CHAPTER 21 Dental materials
Introduction
Throughout recorded equine dental history, exodontia has been the only treatment option for diseased teeth. The goal of equine dentistry is to preserve functional dentition in order to promote the general health, longevity, and productivity of the horse. In an effort to preserve functional dentition, clinicians have begun to apply accepted dental technologies and to adapt dental materials from human and small animal dentistry. This cross-species extrapolation is common in both human and veterinary dentistry since all mammalian dental tissues are similar; however, the equine clinician must understand the differences between brachydont and hypsodont teeth when applying materials designed for use in human teeth.
The purpose of this chapter is to serve as a general resource for equine practitioners to facilitate the incorporation of dental materials into their practice. It includes a basic discussion of dental cements, restorative, endodontic, and periodontal materials, and impression materials. Specific material brand recommendations have been purposely omitted since hundreds of materials are available in each category and since the dentist’s preferences are a major factor in the final selection of a specific brand. However, many experienced dentists advise using a single line of materials for each category. The general clinical application steps for each category of materials is presented, but every dental material has unique properties, handling characteristics, and an application protocol based on the ratios of essential components and the addition of proprietary components. To optimize the clinical properties of any material, the manufacturer’s instructions for storage, mixing, and application must be followed exactly.
Dental cements
Dental cements are primarily used for bonding prosthetics (luting agents) and orthodontic appliances to teeth. Zinc phosphate, zinc oxide eugenol, and polycarboxylate cements are available and still used in dentistry. However, glass ionomer and resin composite cements are primarily used today because of their superior properties and handling characteristics. To select and apply the appropriate cement, practitioners must be familiar with the bonding mechanism for each material (Box 21.1). Since luting applications are rarely practiced in equine dentistry, dental cements will be discussed with respect to their use in the other dental disciplines.
Box 21.1
Types of bonding
1. Mechanical retention: Non-adhesive bonding where the dental material infiltrates the surface irregularities of the dental tissue and cures to interlock with the dental tissue. All cements exhibit mechanical bonding.
2. Chemical bonding: Glass ionomer and polycarboxylate cements form a chemical crystal bond between the carboxyl groups in the polyacid of the cement and the calcium ions of the apatite crystals in the enamel and dentin.
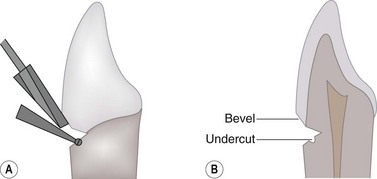
Fig. 21.1 Placement of small retention groove in dentin using a small round burr.
(Courtesy of K-J. Söderholm.)
Restorative dentistry
Restorative dentistry is the dental discipline concerned with the treatment, repair, and conservation of teeth broken down by trauma or decay. The goals of restorative dentistry include returning the diseased tooth to its original shape and function, preventing breakdown of the remaining tooth structure, protecting the pulp from thermal, mechanical, and bacterial insult, and creating an esthetic tooth appearance. The indications for restorative dentistry include dental decay, resorptive lesions, crown fractures, attrition, congenital anomalies, enamel hypoplasia, and access closure of an endodontically treated tooth.1 Restorative dentistry can be divided into two sub-disciplines: direct placement restorations and laboratory-assisted restorations (prosthodontics). The scope of this discussion will be limited to direct placement restorations.
Regardless of the disease etiology, the restoration of a tooth includes two equally important procedures: 1) cavity preparation and 2) selection and application of the restorative materials. Treatment planning for any restoration must include radiographic evaluation of the affected tooth and its surrounding tissues. Radiographic evaluation of the diseased tooth includes evaluation of the pulp, the specific location of the lesion, and the depth and extent of the lesion. Radiographic findings consistent with pulp disease would indicate root canal therapy before tooth restoration. The location of the lesion on each specific tooth determines the forces that will be applied to the restoration. Restorations on the occlusal surface of a tooth must be designed to withstand compressive loading and wear, whereas restorations on the apical aspect of the clinical crown might experience tension or bending stresses. Regardless of the present location of the lesion, all restorations have the potential for eventual occlusal wear due to hypsodontic tooth eruption; therefore, the strength and wear resistance of restorative materials must be considered during material selection. The depth and extent of the lesion must be evaluated to determine the dental tissues involved in the lesion, as well as the proper size and shape of the cavity preparation. Consideration of these factors will determine the required properties of the restorative materials needed to fill the cavity.
During treatment planning, the clinician should also try to identify and eliminate the cause of the lesion, especially in traumatic cases. Failure to eliminate the cause often leads to subsequent destruction of the restoration. Finally, a client’s esthetic demands may influence the practitioner’s choice of the final restorative material and its finish.
Cavity preparation
Cavity preparation is the surgical operation involving the debridement of decayed or diseased dental tissues in order to shape the tooth to receive and retain the restorative material.2 Regardless of the etiology or location of the lesion, the operator must adhere to the following principles. First, the cavity must be prepared so that all diseased and damaged dental tissues are removed without weakening the tooth’s structure. Inherent in this principle is that as much tooth as possible must be preserved so that the restoration does not compromise the structural integrity of the tooth. Secondly, the cavity is extended to prevent further decay or damage to the restoration. The focus of this extension is the removal of any unsupported or undermined dentin and enamel. To achieve this, the walls of the cavity are formed parallel to the enamel rods, which are usually oriented perpendicular to the tooth surface (Fig. 21.4). Finally, the cavity is configured to facilitate filling, retention, and finishing of the restorative material. This step may include dentinal undercutting for mechanically retained materials and marginal beveling to increase the enamel surface bonding area.3 Advances in modern restorative materials make the necessity of dentinal undercutting debatable.4 Since cementum, not enamel, is the peripheral tissue on the crown of equine teeth and since the bonding of restorative materials to cementum has not been studied, the value of marginal beveling of the cavity preparation is also debatable.
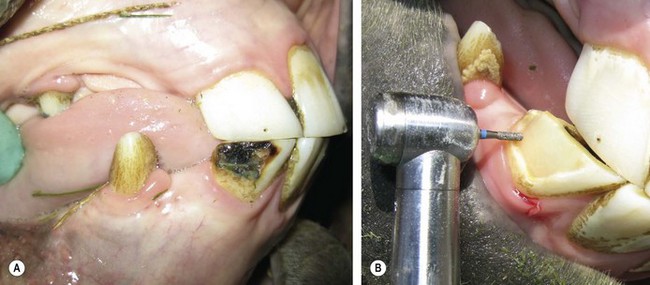
Fig. 21.4 (A) Decay of the mandibular right 3rd incisor (403). (B) Cavity preparation using a diamond burr on a high-speed handpiece. Note that the walls of the preparation are perpendicular to the surface of the tooth. The author elected not to instrument retention grooves (dentinal undercuts) or marginal beveling since this shallow preparation was designed for a resin composite filling.
Bases and liners
Cavity preparations, in which less than 2 mm of dentin remains between the pulpar wall and the pulp (indirect pulp exposure), require the application of a pulp protecting material.5 Cavity varnishes, liners, and bases are used to protect the pulp. Cavity varnishes are organic solvent and resin solutions that seal dentinal tubules. They do not prevent acid penetration or thermal conductivity, and are losing popularity since the organic solvent can interfere with the polymerization of resin composites.
Cavity liners are non-irritating materials that are placed in a thin layer to protect the pulp and decrease dentinal sensitivity. They provide no thermal or mechanical protection and are inadequate as a sole protecting medium. Calcium hydroxide (CaOH), the most popular liner, is supplied as a powder or as commercially prepared pastes. The powder can be applied directly into a cavity or mixed into a paste with water, saline, or an anesthetic. The strong alkalinity (pH 12.5 when mixed with saline) of CaOH is bactericidal, neutralizes acids, and induces reparative dentin formation. CaOH dissolves if contaminated with oral fluids and must be covered by another restorative material.
Cavity bases are used in deep cavities to provide structural support for the final restoration and chemical and thermal protection of the pulp. Dental cements are typically used as bases. Reinforced zinc oxide-eugenol (ZOE) cement (Intermediate Restorative Material, IRM)a has been a historically popular cavity base. This material is losing popularity because eugenol interferes with the bonding of resin composites. ZOE cements have a pH of approximately 7, which is thought to have protective and soothing properties on the pulp. In cases of direct or near pulp exposure, a liner should be used since ZOE cements can cause pulp inflammation. ZOE cements are dispensed as zinc oxide powder and eugenol liquid or as a two paste system. Zinc oxide and eugenol chemically react to form a chelate. The setting time is accelerated by humidity, elevated temperature, and increasing the powder-to-liquid ratio. Therefore, ZOE is typically mixed on a cooled glass slab to slow the setting reaction (Fig. 21.5). Condensation (humidity) on the cooled pad will accelerate the setting reaction and negate the cooling effect. The powder-to-liquid ratio for the mix is dictated by the use of the cement. When mixing a ZOE base material, sufficient powder must be incorporated to produce a stiff, putty-like consistency (Box 21.2).
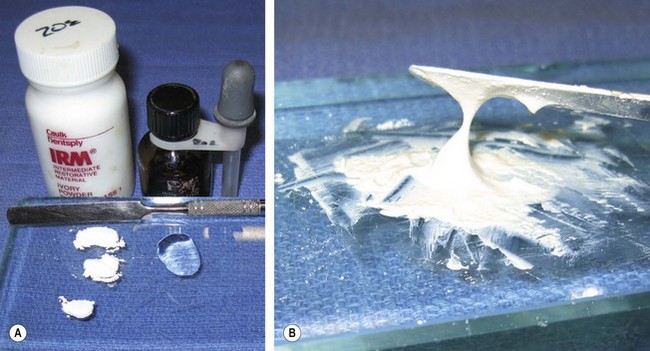
Fig. 21.5 Mixing dental cement. (A) The zinc oxide powder and the eugenol liquid are placed on a glass slab for mixing. (B) Properly mixed cement has a uniform creamy consistency that can be pulled approximately 1 cm.
Box 21.2
Preparation of dental cement (zinc oxide-eugenol)
• A small amount of powder is placed on a mixing slab or pad. A few drops of liquid are placed onto the slab beside the powder.
• Approximately 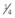 of the powder is pulled into the liquid and mixed with a spatula to a creamy consistency.
of the powder is pulled into the liquid and mixed with a spatula to a creamy consistency.
• Another 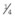 of the powder is incorporated into the mixture and spatulated.
of the powder is incorporated into the mixture and spatulated.
• The remainder of the powder is pulled into the mixture and spatulated.
• The final consistency of the mixture is dictated by the cement’s intended use; however, for most applications, the cement is properly mixed when a one-cm string can be pulled between the mixing pad and the spatula.
Zinc phosphate (ZP) cements are the oldest and least expensive cements. They have high compressive strength and good thermal insulation properties and historically have been used as a restoration intermediate layer between gutta percha (GP) and the final composite restoration. Due to the acidic nature of the material, ZP is not recommended as a base in cases with direct or near pulp exposure.6
Glass ionomer (GI) cement is currently the most popular base material. The properties of GIs are discussed below, under Direct placement restorative materials.
Direct placement restorative materials
The ideal restorative material would allow for conservative cavity preparation, be easy to apply, bond to the substrate (dental tissues), have the similar strength, thermal, and wear characteristic to the tooth, and be the same color as the tooth. No material has all of these ideal characteristics. Therefore, a material, or combination of materials, must be selected based on its specific advantages in a specific situation. Three basic groups of restorative materials are used in veterinary dentistry: amalgam, glass ionomers, and resin composites. These materials have also been combined to produce materials (e.g., resin modified glass ionomers) in an attempt to gain the advantages and minimize the disadvantages of the base materials.
