15 Staphylococcus
Skin infections; osteomyelitis; bloodstream infection; food poisoning; foreign body infections; MRSA
Key points
• Staphylococci are commonly found on the skin of healthy individuals. Staph. aureus is present in the nose of 30% of healthy people but can cause infections where there is lowered host resistance (e.g. damaged skin).
• Many virulence factors have been described for Staph. aureus, but for most a specific role has not been determined. Exceptions include the enterotoxins, toxic shock syndrome toxin, the epidermolytic toxins and perhaps PVL, associated with community-acquired methicillin-resistant Staph. aureus (MRSA).
• Organisms spread from colonized sites (e.g. skin) by hands, clothing, dust and desquamation from the skin.
• MRSA is increasingly prevalent in hospitals and is emerging in the community and causes the same range of infections as methicillin-susceptible isolates.
• Flucloxacillin and vancomycin or teicoplanin are the agents of choice to treat methicillin-susceptible and methicillin-resistant Staph. aureus infections, respectively but newer options include linezolid and daptomycin.
• Coagulase-negative staphylococci (such as Staph. epidermidis) are major pathogens involving prosthetic implants such as intravascular lines or cardiac valves; the pathogenesis involves biofilm production.
• Device removal is usually required for the successful treatment of infections caused by CNS, as well as appropriate antibiotics, such as vancomycin or teicoplanin.
Sir Alexander Ogston, a Scottish surgeon, first showed in 1880 that a number of human pyogenic or pus-forming diseases were associated with a cluster-forming micro-organism. He introduced the name ‘staphylococcus’ (Greek: staphyle, bunch of grapes; kokkos, grain or berry), now used as the genus name for a group of facultatively anaerobic, catalase-positive, Gram-positive cocci. Staphylococci are resistant to dry conditions and high salt concentrations, and are well suited to their ecological niche which is the skin, but can survive for long periods in the environment. They may also be found as part of the normal flora of other sites such as the upper respiratory tract, and are commonly present on animals.
The major pathogen within the genus, Staphylococcus aureus, causes a wide range of major and minor infections in man and animals (Table 15.1) and is characterized by its ability to clot blood plasma by the action of the enzyme coagulase. There are at least 30 other species of staphylococci, all of which lack this enzyme. These coagulase-negative staphylococci (CNS) are skin commensals that can cause opportunistic infections especially associated with prostheses or foreign bodies (usually due to Staph. epidermidis), and urinary tract infections (Staph. saprophyticus). The presence of methicillin-resistant Staph. aureus (MRSA) in many hospitals and increasingly in the community has become a major public health issue, with concern expressed by patients and members of the public about the clinical implications.
Table 15.1 Infections caused by Staph. aureus
| Pyogenic infections | Toxin-mediated infections |
|---|---|
Staphylococcus aureus
Description
Staph. aureus is a Gram-positive coccus about 1 µm in diameter. The cocci are usually arranged in grape-like clusters (Fig. 15.1). The organisms are non-sporing, non-motile and usually non-capsulate. When grown on many types of agar for 24 h at 37°C, individual colonies are circular, 2–3 mm in diameter, with a smooth, shiny surface; colonies appear opaque and are often pigmented (golden-yellow, hence the ‘aureus’). The main distinctive diagnostic features of Staph. aureus are:
• Production of an extracellular enzyme, coagulase, which converts plasma fibrinogen into fibrin, aided by an activator present in plasma.
• Production of thermostable nucleases that break down DNA.
• Production of a surface-associated protein known as clumping factor or bound coagulase that reacts with fibrinogen.
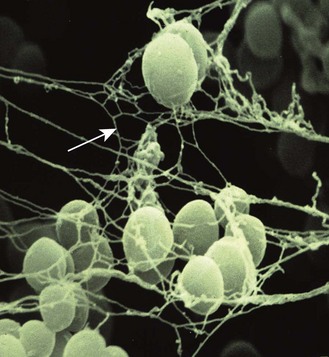
Fig. 15.1 Scanning electron micrograph of staphylococci in a biofilm enmeshed in exopolysaccharide (arrow). Original magnification ×15 000.
(Courtesy of Dr R Bayston, University of Nottingham.)
Various commercial systems, increasingly automated, are available that rapidly identify staphylococci. They are particularly useful for screening large numbers of strains.
Pathogenesis
Staph. aureus is present in the nose of 30% of healthy people and may be found on the skin. It causes infection most commonly at sites of lowered host resistance, such as damaged skin (e.g. surgical site infection) or mucous membranes (e.g. ventilator-associated pneumonia).
