30 Vibrio, mobiluncus, gardnerella and spirillum
Cholera; vaginosis; rat bite fever
Key points
• Vibrio cholerae belonging to serogroup O1 is the causative agent of epidemic cholera.
• V. cholerae O139 is emerging as a cause of epidemic cholera.
• Cholera toxin is the key pathogenic mechanism, causing extensive loss of water and electrolytes in the form of rice-water stools; death from cholera can be prevented with rehydration therapy.
• V. parahaemolyticus is a major cause of diarrhoea in Japan and South-East Asia; infection is associated with the consumption of seafood.
• Infection with V. vulnificus may result in rapid-onset and fatal septicaemia, particularly in people with conditions of iron overload, and is associated with the consumption of seafood.
• Mobiluncus spp. and Gardnerella vaginalis are a major cause of bacterial vaginosis; changes in the normal vaginal flora appear to permit these organisms to cause disease.
Vibrio
The Vibrio genus includes more than 30 species commonly found in aquatic environments. Some cause disease in human beings as well as in marine vertebrates and invertebrates. The most important pathogens of man are Vibrio cholerae, V. parahaemolyticus and V. vulnificus, but various other species are occasionally implicated as opportunist pathogens. Historically, vibrios have been associated almost exclusively with epidemic and pandemic cholera caused by a particular antigenic form of V. cholerae.
Description
Vibrios are short Gram-negative rods, which are often curved and actively motile by a single polar flagellum (Fig. 30.1). Nearly all produce the enzyme oxidase and give a positive indole reaction. The genus can be divided into non-halophilic vibrios, including V. cholerae and other species that are able to grow in media without added salt, and halophilic species such as V. parahaemolyticus and V. vulnificus that require salt for growth. Vibrios grow readily on ordinary media provided that their requirements for electrolytes are met, and grow best when abundant oxygen is present. Most grow at 30°C but some of the halophilic species grow poorly at 37°C, whereas V. cholerae, V. parahaemolyticus and V. alginolyticus grow at 42°C.
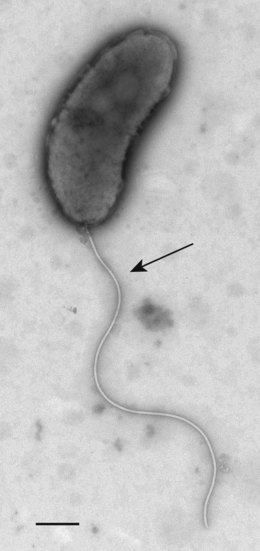
Fig. 30.1 Electron micrograph of Vibrio cholerae showing typical short rod morphology with a single polar flagellum. Bar = 2 µm.
Vibrios have a low tolerance to acid and prefer alkaline conditions (growth range pH 6.8–10.2, optimum pH 7.4–9.6).
Vibrio cholerae
Description
Strains of V. cholerae can be differentiated by their lipopolysaccharide O-antigens into some 130 different serogroups. The classical cause of epidemic cholera possesses the O1 antigen, and is known as V. cholerae O1. Other serogroups are collectively known as ‘non-O1 V. cholerae’ and correspond to strains formerly known as non-agglutinable vibrios or non-cholera vibrios. Some of these strains can cause diarrhoea in man. All strains of V. cholerae share the same flagellar (H) antigen.
Strains of V. cholerae O1 may be further subdivided on the basis of their O antigens into the subtypes Inaba and Ogawa; some strains possess determinants of both of these subtypes and are known as subtype Hikojima.
There are two biotypes of V. cholerae O1: the classical and El Tor biotypes. The El Tor variant is distinguished from the classical biotype by the ability to express a haemolysin on sheep erythrocytes, resistance to polymyxin B, agglutination of chicken erythrocytes and a positive Voges–Proskauer test. The two biotypes can also be recognized by their differential susceptibility to specific phages IV and V.
V. cholerae O139, which has emerged as a new epidemic strain (see below), may have evolved from V. cholerae O1, but with a modified lipopolysaccharide structure.
