7 Acute kidney injury and chronic kidney disease
By the end of the chapter you should be able to:
• Classify the causes of acute kidney injury (AKI)
• Differentiate prerenal and intrinsic renal AKI
• Describe the common causes of chronic kidney disease (CKD)
• Predict the complications of CKD
• Understand the dietary management of CKD
• Give five indications for renal replacement therapy
• Explain how haemodialysis works
• Give the benefits and disadvantages of peritoneal dialysis compared to haemodialysis
Terminology
Acute kidney injury (AKI) and chronic kidney disease (CKD) are now the preferred terms for what often used to be described as acute or chronic renal failure, respectively. Renal failure implies reduced excretory function of the kidney whereas CKD can be present even when excretory function is normal, as long as there is other evidence of kidney damage (for example blood or protein in the urine or structural abnormality).
Acute kidney injury
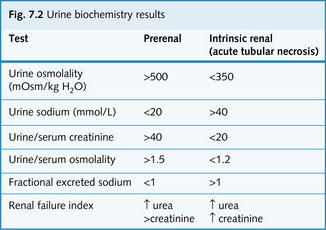
Acute kidney injury is the deterioration of renal function occurring over hours or days. Urea and creatinine rise rapidly. It is usually (but not always) associated with oliguria and is usually (but not always) reversible. The dangerous consequences are volume overload, metabolic acidosis and hyperkalaemia. The causes are classified as shown Fig. 7.1.
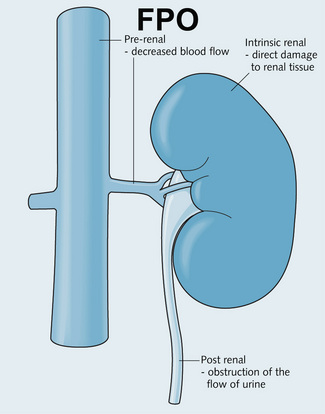
Fig. 7.1 Causes of acute kidney injury. AKI can be caused by decreased blood flow (prerenal), direct damage to the renal tissue (intrinsic renal) or obstruction of the flow of urine (post-renal).
• Reduced effective circulating volume – hypovolaemia (e.g. haemorrhage, dehydration, burns) cardiac failure or liver failure
• Shock (may also lead on to acute tubular necrosis)
• Renal artery stenosis or emboli
• NSAIDs and ACE inhibitors impair the mechanisms of renal autoregulation so can predispose to prerenal AKI.
Intrinsic renal causes of AKI:
• Acute tubular necrosis: ischaemia (can be caused by any prerenal cause), drug toxicity (e.g. gentamicin, aciclovir, methotrexate) and toxins (e.g. myoglobinuria and lipopolysaccharide in Gram-negative sepsis)
• Acute interstitial nephritis (due to drugs, infections, hypercalcaemia, multiple myeloma)
• Glomerular disease – acute glomerulonephritis, rapidly progressive glomerulonephritis
• Vascular disease – vasculitis, malignant hypertension, thrombotic microangiopathies.
• Bladder outflow obstruction (benign prostatic hypertrophy or urethral strictures)
• Tumour (prostate, bladder or gynaecological malignancy)
Note: Obstruction must occur in both kidneys or in a single functioning kidney for renal failure to occur.
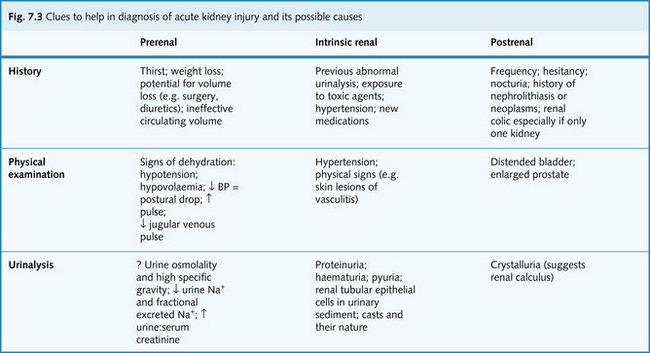
Diagnostic approach to AKI
• Is the renal failure acute or chronic? A history of chronic ill health or signs of CKD such as anaemia may indicate chronicity, as does small kidneys on ultrasound
• Is there urinary tract obstruction? Obstruction should always be considered as a cause because it is reversible and prompt treatment may prevent permanent renal damage
• Is there a rare cause of acute kidney injury? For example, myeloma, systemic vasculitis, haemolytic uraemic syndrome because prompt treatment can be lifesaving.
