40 Rickettsia, orientia, ehrlichia, anaplasma and coxiella
Typhus; spotted fevers; scrub typhus; ehrlichioses; Q fever
Key points
• Rickettsiae are small obligately intracellular bacteria that are associated with insects or ticks during at least part of their transmission cycle.
• Rickettsia and Orientia cause spotted fevers, typhus fevers and scrub typhus by infecting and damaging endothelial cells, resulting in increased vascular permeability, oedema, adult respiratory distress syndrome, meningoencephalitis and rash.
• Ehrlichia and Anaplasma reside in persistently infected vertebrate hosts such as deer, dogs or rodents, and are transmitted by feeding ticks.
• Ehrlichia and Anaplasma target human monocytes or granulocytes, where they grow to microcolonies in cytoplasmic vacuoles.
• The frequent absence of antibodies to rickettsiae, orientiae and ehrlichiae early in the course of illness hinders laboratory diagnosis, making clinico-epidemiologic suspicion of the diagnosis and empirical treatment, preferably with doxycycline, essential.
• Coxiella burnetii thrives in the acidic phagolysosome of macrophages, has a stable extracellular form and infects human beings who inhale aerosols from birth fluids or the placenta of infected animals.
Few diseases have had a greater impact on the course of human history than epidemic typhus. Hans Zinsser’s classic book Rats, Lice and History provides a graphic account of how Rickettsia prowazekii, the aetiological agent of this louse-borne disease, has caused millions of deaths and much human suffering in conditions of famine, poverty and war. Epidemic typhus now occurs mainly in poor populations in developing countries as world conditions have improved, but various other rickettsial diseases are still widely distributed.
The rickettsiae (Rickettsia and Orientia species) and anaplasmas (Anaplasma, Ehrlichia and Neorickettsia species) of medical importance are obligately intracellular bacteria mostly transmitted by arthropod vectors. Molecular studies reveal that Rickettsia species, Orientia tsutsugamushi, Ehrlichia species and Anaplasma species evolved from a common ancestor.
Coxiella burnetii, an intracellular bacterium found in ticks, is more closely related to Legionella pneumophila, but is conveniently considered here alongside the rickettsia group.
Rickettsia and Orientia
Description
The genera Rickettsia and Orientia include organisms responsible for numerous diseases in many parts of the world (Table 40.1). The pioneering research of Ricketts and others in the early twentieth century demonstrated the rickettsial aetiology of Rocky Mountain spotted fever and epidemic louse-borne typhus. Several other diseases, including murine typhus and Mediterranean spotted fever, were later shown to be rickettsial infections. Previously unrecognized spotted fevers caused by R. japonica, R. africae and R. honei were discovered in the 1980s and 1990s in Japan, Africa and Australia, indicating that much remains to be learned about these organisms. Other rickettsiae, poorly understood and presumed to be non-pathogenic, have been isolated, primarily from arthropods, including herbivorous insects.
Rickettsiae are small (0.3–0.5 × 0.8–1.0 µm) Gram-negative bacilli. They reside in the cytosol of host cells (Fig. 40.1). All pathogens are associated with a flea, louse, mite or tick vector. Species pathogenic for man parasitize endothelial cells almost exclusively. Rickettsiae have a small genome (approximately 1 Mb) and lack genes encoding many essential enzymes. Thus they depend on the host for nutrition and building blocks, and are yet to be cultivated outside eukaryotic cells. Like the chlamydiae (see Ch. 39), rickettsiae have a typical Gram-negative bacterial cell wall, including a bilayered outer membrane that contains lipopolysaccharide, and are energy parasites that transport adenosine triphosphate (ATP) with a unique translocase.
Fig. 40.1 Electron micrograph of a cell infected with R. rickettsii, showing the dilatation of the endoplasmic reticulum of host cells that occurs as a result of injury associated with infection by spotted fever group rickettsiae.
The genus Rickettsia is divided into two antigenically distinct groups based on their lipopolysaccharide: the typhus group and the spotted fever group. The immunodominant rickettsial outer membrane protein A (OmpA) exists only in the spotted fever group rickettsiae. Another major outer membrane protein, OmpB, exists in all Rickettsia species. OmpA and OmpB both contain cross-reactive and species-specific epitopes.
