60 Togaviruses and Flaviviruses
A 5-year-old Indonesian girl died of hemorrhagic shock. The presence of dengue virus serotype 3 in her blood was confirmed by reverse transcriptase polymerase chain reaction (RT-PCR).
1. How was the child infected with dengue virus?
2. What are the diseases caused by dengue virus?
3. What types of immune responses are protective? Potentially harmful?
Answers
1. Dengue is a mosquito-borne virus.
2. Dengue hemorrhagic fever and dengue shock syndrome.
3. Neutralizing antibody is protective, but a nonneutralizing antibody can facilitate uptake into macrophages, where the virus replicates and travels throughout the body.
4. Dengue is prevalent where the Aedes mosquito vector is prevalent, in tropical regions of the world.
The members of the Togaviridae and Flaviviridae families are enveloped, positive, single-stranded ribonucleic acid (RNA) viruses (Box 60-1). Alphavirus and Flavivirus are discussed together because of similarities in the diseases that they cause and in their epidemiology. Most are transmitted by arthropods and are therefore arboviruses (arthropod-borne viruses). They differ in size, morphology, gene sequence, and replication.
Box 60-1
Unique Features of Togaviruses and Flaviviruses
Viruses have enveloped, single-stranded, positive-sense RNA.
Togavirus replication includes early (nonstructural) and late (structural) protein synthesis.
Togaviruses replicate in the cytoplasm and bud at the plasma membranes.
Flaviviruses replicate in the cytoplasm and bud at intracellular membranes.
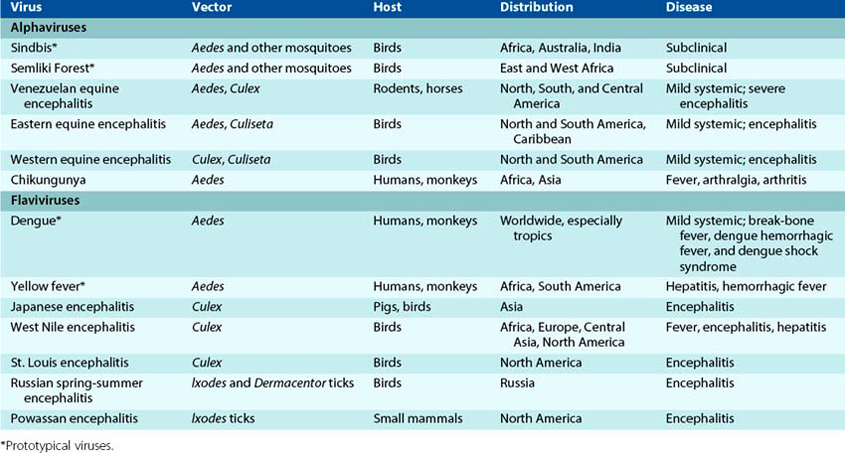
The Togaviridae (togaviruses) can be classified into the following major genera (Table 60-1): Alphavirus, Rubivirus, and Arterivirus. No known arteriviruses cause disease in humans, so this genus is not discussed further. Rubella virus is the only member of the Rubivirus group; it is discussed separately because its disease manifestation (German measles) and its means of spread differ from those of the alphaviruses. The Flaviviridae include the flaviviruses, pestiviruses, and hepaciviruses (hepatitis C and G viruses). Hepatitis C and G are discussed in Chapter 63.
Table 60-1 Togaviruses and Flaviviruses
| Virus Group | Human Pathogens |
|---|---|
| Togaviruses | |
| Alphavirus | Arboviruses |
| Rubivirus | Rubella virus |
| Arterivirus | None |
| Flaviviruses | Arboviruses |
| Hepaciviridae | Hepatitis C virus |
| Pestivirus | None |
Alphaviruses and Flaviviruses
The alphaviruses and flaviviruses are classified as arboviruses because they are usually spread by arthropod vectors. These viruses have a very broad host range, including vertebrates (e.g., mammals, birds, amphibians, reptiles) and invertebrates (e.g., mosquitoes, ticks). Diseases spread by animals or with an animal reservoir are called zoonoses. Examples of pathogenic alphaviruses and flaviviruses are listed in Table 60-2.
Structure and Replication of Alphaviruses
The alphaviruses have an icosahedral capsid and a positive-sense, single-strand RNA genome that resembles messenger RNA (mRNA). They are slightly larger than picornaviruses (45 to 75 nm in diameter) and are surrounded by an envelope (Latin toga, “cloak”). The togavirus genome encodes early and late proteins.
Alphaviruses have two or three glycoproteins that associate to form a single spike. The carboxy (COOH) terminus of the glycoproteins is anchored in the capsid, forcing the envelope to wrap tightly (“shrink-wrap”) and take on the shape of the capsid (Figure 60-1). The capsid proteins of all the alphaviruses are similar in structure and are antigenically cross-reactive. The envelope glycoproteins express unique antigenic determinants that distinguish the different viruses and also express antigenic determinants that are shared by a group, or “complex,” of viruses.
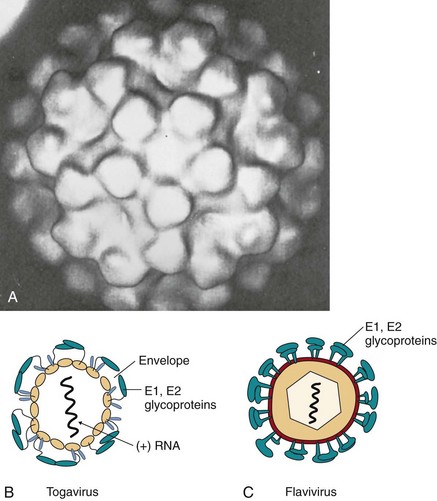
Figure 60-1 Alphavirus morphology. A, Morphology of the alphavirus virion obtained from cryoelectron microscopy and image processing of the micrographs to show that the envelope is held tightly and conforms to the icosahedral shape and symmetry of the capsid. B, Cross-section of alpha-togavirus. C, Cross-section of flavivirus. The envelope protein surrounds the membrane envelope, which encloses an icosahedral nucleocapsid. RNA, Ribonucleic acid.
(A, From From Fuller SD: The T = 4 envelope of Sindbis virus is organized by interactions with a complementary T = 3 capsid, Cell 48:923–934, 1987.)