Basic investigation of acute kidney injury includes urine tests (dipstick, microscopy, culture, cytology), blood tests and renal imaging (KUB (X-ray of the kidney, ureter and bladder), a chest X-ray and an ultrasound of the renal tract). In AKI, the following biochemical changes occur:
• Increased plasma urea and creatinine concentrations
• Increased plasma concentrations of potassium
• Metabolic acidosis and an increased anion gap
• Increased plasma phosphate and decreased plasma calcium (less marked than in CKD)
• Decreased plasma sodium can occur
• Changes in urine biochemistry: these depend on whether prerenal or renal failure exists (Fig. 7.2).
In addition, some serological tests can help clarify the diagnosis:
• Antinuclear antibodies (ANA): SLE-associated nephritis
• Cryoglobulin titre: cryoglobulinaemia
• Complement levels: SLE-associated nephritis, membranoproliferative and acute glomerulonephritis
• Antineutrophil cytoplasmic antibodies (ANCA): vasculitis (Wegener's granulomatosis or polyangiitis).
Fig. 7.3 gives some clues to help in the diagnosis of acute kidney injury and its possible causes.
Management
Management depends on the precipitating cause.
Appropriate fluid replacement therapy is always important to optimize blood flow to the kidneys. Hypovolaemia is a common cause of prerenal failure and will lead to acute tubular necrosis if left uncorrected. Hypovolaemia should be rapidly corrected with intravenous fluids. Clinical signs of dehydration include, decreased skin turgor, a low JVP, a low blood pressure, weight loss. Invasive monitoring of the central venous pressure may be necessary to confirm hypovolaemia.
Obstruction of bladder outflow is relieved with a urinary or suprapubic catheter. If the ureters are obstructed, a drainage tube (nephrostomy) is inserted above the obstruction.
Chronic kidney disease
CKD can result in progressive loss of renal function over months to years. It is usually irreversible because the renal tissue has been replaced by extracellular matrix (scar tissue) in response to damage. However, the deterioration can be slowed with treatment. It can result from renal disease or be secondary to systemic diseases.
Progression of CKD is best monitored by plotting the calculated or estimated glomerular filtration rate (see Chapter 2) against time (Fig. 7.4). At first there may be no symptoms, only biochemical abnormalities but in later stages symptoms develop from the loss of excretory, endocrine and metabolic functions. It is likely that renal replacement therapy will be needed by stage 5. The rate of death from cardiovascular disease is greatly increased in CKD especially stages 4 and 5.
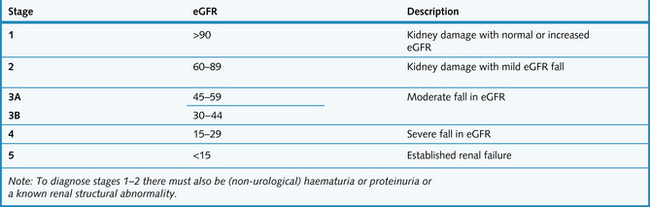
Fig. 7.4 Classification of chronic kidney disease according to estimated glomerular filtration rate (eGFR).
