35 Yersinia, pasteurella and francisella
Plague; pseudotuberculosis; mesenteric adenitis; pasteurellosis; tularaemia
Key points
• Yersinia pestis is the cause of human plague, transmitted to humans from rats and other rodents by their fleas. Pneumonic plague is transmitted from person to person by droplet infection.
• There are three main types of disease: bubonic, pneumonic and septicaemic plague. All are highly fatal without prompt treatment.
• The organism is readily cultured, but a polymerase chain reaction method is preferred for diagnosis as the organism is hazardous to handle.
• Aminoglycosides and chloramphenicol are commonly used in the treatment of plague.
• Y. pseudotuberculosis is an animal pathogen that occasionally causes human infection, which may be subclinical or severe.
• Y. enterocolitica causes a usually mild enteritis, but can give rise to a septicaemia, which may be fatal if untreated.
• Pasteurella multocida sometimes infects man, usually through animal bites.
• Pasteurella infection usually responds to penicillin or other antibiotics.
• Francisella tularensis is the cause of tularaemia, a febrile illness that can be severe and life-threatening. It is usually acquired from infected animals.
The organisms within these three genera are animal pathogens that, under certain conditions, are transmissible to man, either directly, or indirectly through food and water or via insect vectors. They are Gram-negative coccobacilli, formerly contained within one genus, Pasteurella. Molecular genetics has indicated a completely separate identity for the three genera, each with its own disease manifestations in man and animals:
1. Yersinia belongs to the Enterobacteriaceae and includes many non-pathogenic environmental species and three closely related pathogenic species.
2. Pasteurella is closely related to the Actinobacillus–Haemophilus group.
Yersinia
Yersinia pestis
Yersinia pestis, the plague bacillus, is essentially a parasite of rodents. In certain parts of the world, burrowing animals such as ground squirrels, gerbils and voles act as reservoirs of infection that may be transmitted by fleas to susceptible animals such as bandicoots, marmots, squirrels and rats. The animals suffer from outbreaks of plague, and their fleas may transmit the infection to man, giving rise to sporadic disease referred to as wild or sylvatic plague. Farmers or trappers who come into contact with infected animals are at risk.
More serious for man is urban plague, resulting from the spread of infection among rats, especially the black rat, Rattus rattus, which used to flourish around human habitation. Outbreaks of human plague, following epidemics in rats, have in the past sometimes developed into pandemics.
Description
Y. pestis is a Gram-negative, non-sporing, non-motile, short coccobacillus. It occurs singly, in pairs or, when in liquid culture, in chains. Pleomorphism is marked, especially in old cultures in which pear-shaped or globular cells, suggestive of yeast cells (involution forms; see p. 20), may be seen. In smears from exudates and in cultures grown at 37°C they are frequently capsulate. In smears from tissues stained by methylene blue or Giemsa stain they show characteristic bipolar staining (‘safety pin’ appearance).
Y. pestis grows aerobically or anaerobically at 0–37°C (optimum 27°C), although small inocula may not grow aerobically in ordinary culture media. Small, slightly viscid, translucent, non-haemolytic colonies develop on blood agar within 24 h. Growth occurs on MacConkey’s medium but tends to autolyse after 2–3 days.
It is killed at 55°C in 5 min and by 0.5% phenol in 15 min. It is sensitive to drying but may remain viable in moist culture for many months, especially at low temperatures.
Pathogenesis
The heat-stable somatic antigen complex of Y. pestis comprises a rough-type lipopolysaccharide (LPS), which has endotoxin activity and is believed to contribute to the terminal toxaemia of plague. The heat-labile Fraction 1 (F1) protein capsular antigen helps the organism to resist phagocytosis and is a protective immunogen. This, and many other proteins associated with pathogenicity, is encoded by three plasmids. The largest, which is very similar to the virulence plasmids of Y. enterocolitica and Y. pseudotuberculosis, contains genes activated at low calcium concentration that express various outer membrane and secreted proteins with a variety of functions, such as inhibition of phagocytosis and intracellular killing. The V antigen, part of the type III secretion system, helps to suppress the innate immune response and facilitate infection and is an important protective antigen. Y. pestis also produces a plasminogen activator and fibrinolysin, which may play a critical role in the initial stages of infection. A pathogenicity island encodes other proteins associated with virulence including cell surface adhesion and iron acquisition factors, some common to Y. pseudotuberculosis and Y. enterocolitica.
