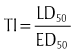Adverse Reactions
1 Define an adverse drug reaction and name five categories of reaction.
2 Discuss the risk-to-benefit ratio of the use of a drug for therapeutic effect and its potential adverse reactions.
Although drugs may act on biologic systems to accomplish a desired effect, they lack absolute specificity in that they can act on many different organs or tissues. This lack of specificity is the reason for undesirable or adverse drug reactions. No drug is free from producing some adverse effects in a certain number of patients. It is estimated that between 5% and 10% of the patients hospitalized annually in the United States are admitted because of adverse reactions to drugs. While hospitalized, 10% to 20% of patients experience adverse reactions caused by drugs.
The dental health care worker is in a good position to observe any adverse reactions or undesirable effects caused by drugs administered in the dental office. Adverse reactions to drugs prescribed by the patient’s physician can be identified in the health history. The dental health care worker should question the patient about any potential oral manifestations of drugs. For example, if the patient is taking phenytoin (Dilantin), questions about enlargement of the patient’s gums should be explored. Because many drugs can produce xerostomia, complaints of dry mouth should direct the dental health care worker to examine the patient’s medications. Knowledge of the typical adverse drug reactions can help dental office personnel identify, minimize, or prevent these types of reactions. Because of the rapport between a patient and the dental health care worker, the patient will often reveal important facts about the health history or ask questions concerning medications prescribed. The dental health care worker must know the terms used to describe adverse reactions to discuss a drug’s undesirable effect accurately with other health professionals. For example, allergy refers to a specific type of reaction to a drug but does not include a complaint of excessive gas, or flatulence.
DEFINITIONS AND CLASSIFICATIONS
Unfortunately, every drug has more than one action. The clinically desirable actions are termed therapeutic effects, and the undesirable reactions are termed adverse effects. Dividing a drug’s effects into two categories is artificial because it depends on the indication for which the drug is being used. For example, when an antihistamine used to relieve hay fever causes drowsiness, the drowsiness can be considered an adverse effect. However, if the antihistamine were being used to induce sleep (over-the-counter [OTC] sleep aid), drowsiness would be considered the therapeutic effect.
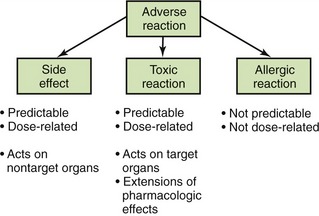
An adverse drug reaction is a response to a drug that is not desired, is potentially harmful, and occurs at usual therapeutic doses. It may be an exaggeration of the desired response, an expected but undesired response, an allergic reaction, a cytotoxic reaction, or an effect on the fetus. Often, adverse drug reactions are divided into the following categories:
• Toxic reaction: A toxic reaction is an extension of the pharmacologic effect resulting from a drug’s effect on the target organs. In this instance, the amount of the desired effect is excessive.
• Side effect: A side effect is a dose-related reaction that is not part of the desired therapeutic outcome. It occurs when a drug acts on nontarget organs to produce undesirable effects. The terms side effect and adverse reaction often are used interchangeably. The upset stomach produced by ibuprofen is an adverse reaction when ibuprofen is given to manage pain.
• Idiosyncratic reaction: An idiosyncratic reaction is a genetically related abnormal drug response. Certain populations, because of their genetic constitution, are more susceptible to certain adverse reactions to specific drugs. Eskimos metabolize certain drugs faster than other populations; therefore a larger dose of those drugs would be needed in that population (e.g., isoniazid).
• Drug allergy: A drug allergy is an immunologic response to a drug resulting in a reaction such as a rash or anaphylaxis. This response accounts for less than 5% of all adverse reactions. Unlike other adverse reactions, allergic reactions are neither predictable nor dose related.
• Interference with natural defense mechanisms: Certain drugs, such as adrenocorticosteroids, can reduce the body’s ability to fight infection. Drugs that interfere with the body’s defenses cause a patient to get infections more easily and have more trouble fighting them.
