24 Salmonella
Food poisoning; enteric fever
Key points
• There are more than 2000 different antigenic types of Salmonella; those pathogenic to man are serotypes of S. enterica.
• Most serotypes of S. enterica cause food-borne gastroenteritis and have animal reservoirs.
• S. enterica serotypes Typhi and Paratyphi cause typhoid fever.
• Typhoid and other serious systemic salmonella infections are treated with amoxicillin, co-trimoxazole, ciprofloxacin or chloramphenicol.
• Antibiotics have no place in the management of salmonella gastroenteritis unless invasive complications are suspected.
• Clean water, sanitation and hygienic handling of foodstuffs are the keys to prevention.
There are well over 2000 different antigenic types of salmonella. They were originally classified as separate species, but it is now generally accepted that they represent serotypes (serovars) of a single species, Salmonella enterica. Various subspecies are recognized, but most of the serotypes that infect mammals are found in a subspecies also designated enterica. For example, the full designation of the serotype formerly called Salmonella enteritidis is: Salmonella enterica subspecies enterica serotype Enteritidis. This cumbersome nomenclature is often abbreviated to Salmonella Enteritidis (S. Enteritidis) and this convention will be followed here in considering those salmonellae responsible for human infections.
Certain serotypes are a major cause of food-borne infection worldwide. Most infections are relatively benign and restricted to the intestinal tract, causing a short-lived diarrhoea, but some S. enterica serotypes, notably Typhi and Paratyphi, cause life-threatening systemic disease.
Description
Salmonellae are typical members of the Enterobacteriaceae: facultatively anaerobic Gram-negative bacilli able to grow on a wide range of relatively simple media and distinguished from other members of the family by their biochemical characteristics and antigenic structure. Their normal habitat is the animal intestine.
Antigens
Typical strains of S. enterica express two sets of antigens, which are readily demonstrable by serotyping. Long-chain lipopolysaccharide (LPS) comprises heat-stable polysaccharide commonly known as the somatic or ‘O’ antigens. These molecules are located in the outer membrane and are anchored into the cell wall by antigenically conserved lipid A and LPS-core regions. The long-chain LPS molecules exhibit considerable variation in sugar composition and degree of polysaccharide branching, and this structural heterogeneity is responsible for the large number of serotypes. Salmonellae are usually highly motile when growing in laboratory media, and flagellar protein subunits contain the epitopes that form the basis of the flagella-based serotyping scheme generally known as the ‘H’ antigens (Fig. 24.1). In most strains of S. enterica the flagella exhibit the property of diphasic variation, whereby one of two genetically distinct flagellar structures are expressed. When one flagellar structure is expressed it contains phase 1 antigens, whereas when the other set is operative phase 2 antigens are synthesized.
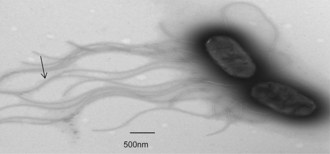
Fig. 24.1 Electron micrograph of S. Typhi. Arrow indicates the flagella that carry the ‘H’ antigens. Bar = 500 nm.
Certain serotypes of S. enterica express a surface polysaccharide, of which the Vi (virulence) antigen of S. Typhi is the most important example. As the polysaccharide may encapsulate the entire bacterium, antibodies designed to recognize the LPS antigens may be prevented from binding; this can occasionally make detection of the O antigens difficult.
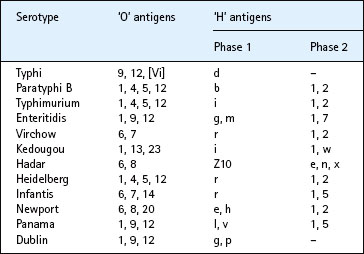
The various O-antigens of salmonellae are numbered with Arabic numerals. The flagellar antigens of phase 1 are designated by lower-case letters, and those of phase 2 by a mixture of lower-case letters and Arabic numerals. The antigenic structure of any serotype of salmonella is thus expressed as an antigenic formula, which has three parts, describing the O antigens, the phase 1 H antigens and the phase 2 H antigens, in that order. The three parts are separated by colons, and the component antigens in each part by commas; for example, the distinctive antigenic formula of S. Enteritidis is 1, 9, 12: g, m: 1, 7.
The original Kauffmann–White scheme, which elegantly catalogued salmonellae (but named them as individual species), placed them into some 30 groups on the basis of shared O antigens, and further subdivided the groups into clusters with H antigens in common. Some salmonellae, such as S. Typhi (9, 12, [Vi]: d), express only one flagellar phase. Some of the commoner serotypes are shown in Table 24.1.