Three severe forms of human plague are described: bubonic, pneumonic and septicaemic plague. All may occur at different stages in the same patient. The disease may also present as pharyngitis or meningitis.
Bubonic plague
The transfer of Y. pestis from rats to man through the bites of infected fleas may occasionally result in a localized infection, known as pestis minor, with mild constitutional symptoms. More often the lymph nodes draining the area of the flea bite become affected, and the resulting adenitis produces intensely painful swellings or buboes in the inguinal, axillary or cervical regions, depending on the position of the bite. From these primary buboes the plague bacilli may spread to all parts of the body. Complications such as bronchopneumonia, septicaemia or meningitis may follow. In the absence of adequate antibiotic therapy administered early in the course of the disease, the case fatality rate may exceed 50%.
Pneumonic plague
This can develop in patients presenting with bubonic or septicaemic plague. It may also be acquired as a primary infection by inhalation of droplets infected with Y. pestis, usually from an individual with pneumonic disease or as a result of exposure to aerosols generated from cultures. A severe bronchopneumonia develops. As disease progresses, the sputum becomes thin and blood stained; numerous plague bacilli are demonstrable in stained films or on culture of the sputum. This type of plague is highly contagious and is almost invariably fatal unless treated very early.
Septicaemic plague
This may occur as a primary infection or as a complication of bubonic or pneumonic plague. The bacilli spread rapidly throughout the body and the outcome is almost invariably fatal, even in treated cases. Purpura may develop in the skin (‘Black Death’), and disseminated intravascular coagulation is usually present. It should be noted that bacteraemia can occur in bubonic or pneumonic plague but is usually intermittent in the early stages.
Pointers to the disease include a sudden onset of high fever accompanied by prostration in individuals recently returned from an endemic area, or with a history of occupational exposure.
Laboratory diagnosis
Pneumonic plague is easily acquired in the laboratory by inhalation of aerosols generated from Y. pestis cultures. These and clinical specimens suspected of containing the organism should be handled only under containment conditions appropriate for hazard group 3 pathogens. Animals used for diagnostic tests must be housed under insect-free containment conditions and handled with strict precautions.
Plague is confirmed by demonstrating the bacilli in fluid from buboes or local skin lesions in the case of bubonic plague, in the sputum in pneumonic plague, and in blood films and by blood culture when septicaemic plague is suspected. Blood culture may be intermittently positive in all forms of the disease. Post mortem, the bacilli can usually be isolated from a wide range of tissues, especially spleen, lung and lymph nodes.
Smears of exudate or sputum are stained with methylene blue, Wayson stain (a mixture of basic fuchsin, methylene blue and phenol), Giemsa stain or with an immunospecific stain. Characteristic bipolar-stained coccobacilli are confirmed as Y. pestis by culturing samples on blood agar and incubating at 27°C. If exudate is inoculated subcutaneously into guinea-pigs or white rats, or on to their nasal mucosa, infection follows and the animals die within 2–5 days. The bacilli may then be isolated from the blood or from smears of spleen tissue taken post mortem.
Characteristic colonies growing on blood agar plates are identified presumptively by various cultural and biological tests, by demonstrating chain formation in broth culture, and by ‘stalactite’ growth from drops of oil layered on the surface of fluid medium. Demonstration of the F1 capsular antigen by immunospecific staining confirms the presence of Y. pestis except in the case of rare non-capsulate strains.
Serology is most likely to be useful in the convalescent stage. The complement fixation and a haemagglutination tests formerly used have now been largely superseded by an enzyme-linked immunosorbent assay (ELISA) with F1 antigen.