The importance of distinguishing between different types of adverse effects can be seen using aspirin as an example. Aspirin can produce adverse reactions such as gastric upset or pain. At higher doses, aspirin can predictably produce toxicity such as tinnitus and hyperthermia (elevated temperature). Another type of reaction to aspirin is allergic, often involving a rash or difficulty in breathing (asthma-like reaction). These differences are significant and become pertinent when discussing an adverse reaction with another health professional. Patients who experience allergic reactions to a medication should not receive that medication or similar medications. Side effects such as gastrointestinal upset, although bothersome, are not reasons to avoid prescribing a medication. It can be given. However, if the gastrointestinal upset is too much for the patient, another drug should be considered. It is important to describe in the patient’s chart the patient’s “problem” in enough detail so that side effects can be separated from allergic reactions. Figure 3-1 describes the types of adverse reactions and notes whether they are predictable or dose dependent.
CLINICAL MANIFESTATIONS OF ADVERSE REACTIONS
Before a drug is used, there must be an assessment of its risk against its benefits (risk-to-benefit ratio). This means that the beneficial effect of the drug must be weighed against its potential for adverse reactions. For example, one would compare the drug’s therapeutic effect (e.g., controlling seizures) with its potential to cause an adverse reaction (e.g., birth defects). In a real-life example, one should compare the therapeutic effect of certain drugs to produce weight loss to their potential for the serious adverse reactions of primary pulmonary hypertension (which is fatal in 50% of patients) or cardiac valvular damage.
Exaggerated Effect on Target Tissues
An exaggerated effect on its target tissue or organ is considered an extension of the therapeutic effect caused by the overreaction of a sensitive patient or by the use of a dose that is too large. For example, a patient may experience exaggerated hypoglycemia when given a therapeutic dose of an oral hypoglycemic agent for the treatment of diabetes. The patient’s blood sugar may fall too low, either because of an unusual sensitivity to the drug or because the dose administered was too high for that patient. Occasionally, this type of adverse reaction may result from liver or kidney disease. Because the disease interferes with the drug’s metabolism or excretion, the drug’s action may be enhanced or prolonged.
Effect on Nontarget Tissues
The effect on nontarget organs or tissues is caused by the nontherapeutic action of the drug. These reactions can occur at usual doses, but they appear more often at higher doses. For example, aspirin may produce gastric upset in usual therapeutic doses, but with higher doses salicylism, characterized by tinnitus, disturbances in the acid-base balance, and confusion, can result. Toxic reactions can affect many parts of the body. A reduction in the dose of a drug usually reduces these adverse reactions.
Effect on Fetal Development (Teratogenic Effect)
The word teratogenic comes from the Greek prefix terato-, meaning “monster,” and the suffix -genic, meaning “producing,” or “producing a malformed fetus.” The relationship between drugs and congenital abnormalities has been recognized since the middle of the twentieth century. In 1961, thalidomide, an OTC drug marketed in Europe, was found to produce phocomelia (short arms and legs) in the exposed fetus. In some cases, only one dose of this drug produced the effect. This incident reinforced the fact that more studies were needed to determine the effect of drugs on pregnant women. For new drugs, there are many more studies on animals and their reproductive capacity before the drugs are placed on the market. Although more information is now available about the safety of drugs in pregnant women, sufficient information is still lacking.
The Food and Drug Administration (FDA) has attempted to address the concerns about the lack of adequate knowledge of drugs by defining five FDA pregnancy categories: A, B, C, D, and X, ranked from least risky to most risky. (See Table 25-2 for a discussion of the meaning of these categories.) They are similar to school grades: A is the “best” or least teratogenic and X is equivalent to F or not ever to be used if someone might get pregnant. Some older drugs may be classified as C because there are insufficient data to place them in higher categories.
Although no drug can be considered “completely safe” for administration to a pregnant woman, many of the drugs used in dentistry are considered to be among the safest. These include the antibiotics penicillin and erythromycin, the pain medication acetaminophen (Tylenol), and the local anesthetic lidocaine. Even these drugs should be administered only if there is a clear need. Elective dental procedures should be conservatively addressed. Drugs that are used in dentistry that are contraindicated during pregnancy include tetracycline, nonsteroidal antiinflammatory agents, the benzodiazepines, and metronidazole. The teratogenic potential for dental drugs is discussed in Chapter 25.
Many drug manufacturers, especially those that produce some of the older drugs, continue to place in their package inserts such statements as the following:
• “There are no adequate and well-controlled studies in pregnant women.”
• “Risks must be balanced against the uncontrolled disease.”
• “Safety during pregnancy has not been established; use requires that potential benefits be weighed against its possible hazard to the mother and child.”
• “Animal reproductive studies are not always indicative of human effects.”
