4 Abdomen
Conceptual overview
GENERAL DESCRIPTION
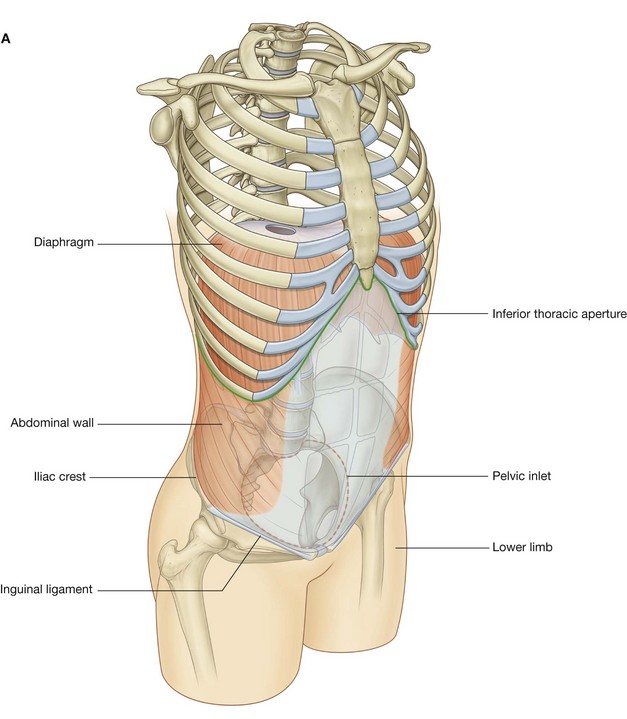
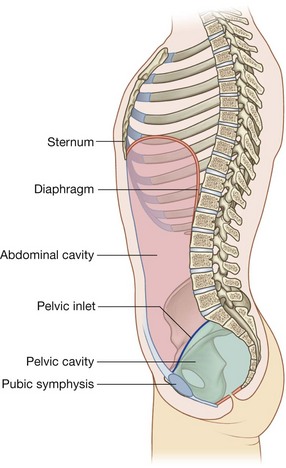
The abdomen is a roughly cylindrical chamber extending from the inferior margin of the thorax to the superior margin of the pelvis and the lower limb (Fig. 4.1A).
The inferior thoracic aperture forms the superior opening to the abdomen, and is closed by the diaphragm. Inferiorly, the deep abdominal wall is continuous with the pelvic wall at the pelvic inlet. Superficially, the inferior limit of the abdominal wall is the superior margin of the lower limb.
The chamber enclosed by the abdominal wall contains a single large peritoneal cavity, which freely communicates with the pelvic cavity.
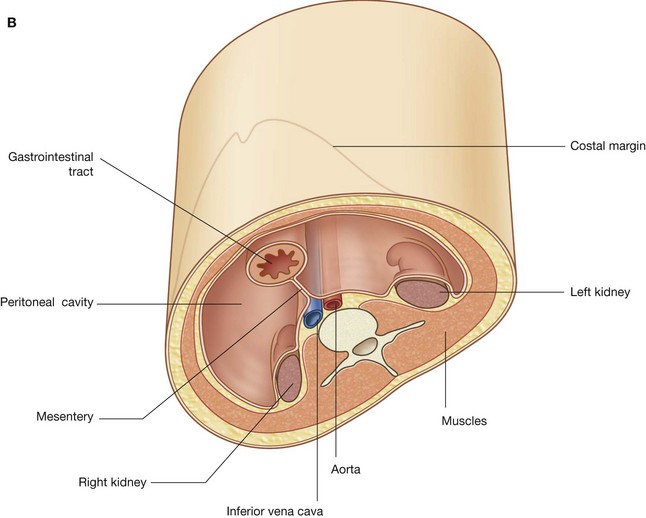
Abdominal viscera are either suspended in the peritoneal cavity by mesenteries or positioned between the cavity and the musculoskeletal wall (Fig. 4.1B).
FUNCTIONS
Houses and protects major viscera
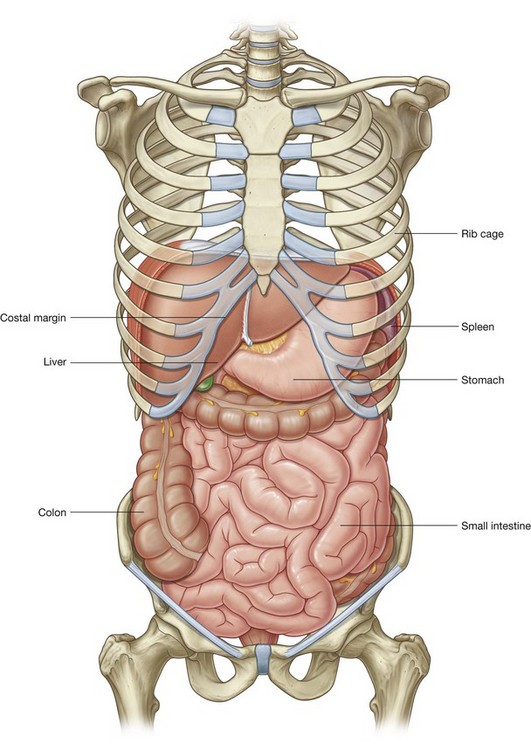
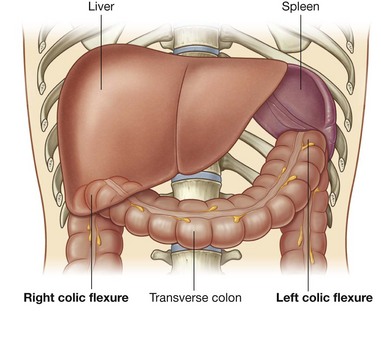
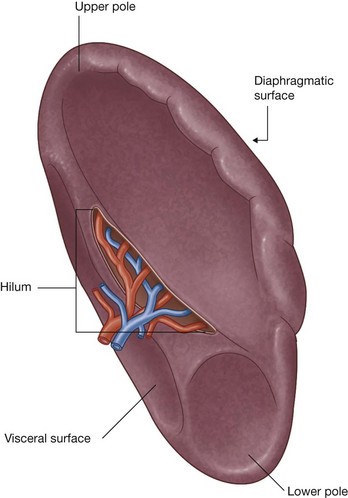
The abdomen houses major elements of the gastrointestinal system (Fig. 4.2), the spleen, and parts of the urinary system.
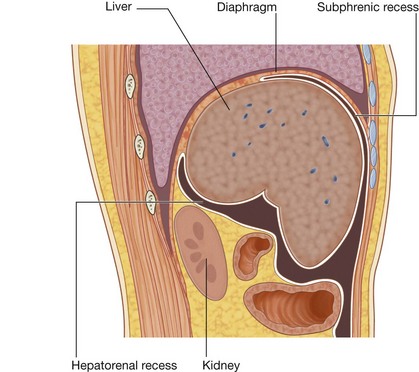
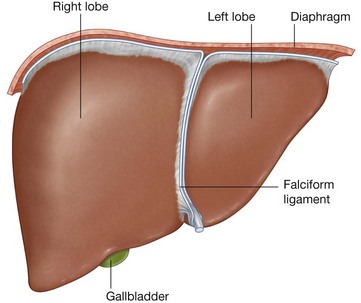
Much of the liver, gallbladder, stomach, and spleen, and parts of the colon are under the domes of the diaphragm, which project superiorly above the costal margin of the thoracic wall, and as a result these abdominal viscera are protected by the thoracic wall. The superior poles of the kidneys are deep to the lower ribs.
Viscera not under the domes of the diaphragm are supported and protected predominantly by the muscular walls of the abdomen.
Breathing
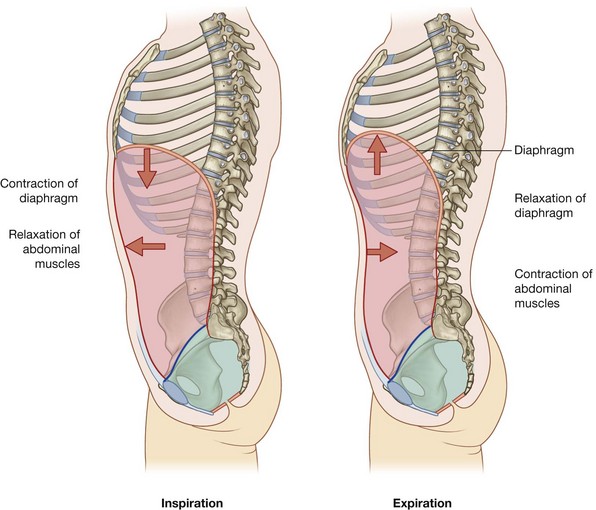
One of the most important roles of the abdominal wall is to assist in breathing:
 it relaxes during inspiration to accommodate expansion of the thoracic cavity and the inferior displacement of abdominal viscera during contraction of the diaphragm (Fig. 4.3);
it relaxes during inspiration to accommodate expansion of the thoracic cavity and the inferior displacement of abdominal viscera during contraction of the diaphragm (Fig. 4.3);Changes in intra-abdominal pressure
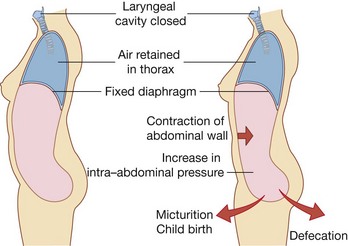
Contraction of abdominal wall muscles can dramatically increase intra-abdominal pressure when the diaphragm is in a fixed position (Fig. 4.4). Air is retained in the lungs by closing valves in the larynx in the neck. Increased intra-abdominal pressure assists in voiding the contents of the bladder and rectum and in giving birth.
COMPONENT PARTS
Wall
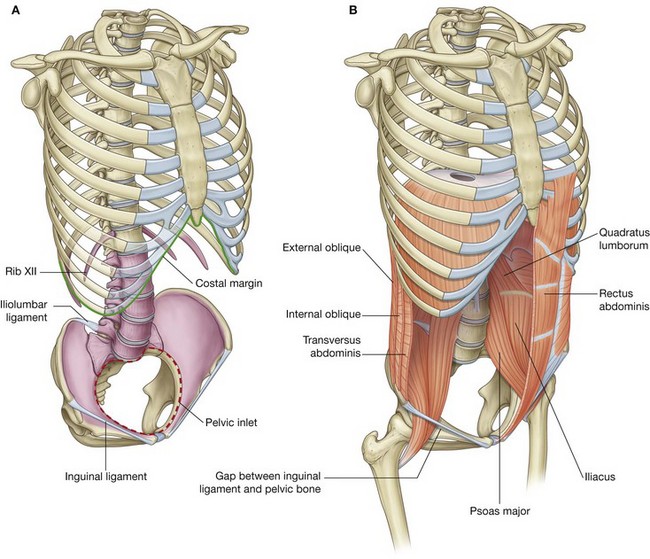
The abdominal wall consists partly of bone but mainly of muscle (Fig. 4.5). The skeletal elements of the wall (Fig. 4.5A) are:
 bony components of the inferior thoracic wall including the costal margin, rib XII, the end of rib XI, and the xiphoid process.
bony components of the inferior thoracic wall including the costal margin, rib XII, the end of rib XI, and the xiphoid process.Muscles make up the rest of the abdominal wall (Fig. 4.5B):
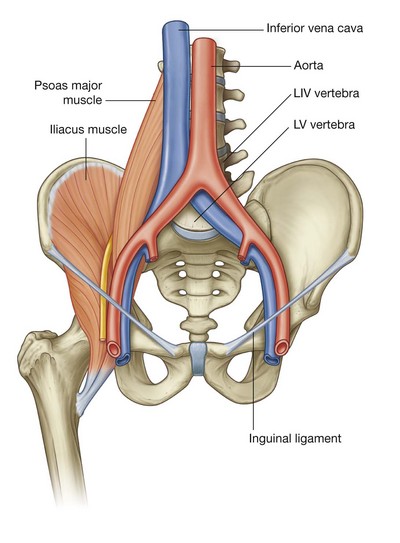
 lateral to the vertebral column, the quadratus lumborum, psoas major, and iliacus muscles reinforce the posterior aspect of the wall. The distal ends of the psoas major and iliacus muscles pass into the thigh and are major flexors of the hip joint;
lateral to the vertebral column, the quadratus lumborum, psoas major, and iliacus muscles reinforce the posterior aspect of the wall. The distal ends of the psoas major and iliacus muscles pass into the thigh and are major flexors of the hip joint; lateral parts of the abdominal wall are predominantly formed by three layers of muscles, which are similar in orientation to the intercostal muscles of the thorax—transversus abdominis, internal oblique, and external oblique;
lateral parts of the abdominal wall are predominantly formed by three layers of muscles, which are similar in orientation to the intercostal muscles of the thorax—transversus abdominis, internal oblique, and external oblique; anteriorly, a segmented muscle (the rectus abdominis) on each side spans the distance between the inferior thoracic wall and the pelvis.
anteriorly, a segmented muscle (the rectus abdominis) on each side spans the distance between the inferior thoracic wall and the pelvis.Structural continuity between posterior, lateral, and anterior parts of the abdominal wall is provided by thick fascia posteriorly and by flat tendinous sheets (aponeuroses) derived from muscles of the lateral wall. A fascial layer of varying thickness separates the abdominal wall from the peritoneum, which lines the abdominal cavity.
Abdominal cavity
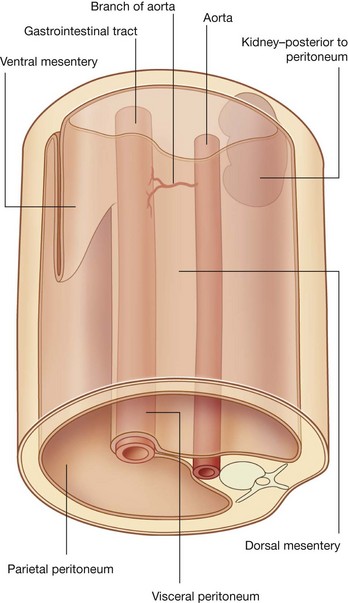
The general organization of the abdominal cavity is one in which a central gut tube (gastrointestinal system) is suspended from the posterior abdominal wall and partly from the anterior abdominal wall by thin sheets of tissue (mesenteries; Fig. 4.6):
Different parts of these two mesenteries are named according to the organs they suspend or with which they are associated.
Major viscera, such as the kidneys, that are not suspended in the abdominal cavity by mesenteries are associated with the abdominal wall.
The abdominal cavity is lined by peritoneum, which consists of an epithelial-like single layer of cells (the mesothelium) together with a supportive layer of connective tissue. Peritoneum is similar to the pleura and serous pericardium in the thorax.
The peritoneum reflects off the abdominal wall to become a component of the mesenteries that suspend the viscera.
Normally, elements of the gastrointestinal tract and its derivatives completely fill the abdominal cavity, making the peritoneal cavity a potential space, and visceral peritoneum on organs and parietal peritoneum on the adjacent abdominal wall slide freely against one another.
Abdominal viscera are either intraperitoneal or retroperitoneal:
 intraperitoneal structures, such as elements of the gastrointestinal system, are suspended from the abdominal wall by mesenteries;
intraperitoneal structures, such as elements of the gastrointestinal system, are suspended from the abdominal wall by mesenteries; structures that are not suspended in the abdominal cavity by a mesentery and that lie between the parietal peritoneum and abdominal wall are retroperitoneal in position.
structures that are not suspended in the abdominal cavity by a mesentery and that lie between the parietal peritoneum and abdominal wall are retroperitoneal in position.Retroperitoneal structures include the kidneys and ureters, which develop in the region between the peritoneum and the abdominal wall and remain in this position in the adult.
During development, some organs, such as parts of the small and large intestines, are suspended initially in the abdominal cavity by a mesentery, and later become retroperitoneal secondarily by fusing with the abdominal wall (Fig. 4.7).
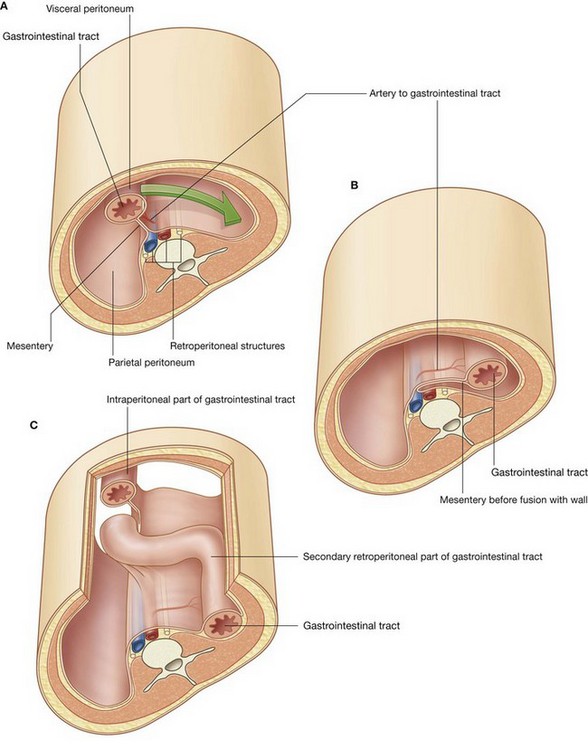
Fig. 4.7 A series showing the progression (A to C) from an intraperitoneal organ to a secondarily retroperitoneal organ.
Large vessels, nerves, and lymphatics are associated with the posterior abdominal wall along the median axis of the body in the region where, during development, the peritoneum reflects off the wall as the dorsal mesentery, which supports the developing gut tube. As a consequence, branches of the neurovascular structures that pass to parts of the gastrointestinal system are unpaired, originate from the anterior aspects of their parent structures, and travel in mesenteries or pass retroperitoneally in areas where the mesenteries secondarily fuse to the wall.
Generally, vessels, nerves, and lymphatics to the abdominal wall and to organs that originate as retroperitoneal structures branch laterally from the central neurovascular structures and are usually paired, one on each side.
Inferior thoracic aperture
The superior aperture of the abdomen is the inferior thoracic aperture, which is closed by the diaphragm (see pp. 126–127). The margin of the inferior thoracic aperture consists of vertebra TXII, rib XII, the distal end of rib XI, the costal margin, and the xiphoid process of the sternum.
Diaphragm
The musculotendinous diaphragm separates the abdomen from the thorax.
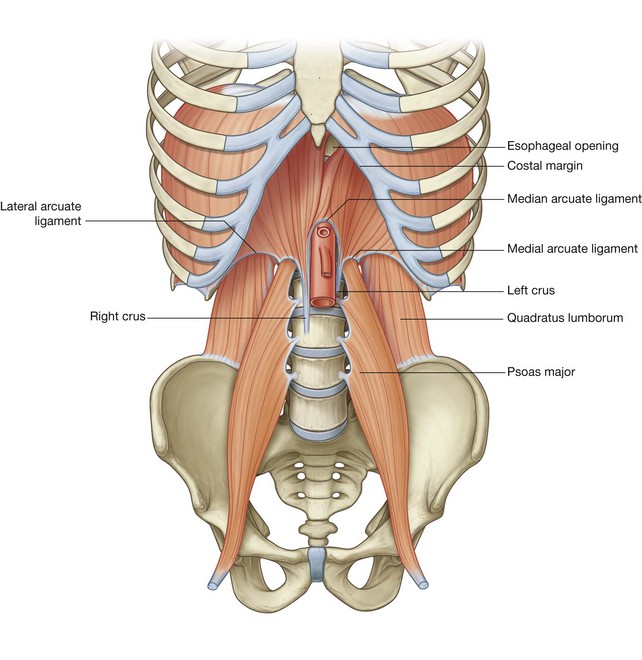
The diaphragm attaches to the margin of the inferior thoracic aperture, but the attachment is complex posteriorly and extends into the lumbar area of the vertebral column (Fig. 4.8). On each side, a muscular extension (crus) firmly anchors the diaphragm to the anterolateral surface of the vertebral column as far down as vertebra LIII on the right and vertebra LII on the left.
Because the costal margin is not complete posteriorly, the diaphragm is anchored to arch-shaped (arcuate) ligaments, which span the distance between available bony points and the intervening soft tissues:
 medial and lateral arcuate ligaments cross muscles of the posterior abdominal wall and attach to vertebrae, the transverse processes of vertebra LI and rib XII, respectively; and
medial and lateral arcuate ligaments cross muscles of the posterior abdominal wall and attach to vertebrae, the transverse processes of vertebra LI and rib XII, respectively; andThe posterior attachment of the diaphragm extends much farther inferiorly than the anterior attachment. Consequently, the diaphragm is an important component of the posterior abdominal wall, to which a number of viscera are related.
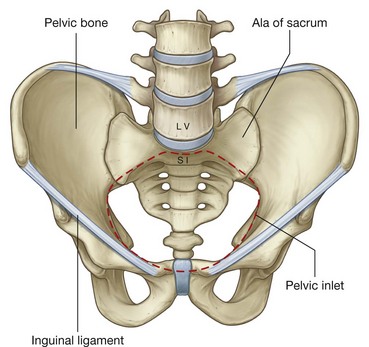
Pelvic inlet
The abdominal wall is continuous with the pelvic wall at the pelvic inlet, and the abdominal cavity is continuous with the pelvic cavity.
The circular margin of the pelvic inlet is formed entirely by bone:
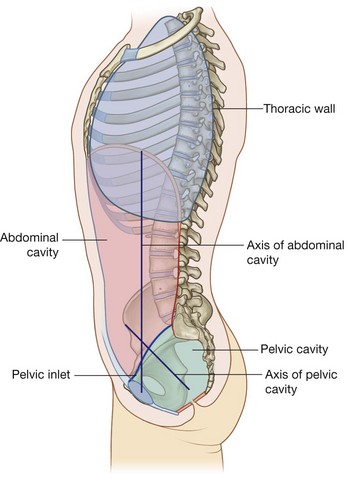
Because of the way in which the sacrum and attached pelvic bones are angled posteriorly on the vertebral column, the pelvic cavity is not oriented in the same vertical plane as the abdominal cavity. Instead, the pelvic cavity projects posteriorly, and the inlet opens anteriorly and somewhat superiorly (Fig. 4.10).
RELATIONSHIP TO OTHER REGIONS
Thorax
The abdomen is separated from the thorax by the diaphragm. Structures pass between the two regions through or posterior to the diaphragm (see Fig. 4.8).
Pelvis
The pelvic inlet opens directly into the abdomen and structures pass between the abdomen and pelvis through it.
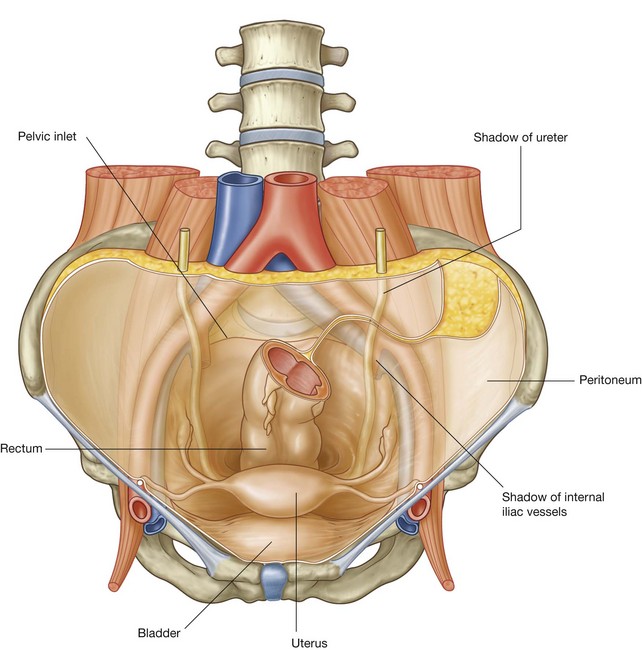
The peritoneum lining the abdominal cavity is continuous with the peritoneum in the pelvis. Consequently, the abdominal cavity is entirely continuous with the pelvic cavity (Fig. 4.11). Infections in one region can therefore freely spread into the other.
The bladder expands superiorly from the pelvic cavity into the abdominal cavity and, during pregnancy, the uterus expands freely superiorly out of the pelvic cavity into the abdominal cavity.
Lower limb
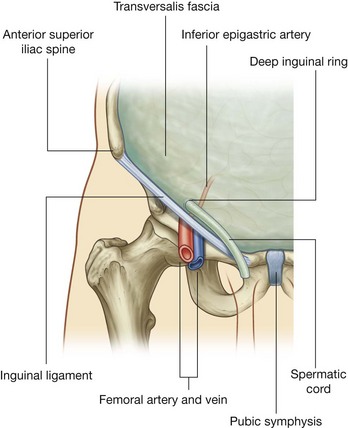
The abdomen communicates directly with the thigh through an aperture formed anteriorly between the inferior margin of the abdominal wall (marked by the inguinal ligament) and the pelvic bone (Fig. 4.12). Structures that pass through this aperture are:
As vessels pass inferior to the inguinal ligament, their names change—the external iliac artery and vein of the abdomen become the femoral artery and vein of the thigh.
KEY FEATURES
Arrangement of abdominal viscera in the adult
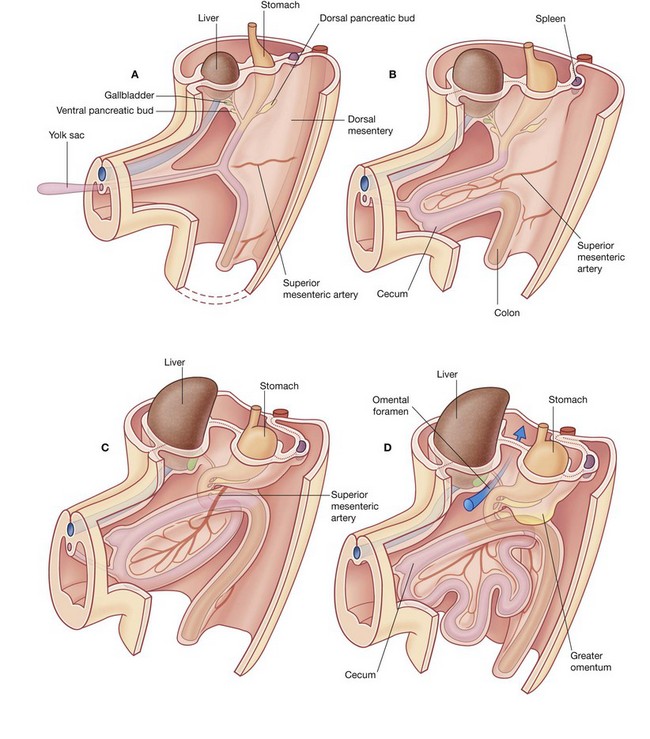
A basic knowledge of the development of the gastrointestinal tract is needed to understand the arrangement of viscera and mesenteries in the abdomen (Fig. 4.13).
The early gastrointestinal tract is oriented longitudinally in the body cavity and is suspended from surrounding walls by a large dorsal mesentery and a much smaller ventral mesentery.
Superiorly, the dorsal and ventral mesenteries are anchored to the diaphragm.
The primitive gut tube consists of the foregut, the midgut, and the hindgut. Massive longitudinal growth of the gut tube, rotation of selected parts of the tube, and secondary fusion of some viscera and their associated mesenteries to the body wall participate in generating the adult arrangement of abdominal organs.
Development of the foregut
In abdominal regions, the foregut gives rise to the distal end of the esophagus, the stomach, and the proximal part of the duodenum. The foregut is the only part of the gut tube suspended from the wall by both the ventral and dorsal mesenteries.
A diverticulum from the anterior aspect of the foregut grows into the ventral mesentery, giving rise to the liver and gallbladder, and, ultimately, to the ventral part of the pancreas.
The dorsal part of the pancreas develops from an outgrowth of the foregut into the dorsal mesentery. The spleen develops in the dorsal mesentery in the region between the body wall and presumptive stomach.
In the foregut, the developing stomach rotates clockwise and the associated dorsal mesentery, containing the spleen, moves to the left and greatly expands. During this process, part of the mesentery becomes associated with, and secondarily fuses with, the left side of the body wall.
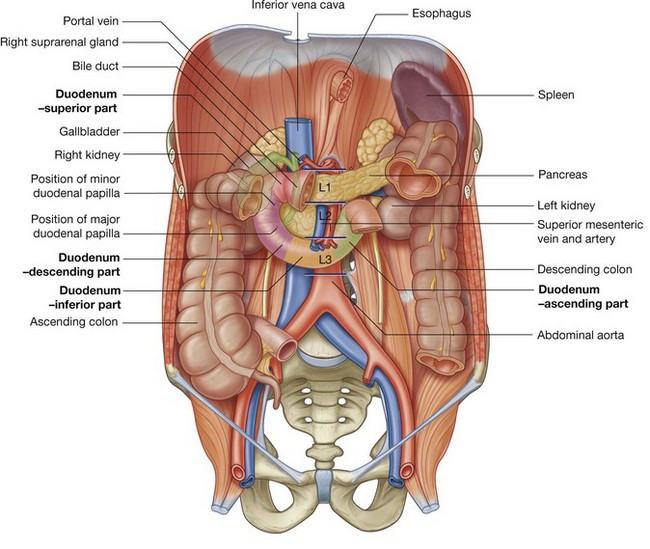
At the same time, the duodenum, together with its dorsal mesentery and an appreciable part of the pancreas, swings to the right and fuses to the body wall.
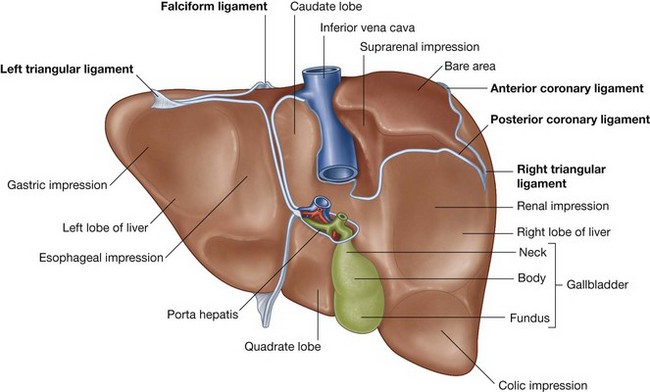
Secondary fusion of the duodenum to the body wall, massive growth of the liver in the ventral mesentery, and fusion of the superior surface of the liver to the diaphragm restrict the opening to the space enclosed by the ballooned dorsal mesentery associated with the stomach. This restricted opening is the omental foramen (epiploic foramen).
