CHAPTER 22 Endodontic therapy
Introduction
Endodontics, from the Greek endo (inside) and odons (tooth), is a specialist sub-field of dentistry that deals with the treatment of the tooth pulp and the tissues surrounding the apex of a tooth. In contrast to exodontia, endodontic treatment is aimed at the preservation of teeth affected with pulp or apical infection. In man, dental caries and dental trauma are the most common causes of acute pulpitis.1 Because equine dentition is anatomically very different from human, canine or feline teeth,2–5 endodontic techniques that are well established in such brachydont species have to be critically evaluated before they can be applied in equine (hypsodont) teeth.
Anachoresis, fissure and gross idiopathic fractures; external traumatic fractures; extension of periodontal disease, and extension of infundibular caries have been described as the most common causes of apical infection in the horse,1,6,7 and the etiopathogenesis of these disorders is discussed more fully in Chapters 9 and 10. Direct exposure of pulp, or reduction of dentin so close to the pulp as to effectively expose it, can occur during equine dental procedures. Excessive heat production by power equipment during routine dental corrections or diastema widening can also cause thermal pulpitis or even pulp necrosis.8
Acute pulp exposure
As noted above, vital pulp can be exposed secondary to dental fractures9–11 or iatrogenically during dental procedures. Dental fractures are not always recognized by horse owners because some affected horses do not show obvious signs of dental pain. Whether the equine species is less susceptible to dental pain or the suppression of pain is a simple phylogenetic necessity in horses, in order not to stand out as prey, remains unclear. Due to absence of signs of dental pain in some horses, idiopathic fractures with pulp exposure are often not detected or treated immediately, and some fractured teeth spontaneously heal by deposition of tertiary dentin on the occlusal aspect of the exposed pulps, as discussed in Chapter 10. Any deficits in this natural repair mechanism result in extension of the pulpitis with probable development of apical abscess formation, or pulp necrosis. Only a small percentage of horses with idiopathic fractures develop acute masticatory problems, fever, or swelling of the supporting bones. When recent dental fractures are diagnosed, the resultant pulp exposure can be managed as described below. At present, dental extraction is still the therapy of choice in the majority of sub-acute or chronic apically infected cheek teeth.12 In selected cases, affected teeth may be preserved with endodontic techniques, such as vital pulpotomy, where a sufficiently viable pulp is saved, or alternatively, by apical resection with pulpectomy (extirpation of pulp).1,12,13
As noted above, acute pulp exposure can occur accidentally during routine dental procedures, in particular during the reduction of incisor overgrowths, including mandibular or maxillary pro- or brachygnathism overgrowths, ‘slantmouth’ (diagonal bite), and during reduction of canines (Fig. 22.1) or cheek teeth overgrowths (‘hooks’, ‘ramps’, ‘tall teeth’, wave- and step-mouth formation). Such iatrogenic pulp exposure actually occurs more often than is currently assumed.1 It has been claimed that pulp exposure under such conditions does not require treatment because the normal deposition of reparative dentin is adequate, and no complications result from pulp exposure in equine teeth. These suppositions are due largely to lack of observation.1 A recent study found the depth of secondary dentin in equine cheek teeth to be highly variable, ranging from 2.5 to 15 mm, and consequently the distance from the occlusal surface to the vital pulp is unpredictable in normal teeth.14
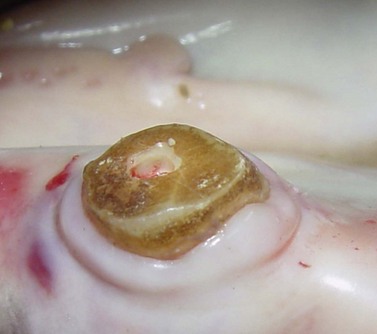
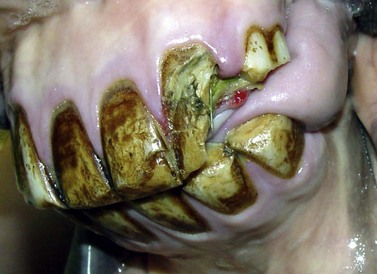
Fig. 22.1 An open pulp cavity in a left lower canine (Triadan 304) caused by excessive reduction of the tooth, following debridement and hemorrhage control.
