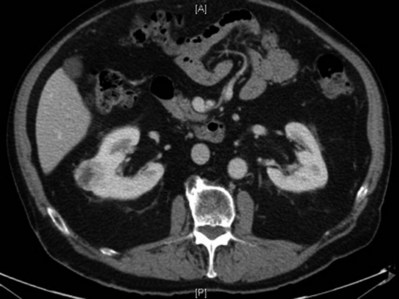
Figure 55–35 CT scan with IV contrast demonstrating a partially exophytic midpole clinical T1a lesion in the right kidney.
Expanded Indications
Clinical Stage T1b Tumors (4 to 7 cm)
LPN was initially restricted to patients with tumors less than or equal to 4 cm in size. Technical advances in laparoscopic techniques, along with demonstration of equivalent results to open surgery for T1a tumors, provided the groundwork for approaching larger lesions (Leibovich et al, 2004; Dash et al, 2006; Mitchell et al, 2006). Simmons and colleagues (2009) reported perioperative outcomes of LPN in 58 patients for T1b tumors. Mean tumor size was 6 cm, and 55% of tumors were centrally located. Although patients with pT1b tumors more often underwent pelvicalyceal repair (P = .004) and heminephrectomy (P < .001), they had similar operative time, blood loss, and hospital stay but longer warm ischemia time than patients with tumors less than 4 cm. Tumor size greater than 4 cm did not increase risk for positive cancer margins, intraoperative complications, or postoperative urologic complications. Of note, in patients with tumors less than 2 cm, 2 to 4 cm, and 4 to 7 cm (stage ≥III), chronic kidney disease (CKD) existed preoperatively in 31%, 35%, and 44% of patients, respectively, and postoperatively in 52%, 53%, and 63%, respectively (P = ns). This underscores the importance of attempting to provide a nephron-sparing approach to all patients. Given adequate laparoscopic experience and judicious patient selection, the perioperative outcomes of LPN for clinical T1b tumors greater than 4 cm appear comparable to those achieved for clinical T1a tumors less than or equal to 4 cm.
Laparoscopic Heminephrectomy
Finelli and colleagues (2005) compared outcomes of laparoscopic heminephrectomy (excising greater than 30% of renal parenchyma) in 41 patients, with a contemporary group of 41 consecutive patients who underwent LPN with less than 30% resection. Except for a longer ischemia time (39 vs. 33 minutes) in the heminephrectomy cohort, there were no differences between the two groups regarding blood loss, operation room (OR) time, analgesic requirement, hospital stay, postoperative serum creatinine, and overall complications. All surgical margins were negative. Specific technical considerations inherent to laparoscopic heminephrectomy include routinely performing deeper renal parenchymal resections, transection of sizable intraparenchymal blood vessels, and intentional entry into the pelvicalyceal system. The primary goals of laparoscopic heminephrectomy and LPN are the same: achieve negative surgical margins, perform clipping or suture repair to secure renal vessels, and where necessary, repair the collecting system while minimizing ischemia time.
Central and Hilar Tumors
Central tumors are defined as those abutting or invading the central renal sinus fat and/or the collecting system on preoperative CT. These tumors deeply infiltrate the renal parenchyma, and their excision requires intentional entry into and potentially suture-repair of the pelvicalyceal system along with complex parenchymal reconstruction, all within the time constraints of renal ischemia. The technical complexity of such cases depends upon the location of the individual tumor, and the type of suturing angles available for laparoscopic instruments in the dominant and nondominant hand. Frank and colleagues (2006) compared experience with LPN for 154 central tumors with LPN for 209 peripheral tumors. Although blood loss was similar, central tumors were associated with somewhat longer operative time, ischemia time, and hospital stay, and more early postoperative complications. There was only one positive margin for cancer in each group. These data reflect the initial outcomes of the LPN experience; at this writing, the authors routinely perform LPN for central tumors without any increase in operative morbidity as noted above.
Hilar tumors, defined as tumors located in the renal hilum in direct contact with the renal artery and/or vein on cross-sectional imaging, were initially considered by many to be a contraindication to LPN. In 2005, an initial experience with LPN outcomes for hilar tumors in 25 patients was reported (Gill et al, 2005). Mean tumor size was 3.7 cm (range 1 to 10.3). LPN was successful in all cases, without any open conversions or operative reinterventions. Postoperative hemorrhage occurred in three early patients. Preoperative 3D video reconstruction of triphasic spiral CT was important in detailing the number, interrelationship, anatomical course, and position of the renal vessels in relation to the tumor.
Tumor in a Solitary Kidney
Partial nephrectomy for tumor in a solitary kidney, open or laparoscopic, is challenging. The margin for error is small because a complication could result in temporary dialysis, or worse, render the patient anephric. In the largest reported series of patients (n = 22) undergoing LPN for a tumor in a solitary kidney mean warm ischemia time was 29 minutes (Gill et al, 2006). Two patients (9%) were electively converted to open surgery. Median pre- and postoperative serum creatinine (1.2 and 1.5 mg/dL, respectively) and estimated glomerular flow rate (eGFR) (67.5 and 50 mL/min/1.73 m2, respectively) reflected changes of 33% and 27%, respectively, which correlates with approximately 25% nephron volume loss. One kidney was lost due to hemorrhage. The authors concluded that LPN for tumor in a solitary kidney was feasible.
Multiple Tumors
Increasingly, nephron-sparing surgery (NSS) is the preferred treatment for patients with multiple, small ipsilateral tumors, due to the potential for contralateral recurrence. LPN has been applied in this clinical setting as well. Steinberg and colleagues (2004) published their experience in 13 patients (92% of patients had an imperative indication for nephron preservation) undergoing laparoscopic NSS for two or more ipsilateral renal tumors. LPN was performed in six patients either by itself, or in conjunction with cryotherapy. At a mean follow-up of 16.4 months (range 1 to 54), no ipsilateral recurrences were noted. When performing LPN for multiple tumors, depending upon their geographic proximity, tumor excision can be performed either en bloc or individually. With the short-term oncologic outcome data available to date, LPN, when judiciously combined with cryotherapy, can provide clearance of the ipsilateral tumor burden, while keeping ischemia time to a minimum.
Cystic Tumors
LPN for radiographically suspicious cystic tumors is technically more challenging due to the potential for inadvertent cyst rupture and tumor spillage. Spaliviero and colleagues (2005) compared 50 patients undergoing LPN for a cystic renal lesion with 50 consecutive patients undergoing LPN for a solid renal mass. LPN was successful in all cases, and intraoperative complications were similar in the two groups. All surgical margins were negative. However, one patient in the cystic tumor group developed retroperitoneal recurrence at 1 year, despite an unremarkable intraoperative course and negative histological margins. The authors concluded that extreme caution and careful technique is necessary to avoid inadvertent cyst rupture and local tumor spillage.
Other Indications
LPN has also been performed in the following unique clinical settings: adrenal involvement from an upper pole tumor requiring excision with concomitant adrenalectomy (Ramani et al, 2003), repair of concomitant renal artery disease (Steinberg et al, 2003), tumor in congenitally anomalous kidney, such as horseshoe kidney (Tsivian et al, 2007), in obese patients (Romero et al, 2008), and after prior ipsilateral renal surgery (Turna et al, 2008).
Procedure
Technical Issues
The principal technical challenge during LPN stems from the complexity of laparoscopic tumor excision and sutured renal reconstruction in a time-sensitive manner. The primary objectives are to complete tumor excision with negative margins, obtain hemostasis, and minimize warm ischemia time. Successful LPN for complex tumors requires an in-depth understanding of 3D renal anatomy, real-time appreciation of visual cues during laparoscopy, and facility with precise and relatively efficient suturing.
Transperitoneal LPN
The transperitoneal approach offers many features that are crucial for performance of advanced LPN: larger working space, more familiar landmarks, greater versatility of instrument angles, and technical ease of suturing. The initial portion of the procedure is performed as previously described for transperitoneal LRN.
Tumor Localization and Excision
Once the initial dissection is complete, including isolation of the hilar vessels, intraoperative ultrasonography is used to confirm location, width, and depth of the tumor (Fig. 55–36). Ultrasonography may also be used to confirm absence of additional lesions in the kidney. The Gerota fascia is entered away from the lesion to expose the renal capsule. Using the monopolar scissor, the capsule is scored circumferentially around the tumor (Fig. 55–37), and the hilum is then clamped using laparoscopic bulldog clamps (Fig. 55–38). The scored line may then be incised using cold shears (Fig. 55–39), and with the assistance of a suction-irrigator device to provide both countertraction and a clear operative field, the tumor excision is completed. In some patients, adherent fat will necessitate a subcapsular dissection to identify the visual borders of the tumor.
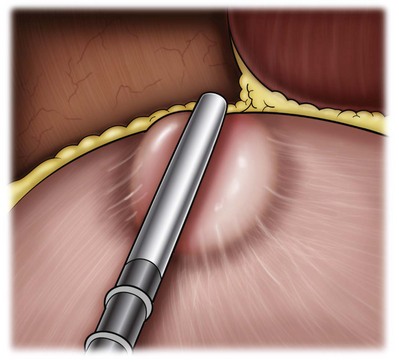
Figure 55–36 Intraoperative ultrasonography is used to confirm location, width, and depth of the tumor.
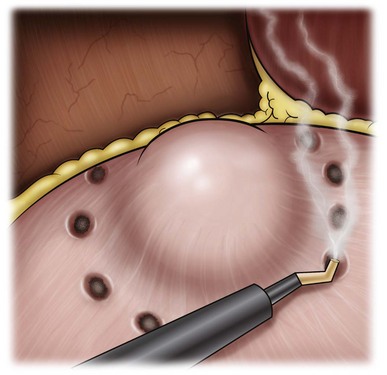
Figure 55–37 After Gerota fascia is cleared to expose the lesion and the renal capsule. Using the monopolar scissor or hook cautery, the capsule is scored circumferentially around the tumor.
Hemostasis
Achieving hemostasis is very important during LPN. Several strategies have been advocated for this purpose. The most widely employed technique to achieve hemostasis of the partial nephrectomy bed is by primarily using freehand laparoscopic suturing with or without adjunctive biologic hemostatic/sealant agents, or a Surgicel bolster (Johnson and Johnson, New Brunswick, NJ) (Fig. 55–40). Alternatively, surgical clips can be used while coming across the surface of the parenchyma. A number of tissue sealants are available: gelatin matrix thrombin sealant (Floseal; Baxter, Deerfield, IL), fibrin glue (Tisseel; Baxter), polyethylene glycol hydrogel (Coseal; Baxter), cyanoacrylate glue (Dermabond; Ethicon, Somerville, NJ), and Bioglue (CryoLife, Atlanta, GA).
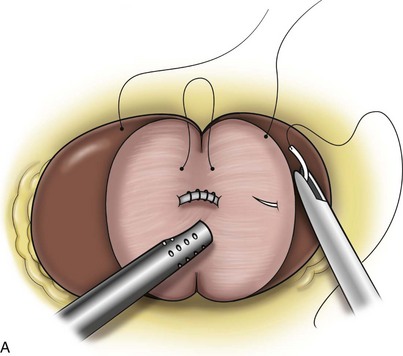
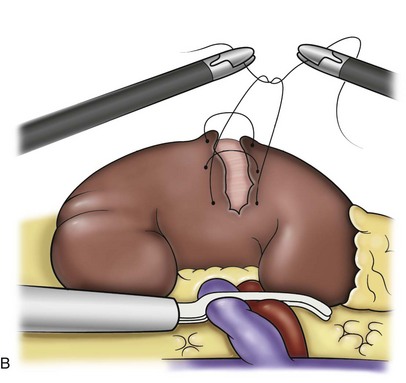
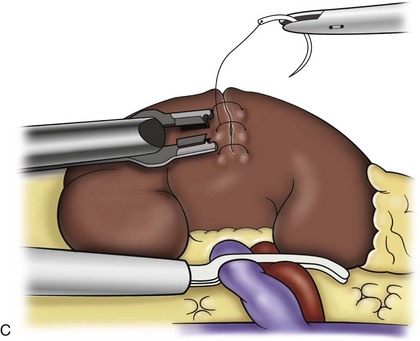
Figure 55–40 A, After use of the argon beam coagulator on the exposed parenchyma, interrupted absorbable sutures are placed for the renorrhaphy. The collecting system has already been repaired and the suction-irrigator serves to provide countertraction and maintain a clear operative field. B, The sutures may be laparoscopically tied with optional pledgets to help prevent capsular tearing during closure. C, Alternatively, sutures with preplaced Lapra-Ty clips (Ethicon Endosurgery, Cincinnati, OH) at the tail are used and secured with an additional Lapra-Ty clip after passing the needle and adjusting tension on the closure.
During LPN for selected small, superficial, exophytic tumors, various thermal (radiofrequency, microwave, ultrasonic) and novel alternative (laser, water-jet) energy sources have been employed for hemostasis clinically and in the laboratory. Herrell and Levin (2005) evaluated the TissueLink radiofrequency device (TissueLink Medical, Dover, DE) during unclamped LPN in 25 human cases. Fibrin glue was used as a hemostatic adjunct. There were no intraoperative complications. Mean estimated blood loss was 98 mL (range 10 to 337). This device could potentially have a role in unclamped LPN for small, peripheral, and exophytic tumors. Lasers have been used as an energy source for performing unclamped LPN in the animal model. The potassium-titanyl-phosphate laser (KTP) (GreenLight PVP; Laserscope, San Jose, CA) and the holmium : yttrium aluminum garnet (Ho : YAG) laser have been employed with initial success (Lotan et al, 2004; Hindley et al, 2006; Liu et al, 2006). Moinzadeh and colleagues (2005) also evaluated the water-jet device (Helix Hydro-jet; Erbe-USA, Marietta, GA) to perform LPN without renal hilar control in the calf model. They were able to perform 18 of 20 cases without hilar control, with a mean estimated blood loss of 174 mL. Further technological improvements are necessary to achieve reliable hemostasis using these technologies.
Hilar Control
Select superficial, exophytic, noninfiltrating tumors can be excised during LPN without hilar clamping. However, tumors that are larger, deeper, central, or hilar require a substantial LPN, necessitating transient hilar clamping to provide a bloodless operative field. Guillonneau and colleagues (2003) compared LPN with (n = 12) and without (n = 16) hilar clamping using ultrasonic shears and bipolar cautery. The authors concluded that hilar clamping provided decreased blood loss, shorter operating time, and superior surgical performance. From a technical standpoint, hilar control can be achieved by using bulldog clamps individually on the renal artery and vein, or by using a Satinsky clamp across both vessels to achieve en bloc hilar clamping.
Renal hypothermia
Three techniques have been described for laparoscopic renal hypothermia: surface cooling with ice slush, instillation of cold saline through a retrograde ureteral catheter, and intra-arterial perfusion of cold saline or lactated Ringer solution (Gill et al, 2003; Landman et al, 2003; Janetschek et al, 2004). Although all of these techniques are clinically feasible and reasonably effective, they are rarely employed during clinical LPN. This is not only because of their complexity, but also because the majority of tumors subjected to LPN do not currently require an inordinately long period of warm ischemia.
Collecting System Repair
Central tumors abutting the renal sinus fat and collecting system may require deliberate entry into the pelvicalyceal system (PCS) to ensure negative surgical margins during tumor excision. For this reason, PCS entry is a common occurrence in contemporary LPN practice. Retrograde injection of dilute indigo carmine through a 5-Fr ureteral catheter inserted into the renal pelvis can be used to help identify the site of PCS entry and confirm repair. Prospective comparison of perioperative outcomes in 27 LPN with pelvicalyceal entry to 37 LPN with no pelvicalyceal entry (Desai et al, 2003) revealed similar operating room (OR) time, tumor excision time, and blood loss. However, PCS suture repair was associated with a somewhat longer warm ischemia time and hospital stay. None of the patients undergoing PCS suture repair developed a urinary leak. The results of this early study showed that intentional entry into the PCS system for central tumors could be safely and effectively repaired (Fig. 55–41). Suture repair of the PCS with a running 3-0 polyglactin suture can be accomplished, and the integrity of the repair tested with retrograde injection of dilute methylene blue. Alternatively, one may close the renal capsule over the defect. In either case, a drain should be placed for postoperative monitoring.
