70 Immunization
Key points
• Immunization is a major means by which infections may be controlled and, in some cases, eradicated.
• Passive immunization involves the administration of immunoglobulin-containing preparations that provide protection against one or more infections. The immunity is short-lived. Preparations providing protection against tetanus, hepatitis B, rabies, varicella–zoster and vaccinia are available.
• Active immunization produces more enduring immunity. Live-attenuated, killed (inactivated) and subunit (e.g. toxoid or capsular antigen) vaccines are used widely.
• The use of particular vaccines is contraindicated in certain individuals, such as the use of live vaccines in immunocompromised individuals.
• Where immunization prevents transmission, the objective of immunization programmes is to achieve herd immunity such that, once a threshold level of immunization has been achieved, no further transmission can take place in that community.
• The standard immunization schedule in the UK provides for active immunization in childhood against diphtheria, tetanus, pertussis, seven serotypes of pneumococci, Haemophilus influenzae capsule type B (Hib), Neisseria meningitidis capsule type C, polio, and measles, mumps and rubella.
• Additional active immunization may be considered against hepatitis B, varicella, tuberculosis and influenza for specific groups, and travellers are routinely offered additional vaccines specific to the locations visited.
It is appropriate that the last chapter of this book should be devoted to immunization, which is only one of the measures that may be used for the control of infectious diseases (see Ch. 68) yet has dramatically and successfully rid the world of the scourge of smallpox and is well on the way to eradicating poliomyelitis. With rapid advances in understanding host responses to antigens and the means to make and deliver vaccines, the future looks exciting for eradicating other communicable diseases in humans and animals. The cost and efficacy of immunization programmes must be assessed against other forms of defence, (Box. 70.1), such as:
• clean air and adequate ventilation
• good animal husbandry with effective quarantine arrangements where necessary
Box 70.1 Approaches to the prevention of infectious disease
An indication of priorities
• Preventive engineering: water, sewage, ventilation, food production and food processing
• Surveillance and diagnostic awareness
• Prompt management: recognition, treatment, isolation and contact tracing where necessary
• Improvement and maintenance of good socio-economic conditions: housing, nutrition, education, medical and social care
• Increased resistance to infection by appropriate active immunization policies and in some special circumstances by passive immunization
Much of the morbidity and mortality of infection is still not preventable by immunization or presents great difficulties. For example, at present, many of the parasitic, diarrhoeal and respiratory infections that take a heavy toll of life and health among young children in poor and overcrowded communities are not amenable to control by specific vaccines; nor are the common bacterial infections associated with haemolytic streptococci, staphylococci and coliform bacilli.
Rationale of immunization
The objective is to produce, without harm to the recipient, a degree of resistance sufficient to prevent a clinical attack of the natural infection and to prevent the spread of infection to susceptibles in the community. There is therefore both a personal gain from being immunized and a public health benefit to the population. The degree of resistance conferred may not protect against an overwhelming challenge, but exposure may help to boost immunity.
Passive immunization
Artificial passive immunization is used in clinical practice when it is considered necessary to protect a patient at short notice and for a limited period. Antibodies, which may be antitoxic, antibacterial or antiviral, in preparations of human or animal serum are injected to give temporary protection. Human preparations are referred to as homologous, and are much less likely to give rise to the adverse reactions occasionally associated with the injection of animal (heterologous) sera. An additional advantage of homologous antisera is that, although they do not confer durable protection, their effect may persist for 3–6 months, whereas the protection afforded by a heterologous serum is likely to last for only a few weeks. The use of monoclonal antibodies against specific pathogens, such as respiratory syncytial virus, is likely to widen the scope of passive immunization in future.
Antiserum raised in the horse against diphtheria toxin (equine diphtheria antitoxin) is available in some countries for the prophylaxis and treatment of diphtheria. A similar heterologous antiserum is available for emergency use in cases of suspected botulism and to protect those thought to be at risk. Equine tetanus antitoxin is still used in some parts of the world, but it should be abandoned in favour of human tetanus immunoglobulin (see Ch. 22, p. 251). It is most important to give an intended recipient of equine serum a prior test dose to exclude hypersensitive subjects who may have been sensitized by a previous dose of equine serum and may suffer ‘serum sickness’.
