31 Haemophilus
Respiratory infections; meningitis; chancroid
Key points
• Most strains of Haemophilus influenzae are non-capsulate but some strains possess a polysaccharide capsule (types a–f).
• H. influenzae type b (Hib) is a major human pathogen that causes invasive infections, including meningitis and epiglottitis.
• Non-capsulate strains cause 90% of non-invasive respiratory infections, including otitis media and acute exacerbations of chronic obstructive airway disease.
• Following the introduction of Hib conjugate vaccine, the number of invasive H. influenzae infections declined dramatically; most invasive infections are now caused by non-typable strains.
• 15–20% of H. influenzae strains are ampicillin resistant (β-lactamase-mediated); ceftriaxone is the treatment of choice for invasive disease.
• Conjugate Hib vaccine is routinely offered to infants at 2, 3, 4 and 12 months in the UK.
• H. ducreyi causes chancroid, sexually transmitted genital ulcers that are common in Africa and South-East Asia but rare in the UK. Single-dose therapy with azithromycin or ceftriaxone is effective.
Haemophilus influenzae is associated with a variety of invasive infections such as meningitis, epiglottitis, pneumonia and septic arthritis, and localized disease of the respiratory tract including bronchitis and otitis media. Other haemophili of medical importance include H. ducreyi, the causative organism of chancroid, and H. parainfluenzae. Two related organisms, H. aphrophilus and H. paraphrophilus, are now combined as a single species within the genus Aggregatibacter as A. aphrophilus; like H. parainfluenzae they are occasionally encountered in patients with infective endocarditis and other miscellaneous conditions.
During the influenza pandemic of 1889–1892, Pfeiffer noted the constant presence of large numbers of small bacilli in the sputum of patients affected with the disease and suggested that the bacillus was the causative agent. Although the true aetiological agent of influenza was shown in 1933 to be a virus, it remains possible that secondary infection with H. influenzae contributed to the high mortality rate seen in the 1889–92 and 1918–19 pandemics.
Description
Haemophili are small, pleomorphic, Gram-negative rods or coccobacilli with occasional longer, filamentous forms (Fig. 31.1). Some strains of H. influenzae produce a polysaccharide capsule, which is demonstrable by capsule stains and a Quellung reaction (swelling of the capsule) with type-specific antisera. There are six capsular types, designated a–f, which can be identified by a polymerase chain reaction (PCR) method. The most important is type b, a polymer of ribosyl ribitol phosphate.
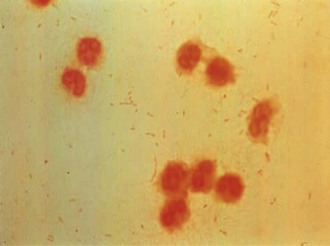
Fig. 31.1 Gram stain of cerebrospinal fluid showing Haemophilus influenzae: Gram-negative pleomorphic coccobacilli.
All species of Haemophilus are catalase and oxidase positive; they reduce nitrate to nitrite and ferment glucose. Patterns of acid production from other carbohydrates are used to differentiate the species. H. influenzae can be divided into eight biotypes on the basis of indole production, urease activity and ornithine decarboxylase reactions. Biotypes I–III are the most common, and most invasive (type b) organisms are biotype I.
Growth requirements
Growth depends on a requirement for two factors, termed X and V:
• X factor (haemin) is required for the synthesis of cytochrome c and other iron-containing respiratory enzymes.
• V factor is nicotinamide adenine dinucleotide (NAD), NAD phosphate or certain unidentified precursor compounds. It is essential for oxidation-reduction processes in cell metabolism.
Different species of Haemophilus require either or both of these factors, and growth factor dependence forms an important step in identification.
Ordinary blood agar contains X and V factors, but growth of H. influenzae, which requires both factors, is poor. Growth is enhanced if the medium is supplemented with NAD. Streaking an organism that excretes this substance (e.g. Staphylococcus aureus) across the surface of the agar stimulates growth in its vicinity (satellitism). Heating blood agar for a few minutes at 70–80°C until it turns brown (chocolate agar) much improves the growth of H. influenzae. This process removes serum NADase, which limits the amount of V factor, and also liberates extra X and V factors from the red cells into the medium. X factor is heat stable, but heating of media at 120°C for several minutes destroys V factor.
