39 Chlamydia
Genital and ocular infections; infertility; atypical pneumonia
Key points
• Chlamydiae are obligate intracellular bacterial pathogens with a unique growth cycle.
• C. trachomatis is the most common bacterial sexually transmitted infection, and the leading infectious cause of blindness.
• C. pneumoniae is an important cause of community acquired pneumonia.
• Chlamydial infections are frequently asymptomatic.
• Serious sequelae of chlamydial infection (blindness, pelvic inflammatory disease, infertility) are caused by immune response-driven scarring and fibrosis.
• Diagnosis requires laboratory tests, preferably nucleic acid amplification tests.
• Treatment is with doxycycline, erythromycin or azithromycin.
• It is important to treat sexual partners of patients with chlamydial genital tract infection.
• Prevention depends on interrupting the transmission chain: there are no effective vaccines.
Chlamydiae are obligate intracellular bacterial pathogens of eukaryotic cells with a characteristic dimorphic growth cycle quite distinct from that of other bacteria, involving alternation between a metabolically inert, infectious, spore-like elementary body, which can survive in the extracellular environment, and a metabolically active, replicating reticulate body, which cannot. They are widely distributed in nature and are responsible for a variety of human infections affecting the eye, and the genitourinary and respiratory tracts.
Chlamydiae were first described in 1907 by Halberstaedter and von Prowazek, who observed cytoplasmic inclusions in conjunctival scrapings taken from children with trachoma and from monkeys inoculated with ocular material from these children. They named them Chlamydozoa, from the Greek words χλαµuς (cloak) and ζooν (animal), because of the way in which the inclusions were draped around the nucleus. Similar inclusions were soon observed in conjunctival scrapings taken from neonates with conjunctivitis, and from the cervix of their mothers. Most human infections are caused by Chlamydia trachomatis, which was first grown, in mouse brain and subsequently in eggs, from a patient with lymphogranuloma venereum (LGV) in the 1930s. The more fastidious trachoma biovar was not isolated until 1957. It was first grown in tissue culture in 1965, making it possible for the first time to study the epidemiology and clinical features of C. trachomatis infection on a large scale.
Description
Classification
The Chlamydia genus (order Chlamydiales; family Chlamydiaceae) comprises nine species of which two are primarily human pathogens: C. trachomatis, causing ocular and genital infections; and C. pneumoniae, causing mainly respiratory disease. The other species infect animals: C. psittaci (chiefly birds); C. abortus (sheep); C. felis (cats); C. pecorum (cattle); C. suis (pigs); C. muridarum (mice) and C. caviae (guinea pigs). C. psittaci, C. abortus and C. felis are occasionally transmitted to man. A taxonomic reclassification, based on ribosomal DNA sequence data, assigned C. pneumoniae, C. psittaci and C. abortus to a new genus, Chlamydophila. However, this new taxonomy has not been universally accepted and is not used here.
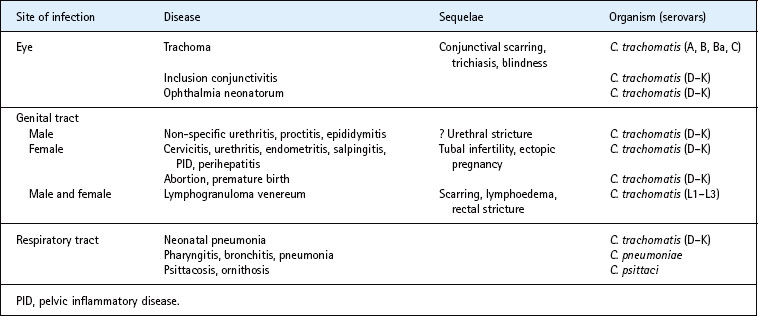
The species C. trachomatis contains two biovars: the more invasive LGV biovar (serovars L1–L3) replicates in macrophages, invades lymph nodes and causes a systemic infection; the more common trachoma biovar is largely confined to squamo-columnar epithelial cells of the eye (serovars A–C) and genital tract (serovars D–K) (Table 39.1). Serovars are defined by the presence of specific epitopes on the major outer membrane protein. Serovars A–C differ from serovars D–K in that they are unable to synthesize tryptophan, owing to disruption of the trpA gene.
