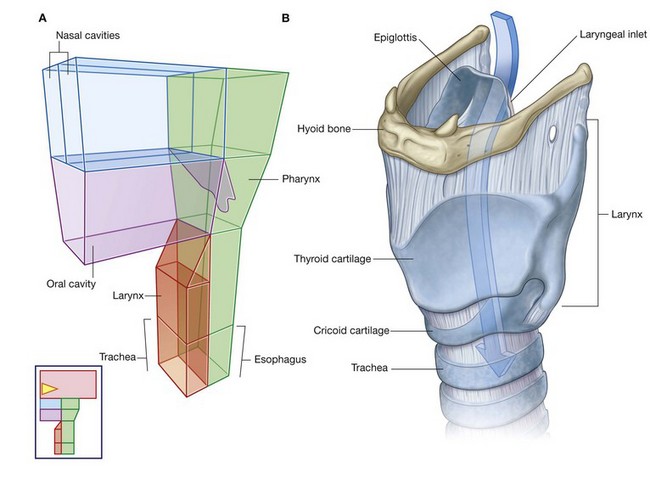
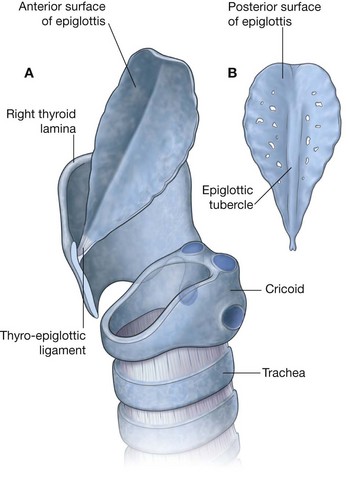
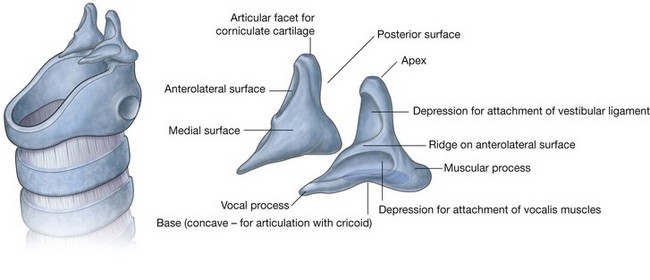
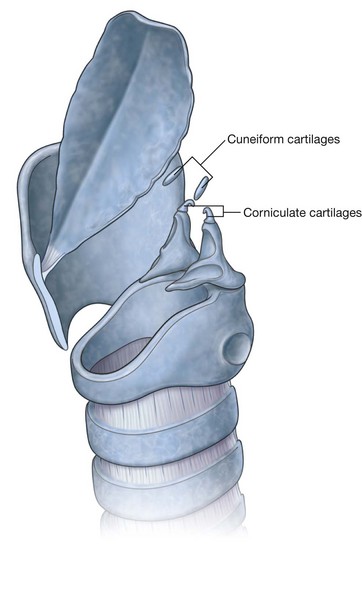
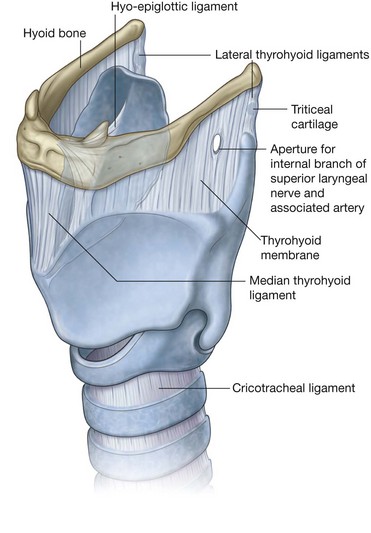
NASAL CAVITIES
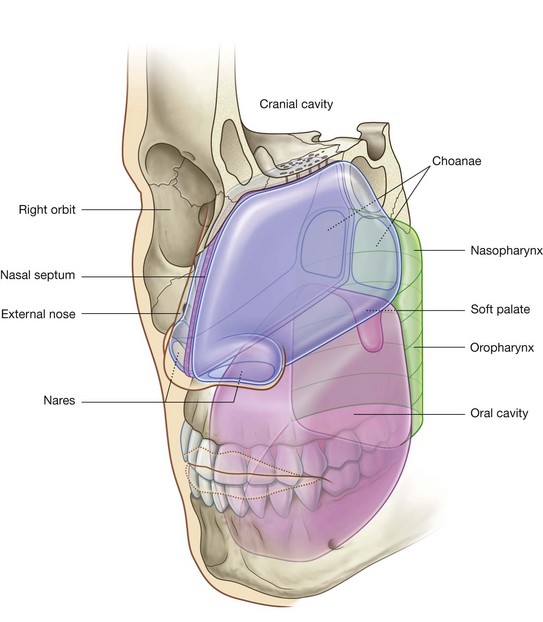
The two nasal cavities are the uppermost parts of the respiratory tract and contain the olfactory receptors. They are elongated wedge-shaped spaces with a large inferior base and a narrow superior apex (Figs. 8.217 and 8.218) and are held open by a skeletal framework consisting mainly of bone and cartilage.
The smaller anterior regions of the cavities are enclosed by the external nose, whereas the larger posterior regions are more central within the skull. The anterior apertures of the nasal cavities are the nares, which open onto the inferior surface of the nose. The posterior apertures are the choanae, which open into the nasopharynx.
The nasal cavities are separated:

from each other by a midline nasal septum;

from the oral cavity below by the hard palate; and

from the cranial cavity above by parts of the frontal, ethmoid, and sphenoid bones.
Lateral to the nasal cavities are the orbits.
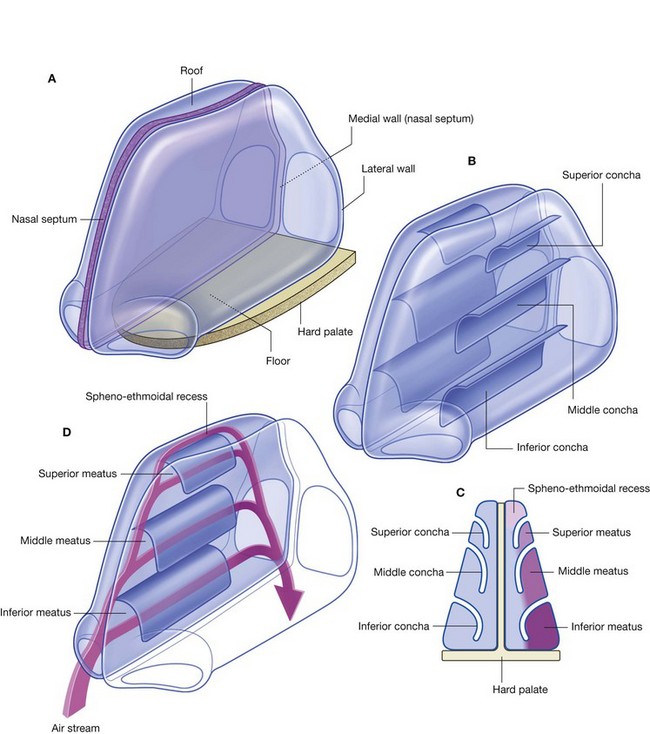
Each nasal cavity has a floor, roof, medial wall, and lateral wall (Fig. 8.218A).
Lateral wall
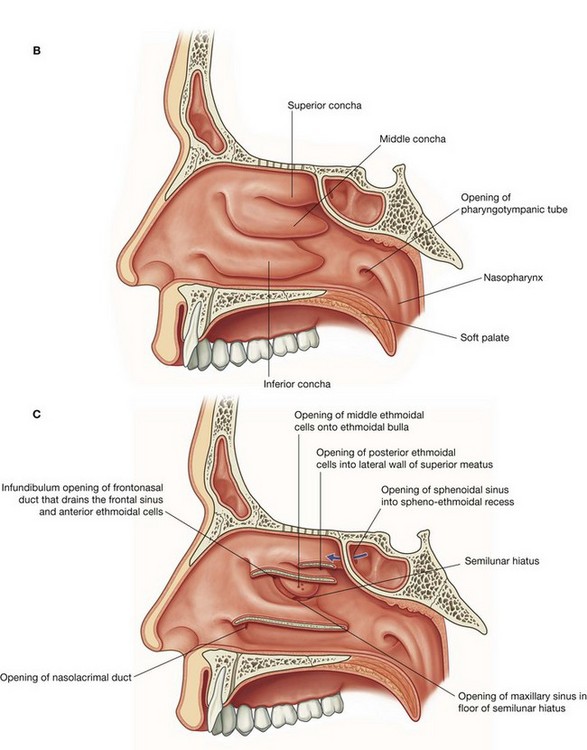
The lateral wall is characterized by three curved shelves of bone (conchae), which are one above the other and project medially and inferiorly across the nasal cavity (Fig. 8.218B). The medial, anterior, and posterior margins of the conchae are free.
The conchae divide each nasal cavity into four air channels (Fig. 8.218C and 8.218D):

an
inferior nasal meatus between the
inferior concha and the nasal floor;

a
middle nasal meatus between the inferior and
middle concha;

a
superior nasal meatus between the middle and
superior concha; and

a
spheno-ethmoidal recess between the superior concha and the nasal roof.
These conchae increase the surface area of contact between tissues of the lateral wall and the respired air.
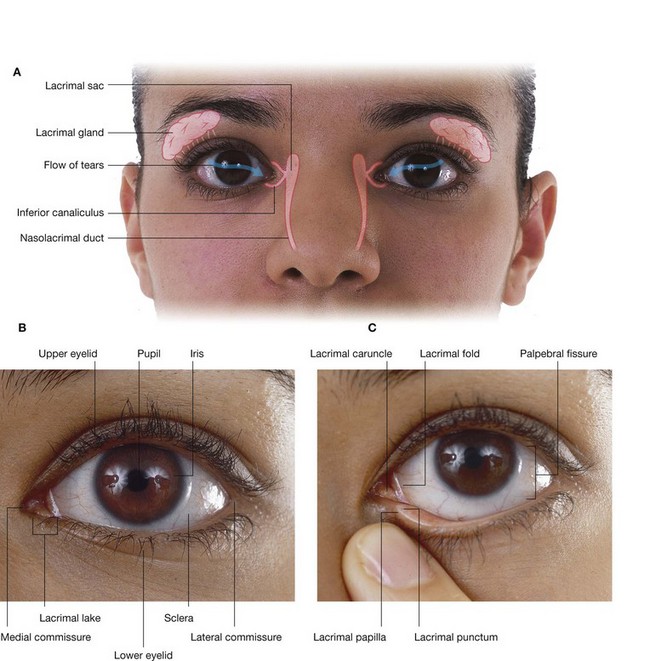
The openings of the paranasal sinuses, which are extensions of the nasal cavity that erode into the surrounding bones during childhood and early adulthood, are on the lateral wall and roof of the nasal cavities (Fig. 8.219). In addition, the lateral wall also contains the opening of the nasolacrimal duct, which drains tears from the eye into the nasal cavity.
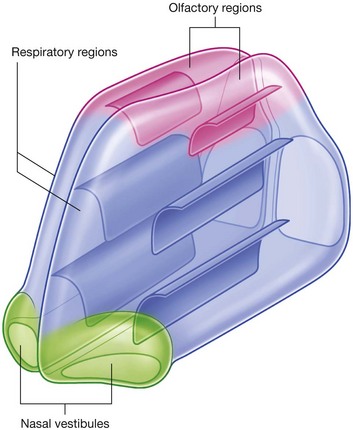
Regions
Each nasal cavity consists of three general regions—the nasal vestibule, the respiratory region, and the olfactory region (Fig. 8.220):

the
nasal vestibule is a small dilated space just internal to the naris that is lined by skin and contains hair follicles;

the
respiratory region is the largest part of the nasal cavity, has a rich neurovascular supply, and is lined by respiratory epithelium composed mainly of ciliated and mucous cells;

the
olfactory region is small, is at the apex of each nasal cavity, is lined by olfactory epithelium, and contains the olfactory receptors.
In addition to housing receptors for the sense of smell (olfaction), the nasal cavities adjust the temperature and humidity of respired air by the action of a rich blood supply, and trap and remove particulate matter from the airway by filtering the air through hair in the vestibule and by capturing foreign material in abundant mucus. The mucus normally is moved posteriorly by cilia on epithelial cells in the nasal cavities and is swallowed.
Innervation and blood supply
Innervation of the nasal cavities is by three cranial nerves:

olfaction is carried by the olfactory nerve [I];

general sensation is carried by the trigeminal nerve [V], the anterior region by the ophthalmic nerve [V
1], and the posterior region by the maxillary nerve [V
2];

all glands are innervated by parasympathetic fibers in the facial nerve [VII] (greater petrosal nerve), which join branches of the maxillary nerve [V
2] in the pteryatine fossa.
Sympathetic fibers are ultimately derived from the T1 spinal cord level. They synapse mainly in the superior cervical sympathetic ganglion, and postganglionic fibers reach the nasal cavities along blood vessels, or by joining branches of the maxillary nerve [V2] in the pteryatine fossa.
Blood supply to the nasal cavities is by:

terminal branches of the maxillary and facial arteries, which originate from the external carotid artery; and

from ethmoidal branches of the ophthalmic artery, which originates from the internal carotid artery.
Skeletal framework
Bones that contribute to the skeletal framework of the nasal cavities include:

the unpaired ethmoid, sphenoid, frontal bone, and vomer;

the paired nasal, maxillary, palatine and lacrimal bones, and inferior conchae.
Of all the bones associated with the nasal cavities, the ethmoid is a key element.
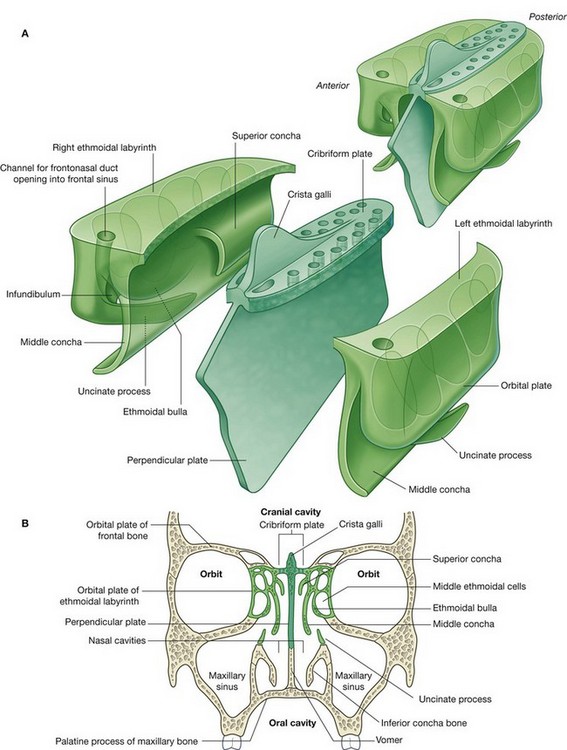
Ethmoid bone
The single ethmoid bone is one of the most complex bones in the skull. It contributes to the roof, lateral wall, and medial wall of both nasal cavities, and contains the ethmoidal cells (ethmoidal sinuses).
The ethmoid bone is cuboidal in overall shape (Fig. 8.221A) and is composed of two rectangular box-shaped ethmoidal labyrinths, one on each side, united superiorly across the midline by a perforated sheet of bone (the cribriform plate). A second sheet of bone (the perpendicular plate) descends vertically in the median sagittal plane from the cribriform plate to form part of the nasal septum.
Each ethmoidal labyrinth is composed of two delicate sheets of bone, which sandwich between them the ethmoidal cells.

the lateral sheet of bone (the
orbital plate) is flat and forms part of the medial wall of the orbit;

the medial sheet of bone forms the upper part of the lateral wall of the nasal cavity and is characterized by two processes and a swelling (
Fig. 8.221B)—the two processes are curved shelves of bone (the superior and middle conchae), which project across the nasal cavity and curve downward ending in free medial margins, while inferior to the origin of the middle concha, the middle ethmoidal cells form a prominent bulge (the
ethmoidal bulla), on the medial wall of the labyrinth.
Extending anterosuperiorly from just under the bulla is a groove (the ethmoidal infundibulum), which continues upward, and narrows to form a channel that penetrates the ethmoidal labyrinth and opens into the frontal sinus. This channel is for the frontonasal duct, which drains the frontal sinus.
The superior surface of the ethmoidal labyrinth articulates with the frontal bone, which usually completes the roof of the ethmoidal cells, while the anterior surface articulates with the frontal process of the maxilla and with the lacrimal bone. The inferior surface articulates with the upper medial margin of the maxilla.
A delicate irregularly shaped projection (the uncinate process) on the anterior aspect of the inferior surface of the ethmoidal labyrinth extends posteroinferiorly across a large defect (maxillary hiatus) in the medial wall of the maxilla to articulate with the inferior concha.
The cribriform plate is at the apex of the nasal cavities and fills the ethmoidal notch in the frontal bone (Fig. 8.221) and separates the nasal cavities below from the cranial cavity above. Small perforations in the bone allow the fibers of the olfactory nerve [I] to pass between the two regions.
A large triangular process (the crista galli) at the midline on the superior surface of the cribriform plate anchors a fold (falx cerebri) of dura mater in the cranial cavity.
The perpendicular plate of the ethmoid bone is quadrangular in shape, descends in the midline from the cribriform plate, and forms the upper part of the median nasal septum (Fig. 8.221). It articulates:

posteriorly with the sphenoidal crest on the body of the sphenoid bone;

anteriorly with the nasal spine on the frontal bone and with the site of articulation at the midline between the two nasal bones; and

inferiorly, anteriorly with the septal cartilage and posteriorly with the vomer.
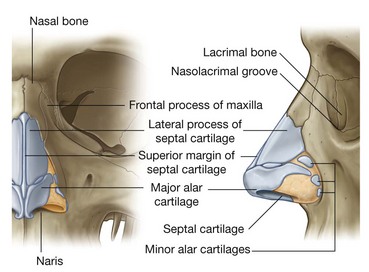
External nose
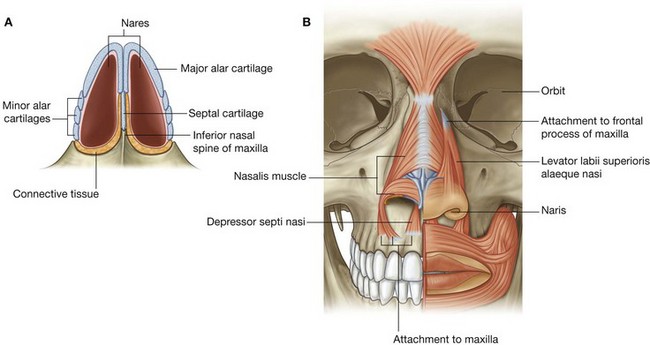
The external nose extends the nasal cavities onto the front of the face and positions the nares so that they point downward (Fig. 8.222). It is pyramidal in shape with its apex anterior in position. The upper angle of the nose between the openings of the orbits is continuous with the forehead.
Like posterior regions, the anterior parts of the nasal cavities found within the nose are held open by a skeletal framework, which is composed partly of bone and mainly of cartilage:

the bony parts are where the nose is continuous with the skull—here the nasal bones and parts of the maxillae and frontal bones provide support;

anteriorly, and on each side, support is provided by
lateral processes of the septal cartilage,
major alar and three or four
minor alar cartilages, and a single septal cartilage in the midline that forms the anterior part of the nasal septum.
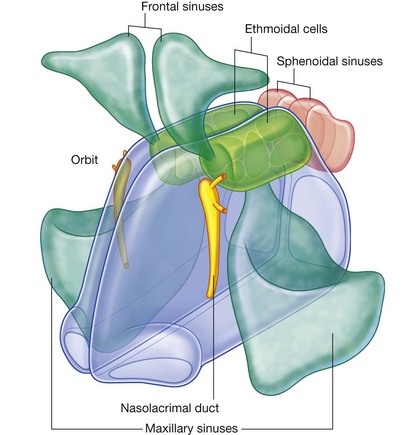
Paranasal sinuses
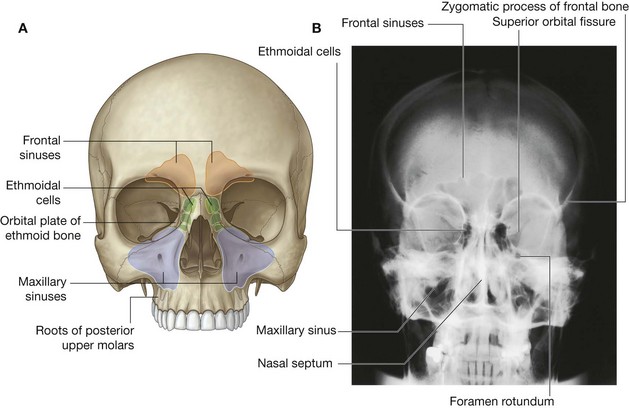
There are four paranasal air sinuses—the ethmoidal cells, and the sphenoidal, maxillary, and frontal sinuses (Fig. 8.223A and 8.223B). Each is named according to the bone in which it is found.
The paranasal sinuses develop as outgrowths from the nasal cavities and erode into the surrounding bones. All are:

