59 Togaviruses
Rubella
Key points
• Rubella (German measles) is caused by infection with a single-stranded, enveloped RNA virus of a single antigenic type. It is characterized by a widespread macular rash.
• In the first 3 months of pregnancy, rubella can infect the fetus, causing the congenital rubella syndrome in 70% of cases and leading to abnormalities of the eye, ear and heart, as well as a range of other problems including mental handicap and purpura.
• Clinical diagnosis is unreliable. Serological detection of specific IgM is the investigation of choice.
• Rubella is now uncommon in the UK owing to widespread immunization with a live-attenuated vaccine included in the MMR preparation.
The Togaviridae comprise of two genera, the Alphavirus genus which contains arthropod-borne viruses (see Chapter 51) and the rubivirus genus, which contains rubella virus as its sole member.
In the mid eighteenth century, two German physicians described the disease now known as rubella or German measles. Commonly considered to be a mild disease with few complications, little attention was paid to rubella until the mid 1940s. However, in 1941, an Australian ophthalmologist, Sir Norman Gregg, described the association between maternal rubella in pregnancy and congenital abnormalities in the infant. He noted an increased incidence of congenital cataracts in children. When questioned, the mothers gave a history of having had rubella in early pregnancy when there had been an epidemic in Australia. Since then the importance of maternal rubella for the fetus has been confirmed by many studies.
Description
Rubella virus is classified as a member of the togavirus family, and is the only member of the genus Rubivirus. It has a single stranded positive sense RNA genome and replication occurs in the cytoplasm of infected cells. It has a lipid containing envelope and a pleomorphic shape with a diameter of 60–70 nm and the nucleocapsid has icosohedral symmetry. The envelope is acquired by the virions by budding from cell membranes into intracellular vesicles or to the exterior. There are 3 major virion polypeptides: C, and the envelope glycoproteins E1 and E2. The virus envelope carries a haemagglutinin (E1), present as 5–6-nm projections, which agglutinates the erythrocytes of 1-day-old chicks, pigeons, sheep and human beings, a characteristic that is utilized in the haemagglutination inhibition test for specific antibodies.
Several chemical agents such as detergents and organic solvents inactivate rubella. The virus is stable at 4°C for over 7 days.
Rubella virus was first isolated in cell culture in 1962 in primary vervet monkey kidney and primary human amnion cultures from throat washings, blood and urine. The virus can be isolated in a variety of primary and continuous cell lines (e.g. Vero, RK13, and baby hamster kidney-21 cells) but only certain lines display a cytopathic effect (e.g. RK13). In other cell lines, the presence of virus is demonstrated by immunofluorescence with specific antibody or resistance to superinfection with another unrelated virus such as echovirus 11.
There is only one antigenic type of rubella virus (although there are minor differences between strains). The World Health Organisation has subdivided rubella virus into two clades and thirteen genotypes. In the laboratory, rubella virus can be transmitted to rhesus monkeys, rabbits and some other animals but man is the only naturally infected species.
Clinical features
Postnatal primary rubella
The incubation period for postnatal primary rubella is 12–21 days, with an average of 16–17 days. Virus may be excreted in the throat for up to a week before and after the rash, and this covers the period of infectivity. High concentrations of virus may be excreted but close and personal contact is needed for onwards transmission. Infection has several characteristic clinical features.
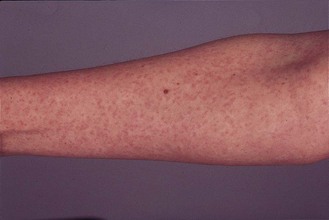
• A macular rash (Fig. 59.1), which usually appears first on the face and then spreads to the trunk and limbs. Particularly in childhood, the rash may be fleeting and perhaps 50% of infections in children are asymptomatic. In adults, asymptomatic rubella is less common.
• General features such as minor pyrexia, malaise and lymphadenopathy also occur, with the suboccipital nodes being those most commonly enlarged and tender.
• Arthralgia is uncommon in children but may occur in up to 60% of women. The joints commonly involved are the fingers, wrists, ankles and knees, and, although arthralgia usually lasts for only a few days, it may occasionally persist for some months.
• Encephalitis and thrombocytopenia are rare complications of rubella and recovery is usually complete.
The clinical features of rubella in an immunocompromised patient are similar to those seen in normal individuals.
Rubella is difficult to diagnose clinically as other virus infections such as some enteroviruses and erythroviruses can present with identical clinical features. Hence laboratory investigation is essential. In the UK, rubella is a notifiable infection; if a doctor suspects a patient has rubella s/he has a legal duty to report it.
Rubella reinfection
Some patients who have had natural infection or have been successfully immunised can display an increase in rubella antibodies, usually only discovered following investigation of a rubella contact. This reinfection is seldom clinically apparent, but laboratory diagnosis is important. Asymptomatic reinfection in pregnancy presents a negligible risk to the fetus whereas clinically apparent reinfection, although rare, may present a risk similar to that of primary infection.