The part of the abdominal cavity enclosed by the expanded dorsal mesentery, and posterior to the stomach, is the omental bursa (lesser sac). Access, through the omental foramen, to this space from the rest of the peritoneal cavity (greater sac) is inferior to the free edge of the ventral mesentery.
Part of the dorsal mesentery that initially forms part of the lesser sac greatly enlarges in an inferior direction, and the two opposing surfaces of the mesentery fuse to form an apron-like structure (the greater omentum). The greater omentum is suspended from the greater curvature of the stomach, lies over other viscera in the abdominal cavity, and is the first structure observed when the abdominal cavity is opened anteriorly.
Development of the midgut
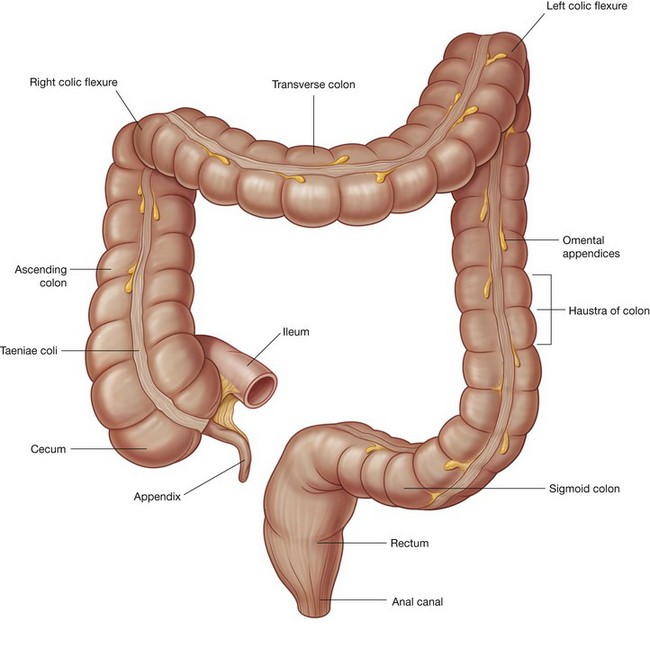
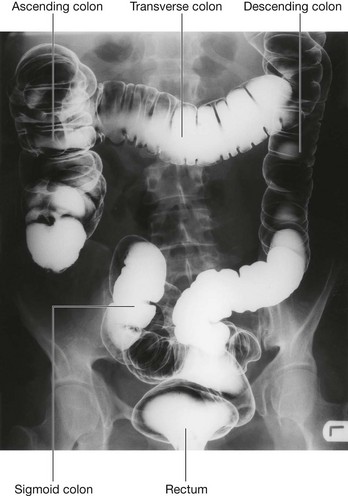
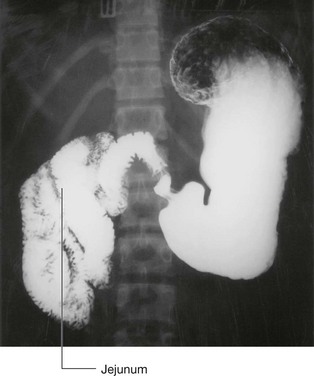
The midgut develops into the distal part of the duodenum, the jejunum, ileum, ascending colon, and proximal two-thirds of the transverse colon. A small yolk sac projects anteriorly from the developing midgut into the umbilicus.
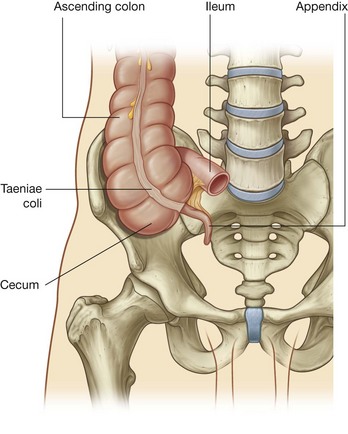
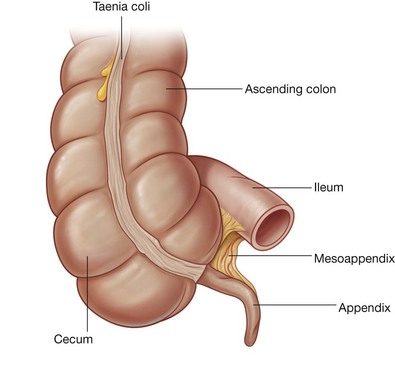
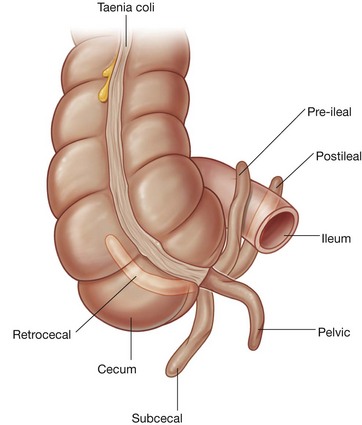
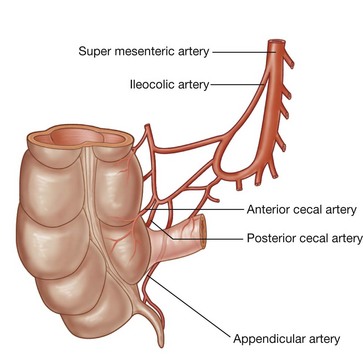
Rapid growth of the gastrointestinal system results in a loop of the midgut herniating out of the abdominal cavity and into the umbilical cord. As the body grows in size and the connection with the yolk sac is lost, the midgut returns to the abdominal cavity. While this process is occurring, the two limbs of the midgut loop rotate counterclockwise around their combined central axis, and the part of the loop that becomes the cecum descends into the inferior right aspect of the cavity. The superior mesenteric artery, which supplies the midgut, is at the center of the axis of rotation.
The cecum remains intraperitoneal, the ascending colon fuses with the body wall becoming secondarily retroperitoneal, and the transverse colon remains suspended by its dorsal mesentery (transverse mesocolon). The greater omentum hangs over the transverse colon and the mesocolon and usually fuses with these structures.
Development of the hindgut
The distal one-third of the transverse colon, descending colon, sigmoid colon, and the superior part of rectum develop from the hindgut.
Proximal parts of the hindgut swing to the left and become the descending colon and sigmoid colon. The descending colon and its dorsal mesentery fuse to the body wall, while the sigmoid colon remains intraperitoneal. The sigmoid colon passes through the pelvic inlet and is continuous with the rectum at the level of vertebra SIII.
Skin and muscles of the anterior and lateral abdominal wall and thoracic intercostal nerves
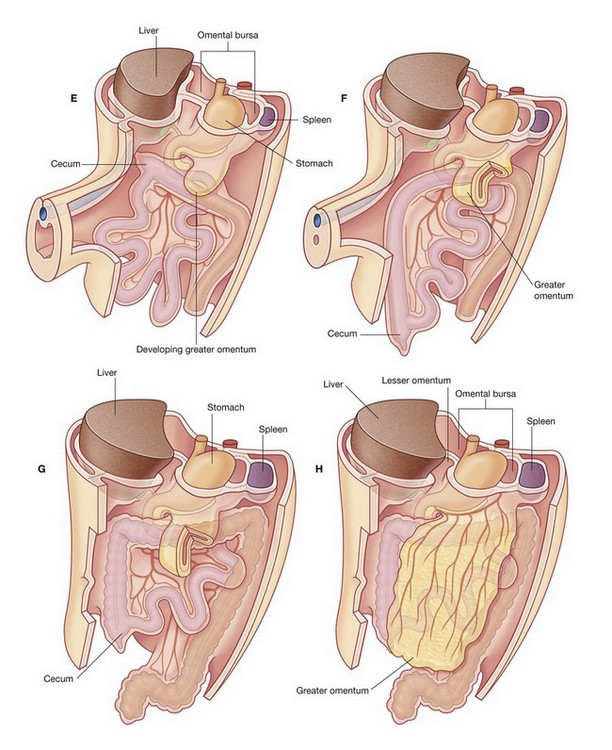
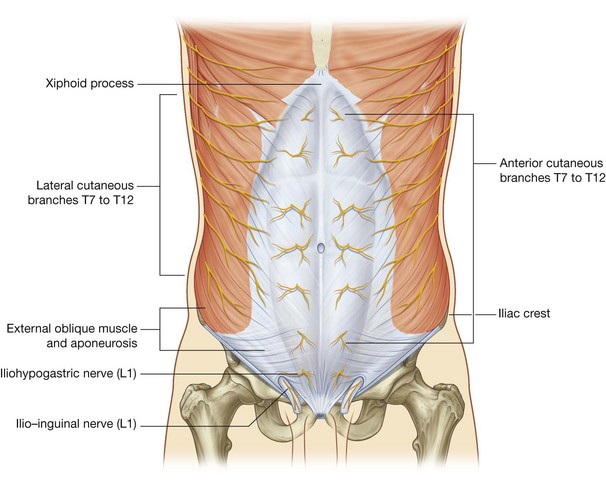
The anterior rami of thoracic spinal nerves T7 to T12 follow the inferior slope of the lateral parts of the ribs and cross the costal margin to enter the abdominal wall (Fig. 4.14). Intercostal nerves T7 to T11 supply skin and muscle of the abdominal wall, as does the subcostal nerve T12. In addition, T5 and T6 supply upper parts of the external oblique muscle of the abdominal wall; T6 also supplies cutaneous innervation to skin over the xiphoid.
Skin and muscle in the inguinal and suprapubic regions of the abdominal wall are innervated by L1 and not by thoracic nerves.
Dermatomes of the anterior abdominal wall are indicated in Figure 4.14. In the midline, skin over the infrasternal angle is T6 and that around the umbilicus is T10. L1 innervates skin in the inguinal and suprapubic regions.
Muscles of the abdominal wall are innervated segmentally in patterns that generally reflect the patterns of the overlying dermatomes.
The groin is a weak area in the anterior abdominal wall
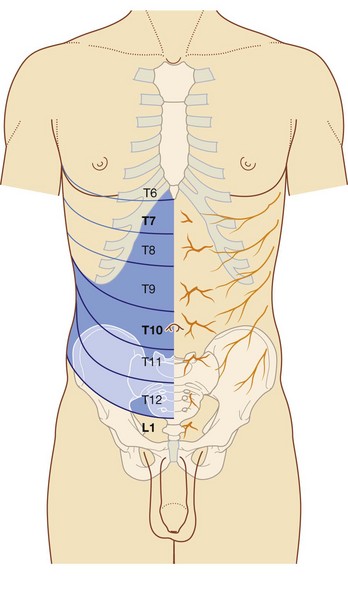
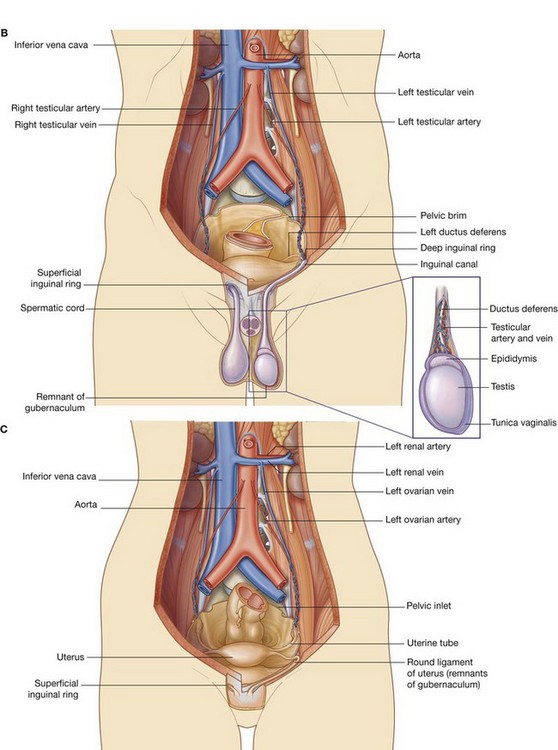
During development, the gonads in both sexes descend from their sites of origin on the posterior abdominal wall into the pelvic cavity in women and the developing scrotum in men (Fig. 4.15).
Before descent, a cord of tissue (the gubernaculum) passes through the anterior abdominal wall and connects the inferior pole of each gonad with primordia of the scrotum in men and the labia majora in women (labioscrotal swellings).
A tubular extension (the processus vaginalis) of the peritoneal cavity and the accompanying muscular layers of the anterior abdominal wall project along the gubernaculum on each side into the labioscrotal swellings.
In men, the testis, together with its neurovascular structures and its efferent duct (the ductus deferens) descends into the scrotum along a path, initially defined by the gubernaculum, between the processus vaginalis and the accompanying coverings derived from the abdominal wall. All that remains of the gubernaculum is a connective tissue remnant that attaches the caudal pole of the testis to the scrotum.
The inguinal canal is the passage through the anterior abdominal wall created by the processus vaginalis. The spermatic cord is the tubular extension of the layers of the abdominal wall into the scrotum that contains all structures passing between the testis and the abdomen.
The distal sac-like terminal end of the spermatic cord on each side contains the testis, associated structures, and the now isolated part of the peritoneal cavity (the cavity of the tunica vaginalis).
In women, the gonads descend to a position just inside the pelvic cavity and never pass through the anterior abdominal wall. As a result, the only major structure passing through the inguinal canal is a derivative of the gubernaculum (the round ligament of uterus).
In both men and women, the groin (inguinal region) is a weak area in the abdominal wall (Fig. 4.15) and is the site of inguinal hernias.
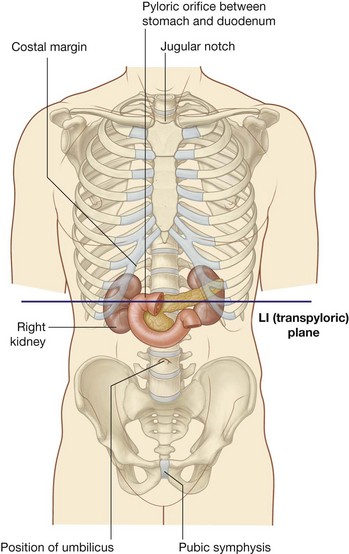
Vertebral level LI
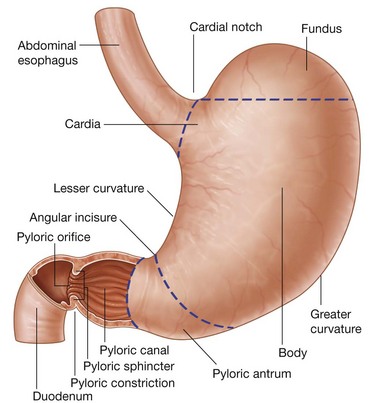
The transpyloric plane is a horizontal plane that transects the body through the lower aspect of vertebra LI (Fig. 4.16). It:
 is about midway between the jugular notch and the pubic symphysis, and crosses the costal margin on each side at roughly the ninth costal cartilage;
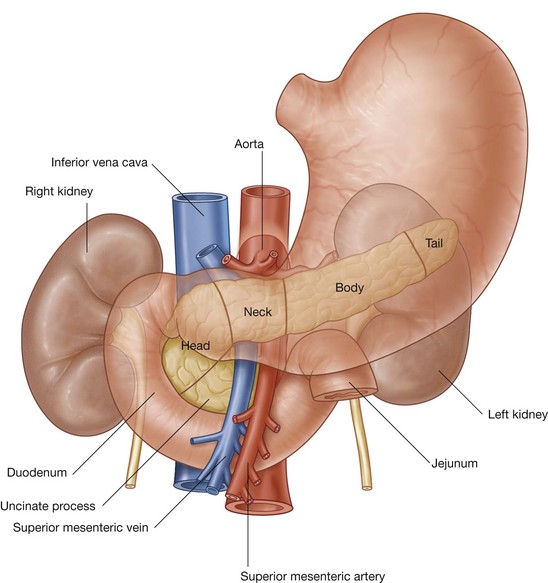
is about midway between the jugular notch and the pubic symphysis, and crosses the costal margin on each side at roughly the ninth costal cartilage; crosses through the opening of the stomach into the duodenum (the pyloric orifice), which is just to the right of the body of LI; the duodenum then makes a characteristic C-shaped loop on the posterior abdominal wall and crosses the midline to open into the jejunum just to the left of the body of vertebra LII, whereas the head of the pancreas is enclosed by the loop of the duodenum, and the body of the pancreas extends across the midline to the left;
crosses through the opening of the stomach into the duodenum (the pyloric orifice), which is just to the right of the body of LI; the duodenum then makes a characteristic C-shaped loop on the posterior abdominal wall and crosses the midline to open into the jejunum just to the left of the body of vertebra LII, whereas the head of the pancreas is enclosed by the loop of the duodenum, and the body of the pancreas extends across the midline to the left;The gastrointestinal system and its derivatives are supplied by three major arteries
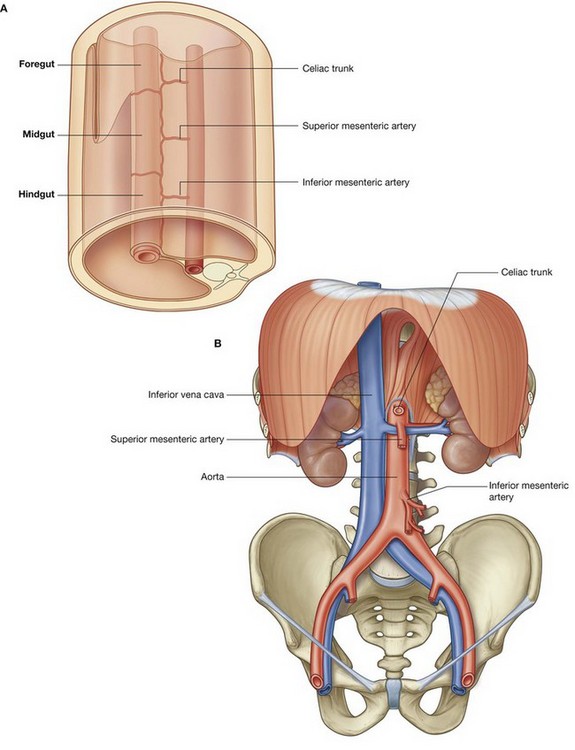
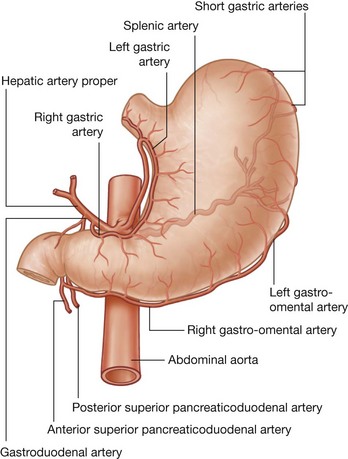
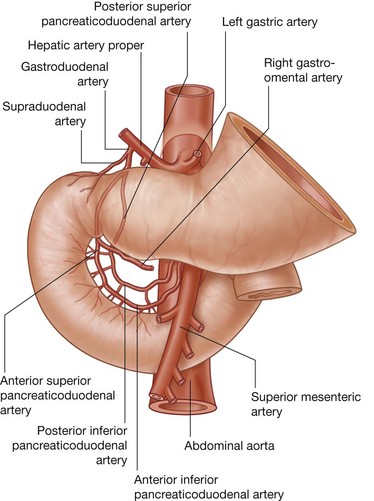
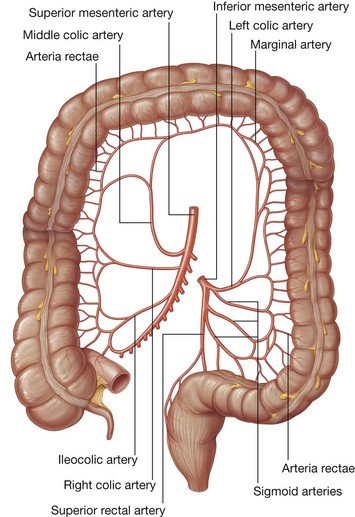
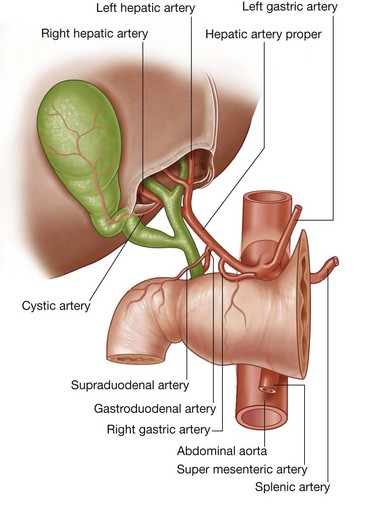
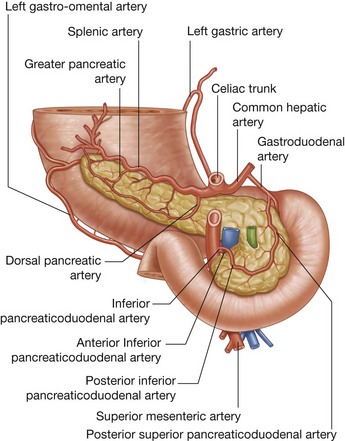
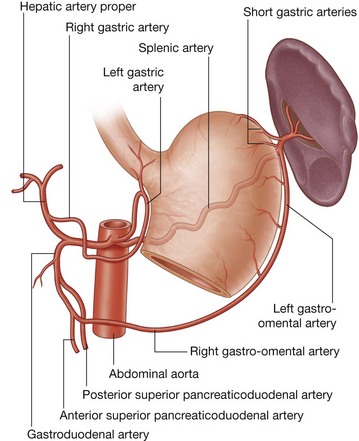
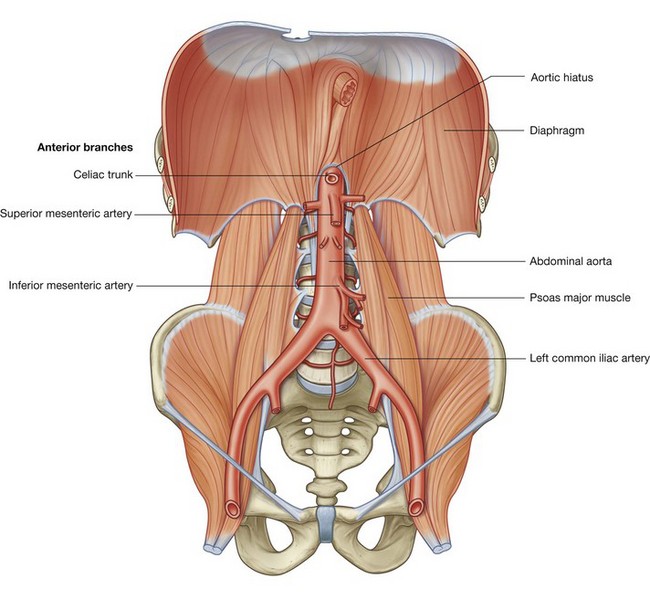
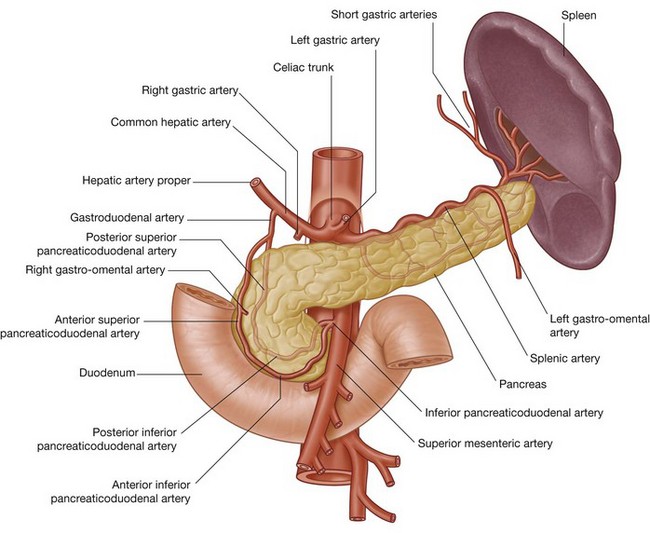
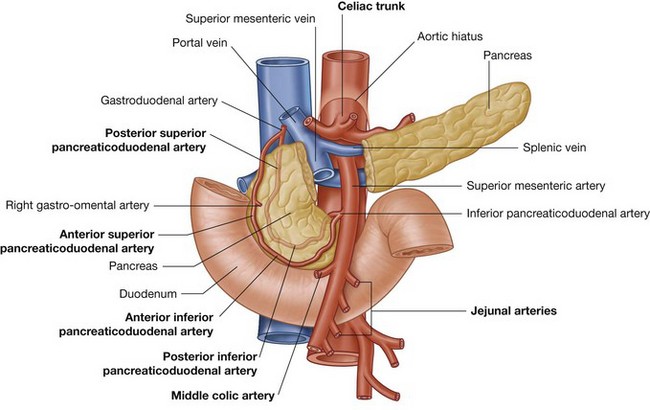
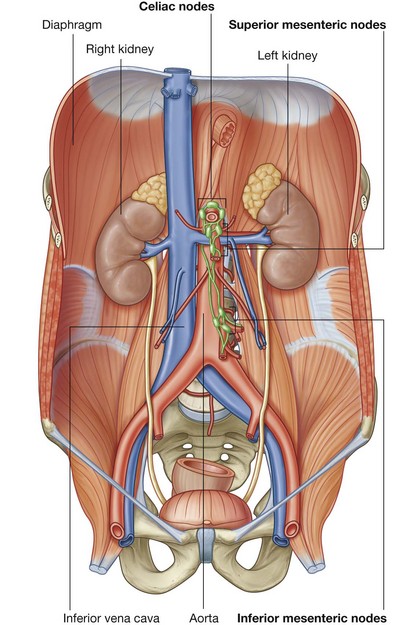
Three large unpaired arteries branch from the anterior surface of the abdominal aorta to supply the abdominal part of the gastrointestinal tract and all of the structures (liver, pancreas, and gallbladder) to which this part of the gut gives rise to during development (Fig. 4.17). These arteries pass through derivatives of the dorsal and ventral mesenteries to reach the target viscera. These vessels therefore also supply structures such as the spleen and lymph nodes that develop in the mesenteries. These three arteries are:
 the celiac artery, which branches from the abdominal aorta at the upper border of vertebra LI and supplies the foregut;
the celiac artery, which branches from the abdominal aorta at the upper border of vertebra LI and supplies the foregut; the superior mesenteric artery, which arises from the abdominal aorta at the lower border of vertebra LI and supplies the midgut; and
the superior mesenteric artery, which arises from the abdominal aorta at the lower border of vertebra LI and supplies the midgut; andVenous shunts from left to right
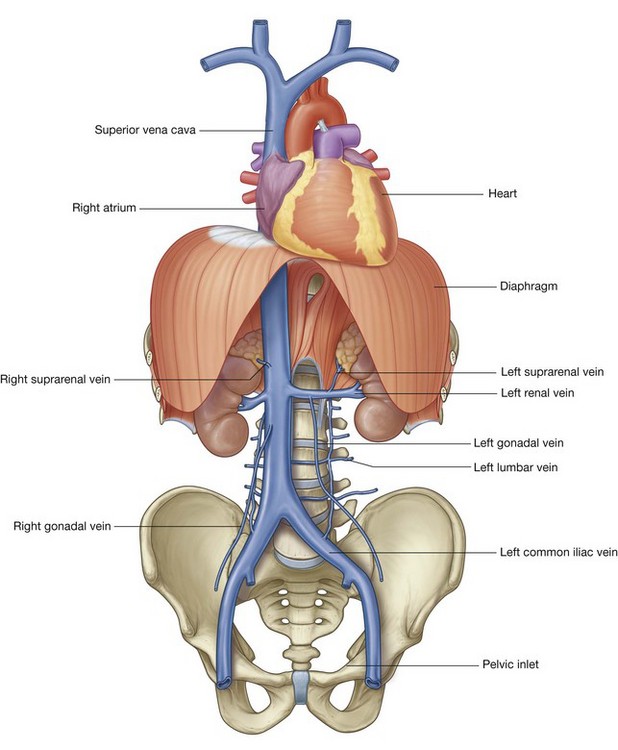
All blood returning to the heart from regions of the body other than the lungs flows into the right atrium of the heart. The inferior vena cava is the major systemic vein in the abdomen and drains this region together with the pelvis, perineum, and both lower limbs (Fig. 4.18).
The inferior vena cava lies to the right of the vertebral column and penetrates the central tendon of the diaphragm at approximately vertebral level TVIII. A number of large vessels cross the midline to deliver blood from the left side of the body to the inferior vena cava.
 One of the most significant is the left renal vein, which drains the kidney, suprarenal gland, and gonad on the same side.