Pathogenesis
Clinical manifestations
Cholera is characterized by the sudden onset of effortless vomiting and profuse watery diarrhoea. Although vomiting is a common feature, the rapid dehydration and hypovolaemic shock, which may cause death in 12–24 h, are related mainly to the profuse, watery, colourless stools with flecks of mucus and a distinctive fishy odour – rice water stools – which contain little protein and are very different from the mucopurulent blood-stained stools of bacillary dysentery. Anuria develops, muscle cramps occur, and the patient quickly becomes weak and lethargic with loss of skin turgor, low blood pressure and an absent or thready pulse. There are, however, all grades of severity, and milder cases cannot be distinguished clinically from other secretory diarrhoeas. Symptomless infections are common.
Pathogenic mechanisms
V. cholerae requires two major pathogenic mechanisms to cause disease:
The sequence of events leading to cholera is confined to the gut. The cholera vibrios are ingested in drink or food and, in natural infections, the dose must often be small. After passing the acid barrier of the stomach the organisms begin to multiply in the alkaline environment of the small intestine, where they migrate towards epithelial cells, facilitated by active motility and the production of mucinase and other proteolytic enzymes. Once the organism has penetrated the mucous layer it adheres to the enterocyte surface. In addition to toxin, co-regulated pili, haemagglutinins and lipopolysaccharide have also been implicated in adhesion.
Once adherent, the bacteria produce a potent enterotoxin known as cholera toxin. The toxin consists of five B subunits (molecular weight 11 600 Da) and a single A subunit (molecular weight 27 200 Da), and has structural, functional and antigenic similarity to heat-labile toxin expressed by strains of enterotoxigenic Escherichia coli (ETEC; see p. 283–4). The A subunit is made up of two peptides (A1 and A2) linked by a single disulphide bridge. The B subunit binds to sugar residues of ganglioside GM1 on the cells lining the villi and crypts of the small intestine. It is thought that insertion of the B subunit into the host cell membrane forms a hydrophilic transmembrane channel through which the toxic A subunit can pass into the cytoplasm. Reduction of the disulphide bond releases the A1 portion of the molecule, which has enterotoxic activity. The cholera enterotoxin causes the transfer of adenosine diphosphoribose (ADP ribose) from nicotinamide adenine dinucleotide (NAD) to a regulatory protein, which is part of the adenylate cyclase enzyme responsible for the generation of intracellular cyclic adenosine monophosphate (cAMP). The result is irreversible activation of adenylate cyclase and overproduction of cAMP. This in turn causes inhibition of uptake of Na+ and Cl− ions by cells lining the villi, together with hypersecretion of Cl− and HCO3− ions. This blocks the uptake of water, which normally accompanies Na+ and Cl− absorption, and there is a passive net outflow of water across mucosal cells, leading to serious loss of water and electrolytes.
V. cholerae may express one or more of four haemolysins: thermo-stable direct haemolysin, El Tor haemolysin, thermo-labile haemolysin and thermo-stable haemolysin. The latter three are found in pathogenic strains of V. cholerae, and all are thought to contribute to virulence.
Some strains of V. cholerae O1 that cause diarrhoea produce a toxin that differs in antigenic nature, receptor site, mode of action and genetic homology from cholera toxin.
Non-O1 V. cholerae
Non-O1 strains of V. cholerae cause mild, sometimes bloody, diarrhoea, often accompanied by abdominal cramps. Symptoms may occasionally be severe, in which case the disease resembles cholera. Wound infections may occur in patients exposed to aquatic environments, and bacteraemia and meningitis have been reported.
These strains may elaborate a wide range of virulence factors, including enterotoxins, cytotoxins, haemolysins and colonizing factors. A few produce cholera toxin.
Laboratory diagnosis
Stool specimens are inoculated into alkaline peptone water, in which vibrios grow rapidly and accumulate on the surface. After incubation for 3–6 h a loopful from the surface is inoculated on to a suitable solid medium such as thiosulphate–citrate–bile salts–sucrose (TCBS) agar. On this medium V. cholerae forms yellow sucrose-fermenting colonies, which are tested for the enzyme oxidase and for agglutination with rabbit antibodies specific for the O1 lipopolysaccharide antigens before biochemical confirmation. A fluorogenic test based on the production of lysyl aminopeptidase by Vibrio spp. has been developed.
