37 Treponema and borrelia
Syphilis; yaws; relapsing fever; Lyme disease
Key points
• The genus Treponema includes the agents of syphilis (T. pallidum), yaws (T. pertenue), bejel (T. endemicum) and pinta (T. carateum). All are essentially morphologically and antigenically identical spirochaetes, which cannot be cultivated in vitro.
• These diseases, if untreated, characteristically progress to chronic disease through distinct early and late stages and pathologies whose appearance is separated by latent periods of variable length.
• Diagnosis of syphilis, which is primarily sexually transmitted, is made by clinical observation and confirmed serologically.
• All of these organisms are sensitive to benzylpenicillin, which can be used to treat the early stages of disease.
• The genus Borrelia includes agents of Lyme disease (B. burgdorferi sensu lato) and relapsing fevers (B. recurrentis and others), all of which are transmitted to man by ticks or lice.
• Lyme disease, if untreated, may progress through distinct clinical phases (stages 1–3), resulting in later pathology (e.g. arthritis, neurological damage) in some individuals.
• Relapsing fevers are characterized by recurring periods of fever and remission associated with antigenic variation of the borreliae. Infection may be fatal.
• Lyme disease is diagnosed clinically and confirmed serologically. Doxycycline may be used to treat the early stages.
Members of the genera Treponema and Borrelia are spirochaetes. Human diseases caused by these bacteria (Table 37.1) include syphilis, which has been known for thousands of years, and infections such as Lyme disease, the true prevalence and geographical distribution of which are still being evaluated.
Treponemal infections may be spread from person to person by intimate physical contact, by contact with infectious body fluids or, in some instances, by fomites. The treponemes that infect man are obligate human parasites, and no other natural hosts are known. In contrast, borreliae are transmitted to man by infected ticks or lice. The borreliae that cause Lyme disease and endemic relapsing fever also infect many other animal species, which act as reservoirs of infection; man is an unfortunate incidental host in the natural history of these pathogens.
Characteristically, treponemal and borrelial infections occur in several distinct clinical stages. These may be separated by periods of remission, and each stage may have a particular associated pathology. Commonly the causative organism is detectable in early lesions but is much more difficult to identify in later disease. The pathogen spreads from the initial site of infection to many organs via the bloodstream and, despite a vigorous immune response, in some untreated cases these infections may be progressive, destructive and, in some instances (e.g. tertiary syphilis), fatal. In other cases only the early symptoms are apparent and the later pathology is not seen.
Antigenic variation contributes to bacterial virulence for the relapsing fever borreliae but in general the pathogenic mechanisms employed by spirochaetes are poorly understood. No extracellular toxins have yet been identified, and the mechanisms that enable these organisms to persist in tissues despite vigorous immune responses remain unclear. Borrelia burgdorferi and other borreliae can vary their surface lipoproteins to avoid the immune system. The paucity of exposed antigenic proteins on the surface of Treponema pallidum may contribute to immune evasion. It is also likely that the later manifestations of the treponemal and some borrelial infections involve auto-immune phenomena.
In addition to the pathogenic species, many other spirochaetes form part of the normal bacterial flora of the mouth, gut and genital tract. Morphological and antigenic similarities between pathogenic and commensal spirochaetes may cause problems in the clinical and serological diagnosis.
Description
Spirochaetes are slender unicellular helical or spiral rods (Fig. 37.1) with a number of distinctive ultrastructural features used in the differentiation of the genera (Fig. 37.2). The cytoplasm is surrounded by a cytoplasmic membrane, and a peptidoglycan layer contributes to cell rigidity and shape. In Treponema species, fine cytoplasmic filaments are visible in the bacterial cytoplasm (Fig. 37.3), but these are absent in Borrelia species. Members of both genera are actively motile; several flagella are attached at each pole of the cell and wrap around the bacterial cell body. In contrast to other motile bacteria, these flagella do not protrude into the surrounding medium but are enclosed within the bacterial outer membrane. Treponemal flagella are complex, comprising a sheath and core (Fig. 37.4), whereas those of Borrelia species are simpler and similar to the flagella of other bacteria. The spirochaetal outer membrane is unusually lipid rich and, at least in some treponemes, appears to be protein deficient and to lack lipopolysaccharide. This may account for the susceptibility of these organisms to killing by detergents and desiccation.