Typing methods
Phage typing schemes have proved extremely useful for discriminating within strains of S. enterica serotypes Typhimurium, Virchow, Enteritidis and Typhi. The numbers of phages comprising the various schemes are constantly being increased to improve strain discrimination.
Characterization of strains of S. enterica is also assisted by determining their distinctive patterns of resistance to a range of antibiotics. For example, certain strains of S. Typhimurium definitive type (DT) 104 are characterized by resistance to ampicillin, chloramphenicol, aminoglycosides and co-trimoxazole.
Pulsed-field gel electrophoresis analysis, of the entire Salmonella genome following digestion with selected enzymes, has facilitated strain discrimination in outbreak situations.
Host range and pathogenicity
Strains of S. enterica are widely distributed in nature. All vertebrates appear capable of harbouring these bacteria in their gut, and certain serotypes have also been isolated from a wide range of arthropods, including flies and cockroaches. Most animal infections seem to range from those without symptoms to those resulting in self-limiting gastroenteritis of variable severity. Some strains, such as those belonging to serotype Typhimurium, show a wide host range and can be isolated from many different animal species. A small number of strains, the host-adapted serotypes, are much more restricted in the species they inhabit, and show a different spectrum of illness.
Among the host-adapted serotypes, Typhi and Paratyphi A and C are rarely, if ever, isolated from animals other than man. Paratyphi B, although essentially a human pathogen, is occasionally isolated from cattle, pigs, poultry, exotic reptiles and other animals, although cycles of transmission in these hosts have not been demonstrated. Human infection with these organisms is characterized by a long incubation period of 10–14 days, followed by a septicaemic illness, enteric fever, quite unlike the diarrhoea and vomiting that are characteristic of food poisoning.
Other salmonellae adapted to particular animal hosts include Cholerae-suis (pigs), Dublin (cattle), Gallinarum-pullorum (poultry), Abortus-equi (horses) and Abortus-ovis (sheep). These are all responsible for considerable morbidity, mortality and economic loss among domestic animals. All can cause human illness, but only strains of Cholerae-suis and Dublin do so regularly. Strains of S. Typhimurium DT 104 isolated from human infection appear to have originated from bovine sources; but whether these strains can be considered as host-adapted to farm animals remains to be established. The rest of the 2000 or so serotypes of salmonellae show no apparent host preference. The extent to which they cause human infection appears to result from their prevalence in domestic food animals at any particular time and on the opportunity for contamination of food in which further multiplication can take place. In developed countries most human infections are caused by a relatively small number of locally prevalent serotypes.
Pathogenesis
In common with many pathogenic enteric bacteria, strains of S. enterica require a range of pathogenic mechanisms to enable them to survive passage through the digestive tract, colonize a host and cause disease. The lack of a good animal model of Salmonella pathogenicity has impeded research into how these bacteria cause disease; however, it is anticipated that the availability of gene sequences will allow the mechanisms to be understood more fully. In general terms, infection is initiated by the ingestion of a sufficient number of organisms to survive the stomach acid barrier and the effects of digestive bile prior to colonization of the gut mucosa, and to express the mechanisms resulting in overt disease.
Infective dose
For human infections, the number of bacteria that must be swallowed in order to cause infection is uncertain and varies with the serotype. The accepted dictum that large inocula of these bacteria are required for induction of human illness is based largely on volunteer studies. In most of these the median infective dose for most serotypes, including Typhi, has varied from 106 to 109 viable organisms. However, investigation of outbreaks suggests that in natural infection the infective dose might be fewer than 1000 viable organisms.
Many factors are thought to influence the infective dose. There appears to be considerable strain-to-strain variation in virulence even within a single serotype. Systematic variation in pathogenicity between serotypes is less easy to demonstrate outside the host-adapted strains. The vehicle of ingestion may also influence pathogenesis. Organisms ingested in water and other drinks may be carried through the stomach relatively rapidly, and evade the effect of gastric acid. Similarly, the administration of antacids, or the effects of gastric resection, reduces the infective dose. Bacteria within particles of food would also evade the action of stomach acids.
Host factors
Host factors are also likely to be important, particularly the nutritional and immune status of the host. Host variables can, however, be confusing; for example, age-specific isolation rates for salmonellae, as for some other gut pathogens, are higher for children less than 1 year of age than for any other age group, but this reflects the fact that a higher proportion of infections are investigated in this age group.
Initiation of infection
Once salmonellae enter the lumen of the intestine they need to be able to tolerate the action of digestive bile and compete with the prevailing gut flora for adhesion sites on the gut mucosa. Certain serotypes, such as S. Typhimurium, express type-1 fimbriae, which enable them to adhere to α-mannose-containing molecules on the microvilli of the ileal mucosa; however, surprisingly little is known about the range of fimbriae expressed. Strains of S. Enteritidis are thought to express at least three different fimbrial structures (Fig. 24.2).