Screening of people at risk
NICE have recommended that people in the following at risk groups should be offered testing for CKD:
• Cardiovascular disease (ischaemic heart disease, chronic heart failure, peripheral vascular disease and cerebral vascular disease)
• Structural renal tract disease, renal calculi or prostatic hypertrophy
• Multisystem diseases with potential kidney involvement, e.g. systemic lupus erythematosus (SLE)
• Family history of stage 5 CKD or hereditary kidney disease
Annual follow-up testing is recommended in all patients with CKD or at risk of CKD, with more frequent testing being required according to clinical circumstance or during an intercurrent illness.
Causes
• Intrinsic renal: glomerulonephritis, chronic pyelonephritis, polycystic kidneys, bladder or urethral obstruction, interstitial nephritis, amyloid, myeloma, renal vascular disease, Alport's syndrome
• Systemic (extrarenal): diabetes mellitus, hypertension (especially if accelerated-malignant), heart failure, SLE, gout, hypercalcaemia, renovascular disease (atheroma), vasculitis
The commonest causes of CKD are diabetes, hypertension, reflux nephropathy and polycystic kidney disease.
The diagnostic approach to CKD can involve:
• Urine: urinalysis (haematuria, proteinuria), microscopy (white cells, eosinophilia, granular casts, red cell casts, red cells), biochemistry (protein or albumin to creatinine ration, urinary electrolytes, osmolality, protein electrophoresis)
• Blood: urea and creatinine and eGFR, electrolytes, glucose, calcium (decreased), phosphate (increased), urate (increased), protein, osmolality, full blood count, markers of inflammation (C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR)), protein electrophoresis, autoantibody screen, complement components
• Radiology: ultrasound (to assess the kidney size, identify obstruction, mass lesions or polycystic kidney disease). Plain radiography or CT of the abdomen (calculi) and chest X-ray (pulmonary oedema). Hand radiographs can show evidence of osteodystrophy in advanced cases but are now rarely performed
• Renal biopsy: consider if kidneys are of normal size and the cause of CKD is not clear from other investigations.
Preventing progression of CKD
Damage which has resulted in CKD cannot be reversed but it is important to effectively treat any cause to prevent further damage, for example relieving any obstruction of urine flow or stopping any nephrotoxic drugs. Hypertension must be effectively treated and this may require several antihypertensives. Diabetics need tight control of their blood glucose and benefit from ACE inhibitors or ARBs, as do patients with proteinuria. The presence of proteinuria is a poor prognostic sign.
Protein restriction to prevent progression of CKD is now rarely recommended as it is likely to cause malnutrition.
Complications and management
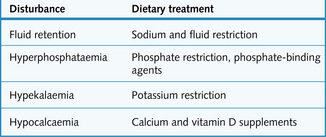
Fluid retention
This can cause pulmonary oedema or peripheral oedema. Treatment is with diuretics which are less effective in renal failure so bigger doses may be needed.
Hypertension
This can be caused by CKD and is an important cause of progression in CKD. It must therefore be carefully treated. Drugs which interfere with the renin–angiotensin system may be particularly effective if they can be tolerated. The targets for treatment are shown in Fig. 7.5.
Increased cardiovascular risk
This is another reason to tightly control blood pressure. Other cardiovascular risk factors must also be addressed and many patients will benefit from lifestyle advice, aspirin and a statin.
Osteodystrophy
In advanced CKD failure to activate vitamin D leads to hypocalcaemia and impaired mineralization of bone. Also the kidneys do not excrete enough phosphate. This stimulates PTH release which increases bone reabsorption. Bone pains are experienced in the lower back and legs. Treatment is with vitamin D analogues, calcium supplements, reducing phosphate intake in the diet and reducing phosphate absorption with phosphate-binding agents.
Anaemia
In advanced CKD there is reduced erythropoietin production by the kidney. In addition retained toxins reduced red blood cell survival and impair bone marrow responsiveness to erythropoietin. This leads to a normocytic normochromic anaemia. Symptoms are lethargy and dyspnoea. Anaemia of CKD can be effectively reversed by (expensive) erythropoietin injections, but it is essential to ensure iron stores are adequate prior to starting treatment.