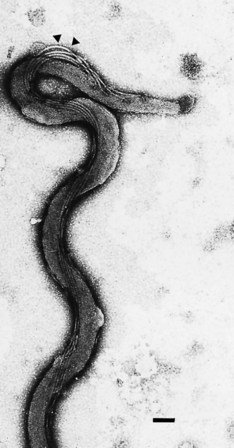
Fig. 37.1 Electron micrograph of T. pallidum. The flagella (arrowheads) are inserted at the tip and follow the helical contour of the bacterial cell enclosed within the outer membrane. Bar = 0.1 µm.
(Photograph courtesy of Professor CW Penn.)
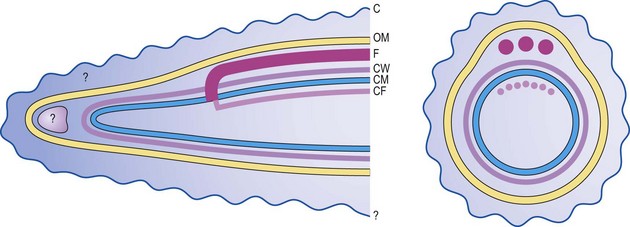
Fig. 37.2 Schematic representation of the structure of T. pallidum in longitudinal and cross-section: C, postulated capsular layer; OM, outer membrane; F, flagellum; CW, cell wall peptidoglycan; CM, cytoplasmic membrane; CF, cytoplasmic filaments (absent in borreliae). Areas of uncertainty (indicated by question marks) include the existence and form of the capsule, the continuity or otherwise of the outer membrane over the tip of the organism, the nature and form of the tip structure, and the exact juxtaposition of the ends of the cytoplasmic filaments with the bacterial flagellar basal bodies.
(After Strugnell R, Cockayne A, Penn CW 1990 Molecular and antigenic analysis of treponemes. Critical Reviews in Microbiology 17: 231–250.)
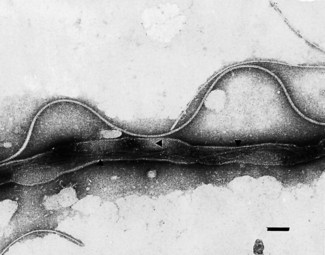
Fig. 37.3 Electron micrograph of a detergent- and protease-treated T. pallidum cell showing cytoplasmic filaments (arrowheads) in the bacterial cytoplasm. Bar = 0.1 µm.
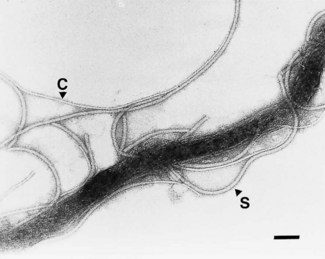
Fig. 37.4 Electron micrograph of a detergent-treated T. pallidum cell showing the complex structure of the treponemal flagellum. Both sheathed (S) flagella and the thinner flagellar cores (C) are visible. Bar = 0.1 µm.
Although the treponemes are distantly related to Gram-negative bacteria, they do not stain by Gram’s method, and modified staining procedures are used. Moreover, the pathogenic treponemes cannot be cultivated in laboratory media and are maintained by subculture in susceptible animals. In contrast, borreliae stain Gram-negative, and many pathogenic species can be cultured in vitro in enriched, serum-containing, media.
Treponema
Treponema species pathogenic for man include the causative agents of venereal syphilis and the non-venereal treponematoses, yaws, bejel and pinta.
The spirochaetes causing these different infections are micro-aerobic and morphologically identical: tightly coiled helical rods, 5–15 µm long and 0.1–0.5 µm diameter. They show only subtle antigenic differences and are characterized primarily by the clinical syndromes they cause and minor differences in the pathology induced in experimental animals.
Treponema pallidum ssp. Pallidum (T. pallidum)
T. pallidum, the causative agent of syphilis, was first isolated from syphilitic lesions in 1905. Infection is usually acquired by sexual contact with infected individuals and is most common in the most sexually active age group of 15–30-year-olds. Congenital syphilis usually occurs following vertical transmission of T. pallidum from the infected mother to the fetus in utero, but neonates may also be infected during passage through the infected birth canal at delivery. Infection in utero may have serious consequences for the fetus. Rarely, syphilis has been acquired by transfusion of infected fresh human blood.