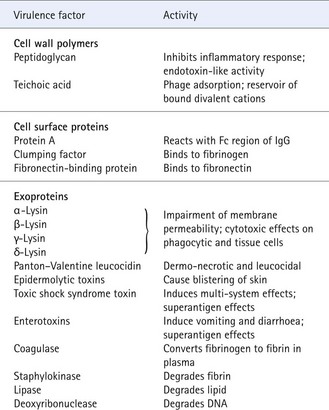
Virulence factors
Recent years have seen a greater understanding of the pathogenic interaction between the host and Staph. aureus. Most strains possess a large number of cell-associated and extracellular factors, some of which contribute to the ability of the organism to overcome the body’s defences and to invade, survive in and colonize the tissues (Table 15.2). Although the role of each factor is not fully understood individually, it is likely that they are responsible for the establishment of infection, enabling the organism to bind to connective tissue, opposing destruction by the bactericidal activities of humoral factors such as complement, and overcoming uptake and intracellular killing by phagocytes.
Table 15.2 Some virulence factors of Staph. aureus
Neutrophils are critical in the innate immune response and the primary cellular defence against Staph. aureus. A multi-step process results in the mobilization of neutrophils from peripheral blood and or the bone marrow in response to a variety of factors. The staphylococcal capsule can stimulate CD4+ T lymphocytes resulting in the production of chemokines that facilitate neutrophil mobilization and lipoteichoic acid in the cell wall interacts with toll-like receptors, which participate in this process. Subsequently, neutrophils use a variety of mechanisms to kill the ingested bacteria, e.g. superoxides and degranulation.
Staphylococcal toxins
Enterotoxins
Enterotoxins, types A–E, G, H, I and J, are commonly produced by up to 65% of strains of Staph. aureus, sometimes singly and sometimes in combination. These toxic proteins withstand exposure to 100°C for several minutes. When ingested as preformed toxins in contaminated food, microgram amounts of toxin can, within a few hours, induce the symptoms of staphylococcal food poisoning: nausea, vomiting and diarrhoea. However, enterotoxins, which are superantigens (see below) probably also play an important role in other serious staphylococcal infections, e.g. bloodstream infection (BSI), especially when accompanied by septic shock.
Toxic shock syndrome toxin (TSST-1)
This was discovered in the early 1980s as a result of epidemiological and microbiological investigations in the USA of toxic shock syndrome, a multi-system disease caused by staphylococcal TSST-1 or enterotoxin, or both. A link was established with the use of highly absorbent tampons in menstruating women, although non-menstrual cases are now as common. The absence of circulating antibodies to TSST-1 is a factor in the pathogenesis of this syndrome.
TSST-1 and the enterotoxins are now recognized as superantigens, that is, they are potent activators of T lymphocytes resulting in the liberation of cytokines such as tumour necrosis factor, and they bind with high affinity to mononuclear cells. These characteristics partly explain the florid and multi-system nature of the clinical conditions associated with these toxins.
Epidermolytic toxins
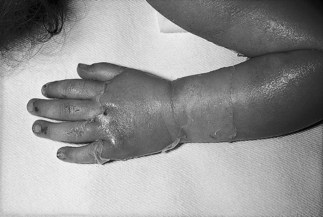
Two kinds of epidermolytic toxin (types A and B) are commonly produced by strains that cause blistering diseases. These toxins induce intraepidermal blisters at the granular cell layer. Such blisters range in severity from the trivial to the distended blisters of pemphigus neonatorum. The most dramatic manifestation of epidermolytic toxin is the scalded skin syndrome in small children, where the toxin spreads systemically in individuals who lack neutralizing antitoxin. Extensive areas of skin are affected, which, after the development of a painful rash, slough off; the skin surface resembles scalding (Fig. 15.2).
Panton-Valentine leukocidin (PVL)
This toxin was recognized some decades back but its potential contribution to the clinical manifestations and outcome have been increasingly described in the context of community-acquired MRSA (CA-MRSA). As the name suggests PVL can adversely affect cells, resulting in leucopenia, but animal studies do not suggest high virulence. Nonetheless, epidemiological data in many countries reveal an association between necrotizing pneumonia and some complicated skin and soft tissue infections (cSSTI) caused by PVL-positive strains of CA-MRSA.
Epidemiology
Sources and acquisition of infection
Infected lesions
Large numbers of staphylococci are disseminated in pus and dried exudate discharged from large infected wounds, burns and secondarily infected skin lesions, and in sputum coughed from the lung of patients with pneumonia. Direct contact is the most important mode of spread, but airborne dissemination may also occur. Cross-infection is an important method of spread of staphylococcal disease, particularly in hospitals, and scrupulous hand hygiene is essential in preventing spread. Food handlers may similarly introduce enterotoxin-producing food poisoning strains into food.