lined by respiratory mucosa, which is ciliated and mucus secreting;

open into the nasal cavities; and

innervated by branches of the trigeminal nerve [V].
Frontal sinuses
The frontal sinuses, one on each side, are variable in size and are the most superior of the sinuses (Fig. 8.223A and 8.223B). Each is triangular in shape and is in the part of the frontal bone under the forehead. The base of each triangular sinus is oriented vertically in the bone at the midline above the bridge of the nose and the apex is laterally approximately one-third of the way along the upper margin of the orbit.
Each frontal sinus drains onto the lateral wall of the middle meatus via the frontonasal duct, which penetrates the ethmoidal labyrinth and continues as the ethmoidal infundibulum at the front end of the semilunar hiatus.
The frontal sinuses are innervated by branches of the supra-orbital nerve from the ophthalmic nerve [V1]. Their blood supply is from branches of the anterior ethmoidal arteries.
Ethmoidal cells
The ethmoidal cells on each side fill the ethmoidal labyrinth (Fig. 8.223A and 8.223B). Each cluster of cells is separated from the orbit by the thin orbital plate of the ethmoidal labyrinth, and from the nasal cavity by the medial wall of the ethmoidal labyrinth.
The ethmoidal cells are formed by a variable number of individual air chambers, which are divided into anterior, middle, and posterior ethmoidal cells based on the location of their apertures on the lateral wall of the nasal cavity:

the anterior ethmoidal cells open into the ethmoidal infundibulum or the frontonasal duct;

the middle ethmoidal cells open onto the ethmoidal bulla, or onto the lateral wall just above this structure;

the posterior ethmoidal cells open onto the lateral wall of the superior nasal meatus.
Because the ethmoidal cells often erode into bones beyond the boundaries of the ethmoidal labyrinth, their walls may be completed by the frontal, maxillary, lacrimal, sphenoid, and palatine bones.
The ethmoidal cells are innervated by:

the
anterior and
posterior ethmoidal branches of the nasociliary nerve from the ophthalmic nerve [V
1]; and

the maxillary nerve [V
2] via orbital branches from the pteryatine ganglion.
The ethmoidal cells receive their blood supply through branches of the anterior and posterior ethmoidal arteries.
Maxillary sinuses
The maxillary sinuses, one on each side, are the largest of the paranasal sinuses and completely fill the bodies of the maxillae (Fig. 8.223A and 8.223B). Each is pyramidal in shape with the apex directed laterally and the base deep to the lateral wall of the adjacent nasal cavity. The medial wall or base of the maxillary sinus is formed by the maxilla, and by parts of the inferior concha and palatine bone that overlie the maxillary hiatus.
The opening of the maxillary sinus is near the top of the base, in the center of the semilunar hiatus, which grooves the lateral wall of the middle nasal meatus.
Relationships of the maxillary sinus are as follows:

the superolateral surface (roof) is related above to the orbit;

the anterolateral surface is related below to the roots of the upper molar and premolar teeth and in front to the face;

the posterior wall is related behind to the infratemporal fossa.
The maxillary sinuses are innervated by infra-orbital and alveolar branches of the maxillary nerve [V2], and receive their blood through branches from the infra-orbital and superior alveolar branches of the maxillary arteries.
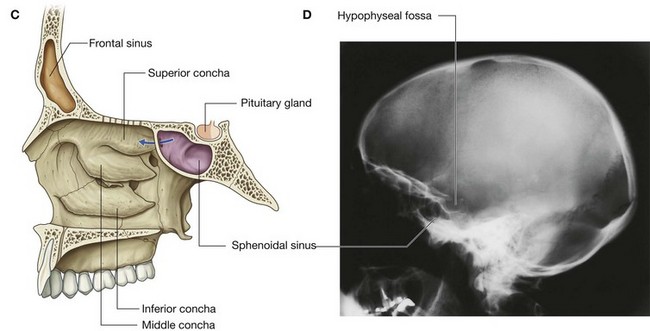
Sphenoidal sinuses
The sphenoidal sinuses, one on either side within the body of the sphenoid, open into the roof of the nasal cavity via apertures on the posterior wall of the spheno-ethmoidal recess (Fig. 8.223C and 8.223D). The apertures are high on the anterior walls of the sphenoid sinuses.
The sphenoidal sinuses are related:

above to the cranial cavity, particularly to the pituitary gland and to the optic chiasm;

laterally, to the cranial cavity, particularly to the cavernous sinuses; and

below and in front, to the nasal cavities.
Because only thin shelves of bone separate the sphenoidal sinuses from the nasal cavities below and hypophyseal fossa above, the pituitary gland can be surgically approached through the roof of the nasal cavities by passing first through the anteroinferior aspect of the sphenoid bone and into the sphenoidal sinuses and then through the top of the sphenoid bone into the hypophyseal fossa.
Innervation of the sphenoidal sinuses is provided by:

the posterior ethmoidal branch of the ophthalmic nerve [V
1]; and

the maxillary nerve [V
2] via orbital branches from the pteryatine ganglion.
The sphenoidal sinuses are supplied by branches of the pharyngeal arteries from the maxillary arteries.
Walls, floor, and roof
Medial wall
The medial wall of each nasal cavity is the mucosa-covered surface of the thin nasal septum, which is oriented vertically in the median sagittal plane and separates the right and left nasal cavities from each other.
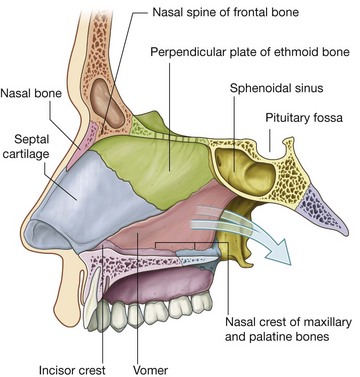
The nasal septum (Fig. 8.224) consists of:

the
septal nasal cartilage anteriorly;

posteriorly, mainly the vomer and the perpendicular plate of the ethmoid bone;

small contributions by the nasal bones where they meet in the midline, and the nasal spine of the frontal bone; and

contributions by the nasal crests of the maxillary and palatine bones, rostrum of the sphenoid bone, and the incisor crest of the maxilla.
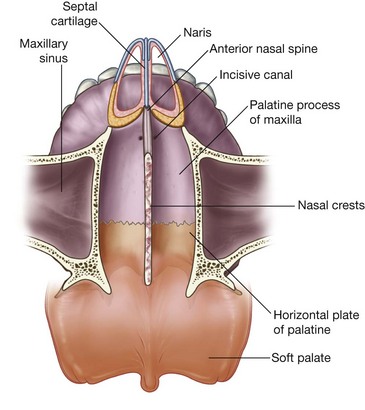
Floor
The floor of each nasal cavity (Fig. 8.225) is smooth, concave, and much wider than the roof. It consists of:

soft tissues of the external nose; and

the upper surface of the palatine process of the maxilla, and the horizontal plate of the palatine bone, which together form the hard palate.
The naris opens anteriorly into the floor, and the superior aperture of the incisive canal is deep to the mucosa immediately lateral to the nasal septum near the front of the hard palate.
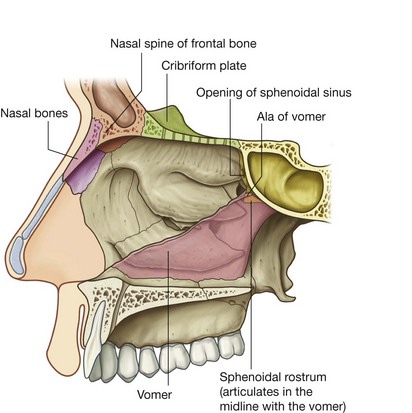
Roof
The roof of the nasal cavity is narrow and is highest in central regions where it is formed by the cribriform plate of the ethmoid bone (Fig. 8.226).
Anterior to the cribriform plate the roof slopes inferiorly to the nares and is formed by:

the nasal spine of the frontal bone and the nasal bones; and

the lateral processes of the septal cartilage and major alar cartilages of the external nose.
Posteriorly, the roof of each cavity slopes inferiorly to the choana and is formed by:

the anterior surface of the sphenoid bone;

the ala of the vomer and adjacent sphenoidal process of the palatine bone; and

the vaginal process of the medial plate of the pterygoid process.
Underlying the mucosa, the roof is perforated superiorly by openings in the cribriform plate, and anterior to these openings by a separate foramen for the anterior ethmoidal nerve and vessels.
The opening between the sphenoidal sinus and the spheno-ethmoidal recess is on the posterior slope of the roof.
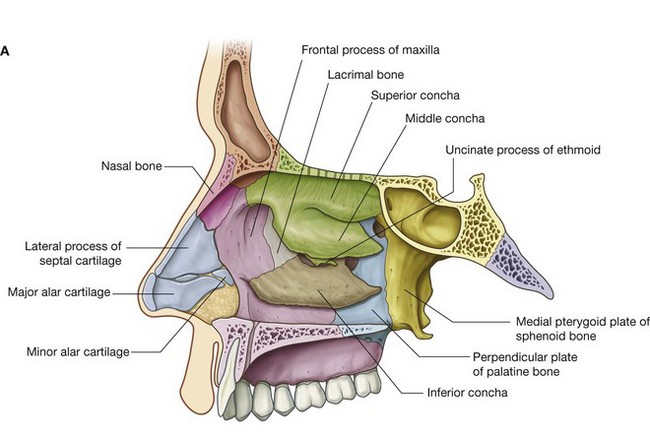
Lateral wall
The lateral wall of each nasal cavity is complex and is formed by bone, cartilage, and soft tissues.
Bony support for the lateral wall (Fig. 8.227A) is provided by:

the ethmoidal labyrinth and uncinate process;

the perpendicular plate of the palatine bone;

the medial plate of the pterygoid process of the sphenoid bone;

the medial surfaces of the lacrimal bones and maxillae; and

the inferior concha.
In the external nose, the lateral wall of the cavity is supported by cartilage (lateral process of the septal cartilage and major and minor alar cartilages) and by soft tissues. The surface of the lateral wall is irregular in contour and is interrupted by the three nasal conchae.
The inferior, middle, and superior conchae (Fig. 8.227B) extend medially across the nasal cavity, separating it into four air channels, an inferior, middle, and superior meatus, and a spheno-ethmoidal recess. The conchae do not extend forward into the external nose. The anterior end of each concha curves inferiorly to form a lip that overlies the end of the related meatus.
Immediately inferior to the attachment of the middle concha and just anterior to the midpoint of the concha, the lateral wall of the middle meatus elevates to form the dome-shaped ethmoidal bulla (Fig. 8.227C). This is formed by the underlying middle ethmoidal cells, which expand the medial wall of the ethmoidal labyrinth.
Inferior to the ethmoidal bulla is a curved gutter (the semilunar hiatus), which is formed by the mucosa covering the lateral wall as it spans a defect in the bony wall between the ethmoidal bulla above and the uncinate process below.
The anterior end of the semilunar hiatus forms a channel (the ethmoidal infundibulum), which curves upward and continues as the frontonasal duct through the anterior part of the ethmoidal labyrinth to open into the frontal sinus.
The nasolacrimal duct and most of the paranasal sinuses open onto the lateral wall of the nasal cavity:

the nasolacrimal duct opens onto the lateral wall of the inferior nasal meatus under the anterior lip of the inferior concha—it drains tears from the conjunctival sac of the eye into the nasal cavity and originates at the inferior end of the lacrimal sac on the anteromedial wall of the orbit;

the frontal sinus drains via the frontonasal duct and ethmoidal infundibulum into the anterior end of the semilunar hiatus on the lateral wall of the middle nasal meatus—the anterior ethmoidal cells drain into the frontonasal duct or ethmoidal infundibulum (in some cases, the frontal sinus drains directly into the anterior end of the middle nasal meatus and the frontonasal duct ends blindly in the anterior ethmoidal cells);

the middle ethmoidal cells open onto or just above the ethmoidal bulla;

the posterior ethmoidal cells usually open onto the lateral wall of the superior nasal meatus;

the large maxillary sinus opens into the semilunar hiatus, usually just inferior to the center of the ethmoidal bulla—this opening is near the roof of the maxillary sinus.
The only paranasal sinus that does not drain onto the lateral wall of the nasal cavity is the sphenoidal sinus, which usually opens onto the sloping posterior roof of the nasal cavity.
Nares
The nares are oval apertures on the inferior aspect of the external nose and are the anterior openings of the nasal cavities (Fig. 8.228A). They are held open by the surrounding alar cartilages and septal cartilage, and by the inferior nasal spine and adjacent margins of the maxillae.
Although the nares are continuously open, they can be widened further by the action of the related muscles of facial expression (nasalis muscle, depressor septi nasi, and levator labii superioris alaeque nasi; Fig. 8.228B).
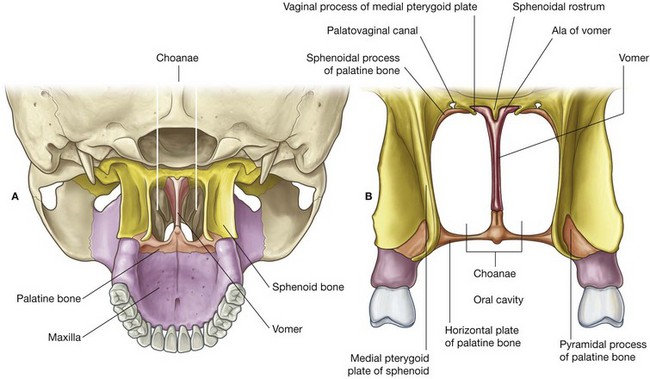
Choanae
The choanae are the oval-shaped openings between the nasal cavities and the nasopharynx (Fig. 8.229). Unlike the nares, which have flexible borders of cartilage and soft tissues, the choanae are rigid openings completely surrounded by bone, and their margins are formed:

inferiorly, by the posterior border of the horizontal plate of the palatine bone;

laterally, by the posterior margin of the medial plate of the pterygoid process; and

medially, by the posterior border of the vomer.
The roof of the choanae is formed:

anteriorly by the ala of the vomer and the vaginal process of the medial plate of the pterygoid process; and

posteriorly by the body of the sphenoid bone.
Gateways
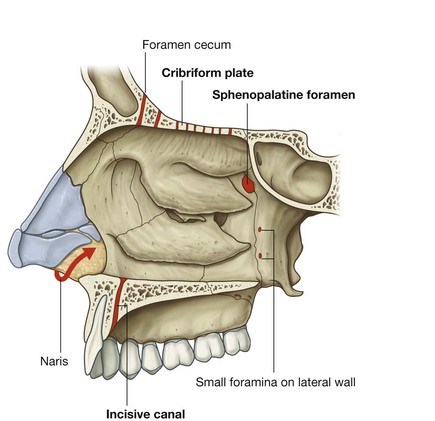
There are a number of routes by which nerves and vessels enter and leave the soft tissues lining each nasal cavity (Fig. 8.230), and these include the cribriform plate, sphenopalatine foramen, the incisive canal, small foramina in the lateral wall, and around the margin of the nares.
Cribriform plate
The fibers of the olfactory nerve [I] exit the nasal cavity and enter the cranial cavity through perforations in the cribriform plate. In addition, small foramina between the cribriform plate and surrounding bone allow the anterior ethmoidal nerve, a branch of the ophthalmic nerve [V1], and accompanying vessels to pass from the orbit into the cranial cavity and then down into the nasal cavity.
In addition, there is a connection in some individuals between nasal veins and the superior sagittal sinus of the cranial cavity through a prominent foramen (the foramen cecum) in the midline between the crista galli and frontal bone.
Sphenopalatine foramen
One of the most important routes by which nerves and vessels enter and leave the nasal cavity is the sphenopalatine foramen in the posterolateral wall of the superior nasal meatus. This foramen is just superior to the attachment of the posterior end of the middle nasal concha and is formed by the sphenopalatine notch in the palatine bone and the body of the sphenoid bone.
The sphenopalatine foramen is a route of communication between the nasal cavity and the pteryatine fossa. Major structures passing through the foramen are:

the sphenopalatine branch of the maxillary artery;

the nasopalatine branch of the maxillary nerve [V
2]; and

superior nasal branches of the maxillary nerve [V
2].
Incisive canal
Another route by which structures enter and leave the nasal cavities is through the incisive canal in the floor of each nasal cavity. This canal is immediately lateral to the nasal septum and just posterosuperior to the root of the central incisor in the maxilla. The two incisive canals, one on each side, both open into the single unpaired incisive fossa in the roof of the oral cavity and transmits:

the nasopalatine nerve from the nasal cavity into the oral cavity; and

the terminal end of the greater palatine artery from the oral cavity into the nasal cavity.
Small foramina in the lateral wall
Other routes by which vessels and nerves get into and out of the nasal cavity include the nares and small foramina in the lateral wall:

internal nasal branches of the infra-orbital nerve of the maxillary nerve [V
2] and alar branches of the nasal artery from the facial artery loop around the margin of the naris to gain entry to the lateral wall of the nasal cavity from the face;

inferior nasal branches from the greater palatine branch of the maxillary nerve [V
2] enter the lateral wall of the nasal cavity from the palatine canal by passing through small foramina on the lateral wall.
Vessels
The nasal cavities have a rich vascular supply for altering the humidity and temperature of respired air. In fact, the submucosa of the respiratory region, particularly that related to the conchae and septum, is often described as “erectile” or “cavernous” because the tissue enlarges or shrinks depending on the amount of blood flowing into the system.
Arteries
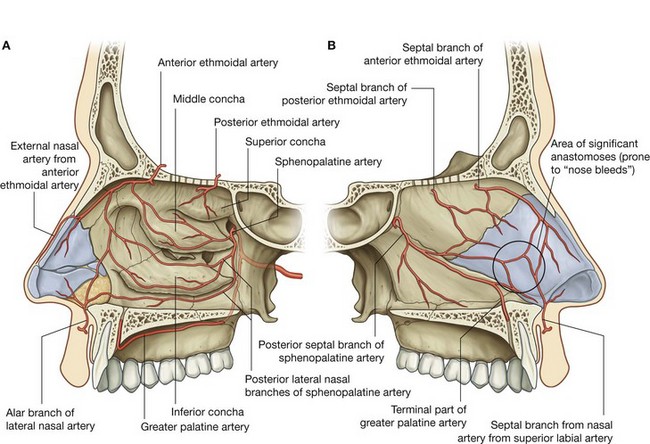
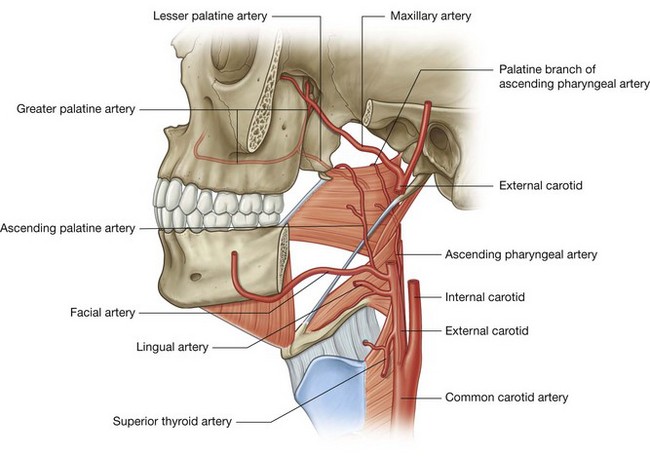
Arteries that supply the nasal cavity include vessels that originate from both the internal and external carotid arteries (Fig. 8.231):