Cholera toxin can be detected by the same techniques described for E. coli heat-labile toxin (see p. 284), but these assays have largely been replaced by antibody-based commercial kits or by rapid molecular methods. The haemolysins expressed by V. cholerae can be detected in polymerase chain reaction (PCR) assays with primers for the tcpA genes and cholera toxin can be characterized by targeting the ctxB genes: ctxB1 has been associated with the classical biotypes; ctxB2 has been linked with El Tor strains isolated in Australia; and ctxB3 has been associated with strains from South America.
Epidemiology
V. cholerae O1
A series of six pandemics of cholera, originating in the Bengal basin, ravaged the world in the nineteenth and twentieth centuries. Subsequently, cholera was contained within the endemic foci and surrounding areas of India and Bangladesh until 1961, when a seventh pandemic due to the El Tor biotype of V. cholerae O1, originally isolated from pilgrims at the quarantine station known as El Tor, began to supplant the classical biotype in India. By 1973 the El Tor biotype had entirely displaced the classical biotype in Bangladesh and spread to Indonesia, the Far East and Africa. In 1991 it reached South America, where the first epidemic in that subcontinent of the twentieth century occurred in Peru. By December 1993 more than 820 000 cases of cholera, with almost 7000 deaths, had occurred, and the epidemic had involved all Latin American countries except Uruguay (Fig. 30.2). Since 1993, the number of cases in the western hemisphere has continued to fall. The vast majority of cases of cholera now occur in Africa and Asia.
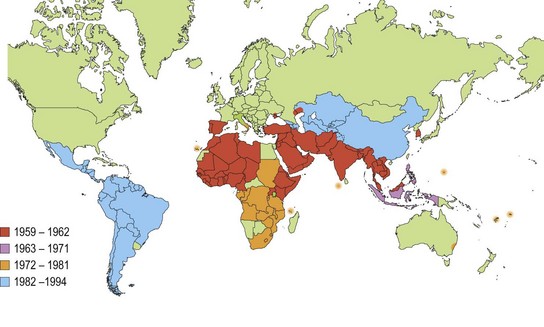
Fig. 30.2 Areas reporting indigenous cholera to the World Health Organization 1959–1994.
(Redrawn from data in: Public Health Laboratory Service 1991 Communicable Disease Report Review 1: R48–R50 and WHO 1995 Weekly Epidemiological Record 70: 201–208.)
Infection is generally spread by contaminated water or foods such as uncooked seafood or vegetables. The source of the contamination is usually the faeces of carriers or patients with cholera, but contamination can probably sometimes occur from natural aquatic reservoirs. Cholera is characteristically an infection of crowded communities with poor standards of hygiene and shared communal water supplies such as tanks, ponds, canals or rivers used for bathing, washing and household use. Outbreaks occur either as explosive epidemics, usually in non-endemic areas, or as protracted epidemic waves in endemic areas. The seasonal incidence is fairly consistent in different endemic regions, although the climatic conditions during epidemic waves may be distinctive for each region. For example, in Bangladesh the cholera season (November to February) follows the monsoon rains and ends with the onset of the hot dry months. In Calcutta the main epidemic wave (May to July) rises to its peak in the hot dry season and ends with the onset of the monsoon, but extends inland to neighbouring states during the rainy season.
Spread of infection is facilitated by the high ratio of symptomless carriers to clinical cases, which varies from 10 : 1 to 100 : 1 depending on living conditions and biotype. Symptomless carriers occur much more frequently in El Tor than in classical infections.
V. cholerae O139
In the 1990s cases of cholera indistinguishable from disease caused by V. cholerae O1 were reported in India and Bangladesh.
The new strain did not agglutinate with antisera to any of the O serogroups and was assigned to a new serogroup, O139 Bengal. However, it closely resembles V. cholerae O1 El Tor biochemically and physiologically, and may eventually attain the status of a subtype (Bengal) of that organism alongside the subtypes Inaba and Ogawa.
V. cholerae O139 is associated with free-living aquatic amoebae and other members of the zooplanktonic flora, which may act as a reservoir for the organism. In Spring 2000, an estimated 30 000 cases occurred in Dhaka, Bangladesh.