Dental amalgam is the alloy of mercury mixed with other metals (usually silver, tin, and copper). This material has been the primary direct restorative material in human dentistry for over a century due to its ease of use, low technique sensitivity, ability to maintain cavity form, and wear resistance. Amalgam is considered the gold standard for load-bearing (occlusal) restorations. This material is losing favor among human dentists and has seen minimal veterinary use due to its material disadvantages and also due to the popularity of modern composite materials. The toxicity of mercury requires special handling. Current evidence does not support the popular concerns about systemic toxicity secondary to dental fillings.7 Amalgam corrodes over time, and the metallic color is esthetically displeasing. Amalgam does not bond to dental tissues and is retained by macromechanical forces, which necessitates additional cavity preparation steps, such as dentinal undercutting, and makes the restoration susceptible to marginal leakage. Finally, the applicability of amalgam in equine teeth is uncertain due to the enamel/cementum configuration on the occlusal surface of equine hypsodontic teeth.8
Dental composites are the most commonly used restorative materials in veterinary dentistry. They are easy to apply, provide acceptable strength and wear resistance, and are esthetically pleasing. Modern composite bonding systems to dentin and enamel require limited cavity preparation and greatly reduce marginal leakage. A composite is a solid material formed from multiphased materials that have been combined to produce properties superior to the individual constituents.9 Dental composites contain three major components:
1. Matrix: an acrylic resin consisting of monomers polymerized using free radical initiators to form a solid material
2. Fillers: glass, ceramic, or composite particles that reinforce the resin matrix
3. Coupling agents: an organosilane, coats the filler particles, and covalently bonds to the matrix resin.
Other components in dental composites include tooth colored pigments, the polymerization activator-initiator system, and polymerization inhibitors to control the working time.
All commercial dental composites use free radical initiators to start an addition polymerization reaction. These free radicals are activated either chemically, by an external energy source (e.g., a curing light), or by a combination of the two mechanisms.
Light-activated resins (light-cured) are packaged as a single paste in a light-proof container (i.e., syringe or compule). Light in the visible blue range (450–475 nm) excites a photosensitizer, commonly camphorquinone, which reacts to the amine activator to produce free radicals. The advantages of light-activated resins are an unlimited working time for material placement and a short, ‘on demand’ set time (usually 30–60 seconds). The depth of cure for light-activated resins is accepted to be approximately 2 mm; therefore deep restorations must be applied using a layering technique (incremental buildup).10,11 In addition to ensuring maximum polymerization conversion, the layering technique minimizes resin shrinkage. The curing light should be held within 1 mm of the restoration to optimize light exposure, or the activation time should be extended. Light-activated resins are initiated by visible light and must be protected from room lights, especially surgical lamps. The minimum energy requirement to initiate the photosensitizer is 300 mW/cm2. Curing lamps should be tested periodically with a radiometer to ensure adequate emission. Because of the intensity of the light produced by curing lamps, operators should never look at the blue light and should use an orange protective shield or glasses to protect against retinal damage.
Chemically activated resins (self-cured, auto-cured) are packaged as two paste systems. One paste contains a benzyl peroxide initiator, and the other paste contains an aromatic tertiary amine activator. Upon mixing, polymerization begins, and the composite sets into a solid state within 3–5 minutes. Heat increases both the rate and degree of polymerization. Chemically activated resins are usually used for large, bulk fill restorations or restorations with limited light access.
Dual-cure resins are chemically activated resins in which a light activation system has been added to each paste and are indicated in restorations where light cannot penetrate the entire depth of the restoration. Light activation attains the initial set of the restoration, and the chemical activator completes the polymerization. Whether the composite is light or chemically activated, the polymerization reaction continues for at least 24 hours before the resin is completely cured. An unfilled resin coating is applied to protect the restoration from air and oral fluids during this curing period. This technique is referred to as ‘rebonding.’
Historically, the most significant problem with dental composites has been shrinkage of the matrix material during polymerization. This shrinkage creates a gap between the restoration and the cavity wall referred to as marginal leakage. In order to reduce the volumetric change within the matrix, high molecular weight monomers, which covalently bond to other polymer chains, are used. Most contemporary dental resins use a combination of bisphenol A epoxy and glycidylmethacrylate (Bis-GMA) and triethylene glycol dimethacrylate (TEGDMA) monomers to limit matrix shrinkage. These monomers also cross-link between polymerization chains to produce a composite with increased physical and mechanical properties.12 Additionally, high levels of filler particles reduce the amount of matrix in the composite, which also limits the polymerization volumetric change. Increased filler loading increases the restoration hardness, fracture strength, and wear resistance and reduces thermal expansion and contraction. While the combination of high levels of Bis-GMA and filler loading minimizes marginal leakage and improves the mechanical properties of the restoration, it also results in a viscous material with poor handling characteristics. Therefore, numerous composite materials are manufactured in an attempt to maximize the physical, mechanical, and handling properties required for different restorative applications.
Dental composites are commonly classified by the filler particle size:
• Conventional (traditional, macrofilled) composites. These stress-bearing composites have the largest particles (8–12 µm) and are rarely used because newer composites outperform them. Although they are strong, their surface is notably rough, discolors, and wears unevenly.
• Microfilled composites. These composites were designed for superior polishability and contain filler particles in the 0.04–0.4 µm range. They are indicated for low-stress, esthetic restorations and are not popular in veterinary medicine because they lack strength and wear resistance.
• Hybrid composites. These composites have a high filler content and contain various sizes of particles ranging from 0.2–3 µm. They are currently the preferred restoration material in human and veterinary dentistry because of their wide range of uses, their superior clinical properties, wear resistance, and acceptable polishability. They are used in stress-bearing and esthetic restorations. The following hybrid composites are further grouped into subcategories.
• Microhybrid composites. This subcategory of hybrid composites combines filler particles of submicron (0.04 µm) and small (0.1–1.0 µm) sizes. They were developed to offer a composite for high stress as well as esthetic restorations. In general, they have superior strength, but polishability is not better than traditional hybrids. This is the most popular category of composites because of their versatility.
• Flowable composites. This subcategory of hybrid composites consists of low viscosity (syringeable) composites with reduced filler content that flow and adapt intimately to the cavity walls. They are only recommended in low stress restorations and restorations with poor accessibility because they lack strength and wear resistance.
• Packable composites. This subcategory of hybrid composites is highly viscous and was designed to be placed similarly to amalgam. They are strong, wear-resistant, and polishable. Packable composites have no superior properties to other hybrid composites and adaptation to the cavity walls is very technique sensitive.
• Nanofilled composites. Recent advances in sol-gel technology have made submicron-sized particle production possible. The nano-particle size (0.005–0.01µm) allows for increased filler loading, which improves strength and wear resistance, as well as minimizing shrinkage.
• Nanohybrid composites. These composites combine nano particles and conventional fillers to produce a microhybrid composite with the strength and wear resistance of a traditional composite and the polishability of a microfilled composite.
• Core (buildup) composites. These high-strength composites were designed for placement under prosthodontic crown restorations where significant tooth structure has been lost. Filler particle sizes vary from micro to macro, and polishability is poor. Anecdotal success in restorations of incisor fractures and extensive decay has been reported.
• Compomers (polyacid modified resin composites). These composites have a polyacid modified resin matrix with composite and glass ionomer fillers. They release low levels of fluoride and are indicated for low-stress restorations in patients at risk for caries. Since they exhibit poor physical properties and wear resistance and release lower levels of fluoride than traditional or resin modified glass ionomers, compomers have seen little clinical use.
No clinical trials have been performed to study the use of any restorative material in equine hypsodontic teeth. However, clinical success using microhybrid composites to restore infundibular cavities was first reported in 2001.13 The use of core composite material in large defects has also been reported.14 The use of flowable composites in equine teeth has become a common practice because of the material’s handling characteristics and the adaptability of the material to the cavity; however, this application must be questioned due to the poor mechanical properties of the material. Nanocomposites have the potential to make a significant improvement over the available composite materials in both human and veterinary patients but have seen limited clinical use.
Dentin-enamel adhesives (bonding agents)
Except for compomers, composite restorative materials are hydrophobic and will not bond to hydrophilic dental tissues. Therefore, composite restorations require an adhesive application to which the composite resin can copolymerize. Adhesion is the bonding or attachment of dissimilar materials so that the materials resist separation and transmit mechanical forces across the bond. Regardless of the substrates being joined, adhesion promotion follows a prescribed generic methodology: 1) substrate preparation; 2) surface priming; and 3) placement of application-specific overlayers that react with the primer. With the exception of glass ionomers, which chemically bond to dental tissues, all modern adhesive systems follow this generic adhesive methodology.15
Substrate preparation dissolves the barrier layers that inhibit primer interaction with the substrate. In heterogeneous substrates, such as dentin and enamel, selective removal of substrate components also enables more efficient surface reactions, alters surface conformation, and changes the surface energy. Acid etching (also called conditioning) is the required surface preparation technique for bonding of restorative materials to dental tissues. Acid etching enamel removes the smear layer created by instrumentation, dissolves apatite crystals to create a microporous surface, and lowers the surface energy, which facilitates spreading (wetting) of the primer. In addition to the surface preparation effects noted for enamel, acid etching dentin increases permeability by widening the dentinal tubules and exposes acid insoluble type 1 collagen fibers, which represent approximately 90 % of the organic phase of dentin (Box 21.3).
Box 21.3
Acid-etch technique
35 % phosphoric acid is the ‘gold standard’ etchant, although other acids (e.g., polyacrylic acid) and varying acid concentrations are available. Etchants are available in liquid and gel forms, with the gel being the most popular because it is easier to dispense and because it retains its placement during vertical applications.
The etching procedure includes the following steps:
1. The preparation is isolated to prevent contamination from blood or oral fluids.
2. The tooth and cavity preparation are cleaned with non-fluoride flour pumice to remove the organic pellicle, plaque, food, and other oral fluids. The pumice is mixed with water into a thick paste and applied with a prophy cup on a low-speed handpiece. Fluoride polishing paste is contraindicated because it interferes with the etching reaction.
3. The cleaned area is rinsed and gently air dried.
4. The etchant is applied to the preparation for appropriate contact time, and then thoroughly rinsed off with water. The standard contact time for dentin is 10–15 seconds and for enamel is 30–40 seconds. The etching time for coronal cementum has not been established; however, the author (SSG) allows 20–30 seconds contact time for cementum. Over-etching should be avoided since a contact time over 120 seconds leaves insoluble calcium precipitates on the surface of enamel.
5. The etched surface is dried according to the adhesive material instructions. Most enamel bonding systems require a dry etch surface, and properly conditioned enamel has a chalky-white or frosty appearance. If this appearance is not achieved, the surface should be re-etched. Most dentin bonding systems require a moist surface with a glistening appearance. Drying the dentin desiccates and collapses the collagen fibrils, which prevents proper bonding.
6. The conditioned tooth is protected from contamination until the restoration material is applied. In the sedated horse, this often necessitates that an assistant cover the prepared tooth with sterile gauze while the operator prepares the restorative material.
After a substrate is prepared, a primer is applied to the substrate. The primer consists of molecules with chemically functional terminal groups that react to the adherends. Dental primer monomers have an adhesive hydrophilic group, which reacts with the enamel or dentin, and a hydrophobic polymerizable group that cross-links with the restorative resin. The primer is carried in acetone, ethanol, or water. The function of the primer on enamel is to completely wet the surface of the enamel and to penetrate the microporosities created by the etchant. Polymerization of the bonding agent interlocks the primer into the microporosities and forms a micromechanical bond. In dentin, the primer infiltrates the dentinal tubules (micromechanical bonding) and entangles the collagen fibers exposed during etching, which upon polymerization, forms the hybrid layer. Hybrid layer formation is the primary bonding mechanism in dentin bonding systems (Fig. 21.6). Finally, interfacial resin overlayers, with extensive crosslinking capabilities, are applied which copolymerize with both the primer and composite resin.