Anaerobic growth considerably reduces the haemin requirement of X-dependent species. A. aphrophilus has a requirement for carbon dioxide, but this character may be lost on subculture. H. influenzae does not require a carbon dioxide-enriched atmosphere, but often grows better in such conditions.
Haemophilus influenzae
Pathogenesis
H. influenzae is an obligate parasite of human mucous membranes; it is not found in any other animal species. It colonizes the throat and nasopharynx, and to a lesser extent the conjunctivae and genital tract. The species is associated with two types of infection which are quite distinct in their epidemiological profiles: invasive infections and non-invasive infections (Table 31.1).
Table 31.1 Clinical spectrum of Haemophilus influenzae infections
| Type of infection | Age group | Strains |
|---|---|---|
| Invasive | 90% children <4 yearsa | 90% H. influenzae type ba 10% non-capsulate strains 1% types e and f |
| Neonatal and maternal | Neonates; pregnant and parturient women | >90% non-capsulate strains |
| Non-invasive respiratory | Children and adults | >90% non-capsulate strains |
a Percentages observed before the introduction of Hib conjugate vaccine; since the introduction of routine vaccination, the epidemiology has changed (see text).
Invasive infections
Most invasive infections are caused by H. influenzae type b (Hib). Meningitis is the most common manifestation, but H. influenzae also causes epiglottitis, septic arthritis, osteomyelitis, pneumonia and cellulitis. In some cases the patient develops a bacteraemia without a clearly defined focus of infection.
These infections are unusual in the first 2 months of life, but are otherwise seen mainly in early childhood. Most cases occur in children under 2 years of age, but acute epiglottitis has a peak incidence between 2 and 4 years of age. The incidence of invasive disease in children has been reduced dramatically in countries where a conjugate Hib vaccine has been introduced, and consequently the epidemiology is changing (see below).
The polysaccharide capsule is the major virulence factor for Hib. When the organism invades the bloodstream, the capsule enables the organisms to evade phagocytosis and complement-mediated lysis in the non-immune host. The rarity of infections in the first 2 months of life correlates with the presence of maternal capsular antibodies, and the occurrence of infection in early infancy with the absence of such antibodies. As the prevalence and mean level of capsular antibodies in the population rise, H. influenzae type b infections become less common (Fig. 31.2).
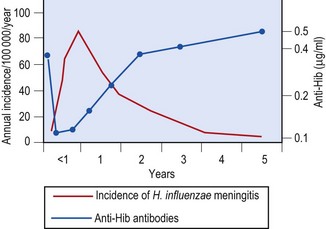
Fig. 31.2 Incidence of H. influenzae meningitis during the first 5 years of life and the corresponding mean level of anti-H. influenzae type b (Hib) capsular polysaccharide antibodies.
(From Peltola H, Käyhty H, Sivonen A, Mäkelä H 1977 Haemophilus influenzae type b capsular polysaccharide vaccine in children: a double blind field study of 100 000 vaccinees 3 months to 5 years of age in Finland. Pediatrics 60: 730–737.
What determines whether acquisition of type b organisms in a susceptible host will lead to asymptomatic carriage and the stimulation of protective antibodies, or to the induction of invasive disease, is unclear. However, animal experiments suggest that when invasion occurs the organism penetrates the submucosa of the nasopharynx and establishes systemic infection through the bloodstream.
The type b capsular polysaccharide facilitates all phases of the invasion process. Other virulence factors that may be involved include:
• fimbriae, which assist attachment to epithelial cells
• immunoglobulin A (Ig A) proteases, which are also involved in colonization
• outer membrane proteins and lipopolysaccharide, which may contribute to invasion at several stages.
Initiation of invasive infection may be potentiated by intercurrent viral infection. Host genetic factors and immunosuppression may also play a role. It is unclear whether it is exposure to H. influenzae type b, or some other organism (e.g. Escherichia coli K100) possessing cross-reacting antigens, that usually stimulates natural protective antibody production.
Since the introduction of routine infant immunization with conjugate Hib vaccine, invasive disease caused by non-capsulate (non-typable) strains has become more common than that caused by Hib in the UK. Meningitis and bacteraemia due to non-capsulate H. influenzae is sometimes seen in the neonate. Infections with non-capsulate strains may also occur in children and adults. Pneumonia and bacteraemia are the most common manifestations, often in patients with an underlying disease, notably chronic lung disease or malignancy. The highest rates occur in patients aged over 60 years, and the case fatality rate is high.