Pathogenesis
Untreated syphilis may be a progressive disease with primary, secondary, latent and tertiary stages. T. pallidum enters tissues by penetration of intact mucosae or through abraded skin.
The bacteria rapidly enter the lymphatics, are widely disseminated via the bloodstream and may lodge in any organ. The exact infectious dose for man is not known, but in experimental animals fewer than ten organisms are sufficient to initiate infection. The bacteria multiply at the initial entry site forming a chancre, a lesion characteristic of primary syphilis, after an average incubation period of 3 weeks. The chancre is painless and most frequently on the external genitalia, but it may occur on the cervix, peri-anal area, in the mouth or anal canal. Chancres usually occur singly, but in immunocompromised individuals, such as those infected with the human immunodeficiency virus (HIV), multiple or persistent chancres may develop. The chancre usually heals spontaneously within 3–6 weeks, and 2–12 weeks later the symptoms of secondary syphilis develop. These are highly variable and widespread but most commonly involve the skin where macular or pustular lesions develop, particularly on the trunk and extremities. The lesions of secondary syphilis are highly infectious.
These lesions gradually resolve and a period of latent infection is entered, in which no clinical manifestations are evident, but serological evidence of infection persists. Relapse of the lesions of secondary syphilis is common, and latent syphilis is classified as early (high likelihood of relapse) or late (recurrence unlikely). Individuals with late latent syphilis are not generally considered infectious, but may still transmit infection to the fetus during pregnancy and their blood may remain infectious.
Late or tertiary syphilis may develop decades after the primary infection. It is a slowly progressive, destructive, inflammatory disease that may affect any organ. The three most common forms are neurosyphilis, cardiovascular syphilis and gummatous syphilis – a rare granulomatous lesion of the skeleton, skin or mucocutaneous tissues. Isolation of T. pallidum from patients with late syphilis is usually impossible, and much of the observed pathology may be due to auto-immune phenomena.
Treponema pallidum ssp. Pertenue (T. pertenue)
T. pertenue is the causative agent of yaws, a disease that is endemic among rural populations in tropical and subtropical countries such as Africa, South America, South-East Asia and Oceania. Eradication programmes sponsored by the World Health Organization reduced the number of cases to fewer than 2 million in the 1970s, but termination of the programmes led to a resurgence of pockets of disease, particularly in West Africa.
Infection with T. pertenue occurs non-venereally following contact of traumatized skin with exudate from early yaws lesions. Infection is usually acquired before puberty. The incubation period is 3–5 weeks, and the initial lesions usually occur on the legs. The papular lesions enlarge, erode and usually heal spontaneously within 6 months. Eruption of similar lesions occurs weeks to months later, and relapse is common. Secondary lesions may involve bones, particularly the fingers, long bones and the jaw. Late yaws is characterized by cutaneous plaques and ulcers, and thickening of the skin on the palms and soles of the feet. Gummatous lesions may also develop. In contrast to syphilis, neurological and cardiovascular damage does not occur. As affected individuals acquire infection early in life, they are essentially non-infectious at childbearing age, and congenital yaws is unknown.
Treponema pallidum ssp. Endemicum (T. endemicum)
This organism causes a non-venereal, syphilis-like disease called endemic syphilis or bejel. Bejel is endemic in Africa, western Asia and Australia, and affects mainly children in rural populations where living conditions and personal hygiene are poor. Transmission is by direct person-to-person contact and by sharing of contaminated eating or drinking utensils.
The initial lesion is usually oral and may not be detected. Secondary lesions include oropharyngeal mucous patches, condyloma lata and periostitis. Late lesions involve gummata in the skin, nasopharynx and bones. As in yaws, the cardiovascular and central nervous systems are not involved, and congenital infection is rare because of the early age of infection.
Treponema carateum
Unlike the other treponematoses, the manifestations of pinta, caused by T. carateum, are confined to the skin. Although these lesions are non-destructive, they cause disfigurement with associated social problems for infected individuals. Pinta is probably the oldest human treponemal infection, with distribution now restricted to arid rural inland regions of Mexico, Central America and Colombia.