Fig. 21.6 The bonded composite resin. Resin flags are formed from the flow of the dentin bonding agent into the dentinal tubules. A hybrid layer is formed by the interaction of the resin and the collagen fibers of the decalcified dentin. The composite resin is bonded to this hybrid layer interface.
(From Gladwin M and Bagby M 2000. Clinical aspects of dental materials. Lippincott, Williams and Wilkins.)
Bonding agents have traditionally been classified based on generational sequencing, chronologically based on market introduction (Box 21.4). Advances in this classification system are generally indicative of a reduction in the number of application steps in the bonding procedure and are not indicative of improved bonding performance. Since the generational classification system has no correlation to improved bonding strength, Stangel et al proposed a contemporary bonding system classification based on whether the acid conditioner (etchant) is rinsed off the dental tissue or left in situ.15
Box 21.4
Generational classification of bonding agents
Since dentists commonly refer to bonding agents with respect to the generational classification system, the following outline is included:
• Introduced in the early 1980s
• Application of the dentin conditioner (etchant), primer and adhesive in distinct steps.
• The Total-Etch Technique (simultaneous aggressive etching of enamel and dentin with phosphoric acid) and the dentin ‘Wet Bonding’ process were introduced
The ‘etch and rinse’ (ER) category is divided into two- or three-step systems in which the first application step is always the etching step (Step 1). In the Two-Step ER system (One Bottle System), the primer and adhesive resin overlayer is applied in a single application (Step 2). In the Three-Step ER system, the primer and adhesive resin overlayer is applied separately (Steps 2 and 3). Within the ‘etch and rinse’ category of bonding agents, the Two-Step (One Bottle) system is the most popular. While both the Two- and Three-Step ER systems produce acceptable bonding strengths to both enamel and dentin, the Three-Step ER system has superior bonding to dentin (Fig. 21.7).
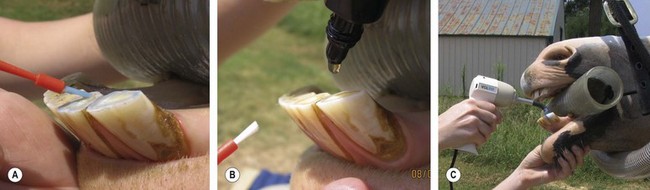
Fig. 21.7 Placement of an ‘Etch and Rinse’ (5th generation) bonding agent (BA) after incisor odontoplasty. (A) The occlusal surface is acid etched, and then rinsed and left damp. (B) The BA is applied and brushed onto the surface. (C) The BA is light cured, and then a second application is made.
The ‘no rinse’ (NR) (self-etch, self-priming) category is divided into one- or two-step systems. In the One-Step NR system, the conditioner, primer, and adhesive resin overlayer is applied together from a single bottle. In the Two-Step NR system, the combined conditioner and primer components (first bottle) are applied, followed by the application of the adhesive resin overlayer (second bottle). The NR bonding systems have failed to produce clinically acceptable bonding strength when compared to the ER systems due to poor removal of the dentin smear layer. If a ‘no rinse’ bonding agent is used for bonding to enamel, a preparatory ‘etch and rinse’ step has been recommended.16 However, this additional step defeats the entire purpose for the ‘no rinse’ system.
In 2005, a review of 85 performance trials evaluated the clinical effectiveness of six dental adhesives systems (2-Step ER, 3-Step ER, 1-Step NR, 2-Step NR, and glass ionomers (GI) with and without conditioning).17 The study found a high degree of variability within each group but made several general conclusions about contemporary bonding systems (Box 21.5).
Box 21.5
Conclusions about contemporary bonding systems based on a review of clinical trials17
1. The 3-Step ER systems out-performed the 2-Step ER systems due to phase separation and incomplete infiltration into the demineralized zone with the latter system
2. Several NR systems required selective enamel etching to be effective; therefore, they are not a true NR system
3. 2-Step NR systems showed clinically reliable performance in non-load-bearing restorations
4. 1-Step NR systems had ineffective clinical performance and had the highest failure rate of all systems
5. Resin Modified GIs performed comparably to 3-Step ER systems and better than conventional GIs
6. Although anecdotal reports may support the use of these bonding systems in load-bearing restorations, no systematic data exist to recommend this application.
Box 21.6
The basic technique for a composite restoration
1. The cavity or endodontic access is prepared (cavity preparation; Fig. 21.4).
2. In deep cavity preparations and endodontic access restorations, a liner and/or base material (e.g., calcium hydroxide, glass ionomer, and Reinforced Zinc Oxide-Eugenol Cement) may be applied.
3. The walls of the cavity are conditioned (Acid Etch Technique; Fig. 21.8).
4. A bonding agent is applied to all etched surfaces with a disposable brush and light cured. Most manufactures suggest two applications of the bonding agent (Fig. 21.8).
5. The resin composite is applied into the cavity and shaped with a plastic instrument. Chemical cure composites are typically applied in bulk, while light-curing and dual-curing composites are applied and cured in 2 mm increments to allow for proper curing of composite and to minimize the shrinkage of the restoration (incremental buildup). Low viscosity materials in vertical restorations can be held in place with a mylar strip (Fig. 21.9).
6. The cavity is filled to the coronal margin, or slightly overfilled.
7. The restoration surface is contoured with a diamond finishing burr on a high-speed water-cooled handpiece and then finished with finishing stones and discs on a low-speed hand piece (Fig. 21.10).
8. The restoration surface and marginal tissues are sealed by re-etching and applying two coats of bonding agent (Rebonding; Fig. 21.11).
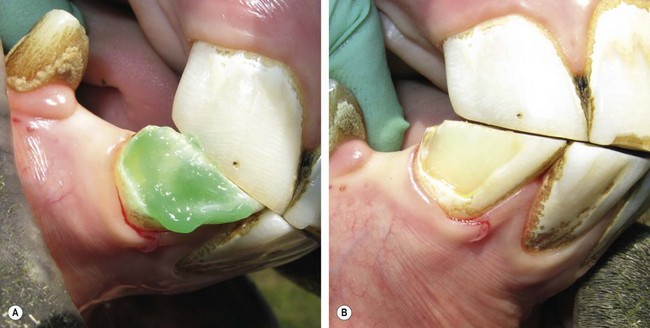
Fig. 21.8 Conditioning and bonding of the cavity. (A) The cavity preparation is conditioned with 37 % phosphoric acid, and then rinsed and left damp. (B) Two layers of an ‘Etch and Rinse’ bonding agent are applied to the cavity preparation. Note the shiny appearance of the bonded dentin.
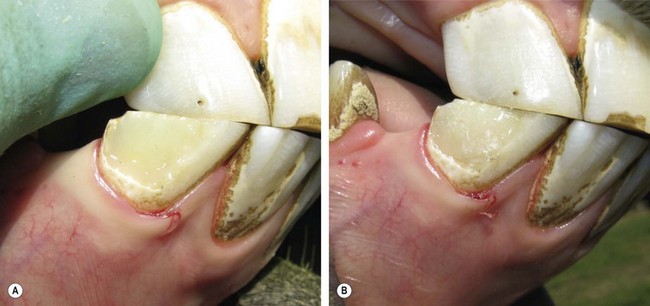
Fig. 21.9 Incremental filling of the cavity with a light-cured resin composite. (A) The first layers are placed and adapted to the walls of the cavity. (B) The remainder of the cavity is filled in 2-mm increments.
Glass ionomer cements
Glass ionomer (GI) cements are a group of materials based on the reaction of silicate glass powder and polyacrylic acid. GIs chemically bond to dentin and enamel by crystal formation when the GI’s carboxyl group chelates with the calcium in the apatite in dentin and enamel. Although this bond is not as strong as that formed by resin-based dentin bonding systems, the clinical retention of GIs in low-stress applications is excellent.18 Additional bond strength can be attained by conditioning the walls of the cavity with 34–37 % phosphoric acid or 10–20 % polyacrylic acid to remove the smear layer to allow for micromechanical bonding to the dentin and enamel.19 The chemical bonding of GIs allows for conservative cavity preparation and placement into moist fields, and GIs have shown clinical success when placed in incompletely debrided cavities (atraumatic restorative treatment).20 GIs have a modulus of elasticity similar to dentin and a coefficient of thermal expansion comparable to tooth structure, which minimizes marginal microleakage and thermal conduction. The unique property of GIs is the release of high levels of fluoride ions over the life of the restoration, which is known to strengthen enamel, decrease dentin sensitivity, and provide an antibacterial and cariostatic effect to the surrounding tissues. Finally, GIs are relatively biocompatible with pulp.
GIs also have several disadvantages that must be considered during treatment planning. Their use is limited to low-stress applications because low compression and flexural strength make them brittle and susceptible to fracture and cause poor wear resistance. GIs are technique sensitive during preparation and placement and must be mixed and applied exactly according to the manufacturer’s specifications for handling and working time. GIs should be manipulated as little as possible during the initial setting period, usually four to five minutes. GIs are often provided as a liquid and a powder which are mixed and applied as a tacky liquid, which lumps upon placement into a cavity. The consistency of the prepared material often necessitates the use of a mylar strip to hold the material in place during initial setting when the cavity is located on a vertical wall (i.e., a peripheral cavity on an incisor) and makes application into an occlusal maxillary cavity difficult (i.e., incisor root canal therapy restoration) or impossible (i.e., cheek tooth infundibulum). To improve handling sensitivity, some GIs are packaged in syringes that dispense a premeasured volume of two gels, which are then mixed or in capsules which are mixed with an amalgamator and dispensed directly into the cavity. Resin modified glass ionomers have also been developed to allow for instant light-cured initial setting of the material, as well as improving strength. The final disadvantage of GIs is their extended curing time (months), which necessitates protection of the restoration surface from desiccation. Protection during curing is commonly provided by the placement of an unfilled resin on the restoration surface and peripheral dental tissues (enamel and cementum in the horse; rebonding technique).
Because of the unique chemical bonding and fluoride releasing properties, GIs are formulated for many dental applications (Box 21.6, 21.7 and 21.8). Although GIs are very popular in human dentistry in luting applications (Fig. 21.12) and in restorative applications in patients with high risk for caries (Fig. 21.13), in veterinary medicine GIs are primarily used as liners under composite restorations to protect the pulp and to augment marginal sealing (Fig. 21.14). This application is commonly referred to as the ‘sandwich technique.’21 Before using a GI, practitioners must consider that the hypsodontic eruption of equine teeth might eventually put a restoration into an occlusal, load-bearing location, for which a GI is inappropriate.
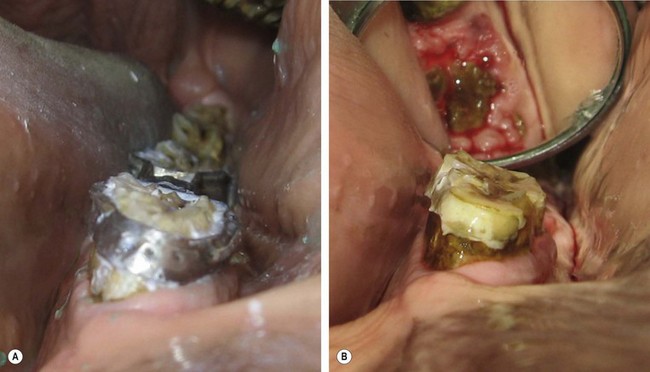
Fig. 21.12 Application of Type I glass ionomer cement. (A) A resin modified glass ionomer was used to cement an orthodontic appliance to the cheek teeth of a young horse. (B) After appliance failure, the glass ionomer cement remains bonded to the cheek tooth peripheral cementum.