Pooled immunoglobulins
Protective levels of antibody to a range of diseases are present in pooled normal human serum. Human normal immunoglobulin (HNIG) is available for the short-term prophylaxis of hepatitis A in contacts or travellers who intend to visit countries where hepatitis A is common. However, active immunization is a much better alternative against hepatitis A for frequent travellers to endemic areas. HNIG also protects those with agammaglobulinaemia and has been used in cases of Gram-negative sepsis and C. difficile infection.
Active immunization
Types of vaccine
Toxoids
If the signs and symptoms of a disease can be attributed essentially to the effects of a single toxin, a modified form of the toxin that preserves its antigenicity but has lost its toxicity (a toxoid) provides the key to successful active immunization against the disease. This has been spectacularly successful with tetanus and diphtheria.
Inactivated vaccines
If the disease is not mediated by a single toxin, it may be possible to stimulate the production of protective antibodies by using the killed (inactivated) organisms. This is done as a routine with vaccines against pertussis (whooping cough), influenza and the inactivated polio (Salk) vaccine.
Attenuated live vaccines
In some cases, the inactivation procedure to make a killed vaccine destroys or modifies the protective antigenicity (immunogenicity) of the organisms. Hence, another approach is to use suspensions of living organisms that are reduced in their virulence (attenuated) but still immunogenic. This strategy has yielded mumps, measles, rubella and varicella vaccines (now combined), the live-virus polio (Sabin) and yellow fever vaccines.
Sometimes, it is possible to use a related organism with shared antigens. Thus, the vaccinia virus vaccine was used to eradicate smallpox, and a bovine tubercle bacillus was modified by Calmette and Guérin to make bacille Calmette–Guérin (BCG), which protects humans against tuberculosis and leprosy (see pp. 220 & 225).
Special procedures
Some vaccines, such as influenza (virus) vaccine, can be refined by a process that removes unwanted protein and other reactive material but retains the important protective antigens. Some others must be conjugated to proteins to render them immunogenic. Some vaccines, such as hepatitis B vaccine, can be bioengineered. There is much current interest in the possibility of developing subunit vaccines consisting of purified fragments of the major immunogenic components of micro-organisms, particularly viruses.
Immune response
Antibodies against the agents of some bacterial and viral infections may be present in the mother’s blood and be passively acquired by the baby (see Part 2 of this book). This gives some protection to the infant at a time when it is poorly equipped to produce specific antibodies, but it may interfere to a varying extent with the infant’s capacity to respond to the stimulus of injected or ingested vaccines in the very early months of life. Although the capacity of the infant to produce specific antibody to injected antigens is poorly developed in the first few months of life, this problem can often be resolved by the use of adjuvants. Thus, effective responses are produced to powerful antigens such as alum-adsorbed toxoids. Pertussis whole-cell vaccines have an adjuvant effect of their own, so the combination of adsorbed toxoids and whole-cell pertussis vaccine (the triple diphtheria, tetanus and pertussis vaccine; DTP) is an effective immunizing complex that used to be given in the UK at 2, 3 and 4 months of age to cover the period when the lethal potential of pertussis is greatest (see below and Ch. 32). The tissues of the newborn respond effectively to BCG vaccine because the protection here is cell-mediated. The use of a high-potency measles vaccine at 6–9 months of age is likely to be exploited in countries where measles kills or severely injures the very young.
When a good specific antibody response is being sought to a toxoid or a killed antigen, the usual procedure is to give three doses of the antigen at spaced intervals. The first or ‘priming’ dose evokes a low level of antibody after a latent period of about 2 weeks, but the second dose elicits a much greater (secondary) antibody response, and this is further boosted by the third dose. The efficacy of injected antigen preparations can be enhanced by slow-release agents such as mineral carriers which have adjuvant effects. With most antigens, the response is better if the first two doses are separated by an interval of a month or two. A third dose is generally recommended at some time thereafter, and further booster doses may be given to maintain immunity.