One of the most significant is the left renal vein, which drains the kidney, suprarenal gland, and gonad on the same side.All venous drainage from the gastrointestinal system passes through the liver
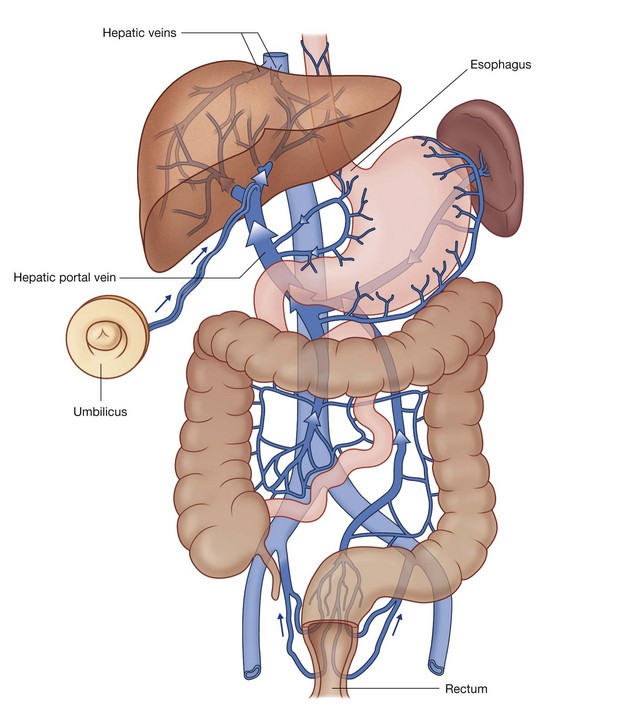
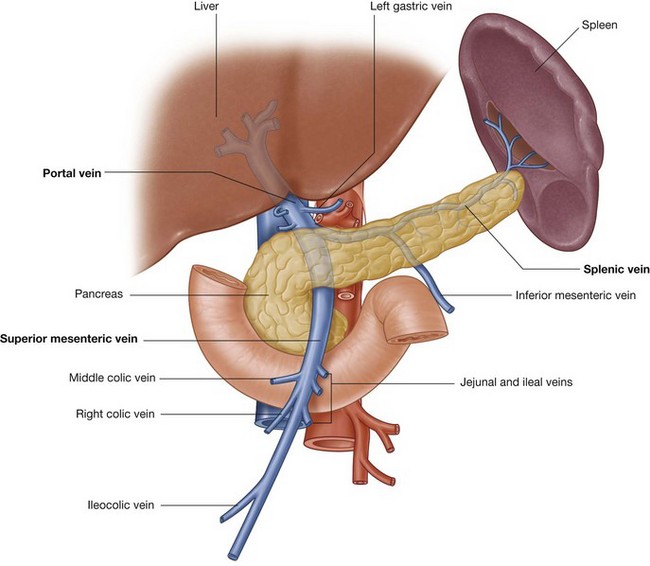
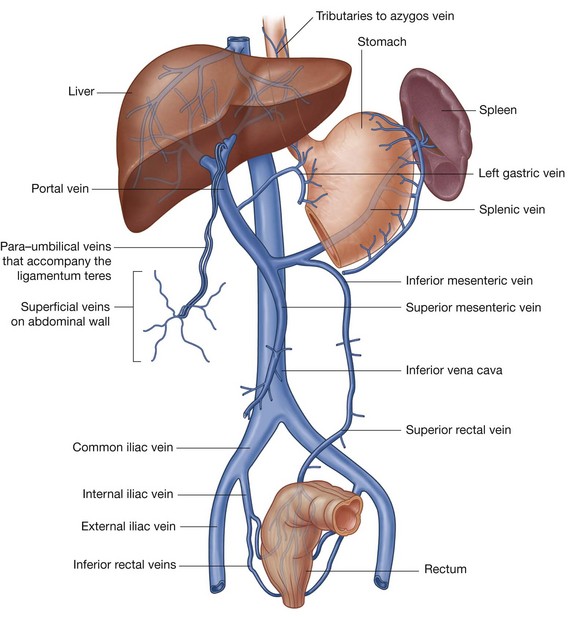
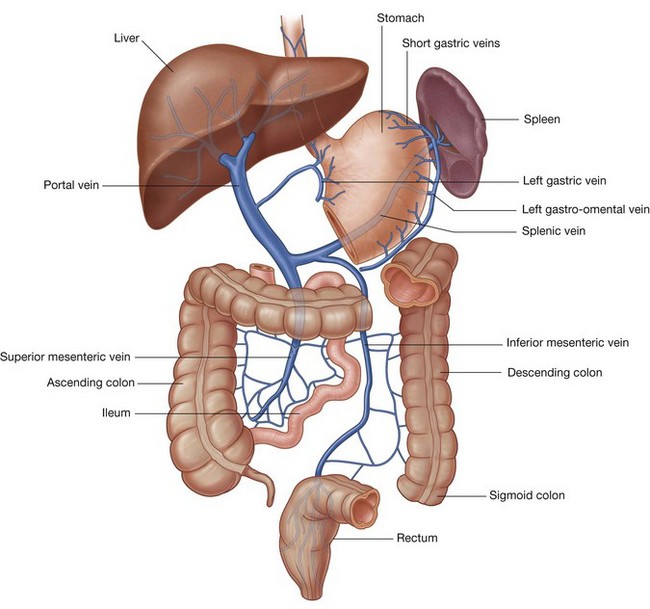
Blood from abdominal parts of the gastrointestinal system and the spleen passes through a second vascular bed, in the liver, before ultimately returning to the heart (Fig. 4.19).
Venous blood from the digestive tract, pancreas, gallbladder, and spleen enters the inferior surface of the liver through the large hepatic portal vein. This vein then ramifies like an artery to distribute blood to small endothelial-lined hepatic sinusoids, which form the vascular exchange network of the liver.
After passing through the sinusoids, the blood collects in a number of short hepatic veins, which drain into the inferior vena cava just before the inferior vena cava penetrates the diaphragm and enters the right atrium of the heart.
Normally, vascular beds drained by the hepatic portal system interconnect, through small veins, with beds drained by systemic vessels, which ultimately connect directly with either the superior or inferior vena cava.
Portacaval anastomoses
Among the clinically most important regions of overlap between the portal and caval systems are those at each end of the abdominal part of the gastrointestinal system:
Small veins that accompany the degenerate umbilical vein (round ligament of the liver) establish another important portacaval anastomosis.
The round ligament of the liver connects the umbilicus of the anterior abdominal wall with the left branch of the portal vein as it enters the liver. The small veins that accompany this ligament form a connection between the portal system and para-umbilical regions of the abdominal wall, which drain into systemic veins.
Other regions where portal and caval systems interconnect include:
Blockage of the hepatic portal vein or of vascular channels in the liver
Blockage of the hepatic portal vein or of vascular channels in the liver can affect the pattern of venous return from abdominal parts of the gastrointestinal system. Vessels that interconnect the portal and caval systems can become greatly enlarged and tortuous, allowing blood in tributaries of the portal system to bypass the liver, enter the caval system, and thereby return to the heart. Portal hypertension can result in esophageal varices and hemorrhoids at the esophageal and rectal ends of the gastrointestinal system, respectively, and in caput medusae in which systemic vessels that radiate from para-umbilical veins enlarge and become visible on the abdominal wall.
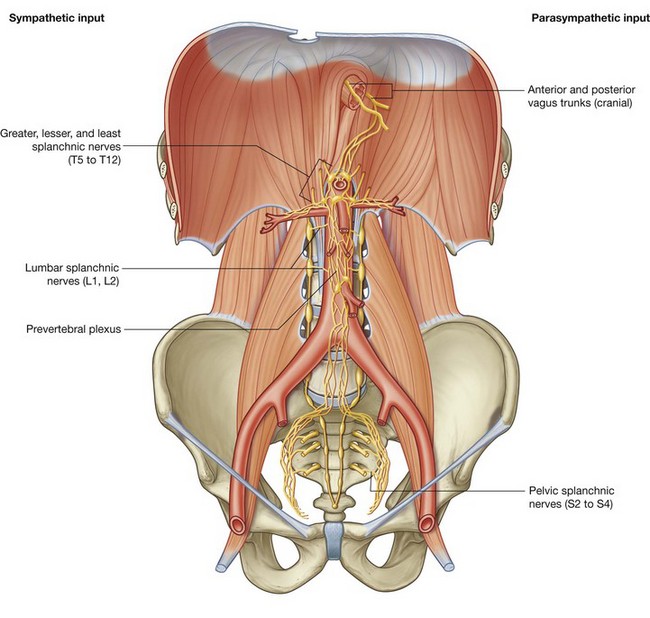
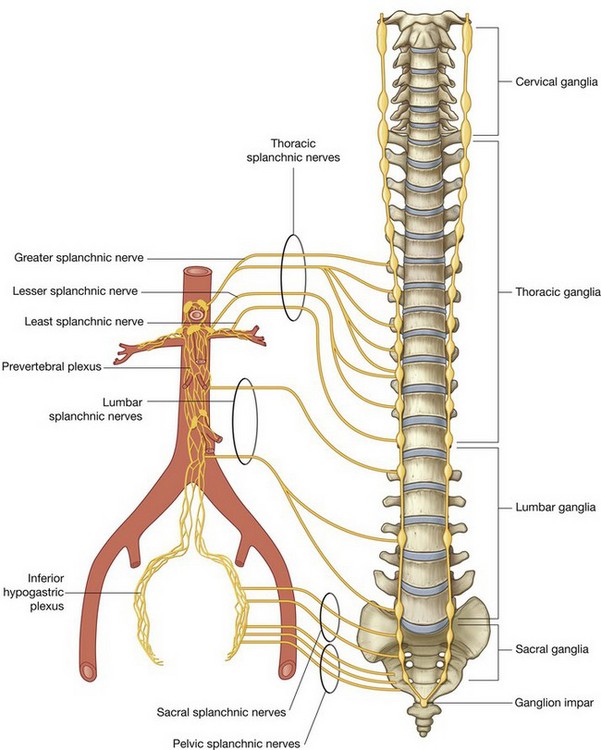
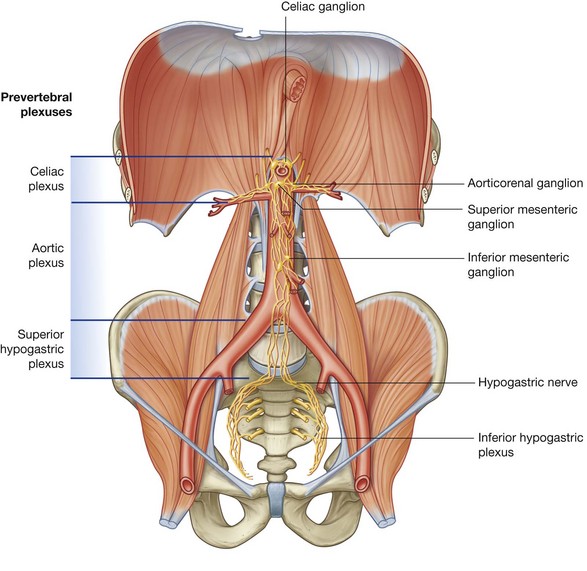
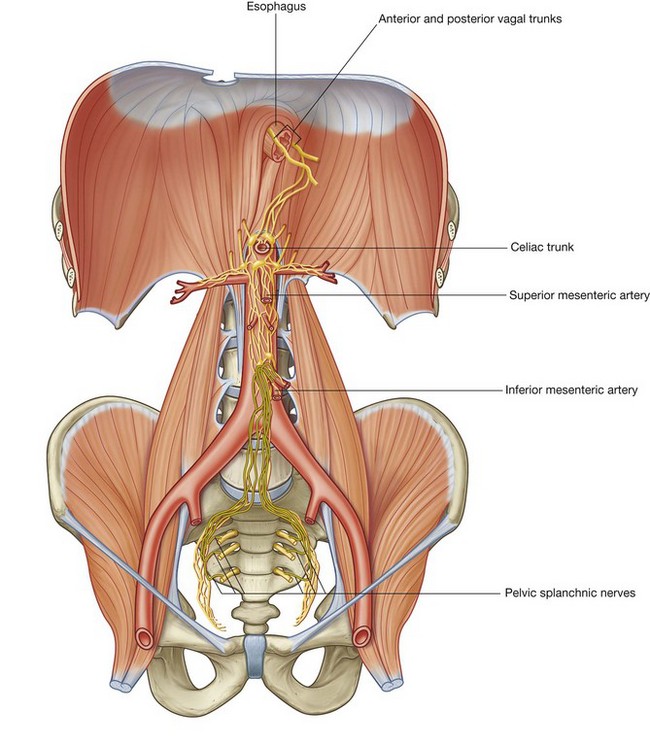
Abdominal viscera are supplied by a large prevertebral plexus
Innervation of the abdominal viscera is derived from a large prevertebral plexus associated mainly with the anterior and lateral surfaces of the aorta (Fig 4.20). Branches are distributed to target tissues along vessels that originate from the abdominal aorta.
The prevertebral plexus contains sympathetic, parasympathetic, and visceral sensory components:
Regional anatomy
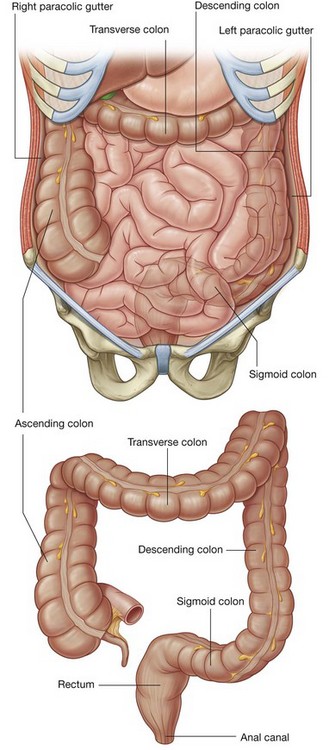
The abdomen is the part of the trunk inferior to the thorax (Fig. 4.21). Its musculomembranous walls surround a large cavity (the abdominal cavity), which is bounded superiorly by the diaphragm and inferiorly by the pelvic inlet.
The abdominal cavity may extend superiorly as high as the fourth intercostal space, and is continuous inferiorly with the pelvic cavity. It contains the peritoneal cavity and the abdominal viscera.
SURFACE TOPOGRAPHY
Topographical divisions of the abdomen are used to describe the location of abdominal organs and the pain associated with abdominal problems. The two schemes most often used are:
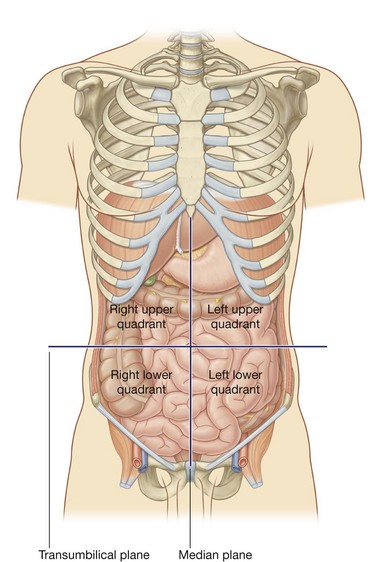
Four-quadrant pattern
A horizontal transumbilical plane passing through the umbilicus and the intervertebral disc between vertebrae LIII and LIV and intersecting with the vertical median plane divides the abdomen into four quadrants—the right upper, left upper, right lower, and left lower quadrants (Fig. 4.22).
Nine-region pattern
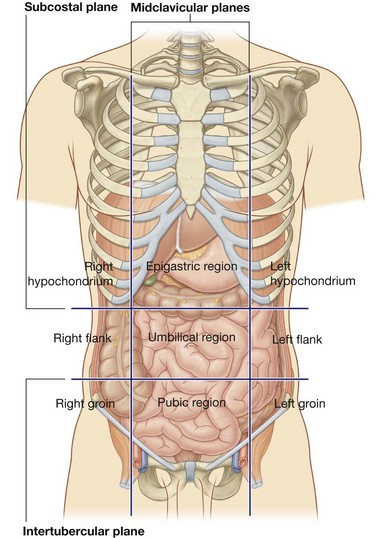
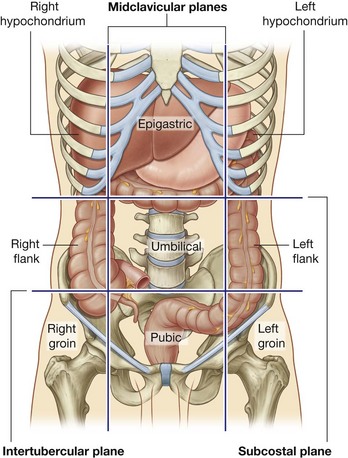
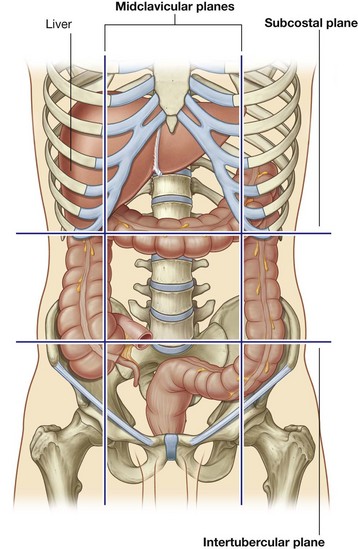
The nine-region pattern is based on two horizontal and two vertical planes (Fig. 4.23).
 The superior horizontal plane (the subcostal plane) is immediately inferior to the costal margins, which places it at the lower border of the costal cartilage of rib X and passes posteriorly through the body of vertebra LIII. (Note, however, that sometimes the transpyloric plane, halfway between the jugular notch and the symphysis pubis or halfway between the umbilicus and the inferior end of the body of the sternum, passing posteriorly through the lower border of vertebrae LI and intersecting with the costal margin at the ends of the ninth costal cartilages, is used instead.)
The superior horizontal plane (the subcostal plane) is immediately inferior to the costal margins, which places it at the lower border of the costal cartilage of rib X and passes posteriorly through the body of vertebra LIII. (Note, however, that sometimes the transpyloric plane, halfway between the jugular notch and the symphysis pubis or halfway between the umbilicus and the inferior end of the body of the sternum, passing posteriorly through the lower border of vertebrae LI and intersecting with the costal margin at the ends of the ninth costal cartilages, is used instead.) The inferior horizontal plane (the intertubercular plane) connects the tubercles of the iliac crests, which are palpable structures 5 cm posterior to the anterior superior iliac spines, and passes through the upper part of the body of vertebra LV.
The inferior horizontal plane (the intertubercular plane) connects the tubercles of the iliac crests, which are palpable structures 5 cm posterior to the anterior superior iliac spines, and passes through the upper part of the body of vertebra LV. The vertical planes pass from the midpoint of the clavicles inferiorly to a point midway between the anterior superior iliac spine and pubic symphysis.
The vertical planes pass from the midpoint of the clavicles inferiorly to a point midway between the anterior superior iliac spine and pubic symphysis.These four planes establish the topographical divisions in the nine-region organization. The following designations are used for each region: superiorly the right hypochondrium, the epigastric region, and the left hypochondrium; inferiorly the right groin (inguinal region), pubic region, and left groin (inguinal region); and in the middle the right flank (lateral region), the umbilical region, and the left flank (lateral region) (Fig. 4.23).
In the clinic
Surgical incisions
Access to the abdomen and its contents is usually obtained through incisions in the anterior abdominal wall. Traditionally, incisions have been placed at and around the region of surgical interest. The size of these incisions was usually large to allow good access and optimal visualization of the abdominal cavity. As anesthesia has developed and muscle-relaxing drugs have become widely used, the abdominal incisions have become smaller.
Currently, the most commonly used large abdominal incision is a central craniocaudad incision from the xiphoid process to the symphysis pubis, which provides wide access to the whole of the abdominal contents and allows an exploratory procedure to be performed (laparotomy).
Other approaches use much smaller incisions. With the advent of small cameras and the development of minimal access surgery, tiny incisions can be made in the anterior abdominal wall and cameras inserted. The peritoneal cavity is “inflated” with carbon dioxide to increase the space in which the procedure is performed. Further instruments may be inserted through small portholes, and procedures such as cholecystectomy (removal of the gallbladder) and appendectomy (removal of the appendix) can be carried out, allowing the patient to return home sooner than a large abdominal incision would allow.
ABDOMINAL WALL
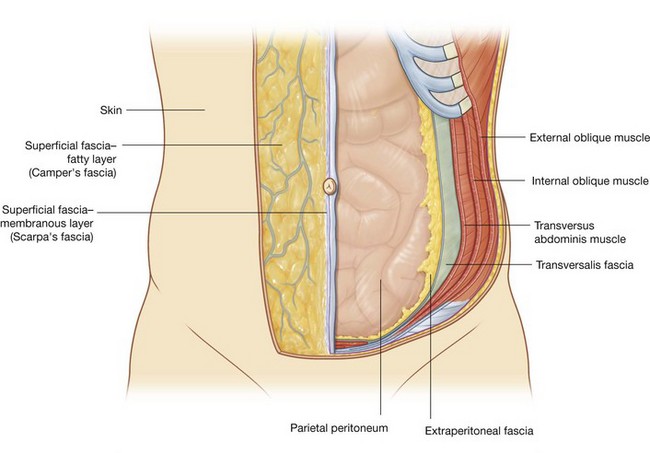
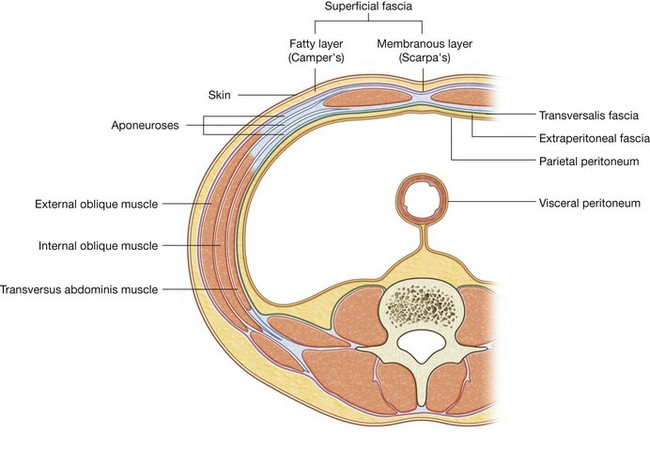
The abdominal wall covers a large area. It is bounded superiorly by the xiphoid process and costal margins, posteriorly by the vertebral column, and inferiorly by the upper parts of the pelvic bones. Its layers consist of skin, superficial fascia (subcutaneous tissue), muscles and their associated deep fascias, extraperitoneal fascia, and parietal peritoneum (Fig. 4.24).
Superficial fascia
The superficial fascia of the abdominal wall (subcutaneous tissue of abdomen) is a layer of fatty connective tissue. It is usually a single layer similar to, and continuous with, the superficial fascia throughout other regions of the body. However, in the lower region of the anterior part of the abdominal wall, below the umbilicus, it forms two layers: a superficial fatty layer and a deeper membranous layer.
Superficial layer
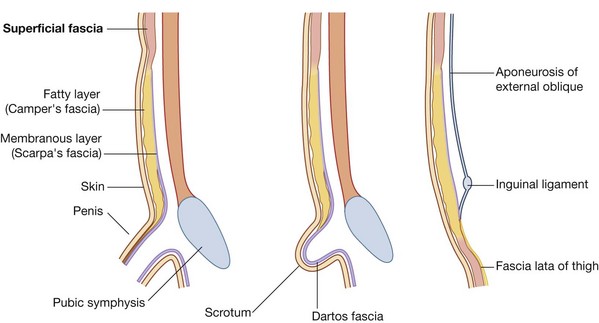
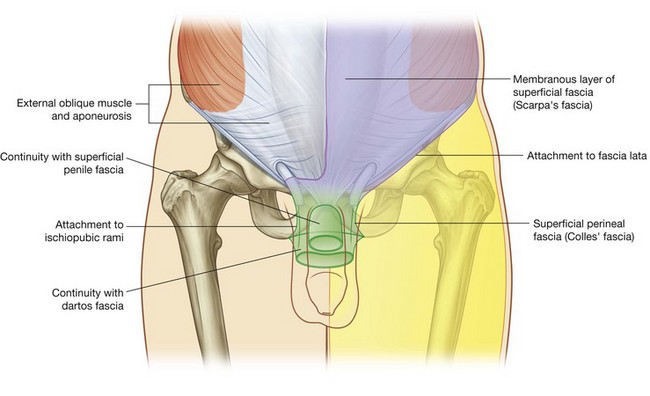
The superficial fatty layer of superficial fascia (Camper’s fascia) contains fat and varies in thickness (Figs. 4.25 and 4.26). It is continuous over the inguinal ligament with the superficial fascia of the thigh and with a similar layer in the perineum.
In men, this superficial layer continues over the penis and, after losing its fat and fusing with the deeper layer of superficial fascia, continues into the scrotum where it forms a specialized fascial layer containing smooth muscle fibers (the dartos fascia). In women, this superficial layer retains some fat and is a component of the labia majora.
Deeper layer
The deeper membranous layer of superficial fascia (Scarpa’s fascia) is thin and membranous, and contains little or no fat (Fig. 4.25). Inferiorly, it continues into the thigh, but just below the inguinal ligament, it fuses with the deep fascia of the thigh (the fascia lata; Fig. 4.26). In the midline, it is firmly attached to the linea alba and the symphysis pubis. It continues into the anterior part of the perineum where it is firmly attached to the ischiopubic rami and to the posterior margin of the perineal membrane. Here, it is referred to as the superficial perineal fascia (Colles’ fascia).
In men, the deeper membranous layer of superficial fascia blends with the superficial layer as they both pass over the penis, forming the superficial fascia of the penis, before they continue into the scrotum where they form the dartos fascia (Fig. 4.25). Also in men, extensions of the deeper membranous layer of superficial fascia attached to the pubic symphysis pass inferiorly onto the dorsum and sides of the penis to form the fundiform ligament of penis. In women, the membranous layer of the superficial fascia continues into the labia majora and the anterior part of the perineum.
Anterolateral muscles
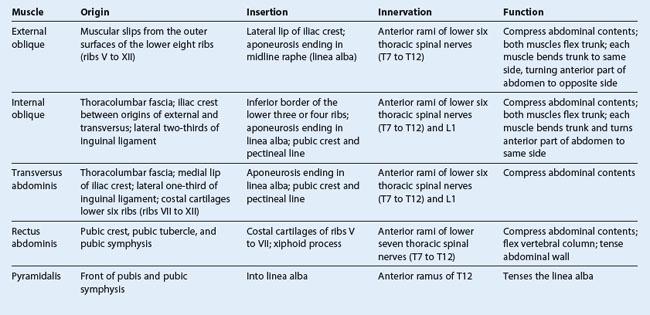
There are five muscles in the anterolateral group of abdominal wall muscles:
 three flat muscles whose fibers begin posterolaterally, pass anteriorly, and are replaced by an aponeurosis as the muscle continues towards the midline—the external oblique, internal oblique, and transversus abdominis muscles;
three flat muscles whose fibers begin posterolaterally, pass anteriorly, and are replaced by an aponeurosis as the muscle continues towards the midline—the external oblique, internal oblique, and transversus abdominis muscles; two vertical muscles, near the midline, which are enclosed within a tendinous sheath formed by the aponeuroses of the flat muscles—the rectus abdominis and pyramidalis muscles.
two vertical muscles, near the midline, which are enclosed within a tendinous sheath formed by the aponeuroses of the flat muscles—the rectus abdominis and pyramidalis muscles.Each of these five muscles has specific actions, but together the muscles are critical for the maintenance of many normal physiological functions. By their positioning, they form a firm, but flexible, wall that keeps the abdominal viscera within the abdominal cavity, protects the viscera from injury, and helps maintain the position of the viscera in the erect posture against the action of gravity.
In addition, contraction of these muscles assists in both quiet and forced expiration by pushing the viscera upward (which helps push the relaxed diaphragm further into the thoracic cavity) and in coughing and vomiting.
All these muscles are also involved in any action that increases intra-abdominal pressure, including parturition (childbirth), micturition (urination), and defecation (expulsion of feces from the rectum).
Flat muscles
External oblique
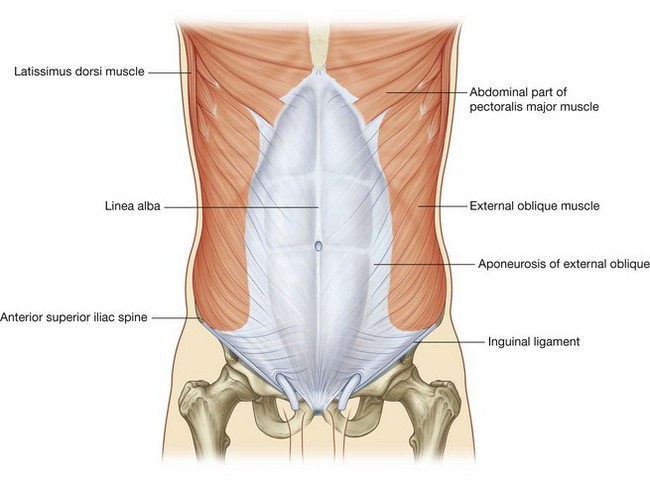
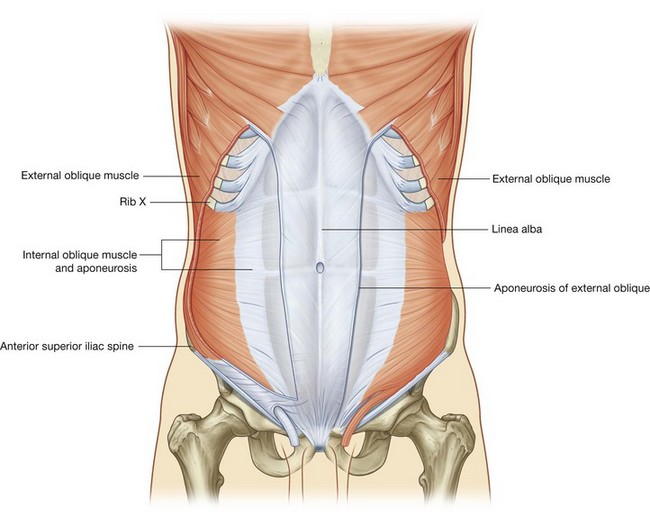
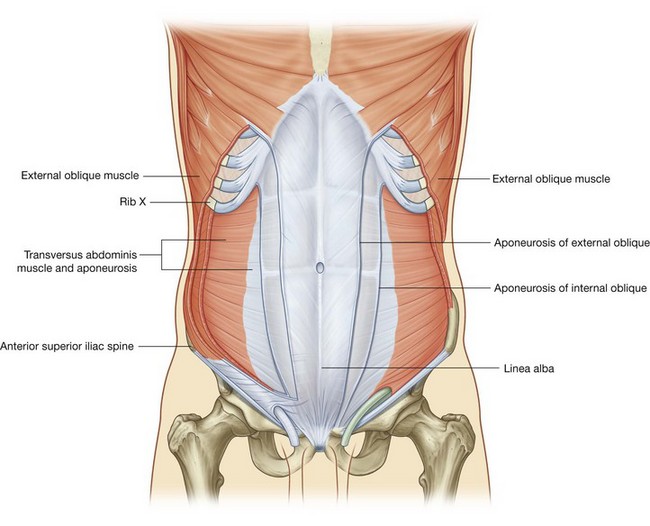
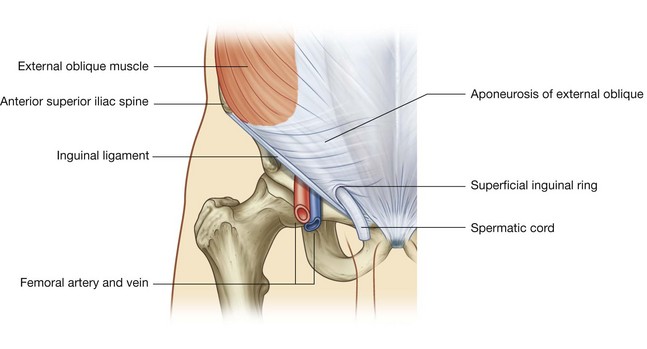
The most superficial of the three flat muscles in the anterolateral group of abdominal wall muscles is the external oblique, which is immediately deep to the superficial fascia (Fig. 4.27). Its laterally placed muscle fibers pass in an inferomedial direction, while its large aponeurotic component covers the anterior part of the abdominal wall to the midline. Approaching the midline, the aponeuroses are entwined, forming the linea alba, which extends from the xiphoid process to the pubic symphysis.