(Bonding to cementum has not been scientifically evaluated.)
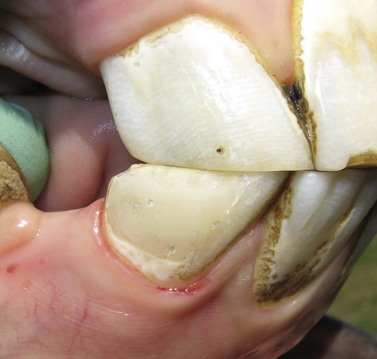
Fig. 21.13 Application of Type II glass ionomer (GI) cement restoration. GI restorations should only be applied after careful and cautious planning because of the hypsodontic eruption of equine teeth. (A) Tooth resorption on the vestibular aspect of the 3rd incisor of a senior horse. (B & C) Cavity preparation of the lesion involved osteoplasty and extensive subgingival debridement of all three dental tissues
(Modified Honma Stage 3 lesion).
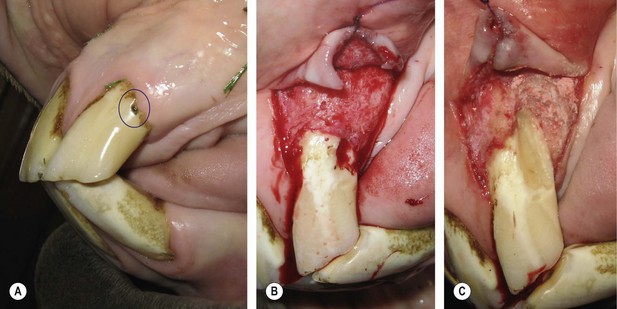
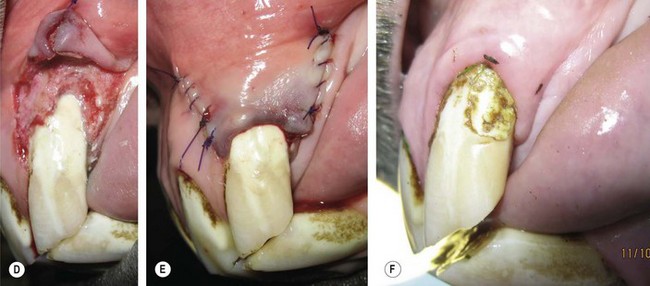
(D & E) A glass ionomer restoration was selected due to the uncertain etiology of the lesion and based on extrapolated applications in human and small animal patients. (F) 1-year follow-up demonstrates retention of the restorative with surface pitting and possible marginal leakage.
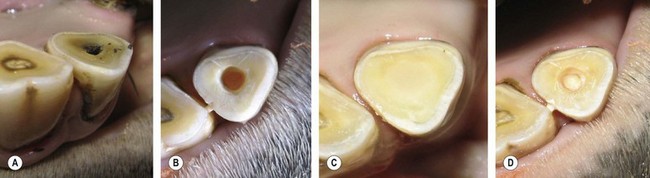
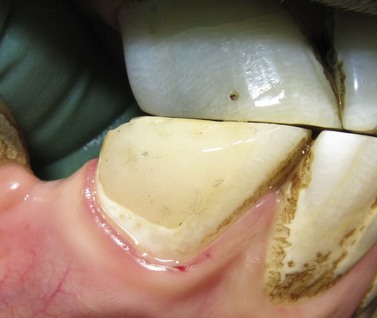
Fig. 21.14 Application of Type III glass ionomer cement (Sandwich Technique). (A) Non-vital pulp exposure of a mandibular 3rd incisor in a teenage horse. (B) Gutta percha (orange) and ZOE obturation of the root canal. (C) Glass ionomer liner application over the gutta percha. (D) The final resin composite restoration of the root canal access.
Box 21.8
The basic technique for glass ionomer restoration
1. The tooth and cavity preparation are cleaned with non-fluoride, flour pumice.
2. If the manufacturer recommends, or if increased bonding strength is required, condition the cavity with polyacrylic acid (acid etch technique).
3. Mix, or activate (encapsulated GI), exactly according to the manufacturer’s instruction. Remove one level scoop of powder and place it on a mixing pad. Divide the powder into three to four aliquots. Dispense the liquid next to the first aliquot and rapidly mix with a mixing spatula. Continue by drawing each aliquot into the liquid until the material is thoroughly mixed. The typical mixing time is approximately 30 seconds; however, mixing on a chilled surface extends the working time. The prepared material should have a uniform, tacky, glossy liquid consistency (Fig. 21.15).
4. Apply the GI to the restoration with a plastic instrument or a compule syringe. In vertical restorations, a mylar strip is usually required to hold the material in place. The initial setting time for GIs is approximately 4 minutes, during which time the material can be manipulated; however, overworking the material should be avoided.
5. The GI must be protected from contamination and drying during the initial setting period (about 20 minutes) by covering the restoration surface with a varnish or unfilled resin.
6. After the initial set, the restoration surface is contoured with a diamond finishing burr on a high-speed, water-cooled handpiece, and then finished with finishing stones and discs on a low-speed hand piece.
7. The restoration surface and marginal tissues are sealed by re-etching and applying a bonding agent (rebonding).
Endodontic materials
Endodontic irrigants
Endodontic therapy involves the preparation, sterilization, and obturation of a diseased pulp canal (see Ch. 22). Irrigation during root canal therapy is required to remove the smear layer of dentin shavings, cellular debris, and pulp remnants created during instrumentation and to disinfect the pulp canal. The chemical debridement provided by endodontic irrigants during equine root canal therapy is critical since the shape of equine pulp canals rarely allows for complete instrumentation. While the type and the concentration of endodontic irrigants are continuously debated, a SEM study concluded that the volume of the irrigant was the most important factor in removing debris from the canal.22
The effectiveness of all endodontic irrigants is limited by their ability to penetrate to the apex of the canal and into the dentin tubules; therefore, all irrigants should be replenished frequently to ensure effective chemical concentrations and removal of debris. Replenishment after each instrument change, or at least every two minutes, is commonly accepted. Ultrasonic activation of endodontic irrigants augments penetration into small pulp canal spaces23 and, theoretically, should improve irrigation of the irregularly-shaped equine pulp canals.24
Liquid irrigants are delivered into the pulp canal using a blunt needle on a syringe to prevent extrusion into the periapical tissue. Needles with a closed tip and a side port should be used when irrigating with caustic irrigants (see sodium hypochlorite). Irrigants are removed by suction, with a paper point, or by flushing with sterile saline solution. Compressed air should not be used to evacuate irrigants from the root canal since air may be extruded through the apex. Fatalities secondary to air embolism have been reported in a human25 and in dogs.26
The most commonly used irrigant is sodium hypochlorite (NaOCl). It has broad-spectrum antimicrobial efficacy and is a proteolytic solvent that dissolves the organic portion of the smear layer and the predentin layer of the dentin. Free Cl− ions are responsible for NaOCl’s antimicrobial effects. Proteolytic reactions deplete free Cl− ions; therefore, NaOCl must be replenished frequently to maintain chemical efficacy. Concentrations from 1 to 5.25 % (household bleach) have been shown to be clinically effective.27 At room temperature, a total contact time of 20–30 minutes is required to completely dissolve the pulp.28 However, heating increases both the tissue solvent and antimicrobial effects.29,30 The effervescence created by mixing sodium hypochlorite and hydrogen peroxide was once a popular flushing technique but this method has been shown to be ineffective.31 During conventional root canal therapy (coronal access), slow irrigation with light pressure through a non-binding or side-port endodontic needle is advisable since injection of NaOCl through the apical foramen into the periradicular tissue is extremely caustic and causes severe pain, periradicular hemorrhage, swelling, and possible abscess. During retrograde procedures, the periapical tissues should be protected and rinsed frequently with saline solution, and the endodontist should consider using a lower concentration of NaOCl. During procedures on maxillary incisors in the sedated horse, gravity evacuates the irrigant from the root canal. For this reason, higher concentrations of irrigant should be considered.
Ethylenediaminetetraacetic acid (EDTA) is a decalcifying agent that is commonly used to dissolve the inorganic component of the smear layer. EDTA also increases the diameter of the dentinal tubules to allow penetration of disinfectants. A 17 % solution is the most common endodontic concentration and removes the smear layer in less than one minute of contact time. Another popular formulation of EDTA adds urea peroxidase for its antibacterial properties in propylene glycol base (RC Prep).b Although the use of gel and paste formulations as a file lubricant is commonly practiced, the solvent efficacy of these preparations is questionable. Since the chemical efficacy of EDTA is self-limiting, it also must be intermittently replenished. For maximum clinical efficacy, EDTA should have a total contact time of at least 15 minutes.32 NaOCl and EDTA are most effective if their use is alternated.33
Chlorhexidine, 2 % solution, (CHX) is used as an endodontic disinfectant because of its antimicrobial properties, and like sodium hypochlorite, heating enhances this property;34 however, CHX has no tissue solvent properties. The use of CHX has had mixed acceptance. Some dentists irrigate with alternating flushes of NaOCl, EDTA, and CHX for increased disinfection, while others support the use of CHX as the final canal rinse before obturation because CHX binds to dental tissues and has persistent antimicrobial effect.35
When using multiple irrigants the root canal is typically rinsed with sterile saline solution between irrigants, and this is especially important when using CHX. CHX is incompatible with NaOCl and decomposes into a potentially carcinogenic precipitate, parachloroaniline,36 and when CHX is mixed with EDTA, a CHX/EDTA salt precipitates.37 The combination of NaOCl and EDTA inactivates the NaOCl, while the EDTA remains active for a few minutes.38
Intracanal medicaments
Since instrumentation and irrigation of non-vital pulps often leaves viable bacteria in the pulp canal, some endodontists perform staged root canal therapy (multiple visits) to ensure disinfection of the canal. Antimicrobial intracanal medicaments are used between treatments. Historically, volatile medicaments (i.e., formocresol and phenol derivatives) were used for their strong antibacterial properties, but these materials have lost popularity due to their potential toxicity.
Calcium hydroxide (CaOH) has both scientific and popular support in several endodontic applications. CaOH has traditionally been the material of choice for the treatment of exposed pulp tissue (direct pulp capping) and for apexification of non-vital pulp canals in immature teeth (Fig. 21.16) because of its biocompatibility and reparative dentin induction property; however, Mineral Trioxide Aggregate (discussed below) is rapidly replacing CaOH in these traditional applications. Because of its potent bactericidal properties, CaOH is also the material of choice for intracanal medicaments during staged (multiple visits) root canal therapy. In addition to the bactericidal effect of strong alkalinity, CaOH also hydrolyzes the lipid component of the lipopolysaccharides in bacterial cell walls.39 In equine dentistry, CaOH has been routinely used as a stand-alone pulp capping material after iatrogenic pulp exposures during occlusal equilibration. As discussed above (Bases and liners), this application is inappropriate.
