22 Clostridium
Gas gangrene; tetanus; food poisoning; pseudomembranous colitis
Key points
• All clostridial infections are characterized by toxin production by the infecting species.
• The major toxins produced by the species are neurotoxins affecting nervous tissue, histotoxins affecting soft tissue and enterotoxins affecting the gut.
• C. perfringens is the major cause of gas gangrene.
• Tetanus, particularly neonatal tetanus, is still a major public health issue in developing countries.
• Botulism occurs predominantly as a severe form of food poisoning.
• C. difficile is the most common cause of diarrhoea in hospital patients in the developed world.
The clostridia are Gram-positive spore-bearing anaerobic bacilli. Most species are saprophytes that normally occur in soil, water and decomposing plant and animal matter; they play an important part in natural processes of putrefaction. Some, such as Clostridium perfringens and C. sporogenes, are commensals of the animal and human gut. On the death of the host, these organisms and other members of the intestinal flora rapidly invade the blood and tissues, and initiate the decomposition of the corpse. The genus has undergone a major taxonomic revision, but this has had little impact on clostridia of medical significance.
• C. perfringens, C. septicum and C. novyi, the causes of gas gangrene and other infections. C. perfringens is also associated with a form of food poisoning.
• C. tetani, the cause of tetanus.
• C. botulinum, the cause of botulism.
• C. difficile, the cause of pseudomembranous colitis and antibiotic-associated diarrhoea.
Genome sequencing of the toxin-producing pathogens C. perfringens, C. tetani, C. botulinum and C. difficile has provided important data on pathogenic determinants and the regulatory events governing their expression as well as revealing the contribution of extra-chromosomal elements to a pathogenic phenotype.
General description
The clostridia are typically large, straight rods with slightly rounded ends. Pleomorphic forms, including filaments or elongated cells, club and spindle-shaped forms (clostridium is Latin for ‘little spindle’) are commonly seen in stained smears from cultures or wounds. They are Gram positive, but may appear to be Gram negative. All produce spores, which enable the organisms to survive in adverse conditions, for example in soil and dust and on skin.
Most species are obligate anaerobes: their spores do not germinate and growth does not normally proceed unless a suitably low redox potential (Eh) exists. A few species grow in the presence of trace amounts of air, and some actually grow slowly under normal atmospheric conditions.
Clostridia are biochemically active, frequently possessing both saccharolytic and proteolytic properties, although in varying degrees. Many species are highly toxigenic. The toxins produced by the organisms of tetanus and botulism attack nervous pathways and are referred to as neurotoxins. The organisms associated with gas gangrene attack soft tissues by producing toxins and aggressins, and are referred to as histotoxic. C. difficile and some strains of C. perfringens produce enterotoxins.
Clostridium perfringens
Description
C. perfringens is a relatively large Gram-positive bacillus (about 4–6 × 1 µm) with blunt ends. It is capsulate and non-motile. It grows quickly on laboratory media, particularly at high temperatures (approximately 42°C), when the doubling time can be as short as 8 min. It can be identified by the Nagler reaction, which exploits the action of its phospholipase on egg yolk medium; colonies are surrounded by zones of turbidity, and the effect is specifically inhibited if C. perfringens antiserum containing α-antitoxin is present on the medium. Typical food-poisoning strains produce heat-resistant spores that can survive boiling for several hours, whereas the spores of the type A strains that cause gas gangrene are inactivated within a few minutes by boiling.
Gas gangrene
C. perfringens is the most common cause of gas gangrene, although various other species of clostridia, including C. septicum, C. novyi type A, C. histolyticum and C. sordellii are occasionally implicated, either alone or in combination. Gas gangrene is almost always a polymicrobial infection involving anaerobes and facultative organisms.
The disease is characterized by rapidly spreading oedema, myositis, necrosis of tissues, gas production and profound toxaemia occurring as a complication of wound infection. The diagnosis is made primarily on clinical grounds with laboratory confirmation.
The main source of the organisms is animal and human excreta, and spores of the causative clostridia are distributed widely. Infection usually results from contamination of a wound with soil, particularly from manured and cultivated land. However, it may be derived indirectly from dirty clothing, street dust, and even the air of an operating theatre if the ventilating system is poorly designed or improperly maintained. The skin often bears spores of C. perfringens, especially in areas of the body that may be contaminated with intestinal organisms.