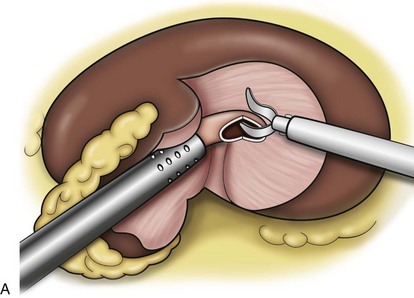
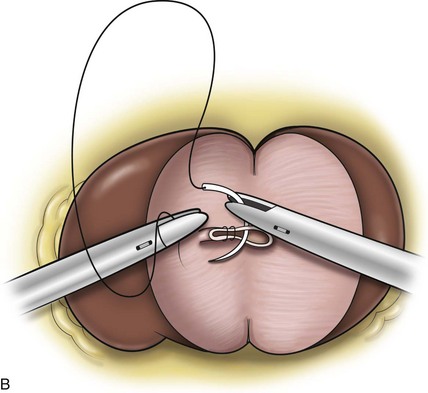
Figure 55–41 A, When deep resection is required, the collecting system will often be transected. With occlusion of the renal vessels, these defects can easily be identified and closed using absorbable sutures. The cut edge of the collecting system is identified with the tip of the needle and elevated. B, An interrupted figure-of-eight suture or running suture is used to completely close the collecting system. The integrity of the repair can be determined by intravenous indigo carmine administration or retrograde instillation, if a ureteral catheter was placed at the beginning of the case. Care must be taken not to destroy the suture, if argon beam coagulation of the parenchyma surface will be performed.
Retroperitoneal LPN
Although most LPN surgeons prefer the transperitoneal approach for most tumors, some employ the retroperitoneal approach, which has advantages for select posteriorly located upper pole apical tumors. After entry into the retroperitoneum and establishment of a working space as previously described, the kidney may be lifted anteriorly off the psoas muscle to allow visualization of the arterial pulsation. The dissection of the renal hilum can then proceed to facilitate bulldog clamp placement when deemed necessary. Tumor localization and excision, hemostatic control, collecting system repair, and renorraphy are performed as described for the transperitoneal approach. In a comparison of 32 retroperitoneal with 19 transperitoneal LPN, choice of approach was based on tumor location (Wright and Porter, 2005). The retroperitoneal approach was associated with shorter operating time, decreased blood loss, quicker return of bowel function, and shorter hospitalization. These authors preferred the retroperitoneal approach for polar and posterolateral masses, while the transperitoneal approach was preferred for anterior and medial lesions. Another comparison of 100 transperitoneal with 63 retroperitoneal LPN demonstrated that blood loss, perioperative complications, postoperative serum creatinine, analgesic requirements, and histological outcomes were comparable in the two groups (Ng et al, 2005). As such, the choice of LPN approach is dictated primarily by surgeon experience and tumor location. Other factors include tumor size, number of tumors, number of arteries supplying the kidney, amount of visceral fat surrounding the kidney, and route of any prior open surgery on the quadrant of interest. The transperitoneal approach for all renal tumors is usually easier, except those that are located posteriorly or posteromedially on the upper pole.
Early Unclamping LPN Technique
The standard LPN technique involves en bloc hilar clamping of the renal artery and vein followed by cold endoshear excision of the tumor with adequate margins. Control of transected intrarenal blood vessels and pelvicalyceal repair are obtained with an initial, central, running suture. Subsequent parenchymal reconstruction involves parenchymal mattress sutures tied over a bolster, with adjunctive use of biologic hemostatic agent. Thus the entire renal repair is performed in the ischemic kidney, with the hilar vessels clamped.
More recently, an early unclamping technique has been developed in an attempt to decrease ischemia time (Nguyen and Gill, 2008). In this technique, the renal hilum is clamped only for the duration of tumor excision and placement of the initial, central, running suture. In conjunction with suturing, clips are used selectively for preemptive control of the larger intrarenal blood vessels before transection. The hilum is then unclamped, reperfusing the kidney. All subsequent suturing in the partial nephrectomy bed to ensure parenchymal hemostasis and pelvicalyceal repair is now performed in the perfused, revasularized kidney. This includes specific figure-of-eight sutures to control any remaining active arterial and/or venous bleeding sites. Horizontal mattress sutures are also placed, if indicated, to undersew various clips, thereby excluding them from immediate proximity to the underlying pelvicalyceal system. If desired, the partial nephrectomy bed can be covered with biologic, hemostatic, or adhesive agents. Given that this technique allows hemostasis to be visually confirmed in the perfused kidney, the compression bolster is rarely used in contemporary practice. The benefits of early unclamping are twofold: (1) it decreases ischemia time, and (2) it leads to decreased incidence of postoperative hemorrhage.
Robotic-Assisted Partial Nephrectomy
Robotic-assisted PN (RaPN) is being explored at several centers with the aim of facilitating the precise intracorporeal suturing necessary during LPN. In its early days, RaPN remains a procedure in evolution (Gettman et al, 2004; Caruso et al, 2006; Kaul et al, 2007; Rogers et al, 2008).
The responsibilities of the table-side assistant include clamping the renal hilum, providing suction and retraction to maintain a clean operative field, delivery and cutting of sutures, and clip placement as needed. The console-side surgeon performs tumor excision, hemostatic suturing, and pelvicalyceal and parenchymal suture reconstruction robotically. Upon completion of renal reconstruction, the hilum is unclamped, and additional parenchymal sutures are placed, as needed, to ensure hemostasis. The robot is undocked and laparoscopic exit completed. The early unclamping LPN technique may be easily adapted for use during RaPN with good success.
Since 2004, several centers have reported initial experiences with RaPN (Gettman et al, 2004; Caruso et al, 2006; Kaul et al, 2007; Rogers et al, 2007). As such, the technical feasibility of RaPN has been established. More recently, initial data comparing outcomes with LPN have become available in a matched-pair analysis comparing RaPN and LPN (Aron et al, 2008). Patients were matched for age, gender, body mass index (BMI), ASA score, tumor side, tumor size, tumor location, and specific technique employed (early vs. conventional unclamping). Perioperative outcomes were similar between the RaPN and LPN cohorts with regard to estimated blood loss, warm ischemia time, OR time, and length of stay. Renal functional outcomes, transfusion rate, and complication rates were also comparable. Two RaPN cases required conversion to standard LPN. In a subset analysis of patients undergoing the early unclamping technique, the LPN cohort had a shorter warm ischemia time (14 vs. 21 minutes, P = .05), despite larger tumors in the LPN cohort (3 cm vs. 2.4 cm, P < .01). The authors concluded that RaPN, an evolving procedure, is technically feasible and safe, albeit with a longer ischemia time compared to LPN in the initial experience. Analysis of a larger number of cases is necessary to determine the relative merits of RaPN. However, it is likely that the growing familiarity of urologists with robotic surgery will facilitate the application of minimally invasive nephron-sparing surgery in a larger cohort of patients. Unfortunately, the robotic approach is associated with increased cost and cosmetic inferiority due to the need for additional access ports.
Warm Ischemia
The limit of safe renal warm ischemia time has historically been considered to be 30 minutes. Although supported by canine and anecdotal clinical data, no scientifically rigorous clinical study has defined an ischemic dose-response curve to date. Indeed, recent data suggests that up to 90 minutes may be reasonable (Orvieto et al, 2005). Until this issue is better understood, all efforts should be made to keep warm ischemia time as short as possible. With regard to surgical renal ischemia, various myths abound due to insufficient data.
Pneumoperitoneum
There is ongoing debate regarding the effects of pneumoperitoneum on renal functional outcomes. It has been demonstrated that 20 mm Hg of intraperitoneal pressure can cause decreased renal cortical capillary perfusion, increased renal venous outflow resistance, and transient oliguria in pigs (Shuto et al, 1995). In contrast, a report by Cisek and colleagues (1998) showed that 6 hours of 20-mm Hg pneumoperitoneum in pigs, rendered renally insufficient with prior left nephrectomy and right heminephrectomy, had no long-term impact on renal function. The debate regarding the negative impact of pneumoperitoneum remains unresolved; however, these studies suggest that when applied for less than 6 hours in patients with normal preoperative renal function, its ultimate impact is likely negligible and reversible. There is general consensus, however, that pneumoperitoneum during laparoscopic surgery should not be maintained with sustained pressures of greater than 20 mm Hg. Attempts at completely eliminating potential effects of pneumoperitoneum on the kidney have also been reported with donor nephrectomy using gasless laparoscopy (Watanabe et al, 2002), but it is unclear whether there are any long-term benefits of this approach.
Renal Artery-Only versus Artery-Plus-Vein Clamping
It is theorized that artery-only clamping during partial nephrectomy would allow retrograde venous blood flow with potentially partial oxygenation to the renal parenchyma. In a solitary kidney pig model, Orvieto and associates (2007) found that artery-only clamping resulted in lower serum creatinine rises, during postoperative days 1 to 3, than in animals that underwent complete hilar occlusion. Interestingly, this effect was not observed in animals that underwent laparoscopic surgery. It was concluded that artery-only clamping provided immediate postoperative benefit, and that this benefit was likely offset by pneumoperitoneum-induced venous compression during laparoscopy. In contrast, the same group found a benefit to artery-only clamping in a case-control study of patients undergoing LPN with artery-only (n = 25) versus simultaneous clamping of the artery and vein (n = 53) (Gong et al, 2008). A significant decrease in serum creatinine and creatinine clearance was observed in those patients undergoing simultaneous clamping of the artery and vein compared to preoperative levels. This effect was not observed in those undergoing artery-only clamping. Additionally, there were no statistically significant differences observed in either blood loss or positive margin rate between the groups. These findings need validation in larger patient cohorts. From a practical standpoint, leaving the renal vein unclamped can lead to increased intraoperative renal parenchymal bleeding, which can obscure the surgical field, leading to increased blood loss and prolonged ischemia time. In the majority of partial nephrectomy cases at the authors’ institutions, both the renal artery and vein are clamped routinely to achieve vascular control and optimize visualization during tumor excision and renorrhaphy.
Manual Compression of Renal Parenchyma versus Vascular Clamping
Some advocate the use of manual compression of the kidney during tumor excision and renorrhaphy, while others contend that vascular clamping is preferable. Manual compression can be effective for small exophytic tumors, although the renal vasculature should always be accessible to allow emergent clamping in the case of hemorrhage. No controlled clinical study examining this issue has been conducted. Issues to consider include the possibility of tissue trauma due to excessive compression, and the limitation of this approach to peripheral tumors during open and hand-assisted laparoscopic procedures only. Manual compression techniques would not be feasible in partial nephrectomy of central, hilar, or large tumors, or for pure laparoscopic surgery.
Intermittent versus Continuous Clamping
Controversy exists regarding the possible deleterious effects of intermittent perfusion by sequential clamping and unclamping during partial nephrectomy. Intermittent ischemic “preconditioning” has been extensively studied as a method for protecting against ischemic organ injury, especially in liver surgery (Centurion et al, 2007; Saidi et al, 2007). Wilson and colleagues (1971) compared renal function in canine kidneys subjected to intermittent versus continuous occlusion for 1 to 2 hours. Occlusion and reperfusion times were 15 minutes and 3 minutes, respectively, repeated for 2 hours of occlusion time. In subjects with intermittent occlusion, GFR was significantly decreased after 7 days compared with continuous occlusion subjects. Contrary to these findings, Frank and colleagues (1993) found that rats undergoing cycled occlusion-reperfusion (15 minutes occlusion followed by 5 minutes reperfusion) for 45 minutes had lower serum creatinine levels and decreased pathologic evidence of tubular injury at 3 days compared with the continuous occlusion subjects. Another study confirmed a protective effect of intermittent ischemic preconditioning in rats, and established the optimal occlusion and reperfusion intervals at 15 and 10 minutes, respectively (Wu et al, 2009). It is possible that species differences may account for discrepancies within the literature. No clinical trials of renal ischemic preconditioning have been conducted; only anecdotal reports guide practice. Because of insufficient and conflicting data regarding intermittent clamping, ischemic preconditioning is not recommended.
Parenchymal Clamping
Several authors have published their initial experiences in a small number of patients using techniques of parenchymal clamping in selected patients with peripheral renal cortical tumors (Verhoest et al, 2007; Simon et al, 2009). Use of a parenchymal clamp can allow the surgeon to induce ischemia only in the immediate area surrounding the tumor. All patients in these two studies had negative surgical margins, and no changes in renal function were reported. The potential benefits of this technique are multiple: It facilitates a bloodless operative field in the area of the tumor; the majority of the normal parenchyma is spared from ischemic injury; and the time-sensitivity of tumor excision and renorrhaphy is markedly reduced. However, the limitation of the technique is that only selected patients with peripherally located tumors are candidates. Larger numbers of patients are necessary to validate this technique, but due to the reduction in time sensitivity, parenchymal clamping may allow a larger cohort of surgeons to perform LPN with increased confidence.
Solitary Kidney May Be “More Tolerant” to Ischemic Injury
It is known that contralateral normally-functioning kidneys will hypertrophy to compensate for decreased function after unilateral nephrectomy. The extent of this response has been shown to be species- and age-dependent (Funahashi et al, 2009). Fried and colleagues (1984) studied functional recovery in rats with or without nephrectomy 2 weeks before a 40-minute ischemic insult. Nephrectomized rats demonstrated improved inulin clearance at 48 hours compared with non-nephrectomized controls. In contrast, Stackl and colleagues (1983) failed to show a difference in renal blood flow (RBF) or GFR in rabbits with or without preischemic nephrectomy. For obvious reasons, a human clinical study of this effect is impossible. Clinical anecdotal evidence points to the possibility that solitary kidneys may be more resistant to ischemic injury. It is plausible that some aspect of this increased tolerance to ischemic injury is attributable to the increased intraoperative vigilance and postoperative support measures taken in this high-risk patient cohort.
Laparoscopic Partial Nephrectomy: Contemporary Outcomes
An 1800-patient retrospective multi-institutional study compared a mature series of 1029 OPN cases with the initial LPN cases for solitary T1 tumors less than or equal to 7cm (Gill et al, 2007). Tumors in the OPN group were larger (3.3 vs. 2.6 cm), and more often located centrally (53% vs. 34%) or in a solitary kidney (P < .001 for all comparisons). LPN had less blood loss, and shorter operative time, hospital stay, and convalescence (P < .001 for all comparisons). Overall postoperative complications (25% vs. 19%) and conversion to radical nephrectomy (1% vs. 0%) were somewhat greater in the LPN group. Importantly, LPN and OPN were similar with regard to intraoperative complications (1.8% vs. 1%), positive surgical margins for cancer (1.6% versus 1%), 3-year oncological outcomes, and 3-year renal functional outcomes. However, LPN had a 10-minute longer ischemia time (30 vs. 20 minutes), and somewhat higher postoperative hemorrhage (4.2% vs. 2%) and reintervention rate.
With increasing LPN experience and standardization of the early unclamping technique, contemporary LPN outcomes have improved significantly. Specifically, the two remaining concerns for higher postoperative hemorrhage and longer ischemia time with LPN have now been addressed, leading to significantly decreased ischemia time and reduced occurrence of post-operative hemorrhage. The initial report of the early unclamping technique in LPN has decreased ischemia to a mean of 13.9 minutes (55% reduction), and reduced postoperative hemorrhage rate to 2% (Nguyen and Gill, 2008).
Gill and colleagues (2010) reported a single-surgeon series of 800 laparoscopic partial nephrectomy (LPN) cases encompassing a 9-year period (1999 to 2008). The authors divided the entire cohort into three chronologic eras: era I: 1999 to 2003 (n = 276), era II: 2004 to 2006 (n = 289), and era III: 2007 to 2008 (n = 235). On comparing eras I, II, and III, tumors in the most recent era were larger, more commonly greater than or equal to 4 cm and central, and, less often, peripheral and less than 4 cm (P value significant for all). Despite such increasing tumor complexity, mean warm ischemia times were shorter in the most recent era: 32 minutes, 32 minutes, and 14 minutes, respectively, (P < .0001). Also, overall, postoperative and urologic complications were significantly lower in the most recent era. Finally, renal functional outcomes were superior in era III, as documented by lesser percent decreases in estimated GFR (18%, 20%, and 11%, respectively).