Duration of immunity
After an effective course of active immunization, a protective amount of antibody may persist in the blood for some years and a subsequent booster injection may maintain protection for a further decade. Much depends upon circumstances that will vary for different vaccines and different groups of people. The duration of active protection cannot be absolutely equated with the presence of demonstrable antibody because factors such as the sensitivity of the test and the actual protective role of the antibody detected have to be taken into consideration.
Age of commencement of active immunization
This must take account of the immaturity of the antibody-forming system in the very early months (see above) and the infectious challenges that a person may encounter early in life and in later years. The start of any immunization programme must be adjusted to the known epidemiology of the diseases that are prevalent in the country in which it is to be instituted, and it must also be related to any serious infective challenges that may be imported from time to time. Poliomyelitis is a good example of a disease that has been largely eradicated from many communities; however, the disease is quick to strike back if the immunization shield is lowered.
Controlled studies of vaccines
Combined field and laboratory studies aim to provide confidence in the efficacy of vaccines. A field trial can show only whether or not the actual preparation of vaccine used was successful under the circumstances prevailing at the time. Accordingly, trials require very sophisticated design and much care in their execution. In order to satisfy the requirement for reproducibility, the method of preparation of the vaccine must be meticulously described and controlled. Much work continues to define and to refine laboratory tests for the protectiveness of a particular vaccine that might be correlated with its efficacy in field trials. The laboratory test that gives results most closely corresponding to the protective value in the field trials may then be adopted as the test for standardizing future batches of vaccine.
Manufacturers of vaccines for commercial use are required in the UK and in many other countries to satisfy certain standards relating to the purity, safety, potency and stability of their products. In addition, the World Health Organization (WHO) has established internationally agreed requirements. At the national level, a system for continuing surveillance of the efficacy of prophylactic vaccines with continuing notification of any suspected adverse reactions is essential.
Contraindications to the use of vaccines
In the delivery of an important immunization programme there is a balance between the rights of the individual (and often this is the parent or carer not the person being immunized) and the benefit to the public. It is necessary not to take too legalistic a view of theoretical hazards and thus err on the side of opting out. The risk of opting out of an immunization schedule should be clearly appreciated and shown to be greater than the adverse effects.
There are some useful general principles about contraindications:
• do not give a vaccine to a patient with an acute illness
• do be sure that the postponed immunization is subsequently given
• do not give a live vaccine to a pregnant woman, unless there is a clear balance of risk in favour of vaccination
• avoid giving any vaccine in the first trimester of pregnancy
• do not give live vaccines to patients receiving immunosuppressive drugs or irradiation, or to patients suffering from malignant conditions of the reticulo-endothelial system – delay until after successful therapy when they are in remission.
Experience with human immunodeficiency virus (HIV) antibody-positive patients with or without the signs and symptoms of the acquired immune deficiency syndrome (AIDS) indicates so far that measles, mumps, rubella and polio (live virus) vaccines can be given, but that BCG vaccine should not be given. Inactivated vaccines, e.g. pneumococcal, are not contraindicated and are of great benefit.
Hazards of immunization
Possible adverse reactions to immunization are:
• mild or moderate pain at the site of injection
• fever and malaise for a day or two after
• anaphylactic reactions are very rare but, as they may be fatal, doctors and nurses should be aware of the possibility and should be prepared for such an emergency by carrying drugs and equipment for resuscitation (a ‘shock box’) to all immunization sessions.
