26 Escherichia
Urinary tract infection; travellers’ diarrhoea; haemorrhagic colitis; haemolytic uraemic syndrome
Key points
• E. coli forms a consistent component of the normal intestinal microbiota.
• Different strains of E. coli carry a range of chromosomal and/or episomal genes encoding pathogenic mechanisms that enable them to cause a diverse range of infections.
• E. coli is the most common cause of urinary tract infection.
• Enterotoxigenic E. coli causes a cholera-like illness.
• Verocytotoxin-producing E. coli belonging to serogroup O157 cause major outbreaks of disease and are a major cause of kidney failure in young children.
• Enteropathogenic E. coli cause infantile enteritis.
• Infection with entero-invasive E. coli resembles that caused by Sh. dysenteriae type 1.
• Enteroaggregative E. coli cause chronic diarrhoeal illness.
• Antibiotic treatment is appropriate in urinary infection and serious sepsis, but most enteric infections are managed conservatively.
Strains of Escherichia coli and related Gram-negative bacteria predominate among the aerobic commensal flora in the gut of human beings and animals. These bacteria are present wherever there is faecal contamination, a phenomenon that is exploited by public health microbiologists as an indicator of faecal pollution of water sources, drinking water and food. The species encompasses a variety of strains, which may be purely commensal or possess combinations of pathogenic mechanisms that enable them to cause disease in man and other animals.
Description
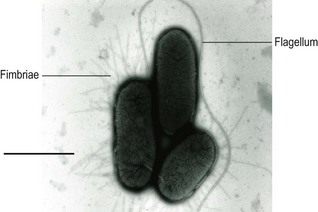
Strains of E. coli are usually motile and may express fimbriae (Fig. 26.1). Some, especially those from extra-intestinal infections, may produce a polysaccharide capsule. They grow well on non-selective media and usually (with the exception of certain Verocytotoxin-producing strains) ferment lactose, producing large red colonies on MacConkey agar. They grow over a wide range of temperature (15–45°C); some strains are more heat-resistant than other members of the Enterobacteriaceae and may survive 60°C for 15 min or 55°C for 60 min. Certain strains are haemolytic when grown on media containing suitable erythrocytes.
E. coli can be differentiated from other enteric Gram-negative bacteria by the ability to utilize certain sugars and by a range of other biochemical reactions. Many characteristic reactions, such as indole production and the formation of acid and gas from lactose and other carbohydrates, take place at 44°C as well as at 37°C.
DNA–DNA recombination studies show that E. coli and Shigella spp. form a single genetic group, and it is therefore to be expected that intermediate strains will occur. Certain atypical types of E. coli are non-motile and anaerogenic, and often ferment lactose late or not at all.
Antigenic structure
Serotyping is based on the somatic lipopolysaccharide (LPS) (O), flagellar (H) and capsular (K) antigens, as detected in agglutination assays with specific rabbit antibodies. More than 200 different O-antigens have been described and others continue to emerge. Serotyping detects cross-reactions as a result of shared epitopes on the LPS expressed by strains of E. coli, and may occur with organisms belonging to the genera Brucella, Citrobacter, Providencia, Salmonella, Shigella and Yersinia.
More than 50 H antigens have been identified. Most are monophasic, but rare diphasic strains have been reported. There are only a few significant cross-reactions between them and with the H antigens of other members of the Enterobacteriaceae. Because certain strains of E. coli cease to express flagella during growth in vitro, strains may need to be grown in semi-solid agar (Craigie tubes) to induce flagella expression. Obtaining a motile phenotype can take several days and sequence-typing of the fliC gene has been considered as a rapid alternative to serotyping.
The term ‘K antigen’ was formerly used collectively for surface or capsular antigens (designated L, A and B) that prevent flagella-specific antibodies from binding to the somatic antigens. In modern usage, ‘K antigen’ refers to the acidic polysaccharide capsular antigens, and those of E. coli may be divided into two groups (groups I and II; Table 26.1) that largely correspond to the former A and L antigens. Due to the complexity of available tests, strains of E. coli are not routinely K-typed.
Table 26.1 K antigens of E. Coli
| Property | Group I | Group II |
|---|---|---|
| Molecular weight (Da) | >100 000 | <50 000 |
| Acidic component | Hexuronic acid, pyruvate | Glucuronic acid, phosphate, KDO, NeuNAc |
| Heat stability (100°C, pH 6) | All stable | Mostly labile |
| O groups | O8, O9 | Many |
| Chromosome site | His | SerA |
| Expressed at 17–20°C | Yes | No |
| Electrophoretic mobility | Low | High |
KDO, ketodeoxyoctonate; NeuNAc, N-acetylneuraminic acid.