Pathogenesis of gas gangrene
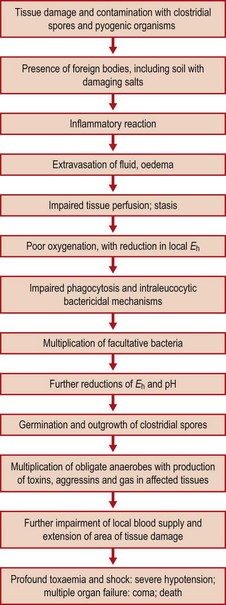
Impairment of the normal blood supply of tissue with a consequent reduction in oxygen tension may allow an anaerobic focus to develop. The patient’s condition may deteriorate rapidly with the development of severe shock (Fig. 22.1).
Crushing of tissue and the severing of arteries in accidental injuries, rough handling of tissue and over-zealous clamping during surgery, or shock waves from gunshot injuries may compromise the microcirculation in an extensive area of tissue and prejudice tissue perfusion. The presence of devitalized or dead tissue, blood clot, extravasated fluid, foreign bodies and coincident pyogenic infection are all factors that promote the occurrence of gas gangrene in a wound. The spores of the clostridia and their vegetative bacilli cannot readily initiate infection in healthy tissues, presumably because the Eh is too high, and the organisms are unable to avoid destruction and clearance by phagocytosis. Predisposing host factors include debility, old age and diabetes.
When clostridial infection has been initiated in a focus of devitalized anaerobic tissue, the organisms multiply rapidly and produce a range of toxins and aggressins. These damage tissue by various necrotizing effects, and some have demonstrable lethal effects. They spread into adjacent viable tissue, particularly muscle, kill it, and render it anaerobic and vulnerable to further colonization, with the production of more toxins and aggressins.
• Hyaluronidase produced by C. perfringens breaks down intercellular cement substance and promotes the spread of the infection along tissue planes.
• Collagenase and other proteinases break down tissues and virtually liquefy muscles. The whole of a muscle group or segment of a limb may be affected.
• α-Toxin, a phospholipase C (lecithinase), is generally considered to be the main cause of the toxaemia associated with gas gangrene, although other clostridial species can produce similar manifestations.
In puerperal infections or in cases of septic abortion, the organisms may gain access from faeces-contaminated perineal skin or contaminated instruments to necrotic or devitalized tissues in the uterus or adnexa. Here they set up a dangerous and often fulminating pelvic infection, possibly with prompt invasion of the bloodstream. There may be intravascular haemolysis and anuria.
C. perfringens may also participate in peritoneal infections that occur as a result of extension of pathogens from the alimentary tract, as in cases of gangrenous appendicitis or intestinal obstruction or mesenteric thrombosis.
If a preparation of adrenaline (epinephrine) used for injection is contaminated with clostridial spores, the combination of an infective inoculum with the local ischaemia that follows the injection may be catastrophic. Gas gangrene may complicate surgical operations on the lower limb or hip of patients whose blood supply is inadequate to maintain oxygenation in the post-operative period.
Clostridia may be associated with less severe forms of infection without the toxaemia and aggression of gas gangrene. Moreover, potentially pathogenic anaerobes may be cultivated from a wound that never shows any sign of gas gangrene, and sometimes the laboratory isolation may be attributed to the germination of a few contaminating spores when the specimen was being processed. Thus, the onus is on the clinician to relate a laboratory report to the patient’s circumstances.
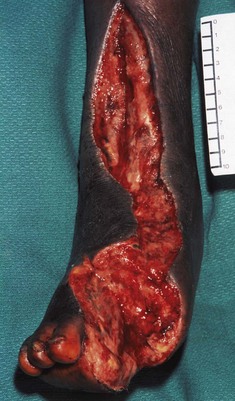
Clinical clues include crepitus, the sponge-cake consistency caused by small bubbles of gas in adjacent tissues. In the early stages, the patient has an anxious, frightened appearance. Local pain is increased, and there is swelling of the affected tissues. Toxaemia and shock supervene, and the patient becomes drowsy and drifts into coma. Prompt diagnosis and intensive surgical and antimicrobial treatment greatly influence the patient’s chance of survival and may avoid the loss of the affected limb, but all devitalized tissue must be excised (see below).
Laboratory diagnosis
If there are sloughs of necrotic tissue in the wound, small pieces should be transferred aseptically into a sterile screw-capped bottle and examined immediately by microscopy and culture. Specimens of exudate should be taken from the deeper areas of the wound where the infection seems to be most pronounced. Gram smears are prepared. If gas gangrene exists, typical Gram-positive bacilli may predominate, often with other bacteria present in a mixed infection. However, there is usually a pronounced lack of inflammatory cells. Initiation of treatment should not await a full laboratory report and early discussion with the bacteriologist is crucial. A direct smear of a wound exudate is often of great help in providing evidence of the relative numbers of different bacteria that may be participating in a mixed infection or may merely be present as contaminants, but the distinction is not invariably easy and joint discussions are important.
Treatment
Prompt and adequate surgical attention to the wound is of the utmost importance (Fig. 22.2).