The consequence for the equine dental practitioner is that pulp exposure may be detected at any time, especially when performing dental procedures. It is desirable that some equine veterinarians have a theoretical knowledge of endodontics as well as the practical skills and adequate equipment to enable satisfactory management of pulpar exposure.
Diagnosis
The diagnosis of iatrogenic pulp exposure can be difficult in some cases, especially when it occurs in caudal cheek teeth. Nevertheless, the detection and adequate management of iatrogenic pulp exposure are the responsibility of all equine dental practitioners. A final examination at the end of any dental correction using a mirror or an endoscope15,16 is mandatory to detect iatrogenically exposed pulp cavities. Bleeding from freshly opened pulp cavities may subside quickly, especially in older animals with narrow pulp horns. Consequently, major dental reductions should be performed slowly in stages, interrupted by examinations of the occlusal surface with a mirror or endoscope. The use of power equipment for dental correction creates a dough-like, viscous layer of wet dental dust which can temporarily occlude pulp cavities, thus masking pulpar exposure. Consequently, dental debris should be flushed from the occlusal surface before examining the occlusal surface.
Management of pulpar exposure
In the case of dental fractures, X-rays should be taken to evaluate the full extent of the fracture, including assessment of the supporting bones. If the extent of the fracture does not preclude dental preservation, preparations for endodontic surgery, (that can be performed in the standing, sedated horse) can proceed. Local analgesia via blocking of the ipsilateral maxillary or mandibular nerve significantly facilitates these endodontic procedures.17 General anesthesia is only required in a minority of cases, including those occasions when adequate local analgesia cannot be obtained.
Initially, loose dental fragments are removed from fractured teeth (Fig. 22.2), and then 2–3 mm of the clinical crown should be removed if the affected tooth is still in occlusal contact, to avoid occlusal pressure from the opposing teeth for a minimum of 2–3 months.1 Bleeding resulting from these procedures can be controlled using locally applied hemostyptic drugs (e.g., adrenaline) or electrosurgical devices.
A fine, sterile dental pick should now be used to gently probe the open pulp cavity. Bleeding indicates the likely presence of vital (may be inflamed or infected beyond redemption) pulp tissue and thus a chance of preserving the tooth. In order to avoid additional trauma to the pulp, the probe is then retracted, and the distance between the occlusal surface of the fracture and the underlying viable pulp is measured. High-speed dental burrs or spoon excavators should now be used to enlarge the pulp cavity1 in order to create better access to the diseased pulp. Simultaneously, contaminated dentin lining the walls of the pulp chambers is removed with a dental burr. This procedure can be difficult in curved incisor root canals. In this instance, the pulp cavity can be accessed from an opening in the intact labial aspect of the clinical crown, which enables a straight drill plane path to be achieved.12
The tooth should be thoroughly disinfected, and the burr should be sharp and clean (new – preferably sterile). When the surgeon is drilling into an exposed pulp, bleeding indicates that the vital pulp has been reached, and drilling should be extended for some additional millimeters if possible to remove any adjacent diseased pulp.