Associated ligaments
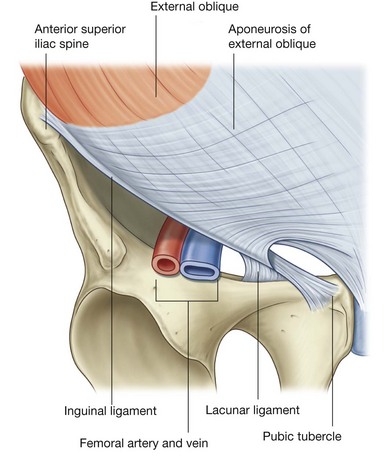
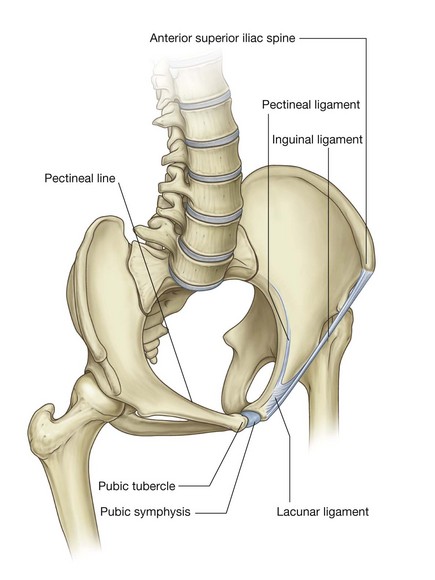
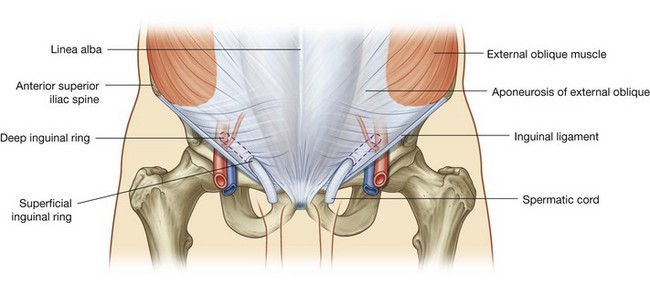
The lower border of the external oblique aponeurosis forms the inguinal ligament on each side (Fig. 4.27). This thickened reinforced free edge of the external oblique aponeurosis passes between the anterior superior iliac spine laterally and the pubic tubercle medially (Fig. 4.28). It folds under itself forming a trough, which plays an important role in the formation of the inguinal canal.
Several other ligaments are also formed from extensions of the fibers at the medial end of the inguinal ligament:
 the lacunar ligament is a crescent-shaped extension of fibers at the medial end of the inguinal ligament that pass backward to attach to the pecten pubis on the superior ramus of the pubic bone (Figs. 4.28 and 4.29);
the lacunar ligament is a crescent-shaped extension of fibers at the medial end of the inguinal ligament that pass backward to attach to the pecten pubis on the superior ramus of the pubic bone (Figs. 4.28 and 4.29);Internal oblique
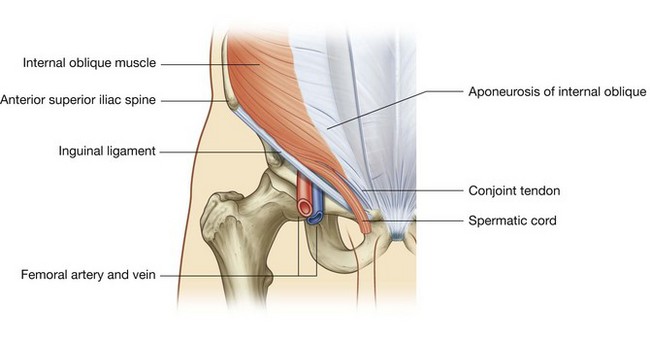
Deep to the external oblique muscle is the internal oblique muscle, which is the second of the three flat muscles (Fig. 4.30). This muscle is smaller and thinner than the external oblique, with most of its muscle fibers passing in a superomedial direction. Its lateral muscular components end anteriorly as an aponeurosis that blends into the linea alba at the midline.
Transversus abdominis
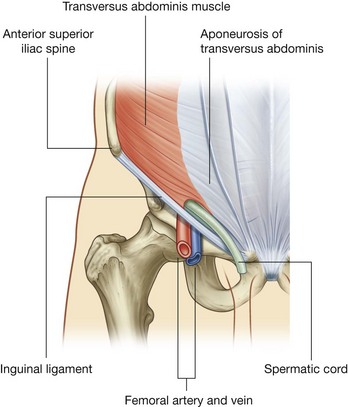
Deep to the internal oblique muscle is the transversus abdominis muscle (Fig. 4.31), so named because of the direction of most of its muscle fibers. It ends in an anterior aponeurosis, which blends with the linea alba at the midline.
Transversalis fascia
Each of the three flat muscles is covered on its anterior and posterior surfaces by a layer of deep (or investing) fascia. In general, these layers are unremarkable except for the layer deep to the transversus abdominis muscle (the transversalis fascia), which is better developed.
The transversalis fascia is a continuous layer of deep fascia that lines the abdominal cavity and continues into the pelvic cavity. It crosses the midline anteriorly, associating with the transversalis fascia of the opposite side, and is continuous with the fascia on the inferior surface of the diaphragm. It is continuous posteriorly with the deep fascia covering the muscles of the posterior abdominal wall and attaches to the thoracolumbar fascia.
After attaching to the crest of the ilium, the transversalis fascia blends with the fascia covering the muscles associated with the upper regions of the pelvic bones and with similar fascia covering the muscles of the pelvic cavity. At this point, it is referred to as the parietal pelvic (or endopelvic) fascia.
There is therefore a continuous layer of deep fascia surrounding the abdominal cavity that is thick in some areas, thin in others, attached or free, and participates in the formation of specialized structures.
Vertical muscles
The two vertical muscles in the anterolateral group of abdominal wall muscles (Table 4.1) are the large rectus abdominis and the small pyramidalis (Fig. 4.32).
Rectus abdominis
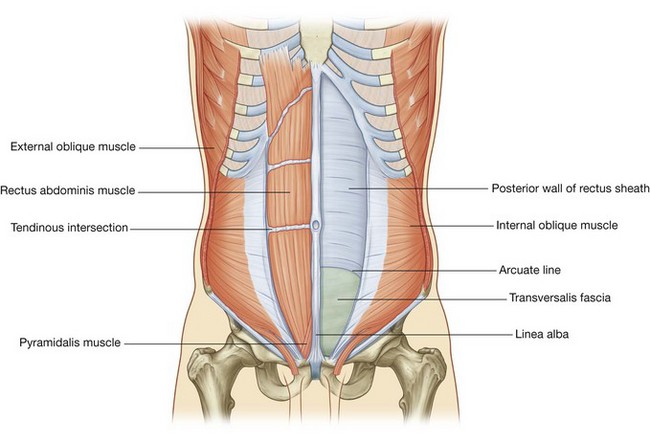
The rectus abdominis is a long, flat muscle and extends the length of the anterior abdominal wall. It is a paired muscle, separated in the midline by the linea alba, and it widens and thins as it ascends from the pubic symphysis to the costal margin. Along its course, it is intersected by three or four transverse fibrous bands or tendinous intersections (Fig. 4.32). These are easily visible on individuals with a well-developed rectus abdominis.
Pyramidalis
The second vertical muscle is the pyramidalis. This small, triangular muscle, which may be absent, is anterior to the rectus abdominis, has its base on the pubis, and its apex is attached superiorly and medially to the linea alba (Fig. 4.32).
Rectus sheath
The rectus abdominis and pyramidalis muscles are enclosed in an aponeurotic tendinous sheath (the rectus sheath) formed by a unique layering of the aponeuroses of the external and internal oblique, and transversus abdominis muscles (Fig. 4.33).
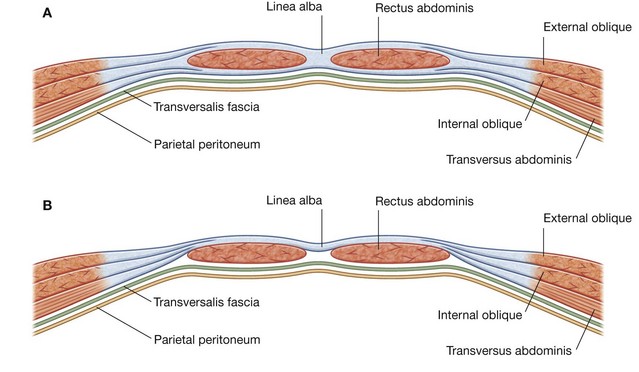
Fig. 4.33 Organization of the rectus sheath. A. Transverse section through the upper three-quarters of the rectus sheath. B. Transverse section through the lower one-quarter of the rectus sheath.
The rectus sheath completely encloses the upper three-quarters of the rectus abdominis and covers the anterior surface of the lower one-quarter of the muscle. As no sheath covers the posterior surface of the lower quarter of the rectus abdominis muscle, the muscle at this point is in direct contact with the transversalis fascia.
The formation of the rectus sheath surrounding the upper three-quarters of the rectus abdominis muscle has the following pattern:
 the anterior wall consists of the aponeurosis of the external oblique and half of the aponeurosis of the internal oblique, which splits at the lateral margin of the rectus abdominis;
the anterior wall consists of the aponeurosis of the external oblique and half of the aponeurosis of the internal oblique, which splits at the lateral margin of the rectus abdominis; the posterior wall of the rectus sheath consists of the other half of the aponeurosis of the internal oblique and the aponeurosis of the transversus abdominis.
the posterior wall of the rectus sheath consists of the other half of the aponeurosis of the internal oblique and the aponeurosis of the transversus abdominis.At a point midway between the umbilicus and the pubic symphysis, corresponding to the beginning of the lower one-quarter of the rectus abdominis muscle, all of the aponeuroses move anterior to the rectus muscle. There is no posterior wall of the rectus sheath and the anterior wall of the sheath consists of the aponeuroses of the external oblique, the internal oblique, and the transversus abdominis muscles. From this point inferiorly, the rectus abdominis muscle is in direct contact with the transversalis fascia. Marking this point of transition is an arch of fibers (the arcuate line; see Fig. 4.32).
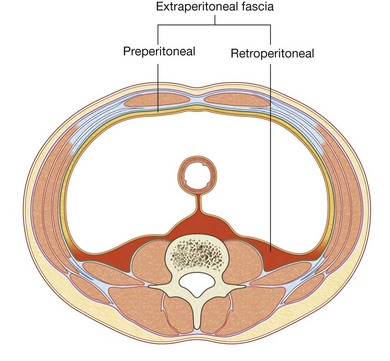
Extraperitoneal fascia
Deep to the transversalis fascia is a layer of connective tissue, the extraperitoneal fascia, which separates the transversalis fascia from the peritoneum (Fig. 4.34). Containing varying amounts of fat, this layer not only lines the abdominal cavity but is also continuous with a similar layer lining the pelvic cavity. It is abundant on the posterior abdominal wall, especially around the kidneys, continues over organs covered by peritoneal reflections, and, as the vasculature is located in this layer, extends into mesenteries with the blood vessels. Viscera in the extraperitoneal fascia are referred to as retroperitoneal.
In the description of specific surgical procedures, the terminology used to describe the extraperitoneal fascia is further modified. The fascia toward the anterior side of the body is described as preperitoneal (or, less commonly, properitoneal) and the fascia towards the posterior side of the body has been described as retroperitoneal (Fig. 4.35). Examples of the use of these terms would be the continuity of fat in the inguinal canal with the preperitoneal fat and a transabdominal preperitoneal laparoscopic repair of an inguinal hernia.
Peritoneum
Deep to the extraperitoneal fascia is the peritoneum (see Figs. 4.6 and 4.7 on pp. 251–252). This thin serous membrane lines the walls of the abdominal cavity and, at various points, reflects onto the abdominal viscera, providing either a complete or a partial covering. The peritoneum lining the walls is the parietal peritoneum; the peritoneum covering the viscera is the visceral peritoneum.
The continuous lining of the abdominal walls by the parietal peritoneum forms a sac. This sac is closed in men, but has two openings in women where the uterine tubes provide a passage to the outside. The closed sac in men and the semi-closed sac in women is called the peritoneal cavity.
Innervation
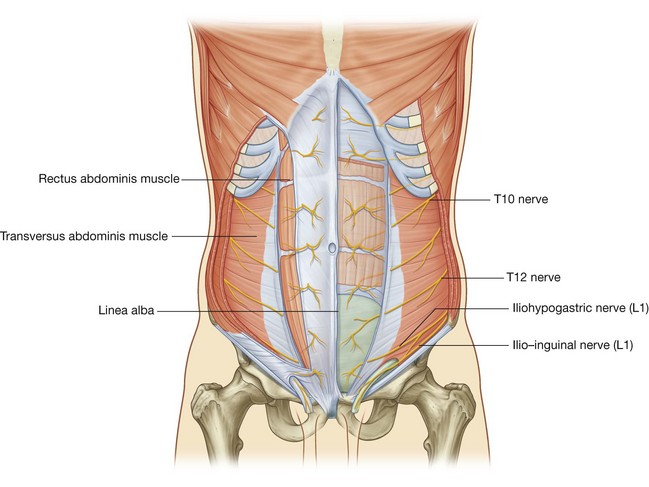
The skin, muscles, and parietal peritoneum of the anterolateral abdominal wall are supplied by T7 to T12 and L1 spinal nerves. The anterior rami of these spinal nerves pass around the body, from posterior to anterior, in an inferomedial direction (Fig. 4.36). As they proceed, they give off a lateral cutaneous branch and end as an anterior cutaneous branch.
The intercostal nerves (T7 to T11) leave their intercostal spaces, passing deep to the costal cartilages, and continue onto the anterolateral abdominal wall between the internal oblique and transversus abdominis muscles (Fig. 4.37). Reaching the lateral edge of the rectus sheath, they enter the rectus sheath and pass posterior to the lateral aspect of the rectus abdominis muscle. Approaching the midline, an anterior cutaneous branch passes through the rectus abdominis muscle and the anterior wall of the rectus sheath to supply the skin.
Spinal nerve T12 (the subcostal nerve) follows a similar course as the intercostals. Branches of L1 (the iliohypogastric nerve and ilio-inguinal nerve), which originate from the lumbar plexus, follow similar courses initially, but deviate from this pattern near their final destination.
Along their course, nerves T7 to T12 and L1 supply branches to the anterolateral abdominal wall muscles and the underlying parietal peritoneum. All terminate by supplying skin:
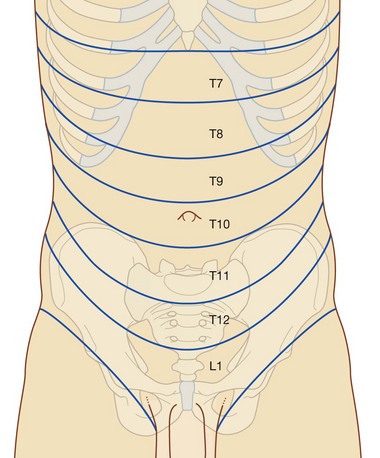
 T11, T12, and L1 supply the skin from just below the umbilicus to, and including, the pubic region (Fig. 4.38);
T11, T12, and L1 supply the skin from just below the umbilicus to, and including, the pubic region (Fig. 4.38);Arterial supply and venous drainage
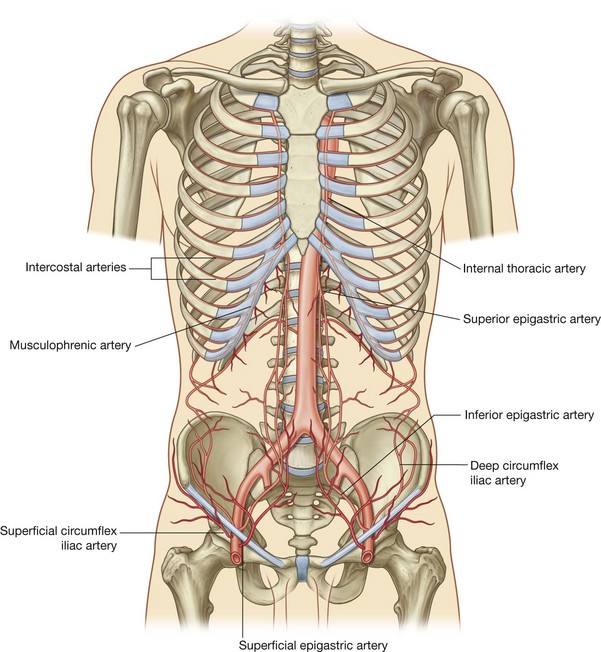
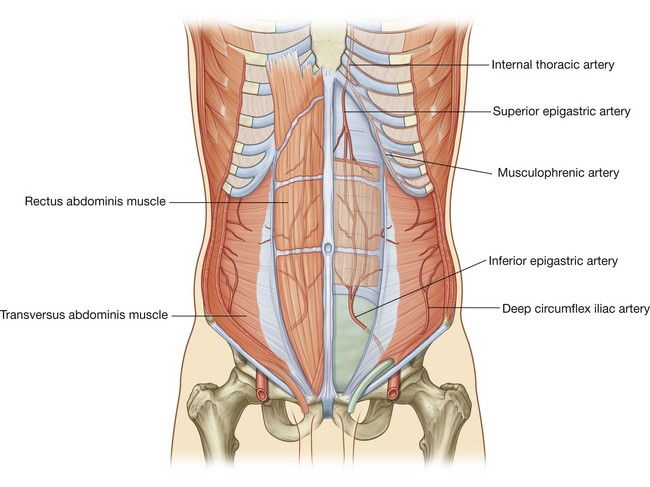
Numerous blood vessels supply the anterolateral abdominal wall. Superficially:
 the superior part of the wall is supplied by branches from the musculophrenic artery, a terminal branch of the internal thoracic artery; and
the superior part of the wall is supplied by branches from the musculophrenic artery, a terminal branch of the internal thoracic artery; and the inferior part of the wall is supplied by the medially placed superficial epigastric artery and the laterally placed superficial circumflex iliac artery, both branches of the femoral artery (Fig. 4.39).
the inferior part of the wall is supplied by the medially placed superficial epigastric artery and the laterally placed superficial circumflex iliac artery, both branches of the femoral artery (Fig. 4.39). the superior part of the wall is supplied by the superior epigastric artery, a terminal branch of the internal thoracic artery;
the superior part of the wall is supplied by the superior epigastric artery, a terminal branch of the internal thoracic artery; the lateral part of the wall is supplied by branches of the tenth and eleventh intercostal arteries and the subcostal artery; and
the lateral part of the wall is supplied by branches of the tenth and eleventh intercostal arteries and the subcostal artery; and the inferior part of the wall is supplied by the medially placed inferior epigastric artery and the laterally placed deep circumflex iliac artery, both branches of the external iliac artery.
the inferior part of the wall is supplied by the medially placed inferior epigastric artery and the laterally placed deep circumflex iliac artery, both branches of the external iliac artery.The superior and inferior epigastric arteries both enter the rectus sheath. They are posterior to the rectus abdominis muscle throughout their course, and anastomose with each other (Fig. 4.40).
Veins of similar names follow the arteries and are responsible for venous drainage.
Lymphatic drainage
Lymphatic drainage of the anterolateral abdominal wall follows the basic principles of lymphatic drainage:
GROIN
The groin (inguinal region) is the area of junction between the anterior abdominal wall and the thigh. In this area, the abdominal wall is weakened from changes that occur during development and a peritoneal sac or diverticulum, with or without abdominal contents, can therefore protrude through it, creating an inguinal hernia. This type of hernia can occur in both sexes, but it is most common in males.
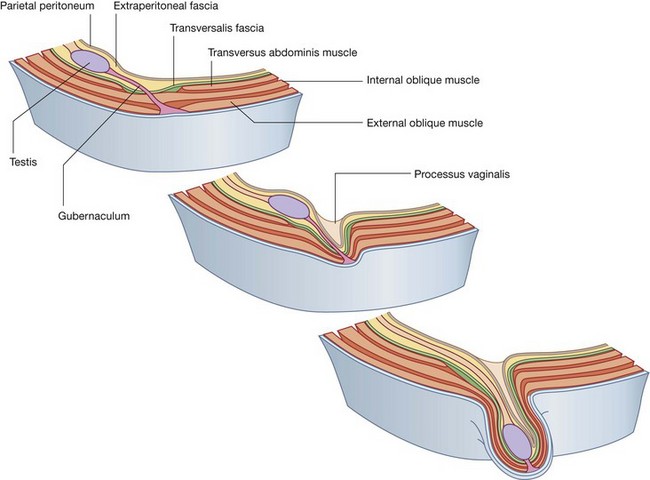
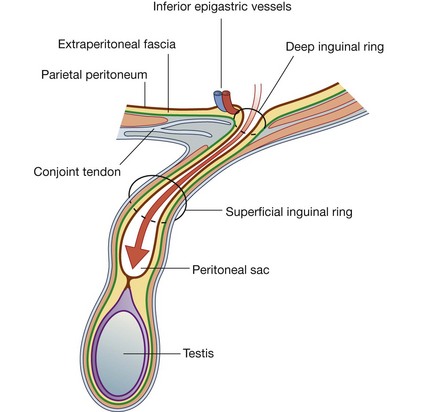
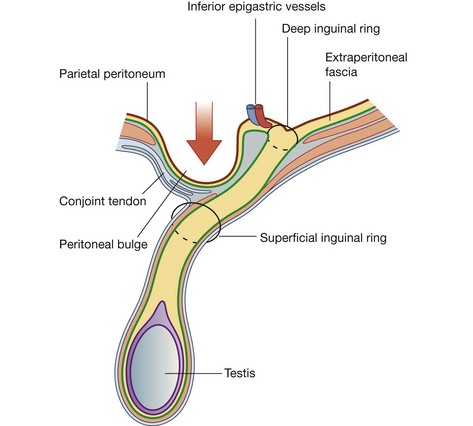
The inherent weakness in the anterior abdominal wall in the groin is caused by changes that occur during the development of the gonads. Before the descent of the testis and ovaries from their initial position high in the posterior abdominal wall, a peritoneal outpouching (the processus vaginalis) forms (Fig. 4.41), protruding through the various layers of the anterior abdominal wall and acquiring coverings from each:
 the second covering is formed by the musculature of the internal oblique (a covering from the transversus abdominis muscle is not acquired because the processus vaginalis passes under the arching fibers of this abdominal wall muscle);
the second covering is formed by the musculature of the internal oblique (a covering from the transversus abdominis muscle is not acquired because the processus vaginalis passes under the arching fibers of this abdominal wall muscle);As a result the processus vaginalis is transformed into a tubular structure with multiple coverings from the layers of the anterior abdominal wall. This forms the basic structure of the inguinal canal.
The final event in this development is the descent of the testes into the scrotum or of the ovaries into the pelvic cavity. This process depends on the development of the gubernaculum, which extends from the inferior border of the developing gonad to the labioscrotal swellings (Fig. 4.41).
The processus vaginalis is immediately anterior to the gubernaculum within the inguinal canal.
In men, as the testes descend, the testes and their accompanying vessels, ducts, and nerves pass through the inguinal canal and are therefore surrounded by the same fascial layers of the abdominal wall. Testicular descent completes the formation of the spermatic cord in men.
In women, the ovaries descend into the pelvic cavity and become associated with the developing uterus. Therefore, the only remaining structure passing through the inguinal canal is the round ligament of the uterus, which is a remnant of the gubernaculum.
The development sequence is concluded in both sexes when the processus vaginalis obliterates. If this does not occur or is incomplete, a potential weakness exists in the anterior abdominal wall and an inguinal hernia may develop. In males, only proximal regions of the tunica vaginalis obliterate. The distal end expands to enclose most of the testis in the scrotum. In other words, the cavity of the tunica vaginalis in men forms as an extension of the developing peritoneal cavity that becomes separated off during development.
Inguinal canal
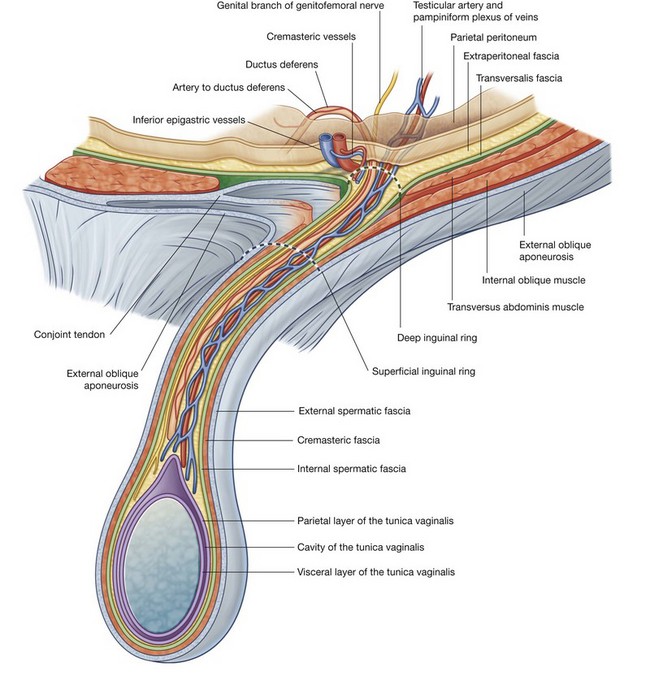
The inguinal canal is a slit-like passage that extends in a downward and medial direction, just above and parallel to the lower half of the inguinal ligament. It begins at the deep inguinal ring and continues for approximately 4 cm, ending at the superficial inguinal ring (Fig. 4.42). The contents of the canal are the genital branch of the genitofemoral nerve, the spermatic cord in men and the round ligament of the uterus in women. Additionally, in both sexes, the ilio-inguinal nerve passes through part of the canal, exiting through the superficial inguinal ring with the other contents.
Deep inguinal ring
The deep (internal) inguinal ring is the beginning of the inguinal canal and is at a point midway between the anterior superior iliac spine and the pubic symphysis (Fig. 4.43). It is just above the inguinal ligament and immediately lateral to the inferior epigastric vessels. Although sometimes referred to as a defect or opening in the transversalis fascia, it is actually the beginning of the tubular evagination of transversalis fascia that forms one of the coverings (the internal spermatic fascia) of the spermatic cord in men or the round ligament of the uterus in women.
Superficial inguinal ring
The superficial (external) inguinal ring is the end of the inguinal canal and is superior to the pubic tubercle (Fig. 4.44). It is a triangular opening in the aponeurosis of the external oblique, with its apex pointing superolaterally and its base formed by the pubic crest. The two remaining sides of the triangle (the medial crus and the lateral crus) are attached to the pubic symphysis and the pubic tubercle, respectively. At the apex of the triangle the two crura are held together by crossing (intercrural) fibers, which prevent further widening of the superficial ring.
As with the deep inguinal ring, the superficial inguinal ring is actually the beginning of the tubular evagination of the aponeurosis of the external oblique onto the structures traversing the inguinal canal and emerging from the superficial inguinal ring. This continuation of tissue over the spermatic cord is the external spermatic fascia.
Anterior wall
The anterior wall of the inguinal canal is formed along its entire length by the aponeurosis of the external oblique muscle (Fig. 4.44). It is also reinforced laterally by the lower fibers of the internal oblique that originate from the lateral two-thirds of the inguinal ligament (Fig. 4.45). This adds an additional covering over the deep inguinal ring, which is a potential point of weakness in the anterior abdominal wall. Furthermore, as the internal oblique muscle covers the deep inguinal ring, it also contributes a layer (the cremasteric fascia containing the cremasteric muscle) to the coverings of the structures traversing the inguinal canal.
Posterior wall
The posterior wall of the inguinal canal is formed along its entire length by the transversalis fascia (see Fig. 4.43). It is reinforced along its medial one-third by the conjoint tendon (inguinal falx; Fig. 4.45). This tendon is the combined insertion of the transversus abdominis and internal oblique muscles into the pubic crest and pectineal line.
As with the internal oblique muscle’s reinforcement of the area of the deep inguinal ring, the position of the conjoint tendon posterior to the superficial inguinal ring provides additional support to a potential point of weakness in the anterior abdominal wall.
Roof
The roof (superior wall) of the inguinal canal is formed by the arching fibers of the transversus abdominis and internal oblique muscles (Figs. 4.45 and 4.46). They pass from their lateral points of origin from the inguinal ligament to their common medial attachment as the conjoint tendon.
Floor
The floor (inferior wall) of the inguinal canal is formed by the medial one-half of the inguinal ligament. This rolled-under, free margin of the lowest part of the aponeurosis of the external oblique forms a gutter or trough on which the contents of the inguinal canal are positioned. The lacunar ligament reinforces most of the medial part of the gutter.
