8 Clinical assessment of the renal system
By the end of the chapter you should be able to:
• List the symptoms typically relevant to the urinary system
• Explain why family history is sometimes important in kidney disease
• Find relevant signs in a patient's hands
• Describe how to measure the jugular venous pulse
• Differentiate an enlarged kidney from splenomegaly
• Describe the test for ascites and the renal causes of it
• Name five tests that can be performed on a urine sample, and explain the importance of a mid-stream urine sample
• List the causes of haematuria and proteinuria and explain how they might be investigated
• List the blood tests that can be done to investigate renal function, and give the possible causes of abnormal results
• Explain when you would use ultrasonography in preference to plain radiography
• List the contraindications for renal biopsy, and four potential complications
• Describe the complications of using intravenous contrast medium
• Explain the main differences between a diagnostic and a therapeutic cystoscopy
• Explain how urodynamics studies can be used to differentiate between genuine stress incontinence and urge incontinence
History
This section is a guide to taking a history for the renal and urinary systems. Not all will be relevant to each patient and you may need to inquire about other systems to investigate a differential diagnosis.
History of presenting complaint
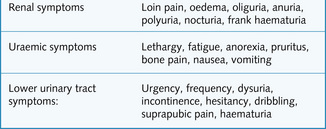
Fig. 8.1 shows typical symptoms of diseases of the urinary system. As always, enquire deeper about the presenting complaint. Use open questions and non-verbal prompting where possible. Use the mnemonics SQITARS (Site, Quality, Intensity, Timing, Aggravating factors, Relieving factors, and associated Symptoms) or SOCRATES (Site, Onset, Character, Radiation, Associated symptoms, Timing, Exacerbating and alleviating factors, Severity) to help you to gather as much information as possible.
Past medical history
Find out any past or current medical illnesses, operations or trauma. Past history of any renal or urological disease is obviously important. Diabetes and hypertension are risk factors for CKD. Systemic diseases such as vasculitis can cause glomerulonephritis. CKD is related to a higher risk of cardiovascular disease. A recent streptococcal throat infection can trigger post-streptococcal glomerulonephritis.
Drug history
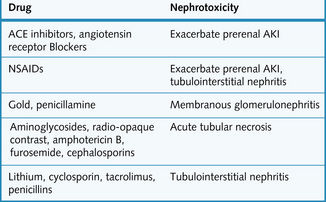
Find out all the medications that the patient takes. Also ask about over-the-counter medicine and homeopathic medicine. Many medications can affect renal function (Fig. 8.2). Also remember that the pharmacokinetics of drugs can be altered in renal disease.
Family history
Polycystic kidney disease, Alport's syndrome and Fabry's syndrome are inherited renal diseases. Many other diseases will have some genetic component. A family history of CKD stage 5 increases the risk of an individual having CKD. Some families have a tendency for IgA nephropathy. Also ask about family history of diabetes and hypertension. Ethnicity is important in the incidence of some diseases, e.g. SLE and diabetic nephropathy in South Asian populations.
Social history
A patient's social situation can be very important in deciding management. Ask about their marital status, social support network, hobbies and how they are coping at work. Other factors in the social history:
• Occupation. Has the patient been exposed to any toxins? Workers in rubber and dye factories can be exposed to aromatic amines that can cause bladder cancer
• Smoking is a risk factor for bladder cancer and renal vascular disease
• Alcohol can compromise kidney function
• Dietary habits can be relevant in urolithiasis (calcium, oxalate and fluid intake) and CKD (protein, fluid, sodium, phosphate, iron intake)
• Sexual history may be relevant if there is a possibility of STI.
Examination
General inspection
Whenever you examine a patient it is important to remember the following points:
• Introduce yourself to the patient and ask permission to examine them
• Position the patient appropriately for the examination – at 45° for cardiovascular or respiratory system examination or lying flat for abdominal examination
• Expose the appropriate part of the body only and ensure the patient is comfortable
• Observe the patient carefully from the end of the bed before you begin the examination
• Look around the patient for any extra clues – dialysis equipment, sputum pot or oxygen mask
• Notice if the patient is conscious or looks well, is in pain, appears anxious or depressed, smells of urine or has any obvious skeletal abnormalities
• Look at the patient's stature (CKD in childhood causes growth retardation)
• Weighing the patient regularly can be helpful if they are retaining fluid.
Hands
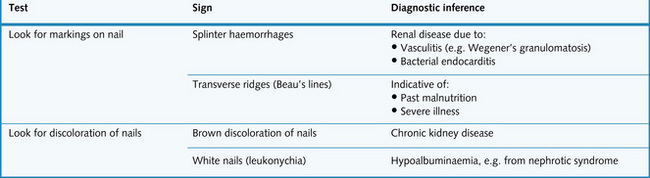
Abnormalities of the nails that indicate underlying renal disease are summarized in Fig. 8.3. To examine the hands, spread out the fingers on a flat, white surface. This will highlight any shortening of the distal phalanges; a difference of length between the fingers is often seen in severe renal osteodystrophy secondary to chronic renal failure. This is due to chronic high circulating levels of parathyroid hormone. With the patient holding their hands straight out, look for a course flapping movement called asterexis. Whilst this could be caused by severe uraemia it cannot be distinguished from asterexis caused by CO2 retention or liver failure.
Pinch the skin on the back of the hand to determine its elasticity or turgor. Reduced skin turgor can be caused by old age but can also indicate dehydration.
Arms
Look in the arms for an arteriovenous fistula, used for dialysis access. If you find a fistula:
Bruising can occur in uraemia as can scratch marks associated with pruritus.