The exposed pulp canal should be shaped like an inverse cone (undercut) close to the occlusal surface to prevent later loss of filling material. The exposed canal is then carefully cleaned using Ringer’s solution and sterile paper points. Clean (preferably sterile) compressed air can also be used to dry the pulp canal, but drying of the pulp must be avoided. Hemostasis is subsequently performed using small cotton pellets or paper points soaked in adrenaline (Fig. 22.3). A couple of minutes after the bleeding has stopped, the pellets are removed carefully, and the pulp can now be capped with calcium hydroxide or similar materials. Calcium hydroxide (Ca(OH)2), which is used in paste form (calcium hydroxide and sterile water) in this situation, has a strong anti-microbial effect18 (due to its basic pH) and also acts as a tertiary dentin stimulant. It is preferable to cover softer, water-based calcium hydroxide with resin-containing calcium hydroxide such as Dycal, but it is inadvisable to place resin-containing preparations directly on to pulp. In human dentistry, other products, such as MTA (mineral trioxide aggregate) are used.19 MTA is a biocompatible endodontic cement that is also capable of stimulating healing and dentogenesis and sets in the presence of moisture.20
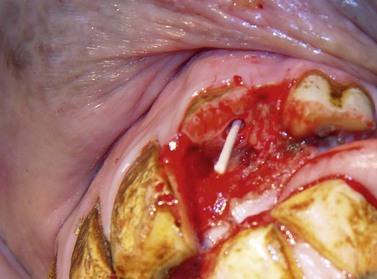
Fig. 22.3 Same horse as in Fig. 22.2. The fracture fragments and debris have been removed; the pulp canal of 202 has been drilled and curetted, and a paper point soaked in 0.8 % adrenaline solution is inserted into its pulp cavity for hemostasis. Filling with calcium hydroxide paste will now be performed.
In the horse, provided occlusal contact is avoided, additional restorative covers may not be necessary over the calcium hydroxide,1 especially if covered with a resin-containing calcium hydroxide preparation. Alternatively, the more occlusal endodontic calcium hydroxide cement can be removed, and a few millimeters of the root canal close to the masticatory surface can be sealed with glass ionomere21 or a resin-composite endodontic material. In show horses, incisors that are fractured at gingival level can be reconstructed using parapulpar pins and composite (Figs 22.4–22.7) to avoid protrusion of the tongue. The owners should be informed that the reconstructed crown has to be reduced at intervals to prevent occlusal contact with the opposite incisor; otherwise the artificial crown will inevitably break as human parapulpar pins and composite cannot withstand the forces of equine prehension. Owners should also be advised not to feed hay from nets, in order to reduce forces on the incisor restorations.
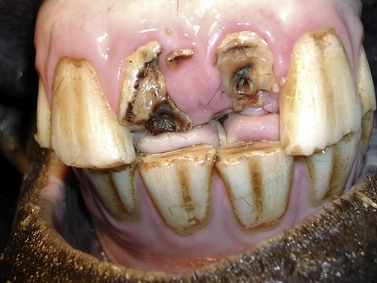
Fig. 22.4 Longstanding (note necrosis of circumpulpar dentin) traumatic fractures of 101 and 201 in a 7-year-old horse. The fractured teeth have been cleaned. An isolated fracture fragment lying between 101 and 102 has been left in situ in order to prevent excessive local hemorrhage prior to endodontic therapy.
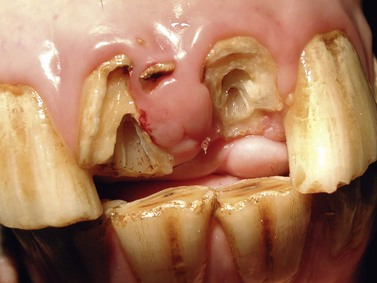
Fig. 22.5 Same horse as in Fig. 22.4. All visible infected and discolored dental tissues have been removed with a diamond burr. The pulp canals have also been debrided and pulpotomy has been performed. Calcium hydroxide paste will now be applied.
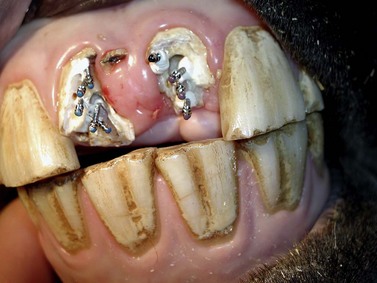
Fig. 22.6 Parapulpar pins have been placed into the dentin of the endodontically treated incisors to facilitate partial reconstruction of the crowns.