During the first few years of life when many vaccines are given, children tend to have various health problems that include occasional febrile convulsions and may, sadly, include an unexplained cot death or other tragedy. It is inevitable that some of these events will coincide with the period shortly after a vaccine was given to a child and the possibility of a causal relationship will be entertained. The probability of such a link certainly deserves to be considered, but it is most important to bear in mind that the issue is emotive and that ill-balanced or ill-informed adverse publicity can do irreparable harm to an immunization programme. For example, annual notifications of whooping cough in England and Wales dropped from more than 100 000 in the early 1950s to some tens of thousands in the late 1950s as the vaccine gained ground. By the early 1970s (and after modification of the vaccine to take account of a possible deficiency in its protective cover), whooping cough was largely controlled in the UK, with about 75% of children immunized. In the late 1970s, after some ill-informed adverse publicity, acceptance rates for the vaccine fell steeply to 30% or lower, and the uptake of other vaccines was also affected. Epidemics of whooping cough followed in the UK in 1978 and 1982, with tens of thousands of children affected and a number of deaths. As a result of efforts to restore confidence in pertussis vaccine, uptake figures increased in the 1980s and the disease again began to be brought under control. Preliminary results of tests with new (acellular) pertussis vaccines indicate that these are safe and probably more effective.
Over the past few years there has been considerable publicity about claims that autism and inflammatory bowel disease were associated with MMR vaccine. Although several well-conducted studies have failed to confirm the original study, which first drew attention to this hazard, the media in the UK have supported the hypothesis and cover of the vaccine fell to the point that cases and deaths from measles reemerged. Mathematical models of the dynamics of measles in a population show that if the number of susceptibles in the population exceeds 5% there is a possibility of transmission.
Children who are most vulnerable are likely to be least protected. Pertussis, polio and measles spread most effectively when living conditions are overcrowded and unhygienic. To some extent, these diseases depend upon population density for their spread. Some underprivileged groups in a community are in very real danger when the average rate of vaccine uptake falls, because they often have the lowest cover.
In view of the stringency of the regulations that control the quality and efficacy of vaccines, the probability of error lies more with the vaccinator than with the producer. There is a special obligation to ensure that vaccines are properly stored, reconstituted (if relevant) and correctly administered (Box 70.2). Many preparations rapidly lose their potency if frozen and thawed or exposed to temperature variation. This means it is vital to maintain the ‘cold chain’ in which the vaccines are held between 2–8°C. The appropriate instructions and local protocols should be followed in detail. An injectable vaccine must be given with a sterile syringe and needle, a separate sterile syringe and needle must be used for each injection, and the equipment must be disposed of properly. The dangers of contamination with blood-borne viruses such as HIV and hepatitis B and C have led to the search for alternative methods of delivery by air jets or via mucosal surfaces.
Site of injection
This will vary with the vaccine to be given. Specific instructions should be followed. In general, and with the exception of BCG, injectable vaccines are given by intramuscular or deep subcutaneous injection. The anterolateral aspect of the thigh or upper arm is the preferred site for infants. Some doctors and nurses use the upper outer quadrant of the buttock, but fat in this area may interfere with the efficacy of the vaccine, especially hepatitis A and B. In future, the technological advances in delivery methods mentioned above will revolutionize the immunization clinics feared by countless school children.
Herd immunity
When most people in a community are immune to a particular infection that is spread from person to person, the natural transmission of the infection is effectively inhibited. Thus, if almost all children in a residential school have been immunized against measles, the school is most unlikely to have an outbreak of measles; even the few children who have not been immunized will enjoy a measure of protection afforded by the general herd immunity in that they will not be challenged within the school. This will only apply so long as the school population is largely composed of immune pupils and a nonimmune pupil does not encounter a visitor who is infected with measles. If there is an influx of susceptibles, the level of herd immunity will fall and the general protection will be lost. When the pupils go into other communities at holiday times, the nonimmune individuals are liable to get measles at their first contact with an infective case.
For herd immunity to operate well in a community or a country, vaccine uptake rates must exceed 90%. For some highly transmissible infections, such as measles, uptake rates above 95% are the target. Bear in mind that herd immunity operates only for infections transmitted from person to person. Tetanus is not transmitted in this way; a nonimmune person is fully vulnerable to tetanus even if he or she is surrounded by fully immunized colleagues in a closed community (Fig. 70.1).