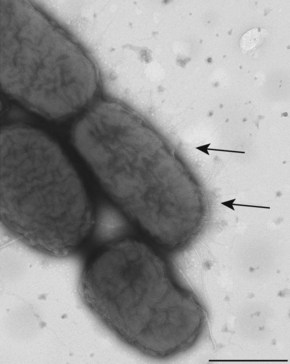
Fig. 24.2 Electron micrograph of S. Enteritidis. Arrows indicate fimbriae. Bar = 0.5 µm.
(From Zuckerman AJ 2004 Principles and practice of clinical virology, 5th edn. John Wiley & Sons Ltd, Chichester.)
Salmonella serotypes such as Typhimurium and Enteritidis also express an adhesion mechanism that does not involve fimbriae. Certain strains of enteric bacteria carry deoxyribonucleic acid (DNA) sequences that encode several pathogenic mechanisms, termed pathogenicity islands. In common with strains of Verocytotoxin-producing Escherichia coli belonging to serogroup O157 (see Ch. 26), strains of S. Typhimurium and S. Enteritidis have pathogenicity islands that encode an adhesion mechanism comprising both a bacterial adhesin and the adhesin receptor, which is translocated into the host intestine. This process enables these bacteria to insert their own binding site into the gut, unlike fimbriae, which require host-derived binding sites located in the intestinal wall.
Attachment to the host mucosa is followed by degeneration of the microvilli to form breaches in the cell membrane through which the salmonellae enter the intestinal epithelial cells. For certain strains, further multiplication in these cells and in macrophages of the Peyer’s patches follows. Some bacteria penetrate into the submucosa and pass to the local mesenteric lymph nodes. All of the clinical manifestations of infection with salmonella, including diarrhoea, begin after ileal penetration where inflammation of the ileal mucosa results in the efflux of water and electrolytes resulting in diarrhoea. For strains of S. Typhi, infection involves invasion of the bloodstream and various organs.
Clinical syndromes
Although salmonellae can cause a wide spectrum of clinical illness there are four major syndromes, each with its own diagnostic and therapeutic problems:
Although uncommon, infection with Salmonella can result in sequelae, including reactive arthritis (Reiter’s syndrome).
Enteric fever
Enteric fever is caused by strains of S. Typhi or S. Paratyphi A, B or C; although S. Paratyphi B, which gene sequence analysis suggests is a variant of S. Java, is more likely to cause non-typhoidal diarrhoea. The clinical features tend to be more severe with S. Typhi (typhoid fever). After penetration of the ileal mucosa the organisms pass via the lymphatics to the mesenteric lymph nodes, whence after a period of multiplication they invade the bloodstream via the thoracic duct. The liver, gall bladder, spleen, kidney and bone marrow become infected during this primary bacteraemic phase in the first 7–10 days of the incubation period. After multiplication in these organs, bacilli pass into the blood, causing a second and heavier bacteraemia, the onset of which approximately coincides with that of fever and other signs of clinical illness. From the gall bladder, a further invasion of the intestine results. Peyer’s patches and other gut lymphoid tissues become involved in an inflammatory reaction, and infiltration with mononuclear cells, followed by necrosis, sloughing and the formation of characteristic typhoid ulcers occurs.
Onset
The interval between ingestion of the organisms and the onset of illness varies with the size of the infecting dose. It can be as short as 3 days or as long as 50 days, but is usually about 2 weeks. The onset is usually insidious. Early symptoms are often vague: a dry cough and epistaxis associated with anorexia, a dull continuous headache, abdominal tenderness and discomfort are among the most common symptoms. Diarrhoea is uncommon and early in the illness many patients complain of constipation.
Progression
In the untreated case the temperature shows a stepladder rise over the first week of the illness, remains high for 7–10 days and then falls during the third or fourth week. Physical signs include a relative bradycardia at the height of the fever, hepatomegaly, splenomegaly and often a rash of rose spots. These are slightly raised, discrete, irregular, blanching, pink macules, 2–4 mm in diameter, most often found on the front of the chest. They appear in crops of up to a dozen at a time and fade after 3–4 days, leaving no scar. They are characteristic of, but not specific for, enteric fever.
Relapse
Apparent recovery can be followed by relapse in 5–10% of untreated cases. Relapse is usually shorter and of milder character than the initial illness, but can be severe and may be fatal. Severe intestinal haemorrhage and intestinal perforation are serious complications that can occur at any stage of the illness.