Blood pressure
This is very important in renal disease. When measuring blood pressure (which needs to be done on several occasions) it is important to measure it on the same limb each time to ensure consistent and comparable results. To ensure measurement is accurate:
• Select the correct cuff size
• Use the cuff on a fully extended arm with the stethoscope applied lightly to the brachial artery
• Always take the blood pressure with the patient sitting and standing. This is because a 5–10 mmHg increase in diastolic pressure is usually seen on standing in a patient with a healthy cardiovascular system (CVS). Postural hypotension (i.e. a drop in diastolic pressure on standing) can be detected only if both measurements are made.
The World Health Organization (WHO) defines hypertension as maintained systolic pressure of 140 mmHg or above and a diastolic pressure of 90 mmHg or above. However, blood pressure levels lower than this are associated with improved survival in epidemiological studies. Most renal diseases are associated with the development of hypertension.
Face
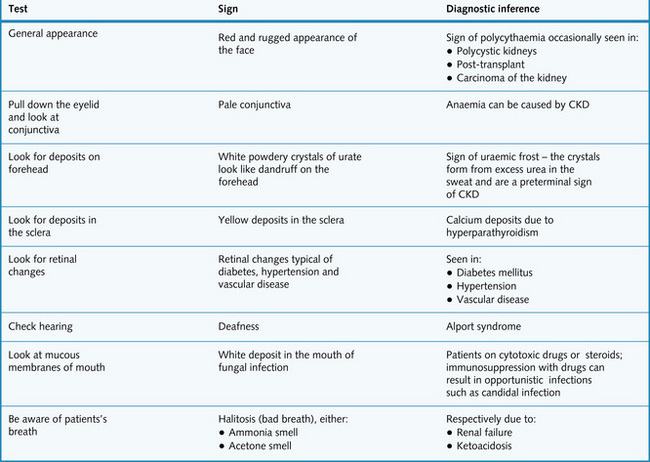
In the face you will want to check the general appearance, look at the conjunctivae for pallor and look in the mouth for ulcers, infections and fetor. You may want to carry out ophthalmoscopy and check the patient's hearing. The facial signs related to renal disease are listed in Fig. 8.4.
Neck
Identify any central venous lines placed that may be used for dialysis access.
Measure the jugular venous pressure (JVP) which is a good indicator of fluid load. The patient should be at 45° and with the head turned to the left. Look between the two heads of the sternocleidomastoid on the left side and note any pulsations in the internal jugular vein. This can be differentiated from visible arterial pulsations because:
If a waveform is visible then measure the height from the top of the fluid level vertically down to the angle of Louis. More than 4 cm is considered raised.
Thorax
Observe the shape of the chest which can be deformed by renal osteodystrophy (a late sign).
Respiratory system
Few signs in a respiratory examination indicate renal disease. A key change to watch for is the pattern and rate of respiration – patients with a metabolic acidosis associated with chronic kidney disease often have a deep, sigh-like respiration, with a rapid breathing rate. This is known as Kussmaul's respiration, and is caused by direct stimulation of the respiratory centre in an attempt to correct the systemic acidosis.
Bilateral pleural effusions can occur in nephrotic syndrome or fluid overload. The signs are dullness to percussion, reduced breath sounds and reduced vocal resonance.
Listen for fine inspiratory crackles indicating pulmonary oedema. This can be caused by volume overload causing congestive cardiac failure.
Cardiovascular system
The kidneys are linked to the cardiovascular examination because volume overload, hypertension and anaemia can produce cardiovascular signs. Palpate the apex beat which might be laterally displaced in left ventricular dilatation, thrusting in hypertension and left ventricular hypertrophy.
Abdomen
The patient should be lying as flat as possible on a firm mattress with arms by the side and head supported with one or two pillows – check that the patient is comfortable. This position ensures that the abdominal muscles are relaxed, making palpation much easier.
Stand on the right-hand side of the bed, ideally with the patient exposed from ‘nipples to knees’; to maintain privacy expose the patient from the xiphisternum to the level of the symphysis pubis.
Inspection
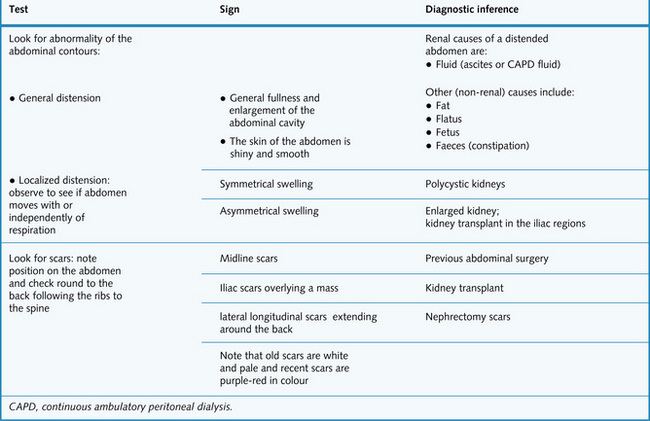
Spend the first 20–30 s inspecting the patient from the end of the bed. Look around the patient for any clues – drips, drains and dialysis machines. Fig. 8.5 gives the signs usually seen on general inspection of the abdomen in a patient with renal disease.
Palpation
Before palpating, check if the patient is in pain. If the answer is yes, ask where the painful or tender area is and start palpating from the point furthest from the locus of the pain. Tell the patient to breathe normally and relax – you will be able to feel much more through relaxed abdominal muscles. It can help to kneel at the bedside so that you are at the same level as the patient – always demonstrate this in an examination.