Fimbrial antigens
Strains of E. coli may express both sex pili and more than one type of fimbrial structure (see p. 159). Within a given culture, there may exist individual cells with fimbriae and others with none, and there is reversible variation between the fimbriate and the non-fimbriate phase.
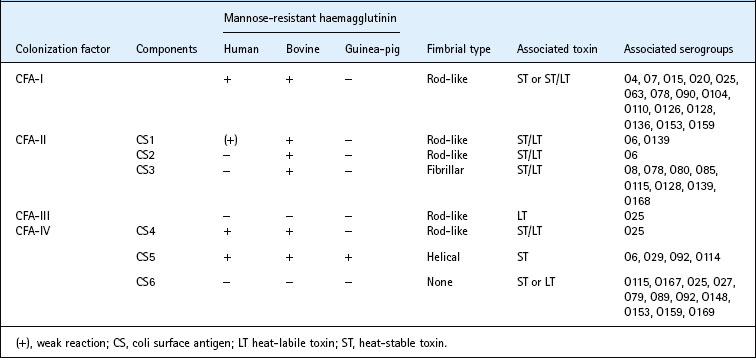
Type-1 fimbriae can mediate adhesion to a wide range of human and animal cells that contain the sugar mannose. Such adhesion might be involved in pathogenicity and there are some examples of this. Filamentous protein structures resembling fimbriae cause mannose-resistant haemagglutination, and probably play an important part in the pathogenesis of diarrhoeal disease and urinary tract infection. They include the K88 antigen found in strains causing enteritis of pigs, the K99 antigen found in strains causing enteritis of calves and lambs, and the colonization factor antigens (CFAs) or coli surface (CS) antigens expressed by enterotoxigenic E. coli (ETEC) that cause human diarrhoeal disease.
Fimbriae that are of importance in urinary tract infection and cause mannose-resistant haemagglutination are distinguished according to their receptor specificities. These include the P fimbriae that bind specifically to receptors present on the P blood group antigens of human erythrocytes and uroepithelial cells.
Pathogenesis
Strains of E. coli possess a range of different pathogenic mechanisms. The polysaccharides of the O and K antigens protect the organism from the bactericidal effect of complement and phagocytes in the absence of specific antibodies. However, in the presence of antibody to K antigens alone, or to both O and K antigens, opsonization may occur.
Many strains express haemolysin(s), and in general strains of E. coli isolated from human extra-intestinal infections are more likely to be haemolytic than strains isolated from the faeces of healthy human beings. Haemolysin production is an important pathogenic mechanism for releasing essential ferric ions bound to haemoglobin, and the expression of certain haemolysins has been shown to be regulated by iron.
Strains of E. coli can express siderophores, such as enterobactin, which remove ferric ions from mammalian iron transport proteins like transferrin and lactoferrin (see Ch. 13). Some strains also express the siderophore, aerobactin; this may be plasmid-mediated. The ability of strains of E. coli to acquire ferric ions is a recognized pathogenic mechanism. Expression of the aerobactin-mediated iron-uptake system is a common feature of strains isolated from patients with septicaemia, pyelonephritis and lower urinary tract infection. Some strains may also utilize siderophores produced by certain species of fungi (e.g. ferrichrome, coprogen, rhodoturulic acid) to acquire iron from environmental sources.
Clinical syndromes
Urinary tract and septic infections
E. coli is the most common cause of acute, uncomplicated urinary tract infection outside hospitals, as well as causing hospital-associated urinary tract sepsis. These bacteria may also cause neonatal meningitis and septicaemia, sepsis in operation wounds and abscesses in a variety of organs.
As many as 80% of E. coli strains that cause neonatal meningitis and 40% of those isolated from infants with septicaemia but without meningitis express a K1 antigen. Strains possessing the K1 or the K5 antigen may be more virulent than those with other K antigens, as they share structural identity with host components.
Strains that cause urinary tract infection often originate from the gut of the patient, with infection occurring in an ascending manner. The ability of E. coli to infect the urinary tract may be associated with fimbriae that specifically mediate adherence to uroepithelial cells.
Epidemiology
Urinary tract infection occurs more frequently in women than in men because the shorter, wider, female urethra appears to be less effective in preventing access of the bacteria to the bladder. Sexual intercourse may be a predisposing factor. The high incidence in pregnant women can be attributed to impairment of urine flow due partly to hormonal changes and partly to pressure on the urinary tract. Other causes of urinary stagnation that may predispose to urinary tract infection include urethral obstruction, urinary stones, congenital malformations and neurological disorders, all of which occur in both sexes. In men, prostatic enlargement is the most common predisposing factor. Catheterization and cystoscopy may introduce bacteria into the bladder and therefore carry a risk of infection.