Morbidity and mortality
Classical typhoid fever is a serious infection that, when untreated, has a mortality rate approaching 20%. It is notoriously unpredictable in its presentation and course. Mild and asymptomatic infections are not uncommon. In endemic areas, and particularly where it co-exists with schistosomiasis, chronic infection can present with fever of many months’ duration, accompanied by chronic bacteraemia. Occasionally, diarrhoea may dominate the picture from the outset, particularly in paratyphoid infections, which sometimes present as typical gastroenteritis no different from that caused by most S. enterica serotypes.
Gastroenteritis and food poisoning
Acute gastroenteritis is characterized by vomiting, abdominal pain, fever and diarrhoea. It can be caused by ingestion of a wide variety of bacteria or their products, several viruses, and a number of vegetable toxins and inorganic chemicals. The term bacterial food poisoning is conveniently restricted to cases and epidemics of acute gastroenteritis that are caused by the ingestion of food contaminated by bacteria or their toxins. In bacterial food poisoning, the bacteria need the opportunity to multiply in the food to reach an infective concentration before being eaten. Infections such as hepatitis A or bacillary dysentery, in which food may be an incidental vector, are not usually considered to be examples of food poisoning. Strains of S. enterica commonly cause food poisoning worldwide.
Clinical features
The most common clinical manifestation of infection with non-invasive salmonella serotypes is diarrhoea, often accompanied by headache, malaise and nausea. The incubation period is usually 8–48 h, the onset abrupt, and the clinical course short and self-limiting. Symptoms vary from the passage of two or three loose stools, which may be disregarded by the sufferer, to a severe and prostrating illness with the frequent passage of watery, green, offensive stools, fever, shivering, abdominal pain and, in the most severe cases, dehydration leading to hypotension, cramps and renal failure. Vomiting is rarely a prominent feature of the illness.
Severe infections occur most often in the very young and the elderly, although mild subclinical infections also occur in these age groups. Infections with certain serotypes in those already ill or debilitated from other causes are likely to be more severe and life-threatening. In most cases the acute stage is over within 2–3 days, although it may be more prolonged. Persistent or high fever suggests bacteraemia, possibly with metastatic infection.
Bacteraemia and metastatic disease
Bacteraemia is a constant feature of enteric fever caused by strains of S. Typhi and Paratyphi A and C. Rarely, complications of infection with other salmonellae can occur. Transient bacteraemia occurs in up to 4% of cases of acute gastroenteritis, but in most cases the organisms are cleared from the bloodstream without ill effect.
Occasionally, dissemination of the bacilli throughout the body results in the establishment of one or more localized foci of persisting infection, especially where pre-existing abnormality makes a tissue or organ vulnerable. Atherosclerotic plaques within large arteries, damaged heart valves, joint prostheses and other implants are all susceptible to metastatic infection.
Osteomyelitis is most often found in long bones, costochondral junctions and the spine. Multiple bony sites may be affected, and sickle cell anaemia is an important predisposing factor. Suppurative arthritis can occur either as an extension of contiguous osteomyelitis or as a primary infection.
Meningitis is a particularly serious complication of infection in neonates and very young children. Abscess formation can occur in almost any organ or tissue. Even in the absence of obvious tissue damage, the ability of salmonellae to enter and survive within macrophages and other cells, particularly in the liver and biliary tree, but also in bone marrow and the kidney, leads occasionally to persistent infection and the chronic carrier state.
The prolonged carrier state
Most people infected with salmonella continue to excrete the organism in their stools for days or weeks after complete clinical recovery, but eventual clearance of the bacteria from the body is usual. A few patients continue to excrete the salmonellae for prolonged periods. The term chronic carrier is reserved for those who excrete salmonellae for a year or more. Chronic carriage can follow symptomatic illness or may be the only manifestation of infection. It can occur with any serotype, but is a particularly important feature of enteric fever: up to 5% of convalescents from typhoid and a smaller number of those who have recovered from paratyphoid fever become chronic carriers, many for a lifetime. The bacilli are most commonly present in the gall bladder, less often in the urinary tract, and are shed in faeces and sometimes in urine. The long duration of the carrier state enables the enteric fever bacilli to survive in the community in non-epidemic times and to persist in small and relatively isolated communities.
Age and sex are important determinants of the frequency of carriage, at least of S. Typhi. After enteric fever, fewer than 1% of patients under 20 years old become carriers, but this proportion rises to more than 10% in patients over 50 years of age. At all ages women become carriers twice as often as men.
