51 Arboviruses: alphaviruses, flaviviruses and bunyaviruses
Encephalitis; yellow fever; dengue; haemorrhagic fever; miscellaneous tropical fevers; undifferentiated fever
Key points
• Arboviruses are transmitted biologically by arthropod vectors (mosquitoes, ticks and biting flies).
• There are three major types of clinical disease caused by these viruses: central nervous system, visceral organs/haemorrhagic fever, and febrile infections, with many progressing from the latter to the former syndromes. Many arboviruses are highly pathogenic and require a high level of biocontainment.
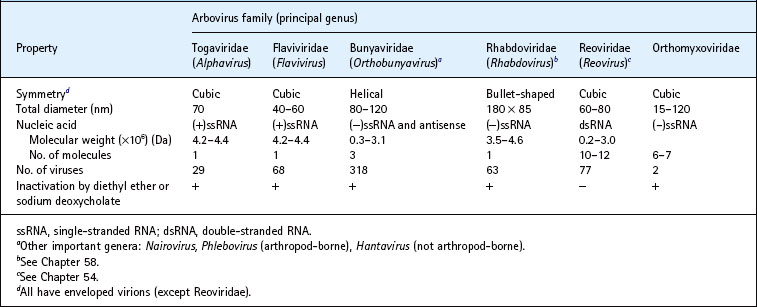
• There are more than 500 recognized arbovirus species classified in six virus families: Togaviridae, Flaviviridae, Bunyaviridae, Rhabdoviridae, Reoviridae and Orthomyxoviridae.
• In the UK arboviruses are overwhelmingly of concern to travellers. Yellow fever and dengue viruses are two widely distributed flaviviruses of global concern, but numerous other agents must be considered in specific locations.
• The Hantavirus genus of the Bunyaviridae contains many pathogens associated with haemorrhagic fever with renal syndrome or hantavirus pulmonary syndrome. Hantaviruses are transmitted by rodents, not arthropods.
• Commercial vaccines are available only for yellow fever, Japanese encephalitis and tick-borne encephalitis, but a number of experimental vaccines show promise.
• The only effective antiviral treatment is ribavirin, which has efficacy only against selected bunyaviruses and alphaviruses.
The name ‘arbo’ (arthropod-borne) virus denotes viruses transmitted biologically by arthropod (mainly insect and tick) vectors. Arboviruses are found in many different taxa, and over 500 individual arbovirus species that are now officially classified in six virus families. Many arboviruses are highly pathogenic and are classified at biosafety level 3 or 4. As there are many similarities in their transmission cycles and in the diseases that they cause, they are considered together in this chapter.
Arboviruses were defined by a World Health Organization Scientific Group as ‘viruses that are maintained in nature principally, or to an important extent, through biological transmission between susceptible vertebrate hosts by haemotophagous arthropods or through transovarian and possible venereal transmission in arthropods; the viruses multiply and produce viraemia in the vertebrates, multiply in the tissues of arthropods, and are passed on to new vertebrates by the bites of arthropods after a period of extrinsic incubation.’
Certain viruses within the six families containing arboviruses are not transmitted by arthropods, but are maintained in nature within rodent reservoirs that may transmit infection directly to man. These include the Hantavirus genus of the family Bunyaviridae.
Description
Classification
Most arboviruses are members of the families Togaviridae, Flaviviridae and Bunyaviridae; some are assigned to the families Reoviridae (genera Coltivirus (e.g. Colorado tick fever virus) and Orbivirus (e.g. Bluetongue viruses)), Orthomyxoviridae (e.g. Thogoto virus) and Rhabdoviridae (members of the genera Vesiculovirus (e.g. vesicular stomatitis virus) and Lyssavirus) (Table 51.1). Within the Togaviridae, only one (Alphavirus) of the two genera contains arthropod-borne viruses; the other genus, Rubivirus, contains rubella virus, which is not arthropod-borne, as its sole member (see Ch. 59). The Flaviviridae contain three genera (Flavivirus, Pestivirus and Hepacivirus), but only the Flavivirus genus contains arthropod-borne viruses. Pestiviruses infect only vertebrate animals (e.g. bovine viral diarrhoea virus), and hepatitis C virus is described in Chapter 52. The Bunyaviridae consist of five genera (Orthobunyavirus, Hantavirus, Nairovirus, Phlebovirus and Tospovirus), containing a total of over 300 species, and is the largest virus family. The Tospovirus genus contains plant viruses that are transmitted by vectors (thrips), whereas the Hantavirus genus contains viruses that are transmitted by rodents rather than arthropods.
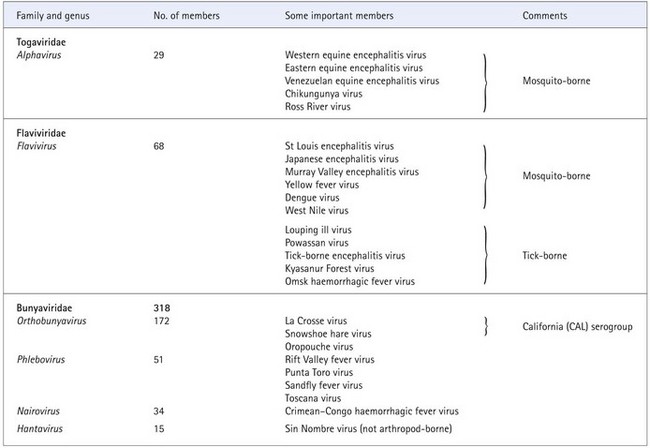
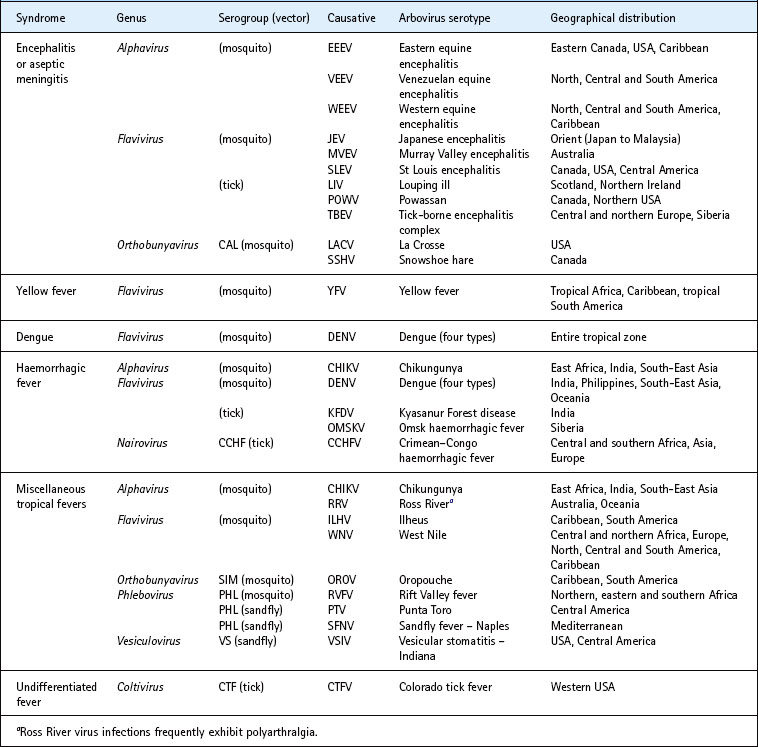
The classification of arboviruses into individual species is now made on the basis of polythetic criteria including genetic as well as antigenic and other phenotypic characteristics. Clusters of viruses that show antigenic overlap are termed serogroups or antigenic complexes. Table 51.2 lists some of the important members.