Chlamydiae have one of the smallest bacterial genomes, containing around 1 million base pairs. Virtually all strains of C. trachomatis also contain a 4.4-MDa plasmid of unknown function. Genomes of several C. trachomatis serovars have been sequenced, and show a high level of conservation of gene order and content (>99%). A high degree of genetic conservation is also seen across Chlamydia species, with C. trachomatis and C. muridarum, for example, being >95% identical. The fact that chlamydiae replicate within an intracellular vacuole probably explains the high degree of conservation, since it does not allow them to exchange genetic material with other bacteria.
Biology
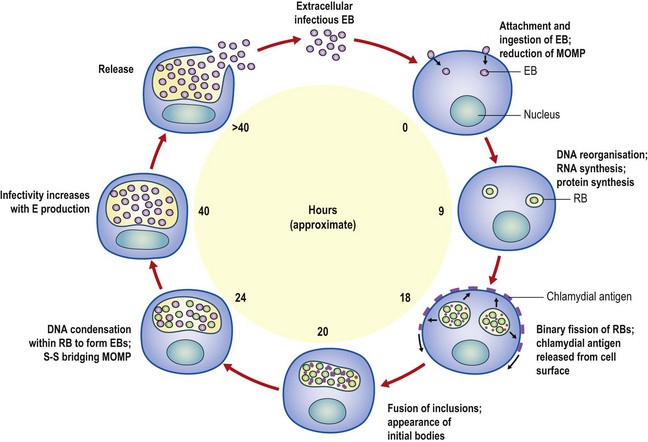
Chlamydiae probably evolved from host-independent, Gram-negative ancestors. They are ‘energy parasites’ relying on the host cell for synthesis of ATP. The chlamydial envelope possesses bacteria-like inner and outer membranes. The infectious elementary body is electron dense, DNA rich and approximately 300 nm in diameter. The cell wall does not contain peptidoglycan, and its rigidity is maintained by extensive disulphide linking of the major outer membrane protein, which makes up some 60% of the outer membrane. The elementary body binds to the host cell and enters by ‘parasite-specified’ endocytosis. Fusion of the chlamydia-containing endocytic vesicle with lysosomes is inhibited and the elementary body begins its unique developmental cycle within the eukaryotic cell. The major outer membrane protein is reduced to a monomeric form and acts as a porin, allowing nutrients to enter the organism from the host cell. After about 8 h the elementary body differentiates into the larger (800–1000 nm), non-infectious, metabolically active reticulate body, which divides by binary fission. By 20 h post-infection, a proportion of reticulate bodies has begun to reorganize into a new generation of elementary bodies (Fig. 39.1). These reach maturity up to 30 h after entry into the cell and rapidly accumulate within the endocytic vacuole, which may contain more than 1000 organisms. They are released by lysis of the host cell 30–48 h after the start of the cycle.
Pathogenesis
After an incubation period of 5–10 days, C. trachomatis elicits an acute inflammatory response with a purulent exudate. A period of chronic inflammation ensues, with the development of sub-epithelial follicles, and this leads eventually, in some cases, to fibrosis and scarring. This scarring process is responsible for much of the morbidity associated with C. trachomatis, in both the genital tract and the eye. It is particularly likely to be seen after repeated infections.
Study of virulence determinants of C. trachomatis is difficult, since it has not so far proved possible to manipulate chlamydiae genetically. However, the availability of the complete genome of several C. trachomatis strains has provided some insights. The serovar D genome contains genes homologous with those coding for virulence factors in other bacteria, including a cytotoxin gene, and genes encoding a type III secretion pathway (see p. 17). A conserved chlamydial protease, proteasome-like activity factor, is secreted into the host cell cytoplasm, where it interferes with the assembly and surface expression of HLA (human leucocyte antigen) molecules and inhibits apoptosis. In a non-human primate model genetic variations in six C. trachomatis genes that appear to be associated with increased virulence have been identified.