A polymerase chain reaction (PCR), with primers based on F1 gene sequences, offers a rapid and less hazardous means of diagnosis than culture.
Treatment
Y. pestis is sensitive to many antibiotics, including aminoglycosides, fluoroquinolones, chloramphenicol, co-trimoxazole and tetracyclines, but not penicillin.
When plague is suspected, patients should be isolated and respiratory precautions observed for at least the first 48 h of treatment. Antibiotic therapy should be started without waiting for confirmation of the diagnosis.
Intramuscular streptomycin or intravenous gentamicin is highly effective. Chloramphenicol (given intravenously for the first 4 days) is recommended in patients with meningitic symptoms. Tetracycline may be adequate in uncomplicated bubonic plague if given in large doses within 48 h of onset, and continued for 10 days. Experience with other antibiotics is limited, but there are indications that ciprofloxacin is effective.
Although monotherapy is usually adequate, strains carrying antibiotic resistance plasmids have been reported, and combined therapy may be advisable until the sensitivity of the strain is known.
Plague is a toxigenic infection and antibiotics will not prevent death once the bacteraemia has exceeded a certain threshold. Most patients treated within 18 h of onset can be expected to survive.
Epidemiology
Plague was introduced into Europe from Asia in the thirteenth century and led to the great pandemic known as the Black Death, when about a quarter of the population of Europe succumbed to the disease.
Plague largely disappeared from Europe in the seventeenth century, perhaps because the black rat was displaced by the spread of the brown (sewer) rat, Rattus norvegicus, which is susceptible to plague but does not commonly frequent human dwellings. Improvements in housing may also have played an important part in the elimination of plague from Europe.
The bacilli are transmitted between animals and from animals to man by fleas, notably, but not exclusively, Xenopsylla cheopsis, an ectoparasite of rats. In cool humid weather fleas multiply and plague spreads readily among susceptible rats. In hot dry weather, on the other hand, the fleas die out, limiting the spread of infection. The persistence of endemic plague requires a fine balance between maintenance hosts with a relatively high resistance to lethal infection and transmission to more susceptible hosts such as rats. These conditions occur in a limited number of locations.
When a flea feeds on the blood of a sick animal, plague bacilli are sucked into the insect’s midgut, where they multiply to produce a biofilm that may block the proventriculus, a process promoted by secreted bacterial proteins. When the host animal dies, the flea seeks an alternative host, which may be another rodent or human being. Because the ‘blocked’ flea is unable to suck readily, some of the infected midgut content is regurgitated and injected into the bite wound of the new victim.
When the epizootic among rats has reached a stage at which the number of susceptible animals has greatly decreased through death or immunity, it tends to die out, as does any human epidemic associated with it.
Domestic cats may become infected through contact with rodents. The animals may develop atypical disease and then transmit the infection to their owners or to veterinarians by the percutaneous or respiratory routes.
The sputum of persons suffering from pneumonic plague contains large numbers of plague bacilli, and under favourable conditions the disease spreads rapidly through the community by droplet infection, independently of rodents or fleas. Close contact is required and epidemics are most likely to occur when overcrowding in insanitary accommodation allows the infected droplets to spread readily from person to person. Cool, humid conditions favour transmission.
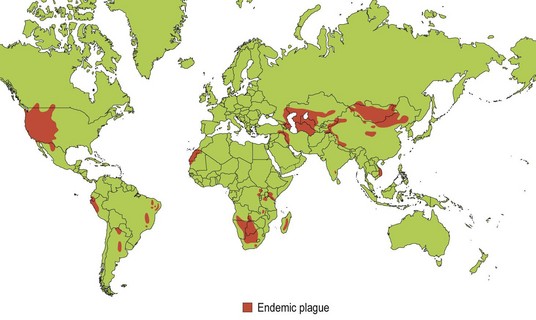
Endemic foci of wild rodent plague persist in many rural parts of the world, including North and South America, Africa and many parts of Asia (Fig. 35.1). Constant surveillance must be maintained to prevent its spread to urban populations, especially in areas where living conditions are below standard.