Table 51.2 Some important arboviruses
Properties
Arboviruses share common biological attributes (see Table 51.1):
1. Most induce fatal encephalitis 1–10 days after intracerebral inoculation of mice aged less than 48 h; some also induce fatal encephalitis after intracerebral inoculation of weaned mice aged 3–4 weeks.
2. Haemagglutination. Most arboviruses can agglutinate erythrocytes, an ability that is inhibited by antiserum against viruses within the same serogroup. Seroreactivity against viruses from dissimilar serogroups is generally weak.
3. Many arboviruses multiply in continuous polyploid tissue cultures of mammalian cells incubated at 37°C, such as grivet monkey kidney (Vero) and baby hamster kidney (BHK).
4. Many arboviruses, such as dengue and Ross River viruses, multiply in continuous cultures of mosquito cells when incubated at 34°C or lower temperatures; Aedes albopictus C6/36 mosquito cells are often used. In general, mosquito-borne viruses do not replicate in tick cell cultures and vice versa.
5. Mosquito-borne arboviruses multiply after oral feeding or intrathoracic injection of several Aedes and Culex mosquito species after incubation at 4–28°C (depending on the mosquito species). Intrathoracic susceptibility of mosquitoes to dengue and California serogroup viruses is 10 to 100 times higher than that of mammalian tissue cultures or suckling mice. Some arboviruses, including members of the Orthobunyavirus and Phlebovirus genera of the Bunyaviridae, Flavivirus genus of the Flaviviridae, Alphavirus genus of the family Togaviridae and some members of the Vesiculovirus genus of the family Rhabdoviridae, are also transmitted transovarially by vectors. Ticks do not normally transmit mosquito-borne arboviruses and vice versa. Sandfly-borne viruses are transmitted only by sandflies (Phlebotomus spp. and Lutzomyia spp.).
6. Tick-borne arboviruses multiply after oral feeding to larval or nymphal ixodid ticks (hard ticks of the genera Dermacentor and Ixodes). The virus is transferred trans-stadially to the next developmental stage (nymph or adult, respectively), which then transmits virus by biting susceptible vertebrates.
Replication
The replication of the various arboviruses differs significantly and is one of the major criteria used in their classification (see Ch. 7 for a general account).
Alphaviruses
Alphaviruses are 70 nm in diameter and enter cells by receptor-mediated endocytosis but co-receptors or other factors probably also contribute to entry and cell specificity. The virion fuses with an endosomal membrane via a fusion peptide in the E1 envelope glycoprotein. The nucleocapsid is released and binds to ribosomes, and the non-structural proteins are translated directly from the genomic RNA. RNA replication occurs in complexes comprised of the non-structural proteins and cellular proteins in association with cytoplasmic membranes. Genomic RNA is messenger sense (positive strand) and serves as a template for full-length negative sense RNA synthesis; these negative-sense RNAs are templates for the production of positive-sense genomic RNA, as well as a subgenomic mRNA that encodes the structural proteins. Both of these RNAs are capped at the 5′ end and polyadenylated at the 3′ end. Regulation of positive- versus negative-strand synthesis occurs via changes in the non-structural protease activity mediated by different cleavage patterns of the non-structural polyprotein. The subgenomic message is translated to yield a polyprotein comprised of the capsid and envelope glycoproteins. The capsid is cleaved co-translationally in the cytoplasm via its own protease activity, and the remaining polyprotein enters the endoplasmic reticulum, where it is processed through the secretory pathway to yield glycosylated E2 and E1 protein heterodimers in the plasma membrane. Genomic RNA combines in the cytoplasm with 240 copies of the capsid protein to form a nucleocapsid, and nucleocapsids interact with the cytoplasmic tail of the E2 envelope protein to mediate budding, whereby 240 E2/E1 protein heterodimers and a portion of the plasma membrane are incorporated into the mature virion.
Flaviviruses
Flavivirus virions are 50 nm in diameter and have three structural proteins; the genome is encapsidated by a small core protein and there are two proteins, the membrane (M) and envelope (E), on the outside of the virus particle. The E protein is the major protein of the virus. It is normally glycosylated, has haemagglutination activity, and is the target of neutralizing antibodies. The NS3 protein (see below) contains the majority of T cell epitopes. The virus genome is one single-stranded, positive-sense RNA molecule. Flavivirus RNA is not polyadenylated. The 5′ one-third of the genome encodes the three structural proteins, and the remaining two-thirds encodes seven non-structural (NS) proteins involved in virus replication, including an RNA-dependent RNA polymerase. Flaviviruses replicate in the cytoplasm of cells. The input virion RNA is translated as a single open reading frame to generate a polyprotein precursor that is rapidly co- and post-translationally processed by viral and cellular proteases to yield the structural and NS proteins. Flavivirus particles assemble by budding through Golgi vesicles and contain prM, a precursor to M protein, as a chaperone for the E protein. Mature virions are produced at the cell surface where the ‘pr’ portion of prM is cleaved by the cell enzyme furin to yield the mature M protein found in virions.
Bunyaviruses
The Bunyaviridae are icosahedral enveloped viruses, with diameters of 100–120 nm. They have tripartite RNA genomes of negative sense, termed the large (L), medium (M) and small (S) segments. The L RNA encodes the RNA-dependent RNA polymerase carried in the virion. The L protein of Crimean–Congo haemorrhagic fever virus encodes an ovarian tumour-like cysteine protease motif, suggesting that the L polyprotein is cleaved autoproteolytically. The M RNA encodes two glycoproteins, Gc and Gn, found on the surface of virions. The S RNA encodes a nucleocapsid (N) protein.
The Orthobunyavirus genus also contains a non-structural protein, NSs, which is an interferon antagonist that also inhibits host cell protein synthesis. The tripartite genome enables bunyaviruses to undergo genetic reassortment that occurs naturally between closely related bunyaviruses and contributes to genetic variation and evolution. The NSm gene of the Tospovirus genus and the NSs gene of the Phlebovirus and Tospovirus genera are encoded as genes in the positive-sense orientation. Thus, the S- and M-RNA segments of tospoviruses and the S-RNA of phleboviruses are termed ambisense RNAs.
Bunyaviruses replicate in the cytoplasm of cells and assemble by budding through Golgi vesicles.
Pathogenesis
Natural vertebrate infection by arboviruses is initiated when mosquitoes or other arthropods deposit saliva in extravascular tissues and blood vessels while blood-feeding. For some alphaviruses, murine model systems using needle inoculations indicate that the initial site of replication is the Langerhans cell. Alphavirus replication appears to stimulate both the migratory response of these cells to, and the accumulation of leucocytes in, the draining lymph nodes, where local replication produces viraemia. Arboviruses induce high titres of viraemia in many susceptible vertebrates 1–4 days after parenteral inoculation or following bites by infected arthropods, resulting in the potential for infection of additional vectors. Invasion of the central nervous system (CNS) via the olfactory nervous tract may ensue in some infections, whereas other viruses cross the blood–brain barrier. In alphavirus infections accompanied by rash and arthritis, virus replication and necrosis occur in the epidermis and possibly the muscles, tendons and connective tissue. Infection of macrophages may mediate musculoskeletal pathology via the release of inflammatory mediators. Wild bird and mammal reservoir hosts regularly exhibit viraemia without symptoms.
Antibodies are first detected when the fever subsides, usually within 5–10 days after infection, and may persist for many years. Antibodies are of the immunoglobulin (Ig) M class for 1–7 weeks after infection; subsequently they are of the IgG class, which appears 2–3 weeks after infection.