Most urinary tract infections are thought to be caused by organisms originating from the patient’s own faecal flora. However, the prevalence of various serotypes of E. coli in urinary tract infections varies with geographical location, suggesting that E. coli causing such infections are specific pathogens for the urinary tract. Pathogenic strains, possibly transmitted in contaminated foods, are able to colonize the bowel, and in individuals with predisposing factors may cause a urinary tract infection. The prevalence of infections due to a particular strain may therefore increase for a time in a locality.
Laboratory diagnosis
Clinical specimens may be stained by Gram’s method for microscopical examination, and are cultured on MacConkey agar or other suitable media. In the case of suspected urinary tract infection, culture is semi-quantitative; in acute E. coli infections the organism is generally present in pure culture at a count of 105 or more per millilitre of urine. For more serious infections, strains should be referred to a reference laboratory for serotyping.
Treatment and control
In the absence of acquired resistance, E. coli is susceptible to many antibacterial agents, including ampicillin, cephalosporins, tetracyclines, quinolones, aminoglycosides, trimethoprim and sulphonamides. Many strains, however, have acquired plasmids conferring resistance to one or more of these drugs, and antimicrobial therapy should be guided by laboratory tests of sensitivity if possible.
Uncomplicated cystitis usually responds to minimal treatment with oral agents such as trimethoprim or nitrofurantoin, but more serious infections require specific antimicrobial therapy based on laboratory results. In particular, bacterial meningitis is a medical emergency and vigorous early treatment with cefotaxime and gentamicin is required.
Urinary catheterization and cystoscopy require rigorous aseptic technique to minimize the introduction of bacteria into the bladder. Bladder irrigation and systemic treatment with antimicrobial agents has been used in catheter-associated infections, but such treatment is seldom more than palliative and encourages infections with resistant organisms.
Diarrhoea
Although E. coli is normally carried in the gut as a harmless commensal, it may cause gastrointestinal disease ranging in severity from mild, self-limiting diarrhoea to haemorrhagic colitis and the associated, potentially life-threatening, haemolytic uraemic syndrome. Such strains fall into at least five groups, each associated with specific serotypes (Table 26.2) and with different pathogenic mechanisms:
1. Enteropathogenic E. coli (EPEC), which cause infantile enteritis, especially in tropical countries.
2. Enterotoxigenic E. coli (ETEC), which are responsible for community-acquired diarrhoeal disease in areas of poor sanitation and are the most common cause of travellers’ diarrhoea.
3. Enteroinvasive E. coli (EIEC), which cause an illness resembling shigella dysentery in patients of all ages.
4. Verocytotoxin-producing E. coli (VTEC), which cause symptoms ranging from mild, watery diarrhoea to haemorrhagic colitis and haemolytic uraemic syndrome.
5. Enteroaggregative E. coli (EAggEC), which cause chronic diarrhoeal disease in certain developing countries.
Table 26.2 The major groups of diarrhoea-causing E. coli
| Pathogenic group | Common serogroups |
|---|---|
| Enteropathogenic E. coli (EPEC) | O26, O55, O86, O111, O114, O119, O125,O126, O127, O128, O142, O158 |
| Enterotoxigenic E. coli (ETEC) | O6, O8, O15, O25, O27, O63, O119, O125, O126, O127, O128, O142 |
| Enteroinvasive E. coli (EIEC) | O78, O115, O148, O153, O159, O167 |
| Verocytotoxin-producing E. coli (VTEC) | O26, O111, O112ac, O124, O136, O143, O144, O152, O157a, O164 |
| Enteroaggregative E. coli (EAggEC) | More than 50 ‘O’ serogroups |
a Other serogroups are far less common than 0157 in human disease.
Enteropathogenic E. coli (EPEC)
Pathogenesis
EPEC strains belonging to characteristic serogroups (Table 26.2) were originally identified epidemiologically as a cause of diarrhoeal disease in infants. Some such strains, notably serogroup O26, have acquired the genes for expression of Verocytotoxin and are classified as VTEC (see below). EPEC may adhere to HEp-2 tissue culture cells in small discrete ‘localized adhesion’ clusters, a phenotype encoded by the adherence factor plasmid (pEAF); strains which carry the pEAF are termed ‘typical’ EPEC (tEPEC) while those without this plasmid are termed ‘atypical’ EPEC (aEPEC).