Other non-O1 V. cholerae serogroups
Strains belonging to serogroups other than O1 and O139 occur widely in aquatic environments, and many infections are associated with exposure to saline environments or consumption of seafood. They appear to survive and multiply in a wide range of foods, and it is likely that food-borne outbreaks occur as with other enteric pathogens.
Treatment
In cholera absolute priority must be given to the life-saving replacement of fluid and electrolytes. Oral rehydration therapy is often sufficient, but severe cases may require intravenous rehydration. The World Health Organization (WHO) promotes the use of oral rehydration therapy in all cases of severe diarrhoea, including that due to V. cholerae. The recommended formula is shown in Table 30.1.
Table 30.1 Formulation of oral rehydration solution recommended by the World Health Organization
| Constituent | Amount (g)a |
|---|---|
| Sodium chloride | 2.6 |
| Potassium chloride | 1.5 |
| Trisodium citrate | 2.9 |
| Glucose (anhydrous) | 13.5 |
a To be dissolved in 1 L of clean drinking water.
Tetracyclines, chloramphenicol and co-trimoxazole reduce the period of excretion of V. cholerae in the stools of patients with cholera. Tetracycline is often given to reduce environmental contamination and the risk of cross-infection. Tetracycline-resistant strains of V. cholerae O1 El Tor began to appear in the 1970s and were soon followed by the appearance of strains resistant to a wide range of antimicrobial agents, including penicillins, streptomycin, chloramphenicol, sulphonamides and trimethoprim.
Control
Public health measures used in the control of any disease spread by faecal contamination are of value in the control of cholera. Most important are the provision of safe drinking water supplies and the proper disposal of human faeces.
Although cholera can be life threatening it is easily prevented and treated. Cholera among travellers is rare and few infections are reported from industrialized countries.
Vaccines
The immune response to cholera is directed against the bacterium rather than the toxin. It is specific for a given serotype. Infection with the classical biotype is followed by almost complete immunity for several years, but infection with the El Tor biotype confers little or no immunity. It is not surprising, therefore, that the development of effective vaccines has proved difficult.
Traditional whole-cell vaccines are not very effective and are no longer recommended for travellers. Oral vaccines that combine purified B subunit and killed whole cells are now widely available and appear to be safe and protective.
Vibrio parahaemolyticus
Description
V. parahaemolyticus is a halophilic vibrio and does not grow in the absence of sodium chloride. This property is conveniently demonstrated by inoculating the organism on to cystine–lactose–electrolyte-deficient (CLED) agar, which supports the growth of non-halophilic vibrios but not halophilic species. Strains of V. parahaemolyticus from clinical specimens generally form green, non-sucrose-fermenting colonies on TCBS agar, but sucrose-fermenting strains are found in estuarine and coastal waters.
Strains associated with gastroenteritis usually express a thermostable haemolysin which causes β-haemolysis of human red cells in Wagatsuma’s agar, a special medium containing mannitol. This haemolysis is known as the Kanagawa phenomenon.
Pathogenesis
V. parahaemolyticus can cause explosive diarrhoea, but symptoms usually abate after 3 days. Other symptoms include abdominal pain, nausea and vomiting, and there may be blood in the stools. A few extra-intestinal infections have been reported, particularly from wounds.
Kanagawa-positive strains cause diarrhoea in volunteers. In contrast, volunteers have ingested 1010 cells of Kanagawa-negative strains without ill effects. Kanagawa-positive strains also adhere to human intestinal cells, whereas Kanagawa-negative strains do not. The heat-stable haemolysin disrupts the cytoskeleton of cell membranes, and causes fluid accumulation in the rabbit ileal-loop toxin test. V. parahaemolyticus also produces a thermo-labile haemolysin that causes morphological changes in tissue culture cells resembling those caused by cholera toxin and the heat-labile enterotoxin of E. coli.
Most strains from seafood and the environment are Kanagawa negative, although positive colonies can usually be found if a sufficient number are tested. It is likely that a few Kanagawa-positive strains multiply selectively in the human intestine as infection develops and predominate in the stools of patients with diarrhoea.