Arboviruses cause a spectrum of disease ranging from inapparent infection (often the most likely outcome) to acute encephalitis. Within the CNS, arboviruses multiply in and induce necrosis of neurones, which in turn become surrounded by microglia, forming glial knots. There is also evidence of apoptosis for some virus infections. An age dependence of CNS disease has been observed for many arboviruses, and animal model systems indicate that age-dependent apoptosis of neurones may explain this phenomenon. Perivascular cuffing with mononuclear cells affects many cerebral blood vessels. Usually there is concomitant meningitis with accumulation of mononuclear cells in the subarachnoid space and hyperaemia of adjacent capillaries. Invasion of the CNS appears to be a critical determinant in the pathogenesis and is due in some cases to the level of viraemia. The role of the immune system is not clear, although it may be involved in the pathogenesis of dengue haemorrhagic fever and dengue shock syndrome. Antigen–antibody complex formation may underlie the syndrome, which is associated with increased capillary permeability and shock, often with haemorrhage. It is known that the uptake of virus into macrophages is enhanced in the presence of non-neutralising antibody, as the virus–antibody complexes bind to Fc receptors, so there is likely to be a great increase in the uptake and release of virus from macrophages.
Some arboviruses including alphaviruses (e.g. Ross River virus), flaviviruses (e.g. West Nile virus) and bunyaviruses (e.g. Bunyamwera virus) can disrupt the activation of specific innate immunity antiviral pathways, including interferon induction, to enhance their replication.
Clinical features
The tissue tropism of arboviruses can be divided into three categories:
1. the CNS (e.g. encephalitis, aseptic meningitis)
Human arbovirus infections become clinically manifest according to the target organ principally infected (Table 51.3). These include the following syndromes.
Table 51.3 Clinical syndromes associated with selected arboviruses and their geographical distribution
Encephalitis
For many arboviruses, encephalitis is most common in children and/or the elderly. Illness typically has an abrupt onset within 1 week of infection, with headache, fever, myalgia and dysthesias, and sometimes lethargy, chills, dizziness, nausea, vomiting and prostration. Inflammation of the throat, cervical lymphadenitis and abdominal tenderness are also common. Signs and symptoms usually subside after several days, but may recrudesce. Progression to encephalitis may occur rapidly, or a prodromal illness may last for 1 week or more. Severe CNS disease is accompanied by neck stiffness, motor weakness and paralysis, meningismus, cranial nerve palsy, confusion, convulsions and somnolescence, leading to coma. Rigidity or weakness of the limbs may occur with reduction in reflexes. White blood cells, predominantly lymphocytes, and raised glucose levels may occur in the cerebrospinal fluid, which can exhibit increased pressure. Peripheral blood cell counts may be raised, with a left shift. During most outbreaks of arboviral encephalitis, a proportion of patients develop aseptic meningitis alone, without significant neuronal involvement, whereas up to 50% of patients recover from acute encephalitis to suffer from neuropsychiatric sequelae varying from physiological impairment to mental disorder, which may last from months to years.
Yellow fever
Yellow fever is caused by a mosquito-borne flavivirus that is found in tropical South America and Africa. The disease has an incubation period of 3–6 days, characterized by the sudden onset of headache and fever (temperatures may exceed 39°C), with generalized myalgia, nausea and vomiting. Jaundice may appear by the third day of illness, but frequently is mild or absent. Haematemesis, melaena, epistaxis and bleeding gums may also be noted. Albuminuria and oliguria may also begin suddenly during the first week of illness. In severe cases, death may occur 3–6 days after the onset of illness, and mid-zonal necrosis is observed in the liver. The case fatality rate is estimated as 20–50%.
Dengue
Dengue is caused by four serologically related flaviviruses called dengue-1, -2, -3 and -4. These viruses are found in most tropical parts of the world. Dengue presents as an acute febrile illness with chills, headache, retro-ocular pain, body aches and arthralgia in more than 90% of apparent cases, with nausea or vomiting and a maculopapular rash resembling measles lasting for 2–7 days in about 60% of cases. Illness persists for 7 days, fever remitting after 3–5 days, followed by relapse (‘saddleback fever’) and pains in the bones, muscles and joints sufficiently severe to earn the epithet ‘breakbone fever’. Rash occurs more commonly in patients aged less than 14 years. Complete recovery is the rule. The incubation period is 5–11 days.
Dengue haemorrhagic fever
This is a less common manifestation of dengue, with about 500 000 cases per year and a case fatality rate of 15%, mainly affecting children. It is occasionally accompanied by a shock syndrome, known as dengue shock syndrome, with a case fatality rate of 50%. These two severe forms of dengue are observed in patients who undergo successive infection with two different dengue viruses (e.g. a primary dengue-1 infection followed by a secondary infection with dengue-2 virus). After an acute onset, fever of 40°C, accompanied by vomiting and anorexia, enlarged liver and petechiae persists for 5–10 days. This is followed by a complete recovery unless shock supervenes, as occurs in 7–10% of patients 2–7 days after onset, usually accompanied by haematemesis and melaena.
Miscellaneous tropical fevers
These comprise an increased temperature to above 39°C with any combination of headache, myalgia, malaise, nausea or vomiting, and sometimes accompanied by maculopapular rash or polyarthralgia, that is, a dengue-like syndrome but without haemorrhagic manifestations or the shock syndrome. Tropical fevers arise from infection with a wide variety of arboviruses (see Table 51.3) and clinical diagnosis is impossible without access to serological tests for diagnosis. Many of the Old World alphaviruses such as Ross River, chikungunya, o’nyong-nyong and Sindbis viruses cause an arthritic syndrome accompanied by rash, which can persist for months. Chronic or relapsing arthritic symptoms may be caused by inflammation associated with periodic increase in replication within peristently infected synovial macrophages, which persist despite neutralizing antibodies and antiviral cytokine responses. Inhibition of cytokine responses by virus–antibody complexes binding to Fc receptors, and interleukin-10 induction, may facilitate persistence.
Undifferentiated fever
A US example is Colorado tick fever (genus Coltivirus) in which symptoms of chilliness, headaches, retro-orbital pain and generalized aches, especially in the back and limbs, appear 3–6 days after bites by infected Dermacentor andersoni ticks in wooded areas of the Rocky Mountain region. Fever of 39–40°C often shows a biphasic course, with eventual defervescence within 1 week, followed by complete recovery.
Hantavirus pulmonary syndrome
This syndrome, due to the hantavirus Sin Nombre, was first recognized in south-western USA during 1993. Sudden onset of fever, myalgia, headache, cough, and nausea or vomiting is accompanied by rapid respirations exceeding 20 per minute, temperature above 38°C and hypotension. Extensive interstitial and alveolar infiltrates are observed in the lungs, accompanied by reduced oxygen saturation. Fatal cases develop progressive pulmonary oedema with hypoxia and severe hypotension, and die 2–16 days after onset of symptoms; the case fatality rate may exceed 50%. Patients with non-fatal infection usually recover within 1–3 weeks.
Laboratory diagnosis
Diagnosis of arbovirus infections depends on:
• the isolation of virus from blood, cerebrospinal fluid or tissues
• detection of arbovirus-specific RNA in blood, cerebrospinal fluid or tissues
• antigen detection by indirect immunofluorescence, commonly used as a rapid diagnostic method
• serology – haemagglutination inhibition and enzyme-linked immunosorbent assays (ELISAs) and neutralization tests can be used to detect serum antibodies from patients. However, these assays are dependent on availability of virus and/or antigens.