Kamoi and Gill also retrospectively compared 150 contemporary patients undergoing OPN (2006 to 2008) with 150 contemporary patients undergoing LPN (2007 to 2008) (author’s unpublished data). Notably, all OPNs were performed by one surgeon experienced with open procedures, and all LPNs were performed by one surgeon experienced with laparoscopy procedures. LPN patients had a shorter ischemia time (21 vs. 13 minutes, P < .0001); more LPN patients had ischemia time of less than or equal to 20 minutes (52% vs. 97%); and fewer had an ischemia time greater than 30 minutes (9% vs. 0%; P < .0001 for both). Postoperative complications were fewer in the LPN group (19% vs. 8.7%; P = .01), including hemorrhage (3.3% vs. 2.7%; P = ns) and urine leak (7.3% vs. 1.3%; P = .02). Patients undergoing elective PN had similar outcomes, and those undergoing imperative PN or PN in a solitary kidney had superior renal functional outcomes in the LPN cohort, probably because of the reduced ischemic insult during LPN. A recent study compared 86 off-clamp LPN with traditional clamped LPN (Herati et al, submitted). Significant differences (P < .05) in the off-clamp group relative to the on-clamp group included shorter operative time (132 vs. 146 minutes), a higher proportion of exophytic lesions (51% vs. 28%), smaller tumor volume (26 vs. 41 cm3), and fewer hilar tumors (15% vs. 29%). Short-term elevation in serum creatinine was significantly less (6% vs. 29%) although long-term significance of this finding is unknown. There were no significant differences in blood loss, transfusion rate, or complications. As experience improves, an increasing number of procedures may be performed off clamp.
These new data indicate that, despite increasing tumor complexity, three key outcomes of contemporary LPN (ischemia time, complications, and renal function) have improved significantly and now approach outcomes of open partial nephrectomy.
Positive Surgical Margin
The clinical significance of a pathologic positive surgical margin after partial nephrectomy, whether laparoscopic or open, has prompted several studies evaluating outcomes specifically in these patients. In a group of 1344 patients undergoing open partial nephrectomy, positive surgical margins were noted in 77 cases (5.5%) (Yossepowitch et al, 2008). The 10-year probability of freedom from local recurrence and progression to metastatic disease was 93% for both. There were no significant differences noted between those with positive and negative margins. Several laparoscopic surgeons have reported similar results. Of 511 patients undergoing LPN for renal cell carcinoma, 9 patients (1.8%) had a positive surgical margin (Permpongkosol et al, 2006a). Two underwent completion nephrectomy with no evidence of residual tumor, one patient with von Hippel-Lindau disease died of metastatic disease at 10 months, and six patients were observed to have no evidence of recurrence at mean follow-up of 32 months. Although these data demonstrate that many patients with pathologically positive margins may be observed, negative surgical margins should always be the goal in any oncologic procedure.
Long-Term Outcomes
Long-term data on LPN are now available (Table 55–4), and appear similar to open partial nephrectomy (OPN). Five-year cancer-specific survival after LPN is 100% (Lane et al, 2007). Most recently, a retrospective comparative study of 7-year outcomes compared 77 patients undergoing LPN with 310 patients undergoing open partial nephrectomy (OPN) for single clinical stage T1 (≤7 cm) tumors between 1999 and 2001 (Lane and Gill, 2010). Cancer recurred infrequently, and only rarely caused mortality, after either LPN or OPN. At 7 years, metastasis-free survival was 97.5% and 97.3% (P = 0.47) after LPN and OPN, respectively. After accounting for baseline differences between the cohorts, using propensity score–matching, 7-year metastasis-free survival was similar after LPN and OPN. On multivariate analysis, predictors of all-cause mortality included advancing age (P < .0001), comorbidity (P < .0001), preoperative renal dysfunction (P = .0001), but not tumor size (P = .6) or operative approach (LPN vs. OPN, P = .06). The authors concluded that LPN and OPN provide similarly excellent long-term overall and cancer-specific survival, with the vast majority (97%) of patients experiencing metastasis-free survival.
In experienced hands, LPN now is equivalent to OPN, with shorter ischemia times, and equivalent complication rates and renal functional outcomes. As a result, several centers routinely offer LPN for the majority of tumors (Permpongkosol et al, 2006b; Kamoi et al, submitted). These include technically challenging SRMs, including tumors which are hilar, central, completely intrarenal, larger (4 to 7 cm, pT1b) or located in a solitary kidney (Turna et al, 2008).
Laparoscopic Ablative Techniques
As the incidence of the small renal mass has increased with the prevalence of cross-sectional imaging, a stage migration has occurred such that a rising number of patients are presenting with low-stage disease. Although the standard of care for these lesions remains surgical excision, due to cancer-specific survival rates in excess of 95% (Frank et al, 2005; Lane et al, 2007), cryoablation and radiofrequency ablation (RFA) have emerged as alternative treatment options for patients with a small renal mass. High-intensity focused ultrasonography, microwave therapy, and high-intensity radiation have also been investigated. The primary goals of these techniques are complete tumor destruction while minimizing morbidity. The potential advantages include less blood loss, less dissection, and fewer complications. Indications are similar for all of the ablative technologies and include lesions in patients with significant comorbidities, solitary kidneys, and hereditary renal cell carcinoma. The percutaneous approach is preferred for treatment of small renal masses due to its lower morbidity, but in some instances may not be possible due to tumor location or proximity to adjacent organs. For these reasons, laparoscopy is required for direct visualization and manipulation to make treatment delivery feasible. As cryoablation and RFA are the most prevalent in clinical applications, these ablative techniques will be discussed here in further detail.
Cryoablation
Laparoscopic cryoablation may be delivered using either a transperitoneal or retroperitoneal approach, with the decision resting primarily on tumor location. The kidney is mobilized using a three-port technique, and Gerota fascia is opened in a manner similar to that employed for LPN. The fat overlying the tumor may be excised and placed in a specimen bag for extraction and pathologic analysis. Biopsy samples of the tumor itself may also be taken with a 14- or 18-gauge biopsy needle for pathologic diagnosis. Placement of the cryoablation probes into the tumor can be performed percutaneously, leaving the ports free for instrumentation. Direct visualization of probe placement and depth of placement are confirmed with the laparoscope and intraoperative ultrasonography, respectively. Number and spacing of probes is dictated by probe-specific ablative diameter, and they should be positioned to ensure cryolesion overlap, typically parallel to one another in a triangular or quadratic configuration. The tip of the probes should be advanced just beyond the deepest margin of the tumor.
Cryoablation is believed to work by two distinct mechanisms: at the cellular level by intracellular ice formation and solute damage, and at the vascular level, secondary to thrombosis and subsequent coagulative and ischemic necrosis. It uses thin probes cooled with compressed gas, such as argon or carbon dioxide, to achieve very low temperatures. Tissue destruction and necrosis occur consistently at temperatures at or below −19.4° C and temperatures of −20° C are achieved 3.1 mm inside of the leading edge of a 3.2-cm iceball (Campbell et al, 1998; Chosy et al, 1998). Typically, an active double freeze-thaw cycle is used to enhance tumor destruction (Gill et al, 1998; Nakada et al, 1998). Clamping of the renal artery during freezing, to theoretically avoid the heat-sink effect of renal blood flow, has been shown to make no difference in the freezing effect (Orihuela et al, 1999).
The progress of the ice-ball formation may be monitored in real time using intraoperative ultrasonography, and the iceball should extend approximately 1 cm beyond the edge of the tumor. Keeping in mind that the progress of the iceball cannot be abruptly stopped, caution should be exercised to avoid contact of the iceball with the renal collecting system, ureter, renal vasculature, or adjacent organs. After the freeze-thaw cycles are complete, the probes are removed with a gentle twisting motion. If any bleeding occurs, it can usually be controlled by applying pressure, or, if necessary, hemostatic agents such as fibrin glue or Floseal.
Radiofrequency Ablation
Similar to cryoablation, RFA may be administered laparoscopically using either a transperitoneal or retroperitoneal approach. After ultrasound confirmation of tumor location and size, the RFA probe is introduced into the tumor, and the tines are deployed to a diameter that ensures ablation of the tumor and a 1-cm margin of normal renal tissue. The size of the thermal lesion is determined by temperature- or impedance-based monitoring. The probe uses an alternating current of high-frequency radio waves, causing ion vibration. The resistance in the tissue causes generation of sufficient heat to result in thermal tissue damage—tumor coagulation, protein denaturation, and cell membrane disintegration all occur (Goldberg et al, 2000; Aron and Gill, 2007). Immediate histopathology after RFA shows hypereosinophila and pyknosis, which is subsequently replaced by coagulative necrosis within days to weeks (Crowley et al, 2001). To achieve these effects, optimal temperatures for ablation range between 60° C and 100° C and avoid tissue vaporization, which may occur at temperatures over 105° C (Goldberg et al, 2000; Crowley et al, 2001).
Unfortunately, unlike cryoablation, real-time ultrasonography cannot be used to monitor the thermal lesion induced by RFA. RFA itself may interfere with ultrasound imaging, and the affected tissue does not have any immediate change in echotexture. Color Doppler ultrasonography has been evaluated during RFA, but does not reliably contribute to monitoring the lesion (Crowley et al, 2001). Although MRI has been used to monitor the changing appearance of ablated lesions at the time of percutaneous treatment (Lewin et al, 2004), there is no current imaging technique that effectively monitors the progress of RFA lesions intraoperatively. Questions have been raised about temperature-based monitoring of the lesion, due to the observation that temperatures measured at the limit of the ablated area are actually 20° C to 30° C cooler than what is measured by the probe thermocouples. A potential solution involves the use of independent temperature probes to monitor temperature at the edge of the desired treatment area (Wingo et al, 2008). This allows a more definitive end point in the ablation cycle. Alternatively, an impedance-based system may be used. Instead of direct temperature measurement, this method relies on tissue impedance; sufficiently desiccated tissue becomes an insulator, and at an impedance level of 200 Ω, further progression of the thermal lesion is unlikely to occur (Lewin et al, 1998).
Imaging Follow-up
While cross-sectional imaging serves as the mainstay to determine treatment efficacy, there is debate about how to interpret the appearance of ablated lesions on postoperative imaging studies. Lack of enhancement on postprocedure CT scan or MRI is generally considered to define success for both cryoablation and RFA. Studies evaluating the appearance of cryolesions by CT or MRI demonstrated that peripheral rim enhancement is a common finding after laparoscopic cryoablation, and not necessarily cause for concern as demonstrated by follow-up percutaneous biopsy of these lesions (Remer et al, 2000; Bolte et al, 2006; Beemster et al, 2008). Cryolesions were generally noted to decrease in size over time, up to 94% at one year (Remer et al, 2000). Nonenhancing infiltrated fat may also be noted in the area overlying the lesion. Findings to elicit cause for concern for residual or recurrent disease were new rim enhancement, internal enhancement, or increase in size of the cryolesion. RFA lesions followed with MRI tend to be hypointense with a surrounding bright rim on T2-weighted imaging, and hyperintense on T1-weighted images. Tumor recurrence by most authors is defined as hyperintense soft tissue signal within the ablation zone, along its margin on T2-weighted images, or in areas of abnormal contrast enhancement within the treated region on the postcontrast images. Over time, RFA lesions also involute, causing a decrease in their size as measured by MRI or CT.
Treatment Outcomes
Several studies have reported promising short-term outcomes of laparoscopic cryoablation (Gill et al, 2000; Cestari et al, 2004). A study of 59 patients undergoing laparoscopic cryoablation, with a median tumor size of 2.5 cm and median follow-up of 26.8 months, documented 2 patients with tumor recurrence (Lawatsch et al, 2006). In a study of 81 patients undergoing laparoscopic cryoablation, 31 were presented with a minimum 3-year follow up (Weld et al, 2007). Biopsy of these lesions demonstrated that 61% were malignant and 39% were benign. Follow-up imaging performed at 1, 3, 6, 12, 24, and 36 months showed one patient who developed return of enhancement that was suspicious for recurrence at 36 months.
A comparison of 66 patients undergoing laparoscopic or percutaneous cryoablation demonstrated equivalent oncologic control at 30-months mean follow-up with no progression to metastasis (Malcolm et al, 2009). However, the percutaneous approach had a significantly higher primary treatment failure rate (25% vs. 3.8%). These patients either underwent salvage treatment or were observed with serial imaging. A similar comparative study of 37 patients with median follow-up of approximately 12 months demonstrated no difference in recurrence (4.2% vs. 5.3%) or need for re-treatment between the laparoscopic and percutaneous groups (Finley et al, 2008).
A multi-institutional study of RFA and cryoablation outcomes in 616 patients demonstrated residual or recurrent disease in 13.4% of patients undergoing RFA and 3.9% of patients undergoing cryoablation (Matin et al, 2006). Overall, 8.7% of patients failed primary therapy, and, after salvage ablative therapy, the failure rate was reduced to 4.2%. The authors noted that the majority of failures were detected after less than 3 months, and that cross-sectional imaging should be obtained 3 to 4 times at spaced intervals for the first year after undergoing treatment. A subsequent meta-analysis of patients undergoing partial nephrectomy, ablative therapies, or observation noted a higher risk of recurrence in patients undergoing cryoablation (RR = 7.45) or RFA (RR = 18.23) when compared with partial nephrectomy (Kunkle et al, 2008). Treatment failure was also linked to tumor size. However, no significant difference in rates of progression to metastatic disease was observed, regardless of treatment modality (Kunkle et al, 2008). Other studies have demonstrated that endophytic status may be linked to treatment failure after laparoscopic cryoablation, perhaps explained by increased difficulty with ultrasound-guided targeting of the lesion without direct visualization afforded by exophytic lesions (Wright et al, 2007).
Complications
A multi-institutional experience with 148 laparoscopic cryoablation procedures on 144 patients reported a complication rate of 15.5% (Laguna et al, 2009). Significant independent predictors of negative outcomes and complications included tumor size, preexisting cardiac disease, and female gender. Although larger tumor size and cardiac disease would intuitively lead one to predict increased complication rate, the association of female gender is unclear. A second multi-institutional study investigated complications of both percutaneous and laparoscopic cryoablation and radiofrequency ablation of small renal tumors (Johnson et al, 2004). A total of 139 cryoablations were performed along with 133 RF ablations. An overall 11% complication rate was observed with 1.8% classified as major, 9.2% as minor. Major complications included significant hemorrhage, ileus, uteropelvic junction (UPJ) obstruction necessitating nephrectomy, urinoma, conversion to open surgery, and death (aspiration pneumonia). In the laparoscopic group (90 patients), a 9% complication rate was reported, with the most common complication being pain or paresthesia at the ablation probe insertion site.
Laparoendoscopic Single-Site (Less) Surgery of the Kidney
Clinical Experience of Renal LESS Surgery
Initially reported for nephrectomy in the urologic literature (Raman et al, 2007), LESS has now been used to perform a variety of transumbilical procedures. Adjunctive 3-mm subxiphoid ports and 2-mm needlescopic ports have been required in some cases for liver retraction and laparoscopic suturing, respectively. Reported operative times and complication rates have been comparable to traditional laparoscopic approaches.
After gaining substantial experience with LESS techniques, several authors have now reported the use of LESS with more time-sensitive surgeries such as donor nephrectomy and partial nephrectomy. A retrospective comparative study between 17 LESS donor nephrectomies and 17 matched patients undergoing a standard laparoscopic approach has now been reported (Canes et al, 2010). All LESS procedures were completed with the use of a purpose-specific access device; at the discretion of the surgeon, an adjunctive 2-mm needlescopic port was used to aid in retraction, hilar dissection, and extraction (Fig. 55–42). Mean warm ischemia time was longer in the LESS group (3 vs. 6.1 minutes), while blood loss and operative times were similar. The median length of harvested renal artery, vein, and ureter were 3.2 cm, 3.6 cm, and 14.9 cm, respectively, in the LESS cohort; measurements for standard laparoscopy were not reported. Complications included one corneal abrasion and one postoperative allograft thrombosis. A more rapid return to convalescence was noted in the LESS donor nephrectomy group, with fewer days on oral pain medication, faster return to work, and days to “100% recovery” (self determined). LESS Pfannenstiel donor nephrectomy has also been reported and may add to the cosmetic benefit afforded by the single-site approach (Andonian et al, 2009). Prospective studies are certainly needed to determine if this approach is warranted in this patient population.