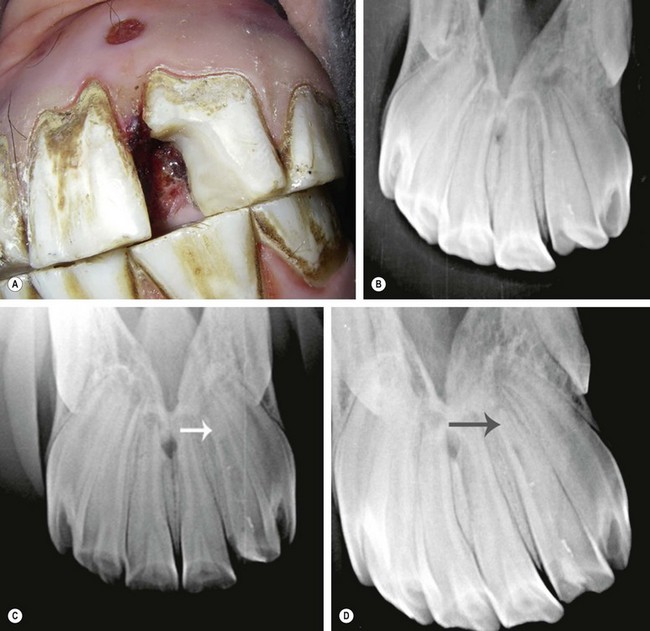
Fig. 21.16 Calcium hydroxide apexification of an equine incisor. (A) Traumatic crown fracture with pulp exposure of an immature 2nd maxillary incisor (202) in a 5-year-old horse. Note the mucosal draining tract. (B) Radiograph demonstrating an apical lucency associated with the open apex of 202. (C) At the five-month follow-up examination, the apex of the 2nd maxillary incisor has mineralized. (D) At the 1-year follow-up examination, the apical periodontium of 202 has re-attached, and root canal therapy was subsequently performed.
(Case by permission from Robert M. Baratt DVM.)
Obturation materials
Obturation is the complete filling and hermetic sealing of the prepared and sterilized root canal. Obturation systems typically require the combination of a solid obturation material and a sealer. Endodontic sealers bond to dentin to provide a hermetic seal, but excessive shrinkage during curing can cause seal failure; therefore, the solid obturation material provides a base for the sealer to minimize shrinkage. Solid obturation materials include gutta percha (GP), silver, and synthetic polymers. Compared to GP, silver has poor sealing properties, and corrosion produces cytotoxic salts. Consequently, the use of silver as an obturation material is below the current endodontic standard of care. A polyurethane based obturation material (Resilon)c is being used in both human and small animal dentistry in resin-based obturation systems. This material is non-toxic, nonmutagenic, and biocompatible, has superior coronal sealing compared to GP, and may strengthen the root.40 However, no clinical successes using resin-based obturation systems in the horse have been reported.
GP is the oldest, least cytotoxic, and most commonly used obturation material. It is also the only solid obturation material with reported use in equine endodontics. Natural rubber and GP are the cis and trans isomers, respectively, of the isoprene monomer, which is extracted from the juices of trees in the sapodilla family. Dental GP typically consists of approximately 20 % GP, 75 % zinc oxide, metallic sulfates for radio-opacity, and other waxes and resins. GP is supplied in cones of various shapes and lengths, but also in cones tapered to match standardized endodontic files. 60-mm lengths are appropriate for most equine application (Fig. 21.17). Most manufacturers package sterilized cones; however, the most common sterilization technique for GP is soaking the cone in 5.25 % sodium hypochlorite (NaOCl) for 1 minute.41 After NaOCl sterilization, the cone is typically rinsed with sterile saline solution. Some dentists believe NaOCl sterilized GP must be rinsed with ethyl alcohol to remove NaOCl crystals, which interfere with the obturation seal.42 GP oxidizes if exposed to air, light, and elevated temperatures; therefore, refrigeration is recommended for prolonged storage. GP is incompressible but can be compacted under pressure, and heating it to a temperature above 147° F (64° C) softens GP to facilitate mechanical packing.43 GP dissolves in organic solvents (e.g., chloroform, halothane, xylene), and a GP cone can be softened with a solvent to facilitate placement into irregularly shaped canals by dipping the apical 2–4 mm of the GP cone into the solvent for 1–6 seconds (chloroform dip technique).44 GP alone has no adhesive properties and cannot hermetically seal a canal. Endodontic sealer cements are always used in combination with the solid obturation material.
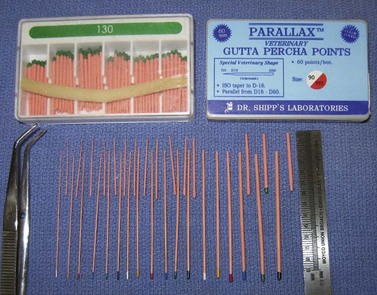
Fig. 21.17 Gutta percha, which is supplied in a variety of diameters and lengths. 30-mm and 60-mm lengths are commonly used in veterinary medicine, with the latter being more appropriate for equine applications.
Endodontic sealers are classified as zinc oxide-eugenol-based, calcium hydroxide-based, glass ionomer cements, and polymers. Calcium hydroxide-based sealers are promoted for their therapeutic effects, but have low adhesion to dentin, low water solubility, and have not passed scientific scrutiny. Glass ionomer endodontic cements have excellent biocompatibility and chemical bonding to dentin but are technique sensitive. Polymer sealers are very popular in small animal and human endodontics because of their handling characteristics and dentin bonding, but their use in equine endodontics is unreported. Zinc oxide-eugenol-based sealers are ZOE cements that have been modified with germicides, rosins, resin acids, and other chemicals for endodontic application. They have a long history of success, are the most commonly used sealers, and are the ‘gold standard’ for scientific comparison. They are mixed as previously described, and introduced into the pulp canal with a spiral filler on a low-speed handpiece or on an endodontic file. Additionally, each GP cone is usually coated with sealer before placement into the canal.
Thermaplasticized GP obturation systems are widely used in small animal and human endodontics, but no clinical successes in horses have been reported. The small size of the system instrumentation and the complex equine endodontic anatomy limit the application of the thermaplasticized systems in horses. A cold filling obturation system (GuttaFlow)d has been introduced, which might facilitate the filling of irregularly shaped equine pulp canals. The system combines particulate GP with a silicon-based sealer in a prepackaged capsule. After mixing with an amalgamator, the material is injected into the pulp canal, and has a 30 minute curing time before a restoration can be applied. The manufacturer claims that the product expands upon curing. However, this expansion is probably clinically insignificant. Research is needed to validate this product, but initial use in equine incisors and canine teeth has produced inconsistent results. (R Baratt, L Kimberlin, S Galloway, personal communication.)
Endodontic treatment of equine cheek teeth requires apicoectomy and retrograde obturation of the tooth. Historically, amalgam has been the preferred material for apical sealing, but it has lost popularity due to toxicity concerns and the availability of superior materials. Glass ionomer cements, resin composites, and reinforced ZOE cements (Super EBAe and IRM) have demonstrated better sealing properties than amalgam, while the latter is noted for less technique sensitivity.45 Mineral Trioxide Aggregate (MTA)f is an endodontic material that has recently shown superior performance in numerous endodontic applications, including pulp capping, orthograde apical closure (apexification), perforation repair, and retrograde root-end filling. MTA contains approximately 75 % Portland cement, and upon hydration with water or saline solution, forms a colloid gel that hardens over a 4-hour period. MTA can be placed in a wet environment and is not affected by blood contamination.46 The material properties of MTA have withstood extensive scientific testing. Compared to amalgam and reinforced ZOE cements, MTA showed significantly less marginal leakage47 and superior marginal adaptation,48 and a recent in vitro study showed that mixing MTA with chlorhexidine gluconate did not inhibit its sealing properties.49 MTA has a high alkalinity (pH 10.5–12) but is less cytotoxic than calcium hydroxide and induces the formation of a higher quality of and greater amount of reparative dentin than CaOH.50,51 Finally, MTA produces minimal periradicular tissue inflammation and is cementogenic.52–54 This unique final property establishes the ideal healing environment for the periodontium. In the horse, initial clinical successes have been reported using MTA as a retrograde filling material in a mandibular cheek tooth;55 however, this report lacks long-term follow-up.
Periodontal materials
Oral and periodontal irrigants
The treatment of periodontal disease requires the debridement of the periodontal pocket or, in advanced cases, periodontal surgery. Several dental materials are used to augment the effects of periodontal debridement. Pocket irrigation is an important procedure following periodontal pocket debridement to remove the loose debris created by the scaling and root planing procedures. Water, saline solution, and chlorhexidine are commonly used oral irrigants in both human and veterinary dentistry. Chlorhexidine has several advantages including: 1) adherence to dental tissues, oral soft tissues, and the pellicle, which prevents bacterial and plaque adherence; 2) a broad antimicrobial spectrum with no reported antimicrobial resistance; and 3) a residual antimicrobial effect.56 Chlorhexidine concentrations between 0.02 and 0.12 % have been shown in clinical trials to be effective oral irrigants.57 A 0.05 % solution has also been reported to be effective and non-toxic to tissues as a surgical wound irrigant,58 and a concentration of 0.12 % was shown to have synergistic antibacterial properties with locally administered tetracycline.59 However, if chlorhexidine is used as a surgical irrigant during periodontal surgery (i.e., guided tissue regeneration), it should be thoroughly rinsed from the periodontal pocket at the conclusion of the procedure because chlorhexidine can devitalize periodontal cells and interfere with reattachment to cementum.60
Local antibiotic administration (perioceutics) (Box 21.9 and 21.10)
Box 21.9
Application of a local antibiotic (perioceutic)
Doxirobe Gel is supplied in a premeasured (1 ml) two-syringe system. For the product to be effective, the periodontal pocket must be debrided and should be dried.
1. The product is mixed according to the labeled instructions using the two-syringe system.
2. A blunt needle is placed onto syringe A for delivery into the periodontal pocket.
3. The pocket is filled to the level of the gingival margin. Many stage 2 periodontal pockets in horses exceed the 1 ml prepackaged volume, and multiple syringes are often required for treatment.
4. Upon contact with crevicular fluid, the gel begins to polymerize. Water drops are typically administered to any exposed gel to accelerate the setting reaction.
5. A plastic instrument can be used to pack any escaping gel back into the periodontal pocket.
6. Covering Doxirobe with an impression material has been described; however, the authors believe that this step is unnecessary since properly placed gel is well retained. Premature dislodging of a local antibiotic administration is probably the result of poor handling technique.
In cases of Stage 2 periodontitis (up to 25 % attachment loss),61 local antibiotic administration (LAA) may be indicated as an ancillary treatment after completion of pocket debridement and irrigation. In human dentistry, tetracycline impregnated fibers and doxycycline, minocycline, and metronidazole gels are commercially prepared for LAA. A biodegradable, polymerized 8.5 % doxycycline gel (Doxirobe Gel)g is labeled for veterinary use in dogs and has been empirically used in other species, including horses (Fig. 21.18). Doxycycline has a broad spectrum of antibiotic activity against several known bacteria associated with periodontal disease, and the degradation of doxycycline gel provides localized, sustained release of antibiotic for approximately 2 weeks. Additionally, doxycycline binds to dentin, cementum, and bone for prolonged antibiotic release, inhibits the collagenase enzyme (an enzyme that slows the healing of the periodontal tissues), and stimulates fibroblast activity to re-establish the periodontium.62,63 The polymer gel also provides a physical barrier for reinfiltration of food and debris into the periodontal pocket, which is an important goal in the treatment of cheek tooth periodontal disease. Although the use of LAAs has become a popular practice in equine dentistry, this ancillary procedure has received no critical evaluation and, in the authors’ opinion, has limited indications in the equine patient. Other less expensive materials have shown clinical effectiveness as temporary barrier materials (e.g., impression materials, calcium sulfate, calcium alginate).