Virus isolation
Virus isolation is an important diagnostic tool, but is normally a slow process and unlikely to generate results until after an acute disease. The causative virus can often be isolated from blood collected during the initial 3–4-day febrile illness when viraemia titres peak. Some arboviruses can also be isolated from cerebrospinal fluid or brain biopsy, or from brain at autopsy of fatal encephalitis cases. Liver may yield virus isolation from fatal cases of yellow fever.
Arbovirus-specific RNA detection
Detection of virus-specific RNA, by reverse transcriptase–polymerase chain reaction (RT-PCR) and the more sensitive real-time RT-PCR, are rapid approaches to diagnosis but depend on the availability of specific oligonucleotide primers. Given the large number of arboviruses, there must be significant differential diagnosis before selection of appropriate primers. Genus-reactive primers have been described for alphaviruses, flaviviruses and bunyaviruses.
After amplification of extracted RNA, the product is analysed by restriction enzyme digestion and/or determination of the nucleotide sequence of the PCR product and comparison with sequences in Genbank or other nucleotide sequence databases.
Serology
Serological testing is often the only available means of laboratory diagnosis of encephalitis. The detection of a four-fold or greater rise of antibody titre by haemagglutination inhibition tests or ELISAs on paired sera collected during the initial week and several days later may provide good, but not definitive, evidence of concurrent infection. Antibodies detected by complement fixation testing first appear 2 weeks or more after onset and become undetectable by 3 years. Haemagglutination inhibition and complement fixation test results are usually not as specific as those obtained by neutralization. Virus-specific IgM antibody may be detected within 1 day of onset of clinical symptoms using an IgM capture ELISA. IgM antibody generally wanes 1–3 months after onset and is replaced by IgG antibodies beginning 2–3 weeks after infection. IgM antibodies are indicative of a recent infection.
Treatment
Currently, no specific anti-arboviral therapeutic agent is available. Patients with encephalitis are managed supportively, using anticonvulsants as required, and ice packs are applied when indicated to reduce hyperthermia. Raised intracranial pressure can also be treated, and airway protection may be needed in unconscious patients, with hyperventilation accompanied by anaesthesia and sedation. Brain swelling can be minimized by regulating serum sodium levels and osmolarity. Nosocomial infections, especially pneumonia, should be prevented and treated aggressively when they occur. Similarly, in dengue and haemorrhagic fevers, supportive measures may include careful maintenance of fluid and electrolyte balance. Ribavirin shows activity against some bunyaviruses (and in combination with interferon-α), but has not been well evaluated and is not therapeutic in many arbovirus infections.
Epidemiology
Natural cycles
Arboviruses are maintained in natural transmission cycles involving reservoir hosts and arthropod vectors, typically:
Except for dengue virus and chikungunya virus in some locations, arboviruses are zoonotic pathogens that utilize wild animals as reservoir hosts. Many arboviruses also use non-human animals as amplification hosts during epidemics, and human beings are often tangentially infected, dead-end hosts during these outbreaks. Mosquitoes or other arthropods become infected by engorging on a viraemic vertebrate. In mosquitoes, infection begins in the midgut epithelium and spreads to the haemocele or open body cavity, where it may disseminate to other tissues and organs including the salivary glands. This extrinsic incubation period is completed when replication in salivary gland acinar cells leads to virus release into the apical cavities and salivary ducts. Transmission may then occur upon a subsequent blood meal, when mosquitoes deposit saliva in extravascular tissues while probing to locate a venule, or intravascularly. Infection of the vertebrate then leads to viraemia and the opportunity for infection of additional vectors. Nonviraemic transmission, where by an infected vector transmits to an uninfected vector feeding at the same time on the same host before replicative viraemia can occur, has been described for tick-borne viruses and West Nile virus (WNV).
Examples of natural cycles are:
• human–mosquito cycle, as in DENV, CHIKV and urban YFV (Fig. 51.1)
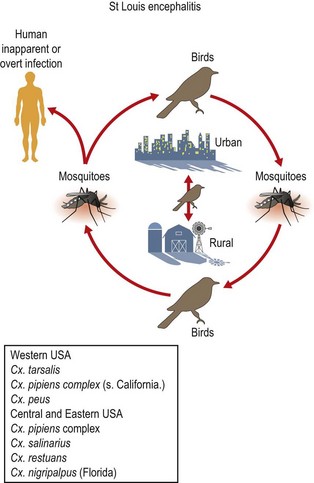
• mosquito–bird cycle, as in SLEV and WNV (Fig. 51.2)
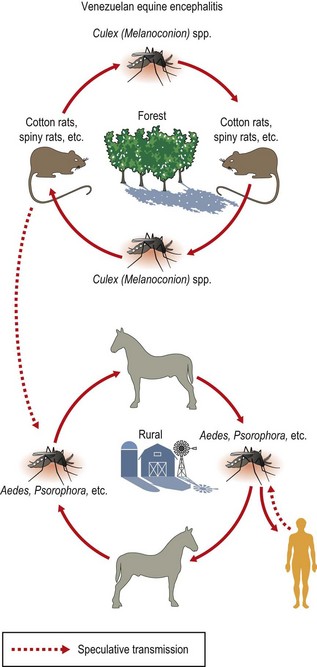
• mosquito–mammal cycle, as in Venezuelan equine encephalitis virus (VEEV; Fig. 51.3).
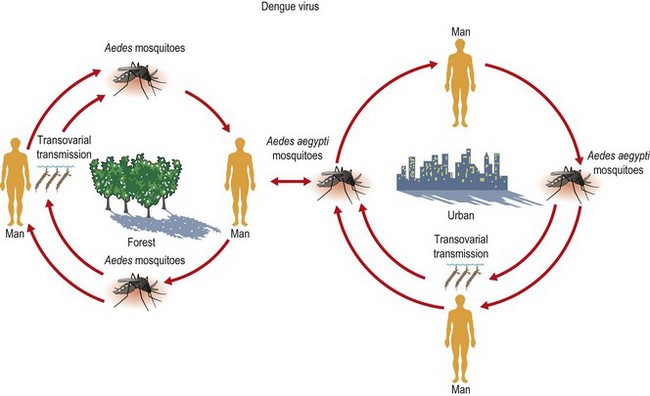
Fig. 51.1 Natural cycle of arbovirus infection: human–mosquito cycle (e.g. dengue and urban yellow fever).
Epidemiological aspects of arbovirus infections with major impact on human health are described according to syndrome and infecting serotype (see also Table 51.2).
Alphaviruses
Western equine encephalitis virus
Western equine encephalitis virus (WEEV) has caused periodic outbreaks of equine and human encephalitis in the western half of North America, as well as in Brazil and Argentina. Although massive outbreaks occurred during the mid-20th century in western North America, only one human case has been documented since 1998. In North America, cases occur only during June to September, when mosquitoes are abundant. Outbreaks of infection occur at intervals of a few years and are preceded by epizootic peaks of encephalitis among horses. The principal mosquito vector species in Canada and the western USA is Culex tarsalis. Passerine birds constitute the principal reservoirs. Human beings and equids are considered dead-end hosts because they produce little viraemia. Although all age groups may be involved, encephalitis due to WEEV (and California serogroup agents) commonly affects children, in whom the disease is often severe.
Eastern equine encephalitis virus
Eastern equine encephalitis virus (EEEV) causes human and horse cases in Atlantic coastal areas extending from Massachusetts and New Jersey to Florida and Texas and northeastern Mexico, and in a few inland locations such as Michigan and Wisconsin. Occasionally, disease extends northwards into Canada. In the United States, an average of 6 human cases of EEEV are reported annually. Illness affects mainly those aged less than 14 years and more than 55 years, with overall case fatality rates as high as 69%. Surviving patients usually have severe neurological sequelae. Most human cases occur during late August and September, about 3 weeks after the peak of horse cases. Distinct forms of EEEV occur throughout South and Central America, but are rarely associated with human disease. In North America, EEEV is maintained enzootically in hardwood swamp habitats, where the mosquito Culiseta melanura transmits among passerine birds. During years of hyper-enzootic transmission, horses and human beings residing near the swamp habitats become infected via bridge vectors such as Aedes spp. (Cs. melanura feeds primarily on birds). Equids and humans are considered dead-end hosts because they develop little viraemia, and outbreaks are therefore confined to regions of enzootic activity.