Congenital rubella
If the fetus is infected during a primary maternal infection, a wide spectrum of abnormalities may occur. The classical congenital rubella syndrome (CRS) triad consists of abnormalities of the eyes, ears and heart.
• Abnormalities of the eyes, which may be bilateral or unilateral, include cataracts, micro-ophthalmia, glaucoma and pigmentary retinopathy, which may result in blindness.
• Bilateral or unilateral sensorineural deafness may be present at birth, although not detected until later in life; it may increase in severity as the child gets older.
• There are many possible heart defects, with patent ductus arteriosus, pulmonary artery and valvular stenosis and ventricular septal defect being the most common.
The baby often has a low birth weight due to intrauterine growth retardation. There may be a purpuric rash due to thrombocytopenia but this usually resolves, as does hepatosplenomegaly. Microcephaly, psychomotor retardation and behavioural disorders can occur. Rarely there may be a persistent infection of the central nervous system (progressive rubella subacute panencephalitis) similar clinically to subacute sclerosing panencephalitis due to measles virus infection. Other problems that may present later in life include pneumonitis, diabetes mellitus, growth hormone deficiency, and thyroid function abnormalities.
Some fetuses may be so severely affected that intrauterine death or stillbirth occurs. Many babies are born with no abnormalities; the risk is associated with the gestational age.
• If maternal infection occurs in the first trimester, the risk of CRS is at least 70%, and many babies have multiple developmental defects.
• In the fourth month of pregnancy the risk reduces to around 20%, and the only abnormality likely to be seen is sensorineural deafness.
• After the 16th week of pregnancy, although fetal infection still occurs, congenital abnormalities are infrequent and the risk is no more than in an apparently uncomplicated pregnancy.
Pathogenesis
Postnatal rubella
The virus is transmitted by the airborne route. The virus first replicates in the epithelium of the buccal mucosa and the lymphoid tissue of the nasopharynx and upper respiratory tract. Towards the end of the incubation period a viraemia occurs and seeds target organs such as skin and joints. Most of the clinical features are probably due to the host’s immune response, e.g. virus can be demonstrated in both unaffected skin and the macules of the rash, suggesting that the presence of virus per se is not sufficient to cause the rash.
Congenital rubella
During viraemia the virus infects and crosses the placenta (in early gestation this is an ineffective barrier to infection) to infect the differentiating cells of the fetus. If such fetal infection occurs in early pregnancy a persistent infection is likely. The fetus cannot mount an immune response in early gestation.
The congenital abnormalities arise from a number of effects of rubella infection. Cell division is slowed, cell differentiation becomes disordered and damage to small blood vessels can occur. Such effects may lead to the abnormalities seen at birth but the persistence of infection may result in the clinical problems presenting later in life, possibly due to direct damage such as in late-onset deafness or because of immunopathological mechanisms such as in pneumonitis. Virus can persist for many years in babies infected during early gestation, but persistent infection is rare in later pregnancy, when the babies are not damaged.
Diagnosis
Postnatal rubella
Given the unreliability of clinical diagnosis, laboratory investigation is required to diagnose rubella and this is of paramount importance for the pregnant patient. As subclinical rubella may occur pregnant women should also be investigated if they are in contact with someone who has possible rubella.
Virus isolation has low sensitivity and is time consuming and so has no role to play. Serology is the method of choice. Assays to detect total rubella antibody or rubella specific immunoglobulin G (IgG) and M (IgM) are used together. The assays chosen should have a high level of sensitivity and specificity.
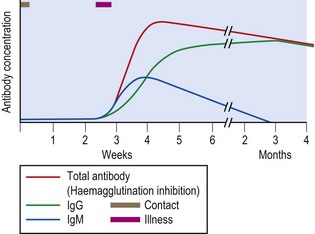
In primary rubella, specific antibody becomes detectable at about the time of the rash, although there may be a delay of 7–10 days, and rapidly increases in concentration (Fig. 59.2). Specific IgM usually (but not always) precedes specific IgG by 1–2 days.
IgG persists for life but IgM generally is only detectable for 1–3 months. If a serum obtained soon after contact but prior to illness is examined in parallel with a sample soon after the appearance of the rash, primary infection can be demonstrated by the development of specific IgM.
Problems in diagnosis can arise if serum is collected after the rash appears or more than 12–14 days after contact as any total rubella antibody or specific IgG may be from recent infection or infection many years prior. Again IgM testing must be performed in order to delineate recent infection. Rubella IgG avidity (the tightness of the bond between antibody and antigen) can also be used to differentiate between recent (low avidity) and remote (high avidity) rubella.
The diagnosis of recent rubella can now also be made by using a genome amplification technique such as Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) on specimens such as throat washings and oral fluids.