vessels that originate from branches of the external carotid artery include the sphenopalatine, greater palatine, superior labial, and lateral nasal arteries;

vessels that originate from branches of the internal carotid artery are the anterior and posterior ethmoidal arteries.
Sphenopalatine artery
The largest vessel supplying the nasal cavity is the sphenopalatine artery (Fig. 8.231), which is the terminal branch of the maxillary artery in the pteryatine fossa. It leaves the pteryatine fossa and enters the nasal cavity by passing medially through the sphenopalatine foramen and onto the lateral wall of the nasal cavity.
Posterior lateral nasal branches supply a large part of the lateral wall and anastomose anteriorly with branches from the anterior and posterior ethmoidal arteries, and with lateral nasal branches of the facial artery.
Posterior septal branches of the sphenopalatine artery pass over the roof of the cavity and onto the nasal septum where they contribute to the blood supply of the medial wall. One of these latter branches continues forward down the nasal septum to anastomose with the terminal end of the greater palatine artery and septal branches of the superior labial artery.
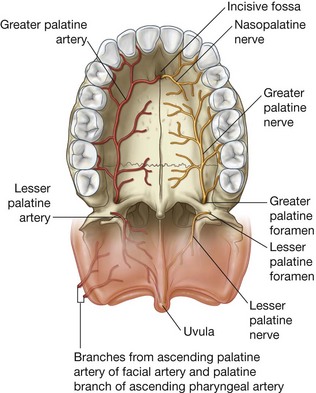
Greater palatine artery
The terminal end of the greater palatine artery enters the anterior aspect of the floor of the nasal cavity by passing up through the incisive canal from the roof of the oral cavity (Fig. 8.231).
Like the sphenopalatine artery, the greater palatine artery arises in the pteryatine fossa as a branch of the maxillary artery. It passes first onto the roof of the oral cavity by passing down through the palatine canal and greater palatine foramen to the posterior aspect of the palate, then passes forward on the undersurface of the palate, and up through the incisive fossa and canal to reach the floor of the nasal cavity. The greater palatine artery supplies anterior regions of the medial wall and adjacent floor of the nasal cavity, and anastomoses with the septal branch of the sphenopalatine artery.
Superior labial and lateral nasal arteries
The superior labial artery and the lateral nasal artery originate from the facial artery on the front of the face.
The superior labial artery originates from the facial artery near the lateral end of the oral fissure and passes medially in the lip, supplying the lip and giving rise to branches that supply the nose and nasal cavity. An alar branch supplies the region around the lateral aspect of the naris and a septal branch passes into the nasal cavity and supplies anterior regions of the nasal septum.
The lateral nasal artery originates from the facial artery in association with the margin of the external nose and contributes to the blood supply of the external nose. Alar branches pass around the lateral margin of the naris and supply the nasal vestibule.
Anterior and posterior ethmoidal arteries
The anterior and posterior ethmoidal arteries (Fig. 8.231) originate in the orbit from the ophthalmic artery, which originates in the cranial cavity as a major branch of the internal carotid artery. They pass through canals in the medial wall of the orbit between the ethmoidal labyrinth and frontal bone, supply the adjacent paranasal sinuses, and then enter the cranial cavity immediately lateral and superior to the cribriform plate.
The posterior ethmoidal artery descends into the nasal cavity through the cribriform plate and has branches to the upper parts of the medial and lateral walls.
The anterior ethmoidal artery passes forward, with the accompanying anterior ethmoidal nerve, in a groove on the cribriform plate and enters the nasal cavity by descending through a slit-like foramen immediately lateral to the crista galli. It gives rise to branches that supply the medial (septal) and lateral wall of the nasal cavity and then continues forward on the deep surface of the nasal bone, and terminates by passing between the nasal bone and lateral nasal cartilage to emerge on the external nose as the external nasal branch to supply skin and adjacent tissues.
Vessels that supply the nasal cavities form extensive anastomoses with each other. This is particularly evident in the anterior region of the medial wall where there are anastomoses between branches of the greater palatine, sphenopalatine, superior labial, and anterior ethmoidal arteries, and where the vessels are relatively close to the surface (Fig. 8.231B). This area is the major site of nosebleeds, or epistaxis.
Veins
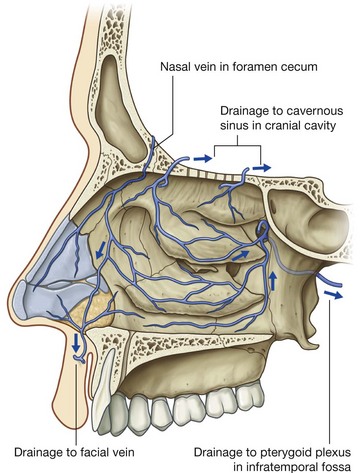
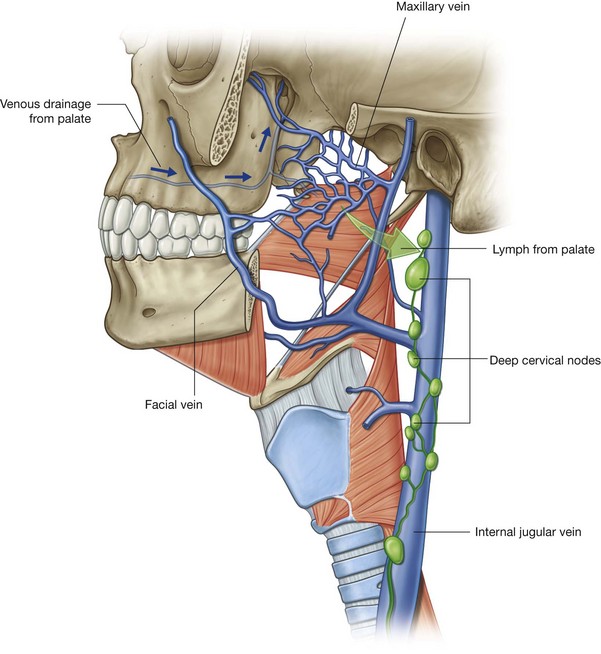
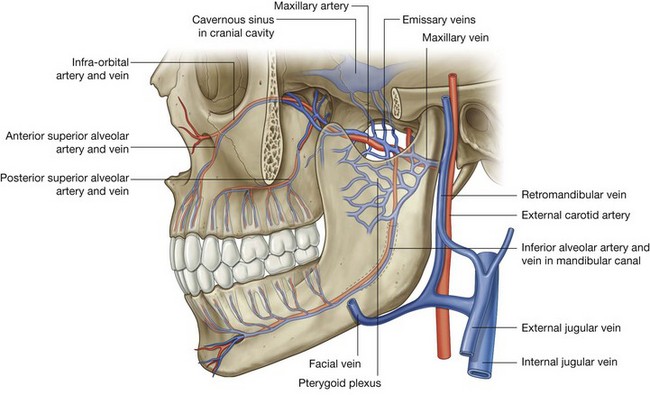
Veins draining the nasal cavities generally follow the arteries (Fig. 8.232):

veins that pass with branches that ultimately originate from the maxillary artery drain into the pterygoid plexus of veins in the infratemporal fossa;

veins from anterior regions of the nasal cavities join the facial vein.
In some individuals, an additional nasal vein passes superiorly through a midline aperture (the foramen cecum), in the frontal bone anterior to the crista galli, and joins with the anterior end of the superior sagittal sinus. Because this nasal vein connects an intracranial venous sinus with extracranial veins, it is classified as an emissary vein. Emissary veins in general are routes by which infections can track from peripheral regions into the cranial cavity.
Veins that accompany the anterior and posterior ethmoidal arteries are tributaries of the superior ophthalmic vein, which is one of the largest emissary veins and drains into the cavernous sinus on either side of the hypophyseal fossa.
Innervation
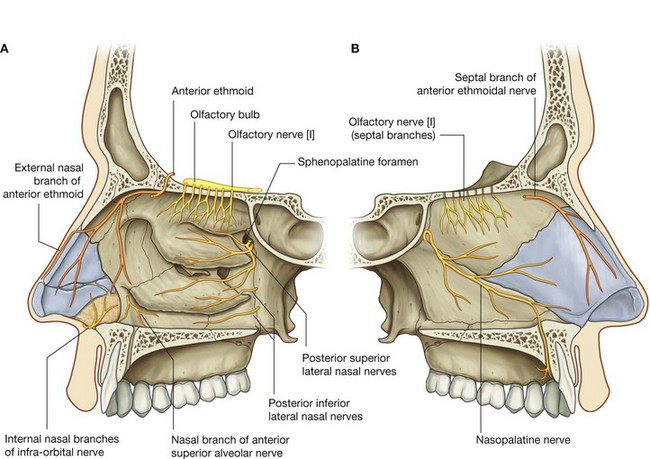
Nerves that innervate the nasal cavities (Fig. 8.233) are:

the olfactory nerve [I] for olfaction; and

branches of the ophthalmic [V
1] and maxillary [V
2] nerves for general sensation.
Secretomotor innervation of mucous glands in the nasal cavities and paranasal sinuses is by parasympathetic fibers from the facial nerve [VII], which mainly join branches of the maxillary nerve [V2] in the pteryatine fossa.
Olfactory nerve [I]
The olfactory nerve [I] is composed of axons from receptors in the olfactory epithelium at the top of each nasal cavity. Bundles of these axons pass superiorly through perforations in the cribriform plate to synapse with neurons in the olfactory bulb of the brain.
Branches from the ophthalmic nerve [V1]
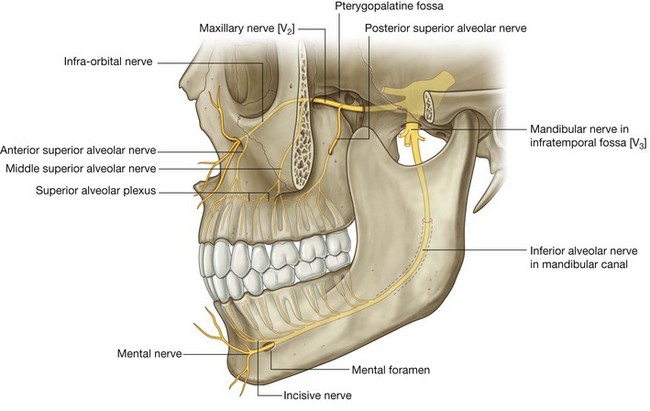
Branches from the ophthalmic nerve [V1] that innervate the nasal cavity are the anterior and posterior ethmoidal nerves, which originate from the nasociliary nerve in the orbit.
Anterior and posterior ethmoidal nerves
The anterior ethmoidal nerve (Fig. 8.233) travels with the anterior ethmoidal artery and leaves the orbit through a canal between the ethmoidal labyrinth and the frontal bone. It passes through and supplies the adjacent ethmoidal cells and frontal sinus, and then enters the cranial cavity immediately lateral and superior to the cribriform plate.
The anterior ethmoidal nerve travels forward in a groove on the cribriform plate and then enters the nasal cavity by descending through a slit-like foramen immediately lateral to the crista galli. It has branches to the medial and lateral wall of the nasal cavity and then continues forward on the undersurface of the nasal bone. It passes onto the external surface of the nose by traveling between the nasal bone and lateral nasal cartilage, and then terminates as the external nasal nerve, which supplies skin around the naris, in the nasal vestibule, and on the tip of the nose.
Like the anterior ethmoidal nerve, the posterior ethmoidal nerve leaves the orbit through a similar canal in the medial wall of the orbit. It terminates by supplying the mucosa of the ethmoidal cells and sphenoidal sinus and normally does not extend into the nasal cavity itself.
Branches from the maxillary nerve [V2]
A number of nasal branches from the maxillary nerve [V2] innervate the nasal cavity. Many of these nasal branches (Fig. 8.233) originate in the pteryatine fossa, which is just lateral to the lateral wall of the nasal cavity, and leave the fossa to enter the nasal cavity by passing medially through the sphenopalatine foramen:

a number of these nerves (
posterior superior lateral nasal nerves) pass forward on and supply the lateral wall of the nasal cavity;

others (
posterior superior medial nasal nerves) cross the roof to the nasal septum and supply both these regions;

the largest of these nerves is the
nasopalatine nerve, which passes forward and down the medial wall of the nasal cavity to pass through the incisive canal onto the roof of the oral cavity, and terminates by supplying the oral mucosa posterior to the incisor teeth;

other nasal nerves (
posterior inferior nasal nerves) originate from the greater palatine nerve, descending from the pteryatine fossa in the palatine canal just lateral to the nasal cavity, and pass through small bony foramina to innervate the lateral wall of the nasal cavity;

a small nasal nerve also originates from the anterior superior alveolar branch of the infra-orbital nerve and passes medially through the maxilla to supply the lateral wall near the anterior end of the inferior concha.
Parasympathetic innervation
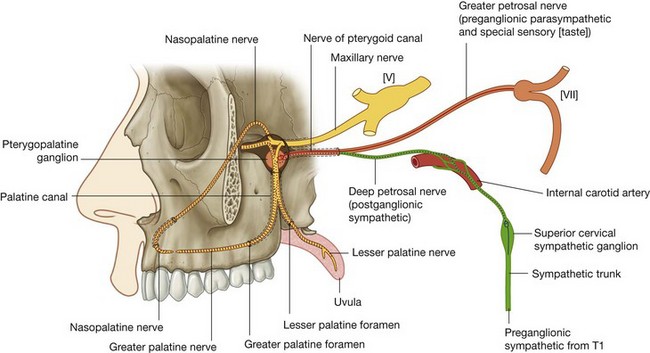
Secretomotor innervation of glands in the mucosa of the nasal cavity and paranasal sinuses is by preganglionic parasympathetic fibers carried in the greater petrosal branch of the facial nerve [VII]. These fibers enter the pteryatine fossa and synapse in the pteryatine ganglion (see p. 940). Postganglionic parasympathetic fibers then join branches of the maxillary nerve [V2] to leave the fossa and ultimately reach target glands.
Sympathetic innervation
Sympathetic innervation, mainly involved with regulating blood flow in the nasal mucosa, is from the spinal cord level T1. Preganglionic sympathetic fibers enter the sympathetic trunk and ascend to synapse in the superior cervical sympathetic ganglion. Postganglionic sympathetic fibers pass onto the internal carotid artery, enter the cranial cavity, and then leave the internal carotid artery to form the deep petrosal nerve, which joins the greater petrosal nerve of the facial nerve [VII] and enters the pteryatine fossa (see Fig. 8.148 and p. 945).
Like the parasympathetic fibers, the sympathetic fibers follow branches of the maxillary nerve [V2] into the nasal cavity.
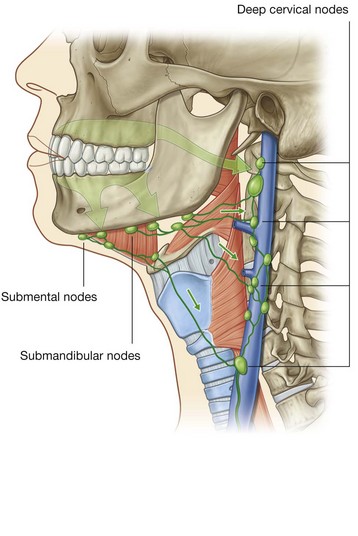
Lymphatics
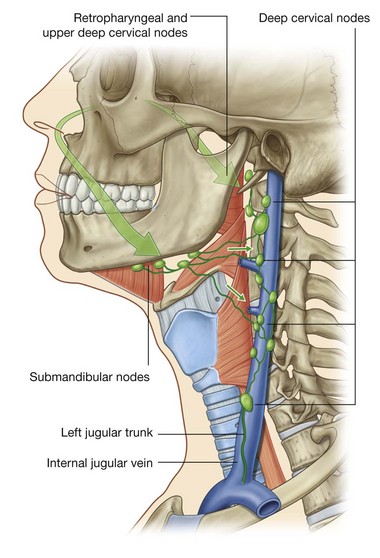
Lymph from anterior regions of the nasal cavities drains forward onto the face by passing around the margins of the nares (Fig. 8.234). These lymphatics ultimately connect with the submandibular nodes.
Lymph from posterior regions of the nasal cavity and the paranasal sinuses drains into upper deep cervical nodes. Some of this lymph passes first through the retropharyngeal nodes.
ORAL CAVITY
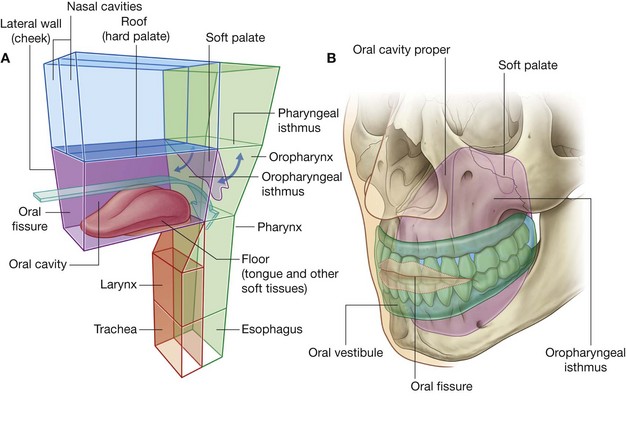
The oral cavity is inferior to the nasal cavities (Fig. 8.235A). It has a roof and floor, and lateral walls, opens onto the face through the oral fissure, and is continuous with the cavity of the pharynx at the oropharyngeal isthmus.
The roof of the oral cavity consists of the hard and soft palates. The floor is formed mainly of soft tissues, which include a muscular diaphragm and the tongue. The lateral walls (cheeks) are muscular and merge anteriorly with the lips surrounding the oral fissure (the anterior opening of the oral cavity).
The posterior aperture of the oral cavity is the oropharyngeal isthmus, which opens into the oral part of the pharynx.
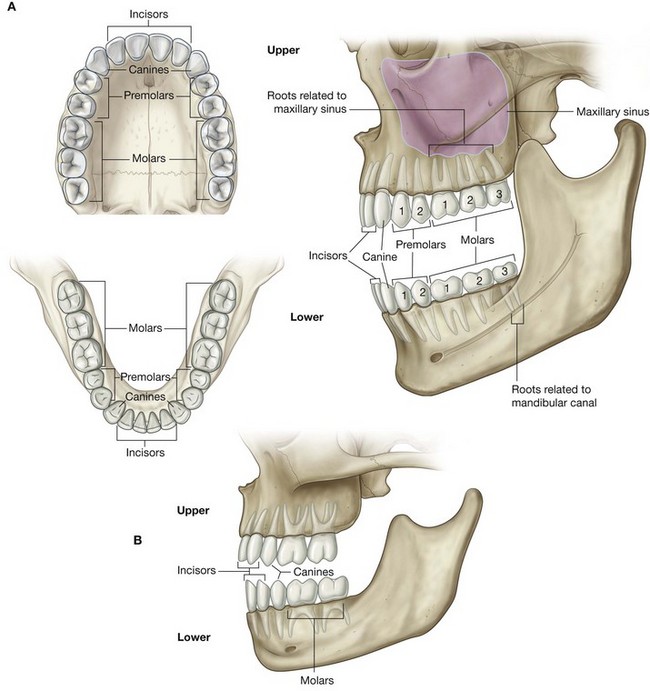
The oral cavity is separated into two regions by the upper and lower dental arches consisting of the teeth and alveolar bone that supports them (Fig. 8.235B):

the outer
oral vestibule, which is horseshoe-shaped, is between the dental arches and the deep surfaces of the cheeks and lips—the oral fissure opens into it and can be opened and closed by muscles of facial expression, and by movements of the lower jaw;

the inner
oral cavity proper, which is enclosed by the dental arches.
The degree of separation between the upper and lower arches is established by elevating or depressing the lower jaw (mandible) at the temporomandibular joint.
The oropharyngeal isthmus at the back of the oral cavity proper can be opened and closed by surrounding soft tissues, which include the soft palate and tongue.
The oral cavity has multiple functions:

it is the inlet for the digestive system involved with the initial processing of food, which is aided by secretions from salivary glands;

it manipulates sounds produced by the larynx and one outcome of this is speech;