Invasive infections due to H. influenzae of serotypes other than b (principally types a, e and f) are uncommon. The spectrum of disease is similar to that seen with type b.
Non-invasive disease
H. influenzae produces a variety of local infections, which are often associated with some underlying physiological or anatomical abnormality. Most are caused by non-capsulate strains. The most common are:
Acute sinusitis and otitis media are usually initiated by viral infections, which predispose to secondary infection with potentially pathogenic components of the resident microbial flora. The mechanisms may involve:
• obstruction to the outflow of respiratory secretions
• decreased clearance of micro-organisms via the normal mucociliary mechanism
Acute exacerbations of chronic obstructive airway disease are similarly initiated by acute viral infections. Respiratory viruses compromise an already impaired mucociliary clearance mechanism in patients with chronic lung disease and allow bacterial colonization of the lower respiratory tract. In this situation H. influenzae can establish purulent infection and further damages pulmonary function by a direct toxic effect on cilia.
Laboratory diagnosis
Gram-stained smears of cerebrospinal fluid, pus, sputum or aspirates from joints, middle ears or sinuses can provide a rapid presumptive identification. Haemophili tend to stain poorly, and dilute carbol fuchsin is a better counterstain than neutral red or safranin.
The viability of H. influenzae in clinical specimens declines with time, particularly at 4°C. Specimens should therefore be transported to the laboratory and cultured without delay. Chocolate agar is a good, general purpose, culture medium and can be used without further supplementation for specimens obtained from sites that would normally be expected to be sterile. Plates should be incubated in an aerobic atmosphere enriched with 5–10% carbon dioxide.
Specimens of expectorated sputum inevitably become contaminated by upper respiratory flora, commonly including H. influenzae, and the finding of the organism in such specimens does not necessarily signify involvement in disease. Support for the significance of H. influenzae is provided if, in a purulent sample, the organism is present as the predominant isolate, or in a viable count of over 106 colony-forming units per mL. Addition of bacitracin (10 international units/mL) facilitates the selective isolation of H. influenzae from mixed cultures of respiratory organisms. Obtaining bronchial secretions by broncho-alveolar lavage reduces the problem of contamination with commensal organisms.
The temptation to obtain throat swabs in patients with suspected acute epiglottitis should be resisted, as attempts to obtain the sample may precipitate complete airway obstruction. Blood culture is indicated for patients with suspected invasive disease and is usually positive in those with acute epiglottitis.
Identification
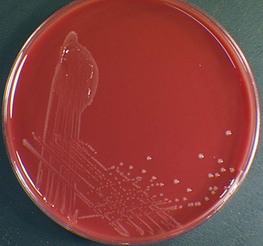
H. influenzae grows poorly on blood agar. On chocolate agar the colonies are smooth, grey or colourless (Fig. 31.3), with a characteristic seminal odour. Confirmation of the identity depends on demonstrating a requirement for one or both of the growth factors, X and V:
• H. influenzae requires both X and V factors.
• H. parainfluenzae requires V factor alone.
• A. aphrophilus (H. aphrophilus) and H. ducreyi require X factor alone.
The culture is plated on nutrient agar that is deficient in both X and V factor, and paper discs containing X factor, V factor and X + V factor are placed on the surface of the agar. After overnight incubation, growth is observed around the discs supplying the necessary growth factors (Fig. 31.4).
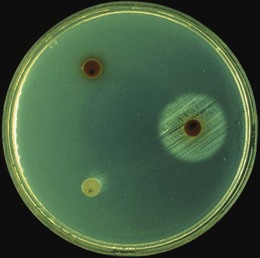
Fig. 31.4 Determination of the growth factor requirement of H. influenzae. Growth around the disc containing both X and V factors (right-hand disc), but not round discs of the individual factors (left-hand discs), indicates that the organism is H. influenzae (see text).
PCR techniques are used to identify Haemophilus species in clinical specimens and as confirmatory tests on isolates. The capsular type of H. influenzae isolates is determined by slide agglutination with type-specific antisera, or a PCR-based method.