Spread of infection is by direct contact with infectious lesions. After a 7–21-day incubation period, small erythematous pruritic primary lesions develop, most commonly on the extremities, face, neck, chest or abdomen. The primary lesions enlarge and coalesce, and once healed may leave areas of hypopigmentation. Disseminated secondary lesions appear 3–12 months later and may become dyschromic. Recurrence of lesions is common for up to 10 years after the initial infection. The depigmented lesions are characteristic of the later stages of pinta but do not cause any serious harm.
Other Treponema species
Treponemes are implicated in several other human infections. The microbial flora associated with these conditions is complex, and the exact role of the spirochaetes in the aetiology of infection remains to be determined. Moreover, the taxonomic status of some of these organisms is uncertain.
Oral infections
T. denticola, T. socranskii and T. pectinovorum form part of the normal flora, and the numbers of these organisms increase in acute necrotizing ulcerative gingivitis and chronic adult periodontal disease. T. vincentii (or Vincent’s spirillum) is similarly associated with ulceromembranous gingivitis or pharyngitis, Vincent’s angina. Several spirochaetes appear to be involved in the aetiology of a similar condition called trench mouth.
Gastrointestinal infections
Several as yet unidentified weakly haemolytic spirochaetes have been implicated in the aetiology of persistent diarrhoea and rectal bleeding in certain human populations. A morphologically similar but genetically distinct organism, Brachyspira (formerly Serpulina) hyodysenteriae, is the cause of swine dysentery.
Skin lesions
Tropical ulcer is a chronic skin condition in which spirochaetes of unknown identity have been implicated, usually in association with fusiform bacteria (see p. 361) and other organisms.
Laboratory diagnosis
The inability to grow most pathogenic treponemes in vitro, coupled with the transitory nature of many of the lesions, makes diagnosis of treponemal infection impossible by routine bacteriological methods. Although spirochaetes are detectable by microscopy in primary and secondary lesions, diagnosis is based primarily on clinical observations and confirmed by serological tests. For practical purposes, the serological responses to all these pathogens are identical, and only their use in the serodiagnosis of syphilis is considered here.
Direct microscopy
Treponemes can be visualized directly in freshly collected exudate from primary or secondary lesions by dark-ground or phase-contrast microscopy. Although this method allows a rapid definitive diagnosis to be made, it is rather insensitive because primary lesions may contain relatively few bacteria. In addition, care must be taken to differentiate between pathogenic and commensal spirochaetes, which may occasionally contaminate such material. More sensitive and specific results may be obtained using fixed material in an immunofluorescence assay with an anti-treponemal antibody.
Serological tests
Infection with T. pallidum results in the rapid production of two types of antibody:
1. Specific antibodies directed primarily at polypeptide antigens of the bacterium
2. Non-specific antibodies (reagin antibodies) that react with a non-treponemal antigen called cardiolipin.
The mechanism of induction of non-specific antibodies remains unclear. Cardiolipin is a phospholipid extracted from beef heart, and it is possible that a similar substance, present in the treponemal cell or released from host cells damaged by the bacterium, may stimulate antibody production.
Historically, assays for non-specific antibody were used as routine screening tests for evidence of syphilis because of their low cost and technical simplicity. Enzyme immunosorbent assays that detect specific treponemal antibodies are increasingly replacing the older tests for screening purposes.
Non-specific serological tests for syphilis
The rapid plasma reagin (RPR) test, which has now largely superseded the earlier Venereal Disease Research Laboratory (VDRL) test, is a non-specific serological test for syphilis that uses cardiolipin as antigen. Immunoglobulin (Ig) M or IgG antibody present in positive sera causes a suspension of this lipoidal antigen to flocculate, and the result can be read rapidly by eye. Both of these assays may be used as screening tests, and are positive in approximately 70% of primary and 99% of secondary syphilitics, but are negative in individuals with late syphilis. These tests can be used quantitatively, and increases in antibody titres with time may be used to confirm a diagnosis of congenital syphilis. The RPR assay is not suitable for use with cerebrospinal fluid.
As a positive result in these tests usually indicates active infection, they can also be used to monitor the efficacy of antibacterial therapy.