Develop a routine for examining all the regions and organs to avoid omitting anything:
• Begin with gentle palpation of the nine regions of the abdomen; then repeat using deeper palpation – keep looking at the patient's face for any signs of discomfort
• Feel for the liver and spleen while the patient is breathing deeply in and out
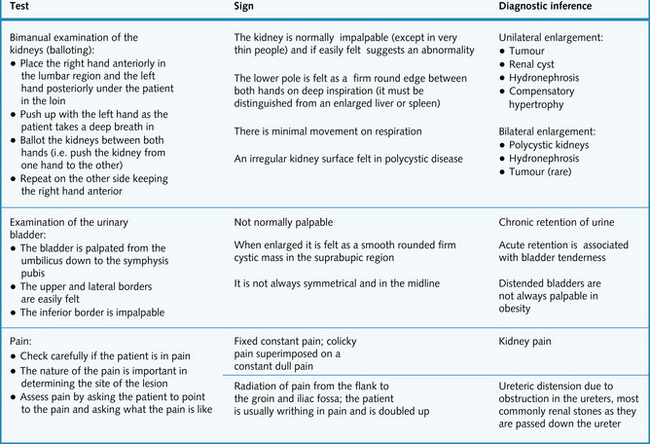
• Next examine the kidneys and urinary bladder, feeling for the aorta, and then check the hernial orifices (inguinal and femoral) while the patient is coughing
• If you feel a mass, define its site, size, shape, edge, surface (regular or irregular), consistency, mobility, movement with respiration and whether it is pulsatile or resonant to percussion. Also check for associated scars and listen over the mass for a bruit.
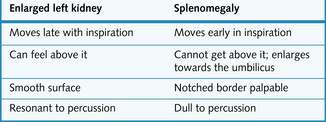
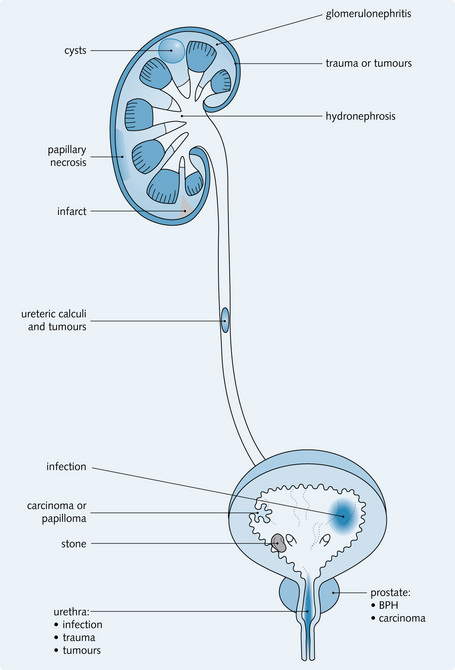
Fig. 8.6 explains how the kidneys are examined and gives the relevant findings in various renal conditions. The differentiating features between an enlarged left kidney and splenomegaly are summarized in Fig. 8.7.
Percussion
Percussion in the abdomen is used to elicit the cause of any distension and the composition of any mass – fluid-filled cysts and solid tumours are dull on percussion. The presence of ascites (excess free fluid in the peritoneal cavity) is shown by shifting dullness. To examine for this the abdomen is percussed from the midline into the flank for any areas of dullness. Keep your finger on the area of dullness whilst asking the patient to roll away from you on to their side. Percuss this area with the patient in the new position and see if it is now resonant to percussion. This is due to redistribution of fluid in the peritoneal cavity.
Auscultation
A bruit is heard if there is rapid turbulent movement of blood through a narrowed artery. Causes of renal bruits include:
Renal bruits can be heard by auscultating just superior to the umbilicus or in the flank. However, it can be difficult to distinguish a renal bruit from one originating in the aorta.
Digital rectal examination
Always obtain permission and have a chaperone. The patient should lie on the left side, with the knees drawn up to the chest.
• Inspect the perianal area for haemorrhoids, fissures, inflammation, prolapse and ulcers
• Put lubricant on the glove and insert finger into the rectum
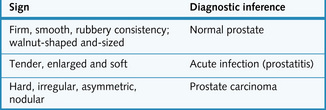
• Assess the tone of the anal sphincter and the size and shape of the prostate (normally walnut-sized)
• After removing your finger, inspect for any blood or faeces.
Palpable changes in the prostate and their clinical significance are summarized in Fig. 8.8. A vaginal examination should also be performed in female patients. (See Crash Course: Obstetrics and Gynaecology for further details.)
Legs
Examine the legs for rashes which might indicate vasculitis related to the kidneys, e.g. Henoch-Schönlein purpura.
If the legs are swollen confirm that it is pitting oedema by pressing for a few seconds and seeing if the indentation remains afterwards. This is associated with nephrotic syndrome and volume overload.
