chapter 56 Ablative Therapy for Renal Tumors
The management of renal cell carcinoma has undergone a paradigm shift over the last two decades. The widespread use and refinement of modern imaging during this period has led to a dramatic increase in the number of incidentally discovered and low-stage renal lesions (Jayson and Sanders, 1998; Volpe et al, 2004). This fortunate trend has allowed urologists to explore alternative and less invasive forms of therapy, among them nephron-sparing surgery and renal ablative technologies (Colombo and Kaouk, 2007; Pinto, 2009). Cancer-specific survival, preservation of renal function, avoidance of treatment-related morbidity, and patient-centered quality-of-life outcomes now constitute the ultimate goals for management of early-stage renal cell carcinoma.
The preponderance of evidence-based data champions the role of partial nephrectomy in the treatment of localized unilateral small renal masses (Herr, 1999; Fergany et al, 2000). Nephron-sparing surgery, in this setting (tumors < 4 cm), offers comparable oncologic outcomes (>95% cancer-specific survival) as compared with radical nephrectomy and is associated with improved preservation of renal function, superior cardiac outcomes, and improved overall survival (Thompson et al, 2008; Huang et al, 2009; Zini et al, 2009). Moreover, nephron-sparing surgery avoids overtreatment of indolent or benign tumors, a particularly germane concern, because nearly 20% of malignant-appearing, small renal masses are pathologically benign (Frank et al, 2003). For these reasons, open partial nephrectomy is currently considered the gold standard treatment for radiographically enhancing small renal masses (Novick, 2004).
Despite its aforementioned long-term advantages, open partial nephrectomy is nevertheless associated with significant short-term morbidity, namely substantial postoperative pain, prolonged hospitalization, and a protracted convalescence (Gill et al, 2007). Because a large proportion of patients who are candidates for partial nephrectomy demonstrate concomitant co-morbidities or are of advanced age, this short-term morbidity must be taken into consideration during patient counseling, and less invasive alternatives, if appropriate, should be discussed.
Laparoscopic partial nephrectomy (LPN) replicates the principles of open nephron-sparing surgery but with less attendant surgical morbidity (Gill et al, 2002). Indeed, laparoscopic partial nephrectomy offers markedly improved convalescence and equivalent disease-free survival as compared with open partial nephrectomy (Aron and Gill, 2007; Andonian et al, 2008; Marszalek et al, 2009). However, laparoscopic partial nephrectomy remains a technically challenging procedure with significantly higher complication rates, even among experienced surgeons at centers of excellence (Gill et al, 2007). Further, renal hilar occlusion is nearly universally required during LPN to minimize blood loss and facilitate tumor resection in a bloodless field. In expert hands, warm ischemia times during LPN approach 30 minutes (Kural et al, 2009; Thomas et al, 2009). Although controversial and dependent on the inherent renal function status, warm ischemia times that approach or exceed 30 minutes may place the kidney at considerable risk for postischemic injury (Thompson et al, 2007; Lane et al, 2008).
Irrespective of the surgical approach chosen, nephron-sparing surgery is underutilized in the United States owing to the comparative risks and attendant technical demands associated with the procedure (Abouassaly et al, 2009). Recent Surveillance, Epidemiology, and End Results (SEER) data found that partial nephrectomy is performed in only 11.1% of patients, with the remaining 88.9% undergoing radical nephrectomy (Miller et al, 2008). Furthermore, when adjusted for patient demographics and tumor size, surgeon preference was significantly more important than patient characteristics with regard to the type of operation selected.
Energy-based, in-situ tumor ablation was developed in an attempt to broaden the minimally invasive treatment options available to patients with ostensibly localized, low-stage renal tumors (Goel and Kaouk, 2008a). Focal ablative therapy confers myriad advantages compared with extirpative surgery. Principally, renal tumor ablation is associated with fewer complications, less morbidity, and a shortened convalescence (Desai et al, 2005). Second, prospective studies have demonstrated no significant impact on renal function following treatment (Shingleton and Sewell, 2003; Raman et al, 2008). Although a modicum of parenchymal loss does occur, renal hilar occlusion is not performed during tumor ablation, and postischemic injury is therefore avoided. Next, ablation offers considerable treatment flexibility. That is, tumors may be treated open, laparoscopically, or percutaneously with the approach tailored to the specifics of the patient and tumor (Rukstalis et al, 2001; Deane and Clayman, 2006). Finally, renal tumor ablation is technically less demanding to perform than either open or laparoscopic partial nephrectomy. Complex reconstruction is not required following treatment, and dissection and isolation of the renal hilum is not requisite. Given these advantages, ideal candidates for ablative treatment have traditionally included patients with small multifocal tumors, those with one or more tumors in a solitary kidney in which surgical warm ischemia would place the kidney at risk, and those who desired treatment but were not appropriate candidates for extirpative surgery (Lehman and Landman, 2008).
Although well tolerated and technically less challenging than alternative extirpative options, concern exists regarding the oncologic efficacy of ablative techniques. A recent meta-analysis demonstrated a higher relative risk of local recurrence with cryoablation (RR = 7.45) and radiofrequency ablation (RR = 18.23) as compared with partial nephrectomy, but with no significant difference in progression to metastatic disease between the three groups (Kunkle et al, 2008). However, nonuniform criteria were used to define tumor recurrence in these series and pathology was not obtained for all specimens.
Evolving technology that includes treatment guidance systems, more robust operator experience, and improved patient selection may position renal ablative technologies as a treatment of choice for small renal masses. Probe ablative treatments are considerably less invasive than open partial nephrectomy, are less technically challenging and more easily learned than laparoscopic partial nephrectomy, and are associated with fewer complications as compared with open or laparoscopic partial nephrectomy.
Key Points: Management of the Small Renal Mass
Cryoablation
Mode of Action and Experimental Data
The concept of applying cold temperatures to destroy tissue is credited to an English physician, Dr. James Arnott (1797 to 1883). Using a combination of ice and salt, he was able to topically palliate tumors of the cervix and breast with variable success. Over the course of the ensuing decades, investigators developed more sophisticated, reproducible, and pragmatic methods of handling and applying topical cryogens (Gage, 1998). In 1963, a neurosurgeon from New York, Dr. Irving S. Cooper, introduced a probe capable of achieving a controlled temperature of −196° C in deep-seated tissues (Cooper, 1963). Using pressurized liquid nitrogen, this self-contained probe was employed to ablate otherwise inoperable brain tumors, as well as portions of the thalamus in patients with Parkinson disease. Cooper’s innovative concept ushered in a new era in cryosurgery in which larger volumes of tissue in less accessible locations could be successfully and reproducibly ablated.
Although Cooper’s experience provided the proof in principle for cryoablation, its practical application for abdominopelvic tumors was not immediately realized. Early cryogen delivery systems could not unfailingly control the extent of tissue death owing to a generalized lack of cryoprobe sophistication and an unreliable system of monitoring the freezing process (Weber and Lee, 2005). As such, irreparable collateral damage was often a consequence of treatment, and the technique was transiently abandoned. It was not until the development of cryoablation under real-time image guidance with intraoperative ultrasonography that the procedure became a safe and pragmatic treatment choice.
The application of intraoperative ultrasonography during cryosurgery addressed many of its aforementioned short-comings (Onik et al, 1984, 1985). First, ultrasonography offered an excellent method for evaluating the patient’s anatomy. The relative size and location of the area to be treated could be visualized and documented, and the entirety of the organ could be evaluated for occult metastatic disease. Second, precise probe placement could be assured by following its echogenic tip under ultrasound guidance. Lastly and most importantly, the freezing process could finally be monitored in real time by observing the formation and propagation of the highly echogenic leading edge of the iceball (Fig. 56-1). Because animal studies had already confirmed a close correlation between the sonographically visible iceball and the zone of cell death, surgeons now had a reliable and reproducible method of targeting and destroying tumors without attendant collateral damage (Steed et al, 1997; Campbell et al, 1998; Weber et al, 1998).
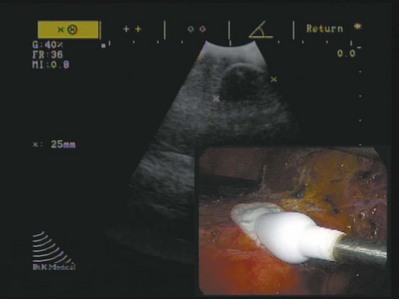
Figure 56–1 Intraoperative ultrasound image during renal tumor laparoscopic cryoablation. Note the hyperechoic semicircular edge with a posterior shadow of the developing iceball. Inset: laparoscopic view of the cryoablated mass with the cryoprobe positioned in the center of the tumor that is covered with ice.
Following treatment under image guidance, the next significant breakthrough with cryoablation was the introduction of argon gas–driven treatment systems (Rewcastle et al, 1999). Through the mid- to late 1990s, cryoablation was performed exclusively with liquid nitrogen-based systems that were relatively inefficient. Newer argon gas systems were developed, ones that use the Joule-Thomson principle (low temperatures are achieved by the rapid expansion of high-pressure, inert gas) to generate temperatures of −185.7° C within the treatment tissues. These systems require shorter treatment times and allow for smaller, more proficient cryoprobes that can be employed laparoscopically with fused ultrasound guidance or percutaneously under computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound guidance. The majority of commercially available cryoablation units (CryoHit, Galil Medical, Plymouth Meeting, PA; CryoCare, CryoCare CS, Endocare, Irvine, CA; SeedNet, Oncura, Philadelphia, PA) now employ argon gas–based systems as described.
The mechanism of tissue injury with cryoablation is multifaceted, reflecting direct cellular damage during the freezing phase and indirect reperfusion injury during the thawing phase (Mazur, 1977; Ishiguro and Rubinsky, 1994; Hoffman and Bischof, 2002). During rapid freezing or freezing at extremely low temperatures, ice crystals form within the intracellular spaces, causing cell death by direct injury to either the cellular membrane or structures within the cell. With gradual freezing, extracellular ice formation creates an osmotic gradient that results in intracellular fluid moving into the extracellular space. The result of this fluid shift is dehydration, cell membrane rupture, and subsequent cell death. During the thawing phase, reperfusion injury occurs that results in microcirculatory failure and small-vessel thrombosis (Weber et al, 1997; Kahlenberg et al, 1998). Although speculative, this may represent the dominant mechanism of cell kill during cryosurgery. The summative pathologic consequence of treatment is coagulative necrosis and fibrous scar formation.