Y. pestis has been used as a biological warfare agent. Its potential application in bio-terrorism is of major concern.
Control
Bubonic plague
Periodic surveys are advocated in endemic areas to determine the prevalence of rodents and fleas so that control measures can be taken. Rats may be destroyed by rat poison and fleas by the liberal application of insecticide to rat runs.
Other control measures include the construction of rat-proof dwelling houses and buildings such as warehouses in dockland areas. The fumigation of ships and measures to prevent rats gaining access to ships and aircraft help to prevent the spread of plague from one country to another.
Pneumonic plague
Patients suffering from pneumonic plague should be isolated, with full respiratory precautions. Overcrowding of houses and other accommodation should be avoided. Co-trimoxazole, ciprofloxacin or tetracyclines administered to immediate contacts may afford some degree of protection.
Vaccination
Killed vaccines confer significant protection against bubonic but not pneumonic plague, but are no longer widely available. Live vaccines prepared from avirulent strains such as Y. pestis EV76 are used in some countries, but can cause severe reactions. Neither type reliably confer long-term immunity, and revaccination is necessary at 6-month intervals if exposure to infection continues. Candidate vaccines based on recombinant F1 and V antigens or fusion proteins have been submitted for licensing but are not yet commercially available.
Yersinia pseudotuberculosis
Y. pseudotuberculosis can cause disease in many species of wild and domesticated animals and birds. Although presentation can vary widely, it typically causes a fatal septicaemia, often accompanied by formation of small whitish nodules in the viscera (‘pseudotuberculosis’).The infection is indirectly transferable to man, usually through contaminated food or water.
Description
Y. pseudotuberculosis is a small, ovoid, Gram-negative bacillus, with a tendency to bipolar staining. Genetically it is very similar to Y. pestis, which is probably a rough variant that has acquired additional plasmids encoding virulence factors. Initial growth may be best under anaerobic conditions. Isolation can be improved by ‘cold enrichment’ in buffered saline incubated at 4°C with periodic subculture for up to 6 weeks.
The organisms may be differentiated from Y. pestis by:
There are eight major O serotypes, several of which can be separated into subtypes based on thermo-stable LPS somatic antigens. Unlike Y. pestis LPS, these are of smooth type and their specificity is determined by the O chain structure. The core regions are common to all serotypes and to Y. pestis. Thermo-labile flagellar antigens are present in cultures grown at 18–26°C. Many other protein antigens are shared with Y. pestis and Y. enterocolitica.
Pathogenesis
Like other yersiniae, Y. pseudotuberculosis carries a plasmid encoding factors essential for pathogenicity including a type III secretion system. At least one enterotoxin is also produced, as well as invasin and iron-regulated proteins encoded by a chromosomal pathogenicity island.
Clinical disease ranges in severity from subclinical to severe. Gastrointestinal manifestations are common; acute ileitis and mesenteric lymphadenitis are the most characteristic usually accompanied by fever, diarrhoea and pain simulating acute or subacute appendicitis. Infection occasionally results in a severe typhoid-like illness with fever, purpura and enlargement of the liver and spleen, which is usually fatal.
All age groups may be attacked but young males aged 5–15 years seem to be more frequently affected. Recovery is usually uneventful, although immunological sequelae such as erythema nodosum or reactive arthritis develop in some patients.
Laboratory diagnosis
Infection in man is confirmed by isolation of the organism in culture from blood, local lesions or mesenteric nodes, particularly the ileocaecal nodes. PCR has been used experimentally.
Specific serum antibodies are detected and measured by tube or micro-agglutination tests performed during the acute phase of the illness with smooth suspensions of strains of serotypes I–VI grown at 22°C. ELISA or haemagglutination of red cells sensitized with LPS can also be used. The antibodies decline rapidly and reach low levels within 3–5 months.