The alphaviruses attach to specific receptors expressed on many different cell types from many different species (Figure 60-2). The host range for these viruses includes vertebrates, such as humans, monkeys, horses, birds, reptiles, and amphibians, and invertebrates, such as mosquitoes and ticks. However, the individual viruses have different tissue tropisms, accounting somewhat for the different disease presentations.
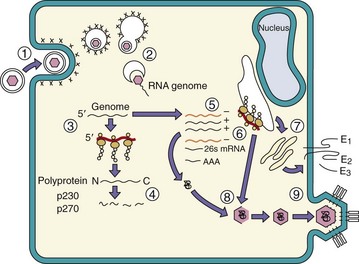
Figure 60-2 Replication of a togavirus. Semliki Forest virus. 1, Semliki Forest virus binds to cell receptors and is internalized in a coated vesicle. 2, On acidification of the endosome, the viral envelope fuses with the endosomal membrane to release the nucleocapsid into the cytoplasm. 3, Ribosomes bind to the positive-sense ribonucleic acid (RNA) genome, and the p230 or p270 (full-length) early polyproteins are made. 4, The polyproteins are cleaved to produce nonstructural proteins 1 to 4 (NSP1 to NSP4), which include a polymerase to transcribe the genome into a negative-sense RNA template. 5, The template is used to produce a full-length 42S positive-sense mRNA genome and a late 26S mRNA for the structural proteins. 6, The capsid (C) protein is translated first, exposing a protease cleavage site and then a signal peptide for association with the endoplasmic reticulum. 7, The E glycoproteins are then synthesized, glycosylated, processed in the Golgi apparatus, and transferred to the plasma membrane. 8, The capsid proteins assemble on the 42S genomic RNA and then associate with regions of cytoplasmic and plasma membranes containing the E1, E2, and E3 spike proteins. 9, Budding from the plasma membrane releases the virus. AAA, Polyadenylate; mRNA, messenger ribonucleic acid.
The virus enters the cell by means of receptor-mediated endocytosis (see Figure 60-2). The viral envelope then fuses with the membrane of the endosome on acidification of the vesicle to deliver the capsid and genome into the cytoplasm.
Once released into the cytoplasm, the alphavirus genomes bind to ribosomes as mRNA. The alphavirus genome is translated in early and late phases. The initial two thirds of the alphavirus RNA is translated into a polyprotein, which is subsequently cleaved into four nonstructural early proteins (NSPs 1 through 4). The protease is part of the polyprotein and precedes the site of cleavage. Each of these proteins is a portion of the RNA-dependent RNA polymerase. A full-length, 42S, negative-sense RNA is synthesized as a template for replication of the genome, and more 42S positive-sense mRNA is produced. In addition, a 26S late mRNA, corresponding to one third of the genome, is transcribed from the template. The 26S RNA encodes the capsid (C) and envelope (E1 through E3) proteins. Late in the replication cycle, viral mRNA can account for as much as 90% of the mRNA in the infected cell. The abundance of late mRNAs allows the production of a large amount of the structural proteins required for packaging the virus.
The structural proteins are produced by protease cleavage of the late polyprotein that was produced from the 26S mRNA. The C protein is translated first and is cleaved from the polyprotein. A signal sequence is then made that associates the nascent polypeptide with the endoplasmic reticulum. Thereafter, envelope glycoproteins are translated, glycosylated, and cleaved from the remaining portion of the polyprotein to produce the E1, E2, and E3 glycoprotein spikes. The E3 is released from most alphavirus glycoprotein spikes. The glycoproteins are processed by the normal cellular machinery in the endoplasmic reticulum and Golgi apparatus and are also acetylated and acylated with long-chain fatty acids. Alphavirus glycoproteins are then transferred efficiently to the plasma membrane.
The C proteins associate with the genomic RNA soon after their synthesis and form an icosahedral capsid. Once this step is completed, the capsid associates with portions of the membrane expressing the viral glycoproteins. The alphavirus capsid has binding sites for the C-terminus of the glycoprotein spike, which pulls the envelope tightly around itself in a manner like shrink-wrapping (see Figures 60-1 and 60-2). Alphaviruses are released on budding from the plasma membrane.
Of interest, the western equine encephalitis virus (WEEV) was created by recombination of two alphaviruses, the eastern equine encephalitis virus (EEEV) and the Sindbis virus. The beginning of the WEEV genome is almost identical to EEEV, with similar glycoproteins and virulence genes, while the end of the genome resembles Sindbis.
Structure and Replication of Flaviviruses
The flaviviruses also have a positive-strand RNA genome, an icosahedral capsid, and an envelope but are slightly smaller than an alphavirus (40 to 65 nm in diameter). The E viral glycoprotein folds over, pairs up with another E glycoprotein, and lies flat across the surface of the virion to form an outer protein layer (see Figure 60-1). Most of the flaviviruses are serologically related, and antibodies to one virus may neutralize another virus.
The attachment and penetration of the flaviviruses occur in the same way as described for the alphaviruses. Flaviviruses also enter macrophages, monocytes, and other cells that have Fc receptors, when the virus is coated with antibody. The antibody actually enhances the infectivity of these viruses by providing new receptors for the virus and promoting viral uptake into these target cells.
The major differences between alphaviruses and flaviviruses are in the organization of their genomes and their mechanisms of protein synthesis. The entire flavivirus genome is translated into a single polyprotein in a manner more similar to the process for picornaviruses than for alphaviruses (Figure 60-3). As a result, there is no temporal distinction in the translation of the different viral proteins. The polyprotein produced from the yellow fever genome contains five nonstructural proteins, including a protease and components of the RNA-dependent RNA polymerase, plus the capsid and envelope structural proteins.
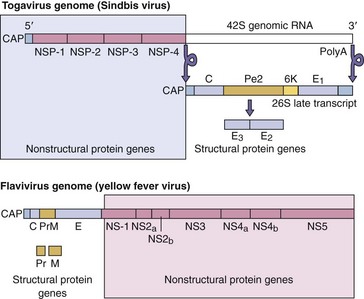
Figure 60-3 Comparison of the togavirus (alphavirus) and flavivirus genomes. Alphavirus: The enzymatic activities are translated from the 5′-end of the input genome, promoting their early rapid translation. The structural proteins are translated later from a smaller messenger ribonucleic acid (mRNA) transcribed from the genomic template. Flavivirus: The genes for the structural proteins of the flaviviruses are at the 5′-end of the genome/mRNA, and only one species of polyprotein is made, which represents the entire genome. PolyA, Polyadenylate.