The scrub typhus rickettsiae are antigenically distinct and appear to be fundamentally different. They have been classified into a related but distinct genus as Orientia tsutsugamushi (see Table 40.1). The cell wall lacks lipopolysaccharide, peptidoglycan and a slime layer, and appears to derive its structural integrity from proteins linked by disulphide bonds. O. tsutsugamushi exhibits multiple major antigenic proteins with both strain-specific and cross-reactive epitopes.
Pathogenesis
Invasion and destruction of target cells
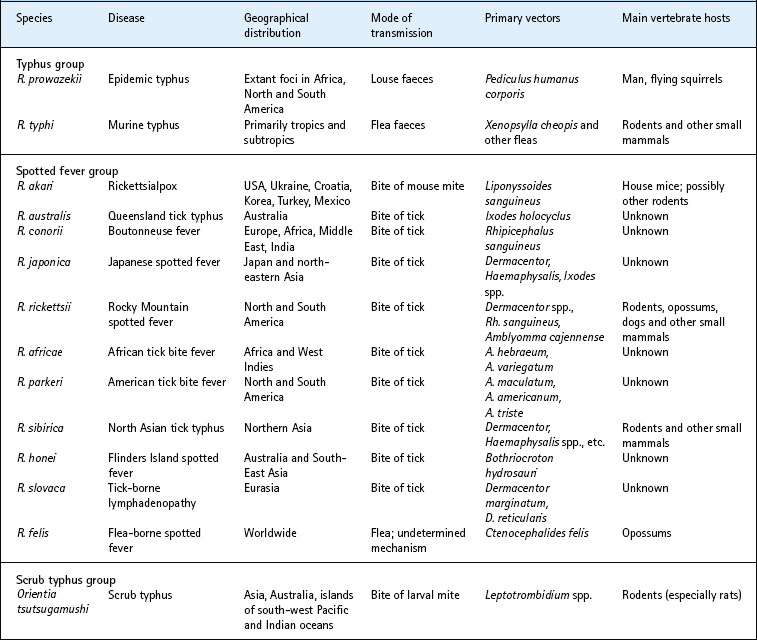
Rickettsiae normally enter the body through the bite or faeces of an infected arthropod vector. They are disseminated through the bloodstream, attach to endothelial cells by OmpA, OmpB and other autotransporter surface proteins, enter endothelial cells by induced phagocytosis, escape from the phagosome, multiply intracellularly and eventually destroy their host cells. Cell culture studies suggest that spotted fever and typhus group rickettsiae destroy the host cell by different mechanisms. After infection with R. prowazekii or R. typhi, the rickettsiae continue to multiply until the cell is packed with organisms (Fig. 40.2) and then bursts, possibly as a result of membranolytic activity; before lysis, host cells appear ultrastructurally normal.
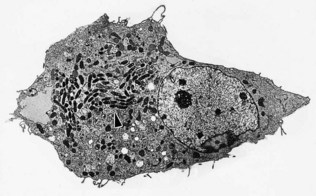
Fig. 40.2 Electron micrograph of a cell infected with R. prowazekii. The rickettsiae continue to multiply within the cell until it is completely packed with organisms and bursts. In contrast to cells infected with spotted fever group rickettsiae, the ultrastructural appearance of cells infected with typhus group rickettsiae remains normal until the cell lyses. The region of the cell containing rickettsiae is indicated by the arrowhead.
Spotted fever group rickettsiae seldom accumulate in large numbers and do not burst the host cells, but stimulate polymerization of F-actin tails which propel them through the cytoplasm and into filopodia, from which they escape the cell or spread into an adjacent cell (Fig. 40.3). Two proteins, RickA and Sca2, act as critical regulators of actin-based movement in spotted fever group, but not in typhus rickettsiae. Infected cells exhibit signs of membrane damage associated with an influx of water, which is sequestered within cisternae of dilated rough endoplasmic reticulum (see Fig. 40.1). Rickettsiae damage host cell membranes at least in part by stimulating production of free oxygen radicals by endothelial cells.
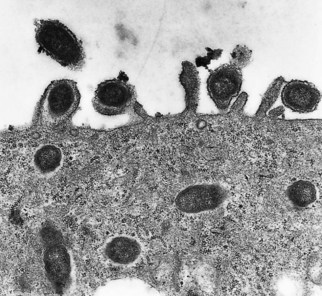
Fig. 40.3 Electron micrograph of R. conorii escaping from a host cell. Note the location of the rickettsiae within host cell filopodia.