• Sutures are removed, and necrotic and devitalized tissue is excised with careful debridement.
• Fascial compartments are incised to release tension.
• Any foreign body is found and removed.
• The wound is not resutured but is left open after thorough cleansing, and loosely packed.
Antibiotic therapy is started immediately in very high doses. This must take account of the likely coexistence of coliform organisms, Gram-positive cocci and faecal anaerobes. Accordingly, penicillin, metronidazole and an aminoglycoside may be given in combination. Alternatively, clindamycin plus an aminoglycoside or a broad-spectrum antibiotic, such as meropenem or imipenem, may be considered. Much intensive supportive therapy is needed.
Enthusiastic claims have been made for the efficacy of hyperbaric oxygen therapy, but clinical trials have given conflicting results. Patients are placed in a special pressurized chamber where they breathe oxygen at 2–3 atm pressure for periods of 1–2 h twice daily on several successive days. This may limit the amount of radical surgery needed.
A polyvalent antiserum containing C. perfringens, C. septicum and C. novyi antitoxins was used formerly, but has been replaced by intensive antimicrobial therapy.
Prophylaxis
Surgical wounds
C. perfringens is normally present in large numbers in human faeces, and its spores are found regularly on the skin, especially of the buttocks and thighs. As clostridial spores are very resistant to most disinfectants, they are likely to survive normal pre-operative skin preparation and persist in the area of the planned incision. The numbers can be reduced by more prolonged skin preparation with the sustained action of an antiseptic such as povidone–iodine for a day or two; this procedure has a place in orthopaedic surgery.
When inevitable skin contamination is combined with circumstances that predispose to devitalization of tissue and reduced oxygen tension, a patient may be vulnerable to the development of post-operative gas gangrene. These circumstances arise when an elderly patient or a patient with vascular insufficiency undergoes major surgery to the hip or lower limb. Perioperative antimicrobial prophylaxis with penicillin is justified in such cases.
Accidental wounds
The prevention of gas gangrene in accidentally sustained wounds must take account of the endogenous factors noted above and the exogenous sources of clostridial spores and vegetative forms in soil, on contaminated clothing, etc. In addition, there is an increased risk of anaerobic infection developing when foreign bodies such as soil, clothing, metal (nails, wire, bullets, shrapnel) and skin are driven into devitalized tissues. Prompt and adequate surgical attention is of paramount importance, but prophylactic administration of benzylpenicillin for patients presenting with serious contaminated wounds is also worthwhile. Prophylaxis may be omitted if the state of the wound and the patient’s general condition are expertly and frequently monitored throughout the recovery period.
Food poisoning
Carrier rates for ‘typical food poisoning strains’ of C. perfringens range from about 2% to more than 30% in different surveys across the world. These bacteria also occur in animals; thus, meat is often contaminated with heat-resistant spores. When meat is cooked in bulk, heat penetration and subsequent cooling is slow unless special precautions are taken. During the cooling period surviving spores may germinate and multiply in the anaerobic environment produced by the cooked meat. Anyone eating this will consume the equivalent of a cooked meat broth culture of the organism. The organisms are protected from the gastric acid by the protein in the meal and pass in large numbers into the intestine.
Ingestion of large numbers of viable organisms is necessary for the production of the typical disease syndrome, which is mediated by an enterotoxin that is released when sporulation occurs in the gut. Typical symptoms are abdominal cramps beginning about 8–12 h after ingestion, followed by diarrhoea. Fever and vomiting are not usually encountered and symptoms generally subside within a day or two. No specific treatment is indicated. The carrier state persists for several weeks, but this should not be regarded as an indication for exclusion from any duties, as carriers may be quite numerous in various communities.
The vehicle of infection is usually a pre-cooked meat food that has been allowed to stand at a temperature conducive to the multiplication of C. perfringens. Although the heat resistance of spores of typical food-poisoning strains ensures their survival in cooked foods, and presumably accounts for the association of these strains with most of the reported outbreaks of C. perfringens food poisoning, similar trouble can be caused by classical heat-sensitive type A strains if they gain access to food during the cooling period under conditions suitable for their subsequent multiplication.
Laboratory diagnosis
This can be difficult as some people carry large numbers of C. perfringens. Diagnosis depends upon the isolation of similar strains of C. perfringens from the faeces of patients and from others at risk who have eaten the suspected food, and from the food itself. Numbers usually exceed 106 organisms/g faeces. The isolates can be sent to a reference laboratory for special typing to prove their relatedness.
Colitis
A sporadic diarrhoeal syndrome, usually occurring in elderly patients during treatment with antibiotics, has been described. The circumstances differ substantially from those of C. perfringens food poisoning. A cytopathic enterotoxin can be detected in the patient’s faeces.