Unlike in the alphavirus genome, the structural genes are at the 5′-end of the flavivirus genome. As a result, the portions of the polyprotein containing the structural (not the catalytic) proteins are synthesized first and with the greatest efficiency. This arrangement may allow the production of more structural proteins, but it decreases the efficiency of nonstructural protein synthesis and the initiation of viral replication. This feature of flaviviruses may contribute to the lag before detection of their replication.
The entire flavivirus polyprotein associates with the endoplasmic reticulum membrane and then is cleaved into its components. Unlike the togaviruses, the flaviviruses acquire their envelope by budding into the endoplasmic reticulum rather than at the cell surface. The virus is then released by exocytosis or cell lysis mechanisms. This route is less efficient, and the virus may remain cell-associated.
Pathogenesis and Immunity
Because the arboviruses are acquired from the bite of an arthropod such as a mosquito, knowledge of the course of infection in both the vertebrate host and the invertebrate vector is important for an understanding of the diseases. These viruses can cause lytic or persistent infections of both vertebrate and invertebrate hosts (Box 60-2). Infections of invertebrates are usually persistent, with continued virus production.
Box 60-2
Disease Mechanisms of Togaviruses and Flaviviruses
Viruses are cytolytic, except for rubella and hepatitis C.
Viruses establish viremia and systemic infection.
Viruses are good inducers of interferon, which can account for the flulike symptoms during prodrome.
Viruses, except rubella and hepatitis C, are arboviruses.
Flaviviruses can infect cells of the monocyte-macrophage lineage. Nonneutralizing antibody can enhance flavivirus infection via Fc receptors on the cells.
The death of an infected cell results from a combination of virus-induced insults. The large amount of viral RNA produced on the replication and transcription of the genome blocks cellular mRNA from binding to ribosomes. Increased permeability of the target cell membrane and changes in ion concentrations can alter enzyme activities and favor the translation of viral mRNA over cellular mRNA. The displacement of cellular mRNA from the protein synthesis machinery prevents rebuilding and maintenance of the cell and is a major cause of the death of the virus-infected cell. Some alphaviruses, such as WEEV, make a nucleotide triphosphatase that degrades deoxyribonucleotides, depleting even the substrate pool for deoxyribonucleic acid (DNA) production.
Female mosquitoes acquire the alphaviruses and flaviviruses by taking a blood meal from a viremic vertebrate host. A sufficient viremia must be maintained in the vertebrate host to allow acquisition of the virus by the mosquito. The virus then infects the epithelial cells of the midgut of the mosquito, spreads through the basal lamina of the midgut to the circulation, and infects the salivary glands. The virus sets up a persistent infection and replicates to high titers in these cells. The salivary glands can then release virus into the saliva. Not all arthropod species support this type of infection, however. For example, the normal vector for the WEEV is the Culex tarsalis mosquito, but certain strains of virus are limited to the midgut of this mosquito, cannot infect its salivary glands, and therefore cannot be transmitted to humans.
On biting a host, the female mosquito regurgitates virus-containing saliva into the victim’s bloodstream. The virus then circulates freely in the host’s plasma and comes into contact with susceptible target cells, such as the endothelial cells of the capillaries, monocytes, dendritic cells, and macrophages.
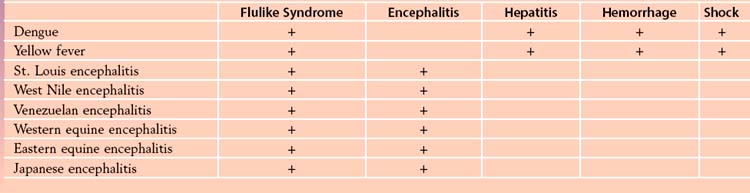
The nature of alphavirus and flavivirus disease is determined primarily by (1) the specific tissue tropisms of the individual virus type, (2) the concentration of infecting virus, and (3) individual responses to the infection. These viruses are associated with mild systemic disease, encephalitis, arthrogenic disease, or hemorrhagic disease.
The initial viremia produces systemic symptoms, such as fever, chills, headaches, backaches, and other flulike symptoms, within 3 to 7 days of infection. Some of these symptoms can be attributed to the effects of the interferon produced in response to the viremia and infection of host cells. The viremia is considered a mild systemic disease, and most viral infections do not progress beyond this point. A secondary viremia can produce sufficient virus to infect target organs, such as the brain, liver, skin, and vasculature, depending on the tissue tropism of the virus (Figure 60-4). The virus gains access to the brain by infecting the endothelial cells lining the small vessels of the brain or the choroid plexus.
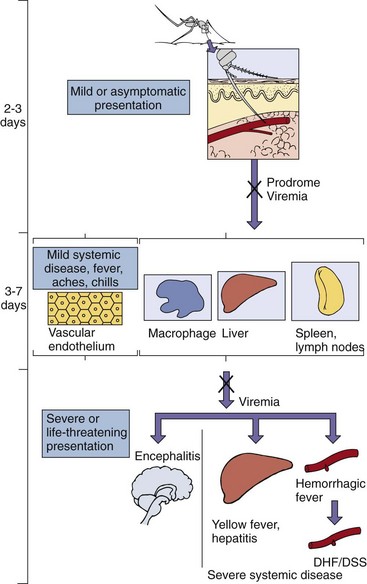
Figure 60-4 Disease syndromes of the alphaviruses and flaviviruses. Primary viremia may be associated with mild systemic disease. Most infections are limited to this. If sufficient virus is produced during the secondary viremia to escape innate and immune protection and to reach critical target tissues, severe systemic disease or encephalitis may result. If antibody is present (X), viremia is blocked. For dengue virus, rechallenge with another strain can result in severe dengue hemorrhagic fever (DHF), which can cause dengue shock syndrome (DSS) because of the loss of fluids from the vasculature.
The primary target cells of the flaviviruses are of the monocyte-macrophage lineage. Although these cells are found throughout the body and may have different characteristics, they express Fc receptors for antibody and release cytokines on challenge. Flavivirus infection is enhanced 200- to 1000-fold by nonneutralizing antiviral antibody that promotes binding of the virus to the Fc receptors and its uptake into the cell.
Immune Response
Both humoral immunity and cellular immunity are elicited and are important to the control of primary infection and the prevention of future infections with the alphaviruses and flaviviruses.