Electrolyte disturbances
Some electrolyte imbalances can be managed with dietary changes (Fig. 7.6).
Acidosis
Metabolic acidosis can be treated with sodium bicarbonate or calcium carbonate supplements.
Uraemia
Inability to excrete nitrogenous waste products can lead to uraemic symptoms. This may include anorexia, nausea and vomiting. Itching is also a common complaint (this is also caused by hyperphosphataemia). When advanced uraemia can cause neuromuscular symptoms (restless legs and fitting). The only effective treatment for uraemic symptoms is renal replacement therapy.
Renal replacement therapy
In end-stage kidney disease it is necessary to replace the excretory function of the kidneys with renal replacement therapy (RRT). The four forms of RRT are: haemodialysis, haemofiltration, peritoneal dialysis and renal transplantation (Fig. 7.7). These remove the waste products and excess fluids that accumulate in renal disease, but only renal transplantation can replace all the kidney's functions.
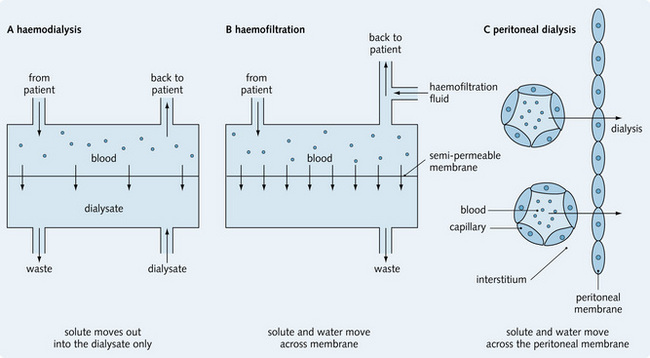
Fig. 7.7 Comparison of the three different methods of dialysis. (A) haemodialysis; (B) haemofiltration and (C) peritoneal dialysis.
(Adapted from O'Callaghan CA, Bremmer BM, 2001. The kidney at a glance. Blackwell Science, pp. 96, 98.)
In CKD the indications for RRT are not clear cut and the onset should be planned in accordance with the patient's symptoms and eGFR. The indications for RRT in AKI include:
Haemodialysis
This involves using a dialysis machine to pump blood through an artificial kidney. Blood flows on one side of a semipermeable membrane, with dialysis fluid being passed in the opposite direction on the other side. Dialysis occurs across the semipermeable membrane removing toxins from the blood down a concentration gradient.
The dialysate is made of purified water with a solute composition similar to plasma, but without any of the waste products, so solutes move along their concentration gradient out of the blood. Smaller solutes diffuse across the semipermeable membrane faster than larger solutes. Dialysis ‘dose’ can be adjusted by altering the blood flow, the area of the semipermeable membrane, or the duration of treatment. On average, patients require at least 4 hours of treatment three times a week. Coagulation within the dialysis machine is prevented with heparin. Treatment may be performed in a hospital environment supervised by a nursing team or at home by the patient themselves after a period of training.
Access to the circulation is gained by an arteriovenous (AV) fistula, which is constructed surgically, usually by joining the radial artery and cephalic vein. Over a few weeks the venous system ‘arterializes’ and the high blood flow required for dialysis can be obtained by ‘needling’ the venous system. Complications of the AV fistula include infection and thrombosis. A fistula should be prepared well in advance of the onset of RRT. If a fistula is not possible then quick access to the circulation can be gained through a double lumen central venous catheter.
Haemofiltration
This involves filtering blood at high pressure across a semipermeable membrane allowing removal of small molecules. The fluid lost across the membrane is discarded and replacement with an appropriate biochemical composition is added back to the blood. This replacement fluid is commonly buffered by lactate.