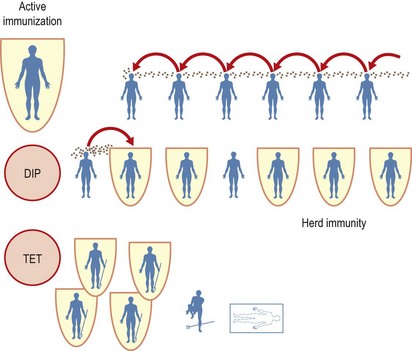
Fig. 70.1 If almost all of the members of a community receive active immunization against a disease that is normally transmitted from person-to-person, the resulting herd immunity confers some advantage even upon an occasional nonimmune member because rapid transmission of the disease through the community is prevented. This is true for diphtheria, but not for tetanus, which is not dependent on person-to-person spread.
Immunization programmes
An immunization campaign carried out without provision for its continuation as a routine procedure will not give satisfactory results unless complete eradication of the disease is achieved. Thus, in planning immunization schedules, consideration must be given to ensuring that the general public is receptive and understands the policy. It is essential to secure the trust and cooperation of parents who have to bring their children to the doctor or clinic for a series of inoculations and who will undoubtedly seek reassurance that the benefits are considerable and the risks negligible. Appropriate consent must be obtained for each immunization.
In the planning and execution of a programme, immunological points that merit special attention are:
Immunization schedules
The provision of a programme of active immunization to a community should be governed by considerations of need, efficacy, safety and ease of administration. An overriding consideration is the cost and the availability of skilled manpower and the safe delivery and supply of vaccines. Circumstances vary widely in different countries, and priorities vary. The WHO Expanded Programme on Immunization (EPI) has been adopted by most countries of the world as a minimum schedule to protect children in parts of the world where transmission rates are particularly high. Single measles is given at about 9 months rather than the combined MMR given at 12–15 months in industrialized countries. This is a balance between mean age of infection and response to the vaccine. Also, the EPI recommends hepatitis B immunization from birth (see below).
In the UK, it is generally agreed that protection of the susceptible population against diphtheria, whooping cough and tetanus, poliomyelitis, Haemophilus influenzae type b (with an inactivated conjugated Hib vaccine), Neisseria meningitidis group C (with protein conjugated to group polysaccharide as menC), common serogroups of Streptococcus pneumoniae, and mumps, measles and rubella (with a combined live vaccine) merit priority in the very early years of life. BCG vaccine is offered to some high-risk populations neonatally. In the UK it is now recommended that the DTP Hib and polio vaccine are started early as a single injection given at the same time as menC, at 2 months, with further doses at 3 and 4 months (Table 70.1). This secures earlier protection against pertussis in the months when the disease is most dangerous to the young child.
Table 70.1 Schedule of immunization for children in the UK*

Notes on some vaccines in common use
Adsorbed tetanus toxoid
The preparation in routine use in the UK is adsorbed onto an aluminium salt; it is more effective and less reactive than simple toxoid. A course of three injections given at intervals of about 4 weeks and boosters prior to, and on leaving, school, i.e. a total of five doses, should protect for life, although studies show that the elderly have low levels of protective antibody. Prevention of tetanus should be considered in relation to the management of wounds (see Ch. 22, p. 251).
Diphtheria toxoid
The adsorbed preparation used in the UK and incorporated into the triple DTP vaccine contains aluminium phosphate and affords good protection which is boosted at school entry age. This preparation is not used for adults; a dilute adsorbed diphtheria vaccine preparation is used for people who are more than 10 years old and is now incorporated with tetanus toxoid for school leavers as Td. Active immunization of adults against diphtheria is not practised in the UK as a routine, unless there is a potentially high occupational risk of encountering the organism in laboratory or clinical work, or unless a person has been in contact with a case of diphtheria or is travelling to an endemic area and protection is deemed necessary. A course of erythromycin gives further protection to a close contact, and this has replaced the use of diphtheria antitoxin in contacts. The falling level of immunity to diphtheria among adults in communities that regard themselves as having good preventive medical services gives some cause for concern and calls for continuing vigilance.