Replication of the alphaviruses and flaviviruses produces a double-stranded RNA replicative intermediate that is a good inducer of interferon-α and interferon-β. The interferon is released into the bloodstream and limits replication of the virus; it also stimulates innate and immune responses but in doing so causes the rapid onset of the flulike symptoms characteristic of mild systemic disease.
Circulating immunoglobulin M (IgM) is produced within 6 days of infection, followed by the production of IgG. The antibody blocks the viremic spread of the virus and the subsequent infection of other tissues. Through recognition of the type-common antigens expressed on all viruses in the family, immunity to one flavivirus can provide some protection against infection with other flaviviruses. Cell-mediated immunity is also important in controlling the primary infection.
Immunity to these viruses is a double-edged sword. Inflammation resulting from the cell-mediated immune response can destroy tissues and significantly contribute to the pathogenesis of encephalitis. Hypersensitivity reactions initiated by the formation of immune complexes with virions and viral antigens, and the activation of complement, can also occur. They can weaken the vasculature and cause it to rupture, leading to hemorrhagic symptoms. A nonneutralizing antibody can enhance the uptake of flaviviruses into macrophages and other cells that express Fc receptors. Such an antibody can be generated to a related strain of virus in which the neutralizing epitope is not expressed or is different. Immune responses to a related strain of dengue virus that do not prevent infection can promote immunopathogenesis, leading to dengue hemorrhagic fever or dengue shock syndrome.
Epidemiology
Alphaviruses and most flaviviruses are prototypical arboviruses (Box 60-3). To be an arbovirus, the virus must be able to (1) infect both vertebrates and invertebrates, (2) initiate a sufficient viremia in a vertebrate host for a sufficient time to allow acquisition of the virus by the invertebrate vector, and (3) initiate a persistent productive infection of the salivary gland of the invertebrate to provide virus for the infection of other host animals. Humans are usually “dead-end” hosts, in that they cannot spread the virus back to the vector because they do not maintain a persistent viremia. If the virus is not in the blood, the mosquito cannot acquire it. A full cycle of infection occurs when the virus is transmitted by the arthropod vector and amplified in a susceptible, immunologically naïve host (reservoir) that allows the reinfection of other arthropods (Figure 60-5). The vectors, natural hosts, and geographic distribution of representative alphaviruses and flaviviruses are listed in Table 60-2.
Box 60-3
Epidemiology of Alphavirus and Flavivirus Infection
Geography/Season
Endemic regions for each arbovirus are determined by habitat of mosquito or other vector
Aedes mosquito, which carries dengue and yellow fever, is found in urban areas and in pools of water
Culex mosquito, which carries St. Louis encephalitis and West Nile encephalitis viruses, is found in forest and urban areas
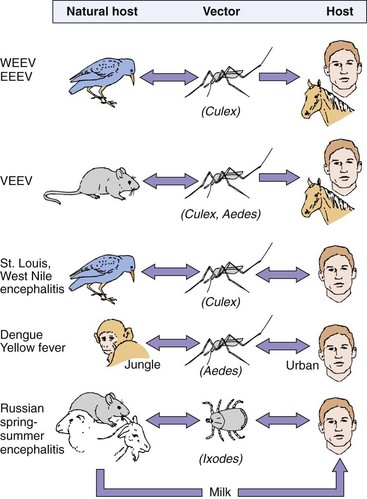
Figure 60-5 Patterns of alphavirus and flavivirus transmission. Birds and small mammals are the hosts that maintain and amplify an arbovirus, which is spread by the insect vector upon a blood meal. A double arrow indicates a cycle of replication in both host (including man) and vector. “Dead-end” infections with no transmission of the virus back to the vector are indicated by the single arrow. EEEV, Eastern equine encephalitis virus; VEEV, Venezuelan equine encephalitis virus; WEEV, western equine encephalitis virus.
These viruses are usually restricted to a specific arthropod vector, its vertebrate host, and their ecologic niche. The most common vector is the mosquito, but ticks and sandflies spread some arboviruses. Even in a tropical region overrun with mosquitoes, the spread of these viruses is still restricted to a specific genus of mosquitoes. Not all arthropods can act as good vectors for each virus. For example, Culex quinquefasciatus is resistant to infection by the WEEV (alphavirus) but is an excellent vector for St. Louis encephalitis virus (flavivirus).
Birds and small mammals are the usual reservoir hosts for the alphaviruses and flaviviruses, but reptiles and amphibians can also act as hosts. A large population of viremic animals can develop in these species to continue the infection cycle of the virus. For example, West Nile encephalitis virus (WNV) was first noted in 1999 as an outbreak in New York by the unusual deaths of captive birds at the Bronx Zoo. Reverse transcriptase polymerase chain reaction (RT-PCR) analysis identified the virus as WNV. The virus is transmitted by Culex pipiens mosquitoes, and crows, blue jays, and other wild birds are the reservoir. The virus spread throughout the United States, and by 2006, the virus and human disease had been noted in almost every state. WNV establishes a sufficient viremia in humans to be a risk factor for transmission through blood transfusions. Documentation of two such cases has led to screening blood donors for WNV and rejecting donors who have fever and headache during the week of blood donation.
Arbovirus diseases occur during the summer months and rainy seasons, when the arthropods breed, and the arboviruses are cycled among a host reservoir (birds), an arthropod (e.g., mosquitoes), and human hosts. This cycle maintains and increases the amount of virus in the environment. In the winter, the vector is not present to maintain the virus. The virus may either (1) persist in arthropod larvae or eggs or in reptiles or amphibians that remain in the locale or (2) migrate with the birds and then return during the summer.
When humans travel into the ecologic niche of the mosquito vector, they risk being infected by the virus. Pools of standing water, drainage ditches, and trash dumps in cities can also provide breeding grounds for mosquitoes such as Aedes aegypti, the vector for yellow fever, dengue, and chikungunya. An increase in the population of these mosquitoes therefore puts the human population at risk for infection. Health departments in many areas monitor birds and mosquitoes caught in traps for arboviruses and initiate control measures, such as insecticide spraying, when necessary.