Tests for specific antibody
Fluorescent treponemal antibody absorption (FTA-Abs) test
This is an indirect immunofluorescence assay in which T. pallidum is used as an antigen. Acetone-fixed treponemes are incubated with heat-treated sera, and bound antibody is detected with a fluorescein-labelled conjugate and ultraviolet microscopy. The serum is first absorbed with a suspension of a non-pathogenic treponeme, which removes non-specific cross-reactive antibodies that may be directed against commensal spirochaetes. The FTA-Abs test is positive in approximately 80, 100 and 95% of primary, secondary and late syphilitics, respectively, and, unlike the RPR and VDRL tests, remains positive following successful therapy.
T. pallidum haemagglutination assay (TPHA)
In this test, T. pallidum antigen is coated on to the surface of red blood cells, and specific antibody in test sera causes haemagglutination. As in the FTA-Abs assay, sera are pre-absorbed with a non-pathogenic treponeme to remove antibody against commensal spirochaetes. The TPHA test is less sensitive than the FTA-Abs test in primary syphilis (positive in 65%), but both give similar results for secondary and late syphilis; the TPHA also remains positive for life following infection. This assay can be used to detect localized production of anti-treponemal antibodies in cerebrospinal fluid, a marker of neurosyphilis. The T. pallidum particle agglutination (TPPA) test works on the same principle as the TPHA, but treponemal antigen is coated on to coloured gelatin particles rather than red blood cells.
Enzyme immunoassay
In these tests monoclonal anti-T. pallidum antibodies are used to detect antibody responses to individual treponemal antigens. This allows rapid screening of large numbers of samples with potentially enhanced specificity. Assays that detect either IgM or IgG are available. Positive results should be confirmed by a second specific test such as the TPHA.
Problems in the serological diagnosis of syphilis
Occasionally, both the non-specific and specific tests produce false-positive results. The RPR and VDRL assays may give a transient positive result following any strong immunological stimulus such as acute bacterial or viral infection or after immunization. More persistent false-positive results occur in individuals with autoimmune or connective tissue disease, in drug abusers and in individuals with hypergammaglobulinaemia. False-positive results usually become apparent when negative results are found in specific serological tests, but in some cases FTA-Abs results may also be positive or borderline.
Rarely, the FTA-Abs test may be positive and the nonspecific VDRL test negative. Lyme disease (see below) induces antibodies that react in the FTA-Abs but not in the VDRL assay. Other spirochaetal diseases such as relapsing fever, yaws, pinta and leptospirosis may give positive results in both specific and non-specific tests. Of particular difficulty is the differential diagnosis of syphilis and yaws in immigrants from areas in which yaws is endemic.
Some of the newer enzyme immunoassays are less sensitive in cases of primary syphilis.
Direct detection of spirochaetal deoxyribonucleic acid (DNA) in clinical material by molecular methods, such as the polymerase chain reaction (PCR), may have a future role in confirming a diagnosis of syphilis in difficult or atypical cases.
Treatment
All the pathogenic treponemes are sensitive to benzylpenicillin, and prolonged high-dose therapy with procaine penicillin has been the traditional method of treatment for primary and secondary syphilis. So far there have been no reports of penicillin resistance. If penicillin allergy is a problem, erythromycin, tetracycline or chloramphenicol may be used. There are reports of treatment failure with erythromycin, and an erythromycin-resistant variant of T. pallidum has been isolated. In late syphilis, aqueous benzylpenicillin is used, as this penetrates better into the central nervous system. In neurosyphilis, successful eradication of the organism may not result in a clinical cure. More aggressive and prolonged antibiotic therapy may be required in HIV-positive patients with syphilis owing to impaired immune function.
Antibiotic therapy of syphilitics, particularly with penicillin, characteristically induces a systemic response called the Jarisch–Herxheimer reaction. This is characterized by the rapid onset (within 2 h) of fever, chills, myalgia, tachycardia, hyperventilation, vasodilatation and hypotension. The response is thought to be due to release of an endogenous pyrogen from the spirochaetes.
Epidemiology and control
Syphilis
The widespread introduction of antibiotic therapy shortly after the Second World War produced a dramatic decrease in the incidence of syphilis, but the disease remained endemic within the general population. Geographically localized outbreaks of syphilis, associated with specific recreational activities and lifestyles, now occur in countries such as the UK.