Healthy carriers
Staph. aureus grows harmlessly on the moist skin of the nostrils in about 30% of healthy persons, and the perineum is also commonly colonized. Organisms are spread from these sites into the environment by the hands, clothing, and dust consisting of skin squames and cloth fibres. Some carriers, called shedders, disseminate exceptionally large numbers of staphylococci.
During the first day or two of life most babies become colonized in the nose and skin by staphylococci, and transmission from babies to nursing mothers, who then develop mastitis, is well described.
Animals
Animals may disseminate Staph. aureus and so cause human infection, e.g. milk from a dairy cow with mastitis, causing staphylococcal food poisoning.
Environment
Although not spore forming, staphylococci may remain alive in a dormant state for several months when dried in pus, sputum, bed clothes or dust, or on inanimate surfaces such as floors. Environmental reservoirs are therefore increasingly recognized as important in hospitals in contributing to endemic MRSA. Staphylococci are fairly readily killed by heat (e.g. moist heat at 65°C for 30 min), by exposure to light and by common disinfectants, hence the emphasis on regular and effective environmental decontamination in controlling MRSA.
The acquisition of Staph. aureus infection may be exogenous (from an external source such as the environment) and more theoretically preventable and endogenous (from a carriage site, or minor lesion elsewhere in the patient’s own body). It is important to remember that the body surfaces of human beings and animals are the main reservoir.
Methicillin-resistant Staph. aureus (MRSA)
MRSA produces a penicillin binding protein 2a (mediated through the mecA gene), which is carried on the staphylococcal cassette chromosome mec (SCCmec) of which there are at least six different types recognized, and this results in resistance to all beta-lactam antibiotics. There is much debate on whether strains of MRSA are intrinsically more virulent than methicillin-susceptible isolates but it is agreed that MRSA causes the same range of infections resulting in excess healthcare costs, prolonged hospital stay and significant mortality. MRSA is endemic in hospitals globally except in Scandinavia and in the Netherlands although declining rates of MRSA BSI have been seen in recent years in the UK, France and other European countries. Vulnerable patients particularly at risk are those who have undergone major surgery and patients in the intensive care unit. Although 50–60% of patients with MRSA are merely colonized, i.e. representing asymptomatic carriage, serious infections occur such as BSI, respiratory tract and bone/joint infections. These infections are then more difficult to treat than infections caused by methicillin-susceptible isolates, and MRSA can spread easily among patients in hospital.
Community-acquired MRSA is increasing, especially in the USA, where 50% or more of Staph. aureus infections presenting to the Emergency Department may be methicillin-resistant. These occur often in otherwise healthy individuals with no recent healthcare contact. The production of PVL, alpha toxin and secreted proteases are implicated as particular virulence determinants. Community-acquired strains may cause the same range of infections as healthcare-associated strains such as BSI, but particularly cSSTI and severe pneumonia.
The control and prevention of MRSA involves the education of all healthcare professionals and the public, fast and reliable detection in the laboratory (including perhaps the use of molecular methods, see below), active surveillance (even universal surveillance), prompt patient isolation or cohorting when admitted to hospital, standard precautions and good professional practice by all health-care workers (including compliance with hand hygiene guidelines), effective hospital hygiene programmes and antibiotic stewardship programmes, e.g. avoidance of the excess use of cephalosporins and fluoroquinolones. Such measures have been very successful in Scandinavia and in the Netherlands where an aggressive ‘search and destroy’ approach involving the extensive screening of all MRSA contacts is employed.
Laboratory diagnosis
One or more of the following specimens should be collected to confirm a diagnosis:
• Pus from abscesses, wounds, burns, etc. is much preferred to swabs.
• Sputum from patients with pneumonia (e.g. postinfluenzal or ventilator-associated pneumonia); bronchoscopic specimens, e.g. bronchoscopic lavage, are increasingly used in critically ill patients.
• Faeces or vomit from patients with suspected food poisoning, or the remains of implicated foods.
• Blood from patients with suspected BSI such as septic shock, osteomyelitis or endocarditis.
• Mid-stream urine from patients with suspected cystitis or pyelonephritis.
• Anterior nasal and perineal swabs (moistened in saline or sterile water) from suspected carriers; nasal swabs should be rubbed in turn over the anterior walls of both nostrils.