Contents
The contents of the inguinal canal are:
These structures enter the inguinal canal through the deep inguinal ring and exit it through the superficial inguinal ring.
Additionally, the ilio-inguinal nerve (L1) passes through part of the inguinal canal. This nerve is a branch of the lumbar plexus, enters the abdominal wall posteriorly by piercing the internal surface of the transversus abdominis muscle, and continues through the layers of the anterior abdominal wall by piercing the internal oblique muscle. As it continues to pass inferomedially, it enters the inguinal canal. It continues down the canal to exit through the superficial inguinal ring.
Spermatic cord
The spermatic cord begins to form proximally at the deep inguinal ring and consists of structures passing between the abdominopelvic cavities and the testis, and the three fascial coverings that enclose these structures (Fig. 4.47).
The structures in the spermatic cord include:
These structures enter the deep inguinal ring, proceed down the inguinal canal, and exit from the superficial inguinal ring, having acquired the three fascial coverings during their journey. This collection of structures and fascias continues into the scrotum where the structures connect with the testes and the fascias surround the testes.
The fascias enclosing the contents of the spermatic cord include:
 the internal spermatic fascia, which is the deepest layer, arises from the transversalis fascia, and is attached to the margins of the deep inguinal ring;
the internal spermatic fascia, which is the deepest layer, arises from the transversalis fascia, and is attached to the margins of the deep inguinal ring; the cremasteric fascia with the associated cremasteric muscle, which is the middle fascial layer and arises from the internal oblique muscle; and
the cremasteric fascia with the associated cremasteric muscle, which is the middle fascial layer and arises from the internal oblique muscle; and the external spermatic fascia, which is the most superficial covering of the spermatic cord, arises from the aponeurosis of the external oblique muscle, and is attached to the margins of the superficial inguinal ring (Fig. 4.47).
the external spermatic fascia, which is the most superficial covering of the spermatic cord, arises from the aponeurosis of the external oblique muscle, and is attached to the margins of the superficial inguinal ring (Fig. 4.47).Round ligament of the uterus
The round ligament of the uterus is a cord-like structure that passes from the uterus to the deep inguinal ring where it enters the inguinal canal. It passes down the inguinal canal and exits through the superficial inguinal ring. At this point, it has changed from a cord-like structure to a few strands of tissue, which attach to the connective tissue associated with the labia majora. As it traverses the inguinal canal, it acquires the same coverings as the spermatic cord in men.
The round ligament of the uterus is the long distal part of the original gubernaculum in the fetus that extends from the ovary to the labioscrotal swellings. From its attachment to the uterus, the round ligament of the uterus continues to the ovary as the ligament of the ovary that develops from the short proximal end of the gubernaculum.
In the clinic
Cremasteric reflex
In men, the cremaster muscle and cremasteric fascia form the middle or second covering of the spermatic cord. This muscle and its associated fascia are supplied by the genital branch of the genitofemoral nerve (L1/L2). Contraction of this muscle can be stimulated by a reflex arc. Gentle touch at and around the skin of the medial aspect of the superior part of the thigh stimulates the sensory fibers in the ilio-inguinal nerve. These sensory fibers enter the spinal cord at level L1. At this level, the sensory fibers stimulate the motor fibers carried in the genital branch of the genitofemoral nerve.
The cremasteric reflex is more active in children, tending to diminish with age. As with many reflexes, it may be absent in certain neurological disorders. Although it can be used for testing spinal cord function at level L1 in men, its clinical use is limited.
Inguinal hernias
An inguinal hernia is the protrusion or passage of a peritoneal sac, with or without abdominal contents, through a weakened part of the abdominal wall in the groin. It occurs because the peritoneal sac enters the inguinal canal either:
Inguinal hernias are therefore classified as either indirect or direct.
Indirect inguinal hernias
The indirect inguinal hernia is the most common of the two types of inguinal hernia and is much more common in men than in women (Fig. 4.48). It occurs because some part, or all, of the embryonic processus vaginalis remains open or patent. It is therefore referred to as being congenital in origin.
The protruding peritoneal sac enters the inguinal canal by passing through the deep inguinal ring, just lateral to the inferior epigastric vessels. The extent of its excursion down the inguinal canal depends on the amount of processus vaginalis that remains patent. If the entire processus vaginalis remains patent, the peritoneal sac may traverse the length of the canal, exit the superficial inguinal ring, and continue into the scrotum in men or the labia majus in women. In this case, the protruding peritoneal sac acquires the same three coverings as those associated with the spermatic cord in men or the round ligament of the uterus in women.
Direct inguinal hernias
A peritoneal sac that enters the medial end of the inguinal canal directly through a weakened posterior wall is a direct inguinal hernia (Fig. 4.49). It is usually described as acquired because it develops when abdominal musculature has been weakened, and is commonly seen in mature men. The bulging occurs medial to the inferior epigastric vessels in the inguinal triangle (Hesselbach’s triangle), which is bounded:
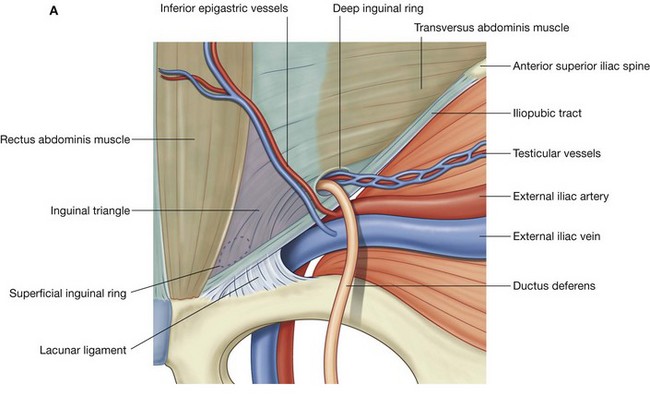
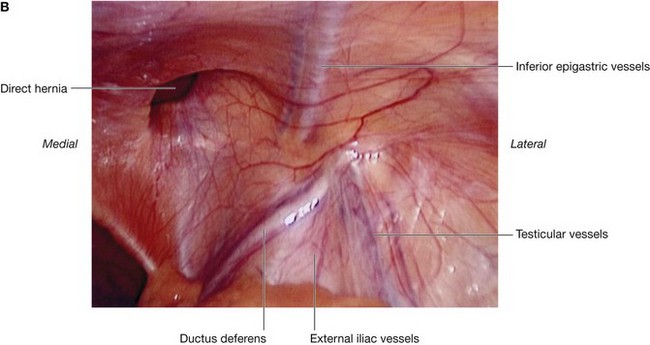
Fig. 4.50 Right inguinal triangle. A. Internal view. B. Laparoscopic view showing the parietal peritoneum still covering the area.
Internally, a thickening of the transversalis fascia (the iliopubic tract) follows the course of the inguinal ligament (Fig. 4.50).
This type of inguinal hernia does not traverse the entire length of the inguinal canal, but may exit through the superficial inguinal ring. When this occurs, the peritoneal sac acquires a layer of external spermatic fascia and can extend, like an indirect hernia, into the scrotum.
In the clinic
Masses around the groin
Around the groin there is a complex confluence of anatomical structures. Careful examination and good anatomical knowledge allows determination of the correct anatomical structure from which the mass arises and therefore the diagnosis. The most common masses in the groin are hernias.
The key to groin examination is determining the position of the inguinal ligament. The inguinal ligament passes between the anterior superior iliac spine laterally and the pubic tubercle medially. Inguinal hernias are above the inguinal ligament and are usually more apparent on standing. A visual assessment of the lump is necessary, bearing in mind the anatomical landmarks of the inguinal ligament.
In men, it is wise to examine the scrotum to check for a lump. If an abnormal mass is present, an inability to feel its upper edge suggests that it may originate from the inguinal canal and might be a hernia. By placing the hand over the lump and asking the patient to cough, the lump bulges outward.
An attempt should be made to reduce the swelling by applying gentle, firm pressure over the lump. If the lump is reducible, the hand should be withdrawn and careful observation will reveal recurrence of the mass.
The position of an abnormal mass in the groin relative to the pubic tubercle is very important, as are the presence of increased temperature and pain, which may represent early signs of strangulation or infection.
 an inguinal hernia appears through the superficial inguinal ring above the pubic tubercle and crest; and
an inguinal hernia appears through the superficial inguinal ring above the pubic tubercle and crest; and a femoral hernia (see below) appears through the femoral canal below and lateral to the pubic tubercle.
a femoral hernia (see below) appears through the femoral canal below and lateral to the pubic tubercle.A hernia is the protrusion of a viscus, in part or in whole, through a normal or abnormal opening. The viscus usually carries a covering of parietal peritoneum, which forms the lining of the hernial sac.
Inguinal hernias
Hernias occur in a variety of regions. The commonest site is the groin of the lower anterior abdominal wall. In some patients, inguinal hernias are present from birth (congenital) and are caused by the persistence of the processus vaginalis and the passage of viscera through the inguinal canal. Acquired hernias occur in older patients and causes include raised intra-abdominal pressure (e.g., from repeated coughing associated with lung disease), damage to nerves of the anterior abdominal wall (e.g., from surgical abdominal incisions), and weakening of the walls of the inguinal canal.
One of the potential problems with hernias is that bowel and fat may become stuck within the hernial sac. This can cause appreciable pain and bowel obstruction, necessitating urgent surgery. Another potential risk is strangulation of the hernia, in which the blood supply to the bowel is cut off at the neck of the hernial sac, rendering the bowel ischemic and susceptible to perforation.
The hernial sac of an indirect inguinal hernia enters the deep inguinal ring and passes through the inguinal canal. If the hernia is large enough, the hernial sac may emerge through the superficial inguinal ring. In men, such a hernia may extend into the scrotum (Fig. 4.51).
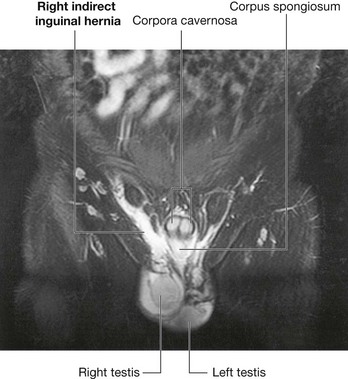
Fig. 4.51 Right indirect inguinal hernia. T2, fat saturated, weighted magnetic resonance image in the coronal plane of a male groin.
The hernial sac of a direct inguinal hernia pushes forward through the posterior wall of the inguinal canal immediately posterior to the superficial inguinal ring. The hernia protrudes directly forward medial to the inferior epigastric vessels and through the superficial inguinal ring.
The differentiation between an indirect and a direct inguinal hernia is made during surgery when the inferior epigastric vessels are identified at the medial edge of the deep internal ring:
Inguinal hernias occur more commonly in men than in women possibly because men have a much larger inguinal canal than women.
Femoral hernias
A femoral hernia passes through the femoral canal and into the medial aspect of the anterior thigh. The femoral canal lies at the medial edge of the femoral sheath, which contains the femoral artery, femoral vein, and lymphatics. The neck of the femoral canal is extremely narrow and is prone to trapping bowel within the sac, so making this type of hernia irreducible and susceptible to bowel strangulation. Femoral hernias are usually acquired, are not congenital, and most commonly occur in middle-aged and elderly populations. In addition, because women generally have wider pelvises than men, they tend to occur more commonly in women.
Umbilical hernias
Umbilical hernias are rare. Occasionally, they are congenital and result from failure of the small bowel to return to the abdominal cavity from the umbilical cord during development. After birth, umbilical hernias may result from incomplete closure of the umbilicus (navel). Overall, most of these hernias close in the first year of life, and surgical repair is not generally attempted until later.
Para-umbilical hernias may occur in adults at and around the umbilicus and often have small necks, so requiring surgical treatment.
Incisional hernias
Incisional hernias occur through a defect in a scar of a previous abdominal operation. Usually, the necks of these hernias are wide and do not therefore strangulate the viscera they contain.
Other hernias
A spigelian hernia passes upward through the arcuate line into the lateral border at the lower part of the posterior rectus sheath. It may appear as a tender mass on one side of the lower anterior abdominal wall.
Abdominopelvic cavity hernias can also develop in association with the pelvic walls, and sites include the obturator canal, the greater sciatic foramen, above and below the piriformis muscle.
ABDOMINAL VISCERA
Peritoneum
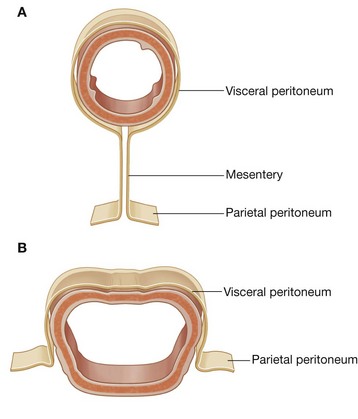
A thin membrane (the peritoneum) lines the walls of the abdominal cavity and covers much of the viscera. The parietal peritoneum lines the walls of the cavity and the visceral peritoneum covers the viscera. Between the parietal and visceral layers of peritoneum is a potential space (the peritoneal cavity). Abdominal viscera either are suspended in the peritoneal cavity by folds of peritoneum (mesenteries) or are outside the peritoneal cavity. Organs suspended in the cavity are referred to as intraperitoneal (Fig. 4.52); organs outside the peritoneal cavity, with only one surface or part of one surface covered by peritoneum, are retroperitoneal.
Innervation of the peritoneum
The parietal peritoneum associated with the abdominal wall is innervated by somatic afferents carried in branches of the associated spinal nerves and is therefore sensitive to well-localized pain. The visceral peritoneum is innervated by visceral afferents that accompany autonomic nerves (sympathetic and parasympathetic) back to the central nervous system. Activation of these fibers can lead to referred and poorly localized sensations of discomfort, and to reflex visceral motor activity.
Peritoneal cavity
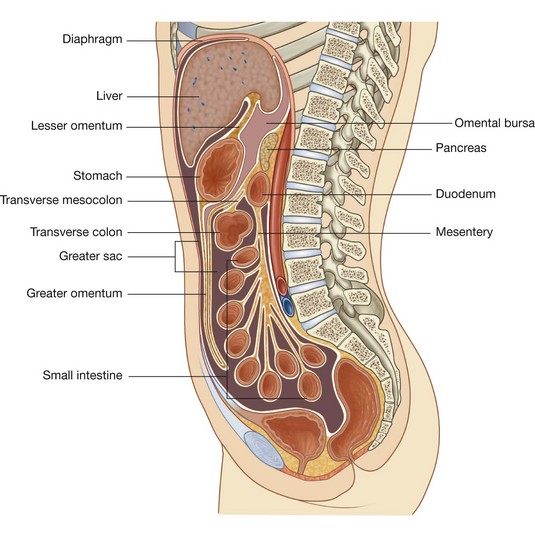
The peritoneal cavity is subdivided into the greater sac and the omental bursa (lesser sac; Fig. 4.53).
 The greater sac accounts for most of the space in the peritoneal cavity, beginning superiorly at the diaphragm and continuing inferiorly into the pelvic cavity. It is entered once the parietal peritoneum has been penetrated.
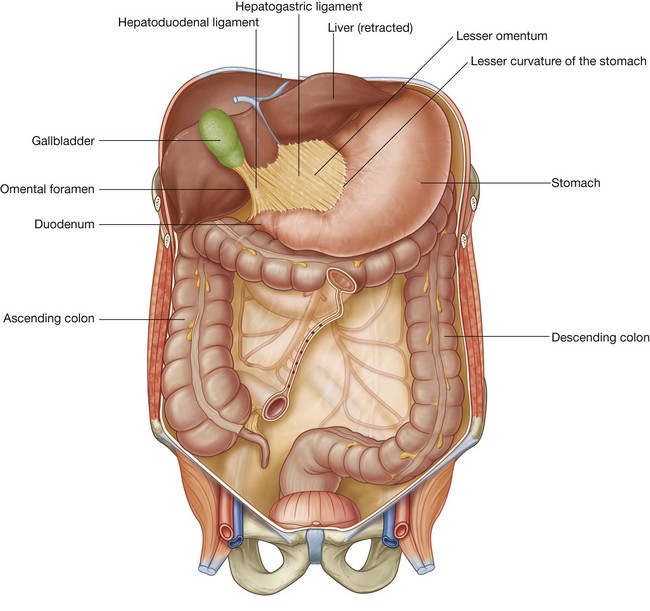
The greater sac accounts for most of the space in the peritoneal cavity, beginning superiorly at the diaphragm and continuing inferiorly into the pelvic cavity. It is entered once the parietal peritoneum has been penetrated. The omental bursa is a smaller subdivision of the peritoneal cavity posterior to the stomach and liver and is continuous with the greater sac through an opening, the omental (epiploic) foramen (Fig. 4.54).
The omental bursa is a smaller subdivision of the peritoneal cavity posterior to the stomach and liver and is continuous with the greater sac through an opening, the omental (epiploic) foramen (Fig. 4.54).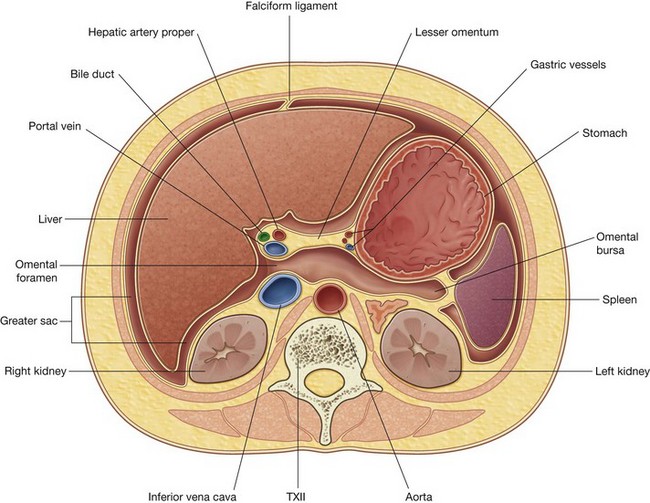
Fig. 4.54 Transverse section illustrating the continuity between the greater and lesser sacs through the omental (epiploic foramen).
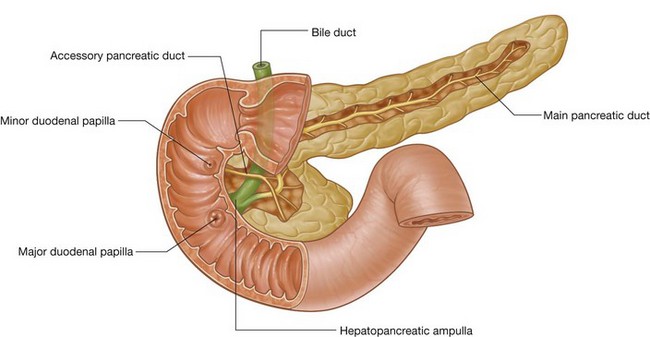
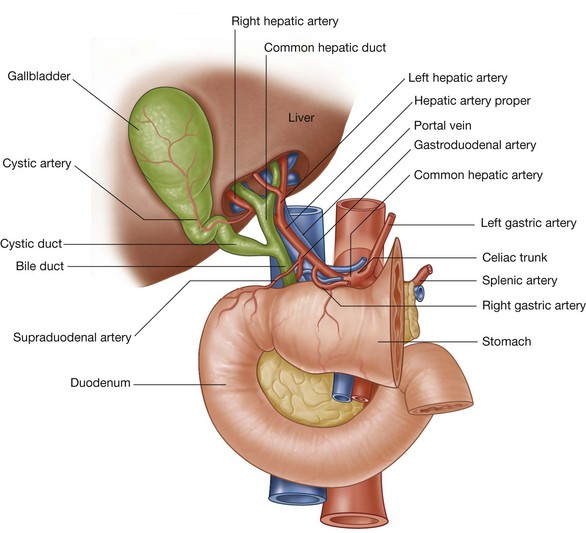
Surrounding the omental (epiploic) foramen are numerous structures covered with peritoneum. They include the portal vein, hepatic artery proper, and bile duct anteriorly; the inferior vena cava posteriorly; the caudate lobe of the liver superiorly; and the first part of the duodenum inferiorly.
In the clinic
Peritoneum
A small volume of peritoneal fluid within the peritoneal cavity lubricates movement of the viscera suspended in the abdominal cavity.
The peritoneal space has a large surface area, which facilitates the spread of disease through the peritoneal cavity and over the bowel and visceral surfaces. Conversely, this large surface area can be used for administering certain types of treatment and a number of procedures.
Ventriculoperitoneal shunts
Patients with obstructive hydrocephalus (an excessive accumulation of cerebrospinal fluid within the cerebral ventricular system) require continuous drainage of this fluid. This is achieved by placing a fine-bore catheter through the skull into the cerebral ventricles and placing the extracranial part of the tube beneath the scalp and skin of the chest wall and then passing it through the abdominal wall into the peritoneal cavity. Cerebrospinal fluid drains through the tube into the peritoneal cavity where it is absorbed.
Dialysis and peritoneal dialysis
People who develop renal failure require dialysis to live. There are two methods.
In the first method (hemodialysis), blood is taken from the circulation, dialyzed through a complex artificial membrane, and returned to the body. A high rate of blood flow is required to remove excess body fluid, exchange electrolytes, and remove noxious metabolites. To accomplish this, either an arteriovenous fistula is established surgically (by connecting an artery to a vein, usually in the upper limb, and requiring approximately six weeks to “mature”) and is cannulated each time the patient returns for dialysis, or a large-bore cannula is placed into the right atrium, through which blood can be aspirated and returned.
In the second method of dialysis, the peritoneum is used as the dialysis membrane. The large surface area of the peritoneal cavity is an ideal dialysis membrane for fluid and electrolyte exchange. To accomplish dialysis, a small tube is inserted through the abdominal wall and dialysis fluid is injected into the peritoneal cavity. Electrolytes and molecules are exchanged across the peritoneum between the fluid and blood. Once dialysis is completed, the fluid is drained.
Peritoneal spread of disease
The large surface area of the peritoneal cavity allows infection and malignant disease to spread easily throughout the abdomen (Fig. 4.55). If malignant cells enter the peritoneal cavity by direct invasion (e.g., from colon or ovarian cancer) spread may be rapid. Similarly, a surgeon excising a malignant tumor and releasing malignant cells into the peritoneal cavity may cause an appreciable worsening of the patient’s prognosis. Infection can also spread across the large surface area.
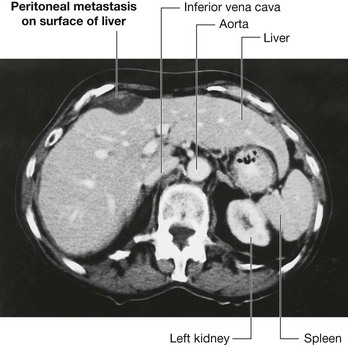
Fig. 4.55 Peritoneal metastasis on the surface of the liver. Computed tomogram in the axial plane of the upper abdomen.
The peritoneal cavity can also act as a barrier to, and container of, disease. Intra-abdominal infection therefore tends to remain below the diaphragm rather than spread into other body cavities.
A perforated bowel (e.g., caused by a perforated duodenal ulcer) often leads to the release of gas into the peritoneal cavity. This peritoneal gas can be easily visualized on an erect chest radiograph—gas can be demonstrated in extremely small amounts beneath the diaphragm. A patient with severe abdominal pain and subdiaphragmatic gas needs a laparotomy.
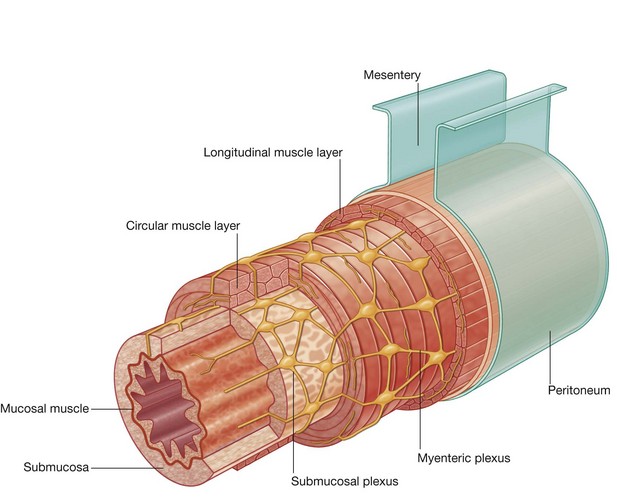
Omenta, mesenteries, and ligaments
Throughout the peritoneal cavity numerous peritoneal folds connect organs to each other or to the abdominal wall. These folds (omenta, mesenteries, and ligaments) develop from the original dorsal and ventral mesenteries, which suspend the developing gastrointestinal tract in the embryonic coelomic cavity. Some contain vessels and nerves supplying the viscera, while others help maintain the proper positioning of the viscera.
Omenta
The omenta consist of two layers of peritoneum, which pass from the stomach and the first part of the duodenum to other viscera. There are two:
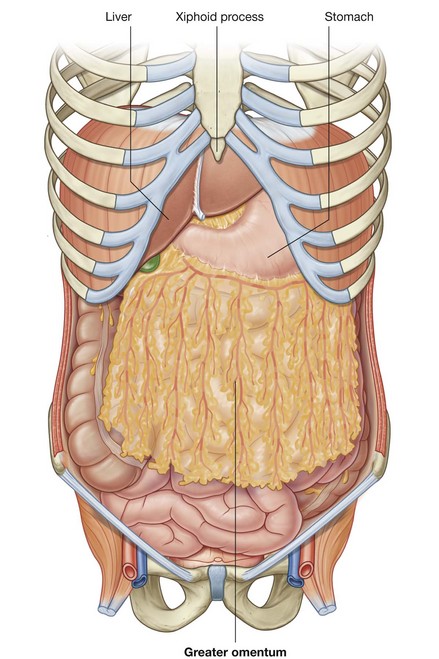
Greater omentum
The greater omentum is a large, apron-like, peritoneal fold that attaches to the greater curvature of the stomach and the first part of the duodenum (Fig. 4.56). It drapes inferiorly over the transverse colon and the coils of the jejunum and ileum (see Fig. 4.53). Turning posteriorly, it ascends to associate with, and become adherent to, the peritoneum on the superior surface of the transverse colon and the anterior layer of the transverse mesocolon before arriving at the posterior abdominal wall.
Usually a thin membrane, the greater omentum always contains an accumulation of fat, which may become substantial in some individuals. Additionally, there are two arteries and accompanying veins, the right and left gastro-omental vessels, between this double-layered peritoneal apron just inferior to the greater curvature of the stomach.
Lesser omentum
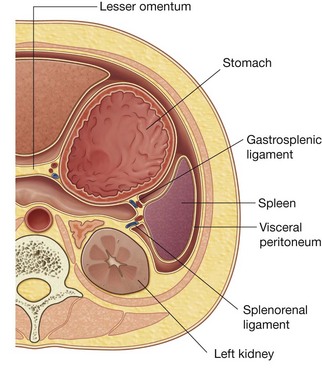
The other two-layered peritoneal omentum is the lesser omentum (Fig. 4.57). It extends from the lesser curvature of the stomach and the first part of the duodenum to the inferior surface of the liver (Figs. 4.53 and 4.57).
A thin membrane continuous with the peritoneal coverings of the anterior and posterior surfaces of the stomach and the first part of the duodenum, the lesser omentum is divided into:
The hepatoduodenal ligament ends laterally as a free margin and serves as the anterior border of the omental foramen (Fig. 4.54). Enclosed in this free edge are the hepatic artery proper, the bile duct, and the portal vein. Additionally, the right and left gastric vessels are between the layers of the lesser omentum near the lesser curvature of the stomach.
In the clinic
When a laparotomy is performed and the peritoneal cavity is opened, the first structure usually encountered is the greater omentum. This fatty double-layered vascular membrane hangs like an apron from the greater curvature of the stomach, drapes over the transverse colon, and lies freely suspended within the abdominal cavity. It is often referred to as the “policeman of the abdomen” because of its apparent ability to migrate to any inflamed area and wrap itself around the organ to wall off inflammation. When a part of bowel becomes inflamed, it ceases peristalsis. This aperistaltic area is referred to as a local paralytic ileus. The remaining noninflamed part of the bowel continues to move and “massages” the greater omentum to the region where there is no peristalsis. The localized inflammatory reaction spreads to the greater omentum, which then adheres to the diseased area of bowel.
The greater omentum is also an important site for metastatic tumor spread. Direct omental spread by a transcoelomic route is common for carcinoma of the ovary. As the metastases develop within the greater omentum, it becomes significantly thickened.
In computed tomography imaging and during laparotomy, the thickened omentum is referred to as an “omental cake.”
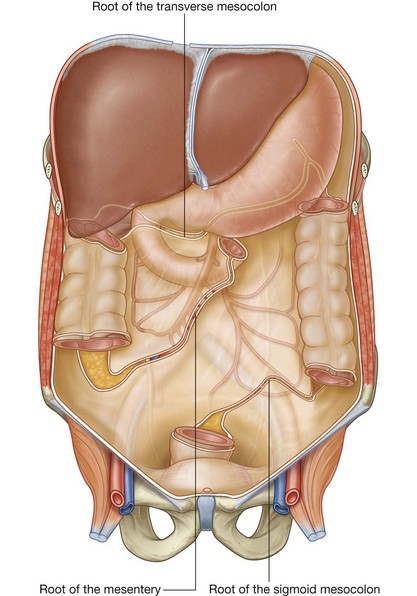
Mesenteries
Mesenteries are peritoneal folds that attach viscera to the posterior abdominal wall. They allow some movement and provide a conduit for vessels, nerves, and lymphatics to reach the viscera and include:
All of these are derivatives of the dorsal mesentery.