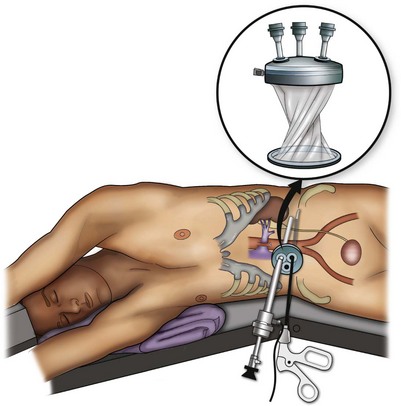
Figure 55–42 Laparoendoscopic single-site surgery donor nephrectomy using a purpose-specific device with multichannel instrument access. A 2-mm instrument is also used to aid in retraction, hilar dissection, and extraction.
An initial series of LESS partial nephrectomy was reported in four patients with a median tumor size of 3-cm (range, 1 to 5.9 cm) (Aron et al, 2009). Median operating time was 270 minutes, and median blood loss was 150 cc. Median warm ischemia time was 20 minutes (range 11 to 29 minutes), and median hospital stay was 3 days. One patient had postoperative hemorrhage and pulmonary embolism.
The two largest LESS series each encompass 100 patients, where LESS has been employed to perform a variety of urologic surgeries (Desai et al, 2009; White et al, 2009). The cases performed have included nephrectomy (simple, partial, radical, and donor), nephroureterectomy, renal cyst excision, renal tumor cryoablation, pyeloplasty, ureteroneocystostomy, ileal ureter, adrenalectomy, hysterectomy, varicocelectomy, radical prostatectomy, transvesical simple prostatectomy, radical cystectomy, sacral colpopexy, and mesh sling removal. These series demonstrate the breadth of urologic procedures that can be performed with LESS techniques.
Raman and colleagues (2009) reported the first comparative study between traditional laparoscopic and LESS applications. The investigators compared 11 LESS nephrectomies with 22 standard laparoscopic nephrectomies. The groups were matched at a 2 : 1 ratio for patient age, surgical indication, and tumor size. Similar outcomes between the two groups were noted regarding operative time (122 vs. 125 minutes), analgesic use, length of stay, and complication rate.
Robotic LESS (R-LESS) Surgery
The da Vinci-S Robotic System (Intuitive Surgical, Sunnyvale, CA) has been used in conjunction with a Tri-Port single-port access device to perform radical prostatectomy, pyeloplasty, and radical nephrectomy (Kaouk et al, 2009). A 12-mm 3D camera and 5-mm grasper were placed through the Tri-Port, while an additional 8-mm port was placed alongside the port in the same incision for another robotic instrument. Operative times (radical prostatectomy = 5 hours, pyeloplasty = 4.5 hours, radical nephrectomy = 2.5 hours), estimated blood loss (80 to 250 cc), and hospital stay (2 days) were acceptable. Significant “clashing” of robotic arms was noted. R-LESS surgery has also been performed using the GelPort laparoscopic access system (Stein et al, 2010). The authors reported more adequate spacing for the robotic instruments and easier assistant access. However, the need for novel purpose-specific, possibly flexible, robotic instrumentation or platforms was noted in both studies.
The prospect of LESS surgery is potentially attractive from the patient perspective, with possible benefit in decreasing postoperative pain and improving cosmesis. This may be especially true in the case of extirpative surgery, when an extraction site is required or if cosmesis is of particular concern. However, these potential benefits have not yet been proven in prospective trials. Moreover, although a large body of surgical literature clearly demonstrates benefits of traditional laparoscopy versus open surgical techniques for analgesic requirements, length of stay, return to convalescence, and cosmesis, the benefits of LESS versus traditional laparoscopy remain in question at this time. Larger comparative series between traditional laparoscopic techniques and LESS are required to further study outcomes relevant to each specific procedure and to evaluate for potential benefits.
Complications of Laparoscopic Renal Surgery
Complications are an unavoidable consequence of surgical practice and even the most experienced clinicians will face problems. Organic factors related to the patient, operating room environment, and chaotic forces can lead to an untoward event. Thus efforts at prevention through knowledge of each given procedure and its potential pitfalls should be maximized. Moreover, patient education about the risks of surgery is essential.
Patient selection is important to minimize risk of complications. This needs to be paired with each surgeon’s experience and ability. Several situations require caution when considering a laparoscopic approach. True contraindications include an uncorrected coagulopathy, untreated infection, and hypovolemic shock (Capelouto and Kavoussi, 1993). Previous surgery is not a contraindication to laparoscopic renal surgery. Prior abdominal surgery may have caused intra-abdominal adhesions and increased the possibility of injury to bowel during insufflation, trocar placement, or dissection. The initial entry site in these patients should be away from scars and prior surgical fields. Open trocar placement or a retroperitoneal approach, may be necessary to minimize access injuries (Hasson, 1971). Extraperitoneal access may also help avoid adhesions. If additional space is required, the peritoneum may be opened under direct vision.
Patients with large, dilated loops of bowel from either functional or obstructive ileus should be approached cautiously, because the dilated intestinal segments can limit the working space and may be injured during access, dissection, and trocar site closure (Borten, 1986).
Care is taken to keep anatomic orientation at all times, because confusion in landmarks can lead to catastrophic consequences. Prior surgery or bulky pathology can alter normal anatomic relationships. It is thus necessary to have preoperative imaging studies available in the room. Intraoperative ultrasonography can be a valuable tool to help identify structures. When visual clues are inadequate to allow safe progress, palpation by hand assistance may provide additional information. Alternatively, open conversion may be indicated.
When complications occur, the consequences can often be minimized through early recognition and appropriate intervention. Laparoscopic renal surgery shares several potential risks with traditional open approaches; however, there are differences in the type and presentation of these complications. It must be kept in mind that all situations are individual, and unique problems may arise and call for innovative actions.
General complications of laparoscopic surgery have been covered in Chapter 9, however, specific pitfalls require review. Reported complications from laparoscopic kidney surgery are reviewed in Table 55–5. The combined incidence of bowel injury in the urologic literature is 0.8%, and injury may occur at any point during the dissection (Schwartz et al, 2010). When reflecting the colon or duodenum, avoid thermal energy adjacent to the bowel. This is the most common cause of unrecognized injury and may not be diagnosed until postoperative day 3 to 5. When recognized, superficial thermal injuries may be oversewn with 3-0 silk suture to imbricate the affected area. Transmural injuries should be debrided and, as with primary sharp injury, may be closed primarily in two layers. The area should be irrigated thoroughly and inspected to rule out a through and through injury. Drain placement is optional and oral intake is held until bowel function has returned.
Table 55–5 Reported Complications of Laparoscopic Kidney Surgery
One of the most devastating complications occurring as a result of laparoscopic surgery is unrecognized bowel injury (Fig. 55–43). Only a small portion of the laparoscopic instrument is in the visual field so that injuries can occur out of the surgeon’s view during introduction or retraction. In the urologic literature, the overall incidence of bowel injury during laparoscopic surgery of the retroperitoneum, both recognized and unrecognized, is 0.65% (Schwartz et al, 2010). Unrecognized injuries result in high-grade complications in 100% of cases in series reporting on laparoscopic bowel injuries. Blunt, sharp, and cautery dissection account for the majority of bowel injuries (60%), while access-related injuries are far less common (6%). The presentation of bowel injuries in patients undergoing laparoscopy differs from that described with open surgery. Patients with unrecognized bowel injury after laparoscopy typically present with persistent and increased trocar-site pain at the site closest to the bowel injury. The area around this site becomes edematous and doughy in consistency. Signs and symptoms may also include abdominal distention, nausea, diarrhea, anorexia, low-grade fever, persistent bowel sounds, and a low or normal white blood cell count. The patient’s condition can rapidly deteriorate to hemodynamic instability and death, if the injury is not recognized and treated (Bishoff et al, 1999). CT with oral contrast is the initial diagnostic modality of choice (Cadeddu et al, 1997), and open exploration is usually required to evacuate bowel spillage and perform the necessary repair. In rare cases, when a controlled fistula develops, conservative management with bowel rest and hyperalimentation may be used, but this can take months to resolve.
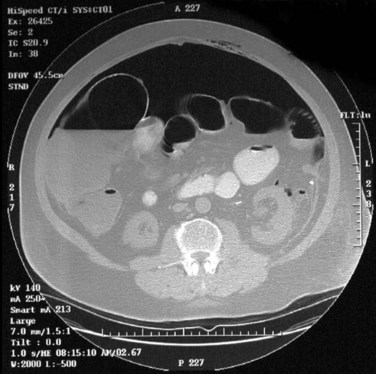
Figure 55–43 CT scan taken 9 days after partial nephrectomy when the patient presented to the clinic for routine follow-up complaining of distention and worsening abdominal pain for the past 3 days, low-grade fever, leukopenia, and pain out of proportion at a single trocar site. CT shows dilated loops of large bowel and significant amounts of free air. Exploration revealed a small perforation in the cecum.
In reflecting the bowel on the left side, care must be taken to avoid making a hole in the mesentery. Any mesenteric defects should be closed as postoperative bowel herniation is possible (Regan et al, 2003). During closure of the mesentery, care also should be taken to avoid compromising the vascular supply to the colon. Retractors not in the operative field may also injure the bowel, and one should check for inadvertent injury at the conclusion of the procedure.
Vascular injuries are the most common complication of urologic laparoscopy (Permpongkosol et al, 2007). Life-threatening vascular injuries can occur during laparoscopic renal surgery and usually occur during dissection of the renal hilum. Injury to arteries, veins, branches, and accessory vessels can result in bleeding that may require conversion to open surgery. The renal vein can have multiple branches that can easily be torn. Care should be taken in assuring ligation and transection without tension. Venous bleeding can be brisk. Many times, applying direct pressure with gauze for several minutes will be sufficient to control bleeding. Resist the temptation to continually explore the area of venous bleeding, if all is quiescent once the gauze is removed. On the right side, the vena cava can be injured. Avulsion of the gonadal or adrenal vein can cause significant bleeding. If a hole is visible, placement of a clip or suture may be attempted once a grasper has controlled the situation. Blind clip placement or suturing can lead to a worsening of the situation and additional complications. Again, direct pressure with gauze over several minutes may abate bleeding. Dissection may continue with the gauze in place.
Arterial injuries can occur when structures are not fully identified before transection. Also, past pointing of scissors can cut an underlying vessel. If the opening is identified, suture placement or clips may be used for control. A hand may be placed in a lower abdominal midline incision to hold pressure if bleeding is brisk. In this manner, laparoscopic suturing or open conversion can proceed in a controlled manner.
Cases of inadvertent stapling of important anatomic structures have been reported. The vena cava and aorta have been mistaken for the renal vessels (McAllister et al, 2004). Several instances of transection of the small mesenteric artery (SMA) or contralateral renal vessels have also occurred. This can occur readily with the novice who is unfamiliar with the retroperitoneal approach. Unfortunately, many of these are not recognized intraoperatively and mortality is high. The best way to avoid this is through continuous anatomic orientation and vigilant self-questioning.
Equipment failure can result in bleeding. A multi-institutional review of endovascular stapler complications showed a malfunction rate of 1.7% (10/565), with eight cases involving the renal vein and two cases the renal artery (Chan et al, 2000). Blood loss resulting from the malfunction was between 200 to 1200 cc. Conversion to open surgery for hemostasis was required in 20% of the malfunction cases. The etiology of stapler failure was due directly to the instrument in three cases and due to preventable causes in seven cases. Preventable causes included stapling over clips or incomplete transection due to incorrect placement. The abdominal cavity should be inspected for bleeding at the conclusion of surgery. Common areas of postoperative intra-abdominal bleeding include the bed of the dissection, adrenal gland, mesentery, gonadal vessels, and ureteral stump.
Postoperative hemorrhage can occur following partial nephrectomy. Hypotension with associated tachycardia and a drop in hematocrit may imply postoperative bleeding. Following partial nephrectomy, an arteriovenous malformation or pseudoaneurysm may form (Benway et al, 2009; Shapiro et al, 2009). These patients present with persistent gross hematuria, hypotension, and tachycardia. If there is a question, CT scanning may be appropriate in identifying the source of bleeding; however, in the majority of patients, immediate renal angiography with embolization of the bleeding site is indicated.
Persistent urine leakage may occur following a partial nephrectomy or cyst ablation. Unless there is distal obstruction to the site of leakage, most will resolve with conservative therapy after several weeks (Meeks et al, 2008). After approximately one week, a controlled fistula develops, and the drain can be taken off continous suction and checked intermittantly to be sure fluid is not accumulating. If conservative management fails, such as in cases of distal obstruction, additional intervention may be required, such as percutaneous drainage of a urinoma or ureteral stent and bladder decompression.
Upper pole renal dissection can result in diaphragmatic injury. This is usually immediately recognized because peak airway pressures suddenly increase, and ventilation of the patient becomes difficult. The diaphragm can be seen billowing on endoscopic inspection. Immediate treatment is needed to prevent a tension pneumothorax. The diaphragm can be sutured directly while a central line catheter is placed into the ipsilateral anterior second intercostals space and placed to a water seal. At the conclusion of the procedure, the patient is ventilated, a chest radiograph obtained, and, if resolved, the catheter is removed. When significant gas remains, a chest tube can be inserted (Del Pizzo et al, 2003; Aron et al, 2007).
On the left side, splenic and pancreatic injuries may occur. Bleeding from the spleen is usually controlled with topical agents and argon beam coagulations (Canby-Hagino et al, 2000; McGinnis et al, 2000). Injuries to the pancreas may be insidious, and inspection is needed at the conclusion of surgery. Superficial pancreatic injuries are observed with drain placement. Deeper injuries may require formal repair or isolation of the segment with a GIA stapler (Varkarakis et al, 2004). Right-sided dissections may cause injury to the liver or gall bladder. Liver injuries are managed with topical therapy and argon beam coagulation. Gall bladder injuries are best managed by cholecystectomy.
Patients undergoing laparoscopic renal surgery are at risk of intravascular volume overload if fluid replacement is not modified relative to open surgery. The laparoscopic approach is associated with far less insensible fluid loss compared with open procedures, and there is also a vascular-mediated oliguria. As such, urine output should not be a barometer of fluid status. Typically, intravenous fluids should be minimized with the exception of laparoscopic donor nephrectomy. Aggressive replacement can cause volume overload in patients with diminished cardiac reserve and can result in postoperative congestive heart failure. Poor urine output or hemodynamic instability in the postoperative period should initiate an evaluation to rule out bleeding; if the work-up is negative, diuresis can be induced if clinically indicated.
Several authors have reported cases of chronic pain syndrome or nerve injury after laparoscopic radical nephrectomy. Patients may experience a burning discomfort in the ipsilateral flank; paresthesias around port sites or over the thigh and upper extremity can occur (Wolf et al, 2000; Oefelein and Bayazit, 2003). In a series of 381 laparoscopic donor nephrectomies, ipsilateral orchialgia was reported in 10% of patients (Kim et al, 2003). Onset of pain occurred at a mean of 5 days after surgery (range 6 to 52 months) and at 6 months; 50% had complete spontaneous resolution. Thigh paresthesias may be avoided by preserving the psoas fascia during posterior renal dissection. Additional reported complications include incisional hernia after intact specimen removal, port-site hernia, prolonged ileus, pulmonary embolus, and pneumonia.
In a multi-institutional review of 185 patients, Gill and coworkers (1995) reported an overall complication rate of 12% for benign disease, with 5% of patients requiring conversion to open surgery. In their series, the incidence of complications decreased markedly with increasing experience. In fact, 70% of the complications occurred during the first 20 cases at each institution. A learning curve of approximately 20 laparoscopic nephrectomy cases is also supported by other reports (Keeley and Tolley, 1998; Rassweiler et al 1998; Fahlenkamp et al, 1999). In a recently reported series of partial nephrectomies, the complication rate continued to decrease even after 750 cases. This implies a longer learning curve for more complex procedures.