The adequacy and reproducibility of tissue destruction during cryoablation is dependent upon several parameters:
Radiofrequency Ablation
Mode of Action and Experimental Data
Cushing and Bovie (1928) introduced the Bovie knife, an instrument that employed pulsed or continuous monopolar radiofrequency current to cauterize or cut tissue, respectively. Although this forerunner to modern electrocautery revolutionized and defined contemporary surgical techniques, it also provided the foundation for present-day radiofrequency ablation (RFA).
Radiofrequency energy effects cell kill by transferring alternating radiofrequency current (450 to 1200 kHz) through insulated interstitial needle electrodes (Raj et al, 2003). This energy transfer generates ionic friction and agitation within tissue that results in heating (Organ, 1976-1977; LeVeen, 1997; Rehman et al, 2004). When tissue is heated in excess of 60° C, irreversible coagulative necrosis and tissue desiccation occurs (Hsu et al, 2000). Much like cryoablation, the adequacy and reproducibility of tissue destruction during RFA is dependent upon several parameters:
Surgical Technique
Transperitoneal/Retroperitoneal Laparoscopic Renal Cryoablation/Radiofrequency Ablation
See Chapter 55 for these modalities.
Percutaneous Renal Cryoablation/Radiofrequency Ablation under Ultrasonography, CT, or MRI
Percutaneous tumor ablation can be performed under conscious sedation with local anesthesia or under general endotracheal anesthesia. Controversy exists regarding the optimal method of sedation. General endotracheal anesthesia allows controlled respiration during probe placement that may translate into more accurate targeting and improved overall outcomes. Conversely, conscious sedation minimizes the morbidity of the procedure and allows the procedure to be performed on an outpatient basis. Following induction, a 16-Fr urinary catheter is placed and intravenous antibiotics are administered. The patient is positioned on the CT or MRI gantry in the prone or flank position.
Intravenous contrast (iodinated or gadolinium contrast, depending on the imaging technique chosen and the patient’s renal function) is administered and the lesion identified and characterized. A 20-gauge “finder needle” or access sheath is placed near the expected location of the tumor, and imaging is repeated to confirm localization. It is important to note the depth of the finder needle for subsequent biopsy and probe placement. Using this finder needle as a guide, an 18-gauge Tru-Cut (Cardinal Health, Dublin, OH) core biopsy needle is inserted percutaneously, and positioning is again confirmed with repeat imaging. Specimens are obtained and sent for permanent section. The cryoprobe(s) or RFA probe is/are next inserted in a manner similar to that of the core biopsy needle. Cryoprobe positioning and positioning of the RFA probe, and prongs are again confirmed with repeat imaging. Treatment is carried out in a fashion similar to that described for laparoscopic cryoablation/radiofrequency ablation (Fig. 56-2). Again, a “track burn” can be performed with percutaneous RFA. With percutaneous cryoablation, the cryoprobe is removed and repeat imaging performed to evaluate for potential bleeding. With either technique, a postablation contrasted imaging study is performed to determine the adequacy of treatment.
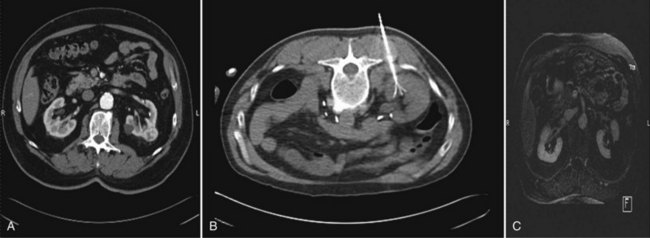
Figure 56–2 A, Preoperative CT scan noting a 2.4-cm solid enhancing mass in the left kidney. B, Intraoperative CT scan with the patient in prone position and a multitine radiofrequency ablation needle deployed into the renal mass. C, Postoperative CT scan at 6 months follow-up showing complete ablation of the left renal mass as demonstrated by the lack of enhancement at the site of the preexisting renal tumor.
Patients who underwent percutaneous ablation under conscious sedation are discharged in a same-day fashion. If the patient required general endotracheal anesthesia or in those patients with significant comorbidities, overnight admission is standard. Again, a CT of the abdomen without contrast is obtained prior to discharge.
Interpretation of Success and Follow-up after Tumor Ablation
Interpretation of treatment success following renal tumor ablation remains a controversial subject. Much of this confusion stems from a lack of uniformity with regard to follow-up protocols (frequency of follow-up and the type of imaging study chosen), as well as a lack of consensus regarding radiographic parameters of success and the role of renal biopsy. The aggregate duration of follow-up with either cryoablation or RFA is inadequate to determine if radiographic follow-up alone is a sufficient surrogate of treatment efficacy (Boorjian and Uzzo, 2009). Routine postablative biopsy may serve a role in corroborating radiographic findings. However, the interpretation of biopsy findings following ablation is highly contentious, and the overall utility of biopsy in this setting is unresolved. Selective biopsy is appropriate and indicated in cases of suspected recurrence. Given these ambiguities, urologists who employ ablative technologies should be familiar with existing and ongoing evidence-based literature and must place these findings in the context of clinical judgment and the overall well being of the patient.
Radiographic Interpretation of Success
As no pathologic margins are rendered with in-situ ablation, imaging characteristics serve as a surrogate marker of treatment efficacy. In general, the complete loss of contrast enhancement on follow-up CT or MRI is considered evidence of complete tissue destruction and attendant treatment success (Matsumoto et al, 2004; McAchran et al, 2005). If a lesion demonstrates persistent contrast enhancement following treatment or requires more than one ablation session to achieve loss of contrast enhancement, this is considered incomplete ablation (Matin et al, 2006). Conversely, if a lesion demonstrates an initial complete loss of contrast enhancement and later demonstrates enlargement of the lesion and/or contrast enhancement, this is considered local tumor recurrence or progression (Novick et al, 2009).
In addition to contrast-related characteristics, lesions that undergo cryoablation or RFA demonstrate characteristic but strikingly different appearances on follow-up imaging. The majority of lesions treated with cryoablation demonstrate a greater than 50% reduction in size in the first year following treatment (Deane and Clayman, 2006; Kawamoto et al, 2009). This contraction is due to cellular breakdown and phagocytosis. Conversely, lesions treated with RFA often do not demonstrate postablative contraction, and failure to do so should not be interpreted as a treatment failure. Enlargement of a lesion, regardless of the treatment modality or the enhancement characteristics, should be construed as an ominous sign of local tumor recurrence with biopsy and/or treatment (observation vs. repeat ablation vs. extirpative surgery) dictated by the size and location of the lesion, the response to the original treatment, and the patient’s overall and renal functional status.
Recommended Radiographic Follow-up Protocol
The optimal radiographic follow-up regimen is not yet defined. The plurality of high-volume centers have determined that CT with intravenous contrast or MRI with gadolinium contrast should be performed at 3 months, 6 months, 9 months, 12 months, 18 months, and 24 months following treatment (Deane, 2006; Matin et al, 2006; Carraway et al, 2009; Crouzet et al, 2009). Imaging should be repeated annually thereafter. There is no available data that cites the superiority of MRI or CT in routine follow-up although some experts contend that CT better distinguishes tumor margins and enhancement in the evaluation of renal tumors (Cadeddu, 2008). Ultrasonography is not routinely employed to evaluate lesions following ablation.
Role of Pre- and Postablation Biopsy
One of the chief criticisms of in-situ renal ablation has been the inability to render definitive pathologic evidence of treatment success. Efficacy with ablative technologies is therefore predicated solely on indirect radiographic cues as previously discussed. In an attempt to address this issue and substantiate the role of postablation radiographic evaluation, biopsy of the lesion before and after treatment has been advocated (Kunkle and Uzzo, 2008).
Historically, biopsy of suspected renal cell carcinoma (RCC) has rarely been performed prior to treatment owing to perceived diagnostic inaccuracies in specimen interpretation, as well as the high overall diagnostic accuracy of cross-sectional imaging (Wang and Wood, 2009). Unfortunately, this reticence to perform biopsy prior to extirpative surgery has likewise carried over to cryoablation and RFA. A recent meta-analysis performed by Kunkle and Uzzo (2008) demonstrated that preablation biopsy was performed in 83.2% of cryoablation cases and 62.2% of RFA cases (P < .0001). Among those that underwent biopsy and subsequent ablation, 53.9% demonstrated pathologic evidence of RCC. The remaining 46.1% of lesions were either benign (12.7%) or indeterminate (33.5%). In light of these ambiguities, accurately interpreting cancer specific outcomes following cryoablation and RFA is extremely challenging. To establish a diagnosis and provide uniformity and improved outcomes-based data, the American Urological Association Small Renal Mass (AUA SRM) Guidelines Panel recently recommended that tumor biopsy be universally performed at the time of ablation (Novick et al, 2009).
Much of the controversy surrounding the oncologic efficacy of ablative technologies has centered on the inherent reliability of postoperative cross-sectional imaging to accurately predict disease-free status. It was originally thought that the high overall diagnostic accuracy of CT or MRI in predicting malignancy preoperatively would translate into equally accurate predictions of postablation tumor destruction or tumor recurrence. However, recent studies have questioned this bias and invoked uncertainty regarding the utility and adequacy of radiographic imaging as the sole surrogate of treatment success, especially following RFA.
In 2002, Rendon and colleagues (2002) performed laparoscopic or percutaneous RFA prior to partial or radical nephrectomy in 10 patients (11 tumors), with a mean tumor size of 2.4 cm. Hematoxylin and eosin (H&E) staining demonstrated residual viable tumor in 7 of the 11 tumors that underwent treatment. Four years later, Park and colleagues (2006b) reported on three patients who underwent delayed nephrectomy at a mean follow-up of 18 months following RFA. Intervention was performed due to new enhancement at the periphery of the treated lesion in two cases and treatment-related ureteropelvic junction obstruction in the remaining patient. No evidence of residual tumor was noted in any of the specimens.