Colonization of the upper part of the small intestine occurs in infantile enteritis associated with EPEC. Electron microscopy of intestinal biopsy specimens shows that the bacteria become intimately associated with the mucosal surface and are partially surrounded by cup-like projections (‘pedestals’) of the enterocyte surface. In areas of EPEC attachment the brush border microvilli are lost. Adhesion to the gut wall and the subsequent mucosal damage has been termed an ‘attaching and effacing’ lesion. The genes responsible are located on a pathogenicity island located on the E. coli chromosome. EPEC use a novel mechanism of adhesion in which the receptor for the adhesin is synthesized by the bacteria and inserted in the host gut wall to provide a binding site. One of the proteins involved, intimin, is expressed on the bacterial cell surface as an adhesin; the other key protein, the translocated intimin receptor (Tir) is synthesised by the EPEC and inserted into the host cell membrane to allow intimin attachment. EPEC-associated toxins have not been detected.
Laboratory diagnosis
Stool specimens are plated on media such as MacConkey agar. Bacteria fermenting lactose are identified as E. coli and serotyped based on somatic and flagellar antigens. Strains belonging to EPEC-associated serogroups may be putative EPEC but detecting the eae genes by PCR can provide an accurate identification.
Epidemiology
Since 1971 few epidemics of EPEC enteritis have been reported in the UK or the USA, although a satisfactory explanation for the decline has not been put forward. Strains responsible for sporadic cases that continue to occur in the UK, especially in the summer months, possess the same pathogenic mechanisms as those that caused earlier outbreaks.
EPEC enteritis is common in communities with poor hygiene where sporadic cases and frequent outbreaks occur in the general community as well as in institutions. The importance of EPEC as a cause of enteritis in adults is difficult to evaluate because few laboratories perform the relevant tests.
Enterotoxigenic E. coli (ETEC)
Pathogenesis
ETEC produce a heat-stable enterotoxin or a heat-labile enterotoxin, or both. In addition, they usually express fimbriae that are specific for the host animal species and that enable the organisms to adhere to the epithelium of the small intestine. Infection is usually of brief duration, often beginning with the rapid onset of loose stools and accompanied by variable symptoms, including nausea, vomiting and abdominal cramps.
Heat-labile enterotoxin (LT) is closely related to the toxin produced by strains of Vibrio cholerae. There are two main forms, termed LT-I and LT-II (Table 26.3). Different forms of LT-I associated with human, porcine and chicken infection have been described; similarly, two forms of LT-II (LT-IIa and LT-IIb) have been detected. Although these toxins have a degree of structural variation, they are all subunit protein toxins comprising one A subunit and five B subunits with molecular weights of 26 000–28 000 Da and 11 500–11 800 Da, respectively. The mechanism by which diarrhoea is caused is identical to that of cholera toxin (see p. 315).
Table 26.3 Differential properties of heat-labile toxins (LT) of E. coli
| LT-I | LT-II | |
|---|---|---|
| Tissue culture changesa | ± | ± |
| Molecular weight (kDa) | ||
| A subunit | 26 | 28 |
| B subunit | 11.5 | 11.8 |
| Isoelectric point | 8.5 | 6.8b, 5.4c |
| Genetic location | Plasmid | Chromosome |
| Action | Binds to gangliosides | Activates cAMP |
cAMP, cyclic adenosine monophosphate.
a Chinese hamster ovary, Y1 cells or Vero cells.
In contrast to LTs, the heat-stable enterotoxins (STs) of E. coli (Table 26.4) have a low molecular weight which confers heat stability and poor antigenicity. There are two major classes, designated ST-I (STa) and ST-II (STb). Variants of ST-I have been associated with porcine and human infections. ST-I is detected by immunoassay or by molecular genetic methods. This toxin activates guanylate cyclase activity, resulting in an increase in the level of cyclic guanosine monophosphate (cGMP). The activity of ST-I is rapid, whereas LTs act after a lag period. The mechanism of secretion caused by ST-I, via cGMP, is not fully understood but calcium appears to play a role. ST-I is plasmid encoded, and these plasmids may also encode the genes for LT, adhesive factors and antibiotic resistance.
Table 26.4 Differential properties of heat-stable toxins (ST) of E. coli
| ST–I (STa) | ST–II (STb) | |
|---|---|---|
| Molecular weight (kDa) | 2 | 5 |
| Infant mouse test | + | − |
| Methanol | Soluble | Insoluble |
| Pig intestinal loop | + | + |
| Rabbit ileal loop | + | − |
| Rat gut loop | + | − |
| Action | Activates cGMP | Unknown (cyclic nucleotides) |
cGMP, cyclic guanosine monophosphate.
ST-II is distinguished from ST-I by its biological activity and its insolubility in methanol. The mechanism of action is not known but it appears not to act via cyclic adenosine monophosphate (cAMP) or cGMP. Molecular methods have superseded animal models for detecting these organisms.
Enterotoxin alone is not sufficient to enable E. coli to cause diarrhoea. The organism must first bind to specific receptors on the mucosal surface of the epithelial cells of the small intestine This adhesion is usually mediated by fimbriae expressing colonization factor antigens (CFAs) or coli surface (CS) antigens. Plasmids that simultaneously carry genes for both CFAs and enterotoxin production have been described.