Scrub typhus rickettsiae also escape from the phagosome, reside free in the cytosol, and are released from host cells soon after infecting them, but little is known about the mechanism(s) by which these organisms damage cells.
Pathological lesions
All members of the genera Rickettsia and Orientia cause widespread microvascular injury leading to the destruction and dysfunction of infected endothelial cells. The pathological manifestations result more from direct rickettsial injury than from immunopathological mechanisms mediated via cytokines and cytotoxic T lymphocytes, inflammation, intravascular coagulation or endotoxin. Interference with normal circulation and increased vascular permeability following damage of blood vessels can cause life-threatening encephalitis and non-cardiogenic pulmonary oedema.
Clinical aspects of rickettsial diseases
Epidemic typhus
Headache and fever develop 6–15 days after infection with R. prowazekii. A macular rash, often noted 4–7 days after the patient becomes ill, first appears on the trunk and axillary folds and then spreads to the extremities. In mild cases the rash may begin to fade after 1–2 days, but in more severe cases it may last much longer and become haemorrhagic. Severe cases may also develop pronounced hypotension and renal dysfunction. The mental state of the patient may progress from dullness to stupor and even coma. Although the prognosis is grave for comatose patients, prompt appropriate treatment may be life-saving.
Individuals who survive a primary infection of louse-borne typhus may develop a relatively milder reactivation of latent infection many years later. This is referred to as recrudescent typhus or Brill–Zinsser disease. Such individuals are nevertheless immune to a second louse-borne infection.
Flea-borne fevers
Patients infected with R. typhi develop symptoms similar to those of epidemic typhus. Fatal cases are uncommon but occasionally occur, particularly in the elderly. Although murine typhus is much milder than epidemic typhus, it is still severe enough to require several months of convalescence. R. felis is maintained transovarially in cat fleas and causes a similar illness.
Tick-borne spotted fever
There are many clinical similarities among the tick-borne rickettsioses of the spotted fever group. The most severe is Rocky Mountain spotted fever, for which the average annual case-fatality rate for 1981–1998 was 3.3%. Risk factors for fatal infection include older age, glucose-6-phosphate dehydrogenase deficiency and delayed tetracycline treatment.
Patients become ill within 2 weeks of infection. Early symptoms include fever, severe headache and myalgia, often accompanied by anorexia, vomiting, abdominal pain, diarrhoea, photophobia and cough. An eschar (‘tache noire’) frequently occurs at the site of the tick bite in all spotted fever group infections except Rocky Mountain spotted fever. A maculopapular rash usually develops within 3–5 days. The rash of spotted fever usually develops first on the extremities rather than on the trunk. Absence of a rash does not exclude rickettsial infection, because a disproportionate number of fatal cases of Rocky Mountain spotted fever are of the ‘spotless’ variety. Spotted fever group rickettsiae are found within the endothelial cells and less often macrophages of vertebrate hosts, but R. rickettsii can also invade vascular smooth muscle.
Vascular damage in severe cases may result in haemorrhagic rash, hypovolaemia, hypotensive shock, non-cardiogenic pulmonary oedema and impairment of central nervous system function. A fulminant form of Rocky Mountain spotted fever sometimes kills the patient within 5 days of the onset of symptoms; this form of the disease is more common in black males who are deficient in glucose-6-phosphate dehydrogenase, and may be related to haemolysis in these patients. Infection confers long-lasting immunity.
Rickettsialpox
The clinical course of rickettsialpox is similar to that of other spotted fever group infections and includes development of fever, headache and an eschar at the site where the infected mite fed. The rash is initially maculopapular but often becomes vesicular. Fever lasts for about a week, and patients usually recover uneventfully.
Scrub typhus
Scrub typhus may be mild or fatal, depending on host factors and presumably the virulence of the infecting strain. Symptoms develop 6–18 days after being fed upon by infected mite larvae (chiggers). The disease is characterized by sudden onset of fever, headache, and myalgia. A maculopapular rash often develops 2–3 days after onset of the illness. An eschar is often apparent at the site of the bite with enlargement of local lymph nodes. Progression of the disease may be accompanied by interstitial pneumonitis, generalized lymphadenopathy and splenomegaly. Death may result from encephalitis, respiratory failure and circulatory failure. Patients who survive generally become afebrile after 2–3 weeks, or sooner if treated appropriately. Scrub typhus confers only transient immunity, and re-infection may occur with heterologous or homologous strains.