Venezuelan equine encephalitis virus
Venezuelan equine encephalitis virus (VEEV) causes outbreaks involving up to hundreds of thousands of human beings and equids, primarily in northern South America, with one outbreak extending as far north as Texas in the USA in 1971. During outbreaks, VEEV is transmitted among equids by a variety of mosquitoes such as Aedes and Psorophora spp. Equids are extremely effective amplifying hosts because they develop high-titred viraemia and are attractive to mosquitoes. Antigenically related viruses occur in sylvatic and swamp habitats through much of the Neotropics and subtropics, as far north as Florida, USA, and as far south as northern Argentina. These viruses can cause febrile illness and sometimes fatal encephalitis in human beings that enter these foci, and one antigenic subtype (designated ID) can mutate to become capable of initiating widespread outbreaks. Rates of encephalitis in apparent infections (generally 5–15% of symptomatic cases) and mortality (around 0.5%) are generally lower than for EEEV, with most fatal cases occurring in young children. Attack rates can exceed 50% and neurological sequelae are common.
Ross River virus
Ross River virus (RRV) infection, known in Australia as epidemic polyarthritis, has also occurred in the South Pacific. In Australia, disease occurs primarily during the summer and autumn as sporadic cases and small outbreaks. An average of 5000 cases is reported each year, mostly involving vacationers and others in rural areas. RRV is maintained in a zoonotic vertebrate–mosquito cycle, with Culex annulirostris and Aedes vigilax serving as the principal vectors, and flying foxes and marsupials implicated as reservoir hosts. Human infections have also been documented in New Guinea, the Solomon Islands, New Caledonia, Fiji, American Samoa and the Cook Islands. During 1979–1980 a large, explosive epidemic of Ross River polyarthritis swept across the South Pacific, with 40–60% of the population affected on some islands. Aedes polynesiensis was implicated as the vector, and epidemiological studies suggested a man–mosquito–man transmission cycle.
Chikungunya virus
Chikungunya virus (CHIKV), named after a Swahili word meaning ‘that which bends up’, referring to the posture of patients with severe joint pains, has probably occurred sporadically in India and South-East Asia for at least 200 years. However, it has its enzootic ancestry in sub-Saharan Africa where a sylvatic transmission cycle occurs between wild primates and arboreal Aedes mosquitoes. Urban CHIKV epidemics in the Indian Ocean, India and South-East Asia, causing hundreds of thousands of cases, involve Ae. aegypti and/or Ae. albopictus transmission in a man–mosquito–man cycle. Unlike dengue, which is endemic in many of these Asian cities, CHIKV may disappear and reappear at irregular intervals. However, CHIKV infection is often overlooked because it is clinically indistinguishable from dengue fever. Since March 2005, epidemics have taken place in the Indian Ocean, Asia and Africa involving millions of people with some fatalities. Disability-adjusted life year (DALY) estimates during the 2006 Indian Ocean epidemic exceeded 265 per million in some regions, accounting for up to 69% of the total DALYs, and case-fatality rates have been estimated at ca. 0.1%. Transmission in many locations has been enhanced by the adaptation of newly emerged strains to Ae. albopictus mosquitoes via a mutation in the E1 envelope glycoprotein gene. The ability of this vector to mediate epidemic transmission in a temperate region of an industrialized nation (Italy), underscores the direct risk of CHIKV to many parts of the world including Europe and the Americas. Like dengue, CHIK intervention using vector control has been ineffective, even in highly regulated and industrialized nations like Singapore.
O’nyong-nyong virus
O’nyong-nyong virus (ONNV), derived from the description by the Acholi tribe, meaning ‘joint breaker’, was first isolated during a 1959–1962 epidemic affecting 2 million people in Uganda, Kenya, Tanzania, Mozambique, Malawi and Senegal. Another major outbreak involving an estimated 1 million cases occurred in 1996 in Uganda and northern Tanzania. Attack rates are generally high and all age groups are affected during ONNV epidemics. The transmission cycle involves Anopheles funestus and Anopheles gambiae mosquitoes; ONNV is the only known alphavirus with Anopheles vectors. The reservoir and amplification hosts are unknown.
Flaviviruses: mosquito-borne
St Louis encephalitis virus
St Louis encephalitis virus (SLEV) was first isolated from the brain of a person dying from acute encephalitis in St Louis, Missouri. SLEV activity is widely distributed throughout the USA. The largest outbreak in recent times occurred in 1975, when 1815 (86%) of 2113 confirmed cases of arbovirus encephalitis were due to SLEV. Mosquito vectors in California, the Rocky Mountains and plains states comprise mainly Cx. tarsalis, but Cx. pipiens and Cx. quinquefasciatus are important along the Mississippi Valley and eastwards. The salt marsh mosquitoes Cx. restuans and Ae. sollicitans may be important vectors in some localities. SLE virus is seen most often in persons aged 55 years or more.
Japanese encephalitis virus
Japanese encephalitis virus (JEV) was first isolated from the brain of a patient with fatal encephalitis in Tokyo in 1935. It continues to cause epidemics of encephalitis, affecting children, particularly in India, Korea, China, South-East Asia and Indonesia, with case fatality rates often exceeding 20%. JEV has recently emerged in the Torres Straits and the tip of northern Australia. Approximately 50 000 cases occur each year, of which 15 000 are fatal. The ratio of apparent to inapparent infection is 1 : 50–400, depending on the geographical area. Cx. tritaeniorhynchus mosquitoes are the principal vectors, and maximal virus isolation rates from mosquitoes occur during late July, simultaneously with human and equine epidemics. Important vertebrate reservoirs are black-crowned night herons and other water birds, and pigs are considered to be an amplifying host.
West Nile virus
West Nile virus (WNV) was first isolated from a febrile patient in the West Nile district of Uganda in 1937. The virus has a wide geographical distribution, including southern Europe, Africa, central and south Asia, and Oceania. Genetic studies have shown that the related Kunjin virus, found in Australasia, is a subtype of West Nile virus. Although the virus infects a wide variety of animals, including horses, cattle and man, the major vertebrate hosts are wild birds, and migratory birds are important in the spread of the disease owing to long-term high-titre viraemia. The principal vectors are considered to be mosquitoes of the Culex genus. Both sylvatic and urban transmission cycles have been reported, with Cx. pipiens implicated as the major urban vector. WNV usually causes a febrile illness, but encephalitis is seen in patients over 50 years of age. Epidemics vary in size, with the largest reported in Israel and South Africa, including an epidemic of 3000 clinical cases in South Africa in 1974 and over 400 cases in Russia in 1998, and an equine epidemic in North Africa in 1998. In the late summer of 1999, the first outbreak of WNV infection was reported in the western hemisphere, in New York. A total of 62 clinical cases was reported, mostly in the elderly, including seven deaths.