it can be used for breathing because it opens into the pharynx, which is a common pathway for food and air. For this reason, the oral cavity can be used by physicians to access the lower airway, and dentists use “rubber dams” to prevent debris such as tooth fragments from passing through the oropharyngeal isthmus and pharynx into either the esophagus or the lower airway.
Multiple nerves innervate the oral cavity
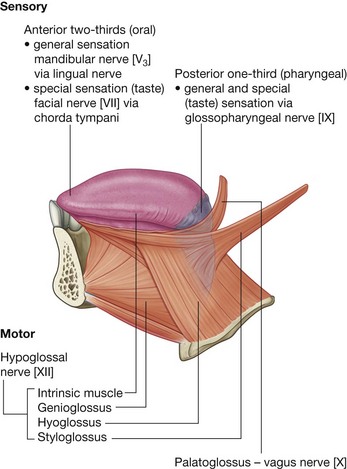
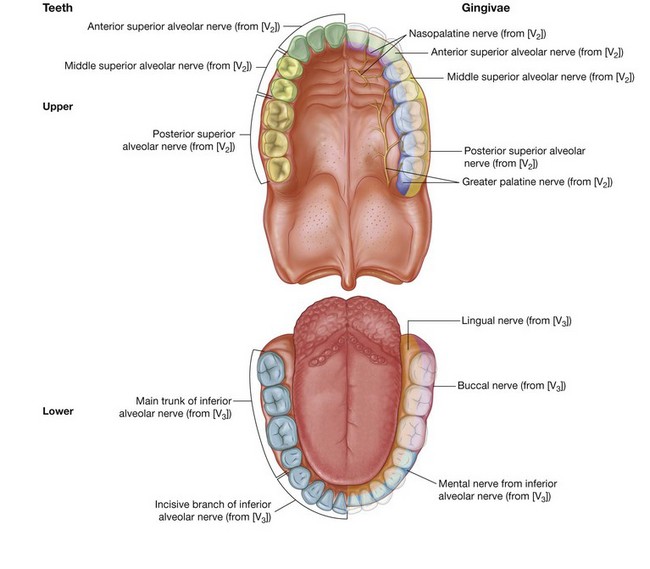
General sensory innervation is carried predominantly by branches of the trigeminal nerve [V]:

the upper parts of the cavity, including the palate and the upper teeth, are innervated by branches of the maxillary nerve [V
2];

the lower parts, including the teeth and oral part of the tongue, are innervated by branches of the mandibular nerve [V
3];

taste (special afferent-SA) from the oral part or anterior two-thirds of the tongue is carried by branches of the facial nerve [VII], which join and are distributed with branches of the trigeminal nerve [V];

parasympathetic fibers to the glands within the oral cavity are also carried by branches of the facial nerve [VII], which are distributed with branches of the trigeminal nerve [V];

sympathetic fibers in the oral cavity ultimately come from spinal cord level T1, synapse in the superior cervical sympathetic ganglion, and are eventually distributed to the oral cavity along branches of the trigeminal nerve [V] or directly along blood vessels.
All muscles of the tongue are innervated by the hypoglossal nerve [XII], except the palatoglossus, which is innervated by the vagus nerve [X].
All muscles of the soft palate are innervated by the vagus nerve [X] except for the tensor veli palatini, which is innervated by a branch from the mandibular nerve [V3]. The muscle (mylohyoid) that forms the floor of the oral cavity is also innervated by the mandibular nerve [V3].
Skeletal framework
Bones that contribute to the skeletal framework of the oral cavity or are related to the anatomy of structures in the oral cavity include:

the paired maxillae, palatine and temporal bones; and

the unpaired mandible, sphenoid, and hyoid bone.
In addition, the cartilaginous parts of the pharyngotympanic tubes on the inferior aspect of the base of the skull are related to the attachment of muscles of the soft palate.
Maxillae
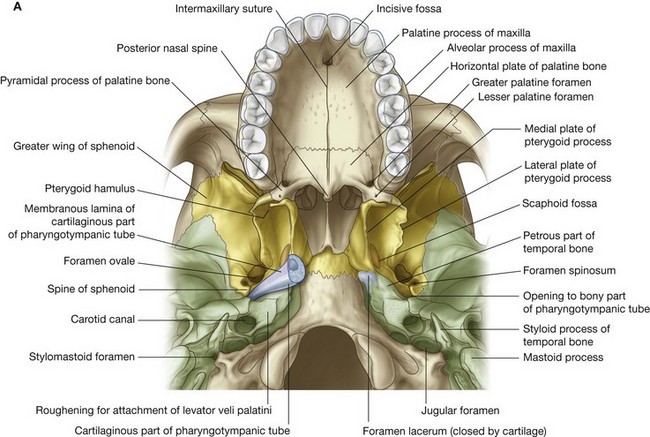
The two maxillae contribute substantially to the architecture of the roof of the oral cavity. The parts involved are the alveolar and palatine processes (Fig. 8.236A).
The palatine process is a horizontal shelf that projects from the medial surface of each maxilla. It originates just superior to the medial aspect of the alveolar process and extends to the midline where it is joined, at a suture, with the palatine process from the other side. Together, the two palatine processes form the anterior two-thirds of the hard palate.
In the midline on the inferior surface of the hard palate and at the anterior end of the intermaxillary suture is a single small fossa (incisive fossa) just behind the incisor teeth. Two incisive canals, one on each side, extend posterosuperiorly from the roof of this fossa to open onto the floor of the nasal cavity. The canals and fossae allow passage of the greater palatine vessels and the nasopalatine nerves.
Palatine bones
The parts of each L-shaped palatine bone that contribute to the roof of the oral cavity are the horizontal plate and the pyramidal process (Fig. 8.236A).
The horizontal plate projects medially from the inferior aspect of the palatine bone and is joined by sutures to its partner in the midline and, on the same side, with the palatine process of the maxilla anteriorly.
A single posterior nasal spine is formed at the midline where the two horizontal plates join and projects backward from the margin of the hard palate. The posterior margin of the horizontal plates and the posterior nasal spine are associated with attachment of the soft palate.
The greater palatine foramen, formed mainly by the horizontal plate of the palatine bone and completed laterally by the adjacent part of the maxilla, opens onto the posterolateral aspect of the horizontal plate. This foramen is the inferior opening of the palatine canal, which continues superiorly into the pteryatine fossa and transmits the greater palatine nerve and vessels to the palate.
Also opening onto the palatine bone is the lesser palatine foramen. This foramen is the inferior opening of the lesser palatine canal, which branches from the greater palatine canal and transmits the lesser palatine nerve and vessels to the soft palate.
The pyramidal process projects posteriorly and fills the space between the inferior ends of the medial and lateral plates of the pterygoid process of the sphenoid bone.
Sphenoid bone
The pterygoid processes and spines of the sphenoid bone are associated with structures related to the soft palate, which forms part of the roof of the oral cavity (Fig. 8.236A).
The pterygoid processes descend, one on each side, from the lateral aspect of the body of the sphenoid bone. Each process has a medial and a lateral plate. These two vertically oriented plates project from the posterior aspect of the process. The V-shaped gap that occurs inferiorly between the two plates is filled by the pyramidal process of the palatine bone.
Projecting posterolaterally from the inferior margin of the medial plate of the pterygoid process is an elongate hook-shaped structure (the pterygoid hamulus). This hamulus is immediately behind the alveolar arch and inferior to the posterior margin of the hard palate. It is:

a “pulley” for one of the muscles (tensor veli palatini) of the soft palate; and

the attachment site for the upper end of the pterygomandibular raphe, which is attached below to the mandible and joins together the superior constrictor of the pharynx and the buccinator muscle of the cheek.
At the root of the medial plate of the pterygoid process on the base of the skull is a small canoe-shaped fossa (scaphoid fossa), which begins just medial to the foramen ovale and descends anteriorly and medially to the root of the medial plate of the pterygoid process (Fig. 8.236A). This fossa is for the attachment of one of the muscles of the soft palate (tensor veli palatini).
The spines of the sphenoid, one on each side, are vertical projections from the inferior surfaces of the greater wings of the sphenoid bone (Fig. 8.236A). Each spine is immediately posteromedial to the foramen spinosum.
The medial aspect of the spine provides attachment for the most lateral part of the tensor veli palatini muscle of the soft palate.
Temporal bone
The styloid process and inferior aspect of the petrous part of the temporal bone provide attachment for muscles associated with the tongue and soft palate, respectively.
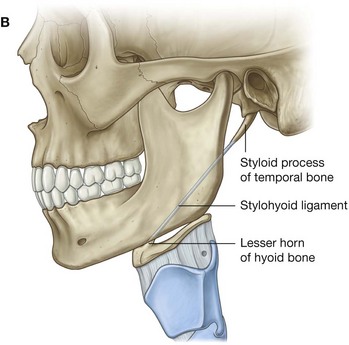
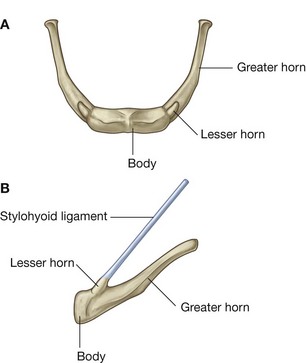
The styloid process projects anteroinferiorly from the underside of the temporal bone. It can be as long as 1 inch (2.5 cm) and points toward the lesser horn of the hyoid bone to which it is attached by the stylohyoid ligament (Fig. 8.236B). The root of the styloid process is immediately anterior to the stylomastoid foramen and lateral to the jugular foramen. The styloglossus muscle of the tongue attaches to the anterolateral surface of the styloid process.
The inferior aspect of the temporal bone has a triangular roughened area immediately anteromedial to the opening of the carotid canal (Fig. 8.236A). The levator veli palatini muscle of the soft palate is attached here.
Cartilaginous part of the pharyngotympanic tube
The trumpet-shaped cartilaginous part of the pharyngotympanic tube is in a groove between the anterior margin of the petrous part of the temporal bone and the posterior margin of the greater wing of the sphenoid (Fig. 8.236A).
The medial and lateral walls of the cartilaginous part of the pharyngotympanic tube are formed mainly of cartilage, whereas the more inferolateral wall is more fibrous and is known as the membranous lamina.
The apex of the cartilaginous part of the pharyngotympanic tube connects laterally to the opening of the bony part in the temporal bone.
The expanded medial end of the cartilaginous part of the pharyngotympanic tube is immediately posterior to the upper margin of the medial plate of the pterygoid process and opens into the nasopharynx.
The cartilaginous part of the pharyngotympanic tube is lateral to the attachment of the levator veli palatini muscle to the petrous part of the temporal bone and medial to the spine of the sphenoid. The tensor veli palatini muscle is attached, in part, to the membranous lamina.
Mandible
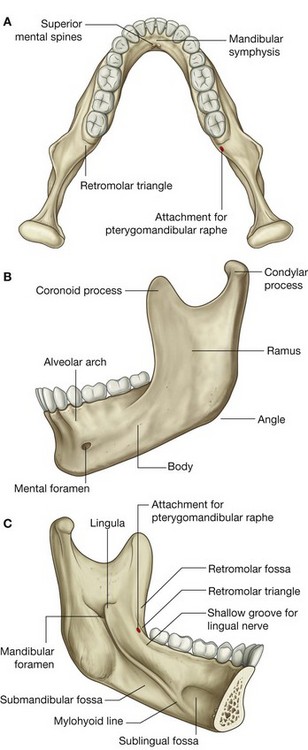
The mandible is the bone of the lower jaw (Fig. 8.237). It consists of a body of right and left parts, which are fused anteriorly in the midline (mandibular symphysis), and two rami. The site of fusion is particularly visible on the external surface of the bone as a small vertical ridge in the midline.
The upper surface of the body of mandible bears the alveolar arch, which anchors the lower teeth, and on its external surface on each side is a small mental foramen.
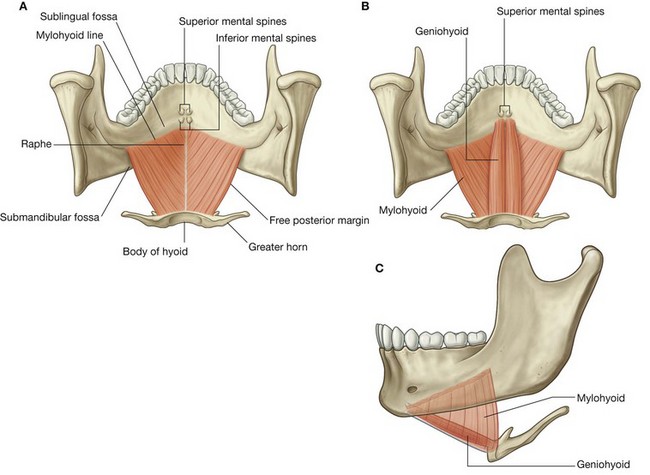
Posterior to the mandibular symphysis on the internal surface of the mandible are two pairs of small spines, one pair immediately above the other pair. These are the superior and inferior mental spines (superior and inferior genial spines), and are attachment sites for a pair of muscles that pass into the tongue and a pair of muscles that connect the mandible to the hyoid bone.
Extending from the midline and originating inferior to the mental spines is a raised line or ridge (the mylohyoid line), which runs posteriorly and superiorly along the internal surface of each side of the body of the mandible to end just below the level of the last molar tooth.
Above the anterior one-third of the mylohyoid line is a shallow depression (the sublingual fossa), and below the posterior two-thirds of the mylohyoid line is another depression (the submandibular fossa).
Between the last molar tooth and the mylohyoid line is a shallow groove for the lingual nerve.
Immediately posterior to the last molar tooth on the medial upper surface of the body of mandible is a small triangular depression (retromolar triangle). The pterygomandibular raphe attaches just medial to the apex of this triangle and extends from here to the tip of the pterygoid hamulus above.
The ramus of mandible, one on each side, is quadrangular shaped and oriented in the sagittal plane. On the medial surface of the ramus is a large mandibular foramen for transmission of the inferior alveolar nerve and vessels.
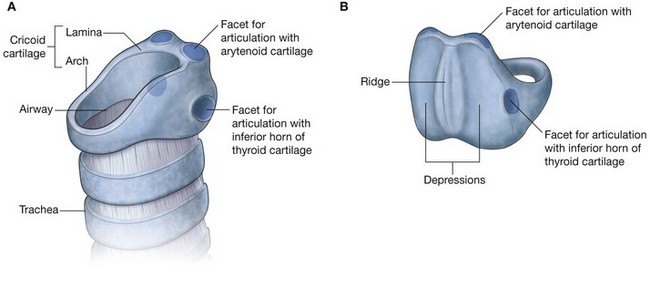
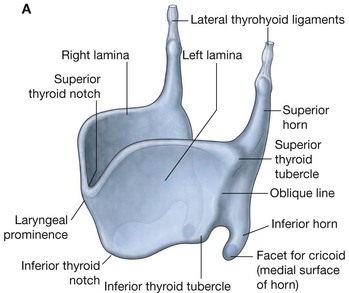
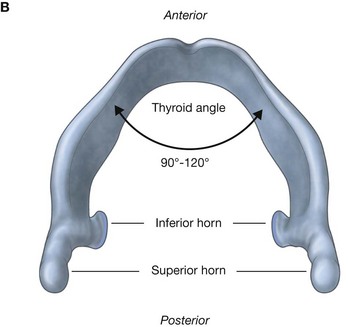
Hyoid bone
The hyoid bone is a small U-shaped bone in the neck between the larynx and the mandible. It has an anterior body of hyoid bone and two large greater horns, one on each side, which project posteriorly and superiorly from the body (Fig. 8.238). There are two small conical lesser horns on the superior surface where the greater horns join with the body. The stylohyoid ligaments attach to the apices of the lesser horns.
The hyoid bone is a key bone in the neck because it connects the floor of the oral cavity in front with the pharynx behind and the larynx below.
Walls: the cheeks
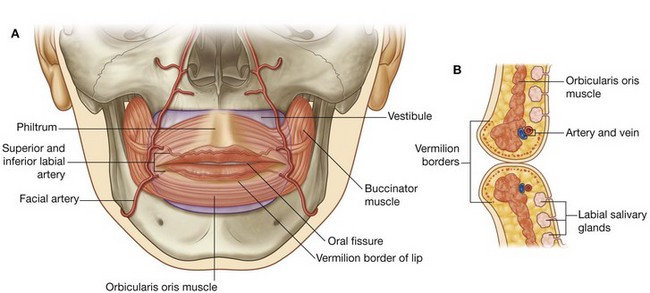
The walls of the oral cavity are formed by the cheeks.
Each cheek consists of fascia and a layer of skeletal muscle sandwiched between skin externally and oral mucosa internally. The thin layer of skeletal muscle within the cheeks is principally the buccinator muscle.
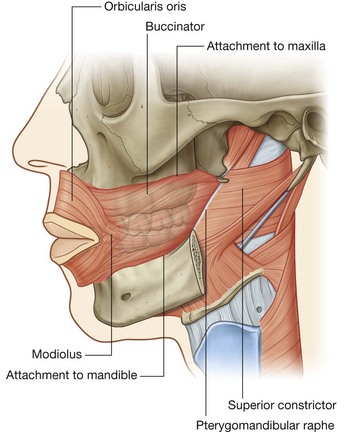
Buccinator
The buccinator muscle is one of the muscles of facial expression (see p. 861 and Fig. 8.239). It is in the same plane as the superior constrictor muscle of the pharynx. In fact, the posterior margin of the buccinator muscle is joined to the anterior margin of the superior constrictor muscle by the pterygomandibular raphe, which runs between the tip of the pterygoid hamulus of the sphenoid bone above and a roughened area of bone immediately behind the last molar tooth on the mandible below.
The buccinator and superior constrictor muscles therefore provide continuity between the walls of the oral and pharyngeal cavities.
The buccinator muscle, in addition to originating from the pterygomandibular raphe, also originates directly from the alveolar part of the mandible and alveolar process of the maxilla.
From its three sites of origin, the muscle fibers of the buccinator run forward to blend with those of the orbicularis oris muscle and to insert into the modiolus, which is a small button-shaped nodule of connective tissue at the interface between the muscles of the lips and cheeks on each side.
The buccinator muscle holds the cheeks against the alveolar arches and keeps food between the teeth when chewing.
The buccinator is innervated by the buccal branch of the facial nerve [VII]. General sensation from the skin and oral mucosa of the cheeks is carried by the buccal branch of the mandibular nerve [V3].
Floor
The floor of the oral cavity proper is formed mainly by three structures:

a muscular diaphragm, which fills the
U-shaped gap between the left and right sides of the body of the mandible and is composed of the paired mylohyoid muscles;

two cord-like geniohyoid muscles above the diaphragm, which run from the mandible in front to the hyoid bone behind;

the tongue, which is superior to the geniohyoid muscles.
Also present in the floor of the oral cavity proper are salivary glands and their ducts. The largest of these glands, on each side, are the sublingual gland and the oral part of the submandibular gland.
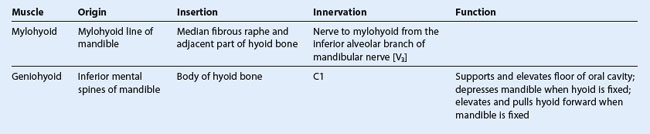
Mylohyoid muscles
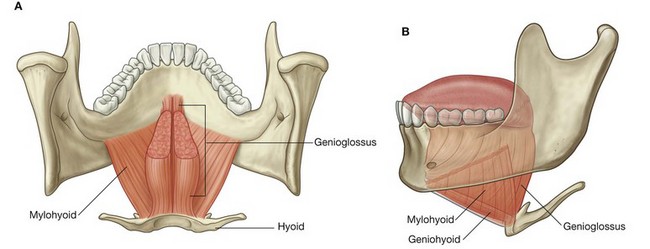
The two thin mylohyoid muscles (Table 8.20), one on each side, together form a muscular diaphragm that defines the inferior limit of the floor of the oral cavity (Fig. 8.240). Each muscle is triangular in shape with its apex pointed forward.
The lateral margin of each triangular muscle is attached to the mylohyoid line on the medial side of the body of the mandible. From here, the muscle fibers run slightly downward to the medial margin at the midline where the fibers are joined together with those of their partner muscle on the other side by a raphe. The raphe extends from the posterior aspect of the mandibular symphysis in front to the body of the hyoid bone behind.
The posterior margin of each mylohyoid muscle is free except for a small medial attachment to the hyoid bone.
The mylohyoid muscles:

contribute structural support to the floor of the oral cavity;

participate in elevating and pulling forward the hyoid bone, and therefore the attached larynx, during the initial stages of swallowing; and

when the hyoid bone is fixed in position, depress the mandible and open the mouth.
Like the muscles of mastication, the mylohyoid muscles are innervated by the mandibular nerve [V3]. The specific branch that innervates the mylohyoid muscles is the nerve to mylohyoid from the inferior alveolar nerve.
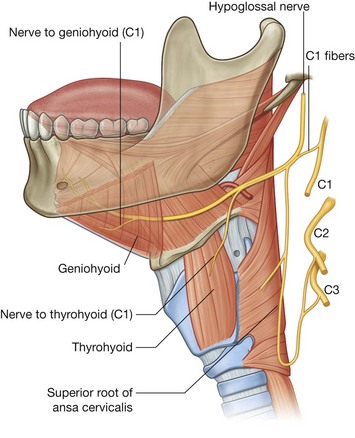
Geniohyoid muscles
The geniohyoid muscles (Table 8.20) are paired cord-like muscles that run, one on either side of the midline, from the inferior mental spines on the posterior surface of the mandibular symphysis to the anterior surface of the body of hyoid bone (Fig. 8.240B&C). They are immediately superior to the mylohyoid muscle in the floor of the mouth and inferior to the genioglossus muscles that form part of the root of the tongue.
The geniohyoid muscles:

mainly pull the hyoid bone, and therefore the attached larynx, up and forward during swallowing; and

because they pass posteroinferiorly from the mandible to the hyoid bone, when the hyoid bone is fixed, they can act with the mylohyoid muscles to depress the mandible and open the mouth.
Unlike other muscles that move the mandible at the temporomandibular joint, the geniohyoid muscles are innervated by a branch of cervical nerve C1, which “hitchhikes” from the neck along the hypoglossal nerve [XII] into the floor of the oral cavity.
Gateway into the floor of the oral cavity
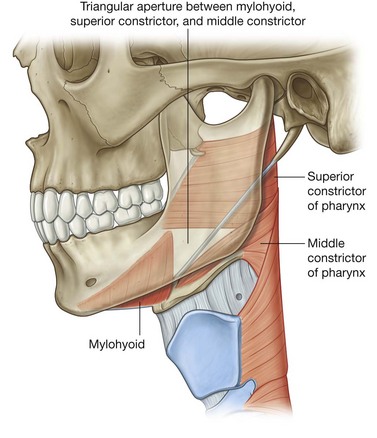
In addition to defining the lower limit of the floor of the oral cavity, the free posterior border of the mylohyoid muscle on each side forms one of the three margins of a large triangular aperture, which is a major route by which structures in the upper neck and infratemporal fossa of the head pass to and from structures in the floor of the oral cavity (Fig. 8.241). The other two muscles that complete the margins of the aperture are the superior and middle constrictor muscles of the pharynx.
Most structures that pass through the aperture are associated with the tongue and include muscles (hyoglossus, styloglossus), vessels (lingual artery and vein), nerves (lingual, hypoglossal [XII], glossopharyngeal [IX]), and lymphatics.
A large salivary gland (the submandibular gland) is “hooked” around the free posterior margin of the mylohyoid muscle and therefore also passes through the opening.
Tongue
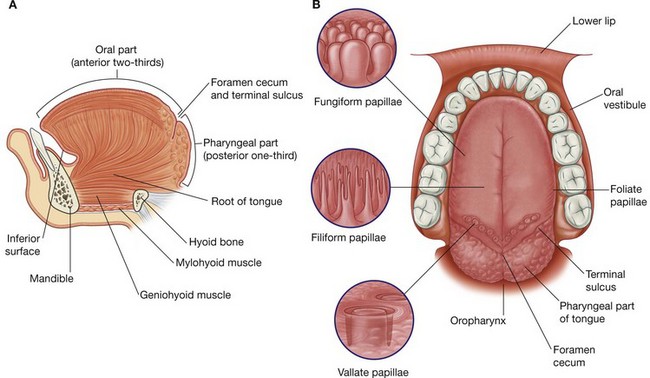
The tongue is a muscular structure that forms part of the floor of the oral cavity and part of the anterior wall of the oropharynx (Fig. 8.242A). Its anterior part is in the oral cavity and is somewhat triangular in shape with a blunt apex of tongue. The apex is directed anteriorly and sits immediately behind the incisor teeth. The root of tongue is attached to the mandible and the hyoid bone.
The superior surface of the oral or anterior two-thirds of the tongue is oriented in the horizontal plane.
The pharyngeal surface or posterior one-third of the tongue curves inferiorly and becomes oriented more in the vertical plane. The oral and pharyngeal surfaces are separated by a v-shaped terminal sulcus of tongue. This terminal sulcus forms the inferior margin of the oropharyngeal isthmus between the oral and pharyngeal cavities. At the apex of the v-shaped sulcus is a small depression (the foramen cecum of tongue), which marks the site in the embryo where the epithelium invaginated to form the thyroid gland.
In some people a thyroglossal duct persists and connects the foramen cecum on the tongue with the thyroid gland in the neck.
Papillae
The superior surface of the oral part of the tongue is covered by hundreds of papillae (Fig. 8.242B):
 filiform papillae
filiform papillae are small cone-shaped projections of the mucosa that end in one or more points;
 fungiform papillae
fungiform papillae are rounder in shape and larger than the filiform papillae, and tend to be concentrated along the margins of the tongue;

the largest of the papillae are the vallate papillae, which are blunt-ended cylindrical papillae invaginations in the tongue’s surface—there are only about 8 to 12 vallate papillae in a single
V-shaped line immediately anterior to the terminal sulcus of tongue;
 foliate papillae
foliate papillae are linear folds of mucosa on the sides of the tongue near the terminal sulcus of tongue.
The papillae in general increase the area of contact between the surface of the tongue and the contents of the oral cavity. All except the filiform papillae have taste buds on their surfaces.
Inferior surface of tongue
The undersurface of the oral part of the tongue lacks papillae, but does have a number of linear mucosal folds (Fig. 8.253). A single median fold (the frenulum of tongue) is continuous with the mucosa covering the floor of the oral cavity, and overlies the lower margin of a midline sagittal septum, which internally separates the right and left sides of the tongue. On each side of the frenulum is a lingual vein, and lateral to each vein is a rough fimbriated fold.
Pharyngeal surface
The mucosa covering the pharyngeal surface of the tongue is irregular in contour because of the many small nodules of lymphoid tissue in the submucosa. These nodules are collectively the lingual tonsil.
There are no papillae on the pharyngeal surface.
Muscles
The bulk of the tongue is composed of muscle (Fig. 8.242 and Table 8.21).
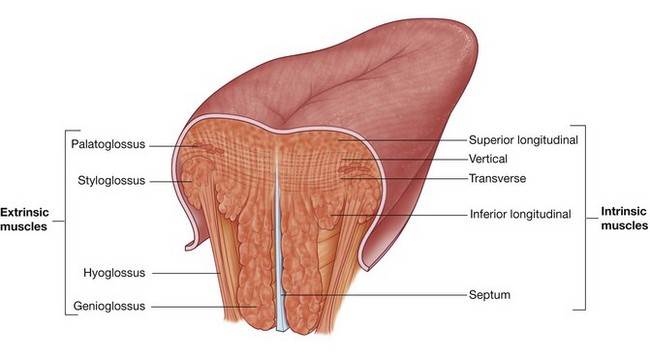
The tongue is completely divided into a left and right half by a median sagittal septum composed of connective tissue. This means that all muscles of the tongue are paired. There are intrinsic and extrinsic lingual muscles.
Except for the palatoglossus, which is innervated by the vagus nerve [X], all muscles of the tongue are innervated by the hypoglossal nerve [XII].
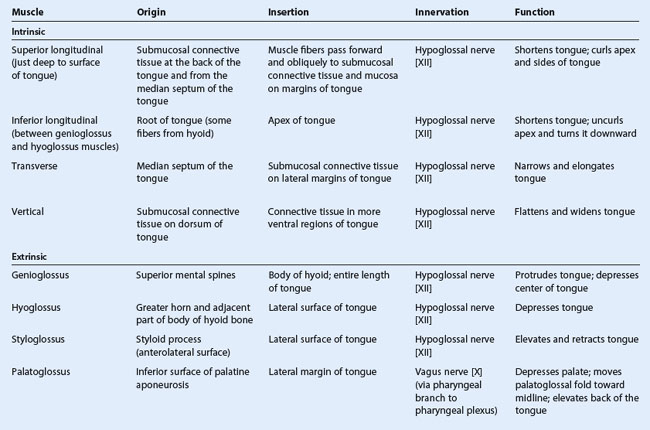
Intrinsic muscles
The intrinsic muscles of the tongue (Fig. 8.243) originate and insert within the substance of the tongue. They are divided into superior longitudinal, inferior longitudinal, transverse, and vertical muscles, and they alter the shape of the tongue by:

lengthening and shortening it;

curling and uncurling its apex and edges; and

flattening and rounding its surface.
Working in pairs or one side at a time the intrinsic muscles of the tongue contribute to precision movements of the tongue required for speech, eating, and swallowing.
Extrinsic muscles
Extrinsic muscles of the tongue (Fig. 8.243 and Table 8.21) originate from structures outside the tongue and insert into the tongue. There are four major extrinsic muscles on each side, the genioglossus, hyoglossus, styloglossus, and palatoglossus. These muscles protrude, retract, depress, and elevate the tongue.
Genioglossus
The thick fan-shaped genioglossus muscles make a substantial contribution to the structure of the tongue. They occur on either side of the midline septum that separates left and right halves of the tongue.
The genioglossus muscles originate from the superior mental spines on the posterior surface of the mandibular symphysis immediately superior to the origin of the geniohyoid muscles from the inferior mental spines (Fig. 8.244). From this small site of origin, each muscle expands posteriorly and superiorly. The most inferior fibers attach to the hyoid bone. The remaining fibers spread out superiorly to blend with the intrinsic muscles along virtually the entire length of the tongue.
The genioglossus muscles:

depress the central part of the tongue; and

protrude the anterior part of the tongue out of the oral fissure (i.e., stick the tongue out).
Like most muscles of the tongue, the genioglossus muscles are innervated by the hypoglossal nerves [XII].
Asking a patient to “stick your tongue out” can be used as a test for the hypoglossal nerves [XII]. If the nerves are functioning normally, the tongue should protrude evenly in the midline. If the nerve on one side is not fully functional, the tip of the tongue will point to that side.
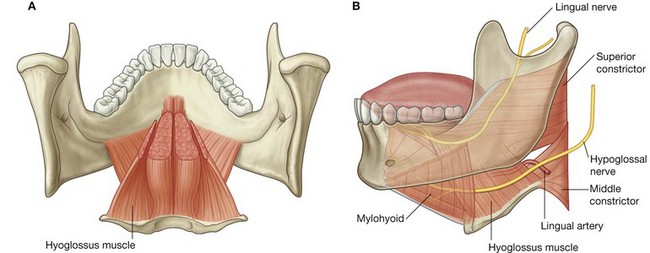
Hyoglossus
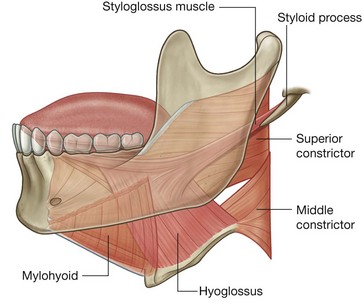
The hyoglossus muscles are thin quadrangular muscles lateral to the genioglossus muscles (Fig. 8.245).
Each hyoglossus muscle originates from the entire length of the greater horn and the adjacent part of the body of the hyoid bone. At its origin from the hyoid bone, the hyoglossus muscle is lateral to the attachment of the middle constrictor muscle of the pharynx. The muscle passes superiorly and anteriorly through the gap between the superior constrictor, middle constrictor, and mylohyoid to insert into the tongue lateral to the genioglossus and medial to the styloglossus.
The hyoglossus muscle depresses the tongue and is innervated by the hypoglossal nerve [XII].
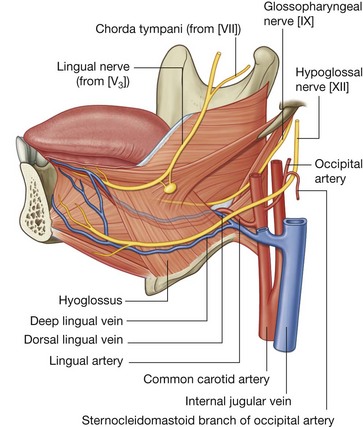
An important landmark
The hyoglossus muscle is an important landmark in the floor of the oral cavity:

the lingual artery from the external carotid artery in the neck enters the tongue deep to the hyoglossus, between the hyoglossus and genioglossus;

the hypoglossal nerve [XII] and lingual nerve (branch of the mandibular nerve [V
3]), from the neck and infratemporal fossa of the head, respectively, enter the tongue on the external surface of the hyoglossus.
Styloglossus
The styloglossus muscles originate from the anterior surface of the styloid processes of the temporal bones. From here, each muscle passes inferiorly and medially through the gap between the middle constrictor, superior constrictor, and mylohyoid muscles to enter the lateral surface of the tongue where they blend with the superior margin of the hyoglossus and with the intrinsic muscles (Fig. 8.246).
The styloglossus muscles retract the tongue and pull the back of the tongue superiorly. They are innervated by the hypoglossal nerves [XII].
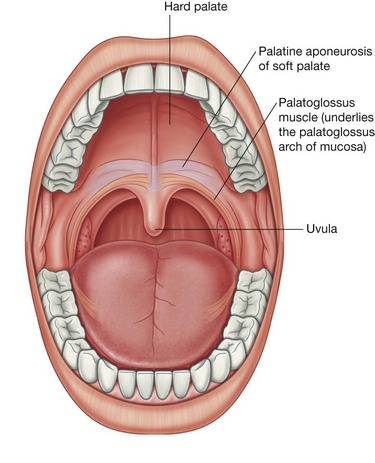
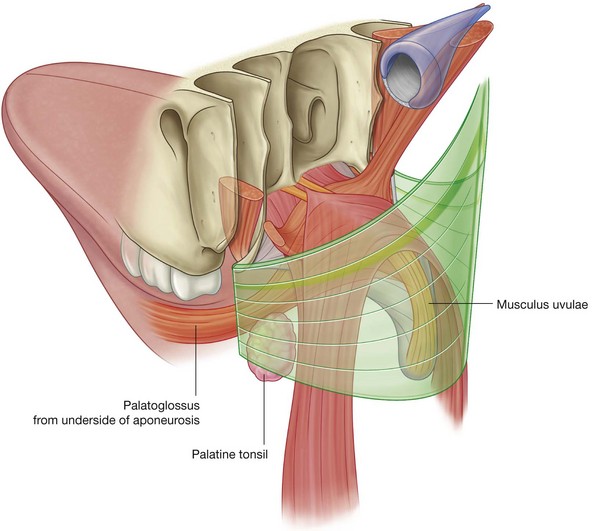
Palatoglossus
The palatoglossus muscles are muscles of the soft palate and the tongue. Each originates from the undersurface of the palatine aponeurosis and passes anteroinferiorly to the lateral side of the tongue (Fig. 8.247).
The palatoglossus muscles:

elevate the back of the tongue;

move the palatoglossal arches of mucosa toward the midline; and

depress the soft palate.
These movements facilitate closing of the oropharyngeal isthmus and as a result separate the oral cavity from the oropharynx.
Unlike other muscles of the tongue, but similar to most other muscles of the soft palate, the palatoglossus muscles are innervated by the vagus nerves [X].
Vessels
Arteries
The major artery of the tongue is the lingual artery (Fig. 8.248).
On each side, the lingual artery originates from the external carotid artery in the neck adjacent to the tip of the greater horn of the hyoid bone. It forms an upward bend and then loops downward and forward to pass deep to the hyoglossus muscle, and accompanies the muscle through the aperture formed by the margins of the mylohyoid, superior constrictor, and middle constrictor muscles, and enters the floor of the oral cavity.
The lingual artery then travels forward in the plane between the hyoglossus and genioglossus muscles to the apex of the tongue.
In addition to the tongue, the lingual artery supplies the sublingual gland, gingiva, and oral mucosa in the floor of the oral cavity.
Veins
The tongue is drained by dorsal lingual and deep lingual veins (Fig. 8.248).
The deep lingual veins are visible through the mucosa on the undersurface of the tongue. Although they accompany the lingual arteries in anterior parts of the tongue, they become separated from the arteries posteriorly by the hyoglossus muscles. On each side, the deep lingual vein travels with the hypoglossal nerve [XII] on the external surface of the hyoglossus muscle and passes out of the floor of the oral cavity through the aperture formed by the margins of the mylohyoid, superior constrictor, and middle constrictor muscles. It joins the internal jugular vein in the neck.
The dorsal lingual vein follows the lingual artery between the hyoglossus and genioglossus muscles and, like the deep lingual vein, drains into the internal jugular vein in the neck.
Innervation
Innervation of the tongue is complex and involves a number of nerves (Figs. 8.248 and 8.249).
Glossopharyngeal nerve [IX]
Taste (SA) and general sensation from the pharyngeal part of the tongue are carried by the glossopharyngeal nerve [IX].
The glossopharyngeal nerve [IX] leaves the skull through the jugular foramen and descends along the posterior surface of the stylopharyngeus muscle. It passes around the lateral surface of the stylopharyngeus and then slips through the posterior aspect of the gap between the superior constrictor, middle constrictor, and mylohyoid muscles. The nerve then passes forward on the oropharyngeal wall just below the inferior pole of the palatine tonsil and enters the pharyngeal part of the tongue deep to the styloglossus and hyoglossus muscles. In addition to taste and general sensation on the posterior one-third of the tongue, branches creep anterior to the terminal sulcus of tongue to carry taste (SA) and general sensation from the vallate papillae.
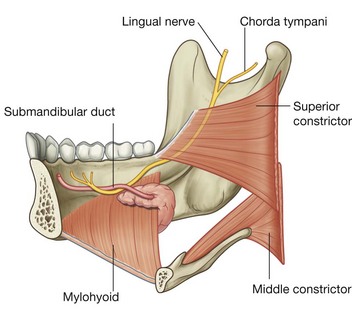
Lingual nerve
General sensory innervation from the anterior two-thirds or oral part of the tongue is carried by the lingual nerve, which is a major branch of the mandibular nerve [V3]. It originates in the infratemporal fossa and passes anteriorly into the floor of the oral cavity by passing through the gap between the mylohyoid, superior constrictor, and middle constrictor muscles (Fig. 8.250).
As it travels through the gap, it passes immediately inferior to the attachment of superior constrictor to the mandible and continues forward on the medial surface of the mandible adjacent to the last molar tooth and deep to the gingiva. In this position, the nerve can be palpated against the bone by placing a finger into the oral cavity.
The lingual nerve then continues anteromedially across the floor of the oral cavity, loops under the submandibular duct, and ascends into the tongue on the external and superior surface of the hyoglossus muscle.
In addition to general sensation from the oral part of the tongue, the lingual nerve also carries general sensation from the mucosa on the floor of the oral cavity and gingiva associated with the lower teeth. The lingual nerve also carries parasympathetic and taste fibers from the oral part of the tongue that are part of the facial nerve [VII].
Facial nerve [VII]
Taste (SA) from the oral part of the tongue is carried into the central nervous system by the facial nerve [VII]. Special sensory (SA) fibers of the facial nerve [VII] leave the tongue and oral cavity as part of the lingual nerve. The fibers then enter the chorda tympani nerve, which is a branch of the facial nerve [VII] that joins the lingual nerve in the infratemporal fossa (Fig. 8.250; also see p. 935).
Hypoglossal nerve [XII]
All muscles of the tongue are innervated by the hypoglossal nerve [XII] except for the palatoglossus muscle, which is innervated by the vagus nerve [X].
The hypoglossal nerve [XII] leaves the skull through the hypoglossal canal and descends almost vertically in the neck to a level just below the angle of mandible (Fig. 8.251). Here it angles sharply forward around the sternocleidomastoid branch of the occipital artery, crosses the external carotid artery, and continues forward, crossing the loop of the lingual artery, to reach the external surface of the lower one-third of the hyoglossus muscle.
The hypoglossal nerve [XII] follows the hyoglossus muscle through the gap between the superior constrictor, middle constrictor, and mylohyoid muscles to reach the tongue.
In the upper neck, a branch from the anterior ramus of C1 joins the hypoglossal nerve [XII]. Most of these C1 fibers leave the hypoglossal nerve [XII] as the superior root of the ansa cervicalis (Fig. 8.251). Near the posterior border of the hyoglossus muscle, the remaining fibers leave the hypoglossal nerve [XII] and form two nerves:

the thyrohyoid branch, which remains in the neck to innervate the thyrohyoid muscle;

the branch to the geniohyoid, which passes into the floor of the oral cavity to innervate the geniohyoid.
Lymphatics
All lymphatic vessels from the tongue ultimately drain into the deep cervical chain of nodes along the internal jugular vein:

the pharyngeal part of the tongue drains through the pharyngeal wall directly into mainly the jugulodigastric node of the deep cervical chain;

the oral part of the tongue drains both directly into the deep cervical nodes, and indirectly into these nodes by passing first through the mylohyoid muscle and into submental and submandibular nodes.
The submental nodes are inferior to the mylohyoid muscles and between the digastric muscles, while the submandibular nodes are below the floor of the oral cavity along the inner aspect of the inferior margins of the mandible.
The tip of the tongue drains through the mylohyoid muscle into the submental nodes and then into mainly the jugulo-omohyoid node of the deep cervical chain.
Salivary glands
Salivary glands are glands that open or secrete into the oral cavity. Most are small glands in the submucosa or mucosa of the oral epithelium lining the tongue, palate, cheeks, and lips, and open into the oral cavity directly or via small ducts. In addition to these small glands are much larger glands, which include the paired parotid, submandibular, and sublingual glands.
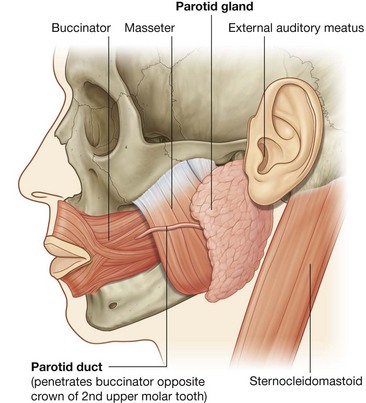
Parotid gland
The parotid gland (see p. 863) on each side is entirely outside the boundaries of the oral cavity in a shallow triangular-shaped trench (Fig. 8.252) formed by:

the sternocleidomastoid muscle behind;

the ramus of mandible in front; and

superiorly, the base of the trench is formed by the external acoustic meatus and the posterior aspect of the zygomatic arch.
The gland normally extends anteriorly over the masseter muscle, and inferiorly over the posterior belly of the digastric muscle.
The parotid duct passes anteriorly across the external surface of the masseter muscle and then turns medially to penetrate the buccinator muscle of the cheek and open into the oral cavity adjacent to the crown of the second upper molar tooth.
The parotid gland encloses the external carotid artery, the retromandibular vein, and the origin of the extracranial part of the facial nerve [VII].
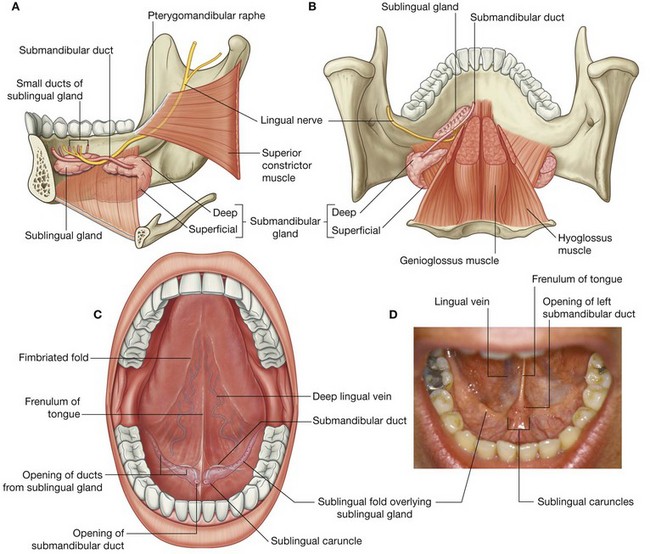
Submandibular glands
The elongate submandibular glands are smaller than the parotid glands, but larger than the sublingual glands. Each is hook shaped (Fig. 8.253A and 8.253B):

the larger arm of the hook is directed forward in the horizontal plane below the mylohyoid muscle and is therefore outside the boundaries of the oral cavity—this larger superficial part of the gland is directly against a shallow impression on the medial side of the mandible (submandibular fossa) inferior to the mylohyoid line;

the smaller arm of the hook (or deep part) of the gland loops around the posterior margin of the mylohyoid muscle to enter and lie within the floor of the oral cavity where it is lateral to the root of the tongue on the lateral surface of the hyoglossus muscle.
The submandibular duct emerges from the medial side of the deep part of the gland in the oral cavity and passes forward to open on the summit of a small sublingual caruncle (papilla) beside the base of frenulum of the tongue (Fig. 8.253C and 8.253D).
The lingual nerve loops under the submandibular duct, crossing first the lateral side and then the medial side of the duct, as the nerve descends anteromedially through the floor of the oral cavity and then ascends into the tongue.
Sublingual glands
The sublingual glands are the smallest of the three major paired salivary glands. Each is almond shaped and is immediately lateral to the submandibular duct and associated lingual nerve in the floor of the oral cavity (Fig. 8.253).
Each sublingual gland lies directly against the medial surface of the mandible where it forms a shallow groove (sublingual fossa) superior to the anterior one-third of the mylohyoid line.
The superior margin of the sublingual gland raises an elongate fold of mucosa (sublingual fold), which extends from the posterolateral aspect of the floor of the oral cavity to the sublingual papilla beside the base of the frenulum of the tongue at the midline anteriorly.
The sublingual gland drains into the oral cavity via numerous small ducts (minor sublingual ducts), which open onto the crest of the sublingual fold. Occasionally, the more anterior part of the gland is drained by a duct (major sublingual duct) that opens together with the submandibular duct on the sublingual caruncle.
Vessels
Vessels that supply the parotid gland originate from the external carotid artery and from its branches that are adjacent to the gland. The submandibular and sublingual glands are supplied by branches of the facial and lingual arteries.
Veins from the parotid gland drain into the external jugular vein, and those from the submandibular and sublingual glands drain into lingual and facial veins.
Lymphatic vessels from the parotid gland drain into nodes that are on or in the gland. These parotid nodes then drain into superficial and deep cervical nodes.
Lymphatics from the submandibular and sublingual glands drain mainly into submandibular nodes and then into deep cervical nodes, particularly the jugulo-omohyoid node.
Innervation
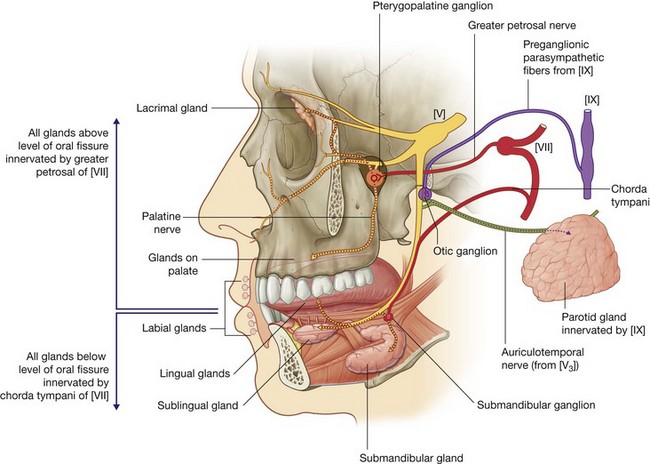
Parasympathetic
Parasympathetic innervation to all salivary glands in the oral cavity is by branches of the facial nerve [VII], which join branches of the maxillary [V2] and mandibular [V3] nerves to reach their target destinations.
The parotid gland receives its parasympathetic innervation from fibers that initially traveled in the glossopharyngeal nerve [IX], which eventually joins a branch of the mandibular nerve [V3] in the infratemporal fossa (Fig. 8.254).
Greater petrosal nerve
All salivary glands above the level of the oral fissure, as well as all mucus glands in the nose and the lacrimal gland in the orbit, are innervated by parasympathetic fibers carried in the greater petrosal branch of the facial nerve [VII] (Fig. 8.254). Preganglionic parasympathetic fibers carried in this nerve enter the pteryatine fossa and synapse with postganglionic parasympathetic fibers in the pteryatine ganglion formed around branches of the maxillary nerve [V2]. Postganglionic parasympathetic fibers join general sensory branches of the maxillary nerve, such as the palatine nerves, destined for the roof of the oral cavity, to reach their target glands.
Chorda tympani
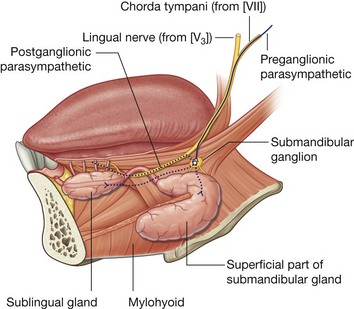
All glands below the level of the oral fissure, which include those small glands in the floor of the oral cavity, in the lower lip, and in the tongue, and the larger submandibular and sublingual glands, are innervated by parasympathetic fibers carried in the chorda tympani branch of the facial nerve [VII] (Fig. 8.254).
The chorda tympani joins the lingual nerve of the mandibular nerve [V3] in the infratemporal fossa and passes with it into the oral cavity. On the external surface of the hyoglossus muscle, preganglionic parasympathetic fibers leave the inferior aspect of the lingual nerve to synapse with postganglionic parasympathetic fibers in the submandibular ganglion, which appears to hang off the lingual nerve (Fig. 8.255). Postganglionic parasympathetic fibers leave the ganglion and pass directly to the submandibular and sublingual glands while others hop back onto the lingual nerve and travel with branches of the lingual nerve to target glands.
Roof—palate
The roof of the oral cavity consists of the palate, which has two parts—an anterior hard palate and a posterior soft palate (Fig. 8.256).
Hard palate
The hard palate separates the oral cavity from the nasal cavities. It consists of a bony plate covered above and below by mucosa:

above, it is covered by respiratory mucosa and forms the floor of the nasal cavities;

below, it is covered by a tightly bound layer of oral mucosa and forms much of the roof of the oral cavity (
Fig. 8.256).
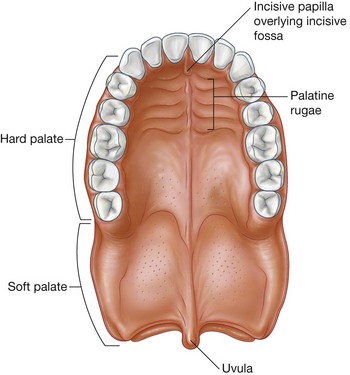
The palatine processes of the maxillae form the anterior three-quarters of the hard palate. The horizontal plates of the palatine bones form the posterior one-quarter. In the oral cavity, the upper alveolar arch borders the hard palate anteriorly and laterally. Posteriorly, the hard palate is continuous with the soft palate.
The mucosa of the hard palate in the oral cavity possesses numerous transverse palatine folds (palatine rugae) and a median longitudinal ridge (palatine raphe), which ends anteriorly in a small oval elevation (incisive papilla). The incisive papilla overlies the incisive fossa formed between the horizontal plates of the maxillae immediately behind the incisor teeth.
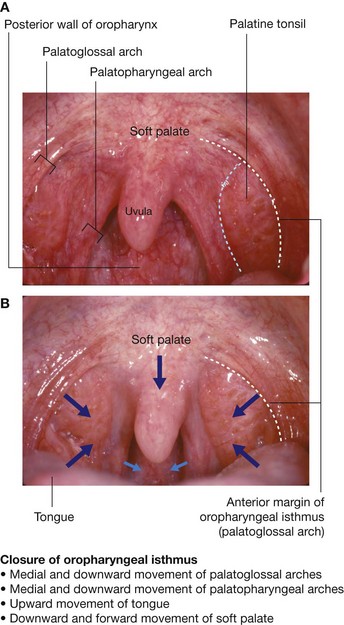
Soft palate
The soft palate (Fig. 8.256) continues posteriorly from the hard palate and acts as a valve that can be:

depressed to help close the oropharyngeal isthmus;

elevated to separate the nasopharynx from the oropharynx.
The soft palate is formed and moved by four muscles and is covered by mucosa that is continuous with the mucosa lining the pharynx and oral and nasal cavities.
The small tear-shaped muscular projection that hangs from the posterior free margin of the soft palate is the uvula.
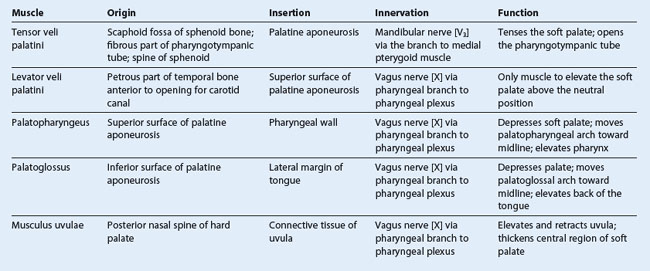
Muscles of the soft palate
Five muscles (Table 8.22) on each side contribute to the formation and movement of the soft palate. Two of these, the tensor veli palatini and levator veli palatini, descend into the palate from the base of the skull. Two others, the palatoglossus and palatopharyngeus, ascend into the palate from the tongue and pharynx, respectively. The last muscle, the musculus uvulae, is associated with the uvula.
All muscles of the palate are innervated by the vagus nerve [X] except for the tensor veli palatini, which is innervated by the mandibular nerve [V3] (via the nerve to medial pterygoid).
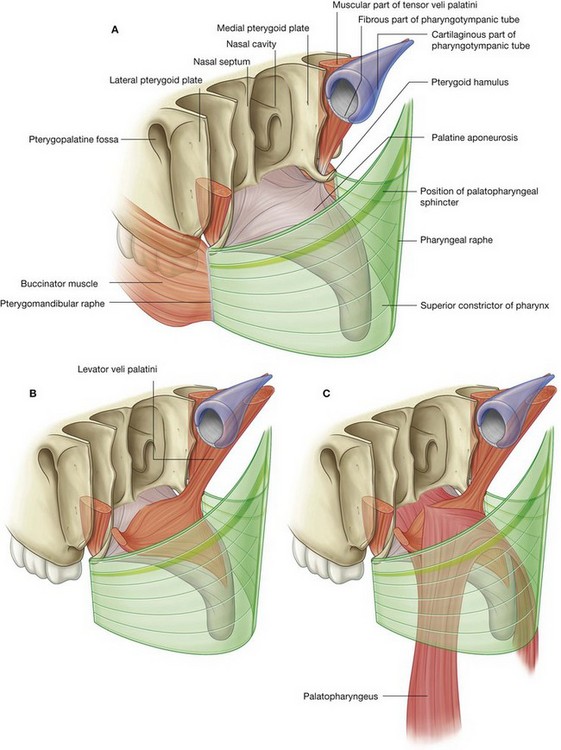
Tensor veli palatini and the palatine aponeurosis
The tensor veli palatini muscle is composed of two parts—a vertical muscular part and a more horizontal fibrous part, which forms the palatine aponeurosis (Fig. 8.257A).
The vertical part of the tensor veli palatini is thin and triangular in shape with its base attached to the skull and its apex pointed inferiorly. The base is attached along an oblique line that begins medially at the scaphoid fossa near the root of the pterygoid process of the sphenoid bone and continues laterally along the membranous part of the pharyngotympanic tube to the spine of the sphenoid bone.
The tensor veli palatini descends vertically along the lateral surface of the medial plate of the pterygoid process and pharyngeal wall to the pterygoid hamulus where the fibers converge to form a small tendon.
The tendon loops 90° medially around the pterygoid hamulus, penetrating the origin of the buccinator muscle as it does, and expands like a fan to form the fibrous horizontal part of the muscle. This fibrous part is continuous across the midline with its partner on the other side to form the palatine aponeurosis.
The palatine aponeurosis is attached anteriorly to the margin of the hard palate, but is unattached posteriorly where it ends in a free margin. This expansive aponeurosis is the major structural element of the soft palate to which the other muscles of the palate attach.
The tensor veli palatini:

tenses (makes firm) the soft palate so that the other muscles attached to the palate can work more effectively; and

opens the pharyngotympanic tube when the palate moves during yawning and swallowing as a result of its attachment superiorly to the membranous part of the pharyngotympanic tube.
The tensor veli palatini is innervated by the nerve to medial pterygoid from the mandibular nerve [V3].
Levator veli palatini
The levator veli palatini muscle originates from the base of the skull and descends to the upper surface of the palatine aponeurosis (Fig. 8.257B). On the skull, it originates from a roughened area on the petrous part of the temporal bone immediately anterior to the opening of the carotid canal. Some fibers also originate from adjacent parts of the pharyngotympanic tube.
The levator veli palatini passes anteroinferiorly through fascia of the pharyngeal wall, passes medial to the pharyngotympanic tube, and inserts onto the palatine aponeurosis. Its fibers interlace at the midline with those of the levator veli palatini on the other side.
Unlike the tensor veli palatini muscles, the levator veli palatini muscles do not pass around the pterygoid hamulus, but course directly from the base of the skull to the upper surface of the palatine aponeurosis. Therefore, they are the only muscles that can elevate the palate above the neutral position and close the pharyngeal isthmus between the nasopharynx and oropharynx.
The levator veli palatini is innervated by the vagus nerve [X] through the pharyngeal branch to the pharyngeal plexus. Clinically, the levator veli palatini can be tested by asking a patient to say “ah.” If the muscle on each side is functioning normally, the palate elevates evenly in the midline. If one side is not functioning, the palate deviates away from the abnormal side.
Palatopharyngeus
The palatopharyngeus muscle originates from the superior surface of the palatine aponeurosis and passes posterolaterally over its margin to descend and become one of the longitudinal muscles of the pharyngeal wall (Fig. 8.257C). It is attached to the palatine aponeurosis by two flat lamellae separated by the levator veli palatini muscle. The more anterior and lateral of these two lamellae is attached to the posterior margin of the hard palate as well as to the palatine aponeurosis.
The two palatopharyngeus muscles, one on each side, underlie the palatopharyngeal arches on the oropharyngeal wall. The palatopharyngeal arches lie posterior and medial to the palatoglossal arches when viewed anteriorly through the oral cavity (Fig. 8.258).
On each side, the palatine tonsil is between the palatopharyngeal and palatoglossal arches on the lateral oropharyngeal wall.
The palatopharyngeus muscles:

depress the palate and move the palatopharyngeal arches toward the midline like curtains—both these actions help close the oropharyngeal isthmus; and