Pertussis vaccine
The vaccines in general use are either whole-cell preparations of killed Bordetella pertussis or acellular vaccines containing antigenic components of the organism. The protection afforded by the whole-cell vaccines is generally acknowledged to be considerable, but the acellular vaccine seems to be more effective and appears to have fewer adverse reactions so it is now included in the UK schedule. Adverse reactions that may be associated with pertussis vaccine include soreness at the site of injection, irritability and pyrexia. Reactions such as persistent screaming, shock, vomiting and convulsions have been reported, but the association with pertussis vaccine is not invariably clear.
Haemophilus influenzae type b (Hib) vaccine
Invasive encapsulated strains of H. influenzae, almost always of serotype b, are associated with meningitis, bacteraemia, epiglottitis and other serious infections particularly affecting young children. The risks of severe morbidity and mortality are highest in children in the age range 3–18 months. Hib conjugate vaccines containing the capsular polysaccharide linked to a protein are immunogenic and reduce pharyngeal carriage of the organism. The Hib vaccines used in the UK are given combined as part of the primary immunization. Children in the age range 1–4 years who have not received Hib vaccine can be protected by a single dose of Hib vaccine. Routine immunization of older children or adults with Hib vaccine is not generally recommended unless they are immunocompromised.
Meningococcal group C (MenC) vaccine
In 1999, following an increase in fatal cases of invasive meningococcal disease especially due to group C, the UK government was the first in the world to introduce MenC vaccine to the routine programme. Although it is now given as part of primary immunization at 3, 4 and 13 months, when the campaign started it was necessary to protect the whole population up to 18 years of age. The design of the antigen is similar to Hib in that the relatively poorly immunogenic polysaccharide is covalently linked to a protein carrier which produces a prolonged immune response in naïve infants. As with Hib there are few adverse reactions and the introduction universally to children has led to a great reduction in cases of meningitis and carriage in the nasopharynx of group C meningococci.
Poliomyelitis vaccines
The control of poliomyelitis is one of the great success stories of active immunization, and the worldwide eradication of poliomyelitis is a goal of the WHO which has nearly been achieved. The Salk vaccine came first; this is a killed mixture of the three types of poliovirus (1, 2 and 3). A course of three injections is given at appropriate intervals and in the UK it is combined with the other infant vaccines.
The oral polio vaccine (OPV) was favoured in the UK and many other countries. It is the live-attenuated form developed by Sabin. This is a mixture of the three types, and it is given orally on three occasions, usually at the same time as the infant vaccinations in the early months. As it is a live preparation, the vaccine must be stored and held according to the manufacturer’s instructions. OPV colonizes the gut and gives rise to local and humoral antibodies. The faeces contain live virus for some time, and poliomyelitis caused by a vaccine strain is a rare, but recognized, hazard which is why at the end stage of eradication of the disease it is recommended for countries to swap to the injectable form.
Pneumococcal vaccines
The 23 valent polysaccharide vaccine has been available for over three decades but its place in universal protection of vulnerable patients has not been established. Splenectomized and immunocompromised people should be routinely immunized preferably before surgery or therapy. A conjugate vaccine, produced initially against 7 types of pneumococci is commercially available in many countries and with the inclusion of other common serotypes, has been used either for universal vaccination or to protect infants at highest risk. There is evidence from North America that the universal infant programme has produced a significant decrease in pneumococcal disease in the elderly. There is hope that these vaccines will become more widely available in resource-poor countries where the need is greatest.
Measles, mumps and rubella (MMR) vaccine
This is a mixture of live-attenuated strains of these three viruses in freeze-dried form; it has to be stored at 2–8°C (not frozen), reconstituted according to the manufacturer’s instructions and used promptly. One injection of the mixture is given between 12 and 15 months in the UK schedule, and the second dose in the fifth year, before entry to school.
The measles component may give rise to fever or malaise and sometimes a rash about a week after inoculation. The mumps component may cause some parotid swelling, seen about 3 weeks after inoculation. Very occasional cases of mumps vaccine-associated meningo-encephalitis occur; these generally occur at 3 weeks after use of the Urabe strain. It is usually benign, but there may be confusion with other more serious syndromes in this age group. The rubella component is not usually associated with reactions in this age group, though temporary malaise, mild fever and arthralgia occurring about the ninth day after vaccination have been noted in some older recipients of rubella vaccine.