The characteristic clusters of Gram-positive cocci can often be demonstrated by microscopy, and the organisms cultured readily on blood agar and most other media within 24 h or less. The tube or slide coagulase test is performed to distinguish Staph. aureus from coagulase-negative species and antimicrobial susceptibility testing with cefoxitin to confirm MRSA using standard methods. Commercially-available molecular methods using the polymerase chain reaction (PCR) have been developed to reduce the time to the detection of MRSA from 48–72 h with culture to less than 12 h to facilitate earlier preventative measures. However, the results from trials with PCR to date are mixed and do not clearly indicate that this more expensive approach can assist in reducing MRSA rates in acute hospitals.
Typing
Most staphylococcal infections are sporadic, but the identification of an outbreak strain, by determining whether all the isolates are of the same type, is an important aspect in the investigation of a source, particularly during outbreaks of MRSA. Traditionally, strains of Staph. aureus were differentiated into different phage types by observation of their pattern of susceptibility to lysis by a standard set of Staph. aureus bacteriophages (viruses that infect bacteria) with international agreement on the interpretation of results. However, many strains of MRSA have become non-typable with this method. Consequently, phage typing has been replaced by genotypic methods such as PCR, pulsed-field gel electrophoresis (PFGE) and gene sequencing (see Ch. 3). Multilocus gene sequence typing (MLST) defines the core genetic population structure but it has only moderate discriminatory power and is expensive. For local outbreak investigations, PFGE has been widely used and it is highly discriminatory but technically demanding. Assessing the gene encoding the staphylococcal surface protein A (spa typing) has become popular for typing MRSA and finally typing the SCCmec elements is used for the study of international MRSA transmission and evolution.
Treatment
Susceptibility to antibiotics
Staph. aureus and other staphylococci are inherently susceptible to many antimicrobial agents (Table 15.3). About 90% of strains found in hospitals are now resistant to benzylpenicillin due to the production of the enzyme penicillinase, a β-lactamase that opens the β-lactam ring. Methicillin (previously used for laboratory testing and initially when first produced for therapy), oxacillin, cloxacillin and flucloxacillin, are stable to the enzyme. Cephalosporins and β-lactamase inhibitors are also stable to penicillinase (see Ch. 5).
Table 15.3 Antibiotics and staphylococci
| Active agents | Agents lacking useful activity |
|---|---|
a Resistance common (see text).
b Usually used in combination, for example with flucloxacillin.
c For categorization of quinolones, see Table 5.3 and associated text.
MRSA strains are resistant to all β-lactam agents, and often to other agents such as the aminoglycosides and fluoroquinolones. Glycopeptides (vancomycin or teicoplanin) are the agents of choice in the treatment of systemic infection with MRSA, but these agents are relatively expensive and may be toxic. Isolates of MRSA with reduced susceptibility or full resistance to glycopeptide antibiotics are uncommon, but have been detected sporadically. These isolates have either thickened cell walls (reduced susceptibility) or the vanA gene (fully resistant), and can be difficult to detect in the routine diagnostic laboratory.
Choice of antibiotic for therapy
Pending receipt of susceptibility test results, the treatment of severe infections suspected to be caused by Staph. aureus should be started with flucloxacillin unless MRSA is endemic locally, in which case a glycopeptide such as vancomycin is indicated. Erythromycin, clindamycin or vancomycin (or teicoplanin) is indicated if the patient is allergic to penicillin. Fusidic acid and rifampicin are not used alone in serious infections, because mutation to resistance arises readily. It is usually necessary to remove an infected source, such as a central intravascular catheter or device, e.g. artificial hip joint, or drain an abscess as part of the treatment.
Other agents have emerged in the last decade and provide alternatives for the treatment of MRSA and can also be used to treat infections caused by isolates with reduced susceptibility to the glycopeptides. These include linezolid, which can be administered parenterally and orally and daptomycin which is bactericidal and is used to treat MRSA BSI. New agents being assessed are glycopeptide derivatives such as telavancin and anti-MRSA beta-lactams such as ceftobiprole.
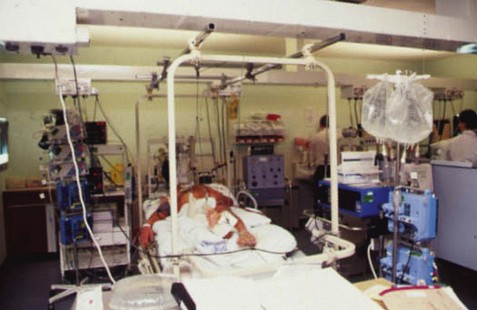
Life-threatening toxin-mediated disease, such as toxic shock syndrome, requires major medical support such as intravenous fluids to prevent multi-organ failure, often best provided in the intensive care unit (Fig. 15.3).