The first colonization factor to be recognized in E. coli was a fimbrial antigen, K88, controlled by a transferable plasmid. Several more have subsequently been found in human strains of ETEC and, no doubt, others remain to be discovered. The properties of some important colonization factors are shown in Table 26.5.
Laboratory diagnosis
Tissue culture assays formerly used for detecting LT have been replaced by immunological techniques. Most laboratories now screen for LT-producing strains of E. coli by a rapid PCR test. Other tests include an enzyme-linked immunoabsorbent assay (ELISA) in which LT present in culture supernates is captured with gangliogide GM1 and detected with toxin-specific rabbit antibodies; a precipitin test (the Biken test) on bacterial colonies growing on an agar medium containing rabbit antibodies specific for LT and commercial latex particle agglutination tests.
ST toxin is usually detected by ELISA tests with specific monoclonal antibody.
Epidemiology
In developing countries ETEC is a major cause of death in children under the age of 5 years. These strains also commonly cause diarrhoea in travellers visiting countries where ETEC are endemic.
The sources and modes of spread of ETEC infection in countries with a warm climate are not well understood, but it seems likely that water contaminated by human or animal sewage plays an important part in the spread of infection.
Enteroinvasive E. coli (EIEC)
Pathogenesis
EIEC, like Shigella dysenteriae-1 (see Ch. 25), cause disease by invading intestinal epithelium. Infection is by ingestion; only a small number of bacteria need to be swallowed as they are relatively resistant to gastric acid and bile, and pass readily into the large intestine where they multiply in the gut lumen. The bacteria pass through the overlying mucous layer, attach to the intestinal epithelial cells and are carried into the cell by endocytosis into an endocytic vacuole, which then lyses. The ability to cause the vacuole to lyse is an important virulence attribute, as organisms unable to do this cannot spread to neighbouring cells. After lysis of the vacuole the bacteria multiply within the epithelial cell and kill it. Spread to neighbouring cells leads to tissue destruction and consequent inflammation, which is the underlying cause of the symptoms of bacillary dysentery.
Pathogenicity depends on both chromosomal and plasmid genes. A large plasmid carries genes for the expression of outer membrane proteins that are required for invasion as well as genes necessary for the insertion of these proteins into the cell membrane. Plasmid genes are also required for the ability to escape from the endocytic vacuole and to invade contiguous host cells. Chromosomal genes encoding pathogenic mechanisms include those required for the expression of long-chain LPS and those encoding an aerobactin-mediated iron-sequestering system.
Laboratory diagnosis
The controversial Serény test, in which the bacteria are tested for the ability to cause conjunctivitis in guinea-pigs, and former tissue culture methods have been superseded by molecular techniques to detect the genes encoding the invasion plasmid antigen (ipaH) and aerobactin expression (iuc).
Epidemiology
The epidemiology and ecology of EIEC have been poorly studied, but there appears to be no evidence of an animal or environmental reservoir. Surveys suggest that they cause about 5% of all diarrhoeas in areas of poor hygiene. In the UK and USA, outbreaks are occasionally described, especially in schools and hospitals for the mentally handicapped. Infections are usually food-borne but there is also evidence of cross-infection. The most common serogroup is O124.
Verocytotoxigenic E. coli (VTEC)
Strains of E. coli expressing a protein cytotoxic for Vero cells were discovered in 1977. Once epidemiologists were aware of VTEC, the importance of these bacteria in human disease became apparent and a link was established with two diseases of previously unknown aetiology: haemorrhagic colitis and haemolytic uraemic syndrome. Outbreaks were first recognized in the USA in 1982, and strains of VTEC belonging to serogroup O157 emerged as the major cause. Since then, outbreaks and sporadic cases have been reported in several other countries and VTEC belonging to many other serotypes have been described. Well publicized major episodes in the USA, Canada, Japan and Scotland have heightened general awareness of the importance of this disease.
Pathogenesis
Human VTEC infection is associated with a range of clinical symptoms from mild, non-bloody diarrhoea to the severe manifestations of haemolytic uraemic syndrome; a wide spectrum of illness can occur even within a single outbreak.
Haemorrhagic colitis is a grossly bloody diarrhoea, usually in the absence of pyrexia. It is usually preceded by abdominal pain and watery diarrhoea. Haemolytic uraemic syndrome is characterized by acute renal failure, micro-angiopathic haemolytic anaemia and thrombocytopenia. It occurs in all age groups, but is more common in infants and young children, and is a major cause of renal failure in childhood. The syndrome is usually associated with a prodromal bloody diarrhoea, but an ‘atypical’ form occurs without a diarrhoeal phase. It may be accompanied by thrombotic thrombocytopenic purpura in which the clinical features are further complicated by neurological involvement and fever.