Weight and colleagues (2008) attempted to correlate the radiographic appearance of ablated renal masses with pathological outcomes. The group examined a total of 109 renal lesions in 88 patients who underwent percutaneous RFA and 192 lesions in 176 patients who underwent laparoscopic cryoablation. At 6 months following treatment, radiographic success (no evidence of contrast enhancement) was noted in 90% of patients who underwent cryoablation and in 85% of patients who underwent RFA. Biopsy at 6 months was then performed in 134 lesions (45%). Pathologic evidence of success was 93.8% with cryoablation and only 64.8% with RFA. Of note, 6 of the 13 patients who underwent RFA and demonstrated viable residual tumor on follow-up biopsy demonstrated no contrast enhancement on follow-up imaging. Conversely, all cryoablation patients who had residual tumor on follow-up biopsy demonstrated definitive contrast enhancement. The authors concluded that radiographic imaging results following cryoablation correlated well with pathologic results, whereas a poor correlation was noted between radiographic imaging and pathologic results following RFA. Routine biopsy following RFA was advocated by the authors.
Although the Weight and colleagues study (2008) was criticized for potential selection bias, it nevertheless highlights the long-standing dilemma of post-RFA biopsy interpretation. Standard H&E staining may be misleading immediately following RFA, because cellular architecture is largely preserved despite cell death (Margulis et al, 2004). The duration of this “architectural preservation” is unknown. Stern attempted to address this issue by performing tumor biopsy of 20 radiographically negative lesions at least 1 year following RFA (Stern et al, 2008). No evidence of disease was noted. The authors concluded that RFA imparts definitive cellular necrosis, and radiographic imaging results correlate well with histopathology. Given such conflicting information, the ultimate utility of H&E staining following RFA remains unknown.
Cell viability stains, primarily reduced nicotinamide adenine dinucleotide (NADH) diaphorase, have been proposed as an alternative and more accurate method of determining cell death following tumor ablation. Much like the aforementioned H&E studies, results with NADH staining have been conflicting. Marcovich and coworkers (2003) performed a porcine study in which renal tumors were ablated and later resected and examined histologically. Tumor architecture was variably preserved on H&E staining (as previously discussed) but no NADH diaphorase staining was noted (implying complete cell death). Conversely, Matlaga and colleagues (2002) performed biopsy following RFA in 10 patients and noted an absence of NADH activity in 8 patients. The remaining two patients stained positive for NADH but technical (maximum treatment temperature of 41° C) and tumor-related (8-cm lesion) confounders may account for these failures. Michaels and colleagues (2002) likewise performed RFA in 15 patients and noted positive NADH staining in 4 of 5 tumors that underwent completion nephrectomy. The inherent value and accuracy of NADH staining following RFA remains controversial.
Key Points: Follow-up After Tumor Ablation
Oncologic Outcomes
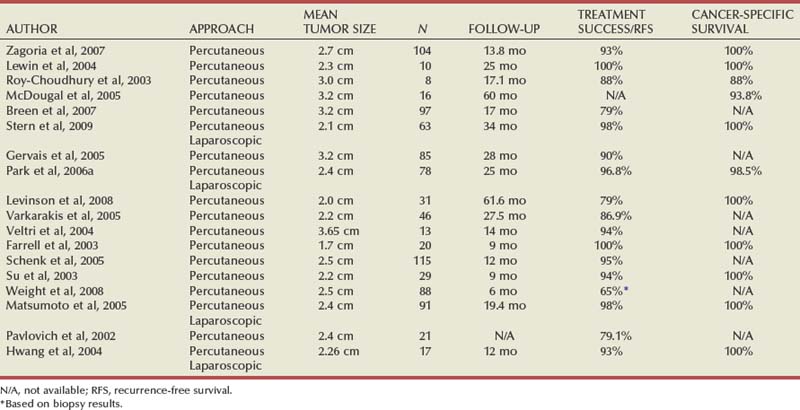
The interpretation of oncologic outcomes following renal tumor ablation is challenging. In general, ablative technologies have been thought to offer inferior cancer control compared to better-established extirpative alternatives. A recent meta-analysis demonstrating a higher relative risk of recurrence with cryoablation and RFA would seem to support this (Kunkle et al, 2008). However, the majority of published studies on cryoablation and RFA have enrolled small numbers of patients and employed disparate operative and follow-up protocols. As was explained, pathology is either not obtained or is difficult to interpret, and the reliability of radiographic imaging remains unknown. A veritable litany of commercially available and ever-evolving treatment systems and probes have further compounded the issue. Perhaps most importantly, collective follow-up is too short to derive meaningful conclusions. Much like radiation treatment for prostate cancer, a discussion of oncologic efficacy regarding ablative technologies must focus less on defined and immediately gratifying end points, such as margin status, and more on long-term markers of success, such as metastasis-free survival, cancer-specific survival, and quality-of-life outcomes. In an attempt to minimize inherent flaws with single institutional series, we have elected to present oncologic outcomes in a more evidence-based and systematic fashion. Individual salient studies of cryoablation and RFA are found in Tables 56-1 and 56-2.
Table 56–1 Cumulative Outcomes of In-Situ Tumor Cryoablation for the Treatment of Small Renal Masses
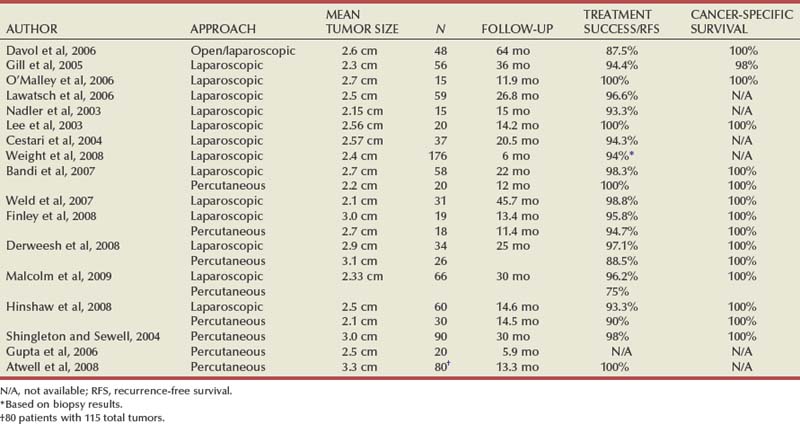
Table 56–2 Cumulative Outcomes of In-Situ Tumor Radiofrequency Ablation for the Treatment of Small Renal Masses
Local Recurrence-Free Survival (LRFS)
Recent meta-analyses have evaluated the risk of local tumor recurrence following cryoablation and RFA (Kunkle et al, 2008; Novick et al, 2009). Local recurrence was defined as “any disease presence in the treated kidney or associated renal fossa posttreatment” and for ablation studies was defined as “any localized disease remaining in the treated kidney at any point after the first ablation.” The AUA SRM Guidelines Panel examined 10 studies of cryoablation and 10 studies of RFA and determined a local recurrence-free survival of 90.6% (83.8% to 94.7%) for cryoablation and 87.0% (83.2% to 90%) for RFA. When compared to alternative extirpative treatments, both cryoablation and RFA demonstrated significantly higher rates of local recurrence despite a shorter overall duration of follow-up. There was no significant difference in local recurrence-free survival between cryoablation and RFA.
Kunkle and colleagues (2008) performed a landmark meta-analysis in which treatment outcomes with partial nephrectomy, cryoablation, RFA, and active surveillance were evaluated. A total of 99 studies and 6,471 tumors were included in the analysis. The relative risk of local recurrence was significantly higher with cryoablation (RR = 7.45) and RFA (RR = 18.23), respectively, when compared to partial nephrectomy. This same group performed a follow-up subanalysis including 1,375 renal tumors (47 total published series from 45 institutions) that underwent cryoablation (n = 600) or RFA (n = 775) alone (Kunkle and Uzzo, 2008). The rate of local tumor progression was 5.2% following cryoablation and 12.9% following RFA. The risk of local recurrence was significantly higher in the RFA cohort on univariate (P = .001) and multivariate (P = .003) analysis.
Metastatic Recurrence-Free Survival (MRFS)
The AUA SRM Guidelines Panel additionally examined the risk of metastatic recurrence, which was defined as “any disease presence in the body other than in the treated kidney or associated renal fossa post-treatment.” Mean metastatic recurrence-free survival was 95.3% (91.1% to 97.5%) for cryoablation and 97.5% (94.8% to 98.8%) for RFA (Novick et al, 2009). Interestingly, there was no significant difference in metastatic recurrence-free survival between the ablative technologies and alternative extirpative treatments including open partial nephrectomy and laparoscopic radical nephrectomy. There was a significant difference in MRFS in favor of laparoscopic partial nephrectomy. The authors did note that duration of follow-up was inadequate to draw conclusions and selection bias may have confounded the analysis.
In Kunkle’s initial meta-analysis, the relative risk of metastatic disease was not significantly different between cryoablation, RFA, active surveillance, and partial nephrectomy (Kunkle et al, 2008). Subsequent analysis of ablation patients alone demonstrated progression to metastatic disease in 1% of patients who underwent cryoablation and 2.5% of patients who underwent RFA (P = .06) (Kunkle and Uzzo, 2008).
Cancer-Specific Survival (CSS)
The AUA meta-analysis additionally examined the risk of dying from RCC following cryoablation (six studies were included) or RFA (eight studies were included). CSS was 95.2% (89.2% to 97.9%) with cryoablation and 98.1% (95.2% to 99.2%) with RFA. CSS was significantly higher with laparoscopic partial nephrectomy than for cryoablation but there was no significant difference in CSS between LPN and RFA. There was no significant difference in CSS between cryoablation and RFA. Again, selection bias and short duration of follow-up may confound these results.
Overall Survival
Mean overall survival rate following cryoablation was 95.8% (87.2% to 98.7%) and 93.7% (85.4% to 97.4%) for RFA. There was no significant difference in overall survival between cryoablation and RFA. Again, limited follow-up and selection bias may confound these results.
Cryoablation versus RFA
Any debate regarding the superiority of cryoablation versus RFA is subject to the same faults, confounders, and ambiguities as the aforementioned comparisons between in-situ tumor ablation and extirpation. Beyond issues of ill-defined radiologic and pathologic end points, there is considerable variability in patient selection, tumor size and location, technique, and approach (laparoscopic vs. percutaneous), as well as inherent bias for a particular ablative modality. To our knowledge, there is no data that directly compares percutaneous and laparoscopic cryoablation to its respective RFA counterparts.