Urban outbreaks of arbovirus infections occur when the reservoirs for the virus are humans or urban animals. Humans can be reservoir hosts for yellow fever, dengue, and chikungunya viruses (see Figure 60-5). These viruses are maintained by Aedes mosquitoes in a sylvatic or jungle cycle, in which monkeys are the natural host, and also in an urban cycle, in which humans are the host. A. aegypti, a vector for each of these viruses, is a household mosquito. It breeds in pools of water, open sewers, and other accumulations of water in cities. The occurrence of numerous inapparent infections in high-density populations provides enough viremic human hosts for the continued spread of these viruses. St. Louis encephalitis and WNV are maintained in an urban environment because their vectors, Culex mosquitoes, breed in stagnant water, including puddles and sewage, and the reservoir group includes common city birds (e.g., crows).
Clinical Syndromes
More humans are infected with alphaviruses and flaviviruses than show significant, characteristic symptoms. The incidence of arbovirus disease is sporadic. Alphavirus infections are usually asymptomatic or cause low-grade disease, such as flulike symptoms (chills, fever, rash, and aches) that correlate with systemic infection during the initial viremia. EEEV, WEEV, and Venezuelan equine encephalitis virus (VEEV) infections can progress to encephalitis in humans. The equine encephalitis viruses are usually more of a problem to livestock than to humans. An affected human may experience fever, headache, and decreased consciousness 3 to 10 days after infection. Unlike herpes simplex virus encephalitis, the disease generally resolves without sequelae, but there is the possibility of paralysis, mental disability, seizures, and death. The name chikungunya (Swahili for “that which bends up”) refers to the crippling arthritis associated with serious disease caused by infection with these viruses. Although prevalent in South America and from western Africa across southern Asia to the Philippines, this disease may spread to the United States because of the return of the A. aegypti mosquito, its vector.
Most flavivirus infections are relatively benign, but serious aseptic meningitis and encephalitic or hemorrhagic disease can occur. The encephalitis viruses include St. Louis, West Nile, Japanese, Murray Valley, and Russian spring-summer viruses. Symptoms and outcomes are similar to those of the togavirus encephalitides. Hundreds to thousands of cases of St. Louis encephalitis virus disease are noted in the United States annually. Approximately 20% of individuals infected with WNV will develop West Nile fever, characterized by fever, headache, tiredness, and body aches, occasionally with a skin rash on the trunk of the body and swollen lymph glands usually lasting only a few days (Clinical Case 60-1). Encephalitis, meningitis, or meningoencephalitis occurs in approximately 1% of WNV-infected individuals, with risk increasing with age.
Clinical Case 60-1
West Nile Encephalitis Virus (WNV)
Hirsch and Warner (N Engl J Med 348:2239–2247, 2003) described the case of a 38-year-old Massachusetts woman who presented with a progressively worsening headache, with photophobia and fever. Because it was August, she was on summer vacation and 10 days earlier (−10) had traveled to St. Louis and stayed for 8 days. While there, she walked in the woods and visited the zoo. A day before the onset of these symptoms (−1), she vacationed along the Atlantic shore and noted that she had been bitten by mosquitos and removed ticks from her dog. Four days later (+4), she was admitted with fever (40° C), chills, rapid heartbeat, confusion, lightheadedness, and lethargy. Although appearing alert, oriented, and only slightly ill, her neck was rigid and Kernig sign was present. The signs of meningitis prompted testing of cerebrospinal fluid, which contained immunoglobulin M (IgM) to WNV and low titers to St. Louis encephalitis virus (SLE). Patient antibody neutralized WNV but not SLE virus infection of tissue culture cells, suggesting that the activity to SLE was due to cross-reactivity between flaviviruses. Tests for other organisms were negative. She was treated empirically for meningitis and for herpes simplex virus (HSV) (acyclovir). Antibacterial and anti-HSV treatment for meningitis and encephalitis were necessary until the laboratory results were available. On day 5 postonset, she became more lethargic and had difficulty answering questions. A magnetic resonance image (MRI) indicated subtle changes in the brain. On day 6, she could not distinguish her right from her left hand, but her headache lessened, and she could respond to commands. On day 7, she had a tremor in her right arm, but her mental status was improving, and by day 8, she was alert and lucid. On day 9, a cranial MRI was normal; on day 10, she was recovered; and on day 11, she was released from the hospital. The season of the year, exposure to insects, and travel by this woman were suggestive of several different arboviral encephalitis diseases, in addition to WNV. Viruses in the differential diagnosis included eastern equine encephalitis, SLE, Powassan virus (tick-borne flavivirus), HSV, and WNV. Unlike HSV encephalitis, flavivirus meningoencephalitis usually resolves with limited sequelae.
The hemorrhagic viruses are dengue and yellow fever viruses. Dengue virus is a major worldwide problem, with up to 100 million cases of dengue fever and 300,000 cases of dengue hemorrhagic fever (DHF) occurring per year. The virus and its vector are present in central and northern South America, and cases have occurred in Puerto Rico, Texas, and Florida. The incidence of the more serious DHF has quadrupled since 1985. Dengue fever is also known as break-bone fever; the symptoms and signs consist of high fever, headache, rash, and back and bone pain that last 6 to 7 days. On rechallenge with another of the four related strains, dengue can also cause DHF and dengue shock syndrome (DSS). Nonneutralizing antibody promotes uptake of the virus into macrophages, which causes memory T cells to become activated, release cytokines, and initiate inflammatory reactions. These reactions and the virus result in weakening and rupture of the vasculature, internal bleeding, and loss of plasma, leading to shock symptoms and internal bleeding. In 1981 in Cuba, dengue-2 virus infected a population previously exposed to dengue-1 virus between 1977 and 1980, leading to an epidemic of more than 100,000 cases of DHF/DSS and 168 deaths.
Yellow fever infections are characterized by severe systemic disease, with degeneration of the liver, kidney, and heart, as well as hemorrhage. Liver involvement causes the jaundice from which the disease gets its name, but massive gastrointestinal hemorrhages (“black vomit”) may also occur. The mortality rate associated with yellow fever during epidemics is as high as 50%.
Laboratory Diagnosis
The alphaviruses and flaviviruses can be grown in both vertebrate and mosquito cell lines, but most are difficult to isolate. Infection can be detected through the use of cytopathologic studies, immunofluorescence, and the hemadsorption of avian erythrocytes. Detection and characterization can be performed by RT-PCR testing of genomic RNA or viral mRNA in blood or other samples. After isolation, the viral RNA can also be distinguished by the finding of RNA “fingerprints” of the genomic RNA. Monoclonal antibodies to the individual viruses have become a useful tool for distinguishing the individual species and strains of viruses.