In the mid-1980s most cases of syphilis in developed countries occurred in male homosexuals. The advent of HIV and the acquired immune deficiency syndrome (AIDS) in the 1980s reduced the incidence among this group owing to changes in sexual practices. The early 1990s saw a resurgence of syphilis among the heterosexual population in the USA, resulting in an increased incidence among women and in the number of cases of congenital syphilis. Subsequent changes in sexual practices among homosexual men have reversed this trend: significantly more cases of primary and secondary syphilis now occur in men than women and, among males, over 60% of cases occur in men who have sex with men. The incidence of congenital syphilis has fallen from 2435 cases in 1994 to 427 in 2009. Total reported numbers of cases of syphilis in the USA were 46 291 in 2008 and 44 828 in 2009.
In the UK ongoing outbreaks of syphilis in various regions have seen the number of cases rise consistently from 2000, peaking at around 3700 cases of infectious syphilis in 2007. Most cases of infectious syphilis (>70%) currently occur among men who have sex with men but significant numbers of cases are still detected in heterosexual men and women. Syphilis therefore continues to pose major public health issues. The potential for congenital infection and the acquisition of syphilis by blood transfusion mean that screening programmes of all pregnant women and blood donations are still required.
Control of syphilis is achieved by treating index cases and any known contacts. Treatment of contacts is important as some may be incubating the infection even if they have no overt signs of disease.
Control of the disease may have additional benefits: primary syphilis increases the risk of HIV infection two- to five-fold, presumably by permitting easier access of the virus through damaged skin or mucosal membranes.
Borrelia
The two principal human diseases associated with borreliae are relapsing fever, caused by Borrelia recurrentis and several other Borrelia species, and Lyme disease or Lyme borreliosis, a multi-system infection caused by B. burgdorferi sensu lato. The bacteria causing these infections are morphologically similar helical rods, 8–30 µm long and 0.2–0.5 µm in diameter, with three to ten loose spirals. Antigenic and genetic differences are used to differentiate the species.
Relapsing fevers
Relapsing fevers are characterized clinically by recurrent periods of fever and spirochaetaemia.
Endemic or tick-borne relapsing fever is a zoonosis caused by several Borrelia species, including B. duttoni, B. hermsii, B. parkeri and B. turicatae, and is transmitted to man by soft-bodied Ornithodoros ticks. The natural hosts for these organisms include rodents and other small mammals on which the ticks normally feed. The disease occurs worldwide, reflecting the distribution of the tick vector.
Epidemic or louse-borne relapsing fever is caused by B. recurrentis, an obligate human pathogen transmitted from person to person by the body louse, Pediculus humanus. The incidence is influenced by socio-economic factors such as lack of personal hygiene, and, historically, increases during periods of war, famine and other social upheaval. The disease still occurs in central and eastern Africa and in the South American Andes.
The spirochaetes causing the two forms of relapsing fever differ in their mode of growth in the arthropod vector, and this influences the way in which human infection is initiated. B. recurrentis grows in the haemolymph of the louse but does not invade tissues. As a result the louse faeces are not infectious and the bacterium is not transferred through eggs to the progeny. Human infection occurs when bacteria released from crushed lice gain entry to tissues through damaged or intact skin, or mucous membranes. Spirochaetes causing tickborne relapsing fever invade all the tissues of the tick, including the salivary glands, genitalia and excretory system. Infection occurs when saliva or excrement is released during feeding. Transovarial transmission to the tick progeny maintains the spirochaete in the tick population.
Pathogenesis
In both forms of relapsing fever, acute symptoms, including high fever, rigors, headache, myalgia, arthralgia, photophobia and cough, develop about 1 week after infection. A skin rash may occur, and there is central nervous system involvement in up to 30% of cases. During the acute phase there may be up to 105 spirochaetes per cubic millimetre of blood. The primary illness resolves within 3–6 days, and terminates abruptly with hypotension and shock, which may be fatal. Relapse of fever occurs 7–10 days later, and several relapses may take place.
Each episode of spirochaetaemia is terminated by the development of specific anti-spirochaete antibody. Subsequent febrile episodes are caused by borreliae that differ antigenically, particularly in outer membrane protein composition, from those causing earlier attacks. As the cycle of fever and relapse continues, the borreliae tend to revert back to the antigenic types that caused the original spirochaetaemia, and ultimate clearance of the infection appears to be due to antibody-mediated killing.