The epidemiology of C. trachomatis infection suggests that a degree of protective immunity follows natural infection. The prevalence and bacterial load of ocular infection is lower in adults than in children in trachoma endemic communities, and the duration of infection is shorter. Similarly, genital C. trachomatis infection is most prevalent in the youngest sexually active age groups, and the chlamydial isolation rate for men with non-gonococcal urethritis is lower in those who have had previous episodes. Killed whole organism vaccines provide some degree of protection against ocular C. trachomatis infection in man and non-human primates. Serovar-specific monoclonal antibodies to the major outer membrane protein neutralize C. trachomatis in vitro, but there are few data to suggest that either IgG or IgA antibody is protective. The intracellular development of C. trachomatis is inhibited by interferon-γ, and evidence from animal models and studies of human ocular infection suggest that cell-mediated immune responses, mediated by CD4+ lymphocytes are important for the clearance of infection.
Vaccine studies in primates suggest that vaccination could provoke more severe disease on subsequent challenge, implying that much of the damage caused by C. trachomatis infection may be immunopathological in origin. This would be in keeping with the histopathology of C. trachomatis infection, in which the lymphoid follicle is the hallmark. Follicles contain typical germinal centres, consisting predominantly of B lymphocytes, with T cells, mostly CD8+, in the parafollicular region. The inflammatory infiltrate between follicles comprises plasma cells, dendritic cells, macrophages, and polymorphonuclear leucocytes, with T and B lymphocytes. Fibrosis is seen at a late stage, typically in trachoma and pelvic inflammatory disease. T lymphocytes are also present and outnumber B cells and macrophages.
A chlamydial heat-shock protein (hsp 60), homologous with the GroEL protein of Escherichia coli, elicits antibody responses that are associated with the damaging sequelae of C. trachomatis infections in both the eye and genital tract. In-vitro interferon-γ interferes with the chlamydial development cycle, leading to persistent infection with continuing release of hsp 60. It is not known whether the immune response to hsp 60 is itself the cause of immunopathological damage, or merely a marker of more severe or prolonged infection. Studies of gene expression at the site of ocular infection have shown the importance of innate immune pathways and NK (natural killer) cell activation, and suggest that matrix metalloproteinases 7 and 9 play an important role in the scarring process. Polymorphisms in immune response genes encoding tumour necrosis factor-α, interferon-γ and interleukin-10 are associated with the development of severe scarring following ocular C. trachomatis infection.
Clinical features
Chlamydia trachomatis
Genital infection
The clinical manifestations of genital C. trachomatis infection are similar to those of gonorrhoea, but are usually less severe, as C. trachomatis infection elicits a less intense acute inflammatory response than Neisseria gonorrhoeae. Many chlamydial infections are asymptomatic. Long term sequelae such as infertility and ectopic pregnancy are generally caused by fibrosis and scarring of the fallopian tubes following prolonged or repeated infections, and may develop even in those with few or no symptoms.
Infection in men
C. trachomatis is detectable in the urethra of up to 50% of men with symptomatic non-gonococcal urethritis. The incubation period is 7–21 days, compared to 2–5 days for gonorrhoea. Patients present with a history of dysuria, usually accompanied by a mild to moderate mucopurulent urethral discharge. C. trachomatis is responsible for a proportion of cases of chronic (persistent or recurrent) non-gonococcal urethritis. Since mixed infections are common, treatment of gonococcal urethritis with an antibiotic ineffective against C. trachomatis may result in post-gonococcal urethritis.
C. trachomatis is responsible for up to 70% of cases of acute epididymitis in young men (35 years of age or less) in developed countries. Patients present with unilateral scrotal pain, swelling and tenderness, often accompanied by fever. Most give a history of current or recent urethral discharge. In older patients, epididymitis and epididymo-orchitis tend to be caused by urinary-tract pathogens. There is no good evidence that chlamydial infection leads to male infertility or to acute or chronic prostatitis.
Both LGV and non-LGV strains of C. trachomatis can cause proctitis in those who practise receptive anal intercourse. Non-LGV strains cause a milder disease, which may be asymptomatic or give rise to rectal pain, bleeding and muco-purulent anal discharge.