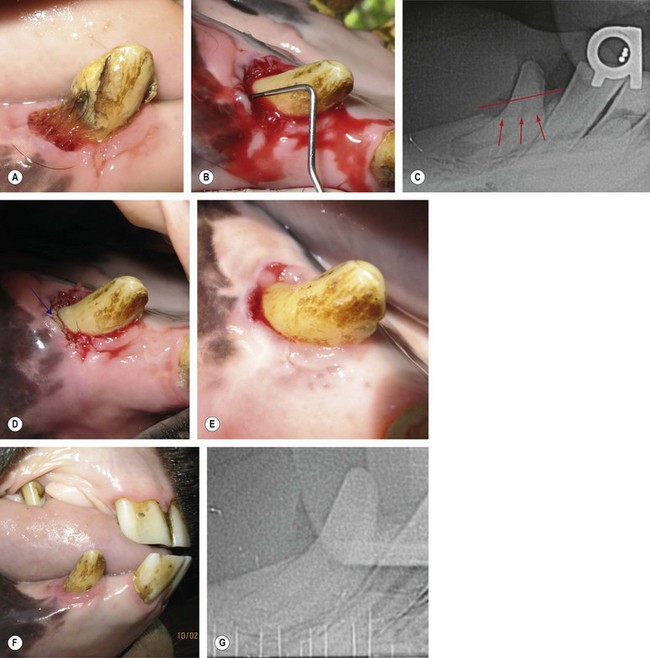
Fig. 21.18 Application of a local antibiotic (perioceutic). (A & B) A 12-mm periodontal pocket (PP) on the vestibulodistal aspect of the right mandibular canine tooth (404) of a young gelding. (C) Radiographs reveal horizontal bone loss of the associated alveolar bone (Stage 2 periodontitis). (D) The blue arrow shows the applied Doxirobe Gel in the PP. (Note that the perioceutic material fills the entire pocket.) (E) At the 2-week follow-up visit, the PP depth measured 4 mm. (F & G) At the 6-month follow-up visit, probing of the PP produced negligible depth and radiographs revealed alveolar bone regrowth.
Periodontal splinting
Box 21.10
The basic technique for fabricating a reinforced composite splint
1. Excessive peripheral cementum is removed from the crowns of the incisors to allow for enamel bonding. This can be accomplished with a whetstone on a low-speed handpiece or a diamond burr on a high-speed handpiece.
2. The teeth are aligned, and a tin foil template is made by molding the foil to the contours of the teeth in the desired application site. The template is then used to cut the reinforcing material to the appropriate length and width. The material should not be handled with bare hands.
3. The teeth are polished with a non-fluoride pumice paste and acid etched.
4. A bonding agent is applied to the teeth and light cured.
5. The mesh is coated with an unfilled resin, and the excess resin is blotted off.
6. A thin layer of filled composite is applied to the bonded surface of the teeth but is not cured.
7. The wetted mesh is applied to the splint site and contoured to the surface of the teeth with a plastic instrument. Excess composite material is removed.
8. The splint over each tooth is light cured. Curing the entire splint requires multiple curing increments.
9. An additional layer of a filled composite is added to the splint, contoured, and cured.
10. The final composite layer of the splint can be shaped or polished to avoid abrasion.
Periodontal splinting is a temporary adjunct appliance used in combination with aggressive periodontal therapy to stabilize diseased permanent teeth with mobility or to stabilize mobile teeth during healing after trauma.64 Splinting creates a stable platform for osseous regeneration by redistributing the forces applied to the diseased teeth to the adjacent healthy teeth. Before splinting, odontoplasty should be performed on the affected teeth to take them out of occlusion since excessive occlusal loading is a frequent cause of tooth mobility.65,66 Splinting materials include acrylics and resin composites used alone or reinforced with interdental wire or fiber mesh (Ribbond).h Since the accumulation of plaque and debris around the splint promotes periodontal disease, periodontal splinting is best suited to the incisors where the appliance can be cleaned daily (Fig. 21.19).
Bone grafting materials
In human and veterinary dentistry, bone grafts are used for guided tissue and bone regeneration (GTR, GBR) in the surgical treatment of advanced periodontal osseous lesions (stage 2–4 periodontitis, >25 % bone loss) and of selected periradicular (endodontic) lesions, and for bone augmentation in association with implant surgery. Although the filling of deep post-extraction alveoli with synthetic bone grafting material has been practiced in veterinary dentistry in order to increase the rate of bony healing and to preserve the alveolar ridge, this practice has limited scientific support and is a source of debate amongst veterinary dentists.
Bone grafting materials are classified by their source and by the type of bone growth that they promote. Material can be obtained from the same patient (autogenous bone graft), from a different patient of the same species (allograft), from a patient of a different species (xenograft), or from artificial or manufactured materials (synthetic grafts/alloplastic grafts). The bone growth potential of a graft material is described as osteogenic, osteoinductive, or osteoconductive. Osteogenic materials contain living osteoblasts that produce new bone within the graft itself. Osteoinductive materials possess bone morphogenic proteins (BMP) that induce the differentiation of osteoblasts in the recipient tissue (which does not have to be bone). These osteoblasts then produce new bone. Osteoconductive materials, when placed into bone, provide physically favorable scaffolding for osteoblasts from the recipient tissue to penetrate and form new bone.
Autogenous bone grafts are transferred from one site to another within the same patient. They can consist of cancellous or cortical bone and are the gold standard bone grafting material. These grafts are usually considered osteoinductive but are potentially osteogenic if tissue remains vital. They rapidly revascularize and lack antigenicity. The collection of autogenous bone graft is also associated with the inherent complications and expense of general anesthesia and significant morbidity. Therefore, the other classifications of bone grafting materials have been developed.
Allografts are typically collected from cadavers. Although not routinely used in veterinary medicine, in human medicine, grafts are collected, commercially prepared, and banked for future use. Preparation of the graft degrades the tissue’s BMPs; therefore, allografts are osteoconductive. The disadvantages of the materials are antigenicity and the potential for disease transmission.
Xenografts are prepared materials of bovine origin and have had extensive clinical use in human periodontics. Like allografts, they are osteoconductive and have the potential for antigenicity and disease transmission.
Several alloplastic (synthetic grafts) materials have been investigated for use in periodontal surgery. All are inert and osteoconductive, with the exception of Pepgen P-15,i a synthetic amino acid sequence (P-15) mixed with a calcium-phosphate matrix, which has demonstrated osteoinductive potential.67 The use of this product has not been reported in an equine patient.
A synthetic, bioactive ceramic derived from calcium salts, phosphates, and silica is labeled for veterinary use ‘in infraboney pockets caused by periodontal disease, endodontic-periodontic lesions, traumatic defects, or intraosseous flaws’(Consil Bioglass)j. The granular material is coated with hydroxylcarbonate apatite (Fig. 21.20) and, when placed in contact with tissue fluids, incorporates ground proteins and attracts osteoblasts.68 This bioactive ceramic is mechanically hemostatic69 and has bacteriostatic properties secondary to its high pH.70 The material is easy to use and can be prepared by mixing with sterile saline solution, sterile water, or the patient’s blood to form a wet sand consistency before placing the material into the host site, or the material can be placed directly into the host site for incorporation with blood.

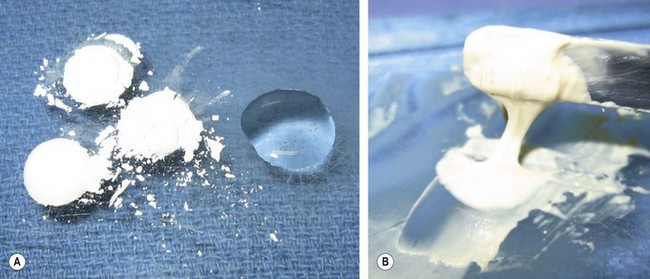
Fig. 21.20 Alloplastic (synthetic) bone graft materials. A granular synthetic, bioactive ceramic (Consil) on the left and powdered β-calcium sulfate hemihydrate (plaster of Paris) on the right.
Due to the large size of the bony defects associated with periodontal disease and dental extraction in the horse, many practitioners use calcium sulfate (plaster of Paris, Dental Stone) as an osteoconductive bone grafting material (Fig. 21.21). Calcium sulfate has been used historically in both human and veterinary medicine in dental and orthopedic applications, and has shown significant regeneration of bone and cementum in the dog.71 Calcium sulfate is readily available, inexpensive, and biocompatible. The material is supplied as a powder (calcium sulfate hemihydrate), which when rehydrated with water, sets into a solid form (calcium sulfate dehydrate, gypsum). The mixing/working time of the material is usually 2–5 minutes, but varies based on the specific product. The mixed material can be applied into a bony defect through a curved-tip syringe as a viscous liquid during the early phase of the setting reaction or later during the setting reaction by hand or with a dental spatula as putty. Once set, the material is porous enough to allow fluid exchange while dense enough to exclude epithelial and gingival connective tissue migration. The porosity of the material also provides for sustained local release of incorporated antibiotics, especially doxycycline, and for the localized release of calcium ions.72 Calcium sulfate resorbs completely over a 2-week period.73 Although calcium sulfate has been used as a bone graft material for over a hundred years, further controlled studies are needed to evaluate its efficacy.
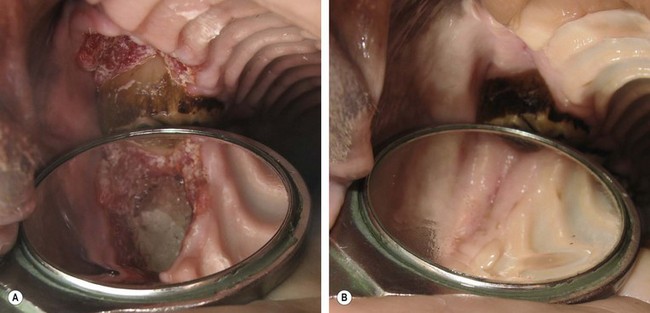
Fig. 21.21 Calcium sulfate used as an alloplastic graft in a post-extraction alveolus. (A) Calcium sulfate placed into the alveolus of the maxillary 1st cheek tooth (106) in a young mare. (B) Gingival healing at 6 weeks postoperatively.
In 2008, the first study reporting the use of bone grafting material in the mouth of a horse was published.74 A methylmethacrylate-based, non-resorbable bone substitute was placed into the post-extraction maxillary alveolus in five ponies. Although the material in this study demonstrated excellent biocompatibility and osteoconductivity, it performed inferiorly to the control alveoli with respect to bone mineral density and bone volume at the one-year follow-up evaluation.
Dental impression and cast materials (Box 21.11)
Box 21.11
The basic technique for making a dental impression with alginate
1. A rigid tray is designed so that the teeth and gingival margins are covered and so that 3–4 mm of material remains between the oral tissues and the tray. Overflow holes are drilled into the tray to allow material relief as the tray is placed.
2. The patient should be sedated to provide at least 10 minutes of access into the mouth without interference from the tongue or cheeks.
3. The mouth is thoroughly rinsed to remove feed material and to wet the teeth.
4. The impression tray is test-fitted.
5. The impression material is mixed according to the manufacturer’s directions using a rubber bowl and spatula and then is spatulated into the impression tray.
6. The tongue, lips, and cheeks are retracted and controlled.
7. The tray is placed onto the teeth and held in position until the impression material sets. Setting can be approximated by leaving a small amount of material in the mixing bowl and observing the setting reaction.
8. The impression is gently removed and inspected for accuracy.
9. At least two impressions, preferably 3 or 4, should be made. You will want study and backup models in addition to the two working models sent to the dental laboratory.
10. Impressions should be wrapped in a damp cloth and stored in an air-tight container until the casts are poured.
Dental impression materials are used to make accurate molds of the oral hard and soft tissues. The impression is the negative reproduction of the tissues. Casting materials are poured into the impression mold to fabricate the dental cast or model, the positive reproduction, of the oral tissues. Study models are used for treatment planning and for treatment documentation. Working models are used to fabricate orthodontic appliances and prosthodontic restorations. Two general categories of elastic impression materials are commonly used in veterinary dentistry: alginate hydrocolloids and elastomeric impression materials. Dental casts are molded from epoxy resin or gypsum. Typically, the impression is sent to a dental laboratory that fabricates both the cast and the prescribed appliance.