Since then WNV has spread across the USA with at least 4161 human cases (277 fatalities) and more than 14 000 equine cases reported in 2002. This is the largest recorded epidemic of arboviral meningo-encephalitis in the USA. Overall, in the USA there had been more than 29 500 human cases and over 1150 deaths by the end of 2009. As of 2006, the distribution of the virus has expanded to include the continental USA and seven Canadian provinces, as well as Mexico, El Salvador and many of the Caribbean Islands. During 2006 the virus spread as far south as Argentina, causing disease in equines. WNV has infected at least 308 species of birds, 30 other vertebrate species and 60 species of mosquitoes. In addition, cases of WNV infection have been imported into European countries from endemic areas.
Usutu virus
Usutu virus, isolated in Africa, is a member of the Japanese encephalitis serogroup of the Flavivirus genus and was not known to cause disease. In 2001 the virus emerged in Austria, in Vienna and surrounding districts, and was responsible for a large epizootic with high mortality in birds, especially Eurasian blackbirds (Turdus merula) and owls. The virus has continued to cause epizootics in birds plus some human cases.
Murray Valley encephalitis virus
Murray Valley encephalitis virus (MVEV) was first isolated from the brain of a patient with fatal encephalitis at Mooroopna, Victoria, Australia. MVEV caused epidemics of encephalitis in irrigated farming regions of the Murray–Darling River basin during the summer months (January to March) of 1951 and 1974, with case fatality rates approaching 40%. MVE virus was isolated from Cx. annulirostris mosquitoes only during 1974 but not during intervening years, which suggests epidemic introduction of virus into this dry temperate region. In the tropical irrigated Ord River region of Western Australia, the virus is endemic and encephalitis cases occurred in 1974, 1978, 1981 and 1986. Another endemic focus exists in the Gulf of Carpentaria region of Queensland. Natural cycles of transmission of MVEV involve Cx. annulirostris as the principal mosquito vector and water birds as reservoirs.
Yellow fever virus
Yellow fever virus (YFV) was first isolated in Ghana from the blood of a male patient with fever, headache, backache and prostration. YFV is found mostly in tropical Africa and tropical South America. There are two epidemiological patterns. In the sylvatic cycle, virus is transmitted among monkeys by mosquitoes (Haemagogus and Sabethes species in South America and Aedes species in Africa). Humans are infected incidentally when entering the area, for example to work as foresters. In the urban cycle, person-to-person transmission is via Ae. aegypti, which uses larval habitats close to human habitation in water, pits and scrap containers such as oil drums.
In the Americas, the majority of YFV cases are reported in Brazil, Peru and Bolivia, and involve males aged 15–45 years who are agricultural and forest workers. YFV usually occurs from December to May, and peaks during March and April, when populations of Haemagogus mosquitoes are highest during the rainy season.
YFV is endemic in many parts of West, Central and East Africa, principally between latitudes 15°N and 15°S, extending northwards into Ethiopia and Sudan. Continuing activity has been encountered in parts of West Africa. There are relatively few outbreaks in East Africa with the last major outbreaks in Sudan in 2003 and 2005. Ae. africanus is the principal vector in Africa.
Dengue viruses
Dengue virus (DENV) infection is endemic in all tropical regions between latitudes 23.5°N and 23.5°S. Four serologically related viruses termed dengue-1, -2, -3 and -4 cause the disease dengue. The febrile clinical symptoms associated with dengue are similar to those of other arboviruses from other families; this has resulted in confusion in the diagnosis of dengue. In particular, the high incidence of dengue has resulted in the misdiagnosis of some arbovirus outbreaks as discussed above. Serological studies are needed to differentiate these.
DENV are thought to have originated in Asia in a sylvatic cycle involving arboreal mosquitoes and monkeys. However, endemic viruses have evolved and are now maintained in nature by a cycle involving man as both reservoir and amplification host, and domestic mosquitoes, principally Ae. aegypti, as vectors.
Dengue virus has caused numerous outbreaks throughout the south-west Pacific region since it was first recognized among servicemen during the Second World War. Subsequently, in the south-west Pacific, multiple dengue viruses have affected Tahiti, with haemorrhagic dengue first encountered in 1971. In addition to Ae. aegypti other species have been implicated as vectors, including Ae. scutellaris hebrideus in New Guinea, Ae. polynesiensis in Tahiti and Ae. cooki in Niue. Hawaii was dengue-free from 1944 to 2001 until, in 2001–2002, there was a DENV-1 outbreak involving at least 122 cases and Ae. albopictus as the vector.
DENV activity continues in many tropical countries of the Pacific rim, including northern Australia. Most South-East Asian countries, including Indonesia, Malaysia, Thailand, Vietnam, China, the Philippines and India, experience repeated epidemics of dengue caused by all four viruses, with most cases occurring between June and November. Mostly children are affected; in some outbreaks up to 25% may develop haemorrhagic fever. Most epidemics occur in urban areas and villages where Ae. aegypti is abundant, but not in rural environments. Up to 100 million infections may occur each year, mostly in children; this is a major public health problem.
Dengue is endemic throughout tropical Africa, including Nigeria in the west and Mozambique in the east, and also in Middle Eastern countries such as Saudi Arabia. Dengue disease is seldom severe in Africa and dengue haemorrhagic fever is rare.
Caribbean countries have been involved in epidemic waves of dengue since 1827, with little evidence of clinical dengue during interepidemic periods. All four DENV have been implicated in outbreaks affecting residents of Caribbean islands and adjacent areas of Central and South America. Each year cases of dengue are imported into continental USA and Europe following visits to dengue-endemic countries. Although most infections in the USA occur among travellers returning from the Caribbean, small numbers of cases (less than 30 per year) of indigenous dengue occur regularly among residents of southern Texas who live near the border with Mexico. The semi-tropical climate in this area allows Ae. aegypti to flourish during the nine warmer months of each year. Recently, a small epidemic was detected in the Florida Keys, another subtropical area inhabited by Ae. aegypti. In 1985, Ae. albopictus was introduced from Asia into the Americas via used motor tyre casings that had been imported for retreading into Texas, from Japan, South Korea and several South-East Asian countries. The mosquito rapidly established itself in Texas and spread into mid-western, north-eastern and north-western states of the USA, and also into Mexico and other countries. A peri-domestic mosquito, Ae. albopictus, was first identified as a dengue vector in Malaysia during the 1960s, and it transmits dengue both in the human–mosquito cycle and by transovarial transfer. However, to date, there have been few reports of dengue virus transmission in the Americas by Ae. albopictus.
Zika virus
Zika virus (ZIKV) is a flavivirus related to yellow fever virus that until recently has been considered endemic to Africa and Southeast Asia, but human cases are rare. In 2007 ZIKV caused an outbreak of 49 confirmed, and 59 probable cases, of a relatively mild disease characterized by rash, arthralgia, and conjunctivitis on Yap Island, Federated States of Micronesia, in the southwestern Pacific Ocean. Serologic studies suggest that 73% of Yap Island residents aged 3 or older had been recently infected with ZIKV. This was the first time that ZIKV was detected outside of Africa and Asia. The emergence of ZIKV outside of its previously known geographic range demonstrates another example of an emerging viral disease.
Flaviviruses: tick-borne
Powassan virus
Powassan virus, the sole North American tick-borne flavivirus, was first isolated from the brain of a fatal human case of encephalitis in Powassan, Ontario, Canada. To date, it has caused approximately 40 cases of encephalitis and four deaths among residents of forested areas of Ontario, Quebec and Nova Scotia in Canada, as well as Massachusetts, New York State and Pennsylvania in the USA. All of these cases occurred between May and October. Principal tick vector species in Ontario are Ixodes cookei, which feeds on groundhogs, and I. marxi, which feeds on tree squirrels; both of these mammals serve as reservoirs.