VTEC have also been implicated as a cause of disease in animals, particularly calves and pigs.
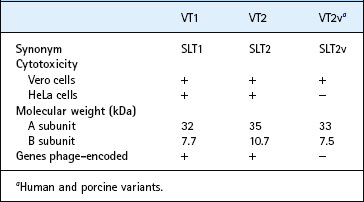
The biological properties, physical characteristics and antigenicity of Verocytotoxin (VT) are very similar to those of Shiga toxin, produced by strains of Sh. dysenteriae type 1 (see p. 276), but the genes encoding VT in E. coli are carried on a lambda-like bacteriophage whereas those encoding Shiga toxin in Sh. dysenteriae type 1 are located on the chromosome. There are two antigenically distinct forms: VT1 and VT2. Antibodies prepared to VT1 neutralize Shiga toxin, whereas antibodies specific for VT2 do not. Variant forms of VT2 have been described in strains of human and porcine origin. The genes controlling production of these variant toxins are not phage encoded, and the toxin receptor also differs from that used by VT1 and VT2 (Table 26.6).
Like Shiga toxin, VT1 and VT2 comprise A and B subunits. For both toxins the A subunit possesses the biological activities of the toxin and the B subunits mediate specific binding and receptor-mediated uptake of the toxin. VT1 and VT2 bind to globotriosylceramide (Gb3) molecules present on the surface of certain eukaryotic cells. In contrast VT2 variant toxins bind to globotetraosylceramide (Gb4). During infection with VTEC the inflammatory mediators, tumour necrosis factor and interleukin-1, in combination with LPS, increase the number of ceramide receptors on the surface of eukaryotic cells, enhancing the binding of VT to these cells. Infection also results in expression of VT molecules that bind to Gb3 receptors located in the kidneys, leading to haemolytic uraemic syndrome. Molecules of Gb3 have been detected on mammalian erythrocytes that have Pk antigens. About 75% of the human population carries this antigen, and such people may have protection from developing haemolytic uraemic syndrome.
VT1 and VT2, like Shiga toxin, are cytotoxic for Vero and HeLa cells, although certain VT2 variant toxins do not bind to HeLa cells, which do not express Gb3 receptors.
VT causes a direct, dose-dependent, cytotoxic effect on human umbilical cord endothelial cells in culture; actively dividing cells are the most sensitive. Micro-angiopathy of the capillaries is a characteristic renal lesion in haemolytic uraemic syndrome, supporting the hypothesis that vascular endothelial cells are primary targets for VT.
Once bound to the eukaryotic cell surface, the holotoxin becomes internalized by host cells and remains active within endosomes. The toxin eventually reaches the Golgi apparatus by mechanisms as yet unknown. At some point within the host cell, the A subunit becomes enzymically ‘nicked’ to form portions A1 (28 kDa) and A2 (4 kDa); the A1 portion of the toxin prevents protein synthesis and results in cell death.
Strains of O157 VTEC express an ‘attaching and effacing’ phenotype and, in common with strains of EPEC, the genes involved are located on a pathogenicity island located on the E. coli chromosome (see p. 283). Although the mechanisms of bacterial adhesion expressed by O157 VTEC are similar to those of EPEC, antigenic variation in, for example, the respective intimin and Tir proteins, has been detected. Owing to the similarity in structure between Verocytotoxin and Shiga-toxin expressed by Shigella dysenteriae-1, VTEC have been termed Shiga-toxin-producing E. coli or STEC; however this can be misleading since VT2 is quite distinct from VT1 and Shiga toxin. Similarly, strains of VTEC have been termed ‘enterohaemorrhagic’ owing to the symptoms of disease they cause, but since few strains of VTEC cause this form of disease the term must be used with caution.
Laboratory diagnosis
The proportion of VTEC in the faecal flora may be low, often less than 1%, so that testing of individual colonies from culture plates may not always detect the pathogen. In foods or faecal extracts VTEC can be concentrated with immunomagnetic beads, small magnetic spheres coated with O157-specific rabbit antibodies which ‘capture’ the VTEC O157 and can be harvested with a magnet. The vtx genes encoding VT1 and VT2 can be detected with DNA probes and PCR. Electrophoretic and gene sequence analysis techniques provide strain discrimination and characterization in outbreaks of infection.
Although 95% of E. coli ferment sorbitol, O157 VTEC do so only slowly. This property is exploited by replacing the lactose present in MacConkey agar with sorbitol. Most strains of O157 VTEC produce colourless colonies after overnight incubation and these can be tested with an O157 LPS-specific antiserum in a simple agglutination assay. An O157-specific PCR assay has also been developed but putative strains of E. coli O157 should be confirmed by routine bacteriological examination and serotyping. Toxigenicity is confirmed by gene probes, by PCR, by testing strains for a cytotoxic effect on Vero cells or by a VT-specific ELISA.