It is hoped that high vaccine coverage with two doses of MMR vaccine in the UK schedule will eradicate measles, mumps and rubella in this country as it nearly has done in North America. Much depends upon public faith in the immunization programme, which has been undermined by media-led campaigns about the safety of MMR.
All seronegative women of childbearing age and seronegative professional attendants who might come into contact with pregnant women should be offered rubella vaccine after a test for immunity. Although it is not thought to be teratogenic, any woman of childbearing age who is given the vaccine should avoid pregnancy for a month.
BCG
The attenuated strain of bovine tubercle bacillus known as bacille Calmette–Guérin (BCG) produces cross-immunity to human tuberculosis and has significantly contributed to the control of that disease and to the other important mycobacterial disease, leprosy, in many countries. An intradermal injection of the live-attenuated vaccine is given on the lateral aspect of the arm at the level of the deltoid insertion, but not higher, or on the upper lateral surface of the thigh. Instruction on the reconstitution of the freeze-dried vaccine, the dosage and the detailed technique of giving a truly intradermal injection, should be most carefully observed. With the exception of newborn children, any recipient of BCG vaccination should have been tested for hypersensitivity to tuberculin and found to be negative. Tuberculin tests include the Mantoux test, in which diluted tuberculin is injected intradermally, and the Heaf test, which is done with a multiple puncture apparatus (see Ch. 18).
Hepatitis B vaccine
For the protection of groups of people considered to be at special risk of acquiring hepatitis B, a bioengineered vaccine is now in common use. A course of three intramuscular injections is given (not into the buttock) at intervals of 1 and 5 months; thus, it takes 6 months to complete the course, and this is of practical importance. Following needlestick injuries or known contact with the virus it is possible to give an accelerated course although efficacy may not be as good. Protective antibody responses are generally achieved in about 90% of those given the vaccine, with a range of responses from weak to strong, but there is a worrying minority of nonresponders (10–15% in those over 40 years of age). Antibody responses should be checked at least 6 weeks after completion of a course, and it may be prudent to give poor responders or nonresponders a booster dose or a repeat course. Nonresponse rates are particularly worrying among patients on maintenance haemodialysis, for whom a higher dose preparation is available. In the general population, the protection afforded to responders is thought to last for about 5 years, when a booster dose of vaccine may be given. Established policy must await further experience with hepatitis vaccines, which in many countries are being given universally as part of the routine schedule. Great success has been claimed in Asia in reducing prevalence and preventing cancer.
Other vaccines
Various vaccines are available for the protection of special groups of people or for individuals in special circumstances (Box 70.3). The reader is referred to the appropriate chapters for more detailed consideration of such topics as:
• the prevention of tetanus in wounded patients (Ch. 22)
• the indications for pneumococcal vaccine (p. 197)
• the management of rabies (Ch. 58)
• the control of Q fever (Ch. 40)
• the protection of those at special risk from chickenpox or hepatitis B (Chs. 43 & 46)
• the protection of the immunocompromised such as HIV/AIDS patients (Ch. 55)
• the prevention of cervical cancer by human papillomavirus vaccines (Ch. 45)
• the indications for rotavirus vaccine (Ch. 54).
Protecting the traveller
An intending traveller to another country should seek advice in advance about the prevailing diseases and the precautions that should be taken. Advice on immunization is unlikely to be of much help if the traveller unwisely runs the risk of drinking raw water or eating uncooked salads and vegetables in a country where sanitation is inadequate and water supplies are insecure.
In advising travellers about exotic diseases, do not forget that diseases now uncommon in countries with developed medical services may still be common in countries that lack such services. Healthcare workers need to include the risk of diphtheria and check immunity to tuberculosis, hepatitis B, measles and poliomyelitis. If the traveller is going to Central Africa or Central America, bear in mind the need for protection against yellow fever or to Mecca for the Hajj to include meningitis vaccine. Other specific vaccines may be indicated such as those against rabies, typhoid and encephalitis. Some of the special vaccines listed in Box 70.3 may also merit inclusion, especially if the traveller’s activities when abroad are likely to expose him or her to the relevant diseases. Japanese B encephalitis vaccine may provoke more adverse events than the risk entailed. Travellers to rural areas in the tropics must always put insecticides and antimalarials on the top of their list of necessities because although there is some progress in the development of an effective malaria vaccine none is available commercially. Practical, simple advice is often of more value than a typhoid or hepatitis immunization.