Treatment
Unlike Y. pestis, Y. pseudotuberculosis is usually sensitive in vitro to penicillins; it is also usually sensitive to aminoglycosides, chloramphenicol, tetracyclines, co-trimoxazole and fluoroquinolones.
Ileitis and mesenteric adenitis are usually self-limiting. Septicaemia demands parenteral treatment with ampicillin, chloramphenicol, gentamicin or tetracycline.
Epidemiology
Many animal species suffer from the infection, but there is little proof of direct transmission to man. Most human infections probably result from the ingestion of contaminated water, vegetables or other food.
About 90% of all human cases in Australia, Europe and North America are attributed to strains of serotype I, followed by serotypes II and III, whereas in Japan serotypes IV and V predominate.
Yersinia enterocolitica
By far the most common manifestation of Y. enterocolitica infection is acute enteritis, which may simulate acute appendicitis. Like many environmental species of Yersinia, it may also occasionally cause opportunist infection in compromised patients, and rarely may present as a plague-like syndrome with fulminant septicaemia.
Description
Morphologically and culturally, Y. enterocolitica resembles Y. pestis and Y. pseudotuberculosis but grows more readily; it differs from them antigenically, biochemically and genetically.
At least 54 different O antigens and 19 H factors have been identified, so that a large number of serotypes are recognized. Serotypes O:3, O:5,27, O:8 and O:9 account for most human infections; other serotypes are probably non-pathogenic in immunocompetent individuals.
Pathogenesis
Y. enterocolitica infects primarily the lymphoid tissue of the small intestine and ileocaecal junction. It carries a low calcium response plasmid and pathogenicity island encoding factors similar to those of Y. pseudotuberculosis. It causes mild and occasionally severe enteritis, mesenteric lymphadenitis and terminal ileitis. Septicaemia, which is often fatal, is most common in the elderly or in patients with predisposing conditions such as cirrhosis, iron overload or immunosuppression. Pneumonia and meningitis are rare presentations. Post-infectious complications include erythema nodosum, polyarthritis, Reiter’s syndrome and thyroiditis. In young children the infection may produce fever, diarrhoea, abdominal pain and vomiting. The symptoms may last for several weeks.
Laboratory diagnosis
The organism is isolated from blood, lymph nodes or other tissues on blood agar or MacConkey’s agar. Isolation from contaminated sources such as faeces is best done by cold enrichment in buffered saline incubated at 4°C for up to 6 weeks, followed by plating on a selective medium. Identity is confirmed by biochemical tests and motility.
The serotype may be determined by slide agglutination with specific rabbit antisera. Serum antibodies are measured by agglutination tests against appropriate O antigens. A significant rise in the titre to 160 or more over a 10-day period indicates acute infection. ELISA may also be used. Cross-reactions occur between serotype O9 and smooth Brucella strains. These are very difficult to differentiate. PCR may be of value but is difficult to apply to highly contaminated materials such as faeces.
Treatment
Y. enterocolitica is sensitive to many antibiotics, including aminoglycosides, chloramphenicol, co-trimoxazole, fluoroquinolones and tetracyclines, but is resistant to penicillin. Sensitivity to other β-lactam antibiotics is variable.
Uncomplicated gastrointestinal infection is usually self-limiting and treatment is indicated only in severe cases. Tetracycline is probably the drug of choice. Invasive infections such as septicaemia require intensive parenteral antibiotic treatment. Limited data are available on the optimum treatment but fluoroquinolones appear effective.
Epidemiology
Y. enterocolitica has been isolated from caseous abscesses resembling those of pseudotuberculosis, from blood and infected wounds, and from the intestinal contents of apparently healthy animals of many species throughout the world. Pigs carry pathogenic serotypes quite frequently, cattle, sheep and goats less so.
Human disease usually results from ingestion of contaminated food or from contact with the environment. Raw pork, milk and drinking water have been implicated as sources. Person-to-person transmission also occurs.
Blood transfusion is a significant hazard as the organism can grow in refrigerated blood from donors with ‘silent’ bacteraemia. Flies are believed to play a role in transmission by contaminating food, and infection has been demonstrated in fleas and lice. However, enteric infection is the usual route of transmission and preventive measures are those appropriate for food-borne disease.