Hegarty and colleagues performed the only published comparative study of cryoablation and RFA in 2006. This retrospective cohort study was performed to determine complications, impact on renal function, follow-up imaging, and oncologic outcomes comparing 164 laparoscopic cryoablation procedures and 82 percutaneous RFA procedures. There was no significant difference in tumor size between the two groups, but the RFA cohort demonstrated more central tumors and tumors in solitary renal units. Radiographic evidence of disease persistence or recurrence was noted in 1.8% of patients who underwent cryoablation and in 11.1% of patients who underwent RFA. There was no significant impact on renal function with either modality. At a median follow-up of 3 years, the CSS for cryoablation was 98%; at a median follow-up of 1 year, the CSS for RFA was 100%. Although the study was successful in articulating the need for improved comparative analysis, its results are difficult to fully embrace given the incongruent patient cohorts and treatment approaches.
Ultimately, the results of the three recent meta-analyses discussed previously offer the most insight into the comparative oncologic merit and potential limitations of cryoablation and RFA (Kunkle et al, 2008; Kunkle and Uzzo, 2008; Novick et al, 2009). Although some data is conflicting, it appears that there is no significant difference in the risk of local tumor recurrence, disease progression/metastasis, cancer-specific survival, and overall survival between the two modalities.
Laparoscopic versus Percutaneous Renal Tumor Ablation
One proposed explanation for the higher local recurrence rate with RFA in some series centers on the adequacy of tumor ablation by the percutaneous approach. Indeed, the vast majority of cryoablation procedures are approached laparoscopically, while nearly 90% of RFA procedures are performed percutaneously (Deane, 2006; Carraway et al, 2009). Crouzet and colleagues (2009) retrospectively compared 244 laparoscopic renal cryoablation procedures with 63 percutaneous renal cryoablation procedures. Patient and tumor characteristics were similar with both cohorts. There was a trend toward prior renal surgery or a solitary renal unit with percutaneous cryoablation. There was no significant difference in renal functional outcomes between the two groups. Duration of hospitalization was shorter in the percutaneous cohort. With a minimum follow-up of 2 years, the percutaneous cohort demonstrated a higher incomplete treatment rate and the need for re-treatment. There was no difference in overall survival, CSS, and recurrence-free survival between the two groups. Similar studies comparing laparoscopic and percutaneous tumor ablation have confirmed these findings (Derweesh et al, 2008; Hinshaw et al, 2008; Hui et al, 2008; Malcolm et al, 2009).
In contradistinction to these studies, Finley and colleagues (2008) reported their 4-year experience with laparoscopic and percutaneous renal cryoablation in 37 patients with 43 tumors. Operative time and duration of hospitalization were significantly shorter with the percutaneous approach, and there was no significant difference in the number of incomplete ablations.
Researchers have investigated exclusive use of general anesthesia and advanced navigational systems as mechanisms to improve outcomes with percutaneous ablation (Gupta et al, 2009; Haber et al, 2009).
Key Points: Oncologic Outcomes
Complications
Complications with in-situ tumor ablation are relatively low. A recent meta-analysis compared urologic and nonurologic complications following cryoablation, radiofrequency ablation, and alternative extirpative approaches (Novick et al, 2009). Major urologic complications were defined as postoperative hemorrhage requiring transfusion or intervention, urinary leak, abscess, and unanticipated loss of renal function. The incidence of major urologic complications with renal cryoablation was 4.9% (range 3.3% to 7.4%). Postoperative hemorrhage is the most commonly cited major adverse risk, with ureteral obstruction, splenic hematoma, and perirenal abscess additionally reported. The risk of major urologic complications with RFA is 6.0% (4.3% to 8.2%). Reported major complications with RFA include ureteral obstruction, pyelocalyceal injury, bowel injury, pancreatic injury, and/or gross hematuria requiring intervention. Of note, the risk of major urologic complications was lower with ablative techniques than with either laparoscopic or open partial nephrectomy, and there was no significant difference in urologic complications with cryoablation versus RFA.
Major nonurologic complications occurred in 4.9% (3.4% to 7.2%) of cryoablation patients and in 4.4% (3.2% to 6.2%) of RFA patients. Of note, nonurologic complications were significantly more common with cryoablation than with RFA or open partial nephrectomy. This higher complication rate with cryoablation may reflect the older patient population in the cryoablation cohort. Commonly cited minor complications with cryoablation and RFA include pain and paresthesia at the probe insertion site, urinary tract infections, and self-limited hematuria.
Conversion was defined in the meta-analysis as a deviation from the intended surgical procedure or approach. The overall conversion rate for cryoablation was 3.5% with bleeding, adhesions, and respiratory difficulty cited as common reasons for conversion. The conversion rate for RFA was 1.6% with proximity to bowel as the only cited reason. Of note, only one RFA study was included in the analysis. Deriving meaningful conclusions regarding the superiority of RFA versus cryoablation with regard to conversions is therefore difficult.
The cited rate of transfusion with cryoablation is 3.2% (2.0% and 20%) and is 2.4% (1.4% to 40%) with RFA. Indeed, the most commonly encountered complication with cryoablation is bleeding, especially in the setting of tumor or renal fracture (up to 17% of patients with renal fracture require transfusion). Lehman and colleagues (2008) additionally noted that tumors greater than 3.0 cm in size were significantly more likely to require a blood transfusion (38%). Bleeding following RFA is relatively uncommon given the ability to perform a hemostatic “track burn” on probe withdrawal.
Reintervention was defined in the meta-analysis as any unplanned operation occurring during or after the planned renal surgery. Reintervention occurred in 2.6% (1.5% to 4.3%) of cryoablation patients and 3.2% (1.9% to 5.1%) of RFA patients. The risk of reintervention was significantly higher in patients undergoing laparoscopic partial nephrectomy than with either ablative technique.
Key Points: Complications Following Tumor Ablation
Future Trends
Extracorporeal High-Intensity Focused Ultrasonography (HIFU)
As an acoustic wave is propagated through tissue, a portion of its energy is absorbed and converted into heat (Madersbacher et al, 1995; Vricella et al, 2009). When ultrasound beams are tightly concentrated, temperature levels approach or exceed the threshold for cell kill (65° C to 85° C) within this focused field, while adjacent tissue is spared. At high enough intensities (>3500 W/cm3), cavitation and microbubble formation occurs that yields extremely high temperatures and a mechanically disrupting “shock wave” effect similar to that seen with extracorporeal shock wave lithotripsy (Kieran et al, 2007). Cavitary forces are extremely difficult to control during HIFU and are generally avoided.
HIFU employs a transducer that is used for treatment and monitoring. The ultrasound beams are focused geometrically through the transducer by employing an acoustic lens or a concave shape. Under real-time guidance, the HIFU beam is focused on the treatment zone and a defined area is ablated. The transducer can then be refocused to ablate a larger overall volume of tissue. Treatment times can be lengthy with a mean reported duration of nearly 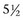 hours (1.5 to 9 hours) (Köhrmann et al, 2002; Marberger et al, 2005; Häcker et al, 2006). Myriad parameters, including focal length, type of transducer employed, and type of treatment system, have been investigated and are beyond the scope of this chapter.
hours (1.5 to 9 hours) (Köhrmann et al, 2002; Marberger et al, 2005; Häcker et al, 2006). Myriad parameters, including focal length, type of transducer employed, and type of treatment system, have been investigated and are beyond the scope of this chapter.
Certainly, the promise of a noninvasive ablative technology that offers favorable and reproducible outcomes is extremely attractive. However, existing studies have yielded mixed results. Vallancien and colleagues (1993) performed a pilot study in which eight patients underwent targeted transcutaneous HIFU ablation followed by specimen excision. Approximately 10% of the cohort exhibited skin burns, and all specimens demonstrated pathologic evidence of viable tumor. Marberger and colleagues (2005) evaluated 18 renal units treated with HIFU followed by specimen extraction. Similar to the findings of Vallencien, incomplete ablation was noted in all cases. Häcker and colleagues (2006) additionally assessed the efficacy of HIFU by treating normal portions of 24 kidneys prior to preplanned completion radical nephrectomy. Coagulative necrosis was sporadic and inconsistent with the number of treatments applied. Again, skin burns were noted in two patients. Purported explanations for these collective incomplete treatments have included poor targeting secondary to respiratory movement and acoustic interference (acoustic shadowing, reverberation, and refraction), as well as system limitations including limited focal zone depth and an inability to monitor treatment progression in real time. Recently, laparoscopic HIFU has been investigated in an attempt to circumvent issues related to targeting, acoustic interference, and respiratory movement (Klingler et al, 2008). Results are anecdotal but favorable. In summary, outcomes with renal HIFU have proven inferior to alternative ablative technologies, and its use in this regard should be considered investigational.
Radiation Therapy
Historically, radiation therapy has been considered ineffective in the treatment of renal cell carcinoma. Although RCC is widely considered “radioresistant,” it is unclear if this assertion is justifiable. It remains unclear if poor outcomes with radiation therapy for RCC are due to an inherent resistance to radiation or due to limitations with radiation delivery (Camphausen and Coia, 2008). Modern external beam radiation systems are inadequately designed to deliver high doses in a focal manner. With conventional treatment systems, high-dose radiation is associated with significant radiation scatter and attendant damage to adjacent tissues. Brachytherapy is better tailored for focal ablation, but its logistic application (seed placement in the kidney) for renal indications remains impractical.
Stereotactic radiosurgery (SRS) employs 3-dimensional coordinates to target and focally ablate tissue using high-dose external beam radiation. As opposed to conventional radiation delivery techniques, modern stereotactic treatment systems compensate for respiratory movement and radiation scatter by automatically tracking, detecting, and correcting for tumor and/or organ movement without interrupting the treatment or repositioning the patient. This tracking system is image guided and dependent on a constant reference point (fiducial marker) that is continually recognized by the linear accelerator. High-dose radiation beams move in real time with the respiratory cycle and are therefore extremely accurate (Ponsky et al, 2007). Not only is radiation scatter minimized, but higher doses may be applied in a focal manner that effectively ablates masses in the kidney without compromising overall renal function. The most commonly used stereotactic radiosurgical system is termed CyberKnife (Accuray, Palo Alto, CA). The CyberKnife is a 6-MV frameless linear accelerator mounted on a computer-controlled robotic arm. An image-guidance system as described above is employed to continually monitor movement of the target organ and deliver high-dose radiation in a focused fashion.