Enteritis necroticans (pigbel)
A subgroup of C. perfringens type C that produces heat-resistant spores is the cause of a disease that affects New Guinea natives when they have pork feasts. The method of cooking the pork allows the clostridia to survive. When the contaminated meat is eaten along with a sweet potato vegetable that contains a proteinase inhibitor, a toxin (the β-toxin) is able to act on the small intestine to produce a necrotizing enteritis. A successful vaccination programme has reduced the incidence of pigbel dramatically.
Clostridium septicum
Description
The bacilli are generally large and actively motile with numerous flagella. It is one of the less exacting anaerobes and grows well at 37°C on ordinary media. Spores are readily formed and, as they develop, various pleomorphic forms arise ranging from swollen Gram-positive ‘citron bodies’ to obviously sporing forms in which the oval spores may be central or subterminal and are clearly bulging.
Pathogenesis
C. septicum is one of the gas gangrene group of clostridia. It occurs harmlessly in the human intestine, but if the integrity of the gut epithelium is impaired, for instance by leukaemic infiltration, bacteraemia may occur. Cyclic or other forms of neutropenia are also associated with spontaneous, non-traumatic gas gangrene that begins with a bacteraemic phase. Typhlitis, a rapidly fatal terminal ileal infection and septicaemia in immunocompromised patients, is most commonly associated with C. septicum.
Intramuscular injection of cultures into laboratory animals produces a spreading inflammatory oedema, with slight gas formation in the tissues. Organisms invade the blood and the animal dies within a day or two. Smears from the liver show long filamentous forms and citron bodies. C. septicum produces several toxins (α, β, δ and ε). The α-toxin, which has lethal, haemolytic and necrotizing activity, appears to be the most important; it does not have phospholipase C activity and thus differs from the α-toxin of C. perfringens.
Clostridium novyi
C. novyi resembles C. perfringens in morphology, but is larger, more pleomorphic and more strictly anaerobic; it is readily killed when vegetative cells are exposed to air. The spores are oval, central or subterminal. The organism occurs widely in soil and is associated with disease in man and animals. There are four types – A, B, C and D – distinguished on the basis of permutations of the toxins and other soluble antigens they produce. Only type A strains are of medical interest as they cause some cases of gas gangrene in man. In 2000, an outbreak of C. novyi type A infections among injecting heroin users killed 13 people in the UK.
C. novyi gas gangrene is associated with profound toxaemia. Culture filtrates are highly toxic and possess at least four active substances (α, β, δ and ε toxins) that account for the various haemolytic, necrotizing, lethal, phospholipase and lipase activities of this organism.
Clostridium sporogenes
This clostridium is widely distributed in nature as a harmless saprophyte. Its spores may survive boiling for periods up to 6 h and the organism is often encountered in mixed cultures after preliminary heating to select heat-resistant pathogens. Although its presence in wound exudates may accelerate an established anaerobic infection by enhancing local conditions, it is not a pathogen in its own right and does not cause gas gangrene.
Clostridium tetani
Description
The tetanus bacillus is a motile, straight, slender, Gram-positive rod. A fully developed terminal spore gives the organism the appearance of a drumstick with a large round end. Gram-negative forms are usually encountered in stained smears. It is an obligate anaerobe that grows well in cooked meat broth and produces a thin spreading film when grown on enriched blood agar. The spores may be highly resistant to adverse conditions, but the degree of resistance varies with the strain. Spores of some strains resist boiling in water for up to 3 h. They may resist dry heat at 160°C for 1 h, and 5% phenol for 2 weeks or more. Iodine (1%) in water is said to kill the spores within a few hours, but glutaraldehyde is one of the few chemical disinfectants that is assuredly sporicidal.
Pathogenesis
As in gas gangrene (see p. 246), germination of spores and their outgrowth depend upon reduced oxygen tension in devitalized tissue and non-viable material in a wound. When infection occurs, often assisted by the simultaneous growth of facultatively anaerobic organisms in a mixed inoculum, the tetanus bacillus remains strictly localized, but tetanus toxin is elaborated and diffuses, as described below.
Toxins
C. tetani produces an oxygen-labile haemolysin (tetanolysin), but the organism’s neurotoxin (tetanospasmin) is the essential pathogenic product. Strains vary in their toxigenicity; some are highly toxigenic. Most strains produce demonstrable toxin after culture in broth for a few days.
The gene encoding the neurotoxin is located on a plasmid. The toxin is synthesized as a single polypeptide with a molecular weight of 150 000 Da, which undergoes post-translational cleavage into a heavy chain and a light chain linked by a disulphide bond. The estimated lethal dose for a mouse of pure tetanospasmin is 0.0001 µg. It is toxic to man and various animals when injected parenterally, but not by the oral route.