Laboratory diagnosis
Timely and accurate diagnosis of rickettsial disease followed by administration of an appropriate antibiotic may mean the difference between death of the patient and uneventful recovery. The lack of widely available, reliable diagnostic tests that can detect the disease in its early stages remains a problem, particularly as symptoms are often non-specific. The rash may appear at a late stage in the infection or not at all, and may resemble exanthemata of many other diseases. The presence or significance of an eschar, if present, is also commonly overlooked.
Serological methods
Rickettsial diseases are usually acute and short-lasting. Antibodies appear in the second week of illness, when the patient is usually on the way to recovery. Death may occur before detectable levels of antibody are present. Serology is therefore not suitable for early diagnosis of rickettsial infections and is used mainly to confirm the diagnosis for epidemiologic investigations.
The traditional Weil–Felix test, which relies on agglutination of the somatic antigens of non-motile Proteus species, is no longer recommended because of unacceptably poor sensitivity and specificity. More reliable diagnostic tests including immunofluorescence and enzyme immunoassay are now commercially available.
Isolation of rickettsiae
Isolation of the organism provides conclusive proof of rickettsial infection. However, it is seldom attempted because of lack of facilities or expertise and because of the presumed danger to laboratory personnel. Such dangers have been overemphasized in this era of antibiotics, but use of containment facilities is appropriate.
Rickettsiae can be isolated in cell culture, in laboratory animals such as mice or guinea-pigs, or in embryonated chicken eggs. Cell culture yields timely results and is widely used for isolation of rickettsiae from clinical samples. Rickettsiae can be detected in cell culture 48–72 h after inoculation by the shell vial assay.
Detection of rickettsiae in tissue
Skin biopsies from the centre of petechial lesions can be examined by immunohistochemistry. This approach is virtually 100% specific and has a sensitivity of 70%. Rickettsiae can be visualized for up to 48 h after the administration of appropriate drugs, allowing diagnosis of infection post mortem. A method has been developed for capturing detached, circulating endothelial cells by antibody-coated magnetic beads and immunocytological detection of intracellular rickettsiae.
Polymerase chain reaction (PCR)
Detection of rickettsial DNA by PCR is more rapid than isolation and allows specific identification, but the test is not generally available. It is also insensitive, particularly early in the course of the infection when therapeutic decisions should be made. Peripheral blood mononuclear cells, skin biopsy and tache noire specimens from the site of the bite can be used.
PCR amplification of highly conserved rickettsial genes can also be used. A single primer pair that amplifies all or most rickettsiae can be designed from the genes encoding 16S ribosomal ribonucleic acid (rrs), a 17-kDa protein (htr), citrate synthase (gltA) or ompB. A suitable approach is to use a conserved genus-specific primer pair to amplify a rickettsial gene and then to identify the species by restriction fragment length polymorphism analysis or DNA sequencing. Scrub typhus can be diagnosed by PCR amplification of the 56-kDa protein gene of O. tsutsugamushi.
Treatment
The drug of choice for treating rickettsial infections of all types and in all age groups is doxycycline. Chloramphenicol is an alternative, but is less effective and carries a higher risk of death. Both drugs are rickettsiostatic and allow the patient’s immune system time to respond and control the infection. Sulphonamides should not be administered as they exacerbate rickettsial infections; β-lactam and aminoglycoside antibiotics are ineffective. Some new quinolones and macrolides have antirickettsial effects in vitro, but clinical experience in severe rickettsioses is lacking. Short courses of doxycycline do not cause significant dental staining in young children.
Owing to the difficulties of accurate diagnosis and the risks involved in misdiagnosis, empirical tetracycline therapy is appropriate for patients who have had a fever for 3 days or more and a history consistent with the epidemiological and clinical features of rickettsial disease. The case fatality rate is increased significantly if treatment is delayed for more than 5 days.
Intensive nursing care, management of fluids and electrolytes, and administration of red blood cells to patients who develop anaemia may be needed. Surgery may also be necessary to remove digits and extremities that develop ischaemic necrosis.