In a series of 482 laparoscopic nephrectomies (444 procedures for benign disease) performed by 20 surgeons at 14 different European medical centers, a 6% overall complication rate was reported, with 10% of the cases converted to open surgery (Rassweiler et al, 1998). The majority of patients converted to open surgery had infectious causes of renal abnormality as the leading indication for kidney removal. Bleeding was the most common cause of open conversion in these cases, followed by the surgeon’s inability to visualize the renal hilum for dissection.
In series comparing open, hand-assisted, and laparoscopic nephrectomy for malignancy, the complication rates were 10%, 17%, and 12% respectively (P = .133) (Chan et al, 2001; Shuford et al, 2004).
Two comparative studies examining complication rates in the elderly population, greater than 75 and greater than 80 years, respectively, found no difference in surgical or long-term morbidity when compared with younger patient populations (Varkarakis et al, 2004b; Thomas et al, 2009). In patients at high risk for perioperative complications, as determined by an American Society of Anesthesiologists score greater than or equal to 3, there were no significant differences in complication rates between hand-assisted, laparoscopy, and open radical nephrectomy (Baldwin et al, 2003).
Penetrance of Laparoscopic Renal Surgery for the Community Urologist
A recent study using information gathered from the SEER-Medicare–linked database showed the serious underutilization of laparoscopic and nephron-sparing techniques (Miller et al, 2008). Of 4872 radical nephrectomies performed for kidney cancer between 1997 and 2002, only 515 (10.6%) were performed laparoscopically. After controlling for variables such as demographics, tumor size, and comorbidities, surgeon-attributable factors were consistently the most significant predictor of the type of surgery performed. Stated otherwise, the surgeon’s practice style was the determining factor for the surgical approach, not patient characteristics or characteristics of the renal cancer. This conclusion should be cause for some alarm in the urologic community. Laparoscopic procedures such as cholecystectomy and appendectomy are quite commonplace and were rapidly adopted by general surgeons. Historically, urologists have been receptive to new technology, a fact most recently demonstrated by the rapid increase in the number of robotic prostatectomies performed in the United States. However, the same does not hold true for laparoscopic nephrectomy, despite its longevity and proven benefits. This would suggest significant barriers to the diffusion of laparoscopy specifically for renal surgery. A complex array of reasons may account for this observation, including the differential incidence of kidney and prostate cancer, marketing of robotics, referral patterns, consumer demand, etc. (Richstone et al, 2008). As physicans, we must be aware of barriers that affect the use of procedures with proven and meaningful patient benefits, and work toward dismantling them such that every patient may gain from progress in our field.
Summary
Laparoscopy has emerged as the preferred treatment modality for many types of renal pathology. Patients have undoubtedly gained from the benefits laparoscopy offers in terms of perioperative morbidity without sacrificing therapeutic outcomes. As surgical tools continue to evolve, even more minimally invasive options such as laparoendoscopic single-site surgery and natural orifice transluminal endoscopic surgery may become more pervasive and potentially offer additional perioperative benefit to patients.
Key Points
Berger A, Brandina R, Atalla MA, et al. Laparoscopic radical nephrectomy for renal cell carcinoma: oncological outcomes at 10 years or more. J Urol. 2009;182:2172-2176.
Bishoff JT, Allaf ME, Kirkels W, et al. Laparoscopic bowel injury: incidence and clinical presentation. J Urol. 1999;161:887-890.
Blom JH, van Poppel H, Maréchal JM, et al. Radical nephrectomy with and without lymph-node dissection: final results of European Organization for Research and Treatment of Cancer (EORTC) Randomized Phase 3 Trial 30881. Eur Urol. 2009;55:28-34.
Blute ML, Leibovich BC, Cheville JC, et al. A protocol for performing extended lymph node dissection using primary tumor pathological features for patients treated with radical nephrectomy for clear cell renal cell carcinoma. J Urol. 2004;172:465-469.
Fahlenkamp D, Rassweiler J, Fornara P, et al. Complications of laparoscopic procedures in urology: experience with 2,407 procedures at 4 German centers. J Urol. 1999;162:765-770.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178:41-46.
Huang WC, Levey AS, Serio AM, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7:735-740.
Kunkle DA, Egleston BL, Uzzo RG. Excise, ablate or observe: the small renal mass dilemma—a meta-analysis and review. J Urol. 2008;179:1227-1233.
Lane BR, Gill IS. Seven-year oncological outcomes after laparoscopic and open partial nephrectomy. J Urol. 2010;183:473-479.
Miller DC, Saigal CS, Banerjee M, et al. Diffusion of surgical innovation among patients with kidney cancer. Cancer. 2008;112:1708-1717.
Permpongkosol S, Link RE, Su LM, et al. Complications of 2,775 urological laparoscopic procedures: 1993 to 2005. J Urol. 2007;177:580-585.
Rassweiler J, Tsivian A, Kumar AV, et al. Oncologic safety of laparoscopic surgery for urological malignancy: experience with more than 1,000 operations. J Urol. 2003;169:2072-2075.
Yossepowitch O, Thompson RH, Leibovich BC, et al. Positive surgical margins at partial nephrectomy: predictors and oncological outcomes. J Urol. 2008;179:2158-2163.
Chatterjee S, Nam R, Fleshner N, et al. Permanent flank bulge is a consequence of flank incision for radical nephrectomy in one half of patients. Urol Oncol. 2004;22:36-39.
Clayman RV, Kavoussi LR, Soper NJ, et al. Laparoscopic nephrectomy: initial case report. J Urol. 1991;146:278-282.
Dunn MD, Portis AJ, Shalhav AL, et al. Laparoscopic versus open radical nephrectomy: a 9-year experience. J Urol. 2000;164:1153-1159.
Gill IS, Matin SF, Desai MM, et al. Comparative analysis of laparoscopic versus open partial nephrectomy for renal tumors in 200 patients. J Urol. 2003;170:64-68.
Kerbl K, Clayman RV, McDougall EM, et al. Laparoscopic nephrectomy: the Washington University experience. Br J Urol. 1994;73:231-236.
Kobayashi T, Terai A, Yoshimura K, et al. Assessment of body image alteration after renal surgery using a novel three-dimensional laser scanner. Urology. 2004;64:264-268.
Patient Evaluation and Preparation
Cadeddu JA, Chan DY, Hedican SP, et al. Retroperitoneal access for transperitoneal laparoscopy in patients at high risk for intra-abdominal scarring. J Endourol. 1999;13:567-570.
Chen RN, Moore RG, Cadeddu JA, et al. Laparoscopic renal surgery in patients at high risk for intra-abdominal or retroperitoneal scarring. J Endourol. 1998;12:143-147.
Fugita OE, Chan DY, Roberts WW, et al. Laparoscopic radical nephrectomy in obese patients: outcomes and technical considerations. Urology. 2004;63:247-252.
Hedican SP, Moon TD, Lowry PS, et al. Hand-assisted laparoscopic renal surgery in the morbidly and profoundly obese. J Endourol. 2004;18:241-244.
Kapoor A, Nassir A, Chew B, et al. Comparison of laparoscopic radical renal surgery in morbidly obese and non-obese patients. J Endourol. 2004;18:657-660.
Mannucci PM, Remuzzi G, Pusineri F, et al. Deamino-8-D-arginine vasopressin shortens the bleeding time in uremia. N Engl J Med. 1983;308:8-12.
Pruthi RS, Kang J, Vick R. Desmopressin induced hyponatremia and seizures after laparoscopic radical nephrectomy. J Urol. 2002;168:187.
Anesthesic Considerations in Laparoscopy
Arthure H. Laparoscopy hazard. Br Med J. 1970;4:492-493.
Herati AS, Atalla MA, Rais-Bahrami S, et al. A new valveless trocar for urologic laparoscopy: initial evaluation. J Endourol. 2009;23:1535-1539.
Hodgson C, McClelland RM, Newton JR. Some effects of the peritoneal insufflation of carbon dioxide at laparoscopy. Anaesthesia. 1970;25:382-390.
Lew JK, Gin T, Oh TE. Anaesthetic problems during laparoscopic cholecystectomy. Anaesth Intensive Care. 1992;20:91-92.
Makarov DV, Kainth D, Link RE, et al. Physiologic changes during helium insufflation in high-risk patients during laparoscopic renal procedures. Urology. 2007;70:35-37.
Monk TG, Weldon BC. Anesthetic considerations for laparoscopic surgery. J Endourol. 1992;6:89.
Nunn J. Respiratory aspects of anesthesia. In: Nunn J, editor. Applied respiratory physiology. London: Butterworths; 1987:350.
Wolf JSJr, Clayman RV, McDougall EM, et al. Carbon dioxide and helium insufflation during laparoscopic radical nephrectomy in a patient with severe pulmonary disease. J Urol. 1996;155:2021.
Considerations in Obese Patients
Doublet J, Belair G. Retroperitoneal laparoscopic nephrectomy is safe and effective in obese patients: a comparative study of 55 procedures. Urology. 2000;56:63-66.
Fazeli-Matin S, Gill IS, Hsu TH, et al. Laparoscopic renal and adrenal surgery in obese patients: comparison to open surgery. J Urol. 1999;162:665-669.
Jacobs SC, Cho E, Dunkin BJ, et al. Laparoscopic nephrectomy in the markedly obese living renal donor. Urology. 2000;56:926-929.
Kapoor A, Nassir A, Chew B, et al. Comparison of laparoscopic radical renal surgery in morbidly obese and non-obese patients. J Endourol. 2004;18:657-660.
Mendoza D, Newman RC, Albala D, et al. Laparoscopic complications in markedly obese urologic patients (a multi-institutional review). Urology. 1996;48:562-567.
Montgomery JS, Johnston WK3rd, Wolf JSJr. Wound complications after hand assisted laparoscopic surgery. J Urol. 2005;174:2226-2230.
Troppmann C, Perez RV. Rhabdomyolysis associated with laparoscopic live donor nephrectomy and concomitant surgery: a note of caution. Am J Transplant. 2003;3:1457-1458.
Surgical Approaches and Obtaining Access
Alcaraz A, Peri L, Molina A, et al. Feasibility of transvaginal NOTES-assisted laparoscopic nephrectomy. Eur Urol. 2010;57:233-237.
Aron M, Koenig P, Kaouk JH, et al. Robotic and laparoscopic partial nephrectomy: a matched-pair comparison from a high-volume centre. BJU Int. 2008;102:86-92.
Benway BM, Wang AJ, Cabello JM, et al. Robotic partial nephrectomy with sliding-clip renorrhaphy: technique and outcomes. Eur Urol. 2009;55:592-599.
Box G, Averch T, Cadeddu J, et al. Nomenclature of natural orifice translumenal endoscopic surgery (NOTES) and laparoendoscopic single-site surgery (LESS) procedures in urology. J Endourol. 2008;22:2575-2581.
Branco AW, Branco Filho AJ, Kondo W, et al. Hybrid transvaginal nephrectomy. Eur Urol. 2008;53:1290-1294.
Deane LA, Lee HJ, Box GN, et al. Robotic versus standard laparoscopic partial/wedge nephrectomy: a comparison of intraoperative and perioperative results from a single institution. J Endourol. 2008;22:947-952.
Kaouk JH, White WM, Goel RK, et al. NOTES transvaginal nephrectomy: first human experience. Urology. 2009;74:5-8.
Monga M, Premoli J, Skemp N, et al. Forearm compression by laparoscopic hand-assist devices. J Endourol. 2004;18:654-656.
Nakada SY, Moon TD, Gist M, et al. Use of the pneumo sleeve as an adjunct in laparoscopic nephrectomy. Urology. 1997;49:612-613.
Palanivelu C, Rajan PS, Rangarajan M, et al. Transvaginal endoscopic appendectomy in humans: a unique approach to NOTES-world’s first report. Surg Endosc. 2008;22:1343-1347.
Raman JD, Bagrodia A, Cadeddu JA. Single-incision, umbilical laparoscopic versus conventional laparoscopic nephrectomy: a comparison of perioperative outcomes and short-term measures of convalescence. Eur Urol. 2009;55:1198-1204.
Rao GV, Reddy DN. Transgastric appendectomy in humans. Presented at the World Congress of Gastroenterology meeting, Montreal, Canada, September 2006.
Sotelo R, de Andrade R, Fernández G, et al. NOTES hybrid transvaginal radical nephrectomy for tumor: stepwise progression toward a first successful clinical case. Eur Urol. 2010;57:138-144.
Zorron R, Filgueiras M, Maggioni LC, et al. NOTES. Transvaginal cholecystectomy: report of the first case. Surg Innov. 2007;14:279-283.
Baba S, Nakagawa K, Nakamura K, et al. Experience of 143 cases of laparoscopic surgery in urology–clinical outcome in comparison to open surgery. Nippon Hinyokika Gakkai Zasshi. 1996;87:842-850.
Bercowsky E, Shalhav AL, Portis A, et al. Is the laparoscopic approach justified in patients with xanthogranulomatous pyelonephritis? Urology. 1999;54:437-442.
Gupta NP, Agrawal AK, Sood S. Tubercular pyelonephritic nonfunctioning kidney—another relative contraindication for laparoscopic nephrectomy: a case report. J Laparoendosc Adv Surg Tech A. 1997;7(2):131-134.
Kerbl K, Clayman RV, McDougall EM, et al. Transperitoneal nephrectomy for benign disease of the kidney: a comparison of laparoscopic and open surgical techniques. Urology. 1994;43:607-613.
Kerbl K, Clayman RV, McDougall EM, et al. Laparoscopic nephrectomy: the Washington University experience. Br J Urol. 1994;73:231-236.
Landman J, Collyer WC, Olweny E, et al. Laparoscopic renal ablation: an in vitro comparison of currently available electrical tissue morcellators. Urology. 2000;56:677-681.
Lee KS, Kim HH, Byun SS, et al. Laparoscopic nephrectomy for tuberculous nonfunctioning kidney: comparison wth laparoscopic simple nephrectomy for other diseases. Urology. 2002;60:411-414.
Moore RG, Chen RN, Hedican SP. Laparoscopic subcapsular nephrectomy. J Endourol. 1998;12:263-264.
Nicol DL, Winkle DC, Nathanson LK, et al. Laparoscopic nephrectomy for benign renal disease. Br J Urol. 1994;73:237-241.
Parra RO, Perez MG, Boullier JA, et al. Comparison between standard flank versus laparoscopic nephrectomy for benign renal disease. J Urol. 1995;153:1171-1173.
Pautler SE, Harrington FS, McWilliams GW, et al. A novel laparoscopic specimen entrapment device to facilitate morcellation of large renal tumors. Urology. 2002;59:591-593.
Rassweiler J, Frede T, Henkel TO, et al. Nephrectomy: a comparative study between the transperitoneal and retroperitoneal laparoscopic versus the open approach. Eur Urol. 1998;33:489-496.
Rosoff JS, Raman JD, Del Pizzo JJ. Feasibility of laparoscopic approach in management of xanthogranulomatous pyelonephritis. Urology. 2006;68:711-714.
Urban DA, Kerbl K, McDougall EM, et al. Organ entrapment and renal morcellation: permeability studies. J Urol. 1993;150:1792-1794.
Bendavid Y, Moloo H, Klein L, et al. Laparoscopic nephrectomy for autosomal dominant polycystic kidney disease. Surg Endosc. 2004;18:751-754.
Brown JA, Torres VE, King BF, et al. Laparoscopic marsupialization of symptomatic polycystic kidney disease. J Urol. 1996;156:22-27.
Camargo AH, Cooperberg MR, Ershoff BD, et al. Laparoscopic management of peripelvic renal cysts: University of California, San Francisco, experience and review of literature. Urology. 2005;65:882-887.
Carrim ZI, Murchison JT. The prevalence of simple renal and hepatic cysts detected by spiral computed tomography. Clin Radiol. 2003;58:626-629.
Cherullo EE, Hobart MG, Chow GK, et al. Caliceal injury during laparoscopic cyst decortication in adult polycystic kidney disease. J Urol. 1999;162:1367-1368.