Antigen detection
The detection of type b polysaccharide antigen in body fluids or pus is useful, particularly in patients who received antibiotics before specimens were obtained. A rapid latex agglutination test with rabbit antibody to type b antigen is used most commonly.
In the absence of confirmatory cultures, the results should be regarded with caution as some serotypes of Streptococcus pneumoniae and E. coli may share similar antigens.
Antibiotic sensitivity tests
Accurate determination of the antibiotic susceptibility of H. influenzae requires careful standardization of the methodology. Disc tests are less reliable for detecting enzyme-mediated ampicillin (β-lactamase) and chloramphenicol (chloramphenicol acetyltransferase) resistance than microbiological or biochemical techniques that demonstrate antibiotic inactivation.
Treatment
H. influenzae is usually susceptible to ampicillin (or amoxicillin), chloramphenicol and tetracyclines. Among cephalosporins, compounds such as cefuroxime, cefotaxime and ceftriaxone are highly active. Other antibiotics active against H. influenzae include co-amoxiclav, ciprofloxacin, azithromycin and clarithromycin.
Ceftriaxone (or a related cephalosporin such as cefotaxime) is the antibiotic of first choice for the treatment of meningitis and acute epiglottitis. It is bactericidal for H. influenzae, achieves good concentrations in the meninges and cerebral tissues, and is highly effective.
β-Lactamase mediated resistance to ampicillin is now encountered in up to 15% of type b strains and about 20% of invasive non-capsulate isolates in the UK. Occasional strains are resistant to ampicillin through alterations in penicillin binding protein 3 (β-lactamase-negative ampicillin resistance–BLNAR)and a few strains demonstrate both mechanisms of resistance. For these reasons ampicillin should not be used as a single agent in meningitis when H. influenzae is a possibility and the results of sensitivity tests are not available. Resistance to chloramphenicol may also be encountered, but in most parts of the world this remains uncommon.
Antibiotic therapy is only one component of the clinical management of patients with haemophilus meningitis and full supportive care is required to achieve the most favourable outcome. Skilled medical and nursing care is also vital in the management of acute epiglottitis, where maintenance of a patent airway is crucial.
For the treatment of less serious respiratory infections, such as otitis media, sinusitis and acute exacerbations of chronic bronchitis, oral antibiotics such as amoxicillin, co-amoxiclav and clarithromycin are all effective.
Epidemiology of invasive disease
Non-capsulate H. influenzae are present in the nasopharynx or throat of 25–80% of healthy people; capsulate strains (about half of which are capsular type b) are present in 5–10%. H. influenzae type b is an important cause of serious systemic infection in children throughout the world. Meningitis is more common in winter months, in families of low socio-economic status and in household contacts of a case. The disease is usually seen in the youngest member of a family and uncommonly in children who have no siblings. Household contacts of patients with invasive disease have an increased risk of acquiring infection if they are less than 5 years of age. The risk for children under 2 years of age is 600–800-fold higher than the age-adjusted risk for the general population.
Outbreaks of infection have been described in close communities, such as nursery schools. Contact with a case in a day care centre or nursery has also been associated with increased attack rates in children under 2 years of age, although the calculated risk is lower than that seen in household contacts.
Very high incidence rates have been reported in certain populations of Australian Aborigines, American Indians and Inuits. In these racial groups the peak incidence of infection occurs at a younger age. By contrast, very low rates have been reported in Hong Kong Chinese. It is possible that socio-economic considerations are important in determining such racial differences, but host genetic factors may also play a role. Immunosuppression, whether iatrogenic or associated with malignancies (especially Hodgkin’s disease), asplenia or agammaglobulinaemia, also predispose to invasive disease. There is seasonal variation, with most cases occurring during the winter months.
The mortality rate associated with H. influenzae meningitis is around 5%. Neurological sequelae, including intellectual impairment, seizures and profound or severe hearing loss, may be present in 10–20% of survivors.