Mesentery
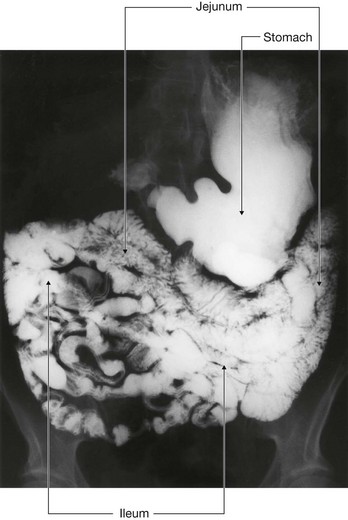
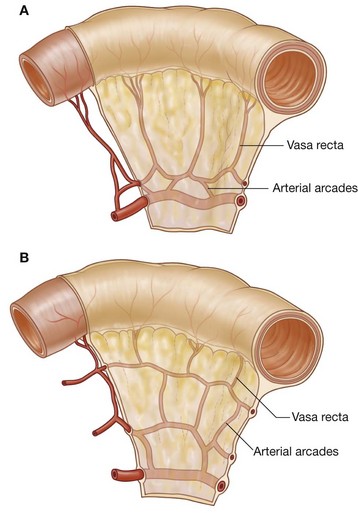
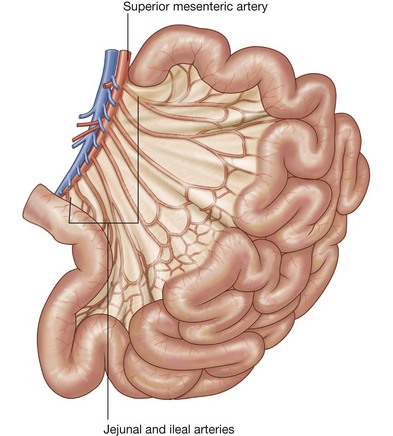
The mesentery is a large, fan-shaped, double-layered fold of peritoneum that connects the jejunum and ileum to the posterior abdominal wall (Fig. 4.58). Its superior attachment is at the duodenojejunal junction, just to the left of the upper lumbar part of the vertebral column. It passes obliquely downward and to the right, ending at the ileocecal junction near the upper border of the right sacro-iliac joint. In the fat between the two peritoneal layers of the mesentery are the arteries, veins, nerves, and lymphatics that supply the jejunum and ileum.
Transverse mesocolon
The transverse mesocolon is a fold of peritoneum that connects the transverse colon to the posterior abdominal wall (Fig. 4.58). Its two layers of peritoneum leave the posterior abdominal wall across the anterior surface of the head and body of the pancreas and pass outward to surround the transverse colon. Between its layers are the arteries, veins, nerve, and lymphatics related to the transverse colon. The anterior layer of the transverse mesocolon is adherent to the posterior layer of the greater omentum.
Sigmoid mesocolon
The sigmoid mesocolon is an inverted, V-shaped peritoneal fold that attaches the sigmoid colon to the abdominal wall (Fig. 4.58). The apex of the V is near the division of the left common iliac artery into its internal and external branches, with the left limb of the descending V along the medial border of the left psoas major muscle and the right limb descending into the pelvis to end at the level of vertebra SIII. The sigmoid and superior rectal vessels, along with the nerves and lymphatics associated with the sigmoid colon, pass through this peritoneal fold.
Ligaments
Peritoneal ligaments consist of two layers of peritoneum that connect two organs to each other or attach an organ to the body wall, and may form part of an omentum. They are usually named after the structures being connected. For example, the splenorenal ligament connects the left kidney to the spleen and the gastrophrenic ligament connects the stomach to the diaphragm.
Organs
Abdominal esophagus
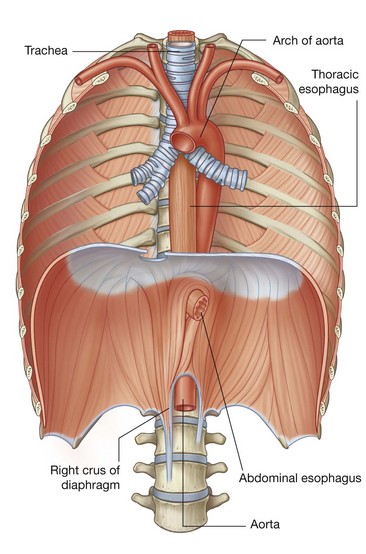
The abdominal esophagus represents the short distal part of the esophagus located in the abdominal cavity. Emerging through the right crus of the diaphragm, usually at the level of vertebra TX, it passes from the esophageal hiatus to the cardial orifice of the stomach just left of the midline (Fig. 4.59).
Associated with the esophagus, as it enters the abdominal cavity, are the anterior and posterior vagal trunks:
 the anterior vagal trunk consists of several smaller trunks whose fibers mostly come from the left vagus nerve; rotation of the gut during development moves these trunks to the anterior surface of the esophagus;
the anterior vagal trunk consists of several smaller trunks whose fibers mostly come from the left vagus nerve; rotation of the gut during development moves these trunks to the anterior surface of the esophagus; similarly, the posterior vagal trunk consists of a single trunk whose fibers mostly come from the right vagus nerve, and rotational changes during development move this trunk to the posterior surface of the esophagus.
similarly, the posterior vagal trunk consists of a single trunk whose fibers mostly come from the right vagus nerve, and rotational changes during development move this trunk to the posterior surface of the esophagus.The arterial supply to the abdominal esophagus (Fig. 4.60) includes:
Stomach
The stomach is the most dilated part of the gastrointestinal tract and has a J-like shape (Figs. 4.61 and 4.62). Positioned between the abdominal esophagus and the small intestine, the stomach is in the epigastric, umbilical, and left hypochondrium regions of the abdomen.
Fig. 4.62 Radiograph, using barium, showing the stomach and duodenum. A. Double contrast radiograph of the stomach. B. Double contrast radiograph showing the duodenal cap.
The stomach is divided into four regions:
 the pyloric part, which is divided into the pyloric antrum and pyloric canal and is the distal end of the stomach (Figs. 4.61 and 4.62B).
the pyloric part, which is divided into the pyloric antrum and pyloric canal and is the distal end of the stomach (Figs. 4.61 and 4.62B).The most distal portion of the pyloric part of the stomach is the pylorus (Fig. 4.61). It is marked on the surface of the organ by the pyloric constriction and contains a thickened ring of gastric circular muscle, the pyloric sphincter, that surrounds the distal opening of the stomach, the pyloric orifice. The pyloric orifice is just to the right of midline in a plane that passes through the lower border of vertebra LI (the transpyloric plane).
Other features of the stomach include:
 the greater curvature, which is a point of attachment for the gastrosplenic ligament and the greater omentum;
the greater curvature, which is a point of attachment for the gastrosplenic ligament and the greater omentum;The arterial supply to the stomach (Fig. 4.60) includes:
Small intestine
The small intestine is the longest part of the gastrointestinal tract and extends from the pyloric orifice of the stomach to the ileocecal fold. This hollow tube, which is approximately 6–7 m long with a narrowing diameter from beginning to end, consists of the duodenum, the jejunum, and the ileum.
Duodenum
The first part of the small intestine is the duodenum. This C-shaped structure, adjacent to the head of the pancreas, is 20–25 cm long and is above the level of the umbilicus; its lumen is the widest of the small intestine (Fig. 4.63). It is retroperitoneal except for its beginning, which is connected to the liver by the hepatoduodenal ligament, a part of the lesser omentum.
The duodenum is divided into four parts (Fig. 4.63).
 The superior part (first part) extends from the pyloric orifice of the stomach to the neck of the gallbladder, is just to the right of the body of vertebra LI, and passes anteriorly to the bile duct, gastroduodenal artery, portal vein, and inferior vena cava.
The superior part (first part) extends from the pyloric orifice of the stomach to the neck of the gallbladder, is just to the right of the body of vertebra LI, and passes anteriorly to the bile duct, gastroduodenal artery, portal vein, and inferior vena cava.
 The descending part (second part) of the duodenum is just to the right of midline and extends from the neck of the gallbladder to the lower border of vertebra LIII. Its anterior surface is crossed by the transverse colon, posterior to it is the right kidney, and medial to it is the head of the pancreas. This part of the duodenum contains the major duodenal papilla, which is the common entrance for the bile and pancreatic ducts, and the minor duodenal papilla, which is the entrance for the accessory pancreatic duct, and the junction of the foregut and the midgut just below the major duodenal papilla.
The descending part (second part) of the duodenum is just to the right of midline and extends from the neck of the gallbladder to the lower border of vertebra LIII. Its anterior surface is crossed by the transverse colon, posterior to it is the right kidney, and medial to it is the head of the pancreas. This part of the duodenum contains the major duodenal papilla, which is the common entrance for the bile and pancreatic ducts, and the minor duodenal papilla, which is the entrance for the accessory pancreatic duct, and the junction of the foregut and the midgut just below the major duodenal papilla. The inferior part (third part) of the duodenum is the longest section, crossing the inferior vena cava, the aorta, and the vertebral column (Figs. 4.62B and 4.63). It is crossed anteriorly by the superior mesenteric artery and vein.
The inferior part (third part) of the duodenum is the longest section, crossing the inferior vena cava, the aorta, and the vertebral column (Figs. 4.62B and 4.63). It is crossed anteriorly by the superior mesenteric artery and vein. The ascending part (fourth part) of the duodenum passes upward on, or to the left of, the aorta to approximately the upper border of vertebra LII and terminates at the duodenojejunal flexure.
The ascending part (fourth part) of the duodenum passes upward on, or to the left of, the aorta to approximately the upper border of vertebra LII and terminates at the duodenojejunal flexure.This duodenojejunal flexure is surrounded by a fold of peritoneum containing muscle fibers called the suspensory muscle (ligament) of duodenum (ligament of Treitz).
The arterial supply to the duodenum (Fig. 4.64) includes:
 duodenal branches from the anterior superior pancreaticoduodenal artery (from the gastroduodenal artery);
duodenal branches from the anterior superior pancreaticoduodenal artery (from the gastroduodenal artery); duodenal branches from the posterior superior pancreaticoduodenal artery (from the gastroduodenal artery);
duodenal branches from the posterior superior pancreaticoduodenal artery (from the gastroduodenal artery); duodenal branches from the anterior inferior pancreaticoduodenal artery (from the inferior pancreaticoduodenal artery—a branch of the superior mesenteric artery);
duodenal branches from the anterior inferior pancreaticoduodenal artery (from the inferior pancreaticoduodenal artery—a branch of the superior mesenteric artery);Jejunum
The jejunum and ileum make up the last two sections of the small intestine (Fig. 4.65). The jejunum represents the proximal two-fifths. It is mostly in the left upper quadrant of the abdomen and is larger in diameter and has a thicker wall than the ileum. Additionally, the inner mucosal lining of the jejunum is characterized by numerous prominent folds that circle the lumen (plicae circulares). The less prominent arterial arcades and longer vasa recta (straight arteries) compared to those of the ileum are a unique characteristic of the jejunum (Fig. 4.66).
The arterial supply to the jejunum includes jejunal arteries from the superior mesenteric artery.
Ileum
The ileum makes up the distal three-fifths of the small intestine and is mostly in the right lower quadrant. Compared to the jejunum, the ileum has thinner walls, fewer and less prominent mucosal folds (plicae circulares), shorter vasa recta, more mesenteric fat, and more arterial arcades (Fig. 4.66).
The ileum opens into the large intestine where the cecum and ascending colon join together. Two flaps projecting into the lumen of the large intestine (the ileocecal fold) surround the opening (Fig. 4.67). The flaps of the ileocecal fold come together at their end forming ridges. Musculature from the ileum continues into each flap, forming a sphincter. Possible functions of the ileocecal fold include preventing reflux from the cecum to the ileum, and regulating the passage of contents from the ileum to the cecum.
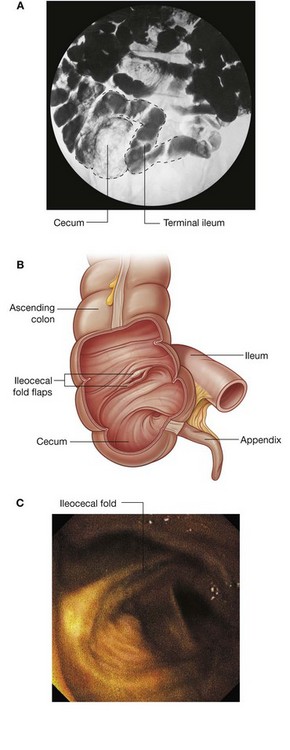
Fig. 4.67 Ileocecal junction. A. Radiograph showing ileocecal junction. B. Illustration showing ileocecal junction and the ileocecal fold. C. Endoscopic image of the ileocecal fold.
The arterial supply to the ileum (Fig. 4.68) includes:
In the clinic
Epithelial transition between the abdominal esophagus and stomach
The gastroesophageal junction is demarcated by a transition from one epithelial type to another epithelial type. In some people, the histological junction does not lie at the physiological gastroesophageal junction, but is in the lower one-third of the esophagus. This may predispose to esophageal ulceration, and is also associated with an increased risk of adenocarcinoma.
In the clinic
Duodenal ulceration
Duodenal ulcers usually occur in the superior part of the duodenum and are much less common than they were 50 years ago. At first, there was no treatment and patients died from hemorrhage or peritonitis. As surgical techniques developed, patients with duodenal ulcers were subjected to extensive upper gastrointestinal surgery to prevent ulcer recurrence and for some patients the treatment was dangerous. As knowledge and understanding of the mechanisms for acid secretion in the stomach increased, drugs were developed to block acid stimulation and secretion indirectly (histamine H2-receptor antagonists) and these have significantly reduced the morbidity and mortality rates of this disease. Pharmacological therapy can now directly inhibit the cells of the stomach that produce acid with, for example, proton pump inhibitors. Patients are also screened for the bacteria Helicobacter pylori, which when eradicated (by antibiotic treatment) significantly reduces duodenal ulcer formation.
Anatomically, duodenal ulcers tend to occur either anteriorly or posteriorly.
Posterior duodenal ulcers erode either directly onto the gastroduodenal artery or, more commonly, onto the posterior superior pancreaticoduodenal artery, which can produce torrential hemorrhage, which may be fatal in some patients. Treatment may involve extensive upper abdominal surgery with ligation of the vessels or by endovascular means whereby the radiologist may place a very fine catheter retrogradely from the femoral artery into the celiac artery. The common hepatic artery and the gastroduodenal artery are cannulated and the bleeding area may be blocked using small coils, which stem the flow of blood.
Anterior duodenal ulcers erode into the peritoneal cavity, causing peritonitis. This intense inflammatory reaction and the local ileus promote adhesion of the greater omentum, which attempts to seal off the perforation. The stomach and duodenum usually contain considerable amounts of gas, which enters the peritoneal cavity and can be observed on a chest radiograph of an erect patient as subdiaphragmatic gas. In most instances, treatment for the ulcer is surgical.
In the clinic
Examination of the upper gastrointestinal tract
It is often necessary to examine the esophagus, stomach, duodenum, and proximal jejunum for disease. After taking an appropriate history and examining the patient, most physicians arrange a series of simple blood tests to look for bleeding, inflammation, and tumors. The next steps in the investigation assess the three components of any loop of bowel, namely, the lumen, the wall, and masses extrinsic to the bowel, which may compress or erode into it.
Examination of the bowel Lumen
Barium sulfate solutions may be swallowed by the patient and can be visualized using an X-ray fluoroscopy unit. The lumen can be examined for masses (e.g., polyps and tumors) and peristaltic waves can be assessed. Patients may also be given carbon dioxide–releasing granules to fill the stomach so that the barium thinly coats the mucosa, resulting in images displaying fine mucosal detail. These tests are relatively simple and can be used to image the esophagus, stomach, duodenum, and small bowel.
Examination of the bowel wall and extrinsic masses
Endoscopy is a minimally invasive diagnostic medical procedure that can be used to assess the interior surfaces of an organ by inserting a tube into the body. The instrument is typically made of a flexible plastic material through which a light source and eye piece are attached at one end. Some systems allow passage of small instruments through the main bore of the endoscope to obtain biopsies and to also undertake small procedures (e.g., the removal of polyps).
In gastrointestinal and abdominal medicine an endoscope is used to assess the esophagus, stomach, duodenum and proximal small bowel (Figs. 4.69-4.72). The tube is swallowed by the patient under light sedation and is extremely well tolerated.
Fig. 4.69 The endoscope is a flexible plastic tube that can be controlled from the proximal end. Through a side portal various devices can be inserted, which run through the endoscope and can be used to obtain biopsies and to perform small endoluminal surgery (e.g., excision of polyps).
Fig. 4.70 Endoscopic images of the gastroesophageal junction. A. Normal. B. Esophageal cancer at esophageal junction.
Assessment of the colon is performed by passage of the tube through the anus and into the rectum. The whole of the colon can be readily assessed; biopsies and stents can also be performed using this device.
In the clinic
Meckel’s diverticulum
A Meckel’s diverticulum (Fig. 4.73) is the remnant of the proximal part of the yolk stalk (vitelline duct), which extends into the umbilical cord in the embryo and lies on the antimesenteric border of the ileum. Although it is an uncommon finding (occurring in approximately 2% of the population), it is always important to consider the diagnosis of Meckel’s diverticulum because it does produce symptoms in a small number of patients. Typical findings include hemorrhage, intussusception, diverticulitis, ulceration, and obstruction.
In the clinic
Computed tomography (CT) scanning and magnetic resonance imaging (MRI)
These imaging techniques can provide important information about the wall of the bowel that may not be obtained from barium or endoscopic studies. Thickening of the wall may indicate inflammatory change or tumor and is always regarded with suspicion. If a tumor is demonstrated, the locoregional spread can be assessed, along with lymphadenopathy and metastatic spread.
In the clinic
Carcinoma of the stomach
Carcinoma of the stomach is a common gastrointestinal malignancy. Chronic gastric inflammation (gastritis), pernicious anemia, and polyps predispose to the development of this aggressive cancer, which is usually not diagnosed until late in the course of the disease. Symptoms include vague epigastric pain, early fullness with eating, bleeding leading to chronic anemia, and obstruction.
The diagnosis may be made using barium and conventional radiology or endoscopy, which allows a biopsy to be obtained at the same time. Ultrasound scanning is used to check the liver for metastatic spread, and, if negative, computed tomography is carried out to assess for surgical resectability. If carcinoma of the stomach is diagnosed early, a curative surgical resection is possible. However, because most patients don’t seek treatment until late in the disease, the overall 5-year survival rate is between 5% and 20%, with a mean survival time of between 5 and 8 months.
Large intestine
The large intestine extends from the distal end of the ileum to the anus, a distance of approximately 1.5 m in adults. It absorbs fluids and salts from the gut contents, thus forming feces, and consists of the cecum, appendix, colon, rectum, and anal canal (Figs. 4.74 and 4.75).
Beginning in the right groin as the cecum, with its associated appendix, the large intestine continues upward as the ascending colon through the right flank and into the right hypochondrium (Fig. 4.76). Just below the liver, it bends to the left, forming the right colic flexure (hepatic flexure), and crosses the abdomen as the transverse colon to the left hypochondrium. At this position, just below the spleen, the large intestine bends downward, forming the left colic flexure (splenic flexure), and continues as the descending colon through the left flank and into the left groin.
It enters the upper part of the pelvic cavity as the sigmoid colon, continues on the posterior wall of the pelvic cavity as the rectum, and terminates as the anal canal.
The general characteristics of most of the large intestine (Fig. 4.74) are:
 the segregation of longitudinal muscle in its walls into three narrow bands (the taeniae coli), which are primarily observed in the cecum and colon and less visible in the rectum; and
the segregation of longitudinal muscle in its walls into three narrow bands (the taeniae coli), which are primarily observed in the cecum and colon and less visible in the rectum; andCecum and appendix
The cecum is the first part of the large intestine (Fig. 4.77). It is inferior to the ileocecal opening and in the right iliac fossa. It is an intraperitoneal structure because of its mobility not because of its suspension by a mesentery.
The cecum is continuous with the ascending colon at the entrance of the ileum and is usually in contact with the anterior abdominal wall. It may cross the pelvic brim to lie in the true pelvis. The appendix is attached to the posteromedial wall of the cecum, just inferior to the end of the ileum (Fig. 4.77).
The appendix is a narrow, hollow, blind-ended tube connected to the cecum. It has large aggregations of lymphoid tissue in its walls and is suspended from the terminal ileum by the mesoappendix (Fig. 4.78), which contains the appendicular vessels. Its point of attachment to the cecum is consistent with the highly visible free taenia leading directly to the base of the appendix, but the location of the rest of the appendix varies considerably (Fig. 4.79). It may be:
 posterior to the cecum or the lower ascending colon, or both, in a retrocecal or retrocolic position;
posterior to the cecum or the lower ascending colon, or both, in a retrocecal or retrocolic position; anterior to the terminal ileum, possibly contacting the body wall, in a pre-ileal position or posterior to the terminal ileum in a postileal position.
anterior to the terminal ileum, possibly contacting the body wall, in a pre-ileal position or posterior to the terminal ileum in a postileal position.The surface projection of the base of the appendix is at the junction of the lateral and middle one-third of a line from the anterior superior iliac spine to the umbilicus (McBurney’s point). People with appendicular problems may describe pain near this location.
The arterial supply to the cecum and appendix (Fig. 4.80) includes:
In the clinic
Appendicitis
Acute appendicitis is an abdominal emergency. It usually occurs when the appendix is obstructed by either a fecalith or enlargement of the lymphoid nodules. Within the obstructed appendix, bacteria proliferate and invade the appendix wall, which becomes damaged by pressure necrosis. In some instances, this may resolve spontaneously; in other cases, inflammatory change (Fig. 4.81) continues and perforation ensues, which may lead to localized or generalized peritonitis.
Most patients with acute appendicitis have localized tenderness in the right groin. Initially, the pain begins as a central, periumbilical, colicky type of pain, which tends to come and go. After 6–10 hours, the pain tends to localize in the right iliac fossa and becomes constant. Patients may develop a temperature, nausea, and vomiting. The etiology of the pain for appendicitis is described in Chapter 1 on p. 53.
Colon
The colon extends superiorly from the cecum and consists of the ascending, transverse, descending, and sigmoid colon (Fig. 4.82). Its ascending and descending segments are (secondarily) retroperitoneal and its transverse and sigmoid segments are intraperitoneal.
At the junction of the ascending and transverse colon is the right colic flexure, which is just inferior to the right lobe of the liver (Fig. 4.83). A similar, but more acute bend (the left colic flexure) occurs at the junction of the transverse and descending colon. This bend is just inferior to the spleen, higher and more posterior than the right colic flexure, and is attached to the diaphragm by the phrenicocolic ligament.
Immediately lateral to the ascending and descending colons are the right and left paracolic gutters (Fig. 4.82). These depressions are formed between the lateral margins of the ascending and descending colon and the posterolateral abdominal wall and are gutters through which material can pass from one region of the peritoneal cavity to another.
Because major vessels and lymphatics are on the medial or posteromedial sides of the ascending and descending colon, a relatively blood-free mobilization of the ascending and descending colon is possible by cutting the peritoneum along these lateral paracolic gutters.
The final segment of the colon (the sigmoid colon) begins above the pelvic inlet and extends to the level of vertebra SIII, where it is continuous with the rectum (Fig. 4.82). This S-shaped structure is quite mobile except at its beginning, where it continues from the descending colon, and at its end, where it continues as the rectum. Between these points, it is suspended by the sigmoid mesocolon.
The arterial supply to the ascending colon (Fig. 4.84) includes:
The arterial supply to the transverse colon (Fig. 4.84) includes:
The arterial supply to the descending colon (Fig. 4.84) includes the left colic artery from the inferior mesenteric artery.
The arterial supply to the sigmoid colon (Fig. 4.84) includes sigmoidal arteries from the inferior mesenteric artery.
Rectum and anal canal
Extending from the sigmoid colon is the rectum (Fig. 4.85). The rectosigmoid junction is usually described as being at the level of vertebra SIII or at the end of the sigmoid mesocolon because the rectum is a retroperitoneal structure.
The anal canal is the continuation of the large intestine inferior to the rectum.
The arterial supply to the rectum and anal canal (Fig. 4.86) includes:
In the clinic
Congenital disorders of the gastrointestinal tract
The normal positions of the abdominal viscera result from a complex series of rotations that the gut tube undergoes and from the growth of the abdominal cavity to accommodate changes in the size of the developing organs. A number of developmental anomalies can occur during gut development, many of which appear in the neonate or infant, and some of which are surgical emergencies. Occasionally, such disorders are diagnosed only in adults.
Malrotation and midgut volvulus
Malrotation is incomplete rotation and fixation of the midgut after it has passed from the umbilical sac and returned to the abdominal coelom (Figs. 4.87 and 4.88). The proximal attachment of the small bowel mesentery begins at the suspensory muscle of duodenum (ligament of Treitz), which determines the position of the duodenojejunal junction. The mesentery of the small bowel ends at the level of the ileocecal junction in the right lower quadrant. This long line of fixation of the mesentery prevents accidental twists of the gut.
Fig. 4.87 Small bowel malrotation and volvulus. Radiograph of stomach, duodenum, and upper jejunum using barium.
If the duodenojejunal flexure or the cecum does not end up in its usual site, the origin of the small bowel mesentery shortens, which permits twisting of the small bowel around the axis of the superior mesenteric artery. Twisting of the bowel, in general, is termed volvulus. Volvulus of the small bowel may lead to a reduction of blood flow and infarction.
In some patients, the cecum ends up in the midabdomen. From the cecum and the right side of the colon a series of peritoneal folds (Ladd’s bands) develop that extend to the right undersurface of the liver and compress the duodenum. A small bowel volvulus may then occur as well as duodenal obstruction. Emergency surgery may be necessary to divide the bands.
In the clinic
Bowel obstruction
A bowel obstruction can be either functional or due to a true obstruction. Mechanical obstruction is caused by an intraluminal, mural or extrinsic mass which can be secondary to a foreign body, obstructing tumor in the wall, or extrinsic compression from an adhesion, or embryological band (Fig. 4.89).
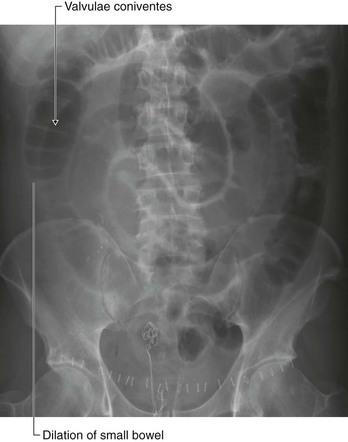
Fig. 4.89 This radiograph of the abdomen, anterior-posterior view, demonstrates a number of dilated loops of small bowel. Small bowel can be identified by the valvulae coniventes that pass from wall to wall as indicated. The large bowel is not dilated. The cause of the small bowel dilatation is an adhesion after pelvic surgery.
A functional obstruction is usually due to an inability of the bowel to peristalse, which again has a number of causes, and most frequently is a postsurgical state due to excessive intraoperative bowel handling. Other causes may well include abnormality of electrolytes (e.g., sodium and potassium) rendering the bowel paralyzed until correction has occurred.
The signs and symptoms of obstruction depend on the level at which the obstruction has occurred. The primary symptom is central abdominal, intermittent, colicky pain as the peristaltic waves try to overcome the obstruction. Abdominal distention will occur if it is a low obstruction (distal), allowing more proximal loops of bowel to fill with fluid. A high obstruction (in the proximal small bowel) may not produce abdominal distention.
Vomiting and absolute constipation, including the inability to pass flatus, will ensue.
Early diagnosis is important because considerable fluid and electrolytes enter the bowel lumen and fail to be reabsorbed, which produces dehydration and electrolyte abnormalities. Furthermore, the bowel continues to distend, compromising the blood supply within the bowel wall, which may lead to ischemia and perforation. The symptoms and signs are variable and depend on the level of obstruction.
Small bowel obstruction is typically caused by adhesions following previous surgery, and history should always be sought for any operations or abdominal interventions (e.g., previous appendectomy). Other causes include bowel passing into hernias (e.g., inguinal), and bowel twisting on its own mesentery (volvulus). Examination of hernial orifices is mandatory in patients with bowel obstruction.
Large bowel obstruction is commonly caused by a tumor. Other potential causes include hernias and inflammatory diverticular disease of the sigmoid colon.
The treatment is intravenous replacement of fluid and electrolytes, analgesia, and relief of obstruction. The passage of a nasogastric tube allows aspiration of fluid from the stomach. In many instances, small bowel obstruction, typically secondary to adhesions, will settle with nonoperative management. Large bowel obstruction may require an urgent operation to remove the obstructing lesion, or a temporary bypass procedure (e.g., defunctioning colostomy) (Fig. 4.90).
In the clinic
Diverticular disease
Diverticular disease is the development of multiple colonic diverticula, predominantly throughout the sigmoid colon, though the whole colon may be affected (Fig. 4.91). The sigmoid colon has the smallest diameter of any portion of the colon and is therefore the site where intraluminal pressure is potentially the highest. Poor dietary fiber intake and obesity are also linked to diverticular disease.
Fig. 4.91 This double-contrast barium enema demonstrates numerous small outpouchings throughout the distal large bowel predominantly within the descending colon and the sigmoid colon. These small outpouchings are diverticula and in most instances remain quiescent.
The presence of multiple diverticula does not necessarily mean the patient requires any treatment. Moreover, many patients have no other symptoms or signs.
Patients tend to develop symptoms and signs when the neck of the diverticulum becomes obstructed by feces and becomes infected. Inflammation may spread along the wall, causing abdominal pain. When the sigmoid colon becomes inflamed (diverticulitis) abdominal pain and fever ensue.
Because of the anatomical position of the sigmoid colon there are a number of complications that may occur. The diverticula can perforate to form an abscess in the pelvis. The inflammation may produce an inflammatory mass, obstructing the left ureter. Inflammation may also spread to the bladder, producing a fistula between the sigmoid colon and the bladder. In these circumstances patients may develop a urinary tract infection and rarely have fecal material and gas passing per urethra.
The diagnosis is based upon clinical examination and often CT scanning. In the first instance, patients will be treated with antibiotic therapy; however, a surgical resection may be necessary if symptoms persist.
In the clinic
Ostomies
It is occasionally necessary to surgically externalize bowel to the anterior abdominal wall. Externalization of bowel plays an important role in patient management. These extra-anatomical bypass procedures use our anatomical knowledge and in many instances are life saving.