A variety of serologic methods can be used to diagnose infections, including hemagglutination inhibition, enzyme-linked immunosorbent assays, and latex agglutination. The presence of specific IgM or a fourfold increase in titer between acute and convalescent sera is used to indicate a recent infection. The serologic cross-reactivity among viruses limits distinction of the actual viral species in many cases.
Treatment, Prevention, and Control
No treatments exist for arbovirus diseases, other than supportive care. The easiest means of preventing the spread of any arbovirus is elimination of its vector and breeding grounds. After 1900, when Walter Reed and his colleagues discovered that yellow fever was spread by A. aegypti, the number of cases was reduced from 1400 to none within 2 years, purely through control of the mosquito population. Many public health departments monitor the bird and mosquito populations in a region for arboviruses and periodically spray to reduce the mosquito population. Avoidance of the breeding grounds of a mosquito vector is also a good preventive measure.
A live vaccine against yellow fever virus and killed vaccines against EEEV, WEEV, Japanese encephalitis virus, and Russian spring-summer encephalitis virus are available. A live Japanese encephalitis virus vaccine is used in China. These vaccines are meant for people working with the virus or at risk for contact. A live vaccine against VEEV is available but only for use in domestic animals. Vaccines consisting of all four strains of dengue virus are being developed to ensure that immune enhancement of the disease on subsequent challenge does not occur.
The yellow fever vaccine is prepared from the 17D strain isolated from a patient in 1927 and grown for long periods in monkeys, mosquitoes, embryonic tissue culture, and embryonated eggs. The vaccine is administered intradermally and elicits lifelong immunity to yellow fever and possibly other cross-reacting flaviviruses.
Rubella Virus
Rubella virus has the same structural properties and mode of replication as the other togaviruses. However, unlike the other togaviruses, rubella is a respiratory virus and does not cause readily detectable cytopathologic effects.
Rubella is one of the five classic childhood exanthems, along with measles, roseola, fifth disease, and chickenpox. Rubella, meaning “little red” in Latin, was first distinguished from measles and other exanthems by German physicians; thus the common name for the disease, German measles. In 1941, an astute Australian ophthalmologist, Norman McAlister Gregg, recognized that maternal rubella infection was the cause of congenital cataracts. Maternal rubella infection has since been correlated with several other severe congenital defects. This finding prompted the development of a unique program to vaccinate children to prevent infection of pregnant women and neonates.
Pathogenesis and Immunity
Rubella virus is not cytolytic but does have limited cytopathologic effects in certain cell lines, such as Vero and RK13. The replication of rubella prevents (in a process known as heterologous interference) the replication of superinfecting picornaviruses. This property allowed the first isolations of rubella virus in 1962.
Rubella infects the upper respiratory tract and then spreads to local lymph nodes, which coincides with a period of lymphadenopathy (Figure 60-6). This stage is followed by establishment of viremia, which spreads the virus throughout the body. Infection of other tissues and the characteristic mild rash occur. The prodromal period lasts approximately 2 weeks (Figure 60-7). The infected person can shed virus in respiratory droplets during the prodromal period and for as long as 2 weeks after the onset of the rash.
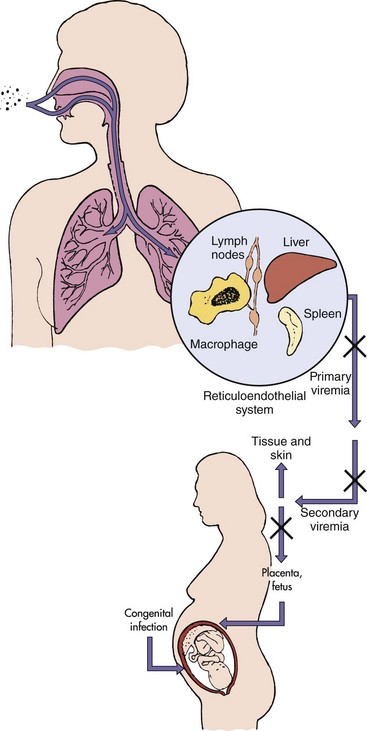
Figure 60-6 Spread of rubella virus within the host. Rubella enters and infects the nasopharynx and lung and then spreads to the lymph nodes and monocyte-macrophage system. The resulting viremia spreads the virus to other tissues and the skin. Circulating antibody can block the transfer of virus at the indicated points (X). In an immunologically deficient pregnant woman, the virus can infect the placenta and spread to the fetus.
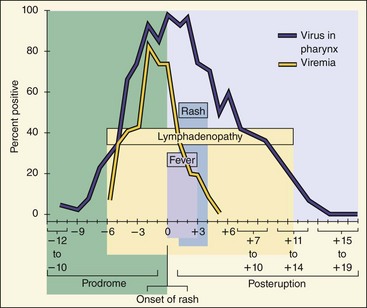
Figure 60-7 Time course of rubella disease. Rubella production in the pharynx precedes the appearance of symptoms and continues throughout the course of the disease. The onset of lymphadenopathy coincides with the viremia. Fever and rash occur later. The person is infectious as long as the virus is produced in the pharynx.
(Modified from Plotkin SA: Rubella vaccine. In Plotkin SA, Mortimer EA, editors: Vaccines, Philadelphia, 1988, WB Saunders.)
Immune Response
Antibody is generated after the viremia, and its appearance correlates with the appearance of the rash. The antibody limits viremic spread, but cell-mediated immunity plays an important role in resolving the infection. Only one serotype of rubella exists, and natural infection produces lifelong protective immunity. Most important, serum antibody in a pregnant woman prevents spread of the virus to the fetus. Immune complexes most likely cause the rash and arthralgia associated with rubella infection.
Congenital Infection
Rubella infection in a pregnant woman can result in serious congenital abnormalities in the child. If the mother does not have antibody, the virus can replicate in the placenta and spread to the fetal blood supply and throughout the fetus. Rubella can replicate in most tissues of the fetus. The virus may not be cytolytic, but the normal growth, mitosis, and chromosomal structure of the cells of the fetus can be altered by the infection. The alterations can lead to improper development of the fetus, small size of the infected baby, and the teratogenic effects associated with congenital rubella infection. The nature of the disorder is determined by (1) the tissue affected and (2) the stage of development disrupted.