Infection in women
C. trachomatis typically infects the columnar epithelial cells of the endocervix. It does not affect the squamous epithelium of the vagina. Infection is associated with a mucopurulent discharge from the cervix visible on speculum examination, and with hypertrophic cervical ectopy that tends to bleed on contact. Most infected women have no symptoms. The prevalence of cervical infection is no higher among women who complain of vaginal discharge than among those who do not, suggesting that it is not a cause of symptomatic vaginal discharge.
C. trachomatis has been implicated as a cause of the urethral syndrome, characterized by dysuria, frequency and sterile pyuria. Clinical signs of urethritis, such as urethral discharge or meatal redness, are not usually found.
Infection may spread from the endocervix to the endometrium and fallopian tubes, causing pelvic inflammatory disease. This is more likely to occur after trauma to the cervix due, for example, to termination of pregnancy, insertion of an intra-uterine contraceptive device, or delivery. Histologic evidence of endometritis can be found in up to 50% of women with mucopurulent cervicitis due to C. trachomatis, and is more common in those with a history of abnormal vaginal bleeding. Classic signs of pelvic inflammatory disease may be present (fever, lower abdominal pain and tenderness, and cervical motion tenderness), but chlamydial pelvic inflammatory disease may be subclinical. Spread to the peritoneum may result in perihepatitis (the Curtis–Fitz-Hugh syndrome), which may be confused with acute cholecystitis in young women. C. trachomatis infection has also been associated with post-partum endometritis.
C. trachomatis is the major cause of pelvic inflammatory disease in developed countries. Infertility may be the first indication of asymptomatic tubal disease. It occurs in about 10% of women following a single upper genital tract infection and in up to 50% after two or three episodes. Infertility may result from endometritis, from blocked or damaged fallopian tubes, or from abnormalities of ovum transportation caused by damage to the ciliated epithelial surface. Other consequences of salpingitis are chronic pelvic pain and ectopic pregnancy. Following chlamydial pelvic inflammatory disease, the risk of ectopic pregnancy increases 7–10-fold.
Some studies have shown C. trachomatis infection to be associated with low birth weight and pre-term delivery, but others have failed to confirm this. In general, infection was diagnosed and treated at a later stage of gestation in those studies which found a correlation between infection and adverse birth outcome than in those that did not.
Infection has been weakly associated with bartholinitis and should be considered in the absence of other known pathogens. A significant association between cervical chlamydial infection and cervical squamous cell carcinoma, but not adenocarcinoma, has been established, and it has been suggested that chlamydial infection may enhance the effect of oncogenic papillomaviruses.
Adult paratrachoma (inclusion conjunctivitis) and otitis media
Adult chlamydial ophthalmia commonly results from the accidental transfer of infected genital discharge to the eye. It usually presents as a unilateral follicular conjunctivitis, acute or subacute in onset. The features are swollen lids, mucopurulent discharge, papillary hyperplasia and later, follicular hypertrophy, and occasionally punctate keratitis. About one-third of patients have otitis media, and complain of blocked ears and hearing loss. The disease is generally benign and self-limiting. Patients and their sexual contacts should be investigated for genital chlamydial infection and managed appropriately.
Reactive arthritis
Arthritis occurring with or soon after non-gonococcal urethritis is termed ‘sexually acquired reactive arthritis’. Conjunctivitis and other features characteristic of Reiter’s syndrome are seen in about one-third of patients. C. trachomatis has also been associated with ‘seronegative’ arthritis in women. Viable chlamydiae have not been detected in the joints of patients with this condition, which is probably the result of immunopathology. Despite this, early tetracycline therapy has been advocated by some investigators.
Neonatal infections
Conjunctivitis appears in 20–50% of infants exposed to C. trachomatis infecting the cervix at birth. A mucopurulent discharge and occasionally pseudomembrane formation occur 1–3 weeks later. It usually resolves without visual impairment.
About half of the infants who have conjunctivitis also develop pneumonia, although a history of recent conjunctivitis and bulging eardrums are found in only half of the cases. Chlamydial pneumonia usually begins between the fourth and eleventh week of life, preceded by upper respiratory symptoms. There is tachypnoea, a prominent, staccato cough but usually no fever, and the illness is protracted. Radiographs show hyperinflation of the lungs with bilateral diffuse, symmetrical, interstitial infiltration and scattered areas of atelectasis. Children infected during infancy are at increased risk of obstructive lung disease and asthma.