H. influenzae type b disease in the UK
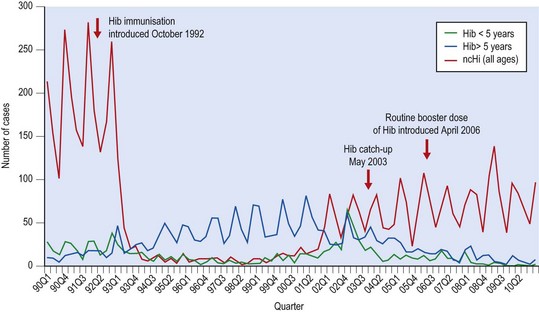
Before the introduction of conjugate Hib vaccine into the infant immunization schedule in 1992, approximately 1500 cases of invasive Hib disease, including 900 cases of meningitis, occurred in the UK every year, with 60 deaths. Immunization rates have remained high (about 93%), and between 1992 and 1999 H. influenzae type b disease in children less than 5 years old fell by 95% (Fig. 31.5). In 1998 only 21 cases of invasive Hib disease in children under 5 years were reported in England and Wales. From 1999 there was a small but gradual increase in the number of cases, most notably in fully immunized children born in 2000 and 2001, but also in older children and adults. In 2003 children over 6 months and under 4 years of age were offered a booster dose of Hib vaccine and in 2006 a routine booster dose of Hib vaccine, given in combination with meningococcus group C conjugate vaccine, was incorporated into the UK infant immunization schedule. These campaigns have had a marked effect on the incidence of invasive Hib disease, most dramatically in 1–4-year-olds, but also in older children and adults.
Control
Active immunization
Early haemophilus vaccines consisting of purified type b capsular polysaccharide were poorly immunogenic in children less than 2 years old and in patients with immune deficiency. Conjugate vaccines in which the polysaccharide is covalently coupled to proteins such as tetanus toxoid, a non-toxic variant of diphtheria toxin, Neisseria meningitidis outer membrane protein or diphtheria toxoid produce a lasting anamnestic response, which is not age-related and may be effective in high-risk patients who respond poorly to polysaccharide vaccine alone. In the UK, H. influenzae type b vaccine is offered routinely to infants at 2, 3 and 4 months of age as part of a pentavalent diphtheria, tetanus, pertussis, polio, Hib vaccine (see Ch. 70). A Hib booster dose (in combination with meningococcus C vaccine), given at 12 months of age, was introduced in 2006. Immunization of infants significantly reduces pharyngeal carriage of Hib, but has no effect on the carriage of other capsular types or non-capsulate strains.
Conjugate Hib vaccine is recommended for children and adults with splenic dysfunction, because they are at increased risk of invasive Hib infection.
Prophylaxis
Rifampicin (20 mg/kg (maximum 600 mg) for children >3 months and adults; 10 mg/kg for infants <3 months) given orally once daily for 4 days eradicates carriage of H. influenzae and prevents secondary infection in household and nursery contacts.
Unvaccinated or partially vaccinated (including those who were immunized only in infancy) siblings of the index case who are younger than 10 years old should be appropriately immunized with the Hib conjugate vaccine. The index case should also be given a dose of Hib vaccine before discharge from hospital to ensure high levels of antibodies and long-term protection against Hib. Household contacts and the index case should be given rifampicin chemoprophylaxis to eradicate carriage if there is a vulnerable individual in the household (an immunosuppressed or asplenic person of any age, or any child younger then ten years – this may be the index case).
When two or more cases of Hib disease occur in a nursery or playgroup within 120 days, chemoprophylaxis should be offered to all room contacts – carers and children. Any unvaccinated or partially vaccinated children under 10 years of age should be appropriately immunized against Hib.
The widespread use of conjugate vaccine may soon render chemoprophylaxis unnecessary.
Other haemophili
H. influenzae biogroup aegyptius
This organism, formerly known as the Koch–Weeks bacillus and H. aegyptius, is now classified as a subgroup of H. influenzae. It is indistinguishable from H. influenzae biotype III in routine tests, but can be identified by a PCR method. It causes a purulent conjunctivitis and Brazilian purpuric fever, a clinical syndrome first recognized in Brazil in 1984, in which conjunctivitis proceeds to an overwhelming septicaemia resembling fulminating meningococcal infection. This virulent clone has not been reported since the early 1990s. Ampicillin in combination with chloramphenicol has been successful when treatment has been started sufficiently early.