Epidemiology
Typhus group infections
Epidemic typhus
R. prowazekii is transmitted from person to person by the body louse, Pediculus humanus corporis; the organisms are present in the faeces of infected lice and enter through the bite wound or skin abrasions. R. prowazekii causes a fatal infection of the louse, which is therefore incapable of long-term maintenance of the rickettsiae, and human beings appear to be reservoirs of epidemic typhus. Patients who suffer a bout of recrudescent typhus (Brill–Zinsser disease) circulate sufficient rickettsiae in their blood to infect approximately 1–5% of lice that feed on them – enough to initiate new epidemics of the disease. R. prowazekii is maintained in an enzootic cycle in North America involving flying squirrels and their fleas and lice.
Murine typhus
Murine typhus is distributed worldwide, particularly in tropical and subtropical coastal regions where the disease is an occupational hazard of working in rat-infested areas such as markets or ports. This disease is maintained in an enzootic cycle involving rats and their fleas, which remain infected for life. Even the inefficient rate of transovarial transmission in fleas may play an important role in maintaining the rickettsiae in nature. Man is infected by the contamination of abraded skin, respiratory tract or conjunctiva with infective flea faeces, in which the rickettsiae can survive for as long as 100 days under optimal conditions of temperature and humidity.
Spotted fever group infections
Tick-borne infections
Tick-borne rickettsiae of the spotted fever group are maintained in enzootic cycles involving ticks and their wild animal hosts. Ticks are the primary reservoirs of the rickettsiae, and maintain the organisms by both trans-stadial transmission (during moulting of larva to nymph and thence to adult tick) and transovarial or vertical transmission. Some horizontal transmission (tick to rodent to tick) is likely to be essential to the survival of virulent rickettsiae in nature because these rickettsiae are somewhat pathogenic to ticks.
Man becomes infected following the bite of infected ticks or through contamination of abraded skin or mucous membranes. People place themselves at risk when they enter areas infested with infected ticks. Infection may also occur through bites of ticks of domestic dogs or if partially fed ticks rupture during manual deticking of dogs. Rocky Mountain spotted fever is endemic in the Americas, especially in the southeastern and south-central USA. In 2007, 2221 cases were reported. A nonfatal illness, African tick bite fever, is often observed in travellers returning from safari in southern Africa.
Rickettsialpox
R. akari is maintained in an enzootic cycle that involves house mice (Mus musculus) and their mites. The arthropod vector is also the primary reservoir and can maintain the organism by trans-stadial and transovarial transmission. Other rodents and their ectoparasites may be able to maintain the rickettsiae in rural areas, but their importance remains unknown. Rickettsialpox is primarily an urban disease associated with mice-infested buildings.
Scrub typhus
The nymphal and adult stages of the mites transmitting O. tsutsugamushi are free living and do not feed on animals. The parasitic larvae (chiggers) occur in habitats that have been disturbed by the loss or removal of the natural vegetation. The area becomes covered with scrub vegetation, which is the preferred habitat for chiggers and their mammalian hosts, and gives the disease its name. The disease is often localized because of the restricted habitat of the chiggers. Persons entering infected areas are at risk.
Control
It is virtually impossible to eradicate rickettsial infections because of their enzootic nature. Measures aimed at reducing rodent or ectoparasite populations may help to reduce the risk of infection. In addition to delousing infested persons, their clothing and bedding should be decontaminated.
Persons entering areas endemic for spotted fever group infections should wear protective clothing treated with tick repellent. Individuals should also examine themselves carefully for ticks as soon as possible after returning from tick-infested areas. The probability of infection is decreased if the tick is removed soon after it attaches. Transmission may require up to 24 h of feeding, perhaps because starved ticks require a partial blood meal if they are to reactivate the virulence of the rickettsiae as well as inject organisms into the feeding site in their saliva.
There is no safe, effective vaccine for any of the rickettsial diseases. The attenuated E strain of R. prowazekii induces protective immunity, but is unsuitable for general use because it causes a mild form of typhus in 10–15% of those inoculated and reverts to a virulent state after animal passage. Inactivated Rocky Mountain spotted fever vaccines afford incomplete protection and are no longer available. A recombinant or attenuated vaccine that contains cross-protective antigens stimulating cellular immunity could protect against both typhus and spotted fever rickettsioses. Scrub typhus vaccines derived from killed rickettsiae do not prevent infection, and experimental recombinant subunit vaccines are ineffective in animals.