elevate the pharynx during swallowing.
The palatopharyngeus is innervated by the vagus nerve [X] through the pharyngeal branch to the pharyngeal plexus.
Palatoglossus
The palatoglossus muscle attaches to the inferior (oral surface) of the palatine aponeurosis and passes inferiorly and anteriorly into the lateral surface of the tongue (Fig. 8.259).
The palatoglossus muscle underlies a fold of mucosa that arches from the soft palate to the tongue. These palatoglossal arches, one on each side, are lateral and anterior to the palatopharyngeal arches and define the lateral margins of the oropharyngeal isthmus (Fig. 8.258).
The palatine tonsil is between the palatoglossal and palatopharyngeal arches on the lateral oropharyngeal wall.
The palatoglossus muscles depress the palate, move the palatoglossal arches toward the midline like curtains, and elevate the back of the tongue. These actions help close the oropharyngeal isthmus.
The palatoglossus is innervated by the vagus nerve [X] through the pharyngeal branch to the pharyngeal plexus.
Musculus uvulae
The musculus uvulae originates from the posterior nasal spine on the posterior margin of the hard palate and passes directly posteriorly over the dorsal aspect of the palatine aponeurosis to insert into connective tissue underlying the mucosa of the uvula (Fig. 8.259). It passes between the two lamellae of the palatopharyngeus superior to the attachment of the levator veli palatini. Along the midline, the musculus uvulae blends with its partner on the other side.
The musculus uvulae elevates and retracts the uvula. This action thickens the central part of the soft palate and helps the levator veli palatini muscles close the pharyngeal isthmus between the nasopharynx and oropharynx.
The musculus uvulae is innervated by the vagus nerve [X] through the pharyngeal branch to the pharyngeal plexus.
Vessels
Arteries
Arteries of the palate include the greater palatine branch of the maxillary artery, the ascending palatine branch of the facial artery, and the palatine branch of the ascending pharyngeal artery. The maxillary, facial, and ascending pharyngeal arteries are all branches that arise in the neck from the external carotid artery (Fig. 8.260).
Ascending palatine artery and palatine branch
The ascending palatine artery of the facial artery ascends along the external surface of the pharynx. The palatine branch loops medially over the top of the superior constrictor muscle of the pharynx to penetrate the pharyngeal fascia with the levator veli palatini muscle and follow the levator veli palatini to the soft palate.
The palatine branch of the ascending pharyngeal artery follows the same course as the palatine branch of the ascending palatine artery from the facial artery and may replace the vessel.
Greater palatine artery
The greater palatine artery originates from the maxillary artery in the pteryatine fossa. It descends into the palatine canal where it gives origin to a small lesser palatine branch, and then continues through the greater palatine foramen onto the inferior surface of the hard palate (Fig. 8.261). The greater palatine artery passes forward on the hard palate and then leaves the palate superiorly through the incisive canal to enter the medial wall of the nasal cavity where it terminates. The greater palatine artery is the major artery of the hard palate. It also supplies palatal gingiva. The lesser palatine branch passes through the lesser palatine foramen just posterior to the greater palatine foramen, and contributes to the vascular supply of the soft palate.
Veins
Veins from the palate generally follow the arteries and ultimately drain into the pterygoid plexus of veins in the infratemporal fossa (Fig. 8.262), or into a network of veins associated with the palatine tonsil, which drain into the pharyngeal plexus of veins or directly into the facial vein.
Lymphatics
Lymphatic vessels from the palate drain into deep cervical nodes (Fig. 8.262).
Innervation
The palate is supplied by the greater and lesser palatine nerves and the nasopalatine nerve (Figs. 8.261 and 8.263).
General sensory fibers carried in all these nerves originate in the pteryatine fossa from the maxillary nerve [V2].
Parasympathetic (to glands) and SA (taste on soft palate) fibers from a branch of the facial nerve [VII] join the nerves in the pteryatine fossa, as do the sympathetics (mainly to blood vessels) ultimately derived from the T1 spinal cord level.
Greater and lesser palatine nerves
The greater and lesser palatine nerves descend through the pteryatine fossa and palatine canal to reach the palate:

the greater palatine nerve travels through the greater palatine foramen and turns anteriorly to supply the hard palate and gingiva as far as the first premolar;

the lesser palatine nerve passes posteromedially to supply the soft palate.
Nasopalatine nerve
The nasopalatine nerve also originates in the pteryatine fossa, but passes medially into the nasal cavity. It continues medially over the roof of the nasal cavity to reach the medial wall, then anteriorly and obliquely down the wall to reach the incisive canal in the anterior floor, and descends through the incisive canal and fossa to reach the inferior surface of the hard palate.
The nasopalatine nerve supplies gingiva and mucosa adjacent to the incisors and canine.
Oral fissure and lips
The oral fissure is the slit-like opening between the lips that connects the oral vestibule to the outside (Fig. 8.264). It can be opened and closed, and altered in shape by the movements of the muscles of facial expression associated with the lips and surrounding regions, and by movements of the lower jaw (mandible).
The lips are entirely composed of soft tissues (Fig. 8.264B). They are lined internally by oral mucosa and covered externally by skin. Externally, there is an area of transition from the thicker skin that covers the face to the thinner skin that overlies the margins of the lips and continues as oral mucosa onto the deep surfaces of the lips.
Blood vessels are closer to the surface in areas where the skin is thin and as a consequence there is a vermilion border that covers the margins of the lips.
The upper lip has a shallow vertical groove on its external surface (the philtrum) sandwiched between two elevated ridges of skin. The philtrum and ridges are formed embryologically by fusion of the medial nasal processes.
On the inner surface of both lips, a fold of mucosa (the median labial frenulum) connects the lip to the adjacent gum.
The lips enclose the orbicularis oris muscle, neurovascular tissues, and labial glands. The small pea-shaped labial glands are between the muscle tissue and the oral mucosa and open into the oral vestibule.
A number of muscles of facial expression control the shape and size of the oral fissure. The most important of these is the orbicularis oris muscle, which encircles the orifice and acts as a sphincter. A number of other muscles of facial expression blend into the orbicularis oris or other tissues of the lips and open or adjust the contours of the oral fissure. These include buccinator, levator labii superioris, zygomaticus major and minor, levator anguli oris, depressor labii inferioris, depressor anguli oris, and platysma (see pp. 859–862).
Oropharyngeal isthmus
The oropharyngeal isthmus is the opening between the oral cavity and the oropharynx (see Fig. 8.258). It is formed:

laterally by the palatoglossal arches;

superiorly by the soft palate; and

and inferiorly by the sulcus terminalis of the tongue that divides the oral surface of the tongue (anterior two-thirds) from the pharyngeal surface (posterior one-third).
The oropharyngeal isthmus can be closed by elevation of the posterior aspect of the tongue, depression of the palate, and medial movement of the palatoglossal arches toward the midline.
Medial movement of the palatopharyngeal arches medial and posterior to the palatoglossal arches is also involved in closing the oropharyngeal isthmus. By closing the oropharyngeal isthmus, food or liquid can be held in the oral cavity while breathing.
Teeth and gingivae
The teeth are attached to sockets (alveoli) in two elevated arches of bone on the mandible below and the maxillae above (alveolar arches).
If the teeth are removed, the alveolar bone is resorbed and the arches disappear.
The gingivae (gums) are specialized regions of the oral mucosa that surround the teeth and cover adjacent regions of the alveolar bone.
The different types of teeth are distinguished on the basis of morphology, position, and function (Fig. 8.265A).
In adults, there are 32 teeth, 16 in the upper jaw and 16 in the lower jaw. On each side in both maxillary and mandibular arches are two incisor, one canine, two premolar, and three molar teeth.

the
incisor teeth are the “front teeth” and have one root and a chisel-shaped crown, which “cuts”;

the
canine teeth are posterior to the incisors, are the longest teeth, have a crown with a single pointed cusp, and “grasp”;

the
premolar teeth (bicuspids) have a crown with two pointed cusps, one on the buccal (cheek) side of the tooth and the other on the lingual (tongue) or palatal (palate) side, generally have one root (but the upper first premolar next to the canine may have two), and “grind”;

the
molar teeth are behind the premolar teeth, have three roots and crowns with three to five cusps, and “grind.”
Two successive sets of teeth develop in humans, deciduous teeth (“baby” teeth) (Fig. 8.265B) and permanent teeth (“adult” teeth). The deciduous teeth emerge from the gingivae at between six months and two years of age. Permanent teeth begin to emerge and replace the deciduous teeth at around age six years, and can continue to emerge into adulthood.
The 20 deciduous teeth consist of two incisor, one canine, and two molar teeth on each side of the upper and lower jaws. These teeth are replaced by the incisor, canine, and premolar teeth of the permanent teeth. The permanent molar teeth erupt posterior to the deciduous molars and require the jaws to elongate forward to accommodate them.
Vessels
Arteries
All teeth are supplied by vessels that branch either directly or indirectly from the maxillary artery (Fig. 8.266).
Inferior alveolar artery
All lower teeth are supplied by the inferior alveolar artery, which originates from the maxillary artery in the infratemporal fossa. The vessel enters the mandibular canal of the mandible, passes anteriorly in bone supplying vessels to the more posterior teeth, and divides opposite the first premolar into incisor and mental branches. The mental branch leaves the mental foramen to supply the chin, while the incisor branch continues in bone to supply the anterior teeth and adjacent structures.
Anterior and posterior superior alveolar arteries
All upper teeth are supplied by anterior and posterior superior alveolar arteries.
The posterior superior alveolar artery originates from the maxillary artery just after the maxillary artery enters the pteryatine fossa and it leaves the fossa through the pterygomaxillary fissure. It descends on the posterolateral surface of the maxilla, branches, and enters small canals in the bone to supply the molar and premolar teeth.
The anterior superior alveolar artery originates from the infra-orbital artery, which arises from the maxillary artery in the pteryatine fossa. The infra-orbital artery leaves the pteryatine fossa through the inferior orbital fissure and enters the inferior orbital groove and canal in the floor of the orbit. The anterior superior alveolar artery originates from the infra-orbital artery in the infra-orbital canal. It passes through bone and branches to supply the incisor and canine teeth.
Gingival supply
The gingivae are supplied by multiple vessels and the source depends on which side of each tooth the gingiva is—the side facing the oral vestibule or cheek (vestibular or buccal side), or the side facing the tongue or palate (lingual or palatal side):

buccal gingiva of the lower teeth is supplied by branches from the inferior alveolar artery, whereas the lingual side is supplied by branches from the lingual artery of the tongue;

buccal gingiva of the upper teeth is supplied by branches of the anterior and posterior superior alveolar arteries;

palatal gingiva is supplied by branches from the nasopalatine (incisor and canine teeth) and greater palatine (premolar and molar teeth) arteries.
Veins
Veins from the upper and lower teeth generally follow the arteries (Fig. 8.266).
Inferior alveolar veins from the lower teeth, and superior alveolar veins from the upper teeth drain mainly into the pterygoid plexus of veins in the infratemporal fossa, although some drainage from the anterior teeth may be via tributaries of the facial vein.
The pterygoid plexus drains mainly into the maxillary vein and ultimately into the retromandibular vein and jugular system of veins. In addition, small communicating vessels pass superiorly, from the plexus, and pass through small emissary foramina in the base of the skull to connect with the cavernous sinus in the cranial cavity. Infection originating in the teeth can track into the cranial cavity through these small emissary veins.
Venous drainage from the teeth can also be via vessels that pass through the mental foramen to connect with the facial vein.
Veins from the gingivae also follow the arteries and ultimately drain into the facial vein or into the pterygoid plexus of veins.
Lymphatics
Lymphatic vessels from the teeth and gingivae drain mainly into submandibular, submental, and deep cervical nodes (Fig. 8.267).
Innervation
All nerves that innervate the teeth and gingivae are branches of the trigeminal nerve [V] (Figs. 8.268 and 8.269).
Inferior alveolar nerve
The lower teeth are all innervated by branches from the inferior alveolar nerve, which originates in the infratemporal fossa from the mandibular nerve [V3] (Figs. 8.268 and 8.269). The inferior alveolar nerve and its accompanying vessels enter the mandibular foramen on the medial surface of the ramus of mandible and travel anteriorly through the bone in the mandibular canal. Branches to the back teeth originate directly from the inferior alveolar nerve.
Adjacent to the first premolar tooth, the inferior alveolar nerve divides into incisive and mental branches:

the
incisive branch innervates the first premolar, the canine, and the incisor teeth, together with the associated vestibular (buccal) gingiva;

the
mental nerve exits the mandible through the mental foramen and innervates the chin and lower lip.
Anterior, middle, and posterior superior alveolar nerves
All upper teeth are innervated by the anterior, middle, and posterior superior alveolar nerves, which originate directly or indirectly from the maxillary nerve [V2] (Figs. 8.268 and 8.269).
The posterior superior alveolar nerve originates directly from the maxillary nerve [V2] in the pteryatine fossa, exits the pteryatine fossa through the pterygomaxillary fissure, and descends on the posterolateral surface of the maxilla. It enters the maxilla through a small foramen approximately midway between the pterygomaxillary fissure and the last molar tooth, and passes through the bone in the wall of the maxillary sinus. The posterior superior alveolar nerve then innervates the molar teeth through the superior alveolar plexus formed by the posterior, middle, and anterior alveolar nerves.
The middle and anterior superior alveolar nerves originate from the infra-orbital branch of the maxillary nerve [V2] in the floor of the orbit:

the middle superior alveolar nerve arises from the infra-orbital nerve in the infra-orbital groove, passes through the bone in the lateral wall of the maxillary sinus, and innervates the premolar teeth via the superior alveolar plexus;

the anterior superior alveolar nerve originates from the infra-orbital nerve in the infra-orbital canal, passes through the maxilla in the anterior wall of the maxillary sinus, and via the superior alveolar plexus, supplies the canine and incisor teeth.
Innervation of gingivae
Like the teeth, the gingivae are innervated by nerves that ultimately originate from the trigeminal nerve [V] (Fig. 8.269):

gingiva associated with the upper teeth is innervated by branches derived from the maxillary nerve [V
2];

gingiva associated with the lower teeth is innervated by branches of the mandibular nerve [V
3].
The gingiva on the buccal side of the upper teeth is innervated by the anterior, middle, and superior alveolar nerves, which also innervate the adjacent teeth. Gingiva on the palatal (lingual) side of the same teeth is innervated by the nasopalatine and the greater palatine nerves:

the nasopalatine nerve innervates gingiva associated with the incisor and canine teeth;