H. ducreyi
H. ducreyi is responsible for a sexually transmitted infection, chancroid, which is most prevalent in tropical regions, particularly Africa and South-East Asia. Patients present with painful penile ulcers (soft sore or soft chancre) and inguinal lymphadenitis. Typical small Gram-negative bacilli can be seen in material from the ulcers or in pus from lymph node aspirates. It is likely that the lesions of chancroid have facilitated the transmission of human immunodeficiency virus (HIV) in some tropical countries.
H. ducreyi is an extremely fastidious organism requiring specialized culture media. A multiplex PCR has been developed for the simultaneous amplification of DNA targets from H. ducreyi, Treponema pallidum and herpes simplex virus types 1 and 2.
Susceptibility to antimicrobial agents varies geographically. Many strains are β-lactamase producers and resistance to tetracycline and co-trimoxazole is common. Chancroid is best treated with azithromycin, ceftriaxone, ciprofloxacin or erythromycin. Azithromycin and ceftriaxone have the advantage of single-dose therapy. Strains with intermediate resistance to ciprofloxacin or erythromycin have been reported. Treatment failures are much more likely in patients with concurrent HIV infection. The use of condoms dramatically reduces the transmission of H. ducreyi. All sexual partners should be identified by contact tracing and treated.
An unrelated Gram-negative rod, Calymmatobacterium granulomatis, causes a somewhat similar sexually transmitted disease, granuloma inguinale or donovanosis, in parts of the tropics. Intracellular organisms, known as Donovan bodies (not to be confused with the Leishman–Donovan bodies of leishmaniasis), can be demonstrated in the stained smears from the lesions. Tetracyclines are usually used in treatment.
Related organisms
H. parainfluenzae, A. aphrophilus (formerly H. aphrophilus and H. paraphrophilus; see p. 324) and A. actinomycetemcomitans (formerly Actinobacillus actinomycetemcomitans) are occasionally implicated in human disease, notably infective endocarditis, but also dental infections, lung abscess and brain abscess. Endocarditis is usually treated successfully with a combination of ampicillin and gentamicin.
Alfa M. The laboratory diagnosis of Haemophilus ducreyi. Canadian Journal of Infectious Diseases and Medical Microbiology. 2005;16:31–34.
Coker TR, Chan LS, Newberry SJ, et al. Diagnosis, microbial epidemiology and antibiotic treatment of acute otitis media in children. Journal of the American Medical Association. 2010;304:2161–2169.
Harrison LH, Simonson V, Waldman EA. Emergence and disappearance of a virulent clone of Haemophilus influenzae biogroup aegyptius, cause of Brazilian Purpuric Fever. Clinical Microbiology Reviews. 2008;21:594–605.
Kelly DF, Moxon ER, Pollard AJ. Haemophilus influenzae type b conjugate vaccines. Immunology. 2004;113:163–174.
Ladhani S, Neely F, Heath PT, et al. Recommendations for the prevention of secondary Haemophilus influenzae type b (Hib) disease. Journal of Infection. 2009;58:3–14.
Ladhani S, Slack MPE, Heath PT, et al. Invasive Haemophilus influenzae disease, Europe, 1996–2006. Emerging Infectious Diseases. 2010;16:455–462.
Peltola H. Worldwide Haemophilus influenzae type b disease at the beginning of the 21st century: global analysis of the disease burden 25 years after the use of the polysaccharide vaccine and a decade after the advent of conjugates. Clinical Microbiology Reviews. 2000;13:302–317.
Tristram S, Jacobs MR, Appelbaum PC. Antimicrobial resistance in Haemophilus influenzae. Clinical Microbiology Reviews. 2007;20:368–389.
Ulanova M, Tsang RSW. Invasive Haemophilus influenzae disease: changing epidemiology and host-parasite interactions in the 21st century Infection. Genetics and Evolution. 2009;9:594–605.
Watt JP, Wolfson LJ, O’Brien KL, et al. Burden of disease caused by Haemophilus influenzae type b in children younger than 5 years: global estimates. Lancet. 2009;374:903–911.
Clinical Effectiveness Group (British Association for Sexual Health and HIV, BASSH). National Guideline for the Management of Chancroid. http://www.bashh.org/documents/85/85.pdf, 2007.
UK Department of Health. Haemophilus influenzae type b (Hib) Immunization. http://www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/@dh/@en/documents/digitalasset/dh_125943.pdf.