Testing the blood and urine
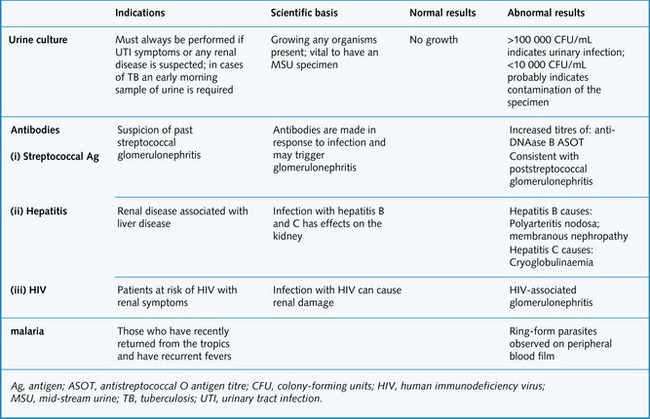
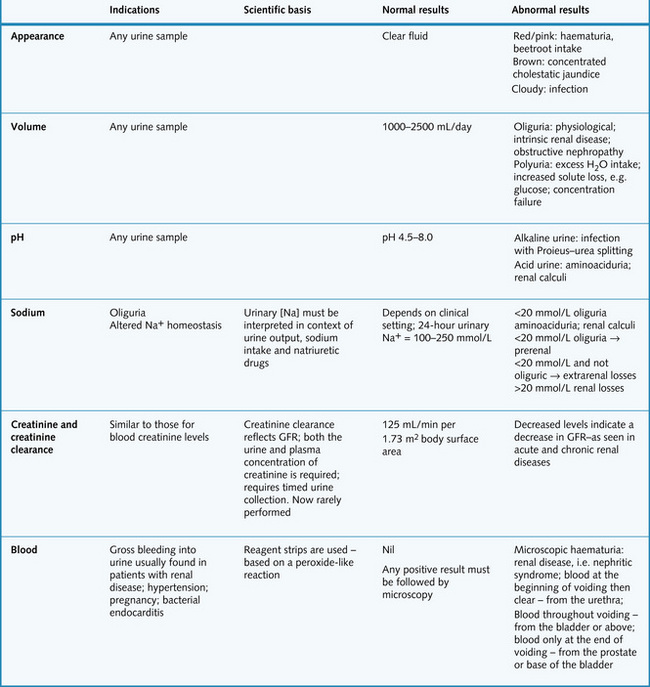
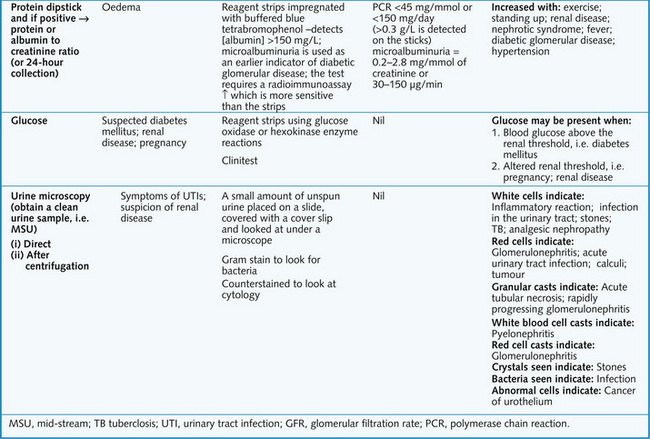
Diseases of the renal and urinary tract are suggested by symptoms linked to the urinary tract, abnormal urinalysis or abnormal serum urea or creatinine concentration. Testing the urine is the simplest investigation and should always be done in suspected renal disease. This is done with a midstream urine (MSU) sample, and assessment includes appearance, pH, dipstick, microscopy and cytological examination (Figs 8.9 and 8.10).
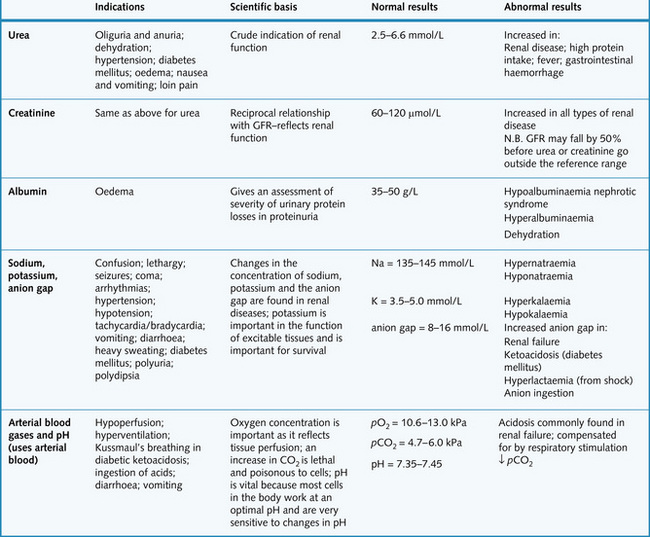
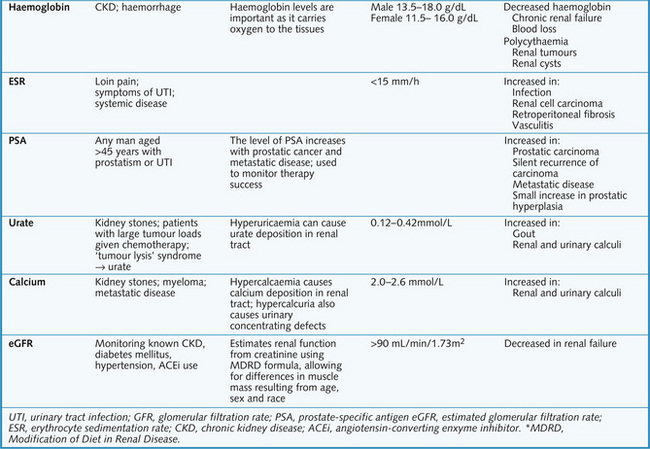
Plasma urea and creatinine are used to assess renal function. However, a significant amount of renal damage can occur before abnormal values are detected in the blood. The preferred option to assess renal function is the calculation of estimated glomerular filtration rate (eGFR) which closely reflects true GFR especially at the lower (clinically significant) end of the range (see Chapter 2). A full blood count may show anaemia (due to blood loss or impaired renal function). Other important blood results are shown in Fig. 8.11.