Infection with VTEC O157 results in high levels of serum antibodies to the O157 LPS antigens but not to the flagellar proteins. These antibodies may be detected for several months after infection, providing valuable retrospective evidence of exposure or recovery from acute infection. Polyacrylamide gel electrophoresis and immunoblotting remain the most sensitive means of detecting antibodies to E. coli O157 LPS, techniques which can also be used to detect salivary antibodies.
Epidemiology
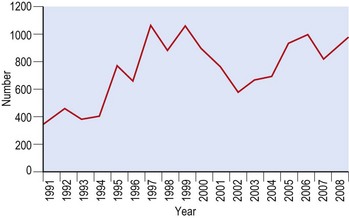
In England and Wales numbers of isolates of VTEC O157 have ranged from 361 cases in 1991 to a peak of 1087 cases in 1997 (Fig. 26.2). Outbreaks have occurred in the community, in nursing homes for the elderly, in day care centres for young children and following visits to petting farms. The most severe clinical manifestations are usually seen in the young and the elderly.
Food is an important source and outbreaks of disease due to VTEC O157 have been associated with hamburger meat, cooked meat products, unpasteurized apple juice, unpasteurized milk and radish sprouts. VTEC have also been isolated from healthy heifers on farms associated with milk-borne incidents, and it is now generally accepted that cattle are a major reservoir.
Enteroaggregative E. coli (EAggEC)
EAggEC are characterized by their ability to adhere to tissue culture cells, such as HEp-2, in an aggregative or ‘stacked brick’ pattern, a property usually encoded on a 60-MDa plasmid. Such strains were first reported in 1987 as a cause of chronic diarrhoea in malnourished children in Chile, and were reported subsequently in many other countries. Occasional outbreaks have occurred in Europe, including the UK, and travellers to endemic regions may become infected. EAggEC may be present in the faeces of apparently healthy members of the population, but EAggEC diarrhoea is comparatively rare in industrialized countries.
Pathogenesis
How EAggEC causes diarrhoea is poorly understood. Volunteer studies have failed to identify the infective dose, and the site of adhesion within the human host is not known. The characteristic pattern of adhesion to HEp-2 cells may be a putative pathogenic mechanism. Strains of EAggEC may express fimbriae, but adhesion to HEp-2 cells can also occur in the absence of these structures and for certain strains adhesion involves cell surface charge. Some, but not all, strains produce an ST-like toxin. Similarly, some isolates express haemolysins, an aerobactin-mediated high-affinity iron uptake system and a range of haemagglutinins; however, apart from the ability to adhere to HEp-2 cells in the stacked-brick formation, these strains are generally quite distinct.
Epidemiology
Strains of EAggEC belong to very diverse combinations of O and H type, and even within an outbreak of diarrhoeal disease strains with several different serotypes may be isolated. The diversity of serotypes and pathogenic mechanisms observed suggests that the genes encoding the aggregative phenotype may be accepted readily by strains of commensal and potentially pathogenic strains of E. coli.
Laboratory diagnosis
The pattern of adhesion to HEp-2 cells on a glass slide has been considered as the ‘gold standard’ for detecting EAggEC; however, since the bacteria also stick to the glass non-specifically, the significance of the adhesion assay has come into question. Aggregative adhesion gene probes and PCRs based on the aat genes, encoding the anti-aggregation protein transporter, have proved useful as rapid screening methods.
Treatment and control of E. coli enteritis
General measures
As with most diarrhoeal disease, the early administration of fluid and electrolytes is the most important single factor in preventing the death of the patient in severe infections. Antimicrobial drugs play a minor role. Despite the potentially serious consequences of VTEC infection, the use of antibiotics in this condition is controversial; certain antimicrobial drugs have been shown to increase expression of Verocytotoxin, so that administration of antibiotics may be counterproductive.
The most effective means of preventing infection is to avoid exposure to the infecting agent. Contaminated food and water are probably the most important vehicles of ETEC infection in developing countries. The provision of safe supplies of water together with education in hygienic practice in the handling and production of food particularly that given to young children, are essential. Travellers to countries with poor hygiene, especially in the tropics, should select eating places with care and, if possible, should consume only hot food and drinks, or bottled water. Self-peeled fruits are probably safe, but salads should be avoided. Unheated milk should always be considered unsafe.