The virus may persist in tissues, such as the lens of the eye, for 3 to 4 years and may be shed up to a year after birth. The presence of the virus during the development of the baby’s immune response may even have a tolerogenic effect on the system, preventing effective clearance of the virus after birth. Immune complexes that produce further clinical abnormalities may also form in the neonate or infant.
Epidemiology
Humans are the only host for rubella (Box 60-4). The virus is spread in respiratory secretions and is generally acquired during childhood. Spread of virus, before or in the absence of symptoms, and crowded conditions, such as those in day-care centers, promote contagion.
Approximately 20% of women of childbearing age escape infection during childhood and are susceptible to infection unless vaccinated. Programs in many states in the United States test expectant mothers for antibodies to rubella.
Before the development and use of the rubella vaccine, cases of rubella in schoolchildren would be reported every spring, and major epidemics of rubella occurred at regular 6- to 9-year intervals. The severity of the 1964 to 1965 epidemic in the United States is indicated in Table 60-3. Congenital rubella occurred in as many as 1% of all the children born in cities, such as Philadelphia, during this epidemic. The immunization program has succeeded in eliminating endemic rubella virus infection in the United States.
Table 60-3 Estimated Morbidity Associated with the 1964-1965 U.S. Rubella Epidemic
| Clinical Events | Number Affected |
|---|---|
| Rubella cases | 12,500,000 |
| Arthritis-arthralgia | 159,375 |
| Encephalitis | 2084 |
| Deaths | |
| Excess neonatal deaths | 2100 |
| Other deaths | 60 |
| TOTAL DEATHS | 2160 |
| Excess fetal wastage | 6250 |
| Congenital rubella syndrome | |
| Deaf children | 8055 |
| Deaf/blind children | 3580 |
| Mentally retarded children | 1790 |
| Other congenital rubella syndrome symptoms | 6575 |
| TOTAL CONGENITAL RUBELLA SYNDROME | 20,000 |
| Therapeutic abortions | 5000 |
From National Communicable Disease Center: Rubella surveillance, Report No. 1, Washington, DC, June 1969, U.S. Department of Health, Education, and Welfare.
Clinical Syndromes
Rubella disease is normally benign in children. After a 14- to 21-day incubation period, the symptoms in children consist of a 3-day maculopapular or macular rash and swollen glands (Figure 60-8). Infection in adults, however, can be more severe and include problems such as bone and joint pain (arthralgia and arthritis) and (rarely) thrombocytopenia or postinfectious encephalopathy. Immunopathologic effects resulting from cell-mediated immunity and hypersensitivity reactions are a major cause of the more severe forms of rubella in adults.
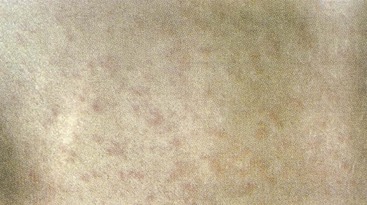
Figure 60-8 Close-up of the rubella rash. Small erythematous macules are visible.
(From Hart CA, Broadwell RL: A color atlas of pediatric infectious disease, London, 1992, Wolfe.)
Congenital disease is the most serious outcome of rubella infection. The fetus is at major risk until the 20th week of pregnancy. Maternal immunity to the virus resulting from prior exposure or vaccination prevents spread of the virus to the fetus. The most common manifestations of congenital rubella infection are cataracts, mental retardation, cardiac abnormalities, and deafness (Boxes 60-5 and 60-6; see Table 60-3). The mortality in utero and within the first year after birth is high for affected babies.
Box 60-6
Clinical Summaries
West Nile encephalitis: During August, a 70-year-old man from a swampy area of Louisiana develops fever, headache, muscle weakness, nausea, and vomiting. He has difficulty answering questions. He progresses into a coma. Magnetic resonance imaging results show no specific localization of lesions (unlike in herpes simplex virus encephalitis). His disease progresses to respiratory failure and death. His 25-year-old niece, living next door, complains of sudden onset of fever (39° C [102.2° F]), headache, and myalgias, with nausea and vomiting lasting 4 days. (See website www.postgradmed.com/issues/2003/07_03/gelfand.shtml.)
Yellow fever: A 42-year-old man had fever (103° F), headache, vomiting, and backache, which started 3 days after returning from a trip to Central America. He appeared normal for a short time, but then his gums started to bleed, and he had bloody urine, vomited blood, and developed petechiae, jaundice, and a slower and weakened pulse. He started to improve 10 days after the onset of disease.
Rubella: A 6-year-old girl from Romania develops a faint rash on her face, accompanied by mild fever and lymphadenopathy. Over the next 3 days, the rash progresses to other parts of the body. She has no history of rubella immunization.
Laboratory Diagnosis
Isolation of the rubella virus is difficult and rarely attempted. The presence of the virus can be detected by RT-PCR detection of viral RNA. The diagnosis is usually confirmed by the presence of antirubella-specific IgM. A fourfold increase in specific IgG antibody titer between acute and convalescent sera is also used to indicate a recent infection. Antibodies to rubella are assayed early in pregnancy to determine the immune status of the woman; this test is required in many states.
When isolation of the virus is necessary, the virus is usually obtained from urine and is detected as interference with replication of echovirus 11 in primary African green monkey kidney cell cultures.
Treatment, Prevention, and Control
No treatment is available for rubella. The best means of preventing rubella is vaccination with the live cold-adapted RA27/3 vaccine strain of virus (Figure 60-9). The live rubella vaccine is usually administered with the measles and mumps vaccines (MMR vaccine) at 24 months of age. The triple vaccine is included routinely in well-baby care. Vaccination promotes both humoral and cellular immunity.
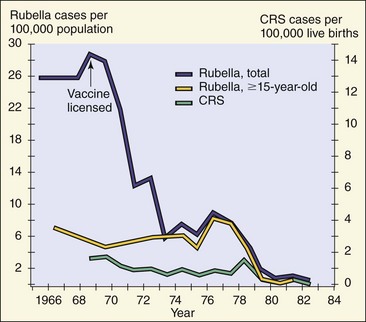
Figure 60-9 Effect of rubella virus vaccination on the incidence of rubella and congenital rubella syndrome (CRS).
(Modified from Williams MN, Preblud SR: Current trends: rubella and congenital rubella—United States, 1983, MMWR Morb Mortal Wkly Rep 33:237–247, 1984.)