Elastomeric impression materials
Elastomeric impression materials (EIM) are primarily used for prosthodontic impressions. Four types of EIM are available: polysulfides, condensation silicones, addition silicones, and polyethers. These products are usually dispensed as two pastes or putties, which upon mixing begin to set into a firm, but elastic consistency. All EIM are set by catalyst-initiated polymerization and have been formulated to minimize shrinkage. The appropriate ratio of the pastes must be measured and spatulated in a manner that minimizes air entrapment to produce accurate impressions; therefore, many EIM are supplied in auto-mixing cartridges that accurately measure and mix the pastes, on demand, and facilitate delivery into the impression tray or into the mouth. EIM have a working time of 2.5–7 minutes and a set time of 3–10 minutes. The expense of EIM is rarely commensurate with the degree of accuracy required for equine impressions; however, some equine practitioners commonly use EIM as temporary bandages to prevent food contamination of extraction site alveoli and periodontal pockets. EIM adapt well to the oral hard tissue walls and can be removed more easily than rigid acrylics, such as polymethylmethacrylate (PPMA). Condensation silicones (silicone, silicone rubber) have significant limitations compared to the other EIM and have lost popularity. The clinical properties of the other EIM will be briefly discussed.
The addition silicones (AS) (polyvinyl siloxanes, polyvinyls, vinyls) are currently the most popular impression materials for fixed prosthetics (Fig. 21.22). AS impressions have excellent detail and remain dimensionally stable for weeks. They are hydrophobic, and moisture (e.g., saliva, blood) can significantly degrade impression accuracy; therefore, AS must be used in a dry field. AS are thermally sensitive, and the rate of cure is accelerated by heating and decelerated by cooling. Several materials can contaminate AS, retarding polymerization and creating unacceptable impressions; therefore, rinsing the mouth with 2 % chlorhexidine to remove contaminants before placing an AS impression is recommended. The most common contaminant is the sulfides in latex gloves; therefore, polyethelene gloves should be worn when handling AS. Other sources of contamination are recently placed restorative resins and the residual films left on the teeth by polyether and polysulfide impression materials.
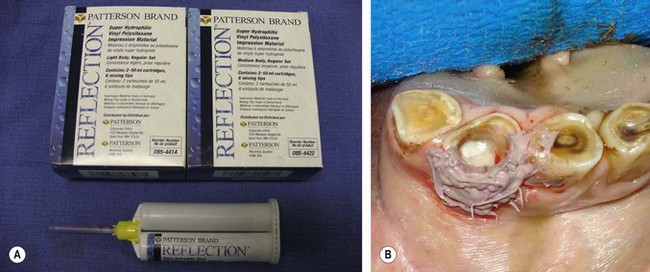
Fig. 21.22 Polyvinyl siloxane (PVS) is the most popular elastomeric impression material. (A) A generic material supplied in an auto-mixing cartridge. (B) PVS used as a mold for a resin composite restoration of a mandibular incisor.
(Courtesy of Edward T. Early, DVM.)
Polyethers (PE) are the second most popular EIM, and the preferred material for full bite registrations. They produce impressions of excellent detail and retain dimensional stability for one to two weeks. PE are hydrophilic and make accurate impressions in a moist environment. The ‘snap-set’ behavior of PE allows the material to flow into an area during the entire working time and then rapidly set.
Polysulfides (PS) are also used for full-bite registrations in human dentistry. They are relatively inexpensive and make accurate impressions in a moist environment; however, the dimensional stability of PS is inferior to that of both AS and PE. These materials are both temperature and moisture sensitive, and heat and humidity accelerate polymerization.
Alginate hydrocolloid impression materials
Alginate hydrocolloid impression materials (AHIM) are inexpensive and appropriate for dental impressions in equine patients. AHIM are dispensed as a powder containing soluble alginate (derived from marine plants), calcium sulfate dihydrate, and sodium phosphate. Upon mixing with water, alginic acid reacts with calcium sulfate to form an insoluble elastic gel. Sodium phosphate retards the reaction and provides for the working time of the material. Tap water with high mineral content also retards the setting time. Fast-setting alginates (Type 1) have a 1–2 minute working time and are appropriate for use in sedated equine patients. Regular setting alginates (Type 2) have a 2–4.5 minute working time. AHIM are highly hydrophilic and should be applied in a moist field. Drying or polishing the pellicle off the teeth may cause the alginate to stick to the teeth. AHIM are easily removed from the mouth; however, their dimensional stability is short, and usually only one cast can be made from each impression. Ideally, casts should be poured within 15 minutes of making the impression; however, casting can be delayed until returning to the laboratory if the impressions are wrapped in a damp cloth and stored in an air-tight container. Although AHIM lack the detailed accuracy and the auto-mixing systems of EIM, they produce impressions of acceptable accuracy for diagnostic bite registrations and orthodontic models, at a fraction of the cost of EIM. Only gypsum casting materials can be used with alginate impressions (Fig. 21.23).
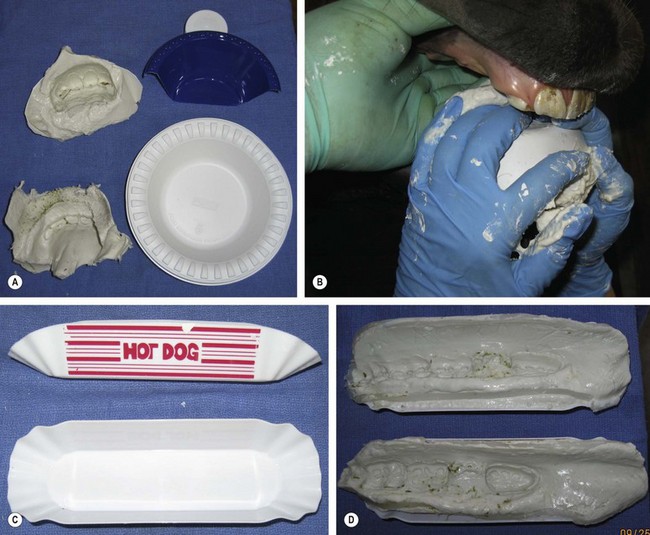
Fig. 21.23 Alginate hydrocolloid impression materials. (A) Improvised incisor impression trays (right) and alginate impressions (left). (B) Making an alginate impression of the mandibular incisors in a sedated horse. (C) Improvised cheek teeth impression trays. (D) The alginate impressions of the mandibular cheek teeth.
Cast materials
Dental plaster (plaster of Paris) and dental stone are used to fabricate dental casts from alginate impressions. Orthodontic casts are typically referred to as models. Both of these casting materials are made from gypsum (calcium sulfate dihydrate). The physical properties of the materials vary greatly based on the dehydration processes used to manufacture each material’s base powder, calcium sulfate hemihydrate. The crystals of dental plasters (β-calcium sulfate hemihydrate) are large, irregularly shaped, and porous, whereas the crystals of dental stone (α-calcium sulfate hemihydrate) are smaller, regularly shaped rods and prisms. Stone produces more detailed casts. However, both produce enough detail for orthodontic models and bite registrations in equine patients (Fig. 21.24).
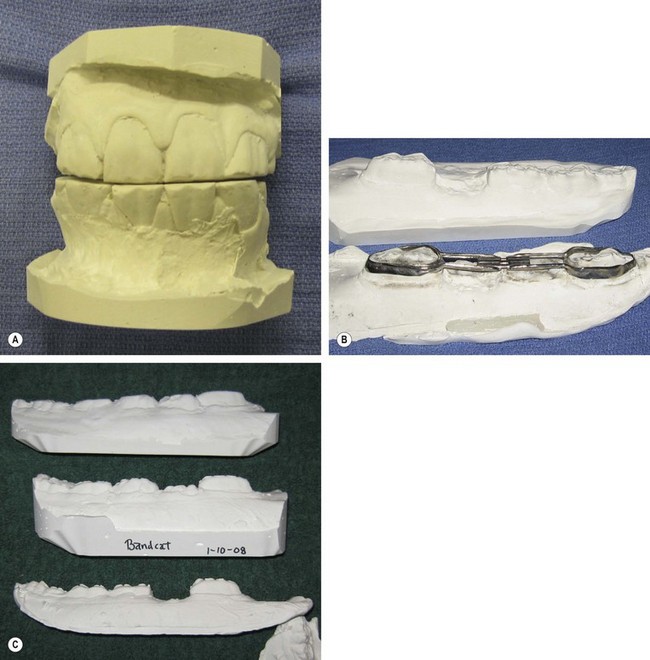
Fig. 21.24 Dental stone casts of the impressions from Fig. 21.23. (A) Casts of the incisors. (B) Working models of the mandibular cheek teeth used to construct an orthodontic appliance. (C) Study models of the mandibular cheek teeth used for treatment planning and for monitoring case progression.
Summary
Equine dentistry saw minimal change through most of the 20th century, with the disciplines of occlusal equilibration and exodontia being the standard of equine dental care. The resurgence of veterinary dental care in the 1990s stimulated practitioners to practice other dental disciplines (endodontics, orthodontics, periodontics, and restorative dentistry) in order to preserve the dentition of their patients. These disciplines require the application of dental materials.
The first decade of the 21st century has seen exponential changes in the practice of equine dentistry. Accepted dental procedures and material applications have been extrapolated from human and small animal veterinary dentistry for use in the equine patient, and anecdotal reports of success support the continuation of these practices. However, failures and the inappropriate application of dental materials demonstrate the need for scientific investigation. With continued case reporting by practitioners and clinical research by universities, our dental materials decisions will become evidence-based.
Acknowledgments
Robert M. Baratt DVM (Salem, CT) and Edward T. Early DVM (Williamsport, PA) for critical review of the manuscript and for photographs.
Anusavice KJ, Phillips’ Science of Dental Materials, 11th ed. Saunders, St. Louis, 2003.
Cohen S, Hargreaves KM. Pathways of the Pulp, 9th ed, St. Louis: CV Mosby, 2006.
Newman MG, Takei HH, Klokkevold PP, Carranza FA, Carranza’s Clinical Periodontology, 10th ed., Saunders Elsevier, St. Louis, 2006.
Powers JM, Sakaguchi RL. Craig’s Restorative Dental Materials, 12th ed, St. Louis: Mosby Elsevier, 2006.
Wiggs RB, Lobprise HB. Veterinary Dentistry: Principles and Practice. Philadelphia, PA: Lippincott-Raven, 1997.
Zardiackas LD, Dellinger TM, Livingston M. Dental Clinics of North America, Dental Materials. Philadelphia, PA: Saunders Elsevier, 2007.
1 Harvey CE, Emily PP. Small animal dentistry. St Louis: Mosby; 1993. pp 215–232
2 Wiggs RB, Lobprise HB. Glossary of terms. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1993:634.
3 Taney KG. Composite restoration of enamel defects. J Vet Dent. 2007;24(2):130–134.
4 Bellows J. Small animal dental equipment, materials and techniques. Ames, Iowa: Blackwell; 2004. p. 129
5 Harvey CE, Emily PP. Small animal dentistry. St Louis: Mosby Year Book; 1993. p. 235
6 Wiggs RB, Lobprise HB, Hefferren JJ. Basic materials and supplies. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1997:38.
7 St.John KR. Biocompatibility of dental materials. Dent Clin N Am. 2007;51(3):747–760.
8 Brannan RD. Restorative Materials in Equine Dentistry. Annual Vet Dental Forum, 2005, pp 135–139
9 Rawls HR, Esquivel-Upshaw J. Restorative Resins. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:399.
10 Kuijs RH, Fennis WM, Kreulen CM, et al. Does layering minimize shrinkage stresses in composite restorations? J Dent Res. 2003 Dec;82(12):967–971.