The spread of infantile enteritis in hospitals and nurseries is mainly from patient to patient, generally on the hands of attendants, or from contaminated infant feeds. It can be prevented only by very strict hygiene. Infected patients, and recently admitted patients suspected of being infected, must be isolated by barrier nursing techniques to prevent faecal spread. In some cases outbreaks can be terminated only by closing the ward or nursery and cleaning thoroughly before reopening.
VTEC infections are acquired most frequently from meat, unpasteurized milk and direct contact with animals. Food-borne infections should be avoided by normal food hygiene with particular attention to processing and handling cooked meat products separately from raw meat, and the thorough cooking of raw meats, especially if minced.
Vaccination
The most extensive studies of the use of vaccines have so far been in the veterinary field. Trials of a potential vaccine for human use, prepared by cross-linking a synthetically produced ST with the non-toxic B subunit of LT, were inconclusive.
Inhibition of enterotoxin activity
Activated charcoal, bismuth subsalicylate and non-steroidal anti-inflammatory drugs inhibit or reverse the secretory effects of enterotoxins in experimental animals and may be of value in the prevention or treatment of diarrhoea.
An experimental silica-based compound designed to bind VT synthesized by bacteria in the intestine, preventing toxin from entering the patient’s tissues has been advocated for infection with VTEC. However, as symptoms follow the effects of toxin, such treatment may have only limited value.
Antimicrobial prophylaxis
Several antimicrobial drugs, including doxycycline, trimethoprim and fluoroquinolones reduce the incidence of diarrhoea in travellers to tropical areas. However, the widespread use of antibiotic prophylaxis has been criticized on the grounds of drug toxicity and because of the rick of encouraging the development and spread of drug resistance.
Other Escherichia species
E. blattae was first described among bacteria isolated from the gut of the cockroach. It differs from E. coli in several ways and would probably be better placed in another genus. It has not been reported from human clinical specimens. E. fergusonii, E. hermanii and E. vulneris have been recovered from various clinical specimens, especially faeces and wounds, but their clinical significance is usually unclear.
Chart H. Clinical significance of Verocytotoxin-producing Escherichia coli O157. World Journal of Microbiology and Biochemistry. 2001;16:719–724.
Chart H, Cheasty T. Human infections with Verocytotoxin-producing Escherichia coli O157 – 10 years of E. coli O157 serodiagnosis. Journal of Medical Microbiology. 2008;57:1389–1393.
Croxen MA, Finlay BB. Molecular Mechanisms of Escherichia coli pathogenicity. Nature Reviews Microbiology. 2010;8:26–38.
Deisingh AK, Thompson M. Strategies for the detection of Escherichia coli O157:H7. Journal of Applied Microbiology. 2004;96:419–429.
Gunn GJ, McKendrick IJ, Ternent HE, et al. An investigation of factors associated with the prevalence of verocytotoxin producing Escherichia coli O157 shedding in Scottish beef cattle. The Veterinary Journal. 2007;174:554–564.
Huang DB, Okhuysen PC, Jiang Z-D, DuPont HL. Enteroaggregative Escherichia coli: an emerging enteric pathogen. American Journal of Gastroenterology. 2004;99:383–389.
Jenkins C, Chart H, Willshaw GA, et al. Association of putative pathogenicity genes with adherence characteristics and fimbrial genotypes in typical enteroaggregative Escherichia coli from patients with and without diarrhoea in the United Kingdom. European Journal of Clinical Microbiology and Infectious Disease. 2007;26:901–906.
Jenkins C, Tembo M, Chart H, et al. Detection of enteroaggregative Escherichia coli in faecal samples from patients in the community with diarrhoea. Journal of Medical Microbiology. 2006;55:1493–1497.
Kaper JB, Nataro JP, Mobley HLT. Pathogenic Escherichia coli. Nature Reviews: Microbiology. 2004;2:123–140.
Lynn RM, O’Brien SJ, Taylor CM, et al. Childhood hemolytic uremic syndrome, United Kingdom and Ireland. Emerging Infectious Diseases. 2005;11:590–596.
Matthews L, McKendrick IJ, Ternent H, et al. Super-shedding cattle and the transmission dynamics of Escherichia coli O157. Epidemiology and Infection. 2006;134:131–142.
Sanchez J, Holmgren J. Virulence factors, pathogenesis and vaccine protection in cholera and ETEC diarrhea. Current Opinions in Immunology. 2005;17:1–11.
Centers for Disease Control and Prevention. Escherichia coli infection. http://www.cdc.gov/ecoli/.
Health Protection Agency. Escherichia coli. http://www.hpa.org.uk/Topics/InfectiousDiseases/InfectionsAZ/EscherichiaColi/.
Health Protection Agency. Escherichia coli O157. http://www.hpa.org.uk/Topics/InfectiousDiseases/InfectionsAZ/EscherichiaColiO157/.