The duration of excretion following infection with other salmonellae is less well documented, but more than 50% of patients stop excreting the organisms within 5 weeks of infection, and 90% of adults are culture negative at 9 weeks. The duration of excretion is significantly greater in children aged under 5 years, but virtually all permanent carriers are adults.
Laboratory diagnosis
Selective media, such as desoxycholate–citrate agar or xylose–lysine desoxycholate agar, are used for the isolation of salmonella bacteria from faeces. Fluid enrichment media, such as tetrathionate or selenite broth, are also useful to detect small numbers of salmonellae in faeces, foods or environmental samples. Suspicious colonies from the culture plates are tested directly for the presence of Salmonella somatic (O) antigens by slide agglutination and subcultured to peptone water for the determination of flagellar (H) antigen structure and further biochemical analysis. A presumptive diagnosis of salmonellosis can often be made within 24 h of the receipt of a specimen, although confirmation may take another day, and formal identification of the serotype takes several more days. A negative report must await the result of enrichment cultures – at least 48 h.
Several commercial PCR assays are available. In common with all PCR assays involving clinical, environmental and food samples, the numbers of bacteria and amounts of DNA can be below the levels required for successful amplification.
Enteric fever
Blood culture
Bone-marrow culture is the most reliable method for the diagnosis of enteric fever. The organisms may also be recovered from the bloodstream at any stage of the illness, but are most commonly found during the first 7–10 days and during relapses. The organisms can also be recovered from the blood clot from a sample taken for serological tests. The clot is digested with streptokinase or minced, and incubated in broth.
Stool and urine culture
Specimens of faeces and urine should be submitted for examination, although the isolation of salmonella from these specimens may indicate merely that the patient is a carrier. In typhoid fever, patients’ stools may contain salmonella from the second week and urine cultures from the third week of the infection. In paratyphoid B infections the clinical course may be much shorter than in typhoid; diarrhoea may occur early and stool cultures are often positive in the first week of the illness.
Serological tests
Infections with both invasive and non-invasive serotypes may induce specific serum antibodies to Salmonella surface antigens, although serological tests have been applied extensively only in the routine diagnosis of infection with S. Typhi and S. Paratyphi A, B and C. The Widal agglutination test, formerly used for the detection of specific O, H and Vi antigens, has been largely replaced by sensitive and specific methods such as enzyme-linked immunosorbent assay (ELISA) and immunoblotting. In interpreting the results of serology, account must be taken of antibodies acquired by previous infection or vaccination.
Cross-reacting antibodies from previous exposure to other salmonellae may confuse the results of serodiagnosis. For example, the O antigens of S. Typhi are expressed by other serotypes such as Enteritidis (see Table 24.1). Antibodies specific for group ‘d’ flagellar antigens can be used to differentiate infections caused by S. Typhi from other serotypes that share O antigens.
Use of serology in the search for typhoid carriers, for example in the routine examination of food handlers and waterworks employees, is of doubtful value.
Food poisoning
The laboratory diagnosis of bacterial food poisoning depends on isolation of the causal organism from samples of faeces or suspected foodstuffs. The more common food-poisoning serotypes, such as Enteritidis or Typhimurium, may be characterized more fully by phage typing and antibiotic resistance typing (see above). Strains can be differentiated further by plasmid and pulsed-field gel electrophoresis typing so that the isolates from patients may be matched with those from the infected food and from a suspected animal source.
Treatment
Enteric fever
The introduction of chloramphenicol in 1948 transformed a life-threatening illness of several weeks’ duration associated with a mortality rate of more than 20% into a short-lasting febrile illness with a mortality rate of less than 2%. Many patients can be treated adequately with oral chloramphenicol from the outset. Initial intravenous therapy with the drug may be necessary for the more severely ill patient, who may have anorexia, abdominal distension and, perhaps, vomiting. The intramuscular route gives inadequate blood levels. Treatment should be maintained for 14 days because relapse is more frequent with a shorter course.
The problem of bone marrow toxicity and the emergence of plasmid-mediated chloramphenicol resistance in many parts of the world prompted the search for alternative agents. Amoxicillin and co-trimoxazole are as effective as chloramphenicol, and are used widely. However, simultaneous resistance to these drugs has become increasingly common in strains of S. Typhi in several endemic areas, and imported multiresistant typhoid is being encountered worldwide. Fluoroquinolones such as ciprofloxacin and cephalosporins such as ceftriaxone and cefixime have emerged as the drugs of choice for the treatment of typhoid.
Gastroenteritis
Management of salmonella gastroenteritis includes replacement of fluids and electrolytes, and control of nausea, vomiting and pain. Drugs to control the hypermotility of the gut are contra-indicated; they may give symptomatic relief for a while, but it is easy to transform a trivial gastroenteritis into a life-threatening bacteraemia by paralysing the bowel.