Lymphogranuloma venereum
The clinical course of LGV can be divided into three stages. The primary stage at the site of inoculation; the secondary stage in the regional lymph nodes, and/or the anorectum; and the tertiary stage of late sequelae affecting the genitalia and/or rectum.
Primary stage
After an incubation period of 3–30 days, a small, painless papule, which may ulcerate, occurs at the site of inoculation. The primary lesion is self-limiting and may pass unnoticed by the patient. Among patients with LGV presenting with buboes in Thailand, more than half had not been aware of an ulcer.
Secondary stage
This occurs some weeks after the primary lesion. It may involve the inguinal lymph nodes, or the anus and rectum. The inguinal form is more common in men than women, since the lymphatic drainage of the upper vagina and cervix is to the retro-peritoneal rather than the inguinal lymph nodes. LGV proctitis occurs in those who practise receptive anal intercourse, probably due to direct inoculation.
The cardinal feature of the inguinal form of LGV is painful, usually unilateral, inguinal and/or femoral lymphadenopathy (bubo). Enlarged lymph nodes are usually firm and often accompanied by fever, chills, arthralgia and headache. Biopsy reveals small discrete areas of necrosis surrounded by proliferating epithelioid and endothelial cells, which may enlarge to form stellate abscesses that may coalesce and break down to form discharging sinuses. In women, signs include a hypertrophic suppurative cervicitis, backache and adnexal tenderness.
Clinical features of anorectal disease include a purulent anal discharge, pain and bleeding due to an acute haemorrhagic proctitis or proctocolitis, often with fever, chills and weight loss. Proctoscopy reveals a granular or ulcerative proctitis. Computed tomography or magnetic resonance imaging scans may show pronounced thickening of the rectal wall, with enlargement of iliac lymph nodes. Enlarged inguinal nodes may also be palpable.
Cervical adenopathy due to LGV has been reported after oral sex. A follicular conjunctivitis has also been described following direct inoculation of the eye, which may be accompanied by pre-auricular lymphadenopathy. Other rare manifestations of the secondary stage include acute meningoencephalitis, synovitis and cardiac involvement.
Tertiary stage
This appears after a latent period of several years, but is rare. Chronic untreated LGV leads to fibrosis, which may cause lymphatic obstruction and elephantiasis of the genitalia in either sex, or rectal strictures and fistulae. Rarely, it can give rise to the syndrome of esthiomene (Greek: ‘eating away’) with widespread destruction of the external genitalia.
Trachoma
The clinical signs of trachoma are best seen in the conjunctival surface of the everted upper eyelid. Active or inflammatory trachoma, which is usually seen in children in endemic communities, is a follicular kerato-conjunctivitis. Subjects in whom five or more follicles of >0.5 mm diameter are seen in the central subtarsal conjunctiva are defined by the World Health organization (WHO) as having follicular trachoma. In some cases the inflammation is severe enough to obscure the conjunctival blood vessels. If more than half the blood vessels are obscured, this is defined as intense inflammatory trachoma. Blood vessels may be seen growing into the cornea, usually at its superior margin; this is known as pannus. C. trachomatis can be detected in a proportion of cases of follicular trachoma, but not in all cases, since the follicles can persist for weeks or months after the infection has resolved. Repeated episodes of inflammatory trachoma lead eventually to conjunctival scarring. As the scars contract they cause the lid margin to turn inwards (entropion), and the lashes to abrade the cornea (trichiasis). This causes extreme discomfort, damages the cornea, and leads eventually to blindness due to corneal opacity.
C. pneumoniae
C. pneumoniae causes pneumonia, pharyngitis, bronchitis, otitis and sinusitis with an incubation period of about 21 days. It may be a significant cause of acute exacerbations of asthma, and is one of the most common causes of community-acquired pneumonia, but is seldom identified as the causal agent because laboratory tests for its diagnosis are not widely used. It is a chronic, often insidious, respiratory pathogen to which there appears to be little immunity. Sero-epidemiological studies indicate that some 60–80% of people worldwide become infected with C. pneumoniae during their life.