Gastrostomy is performed when the stomach is attached to the anterior abdominal wall and a tube is placed through the skin into the stomach. Typically this is performed to feed the patient when it is impossible to take food and fluid orally (e.g., complex head and neck cancer). The procedure can be performed either surgically or through a direct needlestick puncture under sedation in the anterior abdominal wall.
Similarly the jejunum is brought to the anterior abdominal wall and fixed. The jejunostomy is used as a site where a feeding tube is placed through the anterior abdominal wall into the proximal efferent small bowel.
An ileostomy is performed when small bowel contents need to be diverted from the distal bowel. An ileostomy is often performed to protect a distal surgical anastomosis, such as in the colon to allow healing after surgery.
There are a number of instances when a colostomy may be necessary. In many circumstances it is performed to protect the distal large bowel after surgery. A further indication would include large bowel obstruction with imminent perforation wherein a colostomy allows decompression of the bowel and its contents. This is a safe and temporizing procedure performed when the patient is too unwell for extensive bowel surgery. It is relatively straightforward and carries reduced risk preventing significant morbidity and mortality.
An end colostomy is necessary when the patient has undergone a surgical resection of the rectum and anus (typically for cancer).
An ileal conduit is an extra-anatomical procedure and is performed after resection of the bladder for tumor. In this situation a short segment of small bowel is identified. The bowel is divided twice to produce a 20-cm segment of small bowel on its own mesentery. This isolated segment of bowel is used as a conduit. The remaining bowel is joined together. The proximal end is anastomosed to the ureters and distal end to the anterior abdominal wall. Hence, urine passes from the kidneys into the ureters and through the short segment of small bowel to the anterior abdominal wall.
When patients have either an ileostomy, colostomy, or ileal conduit it is necessary for them to fix a collecting bag onto the anterior abdominal wall. Contrary to one’s initial thoughts these bags are tolerated extremely well by most patients and allow patients to live a near normal and healthy life.
Liver
The liver is the largest visceral organ in the body and is primarily in the right hypochondrium and epigastric region, extending into the left hypochondrium (or in the right upper quadrant, extending into the left upper quadrant) (Fig. 4.92).
Surfaces of the liver include:
Diaphragmatic surface
The diaphragmatic surface of the liver, which is smooth and domed, lies against the inferior surface of the diaphragm (Fig. 4.94). Associated with it are the subphrenic and hepatorenal recesses (Fig. 4.93):
 the subphrenic recess separates the diaphragmatic surface of the liver from the diaphragm and is divided into right and left areas by the falciform ligament, a structure derived from the ventral mesentery in the embryo;
the subphrenic recess separates the diaphragmatic surface of the liver from the diaphragm and is divided into right and left areas by the falciform ligament, a structure derived from the ventral mesentery in the embryo; the hepatorenal recess is a part of the peritoneal cavity on the right side between the liver and the right kidney and right suprarenal gland.
the hepatorenal recess is a part of the peritoneal cavity on the right side between the liver and the right kidney and right suprarenal gland.The subphrenic and hepatorenal recesses are continuous anteriorly.
Visceral surface
The visceral surface of the liver is covered with visceral peritoneum except in the fossa for the gallbladder and at the porta hepatis (gateway to the liver; Fig. 4.95), and structures related to it include the following (Fig. 4.96):
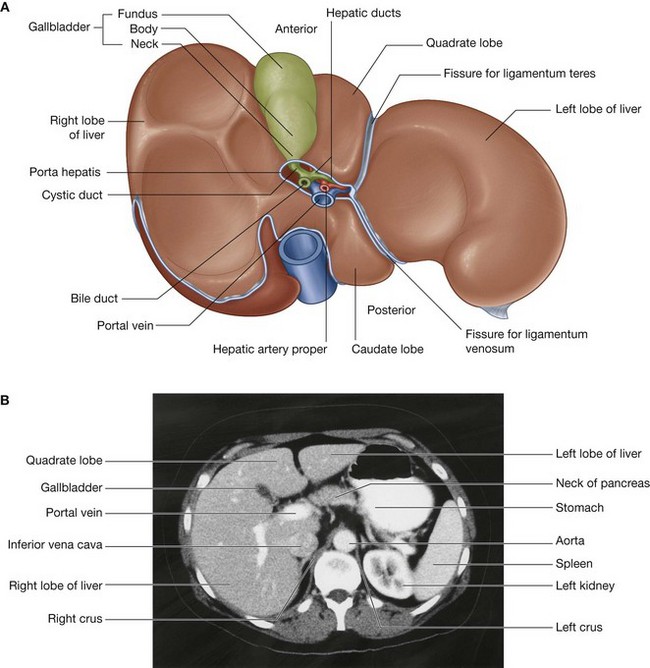
Fig. 4.95 Visceral surface of the liver. A. Illustration. B. Abdominal computed tomogram, with contrast, in the axial plane.
The porta hepatis serves as the point of entry into the liver for the hepatic arteries and the portal vein, and the exit point for the hepatic ducts (Fig. 4.95).
Associated ligaments
The liver is attached to the anterior abdominal wall by the falciform ligament and, except for a small area of the liver against the diaphragm (the bare area), the liver is almost completely surrounded by visceral peritoneum (Fig. 4.96). Additional folds of peritoneum connect the liver to the stomach (hepatogastric ligament), the duodenum (hepatoduodenal ligament), and the diaphragm (right and left triangular ligaments and anterior and posterior coronary ligaments).
The bare area of the liver is a part of the liver on the diaphragmatic surface where there is no intervening peritoneum between the liver and the diaphragm (Fig. 4.96):
 the anterior boundary of the bare area is indicated by a reflection of peritoneum—the anterior coronary ligament;
the anterior boundary of the bare area is indicated by a reflection of peritoneum—the anterior coronary ligament;Lobes
The liver is divided into right and left lobes by fossae for the gallbladder and the inferior vena cava (Fig. 4.95). The right lobe of liver is the largest lobe, whereas the left lobe of liver is smaller. The quadrate and caudate lobes are described as arising from the right lobe of liver, but functionally are distinct.
 The quadrate lobe is visible on the anterior part of the visceral surface of the liver and is bounded on the left by the fissure for ligamentum teres and on the right by the fossa for the gallbladder. Functionally it is related to the left lobe of the liver.
The quadrate lobe is visible on the anterior part of the visceral surface of the liver and is bounded on the left by the fissure for ligamentum teres and on the right by the fossa for the gallbladder. Functionally it is related to the left lobe of the liver. The caudate lobe is visible on the posterior part of the visceral surface of the liver. It is bounded on the left by the fissure for the ligamentum venosum and on the right by the groove for the inferior vena cava. Functionally, it is separate from the right and the left lobes of the liver.
The caudate lobe is visible on the posterior part of the visceral surface of the liver. It is bounded on the left by the fissure for the ligamentum venosum and on the right by the groove for the inferior vena cava. Functionally, it is separate from the right and the left lobes of the liver.Gallbladder
The gallbladder is a pear-shaped sac lying on the visceral surface of the right lobe of the liver in a fossa between the right and quadrate lobes (Fig. 4.95). It has:
 a major part in the fossa (body of gallbladder), which may be against the transverse colon and the superior part of the duodenum; and
a major part in the fossa (body of gallbladder), which may be against the transverse colon and the superior part of the duodenum; andThe arterial supply to the gallbladder (Fig. 4.97) is the cystic artery from the right hepatic artery (a branch of the hepatic artery proper).
The gallbladder receives, concentrates, and stores bile from the liver.
Pancreas
The pancreas lies mostly posterior to the stomach (Figs. 4.98 and 4.99). It extends across the posterior abdominal wall from the duodenum, on the right, to the spleen, on the left.
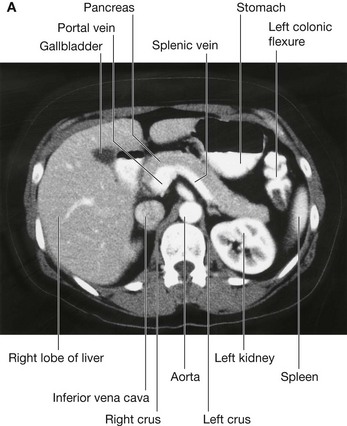
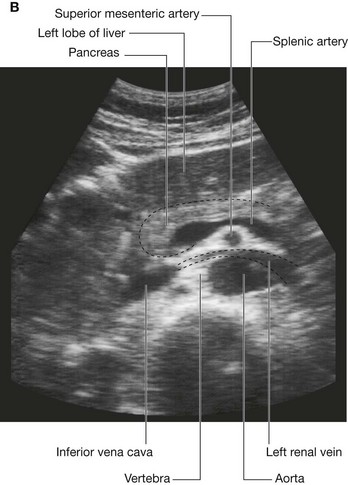
Fig. 4.99 Abdominal images. A. Abdominal computed tomogram, with contrast, in the axial plane. B. Abdominal ultrasound scan.
The pancreas is (secondarily) retroperitoneal except for a small part of its tail and consists of a head, uncinate process, neck, body, and tail.
 Projecting from the lower part of the head is the uncinate process, which passes posterior to the superior mesenteric vessels.
Projecting from the lower part of the head is the uncinate process, which passes posterior to the superior mesenteric vessels. The neck of pancreas is anterior to the superior mesenteric vessels. Posterior to the neck of the pancreas, the superior mesenteric and the splenic veins join to form the portal vein.
The neck of pancreas is anterior to the superior mesenteric vessels. Posterior to the neck of the pancreas, the superior mesenteric and the splenic veins join to form the portal vein.The pancreatic duct begins in the tail of the pancreas (Fig. 4.100). It passes to the right through the body of the pancreas and, after entering the head of the pancreas, turns inferiorly. In the lower part of the head of pancreas, the pancreatic duct joins the bile duct. The joining of these two structures forms the hepatopancreatic ampulla (ampulla of Vater), which enters the descending (second) part of the duodenum at the major duodenal papilla. Surrounding the ampulla is the sphincter of ampulla (sphincter of Oddi), which is a collection of smooth muscle.
The accessory pancreatic duct empties into the duodenum just above the major duodenal papilla at the minor duodenal papilla (Fig. 4.100). If the accessory duct is followed from the minor papilla into the head of the pancreas, a branch point is discovered:
 one branch continues to the left, through the head of the pancreas, and may connect with the pancreatic duct at the point where it turns inferiorly;
one branch continues to the left, through the head of the pancreas, and may connect with the pancreatic duct at the point where it turns inferiorly; a second branch descends into the lower part of the head of pancreas, anterior to the pancreatic duct, and ends in the uncinate process.
a second branch descends into the lower part of the head of pancreas, anterior to the pancreatic duct, and ends in the uncinate process.The main and accessory pancreatic ducts usually communicate with each other. The presence of these two ducts reflects the embryological origin of the pancreas from dorsal and ventral buds from the foregut.
The arterial supply to the pancreas (Fig. 4.101) includes the:
 anterior inferior pancreaticoduodenal artery from the inferior pancreaticoduodenal artery (a branch of the superior mesenteric artery); and
anterior inferior pancreaticoduodenal artery from the inferior pancreaticoduodenal artery (a branch of the superior mesenteric artery); and posterior inferior pancreaticoduodenal artery from the inferior pancreaticoduodenal artery (a branch of the superior mesenteric artery).
posterior inferior pancreaticoduodenal artery from the inferior pancreaticoduodenal artery (a branch of the superior mesenteric artery).In the clinic
Annular pancreas
The pancreas develops from ventral and dorsal buds from the foregut. The dorsal bud forms most of the head, neck, and body of pancreas. The ventral bud rotates around the bile duct to form part of the head and the uncinate process. If the ventral bud splits (becomes bifid) the two segments may encircle the duodenum. The duodenum is therefore constricted and may even undergo atresia, and be absent at birth because of developmental problems. After birth, the child may fail to thrive and vomit due to poor gastric emptying.
Sometimes annular pancreas is diagnosed in utero by ultrasound scanning. The obstruction of the duodenum may prevent the fetus from swallowing enough amniotic fluid, which may increase the overall volume of amniotic fluid in the amniotic sac surrounding the fetus (polyhydramnios).
Duct system for bile
The duct system for the passage of bile extends from the liver, connects with the gallbladder, and empties into the descending part of the duodenum (Fig. 4.102). The coalescence of ducts begins in the liver parenchyma and continues until the right and left hepatic ducts are formed. These drain the respective lobes of the liver.
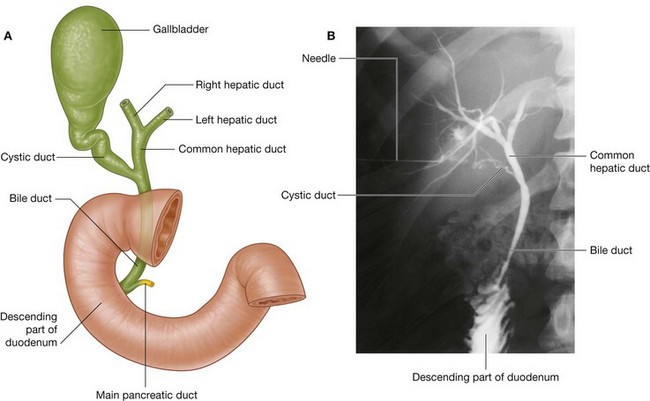
Fig. 4.102 Bile drainage. A. Duct system for passage of bile. B. Percutaneous transhepatic cholangiogram demonstrating the bile duct system.
The two hepatic ducts combine to form the common hepatic duct, which runs, near the liver, with the hepatic artery proper and portal vein in the free margin of the lesser omentum.
As the common hepatic duct continues to descend, it is joined by the cystic duct from the gallbladder. This completes the formation of the bile duct. At this point, the bile duct lies to the right of the hepatic artery proper and usually to the right of, and anterior to, the portal vein in the free margin of the lesser omentum. The omental foramen is posterior to these structures at this point.
The bile duct continues to descend, passing posteriorly to the superior part of the duodenum before joining with the pancreatic duct to enter the descending part of the duodenum at the major duodenal papilla (Fig. 4.102).
Spleen
The spleen develops as part of the vascular system in the part of the dorsal mesentery that suspends the developing stomach from the body wall. In the adult, the spleen lies against the diaphragm, in the area of rib IX to rib X (Fig. 4.103). It is therefore in the left upper quadrant, or left hypochondrium, of the abdomen.
The spleen is connected to the:
 greater curvature of the stomach by the gastrosplenic ligament, which contains the short gastric and gastro-omental vessels; and
greater curvature of the stomach by the gastrosplenic ligament, which contains the short gastric and gastro-omental vessels; andBoth these ligaments are parts of the greater omentum.
The spleen is surrounded by visceral peritoneum except in the area of the hilum on the medial surface of the spleen (Fig. 4.105). The splenic hilum is the entry point for the splenic vessels and occasionally the tail of the pancreas reaches this area.
The arterial supply to the spleen (Fig. 4.106) is the splenic artery from the celiac trunk.
In the clinic
Segmental anatomy of the liver
For many years the segmental anatomy of the liver was of little importance. However, since the development of liver resection surgery, the size, shape, and segmental anatomy of the liver has become clinically important, especially with regard to liver resection for metastatic disease. Indeed, with detailed knowledge of the segments, curative surgery can be performed in patients with tumor metastases.
The liver is divided by the principal plane which divides the organ into halves of approximately equal size. This imaginary line is defined by a parasagittal line that passes through the gallbladder fossa to the inferior vena cava. It is in this plane that the middle hepatic vein is found. Importantly, the principal plane divides the left half of the liver from the right half. The lobes of the liver are unequal in size and bear only little relevance to operative anatomy.
The traditional eight segment anatomy of the liver relates to the hepatic arterial, portal, and biliary drainage of these segments (Fig. 4.107).
Fig. 4.107 Division of the liver into segments based upon the distributions of the bile ducts and hepatic vessels (Couinaud’s segments).
The caudate lobe is defined as segment I, the remaining segments are numbered in a clockwise fashion up to segment VIII. The features are extremely consistent between individuals.
From a surgical perspective a right hepatectomy would involve division of the liver in the principal plane in which segments V, VI, VII, and VIII would be removed, leaving segments I, II, III, and IV.
In the clinic
Gallstones
Gallstones are present in approximately 10% of people over the age of 40 and are more common in women. They consist of a variety of components, but are predominantly a mixture of cholesterol and bile pigment. They may undergo calcification, which can be demonstrated on plain radiographs. Gallstones may be visualized incidentally as part of a routine abdominal ultrasound scan (Fig. 4.108) or on a plain radiograph.
From time to time, gallstones impact in the region of Hartmann’s pouch, which is a bulbous region of the neck of the gallbladder. When the gallstone lodges in this area, the gallbladder cannot empty normally and contractions of the gallbladder wall produce severe pain. If this persists, a cholecystectomy (removal of gallbladder) may be necessary.
Sometimes the gallbladder may become inflamed (cholecystitis). If the inflammation involves the related parietal peritoneum of the diaphragm, pain may not only occur in the right upper quadrant of the abdomen but may also be referred to the shoulder on the right side. This referred pain is due to the innervation of the visceral peritoneum of the diaphragm by spinal cord levels (C3 to C5) that also innervate skin over the shoulder. In this case, one somatic sensory region of low sensory output (diaphragm) is referred to another somatic sensory region of high sensory output (dermatomes).
From time to time, small gallstones pass into the bile duct and are trapped in the region of the sphincter of the ampulla, which obstructs the flow of bile into the duodenum. This, in turn, produces jaundice.
In the clinic
Jaundice
Jaundice is a yellow discoloration of the skin caused by excess bile pigment (bilirubin) within the plasma. The yellow color is best appreciated by looking at the normally white sclerae of the eyes, which turn yellow.
The extent of the elevation of the bile pigments and the duration for which they have been elevated account for the severity of jaundice.
Simplified explanation to understanding the types of jaundice and their anatomical causes
When red blood cells are destroyed by the reticuloendothelial system the iron from the hemoglobin molecule is recycled, whereas the porphyrin ring (globin) compounds are broken down to form fat-soluble bilirubin. On reaching the liver via the blood stream the fat-soluble bilirubin is converted to a water-soluble form of bilirubin. This water-soluble bilirubin is then secreted into the biliary tree and then in turn into the bowel where it forms the dark color of the stool.
Prehepatic jaundice
This type of jaundice is usually produced by conditions where there is an excessive breakdown of red blood cells (e.g., in incompatible blood transfusion and hemolytic anemia).
In the clinic
Spleen disorders
From a clinical point of view, there are two main categories of spleen disorders: rupture and enlargement.
Splenic rupture
This tends to occur when there is localized trauma to the left upper quadrant. It may be associated with left lower rib fractures. Because the spleen has such an extremely thin capsule it is susceptible to injury even when there is no damage to surrounding structures, and because the spleen is highly vascular, when ruptured, it bleeds profusely into the peritoneal cavity. Splenic rupture should always be suspected with blunt abdominal injury. Current treatments preserve as much of the spleen as possible, but some patients require splenectomy.
Arterial supply
The abdominal aorta begins at the aortic hiatus of the diaphragm, anterior to the lower border of vertebra TXII (Fig. 4.109). It descends through the abdomen, anterior to the vertebral bodies, and by the time it ends at the level of vertebra LIV it is slightly to the left of midline. The terminal branches of the abdominal aorta are the two common iliac arteries.
Anterior branches of the abdominal aorta
The abdominal aorta has anterior, lateral, and posterior branches as it passes through the abdominal cavity. The three anterior branches supply the gastrointestinal viscera: the celiac trunk and the superior mesenteric and inferior mesenteric arteries (Fig. 4.109).
The primitive gut tube can be divided into foregut, midgut, and hindgut regions. The boundaries of these regions are directly related to the areas of distribution of the three anterior branches of the abdominal aorta (Fig. 4.110).
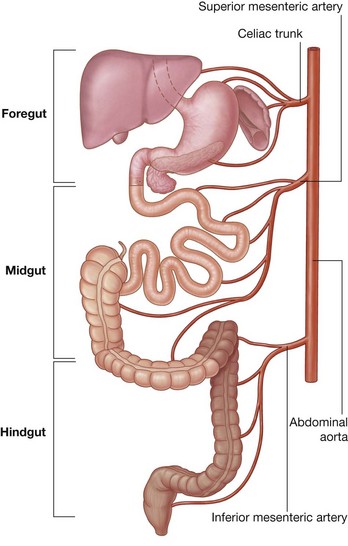
Fig. 4.110 Divisions of the gastrointestinal tract into foregut, midgut, and hindgut, summarizing the primary arterial supply to each segment.
 The foregut begins with the abdominal esophagus and ends just inferior to the major duodenal papilla, midway along the descending part of the duodenum. It includes the abdominal esophagus, stomach, duodenum (superior to the major papilla), liver, pancreas, and gallbladder. The spleen also develops in relation to the foregut region. The foregut is supplied by the celiac trunk.
The foregut begins with the abdominal esophagus and ends just inferior to the major duodenal papilla, midway along the descending part of the duodenum. It includes the abdominal esophagus, stomach, duodenum (superior to the major papilla), liver, pancreas, and gallbladder. The spleen also develops in relation to the foregut region. The foregut is supplied by the celiac trunk. The midgut begins just inferior to the major duodenal papilla, in the descending part of the duodenum, and ends at the junction between the proximal two-thirds and distal one-third of the transverse colon. It includes the duodenum (inferior to the major duodenal papilla), jejunum, ileum, cecum, appendix, ascending colon, and the right two-thirds of the transverse colon. The midgut is supplied by the superior mesenteric artery (Fig. 4.110).
The midgut begins just inferior to the major duodenal papilla, in the descending part of the duodenum, and ends at the junction between the proximal two-thirds and distal one-third of the transverse colon. It includes the duodenum (inferior to the major duodenal papilla), jejunum, ileum, cecum, appendix, ascending colon, and the right two-thirds of the transverse colon. The midgut is supplied by the superior mesenteric artery (Fig. 4.110). The hindgut begins just before the left colic flexure (the junction between the proximal two-thirds and distal one-third of the transverse colon) and ends midway through the anal canal. It includes the left one-third of the transverse colon, descending colon, sigmoid colon, rectum, and upper part of the anal canal. The hindgut is supplied by the inferior mesenteric artery (Fig. 4.110).
The hindgut begins just before the left colic flexure (the junction between the proximal two-thirds and distal one-third of the transverse colon) and ends midway through the anal canal. It includes the left one-third of the transverse colon, descending colon, sigmoid colon, rectum, and upper part of the anal canal. The hindgut is supplied by the inferior mesenteric artery (Fig. 4.110).Celiac trunk
The celiac trunk is the anterior branch of the abdominal aorta supplying the foregut. It arises from the abdominal aorta immediately below the aortic hiatus of the diaphragm (Fig. 4.111), anterior to the upper part of vertebra LI. It immediately divides into the left gastric, splenic, and common hepatic arteries.
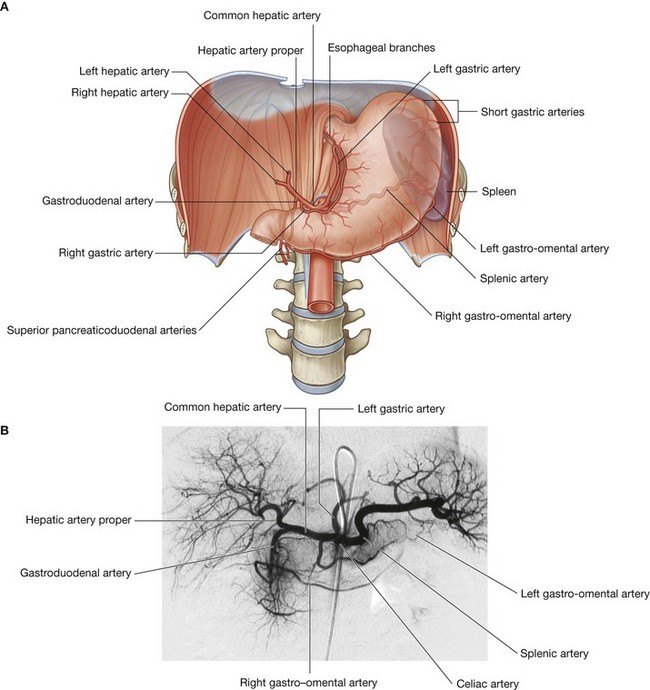
Fig. 4.111 Celiac trunk. A. Distribution of the celiac trunk. B. Digital subtraction angiography of the celiac trunk and its branches.
Left gastric artery
The left gastric artery is the smallest branch of the celiac trunk. It ascends to the cardioesophageal junction and sends esophageal branches upward to the abdominal part of the esophagus (Fig. 4.111). Some of these branches continue through the esophageal hiatus of the diaphragm and anastomose with esophageal branches from the thoracic aorta. The left gastric artery itself turns to the right and descends along the lesser curvature of the stomach in the lesser omentum. It supplies both surfaces of the stomach in this area and anastomoses with the right gastric artery.
Splenic artery
The splenic artery, the largest branch of the celiac trunk, takes a tortuous course to the left along the superior border of the pancreas (Fig. 4.111). It travels in the splenorenal ligament and divides into numerous branches, which enter the hilum of the spleen. As the splenic artery passes along the superior border of the pancreas, it gives off numerous small branches to supply the neck, body, and tail of the pancreas (Fig. 4.112).
Approaching the spleen, the splenic artery gives off short gastric arteries, which pass through the gastrosplenic ligament to supply the fundus of the stomach. It also gives off the left gastro-omental artery, which runs to the right along the greater curvature of the stomach, and anastomoses with the right gastro-omental artery.
Common hepatic artery
The common hepatic artery is a medium-sized branch of the celiac trunk that runs to the right and divides into its two terminal branches, the hepatic artery proper and the gastroduodenal artery (Fig. 4.111 and 4.112).
The hepatic artery proper ascends towards the liver in the free edge of the lesser omentum. It runs to the left of the bile duct and anterior to the portal vein, and divides into the right and left hepatic arteries near the porta hepatis (Fig. 4.113).
As the right hepatic artery nears the liver, it gives off the cystic artery to the gallbladder.
The gastroduodenal artery may give off the supraduodenal artery and does give off the posterior superior pancreaticoduodenal artery near the upper border of the superior part of the duodenum. After these branches the gastroduodenal artery continues descending posterior to the superior part of the duodenum. Reaching the lower border of the superior part of the duodenum, the gastroduodenal artery divides into its terminal branches, the right gastro-omental artery and the anterior superior pancreaticoduodenal artery (Fig. 4.112).
The right gastro-omental artery passes to the left, along the greater curvature of the stomach, eventually anastomosing with the left gastro-omental artery from the splenic artery. The right gastro-omental artery sends branches to both surfaces of the stomach and additional branches descend into the greater omentum.
The anterior superior pancreaticoduodenal artery descends and, along with the posterior superior pancreaticoduodenal artery, supplies the head of the pancreas and the duodenum (Fig. 4.112). These vessels eventually anastomose with the anterior and posterior branches of the inferior pancreaticoduodenal artery.
Superior mesenteric artery
The superior mesenteric artery is the anterior branch of the abdominal aorta supplying the midgut. It arises from the abdominal aorta immediately below the celiac artery (Fig. 4.114), anterior to the lower part of vertebra LI.
The superior mesenteric artery is crossed anteriorly by the splenic vein and the neck of pancreas. Posterior to the artery are the left renal vein, the uncinate process of the pancreas, and the inferior part of the duodenum. After giving off its first branch (the inferior pancreaticoduodenal artery) the superior mesenteric artery gives off jejunal and ileal arteries on its left (Fig. 4.114). Branching from the right side of the main trunk of the superior mesenteric artery are three vessels—the middle colic, right colic, and ileocolic arteries—which supply the terminal ileum, cecum, ascending colon, and two-thirds of the transverse colon.
Inferior pancreaticoduodenal artery
The inferior pancreaticoduodenal artery is the first branch of the superior mesenteric artery. It divides immediately into anterior and posterior branches, which ascend on the corresponding sides of the head of the pancreas. Superiorly, these arteries anastomose with anterior and posterior superior pancreaticoduodenal arteries (see Figs. 4.112 and 4.114). This arterial network supplies the head and uncinate process of the pancreas and the duodenum.
Jejunal and ileal arteries
Distal to the inferior pancreaticoduodenal artery, the superior mesenteric artery gives off numerous branches. Arising on the left is a large number of jejunal and ileal arteries supplying the jejunum and most of the ileum (Fig. 4.115). These branches leave the main trunk of the artery, pass between two layers of the mesentery, and form anastomosing arches or arcades as they pass outward to supply the small intestine. The number of arterial arcades increases distally along the gut.
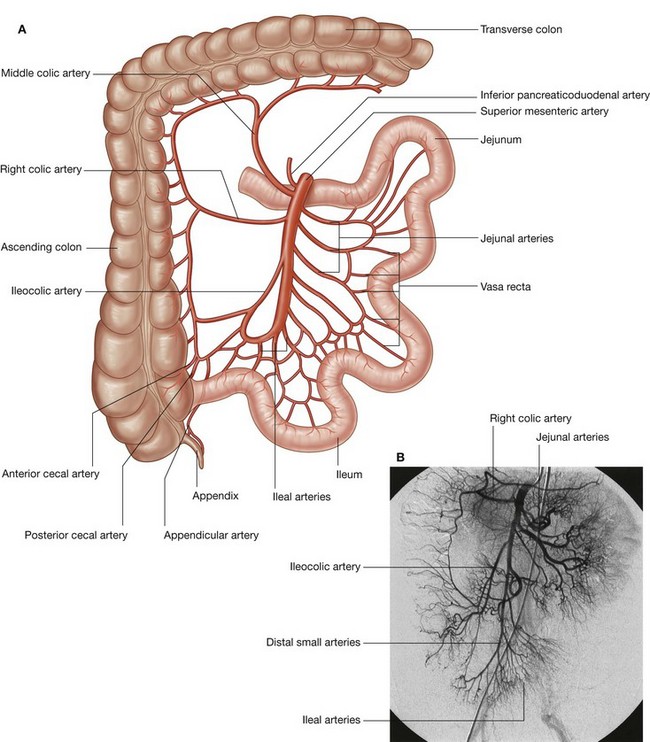
Fig. 4.115 Superior mesenteric artery. A. Distribution of the superior mesenteric artery. B. Digital subtraction angiography of the superior mesenteric artery and its branches.