Antimicrobial prophylaxis is not recommended for infections with Rickettsia species, as they are only rickettsiostatic and disease develops as soon as the antibiotic is discontinued. Prolonged prophylaxis with weekly doses of doxycycline is effective against scrub typhus, but is probably inappropriate except under exceptional circumstances, for instance during military operations.
Ehrlichia and Anaplasma
Description
The first human ehrlichial disease was recognized in 1954 when Neorickettsia (formerly Ehrlichia) sennetsu was identified as the cause of an illness resembling glandular fever in Japan. Ehrlichia chaffeensis, Anaplasma phagocytophilum and E. ewingii later emerged as the causes of tick-borne diseases in the USA. Ehrlichia and Anaplasma species are transmitted through the bite of ticks. N. sennetsu is suspected to infect a fluke and to cause infection when ingested with parasitized raw fish (Table 40.2).
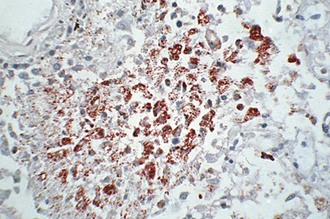
These organisms are small Gram-negative bacteria. They multiply within membrane-bound cytoplasmic vacuoles, usually in various phagocytes, and form characteristic microcolonies resembling mulberries, termed morulae (Latin: morum, mulberry) (Fig. 40.4). Electron microscopy reveals two distinct morphological forms, larger reticulate and smaller dense-core cells, which are the replicating and infectious forms, respectively, in a developmental cycle.
Pathogenesis
Anaplasma and Ehrlichia species can establish prolonged, even persistent, infections in vivo, and some species, including E. chaffeensis, kill heavily infected cells in vitro. Cytokine-associated immunopathological mechanisms are probably important. A. phagocytophilum and E. chaffeensis evade the host immune system by antigenic variation and by modulating the host defences, respectively.
The tropism for phagocytes indicates that these organisms have evolved strategies for evading the microbicidal activities of the macrophage or granulocyte. Within phagocytes A. phagocytophilum and E. chaffeensis block the fusion of phagosome-containing bacteria with lysosomes to prevent killing by lysosomal enzymes. A. phagocytophilum prevents killing mediated by reactive oxygen species by lowering reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity in neutrophils. To accommodate a slow generation time (about 8 h), Anaplasma and Ehrlichia prolong their host cells’ lifespan by inhibiting apoptosis.
Antibody to ehrlichiae confers passive protection, and cellular immunity is crucial to recovery. Suppression of neutrophil function by A. phagocytophilum may predispose to opportunistic infection.
Clinical aspects of infection
Human monocytic ehrlichiosis
The disease begins 1–2 weeks after a bite of an infected tick. Clinical features frequently include fever, headache, myalgias, nausea, arthralgias and malaise. Other manifestations include cough, pharyngitis, lymphadenopathy, diarrhoea, vomiting, abdominal pain and changes in mental status. A fleeting or transient rash involving the extremities, trunk, face or, rarely, the palms and soles appears in 30–40% of patients about 5 days after onset. The rash may be petechial, macular, maculopapular or diffusely erythematous.
Cytopenia early in the course of the illness may provide presumptive clues to the diagnosis. Mild to moderate leucopenia is observed in approximately 60–70% of patients during the first week of illness, with the largest decreases occurring in the total neutrophil count. Thrombocytopenia occurs in 70–90% of patients. Mildly or moderately raised hepatic transaminase levels are noted in most patients at some point during their illness.
About 50% of patients need admission to hospital. Those with severe disease may develop acute renal failure, metabolic acidosis, respiratory failure, profound hypotension, disseminated intravascular coagulopathy, myocardial dysfunction and meningoencephalitis; about 3% die. Death is more common in elderly men and immunocompromised individuals, including those infected with human immunodeficiency virus (HIV).
Human granulocytic anaplasmosis
The incubation period is 7–10 days after a bite by an infected tick. Clinical signs and symptoms are similar to those of human monocytic ehrlichiosis, but the disease is less severe and the mortality rate is less than 1%. Rash and central nervous system involvement are rare.