Unresolved problems
No product is perfect (Box 70.4). Some vaccines are more imperfect than others, and some circumstances pose special problems. The influenza virus has shifts and drifts in its antigenic pattern, so vaccines must be frequently updated. Then decisions have to be made on the patients who merit this special protection. As these include old people and many other patients with chronic medical conditions, the logistics are quite difficult. Most temperate climate countries immunize the elderly and vulnerable before winter and this has been shown in the USA to reduce mortality and hospital admissions.
Cholera vaccines have had a chequered record, with very little evidence of real efficacy for the traveller, who is much better protected by a basic knowledge of hygiene. Results with the use of the oral vaccine are more promising and may protect against ETEC (enterotoxigenic E. coli) as well as cholera.
Acute purulent (bacterial) meningitis is a dramatic clinical problem. The MenC vaccine has afforded protection against group C meningitis and group A has been widely used in sub-Saharan Africa, but a group B vaccine is not routinely available at the time of writing and group B strains are more common in many areas.
Several vaccines have been developed against various herpesviruses, including herpes simplex, varicella–zoster and Epstein–Barr viruses. There have been many difficulties, but a live-attenuated varicella vaccine has been developed which is useful for susceptible staff who work in paediatric or maternity wards and is given universally to all infants in some countries.
A vaccine effective against HIV is being urgently sought in the fight against AIDS. There have been many disappointments, and there is now a glimmer of hope. Advances in molecular technology will herald a new generation of immunogens such as DNA vaccines. Delivery of products without using traditional needles and syringes is another target that is getting nearer and will save infections caused by the reuse of unsterile equipment.
Anonymous: Immunological products and vaccines. In: British National Formulary. British Medical Association and Royal Pharmaceutical Society of Great Britain, London (revised at intervals of about 6 months)
Centers for Disease Control and Prevention. Brunette GW, ed. CDC Health Information for International Travel 2012. Oxford and New York: Oxford University Press, 2011.
Department of Health, Welsh Office, Scottish Home and Health Department. Health Information for Overseas Travel. London: HMSO; 2001.
Department of Health, Welsh Office, Scottish Home and Health Department, DHSS (Northern Ireland). Immunisation against Infectious Disease. London: HMSO; 2006.
Kassianos GC. Immunization: Childhood and Traveller’s Health, ed 4. Oxford: Blackwell; 2001.
Mackett M, Williamson JD. Human Vaccines and Vaccination. Oxford: Bios Scientific Publications; 1995.
Plotkin S, Orenstein W, Offit P. Vaccines, ed 5, Philadelphia: WB Saunders, 2008.
World Health Organization. International Travel and Health. Vaccination Requirements and Travel Advice. Geneva: WHO; 2000.
British National Formulary. http://www.bnf.org/.
Centers for Disease Control and Prevention. Travelers’Health. http://www.cdc.gov/travel/.
Centers for Disease Control and Prevention. Vaccines & Immunizations. http://www.nc.gov/vaccines/.
Department of Health (England). Green book. Immunisation against infectious disease. http://www.dh.gov.uk/en/Publichealth/Immunisation/Greenbook/index.htm.
Department of Health (England). Immunisation. http://www.dh.gov.uk/en/Publichealth/Immunisation/index.htm.
Health Protection Agency. http://www.hpa.org.uk/.
Health Protection Scotland. http://www.hps.scot.nhs.uk/.
National Travel Health Network and Centre. http://www.nathnac.org/.
NHS (Scotland). Fit For Travel. http://www.fitfortravel.nhs.uk/.
World Health Organization. Immunization, Vaccines and Biologicals. http://www.who.int/immunization/.
World Health Organization. International travel and health. http://www.who.int/ith/.