the greater palatine nerve supplies gingiva associated with the remaining teeth.
The gingiva associated with the (buccal) side of the mandibular incisor, canine, and premolar teeth is innervated by the mental branch of the inferior alveolar nerve. Gingiva on the buccal side of the mandibular molar teeth is innervated by the buccal nerve, which originates in the infratemporal fossa from the mandibular nerve [V3]. Gingiva adjacent to the lingual surface of all lower teeth is innervated by the lingual nerve.
Clinical cases
Case 1 MULTINODULAR GOITER
A 50-year-old overweight woman came to the doctor complaining of hoarseness of voice and noisy breathing. She was also concerned at the increase in size of her neck. On examination she had a slow pulse rate (45 beats per minute). She also had an irregular knobby mass in the anterior aspect of the lower neck, which deviated the trachea to the right.
A clinical diagnosis of a multinodular goiter and hypothyroidism was made.
Enlargement of the thyroid gland is due to increased secretion of thyroid stimulating hormone, which is usually secondary to diminished output of thyroid hormones. The thyroid undergoes periods of activity and regression, which can lead to the formation of nodules, some of which are solid and some of which are partially cystic (colloid cysts). This nodule formation is compounded by areas of fibrosis within the gland. Other causes of multinodular goiter include iodine deficiency, and in certain circumstances, drugs that interfere with the metabolism and production of thyroxine. The typical symptom of a goiter is a painless swelling of the thyroid gland. It may be smooth or nodular, and occasionally it may extend into the superior mediastinum as a retrosternal goiter.
The trachea was deviated.
The enlargement of the thyroid gland due to a multinodular goiter may not be symmetrical. In this case there was significant asymmetrical enlargement of the left lobe of the thyroid deviating the trachea to the right.
The patient had a hoarse voice and noisy breathing.
If the thyroid gland enlargement is significant it can compress the trachea, narrowing it to such an extent that a “crowing sound” is heard during inspiration (stridor).
Other possible causes for hoarseness include paralysis of the vocal cord due to compression of the left recurrent laryngeal nerve from the goiter. Of concern is the possibility of malignant change within the goiter directly invading the recurrent laryngeal nerve. Fortunately, malignant change is rare within the thyroid gland.
When patients have a relatively low production of thyroxine such that the basal metabolic rate is reduced they become more susceptible to infection, including throat and upper respiratory tract infections.
On examination the thyroid gland moved during swallowing.
Characteristically, an enlarged thyroid gland is evident as a neck mass arising on one or both sides of the trachea. The enlarged thyroid gland moves on swallowing because it is attached to the larynx by the pretracheal fascia.
The patient was hypothyroid.
Hypothyroidism refers to the clinical and biochemical state in which the thyroid gland is underactive (hyperthyroidism refers to an overactive thyroid gland). Some patients have thyroid masses and no clinical or biochemical abnormalities—these patients are euthyroid.
The hormone thyroxine controls the basal metabolic rate; therefore, low levels of thyroxine affect the resting pulse rate and may produce other changes, including weight gain, and in some cases depression.
The patient was insistent upon surgery.
After discussion about the risks and complications, a subtotal thyroidectomy was performed. After the procedure the patient complained of tingling in her hands and feet and around her mouth, and carpopedal spasm. These symptoms are typical of tetany and are caused by low serum calcium levels.
The etiology of the low serum calcium level was trauma and bruising of the four parathyroid glands left in situ after the operation. Undoubtedly the trauma of removal of such a large thyroid gland produced a change within the parathyroid gland, which failed to function appropriately. The secretion of parathyroid hormone rapidly decreased over the next 24 hours, resulting in increased excitability of peripheral nerves, manifest by carpopedal spasm and orofacial tingling. Muscle spasms can also be elicited by tapping the facial nerve [VII] as it emerges from the parotid gland to produce twitching of the facial muscles (Chvostek’s sign).
The patient recovered from these symptoms due to a low calcium level over the next 24 hours.
At her return to clinic the patient was placed on supplementary oral thyroxine, which is necessary after removal of the thyroid gland.
The patient also complained of a hoarse voice.
The etiology of her hoarse voice was damage to the recurrent laryngeal nerve.
The recurrent laryngeal nerve lies close to the thyroid gland. It may be damaged in difficult surgical procedures, and this may produce unilateral spasm of the ipsilateral vocal cord to produce a hoarse voice.
Since the thyroidectomy and institution of thyroxine treatment, the patient has lost weight and has no further complaints.
Case 2 PAROTID DUCT CALCULUS
A 25-year-old man complained of significant swelling in front of his right ear before and around mealtimes. This swelling was associated with considerable pain, which was provoked by the ingestion of lemon sweets. On examination he had tenderness around the right parotid region and a hard nodule was demonstrated in the buccal mucosa adjacent to the right upper molar teeth.
A diagnosis of parotid duct calculus was made.
The formation of stones in the salivary glands is not uncommon, but it is more likely in the submandibular gland than in the parotid gland because the saliva is more mucinous and the duct has a long upward course from the floor of the mouth. Nevertheless, stones do form in the parotid gland and the parotid ducts. Notably, most parotid duct calculi and submandibular duct calculi occur in mouths with excellent dental hygiene and mucosa.
An ultrasound scan was performed.
An initial ultrasound scan demonstrated a stone in the distal end of the right parotid duct with evidence of ductal dilatation. Assessment of the gland also demonstrated dilated ducts within the gland and evidence of intraparotid lymphadenopathy (Fig. 8.281).
The patient was treated with antibiotics.
A course of antibiotics was given to remove the bacteria that had produced the inflammation. On return to the doctor some days later the gland was normal in size and there was no evidence of inflammation or infection.
An operation was necessary.
The stone was at the distal end of the parotid duct and it would seem logical and straightforward to make a small incision at the sphincter in the buccal mucosa and deliver the stone, thus permitting the gland to drain normally. Unfortunately, in this patient’s case the gland was significantly destroyed by the chronic obstruction and bacterial infection. Furthermore, smaller calculi were also demonstrated in the gland at ultrasound. On direct questioning it appeared that the patient had had numerous attacks over the previous 4–5 years and it was decided that the parotid gland should be removed surgically.
The patient consented for removal of the parotid gland and a discussion of the possibility for loss of facial function and facial paralysis was had with the patient at this time.
Within the parotid gland the facial nerve [VII] divides into its five terminal branches. At operation the gland is displayed and extremely careful dissection is necessary to peel away the parotid gland from the branches of the facial nerve [VII]. This procedure was made more difficult by the chronic inflammatory change within the gland. After the procedure the patient made a good recovery, though there was some mild paralysis of the whole of the right side of the face. Importantly, taste to the anterior two-thirds of the tongue was preserved. The taste fibers to the anterior two-thirds of the tongue travel in the chorda tympani nerve, which is a branch of the facial nerve [VII]. This nerve leaves the facial nerve [VII] to join the lingual nerve proximal to the parotid gland; therefore, any damage to the facial nerve [VII] within the parotid gland does not affect special sensation (taste).
Over the following week the paralysis improved and was likely due to nerve bruising during the procedure. The patient remained asymptomatic.
Case 3 EXTRADURAL HEMATOMA
A 33-year-old man was playing cricket for his local Sunday team. As the new bowler pitched the ball short, it bounced higher than he anticipated and hit him on the side of his head. He immediately fell to the ground unconscious, but after about 30 seconds he was helped to his feet and felt otherwise well. It was noted he had some bruising around his temple. He decided not to continue playing and went to watch the match from the side. Over the next hour he became extremely sleepy and was eventually unrousable. He was rushed to hospital.
When he was admitted to hospital, the patient’s breathing was shallow and irregular and it was necessary to intubate him. A skull radiograph demonstrated a fracture in the region of the pterion. No other abnormality was demonstrated other than minor soft tissue bruising over the left temporal fossa.
A CT scan was performed.
The CT scan demonstrated a lentiform area of high density within the left cranial fossa.
A diagnosis of extradural hemorrhage was made.
Fractures in the region of the pterion are extremely dangerous. A division of the middle meningeal artery passes deep to this structure and is subject to laceration and disruption, especially in conjunction with a skull injury in this region. In this case the middle meningeal artery was torn and started to bleed, producing a large extradural clot.
The patient’s blood pressure began to increase.
Within the skull there is a fixed volume and clearly what goes in must come out (e.g., blood, cerebrospinal fluid). If there is a space-occupying lesion, such as an extradural hematoma, there is nowhere for this to decompress into. As the lesion expands, the brain becomes compressed and the intracranial pressure increases. This pressure compresses vessels, so lowering the cerebral perfusion pressure. To combat this the homeostatic mechanisms of the body increase the blood pressure to overcome the increase in intracerebral pressure. Unfortunately, the increase in intracranial pressure is compounded by the cerebral edema that occurs at and after the initial insult.
An urgent surgical procedure was performed.
Burr holes were placed around the region of the hematoma and it was evacuated. The small branch of the middle meningeal artery was ligated and the patient spent a few days on the intensive care unit. Fortunately the patient made an uneventful recovery.
Case 4 STENOSIS OF THE INTERNAL CAROTID ARTERY
A 60-year-old woman was brought to the emergency department with acute right-sided weakness, predominantly in the upper limb, which lasted for 24 hours. She made an uneventful recovery, but was extremely concerned about the nature of her illness and went to see her local doctor.
A diagnosis of a transient ischemic attack (TIA) was made.
A TIA is a neurological deficit resolving within 24 hours. It is a type of stroke.
Neurological deficit may be permanent or transient. Most transient events resolve within 21 days; any failure of resolution beyond 21 days is an established stroke.
An investigation into the cause of the TIA was undertaken.
Eighty-five percent of all strokes result from cerebral infarction, of which most are due to embolization.
A duplex Doppler scan of the carotid vessels was performed.
The majority of emboli originate from plaques that develop at and around the carotid bifurcation. Emboli consist of platelet aggregates, cholesterol, and atheromatous debris. Emboli may also arise from the heart secondary to cardiac tumors or myocardial infarction.
The lesion in the brain was on the left side.
The motor cortex for the whole of the right side of the body is represented in the left motor strip of the brain, which sits on the precentral gyrus.
The duplex Doppler ultrasound scan demonstrated a significant narrowing (stenosis) of the left internal carotid artery with evidence of plaque formation and abnormal flow in this region. The narrowing was approximately 90%.
Treatment required an operation.
A carotid endarterectomy (removal of the stenosis and the atheromatous plaque) was planned. This procedure is indicated in the presence of an ulcerating plaque with stenosis. The procedure was carried out under general anesthetic and a curvilinear incision was placed in the left side of the neck. The common carotid, external carotid, and internal carotid arteries were displayed. All vessels were clamped and a shunt was placed from the common carotid artery into the internal carotid artery to maintain cerebral blood flow during the procedure. The internal carotid artery was opened and the plaque excised.
After the procedure the patient did extremely well and suffered no further cerebral events. However, a new medical student examined the patient the following day and demonstrated a number of interesting findings. These included altered skin sensation inferior to the left mandible, altered sensation on the left side of the soft palate, an paralyzed left vocal cord, inability to shrug the left shoulder, and a tongue that deviated to the left.
The etiology of these injuries was due to localized nerve trauma.
This constellation of neurological deficits can be accounted for by trauma to the nerves that are close to the carotid bifurcation. The changes in skin sensation can be accounted for by a neurapraxia due to damage to branches of the narrower cervical nerve. The alteration in sensation in the soft palate is due to neurapraxia of the glossopharyngeal nerve [IX]. The paralyzed left cord results from neurapraxia of the recurrent laryngeal nerve, while the inability to shrug the shoulder is due to neurapraxia of the accessory nerve [XI]. Deviation of the tongue can be accounted for by damage to the hypoglossal nerve [XII].
Most of these changes are transient and are usually due to traction injuries during the surgical procedure.
Case 5 POSTERIOR COMMUNICATING ARTERY ANEURYSM
A 33-year-old fit and well woman came to the emergency department complaining of double vision and pain behind her right eye. She had no other symptoms. On examination of the right eye the pupil was dilated. There was a mild ptosis. Testing of eye movement revealed that the eye turned down and out and the pupillary reflex was not present.
These findings revealed that the patient has an ipsilateral third nerve palsy (palsy of the oculomotor nerve [III]).
The oculomotor nerve [III] is the main motor nerve to the ocular and extra-ocular muscles. It arises from the midbrain and pierces the dura mater to run in the lateral wall of the cavernous sinus. The oculomotor nerve [III] leaves the cranial cavity and enters the orbit through the superior orbital fissure. Within this fissure it divides into its superior and inferior divisions.
The site of the nerve lesion needs to be assessed.
Third nerve palsy may involve the nucleus of the oculomotor nerve [III], which typically spares the pupil and is painless. The pupillary reflexes are supplied from the autonomic fibers of the Edinger–Westphal nucleus, which pass through the ciliary ganglion.
The lesion cannot be a primary oculomotor nerve [III] nuclear injury.
As both the pupillary reflexes and vision are affected, the lesion is likely to be along the course of the oculomotor nerve [III]. Medical conditions such as diabetes mellitus and vascular disease may produce an isolated oculomotor nerve [III] injury, but they are not associated with pain.
The lesion was caused by an aneurysm.
One of the commonest causes of a third nerve palsy is pressure on the nerve from a posterior communicating artery aneurysm, which lies parallel to the nerve on the anterior aspect of the brainstem. As the aneurysm abuts the outside of the oculomotor nerve [III], it involves the parasympathetic fibers, which lead to a predominance of the loss of pupillary function over general function.
The aneurysm was imaged with an angiogram.
The patient initially underwent CT and MRI scanning. Currently, the definitive test for assessment of aneurysms arising from the circle of Willis and its branches is a digital subtraction angiogram. The angiogram demonstrated the posterior communicating artery aneurysm. The patient underwent surgery and made an excellent recovery.
Case 6 RECURRENT EPISTAXIS
A 10-year-old boy was brought to an ENT surgeon (ear, nose, and throat surgeon) with epistaxis (nose bleeding). The bleeding was associated with his nose picking habit. However, the bleeding was profuse and on two occasions required hospital admission and nasal packing.
On inspection an indurated area was noted.
The typical findings are an indurated area in the anterior inferior aspect of the nasal septum (Kiesselbach’s area). This is a very vascular area that has a considerable number of veins, which are often traumatized during nose picking.
The patient underwent treatment.
Typical treatment is cauterization of these prominent veins in Kiesselbach’s area, which is usually performed by a simple local analgesia and the application of silver nitrate.
Unfortunately, the boy was involved in a fight the next day and again developed severe epistaxis, which again was difficult to control.
Not only is there a rich venous plexus around Kiesselbach’s area, but there is also a significant arterial supply, which is provided from the nasal septal branches of the posterior and anterior ethmoidal arteries and the branches of the greater palatine artery. These are supplemented from the septal branches of the superior labial artery.
In most cases treatment is conservative.
Conservative treatment usually involves packing the nasal cavity until bleeding has stopped and correcting any bleeding abnormality. In patients with bleeding refractory to medical treatment a series of maneuvers have been employed, including ligating the anterior and posterior ethmoidal arteries through a medial incision in the canthus orbit. Even more drastic measures have included ligation of the internal carotid artery. Unfortunately, many of these procedures fail because of the rich and diverse blood supply to the nasal cavity. Blood arises not only from branches of the internal carotid artery, but also from the external carotid artery, and by simply ligating one or the other of these branches blood flow may not be stemmed.
Determination of the specific site of bleeding can be achieved radiologically.
By placing a catheter from the femoral artery through the aorta and into the carotid circulation the sphenopalatine artery can be easily cannulated from the maxillary branch of the external carotid artery. Bleeding can usually be demonstrated and the vessel can be embolized using small particles.
Fortunately in this young boy’s case, bleeding stopped after further medical management and he remained asymptomatic.
Case 7 COMPLICATION OF ORBITAL FRACTURE
A 35-year-old man was involved in a fight and sustained a punch to the right orbit. He came to the emergency department with double vision.
The double vision was only in one plane.
Examination of the orbits revealed that when the patient was asked to look upward the right eye was unable to rotate superiorly. There was some limitation in general eye movement. Specifically, assessment of the lateral rectus muscle (abducent nerve [VI]), superior oblique muscle (trochlear nerve [IV]), and the rest of the eye muscles (oculomotor nerve [III]) was otherwise unremarkable.
The patient underwent a CT scan.
A CT scan of the facial bones demonstrated a fracture through the floor of the orbit (Fig. 8.282).
A careful review of this CT scan demonstrated that the inferior oblique muscle had been pulled inferiorly with the fragment of bone in the fracture. This produced a tethering effect, so when the patient was asked to gaze in the upward direction the left eye did, but the right eye was unable to because of the tethered inferior oblique muscle.
The patient underwent surgical exploration to elevate the small bony fragment and return the inferior oblique to its appropriate position. On follow-up the patient had no complications.
Case 8 BRAINSTEM TUMOR
A 30-year-old man came to the emergency department with increasing headaches, which were worse in the morning. He also complained of some blurring of vision and a nonspecific alteration in his overall motor abilities.
Examination of the patient by a medical student did not reveal any significant motor or sensory abnormality. The student was diligent and examined all cranial nerves.
Ophthalmoscopy revealed marked bulging of the peripheral margins of the optic nerve and the appearance of vessels hooking over the edge of this bulging periphery.
A diagnosis of raised intracranial pressure was made and a CT scan was performed.
The CT scan demonstrated dilated lateral and third ventricles, and a normal fourth ventricle.
A diagnosis of hydrocephalus was made, and given the absence of dilatation of the fourth ventricle a blockage was suspected at the level of the aqueduct (noncommunicating hydrocephalus).
An MRI scan was obtained.
An MRI scan with intravenous contrast demonstrated a small mass in the midbrain at and around the region of the aqueduct.
A brainstem tumor was diagnosed and the patient underwent treatment.
The medical student’s findings and the site of the tumor do not “add up.”
In the presence of hydrocephalus the ventricles dilate in series from the lateral ventricles onward. If this obstruction is in the region of the foramen of Monro, only the lateral ventricles are dilated. If the obstruction is in the region of the aqueduct, the lateral and third ventricles are dilated. It is unusual for the lateral and median foramina of the fourth ventricle to be obstructed and usually the next point of obstruction is in the region of the arachnoid granulations, secondary to blood and pertinacious fluid blocking the resorption of cerebrospinal fluid. In these cases the cerebrospinal fluid pressure around the brain is typically elevated, producing the changes around the optic nerve described and demonstrated at ophthalmoscopy.
An ophthalmologist examined the eye.
The eye was actually normal and it was an unfortunate misdiagnosis (though a good learning exercise) by the medical student. Importantly, the patient’s tumor was diagnosed and treatment was instituted. Ophthalmoscopy is a difficult skill to acquire and takes many hours of practice.
Case 9 PITUITARY MACROADENOMA
A 30-year-old woman came to her doctor with a history of amenorrhea (absence of menses) and galactorrhea (the production of breast milk). She was not pregnant and appeared otherwise fit and well.
Serum prolactin was measured.
Prolactin is a hormone produced by the pituitary gland and necessary for the production of breast milk postpartum. This hormone was markedly elevated.
Further clinical tests demonstrated visual field defects.
The patient went to see an optometrist who performed a visual field assessment and demonstrated a reduction in the lateral aspects of the normal visual fields. This was bilateral and symmetrical—a bilateral temporal hemianopia.
The visual pathways have now determined the site of the lesion.
Visual information from the temporal fields is projected onto the medial aspect of the retina bilaterally. The visual information from the medial aspects of the retina is carried in fibers that cross the midline through the optic chiasm to the opposite side.
The lesion is in the area of the optic chiasm.
Any disruption of the optic chiasm produces the field defect of bitemporal hemianopia. Tumors of the optic chiasm are unusual, though gliomas do occur. More frequently, compression of the optic chiasm by tumors in the vicinity is the usual cause for bitemporal hemianopia.
A pituitary tumor was diagnosed.
The optic chiasm is anterior and extremely close to the pituitary gland. Given that the patient is producing excessive amounts of prolactin (a pituitary tumor) and there is loss of the function of the chiasm, the most likely clinical explanation is an exophytic pituitary tumor compressing the optic chiasm.
An MRI scan was performed and demonstrated a large tumor (macroadenoma) of the pituitary gland.
Drug treatment was commenced and the tumor shrank. The endocrinological effects of the prolactin secretion also stopped.
Follow-up scans were performed.
Over the ensuing few years the tumor shrank. Unfortunately, the patient again began to secrete prolactin and surgery was performed (Fig. 8.283).
A transsphenoidal approach was undertaken.
With meticulous accuracy a series of very fine instruments was passed through the nasal cavity into the sphenoid bone. The bone was drilled and via this approach the pituitary gland was removed.
Extreme care must be taken because on both sides of the pituitary gland is the cavernous sinus through which the internal carotid artery, oculomotor nerve [III], trochlear nerve [IV], trigeminal nerve [V], and abducent nerve [VI] pass.
 the superior horn is connected by a lateral thyrohyoid ligament to the posterior end of the greater horn of the hyoid bone.
the superior horn is connected by a lateral thyrohyoid ligament to the posterior end of the greater horn of the hyoid bone. the base is concave and articulates with the sloping articular facet on the superolateral surface of the lamina of cricoid cartilage;
the base is concave and articulates with the sloping articular facet on the superolateral surface of the lamina of cricoid cartilage; the anterolateral surface has two depressions, separated by a ridge, for muscle (vocalis) and ligament (vestibular ligament) attachment.
the anterolateral surface has two depressions, separated by a ridge, for muscle (vocalis) and ligament (vestibular ligament) attachment.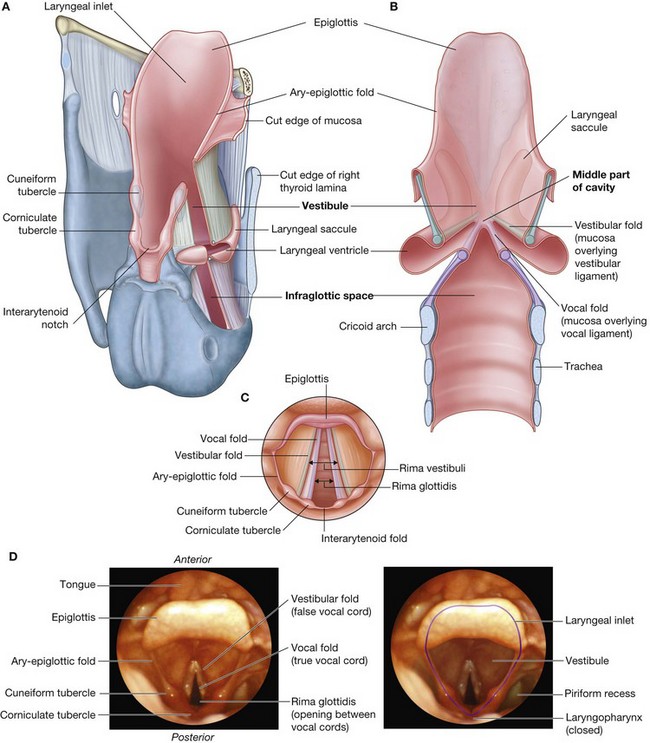
 its lateral borders are formed by mucosal folds (aryepiglottic folds), which enclose the superior margins of the quadrangular membranes and adjacent soft tissues, and two tubercles on the more posterolateral margin of the laryngeal inlet on each side mark the positions of the underlying cuneiform and corniculate cartilages;
its lateral borders are formed by mucosal folds (aryepiglottic folds), which enclose the superior margins of the quadrangular membranes and adjacent soft tissues, and two tubercles on the more posterolateral margin of the laryngeal inlet on each side mark the positions of the underlying cuneiform and corniculate cartilages; its posterior border in the midline is formed by a mucosal fold that forms a depression (interarytenoid notch) between the two corniculate tubercles.
its posterior border in the midline is formed by a mucosal fold that forms a depression (interarytenoid notch) between the two corniculate tubercles. the vestibule is the upper chamber of the laryngeal cavity between the laryngeal inlet and the vestibular folds, which enclose the vestibular ligaments and associated soft tissues;
the vestibule is the upper chamber of the laryngeal cavity between the laryngeal inlet and the vestibular folds, which enclose the vestibular ligaments and associated soft tissues; forcing soft tissues associated with the quadrangular membranes and vestibular ligaments toward the midline.
forcing soft tissues associated with the quadrangular membranes and vestibular ligaments toward the midline. the oblique part runs in a posterior direction from the arch of the cricoid cartilage to the inferior horn of the thyroid cartilage;
the oblique part runs in a posterior direction from the arch of the cricoid cartilage to the inferior horn of the thyroid cartilage; the straight part runs more vertically from the arch of the cricoid cartilage to the posteroinferior margin of the thyroid lamina.
the straight part runs more vertically from the arch of the cricoid cartilage to the posteroinferior margin of the thyroid lamina.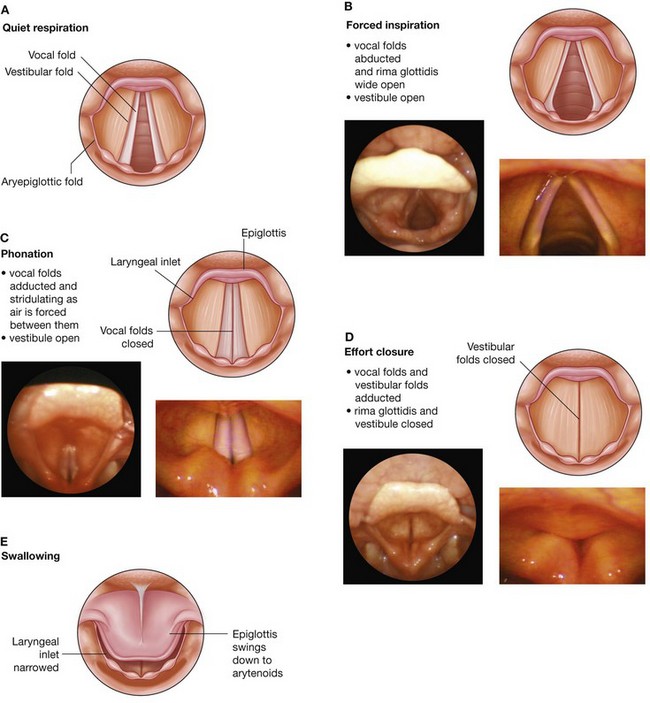
 the superior laryngeal artery originates near the upper margin of the thyroid cartilage from the superior thyroid branch of the external carotid artery, and accompanies the internal branch of the superior laryngeal nerve through the thyrohyoid membrane to reach the larynx;
the superior laryngeal artery originates near the upper margin of the thyroid cartilage from the superior thyroid branch of the external carotid artery, and accompanies the internal branch of the superior laryngeal nerve through the thyrohyoid membrane to reach the larynx; the inferior laryngeal artery originates from the inferior thyroid branch of the thyrocervical trunk of the subclavian artery low in the neck and, together with the recurrent laryngeal nerve, ascends in the groove between the esophagus and trachea—it enters the larynx by passing deep to the margin of the inferior constrictor muscle of the pharynx.
the inferior laryngeal artery originates from the inferior thyroid branch of the thyrocervical trunk of the subclavian artery low in the neck and, together with the recurrent laryngeal nerve, ascends in the groove between the esophagus and trachea—it enters the larynx by passing deep to the margin of the inferior constrictor muscle of the pharynx. superior laryngeal veins drain into superior thyroid veins, which in turn drain into the internal jugular veins (Fig. 8.215);
superior laryngeal veins drain into superior thyroid veins, which in turn drain into the internal jugular veins (Fig. 8.215);