Haematuria
• Microscopic: blood is visible only under a microscope or on dipstick analysis
• Macroscopic (‘frank’ haematuria): blood is visible with the naked eye (> 5 red blood cells per high-power field).
The degree of haematuria does not always reflect the severity of the underlying disorder.
Causes
• Renal causes: glomerular disease such as primary glomerulonephritis (e.g. IgA nephropathy), disorders secondary to systemic illness (e.g. vasculitis, systemic lupus erythematosus (SLE)), carcinoma (both renal and transitional cell), trauma, cystic disease, emboli
• Extrarenal causes: urinary tract infection (UTI)*, ureteral calculi*, prostatic hypertrophy*, carcinoma of the bladder*, renal stone*, trauma, urethritis, catheterization, post-cyclophosphamide
• Systemic causes: coagulation disorders, sickle-cell trait or disease
(*Indicates the most common causes.)
Dipsticks detect haemoglobin (not red blood cells) and will give positive results if there is intravascular haemolysis, since haemoglobin is filtered freely by glomeruli (haemoglobinuria). This can occur physiologically, after heavy exercise, during pregnancy or with prosthetic heart valves. If haemolysis is severe (i.e. in haemolytic crisis), the urine can become red.
Other conditions can cause a red-brown discoloration of the urine that can be confused with haematuria (e.g. porphyria, myoglobinuria, ingestion of some foods (beetroot) or drugs (phenolphthalein)).
Diagnostic approach
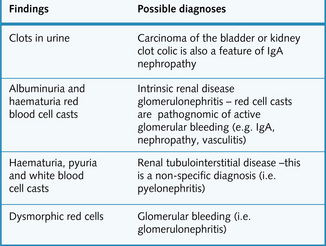
Fig. 8.12 shows the common sites of lesions causing haematuria. The significance of urinalysis is summarized in Fig. 8.13. Transient causes of haematuria should first be excluded with a MSU culture. eGFR should be measured.
Macroscopic haematuria is investigated with MSU culture, urine cytology and urological imaging, e.g. ultrasound and intravenous urogram or CT urogram. Finally diagnostic flexible cystoscopy is performed.
Microscopic haematuria should be measured with dipstick tests. One + of blood or more on two dipstick tests or any symptoms requires further investigation. This would include eGFR, urine ACR and blood pressure. Then renal ultrasound would be performed if the patient was young or urological imaging and cystoscopy if old or had risk factors for urological malignancy.
Proteinuria
Proteinuria is the presence of excess protein in the urine. It is usually assessed using a dipstick, which detects protein levels above 300 mg/L. ‘Microalbuminuria’ is the presence of excess urinary albumin but in amounts insufficient to cause a positive dipstick analysis. Proteinuria is best measured as the protein concentration on a ‘spot’ (early morning) urine sample corrected for urine creatinine concentration (protein to creatinine ratio or albumin to creatinine ratio). It may also be quantified on 24-h urine collections to give the amount excreted in 24 h but this is difficult to perform accurately and no longer routinely recommended. The amount of protein excreted can vary through the day and may increase with up-right posture (orthostatic proteinuria). Urine usually contains < 20 mg/L of albumin and < 200 mg/day of protein (exact values vary from laboratory to laboratory according to methods used to measure protein).
Proteinuria is seen in diabetic nephropathy. As well as being a risk factor for progressive chronic kidney disease (CKD), proteinuria is also associated with increased cardiovascular risk in hypertension and ischaemic heart disease.
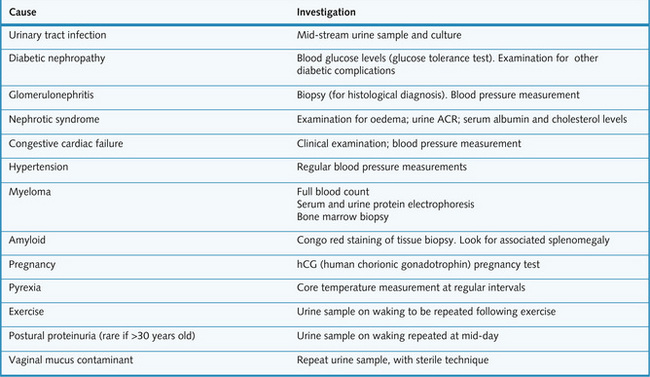
The causes of proteinuria are summarized, together with relevant investigations, in Fg. 8.14.
Imaging and other investigations
Imaging is a very useful investigation in renal disease when used in conjunction with other investigative techniques. Radiological imaging of the upper and lower urinary tract can be used to:
Plain radiography
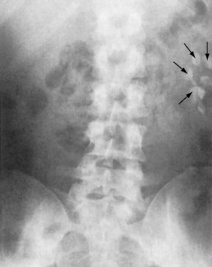
Plain radiography of the kidney, ureters and bladder (KUB) is a simple, non-invasive test that can be used before specialized imaging. It is used to detect calcification in the kidney, such as renal and urinary tract stones – uric acid stones cannot be detected, but in general 90% of stones are radio-opaque (Figs 8.15 and 8.16). It also shows the size and position of the kidneys (this is unreliable), and any secondary bony deposits (such as can be associated with prostatic cancer).