Laboratory diagnosis
The faeces of patients with a history of recent consumption of seafood may be examined by the methods used for V. cholerae. In the examination of seafood and sea or estuarine waters for halophilic species, including V. parahaemolyticus, enrichment culture in alkaline peptone water containing 1% sodium chloride is used. Around 13 O-types (somatic antigens) and 71 K-types (capsular antigens) are recognized; serotype O3:K6 has been detected widely in India, Asia, Africa and Latin America.
Epidemiology
V. parahaemolyticus is a common cause of diarrhoea in Japan and South-East Asia. It also causes illness associated with seafood in many other countries, including the USA and the UK. The organisms are common in fish and shellfish and in the waters from which they are harvested. Infections occur more frequently in the warmer months when the organisms are most prevalent in the aquatic environment. There is a particular risk associated with the consumption of raw seafood prepared and eaten in Japanese-style restaurants.
Extra-intestinal infections are always associated with exposure to the aquatic environment or handling of contaminated seafood.
Treatment and control
Patients with diarrhoea generally require only fluid replacement therapy. Infection can be avoided by normal food hygiene procedures and by refrigeration of seafood to reduce the possibility of bacterial multiplication. For wound infections and septicaemia, the most effective antimicrobial agents include tetracyclines, ciprofloxacin, ceftazidime and gentamicin.
Vibrio vulnificus
This halophilic species differs from other vibrios by utilizing lactose. There are three biotypes of V. vulnificus based on physiological, biochemical and serological properties. Biotype 1 is the predominant human pathogen. Biotype 2 infects eels and biotype 3 causes human wound infections.
Pathogenesis
There are three distinct clinical syndromes:
1. Rapid onset of fulminating septicaemia followed by the appearance of cutaneous lesions. More than 50% of those with primary septicaemia die. The condition is invariably associated with the consumption of raw shellfish. It is thought that the organisms enter the bloodstream by way of the portal vein or the intestinal lymph system. Elderly males with liver function defects due to alcohol abuse and people with iron-overload conditions are particularly susceptible, but any deficiency in the immune system may be a contributing factor.
2. A rapidly progressing cellulitis following contamination of a wound sustained during exposure to salt water. Infections of this kind occur in otherwise healthy persons as well as in the debilitated, and are characterized by wound oedema, erythema and necrosis, which progresses to septicaemia only occasionally. The infection can be rapidly fatal.
3. Acute diarrhoea following the consumption of shellfish. This is less common; victims generally have mildly debilitating underlying conditions. Death is rare.
Pathogenic mechanisms
Strains of V. vulnificus are acid tolerant, surviving in stomach acid by breaking down amino acids to produce amines and CO2. A capsular polysaccharide and long-chain lipopolysaccharide enable pathogenic strains to resist phagocytosis and the killing effects of human serum complement. Two siderophores, a catechol (vulnibactin) and a hyroxamate, have been reported indicating the presence of high-affinity iron-uptake systems; however, many strains are unable to obtain ferric ions bound to human transferrin. Patients with iron overload disorders such as haemochromatosis are highly susceptible; high levels of free serum iron are thought to facilitate the rapid septicaemia observed in patients infected with this organism.
Several toxins may contribute to tissue damage. They include a metalloprotease, a collagenase, a mucinase and a cytotoxin. A vascular permeability factor has also been described. Strains of V. vulnificus express El Tor haemolysin and thermo-labile haemolysin, but a role in pathogenesis has not been demonstrated.
Epidemiology
Infections occur most frequently in areas where the water temperature remains high throughout the year, such as the mid-Atlantic and Gulf coast states of the USA. They are much more common during the warmer months of the year when V. vulnificus is most abundant. Wound infections are associated with injuries sustained in the aquatic environment, whereas septicaemic infections are associated with the consumption of raw shellfish.
Treatment
V. vulnificus wound infections and primary septicemia require early antimicrobial treatment to reduce morbidity and mortality from the illness and to prevent complications. The most effective antimicrobial agents include: tetracyclines; fluoroquinolones such as ciprofloxacin; ceftazidime; and gentamicin. Because of the high case-fatality rates, it is particularly important for clinicians to suspect V. vulnificus wound or bloodstream infections in persons with shellfish or warm seawater exposure and a history of chronic liver disease or conditions of iron overload.