• “Animal reproduction studies have not been conducted with …”
• “… potential benefit must be weighed against the possible hazards to the mother and fetus.”
Encouragement of increased information, especially for newer agents, has resulted in more information about use during pregnancy such as the following statements:
• “Shown to decrease fetal birth weight when given at 50 times the dose recommended for humans.”
• “No evidence of birth abnormalities or impaired fertility in doses two times the usual human dose.”
However, there are still plenty of statements such as the following:
With regard to teratogenesis, it is important to remember that the risk to the fetus must always be considered when considering the benefit to the mother.
In addition, dental patients do not always announce that they are pregnant, so it is important to ask any woman of child-bearing age (between approximately 11 and 63 years of age [a 63-year-old French woman gave birth in 1997]) if she is pregnant. Problem drugs should be avoided as early as possible during the pregnancy. The greatest risk from exposure to drugs occurs before the pregnancy status is known.
Local Effect
Local reactions are characterized by local tissue irritation. Occasionally, injectable drugs can produce irritation, pain, and tissue necrosis at the site of injection. Topically applied agents can produce irritation at the site of application. Drugs taken orally can produce gastrointestinal symptoms, such as nausea or dyspepsia, because of their local actions on the gastrointestinal tract.
Drug Interactions
A drug interaction can occur when the effect of one drug is altered by another drug. These interactions may result in undesirable effects such as toxicity or lack of efficacy. Drug interactions can also produce beneficial effects. Whenever prescribing or suggesting a drug to a dental patient, the chance of drug interactions must be considered. The likelihood that a drug interaction would occur increases with the number of drugs that a patient is taking. Drug-food and drug-disease interactions may also occur.
Hypersensitivity (Allergic Reaction)
Hypersensitivity reactions occur when the immune system of an individual responds to the drug administered or applied. One example of an allergic (hypersensitivity) reaction is when a patient is given a drug and develops hives. For a drug to produce an allergic reaction, it must act as an antigen and react with an antibody in a previously sensitized patient. This reaction is neither dose dependent nor predictable. For an allergic reaction to occur, an ingested drug may be metabolized to a reactive metabolite known as a hapten. This hapten can act as an antigen after combining with proteins in the body. The antigen formed then stimulates the production of an antibody. With subsequent exposure to the drug, the antibodies formed will react with the antigen (drug or metabolite) administered and elicit an antigen-antibody reaction. This reaction triggers a series of biochemical and physiologic events that can be life threatening.
Drug allergy can be divided into the following four types of reactions, depending on the type of antibody produced or the cell mediating the reaction (Table 3-1):
TABLE 3-1
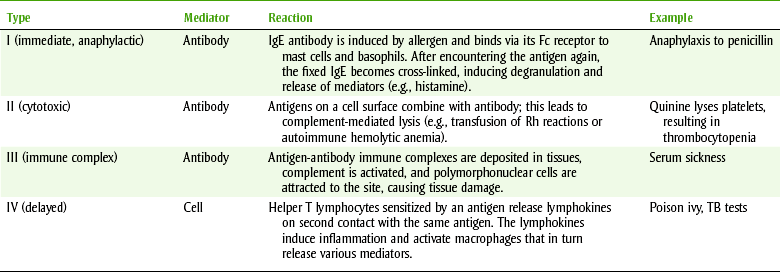
Ig, Immunoglobulin; TB, tuberculosis.
From Levinson WE: Review of medical microbiology and immunology, ed 10, New York, 2008, McGraw-Hill Medical.
• Type I reactions are mediated by immunoglobulin E (IgE) antibodies. When a drug antigen binds to IgE antibody, histamine, leukotrienes, and prostaglandins are released, producing vasodilation, edema, and the inflammatory response. The targets of this reaction are the bronchioles, resulting in anaphylactic shock; the respiratory system, resulting in rhinitis and asthma; and the skin, resulting in urticaria and dermatitis. Because these reactions can occur relatively quickly after drug exposure, they are known as immediate hypersensitivity reactions. Anaphylaxis is an acute, life-threatening allergic reaction characterized by hypotension, bronchospasm, laryngeal edema, and cardiac arrhythmias that can occur within a few minutes after drug administration. Drugs used in dentistry that have produced fatal anaphylaxis include the penicillins, ester local anesthetics, and aspirin. Unexpected anaphylaxis may occur such as after a patient has been given a dose of penicillin by injection. Oral penicillin can also produce anaphylaxis, but it is much less common.