Ponsky and colleagues (2003) evaluated stereotactic radiosurgery in the treatment of renal tissue. Eight pigs underwent bilateral renal irradiation using the CyberKnife treatment system. Treatment doses of between 24 to 40 Gy were administered. The pigs were sacrificed 4 to 8 weeks following ablation. The treatment zone demonstrated complete tissue ablation with no collateral damage to adjacent tissue. Building on this initial animal experiment, Ponsky subsequently performed a Phase I study on three human patients with a mean renal tumor size of 2.03 cm. A total of 16 Gy were administered in a fractionated fashion. Patients were followed for 8 weeks, after which a partial nephrectomy was performed (a so-called treat and cut study). No adverse events or radiation toxicities were noted. Examination for pathology demonstrated residual RCC in two patients and no evidence of viable tumor in the remaining patient. However, low doses were used in this initial feasibility study, and dose escalation trials are ongoing.
Svedman and colleagues (2006) performed a retrospective study evaluating the efficacy and safety of stereotactic radiosurgery in the management of inoperable or metastatic primary renal cell carcinoma. Thirty patients with 82 lesions underwent treatment with varied dose/fractionation schedules. At a median follow-up of 52 months, complete response was noted in 21% of patients, with another 58% demonstrating a partial/stable response. Overall survival was 32 months.
Certainly, the responsiveness of RCC to stereotactic radiosurgery in the aforementioned trials argues against its radioresistant reputation. Although its use should still be considered experimental, with improved treatment protocols and well-designed prospective trials, SRS may ultimately play a significant role in the treatment of RCC.
Microwave Thermotherapy (MWT)
MWT delivers energy through flexible antennae that are inserted directly into the target lesion. Microwave energy creates an electromagnetic field that causes rapid ion oscillation and frictional heat. Although ostensibly similar in design and technique to RFA, MWT is capable of achieving treatment temperatures (>60° C) with greater rapidity. Moreover, MWT is not limited by tissue charring and desiccation and is able to achieve high treatment temperatures over a broader area. These advantages may translate into more efficient treatments and improved cell kill. Although theoretical, there is also some evidence that MWT is less susceptible to the heat-sink phenomenon and may therefore be applied in the treatment of highly vascular lesions (Liang and Wang, 2007).
MWT was initially designed for the percutaneous treatment of hepatocellular carcinoma and has enjoyed considerable success in this capacity. Its use in the management of renal tumors remains investigational, with sporadic feasibility studies supporting its use. MWT was initially employed during unclamped open partial nephrectomy to create an avascular plane prior to tumor resection. More recently, MWT has been investigated as a therapeutic in-situ renal ablative device. Clark and colleagues (2007) performed a Phase I study in which 10 patients underwent MWT of suspected RCC at the time of radical nephrectomy. When examined pathologically, ablation lesions as large as 5.7 cm × 4.7 cm × 3.8 cm were achieved with complete and uniform tissue necrosis. The authors advocated “treat and leave” studies to confirm their findings. In 2008, Liang and colleagues published their series of 12 patients who underwent percutaneous MWT under ultrasound guidance (a so-called treat and leave protocol). All patients demonstrated pathologically proven unilateral renal cell carcinomas that were less than 4 cm, not adjacent to the collecting system or bowel, and were accessible through a percutaneous approach. One or two antennae were employed (depending on tumor size) to ablate the renal lesion, and thermocouples were used to monitor treatment temperatures in real time. No significant adverse events were reported following treatment. All patients underwent follow-up imaging at 1 day, 1 month, 3 months, and 6 months following treatment. At a median follow-up of 11 months, no cancer recurrence was noted on imaging.
MWT offers considerable promise as yet another minimally invasive ablative technology. However, until prospective studies are available, its use should remain investigational.
Laser Interstitial Thermal Therapy (LITT)
LITT employs specialized laser fibers to delivery energy directly into tissue. These fibers emit laser light that is converted to heat. Treatment temperatures of greater than 55° C are achieved, and tissue necrosis results. Thus far, LITT has relied on neodymium : yttrium-aluminum-garnet (Nd : YAG) lasers and diode lasers. Results have been difficult to interpret owing to the small number of treated patients and a lack of clinical follow-up (Dick et al, 2002; Gettman et al, 2002b; Williams et al, 2000). The use of LITT remains investigational.
Navigational Systems
In an attempt to better localize, control, and predict the area of treatment during percutaneous cryoablation, novel navigational and targeting systems have been developed. Haber and colleagues (2009) performed percutaneous renal cryoablation of 13 lesions in 10 patients using a stereotactic navigational system (CT-Nav; Keolis, La Tronche, France) designed to direct renal biopsy and probe placement. Patients underwent preoperative CT scan with a preplaced tracking sensor taped to the body of the patient. Using a stereoscopic infrared camera, the tracking sensor was located three-dimensionally and a tracking handle used to guide probe placement. The mean calculated error in targeting with use of the navigational device was 4.2 mm. The most significant variability occurred in the cephalad/caudad axis consistent with respiratory movement. Although purely experimental, these tracking and targeting systems may represent a significant advance in the percutaneous application of ablative technologies.
Single Port Surgery/NOTES
Developed as an extension of standard laparoscopy, laparoendoscopic single-site (LESS) surgery seeks to minimize patient discomfort, shorten convalescence, and improve cosmesis by placing all instruments through a 2 cm incision, sometimes concealed in the umbilicus (Kaouk et al, 2008).
In 2008, Goel and colleagues published their clinical experience with single-port access renal cryoablation (SPARC) in six patients with small renal masses (Goel and Kaouk, 2008b). Using either a transperitoneal or retroperitoneal approach, all procedures were successfully completed without intraoperative complications. A subsequent publication from the same group cited eight total single-port cryoablation procedures (White et al, 2009). Mean tumor size was 2.64 cm. Mean operative time was 171 minutes. Two patients required postoperative blood transfusions. No recurrences were noted on follow-up imaging. The authors stated that renal ablative procedures may represent an attractive and natural transition into single-port surgery as limited dissection and reconstruction are needed.
Natural orifice translumenal endoscopic surgery (NOTES) employs a natural orifice, such as the vagina, bladder, stomach, or colon, as the entry point to the peritoneal cavity. Ostensibly, NOTES should offer superior cosmesis and decreased pain compared to conventional laparoscopy. However, operating through a natural orifice is technically very challenging with only two NOTES extirpative urologic procedures successfully completed (Kaouk et al, 2009). Much like single-port surgery, NOTES is ideally suited for ablative procedures that require minimal dissection and reconstruction. Crouzet and colleagues (2009) reported bilateral transgastric and transvesical renal cryoablation in two female farm pigs. All four procedures were successfully completed without the need for additional ports. There were no intraoperative complications.
Chemoablation and Combined Chemoablation with RFA
The percutaneous injection of 95% ethanol to ablate renal cysts was originally described by Bean in 1981. This conceptual approach was later applied in the treatment of hepatocellular carcinoma with moderate success. Clayman and colleagues later evaluated injectable 95% ethanol, 24% hypertonic saline gel, and 50% acetic acid gel alone or in combination with RFA in a porcine study (Rehman et al, 2004). The authors demonstrated inadequate tissue necrosis with any of the injectables alone but noted enhanced tissue necrosis with the use of chemoablative agents in conjunction with RFA. The combined use of hypertonic saline with RFA produced the largest overall lesions. A second study from Fotiadis and colleagues (2007), in the United Kingdom, evaluated combined RFA and ethanol injection in 27 patients (28 total tumors) with a mean tumor size of 2.87 cm. Absolute ethanol was injected into the tumor just prior to RFA treatment. At a mean follow-up of approximately 18 months, 27 of 28 tumors were completely ablated with one (21 tumors) or two (6 tumors) treatments. No local recurrence or metastases occurred. Currently, injectable chemoablation agents are difficult to reproducibly control and provide inferior tissue necrosis. Combined RFA/injectable chemoablation remains investigational.
Targeted Embolization and Ablation
Owing to the heat-sink phenomenon with RFA, highly vascular central lesions or lesions positioned adjacent to the renal hilum are often inadequately ablated. Studies estimate treatment failures as high as 40%. In an attempt to address conductive heat loss, investigators have performed selective arterial embolization prior to RFA (Yamakado et al, 2006; Gebauer et al, 2007; Mahnken et al, 2009). Theoretically, selective embolization should allow for more homogenous heating and improved tissue necrosis. Clinical reports are sporadic and anecdotal. Therefore the use of targeted angioembolization prior to RFA remains investigational.
Conclusions
The role of in-situ ablation in the management of small renal masses is rapidly evolving. Once considered experimental and appropriate only for patients with significant comorbidities, cryoablation and radiofrequency ablation are currently considered viable alternatives to extirpative management. In-situ ablation confers less treatment-related morbidity than either open or laparoscopic partial nephrectomy and offers superior renal preservation compared with open or laparoscopic nephrectomy. Cryoablation and RFA are technically less challenging than other nephron-sparing approaches and may allow general urologists without advanced laparoscopic training to selectively provide their patients with a minimally invasive nephron-sparing treatment. Results from recent meta-analyses demonstrate inferior local tumor control compared with partial and radical nephrectomy, but with equivalent or superior cancer-specific and overall survival. Interpretation of treatment success remains challenging with existing protocols, and long-term follow-up is needed to confirm these findings. No prospective literature currently exists that addresses the superiority of cryoablation or RFA. Some evidence exists that percutaneous tumor ablation offers incomplete primary treatment, but with improved recovery and decreased morbidity. Without question, prospective, comparative trials with uniform radiographic and pathologic follow-up are needed to address many of these uncertainties. Ultimately, the decision to treat a small renal mass with an ablative technology should take into account tumor-related characteristics, patient demographics and comorbidities, and the values and desires of the patient.
Carraway WA, Raman JD, Cadeddu JA. Current status of renal radiofrequency ablation. Curr Opin Urol. 2009;19:143-147.
Deane LA, Clayman RV. Review of minimally invasive renal therapies: needle-based and extracorporeal. Urology. 2006;68:26-37.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178:41-46.
Hegarty NJ, Gill IS, Desai MM, et al. Probe-ablative nephron-sparing surgery: cryoablation versus radiofrequency ablation. Urology. 2006;68:7-13.
Huang WC, Elkin EB, Levey AS, et al. Partial nephrectomy versus radical nephrectomy in patients with small renal tumors—is there a difference in mortality and cardiovascular outcomes? J Urol. 2009;181:55-62.
Kunkle DA, Egleston BL, Uzzo RG. Excise, ablate or observe: the small renal mass dilemma—a meta-analysis and review. J Urol. 2008;179:1227-1234.
Kutikov A, Kunkle DA, Uzzo RG. Focal therapy for kidney cancer: a systematic review. Curr Opin Urol. 2009;19:148-153.