Pasteurella
Pasteurella multocida
Pasteurella multocida (formerly P. septica) is a commensal or opportunist pathogen of many species of domestic and wild animals and birds. Human beings occasionally become infected, especially following animal bites. It is subdivided into three subspecies.
Description
P. multocida organisms are aerobic and facultatively anaerobic coccobacilli, which are appreciably smaller than those of Yersinia species, although they are often pleomorphic in culture. They are Gram-negative, non-motile, non-sporing and capsulate in culture at the optimal growth temperature of 37°C. In smears of blood or tissue stained with methylene blue they show bipolar staining. P. multocida does not grow on MacConkey’s medium.
Five capsular antigens A, B, D, E and F (C is not valid) and at least 11 somatic LPS antigens have been identified. The expression of the capsule is affected by cultural conditions and is lost in rough strains, which also fail to express smooth type O antigens.
The organisms are killed in a few minutes at 55°C and by 0.5% phenol in 15 min. They may survive and remain virulent in dried blood for about 3 weeks, and in culture or infected tissues for many months if kept frozen.
Pathogenesis
P. multocida can be extremely virulent to many species of animals and birds, causing fowl cholera and haemorrhagic septicaemia, which are usually fatal. It also causes respiratory infections and contributes to the pathogenesis of atrophic rhinitis in pigs. Carriage of the organism is usually asymptomatic but stress may provoke fatal systemic infection.
The capsule is essential for full virulence, at least in mice and rabbits, and is the major protective antigen. Iridescent smooth strains show the greatest pathogenicity; mucoid strains are of reduced virulence and rough strains are avirulent. A dermonecrotic protein toxin, a cytotoxin and a neuraminidase probably account for many of the local manifestations of infection, but the bacteria also contain LPS with endotoxin activity.
Human infections usually present as a local abscess at the site of a cat or dog bite, with cellulitis, adenitis and, sometimes, osteomyelitis. P. multocida is also implicated in infections of the respiratory system such as pleurisy, pneumonia, empyema, bronchitis, bronchiectasis and nasal sinusitis.
Rare manifestations of disease include meningitis or cerebral abscess (usually following head injury), endocarditis, pericarditis or septicaemia, and infections of the eye, liver, kidney, intestine and genital tract.
A history of a recent animal bite or of occupational exposure are indicators for suspecting a Pasteurella infection. The organisms may also be carried commensally in the respiratory tract and can cause infection after surgical operation or cranial fracture.
Laboratory diagnosis
Material from bite wounds, blood cultures, cerebrospinal fluid (in cases of meningitis) or respiratory secretions (in suppurative chest infections) are cultured on blood agar. The organisms are identified by various cultural and biochemical tests. Serology is unhelpful. PCR is potentially useful but rarely available.
Treatment
Infections usually respond to penicillin but β-lactamase positive strains occur. Tetracycline, erythromycin or co-trimoxazole are suitable alternatives. In cases of osteomyelitis following dog or cat bites, antibiotic therapy must be continued for at least 8 weeks.
Epidemiology
P. multocida is carried in the nasopharyngeal region of many species of wild and domestic animals. In human infections following animal bites, the organism passes directly to the person in the animal’s saliva. Cat bites are particularly hazardous. Human beings may also become infected through breathing droplets generated by the coughing of animals suffering from respiratory infection. Pig farmers may be particularly at risk.
The disease in farm animals can be prevented by vaccination with preparations derived from killed capsulate bacteria. This is not practicable for human infections because of their rarity.
Other Pasteurella species
Pasteurella spp. other than P. multocida and closely related bacteria formerly classified as Pasteurella, including Avibacterium, Bibersteinia, Mannheimia, Gallibacterium and Phocoenobacter spp., are rarely implicated as human pathogens.
• P. caballi causes respiratory and genital infections in horses and has caused bite wound infections in man.