Vibrio alginolyticus
Description
Vibrio alginolyticus is a halophilic organism formerly regarded as biotype 2 of V. parahaemolyticus. It fails to grow on CLED agar but grows in the presence of 10% sodium chloride. It forms large, yellow (sucrose-fermenting) colonies on TCBS. There is pronounced swarming on non-selective solid media.
Pathogenesis
V. alginolyticus causes wound and ear infections. Clinical features include mild cellulitis and a seropurulent exudate. The pathogenic mechanisms are not fully understood although genetic homology between V. alginolyticus, V. cholerae and V. parahaemolyticus has been shown.
Epidemiology
This organism is widely distributed in seawater and seafood and is probably the most common vibrio found in these sources in the UK. It occurs in large numbers throughout the year. Infections are invariably associated with exposure to sea water. Strains appear to be sensitive to ciprofloxacin.
Other vibrios
• Photobacterium damsela is a halophilic marine vibrio found in tropical and semitropical aquatic environments. It is associated with severe infections of wounds acquired in warm coastal areas.
• Vibrio fluvialis is easily confused with Aeromonas hydrophila, but can be differentiated by growth on media containing 6% sodium chloride. Patients experience diarrhoea, abdominal pain, fever and dehydration. Low numbers of V. fluvialis can be isolated from fish and shellfish, and from warm seawater. It seems likely that infection is from contaminated seafood. Strains of V. fluvialis express El Tor haemolysin, and a vacuolating toxin acting on HeLa cells, but the role of these in pathogenesis has not been demonstrated.
• Vibrio hollisae is associated with bacteraemia and diarrhoea, especially in warm coastal areas, such as the Gulf of Mexico. Infections are strongly associated with the consumption of raw seafood. The organism is difficult to isolate because it grows poorly on TCBS agar. Strains of V. hollisae exhibit gene sequences homologous with those encoding the thermo-stable haemolysin of V. parahaemolyticus.
• Vibrio mimicus is a non-halophilic vibrio named for its similarity to V. cholerae and occurrence in similar environments. Most isolates are from the stools of patients who develop gastroenteritis after consumption of raw oysters, although a few cases of otitis media have also been reported. In vitro, strains of V. mimicus express thermo-stable direct haemolysin, El Tor haemolysin and thermo-labile haemolysin.
• Vibrio furnissii is most commonly isolated from stool samples. It has occasionally been implicated in gastroenteritis.
Other aquatic organisms that are probably related to vibrios include Aeromonas spp. and Plesiomonas shigelloides. Aeromonas spp., notably A. hydrophila, have been implicated in diarrhoea and occasionally cause more serious infection in compromised individuals. A. salmonicida is an economically important pathogen of fish. P. shigelloides is an organism of uncertain taxonomic status that sometimes causes water-borne outbreaks of diarrhoea in warm countries.
Mobiluncus
The name Mobiluncus was first proposed for a group of curved, motile, Gram-variable, anaerobic bacteria isolated from the vagina of women with bacterial vaginosis. Its taxonomic position is uncertain. Studies of 16S RNA suggest that the genus is most closely related to Actinomyces.
Description
There are two subspecies: M. curtisii and M. mulieris; M. curtisii can be divided further into two sub-species M. curtisii ssp. curtisii and M. curtisii ssp. holmesii. M. curtisii is short rod (mean length 1.5 µm) and Gram-variable, whereas M. mulieris is longer (mean length 3.0 µm) and Gram-negative. Both have multiple flagella originating from the concave aspect of the cells. Cell wall studies have revealed no outer membrane, but both species are thought to be Gram-negative.