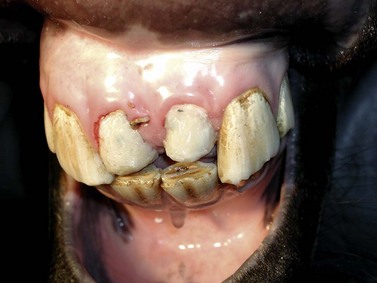
Fig. 22.7 Partial reconstruction of the crowns. Several layers of self curing composite have been attached to the parapulpar pins. The artificial crowns do not reach the occlusal surface of the opposing mandibular incisors. Finally the composite is polished and the dental fracture fragment is removed.
Endodontic procedures in apically infected cheek teeth
Oral approach
Whilst the technique described above can easily be performed in equine incisors, pulp canal treatment of infected cheek teeth using an intraoral approach is significantly more demanding due to difficulties in visualization and limited access to the equine oral cavity. Long-handled instruments and long-shafted, angled dental drills are required, as well as dental mirrors or, preferably, a 90° oral endoscope to visualize the surgical site. A skilled assistant is needed to direct the endoscope, if the procedure is performed under direct video control. If the procedure is performed by a single surgeon, the use of a dental burr has to be alternated with use of other instruments and/or visual control.
Although the endodontic treatment of cheek teeth using an intraoral approach is often discussed anecdotally at dental conferences, no scientific studies or objective long-term reports on the outcome of such attempts appear to have been published to date on this technique. Indeed, when one considers the length (up to 10 cm) which might be required to instrument an equine pulp canal, in relation to the space available when the mouth is fully open, such an approach is often likely to be impracticable.
Apical approach (apicoectomy)
Apicoectomy (radiculectomy) of equine cheek teeth has been described by several authors.1,13,22–25 Apicoectomy involves the resection of the tooth apex, followed by removal of the affected pulps and sealing of the pulp canal system to remove any possible communication between the oral cavity and the periapical tissues.25 Careful case selection of cases for such endodontic surgery is mandatory. A thorough oral examination (see Ch.12) in conjunction with high quality radiographs26 and/or computed tomography (Ch. 13) are required to identify suitable cases. Teeth showing signs of extensive periodontal disease, large fractures, evidence of dental decay, multiple pulpar exposure or long-standing apical infection (with subsequent tooth demineralization) are unsuitable for endodontic surgery.13 In such cases, dental extraction should be recommended.
The apicoectomy technique used by the author has been recently described.13 Surgery is usually performed under general anesthesia with the patient in lateral recumbency. To definitively identify the site of the affected apex, intra-operative radiography, using surface or sinus tract metallic markers should be performed. Access to the affected cheek teeth apices is gained, either via trephination27 for mandibular and rostral maxillary cheek teeth, or via a maxillary bone flap for more caudal maxillary cheek teeth. Bacteriological samples should be taken from the infected periapical regions to allow effective postoperative antimicrobial therapy. The infected apical region is then debrided, and infected tissue is removed using curettes. The surgical site can be obscured by hemorrhage, and even moderate bleeding significantly prolongs surgery and can compromise the quality of root canal sealing. Packing the hemorrhaging area with gauze or bone wax,25 the use of local vasoconstrictors, such as adrenaline solution (0.8 %), and continuous suction13 can keep the apical area blood-free during endodontic treatment.
Diamond burrs mounted on a sterile, high-speed dental drill are used to resect the apex of the affected tooth. Constant irrigation with sterile Ringer’s solution throughout the procedure is essential to prevent heat damage of adjacent dental tissues. The apex is cut at an angle of 15°–20° in a buccolingual (or buccopalatal) plane, so that the cut surface faces buccally (Fig. 22.8). All five (or six) pulp canals are then visualized and enlarged with a conical diamond burr. The contents of the pulp canals (necrotic, infected, or healthy pulps, or food material) are then removed as completely as possible using barbed broaches (Fig. 22.9). In contrast to vital pulps, which are easily removed in one piece, debridement of necrotic pulp debris and cleansing of infected pulp canals are frequently time-consuming and technically difficult. Nevertheless these procedures must be meticulously performed on all affected root canals. The empty canals are then filed with Hedstrøm files of ascending diameter to remove infected and carious circumpulpar dentin, and the pulp canals are alternately flushed with 2.5 % sodium hypochlorite and 3 % hydrogen peroxide solutions until no further debris or discolored dentin shavings are extracted from the canal (Fig. 22.10). The final pulp canal flush is performed with 70 % ethyl alcohol. The pulp canals are then dried using pressurized air and paper points (Fig. 22.11).