Ultrasonography
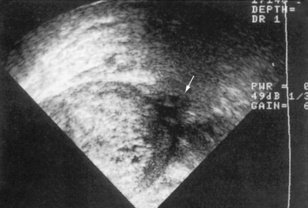
Ultrasonography is a non-invasive technique that involves high-frequency sound waves. It can accurately assess the size, shape and position of the kidney, and can also distinguish solid masses and renal cysts (Figs 8.17 and 8.18). Dilatation of the pelvicalyceal system and upper ureters can also be detected – suggesting the presence of urinary tract obstruction. This is a major cause of reversible renal failure, and can be treated if detected early enough. Transrectal ultrasound (TRUS) can also assess prostate size and be used to guide a prostate biopsy (Fig. 8.19). Renal vein thrombosis can be detected with Doppler ultrasonography, and arterial Doppler studies can be used to identify renal artery stenosis. The specificity and sensitivity of ultrasound investigations are very operator-dependent.
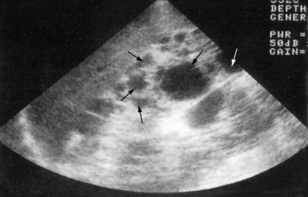
Fig. 8.17 Ultrasound scan showing the typical appearance of polycystic kidneys. There are multiple cysts (arrows) in the parenchyma.
(From Lloyd-Davis RW et al, 1994. Color atlas of urology, 2nd edn. Mosby Year Book.)
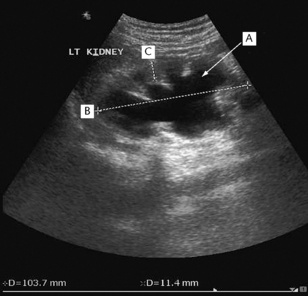
Fig. 8.18 Hydronephrosis of the left kidney demonstrated by ultrasonography. The echolucent (black) areas within the kidney are caused by dilated calyces (A). The bipolar length (B) of the kidney is normal and the cortical thickness (C) is well preserved, suggesting that prompt relief of the obstruction will allow good functional recovery.
Computed tomography
Computed tomography (CT) is a quick and non-invasive technique, which can be used with or without contrast. It is used to define renal and retroperitoneal masses and is ideal for locating and staging renal tumours (Fig. 8.20). It is also used to show polycystic kidney disease and has the advantage of also highlighting non-renal pathology. Modern techniques involving spiral CT can be used to visualize the anatomy of the renal arteries, renal vein and inferior vena cava, as well as retroperitoneal studies. Increasingly, CT without contrast is the investigation of choice to diagnose obstruction to the urinary tract or renal calculi (Fig. 8.21).
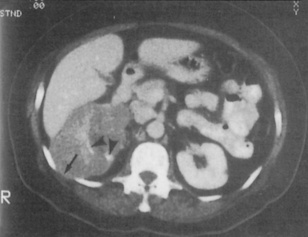
Fig. 8.20 CT scan highlighting a right renal cell carcinoma that extends through the intercostal space between ribs 11 and 12 (arrow) and medially along the renal vein. The high density (white) areas (arrows) indicate calcification.
(From Williams G, Mallick NP, 1994. Color atlas of renal diseases, 2nd edn. Mosby Year Book.)
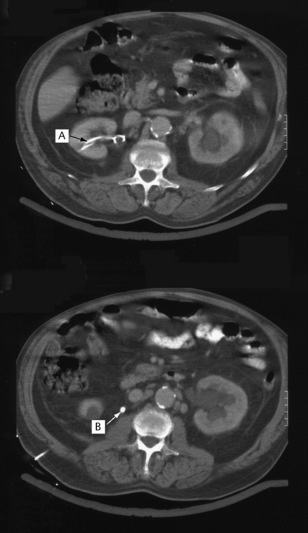
Fig. 8.21 An abdominal CT scan of a patient who presented with acute renal failure and bilateral loin pain. There is bilateral hydronephrosis secondary to bilateral ureteric stones. In the upper image a nephrostomy tube is seen in the right renal pelvis (A). The lower image demonstrates a dense opacity (calculus) lying in the ureter approximately at the level of L2 (B). Subsequent images demonstrated a similar opacity at L3 on the left.
Intravenous urography and intravenous pyelography
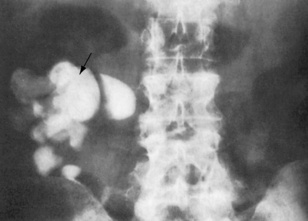
Intravenous urography (IVU) and pyelography (IVP) involve serial radiographs taken after intravenous injection of radio-opaque contrast medium (Figs 8.22 and 8.23). Normal kidney function is required, and the patient must not be pregnant. An IVU can assess kidney size and shape as well as the anatomy and patency of the calyces, pelvis and ureters. It can also be used to localize fistulae and highlight filling defects in the bladder.
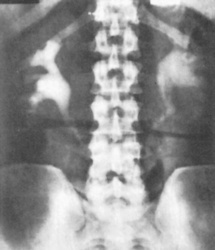
Fig. 8.22 An intravenous urogram showing bilateral hydronephrosis in response to bladder neck obstruction caused by dense granulation and fibrous tissue in a patient with schistosomiasis of the bladder.
(From Williams G, Mallick NP, 1994. Color atlas of renal diseases, 2nd edn. Mosby Year Book.)
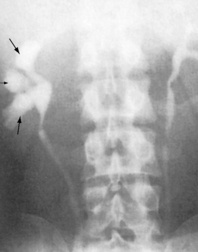
Fig. 8.23 An intravenous urogram showing marked calyceal clubbing in the right kidney (arrows). There is gross dilatation of the calyces, which is pronounced in all poles of the kidney. These findings are the result of unilateral reflux of urine and chronic infection.