When tetanus occurs naturally, the tetanus bacilli stay at the site of the initial infection and are not generally invasive. Toxin diffuses to affect the relevant level of the spinal cord (local tetanus) and then to affect the entire system (generalized tetanus). These stages, including the intermediate one of ‘ascending tetanus’, are demonstrable in experimental animals, but the stages tend to merge in their clinical presentation in man.
The toxin is absorbed from the site of its production in an infective focus, but may be delivered via the blood to all nerves in the body. The heavy chain mediates attachment to gangliosides and the toxin is internalized. It is then moved from the peripheral to the central nervous system by retrograde axonal transport and trans-synaptic spread. The tendency for the first signs of human tetanus to be in the head and neck is attributed to the shorter length of the cranial nerves. In fact, descending involvement of the nervous system is seen as the tetanus toxin takes longer to traverse the longer motor nerves and also diffuses in the spinal cord.
Once the entire toxin molecule has been internalized into presynaptic cells, the light chain is released and affects the membrane of synaptic vesicles. This prevents the release of the neurotransmitter γ-aminobutyric acid. Motor neurones are left under no inhibitory control and undergo sustained excitatory discharge, causing the characteristic motor spasms of tetanus. The toxin exerts its effects on the spinal cord, brainstem, peripheral nerves, at neuromuscular junctions and directly on muscles.
Clinical features of tetanus
Cases of tetanus have been reported in which the infection was apparently associated with a superficial abrasion, a contaminated splinter or a minor thorn prick. Otogenic tetanus may be attributable to over-zealous cleansing of the external auditory meatus with a small stick. In other patients, the site of infection remains undiscovered (cryptogenic tetanus). Tetanus infection may also occur in or near the uterus in cases of septic abortion.
Tetanus neonatorum follows infection of the umbilical wound of newborn infants (see below). Cases of post-operative tetanus have been attributed to imperfectly sterilized catgut, dressings or glove powder, and sometimes to dust-borne infection of the wound at operation.
The onset of signs and symptoms is gradual, usually starting with some stiffness and perhaps pain in or near a recent wound. In some cases the initial complaint may be of stiffness of the jaw (lockjaw). Pain and stiffness in the neck and back may follow. The stiffness spreads to involve all muscle groups; facial spasms produce the ‘sardonic grin’, and in severe cases spasm of the back muscles produces extreme arching of the back (opisthotonos). The period between injury and the first signs is usually about 10–14 days, but there is a considerable range. A severe case with a relatively poor prognosis shows rapid progression from the first signs to the development of generalized spasms. Sweating, tachycardia and arrhythmia, and swings in blood pressure, reflect sympathetic stimulation, which is not well understood but creates problems of management.
Treatment
The patient remains conscious and requires skilled sedation and constant nursing. If generalized spasms are worrying, the patient is paralysed and ventilated mechanically until the toxin that has been taken up has decayed; this may take some weeks.
The patient is given 10 000 units of human tetanus immunoglobulin (HTIG) in saline by slow intravenous infusion. Full wound exploration and debridement is arranged, and the wound is cleansed and left open with a loose pack. Penicillin or metronidazole is given for as long as considered necessary to ensure that bacterial growth and toxin production are stopped. The antitoxin and antibiotics are given immediately, and preferably before surgical excision, but delay must be avoided.
Laboratory diagnosis
Gram smears of the wound exudate and any necrotic material may show the typical ‘drumstick’ bacilli, but this is not invariably so, and thus provides only presumptive evidence as other organisms that resemble C. tetani have terminal spores. Simple light microscopy is often unsuccessful; immunofluorescence microscopy with a specific stain is possible but not generally available.
Direct culture of unheated material on blood agar incubated anaerobically is often the best method of detecting C. tetani. There are various other tricks that exploit the organism’s motility and fine spreading growth; sometimes these are vitiated by overgrowth with Proteus species. Material from the wound or from a mixed sporing subculture may be heated at various temperatures and for various times to exclude non-sporing bacteria; the heated specimens are then seeded on to solid media and incubated anaerobically. Tetanus may be produced in mice by subcutaneous injection of an anaerobic culture prepared from wound material; control mice are protected with tetanus antitoxin.
Epidemiology
Tetanus bacilli may be found in the human intestine, but infection seems to be derived primarily from animal faeces and soil. The organism is especially prevalent in manured soil and for this reason a wound through skin contaminated with soil or manure deserves special attention. However, tetanus spores occur very widely and are commonly present in gardens, sports fields and roads, in the dust, plaster and air of hospitals and houses, on clothing and on articles of common use.