Pathogenesis
Bacterial vaginosis
Mobiluncus spp. are isolated from 97% of women with bacterial vaginosis (non-specific vaginitis) and is rarely found in the vagina of healthy women. Bacterial vaginosis appears to be a polymicrobial infection, with certain organisms playing a key role, especially when they overgrow the lactobacilli of the normal flora, raising the vaginal pH above 4.5. The condition is characterized by the presence of a thin, homogeneous vaginal discharge with a characteristic ‘rotten fish’ smell. This becomes more pronounced on alkalization, and can be evoked by placing a drop of potassium hydroxide solution on the fresh exudate on a slide or the speculum used for the vaginal examination. The characteristic smell is ascribed to amines produced by one or more of the bacterial species that form the complex microbial flora of the vagina.
Mobiluncus are frequently found in association with Gardnerella vaginalis (see below) and with other organisms that may also be of aetiological importance. It appears that both the combination of species and their relative numbers are of importance in the development of the syndrome. The organisms may be isolated from the urethra of male consorts of infected women but do not persist in men once condom use is implemented.
Mobiluncus spp. are occasionally isolated from extragenital sites, especially from breast abscesses. A fatal case of bacteraemia caused by M. curtisii has been reported.
Pathogenic mechanisms
The mechanisms that allow Mobiluncus spp. to cause disease are poorly understood. They express pili and are able to obtain iron from lactoferrin, but the role of these in the pathogenesis of disease is speculative. Strains of Mobiluncus spp. express a relatively thermo-stable toxin that is cytotoxic for Vero (African green monkey) cells, but a role in pathogenesis has not been demonstrated. Primates can be infected experimentally and animal studies may help to elucidate the pathogenesis of vaginitis.
Laboratory diagnosis
Microscopy of fresh unstained vaginal exudate reveals epithelial cells covered with adherent bacteria (clue cells). Mobiluncus spp. can be grown in Columbia blood broth and peptone–starch–dextrose broth containing 10% horse serum. The organisms are essentially anaerobic but grow slowly in 5% oxygen in nitrogen. They do not produce oxidase or catalase but ferment sugars.
A multiplex PCR comprising primers for both Mobiluncus spp. and G. vaginalis has been applied to cases of bacterial vaginosis.
Treatment
Treatment of bacterial vaginosis aims to restore the normal vaginal flora by eliminating Mobiluncus and other organisms that may be involved. Oral or intravaginal metronidazole and clindamycin have been successfully used. Although M. mulieris is more susceptible than M. curtisii to metronidazole, treatment with this drug appears to eliminate all Mobiluncus species in patients with vaginosis.
Gardnerella
Gardnerella vaginalis (formerly Corynebacterium vaginale or Haemophilus vaginalis) is commonly isolated together with Mobiluncus spp. in bacterial vaginosis. It has been implicated in cases of cervical cancer and infections of the urinary tract, but because the organism is frequently present in the vagina of asymptomatic patients its role in disease is equivocal.
Description
G. vaginalis is a non-motile, non-sporing, micro-aerophilic coccobaccilus. It is Gram-variable, but because the cell wall contains lipopolysaccharide it appears to be Gram-negative.
Laboratory diagnosis
G. vaginalis grows on various media, such as Columbia agar containing colistin and nalidixic acid as selective agents. Plates are incubated at 35–37°C in 5–10% carbon dioxide for 2–3 days. On media containing human erythrocytes, the organism produces zones of β-haemolysis. G. vaginalis does not produce oxidase or catalase, but ferments starch and hydrolyses hippurate, and these properties provide a means of presumptive identification. Genes encoding the 60 kDa heat-shock protein chaperonin Cpn 60 can be used to detect G. vaginalis by PCR.
Pathogenesis
Whether G. vaginalis can cause disease in isolation is unclear. The organisms may simply flourish in the vaginal environment provided by other bacteria. Infections with G. vaginalis are associated with proteolysis yielding nitrous products such as cadavarines and putrescines which contribute to the characteristic odour resulting from these infections. The ability to lyse human red cells offers a mechanism for acquiring metabolic iron and may aid multiplication. Similarly, G. vaginalis can acquire ferric ions from human lactoferrin. Strains produce various mucinases such as sialidase and proline dipeptidase, which are thought to damage the vaginal mucosa as part of the pathogenic process. Patients infected with G. vaginalis produce IgA antibodies to the haemolysin, but whether these can be used for serodiagnosis is not known.