• Type II, or cytotoxic/cytolytic, reactions are complement-dependent reactions involving either immunoglobulin G (IgG) or immunoglobulin M (IgM) antibodies. The antigen-antibody complex is fixed to a circulating blood cell, resulting in lysis. Examples of this reaction are penicillin-induced hemolytic anemia and methyldopa-induced autoimmune hemolytic anemia.
• Type III, or Arthus, reactions are mediated by IgG. In these reactions, the drug antigen-antibody complex fixes complement and deposits in the vascular endothelium. The reaction is manifested as serum sickness and includes urticarial skin eruptions, arthralgia, arthritis, lymphadenopathy, and fever. This reaction can be caused by the penicillins and sulfonamides.
• Type IV, or delayed hypersensitivity, reactions are mediated by sensitized T lymphocytes and macrophages. When the cells contact the antigen, an inflammatory reaction is produced by lymphokines, neutrophils, and macrophages. An example of a type IV reaction is allergic contact dermatitis caused by topical application of drugs. Topical benzocaine, penicillin, poison oak, and poison ivy can produce this type of reaction. Reaction to “cheap” jewelry is another example.
Idiosyncrasy
An idiosyncratic reaction is a reaction that is neither the drug’s side effect nor an allergic reaction. Some idiosyncrasies have been found to be genetically determined abnormal reactions, whereas others may be the result of an immunologic mechanism. About 10% of black males can develop severe hemolytic anemia when given the antimalarial drug primaquine. This is because of a deficiency in an enzyme, glucose-6-phosphate dehydrogenase.
Interference with Natural Defense Mechanisms
A drug’s effect on the body’s defense mechanisms can result in an adverse reaction. Long-term systemic administration of corticosteroids can result in decreased resistance to infection. Because periodontal disease involves both infection and an immune response, drugs that are immunosuppressive could exacerbate a patient’s poor oral health.
TOXICOLOGIC EVALUATION OF DRUGS
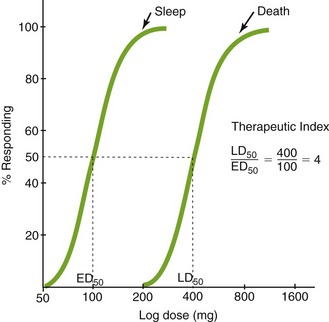
Optimally, evaluations of the toxic effects of drugs are based on experiments that are performed with lower animals and clinical trials conducted in humans. Animal experiments can often elicit adverse reactions that could occur in humans, but unfortunately, drug reactions in animals do not always predict reactions in humans. The lethal dose (LD50), one measure of the toxicity of a drug, is the dose of a drug that kills 50% of the experimental animals. The median effective dose (ED50) is the dose required to produce a specified intensity of effect in 50% of the animals. Figure 3-2 shows a plot of the dose of a drug against the percentage of maximum response (sleep or death) in animals. A dose-response curve is then obtained. The value on the dose axis that corresponds to the 50% intensity level on the response axis can be read directly from the curve. This figure illustrates the ED50 and LD50.
Because all drugs are toxic at some dose, the LD50 is meaningless unless the ED50 is also known. The ratio LD50/ED50 is the therapeutic index (TI) of a drug:
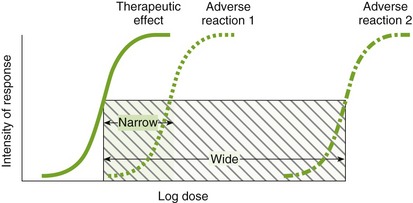
If the value of the TI is small (narrow TI), then toxicity is more likely. If the TI is large (wide TI), then the drug will be safer (Figure 3-3). A drug with a wide TI will have a large LD50 and a small ED50 (the distance between these curves is large). A TI of greater than 10 is usually needed to produce a therapeutically useful drug. The TI derived from animal studies determines both the LD50 and the ED50 from a variety of animals.
CLINICAL SKILLS ASSESSMENT
1. Name four classifications of adverse drug reactions.
2. Describe the problem with identifying teratogenic agents (see also Chapter 25).
3. Describe the types of adverse reactions.
4. Explain the four mechanisms by which an allergic reaction can occur.
5. Describe why the risk-to-benefit ratio is important and helpful in deciding whether to administer a drug to a patient.
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.