C. psittaci
C. psittaci is an important cause of infections in a wide range of birds and is shed in nasal secretions and droppings. Nasal secretions contaminate the feathers, where they dry and produce a highly infectious dust in which the organism can survive for months. This may give rise to severe pneumonia in man, called ornithosis or psittacosis depending on the bird species from which the infection was derived. The agricultural economy is also affected, as large outbreaks of ornithosis have been reported in turkeys, geese and ducks. There are import controls in many countries to restrict the movement of birds, which are rendered more infectious by travel-induced stress.
The incubation period is about 10 days, and the illness ranges from an ‘influenza-like’ syndrome, with general malaise, fever, anorexia, rigors, sore throat, headache and photophobia, to a severe illness with delirium and pneumonia. The illness may resemble bronchopneumonia, but the bronchioles are involved as a secondary event and sputum is scanty. The organism disseminates through the body, and there may be meningoencephalitis, arthritis, pericarditis or myocarditis, or a predominantly typhoidal state with enlarged liver and spleen. Endocarditis has been described.
Laboratory diagnosis
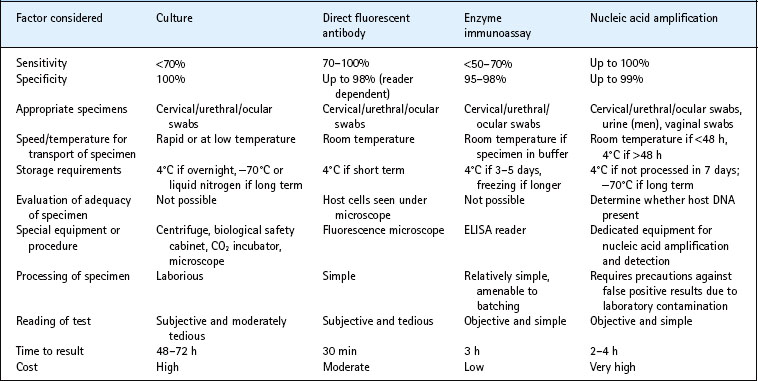
The laboratory diagnosis of chlamydial infection depends on detection of the organisms or their antigens or nucleic acid and, to a much lesser extent, on serology (Table 39.2). In urogenital infection the highest bacterial load of C. trachomatis is found in the endocervix in women and in the urethra in men. An endocervical swab is therefore needed for the diagnosis of infection by culture or antigen detection assay. However, the greater sensitivity of nucleic acid amplification tests for C. trachomatis means that self-administered vaginal swabs and ‘first-catch’ urine specimens give equivalent results to endocervical swabs when using these assays. Samples to be tested can be transported to the laboratory at room temperature, making home-based screening for C. trachomatis possible.
Culture
Centrifugation of specimens onto cycloheximide-treated McCoy or HeLa cell monolayers, followed by incubation and then staining with a fluorescent monoclonal antibody or with a vital dye, to detect inclusions, has been widely used for the diagnosis of C. trachomatis infection. One blind passage may increase sensitivity. However, cell-culture techniques are no more than 70% sensitive compared to nucleic acid amplification tests and are slow and labour intensive. Because culture is essentially 100% specific, it still has a role in medico-legal cases. C. pneumoniae is even more difficult to grow than C. trachomatis. C. psittaci is a hazard group 3 pathogen and few laboratories attempt to grow it.
Direct immunofluorescence
Microscopic detection of elementary bodies with species-specific fluorescent monoclonal antibodies is rapid and, for C. trachomatis oculogenital infections, highly sensitive and specific in the hands of skilled observers. However, the test is laborious and interpretation is subjective. It is best used in settings where few specimens are tested, or for confirming positive results obtained with other tests.
Nucleic acid amplification tests
By enabling amplification of a nucleic acid sequence specific to the chlamydial species, the polymerase chain reaction assay, the strand displacement assay and the transcription mediated amplification technique have overcome problems of poor sensitivity. Commercial assays for C. trachomatis based on each of these three amplification methods are available and widely used. The first two assays amplify nucleotide sequences of the cryptic plasmid, which is present in multiple copies in each chlamydial elementary body. However, a rare variant of C. trachomatis has been described which lacks the plasmid, giving rise to false negative results with these assays. The transcription mediated amplification reaction is directed against rRNA, which is also present in multiple copies. These sensitive assays have replaced culture as the ‘gold standard’ for the diagnosis of C. trachomatis infection. Nucleic acid amplification tests for C. pneumoniae and C. psittaci are not commercially available.