Antibiotics have no part to play in the management in most cases. Randomized, placebo-controlled, double-blind studies have failed to show any benefit from any antibiotic on the duration and severity of the diarrhoea or the duration of fever; some antibiotics seemed to prolong the carrier state. If a patient is clearly at increased risk of bacteraemia and generalized invasion, an antibiotic may protect against this serious complication. Such patients include infants under 3 months of age, patients with a malignancy, haemoglobinopathy or chronic gastrointestinal disease such as ulcerative colitis (especially when treated with steroids), and patients who are immunosuppressed for other reasons. Treatment with an appropriate agent (see above) should continue until the gastroenteritis has resolved completely.
Salmonella bacteraemia
Established salmonella bacteraemia requires aggressive antimicrobial treatment with ciprofloxacin, chloramphenicol, co-trimoxazole or high-dose ampicillin. Uncomplicated bacteraemia should be treated for 10–14 days. A careful search for focal metastatic disease should be undertaken, especially when relapse follows cessation of treatment. Surgical drainage of metastatic abscesses may be required, with surgical intervention if heart valves or large vessels are affected.
In salmonella meningitis in infancy, treatment with chloramphenicol or ampicillin may be unsuccessful in up to a third of cases caused by sensitive strains. As cefotaxime and ceftriaxone penetrate into the cerebrospinal fluid reasonably well and are highly active against most salmonellae, they offer an effective alternative.
Resistance to any of the drugs used to treat invasive infection may occur, so treatment should be supported by susceptibility testing whenever possible.
Chronic asymptomatic carriers
The chronic carrier state presents a particularly difficult therapeutic challenge. The principal site of carriage is the biliary tract, and the presence of gall stones can influence the carriage of S. Typhi and S. Paratyphi A; concomitant biliary disease also has significant implications for therapy. When the patient has chronic cholecystitis or gallstones, antibiotics alone are most unlikely to eradicate the infection. Cholecystectomy together with appropriate antibiotic treatment results in cure in about 90% of cases, but has significant risk, not least from metastatic infection from dissemination of the organisms during surgery.
In the absence of biliary disease, prolonged courses of ampicillin, amoxicillin, co-trimoxazole or ciprofloxacin may cure up to 80% of carriers. However, it is difficult to justify even moderately heroic efforts to cure a condition that has little if any ill effect on the individual and not much direct public health importance. The chronic human carrier is the principal reservoir of enteric fever salmonellae, but even in the developing world direct person-to-person spread by asymptomatic carriers is uncommon. ‘Typhoid Mary’, who is reputed to have caused many infections by her cooking, was an exception and must have had peculiar personal habits. Normal personal hygiene, adequate sanitation and a reliable supply of potable water are the real safeguards against enteric fever. Prolonged carriage of other S. enterica serotypes is of even less public health importance, and rarely if ever justifies exclusion from any employment, or intrusive efforts to eradicate the infection.
Typhoid carriers may develop high levels of serum IgG-class antibodies to the Vi capsular polysaccharides; detection of such antibodies warrants faecal screening for S. Typhi and Paratyphi.
Epidemiology and control
The typhoid and paratyphoid bacilli are essentially human parasites. Human beings are the reservoir host and most infections can be traced to a human source, or at least to a source of human sewage. All other salmonellae have animal hosts.
Enteric fever
Incidence
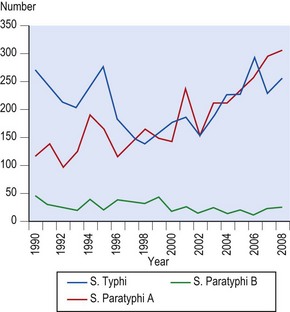
Historically, strains of S. Typhi have been the major cause of typhoidal illness; however, in certain parts of the world, such as Asia, the predominant cause of enteric fever is S. Paratyphi A. In 2000, an estimated 21.7 million cases of typhoid fever occurred with 217 000 deaths. In parts of sub-Saharan Africa, strains of S. Enteritidis and S. Typhimurium are major causes of bloodstream infections. In England and Wales, S. Typhi and S. Paratyphi account for most systemic infections (Fig. 24.3).
Sanitation
The control of enteric fever is in theory straightforward. Cases occur from the ingestion of food or water contaminated with human sewage carrying typhoid or paratyphoid bacilli. Outbreaks occur when numbers of people are infected from a primary source. This may be a single meal or a briefly contaminated water supply, or may be a water supply or source of food contaminated and available for ingestion over a longer period. Secondary transmission from patients infected by the primary source is rare.