Tick-borne encephalitis viruses
Tick-borne encephalitis virus (TBEV) is used to describe a serocomplex of related viruses that are transmitted by ticks and cause similar diseases. These include central European (also known as the western subtype) TBEV that causes central European encephalitis and Russian spring–summer encephalitis (also known as the Far Eastern subtype of TBEV). In addition, a third subtype, Siberian TBEV, has been described mainly on the basis of genetic studies. Russian spring–summer encephalitis occurs in eastern Europe and parts of Asia, including northern Japan, and causes a more severe disease than the central European encephalitis found in western Europe and Scandinavia. The case fatality rate of central European encephalitis is usually below 10%, whereas it can reach 30% for Russian spring–summer encephalitis. In Britain and Ireland and some parts of France and Scandinavia, a mild form of tick-borne encephalitis is caused by a related virus called ‘louping ill’. The latter virus also infects sheep and grouse and gets its name from the leaping gait in infected sheep; the virus rarely infects man.
Human infections with TBEV may range in severity from mild biphasic meningo-encephalitis, which is characteristic of the central European TBEV and louping ill virus, to a severe form of polio-encephalomyelitis that is characteristic of Russian spring–summer encephalitis virus.
Natural cycles involve I. ricinus ticks as vectors, and mice, shrews and other small rodents as reservoirs, with infection transferred tangentially to sheep or other farm animals, and also to human beings. In Siberia, I. persulcatus ticks serve as vectors. Recently, additional viruses have been described from Spain, Turkey and Bulgaria that are related to louping ill virus and are members of the TBEV complex and cause TBEV-like disease symptoms in sheep and other animals.
The TBEV complex contains three viruses associated with haemorrhagic fever: Omsk haemorrhagic fever (OHF) and Kyasanur Forest disease (KFD) and Alkhumra viruses. OHF was identified during the Second World War in Omsk and the virus causes occasional outbreaks in Russia, whereas KFD is found only in India and causes regular outbreaks of haemorrhagic fever. Alkhumra virus was isolated in 1995, and subsequently in 2001 in Saudia Arabia; it caused haemorrhagic fever with a fatal outcome in 25% of patients. Genetic studies suggest that Alkhumra virus is a subtype of KFD virus.
Bunyaviruses: Bunyavirus genus
California (CAL) serogroup
There are at least 14 viruses related antigenically to the prototype California encephalitis virus. In the USA, encephalitis and aseptic meningitis arise commonly from infections with La Crosse virus, first isolated from the brain of a patient with fatal encephalitis in La Crosse, Wisconsin. Other viruses occasionally associated with aseptic meningitis are snowshoe hare virus, isolated from the blood of a snowshoe hare in Montana, and Jamestown Canyon virus (JCV), isolated from Culiseta inornata mosquitoes collected at Jamestown Canyon, Colorado. In central Europe, febrile illness, sometimes with aseptic meningitis, arises from infection with Tahyna virus, isolated from Ae. caspius mosquitoes collected near Tahyna, in the former Czechoslovakia.
Currently, CAL serogroup viruses are the most common arboviruses associated with encephalitis in the USA. The highest attack rates occur in states adjoining the Great Lakes, affecting mainly children aged less than 15 years. Abundant tree holes in wooded areas provide optimal breeding sites for the principal mosquito vector Ae. triseriatus, but rainwater collected in disused motor tyres is a suitable breeding ground for Ae. triseriatus in suburban locations. As adult mosquitoes die in winter, CAL serogroup viruses survive through transovarial transmission. Principal vertebrate reservoirs are tree squirrels and chipmunks.
Oropouche (ORO)
This virus is the only human pathogen in the Simbu serogroup; it was isolated in Trinidad in 1955. Outbreaks of oropouche fever, a febrile illness, have occurred in Brazil since 1961, involving urban transmission by the midge Culicoides paraensis. An outbreak was reported in Panama in 1989, and cases of oropouche fever have been reported in Peru since 1992.
Garissa virus
In 1997–1998 Garrisa virus was described as a reassortant bunyavirus. It was isolated from patients with acute haemorrhagic fever during an epidemic of Rift Valley fever in Kenya and southern Somalia. Acute sera from patients with haemorrhagic fever yielded either virus isolation or PCR evidence of infection. Initial studies indicated that the sequences of the L and S RNA segments were nearly identical to those of Bunyamwera virus, whereas the sequence of the M segment was very different (33% nucleotide and 28% amino acid differences). Very recent studies have shown that Garissa and Ngari virus M segments are nearly identical in sequence, whereas the L and S segments of Bunyamwera virus are similar to those of both Ngari and Garissa virus. These data indicate that Garissa virus is not a reassortant but an isolate of Ngari virus, which is a reassortant of Bunyamwera virus. Previously Ngari virus had not been considered a cause of haemorrhagic fever; further studies are required to investigate the pathogenesis and epidemiology of Ngari virus.
Bunyaviruses: Phlebovirus genus
Rift Valley fever virus
Rift Valley fever virus (RVFV) was first isolated in 1930 from sheep during an epizootic causing abortion and death in the Rift Valley near Lake Niavasha, Kenya, but is present from South Africa to Egypt. The virus infects many large domestic animals and a wide variety of mosquito species, with sheep, cattle, buffaloes and rodents as reservoirs. RVFV is epizootic with long inter-epizootic periods. Outbreaks of Rift Valley fever occurred in Egypt during 1977 involving an estimated 200 000 human cases and 600 deaths. Subsequently, Rift Valley fever has been reported during 1998–1999 in Mauritania and Senegal, and in Yemen and south-west Saudi Arabia in 2000–2001, involving over 2000 clinical cases and a mortality rate of 14%. Genetic studies of the virus are consistent with the theory that infection came from East Africa.
Sandfly fever group
These viruses are distributed throughout the European and North African countries surrounding the Mediterranean Sea, extending eastward through Israel and Iran to West Pakistan and central India. Sandfly fever (Naples and Sicilian) viruses were first isolated from the sera of US servicemen during an outbreak in the Second World War.
Epidemics of dengue-like fever occur during the sandfly season (June to September) and affect mainly visitors rather than residents. Natural vectors are Phlebotomus papatasi and other phlebotomine sandflies. Isolation of virus from male sandflies collected during July suggests transovarial transfer of virus. The natural cycle of sandfly fever appears to involve solely human beings, as definitive host and reservoir, with sandflies as vectors.
Bunyaviruses: Nairovirus genus
Crimean–Congo haemorrhagic fever virus
Crimean–Congo haemorrhagic fever virus (CCHFV) was originally described as two separate viruses: Crimean haemorrhagic fever virus, isolated from the serum of a man with fatal haemorrhagic fever near Samarkand, Uzbekistan, and Congo virus isolated from the serum of a child with fever and arthralgia in Zaire. Antigenic and genetic studies show that the two viruses are identical. CCHFV is distributed widely throughout tropical Africa, from Mauritania to Uganda and Kenya, the Middle East and West Pakistan, and southwards to South Africa. It is also found in Asia, including parts of China. The geographical distribution of CCHFV corresponds to that of Hyalomma sp. ticks, from which the virus can be isolated. Human infection is rare but mortality rates of up to 50% have been reported.
Bunyaviruses: Hantavirus genus
Unlike the other genera in the Bunyaviridae, members of the Hantavirus genus are rodent-associated viruses. They are zoonotic viruses of rodents (mainly mice or voles) that excrete virus in urine for prolonged periods. Virus is transmitted to man by contact with aerosols of rodent urine.