Enzyme immunoassays
Enzyme immunoassays that detect chlamydial antigens, usually the genus specific lipopolysaccharide, have largely been replaced by the more sensitive nucleic acid amplification test.
Point of care tests
Over 20 rapid strip tests based on the immunochromatographic detection of chlamydial lipopolysaccharide are commercially available. They can give a result within 15–20 min of sample collection, but most lack sensitivity compared to nucleic acid amplification methods.
Serological tests
Serological tests are of no value in uncomplicated genital C. trachomatis infection. In pelvic inflammatory disease, in LGV and in the Curtis–Fitz-Hugh syndrome, serology may be useful if a rising titre can be demonstrated. C. trachomatis IgM antibody is the ‘gold standard’ for the diagnosis of chlamydial pneumonia in babies. Pneumonia due to C. pneumoniae and C. psittaci is usually diagnosed serologically, but depends on the demonstration of IgM antibodies or an IgG titre >512 by microimmunofluorescence, or a rise in antibody titre in a convalescent sample. Immunofluorescence and enzyme immuno-assays are commercially available, but have not been rigorously evaluated.
Treatment
Chlamydiae are intracellular and hence insensitive to aminoglycosides and other antibiotics that do not penetrate cells efficiently. Tetracyclines and macrolides are the mainstay of treatment. Treatment is often started before a microbiological diagnosis can be established, so additional broad-spectrum antibiotics are needed to cover gonococcal and, in the case of pelvic inflammatory disease, anaerobic infections. Treatment of sexual partners is essential to prevent reinfection.
Uncomplicated C. trachomatis infections are treated with a single dose of azithromycin 1 g, or with doxycycline 100 mg twice daily for 7 days. Chlamydial pelvic inflammatory disease is treated with a 14-day course of doxycycline 100 mg twice daily. Clinically significant resistance to these antibiotics has not been reported. Doxycycline is contra-indicated in pregnancy. Azithromycin 1 g as a single dose, and amoxicillin 500 mg three times daily for 7 days, are safe and effective in pregnant women. Ofloxacin is active against C. trachomatis at a dose of 300 mg twice daily for 7 days, but is not widely used. Ophthalmia neonatorum and neonatal pneumonia due to C. trachomatis should be treated with erythromycin syrup by mouth, 50 mg/kg daily divided into four doses, for 14 days.
There has been no adequate study comparing antibiotic regimens for LGV, C. pneumoniae or C. psittaci infection. Recommended treatment for LGV is doxycycline 100 mg twice daily, or erythromycin 500 mg four times daily, for 21 days. Azithromycin has been used successfully in some cases, although a 1 g single dose is unlikely to be sufficient. Large collections of pus should be aspirated, using a lateral approach through normal skin. Macrolides or tetracyclines are recommended for the treatment of infection with C. pneumoniae and C. psittaci. Prolonged courses may be required in patients with pneumonia.
Ocular infection can be effectively treated with a single oral dose of azithromycin (20 mg/kg, maximum 1 g) but, in trachoma endemic communities, reinfection rapidly occurs and mass treatment of entire communities is therefore recommended.
Epidemiology
C. trachomatis is the most common bacterial sexually transmitted infection, and the most common infectious cause of blindness. Genital infection is common in all sexually active populations, and prevalence is usually highest in the young. In the UK, the number of reported chlamydial infections trebled between 1996 and 2005. Similar increases were seen in other Western countries over this period, including Sweden and Canada, despite active screening programmes. It is not clear to what extent this is due to an increased incidence, or to an increase in the number of people tested with the sensitive nucleic acid amplification tests that have been widely used since the late 1990s. The overall incidence of reported chlamydial infection in the UK in 2005 was 223 per 100 000 total population, with the highest rate (1300 per 100 000) in women aged 16–19. The WHO has estimated that, in 2005, there were 101 million new cases of genital chlamydial infection.