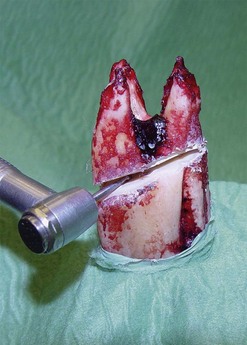
Fig. 22.8 Apicoectomy is demonstrated in the following figures using an extracted maxillary cheek tooth to allow better visualization. The apices are removed with a diamond-coated burr.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
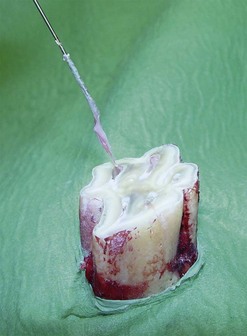
Fig. 22.9 The pulps are extracted using a barbed broach.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
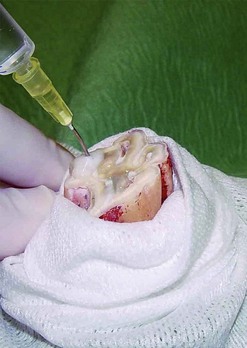
Fig. 22.10 The pulp horns are flushed with sodium hypochlorite.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
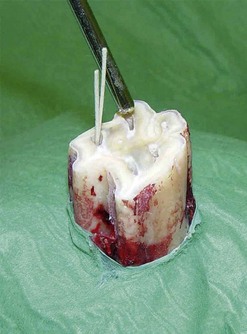
Fig. 22.11 The pulp canals are dried using paper points and compressed air.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
A variety of materials, such as human dental eugenol-based or eugenol-free cements, gutta percha or composite endodontic materials have been used for filling empty pulp canals13,20–24,28 (Fig. 22.12). The choice of endodontic filling material in equine teeth influences the long-term success of the procedure. Teeth on which apicoectomy has been performed continue to erupt (dental eruption is unaffected by endodontic procedures) and are consequently subjected to normal attrition. As all pulps have been removed during surgery, these teeth have lost the ability to produce secondary dentin which normally prevents occlusal pulpar exposure. Consequently, with continued eruption of the treated tooth, the endodontic filling material eventually appears at the occlusal surface after the remaining subocclusal secondary dentin (if present) is worn away. Human endodontic filling materials are not designed to withstand any abrasion whatsoever and so cannot withstand the abrasive forces of equine mastication and so are gradually lost when the material is occlusally exposed. Consequently, food material becomes compressed into the pulp canals which eventually can cause dental decay, secondary fractures, or apical re-infection. The use of filling materials with higher abrasive resistance, such as resin-composite endodontic materials, should lead to better long-term results. Such materials are not designed for obturation of pulp canals, so their performance when so used, especially in the high volume of the equine pulp canal, has yet to be established. In particular, shrinkage which breaks the initial seal may be a problem. The best choice of material for equine apicoectomy is as yet uncertain.
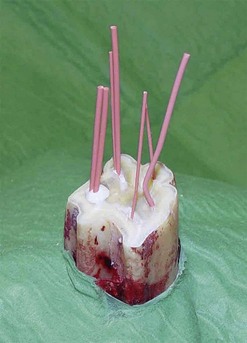
Fig. 22.12 Gutta percha points are used to compress endodontic cement deep into the pulp canals.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
Adequate sealing of the resected apex has also been reported to have a major influence on the outcome of equine apicoectomies.21,28 To prevent the loss or disintegration of the apical seal, it is strongly recommended that an undercut is made with diamond-coated burrs on the apical aspect of each root canal (Fig. 22.13). Apical sealing can then be performed with self-curing, glass ionomer cement, amalgam, MTA or, less satisfactorily, with a resin-based calcium hydroxide cement (Fig. 22.14). Postoperative radiographs should be taken at this stage (Fig. 22.15) to ensure adequate pulp canal sealing is present.29 The surgical wound is closed in routine fashion. Antibiotic and anti-inflammatory drugs are administered for 3–5 days postoperatively.