Cloix P, Martin X, Pangaud C, et al. Surgical management of complex renal cysts: a series of 32 cases. J Urol. 1996;156:28-30.
Desai PJ, Castle EP, Daley SM, et al. Bilateral laparoscopic nephrectomy for significantly enlarged polycystic kidneys: a technique to optimize outcome in the largest of specimens. BJU Int. 2008;101:1019-1023.
Doumas K, Skrepetis K, Lykourinas M. Laparoscopic ablation of symptomatic peripelvic renal cysts. J Endourol. 2004;18:45-48.
Dunn MD, Portis AJ, Naughton C, et al. Laparoscopic cyst marsupialization in patients with autosomal dominant polycystic kidney disease. J Urol. 2001;165:1888-1892.
Gill IS, Kaouk JH, Hobart MG, et al. Laparoscopic bilateral synchronous nephrectomy for autosomal dominant polycystic kidney disease: the initial experience. J Urol. 2001;165:1093-1098.
Hoenig DM, McDougall EM, Shalhav AL, et al. Laparoscopic ablation of peripelvic renal cysts. J Urol. 1997;158:1345-1348.
Hulbert JC, Hunter D, Young AT, et al. Percutaneous intrarenal marsupialization of a perirenal cystic collection—endocystolysis. J Urol. 1988;139:1039-1041.
Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology. 2005;66:484-488.
Jenkins MA, Crane JJ, Munch LC. Bilateral hand-assisted laparoscopic nephrectomy for autosomal dominant polycystic kidney disease using a single midline HandPort incision. Urology. 2002;59:32-36.
Laucks SPJr, McLachlan MS. Aging and simple renal cysts of the kidney. Br J Radiol. 1981;54:12-14.
Lee DI, Andreoni CR, Rehman J, et al. Laparoscopic cyst decortication in autosomal dominant polycystic kidney disease: impact on pain, hypertension, and renal function. J Endourol. 2003;17:345-354.
Lifson BJ, Teichman JM, Hulbert JC. Role and long-term results of laparoscopic decortication in solitary cystic and autosomal dominant polycystic kidney disease. J Urol. 1998;159:702-705.
Limb J, Santiago L, Kaswick J, et al. Laparoscopic evaluation of indeterminate renal cysts: long-term follow-up. J Endourol. 2002;16:79-82.
McDougall EM. Approach to decortication of simple cysts and polycystic kidneys. J Endourol. 2000;14:821-827.
Nieh PT, Bihrle W3rd. Laparoscopic marsupialization of massive renal cyst. J Urol. 1993;150:171-173.
Rané A. Laparoscopic management of symptomatic simple renal cysts. Int Urol Nephrol. 2004;36:5-9.
Rehman J, Landman J, Andreoni C, et al. Laparoscopic bilateral hand assisted nephrectomy for autosomal dominant polycystic kidney disease: initial experience. J Urol. 2001;166:42-47.
Roberts WW, Bluebond-Langner R, Boyle KE, et al. Laparoscopic ablation of symptomatic parenchymal and peripelvic renal cysts. Urology. 2001;58:165-169.
Rubenstein SC, Hulbert JC, Pharand D, et al. Laparoscopic ablation of symptomatic renal cysts. J Urol. 1993;150:1103-1106.
Santiago L, Yamaguchi R, Kaswick J, et al. Laparoscopic management of indeterminate renal cysts. Urology. 1998;52:379-383.
Wehle MJ, Grabstald H. Contraindications to needle aspiration of a solid renal mass: tumor dissemination by renal needle aspiration. J Urol. 1986;136:446-448.
Wolf JSJr. Evaluation and management of solid and cystic renal masses. J Urol. 1998;159:1120-1133.
Renal Biopsy for Benign Medical Renal Disease
Gault MH, Muehrcke RC. Renal biopsy: current views and controversies. Nephron. 1983;34:1-34.
Manaligod JR, Pirani CL. Renal biopsy in 1985. Semin Nephrol. 1985;5:237-239.
Morel-Maroger L. The value of renal biopsy. Am J Kidney Dis. 1982;1:244-248.
Shetye KR, Kavoussi LR, Ramakumar S, et al. Laparoscopic renal biopsy: a 9-year experience. BJU Int. 2003;91:817-820.
Pyelolithotomy, Nephrolithotomy, and Ureterolithotomy
Badani KK, Hemal AK, Fumo M, et al. Robotic extended pyelolithotomy for treatment of renal calculi: a feasibility study. World J Urol. 2006;24:198-201.
Ball AJ, Leveillee RJ, Patel VR, et al. Laparoscopic pyeloplasty and flexible nephroscopy: simultaneous treatment of ureteropelvic junction obstruction and nephrolithiasis. JSLS. 2004;8(3):223-228.
Chang TD, Dretler SP. Laparoscopic pyelolithotomy in an ectopic kidney. J Urol. 1996;156:1753.
Eshghi AM, Roth JS, Smith AD. Percutaneous transperitoneal approach to a pelvic kidney for endourological removal of staghorn calculus. J Urol. 1985;134:525-527.
Gaur DD, Trivedi S, Prabhudesai MR, et al. Laparoscopic ureterolithotomy: technical considerations and long-term follow-up. BJU Int. 2002;89:339-343.
Goel A, Hemal AK. Evaluation of role of retroperitoneoscopic pyelolithotomy and its comparison with percutaneous nephrolithotripsy. Int Urol Nephrol. 2003;35:73-76.
Harmon WJ, Kleer E, Segura JW. Laparoscopic pyelolithotomy for calculus removal in a pelvic kidney. J Urol. 1996;155:2019-2020.
Hemal AK, Goel A, Goel R. Minimally invasive retroperitoneoscopic ureterolithotomy. J Urol. 2003;169:480-482.
Hoenig DM, Shalhav AL, Elbahnasy AM, et al. Laparoscopic pyelolithotomy in a pelvic kidney: a case report and review of the literature. JSLS. 1997;1:163-165.
Jordan GH, McCammon KA, Robey EL. Laparoscopic pyelolithotomy. Urology. 1997;49:131-134.
Kaouk JH, Gill IS, Desai MM, et al. Laparoscopic anatrophic nephrolithotomy: feasibility study in a chronic porcine model. J Urol. 2003;169:691-696.
Simforoosh N, Aminsharifi A, Tabibi A, et al. Laparoscopic anatrophic nephrolithotomy for managing large staghorn calculi. BJU Int. 2008;101:1293-1296.
Stein RJ, Turna B, Nguyen MM, et al. Laparoscopic pyeloplasty with concomitant pyelolithotomy: technique and outcomes. J Endourol. 2008;22:1251-1255.
Tóth C, Holman E, Pásztor I, et al. Laparoscopically controlled and assisted percutaneous transperitoneal nephrolithotomy in a pelvic dystopic kidney. J Endourol. 1993;7:303-305.
Troxel SA, Low RK, Das S. Extraperitoneal laparoscopy-assisted percutaneous nephrolithotomy in a left pelvic kidney. J Endourol. 2002;16:655-657.
Boylu U, Lee BR, Thomas R. Robotic-assisted laparoscopic pyeloplasty and nephropexy for ureteropelvic junction obstruction and nephroptosis. J Laparoendosc Adv Surg Tech A. 2009;19:379-382.
Chueh SC, Hsieh JT, Chen J, et al. Retroperitoneoscopic nephropexy for symptomatic nephroptosis. Surg Endosc. 2002;16:1603-1607.
Gözen AS, Rassweiler JJ, Neuwinger F, et al. Long-term outcome of laparoscopic retroperitoneal nephropexy. J Endourol. 2008;22:2263-2267.
Hübner WA, Schlarp O, Riedl C, et al. Laparoscopic nephropexy using tension-free vaginal tape for symptomatic nephroptosis. Urology. 2004;64:372-374.
Matsui Y, Matsuta Y, Okubo K, et al. Laparoscopic nephropexy: treatment outcome and quality of life. Int J Urol. 2004;11:1-6.
Moss SW. Floating kidneys: a century of nephroptosis and nephropexy. J Urol. 1997;158:699-702.
Plas E, Daha K, Riedl CR, et al. Long-term followup after laparoscopic nephropexy for symptomatic nephroptosis. J Urol. 2001;166:449-452.
Canales B, Monga M. Surgical management of the calyceal diverticulum. Curr Opin Urol. 2003;13:255-260.
Donnellan SM, Harewood LM, Webb DR. Percutaneous management of caliceal diverticular calculi: technique and outcome. J Endourol. 1999;13:83-87.
Gluckman GR, Stoller M, Irby P. Laparoscopic pyelocaliceal diverticula ablation. J Endourol. 1993;7:315-317.
Harewood LM, Agarwal D, Lindsay S, et al. Extraperitoneal laparoscopic caliceal diverticulectomy. J Endourol. 1996;10:425-430.
Jones JA, Lingeman JE, Steidle CP. The roles of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy in management of pyelocaliceal diverticula. J Urol. 1991;146:724-727.
Miller SD, Ng CS, Streem SB, et al. Laparoscopic management of caliceal diverticular calculi. J Urol. 2002;167:1248-1252.
Pang K, David RD, Fuchs GJ. Treatment of stones in caliceal diverticula using retrograde endoscopic approach: critical assessment after 2 years [abstract]. J Endourol. 1992;6:80.
Ruckle HC, Segura JW. Laparoscopic treatment of a stone-filled, caliceal diverticulum: a definitive, minimally invasive therapeutic option. J Urol. 1994;151(1):122-124.
Stream SB, Yost A. Treatment of caliceal diverticular calculi with extracorporeal shock wave lithotripsy: patient selection and extended follow up. J Urol. 1992;148:1043-1046.
Wolf JSJr. Caliceal diverticulum and hydrocalyx. Laparoscopic management. Urol Clin North Am. 2000;27:655-660.
Wyler SF, Bachmann A, Jayet C, et al. Retroperitoneoscopic management of caliceal diverticular calculi. Urology. 2005;65:380-383.
Chiu AW, Chen MT, Chang LS. Laparoscopic nephrolysis for chyluria: case report of long-term success. J Endourol. 1995;9:319-322.
Cohen PG, Glenn JF, Franco NM, et al. Nontropical chyluria secondary to massive mesenteric adenitis. Case report with metabolic and immunologic studies. Am J Med. 1984;77:583-588.
Dalela D, Gupta VP, Goel A, et al. Omental wrap around the renal pedicle: an adjunctive step to minimize morbidity and recurrence after lymphorenal disconnection for chyluria. BJU Int. 2004;94:673-674.
Dalela D, Rastogi M, Goel A, et al. Silver nitrate sclerotherapy for ‘clinically significant’ chyluria: a prospective evaluation of duration of therapy. Urol Int. 2004;72:335-340.
Eisner BH, Tanrikut C, Dahl DM. Chyluria secondary to lymphorenal fistula. Kidney Int. 2009;76:126.
El-Reshaid KA, Madda JP, Sherif MF. Chyluria associated with renal vasculitis. Saudi J Kidney Dis Transpl. 1998;9:152-156.
Garrido P, Arcas R, Bobadilla JF, et al. Thoracic aneurysm as a cause of chyluria: resolution by surgical treatment. Ann Thorac Surg. 1995;60:687-689.
Goel S, Mandhani A, Srivastava A, et al. Is povidone iodine an alternative to silver nitrate for renal pelvic instillation sclerotherapy in chyluria? BJU Int. 2004;94:1082-1085.
Gomella LG, Shenot P, Abdel-Meguid TA. Extraperitoneal laparoscopic nephrolysis for the treatment of chyluria. Br J Urol. 1998;81:320-321.
Kim RJ, Joudi FN. Chyluria after partial nephrectomy: case report and review of the literature. Sci World J. 2009;9:1-4.
McNeil JC, Corbett ST, Kuruvilla S, et al. Metanephric adenoma in a five-year-old boy presenting with chyluria: case report and review of literature. Urology. 2008;72:545-547.
Onyeije CI, Sherer DM, Trambert J. Nonfilarial chyluria during pregnancy. Obstet Gynecol. 1997;90:699-700.
Tandon V, Singh H, Dwivedi US, et al. Filarial chyluria: long-term experience of a university hospital in India. Int J Urol. 2004;11:193-198.
Wilson RS, White RJ. Lymph node tuberculosis presenting as chyluria. Thorax. 1976;31:617-620.
Berger A, Brandina R, Atalla MA, et al. Laparoscopic radical nephrectomy for renal cell carcinoma: oncological outcomes at 10 years or more. J Urol. 2009;182:2172-2176.
Cadeddu JA, Ono Y, Clayman RV, et al. Laparoscopic nephrectomy for renal cell cancer: evaluation of efficacy and safety: a multicenter experience. Urology. 1998;52:773-777.
Desai MM, Strzempkowski B, Matin SF, et al. Prospective randomized comparison of transperitoneal versus retroperitoneal laparoscopic radical nephrectomy. J Urol. 2005;173:38-41.
Gabr AH, Gdor Y, Strope SA, et al. Approach and specimen handling do not influence oncological perioperative and long-term outcomes after laparoscopic radical nephrectomy. J Urol. 2009;182:874-880.
Gill IS, Schweizer D, Hobart MG, et al. Retroperitoneal laparoscopic radical nephrectomy: the Cleveland Clinic experience. J Urol. 2000;163:1665-1670.
Harano M, Eto M, Omoto K, et al. Long-term outcome of hand-assisted laparoscopic radical nephrectomy for localized stage T1/T2 renal-cell carcinoma. J Endourol. 2005;19:803-807.
Hattori R, Osamu K, Yoshino Y, et al. Laparoscopic radical nephrectomy for large renal-cell carcinomas. J Endourol. 2009;23:1523-1526.
Hemal AK, Kumar A, Kumar R, et al. Laparoscopic versus open radical nephrectomy for large renal tumors: a long-term prospective comparison. J Urol. 2007;177:862-866.
Kerbl K, Clayman RV, McDougall EM, et al. Transperitoneal nephrectomy for benign disease of the kidney: a comparison of laparoscopic and open surgical techniques. Urology. 1994;43:607-613.
Martin GL, Castle EP, Martin AD, et al. Outcomes of laparoscopic radical nephrectomy in the setting of vena caval and renal vein thrombus: seven-year experience. J Endourol. 2008;22:1681-1685.
McDougall EM, Clayman CR, Elashry OM. Laparoscopic radical nephrectomy for renal tumor: the Washington University Hospital experience. J Urol. 1996;155:1180-1185.
Molina WR, Desai MM, Ng CS, et al. Retroperitoneoscopic radical nephrectomy with concomitant distal pancreatectomy: case report. J Endourol. 2004;18:665-667.
Nambirajan T, Jeschke S, Al-Zahrani H, et al. Prospective, randomized controlled study: transperitoneal laparoscopic versus retroperitoneoscopic radical nephrectomy. Urology. 2004;64:919-924.
Nelson C, Wolf JS. Comparison of hand-assisted versus standard laparoscopic radical nephrectomy for suspected renal cell carcinoma. J Urol. 2002;167:1989-1994.
Okeke AA, Timoney AG, Keeley FX. Hand-assisted laparoscopic nephrectomy: complications related to the hand-port site. BJU Int. 2002;90:364-367.
Ono Y, Kinukawa T, Hattori R, et al. The long-term outcome of laparoscopic radical nephrectomy for small renal cell carcinoma. J Urol. 2001;165:1867-1870.
Patel VR, Leveillee RJ. Hand-assisted laparoscopic nephrectomy for stage T1 and large stage T2 renal tumors. J Endourol. 2003;17:379-383.
Permpongkosol S, Chan DY, Link RE, et al. Long-term survival analysis after laparoscopic radical nephrectomy. J Urol. 2005;174:1222-1225.
Portis AJ, Yan Y, Landman J, et al. Long-term followup after laparoscopic radical nephrectomy. J Urol. 2002;167:1257-1262.
Saika T, Ono Y, Hattori R, et al. Long-term outcome of laparoscopic radical nephrectomy for pathologic T1 renal cell carcinoma. Urology. 2003;62:1018-1023.
Stifelman MD, Handler T, Nieder AM, et al. Hand-assisted laparoscopy for large renal specimens: a multi-institutional study. Urology. 2003;61:78-82.