LGV is rare in industrialized countries, but is endemic in parts of Africa, Asia, South America and the Caribbean. Its epidemiology is poorly defined, because LGV is often indistinguishable clinically from chancroid and other causes of genital ulceration with bubo formation, and it has been difficult to obtain laboratory confirmation. Among patients presenting with buboes to a sexually transmitted disease clinic in Bangkok 10% were found to have LGV, and an epidemic of LGV has been reported among crack cocaine users in the Bahamas. In 2003, an outbreak of LGV proctitis due to the L2 serovar was reported among homosexual men in the Netherlands, and since then over one thousand cases have been reported in homosexual men in Europe and North America; most affected men were HIV-positive.
Trachoma, caused by C. trachomatis transmitted from eye to eye, disappeared from Europe and North America in the 20th century as living standards improved, but remains endemic in poor rural populations in Africa and Asia. WHO estimates that at least 40 million people have trachoma, and that 8 million are blind or visually impaired as a result.
Molecular epidemiology
Typing isolates of C. trachomatis is potentially of great value. It could help to map sexual networks, and to distinguish between treatment failure and reinfection in clinical trials. If associations could be found between particular strains and particular clinical findings, it could help to identify virulence determinants of C. trachomatis and increase understanding of the pathogenesis of infection.
The first typing method for C. trachomatis, the micro-immunofluoresence test, was based on the ability of monoclonal antibodies to distinguish 13 (later increased to 17) serotypes of C. trachomatis. More recently genital and ocular strains of C. trachomatis have been genotyped following amplification of the ompA gene which encodes the major outer membrane protein, either by sequencing, or by restriction fragment length polymorphism analysis of the amplified product; but ompA genotyping is not sufficiently discriminatory to distinguish between persistent infection and reinfection with a common genotype.
A multi-locus sequence typing method, targeting six variable genes identified through genome sequencing projects, has been used to investigate a variant of C. trachomatis lacking the plasmid detected by commonly used nucleic acid amplification tests.
Control and prevention
Health education and condom promotion, especially for the youngest sexually active age groups, may help to reduce the incidence of genital C. trachomatis infection. Syndromic management of symptomatic infections, and partner notification, may also play a role, but since a high proportion of chlamydial infections is asymptomatic in both sexes, these measures are unlikely to be successful on their own. A screening programme for C. trachomatis at primary health care level in the USA has been shown to reduce the incidence of upper genital tract infection and its complications in women. Screening programmes have been introduced in some European countries, in which young people presenting to health services for any reason are offered a test for C. trachomatis; but the public health impact of such opportunistic screening programmes remains to be demonstrated. Where reinfection rates are high retesting of positive cases 6 months after treatment has been recommended.
No vaccine is presently available. Recent research has focused on the development of a subunit vaccine against C. trachomatis, which provides protection without eliciting immunopathology. Purified preparations of major outer membrane protein were protective in murine models, provided the native trimeric structure of the protein was maintained. In non-human primates, a similar preparation of major outer membrane protein reduced peak shedding from the ocular surface, but had no effect on the duration of infection or on ocular disease.
The strategy for trachoma control recommended by WHO is based on the acronym SAFE: Surgery for trichiasis; mass treatment with Antibiotics; Facial cleanliness; and Environmental improvement to reduce the transmission of C. trachomatis from eye to eye.
Brunham RC, Rey-Ladino J. Immunology of chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nature Reviews Immunology. 2005;5:149–161.
Kuo CC, Jackson LA, Campbell LA, Grayston JT. Chlamydia pneumoniae (TWAR). Clin Microbiol Rev. 1995;8:451–461.
Low N, Bender N, Nartey L, et al. Effectiveness of chlamydia screening: systematic review. Int J Epidemiol. 2009;38:435–448.
Mabey D, Solomon A, Foster A. Trachoma. Lancet. 2003;362:223–229.
Stephens RS, Kalman S, Lammel C, et al. Genome sequence of an obligate intracellular pathogen of humans: Chlamydia trachomatis. Science. 1998;282:754–759.
Van der Bij AK, Spaargaren J, Morré SA, et al. Diagnostic and clinical implications of anorectal lymphogranuloma venereum in men who have sex with men: a retrospective case-control study. Clinical Infectious Diseases. 2006;42:186–194.