Lane BR, Babineau DC, Poggio ED, et al. Factors predicting renal functional outcome after partial nephrectomy. J Urol. 2008;180:2363-2368.
Miller DC, Saigal CS, Banerjee M, et al. Diffusion of surgical innovation among patients with kidney cancer. Cancer. 2008;112:1708-1717.
Novick AC, Campbell SC, Belldegrun A, et al. Guideline for management of the clinical stage 1 renal mass. http://www.auanet.org/content/guidelines-and-quality-care/clinical-guidelines/main-reports/renalmass09.pdf, 2009. [accessed 11.02.11]
Ponsky LE, Crownover RL, Rosen MJ, et al. Initial evaluation of Cyberknife technology for extracorporeal renal tissue ablation. Urology. 2003;61:498-501.
Riggs SB, Larochelle JC, Belldegrun AS. Partial nephrectomy: a contemporary review regarding outcomes and different techniques. Cancer J. 2008;14:302-307.
Weight CJ, Kaouk JH, Hegarty NJ, et al. Correlation of radiographic imaging and histopathology following cryoablation and radio frequency ablation for renal tumors. J Urol. 2008;179:1277-1281.
Abouassaly R, Finelli A, Timilshina N, et al. Treatment trends for renal cell carcinoma in a population-based tumor registry: the potential underuse of partial nephrectomy. J Urol. 2009;181:76.
Andonian S, Janetschek G, Lee BR. Laparoscopic partial nephrectomy: an update on contemporary issues. Urol Clin North Am. 2008;35:385-396.
Aron M, Gill IS. Minimally invasive nephron-sparing surgery (MINSS) for renal tumors part I: laparoscopic partial nephrectomy. Eur Urol. 2007;51:337-346.
Atwell TD, Farrell MA, Leibovich BC, et al. Percutaneous renal cryoablation: experience treating 115 tumors. J Urol. 2008;179:2136-2141.
Auge BK, Santa-Cruz RW, Polascik TJ. Effect of freeze time during renal cryoablation: a swine model. J Endourol. 2006;20:1101-1105.
Bandi G, Wen CC, Hedican SP, et al. Cryoablation of small renal masses: assessment of the outcome at one institution. BJU Int. 2007;100:798-801.
Bean WJ. Renal cysts: treatment with alcohol. Radiology. 1981;138:329-331.
Boorjian SA, Uzzo RG. The evolving management of small renal masses. Curr Oncol Rep. 2009;11:211-217.
Breen DJ, Rutherford EE, Stedman B, et al. Management of renal tumors by image-guided radiofrequency ablation: experience in 105 tumors. Cardiovasc Intervent Radiol. 2007;30:936-942.
Cadeddu JA. Editorial comment: correlation of radiographic imaging and histopathology following cryoablation and radio frequency ablation for renal tumors. J Urol. 2008;179:1281-1282.
Campbell SC, Krishnamurthi V, Chow G, et al. Renal cryosurgery: experimental evaluation of treatment parameters. Urology. 1998;52:29-33.
Camphausen KA, Coia LR. Principles of radiation therapy. In Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ, editors: Cancer management: a multidisciplinary approach, 11th ed, Lawrence (KS): CMP Medica, 2008.
Carraway WA, Raman JD, Cadeddu JA. Current status of renal radiofrequency ablation. Curr Opin Urol. 2009;19:143-147.
Cestari A, Guazzoni G, Dell’Acqua V, et al. Laparoscopic cryoablation of solid renal masses: intermediate term followup. J Urol. 2004;172:1267-1270.
Chang I, Mikityansky I, Wray-Cahen D, et al. Effects of perfusion on radiofrequency ablation in swine kidneys. Radiology. 2004;231:500-505.
Chosy SG, Nakada SY, Lee FTJr, et al. Monitoring renal cryosurgery: predictors of tissue necrosis in swine. J Urol. 1998;159:1370-1374.
Clark PE, Woodruff RD, Zagoria RJ, et al. Microwave ablation of renal parenchymal tumors before nephrectomy: phase I study. AJR. 2007;188:1212-1214.
Collyer WC, Landman J, Olweny EO, et al. Comparison of renal ablation with cryotherapy, dry radiofrequency, and saline augmented radiofrequency in a porcine model. J Am Coll Surg. 2001;193:505-513.
Colombo JRJr, Kaouk JH. Minimally invasive management of renal cancer. Minerva Urol Nefrol. 2007;59:159-165.
Cooper IS. A new method of destruction of extirpation of benign and malignant tissues. N Engl J Med. 1963;263:741-749.
Corwin TS, Lindberg G, Traxer O, et al. Laparoscopic radiofrequency thermal ablation of renal tissue with and without hilar occlusion. J Urol. 2001;166:281-284.
Crouzet S, Haber GP, Kamoi K, et al. Renal cryoablation: a comparative analysis between laparoscopic and percutaneous approaches. J Urol. 2009;181:467.
Curley SA, Izzo F, Delrio P, et al. Radiofrequency ablation of unresectable primary and metastatic hepatic malignancies: results in 123 patients. Ann Surg. 1999;230:1-8.
Curley SA, Izzo F, Ellis LM, et al. Radiofrequency ablation of hepatocellular cancer in 110 patients with cirrhosis. Ann Surg. 2000;232:381-391.
Cushing H, Bovie WT. Electro-surgery as an aid to the removal of intracranial tumors. Surg Gynecol Obstet. 1928;47:751-784.
Davol PE, Fulmer BR, Rukstalis DB. Long-term results of cryoablation for renal cancer and complex renal masses. Urology. 2006;68:2-6.
Deane LA, Clayman RV. Review of minimally invasive renal therapies: needle-based and extracorporeal. Urology. 2006;68:26-37.
Derweesh IH, Malcolm JB, Diblasio CJ, et al. Single center comparison of laparoscopic cryoablation and CT-guided percutaneous cryoablation for renal tumors. J Endourol. 2008;22:2461-2467.
Desai MM, Aron M, Gill IS. Laparoscopic partial nephrectomy versus laparoscopic cryoablation for the small renal tumor. Urology. 2005;66:23-28.
Desai MM, Gill IS. Current status of cryoablation and radiofrequency ablation in the management of renal tumors. Curr Opin Urol. 2002;12:387-393.
Dick EA, Joarder R, DeJode MG, et al. Magnetic resonance imaging-guided laser thermal ablation of renal tumours. BJU Int. 2002;90:814-822.
Djavan B, Partin AW, Hoey MF, et al. Transurethral radiofrequency therapy for benign prostatic hyperplasia using a novel saline-liquid conductor: the virtual electrode. Urology. 2000;55:13-16.
Farrell MA, Charboneau WJ, DiMarco DS, et al. Imaging-guided radiofrequency ablation of solid renal tumors. AJR Am J Roentgenol. 2003;180:1509-1513.
Fergany AF, Hafez KS, Novick AC. Long-term results of nephron sparing surgery for localized renal cell carcinoma: 10 year follow-up. J Urol. 2000;163:442-445.
Finelli A, Rewcastle JC, Jewett MAS. Cryotherapy and radiofrequency ablation: pathophysiologic basis and laboratory studies. Curr Opin Urol. 2003;13:187-191.
Finley DS, Beck S, Box G, et al. Percutaneous and laparoscopic cryoablation of small renal masses. J Urol. 2008;180:492-498.
Fotiadis NI, Sabharwal T, Morales JP, et al. Combined percutaneous radiofrequency ablation and ethanol injection of renal tumors: midterm results. Eur Urol. 2007;52:777-784.
Frank I, Blute ML, Cheville JC, et al. Solid renal tumors: an analysis of pathological features related to tumor size. J Urol. 2003;170:2217-2220.
Gage AA. History of cryosurgery. Semin Surg Oncol. 1998;14:99-109.
Gebauer B, Werk M, Lopez-Hänninen E, et al. Radiofrequency ablation in combination with embolization in metachronous recurrent renal cancer in solitary kidney after contralateral tumor nephrectomy. Cardiovasc Intervent Radiol. 2007;30:644-649.
Gervais DA, McGovern FJ, Arellano RS, et al. Radiofrequency ablation of renal cell carcinoma: part 1, indications, results, and role in patient management over a 6-year period and ablation of 100 tumors. AJR. 2005;185:64-71.
Gettman MT, Lotan Y, Corwin TS, et al. Radiofrequency coagulation of renal parenchyma: comparing effects of energy generation on treatment efficacy. J Endourol. 2002;16:83-88.
Gettman MT, Lotan Y, Lindbverg G, et al. Laparoscopic interstitial laser coagulation of renal tissue with and without hilar occlusion in the porcine model. J Endourol. 2002;16:565-570.
Gill IS, Desai MM, Kaouk JH. Laparoscopic partial nephrectomy for renal tumor: duplicating open surgical techniques. J Urol. 2002;167:469-476.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178:41-46.
Gill IS, Novick AG, Meraney AM, et al. Laparoscopic renal cryoablation in 32 patients. Urology. 2000;56:748-753.
Gill IS, Remer EM, Hasan WA, et al. Renal cryoablation: outcome at 3 years. J Urol. 2005;173:1903-1907.
Goel RK, Kaouk JH. Probe ablative treatment for small renal masses: cryoablation vs. radio frequency ablation. Curr Opin Urol. 2008;18:467-473.
Goel RK, Kaouk JH. Single port access renal cryoablation (SPARC): a new approach. Eur Urol. 2008;53:1115-1116.
Goldberg SN, Dupuy DE. Image-guided radiofrequency tumor ablation: challenges and opportunities: part I. J Vasc Interv Radiol. 2001;12:1021-1032.
Goldberg SN, Gazelle GS, Mueller PR. Thermal ablation therapy for focal malignancy: a unified approach to underlying principles, techniques, and diagnostic imaging guidance. AJR Am J Roentgenol. 2000;174:323-331.
Goldberg SN, Gazelle GS, Solbiati L, et al. Radiofrequency tissue ablation: increased lesion diameter with a perfusion electrode. Acad Radiol. 1996;3:636-644.
Gupta A, Allaf ME, Kavoussi LR, et al. Computerized tomography guided percutaneous renal cryoablation with the patient under conscious sedation: initial clinical experience. J Urol. 2006;175:447-453.
Gupta A, Raman JD, Leveillee RJ, et al. General anesthesia and contrast-enhanced computed tomography to optimize renal percutaneous radiofrequency ablation: multi-institutional intermediate-term results. J Endourol. 2009;23:1099-1105.