(From Lloyd-Davis RW et al, 1994. Color atlas of urology, 2nd edn. Mosby Year Book.)
Investigations involving contrast involve the risks of allergy to the contrast medium and renal damage (especially if there is pre-existing chronic kidney disease). Allergy can range from mild (itching, nausea and vomiting) to severe life-threatening anaphylaxis. Previous reactions to contrast are a contraindication to its further use.
Renal arteriography
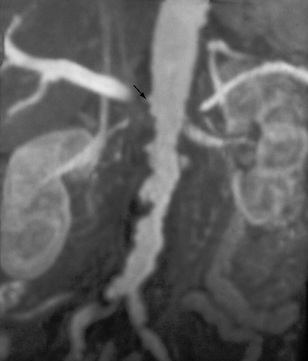
Conventional renal arteriography uses contrast medium to demonstrate the anatomy of the renal arteries. It is used to detect renal artery stenosis or aneurysms (Fig. 8.24). Therapeutic angioplasty may be performed at the same time. It can also be used in the diagnosis of tumours, but this is becoming less common with the increasing use of CT. A catheter is introduced into the femoral artery, through which contrast is injected into the renal artery and a series of radiographs are taken.
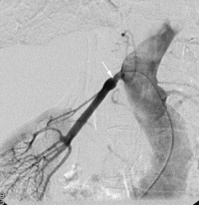
Fig. 8.24 Subtraction arteriogram of a right kidney. There is a single right renal artery with a significant stenosis at the ostium (arrow) with post-stenotic dilatation.
Renal artery stenosis can also be detected using magnetic resonance imaging, avoiding the use of potentially nephrotoxic contrast (Fig. 8.25).
Micturating cystourethrography
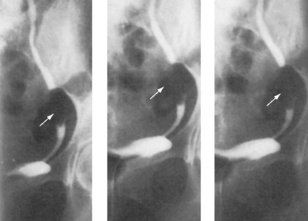
Micturating cystourethrograms are used to demonstrate vesicoureteric reflux from the bladder to the ureters during emptying of the bladder (Fig. 8.26). Reflux can be classified into three grades.
• Grade 1: contrast medium enters the ureter only
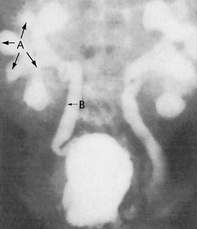
Fig. 8.26 A micturating cystourethrogram showing bilateral ureteric reflux. This patient has early calyceal clubbing (A) and ureteric dilatation (B). This is grade 3 reflux.
(Courtesy of Mr RS Cole.)
This technique was used to investigate patients with recurrent urinary tract infections but has largely been replaced by other techniques because of concerns over ionizing radiation (especially in children).
Urodynamic studies
These are used to distinguish urge incontinence from genuine stress incontinence. They also detect bladder/detrusor muscle instability. The bladder is catheterized and a pressure probe is inserted to measure the bladder pressure. A rectal probe is also inserted to assess intra-abdominal pressure. The detrusor muscle pressure can be calculated by subtracting the bladder pressure from the intra-abdominal pressure. The bladder is then filled with water until the patient feels the urge to void. At this point the relative pressures are recorded. If the patient has stress incontinence, an increase in intra-abdominal pressure (e.g. coughing) leads to involuntary urine leakage, with no bladder/detrusor muscle contraction. If the patient has urge incontinence, the bladder/detrusor muscle contracts either spontaneously or with increased abdominal pressure, and the patient feels an overwhelming urge to urinate immediately (Fig. 8.27).
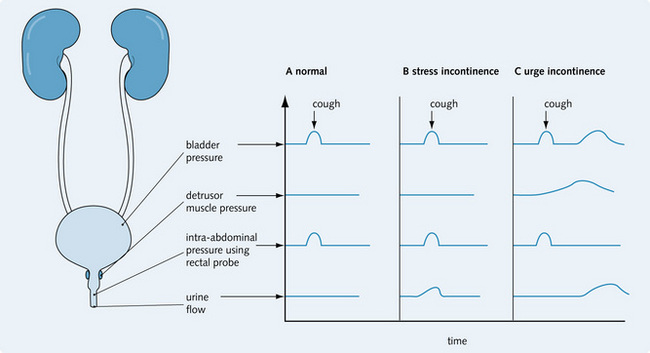
Fig. 8.27 Diagnosing incontinence using urodynamic studies. (A) Normal cough; (B) identifies stress incontinence, in which urine leakage is seen in response to raised intra-abdominal pressure; (C) shows urge incontinence, in which urine leakage is seen in response to detrusor muscle instability following a rise in intra-abdominal pressure.
Retrograde pyelography
Retrograde pyelography is used to define the site of an obstruction (Fig. 8.28) or lesions within the ureter. It does not require functioning kidneys. Under general anaesthesia a ureteric catheter is inserted into the ureter under cystoscopic guidance. Contrast medium is injected into the catheter to identify any lesions. It may be used therapeutically to help dislodge ureteric stones and coax them down the ureter.
Antegrade pyelography
This is used to define the site of obstruction in the upper urinary tract, i.e. mainly within the pelvicalyceal system. Following percutaneous catheterization of a renal calyx contrast medium is injected as described above for retrograde pyelography Percutaneous catheterization of the pelvicalyceal system (nephrostomy) is also used therapeutically to relieve obstruction.