There may be single and then double arcades in the area of the jejunum, with a continued increase in the number of arcades moving into and through the area of the ileum. Extending from the terminal arcade are vasa recta (straight arteries), which provide the final direct vascular supply to the walls of the small intestine. The vasa recta supplying the jejunum are usually long and close together, forming narrow windows visible in the mesentery. The vasa recta supplying the ileum are generally short and far apart, forming low broad windows.
Middle colic artery
The middle colic artery is the first of the three branches from the right side of the main trunk of the superior mesenteric artery (Fig. 4.115). Arising as the superior mesenteric artery emerges from beneath the pancreas, the middle colic artery enters the transverse mesocolon and divides into right and left branches. The right branch anastomoses with the right colic artery while the left branch anastomoses with the left colic artery, which is a branch of the inferior mesenteric artery.
Right colic artery
Continuing distally along the main trunk of the superior mesenteric artery, the right colic artery is the second of the three branches from the right side of the main trunk of the superior mesenteric artery (Fig. 4.115). It is an inconsistent branch, and passes to the right in a retroperitoneal position to supply the ascending colon. Nearing the colon, it divides into a descending branch, which anastomoses with the ileocolic artery, and an ascending branch, which anastomoses with the middle colic artery.
Ileocolic artery
The final branch arising from the right side of the superior mesenteric artery is the ileocolic artery (Fig. 4.115). This passes downward and to the right toward the right iliac fossa where it divides into superior and inferior branches:
 the superior branch passes upward along the ascending colon to anastomose with the right colic artery;
the superior branch passes upward along the ascending colon to anastomose with the right colic artery; the inferior branch continues toward the ileocolic junction dividing into colic, cecal, appendicular, and ileal branches (Fig. 4.115).
the inferior branch continues toward the ileocolic junction dividing into colic, cecal, appendicular, and ileal branches (Fig. 4.115).The specific pattern of distribution and origin of these branches is variable:
 the colic branch crosses to the ascending colon and passes upward to supply the first part of the ascending colon;
the colic branch crosses to the ascending colon and passes upward to supply the first part of the ascending colon;Inferior mesenteric artery
The inferior mesenteric artery is the anterior branch of the abdominal aorta that supplies the hindgut. It is the smallest of the three anterior branches of the abdominal aorta and arises anterior to the body of vertebra LIII. Initially, the inferior mesenteric artery descends anteriorly to the aorta and then passes to the left as it continues inferiorly (Fig. 4.116). Its branches include the left colic artery, several sigmoid arteries, and the superior rectal artery.
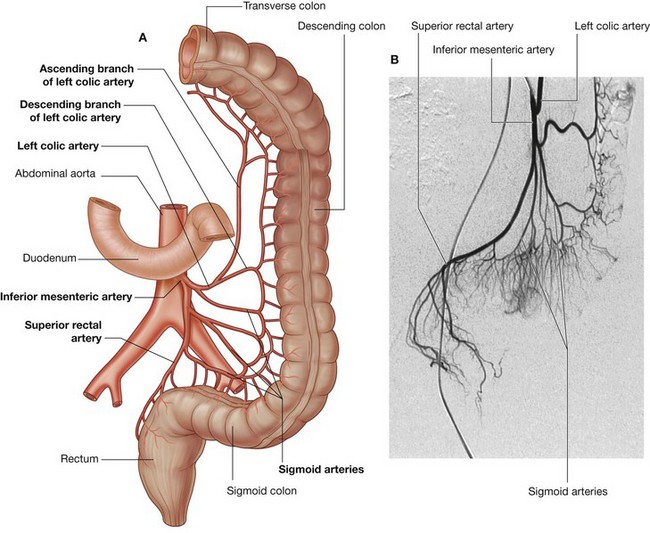
Fig. 4.116 Inferior mesenteric artery. A. Distribution of the inferior mesenteric artery. B. Digital subtraction angiography of the inferior mesenteric artery and its branches.
Left colic artery
The left colic artery is the first branch of the inferior mesenteric artery (Fig. 4.116). It ascends retroperitoneally, dividing into ascending and descending branches:
Sigmoid arteries
The sigmoid arteries consist of two to four branches, which descend to the left, in the sigmoid mesocolon, to supply the lowest part of the descending colon and the sigmoid colon (Fig. 4.116). These branches anastomose superiorly with branches from the left colic artery and inferiorly with branches from the superior rectal artery.
Superior rectal artery
The terminal branch of the inferior mesenteric artery is the superior rectal artery (Fig. 4.116). This vessel descends into the pelvic cavity in the sigmoid mesocolon, crossing the left common iliac vessels. Opposite vertebra SIII, the superior rectal artery divides. The two terminal branches descend on each side of the rectum, dividing into smaller branches in the wall of the rectum. These smaller branches continue inferiorly to the level of the internal anal sphincter, anastomosing along the way with branches from the middle rectal arteries (from the internal iliac artery) and the inferior rectal arteries (from the internal pudendal artery).
In the clinic
Vascular supply to the gastrointestinal system
The abdominal parts of the gastrointestinal system are supplied mainly by the celiac trunk and the superior mesenteric and inferior mesenteric arteries:
 the celiac trunk supplies the lower esophagus, stomach, and the proximal half of the descending part of the duodenum;
the celiac trunk supplies the lower esophagus, stomach, and the proximal half of the descending part of the duodenum; the superior mesenteric artery supplies the rest of the duodenum, the jejunum, the ileum, the ascending colon, and the proximal two-thirds of the transverse colon; and
the superior mesenteric artery supplies the rest of the duodenum, the jejunum, the ileum, the ascending colon, and the proximal two-thirds of the transverse colon; and the inferior mesenteric artery supplies the rest of the transverse colon, the descending colon, the sigmoid colon, and most of the rectum.
the inferior mesenteric artery supplies the rest of the transverse colon, the descending colon, the sigmoid colon, and most of the rectum.Along the descending part of the duodenum there is a potential watershed area between the celiac trunk blood supply and the superior mesenteric arterial blood supply. It is unusual for this area to become ischemic, whereas the watershed area between the superior mesenteric artery and the inferior mesenteric artery, at the splenic flexure, is extremely vulnerable to ischemia.
In certain disease states, the region of the splenic flexure of the colon can become ischemic. When this occurs, the mucosa sloughs off, rendering the patient susceptible to infection and perforation of the large bowel, which then requires urgent surgical attention.
Arteriosclerosis may occur throughout the abdominal aorta and at the openings of the celiac trunk and the superior mesenteric and inferior mesenteric arteries. Not infrequently, the inferior mesenteric artery becomes occluded. Interestingly, many of these patients do not suffer any complications because anastomoses between the right, middle, and left colic arteries gradually enlarge, forming a continuous marginal artery. The distal large bowel therefore becomes supplied by this enlarged marginal artery (marginal artery of Drummond) which replaces the blood supply of the inferior mesenteric artery (Fig. 4.117).
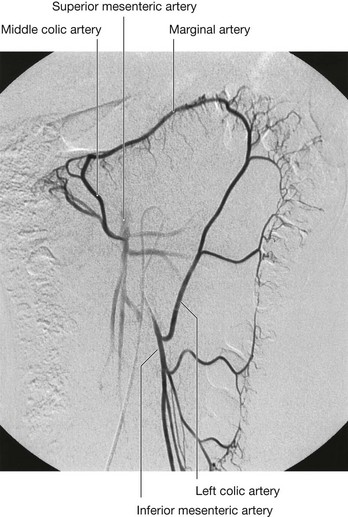
Fig. 4.117 Enlarged marginal artery connecting the superior and inferior mesenteric arteries. Digital subtraction angiogram.
If the openings of the celiac trunk and superior mesenteric artery becomes narrowed, the blood supply to the gut is diminished. After a heavy meal, the oxygen demand of the bowel therefore outstrips the limited supply of blood through the stenosed vessels, resulting in severe pain and discomfort (mesenteric angina). Patients with this condition tend not to eat because of the pain and rapidly lose weight. The diagnosis is determined by aortic angiography and the stenoses of the celiac trunk and superior mesenteric artery are best appreciated in the lateral view.
Venous drainage
Venous drainage of the spleen, pancreas, gallbladder, and the abdominal part of the gastrointestinal tract, except for the inferior part of the rectum, is through the portal system of veins, which deliver blood from these structures to the liver. Once blood passes through the hepatic sinusoids, it passes through progressively larger veins until it enters the hepatic veins, which return the venous blood to the inferior vena cava just inferior to the diaphragm.
Portal vein
The portal vein is the final common pathway for the transport of venous blood from the spleen, pancreas, gallbladder, and the abdominal part of the gastrointestinal tract. It is formed by the union of the splenic vein and the superior mesenteric vein posterior to the neck of the pancreas at the level of vertebra LII (Fig. 4.118).
Ascending toward the liver, the portal vein passes posterior to the superior part of the duodenum and enters the right margin of the lesser omentum. As it passes through this part of the lesser omentum, it is anterior to the omental foramen and posterior to both the bile duct, which is slightly to its right, and the hepatic artery proper, which is slightly to its left (see Fig. 4.113, p. 332).
On approaching the liver, the portal vein divides into right and left branches, which enter the liver parenchyma. Tributaries to the portal vein include:
 the para-umbilical veins, which are associated with the obliterated umbilical vein and connect to veins on the anterior abdominal wall (Fig. 4.120).
the para-umbilical veins, which are associated with the obliterated umbilical vein and connect to veins on the anterior abdominal wall (Fig. 4.120).Splenic vein
The splenic vein forms from numerous smaller vessels leaving the hilum of the spleen (Fig. 4.119). It passes to the right, passing through the splenorenal ligament with the splenic artery and the tail of pancreas. Continuing to the right, the large, straight splenic vein is in contact with the body of the pancreas as it crosses the posterior abdominal wall. Posterior to the neck of the pancreas, the splenic vein joins the superior mesenteric vein to form the portal vein.
Superior mesenteric vein
The superior mesenteric vein drains blood from the small intestine, cecum, ascending colon, and transverse colon (Fig. 4.119). It begins in the right iliac fossa as veins draining the terminal ileum, cecum, and appendix join, and ascends in the mesentery to the right of the superior mesenteric artery.
Posterior to the neck of the pancreas, the superior mesenteric vein joins the splenic vein to form the portal vein.
As a corresponding vein accompanies each branch of the superior mesenteric artery, tributaries to the superior mesenteric vein include jejunal, ileal, ileocolic, right colic, and middle colic veins. Additional tributaries include:
 the anterior and posterior inferior pancreaticoduodenal veins, which pass alongside the arteries of the same name; the anterior superior pancreaticoduodenal vein usually empties into the right gastro-omental vein, and the posterior superior pancreatico-duodenal vein usually empties directly into the portal vein.
the anterior and posterior inferior pancreaticoduodenal veins, which pass alongside the arteries of the same name; the anterior superior pancreaticoduodenal vein usually empties into the right gastro-omental vein, and the posterior superior pancreatico-duodenal vein usually empties directly into the portal vein.Inferior mesenteric vein
The inferior mesenteric vein drains blood from the rectum, sigmoid colon, descending colon, and splenic flexure (Fig. 4.119). It begins as the superior rectal vein and ascends, receiving tributaries from the sigmoid veins and the left colic vein. All these veins accompany arteries of the same name. Continuing to ascend, the inferior mesenteric vein passes posterior to the body of the pancreas and usually joins the splenic vein. Occasionally, it ends at the junction of the splenic and superior mesenteric veins or joins the superior mesenteric vein.
In the clinic
Hepatic cirrhosis
Cirrhosis is a complex disorder of the liver, the diagnosis of which is confirmed histologically. When a diagnosis is suspected, a liver biopsy is necessary.
Cirrhosis is characterized by widespread hepatic fibrosis interspersed with areas of nodular regeneration and abnormal reconstruction of pre-existing lobular architecture. The presence of cirrhosis implies previous or continuing liver cell damage.
The etiology of cirrhosis is complex and includes toxins (alcohol), viral inflammation, biliary obstruction, vascular outlet obstruction, nutritional (malnutrition) causes, and inherited anatomical and metabolic disorders.
As the cirrhosis progresses, the intrahepatic vasculature is distorted, which in turn leads to increased pressure in the portal vein and its draining tributaries (portal hypertension). Portal hypertension produces increased pressure in the splenic venules leading to splenic enlargement. At the sites of portosystemic anastomosis (see below), large dilated varicose veins develop. These veins are susceptible to bleeding and may produce marked blood loss, which in some instances can be fatal.
The liver is responsible for the production of numerous proteins, including those of the clotting cascade. Any disorder of the liver (including infection and cirrhosis) may decrease the production of these proteins and so prevent adequate blood clotting. Patients with severe cirrhosis of the liver have a significant risk of serious bleeding, even from small cuts; in addition, when varices rupture, there is a danger of rapid exsanguination.
As the liver progressively fails, the patient develops salt and water retention, which produces skin and subcutaneous edema. Fluid (ascites) is also retained in the peritoneal cavity, which can hold many liters.
The poorly functioning liver cells (hepatocytes) are unable to break down blood and blood products, leading to an increase in the serum bilirubin level, which manifests as jaundice.
With the failure of normal liver metabolism, toxic metabolic byproducts do not convert to nontoxic metabolites. This buildup of noxious compounds is made worse by the numerous portosystemic shunts, which allow the toxic metabolites to bypass the liver. Patients may develop severe neurological features, which may lead to epileptic fits, dementia, and irreversible neurological damage.
The hepatic portal system drains blood from the visceral organs of the abdomen to the liver. In normal individuals, 100% of the portal venous blood flow can be recovered from the hepatic veins, whereas in patients with elevated portal vein pressure (e.g., from cirrhosis), there is significantly less blood flow to the liver. The rest of the blood enters collateral channels, which drain into the systemic circulation at specific points (Fig. 4.120). The largest of these collaterals occur at:
 the gastroesophageal junction around the cardia of the stomach—where the left gastric vein and its tributaries form a portosystemic anastomosis with tributaries to the azygos system of veins of the caval system;
the gastroesophageal junction around the cardia of the stomach—where the left gastric vein and its tributaries form a portosystemic anastomosis with tributaries to the azygos system of veins of the caval system; the anus—the superior rectal vein of the portal system anastomoses with the middle and inferior rectal veins of the systemic venous system; and
the anus—the superior rectal vein of the portal system anastomoses with the middle and inferior rectal veins of the systemic venous system; and the anterior abdominal wall around the umbilicus—the para-umbilical veins anastomose with veins on the anterior abdominal wall.
the anterior abdominal wall around the umbilicus—the para-umbilical veins anastomose with veins on the anterior abdominal wall.When the pressure in the portal vein is elevated, venous enlargement (varices) tend to occur at and around the sites of portosystemic anastomoses and these enlarged veins are called:
Esophageal varices are susceptible to trauma and, once damaged, may bleed profusely, requiring urgent surgical intervention.
Lymphatics
Lymphatic drainage of the abdominal part of the gastrointestinal tract, as low as the inferior part of the rectum, as well as the spleen, pancreas, gallbladder, and liver, is through vessels and nodes that eventually end in large collections of pre-aortic lymph nodes at the origins of the three anterior branches of the abdominal aorta, which supply these structures. These collections are therefore referred to as the celiac, superior mesenteric, and inferior mesenteric groups of pre-aortic lymph nodes. Lymph from viscera supplied by:
 the celiac trunk (i.e., structures that are part of the abdominal foregut) drains to pre-aortic nodes near the origin of the celiac trunk (Fig. 4.121)—these celiac nodes also receive lymph from the superior mesenteric and inferior mesenteric groups of pre-aortic nodes, and lymph from the celiac nodes enters the cisterna chyli;
the celiac trunk (i.e., structures that are part of the abdominal foregut) drains to pre-aortic nodes near the origin of the celiac trunk (Fig. 4.121)—these celiac nodes also receive lymph from the superior mesenteric and inferior mesenteric groups of pre-aortic nodes, and lymph from the celiac nodes enters the cisterna chyli; the superior mesenteric artery (i.e., structures that are part of the abdominal midgut) drains to pre-aortic nodes near the origin of the superior mesenteric artery (Fig. 4.121)—these superior mesenteric nodes also receive lymph from the inferior mesenteric groups of pre-aortic nodes, and lymph from the superior mesenteric nodes drains to the celiac nodes; and
the superior mesenteric artery (i.e., structures that are part of the abdominal midgut) drains to pre-aortic nodes near the origin of the superior mesenteric artery (Fig. 4.121)—these superior mesenteric nodes also receive lymph from the inferior mesenteric groups of pre-aortic nodes, and lymph from the superior mesenteric nodes drains to the celiac nodes; and the inferior mesenteric artery (i.e., structures that are part of the abdominal hindgut) drains to pre-aortic nodes near the origin of the inferior mesenteric artery (Fig. 4.121), and lymph from the inferior mesenteric nodes drains to the superior mesenteric nodes.
the inferior mesenteric artery (i.e., structures that are part of the abdominal hindgut) drains to pre-aortic nodes near the origin of the inferior mesenteric artery (Fig. 4.121), and lymph from the inferior mesenteric nodes drains to the superior mesenteric nodes.Innervation
Abdominal viscera are innervated by both extrinsic and intrinsic components of the nervous system:
 extrinsic innervation involves receiving motor impulses from, and sending sensory information to, the central nervous system;
extrinsic innervation involves receiving motor impulses from, and sending sensory information to, the central nervous system; intrinsic innervation involves the regulation of digestive tract activities by a generally self-sufficient network of sensory and motor neurons (the enteric nervous system).
intrinsic innervation involves the regulation of digestive tract activities by a generally self-sufficient network of sensory and motor neurons (the enteric nervous system).Abdominal viscera receiving extrinsic innervation include the abdominal part of the gastrointestinal tract, the spleen, the pancreas, the gallbladder, and the liver. These viscera send sensory information back to the central nervous system through visceral afferent fibers and receive motor impulses from the central nervous system through visceral efferent fibers.
The visceral efferent fibers are part of the sympathetic and parasympathetic parts of the autonomic division of the PNS.
Structural components serving as conduits for these afferent and efferent fibers include posterior and anterior roots of the spinal cord, respectively, spinal nerves, anterior rami, white and gray rami communicantes, the sympathetic trunks, splanchnic nerves carrying sympathetic fibers (thoracic, lumbar, and sacral), parasympathetic fibers (pelvic), the prevertebral plexus and related ganglia, and the vagus nerves [X].
The enteric nervous system consists of motor and sensory neurons in two interconnected plexuses in the walls of the gastrointestinal tract. These neurons control the coordinated contraction and relaxation of intestinal smooth muscle and regulate gastric secretion and blood flow.
Sympathetic trunks
The sympathetic trunks are two parallel nerve cords extending on either side of the vertebral column from the base of the skull to the coccyx (Fig. 4.122). As they pass through the neck, they lie posterior to the carotid sheath. In the upper thorax, they are anterior to the necks of the ribs, while in the lower thorax they are on the lateral aspect of the vertebral bodies. In the abdomen, they are anterolateral to the lumbar vertebral bodies and, continuing into the pelvis, they are anterior to the sacrum. The two sympathetic trunks come together anterior to the coccyx to form the ganglion impar.
Throughout the extent of the sympathetic trunks, small raised areas are visible. These collections of neuronal cell bodies outside the CNS are the paravertebral sympathetic ganglia. There are usually:
The ganglia and trunks are connected to adjacent spinal nerves by gray rami communicantes throughout the length of the sympathetic trunk and by white rami communicantes in the thoracic and upper lumbar parts of the trunk (T1 to L2). Neuronal fibers found in the sympathetic trunks include preganglionic and postganglionic sympathetic fibers and visceral afferent fibers.
Splanchnic nerves
The splanchnic nerves are important components in the innervation of the abdominal viscera. They pass from the sympathetic trunk or sympathetic ganglia associated with the trunk, to the prevertebral plexus and ganglia anterior to the abdominal aorta.
There are two different types of splanchnic nerves, depending on the type of visceral efferent fiber they are carrying:
Thoracic splanchnic nerves
Three thoracic splanchnic nerves pass from sympathetic ganglia along the sympathetic trunk in the thorax to the prevertebral plexus and ganglia associated with the abdominal aorta in the abdomen (Fig. 4.123):
 the greater splanchnic nerve arises from the fifth to the ninth (or tenth) thoracic ganglia and travels to the celiac ganglion in the abdomen (a prevertebral ganglion associated with the celiac trunk);
the greater splanchnic nerve arises from the fifth to the ninth (or tenth) thoracic ganglia and travels to the celiac ganglion in the abdomen (a prevertebral ganglion associated with the celiac trunk); the lesser splanchnic nerve arises from the ninth and tenth (or tenth and eleventh) thoracic ganglia and travels to the aorticorenal ganglion;
the lesser splanchnic nerve arises from the ninth and tenth (or tenth and eleventh) thoracic ganglia and travels to the aorticorenal ganglion;Lumbar and sacral splanchnic nerves
There are usually two to four lumbar splanchnic nerves, which pass from the lumbar part of the sympathetic trunk or associated ganglia and enter the prevertebral plexus (Fig. 4.123).
Similarly, the sacral splanchnic nerves pass from the sacral part of the sympathetic trunk or associated ganglia and enter the inferior hypogastric plexus, which is an extension of the prevertebral plexus into the pelvis.
Pelvic splanchnic nerves
The pelvic splanchnic nerves (parasympathetic root) are unique. They are the only splanchnic nerves that carry parasympathetic fibers. In other words, they do not originate from the sympathetic trunks. Rather, they originate directly from the anterior rami of S2 to S4. Preganglionic parasympathetic fibers originating in the sacral spinal cord pass from the S2 to S4 spinal nerves to the inferior hypogastric plexus (Fig. 4.123). Once in this plexus, some of these fibers pass upward, enter the abdominal prevertebral plexus, and distribute with the arteries supplying the hindgut. This provides the pathway for innervation of the distal one-third of the transverse colon, the descending colon, and the sigmoid colon by preganglionic parasympathetic fibers.
Abdominal prevertebral plexus and ganglia
The abdominal prevertebral plexus is a collection of nerve fibers that surrounds the abdominal aorta and is continuous onto its major branches. Scattered throughout the length of the abdominal prevertebral plexus are cell bodies of postganglionic sympathetic fibers. Some of these cell bodies are organized into distinct ganglia, while others are more random in their distribution. The ganglia are usually associated with specific branches of the abdominal aorta and named after these branches.
The three major divisions of the abdominal prevertebral plexus and associated ganglia are the celiac, aortic, and superior hypogastric plexuses (Fig. 4.124).
 The celiac plexus is the large accumulation of nerve fibers and ganglia associated with the roots of the celiac trunk and superior mesenteric artery immediately below the aortic hiatus of the diaphragm. Ganglia associated with the celiac plexus include two celiac ganglia, a single superior mesenteric ganglion, and two aorticorenal ganglia.
The celiac plexus is the large accumulation of nerve fibers and ganglia associated with the roots of the celiac trunk and superior mesenteric artery immediately below the aortic hiatus of the diaphragm. Ganglia associated with the celiac plexus include two celiac ganglia, a single superior mesenteric ganglion, and two aorticorenal ganglia. The aortic plexus consists of nerve fibers and associated ganglia on the anterior and lateral surfaces of the abdominal aorta extending from just below the origin of the superior mesenteric artery to the bifurcation of the aorta into the two common iliac arteries. The major ganglion in this plexus is the inferior mesenteric ganglion at the root of the inferior mesenteric artery.
The aortic plexus consists of nerve fibers and associated ganglia on the anterior and lateral surfaces of the abdominal aorta extending from just below the origin of the superior mesenteric artery to the bifurcation of the aorta into the two common iliac arteries. The major ganglion in this plexus is the inferior mesenteric ganglion at the root of the inferior mesenteric artery. The superior hypogastric plexus contains numerous small ganglia and is the final part of the abdominal prevertebral plexus before the prevertebral plexus continues into the pelvic cavity.
The superior hypogastric plexus contains numerous small ganglia and is the final part of the abdominal prevertebral plexus before the prevertebral plexus continues into the pelvic cavity.Each of these major plexuses gives origin to a number of secondary plexuses, which may also contain small ganglia. These plexuses are usually named after the vessels with which they are associated. For example, the celiac plexus is usually described as giving origin to the superior mesenteric plexus and the renal plexus, as well as other plexuses that extend out along the various branches of the celiac trunk. Similarly, the aortic plexus has secondary plexuses consisting of the inferior mesenteric plexus, the spermatic plexus, and the external iliac plexus.
Inferiorly, the superior hypogastric plexus divides into the hypogastric nerves, which descend into the pelvis and contribute to the formation of the inferior hypogastric or pelvic plexus (Fig. 4.124).
Parasympathetic innervation
Parasympathetic innervation of the abdominal part of the gastrointestinal tract, and of the spleen, pancreas, gallbladder, and liver is from two sources—the vagus nerves [X] and the pelvic splanchnic nerves.
Vagus nerves
The vagus nerves [X] enter the abdomen associated with the esophagus as the esophagus passes through the diaphragm (Fig. 4.125) and provides parasympathetic innervation to the foregut and midgut.
After entering the abdomen as the anterior and posterior vagal trunks, they send branches to the abdominal prevertebral plexus. These branches contain preganglionic parasympathetic fibers and visceral afferent fibers, which are distributed with the other components of the prevertebral plexus along the branches of the abdominal aorta.
Pelvic splanchnic nerves
The pelvic splanchnic nerves, carrying preganglionic parasympathetic fibers from S2 to S4 spinal cord levels, enter the inferior hypogastric plexus in the pelvis. Some of these fibers move upward into the inferior mesenteric part of the prevertebral plexus in the abdomen (Fig. 4.125). Once there, these fibers are distributed with branches of the inferior mesenteric artery and provide parasympathetic innervation to the hindgut.
Enteric system
The enteric system is a division of the visceral part of the nervous system and is a local neuronal circuit in the wall of the gastrointestinal tract. It consists of motor and sensory neurons organized into two interconnected plexuses (the myenteric and submucosal plexuses) between the layers of the gastrointestinal wall, and the associated nerve fibers that pass between the plexuses and from the plexuses to the adjacent tissue (Fig. 4.126).
The enteric system regulates and coordinates numerous gastrointestinal tract activities, including gastric secretory activity, gastrointestinal blood flow, and the contraction and relaxation cycles of smooth muscle (peristalsis).
Although the enteric system is generally independent of the central nervous system, it does receive input from postganglionic sympathetic and preganglionic parasympathetic neurons that modifies its activities.
Sympathetic innervation of the stomach
The pathway of sympathetic innervation of the stomach is as follows:
 A preganglionic sympathetic fiber originating at the T6 level of the spinal cord enters an anterior root to leave the spinal cord.
A preganglionic sympathetic fiber originating at the T6 level of the spinal cord enters an anterior root to leave the spinal cord. At the level of the intervertebral foramen, the anterior root (which contains the preganglionic fiber) and a posterior root join to form a spinal nerve.
At the level of the intervertebral foramen, the anterior root (which contains the preganglionic fiber) and a posterior root join to form a spinal nerve. Outside the vertebral column, the preganglionic fiber leaves the anterior ramus of the spinal nerve through the white ramus communicans.
Outside the vertebral column, the preganglionic fiber leaves the anterior ramus of the spinal nerve through the white ramus communicans. Entering the sympathetic trunk, the preganglionic fiber does not synapse, but passes through the trunk, and enters the greater splanchnic nerve.
Entering the sympathetic trunk, the preganglionic fiber does not synapse, but passes through the trunk, and enters the greater splanchnic nerve. The greater splanchnic nerve passes through the crura of the diaphragm and enters the celiac ganglion.
The greater splanchnic nerve passes through the crura of the diaphragm and enters the celiac ganglion. The postganglionic fiber joins the plexus of nerve fibers surrounding the celiac trunk and continues along its branches.
The postganglionic fiber joins the plexus of nerve fibers surrounding the celiac trunk and continues along its branches. The postganglionic fiber travels through the plexus of nerves accompanying the branches of the celiac trunk supplying the stomach and eventually reaches its point of distribution.
The postganglionic fiber travels through the plexus of nerves accompanying the branches of the celiac trunk supplying the stomach and eventually reaches its point of distribution. This input from the sympathetic system may modify the activities of the gastrointestinal tract controlled by the enteric nervous system.
This input from the sympathetic system may modify the activities of the gastrointestinal tract controlled by the enteric nervous system.In the clinic
Surgery for obesity
Surgery for obesity is also known as weight loss surgery and bariatric surgery. This type of surgery has become increasingly popular over the last few years for patients who are unable to achieve significant weight loss through appropriate diet modification and exercise programs. It is often regarded as a last resort. Importantly, we have to recognize the increasing medical impact that overweight patients pose. With obesity the patient is more likely to develop diabetes and cardiovascular problems and may suffer from increased general health disorders. All of these have a significant impact on health care budgeting and are regarded as serious conditions for the “health of a nation.”
There are a number of surgical options to treat obesity. The American College of Physicians has indicated that, currently, patients with a body mass index of greater than 40 kg per m2 who have failed adequate exercise or diet programs with other complicating factors (e.g., diabetes) may require surgical procedures.
Surgery for patients who are morbidly obese can be categorized into two main groups: malabsorptive procedures and restrictive procedures.
Malabsorptive procedures
There are a variety of bypass procedures that produce a malabsorption state, preventing further weight gain and also producing weight loss. There are complications, which may include anemia, osteoporosis, and diarrhea (e.g., jejunoileal bypass).
Predominantly restrictive procedures
Restrictive procedures involve placing a band or stapling in or around the stomach to decrease the size of the organ. This reduction produces an earlier feeling of satiety and prevents the patient from overeating.
Combination procedures
Probably the most popular procedure currently in the United States, this procedure involves stapling the proximal stomach and joining a loop of small bowel to the small gastric remnant.
Any overweight patient undergoing surgery faces significant risk and increased morbidity with mortality rates from 1% to 5%.