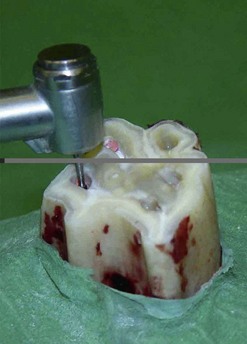
Fig. 22.13 An undercut is created in the apical aspects of the pulp canals using a diamond-tipped burr on a high-speed dental drill.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14

Fig. 22.14 Apical sealing is performed using glass ionomer cement.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J 2008; 178: 411–418. With courtesy of the editor.)14
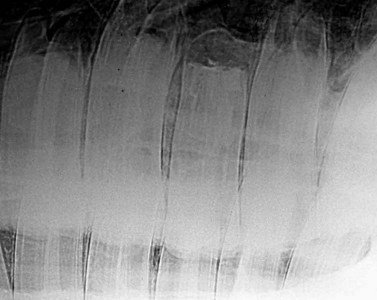
Fig. 22.15 Postoperative X-ray of a 5-year-old Quarterhorse gelding after apical resection and endodontic treatment of 109.
(Reproduced from Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. The Vet J 2008; 178: 411–418. With courtesy of the editor.)14
With careful case selection, success rates of about 80 % have been described.1,13 Reasons for failure of this technique include spread of infection from infected to unaffected pulps via communicating pulp canals, especially in young horses.30 No current diagnostic technique appears to enable a clear differentiation between infected and uninfected pulps prior to surgery, and so all pulp should be removed. In the future, magnetic resonance imaging may provide precise information on the health status of individual pulps.31
Despite the fact that equine endodontic surgery is a sophisticated, costly, and currently controversially technique, the successful outcome of a comparatively high percentage of cases treated to date warrants further objective long-term studies in vitro and in vivo to increase our knowledge of this potentially useful technique.
1 Baker GJ. Endodontic therapy. In: Baker GJ, Easley J. Equine dentistry. 2nd edn. Edinburgh: Elsevier; 2005:295–302.
2 Masson E, Hennet P, Calas P. Apical root canal anatomy in the dog. Endodontics and dental traumatology. 1992;8:109–112.
3 Rochette J. Identification of the endodontic system in carnassial and canine teeth in the dog. J Vet Dent. 1996;13:35–39.
4 Verstraete FJM, Terpak CH. Anatomical variations in the dentition of the domestic cat. J Vet Dent. 1997;14:137–140.
5 Hernández SZ, Negro VB, Maresca BM. Morphologic features of the root canal system of the maxillary fourth premolar and mandibular first molar in dogs. J Vet Dent. 2001;18:9–13.
6 Dacre I, Kempson S, Dixon PM. Pathological studies of cheek teeth apical infections in the horse part 4: aetiopathological findings in 41 apically infected mandibular cheek teeth. Vet J. 2008 Dec;178(3):341–351.
7 Dacre I, Kempson S, Dixon PM. Pathological studies of cheek teeth apical infections in the horse part 5: aetiopathological findings in 57 apically infected maxillary cheek teeth and histological and ultrastructural findings in apically infected cheek teeth. Vet J. 2008 Dec;178(3):352–363.
8 Allen ML, Baker GJ, Freeman DE, et al. In vitro study of heat production during power reduction of equine mandibular teeth. J Am Vet Med Assoc. 2004;224:1128–1132.
9 Dacre I, Kempson S, Dixon PM. Equine idiopathic cheek teeth fractures. Part 1 pathological studies on 35 fractured cheek teeth. Equine Vet J. 2007;39:310–318.
10 Taylor L, Dixon PM. Equine idiopathic cheek teeth fractures part 2: a practice-based survey of 147 affected horses in Britain and Ireland. Equine Vet J. 2007;39:322–326.