11 Chi HH. A posterior composite case utilizing the incremental and stratified layering technique. Oper Dent. 2006 Jul-Aug;31(4):512–516.
12 Rawls HR, Upshaw JE. Restorative resins. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:2003.
13 Klugh DO, Basil T, Brannan R. Infundibular decay in equine maxillary teeth. J Vet Dent. 2001;18(1):26–27. March
14 Earley ET. Restorative dentistry – applied restorative techniques and materials in the equine. In: Proceedings of the 22nd Annual Veterinary Dental Forum. Florida: Jacksonville; 2008:141–162.
15 Stangel I, Ellis TH, Sacher E. Adhesion to tooth structure mediated by contemporary bonding systems. Dent Clin N Am. 2007;51(3):677–694.
16 Miguez PA, Castro PS, Nunes MF, et al. Effect of acid-etching on the enamel bond of two self-etching systems. J Adhes Dent. 2003;5(2):107–112.
17 Pneumans M, Kanumilli P, DeMunck J, et al. Clinical effectiveness of contemporary adhesives: a systemic review of current clinical trials. Dent Mater. 2005;21(9):864–881.
18 Norling BK. Bonding. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:395.
19 Shen C. Dental Cements. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:476.
20 Shen C. Dental Cements. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:481.
21 Shen C. Dental Cements. In: Anusavice KJ, ed. Phillips’ Science of dental materials. 11th edn. St Louis: Saunders; 2003:484.
22 Baker NA, Eleazer PD, Averbach RE, et al. Scanning electron microscope study of the efficacy of various irrigation solutions. J Endod. 1975;1(4):127–135.
23 Himel VT, McSpadden JT, Goodis HE. Instruments, Materials, and Devices. In: Cohen S, Hargreaves KM. Pathways of the pulp. 9th edn. St Louis: CV Mosby; 2006:252.
24 Baratt RM. Equine incisor endodontic therapy. In: Proceedings of the 22nd Annual Veterinary Dental Forum. Florida: Jacksonville; 2008:113–123.
25 Rickles NH, Joshi BA. A death from air embolism during root canal therapy. JADA. 1963;67:39.
26 Michael AH, Miserendio LJ. Instruments and Materials. In: Cohen S, Burns RC. Pathways of the pulp. 4th edn. Washington, DC: CV Mosby; 1987:397–440.
27 Mentz TCF. The use of sodium hypochlorite as a general endodontic medicament. Int Endod J. 1982;15:132–136.
28 Ruddle CJ. Cleaning and shaping the root canal. In: Cohen S, Burns RC. Pathways of the pulp. 8th edn. St Louis: CV Mosby; 2002:258.
29 Cunningham WT, Balekjian AY. Effect of temperature on collagen-dissolving ability of sodium hypochlorite endodontic irrigant. Oral Surg Oral Med Oral Path. 1980;49:175.
30 Cunningham WT, Balekjian AY. Effect of temperature on the bactericidal action of sodium hypochlorite endodontic irrigant. Oral Surg Oral Med Oral Path. 1980;50:569.
31 Svec TA, Harrison JW. The effect of effervescence on debridement of the apical region of root canals in single-rooted teeth. J Endod. 1891;7:335–340.
32 Himel VT, McSpadden JT, Goodis HE. Instruments, materials, and devices. In: Cohen S, Hargreaves KM. Pathways of the pulp. 9th edn. St Louis: CV Mosby; 2006:260.
33 Peters OA, Peters CI. Cleaning and shaping the root canal. In: Cohen S, Hargreaves KM. Pathways of the pulp. 9th edn. St Louis: CV Mosby; 2006:346.
34 Evanov C, Liewehr FR, Buxton TB, Joyce AP. Antibacterial efficacy of calcium hydroxide and chlorhexidine irrigants at 37° and 46° C. J Endod. 2004;30:653.
35 Zehnder M. Root canal irrigants. J Endod. 2006;32(5):389–398.
36 Basrani BR, Manek S, Sodhi RN, et al. Interaction between sodium hypochlorite and chlorhexidine gluconate. J Endod. 2007;33:966–969.
37 Rasimick BJ, Nekich M, Hladek MM, et al. Interaction between chlorhexidine digluconate and EDTA. J Endod. 2008;34(12):1521–1523.
38 Grawehr M, Sener B, Waltimo T, Zehnder M. Interactions of ethylenediamine tetraacetic acid with sodium hypochlorite in aqueous solutions. Int Endod J. 2003;36:411–417.
39 Safavi KE, Nichols FC. Effect of calcium hydroxide on bacterial lipopolysaccharide. J Endod. 1993;19(2):76–78.
40 Peters OA, Peters C. Obturation of the cleaned and shaped canal. In: Cohen S, Hargreaves KM. Pathways of the pulp. 9th edn. St Louis: CV Mosby; 2006:372–375.
41 Senia ES, Marraro RV, Mitchell JL, et al. Rapid sterilization of gutta-purcha cones with 5.25 % sodium hypochlorite. J Endod. 1975;1:136.
42 Himel VT, McSpadden JT, Goodis HE. Instruments, materials, and devices. In: Cohen S, Hargreaves KM. Pathways of the pulp. 9th edn. St Louis: CV Mosby; 2006:263.
43 Goodman A, Schilder H, Aldrich W. The thermomechanical properties of gutta-percha. IV. A thermal profile of the warm gutta-percha packing procedure. Oral Surg Oral Med Oral Path. 1981;51:544.
44 Wiggs RB, Lobprise HB. Basic endodontic therapy. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1997:317.
45 Stabholz A, Friedman S, Abed J. Marginal adaptation of retrograde fillings and its correlation with sealability. J Endod. 1985;11:218–223.
46 Torabinejad M, Higa RK, McKendry DJ, Pitt Ford TR. Dye leakage of four root end filling materials: effects of blood contamination. J Endod. 1994;20:159.
47 Adamo HL, Buruiana R, Schertzer L, Boylan RJ. A comparison of MTA, Super-EBA, composite, and amalgam as root-end filling materials using a bacterial microleakage model. Int Endod J. 1999;32:197.
48 Torabinejad M, Wilder Smith P, Pitt Ford TR. Comparative investigation of marginal adaptation of mineral trioxide aggregate and other commonly used root end filling materials. J Endod. 1995;21:295–299.
49 Shahriar S, Saeed R, Hamid RY, et al. Sealing ability of white and gray mineral trioxide aggregate mixed with distilled water and 0.12 % chlorhexidine gluconate when used as root-end filling materials. J Endod. 2007;33:1429–1432.
50 Pitt Ford TR, Torabinejad M, Abedi HR, et al. Using mineral trioxide aggregate as a pulp-capping material. J Am Dent Assoc. 1996;127:1491–1494.
51 Faraco IM, Jr., Holland R. Response of the pulp of dogs to capping with mineral trioxide aggregate or a calcium hydroxide cement. Dent Traumatol. 2001;17:163–166.
52 Apaydin ES, Shabahang S, Torabinejad M. Hard tissue healing after application of fresh or set MTA as root-end filling material. J Endod. 2004;30:21.
53 Torabinejad M, Hong CU, Lee SJ, et al. Investigation of mineral trioxide aggregate for root-end filling in dogs. J Endod. 1995;21:603.
54 Torabinejad M, Pitt Ford TR, McKendry DJ, et al. Histological assessment of mineral trioxide aggregate as root-end filling in monkeys. J Endod. 1997:223–225.
55 Earley ET. Pulp disease in mandibular cheek teeth. In: Proceedings of the 20th Annual Veterinary Dental Forum, Portland, Oregon, 2006, pp 117–126
56 Bellows J. Small Animal Dental Equipment, Materials and Techniques. Iowa: Blackwell, Ames; 2004. p. 134
57 Jahn C. Supragingival and subgingival irrigation. In: Newman MG, Takei HH, Klokkevold PP, Carranza FA. Carranza’s Clinical periodontology. 10th edn. St Loius: Eslevier; 2006:842.
58 Stashak TS. Equine Wound Management. Baltimore: Williams and Wilkins; 1991. p. 116
59 Morrison SL, Cobb CM, Kazakos GM, et al. Root surface characteristics associated with root surface placement of monolithic tetracycline-impregnated fiber. J Periodont. 1992;63:137.
60 Wiggs RB, Lobprise HB. Periodontology. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1997:225.
61 Klugh DO. Equine periodontal disease. Clin Technique Equine Pract. 2005;4:135–147.
62 Bellows J. Small animal dental equipment, materials and techniques. Ames, Iowa: Blackwell; 2004. p. 136
63 Wiggs RB, Lobprise HB. Periodontology. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1997:224.
64 Vitale MC, Caprioglio C, Martignone A, et al. Combined technique with polyethylene fibers and composite resins in restoration of traumatized anterior teeth. Dent Traumatol. 2004;20:172–177.
65 Klugh DO. A review of equine periodontal disease. In Proceedings, AAEP Annual Convention, 2006; 52: 551–558
66 Spear FM, Cooney JP. Restorative interrelationship. In: Newman MG, Takei HH, Klokkevold PP, Carranza FA. Carranza’s clinical periodontology. 10th edn. St Louis: Elsevier; 2006:1065.
67 Precheur HV. Bone graft materials. In: Zardiackas LD, Dellinger TM, Livingston M, eds. Dent Clin N Am. 2007;51(3):734.
68 Jahn C. Supragingival and subgingival irrigation. In: Newman MG, Takei HH, Klokkevold PP, Carranza FA. Carranza’s Clinical periodontology. 10th edn. Elsevier: St Louis; 2006:982.
69 Deforge DH. Evaluation of Bioglass/PerioGlas (Consil) synthetic bone graft particulate in the dog and cat. J Vet Dent. 1997;14(4):141–145.
70 Allen I, Newman H, Wilson M. Antibacterial activity of particulate bioglass against supra- and subgingival bacteria. Biomaterials. 2001;22:1683–1687.
71 Kim CK, Kim HY, Chai JK, et al. Effect of a calcium sulfate implant with calcium sulfate barrier on periodontal healing in 3-wall intraboney defects in dogs. J Periodontol. 1998;69:982.
72 Wiggs RB, Lobprise HB. Basic materials and supplies. In: Wiggs RB, Lobprise HB. Veterinary dentistry: principles and practice. Philadelphia: Lippincott-Raven; 1997:38.
73 Carranza FA, Takei HH, Cochran DL. Reconstructive periodontal surgery. In: Newman MG, Takei HH, Klokkevold PP, Carranza FA. Carranza’s Clinical periodontology. 10th edn. St Louis: Elsevier; 2006:982.
74 Vlaminck L, Cnudde V, Pieters K, et al. Histologic and micro-computed tomographic evaluation of the osseointegration of a nonresorbable bone substitute in alveoli of ponies after tooth extraction. Am J Vet Res. 2008;69:604–610.
aCaulk IRM, DENTSPLY International Inc., York, PA, USA.
bRC Prep, Stone Pharmaceuticals, Philadelphia, PA, USA.
cResilon, Resilon Research, LLC.
dGuttaFlow, Coltène/Whaledent, Raiffeisenstraße 30, 89129 Langenau, Germany.
eSuper EBA, Harry J. Bosworth Company, Skokie, IL, USA.
fProRoot MTA, DENTSPLY Tulsa Dental Specialties, Tulsa, OK, USA.
gDoxirobe Gel, Pfizer Animal Health, Exton, PA, USA.
hPepgen P-15, DENTSPLY Friadent, Postfach 71 01 11, 68221 Mannheim, Germany.
jRibbond, Ribbond Inc., Seattle, WA, USA.
iConsil Dental Bioglass, Nutramax Laboratories Inc., Edgewood, MD, USA.