Haemofiltration is used predominantly for the treatment of AKI, especially in the intensive care unit setting. When performed slowly (continuous veno–veno haemofiltration), it causes smaller fluid shifts and therefore less hypotension than haemodialysis. Access is obtained through a double lumen central venous catheter. Haemodiafiltration is a combination of dialysis and filtration through the same machine.
Peritoneal dialysis
This is usually undertaken using continuous ambulatory peritoneal dialysis (CAPD). CAPD uses the peritoneal membrane as the semipermeable membrane. Unlike haemodialysis, peritoneal dialysis does not require an AV fistula for circulatory access. Instead, it requires the insertion of a permanent ‘Tenchkoff’ catheter through the anterior abdominal wall into the peritoneal cavity. Dialysate solution is introduced into the peritoneum and exchanged regularly for fresh fluid – up to four or five times a day is necessary to maintain the efficiency of dialysis. Waste products pass into the dialysate along their concentration gradients and water is removed by osmosis. Dialysis solutions with high osmolarity will remove more water. Dextrose is the most commonly used osmotic agent, but is gradually absorbed by the patient. Newer, non-absorbable, osmotic agents are now available (e.g. glucose polymer). CAPD is used in the maintenance dialysis of end-stage renal failure, but technique survival declines to 50% after 5 years due to loss of peritoneal membrane function. The treatment is performed by the patient in the community. A variant of the technique is continuous cycling peritoneal dialysis (CCPD) or automated peritoneal dialysis (APD) in which a machine cycles dialysis fluid in and out of the peritoneal cavity, usually at night whilst the patient is asleep.
Complications of peritoneal dialysis include:
• Peritonitis (50% is caused by Staphylococcus epidermidis). Treatment is with intraperitoneal or intravenous antibiotics
• Mechanical problems with fluid drainage
• Infections or blockage around the site of the catheter
• Other complications include pleural effusions and sclerosing peritonitis (rare but serious).
Renal transplantation
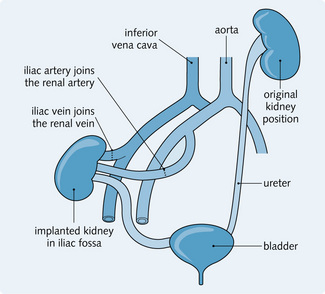
This is the ideal treatment for irreversible stage 5 CKD. It restores near-normal renal function and improves quality of life. The kidney may come from a cadaver, a close living relative or a partner and is usually placed in the iliac fossa. The renal vessels from the donated kidney are anastomosed onto the iliac blood vessels of the recipient and the ureter is inserted into the bladder (Fig. 7.8). Success depends upon:
Short-term complications include:
The risk of rejection is reduced by immunosuppression therapy, which is started at the time of the transplant and continued indefinitely. The patients are at risk of opportunistic infection (e.g. with cytomegalovirus).
Long-term complications include:
Currently, the 1-year graft survival rate is in excess of 80% for cadaveric transplants and 90% for live donor transplants.
Pharmacokinetics in renal disease
There are many factors to think about when prescribing drugs for a patient with kidney disease. Nephrotoxic drugs need to be avoided and the pharmacokinetics of other drugs can be changed.
Absorption
Fluid retention in kidney disease with oedema of the bowel wall can lead to reduced absorption of drugs given orally.
Distribution
The protein binding of drugs may be affected in proteinuric renal disease because of hypoalbuminaemia. Also, with uraemia other retained substances compete for binding sites of the drug. This will increase the concentration of the drug in the blood. Fluid retention in kidney disease can increase the volume of distribution.
Metabolism
The kidney metabolizes some drugs, e.g. insulin. If this function is impaired the half life of the drug will increase. Uraemia can also alter drug metabolism by the liver.
Elimination
Excretion of drugs by the kidney is a major route of elimination. If this function of the kidney is impaired the half life of the drug will be increased and the plasma concentration will rise with each dose of the drug. Renal elimination is particularly important with water-soluble drugs that are minimally metabolized by the liver.