Haber GP, Crouzet S, O’Malley C, et al. stereotactic percutaneous cryoablation for renal tumor: initial experience. J Urol. 2009;181:182.
Häcker A, Michel MS, Marlinghaus E, et al. Extracorporeally induced ablation of renal tissue by high-intensity focused ultrasound. BJU Int. 2006;97:779-785.
Hegarty NJ, Gill IS, Desai MM, et al. Probe-ablative nephron-sparing surgery: cryoablation versus radiofrequency ablation. Urology. 2006;68:7-13.
Herr HW. Partial nephrectomy for unilateral renal carcinoma and a normal contralateral kidney: 10-year follow up. J Urol. 1999;161:33-35.
Hinshaw JL, Shadid AM, Nakada SY, et al. Comparison of percutaneous and laparoscopic cryoablation for the treatment of solid renal mass. AJR. 2008;191:1159-1168.
Hoffmann NE, Bischof JC. The cryobiology of cryosurgical injury. Urology. 2002;60:40-49.
Hsu TH, Fidler ME, Gill IS. Radiofrequency ablation of the kidney: acute and chronic histology in the porcine model. Urology. 2000;56:872-875.
Huang WC, Elkin EB, Levey AS, et al. Partial nephrectomy versus radical nephrectomy in patients with small renal tumors—is there a difference in mortality and cardiovascular outcomes? J Urol. 2009;181:55-62.
Hui GC, Tuncali K, Tatli S, et al. Comparison of percutaneous and surgical approaches to renal tumor ablation: metaanalysis of effectiveness and complications rates. J Vasc Interv Radiol. 2008;19:1311-1320.
Hwang JJ, Walther MM, Paulter SE, et al. Radiofrequency ablation of small renal tumors: intermediate results. J Urol. 2004;171:1814-1818.
Ishiguro H, Rubinsky B. Mechanical interactions between ice crystals and red-blood cells during directional solidification. Cryobiology. 1994;31:483-500.
Jayson M, Sanders H. Increased incidence of serendipitously discovered renal cell carcinoma. Urology. 1998;51:203-205.
Kahlenberg MS, Volpe C, Klippenstein DL, et al. Clinicopathologic effects of cryotherapy on hepatic vessels and bile ducts in a porcine model. Ann Surg Oncol. 1998;5:713-718.
Kaouk JH, Haber GP, Goel RK, et al. Single-port laparoscopic surgery in urology: initial experience. Urology. 2008;71:3-6.
Kaouk JH, White WM, Goel RK, et al. NOTES transvaginal nephrectomy: first human experience. Urology. 2009;74:5-8.
Kawamoto S, Solomon SB, Bluemke DA, et al. Computed tomography and magnetic resonance imaging appearance of renal neoplasms after radiofrequency ablation and cryoablation. Semin Ultrasound CT MR. 2009;30:67-77.
Kieran K, Hall TL, Parsons JE, et al. Refining histotripsy: defining the parameter space for the creation of nonthermal lesions with high intensity, pulsed focused ultrasound of the in vitro kidney. J Urol. 2007;178:672-676.
Klingler HC, Susani M, Seip R, et al. A novel approach to energy ablative therapy of small renal tumours: laparoscopic high-intensity focused ultrasound. Eur Urol. 2008;53:810-816.
Klossner DP, Robilotto AT, Clarke DM, et al. Cryosurgical technique: assessment of the fundamental variables using human prostate cancer model systems. Cryobiology. 2007;55:189-199.
Köhrmann KU, Michel MS, Gaa J, et al. High intensity focused ultrasound as noninvasive therapy for multilocal renal cell carcinoma: case study and review of the literature. J Urol. 2002;167:2397-2403.
Kunkle DA, Egleston BL, Uzzo RG. Excise, ablate or observe: the small renal mass dilemma—a meta-analysis and review. J Urol. 2008;179:1227-1234.
Kunkle DA, Uzzo RG. Cryoablation or radiofrequency ablation of the small renal mass. Cancer. 2008;113:2671-2680.
Kural AR, Atug F, Tufek I, et al. Robot-assisted partial nephrectomy versus laparoscopic partial nephrectomy: comparison of outcomes. J Endourol. 2009;23(9):1491-1497.
Lane BR, Novick AC, Babineau D, et al. Comparison of laparoscopic and open partial nephrectomy for tumor in a solitary kidney. J Urol. 2008;179:847-851.
Larson TR, Robertson DW, Corica A, et al. In vivo interstitial temperature mapping of the human prostate during cryosurgery with correlation to histopathology outcome. Urology. 2000;55:547-552.
Lawatsch EJ, Langenstroer P, Byrd GF, et al. Intermediate results of laparoscopic cryoablation in 59 patients at the Medical College of Wisconsin. J Urol. 2006;175:1225-1229.
Lee DI, McGinnis DE, Feld R, et al. Retroperitoneal laparoscopic cryoablation of small renal tumors: intermediate results. Urology. 2003;61:83-88.
Lehman DS, Hruby GW, Phillips CK, et al. First prize (tie): laparoscopic renal cryoablation: efficacy and complications for larger renal masses. J Endourol. 2008;22(6):1123-1127.
Lehman DS, Landman J. Kidney cancer ablative therapy: indications and patient selection. Curr Urol Rep. 2008;9:34-43.
LeVeen RF. Laser hyperthermia and radiofrequency ablation of hepatic lesions. Semin Interv Radiol. 1997;14:313-324.
Leveillee RJ, Hoey MF. Radiofrequency interstitial tissue ablation: wet electrode. J Endourol. 2003;17:563-577.
Levinson AW, Su L, Agarwal D, et al. Long-term oncological and overall outcomes of percutaneous radio frequency ablation in high risk surgical patients with a solitary small renal mass. J Urol. 2008;180:499-504.
Lewin JS, Nour SG, Connell CF, et al. Phase II clinical trial of interactive MR imaging-guided interstitial radiofrequency thermal ablation of primary kidney tumors: initial experience. Radiology. 2004;232:835-845.
Liang P, Wang Y. Microwave ablation of hepatocellular carcinoma. Oncology. 2007;72:124-131.
Liang P, Wang Y, Zhang D, et al. Ultrasound guided percutaneous microwave ablation for small renal cancer: initial experience. J Urol. 2008;180:844-848.
Lorentzen T, Christensen NE, Nolsoe CP, et al. Radiofrequency tissue ablation with a cooled needle in vitro: ultrasonography, dose response, and lesion temperature. Acad Radiol. 1997;4:292-297.
Madersbacher S, Vingers L, Marberger M, et al. Effect of high-intensity focused ultrasound on human prostate cancer in vivo. Cancer Res. 1995;55:3346-3351.
Mahnken AH, Penzkofer T, Bruners P, et al. Interventional management of a renal cell carcinoma by radiofrequency ablation with tagging and cooling. Korean J Radiol. 2009;10:523-526.
Malcolm JB, Berry TT, Williams MB, et al. Single center experience with percutaneous and laparoscopic cryoablation of small renal masses. J Endourol. 2009;23:907-911.
Marberger M, Schatzl G, Cranston D, et al. Extracorporeal ablation of renal tumors with high intensity focused ultrasound. BJU Int. 2005;95:52-55.
Marcovich R, Aldana J, Morgenstern N, et al. Histological characteristics following radiofrequency ablation of porcine kidney with and without hilar control. J Endourol. 2002;16(Suppl):23.
Marcovich R, Aldana JP, Morgenstern N, et al. Optimal lesion assessment following acute radio frequency ablation of porcine kidney: cellular viability or histopathology? J Urol. 2003;170:1370-1374.
Margulis V, Matsumoto ED, Lindberg G, et al. Acute histologic effects of temperature-based radiofrequency ablation on renal tumor pathologic interpretation. Urology. 2004;64:660-663.
Marszalek M, Meixl H, Polajnar M, et al. Laparoscopic and open partial nephrectomy: a matched-pair comparison of 200 patients. Eur Urol. 2009;55:1171-1178.
Matin SF, Ahrar K, Cadeddu JA, et al. Residual and recurrent disease following renal energy ablative therapy: a multi-institutional study. J Urol. 2006;176:1973-1977.
Matlaga BR, Zagoria RJ, Woodruff RD, et al. Phase II trial of radiofrequency ablation of renal cancer: evaluation of the kill zone. J Urol. 2002;168:2401-2405.
Matsumoto ED, Johnson DB, Ogan K, et al. Short-term efficacy of temperature-based radiofrequency ablation of small renal tumors. Urology. 2005;65:877-881.
Matsumoto ED, Watumull L, Johnson DB, et al. The radiographic evolution of radio-frequency ablated renal tumors. J Urol. 2004;172:45-48.
Mazur P. The role of intracellular freezing in the death of cells cooled at supraoptimal rates. Cryobiology. 1977;14:251-272.
McAchran SE, Lesani A, Resnick MI. Radiofrequency ablation of renal tumors: past, present, and future. Urology. 2005;66:15-22.
McDougal WS, Gervais DA, McGovern FJ, et al. Long-term followup of patients with renal cell carcinoma treated with radio frequency ablation with curative intent. J Urol. 2005;174:61-63.
McGahan JP, Dodd GD3rd. Radiofrequency ablation of the liver: current status. AJR. 2001;176:3-16.
McGahan JP, Scheider P, Brock JM, et al. Treatment of liver tumors by percutaneous radiofrequency electrocautery. Semin Intervent Radiol. 1993;10:143-149.
Miao Y, Ni Y, Mulier S, et al. Ex vivo experiment on radiofrequency liver ablation with saline infusion through a screw-tip cannulated electrode. J Surg Res. 1997;71:19-24.
Michaels MJ, Rhee HK, Mourtzinos AP, et al. Incomplete renal tumor destruction using radio frequency interstitial ablation. J Urol. 2002;168:2046-2049.
Miller DC, Saigal CS, Banerjee M, et al. Diffusion of surgical innovation among patients with kidney cancer. Cancer. 2008;112:1708-1717.
Muvner R, Threatt CB, Delvecchio FC, et al. Hypertonic saline-augmented radiofrequency ablation of the VX-2 tumor implanted in the rabbit kidney: a short-term survival pilot study. Urology. 2002;60:170-175.
Nadler RB, Kim SC, Rubenstein JN, et al. Laparoscopic renal cryosurgery: the Northwestern experience. J Urol. 2003;170:1121-1125.
Nakada SY, Jerde TJ, Warner TF, et al. Bipolar radiofrequency ablation of the kidney: comparison with monopolar radiofrequency ablation. J Endourol. 2003;17:927-933.