Walther MM, Lyne JC, Libutti SK, et al. Laparoscopic cytoreductive nephrectomy as preparation for administration of systemic interleukin-2 in the treatment of metastatic renal cell carcinoma: a pilot study. Urology. 1999;53:496-501.
Wille AH, Roigas J, Deger S, et al. Laparoscopic radical nephrectomy: techniques, results and oncological outcome in 125 consecutive cases. Eur Urol. 2004;45:483-488.
Wolf J. Tips and tricks for hand-assisted laparoscopy. AUA Update Series. 24, 2005. lesson 2
Radical Nephrectomy: Special Considerations
Bandi G, Wen CC, Moon TD, et al. Single center preliminary experience with hand-assisted laparoscopic resection of isolated recurrent renal cell carcinoma fossa recurrences. Urology. 2008;71:495-499.
Berger AD, Kanofsky JA, O’Malley RL, et al. Transperitoneal laparoscopic radical nephrectomy for large (more than 7 cm) renal masses. Urology. 2008;71:421-424.
Blom JH, van Poppel H, Maréchal JM, et al. Radical nephrectomy with and without lymph-node dissection: final results of European Organization for Research and Treatment of Cancer (EORTC) Randomized Phase 3 Trial 30881. Eur Urol. 2009;55:28-34.
Blute ML, Leibovich BC, Cheville JC, et al. A protocol for performing extended lymph node dissection using primary tumor pathological features for patients treated with radical nephrectomy for clear cell renal cell carcinoma. J Urol. 2004;172:465-469.
Bouvy ND, Marquet RL, Jekeel H, et al. Impact of gas(less) laparoscopy and laparotomy on peritoneal tumor growth and abdominal wall metastasis. Ann Surg. 1996;224:694-700.
Burns JM, Matthews BD, Pollinger HS, et al. Effect of carbon dioxide pneumoperitoneum and wound closure technique on port site implantation in a rat model. Surg Endosc. 2005;19:441-447.
Chapman TN, Sharma S, Zhang S, et al. Laparoscopic lymph node dissection in clinically node-negative patients undergoing laparoscopic nephrectomy for renal carcinoma. Urology. 2008;71:287-291.
Desai MM, Gill IS, Ramani AP, et al. Laparoscopic radical nephrectomy for cancer with level I renal vein involvement. J Urol. 2003;169:487-491.
Eisenberg MS, Meng MV, Master VA, et al. Laparoscopic versus open cytoreductive nephrectomy in advanced renal-cell carcinoma. J Endourol. 2006;20:504-508.
Eng MK, Katz MH, Bernstein AJ, et al. Laparoscopic port-site metastasis in urologic surgery. J Endourol. 2008;22:1581-1585.
Granberg CF, Krambeck AE, Leibovich BC, et al. Potential underdetection of pT(3a) renal-cell carcinoma with laparoscopic morcellation. J Endourol. 2007;21:1183-1186.
Gupta A, Watson DI, Ellis T, et al. Tumour implantation following laparoscopy using different insufflation gases. ANZ J Surg. 2002;72:254-257.
Guzzo TJ, Schaeffer EM, McNeil BK, et al. Laparoscopic radical nephrectomy for patients with pathologic T3b renal-cell carcinoma: the Johns Hopkins experience. J Endourol. 2009;23:63-67.
Halpin VJ, Underwood RA, Ye D, et al. Pneumoperitoneum does not influence trocar site implantation during tumor manipulation in a solid tumor model. Surg Endosc. 2005;19:1636-1640.
Hemal AK, Kumar A, Kumar R, et al. Laparoscopic versus open radical nephrectomy for large renal tumors: a long-term prospective comparison. J Urol. 2007;177:862-866.
Hernandez F, Rha KH, Pinto PA, et al. Laparoscopic nephrectomy: assessment of morcellation versus intact specimen extraction. J Urol. 2003;170:412-415.
Hsu TH, Jeffrey RBJr, Chon C, et al. Laparoscopic radical nephrectomy incorporating intraoperative ultrasonography for renal cell carcinoma with renal vein tumor thrombus. Urology. 2003;61:1246-1248.
Ikramuddin S, Lucus J, Ellison EC, et al. Detection of aerosolized cells during carbon dioxide laparoscopy. J Gastrointest Surg. 1998;2:580-583.
Itano NB, Blute ML, Spotts B, et al. Outcome of isolated renal cell carcinoma fossa recurrence after nephrectomy. J Urol. 2000;164:322-325.
Jingli C, Rong C, Rubai X. Influence of colorectal laparoscopic surgery on dissemination and seeding of tumor cells. Surg Endosc. 2006;20:1759-1761.
Landman J, Lento P, Hassen W, et al. Feasiblity of pathological evaluation of morcellated kidneys after radical nephrectomy. J Urol. 2000;164:2086-2089.
Margulis V, McDonald M, Tamboli P, et al. Predictors of oncological outcome after resection of locally recurrent renal cell carcinoma. J Urol. 2009;181:2044-2051.
Martin GL, Castle EP, Martin AD, et al. Outcomes of laparoscopic radical nephrectomy in the setting of vena caval and renal vein thrombus: seven-year experience. J Endourol. 2008;22:1681-1685.
Matin SF, Ahrar K, Cadeddu JA, et al. Residual and recurrent disease following renal energy ablative therapy: a multi-institutional study. J Urol. 2006;176:1973-1977. Erratum in: J Urol 2008;179:2490
Matin SF, Madsen LT, Wood CG. Laparoscopic cytoreductive nephrectomy: the M. D. Anderson Cancer Center experience. Urology. 2006;68:528-532.
Micali S, Celia A, Bove P, et al. Tumor seeding in urological laparoscopy: an international survey. J Urol. 2004;171:2151-2154.
Nguyen CT, Lane BR, Kaouk JH, et al. Surgical salvage of renal cell carcinoma recurrence after thermal ablative therapy. J Urol. 2008;180:104-109.
Pantuck AJ, Zisman A, Dorey F, et al. Renal cell carcinoma with retroperitoneal lymph nodes: role of lymph node dissection. J Urol. 2003;169:2076-2083.
Rabban JT, Meng MV, Yeh B, et al. Kidney morcellation in laparoscopic nephrectomy for tumor: recommendations for specimen sampling and pathologic tumor staging. Am J Surg Path. 2001;25:1158-1166.
Rabets JC, Kaouk J, Fergany A, et al. Laparoscopic versus open cytoreductive nephrectomy for metastatic renal cell carcinoma. Urology. 2004;64:930-934.
Rapp DE, Orvieto MA, Gerber GS, et al. En bloc stapling of renal hilum during laparoscopic nephrectomy and nephroureterectomy. Urology. 2004;64:655-659.
Rassweiler J, Tsivian A, Kumar AV, et al. Oncologic safety of laparoscopic surgery for urological malignancy: experience with more than 1,000 operations. J Urol. 2003;169:2072-2075.
Rosoff JS, Raman JD, Sosa RE, et al. Laparoscopic radical nephrectomy for renal masses 7 centimeters or larger. JSLS. 2009;13:148-153.
Steinberg AP, Finelli A, Desai MM, et al. Laparoscopic radical nephrectomy for large (greater than 7 cm, T2) renal tumors. J Urol. 2004;172:2172-2176.
Tsivian A, Shtabsky A, Issakov J, et al. The effect of pneumoperitoneum on dissemination and scar implantation on intra-abdominal tumor cells. J Urol. 2000;164:2096-2098.
Urban DA, Kerbl K, McDougall EM, et al. Organ entrapment and renal morcellation: permeability studies. J Urol. 1993;150:1792-1794.
Wakabayashi Y, Kataoka A, Koizumi S, et al. A simple technique for facilitating kidney entrapment using a laparoscopic sack during retroperitoneal laparoscopic radical nephrectomy. Urology. 2003;61:828-829.
White WM, Klein FA, Gash J, et al. Prospective radiographic followup after en bloc ligation of the renal hilum. J Urol. 2007;178:1888-1891.
Wu SD, Lesani OA, Zhao LC, et al. A multi-institutional study on the safety and efficacy of specimen morcellation after laparoscopic radical nephrectomy for clinical stage T1 or T2 renal cell carcinoma. J Endourol. 2009;23:1513-1518.
Aron M, Koenig P, Kaouk JH, et al. Robotic and laparoscopic partial nephrectomy: a matched-pair comparison from a high-volume centre. BJU Int. 2008;102:86-92.
Caruso RP, Phillips CK, Kau E, et al. Robot assisted laparoscopic partial nephrectomy: initial experience. J Urol. 2006;176:36-39.
Dash A, Vickers AJ, Schachter LR, et al. Comparison of outcomes in elective partial vs radical nephrectomy for clear cell renal cell carcinoma of 4-7 cm. BJU Int. 2006;97:939-945.
Desai MM, Gill IS, Kaouk JH, et al. Laparoscopic partial nephrectomy with suture repair of the pelvicaliceal system. Urology. 2003;61:99-104.
Finelli A, Gill IS, Desai MM, et al. Laparoscopic heminephrectomy for tumor. Urology. 2005;65:473-478.
Frank I, Colombo JRJr, Rubinstein M, et al. Laparoscopic partial nephrectomy for centrally located renal tumors. J Urol. 2006;175:849-852.
Gettman MT, Blute ML, Chow GK, et al. Robotic-assisted laparoscopic partial nephrectomy: technique and initial clinical experience with DaVinci robotic system. Urology. 2004;64:914-918.
Gill IS, Abreu SC, Desai MM, et al. Laparoscopic ice slush renal hypothermia for partial nephrectomy: the initial experience. J Urol. 2003;170:52-56.
Gill IS, Colombo JRJr, Frank I, et al. Laparoscopic partial nephrectomy for hilar tumors. J Urol. 2005;174:850-853.
Gill IS, Colombo JRJr, Moinzadeh A, et al. Laparoscopic partial nephrectomy in solitary kidney. J Urol. 2006;175:454-458.
Gill IS, Delworth MG, Munch LC. Laparoscopic retroperitoneal partial nephrectomy. J Urol. 1994;152:1539-1542.
Guillonneau B, Bermúdez H, Gholami S, et al. Laparoscopic partial nephrectomy for renal tumor: single center experience comparing clamping and no clamping techniques of the renal vasculature. J Urol. 2003;169:483-486.
Herrell SD, Levin BM. Laparoscopic partial nephrectomy: use of the TissueLink hemostatic dissection device. J Endourol. 2005;19:446-449.
Hindley RG, Barber NJ, Walsh K, et al. Laparoscopic partial nephrectomy using the potassium titanyl phosphate laser in a porcine model. Urology. 2006;67:1079-1083.
Huang WC, Levey AS, Serio AM, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7:735-740.
Janetschek G, Abdelmaksoud A, Bagheri F, et al. Laparoscopic partial nephrectomy in cold ischemia: renal artery perfusion. J Urol. 2004;171:68-71.
Jayson M, Sanders H. Increased incidence of serendipitously discovered renal cell carcinoma. Urology. 1998;51:203-205.
Kaul S, Laungani R, Sarle R, et al. Da Vinci-assisted robotic partial nephrectomy: technique and results at a mean of 15 months of follow-up. Eur Urol. 2007;51:186-191.
Landman J, Venkatesh R, Lee D, et al. Renal hypothermia achieved by retrograde endoscopic cold saline perfusion: technique and initial clinical application. Urology. 2003;61:1023-1025.
Leibovich BC, Blute ML, Cheville JC, et al. Nephron-sparing surgery for appropriately selected renal cell carcinoma between 4 cm and 7 cm results in outcome similar to radical nephrectomy. J Urol. 2004;171:1066-1070.
Leslie JA, Prihoda T, Thompson IM. Serendipitous renal cell carcinoma in the post-CT era: continued evidence in improved outcomes. Urol Oncol. 2003;21:39-44.
Liu M, Rajbabu K, Zhu G, et al. Laparoscopic partial nephrectomy with saline-irrigated KTP laser in a porcine model. J Endourol. 2006;20:1096-1100.
Lotan Y, Gettman MT, Lindberg G, et al. Laparoscopic partial nephrectomy using holmium laser in a porcine model. JSLS. 2004;8:51-55.
Mitchell RE, Gilbert SM, Murphy AM, et al. Partial nephrectomy and radical nephrectomy offer similar cancer outcomes in renal cortical tumors 4 cm or larger. Urology. 2006;67:260-264.
Moinzadeh A, Hasan W, Spaliviero M, et al. Water jet assisted laparoscopic partial nephrectomy without hilar clamping in the calf model. J Urol. 2005;174:317-321.
Ng CS, Gill IS, Ramani AP, et al. Transperitoneal versus retroperitoneal laparoscopic partial nephrectomy: patient selection and perioperative outcomes. J Urol. 2005;174:846-849.
Nguyen MM, Gill IS. Halving ischemia time during laparoscopic partial nephrectomy. J Urol. 2008;179:627-632.
Ramani AP, Abreu SC, Desai MM, et al. Laparoscopic upper pole partial nephrectomy with concomitant en bloc adrenalectomy. Urology. 2003;62:223-226.
Rogers CG, Singh A, Blatt AM, et al. Robotic partial nephrectomy for complex renal tumors: surgical technique. Eur Urol. 2008;53:514-521.
Romero FR, Rais-Bahrami S, Muntener M, et al. Laparoscopic partial nephrectomy in obese and non-obese patients: comparison with open surgery. Urology. 2008;71:806-809.
Simmons MN, Chung BI, Gill IS. Perioperative efficacy of laparoscopic partial nephrectomy for tumors larger than 4 cm. Eur Urol. 2009;55:199-208.
Spaliviero M, Herts BR, Magi-Galluzzi C, et al. Laparoscopic partial nephrectomy for cystic masses. J Urol. 2005;174:614-619.
Steinberg AP, Abreu SC, Desai MM, et al. Laparoscopic nephron-sparing surgery in the presence of renal artery disease. Urology. 2003;62:935-939.
Steinberg AP, Kilciler M, Abreu SC, et al. Laparoscopic nephron-sparing surgery for two or more ipsilateral renal tumors. Urology. 2004;64:255-258.
Tsivian A, Shtricker A, Benjamin S, et al. Laparoscopic partial nephrectomy for tumour excision in a horseshoe kidney. Eur Urol. 2007;51:1132-1133.
Turna B, Aron M, Frota R, et al. Feasibility of laparoscopic partial nephrectomy after previous ipsilateral renal procedures. Urology. 2008;72:584-588.
Winfield HN, Donovan JF, Godet AS, et al. Laparoscopic partial nephrectomy: initial case report for benign disease. J Endourol. 1993;7:521-526.
Wright JL, Porter JR. Laparoscopic partial nephrectomy: comparison of transperitoneal and retroperitoneal approaches. J Urol. 2005;174:841-845.
Centurion SA, Centurion LM, Souza ME, et al. Effects of ischemic liver preconditioning on hepatic ischemia/reperfusion injury in the rat. Transplant Proc. 2007;39:361-364.
Cisek LJ, Gobet RM, Peters CA. Pneumoperitoneum produces reversible renal dysfunction in animals with normal and chronically reduced renal function. J Endourol. 1998;12:95-100.
Frank RS, Frank TS, Zelenock GB, et al. Ischemia with intermittent reperfusion reduces functional and morphologic damage following renal ischemia in the rat. Ann Vasc Surg. 1993;7:150-155.
Fried TA, Hishida A, Barnes JL, et al. Ischemic acute renal failure in the rat: protective effect of uninephrectomy. Am J Physiol. 1984;247:F568-F574.
Funahashi Y, Hattori R, Yamamoto T, et al. Change in contralateral renal parenchymal volume 1 week after unilateral nephrectomy. Urology. 2009;74:708-712.
Gong EM, Zorn KC, Orvieto MA, et al. Artery-only occlusion may provide superior renal preservation during laparoscopic partial nephrectomy. Urology. 2008;72:843-846.
Orvieto MA, Tolhurst SR, Chuang MS, et al. Defining maximal renal tolerance to warm ischemia in porcine laparoscopic and open surgery model. Urology. 2005;66:1111-1115.
Orvieto MA, Zorn KC, Mendiola F, et al. Recovery of renal function after complete renal hilar versus artery alone clamping during open and laparoscopic surgery. J Urol. 2007;177:2371-2374.