Enteric fever is a public health problem only where the wide public availability of wholesome drinking water and the provision of adequate means for the proper disposal of human excreta do not exist. In these circumstances the organisms can be spread widely from carriers and from convalescent and sick persons, possibly helped by cultural factors such as food habits, occupation and personal behaviour. In such communities the selection of control measures may be difficult, and provision of adequate sanitation and pure water may need to be supplemented by prophylactic immunization.
Vaccination
Heat-killed, phenol-preserved whole-cell vaccines containing a mixture of cultures of Typhi, Paratyphi A and Paratyphi B (TAB) have been used for many years in countries with a high endemic level of typhoid fever. Such preparations confer considerable, although not absolute, protection against typhoid for about 3 years. There has always been doubt about the value of the paratyphoid components and monovalent typhoid vaccines are now preferred. Capsular (Vi) polysaccharide vaccines have largely replaced whole-cell vaccines. Alternatively, an oral live-attenuated typhoid vaccine is used.
Travellers to endemic areas in which there are high carriage rates and poor standards of hygiene should be offered immunization, especially if they intend to visit rural areas or to live ‘rough’. However, the risk to the air traveller with full board at a reputable hotel is so small that typhoid immunization may be unnecessary.
Salmonella food poisoning
No other zoonosis is as complex in its epidemiology and control as salmonellosis. The biology of different S. enterica serotypes varies widely and epidemiological patterns differ greatly between geographical areas, depending on climate, population density, land use, farming practices, food-processing technologies and consumer habits.
Sources
The carcasses or products (cooked meats, eggs, milk) of naturally infected domestic animals are the most common sources of food poisoning. Flesh may be infected when an ill, septicaemic animal is slaughtered, but in most cases the salmonellae important in human infection cause only mild or inapparent infection in their animal hosts, and abattoir and shop cross-contamination with intestinal contents from a carrier animal is a more important hazard. Poultry (particularly hens), ducks and turkeys are the most significant reservoirs of food poisoning salmonellae in the UK. Pigs share the honours with poultry in much of northern Europe, whereas beef cattle are important sources in the USA. Duck eggs have always been a problem, as they can be infected in the oviduct before the egg is shelled. Hen eggs acquire their shells higher in the oviduct and are less commonly infected in this way, although they can be contaminated on the outside if laid on soil contaminated by infected hen faeces.
Food contamination
Rats and mice are commonly infected with food-poisoning salmonellae and may contaminate human food with their faeces. Food poisoning may occasionally be caused by food contaminated by a human case or carrier. Thus, even if the foodstuff is initially free from salmonellae, the chance of contamination ‘from the hoof to the home’ is high, and the more sophisticated the manipulation of the food, the greater the chance of contamination. For example, one egg containing salmonellae, if eaten by an individual, probably will not give rise to an infection. On the other hand, if such an egg is pooled with others free from salmonellae, as in preparing a mayonnaise or a Hollandaise sauce for communal consumption, and if conditions of temperature and time allow multiplication of the salmonellae, there will be the potential for an outbreak of infection among those who eat the contaminated food.
A clean carcass can be contaminated at the abattoir by instruments or by hanging in contact with an infected carcass in the chilling hall or during transportation to the wholesale and retail butchers’ premises. The aggregation of calves or pigs in holding pens greatly increases the occurrence of cross-infection before slaughter. Infection in pigs is most often due to the feeding of swill containing infected animal matter, a practice that is difficult to prevent.
There is a close correspondence between the types of salmonellae prevalent in animals, especially pigs and poultry, and the types causing human infection. Strains of S. Typhimurium, which is a primary pathogen in a wide variety of animals, are common, and S. Enteritidis has become prevalent in the UK in poultry flocks. Strains belonging to serotypes such as Heidelberg, Brandenburg, Panama and Virchow are rarely found in animals and yet may be responsible for widespread human infections. More needs to be learned about the epidemiology of such infections. Since 1998 there has been a steady decline in the numbers of salmonellas causing intestinal infection in England and Wales, largely due to vaccination of broiler flocks and restrictions of the importation of eggs from abroad.
Food preparation
Infection of food with salmonellae is not in itself sufficient to cause food poisoning. It is necessary for the infected food to be moist and to be held long enough under conditions that will allow the bacteria to grow, for instance overnight in a warm kitchen or several days in a cool larder. If the food is then eaten without further cooking, infection may follow. Although cooking of liquid foods will render them safe, cooking of solid foods often fails to do so because of the relatively poor rate of heat penetration into the food. A cold or chilled joint of meat, a poultry carcass or a large meat pie may be heated in an oven until the surface is well cooked, while the central part is still insufficiently heated to destroy vegetative bacteria.