In general, louse-borne relapsing fever has longer febrile and afebrile periods than tick-borne infection, but fewer relapses. The case fatality rate varies from 4–40% for louse-borne infection and from 2–5% for tick-borne relapsing fever, with myocarditis, cerebral haemorrhage and liver failure the most common causes of death.
Laboratory diagnosis
Definitive diagnosis of relapsing fevers is made by detection of borreliae in peripheral blood samples. Thick or thin blood smears are stained with Giemsa or other stains such as acridine orange.
Although antibodies to the borreliae are produced during infection, serological tests are complicated by antigenic variation and the tendency to relapse. Serological tests for syphilis are positive in 5–10% of cases.
Treatment
Tetracycline, chloramphenicol, penicillin and erythromycin have been used successfully. As in the treatment of syphilis, antibiotics may elicit a Jarisch–Herxheimer reaction.
Prevention of infection involves avoidance or eradication of the insect vector. Insecticides can be used to eradicate ticks from human dwellings, but elimination from the environment is not feasible. Prevention of louse-borne infection involves maintenance of good personal hygiene, and delousing if necessary.
Lyme disease
Lyme disease, originally called Lyme arthritis, was recognized as an infectious condition in 1975 following an epidemiological investigation of a cluster of cases of suspected juvenile rheumatoid arthritis that occurred in Lyme, Connecticut, USA. A common factor in these cases was a previous history of insect bite, and the infectious agent, B. burgdorferi, was subsequently isolated from an Ixodes tick. Retrospective serological data suggest that Lyme disease was endemic in the USA as early as 1962, and the clinical manifestations of this infection have been known in Europe, including the UK, since the early 1900s. Lyme disease has also been reported in Scandinavia, eastern Europe, China, Japan and Australia.
The natural hosts for B. burgdorferi are wild and domesticated animals, including mice and other rodents, deer, sheep, cattle, horses and dogs. The larger animal hosts such as deer are probably more important in maintaining the size of tick populations rather than acting as a major source of B. burgdorferi. Infection in these animals may be inapparent, although clinical infection has been observed in cattle, horses and dogs.
B. burgdorferi is transmitted to man by ixodid ticks that become infected while feeding on infected animals. The principal vectors in the USA are Ixodes dammini and I. pacificus, and in Europe, I. ricinus. The life cycle of these ticks involves larval, nymph and adult stages, all of which are capable of transmitting infection, although the nymphal stage is most commonly implicated. In areas endemic for Lyme disease, 2–50% of ticks may carry B. burgdorferi. The bacterium grows primarily in the midgut of the tick, and transmission to man occurs during regurgitation of the gut contents during the blood meal. Transmission efficiency appears to be relatively low, but increases with the duration of feeding.
Although there is general similarity, clinical manifestations may differ in the USA and Europe. This variation is due in part to significant differences in the bacterial strains causing infection in the two continents, and has resulted in the division of B. burgdorferi into three distinct genospecies:
• B. burgdorferi sensu stricto is the sole cause of Lyme disease in the USA where Lyme arthritis is a common complication of infection.
• B. afzeli and B. garinii are responsible for most Lyme disease in Europe, and are associated with chronic skin and neurological symptoms, respectively.
Several other genetically distinct isolates of B. burgdorferi have been identified in ticks, but their importance in human infection has yet to be established.
Lyme disease may be a progressive illness, and is divided into three stages:
• Stage 1 is characterized by a spreading annular rash, erythema chronicum migrans (ECM), which occurs at the site of the tick bite 3–22 days after infection. Lesions may contain very small numbers of bacteria, and the disproportionate intensity of the pathology seen may be due to stimulation of cytokines such as tumour necrosis factor-α and secondary mediators. The bacterium also spreads to various other organs. In the USA, secondary lesions similar to those of ECM are common. Malaise, fatigue, headache, rigors and neck stiffness may also be apparent. The rash and secondary lesions fade within 3–4 weeks.
• Stage 2 develops in some patients after several weeks or months. These patients exhibit cardiac or neurological abnormalities, musculoskeletal symptoms or intermittent arthritis.
• Stage 3 may ensue months to years later, when patients present with chronic skin, nervous system or joint abnormalities.
Congenital infection may occur with serious, potentially fatal, consequences for the fetus.