Saidi RF, Chang J, Brooks S, et al. Ischemic preconditioning and intermittent clamping increase the tolerance of fatty liver to hepatic ischemia-reperfusion injury in the rat. Transplant Proc. 2007;39:3010-3014.
Shuto K, Kitano S, Yoshida T, et al. Hemodynamic and arterial blood gas changes during carbon dioxide and helium pneumoperitoneum in pigs. Surg Endosc. 1995;9:1173-1178.
Simon J, Bartsch GJr, Finter F, et al. Laparoscopic partial nephrectomy with selective control of the renal parenchyma: initial experience with a novel laparoscopic clamp. BJU Int. 2009;103:805-808.
Stackl W, Hofmann E, Marberger M. Is the solitary kidney a privileged kidney? Br J Urol. 1983;55:460-464.
Verhoest G, Manunta A, Bensalah K, et al. Laparoscopic partial nephrectomy with clamping of the renal parenchyma: initial experience. Eur Urol. 2007;52:1340-1346.
Watanabe R, Saitoh K, Kurumada S, et al. Gasless laparoscopy-assisted live donor nephrectomy. Transplant Proc. 2002;34:2578-2580.
Wilson DH, Barton BB, Parry WL, et al. Effects of intermittent versus continuous renal arterial occlusion on hemodynamics and function of the kidney. Invest Urol. 1971;8:507-515.
Wu HH, Hsiao TY, Chien CT, et al. Ischemic conditioning by short periods of reperfusion attenuates renal ischemia/reperfusion induced apoptosis and autophagy in the rat. J Biomed Sci. 2009;16:19.
Partial Nephrectomy: Contemporary Outcomes
Gill IS, Kamoi K, Aron M, et al. 800 aparoscopic partial nephrectomies: a single-surgeon series. J Urol. 2010;183:34-41.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178:41-46.
Herati AS, Rais-Bahrami S, Andonian S, et al. Off-clamp versus on-clamp laparoscopic partial nephrectomy: functional and short-term oncological outcomes (submitted).
Lane BR, Gill IS. Five-year oncological outcomes after laparoscopic partial nephrectomy. J Urol. 2007;177:70-74.
Lane BR, Gill IS. Seven-year oncological outcomes after laparoscopic and open partial nephrectomy. J Urol. 2010;183:473-479.
Nguyen MM, Gill IS. Halving ischemia time during laparoscopic partial nephrectomy. J Urol. 2008;179:627-632.
Permpongkosol S, Bagga HS, Romero FR, et al. Trends in the operative management of renal tumors over a 14-year period. BJU Int. 2006;98:751-755.
Permpongkosol S, Colombo JRJr, Gill IS, et al. Positive surgical parenchymal margin after laparoscopic partial nephrectomy for renal cell carcinoma: oncological outcomes. J Urol. 2006;176:2401-2404.
Turna B, Aron M, Gill IS. Expanding indications for laparoscopic partial nephrectomy. Urology. 2008;72:481-487.
Yossepowitch O, Thompson RH, Leibovich BC, et al. Positive surgical margins at partial nephrectomy: predictors and oncological outcomes. J Urol. 2008;179:2158-2163.
Laparoscopic Ablative Techniques
Allaf ME, Bhayani SB, Rogers C, et al. Laparoscopic partial nephrectomy: evaluation of long-term oncological outcome. J Urol. 2004;172:871-873.
Aron M, Gill IS. Minimally invasive nephron-sparing surgery (MINSS) for renal tumours. Part II: probe ablative therapy. Eur Urol. 2007;51:348-357.
Beemster P, Phoa S, Wijkstra H, et al. Follow-up of renal masses after cryosurgery using computed tomography; enhancement patterns and cryolesion size. BJU Int. 2008;101:1237-1242.
Bolte SL, Ankem MK, Moon TD, et al. Magnetic resonance imaging findings after laparoscopic renal cryoablation. Urology. 2006;67:485-489.
Campbell SC, Krishnamurthi V, Chow G, et al. Renal cryosurgery: experimental evaluation of treatment parameters. Urology. 1998;52:29-33.
Cestari A, Guazzoni G, dell’Acqua V, et al. Laparoscopic cryoablation of solid renal masses: intermediate term followup. J Urol. 2004;172:1267-1270.
Chosy SG, Nakada SY, Lee FTJr, et al. Monitoring renal cryosurgery: predictors of tissue necrosis in swine. J Urol. 1998;159:1370-1374.
Crowley JD, Shelton J, Iverson AJ, et al. Laparoscopic and computed tomography-guided percutaneous radiofrequency ablation of renal tissue: acute and chronic effects in an animal model. Urology. 2001;57:976-980.
Finley DS, Beck S, Box G, et al. Percutaneous and laparoscopic cryoablation of small renal masses. J Urol. 2008;180(2):492-498.
Frank I, Blute ML, Leibovich BC, et al. Independent validation of the 2002 American Joint Committee on cancer primary tumor classification for renal cell carcinoma using a large, single institution cohort. J Urol. 2005;173:1889-1892.
Gill IS, Kamoi K, Aron M, et al. 800 laparoscopic partial nephrectomies: a single surgeon series. J Urol. 2010;183:34-41.
Gill IS, Novick AC, Meraney AM, et al. Laparoscopic renal cryoablation in 32 patients. Urology. 2000;56:748-753.
Gill IS, Novick AC, Soble JJ, et al. Laparoscopic renal cryoablation: initial clinical series. Urology. 1998;52:543-551.
Goldberg SN, Gazelle GS, Mueller PR. Thermal ablation therapy for focal malignancy: a unified approach to underlying principles, techniques, and diagnostic imaging guidance. AJR Am J Roentgenol. 2000;174:323-331.
Johnson DB, Solomon SB, Su LM, et al. Defining the complications of cryoablation and radio frequency ablation of small renal tumors: a multi-institutional review. J Urol. 2004;172:874-877.
Kunkle DA, Egleston BL, Uzzo RG. Excise, ablate or observe: the small renal mass dilemma—a meta-analysis and review. J Urol. 2008;179:1227-1233.
Laguna MP, Beemster P, Kumar P, et al. Perioperative morbidity of laparoscopic cryoablation of small renal masses with ultrathin probes: a European multicentre experience. Eur Urol. 2009;56:355-361.
Lane BR, Gill IS. Five-year oncological outcomes after laparoscopic partial nephrectomy. J Urol. 2007;177:70-74.
Lawatsch EJ, Langenstroer P, Byrd GF, et al. Intermediate results of laparoscopic cryoablation in 59 patients at the Medical College of Wisconsin. J Urol. 2006;175:1225-1229.
Lewin JS, Connell CF, Duerk JL, et al. Interactive MRI-guided radiofrequency interstitial thermal ablation of abdominal tumors: clinical trial for evaluation of safety and feasibility. J Magn Reson Imaging. 1998;8:40-47.
Lewin JS, Nour SG, Connell CF, et al. Phase II clinical trial of interactive MR imaging-guided interstitial radiofrequency thermal ablation of primary kidney tumors: initial experience. Radiology. 2004;232:835-845.
Malcolm JB, Berry TT, Williams MB, et al. Single center experience with percutaneous and laparoscopic cryoablation of small renal masses. J Endourol. 2009;23:907-911.
Marszalek M, Meixl H, Polajnar M, et al. Laparoscopic and open partial nephrectomy: a matched-pair comparison of 200 patients. Eur Urol. 2009;55:1171-1178.
Matin SF, Ahrar K, Cadeddu JA, et al. Residual and recurrent disease following renal energy ablative therapy: a multi-institutional study. J Urol. 2006;176:1973-1977. Erratum in: J Urol 2008;179(6):2490
Moinzadeh A, Gill IS, Finelli A, et al. Laparoscopic partial nephrectomy: 3-year followup. J Urol. 2006;175:459-462.
Nakada SY, Lee FT, Warner T, et al. Laparoscopic cryosurgery of the kidney in the porcine model: An acute histological study. Urology. 1998;51(Suppl. 5A):161-166.
Orihuela E, van Sonnenberg E, Motamedi M, et al. Thermodynamics of RCC during cryotherapy: effect of warm renal arterial blood flow [abstract]. J Urol. 1999;161(Suppl):144.
Pyo P, Chen A, Grasso M. Retroperitoneal laparoscopic partial nephrectomy: surgical experience and outcomes. J Urol. 2008;180:1279-1283.
Remer EM, Weinberg EJ, Oto A, et al. MR imaging of the kidneys after laparoscopic cryoablation. AJR Am J Roentgenol. 2000;174:635-640.
Weld KJ, Figenshau RS, Venkatesh R, et al. Laparoscopic cryoablation for small renal masses: three-year follow-up. Urology. 2007;69:448-451.
Wingo MS, Leveillee RJ. Central and deep renal tumors can be effectively ablated: radiofrequency ablation outcomes with fiberoptic peripheral temperature monitoring. J Endourol. 2008;22:1261-1267.
Wright AD, Turk TM, Nagar MS, et al. Endophytic lesions: a predictor of failure in laparoscopic renal cryoablation. J Endourol. 2007;21:1493-1496.
Laparoendoscopic Single-Site (LESS) Surgery
Andonian S, Atalla M, Herati H, et al. Laparoendoscopic single site (LESS) Pfannenstiel donor nephrectomy. Presented at the World Congress of Endourology, Annual Meeting, Munich, Germany, 2009.
Aron M, Canes D, Desai MM, et al. Transumbilical single-port laparoscopic partial nephrectomy. BJU Int. 2009;103(4):516-521.
Canes D, Berger A, Aron M, et al. Laparo-endoscopic single site (LESS) versus standard laparoscopic left donor nephrectomy: matched-pair comparison. Eur Urol. 2010;57:95-101.
Desai MM, Berger A, Brandina A, et al. Laparosndoscopic single-site (LESS) surgery: initial 100 patients. Urology. 2009;74:805-812.
Kaouk JH, Goel RK, Haber GP, et al. Robotic single-port transumbilical surgery in humans: initial report. BJU Int. 2009;103:366-369.
Raman JD, Bagrodia A, Cadeddu JA. Single-incision, umbilical laparoscopic versus conventional laparoscopic nephrectomy: a comparison of perioperative outcomes and short-term measures of convalescence. Eur Urol. 2009;55:1198-1204.
Raman JD, Bensalah K, Bagrodia A, et al. Laboratory and clinical development of single keyhole umbilical nephrectomy. Urology. 2007;70:1039-1042.
Stein RJ, White WM, Goel RK, et al. Robotic laparoendoscopic single-site surgery using Gelport as the access platform. Eur Urol. 2010;57:132-136.
White WM, Haber GP, Goel RK, et al. Single-port urological surgery: single-center experience with the first 100 cases. Urology. 2009;74:801-804.
Complications of Laparoscopic Renal Surgery
Aron M, Colombo JRJr, Turna B, et al. Diaphragmatic repair and/or reconstruction during upper abdominal urological laparoscopy. J Urol. 2007;178:2444-2450.
Baldwin DD, Dunbar JA, Parekh DJ, et al. Single-center comparison of purely laparoscopic, hand-assisted laparoscopic, and open radical nephrectomy in patients at high anesthetic risk. J Endourol. 2003;17:161-167.
Benway BM, Bhayani SB, Rogers CG, et al. Robot-assisted partial nephrectomy versus laparoscopic partial nephrectomy for renal tumors: a multi-institutional analysis of perioperative outcomes. J Urol. 2009;182:866-872.
Bishoff JT, Allaf ME, Kirkels W, et al. Laparoscopic bowel injury: incidence and clinical presentation. J Urol. 1999;161:887-890.
Borten M. Laparoscopic complications: prevention and management. Toronto: BC Decker; 1986. p. 317–29
Cadeddu JA, Regan F, Kavoussi LR, et al. The role of computerized tomography in the evaluation of complications after laparoscopic urological surgery. J Urol. 1997;158:1349-1352.
Canby-Hagino ED, Morey AF, Jatoi I, et al. Fibrin sealant treatment of splenic injury during open and laparoscopic left radical nephrectomy. J Urol. 2000;164:2004-2005.
Capelouto CC, Kavoussi LR. Complications of laparoscopic surgery. Urology. 1993;42:2-12.
Chan D, Bishoff JT, Ratner L, et al. Endovascular gastrointestinal stapler device malfunction during laparoscopic nephrectomy: early recognition and management. J Urology. 2000;164:319-321.
Chan DY, Cadeddu JA, Jarrett TW, et al. Laparoscopic radical nephrectomy: cancer control for renal cell carcinoma. J Urol. 2001;166:2095-2099.
Del Pizzo JJ, Jacobs SC, Bishoff JT, et al. Pleural injury during laparoscopic renal surgery: early recognition and management. J Urol. 2003;169:41-44.
Fahlenkamp D, Rassweiler J, Fornara P, et al. Complications of laparoscopic procedures in urology: experience with 2,407 procedures at 4 German centers. J Urol. 1999;162:765-770.
Gill IS, Kavoussi LR, Clayman RV, et al. Complications of laparoscopic nephrectomy in 185 patients: a multi-institutional review. J Urol. 1995;154:479-483.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178:41-46.
Hasson HM. A modified instrument and method for laparoscopy. Am J Obstet Gynecol. 1971;110:886-887.
Keeley FX, Tolley DA. A review of our first 100 cases of laparoscopic nephrectomy: defining risk factors for complications. Br J Urol. 1998;82:615-618.
Kim FJ, Pinto P, Su LM, et al. Ipsilateral orchialgia after laparoscopic donor nephrectomy. J Endourol. 2003;17:405-409.
McAllister M, Bhayani SB, Ong A, et al. Vena caval transection during retroperitoneoscopic nephrectomy: report of the complication and review of the literature. J Urol. 2004;172:183-185.
McGinnis DE, Strup SE, Gomella LG. Management of hemorrhage during laparoscopy. J Endourol. 2000;14:915-920.
Meeks JJ, Zhao LC, Navai N, et al. Risk factors and management of urine leaks after partial nephrectomy. J Urol. 2008;180:2375-2378.
Oefelein M, Bayazit Y. Chronic pain syndrome after laparoscopic radical nephrectomy. J Urol. 2003;170:1939-1940.
Permpongkosol S, Link RE, Su LM, et al. Complications of 2,775 urological laparoscopic procedures: 1993 to 2005. J Urol. 2007;177:580-585.
Rassweiler J, Fornara P, Weber M, et al. Laparoscopic nephrectomy: the experience of the laparoscopy working group of the German Urologic Association. J Urol. 1998;160:18-21.
Regan JP, Cho ES, Flowers JL. Small bowel obstruction after laparoscopic donor nephrectomy. Surg Endosc. 2003;17:108-110.
Schwartz MJ, Faiena I, Cinman N, et al. Laparoscopic bowel injury in retroperitoneal surgery: current incidence and outcomes. J Urol. 2010;184:589-594.
Shapiro EY, Hakimi AA, Hyams ES, et al. Renal artery pseudoaneurysm following laparoscopic partial nephrectomy. Urology. 2009;74:819-823.
Shuford M, McDougall E, Chang S, et al. Complications of contemporary radical nephrectomy: comparison of open vs. laparoscopic approach. Urol Oncol. 2004;22:121-126.
Thomas AA, Aron M, Hernandez AV, et al. Laparoscopic partial nephrectomy in octogenarians. Urology. 2009;74:1042-1046.
Varkarakis IM, Allaf ME, Bhayani SB, et al. Pancreatic injuries during laparoscopic urologic surgery. Urology. 2004;64:1089-1093.
Varkarakis I, Neururer R, Harabayashi T, et al. Laparoscopic radical nephrectomy in the elderly. BJU Int. 2004;94:517-520.
Wolf JS, Marchovich R, Gill IS, et al. Survey of neuromuscular injuries to the patient and surgeon during urologic laparoscopic surgery. Urology. 2000;55:831-836.
Penetrance of laparoscopic Renal Surgery for the Community Urologist
Miller DC, Saigal CS, Banerjee M, et al. Diffusion of surgical innovation among patients with kidney cancer. Cancer. 2008;112:1708-1717.
Richstone L, Kavoussi LR. Barriers to the diffusion of advanced surgical techniques. Cancer. 2008;112:1646-1649.