Human granulocytic ehrlichiosis caused by E. ewingii elicits similar signs and symptoms. The infection has been observed mainly in immunocompromised patients with no fatalities.
Laboratory diagnosis
Ehrlichia and Anaplasma species grow intracellularly, and isolation is difficult. Human infections are diagnosed mainly by demonstrating the development of specific antibodies during convalescence. Indirect immunofluorescence methods use cell culture-propagated organisms. E. chaffeensis, A. phagocytophilum and E. ewingii are detected diagnostically by PCR with specific primers to amplify the ehrlichial DNA. Human granulocytic anaplasmosis can often be diagnosed by identification of characteristic morulae in Giemsa-stained peripheral blood neutrophils; E. chaffeensis is seldom detected in monocytes in blood smears.
Treatment
Doxycycline shortens the course of infection and reduces mortality. The use of chloramphenicol is not recommended.
Epidemiology
Deer and ticks are involved in the ecology of human monocytic and granulocytic ehrlichioses and of granulocytic anaplasmosis. Deer, canines, rodents and domestic ruminants are important reservoirs. Immature ticks obtain ehrlichiae from the blood of infected animals; the organisms are maintained trans-stadially but not transovarially, and are transmitted during a subsequent blood meal. Human infections are strongly associated with the season of tick activity, history of tick bite and distribution of the vectors. Most cases of human monocytic ehrlichiosis are reported between March and November in the south-central and southeastern USA, where Amblyomma americanum is prevalent. Human granulocytic anaplasmosis occurs from March to December in the northern states.
Coxiella
Description
Query or Q fever, first identified as a distinct clinical entity in 1935 after an outbreak of typhoid-like illness among abattoir workers in Australia, is a widespread disease with an almost global distribution. The aetiological agent, C. burnetii, is a fastidious intracellular prokaryote, genetically related to Legionella species.
C. burnetii is a pleomorphic coccobacillary bacterium with a Gram-negative type of cell wall. The organisms typically grow within the phagolysosome of macrophages of the vertebrate host (Fig. 40.5). Structurally distinct large and small cell variants have been described, suggesting that the organism has a developmental cycle. In acidic conditions, similar to those found within a phagolysosome, it actively metabolizes a variety of substrates and can accomplish significant levels of macromolecular synthesis. Prolonged cultivation in vitro results in phase variation due to deletion of genes involved in the synthesis of lipopolysaccharides analogous to the smooth to rough transitions observed in other bacteria. Phase I organisms are representative of strains in nature, whereas phase II organisms appear in laboratory cultures and are avirulent for laboratory animals.
Pathogenesis
Human infection usually follows inhalation of aerosols containing C. burnetii. Entry into the lungs results in infection of the alveolar macrophages and a brief rickettsaemia. Most infections are subclinical, and only 2% of persons infected with C. burnetii are admitted to hospital. The incubation period for the acute form of the disease is usually about 2 weeks but can be longer.
Typical acute Q fever is a self-limiting flu-like syndrome with high fever (40°C), fatigue, headache and myalgia. The patient may also suffer pneumonitis, hepatic and bone marrow granulomata, and meningoencephalitis. Chronic infections can develop, with the organism persisting in cardiac valves and possibly other foci. Endocarditis is rare, but potentially fatal, and may be accompanied by glomerulonephritis, osteomyelitis or central nervous system involvement.
Reactivation of latent infection may occur during pregnancy, and the organism is shed with the placenta or abortus.
Laboratory diagnosis
Diagnosis relies on the demonstration of specific antibodies in an indirect immunofluorescence assay or enzyme immunoassay. Immunofluorescence assay titres peak at 4–8 weeks. PCR amplification has been used to detect C. burnetii DNA in clinical samples from acute and chronic Q fever patients.
Isolation of C. burnetii from patient specimens is a specialized procedure and is not generally recommended because of the extremely infectious nature of the organism.
Treatment
Most infections resolve without antibiotic treatment, but administration of doxycycline reduces the duration of fever in the acute infection and is definitely recommended in cases of chronic infection. In Q-fever endocarditis, long-term administration of a combination of two drugs among doxycycline, ciprofloxacin and rifampicin has been suggested.
C. burnetii may be recovered from some patients after months or even years of continuous treatment. In addition to antibiotic therapy, the haemodynamic status should be monitored. Valve replacement may be necessary in some cases of Q-fever endocarditis.