The primary reason for the rubella vaccination program is to prevent congenital infection by decreasing the number of susceptible people in the population, especially children. As a result, there are fewer seronegative mothers and smaller chance that they will be exposed to the virus from contact with the children. Because only one serotype for rubella exists, and humans are the only reservoir, vaccination of a large proportion of the population can significantly reduce the likelihood of exposure to the virus.
A 27-year-old businessman experienced a high fever, serious retroorbital headache, and severe joint and back pain 5 days after he and his family returned from a trip to Malaysia. The symptoms lasted for 4 days, and then a rash appeared on his palms and soles, which lasted for 2 days. At the same time, the man’s 5-year-old son experienced mild flulike symptoms and then collapsed after 2 to 5 days. The boy’s hands were cold and clammy, his face was flushed, and his body was warm. There were petechiae on his forehead and ecchymoses elsewhere. He bruised very easily. He was breathing rapidly and had a weak, rapid pulse. He then rapidly recovered after 24 hours.
1. What features of these cases pointed to the diagnosis of dengue virus infection?
2. Of what significance was the trip to Malaysia?
3. What was the source of infection in the father and son?
4. What were the significance of and the pathogenic basis for the petechiae and ecchymoses in the child?
Two weeks after returning from a trip to Mexico, a 25-year-old man had arthralgia (joint aches) and a mild rash that started on his face and spread to his body. He recalled that he had felt as if he had the flu a few days before the onset of the rash. The rash disappeared in 4 days.
5. What features of this case pointed to the diagnosis of rubella infection?
6. Why is it significant that the symptoms started after a trip outside the United States?
7. What precaution could the man have taken to prevent this infection?
8. How was this infection transmitted?
9. Who was at risk for a serious outcome of this infection?
10. If this disease is normally mild in children, why is their immunization so important?
1. The diagnosis of dengue virus infection is indicated by the disease signs of high fever, severe headache, and joint and back pain. His trip to Malaysia would have increased his risk of exposure to Aedes mosquitoes carrying the virus.
2. The Aedes mosquito is endemic in Malaysia and is a carrier of dengue virus, which is prevalent in Malaysia.
3. The virus was transmitted independently by different mosquitoes to the father and son.
4. Petechiae and ecchymoses are indicators of hemorrhagic disease.
5. The diagnosis of rubella infection is suggested by the arthralgia and especially the mild rash. These immune-mediated responses occur after the virus replication and viremic spread, which induces interferon, causing the influenza-like syndrome.
6. Exposure to rubella in the United States is unlikely because of the effective vaccine program there.
7. If the man had been immunized with the measles-mumps-rubella vaccine and received his booster immunization at 15 years of age, he should have been protected against rubella disease.
8. Rubella is the only togavirus that is transmitted by aerosols as a respiratory virus.
9. All unimmunized individuals are at risk for this infection. However, the most serious outcomes occur to the fetus of women who are infected before the 20th week of pregnancy. Rubella causes severe congenital defects.
10. Immunization of the populace (especially children) for rubella prevents congenital defects in babies.
Chambers TJ, Monath TP. The flaviviruses: detection, diagnosis, and vaccine development, vol 61; The flaviviruses: pathogenesis and immunity, vol 60. In: Adv Virus Res. San Diego: Elsevier Academic; 2003.
Cohen J, Powderly WG. Infectious diseases, ed 2. St Louis: Mosby; 2004.
Fernandez-Garcia M-D, et al. Pathogenesis of flavivirus infections: using and abusing the host cell. Cell Host Microbe. 2009;5:318–328.
Flint SJ, et al. Principles of virology: molecular biology, pathogenesis and control of animal viruses, ed 3. Washington, DC: American Society for Microbiology Press; 2009.
Gelfand MS. West Nile virus infection. What you need to know about this emerging threat. Postgrad Med. 2003;114:31–38.
Gorbach SL, Bartlett JG, Blacklow NR. Infectious diseases, ed 3. Philadelphia: WB Saunders; 2004.
Gould EA, Solomon T. Pathogenic flaviviruses. Lancet. 2008;371:500–509.
Hahn CS, et al. Flavivirus genome organization, expression, and replication. Annu Rev Microbiol. 1990;44:663–688.
Johnson RT. Viral infections of the nervous system. Philadelphia: Lippincott-Raven; 1998.
Knipe DM, et al. Fields virology, ed 5. Philadelphia: Lippincott Williams & Wilkins; 2006.
Koblet H. The “merry-go-round”: alphaviruses between vertebrate and invertebrate cells. Adv Virus Res. 1990;38:343–403.
Kuhn RJ, et al. Structure of dengue virus: implications for flavivirus organization, maturation, and fusion. Cell. 2002;108:717–725.
Mackenzie JS, Barrett ADT, Deubel V. Japanese encephalitis and West Nile viruses. In: Curr Top Microbiol Immunol. Berlin: Springer-Verlag; 2002.
Monath TP. Yellow fever vaccine. Plotkin SA, Orenstein WA. Vaccines, ed 4, Philadelphia: WB Saunders, 2004.
Mukhopadhyay S, et al. Structure of West Nile virus. Science. 2003;302:248.
Nash D, et al. The outbreak of West Nile virus infection in the New York City area in 1999. N Engl J Med. 2001;344:1807–1814.
Plotkin SA, Reef S. Rubella vaccine. Plotkin SA, Orenstein WA. Vaccines, ed 4, Philadelphia: WB Saunders, 2004.
Richman DD, Whitley RJ, Hayden FG. Clinical virology, ed 3. Washington, DC: American Society for Microbiology Press; 2009.
Strauss JM, Strauss EG. Viruses and human disease, ed 2. San Diego: Academic; 2007.
Tsai TF. Arboviral infections in the United States. Infect Dis Clin North Am. 1991;5:73–102.
Centers for Disease Control and Prevention. Arbovirus encephalitides. www.cdc.gov/ncidod/dvbid/arbor/index.htm. Accessed June 1, 2012
Centers for Disease Control and Prevention. Dengue. www.cdc.gov/dengue/. Accessed June 1, 2012
Centers for Disease Control and Prevention. West Nile virus. www.cdc.gov/ncidod/dvbid/westnile/index.htm. Accessed June 1, 2012
Dengue map. www.healthmap.org/dengue/index.php. Accessed June 1, 2012