Congenital rubella
Most babies with CRS excrete large amounts of virus for the first few months of life (and are very infectious to their carers). Virus isolation was used historically from throat swabs or urine but this has largely been superseded by molecular tests such as RT-PCR, or serology. Maternal IgM does not cross the placenta, so the detection of rubella-specific IgM in a newborn baby is diagnostic of intrauterine infection. Specific IgG crosses the placenta so its detection in the first few months of life is of no help. Maternal IgG has a half-life in the infant of 3–4 weeks, however, so persistence to the age of 9–12 months is diagnostic of congenital rubella. Occasionally, requests are made to diagnose congenital rubella in an older infant, e.g. one who presents with deafness. However, the reliable diagnosis of congenital infection in older children is impossible as any rubella-specific IgM generated by congenital infection will have disappeared, and rubella-specific IgG in the patient’s serum may have arisen from postnatal infection or immunisation.
Screening for rubella antibody
Screening for rubella antibodies in pregnant women performs the function of identifying those individuals who are susceptible and offering them vaccine postpartum. Blood is usually obtained at the first antenatal clinic visit and serum samples routinely stored for one year. This could be important if an infant is delivered with congenital abnormalities as the maternal serum collected in early pregnancy can be examined together with a later maternal one and also the infant’s blood. This allows investigation of not only rubella but other important pathogens such as cytomegalovirus later in pregnancy. Sometimes, preconceptual screening is performed especially in those undergoing in vitro fertilization.
Rubella antibody screening is most usually performed by enzyme immunoassay, a format which is sensitive and easily automated.
Negative results in an EIA test should be confirmed by a different assay such as latex agglutination. Some women do not produce protective levels of antibody (defined in the UK as >10 IU/mL) even after several vaccinations and they should be considered immune if they have had two documented doses of vaccine.
Epidemiology
Rubella has a worldwide distribution and infection is endemic in all countries that have not had a successful immunisation policy. Outbreaks usually occur in spring and early summer with major epidemics occurring every 4–8 years. Infection is common in childhood. Currently only 2–3% of young adult females are susceptible in the UK due to immunisation: many of those who are susceptible come from areas of the world such as the Indian subcontinent, where immunisation is uncommon. It is of paramount importance that the prevalence of pregnant women susceptible to rubella is monitored; should it decrease substantially immunisation protocols may have to change.
Control
Passive prophylaxis
There is little evidence that administering normal human immunoglobulin after contact reduces the risk of maternal rubella and fetal infection, although it may attenuate the illness.
Active prophylaxis
Attenuated live rubella vaccines have been available since the early 1970s. They are safe and only have minimal side effects such as a transient rash and arthralgia. Seroconversion occurs in over 95% of susceptible vaccinees and protection is of more than 20 years duration. Vaccine virus can be isolated from the throat of vaccinees but there is no evidence of onward transmission to susceptible contacts. It is advisable that all women of childbearing age are screened for rubella antibody before immunisation so that only susceptible women are offered vaccine. The vaccine is live and therefore contraindicated in pregnancy, and pregnancy should be avoided in the month after vaccination. However, although vaccine virus has been shown to infect the fetus, there is no evidence of teratogenicity. Thus, if a susceptible woman is immunised inadvertently whilst pregnant, she can be reassured that any risk to the baby is very remote.
The objective of rubella immunisation is to eradicate congenital rubella. Historically in the UK there have been two approaches. In the 1970s vaccination was targeted at girls aged 11–14 years and susceptible adult women, identified by screening for rubella antibody in situations such as antenatal care and family planning clinics. The reasons for this policy were two-fold: firstly there were uncertainties about the duration of protection of the vaccine, and secondly it was thought natural rubella, acquired by around 50% of teenage girls, would afford women greater protection. There was also the consideration of exposure to natural rubella boosting vaccine immunity.
However, by the late 1980s it was apparent that this approach was not achieving the aim of eradicating congenital rubella, as there were still 10–20 cases of CRS and 100–200 terminations of pregnancy per annum in the UK. This was because the small number of women who were susceptible to rubella were becoming infected via their own or other people’s young children. This led to a different approach, commencing in October 1988, whereby all children were offered vaccine in an attempt to eradicate circulating rubella from the community and thus avoid exposure of any susceptible pregnant women. In order to make the vaccine more attractive to parents it was combined with measles and mumps (the MMR vaccine) and offered to both girls and boys at around 15 months of age. There was also a preschool ‘catch-up’ programme. In the late 1990s a second dose of MMR was introduced at 3–5 years to boost immunity of those who had received only one dose and to reach some of the children who had missed their first dose. This approach, similar to that adopted in the USA, has now almost eliminated congenital rubella. However there have been recent concerns in the UK about the safety of MMR vaccine and this has led to a fall in immunisation rates to below 95%, which may lead to a resurgence of rubella.
Banatvala JE, Brown DWG. Rubella. The Lancet. 2004;363:1127–1136.
Best JM, Icenogle JP, Brown DWG. Rubella. In: Zuckerman AJ, Banatvala JE, Schoub BD, et al. Principles and Practice of Clinical Virology. ed 6. Chichester: Wiley-Blackwell; 2009:561–592.
Miller E, Cradock-Watson JE, Pollock TM. Consequences of confirmed maternal rubella at successive stages of pregnancy. Lancet. 1982;ii:781–784.
Health Protection Agency (UK). Rubella (German Measles). http://www.hpa.org.uk/Topics/InfectiousDiseases/InfectionsAZ/Rubella/.