Epidemiology
Q fever has been found on all continents except Antarctica, but most cases are reported from the UK and France. Elsewhere, the disease often goes unreported, is misdiagnosed, or causes such a mild infection that treatment is not sought. The main reservoirs of the disease are wild and domestic cattle, sheep and goats. Ticks can maintain C. burnetii by trans-stadial and transovarial transmission. Faeces of infected ticks contain very large numbers of C. burnetii, but arthropods are not an important source of infection.
C. burnetii may be the most infectious of all bacteria. Human infections generally follow inhalation of aerosols or direct contact with the organisms in the milk, urine, faeces or birth products of infected animals. The organism can survive on wool for 7–10 months, in skimmed milk for up to 40 months and in tick faeces for at least 1 year. Most individuals acquire the disease as an occupational hazard. Cases are common among abattoir workers and those associated with livestock rearing or dairy farming. Although Q fever normally occurs as isolated, sporadic cases, well-documented outbreaks have been reported.
Control
Elimination of infected reservoir hosts is probably impossible because of chronic infections among the animals and the ability of the organism to survive for long periods in the external environment. Exposure can be reduced by construction of separate facilities for animal parturition, destruction of suspect placental membranes, heat treatment of milk (74°C for 15 s) and efforts to reduce the tick population. Abattoir workers should take care while handling carcasses, especially in the removal and dissection of mammary glands and inner organs. Animal hides should be kept wet until the salting procedure begins. Appropriate containment procedures should be observed in laboratories working with this highly infectious organism.
Inactivated whole-cell vaccines derived from phase I organisms have been developed and generate a protective response.
Audy JR. Red Mites and Typhus. London: Athlone Press; 1968.
Carlyon JA, Fikrig E. Invasion and survival strategies of Anaplasma phagocytophilum. Cellular Microbiology. 2003;5:743–754.
Dalton MJ, Clarke MJ, Holman RC, et al. National surveillance for Rocky Mountain spotted fever, 1981–1992: epidemiologic summary and evaluation of risk factors for fatal outcome. American Journal of Tropical Medicine and Hygiene. 1995;52:405–413.
Dumler JS. Ehrlichioses and anaplasmosis. In: Guerrant RL, Walker DH, Weller PF. Tropical Infectious Diseases. Principles, Pathogens, and Practice. ed 3. Philadelphia: Elsevier Saunders; 2010:339–341.
Hechemy KE, Avsic-Zupanc T, Childs JE, Raoult DA. Rickettsiology: Present and Future Directions. New York: Academy of Sciences, 2003.
Kawamura A, Tanaka H, Tamura A. Tsutsugamushi Disease. Tokyo: University of Tokyo Press; 1995.
Maurin M, Raoult D. Q fever. Clinical Microbiology Reviews. 1999;12:518–553.
Paddock CD, Childs JE. Ehrlichia chaffeensis: a prototypical emerging pathogen. Clinical Microbiology Reviews. 2003;16:37–64.
Treadwell TA, Holman RC, Clarke MJ, et al. Rocky Mountain spotted fever in the United States, 1993–1996. American Journal of Tropical Medicine and Hygiene. 2000;63:21–26.
Walker DH. Rickettsia rickettsii and other spotted fever group rickettsiae (Rocky Mountain spotted fever and other spotted fevers), ed 7. Mandell GL, Bennett JC, Dolin R, eds. Principles and Practice of Infectious Diseases, Vol. 2. Philadelphia: Elsevier Churchill Livingstone. 2009:2499–2507.
Walker DH, Raoult D. Rickettsia prowazekii (epidemic or louse-borne typhus), ed 7. Mandell GL, Bennett JC, Dolin R, eds. Principles and Practice of Infectious Diseases, Vol. 2. Philadelphia: Elsevier Churchill Livingstone. 2009:2521–2524.
Kim I–S, Walker DH. Scrub typhus. In: Guerrant RL, Walker DH, Weller PF. Tropical Infectious Diseases. Principles, Pathogens, and Practice. ed 3. Philadelphia: Elsevier Saunders; 2010:334–338.
Zhang JZ, Sinha M, Luxon BA, Yu X-J. Survival strategy of obligately intracellular Ehrlichia: novel modulation of immune response and host cell cycles. Infection and Immunity. 2004;72:498–507.