Spirillum minus
The organism commonly known as Spirillum minus is of uncertain taxonomic position since a type-strain for this taxon has not been identified. Along with Streptobacillus moniliformis (see p. 348) it is one of the causes of rat bite fever in humans.
Description
S. minus is a spiral Gram-negative organism about 2–5 µm in length and 0.2 µm in diameter. Longer forms of up to 10 µm may be observed. The regular short coils have a wavelength of 0.8–1.0 µm. The organisms are very actively motile, showing darting movements like those of a vibrio. The movement is due to polar flagella, which vary in number from one to seven at each pole. The organisms can be demonstrated in fresh specimens by dark-ground illumination or by staining with Giemsa or Leishman stains. The organism has not been reliably cultivated on artificial media and many of its properties are unknown. It can be cultured in vivo by intraperitoneal injection into guinea-pigs or mice.
Laboratory diagnosis
In rat bite fever, S. minus may be demonstrated in the local lesion, in the regional lymph glands or in the blood by direct microscopy or by animal inoculation. In theory, S. minus DNA may be amplified by broad range bacterial 16S rRNA gene primers.
Pathogenesis
S. minus is transmitted to humans by animal bite; human-to-human transmission has not been recorded. The clinical syndrome of rat bite fever begins with an acute onset of fever and chills 1–4 weeks after the bite, although infection without fever may occur. The bite usually heals before the onset of symptoms but it often re-ulcerates. Local lymphadenopathy and lymphangitis develop with the onset of fever and systemic disease. A generalized rash with large brown to purple macules is usually observed, but some patients present with urticarial lesions. A roseolar rash may spread from the area of the original bite. Fever usually declines within 1 week before returning again after a few days; the fever may then recur in an episodic fashion for months or even years.
Endocarditis, meningitis, hepatitis, nephritis and myocarditis are rare complications. In most untreated cases, symptoms resolve within 2 months, after 6–8 episodes of fever, and fewer than 6.5% of untreated cases are fatal.
Epidemiology
S. minus occurs naturally in wild rats and other rodents, causing bacteraemia. Human rat bite fever occurs mainly in Africa, Japan (where it is known as sodoku) and the Far East. There have been a few reports from Europe and the USA. The disease is prevalent in laboratory workers who handle rats, and in children who live in rat-infested homes.
Chiang S-R, Chuang Y-C. Vibrio vulnificus: clinical manifestations, pathogenesis and antimicrobial therapy. Journal of Microbiology, Immunology, and Infection. 2003;36:81–88.
Chou TN, Lee YT, Lai YY, et al. Prognostic factors for primary septicemia and wound infection caused by Vibrio vulnificus. American Journal of Emergency Medicine. 2010;28:424–431.
Forsum U, Holst E, Larsson PG, et al. Bacterial vaginosis – a microbiological and immunological enigma. Acta Pathologica, Microbiologica et Immunologica Scandinavica. 2005;113:81–90.
Jenkins SG. Rat-bite fever. Clinical Microbiology Newsletter. 1988;10:57–59.
Jones MK, Oliver JD. Vibrio vulnificus: disease and pathogenesis. Infection and Immunity. 2009;77:1723–1733.
Sack DA, Sack RB, Nair GB, Siddique AK. Cholera. Lancet. 2004;363:223–233.
Safa A, Nair GB, Kong RY. Evolution of new variants of Vibrio cholerae O1. Trends in Microbiology. 2010;18:46–54.
Sanchez J, Holmgren J. Virulence factors, pathogenesis and vaccine protection in cholera and ETEC diarrhea. Current Opinion in Immunology. 2005;17:1–11.
Spiegal CA. Bacterial vaginosis. Reviews in Medical Microbiology. 2002;13:43–51.
Centers for Disease Control and Prevention. Vibrio vulnificus. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/vibriov/.
Centers for Disease Control and Prevention. Vibrio parahaemolyticus. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/vibriop/.
Health Protection Agency. Cholera. http://www.hpa.org.uk/infections/topics_az/cholera/menu.htm.
Todar’s Online Textbook of Bacteriology. Vibrio cholerae and Asiatic Cholera. http://textbookofbacteriology.net/cholera.html.