Magnetic resonance imaging
Magnetic resonance imaging (MRI) is an imaging technique that does not involve ionizing radiation – unlike CT. Instead, it relies on the measurement of the magnetic fields of atomic nuclei. It can differentiate cystic and solid renal masses and is useful for precise staging of tumours. However, MRI cannot be used in patients with pacemakers or other metallic implants. Magnetic resonance has now been developed to provide resolution sufficient to diagnose atheromatous renal artery stenosis (see Fig. 8.25). MR urography can also allow anatomical and functional evaluation of the urinary tract withouth the need for ionizing radiation or iodine-containing contrast agents.
Radionuclide scanning
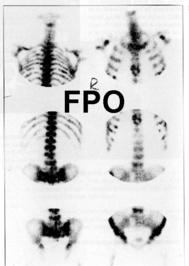
Technetium-labelled dimercaptosuccinic acid (99mTc-DMSA) provides static images of the renal parenchyma. It highlights the localization, shape and function of each individual kidney, and highlights scarring as a result of reflux nephropathy (Fig. 8.29). Technetium-labelled pentetic acid (99mTc-DTPA) is excreted by renal filtration and the changes in the level of 99mTc-DTPA in the kidney over time are quantified using a gamma camera. This provides a dynamic index of blood flow to each kidney. It is used to assess transplant function (Fig. 8.30A) and can demonstrate obstruction to the upper urinary tract (by diuresis renogram; Fig. 8.30B). It can also be used to determine the relative function of each kidney. Both 99mTc-DMSA and 99mTc-DTPA are injected into the venous circulation. Radionuclide techniques can be used to demonstrate reflux nephropathy.
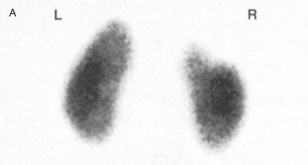
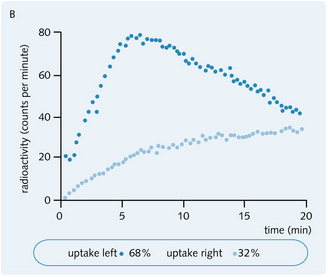
Fig. 8.29 (A) 99mTc-DMSA scan showing a right upper pole scar (courtesy of Dr TO Nunan). (B) The graph shows a diminished uptake of 36% for the right kidney, indicating a degree of loss of function correlating with the scar.
(From Williams G, Mallick NP, 1994. Color atlas of renal diseases, 2nd edn. Mosby Year Book.)
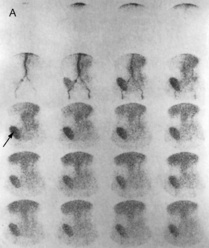
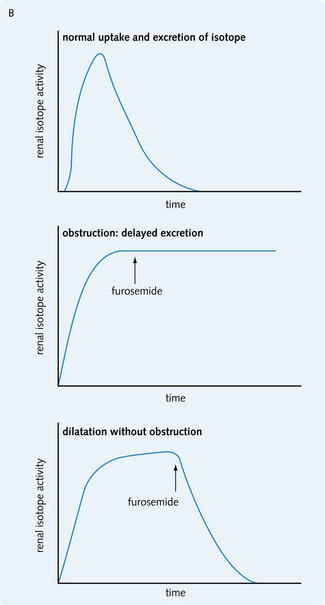
Fig. 8.30 (A)99mTc-DTPA diuretic renogram showing a transplanted kidney functioning normally (arrow). (From Catto GRD et al, 1994. Diagnostic picture tests in renal disease. TMIP.) (B) Diuretic isotopic renography showing tracings for normal, obstructed and dilated (without obstruction) upper urinary tract. Obstruction can be distinguished from dilatation by administering furosemide, which promotes excretion of the isotope in dilatation (without obstruction) but has no effect on excretion rates in obstruction.
(From Johnson RJ, Feehally J, 2000. Comprehensive nephrology. Mosby Year Book.)
Other imaging techniques
Other imaging techniques include scintigraphy. This may be used to investigate vesicoureteric reflux in place of conventional imaging techniques, avoiding exposure to large doses of X-rays. Scintigraphy can also demonstrate secondary deposits in the bone (Fig. 8.31).
Cystoscopy
A rigid or flexible cystoscope is inserted through the urethra to inspect the interior surface of the lower urinary tract (bladder and urethra). This technique is very useful in the diagnosis and treatment of tumours in the bladder. It can also be used to identify stones and fistulae, to take a tissue biopsy and to assess prostatic disease.
Diagnostic cystoscopy can be carried out in the outpatient clinic, and involves a flexible cystoscope examination under local anaesthesia. Therapeutic cystoscopy may require a hospital admission and uses a rigid cystoscope, with the patient under general anaesthesia.
Renal biopsy
Renal biopsy is necessary to classify glomerulonephritis, which can influence the choice of therapy in patients with nephrotic syndrome and acute nephritis. It is also used in the diagnosis and assessment of systemic diseases that affect the kidneys, e.g. sarcoidosis and systemic lupus erythematosus. It can aid the investigation of unexplained acute kidney injury, proteinuria, and haematuria and is vital in the management of patients with renal transplants. A sample of the kidney tissue can be taken by inserting a biopsy needle into the kidney under ultrasound guidance, with the patient lying in the prone position. This is then examined under a microscope, using immunochemical staining to detect complement or immunoglobulins. Relative contraindications to renal biopsy include:
• A bleeding diathesis (absolute unless corrected)
• Single kidney (risk of loss)
• Obesity (technically difficult)
The main complications are pain, bleeding (haematuria or perinephric haematoma) or infection (rare).