Spores of C. tetani and other anaerobes may be embedded in surgical catgut and other dressings. However, the sterility of surgical catgut (prepared from the gut of cattle and sheep) is now rigorously controlled.
Tetanus ranks among the major fatal infections. During the 1980s there were between 800 000 and 1 million deaths annually from tetanus, of which 400 000 were due to neonatal tetanus. The incidence varies enormously from country to country, and is inversely related to socio-economic development and standards of living, preventive medicine and wound management. In developed countries, the reported incidence of adult and childhood tetanus is low. There is a direct relationship with fertile soil and a warm climate; thus, people living in the agricultural areas of developing tropical and subtropical countries are exposed to severe challenges associated with poor hygiene, lack of shoes, neglect of wounds and inadequate immunization.
In addition, some local customs promote the occurrence of tetanus:
• treatment of the umbilical cord stump with primitive applications that include animal dung
• tying of the umbilical cord itself with primitive ligatures
• ear-piercing and other operations performed with unsterile instruments.
Fatality rates may exceed 50% and neonatal tetanus carries a very high mortality rate. Case fatality rates can be greatly reduced to less than 10% by modern methods of treatment in specialist centres. Unfortunately, such expensive skilled help is available for only a small proportion of patients. Under-privileged people in countries with poorly developed or expensive medical services are at greatest risk, and are least likely to receive sophisticated assistance; 80% of deaths occur in Africa and Southeast Asia. A maternal vaccination campaign mounted by the United Nations Children’s Fund (UNICEF) during the past 15 years has reduced the incidence of neonatal tetanus by 50%.
Prevention and control
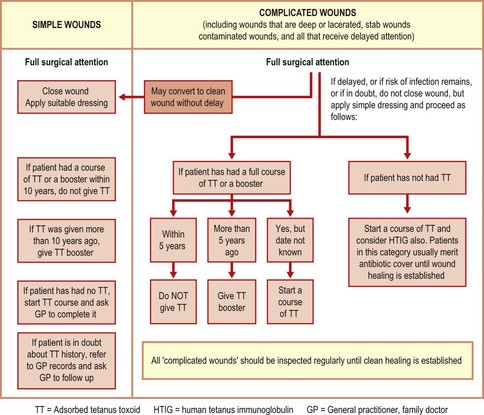
Prompt and adequate wound toilet and proper surgical debridement of wounds are of paramount importance. There is an increased risk that tetanus spores may germinate in a wound if cleansing is delayed and if sepsis develops. Clean superficial wounds that receive prompt attention may not require specific protection against tetanus and it is unreasonable to insist that every small prick or abrasion requires protection with antitoxin or antibiotic.
Routine practice should take account of the local incidence of tetanus and the individual circumstances of the case. It is wise to recommend specific prophylaxis for a non-immunized patient with a deep wound, puncture or stab wound, ragged laceration, a wound with much bruising and any devitalized tissue, a wound that is already septic, or a bite wound or other type of wound that is likely to be heavily contaminated. Figure 22.3 shows an approach that reflects current thinking in the UK.
The need for passive immunization is avoided if the patient is known to be properly immunized against tetanus (see Ch. 70). A patient may be regarded as immune for 6 months after the first two injections, or for 5–10 years after a planned course of three injections (or a subsequent booster injection) of adsorbed tetanus toxoid. Tetanus antitoxin should not be given to immune patients, but their active immunity may be enhanced when necessary by giving a dose of tetanus toxoid at the time of injury if the circumstances justify it.
A patient is considered non-immune if there is no history of having had an injection of tetanus toxoid or if only one injection has been given. Take care: a patient may recall having had ‘a tetanus shot’, but this may have been a previous dose of antitoxin for passive protection (which is transient and cannot be boosted by toxoid). If more than 6 months have elapsed after a course of two injections, or more than 10 years after a full primary course of three injections of adsorbed toxoid (or a booster injection), the patient should be regarded as non-immune. A patient is non-immune if more than 2–3 weeks have elapsed since a previous injection of equine antitoxin, or more than 6–8 weeks in the case of homologous (human) antitoxin. Non-immunity should be assumed if there is any doubt about the immunization history.
Passive immunization with antitoxin
HTIG (homologous antitoxin) is available for passive protection and now supersedes equine antitoxin (heterologous antitoxin), which was associated with occasional adverse reactions. However, equine antiserum should not be prematurely discarded in countries that do not yet have HTIG. The prophylactic dose of HTIG is 250–500 units by intramuscular injection.
Combined active–passive immunization
A non-immune patient receiving passive protection with HTIG after injury may be given the first dose of a course of active immunization with adsorbed toxoid at the same time, provided the injections are given from different syringes and into contralateral sites. The active immunization course must subsequently be completed.