11 Dixon PM, Barakzai SZ, Collins NM, Yates J. Equine idiopathic cheek teeth fractures part 3: a hospital-based survey of 68 referred horses (1999–2005). Equine Vet J. 2007;39:327–332.
12 Garcia F, Sanromán F, Llorens MP. Endodontics in horses. An experimental study. Zentralblatt Veterinaermedizin. Reihe A. 1990;37:205–214.
13 Simhofer H, Stoian C, Zetner K. A long-term study of apicoectomy and endodontic treatment of apically infected cheek teeth in 12 horses. Vet J. 2008;178:411–418.
14 Wiegandt C. Veterinary thesis, Vienna, 2008
15 Tremaine H. Dental endoscopy in the horse. Clinical Techniques in Equine Practice 4, 2005, pp 181–187
16 Simhofer H, Griss R, Zetner K. The use of oral endoscopy for detection of cheek teeth abnormalities in 300 horses. Vet J. 2008 Dec;178(3):396–404.
17 Staszyk C, Bienert A, Bäumer W, et al. Simulation of local anaesthetic nerve block of the infraorbital nerve within the pterygopalatine fossa anatomical landmarks defined by computed tomography. Research in Veterinary Science. 2008;85:399–406.
18 Gomes BPFdA, Ferraz CCR, Vianna ME, et al. In vitro antimicrobial activity of calcium hydroxide pastes and their vehicles against selected microorganisms. Br Dent J. 2002;13:155–161.
19 Carrotte P. Endodontics Part 9. Calcium hydroxide, root resorption, endo-perio lesions. Br Dent J. 2004;197:735–743.
20 Tanomaru MJMG, Barros DB, Watanabe E, Ito IY. In vitro antimicrobial activity of endodontic sealers, MTA-based cements and Portland cement. J Oral Sci. 2007;49:41–45.
21 Pretorius S, van Heerden WF. The use of tricure glass ionomer cement as an apical sealant after apicoectomy. Journal of the South African Dental Association. 1995;50:360–370.
22 Van Foreest AW, Wiemer P. Veterinary dentistry. Apex resection in the horse. Tijdschrift voor diergeneeskunde. 1997;122:670–679.
23 Van den Bogaard A. Apex resection in horses. Tijdschrift voor diergeneeskunde. 1998;123:84–85.
24 Zetner K, Fahrenkrug P. Indikation, Technik und Prognose der Wurzelspitzenresektion im Pferdegebiss. Proceedings. Nürnberg: Jahreskongress des Bundesverbandes der praktischen Tierärzte; 1999.
25 Schramme MC, Boswell JC, Robinson J, et al, Endodontic therapy for periapical infection of cheek teeth a study of 19 horses. Proceedings of the 46th Annual Convention of the American Association of Equine Practitioners, 2000:113–116.
26 Barakzai SZ. Radiology and scintigraphy techniques and normal and abnormal findings. Proceedings Focus on Equine Dentistry. Indianapolis: American Association of Equine Practitioners; 2006. pp 168–172
27 Barakzai SZ, Kane-Smyth J, Lowles J, Townsend N. Trephination of the equine rostral maxillary sinus: efficacy and safety of two trephine sites. Veterinary Surgery. 2008;37:278–282.
28 Steenkamp G, Olivier-Carstens A, van Heerden WF, et al. In vitro comparison of three materials as apical sealants of equine premolar and molar teeth. Equine Vet J. 2005;37:133–136.
29 Carr G, Bentkover S. Surgical endodontics. In: Cohen S, Burns R. Pathways of the pulp. 7th edn. St Louis: Mosby; 1998:608–656.
30 Klugh D. Unpublished data, 2003. Cited by Dacre, 2003
31 Gerlach K. Die Darstellung von Pferdezähnen und benachbarter Strukturen im MRT einschließlich deren Entwicklung und Erkrankungen-derzeitiger Stand. Proceedings of the 6th Annual meeting of the IGFP 8–9 March, Frankfurt, Germany, 2008