Hantaan and Puumala viruses
These viruses induce either (1) a severe illness, termed haemorrhagic fever with renal syndrome, due to Hantaan virus in Japan, Korea, China and Siberia and Puumala virus in Scandinavia, or (2) a mild illness, termed nephropathia epidemica, due to Puumala virus in Scotland, France, Belgium and Germany, the Balkans and Greece. Hantaan virus was first identified in soldiers serving in Korea in 1951 and named after the area where it was detected. Principal vertebrate reservoirs comprise Apodemus agrarius rodents in Asia and Clethrionomys glareolus (bank vole) in Europe.
Sin Nombre virus
Sin Nombre virus induces hantavirus pulmonary syndrome, a severe acute respiratory illness with a case fatality rate exceeding 50%. Initially encountered in May 1993 in the Four Corners region of the USA (Arizona, Colorado, New Mexico and Utah), cases have since occurred elsewhere in the USA, Canada and many countries in South America. The principal rodent reservoir is considered to be Peromyscus maniculatus (deer mouse), but each hantavirus pulmonary syndrome-causing virus occupies a geographical region defined by the rodent species that carries it. Thus, Black Creek Canal virus found in cotton rats (Sigmadon hispidus) in Florida is related to viruses found in South America, with a distinct rodent host.
This was the first occasion on which molecular methods identified an unknown arbovirus before the virus was isolated. Hantavirus group antibodies were detected in sera from human cases and rodents. RNA from lung and liver of fatal human cases and seropositive rodents was amplified by RT-PCR; positive bands were detected using primers for Prospect Hill (North American) and Puumala virus-like hantaviruses, but not Hantaan and Seoul-like (Asian) hantaviruses. This was achieved within 3 weeks after death of the initial human cases. Several months later, Sin Nombre virus, genomically distinct from Prospect Hill and other hantaviruses, was isolated from tissue suspensions, initially from P. maniculatus and subsequently from man.
Control
Strategies for the prevention of arbovirus infections depend on either vector control or active immunization with vaccine.
Vector control
Suppression of populations of vector mosquito species can halt virus transmission in urban and suburban localities as follows:
1. Use of insecticides to kill adult mosquitoes (‘adulticiding’), for instance aerial sprays of malathion; however, nontarget insects may also be affected. The use of persistent insecticides on the interiors of houses has decreased due to environmental concerns.
2. Removal of domestic larval development sites for Ae. aegypti such as tin cans and tyres that could contain rainwater, both near human habitations and in public parks and drainage systems; this has prevented the occurrence of dengue in Singapore and urban yellow fever in metropolitan areas in Caribbean countries.
3. Chemical control of larvae (barricading) by applying insecticides or oil to small larval sites; this has reduced mosquito vector populations substantially in irrigated localities in California and elsewhere.
4. Biological control of larvae by microbiological agents such as Bacillus thuringiensis israeliensis, larvivorous fish, flatworms or mermethid nematodes, or insect growth regulators such as the juvenile hormone that mimics methoprene.
5. Personal protection against bites by mosquitoes involving a combination of protective clothing, preferably impregnated with permethrin, screening of dwellings to prevent entry of vectors and frequent application of repellants such as N,N-diethyl-m-toluamide (DEET) or picaridin to exposed skin. For tick-borne viruses, protective clothing should be worn outdoors, followed by rigorous inspection to remove attached ticks from the skin.
Vaccines
To date relatively few vaccines have been developed to control arbovirus diseases.
Alphaviruses
There are no licensed vaccines for use in man. However, formalin-inactivated vaccines for WEEV, EEEV and VEEV are used to immunize horses, researchers who work with the viruses, and military personnel. A live VEE vaccine, known as TC-83, is also administered to horses and researchers. TC-83 is associated with significant adverse reactions and lack of seroconversion in many human vaccinees, and is not suitable for use in the general population.
Flaviviruses
Vaccines have been licensed against yellow fever, Japanese encephalitis and TBE viruses. A live yellow fever vaccine, 17D, was developed in the 1930s by 176 passages of wild-type strain Asibi through chicken tissue. One dose of vaccine administered subcutaneously and containing 5000–200 000 plaque-forming units of virus gives protective immunity 10 days after immunization; immunity lasts for at least 10 years. The World Health Organization recommends immunization every 10 years to maintain immunity; the vaccine can be given to children over 9 months of age. Immunization is contra-indicated in immunocompromised individuals and pregnant women. More than 550 million doses of vaccine have been administered, leading to only a few cases of yellow fever associated neurotropic disease (YEL-AND) and yellow fever associated viscerotropic disease (YEL-AVD).
Both live and killed vaccines have been developed to control Japanese encephalitis. Formalin-inactivated vaccines were developed in the 1940s based on virus grown in mouse brain but were recently discontinued due to the risk of allergic reaction following immunization. Killed vaccines based on virus grown in Vero cell culture are now being used. Two doses of killed vaccine given 7–28 days apart are required for protective immunity. A booster is given at 1 year and subsequently every 3–4 years to maintain immunity. A live vaccine has been developed: SA14-14-2 was generated in the People’s Republic of China by 126 passages of wild-type strain SA14 in primary hamster kidney cell culture. It is given as two doses and has been administered to more than 300 million people in China without any reports of adverse reactions. To date the vaccine has been used in seven other Asian countries. In addition, a live vaccine is used to immunize pigs.
A formalin-inactivated TBE vaccine using virus grown in chicken eggs was developed in the 1970s to control central European tick-borne encephalitis virus. The vaccine was improved by transferring manufacture to primary chick embryo fibroblast cell culture. Two doses are given, 2 weeks to 3 months apart, followed by a booster given 9 months to 1 year later. Boosters are recommended every 3 years. The vaccine has proved to be very effective, with few adverse reactions, and has resulted in the near elimination of tick-borne encephalitis in Austria. The effectiveness of the vaccine against Russian spring–summer encephalitis is uncertain.
There are no vaccines available to prevent dengue. However, a number of candidate live vaccines are currently undergoing human trials.
Bunyaviruses
Although there are no commercially available vaccines against diseases caused by bunyaviruses, a number of experimental live and killed vaccines have been developed against Rift Valley fever. In addition, experimental deoxyribonucleic acid (DNA)-based vaccines have been developed against several hantaviruses and show promise in preclinical studies.
Barrett ADT. Japanese encephalitis. In: Service MW, ed. The Encyclopedia of Arthropod-Transmitted Infections. Wallingford, UK: CAB International; 2001:239–246.
Elliott RM, ed. The Bunyaviridae. New York: Plenum Press, 1996.
Karabatsos N. International Catalogue of Arboviruses Including Certain Other Viruses of Vertebrates, ed 3. San Antonio: American Society for Tropical Medicine and Hygiene; 1985.
Ksiazek TG, Peters CJ, Rollin PE, et al. Identification of a new North American hantavirus that causes acute pulmonary insufficiency. American Journal of Tropical Medicine and Hygiene. 1995;52:117–123.
Monath TP, ed. The Arboviruses: Ecology and Epidemiology, Vols 1–5. Boca Raton: CRC Press, 1988.
US Department of Health and Human Services. Biosafety in Microbiological and Biomedical Laboratories, ed 5. Washington, DC: US Government Printing Office; 2007.
Weaver SC. Eastern equine encephalitis. In: Service MW, ed. The Encyclopedia of Arthropod-Transmitted Infections. Wallingford, UK: CAB International; 2001:151–159.
Weaver SC, Barrett AD. Transmission cycles, host range, evolution and emergence of arboviral disease. Nature Reviews Microbiology. 2004;2:789–801.
Weaver SC, Ferro C, Barrera R, et al. Venezuelan equine encephalitis. Annual Reviews of Entomology. 2001;49:141–174.
Weaver SC, Reisen WK. Present and future arboviral threats. Antiviral Research. 2009;85:328–345.