Antibiotic protection
The prophylactic administration of antibiotics to all cases of open wounds is not recommended, although the use of penicillin or clindamycin is justified in some cases when there is a significant risk of infection. This precaution must not replace prompt and adequate surgical wound toilet.
Clostridium botulinum
Description
C. botulinum is a strictly anaerobic Gram-positive bacillus. It is motile and has spores that are oval and subterminal. It is a widely distributed saprophyte found in soil, vegetables, fruits, leaves, silage, manure, the mud of lakes and sea mud. Its optimal growth temperature is about 35°C, but some strains can grow and produce toxin at temperatures as low as 1–5°C.
The widespread occurrence of C. botulinum in nature, its ability to produce a potent neurotoxin in food, and the resistance of its spores to inactivation combine to make it a formidable pathogen. Spores of some strains withstand boiling in water (100°C) for several hours. They are usually destroyed by moist heat at 120°C within 5 min. Spores of type E strains (see below) are usually much less heat resistant. Insufficient heating in the process of preserving foods is an important factor in the causation of botulism. The resistance of the spores to radiation is of special relevance to food processing.
Botulinum toxin is categorized as a biothreat level A biological warfare agent. Introduction of toxin into a target population by contaminating food or water is unlikely to succeed because of logistical problems, but botulinum toxin can also cause disease by inhalation.
Carefully controlled injections of toxin are used to treat involuntary muscle disorders, and as an ‘anti-aging’ remedy.
Pathogenesis
Botulism is a severe, often fatal, form of food poisoning characterized by pronounced neurotoxic effects. Botulinum toxins are among the most poisonous natural substances known. Seven main types of C. botulinum, designated A–G, produce antigenically distinct toxins with pharmacologically identical actions. All types can cause human disease, but types A, B and E are most common. The importance of this point is that, if antitoxin is given to a patient in an emergency, only the type-specific antitoxin will be effective.
The disease has been linked to a wide range of foods, including preserved hams, large sausages of the salami type, home-preserved meats and vegetables, canned products such as fish, liver paste, and even hazelnut purée and honey. Traditional dishes such as fish or seal flipper fermented in a barrel buried in the ground cannot be recommended by a bacteriologist! Type E strains are particularly but not invariably associated with a marine source, whereas type A and type B strains are usually associated with soil.
Foods responsible for botulism may not exhibit signs of spoilage. The pre-formed toxin in the food is absorbed from the intestinal tract. Although it is protein, intestinal proteolytic enzymes do not inactivate it. After absorption, botulinum toxin binds irreversibly to the presynaptic nerve endings of the peripheral nervous system and cranial nerves, where it inhibits acetylcholine release.
Clinical features
The period between ingestion of the toxin and the appearance of signs and symptoms is usually 1–2 days, but it may be much longer. There may be initial nausea and vomiting. The oculomotor muscles are affected, and the patient may have diplopia and drooping eyelids with a squint. There may be vertigo and blurred vision.
There is progressive descending motor loss with flaccid paralysis but with no loss of consciousness or sensation, although weakness and sleepiness are often described. The patient is thirsty, with a dry mouth and tongue. There are difficulties in speech and swallowing, with later problems of breathing and despair. There may be abdominal pain and restlessness. Death is due to respiratory or cardiac failure.
Wound botulism
Rare cases of wound infection with C. botulinum resulting in the characteristic signs and symptoms of botulism have been recorded.
Infant botulism
The ‘floppy child syndrome’ describes a young child, usually less than 6 months old, with flaccid paralysis that is ascribed to the growth of C. botulinum in the intestine at a stage in development when the colonization resistance of the gut is poor. Some cases have been attributed to the presence of C. botulinum spores in honey; when the honey was given as an encouragement to feed, the ingested spores were able to germinate and produce toxin in the infant gut.
Laboratory diagnosis
The organism or its toxin may be detected in the suspected food, and toxin may be demonstrated in the patient’s blood by toxin–antitoxin neutralization tests in mice. Samples of faeces or vomit may also yield such evidence. Take care: bear in mind that botulinum toxin is very dangerous – specialist help should be summoned and the laboratory alerted.
Control
Great care must be taken in canning factories to ensure that adequate heating is achieved in all parts of the can contents. Home canning of foodstuffs should be avoided. The amateur preservation of meat and vegetables, especially beans, peas and root vegetables, is dangerous in inexperienced hands. Acid fruits may be bottled safely in the home with heating at 100°C, as a low pH is inhibitory to the growth of C. botulinum.
A prophylactic dose of polyvalent antitoxin should be given intramuscularly to all persons who have eaten food suspected of causing botulism. Injecting three doses of mixed toxoid at intervals of 2 months can produce active immunity, but the very low incidence of the disease under normal conditions does not justify this as a routine. Active immunization should be considered for laboratory staff who might have to handle the organism or specimens containing the organism or its toxin.