Neel HB3rd. Requisites for successful cryogenic surgery of cancer. Arch Surg. 1971;102:45-48.
Novick AC. Laparoscopic and partial nephrectomy. Clin Cancer Res. 2004;10:6322-6327.
Novick AC, Campbell SC, Belldegrun A, et al. Guideline for management of the clinical stage 1 renal mass. http://www.auanet.org/content/guidelines-and-quality-care/clinical-guidelines/main-reports/renalmass09.pdf, 2009. [accessed 11.02.11]
O’Malley RL, Berger AD, Kanofsky JA, et al. A matched-cohort comparison of laparoscopic cryoablation and laparoscopic partial nephrectomy for treating renal masses. BJU Int. 2006;99:395-398.
Onik G, Cooper C, Goldberg HI, et al. Ultrasonic characteristics of frozen liver. Cryobiology. 1984;21:321-328.
Onik G, Gilbert J, Hoddick W, et al. Sonographic monitoring of hepatic cryosurgery in an experimental animal model. AJR. 1985;144:1043-1047.
Organ LW. Electrophysiologic principles of radiofrequency lesion making. Appl Neurophysiol. 1976-1977;39:69-76.
Park S, Anderson JK, Matsumoto ED, et al. Radiofrequency ablation of renal tumors: intermediate-term results. J Endourol. 2006;20:569-573.
Park S, Strup SE, Saboorian H, et al. No evidence of disease after radiofrequency ablation in delayed nephrectomy specimens. Urology. 2006;68:964-967.
Pavlovich CP, Walther MM, Choyke PL, et al. Percutaneous radio frequency ablation of small renal tumors: initial results. J Urol. 2002;167:10-15.
Pennes HH. Analysis of tissue and arterial blood temperatures in the resting human forearm. J Appl Physiol. 1948;1:93-122.
Pereira PL, Trubenbach J, Schenk M, et al. Radiofrequency ablation: in vivo comparison of four commercially available devices in pig livers. Radiology. 2004;232:482-490.
Pinto PA. Renal carcinoma: minimally invasive surgery of the small renal mass. Urol Oncol. 2009;27:335-336.
Ponsky LE, Crownover RL, Rosen MJ, et al. Initial evaluation of Cyberknife technology for extracorporeal renal tissue ablation. Urology. 2003;61:498-501.
Ponsky LE, Mahadevan A, Gill IS, et al. Renal radiosurgery: initial clinical experience with histological evaluation. Surg Innov. 2007;14:265-269.
Raj GV, Reddan DJ, Hoey MF, et al. Management of small renal tumors with radiofrequency ablation. Urology. 2003;61:23-29.
Raman JD, Thomas J, Lucas SM, et al. Radiofrequency ablation for T1a tumors in a solitary kidney: promising intermediate oncologic and renal function outcomes. Can J Urol. 2008;15:3980-3985.
Rehman J, Landman J, Lee D, et al. Needle-based ablation of renal parenchyma using microwave, cryoablation, impedance and temperature based monopolar and bipolar radiofrequency, and liquid and gel chemoablation: laboratory studies and review of the literature. J Endourol. 2004;18:83-104.
Rendon RA, Gertner MR, Sherap MD, et al. Development of a radiofrequency based thermal therapy technique in an in vivo porcine model for the treatment of small renal masses. J Urol. 2001;166:292-298.
Rendon RA, Kachura JR, Sweet JM. The uncertainty of radiofrequency treatment of renal cell carcinoma: findings at immediate and delayed nephrectomy. J Urol. 2002;167:1587-1592.
Renshaw A. The natural history of incidentally detected small renal masses. Cancer. 2004;101:650-652.
Rewcastle JC, Sandison GA, Saliken JC, et al. Considerations during clinical operation of two commercially available cryomachines. J Surg Oncol. 1999;71:106-111.
Rossi S, Buscarini E, Garbagnati F, et al. Percutaneous treatment of small hepatic tumors by an expandable RF needle electrode. AJR Am J Roentgenol. 1998;170:1015-1022.
Rossi S, Garbagnati F, Lencioni R. Unresectable hepatocellular carcinoma: percutaneous radiofrequency thermal ablation after occlusion of tumor blood supply. Radiology. 2000;217:119-126.
Roy-Choudhury SH, Cast JEI, Cooksey G, et al. Early experience with radiofrequency ablation of small solid renal masses. AJR Am J Roentgenol. 2003;180:1055-1061.
Rukstalis DB, Khorsandi M, Garcia FU, et al. Clinical experience with open renal cryoablation. Urology. 2001;57:34-39.
Schenk GS, DiMarco DS, Farrell MA, et al. Image-guided radiofrequency ablation of renal tumors. J Urol. 2005;173:293.
Shepherd JP, Dawber RP. Wound healing and scarring after cryosurgery. Cryobiology. 1984;21:157-169.
Shingleton WB, Sewell PEJr. Cryoablation of renal tumours in patients with solitary kidneys. BJU Int. 2003;92:237-239.
Shingleton WB, Sewell PEJr. Percutaneous renal cryoablation: results in the first 90 patients. J Urol. 2004;171:463.
Steed J, Saliken JC, Donnelly BJ, et al. Correlation between thermosensor temperature and transrectal ultrasonography during prostate cryoablation. Can Assoc Radiol J. 1997;48:186-190.
Stern JM, Gupta A, Raman JD, et al. Radiofrequency ablation of small renal cortical tumours in healthy adults: renal function preservation and intermediate oncological outcome. BJU Int. 2009;104:786-789.
Stern J, Raman JD, Cadeddu JA. Absence of viable carcinoma in biopsies performed greater than 1-year following radiofrequency ablation of renal cortical tumors. J Urol. 2008;179:413.
Su LM, Jarrett TW, Chan DY, et al. Percutaneous computed tomography-guided radiofrequency ablation of renal masses in high surgical risk patients: preliminary results. Urology. 2003;61:26-33.
Sung GT, Gill IS, Hsu TH, et al. Effect of intentional cryo-injury to the renal collecting system. J Urol. 2003;170:619-622.
Svedman C, Sandström P, Pisa P, et al. A prospective Phase II trial of using extracranial stereotactic radiotherapy in primary and metastatic renal cell carcinoma. Acta Oncol. 2006;45:870-875.
Tatsutani K, Rubinsky B, Onik G, et al. Effect of thermal variables on frozen human primary prostatic adenocarcinoma cells. Urology. 1996;48:441-447.
Thomas AA, Aron M, Hernandez AV, et al. Laparoscopic partial nephrectomy in octogenarians. Urology. 2009;74(5):1042-1046.
Thompson RH, Boorjian SA, Lohse CM, et al. Radical nephrectomy for pT1a renal masses may be associated with decreased overall survival compared to partial nephrectomy. J Urol. 2008;179:468-473.
Thompson RH, Frank I, Lohse CM, et al. The impact of ischemia time during open nephron sparing surgery on solitary kidneys: a multi-institutional study. J Urol. 2007;177:471-476.
Vallancien G, Chartier-Kastler E, Bataille N, et al. Focused extracorporeal pyrotherapy. Eur Urol. 1993;23:48-52.
Varkarakis IM, Allaf ME, Inagaki T, et al. Percutaneous radio frequency ablation of renal masses: results at a 2-year mean followup. J Urol. 2005;174:456-460.
Veltri A, De Fazio G, Malfitana V, et al. Percutaneous US-guided RF thermal ablation for malignant renal tumors: preliminary results in 13 patients. Eur Radiol. 2004;14:2303-2310.
Volpe A, Panzarella T, Rendon RA, et al. The natural history of incidentally detected small renal masses. Cancer. 2004;15:738-745.
Vricella GJ, Ponsky LE, Cadeddu JA. Ablative technologies for urologic cancers. Urol Clin N Am. 2009;36:163-178.
Wang R, Wood DPJr. Evolving role of renal biopsy in small renal masses. Urol Oncol. 2009;27:332-334.
Weber SM, Lee FTJr. Cryoablation: history, mechanism of action, and guidance modalities. In: van Sonnenberg E, McMullen W, Solbiati L, editors. Tumor ablation: principles and practice. New York: Springer; 2005:250-265.
Weber SM, Lee FT, Chinn DO, et al. Perivascular and intralesional tissue necrosis after hepatic cryoablation: results in a porcine model. Surgery. 1997;122:742-747.
Weber SM, Lee FT, Warner TF, et al. Hepatic cryoablation: US monitoring of extent of necrosis in normal pig liver. Radiology. 1998;207:73-77.
Weight CJ, Kaouk JH, Hegarty NJ, et al. Correlation of radiographic imaging and histopathology following cryoablation and radio frequency ablation for renal tumors. J Urol. 2008;179:1277-1281.
Weld KJ, Figenshau RS, Venkatesh R, et al. Laparoscopic cryoablation for small renal masses: three-year follow-up. Urology. 2007;69:448-451.
White WM, Haber GP, Goel RK, et al. Single-port urological surgery: single-center experience with the first 100 cases. Urology. 2009;74(4):801-804.
Williams JC, Morrison PM, Swishchuck PN, et al. Laser induced thermotherapy of renal cell carcinoma in mandosimetry, ultrasound and histopathologic correlation. J Urol. 2000;163:9.
Wong WS, Chinn DO, Chinn M, et al. Cryosurgery as a treatment for prostate carcinoma: results and complications. Cancer. 1997;79:963-974.
Woolley ML, Schulsinger DA, Durand DB, et al. Effect of freezing parameters (freeze cycle and thaw process) on tissue destruction following renal cryoablation. J Endourol. 2002;16:519-522.
Yamakado K, Nakatsura A, Kobayashi S, et al. Radiofrequency ablation combined with renal arterial embolization for the treatment of unresectable renal cell carcinoma larger than 3.5 cm: initial experience. Cardiovasc Intervent Radiol. 2006;29:389-394.
Zagoria RJ, Traver MA, Werle DM, et al. Oncologic efficacy of CT-guided percutaneous radiofrequency ablation of renal cell carcinomas. AJR. 2007;189:429-436.
Zevas NT, Kuwayama A. Pathological characteristics of experimental thermal lesions: comparison of induction heating and radiofrequency electrocoagulation. J Neurosurg. 1972;37:418-422.
Zini L, Perrotte P, Capitano U, et al. Radical versus partial nephrectomy. Cancer. 2009;115:1465-1471.