Coagulase-negative staphylococci
Coagulase-negative staphylococci comprise a large group of related species commonly found on the surface of healthy persons, in whom they are rarely the cause of infection. More than 40 species are recognized. Staph. epidermidis accounts for about 75% of all clinical isolates, probably reflecting its preponderance on the normal skin. Other important CNS include Staph. saprophyticus (a cause of urinary infection in young women) and Staph. lugdunensis (may cause severe infections like Staph. aureus). The emergence of CNS as major pathogens reflects the increased use of implants such as intravascular lines and cannulae, cardiac valves, artificial joints, etc and the increasing numbers of severely debilitated patients in hospitals.
Description
Coagulase-negative staphylococci are morphologically similar to Staph. aureus but they do not coagulate plasma and they lack clumping factor and deoxyribonuclease. Because Staph. epidermidis and other CNS may contaminate clinical specimens, care has to be exercised in assessing its significance, especially from superficial sites. When isolated from sites such as blood or cerebrospinal fluid, further specimens should be obtained to confirm its clinical significance.
Coagulase-negative staphylococci are opportunistic pathogens that cause infection in debilitated or compromised patients such as premature neonates and oncology patients, often by colonizing biomedical devices such as intravascular lines. They cause particular problems in:
Pathogenesis
Adherence to the prosthetic device by the production of an exopolysaccharide intercellular adhesion (PIA) is a key step in the formation of a multi-layered biofilm, essential for the pathogenesis of device-related Staph. epidermidis infection. A complex array of inter-related chemical messengers controls expression of polysaccharide and drives intercellular adhesion and biofilm formation. Physiological changes in the biofilm protect Stap. epidermidis from the host immune defence system and restricted penetration, decreased growth rates and persistent bacterial cells as part of the biofilm often render antibiotic treatment unsuccessful. Consequently, there is considerable interest in the development of antimicrobial-impregnated devices such as central intravascular catheters or the use of materials that are less prone to adherence by Staph. epidermidis and subsequent biofilm formation. Important factors in the pathogenesis of Stap. saprophyticus infection include a unique adhesion protein that allows it to adhere to uroepithelial cells and the production of urease. Isolates of Staph. lugdunensis commonly produce thermostable DNase, lipase and haemolysins, not dissimilar to Staph. aureus.
Treatment
The antibiotic treatment of CNS infections is complicated because susceptibility is generally unpredictable. Strains resistant to penicillin, penicillinase-stable penicillins, gentamicin, erythromycin and chloramphenicol are common. If a strain is the cause of systemic infection, vancomycin or teicoplanin should be used. Rifampicin in combination with a glycopeptide is occasionally useful in treating central nervous system infections or device infections associated with a biofilm. Newer agents such as daptomycin and linezolid may have a role but clinical data is awaited to confirm their efficacy in this setting. Uncomplicated urinary tract infection caused by Staph. saprophyticus usually responds to trimethoprim or one of the fluoroquinolones.
Ala’Aldeen D, Hiramatsu K. Staphylococcus aureus: Molecular and Clinical Aspects. Chichester: Horwood Publishing; 2004.
DeLeo FR, Diep BA, Otto M. Host defense and pathogenesis in Staphylococcus aureus infections. Infectious Diseases Clinics of North America. 2009;23:17–34.
DeLeo FR, Otto M, Kreiswirth BN, Chambers HF. Community-associated meticillin-resistant Staphylococcus aureus. Lancet. 2010;375:1557–1568.
Kluytmans J, Struelens M. Meticillin resistant Staphylococcus aureus in the hospital. British Medical Journal. 2009;338:532–537.
Lowy FD. Staphylococcus aureus infections. New England Journal of Medicine. 1998;339:520–532.
McCann MT, Gilmore B, Gorman SP. Staphylococcus epidermidis device-related infections: pathogenesis and clinical management. Journal of Pharmacy and Pharmacology. 2008;60:1551–1571.
Rogers KL, Fey PD, Rupp ME. Coagulase-negative staphylococcal infections. Infectious Diseases Clinics of North America. 2009;23:73–98.
Working Party Report. Revised guidelines for the control of meticillin-resistant Staphylococcus aureus infection in hospitals. Journal of Hospital Infection. 2006;63(Suppl 1):S1–S44.
European Antimicrobial Resistance Surveillance Network (EARS-Net), http://www.ecdc.europa.eu/en/activities/surveillance/EARS-Net/.
UK Health Protection Agency. Staphylococcus aureus. http://www.hpa.org.uk/Topics/InfectiousDiseases/InfectionsAZ/StaphylococcusAureus/.