Clostridium difficile
Description
C. difficile is a motile Gram-positive rod with oval subterminal spores. It commonly occurs in the faeces of neonates and babies until the age of weaning, but it is not generally found in adults.
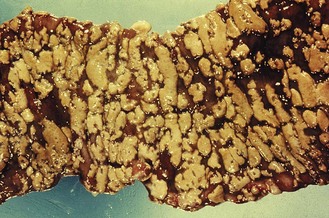
The organism produces an enterotoxin (toxin A) and a cytotoxin (toxin B); some strains produce a third, binary, toxin. It causes antibiotic-associated diarrhoea, occasionally leading to a life-threatening condition, pseudomembranous colitis (Fig. 22.4). There is almost always a history of previous antibiotic therapy although exposure to any agent that perturbs the gut flora, including cytotoxic drugs, may lead to infection. Clindamycin and lincomycin are associated with a particularly high risk but extended-spectrum cephalosporins are also commonly incriminated and are much more commonly used; virtually no antimicrobial drug has escaped blame. A worldwide epidemic of fluoroquinolone-resistant ribotype 027 C. difficile infection is probably due to excessive use of these agents.
Laboratory diagnosis
C. difficile can be isolated from the faeces by enrichment and selective culture procedures. Toxin B can be detected in the patient’s faeces by testing extracts against monolayers of susceptible cells, or both toxins may be demonstrated by immunological methods, such as enzyme-linked immunosorbent assay (ELISA). The sensitivity of many ELISA kits is only 50–70% and results should be interpreted with caution. Moreover, toxin A negative strains, which still cause disease and are prevalent in certain regions of the world, are not detected by some kits. Due to these issues, highly sensitive PCR (polymerase chain reaction) detection of the toxin B gene is now the favoured approach.
Treatment
It is essential to discontinue the antibiotic that is presumed to have precipitated the disease and to suppress the growth and toxin production of C. difficile by giving oral metronidazole or vancomycin. Pseudomembranous colitis may be fatal if it is not quickly recognized and treated.
Epidemiology
The organism is usually acquired from an exogenous source by a patient whose intestinal colonization resistance has been compromised by antibiotic exposure. Patients developing infection have often spent lengthy periods in hospital. Re-infection occurs in 20–50% of cases as it may take the gut 2–3 months to normalize after perturbation.
Many production and companion animals are colonized by C. difficile before weaning and meat and meat products in North America have been found to be contaminated by C. difficile spores. Although the suggestion that C. difficile is a common food-borne pathogen is premature, animals may be an important reservoir of disease. An apparent increase in community-acquired infection in the absence of risk factors such as antibiotic exposure, has led to suggestions that all patients with community-acquired diarrhoea should be tested for C. difficile.
Prevention
Clinical awareness is the keynote. C. difficile is the most common cause of hospital-acquired diarrhoea. If a patient develops diarrhoea after at least 48 h in hospital, especially while taking antibiotics, the possibility of C. difficile infection must be considered. If several cases occur in a hospital unit, cross-infection should be considered as the hospital environment can become extensively contaminated with C. difficile spores. The existing antibiotic and infection control policies of the unit should be reviewed.
Caya JG, Agni R, Miller JE. Clostridium botulinum and the clinical laboratorian. Archives of Pathology and Laboratory Medicine. 2004;128:653–662.
Crobach MJ, Dekkers OM, Wilcox MH, Kuijper EJ. European Society of Clinical Microbiology and Infectious Diseases (ESCMID): data review and recommendations for diagnosing Clostridium difficile-infection (CDI). Clinical Microbiology and Infection. 2009;15:1053–1066.
Farrar JJ, Yen LM, Cook T, et al. Tetanus. Journal of Neurology, Neurosurgery & Psychiatry. 2000;69:292–301.
Freeman J, Bauer MP, Baines S, et al. The changing epidemiology of Clostridium difficile infections. Journal of Clinical Microbiology. 2010;23:529–549.
McLauchlin J, Little C. Hobbs’ 1993 Food Poisoning and Food Hygiene, ed 7. London: Hodder Arnold; 2007.
Popoff MR, Bouvet P. Clostridial toxins. Future Microbiology. 2009;4:1021–1064.
Rood JI, McClane BA, Songer JG, Titball RW. The Clostridia – Molecular Biology and Pathogenesis, ed 1. San Diego: Academic Press; 1997.
Shapiro RL, Hatheway C, Swerdlow DL. Botulism in the United States: a clinical and epidemiologic review. Annals of Internal Medicine. 1998;129:221–228.