• P. dagmatis has been associated with bite wounds and endocarditis.
• P. (Mannheimia) haemolytica causes pneumonia and haemorrhagic septicaemia in sheep, buffalo and cattle, and various diseases in poultry and other domesticated animals. It has been isolated from human cases of endocarditis, septicaemia and wound infection. It differs from P. multocida in forming haemolytic colonies on blood agar and by its ability to grow on MacConkey’s medium.
• P. (Actinobacillus) pneumotropica is frequently isolated from the respiratory tract of laboratory animals. It has occasionally been isolated from human cases of septicaemia, upper respiratory tract infections and from animal bite wounds.
Francisella
Francisella tularensis
Francisella tularensis produces tularaemia in man and certain small mammals, notably rabbits, hares, beavers and various rodent species. It occasionally causes large epizootics in lemmings and other small rodents. It can be transmitted by direct contact, by biting flies, mosquitoes and ticks, by contaminated water or meat, or by aerosols.
Description
When first isolated from infected tissue, F. tularensis is a very small, non-motile, non-sporing, capsulate, Gram-negative coccobacillus. In culture larger, pleomorphic, even filamentous, forms are present. It stains poorly with methylene blue but carbol fuchsin (10%) produces characteristic bipolar staining. Four biovars are recognized:
• Type A (formerly F. tularensis tularensis or F. tularensis nearctica) is found only in North America, is often transmitted by ticks and is highly pathogenic.
• Type B (formerly F. tularensis palaearctica or F. tularensis holarctica) occurs in Europe, Asia and North America, is transmitted by mosquitoes and is much less virulent.
F. novicida and F. philomiragia are now regarded as biovars of F.tularensis and have been reported from North America as rare causes of human disease.
F. tularensis is strictly aerobic. It will not grow on ordinary nutrient media, but grows well on blood agar containing 2.5% glucose and 0.1% cysteine hydrochloride. The novicida and philomiragia biovars are less fastidious.
F. tularensis is killed by moist heat at 55°C in 10 min, but may remain viable for many years in cultures maintained at 10°C, and for many days in moist soil and in water polluted by infected animals.
Pathogenesis
Little is known about mechanisms of pathogenicity. A carbohydrate capsule is essential for virulence. A smooth type LPS is also present in the outer membrane, but apparently has low endotoxin activity. Certain outer membrane proteins elicit protective immunity and presumably contribute to pathogenesis.
In animals suffering from tularaemia the bacteria are present in large numbers within the cells of the liver and spleen, including macrophages.
Most human cases are sporadic, although occasional large outbreaks have been reported. After an acute onset with fever, rigors and headache, the disease develops manifestations that vary according to the route of entry of infection.
Following cutaneous inoculation through direct contact with infected animals or a fly or tick bite, a small punched-out skin ulcer develops at the point of entry, accompanied by enlargement of the draining lymph nodes even to the extent of bubo formation (ulceroglandular form). If entry is via the conjunctiva a similar syndrome will develop involving the eye and pre-auricular nodes (oculoglandular form). A glandular form without ulceration also occurs. Inhalation of infected dust or droplets, or ingestion of contaminated meat or water, is more likely to lead to pulmonary or typhoidal disease, respectively. Either can be preceded or accompanied by painful pharyngitis.
Type A strains cause severe and, in the pre-antibiotic era, often fatal disease. Disease caused by type B strains is much less severe and associated with very low mortality rates, but can cause prolonged disability.
Laboratory diagnosis
F. tularensis is extremely dangerous to handle in the laboratory and hazard group 3 containment is required for all manipulations and animal work. All suspect samples should be labelled ‘High risk’.
Human infections are usually diagnosed by inoculating tissue samples or the discharge from local lesions on to glucose–cystine blood agar or cystine heart agar, and identifying any characteristic small mucoid colonies. Alternatively, the exudate may be inoculated into guinea-pigs or mice and the liver and spleen of the infected animals cultured post mortem. PCR methods targeting outer membrane protein genes are now preferred to culture. Ribosomal sequence typing can aid identification.