Laboratory diagnosis
Once a clinical diagnosis has been made, culture of the spirochaete from suitable biopsy material provides a definitive diagnosis, but this is a lengthy, specialized technique that is not widely available. As the organism is also difficult to detect in histological sections, serological tests are used routinely for the confirmation of Lyme disease, although PCR techniques are used in some laboratories.
Specific IgM antibodies develop within 3–6 weeks of infection. The earliest response appears to be against the bacterial flagellum and later against outer surface proteins. Subsequently, IgG antibodies are produced, and the highest titre is detectable months or years after infection.
An indirect immunofluorescence test is available, but enzyme-linked immunosorbent assay (ELISA) is now widely used. Immunoblotting with a panel of carefully selected recombinant antigens is used to confirm serological results. Serological diagnosis of early Lyme disease may still pose problems, as antibodies to the bacterium are slow to develop in some individuals and the formation of immune complexes may affect the test results. Antibodies that cross-react with B. burgdorferi may be produced after infection with other spirochaetes, and sera from patients with Lyme disease may give a positive FTA-Abs test, although the VDRL test is negative.
Serological evidence of infection may be detectable in the apparent absence of overt disease. The significance of these findings is unclear but it is possible that such individuals may develop late complications of Lyme disease.
Treatment
Penicillins, macrolides, cephalosporins and tetracyclines have all been used successfully. Reports suggest that treatment with tetracyclines (doxycycline) produces fewer late complications than penicillin therapy. About 15% of patients experience a Jarisch–Herxheimer reaction after antibiotic therapy. Despite antibiotic treatment, some patients suffer from minor late complications of the disease, which may be mediated immunologically and may require supplementary immunosuppresive therapy, or may indicate low-level persistence of the organisms.
Antibiotic therapy may reduce or abolish the antibody response, and this may interfere with the serological confirmation of infection.
Epidemiology and control
The geographical distribution of Lyme disease is governed by that of the tick vector and its associated animal hosts. Forestry workers and farmers are particularly at risk, but infection is also increasingly associated with recreational activities. In the UK, Lyme disease occurs in areas that support large populations of wild or domesticated animals on which ixodid ticks feed. Infection may also be acquired after travel to countries where Lyme disease is endemic. It is difficult to accurately assess the true incidence of Lyme disease, because infection may be mild or asymptomatic and consequently not detected. In 2008, there were 813 laboratory-confirmed cases in the UK but estimated case numbers range from 1000–3000 per year. By comparison, approximately 30 000 confirmed cases were reported in the USA in 2009.
Prevention of infection involves avoidance of endemic areas and education of the public regarding the possible risks of infection in these localities. Eradication of the tick vectors or mammalian hosts from such areas is not feasible. A vaccine to protect residents and visitors in areas in which Lyme disease is endemic would be useful, but attempts to develop effective and safe recombinant vaccines against B. burgdorferi have so far been unsuccessful.
Andersson LC, Bergstrom S. Current issues in relapsing fever. Current Opinion in Infectious Disease. 2009;22:443–449.
Antal GM, Lukehart SA, Meheus AZ. The endemic treponematoses. Microbes and Infection. 2002;4:83–94.
Cullen PA, Haake DA, Adler B. Outer membrane proteins of pathogenic spirochetes. FEMS Microbiology Reviews. 2004;28:291–318.
Edwards AM, Dymock D, Jenkinson HF. From tooth to hoof: treponemes in tissue-destructive diseases. Journal of Applied Microbiology. 2003;94:767–780.
Egglestone SI, Turner AJ. Serological diagnosis of syphilis: PHLS Syphilis Serology Working Group. Communicable Disease and Public Health. 2000;3:158–162.
Herremans T, Kortbeek L, Notermans DW. A review of diagnostic tests for congenital syphilis in newborns. European Journal of Clinical Microbiology and Infectious Disease. 2010;29:495–501.
Lee V, Kinghorn G. Syphilis: an update. Clinical Medicine. 2008;8:330–333.
Marques AR. Lyme disease: a review. Current Allergy and Asthma Reports. 2010;10:13–20.
Centers for Disease Control and Prevention. Syphilis. http://www.cdc.gov/std/syphilis/.
Health Protection Agency. Diagnosis and treatment of Lyme borreliosis. http://www.hpa.org.uk/web/HPAweb&HPAwebStandard/HPAweb_C/1204031510081.