Outbreaks
Food-poisoning incidents occur most dramatically as explosive outbreaks among members of a community sharing communal meals, as in factories, hospitals or schools or at a celebratory feast, although sporadic incidents affecting a single family or a single person are much more common and much more difficult to track to a source. In the UK, salmonellae account for about 75% of the incidents of food poisoning in which a causal agent is identified. However, this may only reflect the fact that salmonellosis is relatively easy to confirm bacteriologically and is therefore more readily recognized.
Surveillance
Efficient surveillance of diseases caused by Salmonella has been facilitated by accurate strain discrimination achieved with serotyping, phage typing, antimicrobial resistance typing and pulsed-field gel electrophoresis. Several online networks have been established to enable the rapid exchange of information relating to strains, outbreaks of disease, etc.
Prevention
The principles of the prevention of salmonella food poisoning are:
• raising of animals free from infection
• elimination of contamination by rodents at all levels of food production
• prevention of contamination by human handlers at the wholesale, retail and hotel levels.
The barriers of economic husbandry, out-of-date premises and, perhaps the most important of all, the need for continuing education of food handlers at all levels of production make implementation of these principles difficult. To reduce the incidence of food poisoning, whether due to salmonellae or to other bacteria, two basic precepts must be observed:
1. Raw foodstuffs of animal origin, which are always potentially contaminated, must never have direct or indirect contact with cooked foods.
2. Foodstuff thought to be contaminated should be treated or held under temperature conditions that prevent the organisms from growing.
Cooked foods should be served and eaten immediately after cooking and while still hot, or cooled rapidly and held at refrigerator temperature until eaten, so that at least the inoculum eaten by any individual is small. It is often found that the food incriminated in an outbreak had been cooked several hours or even a day or two before and then left at room temperature before being reheated immediately before serving. This procedure ensures that salmonellae that survived the initial cooking or gained access to the food from contaminated kitchen surfaces or implements had excellent opportunities to multiply in the interval before being warmed up for consumption.
As in most other endeavours to control the spread of infection, the human element is the weakest link in the chain, so that health education, particularly of food handlers, is a most important and continuing requirement.
Chart H, Cheasty T, de Pinna E, et al. Serodiagnosis of Salmonella enterica serovar Typhi and S. enterica serovars Paratyphi A, B and C human infections. Journal of Medical Microbiology. 2007;56:1161–1166.
Crawford RW, Rosales-Reyes R, Ramírez-Aguilar Mde L, et al. Gallstones play a significant role in Salmonella spp. gallbladder colonization and carriage. Proceedings of the National Academy of Sciences USA. 2010;107:4353–4358.
Crump JA, Mintz ED. Global trends in typhoid and paratyphoid Fever. Clinical Infectious Diseases. 2010;50:241–246.
House D, Bishop A, Parry C, et al. Typhoid fever: pathogenesis and disease. Current Opinion in Infectious Diseases. 2001;14:573–578.
Morpeth SC, Ramadhani HO, Crump JA. Invasive non-Typhi Salmonella disease in Africa. Clinical Infectious Diseases. 2009;49:606–611.
Raffatellu M, Wilson RP, Winter SE, Bäumler AJ. Clinical pathogenesis of typhoid fever. Journal of Infection in Developing Countries. 2008;30:260–266.
Threlfall EJ. Antimicrobial drug resistance in Salmonella: problems and perspectives in food- and water-borne infections. FEMS Microbiology Reviews. 2002;26:141–148.
Wain J, House D, Parkhill J, et al. Unlocking the genome of the human typhoid bacillus. Lancet Infectious Diseases. 2002;2:163–170.
Zhang S, Kingsley RA, Santos RL, et al. Molecular pathogenesis of Salmonella enterica serotype typhimurium-induced diarrhea. Infection and Immunity. 2003;71:1–12.
Centers for Disease Control and Prevention. Salmonella Infection (Salmonellosis). http://www.cdc.gov/ncidod/diseases/submenus/sub_salmonella.htm.
Enter-Net. http://www.hpa.org.uk/AboutTheHPA/WhatTheHealthProtectionAgencyDoes/InternationalWork/EnterNet/.
Health Protection Agency. Salmonella. http://www.hpa.org.uk/infections/topics_az/salmonella/menu.htm.
PulseNet. http://www.pulsenetinternational.org.
PulseNet Europe. http://www.pulsenetinternational.org/networks/Pages/europe.aspx.
Sanger Institute. http://www.sanger.ac.uk/resources/downloads/bacteria/salmonella.html.