Serology is most likely to be positive after 3 weeks. Rising F. tularensis antibody titres or individual agglutinin titres of 160 are diagnostic. ELISA with confirmatory western blotting is now replacing agglutination as the preferred method. Serum from cases of brucellosis may cross-react with F. tularensis and vice versa, usually to relatively low titre. Western blotting permits differentiation of cross-reactions.
Treatment
F. tularensis is sensitive to aminoglycosides, chloramphenicol, fluoroquinolones and tetracyclines, but resistant to most β-lactam antibiotics. Streptomycin and gentamicin are the antibiotics of choice in tularaemia and are usually curative. Treatment should be continued for at least 10 days (14 days if ciprofloxacin is used). Tetracyclines or chloramphenicol in high dosage are also effective, but relapse may occur with these bacteriostatic agents unless treatment is prolonged.
Epidemiology
Tularaemia has a worldwide distribution, but occurs mainly in the northern hemisphere. Cases have been reported from North America, from several European countries, including Scandinavia, and from Asia. It has not so far been identified in the UK.
It is a typical zoonosis, spread mainly by insects or ticks among lagomorphs and rodents. It is transmitted to humans through:
• handling of infected animals, such as rabbits or hares
• inhalation of contaminated dust (e.g. during harvesting or mowing)
• ingestion of contaminated water (as a result of pollution with the carcasses or excreta of infected rodents) or meat.
The organism is highly infectious, with a minimum infectious dose of about ten viable bacteria for the most virulent strains. Laboratory workers are especially at risk through handling infected laboratory animals or cultures of the organism. Person-to-person transmission of infection apparently does not occur. F. tularensis has been developed as a biological warfare agent and has potential application in bio-terrorism. A vaccine based on the live-attenuated LVS strain confers some protection. Immunity is dependent on cell mediated responses to outer membrane proteins. Antibodies although diagnostically useful, are not protective.
Abdel-Haq NM, Asmar BI, Abrahamson WM, Brown WJ. Yersinia enterocolitica infection in children. The Pediatric Infectious Disease Journal. 2000;19:954–958.
Agger WA. Tularemia, lawn mowers and rabbits’ nests. Journal of Clinical Microbiology. 2005;43:4304–4305.
Bottone EJ. Yersinia enterocolitica; overview and epidemiologic correlates. Microbes and Infection. 1999;1:323–333.
Butler T. Plague. In: Ledingham JEG, Warrell DA. Concise Oxford Textbook of Medicine. Oxford: Oxford University Press; 2000:1625–1628.
Dennis DT, Campbell GL. Plague and other yersinia infections. In: Kasper DL, Braunwald E, Fauci AS, et al. Harrison’s Principles of Internal Medicine. ed 16. New York: McGraw-Hill; 2005:921–929.
Feodorova VA, Corbel MJ. Prospects for new plague vaccines. Expert Review of Vaccines. 2009;8:1721–1738.
Janda WM, Mutters R. Pasteurella, Mannheimia, Actinobacillus, Eikenella, Kingella, Capnocytophaga and other miscellaneous Gram-negative rods, ed 10. Borriello SP, Murray PR, Funke G, eds. Topley and Wilson’s Microbiology and Microbial Infections, Vol 2. London: Hodder Arnold. 2005:1648–1691.
Schmitt P, Splettstosser W, Porsch-Ozcurumez M, et al. A novel ELISA and a confirmatory western blot useful for diagnosis and epidemiological studies of tularaemia. Epidemiology and Infection. 2005;133:759–766.
Wanger A. Yersinia, ed 6. Borriello SP, Murray PR, Funke G, eds. Topley and Wilson’s Microbiology and Microbial Infections, Vol 2. London: Hodder Arnold. 2005:1458–1473.
Wilson WJ, Erler AM, Nasarabadi SL, et al. A multiplexed PCR-coupled liquid bead array for the simultaneous detection of four biothreat agents. Molecular Cell Probes. 2005;19:137–142.